Manual of Interventional Cardiology
|
|
|
- Nickolas Hubbard
- 5 years ago
- Views:
Transcription
1 782 PLATELET GLYCOPROTEIN IIb/IIIa RECEPTOR ANTAGONISTS (Tables 34.15, 34.16) Activation of the platelet glycoprotein (GP) IIb/IIIa receptor complex constitutes the final common pathway for platelet aggregation, and is a critical event in arterial thrombosis (Figure 40.4, Chapter 40). Potent GP IIb/IIIa receptor antagonists have been shown to improve event-free survival in a variety of interventional and non-interventional settings and are the focus of intensive investigation. is a noncompetive antagonist (monoclonal antibody) that binds 1:1 to the GP IIb/IIIa receptor molecule to induce a conformational change that renders its fibrinogen-binding site inactive. Eptifibatide and tirofiban are competitive antagonists that reversibly inhibit the binding of fibrinogen to the GP IIb/IIIa receptor by directly interacting with its RGD binding site. From a clinical standpoint, noncompetitive inhibitors (such as abciximab) have a longer biological half-life, more cross-reactivity with other cell-surface receptors, and higher dissociation constants (more permanent binding) than competitive inhibitors, which may translate into more durable clinical efficacy (in terms of reducing ischemic complications) and less restenosis (possibly due to cross-reactivity with the vitronectin receptor). Numerous studies have evaluated these agents for acute coronary syndromes and as adjuncts for percutaneous coronary intervention (Tables ). The specific agents and clinical trials are detailed below. A. ABCIXIMAB. (ReoPro, Centocor) is the Fab fragment of the chimeric monoclonal antibody 7E3. binds to and blocks platelet GP IIb/IIIa receptors, the vitronectin receptor (α v β 3 ) on smooth muscle and endothelial cells, and the Mac-1 receptor on leukocytes (Table 34.22). Interaction with these receptors may confer antiplatelet, antiproliferative (i.e., restenosis), and antiinflammatory activity, respectively. Inhibition of clot retraction, factor XIII, and PAI-I, displacement of fibrinogen, and prolongation of the ACT may confer additional anticoagulant and thrombolytic activity unique to abciximab. 1. Trials (Tables ). has been studied in several large-scale, placebo-controlled randomized trials in PTCA (EPIC, EPILOG, CAPTURE), stenting (EPISTENT), acute MI (ADMIRAL, RAPPORT, CADILLAC), and acute coronary syndromes (GUSTO-IV ACS); a comparative trial of abciximab vs. tirofiban (TARGET) has recently been completed. EPIC, EPILOG, EPISTENT, and CAPTURE all demonstrated 35-57% reductions in the primary endpoint (death, MI, TLR) favoring abciximab, 131, which was particularly evident in patients with unstable angina and acute MI. Although EPIC reported more bleeding complications with abciximab, EPILOG demonstrated that low-dose, weight-adjusted heparin (70 U/kg to achieve ACT seconds) plus abciximab could significantly attenuate the risk of bleeding without sacrificing efficacy. Furthermore, benefits observed in high-risk PTCA patients in EPIC were extended to high and low-risk PTCA and stent patients in EPILOG and EPISTENT. Importantly, EPIC continues to demonstrate sustained benefit of abciximab at 30 days, 6 months, and 3 years. 131,132,138 EPILOG has shown persistent benefit at 1 year, 137 and EPISTENT reported sustained benefit at 6 months 135 and at 1 year (mortality: 1% vs. 2.4%). 140 Angiographic restenosis was also lower in diabetics treated with abciximab. 141 In contrast to the abciximab bolus and 12-
2 Chapter 34. Adjunctive Pharmacotherapy 783 hour infusion utilized in EPIC, EPILOG, and EPISTENT, CAPTURE patients received an infusion before and a 1-hour infusion after intervention; initial benefit at 30 days was not sustained at 6 months, 134 suggesting that a 12-hour infusion after intervention may be important for sustained clinical efficacy. has also been studied as an adjunct to thrombolytic therapy for acute MI patients with ST-segment elevation (Table 34.21). In patients with acute MI undergoing primary PTCA or stenting, three trials of abciximab have reported divergent results (Table 34.18): In RAPPORT, 136 abciximab was associated with a nonsignificant 17% reduction in late MACE after primary PTCA (p = 0.07). In ADMIRAL, 142 abciximab was associated with a 47% reduction in late MACE after primary stenting (p = 0.03). In CADILLAC, 143 patients were randomized to primary PTCA vs. stenting, and abciximab vs. no abciximab. Similar to RAPPORT, the 21% reduction in late MACE after primary PTCA was not significant, and in contrast to ADMIRAL, abciximab offered no incremental benefit after primary stenting. In patients with non-st-segment elevation acute coronary syndromes, GUSTO-IV ACS revealed no benefit for abciximab (24 or 48- hour infusion) when added to aspirin and heparin (UFH or dalteparin). 144 Finally, the TARGET trial 145 reported better results for abciximab compared to tirofiban for stent patients (death, MI, or urgent revascularization at 30 days: abciximab 6.0% vs. tirofiban 7.5%, p = 0.037), especially those presenting with an acute coronary syndrome. 2. Dosage. The recommended dose for abciximab for percutaneous intervention is an IV bolus of 0.25 mg/kg administered 10 minutes before intervention, followed by a continuous infusion of mcg/kg/min (to a maximum of 10 mcg/min) for 12 hours after intervention. All patients should also receive standard aspirin therapy. The recommended heparin dose is 70 U/kg to achieve an ACT seconds; additional heparin is not recommended after intervention. Vascular sheaths should be removed when the ACT is seconds, and there is no need to discontinue abciximab for sheath removal. When abciximab is administered after full-dose heparinization ( rescue ReoPro ), additional heparin should be used cautiously to reduce the risk of bleeding; clear guidelines have not been established, and although some suggest partial reversal of full-dose heparin with protamine, this is not routinely recommended. 3. Limitations (Chapter 25). A number of safety concerns have been raised about the use of IIb/IIIa antagonists in general and about abciximab in particular. Most of these concerns are related to bleeding complications, the potential requirement for emergency CABG, severe thrombocytopenia, and potential drug interactions. Virtually all of these considerations are readily prevented and/or treated, and do not constitute significant obstacles to the use of IIb/IIIa inhibitors. a. Bleeding (Table 34.23). In most IIb/IIIa inhibitor trials, ,135,136 major bleeding was defined as a drop in hemoglobin > 5 gm/dl, an absolute drop in hematocrit > 15%, or intracranial hemorrhage; minor bleeding included spontaneous gross hematuria, spontaneous hematemesis, a drop in hemoglobin > 3 gm/dl with evident blood loss, or a drop in hemoglobin > 4 gm/dl without evident blood loss. In abciximab trials utilizing standard-dose heparin to maintain high ACT levels (EPIC, EPILOG, RAPPORT), bleeding was more frequent with abciximab vs. placebo (Table 34.23); independent predictors of bleeding included acute MI, low body
3 784 weight, old age, longer procedural times, repeat PTCA, and failed intervention. In contrast, trials utilizing low-dose heparin, early sheath removal, and ACT seconds (EPILOG, CAPTURE, EPISTENT) reported no difference in major or minor bleeding for patients treated with abciximab or placebo. These studies suggest that the risk of bleeding associated with abciximab can be minimized by low-dose heparin, early sheath removal, avoidance of venous sheaths, and fastidious post-procedure groin care (Table 34.24). Importantly, patients treated with abciximab do not have a higher incidence of intracranial hemorrhage compared to placebo. b. Thrombocytopenia (Table 34.25). A potential safety concern with IIb/IIIa antagonists is thrombocytopenia, which may increase bleeding complications. 146 The incidence of thrombocytopenia appears to be higher with abciximab than with eptifibatide or tirofiban. In the clinical trials, the incidence of mild thrombocytopenia (< 100,000/mm 3 ) was % and severe thrombocytopenia (< 50,000/mm 3 ) was %; platelet transfusions were administered in %. Unlike heparin-induced thrombocytopenia, abciximab-associated thrombocytopenia responds within days of discontinuing the drug, and responds promptly to platelet transfusions (Tables 34.24, 34.25). In contrast, severe thrombocytopenia after abciximab retreatment (which is 2-3 times more frequent than after first time use) may not respond promptly to platelet transfusions. Although abciximab is not FDA-approved for readministration, repeat use is common in clinical practice. One study of readministration of abciximab in 500 patients 147 reported a 3-fold higher incidence of profound thrombocytopenia than with initial administration. Although human IgG anti-chimeric antibodies (HACA) are more common after abciximab readministration, 148 a clear association between HACA and thrombocytopenia has not been identified. If readministration of abciximab is considered, early platelet counts should be obtained and abciximab should be discontinued immediately if the platelet counts fall (Chapter 25). c. Emergency CABG (Table 34.24). The risk of bleeding during emergency CABG is potentiated by abciximab and heparin. 149,150 The hemostatic defect due to abciximab is largely reversible with platelet transfusions, but the benefit is not immediate or complete due to persistent platelet-bound abciximab. Transfused platelets serve as sink to remove excess drug from affected platelets, and it may take 3 hours to completely reverse the antiplatelet effect. 151,152 In EPIC, EPILOG, and EPISTENT, ll153,154 the risk of major bleeding or blood transfusion in emergency CABG patients receiving antecedent abciximab was similar to placebo, although abciximab patients required more platelet transfusions. Key factors in reducing the risk of bleeding during emergency CABG include careful titration of heparin dose and liberal use of platelet transfusions, especially after coming off cardiopulmonary bypass. d. Drug Interactions. Potential drug interactions are described in Table
4 Chapter 34. Adjunctive Pharmacotherapy 785 B. EPTIFIBATIDE (Tables 34.15, 34.16). Eptifibatide (Integrilin; manufactured by COR Therapeutics, distributed by Shering-Plough) is a synthetic cyclic K-G-D (lysine-glycine-aspartic acid) heptapeptide and a competitive antagonist for the IIb/IIIa receptor. Unlike abciximab, eptifibatide has a short halflife (2.5 hours), 155 is very specific for the IIb/IIIa receptor, and does not cross-react with the vitronectin receptor. Platelet function returns to baseline within 4 hours after drug discontinuation. 1. Eptifibatide Trials. The use of eptifibatide during coronary intervention was studied in IMPACT- II, PURSUIT, and ESPRIT (Tables ). In IMPACT-II, 156 there was a nonsignificant 19% reduction in the composite primary endpoint (death, MI, revascularization, or bailout coronary stent) at 30-days in the low-dose eptifibatide group (9.2% vs. 11.4%, p = 0.063) and in the highdose eptifibatide group (9.9% vs. 11.4%, p = 0.22). There were no differences in major bleeding or blood transfusion (Table 34.23). In PURSUIT, 10,948 patients with unstable angina or non-qwave MI received aspirin and intravenous heparin (initial bolus of 5000 U followed by 1000 U/hr to achieve aptt seconds) and were randomly assigned to eptifibatide (180 mcg/kg bolus and 2.0 mcg/kg/min infusion) or placebo. 157 A third arm using a lower dose of eptifibatide was discontinued (as specified in the original protocol) since the higher dose did not cause more bleeding complications. In a subgroup of 1,250 patients undergoing percutaneous intervention within 72 hours of randomization, eptifibatide reduced the 30-day incidence of death and MI by 31% (11.6% vs. 16.7%, p = 0.01), and the incidence of the death and MI before intervention by 69% (1.7% vs. 5.5%, p < 0.001). In ESPRIT, 158 the dose of eptifibatide was further modified to include a second bolus, 10 minutes after the first. In an elective coronary stent population, this second bolus was associated with a 37% reduction in ischemic events at 48 hours, which persisted at 30 days and at 6 months. 2. Dosage. Because IMPACT II was used to support product labeling, the FDA-approved dose of eptifibatide is an IV bolus of 135 mcg/kg immediately before intervention followed by a continuous infusion of 0.5 mcg/kg/min for hours. However, given the dosing concerns in IMPACT II and the favorable experience in PURSUIT, many physicians have adopted the higher dose employed in PURSUIT: an IV bolus of 180 mcg/kg followed by an infusion of 2 mcg/kg/min for patients with acute coronary syndromes. For therapy initiated in the cath lab (as in ESPRIT), a double bolus of eptifibatide (180 mcg/kg x 2, 10 minutes apart) is recommended, followed by an infusion of 2 mcg/kg/min. 3. Limitations. Eptifibatide did not increase the risk of major bleeding complications in IMPACT II, PURSUIT, and ESPRIT. In contrast to abciximab, there may be less need to reduce the heparin dose, there is no known immune response, and the incidence of thrombocytopenia is similar to placebo. Eptifibatide should be discontinued in patients requiring emergency CABG; antiplatelet effects will resolve in 4-6 hours and platelet transfusion is ineffective. Drug clearance is longer in patients with impaired renal function. C. TIROFIBAN (Tables 34.15, 34.16). Tirofiban (Aggrastat; Merck) is a synthetic, small-molecule nonpeptide competitive antagonist of the IIb/IIIa receptor. As with eptifibatide, it is rapidly reversible,
5 786 highly selective, and does not cross-react with the vitronectin receptor. 1. Tirofiban Trials. In RESTORE, 159,160 2,139 patients with acute coronary syndromes undergoing PTCA or DCA receiving aspirin and heparin were randomized to tirofiban (10 mcg/kg bolus and 0.15 mcg/kg/min infusion) or placebo for 36 hours. There was a 40% reduction in death, MI, and urgent revascularization (5.2% vs. 8.7%, p = 0.002) at 2 days, 30% reduction at 7 days (6.9% vs. 9.8%, p = 0.016), and 24% reduction at 30 days (8.0% vs. 10.5%, p = 0.16). The composite endpoint (death, MI, and revascularization) at 6 months was similar (24.1% with tirofiban vs. 27.1% with placebo, p = 0.11), and follow-up angiography in 417 patients showed no difference in restenosis (Tables ). 160 In PRISM-PLUS, 161 patients with acute coronary syndromes were randomized to tirofiban, heparin, or tirofiban plus heparin for hours; the tirofiban-only arm was dropped because of excess mortality at 7 days. The study was completed with 773 patients receiving tirofiban plus heparin and 797 patients receiving heparin plus placebo. The primary endpoint (death, MI, or refractory ischemia within 7 days) was significantly lower in the heparin plus tirofiban group (12.9% vs. 17.9%, p = 0.004). The composite of death and MI was also significantly lower in the heparin plus tirofiban group at 7 days, 30 days, and 6 months. The incidence of major bleeding was similar in the two groups. The recently reported TARGET trial 145 was a direct head-to-head comparison of tirofiban vs. abciximab in 4812 stent patients. Overall, tirofiban was less effective than abciximab, with a 26% relative increase in the primary endpoint of death, MI, or urgent revascularization at 30 days (7.5% vs. 6.0%, p = 0.037). In TACTICS- TIMI-18, 162 2,220 patients with unstable angina or non-st-elevation MI received aspirin, heparin, beta-blockers and tirofiban upon hospital admission, followed by randomization to invasive management (cath within 4 to 48 hours and revascularization where feasible) vs. conservative management (cath only for spontaneous or provokable ischemia). The primary endpoint of the trial (death, MI, or rehospitalization for ACS at 6 months) was reduced by 18% in the invasive arm (15.9% vs. 19.4%, p = 0.025); the beneficial effect was most pronounced in patients with positive troponins (14% vs. 24%). 2. Dosage. The dose of tirofiban used in RESTORE and TARGET was a 10 mcg/kg bolus followed by an infusion of 0.15 mcg/kg/min. In PRISM-PLUS, the dose was slightly different, a 0.4 mcg/kg/min loading infusion for 30 minutes, followed by a 0.1 mcg/kg/min maintenance infusion. In practice, physicians favor the high-dose regimen for patients undergoing coronary intervention. Therapy is usually continued for hours after completion of the case. 3. Limitations. There was no increase in major bleeding complications with tirofiban in the clinical trials (Table 34.23), 159,161 and like eptifibatide, there is no known immune response and the overall incidence of thrombocytopenia is low. If patients require emergency CABG, tirofiban should be discontinued; antiplatelet effects will resolve in 6 hours, but platelet transfusion is ineffective. Clearance is longer in patients with renal failure. D. OTHER IIb/IIIa INHIBITORS. Lamifiban, another intravenous GP IIb/IIIa antagonist, showed benefit for reducing ischemic complications in unstable angina patients in one study, 163 but was less
6 Chapter 34. Adjunctive Pharmacotherapy 787 efficacious in others. 164,165 Lamifiban is not currently under investigation as an adjunct to interventional procedures. Trials of oral GP IIb/IIIa antagonists have been disappointing (Tables 34.17, 34.20, 34.26). In EXCITE, 166 7,232 patients undergoing coronary intervention were randomized to xemilofiban or placebo. The primary endpoint (death, MI and urgent intervention) was similar at 30 days and 6 months, although there appeared to be a benefit in diabetics. In OPUS-TIMI 16, ,302 patients with unstable ischemic syndromes were randomized to oral orbofiban or placebo. Enrollment was terminated because of excess 30-day mortality in the low-dose orofiban group, although patients who underwent PCI appeared to benefit from orbofiban. In SYMPHONY 168 and 2 nd SYMPHONY 169 there was a trend toward higher mortality in patients with acute coronary syndromes receiving sibrafiban. The BRAVO 170 trial with lotrifiban was also terminated because of concerns about excess mortality. E. RECOMMENDATIONS FOR USE OF GP IIb/IIIa INHIBITORS. GP IIb/IIIa inhibitors have been extensively studied in a variety of clinical settings (Tables 34.17, 34.28). There are compelling data to support their routine use in patients undergoing percutaneous intervention and in patients with acute coronary syndromes. Recommendations in these and other clinical setting are described in Table Table Platelet Glycoprotein IIb/IIIa Receptor Antagonists Class Agent Route Type of Inhibition Monoclonal antibody (ReoPro) IV Noncompetitive Cyclic peptides RGD sequence KGD sequence Nonpeptide inhibitors Oral agents MK-852 Eptifibatide (Integrilin) Tirofiban (Aggrastat), fradafiban, lamifiban Xemilofiban, orofiban, sibrafiban, lefradafiban, lotrifiban IV IV Oral Competitive Competitive Competitive Table Comparison of Noncompetitive and Competitive Glycoprotein IIb/IIIa Inhibitors Noncompetitive* Competitive** Biological half life Long Short Duration of effect Continues after infusion During infusion Plasma half-life Short Short GP IIb/IIIa specificity Cross-reactivity with other receptors Highly specific GP IIb/IIIa binding Permanent Reversible * ** Eptifibatide, tirofiban
7 788 Table Overview of Clinical Trials of GP IIb/IIIa Inhibitors Trial IIb/IIIa Inhibitor Setting ACUTE MI - ADJUNCT TO LYTIC THERAPY TAMI-8 IMPACT-AMI PARADIGM TIMI-14A SPEED GUSTO-IV MI INTRO-AMI M7E3 Eptifibatide Lamifiban Eptifibatide t-pa t-pa t-pa, SK, r-pa t-pa, SK r-pa r-pa t-pa ACUTE MI - ADJUNCT TO PRIMARY PTCA/STENT GRAPE RAPPORT ADMIRAL CADILLAC STOP-AMI ISAR-2 Primary PTCA Primary PTCA Primary PTCA Primary stenting or PTCA Stent/abciximab vs. t-pa Primary stenting* PERCUTANEOUS CORONARY INTERVENTION EPIC EPILOG IMPACT-II RESTORE EPISTENT ESPRIT TARGET EXCITE ERASER Eptifibatide Tirofiban Eptifibatide Tirofiban vs. abciximab Xemilofiban PTCA, DCA PTCA, DCA PTCA, DCA, ROTA, ELCA High-risk PTCA, DCA PTCA, PS stent Elective stent Stent (elective and ACS) All percutaneous devices Stent (IVUS to assess restenosis) CAPTURE PURSUIT PARAGON A PARAGON B PRISM PRISM-PLUS GUSTO-IV ACS ACUTE II TACTICS (TIMI 18) UNSTABLE ANGINA/NON-ST-ELEVATION MI Integrilin Lamifiban Lamifiban Tirofiban Tirofiban Tirofiban Tirofiban ACS with intervention ACS with/without intervention ACS with/without intervention ACS with/without intervention ACS: tirofiban vs. UFH ACS with/without intervention ACS without intervention ACS: LMWH vs. UFH ACS: conservative vs. invasive Abbreviations: ACS = acute coronary syndromes; DCA = directional coronary atherectomy; ELCA = excimer laser coronary angioplasty; LMWH = low-molecular-weight heparin; PS = Palmaz-Schatz; r-pa = reteplase; ROTA = Rotablator; SK = streptokinase; t-pa = tissue plasminogen activator; UFH = unfractionated heparin * Included patients with acute MI < 48 hours Acronyms: See Table 34.31, p. 803
8 Chapter 34. Adjunctive Pharmacotherapy 789 Table Major Trials of IIb/IIIa Antagonists for Coronary Intervention in Acute MI Study Antagonist 1 Endpoint Result Comments CADILLAC 143 (n = 2082) (PTCA vs. stent) ischemia-driven TVR at 6 months PTCA: 19.3% PTCA/abcix: 15.2% Stent: 10.9% Stent/abcix: 10.8% (p < ) Stenting superior to PTCA; adverse effects of stents on TIMI flow and late mortality in PAMI-STENT not observed in CADILLAC. was associated with a trend toward better event-free survival in PTCA patients, but no benefit in stent patients RAPPORT 136 (n = 483) (primary PTCA) revascularization at 6 months Placebo: 16.1% : 13.3% (p = 0.32) No benefit for abciximab in primary PTCA patients ADMIRAL 142 (n = 299) (stent 85%, PTCA 15%) ischemia-driven TVR at 30 days Placebo: 20% : 10.7% (p < 0.03) Substantial benefit for abciximab in primary stent patients Munich Trial 222 (n = 200) (primary stenting) Hospital death, MI, revascularization Placebo: 9.2% : 2.0% (p < 0.05) group also had better regional wall motion, global LV function, and peak coronary blood flow. Short-term benefits on primary endpoint were no longer significant at 30 days (p = 0.16) ISAR (n = 401) (primary stenting) Angiographic restenosis No effect on restenosis led to reduction in 30-day MACE (5.0% vs. 10.5%). No additional benefit over time EPIC 181 (n = 64) (direct or rescue PTCA) urgent revascularization at 30 days 30-day: Placebo 26.1% vs. abciximab + 4.8% (p = 0.06) 6-month: Placebo 47.8% vs. abciximab + 4.5% (p = 0.002) Study group represented 64 of 2099 patients in EPIC with acute MI; 42 direct PTCA, 22 rescue PTCA GRAPE 163 (n = 60) (PTCA) Infarct artery patency TIMI-3: 18% before primary PTCA is safe RESTORE 159,160 (n = 134) Tirofiban (primary PTCA) any revascularization 56% reduction at 7 days and 22% reduction at 30 days (p = NS) Study group represented 134 of 2141 patients in RESTORE with acute MI Abbreviations: IVUS = intravascular ultrasound; MI = myocardial infarction; TVR = target vessel revascularization + Bolus plus infusion Acronyms: See Table 34.31, p. 803
9 790 Table Major Trials of IIb/IIIa Antagonists for Acute Coronary Syndromes Study Antagonist Setting Composite Events (30 days) Result (Drug vs. Placebo) RRR (%) PURSUIT 157 (n = 10,984) CANADIAN LAMIFIBAN STUDY 163 (n = 365) Eptifibatide ACS Death, MI 14.2% vs 15.7% (p = 0.04) 10 Lamifiban ACS Death, MI 3.7% vs 8.1% (p = 0.07) 54 PARAGON A 164,189 (n = 2282) Lamifiban ACS Death, MI Placebo: 11.7% Low-dose lamifiban: 10.6%* High-dose lamifiban: 12.0%* - PARAGON B 190 (n = 5225) Lamifiban ACS Death, MI, refractory ischemia 11.8% vs. 12.8%* 8 PRISM 191 (n = 3232) Tirofiban ACS Death, MI, recurrent ischemia 36% risk reduction (p = 0.007) at 48 hours was not maintained at 30 days (15.9% vs. 17.1%*) 7 PRISM-PLUS 161 (n = 1915) Tirofiban High-risk ACS Death, MI, recurrent ischemia 18.5% vs 22.3% (p = 0.03) 17 TACTICS 162 (n = 2220) Tirofiban (all pts) ACS: invasive vs. conservative Death, MI, rehospitalization Invasive 7.4% vs. conservative 10.5% (p < 0.001); benefit persisted at 6 months (18% risk reduction, p = 0.025) 30 CAPTURE 134 (n = 1265) ACS Death, MI, rehospitalization 11.3% vs. 15.9% (p = 0.012) 29 GUSTO-IV ACS 144 (n = 7800) ACS Death, MI Placebo: 8.0% (24-hr): 8.2%* (48-hr): 9.1%* - ACUTE-II 75 Tirofiban ACS: LMWH vs. UFH Death, MI, recurrent ischemia, ischemic CVA Tirofiban, LMWH: 11.7 Tirofiban, UFH: Abbreviations: ACS = acute coronary syndromes; LMWH = low-molecular-weight heparin; MI = myocardial infarction; PCI = percutaneous coronary intervention ; RRR = relative risk reduction; UFH = unfractionated heparin Acronyms: See Table 34.31, p. 803 * p = NS
10 Chapter 34. Adjunctive Pharmacotherapy 791 Table Major Trials of IIb/IIIa Antagonists for Coronary Intervention in Acute Coronary Syndromes or Elective Indications Study Antagonist 1 Endpoint Result Comments EPIC 131,132,138 (n = 2099) EPILOG 133,136,137 (n = 2792) CAPTURE 134 (n = 1265) EPISTENT 135 (n = 2399) ERASER 139 (n = 225) IMPACT-II 156 (n = 4010) ESPRIT 158 (n = 2064) RESTORE 159,160 (n = 2141) TARGET 145 (n = 4802) EXCITE 166 (high risk; PTCA or atherectomy) (PCI) (PCI for refractory unstable angina) (stent) (elective stent) Eptifibatide (PCI) Eptifibatide (elective stent) Tirofiban (PCI for unstable angina) Tirofiban vs. abciximab (stent) Xemilofiban (PCI) urgent TVR at 30 days urgent TVR at 30 days urgent TVR at 30 days urgent TVR at 30 days In-stent restenosis at 6 months (by IVUS) urgent TVR at 30 days urgent TVR at 48 hours any TVR at 48 hours ischemia-driven TVR at 30 days urgent TVR at 30 days Placebo: 12.8% Bolus alone: 11.4% Bolus/infusion: 8.3% (p = 0.008) Placebo: 11.7% SD heparin: 5.2% LD heparin: 5.4% (p < 0.001) Placebo: 15.9% : 11.3% (p = 0.012) Stent alone: 10.8% PTCA/abcix: 6.9% Stent/abcix: 5.3% (p < 0.001) Placebo: 32.4% 12-hr abcix: 37.7% 24-hr abcix: 34.2% (p = NS) Placebo: 11.4% LD eptifib: 9.2% (p = 0.062) HD eptifib: 9.9% (p = 0.22) Placebo: 10.4% Eptifibatide: 6.8% (p = ) Placebo: 8.7 % Tirofiban: 5.4% (p = 0.005) Tirofiban: 7.6% : 6.0% (p = 0.037) Placebo: 8.1% 10 mg: 8.1% 20 mg: 7.3% (p = NS) Short-term benefits persisted at 6 months (RRR 23%) and 3 years (RRR 13%); first trial to show improved survival by decreasing risk of postprocedural CK elevation. Increased bleeding with abciximab Persistent benefit at 6 months (RRR 43%, p < 0.05). This study extended the benefits of abciximab to low-risk patients and proved that a reduction in heparin dose reduced bleeding complications without sacrificing efficacy More major bleeding with abciximab; no difference in outcome at 6-months, suggesting the need for 12-hour drug infusion postintervention No increase in bleeding; significant benefit as an adjunct to stenting; mortality benefit at 1 year No restenosis benefit for abciximab after stenting using IVUS criteria No difference in bleeding; insignificant trend toward benefit with low-dose eptifibatide may have been due to inadequate drug dose Slight increase in bleeding; modified double bolus effective in reducing short-term events in low-risk patients (RRR 35% at 30-days, p = 0.003). Long-term data not yet available No difference in bleeding; used different endpoints that abciximab trials; tirofiban with persistent (but not significant) benefit at 30-days (RRR 16%) Comparable bleeding. Tirofiban was inferior to abciximab, but may be an alternative in low-risk patients without acute coronary syndromes Oral xemilofiban had no benefit; diabetics had slight benefit with xemilofiban Abbreviations: HD = high-dose; IVUS = intravascular ultrasound; LD = low-dose; MI = myocardial infarction; RRR = relative risk reduction; SD = standard dose; TVR = target vessel revascularization Acronyms: See Table 34.31, p. 803
11 792 Table Major Trials of IIb/IIIa Antagonists as Adjuncts to Lytics for Acute MI Study Antagonist Setting Composite Events Results (Drug vs. Placebo) TAMI (n = 70) Acute MI (with t-pa) Rest angina, ECG changes, reinfarction, urgent revascularization, and death at 30 days 13% vs 20%; no excess bleeding in patients receiving t-pa and abciximab TIMI (n = 681) Acute MI (with SK or t-pa) TIMI-3 flow at 90 minutes t-pa + abciximab: 72% Lytic therapy alone: 43% SPEED 186 (n=305) Acute MI (with r-pa) TIMI-3 flow at minutes alone: 28% r-pa + abciximab: 63% GUSTO-IV MI Acute MI Pending STOP-AMI 188 (n = 140) Acute MI (stent + abciximab vs. t-pa Death, MI, stroke at 6 months; infarct size 8.5% vs. 23.2% (p = 0.02); smaller infarct size with stent/abciximab (14.3% vs. 19.4% of LV) Intro-AMI 187 (n=342) Eptifibatide Acute MI (with t-pa) TIMI-3 flow at 60 minutes Better results when eptifibatide given after t-pa; more complete lysis with 50 mg t-pa IMPACT-AMI 184 (n = 180) Eptifibatide Acute MI (with t-pa) TIMI-3 flow at 90 minutes 66% vs 39% (p = 0.006) PARADIGM 165 Lamifiban (n = 353) Acronyms: See Table 34.31, p. 803 Acute MI (with t-pa) Reperfusion (by continuous ECG at 90 minutes) 80.1% vs 62.5% (p = 0.005) Table Effects of Effect Antiplatelet activity Anticoagulant activity Thrombolytic activity Antiproliferative activity Anti-inflammatory activity Mechanism Inhibits platelet cross-linking by fibrinogen Inhibits clot retraction; prolongs ACT Inhibits factor XIII and PAI-1; displaces fibrinogen Nonselective binding to vitronectin receptor Nonselective binding to Mac-1 receptor on white blood cells Abbreviations: ACT = activated clotting time; PAI = plasminogen activator inhibitor; Mac-1 = CR3 integrin
12 Chapter 34. Adjunctive Pharmacotherapy 793 Table Bleeding Complications in Trials of GPIIb/IIIa Inhibitors Bleeding Trial Agent Major Minor Transfusion EPIC 131 (n = 2099) (bolus + infusion) (bolus) Placebo p-value < < < 0.01 IMPACT-II 156 (n = 4010) Eptifibatide (135/0.5)* Eptifibatide (135/0.75)* Placebo p-value NS NS NS CAPTURE 134 (n = 1265) Placebo p-value < < 0.01 RESTORE 159 (n = 2139) Tirofiban Placebo p-value NS EPILOG 133 (n = 2792) (std. dose heparin) (low-dose heparin) Placebo p-value NS < NS EPISTENT 135 (n = 2399) Stent (placebo) Stent (abciximab) PTCA (abciximab) p-value NS NS NS Acronyms: See Table 34.31, p. 803
13 794 Table Safety Issues Related to IIb/IIIa Inhibitors Bleeding Issue Recommendations Reduce Preprocedural Risk Factors: Identify clinical risk factors (acute MI, low body weight, advanced age). Identify contraindications (stroke within 2 years, neurosurgery within 6 months, clinically significant GI or GU bleeding with 6 weeks, active internal bleeding, bleeding diathesis, intracranial neoplasm, AV malformation, or aneurysm, severe uncontrolled hypertension, major surgery or trauma within 6 weeks) For warfarin patients, INR should be < 1.5 Enhance Procedural Safety: Use weight-adjusted heparin (70 U/kg) and abciximab to keep ACT seconds; for other IIb/IIIa inhibitors, see Figure 34.1 Single-wall arterial puncture Avoid venous sheaths if possible Use different arterial access site if recent arterial access was achieved (< 2 weeks) Avoid lytic therapy if possible Postprocedure Care: Avoid post-procedural heparin Sheaths may be removed during infusion of the IIb/IIIa inhibitor when aptt < 50 seconds or ACT < 175 seconds. Apply pressure to access site for at least 30 minutes; check site and distal pulses every 15 minutes for 1 hour, then hourly for 6 hours. Keep patient at complete bed rest with head elevated < 30 for 6-8 hours after discontinuation of IIb/IIIa infusion Nursing care: Avoid IM injections, Foley catheters, NG tubes, and non-compressible sites (e.g., subclavian or jugular vein) for venous access if possible; consider heparin locks for blood drawing Emergency CABG Major hemorrhage Thrombocytopenia Discontinue infusion of drug and heparin Obtain ACT in operating room; adjust heparin dose to target ACT (400 sec). Heparin bolus based on ACT: ACT < 200 sec (heparin 4000 U); ACT sec (heparin 3500 U); ACT sec (heparin 2500 U); ACT sec (heparin 1500 U); ACT sec (heparin 1000 U); ACT > 400 sec (no additional heparin) Prophylactic platelet transfusion (6-10 units) Liberal platelet transfusions after separation from cardiopulmonary bypass Discontinue drug and heparin Platelet transfusions (6-10 units) for abciximab patients Blood transfusions as necessary Identify and correct cause Platelets > 20,000/mm 3 and no Bleeding (Figure 25.1, Table 25.19): Stop drug and other agents Repeat platelet count every 6 hours If platelet count is not normal within 2-3 days, evaluate other causes Platelets < 20,000/mm 3 : Stop drug and other agents Consider prophylactic platelet transfusion (6-10 units) Repeat platelet counts every 6 hours
14 Chapter 34. Adjunctive Pharmacotherapy 795 Table Thrombocytopenia Due to or Heparin (First Exposure) (Re-exposure) Heparin Onset Acute; onset within hours of exposure Immediate onset within hours of exposure Subacute; onset within days of exposure Platelet count Frequently 20,000-50,000/mm 3 ; sometimes < 20,000/mm 3 Frequently < 20,000/mm 3 Usually > 50,000/mm 3 (except HIT-2) Clinical sequelae Bleeding, particularly when platelet count < 20,000/mm 3 Bleeding is common if not identified early Thrombosis with HIT- 2; bleeding is less common Recovery Immediate increase in platelet count 20,000/mm 3 per day after drug discontinuation Initial increase may be followed by subsequent decrease in platelet count after drug discontinuation Slow recovery is common after drug discontinuation Treatment Stop abciximab; platelet transfusions are indicated for bleeding or platelet count < 20,000/mm 3 ; IgG does not enhance recovery of platelet count May not be immediately responsive to initial platelet transfusion; repeat transfusions may be needed; the role of IgG has not been evaluated Stop heparin; platelet transfusions are not useful. Laboratory evaluation Serial platelet counts; identification of abciximab antibodies can be arranged through manufacturer; HACA antibodies may identify patients at risk for thrombocytopenia during readministration Serial platelet counts; identification of abciximab antibodies can be arranged through manufacturer Serial platelet counts (Chapter 25) Abbreviations: HACA = human anti-chimeric antibodies; HIT = heparin-induced thrombocytopenia
15 796 Table Drug Interactions with Drug Clinical Impact Recommendations Aspirin No increase in risk of bleeding Routinely administered with abciximab; no dose adjustment required Warfarin Heparin About 40% of patients with intracranial hemorrhage while on abciximab occurred in patients on warfarin with INR > 1.5 Potentiates bleeding, particularly at vascular access sites Avoid abciximab in warfarin-treated patients with INR > 1.5. If abciximab is mandatory, use FFP (2 units) before abciximab administration Use low-dose weight-adjusted heparin (70 U/kg) and prophylactic (rather than "rescue") abciximab; maintain ACT seconds. Avoid post-procedural heparin Thrombolytics Potentiates bleeding Avoid intraprocedural thrombolytic agents, if possible. For lytic patients with acute MI or rescue-ptca patients after failed lytics, follow heparin recommendations above Ticlopidine Clopidogrel Weak experimental synergy, but bleeding is not increased Clopidogrel monotherapy probably does not increase risk of bleeding. When clopidogrel is given with aspirin, the risk of bleeding is somewhat increased with abciximab No dose adjustment No dose adjustment. Routinely administered with aspirin and abciximab in stent patient Abbreviations: ACT = activated clotting time; FFP = fresh frozen plasma; INR = international normalized ratio
16 Chapter 34. Adjunctive Pharmacotherapy 797 Table Major Trials of Oral GP IIb/IIIa Antagonists Study Drug Setting Endpoints Results Kereiakes 192 (n = 17) Xemilofiban up to 4 weeks (procedural heparin, aspirin, abciximab) PCI Platelet inhibition; safety Magnitude and extent of platelet inhibition enhanced with combination therapy; no increase in major bleeding Simpendorfer 193 (n = 30) Xemilofiban up to 30 days (procedural heparin and aspirin) PCI for unstable angina Platelet inhibition; safety Mucocutaneous bleeding. Severe bleeding in 4 patients (1 death after emergency CABG) Muller 194 (n = 104) Fradafiban or lefradafiban up to 7 days (no aspirin) Healthy volunteers Platelet inhibition Continued profound platelet inhibition with oral drug administration TIMI (n = 329) Sibrafiban up to 28 days (no aspirin) ACS Platelet inhibition; safety Dose-related increase in platelet inhibition; high incidence of minor bleeding ORBIT 196 (n = 549) Xemilofiban up to 4 weeks (with aspirin) After PCI Platelet inhibition; safety Dose-related increase in platelet inhibition; no intracranial hemorrhage; low incidence of major bleeding SOAR 197,198 (n = 259) Orbofiban up to 3 months (with aspirin) ACS Platelet inhibition; safety Dose-related increase in platelet inhibition; no increase in major bleeding; frequent insignificant/minor bleeding TIMI (n = 192) Klerval (IV for hrs, oral up to 4 weeks) ACS Platelet inhibition; safety Relatively high incidence of thrombocytopenia (13%); limited dose response. Less inhibition with oral drug despite similar receptor occupancy EXCITE 166 (n = 7232) Xemilofiban up to 6 months (procedural heparin and aspirin) PCI Death, MI, urgent intervention Placebo vs. xemilofiban 10 mg vs. xemilofiban 20 mg: 30 days (8.1% vs. 8.1% vs. 7.3%, p = NS); 6 months (13.6% vs. 14.1% vs. 12.6%, p = NS). Trend towards higher mortality in low-dose xemilofiban group OPUS-TIMI (n = 10,302) Orbofiban up to 7 months (with aspirin) ACS Death, MI, ischemiadriven or urgent TVR, stroke Placebo vs. orofiban 50/30 vs. orofiban 50/50: 30 days (10.7% vs. 9.7% vs. 9.3%, p = NS); 7 months (20.5% vs. 20.2% vs. 19.5%, p = NS). Trial halted because of significant excess mortality in low-dose group (1.4% vs. 2.3% vs. 1.6%)
17 798 Table Major Trials of Oral GP IIb/IIIa Antagonists Study Drug Setting Endpoints Results FROST 200 (n = 531) Lefradafiban up to 1 month (with heparin, aspirin) ACS Bleeding, death, MI, ischemia-driven TVR High dose (45 mg TID) stopped because of excess major bleeding; no significant difference in composite events; trend toward fewer recurrent ischemic events with 30 mg TID dose SYMPHONY 168 (n = 9233) Sibrafiban up to 3 months (without aspirin) ACS Death, MI, revascularization Aspirin: 9.8% Low-dose sibrafiban: 10.1% High-dose sibrafiban: 10% 2 nd SYMPHONY 169 (n = 6671) Aspirin vs. aspirin plus lowdose sibrafiban vs. high-dose sibrafiban alone ACS Death, MI, severe recurrent ischemia [Trial discontinued after SYMPHONY results available] Aspirin: 9.2 % Aspirin + sibrafiban: 9.3% Sibrafiban alone: 10.5% BRAVO 170 (n = 9200) Lotrifiban vs. placebo (with aspirin) Recent ACS; recent neurologic event Death, MI, stroke, recurrent ischemic TVR Trial discontinued because of excess mortality (2.7% vs. 2.0%, p = 0.022) Abbreviations: ACS = acute coronary syndromes; CABG = coronary artery bypass surgery; MI = myocardial infarction; PCI = percutaneous coronary intervention; TVR = target vessel revascularization Acronyms: See Table 34.31, p. 803
18 Chapter 34. Adjunctive Pharmacotherapy 799 Table Results of Rescue IIb/IIIa Inhibitors and Hirulog for Thrombus ESPIRT 223 (2001) Fuchs 228 (2000) Velianou 224 (2000) de Lemos 225 (2000) Series N Adjunctive Therapy Results Piamsomboon 226 (1999) Ahmed (1999) Fuchs 206 (1999) Haase 208 (1999) Sullebarger 207 (1999) Barsness (1998) Garbarz 214 (1998) Grantham 227 (1998) Henry 213 (1998) Muhlestein 212 (1997) Shah 215 (1997) 77 Rescue eptifibatide for thrombotic complications (42%) 298 Rescue abciximab for thrombus, dissection Type > C, no-reflow, suboptimal result, or distal embolization 186 (planned 45%; rescue for threatened or acute closure 55%) 92 Rescue abciximab (n = 29) vs. no abciximab (n = 63); TIMI 14 substudy 73 (planned 74%; rescue 26%) in acute coronary syndromes with thrombus-containing lesions 45 Rescue abciximab in degenerated SVG 186 Rescue abciximab for thrombus and suboptimal PTCA 63 Rescue abciximab for thrombus and threatened or acute closure 17 Planned abciximab and TEC in SVG 58 Local delivery of abciximab in thrombotic SVG 138 Rescue abciximab for thrombus and suboptimal PTCA 185 Planned and unplanned (rescue) abciximab in thrombotic SVG 16 Rescue abciximab for acute stent thrombosis 29 Rescue abciximab for thrombus after PCI 567 Hirulog vs. heparin for thrombuscontaining lesions (Hirulog Angioplasty Study) Bailout therapy was associated with more MI (30% vs. 7%, p = 0.01) and urgent PCI at 48 hours (14% vs. 0%, p = 0.03) compared to no bailout Stents in 73%. In-hospital results: death (1.3%); Q-MI (0.7%); non-q-mi (31%); TLR (4.6%). Late events: death (1.7%); MI (2.7%); TVR (15.1%); EFS at 1-yr (83%) In-hospital results (planned vs. rescue): death (1.2% vs. 1.0%); Q-MI (2.4% vs. 2.0%); non-q-mi (7% vs. 12.9%); TLR (1.2% vs. 0%). 6-month results (planned vs. rescue): death (2.3% vs. 4.0%); MI (9.4% vs. 14.9%); TVR (4.7% vs. 20.8%, p = 0.001) group had greater ST-segment resolution minutes after PCI Death (1.4%); Q-wave MI (1.4%); non-q-wave MI (18%). No subacute thrombosis, emergency CABG, or repeat PTCA No benefit on procedural success or major ischemic complications compared to no-abciximab ; more non-q-mi in rescue group (40.5% vs. 17.7%) Procedural success (95.3%); in-hospital MACE (4.7%); non-q- MI (24.7%); 1-year TLR (24.9%) and EFS (71%) Repeat PTCA 2 minutes after abciximab. Marked improvement in thrombus score and TIMI flow; MACE (2%); late TVR (15%) was associated with higher procedural success (100% vs. 50%) and less distal embolization/no-reflow (0% vs. 30%) compared to no abciximab Significant improvement in thrombus score and stenosis severity, but not TIMI flow. Adjunctive stent (88%) Angiographic success in 84% (100% success for stent thrombosis); adjunctive PTCA or stent (100%); MACE (17%); bleeding and vascular complications (27%); transfusion (5%) Procedural success higher with planned abciximab; no impact of abciximab on angiographic (distal embolization, no-reflow) or clinical complications Recurrent thrombosis after PTCA for stent thrombosis may respond to abciximab Procedural success (97%), clinical success (93%). Significant improvement in thrombus score and TIMI flow, without distal embolization or no-reflow Thrombus-containing lesions were associated with more MI (5.1% vs. 3.2%) and abrupt closure (13.6% vs. 8.3%); no difference between hirulog and heparin in acute or late outcomes Abbreviations: SVG = saphenous vein graft; Q-MI = Q-wave myocardial infarction; MACE = major adverse cardiac events; TLR= target lesion revascularization; EFS = event-free survival; PCI = percutaneous coronary intervention; TIMI = Thrombolysis in Myocardial Infarction
19 800 Table Recommendations for Platelet Glycoprotein II/IIIa Antagonists Clinical Setting Acute MI (lytic therapy) Acute MI (primary PTCA/Stent) Acute MI (rescue PTCA after failed lytic therapy) Unstable angina, non-qwave MI Percutaneous coronary intervention Rescue use after failed or suboptimal intervention Readministration Recommendations (TIMI-3 flow, ischemic events at 30-days), eptifibatide (TIMI-3 flow, ischemic events at 30-days), and lamifiban (clinical reperfusion but not TIMI flow) all show modest benefit as adjuncts to lytic therapy for ST-elevation acute MI. Further study is needed before GP IIb/IIIa antagonists are routinely used as adjuncts to lytic therapy for acute MI. GUSTO-IV MI results pending. Trials of abciximab as an adjunct to primary PTCA (RAPPORT, CADILLAC) and stenting (ADMIRAL, CADILLAC) demonstrated variable benefit. In stent patients, data from ADMIRAL are favorable, while data from CADILLAC are not. In PTCA patients, data from CADILLAC are favorable, while data from RAPPORT are not. IIb/IIIa inhibitors reduce acute thrombotic events, but routine use in acute MI is controversial. Available anecdotal data in small numbers of patients suggest potential benefit for abciximab; further study is needed. There are compelling data to support the routine use of abciximab, eptifibatide, and tirofiban in patients with acute coronary syndromes without ST-elevation referred for PCI. As adjuncts to heparin, aspirin, and β-blockers, these agents decrease early (30-day) cardiac event rates. Benefits are established for tirofiban and eptifibatide in patients not undergoing PCI, but GUSTO-IV ACS failed to show benefit for abciximab. There are compelling data to support the routine use of abciximab, eptifibatide, or tirofiban as an adjunct to heparin and aspirin in patients undergoing PCI. Only abciximab has shown long-term (> 1 year) mortality benefit in stent patients. Eptifibatide was effective in ESPRIT in reducing short- and mid-term (48-hour, 30-day, 6-month) events. Tirofiban was inferior to abciximab in TARGET. The value of GP IIb/IIIa antagonists as pretreatment before percutaneous intervention is irrefutable; post-hoc use has not been prospectively studied. Several reports in small numbers of patients suggest potential benefit for "rescue" use in thrombus-containing lesions, stent thrombosis, and abrupt closure, but less benefit in degenerated vein grafts. Disadvantage of rescue or post-hoc use is that antecedent high-dose heparin may increase bleeding complications. Readministration is not a concern with tirofiban or eptifibatide. Modest experience with abciximab suggests readministration is feasible, but the risk of severe thrombocytopenia may be higher than after initial exposure. Readministration within 2 weeks of initial exposure is probably safe (before development of HACA). For later re-exposure, H 2 - blockers and steroids are not necessary, but close monitoring of platelet counts is recommended to identify thrombocytopenia. Anaphylactic reactions have not been reported. Acronyms: See Table 34.31, p. 803
Update on Antithrombotic Therapy in Acute Coronary Syndrome
 Update on Antithrombotic Therapy in Acute Coronary Syndrome Laura Tsang November 13, 2006 Objectives: By the end of this session, you should understand: The role of antithrombotics in ACS Their mechanisms
Update on Antithrombotic Therapy in Acute Coronary Syndrome Laura Tsang November 13, 2006 Objectives: By the end of this session, you should understand: The role of antithrombotics in ACS Their mechanisms
An update on the management of UA / NSTEMI. Michael H. Crawford, MD
 An update on the management of UA / NSTEMI Michael H. Crawford, MD New ACC/AHA Guidelines 2007 What s s new in the last 5 years CT imaging advances Ascendancy of troponin and BNP Clarification of ACEI/ARB
An update on the management of UA / NSTEMI Michael H. Crawford, MD New ACC/AHA Guidelines 2007 What s s new in the last 5 years CT imaging advances Ascendancy of troponin and BNP Clarification of ACEI/ARB
Facilitated Percutaneous Coronary Intervention in Acute Myocardial Infarction. Is it beneficial to patients?
 Facilitated Percutaneous Coronary Intervention in Acute Myocardial Infarction Is it beneficial to patients? Seung-Jea Tahk, MD. PhD. Suwon, Korea Facilitated PCI.. background Degree of coronary flow at
Facilitated Percutaneous Coronary Intervention in Acute Myocardial Infarction Is it beneficial to patients? Seung-Jea Tahk, MD. PhD. Suwon, Korea Facilitated PCI.. background Degree of coronary flow at
Chapter 15 Glycoprotein IIb/IIIa Antagonists
 Chapter 15 Glycoprotein IIb/IIIa Antagonists Introduction - GP IIb/IIIa receptors - Pharmacologic approaches - Preparations and dosages Mechanism of Action Clinical Efficacy - UA / NSTEMI - STEMI - PCI
Chapter 15 Glycoprotein IIb/IIIa Antagonists Introduction - GP IIb/IIIa receptors - Pharmacologic approaches - Preparations and dosages Mechanism of Action Clinical Efficacy - UA / NSTEMI - STEMI - PCI
Platelet Glycoprotein IIb/IIIa Antagonists
 REVIEW ARTICLE David C. Warltier, M.D., Ph.D., Editor Anesthesiology 2002; 96:1237 49 2002 American Society of Anesthesiologists, Inc. Lippincott Williams & Wilkins, Inc. Platelet Glycoprotein IIb/IIIa
REVIEW ARTICLE David C. Warltier, M.D., Ph.D., Editor Anesthesiology 2002; 96:1237 49 2002 American Society of Anesthesiologists, Inc. Lippincott Williams & Wilkins, Inc. Platelet Glycoprotein IIb/IIIa
Treatment Strategies for the Prevention of Ischemic Complications in Patients Undergoing Percutaneous Coronary Intervention with Stent Placement
 Treatment Strategies for the Prevention of Ischemic Complications in Patients Undergoing Percutaneous Coronary Intervention with Stent Placement Pharmaceutical Care Project Outcomes Literature Evaluation
Treatment Strategies for the Prevention of Ischemic Complications in Patients Undergoing Percutaneous Coronary Intervention with Stent Placement Pharmaceutical Care Project Outcomes Literature Evaluation
The use of percutaneous coronary intervention (PCI) as
 Antithrombotic Therapy During Percutaneous Coronary Intervention The Seventh ACCP Conference on Antithrombotic and Thrombolytic Therapy Jeffrey J. Popma, MD; Peter Berger, MD; E. Magnus Ohman, MD, FCCP;
Antithrombotic Therapy During Percutaneous Coronary Intervention The Seventh ACCP Conference on Antithrombotic and Thrombolytic Therapy Jeffrey J. Popma, MD; Peter Berger, MD; E. Magnus Ohman, MD, FCCP;
Antiplatelet and Antithrombotic Therapies in PCI Defining the Optimal Strategy
 Antiplatelet and Antithrombotic Therapies in PCI Defining the Optimal Strategy Franz-Josef Neumann Herz-Zentrum Bad Krozingen Antiplatelet and Antithrombotic Therapies in PCI Defining the Optimal Strategy
Antiplatelet and Antithrombotic Therapies in PCI Defining the Optimal Strategy Franz-Josef Neumann Herz-Zentrum Bad Krozingen Antiplatelet and Antithrombotic Therapies in PCI Defining the Optimal Strategy
Myocardial Infarction In Dr.Yahya Kiwan
 Myocardial Infarction In 2007 Dr.Yahya Kiwan New Definition Of Acute Myocardial Infarction The term of myocardial infarction should be used when there is evidence of myocardial necrosis in a clinical setting
Myocardial Infarction In 2007 Dr.Yahya Kiwan New Definition Of Acute Myocardial Infarction The term of myocardial infarction should be used when there is evidence of myocardial necrosis in a clinical setting
S E C T I O N. Cardiovascular Medicine
 S E C T I O N I V Cardiovascular Medicine 102 CME 2004 C h a p t e r 1 5 Gp IIb/IIIa Receptor Antagonists in Intensive Coronary Care Unit Lekha Adik-Pathak 1, Manjeet S. Juneja 2, Shivkumar Bhairappa 3
S E C T I O N I V Cardiovascular Medicine 102 CME 2004 C h a p t e r 1 5 Gp IIb/IIIa Receptor Antagonists in Intensive Coronary Care Unit Lekha Adik-Pathak 1, Manjeet S. Juneja 2, Shivkumar Bhairappa 3
ACUTE CORONARY SYNDROMES
 CLINICAL CARDIOLOGY Current Role of Platelet Glycoprotein IIb/IIIa Inhibitors in Acute Coronary Syndromes Deepak L. Bhatt, MD Eric J. Topol, MD ACUTE CORONARY SYNDROMES (ACSs) span the pathological continuum
CLINICAL CARDIOLOGY Current Role of Platelet Glycoprotein IIb/IIIa Inhibitors in Acute Coronary Syndromes Deepak L. Bhatt, MD Eric J. Topol, MD ACUTE CORONARY SYNDROMES (ACSs) span the pathological continuum
Scottish Medicines Consortium
 Scottish Medicines Consortium bivalirudin 250mg for injection or infusion (Angiox ) (156/05) Nycomed UK Ltd No. 4 February, 2005 The Scottish Medicines Consortium has completed its assessment of the above
Scottish Medicines Consortium bivalirudin 250mg for injection or infusion (Angiox ) (156/05) Nycomed UK Ltd No. 4 February, 2005 The Scottish Medicines Consortium has completed its assessment of the above
NURSING DEPARTMENT CRITICAL CARE POLICY MANUAL CRITICAL CARE PROTOCOLS EPTIFIBATIDE (INTEGRILIN) PROTOCOL
 NURSING DEPARTMENT CRITICAL CARE POLICY MANUAL CRITICAL CARE S EPTIFIBATIDE (INTEGRILIN) I. PURPOSE: A. Integrilin (Eptifibatide) is a specific and potent inhibitor of the platelet receptor glycoprotein
NURSING DEPARTMENT CRITICAL CARE POLICY MANUAL CRITICAL CARE S EPTIFIBATIDE (INTEGRILIN) I. PURPOSE: A. Integrilin (Eptifibatide) is a specific and potent inhibitor of the platelet receptor glycoprotein
Index. Note: Page numbers of article titles are in boldface type.
 Cardiol Clin 24 (2006) 299 304 Index Note: Page numbers of article titles are in boldface type. A Abciximab in PCI, 180 182 ACE trial, 182 Actinomycin D-eluting stent, 224 ACUITY trial, 190 Acute myocardial
Cardiol Clin 24 (2006) 299 304 Index Note: Page numbers of article titles are in boldface type. A Abciximab in PCI, 180 182 ACE trial, 182 Actinomycin D-eluting stent, 224 ACUITY trial, 190 Acute myocardial
Optimal antiplatelet and anticoagulant therapy for patients treated in STEMI network
 Torino 6 Joint meeting with Mayo Clinic Great Innovation in Cardiology 14-15 Ottobre 2010 Optimal antiplatelet and anticoagulant therapy for patients treated in STEMI network Diego Ardissino Ischemic vs
Torino 6 Joint meeting with Mayo Clinic Great Innovation in Cardiology 14-15 Ottobre 2010 Optimal antiplatelet and anticoagulant therapy for patients treated in STEMI network Diego Ardissino Ischemic vs
Learning Objectives. Epidemiology of Acute Coronary Syndrome
 Cardiovascular Update: Antiplatelet therapy in acute coronary syndromes PHILLIP WEEKS, PHARM.D., BCPS-AQ CARDIOLOGY Learning Objectives Interpret guidelines as they relate to constructing an antiplatelet
Cardiovascular Update: Antiplatelet therapy in acute coronary syndromes PHILLIP WEEKS, PHARM.D., BCPS-AQ CARDIOLOGY Learning Objectives Interpret guidelines as they relate to constructing an antiplatelet
FACTOR Xa AND PAR-1 BLOCKER : ATLAS-2, APPRAISE-2 & TRACER TRIALS
 New Horizons In Atherothrombosis Treatment 2012 순환기춘계학술대회 FACTOR Xa AND PAR-1 BLOCKER : ATLAS-2, APPRAISE-2 & TRACER TRIALS Division of Cardiology, Jeonbuk National University Medical School Jei Keon Chae,
New Horizons In Atherothrombosis Treatment 2012 순환기춘계학술대회 FACTOR Xa AND PAR-1 BLOCKER : ATLAS-2, APPRAISE-2 & TRACER TRIALS Division of Cardiology, Jeonbuk National University Medical School Jei Keon Chae,
Acute coronary syndromes refers to a spectrum of
 Appropriate Use of Glycoprotein IIb/IIIa Blockade for Unstable Angina and Non ST Segment Elevation Myocardial Infarction Albert W. Chan, MD, MS, FRCP(C), and Sorin J. Brener, MD Objective: To review the
Appropriate Use of Glycoprotein IIb/IIIa Blockade for Unstable Angina and Non ST Segment Elevation Myocardial Infarction Albert W. Chan, MD, MS, FRCP(C), and Sorin J. Brener, MD Objective: To review the
Guideline for STEMI. Reperfusion at a PCI-Capable Hospital
 MANSOURA. 2015 Guideline for STEMI Reperfusion at a PCI-Capable Hospital Mahmoud Yossof MANSOURA 2015 Reperfusion Therapy for Patients with STEMI *Patients with cardiogenic shock or severe heart failure
MANSOURA. 2015 Guideline for STEMI Reperfusion at a PCI-Capable Hospital Mahmoud Yossof MANSOURA 2015 Reperfusion Therapy for Patients with STEMI *Patients with cardiogenic shock or severe heart failure
Management of Acute Myocardial Infarction
 Management of Acute Myocardial Infarction Prof. Hossam Kandil Professor of Cardiology Cairo University ST Elevation Acute Myocardial Infarction Aims Of Management Emergency care (Pre-hospital) Early care
Management of Acute Myocardial Infarction Prof. Hossam Kandil Professor of Cardiology Cairo University ST Elevation Acute Myocardial Infarction Aims Of Management Emergency care (Pre-hospital) Early care
Cangrelor: Is it the new CHAMPION for PCI? Robert Barcelona, PharmD, BCPS Clinical Pharmacy Specialist, Cardiac Intensive Care Unit November 13, 2015
 Cangrelor: Is it the new CHAMPION for PCI? Robert Barcelona, PharmD, BCPS Clinical Pharmacy Specialist, Cardiac Intensive Care Unit November 13, 2015 Objectives Review the pharmacology and pharmacokinetic
Cangrelor: Is it the new CHAMPION for PCI? Robert Barcelona, PharmD, BCPS Clinical Pharmacy Specialist, Cardiac Intensive Care Unit November 13, 2015 Objectives Review the pharmacology and pharmacokinetic
CHAPTER 17 Antithrombotic Agents Heparins
 CHAPTER 17 Antithrombotic Agents Heparins Structure Mechanism of Action Pharmacokinetics Limitations of Unfractionated Heparin Heparin Induced Thrombocytopenia Heparin Rebound Low Molecular Weight Heparins
CHAPTER 17 Antithrombotic Agents Heparins Structure Mechanism of Action Pharmacokinetics Limitations of Unfractionated Heparin Heparin Induced Thrombocytopenia Heparin Rebound Low Molecular Weight Heparins
Timing of Surgery After Percutaneous Coronary Intervention
 Timing of Surgery After Percutaneous Coronary Intervention Deepak Talreja, MD, FACC Bayview/EVMS/Sentara Outline/Highlights Timing of elective surgery What to do with medications Stopping anti-platelet
Timing of Surgery After Percutaneous Coronary Intervention Deepak Talreja, MD, FACC Bayview/EVMS/Sentara Outline/Highlights Timing of elective surgery What to do with medications Stopping anti-platelet
Platelet-fibrin clot. 50Kd STEMI. Abciximab. Video of a IIb/IIIa inhibitor in action. Unstable Angina and non-stsegment
 Objectives IIb/IIIa, Vitamin K, and Direct Thrombin Inhibition in Cardiology Michael Gulseth, Pharm. D., BCPS Assistant Professor, Duluth Pharmacy 6122 February 14, 2005 Describe the pharmacology, kinetics,
Objectives IIb/IIIa, Vitamin K, and Direct Thrombin Inhibition in Cardiology Michael Gulseth, Pharm. D., BCPS Assistant Professor, Duluth Pharmacy 6122 February 14, 2005 Describe the pharmacology, kinetics,
Prevention of Coronary Stent Thrombosis and Restenosis
 Prevention of Coronary Stent Thrombosis and Restenosis Seong-Wook Park, MD, PhD, FACC Division of Cardiology, Asan Medical Center University of Ulsan College of Medicine, Seoul, Korea 9/12/03 Coronary
Prevention of Coronary Stent Thrombosis and Restenosis Seong-Wook Park, MD, PhD, FACC Division of Cardiology, Asan Medical Center University of Ulsan College of Medicine, Seoul, Korea 9/12/03 Coronary
EPTIFIBATIDE INDUCED SEVERE THROMBOCYTOPENIA IN AN ASYMPTOMATIC PATIENT
 EPTIFIBATIDE INDUCED SEVERE THROMBOCYTOPENIA IN AN ASYMPTOMATIC PATIENT 1 Dr. M. Adnan Raufi MD FACC FSCAI, 2 Ayesha S. BaigMDRama Siddiqui, 3 Nouman Akbar 4 Shakaib Jaffery International Journal of Drug
EPTIFIBATIDE INDUCED SEVERE THROMBOCYTOPENIA IN AN ASYMPTOMATIC PATIENT 1 Dr. M. Adnan Raufi MD FACC FSCAI, 2 Ayesha S. BaigMDRama Siddiqui, 3 Nouman Akbar 4 Shakaib Jaffery International Journal of Drug
Antiplatelet therapy and Coronary Interventions. Georgios I. Papaioannou, MD Hartford Hospital Grand Rounds 4/22/2003
 Antiplatelet therapy and Coronary Interventions Georgios I. Papaioannou, MD Hartford Hospital Grand Rounds 4/22/2003 Objectives Pharmacology of GP IIb/IIIa inhibitors and Monitoring of Platelet Inhibition
Antiplatelet therapy and Coronary Interventions Georgios I. Papaioannou, MD Hartford Hospital Grand Rounds 4/22/2003 Objectives Pharmacology of GP IIb/IIIa inhibitors and Monitoring of Platelet Inhibition
Adults With Diagnosed Diabetes
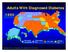 Adults With Diagnosed Diabetes 1990 No data available Less than 4% 4%-6% Above 6% Mokdad AH, et al. Diabetes Care. 2000;23(9):1278-1283. Adults With Diagnosed Diabetes 2000 4%-6% Above 6% Mokdad AH, et
Adults With Diagnosed Diabetes 1990 No data available Less than 4% 4%-6% Above 6% Mokdad AH, et al. Diabetes Care. 2000;23(9):1278-1283. Adults With Diagnosed Diabetes 2000 4%-6% Above 6% Mokdad AH, et
Role of Clopidogrel in Acute Coronary Syndromes. Hossam Kandil,, MD. Professor of Cardiology Cairo University
 Role of Clopidogrel in Acute Coronary Syndromes Hossam Kandil,, MD Professor of Cardiology Cairo University ACS Treatment Strategies Reperfusion/Revascularization Therapy Thrombolysis PCI (with/ without
Role of Clopidogrel in Acute Coronary Syndromes Hossam Kandil,, MD Professor of Cardiology Cairo University ACS Treatment Strategies Reperfusion/Revascularization Therapy Thrombolysis PCI (with/ without
Columbia University Medical Center Cardiovascular Research Foundation
 STEMI and NSTEMI Pharmacology Confusion: How to Choose and Use Antithrombins (Unfractionated and Low Molecular Heparins, Bivalirudin, Fondaparinux) and Antiplatelet Agents (Aspirin, Clopidogrel and Prasugrel)
STEMI and NSTEMI Pharmacology Confusion: How to Choose and Use Antithrombins (Unfractionated and Low Molecular Heparins, Bivalirudin, Fondaparinux) and Antiplatelet Agents (Aspirin, Clopidogrel and Prasugrel)
Clopidogrel and ASA after CABG for NSTEMI
 Clopidogrel and ASA after CABG for NSTEMI May 17, 2007 Justin Lee Pharmacy Resident University Health Network Objectives At the end of this session, you should be able to: Explain the rationale for antiplatelet
Clopidogrel and ASA after CABG for NSTEMI May 17, 2007 Justin Lee Pharmacy Resident University Health Network Objectives At the end of this session, you should be able to: Explain the rationale for antiplatelet
Acute Coronary Syndromes
 Overview Acute Coronary Syndromes Rabeea Aboufakher, MD, FACC, FSCAI Section Chief of Cardiology Altru Health System Grand Forks, ND Epidemiology Pathophysiology Clinical features and diagnosis STEMI management
Overview Acute Coronary Syndromes Rabeea Aboufakher, MD, FACC, FSCAI Section Chief of Cardiology Altru Health System Grand Forks, ND Epidemiology Pathophysiology Clinical features and diagnosis STEMI management
Platelet glycoprotein IIb/IIIa inhibition in acute coronary syndromes
 European Heart Journal (00) 3, 1441 1448 doi:10.1053/euhj.00.3160, available online at http://www.idealibrary.com on Platelet glycoprotein IIb/IIIa inhibition in acute coronary syndromes Gradient of benefit
European Heart Journal (00) 3, 1441 1448 doi:10.1053/euhj.00.3160, available online at http://www.idealibrary.com on Platelet glycoprotein IIb/IIIa inhibition in acute coronary syndromes Gradient of benefit
M/39 CC D. => peak CKMB (12 hr later) ng/ml T.chol/TG/HDL/LDL 180/150/48/102 mg/dl #
 Acute Coronary Syndrome - Case Review - Young-Guk Ko, MD Yonsei Cardiovascular Center Yonsei University College of Medicine Case 1 M/39 #4306212 CC D : Severe squeezing chest pain : 4 hours, aggravated
Acute Coronary Syndrome - Case Review - Young-Guk Ko, MD Yonsei Cardiovascular Center Yonsei University College of Medicine Case 1 M/39 #4306212 CC D : Severe squeezing chest pain : 4 hours, aggravated
THE ECONOMICS OF ADJUNCTIVE THERAPIES IN CORONARY ANGIOPLASTY: DRUGS, DEVICES, OR BOTH?
 THE ECONOMICS OF ADJUNCTIVE THERAPIES IN CORONARY ANGIOPLASTY: DRUGS, DEVICES, OR BOTH? Paul I. Oh, 1 Eric A. Cohen, 2 Nicole Mittmann, 3, 4 Soo Jin Seung 4 1 Division of Clinical Pharmacology, Sunnybrook
THE ECONOMICS OF ADJUNCTIVE THERAPIES IN CORONARY ANGIOPLASTY: DRUGS, DEVICES, OR BOTH? Paul I. Oh, 1 Eric A. Cohen, 2 Nicole Mittmann, 3, 4 Soo Jin Seung 4 1 Division of Clinical Pharmacology, Sunnybrook
ATHEROSCLEROTIC HEART DISease
 CLINICAL CARDIOLOGY Platelet Glycoprotein IIb/IIIa Receptor Antagonists in Cardiovascular Disease David A. Vorchheimer, MD Juan Jose Badimon, PhD Valentin Fuster, MD, PhD ATHEROSCLEROTIC HEART DISease
CLINICAL CARDIOLOGY Platelet Glycoprotein IIb/IIIa Receptor Antagonists in Cardiovascular Disease David A. Vorchheimer, MD Juan Jose Badimon, PhD Valentin Fuster, MD, PhD ATHEROSCLEROTIC HEART DISease
Adjunctive Antithrombotic for PCI. SCAI Fellows Course December 9, 2013
 Adjunctive Antithrombotic for PCI SCAI Fellows Course December 9, 2013 Theodore A Bass, MD FSCAI President SCAI Professor of Medicine, University of Florida Medical Director UF Shands CV Center,Jacksonville
Adjunctive Antithrombotic for PCI SCAI Fellows Course December 9, 2013 Theodore A Bass, MD FSCAI President SCAI Professor of Medicine, University of Florida Medical Director UF Shands CV Center,Jacksonville
QUT Digital Repository:
 QUT Digital Repository: http://eprints.qut.edu.au/ This is the author s version of this journal article. Published as: Doggrell, Sheila (2010) New drugs for the treatment of coronary artery syndromes.
QUT Digital Repository: http://eprints.qut.edu.au/ This is the author s version of this journal article. Published as: Doggrell, Sheila (2010) New drugs for the treatment of coronary artery syndromes.
Defining the Optimal Activated Clotting Time During Percutaneous Coronary Intervention
 Defining the Optimal Activated Clotting Time During Percutaneous Coronary Intervention Aggregate Results From 6 Randomized, Controlled Trials Derek P. Chew, MBBS; Deepak L. Bhatt, MD; A. Michael Lincoff,
Defining the Optimal Activated Clotting Time During Percutaneous Coronary Intervention Aggregate Results From 6 Randomized, Controlled Trials Derek P. Chew, MBBS; Deepak L. Bhatt, MD; A. Michael Lincoff,
A Multicenter Randomized Trial of Immediate Versus Delayed Invasive Strategy in Patients with Non-ST Elevation ACS
 Angioplasty to Blunt the rise Of troponin in Acute coronary syndromes Randomized for an immediate or Delayed intervention A Multicenter Randomized Trial of Immediate Versus Delayed Invasive Strategy in
Angioplasty to Blunt the rise Of troponin in Acute coronary syndromes Randomized for an immediate or Delayed intervention A Multicenter Randomized Trial of Immediate Versus Delayed Invasive Strategy in
COAGULATION, BLEEDING, AND TRANSFUSION IN URGENT AND EMERGENCY CORONARY SURGERY
 COAGULATION, BLEEDING, AND TRANSFUSION IN URGENT AND EMERGENCY CORONARY SURGERY VALTER CASATI, M.D. DIVISION OF CARDIOVASCULAR ANESTHESIA AND INTENSIVE CARE CLINICA S. GAUDENZIO NOVARA (ITALY) ANTIPLATELET
COAGULATION, BLEEDING, AND TRANSFUSION IN URGENT AND EMERGENCY CORONARY SURGERY VALTER CASATI, M.D. DIVISION OF CARDIOVASCULAR ANESTHESIA AND INTENSIVE CARE CLINICA S. GAUDENZIO NOVARA (ITALY) ANTIPLATELET
Asif Serajian DO FACC FSCAI
 Anticoagulation and Antiplatelet update: A case based approach Asif Serajian DO FACC FSCAI No disclosures relevant to this talk Objectives 1. Discuss the indication for antiplatelet therapy for cardiac
Anticoagulation and Antiplatelet update: A case based approach Asif Serajian DO FACC FSCAI No disclosures relevant to this talk Objectives 1. Discuss the indication for antiplatelet therapy for cardiac
Updated and Guideline Based Treatment of Patients with STEMI
 Updated and Guideline Based Treatment of Patients with STEMI Eli I. Lev, MD Director, Cardiac Catheterization Laboratory Hasharon Hospital, Rabin Medical Center Associate Professor of Cardiology Tel-Aviv
Updated and Guideline Based Treatment of Patients with STEMI Eli I. Lev, MD Director, Cardiac Catheterization Laboratory Hasharon Hospital, Rabin Medical Center Associate Professor of Cardiology Tel-Aviv
Treatment of Acute Coronary Syndromes
 Treatment of Acute Coronary Syndromes UC SF Jeffrey Tabas, M.D. sf g h Associate Professor UCSF School of Medicine Emergency Services, San Francisco General Hospital Objectives Review the updated AHA/ACC
Treatment of Acute Coronary Syndromes UC SF Jeffrey Tabas, M.D. sf g h Associate Professor UCSF School of Medicine Emergency Services, San Francisco General Hospital Objectives Review the updated AHA/ACC
Tailoring adjunctive antithrombotic therapy to reperfusion strategy in STEMI
 Tailoring adjunctive antithrombotic therapy to reperfusion strategy in STEMI Adel El-Etriby; MD Professor of Cardiology Ain Shams University President of the Egyptian Working Group of Interventional Cardiology
Tailoring adjunctive antithrombotic therapy to reperfusion strategy in STEMI Adel El-Etriby; MD Professor of Cardiology Ain Shams University President of the Egyptian Working Group of Interventional Cardiology
Adjunctive Antithrombotic for PCI. SCAI Fellows Course December 8, 2014
 Adjunctive Antithrombotic for PCI SCAI Fellows Course December 8, 2014 Theodore A Bass, MD FSCAI Immediate Past-President SCAI Professor of Medicine, University of Florida Medical Director UF Health CV
Adjunctive Antithrombotic for PCI SCAI Fellows Course December 8, 2014 Theodore A Bass, MD FSCAI Immediate Past-President SCAI Professor of Medicine, University of Florida Medical Director UF Health CV
Anti-platelet Therapies in Cardiovascular Disease: From Stable CAD to ACS and Afib!
 Anti-platelet Therapies in Cardiovascular Disease: From Stable CAD to ACS and Afib! Roxana Mehran, MD Columbia University Medical Center Cardiovascular Research Foundation Disclosures Research support
Anti-platelet Therapies in Cardiovascular Disease: From Stable CAD to ACS and Afib! Roxana Mehran, MD Columbia University Medical Center Cardiovascular Research Foundation Disclosures Research support
C.R.E.D.O. Multicenter Multinational (USA, Canada) Prospective Randomized Double Blind Placebo Controlled Trial
 Clopidogrel for the Reduction of Events During Observation Multicenter Multinational (USA, Canada) Prospective Randomized Double Blind Placebo Controlled Trial From Steinhubl et al, JAMA 2002;228:2411-20
Clopidogrel for the Reduction of Events During Observation Multicenter Multinational (USA, Canada) Prospective Randomized Double Blind Placebo Controlled Trial From Steinhubl et al, JAMA 2002;228:2411-20
Thrombolysis, adjunctive pharmacology and interventions
 ESC Guidelines for the management of acute myocardial infarction in patients presenting with ST-segment elevation ESC Annual Congress Munich, 2012 Thrombolysis, adjunctive pharmacology and interventions
ESC Guidelines for the management of acute myocardial infarction in patients presenting with ST-segment elevation ESC Annual Congress Munich, 2012 Thrombolysis, adjunctive pharmacology and interventions
Scottish Medicines Consortium
 Scottish Medicines Consortium bivalirudin, 250mg powder for concentrate for solution for injection or infusion (Angiox ) No. (516/08) The Medicines Company UK Ltd 07 November 2008 The Scottish Medicines
Scottish Medicines Consortium bivalirudin, 250mg powder for concentrate for solution for injection or infusion (Angiox ) No. (516/08) The Medicines Company UK Ltd 07 November 2008 The Scottish Medicines
Optimal antithrombotic therapy:
 Optimal antithrombotic therapy: upstream and during primary PCI. Steen D Kristensen, MD, DMSc, FESC Professor and Consultant Interventional Cardiologist Aarhus University, Denmark UNIVERSITY OF AARHUS
Optimal antithrombotic therapy: upstream and during primary PCI. Steen D Kristensen, MD, DMSc, FESC Professor and Consultant Interventional Cardiologist Aarhus University, Denmark UNIVERSITY OF AARHUS
STEMI update. Vijay Krishnamoorthy M.D. Interventional Cardiology
 STEMI update Vijay Krishnamoorthy M.D. Interventional Cardiology OVERVIEW Current Standard of Care in Management of STEMI Update in management of STEMI Pre-Cath Lab In the ED/Office/EMS. Cath Lab Post
STEMI update Vijay Krishnamoorthy M.D. Interventional Cardiology OVERVIEW Current Standard of Care in Management of STEMI Update in management of STEMI Pre-Cath Lab In the ED/Office/EMS. Cath Lab Post
Disclosures. Inpatient Management of Non-ST Elevation Acute Coronary Syndromes. Edward McNulty MD, FACC. None
 Inpatient Management of Non-ST Elevation Acute Coronary Syndromes Edward McNulty MD, FACC Assistant Clinical Professor UCSF Director, SF VAMC Cardiac Catheterization Laboratory Disclosures None New Guidelines
Inpatient Management of Non-ST Elevation Acute Coronary Syndromes Edward McNulty MD, FACC Assistant Clinical Professor UCSF Director, SF VAMC Cardiac Catheterization Laboratory Disclosures None New Guidelines
Otamixaban for non-st-segment elevation acute coronary syndrome
 Otamixaban for non-st-segment elevation acute coronary syndrome September 2011 This technology summary is based on information available at the time of research and a limited literature search. It is not
Otamixaban for non-st-segment elevation acute coronary syndrome September 2011 This technology summary is based on information available at the time of research and a limited literature search. It is not
(ClinicalTrials.gov ID: NCT ) Title: The Italian Elderly ACS Study Author: Stefano Savonitto. Date: 29 August 2011 Meeting: ESC congress, Paris
 Early aggressive versus initially conservative strategy in elderly patients with non-st- elevation acute coronary syndrome: the Italian randomised trial (ClinicalTrials.gov ID: NCT00510185) Stefano Savonitto,
Early aggressive versus initially conservative strategy in elderly patients with non-st- elevation acute coronary syndrome: the Italian randomised trial (ClinicalTrials.gov ID: NCT00510185) Stefano Savonitto,
Journal of the American College of Cardiology Vol. 44, No. 9, by the American College of Cardiology Foundation ISSN /04/$30.
 Journal of the American College of Cardiology Vol. 44, No. 9, 2004 2004 by the American College of Cardiology Foundation ISSN 0735-1097/04/$30.00 Published by Elsevier Inc. doi:10.1016/j.jacc.2004.05.085
Journal of the American College of Cardiology Vol. 44, No. 9, 2004 2004 by the American College of Cardiology Foundation ISSN 0735-1097/04/$30.00 Published by Elsevier Inc. doi:10.1016/j.jacc.2004.05.085
Horizon Scanning Centre November 2012
 Horizon Scanning Centre November 2012 Cangrelor to reduce platelet aggregation and thrombosis in patients undergoing percutaneous coronary intervention99 SUMMARY NIHR HSC ID: 2424 This briefing is based
Horizon Scanning Centre November 2012 Cangrelor to reduce platelet aggregation and thrombosis in patients undergoing percutaneous coronary intervention99 SUMMARY NIHR HSC ID: 2424 This briefing is based
Bivalirudin Clinical Trials Update Evidence and Future Perspectives
 Bivalirudin Clinical Trials Update Evidence and Future Perspectives Andreas Baumbach Consultant Cardiologist/ hon. Reader in Cardiology Bristol Heart Institute University Hospitals Bristol MY CONFLICTS
Bivalirudin Clinical Trials Update Evidence and Future Perspectives Andreas Baumbach Consultant Cardiologist/ hon. Reader in Cardiology Bristol Heart Institute University Hospitals Bristol MY CONFLICTS
Medicine Dr. Omed Lecture 2 Stable and Unstable Angina
 Medicine Dr. Omed Lecture 2 Stable and Unstable Angina Risk stratification in stable angina. High Risk; *post infarct angina, *poor effort tolerance, *ischemia at low workload, *left main or three vessel
Medicine Dr. Omed Lecture 2 Stable and Unstable Angina Risk stratification in stable angina. High Risk; *post infarct angina, *poor effort tolerance, *ischemia at low workload, *left main or three vessel
Pathology of Cardiovascular Interventions. Body and Disease 2011
 Pathology of Cardiovascular Interventions Body and Disease 2011 Coronary Artery Atherosclerosis Intervention Goals: Acute Coronary Syndromes: Treat plaque rupture and thrombosis Significant Disease: Prevent
Pathology of Cardiovascular Interventions Body and Disease 2011 Coronary Artery Atherosclerosis Intervention Goals: Acute Coronary Syndromes: Treat plaque rupture and thrombosis Significant Disease: Prevent
Role of platelet glycoprotein IIb/IIIa inhibitors in rescue percutaneous coronary interventions
 Role of platelet glycoprotein IIb/IIIa inhibitors in rescue percutaneous coronary interventions Anna Sonia Petronio, Marco De Carlo, Roberta Rossini, Giovanni Amoroso, Ugo Limbruno, Nicola Ciabatti, Caterina
Role of platelet glycoprotein IIb/IIIa inhibitors in rescue percutaneous coronary interventions Anna Sonia Petronio, Marco De Carlo, Roberta Rossini, Giovanni Amoroso, Ugo Limbruno, Nicola Ciabatti, Caterina
Cardiovascular Disorders Lecture 3 Coronar Artery Diseases
 Cardiovascular Disorders Lecture 3 Coronar Artery Diseases By Prof. El Sayed Abdel Fattah Eid Lecturer of Internal Medicine Delta University Coronary Heart Diseases It is the leading cause of death in
Cardiovascular Disorders Lecture 3 Coronar Artery Diseases By Prof. El Sayed Abdel Fattah Eid Lecturer of Internal Medicine Delta University Coronary Heart Diseases It is the leading cause of death in
Diagnosis and Management of Acute Myocardial Infarction
 Diagnosis and Management of Acute Myocardial Infarction Acute Myocardial Infarction (AMI) occurs as a result of prolonged myocardial ischemia Atherosclerosis leads to endothelial rupture or erosion that
Diagnosis and Management of Acute Myocardial Infarction Acute Myocardial Infarction (AMI) occurs as a result of prolonged myocardial ischemia Atherosclerosis leads to endothelial rupture or erosion that
ISAR-LEFT MAIN: A Randomized Clinical Trial on Drug-Eluting Stents for Unprotected Left Main Lesions
 Julinda Mehilli, MD Deutsches Herzzentrum Technische Universität Munich Germany ISAR-LEFT MAIN: A Randomized Clinical Trial on Drug-Eluting Stents for Unprotected Left Main Lesions Background Left main
Julinda Mehilli, MD Deutsches Herzzentrum Technische Universität Munich Germany ISAR-LEFT MAIN: A Randomized Clinical Trial on Drug-Eluting Stents for Unprotected Left Main Lesions Background Left main
Anticoagulation therapy in acute coronary syndromes according to current guidelines
 Acute management of ACS Anticoagulation therapy in acute coronary syndromes according to current guidelines Marcin Grabowski, Marcin Leszczyk, Andrzej Cacko, Krzysztof J. Filipiak, Grzegorz Opolski 1 st
Acute management of ACS Anticoagulation therapy in acute coronary syndromes according to current guidelines Marcin Grabowski, Marcin Leszczyk, Andrzej Cacko, Krzysztof J. Filipiak, Grzegorz Opolski 1 st
Objectives. Treatment of ACS. Early Invasive Strategy. UA/NSTEMI General Concepts. UA/NSTEMI Initial Therapy/Antithrombotic
 Objectives Treatment of ACS Michael P. Gulseth, Pharm. D., BCPS Pharmacotherapy II Spring 2006 Define early invasive strategy and what patients typically receive this approach Compare/contrast the medications
Objectives Treatment of ACS Michael P. Gulseth, Pharm. D., BCPS Pharmacotherapy II Spring 2006 Define early invasive strategy and what patients typically receive this approach Compare/contrast the medications
NURSING DEPARTMENT CRITICAL CARE POLICY MANUAL CRITICAL CARE PROTOCOLS. ACTIVASE (t-pa) INFUSION PROTOCOL FOR ACUTE MYOCARDIAL INFARCTION
 NURSING DEPARTMENT CRITICAL CARE POLICY MANUAL CRITICAL CARE PROTOCOLS ACTIVASE (t-pa) FOR ACUTE MYOCARDIAL INFARCTION I. PURPOSE: A. To reduce the extent of myocardial infarction by lysing the clot in
NURSING DEPARTMENT CRITICAL CARE POLICY MANUAL CRITICAL CARE PROTOCOLS ACTIVASE (t-pa) FOR ACUTE MYOCARDIAL INFARCTION I. PURPOSE: A. To reduce the extent of myocardial infarction by lysing the clot in
Anti-thrombotic management in interventional cardiology
 Chapter 8 Anti-thrombotic management in interventional cardiology James Tcheng, Steve Kindsvater Introduction Since the dawn of the interventional age, the need for anti-thrombotic therapy during percutaneous
Chapter 8 Anti-thrombotic management in interventional cardiology James Tcheng, Steve Kindsvater Introduction Since the dawn of the interventional age, the need for anti-thrombotic therapy during percutaneous
Abstract Background: Methods: Results: Conclusions:
 Two-Year Clinical and Angiographic Outcomes of Overlapping Sirolimusversus Paclitaxel- Eluting Stents in the Treatment of Diffuse Long Coronary Lesions Kang-Yin Chen 1,2, Seung-Woon Rha 1, Yong-Jian Li
Two-Year Clinical and Angiographic Outcomes of Overlapping Sirolimusversus Paclitaxel- Eluting Stents in the Treatment of Diffuse Long Coronary Lesions Kang-Yin Chen 1,2, Seung-Woon Rha 1, Yong-Jian Li
Collectively, the efficacy of the intravenous glycoprotein
 Increased Mortality With Oral Platelet Glycoprotein IIb/IIIa Antagonists A Meta-Analysis of Phase III Multicenter Randomized Trials Derek P. Chew, MBBS; Deepak L. Bhatt, MD; Shelly Sapp, MS; Eric J. Topol,
Increased Mortality With Oral Platelet Glycoprotein IIb/IIIa Antagonists A Meta-Analysis of Phase III Multicenter Randomized Trials Derek P. Chew, MBBS; Deepak L. Bhatt, MD; Shelly Sapp, MS; Eric J. Topol,
bivalirudin 250mg powder for concentrate for solution for injection or infusion (Angiox) SMC No. (638/10) The Medicines Company
 bivalirudin 250mg powder for concentrate for solution for injection or infusion (Angiox) SMC No. (638/10) The Medicines Company 06 August 2010 The Scottish Medicines Consortium (SMC) has completed its
bivalirudin 250mg powder for concentrate for solution for injection or infusion (Angiox) SMC No. (638/10) The Medicines Company 06 August 2010 The Scottish Medicines Consortium (SMC) has completed its
Acute ischemic coronary syndromes (AICS)
 ...CONTINUING MEDICAL EDUCATION... CME ARTICLE Overview of Platelet Glycoprotein IIb/IIIa Receptor Antagonist Treatment in the Management of Coronary Interventions and Acute Ischemic Coronary Syndromes
...CONTINUING MEDICAL EDUCATION... CME ARTICLE Overview of Platelet Glycoprotein IIb/IIIa Receptor Antagonist Treatment in the Management of Coronary Interventions and Acute Ischemic Coronary Syndromes
Interventional Cardiology
 Clinical Investigations Interventional Cardiology Benefit of bolus-only platelet glycoprotein IIb/IIIa inhibition during percutaneous coronary intervention: Insights from the very early outcomes in the
Clinical Investigations Interventional Cardiology Benefit of bolus-only platelet glycoprotein IIb/IIIa inhibition during percutaneous coronary intervention: Insights from the very early outcomes in the
Direct Thrombin Inhibitors for PCI Pharmacology: Role of Bivalirudin in High-Risk PCI
 Direct Thrombin Inhibitors for PCI Pharmacology: Role of Bivalirudin in High-Risk PCI Charles A. Simonton MD, FACC, FSCAI Sanger Clinic Medical Director Clinical Innovation and Research Carolinas Heart
Direct Thrombin Inhibitors for PCI Pharmacology: Role of Bivalirudin in High-Risk PCI Charles A. Simonton MD, FACC, FSCAI Sanger Clinic Medical Director Clinical Innovation and Research Carolinas Heart
The restoration of coronary flow after an
 Pharmacological Reperfusion in Acute Myicardial Infarction after ASSENT 3 and GUSTO V [81] DANIEL FERREIRA, MD, FESC Serviço de Cardiologia, Hospital Fernando Fonseca, Amadora, Portugal Rev Port Cardiol
Pharmacological Reperfusion in Acute Myicardial Infarction after ASSENT 3 and GUSTO V [81] DANIEL FERREIRA, MD, FESC Serviço de Cardiologia, Hospital Fernando Fonseca, Amadora, Portugal Rev Port Cardiol
Clopidogrel Date: 15 July 2008
 These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert in the country of prescription Sponsor/company: sanofi-aventis ClinicalTrials.gov
These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert in the country of prescription Sponsor/company: sanofi-aventis ClinicalTrials.gov
Andreas Baumbach Bristol Heart Institute Bristol Royal Infirmary. London 27/1/2005
 Andreas Baumbach Bristol Heart Institute Bristol Royal Infirmary AB London 27/1/2005 Revascularisation in ACS ICTUS MERLIN REACT 4mins 4mins 4mins AB Tony s Comments 15mins ICTUS Comparison of an early
Andreas Baumbach Bristol Heart Institute Bristol Royal Infirmary AB London 27/1/2005 Revascularisation in ACS ICTUS MERLIN REACT 4mins 4mins 4mins AB Tony s Comments 15mins ICTUS Comparison of an early
Critical Review Form Therapy Objectives: Methods:
 Critical Review Form Therapy Clinical Trial Comparing Primary Coronary Angioplasty with Tissue-Plasminogen Activator for Acute Myocardial Infarction (GUSTO-IIb), NEJM 1997; 336: 1621-1628 Objectives: To
Critical Review Form Therapy Clinical Trial Comparing Primary Coronary Angioplasty with Tissue-Plasminogen Activator for Acute Myocardial Infarction (GUSTO-IIb), NEJM 1997; 336: 1621-1628 Objectives: To
Current Advances and Best Practices in Acute STEMI Management A pharmacoinvasive approach
 Current Advances and Best Practices in Acute STEMI Management A pharmacoinvasive approach Frans Van de Werf, MD, PhD University Hospitals, Leuven, Belgium Frans Van de Werf: Disclosures Research grants
Current Advances and Best Practices in Acute STEMI Management A pharmacoinvasive approach Frans Van de Werf, MD, PhD University Hospitals, Leuven, Belgium Frans Van de Werf: Disclosures Research grants
Geriatric Grand Rounds. Geriatric Grand Rounds. Heart Disease in the Elderly: Pitfalls and Practicalities. Objectives. Conflict of Interest Disclosure
 Geriatric Grand Rounds Tuesday, January 22, 2008 12:00 noon Dr. Bill Black Auditorium Glenrose Rehabilitation Hospital To receive the poster, and handouts via email, subscribe to our E-mailing list at
Geriatric Grand Rounds Tuesday, January 22, 2008 12:00 noon Dr. Bill Black Auditorium Glenrose Rehabilitation Hospital To receive the poster, and handouts via email, subscribe to our E-mailing list at
Journal of the American College of Cardiology Vol. 38, No. 3, by the American College of Cardiology ISSN /01/$20.
 Journal of the American College of Cardiology Vol. 38, No. 3, 2001 2001 by the American College of Cardiology ISSN 0735-1097/01/$20.00 Published by Elsevier Science Inc. PII S0735-1097(01)01423-1 Effect
Journal of the American College of Cardiology Vol. 38, No. 3, 2001 2001 by the American College of Cardiology ISSN 0735-1097/01/$20.00 Published by Elsevier Science Inc. PII S0735-1097(01)01423-1 Effect
DATA SHEET. REOPRO vial 5 ml contains 10 mg abciximab rmc. REOPRO is a clear, colourless, sterile solution for intravenous use.
 DATA SHEET REOPRO Abciximab rmc, solution for injection, vials 2 mg/ml. Presentation REOPRO vial 5 ml contains 10 mg abciximab rmc. REOPRO is a clear, colourless, sterile solution for intravenous use.
DATA SHEET REOPRO Abciximab rmc, solution for injection, vials 2 mg/ml. Presentation REOPRO vial 5 ml contains 10 mg abciximab rmc. REOPRO is a clear, colourless, sterile solution for intravenous use.
Newer Antiplatelets. Asha Moorthy, Jain T Kallarakkal, James Kumar P INTRODUCTION
 ewer Antiplatelets Asha Moorthy, Jain T Kallarakkal, James Kumar P ITRODUCTIO Many therapeutic drugs have been identified that possess clinically important antiplatelet activity. Platelet inhibition can
ewer Antiplatelets Asha Moorthy, Jain T Kallarakkal, James Kumar P ITRODUCTIO Many therapeutic drugs have been identified that possess clinically important antiplatelet activity. Platelet inhibition can
European Heart Journal (2000) doi: /euhj , available online at on
 European Heart Journal (2000) 21 2042 2055 doi:10.1053/euhj.2000.2309, available online at http://www.idealibrary.com on Safety and preliminary efficacy of one month glycoprotein IIb/IIIa inhibition with
European Heart Journal (2000) 21 2042 2055 doi:10.1053/euhj.2000.2309, available online at http://www.idealibrary.com on Safety and preliminary efficacy of one month glycoprotein IIb/IIIa inhibition with
Acute coronary syndromes
 Acute coronary syndromes 1 Acute coronary syndromes Acute coronary syndromes results primarily from diminished myocardial blood flow secondary to an occlusive or partially occlusive coronary artery thrombus.
Acute coronary syndromes 1 Acute coronary syndromes Acute coronary syndromes results primarily from diminished myocardial blood flow secondary to an occlusive or partially occlusive coronary artery thrombus.
Quinn Capers, IV, MD
 Heart Attacks Mended Hearts Presentation, January, 2017 Quinn Capers, IV, MD Associate Professor of Medicine (Cardiovascular Medicine) Director, Transradial Coronary Interventions Division of Cardiovascular
Heart Attacks Mended Hearts Presentation, January, 2017 Quinn Capers, IV, MD Associate Professor of Medicine (Cardiovascular Medicine) Director, Transradial Coronary Interventions Division of Cardiovascular
FastTest. You ve read the book now test yourself
 FastTest You ve read the book...... now test yourself To ensure you have learned the key points that will improve your patient care, read the authors questions below. The answers will refer you back to
FastTest You ve read the book...... now test yourself To ensure you have learned the key points that will improve your patient care, read the authors questions below. The answers will refer you back to
Which drug do you prefer for stable CAD? - P2Y12 inhibitor
 Which drug do you prefer for stable CAD? - P2Y12 inhibitor Jung Rae Cho, MD, PhD Cardiovascular Division, Department of Internal Medicine Kangnam Sacred Heart Hospital, Hallym University Medical Center,
Which drug do you prefer for stable CAD? - P2Y12 inhibitor Jung Rae Cho, MD, PhD Cardiovascular Division, Department of Internal Medicine Kangnam Sacred Heart Hospital, Hallym University Medical Center,
NOVEL ANTI-THROMBOTIC THERAPIES FOR ACUTE CORONARY SYNDROME: DIRECT THROMBIN INHIBITORS
 Judd E. Hollander, MD Professor, Clinical Research Director, Department of Emergency Medicine University of Pennsylvania School of Medicine, Philadelphia, Pennsylvania OBJECTIVES: 1. Discuss the concept
Judd E. Hollander, MD Professor, Clinical Research Director, Department of Emergency Medicine University of Pennsylvania School of Medicine, Philadelphia, Pennsylvania OBJECTIVES: 1. Discuss the concept
New Antithrombotic and Antiplatelet Drugs in CAD : (Factor Xa inhibitors, Direct Thrombin inhibitors and Prasugrel)
 New Antithrombotic and Antiplatelet Drugs in CAD : (Factor Xa inhibitors, Direct Thrombin inhibitors and Prasugrel) Limitations and Advantages of UFH and LMWH Biological limitations of UFH : 1. immune-mediated
New Antithrombotic and Antiplatelet Drugs in CAD : (Factor Xa inhibitors, Direct Thrombin inhibitors and Prasugrel) Limitations and Advantages of UFH and LMWH Biological limitations of UFH : 1. immune-mediated
T he leading cause of morbidity and mortality
 155 CARDIOLOGY UPDATE Antiplatelet therapy in cardiovascular disease M W H Behan, R F Storey... Platelet activation and aggregation are considered to be central to arterial thrombus formation. Antiplatelet
155 CARDIOLOGY UPDATE Antiplatelet therapy in cardiovascular disease M W H Behan, R F Storey... Platelet activation and aggregation are considered to be central to arterial thrombus formation. Antiplatelet
Appendix: ACC/AHA and ESC practice guidelines
 Appendix: ACC/AHA and ESC practice guidelines Definitions for guideline recommendations and level of evidence Recommendation Class I Class IIa Class IIb Class III Level of evidence Level A Level B Level
Appendix: ACC/AHA and ESC practice guidelines Definitions for guideline recommendations and level of evidence Recommendation Class I Class IIa Class IIb Class III Level of evidence Level A Level B Level
Novel Anticoagulation Therapy in Acute Coronary Syndrome
 Novel Anticoagulation Therapy in Acute Coronary Syndrome Soon Jun Hong Korea University Anam Hospital 1 Thrombus Formation Cascade Coagulation Cascade Platelet Cascade TXA2 Aspirin R Inhibitor Fondaparinux
Novel Anticoagulation Therapy in Acute Coronary Syndrome Soon Jun Hong Korea University Anam Hospital 1 Thrombus Formation Cascade Coagulation Cascade Platelet Cascade TXA2 Aspirin R Inhibitor Fondaparinux
Patient characteristics Intervention Comparison Length of followup
 ANTICOAGULANT CHAPTER Low Molecular Weight Heparins compared with Unfractionated Heparin Ref ID: 1903 Reference Antman EM, McCabe CH, Gurfinkel EP et al. Enoxaparin prevents death and cardiac ischemic
ANTICOAGULANT CHAPTER Low Molecular Weight Heparins compared with Unfractionated Heparin Ref ID: 1903 Reference Antman EM, McCabe CH, Gurfinkel EP et al. Enoxaparin prevents death and cardiac ischemic
AGGRASTAT (tirofiban hydrochloride) injection, for intravenous use Initial U.S. Approval: 1998
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use AGGRASTAT (tirofiban hydrochloride) safely and effectively. See full prescribing information for
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use AGGRASTAT (tirofiban hydrochloride) safely and effectively. See full prescribing information for
ACUTE LIMB ISCHEMIA Table of Contents
 ACUTE LIMB ISCHEMIA Table of Contents PERIPHERAL ARTERIAL DISEASE (PAD)... 1 INTERMITTENT CLAUDICATION (IC)... 1 CRITICAL LIMB ISCHEMIA (CLI)... 1 ACUTE LIMB ISCHEMIA (ALI)... 2 Risk Factors...2 Associated
ACUTE LIMB ISCHEMIA Table of Contents PERIPHERAL ARTERIAL DISEASE (PAD)... 1 INTERMITTENT CLAUDICATION (IC)... 1 CRITICAL LIMB ISCHEMIA (CLI)... 1 ACUTE LIMB ISCHEMIA (ALI)... 2 Risk Factors...2 Associated
DOSING AND ADMINISTRATION
 DOSING AND ADMINISTRATION USING 100 ML VIAL OR 250 ML BAG (50 MCG/ML UNITS) 1 2 BOLUS 25 mcg/kg within 5 minutes INFUSION CrCl >60 ml/min: 0.15 mcg/kg/min CrCl
DOSING AND ADMINISTRATION USING 100 ML VIAL OR 250 ML BAG (50 MCG/ML UNITS) 1 2 BOLUS 25 mcg/kg within 5 minutes INFUSION CrCl >60 ml/min: 0.15 mcg/kg/min CrCl
Pathophysiology of ACS
 Pathophysiology of ACS ~ 2.0 MM patients admitted to CCU or telemetry annually 0.6 MM ST-segment elevation MI 1.4 MM Non-ST-segment elevation ACS NSTEMI vs STEMI VANQWISH Boden et al N Engl J Med 1998;338:1785-1792
Pathophysiology of ACS ~ 2.0 MM patients admitted to CCU or telemetry annually 0.6 MM ST-segment elevation MI 1.4 MM Non-ST-segment elevation ACS NSTEMI vs STEMI VANQWISH Boden et al N Engl J Med 1998;338:1785-1792
Timing of Anti-Platelet Therapy for ACS (EARLY-ACS & ACUITY) Mitchell W. Krucoff, MD, FACC
 Timing of Anti-Platelet Therapy for ACS (EARLY-ACS & ACUITY) Mitchell W. Krucoff, MD, FACC Professor, Medicine/Cardiology Duke University Medical Center Director, Cardiovascular Devices Unit Duke Clinical
Timing of Anti-Platelet Therapy for ACS (EARLY-ACS & ACUITY) Mitchell W. Krucoff, MD, FACC Professor, Medicine/Cardiology Duke University Medical Center Director, Cardiovascular Devices Unit Duke Clinical
Clinical Case. Management of ACS Based on ACC/AHA & ESC Guidelines. Clinical Case 4/22/12. UA/NSTEMI: Definition
 Clinical Case Management of ACS Based on ACC/AHA & ESC Guidelines Dr Badri Paudel Mr M 75M Poorly controlled diabetic Smoker Presented on Sat 7pm Intense burning in the retrosternal area Clinical Case
Clinical Case Management of ACS Based on ACC/AHA & ESC Guidelines Dr Badri Paudel Mr M 75M Poorly controlled diabetic Smoker Presented on Sat 7pm Intense burning in the retrosternal area Clinical Case
