Partial purification and biochemical characterization of amylase from Aeromonas caviaenk1isolated from Industrial waste of India
|
|
|
- Jonas Barker
- 5 years ago
- Views:
Transcription
1 Partial purification and biochemical characterization of amylase from Aeromonas caviaenk1isolated from Industrial waste of India Kalyani Khanra Department of Biotechnology, PanskuraBanamali College Panskura RS; PIN ; PurbaMedinipur; West Bengal; INDIA Indranil Choudhuri Department of Biotechnology, Panskura Banamali College Panskura RS; PIN ; Purba Medinipur; West Bengal; INDIA Sudipta Panja 1 Department of Biotechnology, Panskura Banamali College Panskura RS; PIN ; Purba Medinipur; West Bengal; INDIA Nandan Bhattacharyya (Corresponding author) Department of Biotechnology, Panskura Banamali College Panskura RS; PIN ; Purba Medinipur; West Bengal; INDIA Tel: bhattacharyya_nandan@rediffmail.com Received: September 26, 2015 Accepted: October 18, 2015 doi: /jbls.v7i URL: Abstract A partial purification and biochemical characterization of theamylase from AeromonascaviaeNK1 were carried out in this study. The extracellular extract was 85
2 concentrated using ammonium sulfate precipitation and optimum operational conditions for the enzyme activity from the strain were evaluated. The optimum ph and temperature were observed 11.5 and37 0 C respectively. Ca 2+, Mn 2+, Mg 2+, Zn 2+, Pb 2+, Co 2+ were found to have effect on amylase activity.furthermore, the analysis of kinetic showed that the enzyme has K m of 2.4mg/mL and V max of μmol/min/mg for starch. The results indicate that the enzyme reflects their potentiality towards industrial utilization. Keywords: Amylase, Partial purification, Km, V max, Zymogram 1. Introduction The enzyme of amylase familyhas a great significance due to its wide area of potential application. Amylases can be of two types, endoamylases and exoamylases. Endoamylaseshydrolyzein a random manner in the interior of starch molecules and thus can produce linear and branched oligosaccharides of various chain lengths. On the other handexoamylaseshydrolyze the starch molecules from the non-reducing end successively which results in short end products (Reddy, Nimmagadda& Rao, 2004). Amylases catalyze the hydrolysis of alpha-1,4-glycosidic linkages of polysaccharides to yield dextrin, oligosaccharides and maltose. Mainly three types of amylases are found differing in the glycoside bond they attack-α, β and γ amylases. Although they can be isolated from different source like animal, plant and microorganism, the microbial amylases are more thermostable and have higher yield(burhan et al., 2003). The production of microbial amylases from bacteria is dependent on type of strain, composition of medium, method of cultivation, cell growth, nutrient requirements, metal ions, ph, temperature, time of incubation and thermo-stability (Haq, Javed, Hameed & Adan, 2004). Spectrum of application of amylase has widened in many sectors such as food, textile, baking and detergent industries. Besides its use in the saccharification or liquefaction of starch that converts starch into fructose and glucose, the enzyme is also used as a partial replacement for the expensive malt in the brewing industry to improve flour in the baking industry. They are used for the warp sizing of textile fibers, the clarification of haze formed in beer or fruit juices and for pre-treatment of animal feed to improve digestibility (Nusrat& Rahman, 2008). Amylases have been derived from several fungi, yeast, bacteria and actinomycetes but members of the genus Bacillus are heterogeneous and they are very versatile in their adaptability to the environment.amylases have been purified earlier from various Bacillus species such as Bacillusmegaterium(Oyeleke, Auta&Egwim, 2010) from Bacillussubtilis (Riaz,Haq&Qadeer, 2003), from Bacilluslicheniformis SPT 27 (Aiyer, 2004), from Bacillus licheniformis, Bacillus amyloliquifaciens (Vidyalakshmi, Paranthaman&Indhumathi, 2009). Diverse fungal strains are able to produce amylase both intracellularly and extracellularly depending upon the fermentation process (Ellaiah,Adinarayana, Sunitha, &Bhavani Devi, 2003; Vahidi, Shafagi&Mirzabeigi, 2005; Chadha, Rubinder&Saini, 2005). In the present study we report partial purification and characterization of wide ph tolerant, amylase produced by AeromonascaviaeNK1which was isolated from industrial waste of an industrial zone in West Bengal, India. 86
3 2. Materials and Metdods 2.1 Strain A new strain of AeromonascaviaeNK1 was identified from the industrial waste. The strain was maintained in the nutrient agar slant at 4 0 C. The bacteria were revived by inoculating in LB (0.1% v/v) and incubated at 37 0 C in a shaker incubator for overnight. 2.2 Extraction and Partial Purification of Amylase Enzyme from the Isolated Bacteria The isolated bacteria were cultured in 100 ml starch containing mineral medium for 30h in shaker incubator at 120 rpm and 37 0 C. The extracellular enzyme was collected by centrifugation at 3000 X g for 20 min using cooling centrifuge (REMI C30 plus). The supernatant was collected as crude extract of enzyme. The enzyme was purified by ammonium sulphate precipitation up to 80% saturation. The precipitates were collected from different concentration of ammonium sulphate. Then the precipitates were re-suspended in 10 ml of 100mM Tris buffer of ph 10 and dialyzed overnight. 2.3 Protein Content Estimation For protein content estimation standardlowrey method of protein estimation was used (Lowry, Rosebrough, Farr & Randall, 1951). Different dilutions from 0.2 mg/ml to 0.8 mg/ml of Bovine Serum Albumin (BSA) solutions were prepared by mixing of stock BSA solution (1 mg/ ml) and distilled water in test tubes to final volume of 5ml. 0.2 mlproteinsolutionsfrom each were pipetted in fresh test tubes and 2 ml of alkaline copper sulphate reagent was added to each. The solutions were mixed well and incubated at room temperature for 10 min. 0.2 ml of FolinCiocalteau solution was then added to each tube and incubated for 30 min. The absorbance of each mixture was then measured using spectrophotometer (Bio Spectrometer, Eppendorf) at 600 nm. The absorbance was plotted against protein concentration to get a standard calibration curve. The concentration of the unknown samples was determined from the plotted standard curve. 2.4 Molecular weight determination of the purified amylase by SDS- PAGE The molecular weight of the partially purified amylase was determined by using SDS-PAGE as per description of Laemmli(Laemmli, 1970).The concentrations of staking gel and resolving gel in this experiment were 4.5% and 12.5% respectively. The purified amylase was loaded into the well parallel to standard protein marker. The protein band was detected by staining with 0.2% Coomassie brilliant Blue R250 and destining by methanol-acetic acid-water solution (4:1:5). 2.5 Zymogram of Amylase Activity For determination of Zymogram, enzyme was subjected to native gel electrophoresis with the use of 10% acrylamide gel. 0.2% soluble starch was incorporated into the separating gel. After the electrophoresis,washing the gel at room temperature in solutions containing 50 mm Na 2 HPO 4, 50 mm NaH 2 PO 4 (ph 7.2 for 1 h for activity of enzyme.gel was stained in a solution of iodine (Iodine 5 g/l, KI 50 g/l) (Lee,Morikawa, TakagiandImanakawa, 1994; Lin, 87
4 Chyau and Hsu1998). 2.6 Enzyme Assay Amylase activity was determined by measuring the release of reducing sugar from soluble starch. The reaction mixture contained 0.5 ml of crude enzyme and 1 ml of sodium phosphate buffer (ph 7.0). 1% soluble starch was then added and incubated at 25 C for 10 min. The amount of reducing sugar released in the mixture was determined by the addition of 2 ml of 3,5-dinitrosalicylic acid (Miller, 1959) followed by boiling for 10 min to develop color. The absorbance of the mixture was measured at 540 nm, and D-glucose was used to create a standard curve. One unit of enzyme activity was defined as the amount of enzyme releasing reducing sugar equivalent to 1 μmol glucose per minute under the assay condition. The effect of temperature, ph and substrate concentration on amylase activity was studied and Km and Vmax values of the enzyme were calculated from Lineweaver-Burk (double-reciprocal) plot (Mishra &Behera, 2008; Najafi, Deobagkar&Deobagkar, 2005; Sudharhsan, Senthilkumar&Ranjith, 2007). 2.7 Optimization of Enzyme Activity Amylase activity was optimized in respect of ph, temperature, substrate concentration and metal ions Effect of ph for on the activity and stability of amylase Enzyme activity at the ph range from 4 to 12 were determined by 3,5- dinitrosalicylic acid reagent method as described earlier in Effect of temperature on the activity and stability of amylase The enzyme activity (U) were measured by 3,5- dinitrosalicylic acid reagent at the temperature of 20 0 C, 30 0 C, 37 0 C, 40 0 C, 50 0 C at the optimum ph,thatwas noted earlier, for the determination of activity and the stability of the enzyme Effect of substrate concentration on the activity and stability of Amylase For the determination of the optimum substrate concentration,2, 4, 6, 8, 10 mg/ml of starch solutions were prepared. Amylase activity was measured at varying concentrations of starch under optimal conditions on the basis of the information obtained in previous experiment (optimum ph, optimum temperature) Effect of cations and heavy metal ions on the activity and stability amylase The effect of cations on enzyme activity was studies by using Calcium ( mm), Magnesium (1-7 mm), Manganese ( mm), and Zinc (0.1-1mM) ions. They have positive effect on amylase production. We used Lead (0.1-1mM) and Cobalt (0.1-1mM) to study the effect of heavy metal ions on enzyme activity. 88
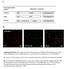
5 3. Results and Discussions 3.1 Molecular Weight of the Purified Amylase The molecular weight of the purified amylase on the basis of SDS-PAGE is ~55kDa as shown in Figure 1. The figure shows that the amylase is partially purified. Figure 1. Molecular weight of the crude enzyme determined by SDS-PAGE. S stands for partially purified sample, M stands for molecular weight marker 3.2 Zymogram The enzyme activity was observed in native PAGE gel containing 0.2% agar. A hallow zone of digested starch was detected in gelindicating activity of amylase enzyme (Figure 2). Figure 2. Zymogram of amylase activity. Proteins were loaded in Lane 1 & 4. Lanes 2 and 3 were empty lanes. 20µg of BSA was loaded in lane 1 and 20 µg of partially purified amylase was loaded in Lane 4. Amylase breakdown the starch and clear zone is appeared in Lane 4 89
6 Relative activity (%) Journal of Biology and Life Science 3.3 Enzyme Kinetics The partially purified α-amylase enzyme was incubated with various concentrations ( mg/mL) and time of soluble potato starch under 37 0 C temperature and ph 7 to determine the kinetic parameter (Roy et al., 2014). The Michaelis-Menten constant (Km) and maximum velocity (Vmax) values were determined from Lineweaver-Burk plots. The Km and Vmax values were calculated from the kinetic data. The enzyme has K m of 2.4 mg/ml and V max of μmol/min/mg for starch (Figure 3). Figure 3. Lineweaver-Burk plot for Kmand Vmax values of the α-amylase in the presence of different concentrations of soluble starch. 3.4 Optimum Condition and Stability of the Enzyme The enzyme showed 100% activity at ph 11.5 in Figure 4a and temperature 37 0 C in Figure 4b. The enzyme is also 70% to 100% active at the ph range from 7 to 12 and temperature 20 0 C to 40 0 C. It was also noted that with increasing the substrate concentration up to 1 mg/ml the activity of the enzyme increases and showed 100% efficiency. With respected to the further increase in substrate concentration there is no change in the enzyme activity. The enzyme showed wide range of ph activity. It may eliminate the requirement of ph adjustment in its industrial application which is an important factor for its industrial application ph Figure 4a. Effect of ph on the activity of purified enzyme 90
7 Figure 4b. Effect of Temperature on the activity of purified enzyme 3.5 Effect of Cationsand Heavy Metal Ions on Amylase Production The data of effect of ions on enzyme activity indicate that the relative activity is 100% in presence of 0.8 mm, 1.5mM, 0.75mM, 5mMof ZnCl 2, calcium chloride, Manganese chloride, Magnesium chloride, and respectively (Fig 5 A, B,C, D). So calcium chloride and magnesium sulfate stimulated the amylase activity. On the other hand, in presence of leadion, cobalt ions (4mM and 0.1mM respectively), relative activity of enzyme is 100 % (Fig 5 E, F). Then after increasing of cobalt ion the activity gradually decreased. So cobalt ion has an inhibitory effect of heavy metal on amylase activity. Figure 5. Effect of cations and heavy metals on the activity of purified enzyme 91
8 4. Conclusion Current results indicate that the ~55kDa amylase enzyme is active at wide range of ph and temperature. This study also revealed that amylolytic activity was affected by cation like calcium and magnesium concentration. In presence of calcium and magnesium ion in a specific range the enzyme activity increased. Lead (Pb) ion has an inhibitory effect of heavy metal on amylase activity. References Aiyer, P. D. (2005). Effect of C: N ratio on alpha amylase production by Bacillus licheniformis SPT 27. African Journal of Biotechnology, 3(10), Burhan, A., Nisa, U., Gökhan, C., Ömer, C., Ashabil, A., & Osman, G. (2003). Enzymatic properties of a novel thermostable, thermophilic, alkaline and chelator resistant amylase from an alkaliphilic Bacillus sp. isolate ANT-6. Process Biochemistry, 38(10), Chadha, B. S., Rubinder, K., & Saini, H. S. (2005). Constitutive α-amylase producing mutant and recombinant haploid strains of thermophilic fungusthermomyceslanuginosus. Folia microbiologica, 50(2), Ellaiah, P., Adinarayana, K., Sunitha, M., &Bhavani Devi, R. (2003). Isolation of α-amylase producing fungi from some soils in India. Indian Journal of Microbiology, 43(2), Haq, I., Javed, M. M., Hameed,U.,& Adnan, F. (2010).Kinetics and thermodynamic studies of alpha amylase from Bacillus lichenoformis mutant. Pak. J. Bot, 42(5), Laemmli, U. K. (1970). Cleavage of structural proteins during the assembly of the head of bacteriophage T4. nature, 227(5259), Lee, S., Morikawa, M., Takagi, M. &Imanakawa, T.(1994). Cloning aapt Gene and Characterization of its product, a-amylase, pullulanase (aapt), from Thermophilic and alkaliphilic Bacillus sp. Strain XAL601. Applied Environmental Microbiology 60, Lin, L. L., Chyau, C. C., & Hsu, W. H. (1998). Production and properties of a raw starch degrading amylase from the thermophilic and alkaliphilic Bacillus sp. TS 23. Biotechnology and Applied Biochemistry, 28(1), Lowry, O. H., Rosebrough, N. J., Farr, A. L., & Randall, R. J. (1951). Protein measurement with the Folin phenol reagent. J biolchem, 193(1), Miller, G. L. (1959). Use of dinitrosalicylic acid reagent for determination of reducing sugar. Analytical chemistry, 31(3), Mishra, S., &Behera, N. (2008). Amylase activity of a starch degrading bacteria isolated from soil receiving kitchen wastes. African Journal of Biotechnology, 7(18), Najafi, M. F., Deobagkar, D., &Deobagkar, D. (2005). Purification and characterization of an extracellular α-amylase from Bacillus subtilis AX20. Protein expression and purification, 92
9 41(2), Nusrat, A., & Rahman, S. (2010). Partial Characterization of Extracellular &alpha-amylase from Three Bacillus Isolates. Bangla. J. Microbiol., 25(1). Oyeleke, S. B., Auta, S. H., &Egwim, E. C. (2010). Production and characterization of amylase produced by Bacillus megaterium isolated from a local yam peel dumpsite in Minna, Niger State. Journal of Microbiology and Antimicrobials, 2(7), Reddy, N. S., Nimmagadda, A., & Rao, K. S. (2004). An overview of the microbial α-amylase family. African Journal of Biotechnology, 2(12), Riaz, N., Haq, I., &Qadeer, M. A. (2003). Characterization of α-amylase by Bacillus subtilis. Int J AgrBiol, 5(3), Roy, A., Khanra, K., Mishra, A., & Bhattacharyya, N. (2014). Partial purification and characterization of amylase from a newly isolated Bacillus megaterium Strain KAN1 from fermented rice Handia. American Journal of Current Microbiology, 2(1), 1-5. Sudharhsan, S., Senthilkumar, S., &Ranjith, K. (2007). Physical and nutritional factors affecting the production of amylase from species of bacillus isolated from spoiled food waste. African Journal of Biotechnology, 6(4), Vahidi, H., Shafagi, B., & Mirzabeigi, Z. (2005). Culture medium optimization of?-amylase producing organism Mocur spp. using the variable size-simplex algorithm. DARU Journal of Pharmaceutical Sciences, 13(1), Vidyalakshmi, R., Paranthaman, R., & Indhumathi, J. (2009). Amylase production on submerged fermentation by Bacillus spp.world Journal of Chemistry, 4(1), Copyright Disclaimer Copyright for this article is retained by the author(s), with first publication rights granted to the journal. This is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license ( 93
EXTRACTION OF THERMO-STABLE ALPHA AMYLASE FROM FERMENTED WHEAT BRAN
 BIOLOGIA 2001, 47 (1&2), PP 47 52 ISSN 0006 3096 EXTRACTION OF THERMO-STABLE ALPHA AMYLASE FROM FERMENTED WHEAT BRAN *HAMAD ASHRAF, IKRAM UL HAQ, AND JAVED IQBAL Biotechnology Research Laboratory, Department
BIOLOGIA 2001, 47 (1&2), PP 47 52 ISSN 0006 3096 EXTRACTION OF THERMO-STABLE ALPHA AMYLASE FROM FERMENTED WHEAT BRAN *HAMAD ASHRAF, IKRAM UL HAQ, AND JAVED IQBAL Biotechnology Research Laboratory, Department
Enhanced Production and Characterization of Commercially Important Thermostable Amylolytic Enzyme
 (An ISO 3297: 27 Certified Organization) Vol. 3, Issue 7, July 214 Enhanced Production and Characterization of Commercially Important Thermostable Amylolytic Enzyme Sarita Sharma 1, Rachana Bhatt 2 1 Master
(An ISO 3297: 27 Certified Organization) Vol. 3, Issue 7, July 214 Enhanced Production and Characterization of Commercially Important Thermostable Amylolytic Enzyme Sarita Sharma 1, Rachana Bhatt 2 1 Master
Screening of bacteria producing amylase and its immobilization: a selective approach By Debasish Mondal
 Screening of bacteria producing amylase and its immobilization: a selective approach By Debasish Mondal Article Summary (In short - What is your article about Just 2 or 3 lines) Category: Bacillus sp produce
Screening of bacteria producing amylase and its immobilization: a selective approach By Debasish Mondal Article Summary (In short - What is your article about Just 2 or 3 lines) Category: Bacillus sp produce
Isolation and Screening of Starch Hydrolising Bacteria and its Effect of Different Physiological. Parameters on Amylase Enzyme Activity
 , pp: 79-83 NOVEMBER-2015 Research Article (Open access) Isolation and Screening of Starch Hydrolising Bacteria and its Effect of Different Physiological Parameters on Amylase Enzyme Activity Prerana Min*,
, pp: 79-83 NOVEMBER-2015 Research Article (Open access) Isolation and Screening of Starch Hydrolising Bacteria and its Effect of Different Physiological Parameters on Amylase Enzyme Activity Prerana Min*,
Scholars Research Library. Purification and characterization of neutral protease enzyme from Bacillus Subtilis
 Journal of Microbiology and Biotechnology Research Scholars Research Library J. Microbiol. Biotech. Res., 2012, 2 (4):612-618 (http://scholarsresearchlibrary.com/archive.html) Purification and characterization
Journal of Microbiology and Biotechnology Research Scholars Research Library J. Microbiol. Biotech. Res., 2012, 2 (4):612-618 (http://scholarsresearchlibrary.com/archive.html) Purification and characterization
SUPPLEMENTARY MATERIAL
 SUPPLEMENTARY MATERIAL Purification and biochemical properties of SDS-stable low molecular weight alkaline serine protease from Citrullus Colocynthis Muhammad Bashir Khan, 1,3 Hidayatullah khan, 2 Muhammad
SUPPLEMENTARY MATERIAL Purification and biochemical properties of SDS-stable low molecular weight alkaline serine protease from Citrullus Colocynthis Muhammad Bashir Khan, 1,3 Hidayatullah khan, 2 Muhammad
Β-FRUCTOFURANOSIDASE ENZYME
 KINETICS ANALYSIS OF Β-FRUCTOFURANOSIDASE ENZYME 2-The effects of enzyme concentration on the rate of an enzyme catalyzed reaction. Systematic names and numbers β-fructofuranosidase (EC 3.2.1.26) Reactions
KINETICS ANALYSIS OF Β-FRUCTOFURANOSIDASE ENZYME 2-The effects of enzyme concentration on the rate of an enzyme catalyzed reaction. Systematic names and numbers β-fructofuranosidase (EC 3.2.1.26) Reactions
Experiment 1. Isolation of Glycogen from rat Liver
 Experiment 1 Isolation of Glycogen from rat Liver Figure 35: FIG-2, Liver, PAS, 100x. Note the presence of a few scattered glycogen granules (GG). Objective To illustrate the method for isolating glycogen.
Experiment 1 Isolation of Glycogen from rat Liver Figure 35: FIG-2, Liver, PAS, 100x. Note the presence of a few scattered glycogen granules (GG). Objective To illustrate the method for isolating glycogen.
Amylase Production from Potato and Banana Peel Waste
 Int.J.Curr.Microbiol.App.Sci (213) 2(11): 41-414 ISSN: 2319-776 Volume 2 Number 11 (213) pp. 41-414 http://www.ijcmas.com Original Research Article Amylase Production from Potato and Banana Peel Waste
Int.J.Curr.Microbiol.App.Sci (213) 2(11): 41-414 ISSN: 2319-776 Volume 2 Number 11 (213) pp. 41-414 http://www.ijcmas.com Original Research Article Amylase Production from Potato and Banana Peel Waste
Production of Thermostable and Ca +2 Independent α-amylases from Halphilic Bacteria
 International Journal of Biotechnology and Biochemistry ISSN 0973-2691 Volume 12, Number 2 (2016) pp. 153-159 Research India Publications http://www.ripublication.com Production of Thermostable and Ca
International Journal of Biotechnology and Biochemistry ISSN 0973-2691 Volume 12, Number 2 (2016) pp. 153-159 Research India Publications http://www.ripublication.com Production of Thermostable and Ca
Kinetics analysis of β-fructofuranosidase enzyme. 1-Effect of Time Incubation On The Rate Of An Enzymatic Reaction
 Kinetics analysis of β-fructofuranosidase enzyme 1-Effect of Time Incubation On The Rate Of An Enzymatic Reaction Enzyme kinetics It is the study of the chemical reactions that are catalyzed by enzymes.
Kinetics analysis of β-fructofuranosidase enzyme 1-Effect of Time Incubation On The Rate Of An Enzymatic Reaction Enzyme kinetics It is the study of the chemical reactions that are catalyzed by enzymes.
Production and Characterization of Bacillus firmus Pectinase
 Production and Characterization of Bacillus firmus Pectinase Anna Roosdiana, 1* Sasangka Prasetyawan, 1 Chanif Mahdi, 1 and Sutrisno 1 1 Laboratory of Biochemistry, Department of Chemistry, Faculty of
Production and Characterization of Bacillus firmus Pectinase Anna Roosdiana, 1* Sasangka Prasetyawan, 1 Chanif Mahdi, 1 and Sutrisno 1 1 Laboratory of Biochemistry, Department of Chemistry, Faculty of
Isolation and Screening of Amylase Producing Fungi
 International Journal of Current Microbiology and Applied Sciences ISSN: 2319-7706 Volume 6 Number 4 (2017) pp. 783-788 Journal homepage: http://www.ijcmas.com Original Research Article https://doi.org/10.20546/ijcmas.2017.604.098
International Journal of Current Microbiology and Applied Sciences ISSN: 2319-7706 Volume 6 Number 4 (2017) pp. 783-788 Journal homepage: http://www.ijcmas.com Original Research Article https://doi.org/10.20546/ijcmas.2017.604.098
Effect of ph on the production of protease by Fusarium oxysporum using agroindustrial waste
 Biotechnological Communication Biosci. Biotech. Res. Comm. 8(1): 78-83 (2015) Effect of ph on the production of protease by Fusarium oxysporum using agroindustrial waste Rupali R. Deshmukh and N. N. Vidhale*
Biotechnological Communication Biosci. Biotech. Res. Comm. 8(1): 78-83 (2015) Effect of ph on the production of protease by Fusarium oxysporum using agroindustrial waste Rupali R. Deshmukh and N. N. Vidhale*
INTERNATIONAL JOURNAL OF ENGINEERING SCIENCES & RESEARCH TECHNOLOGY
 [Ravish, 2(2): Feb., 2013] ISSN: 2277-9655 IJESRT INTERNATIONAL JOURNAL OF ENGINEERING SCIENCES & RESEARCH TECHNOLOGY Isolation And Characterization Of Proteolytic Bacteria And Its Protease Himani Ravish
[Ravish, 2(2): Feb., 2013] ISSN: 2277-9655 IJESRT INTERNATIONAL JOURNAL OF ENGINEERING SCIENCES & RESEARCH TECHNOLOGY Isolation And Characterization Of Proteolytic Bacteria And Its Protease Himani Ravish
Aspergillus foetidus BY AQUEOUS TWO PHASE
 33 CHAPTER 3 PARTIAL PURIFICATION OF TANNASE FROM Aspergillus foetidus BY AQUEOUS TWO PHASE EXTRACTION AND ITS CHARACTERIZATION 3.1 INTRODUCTION Partial purification of proteins in general and tannase
33 CHAPTER 3 PARTIAL PURIFICATION OF TANNASE FROM Aspergillus foetidus BY AQUEOUS TWO PHASE EXTRACTION AND ITS CHARACTERIZATION 3.1 INTRODUCTION Partial purification of proteins in general and tannase
4-The effect of sucrose concentration on the rate of reaction catalyzed by β-fructofuranosidase enzyme.
 Kinetics analysis of β-fructofuranosidase enzyme 4-The effect of sucrose concentration on the rate of reaction catalyzed by β-fructofuranosidase enzyme. One of the important parameter affecting the rate
Kinetics analysis of β-fructofuranosidase enzyme 4-The effect of sucrose concentration on the rate of reaction catalyzed by β-fructofuranosidase enzyme. One of the important parameter affecting the rate
Pelagia Research Library
 Available online at www.pelagiaresearchlibrary.com European Journal of Experimental Biology, 211, 1 (3):124-129 ISSN: 2248 9215 Production of Alkaline Protease by Bacillus subtilis (MTCC7312) using Submerged
Available online at www.pelagiaresearchlibrary.com European Journal of Experimental Biology, 211, 1 (3):124-129 ISSN: 2248 9215 Production of Alkaline Protease by Bacillus subtilis (MTCC7312) using Submerged
Studies on Glucose Isomerase from a Streptomyces Species
 APPLIED AND ENVIRONMENTAL MICROBIOLOGY, Oct. 1976, P. 489-493 Copyright ) 1976 American Society for Microbiology Vol. 32, No. 4 Printed in U.S.A. Studies on Glucose Isomerase from a Streptomyces Species
APPLIED AND ENVIRONMENTAL MICROBIOLOGY, Oct. 1976, P. 489-493 Copyright ) 1976 American Society for Microbiology Vol. 32, No. 4 Printed in U.S.A. Studies on Glucose Isomerase from a Streptomyces Species
Effect of metal ions on amylase production using Bacillus subtilis isolated from soil of Almora District, Uttarakhand, India
 Available online at www.ijpab.com Sirohi, R. and Prakash, V. Int. J. Pure App. Biosci. 3 (4): 37-41 (2015) ISSN: 2320 7051 ISSN: 2320 7051 Int. J. Pure App. Biosci. 3 (4): 37-41 (2015) INTERNATIONAL JOURNAL
Available online at www.ijpab.com Sirohi, R. and Prakash, V. Int. J. Pure App. Biosci. 3 (4): 37-41 (2015) ISSN: 2320 7051 ISSN: 2320 7051 Int. J. Pure App. Biosci. 3 (4): 37-41 (2015) INTERNATIONAL JOURNAL
AMYLOGLUCOSIDASE from ASPERGILLUS NIGER, var.
 AMYLOGLUCOSIDASE from ASPERGILLUS NIGER, var. SYNONYMS INS No. 1100 Prepared at the 59 th JECFA (2002) and published in FNP 52 Add 10 (2002), superseding tentative specifications prepared at the 55 th
AMYLOGLUCOSIDASE from ASPERGILLUS NIGER, var. SYNONYMS INS No. 1100 Prepared at the 59 th JECFA (2002) and published in FNP 52 Add 10 (2002), superseding tentative specifications prepared at the 55 th
Production and Optimization of Protease from Aspergillus niger and Bacillus subtilis using Response Surface Methodology
 IOSR Journal of Biotechnology and Biochemistry (IOSR-JBB) ISSN: 2455-264X, Volume 2, Issue 7 (Nov. Dec. 2016), PP 01-07 Production and Optimization of Protease from Aspergillus niger and Bacillus subtilis
IOSR Journal of Biotechnology and Biochemistry (IOSR-JBB) ISSN: 2455-264X, Volume 2, Issue 7 (Nov. Dec. 2016), PP 01-07 Production and Optimization of Protease from Aspergillus niger and Bacillus subtilis
Preliminary studies of cellulase production by Acinetobacter anitratus and Branhamella sp.
 frican Journal of iotechnology Vol. 6 (1), pp. 28-33, 4 January 27 vailable online at http://www.academicjournals.org/j ISSN 1684 5315 27 cademic Journals Full Length Research Paper Preliminary studies
frican Journal of iotechnology Vol. 6 (1), pp. 28-33, 4 January 27 vailable online at http://www.academicjournals.org/j ISSN 1684 5315 27 cademic Journals Full Length Research Paper Preliminary studies
SUMMARY AND CONCLUSION
 SUMMARY AND CONCLUSION A potential lipase producing marine fungus was selected among 14 lipase producers isolated from seawater and sediments of South Indian coastal environments which was identified as
SUMMARY AND CONCLUSION A potential lipase producing marine fungus was selected among 14 lipase producers isolated from seawater and sediments of South Indian coastal environments which was identified as
Pelagia Research Library. Purification and comparison properties of crude enzyme with purified α-amylase from Bacillus licheniformis ATCC 6346
 Available online at www.pelagiaresearchlibrary.com European Journal of Experimental Biology, 2011, 1 (3):58-69 ISSN: 2248 9215 Purification and comparison properties of crude enzyme with purified α-amylase
Available online at www.pelagiaresearchlibrary.com European Journal of Experimental Biology, 2011, 1 (3):58-69 ISSN: 2248 9215 Purification and comparison properties of crude enzyme with purified α-amylase
Evaluation of - amylase enzyme from Bacillus sp. isolated from various soil samples
 ISSN: 2319-7706 Volume 3 Number 10 (2014) pp. 783-789 http://www.ijcmas.com Original Research Article Evaluation of - amylase enzyme from Bacillus sp. isolated from various soil samples M.P.Prasad 1*,
ISSN: 2319-7706 Volume 3 Number 10 (2014) pp. 783-789 http://www.ijcmas.com Original Research Article Evaluation of - amylase enzyme from Bacillus sp. isolated from various soil samples M.P.Prasad 1*,
Experiment 9. NATURE OF α-amylase ACTIVITY ON STARCH
 Experiment 9 NATURE OF α-amylase ACTIVITY ON STARC In Experiment 1 we described the action of α-amylase on starch as that of catalyzing the hydrolysis of α-1,4-glucosidic bonds at random in the interior
Experiment 9 NATURE OF α-amylase ACTIVITY ON STARC In Experiment 1 we described the action of α-amylase on starch as that of catalyzing the hydrolysis of α-1,4-glucosidic bonds at random in the interior
Qualitative test of protein-lab2
 1- Qualitative chemical reactions of amino acid protein functional groups: Certain functional groups in proteins can react to produce characteristically colored products. The color intensity of the product
1- Qualitative chemical reactions of amino acid protein functional groups: Certain functional groups in proteins can react to produce characteristically colored products. The color intensity of the product
This chapter deals with the evaluation of alpha amylase inhibitory
 This chapter deals with the evaluation of alpha amylase inhibitory activity of different extracts isolated from leaves of Aloe vera L. and leaves of Azadiracta indica A Juss. collected from Bharatpur and
This chapter deals with the evaluation of alpha amylase inhibitory activity of different extracts isolated from leaves of Aloe vera L. and leaves of Azadiracta indica A Juss. collected from Bharatpur and
OPTIMISATION OF XYLOSE PRODUCTION USING XYLANASE
 Int. J. Chem. Sci.: 8(2), 2010, 909-913 OPTIMISATION OF XYLOSE PRODUCTION USING XYLANASE T. SATHISH a and N. Y. S. MURTHY * Department of Biotechnology, Malla Reddy Engineering College, HYDERABAD (A.P.)
Int. J. Chem. Sci.: 8(2), 2010, 909-913 OPTIMISATION OF XYLOSE PRODUCTION USING XYLANASE T. SATHISH a and N. Y. S. MURTHY * Department of Biotechnology, Malla Reddy Engineering College, HYDERABAD (A.P.)
Optimization for Production and Partial Purification of Laccase from Ash Gourd Peels
 International Journal of Current Microbiology and Applied Sciences ISSN: 2319-7706 Volume 6 Number 2 (2017) pp. 997-1003 Journal homepage: http://www.ijcmas.com Original Research Article http://dx.doi.org/10.20546/ijcmas.2017.602.112
International Journal of Current Microbiology and Applied Sciences ISSN: 2319-7706 Volume 6 Number 2 (2017) pp. 997-1003 Journal homepage: http://www.ijcmas.com Original Research Article http://dx.doi.org/10.20546/ijcmas.2017.602.112
Screening, Isolation and Characterization of Amylase Producing Bacteria and optimization for Production of Amylase
 IOSR Journal of Biotechnology and Biochemistry (IOSR-JBB) ISSN: 2455-264X, Volume 4, Issue 2 (Mar. - Apr 2018), PP 50-56 www.iosrjournals.org Screening, Isolation and Characterization of Amylase Producing
IOSR Journal of Biotechnology and Biochemistry (IOSR-JBB) ISSN: 2455-264X, Volume 4, Issue 2 (Mar. - Apr 2018), PP 50-56 www.iosrjournals.org Screening, Isolation and Characterization of Amylase Producing
CELLULASE from PENICILLIUM FUNICULOSUM
 CELLULASE from PENICILLIUM FUNICULOSUM Prepared at the 55th JECFA (2000) and published in FNP 52 Add 8 (2000), superseding tentative specifications prepared at the 31st JECFA (1987) and published in FNP
CELLULASE from PENICILLIUM FUNICULOSUM Prepared at the 55th JECFA (2000) and published in FNP 52 Add 8 (2000), superseding tentative specifications prepared at the 31st JECFA (1987) and published in FNP
Principles of Biotechnology INDUSTRIAL BIOTECHNOLOGY WEEKS 8+9
 Principles of Biotechnology INDUSTRIAL BIOTECHNOLOGY WEEKS 8+9 Industrial Microbiology Industrial Microorganisms and Product formation involved: 1- Use microorganisms to produce valuable commercial product
Principles of Biotechnology INDUSTRIAL BIOTECHNOLOGY WEEKS 8+9 Industrial Microbiology Industrial Microorganisms and Product formation involved: 1- Use microorganisms to produce valuable commercial product
Sequential Extraction of Plant Metabolites
 ISSN: 2319-7706 Volume 4 Number 2 (2015) pp. 33-38 http://www.ijcmas.com Original Research Article Sequential Extraction of Plant Metabolites Shankar L. Laware* PG. Department of Botany, Fergusson College
ISSN: 2319-7706 Volume 4 Number 2 (2015) pp. 33-38 http://www.ijcmas.com Original Research Article Sequential Extraction of Plant Metabolites Shankar L. Laware* PG. Department of Botany, Fergusson College
PRO G max Probiotic fermented soybean meal Benefits of PRO G max
 PRO G max Probiotic fermented soybean meal Benefits of PRO G max Probiotic bacteria > 10 10 CFU/kg High protein with low molecular weight protein approaching small peptides enhancing digestion and absorption
PRO G max Probiotic fermented soybean meal Benefits of PRO G max Probiotic bacteria > 10 10 CFU/kg High protein with low molecular weight protein approaching small peptides enhancing digestion and absorption
Statement Starch Cellulose Glycogen glycosidic bonds present polymer of α-glucose unbranched chains only only found in plants
 1 The statements in the table below refer to three polysaccharide molecules. Complete the table. If the statement is correct, place a tick ( ) in the box and if the statement is incorrect place a cross
1 The statements in the table below refer to three polysaccharide molecules. Complete the table. If the statement is correct, place a tick ( ) in the box and if the statement is incorrect place a cross
BIOL 347L Laboratory Three
 Introduction BIOL 347L Laboratory Three Osmosis in potato and carrot samples Osmosis is the movement of water molecules through a selectively permeable membrane into a region of higher solute concentration,
Introduction BIOL 347L Laboratory Three Osmosis in potato and carrot samples Osmosis is the movement of water molecules through a selectively permeable membrane into a region of higher solute concentration,
Screening of Rice Straw Degrading Microorganisms and Their Cellulase Activities
 Research 83 KKU Sci. J.37 (Supplement) 83-88 (2009) Screening of Rice Straw Degrading Microorganisms and Their Cellulase Activities Abstract Atcha Boonmee 1,2* Rice straw is one of the most abundant agricultural
Research 83 KKU Sci. J.37 (Supplement) 83-88 (2009) Screening of Rice Straw Degrading Microorganisms and Their Cellulase Activities Abstract Atcha Boonmee 1,2* Rice straw is one of the most abundant agricultural
Extraction and Characterization of Protease From Senesced Leaves of Papaya (Carica Papaya) and It s Application
 International Journal of Genetic Engineering and Biotechnology. ISSN 0974 3073 Volume 5, Number 1 (2014), pp. 29-34 International Research Publication House http://www.irphouse.com Extraction and Characterization
International Journal of Genetic Engineering and Biotechnology. ISSN 0974 3073 Volume 5, Number 1 (2014), pp. 29-34 International Research Publication House http://www.irphouse.com Extraction and Characterization
MIXED XYLANASE, β-glucanase ENZYME PREPARATION, produced by a strain of HUMICOLA INSOLENS
 MIXED XYLANASE, β-glucanase ENZYME PREPARATION, produced by a strain of HUMICOLA INSOLENS New specifications prepared at the 61st JECFA (2003) and published in FNP 52 Add 11 (2003). An ADI not specified
MIXED XYLANASE, β-glucanase ENZYME PREPARATION, produced by a strain of HUMICOLA INSOLENS New specifications prepared at the 61st JECFA (2003) and published in FNP 52 Add 11 (2003). An ADI not specified
Scholars Research Library. Amylase production by fungi isolated from Cassava processing site
 Journal of Microbiology and Biotechnology Research Scholars Research Library J. Microbiol. Biotech. Res., 14, 4 (4):3-3 (http://scholarsresearchlibrary.com/archive.html) Amylase production by fungi isolated
Journal of Microbiology and Biotechnology Research Scholars Research Library J. Microbiol. Biotech. Res., 14, 4 (4):3-3 (http://scholarsresearchlibrary.com/archive.html) Amylase production by fungi isolated
Amylase Production from Solid State Fermentation and Submerged Liquid Fermentation by Aspergillus niger
 BCSIR Available online at www.banglajol.info Bangladesh J. Sci. Ind. Res. 47(1), 99-104, 2012 Amylase Production from Solid State Fermentation and Submerged Liquid Fermentation by Aspergillus niger G.
BCSIR Available online at www.banglajol.info Bangladesh J. Sci. Ind. Res. 47(1), 99-104, 2012 Amylase Production from Solid State Fermentation and Submerged Liquid Fermentation by Aspergillus niger G.
Spectrophotometric Determination of the Kinetic Parameters of β-fructofuranosidase and the Mechanism of Inhibition by Copper (II) Sulfate
 Spectrophotometric Determination of the Kinetic Parameters of β-fructofuranosidase and the Mechanism of Inhibition by Copper (II) Sulfate Allen Zhang Tyson Miao Science One Program The University of British
Spectrophotometric Determination of the Kinetic Parameters of β-fructofuranosidase and the Mechanism of Inhibition by Copper (II) Sulfate Allen Zhang Tyson Miao Science One Program The University of British
Qualitative chemical reaction of functional group in protein
 Qualitative chemical reaction of functional group in protein Certain functional groups in proteins can react to produce characteristically colored products. The color intensity of the product formed by
Qualitative chemical reaction of functional group in protein Certain functional groups in proteins can react to produce characteristically colored products. The color intensity of the product formed by
AMYLAZYME RED TABLETS
 www.megazyme.com AMYLAZYME RED TABLETS For the measurement of cereal and microbial α-amylases T-AMZRD 08/16 Megazyme International Ireland 2016 INTRODUCTION: The level of α-amylase in wheat and barley
www.megazyme.com AMYLAZYME RED TABLETS For the measurement of cereal and microbial α-amylases T-AMZRD 08/16 Megazyme International Ireland 2016 INTRODUCTION: The level of α-amylase in wheat and barley
Microbial Production of Amylase from Cassava Waste
 Research in Pharmacy, 5: 22-3, 15 ISSN: 2231-539X www.researchinpharmacy.com Regular Article Microbial Production of Amylase from Cassava Waste Murali Vijayan 1*, Mukesh Kumar Dharmalingam Jothinathan
Research in Pharmacy, 5: 22-3, 15 ISSN: 2231-539X www.researchinpharmacy.com Regular Article Microbial Production of Amylase from Cassava Waste Murali Vijayan 1*, Mukesh Kumar Dharmalingam Jothinathan
Prerequisites Protein purification techniques and protein analytical methods. Basic enzyme kinetics.
 Case 19 Purification of Rat Kidney Sphingosine Kinase Focus concept The purification and kinetic analysis of an enzyme that produces a product important in cell survival is the focus of this study. Prerequisites
Case 19 Purification of Rat Kidney Sphingosine Kinase Focus concept The purification and kinetic analysis of an enzyme that produces a product important in cell survival is the focus of this study. Prerequisites
Screening of Aspergillus flavus and Aspergillus fumigatus strains for extracellular protease enzyme production
 Journal of Microbiology and Antimicrobials Vol. 2(7), pp. 83-87, October 2010 Available online http://www.academicjournals.org/jma ISSN 2141-2308 2010 Academic Journals Full Length Research Paper Screening
Journal of Microbiology and Antimicrobials Vol. 2(7), pp. 83-87, October 2010 Available online http://www.academicjournals.org/jma ISSN 2141-2308 2010 Academic Journals Full Length Research Paper Screening
Analysis of Polyphenoloxidase Enzyme Activity from Potato Extract Biochemistry Lab I (CHEM 4401)
 Analysis of Polyphenoloxidase Enzyme Activity from Potato Extract Biochemistry Lab I (CHEM 4401) Background Enzymes are protein molecules (primarily) that serve as biological catalysts. They are responsible
Analysis of Polyphenoloxidase Enzyme Activity from Potato Extract Biochemistry Lab I (CHEM 4401) Background Enzymes are protein molecules (primarily) that serve as biological catalysts. They are responsible
Media Optimization Studies for Enhanced Production of Serratiopeptidase
 Media Optimization Studies for Enhanced Production of Serratiopeptidase from Bacillus Licheniformis (NCIM ) Manasi J. Wagdarikar*, Anagha M. Joshi, Amir A. Shaikh SCES s Indira College of Pharmacy, Tathawade,
Media Optimization Studies for Enhanced Production of Serratiopeptidase from Bacillus Licheniformis (NCIM ) Manasi J. Wagdarikar*, Anagha M. Joshi, Amir A. Shaikh SCES s Indira College of Pharmacy, Tathawade,
Chapter PURIFICATION OF ALKALINE PROTEASES
 Chapter PURIFICATION OF ALKALINE PROTEASES E /xtracellular alkaline proteases produced by Bacillus sp. K 25 and bacillus pumilus K 242, were purified and the homogeneity was examined by electrophoresis.
Chapter PURIFICATION OF ALKALINE PROTEASES E /xtracellular alkaline proteases produced by Bacillus sp. K 25 and bacillus pumilus K 242, were purified and the homogeneity was examined by electrophoresis.
BIOL 305L Laboratory Four
 Please print Full name clearly: BIOL 305L Laboratory Four An investigation of biochemical changes to tomato fruit when it ripens Introduction According to an old country song by John Denver, the only two
Please print Full name clearly: BIOL 305L Laboratory Four An investigation of biochemical changes to tomato fruit when it ripens Introduction According to an old country song by John Denver, the only two
Case 19 Purification of Rat Kidney Sphingosine Kinase
 Case 19 Purification of Rat Kidney Sphingosine Kinase Focus concept The purification and kinetic analysis of an enzyme that produces a product important in cell survival is the focus of this study. Prerequisites
Case 19 Purification of Rat Kidney Sphingosine Kinase Focus concept The purification and kinetic analysis of an enzyme that produces a product important in cell survival is the focus of this study. Prerequisites
Research Article ISSN Vol 2/Issue 4/Oct-Dec 2012 PRAGYA RATHORE*, PRATIK SHAH, HARSHPREET CHANDOK, SATYENDRA PATEL
 THE PHOSPHATE SOLUBILIZING POTENTIAL OF CITROBACTER FREUNDII UNDER VARIOUS PHYSICOCHEMICAL CONDITIONS PRAGYA RATHORE*, PRATIK SHAH, HARSHPREET CHANDOK, SATYENDRA PATEL Sanghvi Institute of Management &
THE PHOSPHATE SOLUBILIZING POTENTIAL OF CITROBACTER FREUNDII UNDER VARIOUS PHYSICOCHEMICAL CONDITIONS PRAGYA RATHORE*, PRATIK SHAH, HARSHPREET CHANDOK, SATYENDRA PATEL Sanghvi Institute of Management &
GLYCOGEN BEFORE THE LAB YOU HAVE TO READ ABOUT:
 GLYCGEN BEFRE THE LAB YU HAVE T READ ABUT:. Glycogen structure. 2. Glycogen synthesis and degradation (reactions with structural formulas and enzymes). 3. The role of glycogen in liver and muscles. INTRDUCTIN
GLYCGEN BEFRE THE LAB YU HAVE T READ ABUT:. Glycogen structure. 2. Glycogen synthesis and degradation (reactions with structural formulas and enzymes). 3. The role of glycogen in liver and muscles. INTRDUCTIN
Partial purification and characterization of α-amylase produced by Aspergillus oryzae using spent-brewing grains
 Indian Journal of Biotechnology Vol. 4, July 2005, pp. 336-341 Partial purification and characterization of α-amylase produced by Aspergillus oryzae using spent-brewing grains Anil Kumar Patel 1, K Madhavan
Indian Journal of Biotechnology Vol. 4, July 2005, pp. 336-341 Partial purification and characterization of α-amylase produced by Aspergillus oryzae using spent-brewing grains Anil Kumar Patel 1, K Madhavan
Enzymes - Exercise 3 - Rockville
 Enzymes - Exercise 3 - Rockville Objectives -Understand the function of an enzyme. -Know what the substrate, enzyme, and the product of the reaction for this lab. -Understand how at various environments
Enzymes - Exercise 3 - Rockville Objectives -Understand the function of an enzyme. -Know what the substrate, enzyme, and the product of the reaction for this lab. -Understand how at various environments
JOURNAL OF INTERNATIONAL ACADEMIC RESEARCH FOR MULTIDISCIPLINARY Impact Factor 1.625, ISSN: , Volume 2, Issue 11, December 2014
 COMPARISON OF ACTIVITY OF PECTINASE USING AGRICULTURAL WASTE SUBSTRATES A STUDY M.P.KUSUMA* DR M.V.V.CHANDANA LAKSHMI** *Assistant Professor, RBVRR College of Pharmacy, Osmania University, India **Associate
COMPARISON OF ACTIVITY OF PECTINASE USING AGRICULTURAL WASTE SUBSTRATES A STUDY M.P.KUSUMA* DR M.V.V.CHANDANA LAKSHMI** *Assistant Professor, RBVRR College of Pharmacy, Osmania University, India **Associate
Production and Partial Purification of Amylase By Aspergillus niger Isolated from Cassava Peel
 Journal of Basic & Applied Sciences, 2014, 10, 287-291 287 Production and Partial Purification of Amylase By Aspergillus niger Isolated from Cassava Peel A.K. Lawal *, A.M. Banjoko, S.O. Olatope, F.A.
Journal of Basic & Applied Sciences, 2014, 10, 287-291 287 Production and Partial Purification of Amylase By Aspergillus niger Isolated from Cassava Peel A.K. Lawal *, A.M. Banjoko, S.O. Olatope, F.A.
International Journal of Technical Research and Applications e-issn: , Volume-2, Special Issue 5 (Nov-Dec 2014), PP.
 ISOLATION AND CHARACTERIZATION OF RAW STARCH DEGRADING AMYLASE FOR IMPROVING ENERGY EFFICIENCY OF STARCH-BASED INDUSTRY Debora Nangin 1, Aji Sutrisno 2 Department of Agricultural Product Technology University
ISOLATION AND CHARACTERIZATION OF RAW STARCH DEGRADING AMYLASE FOR IMPROVING ENERGY EFFICIENCY OF STARCH-BASED INDUSTRY Debora Nangin 1, Aji Sutrisno 2 Department of Agricultural Product Technology University
Optimization of saccharification conditions of prebiotic extracted jackfruit seeds
 Paper Code: fb005 TIChE International Conference 0 November 0, 0 at Hatyai, Songkhla THAILAND Optimization of saccharification conditions of prebiotic extracted jackfruit seeds Sininart Chongkhong *, Bancha
Paper Code: fb005 TIChE International Conference 0 November 0, 0 at Hatyai, Songkhla THAILAND Optimization of saccharification conditions of prebiotic extracted jackfruit seeds Sininart Chongkhong *, Bancha
Efficient hydrolysis of raw starch and ethanol fermentation: a novel raw starch digesting glucoamylase from Penicillium oxalicum
 DOI 10.1186/s13068-016-0636-5 Biotechnology for Biofuels RESEARCH Open Access Efficient hydrolysis of raw starch and ethanol fermentation: a novel raw starch digesting glucoamylase from Penicillium oxalicum
DOI 10.1186/s13068-016-0636-5 Biotechnology for Biofuels RESEARCH Open Access Efficient hydrolysis of raw starch and ethanol fermentation: a novel raw starch digesting glucoamylase from Penicillium oxalicum
Enzyme use for corn fuel ethanol production. Luis Alessandro Volpato Mereles
 Enzyme use for corn fuel ethanol production Luis Alessandro Volpato Mereles July 12 th, 2007 Agenda Global Biofuel Outlook Novozymes at a glance What are enzymes Using Enzymes to produce Fuel Ethanol from
Enzyme use for corn fuel ethanol production Luis Alessandro Volpato Mereles July 12 th, 2007 Agenda Global Biofuel Outlook Novozymes at a glance What are enzymes Using Enzymes to produce Fuel Ethanol from
Expression constructs
 Gene expressed in bebe3 ZmBEa Expression constructs 35S ZmBEa Pnos:Hygromycin r 35S Pnos:Hygromycin r 35S ctp YFP Pnos:Hygromycin r B -1 Chl YFP- Merge Supplemental Figure S1: Constructs Used for the Expression
Gene expressed in bebe3 ZmBEa Expression constructs 35S ZmBEa Pnos:Hygromycin r 35S Pnos:Hygromycin r 35S ctp YFP Pnos:Hygromycin r B -1 Chl YFP- Merge Supplemental Figure S1: Constructs Used for the Expression
Detection and Comparative Analysis of Amylase Activity from Leaves, Seeds and Stem of Purslane (Portulacaoleracea)
 Detection and Comparative Analysis of Amylase Activity from Leaves, Seeds and Stem of Purslane (Portulacaoleracea) Akshay Ware 1, Faiyaz K. Shaikh 2, Sanjay N. Harke 3 Department of Biotechnology, MGM
Detection and Comparative Analysis of Amylase Activity from Leaves, Seeds and Stem of Purslane (Portulacaoleracea) Akshay Ware 1, Faiyaz K. Shaikh 2, Sanjay N. Harke 3 Department of Biotechnology, MGM
Supporting Information
 Supporting Information Dauvillée et al. 10.1073/pnas.0907424106 Fig. S1. Iodine screening of the C. cohnii mutant bank. Each single colony was grown on rich-medium agar plates then vaporized with iodine.
Supporting Information Dauvillée et al. 10.1073/pnas.0907424106 Fig. S1. Iodine screening of the C. cohnii mutant bank. Each single colony was grown on rich-medium agar plates then vaporized with iodine.
EFFECT OF CARBON SOURCES ON FORMATION OF a-amylase AND GLUCOAMYLASE BY
 J. Gen. App!. Microbiol,, 21, 51-59 (1975) EFFECT OF CARBON SOURCES ON FORMATION OF a-amylase AND GLUCOAMYLASE BY CLOSTRIDIUM ACETOBUTYLICUM BURT ENSLEY, JOHN J. McHUGH, AND LARRY L. BARTON Department
J. Gen. App!. Microbiol,, 21, 51-59 (1975) EFFECT OF CARBON SOURCES ON FORMATION OF a-amylase AND GLUCOAMYLASE BY CLOSTRIDIUM ACETOBUTYLICUM BURT ENSLEY, JOHN J. McHUGH, AND LARRY L. BARTON Department
USING CENTRAL COMPOSITE DESIGNS - RESPONSE SURFACE METHODOLOGY TO OPTIMIZE INVERATSE ACTIVITY CONDITIONS FOR FRUCTOSE PRODUCTION
 Conference proceedings of Biotechnology for Green Solutions and Sustainable Environment: 8-87, 010 USING CENTRAL COMPOSITE DESIGNS - RESPONSE SURFACE METHODOLOGY TO OPTIMIZE INVERATSE ACTIVITY CONDITIONS
Conference proceedings of Biotechnology for Green Solutions and Sustainable Environment: 8-87, 010 USING CENTRAL COMPOSITE DESIGNS - RESPONSE SURFACE METHODOLOGY TO OPTIMIZE INVERATSE ACTIVITY CONDITIONS
EFFECT OF ADDITIONAL MINERAL IONS ON CITRIC ACID PRODUCTIVITY BY ASPERGILLUS NIGER NG-110
 BIOLOGIA 21, 47 (1&2), PP 59 65 ISSN 6 396 EFFECT OF ADDITIONAL MINERAL IONS ON CITRIC ACID PRODUCTIVITY BY ASPERGILLUS NIGER NG-11 SIKANDER ALI 1, IKRAM-UL-HAQ 1 AND JAVED IQBAL 2 1 Biotechnology Research
BIOLOGIA 21, 47 (1&2), PP 59 65 ISSN 6 396 EFFECT OF ADDITIONAL MINERAL IONS ON CITRIC ACID PRODUCTIVITY BY ASPERGILLUS NIGER NG-11 SIKANDER ALI 1, IKRAM-UL-HAQ 1 AND JAVED IQBAL 2 1 Biotechnology Research
-Glucan (mixed linkage), colorimetric method
 -Glucan (mixed linkage), colorimetric method Catalogue number: AK0027, 00 tests Introduction -Glucans are common components in cereals, bacteria, yeasts and mushrooms. Mixed linkage -glucans are naturally
-Glucan (mixed linkage), colorimetric method Catalogue number: AK0027, 00 tests Introduction -Glucans are common components in cereals, bacteria, yeasts and mushrooms. Mixed linkage -glucans are naturally
World Journal of Pharmaceutical and Life Sciences WJPLS
 wjpls, 2018, Vol. 4, Issue 1, 80-84 Research Article ISSN 2454-2229 Rajarajan et al. WJPLS www.wjpls.org SJIF Impact Factor: 4.223 PRODUCTION AND CHARACTERIZATION OF AMYLASE FROM ASPERGILLUS NIGER Dr.
wjpls, 2018, Vol. 4, Issue 1, 80-84 Research Article ISSN 2454-2229 Rajarajan et al. WJPLS www.wjpls.org SJIF Impact Factor: 4.223 PRODUCTION AND CHARACTERIZATION OF AMYLASE FROM ASPERGILLUS NIGER Dr.
Journal of Chemical and Pharmaceutical Research, 2012, 4(8): Research Article
 Available online wwwjocprcom Journal of Chemical and Pharmaceutical Research, 212, 4(8):388-3812 Research Article ISSN : 975-7384 CODEN(USA) : JCPRC5 Isolation and partial purification of Protease from
Available online wwwjocprcom Journal of Chemical and Pharmaceutical Research, 212, 4(8):388-3812 Research Article ISSN : 975-7384 CODEN(USA) : JCPRC5 Isolation and partial purification of Protease from
Work-flow: protein sample preparation Precipitation methods Removal of interfering substances Specific examples:
 Dr. Sanjeeva Srivastava IIT Bombay Work-flow: protein sample preparation Precipitation methods Removal of interfering substances Specific examples: Sample preparation for serum proteome analysis Sample
Dr. Sanjeeva Srivastava IIT Bombay Work-flow: protein sample preparation Precipitation methods Removal of interfering substances Specific examples: Sample preparation for serum proteome analysis Sample
N-Glycosidase F Deglycosylation Kit
 For life science research only. Not for use in diagnostic procedures. FOR IN VITRO USE ONLY. N-Glycosidase F Deglycosylation Kit Kit for the deglycosylation of asparagine-linked glycan chains on glycoproteins.
For life science research only. Not for use in diagnostic procedures. FOR IN VITRO USE ONLY. N-Glycosidase F Deglycosylation Kit Kit for the deglycosylation of asparagine-linked glycan chains on glycoproteins.
IJREAT International Journal of Research in Engineering & Advanced Technology, Volume 1, Issue 2, April-May, 2013 ISSN:
 A Comparative Overview of Ethanol Production from Cereal Grains and Potato by Enzymatic Treatment Soumitra Banerjee 1, Debalina Kundu 2 Dept of Food Technology, Techno India Saltlake, Kolkata 700091 Abstract
A Comparative Overview of Ethanol Production from Cereal Grains and Potato by Enzymatic Treatment Soumitra Banerjee 1, Debalina Kundu 2 Dept of Food Technology, Techno India Saltlake, Kolkata 700091 Abstract
Levan production by Bacillus licheniformis NS032 using sugar beet molasses Gojgic-Cvijovic Gojgic Cvijovic Gordana1*, Jakovljevic Dragica1, Kekez Bran
 Levan production by Bacillus licheniformis NS032 using sugar beet molasses Gojgic-Cvijovic Gojgic Cvijovic Gordana1*, Jakovljevic Dragica1, Kekez Branka2, Beskoski Vladimir2, Vrvic M. Miroslav2 1Institute
Levan production by Bacillus licheniformis NS032 using sugar beet molasses Gojgic-Cvijovic Gojgic Cvijovic Gordana1*, Jakovljevic Dragica1, Kekez Branka2, Beskoski Vladimir2, Vrvic M. Miroslav2 1Institute
Digestive Enzyme Lab
 Digestive Enzyme Lab Objectives 1. To describe the function of enzymes 2. To define: reactants, products, activation energy 3. To describe the enzymatic digestion of carbohydrates by salivary amylase 4.
Digestive Enzyme Lab Objectives 1. To describe the function of enzymes 2. To define: reactants, products, activation energy 3. To describe the enzymatic digestion of carbohydrates by salivary amylase 4.
Isolation, Purification, and Characterization of Horseradish Peroxidase (HRP) J. Kane, T. Schweickart
 Isolation, Purification, and Characterization of Horseradish Peroxidase (HRP) J. Kane, T. Schweickart From the Department of Chemistry, Elon University, Elon, North Carolina 27244 Running title: Analysis
Isolation, Purification, and Characterization of Horseradish Peroxidase (HRP) J. Kane, T. Schweickart From the Department of Chemistry, Elon University, Elon, North Carolina 27244 Running title: Analysis
Bioprospecting of Neem for Antimicrobial Activity against Soil Microbes
 ISSN: 2454-132X Impact factor: 4.295 (Volume3, Issue1) Available online at: www.ijariit.com Bioprospecting of Neem for Antimicrobial Activity against Soil Microbes R. Prasanna PRIST University, Tamilnadu
ISSN: 2454-132X Impact factor: 4.295 (Volume3, Issue1) Available online at: www.ijariit.com Bioprospecting of Neem for Antimicrobial Activity against Soil Microbes R. Prasanna PRIST University, Tamilnadu
Quantitative Determination of Proteins
 UV-0003 Introduction One of the easiest and most accurate spectroscopic tool for determining protein concentration is by UV-Visible spectrophotometers. The V-630 is designed for biochemical analysis and
UV-0003 Introduction One of the easiest and most accurate spectroscopic tool for determining protein concentration is by UV-Visible spectrophotometers. The V-630 is designed for biochemical analysis and
Biology 2180 Laboratory #3. Enzyme Kinetics and Quantitative Analysis
 Biology 2180 Laboratory #3 Name Introduction Enzyme Kinetics and Quantitative Analysis Catalysts are agents that speed up chemical processes and the catalysts produced by living cells are called enzymes.
Biology 2180 Laboratory #3 Name Introduction Enzyme Kinetics and Quantitative Analysis Catalysts are agents that speed up chemical processes and the catalysts produced by living cells are called enzymes.
BIOL 305L Spring 2019 Laboratory Six
 Please print Full name clearly: BIOL 305L Spring 2019 Laboratory Six Osmosis in potato and carrot samples Introduction Osmosis is the movement of water molecules through a selectively permeable membrane
Please print Full name clearly: BIOL 305L Spring 2019 Laboratory Six Osmosis in potato and carrot samples Introduction Osmosis is the movement of water molecules through a selectively permeable membrane
Isolation and screening of different isolates of Aspergillus for amylases production
 ISPUB.COM The Internet Journal of Microbiology Volume 7 Number 1 Isolation and screening of different isolates of Aspergillus for amylases production V Morya, D Yadav Citation V Morya, D Yadav.. The Internet
ISPUB.COM The Internet Journal of Microbiology Volume 7 Number 1 Isolation and screening of different isolates of Aspergillus for amylases production V Morya, D Yadav Citation V Morya, D Yadav.. The Internet
Estimation of protein using standard curve & Biochemistry analyzer. Experiment 1 & 2 BBT 314 ACh
 Estimation of protein using standard curve & Biochemistry analyzer Experiment 1 & 2 BBT 314 ACh Introduction Proteins are an abundant component in all cells, and almost all except storage proteins are
Estimation of protein using standard curve & Biochemistry analyzer Experiment 1 & 2 BBT 314 ACh Introduction Proteins are an abundant component in all cells, and almost all except storage proteins are
Amylase: a sample enzyme
 Amylase: a sample enzyme Objectives: After completion of this laboratory exercise you will be able to: 1. Explain the importance of enzymes in biology. 2. Explain the basic properties of an enzyme as a
Amylase: a sample enzyme Objectives: After completion of this laboratory exercise you will be able to: 1. Explain the importance of enzymes in biology. 2. Explain the basic properties of an enzyme as a
AMYLAZYME
 www.megazyme.com AMYLAZYME ALPHA-AMYLASE ASSAY PROCEDURE T-AMZ 05/18 For the Measurement of Cereal and Microbial α-amylases AACC Method 22-05.01 Megazyme 2018 INTRODUCTION: The level of α-amylase in wheat
www.megazyme.com AMYLAZYME ALPHA-AMYLASE ASSAY PROCEDURE T-AMZ 05/18 For the Measurement of Cereal and Microbial α-amylases AACC Method 22-05.01 Megazyme 2018 INTRODUCTION: The level of α-amylase in wheat
ENZYMES QUESTIONSHEET 1
 QUESTIONSHEET 1 The apparatus illustrated below can be used to investigate the activity of the enzyme catalase, which is found in liver. The liver tissue has been ground up and mixed with a buffer solution.
QUESTIONSHEET 1 The apparatus illustrated below can be used to investigate the activity of the enzyme catalase, which is found in liver. The liver tissue has been ground up and mixed with a buffer solution.
HEMICELLULASE from ASPERGILLUS NIGER, var.
 HEMICELLULASE from ASPERGILLUS NIGER, var. Prepared at the 55th JECFA (2000) and published in FNP 52 Add 8 (2000), superseding tentative specifications prepared at the 31st JECFA (1987) and published in
HEMICELLULASE from ASPERGILLUS NIGER, var. Prepared at the 55th JECFA (2000) and published in FNP 52 Add 8 (2000), superseding tentative specifications prepared at the 31st JECFA (1987) and published in
Isolation of raw starch hydrolysing fungi and purification of α-amylase from Geotrichum candidum CMSS06
 J.Natn.Sci.Foundation Sri Lanka 2009 37 (2):93-98 RESEARCH ARTICLE Isolation of raw starch hydrolysing fungi and purification of α-amylase from Geotrichum candidum CMSS06 S.N.T. De Silva, D.P.S.T.G. Attanayaka
J.Natn.Sci.Foundation Sri Lanka 2009 37 (2):93-98 RESEARCH ARTICLE Isolation of raw starch hydrolysing fungi and purification of α-amylase from Geotrichum candidum CMSS06 S.N.T. De Silva, D.P.S.T.G. Attanayaka
Experiment 3: Activity Determination
 Experiment 3: Activity Determination Introduction: Specific activity is a method for measuring enzymatic activity and the enzyme purity in a mixture. In order to determine the specific activity of an enzyme,
Experiment 3: Activity Determination Introduction: Specific activity is a method for measuring enzymatic activity and the enzyme purity in a mixture. In order to determine the specific activity of an enzyme,
(LM pages 91 98) Time Estimate for Entire Lab: 2.5 to 3.0 hours. Special Requirements
 Laboratory 7 Chemical Aspects of Digestion (LM pages 91 98) Time Estimate for Entire Lab: 2.5 to 3.0 hours Special Requirements Incubation. Students should start these sections at the beginning of the
Laboratory 7 Chemical Aspects of Digestion (LM pages 91 98) Time Estimate for Entire Lab: 2.5 to 3.0 hours Special Requirements Incubation. Students should start these sections at the beginning of the
Purification and Characterization of Iso-Ribonucleases from a Novel Thermophilic Fungus
 Int. J. Mol. Sci. 2014, 15, 944-957; doi:10.3390/ijms15010944 Article OPEN ACCESS International Journal of Molecular Sciences ISSN 1422-0067 www.mdpi.com/journal/ijms Purification and Characterization
Int. J. Mol. Sci. 2014, 15, 944-957; doi:10.3390/ijms15010944 Article OPEN ACCESS International Journal of Molecular Sciences ISSN 1422-0067 www.mdpi.com/journal/ijms Purification and Characterization
PFK Activity Assay Kit (Colorimetric)
 PFK Activity Assay Kit (Colorimetric) Catalog Number KA3761 100 assays Version: 02 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Background... 3 General Information...
PFK Activity Assay Kit (Colorimetric) Catalog Number KA3761 100 assays Version: 02 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Background... 3 General Information...
Glucose-6-phosphate Isomerase Activity Assay Kit (Colorimetric)
 ab155897 Glucose-6-phosphate Isomerase Activity Assay Kit (Colorimetric) Instructions for Use For the sensitive and accurate measurement of Glucose-6-phosphate Isomerase activity in various biological
ab155897 Glucose-6-phosphate Isomerase Activity Assay Kit (Colorimetric) Instructions for Use For the sensitive and accurate measurement of Glucose-6-phosphate Isomerase activity in various biological
Exam 3 Fall 2015 Dr. Stone 8:00. V max = k cat x E t. ΔG = -RT lnk eq K m + [S]
![Exam 3 Fall 2015 Dr. Stone 8:00. V max = k cat x E t. ΔG = -RT lnk eq K m + [S] Exam 3 Fall 2015 Dr. Stone 8:00. V max = k cat x E t. ΔG = -RT lnk eq K m + [S]](/thumbs/75/71985573.jpg) Exam 3 Fall 2015 Dr. Stone 8:00 Name There are 106 possible points (6 bonus points) on this exam. There are 8 pages. v o = V max x [S] k cat = kt e - ΔG /RT V max = k cat x E t ΔG = -RT lnk eq K m + [S]
Exam 3 Fall 2015 Dr. Stone 8:00 Name There are 106 possible points (6 bonus points) on this exam. There are 8 pages. v o = V max x [S] k cat = kt e - ΔG /RT V max = k cat x E t ΔG = -RT lnk eq K m + [S]
SCREENING AND PRODUCTION OF α-amylase FROM ASPERGILLUS NIGER USING ZERO VALUE MATERIAL FOR SOLID STATE FERMENTATION
 Academic Sciences International Journal of Pharmacy and Pharmaceutical Sciences ISSN- 975-1491 Vol 5, Issue 1, 13 Research Article SCREENING AND PRODUCTION OF α-amylase FROM ASPERGILLUS NIGER USING ZERO
Academic Sciences International Journal of Pharmacy and Pharmaceutical Sciences ISSN- 975-1491 Vol 5, Issue 1, 13 Research Article SCREENING AND PRODUCTION OF α-amylase FROM ASPERGILLUS NIGER USING ZERO
Purification and Characterization of Amidase from Paracoccus
 Purification and Characterization of Amidase from Paracoccus sp. SKG: Utilization of amidase inhibited whole cells for bioconversion of acrylonitrile to acrylamide By Prof. T. B. Karegoudar Department
Purification and Characterization of Amidase from Paracoccus sp. SKG: Utilization of amidase inhibited whole cells for bioconversion of acrylonitrile to acrylamide By Prof. T. B. Karegoudar Department
STORE AT 4 o C Version 3
 Instruction Manual Advanced Protein Assay Reagent (Cat. # ADV01) ORDERING INFORMATION To order by phone: (303) - 322-2254 To order by Fax: (303) - 322-2257 To order by e-mail: cserve@cytoskeleton.com Technical
Instruction Manual Advanced Protein Assay Reagent (Cat. # ADV01) ORDERING INFORMATION To order by phone: (303) - 322-2254 To order by Fax: (303) - 322-2257 To order by e-mail: cserve@cytoskeleton.com Technical
ALPHA-AMYLASE ASSAY PROCEDURE (CERALPHA METHOD) FOR THE MEASUREMENT OF PLANT AND MICROBIAL ALPHA-AMYLASES (100/200 Assays per Kit)
 www.megazyme.com ALPHA-AMYLASE ASSAY PROCEDURE (CERALPHA METHOD) K-CERA 02/17 FOR THE MEASUREMENT OF PLANT AND MICROBIAL ALPHA-AMYLASES (100/200 Assays per Kit) AOAC Method 2002.01 AACC Method 22-02.01
www.megazyme.com ALPHA-AMYLASE ASSAY PROCEDURE (CERALPHA METHOD) K-CERA 02/17 FOR THE MEASUREMENT OF PLANT AND MICROBIAL ALPHA-AMYLASES (100/200 Assays per Kit) AOAC Method 2002.01 AACC Method 22-02.01
