Effects of [Na + ], [Cl ], carbonic anhydrase, and intracellular ph on corneal endothelial bicarbonate transport
|
|
|
- Cynthia Powell
- 5 years ago
- Views:
Transcription
1 Effects of [Na + ], [Cl ], carbonic anhydrase, and intracellular ph on corneal endothelial bicarbonate transport Keith Green, Stuart Simon, Gordon M. Kelly, Jr., and Karen A. Bowman Sodium removal from the solution bathing the isolated rabbit corneal endothelium caused a reduction in both unidirectional and net flux of bicarbonate, whereas chloride withdrawal from the solution had no effect on the net bicarbonate flux but increased the unidirectional fluxes. These data correlate with previously published data on the effects of similar solution manipulations on both fluid movement and potential difference across the endothelium and strongly implicate bicarbonate as the primary ion involved in the maintenance of corneal hydration. Carbonic anhydrase (1 mg/ml) added to the solution bathing both sides of the endothelium markedly increased unidirectional and net bicarbonate fluxes, possibly by maintaining a high bicarbonate /CO 2 concentration close to the membrane and thereby eliminating chemical gradients in the unstirred layer adjacent to the membrane. Determinations of intracellular ph with the 5,5-dimethyl-2,-oxazolidine-dione method indicate that at more acid ambient conditions there is a lesser gradient between cell and bathing medium for H + ; similar ambient conditions in other experiments resulted in larger unidirectional bicarbonate fluxes than at neutral ph. The data are suggestive of a nonvectorial H + -HCO 3 ~ exchange occurring across the endothelial cellular membranes. (INVEST OPHTHALMOL VIS SCI 21: , 1981.) Key words: rabbit, cornea, endothelium, bicarbonate transport, unidirectional fluxes, ionic dependence, intracellular ph, carbonic anhydrase regulation of rabbit cornea hydration by the endothelium has been demonstrated to be both bicarbonate- and sodium-sensitive 1 " From the Departments of Ophthalmology (K. G., S.S., G.M.K., K.A.B.) and Physiology (K. G.), Medical College of Georgia, Augusta. Supported in part by Public: Health Research Grant EY 0113 from the National Eye Institute, by the J. B. Hall Foundation, and by a Research to Prevent Blindness, Inc., grant. Stuart Simon was the recipient of an American Medical Association Education and Research Foundation Studentship during the course of this work. Keith Green was the recipient of a Research Manpower Award from Research to Prevent Blindness, Inc. Submitted for publication Nov. 17, Reprint requests: Keith Green, Department of Ophthalmology, Medical College of Georgia, Augusta, Ga and to show no dependence on chloride. 3 The measurement of a net bicarbonate movement across the endothelium " 6 has provided impetus to the concept that a bicarbonate transport system is directly linked to fluid movement across the endothelium. 7 Certainly the link between fluid movement, thickness control, and potential difference across the endothelium and the ionic content of the bathing medium appears strong, 3 but a similar dependence between bicarbonate transport and the composition of the bathing medium has not been established. Such a relationship would support the theory of a direct link between endothelial fluid and bicarbonate transport. The endothelial net bicarbonate flux has been shown to be dependent on the bicarbon /81/ $00.60/ Assoc. for Res. in Vis. and Ophthal., Inc.
2 Volume 21 Number Corneal endothelial bicarbonate fluxes 587 ate concentration in the bathing medium, 5 ' 6 yet other studies 3 " 5 have shown that carbonic anhydrase inhibitors influence both corneal thickness 3 ' and net bicarbonate fluxes. 5 These two observations appear to be ambiguous but must be reconciled. Research using artificial bilayer lipid membranes 8 has shown that the unstirred layers immediately adjacent to either side of the membrane are ratelimiting to the movement of CO 2 - In the presence of carbonic anhydrase, which accelerates the hydration-dehydration of CO 2, the concentration of CO 2 in the unstirred layers was increased so that the membrane itself became rate-limiting. The participation of carbonic anhydrase in the process of endothelial net bicarbonate movement required further study to determine whether a similar process occurred in the endothelium as in the artificial membrane. To determine the dependence of corneal endothelial bicarbonate movement on other ions, bicarbonate fluxes were measured after changes of [Na + ] and [Cl~] in the bathing medium as well as in the presence of carbonic anhydrase. Measurements were made of endothelial intracellular ph under various ambient conditions to determine whether proton movement might be related to bicarbonate transport. Materials and methods Adult albino rabbits weighing 2-3 kg were used in three groups of procedures. Endothelial bicarbonate fluxes and ionic changes (group I). The methods and procedures were as described previously. 0 " 6 Briefly, corneas were de-epithelialized and were mounted on specular microscope mounting rods after removal from eyes. The mounted tissue was then placed in water-jacketed Lucite chambers with a volume of about 1.2 ml each. Paired corneas were used, with one unidirectional flux measured on one of each pair (stromal-to-aqueous [JSncio] on or >e cornea and aqueous-to-stromal [Jepf 0 ] on the other). Both corneal surfaces were bathed with Krebs-bicarbonate Ringers solution 9 with added adenosine (0.3 gm/l) and glutathione (0.09 gm/l). 2 The endothelial surface of one cornea of each pair and the stromal surface of the other cornea of the pair were equilibrated with Ringers solution containing H 1 CO 3 on one side for 1 hr prior to sampling and the cold solution on the opposite endothelial surface; the latter was thereafter replaced at 30 min intervals. 0 ' 6 The solutions were sampled and counted in a Searle Isocap 300 with the Nalgene film ware scintillation system. The bathing solutions contained 13 mm NaCl, 50. mm NaCl, 10 mm NaCl, oromm NaCl. Substitution of NaCl was made with choline chloride, and in Na-free and 10 mm NaCl solutions KHCO 3 was substituted for NaHCO 3 ; thus [Cl~] was maintained constant and only [Na + ] changed. For chloride changes, solutions of 50 and 0 mm NaCl were used; substitution of Cl~ was made with Na 2 SO and sucrose. All solutions were isosmotic at 305 mosm, as determined by measurement in a Fiske osmometer, were equilibrated with 95% O 2 :5% CO 2, and were at ph 7.3. The ph was adjusted by adding either NaOH or HC1 as needed; usually only minor adjustments were needed. Carbonic anhydrase effects (group II). Endothelia were allowed to equilibrate with H 1 CO 3 for 1 hr with 1 mg/ml carbonic anhydrase in the bathing solution on both sides, and the appropriate solutions were sampled after replacement every 30 min after equilibration. The solutions used contained 25 mm bicarbonate at either ph 8 or 7.3. Intracellular ph (group III). The 5,5-dimethyl- 2,-oxazolidine-dione (DMO) method of Miller et al. 10 was used with 1 C-labeled material. Solutions were made containing, nominally, 25 or 60 mm bicarbonate. 6 Solution variations, all at ph 7.5, included the presence or absence of 1 mg/ml carbonic anhydrase; the addition of 1 mm acetazolamide (carbonic anhydrase inhibitor), 10" 6 M ouabain, 10~ M furosemide, or 10~ M amiloride; or a sodium-free solution. The effect of de-epithelialization was tested, as was the stability of the intracellular ph with time. Six or eight whole corneas that had been quickly removed from rabbits were placed into a scintillation vial containing Ringer's solution and 1 C-DMO (2 fxcuml). The 25 mm bicarbonate solutions were at ph 7, 7.5, and 8 without additional drugs and at ph 7.5 when containing drugs; the 60 mm bicarbonate solution was at ph 7.5. The vials were sealed without air space in order to maintain the fluid composition constant, and the corneas were incubated for 1 hr at 37 C, except for one set of incubations for 3 hr. At this time the corneas were removed and rinsed in nonlabeled Ringer's solution of the same composition as that in which the cornea was bathed. The endothelium was lightly blotted with filter paper and was scraped off before placement in a tared homogenization tube. Three or four endothelia
3 588 Green et al. Invest. Ophthalmol. Vis. Sci. October 1981 Table I. Unidirectional and net bicarbonate fluxes across rabbit corneal endothelium as a function of solution [Na + ] and [Cl~] Group I: A 13 meq Na (control) 50 meq Na 10 meq Na 0 meq Na 50 meq Cl" 0 meq Cl~ Group II: A ph 7.32 ± 0.05 Pco ±.7 mm Hg HCO ± 1. mm Group III: B ph 8.08 ± 0.07 PCO ± 1.3 mm Hg HCO ± 0.8 mm 7 ep L./endo 3.2 ± ± ± ± 0.02 D 3.67 ± ± ± ± 0.20 Jendo /epl 1.57 ± ± ± 0.07 D 2.30 ± 0.0 DE 2.15 ± ± ± ± 0.29 repl _ rendoc Jendo Jepl 1.67 ± ± 0.15 D 0.79 ± ± 0.17 DE 1.50 ± ± ± 0.33 D 2.95 ± 0.25 D A All unidirectional flux values are /xeq/cmvhr and represent the mean ± S.E.M. of 36 measurements (no less than four per cornea). B Values are the mean ± S.E.M. of at least six experiments (each with at least four determinations per cornea) and the ph, Pco 2 and HCO3 values are those of the solutions at the termination of the incubation. c Values are the mean ± S.E.M. of the arithmetic difference between the unidirectional fluxes of each corneal pair at each time interval (n = 36). "Significantly different from control values (p <0.02). E Statistically different from 10 meq Na + values (p <0.05). were pooled for each wet weight determination, and after weighing, 100 /JL\ of distilled water was added to each tube and the tissues were homogenized. Duplicate 20 fi\ samples were taken, and 10 ml of Aquasol was added prior to counting in a Searle Isocap 300 scintillation counter. Results Group I. The unidirectional and net bicarbonate fluxes across the rabbit corneal endothelium as a function of solution [Na + ] and [Cl~] are given in Table I. Reduction of [Na + ] caused a fall in Jf d0 (active) and an increase in Jep? (passive), with a combined effect to reduce JlSdo net. Reduction from 13 meq [Na + ] to 50 meq [Na + ] caused a large proportion of the total change induced by Na + -free solution. Jendo is not significantly different between 50, 10, or 0 meq [Na + ] solutions. Although 50 and 10 meq [Na + ] solutions resulted in statistically similar values of Jfpf 10, reduction to an Na + -free solution caused a further statistically different increase in the passive flux (Jlpf 0 ) and a consequent decrease in the net flux. Changes in [Cl~] appeared to have no detrimental effect on JfKdo net - Both unidirectional fluxes were significantly increased, albeit the change induced by 50 meq [Cl ] solution was not further altered by Cl~-free solution. Group II. The addition of 1 mg/ml carbonic anhydrase to both sides of the endothelium approximately doubled the net and unidirectional fluxes of H 1 CO 3. The unidirectional fluxes were significantly greater at ph 7.3 than at ph 8, which corresponds to the data found previously in the absence of carbonic anhydrase. 6 There was a trend in both passive and active fluxes at both ph values to decrease as time progressed; for example, at ph 8 the successive 30 min measurements of Jfndo were 8.52 ± 0.27, 7.0 ± 0.22, 6.73 ± 0.17, and 6.12 ± 0.17, and for ]%$ they were 5.03 ± 0.29,.23 ± 0.32, 3.92 ± 0.29, and 3.77 ± 0.3 (n = 6). Group III. The intracellular ph was calculated according to equations given previously by Irvine et al., 11 in which tissue hydration and the ratio of intracellular to extracellular water are included. Tissue hydration was taken as 70% and extracellular water as 10% of total tissue water for all cases. Increased tissue hydration would reduce the calculated intracellular ph by about 0.03 to 0.0 ph
4 Volume 21 Number Corneal endothelial bicarbonate fluxes 589 Table II. Corneal endothelial intracellular ph as a function of ambient conditions Solution 25 mm bicarbonate, ph 7, 1 hr 25 mm bicarbonate, ph 7.5, 1 hr 25 mm bicarbonate, ph 7.5, 3 hr 25 mm bicarbonate, ph 7.5, de-epithelialized, 1 hr 25 mm bicarbonate, ph 7.5, 1 mg/ml carbonic anhydrase, 1 hr 25 mm bicarbonate, ph 7.5, 1 mm acetazolamide, 1 hr 25 mm bicarbonate, ph 7.5, Na + -free, 1 hr 25 mm bicarbonate, ph 7.5, 10~ 6 M ouabain, 1 hr 25 mm bicarbonate, ph 7.5, 10~ M amiloride, 1 hr 25 mm bicarbonate, ph 7.5, 10~ M furosemide, 1 hr 25 mm bicarbonate, ph mm bicarbonate, ph 7.5 Intracellular ph A 6.85 ± 0.09 c 7.20 ± ± ± ± 0.0 c 7.36 ± 0.0 c 7.32 ± ± , , ± 0.0 c 7.36 ± 0.03 c No. B 2 2 A Values are the mean ± S.E.M. B Number of experimental determinations, each consisting of either three or four pooled endothelia (duplicate samples were taken of each pooled set of endothelia, and a mean was obtained and counted as one experimental determination). Where less than four determinations were made, the average of duplicate analyses are given. c Statistically different from 25 mm bicarbonate, ph 7.5, 1 hr. units per 5% increase in hydration; an increase in extracellular water to 20% would reduce the calculated intracellular ph by 0.01 ph unit, and a decrease to 5% extracellular water would increase intracellular ph by 0.01 ph unit. The choice of extracellular water value is therefore of no consequence in the calculation. Only if tissue hydration was significantly changed would an effect be seen on intracellular ph. The primary determinant of the distribution of DMO therefore is intracellular and extracellular ph. The values obtained for intracellular ph are shown in Table II. The intracellular ph was close to that of the ambient solution at low ph (7.0), and the difference between extracellular and intracellular ph was greater as the external ph increased. The difference between intracellular and extracellular ph (ApH) appears to become constant above ambient ph of 7.5. Incubation for 1 or 3 hr under the same ambient conditions gave the same intracellular ph, indicating that equilibrium was complete within 1 hr, and the de-epithelialized cornea data showed that the presence of the epithelium in the majority of determinations had no effect. At 60 mm bicarbonate, ph 7.5, the intracellular ph was closer to that of the bathing solution than that found at the same ph and 25 mm bicarbonate. The intracellular ph increased with sodium removal from the bathing solution, with a concurrent decrease in net bicarbonate flux (Table I), but a similar trend was observed with both carbonic anhydrase, which increased net and unidirectional bicarbonate fluxes (see Table I), and acetazolamide, which decreased net bicarbonate flux. 5 Ouabain, which inhibits (Na + + K + ) adenosine triphosphatase (ATPase), also increased intracellular ph. Amiloride tended to increase the intracellular ph, but furosemide did not cause such a large increase. Discussion Previous research has shown that both the rate of fluid movement across the endothelium (calculated from the rate of stromal deturgescence) and the electrical potential difference across the endothelium are dependent on the sodium content of the bathing medium and that replacement of chloride has no effect on these parameters. 3 Although bicarbonate transport has been implicated as a causative factor in transendothelial fluid movement, " 6 no studies thus far have shown that bicarbonate transport shows the same ionic dependence as fluid movement and potential difference. The results of this study indicate that there is a strong sodium dependence of both net and unidirectional bicarbonate movement and an absence of any chloride dependence of net bicarbonate transfer. Sodium removal from the bathing solution
5 590 Green et al. Invest. Ophthabnol. Vis. Sci. October 1981 not only decreased J ^do but also increased Jepi d0 > implying that the passive properties of the membrane might also be affected by this maneuver. It appears that the effect of sodium reduction is almost fully realized by reduction to at least 50 meq Na +, since the only other significant change in fluxes between 50 and 0 meq Na + was an increase in the passive Jlpf 0 flux. Chloride removal from the bathing solution, on the other hand, had no effect on the net bicarbonate flux but caused a significant increase in both unidirectional fluxes. Perhaps the latter is indicative of either some regulatory role of Cl~ on bicarbonate, such as an exchange mechanism where, in the absence of Cl~, another anion can substitute, or a general effect on endothelial cell membrane permeability. The net flux is unaltered, however, and this correlates with the lack of effect on both potential difference and fluid movement. 3 Both experiments with sodium and chloride changes of the bathing media support the hypothesis that bicarbonate is directly related to potential difference and fluid movement across the rabbit corneal endothelium. The correlation is striking, although with the sodium data the majority of response appeared to be caused by a change from 13 to 50 meq Na, whereas the potential difference showed a more gradual reduction as sodium concentration was decreased. 3 The addition of carbonic anhydrase to the endothelial bathing solutions caused a large increase in net and unidirectional bicarbonate fluxes. The unidirectional fluxes were greatly increased, by between two and three times, over control values and still showed the same ph dependence as noted previously under normal conditions, 6 i.e., greater unidirectional fluxes at lower ambient ph. The addition of carbonic anhydrase did not change the ph, PCO 2, or PO 2 of the bathing solution despite the marked increase in the unidirectional fluxes in the initial 30 min after drug application. Carbonic anhydrase could be maintaining a high concentration of HCO^" or CO 2 in the unstirred layer adjacent to the membrane, thereby providing substrate for the transport system, which normally would remove HCO^" or CO 2 from the unstirred layer and create a gradient from the membrane face to the bulk solution. Since carbonic anhydrase enhances the hydrationdehydration of CO 2, such a gradient could be minimized by the generation of HCO 3 ~ or CO 2 in the unstirred layer as fast as it was removed by the transport system. The latter could well explain the marked elevation of unidirectional fluxes and of the net flux, especially since the stroma is a large unstirred fluid volume. Support for this concept is obtained from the slight increase in intracellular ph after carbonic anhydrase addition to the ambient solution (Table II). These findings with carbonic anyhdrase may also serve to explain how carbonic anhydrase inhibitors affect net movement of radiolabeled bicarbonate across the endothelium, 5 since local inhibition of the cellular enzyme would lead to an inability to maintain a high bicarbonate or CO 2 concentration at the membrane surface for either exchange or transport function. That there is inhibition of measured bicarbonate fluxes by carbonic anhydrase inhibitors 5 and acceleration of fluxes by extracellular carbonic anhydrase supports the concept that both bicarbonate and carbon dioxide are involved in the net transfer of bicarbonate. The relationship between intracellular and extracellular ph is that the intracellular ph is approximately equal to that of the ambient solution when the latter is at ph 7, whereas, with the external ph approaching 8, the intracellular ph does not increase equivalently. Above ph 7.5, however, the difference between intracellular and extracellular ph appears to become constant at 0.3 ph units. The results of a previous study showed that the unidirectional bicarbonatefluxesat ph 7, 25 mm bicarbonate, are greater than those at ph 8. 6 As ph increases in the external medium the [H + ] gradient would tend to assist H + movement from the cell, which might decrease the fluxes of bicarbonate if there were not preferential directional movement of H +. The measured values of intracellular ph at
6 Volume 21 Number Cornea! endothelial bicarbonate fluxes 591 various external ph values, when related to previous flux data, 6 would appear to suggest that a proton-bicarbonate exchange mechanism plays some role in bicarbonate transfer across the corneal endothelium or in exchange at one or both cell surfaces. Nevertheless, the primary determinant of intracellular ph appears to be the ph of the environment. The data at 60 mm bicarbonate, where the unidirectional fluxes are greater, 6 indicate that the intracellular ph more closely resembles that of the extracellular fluid, a situation that again (in combination with the elevated bicarbonate concentration) would provide a more suitable environment for enhanced bicarbonate transfer. Data obtained with various perturbations of the endothelial bathing media indicate that removal of sodium causes an increase of intracellular ph (Table II) concurrent with a fall in net bicarbonate flux (Table I). Both carbonic anhydrase, which accelerates both unidirectional and net bicarbonate fluxes, and acetazolamide, which reduces net bicarbonate flux across the endothelium, 5 increase intracellular ph. Ouabain, which inhibits (Na + + K + )-dependent ATPase 12 ' 13 and causes corneal swelling without alterations of passive endothelial permeability, 1 increased intracellular ph, which may relate to some alteration of bicarbonate fluxes. Amiloride tended to increase intracellular ph compared to controls, which may imply an involvement of sodium in intracellular H + regulation. Furosemide, on the other hand, had little effect, implying that Cl~-H + exchange plays no role in regulation of intracellular ph or bicarbonate fluxes. We thank Mrs. Ilene Prior for her valuable secretarial assistance. REFERENCES 1. Hodson S: Evidence for a bicarbonate dependent sodium pump in corneal endothelium. Exp Eye Res 11:20, Dikstein S and Maurice DM: The metabolic basis to the fluid pump in the cornea. J Physiol (Lond) 221:29, Fischbarg J and Lim JJ: Role of cations, anions and carbonic anhydrase in fluid transport across rabbit endothelium. J Physiol (Lond) 21:67, Hodson S and Miller F: The bicarbonate pump in the endothelium which regulates the hydration of rabbit cornea. J Physiol (Lond) 263:563, Hull DS, Green K, Boyd M, and Wynn HR: Corneal endothelium bicarbonate transport and the effect of carbonic anhydrase inhibitors on endothelial permeability, fluxes and thickness. INVEST OPH- THALMOL Vis SCI 16:883, Kelly G and Green K: Influence of bicarbonate and CO 2 on rabbit corneal transendothelial bicarbonate fluxes. Exp Eye Res 30:61, Mayes KR and Hodson S: Local osmotic coupling to the active transendothelial bicarbonate flux in the rabbit cornea. Biochim Biophys Acta 51:286, Gutnecht J, Bisson MA, and Tosteson DC: Diffusion of carbon dioxide through lipid bilayer membranes. J Gen Physiol 69:779, Green K and Green MA: Permeability to water of rabbit corneal membranes. Am J Physiol 217:635, Miller RB, Tyson I, and Relman AS: ph of isolated resting skeletal muscle and its relation to potassium content. Am J Physiol 20:108, Irvine ROH, Saunders SJ, Milne MD, and Crawford MA: Gradients of potassium and hydrogen ion in potassium-deficient voluntary muscle. Clin Sci 20:1, Rogers KT: Levels of (Na + + Reactivated and Mg 2+ -activated ATPase activity in bovine and feline corneal endothelium and epithelium. Biochim Biophys Acta 163:50, Riley MV: Anion-sensitive ATPase in rabbit corneal endothelium and its relation to corneal hydration. Exp Eye Res 25:83, Trenberth S and Mishima S: The effect of ouabain on the rabbit corneal endothelium. INVEST OPHTHAL- MOL 7:, 1968.
Cornea Endofhelial Rose Bengal Photosensitizafion
 Cornea Endofhelial Rose Bengal Photosensitizafion Effect on Permeability, Sodium Flux, and Ultrastructure David S. Hull,* Keirh Green,*f and Lisa Laughter* Rabbit corneal endothelial cells demonstrated
Cornea Endofhelial Rose Bengal Photosensitizafion Effect on Permeability, Sodium Flux, and Ultrastructure David S. Hull,* Keirh Green,*f and Lisa Laughter* Rabbit corneal endothelial cells demonstrated
REFERENCES. Reports 1117
 Number 11 Reports 1117 lation. The variations were typical of the average specimen. In order to assess survival of cell membrane potential following dissection, two corneas were used for microelectrode
Number 11 Reports 1117 lation. The variations were typical of the average specimen. In order to assess survival of cell membrane potential following dissection, two corneas were used for microelectrode
NaCl is the same as that to sucrose, raffinose and stachyose, i.e. about 1.
 J. Physiol. (1987), 385, pp. 97-106 97 With 4 text-figures Printed in Great Britain - THE APPARENT REFLEXION COEFFICIENT OF THE LEAKY CORNEAL ENDOTHELIUM TO SODIUM CHLORIDE IS ABOUT ONE IN THE RABBIT BY
J. Physiol. (1987), 385, pp. 97-106 97 With 4 text-figures Printed in Great Britain - THE APPARENT REFLEXION COEFFICIENT OF THE LEAKY CORNEAL ENDOTHELIUM TO SODIUM CHLORIDE IS ABOUT ONE IN THE RABBIT BY
Biochemical evaluation of the corneal endothelium
 Isolation of the plasma membrane from corneal endothelial cells Z. Suzanne Zam, James Cerda,* and Frank M. Polack The plasma membranes of normal rabbit endothelail cells were isolated by the use of an
Isolation of the plasma membrane from corneal endothelial cells Z. Suzanne Zam, James Cerda,* and Frank M. Polack The plasma membranes of normal rabbit endothelail cells were isolated by the use of an
Effects of Sodium Lactate on Isolated Rabbit Corneas
 Investigative Ophthalmology & Visual Science, Vol. 31, No. 5, May 1990 Copyright Association for Research in Vision and Ophthalmology Effects of Sodium Lactate on Isolated Rabbit Corneas Joseph W. Huff
Investigative Ophthalmology & Visual Science, Vol. 31, No. 5, May 1990 Copyright Association for Research in Vision and Ophthalmology Effects of Sodium Lactate on Isolated Rabbit Corneas Joseph W. Huff
S,Ipecular microscopy has added a new
 Functional and structural changes in the corneal endothelium during in vitro perfusion Bernard E. McCarey,* Henry F. Edelhauser, and Diane L. Van Horn The endothelium of isolated rabbit corneas was perfused
Functional and structural changes in the corneal endothelium during in vitro perfusion Bernard E. McCarey,* Henry F. Edelhauser, and Diane L. Van Horn The endothelium of isolated rabbit corneas was perfused
The effect of ouabain on the rabbit corneal endothelium. Sterling M. Trenberth* and Saiichi Mishima
 The effect of ouabain on the rabbit corneal endothelium Sterling M. Trenberth* and Saiichi Mishima With the method of in vitro incubation of the isolated rabbit cornea with the modified KEI medium, the
The effect of ouabain on the rabbit corneal endothelium Sterling M. Trenberth* and Saiichi Mishima With the method of in vitro incubation of the isolated rabbit cornea with the modified KEI medium, the
Relationship Between Fluid Transport and In Situ Inhibition of Na + -K + Adenosine Triphosphatase in Corneal Endothelium
 Relationship Between Fluid Transport and In Situ Inhibition of Na + -K + Adenosine Triphosphatase in Corneal Endothelium Michael V. Riley, Barry S. Winkler, Margaret I. Peters, and Catherine A. Czajkowski
Relationship Between Fluid Transport and In Situ Inhibition of Na + -K + Adenosine Triphosphatase in Corneal Endothelium Michael V. Riley, Barry S. Winkler, Margaret I. Peters, and Catherine A. Czajkowski
A,kCetazolamide lowers intraocular pressure
 Ocular and systemic effects of acetazolamide in nephrectomized rabbits Zvi Friedman,* Theodore Krupin, and Bernard Becker The effects of acetazolamide on intraocular pressure (IOP) were studied on rabbits
Ocular and systemic effects of acetazolamide in nephrectomized rabbits Zvi Friedman,* Theodore Krupin, and Bernard Becker The effects of acetazolamide on intraocular pressure (IOP) were studied on rabbits
Hydrogen Peroxide-Mediated Corneol Endothelial Damage
 Hydrogen Peroxide-Mediated Corneol Endothelial Damage Induction by Oxygen Free Radical Dovid S. Hull,* Keith Green,*f Lisa Thomas,* and Nancy Alderman* Polymorphonuclear leukocytes and other inflammatory
Hydrogen Peroxide-Mediated Corneol Endothelial Damage Induction by Oxygen Free Radical Dovid S. Hull,* Keith Green,*f Lisa Thomas,* and Nancy Alderman* Polymorphonuclear leukocytes and other inflammatory
Quantitation of Na/K ATPase Pump Sites in the Rabbit Corneal Endothelium
 Quantitation of Na/K ATPase Pump Sites in the Rabbit Corneal Endothelium Dayle H. Geroski and Henry F. Edelhauser In these experiments, the binding of 3 H ouabain, a specific inhibitor of Na/K ATPase,
Quantitation of Na/K ATPase Pump Sites in the Rabbit Corneal Endothelium Dayle H. Geroski and Henry F. Edelhauser In these experiments, the binding of 3 H ouabain, a specific inhibitor of Na/K ATPase,
(corneal endothelium) separates an ion exchanger (corneal stroma) from. (Received 22 May 1973)
 J. Physiol. (1974), 236, pp. 271-32 271 With 15 text-figure8 Printed in Great Britain THE REGULATION OF CORNEAL HYDRATION BY A SALT PUMP REQUIRING THE PRESENCE OF SODIUM AND BICARBONATE IONS BY STUART
J. Physiol. (1974), 236, pp. 271-32 271 With 15 text-figure8 Printed in Great Britain THE REGULATION OF CORNEAL HYDRATION BY A SALT PUMP REQUIRING THE PRESENCE OF SODIUM AND BICARBONATE IONS BY STUART
Transport of Solutes and Water
 Transport of Solutes and Water Across cell membranes 1. Simple and Facilitated diffusion. 2. Active transport. 3. Osmosis. Simple diffusion Simple diffusion - the red particles are moving from an area
Transport of Solutes and Water Across cell membranes 1. Simple and Facilitated diffusion. 2. Active transport. 3. Osmosis. Simple diffusion Simple diffusion - the red particles are moving from an area
Transport across the cell membrane
 Transport across the cell membrane Learning objectives Body compartments ECF and ICF Constituents Lipid Bilayer: Barrier to water and water-soluble substances ions glucose H 2 O urea CO 2 O 2 N 2 halothane
Transport across the cell membrane Learning objectives Body compartments ECF and ICF Constituents Lipid Bilayer: Barrier to water and water-soluble substances ions glucose H 2 O urea CO 2 O 2 N 2 halothane
Effects of Amiloride on the Transport of Sodium and Other Ions in the Alga Hydrodictyon reticulatum
 Gen. Physiol. Biophys. (1987). 6, 255-263 255 Effects of Amiloride on the Transport of Sodium and Other Ions in the Alga Hydrodictyon reticulatum R. RYBOVÁ, R. METLIČKA and K. JANÁČEK Academy of Sciences.
Gen. Physiol. Biophys. (1987). 6, 255-263 255 Effects of Amiloride on the Transport of Sodium and Other Ions in the Alga Hydrodictyon reticulatum R. RYBOVÁ, R. METLIČKA and K. JANÁČEK Academy of Sciences.
April 1962 Volume 1, Number 2 INVESTIGATIVE OPHTHALMOLOGY. Symposium on the cornea. Introduction: Factors influencing corneal hydration
 April 1962 Volume 1, Number 2 INVESTIGATIVE OPHTHALMOLOGY Symposium on the cornea Introduction: Factors influencing corneal hydration John E. Harris The swelling of the corneal buttons in aqueous solution
April 1962 Volume 1, Number 2 INVESTIGATIVE OPHTHALMOLOGY Symposium on the cornea Introduction: Factors influencing corneal hydration John E. Harris The swelling of the corneal buttons in aqueous solution
I nadequate corneal epithelial nutrition
 Corneal amino acid supply and distribution Richard A. Thoft and Judith Friend The distribution of nonmetabolized carbon ^'labelled aaminoisobutyric acid (aaib) in the anterior segment of the rabbit eye
Corneal amino acid supply and distribution Richard A. Thoft and Judith Friend The distribution of nonmetabolized carbon ^'labelled aaminoisobutyric acid (aaib) in the anterior segment of the rabbit eye
Defect in Urinary Acidification Induced In Vitro by Amphotericin B
 Defect in Urinary Acidification Induced In Vitro by Amphotericin B PILIP R. STEINMETZ and Lois R. LAWSON From the Departments of Medicine, Harvard Medical School and the Beth Israel Hospital, Boston,-
Defect in Urinary Acidification Induced In Vitro by Amphotericin B PILIP R. STEINMETZ and Lois R. LAWSON From the Departments of Medicine, Harvard Medical School and the Beth Israel Hospital, Boston,-
Effect of Luminal Sodium Concentration
 Effect of Luminal Sodium Concentration on Bicarbonate Absorption in Rat Jejunum KENNETH A. HUBEL From the Department of Medicine, University of Iowa, Iowa City, Iowa 52242 A B S T R A C T An exchange of
Effect of Luminal Sodium Concentration on Bicarbonate Absorption in Rat Jejunum KENNETH A. HUBEL From the Department of Medicine, University of Iowa, Iowa City, Iowa 52242 A B S T R A C T An exchange of
Principles of Fluid Balance
 Principles of Fluid Balance I. The Cellular Environment: Fluids and Electrolytes A. Water 1. Total body water (TBW) = 60% of total body weight 2. Fluid Compartments in the Body a. Intracellular Compartment
Principles of Fluid Balance I. The Cellular Environment: Fluids and Electrolytes A. Water 1. Total body water (TBW) = 60% of total body weight 2. Fluid Compartments in the Body a. Intracellular Compartment
Ch 3 Membrane Transports
 Ch 3 Membrane Transports what's so dynamic about cell membranes? living things get nutrients and energy from the envrionment this is true of the entire organism and each cell this requires transport in/out
Ch 3 Membrane Transports what's so dynamic about cell membranes? living things get nutrients and energy from the envrionment this is true of the entire organism and each cell this requires transport in/out
Molecular Cell Biology. Prof. D. Karunagaran. Department of Biotechnology. Indian Institute of Technology Madras
 Molecular Cell Biology Prof. D. Karunagaran Department of Biotechnology Indian Institute of Technology Madras Module 4 Membrane Organization and Transport Across Membranes Lecture 1 Cell Membrane and Transport
Molecular Cell Biology Prof. D. Karunagaran Department of Biotechnology Indian Institute of Technology Madras Module 4 Membrane Organization and Transport Across Membranes Lecture 1 Cell Membrane and Transport
Chapter 4 Cell Membrane Transport
 Chapter 4 Cell Membrane Transport Plasma Membrane Review o Functions Separate ICF / ECF Allow exchange of materials between ICF / ECF such as obtaining O2 and nutrients and getting rid of waste products
Chapter 4 Cell Membrane Transport Plasma Membrane Review o Functions Separate ICF / ECF Allow exchange of materials between ICF / ECF such as obtaining O2 and nutrients and getting rid of waste products
Transport through membranes
 Transport through membranes Membrane transport refers to solute and solvent transfer across both cell membranes, epithelial and capillary membranes. Biological membranes are composed of phospholipids stabilised
Transport through membranes Membrane transport refers to solute and solvent transfer across both cell membranes, epithelial and capillary membranes. Biological membranes are composed of phospholipids stabilised
Characterization of changes observed in the corneal endothelium with the specular microscope
 Characterization of changes observed in the corneal endothelium with the specular microscope Emil S. Sherrard The specular microscope reveals little of the internal features of the corneal endothelium,
Characterization of changes observed in the corneal endothelium with the specular microscope Emil S. Sherrard The specular microscope reveals little of the internal features of the corneal endothelium,
Chapter 2 Transport Systems
 Chapter 2 Transport Systems The plasma membrane is a selectively permeable barrier between the cell and the extracellular environment. It permeability properties ensure that essential molecules such as
Chapter 2 Transport Systems The plasma membrane is a selectively permeable barrier between the cell and the extracellular environment. It permeability properties ensure that essential molecules such as
Acid and Base Balance
 Acid and Base Balance 1 2 The Body and ph Homeostasis of ph is tightly controlled Extracellular fluid = 7.4 Blood = 7.35 7.45 < 7.35: Acidosis (acidemia) > 7.45: Alkalosis (alkalemia) < 6.8 or > 8.0: death
Acid and Base Balance 1 2 The Body and ph Homeostasis of ph is tightly controlled Extracellular fluid = 7.4 Blood = 7.35 7.45 < 7.35: Acidosis (acidemia) > 7.45: Alkalosis (alkalemia) < 6.8 or > 8.0: death
Cellular Physiology. Body Fluids: 1) Water: (universal solvent) Body water varies based on of age, sex, mass, and body composition
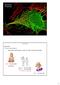 Membrane Physiology Body Fluids: 1) Water: (universal solvent) Body water varies based on of age, sex, mass, and body composition H 2 O ~ 73% body weight Low body fat; Low bone mass H 2 O ( ) ~ 60% body
Membrane Physiology Body Fluids: 1) Water: (universal solvent) Body water varies based on of age, sex, mass, and body composition H 2 O ~ 73% body weight Low body fat; Low bone mass H 2 O ( ) ~ 60% body
Consider the structure of the plasma membrane (fig. 8.6)- phospholipid bilayer with peripheral and integral proteins.
 Topic 8: MEMBRANE TRANSPORT (lectures 11-12) OBJECTIVES: 1. Have a basic appreciation of the chemical characteristics of substances that impact their ability to travel across plasma membranes. 2. Know
Topic 8: MEMBRANE TRANSPORT (lectures 11-12) OBJECTIVES: 1. Have a basic appreciation of the chemical characteristics of substances that impact their ability to travel across plasma membranes. 2. Know
Na + Transport 1 and 2 Linda Costanzo, Ph.D.
 Na + Transport 1 and 2 Linda Costanzo, Ph.D. OBJECTIVES: After studying this lecture, the student should understand: 1. The terminology applied to single nephron function, including the meaning of TF/P
Na + Transport 1 and 2 Linda Costanzo, Ph.D. OBJECTIVES: After studying this lecture, the student should understand: 1. The terminology applied to single nephron function, including the meaning of TF/P
ACTIVE TRANSPORT OF SODIUM BY THE ISOLATED MIDGUT OF HYALOPHORA CECROPIA
 J. Exp. Biol. (1971). 54. 269-374 269 With 1 text-figure Printed in Great Britain ACTIVE TRANSPORT OF SODIUM BY THE ISOLATED MIDGUT OF HYALOPHORA CECROPIA BY W. R. HARVEY AND K. ZERAHN* Department of Biology,
J. Exp. Biol. (1971). 54. 269-374 269 With 1 text-figure Printed in Great Britain ACTIVE TRANSPORT OF SODIUM BY THE ISOLATED MIDGUT OF HYALOPHORA CECROPIA BY W. R. HARVEY AND K. ZERAHN* Department of Biology,
Lactate and force production in skeletal muscle
 J Physiol 562.2 (2005) pp 521 526 521 Lactate and force production in skeletal muscle Michael Kristensen, Janni Albertsen, Maria Rentsch and Carsten Juel Copenhagen Muscle Research Centre, University of
J Physiol 562.2 (2005) pp 521 526 521 Lactate and force production in skeletal muscle Michael Kristensen, Janni Albertsen, Maria Rentsch and Carsten Juel Copenhagen Muscle Research Centre, University of
Duct Acidification Effects of Potassium, HCO3. Harry R. Jacobson. Internal Medicine, 5323 Harry Hines Blvd., Dallas, Texas 75235
 Medullary Collecting Duct Acidification Effects of Potassium, HCO3 Concentration, and pco2 Harry R. Jacobson University of Texas Health Science Center, Department of Internal Medicine, 5323 Harry Hines
Medullary Collecting Duct Acidification Effects of Potassium, HCO3 Concentration, and pco2 Harry R. Jacobson University of Texas Health Science Center, Department of Internal Medicine, 5323 Harry Hines
Interactions Between Cells and the Extracellular Environment
 Chapter 6 Interactions Between Cells and the Extracellular Environment Et Extracellular lll environment Includes all parts of the body outside of cells Cells receive nourishment Cells release waste Cells
Chapter 6 Interactions Between Cells and the Extracellular Environment Et Extracellular lll environment Includes all parts of the body outside of cells Cells receive nourishment Cells release waste Cells
CHAPTER 8 MEMBRANE STRUCTURE AND FUNCTION
 CHAPTER 8 MEMBRANE STRUCTURE AND FUNCTION Section B: Traffic Across Membranes 1. A membrane s molecular organization results in selective permeability 2. Passive transport is diffusion across a membrane
CHAPTER 8 MEMBRANE STRUCTURE AND FUNCTION Section B: Traffic Across Membranes 1. A membrane s molecular organization results in selective permeability 2. Passive transport is diffusion across a membrane
I n a previous article, 1 the membrane potentials
 Direct observation of secretory pumping in vitro of the rabbit eye ciliary processes Influence of ion milieu and carbonic anhydrase inhibition Lennart Berggren The function of the ciliary processes in
Direct observation of secretory pumping in vitro of the rabbit eye ciliary processes Influence of ion milieu and carbonic anhydrase inhibition Lennart Berggren The function of the ciliary processes in
The table indicates how changing the variable listed alone will alter diffusion rate.
 Rate of Diffusion (flux) Concentration gradient substance x surface area of membrane x lipid solubility = Distance (thickness of membrane) x molecular weight Table 3-1: Factors Influencing the Rate of
Rate of Diffusion (flux) Concentration gradient substance x surface area of membrane x lipid solubility = Distance (thickness of membrane) x molecular weight Table 3-1: Factors Influencing the Rate of
Only a small amount of cations is lost from the cells. A considerable. (Received 5 September 1967)
 J. Physiol. (1968), 195, pp. 107-118 107 With 5 text-figures Printed in Great Britain THE EFFECT OF HYPO- AND HYPERTONIC SOLUTIONS ON VOLUME AND ION DISTRIBUTION OF SMOOTH MUSCLE OF GUINEA-PIG TAENIA COLI
J. Physiol. (1968), 195, pp. 107-118 107 With 5 text-figures Printed in Great Britain THE EFFECT OF HYPO- AND HYPERTONIC SOLUTIONS ON VOLUME AND ION DISTRIBUTION OF SMOOTH MUSCLE OF GUINEA-PIG TAENIA COLI
Membrane transport. Pharmacy Dr. Szilvia Barkó
 Membrane transport Pharmacy 04.10.2017 Dr. Szilvia Barkó Cell Membranes Cell Membrane Functions Protection Communication Import and and export of molecules Movement of the cell General Structure A lipid
Membrane transport Pharmacy 04.10.2017 Dr. Szilvia Barkó Cell Membranes Cell Membrane Functions Protection Communication Import and and export of molecules Movement of the cell General Structure A lipid
Acidosis, alkalosis, and aqueous humor dynamics in rabbits. Theodore Krupin, Charles J. Oestrich, Jonathan Bass, Steven M. Podos, 9 and Bernard Becker
 Acidosis, alkalosis, and aqueous humor dynamics in rabbits Theodore Krupin, Charles J. Oestrich, Jonathan Bass, Steven M. Podos, 9 and Bernard Becker Systemic acidosis induced by intravenous administration
Acidosis, alkalosis, and aqueous humor dynamics in rabbits Theodore Krupin, Charles J. Oestrich, Jonathan Bass, Steven M. Podos, 9 and Bernard Becker Systemic acidosis induced by intravenous administration
Chapter MEMBRANE TRANSPORT
 Chapter 3 I MEMBRANE TRANSPORT The cell membrane, or plasma membrane, is the outermost layer of the cell. It completely surrounds the protoplasm or living portion of the cell, separating the cell s interior
Chapter 3 I MEMBRANE TRANSPORT The cell membrane, or plasma membrane, is the outermost layer of the cell. It completely surrounds the protoplasm or living portion of the cell, separating the cell s interior
COMMUNICATION BRIEF EFFECTS OF AMMONIUM AND BICARBONATE-CO2 ON INTRACELLULAR CHLORIDE LEVELS IN APLYSIA NEURONS
 BRIEF COMMUNICATION EFFECTS OF AMMONIUM AND BICARBONATE-CO2 ON INTRACELLULAR CHLORIDE LEVELS IN APLYSIA NEURONS JOHN M. RUSSELL, Department of Physiology and Biophysics, University of Texas Medical Branch,
BRIEF COMMUNICATION EFFECTS OF AMMONIUM AND BICARBONATE-CO2 ON INTRACELLULAR CHLORIDE LEVELS IN APLYSIA NEURONS JOHN M. RUSSELL, Department of Physiology and Biophysics, University of Texas Medical Branch,
Effect of Dexomethasone on Corneol Endotheliol Function in Fuchs' Dystrophy
 Investigative Ophthalmology & Visual Science, Vol. 29, No. 3, March 1988 Copyright Association for Research in Vision and Ophthalmology Effect of Dexomethasone on Corneol Endotheliol Function in Fuchs'
Investigative Ophthalmology & Visual Science, Vol. 29, No. 3, March 1988 Copyright Association for Research in Vision and Ophthalmology Effect of Dexomethasone on Corneol Endotheliol Function in Fuchs'
STEIN IN-TERM EXAM -- BIOLOGY FEBRUARY 16, PAGE
 STEIN IN-TERM EXAM -- BIOLOGY 3058 -- FEBRUARY 16, 2017 -- PAGE 1 of 9 There are 25 questions in this Biology 3058 exam. All questions are "A, B, C, D, E, F, G, H" questions worth one point each. There
STEIN IN-TERM EXAM -- BIOLOGY 3058 -- FEBRUARY 16, 2017 -- PAGE 1 of 9 There are 25 questions in this Biology 3058 exam. All questions are "A, B, C, D, E, F, G, H" questions worth one point each. There
Functions of Proximal Convoluted Tubules
 1. Proximal tubule Solute reabsorption in the proximal tubule is isosmotic (water follows solute osmotically and tubular fluid osmolality remains similar to that of plasma) 60-70% of water and solute reabsorption
1. Proximal tubule Solute reabsorption in the proximal tubule is isosmotic (water follows solute osmotically and tubular fluid osmolality remains similar to that of plasma) 60-70% of water and solute reabsorption
marked secretion ofcatecholamines and a subsequent inhibition ofsecretion although the basal secretion shows an initial rise.
 J. Physiol. (1969), 2, pp. 797-85 797 With 7 text-ftgurem Printed in Great Britain SODIUM IONS AND THE SECRETION OF CATECHOLAMINES By P. BANKS, ROSEMARY BIGGINS, R. BISHOP, B. CHRISTIAN AND N. CURRIE From
J. Physiol. (1969), 2, pp. 797-85 797 With 7 text-ftgurem Printed in Great Britain SODIUM IONS AND THE SECRETION OF CATECHOLAMINES By P. BANKS, ROSEMARY BIGGINS, R. BISHOP, B. CHRISTIAN AND N. CURRIE From
significant replacement of larger intracellular anions by extracellular chloride. The
 J. Physiol. (1979), 297, pp. 387-43 387 With 8 text-ftgurem Printed in Great Britain ANION EXCHANGE AND VOLUME REGULATION DURING METABOLIC BLOCKADE OF RENAL CORTICAL SLICES BY MICHAEL B. PINE, DAVID RHODES,
J. Physiol. (1979), 297, pp. 387-43 387 With 8 text-ftgurem Printed in Great Britain ANION EXCHANGE AND VOLUME REGULATION DURING METABOLIC BLOCKADE OF RENAL CORTICAL SLICES BY MICHAEL B. PINE, DAVID RHODES,
THE EFFECTS OF ION CHANGES ON THE CONTRACTION OF THE RAT UTERUS STIMULATED BY OXYTOCIN
 Brit. J. Pharmacol. (1961), 16, 45-49. THE EFFECTS OF ION CHANGES ON THE CONTRACTION OF THE RAT UTERUS STIMULATED BY OXYTOCIN BY P. J. BENTLEY AND ELEANOR McEWEN From the Department of Physiology, The
Brit. J. Pharmacol. (1961), 16, 45-49. THE EFFECTS OF ION CHANGES ON THE CONTRACTION OF THE RAT UTERUS STIMULATED BY OXYTOCIN BY P. J. BENTLEY AND ELEANOR McEWEN From the Department of Physiology, The
Efflux of Red Cell Water into Buffered Hypertonic Solutions
 Efflux of Red Cell Water into Buffered Hypertonic Solutions EDWIN G. OLMSTEAD From the School of Medicine, University of North Dakota, Grand Forks ABSTRACT Buffered NaCI solutions hypertonic to rabbit
Efflux of Red Cell Water into Buffered Hypertonic Solutions EDWIN G. OLMSTEAD From the School of Medicine, University of North Dakota, Grand Forks ABSTRACT Buffered NaCI solutions hypertonic to rabbit
Bear: Neuroscience: Exploring the Brain 3e
 Bear: Neuroscience: Exploring the Brain 3e Chapter 03: The Neuronal Membrane at Rest Introduction Action potential in the nervous system Action potential vs. resting potential Slide 1 Slide 2 Cytosolic
Bear: Neuroscience: Exploring the Brain 3e Chapter 03: The Neuronal Membrane at Rest Introduction Action potential in the nervous system Action potential vs. resting potential Slide 1 Slide 2 Cytosolic
Other Factors Affecting GFR. Chapter 25. After Filtration. Reabsorption and Secretion. 5 Functions of the PCT
 Other Factors Affecting GFR Chapter 25 Part 2. Renal Physiology Nitric oxide vasodilator produced by the vascular endothelium Adenosine vasoconstrictor of renal vasculature Endothelin a powerful vasoconstrictor
Other Factors Affecting GFR Chapter 25 Part 2. Renal Physiology Nitric oxide vasodilator produced by the vascular endothelium Adenosine vasoconstrictor of renal vasculature Endothelin a powerful vasoconstrictor
Membrane Function. How does the cell membrane control movement of materials? Type 1 Ions Type 2 Molecules Type 3 Molecules Type 4 Molecules H O H
 Why? Membrane Function ow does the cell membrane control movement of materials? The membrane is critical to the maintenance of homeostasis in living organisms. The cell membrane separates the cell from
Why? Membrane Function ow does the cell membrane control movement of materials? The membrane is critical to the maintenance of homeostasis in living organisms. The cell membrane separates the cell from
Acid-base balance is one of the most important of the body s homeostatic mechanisms Acid-base balance refers to regulation of hydrogen ion (H + )
 Acid-base balance is one of the most important of the body s homeostatic mechanisms Acid-base balance refers to regulation of hydrogen ion (H + ) concentration in body fluids Precise regulation of ph at
Acid-base balance is one of the most important of the body s homeostatic mechanisms Acid-base balance refers to regulation of hydrogen ion (H + ) concentration in body fluids Precise regulation of ph at
Acids and Bases their definitions and meanings
 Acids and Bases their definitions and meanings Molecules containing hydrogen atoms that can release hydrogen ions in solutions are referred to as acids. (HCl H + Cl ) (H 2 CO 3 H + HCO 3 ) A base is an
Acids and Bases their definitions and meanings Molecules containing hydrogen atoms that can release hydrogen ions in solutions are referred to as acids. (HCl H + Cl ) (H 2 CO 3 H + HCO 3 ) A base is an
Stimulation of Active K + Transport by Anti-L Antibodies in Trypsin-Treated Low Potassium Sheep Erythrocytes
 LETTER TO THE EDITOR Stimulation of Active K + Transport by Anti-L Antibodies in Trypsin-Treated Low Potassium Sheep Erythrocytes Dear Sir: In this letter we attempt to resolve a discrepancy on the effect
LETTER TO THE EDITOR Stimulation of Active K + Transport by Anti-L Antibodies in Trypsin-Treated Low Potassium Sheep Erythrocytes Dear Sir: In this letter we attempt to resolve a discrepancy on the effect
CHAPTER 27 LECTURE OUTLINE
 CHAPTER 27 LECTURE OUTLINE I. INTRODUCTION A. Body fluid refers to body water and its dissolved substances. B. Regulatory mechanisms insure homeostasis of body fluids since their malfunction may seriously
CHAPTER 27 LECTURE OUTLINE I. INTRODUCTION A. Body fluid refers to body water and its dissolved substances. B. Regulatory mechanisms insure homeostasis of body fluids since their malfunction may seriously
Membrane Transport. Anatomy 36 Unit 1
 Membrane Transport Anatomy 36 Unit 1 Membrane Transport Cell membranes are selectively permeable Some solutes can freely diffuse across the membrane Some solutes have to be selectively moved across the
Membrane Transport Anatomy 36 Unit 1 Membrane Transport Cell membranes are selectively permeable Some solutes can freely diffuse across the membrane Some solutes have to be selectively moved across the
5.6 Diffusion, Membranes, and Metabolism
 5.6 Diffusion, Membranes, and Metabolism Concentration of a substance Number of atoms or molecules in a given volume Concentration gradient of a substance A difference in concentration between two regions
5.6 Diffusion, Membranes, and Metabolism Concentration of a substance Number of atoms or molecules in a given volume Concentration gradient of a substance A difference in concentration between two regions
The kidney. (Pseudo) Practical questions. The kidneys are all about keeping the body s homeostasis. for questions Ella
 The kidney (Pseudo) Practical questions for questions Ella (striemit@gmail.com) The kidneys are all about keeping the body s homeostasis Ingestion Product of metabolism H 2 O Ca ++ Cl - K + Na + H 2 O
The kidney (Pseudo) Practical questions for questions Ella (striemit@gmail.com) The kidneys are all about keeping the body s homeostasis Ingestion Product of metabolism H 2 O Ca ++ Cl - K + Na + H 2 O
ACID-BASE BALANCE URINE BLOOD AIR
 ACIDBASE BALANCE URINE BLOOD AIR H 2 PO 4 NH 4 HCO 3 KIDNEY H H HCO 3 CELLS Hb H LUNG H 2 CO 3 HHb CO 2 H 2 O ph = 7.4 [HCO 3 ] = 24 meq/l PCO 2 = 40 mm Hg CO 2 PRIMARY RENAL MECHANISMS INVOLVED IN ACIDBASE
ACIDBASE BALANCE URINE BLOOD AIR H 2 PO 4 NH 4 HCO 3 KIDNEY H H HCO 3 CELLS Hb H LUNG H 2 CO 3 HHb CO 2 H 2 O ph = 7.4 [HCO 3 ] = 24 meq/l PCO 2 = 40 mm Hg CO 2 PRIMARY RENAL MECHANISMS INVOLVED IN ACIDBASE
HANS H. USSING, THOMAS U. L. BIBER, and NEAL S. BRICKER
 Exposure of the Isolated Frog Skin to High Potassium Concentrations at the Internal Surface II. Changes in epithelial cell volume, resistance, and response to antidiuretic hormone HANS H. USSING, THOMAS
Exposure of the Isolated Frog Skin to High Potassium Concentrations at the Internal Surface II. Changes in epithelial cell volume, resistance, and response to antidiuretic hormone HANS H. USSING, THOMAS
STEIN IN-TERM EXAM -- BIOLOGY FEBRUARY 15, PAGE
 STEIN IN-TERM EXAM -- BIOLOGY 3058 -- FEBRUARY 15, 2018 -- PAGE 1 of 8 There are 25 questions in this Biology 3058 exam. All questions are "A, B, C, D, E, F, G, H" questions worth one point each. There
STEIN IN-TERM EXAM -- BIOLOGY 3058 -- FEBRUARY 15, 2018 -- PAGE 1 of 8 There are 25 questions in this Biology 3058 exam. All questions are "A, B, C, D, E, F, G, H" questions worth one point each. There
Acid Base Balance. Professor Dr. Raid M. H. Al-Salih. Clinical Chemistry Professor Dr. Raid M. H. Al-Salih
 Acid Base Balance 1 HYDROGEN ION CONCENTRATION and CONCEPT OF ph Blood hydrogen ion concentration (abbreviated [H + ]) is maintained within tight limits in health, with the normal concentration being between
Acid Base Balance 1 HYDROGEN ION CONCENTRATION and CONCEPT OF ph Blood hydrogen ion concentration (abbreviated [H + ]) is maintained within tight limits in health, with the normal concentration being between
Module 8: Electrolyte Solutions
 PHARMACEUTICAL CALCULATIONS FALL 207 Contents General Vocab & Definitions... 2 Milliequivalent Calculations... 2 Millimole and Micromole Calculations... 5 Osmolarity... 6 Daily Water Requirement Calculations
PHARMACEUTICAL CALCULATIONS FALL 207 Contents General Vocab & Definitions... 2 Milliequivalent Calculations... 2 Millimole and Micromole Calculations... 5 Osmolarity... 6 Daily Water Requirement Calculations
Acid-Base Balance Dr. Gary Mumaugh
 Acid-Base Balance Dr. Gary Mumaugh Introduction Acid-base balance is one of the most important of the body s homeostatic mechanisms Acid-base balance refers to regulation of hydrogen ion (H + ) concentration
Acid-Base Balance Dr. Gary Mumaugh Introduction Acid-base balance is one of the most important of the body s homeostatic mechanisms Acid-base balance refers to regulation of hydrogen ion (H + ) concentration
Chapter 19 The Urinary System Fluid and Electrolyte Balance
 Chapter 19 The Urinary System Fluid and Electrolyte Balance Chapter Outline The Concept of Balance Water Balance Sodium Balance Potassium Balance Calcium Balance Interactions between Fluid and Electrolyte
Chapter 19 The Urinary System Fluid and Electrolyte Balance Chapter Outline The Concept of Balance Water Balance Sodium Balance Potassium Balance Calcium Balance Interactions between Fluid and Electrolyte
Membrane Structure and Function
 BIOL1040 Page 1 Membrane Structure and Function Friday, 6 March 2015 2:58 PM Cellular Membranes Fluid mosaics of lipids and proteins Phospholipids - abundant Phospholipids are amphipathic molecules (has
BIOL1040 Page 1 Membrane Structure and Function Friday, 6 March 2015 2:58 PM Cellular Membranes Fluid mosaics of lipids and proteins Phospholipids - abundant Phospholipids are amphipathic molecules (has
Electrically Silent Na + and Cl Fluxes Across the Rabbit Ciliary Epithelium
 Electrically Silent Na + and Cl Fluxes Across the Rabbit Ciliary Epithelium Teh-Ching Chu* and Oscar A. Candia*f Unidirectional Na +, Cl~, and mannitol fluxes were measured across the isolated and short-circuited
Electrically Silent Na + and Cl Fluxes Across the Rabbit Ciliary Epithelium Teh-Ching Chu* and Oscar A. Candia*f Unidirectional Na +, Cl~, and mannitol fluxes were measured across the isolated and short-circuited
Membrane Structure, Resting membrane potential, Action potential. Biophysics seminar
 Membrane Structure, Resting membrane potential, Action potential Biophysics seminar 09.09.2013. Membrane structure Biological membranes consists of lipids and proteins to bind with non-covalent bond. Phospholipids
Membrane Structure, Resting membrane potential, Action potential Biophysics seminar 09.09.2013. Membrane structure Biological membranes consists of lipids and proteins to bind with non-covalent bond. Phospholipids
Questions. Question 1!
 Questions Question 1 In a laboratory, scientists often study neurons in isolation, outside of a living creature, in a dish. In this setting, one can have a good deal of control over the local ionic environment
Questions Question 1 In a laboratory, scientists often study neurons in isolation, outside of a living creature, in a dish. In this setting, one can have a good deal of control over the local ionic environment
UNIVERSITY OF MEDICAL SCIENCES, ONDO DEPARTMENT OF PHYSIOLOGY PHS 211 TRANSPORT MECHANISM LECTURER: MR A.O. AKINOLA
 UNIVERSITY OF MEDICAL SCIENCES, ONDO DEPARTMENT OF PHYSIOLOGY PHS 211 TRANSPORT MECHANISM LECTURER: MR A.O. AKINOLA OUTLINE Introduction Basic mechanisms Passive transport Active transport INTRODUCTION
UNIVERSITY OF MEDICAL SCIENCES, ONDO DEPARTMENT OF PHYSIOLOGY PHS 211 TRANSPORT MECHANISM LECTURER: MR A.O. AKINOLA OUTLINE Introduction Basic mechanisms Passive transport Active transport INTRODUCTION
Diffusion & Osmosis - Exercise 4
 Diffusion & Osmosis - Exercise 4 Objectives -Define: Solvent, Solute, and Solution -Define: Diffusion, Selectively permeable membrane, Osmosis, and Dialysis -Understand rule of thumb: Concentration will
Diffusion & Osmosis - Exercise 4 Objectives -Define: Solvent, Solute, and Solution -Define: Diffusion, Selectively permeable membrane, Osmosis, and Dialysis -Understand rule of thumb: Concentration will
Medical Center, Los Angeles, California 90024, U.S.A. (Received 27 July 1970)
 J. Physiol. (1971), 212, pp. 277-286 277 With 1 text-figure Printed in Great Britain GALACTOSE TRANSPORT ACROSS THE HAMSTER SMALL INTESTINE; THE EFFECT OF SODIUM ELECTROCHEMICAL POTENTIAL GRADIENTS By
J. Physiol. (1971), 212, pp. 277-286 277 With 1 text-figure Printed in Great Britain GALACTOSE TRANSPORT ACROSS THE HAMSTER SMALL INTESTINE; THE EFFECT OF SODIUM ELECTROCHEMICAL POTENTIAL GRADIENTS By
What location in the gastrointestinal (GI) tract has tight, or impermeable, junctions between the epithelial cells?
 CASE 32 A 17-year-old boy presents to his primary care physician with complaints of diarrhea for the last 2 days. The patient states that he just returned to the United States after visiting relatives
CASE 32 A 17-year-old boy presents to his primary care physician with complaints of diarrhea for the last 2 days. The patient states that he just returned to the United States after visiting relatives
BIOL 2402 Fluid/Electrolyte Regulation
 Dr. Chris Doumen Collin County Community College BIOL 2402 Fluid/Electrolyte Regulation 1 Body Water Content On average, we are 50-60 % water For a 70 kg male = 40 liters water This water is divided into
Dr. Chris Doumen Collin County Community College BIOL 2402 Fluid/Electrolyte Regulation 1 Body Water Content On average, we are 50-60 % water For a 70 kg male = 40 liters water This water is divided into
The regulation of renal acid secretion: New observations from studies of distal nephron segments
 Kidney International, Vol. 29 (1986), pp. 1099 1109 EDITORIAL REVIEW The regulation of renal acid secretion: New observations from studies of distal nephron segments Forty years ago, in a landmark paper,
Kidney International, Vol. 29 (1986), pp. 1099 1109 EDITORIAL REVIEW The regulation of renal acid secretion: New observations from studies of distal nephron segments Forty years ago, in a landmark paper,
Relaxation responses of aortic rings from salt-loaded high calcium fed rats to potassium chloride, calcium chloride and magnesium sulphate
 Pathophysiology 4 (1998) 275 280 Relaxation responses of aortic rings from salt-loaded high calcium fed rats to potassium chloride, calcium chloride and magnesium sulphate B.J. Adegunloye, O.A. Sofola
Pathophysiology 4 (1998) 275 280 Relaxation responses of aortic rings from salt-loaded high calcium fed rats to potassium chloride, calcium chloride and magnesium sulphate B.J. Adegunloye, O.A. Sofola
Plasma Membrane Function
 Plasma Membrane Function Cells have to maintain homeostasis, they do this by controlling what moves across their membranes Structure Double Layer of phospholipids Head (polar) hydrophiliclikes water -
Plasma Membrane Function Cells have to maintain homeostasis, they do this by controlling what moves across their membranes Structure Double Layer of phospholipids Head (polar) hydrophiliclikes water -
FIGURE A. The phosphate end of the molecule is polar (charged) and hydrophilic (attracted to water).
 PLASMA MEMBRANE 1. The plasma membrane is the outermost part of a cell. 2. The main component of the plasma membrane is phospholipids. FIGURE 2.18 A. The phosphate end of the molecule is polar (charged)
PLASMA MEMBRANE 1. The plasma membrane is the outermost part of a cell. 2. The main component of the plasma membrane is phospholipids. FIGURE 2.18 A. The phosphate end of the molecule is polar (charged)
DIURETICS-2. Dr. Shariq Syed. Shariq AIKC/TYB/2014
 DIURETICS-2 Dr. Syed Structure of Kidney Blood filtered by functional unit: Nephron Except for cells, proteins, other large molecules, rest gets filtered Structure of Kidney 3 major regions of nephron
DIURETICS-2 Dr. Syed Structure of Kidney Blood filtered by functional unit: Nephron Except for cells, proteins, other large molecules, rest gets filtered Structure of Kidney 3 major regions of nephron
Emergency Medical Training Services Emergency Medical Technician Paramedic Program Outlines Outline Topic: Patho Instructor Notes Revised: 11/2013
 Emergency Medical Training Services Emergency Medical Technician Paramedic Program Outlines Outline Topic: Patho Instructor Notes Revised: 11/2013 Cells form 4 basic tissue groups: 1. Epithelial 2. Connective
Emergency Medical Training Services Emergency Medical Technician Paramedic Program Outlines Outline Topic: Patho Instructor Notes Revised: 11/2013 Cells form 4 basic tissue groups: 1. Epithelial 2. Connective
Cellular Neurophysiology I Membranes and Ion Channels
 Cellular Neurophysiology I Membranes and Ion Channels Reading: BCP Chapter 3 www.bioelectriclab All living cells maintain an electrical potential (voltage) across their membranes (V m ). Resting Potential
Cellular Neurophysiology I Membranes and Ion Channels Reading: BCP Chapter 3 www.bioelectriclab All living cells maintain an electrical potential (voltage) across their membranes (V m ). Resting Potential
PHYSIOLOGY AND MAINTENANCE Vol. I Mechanisms of Cell Volume Regulation - Alexander A. Mongin, Sergei N. Orlov MECHANISMS OF CELL VOLUME REGULATION
 MECHANISMS OF CELL VOLUME REGULATION Alexander A. Mongin Albany Medical College, Albany, NY, USA Sergei N. Orlov University Hospital Research Centre (CHUM) and Department of Medicine, Université de Montréal,
MECHANISMS OF CELL VOLUME REGULATION Alexander A. Mongin Albany Medical College, Albany, NY, USA Sergei N. Orlov University Hospital Research Centre (CHUM) and Department of Medicine, Université de Montréal,
Part 1 The Cell and the Cellular Environment
 1 Chapter 3 Anatomy and Physiology Part 1 The Cell and the Cellular Environment 2 The Human Cell The is the fundamental unit of the human body. Cells contain all the necessary for life functions. 3 Cell
1 Chapter 3 Anatomy and Physiology Part 1 The Cell and the Cellular Environment 2 The Human Cell The is the fundamental unit of the human body. Cells contain all the necessary for life functions. 3 Cell
16. Excitability o f Squid Giant Axons in Hypertonic and Hypotonic Solutions
 68 Proc. Japan Acad., 51 (1975) [Vol. 51, 16. Excitability o f Squid Giant Axons in Hypertonic and Hypotonic Solutions By Fumio KUKITA*> and Shunichi YAMAGISHI**) (Comm. by Yasuj i KATSUKI, M. J. A., Jan.
68 Proc. Japan Acad., 51 (1975) [Vol. 51, 16. Excitability o f Squid Giant Axons in Hypertonic and Hypotonic Solutions By Fumio KUKITA*> and Shunichi YAMAGISHI**) (Comm. by Yasuj i KATSUKI, M. J. A., Jan.
DIFFUSON AND OSMOSIS INTRODUCTION diffusion concentration gradient. net osmosis water potential active transport
 DIFFUSON AND OSMOSIS NAME DATE INTRODUCTION The life of a cell is dependent on efficiently moving material into and out of the cell across the cell membrane. Raw materials such as oxygen and sugars needed
DIFFUSON AND OSMOSIS NAME DATE INTRODUCTION The life of a cell is dependent on efficiently moving material into and out of the cell across the cell membrane. Raw materials such as oxygen and sugars needed
Acid-Base Balance 11/18/2011. Regulation of Potassium Balance. Regulation of Potassium Balance. Regulatory Site: Cortical Collecting Ducts.
 Influence of Other Hormones on Sodium Balance Acid-Base Balance Estrogens: Enhance NaCl reabsorption by renal tubules May cause water retention during menstrual cycles Are responsible for edema during
Influence of Other Hormones on Sodium Balance Acid-Base Balance Estrogens: Enhance NaCl reabsorption by renal tubules May cause water retention during menstrual cycles Are responsible for edema during
Diuretics having the quality of exciting excessive excretion of urine. OED. Inhibitors of Sodium Reabsorption Saluretics not Aquaretics
 Diuretics having the quality of exciting excessive excretion of urine. OED Inhibitors of Sodium Reabsorption Saluretics not Aquaretics 1 Sodium Absorption Na Entry into the Cell down an electrochemical
Diuretics having the quality of exciting excessive excretion of urine. OED Inhibitors of Sodium Reabsorption Saluretics not Aquaretics 1 Sodium Absorption Na Entry into the Cell down an electrochemical
CHAPTER. Movement Across Plasma Membrane. Chapter 6 Outline. Diffusion Osmosis. Membrane Potential Cell Signaling
 CHAPTER 6 Interaction Between Cells and the Extracellular Environment Chapter 6 Outline Extracellular Environment Diffusion Osmosis Carrier-Mediated Carrier Mediated Transport Membrane Potential Cell Signaling
CHAPTER 6 Interaction Between Cells and the Extracellular Environment Chapter 6 Outline Extracellular Environment Diffusion Osmosis Carrier-Mediated Carrier Mediated Transport Membrane Potential Cell Signaling
Homeostatic Regulation
 Homeostatic Regulation A hormone is :a Water-soluble hormones: Composed of amino acids and bind a receptor protein on the of the target cell. This starts a signal cascade inside the cell and the signal
Homeostatic Regulation A hormone is :a Water-soluble hormones: Composed of amino acids and bind a receptor protein on the of the target cell. This starts a signal cascade inside the cell and the signal
Multiple Choice Review- Membranes & Enzymes
 Multiple Choice Review- Membranes & Enzymes 1. Cell membranes are and regulate the materials moving into and out of the cell, in order to maintain equilibrium. a. completely permeable b. ionically permeable
Multiple Choice Review- Membranes & Enzymes 1. Cell membranes are and regulate the materials moving into and out of the cell, in order to maintain equilibrium. a. completely permeable b. ionically permeable
Acid - base equilibrium
 Acid base equilibrium ph concept ph = log [H + ] ph [H+] 1 100 mmol/l D = 90 mmol/l 2 10 mmol/l D = 9 mmol/l 3 1 mmol/l 2 ph = log [H + ] 3 ph ph = log [H + ] ph of capillary blood norm: 7,35 7,45 Sorensen
Acid base equilibrium ph concept ph = log [H + ] ph [H+] 1 100 mmol/l D = 90 mmol/l 2 10 mmol/l D = 9 mmol/l 3 1 mmol/l 2 ph = log [H + ] 3 ph ph = log [H + ] ph of capillary blood norm: 7,35 7,45 Sorensen
CONTRACTILITY AND 4s Ca FLUXES IN HEART MUSCLE OF FLOUNDER AT A LOWERED EXTRACELLULAR NaCl CONCENTRATION
 y. exp. Bio/. 9, 2-27 (984) 2 Printed in Great Britain The Company of Biologists Limited 984 CONTRACTILITY AND 4s Ca FLUXES IN HEART MUSCLE OF FLOUNDER AT A LOWERED EXTRACELLULAR NaCl CONCENTRATION BY
y. exp. Bio/. 9, 2-27 (984) 2 Printed in Great Britain The Company of Biologists Limited 984 CONTRACTILITY AND 4s Ca FLUXES IN HEART MUSCLE OF FLOUNDER AT A LOWERED EXTRACELLULAR NaCl CONCENTRATION BY
1.14. Passive Transport
 Passive Transport 1.14 Simple Diffusion Cell s are selectively permeable only certain substances are able to pass through them. As mentioned in section 1.2, cell s are largely composed of a phospholipid
Passive Transport 1.14 Simple Diffusion Cell s are selectively permeable only certain substances are able to pass through them. As mentioned in section 1.2, cell s are largely composed of a phospholipid
to reduce mechanical trauma. In the second method corneae were mounted on specular microscopy rings
 British Journal of Ophthalmology, 1982, 66, 136-14 Assessment and interpretation of corneal endothelial cell morphology and function following cryopreservation P. W. MADDEN AND D. L. EASTY From the Department
British Journal of Ophthalmology, 1982, 66, 136-14 Assessment and interpretation of corneal endothelial cell morphology and function following cryopreservation P. W. MADDEN AND D. L. EASTY From the Department
Cell Structure and Function Exam Study Guide Part I
 Cell Structure and Function Exam Study Guide Part I 1. Which image best depicts the hot water, which the cold? 2. What is the relationship between temperature and the speed of molecular motion? 3. If a
Cell Structure and Function Exam Study Guide Part I 1. Which image best depicts the hot water, which the cold? 2. What is the relationship between temperature and the speed of molecular motion? 3. If a
All biological membranes are bilayers of phospholipid. The proteins in each type of membrane give it its unique properties.
 Many important drugs affect cell surface transport proteins DRUG TARGET PROTEIN DISEASE Omeprazole (Losec) gastric H + /K + ATPase ulcers, gastric reflex oral rehydration therapy intestinal Na + glucose
Many important drugs affect cell surface transport proteins DRUG TARGET PROTEIN DISEASE Omeprazole (Losec) gastric H + /K + ATPase ulcers, gastric reflex oral rehydration therapy intestinal Na + glucose
Managing Acid Base and Electrolyte Disturbances with RRT
 Managing Acid Base and Electrolyte Disturbances with RRT John R Prowle MA MSc MD MRCP FFICM Consultant in Intensive Care & Renal Medicine RRT for Regulation of Acid-base and Electrolyte Acid base load
Managing Acid Base and Electrolyte Disturbances with RRT John R Prowle MA MSc MD MRCP FFICM Consultant in Intensive Care & Renal Medicine RRT for Regulation of Acid-base and Electrolyte Acid base load
Electrical Potential Differences and Electromotive Forces in Epithelial Tissues
 LETTER TO THE EDITOR [Brief letters to the Editor that make specific scientific reference to papers published previously in THE JOURNAL OF GENERAL PHYSIOLOGY are invited. Receipt of such letters will be
LETTER TO THE EDITOR [Brief letters to the Editor that make specific scientific reference to papers published previously in THE JOURNAL OF GENERAL PHYSIOLOGY are invited. Receipt of such letters will be
