The Orbitofrontal Oracle: Cortical Mechanisms for the Prediction and Evaluation of Specific Behavioral Outcomes
|
|
|
- Damon Hicks
- 6 years ago
- Views:
Transcription
1 The Orbitofrontal Oracle: Cortical Mechanisms for the Prediction and Evaluation of Specific Behavioral Outcomes Peter H. Rudebeck 1, * and Elisabeth A. Murray 2, * 1 Friedman Brain Institute, Icahn School of Medicine at Mount Sinai, One Gustave L. Levy Place, New York, NY 114, USA 2 Section on the Neurobiology of Learning and Memory, Laboratory of Neuropsychology, National Institute of Mental Health, Building 49, Suite 1B8, 49 Convent Drive, Bethesda, MD 2892, USA *Correspondence: peter.rudebeck@mssm.edu (P.H.R.), murraye@mail.nih.gov (E.A.M.) The orbitofrontal cortex (OFC) has long been associated with the flexible control of behavior and concepts such as behavioral inhibition, self-control, and emotional regulation. These ideas emphasize the suppression of behaviors and emotions, but OFC s affirmative functions have remained enigmatic. Here we review recent work that has advanced our understanding of this prefrontal area and how its functions are shaped through interaction with subcortical structures such as the amygdala. Recent findings have overturned theories emphasizing behavioral inhibition as OFC s fundamental function. Instead, new findings indicate that OFC provides predictions about specific outcomes associated with stimuli, choices, and actions, especially their moment-to-moment value based on current internal states. OFC function thereby encompasses a broad representation or model of an individual s sensory milieu and potential actions, along with their relationship to likely behavioral outcomes. Introduction Over a century ago, a team of divers discovered an ancient mechanical device in the wreck of a Roman galleon. Called the Antikythera mechanism, its function remained a mystery for decades. The use of new imaging techniques, together with a painstaking analysis of the device s inner workings, revealed that this collection of bronze gears can calculate the future locations of the earth, moon, and sun. As a result, the Antikythera mechanism accurately predicted eclipses and other celestial events (Freeth et al., 28). In ancient times, these prognostications probably contributed to planning important cultural activities, and so it is of some interest that the ancients trusted an analog computer for these prophesies, rather than their oracles, auguries, or authorities. Like the chance discovery of the Antikythera mechanism, the first knowledge about orbitofrontal cortex (OFC) function stemmed from a fortuitous event. As the most ventral part of the prefrontal cortex (PFC) (Figure 1), OFC was directly in harm s way when an accidental explosion led to the passage of an iron rod through the head of a now-famous railroad worker named Phineas Gage (Harlow, 1848). His subsequent lack of self-control, coupled with otherwise fairly normal cognitive capacities, had an enduring influence on ideas about OFC, along with PFC more generally. Based on patients like Phineas Gage and animals with experimental lesions of OFC, many theories hold that its functions primarily involve behavioral inhibition, inhibitory self-control, and emotional regulation (Dias et al., 1996; Mishkin, 1964; Roberts and Wallis, 2; Rolls, 2). Recent research has overturned this view of OFC function. These results show that OFC has a more affirmative function. Rather than primarily suppressing unwanted behaviors and emotions, this new evidence shows that OFC contributes to both by predicting the specific outcomes that should follow either sensory events or behavioral choices and representing an updated valuation of these outcomes. These signals play a particularly important role at the time of a choice, and they underlie both good choices and bad ones. Evidence from monkeys indicates that OFC encodes the predicted value of objects that might serve as goals, perhaps in an abstract common currency (Padoa-Schioppa, 211). In contrast, results from rodents emphasize specific outcomes more than a general outcome like a common currency. These findings suggest that OFC represents the sensory qualities of available outcomes that have become associated with conditioned stimuli (Schoenbaum et al., 211) and potentially maps the entire sensory milieu in terms of potentially reinforcing outcomes (Wilson et al., 214). Although delineating OFC s differences among mammalian species remains an active area of research, an emerging consensus recognizes that OFC in all mammals plays a critical role in signaling predictions about the future outcomes of behavior, including both appetitive and aversive ones. Researchers deciphered the Antikythera device through interplay between a detailed analysis of its inner workings and hypotheses about its function, and we would like to do something similar for OFC. Accordingly, this characterizes the necessary functions of OFC and then delves into its mechanisms. We discuss the information encoded by OFC neurons, the source of these signals, and how distinct parts of OFC contribute to its functions. In doing so, we draw primarily on insights gained from studies of monkeys and rodents, in part because they provide a unique opportunity to examine the causal contribution of OFC to behavior, and partly because they provide insight into its information processing at the Neuron 84, December 17, 214 ª214 Elsevier Inc. 1143
2 Posterior Medial Orbital Sulcus 14r 1o Lateral 11m 13b 14c 13a Sensory Medial OFC 11l 13m 12o single-neuron level. We pose three questions: what does OFC do, what information does OFC process, and how does OFC influence behavior? What Does OFC Do? Prediction of Specific Outcomes Our strongest evidence regarding OFC s function comes from studying animals and humans without an OFC. Observations of patients (like the aforementioned Phineas Gage) and monkeys with damage to OFC have led to the idea that OFC is important for inhibitory control, specifically for suppressing prepotent responses in the face of changing circumstances (Mishkin, 1964). This inhibitory control hypothesis was based in part on the effects of aspiration lesions of OFC on a task called object reversal learning. When humans, monkeys, cats, and rodents with OFC damage face this task, they cannot rapidly reverse their choices after changes in stimulus-reward contingencies (Butter, 1969; Chudasama and Robbins, 23; Dias et al., 1996; Mishkin, 1964; Schoenbaum et al., 23a; Teitelbaum, 1964). Over time, and with repeated findings of this kind, the deficits in object reversal learning became accepted as a pathognomonic sign of OFC damage. The inhibitory control hypothesis of OFC function has had a wide-ranging impact on neuropsychology, 12r Iam Iai AON Iapm Pir Visual Somatic Sensory Gustatory Visceral 12m 13l Ial Limbic Lateral OFC Iapl 12l G Amygdala Hypothalamus Hippocampus Figure 1. Carmichael and Price (1994) s Parcellation of the Ventral Surface of the Frontal Lobe of a Macaque Brain Based on Variation in Chemo- and Cytoarchitecture Red- and purple-shaded regions correspond to the lateral and medial subdivisions of OFC, respectively. These two subdivisions are often used in neuropsychological and neurophysiological investigations in primates to grossly subdivide OFC. The medial and lateral orbital sulci are marked by thick black lines. Thick dark arrows indicate connections, based on Carmichael and Price (1995a, 1995b). neurology, and psychiatry (Blair, 21; Clark et al., 24; Fineberg et al., 21; Roberts and Wallis, 2) and has been used to account for the extreme changes in emotional behavior that often follow OFC damage or dysfunction (Rolls, 2). The link between OFC function, reversal learning, and inhibitory control was based largely on the effects of aspiration lesions of OFC (Iversen and Mishkin, 197; Jones and Mishkin, 1972; cf. Dias et al., 1996), lesions that not only remove the cortex but also may damage fibers of passage axons coursing nearby or through OFC. In a study of macaque monkeys, we recently found that complete, excitotoxic, fiber-sparing lesions of OFC (Figures 1 and 2A) had no effect on either object reversal learning (Figure 2B) or emotional responses to artificial snakes (Rudebeck et al., 213b) (Figure 2C), both of which are severely disrupted by aspiration lesions of OFC (Butter, 1969, Butter et al., 197). This surprising result indicates that, contrary to prevailing expert opinion, OFC is not necessary for choosing objects flexibly in the face of changing stimulus-reward contingencies, at least in this classic task. Thus, the pathognomonic OFC task does not depend on OFC at all, at least not as this anatomical term refers to gray matter as opposed to white matter and fiber tracts. These new results also raise doubts about the links between OFC, emotional regulation, and behavioral flexibility for both monkeys and humans (Butter et al., 197; Damasio et al., 199; Izquierdo et al., 25). The locations of white matter bundles traveling into the frontal lobes (Croxson et al., 25; Jbabdi et al., 213) suggest that humans with damage to OFC whether through strokes, blunt force trauma, tumor excisions, or aneurisms have damage not only to the gray matter of OFC, but also to white matter traveling to frontal regions outside OFC. The close correspondence between macaque and human OFC (Petrides and Pandya, 1994) extends this conclusion to experimental lesions of OFC in monkeys. Two obvious questions follow from our new results: (1) which white matter pathways are damaged by aspiration of OFC; and (2) why do excitotoxic lesions in rodents and marmosets cause deficits in reversal learning (Chudasama and Robbins, 23; Dias et al., 1996; Izquierdo et al., 213; Schoenbaum et al., 23a)? In answer to the first question, aspiration lesions of medial OFC, alone, do not affect emotional responses to snakes (Noonan et al., 21a) or cause deficits in object reversal learning (Noonan et al., 21b). Recall that, in our recent experiment, the selective excitotoxic lesion eliminated both medial and lateral OFC (Figures 1 and 2A). Taken together, these findings suggest that white matter tracts adjacent to the lateral, but not medial, OFC might be the critical pathways. Anatomical tract-tracing studies also show that connections from the medial temporal lobe to the lateral convexity of PFC course near to the fundus of the lateral orbital sulcus, close to where cortex is typically removed by aspiration lesions of OFC (Lehman et al., 211). Alternatively, the severe impairments in reversal learning seen in the older studies might have resulted from a more widespread disconnection, one involving axons located near both medial and lateral OFC. There are a number of potential answers to the second question. One is that different species, by virtue of their different 1144 Neuron 84, December 17, 214 ª214 Elsevier Inc.
3 A C Mean latency to retrieve food (s) Intended lesion T2 MRI - 5 d post-surgery T1 MRI - ~3 yrs post-surgery CON EXC OFC EXC Neutral Objects Moving Spider Stimulus B Errors to criterion Moving Snake Reversal CON EXC OFC EXC CON ASP OFC ASP ACQ D Object difference score Object CON EXC OFC EXC Action Action difference score Figure 2. The Effect of Excitotoxic Lesions of OFC on Reward-Guided Behavior (A) Excitotoxic OFC lesions; location and extent of the intended lesion shown on drawing of a coronal section through the frontal lobe (intended lesion, top), representative case with an excitotoxic lesion of OFC (T2-weighted MRI, taken 5 days after the injection of excitotoxins, middle), and representative case with an excitotoxic lesion of OFC (T1-weighted MRI, taken 3 years after surgery, bottom). (B) Serial object reversal learning. Mean (±SEM) number of errors for unoperated controls (CON EXC and CON ASP, unfilled circles and squares, respectively), macaques with excitotoxic OFC lesions (OFC EXC, shaded triangles), and macaques with aspiration lesions of OFC (OFC ASP, shaded diamonds). Unlike monkeys with aspiration lesions of OFC, monkeys with excitotoxic lesions of OFC do not differ from controls in their performance on this task. (C) Emotional responses to neutral and fearinducing objects. Mean (±SEM) latency of unoperated controls (unfilled bars) and monkeys with excitotoxic OFC lesions (gray bar) to retrieve a desired food reward in the presence of different objects. Monkeys with excitotoxic lesions of OFC do not differ from controls. (D) Devaluation task. Mean (±SEM) difference scores for unoperated controls (unfilled bars) and macaques with excitotoxic lesions of OFC (gray bars) in the object- (left) and action-based (right) devaluation tasks. Same labels as in (C). Monkeys with excitotoxic lesions of OFC are still undergoing behavioral testing; the estimated extents of the OFC lesions as determined from postoperative T2- weighted MR scans ranged from 64% to 96% complete, and later T1-weighted (structural) MR scans are consistent with this picture. There was no correlation of the lesion extent and scores on any behavioral assessment. Adapted from Rudebeck et al. (213b) and Rhodes and Murray (213). foraging niches, solve reversal learning tasks using different strategies. This idea could account for the observation that deficits after excitotoxic lesions of OFC in rats and marmosets differ qualitatively from those caused by aspiration lesions in macaques. Specifically, deficits in rodents and marmosets are usually confined to the first few reversals (Clarke et al., 28), whereas those in macaques emerge only after a number of reversals (Izquierdo et al., 24). Alternatively, the enlargement of PFC during primate evolution (Passingham and Wise, 212; Preuss, 1995) could mean that other parts of PFC, beyond the OFC areas involved in our lesions, can guide choices in reversal learning. Once we identify the cortical areas underlying reversal learning in macaques, we should be in a position to decide among these possibilities. The results discussed so far show something about what OFC does not do, but not much about what it does. Compared with the negative results obtained for the object reversal learning (Figure 2B, triangles) and emotional regulation (Figure 2C) tasks, results from the devaluation task show that OFC plays a critical role in choosing objects based on the expected value of specific outcomes (Figure 2D, left). This impairment, alone among those observed in the tasks we used, did not depend on disrupting fiber pathways near or passing through OFC. In devaluation tasks, animals need to update the value of specific outcomes and to use that information to guide choices. Here, when we say specific outcome, we mean a representation of the constellation of sensory properties that together comprise the food or fluid that is produced by a particular choice. For a food outcome, for example, this would translate to its taste, smell, visual attributes such as color, shape, and texture, and its feel in the mouth. Importantly, whereas object reversal learning examines the ability of animals to respond flexibly to changes in stimulus-reward contingencies when food value is fixed and a single type of reward is available, devaluation tasks examine the ability of animals to respond flexibly to changes in the value of different foods, based on current internal states such as selective satiation. Put another way, reversal learning relates to the likelihood of resource availability given some choice, whereas devaluation tasks probe what a particular resource is worth at that moment. Given the finding that fiber-sparing lesions of OFC disrupted the choice between two objects based on current valuations (Figure 2D, left), we conclude that OFC is necessary for updating the value of specific object-outcome associations, not simply tracking the presence, absence, or likelihood of a reward. Of course, OFC operates in concert with other brain regions to Neuron 84, December 17, 214 ª214 Elsevier Inc. 1145
4 perform this function, including the amygdala and mediodorsal nucleus of the thalamus (Murray and Rudebeck, 213), a point taken up later. In a separate devaluation experiment, monkeys with selective excitotoxic lesions of OFC also had deficits in updating the valuation of expected outcomes in an action-outcome task (Rhodes and Murray, 213) (Figure 2D, right). In fact, these results came from some of the same monkeys tested on the object-outcome task (Figure 2D, left). This result is surprising given that previous results in both rodents (Ostlund and Balleine, 27) and macaque monkeys (Rudebeck et al., 28) suggested that OFC was only required for Pavlovian (stimulus-outcome) or object-reward processes, not action-outcome or instrumental responses. These data indicate that, in primates, OFC may be necessary for all computations where sensory-specific properties of the outcome have to be considered. On this view, OFC functions in evaluating such objects both as choice items (in the object devaluation task) and as object outcomes (e.g., food reward in both object and action devaluation tasks). Taken together, the results from devaluation studies suggest that OFC encodes a unified representation of outcomes, including their specific sensory properties and current biological value, which could enter into a variety of associations. For example, outcomes could be associated not only with objects and actions, but also with abstract concepts, behavioral rules, and strategies. Indeed, one study in macaques that employed an analog of the Wisconsin Card Sort Task implicated OFC in the selection of behavior-guiding rules after rule changes (Buckley et al., 29). Our results appear to fit with data from rodents suggesting that OFC is critical for representing the sensory qualities of specific outcomes associated with actions as well as objects (Burke et al., 28; Gremel and Costa, 213; Keiflin et al., 213) and model-based, as opposed to model-free, reinforcement learning (McDannald et al., 211, 214). Model-based learning draws on an animal s detailed knowledge of the world, in contrast to model-free reinforcement learning, which accounts for behaviors based solely on experience with stimulus-response-outcome associations. Primates and rodents diverged long ago, but we assume that as OFC evolved in both lineages the parts inherited from their common ancestor retained a conserved function and new parts (Passingham and Wise, 212; Preuss, 1995) performed related ones. As a result, we are now in a position to answer the question posed by the title of this section. Summary: What Does OFC Do? Impairments in object reversal learning (Figure 2B) and emotional responding (Figure 2C) in macaques previously ascribed to OFC appear to be due to interrupting fiber tracts running nearby (or through) OFC. Accordingly, the available evidence now argues against the idea that OFC per se plays an important role in behavioral inhibition, inhibitory self-control, or emotional regulation. Instead, OFC, acting together with the amygdala and mediodorsal thalamus, represents the specific identity of predicted outcomes and their up-to-the-moment value, taking into account an animal s or human s current state (Gottfried and Zelano, 211; Holland and Gallagher, 24; Passingham and Wise, 212; Schoenbaum et al., 29). In this way, OFC plays a crucial role in making beneficial choices among either objects or actions (Figure 2D), and perhaps concepts, rules, and strategies, as well. In addition, the outcome representations housed in OFC presumably mediate associations that promote certain Pavlovian responses (i.e., licking). What Information Does OFC Process? Signals Related to Attributes of Future Outcomes Now that we can say what the OFC does, we can explore its mechanisms more fully. Physiological recording studies have demonstrated that many neurons in OFC signal information related to reinforcers that have become associated with stimuli, choices, and actions, specifically foods, fluids, and mildly aversive outcomes. The first neurophysiological studies of OFC showed that its cells encoded stimuli that predicted impending fluid reward (Niki et al., 1972; Rosenkilde et al., 1981; Thorpe et al., 1983), and a myriad of additional outcome-related signals have been reported since then (for a review, see Abe et al., 211; Morrison and Salzman, 211; Padoa-Schioppa and Cai, 211; Pearson et al., 214; Schultz et al., 211; Simmons et al., 27; Wallis and Kennerley, 21). One way to synthesize the diverse array of signals in OFC depends on the idea that its neurons signal predicted outcomes as well as their receipt. Specifically, a substantial proportion of OFC neurons encode the potential for, sensory attributes of, and subjective value of outcomes associated with external stimuli. Other OFC neurons signal similar information when the predicted outcomes actually occur (for example, Padoa-Schioppa and Assad, 26). It is reasonable to think that the stimulus-evoked signals convey the subjective value of biologically significant outcomes because, unlike neurons in other parts of the brain, neurons in OFC distinguish between appetitive and aversive outcomes. In contrast, signals elsewhere appear to code motivational salience, which does not depend as much on whether the outcome is detrimental or beneficial (Morrison and Salzman, 29; Roesch and Olson, 24). These neurophysiological attributes, combined with connections from limbic structures and a variety of sensory modalities (Carmichael and Price, 1995a, 1995b; Ghashghaei et al., 27) (Figure 1), place OFC in a unique position. Indeed, OFC is one of the few places in the brain where visual, gustatory, olfactory, and visceral sensory inputs converge (Price, 25). Thus, neurons in this part of PFC have access to the information essential for evaluating and choosing advantageously among options. Functional brain imaging (fmri) investigations of humans similarly suggest that OFC signals outcome-related attributes (Gottfried and Zelano, 211; O Doherty, 27). One difference between human and animal studies is that the former rarely use primary reinforcement, such as fluid reward or chocolate (cf. Small et al., 21), whereas studies involving animals almost always do. Instead, human participants usually play games or other tasks to earn money or other secondary reinforcers. This difference complicates the comparison of results from different species and raises the possibility that separable parts of OFC encode primary and secondary reinforcement. Two neuroimaging studies indicate that primary and secondary reinforcers may be encoded by separate parts of OFC in humans. A posterior to anterior dissociation in signaling primary and secondary reinforcement, respectively, was first reported 1146 Neuron 84, December 17, 214 ª214 Elsevier Inc.
5 A B C Reward specific (different stimulus - same outcome) z=-2 Stimulus-reward specific (same stimulus - same outcome) z=-1 Figure 3. Encoding of Primary and Secondary Reinforcers in OFC (A) Two stimuli were paired arbitrarily with each of two different foods, equally valued by participants. Stimuli appeared sequentially for 7 ms separated by a 4 ms intertrial interval. (B) Brain regions showing a decrement in BOLD response (green-blue shading) following presentation of two different stimuli that predicted the same food reward (different stimulus, same outcome). This measure is thought to represent primary reinforcers. (C) Brain regions showing a decrement in BOLD response (red-yellow shading) following the sequential presentation of the same stimulusreward pairing (same stimulus, same outcome). This measure is thought to represent secondary reinforcers. Z coordinates denote dorsal-ventral level. Adapted from Klein-Flügge et al. (213). in an experiment comparing OFC activation for sexually explicit images and money (Sescousse et al., 21). The images used in that study were more complicated than suggested by terms like primary and secondary reinforcers, however, and many of them probably operated as both. A recent study provided clearer evidence on this issue (Klein- Flügge et al., 213). Using an experimental design that relied on repetition-suppression effects to map reward-related representations in OFC, the authors showed that representations in anterior OFC are specific to secondary reinforcers, whereas those in the most posterior part of OFC are specific to primary reinforcers. Subjects were taught that two pairs of stimuli predicted specific but different outcomes. For instance, two stimuli predicted strawberries and two other stimuli predicted potato crisps (Figure 3A). To unmask parts of OFC that encoded primary reward, such as strawberries, the authors looked for areas showing a decrease in activation when a different stimulus predicted the same primary reinforcement as on the previous trial (i.e., different stimulus, same outcome, Figure 3B). In this condition, parts of posterior OFC as well as agranular insular cortex showed reduced activation. For secondary reinforcement, the authors searched for decreased activations after repeated presentations of the same stimulus-outcome combinations (i.e., same stimulus, same outcome, Figure 3C). In this condition, parts of anterior OFC showed reduced activation. Thus, primary and secondary reinforcers appear to be signaled in different parts of the human OFC, especially for distinct stimuli associated with specific reinforcers. Future research on nonhuman primates could determine whether these signals contribute causally to goal selection and outcome evaluation. In addition, fmri studies in humans could provide additional detail by investigating the generality of the anterior-posterior dissociation, and whether different types of primary and secondary reinforcers map onto distinct portions of OFC. Outcome Signals in OFC: Beyond Sensory Attributes and Subjective Value Although some neurons in OFC signal the identity and subjective value of reward (Morrison and Salzman, 29; Tremblay and Schultz, 1999), OFC neurons might represent other aspects of reward, as well. For example, OFC is thought to play a role in the hedonic experience of reward through its role in valuation (as reviewed by Kringelbach, 25). If so, then OFC neurons might signal reward irrespective of its context, attributes, or meaning, somewhat similar to theories suggesting that the OFC is important for computing general economic value (Padoa- Schioppa, 27). A recent series of experiments by Tsujimoto and colleagues set out to tackle this question. Their results indicate that a proportion of neural responses in OFC convey the sensory or informational aspects of reward, rather than being directly related to either their identity or hedonic aspects (Tsujimoto et al., 211, 212). Macaques were trained to implement different behavioral response strategies based on instructional cues. In one condition, the orientation of a bar presented on a monitor screen instructed macaques whether to make the same response as on the previous trial or to make a different one (i.e., stay or switch). In another, the delivery of one drop or two half-drops of fluid served as the instruction cues, again indicating whether to stay or switch (Figure 4A). Correct performance in either condition was rewarded with the same fluid reward, delivered in the same way. On trials in which fluid served both as an instructional cue and a reward, it was possible to determine whether neurons in OFC encoded reward for either its instructional meaning, as feedback for a correct response, or irrespective of its meaning. If OFC neurons always had the latter property, it would indicate that this cortical area signals reward for its pleasurable or hedonic aspects. A proportion of the neurons in OFC only encoded the reward when it was presented as an instruction cue. These same neurons did not respond to the reward delivered for correctly performing the instructed strategy (cue only, Figures 4B and 4C). A separate population of OFC neurons signaled reward when it was delivered for a correctly performed trial, and yet another population signaled reward irrespective of when they were delivered (both and feedback only, Figure 4C). Within OFC, roughly equally sized populations of neurons about 15% of the task-related sample responded to reward in these three different categories (Figure 4C). Interestingly, a similar pattern of results was obtained in dorsolateral PFC, but the cortex of the frontal pole differed in that it only signaled reward as feedback (Figure 4C). The presence of separable populations of OFC neurons signaling the occurrence of fluid reward delivery in these different Neuron 84, December 17, 214 ª214 Elsevier Inc. 1147
6 A Task periods Fixation Instruction Shift instruction Feedback Previous goal: Left Delay Pre-feedback Post-feedback B OFC LOS MOS lateral rostral C Percentage of neurons Brain areas recorded FPC DLPFC PS rostral lateral AS 1 cm AS Responses to reward dorsal rostral PS Cue only Both Feedback only Figure 4. OFC Activity Related to Reward Delivered as Instructional Cues or Feedback (A) Trial sequence. Trials started with the presentation of an instructional cue, either a single drop of fluid or two smaller drops. These cues signaled to the monkey whether to stay with the response from the previous trial (left or right) or switch to the alternative option. Once the monkey made a saccade to one of the two potential targets, both targets became solid white, whether correctly or incorrectly performed. Successful performance led to the delivery of additional fluid reward as feedback (reinforcement). Red type indicates sequence on example trial. (B) Brain regions studied: OFC (orange on ventral view of macaque frontal lobe); frontal polar cortex (FPC, red on dorsal view of macaque frontal lobe); dorsolateral prefrontal cortex (DLPFC, green on lateral view of macaque frontal lobe). (C) Percentage of neurons in OFC, FPC, and DLPFC encoding the reward when delivered as an instructional cue (cue only), as feedback (feedback only), or both. Color scheme are as in (B). Dashed line indicates noise/chance level for cue and feedback responses. Adapted from Tsujimoto et al. (212). ways is intriguing. Some neurons in OFC clearly signal the delivery of reward irrespective of its context, potentially suggesting a role for this population in conveying the subjective value of reward or potentially the specific identity of the reward. Neurons encoding reward only as feedback could serve to reinforce or extinguish previously learned associations, a process critical for updating associational knowledge and models of the environment. There is also evidence suggesting that reward-as-feedback encoding in OFC is task specific (Luk and Wallis, 213). Finally, neurons in OFC that signal instructional as opposed to reward-related information probably encode representations in addition to the reward as instruction signals described above. Encoding of task-related signals in OFC unrelated to reinforcement, such as the specific identity of sensory stimuli or the current abstract behavior-guiding rule, have also been reported in a number of studies in humans, macaques, and rodents, but their importance has often received little emphasis (for example, Schoenbaum and Eichenbaum, 1995; Wallis et al., 21; Wallis and Miller, 23; Zelano et al., 211). Instead, studies and reviews (including our own) have concentrated on the rewardrelated attributes of OFC activity (Morrison and Salzman, 211; Rudebeck and Murray, 211a). This was in part done to link the data to neuropsychological results and theories concerning the role of OFC in emotion (Rolls, 2). The existence of nonreinforcement related signals in OFC is potentially important as it suggests a richer representation of the environment beyond reward. It adds weight to the hypothesis that OFC does not simply signal the attributes of a particular reinforcer or stimulus-reinforcer association but instead provides a map of all relevant attributes, related to reinforcement or not, needed to parse the environment (Wilson et al., 214). It is worth noting that both task-relevant and -irrelevant signals have been reported in other parts of PFC in macaques, suggesting that encoding of the whole environment maybe a property of PFC areas in general and not specific to OFC (Genovesio et al., 214; Mann et al., 1988). Despite its intuitive appeal, one issue with the idea that OFC represents all relevant attributes of the environment is that it does not readily provide an explanation of how certain sensory features become behaviorally relevant and therefore encoded by neurons in OFC. One possibility is that, in novel settings, all sensory aspects of the task are represented in OFC. Through learning, these representations are pruned until only those that have led to the desired outcomes will be encoded, an idea that has already gained some attention (Huys et al., 212). The Origin of Outcome-Related Signals Neurophysiological and neuroimaging studies indicate the diverse types of neural signals present in OFC, but they do not indicate how these signals arise. Understanding the source of these signals could prove to be important for future attempts to influence OFC activity therapeutically. OFC receives connections from parts of the limbic system as well as from many of the different sensory modalities (Figure 1; Price, 25). Of all of the areas that interact with OFC, perhaps the amygdala has received the most attention. Interaction between these two structures is thought to be central to emotion and reward-guided behaviors (Gottfried et al., 23; Holland and Gallagher, 24; Murray and Wise, 21; Salzman and Fusi, 21; Schoenbaum and Roesch, 25). Exactly how the amygdala contributes to neural signals in OFC remains unclear. The two available studies that have assessed the impact of amygdala damage on OFC outcome-related activity one in humans and one in rats offer differing perspectives, potentially due to species differences (Hampton et al., 27; Schoenbaum et al., 23b). In addition, studies that involve recording from both the OFC and amygdala in primates have emphasized opposing roles for these two structures in encoding stimulus-outcome associations for reward and punishments (Morrison et al., 211), with OFC encoding appetitive outcomes earlier than the amygdala and the opposite results for punishment. To examine the amygdala s contribution to outcome encoding, we recorded neural activity in lateral OFC (areas 11 and 1148 Neuron 84, December 17, 214 ª214 Elsevier Inc.
7 A Fixation ( s) S1 Onset (1. s) TRIAL SEQUENCE B Firing rate (sp/s) 5.8 ml.4 ml.2 ml.1 ml. ml Firing rate (sp/s) 2 1 S1 - reward S2 - reward C Percent of neurons S2 reward value S1 relative value Interaction S2 Onset (-1.5 s) 1.1 Time from S1 onset (s) 2. Go signal / Choice D OFC OFC Delay (.5 s) Percent of neurons Preoperative Postoperative Percent of neurons S1 On S2 On Reward delivered S1 S2 Reward value Time from S1 onset (s) Figure 5. The Effect of Amygdala Lesions on Reward-Value Encoding in OFC (A) Trial sequence. On each trial monkeys were sequentially presented with two stimuli (S1 and S2) associated with different amounts of reward and were instructed to choose between them. Each stimulus was associated with a different amount of fluid reward and monkeys nearly always chose the stimulus associated with the highest amount. (B) Spike density and raster plots illustrating the activity of one neuron in OFC that exhibited its highest firing rate to stimuli associated with smallest amount of reward. Each dot in the raster plot indicates the time that this neuron discharged. The color of the curves and dots in the raster plot correspond to the amount of reward associated with each stimulus, as noted in the key. Inset figures show the relationship between this neuron s firing rate and reward value, within the periods after S1 and S2 stimulus presentation, respectively. (C) The proportion of OFC neurons encoding the absolute value of S2, relative value of S1 (either higher or lower than S2), or interaction between these factors after the presentation of the second stimulus. (D) Effect of amygdala lesions. Left: proportion of OFC neurons preoperatively (blue/turquoise) and postoperatively (red/orange) encoding the reward value associated with S1 and S2. Right: time course of stimulus-reward value encoding. Green dots indicate significant reductions in reward encoding after amygdala lesions. Red bar: duration of S1 presentation. Purple bar: duration of S2 presentation. Adapted from Rudebeck et al. (213a). 13, Figure 1, red shaded area) and a restricted part of medial prefrontal cortex (MFC, dorsal bank of the cingulate sulcus, corresponding to areas 9v and 24c), of macaques while they performed a choice task for fluid reward (Figure 5A) (Rudebeck et al., 213a). Here, different amounts of fluid reward, which we take to be encoded as an attribute of the outcome, were assigned to the different stimuli available for choice. Recordings were made before and after amygdala lesions. When the amygdala was intact, similar to several previous reports (for example, Wallis and Miller, 23), we found that many neurons in both OFC and MFC signaled reward or outcome-related aspects of the task, including encoding of the value of anticipated outcomes associated with external stimuli (S1 and S2, Figure 5B), as well as the value of received outcomes (not illustrated). Neural activity associated with reward was, however, more prevalent in OFC than in the MFC. For instance, relative to MFC, more neurons in OFC signaled the amount of reward associated with the visual cues that the macaques chose, and they did so earlier. Input from the amygdala to frontal cortex was removed by making complete excitotoxic lesions of the amygdala in both hemispheres. This led to a decrease in the proportion of neurons in OFC encoding outcome-related aspects of the task (Figure 5D). It also altered the latency at which OFC neurons signaled the amount of reward associated with different options. By contrast, outcome-related encoding in MFC was largely unchanged. Thus, despite projecting to both MFC and OFC, the amygdala has a greater influence on outcome encoding in OFC. Importantly, removing input from the amygdala did not abolish outcome-related activity in OFC but merely led to an approximately one-third reduction. Based on the work of Tsujimoto and colleagues, discussed previously, it could be that amygdala input is critical for the cells that signal reward irrespective of its behavioral context, a possibility that could be tested empirically. These data also indicate that the amygdala is not the sole origin for the outcome or reward information being signaled by neurons in OFC. Although the specific task parameters and experimental Neuron 84, December 17, 214 ª214 Elsevier Inc. 1149
8 design could have attenuated the decrement that we observed for example, our monkeys were highly familiar with the choice options it seems likely that other parts of the brain play an integral role in OFC outcome signals. Hypothalamus, agranular insular cortex, inferotemporal cortex, perirhinal cortex, dopaminergic neurons in the midbrain, and parts of the thalamus all send projections to OFC (Goldman-Rakic and Porrino, 1985; Ongür et al., 1998; Saleem et al., 28; Williams and Goldman- Rakic, 1998) and each could provide information related to potential reinforcement or outcomes. Projections from the basal forebrain also target OFC (Kitt et al., 1987). Like input from the amygdala, inputs from the basal forebrain may influence outcome-related signals and, ultimately, the encoding of specific goals for action in OFC. In rats, bursting activity in basal forebrain neurons in response to behaviorally relevant stimuli is time locked to task-related changes in local field potentials (LFPs) in OFC (Nguyen and Lin, 214). In addition, electrical stimulation of the basal forebrain elicits alterations in these LFPs, suggesting that neurons in the basal forebrain directly contribute to OFC encoding of behaviorally relevant stimuli. Although input from basal forebrain neurons is unlikely to directly influence the encoding of specific goals or outcomes in OFC (Croxson et al., 211), temporally discrete signals from this region might serve to heighten the salience of specific options during evaluation and goal selection in OFC. Alternatively, it may support plasticity for behaviorally relevant associations, including stimulus-reward associations, during learning. These findings highlight the fact that many brain structures contribute to encoding specific outcomes in OFC, including the amygdala. Determining the contribution of each of OFC s inputs will undoubtedly be important for gaining a mechanistic and systems-level understanding of this area s function. We think this knowledge is important and has the potential to guide therapeutic interventions aiming to affect OFC processing. Comparison and Choice Signals When making a choice, the costs and benefits of a number of alternatives must be weighed before a final selection can be made. In some situations, the presence of alternative options can influence the valuation of the option currently under consideration (Tversky and Itamar, 1993). Given this, we would like to know whether neural signals in OFC are influenced by the relative value of alternative options presented for choice as well as the current option. If only the latter, then OFC s role in choice would be strictly evaluative. If both, then its role would include relative and evaluative functions. As already indicated, neurons in lateral OFC (Figure 1, red shaded area) encode the predicted subjective value of external stimuli (Kennerley et al., 29; Padoa-Schioppa and Assad, 26; Roesch and Olson, 25; Tremblay and Schultz, 1999). In many studies of macaque OFC in choice behavior, two or more choice options are revealed to the subject at the same time. This aspect of the experimental design makes it difficult to disentangle neuronal responses related to the valuation of each individual option. To get around this problem, a few studies have either held the value of one option stable while varying the other (Padoa-Schioppa and Assad, 28) or they have sequentially presented the options for choice, allowing option values to be dissociated temporally (Luk and Wallis, 213; Rudebeck et al., 213a; Wallis and Miller, 23). In one of these studies, we sequentially presented macaques with two rewarded options, each associated with a different amount of fluid reward (Figure 5A) (Rudebeck et al., 213a). While monkeys were performing the task, neural activity was recorded in lateral OFC. To determine whether reward-related signals in lateral OFC were influenced by the context in which they were presented, we looked at whether encoding of the second option s (S2) reward value was influenced by that of the first (S1). Although a small population of neurons in lateral OFC (about 8%) signaled the reward associated with S2 in a manner that was dependent on S1, a much larger proportion of neurons (about 25%) signaled the reward value of the S2 independently of S1 (Figure 5C). Thus, neurons in lateral OFC predominantly provide an assessment of each individual option independently of the others available. If OFC signals are critical for making choices, then variations in the activity of OFC neurons should be correlated with choice behavior. Padoa-Schioppa (213) has examined this issue for alternatives of similar value. Previously, he had characterized three different types of neurons in lateral OFC: offer value neurons that encode the value of the two offers presented, chosen value neurons that signal the value of the best or chosen option, and chosen juice neurons that signal the juice type (or juice identity) associated with the chosen option. Of these, chosen-juice neurons seem to be most specifically related to the outcome of the trial, as opposed to evaluating alternatives. Surprisingly, Padoa-Schioppa reported that slight fluctuations in either the pretrial or within-trial activity of chosen juice neurons, but not offervalue or chosen-value neurons, correlated with small changes in choice behavior. Later, we take up the finding that it is chosen-juice neurons that show this correlation. For now, we simply note that this study provides evidence that neurons in lateral OFC participate in the process of choosing among options. Evidence from human neuroimaging, macaque lesion, and neurophysiology studies suggest a different view of OFC functional organization, one that questions the role of lateral OFC in choices per se, at least for the part of choice that involves comparisons and final selections. Several studies suggest that whereas lateral parts of OFC are important for evaluating options (Rudebeck and Murray, 211b; Walton et al., 21), its more medial parts are engaged when options have to be compared and selected (Boorman et al., 29; FitzGerald et al., 29; Hunt et al., 212; Noonan et al., 21b; Rudebeck and Murray, 211b). Note that by medial OFC, we refer to the areas shaded in purple in Figure 1, and not to the areas called MFC above. The results of a recent neurophysiology study by Strait et al. (214) add weight to the idea that value comparison occurs in the medial OFC. In their study, monkeys chose between two sequentially presented choice options, each associated with different amounts of reward and different probabilities that the reward would be delivered. Neurons in medial OFC signaled the value of the two offers as they were presented using a common currency value scale to integrate reward size and probability. Notably, after the presentation of the second option, neurons encoded the value of the options using an inversely correlated encoding scheme. Opposing encoding schemes for the two 115 Neuron 84, December 17, 214 ª214 Elsevier Inc.
9 A Spikes/sec B Block switch Spikes/sec Block switch Sham control First 1 Last 1 Trial number OFC lesion Unexpected reward Reward Omission First 1 Last 1 Trial number Figure 6. The Effect of OFC Lesions on Reward-Prediction Errors in Dopaminergic Neurons of the VTA Mean (±SEM) firing rate (spikes/s) of reward responsive ventral tegmental area (VTA) neurons in sham-operated (A) and OFC lesion (B) rats following unexpected reward delivery or omission immediately (first ten trials) and later (last ten trials) after a change in the reward contingencies in the task (block switch). Without input from OFC, VTA neurons do not display the normal patterns of prediction error signaling. Dark solid lines represent responses to unexpected delivery of reward. Gray solid lines represent responses to unexpected omission of reward. Gray dashed lines represent baseline firing. Arrows highlight differences in the VTA neuronal responses in the first and last ten trials after block switches in the sham-operated and lesion rats. Adapted from Takahashi et al. (211). options indicates that the representations of the two offers may be mutually inhibiting each other. Furthermore, after the presentation of the second option, neurons rapidly transitioned to signal the value of the option that would subsequently be chosen. This type of inversely correlated encoding contrasts with the type of signals seen in lateral OFC, where options are encoded largely independent of each other (Figure 5C) and use correlated encoding schemes (P.H.R. and E.A.M., unpublished data). Although these findings are intriguing, we note that only a small proportion of neurons in medial OFC (about 1%) were found to have these properties and that neurons in other brain areas were not examined using the same methods (Strait et al., 214). In addition to providing evidence for a role for medial OFC in guiding choice, the findings of Strait et al. are consistent with the idea that there are two functionally distinct modules in OFC, one for identifying and signaling the specific characteristics of different outcomes associated with objects or actions and one for comparing these options to select a goal for action (Rudebeck and Murray, 211a; Rushworth et al., 211). How the comparison process in medial OFC influences action selection, the next step in the process of attaining ones goals, is an issue we take up later. Even if we accept the idea that medial OFC plays the largest role in choices and lateral OFC does so for evaluations, this still leaves the question of why the activity of chosen-juice neurons in lateral OFC varied with choice behavior and the activity of chosen-value neurons did not. One possibility is that the fluctuations in activity in these neurons reflect feedback coming from medial OFC about the result of the comparison process. Indeed, this explanation was proposed by Padoa-Schioppa (213) and, as he noted, would help to explain why pretrial activity was predictive of the subsequent choice. One way to test this idea could be to record the activity of chosen-juice neurons without input from medial OFC, using temporary interference methods, such as pharmacogenetic inactivation, to specifically suppress the activity of neurons in medial OFC that project to lateral OFC. Summary: What Information Does OFC Process? It is commonly accepted that OFC processes information related to reward value in some way. Although this idea has contributed importantly to understanding OFC, recent results point to the need for more specific concepts. OFC neurons signal reward in multiple ways, which depend on the level of association (Figure 3), the specific sensory properties of the reward, including its instantaneous subjective value (Figures 2D, 5B, and 5D), and the instructional information it conveys (Figure 4). These signals are not homogeneously distributed within OFC (Figure 3), which could account for some of the discrepancies in the literature. Converging lines of evidence point to a role for lateral OFC in evaluating options, fairly independently of each other, and medial OFC in comparing and contrasting them in order to make a choice. How Does OFC Influence Behavior? Outcome Signals: Dopaminergic Prediction Errors For any organism to learn, a prediction of the outcome associated with an action has to be compared to the actual outcome obtained from making that action (Sutton and Barto, 1998). The difference or prediction error can then be used to update expectations and drive learning. Dopaminergic neurons in the ventral tegmental area (VTA) and substantia nigra pars compacta are thought to provide one such prediction-error signal for reward-guided learning (Schultz et al., 1997), but where do the dopamine neurons get information about predicted outcomes? To address this issue, Takahashi and colleagues investigated whether OFC serves as a source of the prediction signal for VTA neurons (Takahashi et al., 211). Recordings were made from putative dopaminergic projection neurons in the VTA of rats with ipsilateral OFC lesions (i.e., unilateral OFC lesions in the same hemisphere as the VTA recordings) while the rats performed a behavioral task designed to generate reward-related error signals (Figure 6A). This task had previously been shown to be dependent on both OFC and VTA (Takahashi et al., Neuron 84, December 17, 214 ª214 Elsevier Inc. 1151
Effects of Amygdala Lesions on Reward-Value Coding in Orbital and Medial Prefrontal Cortex
 Article Effects of Amygdala Lesions on Reward-Value Coding in Orbital and Medial Prefrontal Cortex Peter H. Rudebeck, 1, * Andrew R. Mitz, 1 Ravi V. Chacko, 1 and Elisabeth A. Murray 1 1 Section on the
Article Effects of Amygdala Lesions on Reward-Value Coding in Orbital and Medial Prefrontal Cortex Peter H. Rudebeck, 1, * Andrew R. Mitz, 1 Ravi V. Chacko, 1 and Elisabeth A. Murray 1 1 Section on the
Decision neuroscience seeks neural models for how we identify, evaluate and choose
 VmPFC function: The value proposition Lesley K Fellows and Scott A Huettel Decision neuroscience seeks neural models for how we identify, evaluate and choose options, goals, and actions. These processes
VmPFC function: The value proposition Lesley K Fellows and Scott A Huettel Decision neuroscience seeks neural models for how we identify, evaluate and choose options, goals, and actions. These processes
Double dissociation of value computations in orbitofrontal and anterior cingulate neurons
 Supplementary Information for: Double dissociation of value computations in orbitofrontal and anterior cingulate neurons Steven W. Kennerley, Timothy E. J. Behrens & Jonathan D. Wallis Content list: Supplementary
Supplementary Information for: Double dissociation of value computations in orbitofrontal and anterior cingulate neurons Steven W. Kennerley, Timothy E. J. Behrens & Jonathan D. Wallis Content list: Supplementary
The Frontal Lobes. Anatomy of the Frontal Lobes. Anatomy of the Frontal Lobes 3/2/2011. Portrait: Losing Frontal-Lobe Functions. Readings: KW Ch.
 The Frontal Lobes Readings: KW Ch. 16 Portrait: Losing Frontal-Lobe Functions E.L. Highly organized college professor Became disorganized, showed little emotion, and began to miss deadlines Scores on intelligence
The Frontal Lobes Readings: KW Ch. 16 Portrait: Losing Frontal-Lobe Functions E.L. Highly organized college professor Became disorganized, showed little emotion, and began to miss deadlines Scores on intelligence
Amygdala and Orbitofrontal Cortex Lesions Differentially Influence Choices during Object Reversal Learning
 8338 The Journal of Neuroscience, August 13, 2008 28(33):8338 8343 Behavioral/Systems/Cognitive Amygdala and Orbitofrontal Cortex Lesions Differentially Influence Choices during Object Reversal Learning
8338 The Journal of Neuroscience, August 13, 2008 28(33):8338 8343 Behavioral/Systems/Cognitive Amygdala and Orbitofrontal Cortex Lesions Differentially Influence Choices during Object Reversal Learning
Orbitofrontal Cortex as a Cognitive Map of Task Space
 Neuron Viewpoint Orbitofrontal Cortex as a Cognitive Map of Task Space Robert C. Wilson,, * Yuji K. Takahashi, Geoffrey Schoenbaum,,3,4 and Yael Niv,4, * Department of Psychology and Neuroscience Institute,
Neuron Viewpoint Orbitofrontal Cortex as a Cognitive Map of Task Space Robert C. Wilson,, * Yuji K. Takahashi, Geoffrey Schoenbaum,,3,4 and Yael Niv,4, * Department of Psychology and Neuroscience Institute,
THE PREFRONTAL CORTEX. Connections. Dorsolateral FrontalCortex (DFPC) Inputs
 THE PREFRONTAL CORTEX Connections Dorsolateral FrontalCortex (DFPC) Inputs The DPFC receives inputs predominantly from somatosensory, visual and auditory cortical association areas in the parietal, occipital
THE PREFRONTAL CORTEX Connections Dorsolateral FrontalCortex (DFPC) Inputs The DPFC receives inputs predominantly from somatosensory, visual and auditory cortical association areas in the parietal, occipital
Supplementary materials for: Executive control processes underlying multi- item working memory
 Supplementary materials for: Executive control processes underlying multi- item working memory Antonio H. Lara & Jonathan D. Wallis Supplementary Figure 1 Supplementary Figure 1. Behavioral measures of
Supplementary materials for: Executive control processes underlying multi- item working memory Antonio H. Lara & Jonathan D. Wallis Supplementary Figure 1 Supplementary Figure 1. Behavioral measures of
Reward Systems: Human
 Reward Systems: Human 345 Reward Systems: Human M R Delgado, Rutgers University, Newark, NJ, USA ã 2009 Elsevier Ltd. All rights reserved. Introduction Rewards can be broadly defined as stimuli of positive
Reward Systems: Human 345 Reward Systems: Human M R Delgado, Rutgers University, Newark, NJ, USA ã 2009 Elsevier Ltd. All rights reserved. Introduction Rewards can be broadly defined as stimuli of positive
Neural Basis of Decision Making. Mary ET Boyle, Ph.D. Department of Cognitive Science UCSD
 Neural Basis of Decision Making Mary ET Boyle, Ph.D. Department of Cognitive Science UCSD Phineas Gage: Sept. 13, 1848 Working on the rail road Rod impaled his head. 3.5 x 1.25 13 pounds What happened
Neural Basis of Decision Making Mary ET Boyle, Ph.D. Department of Cognitive Science UCSD Phineas Gage: Sept. 13, 1848 Working on the rail road Rod impaled his head. 3.5 x 1.25 13 pounds What happened
Resistance to forgetting associated with hippocampus-mediated. reactivation during new learning
 Resistance to Forgetting 1 Resistance to forgetting associated with hippocampus-mediated reactivation during new learning Brice A. Kuhl, Arpeet T. Shah, Sarah DuBrow, & Anthony D. Wagner Resistance to
Resistance to Forgetting 1 Resistance to forgetting associated with hippocampus-mediated reactivation during new learning Brice A. Kuhl, Arpeet T. Shah, Sarah DuBrow, & Anthony D. Wagner Resistance to
Taken From The Brain Top to Bottom //
 Taken From The Brain Top to Bottom // http://thebrain.mcgill.ca/flash/d/d_03/d_03_cl/d_03_cl_que/d_03_cl_que.html THE EVOLUTIONARY LAYERS OF THE HUMAN BRAIN The first time you observe the anatomy of the
Taken From The Brain Top to Bottom // http://thebrain.mcgill.ca/flash/d/d_03/d_03_cl/d_03_cl_que/d_03_cl_que.html THE EVOLUTIONARY LAYERS OF THE HUMAN BRAIN The first time you observe the anatomy of the
25 Things To Know. Pre- frontal
 25 Things To Know Pre- frontal Frontal lobes Cognition Touch Sound Phineas Gage First indication can survive major brain trauma Lost 1+ frontal lobe Gage Working on a railroad Gage (then 25) Foreman on
25 Things To Know Pre- frontal Frontal lobes Cognition Touch Sound Phineas Gage First indication can survive major brain trauma Lost 1+ frontal lobe Gage Working on a railroad Gage (then 25) Foreman on
Supplementary Figure 1. Recording sites.
 Supplementary Figure 1 Recording sites. (a, b) Schematic of recording locations for mice used in the variable-reward task (a, n = 5) and the variable-expectation task (b, n = 5). RN, red nucleus. SNc,
Supplementary Figure 1 Recording sites. (a, b) Schematic of recording locations for mice used in the variable-reward task (a, n = 5) and the variable-expectation task (b, n = 5). RN, red nucleus. SNc,
Distinct valuation subsystems in the human brain for effort and delay
 Supplemental material for Distinct valuation subsystems in the human brain for effort and delay Charlotte Prévost, Mathias Pessiglione, Elise Météreau, Marie-Laure Cléry-Melin and Jean-Claude Dreher This
Supplemental material for Distinct valuation subsystems in the human brain for effort and delay Charlotte Prévost, Mathias Pessiglione, Elise Météreau, Marie-Laure Cléry-Melin and Jean-Claude Dreher This
Prefrontal dysfunction in drug addiction: Cause or consequence? Christa Nijnens
 Prefrontal dysfunction in drug addiction: Cause or consequence? Master Thesis Christa Nijnens September 16, 2009 University of Utrecht Rudolf Magnus Institute of Neuroscience Department of Neuroscience
Prefrontal dysfunction in drug addiction: Cause or consequence? Master Thesis Christa Nijnens September 16, 2009 University of Utrecht Rudolf Magnus Institute of Neuroscience Department of Neuroscience
Encoding of Reward and Space During a Working Memory Task in the Orbitofrontal Cortex and Anterior Cingulate Sulcus
 J Neurophysiol 12: 3352 3364, 29. First published September 23, 29; doi:1.1152/jn.273.29. Encoding of Reward and Space During a Working Memory Task in the Orbitofrontal Cortex and Anterior Cingulate Sulcus
J Neurophysiol 12: 3352 3364, 29. First published September 23, 29; doi:1.1152/jn.273.29. Encoding of Reward and Space During a Working Memory Task in the Orbitofrontal Cortex and Anterior Cingulate Sulcus
Neural Basis of Decision Making. Mary ET Boyle, Ph.D. Department of Cognitive Science UCSD
 Neural Basis of Decision Making Mary ET Boyle, Ph.D. Department of Cognitive Science UCSD Phineas Gage: Sept. 13, 1848 Working on the rail road Rod impaled his head. 3.5 x 1.25 13 pounds What happened
Neural Basis of Decision Making Mary ET Boyle, Ph.D. Department of Cognitive Science UCSD Phineas Gage: Sept. 13, 1848 Working on the rail road Rod impaled his head. 3.5 x 1.25 13 pounds What happened
correlates with social context behavioral adaptation.
 REVIEW OF FRONTAL LOBE STRUCTURES Main organization of frontal cortex: 1. Motor area (precentral gyrus). 2. Premotor & supplementary motor areas (immediately anterior to motor area). Includes premotor,
REVIEW OF FRONTAL LOBE STRUCTURES Main organization of frontal cortex: 1. Motor area (precentral gyrus). 2. Premotor & supplementary motor areas (immediately anterior to motor area). Includes premotor,
Selective Ablations Reveal That Orbital and Lateral Prefrontal Cortex Play Different Roles in Estimating Predicted Reward Value
 5878 The Journal of Neuroscience, November 24, 200 30(47):5878 5887 Behavioral/Systems/Cognitive Selective Ablations Reveal That Orbital and Lateral Prefrontal Cortex Play Different Roles in Estimating
5878 The Journal of Neuroscience, November 24, 200 30(47):5878 5887 Behavioral/Systems/Cognitive Selective Ablations Reveal That Orbital and Lateral Prefrontal Cortex Play Different Roles in Estimating
Contributions of Orbitofrontal and Lateral Prefrontal Cortices to Economic Choice and the Good-to-Action Transformation
 Article Contributions of Orbitofrontal and Lateral Prefrontal Cortices to Economic Choice and the Good-to-Action Transformation Xinying Cai 1 and Camillo Padoa-Schioppa 1,2,3, * 1 Department of Anatomy
Article Contributions of Orbitofrontal and Lateral Prefrontal Cortices to Economic Choice and the Good-to-Action Transformation Xinying Cai 1 and Camillo Padoa-Schioppa 1,2,3, * 1 Department of Anatomy
Cortical Control of Movement
 Strick Lecture 2 March 24, 2006 Page 1 Cortical Control of Movement Four parts of this lecture: I) Anatomical Framework, II) Physiological Framework, III) Primary Motor Cortex Function and IV) Premotor
Strick Lecture 2 March 24, 2006 Page 1 Cortical Control of Movement Four parts of this lecture: I) Anatomical Framework, II) Physiological Framework, III) Primary Motor Cortex Function and IV) Premotor
The individual animals, the basic design of the experiments and the electrophysiological
 SUPPORTING ONLINE MATERIAL Material and Methods The individual animals, the basic design of the experiments and the electrophysiological techniques for extracellularly recording from dopamine neurons were
SUPPORTING ONLINE MATERIAL Material and Methods The individual animals, the basic design of the experiments and the electrophysiological techniques for extracellularly recording from dopamine neurons were
The orbitofrontal cortex and the computation of subjective value: consolidated concepts and new perspectives
 Ann. N.Y. Acad. Sci. ISSN 0077-8923 ANNALS OF THE NEW YORK ACADEMY OF SCIENCES Issue: Critical Contributions of the Orbitofrontal Cortex to Behavior The orbitofrontal cortex and the computation of subjective
Ann. N.Y. Acad. Sci. ISSN 0077-8923 ANNALS OF THE NEW YORK ACADEMY OF SCIENCES Issue: Critical Contributions of the Orbitofrontal Cortex to Behavior The orbitofrontal cortex and the computation of subjective
Connect with amygdala (emotional center) Compares expected with actual Compare expected reward/punishment with actual reward/punishment Intuitive
 Orbitofronal Notes Frontal lobe prefrontal cortex 1. Dorsolateral Last to myelinate Sleep deprivation 2. Orbitofrontal Like dorsolateral, involved in: Executive functions Working memory Cognitive flexibility
Orbitofronal Notes Frontal lobe prefrontal cortex 1. Dorsolateral Last to myelinate Sleep deprivation 2. Orbitofrontal Like dorsolateral, involved in: Executive functions Working memory Cognitive flexibility
THE AMYGDALA AND REWARD
 THE AMYGDALA AND REWARD Mark G. Baxter* and Elisabeth A. Murray The amygdala an almond-shaped group of nuclei at the heart of the telencephalon has been associated with a range of cognitive functions,
THE AMYGDALA AND REWARD Mark G. Baxter* and Elisabeth A. Murray The amygdala an almond-shaped group of nuclei at the heart of the telencephalon has been associated with a range of cognitive functions,
Reward prediction based on stimulus categorization in. primate lateral prefrontal cortex
 Reward prediction based on stimulus categorization in primate lateral prefrontal cortex Xiaochuan Pan, Kosuke Sawa, Ichiro Tsuda, Minoro Tsukada, Masamichi Sakagami Supplementary Information This PDF file
Reward prediction based on stimulus categorization in primate lateral prefrontal cortex Xiaochuan Pan, Kosuke Sawa, Ichiro Tsuda, Minoro Tsukada, Masamichi Sakagami Supplementary Information This PDF file
Ventrolateral Prefrontal Neuronal Activity Related to Active Controlled Memory Retrieval in Nonhuman Primates
 Cerebral Cortex 2007;17:i27-i40 doi:10.1093/cercor/bhm086 Ventrolateral Prefrontal Neuronal Activity Related to Active Controlled Memory Retrieval in Nonhuman Primates Genevie` ve Cadoret and Michael Petrides
Cerebral Cortex 2007;17:i27-i40 doi:10.1093/cercor/bhm086 Ventrolateral Prefrontal Neuronal Activity Related to Active Controlled Memory Retrieval in Nonhuman Primates Genevie` ve Cadoret and Michael Petrides
nucleus accumbens septi hier-259 Nucleus+Accumbens birnlex_727
 Nucleus accumbens From Wikipedia, the free encyclopedia Brain: Nucleus accumbens Nucleus accumbens visible in red. Latin NeuroNames MeSH NeuroLex ID nucleus accumbens septi hier-259 Nucleus+Accumbens birnlex_727
Nucleus accumbens From Wikipedia, the free encyclopedia Brain: Nucleus accumbens Nucleus accumbens visible in red. Latin NeuroNames MeSH NeuroLex ID nucleus accumbens septi hier-259 Nucleus+Accumbens birnlex_727
Toward a Mechanistic Understanding of Human Decision Making Contributions of Functional Neuroimaging
 CURRENT DIRECTIONS IN PSYCHOLOGICAL SCIENCE Toward a Mechanistic Understanding of Human Decision Making Contributions of Functional Neuroimaging John P. O Doherty and Peter Bossaerts Computation and Neural
CURRENT DIRECTIONS IN PSYCHOLOGICAL SCIENCE Toward a Mechanistic Understanding of Human Decision Making Contributions of Functional Neuroimaging John P. O Doherty and Peter Bossaerts Computation and Neural
BIOMED 509. Executive Control UNM SOM. Primate Research Inst. Kyoto,Japan. Cambridge University. JL Brigman
 BIOMED 509 Executive Control Cambridge University Primate Research Inst. Kyoto,Japan UNM SOM JL Brigman 4-7-17 Symptoms and Assays of Cognitive Disorders Symptoms of Cognitive Disorders Learning & Memory
BIOMED 509 Executive Control Cambridge University Primate Research Inst. Kyoto,Japan UNM SOM JL Brigman 4-7-17 Symptoms and Assays of Cognitive Disorders Symptoms of Cognitive Disorders Learning & Memory
Emotion Explained. Edmund T. Rolls
 Emotion Explained Edmund T. Rolls Professor of Experimental Psychology, University of Oxford and Fellow and Tutor in Psychology, Corpus Christi College, Oxford OXPORD UNIVERSITY PRESS Contents 1 Introduction:
Emotion Explained Edmund T. Rolls Professor of Experimental Psychology, University of Oxford and Fellow and Tutor in Psychology, Corpus Christi College, Oxford OXPORD UNIVERSITY PRESS Contents 1 Introduction:
The primate amygdala combines information about space and value
 The primate amygdala combines information about space and Christopher J Peck 1,8, Brian Lau 1,7,8 & C Daniel Salzman 1 6 npg 213 Nature America, Inc. All rights reserved. A stimulus predicting reinforcement
The primate amygdala combines information about space and Christopher J Peck 1,8, Brian Lau 1,7,8 & C Daniel Salzman 1 6 npg 213 Nature America, Inc. All rights reserved. A stimulus predicting reinforcement
How rational are your decisions? Neuroeconomics
 How rational are your decisions? Neuroeconomics Hecke CNS Seminar WS 2006/07 Motivation Motivation Ferdinand Porsche "Wir wollen Autos bauen, die keiner braucht aber jeder haben will." Outline 1 Introduction
How rational are your decisions? Neuroeconomics Hecke CNS Seminar WS 2006/07 Motivation Motivation Ferdinand Porsche "Wir wollen Autos bauen, die keiner braucht aber jeder haben will." Outline 1 Introduction
The orbitofrontal cortex, predicted value, and choice
 Ann. N.Y. Acad. Sci. ISSN 0077-8923 ANNALS OF THE NEW YORK ACADEMY OF SCIENCES Issue: Critical Contributions of the Orbitofrontal Cortex to Behavior The orbitofrontal cortex, predicted value, and choice
Ann. N.Y. Acad. Sci. ISSN 0077-8923 ANNALS OF THE NEW YORK ACADEMY OF SCIENCES Issue: Critical Contributions of the Orbitofrontal Cortex to Behavior The orbitofrontal cortex, predicted value, and choice
Brain Mechanisms of Emotion 1 of 6
 Brain Mechanisms of Emotion 1 of 6 I. WHAT IS AN EMOTION? A. Three components (Oately & Jenkins, 1996) 1. caused by conscious or unconscious evaluation of an event as relevant to a goal that is important
Brain Mechanisms of Emotion 1 of 6 I. WHAT IS AN EMOTION? A. Three components (Oately & Jenkins, 1996) 1. caused by conscious or unconscious evaluation of an event as relevant to a goal that is important
Basic definition and Classification of Anhedonia. Preclinical and Clinical assessment of anhedonia.
 Basic definition and Classification of Anhedonia. Preclinical and Clinical assessment of anhedonia. Neurobiological basis and pathways involved in anhedonia. Objective characterization and computational
Basic definition and Classification of Anhedonia. Preclinical and Clinical assessment of anhedonia. Neurobiological basis and pathways involved in anhedonia. Objective characterization and computational
BRAIN MECHANISMS OF REWARD AND ADDICTION
 BRAIN MECHANISMS OF REWARD AND ADDICTION TREVOR.W. ROBBINS Department of Experimental Psychology, University of Cambridge Many drugs of abuse, including stimulants such as amphetamine and cocaine, opiates
BRAIN MECHANISMS OF REWARD AND ADDICTION TREVOR.W. ROBBINS Department of Experimental Psychology, University of Cambridge Many drugs of abuse, including stimulants such as amphetamine and cocaine, opiates
 This article was originally published in the Encyclopedia of Neuroscience published by Elsevier, and the attached copy is provided by Elsevier for the author's benefit and for the benefit of the author's
This article was originally published in the Encyclopedia of Neuroscience published by Elsevier, and the attached copy is provided by Elsevier for the author's benefit and for the benefit of the author's
Cognition in Parkinson's Disease and the Effect of Dopaminergic Therapy
 Cognition in Parkinson's Disease and the Effect of Dopaminergic Therapy Penny A. MacDonald, MD, PhD, FRCP(C) Canada Research Chair Tier 2 in Cognitive Neuroscience and Neuroimaging Assistant Professor
Cognition in Parkinson's Disease and the Effect of Dopaminergic Therapy Penny A. MacDonald, MD, PhD, FRCP(C) Canada Research Chair Tier 2 in Cognitive Neuroscience and Neuroimaging Assistant Professor
The Orbitofrontal Cortex and Reward
 The Orbitofrontal Cortex and Reward Edmund T. Rolls University of Oxford, Department of Experimental Psychology, South Parks Road, Oxford OX1 3UD, UK The primate orbitofrontal cortex contains the secondary
The Orbitofrontal Cortex and Reward Edmund T. Rolls University of Oxford, Department of Experimental Psychology, South Parks Road, Oxford OX1 3UD, UK The primate orbitofrontal cortex contains the secondary
Orbitofrontal cortex. From Wikipedia, the free encyclopedia. Approximate location of the OFC shown on a sagittal MRI
 Orbitofrontal cortex From Wikipedia, the free encyclopedia Approximate location of the OFC shown on a sagittal MRI Orbital surface of left frontal lobe. The orbitofrontal cortex (OFC) is a prefrontal cortex
Orbitofrontal cortex From Wikipedia, the free encyclopedia Approximate location of the OFC shown on a sagittal MRI Orbital surface of left frontal lobe. The orbitofrontal cortex (OFC) is a prefrontal cortex
Anatomy of the basal ganglia. Dana Cohen Gonda Brain Research Center, room 410
 Anatomy of the basal ganglia Dana Cohen Gonda Brain Research Center, room 410 danacoh@gmail.com The basal ganglia The nuclei form a small minority of the brain s neuronal population. Little is known about
Anatomy of the basal ganglia Dana Cohen Gonda Brain Research Center, room 410 danacoh@gmail.com The basal ganglia The nuclei form a small minority of the brain s neuronal population. Little is known about
Chapter 5: Learning and Behavior Learning How Learning is Studied Ivan Pavlov Edward Thorndike eliciting stimulus emitted
 Chapter 5: Learning and Behavior A. Learning-long lasting changes in the environmental guidance of behavior as a result of experience B. Learning emphasizes the fact that individual environments also play
Chapter 5: Learning and Behavior A. Learning-long lasting changes in the environmental guidance of behavior as a result of experience B. Learning emphasizes the fact that individual environments also play
Cerebral Cortex 1. Sarah Heilbronner
 Cerebral Cortex 1 Sarah Heilbronner heilb028@umn.edu Want to meet? Coffee hour 10-11am Tuesday 11/27 Surdyk s Overview and organization of the cerebral cortex What is the cerebral cortex? Where is each
Cerebral Cortex 1 Sarah Heilbronner heilb028@umn.edu Want to meet? Coffee hour 10-11am Tuesday 11/27 Surdyk s Overview and organization of the cerebral cortex What is the cerebral cortex? Where is each
Contributions of the prefrontal cortex to the neural basis of human decision making
 Neuroscience and Biobehavioral Reviews 26 (2002) 631 664 Review Contributions of the prefrontal cortex to the neural basis of human decision making Daniel C. Krawczyk* Department of Psychology, University
Neuroscience and Biobehavioral Reviews 26 (2002) 631 664 Review Contributions of the prefrontal cortex to the neural basis of human decision making Daniel C. Krawczyk* Department of Psychology, University
5th Mini-Symposium on Cognition, Decision-making and Social Function: In Memory of Kang Cheng
 5th Mini-Symposium on Cognition, Decision-making and Social Function: In Memory of Kang Cheng 13:30-13:35 Opening 13:30 17:30 13:35-14:00 Metacognition in Value-based Decision-making Dr. Xiaohong Wan (Beijing
5th Mini-Symposium on Cognition, Decision-making and Social Function: In Memory of Kang Cheng 13:30-13:35 Opening 13:30 17:30 13:35-14:00 Metacognition in Value-based Decision-making Dr. Xiaohong Wan (Beijing
Neuronal Representations of Value
 C H A P T E R 29 c00029 Neuronal Representations of Value Michael Platt and Camillo Padoa-Schioppa O U T L I N E s0020 s0030 s0040 s0050 s0060 s0070 s0080 s0090 s0100 s0110 Introduction 440 Value and Decision-making
C H A P T E R 29 c00029 Neuronal Representations of Value Michael Platt and Camillo Padoa-Schioppa O U T L I N E s0020 s0030 s0040 s0050 s0060 s0070 s0080 s0090 s0100 s0110 Introduction 440 Value and Decision-making
CSE511 Brain & Memory Modeling Lect 22,24,25: Memory Systems
 CSE511 Brain & Memory Modeling Lect 22,24,25: Memory Systems Compare Chap 31 of Purves et al., 5e Chap 24 of Bear et al., 3e Larry Wittie Computer Science, StonyBrook University http://www.cs.sunysb.edu/~cse511
CSE511 Brain & Memory Modeling Lect 22,24,25: Memory Systems Compare Chap 31 of Purves et al., 5e Chap 24 of Bear et al., 3e Larry Wittie Computer Science, StonyBrook University http://www.cs.sunysb.edu/~cse511
Regretful Choices: Neural Representations of Choice in the Orbitofrontal Cortex. A Dissertation SUBMITTED TO THE FACULTY OF UNIVERSITY OF MINNESOTA BY
 Regretful Choices: Neural Representations of Choice in the Orbitofrontal Cortex A Dissertation SUBMITTED TO THE FACULTY OF UNIVERSITY OF MINNESOTA BY Adam Patrick Steiner IN PARTIAL FULFILLMENT OF THE
Regretful Choices: Neural Representations of Choice in the Orbitofrontal Cortex A Dissertation SUBMITTED TO THE FACULTY OF UNIVERSITY OF MINNESOTA BY Adam Patrick Steiner IN PARTIAL FULFILLMENT OF THE
Computational Explorations in Cognitive Neuroscience Chapter 7: Large-Scale Brain Area Functional Organization
 Computational Explorations in Cognitive Neuroscience Chapter 7: Large-Scale Brain Area Functional Organization 1 7.1 Overview This chapter aims to provide a framework for modeling cognitive phenomena based
Computational Explorations in Cognitive Neuroscience Chapter 7: Large-Scale Brain Area Functional Organization 1 7.1 Overview This chapter aims to provide a framework for modeling cognitive phenomena based
Supplementary Figure 1. Example of an amygdala neuron whose activity reflects value during the visual stimulus interval. This cell responded more
 1 Supplementary Figure 1. Example of an amygdala neuron whose activity reflects value during the visual stimulus interval. This cell responded more strongly when an image was negative than when the same
1 Supplementary Figure 1. Example of an amygdala neuron whose activity reflects value during the visual stimulus interval. This cell responded more strongly when an image was negative than when the same
Supplementary Information
 Supplementary Information The neural correlates of subjective value during intertemporal choice Joseph W. Kable and Paul W. Glimcher a 10 0 b 10 0 10 1 10 1 Discount rate k 10 2 Discount rate k 10 2 10
Supplementary Information The neural correlates of subjective value during intertemporal choice Joseph W. Kable and Paul W. Glimcher a 10 0 b 10 0 10 1 10 1 Discount rate k 10 2 Discount rate k 10 2 10
Memory, Attention, and Decision-Making
 Memory, Attention, and Decision-Making A Unifying Computational Neuroscience Approach Edmund T. Rolls University of Oxford Department of Experimental Psychology Oxford England OXFORD UNIVERSITY PRESS Contents
Memory, Attention, and Decision-Making A Unifying Computational Neuroscience Approach Edmund T. Rolls University of Oxford Department of Experimental Psychology Oxford England OXFORD UNIVERSITY PRESS Contents
Cognitive Neuroscience Attention
 Cognitive Neuroscience Attention There are many aspects to attention. It can be controlled. It can be focused on a particular sensory modality or item. It can be divided. It can set a perceptual system.
Cognitive Neuroscience Attention There are many aspects to attention. It can be controlled. It can be focused on a particular sensory modality or item. It can be divided. It can set a perceptual system.
HHS Public Access Author manuscript Neuron. Author manuscript; available in PMC 2017 June 15.
 Orbitofrontal cortex value signals depend on fixation location during free viewing Vincent B. McGinty 1, Antonio Rangel 2, and William T. Newsome 1,3 1 Department of Neurobiology, Stanford University,
Orbitofrontal cortex value signals depend on fixation location during free viewing Vincent B. McGinty 1, Antonio Rangel 2, and William T. Newsome 1,3 1 Department of Neurobiology, Stanford University,
Convergence of Sensory Systems in the Orbitofrontal Cortex in Primates and Brain Design for Emotion
 THE ANATOMICAL RECORD PART A 281A:1212 1225 (2004) Convergence of Sensory Systems in the Orbitofrontal Cortex in Primates and Brain Design for Emotion EDMUND T. ROLLS* Department of Experimental Psychology,
THE ANATOMICAL RECORD PART A 281A:1212 1225 (2004) Convergence of Sensory Systems in the Orbitofrontal Cortex in Primates and Brain Design for Emotion EDMUND T. ROLLS* Department of Experimental Psychology,
Sum of Neurally Distinct Stimulus- and Task-Related Components.
 SUPPLEMENTARY MATERIAL for Cardoso et al. 22 The Neuroimaging Signal is a Linear Sum of Neurally Distinct Stimulus- and Task-Related Components. : Appendix: Homogeneous Linear ( Null ) and Modified Linear
SUPPLEMENTARY MATERIAL for Cardoso et al. 22 The Neuroimaging Signal is a Linear Sum of Neurally Distinct Stimulus- and Task-Related Components. : Appendix: Homogeneous Linear ( Null ) and Modified Linear
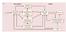 Peripheral facial paralysis (right side). The patient is asked to close her eyes and to retract their mouth (From Heimer) Hemiplegia of the left side. Note the characteristic position of the arm with
Peripheral facial paralysis (right side). The patient is asked to close her eyes and to retract their mouth (From Heimer) Hemiplegia of the left side. Note the characteristic position of the arm with
biological psychology, p. 40 The study of the nervous system, especially the brain. neuroscience, p. 40
 biological psychology, p. 40 The specialized branch of psychology that studies the relationship between behavior and bodily processes and system; also called biopsychology or psychobiology. neuroscience,
biological psychology, p. 40 The specialized branch of psychology that studies the relationship between behavior and bodily processes and system; also called biopsychology or psychobiology. neuroscience,
Still at the Choice-Point
 Still at the Choice-Point Action Selection and Initiation in Instrumental Conditioning BERNARD W. BALLEINE AND SEAN B. OSTLUND Department of Psychology and the Brain Research Institute, University of California,
Still at the Choice-Point Action Selection and Initiation in Instrumental Conditioning BERNARD W. BALLEINE AND SEAN B. OSTLUND Department of Psychology and the Brain Research Institute, University of California,
The Emotional Nervous System
 The Emotional Nervous System Dr. C. George Boeree Emotion involves the entire nervous system, of course. But there are two parts of the nervous system that are especially significant: The limbic system
The Emotional Nervous System Dr. C. George Boeree Emotion involves the entire nervous system, of course. But there are two parts of the nervous system that are especially significant: The limbic system
Academic year Lecture 16 Emotions LECTURE 16 EMOTIONS
 Course Behavioral Economics Academic year 2013-2014 Lecture 16 Emotions Alessandro Innocenti LECTURE 16 EMOTIONS Aim: To explore the role of emotions in economic decisions. Outline: How emotions affect
Course Behavioral Economics Academic year 2013-2014 Lecture 16 Emotions Alessandro Innocenti LECTURE 16 EMOTIONS Aim: To explore the role of emotions in economic decisions. Outline: How emotions affect
Associative learning
 Introduction to Learning Associative learning Event-event learning (Pavlovian/classical conditioning) Behavior-event learning (instrumental/ operant conditioning) Both are well-developed experimentally
Introduction to Learning Associative learning Event-event learning (Pavlovian/classical conditioning) Behavior-event learning (instrumental/ operant conditioning) Both are well-developed experimentally
25/09/2012. Capgras Syndrome. Chapter 2. Capgras Syndrome - 2. The Neural Basis of Cognition
 Chapter 2 The Neural Basis of Cognition Capgras Syndrome Alzheimer s patients & others delusion that significant others are robots or impersonators - paranoia Two brain systems for facial recognition -
Chapter 2 The Neural Basis of Cognition Capgras Syndrome Alzheimer s patients & others delusion that significant others are robots or impersonators - paranoia Two brain systems for facial recognition -
Instrumental Conditioning VI: There is more than one kind of learning
 Instrumental Conditioning VI: There is more than one kind of learning PSY/NEU338: Animal learning and decision making: Psychological, computational and neural perspectives outline what goes into instrumental
Instrumental Conditioning VI: There is more than one kind of learning PSY/NEU338: Animal learning and decision making: Psychological, computational and neural perspectives outline what goes into instrumental
The Role of Orbitofrontal Cortex in Decision Making
 The Role of Orbitofrontal Cortex in Decision Making A Component Process Account LESLEY K. FELLOWS Montreal Neurological Institute, McGill University, Montréal, Québec, Canada ABSTRACT: Clinical accounts
The Role of Orbitofrontal Cortex in Decision Making A Component Process Account LESLEY K. FELLOWS Montreal Neurological Institute, McGill University, Montréal, Québec, Canada ABSTRACT: Clinical accounts
Emotion I: General concepts, fear and anxiety
 C82NAB Neuroscience and Behaviour Emotion I: General concepts, fear and anxiety Tobias Bast, School of Psychology, University of Nottingham 1 Outline Emotion I (first part) Studying brain substrates of
C82NAB Neuroscience and Behaviour Emotion I: General concepts, fear and anxiety Tobias Bast, School of Psychology, University of Nottingham 1 Outline Emotion I (first part) Studying brain substrates of
The previous three chapters provide a description of the interaction between explicit and
 77 5 Discussion The previous three chapters provide a description of the interaction between explicit and implicit learning systems. Chapter 2 described the effects of performing a working memory task
77 5 Discussion The previous three chapters provide a description of the interaction between explicit and implicit learning systems. Chapter 2 described the effects of performing a working memory task
Reward-Dependent Modulation of Working Memory in Lateral Prefrontal Cortex
 The Journal of Neuroscience, March 11, 29 29(1):3259 327 3259 Behavioral/Systems/Cognitive Reward-Dependent Modulation of Working Memory in Lateral Prefrontal Cortex Steven W. Kennerley and Jonathan D.
The Journal of Neuroscience, March 11, 29 29(1):3259 327 3259 Behavioral/Systems/Cognitive Reward-Dependent Modulation of Working Memory in Lateral Prefrontal Cortex Steven W. Kennerley and Jonathan D.
SUPPLEMENTARY INFORMATION
 doi:10.1038/nature11239 Introduction The first Supplementary Figure shows additional regions of fmri activation evoked by the task. The second, sixth, and eighth shows an alternative way of analyzing reaction
doi:10.1038/nature11239 Introduction The first Supplementary Figure shows additional regions of fmri activation evoked by the task. The second, sixth, and eighth shows an alternative way of analyzing reaction
Time Experiencing by Robotic Agents
 Time Experiencing by Robotic Agents Michail Maniadakis 1 and Marc Wittmann 2 and Panos Trahanias 1 1- Foundation for Research and Technology - Hellas, ICS, Greece 2- Institute for Frontier Areas of Psychology
Time Experiencing by Robotic Agents Michail Maniadakis 1 and Marc Wittmann 2 and Panos Trahanias 1 1- Foundation for Research and Technology - Hellas, ICS, Greece 2- Institute for Frontier Areas of Psychology
The Functions of the Orbitofrontal Cortex
 Neurocase (1999) Vol. 5, pp. 301 312 Oxford University Press 1999 REVIEW The Functions of the Orbitofrontal Cortex Edmund T. Rolls University of Oxford, Department of Experimental Psychology, South Parks
Neurocase (1999) Vol. 5, pp. 301 312 Oxford University Press 1999 REVIEW The Functions of the Orbitofrontal Cortex Edmund T. Rolls University of Oxford, Department of Experimental Psychology, South Parks
Reinforcement learning and the brain: the problems we face all day. Reinforcement Learning in the brain
 Reinforcement learning and the brain: the problems we face all day Reinforcement Learning in the brain Reading: Y Niv, Reinforcement learning in the brain, 2009. Decision making at all levels Reinforcement
Reinforcement learning and the brain: the problems we face all day Reinforcement Learning in the brain Reading: Y Niv, Reinforcement learning in the brain, 2009. Decision making at all levels Reinforcement
Chapter 3. Structure and Function of the Nervous System. Copyright (c) Allyn and Bacon 2004
 Chapter 3 Structure and Function of the Nervous System 1 Basic Features of the Nervous System Neuraxis: An imaginary line drawn through the center of the length of the central nervous system, from the
Chapter 3 Structure and Function of the Nervous System 1 Basic Features of the Nervous System Neuraxis: An imaginary line drawn through the center of the length of the central nervous system, from the
Neurobiological Foundations of Reward and Risk
 Neurobiological Foundations of Reward and Risk... and corresponding risk prediction errors Peter Bossaerts 1 Contents 1. Reward Encoding And The Dopaminergic System 2. Reward Prediction Errors And TD (Temporal
Neurobiological Foundations of Reward and Risk... and corresponding risk prediction errors Peter Bossaerts 1 Contents 1. Reward Encoding And The Dopaminergic System 2. Reward Prediction Errors And TD (Temporal
Psych3BN3 Topic 4 Emotion. Bilateral amygdala pathology: Case of S.M. (fig 9.1) S.M. s ratings of emotional intensity of faces (fig 9.
 Psych3BN3 Topic 4 Emotion Readings: Gazzaniga Chapter 9 Bilateral amygdala pathology: Case of S.M. (fig 9.1) SM began experiencing seizures at age 20 CT, MRI revealed amygdala atrophy, result of genetic
Psych3BN3 Topic 4 Emotion Readings: Gazzaniga Chapter 9 Bilateral amygdala pathology: Case of S.M. (fig 9.1) SM began experiencing seizures at age 20 CT, MRI revealed amygdala atrophy, result of genetic
Flavor Physiology q. Flavor Processing in the Primate Brain. The Primary Taste Cortex. The Secondary Taste Cortex
 Flavor Physiology q Edmund T Rolls, Oxford Centre for Computational Neuroscience, Oxford, United Kingdom Ó 2017 Elsevier Inc. All rights reserved. Flavor Processing in the Primate Brain 1 Pathways 1 The
Flavor Physiology q Edmund T Rolls, Oxford Centre for Computational Neuroscience, Oxford, United Kingdom Ó 2017 Elsevier Inc. All rights reserved. Flavor Processing in the Primate Brain 1 Pathways 1 The
Introduction to Systems Neuroscience. Nov. 28, The limbic system. Daniel C. Kiper
 Introduction to Systems Neuroscience Nov. 28, 2017 The limbic system Daniel C. Kiper kiper@ini.phys.ethz.ch http: www.ini.unizh.ch/~kiper/system_neurosci.html LIMBIC SYSTEM The term limbic system mean
Introduction to Systems Neuroscience Nov. 28, 2017 The limbic system Daniel C. Kiper kiper@ini.phys.ethz.ch http: www.ini.unizh.ch/~kiper/system_neurosci.html LIMBIC SYSTEM The term limbic system mean
The Adolescent Developmental Stage
 The Adolescent Developmental Stage o Physical maturation o Drive for independence o Increased salience of social and peer interactions o Brain development o Inflection in risky behaviors including experimentation
The Adolescent Developmental Stage o Physical maturation o Drive for independence o Increased salience of social and peer interactions o Brain development o Inflection in risky behaviors including experimentation
The human brain. of cognition need to make sense gives the structure of the brain (duh). ! What is the basic physiology of this organ?
 The human brain The human brain! What is the basic physiology of this organ?! Understanding the parts of this organ provides a hypothesis space for its function perhaps different parts perform different
The human brain The human brain! What is the basic physiology of this organ?! Understanding the parts of this organ provides a hypothesis space for its function perhaps different parts perform different
Behavioral Neuroscience: Fear thou not. Rony Paz
 Behavioral Neuroscience: Fear thou not Rony Paz Rony.paz@weizmann.ac.il Thoughts What is a reward? Learning is best motivated by threats to survival? Threats are much better reinforcers? Fear is a prime
Behavioral Neuroscience: Fear thou not Rony Paz Rony.paz@weizmann.ac.il Thoughts What is a reward? Learning is best motivated by threats to survival? Threats are much better reinforcers? Fear is a prime
Homework Week 2. PreLab 2 HW #2 Synapses (Page 1 in the HW Section)
 Homework Week 2 Due in Lab PreLab 2 HW #2 Synapses (Page 1 in the HW Section) Reminders No class next Monday Quiz 1 is @ 5:30pm on Tuesday, 1/22/13 Study guide posted under Study Aids section of website
Homework Week 2 Due in Lab PreLab 2 HW #2 Synapses (Page 1 in the HW Section) Reminders No class next Monday Quiz 1 is @ 5:30pm on Tuesday, 1/22/13 Study guide posted under Study Aids section of website
Brain Imaging studies in substance abuse. Jody Tanabe, MD University of Colorado Denver
 Brain Imaging studies in substance abuse Jody Tanabe, MD University of Colorado Denver NRSC January 28, 2010 Costs: Health, Crime, Productivity Costs in billions of dollars (2002) $400 $350 $400B legal
Brain Imaging studies in substance abuse Jody Tanabe, MD University of Colorado Denver NRSC January 28, 2010 Costs: Health, Crime, Productivity Costs in billions of dollars (2002) $400 $350 $400B legal
CISC 3250 Systems Neuroscience
 CISC 3250 Systems Neuroscience Levels of organization Central Nervous System 1m 10 11 neurons Neural systems and neuroanatomy Systems 10cm Networks 1mm Neurons 100μm 10 8 neurons Professor Daniel Leeds
CISC 3250 Systems Neuroscience Levels of organization Central Nervous System 1m 10 11 neurons Neural systems and neuroanatomy Systems 10cm Networks 1mm Neurons 100μm 10 8 neurons Professor Daniel Leeds
SUPPLEMENTARY INFORMATION
 doi:10.1038/nature14066 Supplementary discussion Gradual accumulation of evidence for or against different choices has been implicated in many types of decision-making, including value-based decisions
doi:10.1038/nature14066 Supplementary discussion Gradual accumulation of evidence for or against different choices has been implicated in many types of decision-making, including value-based decisions
Basic Nervous System anatomy. Neurobiology of Happiness
 Basic Nervous System anatomy Neurobiology of Happiness The components Central Nervous System (CNS) Brain Spinal Cord Peripheral" Nervous System (PNS) Somatic Nervous System Autonomic Nervous System (ANS)
Basic Nervous System anatomy Neurobiology of Happiness The components Central Nervous System (CNS) Brain Spinal Cord Peripheral" Nervous System (PNS) Somatic Nervous System Autonomic Nervous System (ANS)
The Somatic Marker Hypothesis: Human Emotions in Decision-Making
 The Somatic Marker Hypothesis: Human Emotions in Decision-Making Presented by Lin Xiao Brain and Creativity Institute University of Southern California Most of us are taught from early on that : -logical,
The Somatic Marker Hypothesis: Human Emotions in Decision-Making Presented by Lin Xiao Brain and Creativity Institute University of Southern California Most of us are taught from early on that : -logical,
If you give any person a prescription of something like Valium and have them take it on
 As always I am happy to do this presentation, which is my favorite topic in addiction medicine. I am an internist, and I have done healthcare for the homeless in Springfield as well as been the medical
As always I am happy to do this presentation, which is my favorite topic in addiction medicine. I am an internist, and I have done healthcare for the homeless in Springfield as well as been the medical
(Visual) Attention. October 3, PSY Visual Attention 1
 (Visual) Attention Perception and awareness of a visual object seems to involve attending to the object. Do we have to attend to an object to perceive it? Some tasks seem to proceed with little or no attention
(Visual) Attention Perception and awareness of a visual object seems to involve attending to the object. Do we have to attend to an object to perceive it? Some tasks seem to proceed with little or no attention
Dissociating Valence of Outcome from Behavioral Control in Human Orbital and Ventral Prefrontal Cortices
 The Journal of Neuroscience, August 27, 2003 23(21):7931 7939 7931 Behavioral/Systems/Cognitive Dissociating Valence of Outcome from Behavioral Control in Human Orbital and Ventral Prefrontal Cortices
The Journal of Neuroscience, August 27, 2003 23(21):7931 7939 7931 Behavioral/Systems/Cognitive Dissociating Valence of Outcome from Behavioral Control in Human Orbital and Ventral Prefrontal Cortices
Henry Molaison. Biography. From Wikipedia, the free encyclopedia
 Henry Molaison From Wikipedia, the free encyclopedia Henry Gustav Molaison (February 26, 1926 December 2, 2008), known widely as H.M., was an American memory disorder patient who had a bilateral medial
Henry Molaison From Wikipedia, the free encyclopedia Henry Gustav Molaison (February 26, 1926 December 2, 2008), known widely as H.M., was an American memory disorder patient who had a bilateral medial
Reward, Context, and Human Behaviour
 Review Article TheScientificWorldJOURNAL (2007) 7, 626 640 ISSN 1537-744X; DOI 10.1100/tsw.2007.122 Reward, Context, and Human Behaviour Clare L. Blaukopf and Gregory J. DiGirolamo* Department of Experimental
Review Article TheScientificWorldJOURNAL (2007) 7, 626 640 ISSN 1537-744X; DOI 10.1100/tsw.2007.122 Reward, Context, and Human Behaviour Clare L. Blaukopf and Gregory J. DiGirolamo* Department of Experimental
TO BE MOTIVATED IS TO HAVE AN INCREASE IN DOPAMINE. The statement to be motivated is to have an increase in dopamine implies that an increase in
 1 NAME COURSE TITLE 2 TO BE MOTIVATED IS TO HAVE AN INCREASE IN DOPAMINE The statement to be motivated is to have an increase in dopamine implies that an increase in dopamine neurotransmitter, up-regulation
1 NAME COURSE TITLE 2 TO BE MOTIVATED IS TO HAVE AN INCREASE IN DOPAMINE The statement to be motivated is to have an increase in dopamine implies that an increase in dopamine neurotransmitter, up-regulation
Brain mechanisms of emotion and decision-making
 International Congress Series 1291 (2006) 3 13 www.ics-elsevier.com Brain mechanisms of emotion and decision-making Edmund T. Rolls * Department of Experimental Psychology, University of Oxford, South
International Congress Series 1291 (2006) 3 13 www.ics-elsevier.com Brain mechanisms of emotion and decision-making Edmund T. Rolls * Department of Experimental Psychology, University of Oxford, South
TMS Disruption of Time Encoding in Human Primary Visual Cortex Molly Bryan Beauchamp Lab
 TMS Disruption of Time Encoding in Human Primary Visual Cortex Molly Bryan Beauchamp Lab This report details my summer research project for the REU Theoretical and Computational Neuroscience program as
TMS Disruption of Time Encoding in Human Primary Visual Cortex Molly Bryan Beauchamp Lab This report details my summer research project for the REU Theoretical and Computational Neuroscience program as
Psychology in Your Life
 Sarah Grison Todd Heatherton Michael Gazzaniga Psychology in Your Life SECOND EDITION Chapter 2 The Role of Biology in Psychology 1 2016 W. W. Norton & Company, Inc. 2.1 How Do Our Nervous Systems Affect
Sarah Grison Todd Heatherton Michael Gazzaniga Psychology in Your Life SECOND EDITION Chapter 2 The Role of Biology in Psychology 1 2016 W. W. Norton & Company, Inc. 2.1 How Do Our Nervous Systems Affect
Nature Neuroscience: doi: /nn.4642
 Supplementary Figure 1 Recording sites and example waveform clustering, as well as electrophysiological recordings of auditory CS and shock processing following overtraining. (a) Recording sites in LC
Supplementary Figure 1 Recording sites and example waveform clustering, as well as electrophysiological recordings of auditory CS and shock processing following overtraining. (a) Recording sites in LC
THE BRAIN HABIT BRIDGING THE CONSCIOUS AND UNCONSCIOUS MIND
 THE BRAIN HABIT BRIDGING THE CONSCIOUS AND UNCONSCIOUS MIND Mary ET Boyle, Ph. D. Department of Cognitive Science UCSD How did I get here? What did I do? Start driving home after work Aware when you left
THE BRAIN HABIT BRIDGING THE CONSCIOUS AND UNCONSCIOUS MIND Mary ET Boyle, Ph. D. Department of Cognitive Science UCSD How did I get here? What did I do? Start driving home after work Aware when you left
Choosing the Greater of Two Goods: Neural Currencies for Valuation and Decision Making
 Choosing the Greater of Two Goods: Neural Currencies for Valuation and Decision Making Leo P. Surgre, Gres S. Corrado and William T. Newsome Presenter: He Crane Huang 04/20/2010 Outline Studies on neural
Choosing the Greater of Two Goods: Neural Currencies for Valuation and Decision Making Leo P. Surgre, Gres S. Corrado and William T. Newsome Presenter: He Crane Huang 04/20/2010 Outline Studies on neural
