A high phylloquinone intake is required to achieve maximal osteocalcin -carboxylation 1 3
|
|
|
- Kerry Harrison
- 6 years ago
- Views:
Transcription
1 A high phylloquinone intake is required to achieve maximal osteocalcin -carboxylation 1 3 Neil C Binkley, Diane C Krueger, Tisha N Kawahara, Jean A Engelke, Richard J Chappell, and John W Suttie ABSTRACT Background: Dietary vitamin K is usually inadequate to maximize serum osteocalcin -carboxylation. Phylloquinone supplementation increases osteocalcin -carboxylation; however, the amount required to maximize carboxylation is not known. Objective: This study assessed the ability of various doses of phylloquinone (vitamin K 1 ) to facilitate osteocalcin -carboxylation. Design: Healthy adults aged y participated in 2 substudies. In an initial dose-finding study (substudy A), 6 women and 4 men received a placebo daily for 1 wk and then phylloquinone daily for 3 wk: 500, 1000, and 2000 g during weeks 2, 3, and 4, respectively. Osteocalcin and undercarboxylated osteocalcin were measured at baseline and after each week of supplementation. Subsequently, to further delineate the -carboxylation response of osteocalcin to various doses of vitamin K, 58 women and 42 men were randomly assigned to receive placebo or phylloquinone supplementation (250, 375, 500, and 1000 g/d) for 2 wk (substudy B). The percentage of undercarboxylated osteocalcin (%ucoc) was measured at baseline and weeks 1 and 2. Results: In substudy A, %ucoc decreased with phylloquinone supplementation (P < ); a greater reduction was observed with 1000 and 2000 g than with 500 g(p < 0.05). In substudy B, %ucoc decreased in all supplemented groups by week 1 (P for the trend < ), which was sustained through week 2. Phylloquinone supplementation decreased %ucoc dose-dependently; %ucoc was significantly different between the 250- g and the placebo groups and between the and 500- g groups but not between the 250-, 375-, and 500- g groups. Conclusion: A daily phylloquinone intake of 1000 g is required to maximally -carboxylate circulating osteocalcin. Am J Clin Nutr 2002;76: KEY WORDS Vitamin K, phylloquinone, osteocalcin, undercarboxylated osteocalcin, -carboxylation INTRODUCTION Vitamin K is a required cofactor for the carboxylase enzyme that converts glutamyl to -carboxyglutamyl (Gla) residues in a small number of proteins (1, 2). With vitamin K inadequacy, less than maximal -carboxylation of these proteins is achieved (3 6). Thus, measurement of undercarboxylated proteins, such as osteocalcin, is a sensitive measure of vitamin K status (7). For the vitamin K dependent proteins, conversion of glutamyl to -carboxyglutamyl conveys the ability to bind calcium ions and is essential for biological activity (8). Because 3 vitamin K dependent proteins (osteocalcin, matrix Gla-protein, and protein S) are present in bone (9) and may play a role in bone metabolism (10, 11), impaired function could potentially have adverse skeletal consequences. Specifically, subclinical vitamin K insufficiency might contribute to the development of osteoporosis (12). Data to support this possibility include epidemiologic observations that a low vitamin K intake is associated with increased hip fracture risk (13, 14). Additionally, some, but not all, reports find antagonism of vitamin K by clinical use of anticoagulants to be associated with low bone mineral density and increased fracture risk (15 18). Furthermore, less than maximal -carboxylation of circulating osteocalcin is common (6, 19 21) and has been reported to correlate with low bone mass (22, 23) and increased hip fracture risk (23 25). However, associations do not necessarily imply causation; alternatively, these indicators of vitamin K insufficiency might simply be surrogate markers of general dietary deficiency (9). Thus, it is not known whether low vitamin K status contributes to bone loss. However, if subclinical vitamin K insufficiency causes adverse skeletal consequences, it is reasonable that this would reflect impaired -carboxylation of vitamin K dependent bone proteins, because the only known role of vitamin K is as a cofactor for the carboxylase enzyme in the synthesis of -carboxyglutamyl, ie, -carboxyglutamic acid (1, 2). Thus, it is plausible that a higher vitamin K intake might improve bone health by enhancing -carboxylation. However, the dose of supplemental vitamin K needed to maximally carboxylate bone proteins, including osteocalcin the most abundant noncollagenous bone matrix protein is unknown. Thus, the purpose of this study was to determine the minimum amount of supplemental phylloquinone (vitamin K 1 ) required to maximize the carboxylation status of serum osteocalcin in healthy young adults as measured by a hydroxyapatite binding assay. SUBJECTS AND METHODS Subjects This study was approved by the University of Wisconsin Health Sciences Institutional Review Board, and informed consent was obtained from all volunteers. All participants were 1 From the Institute on Aging, Department of Medicine (NCB, DCK, and TNK), Department of Biochemistry (JAE and JWS), and Department of Biostatistics (RJC), University of Wisconsin, Madison. 2 Supported in part by NIH grant AG00801 and a grant from Roche Vitamins, Inc, Parsippany, NJ. 3 Address reprint requests to NC Binkley, 2245 MSC, 1300 University Avenue, Madison, WI nbinkley@facstaff.wisc.edu. Received March 23, Accepted for publication October 24, Am J Clin Nutr 2002;76: Printed in USA American Society for Clinical Nutrition 1055
2 1056 BINKLEY ET AL TABLE 1 Demographic data by substudy group and dose of phylloquinone Substudy group and phylloquinone dose Age 1 BMI 1 Compliance No. of subjects replaced y kg/m 2 % Substudy A (n = 4 M, 6 F) 25.8 (24.2, 27.4) 22.3 (21.1, 23.5) 98 2 Substudy B Placebo (n = 9 M, 11 F) 25.8 (24.2, 24.5) 23.3 (21.9, 24.6) g (n = 10 M, 10 F) 25.5 (23.6, 27.5) 25.1 (22.6, 27.7) g (n = 4 M, 16 F) 26.0 (24.3, 27.7) 22.3 (20.9, 23.7) g (n = 10 M, 10 F) 24.5 (23.2, 25.8) 24.1 (22.6, 25.5) g (n = 9 M, 11 F) 26.2 (24.2, 28.3) 25.1 (22.3, 27.8) x ; 95% CI in parentheses. There were no significant differences between groups. healthy volunteers recruited from southern Wisconsin. Eligible subjects were required to have normal screening laboratory values, including a complete blood count, prothrombin time, international normalized ratio, and serum chemistry panel. Volunteers with renal or hepatic disease or a history of malabsorption were excluded. In addition, individuals treated with warfarin or orlistat in the past 4 wk and those consuming foods containing olestra > 3 times/wk were excluded. Six women and 4 men aged y (x ± SD: 25.8 ± 2.9 y) were enrolled in the initial dose-finding study (substudy A). Subsequently, 100 subjects (58 women and 42 men) aged y (25.6 ± 4.0 y) participated in substudy B. Study design Substudy A was conducted to delineate the doses to be used in substudy B. In this 4-wk, open-label study, participants received a placebo daily for the first week followed by doses of 500, 1000, and 2000 g during weeks 2, 3, and 4, respectively. The placebo and phylloquinone tablets were identical in appearance in both substudy A and substudy B and were supplied in light-shielded containers by Roche Vitamins, Inc (Parsippany, NJ). All tablets were composed of > 86% microcrystalline cellulose; magnesium stearate, dry white color, and dry phylloquinone made up the remainder. Because vitamin K is fat soluble, the subjects were instructed to ingest all preparations with their evening meal to maximize absorption. Serum for the measurement of osteocalcin and undercarboxylated osteocalcin (ucoc) concentrations was obtained at baseline and after weeks 1, 2, 3, and 4 of the study. Substudy B was designed to identify the lowest dose of phylloquinone required to maximally -carboxylate osteocalcin. In this single-blind, placebo-controlled, 2-wk trial, the subjects received daily doses of placebo or 250, 375, 500, or 1000 g phylloquinone. The subjects were randomly assigned to receive 3 tablets containing placebo, 125, or 500 g phylloquinone to attain their assigned dose. As in substudy A, the participants were instructed to consume the study preparation with their evening meal. Serum was obtained at baseline and at 1 and 2 wk of treatment for the measurement of osteocalcin, ucoc, and phylloquinone concentrations. These specimens were obtained by routine venipuncture between 0800 and 1100 after a fast of 8 h. Specimens were allowed to clot for 30 min at room temperature while shielded from light and were then centrifuged (200 g, 15 min, room temperature) and quick-frozen in liquid nitrogen. The samples were subsequently maintained at 80 C until thawed for analysis. Subjects in both substudies were asked to provide an estimate of phylloquinone intake by recording in a daily log the number of servings (defined as 0.5 cup) of spinach, lettuce, Brussels sprouts, and broccoli consumed each day. These 4 foods were selected because they provide a large percentage of the total daily phylloquinone intake (26). In addition, to minimize habitual differences in dietary phylloquinone intake, the participants were asked to restrict their diet to one serving or less of the same high vitamin K containing foods each day. Furthermore, to minimize factors that might impair vitamin K absorption, the subjects were asked to avoid olestra-containing products. Compliance was calculated weekly by counting tablets; noncompliant subjects (defined as those who ingested < 85% of the supplement) were allowed to complete the study; however, the data for these subjects were excluded and replacement subjects were enrolled. This approach was taken to minimize the likelihood of compliance falsification. Assays Serum osteocalcin was determined by immunoradiometric assay (ELSA-OSTEO; CisBio International, Gif-sur-Yvette France). ucoc concentrations were determined by using a modification of the hydroxyapatite binding assay (27). Briefly, 0.5 ml serum was treated with 25 mg hydroxyapatite (no. 4280; Mallinkrodt, Inc, Paris, KY) and rotated end over end for 30 min at 4 C. The samples were then centrifuged at g for 5 min at room temperature. The supernatant fluid was removed and analyzed for osteocalcin by immunoradiometric assay. The percentage of ucoc (%ucoc) was calculated as the ratio of unadsorbed osteocalcin (ie, the amount remaining in the supernatant fluid) to total osteocalcin multiplied by 100. Serum phylloquinone concentrations were determined by HPLC separation with the use of fluorescence detection (28). Statistical analysis The groups were defined by substudy (A or B) and treatment (placebo or phylloquinone). The data from subjects in substudy B were analyzed by using a linear regression, with the dose of phylloquinone as a factor. Serum phylloquinone and %ucoc outcomes were transformed to the logarithmic scale to reduce the influence of outliers. Thus, the changes in phylloquinone concentrations and in %ucoc from baseline to week 1 are multiplicative factors rather than differences. The P values were adjusted for multiple comparisons via the Bonferroni method (in this case, multiplied by 4) (29). The test for trend with dose was also performed by fitting a linear regression but with dose considered a continuous predictor. Variables at baseline and after dose increments (substudy A) were examined for significant differences by using a one-way analysis of variance. As above, P values were adjusted by using the Bonferroni method.
3 PHYLLOQUINONE AND MAXIMUM OSTEOCALCIN CARBOXYLATION 1057 TABLE 2 Baseline laboratory values by substudy group and phylloquinone dose 1 Substudy group and Estimated phylloquinone dose Albumin Creatinine ALT Phylloquinone ucoc phylloquinone intake 2 g/l µmol/l U/L nmol/l % µg/d Substudy A (n = 4 M, 6 F) 44 (41.8, 46.2) 79.6 (69.0, 90.2) 20 (12.2, 27.8) NE 7.5 (6.3, 8.7) 109 (77, 140) Substudy B Placebo (n = 9 M, 11 F) 44 (43.1, 45.6) 79.6 (75.1, 84.9) 20 (17.6, 23.0) 0.70 (0.42, 0.98) 7.2 (6.0, 8.4) 85 (57, 113) 250 g (n = 10 M, 10 F) 43 (41.3, 44.2) 79.6 (73.4, 83.1) 19 (15.1, 22.6) 0.61 (0.41, 0.82) 7.9 (6.3, 9.6) 77 (59, 96) 375 g (n = 4 M, 16 F) 43 (41.8, 44.2) 79.6 (70.7, 82.2) 14 (12.7, 15.8) 1.00 (0.61, 1.32) 6.5 (5.5, 7.4) 120 (92, 147) 500 g (n = 10 M, 10 F) 44 (42.6, 45.3) 79.6 (75.1, 87.5) 22 (16.3, 26.7) 0.83 (0.53, 1.13) 8.7 (7.5, 9.8) 94 (75, 113) 1000 g (n = 9 M, 11 F) 44 (42.3, 45.5) 79.6 (72.5, 83.1) 17 (15.1, 19.9) 0.81 (0.59, 1.04) 8.3 (6.7, 10.0) 77 (60, 94) 1 x ; 95% CI in parentheses. ALT, alanine aminotransferase; ucoc, undercarboxylated osteocalcin; NE, not evaluated. There were no significant differences between groups by one-way ANOVA with Bonferroni correction. 2 Estimated by self-report of spinach, broccoli, Brussels sprouts, and lettuce consumption. RESULTS Subjects The demographic characteristics and laboratory results did not differ significantly between groups (Tables 1 and 2). Compliance and adherence were excellent in all groups (Table 1). Five subjects were replaced (2 in substudy A and 3 in substudy B) because of inadequate compliance. No differences in compliance between treatment weeks was observed (data not shown). Substudy A The percentage of ucoc decreased (P < ) from 7.48% at baseline to 2.49%, 1.90%, and 1.75% after sequential supplementation for 1 wk with 500, 1000, and 2000 g phylloquinone/d, respectively (Figure 1). Both the and g/d doses reduced %ucoc more (P < 0.05) than did the 500- g dose. However, %ucoc did not differ after supplementation with 1000 or 2000 g/d. Total serum osteocalcin concentrations were unaffected by phylloquinone supplementation at any dose (data not shown). On the basis of these data, daily doses of phylloquinone between 250 and 1000 g were used in substudy B. Substudy B An increase in serum phylloquinone concentration was observed from baseline to both weeks 1 and 2 in all phylloquinone-supplemented groups (Figure 2). The changes in serum phylloquinone from baseline to week 1 are shown in Table 3. Subjects who received 250 g/d on average had serum phylloquinone concentrations at week 1 that were 3.2 times those at baseline; the true value was likely times the baseline value. Serum phylloquinone concentrations increased with increasing doses of phylloquinone (P < for trend); the greatest increase ( 10-fold, from nmol/l at baseline to nmol/l at week 2) was observed in the subjects who received 1000 g/d. The increase in serum phylloquinone observed with doses of 250, 375, and 500 g/d did not differ (Table 3). In addition, the reduction in %ucoc reflected the change in serum phylloquinone (Figure 3). Specifically, the change in FIGURE 1. Mean percentage of serum undercarboxylated osteocalcin (ucoc) in subjects in substudy A after incremental phylloquinone doses of 250, 500, 1000, and 2000 g/d. Both the and g/d doses reduced ucoc more than did the 500- g/d dose (P < 0.05). No significant difference was observed between the and g/d doses. Bars indicate 95% CIs. * Significantly different from placebo, P < (oneway ANOVA with Bonferroni adjustment). n = 10. FIGURE 2. Mean serum phylloquinone concentrations in subjects in substudy B after placebo ( ) and supplementation with 250 ( ), 375 ( ), 500 ( ), and 1000 ( ) g phylloquinone/d. Concentrations in all supplemented groups were different from those of the placebo group at week 1 (P for trend ), with subsequent stability or further increases in most groups. The greatest increase was observed with 1000 g/d. Bars indicate 95% CIs. n = 20 in all groups.
4 1058 BINKLEY ET AL TABLE 3 Substudy B: changes in serum phylloquinone concentrations from baseline to week 1 by phylloquinone dose 1 Phylloquinone dose group Change Placebo 1.18 (0.85, 1.15) 250 g 3.20 (2.24, 4.57) g 2.91 (2.04, 4.16) 500 g 3.62 (2.53, 5.17) 1000 g 8.18 (5.73, 11.68) 3 1 x ; 95% CI in parentheses. n = 20 per group. The changes are multiplicative factors. 2 Significantly different from placebo, P < (multiple comparisons 3 Significantly different from 500 g, P < 0.01 (multiple comparisons TABLE 4 Substudy B: changes in the percentage of undercarboxylated osteocalcin from baseline to week 1 by phylloquinone dose 1 Phylloquinone dose group Change Placebo 0.99 (0.85, 1.15) 250 g 0.54 (0.46, 0.63) g 0.45 (0.38, 0.52) 500 g 0.44 (0.37, 0.51) 1000 g 0.26 (0.22, 0.30) 3 1 x ; 95% CI in parentheses. n = 20 per group. The changes are multiplicative factors. 2 Significantly different from placebo, P < (multiple comparisons 3 Significantly different from 500 g, P < 0.01 (multiple comparisons %ucoc from baseline to week 1 are shown in Table 4, which depicts changes in %ucoc from baseline to week 1. For example, the subjects who received 250 g/d had serum %ucoc values at week 1 that were only 0.54 times baseline values; on average, the true value was likely to be times the baseline value. Serum %ucoc decreased with increasing doses of phylloquinone supplementation (P < for trend); the greatest reduction was observed with the dose of 1000 g/d. The reduction in %ucoc observed with the doses of 250, 375, and 500 g/d did not differ significantly (Table 4). Total serum osteocalcin concentrations were unaffected by phylloquinone supplementation at any dose (Table 5). DISCUSSION In the current study, circulating osteocalcin was 92% -carboxylated in the specific assay we used. The absolute number is assay dependent; however, it is comparable with prior findings in healthy young persons (6). As was anticipated, supplementation increased circulating phylloquinone concentrations and improved osteocalcin carboxylation, measured as a reduction in %ucoc. Furthermore, whereas daily supplementation with 250 g phylloquinone increased osteocalcin -carboxylation to 96%, 1000 g supplemental phylloquinone/d is required to achieve maximal osteocalcin carboxylation. Although current data are limited, some (30, 31), but not all, (32) studies suggest that the bioavailability of dietary phylloquinone is substantially lower than supplement phylloquinone. As such, to obtain an amount of phylloquinone similar to that supplemented in the current study, ingestion of g phylloquinone/d would be required (4, 30). Because current dietary phylloquinone consumption is often in the range of g/d (33), it seems unlikely that the major increase required to achieve maximal osteocalcin -carboxylation would be attainable by diet alone. Thus, if maximal osteocalcin carboxylation is found to be beneficial in preventing osteoporosis, routine phylloquinone supplementation will probably be required. This study suggests that daily doses of 1000 g phylloquinone may be optimal in studies evaluating the skeletal effects of vitamin K. Previous skeletal studies used supplemental phylloquinone doses of 200 (34), 1000 (20, 35, 36), or (37) g/d. Thus, whereas most previous studies used optimal amounts of phylloquinone, 200 g/d might be insufficient. In addition, many studies have shown beneficial skeletal effects of high doses ( g/d) of vitamin K 2 (menatetrenone) (38 40). These doses are much higher than those required for maximal osteocalcin carboxylation by phylloquinone. Therefore, it seems probable that the vitamin K 2 studies evaluated a pharmacologic effect rather than the physiologic replacement of a deficit. It seems prudent to heed the adage vitamins taken in large doses TABLE 5 Substudy B: changes in total osteocalcin concentrations from baseline to week 1 by phylloquinone dose 1 FIGURE 3. Mean percentage of serum undercarboxylated osteocalcin (ucoc) in subjects in substudy B after placebo ( ) and supplementation with 250 ( ), 375 ( ), 500 ( ), and 1000 ( ) g phylloquinone/d. Values decreased (P for trend < ) in all supplemented groups. The greatest decrease was observed with 1000 g/d. Bars indicate 95% CIs. n = 20 in all groups. Phylloquinone dose group Change Placebo 1.04 (0.97, 1.11) 250 g 1.05 (0.98, 1.13) 375 g 0.93 (0.87, 1.00) 500 g 0.96 (0.89, 1.03) 1000 g 0.99 (0.92, 1.06) 1 x ; 95% CI in parentheses. n = 20 per group. The changes are multiplicative factors. There were no significant differences between groups.
5 PHYLLOQUINONE AND MAXIMUM OSTEOCALCIN CARBOXYLATION 1059 should be considered as drugs (41) rather than assume that vitamin K 2 mediates its effects via improved -carboxylation status. Furthermore, these high-dose vitamin K 2 studies are probably not directly applicable to the dietary requirements of phylloquinone. That this study was limited to healthy young adults must be considered a limitation. Whether maximal osteocalcin -carboxylation is achieved with an intake of 1000 g/d in elderly persons remains unknown. However, in a previous study, a reduction in %ucoc with phylloquinone supplementation was similar in young adults and those aged 65 y (6). Limiting study to a young adult population may have additionally confounded the assessment of a phylloquinone-induced effect on total osteocalcin concentrations. Specifically, our previous study showed that phylloquinone supplementation resulted in a reduction in total osteocalcin concentration, an observation not replicated in the current study. These divergent results may reflect the large degree of between-subject variability produced by high values in some of the young men in this study. It should be emphasized that the physiologic importance of maximizing osteocalcin carboxylation is unknown. Whereas it is reasonable that vitamin K might have skeletal effects mediated via -carboxylation, it is far from clear that vitamin K insufficiency contributes to bone loss or fracture. Although studies correlate low vitamin K intakes or elevated ucoc with low bone mass and increased hip fracture risk, it is possible that these observations are not causally related; low vitamin K intake may simply be a marker of general dietary inadequacy (5). Finally, even if the undercarboxylation of osteocalcin does produce adverse skeletal effects, it is not known whether increasing carboxylation slightly (in the current study from 92% to 98%) would produce beneficial effects. Thus, routine vitamin K supplementation to optimize osteocalcin carboxylation should not be recommended at this time. In summary, this study showed that 1000 g phylloquinone is required to maximally -carboxylate circulating osteocalcin.although there is abundant evidence that less than maximal osteocalcin -carboxylation is extremely common and that dietary phylloquinone supplementation can improve this situation, it remains unknown whether maximal osteocalcin -carboxylation is necessary for optimal skeletal health. Further evaluation of the effect of phylloquinone supplementation is required. Until beneficial skeletal effects are shown by such studies, routine phylloquinone supplementation is unwarranted. REFERENCES 1. Suttie JW. Warfarin and vitamin K. Clin Cardiol 1990;13: Furie B, Bouchard BA, Furie BC. Vitamin K dependent biosynthesis of gamma-carboxyglutamic acid. Blood 1999;93: Sokoll LJ, Sadowski JA. Comparison of biochemical indexes for assessing vitamin K nutritional status in a healthy adult population. Am J Clin Nutr 1996;63: Sokoll LJ, Booth SL, O Brien ME, Davidson KW, Tsaioun KI, Sadowski JA. Changes in serum osteocalcin, plasma phylloquinone, and urinary -carboxyglutamic acid in response to altered intakes of dietary phylloquinone in human subjects. Am J Clin Nutr 1997;65: Jie KS, Hamulyak K, Gijsbers BL, Roumen FJ, Vermeer C. Serum osteocalcin as a marker for vitamin K-status in pregnant women and their newborn babies. Thromb Haemost 1992;68: Binkley NC, Krueger DC, Engelke JA, Foley AL, Suttie JS. Vitamin K supplementation reduces serum concentrations of under-gammacaraboxylated osteocalcin in healthy young and elderly adults. Am J Clin Nutr 2000;72: Rucker RB. Improved functional endpoints for use in vitamin K assessment: important implications for bone disease. Am J Clin Nutr 1997;65: Esmon CT, Suttie JW, Jackson CM. The functional significance of vitamin K action. Difference in phospholipid binding between normal and abnormal prothrombin. J Biol Chem 1975;250: Binkley NC, Suttie JW. Vitamin K nutrition and osteoporosis. J Nutr 1995;125: Booth SL, Gundberg CM, McKeown NM, Morse MO, Wood RJ. Vitamin K depletion increases bone turnover. J Bone Miner Res 1999; 14(suppl):S393 (abstr). 11. Ducy P, Desbois C, Boyce B, et al. Increased bone formation in osteocalcin-deficient mice. Nature 1996;382: Weber P. The role of vitamins in the prevention of osteoporosis a brief status report. Int J Vitam Nutr Res 1999;69: Feskanich D, Weber P, Willett WC, Rockett H, Booth SL, Colditz GA. Vitamin K intake and hip fractures in women: a prospective study. Am J Clin Nutr 1999;69: Booth SL, Tucker KL, Chen H, et al. Dietary vitamin K intakes are associated with hip fracture but not with bone mineral density in elderly men and women. Am J Clin Nutr 2000;71: Jamal SA, Browner WS, Bauer DC, Cummings SR. Warfarin use and risk for osteoporosis in elderly women. Ann Intern Med 1998;128: Caraballo PJ, Gabriel SE, Castro MR, Atkinson EJ, Melton LJ. Changes in bone density after exposure to oral anticoagulants: a metaanalysis. Osteoporos Int 1999;9: Caraballo PJ, Heit JA, Atkinson EJ, et al. Long-term use of oral anticoagulants and the risk of fracture. Arch Intern Med 1999;159: Rosen HN, Maitland LA, Suttie JW, Manning WJ, Glynn RJ, Greenspan SL. Vitamin K and maintenance of skeletal integrity in adults. Am J Med 1993;94: Plantalech L, Chapuy MC, Guillaumont M, Chapuy P, Leclerq M, Delmas PD. Impaired carboxylation of serum osteocalcin in elderly women: effect of vitamin K 1 treatment. In: Christiansen C, Overgaard K, eds. Osteoporosis. Copenhagen: Osteopress, 1990: Knapen MHJ, Hamulyak K, Vermeer C. The effect of vitamin K supplementation on circulating osteocalcin (bone Gla protein) and urinary calcium excretion. Ann Intern Med 1989;111: Knapen MH, Jie KS, Hamulyak K, Vermeer C. Vitamin K induced changes in markers for osteoblast activity and urinary calcium loss. Calcif Tissue Int 1993;53: Vergnaud P, Garnero P, Meunier PJ, Breart G, Kamihagi K, Delmas PD. Undercarboxylated osteocalcin measured with a specific immunoassay predicts hip fracture in elderly women: The EPIDOS study. J Clin Endocrinol Metab 1997;82: Szulc P, Arlot M, Chapuy MC, Duboeuf F, Meunier PJ, Delmas PD. Serum undercarboxylated osteocalcin correlates with hip bone mineral density in elderly women. J Bone Miner Res 1994;9: Szulc P, Chapuy MC, Meunier PJ, Delmas PD. Serum undercarboxylated osteocalcin is a marker of the risk of hip fracture in elderly women. J Clin Invest 1993;91: Szulc P, Chapuy MC, Meunier PJ, Delmas PD. Serum undercarboxylated osteocalcin is a marker of the risk of hip fracture: a three year follow-up study. Bone 1996;18: Booth SL, Pennington JAT, Sadowski JA. Food sources and dietary intakes of vitamin K-1 (phylloquinone) in the American diet: data from the FDA total diet study. J Am Diet Assoc 1996;96: Price PA, Epstein DJ, Lothringer JW, Nishimoto SK, Poser JW, Williamson MK. Structure and function of the vitamin K dependent protein of bone. In: Suttie JW, ed. Vitamin K metabolism and vitamin K dependent proteins. Baltimore: University Park Press, 1980:
6 1060 BINKLEY ET AL 28. Haroon Y, Bacon DS, Sadowski JA. Liquid chromatographic determination of vitamin K 1 in plasma with fluorometric detection. Clin Chem 1986;32: Armitage P, Colton T, eds. The encyclopedia of biostatistics. New York: Wiley, Garber AK, Binkley NC, Krueger DC, Suttie JW. Comparison of phylloquinone bioavailability from food sources or a supplement in human subjects. J Nutr 1999;129: Gijsbers BLM, Jie KS, Vermeer C. Effect of food composition on vitamin K absorption in human volunteers. Br J Nutr 1996;76: Booth SL, O Brien-Morse ME, Dallal GE, Davidson KW, Gundberg CM. Response of vitamin K status to different intakes and sources of phylloquinone-rich foods: comparison of younger and older adults. Am J Clin Nutr 1999;70: Booth SL, Suttie JW. Dietary intake and adequacy of vitamin K. J Nutr 1998;128: Shearer MJ, Harvey JM, Mole P, McMurdo MET, Paterson CR, Bolton-Smith C. Can increased dietary intakes of vitamin K help to prevent osteoporosis? An ongoing intervention trial in the UK. Osteoporosis and vitamin K. Seoul, Korea: Intermedd Inc, 1999: Douglas AS, Robins SP, Hutchison JD, Poter RW, Stewart A, Reid DM. Carboxylation of osteocalcin in postmenopausal osteoporotic women following vitamin K and D supplementation. Bone 1995;17: Jie KS, Gijsbers BL, Knapen MH, Hamulyak K, Frank HL, Vermeer C. Effects of vitamin K and oral anticoagulants on urinary calcium excretion. Br J Haematol 1993;83: Craciun AM, Wolf J, Knapen MH, Brouns F, Vermeer C. Improved bone metabolism in female elite athletes after vitamin K supplementation. Int J Sports Med 1998;19: Orimo H, Shiraki M, Fujita T, Onomura T, Inoue T, Kushida K. Clinical evaluation of menatetrenone in the treatment of involutional osteoporosis a double-blind multicenter comparative study with 1 hydroxyvitamin D. J Bone Miner Res 1992;7:S122 (abstr). 39. Orimo H, Shiraki M, Tomita A, Morii H, Fujita T, Ohata M. Effects of menatetrenone on the bone and calcium metabolism in osteoporosis: a double-blind placebo-controlled study. J Bone Miner Metab 1998;16: Shiraki M, Shiraki Y, Aoki C, Miura M. Vitamin K 2 (menatetrenone) effectively prevents fractures and sustains lumbar bone mineral density in osteoporosis. J Bone Miner Res 2000;15: Thurman JE, Mooradian AD. Vitamin supplementation therapy in the elderly. Drugs Aging 1997;11:
Vitamin K status of healthy Japanese women: age-related vitamin K requirement for -carboxylation of osteocalcin 1 3
 Vitamin K status of healthy Japanese women: age-related vitamin K requirement for -carboxylation of osteocalcin 1 3 Naoko Tsugawa, Masataka Shiraki, Yoshitomo Suhara, Maya Kamao, Kiyoshi Tanaka, and Toshio
Vitamin K status of healthy Japanese women: age-related vitamin K requirement for -carboxylation of osteocalcin 1 3 Naoko Tsugawa, Masataka Shiraki, Yoshitomo Suhara, Maya Kamao, Kiyoshi Tanaka, and Toshio
Vitamin K intake and hip fractures in women: a prospective study 1 3
 Vitamin K intake and hip fractures in women: a prospective study 1 3 Diane Feskanich, Peter Weber, Walter C Willett, Helaine Rockett, Sarah L Booth, and Graham A Colditz ABSTRACT Background: Vitamin K
Vitamin K intake and hip fractures in women: a prospective study 1 3 Diane Feskanich, Peter Weber, Walter C Willett, Helaine Rockett, Sarah L Booth, and Graham A Colditz ABSTRACT Background: Vitamin K
Vitamin K intake and bone mineral density in women and men 1 4
 Vitamin K intake and bone mineral density in women and men 1 4 Sarah L Booth, Kerry E Broe, David R Gagnon, Katherine L Tucker, Marian T Hannan, Robert R McLean, Bess Dawson-Hughes, Peter WF Wilson, L
Vitamin K intake and bone mineral density in women and men 1 4 Sarah L Booth, Kerry E Broe, David R Gagnon, Katherine L Tucker, Marian T Hannan, Robert R McLean, Bess Dawson-Hughes, Peter WF Wilson, L
Efficacy of Vitamin K as a supplement in Osteoporosis
 2014; 2 (6): 25-29 ISSN: 2321-9122 www.biosciencejournals.com EJBB 2014; 2 (6): 25-29 Received: 05-12-2014 Accepted: 23-12-2014 Fazeel ZA Senior Resident, Dept. of Pharmacology, Gandhi Medical College,
2014; 2 (6): 25-29 ISSN: 2321-9122 www.biosciencejournals.com EJBB 2014; 2 (6): 25-29 Received: 05-12-2014 Accepted: 23-12-2014 Fazeel ZA Senior Resident, Dept. of Pharmacology, Gandhi Medical College,
Epidemiological evidence has shown associations between. Effect of Vitamin K Supplementation on Bone Loss in Elderly Men and Women
 ORIGINAL ARTICLE Endocrine Care Effect of Vitamin K Supplementation on Bone Loss in Elderly Men and Women Sarah L. Booth, Gerard Dallal, M. Kyla Shea, Caren Gundberg, James W. Peterson, and Bess Dawson-Hughes
ORIGINAL ARTICLE Endocrine Care Effect of Vitamin K Supplementation on Bone Loss in Elderly Men and Women Sarah L. Booth, Gerard Dallal, M. Kyla Shea, Caren Gundberg, James W. Peterson, and Bess Dawson-Hughes
Age Group and Sex Do Not Influence Responses of Vitamin K Biomarkers to Changes in Dietary Vitamin K 1 3
 The Journal of Nutrition Nutrient Requirements and Optimal Nutrition Age Group and Sex Do Not Influence Responses of Vitamin K Biomarkers to Changes in Dietary Vitamin K 1 3 Jennifer T. Truong, 4 Xueyan
The Journal of Nutrition Nutrient Requirements and Optimal Nutrition Age Group and Sex Do Not Influence Responses of Vitamin K Biomarkers to Changes in Dietary Vitamin K 1 3 Jennifer T. Truong, 4 Xueyan
NEW MK-7 SELECT INTRODUCTION
 NEW MK-7 SELECT INTRODUCTION As we have all known for several years now, the presence of optimal amounts of vitamin D is essential for maintaining optimal bone health and bone calcification, as noted by
NEW MK-7 SELECT INTRODUCTION As we have all known for several years now, the presence of optimal amounts of vitamin D is essential for maintaining optimal bone health and bone calcification, as noted by
Is warfarin usage a risk factor for osteoporotic fractures? A cohort study in the emergency department
 Is warfarin usage a risk factor for osteoporotic fractures? A cohort study in the emergency department Genady Drozdinsky 1, Jonah B. Cohen 2, Zipora Shohat 3,4, Shachaf Shiber-Ofer 3,5, Alon Grossman 3,6
Is warfarin usage a risk factor for osteoporotic fractures? A cohort study in the emergency department Genady Drozdinsky 1, Jonah B. Cohen 2, Zipora Shohat 3,4, Shachaf Shiber-Ofer 3,5, Alon Grossman 3,6
British Journal of Nutrition
 (2009), 102, 1171 1178 q The Authors 2009 doi:10.1017/s0007114509382100 The effect of menaquinone-7 (vitamin K 2 ) supplementation on osteocalcin carboxylation in healthy prepubertal children Marieke J.
(2009), 102, 1171 1178 q The Authors 2009 doi:10.1017/s0007114509382100 The effect of menaquinone-7 (vitamin K 2 ) supplementation on osteocalcin carboxylation in healthy prepubertal children Marieke J.
Evaluation of a Bead-based Enzyme Immunoassay for the Rapid Detection of Osteocalcin in Human Serum
 Clinical Chemistry 46:2 252 257 (2000) Endocrinology and Metabolism Evaluation of a Bead-based Enzyme Immunoassay for the Rapid Detection of Osteocalcin in Human Serum Alexandra M. Crăciun, 1 Cees Vermeer,
Clinical Chemistry 46:2 252 257 (2000) Endocrinology and Metabolism Evaluation of a Bead-based Enzyme Immunoassay for the Rapid Detection of Osteocalcin in Human Serum Alexandra M. Crăciun, 1 Cees Vermeer,
Vitamin K. Amina Ziyad Elaf Sohaib
 Vitamin K Amina Ziyad Elaf Sohaib What is Vitamin K? Fat soluble compound Necessary for the synthesis of several proteins required for blood clotting 1) Vit K 1 (Phylloquinone) - natural form - found in
Vitamin K Amina Ziyad Elaf Sohaib What is Vitamin K? Fat soluble compound Necessary for the synthesis of several proteins required for blood clotting 1) Vit K 1 (Phylloquinone) - natural form - found in
Biochemistry of Vitamin K
 Lecture 4 Biochemistry of Vitamin K The Objectives Types and chemistry of vitamin K Sources and daily requirements Functions Synthesis of γ-carboxyglutamate in: Prothrombin and blood clotting factors Osteocalcin
Lecture 4 Biochemistry of Vitamin K The Objectives Types and chemistry of vitamin K Sources and daily requirements Functions Synthesis of γ-carboxyglutamate in: Prothrombin and blood clotting factors Osteocalcin
Vitamin K Snapshot Monograph
 vitamins minerals nutrients Vitamin K Snapshot Monograph Vitamin K1 and Vitamin K2 Most Frequent Reported Uses: Blood clotting Anticoagulant reversal; bleeding disorders Osteoporosis prevention Arthritis,
vitamins minerals nutrients Vitamin K Snapshot Monograph Vitamin K1 and Vitamin K2 Most Frequent Reported Uses: Blood clotting Anticoagulant reversal; bleeding disorders Osteoporosis prevention Arthritis,
Low serum and bone vitamin K status in patients with longstanding Crohn s disease: another pathogenetic factor of osteoporosis in Crohn s disease?
 Gut 2001;48:473 477 473 Department of Gastroenterology and Hepatology, University Hospital Maastricht, Maastricht, the Netherlands E J Schoon M C A Müller R-J M Brummer R W Stockbrügger Department of Biochemistry,
Gut 2001;48:473 477 473 Department of Gastroenterology and Hepatology, University Hospital Maastricht, Maastricht, the Netherlands E J Schoon M C A Müller R-J M Brummer R W Stockbrügger Department of Biochemistry,
NIH Public Access Author Manuscript Ann Rheum Dis. Author manuscript; available in PMC 2008 November 18.
 NIH Public Access Author Manuscript Published in final edited form as: Ann Rheum Dis. 2008 November ; 67(11): 1570 1573. doi:10.1136/ard.2008.094771. Vitamin K in hand osteoarthritis: results from a randomised
NIH Public Access Author Manuscript Published in final edited form as: Ann Rheum Dis. 2008 November ; 67(11): 1570 1573. doi:10.1136/ard.2008.094771. Vitamin K in hand osteoarthritis: results from a randomised
ORIGINAL ARTICLE. Abstract. Introduction
 ORIGINAL ARTICLE Response of Serum Carboxylated and Undercarboxylated Osteocalcin to Risedronate Monotherapy and Combined Therapy with Vitamin K2 in Corticosteroid-Treated Patients: A Pilot Study Takayoshi
ORIGINAL ARTICLE Response of Serum Carboxylated and Undercarboxylated Osteocalcin to Risedronate Monotherapy and Combined Therapy with Vitamin K2 in Corticosteroid-Treated Patients: A Pilot Study Takayoshi
The most important human clinical trials on Vitamin K2
 References update The most important human clinical trials on Vitamin K2 REFERENCES UPDATE Cardiovascular Health Bone Health Kidney Health Other (Efficacy, Dose Finding, Absorption, etc.) Beneficial Effects
References update The most important human clinical trials on Vitamin K2 REFERENCES UPDATE Cardiovascular Health Bone Health Kidney Health Other (Efficacy, Dose Finding, Absorption, etc.) Beneficial Effects
Sultan Qaboos University. College of Agricultural & Marine Sciences. Vitamin K. Lecture Summary
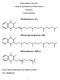 Sultan Qaboos University College of Agricultural & Marine Sciences Vitamin K Lecture Summary Name: Mohsin Mohammed Taqi Mohsin Al-Saleh ID #: M020944-99 0 Introduction: Vitamin K is a group of structurally
Sultan Qaboos University College of Agricultural & Marine Sciences Vitamin K Lecture Summary Name: Mohsin Mohammed Taqi Mohsin Al-Saleh ID #: M020944-99 0 Introduction: Vitamin K is a group of structurally
How can we tell who will fracture? Beyond bone mineral density to the new world of fracture risk assessment
 Copyright 2008 by How can we tell who will fracture? Beyond bone mineral density to the new world of fracture risk assessment Dr. Bone density testing: falling short of expectations More than 25 years
Copyright 2008 by How can we tell who will fracture? Beyond bone mineral density to the new world of fracture risk assessment Dr. Bone density testing: falling short of expectations More than 25 years
Efficacy of Vitamin K2 for Glucocorticoid-induced Osteoporosis in Patients with Systemic Autoimmune Diseases
 ORIGINAL ARTICLE Efficacy of Vitamin K2 for Glucocorticoid-induced Osteoporosis in Patients with Systemic Autoimmune Diseases Kotaro Shikano 1, Kaichi Kaneko 1, Mai Kawazoe 1, Makoto Kaburaki 1, Tomoko
ORIGINAL ARTICLE Efficacy of Vitamin K2 for Glucocorticoid-induced Osteoporosis in Patients with Systemic Autoimmune Diseases Kotaro Shikano 1, Kaichi Kaneko 1, Mai Kawazoe 1, Makoto Kaburaki 1, Tomoko
Vitamin K is the collective term for compounds with vitamin K activity and having the common 2-methyl-1,4-naphtoquinone ring structure.
 Vitamin K NNR 2012 1 (7) 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 27 28 29 30 31 32 33 34 35 36 37 38 39 40 41 42 43 44 45 46 47 48 49 Vitamin K... 1 Dietary sources and intake...
Vitamin K NNR 2012 1 (7) 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 27 28 29 30 31 32 33 34 35 36 37 38 39 40 41 42 43 44 45 46 47 48 49 Vitamin K... 1 Dietary sources and intake...
K K MK-4 MK-4 1,25-D3
 22 5 13 D 25-Hydroxyvitamin D Gla K K D K D K K K -4MK-4 MK-4 MK-4 steriod and xenobiotic receptorsxr MK-4 SXR MK-4 K MK-4 D 1,25-D3 D 1 CYP27B1CYP27B1-KO D CYP27B1-KO D D CYP27B1-KO Ca 1,25-D3 D Vitamin
22 5 13 D 25-Hydroxyvitamin D Gla K K D K D K K K -4MK-4 MK-4 MK-4 steriod and xenobiotic receptorsxr MK-4 SXR MK-4 K MK-4 D 1,25-D3 D 1 CYP27B1CYP27B1-KO D CYP27B1-KO D D CYP27B1-KO Ca 1,25-D3 D Vitamin
The association of vitamin K status with warfarin sensitivity at the onset of treatment
 British Journal of Haematology, 2001, 112, 572±577 The association of vitamin K status with warfarin sensitivity at the onset of treatment Mary Cushman, 1, 2 Sarah L. Booth, 3 Carl J. Possidente, 4 Kenneth
British Journal of Haematology, 2001, 112, 572±577 The association of vitamin K status with warfarin sensitivity at the onset of treatment Mary Cushman, 1, 2 Sarah L. Booth, 3 Carl J. Possidente, 4 Kenneth
Department of Endocrinology, Tokyo Metropolitan Geriatric Medical Center, 35-2 Sakae-cho, Itabashi-ku, Tokyo , Japan 2
 J Bone Miner Metab (2004) 22:26 240 DOI 0.007/s00774-00-047-z Springer-Verlag 2004 Impaired gamma carboxylation of osteocalcin in elderly women with type II diabetes mellitus: relationship between increase
J Bone Miner Metab (2004) 22:26 240 DOI 0.007/s00774-00-047-z Springer-Verlag 2004 Impaired gamma carboxylation of osteocalcin in elderly women with type II diabetes mellitus: relationship between increase
Elecsys bone marker panel. Optimal patient management starts in the laboratory
 bone marker panel Optimal patient management starts in the laboratory Complete solution for osteoporosis The most complete bone metabolism panel on a single platform bone marker assays are important diagnostic
bone marker panel Optimal patient management starts in the laboratory Complete solution for osteoporosis The most complete bone metabolism panel on a single platform bone marker assays are important diagnostic
Two-Year Randomized Controlled Trial of Vitamin K 1 (Phylloquinone) and Vitamin D 3 Plus Calcium on the Bone Health of Older Women
 JOURNAL OF BONE AND MINERAL RESEARCH Volume 22, Number 4, 2007 Published online on January 22, 2007; doi: 10.1359/JBMR.070116 2007 American Society for Bone and Mineral Research Two-Year Randomized Controlled
JOURNAL OF BONE AND MINERAL RESEARCH Volume 22, Number 4, 2007 Published online on January 22, 2007; doi: 10.1359/JBMR.070116 2007 American Society for Bone and Mineral Research Two-Year Randomized Controlled
OPINION OF THE SCIENTIFIC COMMITTEE ON FOOD ON THE TOLERABLE UPPER INTAKE LEVEL OF VITAMIN K
 FOREWORD OPINION OF THE SCIENTIFIC COMMITTEE ON FOOD ON THE TOLERABLE UPPER INTAKE LEVEL OF VITAMIN K (EXPRESSED ON 4 APRIL 2003) This opinion is one in the series of opinions of the Scientific Committee
FOREWORD OPINION OF THE SCIENTIFIC COMMITTEE ON FOOD ON THE TOLERABLE UPPER INTAKE LEVEL OF VITAMIN K (EXPRESSED ON 4 APRIL 2003) This opinion is one in the series of opinions of the Scientific Committee
Calcium and Vitamin D Supplementation is an Ineffective Strategy for the Prevention of Fractures in Older People
 Calcium and Vitamin D Supplementation is an Ineffective Strategy for the Prevention of Fractures in Older People For the Motion: Professor Roger Francis, Institute for Ageing and Health, Newcastle University,
Calcium and Vitamin D Supplementation is an Ineffective Strategy for the Prevention of Fractures in Older People For the Motion: Professor Roger Francis, Institute for Ageing and Health, Newcastle University,
REVIEW ARTICLE. Vitamin K and the Prevention of Fractures. Systematic Review and Meta-analysis of Randomized Controlled Trials
 REVIEW ARTICLE Vitamin K and the Prevention of Fractures Systematic Review and Meta-analysis of Randomized Controlled Trials Sarah Cockayne, MSc; Joy Adamson, PhD; Susan Lanham-New, PhD; Martin J. Shearer,
REVIEW ARTICLE Vitamin K and the Prevention of Fractures Systematic Review and Meta-analysis of Randomized Controlled Trials Sarah Cockayne, MSc; Joy Adamson, PhD; Susan Lanham-New, PhD; Martin J. Shearer,
. The menaquinones, collectively known as vitamin K 2
 Part Fat Soluble Vitamins Risk Assessment Vitamin K General Information Chemistry Vitamin K is not a single compound but a group of homologous fat-soluble compounds derived from -methyl-1,4-naphthoquinone.
Part Fat Soluble Vitamins Risk Assessment Vitamin K General Information Chemistry Vitamin K is not a single compound but a group of homologous fat-soluble compounds derived from -methyl-1,4-naphthoquinone.
Name of Policy: Boniva (Ibandronate Sodium) Infusion
 Name of Policy: Boniva (Ibandronate Sodium) Infusion Policy #: 266 Latest Review Date: April 2010 Category: Pharmacology Policy Grade: Active Policy but no longer scheduled for regular literature reviews
Name of Policy: Boniva (Ibandronate Sodium) Infusion Policy #: 266 Latest Review Date: April 2010 Category: Pharmacology Policy Grade: Active Policy but no longer scheduled for regular literature reviews
Vitamin K (Phylloquinone)
 Vitamin K (Phylloquinone) What is Vitamin K? Vitamin K is a fat-soluble vitamin which plays a key role in blood coagulation. Its name comes from the German name, Koagulationsvitamin. There are three forms,
Vitamin K (Phylloquinone) What is Vitamin K? Vitamin K is a fat-soluble vitamin which plays a key role in blood coagulation. Its name comes from the German name, Koagulationsvitamin. There are three forms,
Opinion of the Scientific Committee on Food on the Tolerable Upper Intake Level of Vitamin K
 EUROPEAN COMMISSION HEALTH & CONSUMER PROTECTION DIRECTORATE-GENERAL Directorate C - Scientific Opinions C2 - Management of scientific committees; scientific co-operation and networks Scientific Committee
EUROPEAN COMMISSION HEALTH & CONSUMER PROTECTION DIRECTORATE-GENERAL Directorate C - Scientific Opinions C2 - Management of scientific committees; scientific co-operation and networks Scientific Committee
Who should receive calcium and vitamin D supplementation?
 Age and Ageing Advance Access published August 8, 2012 Age and Ageing 2012; 0: 1 5 The Author 2012. Published by Oxford University Press on behalf of the British Geriatrics Society. doi: 10.1093/ageing/afs094
Age and Ageing Advance Access published August 8, 2012 Age and Ageing 2012; 0: 1 5 The Author 2012. Published by Oxford University Press on behalf of the British Geriatrics Society. doi: 10.1093/ageing/afs094
9/26/2016. The Impact of Dietary Protein on the Musculoskeletal System. Research in dietary protein, musculoskeletal health and calcium economy
 The Impact of Dietary Protein on the Musculoskeletal System Outline A. The musculoskeletal system and associated disorders Jessica D Bihuniak, PhD, RD Assistant Professor of Clinical Nutrition Department
The Impact of Dietary Protein on the Musculoskeletal System Outline A. The musculoskeletal system and associated disorders Jessica D Bihuniak, PhD, RD Assistant Professor of Clinical Nutrition Department
Benefits of Calcium and Vitamin D: Building and Maintaining Healthy Bones
 Benefits of Calcium and Vitamin D: Building and Maintaining Healthy Bones Prepared by Annette Dickinson, Ph.D. Council for Responsible Nutrition June 2002 There is no question that a generous intake of
Benefits of Calcium and Vitamin D: Building and Maintaining Healthy Bones Prepared by Annette Dickinson, Ph.D. Council for Responsible Nutrition June 2002 There is no question that a generous intake of
THE NEW ZEALAND MEDICAL JOURNAL
 THE NEW ZEALAND MEDICAL JOURNAL Vol 116 No 1179 ISSN 1175 8716 Efficacy of an oral, 10-day course of high-dose calciferol in correcting vitamin D deficiency Fiona Wu, Toni Staykova, Anne Horne, Judy Clearwater,
THE NEW ZEALAND MEDICAL JOURNAL Vol 116 No 1179 ISSN 1175 8716 Efficacy of an oral, 10-day course of high-dose calciferol in correcting vitamin D deficiency Fiona Wu, Toni Staykova, Anne Horne, Judy Clearwater,
Skeletal Manifestations
 Skeletal Manifestations of Metabolic Bone Disease Mishaela R. Rubin, MD February 21, 2008 The Three Ages of Women Gustav Klimt 1905 1 Lecture Outline Osteoporosis epidemiology diagnosis secondary causes
Skeletal Manifestations of Metabolic Bone Disease Mishaela R. Rubin, MD February 21, 2008 The Three Ages of Women Gustav Klimt 1905 1 Lecture Outline Osteoporosis epidemiology diagnosis secondary causes
INTERNATIONAL JOURNAL OF PHARMACOLOGY AND THERAPEUTICS ISSN Original Article
 Original Article THE STUDY OF RELATIONSHIP BETWEEN UNDERCARBOXILATED OSTEOCALCIN AND ADMINISTRATION OF VITAMIN K 2, VITAMIN D 3 AND CALCIUM IN PREVENTION OF OSTEOPOROSIS IN POSTMENOPAUSAL OSTEOPENIC WOMEN
Original Article THE STUDY OF RELATIONSHIP BETWEEN UNDERCARBOXILATED OSTEOCALCIN AND ADMINISTRATION OF VITAMIN K 2, VITAMIN D 3 AND CALCIUM IN PREVENTION OF OSTEOPOROSIS IN POSTMENOPAUSAL OSTEOPENIC WOMEN
British Journal of Nutrition
 (2010), 104, 858 862 q The Authors 2010 doi:10.1017/s0007114510001182 Vitamin K absorption and kinetics in human subjects after consumption of 13 C-labelled phylloquinone from kale Janet A. Novotny*, Anne
(2010), 104, 858 862 q The Authors 2010 doi:10.1017/s0007114510001182 Vitamin K absorption and kinetics in human subjects after consumption of 13 C-labelled phylloquinone from kale Janet A. Novotny*, Anne
Vitamin K. Emily Rohan Sarah Steinmetz
 Vitamin K Emily Rohan Sarah Steinmetz How much do we need? Adequate Intake (AI) Institute of Medicine. Dietary reference intakes for vitamin A, vitamin K, arsenic, boron, chromium, copper, iodine, iron,
Vitamin K Emily Rohan Sarah Steinmetz How much do we need? Adequate Intake (AI) Institute of Medicine. Dietary reference intakes for vitamin A, vitamin K, arsenic, boron, chromium, copper, iodine, iron,
A high menaquinone intake reduces the incidence of coronary heart disease
 Nutrition, Metabolism & Cardiovascular Diseases (2008) xx, 1e7 available at www.sciencedirect.com journal homepage: www.elsevier.com/locate/nmcd A high menaquinone intake reduces the incidence of coronary
Nutrition, Metabolism & Cardiovascular Diseases (2008) xx, 1e7 available at www.sciencedirect.com journal homepage: www.elsevier.com/locate/nmcd A high menaquinone intake reduces the incidence of coronary
Bone Metabolism in Postmenopausal Women Influenced by the Metabolic Syndrome
 Bone Metabolism in Postmenopausal Women Influenced by the Metabolic Syndrome Thomas et al. Nutrition Journal (2015) 14:99 DOI 10.1186/s12937-015-0092-2 RESEARCH Open Access Acute effect of a supplemented
Bone Metabolism in Postmenopausal Women Influenced by the Metabolic Syndrome Thomas et al. Nutrition Journal (2015) 14:99 DOI 10.1186/s12937-015-0092-2 RESEARCH Open Access Acute effect of a supplemented
Dr Tuan V NGUYEN. Mapping Translational Research into Individualised Prognosis of Fracture Risk
 Dr Tuan V NGUYEN Bone and Mineral Research Program, Garvan Institute of Medical Research, Sydney NSW Mapping Translational Research into Individualised Prognosis of Fracture Risk From the age of 60, one
Dr Tuan V NGUYEN Bone and Mineral Research Program, Garvan Institute of Medical Research, Sydney NSW Mapping Translational Research into Individualised Prognosis of Fracture Risk From the age of 60, one
Zeenat Ali, PGY3 Joseph Grisanti, MD June 7 th, 2012
 A Randomized Open Label Trial to Evaluate the Efficacy of Different Dosage Forms of Vitamin D in Patients with Vitamin D Deficiency, and the Effect of Food on Vitamin D Absorption. Zeenat Ali, PGY3 Joseph
A Randomized Open Label Trial to Evaluate the Efficacy of Different Dosage Forms of Vitamin D in Patients with Vitamin D Deficiency, and the Effect of Food on Vitamin D Absorption. Zeenat Ali, PGY3 Joseph
Associations Between Liver Function, Bone Turnover Biomarkers and Adipokines in Older Patients With Hip Fracture
 Elmer ress Original Article J Endocrinol Metab. 2014;4(5-6):121-135 Associations Between Liver Function, Bone Turnover Biomarkers and Adipokines in Older Patients With Hip Fracture Leon Fisher a, d, Alexander
Elmer ress Original Article J Endocrinol Metab. 2014;4(5-6):121-135 Associations Between Liver Function, Bone Turnover Biomarkers and Adipokines in Older Patients With Hip Fracture Leon Fisher a, d, Alexander
High Dietary Sodium Intake Assessed by 24-hour Urine Specimen Increase Urinary Calcium Excretion and Bone Resorption Marker
 J Bone Metab 214;21:189-194 http://dx.doi.org/1.115/jbm.214.21.3.189 pissn 2287-6375 eissn 2287-729 Original Article High Dietary Sodium Intake Assessed by 24-hour Urine Specimen Increase Urinary Calcium
J Bone Metab 214;21:189-194 http://dx.doi.org/1.115/jbm.214.21.3.189 pissn 2287-6375 eissn 2287-729 Original Article High Dietary Sodium Intake Assessed by 24-hour Urine Specimen Increase Urinary Calcium
V t i amin i n D a nd n d Calc l iu i m u : Rol o e l in i n Pr P eve v nt n io i n and n d Tr T eatment n of o Fr F actur u es and n d Fa F ll l s
 Vitamin D and Calcium: Role in Prevention and Treatment of Fractures and Falls Osteoporosis 21: New Insights In Research, Diagnosis, and Clinical Care Deborah Sellmeyer, MD Director, Johns Hopkins Metabolic
Vitamin D and Calcium: Role in Prevention and Treatment of Fractures and Falls Osteoporosis 21: New Insights In Research, Diagnosis, and Clinical Care Deborah Sellmeyer, MD Director, Johns Hopkins Metabolic
Vitamin K The Forgotten Vitamin
 Vitamin K The Forgotten Vitamin Ask anybody what vitamins they know about, and you re likely to hear Vitamin C, Vitamin D, Vitamin E, and many people will even know that there are multiple vitamins called
Vitamin K The Forgotten Vitamin Ask anybody what vitamins they know about, and you re likely to hear Vitamin C, Vitamin D, Vitamin E, and many people will even know that there are multiple vitamins called
Vitamin K. Emily Rohan Sarah Steinmetz
 Vitamin K Emily Rohan Sarah Steinmetz How much do we need? Adequate Intake (AI) Life Stage Group Adequate Intake Level 0-6 months 2.0 ug/day 7-12 months 2.5 ug/day 1-3 years 30 ug/day 4-8 years 55 ug/day
Vitamin K Emily Rohan Sarah Steinmetz How much do we need? Adequate Intake (AI) Life Stage Group Adequate Intake Level 0-6 months 2.0 ug/day 7-12 months 2.5 ug/day 1-3 years 30 ug/day 4-8 years 55 ug/day
Zinc Intake and Biochemical Markers of Bone Turnover in Type 1 Diabetes
 Diabetes Care Publish Ahead of Print, published online September 22, 2008 Zinc and bone markers Zinc Intake and Biochemical Markers of Bone Turnover in Type 1 Diabetes Raelene E. Maser, PhD 1,2; John N.
Diabetes Care Publish Ahead of Print, published online September 22, 2008 Zinc and bone markers Zinc Intake and Biochemical Markers of Bone Turnover in Type 1 Diabetes Raelene E. Maser, PhD 1,2; John N.
Osteocalcin and DM 정호연 강동경희대병원
 Osteocalcin and DM 정호연 강동경희대병원 leptin osteoporosis fractures Endocrine feedback? Obesity, DM Osteocalcin OC is osteoblast specific protein OC -/- shows abnomal visceral fat Is Osteocalcin a candidate?
Osteocalcin and DM 정호연 강동경희대병원 leptin osteoporosis fractures Endocrine feedback? Obesity, DM Osteocalcin OC is osteoblast specific protein OC -/- shows abnomal visceral fat Is Osteocalcin a candidate?
Collagen Crosslinks, Any Method
 190.19 - Collagen Crosslinks, Any Method Collagen crosslinks, part of the matrix of bone upon which bone mineral is deposited, are biochemical markers the excretion of which provides a quantitative measurement
190.19 - Collagen Crosslinks, Any Method Collagen crosslinks, part of the matrix of bone upon which bone mineral is deposited, are biochemical markers the excretion of which provides a quantitative measurement
Bone Turnover Markers for the Diagnosis and Management of Osteoporosis and Diseases Associated with High Bone Turnover. Original Policy Date
 MP 2.04.10 Bone Turnover Markers for the Diagnosis and Management of Osteoporosis and Diseases Associated with High Bone Turnover Medical Policy Section Medicine Issue 12:2013 Original Policy Date 12:2013
MP 2.04.10 Bone Turnover Markers for the Diagnosis and Management of Osteoporosis and Diseases Associated with High Bone Turnover Medical Policy Section Medicine Issue 12:2013 Original Policy Date 12:2013
Postmenopausal osteoporosis is a systemic
 OSTEOPOROSIS: HARD FACTS ABOUT BONES Steven T. Harris, MD, FACP* ABSTRACT As a consequence of the aging process, osteoporosis affects all men and women. Agerelated loss of bone mass leads to skeletal fragility
OSTEOPOROSIS: HARD FACTS ABOUT BONES Steven T. Harris, MD, FACP* ABSTRACT As a consequence of the aging process, osteoporosis affects all men and women. Agerelated loss of bone mass leads to skeletal fragility
Coordinator of Post Professional Programs Texas Woman's University 1
 OSTEOPOROSIS Update 2007-2008 April 26, 2008 How much of our BMD is under our control (vs. genetics)? 1 2 Genetic effects on bone loss: longitudinal twin study (Makovey, 2007) Peak BMD is under genetic
OSTEOPOROSIS Update 2007-2008 April 26, 2008 How much of our BMD is under our control (vs. genetics)? 1 2 Genetic effects on bone loss: longitudinal twin study (Makovey, 2007) Peak BMD is under genetic
Name of Policy: Zoledronic Acid (Reclast ) Injection
 Name of Policy: Zoledronic Acid (Reclast ) Injection Policy #: 355 Latest Review Date: May 2011 Category: Pharmacy Policy Grade: Active Policy but no longer scheduled for regular literature reviews and
Name of Policy: Zoledronic Acid (Reclast ) Injection Policy #: 355 Latest Review Date: May 2011 Category: Pharmacy Policy Grade: Active Policy but no longer scheduled for regular literature reviews and
Annual Rheumatology & Therapeutics Review for Organizations & Societies
 Annual Rheumatology & Therapeutics Review for Organizations & Societies Biochemical Markers of Bone Turnover: Definitions and Recommendations for Monitoring Therapy Learning Objectives for Biochemical
Annual Rheumatology & Therapeutics Review for Organizations & Societies Biochemical Markers of Bone Turnover: Definitions and Recommendations for Monitoring Therapy Learning Objectives for Biochemical
Vitamin K Deficiency Is Associated with Incident Knee Osteoarthritis
 CLINICAL RESEARCH STUDY Vitamin K Deficiency Is Associated with Incident Knee Osteoarthritis Devyani Misra, MD, MS, a Sarah L. Booth, PhD, b Irina Tolstykh, MS, c David T. Felson, MD, MPH, a Michael C.
CLINICAL RESEARCH STUDY Vitamin K Deficiency Is Associated with Incident Knee Osteoarthritis Devyani Misra, MD, MS, a Sarah L. Booth, PhD, b Irina Tolstykh, MS, c David T. Felson, MD, MPH, a Michael C.
Dietary intake patterns in older adults. Katherine L Tucker Northeastern University
 Dietary intake patterns in older adults Katherine L Tucker Northeastern University Changes in dietary needs with aging Lower energy requirement Less efficient absorption and utilization of many nutrients
Dietary intake patterns in older adults Katherine L Tucker Northeastern University Changes in dietary needs with aging Lower energy requirement Less efficient absorption and utilization of many nutrients
Vitamin K deficiency in CKD patients: a modifiable risk factor for vascular calcification?
 http://www.kidney-international.org & 2009 International Society of Nephrology deficiency in CKD patients: a modifiable risk factor for vascular calcification? Thilo Krueger 1, Ralf Westenfeld 2, Markus
http://www.kidney-international.org & 2009 International Society of Nephrology deficiency in CKD patients: a modifiable risk factor for vascular calcification? Thilo Krueger 1, Ralf Westenfeld 2, Markus
The Relationship between Prevalence of Osteoporosis and Proportion of Daily Protein Intake
 Korean J Fam Med. 2013;34:43-48 http://dx.doi.org/10.4082/kjfm.2013.34.1.43 The Relationship between Prevalence of Osteoporosis and Proportion of Daily Protein Intake Original Article Junga Kim, Byungsung
Korean J Fam Med. 2013;34:43-48 http://dx.doi.org/10.4082/kjfm.2013.34.1.43 The Relationship between Prevalence of Osteoporosis and Proportion of Daily Protein Intake Original Article Junga Kim, Byungsung
DRI Concept. DRI Concept
 Risk of adverse effects 1.5 0 DRI Concept DRI Concept EAR RDA UL 1.5 0 Observed level intake Risk of inadequacy Tolerable Upper Intake Level The highest level of daily nutrient intake that is likely to
Risk of adverse effects 1.5 0 DRI Concept DRI Concept EAR RDA UL 1.5 0 Observed level intake Risk of inadequacy Tolerable Upper Intake Level The highest level of daily nutrient intake that is likely to
CASE 1 WHY IS IT IMPORTANT TO TREAT? FACTS CONCERNS
 4:30-5:15pm Ask the Expert: Osteoporosis SPEAKERS Silvina Levis, MD OSTEOPOROSIS - FACTS 1:3 older women and 1:5 older men will have a fragility fracture after age 50 After 3 years of treatment, depending
4:30-5:15pm Ask the Expert: Osteoporosis SPEAKERS Silvina Levis, MD OSTEOPOROSIS - FACTS 1:3 older women and 1:5 older men will have a fragility fracture after age 50 After 3 years of treatment, depending
Calcium Marie Dunford, PhD, MD, Nutrition Consultant, Kingsburg, CA
 Calcium Marie Dunford, PhD, MD, Nutrition Consultant, Kingsburg, CA Calcium is an essential mineral and a major nutrient needed for proper bone health. Absorption of calcium in adults is low. Vitamin D
Calcium Marie Dunford, PhD, MD, Nutrition Consultant, Kingsburg, CA Calcium is an essential mineral and a major nutrient needed for proper bone health. Absorption of calcium in adults is low. Vitamin D
Dementia. Inhibition of vascular calcification
 FLEMENGHO Dementia Inhibition of vascular calcification Role of matrix Gla protein Yan-Ping Liu, Fang-Fei Wei, Cees Vermeer, and Jan A. Staessen, Research Unit Hypertension and Cardiovascular Epidemiology,
FLEMENGHO Dementia Inhibition of vascular calcification Role of matrix Gla protein Yan-Ping Liu, Fang-Fei Wei, Cees Vermeer, and Jan A. Staessen, Research Unit Hypertension and Cardiovascular Epidemiology,
The effect of supplementation with vitamin D on recurrent ischemic events and sudden cardiac death in patients with acute coronary syndrome
 CRC IRB Proposal Matthew Champion PGY-1 8/29/12 The effect of supplementation with vitamin D on recurrent ischemic events and sudden cardiac death in patients with acute coronary syndrome Study Purpose
CRC IRB Proposal Matthew Champion PGY-1 8/29/12 The effect of supplementation with vitamin D on recurrent ischemic events and sudden cardiac death in patients with acute coronary syndrome Study Purpose
Effect of vitamin K supplementation on insulin resistance in older men and women
 Diabetes Care Publish Ahead of Print, published online August 12, 2008 Phylloquinone intake and insulin resistance Effect of vitamin K supplementation on insulin resistance in older men and women Makiko
Diabetes Care Publish Ahead of Print, published online August 12, 2008 Phylloquinone intake and insulin resistance Effect of vitamin K supplementation on insulin resistance in older men and women Makiko
JOURNAL OF INTERNATIONAL ACADEMIC RESEARCH FOR MULTIDISCIPLINARY Impact Factor 1.393, ISSN: , Volume 2, Issue 7, August 2014
 HYPOVITAMINOSIS D IN INDIAN FEMALES WITH POSTMENOPAUSAL OSTEOPOROSIS DR. SHAH WALIULLAH 1 DR. VINEET SHARMA 2 DR. R N SRIVASTAVA 3 DR. YASHODHARA PRADEEP 4 DR. A A MAHDI 5 DR. SANTOSH KUMAR 6 1 Research
HYPOVITAMINOSIS D IN INDIAN FEMALES WITH POSTMENOPAUSAL OSTEOPOROSIS DR. SHAH WALIULLAH 1 DR. VINEET SHARMA 2 DR. R N SRIVASTAVA 3 DR. YASHODHARA PRADEEP 4 DR. A A MAHDI 5 DR. SANTOSH KUMAR 6 1 Research
A breakthrough in the treatment of OSTEOPOROSIS
 Vit. K2 A breakthrough in the treatment of OSTEOPOROSIS OSTEOPOROSIS is a systemic disease of the skeleton apparatus implying compromised bone mineral density and microarchitecture which increases fracture
Vit. K2 A breakthrough in the treatment of OSTEOPOROSIS OSTEOPOROSIS is a systemic disease of the skeleton apparatus implying compromised bone mineral density and microarchitecture which increases fracture
NIH Public Access Author Manuscript Am J Med. Author manuscript; available in PMC 2013 September 01.
 NIH Public Access Author Manuscript Published in final edited form as: Am J Med. 2013 March ; 126(3): 243 248. doi:10.1016/j.amjmed.2012.10.011. Vitamin K Deficiency Is Associated with Incident Knee Osteoarthritis
NIH Public Access Author Manuscript Published in final edited form as: Am J Med. 2013 March ; 126(3): 243 248. doi:10.1016/j.amjmed.2012.10.011. Vitamin K Deficiency Is Associated with Incident Knee Osteoarthritis
Supplementary appendix
 Supplementary appendix This appendix formed part of the original submission and has been peer reviewed. We post it as supplied by the authors. Supplement to: Bolland MJ, Grey A, Gamble GD, Reid IR. The
Supplementary appendix This appendix formed part of the original submission and has been peer reviewed. We post it as supplied by the authors. Supplement to: Bolland MJ, Grey A, Gamble GD, Reid IR. The
Undercarboxylated osteocalcin and bone mass in 8 12 year old children with cystic fibrosis
 Journal of Cystic Fibrosis 7 (2008) 307 312 www.elsevier.com/locate/jcf Undercarboxylated osteocalcin and bone mass in 8 12 year old children with cystic fibrosis M.S. Fewtrell a,, C. Benden b, J.E. Williams
Journal of Cystic Fibrosis 7 (2008) 307 312 www.elsevier.com/locate/jcf Undercarboxylated osteocalcin and bone mass in 8 12 year old children with cystic fibrosis M.S. Fewtrell a,, C. Benden b, J.E. Williams
NIH Public Access Author Manuscript Osteoporos Int. Author manuscript; available in PMC 2011 January 8.
 NIH Public Access Author Manuscript Published in final edited form as: Osteoporos Int. 2011 January ; 22(1): 345 349. doi:10.1007/s00198-010-1179-4. Does Dietary Protein Reduce Hip Fracture Risk in Elders?
NIH Public Access Author Manuscript Published in final edited form as: Osteoporos Int. 2011 January ; 22(1): 345 349. doi:10.1007/s00198-010-1179-4. Does Dietary Protein Reduce Hip Fracture Risk in Elders?
Objectives. Polypharmacy Seminar. Causes of Polypharmacy. Polypharmacy 8/05/12. National Prescribing Curriculum Module
 Polypharmacy Seminar Objectives On completion of this module you should be able to: state the major guiding principles in rational prescribing recognise, and substantiate, over-prescribing or inappropriate
Polypharmacy Seminar Objectives On completion of this module you should be able to: state the major guiding principles in rational prescribing recognise, and substantiate, over-prescribing or inappropriate
Fat Soluble Vitamins. Vitamin A
 Fat Soluble Vitamins Vitamin A The biochemical basis on which vitamin A requirements were established The EAR was set based on data pertaining to the assurance of adequate stores of vitamin A (Table 19).
Fat Soluble Vitamins Vitamin A The biochemical basis on which vitamin A requirements were established The EAR was set based on data pertaining to the assurance of adequate stores of vitamin A (Table 19).
Measurement of osteocalcin
 Review Article Ann Clin Biochem 2000; 37: 432±446 Measurement of osteocalcin Allison Jane Lee, Stephen Hodges and Richard Eastell From the Bone Metabolism Group, Section of Medicine, Division of Clinical
Review Article Ann Clin Biochem 2000; 37: 432±446 Measurement of osteocalcin Allison Jane Lee, Stephen Hodges and Richard Eastell From the Bone Metabolism Group, Section of Medicine, Division of Clinical
FRUIT AND VEGETABLE INTAKE IN POSTMENOPAUSAL WOMEN WITH OSTEOPENIA
 FRUIT AND VEGETABLE INTAKE IN POSTMENOPAUSAL WOMEN WITH OSTEOPENIA Samira Ebrahimof, Anahita Hoshyarrad, Arash Hossein-Nezhad Nahid Zandi, Bagher Larijani, Masoud Kimiagar Abstract INTRODUCTION: Adequate
FRUIT AND VEGETABLE INTAKE IN POSTMENOPAUSAL WOMEN WITH OSTEOPENIA Samira Ebrahimof, Anahita Hoshyarrad, Arash Hossein-Nezhad Nahid Zandi, Bagher Larijani, Masoud Kimiagar Abstract INTRODUCTION: Adequate
FOR CONSUMERS AND PATIENTS
 AVAILABILITY OF VITAMIN D FOR CONSUMERS AND PATIENTS Prof. Heike A. Bischoff-Ferrari, MD, DrPH Centre on Aging and Mobility, University of Zurich Dept. of Rheumatology, University Hospital Zurich Overview
AVAILABILITY OF VITAMIN D FOR CONSUMERS AND PATIENTS Prof. Heike A. Bischoff-Ferrari, MD, DrPH Centre on Aging and Mobility, University of Zurich Dept. of Rheumatology, University Hospital Zurich Overview
The New England Journal of Medicine EFFECT OF CALCIUM AND VITAMIN D SUPPLEMENTATION ON BONE DENSITY IN MEN AND WOMEN 65 YEARS OF AGE OR OLDER
 EFFECT OF CALCIUM AND VITAMIN D SULEMENTATION ON BONE DENSITY IN MEN AND WOMEN 65 YEARS OF AGE OR OLDER BESS DAWSON-HUGHES, M.D., SUSAN S. HARRIS, D.SC., ELIZABETH A. KRALL, H.D., AND GERARD E. DALLAL,
EFFECT OF CALCIUM AND VITAMIN D SULEMENTATION ON BONE DENSITY IN MEN AND WOMEN 65 YEARS OF AGE OR OLDER BESS DAWSON-HUGHES, M.D., SUSAN S. HARRIS, D.SC., ELIZABETH A. KRALL, H.D., AND GERARD E. DALLAL,
Vitamin D and Calcium
 Vitamin D and Calcium American Association of Clinical Endocrinologists Marina Del Rey, CA September 15, 2018 Albert Shieh, MD MS Assistant Clinical Professor Department of Medicine Division of Endocrinology
Vitamin D and Calcium American Association of Clinical Endocrinologists Marina Del Rey, CA September 15, 2018 Albert Shieh, MD MS Assistant Clinical Professor Department of Medicine Division of Endocrinology
This clinical study synopsis is provided in line with Boehringer Ingelheim s Policy on Transparency and Publication of Clinical Study Data.
 abcd Clinical Study for Public Disclosure This clinical study synopsis is provided in line with s Policy on Transparency and Publication of Clinical Study Data. The synopsis which is part of the clinical
abcd Clinical Study for Public Disclosure This clinical study synopsis is provided in line with s Policy on Transparency and Publication of Clinical Study Data. The synopsis which is part of the clinical
Vitamin K in Cystic Fibrosis
 Vitamin K in Cystic Fibrosis Martin J. Shearer PhD, MRCPath Consultant Clinical Scientist Honorary Senior Lecturer Centre for Haemostasis and Thrombosis, Guy s and St. Thomas Foundation Trust London, UK
Vitamin K in Cystic Fibrosis Martin J. Shearer PhD, MRCPath Consultant Clinical Scientist Honorary Senior Lecturer Centre for Haemostasis and Thrombosis, Guy s and St. Thomas Foundation Trust London, UK
MILK. Nutritious by nature. The science behind the health and nutritional impact of milk and dairy foods
 MILK Nutritious by nature The science behind the health and nutritional impact of milk and dairy foods Muscle mass maintenance in older people There is evidence to suggest a potential role for milk and
MILK Nutritious by nature The science behind the health and nutritional impact of milk and dairy foods Muscle mass maintenance in older people There is evidence to suggest a potential role for milk and
Review Article Vitamins K1 and K2: The Emerging Group of Vitamins Required for Human Health
 Hindawi Journal of Nutrition and Metabolism Volume 2017, Article ID 6254836, 6 pages https://doi.org/10.1155/2017/6254836 Review Article Vitamins K1 and K2: The Emerging Group of Vitamins Required for
Hindawi Journal of Nutrition and Metabolism Volume 2017, Article ID 6254836, 6 pages https://doi.org/10.1155/2017/6254836 Review Article Vitamins K1 and K2: The Emerging Group of Vitamins Required for
The Role of the Laboratory in Metabolic Bone Disease
 The Role of the Laboratory in Metabolic Bone Disease Howard Morris PhD, FAACB, FFSc(RCPA) President, IFCC Professor of Medical Sciences, University of South Australia, Clinical Scientist, SA Pathology
The Role of the Laboratory in Metabolic Bone Disease Howard Morris PhD, FAACB, FFSc(RCPA) President, IFCC Professor of Medical Sciences, University of South Australia, Clinical Scientist, SA Pathology
How to Design, Conduct, and Analyze Vitamin D Clinical Trials
 How to Design, Conduct, and Analyze Vitamin D Clinical Trials William B. Grant, PhD Sunlight, Nutrition and Health Research Center wbgrant@infionline.net Disclosure I receive funding from Bio-Tech Pharmacal,
How to Design, Conduct, and Analyze Vitamin D Clinical Trials William B. Grant, PhD Sunlight, Nutrition and Health Research Center wbgrant@infionline.net Disclosure I receive funding from Bio-Tech Pharmacal,
Collagen Crosslinks, Any Method
 190.19 - Collagen Crosslinks, Any Method Collagen crosslinks, part of the matrix of bone upon which bone mineral is deposited, are biochemical markers the excretion of which provides a quantitative measurement
190.19 - Collagen Crosslinks, Any Method Collagen crosslinks, part of the matrix of bone upon which bone mineral is deposited, are biochemical markers the excretion of which provides a quantitative measurement
Vitamin K Supplementation in Postmenopausal Women with Osteopenia (ECKO Trial): A Randomized Controlled Trial
 PLoS MEDICINE Vitamin K Supplementation in Postmenopausal Women with Osteopenia (ECKO Trial): A Randomized Controlled Trial Angela M. Cheung 1,2,3,4*, Lianne Tile 1,4, Yuna Lee 1, George Tomlinson 1,3,
PLoS MEDICINE Vitamin K Supplementation in Postmenopausal Women with Osteopenia (ECKO Trial): A Randomized Controlled Trial Angela M. Cheung 1,2,3,4*, Lianne Tile 1,4, Yuna Lee 1, George Tomlinson 1,3,
Sang Ling Wu, MD, Wei Li, MD, PhD, Alice Wells, MT(ASCP), and Amitava Dasgupta, PhD
 Clinical Chemistry / DIGOXIN-LIKE AND DIGITOXIN-LIKE IMMUNOREACTIVE SUBSTANCES IN ELDERLY PEOPLE Digoxin-Like and Digitoxin-Like Immunoreactive Substances in Elderly People Impact on Therapeutic Drug Monitoring
Clinical Chemistry / DIGOXIN-LIKE AND DIGITOXIN-LIKE IMMUNOREACTIVE SUBSTANCES IN ELDERLY PEOPLE Digoxin-Like and Digitoxin-Like Immunoreactive Substances in Elderly People Impact on Therapeutic Drug Monitoring
Amani Alghamdi. Slide 1
 Minerals in the body Amani Alghamdi Slide 1 The Minerals Small, naturally occurring, inorganic, chemical elements Serve as structural components Minerals classification The minerals present in the body
Minerals in the body Amani Alghamdi Slide 1 The Minerals Small, naturally occurring, inorganic, chemical elements Serve as structural components Minerals classification The minerals present in the body
BMD: A Continuum of Risk WHO Bone Density Criteria
 Pathogenesis of Osteoporosis Osteoporosis Diagnosis: BMD, FRAX and Assessment of Secondary Osteoporosis AGING MENOPAUSE OTHER RISK FACTORS RESORPTION > FORMATION Bone Loss LOW PEAK BONE MASS Steven T Harris
Pathogenesis of Osteoporosis Osteoporosis Diagnosis: BMD, FRAX and Assessment of Secondary Osteoporosis AGING MENOPAUSE OTHER RISK FACTORS RESORPTION > FORMATION Bone Loss LOW PEAK BONE MASS Steven T Harris
Robin M Daly, Nicole Petrass, Shona Bass, and Caryl A Nowson
 The skeletal benefits of calcium- and vitamin D 3 fortified milk are sustained in older men after withdrawal of supplementation: an 18-mo follow-up study 1 3 Robin M Daly, Nicole Petrass, Shona Bass, and
The skeletal benefits of calcium- and vitamin D 3 fortified milk are sustained in older men after withdrawal of supplementation: an 18-mo follow-up study 1 3 Robin M Daly, Nicole Petrass, Shona Bass, and
Diabetes Care 31: , 2008
 Clinical Care/Education/Nutrition/Psychosocial Research O R I G I N A L A R T I C L E Effect of Vitamin K Supplementation on Insulin Resistance in Older Men and Women MAKIKO YOSHIDA, PHD 1 PAUL F. JACQUES,
Clinical Care/Education/Nutrition/Psychosocial Research O R I G I N A L A R T I C L E Effect of Vitamin K Supplementation on Insulin Resistance in Older Men and Women MAKIKO YOSHIDA, PHD 1 PAUL F. JACQUES,
Assessment and Treatment of Osteoporosis Professor T.Masud
 Assessment and Treatment of Osteoporosis Professor T.Masud Nottingham University Hospitals NHS Trust University of Nottingham University of Derby University of Southern Denmark What is Osteoporosis? Osteoporosis
Assessment and Treatment of Osteoporosis Professor T.Masud Nottingham University Hospitals NHS Trust University of Nottingham University of Derby University of Southern Denmark What is Osteoporosis? Osteoporosis
Compounds with vitamin K activity all have a common. Vitamin K in Parenteral Nutrition. Metabolic Function
 GASTROENTEROLOGY 2009;137:S105 S118 Vitamin K in Parenteral Nutrition MARTIN J. SHEARER Centre for Haemostasis and Thrombosis, St Thomas Hospital, London, United Kingdom Vitamin K (as phylloquinone and
GASTROENTEROLOGY 2009;137:S105 S118 Vitamin K in Parenteral Nutrition MARTIN J. SHEARER Centre for Haemostasis and Thrombosis, St Thomas Hospital, London, United Kingdom Vitamin K (as phylloquinone and
Chapter 1 Introduction
 Chapter 1 Introduction There are several disorders which carry an increased risk of thrombosis, clots that interfere with normal circulation, including: venous thromboembolism (VTE), comprising both deep
Chapter 1 Introduction There are several disorders which carry an increased risk of thrombosis, clots that interfere with normal circulation, including: venous thromboembolism (VTE), comprising both deep
LABORATORY REPORT SAMPLE. Summary of Deficient Test Results. Vitamin B12 Pantothenate Biotin Spectrox Immunidex
 LABORATORY REPORT Account Number: Name: Jon Doe Gender: Male DOB: 12/28/1984 Requisition Number: Summary of Deficient Test Results Testing determined the following functional deficiencies: Oleic Acid Borderline
LABORATORY REPORT Account Number: Name: Jon Doe Gender: Male DOB: 12/28/1984 Requisition Number: Summary of Deficient Test Results Testing determined the following functional deficiencies: Oleic Acid Borderline
Page: 1 of 12. Bone Turnover Markers for Diagnosis and Management of Osteoporosis and Diseases Associated With High Bone Turnover
 Last Review Status/Date: December 2014 Page: 1 of 12 Management of Osteoporosis and Diseases Description Bone turnover markers are biochemical markers of either bone formation or bone resorption. Commercially
Last Review Status/Date: December 2014 Page: 1 of 12 Management of Osteoporosis and Diseases Description Bone turnover markers are biochemical markers of either bone formation or bone resorption. Commercially
Dietary protein intake affects albumin fractional synthesis rate in younger and older adults equally
 Emerging Science Dietary protein intake affects albumin fractional synthesis rate in younger and older adults equally Anna E Thalacker-Mercer and Wayne W Campbell Inclusion of dietary protein in meals
Emerging Science Dietary protein intake affects albumin fractional synthesis rate in younger and older adults equally Anna E Thalacker-Mercer and Wayne W Campbell Inclusion of dietary protein in meals
