EpiCARE a network for rare and complex epilepsies
|
|
|
- Hugh Holland
- 5 years ago
- Views:
Transcription
1 Grant agreement no EpiCARE a network for rare and complex epilepsies HP-ERN-2016 European Reference Networks / Framework Partnership Agreement D15.1: Proposal for EpiCARE clinical database Work Package: Research IV (WP15) Due date of deliverable: 31 Oct 2017 Actual submission date: 31 January 2018 Start date of project: 01 March 2017 Duration: 12 months Lead beneficiary for this deliverable: Azienda Ospedaliero-Universitaria A Meyer, Florence, Italy Contributors: Professor Renzo Guerrini and Dr Carmen Barba Project co-funded by the European Commission HP-ERN-2016 European Reference Networks / Framework Partnership Agreement Grant PU Public Dissemination Level PU Page 1 Version 0.2
2 Disclaimer The content of this deliverable does not reflect the official opinion of the European Union. Responsibility for the information and views expressed herein lies entirely with the author(s). PU Page 2 Version 0.2
3 Table of contents 1. Version log Definition and acronyms Introduction Activities carried out and results Conclusions Bibliography / References... 7 PU Page 3 Version 0.2
4 1. Version log Version Date Released by Nature of Change 01 8 th January st January 2018 R. Guerrini (Meyer Hospital) R. Guerrini (Meyer Hospital) First version Deliverable report was modified in order to make it clear what information is collected in the dataset PU Page 4 Version 0.2
5 2. Definition and acronyms Acronyms EpiCARE RedCap TC Epi25K Definitions European Reference Network for rare and complex epilepsies Secure web application for building and managing online surveys and databases Teleconferences Large-Scale Whole Genome Sequencing in Epilepsy consortium PU Page 5 Version 0.2
6 3. Introduction Research based on clinical practice is an important component of EpiCARE. A database, as reported by the deliverable D 15.1, is the starting point for (clinical) research projects promoted by EpiCARE members, as it may indicate how many patients with a specific rare and complex epilepsy can be enrolled and can also facilitate coordinated efforts for applications to Horizon 2020, E-rare calls, clinical trials and publications on even extremely rare disorders. The deliverable D 15.1 refers to objective 5 of EpiCARE proposal: To enhance opportunities for registries, and collaborative research for the benefit of individuals with rare and complex epilepsies across Europe. EpiCARE utilizing the database/registries may allow natural history studies of individual phenotypes (to include epidemiologic, phenotypic, genotypic, response to medication, natural history, comorbidity, long term outcome, existing treatments, mortality) and determine further key areas of research priority to be addressed, as illustrated with the Dravet syndrome experience, for example ( ). The specific objective of deliverable D 15.1 is: to establish a European clinical database (REDCap) for complex and rare epilepsies. A database is an assortment of data that is organized to be easily accessed, managed and updated. Such a research prone, clinical database will subserve clinical trials, genetic and epidemiologic research. 4. Activities carried out and results The structure of the clinical database reported in the deliverable D 15.1, has been set up through discussions in the context of Steering Committee monthly TC and dedicated face-to face meetings. Prof Lieven Lagae, WP I leader, has significantly contributed to the development of the database. As first step, differences between registries and databases have been pointed out. Secondly, the Epi25K database ( ) has been chosen as a model to build the EpiCARE database, and has been adapted to more general clinical purposes and simplified to make it particularly suitable to transfer already stored data and quickly collect new data even with limited resources available. In order to feed the data in reasonably quick way, the database has been simplified and a modular structure has been used. An agreement has been reached concerning the minimum dataset (i.e. demographics, etiology and syndromic classification of epilepsy, comorbidities and treatment) during a dedicated Steering committee meeting (3 rd December 2017). This scheme has been PU Page 6 Version 0.2
7 circulated to the Steering committee members for further amendments in December Finally, a clinical database fulfilling D 15.1 requirements has been set up (see D15.1 ANNEX 1). As shown in the D15.1 ANNEX 1, this database contains core (clinical) data (limited number of relevant data fields: demographics, age at epilepsy diagnosis, etiology of epilepsy, type of epilepsy syndrome, presence of associated intellectual disability, current medical and dietary treatment, previous surgical treatment) with drop down menus for specific items (i.e different types of epilepsy syndromes and current medical treatment) and does not need to be updated very frequently. It is modular, using the REDCap platform, e.g. the core data set (the core module) is common to all patients, but each center or patient group can add (locally) other data fields for its own use. The major achievement of this deliverable is the proposal of a research prone clinical database which will hopefully include all available patients through participation of HCPs and patient groups included in EpiCARE. Pending issues are as follows: 1) anonymization process (the database should be provided with anonymized codes so that duplication of data by different centers would be impossible), 2) accessibility (the database should be made accessible only after duly signed informed consent forms have been approved and adopted by all centres) 3) ethical approval by local boards 5 Conclusions A proposal for a research prone clinical database has been set up through consensus in the context of the EpiCARE Steering Committee. This database represents the first step to coordinate efforts for applications to European research calls for the benefit of individuals with rare disorders, which is one of the main objectives of EpiCARE. 6 Bibliography / References Lanthaler B, Wieser S, Deutschmann A, Schossig A, Fauth C, Zschocke J, Witsch-Baumgartner M. Genotype-based databases for variants causing rare diseases. Gene. 2014;550(1): PU Page 7 Version 0.2
8 ANNEX 1. EPICARE clinical database CORE MODULE informed consent given yes/no European new consent form : 3 parts Demographics unique patient identifier date of birth sex male female dead/alive specify date and cause of death (SUDEP?) date database entry date last update entry current age years/months/days automatic calculation: current date - birth date age epilepsy diagnosis years/months/days for children< 1year and neonates months and days are possible etiology of the epilepsy structural genetic MRI available? DNA available? specify (possibly with drop down menu) specify metabolic specify immune specify infectious specify unknown epilepsy syndrome not known known drop down list, see epi25k encephalopathy intellectual disability yes/no yes/no treatment AED specify generic names (drop down list) ketogenic diet start date / stop date if stopped VNS specify implantation date surgery specify date and type of surgery PU Page 8 Version 0.2
UAB P30 CORE A: The Hepato-Renal Fibrocystic Diseases Translational Resource
 PKD Foundation UAB P30 CORE A: The Hepato-Renal Fibrocystic Diseases Translational Resource http://www.arpkdstudies.uab.edu/ Director: Co-Director: Lisa M. Guay-Woodford, MD William E. Grizzle, MD, PhD
PKD Foundation UAB P30 CORE A: The Hepato-Renal Fibrocystic Diseases Translational Resource http://www.arpkdstudies.uab.edu/ Director: Co-Director: Lisa M. Guay-Woodford, MD William E. Grizzle, MD, PhD
A European Reference Network for rare and complex epilepsies. J Helen Cross Coordinator
 A European Reference Network for rare and complex epilepsies J Helen Cross Coordinator The epilepsies a group of rare diseases Early myoclonic encephalopathy PIGA, SETBP1, SIK1, SLC25A22 Dravet syndrome
A European Reference Network for rare and complex epilepsies J Helen Cross Coordinator The epilepsies a group of rare diseases Early myoclonic encephalopathy PIGA, SETBP1, SIK1, SLC25A22 Dravet syndrome
EUROPEAN COMMISSION HEALTH AND CONSUMERS DIRECTORATE-GENERAL INTERIM REPORT 02_2013
 EUROPEAN COMMISSION HEALTH AND CONSUMERS DIRECTORATE-GENERAL EXECUTIVE AGENCY FOR HEALTH AND CONSUMERS Health unit Contract number: 2010 21 02 Acronym: QUANDHIP INTERIM REPORT 02_2013 Proposal title: Quality
EUROPEAN COMMISSION HEALTH AND CONSUMERS DIRECTORATE-GENERAL EXECUTIVE AGENCY FOR HEALTH AND CONSUMERS Health unit Contract number: 2010 21 02 Acronym: QUANDHIP INTERIM REPORT 02_2013 Proposal title: Quality
Meeting with EMA: cooperation in the field of ERNs. Enrique Terol DG SANTE European Commission
 Meeting with EMA: cooperation in the field of ERNs Enrique Terol DG SANTE European Commission London 16 May 2017 The road to European Reference Networks (ERNs) Imagine if the best specialists from across
Meeting with EMA: cooperation in the field of ERNs Enrique Terol DG SANTE European Commission London 16 May 2017 The road to European Reference Networks (ERNs) Imagine if the best specialists from across
Gesundheit in Horizont 2020
 Wien, 5, Dezember 2013 Gesundheit in Horizont 2020 Gesellschaftliche Herausforderung 1: Gesundheit, demografischer Wandel und Wohlergehen Dr Elmar Nimmesgern GD Forschung und Innovation Europäische Kommission
Wien, 5, Dezember 2013 Gesundheit in Horizont 2020 Gesellschaftliche Herausforderung 1: Gesundheit, demografischer Wandel und Wohlergehen Dr Elmar Nimmesgern GD Forschung und Innovation Europäische Kommission
Fit4RareFit4All Conference Cluj - Napoca 3-4 September 2017
 Fit4RareFit4All Conference Cluj - Napoca 3-4 September 2017 Rare diseases registries as key tools in research and public health Domenica Taruscio Director, National Centre for Rare Diseases Istituto Superiore
Fit4RareFit4All Conference Cluj - Napoca 3-4 September 2017 Rare diseases registries as key tools in research and public health Domenica Taruscio Director, National Centre for Rare Diseases Istituto Superiore
ECIBC overview. Donata LERDA Healthcare Quality Team leader
 ECIBC overview Donata LERDA Healthcare Quality Team leader Joint Research Centre Institute for Health and Consumer Protection Public Health Policy Support Disclaimer: The contents of this presentation
ECIBC overview Donata LERDA Healthcare Quality Team leader Joint Research Centre Institute for Health and Consumer Protection Public Health Policy Support Disclaimer: The contents of this presentation
Country Participation: An Overview for Interested Investigators
 Country Participation: An Overview for Interested Investigators 1 Introduction This document provides an overview of the Peritoneal Dialysis Outcomes and Practice Patterns Study (PDOPPS). If you have any
Country Participation: An Overview for Interested Investigators 1 Introduction This document provides an overview of the Peritoneal Dialysis Outcomes and Practice Patterns Study (PDOPPS). If you have any
Criteria for the evaluation of crime prevention practices
 European Crime Prevention Network Criteria for the evaluation of crime prevention practices QUALIPREV short manual October 2016 RESEARCHER Anneleen Rummens PROMOTORS Prof. dr. Wim Hardyns Prof. dr. Freya
European Crime Prevention Network Criteria for the evaluation of crime prevention practices QUALIPREV short manual October 2016 RESEARCHER Anneleen Rummens PROMOTORS Prof. dr. Wim Hardyns Prof. dr. Freya
rare diseases research through National Plans and Strategies
 Recommendations to support rare diseases research through National Plans and Strategies Dr Domenica Taruscio EUROPLAN Coordinator Director of the Italian Centre for Rare Diseases, Italian National Institute
Recommendations to support rare diseases research through National Plans and Strategies Dr Domenica Taruscio EUROPLAN Coordinator Director of the Italian Centre for Rare Diseases, Italian National Institute
VectorNet. Pirbright Institute. Bulent Alten
 VectorNet Pirbright Institute Bulent Alten Network project What is VectorNet? Inter-institutional project (EFSA-ECDC) Vectors of pathogens affecting human and/or animal health Field work driven by: Current
VectorNet Pirbright Institute Bulent Alten Network project What is VectorNet? Inter-institutional project (EFSA-ECDC) Vectors of pathogens affecting human and/or animal health Field work driven by: Current
Cancer Research in the EU Framework Programmes for RTD
 Cancer Research in the EU Framework Programmes for RTD Action against cancer: European platform Maria Vidal, MD, PhD Deputy Head of Unit Medical and public health research Health Directorate DG Research
Cancer Research in the EU Framework Programmes for RTD Action against cancer: European platform Maria Vidal, MD, PhD Deputy Head of Unit Medical and public health research Health Directorate DG Research
Deliverable. Grant Agreement number: Open Access Policy Alignment STrategies for European Union Research. FP7 CAPACITIES Science in Society
 Deliverable Grant Agreement number: 611742 Project acronym: PASTEUR4OA Project title: Open Access Policy Alignment STrategies for European Union Research Funding Scheme: FP7 CAPACITIES Science in Society
Deliverable Grant Agreement number: 611742 Project acronym: PASTEUR4OA Project title: Open Access Policy Alignment STrategies for European Union Research Funding Scheme: FP7 CAPACITIES Science in Society
Scheduling a Course as a Professional Development Event
 Scheduling a Course as a Professional Development Event Scheduling an approved course as a professional development event is the second step you will need to complete in order to have your trainings reviewed
Scheduling a Course as a Professional Development Event Scheduling an approved course as a professional development event is the second step you will need to complete in order to have your trainings reviewed
RECOMMENDATION ADOPTED BY THE INTERGOVERNMENTAL COMMITTEE FOR THE NAGOYA PROTOCOL AT ITS FIRST MEETING
 CBD Distr. GENERAL 21 July 2011 OPEN-ENDED AD HOC INTERGOVERNMENTAL COMMITTEE FOR THE NAGOYA PROTOCOL ON ACCESS TO GENETIC RESOURCES AND THE FAIR AND EQUITABLE SHARING OF BENEFITS ARISING FROM THEIR UTILIZATION
CBD Distr. GENERAL 21 July 2011 OPEN-ENDED AD HOC INTERGOVERNMENTAL COMMITTEE FOR THE NAGOYA PROTOCOL ON ACCESS TO GENETIC RESOURCES AND THE FAIR AND EQUITABLE SHARING OF BENEFITS ARISING FROM THEIR UTILIZATION
NASDAQ: ZGNX. Company Presentation. October 2017
 NASDAQ: ZGNX Company Presentation October 2017 2 Forward Looking Statement Zogenix cautions you that statements included in this presentation that are not a description of historical facts are forward-looking
NASDAQ: ZGNX Company Presentation October 2017 2 Forward Looking Statement Zogenix cautions you that statements included in this presentation that are not a description of historical facts are forward-looking
Title 1 Descriptive title identifying the study design, population, interventions, and, if applicable, trial acronym
 SPIRIT 2013 Checklist: Recommended items to address in a clinical trial protocol and related documents* Section/item Item No Description Addressed on page number Administrative information Title 1 Descriptive
SPIRIT 2013 Checklist: Recommended items to address in a clinical trial protocol and related documents* Section/item Item No Description Addressed on page number Administrative information Title 1 Descriptive
PROPOSED WORK PROGRAMME FOR THE CLEARING-HOUSE MECHANISM IN SUPPORT OF THE STRATEGIC PLAN FOR BIODIVERSITY Note by the Executive Secretary
 CBD Distr. GENERAL UNEP/CBD/COP/11/31 30 July 2012 ORIGINAL: ENGLISH CONFERENCE OF THE PARTIES TO THE CONVENTION ON BIOLOGICAL DIVERSITY Eleventh meeting Hyderabad, India, 8 19 October 2012 Item 3.2 of
CBD Distr. GENERAL UNEP/CBD/COP/11/31 30 July 2012 ORIGINAL: ENGLISH CONFERENCE OF THE PARTIES TO THE CONVENTION ON BIOLOGICAL DIVERSITY Eleventh meeting Hyderabad, India, 8 19 October 2012 Item 3.2 of
Business Contributions to Setting New Research Agendas: Business Platform for Nutrition Research (BPNR)
 Business Contributions to Setting New Research Agendas: Business Platform for Nutrition Research (BPNR) Purpose How can we promote more investment into public research? How can we promote improved uptake
Business Contributions to Setting New Research Agendas: Business Platform for Nutrition Research (BPNR) Purpose How can we promote more investment into public research? How can we promote improved uptake
Global reporting system for hepatitis (GRSH) data approval manual
 Global reporting system for hepatitis (GRSH) data approval manual Contents Acknowledgement... 1 General information... 2 1. Logging in... 2 2. Validating aggregated numbers in the reports application...
Global reporting system for hepatitis (GRSH) data approval manual Contents Acknowledgement... 1 General information... 2 1. Logging in... 2 2. Validating aggregated numbers in the reports application...
1. The Working Party on Public Health discussed and agreed the draft Council conclusions as set out in the Annex.
 Council of the European Union Brussels, 26 November 2015 (OR. en) 14395/15 SAN 391 NOTE From: To: Subject: General Secretariat of the Council Permanent Representatives Committee/Council Employment, Social
Council of the European Union Brussels, 26 November 2015 (OR. en) 14395/15 SAN 391 NOTE From: To: Subject: General Secretariat of the Council Permanent Representatives Committee/Council Employment, Social
A European Quality Assurance Scheme for Breast Cancer Services - updates Joint Research Centre
 A European Quality Assurance Scheme for Breast Cancer Services - updates Joint Research Centre Institute for Health and Consumer Protection Public Health Policy Support Donata LERDA Disclaimer: The contents
A European Quality Assurance Scheme for Breast Cancer Services - updates Joint Research Centre Institute for Health and Consumer Protection Public Health Policy Support Donata LERDA Disclaimer: The contents
INTERNATIONAL SYMPOSIUM DRAVET SYNDROME AND OTHER SODIUM CHANNEL RELATED ENCEPHALOPATHIES
 4 HORIZONS FOR DRAVET SYNDROME INTERNATIONAL SYMPOSIUM DRAVET SYNDROME AND OTHER SODIUM CHANNEL RELATED ENCEPHALOPATHIES 15-16 MARCH 2018, VERONA PALAZZO DELLA GRAN GUARDIA info@horizonsdravet.eu www.horizonsdravet.eu
4 HORIZONS FOR DRAVET SYNDROME INTERNATIONAL SYMPOSIUM DRAVET SYNDROME AND OTHER SODIUM CHANNEL RELATED ENCEPHALOPATHIES 15-16 MARCH 2018, VERONA PALAZZO DELLA GRAN GUARDIA info@horizonsdravet.eu www.horizonsdravet.eu
First Interim Report to the European Commission DG-SANCO for: Grant Agreement No.: (790655) EAIS. December Annex 2
 First Interim Report to the European Commission DG-SANCO for: Grant Agreement.:2005112 (790655) EAIS. December 2006 Annex 2 European Autism Information System (EAIS) Project: Design of a Prevalence study.
First Interim Report to the European Commission DG-SANCO for: Grant Agreement.:2005112 (790655) EAIS. December 2006 Annex 2 European Autism Information System (EAIS) Project: Design of a Prevalence study.
11. NATIONAL DAFNE CLINICAL AND RESEARCH DATABASE
 11. NATIONAL DAFNE CLINICAL AND RESEARCH DATABASE The National DAFNE Clinical and Research database was set up as part of the DAFNE QA programme (refer to section 12) to facilitate Audit and was updated
11. NATIONAL DAFNE CLINICAL AND RESEARCH DATABASE The National DAFNE Clinical and Research database was set up as part of the DAFNE QA programme (refer to section 12) to facilitate Audit and was updated
Submission Date: 27 January 2010 at: 12:44
 Submission Date: 27 January 2010 at: 12:44 Questionnaire about Institutions partners SECTION 1 Official name 1 1 Provide the name of the organization such as the Ministry of Health or Public Health Institute
Submission Date: 27 January 2010 at: 12:44 Questionnaire about Institutions partners SECTION 1 Official name 1 1 Provide the name of the organization such as the Ministry of Health or Public Health Institute
NHS public health functions agreement Service specification No.1 Neonatal hepatitis B immunisation programme
 NHS public health functions agreement 2017-18 Service specification No.1 Neonatal hepatitis B immunisation programme NHS public health functions agreement 2017-18 Service specification No.1Neonatal hepatitis
NHS public health functions agreement 2017-18 Service specification No.1 Neonatal hepatitis B immunisation programme NHS public health functions agreement 2017-18 Service specification No.1Neonatal hepatitis
EFSA and Member States
 Scientific Cooperation between EFSA and Member States Taking Stock and Looking Ahead Committed to ensuring that Europe s food is safe Scientific Cooperation between EFSA and Member States Scientific cooperation
Scientific Cooperation between EFSA and Member States Taking Stock and Looking Ahead Committed to ensuring that Europe s food is safe Scientific Cooperation between EFSA and Member States Scientific cooperation
Monitoring Public Health Nutrition in Europe Appendix X (Page 1 of 4) Technical Report, Agreement SI (2000CVG3-507)
 Monitoring Public Health Nutrition in Europe Appendix X (Page 1 of 4) EFCOSUM; Executive summary Introduction The project European Food Consumption Survey Method (EFCOSUM) was undertaken within the framework
Monitoring Public Health Nutrition in Europe Appendix X (Page 1 of 4) EFCOSUM; Executive summary Introduction The project European Food Consumption Survey Method (EFCOSUM) was undertaken within the framework
(Legislative acts) REGULATIONS
 31.12.2010 Official Journal of the European Union L 348/1 I (Legislative acts) REGULATIONS REGULATION (EU) No 1235/2010 OF THE EUROPEAN PARLIAMENT AND OF THE COUNCIL of 15 December 2010 amending, as regards
31.12.2010 Official Journal of the European Union L 348/1 I (Legislative acts) REGULATIONS REGULATION (EU) No 1235/2010 OF THE EUROPEAN PARLIAMENT AND OF THE COUNCIL of 15 December 2010 amending, as regards
POSITION DESCRIPTION. Employment Status: Maternity Leave Contract 5 months (Jan-May 2018) PURPOSE OF POSITION DESCRIPTION
 POSITION DESCRIPTION Position Title: Location: Research Coordinator Clinical Trials National Secretariat, Brisbane-based Employment Status: Maternity Leave Contract 5 months (Jan-May 2018) Reporting Relationships:
POSITION DESCRIPTION Position Title: Location: Research Coordinator Clinical Trials National Secretariat, Brisbane-based Employment Status: Maternity Leave Contract 5 months (Jan-May 2018) Reporting Relationships:
NHS public health functions agreement Service specification No.11 Human papillomavirus (HPV) programme
 NHS public health functions agreement 2018-19 Service specification No.11 Human papillomavirus (HPV) programme 1 NHS public health functions agreement 2018-19 Service specification No.11 Human papillomavirus
NHS public health functions agreement 2018-19 Service specification No.11 Human papillomavirus (HPV) programme 1 NHS public health functions agreement 2018-19 Service specification No.11 Human papillomavirus
RESEARCH AND PRACTICE IN ALZHEIMER S DISEASE VOL 10 EADC OVERVIEW B. VELLAS & E. REYNISH
 EADC BRUNO VELLAS 14/01/05 10:14 Page 1 EADC OVERVIEW B. VELLAS & E. REYNISH (Toulouse, France, EU) Bruno Vellas: The European Alzheimer's Disease Consortium is a European funded network of centres of
EADC BRUNO VELLAS 14/01/05 10:14 Page 1 EADC OVERVIEW B. VELLAS & E. REYNISH (Toulouse, France, EU) Bruno Vellas: The European Alzheimer's Disease Consortium is a European funded network of centres of
A practical approach to the E-IMD patient registry (WP4) (Deliverable #6, milestones #5-7)
 A practical approach to the E-IMD patient registry (WP4) (Deliverable #6, milestones #5-7) Major aims of the registry (1) To describe the natural history and outcome of OADs and UCDs (2) To describe and
A practical approach to the E-IMD patient registry (WP4) (Deliverable #6, milestones #5-7) Major aims of the registry (1) To describe the natural history and outcome of OADs and UCDs (2) To describe and
COMMISSION REGULATION (EU) / of XXX
 Ref. Ares(2016)5616438-28/09/2016 EUROPEAN COMMISSION Brussels, XXX [ ](2016) XXX draft COMMISSION REGULATION (EU) / of XXX amending Annex III to Directive 2008/98/EC of the European Parliament and of
Ref. Ares(2016)5616438-28/09/2016 EUROPEAN COMMISSION Brussels, XXX [ ](2016) XXX draft COMMISSION REGULATION (EU) / of XXX amending Annex III to Directive 2008/98/EC of the European Parliament and of
ProScript User Guide. Pharmacy Access Medicines Manager
 User Guide Pharmacy Access Medicines Manager Version 3.0.0 Release Date 01/03/2014 Last Reviewed 11/04/2014 Author Rx Systems Service Desk (T): 01923 474 600 Service Desk (E): servicedesk@rxsystems.co.uk
User Guide Pharmacy Access Medicines Manager Version 3.0.0 Release Date 01/03/2014 Last Reviewed 11/04/2014 Author Rx Systems Service Desk (T): 01923 474 600 Service Desk (E): servicedesk@rxsystems.co.uk
REGIONAL ALLIANCE FOR NATIONAL REGULATORY AUTHORITIES FOR VACCINES IN THE WESTERN PACIFIC. second edition
 REGIONAL ALLIANCE FOR NATIONAL REGULATORY AUTHORITIES FOR VACCINES IN THE WESTERN PACIFIC second edition World Health Organization 2014 The designations employed and the presentation of the material in
REGIONAL ALLIANCE FOR NATIONAL REGULATORY AUTHORITIES FOR VACCINES IN THE WESTERN PACIFIC second edition World Health Organization 2014 The designations employed and the presentation of the material in
E-RARE & THE EUROPEAN JOINT PROGRAMME ON RARE DISEASES. RE(ACT) 2018 Congress 7-10 of March 2018 Bologna, ITALY
 E-RARE & THE EUROPEAN JOINT PROGRAMME ON RARE DISEASES RE(ACT) 2018 Congress 7-10 of March 2018 Bologna, ITALY E-RARE THE ERA-NET FOR RESEARCH PROGRAMMES ON RARE DISEASES MAJOR FACTS ABOUT E-RARE ERA-NET
E-RARE & THE EUROPEAN JOINT PROGRAMME ON RARE DISEASES RE(ACT) 2018 Congress 7-10 of March 2018 Bologna, ITALY E-RARE THE ERA-NET FOR RESEARCH PROGRAMMES ON RARE DISEASES MAJOR FACTS ABOUT E-RARE ERA-NET
EUROPA DONNA Advocates Guide to the ECIBC European Commission Initiative on Breast Cancer
 EUROPA DONNA Advocates Guide to the ECIBC European Commission Initiative on Breast Cancer Part 1 About EUROPA DONNA EUROPA DONNA The European Breast Cancer Coalition is an independent non-profit organisation
EUROPA DONNA Advocates Guide to the ECIBC European Commission Initiative on Breast Cancer Part 1 About EUROPA DONNA EUROPA DONNA The European Breast Cancer Coalition is an independent non-profit organisation
WHO s code of conduct for open and timely sharing of pathogen genetic sequence data during outbreaks of infectious disease
 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 27 28 29 30 31 32 33 34 WHO s code of conduct for open and timely sharing of pathogen genetic sequence data during outbreaks of infectious
1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 27 28 29 30 31 32 33 34 WHO s code of conduct for open and timely sharing of pathogen genetic sequence data during outbreaks of infectious
Real world Outcomes across the AD spectrum for better care: Multi-modal data Access Platform
 Real world Outcomes across the AD spectrum for better care: Multi-modal data Access Platform Catherine Reed, Lilly on behalf of ROADMAP consortium 3 rd Nordic conference on Real World Data, 28-29 November
Real world Outcomes across the AD spectrum for better care: Multi-modal data Access Platform Catherine Reed, Lilly on behalf of ROADMAP consortium 3 rd Nordic conference on Real World Data, 28-29 November
COMMISSION REGULATION (EU) / of XXX
 EUROPEAN COMMISSION Brussels, XXX SANTE/11992/2017 Rev. 0 [ ](2017) XXX draft COMMISSION REGULATION (EU) / of XXX amending Annex II to Regulation (EC) No 1107/2009 by setting out scientific criteria for
EUROPEAN COMMISSION Brussels, XXX SANTE/11992/2017 Rev. 0 [ ](2017) XXX draft COMMISSION REGULATION (EU) / of XXX amending Annex II to Regulation (EC) No 1107/2009 by setting out scientific criteria for
Practical guidance for applicants on the submission of applications on food additives, food enzymes and food flavourings
 Version 2 Updated on 29/11/2011 Practical guidance for applicants on the submission of applications on food additives, food enzymes and food flavourings Valid as of: 11 September 2011 Disclaimer: This
Version 2 Updated on 29/11/2011 Practical guidance for applicants on the submission of applications on food additives, food enzymes and food flavourings Valid as of: 11 September 2011 Disclaimer: This
NHS public health functions agreement
 NHS public health functions agreement 2016-17 Service specification No.1 Neonatal hepatitis B immunisation programme Classification: official NHS England INFORMATION READER BOX Directorate Medical Commissioning
NHS public health functions agreement 2016-17 Service specification No.1 Neonatal hepatitis B immunisation programme Classification: official NHS England INFORMATION READER BOX Directorate Medical Commissioning
REPORT FROM THE COMMISSION TO THE EUROPEAN PARLIAMENT AND THE COUNCIL
 EUROPEAN COMMISSION Brussels, 22.8.2017 COM(2017) 438 final REPORT FROM THE COMMISSION TO THE EUROPEAN PARLIAMENT AND THE COUNCIL on the exercise of the delegation conferred on the Commission pursuant
EUROPEAN COMMISSION Brussels, 22.8.2017 COM(2017) 438 final REPORT FROM THE COMMISSION TO THE EUROPEAN PARLIAMENT AND THE COUNCIL on the exercise of the delegation conferred on the Commission pursuant
Actual submission date 29/03/2018
 Page 1 of 7 Project acronym PROMISS Project full title PRevention Of Malnutrition In Senior Subjects in the EU Grant agreement n 678732 Responsible AGE Platform Europe Contributors Coordinator, WP9 partners
Page 1 of 7 Project acronym PROMISS Project full title PRevention Of Malnutrition In Senior Subjects in the EU Grant agreement n 678732 Responsible AGE Platform Europe Contributors Coordinator, WP9 partners
The PhenX Toolkit: Standard Measures for Collaborative Research
 The PhenX Toolkit: Standard Measures for Collaborative Research Clinical Common Data Elements Task Force (CDETF) November 4, 2016 Carol M. Hamilton, PhD RTI International is a trade name of Research Triangle
The PhenX Toolkit: Standard Measures for Collaborative Research Clinical Common Data Elements Task Force (CDETF) November 4, 2016 Carol M. Hamilton, PhD RTI International is a trade name of Research Triangle
SETPEG GENETIC TESTING GUIDELINES Version 1.0, 5 th October 2017
 SETPEG GENETIC TESTING GUIDELINES Version 1.0, 5 th October 2017 1. The Epilepsy Genetic Diagnostic & Counselling Service at King s Health Partners Professor Deb Pal PhD MRCP (Consultant) deb.pal@nhs.net
SETPEG GENETIC TESTING GUIDELINES Version 1.0, 5 th October 2017 1. The Epilepsy Genetic Diagnostic & Counselling Service at King s Health Partners Professor Deb Pal PhD MRCP (Consultant) deb.pal@nhs.net
American Joint Replacement Registry Introductory Webinar for the National Association of Orthopaedic Nurses
 American Joint Replacement Registry Introductory Webinar for the National Association of Orthopaedic Nurses June 8, 2017 *Pick up your copy of the slides on our website www.ajrr.net under Events & Webinars*
American Joint Replacement Registry Introductory Webinar for the National Association of Orthopaedic Nurses June 8, 2017 *Pick up your copy of the slides on our website www.ajrr.net under Events & Webinars*
Epilepsy Syndromes: Where does Dravet Syndrome fit in?
 Epilepsy Syndromes: Where does Dravet Syndrome fit in? Scott Demarest MD Assistant Professor, Departments of Pediatrics and Neurology University of Colorado School of Medicine Children's Hospital Colorado
Epilepsy Syndromes: Where does Dravet Syndrome fit in? Scott Demarest MD Assistant Professor, Departments of Pediatrics and Neurology University of Colorado School of Medicine Children's Hospital Colorado
The EU rare diseases policy framework: the context for ERNs
 The EU rare diseases policy framework: the context for ERNs Jaroslaw Waligora DG Health and Food Safety, European Commission Unit C.1, Programme Management and Diseases Report from an expert group of the
The EU rare diseases policy framework: the context for ERNs Jaroslaw Waligora DG Health and Food Safety, European Commission Unit C.1, Programme Management and Diseases Report from an expert group of the
Site Training. The 39 th Multicenter Airway Research Collaboration (MARC-39) Study
 Site Training The 39 th Multicenter Airway Research Collaboration (MARC-39) Study 1 Study Leadership Kohei Hasegawa, MD, MPH (PI) Carlos Camargo, MD, DrPH (Co-I) Project Coordination: Catalina Gimenez-Zapiola
Site Training The 39 th Multicenter Airway Research Collaboration (MARC-39) Study 1 Study Leadership Kohei Hasegawa, MD, MPH (PI) Carlos Camargo, MD, DrPH (Co-I) Project Coordination: Catalina Gimenez-Zapiola
SOCIABLE DELIVERABLE D7.2a Interim Assessment of the SOCIABLE Platform and Services
 SOCIABLE ICT-PSP No. 238891 SOCIABLE DELIVERABLE D7.2a Interim Assessment of the SOCIABLE Platform and Services Project Acronym SOCIABLE Grant Agreement No. 238891 Project Title Motivating platform for
SOCIABLE ICT-PSP No. 238891 SOCIABLE DELIVERABLE D7.2a Interim Assessment of the SOCIABLE Platform and Services Project Acronym SOCIABLE Grant Agreement No. 238891 Project Title Motivating platform for
ClinicalTrials.gov a programmer s perspective
 PhUSE 2009, Basel ClinicalTrials.gov a programmer s perspective Ralf Minkenberg Boehringer Ingelheim Pharma GmbH & Co. KG PhUSE 2009, Basel 1 Agenda ClinicalTrials.gov Basic Results disclosure Initial
PhUSE 2009, Basel ClinicalTrials.gov a programmer s perspective Ralf Minkenberg Boehringer Ingelheim Pharma GmbH & Co. KG PhUSE 2009, Basel 1 Agenda ClinicalTrials.gov Basic Results disclosure Initial
Report on the Transplantation Transmission Sentinel Network (TTSN)
 Report on the Transplantation Transmission Sentinel Network (TTSN) CATB Session - AATB s 31st Annual Meeting, Boston September 15, 2007 Scott Brubaker, CTBS AATB Representative, TTSN Advisory Group TTSN
Report on the Transplantation Transmission Sentinel Network (TTSN) CATB Session - AATB s 31st Annual Meeting, Boston September 15, 2007 Scott Brubaker, CTBS AATB Representative, TTSN Advisory Group TTSN
PanCareSurFup Bulletin, Issue nr. 5
 March 2014 PanCareSurFup Bulletin, Issue nr. 5 PanCare Childhood and Adolescent Cancer Survivor Care and Follow-Up Studies Dear Friends, Since the last Bulletin in December 2013, the project has been busy
March 2014 PanCareSurFup Bulletin, Issue nr. 5 PanCare Childhood and Adolescent Cancer Survivor Care and Follow-Up Studies Dear Friends, Since the last Bulletin in December 2013, the project has been busy
Workshop on Extrapolation and Evidence Synthesis in the Development and Therapeutic Use of Medicines in Children
 Workshop on Extrapolation and Evidence Synthesis in the Development and Therapeutic Use of Medicines in Children Dr Oscar Della Pasqua WP 4 11th June 2013 Strathclyde University, Glasgow, UK Project AIMS
Workshop on Extrapolation and Evidence Synthesis in the Development and Therapeutic Use of Medicines in Children Dr Oscar Della Pasqua WP 4 11th June 2013 Strathclyde University, Glasgow, UK Project AIMS
National Surveillance System for Dialysis Centre and Dialysis Associated Diseases
 National Surveillance System for Dialysis Centre and Dialysis Associated Diseases Dr Richard Fluck Consultant Renal Physician & Clinical Director, Royal Derby Hospital UK RA Vascular Access Survey 2005
National Surveillance System for Dialysis Centre and Dialysis Associated Diseases Dr Richard Fluck Consultant Renal Physician & Clinical Director, Royal Derby Hospital UK RA Vascular Access Survey 2005
MEAT CONTENT CALCULATION
 MEAT CONTENT CALCULATION Introduction Amendments to the European Labelling Directive have resulted in the need to harmonise the definition of meat across Europe. This new definition attempts to ensure
MEAT CONTENT CALCULATION Introduction Amendments to the European Labelling Directive have resulted in the need to harmonise the definition of meat across Europe. This new definition attempts to ensure
Discovery and validation of novel. Webinar IMI2 - Call 9 Joint influenza vaccine effectiveness studies
 Discovery and validation of novel Webinar IMI2 - Call 9 Joint influenza vaccine effectiveness studies Dr. Angela Wittelsberger, IMI IMI webinar 29.04.2016 Today s webinar Will cover all aspects of the
Discovery and validation of novel Webinar IMI2 - Call 9 Joint influenza vaccine effectiveness studies Dr. Angela Wittelsberger, IMI IMI webinar 29.04.2016 Today s webinar Will cover all aspects of the
Cochrane Rehabilitation Report sullo stato attuale di Cochrane Rehabilitation field e prospettive future nell ottica di ISPRM
 Cochrane Rehabilitation Report sullo stato attuale di Cochrane Rehabilitation field e prospettive future nell ottica di ISPRM Francesca Gimigliano, MD, PhD Cochrane Rehabilitation Communication Committee
Cochrane Rehabilitation Report sullo stato attuale di Cochrane Rehabilitation field e prospettive future nell ottica di ISPRM Francesca Gimigliano, MD, PhD Cochrane Rehabilitation Communication Committee
SUNFRAIL PROJECT Understanding and Caring for Frailty and Multimorbidity. Speaker/Conference/Date
 SUNFRAIL PROJECT Understanding and Caring for Frailty and Multimorbidity Speaker/Conference/Date ACKNOWLEDGEMENT This presentation is part of SUNFRAIL (project 664291), which has received funding from
SUNFRAIL PROJECT Understanding and Caring for Frailty and Multimorbidity Speaker/Conference/Date ACKNOWLEDGEMENT This presentation is part of SUNFRAIL (project 664291), which has received funding from
The EU PIP - a step in Pediatric Drug Development. Thomas Severin Bonn,
 The EU PIP - a step in Pediatric Drug Development Thomas Severin Bonn, 13.01.2009 Agenda Implications for Industry Company Preparation Time of PIP Submission Content of the PIP The PIP Process and first
The EU PIP - a step in Pediatric Drug Development Thomas Severin Bonn, 13.01.2009 Agenda Implications for Industry Company Preparation Time of PIP Submission Content of the PIP The PIP Process and first
2013 REPORT ON THE STATE OF THE ART OF RARE DISEASE ACTIVITIES IN EUROPE
 2013 REPORT ON THE STATE OF THE ART OF RARE DISEASE ACTIVITIES IN EUROPE STATE OF THE ART OF RARE DISEASE ACTIVITIES IN CYPRUS This work was financed by the EUCERD Joint Action: Working for Rare Diseases
2013 REPORT ON THE STATE OF THE ART OF RARE DISEASE ACTIVITIES IN EUROPE STATE OF THE ART OF RARE DISEASE ACTIVITIES IN CYPRUS This work was financed by the EUCERD Joint Action: Working for Rare Diseases
Measuring the impact of patient and public involvement in the JLA Kidney Transplant PSP
 Measuring the impact of patient and public involvement in the JLA Kidney Transplant PSP Contents Measuring the impact of patient and public involvement in the JLA Kidney Transplant PSP... 1 Overview and
Measuring the impact of patient and public involvement in the JLA Kidney Transplant PSP Contents Measuring the impact of patient and public involvement in the JLA Kidney Transplant PSP... 1 Overview and
WP01 Longitudinal Onset and Course Cohort Studies (Leader: UCL / A. Schrag): Objectives
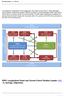 To facilitate the organisation and management, the project is structured in Work packages which together comprise the project. EMTICS consists of 12 work packages (WPs). Each WP is carried out by a varying
To facilitate the organisation and management, the project is structured in Work packages which together comprise the project. EMTICS consists of 12 work packages (WPs). Each WP is carried out by a varying
No relevant disclosures
 No relevant disclosures - Epileptic Encephalopathy (EE): Epileptic activity itself contributes to cognitive and behavioural impairments - Developmental and Epileptic Encephalopathy (DEE): Impairments occur
No relevant disclosures - Epileptic Encephalopathy (EE): Epileptic activity itself contributes to cognitive and behavioural impairments - Developmental and Epileptic Encephalopathy (DEE): Impairments occur
PET Steering Committee Meeting Minutes. Wednesday, January 20 th, 2016 Time: 1:30-3:30pm
 PET Steering Committee Meeting Minutes Wednesday, January 20 th, 2016 Time: 1:30-3:30pm Committee Members Present: U. Metser (Chair), R. dekemp, M. Freeman, M. Greenberg, D. Hussey, A. Singnurkar, Y. Ung
PET Steering Committee Meeting Minutes Wednesday, January 20 th, 2016 Time: 1:30-3:30pm Committee Members Present: U. Metser (Chair), R. dekemp, M. Freeman, M. Greenberg, D. Hussey, A. Singnurkar, Y. Ung
Table Of Content. Appropriate care paths for frail elderly patients: a comprehensive model... 2 Summary... 3 Work Package... 7
 Table Of Content Appropriate care paths for frail elderly patients: a comprehensive model... 2 Summary... 3 Work Package... 7 Coordination of the project... 7 Dissemination of the project... 7 Evaluation
Table Of Content Appropriate care paths for frail elderly patients: a comprehensive model... 2 Summary... 3 Work Package... 7 Coordination of the project... 7 Dissemination of the project... 7 Evaluation
Market surveillance of medical devices
 Market surveillance of medical devices A joint action on market surveillance of medical devices to reinforce public health protection Information for healthcare professionals Introduction The European
Market surveillance of medical devices A joint action on market surveillance of medical devices to reinforce public health protection Information for healthcare professionals Introduction The European
Performance Indicators for Foodborne Disease Programs
 Chapter A long-standing goal of CDC and national public health professional organizations has been to build state and local capacity for detecting and preventing illness. In 1997, CDC convened an expert
Chapter A long-standing goal of CDC and national public health professional organizations has been to build state and local capacity for detecting and preventing illness. In 1997, CDC convened an expert
SUMMARY REPORT OF THE STANDING COMMITTEE ON THE FOOD CHAIN AND ANIMAL HEALTH HELD IN BRUSSELS ON 10 DECEMBER 2012 (Section General Food Law)
 EUROPEAN COMMISSION HEALTH & CONSUMERS DIRECTORATE-GENERAL Brussels, SANCO E 1718316 SUMMARY REPORT OF THE STANDING COMMITTEE ON THE FOOD CHAIN AND ANIMAL HEALTH HELD IN BRUSSELS ON 10 DECEMBER 2012 (Section
EUROPEAN COMMISSION HEALTH & CONSUMERS DIRECTORATE-GENERAL Brussels, SANCO E 1718316 SUMMARY REPORT OF THE STANDING COMMITTEE ON THE FOOD CHAIN AND ANIMAL HEALTH HELD IN BRUSSELS ON 10 DECEMBER 2012 (Section
Pre Accession Programme support and Capacity Building
 Pre Accession Programme support and Capacity Building Andrea Gross-Bošković Director of Croatian Food Agency 64 th meeting of the EFSA Advisory Forum Parma, 7 8 June 2017 History 2006. IPA Pre- Accession
Pre Accession Programme support and Capacity Building Andrea Gross-Bošković Director of Croatian Food Agency 64 th meeting of the EFSA Advisory Forum Parma, 7 8 June 2017 History 2006. IPA Pre- Accession
REGULATION (EC) No.141/2000
 REGULATION (EC) No.141/2000 Community legislation in force Document 300R0141 Directory chapters where this document can be found: [15.30 Health promotion] Regulation (EC) No.141/2000 of the European Parliament
REGULATION (EC) No.141/2000 Community legislation in force Document 300R0141 Directory chapters where this document can be found: [15.30 Health promotion] Regulation (EC) No.141/2000 of the European Parliament
COMMISSION REGULATION (EU) / of XXX
 EUROPEAN COMMISSION Brussels, XXX SANTE/131/2015 CIS REV2 (POOL/G4/2015/131/131-EN CIS.doc) [ ](2015) XXX draft COMMISSION REGULATION (EU) / of XXX amending Annex III and Annex IV to Regulation (EC) No
EUROPEAN COMMISSION Brussels, XXX SANTE/131/2015 CIS REV2 (POOL/G4/2015/131/131-EN CIS.doc) [ ](2015) XXX draft COMMISSION REGULATION (EU) / of XXX amending Annex III and Annex IV to Regulation (EC) No
COMMITTEE OF EXPERTS ON THE CLASSIFICATION OF MEDICINES AS REGARDS THEIR SUPPLY (CD-P-PH/PHO) PROGRAMME RESULTS
 COMMITTEE OF EXPERTS ON THE CLASSIFICATION OF MEDICINES AS REGARDS THEIR SUPPLY (CD-P-PH/PHO) PROGRAMME RESULTS 2016-2017 Introduction This summary provides an overview of the activities carried out by
COMMITTEE OF EXPERTS ON THE CLASSIFICATION OF MEDICINES AS REGARDS THEIR SUPPLY (CD-P-PH/PHO) PROGRAMME RESULTS 2016-2017 Introduction This summary provides an overview of the activities carried out by
COMMISSION OF THE EUROPEAN COMMUNITIES
 COMMISSION OF THE EUROPEAN COMMUNITIES Brussels, 22.12.2008 COM(2008) 882 final REPORT FROM THE COMMISSION TO THE COUNCIL, THE EUROPEAN PARLIAMENT, THE EUROPEAN ECONOMIC AND SOCIAL COMMITTEE AND THE COMMITTEE
COMMISSION OF THE EUROPEAN COMMUNITIES Brussels, 22.12.2008 COM(2008) 882 final REPORT FROM THE COMMISSION TO THE COUNCIL, THE EUROPEAN PARLIAMENT, THE EUROPEAN ECONOMIC AND SOCIAL COMMITTEE AND THE COMMITTEE
State of the Art of Rare Disease Activities in EU Member States and Other European Countries Croatia Report
 State of the Art of Rare Disease Activities in EU Member States and Other European Countries Croatia Report Definition of a Rare Disease Croatia has adopted the European Commission definition of a rare
State of the Art of Rare Disease Activities in EU Member States and Other European Countries Croatia Report Definition of a Rare Disease Croatia has adopted the European Commission definition of a rare
2014 REPORT ON THE STATE OF THE ART OF RARE DISEASE ACTIVITIES IN EUROPE
 2014 REPORT ON THE STATE OF THE ART OF RARE DISEASE ACTIVITIES IN EUROPE STATE OF THE ART OF RARE DISEASE ACTIVITIES IN CYPRUS This work was financed by the EUCERD Joint Action: Working for Rare Diseases
2014 REPORT ON THE STATE OF THE ART OF RARE DISEASE ACTIVITIES IN EUROPE STATE OF THE ART OF RARE DISEASE ACTIVITIES IN CYPRUS This work was financed by the EUCERD Joint Action: Working for Rare Diseases
CONGENITAL HEART PUBLIC HEALTH CONSORTIUM
 CONGENITAL HEART PUBLIC HEALTH CONSORTIUM Impact and opportunity of a public health approach to congenital heart defects WHO WE ARE In 2009, various organizations across federal, state and local communities
CONGENITAL HEART PUBLIC HEALTH CONSORTIUM Impact and opportunity of a public health approach to congenital heart defects WHO WE ARE In 2009, various organizations across federal, state and local communities
Using the NWD Integrative Screener as a Data Collection Tool Agency-Level Aggregate Workbook
 Using the NWD Integrative Screener as a Data Collection Tool Agency-Level Aggregate Workbook The NWD Integrative Screener was designed to be user friendly for providers and agencies. Many items and health
Using the NWD Integrative Screener as a Data Collection Tool Agency-Level Aggregate Workbook The NWD Integrative Screener was designed to be user friendly for providers and agencies. Many items and health
PREPARE. Herman Goossens, Coordinator. Panel discussion, GLoPID-R meeting, Sao Paulo, Brazil December 1, 2016
 PREPARE Herman Goossens, Coordinator Panel discussion, GLoPID-R meeting, Sao Paulo, Brazil December 1, 2016 PREPARE is funded by the European Commission under grant number 602525 Agenda Network structure
PREPARE Herman Goossens, Coordinator Panel discussion, GLoPID-R meeting, Sao Paulo, Brazil December 1, 2016 PREPARE is funded by the European Commission under grant number 602525 Agenda Network structure
B.I.R.O. Best Information through Regional Outcomes
 B.I.R.O. Best Information through Regional Outcomes The role of BIRO in the implementation of the Diabetes EU Policy Recommendations Fabrizio Carinci, BIRO Technical Coordinator, University of Perugia
B.I.R.O. Best Information through Regional Outcomes The role of BIRO in the implementation of the Diabetes EU Policy Recommendations Fabrizio Carinci, BIRO Technical Coordinator, University of Perugia
Innovation in accessibility. How UniEvangélica promoted the inclusion of 22 employees who are deaf or hard of hearing within its hearing community
 Innovation in accessibility How UniEvangélica promoted the inclusion of 22 employees who are deaf or hard of hearing within its hearing community Overview UniEvangélica is a higher-education institution
Innovation in accessibility How UniEvangélica promoted the inclusion of 22 employees who are deaf or hard of hearing within its hearing community Overview UniEvangélica is a higher-education institution
Programme of community action in the field of health ( ) 2012 ACTIVITY REPORT DELIVERABLE 8: EVALUATION REPORT
 Page 1 Programme of community action in the field of health (2008-2013) 2012 ACTIVITY REPORT DELIVERABLE 8: EVALUATION REPORT Page 2 Description of the deliverable An end of year external evaluation report
Page 1 Programme of community action in the field of health (2008-2013) 2012 ACTIVITY REPORT DELIVERABLE 8: EVALUATION REPORT Page 2 Description of the deliverable An end of year external evaluation report
Guideline on good pharmacovigilance practices (GVP)
 19 April 2013 EMA/169546/2012 Guideline on good pharmacovigilance practices (GVP) Module X Additional monitoring Draft finalised by the Agency in collaboration with Member States 25 May 2012 Draft agreed
19 April 2013 EMA/169546/2012 Guideline on good pharmacovigilance practices (GVP) Module X Additional monitoring Draft finalised by the Agency in collaboration with Member States 25 May 2012 Draft agreed
ATMPs & EU GMP Update. Bryan J Wright July 2017
 ATMPs & EU GMP Update Bryan J Wright July 2017 Slide 1 PharmOut 2017 Outline ATMPs What they are? Why are we looking at this subject? The Licensing position of ATMPs and use of PRIME ATMPs and GMP Inspections
ATMPs & EU GMP Update Bryan J Wright July 2017 Slide 1 PharmOut 2017 Outline ATMPs What they are? Why are we looking at this subject? The Licensing position of ATMPs and use of PRIME ATMPs and GMP Inspections
Legal Status and Regulations
 Legal Status and Regulations Presentation Prof. Dr. Vinjar Fønnebø Leader Work Package 2 Partner NAFKAM, University of Tromsø,Norway ComCAM, Ministry of Health, Italy KI, Karolinska Institutet, Sweden
Legal Status and Regulations Presentation Prof. Dr. Vinjar Fønnebø Leader Work Package 2 Partner NAFKAM, University of Tromsø,Norway ComCAM, Ministry of Health, Italy KI, Karolinska Institutet, Sweden
NEWSLETTER Partners. 43 Months WPs Deliverables. 8 Use Cases Companies RTDs. Contents. Keywords
 Welcome to the first issue of the OActive Newsletter! NEWSLETTER Issue 1: December 2018 Contents 1. OActive at a glance 2. OActive website, video & milestones 3. OActive Physical Meetings 4. Attendance
Welcome to the first issue of the OActive Newsletter! NEWSLETTER Issue 1: December 2018 Contents 1. OActive at a glance 2. OActive website, video & milestones 3. OActive Physical Meetings 4. Attendance
COMMISSION DELEGATED REGULATION (EU).../... of XXX
 EUROPEAN COMMISSION Brussels, XXX SANTE/10355/2015 (POOL/E4/2015/10355/10355-EN. doc) [...1(2015) XXX draft COMMISSION DELEGATED REGULATION (EU).../... of XXX supplementmg Regulation (EU) No 609/2013 of
EUROPEAN COMMISSION Brussels, XXX SANTE/10355/2015 (POOL/E4/2015/10355/10355-EN. doc) [...1(2015) XXX draft COMMISSION DELEGATED REGULATION (EU).../... of XXX supplementmg Regulation (EU) No 609/2013 of
National Clinical Programme for Rare Diseases Prof. Eileen Treacy April 7 th,
 National Clinical Programme for Rare Diseases Prof. Eileen Treacy April 7 th, 2017 1 European Reference Networks http://ec.europa.eu/health/ern/toolkit_en A European Framework for Rare Disease Services:
National Clinical Programme for Rare Diseases Prof. Eileen Treacy April 7 th, 2017 1 European Reference Networks http://ec.europa.eu/health/ern/toolkit_en A European Framework for Rare Disease Services:
L 256/32 Official Journal of the European Union
 L 256/32 Official Journal of the European Union 1.10.2005 COMMISSION DIRECTIVE 2005/61/EC of 30 September 2005 implementing Directive 2002/98/EC of the European Parliament and of the Council as regards
L 256/32 Official Journal of the European Union 1.10.2005 COMMISSION DIRECTIVE 2005/61/EC of 30 September 2005 implementing Directive 2002/98/EC of the European Parliament and of the Council as regards
User Instruction Guide
 User Instruction Guide Table of Contents Logging In and Logging Out of MMSx 1 Creating a TPN (Terminal Profile Number) 2 Single Merchant 2 From Navigation Bar 2 From Home Page Link 4 Multiple Merchants
User Instruction Guide Table of Contents Logging In and Logging Out of MMSx 1 Creating a TPN (Terminal Profile Number) 2 Single Merchant 2 From Navigation Bar 2 From Home Page Link 4 Multiple Merchants
3rd Meeting of the European Research Initiative on CLL (ERIC) during the 10th Meeting of the International Working Group on CLL (IWCLL)
 3rd Meeting of the European Research Initiative on CLL (ERIC) during the 10th Meeting of the International Working Group on CLL (IWCLL) Stresa, Italy Regina Palace Hotel Thursday, 9 October 2003, 18.00-21.00
3rd Meeting of the European Research Initiative on CLL (ERIC) during the 10th Meeting of the International Working Group on CLL (IWCLL) Stresa, Italy Regina Palace Hotel Thursday, 9 October 2003, 18.00-21.00
Common Criteria. for. CGIAR Research Proposal (CRP) Design and Assessment
 Common Criteria for CGIAR Research Proposal (CRP) Design and Assessment 30 August 2010 1 CGIAR Research Proposals (CRPs) are reviewed at various stages of their development and approval. These assessments
Common Criteria for CGIAR Research Proposal (CRP) Design and Assessment 30 August 2010 1 CGIAR Research Proposals (CRPs) are reviewed at various stages of their development and approval. These assessments
Health Task Force Workplan
 2006/SOM 3/HTF/021 Agenda Item: VI Health Task Force Workplan 2006-2007 Purpose: Information Submitted by: Chair Health Task Force Meeting Da Nang, Viet Nam 14 15 September 2006 APEC HEALTH TASK FORCE
2006/SOM 3/HTF/021 Agenda Item: VI Health Task Force Workplan 2006-2007 Purpose: Information Submitted by: Chair Health Task Force Meeting Da Nang, Viet Nam 14 15 September 2006 APEC HEALTH TASK FORCE
State of the Art of Rare Disease Activities in EU Member States and Other European Countries. Hungary Report
 State of the Art of Rare Disease Activities in EU Member States and Other European Countries Definition of a Rare Disease Hungary Report Hungary has adopted the European Commission definition of a rare
State of the Art of Rare Disease Activities in EU Member States and Other European Countries Definition of a Rare Disease Hungary Report Hungary has adopted the European Commission definition of a rare
EuroHIV: HIV/AIDS surveillance in Europe
 EuroHIV 2006 Annual Report (2004203).rev Page 1 of 14 EUROPEAN COMMISSION DG SANCO PROGRAMME OF COMMUNITY ACTION ON THE PREVENTION OF AIDS AND CERTAIN OTHER COMMUNICABLE DISEASES EuroHIV: HIV/AIDS surveillance
EuroHIV 2006 Annual Report (2004203).rev Page 1 of 14 EUROPEAN COMMISSION DG SANCO PROGRAMME OF COMMUNITY ACTION ON THE PREVENTION OF AIDS AND CERTAIN OTHER COMMUNICABLE DISEASES EuroHIV: HIV/AIDS surveillance
JPIAMR WP 5 Research Alignment Task 5.2 Virtual Research Institute Québec City, 28 March 2017
 JPIAMR WP 5 Alignment Task 5.2 Virtual Institute Québec City, 28 March 2017 Édith Brochu Institute of Infection and Immunity of CIHR Work package 5: Alignment Task 5.2: JPIAMR Virtual Institute Task Contributors
JPIAMR WP 5 Alignment Task 5.2 Virtual Institute Québec City, 28 March 2017 Édith Brochu Institute of Infection and Immunity of CIHR Work package 5: Alignment Task 5.2: JPIAMR Virtual Institute Task Contributors
Deliverable 2.1 List of relevant genetic variants for pre-emptive PGx testing
 GA N 668353 H2020 Research and Innovation Deliverable 2.1 List of relevant genetic variants for pre-emptive PGx testing WP N and Title: WP2 - Towards shared European Guidelines for PGx Lead beneficiary:
GA N 668353 H2020 Research and Innovation Deliverable 2.1 List of relevant genetic variants for pre-emptive PGx testing WP N and Title: WP2 - Towards shared European Guidelines for PGx Lead beneficiary:
