Anesthesia: Essays and Researches
|
|
|
- Andra Barton
- 6 years ago
- Views:
Transcription
1 A E R Editor-in-Chief : Mohamad Said Maani Takrouri (KSA) Open Access HTML Format For entire Editorial Board visit : Anesthesia: Essays and Researches Review Article Sugammadex: A revolutionary drug in neuromuscular pharmacology Kusha Nag, Dewan Roshan Singh, Akshaya N. Shetti 1, Hemanth Kumar, T. Sivashanmugam, S. Parthasarathy Department of Anaesthesiology and Critical Care, Mahatma Gandhi Medical College, Pillaiyarkuppam, Puducherry, 1 Department of Anaesthesiology, Krishna Institute of Medical Sciences, Karad, Maharashtra, India Corresponding author: Dr. Dewan Roshan Singh, Department of Anaesthesiology and Critical Care, Mahatma Gandhi Medical College, Pillaiyarkuppam, Puducherry , India. droshansingh@yahoo.com Abstract Sugammadex (ORG 25969) is a unique neuromuscular reversal drug; a novel cyclodextrin, the fi rst in a new class of selective relaxant binding agents, which reverse neuromuscular blockade (NMB) with the aminosteroid non-depolarizing muscle relaxants rocuronium and vecuronium. Sugammadex can reverse moderate or deep NMB. The clinical use of sugammadex promises to eliminate many of the shortcomings in current anesthetic practice with regard to antagonism of rocuronium and other aminosteroid muscle relaxants. Key words: Encapsulation, residual paralysis, rocuronium, selective relaxant binding agent, sugammadex INTRODUCTION Muscle relaxants are used during intraoperative period to facilitate endotracheal intubation, ensure patient immobility and improve surgical exposure. [1] Reversal agents are used to terminate the action of muscle relaxants. The ideal reversal agent should have a fast onset; should be efficient at any time, even soon after paralyzing the patient; it should be able to provide complete reversal either for light or profound blockade; it should have a longer half-life than the muscle relaxants and it should not have any side-effects. The ideal reversal Access this article online Website DOI Quick Response Code / agent is yet to be found, but sugammadex is very close to being an ideal reversal agent. [2] Sugammadex is a synthetic cyclodextrin molecule, which acts by encapsulation of rocuronium. [3-6] The discovery of encapsulation for reversal of aminosteroid muscle relaxants is the result of work done by Bom, a research scientist at Organon International (Oss, Netherlands). [7-9] Literature search for the article was carried out using Google Scholar, Proquest database and on wikepedia using the term Sugammadex. CHEMICAL STRUCTURE Sugammadex [6A,6B,6C,6D,6E,6F,6G,6H-octakis-S- (2-carboxyethyl)-6A,6B,6C,6D,6E,6F,6G,6Hoctathio-γ-cyclodextrin octasodium salt] is a modified γ-cyclodextrin [Figure 1]. Chemical formula is C 72 H 104 Na 8 O 48 S 8. Su stands for sugar and gammadex stands for structural molecule gamma-cyclodextrin. [1] Cyclodextrins are cyclic dextrose units joined through 1-4 glycosyl bonds that are produced from starch or 302
2 starch derivates using cyclodextrin glycosyltransferase. The three natural unmodified cyclodextrins are α-, β-or γ-cyclodextrin. Compared with α-and β-cyclodextrins, γ-cyclodextrin exhibits more favorable properties in terms of the size of its internal cavity, water solubility and bioavailability. To have a better fit of the larger rigid structure of the aminosteroid muscle relaxant molecule (e.g., rocuronium or vecuronium) within the cavity of γ-cyclodextrin, the latter was modified by adding eight side chains to extend the cavity. This modification allowed the four hydrophobic steroidal rings of rocuronium to be better accommodated within the hydrophobic cavity. In addition, adding negatively charged carboxyl groups at the end of the eight side chains served two purposes. First, the repellent forces of the negative charges keep propionic acid side chains from being disordered, thereby allowing the cavity to remain open and maintain structural integrity. Second, adding these negatively charged carboxyl groups enhances electrostatic binding to the positively charged quaternary nitrogen of rocuronium [Figure 1]. Three-dimensional structure resembles a hollow, truncated cone or a doughnut. [9] Mechanism of action Inactivates rocuronium by encapsulating (chelating) the free molecule to form a stable complex. [10] The structure has a hydrophobic cavity and a hydrophilic exterior because of the presence of polar hydroxyl groups. Hydrophobic interactions trap the drug into the cyclodextrin cavity, resulting in the formation of a water-soluble guest host complex. Sugammadex exerts its effect by forming very tight complexes at a 1:1 ratio with aminosteroid muscle relaxants (rocuronium > vecuronium >> pancuronium). [9-12] The intermolecular (van der Waals ) forces, thermodynamic (hydrogen) bonds and hydrophobic interactions make the sugammadex rocuronium complex very tight. The sugammadex rocuronium complex has a very high association rate (an association constant of 107 M-1) and a very low dissociation rate. It is estimated that, for every 25 million sugammadex rocuronium complexes, only one complex dissociates. The resulting reduction in free rocuronium plasma concentration creates a gradient between the tissue compartment (including the NMJ) and plasma; free rocuronium moves from tissue to plasma, with a reduction in nicotinic receptor occupancy at the NMJ. [10,13,14] Distribution The volume of distribution of sugammadex is approximately 18 L and volume of distribution at steady state is L ( L/kg). The drug does not bind to plasma proteins or red blood cells. [2] Metabolism and elimination The drug does not produce any metabolites and is mostly excreted through urine in the unchanged form within 24 h. An insignificant part is excreted through feces or in expired air (0.02%). The plasma clearance of the drug ranges from 84 to 138 ml/min. [2] Dosage and formulation Table 1: Recommended dosage of sugammadex Age NMB drug Rocuronium/ vecuronium Rocuronium/ vecuronium Type of reversal Routine (for moderate blockade) Routine (for deep blockade) Dosage (mg/kg) a 2 mg/kg b 4 mg/kg c 18 Rocuronium Immediate 16 mg/kg 2-17 Rocuronium Routine (for moderate blockade) 2 mg/kg a The most appropriate dose in dependent on the level of NMB to be reversed and is independent of the anesthetic regimen; b Recommended if spontaneous recovery has occurred, up to the reappearance of T2 (second twitch in the TOF stimulation) following NMB; c Recommended if recovery has reached at least 1-2 post-tetanic counts following neuromuscular blockade. [15-18] TOF = Train of four; NMB = Neuromuscular blockade Figure 1: Structure of sugammadex Minimum effective reversal dose: 2 mg/kg IV. [12] Administration: The drug should be given as a single IV bolus, delivered within 10 s. [13] The drug is available in IV formulation only; vials of 200 mg (2 ml) or 500 mg (5 ml) and is for non-refrigerated storage. [19] The drug was originally designated as Org 25,969. Trade name is Bridion. Sugammadex was discovered at the Newhouse research site in Scotland. Scientists who discovered Sugammadex worked for the pharmaceutical company, Organon. Organon was acquired by Schering-Plough in 2007; Schering-Plough merged with Merck in Sugammadex is now owned and sold by Merck. On January 3, 2008, Schering-Plough submitted a New Drug 303
3 Application to the US FDA for sugammadex, but was rejected on August [20,21] It was approved for use in the European Union on July 29, [17] The drug is also approved for use in Australia, Iceland, New Zealand and Norway. Indication Reversal of neuromuscular blockade (NMB) in the operation theater and in the intensive care unit (ICU). [17] Drug interactions Toremifene, a chemotherapeutic drug, which is a selective estrogen receptor modulator, can delay the reversal by displacing the formation of the rocuronium-sugammadex complex. The drug can theoretically bind with contraceptive steroids. As endogenous steroids and steroidal drugs lack the quaternary nitrogen of the aminosteroid muscle relaxants, they show a much lower affinity. Rocuronium-sugammadex complex can also be displaced by fusidic acid or high doses of flucloxacillin. Drugs like magnesium, aminoglycosides potentiate muscle relaxants; to counteract these effects, a larger dose of sugammadex may be required. Among anesthetic drugs the highest affinity constant was observed with remifentanil (0.2% of the affinity constant of sugammadex with rocuronium); it has not been shown to be clinically significant. Physical incompatibility has been reported with verapamil, ondansetron and ranitidine. Further investigations are required to assess other possible drug interactions. [1,2,9] Sugammadex does not alter laboratory tests in general, but some modification of the serum progesterone assay and of the coagulation parameters like activated partial thromboplastin time, prothrombin time, INR has been noticed. [17] Contraindications Hypersensitivity to sugammadex or to any excipients. [13] Special considerations Patients with severe renal impairment: Clearance of the drug is reduced in patients with creatinine clearance below 30 ml/min. The efficacy of the drug is not reduced in these patients. [22] There are conflicting reports of its safety in these subset of patients; hence, it is recommended to avoid its use in such patients. [2] Patients with hepatic impairment: Sugammadex-rocuronium complex is not eliminated by hepatic metabolism; therefore, hepatic impairment does not affect its excretion. However, it is advised to be cautious regarding its use in these patients. [2] Obese patients: Use adult dose based on actual body weight. Patients in ICU: Sugammadex has not been investigated in patients receiving rocuronium or vecuronium in the ICU. Elderly: Use adult dose although recovery times are slower. Pediatric patients: Pharmacokinetic profile is quite similar to that of adults. There is no clinical trial in children below 2 years of age. Pregnancy and lactation: Caution in pregnant women. Sugammadex can be used during lactation. Sex, race and body weight: They have no effect on the use of sugammadex. Recurarization A phenomenon of recurrence of NMB may occur where the reversal agents wear off before a muscle relaxant is completely cleared. It has been demonstrated to occur only rarely with sugammadex and only when insufficient doses were administered. The underlying mechanism is thought to be related to redistribution of relaxant after reversal. It may occur for a limited range of sugammadex doses, which are sufficient for complex formation with relaxant in the central compartment, but insufficient for additional relaxant returning to the central compartment from peripheral compartments. [16-19] COMPARISON WITH NEOSTIGMINE Sugammadex acts by encapsulation of rocuronium, whereas neostigmine is an anticholinesterase. Neostigmine has a ceiling effect and when given at a deep level of NMB, can result in inadequate reversal. Neostigmine is also associated with adverse effects like autonomic disturbances including bradycardia, which can be particularly troublesome during extubation in patients for cardiothoracic surgery, nausea and vomiting; these adverse effects are not noticed with sugammadex. [9,15,18,23-25] LIMITATIONS The drug is expensive. The cost of reversal of NMB with sugammadex in a 75 kg person is for 2 mg/kg, for 4 mg/kg and for 16 mg/kg compared to 0.71 for spontaneous recovery from suxamethonium. [26] However, studies have shown that use of sugammadex can be cost-effective by reduced incidence of post-operative residual paralysis reducing operative time, post-operative recovery stay and also decreased side-effects associated with the use of suxamethonium. Adverse effects Sugammadex is a well-tolerated and relatively free of adverse effects. Some of the adverse reported in some of the trials are dysgeusia (metal or bitter taste) mainly seen after doses of 32 mg/kg or higher, recurrence of block, movement of limbs or body or coughing during anesthesia, coughing, grimacing or suckling on the endotracheal tube, procedural hypotension. [17,27,28] 304
4 Precautions If re-administration of rocuronium or vecuronium is required a waiting time of 24 h is recommended. If NMB is required before the recommended waiting time has passed, a non-steroidal muscle relaxant should be used. Sugammadex should not be used to reverse block induced by non-steroidal blockers such as suxamethonium or benzylisoquinolinium compounds (because it cannot form inclusion complexes with these drugs) or aminosteroid muscle relaxants other than rocuronium or vecuronium. [9,16] ADVANTAGES 1. Use of sugammadex will decrease the incidence of residual muscle paralysis and hence improve patient safety [29] 2. Anesthesiologists will not hesitate to give incremental doses of muscle relaxant if required towards the end of surgery 3. High dose rocuronium for rapid sequence induction, i.e., 1.2 mg/kg can be used safely in cases of short duration. 4. In case of need to rapidly terminate the effect of muscle relaxants, i.e., in cannot intubate cannot ventilate scenario, rocuronium would be preferred to suxamethonium 5. No additional anticholinestrases or anticholinergics would be needed for reversal of residual NMB, thus avoiding unwanted cardiovascular effects and other adverse effects of the drugs. [24] WILL SUGAMMADEX BRING A FINAL DEMISE OF SUXAMETHONIUM? Use of suxamethonium has declined in modern anesthetic practice. Suxamethonium has numerous adverse effects such as fasciculations, myalgia, hyperkalemia, heart rate changes and increased intragastric and intraocular pressures, malignant hyperthermia. Use of suxamethonium in children is not preferred due to the danger of hyperkalemic cardiac arrest in children with unrecognized muscular dystrophy. [30] Suxamethonium is still used in difficult airway scenario, full stomach and in cases under general anesthesia of short duration. If sugammadex becomes widely available, rocuronium can be used as a better alternative in the above situations. POTENTIAL IMPACT OF SUGAMMADEX ON DAY TO DAY ANESTHETIC PRACTICE a. Sugammadex cannot reverse NMB with benzylisoquinolinium group of muscle relaxants or with suxamethonium b. After sugammadex administration, reblock with rocuronium will be difficult and the dose required will be unpredictable. Benzylisoquinolinium group of muscle relaxants or suxamethonium should be used in such a scenario c. Sugammadex does not have equal affinity with all aminosteroid relaxants. Reversal of pancuronium will require large doses of sugammadex d. It is necessary to know the extent of NMB before sugammadex administration. Train of four (TOF) count or post tetanic count (PTC) may be sufficient to decide the dosage of sugammadex. Reversal of profound block with inadequate doses of sugammadex will result in incomplete reversal. TOF ratio will give an objective assessment to help determine if antagonism of residual block is actually required. [31,32] CONCLUSION Sugammadex may potentially promote the increased use of non-depolarizing aminosteroid muscle relaxants, but economic constraints may prevent its wide spread use and poses great limitations to its use. [33] It will prove useful for very short surgeries when an intermediate acting NMB is used or when the dose, timing or administration of neostigmine is inadequate. Surgeons should no longer encounter inadequate muscle relaxation and anesthesiologists should no longer encounter patients whose NMB is difficult to reverse at the end of surgery. [34] It should eliminate the occurrence of postoperative residual NMB. Assurance of no residual paralysis may enable anesthesiologists to better evaluate the success or failure when attempting to wean and extubate patients who have received aminosteroid muscle relaxants. Side-effects like nausea/vomiting or unwanted autonomic adverse effects encountered with neostigmine will be eliminated. [35] Improvements in patient outcome must have a dollar value that offsets the cost of the drug. FDA has still not approved the use of sugammadex due to some concerns regarding allergic reactions. The drug is being extensively used in the EU and Australia at present. Sugammadex should not replace good clinical practice titration of muscle relaxants to clinical needs and objective monitoring of NMB in the OT or ICU. [36] The introduction of propofol two decades ago changed anesthetic practice, no other drug has had the same impact. Undoubtedly, sugammadex is a milestone and is likely to change the face of clinical neuromuscular pharmacology. REFERENCES 1. Kovac AL. Sugammadex: The fi rst selective binding reversal agent for neuromuscular block. J Clin Anesth 2009;21: Hemmerling TM, Zaouter C, Geldner G, Nauheimer D. Sugammadex: A short review and clinical recommendations for the cardiac anesthesiologist. Ann Card Anaesth 2010;13: Bhaskar SB. Emergence from anaesthesia: Have we got it all smoothened out? Indian J Anaesth 2013;57: Lee C, Jahr JS, Candiotti KA, Warriner B, Zornow MH, Naguib M. Reversal of profound neuromuscular block by sugammadex administered three minutes after rocuronium: A comparison with spontaneous recovery from succinylcholine. Anesthesiology 2009;110:
5 5. Gijsenbergh F, Ramael S, Houwing N, van Iersel T. First human exposure of Org 25969, a novel agent to reverse the action of rocuronium bromide. Anesthesiology 2005;103: Nicholson WT, Sprung J, Jankowski CJ. Sugammadex: A novel agent for the reversal of neuromuscular blockade. Pharmacotherapy 2007;27: Cameron KS, Fletcher L, Clark JK, Zhang MQ, Orbons LPM. Chemical chelation as a novel method of NMB reversal characterization of the Org NMB complex: ;18: Zhang MQ. Drug specifi c cyclodextrins: The future of rapid neuro-muscular block reversal? Drugs Future 2003;28: Naguib M. Sugammadex: Another milestone in clinical neuromuscular pharmacology. Anesth Analg 2007;104: Bom A, Bradley M, Cameron K, Clark JK, Van Egmond J, Feilden H, et al. A novel concept of reversing neuromuscular block: Chemical encapsulation of rocuronium bromide by a cyclodextrin-based synthetic host. Angew Chem Int Ed Engl 2002;41: Adam JM, Bennett DJ, Bom A, Clark JK, Feilden H, Hutchinson EJ, et al. Cyclodextrin-derived host molecules as reversal agents for the neuromuscular blocker rocuronium bromide: Synthesis and structure-activity relationships. J Med Chem 2002;45: Tarver GJ, Grove SJ, Buchanan K, Bom A, Cooke A, Rutherford SJ, et al. 2-O-substituted cyclodextrins as reversal agents for the neuromuscular blocker rocuronium bromide. Bioorg Med Chem 2002;10: Cameron KS, Clark JK, Cooper A, Fielding L, Palin R, Rutherford SJ, et al. Modifi ed gamma-cyclodextrins and their rocuronium complexes. Org Lett 2002;4: Epemolu O, Bom A, Hope F, Mason R. Reversal of neuromuscular blockade and simultaneous increase in plasma rocuronium concentration after the intravenous infusion of the novel reversal agent Org Anesthesiology 2003;99: Sparr HJ, Vermeyen KM, Beaufort AM, Rietbergen H, Proost JH, Saldien V, et al. Early reversal of profound rocuronium-induced neuromuscular blockade by sugammadex in a randomized multicenter study: Effi cacy, safety, and pharmacokinetics. Anesthesiology 2007;106: Yang LP, Keam SJ. Sugammadex: A review of its use in anaesthetic practice. Drugs 2009;69: BRIDION(R) (Sugammadex) Injection-First and Only Selective Relaxant Binding Agent-Approved in European Union. Schering-Plough, Sacan O, White PF, Tufanogullari B, Klein K. Sugammadex reversal of rocuronium-induced neuromuscular blockade: A comparison with neostigmine-glycopyrrolate and edrophonium-atropine. Anesth Analg 2007;104: Kovac AL. Sugammadex: A selective relaxant binding agent for neuromuscular block reversal. Formulary 2009;44: US FDA Issues Action Letter for Sugammadex. Schering-Plough (press release), Naguib M, Mohamed, Brull, Sorin J. Update on neuromuscular pharmacology. Curr Opin Anaesthesiol 2009;26: Staals LM, Snoeck MM, Driessen JJ, Flockton EA, Heeringa M, Hunter JM. Multicentre, parallel-group, comparative trial evaluating the effi cacy and safety of sugammadex in patients with end-stage renal failure or normal renal function. Br J Anaesth 2008;101: Dahl V, Pendeville PE, Hollmann MW, Heier T, Abels EA, Blobner M. Safety and effi cacy of sugammadex for the reversal of rocuronium-induced neuromuscular blockade in cardiac patients undergoing noncardiac surgery. Eur J Anaesthesiol 2009;26: Hemmerling TM, Russo G, Bracco D. Neuromuscular blockade in cardiac surgery: An update for clinicians. Ann Card Anaesth 2008;11: Jones RK, Caldwell JE, Brull SJ, Soto RG. Reversal of profound rocuronium-induced blockade with sugammadex: A randomized comparison with neostigmine. Anesthesiology 2008;109: Chambers D, Paulden M, Paton F, Heirs M, Duffy S, Hunter JM, et al. Sugammadex for reversal of neuromuscular block after rapid sequence intubation: A systematic review and economic assessment. Br J Anaesth 2010;105: Bajaj P. Reversal by sugammadex. Indian J Anaesth 2009;53: Groudine SB, Soto R, Lien C, Drover D, Roberts K. A randomized, dose-fi nding, phase II study of the selective relaxant binding drug, Sugammadex, capable of safely reversing profound rocuronium-induced neuromuscular block. Anesth Analg 2007;104: Abrishami A, Ho J, Wong J, Yin L, Chung F. Cochrane corner: Sugammadex, a selective reversal medication for preventing postoperative residual neuromuscular blockade. Anesth Analg 2010;110: Miller RD. Sugammadex: An opportunity to change the practice of anesthesiology? Anesth Analg 2007;104: Suy K, Morias K, Cammu G, Hans P, van Duijnhoven WG, Heeringa M, et al. Effective reversal of moderate rocuronium- or vecuronium-induced neuromuscular block with sugammadex, a selective relaxant binding agent. Anesthesiology 2007;106: Eleveld DJ, Kuizenga K, Proost JH, Wierda JM. A temporary decrease in twitch response during reversal of rocuronium-induced muscle relaxation with a small dose of sugammadex. Anesth Analg 2007;104: Ledowski T, Hillyard S, O Dea B, Archer R, Vilas-Boas F, Kyle B. Introduction of sugammadex as standard reversal agent: Impact on the incidence of residual neuromuscular blockade and postoperative patient outcome. Indian J Anaesth 2013;57: Tayal G, Kundra S, Grewal A. Sugammadex-New neuromuscular block reversal. J Anaesth Clin Pharmacol 2008;24: Naguib M, Magboul MM. Adverse effects of neuromuscular blockers and their antagonists. Drug Saf 1998;18: Duvaldestin P, Kuizenga K, Saldien V, Claudius C, Servin F, Klein J, et al. A randomized, dose-response study of sugammadex given for the reversal of deep rocuronium- or vecuronium-induced neuromuscular blockade under sevofl urane anesthesia. Anesth Analg 2010;110: How to cite this article: Nag K, Singh DR, Shetti AN, Kumar H, Sivashanmugam T, Parthasarathy S. Sugammadex: A revolutionary drug in neuromuscular pharmacology. Anesth Essays Res 2013;7:302-6 Source of Support: Nil, Conflict of Interest: None declared. 306
Anesthesia and Neuromuscular Blockade: A Guide for Hospital Pharmacists. Upon completion of this activity, participants will be better able to:
 Anesthesia and Neuromuscular Blockade: A Guide for Hospital Pharmacists EDUCATIONAL OBJECTIVES Upon completion of this activity, participants will be better able to: 1. Understand the use of neuromuscular
Anesthesia and Neuromuscular Blockade: A Guide for Hospital Pharmacists EDUCATIONAL OBJECTIVES Upon completion of this activity, participants will be better able to: 1. Understand the use of neuromuscular
Sugammadex: A Comprehensive Review of the Published Human Science, Including Renal Studies
 Sugammadex: A Comprehensive Review of the Published Human Science, Including Renal Studies Kelsey Martin, MD. CA3 Resident, Indiana University School of Medicine Department of Anesthesia Article Abstract
Sugammadex: A Comprehensive Review of the Published Human Science, Including Renal Studies Kelsey Martin, MD. CA3 Resident, Indiana University School of Medicine Department of Anesthesia Article Abstract
1. NAME OF THE MEDICINAL PRODUCT
 08-15 BRIDIO Solution for Injection ORGAO Brand of sugammadex FOR ITRAVEOUS IJECTIO 1. AME OF THE MEDICIAL PRODUCT Bridion 100 mg/ml solution for injection 2. QUALITATIVE AD QUATITATIVE COMPOSITIO 1 ml
08-15 BRIDIO Solution for Injection ORGAO Brand of sugammadex FOR ITRAVEOUS IJECTIO 1. AME OF THE MEDICIAL PRODUCT Bridion 100 mg/ml solution for injection 2. QUALITATIVE AD QUATITATIVE COMPOSITIO 1 ml
NEOSTIGMINE VERSUS SUGAMMADEX FOR REVERSAL OF NEUROMUSCULAR BLOCK
 EDITORIAL NEOSTIGMINE VERSUS SUGAMMADEX FOR REVERSAL OF NEUROMUSCULAR BLOCK Neostigmine is the classic acetylcholinesterase antagonist, which is widely used for reversal of neuromuscular block of all nondepolarising
EDITORIAL NEOSTIGMINE VERSUS SUGAMMADEX FOR REVERSAL OF NEUROMUSCULAR BLOCK Neostigmine is the classic acetylcholinesterase antagonist, which is widely used for reversal of neuromuscular block of all nondepolarising
The cholinesterase inhibitors, neostigmine and edrophonium,
 Reversal of Rocuronium-Induced Neuromuscular Blockade: A Comparison with Glycopyrrolate and Atropine Ozlem Sacan, MD Paul F. White, MD, PhD Burcu Tufanogullari, MD Kevin Klein, MD BACKGROUND: is a modified
Reversal of Rocuronium-Induced Neuromuscular Blockade: A Comparison with Glycopyrrolate and Atropine Ozlem Sacan, MD Paul F. White, MD, PhD Burcu Tufanogullari, MD Kevin Klein, MD BACKGROUND: is a modified
The Latest Approaches to Reversal of Neuromuscular Blocking Agents
 The Latest Approaches to Reversal of Neuromuscular Blocking Agents Janay Bailey, Pharm.D. Anesthesiology 2017; 126:173-90 Objectives Pharmacists Determine optimal paralytic choices in knowing if reversal
The Latest Approaches to Reversal of Neuromuscular Blocking Agents Janay Bailey, Pharm.D. Anesthesiology 2017; 126:173-90 Objectives Pharmacists Determine optimal paralytic choices in knowing if reversal
Sugammadex: A New Wheel Has Been Invented. Ashlee A. Wheeler, BSN, RN, SRNA
 Sugammadex: A New Wheel Has Been Invented Ashlee A. Wheeler, BSN, RN, SRNA Disclaimer Statement Conflict of Interest: None I have not received any compensation for this presentation. Objectives The participant
Sugammadex: A New Wheel Has Been Invented Ashlee A. Wheeler, BSN, RN, SRNA Disclaimer Statement Conflict of Interest: None I have not received any compensation for this presentation. Objectives The participant
Bridion 100 mg/ml Solution for injection Sugammadex
 Bridion 100 mg/ml Solution for injection Sugammadex 1. NAME OF THE MEDICINAL PRODUCT Bridion 100 mg/ml solution for injection 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 1 ml contains sugammadex sodium
Bridion 100 mg/ml Solution for injection Sugammadex 1. NAME OF THE MEDICINAL PRODUCT Bridion 100 mg/ml solution for injection 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 1 ml contains sugammadex sodium
British Journal of Anaesthesia 101 (4): (2008) doi: /bja/aen216 Advance Access publication July 23, 2008
 British Journal of Anaesthesia 101 (4): 492 7 (2008) doi:10.1093/bja/aen216 Advance Access publication July 23, 2008 Multicentre, parallel-group, comparative trial evaluating the efficacy and safety of
British Journal of Anaesthesia 101 (4): 492 7 (2008) doi:10.1093/bja/aen216 Advance Access publication July 23, 2008 Multicentre, parallel-group, comparative trial evaluating the efficacy and safety of
New York Science Journal 2017;10(6) Efficacy And Safety Of Sugammadex In Reversing Nmb (Rocuronium) In Adults
 Efficacy And Safety Of Sugammadex In Reversing Nmb (Rocuronium) In Adults Mahmoud Abd Elrahman El Sherbeny; Ehab Ahmed Abd Elrahman; Reda Khalil Kamal; Mohamed Ahmed Mohamed Abozena Anesthesia and ICU
Efficacy And Safety Of Sugammadex In Reversing Nmb (Rocuronium) In Adults Mahmoud Abd Elrahman El Sherbeny; Ehab Ahmed Abd Elrahman; Reda Khalil Kamal; Mohamed Ahmed Mohamed Abozena Anesthesia and ICU
TRANSPARENCY COMMITTEE OPINION. 21 January 2009
 The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 21 January 2009 BRIDION 100 mg/ml, solution for injection Box containing 10 x 2 ml bottles, CIP: 573 553-9 Box containing
The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 21 January 2009 BRIDION 100 mg/ml, solution for injection Box containing 10 x 2 ml bottles, CIP: 573 553-9 Box containing
Effects of dexamethasone on sugammadex reversal times of rocuronium: a systematic review protocol
 Effects of dexamethasone on sugammadex reversal times of rocuronium: a systematic review protocol Cassie R. Held Mackenzie D. Sullivan The Center for Translational Research: a Joanna Briggs Institute Center
Effects of dexamethasone on sugammadex reversal times of rocuronium: a systematic review protocol Cassie R. Held Mackenzie D. Sullivan The Center for Translational Research: a Joanna Briggs Institute Center
Sugammadex in patients with chronic renal failure: two case reports
 ISSN: 2203-1413 Vol.04 No.02 Sugammadex in patients with chronic renal failure: two case reports Gwi Eun Yeo 1, Byung Gun Lim 1*, So Hyun Lee 1, Won Joon Lee 1, Il Ok Lee 1 1. Department of Anesthesiology
ISSN: 2203-1413 Vol.04 No.02 Sugammadex in patients with chronic renal failure: two case reports Gwi Eun Yeo 1, Byung Gun Lim 1*, So Hyun Lee 1, Won Joon Lee 1, Il Ok Lee 1 1. Department of Anesthesiology
BRIDION sugammadex (as sodium salt)
 NEW ZEALAND DATA SHEET BRIDION sugammadex (as sodium salt) (i) NAME OF THE MEDICINE Bridion 100 mg/ml Solution for Injection Molecular Formula: C 72 H 104 O 48 S 8 Na 8 Molecular mass: 2178.01 Cas. Registry
NEW ZEALAND DATA SHEET BRIDION sugammadex (as sodium salt) (i) NAME OF THE MEDICINE Bridion 100 mg/ml Solution for Injection Molecular Formula: C 72 H 104 O 48 S 8 Na 8 Molecular mass: 2178.01 Cas. Registry
An Overview of Sugammadex
 B A S I C S C I E N C E Tutorial 332 An Overview of Sugammadex Dr Ciara Mitchell Anaesthetic Registrar, Royal Victoria Hospital, Belfast, Northern Ireland Dr Steve Lobaz Consultant in Anaesthetics and
B A S I C S C I E N C E Tutorial 332 An Overview of Sugammadex Dr Ciara Mitchell Anaesthetic Registrar, Royal Victoria Hospital, Belfast, Northern Ireland Dr Steve Lobaz Consultant in Anaesthetics and
The influence of unrestricted use of sugammadex on clinical anaesthetic practice in a tertiary teaching hospital
 Anaesth Intensive Care 2012; 40: 333-339 The influence of unrestricted use of sugammadex on clinical anaesthetic practice in a tertiary teaching hospital R. W. WATTS*, J. A. LONDON, R. M. A. W. van WIJK,
Anaesth Intensive Care 2012; 40: 333-339 The influence of unrestricted use of sugammadex on clinical anaesthetic practice in a tertiary teaching hospital R. W. WATTS*, J. A. LONDON, R. M. A. W. van WIJK,
Sugammadex sodium (Organon/Schering-
 An Update on Sugammadex Sodium Mark Welliver, CRNA, DNP Dennis Cheek, RN, PhD, FAHA Sugammadex sodium is the generic drug name for the novel modified gamma cyclodextrin that terminates neuromuscular blockade
An Update on Sugammadex Sodium Mark Welliver, CRNA, DNP Dennis Cheek, RN, PhD, FAHA Sugammadex sodium is the generic drug name for the novel modified gamma cyclodextrin that terminates neuromuscular blockade
Is Sugammadex the right choice for reversal? YES NO
 Is Sugammadex the right choice for reversal? YES NO Residual Neuromuscular Blockade (rnmb) rnmb after GA using nondepolarizing NMBA s has pathophysiological, clinical, and economic consequences. Significant
Is Sugammadex the right choice for reversal? YES NO Residual Neuromuscular Blockade (rnmb) rnmb after GA using nondepolarizing NMBA s has pathophysiological, clinical, and economic consequences. Significant
4 February 2011 SUGAMMADEX. S Maharaj. Department of Anaesthetics
 4 February 2011 SUGAMMADEX S Maharaj Commentator: S Belhaj Moderator: A Hunter Department of Anaesthetics CONTENTS INTRODUCTION... 3 NEOSTIGMINE AND ATTEMPTS TO REPLACE IT... 4 SUCCINYLCHOLINE AND ATTEMPTS
4 February 2011 SUGAMMADEX S Maharaj Commentator: S Belhaj Moderator: A Hunter Department of Anaesthetics CONTENTS INTRODUCTION... 3 NEOSTIGMINE AND ATTEMPTS TO REPLACE IT... 4 SUCCINYLCHOLINE AND ATTEMPTS
BRIDION sugammadex (as sodium salt)
 BRIDION sugammadex (as sodium salt) (i) NAME OF THE MEDICINE Bridion 100 mg/ml Solution for Injection Molecular Formula: C 72 H 104 O 48 S 8 Na 8 Molecular mass: 2178.01 Cas. Registry No: 343306-79-6 Chemical
BRIDION sugammadex (as sodium salt) (i) NAME OF THE MEDICINE Bridion 100 mg/ml Solution for Injection Molecular Formula: C 72 H 104 O 48 S 8 Na 8 Molecular mass: 2178.01 Cas. Registry No: 343306-79-6 Chemical
Core Safety Profile. Pharmaceutical form(s)/strength: Solution for Injection CZ/H/PSUR/0005/002 Date of FAR:
 Core Safety Profile Active substance: Rocuronium bromide Pharmaceutical form(s)/strength: Solution for Injection P-RMS: CZ/H/PSUR/0005/002 Date of FAR: 31.05.2012 4.3 Contraindications Hypersensitivity
Core Safety Profile Active substance: Rocuronium bromide Pharmaceutical form(s)/strength: Solution for Injection P-RMS: CZ/H/PSUR/0005/002 Date of FAR: 31.05.2012 4.3 Contraindications Hypersensitivity
NERVOUS SYSTEM NERVOUS SYSTEM. Somatic nervous system. Brain Spinal Cord Autonomic nervous system. Sympathetic nervous system
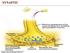 SYNAPTIC NERVOUS SYSTEM NERVOUS SYSTEM CENTRAL NERVOUS SYSTEM PERIPHERAL NERVOUS SYSTEM Brain Spinal Cord Autonomic nervous system Somatic nervous system Sympathetic nervous system Parasympathetic nervous
SYNAPTIC NERVOUS SYSTEM NERVOUS SYSTEM CENTRAL NERVOUS SYSTEM PERIPHERAL NERVOUS SYSTEM Brain Spinal Cord Autonomic nervous system Somatic nervous system Sympathetic nervous system Parasympathetic nervous
Anesthesiology, V 106, No 2, Feb
 Anesthesiology 2007; 106:283 8 Copyright 2007, the American Society of Anesthesiologists, Inc. Lippincott Williams & Wilkins, Inc. Effective Reversal of Moderate Rocuronium- or Vecuronium-induced Neuromuscular
Anesthesiology 2007; 106:283 8 Copyright 2007, the American Society of Anesthesiologists, Inc. Lippincott Williams & Wilkins, Inc. Effective Reversal of Moderate Rocuronium- or Vecuronium-induced Neuromuscular
Comparative study of intubating conditions after Rocuronium and Suxamethonium (study of 80 cases)
 ISPUB.COM The Internet Journal of Anesthesiology Volume 20 Number 1 Comparative study of intubating conditions after Rocuronium and Suxamethonium (study of 80 cases) K Bhati, V Parmar Citation K Bhati,
ISPUB.COM The Internet Journal of Anesthesiology Volume 20 Number 1 Comparative study of intubating conditions after Rocuronium and Suxamethonium (study of 80 cases) K Bhati, V Parmar Citation K Bhati,
Safety and tolerability of single intravenous doses of sugammadex administered simultaneously with rocuronium or vecuronium in healthy volunteers
 British Journal of Anaesthesia 100 (3): 373 9 (2008) doi:10.1093/bja/aem402 Advance Access publication January 31, 2008 Safety and tolerability of single intravenous doses of sugammadex administered simultaneously
British Journal of Anaesthesia 100 (3): 373 9 (2008) doi:10.1093/bja/aem402 Advance Access publication January 31, 2008 Safety and tolerability of single intravenous doses of sugammadex administered simultaneously
Succinycholine: Succinylcholine has no place in pediatric anesthesia. Wads Ames MBBS FRCA
 Succinycholine: Succinylcholine has no place in pediatric anesthesia Wads Ames MBBS FRCA Food And Drug Administration Created in 1906 Responsible for protecting and promoting public health through the
Succinycholine: Succinylcholine has no place in pediatric anesthesia Wads Ames MBBS FRCA Food And Drug Administration Created in 1906 Responsible for protecting and promoting public health through the
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS
 ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1 1. NAME OF THE MEDICINAL PRODUCT Bridion 100 mg/ml solution for injection 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 1 ml contains sugammadex sodium equivalent
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1 1. NAME OF THE MEDICINAL PRODUCT Bridion 100 mg/ml solution for injection 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 1 ml contains sugammadex sodium equivalent
Sux Rocs and Roc Suks? Succinocholyne Vs Rocuronium in RSI
 Sux Rocs and Roc Suks? Succinocholyne Vs Rocuronium in RSI Immediately declare a conflict of interest: I am in love with Succinocholyne It 'was a love at first sight. I use it often and, in my clinical
Sux Rocs and Roc Suks? Succinocholyne Vs Rocuronium in RSI Immediately declare a conflict of interest: I am in love with Succinocholyne It 'was a love at first sight. I use it often and, in my clinical
Neuromuscular Junction
 Muscle Relaxants Neuromuscular Junction Cholinergic antagonists Neuromuscular-blocking agents (mostly nicotinic antagonists): interfere with transmission of efferent impulses to skeletal muscles. These
Muscle Relaxants Neuromuscular Junction Cholinergic antagonists Neuromuscular-blocking agents (mostly nicotinic antagonists): interfere with transmission of efferent impulses to skeletal muscles. These
Chapter 18. Skeletal Muscle Relaxants (Neuromuscular Blocking Agents)
 Chapter 18 Skeletal Muscle Relaxants (Neuromuscular Blocking Agents) Uses of Neuromuscular Blocking Facilitate intubation Surgery Agents Enhance ventilator synchrony Reduce intracranial pressure (ICP)
Chapter 18 Skeletal Muscle Relaxants (Neuromuscular Blocking Agents) Uses of Neuromuscular Blocking Facilitate intubation Surgery Agents Enhance ventilator synchrony Reduce intracranial pressure (ICP)
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS
 ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1 1. NAME OF THE MEDICINAL PRODUCT Bridion 100 mg/ml solution for injection 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 1 ml contains sugammadex sodium equivalent
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1 1. NAME OF THE MEDICINAL PRODUCT Bridion 100 mg/ml solution for injection 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 1 ml contains sugammadex sodium equivalent
Joseph F. Answine, MD
 Joseph F. Answine, MD Joseph F. Answine, MD Staff Anesthesiologist Pinnacle Health Hospitals Harrisburg, PA Clinical Associate Professor of Anesthesiology Pennsylvania State University Hospital Reversal
Joseph F. Answine, MD Joseph F. Answine, MD Staff Anesthesiologist Pinnacle Health Hospitals Harrisburg, PA Clinical Associate Professor of Anesthesiology Pennsylvania State University Hospital Reversal
New Drugs in Pediatric Anesthesia
 New Drugs in Pediatric Anesthesia Anne M. Lynn MD Seattle Children s Hospital University of Washington School of Medicine There s nothing like a nice, focused topic to start this meeting. So many drugs,
New Drugs in Pediatric Anesthesia Anne M. Lynn MD Seattle Children s Hospital University of Washington School of Medicine There s nothing like a nice, focused topic to start this meeting. So many drugs,
ACCUMULATING evidence indicates that residual
 Reversal of Neuromuscular Blockade with at the Reappearance of Four Twitches to Train-of-four Stimulation Adrienn Pongrácz, M.D.,* Szilárd Szatmári, M.D., Ph.D., Réka Nemes, M.D., Béla Fülesdi, M.D., Ph.D.,
Reversal of Neuromuscular Blockade with at the Reappearance of Four Twitches to Train-of-four Stimulation Adrienn Pongrácz, M.D.,* Szilárd Szatmári, M.D., Ph.D., Réka Nemes, M.D., Béla Fülesdi, M.D., Ph.D.,
Disclosure Statement:
 Quite possibly the most interesting drug NOT approved by the FDA Yet. Sal J Morana RPh, PhD Chief Operating Officer (formerly the Pharmacy Director) Porter Medical Center Middlebury, VT Adjunct Faculty/Preceptor
Quite possibly the most interesting drug NOT approved by the FDA Yet. Sal J Morana RPh, PhD Chief Operating Officer (formerly the Pharmacy Director) Porter Medical Center Middlebury, VT Adjunct Faculty/Preceptor
PRODUCT MONOGRAPH BRIDION. solution for injection, 100 mg/ml sugammadex (as sugammadex sodium) Selective Relaxant Binding Agent
 PRDUCT MNGRAPH BRIDIN solution for injection, 100 mg/ml sugammadex (as sugammadex sodium) Selective Relaxant Binding Agent Merck Canada Inc. 16750 route Transcanadienne Kirkland, QC Canada H9H 4M7 www.merck.ca
PRDUCT MNGRAPH BRIDIN solution for injection, 100 mg/ml sugammadex (as sugammadex sodium) Selective Relaxant Binding Agent Merck Canada Inc. 16750 route Transcanadienne Kirkland, QC Canada H9H 4M7 www.merck.ca
Neuromuscular Monitoring and Patient Safety:
 Neuromuscular Monitoring and Patient Safety: Pulmonary Complications of Residual Block CEEA Course Tuesday, Sept 4, 2012 Târgu Mureş, Romania Sorin J. Brull, MD, FCARCSI (Hon) Editor, Patient Safety Section
Neuromuscular Monitoring and Patient Safety: Pulmonary Complications of Residual Block CEEA Course Tuesday, Sept 4, 2012 Târgu Mureş, Romania Sorin J. Brull, MD, FCARCSI (Hon) Editor, Patient Safety Section
FOR REPRESENTATIVE EDUCATION
 Neuromuscular Blockade in the ICU NIMBEX Indication 1 NIMBEX (cisatracurium besylate) is indicated as an adjunct to general anesthesia to facilitate tracheal intubation in adults and in pediatric patients
Neuromuscular Blockade in the ICU NIMBEX Indication 1 NIMBEX (cisatracurium besylate) is indicated as an adjunct to general anesthesia to facilitate tracheal intubation in adults and in pediatric patients
Effect of Vecuronium in different age group
 Original Research Article Effect of Vecuronium in different age group Bharti Rajani 1, Hitesh Brahmbhatt 2, Hemlata Chaudhry 2, Hiren Parmar 3* 1 Associate Professor, Department of Anesthesiology, GMERS
Original Research Article Effect of Vecuronium in different age group Bharti Rajani 1, Hitesh Brahmbhatt 2, Hemlata Chaudhry 2, Hiren Parmar 3* 1 Associate Professor, Department of Anesthesiology, GMERS
In vivo animal studies with sugammadex
 In vivo animal studies with sugammadex L. H. D. J. Booij, 1 J. van Egmond, 2 J. J. Driessen 3 and H. D. de Boer 4 1 Professor of Anaesthesiology, 2 Physicist, 3 Associate Professor of Paediatric Anaesthesiology,
In vivo animal studies with sugammadex L. H. D. J. Booij, 1 J. van Egmond, 2 J. J. Driessen 3 and H. D. de Boer 4 1 Professor of Anaesthesiology, 2 Physicist, 3 Associate Professor of Paediatric Anaesthesiology,
Reversal of Rocuronium-induced Neuromuscular Block by the Selective Relaxant Binding Agent Sugammadex
 Anesthesiology 2006; 104:667 74 2006 American Society of Anesthesiologists, Inc. Lippincott Williams & Wilkins, Inc. Reversal of Rocuronium-induced Neuromuscular Block by the Selective Relaxant Binding
Anesthesiology 2006; 104:667 74 2006 American Society of Anesthesiologists, Inc. Lippincott Williams & Wilkins, Inc. Reversal of Rocuronium-induced Neuromuscular Block by the Selective Relaxant Binding
Pharmacology of the Neuromuscular Junction (NMJ)
 Pharmacology of the Neuromuscular Junction (NMJ) Edward JN Ishac, Ph.D. Professor Smith Building, Room 742 eishac@vcu.edu 828 2127 Department of Pharmacology and Toxicology Medical College of Virginia
Pharmacology of the Neuromuscular Junction (NMJ) Edward JN Ishac, Ph.D. Professor Smith Building, Room 742 eishac@vcu.edu 828 2127 Department of Pharmacology and Toxicology Medical College of Virginia
Reversal of neuromuscular block
 British Journal of Anaesthesia 103 (1): 115 29 (2009) doi:10.1093/bja/aep093 Advance Access publication May 24, 2009 Reversal of neuromuscular block A. rivastava* and J. M. Hunter University of Liverpool
British Journal of Anaesthesia 103 (1): 115 29 (2009) doi:10.1093/bja/aep093 Advance Access publication May 24, 2009 Reversal of neuromuscular block A. rivastava* and J. M. Hunter University of Liverpool
Pharmacology of the Neuromuscular Junction (NMJ)
 Pharmacology of the Neuromuscular Junction (NMJ) Edward JN Ishac, Ph.D. Professor Smith Building, Room 742 eishac@vcu.edu 828 2127 Department of Pharmacology and Toxicology Medical College of Virginia
Pharmacology of the Neuromuscular Junction (NMJ) Edward JN Ishac, Ph.D. Professor Smith Building, Room 742 eishac@vcu.edu 828 2127 Department of Pharmacology and Toxicology Medical College of Virginia
The Neuromuscular Junction: Pharmacologic Applications During Anesthesia
 Page 1 The Neuromuscular Junction: Pharmacologic Applications During Anesthesia Cynthia A. Lien, M.D. New York, NewYork Introduction The neuromuscular junction and neuromuscular transmission are well-studied
Page 1 The Neuromuscular Junction: Pharmacologic Applications During Anesthesia Cynthia A. Lien, M.D. New York, NewYork Introduction The neuromuscular junction and neuromuscular transmission are well-studied
Monitoring of neuromuscular block in operative room and ICU Josep Rodiera M.D. Ph.D. MsC
 Monitoring of neuromuscular block in operative room and ICU Josep Rodiera M.D. Ph.D. MsC Marrakech 2016 Anesthesia Department Centro Medico Teknon Barcelona Conflict of interest Creator of the TOFCuff
Monitoring of neuromuscular block in operative room and ICU Josep Rodiera M.D. Ph.D. MsC Marrakech 2016 Anesthesia Department Centro Medico Teknon Barcelona Conflict of interest Creator of the TOFCuff
ACETYLCHOLINESTERASE inhibitors, such as
 PERIOPERATIVE MEDICINE Anesthesiology 2010; 113:1054 60 Copyright 2010, the American Society of Anesthesiologists, Inc. Lippincott Williams & Wilkins Sugammadex and Neostigmine Dose-finding Study for Reversal
PERIOPERATIVE MEDICINE Anesthesiology 2010; 113:1054 60 Copyright 2010, the American Society of Anesthesiologists, Inc. Lippincott Williams & Wilkins Sugammadex and Neostigmine Dose-finding Study for Reversal
Package leaflet: Information for the user. Bridion 100 mg/ml solution for injection sugammadex
 Package leaflet: Information for the user Bridion 100 mg/ml solution for injection sugammadex Read all of this leaflet carefully before you are given this medicine because it contains important information
Package leaflet: Information for the user Bridion 100 mg/ml solution for injection sugammadex Read all of this leaflet carefully before you are given this medicine because it contains important information
New Zealand Data Sheet DBL TM Rocuronium Bromide Injection
 New Zealand Data Sheet DBL TM Rocuronium Bromide Injection NAME OF THE MEDICINE DBL TM Rocuronium Bromide Injection 50 mg/5 ml Vial PRESENTATION Rocuronium bromide is a quaternary aminosteroid and an analogue
New Zealand Data Sheet DBL TM Rocuronium Bromide Injection NAME OF THE MEDICINE DBL TM Rocuronium Bromide Injection 50 mg/5 ml Vial PRESENTATION Rocuronium bromide is a quaternary aminosteroid and an analogue
NEUROMUSCULAR BLOCKING AGENTS
 NEUROMUSCULAR BLOCKING AGENTS Edward JN Ishac, Ph.D. Associate Professor, Pharmacology and Toxicology Smith 742, 828-2127, Email: eishac@vcu.edu Learning Objectives: 1. Understand the physiology of the
NEUROMUSCULAR BLOCKING AGENTS Edward JN Ishac, Ph.D. Associate Professor, Pharmacology and Toxicology Smith 742, 828-2127, Email: eishac@vcu.edu Learning Objectives: 1. Understand the physiology of the
(PP XI) Dr. Samir Matloob
 DRUGS ACTING ON THE CHOLINERGIC SYSTEM AND THE NEUROMUSCULAR BLOCKING DRUGS IV (NICOTINIC ANTAGONISTS) (PP XI) Dr. Samir Matloob Dept. of Pharmacology Baghdad College of Medicine Drugs acting on the cholinergic
DRUGS ACTING ON THE CHOLINERGIC SYSTEM AND THE NEUROMUSCULAR BLOCKING DRUGS IV (NICOTINIC ANTAGONISTS) (PP XI) Dr. Samir Matloob Dept. of Pharmacology Baghdad College of Medicine Drugs acting on the cholinergic
New Drug Developments for Neuromuscular Blockade and Reversal: Gantacurium, CW002, CW011, and Calabadion
 Current Anesthesiology Reports (2018) 8:119 124 https://doi.org/10.1007/s40140-018-0262-9 NEUROMUSCULAR BLOCKADE (GS MURPHY, SECTION EDITOR) New Drug Developments for Neuromuscular Blockade and Reversal:
Current Anesthesiology Reports (2018) 8:119 124 https://doi.org/10.1007/s40140-018-0262-9 NEUROMUSCULAR BLOCKADE (GS MURPHY, SECTION EDITOR) New Drug Developments for Neuromuscular Blockade and Reversal:
A Nondepolarizing Neuromuscular Blocking (NMB) Agent
 DOSING GUIDE A Nondepolarizing Neuromuscular Blocking (NMB) Agent Easy to remember dosing for the 0.20 mg/kg adult intubating doses of NIMBEX 1 *: For every 10 kg, give 1 ml of NIMBEX (2 mg/ml concentration)
DOSING GUIDE A Nondepolarizing Neuromuscular Blocking (NMB) Agent Easy to remember dosing for the 0.20 mg/kg adult intubating doses of NIMBEX 1 *: For every 10 kg, give 1 ml of NIMBEX (2 mg/ml concentration)
Guide to Neuromuscular Blocking Agents
 Guide to Neuromuscular Blocking Agents EMILY STERLING, PHARMD Associate Clinical Instructor University of Washington School of Pharmacy Seattle, Washington P. SHANE WINSTEAD, PHARMD ICU Clinical Pharmacy
Guide to Neuromuscular Blocking Agents EMILY STERLING, PHARMD Associate Clinical Instructor University of Washington School of Pharmacy Seattle, Washington P. SHANE WINSTEAD, PHARMD ICU Clinical Pharmacy
TRIALS. Martijn Boon 1, Christian H Martini 1, Leon PHJ Aarts 1, Rob FM Bevers 2 and Albert Dahan 1*
 Boon et al. Trials 2013, 14:63 TRIALS STUDY PROTOCOL Open Access Effect of variations in depth of neuromuscular blockade on rating of surgical conditions by surgeon and anesthesiologist in patients undergoing
Boon et al. Trials 2013, 14:63 TRIALS STUDY PROTOCOL Open Access Effect of variations in depth of neuromuscular blockade on rating of surgical conditions by surgeon and anesthesiologist in patients undergoing
Benztropine and trihexyphenidyl: Centrally acting antimuscarinic agents used for treatment of Parkinson disease & extrapyramidal symptoms.
 Scopolamine: Tertiary amine plant alkaloid. Produces peripheral effects similar to those of atropine. Unlike atropine, scopolamine has greater action on the CNS (observed at therapeutic doses). It has
Scopolamine: Tertiary amine plant alkaloid. Produces peripheral effects similar to those of atropine. Unlike atropine, scopolamine has greater action on the CNS (observed at therapeutic doses). It has
Comparison of Intubating Conditions of Succinylcholine and Rocuronium
 Comparison of Intubating Conditions of Succinylcholine and Rocuronium 1 Dr. Vaishali Kotambkar, 2 Dr. Sunil Tuljapure Abstract: The aim of study on neuromuscular drugs was to have nondepolarising muscle
Comparison of Intubating Conditions of Succinylcholine and Rocuronium 1 Dr. Vaishali Kotambkar, 2 Dr. Sunil Tuljapure Abstract: The aim of study on neuromuscular drugs was to have nondepolarising muscle
Sugammadex: Pharmacology, Safety, and Associated Anesthetic Implications
 Sugammadex: Pharmacology, Safety, and Associated Anesthetic Implications LESSON 24 Volume 39* 4/06/2017 L. Harold Barnwell, DNAP, CRNA Staff Anesthetist and Affiliate Faculty Department of Anesthesiology
Sugammadex: Pharmacology, Safety, and Associated Anesthetic Implications LESSON 24 Volume 39* 4/06/2017 L. Harold Barnwell, DNAP, CRNA Staff Anesthetist and Affiliate Faculty Department of Anesthesiology
The effect of desflurane on rocuronium onset, clinical duration and maintenance requirements
 (Acta Anaesth. Belg., 2006, 57, 349-353) The effect of desflurane on rocuronium onset, clinical duration and maintenance requirements R. G. STOUT (*), T. J. GAN (**), P. S. A. GLASS (***), D. G. SILVERMAN
(Acta Anaesth. Belg., 2006, 57, 349-353) The effect of desflurane on rocuronium onset, clinical duration and maintenance requirements R. G. STOUT (*), T. J. GAN (**), P. S. A. GLASS (***), D. G. SILVERMAN
ADVANCES IN ANESTHESIA
 Advances in Anesthesia 29 (2011) 19 37 ADVANCES IN ANESTHESIA Sugammadex: Past, Present, and Future James E. Caldwell, MB, ChB Department of Anesthesia and Perioperative Medicine, University of California,
Advances in Anesthesia 29 (2011) 19 37 ADVANCES IN ANESTHESIA Sugammadex: Past, Present, and Future James E. Caldwell, MB, ChB Department of Anesthesia and Perioperative Medicine, University of California,
Active ingredients: Metoclopramide Hydrochloride mg Equivalent to metoclopramide hydrochloride anhydrous mg
 Name Primperan 10 mg / 2 ml Metoclopramide hydrochloride anhydrous Solution for I.M. or I.V. injection (Ampoules) Composition Each 2 ml ampoule contains: Active ingredients: Metoclopramide Hydrochloride.
Name Primperan 10 mg / 2 ml Metoclopramide hydrochloride anhydrous Solution for I.M. or I.V. injection (Ampoules) Composition Each 2 ml ampoule contains: Active ingredients: Metoclopramide Hydrochloride.
Is lower-dose sugammadex a cost-saving strategy for reversal of deep neuromuscular block? Facts and fiction
 de Boer et al. BMC Anesthesiology (2018) 18:159 https://doi.org/10.1186/s12871-018-0605-6 REVIEW Open Access Is lower-dose sugammadex a cost-saving strategy for reversal of deep neuromuscular block? Facts
de Boer et al. BMC Anesthesiology (2018) 18:159 https://doi.org/10.1186/s12871-018-0605-6 REVIEW Open Access Is lower-dose sugammadex a cost-saving strategy for reversal of deep neuromuscular block? Facts
MUSCLE RELAXANTS. Mr. D.Raju, M.pharm, Lecturer
 MUSCLE RELAXANTS Mr. D.Raju, M.pharm, Lecturer Muscle Relaxants are classified as: I)Peripherally acting A.Neuromuscular blocking agents:- 1) Depolarizing muscle relaxants. 2) Non-depolarizing muscle relaxants
MUSCLE RELAXANTS Mr. D.Raju, M.pharm, Lecturer Muscle Relaxants are classified as: I)Peripherally acting A.Neuromuscular blocking agents:- 1) Depolarizing muscle relaxants. 2) Non-depolarizing muscle relaxants
08-15 NORCURON ORGANON
 08-15 NORCURON ORGANON 1. NAME OF THE MEDICINAL PRODUCT Norcuron 4 mg, powder for solution for injection Norcuron 10 mg, powder for solution for injection 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Norcuron
08-15 NORCURON ORGANON 1. NAME OF THE MEDICINAL PRODUCT Norcuron 4 mg, powder for solution for injection Norcuron 10 mg, powder for solution for injection 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Norcuron
DO WE NEED TO USE SUGAMMADEX AT THE END OF A GENERAL ANESTHESIA TO REVERSE THE ACTION OF NEUROMUSCULAR BLOKING AGENTS?
 1 Experts opinion and Point of view are papers reviewing the literature, usually invited by the Editor in Chief and do not exceed 3000 and 2000 words with 50 and 40 references respectively. parole: 2666
1 Experts opinion and Point of view are papers reviewing the literature, usually invited by the Editor in Chief and do not exceed 3000 and 2000 words with 50 and 40 references respectively. parole: 2666
*Author for Correspondence
 COMPARATIVE EVALUATION OF ONSET TIME INTUBATING CONDITION JUDGED BY CLINICAL CRITERIA AND HAEMODYNAMIC RESPONSE AFTER THE INTUBATING DOSE OF ROCURONIUM AND VECURONIUM *Namita Gupta 1, Mamta Sharma 2, Pusplata
COMPARATIVE EVALUATION OF ONSET TIME INTUBATING CONDITION JUDGED BY CLINICAL CRITERIA AND HAEMODYNAMIC RESPONSE AFTER THE INTUBATING DOSE OF ROCURONIUM AND VECURONIUM *Namita Gupta 1, Mamta Sharma 2, Pusplata
Running head: SUGAMMADEX AND MYASTHENIA GRAVIS 1
 Running head: SUGAMMADEX AND MYASTHENIA GRAVIS 1 Sugammadex in Patients with Myasthenia Gravis Jennifer A. Madsen University of Kansas SUGAMMADEX AND MYASTHENIA GRAVIS 2 Title of Proposed Research Project
Running head: SUGAMMADEX AND MYASTHENIA GRAVIS 1 Sugammadex in Patients with Myasthenia Gravis Jennifer A. Madsen University of Kansas SUGAMMADEX AND MYASTHENIA GRAVIS 2 Title of Proposed Research Project
Ganglion blocking agents
 Ganglion blocking agents -out of date -Specifically act on the nicotinic receptors of both parasymphatetic and sympathetic ganglia - no selectivity toward PG or SG -These drugs are non-depolarizing, competitive
Ganglion blocking agents -out of date -Specifically act on the nicotinic receptors of both parasymphatetic and sympathetic ganglia - no selectivity toward PG or SG -These drugs are non-depolarizing, competitive
INTUBATING CONDITIONS AND INJECTION PAIN
 INTUBATING CONDITIONS AND INJECTION PAIN - Cisatracurium or Rocuronium versus Rocuronium-Cisatracurium Combination - AHED ZEIDAN *, NAZIH NAHLE *, HILAL MAALIKI ** AND ANIS BARAKA *** Summary The present
INTUBATING CONDITIONS AND INJECTION PAIN - Cisatracurium or Rocuronium versus Rocuronium-Cisatracurium Combination - AHED ZEIDAN *, NAZIH NAHLE *, HILAL MAALIKI ** AND ANIS BARAKA *** Summary The present
Cholinergic antagonists
 Cholinergic antagonists Cholinergic antagonists: They are also called anticholinergic drugs or cholinergic blockers, this group include: 1.Antimuscarinic agents ( atropine, ipratropium, scopolamine) 2.
Cholinergic antagonists Cholinergic antagonists: They are also called anticholinergic drugs or cholinergic blockers, this group include: 1.Antimuscarinic agents ( atropine, ipratropium, scopolamine) 2.
MINERVA ANESTESIOLOGICA EDIZIONI MINERVA MEDICA
 MINERVA ANESTESIOLOGICA EDIZIONI MINERVA MEDICA This provisional PDF corresponds to the article as it appeared upon acceptance. A copyedited and fully formatted version will be made available soon. The
MINERVA ANESTESIOLOGICA EDIZIONI MINERVA MEDICA This provisional PDF corresponds to the article as it appeared upon acceptance. A copyedited and fully formatted version will be made available soon. The
SUMMARY OF PRODUCT CHARACTERISTICS. 1. NAME OF THE MEDICINAL PRODUCT Rocuronium Kabi 10 mg/ml solution for injection/infusion
 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Rocuronium Kabi 10 mg/ml solution for injection/infusion 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each ml of solution for injection
SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Rocuronium Kabi 10 mg/ml solution for injection/infusion 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each ml of solution for injection
Suggamadex. Suggamadex. Suggamadex 4/28/2015. Goals: By the End of this Lecture, the Participant should be able to:
 Presented April, 2015 - OOA Convention by Ronald S. Stevens DO Board Certified Anesthesiologist Member Board of Trustees, OOA President/CEO Green Country Anesthesiology Associates PC. Conflict of Interest
Presented April, 2015 - OOA Convention by Ronald S. Stevens DO Board Certified Anesthesiologist Member Board of Trustees, OOA President/CEO Green Country Anesthesiology Associates PC. Conflict of Interest
University of Mississippi Medical Center University of Mississippi Health Care. Pharmacy and Therapeutics Committee Medication Use Evaluation
 University of Mississippi Medical Center University of Mississippi Health Care Pharmacy and Therapeutics Committee Medication Use Evaluation Cisatracurium (Nimbex ) and Vecuronium (Norcuron ) PURPOSE The
University of Mississippi Medical Center University of Mississippi Health Care Pharmacy and Therapeutics Committee Medication Use Evaluation Cisatracurium (Nimbex ) and Vecuronium (Norcuron ) PURPOSE The
Introduction Corresponding author:
 Anesthetic Management Of Aortic Valve Replacement In A Myasthenia Gravis Patient, The Era Of A New Reversal Alia S. Dabbous 1*, Patricia W Nehme 1 and Ahmad M Abou Leila 2 A 66 year old man diagnosed with
Anesthetic Management Of Aortic Valve Replacement In A Myasthenia Gravis Patient, The Era Of A New Reversal Alia S. Dabbous 1*, Patricia W Nehme 1 and Ahmad M Abou Leila 2 A 66 year old man diagnosed with
Farmadol. Paracetamol 10 mg/ml INFUSION SOLUTION
 Farmadol Paracetamol 10 mg/ml INFUSION SOLUTION Composition Each ml contains: Paracetamol 10 mg Pharmacology Pharmacodynamic properties The precise mechanism of the analgesic and antipyretic properties
Farmadol Paracetamol 10 mg/ml INFUSION SOLUTION Composition Each ml contains: Paracetamol 10 mg Pharmacology Pharmacodynamic properties The precise mechanism of the analgesic and antipyretic properties
Succinylcholine born 1951 and still going faster. Matthias W. König, MD FAAP
 Succinylcholine born 1951 and still going faster Matthias W. König, MD FAAP What kind of quack is he? Dr. Lupa never does this! OMG, you want to give SUX? I hope I don t get this question on the oral boards
Succinylcholine born 1951 and still going faster Matthias W. König, MD FAAP What kind of quack is he? Dr. Lupa never does this! OMG, you want to give SUX? I hope I don t get this question on the oral boards
Zekeriyya Alanoglu. Definition of Residual Neuromuscular Block
 Postoperative Residual Neuromuscular Block Zekeriyya Alanoglu Almost 60 years ago the use neuromuscular blocking agents (NMBA) was associated with significantly increased risk of perioperative (approx.
Postoperative Residual Neuromuscular Block Zekeriyya Alanoglu Almost 60 years ago the use neuromuscular blocking agents (NMBA) was associated with significantly increased risk of perioperative (approx.
Comparative study of Succinylcholine, Rocuronium and Vecuronium for intubation and hemodynamic changes during general anaesthesia
 IOSR Journal of Dental and Medical Sciences (IOSR-JDMS) e-issn: 2279-0853, p-issn: 2279-0861.Volume 15, Issue 10 Ver. I (October. 2016), PP 14-19 www.iosrjournals.org Comparative study of Succinylcholine,
IOSR Journal of Dental and Medical Sciences (IOSR-JDMS) e-issn: 2279-0853, p-issn: 2279-0861.Volume 15, Issue 10 Ver. I (October. 2016), PP 14-19 www.iosrjournals.org Comparative study of Succinylcholine,
SUMMARY OF PRODUCT CHARACTERISTICS. 1 ml solution contains 75 micrograms of sufentanilcitrate, corresponding to 50 micrograms of sufentanil.
 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Sufentanil Narcomed, 50 microgram / ml, solution for injection 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 1 ml solution contains 75
SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Sufentanil Narcomed, 50 microgram / ml, solution for injection 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 1 ml solution contains 75
Shaded areas=not MARKETED 24/2/09
 PACKAGE INSERT SCHEDULING STATUS Schedule 6 PROPRIETARY NAME AND DOSAGE FORM RAPIFEN 2 ml IV injection RAPIFEN 10 ml IV injection COMPOSITION Each ml contains alfentanil hydrochloride 0,544 mg (equivalent
PACKAGE INSERT SCHEDULING STATUS Schedule 6 PROPRIETARY NAME AND DOSAGE FORM RAPIFEN 2 ml IV injection RAPIFEN 10 ml IV injection COMPOSITION Each ml contains alfentanil hydrochloride 0,544 mg (equivalent
MUSCLE relaxants are used to provide better operating conditions for
 CLEVELAND CLINIC QUARTERLY Copyright 1970 by The Cleveland Clinic Foundation Volume 37, July 1970 Printed in U.S.A. Pancuronium bromide A new non-depolarizing muscle relaxant Preliminary in fifty patients
CLEVELAND CLINIC QUARTERLY Copyright 1970 by The Cleveland Clinic Foundation Volume 37, July 1970 Printed in U.S.A. Pancuronium bromide A new non-depolarizing muscle relaxant Preliminary in fifty patients
Neuromuscular Blockers
 Neuromuscular Blockers Joanne Leung joanneleung22@hotmail.com Oct 14, 2014 Objectives After this lecture, you should be able to: Describe the physiology of the neuromuscular junction Differentiate the
Neuromuscular Blockers Joanne Leung joanneleung22@hotmail.com Oct 14, 2014 Objectives After this lecture, you should be able to: Describe the physiology of the neuromuscular junction Differentiate the
HST-151 Clinical Pharmacology in the Operating Room
 Harvard-MIT Division of Health Sciences and Technology HST.151: Principles of Pharmocology Instructors: Dr. Carl Rosow, Dr. David Standaert and Prof. Gary Strichartz 1 HST-151 Clinical Pharmacology in
Harvard-MIT Division of Health Sciences and Technology HST.151: Principles of Pharmocology Instructors: Dr. Carl Rosow, Dr. David Standaert and Prof. Gary Strichartz 1 HST-151 Clinical Pharmacology in
Childhood Obesity: Anesthetic Implications
 Childhood Obesity: Anesthetic Implications The Changing Practice of Anesthesia 2015 UCSF Department of Anesthesia and Perioperative Care Marla Ferschl, MD Associate Professor of Anesthesia University of
Childhood Obesity: Anesthetic Implications The Changing Practice of Anesthesia 2015 UCSF Department of Anesthesia and Perioperative Care Marla Ferschl, MD Associate Professor of Anesthesia University of
2 QUALITATIVE AND QUANTITATIVE COMPOSITION
 SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Rocuronium bromide 10 mg/ml solution for injection/ infusion 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each ml of solution of Rocuronium
SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Rocuronium bromide 10 mg/ml solution for injection/ infusion 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each ml of solution of Rocuronium
AUSTRALIAN PRODUCT INFORMATION REMIFENTANIL APOTEX (REMIFENTANIL HYDROCHLORIDE) POWDER FOR INJECTION
 AUSTRALIAN PRODUCT INFORMATION REMIFENTANIL APOTEX (REMIFENTANIL HYDROCHLORIDE) POWDER FOR INJECTION 1 NAME OF THE MEDICINE Remifentanil (as hydrochloride) 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each
AUSTRALIAN PRODUCT INFORMATION REMIFENTANIL APOTEX (REMIFENTANIL HYDROCHLORIDE) POWDER FOR INJECTION 1 NAME OF THE MEDICINE Remifentanil (as hydrochloride) 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each
NEW ZEALAND DATA SHEET
 NEW ZEALAND DATA SHEET VECURE 1. Product Name VECURE 10 mg, powder for injection. 2. Qualitative and Quantitative Composition VECURE 10 mg, 1 vial contains 10 mg vecuronium bromide. When reconstituted
NEW ZEALAND DATA SHEET VECURE 1. Product Name VECURE 10 mg, powder for injection. 2. Qualitative and Quantitative Composition VECURE 10 mg, 1 vial contains 10 mg vecuronium bromide. When reconstituted
Glycopyrronium Bromide and Neostigmine Metilsulfate 0.5mg/2.5mg per ml
 New Zealand Datasheet Glycopyrronium Bromide and Neostigmine Metilsulfate 0.5mg/2.5mg per ml Solution for injection Glycopyrronium bromide 0.5 mg/ml & Neostigmine metilsulfate 2.5 mg/ml Presentation is
New Zealand Datasheet Glycopyrronium Bromide and Neostigmine Metilsulfate 0.5mg/2.5mg per ml Solution for injection Glycopyrronium bromide 0.5 mg/ml & Neostigmine metilsulfate 2.5 mg/ml Presentation is
Dialysability of sugammadex and its complex with rocuronium in intensive care patients with severe renal impairment
 British Journal of Anaesthesia 109 (3): 382 90 (2012) Advance Access publication 24 June 2012. doi:10.1093/bja/aes207 Dialysability of sugammadex and its complex with rocuronium in intensive care patients
British Journal of Anaesthesia 109 (3): 382 90 (2012) Advance Access publication 24 June 2012. doi:10.1093/bja/aes207 Dialysability of sugammadex and its complex with rocuronium in intensive care patients
Remifentanil. Addressing the challenges of ambulatory orthopedic procedures 1-3
 Remifentanil Addressing the challenges of ambulatory orthopedic procedures 1-3 INDICATIONS AND IMPORTANT RISK INFORMATION INDICATIONS ULTIVA (remifentanil HCl) for Injection is indicated for intravenous
Remifentanil Addressing the challenges of ambulatory orthopedic procedures 1-3 INDICATIONS AND IMPORTANT RISK INFORMATION INDICATIONS ULTIVA (remifentanil HCl) for Injection is indicated for intravenous
Neostigmine but not sugammadex impairs upper airway dilator. activity and breathing.
 British Journal of Anaesthesia 101 (3): 344 9 (2008) doi:10.1093/bja/aen176 Advance Access publication June 16, 2008 Neostigmine but not sugammadex impairs upper airway dilator muscle activity and breathing
British Journal of Anaesthesia 101 (3): 344 9 (2008) doi:10.1093/bja/aen176 Advance Access publication June 16, 2008 Neostigmine but not sugammadex impairs upper airway dilator muscle activity and breathing
Tracheal intubation in children after induction of anesthesia with propofol and remifentanil without a muscle relaxant
 Original Article Tracheal intubation in children after induction of anesthesia with propofol and remifentanil without a muscle relaxant Mirmohammad Taghi Mortazavi, 1 Masood Parish, 1 Naghi Abedini, 2
Original Article Tracheal intubation in children after induction of anesthesia with propofol and remifentanil without a muscle relaxant Mirmohammad Taghi Mortazavi, 1 Masood Parish, 1 Naghi Abedini, 2
Ganglionic Blocking Agents
 Ganglionic Blocking Agents 1- Depolarizing Ganglionic Blocking Agents Depolarizing blocking agents are actually ganglionic stimulants. Thus, for nicotine, small doses give an action similar to that of
Ganglionic Blocking Agents 1- Depolarizing Ganglionic Blocking Agents Depolarizing blocking agents are actually ganglionic stimulants. Thus, for nicotine, small doses give an action similar to that of
PRODUCT MONOGRAPH. Solution for Injection 20 mg/10 ml (2 mg/ml) Cisatracurium. Non-depolarizing Skeletal Neuromuscular Blocking Agent
 PRODUCT MONOGRAPH Pr CISATRACURIUM BESYLATE INJECTION Solution for Injection 20 mg/10 ml (2 mg/ml) Cisatracurium Non-depolarizing Skeletal Neuromuscular Blocking Agent This drug should be administered
PRODUCT MONOGRAPH Pr CISATRACURIUM BESYLATE INJECTION Solution for Injection 20 mg/10 ml (2 mg/ml) Cisatracurium Non-depolarizing Skeletal Neuromuscular Blocking Agent This drug should be administered
NEW ZEALAND DATA SHEET. Injection solution 2.5 mg/ml: a clear, colourless, particle-free solution containing 2.5 mg/ml Neostigmine methylsulphate.
 NEW ZEALAND DATA SHEET NAME OF MEDICINE NEOSTIGMINE METHYLSULPHATE INJECTION B.P. Solution for injection 2.5 mg/ml. PRESENTATIONS Injection solution 2.5 mg/ml: a clear, colourless, particle-free solution
NEW ZEALAND DATA SHEET NAME OF MEDICINE NEOSTIGMINE METHYLSULPHATE INJECTION B.P. Solution for injection 2.5 mg/ml. PRESENTATIONS Injection solution 2.5 mg/ml: a clear, colourless, particle-free solution
Effect of Sugammadex on Postoperative Bleeding and Coagulation Parameters After Septoplasty: A Randomized Prospective Study
 e-issn 1643-3750 DOI: 10.12659/MSM.894971 Received: 2015.06.11 Accepted: 2015.07.24 Published: 2015.08.14 Effect of Sugammadex on Postoperative Bleeding and Coagulation Parameters After Septoplasty: A
e-issn 1643-3750 DOI: 10.12659/MSM.894971 Received: 2015.06.11 Accepted: 2015.07.24 Published: 2015.08.14 Effect of Sugammadex on Postoperative Bleeding and Coagulation Parameters After Septoplasty: A
SUMMARY OF PRODUCT CHARACTERISTICS
 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT ACURMIL 25 mg/2.5 ml solution for intravenous injection ACURMIL 50 mg/5 ml solution for intravenous injection 2. QUALITATIVE AND QUANTITATIVE
SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT ACURMIL 25 mg/2.5 ml solution for intravenous injection ACURMIL 50 mg/5 ml solution for intravenous injection 2. QUALITATIVE AND QUANTITATIVE
Discovery, development, and clinical application of sugammadex sodium, a selective relaxant binding agent
 REVIEW Discovery, development, and clinical application of sugammadex sodium, a selective relaxant binding agent Mark Welliver 1,3 John McDonough 1,3 Nicholas Kalynych 1,2,3 Robert Redfern 3 1 University
REVIEW Discovery, development, and clinical application of sugammadex sodium, a selective relaxant binding agent Mark Welliver 1,3 John McDonough 1,3 Nicholas Kalynych 1,2,3 Robert Redfern 3 1 University
Quelicin TM SUCCINYLCHOLINE CHLORIDE INJECTION, USP
 Quelicin TM SUCCINYLCHOLINE CHLORIDE INJECTION, USP A short-acting depolarizing skeletal muscle relaxant. Fliptop Vial Rx only WARNING RISK OF CARDIAC ARREST FROM HYPERKALEMIC RHABDOMYOLYSIS There have
Quelicin TM SUCCINYLCHOLINE CHLORIDE INJECTION, USP A short-acting depolarizing skeletal muscle relaxant. Fliptop Vial Rx only WARNING RISK OF CARDIAC ARREST FROM HYPERKALEMIC RHABDOMYOLYSIS There have
