Origins and molecular biology of testicular germ cell tumors
|
|
|
- Calvin McDaniel
- 6 years ago
- Views:
Transcription
1 & 2005 USCAP, Inc All rights reserved /05 $ Origins and molecular biology of testicular germ cell tumors Victor E Reuter Department of Pathology, Memorial Sloan-ettering Cancer Center, New York, NY, USA Testicular germ cell tumors can be divided into three groups (infantile/prepubertal, adolescent/young adult and spermatocytic seminoma), each with its own constellation of clinical histology, molecular and clinical features. They originate from germ cells at different stages of development. The most common testicular cancers arise in postpubertal men and are characterized genetically by having one or more copies of an isochromosome of the short arm of chromosome 12 [i(12p)] or other forms of 12p amplification and by aneuploidy. The consistent gain of genetic material from chromosome 12 seen in these tumors suggests that it has a crucial role in their development. Intratubular germ cell neoplasia, unclassified type (IGCNU) is the precursor to these invasive tumors. Several factors have been associated with their pathogenesis, including cryptorchidism, elevated estrogens in utero and gonadal dysgenesis. Tumors arising in prepubertal gonads are either teratomas or yolk sac tumors, tend to be diploid and are not associated with i(12p) or with IGCNU. Spermatocytic seminoma (SS) arises in older patients. These benign tumors may be either diploid or aneuploid and have losses of chromosome 9 rather than i(12p). Intratubular SS is commonly encountered but IGCNU is not. The pathogenesis of prepubertal GCT and SS is poorly understood.. doi: /modpathol eywords: germ cell tumors; pathogenesis; intratubular germ cell neoplasia; molecular genetics Incidence Germ cell tumors are a heterogeneous group of neoplasms that arise primarily in the gonads and rarely from specific extragonadal sites along the midline, the pineal gland, mediastinum, retroperitoneum and sacrum. This particular distribution is thought to be due to the migration route of the primordial germ cells to the genital ridge. 1 3 Testicular germ cell tumors (TGCT) comprise approximately 98% of all testicular neoplasms and are the most common malignancy in males between the ages of 15 and 35 years. 4 It is possible to define three epidemiologically, clinically and histologically diverse groups of testicular germ cell tumors (Table 1). 1 The first group includes teratomas and yolk sac tumors of neonates and infants. These tumors occur well before puberty and have an annual incidence of approximately 0.12 per The second group includes seminomas and nonseminomatous germ cell tumors in postpubertal patients, by far the most Correspondence: Dr, MD, Department of Pathology, Memorial Sloan-ettering Cancer Center, 1275 York Avenue, New York, NY 10021, USA. reuterv@mskcc.org Received and accepted 29 August 2004 common form of TGCT, with an incidence of approximately 6.0 per per year and occurring usually between 15 and 40 years of age. The peak incidence for seminomas is in the fourth decade while for nonseminomas it is a decade earlier. The final group is spermatocytic seminoma, which usually occurs in an older age group and has an annual incidence of approximately 0.2 per It is clear that the incidence of TGCTs varies with geographic location and race (Table 2). 5 7 The highest incidence reported is from Scandinavia and Switzerland with an annual rate of over nine cases per males. Significantly lower rates have been reported from other locations such as Asia and Latin America. Evidence that race plays a role in this disease is provided by the fact that the incidence is over five times greater in Caucasian Americans compared to African Americans. 8 The only nonwhite population with a relatively high incidence of TGCT is the Maoris of New Zealand. 9 Interestingly, the incidence of TGCT in many countries has increased significantly during the last century. 8,10 12 Based on SEER data, McGlynn and collaborators reported an overall rise of 44% in the incidence in the United States from 1973 to 1998, from 3.35 per men to 4.84 per Although the rates increased among both white and
2 Origins and molecular biology of testicular germ cell tumors S52 Table 1 Testicular germ cell tumors Age Histology Incidence (per population) Genetic abnormalities Ploidy Prepubertal Teratoma/YST p, -6q Teratoma: diploid YST: aneuploid Postpubertal Seminoma/nonseminoma 6.0 IGCNU: 7i(12p) IGCNU: hypertriploid Seminomai: (12p) Seminoma: hypertriploid Nonseminoma: i(12p) Nonseminoma: hypotriploid Older (440 years) Spermatocytic seminoma Tetraploid/diploid YST ¼ Yolk sac tumor; IGCNU¼ intratubular germ cell neoplasia, unclassified type. Table 2 Factors implicated in the pathogenesis of TGCT black males, the increases observed were different in each group. Among white males, the incidence rose 52% (from 3.69 to 5.62 per ) whereas among black males the incidence rose 25% (from 0.83 to 1.04 per ). An unexpected finding was that the increase differed among histologic groups. Although the overall incidence of seminoma increased 72% in white and 42% in black males, the increase in non-seminoma for white males was only 32% while in black males there was a modest decrease of 3.0%. Most investigators have concluded that this increase in overall incidence of TGCT is due to changes in exposure to etiologic factors. Pathogenesis Race Familial/genetic predisposition Socioeconomic class/occupation Infertility Cryptorchidism High levels of maternal estrogens Low and high birth weights Neonatal jaundice Androgen insensitivity syndrome Gonadal dysgenesis The relative importance of susceptibility genes vs environmental factors in the etiology of TGCT is a source of great debate among investigators. 2,13 20 While both factors seem to play a role in this disease, at present their relative importance is not clear. We have previously mentioned differences among races. Some investigators have suggested a higher incidence among professionals and higher socioeconomic classes, but it is hard to evaluate the effect of race on these findings (Table 2). Some reports have suggested that specific occupations are associated with an increased risk but the data are conflicting and poorly controlled for confounding factors. Infertility has been associated with an increased risk of TGCT with intratubular germ cell neoplasia, unclassified type (IGCNU) being identified in 1 2% of cases. Nevertheless, infertility is itself associated with other conditions linked to the genesis of TGCT such as cryptorchidism and gonadal dysgenesis. Thus its importance as an independent risk factor is still in doubt. Much stronger associations with the development of TGCT exist with factors such as exposure to high levels of maternal estrogen during pregnancy, neonatal jaundice and low and high birth weights. These findings suggest that the initiating events that lead to tumor development occur early, possibly during the prenatal period The most widely accepted risk factor for TGCT is cryptorchidism, 24,25 approximately 10% of the cases having this association. Cryptorchid patients have a four-fold elevated risk of developing TGCT, whether the cryptorchidism was corrected or not There is some evidence that testicular maldescent is associated with prenatal exposure to estrogens. As a result of the elevated risk of germ cell neoplasia in cryptorchid gonads, some have suggested performing testicular biopsies in an attempt to identify IGCNU, which has been reported in up to 4% of cases. Patients with a history of TGCT have an increased incidence of contralateral disease. The reported incidence varies between 2 and 5%, the higher percentage having been reported from Scandinavian countries The incidence is also higher if the contralateral testis is atrophic or maldescended. Other investigators have suggested that bilateral disease is more common in patients with a positive family history for TGCT and if the first tumor occurred at an early age. 32 Bilateral tumors may develop synchronously or metachronously with approximately 50% occurring within 5 years. Some clinicians advocate biopsies of the contralateral gonad to identify IGCNU, which reliably predicts the subsequent development of an invasive GCT. Some testicular tumors appear to be familial with approximately 2% of patients having an affected family member These susceptible families rarely have more than two affected members and these are usually siblings rather than father and son. Early onset of disease and bilaterality are more common in this group of patients, with a reported frequency of 8 14%. Patients with various intersex syndromes are at increased risk for the development of TGCT
3 Patients with androgen insensitivity syndrome (testicular feminization) have a 5 10% risk of developing GCT, usually after the complete development of secondary female sexual characteristics. Gonadal dysgenesis in patients who carry a Y chromosome is a significant risk factor for germ cell tumor development. Linkage studies by Chemes et al 43 have shown a strong correlation between the presence of the Y chromosome and the severity of the gonadal dysgenesis. Gonadoblastoma acts as an in situ precursor in these patients. Recently, Skakkebaek and co-workers 37,42 suggested that many patients with testicular GCT have some evidence of gonadal dysgenesis in the contralateral gonad, be it microlithiasis, Sertoli-cell-only tubules, seminiferous tubules with undifferentiated Sertoli cells or IGCNU. If this is true, it suggests that a specific genetic mutation may not be required to initiate the process of tumor development, but rather environmental factors are sufficient as initiating events. Testicular biopsies in at-risk patients with intersex syndromes may aid in identifying IGCNU. Intratubular germ cell neoplasia This term refers to the lesion initially described by Skakkebaek as carcinoma in situ as well as to other differentiated forms of intratubular germ cell neoplasia Strictly speaking, the lesion originally described by Skakkebaek is now called intratubular germ cell neoplasia, unclassified by most, at least in the Western Hemisphere. The story of testicular carcinoma in situ /intratubular germ cell neoplasia is fascinating and serves as a paradigm for the concept of progression from incipient or preinvasive neoplasia to invasive disease. 46,48 In 1972, Skakkebaek reported atypical spermatogonia in two men undergoing testicular biopsies during the work-up for infertility, who subsequently developed invasive TGCT. He hypothesized that these cells constituted carcinoma in situ. Two subsequent seminal studies by his group proved that this was indeed the case. In 1978, he reported a series of 555 men who underwent testicular biopsies for infertility. 48 They identified six patients with evidence of carcinoma in situ. With a median follow-up period of approximately 3 years, three of these patients developed evidence of an invasive germ cell tumor; one of them with bilateral disease. The remaining 449 patients were tumor free during the same follow-up period. In 1986, the Skakkebaek group reported their experience with contralateral biopsies in 500 patients with unilateral GCT. 49 A total of 27 patients (5.4%) were found to have carcinoma in situ. Eight patients received systemic chemotherapy for advanced disease. Of the remaining 19 patients, seven (37%) developed invasive GCT at this site within the follow-up period. Mathematical modeling suggested that 50% of biopsy-positive cases would develop Origins and molecular biology of testicular germ cell tumors disease within 5 years. Remarkably, not a single case of contralateral GCT developed in the remaining 463 biopsy-negative patients during the same follow-up period. In a subsequent report, the authors revealed that at least two of the biopsy-positive cases that received systemic therapy subsequently developed contralateral tumors, suggesting that systemic therapy is not always effective against preinvasive disease. 50 It is clear that the original lesion described by Skakkebaek is the precursor to all types of germ cell tumors, at least for those that originate in postpubertal gonads, other than spermatocytic seminoma. In early 1980, a group of distinguished pathologists, including Drs Robert Scully, Juan Rosai, F Mostofi and Robert urman, met in Minnesota to discuss nomenclature of incipient germ cell neoplasia. They agreed that carcinoma in situ was a less than optimal choice to describe this lesion since it had no features of epithelial differentiation. They suggested the term intratubular germ cell neoplasia, unclassified (IGCNU) because it was associated with all morphologic types of GCT with the exception of spermatocytic seminoma. It also underscores the fact that differentiated forms of intratubular germ cell neoplasia may occur, including intratubular embryonal carcinoma. IGCNU can be seen adjacent to invasive germ cell tumors in virtually all cases in which residual testicular parenchyma is present. 44,51 As previously mentioned, it is present in up to 4% of cryptorchid patients, in up to 5% of contralateral gonads in patients with unilateral GCT and in up to 1% of patients biopsied for oligospermic infertility. Its association with TCGT arising in prepubertal patients is still a source of controversy. 1,52,53 While some authors suggest that it does not occur, others state that it does. In either case, we can state with reasonable certainty that, if IGCNU does occur in childhood tumors, it is certainly less apparent. IGCNU is characterized morphologically by the presence of enlarged, atypical germ cells located immediately above a usually irregularly thickened basement membrane (Figure 1). The atypical cells are either isolated or form a single row along the basement membrane. They are typically larger than spermatogonia, the other cell that usually resides near the basement membrane. IGCNU cells have clear cytoplasm, irregular nuclear contours, coarse chromatin, and enlarged nucleoli, which may be single or multiple. On the other hand, spermatogonia may also have clear cytoplasm but the cells are small, have round and regular nuclear contours, densely packed chromatin and no nucleoli. In most cases, tumor-bearing tubules do not have active spermatogenesis and contain mostly Sertoli cells. Sertoli cells may be displaced towards the tubular lumen. Characteristically they contain a single nucleolus which is small and regular. The nuclei S53
4 Origins and molecular biology of testicular germ cell tumors S54 Figure 2 Intratubular seminoma. The seminiferous tubules are filled with tumor cells cytologically indistinguishable from IGCNU, the latter evident at the right. Figure 1 (a) IGCNU. The malignant cells are located towards the periphery of the lumen. They exhibit cytoplasmic clearing and the nuclei are enlarged, have irregular contours, coarse chromatin and prominent nucleoli. (b) IGCNU (arrows) associated with microlithiasis. (c) Isolated neoplastic cells (IGCNU) within hyalinized tubules. are oval or round with regular borders and the chromatin is fine. The cytoplasm is amphophilic/ eosinophilic and not vacuolated. In essence, the cytologic features of classic IGCNU are those of seminoma. The relationship is supported by the coexpression of a host of histochemical and immunohistochemical markers among both cell types. Further evidence comes from electron microscopy, which has shown that both share common ultrastructural features including the absence of well-developed cytoplasmic intermediate filaments, inconspicuous organelles, glycogen particles, lack of mature desmosomes and cell junctions, and nucleoli with ropy nucleolonema. Tubules whose lumen are filled with these cells may be regarded as intratubular seminoma (Figure 2). IGCNU may extend into the rete testis, usually undermining the epithelium in a pagetoid pattern (Figure 3a). At times the epithelium may become hyperplastic and in this setting it is important not to confuse this finding with the presence of nonseminomatous germ cell tumor (Figure 3b). IGCNU cells contain glycogen and thus are PASpositive, diastase-sensitive. Rarely will other intratubular cells, either spermatogonia or Sertoli cells, show similar positivity. Placental-like alkaline phosphatase (PLAP) is one of the isoforms of alkaline phosphatase. PLAP antibodies will stain IGCNU and the majority of seminomas and embryonal carcinomas as well as a smaller percentage of yolk sac tumors. Immunoreactivity is seen in virtually all cases of IGCNU and the staining pattern is usually membranous or cytoplasmic (Figure 4a). No other non-neoplastic intratubular cells are immunoreactive for PLAP, but immunoreactivity may be seen in other types of nongerm cell malignancies C-kit (CD 117) is expressed in a large percentage of IGCNU as well as seminomas, but not in other germ cell tumors. 58 Once again, the staining pattern is cytoplasmic/membranous (Figure 4b). Despite the overexpression of this antigen, c-kit is rarely mutated in these tumors. Other antibodies,
5 Origins and molecular biology of testicular germ cell tumors S55 Figure 3 (a) Pagetoid extension of IGCNU into the rete testis. (b) Pagetoid extension of IGCNU into the rete testis associated with reactive hyperplasia of the rete epithelium. This finding may be confused with nonseminomatous GCT. Figure 4 Immunoreactivity in IGCNU. (a) Placental-like alkaline phosphatase. (b) CD-117 (c-kit). Note cytoplasmic staining with accentuation of the cytoplasmic membrane with both markers. (c) Oct3/4. Strong nuclear immunoreactivity is limited to the neoplastic cells. which immunoreact with IGCNU but are rarely used in clinical practice, include M2A and 43-F. 57,59,60 POU5F1 (Oct3/4) is a very interesting marker that was recently described. 61 The gene serves as a transcription factor and its product is expressed in pluripotent mouse and human embryonic stem cells and is downregulated during differentiation. Since the gene is also required for self-renewal of embryonic stem cells, knocking out the gene is lethal. Early reports suggest that this antigen is expressed solely in IGCNU, seminoma and embryonal carcinoma, suggesting that these are the types of GCT cells with pluripotency, that is, with capacity to differentiate. In any event, it provides us with yet another marker for IGCNU (Figure 4c). It is important to keep in mind that the presence of neoplastic cells within tubules does not always constitute IGCNU and that one must adhere strictly to the established diagnostic criteria. Other forms of intratubular germ cell neoplasia (IGCN) include intratubular seminoma (Figure 2), intratubular embryonal carcinoma (Figure 5) and intratubular spermatocytic seminoma (Figure 6). Secondary tumors may sometimes grow within the seminiferous tubules and mimic IGCN, including melanoma
6 Origins and molecular biology of testicular germ cell tumors S56 Figure 5 Intratubular embryonal carcinoma. The tumor cells are cytologically high grade and demonstrate comedo-type necrosis. A fibroinflammatory reaction characteristically surrounds the effected tubules. Figure 7 Intratubular metastatic melanoma. This lesion could be confused with intratubular seminoma in the absence of the clinical history, although greater nuclear variability represents a subtle clue. Figure 6 Intratubular spermatocytic seminoma. The tubules are distended by a polymorphous population of cells and lack the characteristic features of IGCNU. (Figure 7), prostatic carcinoma, lymphoma and even mesothelioma. Molecular biology of germ cell tumors The molecular biology of GCTs is fascinating and fertile ground for basic and translational research. As GCTs are for the most part curable due to effective chemotherapy and in some cases by surgery alone, lessons learned from this disease may help treat other solid tumors more effectively. The initiating genetic events that lead to IGCNU and subsequent invasive GCT have yet to be agreed upon. 1,2,13,62 64 There are two major theories, the first being championed by Chaganti and Houldsworth. 63,64 They have postulated that the most likely target cell for transformation is the zygotene-pachytene spermatocyte where there appears to be a recombination checkpoint. At this stage, there is replicated DNA and upregulated p53, the latter to allow prolongation of the phase and recombination. If unresolved, DNA strand breaks occur and the elevated wild-type p53 can provide an apoptotic trigger. In this model, aberrant chromatid exchange events associated with crossing-over may lead to increased 12p copy number. Possibly through the oncogenic effect of cyclin D2, this aberrant, genomically unstable cell is now able to escape the apoptotic effects of p53 and may re-enter the cell cycle, now as a neoplastic cell. A second and more widely accepted hypothesis is promulgated by Skakkebaek and others. 14,62,65 This model suggests that fetal gonocytes (primordial germ cells) may undergo abnormal cell division (polyploidization) due to mostly environmental factors in utero and give rise to IGCNU. Polyploidization precedes the formation of i(12p). These cells are now susceptible to postnatal and pubertal gonadotropin stimulation and the development of invasive tumors. This hypothesis is based primarily on immunohistochemical data linking gonocytes to IGCNU, developmental abnormalities associated with GCT and epidemiological data. Once again I must emphasize that there are three distinct categories of testicular GCT, each with its own clinical, pathologic and genetic characteristics. Most of what is known about the genetics of this tumor deals with the most common type, those that arise in adolescents and young adults. Seminomas and nonseminomas that arise in this group are consistently aneuploid. The median DNA content of IGCNU and seminoma is hypertriploid and that of nonseminomas is hypotriploid. The characteristic genetic abnormality of these tumors is excess genetic material of the short arm of chromosome
7 12, usually in the form of an isochromosome; i(12p) (Figure 8). This genetic abnormality occurs in all primary sites and histologic types. 66,67 Approximately 80% of cases have i(12p) and the remainder have excess 12p genetic material in derivative chromosomes. 68 The consistent gain of 12p genetic material is supported by gene array and comparative genomic hybridization data. 69,70 The presence of i(12p) in IGCNU is a matter of controversy with most investigators suggesting it is not present. 2,14 This finding tends to support the Skakkebaek hypothesis that polyploidization is an initiating event followed by i(12p) in the invasive phenotype. Since the formation of the isochromosome includes the centromeric/pericentromeric region of chromosome 12, the abnormality can be detected using commercially available chromosome 12 centromeric fluorescent in situ hybridization (FISH) probes. 67,68,71 In a metaphase spread these will show the isochromosome nicely. In interphase cells one can detect a numerical abnormality (normal chromosome 12 plus the number of isochromosomes present) but it is more difficult if not impossible to state that an isochromosome is indeed present. On the other hand, a library probe of the short arm of chromosome 12 can highlight the presence of the isochromosome more easily in metaphase spreads as well as in resting cells (Figure 9). Data suggest that the consistent gain of genetic material from chromosome 12 is crucial for the development of an invasive GCT resulting in efforts to identify the specific genes involved. Some investigators have identified an amplified region at 12p Several genes of interest are located in this region including SOX5, JAW1 and -RAS. 14 More recent data suggest that this minimally deleted region applies preferentially to seminomas, suggesting that the search for putative relevant genes should not be restricted to this region. For example, Houldsworth et al 72 implicated cyclin D2, which is mapped to region 12p13, as the most likely gene to be involved. To date, 247 transcripts mapped to 12p have been evaluated for expression using microarray technology and 24 were found to be overexpressed Figure 8 Diagram of chromosome 12 and isochromosome 12p. The image on the right shows an i(12p) as seen in a karyotype. Origins and molecular biology of testicular germ cell tumors Figure 9 FISH using a 12p specific probe. The large, irregular signals are indicative of the presence of excess genetic material from the short arm of chromosome 12. in over 70% of tumor samples relative to normal testis. A significant number of these transcripts are located telomeric to the 12p11 12p12 region. A few studies have suggested the presence of relevant tumor suppressor genes but the data have not been consistent. Murty et al 73 described two regions of interest on chromosome 12 (q13 and q22) although no candidate genes have been proposed. Homozygous deletions in the region of DCC on 18q have also been identified and associated to progression. Extensive molecular analyses have identified genomic and/or functional (expression) loss of several other known tumor suppressor genes such as RB1, NME, WT1, APC, and p16 although no consistent, reproducible and mechanistic data are available to date linking these genes with tumor development or progression. 2,14 Mutation of the p53 gene is a rare event in GCT, yet a few investigators have suggested mutation of this gene in some chemotherapy-resistant tumors. 74 Whether this mutation is responsible for chemoresistance rather than an epiphenomenon has not been resolved. As previously mentioned, GCTs in infants and adults are genetically distinct. 1,2,75 80 While virtually all adult cases are aneuploid, pediatric cases are mostly diploid, particularly teratomas, although yolk sac tumors may be nondiploid. Imprinting studies suggest that both arise from primordial germ cells but at a different stage of germ cell development. 76 It is quite likely that initiation of childhood GCT occurs during germ cell migration to the genital ridge while this step takes place at a later stage in adults. i(12p) and 12p amplification are rare events in pediatric tumors. Bussey et al 77 found that 12p amplification in childhood GCT was age dependent S57
8 S58 and only seen in patients above the age of 9 years. On the other hand, pediatric tumors are characterized by deletions of 1p, particularly region p36, loss of 6q, and structural abnormalities of chromosome 2 and 3p. Published reports point towards a significant heterogeneity in the structural chromosomal constitution of infantile teratomas. Genetic studies on spermatocytic seminoma are few; it appears that the most common chromosomal abnormality is gains in chromosome 9. 2 A combined immunohistochemical and molecular study by Stoop et al 81 supports that these tumors are likely to originate in a cell farther along the developmental pathway than those that give rise to other germ cell tumors. References Origins and molecular biology of testicular germ cell tumors 1 Looijenga LH, Oosterhuis JW. Pathogenesis of testicular germ cell tumours. Rev Reprod 1999;4: Oosterhuis JW, Looijenga LH. Current views on the pathogenesis of testicular germ cell tumours and perspectives for future research: highlights of the 5th Copenhagen Workshop on Carcinoma in situ and Cancer of the Testis. Apmis 2003;111: Slowikowska-Hilczer J, Romer TE, ula. Neoplastic potential of germ cells in relation to disturbances of gonadal organogenesis and changes in karyotype. J Androl 2003;24: Bosl GJ, Motzer RJ. Testicular germ-cell cancer. N Engl J Med 1997;337: Moller H. Trends in incidence of testicular cancer and prostate cancer in Denmark. Hum Reprod 2001;16: Bergstrom R, Adami HO, Mohner M, et al. Increase in testicular cancer incidence in six European countries: a birth cohort phenomenon. J Natl Cancer Inst 1996;88: Adami HO, Bergstrom R, Mohner M, et al. Testicular cancer in nine northern European countries. Int J Cancer 1994;59: McGlynn A, Devesa SS, Sigurdson AJ, et al. Trends in the incidence of testicular germ cell tumors in the United States. Cancer 2003;97: Wilkinson TJ, Colls BM, Schluter PJ. Increased incidence of germ cell testicular cancer in New Zealand Maoris. Br J Cancer 1992;65: Stone JM, Cruickshank DG, Sandeman TF, et al. Trebling of the incidence of testicular cancer in victoria, Australia ( ). Cancer 1991;68: Boyle P, aye SB, Robertson AG. Changes in testicular cancer in Scotland. Eur J Cancer Clin Oncol 1987;23: Power DA, Brown RS, Brock CS, et al. Trends in testicular carcinoma in England and Wales, Br J Urol Int 2001;87: Looijenga LH, Oosterhuis JW. Pathobiology of testicular germ cell tumors: views and news. Anal Quant Cytol Histol 2002;24: Skotheim RI, Lothe RA. The testicular germ cell tumour genome. Apmis 2003;111: ; discussion Forman D, Gallagher R, Moller H, et al. Aetiology and epidemiology of testicular cancer: report of consensus group. Prog Clin Biol Res 1990;357: Swerdlow AJ. The epidemiology of testicular cancer. Eur Urol 1993;23(Suppl 2): Hardell L, Nasman A, Ohlson CG, et al. Case control study on risk factors for testicular cancer. Int J Oncol 1998;13: Coupland CA, Chilvers CE, Davey G, et al. Risk factors for testicular germ cell tumours by histological tumour type. United ingdom Testicular Cancer Study Group. Br J Cancer 1999;80: Richiardi L, Akre O, Bellocco R, et al. Perinatal determinants of germ-cell testicular cancer in relation to histological subtypes. Br J Cancer 2002;87: Richiardi L, Askling J, Granath F, et al. Body size at birth and adulthood and the risk for germ-cell testicular cancer. Cancer Epidemiol Biomarkers Prev 2003;12: Ekbom A. Growing evidence that several human cancers may originate in utero. Semin Cancer Biol 1998;8: Ekbom A, Akre O. Increasing incidence of testicular cancer birth cohort effects. Apmis 1998;106: ; discussion Depue RH, Pike MC, Henderson BE. Estrogen exposure during gestation and risk of testicular cancer. J Natl Cancer Inst 1983;71: Halme A, ellokumpu-lehtinen P, Lehtonen T, et al. Morphology of testicular germ cell tumours in treated and untreated cryptorchidism. Br J Urol 1989;64: Schottenfeld D, Warshauer ME, Sherlock S, et al. The epidemiology of testicular cancer in young adults. Am J Epidemiol 1980;112: Pike MC, Chilvers C, Peckham MJ. Effect of age at orchidopexy on risk of testicular cancer. Lancet 1986;1: Pottern LM, Brown LM, Hoover RN, et al. Testicular cancer risk among young men: role of cryptorchidism and inguinal hernia. J Natl Cancer Inst 1985;74: Giwercman A, Grindsted J, Hansen B, et al. Testicular cancer risk in boys with maldescended testis: a cohort study. J Urol 1987;138: Dieckmann P, Loy V, Buttner P. Prevalence of bilateral testicular germ cell tumours and early detection based on contralateral testicular intra-epithelial neoplasia. Br J Urol 1993;71: Dieckmann P, Loy V. The value of the biopsy of the contralateral testis in patients with testicular germ cell cancer: the recent German experience. Apmis 1998;106:13 20; discussion Osterlind A, Berthelsen JG, Abildgaard N, et al. Incidence of bilateral testicular germ cell cancer in Denmark, : preliminary findings. Int J Androl 1987;10: Lutke Holzik MF, Sijmons RH, Sleijfer DT, et al. Syndromic aspects of testicular carcinoma. Cancer 2003;97: Dieckmann P, Becker T, Jonas D, et al. Inheritance and testicular cancer. Arguments based on a report of 3 cases and a review of the literature. Oncology 1987;44: Classen J, Dieckmann P, Loy V, et al. [Testicular intraepithelial neoplasms (TIN). An indication for radiotherapy?]. Strahlenther Onkol 1998;174: Patel SR, vols L, Richardson RL. Familial testicular cancer: report of six cases and review of the literature. Mayo Clin Proc 1990;65:
9 36 Heimdal, Olsson H, Tretli S, et al. Familial testicular cancer in Norway and southern Sweden. Br J Cancer 1996;73: Skakkebaek NE, Holm M, Hoei-Hansen C, et al. Association between testicular dysgenesis syndrome (TDS) and testicular neoplasia: evidence from 20 adult patients with signs of maldevelopment of the testis. Apmis 2003;111: Rutgers JL, Scully RE. Pathology of the testis in intersex syndromes. Semin Diagn Pathol 1987;4: Rutgers JL, Scully RE. The androgen insensitivity syndrome (testicular feminization): a clinicopathologic study of 43 cases. Int J Gynecol Pathol 1991;10: Collins GM, im DU, Logrono R, et al. Pure seminoma arising in androgen insensitivity syndrome (testicular feminization syndrome): a case report and review of the literature. Mod Pathol 1993;6: Ramani P, Yeung C, Habeebu SS. Testicular intratubular germ cell neoplasia in children and adolescents with intersex. Am J Surg Pathol 1993;17: Hoei-Hansen CE, Holm M, Rajpert-De Meyts E, et al. Histological evidence of testicular dysgenesis in contralateral biopsies from 218 patients with testicular germ cell cancer. J Pathol 2003;200: Chemes HE, Muzulin PM, Venara MC, et al. Early manifestations of testicular dysgenesis in children: pathological phenotypes, karyotype correlations and precursor stages of tumour development. Apmis 2003; 111: Dieckmann P, Skakkebaek NE. Carcinoma in situ of the testis: review of biological and clinical features. Int J Cancer 1999;83: Montironi R. Intratubular germ cell neoplasia of the testis: testicular intraepithelial neoplasia. Eur Urol 2002;41: Gondos B, Berthelsen JG, Skakkebaek NE. Intratubular germ cell neoplasia (carcinoma in situ): a preinvasive lesion of the testis. Ann Clin Lab Sci 1983;13: Skakkebaek NE. Atypical germ cells in the adjacent normal tissue of testicular tumours. Acta Pathol Microbiol Scand [A] 1975;83: Skakkebaek NE. Carcinoma in situ of the testis: frequency and relationship to invasive germ cell tumours in infertile men. Histopathology 1978;2: von der Maase H, Rorth M, Walbom-Jorgensen S, et al. Carcinoma in situ of contralateral testis in patients with testicular germ cell cancer: study of 27 cases in 500 patients. Br Med J (Clin Res Ed) 1986;293: von der Maase H, Meinecke B, Skakkebaek NE. Residual carcinoma-in-situ of contralateral testis after chemotherapy. Lancet 1988;1: Jacobsen G, Henriksen OB, von der Maase H. Carcinoma in situ of testicular tissue adjacent to malignant germ-cell tumors: a study of 105 cases. Cancer 1981;47: Manivel JC, Simonton S, Wold LE, et al. Absence of intratubular germ cell neoplasia in testicular yolk sac tumors in children. A histochemical and immunohistochemical study. Arch Pathol Lab Med 1988;112: Hu LM, Phillipson J, Barsky SH. Intratubular germ cell neoplasia in infantile yolk sac tumor. Verification by tandem repeat sequence in situ hybridization. Diagn Mol Pathol 1992;1: Origins and molecular biology of testicular germ cell tumors 54 Manivel JC, Jessurun J, Wick MR, et al. Placental alkaline phosphatase immunoreactivity in testicular germ-cell neoplasms. Am J Surg Pathol 1987;11: Wick MR, Swanson PE, Manivel JC. Placental-like alkaline phosphatase reactivity in human tumors: an immunohistochemical study of 520 cases. Hum Pathol 1987;18: Burke AP, Mostofi F. Intratubular malignant germ cells in testicular biopsies: clinical course and identification by staining for placental alkaline phosphatase. Mod Pathol 1988;1: Giwercman A, Cantell L, Marks A. Placental-like alkaline phosphatase as a marker of carcinoma-in-situ of the testis. Comparison with monoclonal antibodies M2A and 43-9F. Apmis 1991;99: Leroy X, Augusto D, Leteurtre E, et al. CD30 and CD117 (c-kit) used in combination are useful for distinguishing embryonal carcinoma from seminoma. J Histochem Cytochem 2002;50: Giwercman A, Lindenberg S, imber SJ, et al. Monoclonal antibody 43-9F as a sensitive immunohistochemical marker of carcinoma in situ of human testis. Cancer 1990;65: Marks A, Sutherland DR, Bailey D, et al. Characterization and distribution of an oncofetal antigen (M2A antigen) expressed on testicular germ cell tumours. Br J Cancer 1999;80: Looijenga LH, Stoop H, de Leeuw HP, et al. POU5F1 (OCT3/4) identifies cells with pluripotent potential in human germ cell tumors. Cancer Res 2003;63: Looijenga LH, de Munnik H, Oosterhuis JW. A molecular model for the development of germ cell cancer. Int J Cancer 1999;83: Chaganti RS, Houldsworth J. The cytogenetic theory of the pathogenesis of human adult male germ cell tumors. Review article. Apmis 1998;106:80 83; discussion Chaganti RS, Houldsworth J. Genetics and biology of adult human male germ cell tumors. Cancer Res 2000;60: Grigor M, Skakkebaek NE. Pathogenesis and cell biology of germ cell neoplasia: general discussion. Eur Urol 1993;23: Bosl GJ, Ilson DH, Rodriguez E, et al. Clinical relevance of the i(12p) marker chromosome in germ cell tumors. J Natl Cancer Inst 1994;86: Rodriguez E, Mathew S, Mukherjee AB, et al. Analysis of chromosome 12 aneuploidy in interphase cells from human male germ cell tumors by fluorescence in situ hybridization. Genes Chromosomes Cancer 1992;5: Rodriguez E, Houldsworth J, Reuter VE, et al. Molecular cytogenetic analysis of i(12p)-negative human male germ cell tumors. Genes Chromosomes Cancer 1993;8: Rodriguez S, Jafer O, Goker H, et al. Expression profile of genes from 12p in testicular germ cell tumors of adolescents and adults associated with i(12p) and amplification at 12p11.2 p12.1. Oncogene 2003;22: raggerud SM, Skotheim RI, Szymanska J, et al. Genome profiles of familial/bilateral and sporadic testicular germ cell tumors. Genes Chromosomes Cancer 2002;34: Blough RI, Heerema NA, Ulbright TM, et al. Interphase chromosome painting of paraffin-embedded tissue in S59
10 S60 Origins and molecular biology of testicular germ cell tumors the differential diagnosis of possible germ cell tumors. Mod Pathol 1998;11: Houldsworth J, Reuter V, Bosl GJ, et al. Aberrant expression of cyclin D2 is an early event in human male germ cell tumorigenesis. Cell Growth Differ 1997; 8: Murty VV, Li RG, Houldsworth J, et al. Frequent allelic deletions and loss of expression characterize the DCC gene in male germ cell tumors. Oncogene 1994;9: Houldsworth J, Xiao H, Murty VV, et al. Human male germ cell tumor resistance to cisplatin is linked to TP53 gene mutation. Oncogene 1998;16: Schneider DT, Schuster AE, Fritsch M, et al. Genetic analysis of childhood germ cell tumors with comparative genomic hybridization. lin Padiatr 2001;213: Schneider DT, Schuster AE, Fritsch M, et al. Multipoint imprinting analysis indicates a common precursor cell for gonadal and nongonadal pediatric germ cell tumors. Cancer Res 2001;61: Bussey J, Lawce HJ, Himoe E, et al. Chromosomes 1 and 12 abnormalities in pediatric germ cell tumors by interphase fluorescence in situ hybridization. Cancer Genet Cytogenet 2001;125: Silver SA, Wiley JM, Perlman EJ. DNA ploidy analysis of pediatric germ cell tumors. Mod Pathol 1994;7: Jenderny J, oster E, Borchers O, et al. Interphase cytogenetics on paraffin sections of paediatric extragonadal yolk sac tumours. Virchows Arch 1996;428: Stock C, Strehl S, Fink FM, et al. Isochromosome 12p and maternal loss of 1p36 in a pediatric testicular germ cell tumor. Cancer Genet Cytogenet 1996;91: Stoop H, van Gurp R, de rijger R, et al. Reactivity of germ cell maturation stage-specific markers in spermatocytic seminoma: diagnostic and etiological implications. Lab Invest 2001;81:
Intratubular Germ Cell Neoplasia of the Testis
 Intratubular Germ Cell Neoplasia of the Testis KS Ngoo Department of Urology Hospital Selayang Advanced Urology Course 15 Aug 2014 MUA Office Clinical scenario A 33 years old man has bilateral testicular
Intratubular Germ Cell Neoplasia of the Testis KS Ngoo Department of Urology Hospital Selayang Advanced Urology Course 15 Aug 2014 MUA Office Clinical scenario A 33 years old man has bilateral testicular
Note: The cause of testicular neoplasms remains unknown
 - In the 15- to 34-year-old age group, they are the most common tumors of men. - Tumors of the testis are a heterogeneous group of neoplasms that include: I. Germ cell tumors : 95%; all are malignant.
- In the 15- to 34-year-old age group, they are the most common tumors of men. - Tumors of the testis are a heterogeneous group of neoplasms that include: I. Germ cell tumors : 95%; all are malignant.
-The cause of testicular neoplasms remains unknown
 - In the 15- to 34-year-old age group, they are the most common tumors of men. - include: I. Germ cell tumors : (95%); all are malignant. II. Sex cord-stromal tumors: from Sertoli or Leydig cells; usually
- In the 15- to 34-year-old age group, they are the most common tumors of men. - include: I. Germ cell tumors : (95%); all are malignant. II. Sex cord-stromal tumors: from Sertoli or Leydig cells; usually
Genetic Studies of Dysgerminoma
 Genetic Studies of Chromosome 12p abnormalities are characteristic of germ cell tumors isochromosome 12p and 12p overrepresentation Some can be detected by karyotyping FISH study of 21 dysgerminomas showed
Genetic Studies of Chromosome 12p abnormalities are characteristic of germ cell tumors isochromosome 12p and 12p overrepresentation Some can be detected by karyotyping FISH study of 21 dysgerminomas showed
Male Genital Cancers in the US in Frequency of Types
 Germ Cell Tumors of the Testis Pathology, Immunohistochemistry, and the Often Confusing Appearance of Their Metastases Charles Zaloudek, MD Department of Pathology UCSF Male Genital Cancers in the US in
Germ Cell Tumors of the Testis Pathology, Immunohistochemistry, and the Often Confusing Appearance of Their Metastases Charles Zaloudek, MD Department of Pathology UCSF Male Genital Cancers in the US in
(Epi)Genetics in normal and malignant germ cell development.
 (Epi)Genetics in normal and malignant germ cell development. Leendert Looijenga, Department of Pathology, Lab. Exp. Patho-Oncol. Erasmus MC, Rotterdam OPTIMAL PATIENT CARE MM Basic and Transl. Oncol.,
(Epi)Genetics in normal and malignant germ cell development. Leendert Looijenga, Department of Pathology, Lab. Exp. Patho-Oncol. Erasmus MC, Rotterdam OPTIMAL PATIENT CARE MM Basic and Transl. Oncol.,
Bilateral Testicular Germ Cell Tumors
 1228 Bilateral Testicular Germ Cell Tumors Twenty-Year Experience at M. D. Anderson Cancer Center Mingxin Che, M.D., Ph.D. 1 Pheroze Tamboli, M.D. 1 Jae Y. Ro, M.D., Ph.D. 1 Dong Soo Park, M.D. 2 Jung
1228 Bilateral Testicular Germ Cell Tumors Twenty-Year Experience at M. D. Anderson Cancer Center Mingxin Che, M.D., Ph.D. 1 Pheroze Tamboli, M.D. 1 Jae Y. Ro, M.D., Ph.D. 1 Dong Soo Park, M.D. 2 Jung
Testicular Germ Cell Tumors; A Simplistic Approach
 Testicular Germ Cell Tumors; A Simplistic Approach Merce Jorda, MD, PhD, MBA Professor and Vice Chair, Director of Anatomic Pathology Director of Genitourinary Pathology Service Interim Director of Cytopathology
Testicular Germ Cell Tumors; A Simplistic Approach Merce Jorda, MD, PhD, MBA Professor and Vice Chair, Director of Anatomic Pathology Director of Genitourinary Pathology Service Interim Director of Cytopathology
Published in: Cancer Causes & Control. DOI: /s z. Document Version Peer reviewed version
 Trends in testicular germ cell cancer incidence in Australia Baade, P., Carriere, P., & Fritschi, L. (2008). Trends in testicular germ cell cancer incidence in Australia. Cancer Causes & Control, 19(10),
Trends in testicular germ cell cancer incidence in Australia Baade, P., Carriere, P., & Fritschi, L. (2008). Trends in testicular germ cell cancer incidence in Australia. Cancer Causes & Control, 19(10),
Morphologic Aspects Aspects
 Testicular Testicular Germ Germ Cell Cell Neoplasia: Neoplasia: Molecular Molecular Pathways Pathways & Challenging Morphologic Aspects Aspects George George J. J. Netto, Netto, M.D. M.D. Johns Johns Hopkins
Testicular Testicular Germ Germ Cell Cell Neoplasia: Neoplasia: Molecular Molecular Pathways Pathways & Challenging Morphologic Aspects Aspects George George J. J. Netto, Netto, M.D. M.D. Johns Johns Hopkins
Testicular Tumors: What s New, True, Important Cristina Magi-Galluzzi, MD, PhD
 Testicular Tumors: What s New, True, Important Cristina Magi-Galluzzi, MD, PhD Director, Genitourinary Pathology R.J. Tomsich Pathology & Laboratory Medicine Institute Professor of Pathology, Lerner College
Testicular Tumors: What s New, True, Important Cristina Magi-Galluzzi, MD, PhD Director, Genitourinary Pathology R.J. Tomsich Pathology & Laboratory Medicine Institute Professor of Pathology, Lerner College
Mediastinal Germ Cell Tumors
 Mediastinal Germ Cell Tumors Anja C. Roden, M.D. Department of Laboratory Medicine and Pathology, Mayo Clinic, Rochester, MN, USA 2018 MFMER slide-1 Disclosure I have no relevant financial relationships
Mediastinal Germ Cell Tumors Anja C. Roden, M.D. Department of Laboratory Medicine and Pathology, Mayo Clinic, Rochester, MN, USA 2018 MFMER slide-1 Disclosure I have no relevant financial relationships
DAX1, testes development role 7, 8 DFFRY, spermatogenesis role 49 DMRT genes, male sex differentiation role 15
 Subject Index N-Acetylcysteine, sperm quality effects 71 Ambiguous genitalia, origins 1, 2 Anti-Müllerian hormone function 13 receptors 13 Sertoli cell secretion 10, 38 Apoptosis assays in testes 73, 74
Subject Index N-Acetylcysteine, sperm quality effects 71 Ambiguous genitalia, origins 1, 2 Anti-Müllerian hormone function 13 receptors 13 Sertoli cell secretion 10, 38 Apoptosis assays in testes 73, 74
Diseases of the penis & testis
 Diseases of the penis & testis Done by : Saef B AL-Abbadi Diseases of penis, Condyloma Acuminatum A benign tumor *Tend to recur but only rarely progress into in situ or invasive cancers read this = genital
Diseases of the penis & testis Done by : Saef B AL-Abbadi Diseases of penis, Condyloma Acuminatum A benign tumor *Tend to recur but only rarely progress into in situ or invasive cancers read this = genital
HISTOPATHOLOGICAL STUDY OF TESTICULAR TUMORS IN NORTH-WEST RAJASTHAN (BIKANER REGION)
 HISTOPATHOLOGICAL STUDY OF TESTICULAR TUMORS IN NORTH-WEST RAJASTHAN (BIKANER REGION) *Rankawat Madhu Sudan, Vyas S.P. and Swami Sarika Department of Pathology, Sardar Patel Medical College, Bikaner *Author
HISTOPATHOLOGICAL STUDY OF TESTICULAR TUMORS IN NORTH-WEST RAJASTHAN (BIKANER REGION) *Rankawat Madhu Sudan, Vyas S.P. and Swami Sarika Department of Pathology, Sardar Patel Medical College, Bikaner *Author
Noninvasive Detection of Testicular Carcinoma In Situ in Semen Using OCT3/4
 european urology 54 (2008) 153 160 available at www.sciencedirect.com journal homepage: www.europeanurology.com Testis Cancer Noninvasive Detection of Testicular Carcinoma In Situ in Semen Using OCT3/4
european urology 54 (2008) 153 160 available at www.sciencedirect.com journal homepage: www.europeanurology.com Testis Cancer Noninvasive Detection of Testicular Carcinoma In Situ in Semen Using OCT3/4
Tinh hoàn
 Tinh hoàn Tinh hoàn Tinh hoàn Tiền liệt tuyến Tiền liệt tuyến Mào tinh hoàn Mào tinh hoàn Túi tinh Túi tinh Túi tinh Túi tinh So-called cystadenoma of seminal vesicle. Gross appearance of granulomatous
Tinh hoàn Tinh hoàn Tinh hoàn Tiền liệt tuyến Tiền liệt tuyến Mào tinh hoàn Mào tinh hoàn Túi tinh Túi tinh Túi tinh Túi tinh So-called cystadenoma of seminal vesicle. Gross appearance of granulomatous
Updates in Urologic Pathology WHO Made Those Changes?! Peyman Tavassoli Pathology Department BC Cancer Agency
 Updates in Urologic Pathology WHO Made Those Changes?! Peyman Tavassoli Pathology Department BC Cancer Agency World Health Organization Available in Feb 2016 Frame work for reporting Major contributing
Updates in Urologic Pathology WHO Made Those Changes?! Peyman Tavassoli Pathology Department BC Cancer Agency World Health Organization Available in Feb 2016 Frame work for reporting Major contributing
We are IntechOpen, the world s leading publisher of Open Access books Built by scientists, for scientists. International authors and editors
 We are IntechOpen, the world s leading publisher of Open Access books Built by scientists, for scientists 4,100 116,000 120M Open access books available International authors and editors Downloads Our
We are IntechOpen, the world s leading publisher of Open Access books Built by scientists, for scientists 4,100 116,000 120M Open access books available International authors and editors Downloads Our
The complex and diverse nature of teratomas causes confusion on several levels diagnostically,
 Diversity Among Teratomas, with a Historical Perspective Thomas M. Ulbright, M.D. Department of Pathology Indiana University School of Medicine Indianapolis, Indiana The complex and diverse nature of teratomas
Diversity Among Teratomas, with a Historical Perspective Thomas M. Ulbright, M.D. Department of Pathology Indiana University School of Medicine Indianapolis, Indiana The complex and diverse nature of teratomas
ACCME/Disclosures. Cribriform Lesions of the Prostate. Case
 Cribriform Lesions of the Prostate Ming Zhou, MD, PhD Departments of Pathology and Urology New York University Langone Medical Center New York, NY Ming.Zhou@NYUMC.ORG ACCME/Disclosures The USCAP requires
Cribriform Lesions of the Prostate Ming Zhou, MD, PhD Departments of Pathology and Urology New York University Langone Medical Center New York, NY Ming.Zhou@NYUMC.ORG ACCME/Disclosures The USCAP requires
N-cadherin Expression in Testicular Germ Cell and Gonadal Stromal Tumors
 381 Ivyspring International Publisher Research Paper Journal of Cancer 2012; 3: 381-389. doi: 10.7150/jca.5017 N-cadherin Expression in Testicular Germ Cell and Gonadal Stromal Tumors Daniel J. Heidenberg
381 Ivyspring International Publisher Research Paper Journal of Cancer 2012; 3: 381-389. doi: 10.7150/jca.5017 N-cadherin Expression in Testicular Germ Cell and Gonadal Stromal Tumors Daniel J. Heidenberg
Best Practice & Research Clinical Endocrinology & Metabolism
 Best Practice & Research Clinical Endocrinology & Metabolism 24 (2010) 291 310 Contents lists available at ScienceDirect Best Practice & Research Clinical Endocrinology & Metabolism journal homepage: www.elsevier.com/locate/beem
Best Practice & Research Clinical Endocrinology & Metabolism 24 (2010) 291 310 Contents lists available at ScienceDirect Best Practice & Research Clinical Endocrinology & Metabolism journal homepage: www.elsevier.com/locate/beem
Germ cell tumours UK SH. Ivo Leuschner. Kiel Pediatric Tumor Registry, Institute of Pathology University Hospital of Schleswig-Holstein Campus Kiel
 Germ cell tumours Ivo Leuschner Kiel Pediatric Tumor Registry, Institute of Pathology University Hospital of Schleswig-Holstein Campus Kiel UK SH Old histogenetic Concept of Germ cell tumours Pluripotent
Germ cell tumours Ivo Leuschner Kiel Pediatric Tumor Registry, Institute of Pathology University Hospital of Schleswig-Holstein Campus Kiel UK SH Old histogenetic Concept of Germ cell tumours Pluripotent
Extratesticular Extension of Germ Cell Tumors Preferentially Occurs at the Hilum
 Anatomic Pathology / EXTRATESTICULAR EXTENSION OF GERM CELL TUMORS Extratesticular Extension of Germ Cell Tumors Preferentially Occurs at the Hilum Sarah M. Dry, MD, and Andrew A. Renshaw, MD Key Words:
Anatomic Pathology / EXTRATESTICULAR EXTENSION OF GERM CELL TUMORS Extratesticular Extension of Germ Cell Tumors Preferentially Occurs at the Hilum Sarah M. Dry, MD, and Andrew A. Renshaw, MD Key Words:
More than 90% testicular tumors are of germ cell
 ORIGINAL ARTICLE SALL4 Is a Novel Diagnostic Marker for Testicular Germ Cell Tumors Dengfeng Cao, MD, PhD,* Jianping Li, BS,* Charles C. Guo, MD,w Robert W. Allan, MD,z and Peter A. Humphrey, MD, PhD*
ORIGINAL ARTICLE SALL4 Is a Novel Diagnostic Marker for Testicular Germ Cell Tumors Dengfeng Cao, MD, PhD,* Jianping Li, BS,* Charles C. Guo, MD,w Robert W. Allan, MD,z and Peter A. Humphrey, MD, PhD*
Age at Surgery for Undescended Testis and Risk of Testicular Cancer
 original article Age at Surgery for Undescended Testis and Risk of Testicular Cancer Andreas Pettersson, M.D., Lorenzo Richiardi, M.D., Ph.D., Agneta Nordenskjold, M.D., Ph.D., Magnus Kaijser, M.D., Ph.D.,
original article Age at Surgery for Undescended Testis and Risk of Testicular Cancer Andreas Pettersson, M.D., Lorenzo Richiardi, M.D., Ph.D., Agneta Nordenskjold, M.D., Ph.D., Magnus Kaijser, M.D., Ph.D.,
Dr Rodney Itaki Lecturer Anatomical Pathology Discipline. University of Papua New Guinea School of Medicine & Health Sciences Division of Pathology
 Neoplasia Dr Rodney Itaki Lecturer Anatomical Pathology Discipline University of Papua New Guinea School of Medicine & Health Sciences Division of Pathology General Considerations Overview: Neoplasia uncontrolled,
Neoplasia Dr Rodney Itaki Lecturer Anatomical Pathology Discipline University of Papua New Guinea School of Medicine & Health Sciences Division of Pathology General Considerations Overview: Neoplasia uncontrolled,
Pathogenesis of testicular germ cell tumours
 Reviews of Reproduction (1999) 4, 90 100 Pathogenesis of testicular germ cell tumours Leendert H. J. Looijenga and J. Wolter Oosterhuis Laboratory for Experimental Patho-Oncology, Daniel den Hoed Cancer
Reviews of Reproduction (1999) 4, 90 100 Pathogenesis of testicular germ cell tumours Leendert H. J. Looijenga and J. Wolter Oosterhuis Laboratory for Experimental Patho-Oncology, Daniel den Hoed Cancer
MAGEC2 is a sensitive and novel marker for seminoma: a tissue microarray analysis of 325 testicular germ cell tumors
 & 2011 USCAP, Inc. All rights reserved 0893-3952/11 $32.00 829 MAGEC2 is a sensitive and novel marker for seminoma: a tissue microarray analysis of 325 testicular germ cell tumors Peter K Bode*,1, André
& 2011 USCAP, Inc. All rights reserved 0893-3952/11 $32.00 829 MAGEC2 is a sensitive and novel marker for seminoma: a tissue microarray analysis of 325 testicular germ cell tumors Peter K Bode*,1, André
DISORDERS OF MALE GENITALS
 Wit JM, Ranke MB, Kelnar CJH (eds): ESPE classification of paediatric endocrine diagnosis. 9. Testicular disorders/disorders of male genitals. Horm Res 2007;68(suppl 2):63 66 ESPE Code Diagnosis OMIM ICD10
Wit JM, Ranke MB, Kelnar CJH (eds): ESPE classification of paediatric endocrine diagnosis. 9. Testicular disorders/disorders of male genitals. Horm Res 2007;68(suppl 2):63 66 ESPE Code Diagnosis OMIM ICD10
Clues to the aetiological heterogeneity of testicular seminomas and non-seminomas: time trends and age-period-cohort effects
 International Epidemiological Association 2000 Printed in Great Britain International Journal of Epidemiology 2000;29:826 831 Clues to the aetiological heterogeneity of testicular seminomas and non-seminomas:
International Epidemiological Association 2000 Printed in Great Britain International Journal of Epidemiology 2000;29:826 831 Clues to the aetiological heterogeneity of testicular seminomas and non-seminomas:
EARLY ONLINE RELEASE
 EARLY ONLINE RELEASE Note: This article was posted on the Archives Web site as an Early Online Release. Early Online Release articles have been peer reviewed, copyedited, and reviewed by the authors. Additional
EARLY ONLINE RELEASE Note: This article was posted on the Archives Web site as an Early Online Release. Early Online Release articles have been peer reviewed, copyedited, and reviewed by the authors. Additional
Neoplastic Potential of Germ Cells in Relation to Disturbances of Gonadal Organogenesis and Changes in Karyotype
 Journal of Andrology, Vol. 24, No. 2, March/April 23 Copyright American Society of Andrology Neoplastic Potential of Germ Cells in Relation to Disturbances of Gonadal Organogenesis and Changes in Karyotype
Journal of Andrology, Vol. 24, No. 2, March/April 23 Copyright American Society of Andrology Neoplastic Potential of Germ Cells in Relation to Disturbances of Gonadal Organogenesis and Changes in Karyotype
Cardiff MRCS OSCE Courses Testicular Cancer
 Testicular Cancer Scenario: A 40-year-old male presents to the surgical out-patient clinic with a 6-8 week history of a painless lump in his left scrotum. He however complains of a dull ache in the scrotum
Testicular Cancer Scenario: A 40-year-old male presents to the surgical out-patient clinic with a 6-8 week history of a painless lump in his left scrotum. He however complains of a dull ache in the scrotum
Solid Tumour Section Review
 Atlas of Genetics and Cytogenetics in Oncology and Haematology OPEN ACCESS JOURNAL AT INIST-CNRS Solid Tumour Section Review Testis: Germ cell tumors Leendert HJ Looijenga Pathology/Lab. Exp. Patho-Oncology,
Atlas of Genetics and Cytogenetics in Oncology and Haematology OPEN ACCESS JOURNAL AT INIST-CNRS Solid Tumour Section Review Testis: Germ cell tumors Leendert HJ Looijenga Pathology/Lab. Exp. Patho-Oncology,
TitleA case of metachronous bilateral te. Citation 泌尿器科紀要 (1993), 39(6):
 TitleA case of metachronous bilateral te Takashi, Munehisa; Hirata, Yoshifum Author(s) Hideo; Shimoji, Toshio; Miyake, Koj Nagasaka, Tetsuro Citation 泌尿器科紀要 (1993), 39(6): 577-580 Issue Date 1993-06 URL
TitleA case of metachronous bilateral te Takashi, Munehisa; Hirata, Yoshifum Author(s) Hideo; Shimoji, Toshio; Miyake, Koj Nagasaka, Tetsuro Citation 泌尿器科紀要 (1993), 39(6): 577-580 Issue Date 1993-06 URL
Edinburgh Research Explorer
 Edinburgh Research Explorer Intratubular germ cell neoplasia of the human testis Citation for published version: Mitchell, RT, E Camacho-Moll, M, Macdonald, J, Anderson, R, Kelnar, CJH, O'Donnell, M, Sharpe,
Edinburgh Research Explorer Intratubular germ cell neoplasia of the human testis Citation for published version: Mitchell, RT, E Camacho-Moll, M, Macdonald, J, Anderson, R, Kelnar, CJH, O'Donnell, M, Sharpe,
Protocol for the Examination of Lymphadenectomy Specimens From Patients With Malignant Germ Cell and Sex Cord-Stromal Tumors of the Testis
 Protocol for the Examination of Specimens From Patients With Malignant Germ Cell and Sex Cord-Stromal Tumors of the Testis Version: Testis 4.0.1.1 Protocol Posting Date: February 2019 Accreditation Requirements
Protocol for the Examination of Specimens From Patients With Malignant Germ Cell and Sex Cord-Stromal Tumors of the Testis Version: Testis 4.0.1.1 Protocol Posting Date: February 2019 Accreditation Requirements
GERM CELL TUMOR. Edited by Angabin Matin
 GERM CELL TUMOR Edited by Angabin Matin Germ Cell Tumor Edited by Angabin Matin Published by InTech Janeza Trdine 9, 51000 Rijeka, Croatia Copyright 2012 InTech All chapters are Open Access distributed
GERM CELL TUMOR Edited by Angabin Matin Germ Cell Tumor Edited by Angabin Matin Published by InTech Janeza Trdine 9, 51000 Rijeka, Croatia Copyright 2012 InTech All chapters are Open Access distributed
Citation for published version (APA): Lutke Holzik, M. F. (2007). Genetic predisposition to testicular cancer s.n.
 University of Groningen Genetic predisposition to testicular cancer Lutke Holzik, Martijn Frederik IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's PDF) if you wish to cite
University of Groningen Genetic predisposition to testicular cancer Lutke Holzik, Martijn Frederik IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's PDF) if you wish to cite
1. Background Gonadoblastoma (GB) is regarded as an in situ form of germ cell tumor in dysgenetic gonads (type
 Disease Markers 34 (2013) 419 424 419 DOI 10.3233/DMA-130972 IOS Press Utility of OCT3/4, TSPY and β-catenin as biological markers for gonadoblastoma formation and malignant germ cell tumor development
Disease Markers 34 (2013) 419 424 419 DOI 10.3233/DMA-130972 IOS Press Utility of OCT3/4, TSPY and β-catenin as biological markers for gonadoblastoma formation and malignant germ cell tumor development
International Society of Gynecological Pathologists Symposium 2007
 International Society of Gynecological Pathologists Symposium 2007 Anais Malpica, M.D. Department of Pathology The University of Texas M.D. Anderson Cancer Center Grading of Ovarian Cancer Histologic grade
International Society of Gynecological Pathologists Symposium 2007 Anais Malpica, M.D. Department of Pathology The University of Texas M.D. Anderson Cancer Center Grading of Ovarian Cancer Histologic grade
A COMPARATIVE STUDY OF GERM CELL KINETICS IN THE TESTES OF CHILDREN WITH UNILATERAL CRYPTORCHIDISM: A PRELIMINARY REPORT*
 FERTILITY AND STERILITY Copyright 1970 by the Williams & Wilkins Co. Vol. 21, No. 11, November 1970 Printed in U.S.A. A COMPARATIVE STUDY OF GERM CELL KINETICS IN THE TESTES OF CHILDREN WITH UNILATERAL
FERTILITY AND STERILITY Copyright 1970 by the Williams & Wilkins Co. Vol. 21, No. 11, November 1970 Printed in U.S.A. A COMPARATIVE STUDY OF GERM CELL KINETICS IN THE TESTES OF CHILDREN WITH UNILATERAL
Cell Divisions. The autosomes represent the whole body. * Male Sex Chromosomes: XY * Female Sex Chromosomes: XX
 Cell Divisions Each Cell (including gonads) has 46 chromosomes (23 pairs of chromosomes: 22 pairs of autosomes, 1 pair of sex chromosomes) which are located in the nucleus). The autosomes represent the
Cell Divisions Each Cell (including gonads) has 46 chromosomes (23 pairs of chromosomes: 22 pairs of autosomes, 1 pair of sex chromosomes) which are located in the nucleus). The autosomes represent the
Chromosome 12p abnormalities in dysgerminoma of the ovary: a FISH analysis
 & 2006 USCAP, Inc All rights reserved 0893-3952/06 $30.00 www.modernpathology.org Chromosome 12p abnormalities in dysgerminoma of the ovary: a FISH analysis Paolo Cossu-Rocca 1,2, Shaobo Zhang 1, Lawrence
& 2006 USCAP, Inc All rights reserved 0893-3952/06 $30.00 www.modernpathology.org Chromosome 12p abnormalities in dysgerminoma of the ovary: a FISH analysis Paolo Cossu-Rocca 1,2, Shaobo Zhang 1, Lawrence
CODING TUMOUR MORPHOLOGY. Otto Visser
 CODING TUMOUR MORPHOLOGY Otto Visser INTRODUCTION The morphology describes the tissue of the tumour closest to normal tissue Well differentiated tumours are closest to normal Undifferentiated tumours show
CODING TUMOUR MORPHOLOGY Otto Visser INTRODUCTION The morphology describes the tissue of the tumour closest to normal tissue Well differentiated tumours are closest to normal Undifferentiated tumours show
Carcinoma-in-situ of the Testis Is Ultrasound of the Testes Useful as a Screening Method?
 R E V I E W A R T I C L E Carcinoma-in-situ of the Testis Is Ultrasound of the Testes Useful as a Screening Method? Suzan Lenz 1 *, Aleksander Giwercman 2 Testicular germ cell cancer is preceded by a pre-invasive
R E V I E W A R T I C L E Carcinoma-in-situ of the Testis Is Ultrasound of the Testes Useful as a Screening Method? Suzan Lenz 1 *, Aleksander Giwercman 2 Testicular germ cell cancer is preceded by a pre-invasive
CHAPTER 4X. Tumours of the of the Testis Xxxand Paratesticular Tissue. steep Xxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxx
 CHPTER 4X Tumours of the of the Testis Xxxand Paratesticular Tissue Germ Xxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxx cell tumours are the most frequent and important neoplasms xxxxxxxx. at
CHPTER 4X Tumours of the of the Testis Xxxand Paratesticular Tissue Germ Xxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxx cell tumours are the most frequent and important neoplasms xxxxxxxx. at
Prof. Dr. med. Beata BODE-LESNIEWSKA Institute of Pathology and Molecular Pathology University Hospital; Zurich
 Prof. Dr. med. Beata BODE-LESNIEWSKA Institute of Pathology and Molecular Pathology University Hospital; Zurich 32 year old man 2 months history of growing left supraclavicular lymph nodes Antibiotic treatment
Prof. Dr. med. Beata BODE-LESNIEWSKA Institute of Pathology and Molecular Pathology University Hospital; Zurich 32 year old man 2 months history of growing left supraclavicular lymph nodes Antibiotic treatment
Disclosure of Relevant Financial Relationships
 Evening Specialty Conference - Genitourinary Pathology Case 2 Disclosure of Relevant Financial Relationships Sean R Williamson, MD Henry Ford Health System, Detroit, MI @Williamson_SR USCAP requires that
Evening Specialty Conference - Genitourinary Pathology Case 2 Disclosure of Relevant Financial Relationships Sean R Williamson, MD Henry Ford Health System, Detroit, MI @Williamson_SR USCAP requires that
Leukaemia 35% Lymphoma 14%
 Distribution ib ti of Cancers in Children under 15 years Leukaemia 35% Lymphoma 14% Neuroblastoma 9% Other 5% Liver 1% Retinoblastoma 3% Bone and STS 15% CNS 20% Wilms' 8% 30-40% Mortality Germ Cell Tumours
Distribution ib ti of Cancers in Children under 15 years Leukaemia 35% Lymphoma 14% Neuroblastoma 9% Other 5% Liver 1% Retinoblastoma 3% Bone and STS 15% CNS 20% Wilms' 8% 30-40% Mortality Germ Cell Tumours
To General Embryology Dr: Azza Zaki
 Introduction To General Embryology The Human Development is a continuous process that begins when an ovum from a female is fertilized by a sperm from a male. Cell division, growth and differentiation transform
Introduction To General Embryology The Human Development is a continuous process that begins when an ovum from a female is fertilized by a sperm from a male. Cell division, growth and differentiation transform
USCAP 2012: Companion Meeting of the AAOOP. Update on lacrimal gland neoplasms: Molecular pathology of interest
 USCAP 2012: Companion Meeting of the AAOOP Vancouver BC, Canada, March 17, 2012 Update on lacrimal gland neoplasms: Molecular pathology of interest Valerie A. White MD, MHSc, FRCPC Department of Pathology
USCAP 2012: Companion Meeting of the AAOOP Vancouver BC, Canada, March 17, 2012 Update on lacrimal gland neoplasms: Molecular pathology of interest Valerie A. White MD, MHSc, FRCPC Department of Pathology
2 to 3% of All New Visceral Cancers Peak Incidence is 6th Decade M:F = 2:1 Grossly is a Bright Yellow, Necrotic Mass with a Pseudocapsule
 GENITOURINARY PATHOLOGY Kathleen M. O Toole, M.D. Renal Cell Carcinoma 2 to 3% of All New Visceral Cancers Peak Incidence is 6th Decade M:F = 2:1 Grossly is a Bright Yellow Necrotic Mass Grossly is a Bright
GENITOURINARY PATHOLOGY Kathleen M. O Toole, M.D. Renal Cell Carcinoma 2 to 3% of All New Visceral Cancers Peak Incidence is 6th Decade M:F = 2:1 Grossly is a Bright Yellow Necrotic Mass Grossly is a Bright
Management of gonads in DSD
 Management of gonads in DSD Martine Cools, paediatric endocrinologist, Katja Wolffenbuttel and Piet Hoebeke, paediatric urologists, all at University Hospital Ghent, Belgium Sten Drop, paediatric endocrinologist
Management of gonads in DSD Martine Cools, paediatric endocrinologist, Katja Wolffenbuttel and Piet Hoebeke, paediatric urologists, all at University Hospital Ghent, Belgium Sten Drop, paediatric endocrinologist
Understanding the Human Karyotype Colleen Jackson Cook, Ph.D.
 Understanding the Human Karyotype Colleen Jackson Cook, Ph.D. SUPPLEMENTAL READING Nussbaum, RL, McInnes, RR, and Willard HF (2007) Thompson and Thompson Genetics in Medicine, 7th edition. Saunders: Philadelphia.
Understanding the Human Karyotype Colleen Jackson Cook, Ph.D. SUPPLEMENTAL READING Nussbaum, RL, McInnes, RR, and Willard HF (2007) Thompson and Thompson Genetics in Medicine, 7th edition. Saunders: Philadelphia.
Testicular tumours: What s new and what s
 USCAP Vancouver 2012 Testicular tumours: What s new and what s relevant? Dan Berney Introduction The rarity and complexity of testicular tumours results in unique challenges for pathologists. The difficulty
USCAP Vancouver 2012 Testicular tumours: What s new and what s relevant? Dan Berney Introduction The rarity and complexity of testicular tumours results in unique challenges for pathologists. The difficulty
Perinatal determinants of germ-cell testicular cancer in relation to histological subtypes
 British Journal of Cancer (2002) 87, 545 550 All rights reserved 0007 0920/02 $25.00 www.bjcancer.com Perinatal determinants of germ-cell testicular cancer in relation to histological subtypes L Richiardi*,1,2,
British Journal of Cancer (2002) 87, 545 550 All rights reserved 0007 0920/02 $25.00 www.bjcancer.com Perinatal determinants of germ-cell testicular cancer in relation to histological subtypes L Richiardi*,1,2,
Testicular Malignancies /8/15
 Collecting Cancer Data: Testis 2014-2015 NAACCR Webinar Series January 8, 2015 Q&A Please submit all questions concerning webinar content through the Q&A panel. Reminder: If you have participants watching
Collecting Cancer Data: Testis 2014-2015 NAACCR Webinar Series January 8, 2015 Q&A Please submit all questions concerning webinar content through the Q&A panel. Reminder: If you have participants watching
IN SUMMARY HST 071 NORMAL & ABNORMAL SEXUAL DIFFERENTIATION Fetal Sex Differentiation Postnatal Diagnosis and Management of Intersex Abnormalities
 Harvard-MIT Division of Health Sciences and Technology HST.071: Human Reproductive Biology Course Director: Professor Henry Klapholz IN SUMMARY HST 071 Title: Fetal Sex Differentiation Postnatal Diagnosis
Harvard-MIT Division of Health Sciences and Technology HST.071: Human Reproductive Biology Course Director: Professor Henry Klapholz IN SUMMARY HST 071 Title: Fetal Sex Differentiation Postnatal Diagnosis
Citation for published version (APA): Lutke Holzik, M. F. (2007). Genetic predisposition to testicular cancer s.n.
 University of Groningen Genetic predisposition to testicular cancer Lutke Holzik, Martijn Frederik IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's PDF) if you wish to cite
University of Groningen Genetic predisposition to testicular cancer Lutke Holzik, Martijn Frederik IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's PDF) if you wish to cite
Disclosure. Relevant Financial Relationship(s) None. Off Label Usage None MFMER slide-1
 Disclosure Relevant Financial Relationship(s) None Off Label Usage None 2013 MFMER slide-1 Case Presentation A 43 year old male, with partial nephrectomy for a right kidney mass 2013 MFMER slide-2 2013
Disclosure Relevant Financial Relationship(s) None Off Label Usage None 2013 MFMER slide-1 Case Presentation A 43 year old male, with partial nephrectomy for a right kidney mass 2013 MFMER slide-2 2013
Male Reproductive System
 Male Reproductive System Constitution of male reproductive system Genital gland ----testis Genital ducts epididymis / ductus deferens / urinary duct Accessory sex glands Penis prostate gland Seminal vesicle
Male Reproductive System Constitution of male reproductive system Genital gland ----testis Genital ducts epididymis / ductus deferens / urinary duct Accessory sex glands Penis prostate gland Seminal vesicle
ONCOLOGY. Csaba Bödör. Department of Pathology and Experimental Cancer Research november 19., ÁOK, III.
 ONCOLOGY Csaba Bödör Department of Pathology and Experimental Cancer Research 2018. november 19., ÁOK, III. bodor.csaba1@med.semmelweis-univ.hu ONCOLOGY Characteristics of Benign and Malignant Neoplasms
ONCOLOGY Csaba Bödör Department of Pathology and Experimental Cancer Research 2018. november 19., ÁOK, III. bodor.csaba1@med.semmelweis-univ.hu ONCOLOGY Characteristics of Benign and Malignant Neoplasms
International Journal of Research and Review E-ISSN: ; P-ISSN:
 International Journal of Research and Review www.ijrrjournal.com E-ISSN: 2349-9788; P-ISSN: 2454-2237 Case Report Rare Combinations of Testicular Mixed Germ Cell Tumors - 2 Case Reports Dr. Byna Syam Sundara
International Journal of Research and Review www.ijrrjournal.com E-ISSN: 2349-9788; P-ISSN: 2454-2237 Case Report Rare Combinations of Testicular Mixed Germ Cell Tumors - 2 Case Reports Dr. Byna Syam Sundara
International Journal of Case Reports in Medicine
 International Journal of Case Reports in Medicine Vol. 2013 (2013), Article ID 616832, 20 minipages. DOI:10.5171/2013.616832 www.ibimapublishing.com Copyright 2013 A. L. Hemalatha, Nishtha Batra, Deepthi.
International Journal of Case Reports in Medicine Vol. 2013 (2013), Article ID 616832, 20 minipages. DOI:10.5171/2013.616832 www.ibimapublishing.com Copyright 2013 A. L. Hemalatha, Nishtha Batra, Deepthi.
Case 2. Dr. Sathima Natarajan M.D. Kaiser Permanente Medical Center Sunset
 Case 2 Dr. Sathima Natarajan M.D. Kaiser Permanente Medical Center Sunset History 24 year old male presented with a 3 day history of right flank pain, sharp in nature Denies fever, chills, hematuria or
Case 2 Dr. Sathima Natarajan M.D. Kaiser Permanente Medical Center Sunset History 24 year old male presented with a 3 day history of right flank pain, sharp in nature Denies fever, chills, hematuria or
Intraductal carcinoma of the prostate on needle biopsy: histologic features and clinical significance
 & 2006 USCAP, Inc All rights reserved 0893-3952/06 $30.00 www.modernpathology.org Intraductal carcinoma of the prostate on needle biopsy: histologic features and clinical significance Charles C Guo 1 and
& 2006 USCAP, Inc All rights reserved 0893-3952/06 $30.00 www.modernpathology.org Intraductal carcinoma of the prostate on needle biopsy: histologic features and clinical significance Charles C Guo 1 and
Role of FISH in Hematological Cancers
 Role of FISH in Hematological Cancers Thomas S.K. Wan PhD,FRCPath,FFSc(RCPA) Honorary Professor, Department of Pathology & Clinical Biochemistry, Queen Mary Hospital, University of Hong Kong. e-mail: wantsk@hku.hk
Role of FISH in Hematological Cancers Thomas S.K. Wan PhD,FRCPath,FFSc(RCPA) Honorary Professor, Department of Pathology & Clinical Biochemistry, Queen Mary Hospital, University of Hong Kong. e-mail: wantsk@hku.hk
Instructions for Coding Grade for 2014+
 Instructions for Coding for 2014+ GRADE, DIFFERENTIATION OR CELL INDICATOR Item Length: 1 NAACCR Item #: 440 NAACCR Name:, Differentiation for solid tumors (Codes 1, 2, 3, 4, 9) and Cell Indicator for
Instructions for Coding for 2014+ GRADE, DIFFERENTIATION OR CELL INDICATOR Item Length: 1 NAACCR Item #: 440 NAACCR Name:, Differentiation for solid tumors (Codes 1, 2, 3, 4, 9) and Cell Indicator for
Maldescended testis in Adults. Dr. BG GAUDJI Urologist STEVE BIKO ACADEMIC HOSPITAL
 Maldescended testis in Adults Dr. BG GAUDJI Urologist STEVE BIKO ACADEMIC HOSPITAL Definitions Cryptorchid: testis neither resides nor can be manipulated into the scrotum Ectopic: aberrant course Retractile:
Maldescended testis in Adults Dr. BG GAUDJI Urologist STEVE BIKO ACADEMIC HOSPITAL Definitions Cryptorchid: testis neither resides nor can be manipulated into the scrotum Ectopic: aberrant course Retractile:
Citation for published version (APA): Lutke Holzik, M. F. (2007). Genetic predisposition to testicular cancer s.n.
 University of Groningen Genetic predisposition to testicular cancer Lutke Holzik, Martijn Frederik IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's PDF) if you wish to cite
University of Groningen Genetic predisposition to testicular cancer Lutke Holzik, Martijn Frederik IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's PDF) if you wish to cite
1 NORMAL HISTOLOGY AND METAPLASIAS
 1 NORMAL HISTOLOGY AND METAPLASIAS, MD Anatomy and Histology 1 Metaplasias 2 ANATOMY AND HISTOLOGY The female breast is composed of a branching duct system, which begins at the nipple with the major lactiferous
1 NORMAL HISTOLOGY AND METAPLASIAS, MD Anatomy and Histology 1 Metaplasias 2 ANATOMY AND HISTOLOGY The female breast is composed of a branching duct system, which begins at the nipple with the major lactiferous
CINtec p16 INK4a Staining Atlas
 CINtec p16 INK4a Staining Atlas Rating Rating Positive The rating positive will be assigned if the p16 INK4a -stained slide shows a continuous staining of cells of the basal and parabasal cell layers of
CINtec p16 INK4a Staining Atlas Rating Rating Positive The rating positive will be assigned if the p16 INK4a -stained slide shows a continuous staining of cells of the basal and parabasal cell layers of
Normal Morphology. Anatomic Considerations. Normal Urothelial Histology and Cytology
 1 Normal Morphology Anatomic Considerations The urinary tract can be divided into three regions: the kidney; the calyces, pelves and ureters (upper collecting system or upper tract); and the bladder and
1 Normal Morphology Anatomic Considerations The urinary tract can be divided into three regions: the kidney; the calyces, pelves and ureters (upper collecting system or upper tract); and the bladder and
Hans Stoop, Wim Kirkels, Gert Dohle, Ad Gillis, Michael Den Bakker, Katharina Biermann, J Wolter Oosterhuis, Leendert Looijenga
 Diagnosis of testicular carcinoma in situ, (intratubularand micro-invasive) seminoma and embryonal carcinoma using direct enzymatic alkaline phosphatase reactivity on frozen histological sections. Hans
Diagnosis of testicular carcinoma in situ, (intratubularand micro-invasive) seminoma and embryonal carcinoma using direct enzymatic alkaline phosphatase reactivity on frozen histological sections. Hans
Pathology of Lobular & Ductal Preneoplasia. Syed A Hoda, MD Weill-Cornell, New York, NY
 Pathology of Lobular & Ductal Preneoplasia Syed A Hoda, MD Weill-Cornell, New York, NY Proliferative Epithelial Changes in Breast A wide range of proliferative epithelial changes occur in the breast There
Pathology of Lobular & Ductal Preneoplasia Syed A Hoda, MD Weill-Cornell, New York, NY Proliferative Epithelial Changes in Breast A wide range of proliferative epithelial changes occur in the breast There
Ductal Proliferations of the Breast: The Good, the Bad, and the Ugly
 Ductal Proliferations of the Breast: The Good, the Bad, and the Ugly Melinda F. Lerwill, MD CRITERIA FOR DISTINGUISHING LOW-GRADE DUCTAL CARCINOMA IN SITU FROM USUAL DUCTAL HYPERPLASIA CYTOLOGY Low-grade
Ductal Proliferations of the Breast: The Good, the Bad, and the Ugly Melinda F. Lerwill, MD CRITERIA FOR DISTINGUISHING LOW-GRADE DUCTAL CARCINOMA IN SITU FROM USUAL DUCTAL HYPERPLASIA CYTOLOGY Low-grade
Fellow GU Lecture Series, Testicular Cancer. Asit Paul, MD, PhD 02/06/2018
 Fellow GU Lecture Series, 2018 Testicular Cancer Asit Paul, MD, PhD 02/06/2018 Rare cancer worldwide, approximately 1% of all male cancers There is a large difference among ethnic/racial groups. Rates
Fellow GU Lecture Series, 2018 Testicular Cancer Asit Paul, MD, PhD 02/06/2018 Rare cancer worldwide, approximately 1% of all male cancers There is a large difference among ethnic/racial groups. Rates
3/27/2017. Pulmonary Pathology Specialty Conference. Disclosure of Relevant Financial Relationships. Clinical History:
 Pulmonary Pathology Specialty Conference Saul Suster, M.D. Medical College of Wisconsin Disclosure of Relevant Financial Relationships USCAP requires that all planners (Education Committee) in a position
Pulmonary Pathology Specialty Conference Saul Suster, M.D. Medical College of Wisconsin Disclosure of Relevant Financial Relationships USCAP requires that all planners (Education Committee) in a position
Spermatogenesis. What is it and what does it look like? How do hormones regulate spermatogenesis?
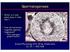 Spermatogenesis What is it and what does it look like? How do hormones regulate spermatogenesis? FSH, androgens, growth factors Animal Physiology (Hill, Wise, Anderson): Ch. 15 435-438 1 Spermatogenesis:
Spermatogenesis What is it and what does it look like? How do hormones regulate spermatogenesis? FSH, androgens, growth factors Animal Physiology (Hill, Wise, Anderson): Ch. 15 435-438 1 Spermatogenesis:
The Pathology of Germ Cell Tumours of the Ovary
 The Pathology of Germ Cell Tumours of the Ovary Professor Mike Wells University of Sheffield Amman, Jordan November 2013 Professor Francisco Paco Nogales I. Primitive germ cell tumors A. Dysgerminoma
The Pathology of Germ Cell Tumours of the Ovary Professor Mike Wells University of Sheffield Amman, Jordan November 2013 Professor Francisco Paco Nogales I. Primitive germ cell tumors A. Dysgerminoma
The Developing Testis. Physiology and Pathophysiology
 The Developing Testis. Physiology and Pathophysiology Endocrine Development Vol. 5 Series Editor Martin O. Savage London The Developing Testis Physiology and Pathophysiology Volume Editor Olle Söder Stockholm
The Developing Testis. Physiology and Pathophysiology Endocrine Development Vol. 5 Series Editor Martin O. Savage London The Developing Testis Physiology and Pathophysiology Volume Editor Olle Söder Stockholm
HPV and Head and Neck Cancer: What it means for you and your patients
 HPV and Head and Neck Cancer: What it means for you and your patients Financial Disclosure: None November 8, 2013 Steven J. Wang, MD Associate Professor Department of Otolaryngology-Head and Neck Surgery
HPV and Head and Neck Cancer: What it means for you and your patients Financial Disclosure: None November 8, 2013 Steven J. Wang, MD Associate Professor Department of Otolaryngology-Head and Neck Surgery
Embryonic Stem Cell Transcription Factor Signatures in the Diagnosis of Primary and Metastatic Germ Cell Tumors
 ORIGINAL ARTICLE Embryonic Stem Cell Transcription Factor Signatures in the Diagnosis of Primary and Metastatic Germ Cell Tumors Sandro Santagata, MD, PhD,* Keith L. Ligon, MD, PhD,*w and Jason L. Hornick,
ORIGINAL ARTICLE Embryonic Stem Cell Transcription Factor Signatures in the Diagnosis of Primary and Metastatic Germ Cell Tumors Sandro Santagata, MD, PhD,* Keith L. Ligon, MD, PhD,*w and Jason L. Hornick,
CYTOGENETICS Dr. Mary Ann Perle
 CYTOGENETICS Dr. Mary Ann Perle I) Mitosis and metaphase chromosomes A) Chromosomes are most fully condensed and clearly distinguishable during mitosis. B) Mitosis (M phase) takes 1 to 2 hrs and is divided
CYTOGENETICS Dr. Mary Ann Perle I) Mitosis and metaphase chromosomes A) Chromosomes are most fully condensed and clearly distinguishable during mitosis. B) Mitosis (M phase) takes 1 to 2 hrs and is divided
A. Incorrect! All the cells have the same set of genes. (D)Because different types of cells have different types of transcriptional factors.
 Genetics - Problem Drill 21: Cytogenetics and Chromosomal Mutation No. 1 of 10 1. Why do some cells express one set of genes while other cells express a different set of genes during development? (A) Because
Genetics - Problem Drill 21: Cytogenetics and Chromosomal Mutation No. 1 of 10 1. Why do some cells express one set of genes while other cells express a different set of genes during development? (A) Because
ARTHUR PURDY STOUT SOCIETY COMPANION MEETING: DIFFICULT NEW DIFFERENTIAL DIAGNOSES IN PROSTATE PATHOLOGY. Jonathan I. Epstein.
 1 ARTHUR PURDY STOUT SOCIETY COMPANION MEETING: DIFFICULT NEW DIFFERENTIAL DIAGNOSES IN PROSTATE PATHOLOGY Jonathan I. Epstein Professor Pathology, Urology, Oncology The Reinhard Professor of Urological
1 ARTHUR PURDY STOUT SOCIETY COMPANION MEETING: DIFFICULT NEW DIFFERENTIAL DIAGNOSES IN PROSTATE PATHOLOGY Jonathan I. Epstein Professor Pathology, Urology, Oncology The Reinhard Professor of Urological
Histological pattern of ovarian tumors and their age distribution
 Original Article Nepal Med Coll J 2008; 10(2): 81-85 Histological pattern of ovarian s and their age distribution R Jha and S Karki Department of Pathology, TUTH, Maharajgunj, Kathmandu, Nepal Corresponding
Original Article Nepal Med Coll J 2008; 10(2): 81-85 Histological pattern of ovarian s and their age distribution R Jha and S Karki Department of Pathology, TUTH, Maharajgunj, Kathmandu, Nepal Corresponding
Testicular Germ Cell Tumours: A Review of Genetics, Morphology and Immunohistochemistry
 Review Article Testicular Germ Cell Tumours: A Review of Genetics, Morphology and Immunohistochemistry DOI: NJLM/2015/13783:2046 Pathology Section Kanwardeep Kaur Tiwana, Sarita Nibhoria, Ashish Yadav,
Review Article Testicular Germ Cell Tumours: A Review of Genetics, Morphology and Immunohistochemistry DOI: NJLM/2015/13783:2046 Pathology Section Kanwardeep Kaur Tiwana, Sarita Nibhoria, Ashish Yadav,
Case Report Primary Malignancy in a Supernumerary Testicle Presenting as a Large Pelvic Mass
 Hindawi Volume 2017, Article ID 4529853, 4 pages https://doi.org/10.1155/2017/4529853 Case Report Primary Malignancy in a Supernumerary Testicle Presenting as a Large Pelvic Mass Justin Noroozian, 1 Daniel
Hindawi Volume 2017, Article ID 4529853, 4 pages https://doi.org/10.1155/2017/4529853 Case Report Primary Malignancy in a Supernumerary Testicle Presenting as a Large Pelvic Mass Justin Noroozian, 1 Daniel
Applications of IHC. Determination of the primary site in metastatic tumors of unknown origin
 Applications of IHC Determination of the primary site in metastatic tumors of unknown origin Classification of tumors that appear 'undifferentiated' by standard light microscopy Precise classification
Applications of IHC Determination of the primary site in metastatic tumors of unknown origin Classification of tumors that appear 'undifferentiated' by standard light microscopy Precise classification
2% of all malignancies Male predominance Patients usually more than 60 years old
 Benign Bladder Tumors Transitional Cell Papilloma 2-3% Inverted Papilloma Rare Malignant Transitional (Urothelial) Carcinoma 90% Carcinoma In-Situ (By Itself) 5-10% Squamous Cell Carcinoma 3-7% Adenocarcinoma
Benign Bladder Tumors Transitional Cell Papilloma 2-3% Inverted Papilloma Rare Malignant Transitional (Urothelial) Carcinoma 90% Carcinoma In-Situ (By Itself) 5-10% Squamous Cell Carcinoma 3-7% Adenocarcinoma
Teratocarcinoma In A Young Boy- An Unusual Presentation
 Human Journals Case Report November 2015 Vol.:2, Issue:1 All rights are reserved by Atia Zaka-ur-Rab et al. Teratocarcinoma In A Young Boy- An Unusual Presentation Keywords: Boy, Testicular Mass, Teratocarcinoma
Human Journals Case Report November 2015 Vol.:2, Issue:1 All rights are reserved by Atia Zaka-ur-Rab et al. Teratocarcinoma In A Young Boy- An Unusual Presentation Keywords: Boy, Testicular Mass, Teratocarcinoma
Chapter 8. Summary. Chapter 8. Summary
 Chapter 8 Chapter 8 Summary Summary Summary In CHAPTER 1 an introduction to several aspects of cervical cancer, HPV and their relation with immunology is given. It took almost one-and-a-half centuries
Chapter 8 Chapter 8 Summary Summary Summary In CHAPTER 1 an introduction to several aspects of cervical cancer, HPV and their relation with immunology is given. It took almost one-and-a-half centuries
ROLE OF PROSTATIC BASAL CELL MARKER IN DIAGNOSIS OF PROSTATIC LESIONS
 Original Research Article Pathology International Journal of Pharma and Bio Sciences ISSN 0975-6299 ROLE OF PROSTATIC BASAL CELL MARKER IN DIAGNOSIS OF PROSTATIC LESIONS SUBATHRA K* Department of pathology,
Original Research Article Pathology International Journal of Pharma and Bio Sciences ISSN 0975-6299 ROLE OF PROSTATIC BASAL CELL MARKER IN DIAGNOSIS OF PROSTATIC LESIONS SUBATHRA K* Department of pathology,
