July 2016 Corporate Presentation. DAVID P. SOUTHWELL, President and CEO Cantor Fitzgerald 2 nd Annual Health Care Conference
|
|
|
- Robert Walton
- 5 years ago
- Views:
Transcription
1 July 2016 Corporate Presentation DAVID P. SOUTHWELL, President and CEO Cantor Fitzgerald 2 nd Annual Health Care Conference
2 Forward Looking Statements This presentation contains forward-looking statements that are based on our management s belief and assumptions and on information currently available to our management. Although we believe that the expectations reflected in these forward-looking statements are reasonable, these statements relate to future events or our future financial performance, and involve known and unknown risks, uncertainties and other factors that may cause our actual results, levels of activity, performance or achievements to be materially different from any future results, levels of activity, performance or achievements expressed or implied by these forward-looking statements. In some cases, you can identify forward-looking statements by terminology such as may, might, could, would, will, should, expect, intend, plan, anticipate, believe, estimate, predict, project, target, potential, continue or the negative of these terms or other comparable terminology. These statements are only predictions. You should not place undue reliance on forward-looking statements because they involve known and unknown risks, uncertainties and other factors, which are, in some cases, beyond our control and which could materially affect results. If one or more of these risks or uncertainties occur, or if our underlying assumptions prove to be incorrect, actual events or results may vary significantly from those implied or projected by the forward-looking statements. No forwardlooking statement is a guarantee of future performance. The forward-looking statements in this presentation represent our views as of the date of this presentation. We anticipate that subsequent events and developments will cause our views to change. However, while we may elect to update these forward-looking statements at some point in the future, we have no current intention of doing so except to the extent required by applicable law. You should therefore not rely on these forward-looking statements as representing our views as of any date subsequent to the date of this presentation. All trademarks and registered trademarks are the property of their respective owners. Trabodenoson is an investigational compound and is not yet approved by the FDA for any indication. 2
3 Inotek: Transforming Glaucoma Treatment Trabodenoson: Novel adenosine mimetic that stimulates a natural pathway Glaucoma: *$5.6 billion worldwide market with low compliance Phase 1 Monotherapy showed no dose related side effects (ocular or systemic) at greater than Phase 3 doses Phase 2 Monotherapy showed clear dose response, good ocular and systemic safety, ability to dose QD or BID, additive efficacy to prostaglandins First Phase 3 Monotherapy, MATrX-1, tests QD and BID doses against placebo. Results 4Q 16 First fixed-dose combination trial to initiate in 2016 Adenosine A 1 activation is neuroprotective in the retina and brain neuroprotective in the back of the eye *Source: IMS Health in Schwartz, Quigley, Survey of Ophthalmology, 2008, in press 2 Gomes et al. 2011, 3 Zhong et al. 2013, 4 Cunha
4 Leadership with History Together David P. Southwell, MBA President & CEO Rudolf Baumgartner, M.D. Chief Medical Officer William McVicar, Ph.D. Chief Scientific Officer Claudine Prowse, Ph.D. VP Strategy Cadmus Rich, M.D., MBA VP Clinical and Medical Affairs 4
5 $2 Billion U.S. Glaucoma Market Unmet Need: Effective QD treatment with minimal side effects GLAUCOMA PRESCRIPTION MARKET SHARE First Line: Prostaglandin Analogs Monotherapy QD Dosing IOP Drop from Baseline: 6 8 mmhg* Ocular Side Effects Red eye (hyperemia) Iris discoloration Blurred vision Darkening of the eyelids Adipose tissue shrinkage Eye pain Latanoprost 31% 51% Travatan Travoprost 10% Lumigan Bimatoprost 10% beta blockers (combo) 14% 49% beta blockers (alone) 14% alpha agonist 10% carbonic anhydrase inhibitor Other 6% 5% Second Line: Adjunctive Agents Monotherapy BID or TID Dosing Ocular and Systemic Side Effects Severe respiratory and cardiac reactions Blurred vision Allergic conjunctivitis Itching (pruritus), burning, stinging * Per Latanoprost Package Insert Source: Share data represents total prescriptions for IMS Health in
6 Probability of Being Treated IOP Elevation Drives Treatment Objective: Normalized Intraocular Pressure (IOP) High Usually Treated >25 mmhg Medium Low Normal IOP mmhg Usually not treated without serious risk factors Suspected Glaucoma >22-25 mmhg Medical treatment based on associated risk factors First Line Monotherapy First Line Second Line Co-Administration or Fixed-Dose Combination Increasing IOP Market Opportunity as First or Second Line First Line: IOP Lowering; Safety/Tolerability = Compliance Second Line: Additive efficacy to latanoprost; Minimal added side effects, QD dosing 6
7 Trabecular Meshwork: The Natural Mechanism for Regulating IOP Natural pressure regulating outflow 70% via trabecular meshwork Trabecular Meshwork 30% Secondary outflow via uveoscleral pathway Aqueous humor production 7
8 Trabodenoson s Novel Mechanism* Mechanism: Binds to A1 receptors on Trabecular Meshwork Upregulates MMP-2, digesting extracellular matrix proteins that clog the TM Research supporting trabodenoson s MOA presented at the 2016 American Glaucoma Society *Increased secretion of MMPs contributes to trabodenoson-induced changes in conventional outflow facility; DS Albers, CE Crosson, JS Myers, CC Rich, R Baumgartner, and WK McVicar; American Glaucoma Society Annual Meeting, March 2016, Poster #: PO047 8
9 From Adenosine to Trabodenoson Trabodenoson Rational Design Trabodenoson is an adenosine mimetic optimized to selectively target the A 1 receptor Developed by medicinal chemists at Inotek 9
10 From Adenosine to Trabodenoson Trabodenoson Rational Design Trabodenoson is an adenosine mimetic optimized to selectively target the A 1 receptor Potency High affinity for A 1 receptor Corneal Penetration Lipid solubility = ocular penetration Selectivity Non-target receptor interactions systematically removed Trabodenoson bound to A 1 receptor Eye Tissue Compatibility High compatibility with sensitive tissues in front of eye Compound A 1 (Ki, nm) A 2a (Ki, nm) A 3 (Ki, nm) Trabodenoson ,
11 Trabodenoson mcg Phase 1: Good Safety Profile and Tolerable Design Subjects Cohort Cohort Cohort Cohort Cohort Cohort Cohort Results No Dose Limiting Toxicity; no doserelated ocular or systemic side effects; limited systemic exposure at high doses , ,600 2,400 3,200 Treatment 14-day BID 14-day BID 14-day BID 14-day BID 14-day BID 14-day BID Escalating SD BID: Twice Daily SD: Single Dose Journal of Ocular Pharmacology and Therapeutics. April 2016, ahead of print. doi: /jop Trial authors; Alan Laties 1, Cadmus C. Rich 2, Randall Stoltz 3, Vernon Humbert 4, Chaim Brickman 2, William McVicar 2, and Rudolf A. Baumgartner 2 Journal of Ocular Pharmacology and Therapeutics. April 2016, ahead of print. doi: /jop Trial authors; Alan Laties 1, Cadmus C. Rich 2, Randall Stoltz 3, Vernon Humbert 4, Chaim Brickman 2, William McVicar 2, and Rudolf A. Baumgartner 2 11
12 No Clinically Significant Safety Signals in Trials to Date Phase 1 Comprehensive Safety Assessments included: Continuous cardiac monitor 12-lead ECG Orthostatic BP and heart rate Vital signs Blood cardiac troponin I Spirometry FEV 1 Blood pharmacokinetic sampling Urine pharmacokinetic sampling Renal biomarkers Karolinska sleepiness scale Adverse events Physical examinations Clinical laboratory assessments Toxicology screen Ophthalmology assessments Slit lamp exam/hyperemia Fundus exam Best-corrected visual acuity Intraocular pressure 12
13 IOP Drop SEM (mmhg) Monotherapy Phase 2 Results Dose-Dependent IOP Reduction, Increases with Treatment Duration Day 14 Day mcg 100 mcg 200 mcg 500 mcg 500 mcg * * * ** p=0.016 IOP Drop from Diurnal Baseline on Day -1 IOP Drop from Study Baseline on Day 1 * * *Indicates comparison to placebo group; p-value <0.05 ** p-value refers to Signed Rank test Day 28 versus Day 14 Journal of Ocular Pharmacology and Therapeutics. March 2016, ahead of print. doi: /jop Trial authors; Jonathan S. Myers 1, Kenneth N. Sall 2, Harvey DuBiner 3, Natanya Slomowitz 4, William McVicar 4, Cadmus C. Rich 4 and Rudolf A. Baumgartner 4 13
14 Phase 2: IOP Statistically Lowered at All Timepoints on Day 28 FIG. 2. Mean IOP for the trabodenoson 500 mcg and placebo groups before randomization (Day 1) and after randomization (Days 14, 28, and 29). Published in Journal of Ocular Pharmacology and Therapeutics. Ahead of Print DOI: /jop
15 MATrX-1 Phase 3 Trial Design Trabodenoson 1000 mcg (3.0%) QD OU Identical population to Phase 2 IOP >24 mmhg Trabodenoson 2000 mcg (6.0%) QD OU ~ 360 patients treated for 12 weeks Three trabodenoson doses: 1000 mcg QD 2000 mcg QD 1500 mcg BID Placebo controlled Statistical comparator Timolol 0.5% BID Internal control Not part of statistical comparison Screening Period 1 to 14 Days Washout Period 1 to 39 Days Placebo BID Run-in Period 5 to 9 Days Trabodenoson 1500 mcg (4.5%) BID OU Timolol BID OU Placebo BID OU Day 28* Day 42* Day 84* 3 Month Treatment Period Day 1 to Day 84 AM No Ocular Treatment Observation Period 7 Days ClinicalTrials.gov Identifier: NCT *Endpoint. 15
16 Additional Phase 3 Trials MATrX-2 Similar design as MATrX-1 Optimized dose to be selected from MATrX-1 Large sample size Long-Term Safety Trial Primary endpoint = safety Greater IOP range At least 300 patients treated for at least 6 months, and 100 patients for at least 12 months Phase 3 Program Optimized for Success: Similar patient population to Phase 2 Primary endpoint versus placebo Higher doses than in Phase 2 Below the maximum dose tested in Phase 1 16
17 Hyperemia Rate (%) IOP Drop from BL (mmhg) Path to Approval for Fixed-Dose Combination (FDC) U.S. Regulatory Requirements: Each individual active component adds to efficacy FDA determination of FDC benefit/risk profile Objectives: Phase 2 optimizes dose for Phase 3 Differentiated on efficacy and safety Latanoprost efficacy and hyperemia are dose-related Latanoprost IOP Reduction Latanoprost Hyperemia Raber, Mandema, Li, Nickens, 2015, J. Ocul. Pharmacol. Ther. 17
18 FDC Trial Progress Phase 2 trial of trabodenoson co-administered with latanoprost completed Additive efficacy to latanoprost in PGA poor-responders Phase 2 dose-ranging trial to begin in 2016 top line data in 2017 Determine optimal combination of trabodenoson and latanoprost Phase 3 program to commence following Phase 2 18
19 Back of Eye: Potential for Orphan Indications Optic Neuropathies and Degenerative Retinal Diseases Trabodenoson shows potential in treating the back of the eye: Preclinical data support effect: High Pressure Optic Neuropathy Model Eye drops shown to deliver drug to retina in rabbits and monkeys Orphan indications being evaluated: Retinitis Pigmentosa Non-Arteritic Ischemic Optic Neuropathy (NAION) Adenosine receptors in the back of the eye/retina Retinal Layers Nerve fiber layer Ganglion cell layer Inner plexiform layer Inner nuclear layer Outer plexiform layer Outer nuclear layer Photoreceptor layer Pigment epthelium Section through retina Cells Inner limiting membrane Axons at surface of retina passing via optic nerve, chiasm, and tract to lateral geniculate body Ganglion cell Müller cell (supporting glial cell) Bipolar cell Amarcine cell Horizontal cell Rod Cone Pigment cells of choroid A1 A1 Receptors 19
20 Inotek Value Drivers End-of Phase 2 Meeting Phase 3 MATrX-1 Initiation Presented at Glaucoma 360 Presented Preclinical Research at AGS Presented at GTC BIO ARVO Poster Presentations Publication of Phase 1/2 Clinical Data Initiation of FDC Dose Ranging Trial Presenting at OIS Top-Line Phase 3 MATrX-1 Results Initiation of Phase 3 MATrX-2 Initiation of Monotherapy Long- Term Safety Trial FDC Dose Ranging Results Results of Neuroprotection Clinical PoP Trial Initiation of FDC Phase 3 Trial Phase 3 MATrX-2 Results Monotherapy NDA Submission 20
21 Thank you
Treatments on the Horizon
 Latanoprostene bunod (Vesneo) Treatments on the Horizon Dominick L Opitz, OD, FAAO Associate Professor Illinois College of Optometry Valeant (B+L) Nitrous oxide-donating prostaglandin F2-alpha analogue
Latanoprostene bunod (Vesneo) Treatments on the Horizon Dominick L Opitz, OD, FAAO Associate Professor Illinois College of Optometry Valeant (B+L) Nitrous oxide-donating prostaglandin F2-alpha analogue
Building a Major Ophthalmic Pharmaceutical Company. Aerie Pharmaceuticals, Inc. Company Overview June 2-3, 2015
 Building a Major Ophthalmic Pharmaceutical Company Aerie Pharmaceuticals, Inc. Company Overview June 2-3, 2015 1 Important Information Any discussion of the potential use or expected success of our product
Building a Major Ophthalmic Pharmaceutical Company Aerie Pharmaceuticals, Inc. Company Overview June 2-3, 2015 1 Important Information Any discussion of the potential use or expected success of our product
Building a Major Ophthalmic Pharmaceutical Company. Aerie Pharmaceuticals, Inc. July 13, 2016
 Building a Major Ophthalmic Pharmaceutical Company Aerie Pharmaceuticals, Inc. July 13, 2016 1 Important Information Any discussion of the potential use or expected success of our product candidates is
Building a Major Ophthalmic Pharmaceutical Company Aerie Pharmaceuticals, Inc. July 13, 2016 1 Important Information Any discussion of the potential use or expected success of our product candidates is
Rhopressa TM Rocket 2 Phase 3 Topline Results
 Rhopressa TM Rocket 2 Phase 3 Topline Results 1 Important Information Any discussion of the potential use or expected success of our product candidates is subject to our product candidates being approved
Rhopressa TM Rocket 2 Phase 3 Topline Results 1 Important Information Any discussion of the potential use or expected success of our product candidates is subject to our product candidates being approved
Aerie Pharmaceuticals, Inc. Additional Rhopressa Rocket 1 Analyses and Path Forward
 Aerie Pharmaceuticals, Inc. Additional Rhopressa Rocket 1 Analyses and Path Forward May 7, 2015 1 Important Information Any discussion of the potential use or expected success of our product candidates
Aerie Pharmaceuticals, Inc. Additional Rhopressa Rocket 1 Analyses and Path Forward May 7, 2015 1 Important Information Any discussion of the potential use or expected success of our product candidates
VI.2.2 Summary of treatment benefits
 EU-Risk Management Plan for Bimatoprost V01 aetiology), both OAG and ACG can be secondary conditions. Secondary glaucoma refers to any case in which another disorder (e.g. injury, inflammation, vascular
EU-Risk Management Plan for Bimatoprost V01 aetiology), both OAG and ACG can be secondary conditions. Secondary glaucoma refers to any case in which another disorder (e.g. injury, inflammation, vascular
3,4 In eyes with ocular hypertension or POAG, medical
 JOURNAL OF OCULAR PHARMACOLOGY AND THERAPEUTICS Volume 32, Number 8, 2016 Mary Ann Liebert, Inc. DOI: 10.1089/jop.2015.0148 A Dose-Escalation Study to Evaluate the Safety, Tolerability, Pharmacokinetics,
JOURNAL OF OCULAR PHARMACOLOGY AND THERAPEUTICS Volume 32, Number 8, 2016 Mary Ann Liebert, Inc. DOI: 10.1089/jop.2015.0148 A Dose-Escalation Study to Evaluate the Safety, Tolerability, Pharmacokinetics,
Managing the Patient with POAG
 Managing the Patient with POAG Vision Institute Annual Fall Conference Mitchell W. Dul, OD, MS, FAAO mdul@sunyopt.edu Richard J. Madonna, MA, OD, FAAO rmadonna@sunyopt.edu Ocular Hypertension (OHT) Most
Managing the Patient with POAG Vision Institute Annual Fall Conference Mitchell W. Dul, OD, MS, FAAO mdul@sunyopt.edu Richard J. Madonna, MA, OD, FAAO rmadonna@sunyopt.edu Ocular Hypertension (OHT) Most
Company Overview. Investor Presentation November 2018
 Company Overview Investor Presentation November 2018 Important Information The information in this presentation does not contain all of the information that a potential investor should review before investing
Company Overview Investor Presentation November 2018 Important Information The information in this presentation does not contain all of the information that a potential investor should review before investing
Company Overview. Investor Presentation March 2018
 Company Overview Investor Presentation March 2018 Important Information The information in this presentation does not contain all of the information that a potential investor should review before investing
Company Overview Investor Presentation March 2018 Important Information The information in this presentation does not contain all of the information that a potential investor should review before investing
Transforming Glaucoma Care with OTX-TP Sustained Release Travaprost Plug
 Transforming Glaucoma Care with OTX-TP Sustained Release Travaprost Plug Jonathan H. Talamo, M.D. Chief Medical Officer Glaucoma 360 January 29, 2016 Page 1 Forward-Looking Statements Any statements in
Transforming Glaucoma Care with OTX-TP Sustained Release Travaprost Plug Jonathan H. Talamo, M.D. Chief Medical Officer Glaucoma 360 January 29, 2016 Page 1 Forward-Looking Statements Any statements in
Roclatan TM Mercury 1 Phase 3 12-month Topline Results. For Investor Use
 Roclatan TM Mercury 1 Phase 3 12-month Topline Results For Investor Use 1 Important Information Any discussion of the potential use or expected success of our product candidates is subject to our product
Roclatan TM Mercury 1 Phase 3 12-month Topline Results For Investor Use 1 Important Information Any discussion of the potential use or expected success of our product candidates is subject to our product
Update on Rhopressa TM QD (netarsudil ophthalmic solution) 0.02% and Roclatan TM (netarsudil/latanoprost ophthalmic solution) 0.02%/0.
 Update on Rhopressa TM QD (netarsudil ophthalmic solution) 0.02% and Roclatan TM (netarsudil/latanoprost ophthalmic solution) 0.02%/0.005% 1 Important Information Any discussion of the potential use or
Update on Rhopressa TM QD (netarsudil ophthalmic solution) 0.02% and Roclatan TM (netarsudil/latanoprost ophthalmic solution) 0.02%/0.005% 1 Important Information Any discussion of the potential use or
3/31/2019. Role of IOP. Role of IOP. Role of IOP. Role of IOP. Evaluation of glaucoma has changed Why hasn t treatment?
 Glaucoma Update ROLE FOR RHOPRESSA Gregory D. Searcy, M.D. 1857: German ophthalmologist Albrecht Van Graefe concluded all glaucomatous optic disc excavation is associated with high IOP based on the only
Glaucoma Update ROLE FOR RHOPRESSA Gregory D. Searcy, M.D. 1857: German ophthalmologist Albrecht Van Graefe concluded all glaucomatous optic disc excavation is associated with high IOP based on the only
Phase 3c Topline Results. Page 1
 Phase 3c Topline Results Page 1 Important Information Any statements in this presentation about future expectations, plans and prospects for the Company including the development and regulatory status
Phase 3c Topline Results Page 1 Important Information Any statements in this presentation about future expectations, plans and prospects for the Company including the development and regulatory status
PRACTICAL APPROACH TO MEDICAL MANAGEMENT OF GLAUCOMA
 PRACTICAL APPROACH TO MEDICAL MANAGEMENT OF GLAUCOMA DR. RAVI THOMAS, DR. RAJUL PARIKH, DR. SHEFALI PARIKH IJO MAY 2008 PRESENTER AT JDOS : DR. RAHUL SHUKLA T.N. SHUKLA EYE HOSPITAL TERMINOLOGY POAG: PRIMARY
PRACTICAL APPROACH TO MEDICAL MANAGEMENT OF GLAUCOMA DR. RAVI THOMAS, DR. RAJUL PARIKH, DR. SHEFALI PARIKH IJO MAY 2008 PRESENTER AT JDOS : DR. RAHUL SHUKLA T.N. SHUKLA EYE HOSPITAL TERMINOLOGY POAG: PRIMARY
Innovation In Ophthalmology
 Innovation In Ophthalmology INVELTYS TM Approval August 2018 Disclaimers and Notices This presentation contains forward-looking statements within the meaning of the Private Securities Litigation Reform
Innovation In Ophthalmology INVELTYS TM Approval August 2018 Disclaimers and Notices This presentation contains forward-looking statements within the meaning of the Private Securities Litigation Reform
East and North Hertfordshire treatment pathway for primary open angle glaucoma and ocular hypertension in adults
 East and North Hertfordshire treatment pathway for primary open angle glaucoma and ocular hypertension in adults Introduction Glaucoma is a group of eye diseases causing optic nerve damage. In most cases
East and North Hertfordshire treatment pathway for primary open angle glaucoma and ocular hypertension in adults Introduction Glaucoma is a group of eye diseases causing optic nerve damage. In most cases
53 year old woman attends your practice for routine exam. She has no past medical history or family history of note.
 Case 1 Normal Tension Glaucoma 53 year old woman attends your practice for routine exam. She has no past medical history or family history of note. Table 1. Right Eye Left Eye Visual acuity 6/6 6/6 Ishihara
Case 1 Normal Tension Glaucoma 53 year old woman attends your practice for routine exam. She has no past medical history or family history of note. Table 1. Right Eye Left Eye Visual acuity 6/6 6/6 Ishihara
OIS Presentation October 25, 2018
 OIS Presentation October 25, 2018 Forward looking statements This presentation has been prepared by Aerpio Pharmaceuticals ( we, us or, the Company ) and includes forward-looking statements. All statements
OIS Presentation October 25, 2018 Forward looking statements This presentation has been prepared by Aerpio Pharmaceuticals ( we, us or, the Company ) and includes forward-looking statements. All statements
Glaucoma Disease Progression Role of Intra Ocular Pressure. Is Good Enough, Low Enough?
 Glaucoma Disease Progression Role of Intra Ocular Pressure Is Good Enough, Low Enough? Glaucoma Diseases Progression Key Considerations Good number of patients may be diagnosed only after some damage the
Glaucoma Disease Progression Role of Intra Ocular Pressure Is Good Enough, Low Enough? Glaucoma Diseases Progression Key Considerations Good number of patients may be diagnosed only after some damage the
Science & Technologies. UNWANTED SIDE EFFECTS OF TRAVOPROST Gazepov Strahil 1, Iljaz Ismaili 2, Goshevska Dashtevska Emilija 2
 UNWANTED SIDE EFFECTS OF TRAVOPROST Gazepov Strahil 1, Iljaz Ismaili 2, Goshevska Dashtevska Emilija 2 1 Clinical Hospital, Shtip 2 University Eye Clinic,Skopje Introduction: Glaucoma is a chronical progressive
UNWANTED SIDE EFFECTS OF TRAVOPROST Gazepov Strahil 1, Iljaz Ismaili 2, Goshevska Dashtevska Emilija 2 1 Clinical Hospital, Shtip 2 University Eye Clinic,Skopje Introduction: Glaucoma is a chronical progressive
IOP measurements: 8.00 am (trough drug levels) and am (peak drug levels)(2 hours post dose)
 This overview of 2% eye drops was presented by Dr. Ravin N. Das, at Hotel Satya Ashoka, on 19-6-2004 in place of Dr. H. S. Ray due to unforseen circumstances. This dinner meeting was sponsored by Cipla
This overview of 2% eye drops was presented by Dr. Ravin N. Das, at Hotel Satya Ashoka, on 19-6-2004 in place of Dr. H. S. Ray due to unforseen circumstances. This dinner meeting was sponsored by Cipla
Efficacy of latanoprost in management of chronic angle closure glaucoma. Kumar S 1, Malik A 2 Singh M 3, Sood S 4. Abstract
 Original article Efficacy of latanoprost in management of chronic angle closure glaucoma Kumar S 1, Malik A 2 Singh M 3, Sood S 4 1 Associate Professor, 2 Assistant Professor, 4 Professor, Department of
Original article Efficacy of latanoprost in management of chronic angle closure glaucoma Kumar S 1, Malik A 2 Singh M 3, Sood S 4 1 Associate Professor, 2 Assistant Professor, 4 Professor, Department of
AR CS205 Clinical Pilot Study Topline Results
 AR-13324-CS205 Clinical Pilot Study Topline Results 1 Important Information The information in this presentation is current only as of its date and may have changed or may change in the future. We undertake
AR-13324-CS205 Clinical Pilot Study Topline Results 1 Important Information The information in this presentation is current only as of its date and may have changed or may change in the future. We undertake
Vision Health: Conditions, Disorders & Treatments GLAUCOMA
 Vision Health: Conditions, Disorders & Treatments GLAUCOMA Glaucoma is a disease of the optic nerve, which transmits the images you see from the eye to the brain. The optic nerve is made up of many nerve
Vision Health: Conditions, Disorders & Treatments GLAUCOMA Glaucoma is a disease of the optic nerve, which transmits the images you see from the eye to the brain. The optic nerve is made up of many nerve
Roclatan TM Mercury 2 Phase 3 Topline Results
 Roclatan TM Mercury 2 Phase 3 Topline Results 1 Important Information Any discussion of the potential use or expected success of our product candidates is subject to our product candidates being approved
Roclatan TM Mercury 2 Phase 3 Topline Results 1 Important Information Any discussion of the potential use or expected success of our product candidates is subject to our product candidates being approved
A. Incorrect! Acetazolamide is a carbonic anhydrase inhibitor given orally or by intravenous injection.
 Pharmacology - Problem Drill 20: Drugs that Treat Glaucoma Question No. 1 of 10 1. is a topical carbonic anhydrase inhibitor. Question #01 (A) Acetazolamide (B) Clonidine (C) Dorzolamide (D) Apraclonidine
Pharmacology - Problem Drill 20: Drugs that Treat Glaucoma Question No. 1 of 10 1. is a topical carbonic anhydrase inhibitor. Question #01 (A) Acetazolamide (B) Clonidine (C) Dorzolamide (D) Apraclonidine
DERBYSHIRE JOINT AREA PRESCRIBING COMMITTEE (JAPC)
 Guidelines for the medical treatment of chronic open angle glaucoma and ocular hypertension Summary: DERBYSHIRE JOINT AREA PRESCRIBING COMMITTEE (JAPC) Diagnosis and management of ocular hypertension (OHT)
Guidelines for the medical treatment of chronic open angle glaucoma and ocular hypertension Summary: DERBYSHIRE JOINT AREA PRESCRIBING COMMITTEE (JAPC) Diagnosis and management of ocular hypertension (OHT)
Vision I. Steven McLoon Department of Neuroscience University of Minnesota
 Vision I Steven McLoon Department of Neuroscience University of Minnesota 1 Eye Cornea Sclera Conjunctiva 2 Eye The conjunctiva lines the inner surface of the eyelids and outer surface of the sclera. 3
Vision I Steven McLoon Department of Neuroscience University of Minnesota 1 Eye Cornea Sclera Conjunctiva 2 Eye The conjunctiva lines the inner surface of the eyelids and outer surface of the sclera. 3
Practical approach to medical management of glaucoma DR. RATHINI LILIAN DAVID
 Practical approach to medical management of glaucoma DR. RATHINI LILIAN DAVID Glaucoma is one of the major causes of visual loss worldwide. The philosophy of glaucoma management is to preserve the visual
Practical approach to medical management of glaucoma DR. RATHINI LILIAN DAVID Glaucoma is one of the major causes of visual loss worldwide. The philosophy of glaucoma management is to preserve the visual
Divakar Gupta Glaucoma Fellow, Duke Eye Center 5/14/16
 Divakar Gupta Glaucoma Fellow, Duke Eye Center 5/14/16 Pathophysiology of glaucoma Consider risk factors of glaucoma Understand the side effects of glaucoma medications Diagnostic testing Leading cause
Divakar Gupta Glaucoma Fellow, Duke Eye Center 5/14/16 Pathophysiology of glaucoma Consider risk factors of glaucoma Understand the side effects of glaucoma medications Diagnostic testing Leading cause
Effect of brimonidine on intraocular pressure in normal tension glaucoma: A short term clinical trial
 European Journal of Ophthalmology / Vol. 13 no. 7, 2003 / pp. 611-615 Effect of brimonidine on intraocular pressure in normal tension glaucoma: A short term clinical trial S.A. GANDOLFI, L. CIMINO, P.
European Journal of Ophthalmology / Vol. 13 no. 7, 2003 / pp. 611-615 Effect of brimonidine on intraocular pressure in normal tension glaucoma: A short term clinical trial S.A. GANDOLFI, L. CIMINO, P.
OCULAR PHARMACOLOGY GLAUCOMA. increased intraocular pressure. normally mm Hg. when to Tx no fixed level.
 OCULAR PHARMACOLOGY GLAUCOMA increased intraocular pressure normally 12 20 mm Hg. when to Tx no fixed level. literature sets ~21 mm Hg as upper limit of normal. some safe at 30 mm Hg some may have damage
OCULAR PHARMACOLOGY GLAUCOMA increased intraocular pressure normally 12 20 mm Hg. when to Tx no fixed level. literature sets ~21 mm Hg as upper limit of normal. some safe at 30 mm Hg some may have damage
Abbreviated Update: Ophthalmic Glaucoma Agents
 Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35, Salem, Oregon 97301-1079 Phone 503-945-5220 Fax 503-947-1119 Abbreviated Update: Ophthalmic Glaucoma Agents Month/Year
Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35, Salem, Oregon 97301-1079 Phone 503-945-5220 Fax 503-947-1119 Abbreviated Update: Ophthalmic Glaucoma Agents Month/Year
Glaucoma Clinical Update. Barry Emara MD FRCS(C) Giovanni Caboto Club October 3, 2012
 Glaucoma Clinical Update Barry Emara MD FRCS(C) Giovanni Caboto Club October 3, 2012 Objectives Understand the different categories of glaucoma Recognize the symptoms and signs of open angle and angle-closure
Glaucoma Clinical Update Barry Emara MD FRCS(C) Giovanni Caboto Club October 3, 2012 Objectives Understand the different categories of glaucoma Recognize the symptoms and signs of open angle and angle-closure
Glaucoma is a heterogeneous
 Old and New Drug Classes Expanding To Include Glaucoma Treatments Troy Kish, PharmD, BCPS Glaucoma is a heterogeneous disease characterized by the development of increased intraocular pressure (IOP) that
Old and New Drug Classes Expanding To Include Glaucoma Treatments Troy Kish, PharmD, BCPS Glaucoma is a heterogeneous disease characterized by the development of increased intraocular pressure (IOP) that
Medical Treatment in Pediatric Glaucoma
 Medical Treatment in Pediatric Glaucoma By Nader Bayoumi, MD Lecturer of Ophthalmology Ophthalmology Department Alexandria University Alexandria, Egypt ESG 2012 Pediatric glaucoma is a surgical disease
Medical Treatment in Pediatric Glaucoma By Nader Bayoumi, MD Lecturer of Ophthalmology Ophthalmology Department Alexandria University Alexandria, Egypt ESG 2012 Pediatric glaucoma is a surgical disease
Cavosonstat Phase 2 Trial Results. November 28 th, 2016
 Phase 2 Trial Results November 28 th, 2016 Nivalis Therapeutics 2016 Agenda Introduction Mike Carruthers, Chief Financial Officer Initial Remarks Jon Congleton, President and CEO Data Highlights David
Phase 2 Trial Results November 28 th, 2016 Nivalis Therapeutics 2016 Agenda Introduction Mike Carruthers, Chief Financial Officer Initial Remarks Jon Congleton, President and CEO Data Highlights David
Goals. Glaucoma PARA PEARL TO DO. Vision Loss with Glaucoma
 Glaucoma Janet R. Fett, OD Drs. Kincaid, Fett and Tharp So Sioux City, NE eyewear21@hotmail.com Goals Understand Glaucoma Disease process Understand how your data (objective and subjective) assists in
Glaucoma Janet R. Fett, OD Drs. Kincaid, Fett and Tharp So Sioux City, NE eyewear21@hotmail.com Goals Understand Glaucoma Disease process Understand how your data (objective and subjective) assists in
Elements for a Public Summary. Overview of disease epidemiology
 VI.2 VI.2.1 Elements for a Public Summary Overview of disease epidemiology Epidemiology of the disease Incidence and prevalence Increased pressure in the eye occurs in more than 100 million people, and
VI.2 VI.2.1 Elements for a Public Summary Overview of disease epidemiology Epidemiology of the disease Incidence and prevalence Increased pressure in the eye occurs in more than 100 million people, and
Elements for a public summary. Overview of disease epidemiology
 VI.2 VI.2.1 Elements for a public summary Overview of disease epidemiology Glaucoma is an eye disease that can result in damage to the optic nerve and loss of vision (blindness). It is the major cause
VI.2 VI.2.1 Elements for a public summary Overview of disease epidemiology Glaucoma is an eye disease that can result in damage to the optic nerve and loss of vision (blindness). It is the major cause
Summary of the risk management plan (RMP) for Izba (travoprost)
 EMA/14138/2014 Summary of the risk management plan (RMP) for Izba (travoprost) This is a summary of the risk management plan (RMP) for Izba, which details the measures to be taken in order to ensure that
EMA/14138/2014 Summary of the risk management plan (RMP) for Izba (travoprost) This is a summary of the risk management plan (RMP) for Izba, which details the measures to be taken in order to ensure that
PATIENT GUIDE TO LUMIGAN 0.01%
 Treatment Tracker enclosed PATIENT GUIDE TO LUMIGAN 0.01% LUMIGAN (bimatoprost ophthalmic solution) 0.01% is used for the treatment of high eye pressure, also called intraocular pressure (IOP), in people
Treatment Tracker enclosed PATIENT GUIDE TO LUMIGAN 0.01% LUMIGAN (bimatoprost ophthalmic solution) 0.01% is used for the treatment of high eye pressure, also called intraocular pressure (IOP), in people
Vision loss in elderly. Erica Weir, April 2015
 Vision loss in elderly Erica Weir, April 2015 1 Burden Enter nursing homes 3 years earlier Twice the risk of falling 4x the risk of hip fracture Independent risk factor for delirium What are the leading
Vision loss in elderly Erica Weir, April 2015 1 Burden Enter nursing homes 3 years earlier Twice the risk of falling 4x the risk of hip fracture Independent risk factor for delirium What are the leading
Vision loss in elderly
 Vision loss in elderly Erica Weir, February 2016 1 Burden Enter nursing homes 3 years earlier Twice the risk of falling 4x the risk of hip fracture Independent risk factor for delirium What are the leading
Vision loss in elderly Erica Weir, February 2016 1 Burden Enter nursing homes 3 years earlier Twice the risk of falling 4x the risk of hip fracture Independent risk factor for delirium What are the leading
LOWPROST Eye Drops (Bimatoprost Ophthalmic Solution 0.01%)
 Published on: 14 Apr 2017 LOWPROST Eye Drops (Bimatoprost Ophthalmic Solution 0.01%) For the use of a Registered Medical Practitioner or a Hospital or a Laboratory only Composition Each ml contains: Bimatoprost...
Published on: 14 Apr 2017 LOWPROST Eye Drops (Bimatoprost Ophthalmic Solution 0.01%) For the use of a Registered Medical Practitioner or a Hospital or a Laboratory only Composition Each ml contains: Bimatoprost...
Xalatan and Xalatan /betablocker fixed combination : role in glaucoma therapy
 Xalatan and Xalatan /betablocker fixed combination : role in glaucoma therapy Grant McLaren St John Eye Hospital Division of Ophthalmology University of Witwatersrand Johannesburg South Africa 1 Goals
Xalatan and Xalatan /betablocker fixed combination : role in glaucoma therapy Grant McLaren St John Eye Hospital Division of Ophthalmology University of Witwatersrand Johannesburg South Africa 1 Goals
The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION. 28 May 2008
 The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 28 May 2008 COSOPT 20 mg/5 mg/ml, eye drops, solution in single dose container Box of 60 0.2 ml single dose containers
The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 28 May 2008 COSOPT 20 mg/5 mg/ml, eye drops, solution in single dose container Box of 60 0.2 ml single dose containers
GLAUCOMA. An Overview
 GLAUCOMA An Overview Compiled by Campbell M Gold (2004) CMG Archives http://campbellmgold.com --()-- IMPORTANT The health information contained herein is not meant as a substitute for advice from your
GLAUCOMA An Overview Compiled by Campbell M Gold (2004) CMG Archives http://campbellmgold.com --()-- IMPORTANT The health information contained herein is not meant as a substitute for advice from your
Glaucoma. How is Glaucoma Diagnosed? Glaucoma Testing
 Glaucoma How is Glaucoma Diagnosed? Glaucoma Testing There is no single test for glaucoma. The diagnosis is made by evaluating the patient from a number of perspectives, using specialized instruments.
Glaucoma How is Glaucoma Diagnosed? Glaucoma Testing There is no single test for glaucoma. The diagnosis is made by evaluating the patient from a number of perspectives, using specialized instruments.
WARNING LETTER DEPARTMENT OF HEALTH & HUMAN SERVICES TRANSMITTED BY FACSIMILE
 DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service Food and Drug Administration Rockville, MD 20857 TRANSMITTED BY FACSIMILE David E.I. Pyott President and Chief Executive Officer PO Box 19534
DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service Food and Drug Administration Rockville, MD 20857 TRANSMITTED BY FACSIMILE David E.I. Pyott President and Chief Executive Officer PO Box 19534
NEW ZEALAND DATA SHEET
 TRUSOPT 2% Ophthalmic Solution dorzolamide hydrochloride 1 Trusopt 2% Opthalmic Solution 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each 1mL of TRUSOPT 2% contains 20 mg dorzolamide (22.3 mg of dorzolamide
TRUSOPT 2% Ophthalmic Solution dorzolamide hydrochloride 1 Trusopt 2% Opthalmic Solution 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each 1mL of TRUSOPT 2% contains 20 mg dorzolamide (22.3 mg of dorzolamide
Glaucoma Basics OVERVIEW GENETICS SIGNALMENT/DESCRIPTION OF PET
 Glaucoma Basics OVERVIEW Glaucoma is a disease of the eye, in which the pressure within the eye is increased (pressure within the eye is known as intraocular pressure or IOP) High intraocular pressure
Glaucoma Basics OVERVIEW Glaucoma is a disease of the eye, in which the pressure within the eye is increased (pressure within the eye is known as intraocular pressure or IOP) High intraocular pressure
psivida Transforms into Commercial Stage Specialty BioPharmaceutical Company AAO October 25, 2018 NASDAQ: EYPT
 psivida Transforms into Commercial Stage Specialty BioPharmaceutical Company OIS @ AAO October 25, 2018 NASDAQ: EYPT Forward Looking SAFE HARBOR STATEMENTS UNDER THE PRIVATE SECURITIES LITIGATION REFORM
psivida Transforms into Commercial Stage Specialty BioPharmaceutical Company OIS @ AAO October 25, 2018 NASDAQ: EYPT Forward Looking SAFE HARBOR STATEMENTS UNDER THE PRIVATE SECURITIES LITIGATION REFORM
The second most common causes of blindness worldwide. ( after cataract) The commonest cause of irreversible blindness in the world Estimated that 3%
 The second most common causes of blindness worldwide. ( after cataract) The commonest cause of irreversible blindness in the world Estimated that 3% of our population age > 40 have glaucoma In the past:
The second most common causes of blindness worldwide. ( after cataract) The commonest cause of irreversible blindness in the world Estimated that 3% of our population age > 40 have glaucoma In the past:
N W KNOW. What You Need to Know. What You Know About Glaucoma Could Save Your Sight
 What You Need to Know is the second leading cause of blindness around the world, so it is important to understand its significance and treatment options. While glaucoma is not curable, there are several
What You Need to Know is the second leading cause of blindness around the world, so it is important to understand its significance and treatment options. While glaucoma is not curable, there are several
5/12/2014. Lynn E. Lawrence, CPOT, ABOC, COA
 Lynn E. Lawrence, CPOT, ABOC, COA Glaucoma is an optic neuropathy characterized by a loss of ganglion cells and their axons, in the RNFL. The loss of retinal ganglion cells in glaucoma is irreversible
Lynn E. Lawrence, CPOT, ABOC, COA Glaucoma is an optic neuropathy characterized by a loss of ganglion cells and their axons, in the RNFL. The loss of retinal ganglion cells in glaucoma is irreversible
Financial Disclosure. Prostaglandin Analogs (PGs) The Glaucoma Grab Bag: Practical Guidelines for Effective Glaucoma Therapy
 The Glaucoma Grab Bag: Practical Guidelines for Effective Glaucoma Therapy Danica J. Marrelli, OD, FAAO University of Houston College of Optometry Financial Disclosure I have received I have received speaking
The Glaucoma Grab Bag: Practical Guidelines for Effective Glaucoma Therapy Danica J. Marrelli, OD, FAAO University of Houston College of Optometry Financial Disclosure I have received I have received speaking
Class Update with New Drug Evaluation: Glaucoma Drugs
 Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
PGAs. Glaucoma Pharmacology A-Z. Selecting Therapy. PGAs Prostaglandin analogs. Prostaglandin Side Effects 1/6/2014
 PGAs Glaucoma Pharmacology A-Z Eric E. Schmidt, O.D. Omni Eye Specialists Wilmington, NC schmidtyvision@msn.com QHS dosing Long duration of action Flatten diurnal curve Effective on trough and peak IOP
PGAs Glaucoma Pharmacology A-Z Eric E. Schmidt, O.D. Omni Eye Specialists Wilmington, NC schmidtyvision@msn.com QHS dosing Long duration of action Flatten diurnal curve Effective on trough and peak IOP
Elements for a public summary. VI.2.1 Overview of disease epidemiology
 VI.2 Elements for a public summary VI.2.1 Overview of disease epidemiology Studies estimated that 3-6 million people in the United States alone, including 4-10% of the population older than 40 years, have
VI.2 Elements for a public summary VI.2.1 Overview of disease epidemiology Studies estimated that 3-6 million people in the United States alone, including 4-10% of the population older than 40 years, have
Latanoprost 0.005% v/s Timolol Maleate 0.5% Pressure Lowering Effect in Primary Open Angle Glaucoma
 Original Article Latanoprost 0.005% v/s Timolol Maleate 0.5% Pressure Lowering Effect in Primary Open Angle Glaucoma Arshad Ali Lodhi, Khalid Iqbal Talpur, Mahtab Alam Khanzada Pak J Ophthalmol 2008, Vol.
Original Article Latanoprost 0.005% v/s Timolol Maleate 0.5% Pressure Lowering Effect in Primary Open Angle Glaucoma Arshad Ali Lodhi, Khalid Iqbal Talpur, Mahtab Alam Khanzada Pak J Ophthalmol 2008, Vol.
Treatments for Open-Angle Glaucoma. A Review of the Research for Adults
 Treatments for Open-Angle Glaucoma A Review of the Research for Adults Is This Information Right for Me? Yes, this information is for you if: Your eye doctor has said that you have open-angle glaucoma,
Treatments for Open-Angle Glaucoma A Review of the Research for Adults Is This Information Right for Me? Yes, this information is for you if: Your eye doctor has said that you have open-angle glaucoma,
effect of previous argon laser trabeculoplasty
 Graefe s Arch Clin Exp Ophthalmol (2006) 244: 1073 1076 CLINICAL INVESTIGATION DOI 10.1007/s00417-005-0198-x Esther Arranz-Marquez Miguel A. Teus Effect of previous argon laser trabeculoplasty on the ocular
Graefe s Arch Clin Exp Ophthalmol (2006) 244: 1073 1076 CLINICAL INVESTIGATION DOI 10.1007/s00417-005-0198-x Esther Arranz-Marquez Miguel A. Teus Effect of previous argon laser trabeculoplasty on the ocular
Update on Glaucoma Medical Therapy
 Update on Glaucoma Medical Therapy Murray Fingeret, OD Disclosures Consultant Alcon, Allergan, Bausch & Lomb, Carl Zeiss Meditec, Dyopsys, Heidelberg Engineering, Reichert, Topcon Glaucoma Therapy An Overview
Update on Glaucoma Medical Therapy Murray Fingeret, OD Disclosures Consultant Alcon, Allergan, Bausch & Lomb, Carl Zeiss Meditec, Dyopsys, Heidelberg Engineering, Reichert, Topcon Glaucoma Therapy An Overview
Building a Major Ophthalmic Pharmaceutical Company. Aerie Pharmaceuticals, Inc. ASRS August 8, 2016
 Building a Major Ophthalmic Pharmaceutical Company Aerie Pharmaceuticals, Inc. OIS @ ASRS August 8, 2016 1 Important Information Any discussion of the potential use or expected success of our product candidates
Building a Major Ophthalmic Pharmaceutical Company Aerie Pharmaceuticals, Inc. OIS @ ASRS August 8, 2016 1 Important Information Any discussion of the potential use or expected success of our product candidates
Glaucoma. Glaucoma. Glaucoma. Trevor Arnold, MS, DVM, DACVO
 Glaucoma Trevor Arnold, MS, DVM, DACVO Glaucoma Physiology of Aqueous Humor Produced in the ciliary body Flows out the iridocorneal angle and ciliary cleft High intraocular pressures are caused by a decreased
Glaucoma Trevor Arnold, MS, DVM, DACVO Glaucoma Physiology of Aqueous Humor Produced in the ciliary body Flows out the iridocorneal angle and ciliary cleft High intraocular pressures are caused by a decreased
Sponsor. Generic Drug Name. Trial Indications. Protocol Number. Protocol Title. Clinical Trial Phase. Study Start/End Dates. Reason for Termination
 Sponsor Alcon Research, Ltd. Generic Drug Name Travoprost/timolol maleate Trial Indications Open-angle glaucoma or ocular hypertension Protocol Number C-09-007 Protocol Title An Evaluation of Patient Reported
Sponsor Alcon Research, Ltd. Generic Drug Name Travoprost/timolol maleate Trial Indications Open-angle glaucoma or ocular hypertension Protocol Number C-09-007 Protocol Title An Evaluation of Patient Reported
Original Article. Abstract. Introduction. C Olali, G Malietzis, S Ahmed, E Samaila 1, M Gupta
 Original Article Real-life experience study of the safety and efficacy of travoprost 0.004% / timoptol 0.50% fixed combination ophthalmic solution in intraocular pressure control C Olali, G Malietzis,
Original Article Real-life experience study of the safety and efficacy of travoprost 0.004% / timoptol 0.50% fixed combination ophthalmic solution in intraocular pressure control C Olali, G Malietzis,
How Strongly Do You Feel That This Patient Has Glaucoma? % % % % %
 My Favorite Cases Anthony B. Litwak, OD, FAAO VA Medical Center Baltimore, Maryland Dr. Litwak is a speaker and on advisory boards for Alcon and Zeiss Meditek CASE CR 35 yohf Neg PMH +FOH mother and grandmother
My Favorite Cases Anthony B. Litwak, OD, FAAO VA Medical Center Baltimore, Maryland Dr. Litwak is a speaker and on advisory boards for Alcon and Zeiss Meditek CASE CR 35 yohf Neg PMH +FOH mother and grandmother
RXi Pharmaceuticals. sd-rxrna Demonstrate Robust Efficacy in the Eye. Dr. Geert Cauwenbergh President & CEO OTCQX: RXII. Next Generation in RNAi
 RXi Pharmaceuticals sd-rxrna Demonstrate Robust Efficacy in the Eye Next Generation in RNAi Dr. Geert Cauwenbergh President & CEO OTCQX: RXII 2 Forward Looking Statements This presentation contains forward-looking
RXi Pharmaceuticals sd-rxrna Demonstrate Robust Efficacy in the Eye Next Generation in RNAi Dr. Geert Cauwenbergh President & CEO OTCQX: RXII 2 Forward Looking Statements This presentation contains forward-looking
My Favourite Cases Anthony B. Litwak, OD, FAAO VA Medical Center Baltimore, MD
 My Favourite Cases Anthony B. Litwak, OD, FAAO VA Medical Center Baltimore, MD Dr. Litwak is a speaker and on advisory boards for Alcon and Zeiss Meditek CASE CR! 35 YOHF! Neg PMH! +FOH mother and grandmother
My Favourite Cases Anthony B. Litwak, OD, FAAO VA Medical Center Baltimore, MD Dr. Litwak is a speaker and on advisory boards for Alcon and Zeiss Meditek CASE CR! 35 YOHF! Neg PMH! +FOH mother and grandmother
Applications of Sustained Release Delivery Systems in Ocular Disease
 Applications of Sustained Release Delivery Systems in Ocular Disease Formulation and Delivery Systems For Peptide and Protein 2012 December 5, 2012 Christopher A. Rhodes, Ph.D. Principal, Christopher A
Applications of Sustained Release Delivery Systems in Ocular Disease Formulation and Delivery Systems For Peptide and Protein 2012 December 5, 2012 Christopher A. Rhodes, Ph.D. Principal, Christopher A
ROBERT L. SHIELDS, M.D. GLAUCOMA SURGERY AND TREATMENT
 1 CURRICULUM VITAE ROBERT L. SHIELDS, M.D. GLAUCOMA SURGERY AND TREATMENT University of Colorado Health Eye Center Cherry Creek 2000 South Colorado Boulevard Annex Building, Suite 100 Denver, Colorado
1 CURRICULUM VITAE ROBERT L. SHIELDS, M.D. GLAUCOMA SURGERY AND TREATMENT University of Colorado Health Eye Center Cherry Creek 2000 South Colorado Boulevard Annex Building, Suite 100 Denver, Colorado
SUPPLEMENTARY INFORMATION
 SUPPLEMENTARY INFORMATION Contents METHODS... 2 Inclusion and exclusion criteria... 2 Supplementary table S1... 2 Assessment of abnormal ocular signs and symptoms... 3 Supplementary table S2... 3 Ocular
SUPPLEMENTARY INFORMATION Contents METHODS... 2 Inclusion and exclusion criteria... 2 Supplementary table S1... 2 Assessment of abnormal ocular signs and symptoms... 3 Supplementary table S2... 3 Ocular
Histology of the Eye
 Histology of the Eye Objectives By the end of this lecture, the student should be able to describe: The general structure of the eye. The microscopic structure of:»cornea.»retina. EYE BULB Three coats
Histology of the Eye Objectives By the end of this lecture, the student should be able to describe: The general structure of the eye. The microscopic structure of:»cornea.»retina. EYE BULB Three coats
Innovation In Ophthalmics
 Innovation In Ophthalmics Ophthalmic Innovation Summit @ AAO 2018 October 25, 2018 Mark Iwicki Chairman & CEO, Kala Pharmaceuticals Disclaimers and Notices This presentation contains forward-looking statements
Innovation In Ophthalmics Ophthalmic Innovation Summit @ AAO 2018 October 25, 2018 Mark Iwicki Chairman & CEO, Kala Pharmaceuticals Disclaimers and Notices This presentation contains forward-looking statements
Long Term Care Formulary RS 14. RESTRICTED STATUS Topical Medical Treatment of Glaucoma 1 of 5
 RESTRICTED STATUS Topical Medical Treatment of Glaucoma 1 of 5 PREAMBLE Significance: Glaucoma occurs in 1-2% of white people aged over 40 years, rising to 5% at 70 years and exponentially with advancing
RESTRICTED STATUS Topical Medical Treatment of Glaucoma 1 of 5 PREAMBLE Significance: Glaucoma occurs in 1-2% of white people aged over 40 years, rising to 5% at 70 years and exponentially with advancing
Phase 3 investigation of aprocitentan for resistant hypertension management. Investor Webcast June 2018
 Phase 3 investigation of aprocitentan for resistant hypertension management Investor Webcast June 2018 The following information contains certain forward-looking statements, relating to the company s business,
Phase 3 investigation of aprocitentan for resistant hypertension management Investor Webcast June 2018 The following information contains certain forward-looking statements, relating to the company s business,
What s New in Glaucoma Treatment! (TPG)
 What s New in Glaucoma Treatment! (TPG) Jane Kuo,OD Author: Dr. Jane Kuo graduated from the Southern California College of Optometry at Marshall B. Ketchum University (SCCO). She completed her residency
What s New in Glaucoma Treatment! (TPG) Jane Kuo,OD Author: Dr. Jane Kuo graduated from the Southern California College of Optometry at Marshall B. Ketchum University (SCCO). She completed her residency
The Visual System. Retinal Anatomy Dr. Casagrande February 2, Phone: Office: T2302 MCN
 The Visual System Retinal Anatomy Dr. Casagrande February 2, 2004 Phone: 343-4538 Email: vivien.casagrande@mcmail.vanderbilt.edu Office: T2302 MCN Reading assignments and Good Web Sites Chapter 2 in Tovée,
The Visual System Retinal Anatomy Dr. Casagrande February 2, 2004 Phone: 343-4538 Email: vivien.casagrande@mcmail.vanderbilt.edu Office: T2302 MCN Reading assignments and Good Web Sites Chapter 2 in Tovée,
Completed Clinical Research Trials Monte Dirks, M.D.
 Completed Clinical Research Trials Monte Dirks, M.D. Monte Dirks, M.D. - Primary Investigator Norfloxacin -Merck (1986) Safety and efficacy of norfloxacin vs. tobramycin in the treatment of external ocular
Completed Clinical Research Trials Monte Dirks, M.D. Monte Dirks, M.D. - Primary Investigator Norfloxacin -Merck (1986) Safety and efficacy of norfloxacin vs. tobramycin in the treatment of external ocular
Ocular Hypotensive Efficacy of Netarsudil Ophthalmic Solution 0.02% Over a 24-Hour Period: A Pilot Study
 Ocular Hypotensive Efficacy of Netarsudil Ophthalmic Solution 0.02% Over a 24-Hour Period: A Pilot Study James H. Peace, M.D. 1, Casey K. Kopczynski, Ph.D. 2, and Theresa Heah, M.D. 2 1 Inglewood, CA 2
Ocular Hypotensive Efficacy of Netarsudil Ophthalmic Solution 0.02% Over a 24-Hour Period: A Pilot Study James H. Peace, M.D. 1, Casey K. Kopczynski, Ph.D. 2, and Theresa Heah, M.D. 2 1 Inglewood, CA 2
Innovations in Glaucoma Drug Delivery What the Future Holds. Justin Schweitzer, OD, FAAO Vance Thompson Vision Sioux Falls, South Dakota
 Innovations in Glaucoma Drug Delivery What the Future Holds Justin Schweitzer, OD, FAAO Vance Thompson Vision Sioux Falls, South Dakota Allergan Glaukos Bausch and Lomb Bio- Tissue Alcon TearScience Reichert
Innovations in Glaucoma Drug Delivery What the Future Holds Justin Schweitzer, OD, FAAO Vance Thompson Vision Sioux Falls, South Dakota Allergan Glaukos Bausch and Lomb Bio- Tissue Alcon TearScience Reichert
THE CHRONIC GLAUCOMAS
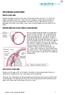 THE CHRONIC GLAUCOMAS WHAT IS GLAUCOMA People with glaucoma have lost some of their field of all round vision. It is often the edge or periphery that is lost. That is why the condition can be missed until
THE CHRONIC GLAUCOMAS WHAT IS GLAUCOMA People with glaucoma have lost some of their field of all round vision. It is often the edge or periphery that is lost. That is why the condition can be missed until
Revefenacin (TD-4208) Phase 3 Efficacy Results
 Revefenacin (TD-4208) Phase 3 Efficacy Results Once-daily, Nebulized Long-Acting Muscarinic Antagonist (LAMA) October 20, 2016 THERAVANCE, the Cross/Star logo and MEDICINES THAT MAKE A DIFFERENCE are registered
Revefenacin (TD-4208) Phase 3 Efficacy Results Once-daily, Nebulized Long-Acting Muscarinic Antagonist (LAMA) October 20, 2016 THERAVANCE, the Cross/Star logo and MEDICINES THAT MAKE A DIFFERENCE are registered
Collaboration in the care of glaucoma patients and glaucoma suspects. Barry Emara MD FRCS(C) Nico Ristorante November 29, 2012
 Collaboration in the care of glaucoma patients and glaucoma suspects Barry Emara MD FRCS(C) Nico Ristorante November 29, 2012 Goals of Collaboration Patient-centred and evidence based approach Timely access
Collaboration in the care of glaucoma patients and glaucoma suspects Barry Emara MD FRCS(C) Nico Ristorante November 29, 2012 Goals of Collaboration Patient-centred and evidence based approach Timely access
KEY MESSAGES. Details of the evidence supporting these recommendations can be found in the above CPG, available on the following websites:
 QUICK REFERENCE FOR HEALTHCARE PROVIDERS KEY MESSAGES 1. Glaucoma is a chronic eye disease that damages the optic nerve, & can result in serious vision loss and irreversible blindness. 2. Glaucoma diagnosis
QUICK REFERENCE FOR HEALTHCARE PROVIDERS KEY MESSAGES 1. Glaucoma is a chronic eye disease that damages the optic nerve, & can result in serious vision loss and irreversible blindness. 2. Glaucoma diagnosis
[TRAVOPROST] 40 MICROGRAMS/ML, EYE DROPS, SOLUTION RISK MANAGEMENT PLAN. TRAVOPR-v
![[TRAVOPROST] 40 MICROGRAMS/ML, EYE DROPS, SOLUTION RISK MANAGEMENT PLAN. TRAVOPR-v [TRAVOPROST] 40 MICROGRAMS/ML, EYE DROPS, SOLUTION RISK MANAGEMENT PLAN. TRAVOPR-v](/thumbs/94/119684929.jpg) [TRAVOPROST] 40 MICROGRAMS/ML, EYE DROPS, SOLUTION RISK MANAGEMENT PLAN TRAVOPR-v2-270214 VI.2 Elements for a public summary VI.2.1 Overview of disease epidemiology Studies estimated that 3-6 million people
[TRAVOPROST] 40 MICROGRAMS/ML, EYE DROPS, SOLUTION RISK MANAGEMENT PLAN TRAVOPR-v2-270214 VI.2 Elements for a public summary VI.2.1 Overview of disease epidemiology Studies estimated that 3-6 million people
(Eye)Drop It Like It s Hot New & Exciting Advancements in Glaucoma
 Disclosures (Eye)Drop It Like It s Hot New & Exciting Advancements in Glaucoma I have no financial ties or relationships to any of the companies or products mentioned in this lecture. Although if one wants
Disclosures (Eye)Drop It Like It s Hot New & Exciting Advancements in Glaucoma I have no financial ties or relationships to any of the companies or products mentioned in this lecture. Although if one wants
THE CHRONIC GLAUCOMAS
 THE CHRONIC GLAUCOMAS WHAT IS GLAUCOMA? People with glaucoma have lost some of their field of all round vision. It is often the edge or periphery that is lost. That is why the condition can be missed until
THE CHRONIC GLAUCOMAS WHAT IS GLAUCOMA? People with glaucoma have lost some of their field of all round vision. It is often the edge or periphery that is lost. That is why the condition can be missed until
Ganglion cell analysis by optical coherence tomography (OCT) Jonathan A. Micieli, MD Valérie Biousse, MD
 Ganglion cell analysis by optical coherence tomography (OCT) Jonathan A. Micieli, MD Valérie Biousse, MD Figure 1. Normal OCT of the macula (cross section through the line indicated on the fundus photo)
Ganglion cell analysis by optical coherence tomography (OCT) Jonathan A. Micieli, MD Valérie Biousse, MD Figure 1. Normal OCT of the macula (cross section through the line indicated on the fundus photo)
Aquinox Q2 /2015 Conference Call: LEADERSHIP Secondary Endpoint Update - AQX-1125 in BPS/IC. August 6, 2015
 Aquinox Q2 /2015 Conference Call: LEADERSHIP Secondary Endpoint Update - AQX-1125 in BPS/IC August 6, 2015 Forward Looking Statements / Safe Harbor This presentation and the accompanying oral commentary
Aquinox Q2 /2015 Conference Call: LEADERSHIP Secondary Endpoint Update - AQX-1125 in BPS/IC August 6, 2015 Forward Looking Statements / Safe Harbor This presentation and the accompanying oral commentary
Opthea Initiates OPT-302 Diabetic Macular Edema Clinical Trial
 ASX and Media Release 3 January 2018 Opthea Initiates OPT-302 Diabetic Macular Edema Clinical Trial Melbourne, Australia; January 3 2018 Opthea Limited (ASX:OPT), a late stage biopharmaceutical company
ASX and Media Release 3 January 2018 Opthea Initiates OPT-302 Diabetic Macular Edema Clinical Trial Melbourne, Australia; January 3 2018 Opthea Limited (ASX:OPT), a late stage biopharmaceutical company
Clinical results of OPT-302 (VEGF-C/D Trap ) Combination Treatment in namd and DME
 Clinical results of OPT-302 (VEGF-C/D Trap ) Combination Treatment in namd and DME Ophthalmology Innovation Summit @ AAO, October 25 2018 Megan Baldwin PhD, CEO & Managing Director Disclaimer Investment
Clinical results of OPT-302 (VEGF-C/D Trap ) Combination Treatment in namd and DME Ophthalmology Innovation Summit @ AAO, October 25 2018 Megan Baldwin PhD, CEO & Managing Director Disclaimer Investment
Preclinical Models of Ocular Glaucoma at CBI
 Preclinical Models of Ocular Glaucoma at CBI Comparative Biosciences, Inc. 786 Lucerne Drive Sunnyvale, CA 94085 Telephone: 408.738.9260 www.compbio.com Premier Preclinical Contract Research Organization
Preclinical Models of Ocular Glaucoma at CBI Comparative Biosciences, Inc. 786 Lucerne Drive Sunnyvale, CA 94085 Telephone: 408.738.9260 www.compbio.com Premier Preclinical Contract Research Organization
Revised: 07/2017. LUMIGAN (bimatoprost ophthalmic solution) 0.01% for topical ophthalmic use Initial U.S. Approval: 2001
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use LUMIGAN 0.01% safely and effectively. See full prescribing information for LUMIGAN 0.01%. LUMIGAN
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use LUMIGAN 0.01% safely and effectively. See full prescribing information for LUMIGAN 0.01%. LUMIGAN
DATA SHEET. Clinical studies have shown mean intraocular pressure decreases of up to 9 mmhg.
 DATA SHEET NAME OF MEDICINE LUMIGAN (bimatoprost) 0.3 mg/ml eye drops Presentation LUMIGAN (bimatoprost) eye drops are a clear, isotonic, colourless, sterile ophthalmic solution. Bimatoprost is a white
DATA SHEET NAME OF MEDICINE LUMIGAN (bimatoprost) 0.3 mg/ml eye drops Presentation LUMIGAN (bimatoprost) eye drops are a clear, isotonic, colourless, sterile ophthalmic solution. Bimatoprost is a white
Clinical results of OPT-302 (VEGF-C/D Trap ) Combination Treatment in namd and DME
 Clinical results of OPT-302 (VEGF-C/D Trap ) Combination Treatment in namd and DME Ophthalmology Innovation Summit @ AAO, October 25 2018 Megan Baldwin PhD, CEO & Managing Director Disclaimer Investment
Clinical results of OPT-302 (VEGF-C/D Trap ) Combination Treatment in namd and DME Ophthalmology Innovation Summit @ AAO, October 25 2018 Megan Baldwin PhD, CEO & Managing Director Disclaimer Investment
