Acute coronary syndromes refers to a spectrum of
|
|
|
- Holly Curtis
- 6 years ago
- Views:
Transcription
1 Appropriate Use of Glycoprotein IIb/IIIa Blockade for Unstable Angina and Non ST Segment Elevation Myocardial Infarction Albert W. Chan, MD, MS, FRCP(C), and Sorin J. Brener, MD Objective: To review the clinical trials of glycoprotein IIb/IIIa (GPIIb/IIIa) inhibitors for unstable angina or non ST segment elevation myocardial infarction (MI) and to provide a practice guide for the use of these agents in this setting. Methods: Clinical trials relating to GPIIb/IIIa antagonists and acute coronary syndromes were identified from a MEDLINE search of articles published between 1994 and 2001 and from presentations made during the official meetings of the American College of Cardiology, American Heart Association, and the European Heart Society in 1999 and Clinical trials related to ST segment elevation were excluded. Results: Eleven clinical trials were identified, 7 relating to intravenous GPIIb/IIIa inhibitors and 4 relating to oral GPIIb/IIIa inhibition. When combined with aspirin and unfractionated heparin, eptifibatide and tirofiban improved clinical outcomes in patients with unstable angina or non ST segment elevation MI, especially in the context of early revascularization. Abciximab was beneficial only as an adjunct to percutaneous coronary intervention. Oral chronic GPIIb/IIIa inhibition was associated with increased mortality in each of the clinical trials that were completed. Conclusion: Barring the inherent differences in their structures and pharmacokinetics, both tirofiban and eptifibatide improve clinical outcomes of patients with unstable angina or non ST segment elevation myocardial infarction, regardless of revascularization strategy, while abciximab plays a pivotal role in percutaneous coronary intervention for acute coronary syndromes. For oral GPIIb/IIIa inhibitors, more investigation is required to better understand the biologic properties of these drugs before further clinical testing. Acute coronary syndromes refers to a spectrum of clinical presentations including unstable angina, non-q wave myocardial infarction (MI), Q wave MI, and sudden ischemic death [1]. The presence of new ST segment elevation or left bundle branch block determines the eligibility of reperfusion therapy. In the absence of these findings, careful monitoring and appropriate use of pharmacotherapy will be required based on individual risk profile. Unstable angina and non ST segment elevation MI have similar presentation, and the distinction between the 2 can only be made several hours later using serum markers for myocardial necrosis (ie, creatine kinase MB, troponin T or I) [1]. Clinical features suggesting short-term high risk include age greater than 75 years, prolonged (> 20 minutes) rest pain or recurrent ischemic pain, left ventricular dysfunction, pulmonary edema, transient electrocardiographic ST-T changes, and elevation of cardiac troponin T, troponin I, or creatine kinase MB [2]. Pivotal to the pathogenesis of acute coronary syndromes is platelet activation and aggregation. Administration of aspirin plus unfractionated heparin or low-molecular-weight heparin has been established in the standard guideline as the initial management for acute coronary syndromes [2]. However, the antiplatelet effect of aspirin is rather modest. For intermediate and high-risk patients presenting with acute coronary syndromes, glycoprotein IIb/IIIa (GPIIb/IIIa) antagonists can provide additional therapeutic benefit. Three classes of intravenous GPIIb/IIIa inhibitors have been tested in the clinical trials for the treatment of acute coronary syndromes: human-murine chimeric monoclonal antibody fragment (abciximab), synthetic peptide (eptifibatide), and synthetic non-peptide forms (tirofiban, lamifiban). All except for lamifiban are commercially available. While all GPIIb/IIIa inhibitors have a rapid onset of antiplatelet action, the biologic profiles of these agents are significantly different (Table 1). In this article, we review the clinical trials related to the use of GPIIb/IIIa antagonists in patients with unstable angina and non ST segment elevation MI. From the Department of Cardiology, The Cleveland Clinic Foundation, Cleveland, OH. Vol. 8, No. 4 April 2001 JCOM 33
2 GLYCOPROTEIN IIb/IIIa BLOCKADE Table 1. Biologic Properties of 4 Glycoprotein IIb/IIIa Antagonists Specificity to Platelet Adjustment for Agent Composition GPIIb/IIIa Receptors Pharmacokinetics Inhibition Renal Insufficiency Abciximab (Reopro) Eptifibatide (Integrilin) Tirofiban HCl (Aggrastat) Lamifaban Fab fragment of the chimeric human-murine monoclonal antibody 7E3 Cyclic heptapeptide with 6 amino acids and 1 mercaptopropiionyl residual Non-peptide tyrosine derivative Non-peptide tyrosine derivative Nonspecific to GPIIb/IIIa receptor; also binds to the vitronectin and Mac- 1 receptors, the significance of which is unclear Specific Specific Specific T1 / 2 = hours Protein binding: no Renal excretion: no T1 / 2 = 2 4 hours Protein binding: 25% Renal excretion: 50% T1 / = 4 8 hours 2 Protein binding: 65% Renal excretion: 65% > 90% > 90%* 40% 50% > 90% No Yes Reduce bolus and infusion rates by 50% if CrCl < 30 ml/min CrCl = creatine clearance. *135 µg/kg bolus and 0.5 µg/kg/min infusion. 180 µg/kg bolus and 2.0 µg/kg/min infusion. Methods Reports published between 1994 and January 2001 relating to GPIIb/IIIa inhibition for acute coronary syndromes were identified on MEDLINE using the keywords glycoprotein IIb/IIIa antagonists, unstable angina, myocardial infarction, and human. Randomized, double-blinded, placebo-controlled trials that enrolled more than 500 patients were included in the review. Reports of the primary studies, as well as those of the subsequent sub-analyses, were retrieved. Relevant abstracts and late-breaking trials presented at the annual scientific sessions of the American College of Cardiology, American Heart Association, and the European Society of Cardiology in the years 1999 and 2000 were also reviewed. Studies that were related to ST segment elevation MI were excluded from analysis. Results A total of 11 major randomized trials were identified [3 13]. Seven trials were related to intravenous GPIIb/IIIa antagonists and 4 were related to oral therapy, enrolling a total of 31,402 and 35,294 patients respectively. Five of the intravenous GPIIb/IIIa antagonist trials (collectively known as the 5Ps) led to the U.S. Food and Drug Administration approval of eptifibatide and tirofiban for initial medical management of acute coronary syndromes. These trials are the Platelet IIb/IIIa Antagonism for the Reduction of Acute Coronary syndrome Events in a Global Organization Network A and B (PARAGON A and B) [3,4], the Platelet Receptor Inhibition in Ischemic Syndrome Management (PRISM) [5], the Platelet Receptor Inhibition in Ischemic Syndrome Management in Patients Limited by Unstable Signs and Symptoms (PRISM-PLUS) [6], and the Platelet Glycoprotein IIb/IIIa in Unstable Angina: Receptor Suppression Using Integrilin Therapy (PURSUIT) [7]. The use of abciximab as an adjunctive therapy for acute coronary syndromes patients scheduled for urgent percutaneous coronary intervention was examined in the c7e3 Fab Antiplatelet Therapy in Unstable Refractory Angina (CAPTURE) trial [9]. The Global Use of Strategies To open Occluded coronary arteries (GUSTO) IV trial, which aimed to study abciximab as a primary medical regimen for acute coronary syndromes, was recently completed [8]. Efficacy of the Synthetic Molecules in Acute Coronary Syndromes: The 5 P Trials Five randomized trials evaluated the role of these highly specific GPIIb/IIIa receptor blockers for initial medical management of unstable angina or non ST segment elevation MI (Tables 2 and 3, Figure 1). Except for PURSUIT, cardiac catheterization and percutaneous coronary intervention were discouraged until 48 to 72 hours after randomization. PARAGON A was a dose-finding study that randomized 2282 patients to 5 regimens: high- and low-dose lamifiban, with or without heparin, and heparin alone [3]. At 30 days, there was no difference in death or MI among the 5 groups. However, at 6-month follow-up, patients assigned to the lowdose lamifiban with or without heparin did better than the placebo group (death or MI, 13.7% versus 17.9%, P = 0.027), but high-dose lamifiban subgroup did not (16.4% versus 17.9%, P = not significant) (Table 3). Postulated mechanisms 34 JCOM April 2001 Vol. 8, No. 4
3 Table 2. Overview of Trials Evaluating Glycoprotein IIb/IIIa Inhibitors in Acute Coronary Syndromes PCI Trial Year n Treatment Patient Profile Dose Regimen Primary Endpoints Performed CAPTURE [9] GUSTO-IV [8] PURSUIT [7] ,948 Abciximab versus placebo Abciximab versus placebo* Eptifibatide versus placebo Refractory UA (within 48 hours) with ST-T changes, and angiographic lesion suitable for PCI Symptoms of angina with troponin T elevation or ST-T changes UA within 24 hours, with CKMB elevation or ST-T changes 1. Abciximab 0.25 mg/kg bolus + 10 µg/min hours before PTCA and continue for 1 hour after PTCA 2. Placebo 1. Abciximab bolus + 48-hour infusion 2. Abciximab bolus + 24-hour infusion 3. Placebo 1. Eptifibatide 180 µg/kg bolus µg/kg/min 72 hours 2. Eptifibatide 180 µg/kg bolus µg/kg/min 72 hours 3. Placebo Death/MI/urgent revascularization at 30 days Death/MI at 30 days Death/MI at 30 days All < 5% 24% PRISM [5] PRISM-PLUS [6] Tirofiban versus placebo Tirofiban (high-dose) versus tirofiban (low-dose) + heparin versus heparin UA/NQMI within 24 hours, with new ST-T changes, elevated CKMB, or history of CAD UA/NQMI within 12 hours and new ischemic ST-T changes 1. Tirofiban 0.6 µg/kg/min 30 min µg/kg/min infusion 47.5 hours 2. Placebo 1. Tirofiban 0.6 µg/kg/min 30 min µg/kg/min infusion 48 hours + placebo heparin 48 hours 2. Tirofiban 0.4 µg/kg/min 30 min µg/kg/min infusion 48 hours + heparin 48 hours 3. Heparin alone Death/MI/refractory ischemia at 48 hours Death/MI/refractory ischemia within 7 days 21% 31% PARAGON-A [3] arms: High-dose lamifiban (± heparin) versus low-dose lamifiban (± heparin) versus heparin UA within 12 hours with ST-T changes 1. Lamifiban 750 µg bolus µg/min infusion (± heparin) 3 5 days 2. Lamifiban 300 µg bolus mg/min infusion (± heparin) 3 5 days 3. Heparin alone Death/MI at 30 days 14% PARAGON-B [4] Lamifiban versus placebo UA within 12 hours with ST-T changes or elevated CKMB (or troponin T) 1. Lamifiban 500 mg bolus + 1.0, 1.5, or 2.0 mg/min infusion (based on SCr) 72 hours 2. Placebo Death/MI/recurrent ischemia at 30 days 27% CAD = coronary artery disease; CKMB = creatine kinase MB; MI = myocardial infarction; NQMI = non Q wave myocardial infarction; PCI = percutaneous coronary intervention; PTCA = percutaneous transluminal coronary angioplasty; SCr = serum creatine; UA = unstable angina. *Low-molecular-weight heparin substudy was performed in a fraction of patients in each group. Unfractionated heparin was given to the rest of the patients. All patients in both treatment and control groups received aspirin, unfractionated heparin bolus, and infusion. Vol. 8, No. 4 April 2001 JCOM 35
4 GLYCOPROTEIN IIb/IIIa BLOCKADE Table 3. Incidence of Death or Nonfatal MI at 6 Months in GPIIb/IIIa Acute Coronary Syndrome Trials Incidence of Death or MI at 6 Months, % Trial GPIIb/IIIa Inhibitor n GPIIb/IIIa Inhibitor Control Risk Reduction, % P PURSUIT [7] Eptifibatide NS PRISM-PLUS [6]* Tirofiban PARAGON-A [3] Lamifiban NS PARAGON-B [4] Lamifiban NS MI = myocardial infarction; NS = not significant. *Tirofiban/heparin versus heparin alone. 18 Relative risk reduction, P value 16 29%, %, 0.04 GPIIb/IIIa antagonist Placebo 14 32%, %, NS 7.8%, NS %, NS % 8 19%, CAPTURE GUSTO IV* PURSUIT PRISM-PLUS PRISM PARAGON-A PARAGON-B Figure 1. Results of the clinical trials examining the use of glycoprotein IIb/IIIa antagonists as medical therapy for patients presenting with unstable angina or non ST segment elevation MI at 30 days. NS = not significant. *48-hour abciximab versus placebo; High-dose eptifibatide versus placebo; Tirofiban/heparin versus heparin alone. 36 JCOM April 2001 Vol. 8, No. 4
5 included paradoxical platelet activation with high-dose lamifiban and possible passivation of the plaque at low-dose. Consequently, PARAGON-B was designed to confirm the benefit with low-dose lamifiban observed in PARAGON-A (Table 2) [4]. In this study, lamifiban was dosed according to patients creatinine clearance. The 30-day primary composite endpoint of death, MI, or recurrent ischemia was not different between the 2 groups (Figure 1). Marked benefit of lamifiban, however, was observed among 1160 patients who had elevated troponin T during hospitalization (30-day death or MI, 11% and 19% for lamifiban and placebo; P = 0.018), and in those who underwent early percutaneous revascularization during lamifiban infusion (11.6% versus 18.5%, P < 0.05). Nevertheless, since lamifiban did not achieve significant treatment effect in 2 clinical trials, it is not currently approved for clinical use. The use of tirofiban for acute coronary syndromes was studied in the PRISM and PRISM-PLUS trials [5,6]. Patients risk profiles differed in the 2 studies: ST segment changes were present in more than 90% of patients enrolled in PRISM-PLUS but in only 39% of patients in PRISM. In addition, tirofiban infusion was continued after percutaneous coronary intervention in PRISM-PLUS but was limited to 48 hours in PRISM, prior to angiography. In the PRISM trial, tirofiban demonstrated efficacy at 48 hours by lowering the primary composite endpoint of death, MI, or refractory ischemia by 33% (P = 0.01), and more marked benefit was observed in patients with electrocardiographic ischemia (ST segment depression or T wave inversion) at initial presentation. Moreover, mortality benefit was observed in the tirofiban group at 30 days (2.3% versus 3.6%, P = 0.02). Although the choice of subsequent treatment strategies (medical therapy, percutaneous coronary intervention, or coronary artery bypass graft [CABG] was not randomized, a reduced rate of death, MI, or refractory ischemia was observed with early percutaneous coronary intervention (21.6% versus 27.3%, P < 0.05) but not with medical therapy (10.0% versus 11.7%, P = not significant). No evidence of rebound phenomenon was observed after cessation of tirofiban infusion. PRISM-PLUS initially randomized acute coronary syndromes patients into 3 arms (Table 2). The tirofiban-alone arm was stopped prematurely after a 4.6% mortality rate was noted in that group at 7 days compared with 1.1% in the control group. The duration of infusion averaged about 3 days (71.3 ± 20 hours) and was continued for an additional 15.4 ± 8 hours in patients who underwent percutaneous coronary intervention. Reduction of death, MI, or refractory ischemia was observed with tirofiban/heparin therapy as early as 48 hours (5.7% versus 7.8%, P = 0.08), and at 7 days (12.9% versus 17.9%, P = 0.004), 30 days (18.5% versus 22.3%, P = 0.03) (Figure 1) and 6 months (27.7% versus 32.1%, P = 0.02) (Table 3) when compared with heparin alone. The benefit resulted mainly from the reduction in recurrent MI. Early divergence of the endpoints suggested the benefit of the drug was independent of subsequent revascularization therapy. Among 475 patients who subsequently underwent percutaneous coronary intervention, death, MI, or refractory ischemia was observed in 8.8% of the tirofiban group and 15.3% of the control group (risk ratio, 0.55; 95% confidence interval, 0.32 to 0.94). For patients who were managed with medical therapy alone, clinical outcome was slightly better with tirofiban/heparin combination therapy, although the difference did not reach statistical significance (composite endpoint at 30 days, 14.8% versus 16.8%; risk ratio, 0.87; 95% confidence interval, 0.60 to 1.25). The PURSUIT trial was the largest of the GPIIb/IIIa acute coronary syndrome trials [7]. The initial study protocol included 3 treatment arms: high-dose eptifibatide, low-dose eptifibatide, or placebo (Table 2). After demonstrating the safety in the high-dose eptifibatide arm, the low-dose treatment arm was discontinued according to the protocol. No restriction on the timing of revascularization was imposed. Reduction of death or MI was consistently observed in favor of eptifibatide at 96 hours (7.6% versus 9.1%, P = 0.01),7 days (10.1% versus 11.6%, P = 0.02), and 30 days (14.2% versus 15.7%, P = Percutaneous coronary intervention appeared to provide com plementary benefits to eptifibatide, since a greater reduction of the composite endpoint at 30 days was observed (11.6% versus 16.7%, P = 0.01). Patients who did not undergo revascularization procedures had a non-statistically significant reduction of the composite endpoint (14.5% versus 15.6%, P = 0.23), a finding similar to PARAGON B, PRISM, and PRISM-PLUS trials (Figure 2). In conclusion, there was a marked clinical benefit of the synthetic GPIIb/IIIa antagonists (eptifibatide, tirofiban, and lamifiban) in high-risk patients presenting with unstable angina or non ST segment elevation MI. Clinical benefits were most evident when GPIIb/IIIa antagonists were given to patients with ST segment changes (as in PRISM and PRISM-PLUS) or troponin elevation (PARAGON, PRISM- PLUS) and to those who were considered for percutaneous coronary intervention during drug infusion phase (PRISM, PRISM-PLUS, PURSUIT, PARAGON B) (Figure 2). Efficacy of Abciximab in Acute Coronary Syndromes The c7e3 Fab Antiplatelet Therapy in Unstable Refractory angina (CAPTURE) trial was designed primarily to examine the clinical efficacy of abciximab in acute coronary syndrome patients undergoing percutaneous coronary intervention [9]. A total of 1266 patients were randomized to either abciximab for 18 to 24 hours before and for 1 hour after percutaneous coronary intervention or placebo. Reduction in MI with abciximab was observed both during the medical stabilization phase (0.6% versus 2.1% in placebo, P = 0.029) and Vol. 8, No. 4 April 2001 JCOM 37
6 GLYCOPROTEIN IIb/IIIa BLOCKADE GPIIb/IIIa antagonist 18 Medical therapy alone Placebo With percutaneous coronary intervention Death/MI, % Death/MI, % PARAGON-B* PRISM PRISM-PLUS PURSUIT GUSTO IV 0 PARAGON-B* PRISM-PLUS PURSUIT CAPTURE Figure 2. Incidence of death or myocardial infarction (MI) at 30 days following the administration of glycoprotein IIb/IIIa inhibitors for acute coronary syndrome, stratified by strategy (invasive versus noninvasive). *Results include composite endpoint of death, MI, or refractory ischemia. within the first 24 hours after percutaneous coronary intervention (2.6% versus 5.5%, P = 0.009). At 30 days, the abciximab group had a 29% reduction in the composite endpoint of death, MI, or urgent intervention compared with placebo (11.3% versus 15.9%, P = 0.012) [9]. Further examination of the trial showed that the marked benefit of abciximab occurred in patients who had elevated troponin T levels at the time of presentation (6-month event rate, 9.5% and 23.9% for abciximab and placebo, P = 0.002), but not in those who had normal levels (9.4% versus 7.5%, P = 0.47), suggesting that abciximab reduced the risk of patients with elevated troponin T to that of patients with normal troponin T [14]. These findings highlight the important role of troponin T in the risk stratification of acute coronary syndromes and the clinical benefit of abciximab in the presence of thrombus reflected by elevated troponin T levels. In a pooled analysis of the CAPTURE (abciximab), PURSUIT (eptifibatide), and PRISM-PLUS (tirofiban) trials, GPIIb/IIIa blockade resulted in a significant reduction of death or MI during medical treatment (2.5% versus 3.8%; risk reduction, 34%; P < 0.001), and percutaneous coronary intervention further reduced the event rate by 41% within the first 48 hours after the procedure (4.9% versus 8.0%, P < 0.001) [15]. The consistent benefits of eptifibatide and tirofiban seen in the acute coronary syndromes trials prompted a study of abciximab as medical therapy for acute coronary syndromes. The main difference between GUSTO IV and the 5 P trials was that early catheterization was prohibited in GUSTO IV unless severe refractory ischemia occurred. In fact, less than 5% patients underwent percutaneous coronary intervention within 3 days after enrollment. The trial randomized 7800 patients who were believed to have acute coronary syndromes based on anginal symptom, plus elevation of troponin T or ischemic electrocardiographic changes. The study randomized patients to 3 arms: abciximab bolus plus 48-hour infusion, abciximab bolus plus 24-hour infusion, or placebo (Table 2). Aspirin and unfractionated heparin were given to all patients. At 30 days, the composite endpoint of death/mi was not different across the 3 treatment arms (48-hour abciximab, 9.1%; 24-hour abciximab, 8.2%; placebo, 8.0%; P = not significant). Paradoxically, a trend of increased death/mi with high-dose abciximab was present. The marked benefit demonstrated consistently in the troponin-positive patients in the other GPIIb/IIIa inhibitor acute cornary syndrome trials was not observed among those patients given abciximab (death/mi at 30 days: 11.7% for 48-hour abciximab, 10.2% for 24-hour abciximab, and 9.7% for placebo). When the results of all GPIIb/IIIa antagonist acute coronary syndromes trials are pooled, GPIIb/IIIa inhibition still confers a significant 9% reduction in death or MI at 30 days (P = 0.04) [16]. Postulated explanations for the negative results from this trial include enrollment of a relatively low-risk population, identified by positive troponin alone without electrocardiographic manifestations of ischemia, uncertainty regarding appropriate dosing, and the possibility of untoward toxic 38 JCOM April 2001 Vol. 8, No. 4
7 effect with prolonged platelet inhibition with abciximab. The difference in efficacy of abciximab in GUSTO IV and in CAPTURE trial underscore the complementary role of percutaneous coronary intervention during GPIIb/IIIa inhibitor administration a finding that has been noted when comparing the patients who were treated with percutaneous coronary intervention or medical therapy in the 5 P trials (Figure 2). Choice of GPIIb/IIIa Inhibitor in Candidates for Percutaneous Coronary Intervention Two recently completed studies further complicated the choice of GPIIb/IIIa antagonists in acute coronary syndrome patients who are candidates for percutaneous coronary intervention. The first was the Treat angina with Aggrastat and determine Cost of Therapy with an Invasive or Conservative Strategy (TACTICS-TIMI 18) trial, which demonstrated the superiority of an early invasive strategy over a conservative strategy in patients with acute coronary syndromes (6-month death, MI, or rehospitalization for acute coronary syndromes, 10.4% versus 15.9%; P = 0.025) [17]. Tirofiban was given to all patients in this study. Patients who were troponin-positive had even more significant reduction in the primary endpoint with early aggressive therapy (14% versus 24%; risk reduction, 42%). The second trial was the Do Tirofiban and ReoPro Give similar Efficacy Trial (TARGET). This trial concluded that abciximab administration during elective or urgent percutaneous coronary intervention resulted in 26% less incidence of death/mi/ refractory angina at 30 days compared with tirofiban [18]. Among the acute coronary syndromes subgroup, the benefit of abciximab was even more substantial (6.3% versus 9.3%; risk reduction, 32%). The outcome of the 3 trials (GUSTO IV, TARGET, and TACTICS-TIMI 18) provided more questions than answers regarding choice of GPIIb/IIIa inhibitor in the management of acute coronary syndromes. The question of which agent should be given and when it should be started has recently been debated. Based on the results of these trials, together with PRISM and PRISM-PLUS, medium- or high-risk acute coronary syndrome patients should be started on tirofiban while being triaged for early catheterization within 48 hours, followed by revascularization by either percutaneous coronary intervention or CABG. If angiography procedures can be arranged within the first few hours of admission, GPIIb/IIIa inhibitor administration could probably be postponed and abciximab should be started in the catheterization laboratory once percutaneous coronary intervention is undertaken, based on the superior results of abciximab compared to tirofiban in TARGET. Another issue, which has never been studied in clinical trials, is whether tirofiban or eptifibatide started before patient arrival to the catheterization laboratory could or should be switched to abciximab at the time of percutaneous coronary intervention. We believe that although rebound phenomenon has not been seen after discontinuation of eptifibatide or tirofiban infusion, the risk of bleeding related to switching from one GPIIb/IIIa inhibitor to another is uncertain and this practice is currently not recommended without appropriate trials addressing this issue. Combination of Low-Molecular-Weight Heparin and GPIIb/IIIa Inhibitors While unfractionated heparin inhibits thrombin formation by binding to antithrombin III, low-molecular-weight heparin such as enoxaparin or dalteparin may be more effective in blocking the coagulation cascade by inhibiting factor Xa in addition to its antithrombin activity. Lowmolecular-weight heparin binds less avidly to plasma and tissue proteins and thus has higher bioavailability and a more predictable therapeutic effect. Large clinical trials have demonstrated its superior efficacy in the setting of unstable angina and non ST segment elevation MI compared to unfractionated heparin [19 21]. The National Investigators Collaborating on Enoxaparin (NICE)-3 was an observational study demonstrating the feasibility of combining enoxaparin and GPIIb/IIIa inhibitors for acute coronary syndrome patients who subsequently underwent percutaneous coronary intervention [22]. The GUSTO IV dalteparin substudy showed the safety, and a trend towards improved efficacy, for the combination of dalteparin and abciximab for medical management of acute coronary syndromes patients [23]. Furthermore, the NICE-4 trial demonstrated the safety of enoxaparin and abciximab combination therapy in the percutaneous coronary intervention setting [24,25]. The Antithrombotic Combination Using Tirofiban and Enoxaparin (ACUTE)-II trial, which randomized 525 acute coronary syndromes patients to tirofiban/enoxaparin or tirofiban/unfractionated heparin, showed no significant difference in the primary endpoint of bleeding and a trend toward reduced cardiac events with enoxaparin at 30 days [26]. Since increasingly more acute coronary syndromes patients are started on the combination of low-molecularweight heparin and GPIIb/IIIa inhibitors during initial hospitalization, current research is focused on the dose regimen and the choices of low-molecular weight heparin and GPIIb/IIIa inhibitor to facilitate a smooth transition and continuation of anticoagulation therapy from medical stabilization to revascularization for acute coronary syndrome patients [27]. Safety of GPIIb/IIIa Blockade Bleeding. A trend towards increased bleeding and a greater need for blood transfusion is observed in the active treatment Vol. 8, No. 4 April 2001 JCOM 39
8 GLYCOPROTEIN IIb/IIIa BLOCKADE Table 4. Safety Profile of Glycoprotein IIb/IIIa Antagonists for Acute Coronary Syndromes in the Clinical Trials Major Bleeding, %* Blood Transfusion, % Thrombocytopenia, % Agent Trial Treatment Control Treatment Control Treatment Control Abciximab CAPTURE [9] GUSTO IV [8] N/A N/A Eptifibatide PURSUIT [7] N/A N/A 0.2 < 0.1 Tirofiban PRISM [5] PRISM-PLUS [6] Lamifiban PARAGON-A [3] PARAGON-B [4] N/A N/A N/A N/A N/A = not available *Defined by > 5.0 g/dl decrease in the blood hemoglobin level, the need for transfusion of 2 units of blood, the need for corrective surgery, the occurrence of an intracranial or retroperitoneal hemorrhage, or a combination of these events. Defined by a decrease in platelet count to 90,000/mm3. arms across all the GPIIb/IIIa trials (Table 4). Fortunately, the incidence was only modestly increased, and bleeding mostly occurred at the femoral access sites of the patients who underwent cardiac catheterization. Considering the trade-off in terms of reduction of the risk of death or MI, the increased risk of bleeding was acceptable. For patients who need to undergo emergency or urgent CABG surgery, considerable concern about postoperative bleeding caused by GPIIb/IIIa inhibitor infusion has been raised. Insight regarding the effect of GPIIb/IIIa inhibition on the safety of CABG was gained from the PURSUIT trial [28]. In this trial, 1558 patients (14.2% of the total population) required emergency or urgent CABG surgery, of which 866 patients (56%) received eptifibatide for a median of 71.8 hours prior to CABG (interquartile range, 36 to 72 hours). Time of drug discontinuation to CABG in the eptifibatide group was 66.5 hours (median; interquartile range, 18 to 184 hours), which was similar to the control group (median, 57.8 hours; P = 0.2). The subsequent risks of major (eptifibatide, 58.2%; placebo, 56.6%) or minor (eptifibatide, 23.5%; placebo, 23.6%) bleeding were not different between the 2 groups. Only 1 intracranial hemorrhage occurred in the study population. Patients who received eptifibatide had 16% less postoperative death or MI at 6-month follow-up. An additional 20% reduction in postoperative death or MI was evident when eptifibatide was given within 24 hours prior to CABG. The rapid reversibility (2 to 4 hours) of eptifibatide explains the absence of increased bleeding after CABG compared with placebo. Analysis of the EPIC patients who required emergency or urgent CABG (56 patients [2.8%], with 46 patients undergoing surgery within 24 hours) indicated that the more prolonged biologic half-life of abciximab was a concern for this group of patients [29]. Comparing abiciximab with placebo, the mortality rate was higher (23.5% versus 8.0% in placebo) as was blood product requirement (76.5% versus 64.0% for packed red blood cells and 76.5% versus 56.0% for platelets). Weightadjusted heparin doses were used in the subsequent EPILOG and EPISTENT trials. Among 42 patients who underwent emergency CABG (abciximab was discontinued within 6 hours of operation in 50% to 60% of patients), there were no significant differences in 30-day clinical outcome (death or MI, 32% and 45% for abciximab and placebo; P = not significant), need of surgical exploration (12% and 3% for abciximab and placebo, P = 0.21), or packed red blood cell transfusion (57% and 50% for abciximab and placebo, P = not significant) between the comparison groups [30]. Renal insufficiency. Eptifibatide and tirofiban are cleared by renal excretion, while abciximab is cleared by the reticuloendothelial system. Patients with renal insufficiency were not well-represented in the clinical trials. Patients with a serum creatinine value greater than 2.0 to 2.5 mg/dl were excluded from PURSUIT, PRISM, and PRISM-PLUS. The dosage of tirofiban should be half of the normal in the presence of renal insufficiency. A reduced dose of eptifibatide (135 µg/kg bolus µg/kg/min) was given to patients with serum creatinine levels between 2 and 4 mg/dl in the percutaneous coronary intervention setting [31], but this dose regimen has not been tried in medical management for acute coronary syndromes in which the infusion period is usually much longer. Both tirofiban and eptifibatide are dialyzable, but this modality is rarely required because of their relatively short half-lives. Abciximab is not currently recommended for patients on dialysis. Thrombocytopenia. Across all the GPIIb/IIIa inhibitor trials, thrombocytopenia occurred slightly more often in the active 40 JCOM April 2001 Vol. 8, No. 4
9 Unstable angina, non ST segment elevation MI Medical stabilization with aspirin, UFH or LMWH, β blocker, ± statins, nitrates Medium- or high-risk profile: age > 75, rest angina, pulmonary edema, CKMB or troponin elevation, transient ST-T changes, LV dysfunction ABSENT Further risk stratification with noninvasive studies PRESENT Catheterization within 4 48 hours Catheterization < 4 hours Tirofiban or eptifibatide bolus and infusion Cardiac catheterization Medical therapy PCI indicated: start abciximab in the lab, followed by 12-hour infusion post-pci CABG PCI, continue tirofiban or eptifabitide infusion for hours CABG; stop tirofiban or eptifibatide at least 4 6 hours prior to surgery Figure 3. Proposed critical pathway for the management of unstable angina or non ST segment elevation MI based on contemporary clinical trials. CABG = coronary artery bypass graft; CKMB= creatine kinase MB; LMWH = low-molecular-weight heparin; LV = left ventricular; MI = myocardial infarction; PCI = percutaneous coronary intervention; UFH = unfractionated heparin. arm (Table 4). The incidence of severe thrombocytopenia (< 50,000 platelets/mm 3 ) was 0.2% in eptifibatide-treated patients (PURSUIT) and 0.4% to 0.5% in tirofiban-treated patients (PRISM, PRISM-PLUS). Although these figures were higher than those in the control groups (0.1% to 0.2%), the absolute number of patients was still small. Abciximab, however, was associated with a higher incidence of thrombocytopenia (0.9% to 1.8% in EPIC, EPILOG, and CAPTURE), and the association increased with the dosage of heparin [32]. Thrombocytopenia during readministration of abciximab, an immunologic phenomenon related to IgG antibodies, was reported to be 2.4% to 4.0% in observational registries [33,34], and the incidence increased with proximity to the previous administration. Efficacy of abciximab did not change during readministration. Our current recommendation is to check platelet count at 2 to 4 hours after beginning the abciximab infusion and repeat in 12 to 24 hours, regardless of a history of prior use. Neither eptifibatide nor tirofiban have been Vol. 8, No. 4 April 2001 JCOM 41
10 GLYCOPROTEIN IIb/IIIa BLOCKADE Table 5. Summary of the Trials of Oral Glycoprotein IIb/IIIa Inhibitors for Acute Coronary Syndromes Mortality, %* MI, %* GPIIb/IIIa GPIIb/IIIa Aspirin GPIIb/IIIa Aspirin Trial Inhibitor n Inhibitor Alone P Inhibitor Alone P OPUS-TIMI 16 Orobofiban [12] 10, SYMPHONY Sibrafiban [10] nd SYMPHONY Sibrafiban [11] BRAVO Lotrafiban [13] N/A N/A MI = myocardial infarction; N/A = not available. *Endpoints at 30 days and beyond. Low-dose sibrafiban + aspirin versus high-dose sibrafiban alone versus aspirin alone. reported to cause immunologic reaction, but it is prudent to check platelet count 6 to 24 hours after therapy initiation. Oral GPIIb/IIIa Blockade for Acute Coronary Syndromes In view of the remarkable benefit of intravenous GPIIb/IIIa agents in acute coronary syndromes and percutaneous coronary intervention, it was hypothesized that moderate chronic inhibition of platelet aggregation with oral agents may further reduce major adverse coronary events. Three different oral agents were tested in 4 trials among 35,294 patients [10 13,35]. Despite the diversity of the patient population and inherent differences in the pharmacokinetics of the drugs, each of the 4 trials strikingly demonstrated lower efficacy (higher mortality) and safety after 30 days of intake (Table 5). All except for the first SYMPHONY trial were terminated prematurely due to increased mortality observed during interim analyses. However, the incidence of MI and urgent revascularization were not different between the comparison groups in any of these trials. The disparity between the increased mortality and neutral effect on MI or revascularization (attributable to a prothrombotic effect) suggests untoward mechanism(s) unrelated to a prothrombotic effect, such as (1) inadequate platelet inhibition (only 30% to 50% platelet inhibition could be achieved by oral therapy) and partial activation of platelet [36,37]; (2) proinflammatory response as a result of release of CD-40 ligand and activation of P-selectin [37,38]; and (3) induction of myocytic apoptosis, resulting in arrhythmias [39,40]. The disappointing results from these trials call for a reexamination of the role of oral GPIIb/IIIa inhibitors in acute coronary syndromes. Economic Considerations In view of the efficacy of GPIIb/IIIa inhibitors for acute coronary syndromes patients demonstrated in CAPTURE, PURSUIT, PRISM, and PRISM-PLUS trials, economic consideration has been the major issue with respect to the choice of agents. The cost savings derived from the reduction of ischemic events may offset the cost of acquisition of these agents. The incremental cost-effectiveness ratio of abciximab for patients undergoing percutaneous coronary intervention for acute coronary syndromes was estimated to be $4000 to $7000 per life-year saved [41]. For eptifibatide, this ratio was $16,491 per year of life saved based on U.S. PURSUIT patients [42]. These data are not comparable since many of the eptifibatide patients in PURSUIT did not undergo percutaneous coronary intervention. However, the reasonable cost-effectiveness ratios suggest that empirical use of GPIIb/IIIa inhibitors for unstable angina or non ST segment elevation MI patients is attractive compared with some other conventional therapies ($19,000 for hypertension, $26,000 for CABG, $32,000 for tissue plasminogen activator in patients with acute MI, and $35,000 for hemodialysis) [43]. Conclusion The current standard of therapy for unstable angina or non ST segment elevation MI should include tirofiban or eptifibatide administration, beginning soon after hospitalization. Since most of the benefits shown in the clinical trials were derived from the complementary usage of percutaneous coronary intervention and intravenous GPIIb/IIIa antagonists, all but low-risk patients should undergo early cardiac catheterization for further risk stratification and possible revascularization while receiving GPIIb/IIIa inhibitor infusion (Figure 3). Abciximab should be reserved for patients who are taken immediately to the catheterization laboratory and have not been treated with other agents. There is currently no role for oral GPIIb/IIIa inhibition for acute coronary syndromes until the pathophysiologic mechanisms responsible for increased mortality are further elucidated. The combination of low-molecular-weight heparin and GPIIb/IIIa inhibitor is currently being investigated in phase III clinical trials. 42 JCOM April 2001 Vol. 8, No. 4
11 Target Vessel Revascularization, %* Major Bleeding, % GPIIb/IIIa Aspirin GPIIb/IIIa Aspirin Inhibitor Alone P Inhibitor Alone P < < < N/A N/A < Corresponding author: Sorin J. Brener, MD, Dept. of Cardiology, The Cleveland Clinic Foundation, 9500 Euclid Avenue, Desk F25, Cleveland, OH 44195, breners@ccf.org. Financial disclosures: The authors disclosed no financial relationships. Author contributions: conception and design, AWC, SJB; analysis and interpretation of data, AWC, SJB; drafting of the article, AWC; critical revision of the article, AWC, SJB; final approval of the article, AWC, SJB; statistical experience, AWC; administrative, technical, or logistic support, SJB; collection and assembly of data, AWC. References 1. Myocardial infarction redefined a consensus document of The Joint European Society of Cardiology/American College of Cardiology Committee for the redefinition of myocardial infarction. J Am Coll Cardiol 2000;36: Braunwald E, Antman EM, Beasley JW, et al. ACC/AHA guidelines for the management of patients with unstable angina and non-st-segment elevation myocardial infarction. A report of the American College of Cardiology/ American Heart Association Task Force on Practice Guidelines (Committee on the Management of Patients With Unstable Angina). J Am Coll Cardiol 2000;36: International, randomized, controlled trial of lamifiban (a platelet glycoprotein IIb/IIIa inhibitor), heparin, or both in unstable angina. The PARAGON Investigators. Platelet IIb/IIIa Antagonism for the Reduction of Acute coronary syndrome events in a Global Organization Network. Circulation 1998;97: Harrington R. Optimal dosing of a platelet glycoprotein antagonist, lamifiban, using renal-based algorithms, in patients with acute coronary syndromes. The Second Platelet IIb/IIIa Antagonism of the Reduction of Acute coronary syndrome events in a Global Organization Network (PARAGON B) [abstract]. Presented at the 49th Annual Scientific Sessions of the American College of Cardiology; 2000 Mar 12 15; Anaheim, CA. Available at org/education/online/trials/acc2000/paragonb.htm. Accessed 27 Mar A comparison of aspirin plus tirofiban with aspirin plus heparin for unstable angina. Platelet Receptor Inhibition in Ischemic Syndrome Management (PRISM) Study Investigators. N Engl J Med 1998;338: Inhibition of the platelet glycoprotein IIb/IIIa receptor with tirofiban in unstable angina and non-q-wave myocardial infarction. Platelet Receptor Inhibition in Ischemic Syndrome Management in Patients Limited by Unstable Signs and Symptoms (PRISM-PLUS) Study Investigators [published erratum appears in N Engl J Med 1998;339:415]. N Engl J Med 1998;338: Inhibition of platelet glycoprotein IIb/IIIa with eptifibatide in patients with acute coronary syndromes. The PURSUIT Trial Investigators. Platelet Glycoprotein IIb/IIIa in Unstable Angina: Receptor Suppression Using Integrilin Therapy. N Engl J Med 1998;339: Simoons M. GUSTO IV: clinical trial results [abstract]. Presented at the 22nd Congress of European Cardiology Society; 2000 Aug 26 30; Amsterdam, Netherlands. Available at hotlines.htm. Accessed 27 Mar Randomised placebo-controlled trial of abciximab before and during coronary intervention in refractory unstable angina: the CAPTURE Study [published erratum appears in Lancet 1997;350:744]. Lancet 1997;349: Comparison of sibrafiban with aspirin for prevention of cardiovascular events after acute coronary syndromes: a randomised trial. The SYMPHONY Investigators. Sibrafiban versus Aspirin to Yield Maximum Protection from Ischemic Heart Events Postacute Coronary Syndromes. Lancet 2000;355: Newby LK. A randomized comparison of sibrafiban, an oral glycoprotein (GP) IIb/IIIa receptor antagonist, with and without aspirin versus aspirin after acute coronary syndromes: results of the second SYMPHONY trial. Sibrafiban versus Aspiran to Yield Maximum Protection from Ischemic Heart Events Post Acute Coronary Syndromes (2nd SYMPHONY) [abstract]. Presented at the 49th Annual Scientific Session of the American College of Cardiology; 2000 Mar 12 15; Anaheim, CA. Available at education/online/trials/acc2000/symphony2.htm. Accessed 27 Mar Cannon CP, McCabe CH, Wilcox RG, et al. Oral glycoprotein IIb/IIIa inhibition with orbofiban in patients with unstable coronary syndromes (OPUS-TIMI 16) trial. Circulation 2000; 102: Topol EJ, Easton JD, Amarenco P, et al. Design of the blockade of the glycoprotein IIb/IIIa receptor to avoid vascular occlusion (BRAVO) trial. Am Heart J 2000;139: Hamm CW, Heeschen C, Goldmann B, et al. Benefit of abciximab in patients with refractory unstable angina in relation to serum troponin T levels. c7e3 Fab Antiplatelet Therapy in Unstable Refractory Angina (CAPTURE) Study Investigators [published erratum appears in N Engl J Med 1999;341: 548]. N Engl J Med 1999;340: Boersma E, Akkerhuis KM, Theroux P, et al. Platelet glycoprotein IIb/IIIa receptor inhibition in non-st-elevation acute coronary syndromes: early benefit during medical treatment only, with additional protection during percutaneous coronary intervention. Circulation 1999;100: Vol. 8, No. 4 April 2001 JCOM 43
12 GLYCOPROTEIN IIb/IIIa BLOCKADE 16. Bhatt DL, Topol EJ. Current role of platelet glycoprotein IIb/IIIa inhibitors in acute coronary syndromes. JAMA 2000; 284: Cannon CP, Weintraub WS, Demopoulos L, et al. Results of the treat angina with aggrastat and determine cost of therapy with an invasive or conservative strategy (TACTICS- TIMI 18) trial: a comparison of invasive versus conservative strategy in patients with unstable angina and non-st segment elevation myocardial infarction. A comparison of invasive versus conservative strategy in patients with unstable angina and non-st segment elevation myocardial infarction [abstract]. Presented at the American Heart Association Scientific Session; 2000 Nov 12 15; New Orleans, LA. Available at abstracts/latebreakingclinicaltrials.oft. Accessed 27 March Topol EJ. Do tirofiban and ReoPro give similar efficacy trial (TARGET) [abstract]? Presented at the American Heart Association Scientific Session 2000; 2000 Nov 12 15; New Orleans, LA. Available at abstracts/latebreakingclinicaltrials.oft. Accessed 27 Mar Antman EM, Cohen M, Radley D, et al. Assessment of the treatment effect of enoxaparin for unstable angina/non-qwave myocardial infarction. TIMI 11B-ESSENCE metaanalysis. Circulation 1999;100: Cohen M, Demers C, Gurfinkel EP, et al. A comparison of low-molecular-weight heparin with unfractionated heparin for unstable coronary artery disease. Efficacy and Safety of Subcutaneous Enoxaparin in Non-Q-Wave Coronary Events Study Group. N Engl J Med 1997;337: Klein W, Buchwald A, Hillis SE, et al. Comparison of lowmolecular-weight heparin with unfractionated heparin acutely and with placebo for 6 weeks in the management of unstable coronary artery disease. Fragmin in unstable coronary artery disease study (FRIC) [published erratum appears in Circulation 1998;97:413]. Circulation 1997;96: Ferguson J. The National Investigators Collaboration on Enoxaparin (NICE) 3 study [abstract]. Presented at the 22nd Congress of European Cardiology Society; 2000 Aug 26 30; Amsterdam, Netherlands. Available at org. Accessed 27 Mar Wallentin L. Global Use of Strategies to open Totally Occluded arteries (GUSTO) IV dalteparin substudy [abstract]. Presented at the 22nd Congress of European Cardiology Society; 2000 Aug 26 30; Amsterdam, Netherlands. Available at hotlines.htm. Accessed 27 Mar Kereiakes DJ, Fry E, Matthai W, et al. Combination enoxaparin and abciximab therapy during percutaneous coronary intervention: NICE guys finish first. J Invasive Cardiol 2000;12 Suppl A:1A 5A. 25. Tramuta DA, Kereiakes DJ, Dippel EJ, et al. Combination enoxaparin-abciximab therapy during coronary intervention: the next standard of care? J Invasive Cardiol 2000;12 Suppl C:3C 6C. 26. Cohen M, Theroux P, Frey MJ, et al. Antithrombotic combination using tirofiban and enoxaparin: the (ACUTE)-II study [abstract]. Presented at the American Heart Association Scientific Session 2000; 2000 Nov 12 15; New Orleans, LA. Accessed 27 Mar Kereiakes DJ, Grines C, Fry E, et al. Combination enoxaparin and abciximab during percutaneous coronary intervention: A new standard of care? Curr Interv Cardiol Rep 2000;2: Marso SP, Bhatt DL, Roe MT, et al. Enhanced efficacy of eptifibatide administration in patients with acute coronary syndrome requiring in-hospital coronary artery bypass grafting. PURSUIT Investigators. Circulation 2000;102: Boehrer JD, Kereiakes DJ, Navetta FI, et al. Effects of profound platelet inhibition with c7e3 before coronary angioplasty on complications of coronary bypass surgery. EPIC Investigators. Evaluation Prevention of Ischemic Complications. Am J Cardiol 1994;74: Lincoff AM, LeNarz LA, Despotis GJ, et al. Abciximab and bleeding during coronary surgery: results from the EPILOG and EPISTENT trials. Improve Long-term Outcome with abciximab GP IIb/IIIa blockade. Evaluation of Platelet IIb/IIIa Inhibition in STENTing. Ann Thorac Surg 2000;70: Randomised placebo-controlled trial of effect of eptifibatide on complications of percutaneous coronary intervention: IMPACT-II. Integrilin to Minimise Platelet Aggregation and Coronary Thrombosis-II. Lancet 1997;349: Tcheng JE. Clinical challenges of platelet glycoprotein IIb/IIIa receptor inhibitor therapy: bleeding, reversal, thrombocytopenia, and retreatment. Am Heart J 2000;139(2 Pt 2):S Tcheng JE, Kereiakes DJ, Braden GA, et al. Readministration of abciximab: interim report of the ReoPro readministration registry. Am Heart J 1999;138(1 Pt 2):S Madan M, Kereiakes DJ, Hermiller JB, et al. Efficacy of abciximab readministration in coronary intervention. Am J Cardiol 2000;85: Chew DP, Bhatt DL, Sapp S, Topol EJ. Increased mortality with oral platelet glycoprotein IIb/IIIa antagonists: a metaanalysis of phase III multicenter randomized trials. Circulation 2001;103: Coulter S, Cannon CP, Cooper R, et al. Thrombocytopenia, bleeding and thrombotic events with oral glycoprotein IIb/IIIa inhibition. Results from OPUS TIMI 16. J Am Coll Cardiol 2000;35 (2 Suppl 1):393A. 37. Peter K, Schwarz M, Ylanne J, et al. Induction of fibrinogen binding and platelet aggregation as a potential intrinsic property of various glycoprotein IIb/IIIa (alphaiibbeta3) inhibitors. Blood 1998;92: Holmes MB, Sobel BE, Cannon CP, Schneider DJ. Increased platelet reactivity in patients given orbofiban after an acute coronary syndrome: an OPUS-TIMI 16 substudy. Am J Cardiol 2000;85: Buckley CD, Pilling D, Henriquez NV, et al. RGD peptides induce apoptosis by direct caspase 3 activation. Nature 1999;397: JCOM April 2001 Vol. 8, No. 4
Platelet glycoprotein IIb/IIIa inhibition in acute coronary syndromes
 European Heart Journal (00) 3, 1441 1448 doi:10.1053/euhj.00.3160, available online at http://www.idealibrary.com on Platelet glycoprotein IIb/IIIa inhibition in acute coronary syndromes Gradient of benefit
European Heart Journal (00) 3, 1441 1448 doi:10.1053/euhj.00.3160, available online at http://www.idealibrary.com on Platelet glycoprotein IIb/IIIa inhibition in acute coronary syndromes Gradient of benefit
QUT Digital Repository:
 QUT Digital Repository: http://eprints.qut.edu.au/ This is the author s version of this journal article. Published as: Doggrell, Sheila (2010) New drugs for the treatment of coronary artery syndromes.
QUT Digital Repository: http://eprints.qut.edu.au/ This is the author s version of this journal article. Published as: Doggrell, Sheila (2010) New drugs for the treatment of coronary artery syndromes.
An update on the management of UA / NSTEMI. Michael H. Crawford, MD
 An update on the management of UA / NSTEMI Michael H. Crawford, MD New ACC/AHA Guidelines 2007 What s s new in the last 5 years CT imaging advances Ascendancy of troponin and BNP Clarification of ACEI/ARB
An update on the management of UA / NSTEMI Michael H. Crawford, MD New ACC/AHA Guidelines 2007 What s s new in the last 5 years CT imaging advances Ascendancy of troponin and BNP Clarification of ACEI/ARB
ACUTE CORONARY SYNDROMES
 CLINICAL CARDIOLOGY Current Role of Platelet Glycoprotein IIb/IIIa Inhibitors in Acute Coronary Syndromes Deepak L. Bhatt, MD Eric J. Topol, MD ACUTE CORONARY SYNDROMES (ACSs) span the pathological continuum
CLINICAL CARDIOLOGY Current Role of Platelet Glycoprotein IIb/IIIa Inhibitors in Acute Coronary Syndromes Deepak L. Bhatt, MD Eric J. Topol, MD ACUTE CORONARY SYNDROMES (ACSs) span the pathological continuum
Chapter 15 Glycoprotein IIb/IIIa Antagonists
 Chapter 15 Glycoprotein IIb/IIIa Antagonists Introduction - GP IIb/IIIa receptors - Pharmacologic approaches - Preparations and dosages Mechanism of Action Clinical Efficacy - UA / NSTEMI - STEMI - PCI
Chapter 15 Glycoprotein IIb/IIIa Antagonists Introduction - GP IIb/IIIa receptors - Pharmacologic approaches - Preparations and dosages Mechanism of Action Clinical Efficacy - UA / NSTEMI - STEMI - PCI
Collectively, the efficacy of the intravenous glycoprotein
 Increased Mortality With Oral Platelet Glycoprotein IIb/IIIa Antagonists A Meta-Analysis of Phase III Multicenter Randomized Trials Derek P. Chew, MBBS; Deepak L. Bhatt, MD; Shelly Sapp, MS; Eric J. Topol,
Increased Mortality With Oral Platelet Glycoprotein IIb/IIIa Antagonists A Meta-Analysis of Phase III Multicenter Randomized Trials Derek P. Chew, MBBS; Deepak L. Bhatt, MD; Shelly Sapp, MS; Eric J. Topol,
Acute coronary syndrome (ACS) is an
 OVERVIEW OF MEDICAL MANAGEMENT OF ACUTE CORONARY SYNDROMES Robert B. Parker, PharmD * Acute coronary syndrome (ACS) is an umbrella term used to describe any group of symptoms of acute myocardial ischemia
OVERVIEW OF MEDICAL MANAGEMENT OF ACUTE CORONARY SYNDROMES Robert B. Parker, PharmD * Acute coronary syndrome (ACS) is an umbrella term used to describe any group of symptoms of acute myocardial ischemia
Otamixaban for non-st-segment elevation acute coronary syndrome
 Otamixaban for non-st-segment elevation acute coronary syndrome September 2011 This technology summary is based on information available at the time of research and a limited literature search. It is not
Otamixaban for non-st-segment elevation acute coronary syndrome September 2011 This technology summary is based on information available at the time of research and a limited literature search. It is not
2003 by the American College of Cardiology Foundation ISSN /03/$30.00
 Journal of the American College of Cardiology Vol. 41, No. 4 Suppl S 2003 by the American College of Cardiology Foundation ISSN 0735-1097/03/$30.00 Published by Elsevier Science Inc. doi:10.1016/s0735-1097(02)02963-7
Journal of the American College of Cardiology Vol. 41, No. 4 Suppl S 2003 by the American College of Cardiology Foundation ISSN 0735-1097/03/$30.00 Published by Elsevier Science Inc. doi:10.1016/s0735-1097(02)02963-7
Update on Antithrombotic Therapy in Acute Coronary Syndrome
 Update on Antithrombotic Therapy in Acute Coronary Syndrome Laura Tsang November 13, 2006 Objectives: By the end of this session, you should understand: The role of antithrombotics in ACS Their mechanisms
Update on Antithrombotic Therapy in Acute Coronary Syndrome Laura Tsang November 13, 2006 Objectives: By the end of this session, you should understand: The role of antithrombotics in ACS Their mechanisms
THE ECONOMICS OF ADJUNCTIVE THERAPIES IN CORONARY ANGIOPLASTY: DRUGS, DEVICES, OR BOTH?
 THE ECONOMICS OF ADJUNCTIVE THERAPIES IN CORONARY ANGIOPLASTY: DRUGS, DEVICES, OR BOTH? Paul I. Oh, 1 Eric A. Cohen, 2 Nicole Mittmann, 3, 4 Soo Jin Seung 4 1 Division of Clinical Pharmacology, Sunnybrook
THE ECONOMICS OF ADJUNCTIVE THERAPIES IN CORONARY ANGIOPLASTY: DRUGS, DEVICES, OR BOTH? Paul I. Oh, 1 Eric A. Cohen, 2 Nicole Mittmann, 3, 4 Soo Jin Seung 4 1 Division of Clinical Pharmacology, Sunnybrook
NOVEL ANTI-THROMBOTIC THERAPIES FOR ACUTE CORONARY SYNDROME: DIRECT THROMBIN INHIBITORS
 Judd E. Hollander, MD Professor, Clinical Research Director, Department of Emergency Medicine University of Pennsylvania School of Medicine, Philadelphia, Pennsylvania OBJECTIVES: 1. Discuss the concept
Judd E. Hollander, MD Professor, Clinical Research Director, Department of Emergency Medicine University of Pennsylvania School of Medicine, Philadelphia, Pennsylvania OBJECTIVES: 1. Discuss the concept
Cangrelor: Is it the new CHAMPION for PCI? Robert Barcelona, PharmD, BCPS Clinical Pharmacy Specialist, Cardiac Intensive Care Unit November 13, 2015
 Cangrelor: Is it the new CHAMPION for PCI? Robert Barcelona, PharmD, BCPS Clinical Pharmacy Specialist, Cardiac Intensive Care Unit November 13, 2015 Objectives Review the pharmacology and pharmacokinetic
Cangrelor: Is it the new CHAMPION for PCI? Robert Barcelona, PharmD, BCPS Clinical Pharmacy Specialist, Cardiac Intensive Care Unit November 13, 2015 Objectives Review the pharmacology and pharmacokinetic
Antiplatelet and Antithrombotic Therapies in PCI Defining the Optimal Strategy
 Antiplatelet and Antithrombotic Therapies in PCI Defining the Optimal Strategy Franz-Josef Neumann Herz-Zentrum Bad Krozingen Antiplatelet and Antithrombotic Therapies in PCI Defining the Optimal Strategy
Antiplatelet and Antithrombotic Therapies in PCI Defining the Optimal Strategy Franz-Josef Neumann Herz-Zentrum Bad Krozingen Antiplatelet and Antithrombotic Therapies in PCI Defining the Optimal Strategy
FACTOR Xa AND PAR-1 BLOCKER : ATLAS-2, APPRAISE-2 & TRACER TRIALS
 New Horizons In Atherothrombosis Treatment 2012 순환기춘계학술대회 FACTOR Xa AND PAR-1 BLOCKER : ATLAS-2, APPRAISE-2 & TRACER TRIALS Division of Cardiology, Jeonbuk National University Medical School Jei Keon Chae,
New Horizons In Atherothrombosis Treatment 2012 순환기춘계학술대회 FACTOR Xa AND PAR-1 BLOCKER : ATLAS-2, APPRAISE-2 & TRACER TRIALS Division of Cardiology, Jeonbuk National University Medical School Jei Keon Chae,
Glycoprotein (GP) IIb/IIIa antagonists are potent inhibitors
 Current Perspective Platelet Glycoprotein IIb/IIIa Inhibitors Recognition of a Two-Edged Sword? Martin J. Quinn, MD, PhD; Edward F. Plow, PhD; Eric J. Topol, MD Glycoprotein (GP) IIb/IIIa antagonists are
Current Perspective Platelet Glycoprotein IIb/IIIa Inhibitors Recognition of a Two-Edged Sword? Martin J. Quinn, MD, PhD; Edward F. Plow, PhD; Eric J. Topol, MD Glycoprotein (GP) IIb/IIIa antagonists are
Manual of Interventional Cardiology
 782 PLATELET GLYCOPROTEIN IIb/IIIa RECEPTOR ANTAGONISTS (Tables 34.15, 34.16) Activation of the platelet glycoprotein (GP) IIb/IIIa receptor complex constitutes the final common pathway for platelet aggregation,
782 PLATELET GLYCOPROTEIN IIb/IIIa RECEPTOR ANTAGONISTS (Tables 34.15, 34.16) Activation of the platelet glycoprotein (GP) IIb/IIIa receptor complex constitutes the final common pathway for platelet aggregation,
Appendix: ACC/AHA and ESC practice guidelines
 Appendix: ACC/AHA and ESC practice guidelines Definitions for guideline recommendations and level of evidence Recommendation Class I Class IIa Class IIb Class III Level of evidence Level A Level B Level
Appendix: ACC/AHA and ESC practice guidelines Definitions for guideline recommendations and level of evidence Recommendation Class I Class IIa Class IIb Class III Level of evidence Level A Level B Level
Scottish Medicines Consortium
 Scottish Medicines Consortium bivalirudin, 250mg powder for concentrate for solution for injection or infusion (Angiox ) No. (516/08) The Medicines Company UK Ltd 07 November 2008 The Scottish Medicines
Scottish Medicines Consortium bivalirudin, 250mg powder for concentrate for solution for injection or infusion (Angiox ) No. (516/08) The Medicines Company UK Ltd 07 November 2008 The Scottish Medicines
Horizon Scanning Centre November 2012
 Horizon Scanning Centre November 2012 Cangrelor to reduce platelet aggregation and thrombosis in patients undergoing percutaneous coronary intervention99 SUMMARY NIHR HSC ID: 2424 This briefing is based
Horizon Scanning Centre November 2012 Cangrelor to reduce platelet aggregation and thrombosis in patients undergoing percutaneous coronary intervention99 SUMMARY NIHR HSC ID: 2424 This briefing is based
Anticoagulation therapy in acute coronary syndromes according to current guidelines
 Acute management of ACS Anticoagulation therapy in acute coronary syndromes according to current guidelines Marcin Grabowski, Marcin Leszczyk, Andrzej Cacko, Krzysztof J. Filipiak, Grzegorz Opolski 1 st
Acute management of ACS Anticoagulation therapy in acute coronary syndromes according to current guidelines Marcin Grabowski, Marcin Leszczyk, Andrzej Cacko, Krzysztof J. Filipiak, Grzegorz Opolski 1 st
Learning Objectives. Epidemiology of Acute Coronary Syndrome
 Cardiovascular Update: Antiplatelet therapy in acute coronary syndromes PHILLIP WEEKS, PHARM.D., BCPS-AQ CARDIOLOGY Learning Objectives Interpret guidelines as they relate to constructing an antiplatelet
Cardiovascular Update: Antiplatelet therapy in acute coronary syndromes PHILLIP WEEKS, PHARM.D., BCPS-AQ CARDIOLOGY Learning Objectives Interpret guidelines as they relate to constructing an antiplatelet
Percutaneous Coronary Intervention After Subcutaneous Enoxaparin Pretreatment in Patients With Unstable Angina Pectoris
 Percutaneous Coronary Intervention After Subcutaneous Enoxaparin Pretreatment in Patients With Unstable Angina Pectoris J.Ph. Collet, MD, PhD; G. Montalescot, MD, PhD; L. Lison, MD; R. Choussat, MD; A.
Percutaneous Coronary Intervention After Subcutaneous Enoxaparin Pretreatment in Patients With Unstable Angina Pectoris J.Ph. Collet, MD, PhD; G. Montalescot, MD, PhD; L. Lison, MD; R. Choussat, MD; A.
Inter-regional differences and outcome in unstable angina
 European Heart Journal (2000) 21, 1433 1439 doi:10.1053/euhj.1999.1983, available online at http://www.idealibrary.com on Inter-regional differences and outcome in unstable angina Analysis of the International
European Heart Journal (2000) 21, 1433 1439 doi:10.1053/euhj.1999.1983, available online at http://www.idealibrary.com on Inter-regional differences and outcome in unstable angina Analysis of the International
Prospective Comparison of Hemorrhagic Complications After Treatment With Enoxaparin
 Prospective Comparison of Hemorrhagic Complications After Treatment With Versus Unfractionated Heparin for Unstable Angina Pectoris or Non ST-Segment Elevation Acute Myocardial Infarction Scott D. Berkowitz,
Prospective Comparison of Hemorrhagic Complications After Treatment With Versus Unfractionated Heparin for Unstable Angina Pectoris or Non ST-Segment Elevation Acute Myocardial Infarction Scott D. Berkowitz,
Defining the Optimal Activated Clotting Time During Percutaneous Coronary Intervention
 Defining the Optimal Activated Clotting Time During Percutaneous Coronary Intervention Aggregate Results From 6 Randomized, Controlled Trials Derek P. Chew, MBBS; Deepak L. Bhatt, MD; A. Michael Lincoff,
Defining the Optimal Activated Clotting Time During Percutaneous Coronary Intervention Aggregate Results From 6 Randomized, Controlled Trials Derek P. Chew, MBBS; Deepak L. Bhatt, MD; A. Michael Lincoff,
Timing of Surgery After Percutaneous Coronary Intervention
 Timing of Surgery After Percutaneous Coronary Intervention Deepak Talreja, MD, FACC Bayview/EVMS/Sentara Outline/Highlights Timing of elective surgery What to do with medications Stopping anti-platelet
Timing of Surgery After Percutaneous Coronary Intervention Deepak Talreja, MD, FACC Bayview/EVMS/Sentara Outline/Highlights Timing of elective surgery What to do with medications Stopping anti-platelet
2003 by the American College of Cardiology Foundation ISSN /03/$30.00
 Journal of the American College of Cardiology Vol. 41, No. 4 Suppl S 2003 by the American College of Cardiology Foundation ISSN 0735-1097/03/$30.00 Published by Elsevier Science Inc. PII S0735-1097(02)02688-8
Journal of the American College of Cardiology Vol. 41, No. 4 Suppl S 2003 by the American College of Cardiology Foundation ISSN 0735-1097/03/$30.00 Published by Elsevier Science Inc. PII S0735-1097(02)02688-8
Direct Thrombin Inhibitors for PCI Pharmacology: Role of Bivalirudin in High-Risk PCI
 Direct Thrombin Inhibitors for PCI Pharmacology: Role of Bivalirudin in High-Risk PCI Charles A. Simonton MD, FACC, FSCAI Sanger Clinic Medical Director Clinical Innovation and Research Carolinas Heart
Direct Thrombin Inhibitors for PCI Pharmacology: Role of Bivalirudin in High-Risk PCI Charles A. Simonton MD, FACC, FSCAI Sanger Clinic Medical Director Clinical Innovation and Research Carolinas Heart
Role of Clopidogrel in Acute Coronary Syndromes. Hossam Kandil,, MD. Professor of Cardiology Cairo University
 Role of Clopidogrel in Acute Coronary Syndromes Hossam Kandil,, MD Professor of Cardiology Cairo University ACS Treatment Strategies Reperfusion/Revascularization Therapy Thrombolysis PCI (with/ without
Role of Clopidogrel in Acute Coronary Syndromes Hossam Kandil,, MD Professor of Cardiology Cairo University ACS Treatment Strategies Reperfusion/Revascularization Therapy Thrombolysis PCI (with/ without
Patient characteristics Intervention Comparison Length of followup
 ANTICOAGULANT CHAPTER Low Molecular Weight Heparins compared with Unfractionated Heparin Ref ID: 1903 Reference Antman EM, McCabe CH, Gurfinkel EP et al. Enoxaparin prevents death and cardiac ischemic
ANTICOAGULANT CHAPTER Low Molecular Weight Heparins compared with Unfractionated Heparin Ref ID: 1903 Reference Antman EM, McCabe CH, Gurfinkel EP et al. Enoxaparin prevents death and cardiac ischemic
Journal of the American College of Cardiology Vol. 40, No. 6, by the American College of Cardiology Foundation ISSN /02/$22.
 Journal of the American College of Cardiology Vol. 40, No. 6, 2002 2002 by the American College of Cardiology Foundation ISSN 0735-1097/02/$22.00 Published by Elsevier Science Inc. PII S0735-1097(02)02119-8
Journal of the American College of Cardiology Vol. 40, No. 6, 2002 2002 by the American College of Cardiology Foundation ISSN 0735-1097/02/$22.00 Published by Elsevier Science Inc. PII S0735-1097(02)02119-8
ATHEROSCLEROTIC HEART DISease
 CLINICAL CARDIOLOGY Platelet Glycoprotein IIb/IIIa Receptor Antagonists in Cardiovascular Disease David A. Vorchheimer, MD Juan Jose Badimon, PhD Valentin Fuster, MD, PhD ATHEROSCLEROTIC HEART DISease
CLINICAL CARDIOLOGY Platelet Glycoprotein IIb/IIIa Receptor Antagonists in Cardiovascular Disease David A. Vorchheimer, MD Juan Jose Badimon, PhD Valentin Fuster, MD, PhD ATHEROSCLEROTIC HEART DISease
Acute coronary syndrome (ACS) is a potentially
 DIAGNOSING ACUTE CORONARY SYNDROME AND DETERMINING PATIENT RISK Edith A. Nutescu, PharmD* ABSTRACT Acute coronary syndrome is a form of coronary artery disease and has a broad range of clinical presentations.
DIAGNOSING ACUTE CORONARY SYNDROME AND DETERMINING PATIENT RISK Edith A. Nutescu, PharmD* ABSTRACT Acute coronary syndrome is a form of coronary artery disease and has a broad range of clinical presentations.
(ClinicalTrials.gov ID: NCT ) Title: The Italian Elderly ACS Study Author: Stefano Savonitto. Date: 29 August 2011 Meeting: ESC congress, Paris
 Early aggressive versus initially conservative strategy in elderly patients with non-st- elevation acute coronary syndrome: the Italian randomised trial (ClinicalTrials.gov ID: NCT00510185) Stefano Savonitto,
Early aggressive versus initially conservative strategy in elderly patients with non-st- elevation acute coronary syndrome: the Italian randomised trial (ClinicalTrials.gov ID: NCT00510185) Stefano Savonitto,
The Thrombolysis In Myocardial Infarction Risk Score in Unstable Angina/ Non ST-Segment Elevation Myocardial Infarction
 Journal of the American College of Cardiology Vol. 41, No. 4 Suppl S 2003 by the American College of Cardiology Foundation ISSN 0735-1097/03/$30.00 Published by Elsevier Science Inc. doi:10.1016/s0735-1097(02)03019-x
Journal of the American College of Cardiology Vol. 41, No. 4 Suppl S 2003 by the American College of Cardiology Foundation ISSN 0735-1097/03/$30.00 Published by Elsevier Science Inc. doi:10.1016/s0735-1097(02)03019-x
Health technology Abciximab use in high-risk patients undergoing percutaneous transluminal coronary angioplasty.
 Costs and effects in therapy for acute coronary syndromes: the case of abciximab in highrisk patients undergoing percutaneous transluminal coronary angioplasty in the EPIC study van Hout B A, Bowman L,
Costs and effects in therapy for acute coronary syndromes: the case of abciximab in highrisk patients undergoing percutaneous transluminal coronary angioplasty in the EPIC study van Hout B A, Bowman L,
TRANSPARENCY COMMITTEE OPINION. 29 April 2009
 The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 29 April 2009 ANGIOX 250 mg, powder for concentrate for solution for injection or infusion by the intravenous route
The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 29 April 2009 ANGIOX 250 mg, powder for concentrate for solution for injection or infusion by the intravenous route
During the past 15 years, we have learned an enormous amount about the pathogenesis
 REVIEW ARTICLE Unstable Angina Current Concepts of Pathogenesis and Treatment John A. Ambrose, MD; George Dangas, MD During the past 15 years, we have learned an enormous amount about the pathogenesis
REVIEW ARTICLE Unstable Angina Current Concepts of Pathogenesis and Treatment John A. Ambrose, MD; George Dangas, MD During the past 15 years, we have learned an enormous amount about the pathogenesis
Adults With Diagnosed Diabetes
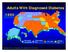 Adults With Diagnosed Diabetes 1990 No data available Less than 4% 4%-6% Above 6% Mokdad AH, et al. Diabetes Care. 2000;23(9):1278-1283. Adults With Diagnosed Diabetes 2000 4%-6% Above 6% Mokdad AH, et
Adults With Diagnosed Diabetes 1990 No data available Less than 4% 4%-6% Above 6% Mokdad AH, et al. Diabetes Care. 2000;23(9):1278-1283. Adults With Diagnosed Diabetes 2000 4%-6% Above 6% Mokdad AH, et
TIMI, PURSUIT, and GRACE risk scores: sustained prognostic value and interaction with revascularization in NSTE-ACS
 European Heart Journal (2005) 26, 865 872 doi:10.1093/eurheartj/ehi187 Clinical research TIMI, PURSUIT, and GRACE risk scores: sustained prognostic value and interaction with revascularization in NSTE-ACS
European Heart Journal (2005) 26, 865 872 doi:10.1093/eurheartj/ehi187 Clinical research TIMI, PURSUIT, and GRACE risk scores: sustained prognostic value and interaction with revascularization in NSTE-ACS
Myocardial Infarction In Dr.Yahya Kiwan
 Myocardial Infarction In 2007 Dr.Yahya Kiwan New Definition Of Acute Myocardial Infarction The term of myocardial infarction should be used when there is evidence of myocardial necrosis in a clinical setting
Myocardial Infarction In 2007 Dr.Yahya Kiwan New Definition Of Acute Myocardial Infarction The term of myocardial infarction should be used when there is evidence of myocardial necrosis in a clinical setting
Anti-thrombotic management in interventional cardiology
 Chapter 8 Anti-thrombotic management in interventional cardiology James Tcheng, Steve Kindsvater Introduction Since the dawn of the interventional age, the need for anti-thrombotic therapy during percutaneous
Chapter 8 Anti-thrombotic management in interventional cardiology James Tcheng, Steve Kindsvater Introduction Since the dawn of the interventional age, the need for anti-thrombotic therapy during percutaneous
Acute Coronary Syndromes
 Overview Acute Coronary Syndromes Rabeea Aboufakher, MD, FACC, FSCAI Section Chief of Cardiology Altru Health System Grand Forks, ND Epidemiology Pathophysiology Clinical features and diagnosis STEMI management
Overview Acute Coronary Syndromes Rabeea Aboufakher, MD, FACC, FSCAI Section Chief of Cardiology Altru Health System Grand Forks, ND Epidemiology Pathophysiology Clinical features and diagnosis STEMI management
Cardiovascular risk and therapeutic benefit of coronary interventions for patients with unstable angina according to the troponin T status
 European Heart Journal (2000) 21, 1159 1166 doi:10.1053/euhj.1999.1986, available online at http://www.idealibrary.com on Cardiovascular risk and therapeutic benefit of coronary interventions for patients
European Heart Journal (2000) 21, 1159 1166 doi:10.1053/euhj.1999.1986, available online at http://www.idealibrary.com on Cardiovascular risk and therapeutic benefit of coronary interventions for patients
Platelet Glycoprotein IIb/IIIa Antagonists
 REVIEW ARTICLE David C. Warltier, M.D., Ph.D., Editor Anesthesiology 2002; 96:1237 49 2002 American Society of Anesthesiologists, Inc. Lippincott Williams & Wilkins, Inc. Platelet Glycoprotein IIb/IIIa
REVIEW ARTICLE David C. Warltier, M.D., Ph.D., Editor Anesthesiology 2002; 96:1237 49 2002 American Society of Anesthesiologists, Inc. Lippincott Williams & Wilkins, Inc. Platelet Glycoprotein IIb/IIIa
Journal of the American College of Cardiology Vol. 44, No. 9, by the American College of Cardiology Foundation ISSN /04/$30.
 Journal of the American College of Cardiology Vol. 44, No. 9, 2004 2004 by the American College of Cardiology Foundation ISSN 0735-1097/04/$30.00 Published by Elsevier Inc. doi:10.1016/j.jacc.2004.05.085
Journal of the American College of Cardiology Vol. 44, No. 9, 2004 2004 by the American College of Cardiology Foundation ISSN 0735-1097/04/$30.00 Published by Elsevier Inc. doi:10.1016/j.jacc.2004.05.085
Percutaneous coronary intervention (PCI), in
 ECONOMIC IMPLICATIONS OF THE USE OF DIRECT THROMBIN INHIBITORS IN PERCUTANEOUS CORONARY INTERVENTION David J. Cohen, MD, MSc * ABSTRACT More than 1.2 million percutaneous coronary intervention (PCI) procedures
ECONOMIC IMPLICATIONS OF THE USE OF DIRECT THROMBIN INHIBITORS IN PERCUTANEOUS CORONARY INTERVENTION David J. Cohen, MD, MSc * ABSTRACT More than 1.2 million percutaneous coronary intervention (PCI) procedures
Facilitated Percutaneous Coronary Intervention in Acute Myocardial Infarction. Is it beneficial to patients?
 Facilitated Percutaneous Coronary Intervention in Acute Myocardial Infarction Is it beneficial to patients? Seung-Jea Tahk, MD. PhD. Suwon, Korea Facilitated PCI.. background Degree of coronary flow at
Facilitated Percutaneous Coronary Intervention in Acute Myocardial Infarction Is it beneficial to patients? Seung-Jea Tahk, MD. PhD. Suwon, Korea Facilitated PCI.. background Degree of coronary flow at
Pathophysiology of ACS
 Pathophysiology of ACS ~ 2.0 MM patients admitted to CCU or telemetry annually 0.6 MM ST-segment elevation MI 1.4 MM Non-ST-segment elevation ACS NSTEMI vs STEMI VANQWISH Boden et al N Engl J Med 1998;338:1785-1792
Pathophysiology of ACS ~ 2.0 MM patients admitted to CCU or telemetry annually 0.6 MM ST-segment elevation MI 1.4 MM Non-ST-segment elevation ACS NSTEMI vs STEMI VANQWISH Boden et al N Engl J Med 1998;338:1785-1792
S E C T I O N. Cardiovascular Medicine
 S E C T I O N I V Cardiovascular Medicine 102 CME 2004 C h a p t e r 1 5 Gp IIb/IIIa Receptor Antagonists in Intensive Coronary Care Unit Lekha Adik-Pathak 1, Manjeet S. Juneja 2, Shivkumar Bhairappa 3
S E C T I O N I V Cardiovascular Medicine 102 CME 2004 C h a p t e r 1 5 Gp IIb/IIIa Receptor Antagonists in Intensive Coronary Care Unit Lekha Adik-Pathak 1, Manjeet S. Juneja 2, Shivkumar Bhairappa 3
Continuing Medical Education Post-Test
 Continuing Medical Education Post-Test Based on the information presented in this monograph, please choose one correct response for each of the following questions or statements. Record your answers on
Continuing Medical Education Post-Test Based on the information presented in this monograph, please choose one correct response for each of the following questions or statements. Record your answers on
Acute ischemic coronary syndromes (AICS)
 ...CONTINUING MEDICAL EDUCATION... CME ARTICLE Overview of Platelet Glycoprotein IIb/IIIa Receptor Antagonist Treatment in the Management of Coronary Interventions and Acute Ischemic Coronary Syndromes
...CONTINUING MEDICAL EDUCATION... CME ARTICLE Overview of Platelet Glycoprotein IIb/IIIa Receptor Antagonist Treatment in the Management of Coronary Interventions and Acute Ischemic Coronary Syndromes
The Role of Low-Molecular-Weight Heparin in the Management of Acute Coronary Syndromes
 Journal of the American College of Cardiology Vol. 41, No. 4 Suppl S 2003 by the American College of Cardiology Foundation ISSN 0735-1097/03/$30.00 Published by Elsevier Science Inc. doi:10.1016/s0735-1097(02)02901-7
Journal of the American College of Cardiology Vol. 41, No. 4 Suppl S 2003 by the American College of Cardiology Foundation ISSN 0735-1097/03/$30.00 Published by Elsevier Science Inc. doi:10.1016/s0735-1097(02)02901-7
The Clopidogrel in Unstable angina to prevent Recurrent ischemic Events (CURE) Trial
 Benefits and Risks of the Combination of Clopidogrel and Aspirin in Patients Undergoing Surgical Revascularization for Non ST-Elevation Acute Coronary Syndrome The Clopidogrel in Unstable angina to prevent
Benefits and Risks of the Combination of Clopidogrel and Aspirin in Patients Undergoing Surgical Revascularization for Non ST-Elevation Acute Coronary Syndrome The Clopidogrel in Unstable angina to prevent
Scottish Medicines Consortium
 Scottish Medicines Consortium bivalirudin 250mg for injection or infusion (Angiox ) (156/05) Nycomed UK Ltd No. 4 February, 2005 The Scottish Medicines Consortium has completed its assessment of the above
Scottish Medicines Consortium bivalirudin 250mg for injection or infusion (Angiox ) (156/05) Nycomed UK Ltd No. 4 February, 2005 The Scottish Medicines Consortium has completed its assessment of the above
Belinda Green, Cardiologist, SDHB, 2016
 Acute Coronary syndromes All STEMI ALL Non STEMI Unstable angina Belinda Green, Cardiologist, SDHB, 2016 Thrombus in proximal LAD Underlying pathophysiology Be very afraid for your patient Wellens
Acute Coronary syndromes All STEMI ALL Non STEMI Unstable angina Belinda Green, Cardiologist, SDHB, 2016 Thrombus in proximal LAD Underlying pathophysiology Be very afraid for your patient Wellens
Is the role of bivalirudin established?
 Is the role of bivalirudin established? Rob Henderson Consultant Cardiologist Trent Cardiac Centre Nottingham University Hospitals Conflicts of Interest: None Declarations: Member NICE Unstable Angina
Is the role of bivalirudin established? Rob Henderson Consultant Cardiologist Trent Cardiac Centre Nottingham University Hospitals Conflicts of Interest: None Declarations: Member NICE Unstable Angina
Medicine Dr. Omed Lecture 2 Stable and Unstable Angina
 Medicine Dr. Omed Lecture 2 Stable and Unstable Angina Risk stratification in stable angina. High Risk; *post infarct angina, *poor effort tolerance, *ischemia at low workload, *left main or three vessel
Medicine Dr. Omed Lecture 2 Stable and Unstable Angina Risk stratification in stable angina. High Risk; *post infarct angina, *poor effort tolerance, *ischemia at low workload, *left main or three vessel
Treatment Strategies for the Prevention of Ischemic Complications in Patients Undergoing Percutaneous Coronary Intervention with Stent Placement
 Treatment Strategies for the Prevention of Ischemic Complications in Patients Undergoing Percutaneous Coronary Intervention with Stent Placement Pharmaceutical Care Project Outcomes Literature Evaluation
Treatment Strategies for the Prevention of Ischemic Complications in Patients Undergoing Percutaneous Coronary Intervention with Stent Placement Pharmaceutical Care Project Outcomes Literature Evaluation
Benefits and Safety of Tirofiban Among Acute Coronary Syndrome Patients With Mild to Moderate Renal Insufficiency
 Benefits and Safety of Tirofiban Among Acute Coronary Syndrome Patients With Mild to Moderate Renal Insufficiency Results From the Platelet Receptor Inhibition in Ischemic Syndrome Management in Patients
Benefits and Safety of Tirofiban Among Acute Coronary Syndrome Patients With Mild to Moderate Renal Insufficiency Results From the Platelet Receptor Inhibition in Ischemic Syndrome Management in Patients
METHODS OBJECTIVES BACKGROUND METHODS
 Journal of the American College of Cardiology Vol. 36, No. 3, 2000 2000 by the American College of Cardiology ISSN 0735-1097/00/$20.00 Published by Elsevier Science Inc. PII S0735-1097(00)00814-7 Platelet
Journal of the American College of Cardiology Vol. 36, No. 3, 2000 2000 by the American College of Cardiology ISSN 0735-1097/00/$20.00 Published by Elsevier Science Inc. PII S0735-1097(00)00814-7 Platelet
Frequency and Clinical Implications of Discordant Creatine Kinase-MB and Troponin Measurements in Acute Coronary Syndromes
 Journal of the American College of Cardiology Vol. 47, No. 2, 2006 2006 by the American College of Cardiology Foundation ISSN 0735-1097/06/$32.00 Published by Elsevier Inc. doi:10.1016/j.jacc.2005.08.062
Journal of the American College of Cardiology Vol. 47, No. 2, 2006 2006 by the American College of Cardiology Foundation ISSN 0735-1097/06/$32.00 Published by Elsevier Inc. doi:10.1016/j.jacc.2005.08.062
Acute Coronary Syndrome. ACC/AHA 2002 Guidelines
 Acute Coronary Syndrome ACC/AHA 2002 Guidelines ACS Unstable Angina Non ST elevation MI ST elevation MI ACS UA and Non STEMI described in these guidelines Management of STEMI described in separate guidelines
Acute Coronary Syndrome ACC/AHA 2002 Guidelines ACS Unstable Angina Non ST elevation MI ST elevation MI ACS UA and Non STEMI described in these guidelines Management of STEMI described in separate guidelines
Journal of the American College of Cardiology Vol. 41, No. 6, by the American College of Cardiology Foundation ISSN /03/$30.
 Journal of the American College of Cardiology Vol. 41, No. 6, 2003 2003 by the American College of Cardiology Foundation ISSN 0735-1097/03/$30.00 Published by Elsevier Science Inc. doi:10.1016/s0735-1097(02)02970-4
Journal of the American College of Cardiology Vol. 41, No. 6, 2003 2003 by the American College of Cardiology Foundation ISSN 0735-1097/03/$30.00 Published by Elsevier Science Inc. doi:10.1016/s0735-1097(02)02970-4
CLINICIAN INTERVIEW RECOGNIZING ACS AND STRATIFYING RISK IN PRIMARY CARE. An interview with A. Michael Lincoff, MD, and Eric R. Bates, MD, FACC, FAHA
 RECOGNIZING ACS AND STRATIFYING RISK IN PRIMARY CARE An interview with A. Michael Lincoff, MD, and Eric R. Bates, MD, FACC, FAHA Dr Lincoff is an interventional cardiologist and the Vice Chairman for Research
RECOGNIZING ACS AND STRATIFYING RISK IN PRIMARY CARE An interview with A. Michael Lincoff, MD, and Eric R. Bates, MD, FACC, FAHA Dr Lincoff is an interventional cardiologist and the Vice Chairman for Research
Interventional Cardiology
 Clinical Investigations Interventional Cardiology Benefit of bolus-only platelet glycoprotein IIb/IIIa inhibition during percutaneous coronary intervention: Insights from the very early outcomes in the
Clinical Investigations Interventional Cardiology Benefit of bolus-only platelet glycoprotein IIb/IIIa inhibition during percutaneous coronary intervention: Insights from the very early outcomes in the
Cardiovascular Disorders Lecture 3 Coronar Artery Diseases
 Cardiovascular Disorders Lecture 3 Coronar Artery Diseases By Prof. El Sayed Abdel Fattah Eid Lecturer of Internal Medicine Delta University Coronary Heart Diseases It is the leading cause of death in
Cardiovascular Disorders Lecture 3 Coronar Artery Diseases By Prof. El Sayed Abdel Fattah Eid Lecturer of Internal Medicine Delta University Coronary Heart Diseases It is the leading cause of death in
Two-Year Follow-Up of Tirofiban-Based Management of Non-ST-Elevation Acute Coronary Syndrome A Single Center Study
 Original Article Follow-Up of Tirofiban-Based NSTEACS Management Acta Cardiol Sin 2010;26:19 27 Coronary Heart Disease Two-Year Follow-Up of Tirofiban-Based Management of Non-ST-Elevation Acute Coronary
Original Article Follow-Up of Tirofiban-Based NSTEACS Management Acta Cardiol Sin 2010;26:19 27 Coronary Heart Disease Two-Year Follow-Up of Tirofiban-Based Management of Non-ST-Elevation Acute Coronary
A Multicenter Randomized Trial of Immediate Versus Delayed Invasive Strategy in Patients with Non-ST Elevation ACS
 Angioplasty to Blunt the rise Of troponin in Acute coronary syndromes Randomized for an immediate or Delayed intervention A Multicenter Randomized Trial of Immediate Versus Delayed Invasive Strategy in
Angioplasty to Blunt the rise Of troponin in Acute coronary syndromes Randomized for an immediate or Delayed intervention A Multicenter Randomized Trial of Immediate Versus Delayed Invasive Strategy in
Timing of Anti-Platelet Therapy for ACS (EARLY-ACS & ACUITY) Mitchell W. Krucoff, MD, FACC
 Timing of Anti-Platelet Therapy for ACS (EARLY-ACS & ACUITY) Mitchell W. Krucoff, MD, FACC Professor, Medicine/Cardiology Duke University Medical Center Director, Cardiovascular Devices Unit Duke Clinical
Timing of Anti-Platelet Therapy for ACS (EARLY-ACS & ACUITY) Mitchell W. Krucoff, MD, FACC Professor, Medicine/Cardiology Duke University Medical Center Director, Cardiovascular Devices Unit Duke Clinical
IN patients with ST elevated myocardial infarction, ST segment resolution following
 The Effect of Tirofiban on ST Segment Resolution in Patients With Non-ST Elevated Myocardial Infarction Özgür BAYTURAN, 1 MD, Ali Riza BILGE, 1 MD, Cevad SEKÜRI, 1 MD, Ozan ÜTÜK, 1 MD, Hakan TIKIZ, 1 MD,
The Effect of Tirofiban on ST Segment Resolution in Patients With Non-ST Elevated Myocardial Infarction Özgür BAYTURAN, 1 MD, Ali Riza BILGE, 1 MD, Cevad SEKÜRI, 1 MD, Ozan ÜTÜK, 1 MD, Hakan TIKIZ, 1 MD,
bivalirudin 250mg powder for concentrate for solution for injection or infusion (Angiox) SMC No. (638/10) The Medicines Company
 bivalirudin 250mg powder for concentrate for solution for injection or infusion (Angiox) SMC No. (638/10) The Medicines Company 06 August 2010 The Scottish Medicines Consortium (SMC) has completed its
bivalirudin 250mg powder for concentrate for solution for injection or infusion (Angiox) SMC No. (638/10) The Medicines Company 06 August 2010 The Scottish Medicines Consortium (SMC) has completed its
Optimal antiplatelet and anticoagulant therapy for patients treated in STEMI network
 Torino 6 Joint meeting with Mayo Clinic Great Innovation in Cardiology 14-15 Ottobre 2010 Optimal antiplatelet and anticoagulant therapy for patients treated in STEMI network Diego Ardissino Ischemic vs
Torino 6 Joint meeting with Mayo Clinic Great Innovation in Cardiology 14-15 Ottobre 2010 Optimal antiplatelet and anticoagulant therapy for patients treated in STEMI network Diego Ardissino Ischemic vs
Keywords Non ST-segment elevation ACS Antithrombotic therapy Glycoprotein IIb/IIIa inhibitor Tirofiban. Introduction
 J Thromb Thrombolysis (2007) 24:241 246 DOI 10.1007/s19-007-0015-y Routine upstream versus selective down stream use of tirofiban in non-st elevation myocardial infarction patients scheduled for early
J Thromb Thrombolysis (2007) 24:241 246 DOI 10.1007/s19-007-0015-y Routine upstream versus selective down stream use of tirofiban in non-st elevation myocardial infarction patients scheduled for early
Clinical Therapeutics/Volume 31, Number 8, 2009
 Clinical Therapeutics/Volume 31, Number 8, 2009 Assessment of the Bioequivalence of Brand and Biogeneric Formulations of Abciximab for the Treatment of Acute Coronary Syndrome: A Prospective, Open-Label,
Clinical Therapeutics/Volume 31, Number 8, 2009 Assessment of the Bioequivalence of Brand and Biogeneric Formulations of Abciximab for the Treatment of Acute Coronary Syndrome: A Prospective, Open-Label,
Acute Coronary Syndromes. January 9, 2013 Chris Chiles M.D. FACC
 Acute Coronary Syndromes January 9, 2013 Chris Chiles M.D. FACC Disclosures None- not even a breakfast burrito from a drug company Hospitalizations in the U.S. Due to ACS Acute Coronary Syndromes* 1.57
Acute Coronary Syndromes January 9, 2013 Chris Chiles M.D. FACC Disclosures None- not even a breakfast burrito from a drug company Hospitalizations in the U.S. Due to ACS Acute Coronary Syndromes* 1.57
Nova Scotia Guidelines for Acute Coronary Syndromes (Updating the 2008 Antiplatelet Section of the Guidelines)
 Cardiovascular Health Nova Scotia Guideline Update Nova Scotia Guidelines for Acute Coronary Syndromes (Updating the 2008 Antiplatelet Section of the Guidelines) Authors: Dr. M. Love, Dr. I. Bata, K. Harrigan
Cardiovascular Health Nova Scotia Guideline Update Nova Scotia Guidelines for Acute Coronary Syndromes (Updating the 2008 Antiplatelet Section of the Guidelines) Authors: Dr. M. Love, Dr. I. Bata, K. Harrigan
AHA Scientific Statement
 AHA Scientific Statement Acute Coronary Care in the Elderly, Part I Non ST-Segment Elevation Acute Coronary Syndromes A Scientific Statement for Healthcare Professionals From the American Heart Association
AHA Scientific Statement Acute Coronary Care in the Elderly, Part I Non ST-Segment Elevation Acute Coronary Syndromes A Scientific Statement for Healthcare Professionals From the American Heart Association
Acute Coronary Syndrome. Cindy Baker, MD FACC Director Peripheral Vascular Interventions Division of Cardiovascular Medicine
 Acute Coronary Syndrome Cindy Baker, MD FACC Director Peripheral Vascular Interventions Division of Cardiovascular Medicine Topics Timing is everything So many drugs to choose from What s a MINOCA? 2 Acute
Acute Coronary Syndrome Cindy Baker, MD FACC Director Peripheral Vascular Interventions Division of Cardiovascular Medicine Topics Timing is everything So many drugs to choose from What s a MINOCA? 2 Acute
The restoration of coronary flow after an
 Pharmacological Reperfusion in Acute Myicardial Infarction after ASSENT 3 and GUSTO V [81] DANIEL FERREIRA, MD, FESC Serviço de Cardiologia, Hospital Fernando Fonseca, Amadora, Portugal Rev Port Cardiol
Pharmacological Reperfusion in Acute Myicardial Infarction after ASSENT 3 and GUSTO V [81] DANIEL FERREIRA, MD, FESC Serviço de Cardiologia, Hospital Fernando Fonseca, Amadora, Portugal Rev Port Cardiol
Journal of the American College of Cardiology Vol. 35, No. 7, by the American College of Cardiology ISSN /00/$20.
 Journal of the American College of Cardiology Vol. 35, No. 7, 2000 2000 by the American College of Cardiology ISSN 0735-1097/00/$20.00 Published by Elsevier Science Inc. PII S0735-1097(00)00684-3 REVIEW
Journal of the American College of Cardiology Vol. 35, No. 7, 2000 2000 by the American College of Cardiology ISSN 0735-1097/00/$20.00 Published by Elsevier Science Inc. PII S0735-1097(00)00684-3 REVIEW
Unstable angina and NSTEMI
 Issue date: March 2010 Unstable angina and NSTEMI The early management of unstable angina and non-st-segment-elevation myocardial infarction This guideline updates and replaces recommendations for the
Issue date: March 2010 Unstable angina and NSTEMI The early management of unstable angina and non-st-segment-elevation myocardial infarction This guideline updates and replaces recommendations for the
Case Study 50 YEAR OLD MALE WITH UNSTABLE ANGINA
 Case Study 50 YEAR OLD MALE WITH UNSTABLE ANGINA Case History A 50-year-old man with type 1 diabetes mellitus and hypertension presents after experiencing 1 hour of midsternal chest pain that began after
Case Study 50 YEAR OLD MALE WITH UNSTABLE ANGINA Case History A 50-year-old man with type 1 diabetes mellitus and hypertension presents after experiencing 1 hour of midsternal chest pain that began after
M/39 CC D. => peak CKMB (12 hr later) ng/ml T.chol/TG/HDL/LDL 180/150/48/102 mg/dl #
 Acute Coronary Syndrome - Case Review - Young-Guk Ko, MD Yonsei Cardiovascular Center Yonsei University College of Medicine Case 1 M/39 #4306212 CC D : Severe squeezing chest pain : 4 hours, aggravated
Acute Coronary Syndrome - Case Review - Young-Guk Ko, MD Yonsei Cardiovascular Center Yonsei University College of Medicine Case 1 M/39 #4306212 CC D : Severe squeezing chest pain : 4 hours, aggravated
Columbia University Medical Center Cardiovascular Research Foundation
 STEMI and NSTEMI Pharmacology Confusion: How to Choose and Use Antithrombins (Unfractionated and Low Molecular Heparins, Bivalirudin, Fondaparinux) and Antiplatelet Agents (Aspirin, Clopidogrel and Prasugrel)
STEMI and NSTEMI Pharmacology Confusion: How to Choose and Use Antithrombins (Unfractionated and Low Molecular Heparins, Bivalirudin, Fondaparinux) and Antiplatelet Agents (Aspirin, Clopidogrel and Prasugrel)
New Antithrombotic and Antiplatelet Drugs in CAD : (Factor Xa inhibitors, Direct Thrombin inhibitors and Prasugrel)
 New Antithrombotic and Antiplatelet Drugs in CAD : (Factor Xa inhibitors, Direct Thrombin inhibitors and Prasugrel) Limitations and Advantages of UFH and LMWH Biological limitations of UFH : 1. immune-mediated
New Antithrombotic and Antiplatelet Drugs in CAD : (Factor Xa inhibitors, Direct Thrombin inhibitors and Prasugrel) Limitations and Advantages of UFH and LMWH Biological limitations of UFH : 1. immune-mediated
Original Article Impact of timing to coronary angiography in acute coronary syndrome on contemporary clinical practice
 Am J Cardiovasc Dis 2012;2(3):248-252 www.ajcd.us /ISSN:2160-200X/AJCD1204002 Original Article Impact of timing to coronary angiography in acute coronary syndrome on contemporary clinical practice Angela
Am J Cardiovasc Dis 2012;2(3):248-252 www.ajcd.us /ISSN:2160-200X/AJCD1204002 Original Article Impact of timing to coronary angiography in acute coronary syndrome on contemporary clinical practice Angela
2007 ACC/AHA GUIDELINES FOR THE MANAGEMENT OF NSTE-ACS: OPTIMAL ANTICOAGULATION AND ANTIPLATELET THERAPY
 2007 ACC/AHA GUIDELINES FOR THE MANAGEMENT OF NSTE-ACS: OPTIMAL ANTICOAGULATION AND ANTIPLATELET THERAPY Charles V. Pollack, Jr., MA, MD, FACEP, FAAEM, FAHA Professor and Chairman, Department of Emergency
2007 ACC/AHA GUIDELINES FOR THE MANAGEMENT OF NSTE-ACS: OPTIMAL ANTICOAGULATION AND ANTIPLATELET THERAPY Charles V. Pollack, Jr., MA, MD, FACEP, FAAEM, FAHA Professor and Chairman, Department of Emergency
CHAPTER 17 Antithrombotic Agents Heparins
 CHAPTER 17 Antithrombotic Agents Heparins Structure Mechanism of Action Pharmacokinetics Limitations of Unfractionated Heparin Heparin Induced Thrombocytopenia Heparin Rebound Low Molecular Weight Heparins
CHAPTER 17 Antithrombotic Agents Heparins Structure Mechanism of Action Pharmacokinetics Limitations of Unfractionated Heparin Heparin Induced Thrombocytopenia Heparin Rebound Low Molecular Weight Heparins
Differential Expression of Cardiac Biomarkers by Gender in Patients With Unstable Angina/Non ST-Elevation Myocardial Infarction
 Differential Expression of Cardiac Biomarkers by Gender in Patients With Unstable Angina/Non ST-Elevation Myocardial Infarction A TACTICS-TIMI 18 (Treat Angina with Aggrastat and determine Cost of Therapy
Differential Expression of Cardiac Biomarkers by Gender in Patients With Unstable Angina/Non ST-Elevation Myocardial Infarction A TACTICS-TIMI 18 (Treat Angina with Aggrastat and determine Cost of Therapy
Novel Risk Markers in ACS (Hyperglycemia, Anemia, GFR)
 Novel Risk Markers in ACS (Hyperglycemia, Anemia, GFR) Shaul Atar, MD Department of Cardiology Faculty of Medicine of the Galilee Western Galilee Medical Center, Nahariya, Israel TIMI Risk Score Age 65
Novel Risk Markers in ACS (Hyperglycemia, Anemia, GFR) Shaul Atar, MD Department of Cardiology Faculty of Medicine of the Galilee Western Galilee Medical Center, Nahariya, Israel TIMI Risk Score Age 65
Bolus-Only Platelet Glycoprotein IIb/IIIa Inhibition During Percutaneous Coronary Intervention
 Original Contribution Bolus-Only Platelet Glycoprotein IIb/IIIa Inhibition During Percutaneous Coronary Intervention Jonathan D. Marmur, MD, Shyam Poludasu, MD, Ajay Agarwal, MD, Pompeiu Vladutiu, MD,
Original Contribution Bolus-Only Platelet Glycoprotein IIb/IIIa Inhibition During Percutaneous Coronary Intervention Jonathan D. Marmur, MD, Shyam Poludasu, MD, Ajay Agarwal, MD, Pompeiu Vladutiu, MD,
Benefit of Early Invasive Therapy in Acute Coronary Syndromes A Meta-Analysis of Contemporary Randomized Clinical Trials
 Journal of the American College of Cardiology Vol. 48, No. 7, 2006 2006 by the American College of Cardiology Foundation ISSN 0735-1097/06/$32.00 Published by Elsevier Inc. doi:10.1016/j.jacc.2006.06.050
Journal of the American College of Cardiology Vol. 48, No. 7, 2006 2006 by the American College of Cardiology Foundation ISSN 0735-1097/06/$32.00 Published by Elsevier Inc. doi:10.1016/j.jacc.2006.06.050
ST-segment Elevation Myocardial Infarction (STEMI): Optimal Antiplatelet and Anti-thrombotic Therapy in the Emergency Department
 ST-segment Elevation Myocardial Infarction (STEMI): Optimal Antiplatelet and Anti-thrombotic Therapy in the Emergency Department decision-making. They have become the cornerstone of many ED protocols for
ST-segment Elevation Myocardial Infarction (STEMI): Optimal Antiplatelet and Anti-thrombotic Therapy in the Emergency Department decision-making. They have become the cornerstone of many ED protocols for
Chronic kidney disease (CKD) is frequently encountered
 AHA Scientific Statement Pharmacotherapy in Chronic Kidney Disease Patients Presenting With Acute Coronary Syndrome A Scientific Statement From the American Heart Association Jeffrey B. Washam, PharmD,
AHA Scientific Statement Pharmacotherapy in Chronic Kidney Disease Patients Presenting With Acute Coronary Syndrome A Scientific Statement From the American Heart Association Jeffrey B. Washam, PharmD,
Impact of Delay to Angioplasty in Patients With Acute Coronary Syndromes Undergoing Invasive Management
 Journal of the American College of Cardiology Vol. 55, No. 1, by the American College of Cardiology Foundation ISSN 735-97//$3. Published by Elsevier Inc. doi:.1/j.jacc.9.11.3 CLINICAL RESEARCH Invasive
Journal of the American College of Cardiology Vol. 55, No. 1, by the American College of Cardiology Foundation ISSN 735-97//$3. Published by Elsevier Inc. doi:.1/j.jacc.9.11.3 CLINICAL RESEARCH Invasive
Asif Serajian DO FACC FSCAI
 Anticoagulation and Antiplatelet update: A case based approach Asif Serajian DO FACC FSCAI No disclosures relevant to this talk Objectives 1. Discuss the indication for antiplatelet therapy for cardiac
Anticoagulation and Antiplatelet update: A case based approach Asif Serajian DO FACC FSCAI No disclosures relevant to this talk Objectives 1. Discuss the indication for antiplatelet therapy for cardiac
FastTest. You ve read the book now test yourself
 FastTest You ve read the book...... now test yourself To ensure you have learned the key points that will improve your patient care, read the authors questions below. The answers will refer you back to
FastTest You ve read the book...... now test yourself To ensure you have learned the key points that will improve your patient care, read the authors questions below. The answers will refer you back to
High-Dose Tirofiban Administered as Bolus-Only during Percutaneous Coronary Intervention
 Original Contribution High-Dose Tirofiban Administered as Bolus-Only during Percutaneous Coronary Intervention Jonathan D. Marmur, MD, Shyam Poludasu, MD, Ajay Agarwal, MD, Nagarathna Manjappa MD, Erdal
Original Contribution High-Dose Tirofiban Administered as Bolus-Only during Percutaneous Coronary Intervention Jonathan D. Marmur, MD, Shyam Poludasu, MD, Ajay Agarwal, MD, Nagarathna Manjappa MD, Erdal
