Prediction of THC Plasma and Brain Concentrations following. Marijuana Administration: Approach and Challenges
|
|
|
- Chester West
- 6 years ago
- Views:
Transcription
1 Prediction of THC Plasma and Brain Concentrations following Marijuana Administration: Approach and Challenges Contents: 1. Background and Significance 1.1. Introduction 1.2. THC - the primary chemical constituent of interest 1.3. Prediction of plasma and brain concentrations in human: Challenges and Approach 1.4. Modeling and Simulations: Importance in Clinical Pharmacology 1.5. Physiologically-based pharmacokinetic model (PBPK) to estimate the plasma and tissue concentration of THC 2. Objectives 2.1. Specific aim and hypothesis 3. Research Strategy and Preliminary Investigation 3.1. PBPK model development tool 3.2. Tissue compartments in PBPK model 4. Results and Discussion 4.1. PBPK model evaluation 4.2. Prediction of human plasma and brain concentrations of THC 5. Future Work 6. Appendix (Figures) 7. References 1
2 1. Background and Significance 1.1. Introduction The National Institute on Drug Abuse (NIDA) identifies marijuana as the most commonly used illicit drug in the United States; it is also one of the most commonly abused drugs throughout the world (1). Smoking (intrapulmonary delivery route) is the preferred mode of marijuana administration for both medicinal and recreational users. The increasing interest in the use of medical marijuana and the movement toward the legalization of recreational marijuana use create an increased need to better understand the pharmacokinetics of the active components of marijuana and their resulting pharmacodynamic/pharmacologic effects. Pharmacokinetics (PK) is the study of the time course of the absorption, distribution, metabolism, and excretion of a drug and is often evaluated using blood/plasma concentrations. Pharmacodynamics (PD) is the study of the relationship between the concentration of drug at the site of action (biophase compartment) and the exerted effect. The purpose of this research is to develop a physiologically-based pharmacokinetic model capable of predicting drug concentrations at sites not directly accessible for sampling followed by developing correlations of the simulated concentrations to clinically observed pharmacological effects THC - the primary chemical constituent of interest Cannabis is composed of a vast number of chemical constituents, with Δ 9 -tetrahydrocannabinol (THC) being the principal mediator of the observed psychoactive and cardiovascular effects (2). THC, once administered, rapidly distributes to highly vascularized tissues, including the brain (3). Because of their lipophilic natures, THC and its metabolites (primary metabolite, 11- hydroxy-thc (THC-OH); secondary metabolite, 11-nor-9-carboxy- Δ 9 -THC (THC-COOH)) 2
3 readily enter less vascularized tissues, where significant concentrations may remain for extended time periods (3,4) Prediction of plasma and brain concentrations in humans: Approach and Challenges THC, Δ 9 -tetrahydrocannabinol, the primary active chemical constituent in marijuana following marijuana smoking is known to produce psychoactive effects. THC is rapidly cleared from the bloodstream, and plasma concentrations cannot be used to directly predict the observed psychoactive effects. Johansson et al. hypothesized that residual levels of THC in the accumulating tissues (e.g. brain), long after the time of administration, might be responsible for the longer-term observed psychoactive effects (5). Hence, it is imperative to be able to predict the concentration of THC in the human brain in order to explain and predict the potential physical and behavioral effects. Since, in most cases, the brain is inaccessible for direct sampling, other methods which allow for the prediction of the brain concentration-time profile need to be developed. Further efforts utilizing modeling and simulation approaches, can be used to predict brain concentrations and correlate those to the observed physiologic effects to better quantify the risks associated with marijuana use. The use of modeling and simulation approaches may successfully predict drug concentrations in otherwise difficult-to-sample anatomical regions Modeling and Simulations: Importance in Clinical Pharmacology With the advancement in computing technologies and their application in the fields of clinical pharmacology and clinical pharmacokinetics, computer-based simulations have evolved as a tool for the prediction of the effects of drugs in physiological systems. In addition to the advantages 3
4 of being less expensive and less labor intensive compared to animal experiments or trials using human subjects, simulations can provide information that is experimentally difficult to obtain. Physiologically-Based Pharmacokinetic Modeling (PBPK) is a computational technique which uses known anatomic and physiologic characteristics, such as organ weight/volume, regional blood flow, and metabolism patterns, to quantitatively describe drug distribution patterns within the body. In these studies, PBPK techniques, along with known THC chemical and biochemical characteristics, are being used to predict brain THC concentrations. Model building was initiated using reported data from a controlled clinical trial which administered THC intravenously to a group of adult males (6) Physiologically-based pharmacokinetic models (PBPK) to estimate the plasma and tissue concentration of THC A PBPK model is a multi-compartment model with each compartment assigned to a specific organ or tissue. These compartments are connected in a flow model using the blood supply (circulatory system) as the system input and output source (7). PBPK modeling is an example of a bottom-up modeling approach wherein the compartmental structure derived using known system (anatomical) characteristics and parameterized using the experimentally determined physicochemical properties of the drug is utilized to predict the concentration-time profile in plasma and other tissues. Given a known input concentration of the drug (the dose), the resulting concentration-time profile of the drug in plasma and other tissues can then be simulated. The strength of the PBPK modeling approach lies in its application to the prediction of drug concentrations in inaccessible tissues; these methods have been widely used in the field of toxicology (8). PBPK models are finding increasing use in drug exposure risk assessment analyses due to the capability of such models to predict age- and gender-specific 4
5 pharmacokinetics for the populations that are difficult to study experimentally (e.g. pediatrics, pregnant women) (9). Frequently, PBPK models are derived and validated based on data obtained from animal species where both blood and tissue sampling may be possible. Interspecies scaling, an extrapolation between animal species and humans that accounts for differences in physiology, can then be applied to enable the prediction of a human profile from preclinical animal data without the necessity for human testing. 2. Objectives: The overall objective of this research is to use modeling-based approaches to better understand the time-dependent relationships between the dose of THC administered following intrapulmonary administration (smoking) and resulting physiologic effects Specific Aim: To develop a PBPK model to estimate the plasma and tissue concentrations of THC following marijuana administration The working hypothesis of this aim is that the concentration-time profile of THC in plasma and in tissue compartments can be predicted using a PBPK modeling approach. The following activities were conducted to initiate model development: a) Develop a human PBPK model using known physiological system parameters and the known physicochemical properties of THC using the PBPKPlus module in GastroPlus TM (Version 8.5, Simulations Plus, Inc., Lancaster, CA). GastroPlus TM is a modeling and simulation software package widely used by pharmaceutical scientists and the US Food and Drug Administration (FDA) for decision-making in drug discovery and development. Model diagnostics and optimization were performed by comparing the predicted (simulated) plasma profile obtained from the model to observed profiles obtained from the literature. 5
6 b) Prediction of THC profile in brain: The concentration-time profile for THC in the brain, the tissue of major interest regarding THC s psychoactive effects, is estimated using the PBPK model. 3. Research strategy and preliminary investigation: The PBPK model will allow the simulation of the effect of varying marijuana/thc dose and population physiology on the concentration-time profile of THC in plasma and other tissues. Development of a PBPK model to estimate the plasma and tissue concentrations of THC following THC administration 3.1. PBPK model development tool: The biopharmaceutical properties of THC were obtained from the literature, and, for those values that were not available, estimates were made using ADMET TM Predictor Version 6.5 (Simulations Plus, Inc., Lancaster, CA). The PBPKPlus TM module works in conjunction with an internal module named PEAR Physiology (Population Estimates for Age-Related Physiology). PEAR enables the construction of a physiological system with specific age, body weight, and race (limited to Western and Asian) features. The whole-body physiological model (Figure 2A) consists of selected tissue compartments (organs) connected via the circulatory system (blood) with the amount of drug in each compartment governed by a unique set of differential equations Tissue compartments in PBPK model: Each tissue compartment in the PBPKPlus TM module can be individually designated as either a Perfusion-limited or a Permeabilitylimited region. Selection of a perfusion-limited tissue compartment is appropriate when the permeability/ uptake of the drug into the tissue is rapid and the transfer of drug into the tissue is governed by the perfusion/ input rate (blood flow). Permeability-limited tissues, in contrast, are those where the transfer of drug to the tissue is low, and uptake of the drug into the tissue is 6
7 limited by the permeability (partitioning) properties of the drug and the total surface area of the tissue exposed to the drug input from the bloodstream. For THC, all tissue compartments in the PBPK model were assumed to be perfusion-limited (Figure 2B) due to its high log P (logarithm of partition-coefficient; high log P indicates high permeability in tissues) value. The mass balance differential equation for a non-eliminating, perfusion-limited tissue is described in the whole body PBPK model by (7,10): ( ) Eq. 1 where and refer to the volume of the tissue (organ), concentration of drug in the tissue, blood flow to the tissue, concentration of drug coming into the tissue (arterial), and the tissue-plasma partition coefficient for tissue i, respectively. Similarly, for eliminating/clearance tissues like the liver, the mass balance differential equation describing its drug tissue concentration is as follows: ( ( )) ( ) Eq. 2 where and are the intrinsic clearance and fraction of the unbound drug in plasma, respectively. The Rodgers and Rowland method (11) was selected for the calculation of the tissue-to-plasma partition coefficient (K p ) values in the PBPK model. This method encompasses a number of processes, including the drug dissolution rate in tissue water, partitioning of drug into neutral lipids and phospholipids, and the distribution process related to the ionization of the drug. 7
8 4. Results and Discussion 4.1. PBPK model evaluation A diagnostic evaluation of the model was performed by selecting a set of physiological conditions consistent with the population used for the investigation of intravenously administered THC pharmacokinetics described in Tseng et al. (6). The comparison was based on the average conditions (average dose = 4mg) in males (N=6, age = 23 ± 3 yr, weight = 72 ± 9 kg) receiving a THC infusion over an average interval of 20 minutes. The predicted and observed plasma concentration-time profiles (Figure 3) show good agreement, thereby supporting the predictive ability of the PBPK model. Additional populations and datasets will need to be evaluated to expand the potential utility of the model Prediction of human plasma and brain concentrations of THC Because of the critical importance of the psychoactive effects of THC, coupled with the inability to directly measure brain concentrations, the PBPK model was used to predict the concentration of THC in plasma and in the brain. Figure 4 shows a simulated concentration-time profile for THC in both plasma and brain using the initial PBPK model. The simulated peak THC concentration in brain was observed to be nearly four-fold higher that in the plasma, which supports the need to link the brain as the biophase compartment when studying the psychoactive effects elicited by THC. The brain concentration-time profile clearly shows higher terminal levels of THC (~50 ng/ml), compared to very low concentrations of THC in the plasma at the same time points. Since the concentration of drug in the brain likely determines the observed effects, the observed longer-term psychoactive effects of THC could be explained by the high THC concentrations predicted in the brain. 8
9 5. Future Work The focus of this research with regard to PBPK modeling has been to develop a basic model that predicts the plasma and the brain concentration profiles for THC. This model will continue to be optimized and will be extended to predict the effects of changes in physiology, e.g. gender, age, body weight, on plasma and tissue concentration-time profiles. In addition, the effect of the route of administration of THC, including the intrapulmonary route of administration (smoking) and the oral route (through the gastrointestinal tract), on the concentration-time profile in plasma and tissues will be examined. 9
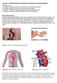
10 6. Appendix: Figures THC in human brain Psychoactive Effects Marijuana Figure 1. Marijuana smoking and psychoactive effects resulting from the concentration of THC in the brain (Images: Microsoft PowerPoint Clip Art). Q C bi Plasma (V p, C p, f up ) C bo Q Tissue (V t, C t, f ut, CL int ) Figure 2A Figure 2B Figure 2: Whole-body PBPK model with various organs connected by circulatory system (2A). Diagrammatic representation of a perfusion-limited tissue model (2B). Q is the blood flow in and out of the tissue. V, C, and f u are the volume, concentration, and fraction of unbound drug, respectively. C bi and C bo are the concentration of drug in the blood moving into the tissue (arterial) and concentration of drug in the blood moving out (venous) from the tissue compartment. 10
11 Figure 3A Figure 3B Figure 3: PBPK Model Diagnosis: Comparison of predicted plasma concentration-time profile with observed data (6) using PBPKPlus TM module. Red line, predicted (simulated) data; blue squares, observed data (average) reported by Tseng et al. (6). Fig. 3B: semi-log plot with concentration on log scale for better visual inspection of the terminal phase of the plot. THC Profile in Brain THC Profile in Plasma Figure 4: Simulation of brain THC profile: Simulated brain (purple line) and plasma (blue line) profile using PBPKPlus TM. 11
12 7. References 1. Substance Abuse and Mental Health Services Administration. Results from the 2010 National Survey on Drug Use and Health: Summary of National Findings, HHS Pub. No. (SMA) , Rockville, MD: SAMHSA, E.L. Karschner, E.W. Schwilke, R.H. Lowe, W.D. Darwin, R.I. Herning, J.L. Cadet, and M.A. Huestis. Implications of plasma Delta9-tetrahydrocannabinol, 11-hydroxy-THC, and 11-nor-9- carboxy-thc concentrations in chronic cannabis smokers. Journal of analytical toxicology. 33: (2009). 3. C.C. Hunault, J.C. van Eijkeren, T.T. Mensinga, I. de Vries, M.E. Leenders, and J. Meulenbelt. Disposition of smoked cannabis with high Delta(9)-tetrahydrocannabinol content: A kinetic model. Toxicology and applied pharmacology (2010). 4. M.A. Huestis, J.M. Mitchell, and E.J. Cone. Urinary excretion profiles of 11-nor-9-carboxy-delta 9-tetrahydrocannabinol in humans after single smoked doses of marijuana. Journal of analytical toxicology. 20: (1996). 5. E. Johansson, M.M. Halldin, S. Agurell, L.E. Hollister, and H.K. Gillespie. Terminal elimination plasma half-life of delta 1-tetrahydrocannabinol (delta 1-THC) in heavy users of marijuana. European journal of clinical pharmacology. 37: (1989). 6. A.H. Tseng, J.W. Harding, and R.M. Craft. Pharmacokinetic factors in sex differences in Delta 9- tetrahydrocannabinol-induced behavioral effects in rats. Behavioural brain research. 154:77-83 (2004). 7. H.M. Jones, I.B. Gardner, and K.J. Watson. Modelling and PBPK simulation in drug discovery. The AAPS journal. 11: (2009). 8. N. Tsamandouras, A. Rostami-Hodjegan, and L. Aarons. Combining the "bottom-up" and "topdown" approaches in pharmacokinetic modelling: Fitting PBPK models to observed clinical data. British journal of clinical pharmacology (2013). 9. H.J. Clewell, P.R. Gentry, T.R. Covington, R. Sarangapani, and J.G. Teeguarden. Evaluation of the potential impact of age- and gender-specific pharmacokinetic differences on tissue dosimetry. Toxicological sciences : an official journal of the Society of Toxicology. 79: (2004). 10. B. Xia, T. Heimbach, T.H. Lin, H. He, Y. Wang, and E. Tan. Novel physiologically based pharmacokinetic modeling of patupilone for human pharmacokinetic predictions. Cancer chemotherapy and pharmacology. 69: (2012). 11. T. Rodgersand M. Rowland. Physiologically based pharmacokinetic modelling 2: predicting the tissue distribution of acids, very weak bases, neutrals and zwitterions. Journal of pharmaceutical sciences. 95: (2006). 12
Pharmacokinetic Modeling & Simulation in Discovery and non-clinical Development
 Pharmacokinetic Modeling & Simulation in Discovery and non-clinical Development Where do we stand? Disclaimer I am not a bioinformatician, mathematician or biomedical engineer. I am a simple minded pharmacist,
Pharmacokinetic Modeling & Simulation in Discovery and non-clinical Development Where do we stand? Disclaimer I am not a bioinformatician, mathematician or biomedical engineer. I am a simple minded pharmacist,
Pharmacokinetic Evaluation of Published Studies on Controlled Smoking of Marijuana
 Pharmacokinetic Evaluation of Published Studies on Controlled Smoking of Marijuana G. Sticht and H. Käferstein Institute of Legal Medicine, University of Cologne, Melatengürtel 60-62, D - 50823 Köln, Germany
Pharmacokinetic Evaluation of Published Studies on Controlled Smoking of Marijuana G. Sticht and H. Käferstein Institute of Legal Medicine, University of Cologne, Melatengürtel 60-62, D - 50823 Köln, Germany
BASIC PHARMACOKINETICS
 BASIC PHARMACOKINETICS MOHSEN A. HEDAYA CRC Press Taylor & Francis Croup Boca Raton London New York CRC Press is an imprint of the Taylor & Francis Group, an informa business Table of Contents Chapter
BASIC PHARMACOKINETICS MOHSEN A. HEDAYA CRC Press Taylor & Francis Croup Boca Raton London New York CRC Press is an imprint of the Taylor & Francis Group, an informa business Table of Contents Chapter
Marijuana and DWI 1/31/2017. Smoking Vaporizing Ingestion of edibles. Hello Mary Jane. Summary. Marijuana and Public Policy
 Hello Mary Jane Marijuana and DWI Using Science to Cut Through the Smoke Marijuana and Public Policy Summary Between 25% and 60% of cartel profits come from Marijuana RAND Corporation study vs. White House
Hello Mary Jane Marijuana and DWI Using Science to Cut Through the Smoke Marijuana and Public Policy Summary Between 25% and 60% of cartel profits come from Marijuana RAND Corporation study vs. White House
Marijuana and DWI. Using Science to Cut Through the Smoke
 Marijuana and DWI Using Science to Cut Through the Smoke Hello Mary Jane Marijuana and Public Policy Between 25% and 60% of cartel profits come from Marijuana RAND Corporation study vs. White House Office
Marijuana and DWI Using Science to Cut Through the Smoke Hello Mary Jane Marijuana and Public Policy Between 25% and 60% of cartel profits come from Marijuana RAND Corporation study vs. White House Office
Use of PBPK in simulating drug concentrations in pediatric populations: Case studies of Midazolam and Gabapentin
 Use of PBPK in simulating drug concentrations in pediatric populations: Case studies of Midazolam and Gabapentin WORKSHOP ON MODELLING IN PAEDIATRIC MEDICINES April 2008 Viera Lukacova, Ph.D. Walter Woltosz,
Use of PBPK in simulating drug concentrations in pediatric populations: Case studies of Midazolam and Gabapentin WORKSHOP ON MODELLING IN PAEDIATRIC MEDICINES April 2008 Viera Lukacova, Ph.D. Walter Woltosz,
MARIHUANA AND DRIVING: WHAT IS THE SIGNIFICANCE OF CANNABINOID CONCENTRATIONS? * * A. McBay, M.D. ; and A. P. Mason SYNOPSIS
 MARIHUANA AND DRIVING: WHAT IS THE SIGNIFICANCE OF CANNABINOID CONCENTRATIONS? * * A. McBay, M.D. ; and A. P. Mason SYNOPSIS It has been inferred that significant driving impairment can result from marihuana
MARIHUANA AND DRIVING: WHAT IS THE SIGNIFICANCE OF CANNABINOID CONCENTRATIONS? * * A. McBay, M.D. ; and A. P. Mason SYNOPSIS It has been inferred that significant driving impairment can result from marihuana
One-Compartment Open Model: Intravenous Bolus Administration:
 One-Compartment Open Model: Intravenous Bolus Administration: Introduction The most common and most desirable route of drug administration is orally by mouth using tablets, capsules, or oral solutions.
One-Compartment Open Model: Intravenous Bolus Administration: Introduction The most common and most desirable route of drug administration is orally by mouth using tablets, capsules, or oral solutions.
Identifying New Cannabis Use with Urine Creatinine- Normalized THCCOOH Concentrations and Time Intervals Between Specimen Collections *
 Identifying New Cannabis Use with Urine Creatinine- Normalized THCCOOH Concentrations and Time Intervals Between Specimen Collections * Michael L. Smith 1, Allan J. Barnes 2, and Marilyn A. Huestis 2,
Identifying New Cannabis Use with Urine Creatinine- Normalized THCCOOH Concentrations and Time Intervals Between Specimen Collections * Michael L. Smith 1, Allan J. Barnes 2, and Marilyn A. Huestis 2,
WHY... 8/21/2013 LEARNING OUTCOMES PHARMACOKINETICS I. A Absorption. D Distribution DEFINITION ADME AND THERAPEUIC ACTION
 PHARMACOKINETICS I Absorption & Distribution LEARNING OUTCOMES By the end of the lecture students will be able to.. Dr Ruwan Parakramawansha MBBS, MD, MRCP(UK),MRCPE, DMT(UK) (2013/08/21) Define pharmacokinetics,
PHARMACOKINETICS I Absorption & Distribution LEARNING OUTCOMES By the end of the lecture students will be able to.. Dr Ruwan Parakramawansha MBBS, MD, MRCP(UK),MRCPE, DMT(UK) (2013/08/21) Define pharmacokinetics,
Pharmacokinetics One- compartment Open Model Lec:2
 22 Pharmacokinetics One- compartment Open Model Lec:2 Ali Y Ali BSc Pharmacy MSc Industrial Pharmaceutical Sciences Dept. of Pharmaceutics School of Pharmacy University of Sulaimani 1 Outline Introduction
22 Pharmacokinetics One- compartment Open Model Lec:2 Ali Y Ali BSc Pharmacy MSc Industrial Pharmaceutical Sciences Dept. of Pharmaceutics School of Pharmacy University of Sulaimani 1 Outline Introduction
Stimulate your kinetic understanding Permeability Binding Metabolism Transporters
 Stimulate your kinetic understanding Permeability Binding Metabolism Transporters www.simulations-plus.com +1-661-723-7723 What is MembranePlus? MembranePlus is an advanced, yet easy-to-use, modeling and
Stimulate your kinetic understanding Permeability Binding Metabolism Transporters www.simulations-plus.com +1-661-723-7723 What is MembranePlus? MembranePlus is an advanced, yet easy-to-use, modeling and
Development of Canagliflozin: Mechanistic Absorption Modeling During Late-Stage Formulation and Process Optimization
 Development of Canagliflozin: Mechanistic Absorption Modeling During Late-Stage Formulation and Process Optimization Nico Holmstock Scientist, Janssen R&D M CERSI 2017, BALTIMORE (USA) Canagliflozin An
Development of Canagliflozin: Mechanistic Absorption Modeling During Late-Stage Formulation and Process Optimization Nico Holmstock Scientist, Janssen R&D M CERSI 2017, BALTIMORE (USA) Canagliflozin An
Basic Pharmacokinetics and Pharmacodynamics: An Integrated Textbook with Computer Simulations
 Basic Pharmacokinetics and Pharmacodynamics: An Integrated Textbook with Computer Simulations Rosenbaum, Sara E. ISBN-13: 9780470569061 Table of Contents 1 Introduction to Pharmacokinetics and Pharmacodynamics.
Basic Pharmacokinetics and Pharmacodynamics: An Integrated Textbook with Computer Simulations Rosenbaum, Sara E. ISBN-13: 9780470569061 Table of Contents 1 Introduction to Pharmacokinetics and Pharmacodynamics.
USING PBPK MODELING TO SIMULATE THE DISPOSITION OF CANAGLIFLOZIN
 USING PBPK MODELING TO SIMULATE THE DISPOSITION OF CANAGLIFLOZIN Christophe Tistaert PDMS Pharmaceutical Sciences Preformulation & Biopharmaceutics AAPS 2015, FLORIDA (USA) Canagliflozin An orally active
USING PBPK MODELING TO SIMULATE THE DISPOSITION OF CANAGLIFLOZIN Christophe Tistaert PDMS Pharmaceutical Sciences Preformulation & Biopharmaceutics AAPS 2015, FLORIDA (USA) Canagliflozin An orally active
BIOPHARMACEUTICS and CLINICAL PHARMACY
 11 years papers covered BIOPHARMACEUTICS and CLINICAL PHARMACY IV B.Pharm II Semester, Andhra University Topics: Absorption Distribution Protein binding Metabolism Excretion Bioavailability Drug Interactions
11 years papers covered BIOPHARMACEUTICS and CLINICAL PHARMACY IV B.Pharm II Semester, Andhra University Topics: Absorption Distribution Protein binding Metabolism Excretion Bioavailability Drug Interactions
Basic Concepts of TDM
 TDM Lecture 1 5 th stage What is TDM? Basic Concepts of TDM Therapeutic drug monitoring (TDM) is a branch of clinical pharmacology that specializes in the measurement of medication concentrations in blood.
TDM Lecture 1 5 th stage What is TDM? Basic Concepts of TDM Therapeutic drug monitoring (TDM) is a branch of clinical pharmacology that specializes in the measurement of medication concentrations in blood.
Viera Lukacova Director, Simulation Sciences
 Use of In Silico Mechanistic Models to Support Interspecies Extrapolation of Oral Bioavailability and Formulation Optimization: Model Example Using GastroPlus Viera Lukacova Director, Simulation Sciences
Use of In Silico Mechanistic Models to Support Interspecies Extrapolation of Oral Bioavailability and Formulation Optimization: Model Example Using GastroPlus Viera Lukacova Director, Simulation Sciences
Simulation of the Nonlinear PK of Gabapentin and Midazolam in. Adult and Pediatric Populations. Population Approach Group in Europe (PAGE)
 Simulation of the Nonlinear PK of Gabapentin and Midazolam in Adult and Pediatric Populations Population Approach Group in Europe (PAGE) Meeting June 2006 Brugge, Belgium Walter Woltosz, M.S., M.A.S. Viera
Simulation of the Nonlinear PK of Gabapentin and Midazolam in Adult and Pediatric Populations Population Approach Group in Europe (PAGE) Meeting June 2006 Brugge, Belgium Walter Woltosz, M.S., M.A.S. Viera
Define the terms biopharmaceutics and bioavailability.
 Pharmaceutics Reading Notes Define the terms biopharmaceutics and bioavailability. Biopharmaceutics: the area of study concerning the relationship between the physical, chemical, and biological sciences
Pharmaceutics Reading Notes Define the terms biopharmaceutics and bioavailability. Biopharmaceutics: the area of study concerning the relationship between the physical, chemical, and biological sciences
Dr. Nele Plock. Dr. N. Plock, March 11, 2008, American Conference on Pharmacometrics
 Parallel PKPD modeling of an endogenous agonist (A) and exogenous antagonist (B) based on research PKPD data to predict an effective first-in-man dose of B A combination of physiologically based PK and
Parallel PKPD modeling of an endogenous agonist (A) and exogenous antagonist (B) based on research PKPD data to predict an effective first-in-man dose of B A combination of physiologically based PK and
A Regional Model of Lung Metabolism for Improving Species Dependent Descriptions of 1,3-Butadiene and its Metabolites
 A Regional Model of Lung Metabolism for Improving Species Dependent Descriptions of 1,3-Butadiene and its Metabolites Harvey Clewell, Jerry Campbell, and Cynthia VanLandingham ENVIRON and The Hamner Institutes
A Regional Model of Lung Metabolism for Improving Species Dependent Descriptions of 1,3-Butadiene and its Metabolites Harvey Clewell, Jerry Campbell, and Cynthia VanLandingham ENVIRON and The Hamner Institutes
DMPK. APRIL 27 TH 2017 Jan Neelissen Scientific Adviser Science & Technology
 DMPK APRIL 27 TH 2017 Jan Neelissen Scientific Adviser Science & Technology What I learned is a good DMPK profile have acceptable water solubility for development be completely absorbed, preferably via
DMPK APRIL 27 TH 2017 Jan Neelissen Scientific Adviser Science & Technology What I learned is a good DMPK profile have acceptable water solubility for development be completely absorbed, preferably via
Determination of bioavailability
 Pharmaceutics 2 Bioavailability Bioavailability is the rate and extent to which an administered drug reaches the systemic circulation. For example, if 100 mg of a drug is administered orally and 70 mg
Pharmaceutics 2 Bioavailability Bioavailability is the rate and extent to which an administered drug reaches the systemic circulation. For example, if 100 mg of a drug is administered orally and 70 mg
Lecture 1: Physicochemical Properties of Drugs and Drug Disposition
 Lecture 1: Physicochemical Properties of Drugs and Drug Disposition Key objectives: 1. Be able to explain the benefits of oral versus IV drug administration 2. Be able to explain the factors involved in
Lecture 1: Physicochemical Properties of Drugs and Drug Disposition Key objectives: 1. Be able to explain the benefits of oral versus IV drug administration 2. Be able to explain the factors involved in
EVE 491/591 Toxicology. Toxicant Entry into the Body 2/19/2018. Absorption and Fate of a Toxicant
 EVE 491/591 Toxicology Lecture #7 1. Absorption of Toxicants 2. Case study Part VI Toxicant Entry into the Body Toxicants must defeat barriers to absorption The respiratory system The digestive system
EVE 491/591 Toxicology Lecture #7 1. Absorption of Toxicants 2. Case study Part VI Toxicant Entry into the Body Toxicants must defeat barriers to absorption The respiratory system The digestive system
Pharmacokinetics of drug infusions
 SA Hill MA PhD FRCA Key points The i.v. route provides the most predictable plasma concentrations. Pharmacodynamic effects of a drug are related to plasma concentration. Both plasma and effect compartments
SA Hill MA PhD FRCA Key points The i.v. route provides the most predictable plasma concentrations. Pharmacodynamic effects of a drug are related to plasma concentration. Both plasma and effect compartments
Muhammad Fawad Rasool Feras Khalil Stephanie Läer
 Clin Pharmacokinet (2015) 54:943 962 DOI 10.1007/s40262-015-0253-7 ORIGINAL RESEARCH ARTICLE A Physiologically Based Pharmacokinetic Drug Disease Model to Predict Carvedilol Exposure in Adult and Paediatric
Clin Pharmacokinet (2015) 54:943 962 DOI 10.1007/s40262-015-0253-7 ORIGINAL RESEARCH ARTICLE A Physiologically Based Pharmacokinetic Drug Disease Model to Predict Carvedilol Exposure in Adult and Paediatric
NOTE FOR GUIDANCE ON TOXICOKINETICS: THE ASSESSMENT OF SYSTEMIC EXPOSURE IN TOXICITY STUDIES S3A
 INTERNATIONAL CONFERENCE ON HARMONISATION OF TECHNICAL REQUIREMENTS FOR REGISTRATION OF PHARMACEUTICALS FOR HUMAN USE ICH HARMONISED TRIPARTITE GUIDELINE NOTE FOR GUIDANCE ON TOXICOKINETICS: THE ASSESSMENT
INTERNATIONAL CONFERENCE ON HARMONISATION OF TECHNICAL REQUIREMENTS FOR REGISTRATION OF PHARMACEUTICALS FOR HUMAN USE ICH HARMONISED TRIPARTITE GUIDELINE NOTE FOR GUIDANCE ON TOXICOKINETICS: THE ASSESSMENT
Cannabis in the Workplace
 Cannabis in the Workplace Ryan Vandrey, PhD Johns Hopkins University School of Medicine Disclosures Paid consultant to Zynerba Pharmaceuticals, Battelle Memorial Institute None of my consulting is directly
Cannabis in the Workplace Ryan Vandrey, PhD Johns Hopkins University School of Medicine Disclosures Paid consultant to Zynerba Pharmaceuticals, Battelle Memorial Institute None of my consulting is directly
1 Introduction: The Why and How of Drug Bioavailability Research
 j1 1 Introduction: The Why and How of Drug Bioavailability Research Han van de Waterbeemd and Bernard Testa Abbreviations ADME EMEA FDA NCE PD P-gp PK R&D Absorption, distribution, metabolism, and excretion
j1 1 Introduction: The Why and How of Drug Bioavailability Research Han van de Waterbeemd and Bernard Testa Abbreviations ADME EMEA FDA NCE PD P-gp PK R&D Absorption, distribution, metabolism, and excretion
Chapter 5. The Actions of Drugs. Origins of Drugs. Names of Drugs. Most drugs come from plants or are chemically derived from plants
 Chapter 5 The Actions of Drugs Origins of Drugs Most drugs come from plants or are chemically derived from plants Names of Drugs Chemical name: Complete chemical description of the molecule Example: N'-[2-[[5-(dimethylaminomethyl)-2-furyl]
Chapter 5 The Actions of Drugs Origins of Drugs Most drugs come from plants or are chemically derived from plants Names of Drugs Chemical name: Complete chemical description of the molecule Example: N'-[2-[[5-(dimethylaminomethyl)-2-furyl]
PFIZER INC. THERAPEUTIC AREA AND FDA APPROVED INDICATIONS: See USPI.
 PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
Chromatography on Immobilized Artificial Membrane
 Chromatography on Immobilized Artificial Membrane Principles of measuring compounds affinity to phospholipids using immobilized artificial membrane chromatography Chromatography on Immobilized Artificial
Chromatography on Immobilized Artificial Membrane Principles of measuring compounds affinity to phospholipids using immobilized artificial membrane chromatography Chromatography on Immobilized Artificial
PHARMACOKINETICS OF DRUG ABSORPTION
 Print Close Window Note: Large images and tables on this page may necessitate printing in landscape mode. Applied Biopharmaceutics & Pharmacokinetics > Chapter 7. Pharmacokinetics of Oral Absorption >
Print Close Window Note: Large images and tables on this page may necessitate printing in landscape mode. Applied Biopharmaceutics & Pharmacokinetics > Chapter 7. Pharmacokinetics of Oral Absorption >
Biopharmaceutics Lecture-11 & 12. Pharmacokinetics of oral absorption
 Biopharmaceutics Lecture-11 & 12 Pharmacokinetics of oral absorption The systemic drug absorption from the gastrointestinal (GI) tract or from any other extravascular site is dependent on 1. 2. 3. In the
Biopharmaceutics Lecture-11 & 12 Pharmacokinetics of oral absorption The systemic drug absorption from the gastrointestinal (GI) tract or from any other extravascular site is dependent on 1. 2. 3. In the
Comparison of Cannabinoid Pharmacokinetic Properties in Occasional and Heavy Users Smoking a Marijuana or Placebo Joint
 Comparison of Cannabinoid Pharmacokinetic Properties in Occasional and Heavy Users Smoking a Marijuana or Placebo Joint Stefan W. Toennes 1, *, Johannes G. Ramaekers 2, Eef L. Theunissen 2, Manfred R.
Comparison of Cannabinoid Pharmacokinetic Properties in Occasional and Heavy Users Smoking a Marijuana or Placebo Joint Stefan W. Toennes 1, *, Johannes G. Ramaekers 2, Eef L. Theunissen 2, Manfred R.
Practical Application of PBPK in Neonates and Infants, Including Case Studies
 Practical Application of PBPK in Neonates and Infants, Including Case Studies Presented at the conference : Innovative Approaches to Pediatric Drug Development and Pediatric Medical Countermeasures: A
Practical Application of PBPK in Neonates and Infants, Including Case Studies Presented at the conference : Innovative Approaches to Pediatric Drug Development and Pediatric Medical Countermeasures: A
PHA5128 Dose Optimization II Case Study I Spring 2013
 Silsamicin is an investigational compound being evaluated for its antimicrobial effect. The route of administration for this drug is via intravenous bolus. Approximately 99.9% of this drug is eliminated
Silsamicin is an investigational compound being evaluated for its antimicrobial effect. The route of administration for this drug is via intravenous bolus. Approximately 99.9% of this drug is eliminated
Introduction to Pharmacokinetics (PK) Anson K. Abraham, Ph.D. Associate Principal Scientist, PPDM- QP2 Merck & Co. Inc., West Point, PA 5- June- 2017
 Introduction to Pharmacokinetics (PK) Anson K. Abraham, Ph.D. Associate Principal Scientist, PPDM- QP2 Merck & Co. Inc., West Point, PA 5- June- 2017 1 Outline Definition & Relevance of Pharmacokinetics
Introduction to Pharmacokinetics (PK) Anson K. Abraham, Ph.D. Associate Principal Scientist, PPDM- QP2 Merck & Co. Inc., West Point, PA 5- June- 2017 1 Outline Definition & Relevance of Pharmacokinetics
Pharmacokinetics: The Basics
 Pharmacokinetics: The Basics 2017 Otsuka Pharmaceutical Development & Commercialization, Inc., Rockville, MD January 2017 MRC2.CORP.D.00200 1 advice or professional diagnosis. Users seeking medical advice
Pharmacokinetics: The Basics 2017 Otsuka Pharmaceutical Development & Commercialization, Inc., Rockville, MD January 2017 MRC2.CORP.D.00200 1 advice or professional diagnosis. Users seeking medical advice
Pharmacokinetics I. Dr. M.Mothilal Assistant professor
 Pharmacokinetics I Dr. M.Mothilal Assistant professor DRUG TRANSPORT For a drug to produce a therapeutic effect, it must reach to its target and it must accumulate at that site to reach to the minimum
Pharmacokinetics I Dr. M.Mothilal Assistant professor DRUG TRANSPORT For a drug to produce a therapeutic effect, it must reach to its target and it must accumulate at that site to reach to the minimum
PHA Final Exam Fall 2006
 PHA 5127 Final Exam Fall 2006 On my honor, I have neither given nor received unauthorized aid in doing this assignment. Name Please transfer the answers onto the bubble sheet. The question number refers
PHA 5127 Final Exam Fall 2006 On my honor, I have neither given nor received unauthorized aid in doing this assignment. Name Please transfer the answers onto the bubble sheet. The question number refers
Applied Biopharmaceutics & Pharmacokinetics Sixth Edition
 Applied Biopharmaceutics & Pharmacokinetics Sixth Edition Hill Leon Shargel, PHD, RPh Applied Biopharmaceutics, LLC Raleigh, North Carolina Affiliate Associate Professor, School of Pharmacy Virginia Commonwealth
Applied Biopharmaceutics & Pharmacokinetics Sixth Edition Hill Leon Shargel, PHD, RPh Applied Biopharmaceutics, LLC Raleigh, North Carolina Affiliate Associate Professor, School of Pharmacy Virginia Commonwealth
Sub Population: The elderly
 Sub Population: The elderly How can Modelling and Simulation inform understanding of safety and efficacy? Amy S. Y. Cheung, Senior Clinical Pharmacometrician Early Clinical Development, AstraZeneca On
Sub Population: The elderly How can Modelling and Simulation inform understanding of safety and efficacy? Amy S. Y. Cheung, Senior Clinical Pharmacometrician Early Clinical Development, AstraZeneca On
UNIVERSITY OF THE WEST INDIES, ST AUGUSTINE
 UNIVERSITY OF THE WEST INDIES, ST AUGUSTINE FACULTY OF MEDICAL SCIENCES SCHOOL OF PHARMACY BACHELOR OF SCIENCE IN PHARMACY DEGREE COURSE SYLLABUS COURSE TITLE: COURSE CODE: BIOPHARMACEUTICS, NEW DRUG DELIVERY
UNIVERSITY OF THE WEST INDIES, ST AUGUSTINE FACULTY OF MEDICAL SCIENCES SCHOOL OF PHARMACY BACHELOR OF SCIENCE IN PHARMACY DEGREE COURSE SYLLABUS COURSE TITLE: COURSE CODE: BIOPHARMACEUTICS, NEW DRUG DELIVERY
Saliva Versus Plasma Bioequivalence of Rusovastatin in Humans: Validation of Class III Drugs of the Salivary Excretion Classification System
 Drugs R D (2015) 15:79 83 DOI 10.1007/s40268-015-0080-1 ORIGINAL RESEARCH ARTICLE Saliva Versus Plasma Bioequivalence of Rusovastatin in Humans: Validation of Class III Drugs of the Salivary Excretion
Drugs R D (2015) 15:79 83 DOI 10.1007/s40268-015-0080-1 ORIGINAL RESEARCH ARTICLE Saliva Versus Plasma Bioequivalence of Rusovastatin in Humans: Validation of Class III Drugs of the Salivary Excretion
Basic Biopharmaceutics, Pharmacokinetics, and Pharmacodynamics
 Basic Biopharmaceutics, Pharmacokinetics, and Pharmacodynamics Learning Outcomes Define biopharmaceutics Describe 4 processes of pharmacokinetics Describe factors that affect medication absorption Describe
Basic Biopharmaceutics, Pharmacokinetics, and Pharmacodynamics Learning Outcomes Define biopharmaceutics Describe 4 processes of pharmacokinetics Describe factors that affect medication absorption Describe
Tissue Dose Based Risk Assessments
 Tissue Dose Based Risk Assessments Melvin Andersen The Hamner Institutes for Health Sciences Workshop on Health Risk Assessment of Essential Metals May 6-7, 2008 Ottawa, CA Outline PBPK modeling and the
Tissue Dose Based Risk Assessments Melvin Andersen The Hamner Institutes for Health Sciences Workshop on Health Risk Assessment of Essential Metals May 6-7, 2008 Ottawa, CA Outline PBPK modeling and the
Plasma Cannabinoid Pharmacokinetics After Controlled Smoking and Ad libitum Cannabis Smoking in Chronic Frequent Users
 Journal of Analytical Toxicology 2015;39:580 587 doi:10.1093/jat/bkv082 Special Issue Plasma Cannabinoid Pharmacokinetics After Controlled Smoking and Ad libitum Cannabis Smoking in Chronic Frequent Users
Journal of Analytical Toxicology 2015;39:580 587 doi:10.1093/jat/bkv082 Special Issue Plasma Cannabinoid Pharmacokinetics After Controlled Smoking and Ad libitum Cannabis Smoking in Chronic Frequent Users
Strategies for Developing and Validating PBPK Models for Extrapolation to Unstudied Population
 Strategies for Developing and Validating PBPK Models for Extrapolation to Unstudied Population Nina Isoherranen Department of Pharmaceutics University of Washington Processes driving drug disposition are
Strategies for Developing and Validating PBPK Models for Extrapolation to Unstudied Population Nina Isoherranen Department of Pharmaceutics University of Washington Processes driving drug disposition are
Altered GI absorption in special populations: An industry perspective
 Altered GI absorption in special populations: An industry perspective Cordula Stillhart and Neil J. Parrott F. Hoffmann La Roche Ltd, Basel (CH) UNGAP WG meeting, Leuven (B) 8 March 2018 Current challenges
Altered GI absorption in special populations: An industry perspective Cordula Stillhart and Neil J. Parrott F. Hoffmann La Roche Ltd, Basel (CH) UNGAP WG meeting, Leuven (B) 8 March 2018 Current challenges
General Principles of Pharmacology and Toxicology
 General Principles of Pharmacology and Toxicology Parisa Gazerani, Pharm D, PhD Assistant Professor Center for Sensory-Motor Interaction (SMI) Department of Health Science and Technology Aalborg University
General Principles of Pharmacology and Toxicology Parisa Gazerani, Pharm D, PhD Assistant Professor Center for Sensory-Motor Interaction (SMI) Department of Health Science and Technology Aalborg University
Pharmacokinetics and bioavailability derived from various body fluids. Saliva samples instead of plasma samples
 Pharmacokinetics and bioavailability derived from various body fluids Saliva samples instead of plasma samples Willi Cawello, Schwarz BioSciences, Monheim am Rhein 1 Overview Introduction Sampling tissues/fluids
Pharmacokinetics and bioavailability derived from various body fluids Saliva samples instead of plasma samples Willi Cawello, Schwarz BioSciences, Monheim am Rhein 1 Overview Introduction Sampling tissues/fluids
Absolute bioavailability and pharmacokinetic studies in early clinical development using microdose and microtracer approaches.
 Absolute bioavailability and pharmacokinetic studies in early clinical development using microdose and microtracer approaches. Dr Lloyd Stevens Senior Research Fellow Pharmaceutical Profiles Nottingham,
Absolute bioavailability and pharmacokinetic studies in early clinical development using microdose and microtracer approaches. Dr Lloyd Stevens Senior Research Fellow Pharmaceutical Profiles Nottingham,
Public Assessment Report. EU worksharing project paediatric data. Valcyte. Valganciclovir
 Public Assessment Report EU worksharing project paediatric data Valcyte Valganciclovir Currently approved indication(s): Pharmaceutical form(s) affected by this project: Strength(s) affected by this variation:
Public Assessment Report EU worksharing project paediatric data Valcyte Valganciclovir Currently approved indication(s): Pharmaceutical form(s) affected by this project: Strength(s) affected by this variation:
Drug and Alcohol Dependence
 Drug and Alcohol Dependence 105 (2009) 24 32 Contents lists available at ScienceDirect Drug and Alcohol Dependence journal homepage: www.elsevier.com/locate/drugalcdep Extended urinary 9-tetrahydrocannabinol
Drug and Alcohol Dependence 105 (2009) 24 32 Contents lists available at ScienceDirect Drug and Alcohol Dependence journal homepage: www.elsevier.com/locate/drugalcdep Extended urinary 9-tetrahydrocannabinol
Detection of Cannabinoids in Oral Fluid with the Agilent 7010 GC-MS/MS System
 Application Note Forensics, Workplace Drug Testing Detection of Cannabinoids in Oral Fluid with the Agilent 7010 GC-MS/MS System Authors Fred Feyerherm and Anthony Macherone Agilent Technologies, Inc.
Application Note Forensics, Workplace Drug Testing Detection of Cannabinoids in Oral Fluid with the Agilent 7010 GC-MS/MS System Authors Fred Feyerherm and Anthony Macherone Agilent Technologies, Inc.
Development of a Human PBBK Model for Mixtures: Trio Mixture of Mercury, Lead, and Selenium
 IOSR Journal of Environmental Science, Toxicology and Food Technology (IOSR-JESTFT) e-issn: 2319-2402,p- ISSN: 2319-2399.Volume 11, Issue 6 Ver. I (June. 2017), PP 06-13 www.iosrjournals.org Development
IOSR Journal of Environmental Science, Toxicology and Food Technology (IOSR-JESTFT) e-issn: 2319-2402,p- ISSN: 2319-2399.Volume 11, Issue 6 Ver. I (June. 2017), PP 06-13 www.iosrjournals.org Development
FDA Use of Big Data in Modeling and Simulations
 FDA Use of Big Data in Modeling and Simulations Jeffry Florian, Ph.D., Division of Pharmacometrics, CDER/OTS/OCP/DPM DISCLAIMER: The views expressed in this presentation are that of the author and do not
FDA Use of Big Data in Modeling and Simulations Jeffry Florian, Ph.D., Division of Pharmacometrics, CDER/OTS/OCP/DPM DISCLAIMER: The views expressed in this presentation are that of the author and do not
Understand the physiological determinants of extent and rate of absorption
 Absorption and Half-Life Nick Holford Dept Pharmacology & Clinical Pharmacology University of Auckland, New Zealand Objectives Understand the physiological determinants of extent and rate of absorption
Absorption and Half-Life Nick Holford Dept Pharmacology & Clinical Pharmacology University of Auckland, New Zealand Objectives Understand the physiological determinants of extent and rate of absorption
Pharmacokinetics and allometric scaling of levormeloxifene, a selective oestrogen receptor modulator
 BIOPHARMACEUTICS & DRUG DISPOSITION Biopharm. Drug Dispos. 24: 121 129 (2003) Published online in Wiley InterScience (www.interscience.wiley.com). DOI: 10.1002/bdd.344 Pharmacokinetics and allometric scaling
BIOPHARMACEUTICS & DRUG DISPOSITION Biopharm. Drug Dispos. 24: 121 129 (2003) Published online in Wiley InterScience (www.interscience.wiley.com). DOI: 10.1002/bdd.344 Pharmacokinetics and allometric scaling
Principles of Toxicokinetics/Toxicodynanics
 Biochemical and Molecular Toxicology ENVR 442; TOXC 442; BIOC 442 Principles of Toxicokinetics/Toxicodynanics Kim L.R. Brouwer, PharmD, PhD kbrouwer@unc.edu; 919-962-7030 Pharmacokinetics/ Toxicokinetics:
Biochemical and Molecular Toxicology ENVR 442; TOXC 442; BIOC 442 Principles of Toxicokinetics/Toxicodynanics Kim L.R. Brouwer, PharmD, PhD kbrouwer@unc.edu; 919-962-7030 Pharmacokinetics/ Toxicokinetics:
Nicotine pharmacokinetics of electronic cigarettes: experimental data and a review of the literature
 Nicotine pharmacokinetics of electronic cigarettes: experimental data and a review of the literature Ian M. Fearon, Alison Eldridge, Nathan Gale, Mike McEwan Paul R. Nelson, Mitch Stiles, Elaine Round
Nicotine pharmacokinetics of electronic cigarettes: experimental data and a review of the literature Ian M. Fearon, Alison Eldridge, Nathan Gale, Mike McEwan Paul R. Nelson, Mitch Stiles, Elaine Round
Pharmacokinetics of ibuprofen in man. I. Free and total
 Pharmacokinetics of ibuprofen in man. I. Free and total area/dose relationships Ibuprofen kinetics were studied in 15 subjects after four oral doses. Plasma levels of both total and free ibuprofen were
Pharmacokinetics of ibuprofen in man. I. Free and total area/dose relationships Ibuprofen kinetics were studied in 15 subjects after four oral doses. Plasma levels of both total and free ibuprofen were
Tutorial. & In case studies 1 and 2, we explore intravenous iv. & Then, we move on to extravascular dosing in case
 The AAPS Journal, Vol. 1, No. 1, January 2016 ( # 2015) DOI: 10.120/s1224-015-917-6 Tutorial Pattern Recognition in Pharmacokinetic Data Analysis Johan Gabrielsson, 1,4 Bernd Meibohm, 2 and Daniel Weiner
The AAPS Journal, Vol. 1, No. 1, January 2016 ( # 2015) DOI: 10.120/s1224-015-917-6 Tutorial Pattern Recognition in Pharmacokinetic Data Analysis Johan Gabrielsson, 1,4 Bernd Meibohm, 2 and Daniel Weiner
Outline. Model Development GLUCOSIM. Conventional Feedback and Model-Based Control of Blood Glucose Level in Type-I Diabetes Mellitus
 Conventional Feedback and Model-Based Control of Blood Glucose Level in Type-I Diabetes Mellitus Barış Ağar, Gülnur Birol*, Ali Çınar Department of Chemical and Environmental Engineering Illinois Institute
Conventional Feedback and Model-Based Control of Blood Glucose Level in Type-I Diabetes Mellitus Barış Ağar, Gülnur Birol*, Ali Çınar Department of Chemical and Environmental Engineering Illinois Institute
Volume 1(3) May-June 2013 Page 351
 ISSN: 2321-5674(Print) BIOAVAILABILITY: CRITERIA FOR APPROVING A DRUG PRODUCT FOR MARKETING Sandhya Singh 1, Faheem Ajmal Ansari 1, Shravan Paswan 2*, Rnjan Kumar Sharma 2, Alok Ranjan Gaur 3 1 Azad Institute
ISSN: 2321-5674(Print) BIOAVAILABILITY: CRITERIA FOR APPROVING A DRUG PRODUCT FOR MARKETING Sandhya Singh 1, Faheem Ajmal Ansari 1, Shravan Paswan 2*, Rnjan Kumar Sharma 2, Alok Ranjan Gaur 3 1 Azad Institute
Pharmacokinetic and absolute bioavailability studies in early clinical development using microdose and microtracer approaches.
 Pharmacokinetic and absolute bioavailability studies in early clinical development using microdose and microtracer approaches. Lloyd Stevens PhD Senior Research Fellow Pharmaceutical Profiles Nottingham,
Pharmacokinetic and absolute bioavailability studies in early clinical development using microdose and microtracer approaches. Lloyd Stevens PhD Senior Research Fellow Pharmaceutical Profiles Nottingham,
Public Assessment Report. Scientific discussion. Ropinirol Actavis. Ropinirole hydrochloride DK/H/1212/ /DC
 Public Assessment Report Scientific discussion Ropinirol Actavis Ropinirole hydrochloride DK/H/1212/001-007/DC This module reflects the scientific discussion for the approval of Ropinirole film-coated
Public Assessment Report Scientific discussion Ropinirol Actavis Ropinirole hydrochloride DK/H/1212/001-007/DC This module reflects the scientific discussion for the approval of Ropinirole film-coated
HTPK: Conducting PK modeling and
 HTPK: Conducting PK modeling and simulations at high speed November 5, 2018 Robert Fraczkiewicz, David Miller, Marvin Waldman, Robert D. Clark Slide 1 Session Description and Objectives HTPK lightens the
HTPK: Conducting PK modeling and simulations at high speed November 5, 2018 Robert Fraczkiewicz, David Miller, Marvin Waldman, Robert D. Clark Slide 1 Session Description and Objectives HTPK lightens the
Pharmaceutics I صيدالنيات 1. Unit 2 Route of Drug Administration
 Pharmaceutics I صيدالنيات 1 Unit 2 Route of Drug Administration 1 Routs of Drug administration The possible routes of drug entry into the body may be divided into two classes: Parenteral Rout Enteral Rout
Pharmaceutics I صيدالنيات 1 Unit 2 Route of Drug Administration 1 Routs of Drug administration The possible routes of drug entry into the body may be divided into two classes: Parenteral Rout Enteral Rout
DOSE SELECTION FOR CARCINOGENICITY STUDIES OF PHARMACEUTICALS *)
 DOSE SELECTION FOR CARCINOGENICITY STUDIES OF PHARMACEUTICALS *) Guideline Title Dose Selection for Carcinogenicity Studies of Pharmaceuticals *) Legislative basis Directive 75/318/EEC as amended Date
DOSE SELECTION FOR CARCINOGENICITY STUDIES OF PHARMACEUTICALS *) Guideline Title Dose Selection for Carcinogenicity Studies of Pharmaceuticals *) Legislative basis Directive 75/318/EEC as amended Date
Principal Investigator: Marion, Alan, S, M.D., MDS Pharma Services (US) Inc., 621 Rose Street, PO Box 80837, Lincoln, NE 68502, USA
 SYNOPSIS Issue Date: 06 October 2008 Document No.: EDMS-PSDB-8954363:2. Name of Sponsor/Company Johnson & Johnson Pharmaceutical Research & Development, L.L.C. Name of Finished Product Name of Active Ingredient(s)
SYNOPSIS Issue Date: 06 October 2008 Document No.: EDMS-PSDB-8954363:2. Name of Sponsor/Company Johnson & Johnson Pharmaceutical Research & Development, L.L.C. Name of Finished Product Name of Active Ingredient(s)
Saião A, Chan B, Isbister GK Department of Clinical Toxicology and Pharmacology, Calvary Mater Newcastle, NSW Emergency Department, Prince of Wales
 Saião A, Chan B, Isbister GK Department of Clinical Toxicology and Pharmacology, Calvary Mater Newcastle, NSW Emergency Department, Prince of Wales Hospital, Sydney, NSW, Australia Paracetamol Poisoning
Saião A, Chan B, Isbister GK Department of Clinical Toxicology and Pharmacology, Calvary Mater Newcastle, NSW Emergency Department, Prince of Wales Hospital, Sydney, NSW, Australia Paracetamol Poisoning
The clinical trial information provided in this public disclosure synopsis is supplied for informational purposes only.
 The clinical trial information provided in this public disclosure synopsis is supplied for informational purposes only. Please note that the results reported in any single trial may not reflect the overall
The clinical trial information provided in this public disclosure synopsis is supplied for informational purposes only. Please note that the results reported in any single trial may not reflect the overall
Impaired Driving in Colorado: First Results from a Data Integration Initiative
 Impaired Driving in Colorado: First Results from a Data Integration Initiative BECKY BUI OFFICE OF RESEARCH & STATISTICS DIVISION OF CRIMINAL JUSTICE COLORADO DEPARTMENT OF PUBLIC SAFETY Presentation to
Impaired Driving in Colorado: First Results from a Data Integration Initiative BECKY BUI OFFICE OF RESEARCH & STATISTICS DIVISION OF CRIMINAL JUSTICE COLORADO DEPARTMENT OF PUBLIC SAFETY Presentation to
A primer on pharmacology
 A primer on pharmacology Universidade do Algarve Faro 2017 by Ferdi Engels, Ph.D. 1 2 1 3 Utrecht university campus de Uithof Dept. of Pharmaceutical Sciences Division of Pharmacology 4 2 Bachelor and
A primer on pharmacology Universidade do Algarve Faro 2017 by Ferdi Engels, Ph.D. 1 2 1 3 Utrecht university campus de Uithof Dept. of Pharmaceutical Sciences Division of Pharmacology 4 2 Bachelor and
Metformin: Mechanistic Absorption Modeling and IVIVC Development
 Metformin: Mechanistic Absorption Modeling and IVIVC Development Maziar Kakhi *, Ph.D. FDA Silver Spring, MD 20993 Maziar.kakhi@fda.hhs.gov Viera Lukacova, Ph.D. Simulations Plus Lancaster, CA 93534 viera@simulations-plus.com
Metformin: Mechanistic Absorption Modeling and IVIVC Development Maziar Kakhi *, Ph.D. FDA Silver Spring, MD 20993 Maziar.kakhi@fda.hhs.gov Viera Lukacova, Ph.D. Simulations Plus Lancaster, CA 93534 viera@simulations-plus.com
PHA Final Exam. Fall On my honor, I have neither given nor received unauthorized aid in doing this assignment.
 PHA 5127 Final Exam Fall 2012 On my honor, I have neither given nor received unauthorized aid in doing this assignment. Name Please transfer the answers onto the bubble sheet. The question number refers
PHA 5127 Final Exam Fall 2012 On my honor, I have neither given nor received unauthorized aid in doing this assignment. Name Please transfer the answers onto the bubble sheet. The question number refers
We are IntechOpen, the world s leading publisher of Open Access books Built by scientists, for scientists. International authors and editors
 We are IntechOpen, the world s leading publisher of Open Access books Built by scientists, for scientists 3,900 116,000 120M Open access books available International authors and editors Downloads Our
We are IntechOpen, the world s leading publisher of Open Access books Built by scientists, for scientists 3,900 116,000 120M Open access books available International authors and editors Downloads Our
Extraction, Isolation and Analysis of Cannabis Extract and Preparation
 Extraction, Isolation and Analysis of Cannabis Extract and Preparation A S S O C.PROF. D R. N A R I SA K A M K AEN D I R EC TO R O F H E R BAL P H A R MACY C O L L EG E O F P H A R MACY, R A N G S I T
Extraction, Isolation and Analysis of Cannabis Extract and Preparation A S S O C.PROF. D R. N A R I SA K A M K AEN D I R EC TO R O F H E R BAL P H A R MACY C O L L EG E O F P H A R MACY, R A N G S I T
Aug 28 th, 2017 Pierre Daublain
 Analyzing the Potential Root Causes of Variability of Pharmacokinetics in Preclinical Species to Inform Derisking Strategies in Discovery and Early Development Aug 28 th, 2017 Pierre Daublain Outline Problem
Analyzing the Potential Root Causes of Variability of Pharmacokinetics in Preclinical Species to Inform Derisking Strategies in Discovery and Early Development Aug 28 th, 2017 Pierre Daublain Outline Problem
Supplemental Information
 Supplemental Information Article Title:The Proton Pump Inhibitor, Omeprazole, but not Lansoprazole or Pantoprazole, is a Metabolism-Dependent Inhibitor of CYP2C19: Implications for Co-Administration with
Supplemental Information Article Title:The Proton Pump Inhibitor, Omeprazole, but not Lansoprazole or Pantoprazole, is a Metabolism-Dependent Inhibitor of CYP2C19: Implications for Co-Administration with
The British Columbia Coroners Service is committed to conducting a thorough, independent examination of the factors contributing to death in order to
 The British Columbia Coroners Service is committed to conducting a thorough, independent examination of the factors contributing to death in order to improve community safety and quality of life in the
The British Columbia Coroners Service is committed to conducting a thorough, independent examination of the factors contributing to death in order to improve community safety and quality of life in the
PHA First Exam Fall 2003
 PHA 5127 First Exam Fall 2003 On my honor, I have neither given nor received unauthorized aid in doing this assignment. Name Question/Points 1. /14 pts 2. /6 pts 3. /15 pts 4. /12 pts 5. /20 pts 6. /10pts
PHA 5127 First Exam Fall 2003 On my honor, I have neither given nor received unauthorized aid in doing this assignment. Name Question/Points 1. /14 pts 2. /6 pts 3. /15 pts 4. /12 pts 5. /20 pts 6. /10pts
Cannabinoid Related Mortality in Denver. James Caruso, MD Chief Medical Examiner/Coroner Denver, Colorado **No commercial/financial disclosures**
 Cannabinoid Related Mortality in Denver James Caruso, MD Chief Medical Examiner/Coroner Denver, Colorado **No commercial/financial disclosures** Cannabis Legal Status March 2014 Fatality Case #1 19 y/o
Cannabinoid Related Mortality in Denver James Caruso, MD Chief Medical Examiner/Coroner Denver, Colorado **No commercial/financial disclosures** Cannabis Legal Status March 2014 Fatality Case #1 19 y/o
ICH Topic S1C(R2) Dose Selection for Carcinogenicity Studies of Pharmaceuticals. Step 5
 European Medicines Agency October 2008 EMEA/CHMP/ICH/383/1995 ICH Topic S1C(R2) Dose Selection for Carcinogenicity Studies of Pharmaceuticals Step 5 NOTE FOR GUIDANCE ON DOSE SELECTION FOR CARCINOGENICITY
European Medicines Agency October 2008 EMEA/CHMP/ICH/383/1995 ICH Topic S1C(R2) Dose Selection for Carcinogenicity Studies of Pharmaceuticals Step 5 NOTE FOR GUIDANCE ON DOSE SELECTION FOR CARCINOGENICITY
The clinical trial information provided in this public disclosure synopsis is supplied for informational purposes only.
 The clinical trial information provided in this public disclosure synopsis is supplied for informational purposes only. Please note that the results reported in any single trial may not reflect the overall
The clinical trial information provided in this public disclosure synopsis is supplied for informational purposes only. Please note that the results reported in any single trial may not reflect the overall
PHARMACOKINETICS SMALL GROUP I:
 PHARMACOKINETICS SMALL GROUP I: Question 1 Absorption of the anti-fungal agent, itraconazole, is dependent on a low gastric ph. Calculate the relative concentrations of a weak acid (with a pka of 5.4)
PHARMACOKINETICS SMALL GROUP I: Question 1 Absorption of the anti-fungal agent, itraconazole, is dependent on a low gastric ph. Calculate the relative concentrations of a weak acid (with a pka of 5.4)
PHA 5127 FINAL EXAM FALL On my honor, I have neither given nor received unauthorized aid in doing this assignment.
 PHA 5127 FINAL EXAM FALL 1997 On my honor, I have neither given nor received unauthorized aid in doing this assignment. Name Question Points 1. /14 pts 2. /10 pts 3. /8 pts 4 /8 pts 5. /12 pts 6. /8 pts
PHA 5127 FINAL EXAM FALL 1997 On my honor, I have neither given nor received unauthorized aid in doing this assignment. Name Question Points 1. /14 pts 2. /10 pts 3. /8 pts 4 /8 pts 5. /12 pts 6. /8 pts
Biomath M263 Clinical Pharmacology
 Training Program in Translational Science Biomath M263 Clinical Pharmacology Spring 2013 www.ctsi.ucla.edu/education/training/webcasts Wednesdays 3 PM room 17-187 CHS 4/3/2013 Pharmacokinetics and Pharmacodynamics
Training Program in Translational Science Biomath M263 Clinical Pharmacology Spring 2013 www.ctsi.ucla.edu/education/training/webcasts Wednesdays 3 PM room 17-187 CHS 4/3/2013 Pharmacokinetics and Pharmacodynamics
Jack Cook, PhD & Vivek Purohit, PhD Clinical Pharmacology Pfizer, Inc.
 Challenges and Strategies to Facilitate Formulation Development of Pediatric Drug Products Biopharmaceutical Considerations in Pediatric Formulation Development Jack Cook, PhD & Vivek Purohit, PhD Clinical
Challenges and Strategies to Facilitate Formulation Development of Pediatric Drug Products Biopharmaceutical Considerations in Pediatric Formulation Development Jack Cook, PhD & Vivek Purohit, PhD Clinical
Varun Goel 1, Nathalie H Gosselin 2, Claudia Jomphe 2, Husain Attarwala 1, Xiaoping (Amy) Zhang 1, JF Marier 2, Gabriel Robbie 1
 Population Pharmacokinetic (PK) / Pharmacodynamic (PD) Model of Serum Transthyretin (TTR) Following Patisiran Administration in Healthy Volunteers and Patients with Hereditary TTR-Mediated (hattr) Amyloidosis
Population Pharmacokinetic (PK) / Pharmacodynamic (PD) Model of Serum Transthyretin (TTR) Following Patisiran Administration in Healthy Volunteers and Patients with Hereditary TTR-Mediated (hattr) Amyloidosis
Effective Date: Approved by: Laboratory Executive Director, Ed Hughes (electronic signature)
 1 Policy #: 803 (PLH-803-02) Effective Date: NA Reviewed Date: 4/11/2008 Subject: URINE DRUG SCREENS Approved by: Laboratory Executive Director, Ed Hughes (electronic signature) Approved by: Laboratory
1 Policy #: 803 (PLH-803-02) Effective Date: NA Reviewed Date: 4/11/2008 Subject: URINE DRUG SCREENS Approved by: Laboratory Executive Director, Ed Hughes (electronic signature) Approved by: Laboratory
Predictive modeling of deposition, dissolution, absorption and systemic exposure
 Predictive modeling of deposition, dissolution, absorption and systemic exposure IPAC-RS/UF Orlando Inhalation Conference March 20, 2014 Per Bäckman and Bo Olsson, AstraZeneca R&D, Mölndal Sweden The views
Predictive modeling of deposition, dissolution, absorption and systemic exposure IPAC-RS/UF Orlando Inhalation Conference March 20, 2014 Per Bäckman and Bo Olsson, AstraZeneca R&D, Mölndal Sweden The views
IPAC-RS/UF Orlando Inhalation Conference March 20, S.T. Horhota 1, C.B. Verkleij 2, P.J.G. Cornelissen 2, L. Bour 3, A. Sharma 3, M.
 IPAC-RS/UF Orlando Inhalation Conference March 20, 2014 Case Study: Pharmacokinetics and Pharmacodynamics of Tiotropium and Salmeterol Following Parallel Administration in COPD Patients Using Different
IPAC-RS/UF Orlando Inhalation Conference March 20, 2014 Case Study: Pharmacokinetics and Pharmacodynamics of Tiotropium and Salmeterol Following Parallel Administration in COPD Patients Using Different
SYNOPSIS. The study results and synopsis are supplied for informational purposes only.
 SYNOPSIS INN : LEFLUNOMIDE Study number : HMR486/1037 et HMR486/3503 Study title : Population pharmacokinetics of A77 1726 (M1) after oral administration of leflunomide in pediatric subjects with polyarticular
SYNOPSIS INN : LEFLUNOMIDE Study number : HMR486/1037 et HMR486/3503 Study title : Population pharmacokinetics of A77 1726 (M1) after oral administration of leflunomide in pediatric subjects with polyarticular
Drug dosing in Extremes of Weight
 Drug dosing in Extremes of Weight The Plump & Heavy versus The Skinny & Light Maria Minerva P. Calimag, MD, MSc, PhD, DPBA, FPSECP PROFESSOR Departments of Pharmacology, Anesthesiology and Clinical Epidemiology
Drug dosing in Extremes of Weight The Plump & Heavy versus The Skinny & Light Maria Minerva P. Calimag, MD, MSc, PhD, DPBA, FPSECP PROFESSOR Departments of Pharmacology, Anesthesiology and Clinical Epidemiology
Nontraditional PharmD Program PRDO 7700 Pharmacokinetics Review Self-Assessment
 Nontraditional PharmD Program PRDO 7700 Pharmacokinetics Review Self-Assessment Please consider the following questions. If you do not feel confident about the material being covered, then it is recommended
Nontraditional PharmD Program PRDO 7700 Pharmacokinetics Review Self-Assessment Please consider the following questions. If you do not feel confident about the material being covered, then it is recommended
