For administration by health care professionals. To be administered under a physicianâ s supervision.
|
|
|
- Laurence Fitzgerald
- 5 years ago
- Views:
Transcription
1 PRANACTINCITRIC urea c13 powder, for solution Otsuka America Pharmaceutical Introduction and Test Instructions Note: This â Introduction and Test Instructionsâ contains information for: Test Collection Kit Breath Test Instrument BreathTekâ UBT for H. pylori 1. UBiT IR 300 Infrared Spectrophotometer 2. POConeâ Infrared Spectrophotometer I. Intended Use The BreathTekâ UBT Collection Kit is intended for use in the qualitative detection of urease associated with Helicobacter pylori in the human stomach and as an aid in the initial diagnosis and posttreatment monitoring of Helicobacter pylori infection in adult patients. The test may be used for monitoring treatment if used at least four (4) weeks following completion of therapy. For these purposes, the system utilizes an Infrared Spectrophotometer for the measurement of the ratio of 13CO to 12CO in breath samples. 2 For administration by health care professionals. To be administered under a physicianâ s supervision. II. Summary and Explanation Since the isolation of the spiral ureaseproducing Helicobacter pylori bacteria (H. pylori) in 1983 by Drs. Marshall and Warren 1, a significant body of evidence has accumulated indicating that the bacteria is an important pathogen in the upper GI tract of humans. 2.3 The causal relationship between H. pylori and chronic active gastritis, duodenal ulcer, and gastric ulcer is well documented. 4.5 Methods available for detecting current infection of the human stomach by H. pylori are generally divided into two (2) general types: Invasive and Noninvasive. Invasive methods are so named because they include, as a first step, an esophagogastroduodenoscopy (â EGDâ ) with collection of gastric biopsies. These biopsies are then examined by one or more detection methods: histological examination of stained tissue, microbiological culture of the organism, or direct detection of urease activity in the tissue (for example, the CLOtest ). Biopsy based methods are expensive, entail some patient risk and discomfort and may give false negative results due to sampling errors when colonization of the gastric mucosa is patchy. 6 The noninvasive, nonradioactive method for detecting current H. pylori infection is based on the BreathTekâ UBT which is described in the next section. Several serological tests that detect serum antibodies to H. pylori are commercially available. A positive result with these tests cannot distinguish between current infection and past exposure to infection and, therefore, is not a conclusive indicator of current gastrointestinal colonization by H. pylori. III. Principle of the BreathTekâ UBT for H. pylori A. Description of the Pranactin Citric Diagnostic Drug Component The diagnostic drug component of the kit is 13Curea, a synthetic urea contained in a granulated powder (PranactinCitric) for reconstitution with potable water to provide a clear solution for oral administration. The carbon in the drug component is predominantly Carbon13, a stable, naturally occurring, nonradioactive isotope of carbon; the relative abundance of Carbon13 is greater than or equal to 99%. Each three (3) gram dose of Pranactin Citric is supplied in a polyethylenelined foil pouch and contains 75 mg of 13Curea, citric acid 7, aspartame and mannitol. 13Curea is the diamide of 13Ccarbonic acid and is highly soluble in water (1 gram per ml at 25Â C). It has the following chemical formula: 13CH N O. An average adult body normally contains about /14 2
2 grams of urea, which is a product of protein metabolism. Urea in the body is referred to as natural isotopic abundance urea since it is composed of 98.9% 12Curea and 1.1% 13Curea. B. Principle of the Test Pranactin Citric drug product is a component of the BreathTekâ UBT for H. pylori kit. Three (3) g of reconstituted Pranactin Citric containing 75 mg of 13Curea is ingested by the patient. In the presence of urease associated with gastric H. pylori, 13Curea is decomposed to 13CO 2 and NH4 + according to the following equation: (NH 2) 2 13 CO + H2O + 2H HP Urease CO + 2NH Curea â 13 The CO 2, is absorbed in the blood, then exhaled in the breath. This results in an increase in the ratio of 13CO to 12CO in a POSTDOSE breath sample taken after the Pranactin 2 2 Citric solution was consumed, compared to a BASELINE sample taken before the Pranactin Citric solution was consumed. Analysis of the breath samples is performed by UBiT IR300 Infrared Spectrophotometer or POConeâ Infrared Spectrophotometer [located at your testing laboratory, physician office or hospital]. The BreathTekâ UBT can detect very low levels of H. pylori colonization and, by assessing the entire gastric mucosa, avoids the risk of sampling errors inherent in biopsy based methods. In the absence of gastric H. pylori, the 13Curea does not produce 13CO 2 in the stomach. The ratio of 13CO in the POSTDOSE breath sample remains essentially the same as the BASELINE. 2 IV. Warnings and Precautions A. For in vitro diagnostic use only. The Pranactin Citric drug solution is taken orally as part of the diagnostic procedure. B. Phenylketonurics: Contains Phenylalanine (one of the protein components of Aspartame), 84 mg per dosage unit. (For reference, 12 ounces of typical diet cola soft drinks contain approximately 80 mg of Phenylalanine.) C. A negative result does not rule out the possibility of Helicobacter pylori infection. False negative results do occur with this procedure. If clinical signs are suggestive of H. pylori infection, retest with a new sample or an alternate method. D. Antimicrobials, proton pump inhibitors, and bismuth preparations are known to suppress H. pylori. Ingestion of these within two (2) weeks prior to performing the BreathTekâ UBT may give false negative results. E. A false positive test may occur due to urease associated with other gastric spiral organisms observed in humans such as Helicobacter heilmannii. F. Premature POSTDOSE breath collection time can lead to a false negative diagnosis for a patient with a marginally positive BreathTekâ UBT result. G. A false positive test could occur in patients who have achlorhydria. 8 H. If particulate matter is visible in the reconstituted Pranactin Citric solution after thorough mixing, the solution should not be used. V. Shelf Life and Storage The BreathTekâ UBT Collection Kit should be stored at 15Â 30Â C (59Â 86Â F). Pranactin has an expiration date. Do not use beyond the expiration date stated on the label. VI. Patient Preparation Citric 2/14
3 A. Remind the patient that Pranactin Citric contains phenylalanine (one of the protein components of Aspartame). Phenylketonurics restrict dietary phenylalanine. B. The patient should have fasted at least one (1) hour before administering the BreathTekâ UBT. C. The patient should not have taken antimicrobials, proton pump inhibitors, or bismuth preparations within two (2) weeks prior to administering the BreathTekâ UBT. VII. Procedure for Collecting Breath Samples Using BreathTekâ UBT Kit, for Analysis by Infrared Spectrophotometer A. Materials 1. Materials provided Each sealed singlepatient BreathTekâ UBT Collection Kit contains: One (1) plastic kit tray containing One (1) â How Toâ guide Test instructions One (1) pouch of Pranactin Citric powder (3 g) A set of four (4) selfadhesive barcode stickers. All barcodes should bear the same number. Two (2) breath collection bags, one (1) blue bag for the BASELINE sample and one (1) pink bag for the POSTDOSE sample. One (1) sample transport bag One (1) plastic straw One (1) plastic drinking cup 2. Materials needed but not provided A timer capable of timing an interval up to fifteen (15) minutes. Scissors for opening the Pranactin Citric pouch. Note: An Infrared Spectrophotometer (UBiT is required for analysis of breath samples. IR3000 or POConeâ, Otsuka Pharmaceutical Co., Ltd.) B. StepByStep Procedure Time intervals listed in the following stepbystep procedure are critical. They are highlighted by the timer icon: 1. Verify that the patient has been prepared for the test as specified in Section VI. 2. Open the BreathTekâ UBT Collection Kit, which should contain all the materials listed in Step VII. Slide out the kit tray. Label each breath collection bag to maintain patient identification using the barcode labels provided, or according to your laboratory or office procedure. 3. Collect the BASELINE breath sample according to the following procedure: a. Remove the blue breath collection bag from the kit tray. b. Remove the pulloff cap from the mouthpiece of the breath collection bag. c. Instruct the patient to: (1) breathe normally; (2) take a deep breath then pause momentarily; (3) exhale into the mouthpiece of the bag. d. Replace the cap firmly until it clicks on the mouthpiece of the bag. 4. Prepare the Pranactin Citric solution no more than sixty (60) minutes before administering it to the patient. Urea slowly decomposes in water. a. Remove the Pranactin Citric pouch from the kit tray. Tap the upright packet of Pranactin Citric to settle the contents in the bottom half. b. With clean scissors, cut off the top of the packet and carefully empty the contents into the drinking cup provided, making sure to transfer all of the contents by tapping on the bottom of the pouch. c. Add potable water to the fill line indicated on the outside of the cup by a raised 3/14
4 tapping on the bottom of the pouch. c. Add potable water to the fill line indicated on the outside of the cup by a raised plastic ridge. d. Replace the lid securely and swirl the mixture for up to two (2) minutes to dissolve the packet contents; typically, only one (1) minute is required for complete dissolution. The resulting solution should be clear with no particulate matter. If particulate matter is present after thorough mixing, the solution should not be used. 5. Instruct the patient to drink all of the solution with the straw provided, without stopping. Advise the patient NOT to â rinseâ the inside of his/her mouth with the solution before swallowing. Discard the straw. 6. Set the timer for fifteen (15) minutes. The patient should sit quietly and should not eat, drink or smoke during the fifteen (15) minute interval. 7. After fifteen (15) minutes have elapsed, remove the pink breath collection bag from the kit tray. Collect the POSTDOSE breath sample according to the procedure described in Steps VII B.3.b through B.3.d. 8. Store the specimens at 15Â 30Â C (59Â 86Â F) until analysis is performed. 9. Perform breath sample analysis within seven (7) days of breath sample collection. If desired, use the plastic sample transport bag for transport of the breath samples. 8/24/ VIII. Quality Control Complete operating information, including selfdiagnostic instrument routines and user maintenance procedures, is provided in the Instruction Manuals for the UBiT IR300 Spectrophotometer, the UBiT AS10 Autosampler and the POConeâ Spectrophotometer, respectively. Additionally, each office laboratory or test facility should follow its own internal procedures for quality control. IX. Test Results A. The Test Method The ratio of 13CO to 12 2 CO 2 in breath samples is determined by Infrared Spectrophotometer, (i.e., UBiT IR300 or POConeâ ). B. Calculation of Results The result of the BreathTekâ UBT is provided as the Delta Over Baseline. No calculations are required by the user. Delta Over Baseline is the difference between the ratio ( 13CO 2 / 12 CO 2) in the POSTDOSE sample and the corresponding ratio in the BASELINE sample. C. Determination of the Cutoff Point The cutoff point is the level of BreathTekâ UBT result used to discriminate between H. pylori infected and uninfected individuals. For the BreathTekâ UBT, the Delta Over Baseline cutoff point was determined to be 2.4 in a controlled study of twentysix (26) infected, and twentythree (23) uninfected adult volunteers. Test subjects were judged to be in acceptable health based on the results of a medical history and physical examination and demonstrated no uncontrolled clinically significant abnormality other than, for some, symptoms of peptic ulcer. The previous version of the Meretek urea breath test, the Meretek UBT was used as the reference standard. The cutoff point was calculated by determining the BreathTekâ UBT result level at which negative and positive subjects were best distinguished by cooptimization of relative sensitivity and specificity. The 2.4 cutoff point for the BreathTekâ UBT was verified in an independent study by retrospective analysis of Clinical Field Trial data collected on 145 H. pylori negative and 105 H. pylori positive test subjects, again using the original Meretek UBT as reference. Asymptomatic subjects and those with dyspepsia were included in the validation study. Figure 1a depicts graphically the BreathTekâ UBT Delta Over Baseline cutoff point which distinguishes H. pylori positive and negative subjects. For the Meretek UBT Breath Test, the Delta Over Baseline cutoff point was determined to be 2.4 in a controlled study of sixtysix (66) infected and fiftythree (53) uninfected asymptomatic, apparently healthy volunteers. Histological examination of biopsy 4/14
5 tissue was used as the reference standard. The cutoff point was evaluated by determining the Meretek UBT Breath Test result level at which histologically negative and positive subjects were best distinguished. Figure 1b graphically depicts the Meretek UBT Breath Test Delta Over Baseline cutoff point which distinguishes histologically positive and negative subjects. Note that in Figures 1a and 1b, the Delta Over Baseline scales are logarithmic. D. Interpretation of Results For the BreathTekâ UBT, a result greater than or equal to 2.4 Delta Over Baseline is interpreted as diagnostically positive indicating the presence of urease associated with H. pylori. A BreathTekâ UBT result less than 2.4 Delta Over Baseline is interpreted as diagnostically negative indicating the absence of urease associated with H. pylori. The 2.4 Delta Over Baseline cutoff point applies to both initial diagnosis and posttreatment monitoring of H. pylori infection. X. Limitations of the Test A. The BreathTekâ UBT should not be used until four (4) weeks or more after the end of treatment for the eradication of H. pylori as earlier posttreatment assessment may give false negative results. B. The performance characteristics for persons under the age of eighteen (18) have not been established for this test. C. The specimen integrity of breath samples and reference gases stored in breath bags under ambient conditions has not been determined beyond seven (7) days. D. A correlation between the number of H. pylori organisms in the stomach and the BreathTekâ UBT result has not been established. E. The predicate device (Meretek UBT ) was standardized in asymptomatic healthy volunteers and subsequently validated in clinical trials limited to patients with documented duodenal ulcer disease. 5/14
6 XI. Expected Values Delta Over Baseline values for the BreathTekâ UBT were determined in a controlled clinical study of twentysix (26) infected and twentythree (23) uninfected adult volunteers. The Meretek UBT Breath Test was used as the reference method in the diagnosis of infection. The range of BreathTekâ UBT Delta Over Baseline values for the uninfected group was determined to be 0.0 to 1.0. A histogram for the distribution of Delta Over Baseline values from the uninfected subjects is shown in Figure 2a. Values for the Meretek UBT Breath Test were determined in a controlled clinical study of sixtysix (66) infected and fiftythree (53) uninfected asymptomatic, apparently healthy volunteers. Histological examination of biopsy tissue was used as the reference method in the determination of infection in this study. The range of Meretek UBT Delta Over Baseline values for the uninfected group was determined to be 0.0 to 2.2. A histogram for the distribution of Delta Over Baseline values from the uninfected subjects is shown in Figure 2b. XII. Performance Characteristics A. Performance Characteristics for the UBiT IR300 Spectrophotometer. Refer to the Instruction Manual for the instrument. B. Performance Characteristics for the POConeâ Spectrophotometer. Refer to the Instruction Manual for the instrument. C. Method Comparisons in Clinical Trials 1. Comparison of the BreathTekâ UBT with the Meretek UBT a. Experimental Design The method comparison data presented here were collected from a prospective, crossover clinical field trial designed to validate the BreathTekâ UBT test procedure and to examine the effect of pretest fasting time on test performance. The study included two hundred fiftytwo (252) adult test subjects from Houston and Galveston, Texas. Subjects were judged to be in acceptable health based on the results of a medical history and physical examination and demonstrated no uncontrolled clinically significant abnormality other than, for some, symptoms of dyspepsia. Test subjects were tested for H. pylori infection using the Meretek UBT Breath Test according to established procedure and with the BreathTekâ UBT under differing conditions of pretest fasting times. Otherwise, no special instructions were given to subjects beyond those listed in the stepbystep procedures for administration of the Meretek UBT and BreathTekâ UBT. To minimize potential bias due to test order, the sequence of urea breath tests administered to each subject was randomized. All breath tests were administered to a given individual within fourteen (14) days of one 6/14
7 another, most often and at a minimum, on successive days. b. Results It was demonstrated in the field trial that the BreathTekâ UBT may be administered at any time beyond one (1) hour after consuming solid and/or liquid food. Method comparison results are presented in a twoway contingency table on the following page (Table 1). Point estimates of Percent Agreement of the BreathTekâ UBT with Meretek UBT positive and negative results are listed in the contingency table (Table 1). The comparative method for determining the true diagnosis was the predicate device (Meretek UBT ) rather than endoscopic methods. The exact binomial distribution was used to calculate the lower and upper limits of the 95% confidence intervals of the performance statistics. The confidence intervals are entered in parentheses following the point estimate of the statistic. Table 1. Comparison of BreathTekâ UBT (â 1hour fast) with Meretek UBT BreathTekâ UBT Results Meretek UBT Positive Negative Total Positive Negative Total Percent Agreement with Meretek UBT positive subjects: 99.1 % [95% CI: (94.9, )] Percent Agreement with Meretek UBT negative subjects: 99.3% [95% CI: (96.2, 100.0)] 2. Comparison of Gas Isotope Ratio Mass Spectrometry (GIRMS) and UBiT IR300 Infrared Spectrophotometry Method A multicenter prospective clinical trial was conducted to compare the UBiT IR300 method with the traditional GIRMS method. The study included a total of three hundred twenty (320) adult test subjects enrolled at four (4) physicansâ office laboratory (POL) settings and at a clinical laboratory. The results of the clinical trial are provided in the Instructional Manual for the UBiT IR300 Infrared Spectrophotometer (refer to the Application Note, 13CUrea Breath Test using the UBiT IR300 Infrared Spectrophotometry System). Table 2 shows the percent agreement of the UBiT IR300 results as compared to the GIRMS method. Overall agreement was excellent at percent. Table 2. Agreement of UBiT IR300 and GIRMS for 13C urea breath test GIRMS Results UBiT IR 300 Results Positive Negative Total Positive /14
8 Negative Total %Overall Agreement: 99.06% [95% CI: (97.35, 99.74)] %Positive Agreement: 98.29% [95% CI: (94.26, 99.70)] %Negative Agreement: 99.51% [95% CI: (97.49, 99.97)] 3. Comparison of UBiT IR300 and POConeâ Infrared Spectrophotometry Methods A multicenter, prospective study was conducted to compare the POConeâ Infrared Spectrophotometer to the UBiT IR300 Infrared Spectrophotometer for measuring 13CO 2 enrichment in breath. The study included a total of two hundred twenty (220) adult test subjects enrolled at five (5) physiciansâ office laboratory (POL) and point of care (POC) settings. The results of the clinical trial are provided in the Instruction Manual for the POConeâ Infrared Spectrophotometer (refer to the Application Note, 13CUrea Breath Test using the POConeâ Infrared Spectrophotometry System). Table 3 shows the percent agreement of the POConeâ results with the UBiT results. Overall agreement was percent IR300 Table 3. Agreement of POConeâ and UBiT IR300 for the 13C urea breath test UBiT IR 300 Results POConeâ Results Positive Negative Total Positive Negative Total %Overall Agreement: 99.55% [95% CI: (97.67, 99.98)] %Positive Agreement: % [95% CI: (95.90, )] %Negative Agreement: 99.25% [95% CI: (96.27, 99.96)] 4. Comparison of Meretek UBT with Endoscopic Methods a. Experimental Design The method comparison data presented here were collected from two (2) independent double blind clinical field trials which involved treatment of H. pylori infection. The studies included four hundred ninetynine (499) adult patients with duodenal ulcer disease at seventyfive (75) clinical sites in the United States. Patients were tested for H. pylori infection initially (using histopathology, microbiological culture, CLOtest, and the Meretek UBT ), and at various posttreatment intervals through out the study (using histopathology, microbiological culture, and the Meretek UBT ). In these clinical trials, patients were treated with various combinations of clarithromycin, omeprazole and placebo. Note, however, that there is no evidence that differing treatment regimens affect the performance of the Meretek UBT. 1) Histopathology Biopsy specimens, fixed with 10% buffered formalin were cut into 4mm sections, stained with Genta stain and examined by an experienced pathologist. 8/14
9 2) 3) 4) Biopsy specimens, fixed with 10% buffered formalin were cut into 4mm sections, stained with Genta stain and examined by an experienced pathologist. Microbiologic culture Culture was performed using fresh bloodbased media, both selective and nonselective, at 37Â C in 12% CO2 in air with 98% humidity. H. pylori were identified by Gram stain, typical colony morphology, and biochemical properties (production of oxidase, catalase and urease). CLOtest (Delta West, Limited, Bently, West Australia) A biopsy specimen was tested for urease activity with the CLOtest according to the instructions in its package insert. The Meretek UBT Breath Test for H. pylori The diagnostic Meretek UBT Breath Test was performed in accordance with procedures described in its package insert. b. Results Method comparison results are presented in twoway contingency tables. In Tables 4, 5, and 6, the Meretek UBT Breath Test results are compared with the CLOtest, histology, and with the combined endoscopic method results (CLOtest, histology, and culture) for the initial patient visit 9. In Table 7, the Meretek UBT Breath Test results are compared with the combined endoscopic method results (histology and culture) for the posttreatment visits which occurred four (4) weeks or more after end of treatment. The exact binomial distribution was used to calculate the lower and upper limits of the 95% confidence intervals of the performance statistics. The confidence intervals are entered in parentheses following the point estimate of the statistic. D. Performance Characteristics For Initial Diagnosis Table 4. Comparison with CLOtest for Initial Visit Meretekâ UBT Results CLOtest Results Positive Negative Total Positive Negative Total Relative Sensitivity: 92.8% [95% CI: (90, 95)] Relative Specificity: 94.1% [95% CI: (71, l00)] Table 5. Comparison with Histology for Initial Visit Meretekâ UBT Results Histology Positive Negative Total Positive Negative Total Sensitivity: 95.2% [95% CI: (93, 97)] Relative Specificity: 90.0% [95% CI: (74, 98)] Table 6. Comparison with Combined Endoscopic 9/14
10 Methods for Initial Visit Combined endoscopic methods used were CLOtest, histology, and culture per DAIDP guidelines 8 for pretreatment diagnosis. Meretekâ UBT Results Endoscopy Positive Negative Total Positive Negative Total Sensitivity: 95.2% [95 % CI: (93, 97)] Specificity: 89.7% [95% CI: (73, 98)] E. Performance Characteristics for PostTreatment Monitoring Table 7. Comparison with Combined Endoscopic Methods* for PostTreatment Visits (four weeks or more after End of Treatment (EOT)) Meretek UBT Breath Test Results 1 Month EOT 3 Months EOT 6 Months EOT 16 Months ComBined Endoscopy Pos Neg Pos Neg Pos Neg Pos Neg Positive Negative Sensitivity (95% CI) Specificity (95% CI) 96.9 (93, 99) 95.1 (89, 98) 93.9 (88, 97) 95.6 (89, 99) 94.8 (88, 98) 97.6 (92, 100) 95.5 (93, 97) 96.0 (93, 98) *Combined endoscopic methods used were histology and culture per DAIDP guidelines 8 for posttreatment monitoring. Please note that the posttreatment performance characteristics at 1, 3 and 6 months after therapy are not statistically different. Therefore, the single best estimates of sensitivity and specificity are presented in the 16 Months Combined column. Negative Predictive Value (NPV) for PostTreatment Monitoring Given the posttreatment sensitivity (95.5%) and specificity (96.0%) observed in these studies, and assuming a treatment efficacy of 90% (10% prevalence of residual H. pylori infection), the NPV of the Meretek UBT is greater than 99%. When efficacy of treatment drops to 50%, the NPV is still greater than 95%. 10/14
11 XIII. Bibliography 1. Marshall, B.J., Warren, J.R. Unidentified curved bacilli on gastric epithelium in active chronic gastritis, Lancet, June 4: ; Northfield T.C., Mendall M., Goggin P.M., (Eds), Helicobacter pylori infection. Pathophysiology, Epidemiology and Management. Kluwer Academic Publisher (1993). 3. Rathbone B.J., Heatley R.V., (Eds) Helicobacter pylori and Gastroduodenal Disease, Blackwell Scientific Publications, 2nd edition (1992). 4. Helicobacter pylori in Peptic Ulcer Diseases, Program and Abstracts. NIH Consensus Development Conference, February 79, 1994, Bethesda, MD. 5. NIH Consensus Development Panel, H. pylori in Peptic Ulcer Disease, JAMA, July 6, 1994 Vol. 272, No.1, Reference 2, page Graham, D.Y., Runke, D., Anderson, S., Malaty, H.M., and Klein, P.D. Citric Acid as the Test Meal for the 13CUrea Breath Test. American Journal of Gastroenterology, 5, ; Borriello, S.P., Reed, P.J., Dolby, J.M., Barclay, F.E. and Webster, A.D.B. Microbial and metabolic profile of achlorhydric stomach: comparison of pernicious anemia and hypogammaglobulinaemia. J. Chin. Pathol. 38, ; FDA, Center for Drug Evaluation and Research, Division of AntiInfective Drug Products, DAIDP Points to consider document Helicobacter pyloriassociated Peptic Ulcer Disease. Indication #25. (March 1995 Addendum to March 15, 1995 Draft). XIV. Name and Place of Business The BreathTekâ UBT for H. pylori Collection Kit is manufactured for Meretek Diagnostics Group of Otsuka America Pharmaceutical, Inc., 2440 Research Boulevard, Rockville, MD XV. Labeling Revision Information Revision: September 2008 Print Code: 0508L0177 Part Number: AC Directions For Use: The PranactinRCitric in this container is intended for use only as a componet of a BreathTek TM UBT for H. pylori. See package insert for instructions on how to prepare the solution and directions for use. Phenylketonurics: Contains Phenylalanine, 84 mg Per Pouch Part No AA Print Code: 0508L0155 US PATENT 4,830,010 NDC R Pranactin Citric 11/14
12 Contains: CUrea, 75mg For in vitro diagnostic use only. The PranactinRCitric drug is taken orally as part of the BreathTek TM UBT for H. pylori. Store at C (59 86 F) Manufactured for Otsuka America Pharmaceutical, Inc. Rockville, MD AA 13 o o o o 12/14
13 PRANACTINCITRIC urea c13 powder, for solution Product Information Product Type HUMAN PRESCRIPTION DRUG NDC Product Code (Source) Route of Administration ORAL DEA Schedule Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength 13/14
14 UREA C13 (UREA C13) UREA C13 75 mg in 3000 mg Inactive Ingredients Ingredient Name ANHYDROUS CITRIC ACID MANNITOL ASPARTAME Strength 2000 mg in 3000 mg 775 mg in 3000 mg 150 mg in 3000 mg Product Characteristics Color Score Shape Flavor Contains Size Imprint Code Packaging # NDC Package Description Multilevel Packaging mg In 1 KIT None Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA /10/2001 Labeler Otsuka America Pharmaceutical ( ) Registrant Otsuka America Pharmaceutical ( ) Revised: 08/2009 Otsuka America Pharmaceutical 14/14
H. pylori Antigen ELISA Kit
 H. pylori Antigen ELISA Kit Catalog Number KA3142 96 assays Version: 04 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle of
H. pylori Antigen ELISA Kit Catalog Number KA3142 96 assays Version: 04 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle of
H.Pylori IgG
 DIAGNOSTIC AUTOMATION, INC. 21250 Califa Street, Suite 102 and116, Woodland Hills, CA 91367 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com
DIAGNOSTIC AUTOMATION, INC. 21250 Califa Street, Suite 102 and116, Woodland Hills, CA 91367 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com
H.Pylori IgG Cat # 1503Z
 DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
H. pylori IgM. Cat # H. pylori IgM ELISA. ELISA: Enzyme Linked Immunosorbent Assay. ELISA - Indirect; Antigen Coated Plate
 DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com H. pylori
DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com H. pylori
H. pylori IgM CLIA kit
 H. pylori IgM CLIA kit Cat. No.:DEEL0251 Pkg.Size:96 tests Intended use Helicobacter pylori IgM Chemiluminescence ELISA is intended for use in evaluating the serologic status to H. pylori infection in
H. pylori IgM CLIA kit Cat. No.:DEEL0251 Pkg.Size:96 tests Intended use Helicobacter pylori IgM Chemiluminescence ELISA is intended for use in evaluating the serologic status to H. pylori infection in
Is it just a bellyache, or could it be an. infection? Please see accompanying BRIEF SUMMARY and enclosed Current Package Insert.
 Is it just a bellyache, or could it be an infection? Please see accompanying BRIEF SUMMARY and enclosed Current Package Insert. I have a bellyache. As a parent or caregiver, anytime you hear your child
Is it just a bellyache, or could it be an infection? Please see accompanying BRIEF SUMMARY and enclosed Current Package Insert. I have a bellyache. As a parent or caregiver, anytime you hear your child
H.pylori IgA Cat #
 DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
TRANSPARENCY COMMITTEE OPINION. 13 December 2006
 The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 13 December 2006 HELIKIT 75 mg, powder for oral solution CIP : 343 132-1 Applicant : MAYOLY SPINDLER 13 Curea anhydrous
The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 13 December 2006 HELIKIT 75 mg, powder for oral solution CIP : 343 132-1 Applicant : MAYOLY SPINDLER 13 Curea anhydrous
H.Pylori IgM Cat # 1504Z
 DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
See external label 2 C-8 C Σ=96 tests Cat # 1505Z. MICROWELL ELISA H.Pylori IgA Cat # 1505Z
 DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
SCIENTIFIC DISCUSSION
 SCIENTIFIC DISCUSSION This module reflects the initial scientific discussion and scientific discussion on procedures, which have been finalised before 1 October 2001. For scientific information on procedures
SCIENTIFIC DISCUSSION This module reflects the initial scientific discussion and scientific discussion on procedures, which have been finalised before 1 October 2001. For scientific information on procedures
Helicobacter Pylori Testing HELICOBACTER PYLORI TESTING HS-131. Policy Number: HS-131. Original Effective Date: 9/17/2009
 Easy Choice Health Plan, Inc. Harmony Health Plan of Illinois, Inc. Missouri Care, Inc. Ohana Health Plan, a plan offered by WellCare Health Insurance of Arizona, Inc. WellCare Health Insurance of Illinois,
Easy Choice Health Plan, Inc. Harmony Health Plan of Illinois, Inc. Missouri Care, Inc. Ohana Health Plan, a plan offered by WellCare Health Insurance of Arizona, Inc. WellCare Health Insurance of Illinois,
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS
 ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1 1. NAME OF THE MEDICINAL PRODUCT Helicobacter Test INFAI 75 mg powder for oral solution 2. QUALITATIVE AND QUANTITATIVE COMPOSITION One jar contains 75 mg of
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1 1. NAME OF THE MEDICINAL PRODUCT Helicobacter Test INFAI 75 mg powder for oral solution 2. QUALITATIVE AND QUANTITATIVE COMPOSITION One jar contains 75 mg of
Helicobacter pylori Antigen Test
 Helicobacter pylori Antigen Test Instructions For Use Format: Cassette For: Catalog Number: VEL-001-HP(s) Specimen: Fecal Specimen * Please read the instructions carefully before use INTENDED USE Helicobacter
Helicobacter pylori Antigen Test Instructions For Use Format: Cassette For: Catalog Number: VEL-001-HP(s) Specimen: Fecal Specimen * Please read the instructions carefully before use INTENDED USE Helicobacter
Gilles Jequier. Commercial Director Organobalance, a Novozymes Company
 "Latest clinical Evidences showing that a proprietary Lactobacillus reuteri Strain can reduce the Symptoms associated with a Helicobacter pylori Infection" Gilles Jequier Commercial Director Organobalance,
"Latest clinical Evidences showing that a proprietary Lactobacillus reuteri Strain can reduce the Symptoms associated with a Helicobacter pylori Infection" Gilles Jequier Commercial Director Organobalance,
PYTEST - urea c-14 capsule Avent, Inc Description
 PYTEST - urea c-4 capsule Avent, Inc. ---------- PYtest ( 4 Description 4 C-Urea Capsules) PYtest ( C-urea capsules) is intended for use in the detection of gastric urease as an aid in the diagnosis of
PYTEST - urea c-4 capsule Avent, Inc. ---------- PYtest ( 4 Description 4 C-Urea Capsules) PYtest ( C-urea capsules) is intended for use in the detection of gastric urease as an aid in the diagnosis of
Urea Breath Test for Diagnosis of Helicobactor pylori. Original Policy Date 12:2013
 MP 2.04.04 Urea Breath Test for Diagnosis of Helicobactor pylori Medical Policy Section Medicine Issue 12:2013 Original Policy Date 12:2013 Last Review Status/Date 12:2013 Return to Medical Policy Index
MP 2.04.04 Urea Breath Test for Diagnosis of Helicobactor pylori Medical Policy Section Medicine Issue 12:2013 Original Policy Date 12:2013 Last Review Status/Date 12:2013 Return to Medical Policy Index
Corporate Medical Policy
 Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: helicobacter_pylori_testing 01/01/2019 N/A 01/01/2020 01/01/2019 Policy Effective April 1, 2019 Description
Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: helicobacter_pylori_testing 01/01/2019 N/A 01/01/2020 01/01/2019 Policy Effective April 1, 2019 Description
H. pylori Stool Rapid Test (Cassette)
 H. pylori Stool Rapid Test (Cassette) Cat. No.:DTS590 Pkg.Size: Intended use The H. pylori Stool Cassette is an immunochromatographic screening assay for the qualitative detectionof Helicobacter pylori
H. pylori Stool Rapid Test (Cassette) Cat. No.:DTS590 Pkg.Size: Intended use The H. pylori Stool Cassette is an immunochromatographic screening assay for the qualitative detectionof Helicobacter pylori
CLIA Complexity for Whole Blood: WAIVED. Intended Use. Summary and Explanation
 CLIA Complexity for Whole Blood: WAIVED Intended Use The is a lateral-flow immunoassay intended for the rapid, qualitative detection of IgG antibodies specific to Helicobacter pylori in whole blood. The
CLIA Complexity for Whole Blood: WAIVED Intended Use The is a lateral-flow immunoassay intended for the rapid, qualitative detection of IgG antibodies specific to Helicobacter pylori in whole blood. The
Catalog # 0W009 0W010 Test Cassettes, murine monoclonal antibody to human IgG (Test Line) and rabbit polyclonal antibody (Control Line)
 For in vitro diagnostic use. CLIA Complexity for Whole Blood: WAIVED INTENDED USE The QuickVue H. pylori Test is a lateral-flow immunoassay intended for the rapid, qualitative detection of IgG antibodies
For in vitro diagnostic use. CLIA Complexity for Whole Blood: WAIVED INTENDED USE The QuickVue H. pylori Test is a lateral-flow immunoassay intended for the rapid, qualitative detection of IgG antibodies
Helicobacter and gastritis
 1 Helicobacter and gastritis Dr. Hala Al Daghistani Helicobacter pylori is a spiral-shaped gram-negative rod. H. pylori is associated with antral gastritis, duodenal (peptic) ulcer disease, gastric ulcers,
1 Helicobacter and gastritis Dr. Hala Al Daghistani Helicobacter pylori is a spiral-shaped gram-negative rod. H. pylori is associated with antral gastritis, duodenal (peptic) ulcer disease, gastric ulcers,
Catalog # 0W009 0W010 Test Cassettes, murine monoclonal antibody to human IgG (Test Line) and rabbit polyclonal antibody (Control Line) Reagents
 For in vitro diagnostic use. CLIA Complexity: MODERATE INTENDED USE The QuickVue H. pylori Test is a lateral-flow immunoassay intended for the rapid, qualitative detection of IgG antibodies specific to
For in vitro diagnostic use. CLIA Complexity: MODERATE INTENDED USE The QuickVue H. pylori Test is a lateral-flow immunoassay intended for the rapid, qualitative detection of IgG antibodies specific to
Helicobacter Connections. Barry Marshall
 Helicobacter Connections Barry Marshall The greatest obstacle to knowledge is not ignorance, it is the illusion of knowledge. Daniel Boorstein - Historian Peptic Ulcers Duodenal Ulcer (DU) Gastric Ulcer
Helicobacter Connections Barry Marshall The greatest obstacle to knowledge is not ignorance, it is the illusion of knowledge. Daniel Boorstein - Historian Peptic Ulcers Duodenal Ulcer (DU) Gastric Ulcer
Helicobacter 2008;13:1-6. Am J Gastroent 2007;102: Am J of Med 2004;117:31-35.
 An Update on Helicobacter pylori and Its Treatment Trenika Mitchell, PharmD, BCPS Clinical Assistant Professor University of Kentucky College of Pharmacy October 18, 2008 Objectives Review the epidemiology
An Update on Helicobacter pylori and Its Treatment Trenika Mitchell, PharmD, BCPS Clinical Assistant Professor University of Kentucky College of Pharmacy October 18, 2008 Objectives Review the epidemiology
Type of intervention Diagnosis. Economic study type Cost-effectiveness analysis.
 Noninvasive tests as a substitute for histology in the diagnosis of Helicobacter pylori infection Hahn M, Fennerty M B, Corless C L, Magaret N, Lieberman D A, Faigel D O Record Status This is a critical
Noninvasive tests as a substitute for histology in the diagnosis of Helicobacter pylori infection Hahn M, Fennerty M B, Corless C L, Magaret N, Lieberman D A, Faigel D O Record Status This is a critical
Rapid-VIDITEST. Helicobacter pylori. One step Helicobacter pylori Blister test. Instruction manual
 Rapid-VIDITEST Helicobacter pylori One step Helicobacter pylori Blister test. Instruction manual Producer: VIDIA spol. s r.o., Nad Safinou II 365, 252 50 Vestec, Czech Republic, Tel.: +420 261 090 565,
Rapid-VIDITEST Helicobacter pylori One step Helicobacter pylori Blister test. Instruction manual Producer: VIDIA spol. s r.o., Nad Safinou II 365, 252 50 Vestec, Czech Republic, Tel.: +420 261 090 565,
ONE-STEP WHOLE BLOOD H.PYLORI TEST. Product Code: 24529
 ONE-STEP WHOLE BLOOD H.PYLORI TEST Product Code: 24529 Contents of Package: - Foil Package (Contains: Test Device and Dropper) - Vial of Test Solution - Finger Stick Device Alcohol Swab Materials required
ONE-STEP WHOLE BLOOD H.PYLORI TEST Product Code: 24529 Contents of Package: - Foil Package (Contains: Test Device and Dropper) - Vial of Test Solution - Finger Stick Device Alcohol Swab Materials required
Comparative study of invasive methods for diagnosis of Helicobacter pylori in humans
 ISSN: 2319-7706 Volume 2 Number 7 (2013) pp. 63-68 http://www.ijcmas.com Original Research Article Comparative study of invasive methods for diagnosis of Helicobacter pylori in humans V.Subbukesavaraja
ISSN: 2319-7706 Volume 2 Number 7 (2013) pp. 63-68 http://www.ijcmas.com Original Research Article Comparative study of invasive methods for diagnosis of Helicobacter pylori in humans V.Subbukesavaraja
Utility of In House made Rapid Urease Broth Test for Detection of Helicobacter pylori Infection in Resource Constraint Settings
 Original article: Utility of In House made Rapid Urease Broth Test for Detection of Helicobacter pylori Infection in Resource Constraint Settings 1.Dr. Swati Rahul Dhope, 2. Dr. Sachinkumar Vasantrao Wankhede
Original article: Utility of In House made Rapid Urease Broth Test for Detection of Helicobacter pylori Infection in Resource Constraint Settings 1.Dr. Swati Rahul Dhope, 2. Dr. Sachinkumar Vasantrao Wankhede
INSTANT-VIEW H. pylori Rapid Test CLIA WAIVED Test for Whole Blood QUALITATIVE IN-VITRO DIAGNOSTIC TEST FOR EXTERNAL USE ONLY
 INTENDED USE INSTANT-VIEW H. pylori Rapid Test CLIA WAIVED Test for Whole Blood QUALITATIVE IN-VITRO DIAGNOSTIC TEST FOR EXTERNAL USE ONLY INSTANT-VIEW H. pylori Rapid Test is a rapid qualitative immunoassay
INTENDED USE INSTANT-VIEW H. pylori Rapid Test CLIA WAIVED Test for Whole Blood QUALITATIVE IN-VITRO DIAGNOSTIC TEST FOR EXTERNAL USE ONLY INSTANT-VIEW H. pylori Rapid Test is a rapid qualitative immunoassay
Setting The setting was community. The economic study was carried out in the USA.
 Costs of managing Helicobacter pylori-infected ulcer patients after initial therapy Griffiths R I, Rabeneck L, Guzman G, Cromwell D M, Strauss M J, Robinson J W, Winston B, Li T, Graham D Y Record Status
Costs of managing Helicobacter pylori-infected ulcer patients after initial therapy Griffiths R I, Rabeneck L, Guzman G, Cromwell D M, Strauss M J, Robinson J W, Winston B, Li T, Graham D Y Record Status
National Digestive Diseases Information Clearinghouse
 Gastritis National Digestive Diseases Information Clearinghouse U.S. Department of Health and Human Services NATIONAL INSTITUTES OF HEALTH What is gastritis? Gastritis is a condition in which the stomach
Gastritis National Digestive Diseases Information Clearinghouse U.S. Department of Health and Human Services NATIONAL INSTITUTES OF HEALTH What is gastritis? Gastritis is a condition in which the stomach
NCCLS. Accutest H. pylori WB Test. (Catalog. No. ID-92A225) For Professional and In Vitro Diagnostic Use Only
 NCCLS Accutest H. pylori WB Test (Catalog. No. ID-92A225) For Professional and In Vitro Diagnostic Use Only A Rapid Visual Test for the Qualitative Detection of IgG Antibodies Specific to Helicobacter
NCCLS Accutest H. pylori WB Test (Catalog. No. ID-92A225) For Professional and In Vitro Diagnostic Use Only A Rapid Visual Test for the Qualitative Detection of IgG Antibodies Specific to Helicobacter
PDF hosted at the Radboud Repository of the Radboud University Nijmegen
 PDF hosted at the Radboud Repository of the Radboud University Nijmegen The following full text is a publisher's version. For additional information about this publication click this link. http://hdl.handle.net/2066/48400
PDF hosted at the Radboud Repository of the Radboud University Nijmegen The following full text is a publisher's version. For additional information about this publication click this link. http://hdl.handle.net/2066/48400
instrument. When 13C-UBT positive value is greater than or equal to / - 0.4, the the subject can be 1. Data and methods details are as follows:
 Application of 13 C-urea breath test in screening helicobacter pylori infection during health examination in Chengdu, Sichuan YANG Yan-hua. LIU Yu-ping. CHENG You-fu, SHUAI Ping. LU Qiao. ZHENG Xiao-xia,
Application of 13 C-urea breath test in screening helicobacter pylori infection during health examination in Chengdu, Sichuan YANG Yan-hua. LIU Yu-ping. CHENG You-fu, SHUAI Ping. LU Qiao. ZHENG Xiao-xia,
Research Article Performance of Routine Helicobacter pylori Invasive Tests in Patients with Dyspepsia
 Gastroenterology Research and Practice Volume 2013, Article ID 184806, 5 pages http://dx.doi.org/10.1155/2013/184806 Research Article Performance of Routine Helicobacter pylori Invasive Tests in Patients
Gastroenterology Research and Practice Volume 2013, Article ID 184806, 5 pages http://dx.doi.org/10.1155/2013/184806 Research Article Performance of Routine Helicobacter pylori Invasive Tests in Patients
The Nobel Prize in Physiology or Medicine for 2005
 The Nobel Prize in Physiology or Medicine for 2005 jointly to Barry J. Marshall and J. Robin Warren for their discovery of "the bacterium Helicobacter pylori and its role in gastritis and peptic ulcer
The Nobel Prize in Physiology or Medicine for 2005 jointly to Barry J. Marshall and J. Robin Warren for their discovery of "the bacterium Helicobacter pylori and its role in gastritis and peptic ulcer
INSTANT-VIEW H. pylori Rapid Test CLIA WAIVED Test for Whole Blood QUALITATIVE IN-VITRO DIAGNOSTIC TEST FOR EXTERNAL USE ONLY
 INENDED USE INSAN-VIEW H. pylori Rapid est LIA WAIVED est for Whole Blood QUALIAIVE IN-VIRO DIAGNOSI ES FOR EXERNAL USE ONLY INSAN-VIEW H. pylori Rapid est is a rapid qualitative immunoassay intended to
INENDED USE INSAN-VIEW H. pylori Rapid est LIA WAIVED est for Whole Blood QUALIAIVE IN-VIRO DIAGNOSI ES FOR EXERNAL USE ONLY INSAN-VIEW H. pylori Rapid est is a rapid qualitative immunoassay intended to
Blood Urea Nitrogen Enzymatic Kit Manual Catalog #:
 Blood Urea Nitrogen Enzymatic Kit Manual Catalog #: 5602-01 TABLE OF CONTENTS GENERAL INFORMATION... 2 Product Description... 2 Procedure Overview... 2 Kit Contents, Storage and Shelf Life... 3 Required
Blood Urea Nitrogen Enzymatic Kit Manual Catalog #: 5602-01 TABLE OF CONTENTS GENERAL INFORMATION... 2 Product Description... 2 Procedure Overview... 2 Kit Contents, Storage and Shelf Life... 3 Required
DERBYSHIRE JOINT AREA PRESCRIBING COMMITTEE (JAPC) MANAGEMENT OF DYSPEPSIA
 DERBYSHIRE JOINT AREA PRESCRIBING COMMITTEE (JAPC) MANAGEMENT OF DYSPEPSIA o Patients of any age with ALARM signs should be referred through the 2-week referral system o Routine endoscopic investigation
DERBYSHIRE JOINT AREA PRESCRIBING COMMITTEE (JAPC) MANAGEMENT OF DYSPEPSIA o Patients of any age with ALARM signs should be referred through the 2-week referral system o Routine endoscopic investigation
Prevalence of Helicobacter pylori in Patients with End Stage Renal Disease
 2000;20:97-102 Helicobacter pylori Prevalence of Helicobacter pylori in Patients with End Stage Renal Disease Do Ha Kim, M.D., Hwoon-Yong Jung, M.D., Suk-Kyun Yang, M.D. Weon-Seon Hong, M.D. and Young
2000;20:97-102 Helicobacter pylori Prevalence of Helicobacter pylori in Patients with End Stage Renal Disease Do Ha Kim, M.D., Hwoon-Yong Jung, M.D., Suk-Kyun Yang, M.D. Weon-Seon Hong, M.D. and Young
Clinical practice - breath tests
 Clinical practice - breath tests G D Bell Gastroenterology Department, Sunderland District General Hospital, Sunderland, UK The underlying principle of the two non-invasive radio-labelled urea breath tests
Clinical practice - breath tests G D Bell Gastroenterology Department, Sunderland District General Hospital, Sunderland, UK The underlying principle of the two non-invasive radio-labelled urea breath tests
T R A I N I N G G U I D E
 TRAINING GUIDE InstaCube Oral Fluid Drug Test For Forensic Use Only Powered By: Contents 3 The information in this presentation is a general overview on performing and interpreting the InstaCube Oral Fluid
TRAINING GUIDE InstaCube Oral Fluid Drug Test For Forensic Use Only Powered By: Contents 3 The information in this presentation is a general overview on performing and interpreting the InstaCube Oral Fluid
PYTEST- urea, c-14 capsule Avent, Inc Des cription
 PYTEST- urea, c-4 capsule Avent, Inc. ---------- 4 PYtes t ( C-Urea Caps ules) Des cription 4 PYtest ( C-urea capsules) is intended for use in the detection of gastric urease as an aid in the diagnosis
PYTEST- urea, c-4 capsule Avent, Inc. ---------- 4 PYtes t ( C-Urea Caps ules) Des cription 4 PYtest ( C-urea capsules) is intended for use in the detection of gastric urease as an aid in the diagnosis
- Helicobacter - THE EASE AND DIFFICULTY OF A NEW DISCOVERY. Robin Warren
 - Helicobacter - THE EASE AND DIFFICULTY OF A NEW DISCOVERY Robin Warren EARLY DAYS First reports 100 years ago considered spirochaetes 1940 Freedburg saw curved organisms in the stomach 1954 Palmer: Freedburg
- Helicobacter - THE EASE AND DIFFICULTY OF A NEW DISCOVERY Robin Warren EARLY DAYS First reports 100 years ago considered spirochaetes 1940 Freedburg saw curved organisms in the stomach 1954 Palmer: Freedburg
COMPARISON OF ONCE-A-DAY VERSUS TWICE-A-DAY CLARITHROMYCIN IN TRIPLE THERAPY FOR HELICOBACTER PYLORI ERADICATION
 Phil J Gastroenterol 2006; 2: 25-29 COMPARISON OF ONCE-A-DAY VERSUS TWICE-A-DAY CLARITHROMYCIN IN TRIPLE THERAPY FOR HELICOBACTER PYLORI ERADICATION Marianne P Collado, Ma Fatima P Calida, Peter P Sy,
Phil J Gastroenterol 2006; 2: 25-29 COMPARISON OF ONCE-A-DAY VERSUS TWICE-A-DAY CLARITHROMYCIN IN TRIPLE THERAPY FOR HELICOBACTER PYLORI ERADICATION Marianne P Collado, Ma Fatima P Calida, Peter P Sy,
For the convenience of storage, Cassette and Sample Tubes can be stored separately. OneStep. H.Pylori Antigen. RapiCard InstaTest.
 CORTEZ DIAGNOSTICS, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 USA Tel: (818) 591-3030 Fax: (818) 591-8383 E-mail: onestep@rapidtest.com Web site: www.rapidtest.com See external label
CORTEZ DIAGNOSTICS, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 USA Tel: (818) 591-3030 Fax: (818) 591-8383 E-mail: onestep@rapidtest.com Web site: www.rapidtest.com See external label
Prevpac Pylera Omeclamox-Pak
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.50.21 Subject: -Pak Page: 1 of 5 Last Review Date: September 20, 2018 -Pak Description (lansoprazole,
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.50.21 Subject: -Pak Page: 1 of 5 Last Review Date: September 20, 2018 -Pak Description (lansoprazole,
Product Training & Certification
 Product Training & Certification Oratect Oral Fluid Drug Screen Device For Professional Point of Care Use Oratect Training Page 1 of 12 MKT-045 (C) Oratect Oral Fluid Drug Screen Device Training and Certification
Product Training & Certification Oratect Oral Fluid Drug Screen Device For Professional Point of Care Use Oratect Training Page 1 of 12 MKT-045 (C) Oratect Oral Fluid Drug Screen Device Training and Certification
PROCEDURE. Sekisui Diagnostics OSOM ifob Rapid Test. Title: Procedure #: Institution: Prepared by: Date: Title: Accepted by: Date adopted: Title:
 SAMPLE PROCEDURE This Sample Procedure is not intended as a substitute for your facility s Procedure Manual or reagent labeling, but rather as a model for your use in customizing for your laboratory s
SAMPLE PROCEDURE This Sample Procedure is not intended as a substitute for your facility s Procedure Manual or reagent labeling, but rather as a model for your use in customizing for your laboratory s
Strep-a-Test Twister Test
 Strep-a-Test Twister Test Code: 24524 A rapid test for the qualitative detection of Strep A antigen in throat swab specimens. For professional in vitro diagnostic use only. INTENDED USE The Strep A Twist
Strep-a-Test Twister Test Code: 24524 A rapid test for the qualitative detection of Strep A antigen in throat swab specimens. For professional in vitro diagnostic use only. INTENDED USE The Strep A Twist
Rotavirus Test Kit. Instructions For Use. Format: Cassette Specimen: Fecal Extract Catalog Number: VEL-001-ROTA
 Rotavirus Test Kit Instructions For Use Format: Cassette Specimen: Fecal Extract Catalog Number: VEL-001-ROTA * Please read the instructions carefully before use INTENDED USE Velotest Rotavirus Test is
Rotavirus Test Kit Instructions For Use Format: Cassette Specimen: Fecal Extract Catalog Number: VEL-001-ROTA * Please read the instructions carefully before use INTENDED USE Velotest Rotavirus Test is
H. Pylori Whole Blood Rapid Test
 H. Pylori Whole Blood Rapid est A LIA Waived Qualitative In-Vitro Diagnostic est for External Use only INENDED USE he LIAwaived, Inc. H. Pylori Whole Blood Rapid est is a rapid qualitative immunoassay
H. Pylori Whole Blood Rapid est A LIA Waived Qualitative In-Vitro Diagnostic est for External Use only INENDED USE he LIAwaived, Inc. H. Pylori Whole Blood Rapid est is a rapid qualitative immunoassay
SAMPLE PROCEDURE , 07/11
 SAMPLE PROCEDURE This Sample Procedure is not intended as a substitute for your facility s Procedure Manual or reagent labeling, but rather as a model for your use in customizing for your laboratory s
SAMPLE PROCEDURE This Sample Procedure is not intended as a substitute for your facility s Procedure Manual or reagent labeling, but rather as a model for your use in customizing for your laboratory s
Blood Urea Nitrogen Enzymatic Kit Manual Catalog #:
 BIOO LIFE SCIENCE PRODUCTS Blood Urea Nitrogen Enzymatic Kit Manual Catalog #: 5602-01 BIOO Scientific Corp. 2010 TABLE OF CONTENTS GENERAL INFORMATION... 1 Product Description... 1 Procedure Overview...
BIOO LIFE SCIENCE PRODUCTS Blood Urea Nitrogen Enzymatic Kit Manual Catalog #: 5602-01 BIOO Scientific Corp. 2010 TABLE OF CONTENTS GENERAL INFORMATION... 1 Product Description... 1 Procedure Overview...
Helicobacter pylori Eradication Therapy Success Regarding Different Treatment Period Based on Clarithromycin or Metronidazole Triple-Therapy Regimens
 Helicobacter ISSN 1523-5378 Filipec Blackwell Oxford, HEL 1083-4389 1523-5378 Journal XXX Original H. 2008 pylori Kanizaj compilation The UK Eradication Publishing Article Authors et al. Ltd 2008 Therapy
Helicobacter ISSN 1523-5378 Filipec Blackwell Oxford, HEL 1083-4389 1523-5378 Journal XXX Original H. 2008 pylori Kanizaj compilation The UK Eradication Publishing Article Authors et al. Ltd 2008 Therapy
Management of Dyspepsia
 MPharm Programme Management of Dyspepsia Slide 1 of 28 Learning Objectives Understand the principles and wider implications underpinning evidence based therapeutics in the key clinical specialities Objectively
MPharm Programme Management of Dyspepsia Slide 1 of 28 Learning Objectives Understand the principles and wider implications underpinning evidence based therapeutics in the key clinical specialities Objectively
Instructions for Use. For use with. 10 mg vial
 Instructions for Use For use with 10 mg vial Table of Contents Parts of the ZOMA-Jet 10... 1 Supplies you will need to mix a ZOMACTON 10 mg Vial... 2 Mix a ZOMACTON 10 mg vial... 3 Reset the ZOMA-Jet 10...
Instructions for Use For use with 10 mg vial Table of Contents Parts of the ZOMA-Jet 10... 1 Supplies you will need to mix a ZOMACTON 10 mg Vial... 2 Mix a ZOMACTON 10 mg vial... 3 Reset the ZOMA-Jet 10...
INSTRUCTIONS FOR USE. INBRIJA (in-brih-jah) (levodopa inhalation powder) For Oral Inhalation Only
 INSTRUCTIONS FOR USE INBRIJA (in-brih-jah) (levodopa inhalation powder) For Oral Inhalation Only Read and follow these instructions before you start using INBRIJA and each time you get a refill. There
INSTRUCTIONS FOR USE INBRIJA (in-brih-jah) (levodopa inhalation powder) For Oral Inhalation Only Read and follow these instructions before you start using INBRIJA and each time you get a refill. There
The PYtest The Cutting Edge Urea Breath Test to detect Helicobacter pylori
 The PYtest The Cutting Edge Urea Breath Test to detect Helicobacter pylori Barry Marshall The Gold-Standard Non-invasive Diagnostic Test for The Detection of H.pylori As a Healthcare Provider... Do you
The PYtest The Cutting Edge Urea Breath Test to detect Helicobacter pylori Barry Marshall The Gold-Standard Non-invasive Diagnostic Test for The Detection of H.pylori As a Healthcare Provider... Do you
NEW! KIBION DYNAMIC 13 C BREATH TEST ANALYSER. Rapid and reliable diagnosis of Helicobacter pylori infections
 NEW! KIBION DYNAMIC 13 C BREATH TEST ANALYSER Rapid and reliable diagnosis of Helicobacter pylori infections Help prevent chronic Helicobacter pylori infections and the development of gastric cancer More
NEW! KIBION DYNAMIC 13 C BREATH TEST ANALYSER Rapid and reliable diagnosis of Helicobacter pylori infections Help prevent chronic Helicobacter pylori infections and the development of gastric cancer More
Fecal H. pylori Antigen Rapid Test (Strip)
 Fecal H. pylori Antigen Rapid Test (Strip) Cat. No.:DTS526 Pkg.Size: Intended use This H. pylori Antigen Rapid Test is intended for the direct qualitative detection of the presence of H. pylori antigen
Fecal H. pylori Antigen Rapid Test (Strip) Cat. No.:DTS526 Pkg.Size: Intended use This H. pylori Antigen Rapid Test is intended for the direct qualitative detection of the presence of H. pylori antigen
Step-by-Step Instructions For OraQuick HCV Rapid Antibody Test
 Step-by-Step Instructions For OraQuick HCV Rapid Antibody Test Complexity: WAIVED for fingerstick whole blood and venipuncture whole blood. A Certificate of CLIA Waiver is required to perform the test
Step-by-Step Instructions For OraQuick HCV Rapid Antibody Test Complexity: WAIVED for fingerstick whole blood and venipuncture whole blood. A Certificate of CLIA Waiver is required to perform the test
Evaluation of a new rapid immunoassay for the detection of Helicobacter pylori in faeces: a prospective pilot study
 Aliment Pharmacol Ther 2005; 21: 485 489. doi: 10.1111/j.1365-2036.2005.02355.x Evaluation of a new rapid immunoassay for the detection of Helicobacter pylori in faeces: a prospective pilot study L. TREVISANI*,
Aliment Pharmacol Ther 2005; 21: 485 489. doi: 10.1111/j.1365-2036.2005.02355.x Evaluation of a new rapid immunoassay for the detection of Helicobacter pylori in faeces: a prospective pilot study L. TREVISANI*,
HBeAg and HBeAg Ab ELISA Kit
 HBeAg and HBeAg Ab ELISA Kit Catalog Number KA0290 96 assays Version: 17 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Principle of the Assay... 3
HBeAg and HBeAg Ab ELISA Kit Catalog Number KA0290 96 assays Version: 17 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Principle of the Assay... 3
American College of Gastroenterology Guideline on the Management of Helicobacter pylori Infection
 American Journal of Gastroenterology ISSN 0002-9270 C 2007 by Am. Coll. of Gastroenterology doi: 10.1111/j.1572-0241.2007.01393.x Published by Blackwell Publishing American College of Gastroenterology
American Journal of Gastroenterology ISSN 0002-9270 C 2007 by Am. Coll. of Gastroenterology doi: 10.1111/j.1572-0241.2007.01393.x Published by Blackwell Publishing American College of Gastroenterology
KEYWORDS Dyspepsia, Acid Peptic Disease, Helicobacter Pylori, Urease, Giemsa, Peptic Ulcer, Non-Ulcer Dyspepsia.
 INCIDENCE OF HELICOBACTER PYLORI WITH ACID PEPTIC DISEASE AND MALIGNANT CONDITIONS OF UPPER GASTROINTESTINAL TRACT IN A TERTIARY CENTRE - A PROSPECTIVE STUDY Karunamoorthy Rajachidambaram 1, Dinkaran Kaarthesan
INCIDENCE OF HELICOBACTER PYLORI WITH ACID PEPTIC DISEASE AND MALIGNANT CONDITIONS OF UPPER GASTROINTESTINAL TRACT IN A TERTIARY CENTRE - A PROSPECTIVE STUDY Karunamoorthy Rajachidambaram 1, Dinkaran Kaarthesan
594 Lewin, Weinstein, and Riddell s Gastrointestinal Pathology and Its Clinical Implications
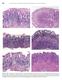 594 Lewin, Weinstein, and Riddell s Gastrointestinal Pathology and Its Clinical Implications Figure 13-20. Stages in the natural history of H. pylori. Biopsies from the antrum are on the left and the oxyntic
594 Lewin, Weinstein, and Riddell s Gastrointestinal Pathology and Its Clinical Implications Figure 13-20. Stages in the natural history of H. pylori. Biopsies from the antrum are on the left and the oxyntic
Oral Specimen Collection Device
 Oral Specimen Collection Device For In Vitro Diagnostic Use NAME AND INTENDED USE The Intercept Oral Specimen Collection Device is intended for use in the collection, preservation and transport of oral
Oral Specimen Collection Device For In Vitro Diagnostic Use NAME AND INTENDED USE The Intercept Oral Specimen Collection Device is intended for use in the collection, preservation and transport of oral
Helicobacter pylori IgA ELISA Kit
 Helicobacter pylori IgA ELISA Kit Catalog Number KA0964 96 assays Version: 03 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle
Helicobacter pylori IgA ELISA Kit Catalog Number KA0964 96 assays Version: 03 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle
MANAGEMENT OF DYSPEPSIA AND GASTRO-OESOPHAGEAL REFLUX DISEASE (GORD)
 DERBYSHIRE JOINT AREA PRESCRIBING COMMITTEE (JAPC) MANAGEMENT OF DYSPEPSIA AND GASTRO-OESOPHAGEAL REFLUX DISEASE (GORD) Routine endoscopic investigation of patients of any age, presenting with dyspepsia
DERBYSHIRE JOINT AREA PRESCRIBING COMMITTEE (JAPC) MANAGEMENT OF DYSPEPSIA AND GASTRO-OESOPHAGEAL REFLUX DISEASE (GORD) Routine endoscopic investigation of patients of any age, presenting with dyspepsia
Low Dose Furazolidone for Eradication of H- pylori Instead of Clarithromycin: A Clinical Trial
 Global Journal of Health Science; Vol. 7, No. 1; 2015 ISSN 1916-9736 E-ISSN 1916-9744 Published by Canadian Center of Science and Education Low Dose Furazolidone for Eradication of H- pylori Instead of
Global Journal of Health Science; Vol. 7, No. 1; 2015 ISSN 1916-9736 E-ISSN 1916-9744 Published by Canadian Center of Science and Education Low Dose Furazolidone for Eradication of H- pylori Instead of
Product Training & Certification
 B R A N A N M E D I C A L C O R P O R A T I O N Product Training & Certification Oratect III Oral Fluid Drug Screen Device Catalog # HM11 & HM12 For Forensic Use Only Branan Medical Corporation 140 Technology
B R A N A N M E D I C A L C O R P O R A T I O N Product Training & Certification Oratect III Oral Fluid Drug Screen Device Catalog # HM11 & HM12 For Forensic Use Only Branan Medical Corporation 140 Technology
CDIA TM Rubella IgG/IgM Rapid Test Kit
 CDIA TM Rubella IgG/IgM Rapid Test Kit Cat.No: DTSJZ024 Lot. No. (See product label) Intended Use The CDIA TM Rubella IgG/IgM Rapid Test Kit is a rapid chromatographic immunoassay for the qualitative detection
CDIA TM Rubella IgG/IgM Rapid Test Kit Cat.No: DTSJZ024 Lot. No. (See product label) Intended Use The CDIA TM Rubella IgG/IgM Rapid Test Kit is a rapid chromatographic immunoassay for the qualitative detection
ReQuest H. Pylori IgG
 Intended use 01-550H 96 Test Set Enzyme Immunoassay for the Detection of Anti- Helicobacter pylori IgG Antibodies in Human Serum The SeraQuest H. Pylori IgG is a qualitative enzyme immunoassay (EIA) kit
Intended use 01-550H 96 Test Set Enzyme Immunoassay for the Detection of Anti- Helicobacter pylori IgG Antibodies in Human Serum The SeraQuest H. Pylori IgG is a qualitative enzyme immunoassay (EIA) kit
Hepatitis C Virus (HCV) Antibody Test
 Hepatitis C Virus (HCV) Antibody Test Instructions For Use Format: Cassette For: Catalog Number: VEL-001-HCV Specimen: Serum/Plasma * Please read the instructions carefully before use INTENDED USE Hepatitis
Hepatitis C Virus (HCV) Antibody Test Instructions For Use Format: Cassette For: Catalog Number: VEL-001-HCV Specimen: Serum/Plasma * Please read the instructions carefully before use INTENDED USE Hepatitis
GlaxoSmithKline. Renal impairment. Hepatic impairment
 RELENZA GlaxoSmithKline Zanamivir QUALITATIVE AND QUANTITATIVE COMPOSITION Each RELENZA ROTADISK consists of four regularly spaced double foil blisters each containing a white to off-white micronised powder
RELENZA GlaxoSmithKline Zanamivir QUALITATIVE AND QUANTITATIVE COMPOSITION Each RELENZA ROTADISK consists of four regularly spaced double foil blisters each containing a white to off-white micronised powder
PEER REVIEW HISTORY ARTICLE DETAILS
 PEER REVIEW HISTORY BMJ Open publishes all reviews undertaken for accepted manuscripts. Reviewers are asked to complete a checklist review form (http://bmjopen.bmj.com/site/about/resources/checklist.pdf)
PEER REVIEW HISTORY BMJ Open publishes all reviews undertaken for accepted manuscripts. Reviewers are asked to complete a checklist review form (http://bmjopen.bmj.com/site/about/resources/checklist.pdf)
Proton Pump Inhibitors Drug Class Prior Authorization Protocol
 Proton Pump Inhibitors Drug Class Prior Authorization Protocol Line of Business: Medi-Cal P&T Approval Date: November 15, 2017 Effective Date: January 1, 2018 This policy has been developed through review
Proton Pump Inhibitors Drug Class Prior Authorization Protocol Line of Business: Medi-Cal P&T Approval Date: November 15, 2017 Effective Date: January 1, 2018 This policy has been developed through review
Correlation Between Endoscopic and Histological Findings in Different Gastroduodenal Lesion and its Association with Helicobacter Pylori
 ORIGINAL ARTICLE Correlation Between Endoscopic and Histological Findings in Different Gastroduodenal Lesion and its Association with Helicobacter Pylori *A. Sultana 1, SM Badruddoza 2, F Rahman 3 1 Dr.
ORIGINAL ARTICLE Correlation Between Endoscopic and Histological Findings in Different Gastroduodenal Lesion and its Association with Helicobacter Pylori *A. Sultana 1, SM Badruddoza 2, F Rahman 3 1 Dr.
Clinical Policy Title: Noninvasive testing for H. pylori
 Clinical Policy Title: Noninvasive testing for H. pylori Clinical Policy Number: 08.01.05 Effective Date: January 1, 2016 Initial Review Date: August 19, 2015 Most Recent Review Date: August 17, 2017 Next
Clinical Policy Title: Noninvasive testing for H. pylori Clinical Policy Number: 08.01.05 Effective Date: January 1, 2016 Initial Review Date: August 19, 2015 Most Recent Review Date: August 17, 2017 Next
Helicobacter pylori: Diagnosis, treatment and risks of untreated infection
 Helicobacter pylori: Diagnosis, treatment and risks of untreated infection Klaus Mönkemüller Department of Gastroenterology, Hepatology und Infectius Diseases Otto-von-Guericke University, Magdeburg bb
Helicobacter pylori: Diagnosis, treatment and risks of untreated infection Klaus Mönkemüller Department of Gastroenterology, Hepatology und Infectius Diseases Otto-von-Guericke University, Magdeburg bb
Monitoring the Health of Transplanted Organs DONOR GENOTYPING MANUAL
 Monitoring the Health of Transplanted Organs DONOR GENOTYPING MANUAL 10101 Innovation Drive. Suite 600. Wauwatosa, WI 53226 1-888-214-3151 MKT-008 v3.0 MKT-008 v3.0 TABLE OF CONTENTS I Intended Use...
Monitoring the Health of Transplanted Organs DONOR GENOTYPING MANUAL 10101 Innovation Drive. Suite 600. Wauwatosa, WI 53226 1-888-214-3151 MKT-008 v3.0 MKT-008 v3.0 TABLE OF CONTENTS I Intended Use...
Clinical Policy Title: Noninvasive testing for H. pylori
 Clinical Policy Title: Noninvasive testing for H. pylori Clinical Policy Number: 08.01.05 Effective Date: January 1, 2016 Initial Review Date: August 19, 2015 Most Recent Review Date: July 3, 2018 Next
Clinical Policy Title: Noninvasive testing for H. pylori Clinical Policy Number: 08.01.05 Effective Date: January 1, 2016 Initial Review Date: August 19, 2015 Most Recent Review Date: July 3, 2018 Next
Clinical Policy: Helicobacter Pylori Serology Testing Reference Number: CP.MP.153
 Clinical Policy: Reference Number: CP.MP.153 Effective Date: 12/17 Last Review Date: 12/17 Coding Implications Revision Log See Important Reminder at the end of this policy for important regulatory and
Clinical Policy: Reference Number: CP.MP.153 Effective Date: 12/17 Last Review Date: 12/17 Coding Implications Revision Log See Important Reminder at the end of this policy for important regulatory and
Review article: 13 C-urea breath test in the diagnosis of Helicobacter pylori infection a critical review
 Aliment Pharmacol Ther 2004; 20: 1001 1017. doi: 10.1111/j.1365-2036.2004.02203.x Review article: 13 C-urea breath test in the diagnosis of Helicobacter pylori infection a critical review J. P. GISBERT
Aliment Pharmacol Ther 2004; 20: 1001 1017. doi: 10.1111/j.1365-2036.2004.02203.x Review article: 13 C-urea breath test in the diagnosis of Helicobacter pylori infection a critical review J. P. GISBERT
MEDICAL POLICY EFFECTIVE DATE: 05/19/11 REVISED DATE: 05/24/12, 05/23/13 ARCHIVED DATE: 05/22/14 EDITED DATE: 05/28/15, 05/25/16, 05/18/17, 05/17/18
 MEDICAL POLICY SUBJECT: NON-INVASIVE HELICOBACTER PAGE: 1 OF: 6 If a product excludes coverage for a service, it is not covered, and medical policy criteria do not apply. If a commercial product, including
MEDICAL POLICY SUBJECT: NON-INVASIVE HELICOBACTER PAGE: 1 OF: 6 If a product excludes coverage for a service, it is not covered, and medical policy criteria do not apply. If a commercial product, including
Disclosures. Co-founder and Chief Science Officer, TechLab
 H. pylori testing Disclosures Co-founder and Chief Science Officer, TechLab Learning Objectives Evaluate the appropriate testing methodology by balancing performance, economics, and workflow. Discuss the
H. pylori testing Disclosures Co-founder and Chief Science Officer, TechLab Learning Objectives Evaluate the appropriate testing methodology by balancing performance, economics, and workflow. Discuss the
SOLUTION TEST STRIPS
 SOLUTION TEST STRIPS Patented AHPHLD8 REF066 (17/055) Virox_InsertBook_PreventionHLD8_06F.indd 1 2017-04-21 11:14 Virox_InsertBook_PreventionHLD8_06F.indd 2 2017-04-21 11:14 INTENDED USE: The Prevention
SOLUTION TEST STRIPS Patented AHPHLD8 REF066 (17/055) Virox_InsertBook_PreventionHLD8_06F.indd 1 2017-04-21 11:14 Virox_InsertBook_PreventionHLD8_06F.indd 2 2017-04-21 11:14 INTENDED USE: The Prevention
Alco-Breath Tube Breath Alcohol Test Training and Certification for Test Administrators
 Alco-Breath Tube Breath Alcohol Test Training and Certification for Test Administrators The information provided is intended to educate test administrators in the use of the Alco-Breath Tube breath alcohol
Alco-Breath Tube Breath Alcohol Test Training and Certification for Test Administrators The information provided is intended to educate test administrators in the use of the Alco-Breath Tube breath alcohol
Treponema Pallidum (TP) Antibody Test
 Treponema Pallidum (TP) Antibody Test Instructions For Use Format: Cassette For: Catalog Number: VEL-001-TP Specimen: Serum/Plasma * Please read the instructions carefully before use INTENDED USE Velotest
Treponema Pallidum (TP) Antibody Test Instructions For Use Format: Cassette For: Catalog Number: VEL-001-TP Specimen: Serum/Plasma * Please read the instructions carefully before use INTENDED USE Velotest
Clinical Policy Title: Noninvasive testing for H. pylori
 Clinical Policy Title: Noninvasive testing for H. pylori Clinical Policy Number: 08.01.04 Effective Date: January 1, 2016 Initial Review Date: August 19, 2015 Most Recent Review Date: August 17, 2016 Next
Clinical Policy Title: Noninvasive testing for H. pylori Clinical Policy Number: 08.01.04 Effective Date: January 1, 2016 Initial Review Date: August 19, 2015 Most Recent Review Date: August 17, 2016 Next
Sputum DNA Collection, Preservation and Isolation Kit 50 Individual Devices
 3430 Schmon Parkway Thorold, ON, Canada L2V 4Y6 Phone: 866-667-4362 (905) 227-8848 Fax: (905) 227-1061 Email: techsupport@norgenbiotek.com Sputum DNA Collection, Preservation and Isolation Kit 50 Individual
3430 Schmon Parkway Thorold, ON, Canada L2V 4Y6 Phone: 866-667-4362 (905) 227-8848 Fax: (905) 227-1061 Email: techsupport@norgenbiotek.com Sputum DNA Collection, Preservation and Isolation Kit 50 Individual
QUICK REFERENCE INSTRUCTIONS For use with Sofia and Sofia 2. Rx only
 QUICK REFERENCE INSTRUCTIONS For use with Sofia and Sofia 2. Rx only Test Procedure Study the Package Insert and User Manual thoroughly before using Quick Reference Instructions. This is not a complete
QUICK REFERENCE INSTRUCTIONS For use with Sofia and Sofia 2. Rx only Test Procedure Study the Package Insert and User Manual thoroughly before using Quick Reference Instructions. This is not a complete
Sequioa Education Systems, Inc. 1
 Functional Diagnostic Medicine Training Program Module 2 The Functional Diagnostic Medicine Approach in the Treatment of Gastrointestinal Dysfunction and Disease Dr. Wayne L. Sodano, D.C., D.A.B.C.I. &
Functional Diagnostic Medicine Training Program Module 2 The Functional Diagnostic Medicine Approach in the Treatment of Gastrointestinal Dysfunction and Disease Dr. Wayne L. Sodano, D.C., D.A.B.C.I. &
Chlamydia trachomatis (CHLa)Test Kit
 Chlamydia trachomatis (CHLa)Test Kit Instructions For Use Format: Cassette Specimen: Urethral/Genital Swab Catalog Number: VEL-001-CHLa * Please read the instructions carefully before use INTENDED USE
Chlamydia trachomatis (CHLa)Test Kit Instructions For Use Format: Cassette Specimen: Urethral/Genital Swab Catalog Number: VEL-001-CHLa * Please read the instructions carefully before use INTENDED USE
INSTRUCTION FOR USE EIA-ANTI-HDV
 GWB-HDVIGG 96 INSTRUCTION FOR USE EIA-ANTI-HDV ENZYME IMMUNOASSAY FOR THE DETECTION OF TOTAL ANTIBODIES TO HEPATITIS DELTA VIRUS Page 1 of 10 Table of contents I. Intended use. 3 II. Contents of the EIA-ANTI-HDV.....
GWB-HDVIGG 96 INSTRUCTION FOR USE EIA-ANTI-HDV ENZYME IMMUNOASSAY FOR THE DETECTION OF TOTAL ANTIBODIES TO HEPATITIS DELTA VIRUS Page 1 of 10 Table of contents I. Intended use. 3 II. Contents of the EIA-ANTI-HDV.....
Realities of Diagnosing Helicobacter pylon Infection in Clinical Practice: A Case for Non-invasive Indirect Methodologies
 YALE JOURNAL OF BIOLOGY AND MEDICINE 71 (1998), pp. 81-90. Copyright 1999. All rights reserved. Realities of Diagnosing Helicobacter pylon Infection in Clinical Practice: A Case for Non-invasive Indirect
YALE JOURNAL OF BIOLOGY AND MEDICINE 71 (1998), pp. 81-90. Copyright 1999. All rights reserved. Realities of Diagnosing Helicobacter pylon Infection in Clinical Practice: A Case for Non-invasive Indirect
