COMICR 378. Addresses 1 The Walter and Eliza Hall Institute of Medical Research, Parkville,
|
|
|
- Valentine Tyler
- 5 years ago
- Views:
Transcription
1 Protein targeting to destinations of the secretory pathway in the malaria parasite Plasmodium falciparum Christopher J Tonkin 1, J Andrew Pearce 1, Geoffrey I McFadden 2 and Alan F Cowman 1 The secretory pathway in the malaria parasite Plasmodium falciparum has many unique aspects in terms of protein destinations and trafficking mechanisms. Recently, several exciting insights into protein trafficking within this intracellular parasite have been unveiled: these include signals that are required for targeting of proteins to the red blood cell and the relict plastid (known as the apicoplast); and the elucidation of the pathways of the haemoglobin proteases targeted to the food vacuole. Protein-targeting to the apical organelles in P. falciparum, however, is still not very well understood, but available research offers a tantalising glimpse of the system. Addresses 1 The Walter and Eliza Hall Institute of Medical Research, Parkville, Melbourne, 3010, Australia 2 The School of Botany, The University of Melbourne, Parkville, Melbourne, 3050, Australia Corresponding author: Cowman, Alan F (cowman@wehi.edu.au) This review comes from a themed issue on Host microbe interactions: parasites Edited by Brendan Crabb /$ see front matter # 2006 Elsevier Ltd. All rights reserved. DOI /j.mib Introduction Plasmodium falciparum is the causative agent of the most severe form of malaria and infects over 300 million people and kills approximately 2 million people annually. Most of the pathology associated with P. falciparum infection occurs during the asexual cycle when this unicellular eukaryotic parasite invades, rapidly grows and divides into multiple daughter cells before finally bursting from the host. The malaria parasite P. falciparum has a range of unique organelles all fed by the protein secretory pathway. Indeed, protein secretory pathways are fundamental to the biology and pathology of P. falciparum infection and have great potential as targets for intervention. Like all other eukaryotes, most secretory proteins in P. falciparum contain an N terminal signal peptide for translocation into the ER and additional protein-targeting motifs that provide essential information for subsequent targeting to secretory destinations. There are at least 12 secretory destinations in P. falciparum, many of them unique and some of the signals and pathways of proteins travelling to such destinations have recently been revealed. The focus of this review is on recent advances in understanding the organization of the P. falciparum secretory pathway, with special emphasis on protein targeting to parasite organelles. Trafficking to the host red blood cell During invasion into the erythrocyte, the parasite constructs a parasitophorous vacuole membrane (PVM) that encloses it within a parasitophorous vacuole (PV) and separates it from the host cell cytosol (Figure 1). In order to grow and develop within this protected compartment, the parasite activates a remarkable host cell remodelling process. This converts a simple, terminally differentiated erythrocyte into an ideal host cell that supports the parasite s nutritional needs, and allows adherence of the cell to vascular endothelia in order to avoid detection by the host immune mechanisms (Figure 2). This remodelling requires trafficking of proteins out of the parasite and into the host cell. Recently, advances have been made in the understanding of this process, such as the elucidation of the PEXEL/VTS (plasmodium export element/vacuolar targeting signal) targeting motif that is responsible for translocation proteins across the PVM [1,2 ] and the identification of the first molecule involved in host cell targeting (Figure 2) [3 ]. There have been several recently published reviews that comprehensively discuss this aspect of the secretory pathway [4,5]: instead of re-reviewing the literature here, the recent findings have been summarized in Figure 1. Trafficking to the food vacuole Rapid growth of the parasite within the host erythrocyte is achieved, in part, by digesting haemoglobin within a lysosomal-like compartment that is referred to as the food vacuole (FV). Haemoglobin proteolysis takes place by a series of parasite-derived proteases [6]. All of the known FV proteases bear a putative N terminal signalpeptide, signifying that they are transported through the endoplasmic reticulum (ER), and a prodomain that might have a role in targeting from the ER to the FV. Recently, the targeting of two FV-targeted proteases has been studied in detail and they have been shown to traffic to the FV by two different routes. COMICR 378
2 2 Host microbe interactions: parasites Figure 1 Intracellular secretory transport in Plasmodium falciparum. P. falciparum (light grey) resides in the PV (white) and is separated from the red blood cell cytoplasm (pink) by the PVM. The parasite ER (red) resembles a simple perinuclear structure (nucleus in blue) with so-called horns protruding from each side. Most secreted proteins require a signal peptide (SP) in order to enter the ER [32 ]. The Golgi complex (yellow) is less elaborate than in model systems and seems to be in a simple unstacked conformation [33 34,35 ]. The apicoplast (light green) has four membranes, and nuclearencoded proteins require a SP and transit peptide (TP) for correct targeting to the apicoplast [8]. Protein traffic to the apicoplast occurs through the secretory pathway but it is likely that they do not pass through the Golgi [13,14 ]. Haemoglobin is endocytosed by the cytostome (Cy) and trafficked to the FV (aqua) in vesicles (orange) [10]. FV haemoglobin proteases traffic through the secretory pathway and there is evidence for direct transport to the cytostome (orange vesicles) and through the PV (yellow vesicles) [6,7 ]. Three apical organelles involved in red blood cell invasion are also synthesised de novo by the secretory pathway. Very little is known about targeting to the rhoptries (Rh; grey), micronemes (Mn; dark green) and dense granules (DG; light green) in P. falciparum. In T. gondii targeting of transmembrane rhoptry proteins involves a tyrosine-based sorting motif (YXXf) in the cytoplasmic tail, through pre-rhoptry compartments (PRh) [27] and this might be true for some P. falciparum proteins. Targeting of T. gondii transmembrane microneme proteins requires tyrosine-based signals and an acidic patch [22], but this is not absolutely required for P. falciparum [23]. Dense granules are the default destination for secretory proteins in T. gondii [30], however, this cannot be the case for P. falciparum as these organelles are not synthesised until late in the intraerythrocytic cycle. It is presumed that vesicular trafficking delivers proteins to the dense granules (grey vesicles). The first was a study of the FV-targeted protease plasmepsin II, an aspartic protease involved in the initial steps of haemoglobin degradation [7 ]. It was shown that plasmepsin II travels through the ER, as an integral membrane protein, and is initially delivered to the cytostome by an uncharacterised mechanism, probably involving vesicular shuttling, before subsequent localisation to the FV. Upon localisation of plasmepsin II to the FV membrane, the proregion, which contains the only transmembrane domain in the protein, is removed by an uncharacterised protease, termed the PM convertase, releasing mature, soluble plasmepsin II into the FV [7 ]. More recently, trafficking of another FV protease, dipeptide aminopeptidase 1 (DPAP1) has been shown to use a different pathway to plasmepsin II [6]. DPAP1 does not traffic directly to the cytostome, as is the case for plasmepsin II. Instead DPAP1 accumulates in the PV before moving to the FV [6]. Like plasmepsin II, DPAP1 also has a signal sequence, but it does not have a transmembrane domain. DPAP1 is also processed differently to plasmepsin II, by excision of an internal domain postulated to be between the so-called exclusion domain and the two catalytic domains [6]. It therefore appears that plasmepsin II and DPAP1 take different routes to reach the FV and, given that they have differences in their prodomain structure and solubility during trafficking, it is possible that the prodomain itself is responsible for the targeting route of proteins to the FV (Figure 1). Because reporter proteins with a signal peptide are secreted into the PV and are also subsequently found in the FV, there appears to be no additional sequence information necessary for FV-targeting [8,9]. It will be interesting to determine the role of the prodomain in FV proteases, and perhaps it specifies another route of targeting or increases the efficiency of targeting to the FV. Digestion of haemoglobin by the parasite first requires the engulfment of portions of host cytoplasm across the PV and the plasma membrane (PM) and into the FV (Figure 1). This process has been morphologically characterised and involves a specialised endocytic-like
3 Protein targeting to destinations of the secretory pathway in the malaria parasite Plasmodium falciparum Tonkin et al. 3 Figure 2 Targeting proteins into and through the red blood cell. There are two routes for red blood cell (pink) targeted proteins (a e) and (1 6). (a) Soluble host targeted proteins contain an N terminal signal peptide for translocation into the ER (red) and transport to the PV [9]. (b and c) Host-targeted proteins probably associate with chaperones to maintain a translocatable conformation, whereas the PEXEL/VTS sequence mediates transport across the PVM [1,2 ]. The putative PEXEL/VTS translocator is presumably within the PVM, however it has not yet been established where PEXEL/VTS recognition takes place. (d) Upon translocation into the red blood cell cytosol proteins that are destined for the underside of the red blood cell membrane (i.e. KAHRP [knob associated histidine-rich protein] and PfEMP3 [P. falciparum erythrocyte membrane protein 3]) associate with parasite-derived membrane-bound structures called Maurer s clefts [9,37]. The Maurer s cleft binding region (or MCBR) of KAHRP and PfEMP3 are unrelated to one another but both mediate transitory binding to the Maurer s clefts by interacting with a membrane-bound protein such as (e)(i) PfEMP1 (see below) or (e)(ii) an unknown receptor. It is not known how proteins move from the Maurer s clefts to the underside of the red blood cell. (1) The variant surface protein PfEMP1 does not contain a typical signal peptide and it is unclear how this protein is exported to the host cell. PfEMP1 contains a C terminal transmembrane domain that probably acts as a stop transfer signal for entry into the ER (1a). Transport of PfEMP1 presumably occurs as a transmembrane protein through the secretory pathway [38], although others have demonstrated that PfEMP1 is initially transported as a soluble protein (1b) [39]. (2 3) Recent work shows Maurer s clefts originate from the PVM and it likely that PfEMP1 is packaged into Maurer s clefts as they are forming (3 enlarged) [40]. (4) PfEMP1 then traffics to the red blood cell membrane through Maurer s clefts with its C terminal tail exposed to the red blood cell cytoplasm [38]. A multiple spanning membrane protein SBP1 (spectrin binding protein 1) is necessary for transport of PfEMP1 from the Maurer s clefts to the red cell surface but not other host-targeted parasite proteins [3]. It is possible that SBP1 acts as a ligand for correct PfEMP1 deposition within the Maurer s cleft or that it acts as a receptor for the PfEMP1 PEXEL or another unidentified sequence (3-englarged). (5 6) It is unknown how PfEMP1 gets from the Maurer s clefts to the surface of the red blood cell. organelle, referred to as the cytostome (Figure 1) [10]. The molecular events mediating this process, however, are completely unknown. The P. falciparum genome encodes a suite of homologues that are involved in endocytosis in mammalian and yeast, such as the Rabs, putative SNARE-like molecules and clathrin coat components. Experimental evidence is required to show that these homologues are involved in the acquisition of haemoglobin from the host cell (Figure 1) [11]. Protein trafficking to the apicoplast P. falciparum contains a relict plastid called the apicoplast that is essential for parasite survival [12]. The apicoplast is surrounded by four membranes and most apicoplast proteins are nuclear-encoded and require a two-part N terminal extension made up of a signal peptide and a transit peptide (Figure 1) [8]. Earlier work has shown that the signal peptide resembles a classical eukaryotic signal peptide and mediates targeting into the ER
4 4 Host microbe interactions: parasites Figure 3 A model for apicoplast targeting. (a) ER vesicles arrive at the apicoplast surface carrying a transit peptide-bearing protein. The positively charged transit peptide is held in unfolded conformation by the Hsp70 chaperone, BiP. (b) Transit peptide bearing proteins might be brought to the surface by an electrophoretic attraction to a negatively charged translocation complex, which is analogous to the Toc/Tic translocation machinery of plant plastids. (c) The positive charge of apicoplast transit peptides is further electrophoretically attracted into the reducing apicoplast lumen. (d,e) Hsp93 (ClpC) might also attach to the transit peptide and contribute to the movement of the protein through the membranes as a ratchet that only allows anterograde transport. (f) Once inside the apicoplast, the transit peptide is removed by the stromal processing peptidase (SPP). (g) The attached protein is folded into an active conformation with help from Cpn60/Cpn10 and the transit peptide is degraded into its amino acid constituents by an unknown protease. whereas the transit peptide mediates all remaining steps (Figure 1)[8]. Recently, two studies have shed light on the pathway apicoplast proteins take through the secretory system [13,14 ]. It has been shown that is signal peptides from other secreted proteins can mediate apicoplast targeting in combination with a transit peptide, therefore demonstrating that no specific targeting information is found in an apicoplast signal peptide [13 ]. It has also been shown that the P. falciparum apicoplast has a close association with parasites ER [13 ] and that apicoplast-targeting is Brefeldin-A (BFA)-insensitive [13,14 ]. Furthermore, an apicoplast-targeted protein with an incorporated ER retention signal that is recognised by ERD2 in the cis-golgi is still targeted to the apicoplast [13,14 ]; however, when the transit peptide is mutated or deleted, the ER retrieval signal takes over signifying a dominance of transit peptide function over cis-golgi retrieval of the ER retention signal [13 ]. Overall, these studies strongly suggest that apicoplast targeted proteins do not traverse the Golgi but rather divert straight from the ER to the apicoplast, in a similar way to some other organisms with multimembrane-bound plastids (Figure 1) [14,15]. After entry into the secretory pathway, the transit peptide mediates the remaining steps to the apicoplast. Apicoplast transit-peptides, like plant chloroplast transitpeptides, have no consensus sequence, are variable in length and are not predicted to have any secondary structure. Transit peptides are identifiable by a predominance of hydrophilic and basic amino acids, and a corresponding lack of acidic residues [16]. Recently, it was shown that plastid transit-peptides from a wide range of organisms share these common features, although it appears that biases in codon usage influence the actual amino acids used in transit peptides. P. falciparum, for instance, is highly AT-rich and therefore uses lysine and asparagine, which can be both coded for by AT-only codons, in its apicoplast transit peptides, whereas other organisms with more balanced codon usages use a wider range of amino acids with equivalent characteristics [16]. Overall, it appears that transit peptides have relatively loose structural constraints, depending primarily on their overall average composition for their function. What features of apicoplast transit-peptides are physiologically important? To test whether positive charge is essential for transit-peptide function, a series of point mutations were created to perturb the most N terminal charge in a P. falciparum transit peptide [17]. By individually substituting the first two N terminal lysine residues with alanine, glutamic acid or aspartic acid it was demonstrated that the positive charge of the N terminal
5 Protein targeting to destinations of the secretory pathway in the malaria parasite Plasmodium falciparum Tonkin et al. 5 is crucial for transit-peptide fidelity. More so, apicoplast targeting was altered more dramatically when the first positive charge was changed when compared to when the second was changed [17], signifying its greater importance in transit-peptide function. Hsp70 chaperones also appear to be important for import of mitochondrial and plastid proteins by unfolding and folding the proteins before and after membrane translocation, respectively. The apicoplast transit peptide contains a region that is predicted to be suitable for Hsp70 chaperone binding, and this putative binding site was crucial for apicoplast transit-peptide function in vivo [17]. A physical interaction between an apicoplast transit peptide and Hsp70 has also recently been demonstrated [18]. Overall, it seems apparent that transit peptides are very simple and confer their information through a simple charge profile, and possibly an Hsp70 chaperone binding site. The identification of important characteristics of apicoplast transit peptides suggests a model of transit-peptide recognition and apicoplast protein import (Figure 3). After entry into the ER, BiP (the resident ER-Hsp70) binds the transit peptide to keep it in an unfolded state. The transit peptide is then be recognised and translocated through the apicoplast membranes by an as yet undefined protein complex that is analogous (or even homologous) to the Toc/Tic (translocon of the outer/inner envelope membrane of chloroplasts) machinery in plants (Figure 3) [19]. Perhaps, a negative-charged patch on a docking protein permits initial interaction with the transit peptide and the reducing environment created by the metabolic processes of the apicoplast provides an electrostatic force that pulls the transit peptide bearing protein through the membranes in a similar manner to the way proteins are translocated into the mitochondria (Figure 3) [20]. Additional mechanical force required for protein import could be provided by other resident chaperones (ClpC) in the apicoplast. The protein could then be folded and the transit peptide removed (Figure 3). Trafficking to the apical organelles Approximately 35 hours after invasion, when most of the haemoglobin has been consumed, the parasite initiates simultaneous synthesis of up to 32 daughter merozoites, each of which are capable of invading a new red blood cell. As part of merozoite formation, the apical organelles consisting of the rhoptries, micronemes and dense granules are formed de novo by the secretory pathway (Figure 1). Micronemes are storage organelles for adhesive proteins and release their contents onto the parasite surface upon host cell recognition (Figure 1) [21]. In T. gondii, transmembrane micronemal proteins use a tyrosine-based sorting motif and an acidic patch of residues in the C terminal tail [22]. These features are also found on several P. falciparum micronemal proteins. However, in the case of the micronemal protein erythrocyte binding antigen 175 (EBA175), it is clear that neither the tyrosinebased motif nor the acidic residues are required for micronemal targeting [23]. Instead, an unknown signal within the cysteine-rich region directs EBA175 to the micronemes and then unknown features located within the cytoplasmic tail enable EBA175 to function as an invasion ligand [23]. Targeting of soluble proteins to the micronemes in T. gondii relies on their interaction with escorter transmembrane molecules, but no such process has been described for any soluble micronemal protein in P. falciparum [24]. Rhoptries are large club-shaped organelles that are found at the apex of the polarized merozoite, and these organelles contribute to the invasion process as well as to the synthesis of the PV (Figure 1). Rhoptry-targeting is BFAsensitive, indicating that rhoptry proteins pass through the ER and Golgi [25,26]. Rhoptry protein targeting in T. gondii relies on a tyrosine-based motif, YXXf (where X is any amino acid and f is a bulky hydrophobic residue), similar to that found in endosomal-targeted proteins in mammalian and yeast cells [27]. Nothing is known of the requirements for rhoptry targeting in P. falciparum, although many of these proteins do contain a C terminal tyrosine motif. Rhoptry-targeting in T. gondii also relies on the packaging of transmembrane proteins into clathrincoated vesicles by an interaction of the C terminal tyrosine-based motif with the m1 subunit of the AP-1 (adaptor protein complex 1) clathrin-coat complex [28]. All homologous components that are necessary for this targeting mechanism are present in P. falciparum, but whether they function in rhoptry targeting is unknown [11]. Soluble rhoptry proteins do not contain a tyrosinebased motif and probably rely on interaction with membrane-anchored rhoptry-targeted proteins in order to be correctly located, in a similar mechanism to that used by soluble micronemal proteins. The soluble P. falciparum rhoptry-associated protein 2 (RAP2), for instance, relies on an interaction with RAP1 for correct targeting [29]. In turn RAP1 might be bound to RAMA (rhoptry-associated membrane antigen), which is a GPI (glycosyl-phosphatidylinositol) protein located in the rhoptries. How such an entourage of proteins is directed to the rhoptries remains unknown. The third set of apical organelles are the dense granules, which probably expel their contents into the PV upon entry into the host cell (Figure 1). All proteins destined for the PV in T. gondii transit through the dense granules [30]. However, in P. falciparum there are many secreted proteins targeted to PV and red blood cells that are expressed even before dense granules are synthesised, implying that there is another route in malaria parasites. The only known requirement for dense granule targeting
6 6 Host microbe interactions: parasites in P. falciparum is timing of expression. Ring infected erythrocyte antigen (RESA) is normally packaged into dense granules late in the cell cycle and correct targeting was only observed under control of an endogenous promoter, but not under a heterologous promoter [31]. The molecular organization of the secretory pathway Little is known about the molecular organization of the secretory pathway of P. falciparum. The ER resembles a simple perinuclear structure with two protruding horns [32 ], whereas the Golgi is rudimentary with a so-called unstacked conformation (Figure 1) [33 34,35 ]. Like all eukaryotic cells, P. falciparum must rely on a huge supply of molecular machinery to shuffle proteins through membranes and in and out of vesicles in order to target proteins to their correct destination [36]. The genome of P. falciparum has many homologues of core secretory proteins such as the Rabs, the ARFs, the COP components and the Sec apparatus [11], but there is very little experimental evidence for the specific role of any of these components. Protein targeting in P. falciparum has been generally understudied and there are many intriguing questions that remain unanswered. Whereas some protein signals have been identified for localisation to organelles such as the apicoplast and export into the host cell, the molecular machinery for these and other membrane translocation events is unknown. The availability of the genome sequence has been important in revealing the conserved trafficking machinery but has not enabled the identification of components for movement of proteins to some of the locations unique to this parasite and its intracellular and host cell compartments. The molecular machinery for targeting to these compartments provides a fascinating avenue for future investigation and potentially might be a useful target for design of novel therapeutics to control this devastating disease of humans. References and recommended reading Papers of particular interest, published within the annual period of review, have been highlighted as: 1. of special interest of outstanding interest Marti M, Good RT, Rug M, Knuepfer E, Cowman AF: Targeting malaria virulence and remodeling proteins to the host erythrocyte. Science 2004, 306: This study identifies a sequence required for export of proteins across the parasitophorous vacuole membrane, which has allowed the identification of over 400 exported proteins. 2. Hiller NL, Bhattacharjee S, van Ooij C, Liolios K, Harrison T, Lopez- Estrano C, Haldar K: A host-targeting signal in virulence proteins reveals a secretome in malarial infection. Science 2004, 306: This study simultaneously identified the sequence required for export to the PVM. 3. Cooke BM, Buckingham DW, Glenister FK, Fernandez KM, Bannister LH, Marti M, Mohandas N, Coppel RL: A Maurer s cleftassociated protein is essential for expression of the major malaria virulence antigen on the surface of infected red blood cells. J Cell Biol 2006, 172: This is the first protein identified to play a role in trafficking of the important virulence protein PfEMP1 to the erythrocyte surface. 4. Marti M, Baum J, Rug M, Tilley L, Cowman AF: Signal-mediated export of proteins from the malaria parasite to the host erythrocyte. J Cell Biol 2005, 171: Lingelbach K, Przyborski JM: The long and winding road: protein trafficking mechanisms in the Plasmodium falciparum infected erythrocyte. Mol Biochem Parasitol 2006, 147: Klemba M, Gluzman I, Goldberg DE: A Plasmodium falciparum dipeptidyl aminopeptidase I participates in vacuolar hemoglobin degradation. J Biol Chem 2004, 279: Klemba M, Beatty W, Gluzman I, Goldberg DE: Trafficking of plasmepsin II to the food vacuole of the malaria parasite Plasmodium falciparum. J Cell Biol 2004, 164: The authors elucidate the trafficking pathway of a FV protease. 8. Waller RF, Reed MB, Cowman AF, McFadden GI: Protein trafficking to the plastid of Plasmodium falciparum is via the secretory pathway. EMBO J 2000, 19: Wickham ME, Rug M, Ralph SA, Klonis N, McFadden GI, Tilley L, Cowman AF: Trafficking and assembly of the cytoadherence complex in Plasmodium falciparum-infected human erythrocytes. EMBO J 2001, 20: Aikawa M, Huff CG, Spinz H: Comparative feeding mechanisms of avian and primate malarial parasites. Mil Med 1966, 131: Gardner MJ, Hall N, Fung E, White O, Berriman M, Hyman RW, Carlton JM, Pain A, Nelson KE, Bowman S et al.: Genome sequence of the human malaria parasite Plasmodium falciparum. Nature 2002, 419: He CY, Shaw MK, Pletcher CH, Striepen B, Tilney LG, Roos DS: A plastid segregation defect in the protozoan parasite Toxoplasma gondii. EMBO J 2001, 20: Tonkin CJ, Struck NS, Mullin KA, Stimmler LM, McFadden GI: Evidence for Golgi-independent transport from the early secretory pathway to the plastid in malaria parasites. Mol Microbiol /j x. Shows that apicoplast-targeted proteins take the default secretory pathway, that the apicoplast shares a close association with the ER and that apicoplast proteins most probably do not pass through the Golgi. 14. DeRocher A, Gilbert B, Feagin JE, Parsons M: Dissection of brefeldin A-sensitive and -insensitive steps in apicoplast protein targeting. J Cell Sci 2005, 118: The authors suggest that apicoplast proteins do not pass through the Golgi apparatus. 15. van Dooren GG, Schwartzbach SD, Osafune T, McFadden GI: Translocation of proteins across the multiple membranes of complex plastids. Biochim Biophys Acta 2001, 1541: Ralph SA, Foth BJ, Hall N, McFadden GI: Evolutionary pressures on apicoplast transit peptides. Mol Biol Evol 2004, 21: Foth BJ, Ralph SA, Tonkin CJ, Struck NS, Fraunholz M, Roos DS, Cowman AF, McFadden GI: Dissecting apicoplast targeting in the malaria parasite Plasmodium falciparum. Science 2003, 299: Yung SC, Unnasch TR, Lang-Unnasch N: Cis and trans factors involved in apicoplast targeting in Toxoplasma gondii. J Parasitol 2003, 89: Soll J, Schleiff E: Protein import into chloroplasts. Nat Rev Mol Cell Biol 2004, 5: Martin J, Mahlke K, Pfanner N: Role of an energized inner membrane in mitochondrial protein import. Delta psi drives the movement of presequences. J Biol Chem 1991, 266: Carruthers VB, Sibley LD: Sequential protein secretion from three distinct organelles of Toxoplasma gondii accompanies invasion of human fibroblasts. Eur J Cell Biol 1997, 73:
7 Protein targeting to destinations of the secretory pathway in the malaria parasite Plasmodium falciparum Tonkin et al Di Cristina M, Spaccapelo R, Soldati D, Bistoni F, Crisanti A: Two conserved amino acid motifs mediate protein targeting to the micronemes of the apicomplexan parasite Toxoplasma gondii. Mol Cell Biol 2000, 20: Gilberger TW, Thompson JK, Reed MB, Good RT, Cowman AF: The cytoplasmic domain of the Plasmodium falciparum ligand EBA-175 is essential for invasion but not protein trafficking. J Cell Biol 2003, 162: Meissner M, Reiss M, Viebig N, Carruthers VB, Toursel C, Tomavo S, Ajioka JW, Soldati D: A family of transmembrane microneme proteins of Toxoplasma gondii contain EGF-like domains and function as escorters. J Cell Sci 2002, 115: Howard RF, Schmidt CM: The secretory pathway of Plasmodium falciparum regulates transport of p82/rap1 to the rhoptries. Mol Biochem Parasitol 1995, 74: Noe AR, Fishkind DJ, Adams JH: Spatial and temporal dynamics of the secretory pathway during differentiation of the Plasmodium yoelii schizont. Mol Biochem Parasitol 2000, 108: Hoppe HC, Ngo HM, Yang M, Joiner KA: Targeting to rhoptry organelles of Toxoplasma gondii involves evolutionarily conserved mechanisms. Nat Cell Biol 2000, 2: Ngo HM, Yang M, Paprotka K, Pypaert M, Hoppe H, Joiner KA: AP-1 in Toxoplasma gondii mediates biogenesis of the rhoptry secretory organelle from a post-golgi compartment. J Biol Chem 2003, 278: Baldi DL, Andrews KT, Waller RF, Roos DS, Howard RF, Crabb BS, Cowman AF: RAP1 controls rhoptry targeting of RAP2 in the malaria parasite Plasmodium falciparum. EMBO J 2000, 19: Chaturvedi S, Qi H, Coleman D, Rodriguez A, Hanson PI, Striepen B, Roos DS, Joiner KA: Constitutive calciumindependent release of Toxoplasma gondii dense granules occurs through the NSF/SNAP/SNARE/Rab machinery. J Biol Chem 1999, 274: Rug M, Wickham ME, Foley M, Cowman AF, Tilley L: Correct promoter control is needed for trafficking of the ring-infected erythrocyte surface antigen to the host cytosol in transfected malaria parasites. Infect Immun 2004, 72: van Dooren GG, Marti M, Tonkin CJ, Stimmler LM, Cowman AF, McFadden GI: Development of the endoplasmic reticulum, mitochondrion and apicoplast during the asexual life cycle of Plasmodium falciparum. Mol Microbiol 2005, 57: This study characterises the structure of the P. falciparum ER throughout the intraerythrocytic cycle. 33. Elmendorf HG, Haldar K: Identification and localization of ERD2 in the malaria parasite Plasmodium falciparum: separation from sites of sphingomyelin synthesis and implications for organization of the Golgi. EMBO J 1993, 12: Van Wye J, Ghori N, Webster P, Mitschler RR, Elmendorf HG, Haldar K: Identification and localization of Rab6, separation of Rab6 from ERD2 and implications for an unstacked Golgi, in Plasmodium falciparum. Mol Biochem Parasitol 1996, 83: Struck NS, de Souza Dias S, Langer C, Marti M, Pearce JA, Cowman AF, Gilberger TW: Re-defining the Golgi complex in Plasmodium falciparum using the novel Golgi marker PfGRASP. J Cell Sci 2005, 118: This study characterises the structure of the P. falciparum ER throughout the intraerythrocytic cycle. 36. Bock JB, Matern HT, Peden AA, Scheller RH: A genomic perspective on membrane compartment organization. Nature 2001, 409: Knuepfer E, Rug M, Klonis N, Tilley L, Cowman AF: Trafficking determinants for PfEMP3 export and assembly under the Plasmodium falciparum-infected red blood cell membrane. Mol Microbiol 2005, 58: Kriek N, Tilley L, Horrocks P, Pinches R, Elford BC, Ferguson DJ, Lingelbach K, Newbold CI: Characterization of the pathway for transport of the cytoadherence-mediating protein, PfEMP1, to the host cell surface in malaria parasite-infected erythrocytes. Mol Microbiol 2003, 50: Papakrivos J, Newbold CI, Lingelbach K: A potential novel mechanism for the insertion of a membrane protein revealed by a biochemical analysis of the Plasmodium falciparum cytoadherence molecule PfEMP-1. Mol Microbiol 2005, 55: Spycher C, Rug M, Klonis N, Ferguson DJ, Cowman AF, Beck HP, Tilley L: Genesis of and trafficking to the Maurer s clefts of P. falciparum infected erythrocytes. Mol Cell Biol 2006, 26:
Protein Trafficking in the Secretory and Endocytic Pathways
 Protein Trafficking in the Secretory and Endocytic Pathways The compartmentalization of eukaryotic cells has considerable functional advantages for the cell, but requires elaborate mechanisms to ensure
Protein Trafficking in the Secretory and Endocytic Pathways The compartmentalization of eukaryotic cells has considerable functional advantages for the cell, but requires elaborate mechanisms to ensure
Intracellular Compartments and Protein Sorting
 Intracellular Compartments and Protein Sorting Intracellular Compartments A eukaryotic cell is elaborately subdivided into functionally distinct, membrane-enclosed compartments. Each compartment, or organelle,
Intracellular Compartments and Protein Sorting Intracellular Compartments A eukaryotic cell is elaborately subdivided into functionally distinct, membrane-enclosed compartments. Each compartment, or organelle,
endomembrane system internal membranes origins transport of proteins chapter 15 endomembrane system
 endo system chapter 15 internal s endo system functions as a coordinated unit divide cytoplasm into distinct compartments controls exocytosis and endocytosis movement of molecules which cannot pass through
endo system chapter 15 internal s endo system functions as a coordinated unit divide cytoplasm into distinct compartments controls exocytosis and endocytosis movement of molecules which cannot pass through
Molecular Cell Biology Problem Drill 16: Intracellular Compartment and Protein Sorting
 Molecular Cell Biology Problem Drill 16: Intracellular Compartment and Protein Sorting Question No. 1 of 10 Question 1. Which of the following statements about the nucleus is correct? Question #01 A. The
Molecular Cell Biology Problem Drill 16: Intracellular Compartment and Protein Sorting Question No. 1 of 10 Question 1. Which of the following statements about the nucleus is correct? Question #01 A. The
Practice Exam 2 MCBII
 1. Which feature is true for signal sequences and for stop transfer transmembrane domains (4 pts)? A. They are both 20 hydrophobic amino acids long. B. They are both found at the N-terminus of the protein.
1. Which feature is true for signal sequences and for stop transfer transmembrane domains (4 pts)? A. They are both 20 hydrophobic amino acids long. B. They are both found at the N-terminus of the protein.
Vesicle Transport. Vesicle pathway: many compartments, interconnected by trafficking routes 3/17/14
 Vesicle Transport Vesicle Formation Curvature (Self Assembly of Coat complex) Sorting (Sorting Complex formation) Regulation (Sar1/Arf1 GTPases) Fission () Membrane Fusion SNARE combinations Tethers Regulation
Vesicle Transport Vesicle Formation Curvature (Self Assembly of Coat complex) Sorting (Sorting Complex formation) Regulation (Sar1/Arf1 GTPases) Fission () Membrane Fusion SNARE combinations Tethers Regulation
Homework Hanson section MCB Course, Fall 2014
 Homework Hanson section MCB Course, Fall 2014 (1) Antitrypsin, which inhibits certain proteases, is normally secreted into the bloodstream by liver cells. Antitrypsin is absent from the bloodstream of
Homework Hanson section MCB Course, Fall 2014 (1) Antitrypsin, which inhibits certain proteases, is normally secreted into the bloodstream by liver cells. Antitrypsin is absent from the bloodstream of
Summary of Endomembrane-system
 Summary of Endomembrane-system 1. Endomembrane System: The structural and functional relationship organelles including ER,Golgi complex, lysosome, endosomes, secretory vesicles. 2. Membrane-bound structures
Summary of Endomembrane-system 1. Endomembrane System: The structural and functional relationship organelles including ER,Golgi complex, lysosome, endosomes, secretory vesicles. 2. Membrane-bound structures
Protein sorting (endoplasmic reticulum) Dr. Diala Abu-Hsasan School of Medicine
 Protein sorting (endoplasmic reticulum) Dr. Diala Abu-Hsasan School of Medicine dr.abuhassand@gmail.com An overview of cellular components Endoplasmic reticulum (ER) It is a network of membrane-enclosed
Protein sorting (endoplasmic reticulum) Dr. Diala Abu-Hsasan School of Medicine dr.abuhassand@gmail.com An overview of cellular components Endoplasmic reticulum (ER) It is a network of membrane-enclosed
Essential Cell Biology
 Alberts Bray Hopkin Johnson Lewis Raff Roberts Walter Essential Cell Biology FOURTH EDITION Chapter 15 Intracellular Compartments and Protein Transport Copyright Garland Science 2014 CHAPTER CONTENTS MEMBRANE-ENCLOSED
Alberts Bray Hopkin Johnson Lewis Raff Roberts Walter Essential Cell Biology FOURTH EDITION Chapter 15 Intracellular Compartments and Protein Transport Copyright Garland Science 2014 CHAPTER CONTENTS MEMBRANE-ENCLOSED
Molecular Cell Biology - Problem Drill 17: Intracellular Vesicular Traffic
 Molecular Cell Biology - Problem Drill 17: Intracellular Vesicular Traffic Question No. 1 of 10 1. Which of the following statements about clathrin-coated vesicles is correct? Question #1 (A) There are
Molecular Cell Biology - Problem Drill 17: Intracellular Vesicular Traffic Question No. 1 of 10 1. Which of the following statements about clathrin-coated vesicles is correct? Question #1 (A) There are
Signal-mediated export of proteins from the malaria parasite to the host erythrocyte
 Published Online: 21 November, 2005 Supp Info: http://doi.org/10.1083/jcb.200508051 Downloaded from jcb.rupress.org on January 13, 2019 JCB: MINI-REVIEW Signal-mediated export of proteins from the malaria
Published Online: 21 November, 2005 Supp Info: http://doi.org/10.1083/jcb.200508051 Downloaded from jcb.rupress.org on January 13, 2019 JCB: MINI-REVIEW Signal-mediated export of proteins from the malaria
Zool 3200: Cell Biology Exam 4 Part I 2/3/15
 Name: Key Trask Zool 3200: Cell Biology Exam 4 Part I 2/3/15 Answer each of the following questions in the space provided, explaining your answers when asked to do so; circle the correct answer or answers
Name: Key Trask Zool 3200: Cell Biology Exam 4 Part I 2/3/15 Answer each of the following questions in the space provided, explaining your answers when asked to do so; circle the correct answer or answers
17/01/2017. Protein trafficking between cell compartments. Lecture 3: The cytosol. The mitochondrion - the power plant of the cell
 ell biology 2017 version 13/1 2017 ote endosome vs lysosome handout Lecture 3: Text book Alberts et al.: hapter 12-14 (Topics covered by the lecture) A lot of reading! Focus on principles ell Biology interactive
ell biology 2017 version 13/1 2017 ote endosome vs lysosome handout Lecture 3: Text book Alberts et al.: hapter 12-14 (Topics covered by the lecture) A lot of reading! Focus on principles ell Biology interactive
Chapter 13: Vesicular Traffic
 Chapter 13: Vesicular Traffic Know the terminology: ER, Golgi, vesicle, clathrin, COP-I, COP-II, BiP, glycosylation, KDEL, microtubule, SNAREs, dynamin, mannose-6-phosphate, M6P receptor, endocytosis,
Chapter 13: Vesicular Traffic Know the terminology: ER, Golgi, vesicle, clathrin, COP-I, COP-II, BiP, glycosylation, KDEL, microtubule, SNAREs, dynamin, mannose-6-phosphate, M6P receptor, endocytosis,
Intracellular vesicular traffic. B. Balen
 Intracellular vesicular traffic B. Balen Three types of transport in eukaryotic cells Figure 12-6 Molecular Biology of the Cell ( Garland Science 2008) Endoplasmic reticulum in all eucaryotic cells Endoplasmic
Intracellular vesicular traffic B. Balen Three types of transport in eukaryotic cells Figure 12-6 Molecular Biology of the Cell ( Garland Science 2008) Endoplasmic reticulum in all eucaryotic cells Endoplasmic
2013 John Wiley & Sons, Inc. All rights reserved. PROTEIN SORTING. Lecture 10 BIOL 266/ Biology Department Concordia University. Dr. S.
 PROTEIN SORTING Lecture 10 BIOL 266/4 2014-15 Dr. S. Azam Biology Department Concordia University Introduction Membranes divide the cytoplasm of eukaryotic cells into distinct compartments. The endomembrane
PROTEIN SORTING Lecture 10 BIOL 266/4 2014-15 Dr. S. Azam Biology Department Concordia University Introduction Membranes divide the cytoplasm of eukaryotic cells into distinct compartments. The endomembrane
Lecture Readings. Vesicular Trafficking, Secretory Pathway, HIV Assembly and Exit from Cell
 October 26, 2006 1 Vesicular Trafficking, Secretory Pathway, HIV Assembly and Exit from Cell 1. Secretory pathway a. Formation of coated vesicles b. SNAREs and vesicle targeting 2. Membrane fusion a. SNAREs
October 26, 2006 1 Vesicular Trafficking, Secretory Pathway, HIV Assembly and Exit from Cell 1. Secretory pathway a. Formation of coated vesicles b. SNAREs and vesicle targeting 2. Membrane fusion a. SNAREs
The Cell Organelles. Eukaryotic cell. The plasma membrane separates the cell from the environment. Plasma membrane: a cell s boundary
 Eukaryotic cell The Cell Organelles Enclosed by plasma membrane Subdivided into membrane bound compartments - organelles One of the organelles is membrane bound nucleus Cytoplasm contains supporting matrix
Eukaryotic cell The Cell Organelles Enclosed by plasma membrane Subdivided into membrane bound compartments - organelles One of the organelles is membrane bound nucleus Cytoplasm contains supporting matrix
PROTEIN TRAFFICKING. Dr. SARRAY Sameh, Ph.D
 PROTEIN TRAFFICKING Dr. SARRAY Sameh, Ph.D Overview Proteins are synthesized either on free ribosomes or on ribosomes bound to endoplasmic reticulum (RER). The synthesis of nuclear, mitochondrial and peroxisomal
PROTEIN TRAFFICKING Dr. SARRAY Sameh, Ph.D Overview Proteins are synthesized either on free ribosomes or on ribosomes bound to endoplasmic reticulum (RER). The synthesis of nuclear, mitochondrial and peroxisomal
Molecular Trafficking
 SCBM 251 Molecular Trafficking Assoc. Prof. Rutaiwan Tohtong Department of Biochemistry Faculty of Science rutaiwan.toh@mahidol.ac.th Lecture outline 1. What is molecular trafficking? Why is it important?
SCBM 251 Molecular Trafficking Assoc. Prof. Rutaiwan Tohtong Department of Biochemistry Faculty of Science rutaiwan.toh@mahidol.ac.th Lecture outline 1. What is molecular trafficking? Why is it important?
Mechanism of Vesicular Transport
 Mechanism of Vesicular Transport Transport vesicles play a central role in the traffic of molecules between different membrane-enclosed enclosed compartments. The selectivity of such transport is therefore
Mechanism of Vesicular Transport Transport vesicles play a central role in the traffic of molecules between different membrane-enclosed enclosed compartments. The selectivity of such transport is therefore
BIOL 4374/BCHS 4313 Cell Biology Exam #2 March 22, 2001
 BIOL 4374/BCHS 4313 Cell Biology Exam #2 March 22, 2001 SS# Name This exam is worth a total of 100 points. The number of points each question is worth is shown in parentheses. Good luck! 1. (2) In the
BIOL 4374/BCHS 4313 Cell Biology Exam #2 March 22, 2001 SS# Name This exam is worth a total of 100 points. The number of points each question is worth is shown in parentheses. Good luck! 1. (2) In the
Lecture 6 - Intracellular compartments and transport I
 01.25.10 Lecture 6 - Intracellular compartments and transport I Intracellular transport and compartments 1. Protein sorting: How proteins get to their appropriate destinations within the cell 2. Vesicular
01.25.10 Lecture 6 - Intracellular compartments and transport I Intracellular transport and compartments 1. Protein sorting: How proteins get to their appropriate destinations within the cell 2. Vesicular
Intracellular Vesicular Traffic Chapter 13, Alberts et al.
 Intracellular Vesicular Traffic Chapter 13, Alberts et al. The endocytic and biosynthetic-secretory pathways The intracellular compartments of the eucaryotic ell involved in the biosynthetic-secretory
Intracellular Vesicular Traffic Chapter 13, Alberts et al. The endocytic and biosynthetic-secretory pathways The intracellular compartments of the eucaryotic ell involved in the biosynthetic-secretory
Cellular compartments
 Cellular compartments 1. Cellular compartments and their function 2. Evolution of cellular compartments 3. How to make a 3D model of cellular compartment 4. Cell organelles in the fluorescent microscope
Cellular compartments 1. Cellular compartments and their function 2. Evolution of cellular compartments 3. How to make a 3D model of cellular compartment 4. Cell organelles in the fluorescent microscope
TRANSPORT PROCESSES. 1b. moving proteins into membranes and organelles
 1b. moving proteins into membranes and organelles SLIDE 1 A typical mammalian cell contains up to 10,000 different kinds of proteins. The vast majority of these proteins are synthesized by cytosolic ribosomes,
1b. moving proteins into membranes and organelles SLIDE 1 A typical mammalian cell contains up to 10,000 different kinds of proteins. The vast majority of these proteins are synthesized by cytosolic ribosomes,
CELL BIOLOGY - CLUTCH CH INTRACELLULAR PROTEIN TRANSPORT.
 !! www.clutchprep.com CONCEPT: MEMBRANE ENCLOSED ORGANELLES Table of eukaryotic organelles and their functions Organelle Function % volume of cell Cytosol Aqueous fluid where metabolic pathways and chemical
!! www.clutchprep.com CONCEPT: MEMBRANE ENCLOSED ORGANELLES Table of eukaryotic organelles and their functions Organelle Function % volume of cell Cytosol Aqueous fluid where metabolic pathways and chemical
BIOLOGY 111. CHAPTER 3: The Cell: The Fundamental Unit of Life
 BIOLOGY 111 CHAPTER 3: The Cell: The Fundamental Unit of Life The Cell: The Fundamental Unit of Life Learning Outcomes 3.1 Explain the similarities and differences between prokaryotic and eukaryotic cells
BIOLOGY 111 CHAPTER 3: The Cell: The Fundamental Unit of Life The Cell: The Fundamental Unit of Life Learning Outcomes 3.1 Explain the similarities and differences between prokaryotic and eukaryotic cells
Endomembrane system, *Chloroplasts, *Mitochondria. *Learn these from text/connect1. Fertilization of a human cell
 Key Concepts: - Cells are the Basic Unit of Life Cell Theory, Surface to Volume - 2 Cell Types Prokaryotic, Eukaryotic - Cell Membrane Membrane Structure - Cell Organelles Endomembrane system, *Chloroplasts,
Key Concepts: - Cells are the Basic Unit of Life Cell Theory, Surface to Volume - 2 Cell Types Prokaryotic, Eukaryotic - Cell Membrane Membrane Structure - Cell Organelles Endomembrane system, *Chloroplasts,
Cell are made up of organelles. An ORGANELLE is a specialized subunit within a cell that has a specific function.
 Plant and Animal Cells The Cell Theory All living things are made up of one or more cells. All cells come from other cells. Organization of Living Things Cell are made up of organelles. An ORGANELLE is
Plant and Animal Cells The Cell Theory All living things are made up of one or more cells. All cells come from other cells. Organization of Living Things Cell are made up of organelles. An ORGANELLE is
Eukaryotic Cell Structures
 Comparing the Cell to a Factory Eukaryotic Cell Structures Structures within a eukaryotic cell that perform important cellular functions are known as organelles. Cell biologists divide the eukaryotic cell
Comparing the Cell to a Factory Eukaryotic Cell Structures Structures within a eukaryotic cell that perform important cellular functions are known as organelles. Cell biologists divide the eukaryotic cell
A. Major parts 1. Nucleus 2. Cytoplasm a. Contain organelles (see below) 3. Plasma membrane (To be discussed in Cellular Transport Lecture)
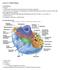 Lecture 5: Cellular Biology I. Cell Theory Concepts: 1. Cells are the functional and structural units of living organisms 2. The activity of an organism is dependent on both the individual and collective
Lecture 5: Cellular Biology I. Cell Theory Concepts: 1. Cells are the functional and structural units of living organisms 2. The activity of an organism is dependent on both the individual and collective
Advanced Cell Biology. Lecture 33
 Advanced Cell Biology. Lecture 33 Alexey Shipunov Minot State University April 22, 2013 Shipunov (MSU) Advanced Cell Biology. Lecture 33 April 22, 2013 1 / 38 Outline Questions and answers Intracellular
Advanced Cell Biology. Lecture 33 Alexey Shipunov Minot State University April 22, 2013 Shipunov (MSU) Advanced Cell Biology. Lecture 33 April 22, 2013 1 / 38 Outline Questions and answers Intracellular
Renata Schipp Medical Biology Department
 Renata Schipp Medical Biology Department Deffinition of cell The cell is the smallest structural and functional unit of all known living organisms The cell was discovered by Robert Hooke in 1665 and also
Renata Schipp Medical Biology Department Deffinition of cell The cell is the smallest structural and functional unit of all known living organisms The cell was discovered by Robert Hooke in 1665 and also
Lysosomes and endocytic pathways 9/27/2012 Phyllis Hanson
 Lysosomes and endocytic pathways 9/27/2012 Phyllis Hanson General principles Properties of lysosomes Delivery of enzymes to lysosomes Endocytic uptake clathrin, others Endocytic pathways recycling vs.
Lysosomes and endocytic pathways 9/27/2012 Phyllis Hanson General principles Properties of lysosomes Delivery of enzymes to lysosomes Endocytic uptake clathrin, others Endocytic pathways recycling vs.
1. endoplasmic reticulum This is the location where N-linked oligosaccharide is initially synthesized and attached to glycoproteins.
 Biology 4410 Name Spring 2006 Exam 2 A. Multiple Choice, 2 pt each Pick the best choice from the list of choices, and write it in the space provided. Some choices may be used more than once, and other
Biology 4410 Name Spring 2006 Exam 2 A. Multiple Choice, 2 pt each Pick the best choice from the list of choices, and write it in the space provided. Some choices may be used more than once, and other
SBI3U7 Cell Structure & Organelles. 2.2 Prokaryotic Cells 2.3 Eukaryotic Cells
 SBI3U7 Cell Structure & Organelles 2.2 Prokaryotic Cells 2.3 Eukaryotic Cells No nucleus Prokaryotic Cells No membrane bound organelles Has a nucleus Eukaryotic Cells Membrane bound organelles Unicellular
SBI3U7 Cell Structure & Organelles 2.2 Prokaryotic Cells 2.3 Eukaryotic Cells No nucleus Prokaryotic Cells No membrane bound organelles Has a nucleus Eukaryotic Cells Membrane bound organelles Unicellular
Endoplasmic Reticulum
 Endoplasmic Reticulum What s ER? How is ER? Why is ER? definition description functions Nissl s bodies neurons Berg s bodies hepatocytes Organelle structure histocytochemical evidences Ergastoplasm pancreatic
Endoplasmic Reticulum What s ER? How is ER? Why is ER? definition description functions Nissl s bodies neurons Berg s bodies hepatocytes Organelle structure histocytochemical evidences Ergastoplasm pancreatic
1. This is the location where N-linked oligosaccharide is initially synthesized and attached to glycoproteins.
 Biology 4410 Name Spring 2006 Exam 2 A. Multiple Choice, 2 pt each Pick the best choice from the list of choices, and write it in the space provided. Some choices may be used more than once, and other
Biology 4410 Name Spring 2006 Exam 2 A. Multiple Choice, 2 pt each Pick the best choice from the list of choices, and write it in the space provided. Some choices may be used more than once, and other
Chapter 7. (7-1 and 7-2) A Tour of the Cell
 Chapter 7 (7-1 and 7-2) A Tour of the Cell Microscopes as Windows to the World of Cells Cells were first described in 1665 by Robert Hooke. By the mid-1800s, the accumulation of scientific evidence led
Chapter 7 (7-1 and 7-2) A Tour of the Cell Microscopes as Windows to the World of Cells Cells were first described in 1665 by Robert Hooke. By the mid-1800s, the accumulation of scientific evidence led
The most valuable lipid ever?
 The most valuable lipid ever? Spermaceti whale oil Used by sperm whales in a special organ in the huge head cavity Largely comprised of cetyl palmitate ww.thisrecording.com www.greenpeace.org Peer Instruction
The most valuable lipid ever? Spermaceti whale oil Used by sperm whales in a special organ in the huge head cavity Largely comprised of cetyl palmitate ww.thisrecording.com www.greenpeace.org Peer Instruction
Cellular control of cholesterol. Peter Takizawa Department of Cell Biology
 Cellular control of cholesterol Peter Takizawa Department of Cell Biology Brief overview of cholesterol s biological role Regulation of cholesterol synthesis Dietary and cellular uptake of cholesterol
Cellular control of cholesterol Peter Takizawa Department of Cell Biology Brief overview of cholesterol s biological role Regulation of cholesterol synthesis Dietary and cellular uptake of cholesterol
Introduction. Biochemistry: It is the chemistry of living things (matters).
 Introduction Biochemistry: It is the chemistry of living things (matters). Biochemistry provides fundamental understanding of the molecular basis for the function and malfunction of living things. Biochemistry
Introduction Biochemistry: It is the chemistry of living things (matters). Biochemistry provides fundamental understanding of the molecular basis for the function and malfunction of living things. Biochemistry
I. Fluid Mosaic Model A. Biological membranes are lipid bilayers with associated proteins
 Lecture 6: Membranes and Cell Transport Biological Membranes I. Fluid Mosaic Model A. Biological membranes are lipid bilayers with associated proteins 1. Characteristics a. Phospholipids form bilayers
Lecture 6: Membranes and Cell Transport Biological Membranes I. Fluid Mosaic Model A. Biological membranes are lipid bilayers with associated proteins 1. Characteristics a. Phospholipids form bilayers
CELL PART OF THE DAY. Chapter 7: Cell Structure and Function
 CELL PART OF THE DAY Chapter 7: Cell Structure and Function Cell Membrane Cell membranes are composed of two phospholipid layers. Cell membrane is flexible, not rigid The cell membrane has two major functions.
CELL PART OF THE DAY Chapter 7: Cell Structure and Function Cell Membrane Cell membranes are composed of two phospholipid layers. Cell membrane is flexible, not rigid The cell membrane has two major functions.
1. to understand how proteins find their destination in prokaryotic and eukaryotic cells 2. to know how proteins are bio-recycled
 Protein Targeting Objectives 1. to understand how proteins find their destination in prokaryotic and eukaryotic cells 2. to know how proteins are bio-recycled As a protein is being synthesized, decisions
Protein Targeting Objectives 1. to understand how proteins find their destination in prokaryotic and eukaryotic cells 2. to know how proteins are bio-recycled As a protein is being synthesized, decisions
Chapter 6. Antigen Presentation to T lymphocytes
 Chapter 6 Antigen Presentation to T lymphocytes Generation of T-cell Receptor Ligands T cells only recognize Ags displayed on cell surfaces These Ags may be derived from pathogens that replicate within
Chapter 6 Antigen Presentation to T lymphocytes Generation of T-cell Receptor Ligands T cells only recognize Ags displayed on cell surfaces These Ags may be derived from pathogens that replicate within
By: Brooke Sheppard
 By: Brooke Sheppard What is a Cell? Cells are the basic structure of life for all organisms. Cells are microscopic, which means we can only view cells under a microscope. There are animal cells and plant
By: Brooke Sheppard What is a Cell? Cells are the basic structure of life for all organisms. Cells are microscopic, which means we can only view cells under a microscope. There are animal cells and plant
Organelles of the Cell & How They Work Together. Packet #7
 Organelles of the Cell & How They Work Together Packet #7 Introduction Introduction Organization of cells is basically similar in all cells. Additionally, most cells are tiny Ranging from 1 1000 cubic
Organelles of the Cell & How They Work Together Packet #7 Introduction Introduction Organization of cells is basically similar in all cells. Additionally, most cells are tiny Ranging from 1 1000 cubic
Cell Quality Control. Peter Takizawa Department of Cell Biology
 Cell Quality Control Peter Takizawa Department of Cell Biology Cellular quality control reduces production of defective proteins. Cells have many quality control systems to ensure that cell does not build
Cell Quality Control Peter Takizawa Department of Cell Biology Cellular quality control reduces production of defective proteins. Cells have many quality control systems to ensure that cell does not build
Chapter 1: Vesicular traffic. Biochimica cellulare parte B 2017/18
 Chapter 1: Vesicular traffic Biochimica cellulare parte B 2017/18 Major Protein-sorting pathways in eukaryotic cells Secretory and endocytic pathways Unifying principle governs all protein trafficking
Chapter 1: Vesicular traffic Biochimica cellulare parte B 2017/18 Major Protein-sorting pathways in eukaryotic cells Secretory and endocytic pathways Unifying principle governs all protein trafficking
Organelles of the Cell & How They Work Together. Packet #5
 Organelles of the Cell & How They Work Together Packet #5 Developed by Mr. Barrow 2018 1 Introduction Organization of cells is basically similar in all cells. Additionally, most cells are tiny Ranging
Organelles of the Cell & How They Work Together Packet #5 Developed by Mr. Barrow 2018 1 Introduction Organization of cells is basically similar in all cells. Additionally, most cells are tiny Ranging
Key Concept B F. How do peptides get loaded onto the proper kind of MHC molecule?
 Location of MHC class I pockets termed B and F that bind P and P9 amino acid side chains of the peptide Different MHC alleles confer different functional properties on the adaptive immune system by specifying
Location of MHC class I pockets termed B and F that bind P and P9 amino acid side chains of the peptide Different MHC alleles confer different functional properties on the adaptive immune system by specifying
B F. Location of MHC class I pockets termed B and F that bind P2 and P9 amino acid side chains of the peptide
 Different MHC alleles confer different functional properties on the adaptive immune system by specifying molecules that have different peptide binding abilities Location of MHC class I pockets termed B
Different MHC alleles confer different functional properties on the adaptive immune system by specifying molecules that have different peptide binding abilities Location of MHC class I pockets termed B
Lecture 6 9/17 Dr. Hirsh Organization of Cells, continued
 Cell structure of Eukaryotic cells Lecture 6 9/17 Dr. Hirsh Organization of Cells, continued Lots of double-membraned organelles Existence of an Endo-membrane system separation of areas of cell, transport
Cell structure of Eukaryotic cells Lecture 6 9/17 Dr. Hirsh Organization of Cells, continued Lots of double-membraned organelles Existence of an Endo-membrane system separation of areas of cell, transport
4/12/17. Cells. Cell Structure. Ch. 2 Cell Structure and Func.on. Range of Cell Sizes BIOL 100
 Ch. 2 Cell Structure and Func.on BIOL 100 Cells Fundamental units of life Cell theory All living things are composed of one or more cells. The cell is the most basic unit of life. All cells come from pre-existing
Ch. 2 Cell Structure and Func.on BIOL 100 Cells Fundamental units of life Cell theory All living things are composed of one or more cells. The cell is the most basic unit of life. All cells come from pre-existing
Endocytosis and Intracellular Trafficking of Notch and Its Ligands
 CHA P T E R F IVE Endocytosis and Intracellular Trafficking of Notch and Its Ligands Shinya Yamamoto, *,1 Wu-Lin Charng, *,1 and Hugo J. Bellen *,,, Contents 1. Notch Signaling and its Regulation by Endocytosis
CHA P T E R F IVE Endocytosis and Intracellular Trafficking of Notch and Its Ligands Shinya Yamamoto, *,1 Wu-Lin Charng, *,1 and Hugo J. Bellen *,,, Contents 1. Notch Signaling and its Regulation by Endocytosis
Export of Plasmodium Proteins via a Novel Secretory Pathway
 Export of Plasmodium Proteins via a Novel Secretory Pathway The intraerythrocytic location of the malaria parasite necessitates modiþcation of the host cell. These alterations are mediated either directly
Export of Plasmodium Proteins via a Novel Secretory Pathway The intraerythrocytic location of the malaria parasite necessitates modiþcation of the host cell. These alterations are mediated either directly
Zool 3200: Cell Biology Exam 4 Part I 2/3/15
 Name: Trask Zool 3200: Cell Biology Exam 4 Part I 2/3/15 Answer each of the following questions in the space provided, explaining your answers when asked to do so; circle the correct answer or answers
Name: Trask Zool 3200: Cell Biology Exam 4 Part I 2/3/15 Answer each of the following questions in the space provided, explaining your answers when asked to do so; circle the correct answer or answers
Organelles of the Cell & How They Work Together. Packet #5
 Organelles of the Cell & How They Work Together Packet #5 Developed by Mr. Barrow 2018 1 Introduction Organization of cells is basically similar in all cells. Additionally, most cells are tiny Ranging
Organelles of the Cell & How They Work Together Packet #5 Developed by Mr. Barrow 2018 1 Introduction Organization of cells is basically similar in all cells. Additionally, most cells are tiny Ranging
Chapter 1 Plasma membranes
 1 of 5 TEXTBOOK ANSWERS Chapter 1 Plasma membranes Recap 1.1 1 The plasma membrane: keeps internal contents of the cell confined to one area keeps out foreign molecules that damage or destroy the cell
1 of 5 TEXTBOOK ANSWERS Chapter 1 Plasma membranes Recap 1.1 1 The plasma membrane: keeps internal contents of the cell confined to one area keeps out foreign molecules that damage or destroy the cell
BIO 5099: Molecular Biology for Computer Scientists (et al)
 BIO 5099: Molecular Biology for Computer Scientists (et al) Lecture 15: Being a Eukaryote: From DNA to Protein, A Tour of the Eukaryotic Cell. Christiaan van Woudenberg Being A Eukaryote Basic eukaryotes
BIO 5099: Molecular Biology for Computer Scientists (et al) Lecture 15: Being a Eukaryote: From DNA to Protein, A Tour of the Eukaryotic Cell. Christiaan van Woudenberg Being A Eukaryote Basic eukaryotes
Chapters 2 and 3. Pages and Pages Prayer Attendance Homework
 Chapters 2 and 3 Pages 44-45 and Pages 59-62 Prayer Attendance Homework The Cell The cell is the basic unit of life on Earth, separated from its environment by a membrane and sometimes an outer wall. Prokaryotic
Chapters 2 and 3 Pages 44-45 and Pages 59-62 Prayer Attendance Homework The Cell The cell is the basic unit of life on Earth, separated from its environment by a membrane and sometimes an outer wall. Prokaryotic
General information. Cell mediated immunity. 455 LSA, Tuesday 11 to noon. Anytime after class.
 General information Cell mediated immunity 455 LSA, Tuesday 11 to noon Anytime after class T-cell precursors Thymus Naive T-cells (CD8 or CD4) email: lcoscoy@berkeley.edu edu Use MCB150 as subject line
General information Cell mediated immunity 455 LSA, Tuesday 11 to noon Anytime after class T-cell precursors Thymus Naive T-cells (CD8 or CD4) email: lcoscoy@berkeley.edu edu Use MCB150 as subject line
AP Biology
 Tour of the Cell (1) 2007-2008 Types of cells Prokaryote bacteria cells - no organelles - organelles Eukaryote animal cells Eukaryote plant cells Cell Size Why organelles? Specialized structures - specialized
Tour of the Cell (1) 2007-2008 Types of cells Prokaryote bacteria cells - no organelles - organelles Eukaryote animal cells Eukaryote plant cells Cell Size Why organelles? Specialized structures - specialized
MCB130 Midterm. GSI s Name:
 1. Peroxisomes are small, membrane-enclosed organelles that function in the degradation of fatty acids and in the degradation of H 2 O 2. Peroxisomes are not part of the secretory pathway and peroxisomal
1. Peroxisomes are small, membrane-enclosed organelles that function in the degradation of fatty acids and in the degradation of H 2 O 2. Peroxisomes are not part of the secretory pathway and peroxisomal
The endoplasmic reticulum is a network of folded membranes that form channels through the cytoplasm and sacs called cisternae.
 Endoplasmic reticulum (ER) The endoplasmic reticulum is a network of folded membranes that form channels through the cytoplasm and sacs called cisternae. Cisternae serve as channels for the transport of
Endoplasmic reticulum (ER) The endoplasmic reticulum is a network of folded membranes that form channels through the cytoplasm and sacs called cisternae. Cisternae serve as channels for the transport of
Biology 12 Cell Structure and Function. Typical Animal Cell
 Biology 12 Cell Structure and Function Typical Animal Cell Vacuoles: storage of materials and water Golgi body: a series of stacked disk shaped sacs. Repackaging centre stores, modifies, and packages proteins
Biology 12 Cell Structure and Function Typical Animal Cell Vacuoles: storage of materials and water Golgi body: a series of stacked disk shaped sacs. Repackaging centre stores, modifies, and packages proteins
A Tour of the Cell Lecture 2, Part 1 Fall 2008
 Cell Theory 1 A Tour of the Cell Lecture 2, Part 1 Fall 2008 Cells are the basic unit of structure and function The lowest level of structure that can perform all activities required for life Reproduction
Cell Theory 1 A Tour of the Cell Lecture 2, Part 1 Fall 2008 Cells are the basic unit of structure and function The lowest level of structure that can perform all activities required for life Reproduction
Protein Targeting to the Malaria Parasite Plastid
 Traffic 2008; 9: 166 175 Blackwell Munksgaard Review # 2007 The Authors Journal compilation # 2007 Blackwell Publishing Ltd doi: 10.1111/j.1600-0854.2007.00660.x Protein Targeting to the Malaria Parasite
Traffic 2008; 9: 166 175 Blackwell Munksgaard Review # 2007 The Authors Journal compilation # 2007 Blackwell Publishing Ltd doi: 10.1111/j.1600-0854.2007.00660.x Protein Targeting to the Malaria Parasite
MeBOP Module 2 Introduction
 MeBOP Module 2 Introduction Karine Frenal: The expert of Toxo glideosome and IMC Damien Jacot: Expert in about everything Sunil Kumar Dogga: TgAsp3 Budhaditya Mukherjee: Biochemistry Apicomplexans are
MeBOP Module 2 Introduction Karine Frenal: The expert of Toxo glideosome and IMC Damien Jacot: Expert in about everything Sunil Kumar Dogga: TgAsp3 Budhaditya Mukherjee: Biochemistry Apicomplexans are
Cell Overview. Hanan Jafar BDS.MSc.PhD
 Cell Overview Hanan Jafar BDS.MSc.PhD THE CELL is made of: 1- Nucleus 2- Cell Membrane 3- Cytoplasm THE CELL Formed of: 1. Nuclear envelope 2. Chromatin 3. Nucleolus 4. Nucleoplasm (nuclear matrix) NUCLEUS
Cell Overview Hanan Jafar BDS.MSc.PhD THE CELL is made of: 1- Nucleus 2- Cell Membrane 3- Cytoplasm THE CELL Formed of: 1. Nuclear envelope 2. Chromatin 3. Nucleolus 4. Nucleoplasm (nuclear matrix) NUCLEUS
Structure & Function of Cells
 Anatomy & Physiology 101-805 Unit 4 Structure & Function of Cells Paul Anderson 2011 Anatomy of a Generalised Cell Attached or bound ribosomes Cilia Cytosol Centriole Mitochondrion Rough endoplasmic reticulum
Anatomy & Physiology 101-805 Unit 4 Structure & Function of Cells Paul Anderson 2011 Anatomy of a Generalised Cell Attached or bound ribosomes Cilia Cytosol Centriole Mitochondrion Rough endoplasmic reticulum
Cytosol the fluid Cytoplasm cell interior, everything outside the nucleus but within the cell membrane, includes the organelles, cytosol, and
 Cell Organelles Plasma Membrane comprised of a phospholipid bilayer and embedded proteins Outer surface has oligosaccharides separates the cells s contents from its surroundings Cytosol the fluid Cytoplasm
Cell Organelles Plasma Membrane comprised of a phospholipid bilayer and embedded proteins Outer surface has oligosaccharides separates the cells s contents from its surroundings Cytosol the fluid Cytoplasm
10/5/2015. Cell Size. Relative Rate of Reaction
 The Cell Biology 102 Fundamental unit of life Smallest unit that displays all the basic elements of life Lecture 5: Cells Cell Theory 1. All living things are made of one or more cells Cell Theory 2. The
The Cell Biology 102 Fundamental unit of life Smallest unit that displays all the basic elements of life Lecture 5: Cells Cell Theory 1. All living things are made of one or more cells Cell Theory 2. The
Lysosomes, Peroxisomes and Centrioles. Hüseyin Çağsın
 Lysosomes, Peroxisomes and Centrioles Hüseyin Çağsın Lysosomes Outline Endosomes Molecule transport to the lysosomes Endocytosis Exocytosis Autophagy Vacuoles Peroxisomes Centrioles Lysosomes Lysosomes
Lysosomes, Peroxisomes and Centrioles Hüseyin Çağsın Lysosomes Outline Endosomes Molecule transport to the lysosomes Endocytosis Exocytosis Autophagy Vacuoles Peroxisomes Centrioles Lysosomes Lysosomes
Chapter 4: Cell Structure and Function
 Chapter 4: Cell Structure and Function Robert Hooke Fig. 4-2, p.51 The Cell Smallest unit of life Can survive on its own or has potential to do so Is highly organized for metabolism Senses and responds
Chapter 4: Cell Structure and Function Robert Hooke Fig. 4-2, p.51 The Cell Smallest unit of life Can survive on its own or has potential to do so Is highly organized for metabolism Senses and responds
LIFE IS CELLULAR. Cell Theory. Cells Are Small. Prokaryotic Cell 10/4/15. Chapter 7 Cell Structure and Function
 Chapter 7 Cell Structure and Function The cell basic unit of life, all living things are made of a cell (unicellular) or more than one cell (multicellular). LIFE IS CELLULAR The invention of the microscope
Chapter 7 Cell Structure and Function The cell basic unit of life, all living things are made of a cell (unicellular) or more than one cell (multicellular). LIFE IS CELLULAR The invention of the microscope
October 26, Lecture Readings. Vesicular Trafficking, Secretory Pathway, HIV Assembly and Exit from Cell
 October 26, 2006 Vesicular Trafficking, Secretory Pathway, HIV Assembly and Exit from Cell 1. Secretory pathway a. Formation of coated vesicles b. SNAREs and vesicle targeting 2. Membrane fusion a. SNAREs
October 26, 2006 Vesicular Trafficking, Secretory Pathway, HIV Assembly and Exit from Cell 1. Secretory pathway a. Formation of coated vesicles b. SNAREs and vesicle targeting 2. Membrane fusion a. SNAREs
CHAPTER 4 - CELLS. All living things are made up of one or more cells. A cell is the smallest unit that can carry on all of the processes of life.
 CHAPTER 4 - CELLS Objectives Name the scientists who first observed living and nonliving cells. Summarize the research that led to the development of the cell theory. State the three principles of the
CHAPTER 4 - CELLS Objectives Name the scientists who first observed living and nonliving cells. Summarize the research that led to the development of the cell theory. State the three principles of the
Organelles Found in a Generalized Animal Cell
 Organelles Found in a Generalized Animal Cell 1. Cell Membrane 2. Cytoplasm 3. Nucleus 4. Nuclear Membrane 5. Nucleoplasm 6. Nucleolus 7. Chromosomes 8. Vacuole 9. Ribosomes 10. Rough Endoplasmic Reticulum
Organelles Found in a Generalized Animal Cell 1. Cell Membrane 2. Cytoplasm 3. Nucleus 4. Nuclear Membrane 5. Nucleoplasm 6. Nucleolus 7. Chromosomes 8. Vacuole 9. Ribosomes 10. Rough Endoplasmic Reticulum
Don t Freak Out. Test on cell organelle on Friday!
 Cell Structure 1 Don t Freak Out Test on cell organelle on Friday! This test should be a buffer test and help raise your overall test score. All information will come from this week! 2 Cells Provide Compartments
Cell Structure 1 Don t Freak Out Test on cell organelle on Friday! This test should be a buffer test and help raise your overall test score. All information will come from this week! 2 Cells Provide Compartments
Questions in Cell Biology
 Name: Questions in Cell Biology Directions: The following questions are taken from previous IB Final Papers on the subject of cell biology. Answer all questions. This will serve as a study guide for the
Name: Questions in Cell Biology Directions: The following questions are taken from previous IB Final Papers on the subject of cell biology. Answer all questions. This will serve as a study guide for the
Basic Structure of a Cell
 Basic Structure of a Cell 1 Introduction to Cells Cells are the basic units of organisms Cells can only be observed under microscope Basic types of cells: Animal Cell Plant Cell Bacterial Cell 2 Number
Basic Structure of a Cell 1 Introduction to Cells Cells are the basic units of organisms Cells can only be observed under microscope Basic types of cells: Animal Cell Plant Cell Bacterial Cell 2 Number
First to View Cells. copyright cmassengale
 CELL THEORY All living things are made of cells Cells are the basic unit of structure and function in an organism (basic unit of life) Cells come from the reproduction of existing cells (cell division)
CELL THEORY All living things are made of cells Cells are the basic unit of structure and function in an organism (basic unit of life) Cells come from the reproduction of existing cells (cell division)
Lesson Overview. 7.2 Cell Structure
 7.2 Cell Organization The eukaryotic cell can be divided into two major parts: the nucleus and the cytoplasm. The cytoplasm is the fluid portion of the cell outside the nucleus. Prokaryotes do not have
7.2 Cell Organization The eukaryotic cell can be divided into two major parts: the nucleus and the cytoplasm. The cytoplasm is the fluid portion of the cell outside the nucleus. Prokaryotes do not have
A Tour of the Cell. Chapter 6. Biology Eighth Edition Neil Campbell and Jane Reece. PowerPoint Lecture Presentations for
 Chapter 6 A Tour of the Cell PowerPoint Lecture Presentations for Biology Eighth Edition Neil Campbell and Jane Reece Lectures by Chris Romero, updated by Erin Barley with contributions from Joan Sharp
Chapter 6 A Tour of the Cell PowerPoint Lecture Presentations for Biology Eighth Edition Neil Campbell and Jane Reece Lectures by Chris Romero, updated by Erin Barley with contributions from Joan Sharp
The Cell. Biology 105 Lecture 4 Reading: Chapter 3 (pages 47 62)
 The Cell Biology 105 Lecture 4 Reading: Chapter 3 (pages 47 62) Outline I. Prokaryotic vs. Eukaryotic II. Eukaryotic A. Plasma membrane transport across B. Main features of animal cells and their functions
The Cell Biology 105 Lecture 4 Reading: Chapter 3 (pages 47 62) Outline I. Prokaryotic vs. Eukaryotic II. Eukaryotic A. Plasma membrane transport across B. Main features of animal cells and their functions
Endomembrane system 11/1/2018. Endomembrane System. Direct physical continuity. Transfer of membrane segments as vesicles. Outer Nuclear envelope
 Endomembrane system Endomembrane System Outer Nuclear envelope Direct physical continuity Transfer of membrane segments as vesicles Endoplasmic reticulum BUT membranes are not identical in structure and
Endomembrane system Endomembrane System Outer Nuclear envelope Direct physical continuity Transfer of membrane segments as vesicles Endoplasmic reticulum BUT membranes are not identical in structure and
Chapter 3. Chapter 3. Bellringer. Objectives. Chapter 3. Chapter 3. Vocabulary. Cell Wall. Identify the different parts of a eukaryotic cell.
 Bellringer Objectives Identify the different parts of a eukaryotic cell. Explain the function of each part of a eukaryotic cell. Plant cells and animal cells have many cell parts in common. But they also
Bellringer Objectives Identify the different parts of a eukaryotic cell. Explain the function of each part of a eukaryotic cell. Plant cells and animal cells have many cell parts in common. But they also
(Please activate your clickers)
 Organelles in prokaryotes and eukaryotes! Plasma membrane! Cell wall (in prokaryotes, plants, fungi, some protists)! Ribosomes! Nucleoid/nucleus! Mitochondria! Plastids (Please activate your clickers)
Organelles in prokaryotes and eukaryotes! Plasma membrane! Cell wall (in prokaryotes, plants, fungi, some protists)! Ribosomes! Nucleoid/nucleus! Mitochondria! Plastids (Please activate your clickers)
Posttranslational Modification and Targeting of Proteins
 Posttranslational Modification and Targeting of Proteins Graduate Biochemistry Term 2/2016 Assist. Prof. Dr. Panida Khunkaewla School of Chemistry, Institute of Science Suranaree University of Technology
Posttranslational Modification and Targeting of Proteins Graduate Biochemistry Term 2/2016 Assist. Prof. Dr. Panida Khunkaewla School of Chemistry, Institute of Science Suranaree University of Technology
Cell Biology. A discipline of biology: 1. Cell structure 2. Cellular processes 3. Cell division
 The Cell Cell Biology 1 A discipline of biology: 1. Cell structure 2. Cellular processes 3. Cell division Tight connection with 1. Molecular biology 2. Biochemistry Cell theory 2 1838, 1839 Theodor Schwann
The Cell Cell Biology 1 A discipline of biology: 1. Cell structure 2. Cellular processes 3. Cell division Tight connection with 1. Molecular biology 2. Biochemistry Cell theory 2 1838, 1839 Theodor Schwann
The Fundamental Unit of Life. Intext Exercise 1
 Intext Exercise 1 Who discovered cells and how? Cells were discovered in 1665 by an English Botanist, Robert Hooke. He used a primitive microscope to observe cells in a cork slice. Why is the cell called
Intext Exercise 1 Who discovered cells and how? Cells were discovered in 1665 by an English Botanist, Robert Hooke. He used a primitive microscope to observe cells in a cork slice. Why is the cell called
Cell Structure & Function. Source:
 Cell Structure & Function Source: http://koning.ecsu.ctstateu.edu/cell/cell.html Definition of Cell A cell is the smallest unit that is capable of performing life functions. http://web.jjay.cuny.edu/~acarpi/nsc/images/cell.gif
Cell Structure & Function Source: http://koning.ecsu.ctstateu.edu/cell/cell.html Definition of Cell A cell is the smallest unit that is capable of performing life functions. http://web.jjay.cuny.edu/~acarpi/nsc/images/cell.gif
BIO 5099: Molecular Biology for Computer Scientists (et al) Lecture 15: Being A Eukaryote. Eukaryotic Cells. Basic eukaryotes have:
 BIO 5099: Molecular Biology for Computer Scientists (et al) Lecture 15: Being a Eukaryote: From DNA to Protein, A Tour of the Eukaryotic Cell. Christiaan van Woudenberg Being A Eukaryote Basic eukaryotes
BIO 5099: Molecular Biology for Computer Scientists (et al) Lecture 15: Being a Eukaryote: From DNA to Protein, A Tour of the Eukaryotic Cell. Christiaan van Woudenberg Being A Eukaryote Basic eukaryotes
Module 3 Lecture 7 Endocytosis and Exocytosis
 Module 3 Lecture 7 Endocytosis and Exocytosis Endocytosis: Endocytosis is the process by which cells absorb larger molecules and particles from the surrounding by engulfing them. It is used by most of
Module 3 Lecture 7 Endocytosis and Exocytosis Endocytosis: Endocytosis is the process by which cells absorb larger molecules and particles from the surrounding by engulfing them. It is used by most of
Cell Structure and Function
 Cell Structure and Function Many Scientists Contributed to the Cell Theory! Hooke discovered cells while looking at cork under the microscope! Leewenhoek was the first to observe bacteria! Schleiden discovered
Cell Structure and Function Many Scientists Contributed to the Cell Theory! Hooke discovered cells while looking at cork under the microscope! Leewenhoek was the first to observe bacteria! Schleiden discovered
Eukaryotic cells contain organelles that allow the specializations and the separation of functions within the cell.
 Section 3: Eukaryotic cells contain organelles that allow the specializations and the separation of functions within the cell. K What I Know W What I Want to Find Out L What I Learned Essential Questions
Section 3: Eukaryotic cells contain organelles that allow the specializations and the separation of functions within the cell. K What I Know W What I Want to Find Out L What I Learned Essential Questions
