IT HAS BECOME apparent, in clinical
|
|
|
- Godwin Hilary Robertson
- 5 years ago
- Views:
Transcription
1 Electrocardiographic Alterations Produced by a Decrease in Plasma ph, Bicarbonate and Sodium as Compared with those Produced by an Increase in Potassium By ATHLEEN E. ROBERTS, M.D., AND MELVILLE G. MAGIDA, M.D. WITH THE TECHNICAL ASSISTANCE OF MRS. HlLDA SATRAN AND MllS. SARAH WARD The effects of a decrease in plasma ph, bicarbonate and sodium on the electrocardiographic pattern are compared with those produced by elevation of scrum potassium and lithium in the dog. The electrocardiographic alterations were found to be similar in form and progression. The alterations can be correlated with those found in isolated nerve and muscle preparations. IT HAS BECOME apparent, in clinical situations accompanied by hyperkalemia, that the electrocardiographic abnormalities and clinical symptomatology cannot be closely correlated with the plasma potassium concentration. ' 3 In many of these conditions, there occurs a coincident decrease in sodium, bicarbonate, and ph accompanied by variable disturbances in chloride, phosphate, and magnesium. " 8 The effects of potassium, calcium, and magnesium on the electrocardiogram have been well defined experimentally. However, the effects of bicarbonate, ph, and sodium, which are known to influence the electric potentials in isolated nerve and muscle have not been delineated in the intact organism. The present experiments in normal dogs were undertaken () to define the electrocardiographic changes which occur as a result of disturbances of those ions known to be altered in clinical situations associated with hyperkalemia, and (2) to correlate these From the Department of Physiology, Cornell University Medical College, New York, N. Y. Aided by grants from the American Heart Association and the tional Heart Institute, tional Institutes of Health. This work was done during Dr. Roberts' tenure of a Research Fellowship of the American Heart Association. Dr. Magida is a Post-Doctorate Research Fellow, tional Heart Institute, United States Public Health Service, Bethesda, Md. Received for publication Dec. 5, 952. changes with those previously ascribed solely to potassium. METHODS AND MATERIALS Forty-two experiments were performed on 3 female mongrel dogs, anesthetized with sodium pentobarbital. Following a preliminary control period, the substance to be investigated was infused through polyethylene tubing into an antecubital vein, utilizing a calibrated constant infusion pump. Serial electrocardiograms and blood samples for the determination of carbon dioxide, ph, sodium, potassium and chloride were taken at intervals of 0 to 20 minutes. Respiratory acidosis was produced by the administration of carbon dioxide-oxygen mixtures delivered by a Palmer pump through a closed tracheotomy site at rates of 7.5 to 0 liters per minute. The carbon dioxide-oxygen mixtures were incremented in stepwise fashion beginning with 20 per cent and terminating with SO per cent carbon dioxide. Electrocardiograms, including the standard limb and augmented unipolar extremity leads, were taken with a Sanborn Viso-Cardiette. In order to avoid axis shifts, the animals were maintained in a fixed position and electrocardiograms were taken in the same phase of respiration. Electrodes were not shifted during the course of an experiment. Arterial blood was obtained from an indwelling needle in the femoral artery. Blood was drawn into an oiled syringe containing potassium oxalate, and agitated with a small quantity of mercury. A part of this blood was transferred to a Van Slyke pipet for determination of ph; the remainder was introduced into a test tube under oil, sealed with paraffin, immediately centrifuged and refrigerated for to 2 hours before determinations of carbon dioxide were made. Heparinized blood samples were analyzed 206 Circulation Research, Volume I, Mav 053
2 ATHLEEN E. ROBERTS AND MELVILLE G. MAGIDA 207 for sodium and potassium with an internal standard flame photometer. Chloride was determined by Van Slyke and iller's modification of Sendroy's method and total carbon dioxide by the method of Van Slyke and Neill. Whole blood ph was determined with a Cambridge condensor type glass electrode surrounded by a constant temperature water jacket at 37 C. Bicarbonate was calculated from the total carbon dioxide and ph utilizing the Henderson-Hasselbach equation. same general pattern described by others. " 8 In order of appearance they consisted of an initial tachycardia, augmentation or peaking of the T wave, obliteration of the ST segment with the T wave arising from the S wave, bradycardia with loss of the P wave, development of a nodal rhythm or ventricular origin of the cardiac impulse with increased duration ph BHCQ, Li > j FTP' I -V- A.! \ J "f -A ] - Fio.. Effect of the Infusion of Potassium Chloride. Each period represents an interval of 40 minutes. Duration of infusion 70 minutes. RESULTS Infusion of Potassium Chloride. Figure illustrates the typical alterations in plasma electrolytes and the electrocardiogram which result from the infusion of potassium chloride at a rate of 0.6 meq. per minute. Similar results were obtained in four additional experiments. The increase in potassium concentration was associated with decreases in plasma bicarbonate and ph, and a small, but definite, fall in sodium concentration. The electrocardiographic alterations followed the -a T A of QRS and T. With continuation of the infusion, bizarre ventricular complexes appeared. Infusion of Lithium Chloride. Because of its similarity to potassium, both as to chemical properties and its influence upon the isolated nerve preparation, and because of its known clinical toxicity, the effect of lithium chloride was examined in relation to the electrocardiogram and electrolyte structure of the body fluids in four dogs. Figure 2 illustrates the similar effects of this ion and of potassium upon the electrocardiogram and the plasma electro-
3 i 208 EFFECT OF DECREASED PH, HCO3, NA AND ON ECG lyte pattern. In these experiments lithium chloride was infused at the rate of 0.95 meq. per minute. The plasma alterations in ph, bicarbonate, and sodium are similar to, but more profound than those produced by the infusion of potassium chloride. Especially striking is the appearance of electrocardiographic changes in the absence of significant ph BHCO: results were obtained in three additional experiments. As anticipated, there was a marked fall in plasma ph and bicarbonate concentration, accompanied by progressive electrocardiographic alterations similar to those seen in figures. and 2. We were unable to demonstrate, however, what specific influences the decreased ph and bicarbonate might have on k \ \r II m FIG. 2. Effect of the Infusion of Lithium Chloride. Each period represents an interval of 40 minutes. Duration of infusion 200 minutes. alteration in the plasma concentration of potassium. Infusion of Ammonium Chloride, Hydrochloric Acid and, Sulfuric Acid. Common to the infusion of either potassium or lithium chloride is a fall in plasma ph and bicarbonate. For this reason, it seemed appropriate to produce an acidosis by the infusion of ammonium chloride, hydrochloric or sulfuric acid and to examine the effects of these agents upon the electrocardiogram. The typical alterations produced by the infusion of ammonium chloride at a rate of.25 meq. per minute are illustrated in figure 3. Similar V. r, %i" i, V.-' the electrocardiogram, since there was a concomitant fall in plasma sodium and a rise in potassium. The specific influence upon the electrocardiogram of a decrease in the ph and bicarbonate of the extracellular compartment is better illustrated by the infusion of hydrochloric acid at a rate of.3 meq. per minute. Figure 4 illustrates the observation, as noted in 0 experiments, that the infusion of hydrochloric acid may produce electrocardiographic changes which are similar to those produced by the infusion of potassium chloride. It is worthy of note that these changes may be unassoci- f A i;
4 BHCQ 7.4 Z O TERMINAL -A. Fio. 3. Effect of the Infusion of Ammonium Chloride. Each period represents an interval of 40 minutes. Duration of infusion 40 minutes. ph BHCQ320.5 n I I ! 4.00 n Fia. 4. Effect of the Infusion of Hydrochloric Acid. Each period represents an interval of 20 minutes. Duration of infusion 0 minutes.
5 20 EFFECT OF DECREASED rh, HCO,, NA AND ON ECG ated with alterations in plasma potassium, and that the changes in sodium are of minor degree. Since antecedent alterations were produced by the infusion of the chloride ion in combination with potassium, lithium, ammonium, or hydrogen, one might infer that this ion was was induced in two dogs. The inhalation of 20 to 60 per cent carbon dioxide produced more striking decrements in ph than were produced by any of the aforementioned technics. Yet, as is evident in figure 0, only minor alterations in the electrocardiogram developed, and little ph BHCQ, m m ti jjji i 4 >l!! i v V v \ I -, -i \.. FIG. 5. Effect of the Infusion of Sulfuric Acid. Each period represents nn interval of 30 minutes. Duration of infusion 65 minutes. responsible for the production of the plasma and electrocardiographic alterations. To eliminate this possibility, sulfuric acid was infused in two experiments at a rate of 0.95 meq. per minute. Such an experiment is presented in figure 5. Examination of the data and records reveals that the same plasma and electrocardiographic alterations may be produced without the infusion of exogenous chloride. Resjriratory Acidosis. In order to determine whether the electrocardiographic changes, described above, result from alterations in plasma ph alone, a severe respiratory acidosis \ r by way of a sequential pattern of change was noted, despite the marked fall in plasma ph to The Influence of the Rale of Injection. Of major importance is the rate at which the various substances are infused. The administration of small total amounts of these solutions at rapid rates was sufficient to produce immediate death in the animal. Under these circumstances the animals frequently developed a terminal electrocardiographic picture without concomitant alteration in plasma electrolytes or the sequential development of electrocardiographic abnormalities, as have
6 ATHLEEN E. ROBERTS AND MELVILLE G. MAGLDA 2 been described. Only with infusion rates such that the infused substances did not reach the heart in amounts greater than.3 meq. per minute were we able to demonstrate progressive alterations in plasma electrolytes and the electrocardiogram. external concentration of this ion or of the lithium ion results in depolarization of the cell. 8 ' 0 Further, the action potential as well as the conduction of nervous impulses is dependent upon the maintenance of an adequate sodium concentration in the extracellular H HI DISCUSSION From the data presented it may be seen that electrocardiographic abnormalities similar to those ascribed to hyperkalemia may be produced by alterations in the plasma concentration of sodium, bicarbonate and hydrogen ion, with or without a concomitant change in the concentration of potassium. It is well established that the concentration gradients of potassium and sodium which exist across the cell membrane are of paramount importance in the maintenance of cellular activity. 9 ' 0 Potassium, is implicated in the maintenance of the resting potential; an elevation of the FIG. 6. Effect of Respiratory Acidosis. fluid. 9 ' 0 Evidence for similar alterations in myocardial units of the intact organism may be deduced from the finding that potassium and lithium produce similar changes in the electrocardiogram. Studies on the distribution of these ions following their infusion into dogs have shown that both enter the intracellular compartment. ' 2 In the case of potassium, extracellular bicarbonate accompanies the intracellular shift, resulting in an increase in the intracellular ph. Results presented in figure 2 suggest that a like transfer of bicarbonate ions occurs with the infusion of lithium chloride. It has been pointed out that the infusion
7 22 EFFECT OF DECREASED i>h, HCO 3, NA AND ON ECG of either lithium or potassium is accompanied by a fall in plasma sodium concentration. That this alteration in plasma sodium may be responsible, in part, for the alterations in cellular function cannot be refuted. Although little is known concerning the influence of hydrogen ion concentration on the bio-electric properties of the cell, certain deductions may be made. It has been established that the intracellular ph is maintained close to a value of 6.8; in the extracellular compartment the concentration of bicarbonate and carbonic acid are such that the ph of this compartment is maintained at the normal of 7.4.' 3 Thus, a hydrogen ion gradient is established across the cell membrane. A change in the extracellular ph from 7.4 to 7.0 approaches the maximum compatible with life. From this it would appear that alterations in the hydrogen ion gradient may influence the electric potentials upon which the fundamental properties of nerve and muscle depend. The infusion of ammonium chloride, hydrochloric acid or sulfuric acid causes a decrease of ph and bicarbonate which evokes an alteration of the hydrogen ion gradient between the extra- and intracellular compartments. No alteration in extracellular concentration of potassium need be implicated as seen from the finding that electrocardiographic aberrations may be produced, as in the case of hydrochloric acid infusion, without concomitant alteration in the concentration of potassium. Although the absence of electrocardiographic abnormalities in the presence of respiratory acidosis would seem incompatible with the thesis that alterations in ph influence cardiac function, this need not be the case. Carbon dioxide permeates all cells of the body and rapid equilibrium is achieved between the extra and intracellular compartments. Since no change occurred in those other ions which were measured, it is conceivable that little alteration in ionic gradient occurred across the cell membrane under the conditions of these experiments. Since carbon dioxide has been reported to repolarize a nerve previously depolarized with potassium, 9 it would be illogical to presume that similar alterations would be produced by carbon dioxide and potassium in the myocardium of the intact animal. The defect in myocardial function which can be implicated in sodium loss is a dual one. As has been pointed out, the action potential in an isolated nerve is dependent upon the concentration of sodium in the extracellular fluid; a decrease in the concentration of this ion is accompanied by a progressive change in action potential, with eventual loss of excitability of the cell. In addition, this ion represents available base in the bicarbonate buffer system. The drainage of this cation which is responsible for the maintenance of ph stability in the extracellular compartment leads to a profound acidosis. Clinical Implications The electrolyte and electrocardiographic abnormalities which were experimentally induced have counterparts in the clinical conditions of diabetic acidosis, adrenal and renal insufficiency and in cases of salt depletion or deprivation. Although the alterations in the acid-base picture in these clinical situations are infinitely more complex than the experimentally induced alterations by which each component has been studied, it is conceivable that certain similarities and interrelationships may be mutually shared. In all situations, experimental or clinical, there is evidence to indicate an alteration in the concentration gradient across the myocardial cell which involves sodium, potassium, bicarbonate, and hydrogen ion and perhaps other components which were not measured by us. Since myocardial function is ultimately dependent upon processes by which depolarization and repolarization occur in sequence with systole and diastole, severe alterations in electric potential must eventuate in cardiac failure. Although it is not possible in these experiments to indict any single alteration in ionic concentration, we infer that the alteration in plasma potassium is not alone responsible for the electrocardiographic alterations which have been described in conjunction with those clinical states in which hyperkalemia is a prominent finding.
8 ATHLEEN E. ROBERTS AND MELVILLE G. MAG IDA 2.3 CONCLUSIONS. Similar elcctrocardiographic alterations can be produced by the infusion of potassium chloride, lithium chloride, ammonium chloride, hydrochloric and sulfuric acid. 2. These electrocardiographic alterations are similar in form and development to those previously ascribed to hyperkalemia, but may be produced with or without an elevation of plasma potassium. ACNOWLEDGMENT We are grateful to Dr. Robert F. Pitts for his helpful advice and assistance in this project. REFERENCES MATIN, H. E., AND WEHTMAN, M.: Electrolyte changes and the electrocardiogram in diabetic acidosis. Am. Heart J. 34: 646, MEIULL, J. P., LEVINE, H. D., SOMERVILLE, W., AND SMITH, S.: Clinical recognition and treatment of acute potassium intoxication in man. Ann. Int. Med. 33: 97, LEVINE, H. D., AND MEILL, J. P.: Advanced disturbances of the cardiac mechanism in potassium intoxication in man. Circulation 3: 889, DAIUIOW, D. C: Body fluid physiology: The role of potassium in clinical disturbances of body water and electrolytes. New England J. Med. 242: 978, 950; 242: 04, NADLER, C. S., BELLET, S., REINHOLD, J. G., AND LANNING, H.: Alterations in the serum potassium and their relations to certain constituents of the blood in diabetic acidosis. Am. J. M. Sc. 28: 30S, J945. c FINCH, C. A., SAWYER, C. G., AND FLYNN, J. M.: The clinical syndrome of potassium intoxication. Am. J. Med.'l: 337, EITH, M. M., OSTERBERG, A. E., AND BURCHALI,, H. B.: Some effects of potassium salts in man. Ann. Int. Med. 6: 879, AVINLEU, W. S., HOFF, H. E., AND SMITH, P..: Electrocardiographic changes and the concentration of serum potassium following the intravenous injection of potassium chloride. Am. J. Physiol. 24: 47S, LORENTE DE No, R.: Studies from the Rockefeller Institute for Medical Research. Vol..3, HODGIN, A. L.: The ionic basis of electrical activity in nerve and muscle. Biol. Rev. 26:, 95. FOXJLS, J., MUDGE, G. II., AND GlLMAN, A.: Renal excretion of cation in the clog during infusion of isotonic solutions of lithium chloride. Am. J. Physiol. 68: 042, BOUHDILLON, J.: Distribution in body fluids and excretion of ingested ammonium chloride, potassium chloride, and sodium chloride. Am. J. Physiol. 20: 4, PETERS, J. P., AND VAN SLYE, D. D.: Quantitative clinical chemistry. Vol., Army Edition. Baltimore, Williams & Wilkins, 943.
WATER, SODIUM AND POTASSIUM
 WATER, SODIUM AND POTASSIUM Attila Miseta Tamás Kőszegi Department of Laboratory Medicine, 2016 1 Average daily water intake and output of a normal adult 2 Approximate contributions to plasma osmolality
WATER, SODIUM AND POTASSIUM Attila Miseta Tamás Kőszegi Department of Laboratory Medicine, 2016 1 Average daily water intake and output of a normal adult 2 Approximate contributions to plasma osmolality
Principles of Anatomy and Physiology
 Principles of Anatomy and Physiology 14 th Edition CHAPTER 27 Fluid, Electrolyte, and Acid Base Fluid Compartments and Fluid In adults, body fluids make up between 55% and 65% of total body mass. Body
Principles of Anatomy and Physiology 14 th Edition CHAPTER 27 Fluid, Electrolyte, and Acid Base Fluid Compartments and Fluid In adults, body fluids make up between 55% and 65% of total body mass. Body
A. Incorrect! The left ventricle receives oxygenated blood from the lungs via the left atrium.
 Anatomy and Physiology - Problem Drill 16: The Cardiovascular System No. 1 of 10 Instruction: (1) Read the problem statement and answer choices carefully (2) Work the problems on paper as needed (3) Pick
Anatomy and Physiology - Problem Drill 16: The Cardiovascular System No. 1 of 10 Instruction: (1) Read the problem statement and answer choices carefully (2) Work the problems on paper as needed (3) Pick
ELECTROCARDIOGRAPHY (ECG)
 ELECTROCARDIOGRAPHY (ECG) The heart is a muscular organ, which pumps blood through the blood vessels of the circulatory system. Blood provides the body with oxygen and nutrients, as well as assists in
ELECTROCARDIOGRAPHY (ECG) The heart is a muscular organ, which pumps blood through the blood vessels of the circulatory system. Blood provides the body with oxygen and nutrients, as well as assists in
NORMOSOL -R MULTIPLE ELECTROLYTES INJECTION TYPE 1, USP For Replacing Acute Losses of Extracellular Fluid Flexible Plastic Container
 NORMOSOL -R MULTIPLE ELECTROLYTES INJECTION TYPE 1, USP For Replacing Acute Losses of Extracellular Fluid Flexible Plastic Container R x only DESCRIPTION Normosol-R is a sterile, nonpyrogenic isotonic
NORMOSOL -R MULTIPLE ELECTROLYTES INJECTION TYPE 1, USP For Replacing Acute Losses of Extracellular Fluid Flexible Plastic Container R x only DESCRIPTION Normosol-R is a sterile, nonpyrogenic isotonic
Acid Base Balance. Professor Dr. Raid M. H. Al-Salih. Clinical Chemistry Professor Dr. Raid M. H. Al-Salih
 Acid Base Balance 1 HYDROGEN ION CONCENTRATION and CONCEPT OF ph Blood hydrogen ion concentration (abbreviated [H + ]) is maintained within tight limits in health, with the normal concentration being between
Acid Base Balance 1 HYDROGEN ION CONCENTRATION and CONCEPT OF ph Blood hydrogen ion concentration (abbreviated [H + ]) is maintained within tight limits in health, with the normal concentration being between
Major intra and extracellular ions Lec: 1
 Major intra and extracellular ions Lec: 1 The body fluids are solutions of inorganic and organic solutes. The concentration balance of the various components is maintained in order for the cell and tissue
Major intra and extracellular ions Lec: 1 The body fluids are solutions of inorganic and organic solutes. The concentration balance of the various components is maintained in order for the cell and tissue
CHAPTER 27 LECTURE OUTLINE
 CHAPTER 27 LECTURE OUTLINE I. INTRODUCTION A. Body fluid refers to body water and its dissolved substances. B. Regulatory mechanisms insure homeostasis of body fluids since their malfunction may seriously
CHAPTER 27 LECTURE OUTLINE I. INTRODUCTION A. Body fluid refers to body water and its dissolved substances. B. Regulatory mechanisms insure homeostasis of body fluids since their malfunction may seriously
Acid-Base Balance 11/18/2011. Regulation of Potassium Balance. Regulation of Potassium Balance. Regulatory Site: Cortical Collecting Ducts.
 Influence of Other Hormones on Sodium Balance Acid-Base Balance Estrogens: Enhance NaCl reabsorption by renal tubules May cause water retention during menstrual cycles Are responsible for edema during
Influence of Other Hormones on Sodium Balance Acid-Base Balance Estrogens: Enhance NaCl reabsorption by renal tubules May cause water retention during menstrual cycles Are responsible for edema during
The Electrocardiogram
 The Electrocardiogram Chapters 11 and 13 AUTUMN WEDAN AND NATASHA MCDOUGAL The Normal Electrocardiogram P-wave Generated when the atria depolarizes QRS-Complex Ventricles depolarizing before a contraction
The Electrocardiogram Chapters 11 and 13 AUTUMN WEDAN AND NATASHA MCDOUGAL The Normal Electrocardiogram P-wave Generated when the atria depolarizes QRS-Complex Ventricles depolarizing before a contraction
Normosol -R and 5% Dextrose Injection MULTIPLE ELECTROLYTES AND 5% DEXTROSE INJECTION TYPE 1, USP For Replacing Acute Losses of Extracellular Fluid
 Normosol -R and 5% Dextrose Injection MULTIPLE ELECTROLYTES AND 5% DEXTROSE INJECTION TYPE 1, USP For Replacing Acute Losses of Extracellular Fluid R x only Flexible Plastic Container DESCRIPTION Normosol-R
Normosol -R and 5% Dextrose Injection MULTIPLE ELECTROLYTES AND 5% DEXTROSE INJECTION TYPE 1, USP For Replacing Acute Losses of Extracellular Fluid R x only Flexible Plastic Container DESCRIPTION Normosol-R
Electrolyte Imbalance and Resuscitation. Dr. Mehmet Okumuş Sütçü Imam University Faculty of Medicine Department of Emergency Medicine
 Electrolyte Imbalance and Resuscitation Dr. Mehmet Okumuş Sütçü Imam University Faculty of Medicine Department of Emergency Medicine Presentation plan Definition of the electrolyte disturbances Conditions
Electrolyte Imbalance and Resuscitation Dr. Mehmet Okumuş Sütçü Imam University Faculty of Medicine Department of Emergency Medicine Presentation plan Definition of the electrolyte disturbances Conditions
MYOCARDIAL INFARCTION AND PREPONDERANCE OF A VENTRICLE
 POTASSIUM EFFECTS ON T WAVE INVERSION IN MYOCARDIAL INFARCTION AND PREPONDERANCE OF A VENTRICLE BY E. P. SHARPEY-SCHAFER From the Department of Medicine, British Postgraduate Medical School, London Received
POTASSIUM EFFECTS ON T WAVE INVERSION IN MYOCARDIAL INFARCTION AND PREPONDERANCE OF A VENTRICLE BY E. P. SHARPEY-SCHAFER From the Department of Medicine, British Postgraduate Medical School, London Received
Normal range of serum potassium is meq/l true hyperkalemia manifests clinically as : Clinical presentation : muscle and cardiac dysfunction
 Potassium Disorders hyperkalemia Potassium is mainly an cation? What is the major physiological role of potassium in the body? What is the major regulatory system of serum potassium level? Which part of
Potassium Disorders hyperkalemia Potassium is mainly an cation? What is the major physiological role of potassium in the body? What is the major regulatory system of serum potassium level? Which part of
Basic Fluid and Electrolytes
 Basic Fluid and Electrolytes Chapter 22 Basic Fluid and Electrolytes Introduction Infants and young children have a greater need for water and are more vulnerable to alterations in fluid and electrolyte
Basic Fluid and Electrolytes Chapter 22 Basic Fluid and Electrolytes Introduction Infants and young children have a greater need for water and are more vulnerable to alterations in fluid and electrolyte
Chapter 26 Fluid, Electrolyte, and Acid- Base Balance
 Chapter 26 Fluid, Electrolyte, and Acid- Base Balance 1 Body Water Content Infants: 73% or more water (low body fat, low bone mass) Adult males: ~60% water Adult females: ~50% water (higher fat content,
Chapter 26 Fluid, Electrolyte, and Acid- Base Balance 1 Body Water Content Infants: 73% or more water (low body fat, low bone mass) Adult males: ~60% water Adult females: ~50% water (higher fat content,
Emergency Medical Training Services Emergency Medical Technician Paramedic Program Outlines Outline Topic: Patho Instructor Notes Revised: 11/2013
 Emergency Medical Training Services Emergency Medical Technician Paramedic Program Outlines Outline Topic: Patho Instructor Notes Revised: 11/2013 Cells form 4 basic tissue groups: 1. Epithelial 2. Connective
Emergency Medical Training Services Emergency Medical Technician Paramedic Program Outlines Outline Topic: Patho Instructor Notes Revised: 11/2013 Cells form 4 basic tissue groups: 1. Epithelial 2. Connective
following experiments were designed to show the effects of changes in CO2 combining power in the blood of dogs after the administration of acid
 OBSERVATIONS ON THE FORMATION OF WHEALS V. THE EFFECTS OF VARIATION OF THE CO2 COMBINING POWER OF THE BLOOD ON HISTAMINE WHEALS By F. S. McCONNELL, W. K. WEAVER AND H. L. ALEXANDER (From the Department
OBSERVATIONS ON THE FORMATION OF WHEALS V. THE EFFECTS OF VARIATION OF THE CO2 COMBINING POWER OF THE BLOOD ON HISTAMINE WHEALS By F. S. McCONNELL, W. K. WEAVER AND H. L. ALEXANDER (From the Department
Chapter 9, Part 2. Cardiocirculatory Adjustments to Exercise
 Chapter 9, Part 2 Cardiocirculatory Adjustments to Exercise Electrical Activity of the Heart Contraction of the heart depends on electrical stimulation of the myocardium Impulse is initiated in the right
Chapter 9, Part 2 Cardiocirculatory Adjustments to Exercise Electrical Activity of the Heart Contraction of the heart depends on electrical stimulation of the myocardium Impulse is initiated in the right
Chapter 27: WATER, ELECTROLYTES, AND ACID-BASE BALANCE
 Chapter 27: WATER, ELECTROLYTES, AND ACID-BASE BALANCE I. RELATED TOPICS Integumentary system Cerebrospinal fluid Aqueous humor Digestive juices Feces Capillary dynamics Lymph circulation Edema Osmosis
Chapter 27: WATER, ELECTROLYTES, AND ACID-BASE BALANCE I. RELATED TOPICS Integumentary system Cerebrospinal fluid Aqueous humor Digestive juices Feces Capillary dynamics Lymph circulation Edema Osmosis
Boston, Mass.) per L. and the bicarbonate 35 meq. per L. The bath was maintained at approximately ph 7.4 by equilibration
 CHANGES IN ACID-BASE BALANCE OF UREMIC PATIENTS DURING HEMODIALYSIS1 By JOHN M. WELLER,2 ROY C. SWAN,3 AND JOHN P. MERRILL 4 (From the Department of Medicine, Peter Bent Brigham Hospital and Harvard Medical
CHANGES IN ACID-BASE BALANCE OF UREMIC PATIENTS DURING HEMODIALYSIS1 By JOHN M. WELLER,2 ROY C. SWAN,3 AND JOHN P. MERRILL 4 (From the Department of Medicine, Peter Bent Brigham Hospital and Harvard Medical
Objectives. Blood Buffers. Definitions. Strong/Weak Acids. Fixed (Non-Volatile) Acids. Module H Malley pages
 Blood Buffers Module H Malley pages 120-126 Objectives Define a buffer system and differentiate between the buffering systems present in the body. Given an arterial blood-gas result, determine the degree
Blood Buffers Module H Malley pages 120-126 Objectives Define a buffer system and differentiate between the buffering systems present in the body. Given an arterial blood-gas result, determine the degree
Principles of Fluid Balance
 Principles of Fluid Balance I. The Cellular Environment: Fluids and Electrolytes A. Water 1. Total body water (TBW) = 60% of total body weight 2. Fluid Compartments in the Body a. Intracellular Compartment
Principles of Fluid Balance I. The Cellular Environment: Fluids and Electrolytes A. Water 1. Total body water (TBW) = 60% of total body weight 2. Fluid Compartments in the Body a. Intracellular Compartment
a lecture series by SWESEMJR
 Electrolyte disturbances Hypokalaemia Decreased extracellular potassium increases excitability in the myocardial cells and consequently the effect of very severe hypokalaemia is ventricular arrhythmia.
Electrolyte disturbances Hypokalaemia Decreased extracellular potassium increases excitability in the myocardial cells and consequently the effect of very severe hypokalaemia is ventricular arrhythmia.
The Induction of Metabolic Alkalosis by Correction of Potassium Deficiency *
 lournal of Clinical Investigation Vol. 45, No. 4, 1966 The Induction of Metabolic Alkalosis by Correction of Potassium Deficiency * HOWARD L. BLEICH,t RICHARD L. TANNEN,t AND WILLIAM B. SCHWARTZ t (From
lournal of Clinical Investigation Vol. 45, No. 4, 1966 The Induction of Metabolic Alkalosis by Correction of Potassium Deficiency * HOWARD L. BLEICH,t RICHARD L. TANNEN,t AND WILLIAM B. SCHWARTZ t (From
CASE 27. What is the response of the kidney to metabolic acidosis? What is the response of the kidney to a respiratory alkalosis?
 CASE 27 A 21-year-old man with insulin-dependent diabetes presents to the emergency center with mental status changes, nausea, vomiting, abdominal pain, and rapid respirations. On examination, the patient
CASE 27 A 21-year-old man with insulin-dependent diabetes presents to the emergency center with mental status changes, nausea, vomiting, abdominal pain, and rapid respirations. On examination, the patient
Part 1 The Cell and the Cellular Environment
 1 Chapter 3 Anatomy and Physiology Part 1 The Cell and the Cellular Environment 2 The Human Cell The is the fundamental unit of the human body. Cells contain all the necessary for life functions. 3 Cell
1 Chapter 3 Anatomy and Physiology Part 1 The Cell and the Cellular Environment 2 The Human Cell The is the fundamental unit of the human body. Cells contain all the necessary for life functions. 3 Cell
INTRAVENOUS SOLUTIONS WITH POTASSIUM CHLORIDE POTASSIUM CHLORIDE IN LACTATED RINGER S AND 5% DEXTROSE INJECTION, USP Flexible Plastic Container
 INTRAVENOUS SOLUTIONS WITH POTASSIUM CHLORIDE POTASSIUM CHLORIDE IN LACTATED RINGER S AND 5% DEXTROSE INJECTION, USP Flexible Plastic Container R x only DESCRIPTION Intravenous solutions with potassium
INTRAVENOUS SOLUTIONS WITH POTASSIUM CHLORIDE POTASSIUM CHLORIDE IN LACTATED RINGER S AND 5% DEXTROSE INJECTION, USP Flexible Plastic Container R x only DESCRIPTION Intravenous solutions with potassium
The equilibrium between basis and acid can be calculated and termed as the equilibrium constant = Ka. (sometimes referred as the dissociation constant
 Acid base balance Dobroslav Hájek dhajek@med.muni.cz May 2004 The equilibrium between basis and acid can be calculated and termed as the equilibrium constant = Ka. (sometimes referred as the dissociation
Acid base balance Dobroslav Hájek dhajek@med.muni.cz May 2004 The equilibrium between basis and acid can be calculated and termed as the equilibrium constant = Ka. (sometimes referred as the dissociation
Acid-base balance is one of the most important of the body s homeostatic mechanisms Acid-base balance refers to regulation of hydrogen ion (H + )
 Acid-base balance is one of the most important of the body s homeostatic mechanisms Acid-base balance refers to regulation of hydrogen ion (H + ) concentration in body fluids Precise regulation of ph at
Acid-base balance is one of the most important of the body s homeostatic mechanisms Acid-base balance refers to regulation of hydrogen ion (H + ) concentration in body fluids Precise regulation of ph at
Mathematical modeling of ischemia and infarction
 Mathematical modeling of ischemia and infarction Mostly based on Cimponeriu, Starmer and Bezerianos: A theoretical analysis of acute ischemia and infarction using ECG reconstruction on a 2-D model of myocardium
Mathematical modeling of ischemia and infarction Mostly based on Cimponeriu, Starmer and Bezerianos: A theoretical analysis of acute ischemia and infarction using ECG reconstruction on a 2-D model of myocardium
Sodium Bicarbonate Injection, USP
 Sodium Bicarbonate Injection, USP FOR THE CORRECTION OF METABOLIC ACIDOSIS AND OTHER CONDITIONS REQUIRING SYSTEMIC ALKALINIZATION. Plastic Ansyr II Syringe R x only DESCRIPTION Sodium Bicarbonate Injection,
Sodium Bicarbonate Injection, USP FOR THE CORRECTION OF METABOLIC ACIDOSIS AND OTHER CONDITIONS REQUIRING SYSTEMIC ALKALINIZATION. Plastic Ansyr II Syringe R x only DESCRIPTION Sodium Bicarbonate Injection,
Water, Electrolytes, and Acid-Base Balance
 Chapter 27 Water, Electrolytes, and Acid-Base Balance 1 Body Fluids Intracellular fluid compartment All fluids inside cells of body About 40% of total body weight Extracellular fluid compartment All fluids
Chapter 27 Water, Electrolytes, and Acid-Base Balance 1 Body Fluids Intracellular fluid compartment All fluids inside cells of body About 40% of total body weight Extracellular fluid compartment All fluids
ECG and Cardiac Electrophysiology
 ECG and Cardiac Electrophysiology Simon Some very basic electrophysiology Intracellular fluid: 10 mm Na, 140 mm K, etc. K Na-K ATPase Extracellular fluid: 140mM Na, 4mM K, etc. Na Ion gradient plus selective
ECG and Cardiac Electrophysiology Simon Some very basic electrophysiology Intracellular fluid: 10 mm Na, 140 mm K, etc. K Na-K ATPase Extracellular fluid: 140mM Na, 4mM K, etc. Na Ion gradient plus selective
Chapter 20 8/23/2016. Fluids and Electrolytes. Fluid (Water) Fluid (Water) (Cont.) Functions
 Chapter 20 Fluids and Electrolytes All items and derived items 2015, 2011, 2006 by Mosby, Inc., an imprint of Elsevier Inc. All rights reserved. Fluid (Water) Functions Provides an extracellular transportation
Chapter 20 Fluids and Electrolytes All items and derived items 2015, 2011, 2006 by Mosby, Inc., an imprint of Elsevier Inc. All rights reserved. Fluid (Water) Functions Provides an extracellular transportation
during the 8 days prior to the first intraperitoneal
 EXPERIMENTAL HYPERTONICITY: ALTERATIONS IN THE DISTRIBUTION OF BODY WATER, AND THE CAUSE OF DEATH 1 By ALEXANDER W. WINKLER, J. RUSSELL ELKINTON,2 JAMES HOPPER, JR.,8 AND HEBBEL E. HOFF (From the Department
EXPERIMENTAL HYPERTONICITY: ALTERATIONS IN THE DISTRIBUTION OF BODY WATER, AND THE CAUSE OF DEATH 1 By ALEXANDER W. WINKLER, J. RUSSELL ELKINTON,2 JAMES HOPPER, JR.,8 AND HEBBEL E. HOFF (From the Department
HUMAN SUBJECT 1. Syracuse, N. Y.) the urine of increasing quantities of these buffers, it has been found in man as in the dog that (1)
 THE RENAL REGULATION OF ACID-BASE BALANCE IN MAN. II. FACTORS AFFECTING THE EXCRETION OF TITRATABLE ACID BY THE NORMAL HUMAN SUBJECT 1 By W. A. SCHIESS, J. L. AYER, W. D. LOTSPEICH AND R. F. PITTS WITH
THE RENAL REGULATION OF ACID-BASE BALANCE IN MAN. II. FACTORS AFFECTING THE EXCRETION OF TITRATABLE ACID BY THE NORMAL HUMAN SUBJECT 1 By W. A. SCHIESS, J. L. AYER, W. D. LOTSPEICH AND R. F. PITTS WITH
HIGH voltage of R waves in left precordial leads of the electrocardiogram
 R-WAVE VOLTAGE CHANGES IN RELATION TO ARTERIAL PRESSURE IN HYPERTENSIVE PATIENTS ATHAN J. GEORGOPOULOS, M.D.,* PHILIP A. VLASTARIS, M.D.,f and WILLIAM L. PROUDFIT, M.D. Department of Cardiovascular Disease
R-WAVE VOLTAGE CHANGES IN RELATION TO ARTERIAL PRESSURE IN HYPERTENSIVE PATIENTS ATHAN J. GEORGOPOULOS, M.D.,* PHILIP A. VLASTARIS, M.D.,f and WILLIAM L. PROUDFIT, M.D. Department of Cardiovascular Disease
Chapter 19 The Urinary System Fluid and Electrolyte Balance
 Chapter 19 The Urinary System Fluid and Electrolyte Balance Chapter Outline The Concept of Balance Water Balance Sodium Balance Potassium Balance Calcium Balance Interactions between Fluid and Electrolyte
Chapter 19 The Urinary System Fluid and Electrolyte Balance Chapter Outline The Concept of Balance Water Balance Sodium Balance Potassium Balance Calcium Balance Interactions between Fluid and Electrolyte
Acid-Base Balance Dr. Gary Mumaugh
 Acid-Base Balance Dr. Gary Mumaugh Introduction Acid-base balance is one of the most important of the body s homeostatic mechanisms Acid-base balance refers to regulation of hydrogen ion (H + ) concentration
Acid-Base Balance Dr. Gary Mumaugh Introduction Acid-base balance is one of the most important of the body s homeostatic mechanisms Acid-base balance refers to regulation of hydrogen ion (H + ) concentration
Managing Acid Base and Electrolyte Disturbances with RRT
 Managing Acid Base and Electrolyte Disturbances with RRT John R Prowle MA MSc MD MRCP FFICM Consultant in Intensive Care & Renal Medicine RRT for Regulation of Acid-base and Electrolyte Acid base load
Managing Acid Base and Electrolyte Disturbances with RRT John R Prowle MA MSc MD MRCP FFICM Consultant in Intensive Care & Renal Medicine RRT for Regulation of Acid-base and Electrolyte Acid base load
Complete Atrioventricular Block Due to Potassium
 Complete Atrioventricular Block Due to Potassium By Charles Fisch, M.D., Kalman Greenspan, Ph.D., and Robert E. Edmandi, M.D. ABSTRACT Intravenous infusion of KC1 at a rate of 1.22 or 2.45 meq per minute
Complete Atrioventricular Block Due to Potassium By Charles Fisch, M.D., Kalman Greenspan, Ph.D., and Robert E. Edmandi, M.D. ABSTRACT Intravenous infusion of KC1 at a rate of 1.22 or 2.45 meq per minute
Interrelationship of Potassium and Vagal Action on the
 fournal of Clinical Investigation Vol. 44, No. 3, 1965 Interrelationship of Potassium and Vagal Action on the Sinoatrial Pacemaker and on Atrioventricular Conduction * HARVEY FEIGENBAUM,t CHARLES M. WUNSCH,t
fournal of Clinical Investigation Vol. 44, No. 3, 1965 Interrelationship of Potassium and Vagal Action on the Sinoatrial Pacemaker and on Atrioventricular Conduction * HARVEY FEIGENBAUM,t CHARLES M. WUNSCH,t
Potassium Efflux from Myocardial Cells Induced by Defibrillator Shock
 Purdue University Purdue e-pubs Weldon School of Biomedical Engineering Faculty Publications Weldon School of Biomedical Engineering 1986 Potassium Efflux from Myocardial Cells Induced by Defibrillator
Purdue University Purdue e-pubs Weldon School of Biomedical Engineering Faculty Publications Weldon School of Biomedical Engineering 1986 Potassium Efflux from Myocardial Cells Induced by Defibrillator
ELECTROLYTE DISTURBANCES IN CONGESTIVE HEART FAILURE*
 ELECTROLYTE DISTURBANCES IN CONGESTIVE HEART FAILURE* DAVID P. B.AUMANN, M.D. Department of Medicine, University of Arkansas School of Medicine, Little Rock, Arkansas The retention of salt and water secondary
ELECTROLYTE DISTURBANCES IN CONGESTIVE HEART FAILURE* DAVID P. B.AUMANN, M.D. Department of Medicine, University of Arkansas School of Medicine, Little Rock, Arkansas The retention of salt and water secondary
Acids, Bases, and Salts
 Acid / Base Balance Objectives Define an acid, a base, and the measure of ph. Discuss acid/base balance, the effects of acidosis or alkalosis on the body, and the mechanisms in place to maintain balance
Acid / Base Balance Objectives Define an acid, a base, and the measure of ph. Discuss acid/base balance, the effects of acidosis or alkalosis on the body, and the mechanisms in place to maintain balance
Cardiovascular system
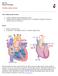 BIO 301 Human Physiology Cardiovascular system The Cardiovascular System: consists of the heart plus all the blood vessels transports blood to all parts of the body in two 'circulations': pulmonary (lungs)
BIO 301 Human Physiology Cardiovascular system The Cardiovascular System: consists of the heart plus all the blood vessels transports blood to all parts of the body in two 'circulations': pulmonary (lungs)
Fluid, Electrolyte, and Acid Base Balance
 25 Fluid, Electrolyte, and Acid Base Balance Lecture Presentation by Lori Garrett Note to the Instructor: For the third edition of Visual Anatomy & Physiology, we have updated our PowerPoints to fully
25 Fluid, Electrolyte, and Acid Base Balance Lecture Presentation by Lori Garrett Note to the Instructor: For the third edition of Visual Anatomy & Physiology, we have updated our PowerPoints to fully
Principles of Anatomy and Physiology
 Principles of Anatomy and Physiology 14 th Edition CHAPTER 20 The Cardiovascular System: The Heart Introduction The purpose of the chapter is to: 1. Learn about the components of the cardiovascular system
Principles of Anatomy and Physiology 14 th Edition CHAPTER 20 The Cardiovascular System: The Heart Introduction The purpose of the chapter is to: 1. Learn about the components of the cardiovascular system
STUDY ON MECHANISM OF ACID AND AMMONIA EXCRETION BY KIDNEY AFTER ACID LOAD. Hisato YOSHIMURA, Mamoru FUJIMOTO AND Junichi SUGIMOTO
 STUDY ON MECHANISM OF ACID AND AMMONIA EXCRETION BY KIDNEY AFTER ACID LOAD Hisato YOSHIMURA, Mamoru FUJIMOTO AND Junichi SUGIMOTO Department of Physiology, Kyoto Prefectural University of Medicine, Kyoto
STUDY ON MECHANISM OF ACID AND AMMONIA EXCRETION BY KIDNEY AFTER ACID LOAD Hisato YOSHIMURA, Mamoru FUJIMOTO AND Junichi SUGIMOTO Department of Physiology, Kyoto Prefectural University of Medicine, Kyoto
The Urinary System PART B
 15 The Urinary System PART B PowerPoint Lecture Slide Presentation by Jerry L. Cook, Sam Houston University ESSENTIALS OF HUMAN ANATOMY & PHYSIOLOGY EIGHTH EDITION ELAINE N. MARIEB Urinary Bladder Smooth,
15 The Urinary System PART B PowerPoint Lecture Slide Presentation by Jerry L. Cook, Sam Houston University ESSENTIALS OF HUMAN ANATOMY & PHYSIOLOGY EIGHTH EDITION ELAINE N. MARIEB Urinary Bladder Smooth,
Carbon Dioxide Transport. Carbon Dioxide. Carbon Dioxide Transport. Carbon Dioxide Transport - Plasma. Hydrolysis of Water
 Module H: Carbon Dioxide Transport Beachey Ch 9 & 10 Egan pp. 244-246, 281-284 Carbon Dioxide Transport At the end of today s session you will be able to : Describe the relationship free hydrogen ions
Module H: Carbon Dioxide Transport Beachey Ch 9 & 10 Egan pp. 244-246, 281-284 Carbon Dioxide Transport At the end of today s session you will be able to : Describe the relationship free hydrogen ions
Slide 1. Slide 2. Slide 3. Learning Outcomes. Acid base terminology ARTERIAL BLOOD GAS INTERPRETATION
 Slide 1 ARTERIAL BLOOD GAS INTERPRETATION David O Neill MSc BSc RN NMP FHEA Associate Lecturer (Non Medical Prescribing) Cardiff University Advanced Nurse Practitioner Respiratory Medicine Slide 2 Learning
Slide 1 ARTERIAL BLOOD GAS INTERPRETATION David O Neill MSc BSc RN NMP FHEA Associate Lecturer (Non Medical Prescribing) Cardiff University Advanced Nurse Practitioner Respiratory Medicine Slide 2 Learning
only. Analysis of human muscle obtained by biopsy suggests that changes
 A PROPOSED MECHANISM OF EXTRACELLLAR REGLATION OF MSCLE COMPOSITION* ROBERT E. COOKE** AND WILLIAM E. SEGAR The changes in muscle composition which occur in disturbances of extracellular electrolyte composition
A PROPOSED MECHANISM OF EXTRACELLLAR REGLATION OF MSCLE COMPOSITION* ROBERT E. COOKE** AND WILLIAM E. SEGAR The changes in muscle composition which occur in disturbances of extracellular electrolyte composition
Human Anatomy and Physiology - Problem Drill 23: The Urinary System, Fluid, Electrolyte and Acid-Base Balance
 Human Anatomy and Physiology - Problem Drill 23: The Urinary System, Fluid, Electrolyte and Acid-Base Balance Question No. 1 of 10 Which of the following statements about the functions of the urinary system
Human Anatomy and Physiology - Problem Drill 23: The Urinary System, Fluid, Electrolyte and Acid-Base Balance Question No. 1 of 10 Which of the following statements about the functions of the urinary system
EI2311 BIOMEDICAL INSTRUMENTATION
 66 EI2311 BIOMEDICAL INSTRUMENTATION 1. What is meant by cell? UNIT I PHYSIOLOGY AND TRANSDUCERS The basic living unit of the body is cell. The function of organs and other structure of the body is understood
66 EI2311 BIOMEDICAL INSTRUMENTATION 1. What is meant by cell? UNIT I PHYSIOLOGY AND TRANSDUCERS The basic living unit of the body is cell. The function of organs and other structure of the body is understood
1/3/2008. Karen Burke Priscilla LeMone Elaine Mohn-Brown. Medical-Surgical Nursing Care, 2e Karen Burke, Priscilla LeMone, and Elaine Mohn-Brown
 Medical-Surgical Nursing Care Second Edition Karen Burke Priscilla LeMone Elaine Mohn-Brown Chapter 7 Caring for Clients with Altered Fluid, Electrolyte, or Acid-Base Balance Water Primary component of
Medical-Surgical Nursing Care Second Edition Karen Burke Priscilla LeMone Elaine Mohn-Brown Chapter 7 Caring for Clients with Altered Fluid, Electrolyte, or Acid-Base Balance Water Primary component of
DBL MAGNESIUM SULFATE CONCENTRATED INJECTION
 DBL MAGNESIUM SULFATE CONCENTRATED INJECTION NAME OF MEDICINE Magnesium Sulfate BP DESCRIPTION DBL Magnesium Sulfate Concentrated Injection is a clear, colourless, sterile solution. Each ampoule contains
DBL MAGNESIUM SULFATE CONCENTRATED INJECTION NAME OF MEDICINE Magnesium Sulfate BP DESCRIPTION DBL Magnesium Sulfate Concentrated Injection is a clear, colourless, sterile solution. Each ampoule contains
The Physiologic Limits of the Defense of ph *
 lournal of Clinical Investigation Vol. 44, No. 2, 1965 The Response of Extracellular Hydrogen Ion Concentration to Graded Degrees of Chronic Hypercapnia: The Physiologic Limits of the Defense of ph * WILLIAM
lournal of Clinical Investigation Vol. 44, No. 2, 1965 The Response of Extracellular Hydrogen Ion Concentration to Graded Degrees of Chronic Hypercapnia: The Physiologic Limits of the Defense of ph * WILLIAM
Chapter 16 Nutrition, Fluids and Electrolytes, and Acid-Base Balance Nutrition Nutrients Water o Functions Promotes metabolic processes Transporter
 Chapter 16 Nutrition, Fluids and Electrolytes, and Acid-Base Balance Nutrition Nutrients Water o Functions Promotes metabolic processes Transporter for nutrients and wastes Lubricant Insulator and shock
Chapter 16 Nutrition, Fluids and Electrolytes, and Acid-Base Balance Nutrition Nutrients Water o Functions Promotes metabolic processes Transporter for nutrients and wastes Lubricant Insulator and shock
BIOL 2402 Fluid/Electrolyte Regulation
 Dr. Chris Doumen Collin County Community College BIOL 2402 Fluid/Electrolyte Regulation 1 Body Water Content On average, we are 50-60 % water For a 70 kg male = 40 liters water This water is divided into
Dr. Chris Doumen Collin County Community College BIOL 2402 Fluid/Electrolyte Regulation 1 Body Water Content On average, we are 50-60 % water For a 70 kg male = 40 liters water This water is divided into
SUMMARY OF PRODUCT CHARACTERISTICS 2. QUALITATIVE AND QUANTITATIVE COMPOSITION
 SUMMARY OF PRODUCT CHARACTERISTICS PRODUCT SUMMARY 1. NAME OF THE MEDICINAL PRODUCT Sterile Potassium Chloride Concentrate 15%. 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 15% of Potassium Chloride in
SUMMARY OF PRODUCT CHARACTERISTICS PRODUCT SUMMARY 1. NAME OF THE MEDICINAL PRODUCT Sterile Potassium Chloride Concentrate 15%. 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 15% of Potassium Chloride in
INTRODUCTION TO ECG. Dr. Tamara Alqudah
 INTRODUCTION TO ECG Dr. Tamara Alqudah Excitatory & conductive system of the heart + - The ECG The electrocardiogram, or ECG, is a simple & noninvasive diagnostic test which records the electrical
INTRODUCTION TO ECG Dr. Tamara Alqudah Excitatory & conductive system of the heart + - The ECG The electrocardiogram, or ECG, is a simple & noninvasive diagnostic test which records the electrical
2.5 per cent solution). A Magill endotracheal tube fitted. with an inflatable balloon was introduced into the trachea
 THE REGULATION OF RENAL BICARBONATE REABSORPTION BY PLASMA CARBON DIOXIDE TENSION ' By ARNOLD S. RELMAN, BENJAMIN ETSTEN, AND WILLIAM B. SCHWARTZ (From the Evans Memorial, Massachusetts Memorial Hospitals
THE REGULATION OF RENAL BICARBONATE REABSORPTION BY PLASMA CARBON DIOXIDE TENSION ' By ARNOLD S. RELMAN, BENJAMIN ETSTEN, AND WILLIAM B. SCHWARTZ (From the Evans Memorial, Massachusetts Memorial Hospitals
PART I. Disorders of the Heart Rhythm: Basic Principles
 PART I Disorders of the Heart Rhythm: Basic Principles FET01.indd 1 1/11/06 9:53:05 AM FET01.indd 2 1/11/06 9:53:06 AM CHAPTER 1 The Cardiac Electrical System The heart spontaneously generates electrical
PART I Disorders of the Heart Rhythm: Basic Principles FET01.indd 1 1/11/06 9:53:05 AM FET01.indd 2 1/11/06 9:53:06 AM CHAPTER 1 The Cardiac Electrical System The heart spontaneously generates electrical
There are number of parameters which are measured: ph Oxygen (O 2 ) Carbon Dioxide (CO 2 ) Bicarbonate (HCO 3 -) AaDO 2 O 2 Content O 2 Saturation
 Arterial Blood Gases (ABG) A blood gas is exactly that...it measures the dissolved gases in your bloodstream. This provides one of the best measurements of what is known as the acid-base balance. The body
Arterial Blood Gases (ABG) A blood gas is exactly that...it measures the dissolved gases in your bloodstream. This provides one of the best measurements of what is known as the acid-base balance. The body
PHYSIOLOGY MeQ'S (Morgan) All the following statements related to blood volume are correct except for: 5 A. Blood volume is about 5 litres. B.
 PHYSIOLOGY MeQ'S (Morgan) Chapter 5 All the following statements related to capillary Starling's forces are correct except for: 1 A. Hydrostatic pressure at arterial end is greater than at venous end.
PHYSIOLOGY MeQ'S (Morgan) Chapter 5 All the following statements related to capillary Starling's forces are correct except for: 1 A. Hydrostatic pressure at arterial end is greater than at venous end.
Solution for cardiac perfusion in viaflex plastic container
 CARDIOPLEGIA SOLUTION A Solution for cardiac perfusion in viaflex plastic container DESCRIPTION Cardioplegia Solution A is a sterile, non-pyrogenic solution in a Viaflex bag. It is used to induce cardiac
CARDIOPLEGIA SOLUTION A Solution for cardiac perfusion in viaflex plastic container DESCRIPTION Cardioplegia Solution A is a sterile, non-pyrogenic solution in a Viaflex bag. It is used to induce cardiac
Introduction. Acids, Bases and ph; a review
 0 P a g e Introduction In this sheet, we discuss acidbase balance in our body and the role of kidneys in its establishment. Arrangement of topics is different from that of the lecture, to assure consistency
0 P a g e Introduction In this sheet, we discuss acidbase balance in our body and the role of kidneys in its establishment. Arrangement of topics is different from that of the lecture, to assure consistency
PRESCRIBING INFORMATION. Sodium Bicarbonate Injection USP 42mg/ml (4.2%), 75mg/ml (7.5%), 84mg/ml (8.4%) Sterile solution ALKALIZER
 PRESCRIBING INFORMATION Sodium Bicarbonate Injection USP 42mg/ml (4.2%), 75mg/ml (7.5%), 84mg/ml (8.4%) Sterile solution Lifeshield ABBOJECT Single-dose Syringes and Flipp Vial ALKALIZER FOR CORRECTION
PRESCRIBING INFORMATION Sodium Bicarbonate Injection USP 42mg/ml (4.2%), 75mg/ml (7.5%), 84mg/ml (8.4%) Sterile solution Lifeshield ABBOJECT Single-dose Syringes and Flipp Vial ALKALIZER FOR CORRECTION
4. The two inferior chambers of the heart are known as the atria. the superior and inferior vena cava, which empty into the left atrium.
 Answer each statement true or false. If the statement is false, change the underlined word to make it true. 1. The heart is located approximately between the second and fifth ribs and posterior to the
Answer each statement true or false. If the statement is false, change the underlined word to make it true. 1. The heart is located approximately between the second and fifth ribs and posterior to the
Ventricular Tachycardia Basics
 Ventricular Tachycardia Basics OVERVIEW Ventricular refers to the ventricles of the heart; tachycardia is the medical term for rapid heart rate The heart of the dog or cat is composed of four chambers;
Ventricular Tachycardia Basics OVERVIEW Ventricular refers to the ventricles of the heart; tachycardia is the medical term for rapid heart rate The heart of the dog or cat is composed of four chambers;
Health Science 20 Circulatory System Notes
 Health Science 20 Circulatory System Notes Functions of the Circulatory System The circulatory system functions mainly as the body s transport system. It transports: o Oxygen o Nutrients o Cell waste o
Health Science 20 Circulatory System Notes Functions of the Circulatory System The circulatory system functions mainly as the body s transport system. It transports: o Oxygen o Nutrients o Cell waste o
Amjad Bani Hani Ass.Prof. of Cardiac Surgery & Intensive Care FLUIDS AND ELECTROLYTES
 Amjad Bani Hani Ass.Prof. of Cardiac Surgery & Intensive Care FLUIDS AND ELECTROLYTES Body Water Content Water Balance: Normal 2500 2000 1500 1000 500 Metab Food Fluids Stool Breath Sweat Urine
Amjad Bani Hani Ass.Prof. of Cardiac Surgery & Intensive Care FLUIDS AND ELECTROLYTES Body Water Content Water Balance: Normal 2500 2000 1500 1000 500 Metab Food Fluids Stool Breath Sweat Urine
Acid and Base Balance
 Acid and Base Balance 1 2 The Body and ph Homeostasis of ph is tightly controlled Extracellular fluid = 7.4 Blood = 7.35 7.45 < 7.35: Acidosis (acidemia) > 7.45: Alkalosis (alkalemia) < 6.8 or > 8.0: death
Acid and Base Balance 1 2 The Body and ph Homeostasis of ph is tightly controlled Extracellular fluid = 7.4 Blood = 7.35 7.45 < 7.35: Acidosis (acidemia) > 7.45: Alkalosis (alkalemia) < 6.8 or > 8.0: death
ECG Interpretation Cat Williams, DVM DACVIM (Cardiology)
 ECG Interpretation Cat Williams, DVM DACVIM (Cardiology) Providing the best quality care and service for the patient, the client, and the referring veterinarian. GOAL: Reduce Anxiety about ECGs Back to
ECG Interpretation Cat Williams, DVM DACVIM (Cardiology) Providing the best quality care and service for the patient, the client, and the referring veterinarian. GOAL: Reduce Anxiety about ECGs Back to
Acid - base equilibrium
 Acid base equilibrium ph concept ph = log [H + ] ph [H+] 1 100 mmol/l D = 90 mmol/l 2 10 mmol/l D = 9 mmol/l 3 1 mmol/l 2 ph = log [H + ] 3 ph ph = log [H + ] ph of capillary blood norm: 7,35 7,45 Sorensen
Acid base equilibrium ph concept ph = log [H + ] ph [H+] 1 100 mmol/l D = 90 mmol/l 2 10 mmol/l D = 9 mmol/l 3 1 mmol/l 2 ph = log [H + ] 3 ph ph = log [H + ] ph of capillary blood norm: 7,35 7,45 Sorensen
RENAL TUBULAR ACIDOSIS An Overview
 RENAL TUBULAR ACIDOSIS An Overview UNIVERSITY OF PNG SCHOOL OF MEDICINE AND HEALTH SCIENCES DISCIPLINE OF BIOCHEMISTRY & MOLECULAR BIOLOGY CLINICAL BIOCHEMISTRY PBL MBBS IV VJ. Temple 1 What is Renal Tubular
RENAL TUBULAR ACIDOSIS An Overview UNIVERSITY OF PNG SCHOOL OF MEDICINE AND HEALTH SCIENCES DISCIPLINE OF BIOCHEMISTRY & MOLECULAR BIOLOGY CLINICAL BIOCHEMISTRY PBL MBBS IV VJ. Temple 1 What is Renal Tubular
The average potassium content during the last 5. solids. This average decrease of 2.2 meq. per 100. initial potassium content of the arteries.
 THE EFFECT OF NOR-EPINEPHRINE ON THE ELECTROLYTE COMPOSITION OF ARTERIAL SMOOTH MUSCLE' By LOUIS TOBIAN 2 AND ADACIE FOX (From the Departments of Pharmacology and Internal Medicine, Southwesters Medical
THE EFFECT OF NOR-EPINEPHRINE ON THE ELECTROLYTE COMPOSITION OF ARTERIAL SMOOTH MUSCLE' By LOUIS TOBIAN 2 AND ADACIE FOX (From the Departments of Pharmacology and Internal Medicine, Southwesters Medical
CASE 10. What would the ST segment of this ECG look like? On which leads would you see this ST segment change? What does the T wave represent?
 CASE 10 A 57-year-old man presents to the emergency center with complaints of chest pain with radiation to the left arm and jaw. He reports feeling anxious, diaphoretic, and short of breath. His past history
CASE 10 A 57-year-old man presents to the emergency center with complaints of chest pain with radiation to the left arm and jaw. He reports feeling anxious, diaphoretic, and short of breath. His past history
Potassium regulation. -Kidney is a major regulator for potassium Homeostasis.
 Potassium regulation. -Kidney is a major regulator for potassium Homeostasis. Normal potassium intake, distribution, and output from the body. Effects of severe hyperkalemia Partial depolarization of cell
Potassium regulation. -Kidney is a major regulator for potassium Homeostasis. Normal potassium intake, distribution, and output from the body. Effects of severe hyperkalemia Partial depolarization of cell
Interpreting Electrocardiograms (ECG) Physiology Name: Per:
 Interpreting Electrocardiograms (ECG) Physiology Name: Per: Introduction The heart has its own system in place to create nerve impulses and does not actually require the brain to make it beat. This electrical
Interpreting Electrocardiograms (ECG) Physiology Name: Per: Introduction The heart has its own system in place to create nerve impulses and does not actually require the brain to make it beat. This electrical
Fluid and Electrolytes P A R T 4
 Fluid and Electrolytes P A R T 4 Mechanisms that control acid-base homeostasis Acids and bases continually enter and leave body Hydrogen ions also result from metabolic activity Acids Hydrogen ion donors
Fluid and Electrolytes P A R T 4 Mechanisms that control acid-base homeostasis Acids and bases continually enter and leave body Hydrogen ions also result from metabolic activity Acids Hydrogen ion donors
IT has become common observation that
 Electrolytes and ph Changes in Relation to Hypothermic Ventricular Fibrillation By BENJAMIN G. COVINO AND A. H. HEGNAUER Coronary aiteriovenous electrolyte differences were studied in normo- and hypercnpneic
Electrolytes and ph Changes in Relation to Hypothermic Ventricular Fibrillation By BENJAMIN G. COVINO AND A. H. HEGNAUER Coronary aiteriovenous electrolyte differences were studied in normo- and hypercnpneic
CRC 431 ECG Basics. Bill Pruitt, MBA, RRT, CPFT, AE-C
 CRC 431 ECG Basics Bill Pruitt, MBA, RRT, CPFT, AE-C Resources White s 5 th ed. Ch 6 Electrocardiography Einthoven s Triangle Chest leads and limb leads Egan s 10 th ed. Ch 17 Interpreting the Electrocardiogram
CRC 431 ECG Basics Bill Pruitt, MBA, RRT, CPFT, AE-C Resources White s 5 th ed. Ch 6 Electrocardiography Einthoven s Triangle Chest leads and limb leads Egan s 10 th ed. Ch 17 Interpreting the Electrocardiogram
Lab #3: Electrocardiogram (ECG / EKG)
 Lab #3: Electrocardiogram (ECG / EKG) An introduction to the recording and analysis of cardiac activity Introduction The beating of the heart is triggered by an electrical signal from the pacemaker. The
Lab #3: Electrocardiogram (ECG / EKG) An introduction to the recording and analysis of cardiac activity Introduction The beating of the heart is triggered by an electrical signal from the pacemaker. The
SAMPLE CHAPTER CHAPTER
 2.1 Introduction CHAPTER 2 Cardiovascular Structure and Function One may question why it is important for a biomedical engineer to study physiology. To answer this question, we can begin by recognizing
2.1 Introduction CHAPTER 2 Cardiovascular Structure and Function One may question why it is important for a biomedical engineer to study physiology. To answer this question, we can begin by recognizing
Singer and Hastings (7) have indicated that the. data of Shock and Hastings (8) gives an apparent
 INTRACELLULAR CATION EXCHANGES IN ACIDOSIS DUE TO RENAL INSUFFICIENCY. EFFECTS OF ALKALI THERAPY1 By J. R. ELKINTON,2 R. D. SQUIRES,3 AND R. B. SINGER (From the Chemical Section of the Department of Medicine,
INTRACELLULAR CATION EXCHANGES IN ACIDOSIS DUE TO RENAL INSUFFICIENCY. EFFECTS OF ALKALI THERAPY1 By J. R. ELKINTON,2 R. D. SQUIRES,3 AND R. B. SINGER (From the Chemical Section of the Department of Medicine,
Physical Fitness Biology Concepts of Biology 13.1
 Physical Fitness Biology 100 - Concepts of Biology 13.1 Name Instructor Lab Section Objectives: To gain an understanding of: What is meant by the term physical fitness Various tests used to measure an
Physical Fitness Biology 100 - Concepts of Biology 13.1 Name Instructor Lab Section Objectives: To gain an understanding of: What is meant by the term physical fitness Various tests used to measure an
-SQA-SCOTTISH QUALIFICATIONS AUTHORITY. Hanover House 24 Douglas Street GLASGOW G2 7NQ NATIONAL CERTIFICATE MODULE DESCRIPTOR
 -SQA-SCOTTISH QUALIFICATIONS AUTHORITY Hanover House 24 Douglas Street GLASGOW G2 7NQ NATIONAL CERTIFICATE MODULE DESCRIPTOR -Module Number- 0099111 -Session-1989-90 -Superclass- PB -Title- PRINCIPLES
-SQA-SCOTTISH QUALIFICATIONS AUTHORITY Hanover House 24 Douglas Street GLASGOW G2 7NQ NATIONAL CERTIFICATE MODULE DESCRIPTOR -Module Number- 0099111 -Session-1989-90 -Superclass- PB -Title- PRINCIPLES
CRITICAL THINKING QUESTIONS AND ANSWERS AND CYCLE 2 LAB EXAM TEMPLATE. There are two main mechanisms that work in conjunction to return the blood
 CRITICAL THINKING QUESTIONS AND ANSWERS AND CYCLE 2 LAB EXAM TEMPLATE There are two main mechanisms that work in conjunction to return the blood THE CARDIAC PUMP 1) The forward pull(vis a fronte) This
CRITICAL THINKING QUESTIONS AND ANSWERS AND CYCLE 2 LAB EXAM TEMPLATE There are two main mechanisms that work in conjunction to return the blood THE CARDIAC PUMP 1) The forward pull(vis a fronte) This
Calcium (Ca 2+ ) mg/dl
 Quick Guide to Laboratory Values Use this handy cheat-sheet to help you monitor laboratory values related to fluid and electrolyte status. Remember, normal values may vary according to techniques used
Quick Guide to Laboratory Values Use this handy cheat-sheet to help you monitor laboratory values related to fluid and electrolyte status. Remember, normal values may vary according to techniques used
SUMMARY OF PRODUCT CHARACTERISTICS. Potassium Chloride 0.15% w/v & Sodium Chloride 0.9% w/v Solution for Infusion
 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Potassium Chloride 0.15% w/v & Sodium Chloride 0.9% w/v Solution for Infusion 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Potassium Chloride
SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Potassium Chloride 0.15% w/v & Sodium Chloride 0.9% w/v Solution for Infusion 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Potassium Chloride
Acid & Alkaline Nutrition: Shattering the Myths By Michael McEvoy, FDN, CNC, CMTA
 Acid & Alkaline Nutrition: Shattering the Myths By Michael McEvoy, FDN, CNC, CMTA According to Guy Schenker, DC: Acid/alkaline imbalances always involve respiratory function Acid/alkaline imbalances always
Acid & Alkaline Nutrition: Shattering the Myths By Michael McEvoy, FDN, CNC, CMTA According to Guy Schenker, DC: Acid/alkaline imbalances always involve respiratory function Acid/alkaline imbalances always
ELECTROLYTES RENAL SHO TEACHING
 ELECTROLYTES RENAL SHO TEACHING Metabolic Alkalosis 2 factors are responsible for generation and maintenance of metabolic alkalosis this includes a process that raises serum bicarbonate and a process that
ELECTROLYTES RENAL SHO TEACHING Metabolic Alkalosis 2 factors are responsible for generation and maintenance of metabolic alkalosis this includes a process that raises serum bicarbonate and a process that
Systolic and Diastolic Currents of Injury
 Systolic and Diastolic Currents of Injury Figure 1 Action Potentials of Normal and Ischemic Tissue In Figure 1 above, the action potential of normal myocardium is represented by the solid lines and the
Systolic and Diastolic Currents of Injury Figure 1 Action Potentials of Normal and Ischemic Tissue In Figure 1 above, the action potential of normal myocardium is represented by the solid lines and the
PACKAGE LEAFLET: INFORMATION FOR THE USER. CALCICLO STEROP 11 meq 10 ml Solution for injection. Dihydrated Calcium Chloride
 PACKAGE LEAFLET: INFORMATION FOR THE USER CALCICLO STEROP 11 meq 10 ml Solution for injection Dihydrated Calcium Chloride Read all of this leaflet carefully before you start using this medicine because
PACKAGE LEAFLET: INFORMATION FOR THE USER CALCICLO STEROP 11 meq 10 ml Solution for injection Dihydrated Calcium Chloride Read all of this leaflet carefully before you start using this medicine because
Introduction. Circulation
 Introduction Circulation 1- Systemic (general) circulation 2- Pulmonary circulation carries oxygenated blood to all parts of the body carries deoxygenated blood to the lungs From Lt. ventricle aorta From
Introduction Circulation 1- Systemic (general) circulation 2- Pulmonary circulation carries oxygenated blood to all parts of the body carries deoxygenated blood to the lungs From Lt. ventricle aorta From
Arterial blood gas analysis
 perioperativecpd.com continuing professional development Arterial blood gas analysis Article based on original by the Resuscitation Council U.K. Introduction Interpreting the analysis of an arterial blood
perioperativecpd.com continuing professional development Arterial blood gas analysis Article based on original by the Resuscitation Council U.K. Introduction Interpreting the analysis of an arterial blood
Collin County Community College
 Collin County Community College BIOL. 2402 Anatomy & Physiology WEEK 5 The Heart 1 The Heart Beat and the EKG 2 1 The Heart Beat and the EKG P-wave = Atrial depolarization QRS-wave = Ventricular depolarization
Collin County Community College BIOL. 2402 Anatomy & Physiology WEEK 5 The Heart 1 The Heart Beat and the EKG 2 1 The Heart Beat and the EKG P-wave = Atrial depolarization QRS-wave = Ventricular depolarization
