Detailed Time Course of Ovum Transport in the Rhesus Monkey (Macaca mulatta)
|
|
|
- Cornelia Sherman
- 6 years ago
- Views:
Transcription
1 BIOLOGY OF REPRODUCTION 13, Detailed Time Course of Ovum Transport in the Rhesus Monkey (Macaca mulatta) CARLTON A. EDDY, RAUL G. GARCIA, DUANE C. KRAEMER2 and CARL J. PAUERSTEIN3 Departments of Obstetrics and Gynecology and Physiology Center for Research and Training in Reproductive Biology4 The University of Texas Health Science Center at San Antonio 7703 Floyd Curl Drive, San Antonio, Texas ABSTRACT The time course of ovum transport was studied in a colony of untreated spontaneously cycling female rhesus monkeys. Plasma estrogen values were determined daily by radioimmunoassay starting on Day 8 of the menstrual cycle to provide predictive indication of impending ovulation. After detection of a well-defined preovulatory increase in estrogen production (levels in excess of pg/mi) serial laparoscopies were initiated and performed every 12 to 24 h to monitor ovarian follicular development and to visually confirm the occurrence of ovulation. Laparotomies were performed approximately 24, 48, or 72 h post-ovulation and the reproductive tracts were flushed in Situ to recover ova. The ampulla, isthmus and uterus were flushed separately, allowing the position of the ova within the tract to be determined. Ova were consistently recovered from the tubal ampulla at 24 and 48 h postovulation. By 72 h ova were recovered from both the tube and the uterus. Our findings, based upon accurately timed ovulation, indicate that normal tubal ovum transport time for the untreated, spontaneously cycling rhesus monkey is approximately 72 h. INTRODUCTION Knowledge of the normal time course of tubal ovum transport is basic to an understanding of tubal function and is a prerequisite for the development of future contraceptive techniques operative at the tubal level. Because of its anatomic, physiologic, and endocrinologic similarity to man, the nonhuman primate constitutes a potentially valuable model in which to investigate tubal transport phenomena. Although the detailed time course of ovum transport has been determined for various laboratory species, most notably the rabbit, similar information for man and nonhuman primates is lacking or incomplete. This lack of knowledge reflects the inherent complexities associated with the use of the primate in ovum transport studies, chief among which is the difficulty in accurately determining the time of Accepted July 16, Rockefeller Foundation Postdoctoral Fellow in Reproductive Biology. 2 Present Address: College of Veterinary Medicine, Department of Veterinary Physiology and Pharmacology, Texas A & M University, College Station, Texas Recipient of NICHHD Research Career Development Award K04 HD Supported by the Rockefeller Foundation. ovulation. A variety of indirect criteria has been evolved to determine the time of ovulation in the nonhuman primate. These include rectal palpation of the ovaries (Uartman, 1932;Mahoney, 1970), sequential changes in vaginal cytology (Mahoney, 1970; Mauro et al., 1970; Uutchinson, 1970) and in cervical mucus composition (Mahoney, 1970; Ovadia et al., 1971), and sex skin changes (Uagino and Goldzieher, 1970). However, extensive variability between and within animals combined with equivocal changes in the above parameters renders temporal correlation of such events with ovulation difficult. Periovulatory changes in gross ovarian follicular morphology observed at laparotomy have been described in the rhesus monkey (Betteridge et al., 1970) and have obviated much of the difficulty in diagnosing ovulation in this species. When such observations are made serially, it becomes possible to both diagnose and time the occurrence of ovulation with accuracy. Serial laparoscopy has been used to time ovulation in the primate for use in ovum transport studies (Jainudeen and Hafez, 1973). These investigators encountered difficulty in recovering natural ova, and speculated 363
2 364 EDDY ET AL. that the low recovery reflected interference with the mechanism of ovum pickup due to frequent and repeated laparoscopic manipulation of the reproductive tract. Thus, attempts to recover natural ova were decreased and emphasis were shifted to the recovery of surrogate ova (Sephadex beads and rabbit ova). Thus the time course of tubal ovum transport of natural primate ova remains largely undefined. Because the primate ovulates spontaneously, an excessive number of serial laparoscopic observations may be required if laparoscopy is initiated prematurely. In the rhesus monkey the preovulatory surge of LH is preceded by a gradual increase in circulating estrogen of approximately three days duration (Uotchkiss et al., 1971). Estrogen levels then rise sharply, peak 9-15 h prior to the LU surge, then decline prior to ovulation. Ovulation follows the estrogen peak by approximately 37 h (Weick et al., 1973). Measurement of a characteristic preovulatory peak in circulating estrogen may be used to predict impending ovulation and thereby decrease the number of laparoscopies required to verify ovulation. The present study was undertaken to define the detailed time course of tubal ovum transport in the spontaneously ovulating rhesus monkey using serial laparoscopy, initiated following detection of a preovulatory estrogen peak, to time ovulation. in place of methanol for precipitation of plasma proteins. The estrogen antibody was prepared and made available by Dr. Burton V. Caldwell. Laparoscopy Following the detection of a preovulatory peak in estrogen (levels in excess of pg/mi), serial Iaparoscopies were initiated in order to monitor sequential changes in follicular morphology. Under Sernylan anesthesia (3-5 mg/kg body weight) the MATERIALS AND METHODS Sexually mature female rhesus monkeys (Macaca mulatta) weighing kg were individually caged under controlled conditions of 75#{176} F and 50 percent relative humidity and exposed to a 14 h fluorescent light photo-period. Commercially prepared pelleted monkey chow was supplemented twice weekly with fresh fruit and vegetables. Tap water was available ad lib. Vaginal swabs were taken each morning and examined for menses. The first day of menstrual bleeding was designated Day 1 of the cycle. Each animal was allowed to establish regular, spontaneous, cyclic activity prior to use. Daily blood samples (2-3 ml) were drawn each morning beginning on Day 8 by femoral puncture with a heparinized syringe following tranquilization with phencyclidine hydrochloride (Sernylan, Parke-Davis, mg/kg body weight, i.m.). The heparinized blood was centrifuged and the plasma assayed for estrogen. Samples were drawn through Day 16 or until a preovulatory estrogen peak was detected. Estrogen Assay Total plasma estrogen levels were determined by radioimmunoassay as described by Wu et al. (1973) with minor modification. Absolute ethanol was used FIG. 1. In situ flushing technique for the recovery of ova from various portions of the Macaca mulatta reproductive tract. (a) Ampullary flush; (b) Isthmic flush; (c) Uterine flush. (Insert: details of fenestrations applied to tip of intracatheter cannula.) )
3 OVUM TRANSPORT IN THE RHESUS MONKEY 365 C a, 0 0 U E 100 a- (2) (6) : ) 6 Days (8) (8) From Ovulation FIG. 2. Mean plasma estrogen pattern of nine ovulatory menstrual cycles in which ova were recovered in Macaca mulatta. Values are normalized to the day of ovulation. Vertical lines about each point represent the standard error of the mean. Numbers in parentheses represent the number of observations. Hatched area defines the average interval of time during which ovulation occurred (range ± 6-15 h). possible. The isthmus, excluding the intramural position and a 3-5 mm proximal segment, was flushed with 3-5 ml of saline, and the effluent collected through the ampulla. To flush the uterus, an 18 gauge polyethylene intracatheter fenestrated at the tip was introduced through the uterine fundus into the uterine cavity. A second intracatheter was inserted through the ventral aspect of the uterus into the uterine cavity at a point several millimeters above the internal cervical Os. Syringes were attached to each intracatheter and successive 6 ml quantities of saline were flushed through the uterine cavity from alternating syringes. At each flushing the effluent was collected in the corresponding empty syringe. The use of a cervical clamp prevented transcervical leakage of fluid during uterine flushing. In the 72 h post ovulation group, the uterus was flushed first, with the TUJ occluded, to avoid artifactual dislocation of ova located in the proximal isthmus into the uterus during tubal flushing. Tubal and uterine effluents were examined for the presence of ova with a binocular dissecting microscope. RESULTS entire surface of both ovaries was visualized using a pediatric laparoscope. The time of ovulation was arbitrarily designated as the midpoint between two successive lararoscopic observations, the former preovulatory and the latter postovulatory. Using this rationale, the time of ovulation was accurately defined within a range of ± 6-15 h. Ovum Recovery Midventral laparotom ies were performed under Sernylan anesthesia.3-5 mg/kg) 24, 48 or 72 h post ovulation and the reproductive tracts flushed in situ to recover the unfertilized ova (Fig. 1). In the majority of cases a polyethylene cannula (P.E. 60, id in., o.d in.) was inserted into the ampulla to a point just beyond the infundibulum and ligated in place. In the remaining cases, the oviduct was flushed directly into a receptacle (Mastroianni and Rosseau, 1965). The ampullary isthmic junction (AIJ) was located and a short, 30 gauge needle mounted on a syringe filled with normal saline was inserted into the lumen of the ampulla at the level of the AlJ or several millimeters to the ovarian side of it. The tubo-uterine junction (TUJ) was digitally occluded and 3-10 ml of saline was flushed through the ampulla. The needle was then introduced into the isthmus as close to the TUJ as Timing of Ovulation Figure 2 shows the periovulatory peripheral plasma estrogen levels of nine ovulatory menstrual cycles in which ova were recovered following in situ flushings of the reproductive tract. In each cycle a well defined preovulatory peak of estrogen in excess of 200 pg/mi (range pg/mi) was observed h prior to ovulation. Serial laparoscopy was initiated h after detection of each peak and was repeated every h until post ovulatory changes were observed. In eight cycles two laparoscopies were sufficient to confirm and time ovulation. In the remaining cycle, two additional laparoscopies were required. During the initial observation the ovary containing the Graafian follicle was easily distinguished from the contralateral nonovulatory ovary. The former was much larger, the entire ovary assuming a fluid-filled, bluish-pink opaque appearance which could occasionally be localized to a specific area which subsequently TABLE 1. Ovum recovery rates following in situ flushing of the reproductive tract of Macaca mulatta. Time ovulation after (hrs) Number flushings of Number of ova recovered Percent recovery Total
4 366 EDDY ET AL. TABLE 2. Results of in situ flushing of the reproductive tract following timed ovulation in Macaca rnulatta. Animal number Time post ovulation Tubal flush Uterine flush Location ovum of ± Ampulla ± Ampulla ± Ampulla ± Ampulla ± Ampulla ± Ampulla All 72 ± Uterus A13 72 ± Tube A17 72 ± Uterus proved to be the site of ovulation. In agreement with Betteridge et al. (1970) we uniformly failed to observe persistent, well defined, protuberant Graafian follicles with preovulatory stigmata (Johnson et al., 1968). Instead, final follicular maturation and ovulation, visually observed as the transition from poorly defined Graafian follicle to protuberant, hemorrhagic, raw, post ovulatory corpus luteum (CL), generally encompassed a period of only hours. Although variety in periovulatory follicular morphology was encountered, the diagnosis of ovulation was always unequivocal, due primarily to the opportunity to view progressive changes, before and after ovulation. Ovum Transport A total of 24 in situ flushings were performed following laparoscopic timing of ovulation and resulted in recovery of nine ova (Table 1) yielding an overall recovery rate of 37.5 percent. In contrast, recovery rates for individual time groups ranged from 75 percent to 25 percent. The location of ova recovered through in situ flushing of the reproductive tract at various times post ovulation is summarized in Table 2. Ova were consistently recovered from the ampulla at both 24 and 48 h after ovulation, in all cases still in cumulus (Figs. 3, 4). By 72 h post ovulation, ova were entering the uterus. Two ova were recovered from this site whereas a third was flushed from the tube. In the latter recovery, the precise location of the ovum within the tube is not known with certainty. Uterine ova were still surrounded by cumulus cells which persisted in much smaller numbers than the cumulus mass seen at 24 and 48 h (Fig. 5). This same sparse cellular investment characterized the single ovum recovered from the tube at 72 h (Fig. 6). On the basis of cumulus investment, it would appear that this FIG. 3. Macaca mulatta ovum recovered 24 h after ovulation. This ovum was surrounded by a cumulus mass when flushed from the ampulla. FIG. 4. Macaca mulatta ovum recovered 48 h after ovulation. This ovum was located in the ampulla and still retained a dense cellular investment of cumulus cells.
5 OVUM TRANSPORT IN THE RHESUS MONKEY 367 FIG. 5. Macaca mulatta ovum recovered from the uterus 72 h after ovulation. Unlike the tubal ova recovered from the anipulla 24 and 48 h after ovulation, this uterine ovum was surrounded by comparatively few cumulus cells. ovum had traveled beyond the AIJ and was located in the isthmus, if denudation occurs during passage through the isthmus. DISCUSSION Discussion of Limitations of Methods As amplified below this report represents, to the best of our knowledge, the first description of the detailed time-course of tubal transport of ova in the rhesus monkey. Before discussing our conclusions, it is pertinent to review possible limitations imposed upon the interpretation of our data. First it may be argued that the administration of transquilizing doses of Sernylan to FIG. 6. Macaca mulatta ovum recovered from the tube 72 h after ovulation. Note the same sparse cellular investment as that seen surrounding the ovum in Fig. 5. facilitate daily blood collection may have modified the process of ovum transport. However, use of Sernylan in Macaca mulatta in conjunction with serial blood drawing has been reported to restore normal fertility in females previously experiencing lowered fertility due to blood collection (Hamner, 1974). The use of Ketamine in conjunction with serial blood drawing has also been shown to have no effect on normal follicular maturation and ovulation in Macaca mulatta (Channing, 1975). Thus, we do not think that the blood sampling interfered with ovulation and ovum transport. Second, one must confront the possibility that the serial laparoscopic examinations, so essential to the accurate determination of ovulation, might have interfered with ovum transport. Jainudeen and Hafez (1973) speculated that excessive manipulation of the Macaca fascicularis genital tract during laparoscopic examination might have interfered with ovum pickup. Although the number of laparoscopies required to time ovulation was not specified in their study, the arbitrary initiation of serial laparoscopy on Day 11 and its continuation until ovulation was confirmed may be expected to have at least sometimes necessitated an excessive number of laparoscopies, which might have contributed to the recovery of only three natural ova. In the present study daily measurement of plasma estrogen levels furnished accurate prospective indication of impending ovulation. As a result, the number of laparoscopics performed was minimized. This fact notwithstanding, the overall recovery rate was only 37.5 percent. Obviously some of the failures to recover ova may reflect limitations of the recovery technique. However, during one recovery attempt an ovum was aspirated from the cul-de-sac 72 ± 12 h post ovulation, prior to any attempt to flush the tract itself. Since only two laparoscopies, 24 h apart, were required to time ovulation in this case, this finding may reflect normal variations in the efficiency of the ovum pick up mechanism. This possibility is strengthened by the successful recovery of a uterine ovum at 72 h post ovulation despite four antecedent laparoscopies. Third, the recovery rates varied with the time of examination. Ova were recovered in 3 of 4 attempts at 24 h (all in the ampulla), in 3 of 8 attempts at 48 h (all in the ampulla) and in 3 of 12 attempts at 72 h (2 from the uterus and one from the tube). Failure to recover ova in 5
6 368 EDDY ET AL. of 8 attempts to flush the isthmus may reflect technical inefficiency in flushing the isthmus. Thus we might have failed to recover those ova which passed into the isthmus prior to 48 h. By the same reasoning, failure to recover more ova from the uterus at 72 h postovulation (9 out of 12) may reflect their continued presence in the isthmus. Recovery rates of uterine ova significantly lower than those for tubal ova have also been repeatedly remarked upon in reports of investigations of ovum transport in the rabbit. Finally, the described time-course is synthesized by noting the location of recovered ova in different animals at each time interval, and thus is intrinsically inferior to tracking the complete transport of individual ova through the same animal. Discussion and Results In the majority of mammalian species studied, tubal transport of the ovum into the uterus requires 3 to 4 days. The few reports of tubal ovum transport in the spontaneously ovulating primate have generally dealt with the overall time of transport from ovary to uterus, and were not based on accurate knowledge of the time of ovulation. Hartman (1944) used rectal palpation to time ovulation and determined tubal ovum transport in the rhesus to require somewhat over three days based on recovery of ova from the uterus. Mastroianni et al. (1967) using ferning of cervical mucus to time ovulation noted the location of ova in the reproductive tract of spontaneously ovulating rhesus monkeys 24 h after presumed ovulation. All ova recovered were in the tube. Marston et al. (1969) using morphologic dating of the CL at the time of flushing of the entire tube noted the persistence of ova within the rhesus oviduct 4 to 7 days postovulation. Ova were also recovered from the uterus during this interval. In none of these studies was the detailed time course of ovum transport investigated. Jainudeen and Hafez (1973) attempted to provide such a detailed description in Macaca fascicularis by excising and segmentally flushing oviducts at 24 h intervals following lararoscopically timed ovulation. Unfortunately early in the experiment they abandoned the attempt to recover natural ova and substituted surrogate ova. Their results therefore are largely predicted upon the movement of artificial ova which were not proved to accurately mimic the transport of natural ova. They noted transport through the distal two-thirds of the tube within 48 h, retention at the AIJ for an additional 48 h, followed by transport through the proximal third of the tube in an additional 48 h. These results are in contrast to the time-course of ovum transport in the rabbit, where newly ovulated ova arrive at the ampullary-isthmic junction within minutes and are delayed at that location for some h, before entering the isthmus. The journey through the isthmus to the uterus takes about 36 h, so that entry into the uterus usually takes place 66 to 72 h after injection of human chorionic gonadotropin. Within the limits discussed above we may safely conclude from our data that at least some rhesus ova are still in the ampulla 48 ± 15 h after ovulation and that some ova arrive in the uterus within 72 ± 12 h after ovulation. Thus, although the overall time-course of ovum transport in Macaca mulatta is apparently not greatly different from that in the rabbit, the details of transit are. Our data suggest that rhesus ova remain in the ampulla much longer and traverse the isthmus much more quickly than do rabbit ova. In addition the cumulus cells persist much longer around unfertilized Macaca mulatta ova than around unfertilized rabbit ova. Our data are in good agreement with those obtained in our own laboratories and by Croxatto et al. (1972) concerning the timecourse of egg transport in women, and support the contention that the rhesus is a good model for studies of human ovum transport. ACKNOWLEDGMENT The authors gratefully acknowledge the excellent technical assistance of Thomas C. Turner and Elizabeth Menchaca, Department of Obstetrics and Gynecology. This work is supported by the Agency for International Development, Subcontract PARFR-56 from the University of Minnesota. REFERENCES Betteridge, K. J., Kelly, W. A., and Marston, J. H. (1970). Morphology of the rhesus monkey ovary near the time of ovulation. J. Reprod. Fertil. 22, Channing, C. P. (1975). Personal communication. Croxatto, H. B., Fuentealba, B., Diaz, S., Pastene, L., and Tatum, H. J. (1972). A simple non-surgical technique to obtain unimplanted eggs from human uteri. Am. J. Obstet. Gynecol. 112, Hagino, N. and Goldzieher, J. W. (1970). Regulation of gonadotrophin release by the corpus luteum in the baboon. Endocrinology 87, Hamner, C. (1974). Personal communication. Hartman, C. G. (1932). Pelvic (rectal) palpation of the female monkey, with special reference to the ascertainment of ovulation time. Am. J. Obstet.
7 OVUM TRANSPORT IN THE RHESUS MONKEY 369 Gynecol. 26, Hartman, C. G. (1944). Recovery of primate eggs and embryos. Methods and data on the time of ovulation. West. J. Surg. Obstet. Gynecol. 52, Hotchkiss, J., Atkinson, L. W., and Knobil, E. (1971). Time course of serum estrogen and luteinizing hormone concentrations during the menstrual cycle of the rhesus monkey. Endocrinology 89, Hutchinson, T. C. (1970). Vaginal cytology and reproduction in the squirrel monkey (Saimiri sciureus). Folia Primat. 12, Jainudeen, M. R. and Hafez, E. S. E. (1973). Egg transport in the macaque (Macaca fascicularis). Biol. Reprod. 9, Johansson, E. D. B., Neill, J. D., and Knobil, E. (1968). Pen-ovulatory progesterone concentration in the peripheral plasma of the rhesus monkey with a methodologic note on the detection of ovulation. Endocrinology 82, Mahoney, C. J. (1970). A study of the menstrual cycle in Macaca irus with special reference to the detection of ovulation. J. Reprod. Fertil. 21, Marston, J. H., Kelly, W. A., and Eckstein, P. (1969). Effect of an intrauterine device on gamete transport and fertilization in the rhesus monkey. J. Reprod. Fertil. 19, Mastroianni, L. and Rosseau, C. H. (1965). Influence of the intrauterine coil on ovum transport and sperm distribution in the monkey. Am. J. Obstet. Gynecol. 93, Mastroianni, L., Suzuki, S., Manabe, V., and Watson, F. (1967). Further observations on the influence of the intrauterine device on ovum and sperm distribution in the monkey. Am. J. Obstet. Gynecol. 99, Mauro, J., Serrone, D., Somsin, P., and Stein, A. A. (1970). Cyclic vaginal cytologic patterns in the Macaca mulatta. Acta Cytol. 14, Ovadia, J., McArthur, J. W., Smith, 0. W., and Bashir-Farahmand, J. (1971). An individualized technique for inducing ovulation in the bonnet monkey, Macaca radiata..j. Reprod. Fertil. 27, Weick, R. F., Dierschke, D. J., Karsch, F. J., Hotchkiss, J., and Knobil, E. (1967). Periovulatory time course of the circulating gonadotropin and ovarian hormones in the rhesus monkey. The Endocrine Society Meetings (Abstr.), July, 1973, Chicago. Wu, C-H., Lundy, L.., and Lee, S. G. (1973). A rapid radioimmunoassay for plasma estrogen. Am. J. Obstet. Gynecol. 115,
(Received 5th July 1968)
 EFFECT OF AN INTRA-UTERINE DEVICE ON CONCEPTION AND OVULATION IN THE RHESUS MONKEY W. A. KELLY, J. H. MARSTON and P. ECKSTEIN Department of Anatomy, Medical School, Birmingham 15 (Received 5th July 1968)
EFFECT OF AN INTRA-UTERINE DEVICE ON CONCEPTION AND OVULATION IN THE RHESUS MONKEY W. A. KELLY, J. H. MARSTON and P. ECKSTEIN Department of Anatomy, Medical School, Birmingham 15 (Received 5th July 1968)
IN VITRO FERTILIZATION OF RABBIT EGGS IN OVIDUCT SECRETIONS FROM DIFFERENT DAYS BEFORE AND AFTER OVULATION*
 FERTILITY AND STERILITY Copyright~ 1975 The American Fertility Society Vol. 26, No.7, July 1975 Printed in U.SA. IN VITRO FERTILIZATION OF RABBIT EGGS IN OVIDUCT SECRETIONS FROM DIFFERENT DAYS BEFORE AND
FERTILITY AND STERILITY Copyright~ 1975 The American Fertility Society Vol. 26, No.7, July 1975 Printed in U.SA. IN VITRO FERTILIZATION OF RABBIT EGGS IN OVIDUCT SECRETIONS FROM DIFFERENT DAYS BEFORE AND
STUDIES OF THE HUMAN UNFERTILIZED TUBAL OVUM*t
 FERTILITY AND STERILITY Copyright @ 1973 by The Williams & Wilkins Co. Vol. 24, No.8, August 1973 Printed in U.S.A. STUDIES OF THE HUMAN UNFERTILIZED TUBAL OVUM*t C. NORIEGA, M.D., AND C. OBERTI, M.D.
FERTILITY AND STERILITY Copyright @ 1973 by The Williams & Wilkins Co. Vol. 24, No.8, August 1973 Printed in U.S.A. STUDIES OF THE HUMAN UNFERTILIZED TUBAL OVUM*t C. NORIEGA, M.D., AND C. OBERTI, M.D.
Female Reproductive System
 Female Reproductive System (Part A-1) Module 10 -Chapter 12 Overview Female reproductive organs Ovaries Fallopian tubes Uterus and vagina Mammary glands Menstrual cycle Pregnancy Labor and childbirth Menopause
Female Reproductive System (Part A-1) Module 10 -Chapter 12 Overview Female reproductive organs Ovaries Fallopian tubes Uterus and vagina Mammary glands Menstrual cycle Pregnancy Labor and childbirth Menopause
10.7 The Reproductive Hormones
 10.7 The Reproductive Hormones December 10, 2013. Website survey?? QUESTION: Who is more complicated: men or women? The Female Reproductive System ovaries: produce gametes (eggs) produce estrogen (steroid
10.7 The Reproductive Hormones December 10, 2013. Website survey?? QUESTION: Who is more complicated: men or women? The Female Reproductive System ovaries: produce gametes (eggs) produce estrogen (steroid
OOCYTE RECOVERY FROM THE HUMAN OV ARY*
 FERTILITY AND STERILITY Copyright by The Williams & Wilkins Co. Vol. 23. No. 10. October 1972 Printed in U.S.A. OOCYTE RECOVERY FROM THE HUMAN OV ARY* LARRY L. MORGENSTERN, M.D.,t AND PIERRE SOUPART, M.D.,
FERTILITY AND STERILITY Copyright by The Williams & Wilkins Co. Vol. 23. No. 10. October 1972 Printed in U.S.A. OOCYTE RECOVERY FROM THE HUMAN OV ARY* LARRY L. MORGENSTERN, M.D.,t AND PIERRE SOUPART, M.D.,
egg or cumulus clot and in another seventeen
 MORPHOLOGY OF THE RHESUS MONKEY OVARY NEAR THE TIME OF OVULATION. J. BETTERIDGE,* W. A. KELLYt and J. H. MARSTONJ Department of Anatomy, The Medical School, Birmingham 15 (Received 25th July 1969) Summary.
MORPHOLOGY OF THE RHESUS MONKEY OVARY NEAR THE TIME OF OVULATION. J. BETTERIDGE,* W. A. KELLYt and J. H. MARSTONJ Department of Anatomy, The Medical School, Birmingham 15 (Received 25th July 1969) Summary.
different ratios of PMSG and HCG on the occurrence of follicular haemorrhage THE induction of ovulation with PMSG and HCG in the rat has been studied
 Q. Jl exp. Physiol. (1968) 53, 129-135 THE INDUCTION OF OVULATION IN IMMATURE RATS TREATED WITH PREGNANT MARE'S SERUM GONADOTROPHIN AND HUMAN CHORIONIC GONADOTROPHIN. By S. F. LUNN and E. T. BELL. From
Q. Jl exp. Physiol. (1968) 53, 129-135 THE INDUCTION OF OVULATION IN IMMATURE RATS TREATED WITH PREGNANT MARE'S SERUM GONADOTROPHIN AND HUMAN CHORIONIC GONADOTROPHIN. By S. F. LUNN and E. T. BELL. From
Sperm Survival in Women. Motile Sperm in the Fundus and Tubes of Surgical Cases
 Sperm Survival in Women Motile Sperm in the Fundus and Tubes of Surgical Cases Boris B. Rubenstein, M.D., Ph.D.; Hermann Strauss, M.D.; Maurice L. Lazarus, M.D., and Henry Hankin, M.D. THE DURATION of
Sperm Survival in Women Motile Sperm in the Fundus and Tubes of Surgical Cases Boris B. Rubenstein, M.D., Ph.D.; Hermann Strauss, M.D.; Maurice L. Lazarus, M.D., and Henry Hankin, M.D. THE DURATION of
Female Reproductive System. Lesson 10
 Female Reproductive System Lesson 10 Learning Goals 1. What are the five hormones involved in the female reproductive system? 2. Understand the four phases of the menstrual cycle. Human Reproductive System
Female Reproductive System Lesson 10 Learning Goals 1. What are the five hormones involved in the female reproductive system? 2. Understand the four phases of the menstrual cycle. Human Reproductive System
(Received 8th October 1973)
 THE INFLUENCE OF A CANNULA IN THE RABBIT OVIDUCT II. EFFECT ON EMBRYO SURVIVAL M. H. SLOAN, S. L. COLEY and A. D. JOHNSON Animal Science Department, Livestock-Poultry Building, University of Georgia, Athens,
THE INFLUENCE OF A CANNULA IN THE RABBIT OVIDUCT II. EFFECT ON EMBRYO SURVIVAL M. H. SLOAN, S. L. COLEY and A. D. JOHNSON Animal Science Department, Livestock-Poultry Building, University of Georgia, Athens,
CYCLIC MOUSE. and NEENA B. SCHWARTZ INTRODUCTION
 TIMING OF LH RELEASE AND OVULATION IN THE CYCLIC MOUSE AUDREY S. BINGEL and NEENA B. SCHWARTZ Department of Physiology, University of Illinois College of Medicine, Chicago, Illinois, U.S.A. (Received 1st
TIMING OF LH RELEASE AND OVULATION IN THE CYCLIC MOUSE AUDREY S. BINGEL and NEENA B. SCHWARTZ Department of Physiology, University of Illinois College of Medicine, Chicago, Illinois, U.S.A. (Received 1st
CLEAVAGE OF HUMAN OVA IN VITRO*
 FERTILITY AND STERn.1TY Copyright., 1971 by The WiUiams & Wilkins Co. Vol. 22, No.4, April 1971 Printed in U.S.A. CLEAVAGE OF HUMAN OVA IN VITRO* H. M. SEITZ, JR., M.D., G. ROCHA, M.D., B. G. BRACKETI,
FERTILITY AND STERn.1TY Copyright., 1971 by The WiUiams & Wilkins Co. Vol. 22, No.4, April 1971 Printed in U.S.A. CLEAVAGE OF HUMAN OVA IN VITRO* H. M. SEITZ, JR., M.D., G. ROCHA, M.D., B. G. BRACKETI,
MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question.
 MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. 1) Which of the following hormones controls the release of anterior pituitary gonadotropins? A) LH
MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. 1) Which of the following hormones controls the release of anterior pituitary gonadotropins? A) LH
Bio 322 Human Anatomy Objectives for the laboratory exercise Female Reproductive System
 Bio 322 Human Anatomy Objectives for the laboratory exercise Female Reproductive System Required reading before beginning this lab: Saladin, KS: Human Anatomy 5 th ed (2017) Chapter 26 For this lab you
Bio 322 Human Anatomy Objectives for the laboratory exercise Female Reproductive System Required reading before beginning this lab: Saladin, KS: Human Anatomy 5 th ed (2017) Chapter 26 For this lab you
CASE 41. What is the pathophysiologic cause of her amenorrhea? Which cells in the ovary secrete estrogen?
 CASE 41 A 19-year-old woman presents to her gynecologist with complaints of not having had a period for 6 months. She reports having normal periods since menarche at age 12. She denies sexual activity,
CASE 41 A 19-year-old woman presents to her gynecologist with complaints of not having had a period for 6 months. She reports having normal periods since menarche at age 12. She denies sexual activity,
Biology of fertility control. Higher Human Biology
 Biology of fertility control Higher Human Biology Learning Intention Compare fertile periods in females and males What is infertility? Infertility is the inability of a sexually active, non-contracepting
Biology of fertility control Higher Human Biology Learning Intention Compare fertile periods in females and males What is infertility? Infertility is the inability of a sexually active, non-contracepting
LUTEINIZED UNRUPTURED FOLLICLE SYNDROME: A SUBTLE CAUSE OF INFERTILITY*
 FERTILITY AND STERILITY Copyright c 1978 The American Fertility Society Vol. 29, No.3, March 1978 Printed in U.S.A. LUTEINIZED UNRUPTURED FOLLICLE SYNDROME: A SUBTLE CAUSE OF INFERTILITY* JAROSLA V MARIK,
FERTILITY AND STERILITY Copyright c 1978 The American Fertility Society Vol. 29, No.3, March 1978 Printed in U.S.A. LUTEINIZED UNRUPTURED FOLLICLE SYNDROME: A SUBTLE CAUSE OF INFERTILITY* JAROSLA V MARIK,
Reproductive System. Testes. Accessory reproductive organs. gametogenesis hormones. Reproductive tract & Glands
 Reproductive System Testes gametogenesis hormones Accessory reproductive organs Reproductive tract & Glands transport gametes provide nourishment for gametes Hormonal regulation in men Hypothalamus - puberty
Reproductive System Testes gametogenesis hormones Accessory reproductive organs Reproductive tract & Glands transport gametes provide nourishment for gametes Hormonal regulation in men Hypothalamus - puberty
9.4 Regulating the Reproductive System
 9.4 Regulating the Reproductive System The Reproductive System to unite a single reproductive cell from a female with a single reproductive cell from a male Both male and female reproductive systems include
9.4 Regulating the Reproductive System The Reproductive System to unite a single reproductive cell from a female with a single reproductive cell from a male Both male and female reproductive systems include
Reproduction and Development. Female Reproductive System
 Reproduction and Development Female Reproductive System Outcomes 5. Identify the structures in the human female reproductive system and describe their functions. Ovaries, Fallopian tubes, Uterus, Endometrium,
Reproduction and Development Female Reproductive System Outcomes 5. Identify the structures in the human female reproductive system and describe their functions. Ovaries, Fallopian tubes, Uterus, Endometrium,
Reproductive Endocrinology. Isabel Hwang Department of Physiology Faculty of Medicine University of Hong Kong Hong Kong May2007
 Reproductive Endocrinology Isabel Hwang Department of Physiology Faculty of Medicine University of Hong Kong Hong Kong May2007 isabelss@hkucc.hku.hk A 3-hormone chain of command controls reproduction with
Reproductive Endocrinology Isabel Hwang Department of Physiology Faculty of Medicine University of Hong Kong Hong Kong May2007 isabelss@hkucc.hku.hk A 3-hormone chain of command controls reproduction with
Web Activity: Simulation Structures of the Female Reproductive System
 differentiate. The epididymis is a coiled tube found along the outer edge of the testis where the sperm mature. 3. Testosterone is a male sex hormone produced in the interstitial cells of the testes. It
differentiate. The epididymis is a coiled tube found along the outer edge of the testis where the sperm mature. 3. Testosterone is a male sex hormone produced in the interstitial cells of the testes. It
Contralateral tubal-ovarian apposition and fertility in hemiovariectomized primates*
 FERTILITY AND STERILITY Copyright 1984 The American Fertility Society Vol. 42, No.4, October 1984 Printed in U.8A. Contralateral tubal-ovarian apposition and fertility in hemiovariectomized primates* Victoria
FERTILITY AND STERILITY Copyright 1984 The American Fertility Society Vol. 42, No.4, October 1984 Printed in U.8A. Contralateral tubal-ovarian apposition and fertility in hemiovariectomized primates* Victoria
Reproductive Hormones
 Reproductive Hormones Male gonads: testes produce male sex cells! sperm Female gonads: ovaries produce female sex cells! ovum The union of male and female sex cells during fertilization produces a zygote
Reproductive Hormones Male gonads: testes produce male sex cells! sperm Female gonads: ovaries produce female sex cells! ovum The union of male and female sex cells during fertilization produces a zygote
Estradiol Levels Near the Time of Ovulation in the Bonnet Monkey (Macaca radiata)1
 BIOLOGY OF REPRoDucTIoN 11, 237-244 (1974) Estradiol Levels Near the Time of Ovulation in the Bonnet Monkey (Macaca radiata)1 B. LASLEY,2 A. G. HENDRICKX, AND G. H. STABENFELDT California Primate Research
BIOLOGY OF REPRoDucTIoN 11, 237-244 (1974) Estradiol Levels Near the Time of Ovulation in the Bonnet Monkey (Macaca radiata)1 B. LASLEY,2 A. G. HENDRICKX, AND G. H. STABENFELDT California Primate Research
SISTEMA REPRODUCTOR (LA IDEA FIJA) Copyright 2004 Pearson Education, Inc., publishing as Benjamin Cummings
 SISTEMA REPRODUCTOR (LA IDEA FIJA) How male and female reproductive systems differentiate The reproductive organs and how they work How gametes are produced and fertilized Pregnancy, stages of development,
SISTEMA REPRODUCTOR (LA IDEA FIJA) How male and female reproductive systems differentiate The reproductive organs and how they work How gametes are produced and fertilized Pregnancy, stages of development,
Female Reproductive Physiology. Dr Raelia Lew CREI, FRANZCOG, PhD, MMed, MBBS Fertility Specialist, Melbourne IVF
 Female Reproductive Physiology Dr Raelia Lew CREI, FRANZCOG, PhD, MMed, MBBS Fertility Specialist, Melbourne IVF REFERENCE Lew, R, Natural History of ovarian function including assessment of ovarian reserve
Female Reproductive Physiology Dr Raelia Lew CREI, FRANZCOG, PhD, MMed, MBBS Fertility Specialist, Melbourne IVF REFERENCE Lew, R, Natural History of ovarian function including assessment of ovarian reserve
REPRODUCCIÓN. La idea fija. Copyright 2004 Pearson Education, Inc., publishing as Benjamin Cummings
 REPRODUCCIÓN La idea fija How male and female reproductive systems differentiate The reproductive organs and how they work How gametes are produced and fertilized Pregnancy, stages of development, birth
REPRODUCCIÓN La idea fija How male and female reproductive systems differentiate The reproductive organs and how they work How gametes are produced and fertilized Pregnancy, stages of development, birth
Outline. Male Reproductive System Testes and Sperm Hormonal Regulation
 Outline Male Reproductive System Testes and Sperm Hormonal Regulation Female Reproductive System Genital Tract Hormonal Levels Uterine Cycle Fertilization and Pregnancy Control of Reproduction Infertility
Outline Male Reproductive System Testes and Sperm Hormonal Regulation Female Reproductive System Genital Tract Hormonal Levels Uterine Cycle Fertilization and Pregnancy Control of Reproduction Infertility
TUBAL PLASTIC SURGERY is an accepted form of therapy in the treatment
 Tubal Plastic Surgery ADNAN MROUEH, M.D., ROBERT H. GLASS, M.D., and C. LEE BUXTON, M.D. TUBAL PLASTIC SURGERY is an accepted form of therapy in the treatment of infertility. However, reports have differed
Tubal Plastic Surgery ADNAN MROUEH, M.D., ROBERT H. GLASS, M.D., and C. LEE BUXTON, M.D. TUBAL PLASTIC SURGERY is an accepted form of therapy in the treatment of infertility. However, reports have differed
me LUTEINIZED UNRUPTURED FOLLICLE SYNDROME AND ENDOMETRIOSIS
 FERTILITY AND STERILITY Copyright c 980 The American Fertility Society Vol. 33,, JanuaEY 980 Printed in U.S.A. me LUTEINIZED UNRUPTURED FOLLICLE SYNDROME AND ENDOMETRIOSIS W. PAULDMOWSKI, M.D.,.PH.D.*
FERTILITY AND STERILITY Copyright c 980 The American Fertility Society Vol. 33,, JanuaEY 980 Printed in U.S.A. me LUTEINIZED UNRUPTURED FOLLICLE SYNDROME AND ENDOMETRIOSIS W. PAULDMOWSKI, M.D.,.PH.D.*
Chapter 28: REPRODUCTIVE SYSTEM: MALE
 Chapter 28: REPRODUCTIVE SYSTEM: MALE I. FUNCTIONAL ANATOMY (Fig. 28.1) A. Testes: glands which produce male gametes, as well as glands producing testosterone 2. Seminiferous tubules (Fig.28.3; 28.5) a.
Chapter 28: REPRODUCTIVE SYSTEM: MALE I. FUNCTIONAL ANATOMY (Fig. 28.1) A. Testes: glands which produce male gametes, as well as glands producing testosterone 2. Seminiferous tubules (Fig.28.3; 28.5) a.
Chapter 14 Reproduction Review Assignment
 Date: Mark: _/45 Chapter 14 Reproduction Review Assignment Multiple Choice Identify the choice that best completes the statement or answers the question. 1. Use the diagram above to answer the next question.
Date: Mark: _/45 Chapter 14 Reproduction Review Assignment Multiple Choice Identify the choice that best completes the statement or answers the question. 1. Use the diagram above to answer the next question.
RECOVERY OF MIDCYCLE HUMAN FOLLICULAR OOCYTES: CORRELATION OF THEIR MORPHOLOGY WITH ENDOMETRIAL AND FOLLICULAR HISTOLOGY
 r FERTILITY AND STERILITY Copyright ~ 1978 The American Fertility Society Vol. 29,.5, May 1978 Printed in U.S.A. RECOVERY OF MIDCYCLE HUMAN FOLLICULAR OOCYTES: CORRELATION OF THEIR MORPHOLOGY WITH ENDOMETRIAL
r FERTILITY AND STERILITY Copyright ~ 1978 The American Fertility Society Vol. 29,.5, May 1978 Printed in U.S.A. RECOVERY OF MIDCYCLE HUMAN FOLLICULAR OOCYTES: CORRELATION OF THEIR MORPHOLOGY WITH ENDOMETRIAL
THE EFFECT OF COPPER IMPLANTS IN THE REMINAL VESICLES ON FERTILITY OF THE RAT, RABBIT, AND HAMSTER*
 FERTILITY A(\O Sn:HILIT'l Copyright 1973 by The Williams & Wilkins Co. Vol. 24, :-';0. 1..January 1973 Printed in U.S.A. THE EFFECT OF COPPER IMPLANTS IN THE REMINAL VESICLES ON FERTILITY OF THE RAT, RABBIT,
FERTILITY A(\O Sn:HILIT'l Copyright 1973 by The Williams & Wilkins Co. Vol. 24, :-';0. 1..January 1973 Printed in U.S.A. THE EFFECT OF COPPER IMPLANTS IN THE REMINAL VESICLES ON FERTILITY OF THE RAT, RABBIT,
MEGESTROL ACETATE. II. EFFECTS ON OVULATION IN NONHUMAN PRIMATES AS DETERMINED BY LAPAROSCOPY*
 FERTLTY AND STEULTY Copyright 1974 The American Fertility Society Vol. 25, No.1, January 1974 Printed in U.S.A. MEGESTROL ACETATE.. EFFECTS ON OVULATON N NONHUMAN PRMATES AS DETERMNED BY LAPAROSCOPY* RCHARD
FERTLTY AND STEULTY Copyright 1974 The American Fertility Society Vol. 25, No.1, January 1974 Printed in U.S.A. MEGESTROL ACETATE.. EFFECTS ON OVULATON N NONHUMAN PRMATES AS DETERMNED BY LAPAROSCOPY* RCHARD
Hormonal Control of Human Reproduction
 Hormonal Control of Human Reproduction Bởi: OpenStaxCollege The human male and female reproductive cycles are controlled by the interaction of hormones from the hypothalamus and anterior pituitary with
Hormonal Control of Human Reproduction Bởi: OpenStaxCollege The human male and female reproductive cycles are controlled by the interaction of hormones from the hypothalamus and anterior pituitary with
SPERM TRANSPORT FROM THE EXTERNAL CERVICAL OS TO THE FALLOPIAN TUBES IN WOMEN: ATIME AND QUANTITATION STUDY*t
 FERTILITY AND STERILITY Copyright 1973 by The Williams & Wilkins Co. Vol. 24, No.9, September 1973 Printed in U.S.A. SPERM TRANSPORT FROM THE EXTERNAL CERVICAL OS TO THE FALLOPIAN TUBES IN WOMEN: ATIME
FERTILITY AND STERILITY Copyright 1973 by The Williams & Wilkins Co. Vol. 24, No.9, September 1973 Printed in U.S.A. SPERM TRANSPORT FROM THE EXTERNAL CERVICAL OS TO THE FALLOPIAN TUBES IN WOMEN: ATIME
1. During the follicular phase of the ovarian cycle, the hypothalamus releases GnRH.
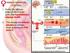 1. During the follicular phase of the ovarian cycle, the hypothalamus releases GnRH. 2. This causes the anterior pituitary to secrete small quantities of FSH and LH. 3. At this time, the follicles in the
1. During the follicular phase of the ovarian cycle, the hypothalamus releases GnRH. 2. This causes the anterior pituitary to secrete small quantities of FSH and LH. 3. At this time, the follicles in the
DATE: NAME: CLASS: Chapter 14 Test
 Multiple Choice Questions Decide which of the choices best completes the statement or answers the question. Locate that question number on the separate answer sheet provided. Use the procedure described
Multiple Choice Questions Decide which of the choices best completes the statement or answers the question. Locate that question number on the separate answer sheet provided. Use the procedure described
Chapter 46 ~ Animal Reproduction
 Chapter 46 ~ Animal Reproduction Overview Asexual (one parent) fission (parent separation) budding (corals) fragmentation & regeneration (inverts) parthenogenesis Sexual (fusion of haploid gametes) gametes
Chapter 46 ~ Animal Reproduction Overview Asexual (one parent) fission (parent separation) budding (corals) fragmentation & regeneration (inverts) parthenogenesis Sexual (fusion of haploid gametes) gametes
ONTARIO PORK RESEARCH PROPOSAL FINAL REPORT
 PROPOSAL # 04/16 ONTARIO PORK RESEARCH PROPOSAL FINAL REPORT Project Leader: R.N. Kirkwood/ R.M. Friendship Project Title: Effect of addition of seminal plasma to thawed semen on sow fertility Objectives
PROPOSAL # 04/16 ONTARIO PORK RESEARCH PROPOSAL FINAL REPORT Project Leader: R.N. Kirkwood/ R.M. Friendship Project Title: Effect of addition of seminal plasma to thawed semen on sow fertility Objectives
Realizing dreams booklet.indd 1 5/20/ :26:52 AM
 Realizing dreams. 18891booklet.indd 1 5/20/2010 11:26:52 AM The Journey To Parenthood The first Gator Baby was born in 1988 through the in vitro fertilization program at the University of Florida. Since
Realizing dreams. 18891booklet.indd 1 5/20/2010 11:26:52 AM The Journey To Parenthood The first Gator Baby was born in 1988 through the in vitro fertilization program at the University of Florida. Since
Bio 12- Ch. 21: Reproductive System
 Bio 12- Ch. 21: Reproductive System 21.1- Male Reproductive System o Male anatomy o Testes and how they relate to sperm production and male sex hormones o Hormone regulation in males 21.2- Female Reproductive
Bio 12- Ch. 21: Reproductive System 21.1- Male Reproductive System o Male anatomy o Testes and how they relate to sperm production and male sex hormones o Hormone regulation in males 21.2- Female Reproductive
EQUINE INTRACYTOPLASMIC SPERM INJECTION PROGRAM
 COLLEGE OF VETERINARY MEDICINE & BIOMEDICAL SCIENCES Department of Large Animal Clinical Sciences 4475 TAMU January 2016 b EQUINE INTRACYTOPLASMIC SPERM INJECTION PROGRAM Section of Theriogenology & the
COLLEGE OF VETERINARY MEDICINE & BIOMEDICAL SCIENCES Department of Large Animal Clinical Sciences 4475 TAMU January 2016 b EQUINE INTRACYTOPLASMIC SPERM INJECTION PROGRAM Section of Theriogenology & the
Molecular BASIS OF FERTILIZATION
 COLLEGE OF HEALTH SCIENCE DEPARTMENT OF PHYSIOLOGY PRESENTATION ON: Molecular BASIS OF FERTILIZATION By TEKETEL ERISTU Kediso 1 Presentation Outline Introduction Fertilization Types of Fertilization Cellular
COLLEGE OF HEALTH SCIENCE DEPARTMENT OF PHYSIOLOGY PRESENTATION ON: Molecular BASIS OF FERTILIZATION By TEKETEL ERISTU Kediso 1 Presentation Outline Introduction Fertilization Types of Fertilization Cellular
Functions of male Reproductive System: produce gametes deliver gametes protect and support gametes
 Functions of male Reproductive System: produce gametes deliver gametes protect and support gametes Spermatogenesis occurs in the testes after puberty. From the testes they are deposited into the epididymas
Functions of male Reproductive System: produce gametes deliver gametes protect and support gametes Spermatogenesis occurs in the testes after puberty. From the testes they are deposited into the epididymas
The Human Menstrual Cycle
 The Human Menstrual Cycle Name: The female human s menstrual cycle is broken into two phases: the Follicular Phase and the Luteal Phase. These two phases are separated by an event called ovulation. (1)
The Human Menstrual Cycle Name: The female human s menstrual cycle is broken into two phases: the Follicular Phase and the Luteal Phase. These two phases are separated by an event called ovulation. (1)
Female reproductive cycle: A Comprehensive Review Rachel Ledden Paper for Bachelors in Science January 20, 2018
 Running head: 1 Female reproductive cycle: A Comprehensive Review Rachel Ledden Paper for Bachelors in Science January 20, 2018 Female reproductive cycle: A Comprehensive Review 2 The reproductive cycle
Running head: 1 Female reproductive cycle: A Comprehensive Review Rachel Ledden Paper for Bachelors in Science January 20, 2018 Female reproductive cycle: A Comprehensive Review 2 The reproductive cycle
LIE ASSAY OF GONADOTROPIN in human blood is one of the most important
 Changes in Human Serum FSH Levels During the Normal Menstrual Cycle MASAO IGARASHI, M.D., JUNJI KAMIOKA, M.D., YOICHI EHARA, M.D., and SEIICHI MATSUMOTO, M.D. LIE ASSAY OF GONADOTROPIN in human blood is
Changes in Human Serum FSH Levels During the Normal Menstrual Cycle MASAO IGARASHI, M.D., JUNJI KAMIOKA, M.D., YOICHI EHARA, M.D., and SEIICHI MATSUMOTO, M.D. LIE ASSAY OF GONADOTROPIN in human blood is
The Female Reproductive System
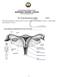 The Female Reproductive System STD X The female reproductive system consists of a pair of ovaries, a pair of oviducts, uterus, a vagina and the outermost part - the vulva. 1 1. Ovaries The ovaries are
The Female Reproductive System STD X The female reproductive system consists of a pair of ovaries, a pair of oviducts, uterus, a vagina and the outermost part - the vulva. 1 1. Ovaries The ovaries are
Animal Reproduction Chapter 46. Fission. Budding. Parthenogenesis. Fragmentation 11/27/2017
 Animal Reproduction Chapter 46 Both asexual and sexual reproduction occur in the animal kingdom Sexual reproduction is the creation of an offspring by fusion of a male gamete (sperm) and female gamete
Animal Reproduction Chapter 46 Both asexual and sexual reproduction occur in the animal kingdom Sexual reproduction is the creation of an offspring by fusion of a male gamete (sperm) and female gamete
DISPENSABILITY OF FIMBRIAE: OVUM PICKUP BY TUBAL FISTULAS IN THE RABBIT
 , I FERTIUTY AND STERIIJTY Copyright" 1979 The American Fertility Society Vol. 32, No.3, September 1979 Printed in U.SA. DISPENSABILITY OF FIMBRIAE: OVUM PICKUP BY TUBAL FISTULAS IN THE RABBIT KAREL G.
, I FERTIUTY AND STERIIJTY Copyright" 1979 The American Fertility Society Vol. 32, No.3, September 1979 Printed in U.SA. DISPENSABILITY OF FIMBRIAE: OVUM PICKUP BY TUBAL FISTULAS IN THE RABBIT KAREL G.
Information Booklet. Exploring the causes of infertility and treatment options.
 Information Booklet Exploring the causes of infertility and treatment options www.ptafertility.co.za info@ptafertility.co.za +27 12 998 8854 Faith is taking the first step even if you don t see the whole
Information Booklet Exploring the causes of infertility and treatment options www.ptafertility.co.za info@ptafertility.co.za +27 12 998 8854 Faith is taking the first step even if you don t see the whole
I. ART PROCEDURES. A. In Vitro Fertilization (IVF)
 DFW Fertility Associates ASSISTED REPRODUCTIVE TECHNOLOGY (ART) Welcome to DFW Fertility Associates/ Presbyterian-Harris Methodist Hospital ARTS program. This document provides an overview of treatment
DFW Fertility Associates ASSISTED REPRODUCTIVE TECHNOLOGY (ART) Welcome to DFW Fertility Associates/ Presbyterian-Harris Methodist Hospital ARTS program. This document provides an overview of treatment
Egg donation in an in vitro fertilization program: an alternative approach to cycle synchronization and timing of embryo transfer
 FERTILITY AND STERILITY Copyright tl 1989 The American Fertility Society Printed on acid-free paper in U. S.A. Egg donation in an in vitro fertilization program: an alternative approach to cycle synchronization
FERTILITY AND STERILITY Copyright tl 1989 The American Fertility Society Printed on acid-free paper in U. S.A. Egg donation in an in vitro fertilization program: an alternative approach to cycle synchronization
Central Progesterone Involvement in Estrogen- Induced Prolactin and Luteinizing Hormone Secretion Surges in Female Rats
 Southern Illinois University Carbondale OpenSIUC Honors Theses University Honors Program 5-10-2014 Central Progesterone Involvement in Estrogen- Induced Prolactin and Luteinizing Hormone Secretion Surges
Southern Illinois University Carbondale OpenSIUC Honors Theses University Honors Program 5-10-2014 Central Progesterone Involvement in Estrogen- Induced Prolactin and Luteinizing Hormone Secretion Surges
Infertility: failure to conceive within one year of unprotected regular sexual intercourse. Primary secondary
 Subfertility Infertility: failure to conceive within one year of unprotected regular sexual intercourse. Primary secondary Infertility affects about 15 % of couples. age of the female. Other factors that
Subfertility Infertility: failure to conceive within one year of unprotected regular sexual intercourse. Primary secondary Infertility affects about 15 % of couples. age of the female. Other factors that
Assisted Reproduction. By Dr. Afraa Mahjoob Al-Naddawi
 Assisted Reproduction By Dr. Afraa Mahjoob Al-Naddawi Learning Objectives: By the end of this lecture, you will be able to: 1) Define assisted reproductive techniques (ART). 2) List indications for various
Assisted Reproduction By Dr. Afraa Mahjoob Al-Naddawi Learning Objectives: By the end of this lecture, you will be able to: 1) Define assisted reproductive techniques (ART). 2) List indications for various
Unit 2 Physiology and Health Part (a) The Reproductive System HOMEWORK BOOKLET
 Unit 2 Physiology and Health Part (a) The Reproductive System HOMEWORK BOOKLET Name: Homework Date Due Mark % Key Area 1 The structure and function of reproductive organs Key Area 2 Hormonal control of
Unit 2 Physiology and Health Part (a) The Reproductive System HOMEWORK BOOKLET Name: Homework Date Due Mark % Key Area 1 The structure and function of reproductive organs Key Area 2 Hormonal control of
INDUCTION OF OVULATION IN URETHANE-TREATED RATS
 5 INDUCTION OF OVULATION IN URETHANE-TREATED RATS Ronald D. Johnson* and Barbara Shirley Faculty of Natural Sciences, University of Tulsa, Tulsa, Oklahoma 74104 Subcutaneous injection of urethane (1 g/kg
5 INDUCTION OF OVULATION IN URETHANE-TREATED RATS Ronald D. Johnson* and Barbara Shirley Faculty of Natural Sciences, University of Tulsa, Tulsa, Oklahoma 74104 Subcutaneous injection of urethane (1 g/kg
REPRODUCTION & GENETICS. Hormones
 REPRODUCTION & GENETICS Hormones http://www.youtube.com/watch?v=np0wfu_mgzo Objectives 2 Define what hormones are; Compare and contrast the male and female hormones; Explain what each hormone in the mail
REPRODUCTION & GENETICS Hormones http://www.youtube.com/watch?v=np0wfu_mgzo Objectives 2 Define what hormones are; Compare and contrast the male and female hormones; Explain what each hormone in the mail
Chapter 14 The Reproductive System
 Biology 12 Name: Reproductive System Per: Date: Chapter 14 The Reproductive System Complete using BC Biology 12, page 436-467 14. 1 Male Reproductive System pages 440-443 1. Distinguish between gametes
Biology 12 Name: Reproductive System Per: Date: Chapter 14 The Reproductive System Complete using BC Biology 12, page 436-467 14. 1 Male Reproductive System pages 440-443 1. Distinguish between gametes
CONTRACTILE PATTERNS OF ISOLATED OVIDUCTAL SMOOTH MUSCLE UNDER DIFFERENT HORMONAL CONDITIONS*
 FERTILITY AND STERILITY Copyright ~ 1980 The American Fertility Society Vol. 33. No.3. March 1980 Printed in U.SA. CONTRACTILE PATTERNS OF ISOLATED OVIDUCTAL SMOOTH MUSCLE UNDER DIFFERENT HORMONAL CONDITIONS*
FERTILITY AND STERILITY Copyright ~ 1980 The American Fertility Society Vol. 33. No.3. March 1980 Printed in U.SA. CONTRACTILE PATTERNS OF ISOLATED OVIDUCTAL SMOOTH MUSCLE UNDER DIFFERENT HORMONAL CONDITIONS*
REPRODUCTION The diagram below shows a section through seminiferous tubules in a testis.
 1. The diagram below shows a section through seminiferous tubules in a testis. Which cell produces testosterone? 2. A function of the interstitial cells in the testes is to produce A sperm B testosterone
1. The diagram below shows a section through seminiferous tubules in a testis. Which cell produces testosterone? 2. A function of the interstitial cells in the testes is to produce A sperm B testosterone
Health Science: the structures & functions of the reproductive system
 Health Science: the structures & functions of the reproductive BELLWORK 1. List (4) careers that are r/t the Reproductive, Urinary, and Endocrine Systems 2. Copy down the following terms: -ologist = one
Health Science: the structures & functions of the reproductive BELLWORK 1. List (4) careers that are r/t the Reproductive, Urinary, and Endocrine Systems 2. Copy down the following terms: -ologist = one
Topic 18- Human Reproductive System. Day 2-Female (and review of) Male Reproductive Systems
 Topic 18- Human Reproductive System Day 2-Female (and review of) Male Reproductive Systems Bell Ringer (5 minutes) Bioblitz WHAT ARE WE LEARNING TODAY? Date: 12/14-12/15 Topic: Human Reproductive System
Topic 18- Human Reproductive System Day 2-Female (and review of) Male Reproductive Systems Bell Ringer (5 minutes) Bioblitz WHAT ARE WE LEARNING TODAY? Date: 12/14-12/15 Topic: Human Reproductive System
Small Ruminant Reproductive Management Workshop
 Small Ruminant Reproductive Management Workshop Animal Nutrition and Physiology Center, North Dakota State University Sponsors: American Sheep and Goat Center, North Dakota State University, University
Small Ruminant Reproductive Management Workshop Animal Nutrition and Physiology Center, North Dakota State University Sponsors: American Sheep and Goat Center, North Dakota State University, University
Investigation: The Human Menstrual Cycle Research Question: How do hormones control the menstrual cycle?
 Investigation: The Human Menstrual Cycle Research Question: How do hormones control the menstrual cycle? Introduction: The menstrual cycle (changes within the uterus) is an approximately 28-day cycle that
Investigation: The Human Menstrual Cycle Research Question: How do hormones control the menstrual cycle? Introduction: The menstrual cycle (changes within the uterus) is an approximately 28-day cycle that
In Vitro Fertilization What to expect
 Patient Education In Vitro Fertilization What to expect This handout describes how to prepare for and what to expect when you have in vitro fertilization. It provides written information about this process,
Patient Education In Vitro Fertilization What to expect This handout describes how to prepare for and what to expect when you have in vitro fertilization. It provides written information about this process,
In Vitro Fertilization
 Patient Education In Vitro Fertilization About the treatment This handout describes how to prepare for and what to expect when you have in vitro fertilization. It provides written information about this
Patient Education In Vitro Fertilization About the treatment This handout describes how to prepare for and what to expect when you have in vitro fertilization. It provides written information about this
Chapter 27 The Reproductive System. MDufilho
 Chapter 27 The Reproductive System 1 Figure 27.19 Events of oogenesis. Before birth Meiotic events 2n Oogonium (stem cell) Mitosis Follicle development in ovary Follicle cells Oocyte 2n Primary oocyte
Chapter 27 The Reproductive System 1 Figure 27.19 Events of oogenesis. Before birth Meiotic events 2n Oogonium (stem cell) Mitosis Follicle development in ovary Follicle cells Oocyte 2n Primary oocyte
STRUCTURE AND FUNCTION OF THE FEMALE REPRODUCTIVE SYSTEM
 Unit 7B STRUCTURE AND FUNCTION OF THE FEMALE REPRODUCTIVE SYSTEM LEARNING OBJECTIVES 1. Learn the structures of the female reproductive tract. 2. Learn the functions of the female reproductive tract. 3.
Unit 7B STRUCTURE AND FUNCTION OF THE FEMALE REPRODUCTIVE SYSTEM LEARNING OBJECTIVES 1. Learn the structures of the female reproductive tract. 2. Learn the functions of the female reproductive tract. 3.
Student Academic Learning Services Page 1 of 5 Reproductive System Practice
 Student Academic Learning Services Page 1 of 5 Reproductive System Practice Fill in the blanks using the words listed: accessory glands, vas deferens, penis, scrotum, fallopian tube, testes, urethra, vagina,
Student Academic Learning Services Page 1 of 5 Reproductive System Practice Fill in the blanks using the words listed: accessory glands, vas deferens, penis, scrotum, fallopian tube, testes, urethra, vagina,
MedStar Health, Inc. POLICY AND PROCEDURE MANUAL Policy Number: PA.018.MH Last Review Date: 08/04/2016 Effective Date: 01/01/2017
 MedStar Health, Inc. POLICY AND PROCEDURE MANUAL PA.018.MH Infertility- Treatment This policy applies to the following lines of business: MedStar Employee (Select) MedStar MA DSNP CSNP (Not Covered) MedStar
MedStar Health, Inc. POLICY AND PROCEDURE MANUAL PA.018.MH Infertility- Treatment This policy applies to the following lines of business: MedStar Employee (Select) MedStar MA DSNP CSNP (Not Covered) MedStar
Methods Used to Self-Predict Ovulation A Comparative Study
 Marquette University e-publications@marquette Nursing Faculty Research and Publications Nursing, College of 5-1-1990 Methods Used to Self-Predict Ovulation A Comparative Study Richard Fehring Marquette
Marquette University e-publications@marquette Nursing Faculty Research and Publications Nursing, College of 5-1-1990 Methods Used to Self-Predict Ovulation A Comparative Study Richard Fehring Marquette
Chapter 36 Active Reading Guide Reproduction and Development
 Name: AP Biology Mr. Croft Chapter 36 Active Reading Guide Reproduction and Development Section 1 1. Distinguish between sexual reproduction and asexual reproduction. 2. Which form of reproduction: a.
Name: AP Biology Mr. Croft Chapter 36 Active Reading Guide Reproduction and Development Section 1 1. Distinguish between sexual reproduction and asexual reproduction. 2. Which form of reproduction: a.
Treatment 3 Days After Ovulation In Mares
 Luteal Regression And Follicle Development Following Prostaglandin-F 2α Treatment 3 Days After Ovulation In Mares D.R. Bergfelt a, R.A. Pierson b, and O.J. Ginther a a University of Wisconsin, Madison,
Luteal Regression And Follicle Development Following Prostaglandin-F 2α Treatment 3 Days After Ovulation In Mares D.R. Bergfelt a, R.A. Pierson b, and O.J. Ginther a a University of Wisconsin, Madison,
Animal Reproductive Systems. Chapter 42
 Animal Reproductive Systems Chapter 42 Impacts, Issues Male or Female? Body or Genes? Body and genes don t always match male or female characteristics also depend on hormones mutations can result in intersex
Animal Reproductive Systems Chapter 42 Impacts, Issues Male or Female? Body or Genes? Body and genes don t always match male or female characteristics also depend on hormones mutations can result in intersex
Infertility F REQUENTLY A SKED Q UESTIONS. Q: Is infertility a common problem?
 Infertility (female factors). In another one third of cases, infertility is due to the man (male factors). The remaining cases are caused by a mixture of male and female factors or by unknown factors.
Infertility (female factors). In another one third of cases, infertility is due to the man (male factors). The remaining cases are caused by a mixture of male and female factors or by unknown factors.
JOHN NEWTON M.B., B.S., M.R.C.O.G. , I E. 2a'~~~~ 600 ~ C} E2 > FIG. 1. Plasma hormone levels in normal menstrual
 Postgraduate Medical Journal (January 1972) 48, 5-9. Hormone excretion patterns in anovulatory infertility BEFORE embarking upon a discussion of anovulatory infertility, it is necessary to define a physiologically
Postgraduate Medical Journal (January 1972) 48, 5-9. Hormone excretion patterns in anovulatory infertility BEFORE embarking upon a discussion of anovulatory infertility, it is necessary to define a physiologically
of Chlormadinone on Amount of Human Cervical Mucus and Its Glycogen Content
 " Effect, of Chlormadinone on Amount of Human Cervical Mucus and Its Glycogen Content A. T. GREGOIRE, PHD, and K. USTAY, MD* THE MODE OF ACTION of orally administered steroids in contraceptive therapy
" Effect, of Chlormadinone on Amount of Human Cervical Mucus and Its Glycogen Content A. T. GREGOIRE, PHD, and K. USTAY, MD* THE MODE OF ACTION of orally administered steroids in contraceptive therapy
Evaluation of the Infertile Couple
 Overview and Definition Infertility is defined as the inability of a couple to fall pregnant after one year of unprotected intercourse. Infertility is a very common condition as in any given year about
Overview and Definition Infertility is defined as the inability of a couple to fall pregnant after one year of unprotected intercourse. Infertility is a very common condition as in any given year about
N. Shirazian, MD. Endocrinologist
 N. Shirazian, MD Internist, Endocrinologist Inside the ovary Day 15-28: empty pyfollicle turns into corpus luteum (yellow body) Immature eggs Day 1-13: 13: egg developing inside the growing follicle Day
N. Shirazian, MD Internist, Endocrinologist Inside the ovary Day 15-28: empty pyfollicle turns into corpus luteum (yellow body) Immature eggs Day 1-13: 13: egg developing inside the growing follicle Day
Unexpected Gynecologic Findings at Laparotomy. Susan A. Davidson, MD University of Colorado, Denver School of Medicine
 Unexpected Gynecologic Findings at Laparotomy Susan A. Davidson, MD University of Colorado, Denver School of Medicine Adnexal Mass: Gyn Etiologies Uterine Leiomyomas Pregnancy Malignancy Tubal Pregnancy
Unexpected Gynecologic Findings at Laparotomy Susan A. Davidson, MD University of Colorado, Denver School of Medicine Adnexal Mass: Gyn Etiologies Uterine Leiomyomas Pregnancy Malignancy Tubal Pregnancy
Female Reproductive System. Justin D. Vidal
 Female Reproductive System Justin D. Vidal If you cannot identify the tissue, then it is probably part of the female reproductive system! Introduction The female reproductive system is constantly changing,
Female Reproductive System Justin D. Vidal If you cannot identify the tissue, then it is probably part of the female reproductive system! Introduction The female reproductive system is constantly changing,
Sample Provincial exam Q s: Reproduction
 Sample Provincial exam Q s: Reproduction 11. Functions Testosterone Makes the male sex organs function normally, and also inhibits hypothalamus s release of GnRH and thus LH & FSH and thus testosterone
Sample Provincial exam Q s: Reproduction 11. Functions Testosterone Makes the male sex organs function normally, and also inhibits hypothalamus s release of GnRH and thus LH & FSH and thus testosterone
Chapter 22 The Reproductive System (I)
 Chapter 22 The Reproductive System (I) An Overview of Reproductive Physiology o The Male Reproductive System o The Female Reproductive System 22.1 Reproductive System Overview Reproductive system = all
Chapter 22 The Reproductive System (I) An Overview of Reproductive Physiology o The Male Reproductive System o The Female Reproductive System 22.1 Reproductive System Overview Reproductive system = all
Female and Male Reproductive Systems
 Female and Male Reproductive Systems Reproductive System: Organs that make possible the production of offspring. Female Reproductive System Female Reproductive System: Words to be familiar with ESTROGEN
Female and Male Reproductive Systems Reproductive System: Organs that make possible the production of offspring. Female Reproductive System Female Reproductive System: Words to be familiar with ESTROGEN
What are the main functions of the male reproductive system? 1. Produce sperm 2. Deposit sperm into the female 3. Provide a pathway for the removal
 What are the main functions of the male reproductive system? 1. Produce sperm 2. Deposit sperm into the female 3. Provide a pathway for the removal of urine Where is sperm produced? -In the 2 testes What
What are the main functions of the male reproductive system? 1. Produce sperm 2. Deposit sperm into the female 3. Provide a pathway for the removal of urine Where is sperm produced? -In the 2 testes What
Human Sexuality - Ch. 2 Sexual Anatomy (Hock)
 Human Sexuality - Ch. 2 Sexual Anatomy (Hock) penis penile glans corona frenulum penile shaft erection foreskin circumcision corpora cavernosa corpus spongiosum urethra scrotum spermatic cords testicles
Human Sexuality - Ch. 2 Sexual Anatomy (Hock) penis penile glans corona frenulum penile shaft erection foreskin circumcision corpora cavernosa corpus spongiosum urethra scrotum spermatic cords testicles
Human Reproduction. Male & Female Systems & Menstration
 Human Reproduction Male & Female Systems & Menstration Make a Sperm & Ovum Female Mons Pubis Labia Urethra Perineum Vagina Cervix Uterus Endometrium Fallopian tubes Ovaries Ovum Male Scrotrum Testicle
Human Reproduction Male & Female Systems & Menstration Make a Sperm & Ovum Female Mons Pubis Labia Urethra Perineum Vagina Cervix Uterus Endometrium Fallopian tubes Ovaries Ovum Male Scrotrum Testicle
HORMONES & REPRODUCTION OUTLINE
 1 HORMONES & REPRODUCTION Dr. Steinmetz OUTLINE 2 The Endocrine System Sexual Reproduction Hormonal Role in Sexual Differentiation Gender Differences and Gender Identity Characterizing Complex Behaviors
1 HORMONES & REPRODUCTION Dr. Steinmetz OUTLINE 2 The Endocrine System Sexual Reproduction Hormonal Role in Sexual Differentiation Gender Differences and Gender Identity Characterizing Complex Behaviors
Human Reproductive System
 Human Reproductive System I. The male reproductive anatomy is a delivery system for sperm. A. The male s external reproductive organs consist of the scrotum and penis. 1. The penis is the external organ
Human Reproductive System I. The male reproductive anatomy is a delivery system for sperm. A. The male s external reproductive organs consist of the scrotum and penis. 1. The penis is the external organ
MULTIPLE CHOICE: match the term(s) or description with the appropriate letter of the structure.
 Chapter 27 Exam Due NLT Thursday, July 31, 2015 Name MULTIPLE CHOICE: match the term(s) or description with the appropriate letter of the structure. Figure 27.1 Using Figure 27.1, match the following:
Chapter 27 Exam Due NLT Thursday, July 31, 2015 Name MULTIPLE CHOICE: match the term(s) or description with the appropriate letter of the structure. Figure 27.1 Using Figure 27.1, match the following:
EVALUATING THE INFERTILE PATIENT-COUPLES. Stephen Thorn, MD
 EVALUATING THE INFERTILE PATIENT-COUPLES Stephen Thorn, MD Overview The field of reproductive medicine continues to evolve rapidly by offering newer diagnostic testing and therapeutic options to improve
EVALUATING THE INFERTILE PATIENT-COUPLES Stephen Thorn, MD Overview The field of reproductive medicine continues to evolve rapidly by offering newer diagnostic testing and therapeutic options to improve
In domestic animals, we have limited period of estrus (sexual receptivity) and the term estrous
 REPRODUCTIVE CYCLES 1. Estrous cycle 2. Menstrual cycle In domestic animals, we have limited period of estrus (sexual receptivity) and the term estrous cycle is used. The onset of proestrus defines the
REPRODUCTIVE CYCLES 1. Estrous cycle 2. Menstrual cycle In domestic animals, we have limited period of estrus (sexual receptivity) and the term estrous cycle is used. The onset of proestrus defines the
COLLECTION OF HUMAN OOCYTES AT LAPAROSCOPY AND LAPAROTOMY
 FERTILITY AND STERILITY Copyright 1974 The American Fertility Society Vol. 25, No. 12, December 1974 Printed in U.s.A. COLLECTION OF HUMAN OOCYTES AT LAPAROSCOPY AND LAPAROTOMY ALEXANDER LOPATA,* M.B.,
FERTILITY AND STERILITY Copyright 1974 The American Fertility Society Vol. 25, No. 12, December 1974 Printed in U.s.A. COLLECTION OF HUMAN OOCYTES AT LAPAROSCOPY AND LAPAROTOMY ALEXANDER LOPATA,* M.B.,
Human and Bovine Sperm Migration
 Human and Bovine Sperm Migration K. S. MOGHSS, M.D. SPERM MGRATON may be accomplished by intrinsic sperm activity, uterotubal contractions, ciliary motions of tubal epithelium, or a combination of these
Human and Bovine Sperm Migration K. S. MOGHSS, M.D. SPERM MGRATON may be accomplished by intrinsic sperm activity, uterotubal contractions, ciliary motions of tubal epithelium, or a combination of these
