BCH 3000 PRINCIPLES OF BIOCHEMISTRY
|
|
|
- Alisha Watkins
- 5 years ago
- Views:
Transcription
1 BCH 3000 PRINCIPLES OF BIOCHEMISTRY (Semester /15) 1
2 CARBOHYDRATE Carbohydrates as the main component of life Classification Structure Chemical reactions and biochemical functions of carbohydrate 2
3 CARBOHYDRATE 3
4 4
5 CARBOHYDRATES 'hydrate of carbon', with structural formula (CH 2 O)n Carbohydrate = saccharides = biological molecules The basic building blocks of carbohydrates are the monosaccharides i.e. simplest = monosaccharides, eg. glucose These are linked together in longer chains to form oligosaccharides (2-20 residues - eg. Maltose) and polysaccharides (> 20 residues eg. starch, cellulose, glicogen ) 5
6 CARBOHYDRATES The root sacchar- comes from the Latin saccharum, "sugar". Why saccharide called carbohydrate? Most have general formula (CH 2 O)n Many saccharides a. Modified b. Contains amino groups, sulphates, phosphates, etc. 6
7 Functions of carbohydrates important source of energy for the body - 1g of carbohydrate provides 4.2 kcal of energy o Almost all of the cells use glucose to distribute energy -brain cells ; erythrocytes (red blood cells) are completely dependant on glucose as an energy source. Act as energy storage - glycogen stores act as a readily available energy reserve. A person weighing 70kg has a glycogen reserve of about g, which is about kcal. 7
8 Functions of carbohydrates Carbohydrates find many uses as structural elements eg. Cellulose cell walls in plants, bacteria, exoskeleton of insects, Carbohydrates are utilized as raw materials for several industries. For e.g., paper, plastics, textiles etc. Marker molecules for cell recognition Blood types A,B,O Found in biological molecules e g coenzymes and nucleic acids 8
9 Classification of Carbohydrates 1. Monosaccharide- one sugar residue. Most well known is glucose, C 6 H 12 O 6 2. Oligosaccharide- a few (2-9) sugar residues. Most well known is cane sugar or sucrose, C 12 H 22 O Polysaccharide - many sugar residues. Most common are glycogen, starch and cellulose, from animals, plants and plants. 9
10 Monosaccharides 1. Two classes- aldoses (aldehyde) and ketoses (keto) 2. In our formula, (CH 2 O)n, n is 3 or more 3. Simplest are dihydroxycetone, a ketose where n=3, and glyceraldehyde, an aldose where n=3 - Triose 4. Simple sugars monomeric 5. can have different number of carbon atoms 6. can be combined to form disaccharides and polysaccharides 7. some can have a linear or ring structure 10
11 Monosaccharides Aldose = an aldehyde with two or more hydroxyl groups. Ketose = a ketone with two or more hydroxyl groups Both are trioses = simplest monosaccharides; threecarbon sugars 11
12 Aldehyde Ketone back 12
13 Aldoses and Ketoses 13
14 Monosaccharides Both have the same compositions = TAUTOMERS = (isomer) Tautomers are organic compounds that are interconvertible by a chemical reaction called tautomerization. Can change from one form to the other but takes a very long time Catalysts can speed up the change Why tautomer?? Change from one form (aldehyde) to another (ketone) 14
15 Isomers = are molecules with the same chemical formula and often with the same kinds of bonds between atoms, but in which the atoms are arranged differently. Many isomers share similar if not identical properties in most chemical contexts 15
16 ENANTIOMER (Optical isomers) In glycerldehyde chiral carbon = 4 different groups get 2 isomers = enantiomer mirror images which are not superimposable - D and L isomer 16
17 17
18 18
19 19
20 Enantiomers -if they are mirror images of each other- one is the mirror image of the other, two stereoisomers are enantiomers if they are different but each can be superimposed on the mirror image of the other. 20
21 Stereoisomers means that the two molecules differ in their three-dimensional shapes only but that they have the same structural formulas. This means they have the same exact groups attached in the same way. Only the three-dimensional orientation of these groups are different. enantiomers are stereoisomers 21
22 Diastereomers (or diastereoisomers) are stereoisomers that are not enantiomers (nonsuperimposable mirror images of each other). can have different physical properties and different reactivity. pairs of isomers that have opposite configurations at one or more of the chiral centers but are not mirror images of each other cis-trans isomerism is a form of diastereomerism 22
23 Diastereomers 23
24 24
25 QUESTION Which is L isomer? Which is D isomer? Fischer Projections Glyceraldehyde is actually the basis for the L and D nomenclature Solution of D-glyceraldehyde rotates polarized light to the right (dextrorotatory) and L-glyceraldehyde rotates light to the left (levorotatory) 25
26 Plane Polarized Light 26
27 27
28 28
29 29
30 Fischer Projections Monosaccharides can have multiple chiral centers need some conventions for drawing their structures. For linear chains, the stereochemistry is often represented as a Fischer Projection: 30
31 In a Fischer projection the carbon chain is oriented in the vertical direction, with a conformation that projects the carbon bonds onto a flat plane, and with all horizontal bonds projecting out, in front of the plane When the molecule is oriented with the C1 aldehyde at the top, pointing away from the viewer, this defines a convention where the C2 hydroxyl group will be on the left for L-glyceraldehyde, and on the right for D- glyceraldehyde 31
32 Stereochemistry of Longer Monosaccharides i. For longer monosaccharides, the assignment of the L and D configuration is determined by the configuration of the chiral carbon farthest away from the C1 carbonyl (ie. Highest chiral number) ii. Eg. - glucose, a 6-carbon sugar, the C5 carbon is used. If the C5 hydroxyl group is on the left, the molecule is L- glucose. If the hydroxyl group is on the right, it is D- glucose iii. In a carbon chain with 2 possible configurations for each chiral center, there are a total of 2n stereoisomers for a compound with n chiral carbons 32
33 33
34 Some D- Aldose Isomers 34
35 Some D-Ketose Isomers 35
36 Under natural conditions, only one enantiomer predominates the D-isomer cf. amino acid L-isomer e.g. monosaccharide monosaccharide - = D- monosaccharide 36
37 Diastereoisomer : Diastereomers are stereoisomers that are not enantiomers or mirror images of each other. Diastereomers can have different physical properties and different reactivity. 37
38 38
39 Chiral carbons Tetrose Are they enantiomers? 39
40 40
41 PENTOSE Contains 5 carbon atoms 3 chiral carbons = 2 3 stereoisomers - 4 pairs of enantiomers - untuk aldose = aldopentose Stereochemistry of Longer Monosaccharides BUT ketopentose only 2 chiral carbons 4 isomers only 41
42 Some D- Aldose Isomers 42
43 Aldopentose Ketopentose 43
44 HEXOSE Monasaccharide with 6 carbon atoms Number of isomers high Common hexosee = glucose & fructose, mannose & galactose also abundant all play important biological roles 44
45 45
46 Cyclic structures Cyclic Structures are the common form of monosaccharides with 5 or 6 carbon atoms. O O form when the hydroxyl group on C-5 reacts with the aldehyde group or ketone group. 46
47 Drawing the Cyclic Structure for Glucose STEP 1 : Number the carbon chain and turn clockwise to form a linear open chain. H HO H H H C C C C C O OH H OH OH CH 2 OH H H OH OH OH HOCH 2 C C C C C H 2 H OH O H 47
48 H HO H H H O 1 C C OH C H C OH C OH CH 2 OH H H OH H HOCH 2 C C C C C OH OH H OH O H 48
49 Drawing the Cyclic Structure for Glucose STEP 2: Fold into a hexagon. Bond the C5 O to C1. Place the C6 group above the ring. Write the OH groups on C2 and C4 below the ring. Write the OH group on C3 above the ring. Write a new OH on C1. 4 OH 5 6 CH 2 OH OH 3 2 O OH 1 OH 49
50 Drawing the Cyclic Structure for Glucose STEP 3 : Write the new OH on C1 down for the form. up for the form. CH 2 OH O CH 2 OH O OH OH OH OH -D-Glucose OH OH OH OH -D-Glucose 50
51 Summary of the Formation of Cyclic Glucose 51
52 -D-Glucose and β-d-glucose in Solution When placed in solution, cyclic structures open and close. -D-glucose converts to β-d-glucose and vice versa. at any time, only a small amount of open chain forms. CH2OH CH 2 OH CH 2 OH H O O OH OH OH OH OH OH OH OH -D-glucose D-glucose (open) β-d-glucose (36%) (trace) (64%) O O C H OH OH OH 52
53 Cyclic Structure of Fructose Fructose is a ketohexose. forms a cyclic structure. reacts the OH on C-5 with the C=O on C-2. CH 2 OH C O CH 2 OH O CH 2 OH CH 2 OH O OH HO C H OH OH H C OH OH CH 2 OH H C OH CH 2 OH OH OH D-fructose α-d-fructose -D-fructose 53
54 CYCLICAL STRUCTURE CHO with 5 & 6 carbon atoms normally exist as ring structures Cyclization = interactions between functional groups at C-1 & C-5 hemiacetal (in aldohexose) Or between C-2 dan C-5 hemiketal (in ketohexose) carbonyl carbon becomes new chiral centre = anomeric carbon Cyclic sugars 2 different forms - dan = Anomers 54
55 55
56 Hemiacetal & Hemiketal 56
57 Drawing the Cyclic Structure for Glucose New Chiral centre Glucose 57
58 Drawing the Cyclic Structure for Fructose 1 New Chiral centre Fructose 58
59 59
60 60
61 61
62 CYCLICAL STRUCTURES CHO 5 carbon = furanose furan CHO 6 carbon = pyranose Pyran Normally CHO > 5 carbon in cyclic form o o Free carbonyl group can anomer Can change from one form to another 62
63 63
64 64
65 Furanose 65
66 pyranose 66
67 Which isomer used for reaction? Certain reactions any isomer Others only one anomer eg. RNA & DNA requires -D-ribose & - D-deoksiribose Fischer Projection usefull to explain `stereochemsitry sugars, But does not give true picture of overall shape accurately. use HAWORTH PROJECTION FORMULA 67
68 Fischer Projection Formula Haworth Projection Formula or? 68
69 69
70 70
71 Important Simple Monosaccharides 1. Glucose 2. Mannose 3. Galactose 4. Fructose 5. Ribose 71
72 RNA - only ribofuranose Dinding sel (polysaccharide) - pyranose KETOPENTOSE almost all in cyclic form, but only furanose eg. a -D-ribulose 72
73 73
74 REACTIONS OF MONOSACCHARIDES 1. Mutarotation. 2. Oxidation to CO 2 + H 2 O Reactions due to aldehyde group 3. Reducing sugars. 4. Reduction to polyols. Reactions due to alcohol group 5. Esterification. 6. Formation of acetals, also called glycosides 74
75 75
76 REACTIONS OF MONOSACCHARIDES 1. Oxidation & Reduction Important in biochemistry provides energy when CHO completely oxidised Photosynthesis reversible process when CO 2 & H 2 O reduced Oxidation reaction can be used to detect the presence of carbohydrates eg. Aldehyde [O] carboxyl basis for test for aldose When aldehyde is oxidised, the oxidising agent is reduced 76
77 Because of his property, they are called reducing agents. Ketose is also a reducing agent Y? Ketoses can also be reducing sugars because they can isomerise (a tautomerisation) to aldoses via an enediol: 77
78 REDUCING SUGARS Sugars that contain aldehyde groups that are oxidised to carboxylic acids are classified as reducing sugars. They are classified as reducing sugars since they reduce the Cu 2+ to Cu + which forms as a red precipitate, copper (I) oxide. 78
79 Common test reagents are : Benedicts reagent (CuSO 4 / citrate) Fehlings reagent (CuSO 4 / tartrate) hemi-acetal Remember that aldehydes (and hence aldoses) are readily oxidised In order for oxidation to occur, the cyclic form must first ring-open to give the reactive aldehyde (?) So any sugar that contains a hemi-acetal will be a reducing sugar. But glycosides which are acetals are not reducing sugars. 79
80 80
81 Another example of reagent for reducing sugars Tollen s Reagent Use silver ammonium complex Ag (NH 3 ) 2+ as oxidising agent mirror precipitate on the walls of test tube RCHO + 2Ag(NH 3 ) 2+ + OH - RCOO Ag + + 3NH 3 + NH 4+ + H 2 O Other methods use enzyme glucose oxidase to detect glucose 81
82 ESTERIFICATION REACTION Hydroxyl group (OH) in CHO reacts with acids ester E.g. Phosphate ester intermediate in the breakdown of CHO energy Phosphate ester is normally formed when the phosphate from ATP reacts with sugars phosphorylated sugar important in the metabolism of CHO 82
83 MONASACCHARIDE DERIVATIVES Monosaccharides have several OH groups can bind/exchange with other groups modify the original structures A. Phosphate esters Phosphate esters found in many metabolsic pathways - ATP, ADP, etc B. Acid & lactones Oxidation of monosaccharides produces 1. Aldonic acids e.g. aldose reacting with alkaline solution of Cu + 2. Lactone & uronic acids - [O] with enzim 83
84 Mannose 84
85 85
86 C. Alditols Reduction of C=O polyhydroxy compounds = eg D- mannitol & D-glucitol Ribitol or Adonitol is a crystalline pentose alcohol (C 5 H 12 O 5 ) formed by the reduction of ribose. It occurs naturally in the plant Adonis vernalis 86
87 87
88 Mannitol or 1,2,3,4,5,6-hexanehexol (C 6 H 8 (OH) 6 ) is a vasodilator which is used mainly to reduce pressure in the cranium, Chemically, mannitol is an alcohol and a sugar, or a polyol; it is similar to xylitol or sorbitol. Mannitol is also used as a sweetener for people with diabetes. 88
89 89
90 D.Amino sugars 2 derivatives of amino sugars in polysaccharides glucosamine & galactosamine Exchange of OH with NH 2 90
91 91
92 E. Glycoside glycosides are certain molecules in which a sugar part is bound to some other part Bond = glycosidic bond Formed when H 2 O is removed from OH of saccharide & other compounds containing OH - Found in plants/animals e.g. - Salicin, a glycoside related to Aspirin NB: OH - must be attached to anomeric carbon 92
93 Salicin, a glycoside related to Aspirin 93
94 94
95 OH - from sugar with another OH - ether bond OH - - must be anomeric bond= glycosidic bond product = glycoside furanose = furanoside, pyranose = pyranoside. 95
96 OLIGOSACCHARIDES Glycosidic bonds between monosaccharides = basis for the formation of oligosaccharides and polysaccharides Bonds = between a anomer or b anomer and another OH - of another sugar lots of combinations - OH - must be numbered to differentiate notation for glycosidic bond which anomeric atom involved e.g.. (1 4), (1 6), (1 1) 96
97 97
98 OLIGOSACCHARIDES Formed when two monosaccharides are joined together by glycosidic bond Plays important roles in living organisms Simplest and most important oligosaccharide = disaccharide Examples of disaccharide = sucrose, lactose, maltose 98
99 4 important characteristics to differentiate disaccharides 1. monomer found and their configuration 2. which carbon involved in the bond 3. the arrangement of the monomers 4. the anomeric configuration of the hydroxyl group 99
100 Chemical characteristics of poly- & oligosaccharides formed will depend on 1. Chemical characteristics of monosaccahrides 2. The type of glycosidic bonds formed (i.e. which anonmer; which carbon atom etc ) e.g. The differences between celluloses and starch is because of the difference in the glycosidic bond formed between glucose Because of the variation in the glycosidic bond, can get various types of polymers branched or linear 100
101 101
102 102
103 Bond: (1 4) 103
104 104
105 105
106 106
107 107
108 108
109 Example. Arrangement starts with non-reducing end left anomeric & enantiomeric with prefixes Cyclic configuration with suffix Atoms between glycosidic bonds the number inside bracket between residues Not all oligosaccharides are dimeric; also possible to have trimers, tetramer and bigger 109
110 110
111 Sucrose - 2 Monosaccharides = -D-Glucose & -D- fructose Glucose = aldohexose = pyranose ; Fructosee = ketohexose = furanose -C-1glucose attached to fructose Not reducing sugar - why? because both anomeric groups are involved in glycosidic bond But free glucose and fructose are reducing sugars. When sucrose is digested, hydrolysed glucose & frctose energy 111
112 112
113 Lactose A disaccharide formed from -D- galactose & - D-glucose Galctose = epimer of glucose - ie. Reverse position at C-4 Glycosidic bond = (1 4) between anomeric C- 1 ( form) of galactose and C-4 carbon of glucose 113
114 Because anomeric carbon of glucose is NOT involved in the bond formation, can be in either & Lactose = reducing sugar because the groups at the anomeric carbon atom (glucose) is not involved in glycosidic bond formation; hence can react with oxidising agents 114
115 115
116 Lactose= milk sugar LACTOSE INTOLERANCE Human can be allergic to milk or milk products why??? lack of lactase (breaks lactose to galactose and glucose) lactose will accumulate Lactose will be acted on by lactase bacteria produce Hydrogen gas, CO 2 & organic acids problems with digestion bloating and diarrhea Although lactose can be degraded to galactose, galactose has to be isomerised to glucose before being absorbed can accumulate GALACTOSEMIA mental retardation 116
117 117
118 Maltose a disaccharide hydrolytic product of starch 2 molecules of D-glucose - -D-glucose & -D-glucose joined by (1 4) bond Different from celobiose o o o hydrolysis of cellulose different glycosidic bond - D-glucose attached through (1 4)bond maltose can be digested by humans, cellobiose cannot 118
119 119
120 Epimers are diastereomers that differ in configuration of only one stereogenic center Diastereomers are a class of stereoisomers that are nonsuperposable, non-mirror images of one another 120
121 Epimers 121
122 POLYSACCAHARIDE various functions sequence of monmer determines the primary structure normally simple monomers 1 type of monomer = homopolysaccharide more = heteropolysaccharide normally not complex not more than 2 residues Cf. Protein & nucleic acids - well defined length polysaccharide chain - random length - 122
123 Storage Polysaccharide 1. Important examples - amylose & amylopectin - starch in plants and & glycogen in mammals and bacterial cells 2. amylose, amylopectin & glycogen = homopolysaccharide (glucan) - deposited in the liver, otot = polymer -D-glucopyranose- Difference bond between residues 123
124 amylose - linear, α(1 4) Amylopectin & glycogen α(1 4) + α(1 6), branched polymer Glycogen - more branched; if not they are very similar 124
125 125
126 Regular and simple structure regular secondary structure α(1 4) bond, each residue leans slightly compared to the previous resisue helical conformation However, the helix is not stable- e.g. amylose form random coils Iodine can stabilise helix because it can fit the core of the helix Complex blue in colour 126
127 127
128 Glycogen & amylopectin cannot get blue colour??? Branches inhibit formation of helix To form a helix need 12 residue for ever turn amylopectin residues, glycogen - 8 residue not enough 128
129 129
130 130
131 STRUCTURAL POLYSACCHARIDE Plants do not use/synthesise structural protein use specialised polysaccharide animal use both Cellulose main polymer in plants - woody/fibrous Polymer linear - D-glucose ( glucan) Joined through (1 4)bond 131
132 132
133 Animals- can digest starch can cleave bond Cellulose - cannot requires symbiotic bacteria - produce enzyme cellulase Ruminant - OK- cellulase is present White ants protozoa can digest cellulse Fungi - eg. mushroom live on rottting wood, etc Other polysaccharides also found. e.g. xylans = polymer (1 4) - linked D- xylopyranose Glucomanan = hemicellulose 133
134 Cellulose Not confined to plants only Marine Invetebrata - eg. Tunicates - cellulose in the mantle in connective tissue - human 134
135 Tunicates 135
136 Tunicates 136
137 137
138 CHITIN Similar to cellulose smalll difference - homopolymer N-asetil-Dglucosamine - minor constituent in fungi and algae- replace cellulose Role in invertebrate - exoskeleton of arthropod & mollusks 138
139 139
140 140
141 141
142 GLYCOSAMINOGLYCAN In vertebrata - previously known as mucopolysacharide chondroitin sulphate keratan sulphate connective tissue dermatan sulphate skin Hyaluronic acid 1. All are polymers = repeating units of disaccharides 2. Sugar (CHO) = N- asetylglucosemine 142
143 143
144 144
145 Main functions of glycosaminoglycan formationn of matrix that bind protein components with connective tissue eg. Proteoglycan inside cartilage - filemental structure synthesised from hyaluronic acid with protein core Protein - keratin sulphate chain and chondroitin sulphate chains are attached Structure - collagen fibers becomes compact and strong Bonds electrostatic between sulphate and basic collagen side chains 145
146 146
147 Non Structural function of glycosaminoglycan Hyaluronic acid very soluble in water- found in-synovial fluid- lubricating agent for joints vitreous humor eyes agent for increasing fluidity Heparin - anticoagulant in body tissues bound strongly to blood proteins (prothrombin III) prevets enziymes in the coagulation of blood 147
148 Polysaccharides in bacterial cell walls Gram + & Gram based on cell wall Gram + = peptidoglycan = polysaccharide peptide complex which are multilayered and crosslinked Gram - = single layer of peptidoglycan covered with membrane layer 148
149 Gram + bacteria 149
150 Gram - bacteria 150
151 Importance of other peptidoglycas A few antibiotics inhibit bacterial growth by preventing peptidoglycan layers Lysozymes can dissolve cell walls cell lysis bacterial death Also found in bacteriophage, white of egg, eyedrops of humans 151
152 GLYCOPROTEIN Many proteins are bound to saccharide = glycoprotein Different functions Saccharide chain (= glycan) bound to protein -2 ways Bound to N of the amino group of asparagine - (N- Iinked Glycans) Through N-acetylgalactosamine Differet structures complex branched structures Different functions protein indicators old proteins that need to be destroyed 152
153 ABO blood group system, the classification of human blood based on the inherited properties of red blood cells (erythrocytes) as determined by the presence or absence of the antigens A and B, which are carried on the surface of the red cells. These antigens may be proteins, carbohydrates, glycoproteins, or glycolipids, depending on the blood group system Persons may thus have type A, type B, type O, or type AB blood 153
154 154
155 eg. immunoglobin sialic acid residues will be cleaved slowly, then receptor will recognise and bind to the protein, engulf the protein O-linked Glycans Different functions e.g.. Antartic fish have glycoprotein which acts as an anti freez fluids do not freeze although temperature below freezing point Mucins - glycoprotein in salive increase viscosity of fluid 155
156 Humans can produce antibodies against the A & B, but NOT O; i.e. O is antigenic antigenic Normally, antibodies react against other antigens. eg. Type A carries antibodies against B - therefore when receiving blood type B - will clot & precipitate Blood type O carries antibodies against A & B cannot receive blood type A and B; but CAN donate to both Bllod Type AB carries A & B antigens; therefore no antibodies against B ; only donatieto AB only 156
157 Oligosaccharides as CELL MARKERS Blood group antigens a cell recognition phenomenon Cells need to be marked (on the surface) so that they can interact with other cells can recognize own cells from other external cells In animals there a layer of saccharides bound to a protein or lupid in the membranes e.g. glycocalyx - can interact with bacteria in the intestine; collagen To act as signal, need to have a specific protein bound specifically - imunoglobulin Other examples lectin interact between cells and proteins in the intercellulr matrix to maintain the structure of tissues and organs 157
Carbohydrates. Chapter 12
 Carbohydrates Chapter 12 Educational Goals 1. Given a Fischer projection of a monosaccharide, classify it as either aldoses or ketoses. 2. Given a Fischer projection of a monosaccharide, classify it by
Carbohydrates Chapter 12 Educational Goals 1. Given a Fischer projection of a monosaccharide, classify it as either aldoses or ketoses. 2. Given a Fischer projection of a monosaccharide, classify it by
Chapter 16: Carbohydrates
 Vocabulary Aldose: a sugar that contains an aldehyde group as part of its structure Amylopectin: a form of starch; a branched chain polymer of glucose Amylose: a form of starch; a linear polymer of glucose
Vocabulary Aldose: a sugar that contains an aldehyde group as part of its structure Amylopectin: a form of starch; a branched chain polymer of glucose Amylose: a form of starch; a linear polymer of glucose
Fundamentals of Organic Chemistry. CHAPTER 6: Carbohydrates
 Fundamentals of Organic Chemistry CHEM 109 For Students of Health Colleges Credit hrs.: (2+1) King Saud University College of Science, Chemistry Department CHEM 109 CHAPTER 6: Carbohydrates Carbohydrates
Fundamentals of Organic Chemistry CHEM 109 For Students of Health Colleges Credit hrs.: (2+1) King Saud University College of Science, Chemistry Department CHEM 109 CHAPTER 6: Carbohydrates Carbohydrates
24.1 Introduction to Carbohydrates
 24.1 Introduction to Carbohydrates Carbohydrates (sugars) are abundant in nature: They are high energy biomolecules. They provide structural rigidity for organisms (plants, crustaceans, etc.). The polymer
24.1 Introduction to Carbohydrates Carbohydrates (sugars) are abundant in nature: They are high energy biomolecules. They provide structural rigidity for organisms (plants, crustaceans, etc.). The polymer
Dr. Nafith Abu Tarboush. Rana N. Talj
 2 Dr. Nafith Abu Tarboush June 19 th 2013 Rana N. Talj Review: Fischer suggested a projection in which the horizontal bonds are projecting towards the viewer and the vertical ones project away from the
2 Dr. Nafith Abu Tarboush June 19 th 2013 Rana N. Talj Review: Fischer suggested a projection in which the horizontal bonds are projecting towards the viewer and the vertical ones project away from the
Carbohydrates. Dr. Mamoun Ahram Summer,
 Carbohydrates Dr. Mamoun Ahram Summer, 2017-2018 Resource This lecture Campbell and Farrell s Biochemistry, Chapter 16 What are they? Carbohydrates are polyhydroxy aldehydes or ketones Saccharide is another
Carbohydrates Dr. Mamoun Ahram Summer, 2017-2018 Resource This lecture Campbell and Farrell s Biochemistry, Chapter 16 What are they? Carbohydrates are polyhydroxy aldehydes or ketones Saccharide is another
Carbohydrates. Learning Objective
 , one of the four major classes of biomolecules, are aldehyde or ketone compounds with multiple hydroxyl groups. They function as energy stores, metabolic intermediates and important fuels for the body.
, one of the four major classes of biomolecules, are aldehyde or ketone compounds with multiple hydroxyl groups. They function as energy stores, metabolic intermediates and important fuels for the body.
Carbohydrates. Chapter 18
 Carbohydrates Chapter 18 Biochemistry an overview Biochemistry is the study of chemical substances in living organisms and the chemical interactions of these substances with each other. Biochemical substances
Carbohydrates Chapter 18 Biochemistry an overview Biochemistry is the study of chemical substances in living organisms and the chemical interactions of these substances with each other. Biochemical substances
Dr. Basima Sadiq Ahmed PhD. Clinical biochemist
 Dr. Basima Sadiq Ahmed PhD. Clinical biochemist MEDICAL AND BIOLOGICAL IMPORTANCE 1. major source of energy for man. e.g, glucose is used in the human body for energy production. 2. serve as reserve food
Dr. Basima Sadiq Ahmed PhD. Clinical biochemist MEDICAL AND BIOLOGICAL IMPORTANCE 1. major source of energy for man. e.g, glucose is used in the human body for energy production. 2. serve as reserve food
BCH 4053 Spring 2001 Chapter 7 Lecture Notes
 BC 4053 Spring 2001 Chapter 7 Lecture Notes 1 Chapter 7 Carbohydrates 2 Carbohydrates: Nomenclature ydrates of carbon General formula (C 2 ) n (simple sugars) or C x ( 2 0) y Monosaccharides (simple sugars)
BC 4053 Spring 2001 Chapter 7 Lecture Notes 1 Chapter 7 Carbohydrates 2 Carbohydrates: Nomenclature ydrates of carbon General formula (C 2 ) n (simple sugars) or C x ( 2 0) y Monosaccharides (simple sugars)
CARBOHYDRATES (SUGARS)
 ARBYDRATES (SUGARS) ARBYDRATES: 1. Most Abundant Molecules on Earth: (100 MILLIN METRI TNS f 2 And 2 0 onverted To ellulose and ther Plant Products/Year) 2. FUNTINS: Diet, Energy, Structural, Signalling
ARBYDRATES (SUGARS) ARBYDRATES: 1. Most Abundant Molecules on Earth: (100 MILLIN METRI TNS f 2 And 2 0 onverted To ellulose and ther Plant Products/Year) 2. FUNTINS: Diet, Energy, Structural, Signalling
Chapter 22 Carbohydrates
 Chapter 22 Carbohydrates Introduction Classification of Carbohydrates Carbohydrates have the general formula C x (H 2 O) y Carbohydrates are defined as polyhydroxy aldehydes or ketones or substances that
Chapter 22 Carbohydrates Introduction Classification of Carbohydrates Carbohydrates have the general formula C x (H 2 O) y Carbohydrates are defined as polyhydroxy aldehydes or ketones or substances that
UNIT 4. CARBOHYDRATES
 UNIT 4. CARBOHYDRATES OUTLINE 4.1. Introduction. 4.2. Classification. 4.3. Monosaccharides. Classification. Stereoisomers. Cyclic structures. Reducing sugars. Sugar derivatives 4.4. Oligosaccharides. Disaccharides.
UNIT 4. CARBOHYDRATES OUTLINE 4.1. Introduction. 4.2. Classification. 4.3. Monosaccharides. Classification. Stereoisomers. Cyclic structures. Reducing sugars. Sugar derivatives 4.4. Oligosaccharides. Disaccharides.
MahaAbuAjamieh. BahaaNajjar. MamoonAhram
 7 MahaAbuAjamieh BahaaNajjar MamoonAhram Carbohydrates (saccharides) can be classified into these main categories: 1. Monosaccharides, they are simplesugars (the simplest units), such as glucose, galactose
7 MahaAbuAjamieh BahaaNajjar MamoonAhram Carbohydrates (saccharides) can be classified into these main categories: 1. Monosaccharides, they are simplesugars (the simplest units), such as glucose, galactose
among the most important organic compounds in the living organisms;
 CARBOHYDRATES Elena Rivneac PhD, Associate Professor Department of Biochemistry and Clinical Biochemistry State University of Medicine and Pharmacy "Nicolae Testemitanu" CARBOHYDRATESare among the most
CARBOHYDRATES Elena Rivneac PhD, Associate Professor Department of Biochemistry and Clinical Biochemistry State University of Medicine and Pharmacy "Nicolae Testemitanu" CARBOHYDRATESare among the most
Chapter 18. Carbohydrates with an Introduction to Biochemistry. Carbohydrates with an Introduction to Biochemistry page 1
 Chapter 18 Carbohydrates with an Introduction to Biochemistry Carbohydrates with an Introduction to Biochemistry page 1 Introduction to Proteins, Carbohydrates, Lipids, and Bioenergetics Metabolism and
Chapter 18 Carbohydrates with an Introduction to Biochemistry Carbohydrates with an Introduction to Biochemistry page 1 Introduction to Proteins, Carbohydrates, Lipids, and Bioenergetics Metabolism and
Carbohydrates 1. Steven E. Massey, Ph.D. Assistant Professor Bioinformatics Department of Biology University of Puerto Rico Río Piedras
 Carbohydrates 1 Steven E. Massey, Ph.D. Assistant Professor Bioinformatics Department of Biology University of Puerto Rico Río Piedras Office & Lab: NCN#343B Tel: 787-764-0000 ext. 7798 E-mail: stevenemassey@gmail.com
Carbohydrates 1 Steven E. Massey, Ph.D. Assistant Professor Bioinformatics Department of Biology University of Puerto Rico Río Piedras Office & Lab: NCN#343B Tel: 787-764-0000 ext. 7798 E-mail: stevenemassey@gmail.com
Chapter 20 Carbohydrates Chapter 20
 Chapter 20 Carbohydrates Chapter 20 1 Carbohydrates Carbohydrate: A polyhydroxyaldehyde or polyhydroxyketone, or a substance that gives these compounds on hydrolysis. Monosaccharide: A carbohydrate that
Chapter 20 Carbohydrates Chapter 20 1 Carbohydrates Carbohydrate: A polyhydroxyaldehyde or polyhydroxyketone, or a substance that gives these compounds on hydrolysis. Monosaccharide: A carbohydrate that
Questions- Carbohydrates. A. The following structure is D-sorbose. (Questions 1 7) CH 2 OH C = O H C OH HO C H H C OH
 Questions- Carbohydrates A. The following structure is D-sorbose. (Questions 1 7) CH 2 C = O H C HO C H H C CH 2 1. 2. 3. 4. 5. Which characteristic is different when comparing the open-chain forms of
Questions- Carbohydrates A. The following structure is D-sorbose. (Questions 1 7) CH 2 C = O H C HO C H H C CH 2 1. 2. 3. 4. 5. Which characteristic is different when comparing the open-chain forms of
CLASS 11th. Biomolecules
 CLASS 11th 01. Carbohydrates These are the compound of carbon, hydrogen and oxygen having hydrogen and oxygen in the same ratio as that of water, i.e. 2 : 1. They are among the most widely distributed
CLASS 11th 01. Carbohydrates These are the compound of carbon, hydrogen and oxygen having hydrogen and oxygen in the same ratio as that of water, i.e. 2 : 1. They are among the most widely distributed
Chapter-8 Saccharide Chemistry
 Chapter-8 Saccharide Chemistry Page 217-228 Carbohydrates (Saccharides) are most abundant biological molecule, riginally produced through C 2 fixation during photosynthesis I (C 2 ) n or - C - I where
Chapter-8 Saccharide Chemistry Page 217-228 Carbohydrates (Saccharides) are most abundant biological molecule, riginally produced through C 2 fixation during photosynthesis I (C 2 ) n or - C - I where
Chapter 23 Carbohydrates and Nucleic Acids. Carbohydrates
 Chapter 23 Carbohydrates and Nucleic Acids Carbohydrates Synthesized by plants using sunlight to convert CO 2 and H 2 O to glucose and O 2. Polymers include starch and cellulose. Starch is storage unit
Chapter 23 Carbohydrates and Nucleic Acids Carbohydrates Synthesized by plants using sunlight to convert CO 2 and H 2 O to glucose and O 2. Polymers include starch and cellulose. Starch is storage unit
Carbohydrates CHAPTER SUMMARY
 14 2 cellulose 2 2 arbohydrates 2 amylose APTER SUMMARY 14.1 hemical Nature of arbohydrates - Polyhydroxy Aldehydes and Ketones arbohydrates are a class of organic biopolymers which consist of polyhydroxy
14 2 cellulose 2 2 arbohydrates 2 amylose APTER SUMMARY 14.1 hemical Nature of arbohydrates - Polyhydroxy Aldehydes and Ketones arbohydrates are a class of organic biopolymers which consist of polyhydroxy
Carbohydrates. Monosaccharides
 Carbohydrates Carbohydrates (also called saccharides) are molecular compounds made from just three elements: carbon, hydrogen and oxygen. Monosaccharides (e.g. glucose) and disaccharides (e.g. sucrose)
Carbohydrates Carbohydrates (also called saccharides) are molecular compounds made from just three elements: carbon, hydrogen and oxygen. Monosaccharides (e.g. glucose) and disaccharides (e.g. sucrose)
Polymers: large molecules made up of repeating smaller units (monomer) peptides and proteins (Chapter 25) nucleic acids (Chapter 26)
 Chapter 23: Carbohydrates hydrates of carbon: general formula C n (H 2 O) n Plants: photosynthesis 6 CO 2 + 6 H 2 O hν C 6 H 12 O 6 + 6 O 2 Polymers: large molecules made up of repeating smaller units
Chapter 23: Carbohydrates hydrates of carbon: general formula C n (H 2 O) n Plants: photosynthesis 6 CO 2 + 6 H 2 O hν C 6 H 12 O 6 + 6 O 2 Polymers: large molecules made up of repeating smaller units
Chem 263 Nov 22, Carbohydrates (also known as sugars or saccharides) See Handout
 hem 263 Nov 22, 2016 arbohydrates (also known as sugars or saccharides) See andout Approximately 0.02% of the sun s energy is used on this planet for photosynthesis in which organisms convert carbon dioxide
hem 263 Nov 22, 2016 arbohydrates (also known as sugars or saccharides) See andout Approximately 0.02% of the sun s energy is used on this planet for photosynthesis in which organisms convert carbon dioxide
Carbohydrate Structure
 IN THE NAME OF GOD Carbohydrate Structure Disaccharides Simple Carbs Sucrose (glucose & fructose) Cookies, candy, cake, soft drinks Maltose (glucose & glucose) Beans Lactose (glucose & galactose) Yogurt,
IN THE NAME OF GOD Carbohydrate Structure Disaccharides Simple Carbs Sucrose (glucose & fructose) Cookies, candy, cake, soft drinks Maltose (glucose & glucose) Beans Lactose (glucose & galactose) Yogurt,
Farah Al-Khaled. Razi Kittaneh. Mohammad Omari
 7 Farah Al-Khaled Razi Kittaneh Mohammad Omari Dr. Mamoun Ahram In this lecture we are going to talk about modified sugars. Remember: The Fischer projection can be turned into a ring structure (which is
7 Farah Al-Khaled Razi Kittaneh Mohammad Omari Dr. Mamoun Ahram In this lecture we are going to talk about modified sugars. Remember: The Fischer projection can be turned into a ring structure (which is
Topic 4 - #2 Carbohydrates Topic 2
 Topic 4 - #2 Carbohydrates Topic 2 Biologically Important Monosaccharide Derivatives There are a large number of monosaccharide derivatives. A variety of chemical and enzymatic reactions produce these
Topic 4 - #2 Carbohydrates Topic 2 Biologically Important Monosaccharide Derivatives There are a large number of monosaccharide derivatives. A variety of chemical and enzymatic reactions produce these
Carbohydrates hydrates of carbon: general formula C n (H 2 O) n. Polymers: large molecules made up of repeating smaller units (monomer)
 Carbohydrates hydrates of carbon: general formula C n ( ) n Plants: photosynthesis hν 6 C + 6 C 6 6 + 6 Polymers: large molecules made up of repeating smaller units (monomer) Biopolymers: carbohydrates
Carbohydrates hydrates of carbon: general formula C n ( ) n Plants: photosynthesis hν 6 C + 6 C 6 6 + 6 Polymers: large molecules made up of repeating smaller units (monomer) Biopolymers: carbohydrates
Long time ago, people who sacrifice their sleep, family, food, laughter, and other joys of life were called SAINTS. But now, they are called STUDENTS!
 Long time ago, people who sacrifice their sleep, family, food, laughter, and other joys of life were called SAINTS. But now, they are called STUDENTS! Monosaccharaides Q. Can hydrolysis occur at anytime
Long time ago, people who sacrifice their sleep, family, food, laughter, and other joys of life were called SAINTS. But now, they are called STUDENTS! Monosaccharaides Q. Can hydrolysis occur at anytime
Dr. Entedhar Carbohydrates Carbohydrates are carbon compounds that have aldehyde (C-H=0) or ketone (C=O) moiety and comprises polyhyroxyl alcohol
 Dr. Entedhar Carbohydrates Carbohydrates are carbon compounds that have aldehyde (C-H=0) or ketone (C=O) moiety and comprises polyhyroxyl alcohol (polyhydroxyaldehyde or polyhyroxyketone); their polymers,which
Dr. Entedhar Carbohydrates Carbohydrates are carbon compounds that have aldehyde (C-H=0) or ketone (C=O) moiety and comprises polyhyroxyl alcohol (polyhydroxyaldehyde or polyhyroxyketone); their polymers,which
HW #9: 21.36, 21.52, 21.54, 21.56, 21.62, 21.68, 21.70, 21.76, 21.82, 21.88, 21.94, Carbohydrates
 Chemistry 131 Lectures 16 & 17: Carbohydrates Chapter 21 in McMurry, Ballantine, et. al. 7 th edition 05/24/18, 05/25/18 W #9: 21.36, 21.52, 21.54, 21.56, 21.62, 21.68, 21.70, 21.76, 21.82, 21.88, 21.94,
Chemistry 131 Lectures 16 & 17: Carbohydrates Chapter 21 in McMurry, Ballantine, et. al. 7 th edition 05/24/18, 05/25/18 W #9: 21.36, 21.52, 21.54, 21.56, 21.62, 21.68, 21.70, 21.76, 21.82, 21.88, 21.94,
Biochemistry: A Short Course
 Tymoczko Berg Stryer Biochemistry: A Short Course Second Edition CHAPTER 10 Carbohydrates 2013 W. H. Freeman and Company Chapter 10 Outline Monosaccharides are aldehydes or ketones that contain two or
Tymoczko Berg Stryer Biochemistry: A Short Course Second Edition CHAPTER 10 Carbohydrates 2013 W. H. Freeman and Company Chapter 10 Outline Monosaccharides are aldehydes or ketones that contain two or
Chapter 7 Overview. Carbohydrates
 Chapter 7 Overview n Carbohydrates main ingredient for energy production Most abundant biomolecule in nature Direct link between solar energy & chemical energy Glucose our main energy source Carbohydrates
Chapter 7 Overview n Carbohydrates main ingredient for energy production Most abundant biomolecule in nature Direct link between solar energy & chemical energy Glucose our main energy source Carbohydrates
CHAPTER 27 CARBOHYDRATES SOLUTIONS TO REVIEW QUESTIONS
 27 09/17/2013 11:12:35 Page 397 APTER 27 ARBYDRATES SLUTINS T REVIEW QUESTINS 1. In general, the carbohydrate carbon oxidation state determines the carbon s metabolic energy content. The more oxidized
27 09/17/2013 11:12:35 Page 397 APTER 27 ARBYDRATES SLUTINS T REVIEW QUESTINS 1. In general, the carbohydrate carbon oxidation state determines the carbon s metabolic energy content. The more oxidized
Carbohydrates. Green plants turn H 2 O, CO 2, and sunlight into carbohydrates.
 Chapter 27 Carbohydrates Green plants turn 2 O, CO 2, and sunlight into carbohydrates. Introduction to General, Organic, and Biochemistry, 10e John Wiley & Sons, Inc Morris ein, Scott Pattison, and Susan
Chapter 27 Carbohydrates Green plants turn 2 O, CO 2, and sunlight into carbohydrates. Introduction to General, Organic, and Biochemistry, 10e John Wiley & Sons, Inc Morris ein, Scott Pattison, and Susan
STRUCTURE OF MONOSACCHARIDES
 Lecture: 2 OCCURRENCE AND STRUCTURE OF MONOSACCHARIDES The simplest monosaccharide that possesses a hydroxyl group and a carbonyl group with an asymmetric carbon atom is the aldotriose -glyceraldehyde.
Lecture: 2 OCCURRENCE AND STRUCTURE OF MONOSACCHARIDES The simplest monosaccharide that possesses a hydroxyl group and a carbonyl group with an asymmetric carbon atom is the aldotriose -glyceraldehyde.
2/25/2015. Chapter 6. Carbohydrates. Outline. 6.1 Classes of Carbohydrates. 6.1 Classes of Carbohydrates. 6.1 Classes of Carbohydrates
 Lecture Presentation Chapter 6 Carbohydrates Julie Klare Fortis College Smyrna, GA Outline 6.7 Carbohydrates and Blood The simplest carbohydrates are monosaccharides (mono is Greek for one, sakkhari is
Lecture Presentation Chapter 6 Carbohydrates Julie Klare Fortis College Smyrna, GA Outline 6.7 Carbohydrates and Blood The simplest carbohydrates are monosaccharides (mono is Greek for one, sakkhari is
Chapter 27 Carbohydrates
 Chapter 27 Carbohydrates Green plants turn 2 O, CO 2, and sunlight into carbohydrates. Introduction to General, Organic, and Biochemistry, 10e John Wiley & Sons, Inc Morris ein, Scott Pattison, and Susan
Chapter 27 Carbohydrates Green plants turn 2 O, CO 2, and sunlight into carbohydrates. Introduction to General, Organic, and Biochemistry, 10e John Wiley & Sons, Inc Morris ein, Scott Pattison, and Susan
I. Carbohydrates Overview A. Carbohydrates are a class of biomolecules which have a variety of functions. 1. energy
 Chapter 22 Carbohydrates Chem 306 Roper I. Carbohydrates Overview A. Carbohydrates are a class of biomolecules which have a variety of functions. 1. energy 2. energy storage 3. structure 4. other functions!
Chapter 22 Carbohydrates Chem 306 Roper I. Carbohydrates Overview A. Carbohydrates are a class of biomolecules which have a variety of functions. 1. energy 2. energy storage 3. structure 4. other functions!
Nafith Abu Tarboush DDS, MSc, PhD
 Nafith Abu Tarboush DDS, MSc, PhD natarboush@ju.edu.jo www.facebook.com/natarboush Two major goals: 1. Monosaccharides: to recognize their structure, properties, & their stereochemistry 2. The nature of
Nafith Abu Tarboush DDS, MSc, PhD natarboush@ju.edu.jo www.facebook.com/natarboush Two major goals: 1. Monosaccharides: to recognize their structure, properties, & their stereochemistry 2. The nature of
Welcome to Class 7. Class 7: Outline and Objectives. Introductory Biochemistry
 Welcome to Class 7 Introductory Biochemistry Class 7: Outline and Objectives l Monosaccharides l Aldoses, ketoses; hemiacetals; epimers l Pyranoses, furanoses l Mutarotation, anomers l Disaccharides and
Welcome to Class 7 Introductory Biochemistry Class 7: Outline and Objectives l Monosaccharides l Aldoses, ketoses; hemiacetals; epimers l Pyranoses, furanoses l Mutarotation, anomers l Disaccharides and
Introduction to Carbohydrates
 Introduction to Carbohydrates 1. A six-carbon aldose has four chiral centers as follows: 2R, 3R, 4S, and 5R. A. Draw the sugar in linear and cyclic form B. Draw the form that would predominate in solution.
Introduction to Carbohydrates 1. A six-carbon aldose has four chiral centers as follows: 2R, 3R, 4S, and 5R. A. Draw the sugar in linear and cyclic form B. Draw the form that would predominate in solution.
Chapter 24: Carbohydrates
 Chapter 24: Carbohydrates [Sections: 24.1 24.10] 1. Carbohydrates definition naturally occuring compounds derived from carbon, oxygen and hydrogen the net molecular formula comes from each carbon having
Chapter 24: Carbohydrates [Sections: 24.1 24.10] 1. Carbohydrates definition naturally occuring compounds derived from carbon, oxygen and hydrogen the net molecular formula comes from each carbon having
Carbohydrates. What are they? What do cells do with carbs? Where do carbs come from? O) n. Formula = (CH 2
 Carbohydrates What are they? Formula = (C 2 O) n where n > 3 Also called sugar Major biomolecule in body What do cells do with carbs? Oxidize them for energy Store them to oxidize later for energy Use
Carbohydrates What are they? Formula = (C 2 O) n where n > 3 Also called sugar Major biomolecule in body What do cells do with carbs? Oxidize them for energy Store them to oxidize later for energy Use
Chapter 23: Carbohydrates hydrates of carbon: general formula C n (H 2 O) n. Polymers: large molecules made up of repeating smaller units (monomer)
 Chapter : Carbohydrates hydrates of carbon: general formula C n ( ) n Plants: photosynthesis hν C + C + Polymers: large molecules made up of repeating smaller units (monomer) Biopolymers: Monomer units:
Chapter : Carbohydrates hydrates of carbon: general formula C n ( ) n Plants: photosynthesis hν C + C + Polymers: large molecules made up of repeating smaller units (monomer) Biopolymers: Monomer units:
Chapter 11. Learning objectives: Structure and function of monosaccharides, polysaccharide, glycoproteins lectins.
 Chapter 11 Learning objectives: Structure and function of monosaccharides, polysaccharide, glycoproteins lectins. Carbohydrates Fuels Structural components Coating of cells Part of extracellular matrix
Chapter 11 Learning objectives: Structure and function of monosaccharides, polysaccharide, glycoproteins lectins. Carbohydrates Fuels Structural components Coating of cells Part of extracellular matrix
Anomeric carbon Erythritol is achiral because of a mirror plane in the molecule and therefore, the product is optically inactive.
 APTER 22 Practice Exercises 22.1 2 2 2 2 2 2 2 2 D-Ribulose L-Ribulose D-Xyulose L-Xyulose (one pair of enantiomers) (a second pair of enantiomers) 22.3 2 Anomeric carbon Glycosidic bond 3 () Methyl -D-mannopyranoside
APTER 22 Practice Exercises 22.1 2 2 2 2 2 2 2 2 D-Ribulose L-Ribulose D-Xyulose L-Xyulose (one pair of enantiomers) (a second pair of enantiomers) 22.3 2 Anomeric carbon Glycosidic bond 3 () Methyl -D-mannopyranoside
CHAPTER 23. Carbohydrates
 CAPTER 23 Carbohydrates 1 Introduction Carbohydrates are naturally occurring compounds of carbon, hydrogen, and oxygen. Carbohydrates have the empirical formula C 2. Carbohydrates have the general formula
CAPTER 23 Carbohydrates 1 Introduction Carbohydrates are naturally occurring compounds of carbon, hydrogen, and oxygen. Carbohydrates have the empirical formula C 2. Carbohydrates have the general formula
I (CH 2 O) n or H - C - OH I
 V. ARBYDRATE arbohydrates (glycans) have the following basic composition: I ( ) n or - - I Many carbohydrates are soluble in water. The usual chemical test for the simpler carbohydrates is heating with
V. ARBYDRATE arbohydrates (glycans) have the following basic composition: I ( ) n or - - I Many carbohydrates are soluble in water. The usual chemical test for the simpler carbohydrates is heating with
Chemistry B11 Chapters 13 Esters, amides and carbohydrates
 Chapters 13 Esters, amides and carbohydrates Esters: esters are derived from carboxylic acids (the hydrogen atom in the carboxyl group of carboxylic acid is replaced by an alkyl group). The functional
Chapters 13 Esters, amides and carbohydrates Esters: esters are derived from carboxylic acids (the hydrogen atom in the carboxyl group of carboxylic acid is replaced by an alkyl group). The functional
Nafith Abu Tarboush DDS, MSc, PhD
 Nafith Abu Tarboush DDS, MSc, PhD natarboush@ju.edu.jo www.facebook.com/natarboush Two major goals: 1. Monosaccharides: to recognize their structure, properties, & their stereochemistry 2. The nature of
Nafith Abu Tarboush DDS, MSc, PhD natarboush@ju.edu.jo www.facebook.com/natarboush Two major goals: 1. Monosaccharides: to recognize their structure, properties, & their stereochemistry 2. The nature of
Ch13. Sugars. What biology does with monosaccharides disaccharides and polysaccharides. version 1.0
 Ch13 Sugars What biology does with monosaccharides disaccharides and polysaccharides. version 1.0 Nick DeMello, PhD. 2007-2015 Ch13 Sugars Haworth Structures Saccharides can form rings. That creates a
Ch13 Sugars What biology does with monosaccharides disaccharides and polysaccharides. version 1.0 Nick DeMello, PhD. 2007-2015 Ch13 Sugars Haworth Structures Saccharides can form rings. That creates a
BIOMOLECULES & SPECTROSCOPY TABLE OF CONTENTS S.NO. TOPIC PAGE NO. i) Carbohydrates B3. ii) Proteins & Nucleic Acids.
 BIOMOLECULES & SPECTROSCOPY TABLE OF CONTENTS S.NO. TOPIC PAGE NO. 1. Biomolecules i) Carbohydrates B3 ii) Proteins & Nucleic Acids iii) Steroids iv) Terpenes & Cartenoids B27 B61 B65 2. Spectroscopy v)
BIOMOLECULES & SPECTROSCOPY TABLE OF CONTENTS S.NO. TOPIC PAGE NO. 1. Biomolecules i) Carbohydrates B3 ii) Proteins & Nucleic Acids iii) Steroids iv) Terpenes & Cartenoids B27 B61 B65 2. Spectroscopy v)
Carbohydrates Learning Objectives
 16-1 16-2 16-3 Carbohydrates Learning bjectives 1. What Are the Structures and the Stereochemistry of Monosaccharides? 2. ow Do Monosaccharides React? 3. What are Disaccharides? 4. What Are Some Important
16-1 16-2 16-3 Carbohydrates Learning bjectives 1. What Are the Structures and the Stereochemistry of Monosaccharides? 2. ow Do Monosaccharides React? 3. What are Disaccharides? 4. What Are Some Important
Number of Carbohydrate Units
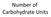 Number of Carbohydrate Units Monosaccharides = single unit Disaccharides = two units Oligiosaccharide = 3 10 units Polysaccharide = 11+ units Bonus: Can you name the most common Mono (4), Di(3), and Poly(4)
Number of Carbohydrate Units Monosaccharides = single unit Disaccharides = two units Oligiosaccharide = 3 10 units Polysaccharide = 11+ units Bonus: Can you name the most common Mono (4), Di(3), and Poly(4)
Sheet #10 Dr. Mamoun Ahram Sec 1,2,3 15/07/2014. Carbohydrates 2
 Carbohydrates 2 A study Guide: Kindly,refer to the slide number,look at the structures and read the sheet notes well,most of the slides content besides all what the doctor said are mentioned here,good
Carbohydrates 2 A study Guide: Kindly,refer to the slide number,look at the structures and read the sheet notes well,most of the slides content besides all what the doctor said are mentioned here,good
Carbohydrates. Dr. Diala Abu-Hassan, DDS, PhD All images were taken from Campbell textbook except where noted
 Carbohydrates Dr. Diala Abu-Hassan, DDS, PhD Dr.abuhassand@gmail.com McCurry et al Dr. Diala Abu-Hassan All images were taken from Campbell textbook except where noted 1 What Are Carbohydrates? Carbohydrates
Carbohydrates Dr. Diala Abu-Hassan, DDS, PhD Dr.abuhassand@gmail.com McCurry et al Dr. Diala Abu-Hassan All images were taken from Campbell textbook except where noted 1 What Are Carbohydrates? Carbohydrates
IntroducKon to Carbohydrates
 Carbohidratos IntroducKon to Carbohydrates Carbohydrates (sugars) are abundant in nature: They are high energy biomolecules. They provide structural rigidity for organisms (plants, crustaceans, etc.).
Carbohidratos IntroducKon to Carbohydrates Carbohydrates (sugars) are abundant in nature: They are high energy biomolecules. They provide structural rigidity for organisms (plants, crustaceans, etc.).
2.2: Sugars and Polysaccharides François Baneyx Department of Chemical Engineering, University of Washington
 2.2: Sugars and Polysaccharides François Baneyx Department of hemical Engineering, University of Washington baneyx@u.washington.edu arbohydrates or saccharides are abundant compounds that play regulatory
2.2: Sugars and Polysaccharides François Baneyx Department of hemical Engineering, University of Washington baneyx@u.washington.edu arbohydrates or saccharides are abundant compounds that play regulatory
Part I => CARBS and LIPIDS. 1.2 Monosaccharides 1.2a Stereochemistry 1.2b Derivatives
 Part I => CARBS and LIPIDS 1.2 Monosaccharides 1.2a Stereochemistry 1.2b Derivatives Section 1.2a: Stereochemistry Synopsis 1.2a monosaccharide (greek) sugar - Monosaccharides are carbonyl polyols (or
Part I => CARBS and LIPIDS 1.2 Monosaccharides 1.2a Stereochemistry 1.2b Derivatives Section 1.2a: Stereochemistry Synopsis 1.2a monosaccharide (greek) sugar - Monosaccharides are carbonyl polyols (or
Pharmacognosy- 1 PHG 222. Prof. Dr. Amani S. Awaad
 Pharmacognosy- 1 PHG 222 Prof. Dr. Amani S. Awaad Professor of Pharmacognosy Pharmacognosy Department, College of Pharmacy Salman Bin Abdulaziz University, Al-Kharj. KSA. Email: amaniawaad@hotmail.com
Pharmacognosy- 1 PHG 222 Prof. Dr. Amani S. Awaad Professor of Pharmacognosy Pharmacognosy Department, College of Pharmacy Salman Bin Abdulaziz University, Al-Kharj. KSA. Email: amaniawaad@hotmail.com
!"#$%&'()*+(!,-./012-,345(
 (!"#$%&'()*+(!,-./012-,345( (!"#"$%&'()$*%#+,'(-(.+/&/*+,%&(01"2+34$5( 6%#+,"(!/$75#38+(92+41( CAPTER 20: Learning Objectives:! >
(!"#$%&'()*+(!,-./012-,345( (!"#"$%&'()$*%#+,'(-(.+/&/*+,%&(01"2+34$5( 6%#+,"(!/$75#38+(92+41( CAPTER 20: Learning Objectives:! >
Organic Chemistry III
 rganic Chemistry III (Yuki Goto, Bioorganic Chemistry Lab.) rganic chemistry of biomolecules rganic chemistry of radicals 6/6 (Wed) 6/13 (Wed) 6/20 (Wed) 6/27 (Wed) 7/4 (Wed) Examples of biomolecules?
rganic Chemistry III (Yuki Goto, Bioorganic Chemistry Lab.) rganic chemistry of biomolecules rganic chemistry of radicals 6/6 (Wed) 6/13 (Wed) 6/20 (Wed) 6/27 (Wed) 7/4 (Wed) Examples of biomolecules?
For more info visit
 Carbohydrates Classification of carbohydrates: Monosaccharides: Polyhydroxy aldehydes or polyhydroxy ketones which cannot be decomposed by hydrolysis to give simpler carbohydrates.examples: Glucose, Fructose,
Carbohydrates Classification of carbohydrates: Monosaccharides: Polyhydroxy aldehydes or polyhydroxy ketones which cannot be decomposed by hydrolysis to give simpler carbohydrates.examples: Glucose, Fructose,
Chemistry 110. Bettelheim, Brown, Campbell & Farrell. Ninth Edition. Introduction to General, Organic and Biochemistry Chapter 20 Carbohydrates
 hemistry 110 Bettelheim, Brown, ampbell & Farrell Ninth Edition Introduction to General, rganic and Biochemistry hapter 20 arbohydrates Polyhydroxy Aldehydes & Ketones arbohydrates A A arbohydrate is a
hemistry 110 Bettelheim, Brown, ampbell & Farrell Ninth Edition Introduction to General, rganic and Biochemistry hapter 20 arbohydrates Polyhydroxy Aldehydes & Ketones arbohydrates A A arbohydrate is a
PAPER No. 16 Bioorganic and biophysical chemistry MODULE No.3: Sugars and polysaccharides
 Subject Chemistry Paper No and Title Module No and Title Module Tag Paper 16, Bioorganic and biophysical chemistry 3, Sugars and polysaccharides CHE_P16_M3 TABLE OF CONTENTS 1. Learning outcomes 2. Introduction
Subject Chemistry Paper No and Title Module No and Title Module Tag Paper 16, Bioorganic and biophysical chemistry 3, Sugars and polysaccharides CHE_P16_M3 TABLE OF CONTENTS 1. Learning outcomes 2. Introduction
CHAPTER 7 Carbohydrates and Glycobiology. Key topics about carbohydrates
 CHAPTER 7 Carbohydrates and Glycobiology Key topics about carbohydrates Structures and names of monosaccharides Open-chain and ring forms of monosaccharides Structures and properties of disaccharides Biological
CHAPTER 7 Carbohydrates and Glycobiology Key topics about carbohydrates Structures and names of monosaccharides Open-chain and ring forms of monosaccharides Structures and properties of disaccharides Biological
Chapter 1. Chemistry of Life - Advanced TABLE 1.2: title
 Condensation and Hydrolysis Condensation reactions are the chemical processes by which large organic compounds are synthesized from their monomeric units. Hydrolysis reactions are the reverse process.
Condensation and Hydrolysis Condensation reactions are the chemical processes by which large organic compounds are synthesized from their monomeric units. Hydrolysis reactions are the reverse process.
IntroducKon to Carbohydrates
 Carbohidratos IntroducKon to Carbohydrates Carbohydrates (sugars) are abundant in nature: They are high energy biomolecules. They provide structural rigidity for organisms (plants, crustaceans, etc.).
Carbohidratos IntroducKon to Carbohydrates Carbohydrates (sugars) are abundant in nature: They are high energy biomolecules. They provide structural rigidity for organisms (plants, crustaceans, etc.).
Disaccharides. Three Important Disaccharides Maltose, Lactose, and Sucrose. The formation of these three common disaccharides are:
 DISACCHARIDES Disaccharides Three Important Disaccharides Maltose, Lactose, and Sucrose The formation of these three common disaccharides are: 2 Disaccharides Maltose (Malt Sugar) Maltose is known as malt
DISACCHARIDES Disaccharides Three Important Disaccharides Maltose, Lactose, and Sucrose The formation of these three common disaccharides are: 2 Disaccharides Maltose (Malt Sugar) Maltose is known as malt
Carbohydrates: structure and Function. Important. 436 Notes Original slides. 438 notes Extra information
 Carbohydrates: structure and Function Important. 436 Notes Original slides. 438 notes Extra information Objectives: To understand: 1- The structure of carbohydrates of physiological significance. 2- The
Carbohydrates: structure and Function Important. 436 Notes Original slides. 438 notes Extra information Objectives: To understand: 1- The structure of carbohydrates of physiological significance. 2- The
Carbohydrates. Organic compounds which comprise of only C, H and O. C x (H 2 O) y
 Carbohydrates Organic compounds which comprise of only C, H and O C x (H 2 O) y Carbohydrates Monosaccharides Simple sugar Soluble in water Precursors in synthesis triose sugars of other (C3) molecules
Carbohydrates Organic compounds which comprise of only C, H and O C x (H 2 O) y Carbohydrates Monosaccharides Simple sugar Soluble in water Precursors in synthesis triose sugars of other (C3) molecules
Dr. Nafith Abu Tarboush. Tarek Khrisat
 1 Dr. Nafith Abu Tarboush June 18 th 2013 Tarek Khrisat 1 Lecture Outline: Aldoses and Ketoses Optical isomers -Fischer projection -Enantiomers, Diastereomers, and Epimers Isomers: Compounds with the same
1 Dr. Nafith Abu Tarboush June 18 th 2013 Tarek Khrisat 1 Lecture Outline: Aldoses and Ketoses Optical isomers -Fischer projection -Enantiomers, Diastereomers, and Epimers Isomers: Compounds with the same
Chemistry 1050 Exam 3 Study Guide
 Chapter 12 Chemistry 1050 Exam 3 Study Guide 12.1 a) Identify alkenes, alkynes and aromatics as unsaturated hydrocarbons. Determine the number of hydrogen atoms needed to complete an alkene structure.
Chapter 12 Chemistry 1050 Exam 3 Study Guide 12.1 a) Identify alkenes, alkynes and aromatics as unsaturated hydrocarbons. Determine the number of hydrogen atoms needed to complete an alkene structure.
An aldose contains an aldehyde functionality A ketose contains a ketone functionality
 RCT Chapter 7 Aldoses and Ketoses; Representative monosaccharides. (a)two trioses, an aldose and a ketose. The carbonyl group in each is shaded. An aldose contains an aldehyde functionality A ketose contains
RCT Chapter 7 Aldoses and Ketoses; Representative monosaccharides. (a)two trioses, an aldose and a ketose. The carbonyl group in each is shaded. An aldose contains an aldehyde functionality A ketose contains
Dr. Mahendra P. Bhatt (BMLT, MS-Ph.D., Post-doctorate) Associate Professor Clinical Biochemistry
 Dr. Mahendra P. Bhatt (BMLT, MS-Ph.D., Post-doctorate) Associate Professor Clinical Biochemistry mahendramlt@gmail.com Students will be able to describe: Biochemical organization of the cell Transport
Dr. Mahendra P. Bhatt (BMLT, MS-Ph.D., Post-doctorate) Associate Professor Clinical Biochemistry mahendramlt@gmail.com Students will be able to describe: Biochemical organization of the cell Transport
May 21 st, 2008 Biochemistry Recitation
 May 21 st, 2008 Biochemistry Recitation MBioS 303 Summer 2008 Outline Carbohydrate basics Aldoses vs. ketoses L and D configurations and anomers Glycosidic bonds, disaccharides Polysaccharides Storage:
May 21 st, 2008 Biochemistry Recitation MBioS 303 Summer 2008 Outline Carbohydrate basics Aldoses vs. ketoses L and D configurations and anomers Glycosidic bonds, disaccharides Polysaccharides Storage:
Chem 60 Takehome Test 2 Student Section
 Multiple choice: 1 point each. Mark only one answer for each question. 1. are composed primarily of carbon and hydrogen, but may also include oxygen, nitrogen, sulfur, phosphorus, and a few other elements.
Multiple choice: 1 point each. Mark only one answer for each question. 1. are composed primarily of carbon and hydrogen, but may also include oxygen, nitrogen, sulfur, phosphorus, and a few other elements.
Definition of a Carbohydrate
 * Atoms held together by covalent bonds Definition of a Carbohydrate * Organic macromolecules * Consist of C, H, & O atoms * Usually in a 1:2:1 ratio of C:H : O Functions Performed by Carbohydrates Used
* Atoms held together by covalent bonds Definition of a Carbohydrate * Organic macromolecules * Consist of C, H, & O atoms * Usually in a 1:2:1 ratio of C:H : O Functions Performed by Carbohydrates Used
B.sc. III Chemistry Paper b. Submited by :- Dr. Sangeeta Mehtani Associate Professor Deptt. Of Chemistry PGGCG, sec11 Chd
 B.sc. III Chemistry Paper b Submited by :- Dr. Sangeeta Mehtani Associate Professor Deptt. Of Chemistry PGGCG, sec11 Chd CARBOYDRATES Carbohydrates polyhydroxyaldehydes or polyhydroxyketones of formula
B.sc. III Chemistry Paper b Submited by :- Dr. Sangeeta Mehtani Associate Professor Deptt. Of Chemistry PGGCG, sec11 Chd CARBOYDRATES Carbohydrates polyhydroxyaldehydes or polyhydroxyketones of formula
2. Structural e.g. bacterial cell walls, cellulose. 3. Information e.g. signals on proteins and membranes.
 hapter 8 - arbohydrates ydrates of arbon: m ( 2 ) n Saccharides: Latin: Saccharum = Sugar 1. Energy transport and storage. 2. Structural e.g. bacterial cell walls, cellulose. 3. Information e.g. signals
hapter 8 - arbohydrates ydrates of arbon: m ( 2 ) n Saccharides: Latin: Saccharum = Sugar 1. Energy transport and storage. 2. Structural e.g. bacterial cell walls, cellulose. 3. Information e.g. signals
Carbohydrates are aldehyde or ketone compounds with multiple hydroxyl groups Have multiple roles in all forms of life
 Carbohydrates 1 Carbohydrates are aldehyde or ketone compounds with multiple hydroxyl groups Have multiple roles in all forms of life Classification Serve as energy stores, fuels, and metabolic intermediates
Carbohydrates 1 Carbohydrates are aldehyde or ketone compounds with multiple hydroxyl groups Have multiple roles in all forms of life Classification Serve as energy stores, fuels, and metabolic intermediates
Lecture 2 Carbohydrates
 Lecture 2 Carbohydrates Sources of CHOs Wholegrains major dietary intake Vegetables, legumes ad fruit contain dietary fibre Milk products provide lactose essential for infants Glycogen is a storage carbohydrate,
Lecture 2 Carbohydrates Sources of CHOs Wholegrains major dietary intake Vegetables, legumes ad fruit contain dietary fibre Milk products provide lactose essential for infants Glycogen is a storage carbohydrate,
Chapter 11 Lecture Notes: Carbohydrates
 Educational Goals Chapter 11 Lecture Notes: Carbohydrates 1. Given a Fischer projection of a monosaccharide, classify it as either aldoses or ketoses. 2. Given a Fischer projection of a monosaccharide,
Educational Goals Chapter 11 Lecture Notes: Carbohydrates 1. Given a Fischer projection of a monosaccharide, classify it as either aldoses or ketoses. 2. Given a Fischer projection of a monosaccharide,
Chapter 7 Carbohydrates
 Chapter 7 Carbohydrates Definition of Carbohydrates carbohydrate: hydrate of carbon ; C n ( 2 ) m Examples: glucose (C 6 12 6 or C 6 ( 2 ) 6 ), sucrose (C 12 22 11 or C 12 ( 2 ) 11 ) saccharide: simple
Chapter 7 Carbohydrates Definition of Carbohydrates carbohydrate: hydrate of carbon ; C n ( 2 ) m Examples: glucose (C 6 12 6 or C 6 ( 2 ) 6 ), sucrose (C 12 22 11 or C 12 ( 2 ) 11 ) saccharide: simple
Name LastName Student ID
 Name LastName Student ID 1) (12 points) Imidazopyridine derivatives such as 1-deaza-9H-purines (like 1) and 3- deaza-9h-purines (like 2) represent privileged structures in medicinal chemistry and they
Name LastName Student ID 1) (12 points) Imidazopyridine derivatives such as 1-deaza-9H-purines (like 1) and 3- deaza-9h-purines (like 2) represent privileged structures in medicinal chemistry and they
Chemistry 1120 Exam 2 Study Guide
 Chemistry 1120 Exam 2 Study Guide Chapter 6 6.1 Know amines are derivatives of ammonia, which is not an amine. Classify amines as primary, secondary or tertiary. Master Tutor Section 6.1 Review Section
Chemistry 1120 Exam 2 Study Guide Chapter 6 6.1 Know amines are derivatives of ammonia, which is not an amine. Classify amines as primary, secondary or tertiary. Master Tutor Section 6.1 Review Section
Basic Biochemistry. Classes of Biomolecules
 Basic Biochemistry ABE 580 Classes of Biomolecules Carbohydrates Lipids Amino Acids Nucleic Acids Other 1 Carbohydrates Sugars Composed of C, H, O (C n H 2n O n ) Biological Uses Energy source/storage
Basic Biochemistry ABE 580 Classes of Biomolecules Carbohydrates Lipids Amino Acids Nucleic Acids Other 1 Carbohydrates Sugars Composed of C, H, O (C n H 2n O n ) Biological Uses Energy source/storage
CARBOHYDRATES (H 2. Empirical formula: C x. O) y
 CARBYDRATES 1 CARBYDRATES Empirical formula: C x ( 2 ) y 2 CARBYDRATES- WERE? In solid parts of: plants, up to 80% animals, do not exceed 2% In plants: main storage material (starch) building material
CARBYDRATES 1 CARBYDRATES Empirical formula: C x ( 2 ) y 2 CARBYDRATES- WERE? In solid parts of: plants, up to 80% animals, do not exceed 2% In plants: main storage material (starch) building material
Lecture Notes Chem 51C S. King. Chapter 28 Carbohydrates. Starch, Glycogen and cellulose are all polymers of glucose.
 Lecture otes hem 51 S. King hapter 28 arbohydrates arbohydrates are the most abundant class of organic compounds in the plant world. They are synthesized by nearly all plants and animals, which use them
Lecture otes hem 51 S. King hapter 28 arbohydrates arbohydrates are the most abundant class of organic compounds in the plant world. They are synthesized by nearly all plants and animals, which use them
Carbohydrates. Haworth Projections
 Carbohydrates There are, for all intents and purposes, 2 classes of carbohydrates: pyranoses and furanoses ( ose is the suffix for a sugar). These two classes are so called because their carbon skeletons
Carbohydrates There are, for all intents and purposes, 2 classes of carbohydrates: pyranoses and furanoses ( ose is the suffix for a sugar). These two classes are so called because their carbon skeletons
Chem 263 Apr 11, 2017
 hem 263 Apr 11, 2017 arbohydrates- emiacetal Formation You know from previous lectures that carbonyl compounds react with all kinds of nucleophiles. ydration and hemiacetal formation are typical examples.
hem 263 Apr 11, 2017 arbohydrates- emiacetal Formation You know from previous lectures that carbonyl compounds react with all kinds of nucleophiles. ydration and hemiacetal formation are typical examples.
What are Carbohydrates? Aldoses and Ketoses
 What are Carbohydrates? Polyhydroxylated aldehydes and ketones Commonly called sugars General formula of common sugars!glucose: C 6 ( 2 ) 6!Glyceraldehyde: C 3 ( 2 ) 3 Talking points: C 2 ACS Division
What are Carbohydrates? Polyhydroxylated aldehydes and ketones Commonly called sugars General formula of common sugars!glucose: C 6 ( 2 ) 6!Glyceraldehyde: C 3 ( 2 ) 3 Talking points: C 2 ACS Division
You know from previous lectures that carbonyl react with all kinds of nucleophiles. Hydration and hemiacetal formation are typical examples.
 hem 263 Nov 17, 2009 D,L onfiguration of Sugars Glyceraldehyde has only one stereogenic center and therefore has two enantiomers (mirror image) forms. A D-sugar is defined as one that has configuration
hem 263 Nov 17, 2009 D,L onfiguration of Sugars Glyceraldehyde has only one stereogenic center and therefore has two enantiomers (mirror image) forms. A D-sugar is defined as one that has configuration
Carbohydrates I. Scheme 1. Carbohydrates are classified into two main classes, sugars and polysaccharides.
 Carbohydrates I 11.1 Introduction Carbohydrates are polyhydroxy aldehydes or ketones. They are primarily produced by plants and form a very large group of naturally occurring organic substances. Some common
Carbohydrates I 11.1 Introduction Carbohydrates are polyhydroxy aldehydes or ketones. They are primarily produced by plants and form a very large group of naturally occurring organic substances. Some common
Carbohydrates - General Description
 arbohydrates - General Description A. Polyhydroxy Aldehydes or Ketones ARBN AIN B. Serve a variety of functions ARBN AIN ARBN AIN 1. Energy storage (Glucose, Glycogen, Starch) 2. Structural Support (ellulose,
arbohydrates - General Description A. Polyhydroxy Aldehydes or Ketones ARBN AIN B. Serve a variety of functions ARBN AIN ARBN AIN 1. Energy storage (Glucose, Glycogen, Starch) 2. Structural Support (ellulose,
Carbohydrates are a large group of organic compounds occurring in and including,, and. They contain hydrogen and oxygen in the same ratio as (2:1).
 Carbohydrates are a large group of organic compounds occurring in and and including,, and. They contain hydrogen and oxygen in the same ratio as (2:1). Why we study carbohydrates 1) carbohydrates are the
Carbohydrates are a large group of organic compounds occurring in and and including,, and. They contain hydrogen and oxygen in the same ratio as (2:1). Why we study carbohydrates 1) carbohydrates are the
A Getting-It-On Review and Self-Test. . Carbohydrates are
 A Getting-It-n Review and Self-Test arbohydrates arbohydrates, one of the three principal classes of foods, contain only three elements: (1), (2), and (3). The name carbohydrate is derived from the French
A Getting-It-n Review and Self-Test arbohydrates arbohydrates, one of the three principal classes of foods, contain only three elements: (1), (2), and (3). The name carbohydrate is derived from the French
