National Institute for Health and Clinical Excellence
|
|
|
- Shauna Russell
- 5 years ago
- Views:
Transcription
1 National Institute for Health and Clinical Excellence 218_2 Hysteroscopic sterilisation by tubal cannulation and placement of intrafallopian implants Consultation Comments table IPAC date: Thursday 16 th April 1 Consultee 1, Sec. Comments Response 1 We would like to recommend that Ensure that patients understand that additional contraception must be used until imaging confirms tubal occlusion and that there is a small risk of pregnancy in the longer term after any form of tubal occlusion procedure. be modified to state: Ensure that patients understand that additional contraception must be used until imaging confirms proper placement of the micro-inserts, and, if necessary, HSG confirms tubal occlusion. Thank you for your comment. Sections 1.2 and of the guidance will be changed. However, section 1.2 will still state that there is a small risk of pregnancy in the longer term after any form of tubal occlusion procedure. Note that there is a small risk of pregnancy in the longer term after any form of permanent birth control procedure. The modification to the statement regarding imaging would be more accurate because the Essure Confirmation Test protocol involves an initial x-ray at 3 months only if the placement appears suspicious is an HSG required to evaluate occlusion. The modification to the risk of pregnancy statement helps patients understand that there is a small risk of pregnancy for any permanent birth control procedure. 1 of 22
2 2 Consultee 2, 3 Consultee 2, 4 Consultee 3, Sec. Comments Response : The Adiana procedure requires verification of bilateral tubal occlusion through an hysterosalpingography (HSG) procedure. Thank you for your comment. Section of the guidance details the use of imaging and HSG after the procedure. The IP programme issues guidance on procedures rather than individual devices. 2.1 no comments Thank you for your comment. 2.1 The commonest female sterilisation technique is Laparosopic Serilisation using Filshie clips to achieve tubal occlusion under general anaesthetic. Thank you for your comment. Section lists laparoscopic sterilisation as a comparator procedure for female sterilisation. 5 Consultee 1, 2.2 We would like to recommend that A small, flexible titanium microinsert is passed through the hysteroscope using a guidewire and placed into each of the fallopian tubes. be changed to: A small, flexible nickel-titanium & stainless steel microinsert is passed through the hysteroscope using a guidewire and placed into each of the fallopian tubes. Â because the micro-insert is composed of nitinol, a nickel-titanium alloy, stainless steel and PET (polyethylene terethalate fibers). We would also like to recommend that An additional form of contraception is used for 3 months until imaging (such as hysterosalpingogram [HSG] or X-ray) confirms correct placement of the microinserts and HSG can ensure that the fallopian tubes have become occluded. Â be changed to An additional form of contraception is used for 3 months until imaging (such as an X-ray) confirms proper placement of the micro-inserts, and, if necessary, HSG confirms tubal occlusion. in order to be consistent with device labeling. Thank you for your comment. The IP programme issues guidance on procedures rather than individual devices. Sections and of the guidance will be changed to become more generic. Section will be added to state that a number of different devices may be used for this procedure. 2 of 22
3 6 Consultee 3, Sec. Comments Response 2.2 It is entirely possible to perform the procedure, in my experience, without the need for local anaesthetic and/or intravenous sedation. Â I advise the patient to take simple analgesia (eg NSAID or paracetamol based analgesic) 1 hr prior to the procedure. I carry out the procedure regularly in the out-patient setting using the vaginoscopic technique. Â In so doing, this obviates the need for the use of a speculum, volsellum. local anaesthetic or intravenous sedation. Â In adopting the vaginoscopic technique, the average procedure time (from introduction to removal of the hysteroscope from the vagina) is 4-5 mins. Provided the placement of the devices appears satisfactory at the time of insertion, device placement confirmation can be made by TA/TV ultrasound at 3 months. If this is satisfactory the patient can be considered adequately sterilised. Â If there is any doubt on the placement of the devices however an HSG can then be done. Â This is the follow up protocol I have adopted in my experience of the technique. Thank you for your comment. Section of the guidance will be changed to include the term usually. Section will be changed to include ultrasound scanning. 3 of 22
4 7 Consultee 2, 8 Consultee 6, Sec. Comments Response : A small, flexible titanium microinsert or polymer matrix is passed through the hysteroscope using a delivery catheter and placed into each of the fallopian tubes. With the titanium microinserts, scar tissue formation is induced which occludes the fallopian tubes and prevents conception. With the polymer matrix, a light application of radio frequency energy is applied to create a superficial lesion around the polymer matrix. This initiates an acute wound healing response, causing healthy tissue to grow into the matrix, anchoring it in place and preventing conception : The polymer matrix procedure requires confirmation of tubal occlusion through an hysterosalpingogram (HSG) Specific Comment An additional form of contraception is used for 3 months until imaging (such as hysterosalpingogram [HSG] or X-ray) confirms correct placement of the microinserts and HSG can ensure that the fallopian tubes have become occluded. COMMENT: The use of pelvic ultrasound be added to the imaging tests stated above Thank you for your comment. Section will be changed to include radiofrequency ablation technique. Section of the guidance includes HSG. Thank you for your comment. Section will be changed to include ultrasound scanning. 4 of 22
5 9 Consultee 2, Sec. Comments Response 2.3 The Pivotal Clinical Trial for the Adiana System was published in the December 2008 edition of Obstetrics & Gynecology. Efficacy data is as follows: Case series of 645 women, successful bilateral placement in 95% (611/645) of women. Tubal occlusion was confirmed by HSG in 94% (570/604) of women 19/645 women had tubal patency at 3 months, but a second HSG at 6 months confirmed occlusion. In 645 women 6 pregnancies were reported. Â Three pregnancies resulted from misread HSGs and faulty reliance and the other three from indeterminate causes. In this study 99% (524 of 531) of women were very satisfied or satisfied with the procedure. 99% (525 of 532) of women reported their overall comfort as excellent or very good. 98% (626 of 636) of women returned to normal activities by 48 hours postprocedure of these, 90% (560 of 626) resumed normal activities the day after the procedure. Reference: Thierry G. Vancaillie, MD, FRANZCOG, Ted L. Anderson, MD, PhD, and D. Alan Johns MD A 12-Month Prospective Evaluation of Transcervical Sterilization Using Implantable Polymer Matrices. Obstet Gynecol : Thank you for your comment. This trial was identified in the post-consultation literature search and will be included in the guidance and table 2 of the overview. 5 of 22
6 10 Consultee 2, Sec. Comments Response 2.4 In the case of 645 women in the polymer matrix study 40.2% reported very little or no discomfort during the procedure and 9.2% were very uncomfortable. 80% (504/629) of women reported that the discomfort they experienced during the procedure and at periopertive intervals was the same or less than anticipated. 98% (619/632) described the tolerance of the procedure to be well, very well, or excellent. No uterine or tubal perforations were reported. No injuries related to RF energy or matrix placement occurred. Most procedure-related adverse events were mild in duration and resolved without intervention. No allergic or adverse reactions to the matrices occurred, and there were no known expulsions of the matrices. There were no requests by patients to have the polymer matrices removed. Hyponatremia (sodium 129 meq/l) occurred in one case (0.15%), and was successfully treated with induced diuresis without sequelae. Thank you for your comment. This trial was identified in the post-consultation literature search and will be included in the guidance and table 2 of the overview. 6 of 22
7 11 Consultee 4, Sec. Comments Response I would like to comment on the provisional recommendations NICE have published on the above technique. Having reviewed the literature it does seem to me that the medium and longterm efficacy of hysteroscopic sterilisation by tubal cannulation and placement of intrafallopian implants is as good as laparoscopic sterilisation although I agree that there is a need for more longterm studies and data to be collected. Essure has a good published safety profile and increasingly units of publishing that abdominal x-rays are an adequate method of assessing correct placement and hysterosalpingography may not necessarily be needed except in cases of initial abdominal x-ray uncertainty. I would certainly support the approval of this method of hysteroscopic sterilisation as another important option for women and their doctors to discuss while considering permanent sterilisation. [Conflict of Interests: consultant to Conceptus] Thank you for your comment. 7 of 22
8 12 Consultee 3, Sec. Comments Response I am a gynaecologist of 20 years standing with a specialist interest in Hysteroscopic Surgery. In my practice, I have also performed numerous Laparoscopic Sterilisation procedures over the years. Being aware from the literature that Essure Hysteroscopic Sterilisation has a superior safety profile as well as being more effective than Laparoscopic Tubal Occlusion, I have worked hard over the last 5 years or so to introduce Essure Hysteroscopic Sterilisation to my unit in Edinburgh. I started the procedure in Edinburgh in December I was funded initially for 75 patients and have completed Essure for 52 patients to date. It is my belief that Essure Hysteroscopic Sterilisation should be offered to all women requesting sterilisation. Its success in my unit has justified the case for ongoing funding. Thank you for your comment. The IP programme issues guidance on procedures rather than individual devices. 8 of 22
9 13 Consultee 3, Sec. Comments Response As noted in the Royal College of Obstetricians and Gynaecologists s guidance on Laparoscopic Tubal Occlusion (LTO), LTO has an estimated lifetime failure rate is 1 in 200. Although Essure s lifetime failure rate has not been calculated, the estimated ten year failure rate is 0.31%, or less than 1 in 300. The manufacturer of Essure has reported that the pregnancies reported in commercial use number 300 out of 259,000 Essure procedures, or less than 1 in 800. Although it can be assumed that there may be underreporting in commercial practice, both the commercial and clinical data demonstrate that essure s long-term effectiveness supports its use as the standard for sterilisation. Thank you for your comment. 9 of 22
10 14 Consultee 3, 15 Consultee 3, Sec. Comments Response The choice of Essure over Laparoscopic Tubal Occlusion is even stronger when comparing the safety of the two procedures. Both procedures carry the risk of failed procedures (failure to gain entry to the abdomen for LTO vs. bilateral placement failure for Essure), the risk of ectopic pregnancy, and the risk of uterine perforation. However, because LTO requires a laparoscopic approach while Essure requires a hysteroscopic approach with minimal or more often no anaesthesia, LTO carries additional risks. For LTO procedures, injuries to the bowel, bladder or blood vessels are serious but infrequent: 2-3 in every 1000 procedures. One study has suggested that 1 woman in every undergoing laparoscopy dies as a result of complications. The use of general anaesthesia in LTO procedures also carries risks which must be considered. In performing Essure Hysteroscopic Sterilisation it is entirely possible in the vast majority of cases to adopt the Vaginoscopic technique whereby there is no need for instrumentation such as introduction of a specululm, use of a volsellum or the need for local anaesthetic or any form of sedation. The average duration for the Vaginoscopic Essure Hysteroscopic Sterilisation procedure in the out-patient setting is 4-5 mins in my current experience. Prior to the procedure I merely advise the patient to take simple analgesia (eg NSAID or paracetamol based analgesia) 1 hour prior to the appointment. Many patients do not require additional analgesia following the procedure. Thank you for your comment. This falls outside the scope of the guidance. Thank you for comment. This falls outside the scope of the guidance. Section of the guidance will be changed to include the term usually. 10 of 22
11 16 Consultee 3, Sec. Comments Response Because the Essure procedure uses a hysteroscoipc approach, risks due to surgical incision, intraabdominal exploration and general anaesthesia can be eliminated. Risks and side effects for the Essure procedure are generally mild, and many do not require further medical intervention for resolution. Risks include pain, cramping, vaginal bleeding and occasionally a vasovagal reaction. Regards the latter I have to date had 1 out of 52 patients who has had a vasovagal response. Thank you for your comment. These outcomes are included in the safety section of the guidance. 11 of 22
12 17 Consultee 3, Sec. Comments Response It is obviously imperative the patient be counseled to continue with their pre-essure contraception following the procedure until they return for the 3 month Confirmation Test to check placement of the devices. If the Essure procedure appears straightforward with 3-8 coils seen trailing into the cavity after positioning and deployment, my practice is for a TA/TV ultrasound scan 3 months later. If device placement at this is satisfactory, the patient is informed she is adequately sterilized although there is still a very small failure rate as aforementioned. She can then discontinue her other method of contraception. I would only suggest a Hysterosalpingogram (HSG) if the Essure procedure is difficult with the risk of suboptimal placement or there is any doubt on the placement at the 3 month Confirmation ultrasound examination. It is because of Essure s safety profile, high effectiveness and patient satisfaction rates that I believe the Essure procedure should be a woman s first choice for sterilisation. Thank you for your comment. Section will be changed to include ultrasound scanning. The IP programme produces guidance on procedures rather than individual devices. 12 of 22
13 18 Consultee 5, 19 Consultee 5, Sec. Comments Response I am the Vice President of Europe for Conceptus, Inc., manufacturer of the Essure System for permanent birth control. Conceptus would like to provide additional information regarding Essure to assist you with finalizing the NICE draft guidance. We are submitting a marked PDF of the draft guidance with small changes that aid in the accuracy of the guidance [see comments from Consultee 1]. More than 275,000 women worldwide have undergone the Essure procedure since Essure was certified with its CE mark in The strength of evidence for the Essure Procedure shows that the procedure offers substantial benefits over the current surgical regimen, tubal ligation. Conceptus clinical trials demonstrated that Essure s effectiveness is unparalleled; preliminary analysis of nine years of long-term clinical trial data and over 45,068 women-months of Essure reliance confirms that there have been zero reported pregnancies among women relying upon Essure.(1) This preliminary data was presented by Dr. Linda Bradley at the AAGL 2008 Global Congress. The following table shows Essure s adjusted failure rate as compared to CREST, the Collaborative Review of Sterilization. Years 9 & 10 are estimates. [See table at the end of this consultation comments table.] Thank you for your comment. Please see responses to consultee 1. The IP programme issues guidance on procedures rather than individual devices. Thank you for your comment. The IP programme issues guidance on procedures rather than individual devices. 13 of 22
14 20 Consultee 5, 21 Consultee 5, 22 Consultee 5, Sec. Comments Response The low rate of pregnancies reported in commercial use supports the results obtained in the clinical trials.(2) From Essure s commercial release in 2001 through December 2008, 300 pregnancies were reported out of an estimated 259,000 procedures performed.(2) Reported pregnancies tend to be related to noncompliance with the Essure instructions for use or a misread Essure confirmation test. (3) Essure also exhibits an exceptional safety profile. One researcher summarized that compared to laparoscopic sterilisation, the hysteroscopic approach obviates admission, general anaesthesia, surgical incisions and a complication rate of 5 per 1,000 procedures. (4) Adverse events observed during the Essure Procedure are generally mild, and many do not require further medical intervention for resolution. Patients are extremely satisfied with Essure. In one recent study, all study subjects were highly satisfied after hysteroscopic sterilisation: 91% of subjects rated the method at 10 (high satisfaction degree on a 0 to 10 visual analog scale) and none of the subjects rated it under 8.(5) More than 97% of the patients said that they would recommend the procedure to others. (5) 86.5% of study subjects considered the procedure painless or scarcely painful.(5) The most valuable aspects of the Essure procedure were stated to be absence of surgery room (52.7%), method's quickness and comfort (19.9%), and permanent sterilisation (18.2%). (5) Thank you for your comment. Thank you for your comment. This falls outside the scope of the guidance. Thank you for your comment. 14 of 22
15 23 Consultee 5, 24 Consultee 6, Sec. Comments Response In summary, both the published literature and Essure s record in commercial use demonstrate the substantial benefits of effectiveness, safety, and patient satisfaction offered by Essure. As concluded by one of the researchers, the Essure procedure should be one of the first-line options for female permanent sterilisation. (5) Thank you for your telephone message agreeing to receive my belated comments regarding the provisional NICE guidance for IP218. I would like to outline my views supporting the more widespread use of outpatient hysteroscopic hysteroscopic sterilisation using the Essure TM technique and also to comment on radiological follow up supporting the use of pelvic ultrasound. Thank you for your comment. The IP programme issues guidance on procedures rather than individual devices. Thank you for your comment. The IP programme issues guidance on procedures rather than individual devices. Section will be changed to include ultrasound scanning. I am a Consultant Obstetrician and Gynaecologist at the Birmingham Women s Hospital and I have a major interest in outpatient hysteroscopic techniques. I have been performing outpatient hysteroscopic sterilisation using the Essure TM permanent birth control system since 2002 and have published our initial experience in the British Journal of Obstetrics and Gynaecology (1). To date I have performed over 350 Essure sterilisation procedures and we are prospectively collecting all our data on a secure database (available on request), from which we hope to publish further work. 15 of 22
16 25 Consultee 6, Sec. Comments Response We, along with many others (2), have demonstrated that outpatient hysteroscopic sterilisation using the Essure TM system is safe, convenient, feasible and effective. It does not require the use of a vaginal speculum, cervical dilatation or local anaesthesia in the majority of women. It has major advantages over traditional laparoscopic sterilisation, in that it avoids the need for admission to hospital, risks from general anaesthesia, need for abdominal incisions and damage to internal organs (bowel, bladder, vasculature etc. estimated 3 in 1000 procedures) associated with the use of closed entry laparoscopy. In my experience, the only complication arising from Essure TM sterilisation is mild, self-limiting vasovagal responses in up to 5% of women (1). To my knowledge (personal and from the published literature), no serious, life-threatening complications have been associated with the technique. This contrasts markedly with traditional laparoscopic sterilisation i.e. the risks of intra-abdominal visceral injury are likely to increase over time in view of the increasing rates of caesarian sections (leading to abdominal adhesions) and obesity which are known to be independent risk factors for bowel and vascular injury at laparoscopy. In addition to potential serious morbidity with laparoscopy, it should be noted that women die every year from complications arising from laparoscopic sterilisation. Thank you for your comment. This falls outside the scope of the guideline. 16 of 22
17 26 Consultee 6, 27 Consultee 6, Sec. Comments Response Laparoscopic sterilisation has an estimated lifetime failure rate of up to is 1 in 200 (3). The failure rate associated with the EssureTM technique compares favourably, with an estimated ten year failure rate of less than 1 in 300. The manufacturer of Essure has reported that the pregnancies reported in commercial use number 300 out of 259,000 Essure procedures, or less than 1 in 800, although this figure reflects reported cases only (source Conceptus). The feasibility and effectiveness rates are consistent across the increasing number of published studies pertaining to EssureTM sterilisation in both the short and medium term (2). Longer-term effectiveness data will inevitably become available over time, but there is no plausible reason to assume that failure rates should dramatically increase in the longer term over what the short and medium term data tell us now. In view of the safety, convenience, short recovery time, avoidance of abdominal entry and incisions and effectiveness, I believe that the evidence supporting the first-line use of outpatient hysteroscopic sterilisation using the Essure TM permanent birth control system is compelling and that the technique should be available to all women requesting permanent birth control. Moreover, with the increasing body of published data supporting the technique, I believe that the outpatient hysteroscopic Essure TM sterilisation should replace laparoscopic approaches as the first line method of female sterilisation. 17 of 22 Thank you for your comment. Thank you for your comment. The IP programme issues guidance on procedures rather than individual devices.
18 28 Consultee 6, 29 Consultee 6, Sec. Comments Response Abdominal X-ray (AXR) is considered the first line confirmation test following the EssureTM procedure in the UK (4-9)(approved by Conceptus notified body for CE marking purposes). An AXR confirms satisfactory placement of the EssureTM microinserts. Where device placement is considered unsatisfactory, additional contraception should be continued and an HSG performed (10). We published the largest series to date from the UK on outpatient hysteroscopic sterilisation using the EssureTM permanent birth control system (1). We chose to use HSG initially to confirm tubal occlusion. We surveyed patient satisfaction with the overall procedure (including radiological follow up with HSG) and found whilst the overall procedure was associated with high levels of satisfaction, some women found the HSG somewhat invasive. Whilst our follow up rates were good (95%), we, along with other published series, had patients who did not attend for follow up. We were concerned that this noncompliance in a minority may, in part, be attributable to the need for an HSG. On completion of our first 200 outpatient procedures, we were going to amend our protocols to using less invasive AXR as the first line confirmation test for uncomplicated procedures (as recommended by the manufacturer s), but following discussions with international colleagues it became clear that many experienced users were utilising pelvic transvaginal ultrasound (TVS) rather than AXR or HSG to confirm satisfactory tubal placement of the EssureTM microinserts. Thank you for your comment. Section describes confirmation tests for the procedure. Thank you for your comment. Section will be changed to include ultrasound scanning. 18 of 22
19 30 Consultee 6, Sec. Comments Response TVS offers potential advantages over AXR in that it has the ability to locate the devices within the enveloping tissue, additional information is gained about surrounding soft-tissue structures and the position of the device within the utero-tubal junction can be displayed on ultrasound while it can be merely be inferred from plain X-ray films. Moreover, TVS is relatively non-invasive (compared to HSG), cheap and avoids the use of potentially harmful ionising radiation. Published data (from the US, Australia and Europe) (11-15) supports the use of TVS to confirm satisfactory tubal placement of the EssureTM microinserts. In view of this, since January 2008, our unit has introduced TVS as the first line imaging technique to confirm satisfactory bilateral tubal placement at 3 months post-procedure, with recourse to HSG for equivocal results or where procedures have been considered not to be straightforward (our protocol and follow up algorithm is available on request includes conservative definitions of what constitutes a straightforward procedure and when to use TVS and HSG). I know that a similar approach, with follow up based upon TVS, is adopted across the Netherlands (Personal communication S Veersema). Thank you for your comment. Section will be changed to include ultrasound scanning. 19 of 22
20 31 Consultee 6, Sec. Comments Response Thus the requirement of radiological follow up, posthysteroscopic sterilisation using the EssureTM system, is to confirm satisfactory placement of the microinserts and this can be achieved by TVS as well as AXR, with the advantages of TVS described above. HSG can be used to confirm tubal occlusion, but given its more invasive nature (as well as associated costs and morbidity), it should be reserved for equivocal AXR or TVS results following standard EssureTM procedures or as a first line where the EssureTM procedure is considered difficult or suboptimal. It is my belief that in time we will be able to dispense altogether with radiological follow up following standard EssureTM procedures (as we did with laparoscopic sterilisations using the Filshie clip). However, for the time being, whilst the procedure is becoming more widely established, we should continue with radiological follow up based upon TVS, AXR or HSG. Thank you for your comment. Section will be changed to include ultrasound scanning. All studies referenced below have been considered. References 1. Sinha D, Kalathy V, Gupta JK, Clark TJ. The feasibility, success and patient satisfaction associated with outpatient hysteroscopic sterilisation. BJOG 2007;114: NICE overview IP 218 (November 2008) 3. National Evidence Based Guideline. Male and Female Sterilisation 2004 (Available at ) 4. Cooper JM, Carignan CS, Cher D, Kerin JF, Microinsert nonincisional 5. hysteroscopic sterilization, Obstet Gynecol, 2003;102: of 22
21 31 cont. Consultee 6, (cont) Sec. Comments Response (cont) 6. Kerin JF, Cooper JM, Price T, et al., Hysteroscopic sterilization using a micro-insert device: results of a multicentre phase II study, Hum Reprod, 2003;18: Ubeda A, Labastida R, Dexeus S, Essure: a new device for hysteroscopic tubal sterilization in an outpatient setting, Fertil Steril, 2004;82(1): Heredia F, Cos R, Moros S,Torrabadella L, Radiological control of Essure placements, Surg, 2004;1: Gynecol 9. Conceptus SAS, Physician Training Manual: X- ray Protocol, 2003; Conceptus USA, US Physician Training Manual: HSG Protocol, TR ; Teoh M, Meagher S, Kovacs G, Ultrasound detection of the Essure permanent birth control device: a case series, Aus NZ J Obstet, 2003:43: Kerin JF, Levy BS, Ultrasound: an effective method for localization of the echogenic Essure sterilization microinsert: correlation with radiologic evaluations, J Minim Invasive Gynecol, 2005;12: "Comments received in the course of consultations carried out by NICE are published in the interests of openness and transparency, and to promote understanding of how recommendations are developed. The comments are published as a record of the submissions that NICE has received, and are not endorsed by NICE, its officers or advisory committees." 21 of 22
22 Essure Long-Term Effectiveness Rate from Clinical Trials (posterior mean, age adjusted) (est) Essure 99.97% 99.93% 99.90% 99.87% 99.84% 99.80% 99.77% 99.74% 99.71% [Table submitted as part of the consultation response from Consultee 4,. This graph will be removed prior to publication]. 22 of 22
Unintended pregnancies after Essure sterilization in the Netherlands
 Unintended pregnancies after Essure sterilization in the Netherlands S. Veersema M.P.H. Vleugels L.M. Moolenaar C.A.H. Janssen H.A.M. Brölmann Fertil Steril. 2010;93:35-8. 13 Hysteroscopic Sterilization
Unintended pregnancies after Essure sterilization in the Netherlands S. Veersema M.P.H. Vleugels L.M. Moolenaar C.A.H. Janssen H.A.M. Brölmann Fertil Steril. 2010;93:35-8. 13 Hysteroscopic Sterilization
Female sterilization by the natural pathways
 info-canada@conceptus.com www.essure.com Female sterilization by the natural pathways magazine 1.Hatcher R et al. Contraceptive Technology, 17 th Edition. New York : Ardent Media, 1998. 2. Bhiwandiwala
info-canada@conceptus.com www.essure.com Female sterilization by the natural pathways magazine 1.Hatcher R et al. Contraceptive Technology, 17 th Edition. New York : Ardent Media, 1998. 2. Bhiwandiwala
ESSURE A RESOURCE FOR CODING
 ESSURE REIMBURSEMENT GUIDE A RESOURCE FOR CODING INDICATION Essure is indicated for women who desire permanent birth control (female sterilization) by bilateral occlusion of fallopian tubes. IMPORTANT
ESSURE REIMBURSEMENT GUIDE A RESOURCE FOR CODING INDICATION Essure is indicated for women who desire permanent birth control (female sterilization) by bilateral occlusion of fallopian tubes. IMPORTANT
Female sterilisation: a cohort controlled comparative study of ESSURE versus laparoscopic sterilisation
 BJOG: an International Journal of Obstetrics and Gynaecology November 2005, Vol. 112, pp. 1522 1528 DOI: 10.1111/j.1471-0528.2005.00726.x Female sterilisation: a cohort controlled comparative study of
BJOG: an International Journal of Obstetrics and Gynaecology November 2005, Vol. 112, pp. 1522 1528 DOI: 10.1111/j.1471-0528.2005.00726.x Female sterilisation: a cohort controlled comparative study of
MEDICAL POLICY SUBJECT: FEMALE STERILIZATION. POLICY NUMBER: CATEGORY: Contract Clarification
 MEDICAL POLICY SUBJECT: FEMALE STERILIZATION PAGE: 1 OF: 5 If a product excludes coverage for a service, it is not covered, and medical policy criteria do not apply. If a commercial product, including
MEDICAL POLICY SUBJECT: FEMALE STERILIZATION PAGE: 1 OF: 5 If a product excludes coverage for a service, it is not covered, and medical policy criteria do not apply. If a commercial product, including
Clinical Experience With Contrast Infusion Sonography as an Essure Confirmation Test
 ORIGINAL RESEARCH Clinical Experience With Contrast Infusion Sonography as an Essure Confirmation Test Viviane F. Connor, MD Received November 3, 2010, from the Department of Gynecology, Section of Minimally
ORIGINAL RESEARCH Clinical Experience With Contrast Infusion Sonography as an Essure Confirmation Test Viviane F. Connor, MD Received November 3, 2010, from the Department of Gynecology, Section of Minimally
Update on the Essure System for Permanent Birth Control
 Update on the Essure System for Permanent Birth Control Heidi Collins Fantasia IIn 2002, the U.S. Food and Drug Administration (FDA) approved Essure (Bayer, Whippany, NJ), a minimally invasive procedure
Update on the Essure System for Permanent Birth Control Heidi Collins Fantasia IIn 2002, the U.S. Food and Drug Administration (FDA) approved Essure (Bayer, Whippany, NJ), a minimally invasive procedure
Satisfaction and tolerance with office hysteroscopic tubal sterilization
 TECHNIQUES AND INSTRUMENTATION Satisfaction and tolerance with office hysteroscopic tubal sterilization Jose E. Arjona, M.D., Ph.D., a Monica Mi~no, M.D., Ph.D., a Javier Cordon, M.D., Ph.D., a Balbino
TECHNIQUES AND INSTRUMENTATION Satisfaction and tolerance with office hysteroscopic tubal sterilization Jose E. Arjona, M.D., Ph.D., a Monica Mi~no, M.D., Ph.D., a Javier Cordon, M.D., Ph.D., a Balbino
Complications of hysteroscopic Essure â sterilisation: report on 4306 procedures performed in a single centre
 DOI: 10.1111/j.1471-0528.2012.03292.x www.bjog.org Fertility control Complications of hysteroscopic Essure â sterilisation: report on 4306 procedures performed in a single centre B Povedano, a JE Arjona,
DOI: 10.1111/j.1471-0528.2012.03292.x www.bjog.org Fertility control Complications of hysteroscopic Essure â sterilisation: report on 4306 procedures performed in a single centre B Povedano, a JE Arjona,
Permanent Sterilization: When you are really sure!
 Permanent Sterilization: When you are really sure! Tony Ogburn MD Department of Ob/Gyn 2006-8 National Survey of Family Growth 6.1% of women had a sterilized male partner The History of Female Sterilization
Permanent Sterilization: When you are really sure! Tony Ogburn MD Department of Ob/Gyn 2006-8 National Survey of Family Growth 6.1% of women had a sterilized male partner The History of Female Sterilization
SIMPLE PERMANENT CONTRACEPTION A HIGHLY EFFECTIVE AND COMPLICATION FREE BIRTH CONTROL DEVICE
 CONSIDER WHAT IS BEST FOR YOU... SIMPLE PERMANENT CONTRACEPTION A HIGHLY EFFECTIVE AND COMPLICATION FREE BIRTH CONTROL DEVICE Effective. Safe. Proven. FILSHIE - THE CLIP, A HIGHLY EFFECTIVE CHOICE FOR
CONSIDER WHAT IS BEST FOR YOU... SIMPLE PERMANENT CONTRACEPTION A HIGHLY EFFECTIVE AND COMPLICATION FREE BIRTH CONTROL DEVICE Effective. Safe. Proven. FILSHIE - THE CLIP, A HIGHLY EFFECTIVE CHOICE FOR
Labeling for Permanent Hysteroscopically-Placed Tubal Implants Intended for Sterilization
 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 Labeling for Permanent Hysteroscopically-Placed Tubal Implants Intended for Sterilization Draft Guidance for Industry and Food and Drug
1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 Labeling for Permanent Hysteroscopically-Placed Tubal Implants Intended for Sterilization Draft Guidance for Industry and Food and Drug
Essure Permanent Birth Control Device: Radiological followup results at our center
 Essure Permanent Birth Control Device: Radiological followup results at our center Poster No.: C-0212 Congress: ECR 2013 Type: Scientific Exhibit Authors: R. Díaz Aguilera, A. M. Higuera Higuera, V. Palomo
Essure Permanent Birth Control Device: Radiological followup results at our center Poster No.: C-0212 Congress: ECR 2013 Type: Scientific Exhibit Authors: R. Díaz Aguilera, A. M. Higuera Higuera, V. Palomo
Consider what is best for you...
 Consider what is best for you... PERMANENT CONTRACEPTION A HIGHLY EFFECTIVE AND COMPLICATION FREE BIRTH CONTROL DEVICE Effective. Safe. Proven. FILSHIE - THE CLIP, EASY TO CORRECTLY APPLY Quick laparoscopic
Consider what is best for you... PERMANENT CONTRACEPTION A HIGHLY EFFECTIVE AND COMPLICATION FREE BIRTH CONTROL DEVICE Effective. Safe. Proven. FILSHIE - THE CLIP, EASY TO CORRECTLY APPLY Quick laparoscopic
Transcervical Sterilization
 Q UESTIONS & ANSWERS A BOUT Transcervical Sterilization A New Choice in Permanent Birth Control Choosing a Birth Control Method Women and their partners now have more birth control choices than ever. How
Q UESTIONS & ANSWERS A BOUT Transcervical Sterilization A New Choice in Permanent Birth Control Choosing a Birth Control Method Women and their partners now have more birth control choices than ever. How
Myometrial Insertion of Essure Mirco-Insert
 Case Report http://www.alliedacademies.org/research-and-reports-in-gynecology-and-obstetrics Myometrial Insertion of Essure Mirco-Insert Jeffrey J Woo 1*, Barbara E Simpson 1, Dale W Stovall 2 1 Department
Case Report http://www.alliedacademies.org/research-and-reports-in-gynecology-and-obstetrics Myometrial Insertion of Essure Mirco-Insert Jeffrey J Woo 1*, Barbara E Simpson 1, Dale W Stovall 2 1 Department
Essure By Mayo Clinic staff
 Page 1 of 5 Reprints A single copy of this article may be reprinted for personal, noncommercial use only. Essure By Mayo Clinic staff Original Article: http://www.mayoclinic.com/health/essure/my00999 Definition
Page 1 of 5 Reprints A single copy of this article may be reprinted for personal, noncommercial use only. Essure By Mayo Clinic staff Original Article: http://www.mayoclinic.com/health/essure/my00999 Definition
Sterilisation for women at the RD&E: what you need to know Reference Number: CW
 Sterilisation for women at the RD&E: what you need to know Royal Devon and Exeter NHS Foundation Trust Patient Information Sterilisation for Women at The Royal Devon and Exeter Hospital What you need to
Sterilisation for women at the RD&E: what you need to know Royal Devon and Exeter NHS Foundation Trust Patient Information Sterilisation for Women at The Royal Devon and Exeter Hospital What you need to
Probability of pregnancy after sterilization: a comparison of hysteroscopic versus laparoscopic sterilization,
 Contraception xx (2014) xxx xxx Original research article Probability of pregnancy after sterilization: a comparison of hysteroscopic versus laparoscopic sterilization, Aileen M. Gariepy a,, Mitchell D.
Contraception xx (2014) xxx xxx Original research article Probability of pregnancy after sterilization: a comparison of hysteroscopic versus laparoscopic sterilization, Aileen M. Gariepy a,, Mitchell D.
Hysteroscopic Tubal Sterilization: An Evidence-Based Analysis
 Hysteroscopic Tubal Sterilization: An Evidence-Based Analysis K McMartin October 2013 Ontario Health Technology Assessment Series; Vol.13: No. 21, pp. 1 35, October 2013 Suggested Citation This report
Hysteroscopic Tubal Sterilization: An Evidence-Based Analysis K McMartin October 2013 Ontario Health Technology Assessment Series; Vol.13: No. 21, pp. 1 35, October 2013 Suggested Citation This report
V. Mijatovic S. Veersema M.H. Emanuel R. Schats P.G. Hompes. Fertil Steril. 2010;93:
 Essure hysteroscopic tubal occlusion device for the treatment of hydrosalpinx prior to in vitro fertilization-embryo transfer in patients with a contraindication for laparoscopy. V. Mijatovic S. Veersema
Essure hysteroscopic tubal occlusion device for the treatment of hydrosalpinx prior to in vitro fertilization-embryo transfer in patients with a contraindication for laparoscopy. V. Mijatovic S. Veersema
THE WOMAN-FRIENDLY STERILIZATION METHOD
 THE WOMAN-FRIENDLY STERILIZATION METHOD Urogyn BV Transistorweg 5a 6534 AT Nijmegen The Netherlands t +31(0) 24 711 41 30 info@urogynbv.com www.urogynbv.com THE MOST WOMAN-FRIENDLY STERILIZATION METHOD
THE WOMAN-FRIENDLY STERILIZATION METHOD Urogyn BV Transistorweg 5a 6534 AT Nijmegen The Netherlands t +31(0) 24 711 41 30 info@urogynbv.com www.urogynbv.com THE MOST WOMAN-FRIENDLY STERILIZATION METHOD
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE
 NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE INTERVENTIONAL PROCEDURES PROGRAMME Interventional procedure overview of sterilisation by insertion of intrafallopian implants Sterilisation is sometimes
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE INTERVENTIONAL PROCEDURES PROGRAMME Interventional procedure overview of sterilisation by insertion of intrafallopian implants Sterilisation is sometimes
INTERVENTIONAL PROCEDURES PROGRAMME
 NATIONAL INSTITUTE FOR CLINICAL EXCELLENCE INTERVENTIONAL PROCEDURES PROGRAMME Interventional procedure overview of microwave endometrial ablation Introduction This overview has been prepared to assist
NATIONAL INSTITUTE FOR CLINICAL EXCELLENCE INTERVENTIONAL PROCEDURES PROGRAMME Interventional procedure overview of microwave endometrial ablation Introduction This overview has been prepared to assist
Clinical Policy: Essure Removal Reference Number: CP.MP.131
 Clinical Policy: Reference Number: CP.MP.131 Effective Date: 11/16 Last Review Date: 11/17 Coding Implications Revision Log See Important Reminder at the end of this policy for important regulatory and
Clinical Policy: Reference Number: CP.MP.131 Effective Date: 11/16 Last Review Date: 11/17 Coding Implications Revision Log See Important Reminder at the end of this policy for important regulatory and
Laparoscopy-Hysteroscopy
 Laparoscopy-Hysteroscopy Patient Information Laparoscopy The laparoscope, a surgical instrument similar to a telescope, is inserted through a small incision (cut) in the belly button during laparoscopy.
Laparoscopy-Hysteroscopy Patient Information Laparoscopy The laparoscope, a surgical instrument similar to a telescope, is inserted through a small incision (cut) in the belly button during laparoscopy.
Laparoscopy and Hysteroscopy
 AMERICAN SOCIETY FOR REPRODUCTIVE MEDICINE Laparoscopy and Hysteroscopy A Guide for Patients PATIENT INFORMATION SERIES Published by the American Society for Reproductive Medicine under the direction of
AMERICAN SOCIETY FOR REPRODUCTIVE MEDICINE Laparoscopy and Hysteroscopy A Guide for Patients PATIENT INFORMATION SERIES Published by the American Society for Reproductive Medicine under the direction of
Outpatient hysteroscopy direct diagnostic access & new therapeutic procedures
 Outpatient hysteroscopy direct diagnostic access & new therapeutic procedures Mrs Katy Clifford Consultant Gynaecologist St Mary s Hospital Imperial College Healthcare NHS Trust Diagnostic outpatient hysteroscopy
Outpatient hysteroscopy direct diagnostic access & new therapeutic procedures Mrs Katy Clifford Consultant Gynaecologist St Mary s Hospital Imperial College Healthcare NHS Trust Diagnostic outpatient hysteroscopy
ESSURE CLINICAL RESOURCE
 ESSURE CLINICAL RESOURCE Physician Training Manual IMPORTANT SAFETY INFORMATION WARNING: Some patients implanted with the Essure System for Permanent Birth Control have experienced and/or reported adverse
ESSURE CLINICAL RESOURCE Physician Training Manual IMPORTANT SAFETY INFORMATION WARNING: Some patients implanted with the Essure System for Permanent Birth Control have experienced and/or reported adverse
If you have any further questions, please speak to a doctor or nurse caring for you.
 Having a laparoscopy This leaflet aims to answer your questions about having a laparoscopy. It explains the benefits, risks and alternatives, as well as what you can expect when you come to hospital. If
Having a laparoscopy This leaflet aims to answer your questions about having a laparoscopy. It explains the benefits, risks and alternatives, as well as what you can expect when you come to hospital. If
Female Sterilization. Kavita Nanda, MD, MHS FHI 360 Expanding Contraceptive Choice December 6, 2018
 Female Sterilization Kavita Nanda, MD, MHS FHI 360 Expanding Contraceptive Choice December 6, 2018 What is female sterilization? Family planning method that provides permanent contraception to women and
Female Sterilization Kavita Nanda, MD, MHS FHI 360 Expanding Contraceptive Choice December 6, 2018 What is female sterilization? Family planning method that provides permanent contraception to women and
Case Report Essure Surgical Removal and Subsequent Resolution of Chronic Pelvic Pain: A Case Report and Review of the Literature
 Case Reports in Obstetrics and Gynecology Volume 2016, Article ID 6961202, 5 pages http://dx.doi.org/10.1155/2016/6961202 Case Report Essure Surgical Removal and Subsequent Resolution of Chronic Pelvic
Case Reports in Obstetrics and Gynecology Volume 2016, Article ID 6961202, 5 pages http://dx.doi.org/10.1155/2016/6961202 Case Report Essure Surgical Removal and Subsequent Resolution of Chronic Pelvic
Aileen M. CON-08309; No of Pages 8. Contraception xx (2014) xxx xxx
 CON-08309; No of Pages 8 1 Original research article 2 Probability of pregnancy after sterilization: a comparison of hysteroscopic 3 versus laparoscopic sterilization, 4Q1 Aileen M. 5 6 7 Contraception
CON-08309; No of Pages 8 1 Original research article 2 Probability of pregnancy after sterilization: a comparison of hysteroscopic 3 versus laparoscopic sterilization, 4Q1 Aileen M. 5 6 7 Contraception
Consent Advice No. XX (Joint with BSGE) Peer Review Draft Spring Morcellation for Laparoscopic Myomectomy or Hysterectomy
 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 27 28 29 30 31 32 33 34 35 36 37 38 39 40 41 42 43 44 45 46 47 48 49 50 51 Consent Advice No. XX (Joint with BSGE) Peer Review Draft
1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 27 28 29 30 31 32 33 34 35 36 37 38 39 40 41 42 43 44 45 46 47 48 49 50 51 Consent Advice No. XX (Joint with BSGE) Peer Review Draft
CLINICAL GUIDELINES ID TAG Female Sterilisation (tubal occlusion) at Caesarean Section- Guideline for counselling and consent
 Title: Author: Designation: Speciality / Division: Directorate: CLINICAL GUIDELINES ID TAG Female Sterilisation (tubal occlusion) at Caesarean Section- Guideline for counselling and consent Dr Meeta Kamath
Title: Author: Designation: Speciality / Division: Directorate: CLINICAL GUIDELINES ID TAG Female Sterilisation (tubal occlusion) at Caesarean Section- Guideline for counselling and consent Dr Meeta Kamath
Hysteroscopic Tubal Occlusion for Contraception
 Hysteroscopic Tubal Occlusion for Contraception (Essure and Adiana Systems) Policy Number: 2014M0056A Effective Date: June 1, 2014 Table of Contents: Page: Cross Reference Policy: POLICY DESCRIPTION 2
Hysteroscopic Tubal Occlusion for Contraception (Essure and Adiana Systems) Policy Number: 2014M0056A Effective Date: June 1, 2014 Table of Contents: Page: Cross Reference Policy: POLICY DESCRIPTION 2
Clinics in diagnostic imaging (110)
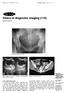 M e d i c a l E d u c a t i o n Singapore Med Med J 2006; J 2006; 47(7) 47(7) : 642 : 1 Clinics in diagnostic imaging (110) Lim S Y, Lam S L Fig. 1 Radiograph of the pelvis. Fig. 2 Sagittal and coronal
M e d i c a l E d u c a t i o n Singapore Med Med J 2006; J 2006; 47(7) 47(7) : 642 : 1 Clinics in diagnostic imaging (110) Lim S Y, Lam S L Fig. 1 Radiograph of the pelvis. Fig. 2 Sagittal and coronal
Information for parents. Selective Salpingogram (SS)
 Information for parents Selective Salpingogram (SS) What is a Selective Salpingogram? A Selective Salpingogram is a special X-ray procedure used to see if you have blocked fallopian tubes, (the tubes that
Information for parents Selective Salpingogram (SS) What is a Selective Salpingogram? A Selective Salpingogram is a special X-ray procedure used to see if you have blocked fallopian tubes, (the tubes that
female steri e sterilisation female sterilisation male and female sterilisation male sterilisation emale sterilisation female male sterilisati
 your guide to male and female sterilisation Helping you choose the method of contraception that is best for you female sterilisati le sterilisation male sterilisation e sterilisation female female ster
your guide to male and female sterilisation Helping you choose the method of contraception that is best for you female sterilisati le sterilisation male sterilisation e sterilisation female female ster
Figure 1b: Essure Insert (NOT TO SCALE)
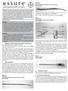 Figure 1b: Essure Insert Expanded configuration, detached from the delivery system (NOT TO SCALE) WARNING: Some patients implanted with the Essure System for Permanent Birth Control have experienced and/or
Figure 1b: Essure Insert Expanded configuration, detached from the delivery system (NOT TO SCALE) WARNING: Some patients implanted with the Essure System for Permanent Birth Control have experienced and/or
Figure 1b: Essure Insert (NOT TO SCALE) (NOT TO SCALE)
 Figure 1b: Essure Insert Expanded configuration, detached from the delivery system (NOT TO SCALE) WARNING: Some patients implanted with the Essure System for Permanent Birth Control have experienced and/or
Figure 1b: Essure Insert Expanded configuration, detached from the delivery system (NOT TO SCALE) WARNING: Some patients implanted with the Essure System for Permanent Birth Control have experienced and/or
ifuse implant system for treating chronic sacroiliac joint pain
 NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Medical technology guidance SCOPE ifuse implant system for treating chronic sacroiliac joint pain 1 Technology 1.1 Description of the technology The ifuse
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Medical technology guidance SCOPE ifuse implant system for treating chronic sacroiliac joint pain 1 Technology 1.1 Description of the technology The ifuse
Chapter 1. Chapter 2. Chapter 3
 Summary To perform IUI some conditions are required. This includes 1) a certain amount of progressively motile spermatozoa, 2) the presence of ovulation, 3) the presence of functional fallopian tubes,
Summary To perform IUI some conditions are required. This includes 1) a certain amount of progressively motile spermatozoa, 2) the presence of ovulation, 3) the presence of functional fallopian tubes,
International Federation of Fertility Societies. Global Standards of Infertility Care. Assessment of tubal patency. Recommendations for Practice
 International Federation of Fertility Societies Name Version number Author Global Standards of Infertility Care Standard 7. Assessment of tubal patency Recommendations for Practice Date of first release
International Federation of Fertility Societies Name Version number Author Global Standards of Infertility Care Standard 7. Assessment of tubal patency Recommendations for Practice Date of first release
Laparoscopy. Department of Gynaecology. Patient information
 Laparoscopy Department of Gynaecology Patient information What is is a a laparoscopy? A laparoscopy is an operation performed under general anaesthetic to help your gynaecologist make a diagnosis by looking
Laparoscopy Department of Gynaecology Patient information What is is a a laparoscopy? A laparoscopy is an operation performed under general anaesthetic to help your gynaecologist make a diagnosis by looking
Excessive menstrual blood loss
 Ian Chilcott Excessive menstrual blood loss >80mls - That interferes with physical, emotional, social and material quality of life 1 in 20 women aged 30 to 49 years consult their GP each year with menorrhagia
Ian Chilcott Excessive menstrual blood loss >80mls - That interferes with physical, emotional, social and material quality of life 1 in 20 women aged 30 to 49 years consult their GP each year with menorrhagia
CODING GUIDELINES FOR CONTRACEPTIVES. Effective June 1, 2017 Version 1.40
 CODING GUIDELINES FOR CONTRACEPTIVES Effective June 1, 2017 Version 1.40 TABLE OF CONTENTS ICD-10 CM Diagnosis Codes: Encounter for Contraception page 2 Coding for IUD Insertion and Removal Procedures
CODING GUIDELINES FOR CONTRACEPTIVES Effective June 1, 2017 Version 1.40 TABLE OF CONTENTS ICD-10 CM Diagnosis Codes: Encounter for Contraception page 2 Coding for IUD Insertion and Removal Procedures
Vascular malformations and their management
 Information for patients Vascular malformations and their management Introduction This leaflet tells you about the condition known as vascular malformation, including venous malformation, arteriovenous
Information for patients Vascular malformations and their management Introduction This leaflet tells you about the condition known as vascular malformation, including venous malformation, arteriovenous
Microinsert Nonincisional Hysteroscopic Sterilization
 Microinsert Nonincisional Hysteroscopic Sterilization Jay M. Cooper, MD, Charles S. Carignan, MD, Daniel Cher, MD, and John F. Kerin, MD, for the Selective Tubal Occlusion Procedure 2000 Investigators
Microinsert Nonincisional Hysteroscopic Sterilization Jay M. Cooper, MD, Charles S. Carignan, MD, Daniel Cher, MD, and John F. Kerin, MD, for the Selective Tubal Occlusion Procedure 2000 Investigators
Your visit to the Outpatient Hysteroscopy Clinic
 Your visit to the Outpatient Hysteroscopy Clinic Department of Gynaecology Patient Information What What is the is an Outpatient outpatient Cystoscopy hysteroscopy? Clinic? An outpatient hysteroscopy means
Your visit to the Outpatient Hysteroscopy Clinic Department of Gynaecology Patient Information What What is the is an Outpatient outpatient Cystoscopy hysteroscopy? Clinic? An outpatient hysteroscopy means
Commissioning Policy Individual Funding Request
 Commissioning Policy Individual Funding Request Female Sterilisation Prior Approval Policy Date Adopted: 6 th February 2017 Version: 1617.1 Individual Funding Request Team - A partnership between Bristol,
Commissioning Policy Individual Funding Request Female Sterilisation Prior Approval Policy Date Adopted: 6 th February 2017 Version: 1617.1 Individual Funding Request Team - A partnership between Bristol,
Surgery to Reduce the Risk of Ovarian Cancer. Information for Women at Increased Risk
 Surgery to Reduce the Risk of Ovarian Cancer Information for Women at Increased Risk Centre for Genetics Education NSW Health 2017 The Centre for Genetics Education NSW Health Level 5 2C Herbert St St
Surgery to Reduce the Risk of Ovarian Cancer Information for Women at Increased Risk Centre for Genetics Education NSW Health 2017 The Centre for Genetics Education NSW Health Level 5 2C Herbert St St
SIMPLE SAFE EFFECTIVE. Your Solution to Outpatient Ablation
 SIMPLE SAFE EFFECTIVE Your Solution to Outpatient Ablation WWW.THERMABLATE-EAS.COM SIMPLE Unique, fully automated design continually controls parameters of time, temperature and pressure to ensure consistent
SIMPLE SAFE EFFECTIVE Your Solution to Outpatient Ablation WWW.THERMABLATE-EAS.COM SIMPLE Unique, fully automated design continually controls parameters of time, temperature and pressure to ensure consistent
Dr John Short. Obstetrician and Gynaecologist Christchurch Women s Hospital Oxford Women's Health Christchurch
 Dr John Short Obstetrician and Gynaecologist Christchurch Women s Hospital Oxford Women's Health Christchurch 16:30-17:30 WS #125: Everything GPs Should Know About Gynaecologists 17:35-18:30 WS #135: Everything
Dr John Short Obstetrician and Gynaecologist Christchurch Women s Hospital Oxford Women's Health Christchurch 16:30-17:30 WS #125: Everything GPs Should Know About Gynaecologists 17:35-18:30 WS #135: Everything
Laparoscopy and Related Procedures
 Laparoscopy and Related Procedures Introduction The operation of laparoscopy is also commonly known as a keyhole operation. Laparoscopy can be used as a diagnostic procedure (i.e. an operation to find
Laparoscopy and Related Procedures Introduction The operation of laparoscopy is also commonly known as a keyhole operation. Laparoscopy can be used as a diagnostic procedure (i.e. an operation to find
Imaging evaluation of gynaecological devices
 Imaging evaluation of gynaecological devices Poster No.: C-0850 Congress: ECR 2013 Type: Educational Exhibit Authors: R. M. Lorente Ramos, J. Azpeitia Arman, P. Aparicio Rodríguez- 1 1 1 1 1 Miñón, F.
Imaging evaluation of gynaecological devices Poster No.: C-0850 Congress: ECR 2013 Type: Educational Exhibit Authors: R. M. Lorente Ramos, J. Azpeitia Arman, P. Aparicio Rodríguez- 1 1 1 1 1 Miñón, F.
INTERVENTIONAL PROCEDURES PROGRAMME
 NATIONAL INSTITUTE FOR CLINICAL EXCELLENCE INTERVENTIONAL PROCEDURES PROGRAMME Interventional procedure overview of laparoscopic laser myomectomy Introduction This overview has been prepared to assist
NATIONAL INSTITUTE FOR CLINICAL EXCELLENCE INTERVENTIONAL PROCEDURES PROGRAMME Interventional procedure overview of laparoscopic laser myomectomy Introduction This overview has been prepared to assist
Advanced 3D Ultrasound Incorporating Fly Thru Virtual Imaging Promotes the Concept of Ultrasound Hysteroscopy
 Advanced 3D Ultrasound Incorporating Fly Thru Virtual Imaging Promotes the Concept of Ultrasound Hysteroscopy Bill Smith Clinical Diagnostics Services, London, UK Introduction Conventional hysteroscopy
Advanced 3D Ultrasound Incorporating Fly Thru Virtual Imaging Promotes the Concept of Ultrasound Hysteroscopy Bill Smith Clinical Diagnostics Services, London, UK Introduction Conventional hysteroscopy
Pelvic Pain. What you need to know. 139 Dumaresq Street Campbelltown Phone Fax
 Pelvic Pain What you need to know 139 Dumaresq Street Campbelltown Phone 4628 5292 Fax 4628 0349 www.nureva.com.au September 2015 PELVIC PAIN This is a common problem and most women experience some form
Pelvic Pain What you need to know 139 Dumaresq Street Campbelltown Phone 4628 5292 Fax 4628 0349 www.nureva.com.au September 2015 PELVIC PAIN This is a common problem and most women experience some form
Investigating HMB- an evidence based approach
 BSGE Meeting: Contemporary management of heavy menstrual bleeding (HMB) in primary and secondary care: (7 th December 2018, RCOG) Investigating HMB- an evidence based approach T. Justin Clark MB ChB, MD(Hons),
BSGE Meeting: Contemporary management of heavy menstrual bleeding (HMB) in primary and secondary care: (7 th December 2018, RCOG) Investigating HMB- an evidence based approach T. Justin Clark MB ChB, MD(Hons),
KUALA LUMPUR SUMMARY MATERIALS AND METHODS INTRODUCTION
 Med. J. Malaysia Vol. 37 No. 3 September 1982. WITH HULKA CLIPS AT THE UNIVERSITY KUALA LUMPUR ASARI ABDUL RAHMAN V. SIVANESARATNAM A. ADLAN NURUDDIN SUMMARY An analysis of 86 patients sterilized laparoscopically
Med. J. Malaysia Vol. 37 No. 3 September 1982. WITH HULKA CLIPS AT THE UNIVERSITY KUALA LUMPUR ASARI ABDUL RAHMAN V. SIVANESARATNAM A. ADLAN NURUDDIN SUMMARY An analysis of 86 patients sterilized laparoscopically
Evaluation of the Infertile Couple
 Overview and Definition Infertility is defined as the inability of a couple to fall pregnant after one year of unprotected intercourse. Infertility is a very common condition as in any given year about
Overview and Definition Infertility is defined as the inability of a couple to fall pregnant after one year of unprotected intercourse. Infertility is a very common condition as in any given year about
WOMEN & INFANTS HOSPITAL Providence, RI CONSENT FOR IVF WITH EMBRYO TRANSFER
 *40639* 40639 WOMEN & INFANTS HOSPITAL Providence, RI 02905 CONSENT FOR IVF WITH EMBRYO TRANSFER I have requested treatment by the physicians and (Print Patient s name) staff of the Women & Infants Fertility
*40639* 40639 WOMEN & INFANTS HOSPITAL Providence, RI 02905 CONSENT FOR IVF WITH EMBRYO TRANSFER I have requested treatment by the physicians and (Print Patient s name) staff of the Women & Infants Fertility
Laparoscopic (keyhole) sterilisation
 Laparoscopic (keyhole) sterilisation Gynaecology Department Patient Information Leaflet Introduction This leaflet is for women who are considering sterilisation to prevent pregnancy. It explains what the
Laparoscopic (keyhole) sterilisation Gynaecology Department Patient Information Leaflet Introduction This leaflet is for women who are considering sterilisation to prevent pregnancy. It explains what the
Surgical treatment of urinary stress incontinence with tension free vaginal tape
 Surgical treatment of urinary stress incontinence with tension free vaginal tape Gynaecology department 01935 384 385 yeovilhospital.nhs.uk Many surgical operations are available for the treatment of
Surgical treatment of urinary stress incontinence with tension free vaginal tape Gynaecology department 01935 384 385 yeovilhospital.nhs.uk Many surgical operations are available for the treatment of
Compare and contrast laparoscopic surgery verses methotrexate in a woman with the diagnosis of ectopic pregnancy
 Compare and contrast laparoscopic surgery verses methotrexate in a woman with the diagnosis of ectopic pregnancy Ectopic pregnancy is defined as the implantation of a conceptus outside of the uterine cavity.
Compare and contrast laparoscopic surgery verses methotrexate in a woman with the diagnosis of ectopic pregnancy Ectopic pregnancy is defined as the implantation of a conceptus outside of the uterine cavity.
Open Radical Removal of the Kidney
 Who can I contact if I have a problem when I get home? If you experience any problems related to your surgery or admission once you have been discharged home. Please feel free to contact 4A, 4B or 4C ward
Who can I contact if I have a problem when I get home? If you experience any problems related to your surgery or admission once you have been discharged home. Please feel free to contact 4A, 4B or 4C ward
FACTSHEET FERTILITY INVESTIGATIONS
 FACTSHEET FERTILITY INVESTIGATIONS Most people nowadays know enough about contraception to expect a pregnancy to occur reasonably soon after contraceptive precautions are stopped. If your own fertility
FACTSHEET FERTILITY INVESTIGATIONS Most people nowadays know enough about contraception to expect a pregnancy to occur reasonably soon after contraceptive precautions are stopped. If your own fertility
Uterine artery embolisation for treating adenomyosis
 Uterine artery embolisation for treating Issued: December 2013 guidance.nice.org.uk/ipg NICE has accredited the process used by the NICE Interventional Procedures Programme to produce interventional procedures
Uterine artery embolisation for treating Issued: December 2013 guidance.nice.org.uk/ipg NICE has accredited the process used by the NICE Interventional Procedures Programme to produce interventional procedures
Male and female sterilization
 Male and female sterilization Catriona Melville Alison Bigrigg Abstract Male and female sterilization is used in many countries worldwide as a permanent method of contraception. Failure rates for female
Male and female sterilization Catriona Melville Alison Bigrigg Abstract Male and female sterilization is used in many countries worldwide as a permanent method of contraception. Failure rates for female
Moneli Golara Consultant Obstetrician and Gynaecologist Barnet Hospital Royal Free NHS Trust
 Moneli Golara Consultant Obstetrician and Gynaecologist Barnet Hospital Royal Free NHS Trust Endometriosis one of the most common conditions requiring treatment Growth of endometrial like tissue outside
Moneli Golara Consultant Obstetrician and Gynaecologist Barnet Hospital Royal Free NHS Trust Endometriosis one of the most common conditions requiring treatment Growth of endometrial like tissue outside
levonorgestrel 13.5mg intrauterine delivery system (Jaydess ) SMC No. (1036/15) Bayer
 levonorgestrel 13.5mg intrauterine delivery system (Jaydess ) SMC No. (1036/15) Bayer 6 March 2015 The Scottish Medicines Consortium (SMC) has completed its assessment of the above product and advises
levonorgestrel 13.5mg intrauterine delivery system (Jaydess ) SMC No. (1036/15) Bayer 6 March 2015 The Scottish Medicines Consortium (SMC) has completed its assessment of the above product and advises
IPAC date: May of 13
 National Institute for Health and Care Excellence IP685/3 Transcatheter aortic valve implantation (transcatheter aortic valve replacement) for aortic stenosis Consultation Comments table IPAC date: May
National Institute for Health and Care Excellence IP685/3 Transcatheter aortic valve implantation (transcatheter aortic valve replacement) for aortic stenosis Consultation Comments table IPAC date: May
Instruction for the patient
 Instruction for the patient Your situation You are a 38-year-old, woman who is pregnant with her third child You and your partner agree that with this child your family is complete In the past, you used
Instruction for the patient Your situation You are a 38-year-old, woman who is pregnant with her third child You and your partner agree that with this child your family is complete In the past, you used
Example CLINICAL GUIDELINES for Postpartum IUD insertion
 Example CLINICAL GUIDELINES for Postpartum IUD insertion Postpartum Intrauterine Device Insertion 1.0 Indications: 1.1 Insertion of an intrauterine device (IUD) for long-acting reversible contraception
Example CLINICAL GUIDELINES for Postpartum IUD insertion Postpartum Intrauterine Device Insertion 1.0 Indications: 1.1 Insertion of an intrauterine device (IUD) for long-acting reversible contraception
Information leaflet on. Laparoscopic Treatment of Endometriosis
 Information leaflet on Laparoscopic Treatment of Endometriosis 1 What is endometriosis? Endometriosis is a condition, which affects many women. It is defined as the presence of endometrial tissue outside
Information leaflet on Laparoscopic Treatment of Endometriosis 1 What is endometriosis? Endometriosis is a condition, which affects many women. It is defined as the presence of endometrial tissue outside
Laparoscopy. Patient information leaflet
 Laparoscopy Division of Adhesions Dye Test Treatment of Endometriosis Cystectomy Oophorectomy, Salpingectomy Salpingostomy Sterilisation Gynaecology Department Patient information leaflet What is a Laparoscopy?
Laparoscopy Division of Adhesions Dye Test Treatment of Endometriosis Cystectomy Oophorectomy, Salpingectomy Salpingostomy Sterilisation Gynaecology Department Patient information leaflet What is a Laparoscopy?
INTERVENTIONAL PROCEDURES PROGRAMME
 NATIONAL INSTITUTE FOR CLINICAL EXCELLENCE INTERVENTIONAL PROCEDURES PROGRAMME Interventional procedure overview of balloon thermal endometrial ablation (Cavaterm) Introduction This overview has been prepared
NATIONAL INSTITUTE FOR CLINICAL EXCELLENCE INTERVENTIONAL PROCEDURES PROGRAMME Interventional procedure overview of balloon thermal endometrial ablation (Cavaterm) Introduction This overview has been prepared
National Institute for Health and Care Excellence
 National Institute for Health and Care Excellence IP956 Insertion of an annular disc implant at lumbar discectomy Consultation Comments table IPAC date: Friday 12 th September 2014 1 of 10 1 Consultee
National Institute for Health and Care Excellence IP956 Insertion of an annular disc implant at lumbar discectomy Consultation Comments table IPAC date: Friday 12 th September 2014 1 of 10 1 Consultee
Radical removal of the kidney (radical nephrectomy): procedure-specific information
 PATIENT INFORMATION Radical removal of the kidney (radical nephrectomy): procedure-specific information What is the evidence base for this information? This leaflet includes advice from consensus panels,
PATIENT INFORMATION Radical removal of the kidney (radical nephrectomy): procedure-specific information What is the evidence base for this information? This leaflet includes advice from consensus panels,
Hysteroscopy. Department of Gynaecology. Patient information
 Hysteroscopy Department of Gynaecology Patient information What is is a hysteroscopy? a Hysteroscopy? A hysteroscopy is a procedure during which the inside of the uterus (womb) is examined using a thin
Hysteroscopy Department of Gynaecology Patient information What is is a hysteroscopy? a Hysteroscopy? A hysteroscopy is a procedure during which the inside of the uterus (womb) is examined using a thin
Birth Control- an Overview. Keith Merritt, MD. Remember, all methods of birth control are safer and have fewer side effects than pregnancy
 Birth Control- an Overview Keith Merritt, MD Basics Remember, all methods of birth control are safer and have fewer side effects than pregnancy Even with perfect use, each method of birth control has a
Birth Control- an Overview Keith Merritt, MD Basics Remember, all methods of birth control are safer and have fewer side effects than pregnancy Even with perfect use, each method of birth control has a
Laparoscopic Radical Removal of the Kidney +/- Ureter
 Who can I contact if I have a problem when I get home? If you experience any problems related to your surgery or admission once you have been discharged home. Please feel free to contact 4A, 4B or 4C ward
Who can I contact if I have a problem when I get home? If you experience any problems related to your surgery or admission once you have been discharged home. Please feel free to contact 4A, 4B or 4C ward
 INTRAUTERINE DEVICE = IUD INTRAUTERINE DEVICE = IUD CONGENITAL DISORDERS Pyometra = pyometrea is a uterine infection, it is accumulation of purulent material in the uterine cavity. Ultrasound is usually
INTRAUTERINE DEVICE = IUD INTRAUTERINE DEVICE = IUD CONGENITAL DISORDERS Pyometra = pyometrea is a uterine infection, it is accumulation of purulent material in the uterine cavity. Ultrasound is usually
Guidelines for performing gynaecological endoscopic procedures
 Guidelines for performing gynaecological endoscopic procedures This statement has been developed by the Women s Health Committee. It has been reviewed by the Endoscopic Surgery Advisory Committee (RANZCOG/AGES)
Guidelines for performing gynaecological endoscopic procedures This statement has been developed by the Women s Health Committee. It has been reviewed by the Endoscopic Surgery Advisory Committee (RANZCOG/AGES)
Laparoscopy. Patient Information. Womens Health
 Laparoscopy Patient Information Womens Health What is a Laparoscopy Laparoscopy is a minimally invasive or key hole surgical procedure performed under general anaesthetic. It enables the surgeon to look
Laparoscopy Patient Information Womens Health What is a Laparoscopy Laparoscopy is a minimally invasive or key hole surgical procedure performed under general anaesthetic. It enables the surgeon to look
Partial Removal of the Kidney
 Who can I contact if I have a problem when I get home? If you experience any problems related to your surgery or admission once you have been discharged home. Please feel free to contact Ward 4A, 4B or
Who can I contact if I have a problem when I get home? If you experience any problems related to your surgery or admission once you have been discharged home. Please feel free to contact Ward 4A, 4B or
Cover Page. The handle holds various files of this Leiden University dissertation.
 Cover Page The handle http://hdl.handle.net/1887/57234 holds various files of this Leiden University dissertation. Author: Chapelle, C.F. la Title: Evidence-based implementation of minimally invasive surgery
Cover Page The handle http://hdl.handle.net/1887/57234 holds various files of this Leiden University dissertation. Author: Chapelle, C.F. la Title: Evidence-based implementation of minimally invasive surgery
Women s & Children s Directorate The TVT Operation - a guide for patients
 Women s & Children s Directorate The TVT Operation - a guide for patients This leaflet was written for women who are considering having a TVT operation. If you have any questions that aren't answered by
Women s & Children s Directorate The TVT Operation - a guide for patients This leaflet was written for women who are considering having a TVT operation. If you have any questions that aren't answered by
100% Highly effective No cost No side effects
 effective? Advantages Disadvantages How do I get Cost Abstinence For some it can mean no sexual contact. For others it is no sexual intercourse or vaginal penetration. A permanent surgical procedure available
effective? Advantages Disadvantages How do I get Cost Abstinence For some it can mean no sexual contact. For others it is no sexual intercourse or vaginal penetration. A permanent surgical procedure available
Tension-free Vaginal Tape (TVT)
 Page 1 of 7 Tension-free Vaginal Tape (TVT) Introduction This leaflet will provide you with basic information about the Tension--free Vaginal Tape (TVT) procedure. What is a TVT? TVT is an operation to
Page 1 of 7 Tension-free Vaginal Tape (TVT) Introduction This leaflet will provide you with basic information about the Tension--free Vaginal Tape (TVT) procedure. What is a TVT? TVT is an operation to
X-Plain Ovarian Cancer Reference Summary
 X-Plain Ovarian Cancer Reference Summary Introduction Ovarian cancer is fairly rare. Ovarian cancer usually occurs in women who are over 50 years old and it may sometimes be hereditary. This reference
X-Plain Ovarian Cancer Reference Summary Introduction Ovarian cancer is fairly rare. Ovarian cancer usually occurs in women who are over 50 years old and it may sometimes be hereditary. This reference
WOMEN & INFANTS HOSPITAL Providence, RI CONSENT FOR IN VITRO FERTILIZATION USING A GESTATIONAL CARRIER (PATIENT/INTENDED PARENTS) 1.
 *40675* 40675 MR-838 (9-2017) WOMEN & INFANTS HOSPITAL Providence, RI 02905 CONSENT FOR IN VITRO FERTILIZATION USING A GESTATIONAL CARRIER (PATIENT/INTENDED PARENTS) 1. I, and (Print Patient s name) (Print
*40675* 40675 MR-838 (9-2017) WOMEN & INFANTS HOSPITAL Providence, RI 02905 CONSENT FOR IN VITRO FERTILIZATION USING A GESTATIONAL CARRIER (PATIENT/INTENDED PARENTS) 1. I, and (Print Patient s name) (Print
Diagnostic laparoscopy in primary and secondary infertility
 Diagnostic laparoscopy in primary and secondary infertility Al-Sakkkal Ghada Saddallah C.A.B.O.G. Department of Obs. And Gyn., Hawler Medical University ABSTRACT Objective: To compare the diagonstic effficacy
Diagnostic laparoscopy in primary and secondary infertility Al-Sakkkal Ghada Saddallah C.A.B.O.G. Department of Obs. And Gyn., Hawler Medical University ABSTRACT Objective: To compare the diagonstic effficacy
Stop Coping. Start Living. Talk to your doctor about pelvic organ prolapse and sacrocolpopexy
 Stop Coping. Start Living Talk to your doctor about pelvic organ prolapse and sacrocolpopexy Did you know? One in three women will suffer from a pelvic health condition in her lifetime. Four of the most
Stop Coping. Start Living Talk to your doctor about pelvic organ prolapse and sacrocolpopexy Did you know? One in three women will suffer from a pelvic health condition in her lifetime. Four of the most
Family Planning Eligibility Program
 INDIANA HEALTH COVERAGE PROGRAMS PROVIDER REFERENCE M ODULE Family Planning Eligibility Program L I B R A R Y R E F E R E N C E N U M B E R : P R O M O D 0 0 0 5 3 P U B L I S H E D : N O V E M B E R 2
INDIANA HEALTH COVERAGE PROGRAMS PROVIDER REFERENCE M ODULE Family Planning Eligibility Program L I B R A R Y R E F E R E N C E N U M B E R : P R O M O D 0 0 0 5 3 P U B L I S H E D : N O V E M B E R 2
FDA-Approved Patient Labeling Patient Information Mirena (mur-ā-nah) (levonorgestrel-releasing intrauterine system)
 FDA-Approved Patient Labeling Patient Information Mirena (mur-ā-nah) (levonorgestrel-releasing intrauterine system) Mirena does not protect against HIV infection (AIDS) and other sexually transmitted infections
FDA-Approved Patient Labeling Patient Information Mirena (mur-ā-nah) (levonorgestrel-releasing intrauterine system) Mirena does not protect against HIV infection (AIDS) and other sexually transmitted infections
Coding for the Contraceptive Implant and IUDs
 LARC Quick Coding Guide 2018 UPDATE Coding for the Contraceptive Implant and IUDs CORRECT CODING can result in more appropriate compensation for services and devices. To help practices receive appropriate
LARC Quick Coding Guide 2018 UPDATE Coding for the Contraceptive Implant and IUDs CORRECT CODING can result in more appropriate compensation for services and devices. To help practices receive appropriate
Ultrasound-enhanced, catheter-directed thrombolysis for pulmonary embolism
 NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Interventional procedure consultation document Ultrasound-enhanced, catheter-directed thrombolysis for pulmonary embolism A pulmonary embolism (PE) is
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Interventional procedure consultation document Ultrasound-enhanced, catheter-directed thrombolysis for pulmonary embolism A pulmonary embolism (PE) is
