Biochimica et Biophysica Acta xxx (2011) xxx xxx. Contents lists available at ScienceDirect. Biochimica et Biophysica Acta
|
|
|
- Melissa Roberts
- 5 years ago
- Views:
Transcription
1 BBAPAP-38660; No. of pages: 10; 4C: 2, 3 Biochimica et Biophysica Acta xxx (2011) xxx xxx Contents lists available at ScienceDirect Biochimica et Biophysica Acta journal homepage: 1 Review 2 Proteases as regulators of pathogenesis: Examples from the apicomplexa 3 Hao Li, Matthew A. Child, Matthew Bogyo Q1 4 Departments of Pathology and Microbiology and Immunology and Graduate program in Chemical and Systems Biology, Stanford University, 300 Pasteur Dr. Stanford, CA 94305, USA 5 article info 6 7 Article history: 8 Received 27 April Received in revised form 1 June Accepted 2 June Available online xxxx Q3 15 Keywords: 16 Apicomplexans 17 Proteases 18 Pathogenesis 19 Plasmodium 20 Toxoplasma 21 Malaria 22 Invasion 23 Egress Contents abstract The diverse functional roles that proteases play in basic biological processes make them essential for virtually 24 all organisms. Not surprisingly, proteolysis is also a critical process required for many aspects of pathogenesis. 25 In particular, obligate intracellular parasites must precisely coordinate proteolytic events during their highly 26 regulated life cycle inside multiple host cell environments. Advances in chemical, proteomic and genetic tools 27 that can be applied to parasite biology have led to an increased understanding of the complex events centrally 28 regulated by proteases. In this review, we outline recent advances in our knowledge of specific proteolytic 29 enzymes in two medically relevant apicomplexan parasites: Plasmodium falciparum and Toxoplasma gondii. 30 Efforts over the last decade have begun to provide a map of key proteotolyic events that are essential for both 31 parasite survival and propagation inside host cells. These advances in our molecular understanding of 32 proteolytic events involved in parasite pathogenesis provide a foundation for the validation of new networks 33 and enzyme targets that could be exploited for therapeutic purposes. This article is part of a Special Issue 34 entitled: Proteolysis 50 years after the discovery of lysosome Published by Elsevier B.V Introduction The apicomplexan parasites: Plasmodium and Toxoplasma Role of proteases in host-cell invasion Subtilases Rhomboid proteases The cysteine proteases The metalloprotease toxolysin Proteases in general catabolism Hemoglobin degradation in Plasmodium Proteases involved in general peptide processing Proteases have central roles in parasite egress Serine-repeat antigen (SERA) family PfSUB DPAP Host cell calpain Proteases as targets for anti-malarials Tools to facilitate dissection of apicomplexan parasite proteases Conclusion Acknowledgements References This article is part of a Special Issue entitled: Proteolysis 50 years after the discovery of lysosome. Corresponding author. Tel.: ; fax: address: mbogyo@stanford.edu (M. Bogyo) /$ see front matter 2011 Published by Elsevier B.V. doi: /j.bbapap Introduction The primary function of all proteases is to catalyze the simple process of peptide bond hydrolysis. While this process is the same for all proteases, each family utilizes a different general mechanism to accomplish this goal
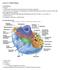
2 2 H. Li et al. / Biochimica et Biophysica Acta xxx (2011) xxx xxx 68 Although proteases were originally thought to act as downstream 69 mediators of protein turn-over, we now know that distinct proteolytic 70 processing events can also regulate or initiate key biological processes 71 such as cell death [1], cell cycle progression [2] and cell migration [3]. 72 Given that proteases regulate such basic biological processes, it is not 73 surprising that apicomplexan parasites, with their complex life cycles, also 74 depend on proteolytically regulated processes. Decades of research in 75 parasite biology have begun to define the roles of proteases in all stages of 76 the parasite life cycle. In this review, we will provide a general overview of 77 the current knowledge of proteases found in the apicomplexan parasites 78 Plasmodium spp. and Toxoplasma spp, with special focus on three 79 important cellular events: host-cell invasion, general catabolism and 80 host cell rupture (also known as egress). While the roles of proteases in 81 various aspects of parasite biology have been reviewed in the past [4 7] 82 we will focus on recent discoveries that have contributed to our 83 understanding of critical proteolytic events in these two important 84 human pathogens The apicomplexan parasites: Plasmodium and Toxoplasma 86 The apicomplexa comprise a phylum of highly diverse eukaryotic 87 protozoa that exists as parasites in animals. Apicomplexans are 88 morphologically characterized by the presence of an eponymous apical 89 complex of organelles consisting of micronemes, rhoptries and dense 90 granules, as well as other less-well characterized secretory organelles such 91 as exonemes. These organelles contain proteins that are secreted around 92 the time of egress and invasion to facilitate parasite motility, host cell 93 adhesion and subsequent remodeling of host cells (see [8] for detailed 94 review). These parasites also have common, yet parasite-specific features 95 such as a chloroplast-like organelle termed the apicoplast that has a role in 96 fatty acid and isoprenoid synthesis [9]. Plasmodium spp., the causative 97 agent of malaria, affects 500 million worldwide and therefore attracts a 98 considerable amount of medical and research attention [10]. Classically at 99 least five species of Plasmodium infect humans: Plasmodium vivax, 100 Plasmodium ovale, Plasmodium malariae, Plasmodium knowlesi and 101 Plasmodium falciparum. Each species has a highly regulated life cycle 102 inside the intermediate human host as well as inside the definitive insect 103 vector. In addition all Plasmodium spp. within the intermediate host 104 primarily infect red blood cells. Toxoplasma gondii is related to Plasmodium 105 spp. however a notable difference is that T. gondii infects virtually any 106 nucleated host cell. Up to one third of the entire population of the world is 107 infected by T. gondii[11] and although primary infection by T. gondii is 108 typically subclinical, it can cause serious illness in immuno-compromised 109 patients and leads to fetal death if acquired during pregnancy [12]. 110 Both Plasmodium and Toxoplasma progress through a complicated 111 series of sexual and asexual life cycles where non-replicative extracel- lular forms of the parasites known as zoites first invade host cells, grow and replicate within these cells, and finally rupture to establish further infections (Fig. 1)[13,14]. This variety of molecular events requires the expression of many specialized proteins to coordinate these diverse biochemical activities. Recent efforts have revealed proteases as crucial players in all stages of the parasite life cycle. The roles of various proteases in Plasmodium and Toxoplasma will be reviewed here, but the importance of proteases is not limited to the apicomplexans, and their roles in the progression of other parasitic diseases have been previously reviewed [15]. 2. Role of proteases in host-cell invasion As obligate intracellular parasites, it is vital for the extra-cellular forms of Plasmodium and Toxoplasma to rapidly invade host cells. This process of invasion occurs within a very short time frame because the extracellular forms cannot survive for long periods of time outside host cells. Invasion proceeds following an initial contact by the polar zoite, which reorients such that its apical end contacts the host cell membrane [16,17]. Atight junction that results from the interaction of multiple receptor proteins forms between the apposed parasite and host membranes, and this junction moves from the apical to the posterior end of the zoite. The active trafficking of the receptor proteins on the parasite surface drags the host cell membrane over the parasite, creating an invagination within the host cell [5,6]. As invasion proceeds, the zoite must shed the surface proteins initially required for attachment and penetration. Invasion is complete when the parasite is fully engulfed within the parasitophorous vacuole, which seals to create a protective environment where it can subsequently grow and replicate (see Fig. 2,Step1). Many apical surface proteins have been confirmed to be required for successful attachment [7,18,19]. In Plasmodium, proteins such as merozoite surface proteins (MSPs), apical membrane antigen 1 (AMA1), rhoptry neck proteins (RONs) and serine repeat antigen (SERA) proteins are thought to play important roles in parasite invasion. In addition, many of these proteins are proteolytically processed either during trafficking to, or arrival at, the parasite surface. However, until recently the identities of the majority of proteases that regulate the maturation and subsequent shedding of the surface proteins during invasion were largely unknown Subtilases The subtilisin-like serine proteases are a class of enzymes present in both P. falciparum and T. gondii. The P. falciparum genome project identified three subtilases but only two, PfSUB1 and PfSUB2, are known to have functional roles in the blood-stages of the parasite life cycle [20]. PfSUB1 has roles in both egress (see Section 4.2) and invasion. This Fig. 1. Asexual life cycle of Plasmodium (A) and Toxoplasma(B). During invasion, polar zoite cells randomly establish contact to host cells, followed by reorientation of apical end to host cell membrane. A tight junction forms between the parasite and host membrane, and this junction moves from the apical to the posterior end of the parasite. This results in formation of an invagination which eventually forms the parasitophorous vacuole. This is accompanied by shedding of surface proteins. Once invasion is complete, parasites rapidly grow in size in the intracellular environment, and replicate their genome to prepare for cellular division. When cell division is complete, parasites exit host cells in a process known as egress to invade other cells. In Toxoplasma, chronic infection can occur via transformation of the active tachyzoites to the inactive form of parasites known as bradyzoites which form tissue cysts in hosts.
3 H. Li et al. / Biochimica et Biophysica Acta xxx (2011) xxx xxx 3 Fig. 2. Schematic depicting the role of proteases in different stages of the parasite lifecycle in A) Plasmodium falciparum and B) Toxoplasma gondii. Different classes of proteases arerepresented in the corresponding color. Proteases are involved in proteolytic maturation of apical organelle proteins prior to invasion (not shown in figure). During invasion (1), the proteins from the apical organelles can facilitate adhesion to the host cell, and these proteins are subsequently shed in a protease-dependent manner. Proteases are also involved in general metabolism (2) to allow growth and replication. After several rounds of cell division, host cells rupture to release parasite zoite cells (3) which then re-invade other host cellsandrepeatthelifecycle. 154 protease has a high degree of substrate specificity [21] and its expression 155 peaks in schizont stage [22]. It was initially postulated that PfSUB1 might 156 be involved in the shedding of micronemal proteins from the surface of 157 the parasite during invasion. However selective inhibitors of PfSUB1 do 158 not interfere with shedding of surface proteins MSP-1 and AMA-1 [6],but 159 can efficiently block invasion [23]. The effects of PfSUB1 inhibitors were 160 suggested to be the result of a block in processing of surface proteins 161 required for productive invasion. PfSUB1 was subsequently shown to 162 effectively process MSP1/6/7 and that the maturation of these surface 163 proteins was required for successful invasion [24]. A detailed bioinfor- 164 matic and proteomic analysis has identified additional proteins processed 165 by PfSUB1 that are putatively involved in egress and invasion [25]. 166 While PfSUB1 is involved in maturation of MSP1 before merozoite 167 rupture, PfSUB2 functions as a sheddase during merozoite invasion. 168 PfSUB2 localizes to the micronemes, and is secreted and translocated to 169 the posterior end of parasite surface during invasion [26]. Duringthis 170 translocation, PfSUB2 proteolytically cleaves the ectodomain of MSP1 and 171 AMA1. This processing may disengage adhesion complexes to release the 172 parasite into the parasitophorous vacuole. Evidence also suggests that 173 PfSUB2 is responsible for the proteolytic processing of Plasmodium 174 thrombospondin related apical merozoite protein (PTRAMP), another 175 adhesive protein on the merozoite surface [27]. Similar to MSP1 and 176 AMA1, PTRAMP is shed from the merozoite during erythrocyte invasion. 177 Given the relatedness of T. gondii and P. falciparum, it is not surprising 178 that subtilisin-like proteases also have important functions in host cell 179 invasion by Toxoplasma[28,29]. Using a gene disruption strategy to 180 knock out the sub1 gene in tachyzoites, Lagal and co-workers [30] 181 showed that loss of GPI-anchored protease TgSUB1 results in defective 182 surface processing of micronemal proteins MIC2, MIC4 and M2AP. Since 183 secretion of mature forms of these proteins is important for attachment 184 to the host cell, tachyzoites lacking TgSUB1 have a reduced ability to 185 invade. Furthermore, deletion of the sub1 gene results in parasites with a 186 defect in gliding motility, a key process required for invasion of the host. 187 The highly related T. gondii subtilase, TgSUB2, also has been linked to 188 invasion. TgSUB2 is a transmembrane protein that localizes to the 189 rhoptry organelle in tachyzoites [31]. Attempts to disrupt the Tgsub2 190 gene suggest that it is essential in the T. gondii life cycle. Interestingly, 191 the peptide sequence where autocleavage occurs within the TgSUB2 192 protein matches that of cleavage sites of other rhoptry protein (e.g. 193 ROP1). This, combined with its known rhoptry localization, is suggestive 194 of a role for TgSUB2 in proteolytic maturation of rhoptry proteins Rhomboid proteases The Rhomboid family of serine proteases was first discovered in 196 Drosophila[32], and it soon became apparent that these intra-membrane 197 proteases are conserved in both prokaryotic and eukaryotic organisms 198 [33,34]. The rhomboid proteases have a mechanism of proteolysis that is 199 unique from the soluble serine proteases. Specifically, the protease 200 active site is buried in the plane of the membrane, and proteolysis occurs 201 in, or adjacent to, the transmembrane domain of the substrate [35]. 202 In Toxoplasma, proteomic analysis of invasion-specific cleavage 203 events first hinted at a role for rhomboid proteases in surface protein 204 shedding [36]. Six rhomboid-like genes were identified and five 205 rhomboid proteases, TgROM1-5, were subsequently cloned and 206 characterized [37,38]. These rhomboid proteases have non-overlapping 207 substrate specificities and localize to different regions of the cell. Of 208 these, TgROM2, TgROM4 and TgROM5 function as sheddases of 209 micronemal adhesin proteins during the process of invasion. Recent 210 work has uncovered a crucial role for TgROM4 in invasion as a result of 211 processing of surface adhesins MIC2, MIC3 and AMA1 [39]. This cleavage 212 of AMA1 from the T. gondii surface during invasion, in turn, triggers a 213 switch of the parasite from an invasive to replicative state [40]. TgROM5, 214 which like TgROM4, is localized to the cell surface of the tachyzoite, 215 cleaves the adhesion protein MIC2 upon its trafficking to the posterior 216 end of the parasite membrane during invasion. The rhomboid proteases 217 in T. gondii thus collectively serve as sheddases that remove adhesion 218 proteins to allow successful host cell invasion. However, it is perhaps 219 worthwhile to note that rhomboid proteases may have functions 220 beyond those required for invasion. For example, TgROM1 has been 221 shown to contribute to intracellular growth of the parasite [41]. 222 Not long after the characterization of TgROMs, rhomboid proteases 223 were identified in P. falciparum and shown to also function in shedding 224 of adhesion proteins during invasion. PfROM1 and PfROM4 are able to 225 cleave a variety of substrates that possess a transmembrane domain, 226 and they are likely to be involved in all invasive stages of P. falciparum 227 [42 44]. PfROM1 has similar substrate preference as the Drosophila 228 Spitz-cleaving rhomboid proteases, while PfROM4 is distinct in that it 229 does not cleave Spitz-type substrate motifs and instead cleaves 230 erythrocyte binding-like (EBL) adhesins [42]. Collectively, these results 231 establish a role for rhomboid proteases in apicomplexan invasion. 232 Further studies will be required to characterize the specific targets of 233 each rhomboid protease in the life cycle of the parasites
4 4 H. Li et al. / Biochimica et Biophysica Acta xxx (2011) xxx xxx The cysteine proteases 236 Although cysteine protease inhibitors have convincingly been 237 shown to block parasite invasion, only a few candidate proteases have 238 been identified. In P. falciparum, the identification of selective inhibitors 239 of falcipain-1, through screening of epoxide inhibitor libraries, provided 240 a tool that implicated this protease in erythrocyte invasion [45]. Unlike 241 the other falcipains that are involved in hemoglobin degradation, 242 falcipain-1 localizes to distinct, granule like structures in newly formed 243 merozoites. However, subsequent gene disruption of falcipain suggests that its role in invasion may be somewhat redundant [46,47]. 245 The fact that broad-spectrum cysteine protease inhibitors block invasion 246 suggests that yet undiscovered cysteine proteases may function 247 together to ensure successful invasion. 248 In T. gondii, a cathepsin B homologue TgCPB (also known as toxopain or TgCP1), may play a role in tachyzoite invasion by processing rhoptry 250 proteins [48]. Immunoelectron microscopy using cathepsin B antibodies 251 provides some evidence that TgCPB might be localized to the rhoptries. 252 The use of small molecule inhibitors of cathepsin B has also been shown 253 to disrupt rhoptry protein maturation resulting in abnormal rhoptry 254 development and defects in tachyzoite invasion [49]. However, the 255 specificity of the compounds used in these studies has not been 256 confirmed suggesting that effects could be mediated by other targets. In 257 particular, T. gondii expresses a highly homologous cathepsin L, TgCPL, 258 which has recently been shown to localize to a specialized secretory 259 organelle termed the vacuolar compartment (VAC) where it is believed 260 to play a role in maturation of secretory proteins targeted to the 261 micronemes [50]. Inparticular,T. gondii deficient for TgCPL showed 262 slower maturation of TgM2AP and TgMIC3 proproteins. As TgM2AP and 263 TgMIC3 are required for efficient host cell invasion, it is not surprising 264 that the TgCPL-deficient strain has reduced ability to invade host cells. 265 Although only a limited number of cysteine proteases have been 266 confirmed to have a role in invasion, recent tools such as the complete 267 genome sequence of P. falciparum and T. gondii, in combination with the 268 use of chemical inhibitors, will hopefully aid in the discovery and 269 characterization of additional important cysteine proteases The metalloprotease toxolysin The metalloprotease toxolysin 4 contains a signature motif present in 272 insulin-degrading enzymes [51]. This protease localizes to the micro- 273 nemes, and its secretion coincides with discharge of micronemal contents. 274 Thus, it is possible that toxolysin 4 can function in invasion. However, 275 further studies will be required to assign specific roles to this protease Proteases in general catabolism 277 Once invasion has occurred, parasites actively metabolize host cell 278 proteins to generate products required for growth and replication. In 279 Plasmodium, this process has been extensively studied due to its potential 280 as a target pathway for malaria drug-therapy (See Fig. 2,Step2)[52] Hemoglobin degradation in Plasmodium 282 During the intraerythrocytic stage of the Plasmodium life cycle, the 283 parasite imports host cell hemoglobin and then progressively degrades it 284 to generate key nutrients as the parasite grows. Degradation of 285 hemoglobin provides free amino acids, frees up space for further growth, 286 and also has a possible role in regulating the osmotic status of the cell [53]. 287 This process occurs mainly in the trophozoite stage, during which time 288 hemoglobin is endocytosed into a parasite organelle known as the food 289 vacuole. Once inside this acidic compartment, a cascade of aspartyl and 290 cysteine proteases degrades the hemoglobin to the final amino acid 291 products [54]. 292 The extended family of plasmepsin aspartyl proteases in P. falciparum 293 has been the focus of intensive research mainly due to the parasite- specific nature of these enzymes [55]. Therefore, plasmepsins were initially thought to be ideal targets for development of anti-parasitic drugs. The plasmepsins are a family of aspartyl proteases made up of 4 main members: plasmepsins I, II, IV and histo-aspartic protease [56,57]. Plasmepsin I and II are thought to initiate hemoglobin degradation, while histo-aspartic protease and plasmepsin IV function in the degradation of peptides generated by the action of upstream members of the family [58 61]. However, the fact that genetic disruption of individual plasmepsins did not result in any difference in P. falciparum morphology suggests that functional redundancy between the family members may make these proteases non-optimal as therapeutic targets [62]. Nevertheless, plasmepsins are important proteases in hemoglobin degradation, especially since a triple or quadruple plasmepsins gene knock-out confers a growth defect in P. falciparum[63]. Other proteases such as the metalloprotease falcilysin [64 66], and the cysteine proteases falcipains 2, 2 and 3 [67 70] and dipeptidyl aminopeptidase 1 (DPAP1), are also involved in degradation of hemoglobin after it has been cleaved into peptide fragments [71]. Falcipains are thought to function downstream of plasmepsin I and II, and evidence suggests that these proteases are involved in activation of the food vacuole plasmepsins [72]. Gene disruptions as well as studies using small molecule inhibitors of falcipain-2, have confirmed a role for this protease in hemoglobin degradation [73 75]. Furthermore, falcipains may be able to replace the functions of plasmepsins [62] making them promising targets for antimalarials [76]. The cysteine dipeptidyl aminopeptidase 1 (DPAP1) is an exopeptidase in the food vacuole [71]. DPAP1 is a homologue of mammalian cathepsin C that also removes dipeptides from protein substrates. DPAP1 functions in an acidic environment (the food vacuole) and has similar substrate specificity as cathepsin C [77]. Studies using specific inhibitors of DPAP1 have shown that inhibition of this protease leads to an accumulation of parasites at the trophozoite stage and a subsequent decrease in parasitemia [78]. These inhibitors were also tested in a mouse model of malaria and found to reduce parasitemia. Furthermore, by linking DPAP1 inhibitors to a synthetic analog of another effective antimalarial drug, it was possible to enhance killing of parasites in culture [79]. These observations, combined with the essential nature of DPAP1 expression in blood stage parasites [71], suggest that it plays a critical role in parasite survival and is therefore a potentially valuable drug target. The final stages of hemoglobin degradation are carried out by aminopeptidases that generate single amino acids from cleaved peptides [80]. An additional family of neutral amino-peptidases PfA- M1 and PfM17LAP [81 83] has recently been suggested to degrade peptide substrates that are exported to the cytosol from the food vacuole to generate free amino acids [84 86]. These proteases may also represent promising new targets for the development of antimalarial drugs, which may be facilitated by the recent structural characterization of members of this family [87] Proteases involved in general peptide processing Proteases that perform essential roles such as protein homeostasis, protein transport and host cell remodeling are also potentially valuable as targets for therapeutic intervention. As these processes are required throughout the parasite life cycle, drugs that block the action of these proteases have the potential to be effective at multiple points during an infection. The proteasome is a large multi-subunit complex that catalyzes the proteolysis of proteins that have been targeted for degradation by ubiquitination. This protease complex was conserved across the eukaryota and is also found in some prokaryotes [88]. Theproteasome is involved in regulatory functions such as protein turnover and degradation of misfolded proteins. In mammalian cells, it also regulates cell cycle progression and inflammation pathways [89].Furthermore,a small molecule proteasome inhibitor has been approved by the FDA for
5 H. Li et al. / Biochimica et Biophysica Acta xxx (2011) xxx xxx the treatment of multiple myeloma, suggesting that targeting the 358 proteasome could be an effective way to treat other diseases [90]. 359 Initial studies using the natural product lactacystin, which inhibits P. 360 falciparum proteasome at nanomolar concentrations, confirmed that a 361 block in proteasome activity leads to parasite death [91].However,these 362 initial studies also pointed out the overall toxicity that resulted from 363 inhibition of the host proteasome. More recent studies with several 364 different classes of proteasome inhibitors suggest that the proteasome 365 may be a viable target in Plasmodium spp. [92 94].Furthermore, atleast 366 two proteasome inhibitors epoxomicin and thiostrepton also reduce 367 growth of the sexually differentiated Plasmodium gametocyte, suggest- 368 ing that inhibition of the proteasome could be an effective way to block 369 parasite transmission [95,96]. Unfortunately, relatively little is known 370 about the biochemical or structural properties of the proteasome from 371 Toxoplasma or Plasmodium and this has hindered the development of 372 parasite-specific inhibitors. Selective inhibitors will almost certainly be 373 required to effectively validate the proteasome as a drug target for 374 malaria and other parasitic diseases. 375 An additional protease that has been suggested to function in T. 376 gondii in general catabolism is cathepsin C (TgCPC). Three isoforms of 377 this protease have been identified, and TgCP1 is implicated in growth 378 and replication of the parasite [97]. Cathepsin C is an exopeptidase that 379 cleaves dipeptides from the N-terminus of proteins. Treatment with a 380 cathepsin C inhibitor led a reduction in parasitemia and reduced protein 381 degradation in the parasitophorous vacuole (PV). However, additional 382 studies to identify the substrates of cathepsin C will be required to define 383 its role in general peptide degradation in T. gondii. 384 The recent placement of plasmepsin V in the Plasmodium protein 385 export pathway also opens up new strategies for antimalarial 386 chemotherapy. Unlike plasmepsin I IV, plasmepsin V is not localized 387 to the food vacuole, but is instead restricted to the endoplasmic 388 reticulum (ER) membrane [98]. Recent work demonstrated the role of 389 this aspartyl protease in cleaving the Plasmodium Export Element 390 (PEXEL) sequence motif, required for the export of PEXEL containing 391 parasite proteins into the erythrocyte cytosol [99,100]. This export of 392 proteins allows the parasite to remodel the host cell for functions such 393 as adhesion to endothelial cells. Such remodeling could be a 394 mechanism to evade host defenses and therefore inhibition of 395 plasmepsin V could be a strategy for clearing parasites [101,102] Proteases have central roles in parasite egress 397 As obligate intracellular parasites, Plasmodium and Toxoplasma 398 spend the majority of their lives within the confines of their host-cell 399 environment. Once replication is complete and several mitotic cycles 400 have taken place, specialized zoite cells are released from the host cell 401 through a lytic process known as egress (See Fig. 2, Step 3). Our current 402 understanding of egress is that it is a rapid and tightly regulated process. 403 However, the exact mechanisms that regulate egress remain unclear. In 404 Plasmodium, there are two popular models; the first model proposes 405 that egress occurs by initial degradation of the parasitophorous vacuole 406 membrane (PVM), and then the erythrocyte membrane. The second 407 model proposes the reverse, where the erythrocyte membrane is 408 degraded prior to the release of the merozoites from the PVM [103]. 409 Both models have experimental support, and thus it is difficult to 410 provide a definite mechanism. However, recent advances with video 411 microscopy support a model whereby the PVM ruptures, followed by 412 the rapid and explosive rupturing of the erythrocyte membrane that is 413 thought to facilitate the dissemination of the daughter zoites from the 414 parent host-cell [104,105]. 415 Despite our lack of understanding of the precise mechanistic 416 processes underlying egress, proteases appear to play pivotal roles. 417 For example, treatment of P. falciparum infected cells with serine and 418 cysteine protease inhibitors produce a block in egress leading to 419 accumulation of late stage schizonts [106]. However, it is important to 420 note that these experiments used broad-spectrum inhibitors that block proteases implicated in both egress and invasion. Subsequent experiments have identified inhibitors that can specifically block egress, and these have served as useful chemical tools in further exploration of the proteases involved in egress [23,107]. The following section reviews the role of several proteases that function in Plasmodium egress. Although studies on host-cell rupture in Toxoplasma have uncovered roles for potassium and calcium signaling [108] and the pore-forming perforin proteins [109], virtually nothing is known about proteases that may regulate egress in this organism Serine-repeat antigen (SERA) family The serine-repeat antigen (SERA) family is a group of highly expressed Plasmodium proteins that are characterized by a central papain-like protease domain. Within the family, only SERA 6 8havethe canonical active site cysteine residue found in classical papain family proteases, while SERA 1 5 and 9 have a serine in place of this residue [6]. Intriguingly, this family of proteins is not found in related apicomplexan parasites such as Toxoplasma. SERA5 is the most abundant of the SERA family [110] and is found in the parasitophorous vacuole where it is processed around the time of egress. Antibodies against the truncated form of SERA5 and a cyclic peptide directed against the predicted enzyme domain of the protein interferes with its function and cause a reduction in parasite growth, suggesting that SERA5 is vital for Plasmodium egress and/or invasion [ ]. SERA5 is one of the two SERA genes that cannot be disrupted, further highlighting its importance in the parasite life cycle [113]. As SERA5 possesses a papain-like fold with serine as the active site residue [114], it could potentially function in egress and invasion by proteolytically processing substrates. The recombinant catalytic domain of SERA5 indeed possesses some weak chymotrypsin-like activity and this activity is sensitive to protease inhibitors [115]. However, it is still presently unclear if endogenous SERA5 has proteolytic activity. Crystallographic evidence suggests that the active site in SERA5 is not conducive to substrate binding [114]. As such, although SERA5 may be an important player in egress, any catalytic function remains a mystery. The only SERA protein with a direct link to parasite egress is SERA8. In Plasmodium berghei, disruption of the SERA8 orthologue, ECP1, resulted in a complete block of sporozoite egress from oocytes [116]. Therefore, the SERA proteins may mediate egress in both the sexual and asexual stages of Plasmodium PfSUB1 During maturation, SERA5 is proteolytically cleaved from the fulllength form of approximately 126 kda, through two intermediates before accumulating as a 50 kda form which is released into culture supernantant during rupture [117]. Investigation of the proteases responsible for the maturation of SERA5 linked the serine protease PfSUB1 and the cysteine protease DPAP3 to this maturation process [23,107]. In addition to the role of PfSUB1 in priming merozoites during invasion, it has a role in parasite egress. PfSUB1 localizes to a merozoite organelle called the exoneme, and is released into the parasitophorous vacuolar space before egress. Further studies demonstrated that PfSUB1 is responsible for cleaving SERA 4, 5 and 6 during egress. Inhibition of PfSUB1 using a specific inhibitor (MRT 12113) led to significant reduction in egress and invasion. The role of PfSUB1 in processing SERA5 was also confirmed by a chemical screen that identified a chloroisocoumarin (JCP104) that serves as both an inhibitor and probe of PfSUB1 activity [107]. JCP104 specifically blocks schizont rupture further demonstrating that PfSUB1 is a regulator of P. falciparum egress DPAP3 In addition to identifying a small molecule inhibitor of PfSUB1, the small molecule screen by Arastu-Kapur and co-workers identified a
6 6 H. Li et al. / Biochimica et Biophysica Acta xxx (2011) xxx xxx 481 cysteine protease inhibitor that specifically blocked host cell rupture 482 [107]. The identified dipeptide vinyl sulfone hit was converted to an 483 active site probe and used to identify the dipeptidyl aminopeptidases 484 (DPAPs) as targets of this compound. Further development efforts 485 identified inhibitors that could specifically target the food vacuole 486 specific protease DPAP1 as well as the related family member DPAP3, a 487 protease with unknown functions. These compounds showed that 488 inhibition of DPAP1 resulted in toxicity to the parasite without affecting 489 host cell rupture, while inhibition of DPAP3 specifically blocked parasite 490 release from the host cell. Inhibition of DPAP3 also led to reduction in 491 levels of the mature form of PfSUB1 without causing accumulation of the 492 precursor protein, suggesting that DPAP3 may be required for maturation 493 of PfSUB1. DPAP3 may therefore regulate host cell egress by controlling 494 maturation of secretory proteins required for this process [107] Host cell calpain 496 In addition to parasite derived proteases, a host protease has also 497 recently been implicated in the regulation of parasite egress in both 498 P. falciparum and T. gondii. Specifically, depletion of host cell calpain leads 499 to a complete block in parasite egress [118]. In this study, human calpain activity in erythrocytes was reduced by both chemical inhibition and 501 immunodepletion. The resulting calpain-1 deficient erythrocytes were 502 infected with P. falciparum and a defect in schizont rupture was observed. 503 To confirm the role of calpain in this process, purified calpain was added 504 back to the calpain-depleted host cells and egress was restored. The role of 505 calpaininreleaseoft. gondii tachyzoites was also investigated using sirna knockdown in U2OS human host cells. Knockdown of both calpain 1 and calpain 2 was achieved by targeting of the regulatory subunit of both calpains. Similar to Plasmodium, calpain knockdown in host-cells blocked T. gondii egress. Therefore, it is likely that host-cell calpain facilitates parasite egress through some type of host cell remodeling, the details of which are yet to be determined. 5. Proteases as targets for anti-malarials Proteases are attractive targets for drug development because the presence of a well-defined active site facilitates interactions with small molecules, and thus many classes of small molecule proteases inhibitors have been developed [119]. Our understanding of the biology of specific proteases in the intra-erythrocytic stages during malaria infection has advanced greatly in the past decade and this information will likely facilitate the design of specific protease inhibitors as anti-parasitic agents (Table 1). Furthermore, the rapid emergence of drug resistance in Plasmodium spp. suggests an urgent need for new anti-malarial compounds. Current approved drugs focus mainly on targeting processes in the apicoplast or food vacuole, which are responsible for parasite growth and metabolism [52]. Proteases involved in hemoglobin metabolism are the most commonly targeted proteases in anti-malarial drugs currently under development, yet it may not be the most optimal pathway due to highly redundant mechanisms and the ability of the parasite to actively export compounds out of the food vacuole. While much effort has been directed towards developing lead compounds to target falcipains and plasmepsins, it is important to note that t1:1 Table 1 List of partially characterized proteases organized by mechanism of proteolysis in Toxoplasma gondii and Plasmodium falciparum. Cited references are representative papers from studies of each protease listed. t1:2 t1:3 Protease Class Protease Apicomplexan species Implicated function in asexual stage Protease function and localization Literature t1:4 Aspartyl Plasmepsins I IV Plasmodium falciparum Growth and Replication Hemoglobin degradation in food vacuole [55,60] t1:5 Plasmepsin V Plasmodium falciparum Growth and Replication PEXEL processing for export into the erythrocytic [99,100] cytosol; localized to the ER t1:6 Cysteine TgCPB/Toxopain-1 Toxoplasma gondii Invasion Rhoptry protein processing; localized to the rhoptries [48] t1:7 TgCPCs Toxoplasma gondii Growth and Replication Cleavage of dipeptides from N-terminal in cytosol, [97] exact functions unknown t1:8 TgCPL Toxoplasma gondii Invasion Maturation of micronemal proteins; localized in the [50] vacuolar compartment t1:9 PfDPAP1 Plasmodium falciparum Growth and Replication Involved in later stages of hemoglobin degradation [77,78] in food vacuole t1:10 PfDPAP3 Plasmodium falciparum Egress Facilitates PfSUB1 maturation in apical end of [107] merozoites (unpublished)l Falcipain-1 Plasmodium falciparum Invasion Exact functions unknown; localized to apical [45,46] end of merozoites Falcipain-2,2,3 Plasmodium falciparum Growth and Replication Hemoglobin degradation in food vacuole [69,70,74] Metalloprotease Falcilysin Plasmodium falciparum Growth and Replication Involved in later stages of hemoglobin degradation [64,66] in food vacuole PfM1AAP and PfM17LAP Plasmodium falciparum Growth and Replication Involved in terminal stages of hemoglobin degradation [80,82] in food vacuole Toxolysin 4 Toxoplasma gondii Invasion/Egress Exact function unknown; localized to micronemes [51] Serine PfSUB1 Plasmodium falciparum Invasion and egress Processing of merozoite surface and parasitophorous [23,24,107] vacuole (PV) constituents prior to egress; localized to the exonemes PfSUB2 Plasmodium falciparum Invasion Surface protein sheddase; localized to the micronemes [26] TgSUB1 Toxoplasma gondii Invasion Processes micronemal proteins involved in host cell [30] attachment; localized to the micronemes TgSUB2 Toxoplasma gondii Invasion Maturation of rhoptry proteins; localized to the rhoptries [31] PfSERA5 Plasmodium falciparum Egress Potential role in egress; localized to the PV [110,114] TgROM2,4,5 Toxoplasma gondii Invasion Surface protein sheddase; localized to tachyzoite [38] plasma membrane PfROM1 and 4 Plasmodium falciparum Invasion Surface protein sheddase; localized to merozoite [42] plasma membrane Threonine PfHslV Plasmodium falciparum Growth and Replication Possible function as peptidase, but targets unknown; [141,142] localized to mitochondria Proteasome Plasmodium and Toxoplasma Growth and Replication General protein degradation; Regulates protein turnover; localization unknown [94,140]
7 H. Li et al. / Biochimica et Biophysica Acta xxx (2011) xxx xxx redundancies exist between these two families of proteases [62,63] and 532 hence effective therapy would most likely require targeting of both. 533 Recent work has highlighted the potential of DPAP1 as a target for drug 534 development efforts [78]. The inability to disrupt the DPAP1 gene 535 highlights its vital role in parasite catabolism. Furthermore a hybrid of a 536 DPAP1 inhibitor and an artemisinin analogwere able to effectively reduce 537 parasite growth at low nano-molar concentrations with low cell toxicity 538 [79]. Such a hybrid drug is less likely to induce resistance since each 539 fragment of the compound uses distinct mechanisms for parasite killing. 540 Other proteases involved in Plasmodium egress and invasion are 541 gaining attention as potential targets for therapy [103]. Arguably, egress 542 and invasion are attractive processes for inhibitor development because 543 Plasmodium spp. are obligate intracellular parasites that are particularly 544 vulnerable outside their host cells [7]. Our lack of understanding of 545 specific players in these processes is probably the reason for slow 546 development of compounds that target egress and invasion. While there 547 have been a recent series of published inhibitors that target DPAP3 and 548 PfSUB1 [23,107], these compounds are generally not optimal drugs and 549 significant additional efforts will be required to convert these lead 550 compounds into more suitable therapeutic agents Tools to facilitate dissection of apicomplexan parasite proteases 552 The post-genomic era offers exciting prospects to begin to further 553 define apicomplexan biology. Genome sequences [ ] and tran- 554 scriptome profiling [123,124] of P. falciparum and T. gondii enable genetic 555 validation of protease activities and increase the rate of discovery of novel 556 proteases with important functions. While T. gondii is a relatively 557 genetically tractable organism [125], P. falciparum remains challenging 558 to study using classical genetic methods. Low transfection efficiency 559 [126,127], coupled with the lack of RNAi machinery [128,129] makes 560 standard genetic tools untenable. Fortunately, recent advances in 561 chemical genetics [107,130] and systems biology are helping to advance 562 the study of basic parasite biology. 563 One particularly exciting area of development is the use of small 564 molecules to conditionally regulate protein stability. This technique 565 makes use of ligands that can stabilize a protein domain known as the 566 destabilizing domain (DD), which is otherwise degraded in the absence 567 of a stabilizing small molecule [131]. Recent success in P. falciparum has 568 allowed the conditional knockdown of falcipain-2 [132],parasite calpain 569 [133], calcium dependent-protein kinase 5 [134] and a proteasome cap 570 regulatory subunit Rpn6 [135]. InP. falciparum, the DD system has 571 therefore proven to be more generally applicable than the previously 572 reported tetracycline-inducible transactivator system [136]. Moreover, 573 the DD system allows the rapid temporal control of protein levels and 574 thus enables the study of protein function at precise stages of the parasite 575 life cycle. 576 The use of activity-based probes (ABPs) as a forward chemical genetic 577 tool [137] also represents an exciting method to study parasite protease 578 biology [78,107,130]. Using inhibitor scaffolds tagged with a reporter, the 579 activity of proteases can be monitored in intact parasites. This technique 580 can also be applied effectively to identify selective substrates that can then 581 be used for high throughput screens of protease targets [138]. In addition, 582 various groups have harnessed mass spectrometry (MS) as a tool to assess 583 global proteomic changes in P. falciparum[25,139]. These approaches 584 allow for a more global mapping of proteolytic events, and could lead to 585 the identification of essential pathways that are controlled by proteases. 586 The regulators of these pathways can then be prioritized as targets for 587 small molecule therapies Conclusion 589 In this review, we have described the roles of various proteases 590 throughout the life cycles of Plasmodium and Toxoplasma. Although we 591 have highlighted many proteases that have been the focus of significant 592 study over the past decade, we also chose to draw attention to proteases where further investigation is required to better understand their function in the parasite. With the help of novel genetic and chemical tools, coupled with systems-based analysis, the functions of poorly understood and novel proteases can hopefully be defined in the near future. As proteases represent very attractive targets in drug development, further studies will facilitate discovery of novel drugs against malaria and toxoplasmosis that will hopefully overcome emerging resistance to current drug therapies. Acknowledgements We thank Dr. J. Boothroyd for helpful discussions. This work was funded by a Burroughs Wellcome New Investigator in Pathogenesis Grant (to MB) and NIH grants R01 AI and R21 AI (to MB). H.L. is supported by the NSS-Ph.D. Scholarship from Agency for Science, Technology and Research Singapore. References [1] G.M. Cohen, Caspases: the executioners of apoptosis, Biochem. J. 326 (1997) [2] S.J. Li, M. Hochstrasser, A new protease required for cell-cycle progression in yeast, Nature 398 (1999) [3] A. Huttenlocher, S.P. Palecek, Q. Lu, W.L. Zhang, R.L. Mellgren, D.A. Lauffenburger, M.H. Ginsberg, A.F. Horwitz, Regulation of cell migration by the calciumdependent protease calpain, J. Biol. Chem. 272 (1997) [4] M. Klemba, D.E. Goldberg, Biological roles of proteases in parasitic protozoa, Annu. Rev. Biochem. 71 (2002) [5] K. Kim, Role of proteases in host cell invasion by Toxoplasma gondii and other apicomplexa, Acta Trop. 91 (2004) [6] M.J. Blackman, Proteases in host cell invasion by the malaria parasite, Cell. Microbiol. 6 (2004) [7] A.F. Cowman, B.S. Crabb, Invasion of red blood cells by malaria parasites, Cell 124 (2006) [8] M.J. Blackman, L.H. Bannister, Apical organelles of apicomplexa: biology and isolation by subcellular fractionation, Mol. Biochem. Parasitol. 117 (2001) [9] R.F. Walter, G.I. McFadden, The apicoplast: a review of the derived plastid of apicomplexan parasites, Curr. Issues Mol. Biol. 7 (2005) [10] R.W. Snow, C.A. Guerra, A.M. Noor, H.Y. Myint, S.I. Hay, The global distribution of clinical episodes of Plasmodium falciparum malaria, Nature 434 (2005) [11] A.M. Tenter, A.R. Heckeroth, L.M. Weiss, Toxoplasma gondii: from animals to humans, Int. J. Parasitol. 30 (2000) [12] J.G. Montoya, O. Liesenfeld, Toxoplasmosis, Lancet 363 (2004) [13] M.W. Black, J.C. Boothroyd, Lytic cycle of Toxoplasma gondii, Microbiol. Mol. Biol. Rev. 64 (2000) [14] B. Striepen, C.N. Jordan, S. Reiff, G.G. van Dooren, Building the perfect parasite: cell division in apicomplexa, PLoS Pathog. 3 (2007) [15] J.H. McKerrow, C. Caffrey, B. Kelly, P. Loke, M. Sajid, Proteases in parasitic diseases, Ann. Rev. Pathol. -Mech. Disease 1 (2006) [16] J.E. Smith, A ubiquitous intracellular parasite the cellular biology of toxoplasma-gondii, Int. J. Parasitol. 25 (1995) [17] D. Soldati, B.J. Foth, A.F. Cowman, Molecular and functional aspects of parasite invasion, Trends Parasitol. 20 (2004) [18] D. Soldati, J.F. Dubremetz, M. Lebrun, Microneme proteins: structural and functional requirements to promote adhesion and invasion by the apicomplexan parasite Toxoplasma gondii, Int. J. Parasitol. 31 (2001) [19] M.H. Huynh, J.M. Harper, V.B. Carruthers, Preparing for an invasion: charting the pathway of adhesion proteins to Toxoplasma micronemes, Parasitol. Res. 98 (2006) [20] C. Withers-Martinez, L. Jean, M.J. Blackman, Subtilisin-like proteases of the malaria parasite, Mol. Microbiol. 53 (2004) [21] M. Sajid, C. Withers-Martinez, M.J. Blackman, Maturation and specificity of Plasmodium falciparum subtilisin-like protease-1, a malaria merozoite subtilisinlike serine protease, J. Biol. Chem. 275 (2000) [22] M.J. Blackman, H. Fujioka, W.H.L. Stafford, M. Sajid, B. Clough, S.L. Fleck, M. Aikawa, M. Grainger, F. Hackett, A subtilisin-like protein in secretory organelles of Plasmodium falciparum merozoites, J. Biol. Chem. 273 (1998) [23] S. Yeoh, R.A. O'Donnell, K. Koussis, A.R. Dluzewski, K.H. Ansell, S.A. Osborne, F. Hackett, C. Withers-Martinez, G.H. Mitchell, L.H. Bannister, J.S. Bryans, C.A. Kettleborough, M.J. Blackman, Subcellular discharge of a serine protease mediates release of invasive malaria parasites from host erythrocytes, Cell 131 (2007) [24] K. Koussis, C. Withers-Martinez, S. Yeoh, M. Child, F. Hackett, E. Knuepfer, L. Juliano, U. Woehlbier, H. Bujard, M.J. Blackman, A multifunctional serine protease primes the malaria parasite for red blood cell invasion, EMBO J. 28 (2009) [25] N.C. Silmon de Monerri, H.R. Flynn, M.G. Campos, F. Hackett, K. Koussis, C. Withers-Martinez, J.M. Skehel, M.J. Blackman, Global identification of multiple substrates for Plasmodium falciparum SUB1, an essential malarial processing protease, Infect. Immun. 79 (2011) [26] P.K. Harris, S. Yeoh, A.R. Dluzewski, R.A. O'Donnell, C. Withers-Martinez, F. Hackett, L.H. Bannister, G.H. Mitchell, M.J. Blackman, Molecular identification of a malaria merozoite surface sheddase, PLoS Pathog. 1 (2005)
MeBOP Module 2 Introduction
 MeBOP Module 2 Introduction Karine Frenal: The expert of Toxo glideosome and IMC Damien Jacot: Expert in about everything Sunil Kumar Dogga: TgAsp3 Budhaditya Mukherjee: Biochemistry Apicomplexans are
MeBOP Module 2 Introduction Karine Frenal: The expert of Toxo glideosome and IMC Damien Jacot: Expert in about everything Sunil Kumar Dogga: TgAsp3 Budhaditya Mukherjee: Biochemistry Apicomplexans are
REGULATION OF ENZYME ACTIVITY. Medical Biochemistry, Lecture 25
 REGULATION OF ENZYME ACTIVITY Medical Biochemistry, Lecture 25 Lecture 25, Outline General properties of enzyme regulation Regulation of enzyme concentrations Allosteric enzymes and feedback inhibition
REGULATION OF ENZYME ACTIVITY Medical Biochemistry, Lecture 25 Lecture 25, Outline General properties of enzyme regulation Regulation of enzyme concentrations Allosteric enzymes and feedback inhibition
Homework Hanson section MCB Course, Fall 2014
 Homework Hanson section MCB Course, Fall 2014 (1) Antitrypsin, which inhibits certain proteases, is normally secreted into the bloodstream by liver cells. Antitrypsin is absent from the bloodstream of
Homework Hanson section MCB Course, Fall 2014 (1) Antitrypsin, which inhibits certain proteases, is normally secreted into the bloodstream by liver cells. Antitrypsin is absent from the bloodstream of
Subcompartmentalisation of Proteins in the Rhoptries Correlates with Ordered Events of Erythrocyte Invasion by the Blood Stage Malaria Parasite
 Subcompartmentalisation of Proteins in the Rhoptries Correlates with Ordered Events of Erythrocyte Invasion by the Blood Stage Malaria Parasite Elizabeth S. Zuccala 1,4, Alexander M. Gout 2,4, Chaitali
Subcompartmentalisation of Proteins in the Rhoptries Correlates with Ordered Events of Erythrocyte Invasion by the Blood Stage Malaria Parasite Elizabeth S. Zuccala 1,4, Alexander M. Gout 2,4, Chaitali
Practice Exam 2 MCBII
 1. Which feature is true for signal sequences and for stop transfer transmembrane domains (4 pts)? A. They are both 20 hydrophobic amino acids long. B. They are both found at the N-terminus of the protein.
1. Which feature is true for signal sequences and for stop transfer transmembrane domains (4 pts)? A. They are both 20 hydrophobic amino acids long. B. They are both found at the N-terminus of the protein.
Glycolysis - Plasmodium
 Apicomplexan Biochemistry Basics Toxoplasma Good cell biology model Genome sequencing not completed Virtual pathways Cryptosporidium The strange one Genome sequence completed Virtual pathways Plasmodium
Apicomplexan Biochemistry Basics Toxoplasma Good cell biology model Genome sequencing not completed Virtual pathways Cryptosporidium The strange one Genome sequence completed Virtual pathways Plasmodium
Chapter 6. Antigen Presentation to T lymphocytes
 Chapter 6 Antigen Presentation to T lymphocytes Generation of T-cell Receptor Ligands T cells only recognize Ags displayed on cell surfaces These Ags may be derived from pathogens that replicate within
Chapter 6 Antigen Presentation to T lymphocytes Generation of T-cell Receptor Ligands T cells only recognize Ags displayed on cell surfaces These Ags may be derived from pathogens that replicate within
Protein Trafficking in the Secretory and Endocytic Pathways
 Protein Trafficking in the Secretory and Endocytic Pathways The compartmentalization of eukaryotic cells has considerable functional advantages for the cell, but requires elaborate mechanisms to ensure
Protein Trafficking in the Secretory and Endocytic Pathways The compartmentalization of eukaryotic cells has considerable functional advantages for the cell, but requires elaborate mechanisms to ensure
1. to understand how proteins find their destination in prokaryotic and eukaryotic cells 2. to know how proteins are bio-recycled
 Protein Targeting Objectives 1. to understand how proteins find their destination in prokaryotic and eukaryotic cells 2. to know how proteins are bio-recycled As a protein is being synthesized, decisions
Protein Targeting Objectives 1. to understand how proteins find their destination in prokaryotic and eukaryotic cells 2. to know how proteins are bio-recycled As a protein is being synthesized, decisions
2013 John Wiley & Sons, Inc. All rights reserved. PROTEIN SORTING. Lecture 10 BIOL 266/ Biology Department Concordia University. Dr. S.
 PROTEIN SORTING Lecture 10 BIOL 266/4 2014-15 Dr. S. Azam Biology Department Concordia University Introduction Membranes divide the cytoplasm of eukaryotic cells into distinct compartments. The endomembrane
PROTEIN SORTING Lecture 10 BIOL 266/4 2014-15 Dr. S. Azam Biology Department Concordia University Introduction Membranes divide the cytoplasm of eukaryotic cells into distinct compartments. The endomembrane
Complexity DNA. Genome RNA. Transcriptome. Protein. Proteome. Metabolites. Metabolome
 DNA Genome Complexity RNA Transcriptome Systems Biology Linking all the components of a cell in a quantitative and temporal manner Protein Proteome Metabolites Metabolome Where are the functional elements?
DNA Genome Complexity RNA Transcriptome Systems Biology Linking all the components of a cell in a quantitative and temporal manner Protein Proteome Metabolites Metabolome Where are the functional elements?
Intracellular Compartments and Protein Sorting
 Intracellular Compartments and Protein Sorting Intracellular Compartments A eukaryotic cell is elaborately subdivided into functionally distinct, membrane-enclosed compartments. Each compartment, or organelle,
Intracellular Compartments and Protein Sorting Intracellular Compartments A eukaryotic cell is elaborately subdivided into functionally distinct, membrane-enclosed compartments. Each compartment, or organelle,
COMICR 378. Addresses 1 The Walter and Eliza Hall Institute of Medical Research, Parkville,
 Protein targeting to destinations of the secretory pathway in the malaria parasite Plasmodium falciparum Christopher J Tonkin 1, J Andrew Pearce 1, Geoffrey I McFadden 2 and Alan F Cowman 1 The secretory
Protein targeting to destinations of the secretory pathway in the malaria parasite Plasmodium falciparum Christopher J Tonkin 1, J Andrew Pearce 1, Geoffrey I McFadden 2 and Alan F Cowman 1 The secretory
Proteasomes. When Death Comes a Knock n. Warren Gallagher Chem412, Spring 2001
 Proteasomes When Death Comes a Knock n Warren Gallagher Chem412, Spring 2001 I. Introduction Introduction The central dogma Genetic information is used to make proteins. DNA RNA Proteins Proteins are the
Proteasomes When Death Comes a Knock n Warren Gallagher Chem412, Spring 2001 I. Introduction Introduction The central dogma Genetic information is used to make proteins. DNA RNA Proteins Proteins are the
CELL BIOLOGY - CLUTCH CH THE IMMUNE SYSTEM.
 !! www.clutchprep.com CONCEPT: OVERVIEW OF HOST DEFENSES The human body contains three lines of against infectious agents (pathogens) 1. Mechanical and chemical boundaries (part of the innate immune system)
!! www.clutchprep.com CONCEPT: OVERVIEW OF HOST DEFENSES The human body contains three lines of against infectious agents (pathogens) 1. Mechanical and chemical boundaries (part of the innate immune system)
Super-Resolution Dissection of Coordinated Events during Malaria Parasite Invasion of the Human Erythrocyte
 Article Super-Resolution Dissection of Coordinated Events during Malaria Parasite Invasion of the Human Erythrocyte David T. Riglar, 1,2,5 Dave Richard, 1,5 Danny W. Wilson, 1,6 Michelle J. Boyle, 1,2,6
Article Super-Resolution Dissection of Coordinated Events during Malaria Parasite Invasion of the Human Erythrocyte David T. Riglar, 1,2,5 Dave Richard, 1,5 Danny W. Wilson, 1,6 Michelle J. Boyle, 1,2,6
Cell Quality Control. Peter Takizawa Department of Cell Biology
 Cell Quality Control Peter Takizawa Department of Cell Biology Cellular quality control reduces production of defective proteins. Cells have many quality control systems to ensure that cell does not build
Cell Quality Control Peter Takizawa Department of Cell Biology Cellular quality control reduces production of defective proteins. Cells have many quality control systems to ensure that cell does not build
Cross-regulation of host STATs for the survival of Toxoplasma gondii
 JITMM2010&IMC2010, 1-3 December 2010 Cross-regulation of host STATs for the survival of Toxoplasma gondii Ho-Woo Nam 1, Dong-Soo Kim 2, Sung-Jong Hong 3 Department of arasitology, College of Medicine,
JITMM2010&IMC2010, 1-3 December 2010 Cross-regulation of host STATs for the survival of Toxoplasma gondii Ho-Woo Nam 1, Dong-Soo Kim 2, Sung-Jong Hong 3 Department of arasitology, College of Medicine,
Broad Spectrum Protease Inhibitor Cocktail
 Broad Spectrum Protease Inhibitor Cocktail Catalog number: AR1182 Boster s Broad Spectrum Protease Inhibitor Cocktail is a complex of various protease inhibitors, which has been tested for inhibiting proteases
Broad Spectrum Protease Inhibitor Cocktail Catalog number: AR1182 Boster s Broad Spectrum Protease Inhibitor Cocktail is a complex of various protease inhibitors, which has been tested for inhibiting proteases
THE ROLE OF ALTERED CALCIUM AND mtor SIGNALING IN THE PATHOGENESIS OF CYSTINOSIS
 Research Foundation, 18 month progress report THE ROLE OF ALTERED CALCIUM AND mtor SIGNALING IN THE PATHOGENESIS OF CYSTINOSIS Ekaterina Ivanova, doctoral student Elena Levtchenko, MD, PhD, PI Antonella
Research Foundation, 18 month progress report THE ROLE OF ALTERED CALCIUM AND mtor SIGNALING IN THE PATHOGENESIS OF CYSTINOSIS Ekaterina Ivanova, doctoral student Elena Levtchenko, MD, PhD, PI Antonella
Ultrastructural Studies on Plasmodium vivax
 Characterization of Human Malaria Parasites Ultrastructural Studies on Plasmodium vivax For the first time a detailed ultrastructural study was carried out on P. vivax. Fine structural analysis of growth
Characterization of Human Malaria Parasites Ultrastructural Studies on Plasmodium vivax For the first time a detailed ultrastructural study was carried out on P. vivax. Fine structural analysis of growth
Why chaperone vectors?
 Why chaperone vectors? A protein folding initiative An open discussion with structural biologists Protein Structure Initiative: Pilot Phase Whether the pilot phase achieved its goal depends on how we measure
Why chaperone vectors? A protein folding initiative An open discussion with structural biologists Protein Structure Initiative: Pilot Phase Whether the pilot phase achieved its goal depends on how we measure
Peptide hydrolysis uncatalyzed half-life = ~450 years HIV protease-catalyzed half-life = ~3 seconds
 Uncatalyzed half-life Peptide hydrolysis uncatalyzed half-life = ~450 years IV protease-catalyzed half-life = ~3 seconds Life Sciences 1a Lecture Slides Set 9 Fall 2006-2007 Prof. David R. Liu In the absence
Uncatalyzed half-life Peptide hydrolysis uncatalyzed half-life = ~450 years IV protease-catalyzed half-life = ~3 seconds Life Sciences 1a Lecture Slides Set 9 Fall 2006-2007 Prof. David R. Liu In the absence
Malaria Life Cycle Life Cycle
 Malaria Malaria Life Cycle Life Cycle Oocyst Zygote Sporogony Sporozoites Mosquito Salivary Gland Gametocytes Exoerythrocytic (hepatic) cycle Hypnozoites (for P. vivax and P. ovale) Erythrocytic Cycle
Malaria Malaria Life Cycle Life Cycle Oocyst Zygote Sporogony Sporozoites Mosquito Salivary Gland Gametocytes Exoerythrocytic (hepatic) cycle Hypnozoites (for P. vivax and P. ovale) Erythrocytic Cycle
General information. Cell mediated immunity. 455 LSA, Tuesday 11 to noon. Anytime after class.
 General information Cell mediated immunity 455 LSA, Tuesday 11 to noon Anytime after class T-cell precursors Thymus Naive T-cells (CD8 or CD4) email: lcoscoy@berkeley.edu edu Use MCB150 as subject line
General information Cell mediated immunity 455 LSA, Tuesday 11 to noon Anytime after class T-cell precursors Thymus Naive T-cells (CD8 or CD4) email: lcoscoy@berkeley.edu edu Use MCB150 as subject line
LECTURE-15. itraq Clinical Applications HANDOUT. Isobaric Tagging for Relative and Absolute quantitation (itraq) is a quantitative MS
 LECTURE-15 itraq Clinical Applications HANDOUT PREAMBLE Isobaric Tagging for Relative and Absolute quantitation (itraq) is a quantitative MS based method for quantifying proteins subject to various different
LECTURE-15 itraq Clinical Applications HANDOUT PREAMBLE Isobaric Tagging for Relative and Absolute quantitation (itraq) is a quantitative MS based method for quantifying proteins subject to various different
Chapter 9. Cellular Signaling
 Chapter 9 Cellular Signaling Cellular Messaging Page 215 Cells can signal to each other and interpret the signals they receive from other cells and the environment Signals are most often chemicals The
Chapter 9 Cellular Signaling Cellular Messaging Page 215 Cells can signal to each other and interpret the signals they receive from other cells and the environment Signals are most often chemicals The
Protein Modification Overview DEFINITION The modification of selected residues in a protein and not as a component of synthesis
 Lecture Four: Protein Modification & Cleavage [Based on Chapters 2, 9, 10 & 11 Berg, Tymoczko & Stryer] (Figures in red are for the 7th Edition) (Figures in Blue are for the 8th Edition) Protein Modification
Lecture Four: Protein Modification & Cleavage [Based on Chapters 2, 9, 10 & 11 Berg, Tymoczko & Stryer] (Figures in red are for the 7th Edition) (Figures in Blue are for the 8th Edition) Protein Modification
Cell Signaling part 2
 15 Cell Signaling part 2 Functions of Cell Surface Receptors Other cell surface receptors are directly linked to intracellular enzymes. The largest family of these is the receptor protein tyrosine kinases,
15 Cell Signaling part 2 Functions of Cell Surface Receptors Other cell surface receptors are directly linked to intracellular enzymes. The largest family of these is the receptor protein tyrosine kinases,
CELLS. Cells. Basic unit of life (except virus)
 Basic unit of life (except virus) CELLS Prokaryotic, w/o nucleus, bacteria Eukaryotic, w/ nucleus Various cell types specialized for particular function. Differentiation. Over 200 human cell types 56%
Basic unit of life (except virus) CELLS Prokaryotic, w/o nucleus, bacteria Eukaryotic, w/ nucleus Various cell types specialized for particular function. Differentiation. Over 200 human cell types 56%
Ph.D. Thesis: Protective immune response in P.falciparum malaria 2011 CHAPTER I: Introduction. S.D. Lourembam 16
 CHAPTER I: Introduction S.D. Lourembam 16 1. INTRODUCTION Malaria remains a major global health problem with 300 to 500 million clinical infections and more than a million deaths reported each year. In
CHAPTER I: Introduction S.D. Lourembam 16 1. INTRODUCTION Malaria remains a major global health problem with 300 to 500 million clinical infections and more than a million deaths reported each year. In
Molecular Graphics Perspective of Protein Structure and Function
 Molecular Graphics Perspective of Protein Structure and Function VMD Highlights > 20,000 registered Users Platforms: Unix (16 builds) Windows MacOS X Display of large biomolecules and simulation trajectories
Molecular Graphics Perspective of Protein Structure and Function VMD Highlights > 20,000 registered Users Platforms: Unix (16 builds) Windows MacOS X Display of large biomolecules and simulation trajectories
Key Concept B F. How do peptides get loaded onto the proper kind of MHC molecule?
 Location of MHC class I pockets termed B and F that bind P and P9 amino acid side chains of the peptide Different MHC alleles confer different functional properties on the adaptive immune system by specifying
Location of MHC class I pockets termed B and F that bind P and P9 amino acid side chains of the peptide Different MHC alleles confer different functional properties on the adaptive immune system by specifying
B F. Location of MHC class I pockets termed B and F that bind P2 and P9 amino acid side chains of the peptide
 Different MHC alleles confer different functional properties on the adaptive immune system by specifying molecules that have different peptide binding abilities Location of MHC class I pockets termed B
Different MHC alleles confer different functional properties on the adaptive immune system by specifying molecules that have different peptide binding abilities Location of MHC class I pockets termed B
Identification and characterization of merozoite surface protein 1 epitope
 Identification and characterization of merozoite surface protein epitope Satarudra Prakash Singh,2, Bhartendu Nath Mishra 2* Amity Institute of Biotechnology, Amity University, Uttar Pradesh, Gomti Nagar,
Identification and characterization of merozoite surface protein epitope Satarudra Prakash Singh,2, Bhartendu Nath Mishra 2* Amity Institute of Biotechnology, Amity University, Uttar Pradesh, Gomti Nagar,
Selective inhibition of a two-step egress of malaria parasites from the host erythrocyte. Melbourne 3050, Australia. Melbourne 3050, Australia
 JBC Papers in Press. Published on July 11, 2003 as Manuscript M305252200 1 Selective inhibition of a two-step egress of malaria parasites from the host erythrocyte Mark E. Wickham 1, Janetta G. Culvenor
JBC Papers in Press. Published on July 11, 2003 as Manuscript M305252200 1 Selective inhibition of a two-step egress of malaria parasites from the host erythrocyte Mark E. Wickham 1, Janetta G. Culvenor
BIOL 4374/BCHS 4313 Cell Biology Exam #1 February 13, 2001
 BIOL 4374/BCHS 4313 Cell Biology Exam #1 February 13, 2001 SS# Name This exam is worth a total of 100 points. The number of points each question is worth is shown in parentheses. Good luck! 1. (2) The
BIOL 4374/BCHS 4313 Cell Biology Exam #1 February 13, 2001 SS# Name This exam is worth a total of 100 points. The number of points each question is worth is shown in parentheses. Good luck! 1. (2) The
number Done by Corrected by Doctor Dr. Diala
 number 30 Done by Dergam Al-Tarawneh Corrected by Zaid Emad Doctor Dr. Diala 1 After we ve finished talking about lipids metabolism pathways, today we will start talking about another pathway that takes
number 30 Done by Dergam Al-Tarawneh Corrected by Zaid Emad Doctor Dr. Diala 1 After we ve finished talking about lipids metabolism pathways, today we will start talking about another pathway that takes
Signal Transduction Cascades
 Signal Transduction Cascades Contents of this page: Kinases & phosphatases Protein Kinase A (camp-dependent protein kinase) G-protein signal cascade Structure of G-proteins Small GTP-binding proteins,
Signal Transduction Cascades Contents of this page: Kinases & phosphatases Protein Kinase A (camp-dependent protein kinase) G-protein signal cascade Structure of G-proteins Small GTP-binding proteins,
Cell Injury MECHANISMS OF CELL INJURY
 Cell Injury MECHANISMS OF CELL INJURY The cellular response to injurious stimuli depends on the following factors: Type of injury, Its duration, and Its severity. Thus, low doses of toxins or a brief duration
Cell Injury MECHANISMS OF CELL INJURY The cellular response to injurious stimuli depends on the following factors: Type of injury, Its duration, and Its severity. Thus, low doses of toxins or a brief duration
Life Science 1A Final Exam. January 19, 2006
 ame: TF: Section Time Life Science 1A Final Exam January 19, 2006 Please write legibly in the space provided below each question. You may not use calculators on this exam. We prefer that you use non-erasable
ame: TF: Section Time Life Science 1A Final Exam January 19, 2006 Please write legibly in the space provided below each question. You may not use calculators on this exam. We prefer that you use non-erasable
AP Biology Cells: Chapters 4 & 5
 AP Biology Cells: Chapters 4 & 5 Multiple Choice Identify the choice that best completes the statement or answers the question. 1. The was the first unifying principle of biology. a. spontaneous generation
AP Biology Cells: Chapters 4 & 5 Multiple Choice Identify the choice that best completes the statement or answers the question. 1. The was the first unifying principle of biology. a. spontaneous generation
A. Major parts 1. Nucleus 2. Cytoplasm a. Contain organelles (see below) 3. Plasma membrane (To be discussed in Cellular Transport Lecture)
 Lecture 5: Cellular Biology I. Cell Theory Concepts: 1. Cells are the functional and structural units of living organisms 2. The activity of an organism is dependent on both the individual and collective
Lecture 5: Cellular Biology I. Cell Theory Concepts: 1. Cells are the functional and structural units of living organisms 2. The activity of an organism is dependent on both the individual and collective
The Nobel Prize in Chemistry 2004
 The Nobel Prize in Chemistry 2004 Ubiquitous Quality Control of Life C S Karigar and K R Siddalinga Murthy The Nobel Prize in Chemistry for 2004 is shared by Aaron Ciechanover, Avram Hershko and Irwin
The Nobel Prize in Chemistry 2004 Ubiquitous Quality Control of Life C S Karigar and K R Siddalinga Murthy The Nobel Prize in Chemistry for 2004 is shared by Aaron Ciechanover, Avram Hershko and Irwin
Summary of Endomembrane-system
 Summary of Endomembrane-system 1. Endomembrane System: The structural and functional relationship organelles including ER,Golgi complex, lysosome, endosomes, secretory vesicles. 2. Membrane-bound structures
Summary of Endomembrane-system 1. Endomembrane System: The structural and functional relationship organelles including ER,Golgi complex, lysosome, endosomes, secretory vesicles. 2. Membrane-bound structures
TRANSPORT PROCESSES. 1b. moving proteins into membranes and organelles
 1b. moving proteins into membranes and organelles SLIDE 1 A typical mammalian cell contains up to 10,000 different kinds of proteins. The vast majority of these proteins are synthesized by cytosolic ribosomes,
1b. moving proteins into membranes and organelles SLIDE 1 A typical mammalian cell contains up to 10,000 different kinds of proteins. The vast majority of these proteins are synthesized by cytosolic ribosomes,
Bone marrow reticulocytes: a Plasmodium vivax affair? Alfredo Mayor a and Pietro Alano b
 Bone marrow reticulocytes: a Plasmodium vivax affair? Alfredo Mayor a and Pietro Alano b a ISGlobal, Barcelona Ctr. Int. Health Res. (CRESIB), Hospital Clínic - Universitat de Barcelona, Barcelona, Spain;
Bone marrow reticulocytes: a Plasmodium vivax affair? Alfredo Mayor a and Pietro Alano b a ISGlobal, Barcelona Ctr. Int. Health Res. (CRESIB), Hospital Clínic - Universitat de Barcelona, Barcelona, Spain;
Cellular compartments
 Cellular compartments 1. Cellular compartments and their function 2. Evolution of cellular compartments 3. How to make a 3D model of cellular compartment 4. Cell organelles in the fluorescent microscope
Cellular compartments 1. Cellular compartments and their function 2. Evolution of cellular compartments 3. How to make a 3D model of cellular compartment 4. Cell organelles in the fluorescent microscope
Cell cycle and apoptosis
 Cell cycle and apoptosis Cell cycle Definition Stages and steps Cell cycle Interphase (G1/G0, S, and G2) Mitosis (prophase, metaphase, anaphase, telophase, karyokinesis, cytokinesis) Control checkpoints
Cell cycle and apoptosis Cell cycle Definition Stages and steps Cell cycle Interphase (G1/G0, S, and G2) Mitosis (prophase, metaphase, anaphase, telophase, karyokinesis, cytokinesis) Control checkpoints
PAPER No. : 16, Bioorganic and biophysical chemistry MODULE No. : 22, Mechanism of enzyme catalyst reaction (I) Chymotrypsin
 Subject Paper No and Title 16 Bio-organic and Biophysical Module No and Title 22 Mechanism of Enzyme Catalyzed reactions I Module Tag CHE_P16_M22 Chymotrypsin TABLE OF CONTENTS 1. Learning outcomes 2.
Subject Paper No and Title 16 Bio-organic and Biophysical Module No and Title 22 Mechanism of Enzyme Catalyzed reactions I Module Tag CHE_P16_M22 Chymotrypsin TABLE OF CONTENTS 1. Learning outcomes 2.
Cellular functions of protein degradation
 Protein Degradation Cellular functions of protein degradation 1. Elimination of misfolded and damaged proteins: Environmental toxins, translation errors and genetic mutations can damage proteins. Misfolded
Protein Degradation Cellular functions of protein degradation 1. Elimination of misfolded and damaged proteins: Environmental toxins, translation errors and genetic mutations can damage proteins. Misfolded
Insulin mrna to Protein Kit
 Insulin mrna to Protein Kit A 3DMD Paper BioInformatics and Mini-Toober Folding Activity Student Handout www.3dmoleculardesigns.com Insulin mrna to Protein Kit Contents Becoming Familiar with the Data...
Insulin mrna to Protein Kit A 3DMD Paper BioInformatics and Mini-Toober Folding Activity Student Handout www.3dmoleculardesigns.com Insulin mrna to Protein Kit Contents Becoming Familiar with the Data...
Fabio T M Costa, PhD. University of Campinas (UNICAMP); Campinas, SP, Brazil. Supported by:
 Fabio T M Costa, PhD University of Campinas (UNICAMP); Campinas, SP, Brazil Supported by: P. falciparum associated pathologies In falciparum malaria cytoadhesion of mature forms are related to fatalities!
Fabio T M Costa, PhD University of Campinas (UNICAMP); Campinas, SP, Brazil Supported by: P. falciparum associated pathologies In falciparum malaria cytoadhesion of mature forms are related to fatalities!
MBios 401/501: Lecture 12.1 Signaling IV. Slide 1
 MBios 401/501: Lecture 12.1 Signaling IV Slide 1 Pathways that require regulated proteolysis 1. Notch and Delta 2. Wnt/ b-catenin 3. Hedgehog 4. NFk-B Our last topic on cell signaling are pathways that
MBios 401/501: Lecture 12.1 Signaling IV Slide 1 Pathways that require regulated proteolysis 1. Notch and Delta 2. Wnt/ b-catenin 3. Hedgehog 4. NFk-B Our last topic on cell signaling are pathways that
Cell Cycle. Trends in Cell Biology
 Cell Cycle Trends in Cell Biology Cell Cycle The orderly sequence of events by which a cell duplicates its contents and divides into two Daughter Cells Activities of a cell from one cell division to the
Cell Cycle Trends in Cell Biology Cell Cycle The orderly sequence of events by which a cell duplicates its contents and divides into two Daughter Cells Activities of a cell from one cell division to the
Propagation of the Signal
 OpenStax-CNX module: m44452 1 Propagation of the Signal OpenStax College This work is produced by OpenStax-CNX and licensed under the Creative Commons Attribution License 3.0 By the end of this section,
OpenStax-CNX module: m44452 1 Propagation of the Signal OpenStax College This work is produced by OpenStax-CNX and licensed under the Creative Commons Attribution License 3.0 By the end of this section,
BACTERIAL PATHOGENESIS
 BACTERIAL PATHOGENESIS A pathogen is a microorganism that is able to cause disease. Pathogenicity is the ability to produce disease in a host organism. Virulence a term which refers to the degree of pathogenicity
BACTERIAL PATHOGENESIS A pathogen is a microorganism that is able to cause disease. Pathogenicity is the ability to produce disease in a host organism. Virulence a term which refers to the degree of pathogenicity
Cover Page. The following handle holds various files of this Leiden University dissertation:
 Cover Page The following handle holds various files of this Leiden University dissertation: http://hdl.handle.net/188/59466 Author: Oudshoorn, D. Title: Wrapping up : nidovirus membrane structures and
Cover Page The following handle holds various files of this Leiden University dissertation: http://hdl.handle.net/188/59466 Author: Oudshoorn, D. Title: Wrapping up : nidovirus membrane structures and
Cell Communication. Local and Long Distance Signaling
 Cell Communication Cell to cell communication is essential for multicellular organisms Some universal mechanisms of cellular regulation providing more evidence for the evolutionary relatedness of all life
Cell Communication Cell to cell communication is essential for multicellular organisms Some universal mechanisms of cellular regulation providing more evidence for the evolutionary relatedness of all life
Parasitic Protozoa, Helminths, and Arthropod Vectors
 PowerPoint Lecture Slides for MICROBIOLOGY ROBERT W. BAUMAN Chapter 23 Parasitic Protozoa, Helminths, and Arthropod Vectors Parasitic Diseases Protozoan and helminthic parasites are emerging as serious
PowerPoint Lecture Slides for MICROBIOLOGY ROBERT W. BAUMAN Chapter 23 Parasitic Protozoa, Helminths, and Arthropod Vectors Parasitic Diseases Protozoan and helminthic parasites are emerging as serious
SHORT LINEAR MOTIFS. Holger Dinkel EMBO Practical Course Computational analysis of protein-protein interactions From sequences to networks
 Holger Dinkel EMBO Practical Course Computational analysis of protein-protein interactions From sequences to networks IMPORTANCE OF 2 / 19 IMPORTANCE OF 2 / 19 IMPORTANCE OF 2 / 19 IMPORTANCE OF 2 / 19
Holger Dinkel EMBO Practical Course Computational analysis of protein-protein interactions From sequences to networks IMPORTANCE OF 2 / 19 IMPORTANCE OF 2 / 19 IMPORTANCE OF 2 / 19 IMPORTANCE OF 2 / 19
I. Fluid Mosaic Model A. Biological membranes are lipid bilayers with associated proteins
 Lecture 6: Membranes and Cell Transport Biological Membranes I. Fluid Mosaic Model A. Biological membranes are lipid bilayers with associated proteins 1. Characteristics a. Phospholipids form bilayers
Lecture 6: Membranes and Cell Transport Biological Membranes I. Fluid Mosaic Model A. Biological membranes are lipid bilayers with associated proteins 1. Characteristics a. Phospholipids form bilayers
FurinDB: A Database of 20-Residue Furin Cleavage Site Motifs, Substrates and Their Associated Drugs
 Int. J. Mol. Sci. 2011, 12, 1060-1065; doi:10.3390/ijms12021060 OPEN ACCESS Technical Note International Journal of Molecular Sciences ISSN 1422-0067 www.mdpi.com/journal/ijms FurinDB: A Database of 20-Residue
Int. J. Mol. Sci. 2011, 12, 1060-1065; doi:10.3390/ijms12021060 OPEN ACCESS Technical Note International Journal of Molecular Sciences ISSN 1422-0067 www.mdpi.com/journal/ijms FurinDB: A Database of 20-Residue
Proteomic Analysis of Fractionated Toxoplasma Oocysts Reveals Clues to Their Environmental Resistance
 Proteomic Analysis of Fractionated Toxoplasma Oocysts Reveals Clues to Their Environmental Resistance Heather M. Fritz 1., Paul W. Bowyer 2,3., Matthew Bogyo 2,3, Patricia A. Conrad 1 *, John C. Boothroyd
Proteomic Analysis of Fractionated Toxoplasma Oocysts Reveals Clues to Their Environmental Resistance Heather M. Fritz 1., Paul W. Bowyer 2,3., Matthew Bogyo 2,3, Patricia A. Conrad 1 *, John C. Boothroyd
Formation of the Food Vacuole in Plasmodium falciparum: A Potential Role for the 19 kda Fragment of Merozoite Surface Protein 1 (MSP1 19 )
 Formation of the Food Vacuole in Plasmodium falciparum: A Potential Role for the 19 kda Fragment of Merozoite Surface Protein 1 (MSP1 19 ) Anton R. Dluzewski 1,3. *, Irene T. Ling 3., John M. Hopkins 1,2,
Formation of the Food Vacuole in Plasmodium falciparum: A Potential Role for the 19 kda Fragment of Merozoite Surface Protein 1 (MSP1 19 ) Anton R. Dluzewski 1,3. *, Irene T. Ling 3., John M. Hopkins 1,2,
Reading Packet 2- Cells Unit. Chapter 6: A Tour of the Cell 1. What is resolving power?
 AP Biology Reading Packet 2- Cells Unit Name Chapter 6: A Tour of the Cell 1. What is resolving power? 2. How is an electron microscope different from a light microscope and what is the difference between
AP Biology Reading Packet 2- Cells Unit Name Chapter 6: A Tour of the Cell 1. What is resolving power? 2. How is an electron microscope different from a light microscope and what is the difference between
2013 W. H. Freeman and Company. 12 Signal Transduction
 2013 W. H. Freeman and Company 12 Signal Transduction CHAPTER 12 Signal Transduction Key topics: General features of signal transduction Structure and function of G protein coupled receptors Structure
2013 W. H. Freeman and Company 12 Signal Transduction CHAPTER 12 Signal Transduction Key topics: General features of signal transduction Structure and function of G protein coupled receptors Structure
Cancer and Tyrosine Kinase Inhibition
 Cancer and Tyrosine Kinase Inhibition 1 Motivation (1) In first world countries cancer is the second most common cause of death after cardiovascular diseases. For most tumors, treatment is limited to surgery,
Cancer and Tyrosine Kinase Inhibition 1 Motivation (1) In first world countries cancer is the second most common cause of death after cardiovascular diseases. For most tumors, treatment is limited to surgery,
10/5/2015. Cell Size. Relative Rate of Reaction
 The Cell Biology 102 Fundamental unit of life Smallest unit that displays all the basic elements of life Lecture 5: Cells Cell Theory 1. All living things are made of one or more cells Cell Theory 2. The
The Cell Biology 102 Fundamental unit of life Smallest unit that displays all the basic elements of life Lecture 5: Cells Cell Theory 1. All living things are made of one or more cells Cell Theory 2. The
Protein sorting (endoplasmic reticulum) Dr. Diala Abu-Hsasan School of Medicine
 Protein sorting (endoplasmic reticulum) Dr. Diala Abu-Hsasan School of Medicine dr.abuhassand@gmail.com An overview of cellular components Endoplasmic reticulum (ER) It is a network of membrane-enclosed
Protein sorting (endoplasmic reticulum) Dr. Diala Abu-Hsasan School of Medicine dr.abuhassand@gmail.com An overview of cellular components Endoplasmic reticulum (ER) It is a network of membrane-enclosed
University of York Department of Biology B. Sc Stage 3 Degree Examinations Human Molecular Parasitology. Submission deadline: 12 noon
 Examination Candidate Number: University of York Department of Biology B. Sc Stage 3 Degree Examinations 2017-18 Human Molecular Parasitology Submission deadline: 12 noon Work should be submitted via the
Examination Candidate Number: University of York Department of Biology B. Sc Stage 3 Degree Examinations 2017-18 Human Molecular Parasitology Submission deadline: 12 noon Work should be submitted via the
Antigen Presentation to T lymphocytes
 Antigen Presentation to T lymphocytes Immunology 441 Lectures 6 & 7 Chapter 6 October 10 & 12, 2016 Jessica Hamerman jhamerman@benaroyaresearch.org Office hours by arrangement Antibodies and T cell receptors
Antigen Presentation to T lymphocytes Immunology 441 Lectures 6 & 7 Chapter 6 October 10 & 12, 2016 Jessica Hamerman jhamerman@benaroyaresearch.org Office hours by arrangement Antibodies and T cell receptors
GPI-anchored glycoproteins Mike Ferguson
 GPI-Anchored Glycoproteins Prof. College of Life Sciences University of Dundee UK 1 What are GPI membrane anchors? GPI stands for glycosylphosphatidylinositol In other words, a phosphatidylinositol (PI)
GPI-Anchored Glycoproteins Prof. College of Life Sciences University of Dundee UK 1 What are GPI membrane anchors? GPI stands for glycosylphosphatidylinositol In other words, a phosphatidylinositol (PI)
BIOL 4374/BCHS 4313 Cell Biology Exam #2 March 22, 2001
 BIOL 4374/BCHS 4313 Cell Biology Exam #2 March 22, 2001 SS# Name This exam is worth a total of 100 points. The number of points each question is worth is shown in parentheses. Good luck! 1. (2) In the
BIOL 4374/BCHS 4313 Cell Biology Exam #2 March 22, 2001 SS# Name This exam is worth a total of 100 points. The number of points each question is worth is shown in parentheses. Good luck! 1. (2) In the
Virus and Prokaryotic Gene Regulation - 1
 Virus and Prokaryotic Gene Regulation - 1 We have discussed the molecular structure of DNA and its function in DNA duplication and in transcription and protein synthesis. We now turn to how cells regulate
Virus and Prokaryotic Gene Regulation - 1 We have discussed the molecular structure of DNA and its function in DNA duplication and in transcription and protein synthesis. We now turn to how cells regulate
1. endoplasmic reticulum This is the location where N-linked oligosaccharide is initially synthesized and attached to glycoproteins.
 Biology 4410 Name Spring 2006 Exam 2 A. Multiple Choice, 2 pt each Pick the best choice from the list of choices, and write it in the space provided. Some choices may be used more than once, and other
Biology 4410 Name Spring 2006 Exam 2 A. Multiple Choice, 2 pt each Pick the best choice from the list of choices, and write it in the space provided. Some choices may be used more than once, and other
1. This is the location where N-linked oligosaccharide is initially synthesized and attached to glycoproteins.
 Biology 4410 Name Spring 2006 Exam 2 A. Multiple Choice, 2 pt each Pick the best choice from the list of choices, and write it in the space provided. Some choices may be used more than once, and other
Biology 4410 Name Spring 2006 Exam 2 A. Multiple Choice, 2 pt each Pick the best choice from the list of choices, and write it in the space provided. Some choices may be used more than once, and other
SIBLINGs, cancer's multifunctional weapons
 SIBLINGs, cancer's multifunctional weapons 6/18/08 Akeila Bellahcène and Vincent Castronovo of the Metastasis Research laboratory of the University of Liège are among the first researchers to have discovered
SIBLINGs, cancer's multifunctional weapons 6/18/08 Akeila Bellahcène and Vincent Castronovo of the Metastasis Research laboratory of the University of Liège are among the first researchers to have discovered
Invasion of Red Blood Cells by Malaria Parasites
 Leading Edge Review Invasion of Red Blood Cells by Malaria Parasites Alan F. Cowman 1, * and Brendan S. Crabb 1, * 1 The Walter and Eliza Hall Institute of Medical Research, Melbourne 3050, Australia *Contact:
Leading Edge Review Invasion of Red Blood Cells by Malaria Parasites Alan F. Cowman 1, * and Brendan S. Crabb 1, * 1 The Walter and Eliza Hall Institute of Medical Research, Melbourne 3050, Australia *Contact:
Molecular Cell Biology Problem Drill 16: Intracellular Compartment and Protein Sorting
 Molecular Cell Biology Problem Drill 16: Intracellular Compartment and Protein Sorting Question No. 1 of 10 Question 1. Which of the following statements about the nucleus is correct? Question #01 A. The
Molecular Cell Biology Problem Drill 16: Intracellular Compartment and Protein Sorting Question No. 1 of 10 Question 1. Which of the following statements about the nucleus is correct? Question #01 A. The
The death receptors: signaling and modulation
 The death receptors: signaling and modulation 1 1 The extrinsic cell death pathway 2 Nat Rev Drug Discov. 2008 Dec;7(12):1001-12. 2 Death receptors Belong to the tumor necrosis factor (TNF) receptor gene
The death receptors: signaling and modulation 1 1 The extrinsic cell death pathway 2 Nat Rev Drug Discov. 2008 Dec;7(12):1001-12. 2 Death receptors Belong to the tumor necrosis factor (TNF) receptor gene
Malaria parasite vaccine development Strategies & Targets
 Malaria parasite vaccine development Strategies & Targets Tulane University Ahmed Aly Most malaria disease deaths are among children and pregnant women A child or a pregnant woman dies of malaria nearly
Malaria parasite vaccine development Strategies & Targets Tulane University Ahmed Aly Most malaria disease deaths are among children and pregnant women A child or a pregnant woman dies of malaria nearly
endomembrane system internal membranes origins transport of proteins chapter 15 endomembrane system
 endo system chapter 15 internal s endo system functions as a coordinated unit divide cytoplasm into distinct compartments controls exocytosis and endocytosis movement of molecules which cannot pass through
endo system chapter 15 internal s endo system functions as a coordinated unit divide cytoplasm into distinct compartments controls exocytosis and endocytosis movement of molecules which cannot pass through
17/01/2017. Protein trafficking between cell compartments. Lecture 3: The cytosol. The mitochondrion - the power plant of the cell
 ell biology 2017 version 13/1 2017 ote endosome vs lysosome handout Lecture 3: Text book Alberts et al.: hapter 12-14 (Topics covered by the lecture) A lot of reading! Focus on principles ell Biology interactive
ell biology 2017 version 13/1 2017 ote endosome vs lysosome handout Lecture 3: Text book Alberts et al.: hapter 12-14 (Topics covered by the lecture) A lot of reading! Focus on principles ell Biology interactive
Determination of the temporal pattern and importance of BALF1 expression in Epstein-Barr viral infection
 Determination of the temporal pattern and importance of BALF1 expression in Epstein-Barr viral infection Melissa Mihelidakis May 6, 2004 7.340 Research Proposal Introduction Apoptosis, or programmed cell
Determination of the temporal pattern and importance of BALF1 expression in Epstein-Barr viral infection Melissa Mihelidakis May 6, 2004 7.340 Research Proposal Introduction Apoptosis, or programmed cell
Biochemistry 2 Recita0on Amino Acid Metabolism
 Biochemistry 2 Recita0on Amino Acid Metabolism 04-20- 2015 Glutamine and Glutamate as key entry points for NH 4 + Amino acid catabolism Glutamine synthetase enables toxic NH 4 + to combine with glutamate
Biochemistry 2 Recita0on Amino Acid Metabolism 04-20- 2015 Glutamine and Glutamate as key entry points for NH 4 + Amino acid catabolism Glutamine synthetase enables toxic NH 4 + to combine with glutamate
Cell Cell
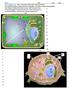 Go to cellsalive.com. Select Interactive Cell Models: Plant and Animal. Fill in the information on Plant and Animal Organelles, then Click on Start the Animation Select Plant or Animal Cell below the box.
Go to cellsalive.com. Select Interactive Cell Models: Plant and Animal. Fill in the information on Plant and Animal Organelles, then Click on Start the Animation Select Plant or Animal Cell below the box.
Cell Communication. Chapter 11. Key Concepts in Chapter 11. Cellular Messaging. Cell-to-cell communication is essential for multicellular organisms
 Chapter 11 Cell Communication Dr. Wendy Sera Houston Community College Biology 1406 Key Concepts in Chapter 11 1. External signals are converted to responses within the cell. 2. Reception: A signaling
Chapter 11 Cell Communication Dr. Wendy Sera Houston Community College Biology 1406 Key Concepts in Chapter 11 1. External signals are converted to responses within the cell. 2. Reception: A signaling
Cell Structure and Function
 Cell Theory Cell Structure and Function Chapter 6 Pg. 94-124 What is a cell? The basic functional unit of all living things. The Cell Theory states All organisms are made of one or more cells. Cells are
Cell Theory Cell Structure and Function Chapter 6 Pg. 94-124 What is a cell? The basic functional unit of all living things. The Cell Theory states All organisms are made of one or more cells. Cells are
Enzymes: Regulation 2-3
 Enzymes: Regulation 2-3 Reversible covalent modification Association with regulatory proteins Irreversible covalent modification/proteolytic cleavage Reading: Berg, Tymoczko & Stryer, 6th ed., Chapter
Enzymes: Regulation 2-3 Reversible covalent modification Association with regulatory proteins Irreversible covalent modification/proteolytic cleavage Reading: Berg, Tymoczko & Stryer, 6th ed., Chapter
G-Protein Signaling. Introduction to intracellular signaling. Dr. SARRAY Sameh, Ph.D
 G-Protein Signaling Introduction to intracellular signaling Dr. SARRAY Sameh, Ph.D Cell signaling Cells communicate via extracellular signaling molecules (Hormones, growth factors and neurotransmitters
G-Protein Signaling Introduction to intracellular signaling Dr. SARRAY Sameh, Ph.D Cell signaling Cells communicate via extracellular signaling molecules (Hormones, growth factors and neurotransmitters
18. PANCREATIC FUNCTION AND METABOLISM. Pancreatic secretions ISLETS OF LANGERHANS. Insulin
 18. PANCREATIC FUNCTION AND METABOLISM ISLETS OF LANGERHANS Some pancreatic functions have already been discussed in the digestion section. In this one, the emphasis will be placed on the endocrine function
18. PANCREATIC FUNCTION AND METABOLISM ISLETS OF LANGERHANS Some pancreatic functions have already been discussed in the digestion section. In this one, the emphasis will be placed on the endocrine function
Apicoplast. Treatments and New drug targets
 Treatments and New drug targets Apicoplast What is the apicoplast? Where does it come from? How are proteins targeted to the organelle? How does the organelle replicate? What is the function of the organelle?
Treatments and New drug targets Apicoplast What is the apicoplast? Where does it come from? How are proteins targeted to the organelle? How does the organelle replicate? What is the function of the organelle?
Lecture 6 9/17 Dr. Hirsh Organization of Cells, continued
 Cell structure of Eukaryotic cells Lecture 6 9/17 Dr. Hirsh Organization of Cells, continued Lots of double-membraned organelles Existence of an Endo-membrane system separation of areas of cell, transport
Cell structure of Eukaryotic cells Lecture 6 9/17 Dr. Hirsh Organization of Cells, continued Lots of double-membraned organelles Existence of an Endo-membrane system separation of areas of cell, transport
COURSE: Medical Microbiology, MBIM 650/720 - Fall TOPIC: Antigen Processing, MHC Restriction, & Role of Thymus Lecture 12
 COURSE: Medical Microbiology, MBIM 650/720 - Fall 2008 TOPIC: Antigen Processing, MHC Restriction, & Role of Thymus Lecture 12 FACULTY: Dr. Mayer Office: Bldg. #1, Rm B32 Phone: 733-3281 Email: MAYER@MED.SC.EDU
COURSE: Medical Microbiology, MBIM 650/720 - Fall 2008 TOPIC: Antigen Processing, MHC Restriction, & Role of Thymus Lecture 12 FACULTY: Dr. Mayer Office: Bldg. #1, Rm B32 Phone: 733-3281 Email: MAYER@MED.SC.EDU
Introduction. Biochemistry: It is the chemistry of living things (matters).
 Introduction Biochemistry: It is the chemistry of living things (matters). Biochemistry provides fundamental understanding of the molecular basis for the function and malfunction of living things. Biochemistry
Introduction Biochemistry: It is the chemistry of living things (matters). Biochemistry provides fundamental understanding of the molecular basis for the function and malfunction of living things. Biochemistry
Protein kinases are enzymes that add a phosphate group to proteins according to the. ATP + protein OH > Protein OPO 3 + ADP
 Protein kinase Protein kinases are enzymes that add a phosphate group to proteins according to the following equation: 2 ATP + protein OH > Protein OPO 3 + ADP ATP represents adenosine trisphosphate, ADP
Protein kinase Protein kinases are enzymes that add a phosphate group to proteins according to the following equation: 2 ATP + protein OH > Protein OPO 3 + ADP ATP represents adenosine trisphosphate, ADP
The Cell and Cellular transport
 Cell theory (1838): The Cell 1. All organisms are composed of one or more cells, and the life processes of metabolism and heredity occur within these cells. 2. Cells are the smallest living things, the
Cell theory (1838): The Cell 1. All organisms are composed of one or more cells, and the life processes of metabolism and heredity occur within these cells. 2. Cells are the smallest living things, the
Cell morphology. Cell organelles structure and function. Chapter 1: UNIT 1. Dr. Charushila Rukadikar
 UNIT 1 Cell morphology Cell organelles structure and function Chapter 1: Dr. Charushila Rukadikar Assistant Professor Department Of Physiology ZMCH, Dahod Physiology The science that is concerned with
UNIT 1 Cell morphology Cell organelles structure and function Chapter 1: Dr. Charushila Rukadikar Assistant Professor Department Of Physiology ZMCH, Dahod Physiology The science that is concerned with
Animal Tissue Culture SQG 3242 Biology of Cultured Cells. Dr. Siti Pauliena Mohd Bohari
 Animal Tissue Culture SQG 3242 Biology of Cultured Cells Dr. Siti Pauliena Mohd Bohari The Culture Environment Changes of Cell s microenvironment needed that favor the spreading, migration, and proliferation
Animal Tissue Culture SQG 3242 Biology of Cultured Cells Dr. Siti Pauliena Mohd Bohari The Culture Environment Changes of Cell s microenvironment needed that favor the spreading, migration, and proliferation
