BIOLOGY OF REPRODUCTION 56, (1997) Warren B. Nothnick, 3 ' 4 Paul Soloway, 5 and Thomas E. Curry, Jr. 2, 4
|
|
|
- Lucas Flynn
- 6 years ago
- Views:
Transcription
1 BIOLOGY OF REPRODUCTION 56, (1997) Assessment of the Role of Tissue Inhibitor of Metalloproteinase-1 (TIMP-1) during the Periovulatory Period in Female Mice Lacking a Functional TIMP-1 Gene' Warren B. Nothnick, 3 ' 4 Paul Soloway, 5 and Thomas E. Curry, Jr. 2, 4 Departments of Obstetrics and Gynecology, 4 University of Kentucky, Lexington, Kentucky Department of Molecular and Cellular Biology ș Roswell Cancer Institute, Buffalo, New York ABSTRACT Tissue inhibitor of metalloproteinase (TIMP)-1 is a multifunctional peptide that has been implicated in the ovulatory process. To assess the function of TIMP-1 during the periovulatory period in vivo, mice incapable of expressing the TIMP-1 gene product were utilized. Twenty-three-day-old TIMP-1-deficient (n = 59) and wild-type (n = 61) female mice were injected with 5 IU ecg, followed 48 h later by an ovulation-inducing dose of hcg (5 IU). Animals were killed at the time of hcg injection (0-h hcg), at 12 h (12-h hcg), or at 24 h post-hcg (24-h hcg) administration. Serum was collected for the assessment of estradiol-1 73 (0-h hcg groups) or progesterone content (12- and 24-h hcg groups), while ovaries were removed for either histological preparation or Northern analysis of TIMP-1, TIMP-2, and TIMP-3. The number of healthy and atretic follicles was determined in the 0-h hcg groups, as was the number of oocytes released in the 24-h hcg group. TIMP-1-deficient females in the 0-h hcg group showed reduced levels of ovarian TIMP-2 (0.29-fold decrease, p < 0.05) and TIMP-3 (3.0-fold decrease, p < 0.05) expression compared to wild-type counterparts. No significant difference was detected between genotypes in the 0-h hcg group for number of healthy or atretic follicles or for serum estradiol-171 concentrations. Additionally, no significant differences were detected between genotypes in the 12- and 24-h hcg groups for serum progesterone concentrations, ovarian TIMP-2 and TIMP-3 expression, or number of oocytes released (24-h hcg group). To assess the effect of TIMP-1 on steroidogenesis in vitro, granulosa cells were obtained from 23-day-old, ecg-primed TIMP-1-deficient and wild-type females. Addition of recombinant human TIMP-1 significantly increased conditioned media estradiol-17p3 concentrations in cell cultures from both mutant (1.32-fold over controls; p = 0.02; n = 4) and wild-type females (1.16-fold over controls; p = 0.04; n = 3). It is concluded from this study that TIMP-1 may modulate ovarian TIMP-2 and TIMP-3 mrna expression during folliculogenesis. In addition, TIMP-1 exhibits steroidogenic activity in vitro, but no evidence was found for regulation of steroidogenesis in vivo. INTRODUCTION The ovulatory process involves a complex cascade of biochemical events that is set in motion by the LH surge. LH stimulates the proteolytic breakdown of the apical follicular wall during the periovulatory period, leading to follicular rupture [1, 2]. Specifically, LH stimulates the matrix metalloproteinases, collagenase [3], proteoglycanase, and gelatinase [4], all of which are believed to play a pivotal role in the tissue remodeling associated with apical follicular degradation and oocyte release. In addition to stimu- Accepted December 13, Received October 16, 'Supported by NIH HD23195 to T.E.C. Jr. and EY11279 to P.S. 2Correspondence. FAX: (606) ; tecurry@pop.uky.edu 3 Current address: Departments of Obstetrics and Gynecology and Physiology, University of Kansas Medical Center, Kansas City, KS lating matrix metalloproteinase activity during the periovulatory and ovulatory period, LH also stimulates expression and activity of the matrix metalloproteinase inhibitor, known as tissue inhibitor of metalloproteinase (TIMP)-1, in several species [5-9]. In fact, LH/hCG stimulates concurrent expression of not only ovarian TIMP-1 but also the metalloproteinase collagenase [3, 6]. This simultaneous expression of enzyme and inhibitor has been demonstrated in the ovary and other systems [10, 11] and is believed to be a mechanism that allows focal control of the tissue-degrading enzyme(s). In addition to regulating matrix metalloproteinase activity, TIMP-1 has been shown to exhibit growth-promoting activity in a variety of cell types or cell lines, e.g., articular chondrocytes, skin epithelial cells, aortic smooth muscle cells, and human skin fibroblasts [12, 13]. TIMP-1 shares sequence homology with erythroid potentiating factor [14] and stimulates proliferation of erythroid progenitor cells [15], thus raising the possibility that this protein may also play a role in erythropoiesis and neovascularization. Additionally, TIMP- 1 has been shown to stimulate collagenase secretion in human skin fibroblasts [16], suggesting that TIMP-1 may regulate metalloproteinase production. Finally, a TIMP-l-like protein has been reported to stimulate steroid production by Leydig cells as well as granulosa cells in vitro [17], and TIMP-1 has been reported to share a 124- base-pair sequence homology with the steroidogenic acute regulatory protein (StAR; [18]). TIMP-1 is a highly abundant ovarian mrna [19] encoding a multifunctional protein potentially capable of modulating the secretion and activity of proteolytic enzymes, neovascularization, growth, and steroidogenesis of the newly developing corpus luteum. In order to directly determine the importance of TIMP-1 in ovarian function in vivo, TIMP-l-deficient mice were utilized in the present study to examine folliculogenesis, steroidogenesis, and ovulation. MATERIALS AND METHODS Animals TIMP-l-deficient animals were generated by homologous recombination of a neo-containing gene-targeting vector in mouse embryonic stem cells [20]. Transmission of the mutant allele and the genotype of mice were determined by polymerase chain reaction analysis of the neo sequences in genomic tail DNA. TIMP-1 deficiency was confirmed at the transcript and protein level by Northern analysis and matrix metalloproteinase inhibitor assays, respectively. Animals were housed and maintained under the supervision of a licensed veterinarian. All animal procedures for these experiments were approved by the University of Kentucky Institutional Animal Care and Use Committee. Mice were maintained on a 14L:10OD cycle. Pups were weaned at 21 days of age, at which time all pups were sexed. Female pups were ear notched for identification purposes, and
2 1182 NOTHNICK ET AL. a 1-cm segment of the tail (not including the keratinized tip) was removed and prepared for isolation of genomic DNA. Treatment Groups, Tissue Collection, and Assessment of Ovulation Homozygous wild-type (+/+) and TIMP-1-deficient (-/ -) females (i.e., mutant animals) were injected at 23 days of age with ecg (5 IU, s.c.) to stimulate follicular growth, followed 48 h later by s.c. administration of hcg (5 IU) to induce ovulation. This regime typically results in ovulation sites per ovary [21]. All animals were then killed by decapitation at 0 h (time of hcg administration), 12 h, or 24 h after hcg administration. Controls consisted of wild-type and TIMP-l-deficient mice receiving vehicle injections at the time of ecg and hcg injections, as well as animals receiving no injections that were killed at the end of the treatment period (age-matched controls). Peripheral blood was collected for assessment of serum estrogen and progesterone concentrations, and ovarian weights were recorded. One ovary from each animal was placed into neutral buffered formalin and prepared for histological assessment as described below, while the other ovary from each animal was snap frozen in liquid nitrogen and stored at -75 C until analyzed for TIMP mrna content. Both TIMP-l-deficient and wild-type females in the 24-h posthcg treatment group (ovulation occurring approximately h post-hcg) were examined for the number of oocytes released; to this end the oviducts were flushed with PBS, the cumulus mass was placed on a glass slide with a coverslip, and the number of oocytes released from both ovaries was counted. Metalloproteinase Inhibitor Assay The assay is based on the inhibition of a uterine neutral metalloproteinase (matrilysin) as described previously [5]. Matrilysin degrades the colorimetric substrate, Azocoll. In the presence of endogenous inhibitors, matrilysin activity is inhibited and there is a decrease in the digestion of the Azocoll substrate, which is quantitated spectrophotometrically. This assay allows for quantitation of both serumborne (e.g., macroglobulin-type inhibitors) and tissue-borne inhibitors of metalloproteinases. To assess only TIMP activity, aliquots of serum from each animal (n = 2 per genotype from the 24-h hcg groups) were treated with 200 mm methylamine hydrochloride. This treatment inactivates alpha macroglobulintype inhibitors [5], and accordingly the inhibition of substrate digestion is attributable to TIMP activity. Samples were treated for 30 min at room temperature, and the samples were then dialyzed overnight at 4C against 2 L of assay buffer with three buffer changes. A volume of matrilysin sufficient to yield a change in absorbance of 0.2 (-20 pl) was mixed with the methylamine-treated serum samples (200 pll) and brought to a total volume of 400 [LI with assay buffer (50 mm Tris-HCl, 10 mm CaC12, 0.2 M NaCl, 0.05% sodium azide, and 0.167% Brij35 [polyoxyethylene 23 lauryl ether, a detergent], ph 7.5). This mixture was preincubated for 1 h at 37 C to allow for binding of matrilysin and endogenous inhibitors present in the serum. Next, 1 mg of Azocoll suspended in 850 pl1 assay buffer was added to each sample and incubated for 18 h at 37 C in a reciprocating water bath. The inhibition of matrilysin activity from the endogenous inhibitors was quantitated on a Beckman DU-64 spectrophotometer (Beckman Instruments, Fullerton, CA) at 520 nm and expressed as inhibitor units; an inhibitor unit was calculated as the percentage inhibition of total matrilysin activity per milligram of total protein in the sample. Total RNA Isolation and Northern Blot Analysis RNA was isolated by the method of Chomczynski and Sacchi [22] using an acid guanidinium thiocyanate-phenolchloroform extraction procedure as modified by Nothnick and colleagues [21]. Ovaries were pooled from 2 to 3 animals to yield a total weight of at least 15 mg. Total RNA was isolated from ovarian tissue by homogenization in 2 ml of denaturing solution (4 M guanidinium thiocyanate, 25 mm sodium citrate [ph 7.0], 0.5% sarcosyl, and 0.1 M 3-mercaptoethanol) followed by extraction with phenol: chloroform:isoamyl alcohol and reprecipitation in ethanol. The resulting pellet was washed in cold diethylpyrocarbonate (DEPC)-treated 75% ethanol, vacuum dried, resuspended in of DEPC-treated water, and stored at -75 C until analyzed by Northern blotting. Northern analysis was performed using up to 25 tpg of total RNA per lane. Samples were electrophoresed through a 1% agarose gel containing 2.2 M formaldehyde and were transferred to a nylon membrane (Nytran; Schleicher and Schuell, Keene, NH) as recommended by the manufacturer. The respective cdna probes for mouse TIMP-1, TIMP-2, and TIMP-3 were excised from their plasmid with the appropriate endonucleases (TIMP-1, BamHIIHindIII; TIMP-2, EcoRIIHindIII; TIMP-3, Pst I) and labeled using a random primer kit (Gibco BRL, Gaithersburg, MD). Each probe was labeled to a specific activity of approximately dpm/lg of DNA using [ot- 3 2 P]dCTP (NEN-DuPont, Boston, MA). Filters were hybridized overnight according to the recommendations of the manufacturer. After probing for the various TIMPs, blots were hybridized for the transcript of the constitutively expressed 18S ribosomal protein using a rat 18S cdna probe. All TIMP Northern data were then normalized to the relative expression of the 18S transcript. Ovarian Histology and Tissue Morphometrics To prepare ovaries for histological assessment, tissues were fixed in toto in 10% neutral buffered formalin. Tissues were then embedded in paraffin, serially sectioned at 7 pim, and counterstained with hematoxylin and eosin as described by Luna [23]. On every tenth section (i.e., 70-[Lm intervals), follicles with an oocyte containing a nucleus (0-h and 12-h hcg groups) or corpora lutea (24-h hcg groups) were measured by using the mean of two right angle determinations through the follicle/corpus luteum. Follicles were grouped according to size (i.e., VIm, ,um, or > 400 jim) and classified as healthy or atretic based upon the presence of 3 pyknotic nuclei per section. Granulosa Cell Isolation and Culture Granulosa cells were isolated from preovulatory follicles and cultured as routinely performed in our laboratory [5, 9] with the exception that 23-day-old, ecg-primed wildtype and TIMP- -deficient mice were used. Briefly, ovaries were cleaned of adnexa (connective tissue, fat, etc.), gap junctions were disrupted, and granulosa cells were expressed by puncturing the ovaries with a 26-gauge needle. Granulosa cells were pooled from a total of 8-12 ovaries (n = 4-6 animals per genotype). Granulosa cells were pelleted by centrifugation (5 min, 600 x g) and placed into
3 OVARIAN TIMP-1 FUNCTION 1183 culture wells containing 0.5 ml Ham's F-12/Dulbecco Minimum Eagle medium containing 25 mm Hepes, 1% (w:v) BSA, and gentamicin (0.05 mg/ml) at a concentration of 1 x 106 viable cells/ml of medium. Cell viability was assessed using trypan blue exclusion before allocation of the cells and again at the end of the culture period. Cells were cultured for 24 h in the presence of 1 Rig/ml recombinant human TIMP-1 (a generous gift from Dr. Paul Cannon, Syntex Corp., Palo Alto, CA) or vehicle (20 mm Tris-HCl [ph 7.5], 100 mm NaCl, 0.01% [w:v] Brij35) under 5.0% CO 2. After incubation, granulosa cells were pelleted by centrifugation (5 min, 600 x g), and steroid content of the conditioned medium was determined by RIA as described below. This experiment was performed a total of three separate times using wild-type mice and four separate times using TIMP-1-deficient mice (n = 3 and 4 for wild-type and TIMP-1-deficient cultures, respectively). Serum and Conditioned Media Steroid Quantitation Because of limited sample volume, estrogen concentrations were analyzed in serum from animals in the 0-h group and matching controls, while progesterone concentrations were analyzed in the 12- and 24-h post-hcg groups and their matching controls. Steroid concentrations were determined by a solid-phase 125I RIA (Diagnostic Products Corp., Los Angeles, CA) previously validated in our laboratory [24]. Samples were analyzed singly or in duplicate (50 Rl) when sufficient serum was available for the latter. Conditioned media steroid content was determined using 100 l of medium, and samples were assayed in duplicate. The detection limits of the assays were 4.0 pg/ml and 0.05 ng/ml for estrogen and progesterone, respectively. The inter- and intraassay coefficients of variation for the estrogen assay were 8.5% and 5.3%, respectively, and for the progesterone assays they were 7.9% and 4.6%, respectively. Recombinant human TIMP-1 showed cross-reactivity with neither the estrogen nor progesterone assays. Statistics Data were analyzed by planned comparisons using twosample t-tests for between-genotype analysis. Data among time points within genotypes were analyzed by one-way ANOVA. When a significant F value was obtained, post hoc analysis was made using the Student-Newman-Keuls procedure [25]. RESULTS All TIMP-1-deficient mice contained the 500-base-pair replacement vector (neo insert) as determined by polymerase chain reaction amplification of genomic DNA. Detection of the neo insert coincided with the inability to express the TIMP-1 transcript, as ovarian tissue from mutant (i.e., -/-) females, regardless of treatment group, failed to demonstrate a hybridization signal for the TIMP-1 transcript (Fig. 1A). TIMP-1 transcript was also not detectable in lung and peritoneum preparations from 23-day-old TIMP-1-deficient animals, confirming that this transcript was not specifically disrupted in the ovary (data not shown). In contrast, TIMP-1 was detected in the lung and peritoneum of wild-type mice (data not shown) as well as in the ovaries of ecg+hcg-treated wild-type animals (Fig. 1A). In fact, TIMP- 1 could be detected in hcg-stimulated (24 h postadministration) ovaries from wild-type females using as little as 0.5 jig of total RNA (Fig. B), but the transcript could FIG. 1. A) Northern analysis of TIMP-1 in ovarian tissue from TIMP-1- deficient (-/-) and wild-type (+/+) females (top transcript). Total RNA was isolated from ovarian tissue, and Northern analysis was performed using 7.5 Ig of total RNA per lane as described in Materials and Methods. A single transcript of approximately 900 base pairs, consistent with TIMP-1, was detected in ovarian tissue from all gonadotropin-stimulated wild-type females but not in ovaries from TIMP-1-deficient females. Approximate 1.9-kilobase transcript, consistent with the 1 8S ribosomal protein transcript, indicates equality of lane loading and capillary transfer of RNA to membranes. B) Northern analysis of TIMP-1 in ovarian tissue with loading of increasing quantities of total RNA. Total RNA was loaded onto an agarose-formaldehyde gel as described in Materials and Methods using 0.5, 2.5, 5.0, or 25.0 pig of RNA from ovaries of wild-type and TIMP-1- deficient females from the 24-h hcg treatment group. TIMP-1 transcript could be detected using as little as 0.5 I.g of RNA from the wild-type ovaries, but no transcript was detected in the TIMP-1-deficient ovaries using as much as 25 VIg of total RNA. not be detected in hcg-stimulated (24 h post-hcg) ovaries of TIMP-l-deficient females using up to 25 jig of total RNA (Fig. 1B). Lastly, the lack of TIMP-1 transcript was also associated with the inability to display functional TIMP-1 activity. Specifically, serum-derived TIMP-1 activity was detected in serum obtained from wild-type mice from the 24-h hcg treatment group but not from TIMP-1- deficient females in this same treatment group (Fig. 2). Basal levels of TIMP activity in the mutants were attributed to TIMP-2 activity, as TIMP-1-deficient females did express TIMP-2 mrna. To examine whether disruption of the TIMP-1 gene product resulted in a compensatory increase in TIMP-2 or TIMP-3 mrna levels, Northern analysis of total ovarian RNA was performed in each of the treatment groups for both TIMP-l-deficient and wild-type females. In the 0-h hcg mutant females, ovarian TIMP-2 (Fig. 3) and TIMP-3 content (Fig. 4) was significantly reduced compared to that in wild-type counterparts. No other differences in ovarian
4 1184 NOTHNICK ET AL..2 I (u*c ++ /+ Genotype FIG. 2. Bioassay of functional TIMP-1 protein in serum of wild-type and TIMP-1-deficient mice. To verify absence of a functional TIMP-1 protein in the TIMP-1-deficient mice, 100 *1l of serum was obtained from wildtype and mutant mice in the 24-h hcg group (n = 2 per genotype) and analyzed using a colorimetric inhibitor of metalloproteinase assay as described in Materials and Methods. The displayed difference in inhibitor activity between wild-type and mutant females is attributed to the absence of TIMP-1 protein in the TIMP-1 -deficient females. The baseline inhibitor value in the serum of TIMP-1-deficient mice is attributed to TIMP-2, as these animals did express TIMP-2 mrna but not TIMP-1 transcript. TIMP-2 (Fig. 3) or TIMP-3 (Fig. 4) mrna expression were detected between genotypes in the 12-h and 24-h hcg groups. Lastly, compensatory alterations in TIMP-2 and TIMP-3 were not detected between genotypes in lung (positive TIMP-2 control) and kidney (positive TIMP-3 control) tissue preparations (data not shown). To assess the effect of disruption of the TIMP-1 gene on ovarian morphology, histologic studies were conducted. In the 0-h hcg group, no differences between genotypes were noted with regard to number of healthy or atretic follicles or follicular size (Table 1). Similarly, no significant differences were detected in serum estradiol concentrations, body weight, or ovary weight between genotypes in the 0-h hcg groups (Table 1). Morphometric analysis of the ovaries from the 12-h hcg group revealed no significant differences in follicular development (tissue morphometrics and number of healthy and atretic follicles; data not shown), serum progesterone, body weight, or ovarian weight between genotypes (Table 2). Similar to findings for the 12-h hcg group, no significant differences were noted between genotypes in the 24-h hcg group with regard to gross luteal development (data not shown), serum progesterone, body weight, ovarian weight, or the number of oocytes released (Table 3). One striking feature of the ovarian histology of the TIMP-l-deficient mice was the lack of erythrocytes within the ovary, especially in the 0-h hcg group (Fig. 5, A and B). To support our in vivo findings that TIMP-1 may not be a regulator of steroidogenesis, granulosa cells were cultured in vitro and the steroidogenic effect of TIMP-1 was examined. In contrast to the in vivo estradiol data, exogenously added TIMP-1 significantly (p < 0.05) stimulated granulosa cell estradiol production in vitro in granulosa cells derived from both wild-type and TIMP-l-deficient mice, with the latter showing a 2-fold greater response compared to granulosa cells obtained from wild-type females (Table 4). In contrast to the in vitro estradiol data, but supporting the in vivo progesterone data, exogenously FIG. 3. Northern analysis of ovarian TIMP-2 mrna in wild-type and TIMP-1-deficient mice. Ovaries were collected from mice in each of the treatment groups, and total RNA was isolated and analyzed for TIMP-2 mrna content using 7.5 )pg of total RNA per lane as described in Materials and Methods. A) Detection of a 1.0- and 3.5-kilobase transcript for TIMP-2. The same blot hybridized for TIMP-1 (Fig. 1) was rehybridized for TIMP-2. The 18S hybridization signals for this membrane are displayed in Figure 1. All TIMP-2 data are normalized to the 18S transcript expression for their respective blots. B) Relative changes in TIMP-2 expression normalized to the 18S transcript for each of the treatment groups. Data are expressed as the mean + SEM for a total of three experiments; the depicted blot is a representation. * p < 0.05 compared to wild-type counterpart. added TIMP-1 had no effect on granulosa cell progesterone production in vitro in granulosa cells derived from either wild-type or TIMP-l-deficient females (Table 4). DISCUSSION The present study is the first to investigate the physiological role of TIMP-1 in the periovulatory period utilizing female mice incapable of expressing the functional TIMP-1 gene product (TIMP-l-deficient mice). As such, these studies allowed us to assess, in an in vivo setting, several potential functions of TIMP-1 that have been attributed to this metalloproteinase inhibitor based on in vitro studies. To ascertain that observed differences between genotypes were not due to a compensatory increase in expression of the other TIMP family members (TIMP-2 and/or TIMP-3), we examined whether disruption of the TIMP-1 gene resulted in an increase in mrna expression of TIMP-2 or TIMP-3. We observed a reduction in TIMP-2 and TIMP-3 ovarian mrna expression in ecg-primed TIMP-l-deficient mice (0-h time point) but were unable to detect an alteration in ovarian TIMP-2 or TIMP-3 mrna expression in unstimulated or ecg+hcg-stimulated (12 h and 24 h post-hcg)
5 OVARIAN TIMP-1 FUNCTION 1185 TABLE 2. Comparison between wild-type (+/+) and TIMP-1-deficient (-/-) mice in the 12-h hcg treatment group (mean SEM). Genotype Parameter assessed +/+ -/- Body weight (g) (n = 10) (n = 8) Combined ovary weight (mg) (n = 10) (n = 8) Serum progesterone (ng/ml) _ 0.7 (n = 9) (n = 6) FIG. 4. Northern analysis of ovarian TIMP-3 mrna in wild-type and TIMP-1-deficient mice. Ovaries were collected from mice in each of the treatment groups, and total RNA was isolated and analyzed for TIMP-3 mrna content using 7.5 jig of total RNA per lane as described in Materials and Methods. A) Detection of a single 4.5-kilobase transcript for TIMP-3. The blot in Figure 1 was cohybridized for TIMP-1 and TIMP-3, and the 18S hybridization signals are displayed in Figure 1. All TIMP-3 data are normalized to the 18S transcript expression for their respective blots. B) Relative changes in TIMP-3 expression normalized to the 18S transcript for each of the treatment groups. Data are expressed as the mean SEM for a total of three experiments; the depicted blot is a representation. * p < 0.05 compared to respective wild-type counterpart. TABLE 1. Comparison between wild-type (+/+) and TIMP-1-deficient (-/-) mice in the 0-h hcg treatment group (mean SEM). No. per Genotype Parameter assessed genotype +/+ -/- Body weight (g) ± Combined ovary weight (mg) ± Serum estradiol (pg/ml) ± Number of healthy follicles [im ± im >400 jim Number of atretic folliclesb M jim >400 ixm a Healthy follicles were defined as follicles that contained granulosa cells having no pyknotic nuclei. b Atretic follicles were defined as follicles that contained at least 3 granulosa cells having a pyknotic nuclei. mutant mice. This finding suggests that TIMP-1 may play either a direct or an indirect modulatory role in TIMP-2 and/or TIMP-3 expression during the latter stages of folliculogenesis and, to our knowledge, is the first report of a potential regulatory mechanism of TIMP-1 on the expression of other tissue inhibitors of metalloproteinase family members. We further speculate that this modulatory role of TIMP- 1 on TIMP-2/TIMP-3 expression may act to increase the inhibitory regulation of tissue remodeling. For instance, TIMP-1 is a secreted protein present in bodily fluids that bathe the tissues [26]. In these fluids, TIMP-1 may act as a first line of defense to regulate matrix metalloproteinase action and subsequent tissue breakdown and remodeling. To assure this defensive action, TIMP-1 may further act to regulate tissue remodeling by increasing the expression of the extracellular matrix-associated TIMP-3 [27-29]. As such, the absence of TIMP- 1 (as occurs in TIMP- 1-deficient mice) may result in lower levels of TIMP-3 expression as seen in findings in the current study. A TIMP-l-like protein has been reported to exhibit steroidogenic activity in cultured rat granulosa cells, specifically stimulating progesterone secretion [17]. The use of TIMP-l-deficient mice provides a unique experimental paradigm to examine the role of TIMP- 1 in regulation of ovarian steroidogenesis. In the current study, assessment of serum concentrations for both estrogen and progesterone in vivo failed to reveal differences among treatment groups between the genotypes, suggesting that TIMP-1 may not play a steroidogenic role in vivo. It should be noted that the previous study of a steroidogenic role for TIMP-1 assessed progesterone production in vitro [17]. To assess whether the lack of TIMP-1 modulation of steroidogenesis observed in our present in vivo study was due to differences between the in vivo and in vitro models, we cultured granulosa cells from wild-type and TIMP-l-deficient mice in the absence and presence of recombinant human TIMP-1. Similar to the in vivo' observations, no differences in estradiol or progesterone concentrations in the conditioned medium were found. Of particular interest were the modest steroidogenic effect of recombinant TIMP-1 on estradiol TABLE 3. Comparison between wild-type (+/+) and TIMP-1-deficient (-/-) mice in the 24-h hcg treatment group (mean SEM). Genotype No. per Parameter assessed genotype +/+ -/- Body weight (g) Combined ovary _ weight (mg) Number of oocytes released Serum progesterone (ng/ml) (n = 13) (n = 9)
6 1186 NOTHNICK ET AL. FIG. 5. Representative light photomicrographs of ovarian tissue from TIMP-1-deficient and wild-type animals in the treatment groups, counterstained with hematoxylin and eosin. Animals were treated and killed, and ovaries were removed and prepared for histologic assessment as described in Materials and Methods. A and B are ovaries from TIMP-1 -deficient and wild-type animals, respectively, in the 0-h hcg group. Arrows indicate high degree of erythrocytes in the ovarian vessels. Each photomicrograph is representative of a minimum of three animals per treatment group. F, follicle (magnification X220). secretion in these cells in vitro and the observation that this effect could be induced using nanomolar to micromolar concentrations. We attribute this discrepancy in the in vivo and in vitro steroidogenic capability of TIMP-1 to the fact that in the TIMP-1-deficient mice, other factors may still be present that stimulate or maintain steroidogenesis. The steroidogenic ability of TIMP-1 in vitro may be apparent when the cells are removed from their in vivo environment such that TIMP-1 may be present in greater quantities in vitro compared to other stimulators of steroidogenesis. Al- TABLE 4. Steroidogenic effect of exogenous TIMP-1 on wild-type (+/+) and TIMP-1 -deficient (-/-) granulosa cell estrogen and progesterone production in vitro (mean ± SEM). Basal steroid level Percentage change from basal level Steroid Genotype No. of trials (without TIMP-1) (with 1 [Lg/ml rtimp-1 added) Estradiol (pg/ml) +/ /b % b Progesterone (ng/ml) +/ % % rtimp-1, recombinant human TIMP-1. b Significantly different compared to basal steroid levels within genotype (p < 0.05).
7 OVARIAN TIMP-1 FUNCTION 1187 ternatively, the presence of cathepsin-l may be a prerequisite for TIMP-1 action and varying levels of this serine proteinase may impact the ability of TIMP-1 to stimulate steroidogenesis [17]. Also of interest is the finding that a 124-base-pair region of the 5' noncoding region of TIMP- 1 shares sequence homology with StAR, an acute controller for the transport of cholesterol to the inner mitochondrial membrane, suggesting possible common regulatory mechanisms between TIMP expression and regulation of steroidogenesis [18]. As TIMP-1 induces modest steroidogenic activity, we postulate that TIMP-1 most likely functions as a coregulator of steroidogenesis in vivo along with several other factors. One interesting observation in the current study was the lack of erythroid cells in the majority (3 of 4) of the ovaries of ecg-stimulated TIMP-l-deficient mice (i.e., 0 h) compared to the wild-type animals. TIMP-1 was initially identified by its ability to potentiate the formation of erythroid colonies from bone marrow precursors in vitro [30]. The difference observed in the present study may result from the impact of TIMP-1 on erythropoiesis, angiogenesis, vasculogenesis, or ovarian blood flow. As the hematocrits from wild-type and TIMP-l-deficient mice were identical (data not shown), the decrease in red blood cells in most of the mutant ovaries is striking. This decrease in erythroid cells is not due to a sampling artifact as ovaries were serially sectioned and subsequently examined. Although all of the tissues were handled in the same manner, the possibility that the difference in the content of ovarian erythrocytes is due to artifactual collection or processing of the tissues cannot be ruled out. Thus, further studies are necessary to elucidate the action of TIMP-1 on ovarian erythropoiesis and blood flow. Previous studies in our laboratory had demonstrated that TIMP-1 mrna expression and TIMP activity increase as ovulation approaches, first suggesting an associational role between the two parameters [3]. The current study revealed no differences between genotypes in ovarian weight, the number of oocytes released, and progesterone production both in vivo and in vitro. Furthermore, there were no differences in litter size, frequency, or total reproductive capacity between the wild-type and mutant mice. Thus, the perplexing question raised is, what is the function of an ovarian protein that is highly abundant and reaches peak levels around the time of oocyte release in the ovulatory process? The obvious possibility is that TIMP-1 functions only as a matrix metalloproteinase inhibitor and as such acts to regulate the extent and localization of follicular degradation. An alternative possibility is that TIMP-1 may stimulate collagenase production that in turn leads to oocyte release. TIMP-1 has been shown to stimulate human skin fibroblast production of collagenase [16], and this matrix metalloproteinase has been implicated in the ovulatory process [1, 3, 6, 21]. In the current study, we did not analyze potential compensatory mechanisms in matrix metalloproteinases that undoubtedly play a vital role in the ovulatory process [1, 3, 4, 6, 21]. Experiments in our laboratory are currently underway to examine the role of TIMP-1 in regulation of collagenase expression as associated with ovulation. Finally, TIMP- 1 has been postulated as a modulator of apoptosis [31, 32]. As there was no apparent difference in the morphology, steroidogenic potential, or number of oocytes released, it would appear that TIMP-1 has little or no impact on ovarian apoptosis. In summary, this is the first study to directly address the role of TIMP-1 in ovarian physiology using TIMP-l-deficient mice. Although TIMP-1 is highly abundant during the periovulatory period, TIMP-1 is not essential for fertility nor does it appear to be prerequisite for the processes that occur during the periovulatory period, such as progesterone production, oocyte release, and early luteal development. TIMP-1 does have a modest regulatory influence on ovarian TIMP-2 and TIMP-3 mrna expression during the period of follicular maturation and growth. Additionally, TIMP- 1 appears to be a coregulator of estradiol production in vitro, most likely functioning in concert with other stimulatory factors to modulate estradiol production during the period of follicular growth; however, there is no evidence for this activity in vivo. ACKNOWLEDGMENTS Gratitude is expressed to Mr. Sam Keeble and Ms. Carole Moore for performing the steroid RIAs. The generous gift of recombinant human TIMP-1 from Dr. Paul Cannon, Syntex Corp., Palo Alto, CA, is acknowledged. REFERENCES 1. Espey LL, Lipner H. Ovulation. In: Knobil E, Neill J (eds.), The Physiology of Reproduction. New York: Raven Press; 1994: Woessner JF Jr. Matrix metalloproteinases and their inhibitors in connective tissue remodeling. FASEB J 1991; 5: Curry TE Jr, LeMaire WJ, Dean DD, Sanders SL, Pedigo NG, Jones PBC. The role of ovarian proteases and their inhibitors in ovulation. Steroids 1989; 54: Curry TE Jr, Mann JS, Huang MH, Keeble SC. Gelatinase and proteoglycanase activity during the periovulatory period in the rat. Biol Reprod 1992; 46: Mann JS, Kindy MS, Edwards DR, Curry TE Jr. Hormonal regulation of matrix metalloproteinase inhibitors in rat granulosa cells and ovaries. Endocrinology 1991; 128: Reich R, Daphna-Iken D, Chun SY, Popliker M, Slager R, Adelmann- Grill BC, Tsafriri A. Preovulatory changes in ovarian expression of collagenase and tissue metalloproteinase inhibitor messenger ribonucleic acid: role of eicosanoids. Endocrinology 1991; 129: Freudenstein J, Wagner S, Luck RM, Eispaniere R, Scheit KH. mrna of bovine tissue inhibitor of metalloproteinases: sequence and expression in bovine ovarian tissue. Biochem Biophys Res Commun 1990; 171: Smith GW, Goetz TL, Anthony RV, Smith ME Molecular cloning of an ovine ovarian tissue inhibitor of metalloproteinases: ontogeny of messenger ribonucleic acid expression and in situ localization within preovulatory follicles and luteal tissue. Endocrinology 1994; 134: Mann JS, Kindy MS, Hyde JE Clark MR, Curry TE Jr. Role of protein synthesis, prostaglandins and estrogen in rat ovarian metalloproteinase inhibitor production. Biol Reprod 1993; 48: Murphy G, Reynolds JJ, Werb Z. Biosynthesis of tissue inhibitor of metalloproteinase by human fibroblasts in culture. Stimulation by 12-O-tetradecanoylphorbol 13-acetate and interleukin-1 in parallel with collagenase. J Biol Chem 1985; 260: Edwards DR, Murphy G, Reynolds JJ, Whitham SE, Docherty AJP, Angel P, Heath JK. Transforming growth factor beta modulates the expression of collagenase and metalloproteinase inhibitor. EMBO J 1987; 6: Bertaux B, Hornebeck W, Eisen AZ, Dubertret L. Growth stimulation of human keratinocytes by tissue inhibitors of metalloproteinases. J Invest Dermatol 1991; 97: Hayakawa T, Yamashita K, Tanazwa K, Uchijima E, Iwata K. Growth promoting activity of tissue inhibitor of metalloproteinase-1 (TIMP-1) for a wide range of cells. A possible new growth factor in serum. FEBS Lett 1992; 298: Gasson JC, Golde DW, Kaufman SE, Westbrook CA, Hewick RM, Kaufman RJ, Wong GG, Temple PA, Leary AC, Brown EL, Orr EC, Clark SC. Molecular characterization and expression of the gene encoding human euthyroid potentiating activity. Nature 1985; 15: Hayakawa T, Yamashita K, Kishi J, Harigaya K. Tissue inhibitor of metalloproteinases from human bone marrow stromal cell line KM 102 has erythroid potentiating activity, suggesting its possibly bifunc-
8 1188 NOTHNICK ET AL. tional role in the hematopoietic environment. FEBS Lett 1990: 268: Clark IM, Powell LK, Cawston TE. Tissue inhibitor of metalloproteinase (TIMP-1) stimulates the secretion of collagenase from human skin fibroblasts. Biochem Biophys Res Commun 1994: 203: Boujrad N, Ogwuegbu SO, Garnier M, Lee CH. Martin BM, Papadopoulos V. Identification of a stimulator of steroid hormone synthesis isolated from testis. Science 1995; 268: Hartung S, Rust W, Balvers M, Ivell R. Molecular cloning and in vivo expression of the bovine steroidogenic acute regulatory protein. Biochem Biophy Res Commun 1995; 215: Rapp G, Freudenstein J, Klaudiny J, Mucha J, Wempe F, Zimmer M, Scheit KH. Characterization of three abundant mrnas from human ovarian granulosa cells. DNA Cell Biol 1990; 9: Soloway PD, Alexander CM, Werb Z. Jaenisch R. Targeted mutagenesis of Timp-I reveals that lung tumor invasion is influenced by Timp-1 genotype of the tumor but not by that of the host. Oncogene 1996; 13: Nothnick WB, Keeble SC, Curry TE Jr. Collagenase, gelatinase, and proteoglycanase messenger ribonucleic acid expression and activity during luteal development, maintenance, and regression in the pseudopregnant rat ovary. Biol Reprod 1996; 54: Chomczynski P, Sacchi N. Single step method of RNA isolation by acid guanidinium thiocyanate-phenol-chloroform extraction. Anal Biochem 1987; 162: Luna LG. Manual of Histologic Staining Methods of the Armed Forces Institute of Pathology. New York: McGraw-Hill: 1968: Jones HM, Vernon MW, Rush ME. Androgenic modulation of periovulatory FSH release in the rat. Biol Reprod 1987; 37: Rosner B. Fundamentals of Biostatistics, 3rd ed. Boston: PWS-Dent; Birkedal-Hansen H, Moore WGI, Bodden MK, Windsor LJ, Birkedal- Hansen B, DeCarlo A, Engler JA. Matrix metalloproteinases: a review. Crit Rev Oral Biol 1993; 4: Leco KJ, Khokha R, Pavloff N, Hawkes SP, Edwards DR. Tissue inhibitor of metalloproteinase-3 (TIMP-3) is an extracellular matrixassociated protein with a distinct pattern of expression in mouse cells and tissues. J Biol Chem 1994; 269: Pavloff N, Staskus PW, Kishnani NS, Hawkes SP A new inhibitor of metalloproteinases from chicken: chlmp-3. A third member of the TIMP family. J Biol Chem 1992; 267: Staskus PW, Masiarz FR, Pallanck LJ, Hawkes SP The 21-kDa protein is a transformation-sensitive metalloproteinase inhibitor of chicken fibroblasts. J Biol Chem 1991; 266: Docherty AJP, Lyons A, Smith BJ, Wright EM, Stephens PE, Harris TJR, Murphy G, Reynolds JJ. Sequence of human tissue inhibitor of metalloproteinase and its identity to erythroid-potentiating activity. Nature 1985; 318: Talhouk RS, Bissell MJ, Werb Z. Coordinated expression of extracellular-matrix-degrading proteinases and their inhibitors regulates mammary epithelial function during involution. J Cell Biol 1992: 118: Li F, Strange R, Friis RR, Dionov V, Altermatt H-J, Saurer S, Niemann H, Andres A-C. Expression of stromelysin-l and TIMP-I in the involuting mammary gland and in early invasive tumors of the mouse. Int J Cancer 1994: 59:
(A) PCR primers (arrows) designed to distinguish wild type (P1+P2), targeted (P1+P2) and excised (P1+P3)14-
 1 Supplemental Figure Legends Figure S1. Mammary tumors of ErbB2 KI mice with 14-3-3σ ablation have elevated ErbB2 transcript levels and cell proliferation (A) PCR primers (arrows) designed to distinguish
1 Supplemental Figure Legends Figure S1. Mammary tumors of ErbB2 KI mice with 14-3-3σ ablation have elevated ErbB2 transcript levels and cell proliferation (A) PCR primers (arrows) designed to distinguish
OVARY The surface of the ovary is covered with surface epithelium
 OVARY Cow The ovary, or female gonad, is: 1. an exocrine gland, producing oocytes 2. an endocrine gland, secreting hormones, i.e., estrogen and progesterone OVARY OVARY The surface of the ovary is covered
OVARY Cow The ovary, or female gonad, is: 1. an exocrine gland, producing oocytes 2. an endocrine gland, secreting hormones, i.e., estrogen and progesterone OVARY OVARY The surface of the ovary is covered
ABSTRACT. Key words: ovulation, ovary, human, follicle, collagen, MMP and TIMP. ISBN-10: ISBN-13:
 HUMAN OVULATION Studies on collagens, gelatinases and tissue inhibitors of metalloproteinases Anna Karin Lind Department of Obstetrics and Gynecology Institute of Clinical Sciences Sahlgrenska University
HUMAN OVULATION Studies on collagens, gelatinases and tissue inhibitors of metalloproteinases Anna Karin Lind Department of Obstetrics and Gynecology Institute of Clinical Sciences Sahlgrenska University
THE MENSTRUAL CYCLE INA S. IRABON, MD, FPOGS, FPSRM, FPSGE OBSTETRICS AND GYNECOLOGY REPRODUCTIVE ENDOCRINOLOGY AND INFERTILITY
 THE MENSTRUAL CYCLE INA S. IRABON, MD, FPOGS, FPSRM, FPSGE OBSTETRICS AND GYNECOLOGY REPRODUCTIVE ENDOCRINOLOGY AND INFERTILITY REFERENCE Comprehensive Gynecology 7 th edition, 2017 (Lobo RA, Gershenson
THE MENSTRUAL CYCLE INA S. IRABON, MD, FPOGS, FPSRM, FPSGE OBSTETRICS AND GYNECOLOGY REPRODUCTIVE ENDOCRINOLOGY AND INFERTILITY REFERENCE Comprehensive Gynecology 7 th edition, 2017 (Lobo RA, Gershenson
REPRODUCTIVE CYCLE OF FEMALE MAMMAL
 REPRODUCTIVE CYCLE OF FEMALE MAMMAL Fig. 8-12 Secondary follicles growing follicles increase in number of layers of granulosa cells Tertiary follicles maturing follicles antrum formation fluid filled space
REPRODUCTIVE CYCLE OF FEMALE MAMMAL Fig. 8-12 Secondary follicles growing follicles increase in number of layers of granulosa cells Tertiary follicles maturing follicles antrum formation fluid filled space
Expression of acid base transporters in the kidney collecting duct in Slc2a7 -/-
 Supplemental Material Results. Expression of acid base transporters in the kidney collecting duct in Slc2a7 -/- and Slc2a7 -/- mice. The expression of AE1 in the kidney was examined in Slc26a7 KO mice.
Supplemental Material Results. Expression of acid base transporters in the kidney collecting duct in Slc2a7 -/- and Slc2a7 -/- mice. The expression of AE1 in the kidney was examined in Slc26a7 KO mice.
Effect of Resistin on Granulosa and Theca Cell Function in Cattle
 1 Effect of Resistin on Granulosa and Theca Cell Function in Cattle D.V. Lagaly, P.Y. Aad, L.B. Hulsey, J.A. Grado-Ahuir and L.J. Spicer Story in Brief Resistin is an adipokine that has not been extensively
1 Effect of Resistin on Granulosa and Theca Cell Function in Cattle D.V. Lagaly, P.Y. Aad, L.B. Hulsey, J.A. Grado-Ahuir and L.J. Spicer Story in Brief Resistin is an adipokine that has not been extensively
Reproductive Endocrinology. Isabel Hwang Department of Physiology Faculty of Medicine University of Hong Kong Hong Kong May2007
 Reproductive Endocrinology Isabel Hwang Department of Physiology Faculty of Medicine University of Hong Kong Hong Kong May2007 isabelss@hkucc.hku.hk A 3-hormone chain of command controls reproduction with
Reproductive Endocrinology Isabel Hwang Department of Physiology Faculty of Medicine University of Hong Kong Hong Kong May2007 isabelss@hkucc.hku.hk A 3-hormone chain of command controls reproduction with
CASE 41. What is the pathophysiologic cause of her amenorrhea? Which cells in the ovary secrete estrogen?
 CASE 41 A 19-year-old woman presents to her gynecologist with complaints of not having had a period for 6 months. She reports having normal periods since menarche at age 12. She denies sexual activity,
CASE 41 A 19-year-old woman presents to her gynecologist with complaints of not having had a period for 6 months. She reports having normal periods since menarche at age 12. She denies sexual activity,
Collagenase Assay Kit
 Collagenase Assay Kit Catalog # 31 and 32 For Research Use Only - Not Human or Therapeutic Use INTRODUCTION The collagenases are members of the matrix metalloproteinase (MMP) family and degrade collagen
Collagenase Assay Kit Catalog # 31 and 32 For Research Use Only - Not Human or Therapeutic Use INTRODUCTION The collagenases are members of the matrix metalloproteinase (MMP) family and degrade collagen
Development Supplementary information. Supplementary Figures * * +/+ +/- -/- +/+ +/- -/-
 Development 144: doi:1.1242/dev.1473: Supplementary information Supplementary Figures A (f) FRT LoxP 2 3 4 B All Males Females I Ovary 1 (+) 77 bps (f) 78 bps (-) >13 bps (-) 2 4 (-) 424 bps M +/f +/-
Development 144: doi:1.1242/dev.1473: Supplementary information Supplementary Figures A (f) FRT LoxP 2 3 4 B All Males Females I Ovary 1 (+) 77 bps (f) 78 bps (-) >13 bps (-) 2 4 (-) 424 bps M +/f +/-
LH (Bovine) ELISA Kit
 LH (Bovine) ELISA Kit Catalog Number KA2280 96 assays Version: 05 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle of the Assay...
LH (Bovine) ELISA Kit Catalog Number KA2280 96 assays Version: 05 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle of the Assay...
Endocrinology of the Female Reproductive Axis
 Endocrinology of the Female Reproductive Axis girlontheriver.com Geralyn Lambert-Messerlian, PhD, FACB Professor Women and Infants Hospital Alpert Medical School at Brown University Women & Infants BROWN
Endocrinology of the Female Reproductive Axis girlontheriver.com Geralyn Lambert-Messerlian, PhD, FACB Professor Women and Infants Hospital Alpert Medical School at Brown University Women & Infants BROWN
Superovulation of Beef Heifers with Follicle Stimulating Hormone or Human Menopausal Gonadotropin: Acute Effects on Hormone Secretion
 Superovulation of Beef Heifers with Follicle Stimulating Hormone or Human Menopausal Gonadotropin: Acute Effects on Hormone Secretion A.S. Leaflet R1362 Acacia A. Alcivar, graduate research assistant,
Superovulation of Beef Heifers with Follicle Stimulating Hormone or Human Menopausal Gonadotropin: Acute Effects on Hormone Secretion A.S. Leaflet R1362 Acacia A. Alcivar, graduate research assistant,
Collagenase Assay Kit
 Collagenase Assay Kit Catalog # 31 and 32 For Research Use Only - Not Human or Therapeutic Use INTRODUCTION Collagenases are members of the matrix metalloproteinase (MMP) family and degrade collagen types
Collagenase Assay Kit Catalog # 31 and 32 For Research Use Only - Not Human or Therapeutic Use INTRODUCTION Collagenases are members of the matrix metalloproteinase (MMP) family and degrade collagen types
SUPPLEMENTARY MATERIAL. Sample preparation for light microscopy
 SUPPLEMENTARY MATERIAL Sample preparation for light microscopy To characterize the granulocytes and melanomacrophage centers, cross sections were prepared for light microscopy, as described in Material
SUPPLEMENTARY MATERIAL Sample preparation for light microscopy To characterize the granulocytes and melanomacrophage centers, cross sections were prepared for light microscopy, as described in Material
Female Reproductive System. Lesson 10
 Female Reproductive System Lesson 10 Learning Goals 1. What are the five hormones involved in the female reproductive system? 2. Understand the four phases of the menstrual cycle. Human Reproductive System
Female Reproductive System Lesson 10 Learning Goals 1. What are the five hormones involved in the female reproductive system? 2. Understand the four phases of the menstrual cycle. Human Reproductive System
Superovulation of Beef Heifers with Follicle Stimulating Hormone or Human Menopausal Gonadotropin: Acute Effects on Hormone Secretion
 Beef Research Report, 1996 Animal Science Research Reports 1997 Superovulation of Beef Heifers with Follicle Stimulating Hormone or Human Menopausal Gonadotropin: Acute Effects on Hormone Secretion Acacia
Beef Research Report, 1996 Animal Science Research Reports 1997 Superovulation of Beef Heifers with Follicle Stimulating Hormone or Human Menopausal Gonadotropin: Acute Effects on Hormone Secretion Acacia
The Distribution of Collagenous Connective Tissue in Rat Ovarian Follicles
 BIOLOGY OF REPRODUCTION 14, 502-506 (1976). The Distribution of Collagenous Connective Tissue in Rat Ovarian Follicles LAWRENCE L ESPEY Reproductive Physiology Laboratory, Trinity University, San Antonio,
BIOLOGY OF REPRODUCTION 14, 502-506 (1976). The Distribution of Collagenous Connective Tissue in Rat Ovarian Follicles LAWRENCE L ESPEY Reproductive Physiology Laboratory, Trinity University, San Antonio,
Treatment 3 Days After Ovulation In Mares
 Luteal Regression And Follicle Development Following Prostaglandin-F 2α Treatment 3 Days After Ovulation In Mares D.R. Bergfelt a, R.A. Pierson b, and O.J. Ginther a a University of Wisconsin, Madison,
Luteal Regression And Follicle Development Following Prostaglandin-F 2α Treatment 3 Days After Ovulation In Mares D.R. Bergfelt a, R.A. Pierson b, and O.J. Ginther a a University of Wisconsin, Madison,
LH (Rodent) ELISA Kit
 LH (Rodent) ELISA Kit Catalog Number KA2332 96 assays Version: 05 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle of the Assay...
LH (Rodent) ELISA Kit Catalog Number KA2332 96 assays Version: 05 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle of the Assay...
CLARITY reveals dynamics of ovarian follicular architecture and vasculature in three-dimensions
 CLARITY reveals dynamics of ovarian follicular architecture and vasculature in three-dimensions Yi Feng, Peng Cui, Xiaowei Lu, Brian Hsueh, Fredrik Möller Billig, Livia Zarnescu Yanez, Raju Tomer, Derek
CLARITY reveals dynamics of ovarian follicular architecture and vasculature in three-dimensions Yi Feng, Peng Cui, Xiaowei Lu, Brian Hsueh, Fredrik Möller Billig, Livia Zarnescu Yanez, Raju Tomer, Derek
Effects of Label-Dose Permethrin Administration on Reproductive Function and Embryo Quality on Superovulated Beef Heifers
 Animal Industry Report AS 662 ASL R3050 2016 Effects of Label-Dose Permethrin Administration on Reproductive Function and Embryo Quality on Superovulated Beef Heifers Tyler M. Dohlman Iowa State University,
Animal Industry Report AS 662 ASL R3050 2016 Effects of Label-Dose Permethrin Administration on Reproductive Function and Embryo Quality on Superovulated Beef Heifers Tyler M. Dohlman Iowa State University,
Fertility Diagnostics
 Fertility Diagnostics Fertility hormones measured on PATHFAST For internal use only Diagnostics PATHFAST Chemiluminescence-immuno-analyzer 1 Content: page 1. Fertility hormones - general aspects 1.1 Reproductive
Fertility Diagnostics Fertility hormones measured on PATHFAST For internal use only Diagnostics PATHFAST Chemiluminescence-immuno-analyzer 1 Content: page 1. Fertility hormones - general aspects 1.1 Reproductive
SISTEMA REPRODUCTOR (LA IDEA FIJA) Copyright 2004 Pearson Education, Inc., publishing as Benjamin Cummings
 SISTEMA REPRODUCTOR (LA IDEA FIJA) How male and female reproductive systems differentiate The reproductive organs and how they work How gametes are produced and fertilized Pregnancy, stages of development,
SISTEMA REPRODUCTOR (LA IDEA FIJA) How male and female reproductive systems differentiate The reproductive organs and how they work How gametes are produced and fertilized Pregnancy, stages of development,
MICROWELL ELISA LUTEINIZING HORMONE (LH) ENZYMEIMMUNOASSAY TEST KIT LH ELISA. Cat # 4225Z
 DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
REPRODUCCIÓN. La idea fija. Copyright 2004 Pearson Education, Inc., publishing as Benjamin Cummings
 REPRODUCCIÓN La idea fija How male and female reproductive systems differentiate The reproductive organs and how they work How gametes are produced and fertilized Pregnancy, stages of development, birth
REPRODUCCIÓN La idea fija How male and female reproductive systems differentiate The reproductive organs and how they work How gametes are produced and fertilized Pregnancy, stages of development, birth
10.7 The Reproductive Hormones
 10.7 The Reproductive Hormones December 10, 2013. Website survey?? QUESTION: Who is more complicated: men or women? The Female Reproductive System ovaries: produce gametes (eggs) produce estrogen (steroid
10.7 The Reproductive Hormones December 10, 2013. Website survey?? QUESTION: Who is more complicated: men or women? The Female Reproductive System ovaries: produce gametes (eggs) produce estrogen (steroid
EVALUATION OF FOLLICULAR ATRESIA AND ELECTROPHORETIC PATTERN OF FOLLICULAR FLUID PROTEINS IN ACYCLIC BUFFALO (Bubalus bubalis)
 Original Article Buffalo Bulletin (December 2013) Vol.32 No.4 EVALUATION OF FOLLICULAR ATRESIA AND ELECTROPHORETIC PATTERN OF FOLLICULAR FLUID PROTEINS IN ACYCLIC BUFFALO (Bubalus bubalis) F.A. Khan 1,*,
Original Article Buffalo Bulletin (December 2013) Vol.32 No.4 EVALUATION OF FOLLICULAR ATRESIA AND ELECTROPHORETIC PATTERN OF FOLLICULAR FLUID PROTEINS IN ACYCLIC BUFFALO (Bubalus bubalis) F.A. Khan 1,*,
Ovarian Follicular Development in the Untreated and
 Ovarian Follicular Development in the Untreated and PMSG-treated Cyclic Rat Hajime MIYAMOTO, Goro KATSUURA and Takehiko ISHIBASHI Department of Animal Science, College of Agriculture, Kyoto University,
Ovarian Follicular Development in the Untreated and PMSG-treated Cyclic Rat Hajime MIYAMOTO, Goro KATSUURA and Takehiko ISHIBASHI Department of Animal Science, College of Agriculture, Kyoto University,
Mouse Cathepsin B ELISA Kit
 GenWay Biotech, Inc. 6777 Nancy Ridge Drive San Diego, CA 92121 Phone: 858.458.0866 Fax: 858.458.0833 Email: techline@genwaybio.com http://www.genwaybio.com Mouse Cathepsin B ELISA Kit Catalog No. GWB-ZZD154
GenWay Biotech, Inc. 6777 Nancy Ridge Drive San Diego, CA 92121 Phone: 858.458.0866 Fax: 858.458.0833 Email: techline@genwaybio.com http://www.genwaybio.com Mouse Cathepsin B ELISA Kit Catalog No. GWB-ZZD154
Leanne J. Bakke, 4 Qinglei Li, 3,5 Carolyn A. Cassar, 3 Mark P.D. Dow, 3 J. Richard Pursley, 3 and George W. Smith 2,3,4,5
 BIOLOGY OF REPRODUCTION 71, 605 612 (2004) Published online before print 28 April 2004. DOI 10.1095/biolreprod.104.027185 Gonadotropin Surge-Induced Differential Upregulation of Collagenase-1 (MMP-1) and
BIOLOGY OF REPRODUCTION 71, 605 612 (2004) Published online before print 28 April 2004. DOI 10.1095/biolreprod.104.027185 Gonadotropin Surge-Induced Differential Upregulation of Collagenase-1 (MMP-1) and
Two important cells in female are the theca cells and the granulose cells. Granulosa cells are affected by the two gonadotropin hormones; FSH and LH.
 1 UGS physiology sheet #13 lecture 3 Dr.Saleem Khresha. Now we will start discussing the female reproductive system Ovarian Steroids Two important cells in female are the theca cells and the granulose
1 UGS physiology sheet #13 lecture 3 Dr.Saleem Khresha. Now we will start discussing the female reproductive system Ovarian Steroids Two important cells in female are the theca cells and the granulose
Human Cathepsin D ELISA Kit
 GenWay Biotech, Inc. 6777 Nancy Ridge Drive San Diego, CA 92121 Phone: 858.458.0866 Fax: 858.458.0833 Email: techline@genwaybio.com http://www.genwaybio.com Human Cathepsin D ELISA Kit Catalog No. GWB-J4JVV9
GenWay Biotech, Inc. 6777 Nancy Ridge Drive San Diego, CA 92121 Phone: 858.458.0866 Fax: 858.458.0833 Email: techline@genwaybio.com http://www.genwaybio.com Human Cathepsin D ELISA Kit Catalog No. GWB-J4JVV9
Effects of Catecholamines and Dibenamine on Ovulation in the Perfused Fowl Ovary
 Effects of Catecholamines and Dibenamine on Ovulation in the Perfused Fowl Ovary Tomoki HIGUCHI, Tomoki SOH, Frank HERTELENDY* and Kousaku TANAKA Faculty of Agriculture, Kyushu University, Higashi-ku,
Effects of Catecholamines and Dibenamine on Ovulation in the Perfused Fowl Ovary Tomoki HIGUCHI, Tomoki SOH, Frank HERTELENDY* and Kousaku TANAKA Faculty of Agriculture, Kyushu University, Higashi-ku,
IN OVARIAN ANTRAL FOLLICULAR FLUID OF BUFFALOES*
 Indian J. Anim. Res., 41 (2): 106-110, 2007 i.exeenzymatic PROFILES OF ACID AND ALKALINE PHOSPHATASES IN OVARIAN ANTRAL FOLLICULAR FLUID OF BUFFALOES* G.P. Kalmath and J.P. Ravindra 1 ** Department of
Indian J. Anim. Res., 41 (2): 106-110, 2007 i.exeenzymatic PROFILES OF ACID AND ALKALINE PHOSPHATASES IN OVARIAN ANTRAL FOLLICULAR FLUID OF BUFFALOES* G.P. Kalmath and J.P. Ravindra 1 ** Department of
Central Progesterone Involvement in Estrogen- Induced Prolactin and Luteinizing Hormone Secretion Surges in Female Rats
 Southern Illinois University Carbondale OpenSIUC Honors Theses University Honors Program 5-10-2014 Central Progesterone Involvement in Estrogen- Induced Prolactin and Luteinizing Hormone Secretion Surges
Southern Illinois University Carbondale OpenSIUC Honors Theses University Honors Program 5-10-2014 Central Progesterone Involvement in Estrogen- Induced Prolactin and Luteinizing Hormone Secretion Surges
MINERAL PROFILES OF OVARIAN ANTRAL FOLLICULAR FLUID IN BUFFALOES DURING FOLLICULAR DEVELOPMENT*
 Indian J. Anim. Res., 41 (2): 87-93, 2007 MINERAL PROFILES OF OVARIAN ANTRAL FOLLICULAR FLUID IN BUFFALOES DURING FOLLICULAR DEVELOPMENT* G.P. Kalmath and J.P. Ravindra** Department of Veterinary Physiology,
Indian J. Anim. Res., 41 (2): 87-93, 2007 MINERAL PROFILES OF OVARIAN ANTRAL FOLLICULAR FLUID IN BUFFALOES DURING FOLLICULAR DEVELOPMENT* G.P. Kalmath and J.P. Ravindra** Department of Veterinary Physiology,
SensoLyte Generic MMP Assay Kit *Colorimetric*
 SensoLyte Generic MMP Assay Kit *Colorimetric* Revision#1.2 Catalog # Kit Size Last updated: May2017 AS-72095 100 Assays (96-well plate) Optimized Performance: This kit is optimized to detect MMP activity
SensoLyte Generic MMP Assay Kit *Colorimetric* Revision#1.2 Catalog # Kit Size Last updated: May2017 AS-72095 100 Assays (96-well plate) Optimized Performance: This kit is optimized to detect MMP activity
Biology of Reproduction- Zool 346 Exam 2
 Biology of Reproduction- Zool 346 Exam 2 ANSWER ALL THE QUESTIONS ON THE ANSWER SHEET. THE ANSWER ON THE ANSWER SHEET IS YOUR OFFICIAL ANSWER. Some critical words are boldfaced. This exam is 7 pages long.
Biology of Reproduction- Zool 346 Exam 2 ANSWER ALL THE QUESTIONS ON THE ANSWER SHEET. THE ANSWER ON THE ANSWER SHEET IS YOUR OFFICIAL ANSWER. Some critical words are boldfaced. This exam is 7 pages long.
FSH (Rodent) ELISA Kit
 FSH (Rodent) ELISA Kit Catalog Number KA2330 96 assays Version: 06 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle of the Assay...
FSH (Rodent) ELISA Kit Catalog Number KA2330 96 assays Version: 06 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle of the Assay...
Ovarian Characteristics, Serum Hormone Concentrations, and Fertility in Lactating Dairy Cows in Response to Equine Chorionic Gonadotropin
 Ovarian Characteristics, Serum Hormone Concentrations, and Fertility in Lactating Dairy Cows in Response to quine Chorionic Gonadotropin S. L. Pulley, L. D. Wallace, H. I. Mellieon, and J. S. Stevenson
Ovarian Characteristics, Serum Hormone Concentrations, and Fertility in Lactating Dairy Cows in Response to quine Chorionic Gonadotropin S. L. Pulley, L. D. Wallace, H. I. Mellieon, and J. S. Stevenson
Supplemental Figure 1: Leydig cells are reduced at multiple stages in both male sterile mutants
 SUPPLEMENTAL FIGURE LEGENDS: Supplemental Figure 1: Leydig cells are reduced at multiple stages in both male sterile mutants (Sgpl1 -/- and Plekha1 -/- ). Using an antibody against CYP11a1 to label Leydig
SUPPLEMENTAL FIGURE LEGENDS: Supplemental Figure 1: Leydig cells are reduced at multiple stages in both male sterile mutants (Sgpl1 -/- and Plekha1 -/- ). Using an antibody against CYP11a1 to label Leydig
1. During the follicular phase of the ovarian cycle, the hypothalamus releases GnRH.
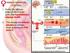 1. During the follicular phase of the ovarian cycle, the hypothalamus releases GnRH. 2. This causes the anterior pituitary to secrete small quantities of FSH and LH. 3. At this time, the follicles in the
1. During the follicular phase of the ovarian cycle, the hypothalamus releases GnRH. 2. This causes the anterior pituitary to secrete small quantities of FSH and LH. 3. At this time, the follicles in the
The Human Menstrual Cycle
 The Human Menstrual Cycle Name: The female human s menstrual cycle is broken into two phases: the Follicular Phase and the Luteal Phase. These two phases are separated by an event called ovulation. (1)
The Human Menstrual Cycle Name: The female human s menstrual cycle is broken into two phases: the Follicular Phase and the Luteal Phase. These two phases are separated by an event called ovulation. (1)
Folliculogenesis: Physiology and pathophysiology
 DIMITRIOS LOUTRADIS Professor of Obstetrics and Gynaecology Head of 1st Department of Obstetrics and Gynaecology University of Athens Medical School Alexandra Hospital Folliculogenesis: Physiology and
DIMITRIOS LOUTRADIS Professor of Obstetrics and Gynaecology Head of 1st Department of Obstetrics and Gynaecology University of Athens Medical School Alexandra Hospital Folliculogenesis: Physiology and
The intra-follicular molecular biology mandating advancement of egg retrieval in some women
 The intra-follicular molecular biology mandating advancement of egg retrieval in some women David H. Barad, USA Director of Assisted Reproductive Technology, The Center for Human Reproduction New York
The intra-follicular molecular biology mandating advancement of egg retrieval in some women David H. Barad, USA Director of Assisted Reproductive Technology, The Center for Human Reproduction New York
Ovarian Expression of a Disintegrin and Metalloproteinase with Thrombospondin Motifs During Ovulation in the Gonadotropin-Primed Immature Rat 1
 BIOLOGY OF REPRODUCTION 62, 1090 1095 (2000) Ovarian Expression of a Disintegrin and Metalloproteinase with Thrombospondin Motifs During Ovulation in the Gonadotropin-Primed Immature Rat 1 Lawrence L.
BIOLOGY OF REPRODUCTION 62, 1090 1095 (2000) Ovarian Expression of a Disintegrin and Metalloproteinase with Thrombospondin Motifs During Ovulation in the Gonadotropin-Primed Immature Rat 1 Lawrence L.
Investigation: The Human Menstrual Cycle Research Question: How do hormones control the menstrual cycle?
 Investigation: The Human Menstrual Cycle Research Question: How do hormones control the menstrual cycle? Introduction: The menstrual cycle (changes within the uterus) is an approximately 28-day cycle that
Investigation: The Human Menstrual Cycle Research Question: How do hormones control the menstrual cycle? Introduction: The menstrual cycle (changes within the uterus) is an approximately 28-day cycle that
The reproductive lifespan
 The reproductive lifespan Reproductive potential Ovarian cycles Pregnancy Lactation Male Female Puberty Menopause Age Menstruation is an external indicator of ovarian events controlled by the hypothalamicpituitary
The reproductive lifespan Reproductive potential Ovarian cycles Pregnancy Lactation Male Female Puberty Menopause Age Menstruation is an external indicator of ovarian events controlled by the hypothalamicpituitary
T Lin, D Wang and D M Stocco 1
 461 Interleukin-1 inhibits Leydig cell steroidogenesis without affecting steroidogenic acute regulatory protein messenger ribonucleic acid or protein levels T Lin, D Wang and D M Stocco 1 Medical and Research
461 Interleukin-1 inhibits Leydig cell steroidogenesis without affecting steroidogenic acute regulatory protein messenger ribonucleic acid or protein levels T Lin, D Wang and D M Stocco 1 Medical and Research
Chapter 14 Reproduction Review Assignment
 Date: Mark: _/45 Chapter 14 Reproduction Review Assignment Multiple Choice Identify the choice that best completes the statement or answers the question. 1. Use the diagram above to answer the next question.
Date: Mark: _/45 Chapter 14 Reproduction Review Assignment Multiple Choice Identify the choice that best completes the statement or answers the question. 1. Use the diagram above to answer the next question.
LH (Canine) ELISA Kit
 LH (Canine) ELISA Kit Catalog Number KA2292 96 assays Version: 05 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle of the Assay...
LH (Canine) ELISA Kit Catalog Number KA2292 96 assays Version: 05 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle of the Assay...
LUTEINIZED UNRUPTURED FOLLICLE SYNDROME: A SUBTLE CAUSE OF INFERTILITY*
 FERTILITY AND STERILITY Copyright c 1978 The American Fertility Society Vol. 29, No.3, March 1978 Printed in U.S.A. LUTEINIZED UNRUPTURED FOLLICLE SYNDROME: A SUBTLE CAUSE OF INFERTILITY* JAROSLA V MARIK,
FERTILITY AND STERILITY Copyright c 1978 The American Fertility Society Vol. 29, No.3, March 1978 Printed in U.S.A. LUTEINIZED UNRUPTURED FOLLICLE SYNDROME: A SUBTLE CAUSE OF INFERTILITY* JAROSLA V MARIK,
Hormonal Control of Human Reproduction
 Hormonal Control of Human Reproduction Bởi: OpenStaxCollege The human male and female reproductive cycles are controlled by the interaction of hormones from the hypothalamus and anterior pituitary with
Hormonal Control of Human Reproduction Bởi: OpenStaxCollege The human male and female reproductive cycles are controlled by the interaction of hormones from the hypothalamus and anterior pituitary with
Chapter 27 The Reproductive System. MDufilho
 Chapter 27 The Reproductive System 1 Figure 27.19 Events of oogenesis. Before birth Meiotic events 2n Oogonium (stem cell) Mitosis Follicle development in ovary Follicle cells Oocyte 2n Primary oocyte
Chapter 27 The Reproductive System 1 Figure 27.19 Events of oogenesis. Before birth Meiotic events 2n Oogonium (stem cell) Mitosis Follicle development in ovary Follicle cells Oocyte 2n Primary oocyte
Female Reproductive System. Justin D. Vidal
 Female Reproductive System Justin D. Vidal If you cannot identify the tissue, then it is probably part of the female reproductive system! Introduction The female reproductive system is constantly changing,
Female Reproductive System Justin D. Vidal If you cannot identify the tissue, then it is probably part of the female reproductive system! Introduction The female reproductive system is constantly changing,
Phases of the Ovarian Cycle
 OVARIAN CYCLE An ovary contains many follicles, and each one contains an immature egg called an oocyte. A female is born with as many as 2 million follicles, but the number is reduced to 300,000 to 400,000
OVARIAN CYCLE An ovary contains many follicles, and each one contains an immature egg called an oocyte. A female is born with as many as 2 million follicles, but the number is reduced to 300,000 to 400,000
Procaspase-3. Cleaved caspase-3. actin. Cytochrome C (10 M) Z-VAD-fmk. Procaspase-3. Cleaved caspase-3. actin. Z-VAD-fmk
 A HeLa actin - + + - - + Cytochrome C (1 M) Z-VAD-fmk PMN - + + - - + actin Cytochrome C (1 M) Z-VAD-fmk Figure S1. (A) Pan-caspase inhibitor z-vad-fmk inhibits cytochrome c- mediated procaspase-3 cleavage.
A HeLa actin - + + - - + Cytochrome C (1 M) Z-VAD-fmk PMN - + + - - + actin Cytochrome C (1 M) Z-VAD-fmk Figure S1. (A) Pan-caspase inhibitor z-vad-fmk inhibits cytochrome c- mediated procaspase-3 cleavage.
Female Reproductive Physiology. Dr Raelia Lew CREI, FRANZCOG, PhD, MMed, MBBS Fertility Specialist, Melbourne IVF
 Female Reproductive Physiology Dr Raelia Lew CREI, FRANZCOG, PhD, MMed, MBBS Fertility Specialist, Melbourne IVF REFERENCE Lew, R, Natural History of ovarian function including assessment of ovarian reserve
Female Reproductive Physiology Dr Raelia Lew CREI, FRANZCOG, PhD, MMed, MBBS Fertility Specialist, Melbourne IVF REFERENCE Lew, R, Natural History of ovarian function including assessment of ovarian reserve
Supplementary Figure 1: Neuregulin 1 increases the growth of mammary organoids compared to EGF. (a) Mammary epithelial cells were freshly isolated,
 1 2 3 4 5 6 7 8 9 10 Supplementary Figure 1: Neuregulin 1 increases the growth of mammary organoids compared to EGF. (a) Mammary epithelial cells were freshly isolated, embedded in matrigel and exposed
1 2 3 4 5 6 7 8 9 10 Supplementary Figure 1: Neuregulin 1 increases the growth of mammary organoids compared to EGF. (a) Mammary epithelial cells were freshly isolated, embedded in matrigel and exposed
Endocrine System Hormones (Ch. 45)
 Endocrine System Hormones (Ch. 45) Regulation Why are hormones needed? chemical messages from one body part to another communication needed to coordinate whole body daily homeostasis & regulation of large
Endocrine System Hormones (Ch. 45) Regulation Why are hormones needed? chemical messages from one body part to another communication needed to coordinate whole body daily homeostasis & regulation of large
SUPPLEMENTAL MATERIAL. Supplementary Methods
 SUPPLEMENTAL MATERIAL Supplementary Methods Culture of cardiomyocytes, fibroblasts and cardiac microvascular endothelial cells The isolation and culturing of neonatal rat ventricular cardiomyocytes was
SUPPLEMENTAL MATERIAL Supplementary Methods Culture of cardiomyocytes, fibroblasts and cardiac microvascular endothelial cells The isolation and culturing of neonatal rat ventricular cardiomyocytes was
(Stratagene, La Jolla, CA) (Supplemental Fig. 1A). A 5.4-kb EcoRI fragment
 SUPPLEMENTAL INFORMATION Supplemental Methods Generation of RyR2-S2808D Mice Murine genomic RyR2 clones were isolated from a 129/SvEvTacfBR λ-phage library (Stratagene, La Jolla, CA) (Supplemental Fig.
SUPPLEMENTAL INFORMATION Supplemental Methods Generation of RyR2-S2808D Mice Murine genomic RyR2 clones were isolated from a 129/SvEvTacfBR λ-phage library (Stratagene, La Jolla, CA) (Supplemental Fig.
Outline. Male Reproductive System Testes and Sperm Hormonal Regulation
 Outline Male Reproductive System Testes and Sperm Hormonal Regulation Female Reproductive System Genital Tract Hormonal Levels Uterine Cycle Fertilization and Pregnancy Control of Reproduction Infertility
Outline Male Reproductive System Testes and Sperm Hormonal Regulation Female Reproductive System Genital Tract Hormonal Levels Uterine Cycle Fertilization and Pregnancy Control of Reproduction Infertility
I. Endocrine System & Hormones Figure 1: Human Endocrine System
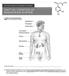 I. Endocrine System & Hormones Figure 1: Human Endocrine System Endocrine System: a) Endocrine glands are ductless since they lack specific vessels for the transport of hormones throughout the body. Instead,
I. Endocrine System & Hormones Figure 1: Human Endocrine System Endocrine System: a) Endocrine glands are ductless since they lack specific vessels for the transport of hormones throughout the body. Instead,
Endocrinology laboratory Department of Zoology Kalyani University Kalyani, West Bengal India
 Epidermal growth factor (EGF) promotes ovarian steroidogenesis and epidermal growth factor receptor (EGFR) signaling is required for gonadotropin-induced steroid production in common carp Cyprinus carpio
Epidermal growth factor (EGF) promotes ovarian steroidogenesis and epidermal growth factor receptor (EGFR) signaling is required for gonadotropin-induced steroid production in common carp Cyprinus carpio
SUPPLEMENTARY INFORMATION
 SUPPLEMENTARY INFORMATION FOR Liver X Receptor α mediates hepatic triglyceride accumulation through upregulation of G0/G1 Switch Gene 2 (G0S2) expression I: SUPPLEMENTARY METHODS II: SUPPLEMENTARY FIGURES
SUPPLEMENTARY INFORMATION FOR Liver X Receptor α mediates hepatic triglyceride accumulation through upregulation of G0/G1 Switch Gene 2 (G0S2) expression I: SUPPLEMENTARY METHODS II: SUPPLEMENTARY FIGURES
Effects of superovulation with FSH. Abstract
 Chapter 2 Effects of superovulation with ofsh and norgestomet/gnrh-controlled release of the LH surge on hormone concentrations, and yield of oocytes and embryos at specific developmental stages Effects
Chapter 2 Effects of superovulation with ofsh and norgestomet/gnrh-controlled release of the LH surge on hormone concentrations, and yield of oocytes and embryos at specific developmental stages Effects
Progesterone and clinical outcomes
 Synchronization of Slowly Developing Embryos Restores Implantation Success Richard T. Scott, Jr, MD, HCLD Clinical and Scientific Director, Reproductive Medicine Associates of New Jersey Professor and
Synchronization of Slowly Developing Embryos Restores Implantation Success Richard T. Scott, Jr, MD, HCLD Clinical and Scientific Director, Reproductive Medicine Associates of New Jersey Professor and
Signaling Vascular Morphogenesis and Maintenance
 Signaling Vascular Morphogenesis and Maintenance Douglas Hanahan Science 277: 48-50, in Perspectives (1997) Blood vessels are constructed by two processes: vasculogenesis, whereby a primitive vascular
Signaling Vascular Morphogenesis and Maintenance Douglas Hanahan Science 277: 48-50, in Perspectives (1997) Blood vessels are constructed by two processes: vasculogenesis, whereby a primitive vascular
Effect of Bovine Follicular Fluid Added to the Maturation Medium on Sperm Penetration in Pig Oocytes Matured In Vitro
 Article Effect of Bovine Follicular Fluid Added to the Maturation Medium on Sperm Penetration in Pig Oocytes Matured In Vitro Abstract Naoki ISOBE Research Associate Graduate School for International Development
Article Effect of Bovine Follicular Fluid Added to the Maturation Medium on Sperm Penetration in Pig Oocytes Matured In Vitro Abstract Naoki ISOBE Research Associate Graduate School for International Development
Manual (Second edition)
 Reagent for RNA Extraction ISOGENⅡ Manual (Second edition) Code No. 311-07361 Code No. 317-07363 NIPPON GENE CO., LTD. Table of contents I Product description 1 II Product content 1 III Storage 1 IV Precautions
Reagent for RNA Extraction ISOGENⅡ Manual (Second edition) Code No. 311-07361 Code No. 317-07363 NIPPON GENE CO., LTD. Table of contents I Product description 1 II Product content 1 III Storage 1 IV Precautions
Effect of addition of exogenous growth factor on in vitro development of preimplantation stage buffalo embryos
 Effect of addition of exogenous growth factor on in vitro development of preimplantation stage buffalo embryos CONTENTS 5. EFFECT OF ADDITION OF EXOGENOUS GROWTH FACTOR ON IN VITRO DEVELOPMENT OF PREIMPLANTATION
Effect of addition of exogenous growth factor on in vitro development of preimplantation stage buffalo embryos CONTENTS 5. EFFECT OF ADDITION OF EXOGENOUS GROWTH FACTOR ON IN VITRO DEVELOPMENT OF PREIMPLANTATION
Reproductive Health and Pituitary Disease
 Reproductive Health and Pituitary Disease Janet F. McLaren, MD Assistant Professor Division of Reproductive Endocrinology and Infertility Department of Obstetrics and Gynecology jmclaren@uabmc.edu Objectives
Reproductive Health and Pituitary Disease Janet F. McLaren, MD Assistant Professor Division of Reproductive Endocrinology and Infertility Department of Obstetrics and Gynecology jmclaren@uabmc.edu Objectives
Department of Obstetrics and Gynecology, IVF Unit, Carmel Medical Center, Haifa, Israel
 FERTILITY AND STERILITY VOL. 79, NO. 3, MARCH 2003 Copyright 2003 American Society for Reproductive Medicine Published by Elsevier Science Inc. Printed on acid-free paper in U.S.A. Decreased expression
FERTILITY AND STERILITY VOL. 79, NO. 3, MARCH 2003 Copyright 2003 American Society for Reproductive Medicine Published by Elsevier Science Inc. Printed on acid-free paper in U.S.A. Decreased expression
Supplementary Information Titles Journal: Nature Medicine
 Supplementary Information Titles Journal: Nature Medicine Article Title: Corresponding Author: Supplementary Item & Number Supplementary Fig.1 Fig.2 Fig.3 Fig.4 Fig.5 Fig.6 Fig.7 Fig.8 Fig.9 Fig. Fig.11
Supplementary Information Titles Journal: Nature Medicine Article Title: Corresponding Author: Supplementary Item & Number Supplementary Fig.1 Fig.2 Fig.3 Fig.4 Fig.5 Fig.6 Fig.7 Fig.8 Fig.9 Fig. Fig.11
Regulation of the IGF axis by TGF-b during periosteal chondrogenesis: implications for articular cartilage repair
 Regulation of the IGF axis by TGF-b during periosteal chondrogenesis: implications for articular cartilage repair Chapter 04 Boek 1_Gie.indb 55 21-05-2007 12:27:33 Chapter 04 Abstract Goal: TGF-b and IGF-I
Regulation of the IGF axis by TGF-b during periosteal chondrogenesis: implications for articular cartilage repair Chapter 04 Boek 1_Gie.indb 55 21-05-2007 12:27:33 Chapter 04 Abstract Goal: TGF-b and IGF-I
Nutritional and metabolic mechanisms. in the ovarian follicle
 Nutritional and metabolic mechanisms in the ovarian follicle Joëlle Dupont Team Leader : «Interaction Metabolism and Reproduction» Unit of Physiology of Reproduction and Behaviors UMR 6175 INRA/CNRS/Université
Nutritional and metabolic mechanisms in the ovarian follicle Joëlle Dupont Team Leader : «Interaction Metabolism and Reproduction» Unit of Physiology of Reproduction and Behaviors UMR 6175 INRA/CNRS/Université
Supplementary Materials and Methods
 Supplementary Materials and Methods Whole Mount X-Gal Staining Whole tissues were collected, rinsed with PBS and fixed with 4% PFA. Tissues were then rinsed in rinse buffer (100 mm Sodium Phosphate ph
Supplementary Materials and Methods Whole Mount X-Gal Staining Whole tissues were collected, rinsed with PBS and fixed with 4% PFA. Tissues were then rinsed in rinse buffer (100 mm Sodium Phosphate ph
Mitochondrial DNA Isolation Kit
 Mitochondrial DNA Isolation Kit Catalog Number KA0895 50 assays Version: 01 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Background... 3 General Information... 4 Materials
Mitochondrial DNA Isolation Kit Catalog Number KA0895 50 assays Version: 01 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Background... 3 General Information... 4 Materials
PRODUCT: RNAzol BD for Blood May 2014 Catalog No: RB 192 Storage: Store at room temperature
 PRODUCT: RNAzol BD for Blood May 2014 Catalog No: RB 192 Storage: Store at room temperature PRODUCT DESCRIPTION. RNAzol BD is a reagent for isolation of total RNA from whole blood, plasma or serum of human
PRODUCT: RNAzol BD for Blood May 2014 Catalog No: RB 192 Storage: Store at room temperature PRODUCT DESCRIPTION. RNAzol BD is a reagent for isolation of total RNA from whole blood, plasma or serum of human
GENERAL SUMMARY Corpus luteum is a transient endocrine structure formed from the ruptured ovarian follicle. Its main function is to secrete P 4, a pro
 Corpus luteum is a transient endocrine structure formed from the ruptured ovarian follicle. Its main function is to secrete P 4, a pro-gestational hormone, essential for establishment and maintenance of
Corpus luteum is a transient endocrine structure formed from the ruptured ovarian follicle. Its main function is to secrete P 4, a pro-gestational hormone, essential for establishment and maintenance of
human Total Cathepsin B Catalog Number: DY2176
 human Total Cathepsin B Catalog Number: DY2176 This DuoSet ELISA Development kit contains the basic components required for the development of sandwich ELISAs to measure natural and recombinant human Total
human Total Cathepsin B Catalog Number: DY2176 This DuoSet ELISA Development kit contains the basic components required for the development of sandwich ELISAs to measure natural and recombinant human Total
CHARACTERISATION OF THE DEVELOPMENT AND HORMONAL REGULATION OF THE OVARIAN LYMPHATIC VASCULATURE
 CHARACTERISATION OF THE DEVELOPMENT AND HORMONAL REGULATION OF THE OVARIAN LYMPHATIC VASCULATURE Hannah Mary Brown Robinson Institute School of Paediatrics and Reproductive Health Research Centre for Reproductive
CHARACTERISATION OF THE DEVELOPMENT AND HORMONAL REGULATION OF THE OVARIAN LYMPHATIC VASCULATURE Hannah Mary Brown Robinson Institute School of Paediatrics and Reproductive Health Research Centre for Reproductive
Human Urokinase / PLAU / UPA ELISA Pair Set
 Human Urokinase / PLAU / UPA ELISA Pair Set Catalog Number : SEK10815 To achieve the best assay results, this manual must be read carefully before using this product and the assay is run as summarized
Human Urokinase / PLAU / UPA ELISA Pair Set Catalog Number : SEK10815 To achieve the best assay results, this manual must be read carefully before using this product and the assay is run as summarized
Supporting Information
 Supporting Information Franco et al. 10.1073/pnas.1015557108 SI Materials and Methods Drug Administration. PD352901 was dissolved in 0.5% (wt/vol) hydroxyl-propyl-methylcellulose, 0.2% (vol/vol) Tween
Supporting Information Franco et al. 10.1073/pnas.1015557108 SI Materials and Methods Drug Administration. PD352901 was dissolved in 0.5% (wt/vol) hydroxyl-propyl-methylcellulose, 0.2% (vol/vol) Tween
Supplemental Experimental Procedures
 Cell Stem Cell, Volume 2 Supplemental Data A Temporal Switch from Notch to Wnt Signaling in Muscle Stem Cells Is Necessary for Normal Adult Myogenesis Andrew S. Brack, Irina M. Conboy, Michael J. Conboy,
Cell Stem Cell, Volume 2 Supplemental Data A Temporal Switch from Notch to Wnt Signaling in Muscle Stem Cells Is Necessary for Normal Adult Myogenesis Andrew S. Brack, Irina M. Conboy, Michael J. Conboy,
Tissue repair. (3&4 of 4)
 Tissue repair (3&4 of 4) What will we discuss today: Regeneration in tissue repair Scar formation Cutaneous wound healing Pathologic aspects of repair Regeneration in tissue repair Labile tissues rapid
Tissue repair (3&4 of 4) What will we discuss today: Regeneration in tissue repair Scar formation Cutaneous wound healing Pathologic aspects of repair Regeneration in tissue repair Labile tissues rapid
A comparison of the effects of estrus cow. nuclear maturation of bovine oocytes
 A comparison of the effects of estrus cow serum and fetal calf serum on in vitro nuclear maturation of bovine oocytes J Spiropoulos, SE Long University of Bristol, School of Veterinary Science, Department
A comparison of the effects of estrus cow serum and fetal calf serum on in vitro nuclear maturation of bovine oocytes J Spiropoulos, SE Long University of Bristol, School of Veterinary Science, Department
Human Immunodeficiency Virus type 1 (HIV-1) p24 / Capsid Protein p24 ELISA Pair Set
 Human Immunodeficiency Virus type 1 (HIV-1) p24 / Capsid Protein p24 ELISA Pair Set Catalog Number : SEK11695 To achieve the best assay results, this manual must be read carefully before using this product
Human Immunodeficiency Virus type 1 (HIV-1) p24 / Capsid Protein p24 ELISA Pair Set Catalog Number : SEK11695 To achieve the best assay results, this manual must be read carefully before using this product
Analysis of small RNAs from Drosophila Schneider cells using the Small RNA assay on the Agilent 2100 bioanalyzer. Application Note
 Analysis of small RNAs from Drosophila Schneider cells using the Small RNA assay on the Agilent 2100 bioanalyzer Application Note Odile Sismeiro, Jean-Yves Coppée, Christophe Antoniewski, and Hélène Thomassin
Analysis of small RNAs from Drosophila Schneider cells using the Small RNA assay on the Agilent 2100 bioanalyzer Application Note Odile Sismeiro, Jean-Yves Coppée, Christophe Antoniewski, and Hélène Thomassin
Nature Structural & Molecular Biology: doi: /nsmb Supplementary Figure 1. Generation and validation of mtef4-knockout mice.
 Supplementary Figure 1 Generation and validation of mtef4-knockout mice. (a) Alignment of EF4 (E. coli) with mouse, yeast and human EF4. (b) Domain structures of mouse mtef4 compared to those of EF4 (E.
Supplementary Figure 1 Generation and validation of mtef4-knockout mice. (a) Alignment of EF4 (E. coli) with mouse, yeast and human EF4. (b) Domain structures of mouse mtef4 compared to those of EF4 (E.
We are IntechOpen, the world s leading publisher of Open Access books Built by scientists, for scientists. International authors and editors
 We are IntechOpen, the world s leading publisher of Open Access books Built by scientists, for scientists 3,700 108,500 1.7 M Open access books available International authors and editors Downloads Our
We are IntechOpen, the world s leading publisher of Open Access books Built by scientists, for scientists 3,700 108,500 1.7 M Open access books available International authors and editors Downloads Our
Single Cell Quantitative Polymer Chain Reaction (sc-qpcr)
 Single Cell Quantitative Polymer Chain Reaction (sc-qpcr) Analyzing gene expression profiles from a bulk population of cells provides an average profile which may obscure important biological differences
Single Cell Quantitative Polymer Chain Reaction (sc-qpcr) Analyzing gene expression profiles from a bulk population of cells provides an average profile which may obscure important biological differences
FSH (Human) ELISA Kit
 FSH (Human) ELISA Kit Catalog Number KA0213 96 assays Version: 03 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle of the Assay...
FSH (Human) ELISA Kit Catalog Number KA0213 96 assays Version: 03 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle of the Assay...
9.4 Regulating the Reproductive System
 9.4 Regulating the Reproductive System The Reproductive System to unite a single reproductive cell from a female with a single reproductive cell from a male Both male and female reproductive systems include
9.4 Regulating the Reproductive System The Reproductive System to unite a single reproductive cell from a female with a single reproductive cell from a male Both male and female reproductive systems include
