ORIGINAL ARTICLE. neuroimaging studies have provided evidence consistent
|
|
|
- Peregrine Roy Robbins
- 5 years ago
- Views:
Transcription
1 ORIGINAL ARTICLE A Functional Magnetic Resonance Imaging Study of Amygdala and Medial Prefrontal Cortex Responses to Overtly Presented Fearful Faces in Posttraumatic Stress Disorder Lisa M. Shin, PhD; Christopher I. Wright, MD, PhD; Paul A. Cannistraro, MD; Michelle M. Wedig, BS; Katherine McMullin, BA; Brian Martis, MD; Michael L. Macklin, BA; Natasha B. Lasko, PhD; Sarah R. Cavanagh, MA; Terri S. Krangel, PhD; Scott P. Orr, PhD; Roger K. Pitman, MD; Paul J. Whalen, PhD; Scott L. Rauch, MD Background: Previous functional neuroimaging studies have demonstrated exaggerated amygdala responses and diminished medial prefrontal cortex responses during the symptomatic state in posttraumatic stress disorder (PTSD). Objectives: To determine whether these abnormalities also occur in response to overtly presented affective stimuli unrelated to trauma; to examine the functional relationship between the amygdala and medial prefrontal cortex and their relationship to PTSD symptom severity in response to these stimuli; and to determine whether responsivity of these regions habituates normally across repeated stimulus presentations in PTSD. Design: Case-control study. Setting: Academic medical center. Participants: Volunteer sample of 13 men with PTSD (PTSD group) and 13 trauma-exposed men without PTSD (control group). Main Outcome Measures: We used functional magnetic resonance imaging (fmri) to study blood oxygenation level dependent signal during the presentation of emotional facial expressions. Results: The PTSD group exhibited exaggerated amygdala responses and diminished medial prefrontal cortex responses to fearful vs happy facial expressions. In addition, in the PTSD group, blood oxygenation level dependent signal changes in the amygdala were negatively correlated with signal changes in the medial prefrontal cortex, and symptom severity was negatively related to blood oxygenation level dependent signal changes in the medial prefrontal cortex. Finally, relative to the control group, the PTSD group tended to exhibit diminished habituation of fearful vs happy responses in the right amygdala across functional runs, although this effect did not exceed our a priori statistical threshold. Conclusions: These results provide evidence for exaggerated amygdala responsivity, diminished medial prefrontal cortex responsivity, and a reciprocal relationship between these 2 regions during passive viewing of overtly presented affective stimuli unrelated to trauma in PTSD. Arch Gen Psychiatry. 2005;62: Author Affiliations are listed at the end of this article. SEVERAL RECENT FUNCTIONAL neuroimaging studies have provided evidence consistent with amygdala hyperresponsivity during exposure to traumatic reminders in posttraumatic stress disorder (PTSD). 1-6 In addition, PTSD symptom severity is positively correlated with blood flow in the amygdala during such exposure. 3,5,7 To characterize the scope of amygdala hyperresponsivity in PTSD, it is important to determine whether amygdala responses to stimuli unrelated to trauma are also exaggerated in this disorder. Previous research has established that the normal human amygdala is responsive to facial expressions of fear 8-11 and that individuals with PTSD exhibit exaggerated amygdala responses to these facial expressions. Using functional magnetic resonance imaging (fmri), Rauch and colleagues 12 measured blood oxygenation level dependent (BOLD) signal responses to backwardly masked fearful and happy facial expressions in combat veterans with and without PTSD. In that study, the fearful and happy facial expressions were presented very briefly (33 milliseconds) and were followed immediately by neutral facial expressions. 11 Relative to the control group, the PTSD group exhibited greater BOLD signal increases in the right amygdala in response to masked fearful vs 273
2 happy facial expressions. In addition, PTSD symptom severity was positively correlated with BOLD signal changes in the amygdala. Whether amygdala hyperresponsivity in PTSD can be demonstrated when participants are explicitly aware of the presence of emotional facial expressions is not known. In addition, whether this amygdala hyperresponsivity declines normally across repeatedstimulus presentations has never been assessed in PTSD. Medial prefrontal structures also have been implicated in the pathophysiology of PTSD. Several functional neuroimaging studies have demonstrated relatively diminished activation in the subcallosal, 13,14 anterior cingulate, and medial frontal gyri 5,16 in this disorder. In addition, PTSD symptom severity is negatively correlated with blood flow in the medial frontal gyrus during traumatic imagery and recollection. 5 Furthermore, blood flow changes in the medial frontal gyrus appear to be negatively correlated with changes in the amygdala during traumatic imagery in PTSD. 5 These findings highlight the potential importance of interactions between the amygdala and medial prefrontal structures in this disorder. However, whether such interactions can be observed during the presentation of more general, affective stimuli unrelated to trauma is unknown. The goals of the present study were to determine whether (1) individuals with PTSD exhibit exaggerated amygdala responses and diminished medial prefrontal responses to nonmasked (ie, overtly presented) fearful vs happy facial expressions; (2) there is a reciprocal relationship between BOLD signal changes in the amygdala and medial prefrontal regions in this contrast; (3) symptom severity is related to signal changes in the amygdala and medial prefrontal regions; and (4) the amygdala and medial prefrontal responses to fearful vs happy faces habituate normally in patients with PTSD. We studied fmri BOLD signal in trauma-exposed men with and without PTSD while they viewed blocks of emotional facial expressions. In the fearful vs happy comparison, we predicted that participants with PTSD would exhibit greater activation in the amygdala 12 and diminished activation in medial prefrontal regions (including medial frontal, anterior cingulate, and subcallosal gyri), compared with participants without PTSD. In addition, we predicted that fmri BOLD signal changes in the amygdala would be negatively correlated with signal changes in medial prefrontal regions in the fearful vs happy contrast. 5 We also predicted that symptom severity would be positively correlated with responses in the amygdala 5,7,12 and negatively correlated with those in medial prefrontal regions. 5 Finally, we predicted that, relative to the control group, the PTSD group would show diminished habituation of amygdala responses over repeated presentations of fearful vs happy expressions. METHODS PARTICIPANTS Participants included 26 trauma-exposed men without a history of head injury, neurological disorders, or other major medical conditions. Thirteen participants met DSM-IV diagnostic criteria for current PTSD (PTSD group), and 13 never had PTSD (control group) according to a structured clinical interview (the Clinician-Administered PTSD Scale [CAPS]). 18 Twenty-four participants were right-handed, and 2 (1 in the PTSD and 1 in the control group) were left-handed. 19 Participants had served in combat in Vietnam (10 in the PTSD and 8 in the control group) or were firefighters (3 in the PTSD and 5 in the control group). Examples of Vietnam experiences included being wounded or in mortal danger in combat, witnessing the injury or death of friends, and handling body parts. Examples of firefighter experiences included being trapped and injured in a collapsed burning building, witnessing the death and/or disfigurement of others including children and coworkers, and witnessing an accident scene of a family member. All cases of PTSD were chronic. Duration of illness ranged from 10 to 35 years. No participant was taking psychotropic or cardiovascular medication at the time of the study. Seven participants (5 in the PTSD and 2 in the control group) had been taking such medications before the study but had discontinued them for 2 to 6 weeks, depending on the type of medication. The groups did not differ with regard to age (mean ± SD age, 52.8 ±7.3 [PTSD group] vs 49.7 ±8.9 years [control group]; F 1,24 =0.9;P=.35). The control group had an average of 1.8 more years of education than the PTSD group (mean±sd, 13.8±2.3 [PTSD group] vs 15.6±1.9 years [control group]; F 1,24 =2.2; P=.04). The groups did not differ in terms of marital status (69% [PTSD group] vs 85% [control group] married; 2 1=0.9; P=.35), but a greater proportion of participants in the PTSD group were unemployed (38% [PTSD group] vs 0% [control group]; 2 1=6.2; P=.02). Relative to the control group, the PTSD group had significantly higher mean±sd scores (indicating greater symptom severity) on the following clinical measures: CAPS (62.0±25.2 [PTSD group] vs 3.3±6.0 [control group]; F 1,22 =61.9; P.001); the Beck Depression Inventory 20 (BDI) (20.7±17.0 [PTSD group] vs 4.2±4.3 [control group]; F 1,24 =11.5; P=.003); and Beck Anxiety Inventory 21 (18.8±14.5 [PTSD group] vs 5.0±6.2 [control group]; F 1,24 =10.1; P=.004). All symptoms of PTSD were represented among the various cases of PTSD, although not every symptom in each case. Flashbacks were less frequent, and amnesia was rare. The PTSD group s mean CAPS score represents moderately severe PTSD symptoms, and the control group s mean CAPS score represents minimal PTSD symptoms. The presence of other Axis I mental disorders was assessed with the Structured Clinical Interview for DSM-IV. 22 Participants in the PTSD group met criteria for the following current comorbid diagnoses: major depression (n=4), dysthymia (n=2), bipolar disorder II (n=1), panic disorder (n=3), social phobia (n=2), and specific phobia (n=1). None of the participants in the control group met criteria for a current Axis I diagnosis. This study was approved by the institutional review boards of the Massachusetts General Hospital and the Veterans Affairs Medical Center. Written informed consent was obtained from each participant. STIMULUS PRESENTATION Stimuli were gray scale images of 6 fearful, 6 happy, and 6 neutral facial expressions selected from a well-validated set. 23 Facial expressions were posed by 3 men and 3 women. Each face was presented for 200 milliseconds, with a 300-millisecond interstimulus interval, in a pseudorandom order such that facial expressions of a single identity were never presented in succession. Across each run, each fearful and happy face was presented an equal number of times. Subjects viewed 4 runs of these facial expressions, with each run consisting of ten 28-second alternating blocks. Each run began and ended with a 28- second block of low-level fixation (eg, +NHFHFHFN+). The 274
3 order of conditions and runs was counterbalanced across subjects and groups. The paradigm was modeled after those used in previous studies. 8,24 Although it does not permit the acquisition of online behavioral data, we chose to use a passive viewing paradigm because it has been found to be associated with relatively robust amygdala responses. 25,26 The facial stimuli were displayed using standardized software (MacStim 2.5.9; WhiteAnt Occasional Publishing, West Melbourne, Australia) and an XG-2000V color liquid crystal display projector (Sharp, Osaka, Japan). Immediately after each scanning session, outside the scanner, participants rated the facial expressions on scales of valence (negative to positive, 3 to +3) and arousal (low to high, 0 to 6). fmri PROCEDURES Scans were obtained from a Symphony/Sonata 1.5-T wholebody high-speed imaging device equipped for echo planar imaging (Siemens Medical Systems, Iselin, NJ) with a 3-axis gradient head coil. Head movement was restricted using expandable foam cushions. After an automated scout image was acquired and shimming procedures were performed to optimize field homogeneity, 27 high-resolution 3-dimensional magnetizationprepared rapid acquisition gradient echo sequences (repetition time/echo time/flip angle, 7.25 milliseconds/3 milliseconds/ 7 ) with an in-plane resolution of 1.3 mm and 1-mm-slice thickness were collected for spatial normalization and for positioning the slice prescription of the subsequent sequences. Then T1- (repetitition time/echo time/flip angle, 8 seconds/39 milliseconds/90 ) and T2-weighted sequences (repetition time/ echo time/flip angle, 10 seconds/48 milliseconds/120 ) were gathered. Functional MRI images 28 were acquired using a gradient echo T2-weighted sequence (repetition time/echo time/ flip angle, 2.8 seconds/40 milliseconds/90 ). Before each scan, 4 images were acquired and discarded to allow longitudinal magnetization to reach equilibrium. The T1- and T2-weighted and functional images were collected in the same plane (24 coronal slices angled perpendicular to the anterior commissure posterior commissure line) with the same slice thickness (7 mm, skip 1 mm; voxel size, mm), excitation order (interleaved), and phase encoding (foot-to-head). DATA ANALYSIS We performed preprocessing and statistical analysis of the fmri data using a statistical parametric mapping method (SPM99 software package; Wellcome Department of Cognitive Neurology, London, England 29 ). Within SPM99, images were motion corrected (sinc interpolation) and transformed into a standard (Montreal Neurological Institute [MNI] coordinate system; McGill University, Montreal, Quebec) stereotactic space (bilinear interpolation). Images were then smoothed with a 4-mm gaussian kernel. At each voxel, the data were fit to a linear statistical model using the least squares method. The design was modeled using a boxcar function convolved with the hemodynamic response function. Hypotheses were tested as contrasts in which linear compounds of the model parameters were evaluated using t statistics, which were then transformed to z scores. We chose to focus on the fearful vs happy contrast to facilitate comparison of our present results with those of Rauch et al. 12 The fearful vs neutral contrasts yielded results similar to those of the fearful vs happy contrasts but will not be discussed further herein for the sake of brevity. We first computed the voxelwise fearful vs happy contrast within each group, and then assessed our prediction of exaggerated amygdala responses and diminished medial prefrontal responses to fearful vs happy facial expressions in PTSD with a voxelwise test of the condition diagnosis interaction. These analyses were conducted within the first functional run to avoid any habituation or sensitization effects. We chose to conduct the fearful vs happy contrasts within a fixed-effects model because this procedure minimizes type II error. Although fixedeffects analyses limit our ability to generalize from the study sample to the larger population of patients with PTSD, the present findings in the amygdala presented in the Results section) are similar to those of a previous fmri study. 12 Furthermore, random-effects analyses (not shown) revealed similar (although less robust) findings in the amygdala and medial prefrontal regions. To determine whether fmri BOLD signal changes in the amygdala were significantly related to signal changes in the medial prefrontal cortex in PTSD, we (1) defined a functional region of interest (diameter, 6 mm) around the amygdala activations in run 1 in the PTSD group (MNI coordinates,+22, +4, 14; +18, 6, 20; and 20, 8, 18), (2) extracted signal values per condition per subject from that region of interest, 30 (3) calculated the fearful vs happy signal change value per subject, and (4) determined whether those change values were associated with signal changes in other brain areas in the fearful vs happy comparison (using individual subject contrast images) via a voxelwise correlational analysis. To determine the relationship between PTSD symptom severity (CAPS scores) and fearful vs happy signal change in the amygdala and medial prefrontal regions in the PTSD group, we conducted voxelwise correlational analyses within SPM99. We chose to focus specifically on the amygdala and medial prefrontal regions in these correlational analyses. Finally, to examine habituation differences between groups, we compared fearful vs happy responses in early (run 1) vs late (run 4) functional runs. 31,32 STATISTICS The statistical parametric maps resulting from these analyses were inspected for activations in our a priori regions of interest. Given our strong, directional hypotheses, we used a significance threshold of P.001, uncorrected (z score, 3.09) for activations in these regions. Because the procedure of correcting P values based on the region size is biased toward finding significance in small structures, we chose to use a constant significance threshold. For regions about which we had no a priori prediction, we used a more conservative constant significance threshold of P.00001, uncorrected (z score, 4.27). Ratings data are expressed as mean±sd. RESULTS FACIAL EXPRESSION RATINGS Ratings of valence and arousal were submitted to separate 2 3 (diagnosis [PTSD and control] expression [fearful, happy, and neutral]) analyses of variance. With regard to valence, only the main effects of group (F 1,24 =9.3; P=.006) and expression (F 2,48 =64.5; P.001) were significant. The PTSD group gave more negative ratings across all facial expressions than the control group. Post hoc testing showed significant valence ratings differences among all 3 types of facial expressions: fearful ( 1.8±1.4) vs happy (1.7±1.1) (t 25 =9.2; P.001); fearful vs neutral (0.0±0.6) (t 25 =6.0; P.001); and neutral vs happy (t 25 =7.7; P.001). Analysis of the arousal ratings revealed only a significant main effect of expression (F 2,46 =17.9; P.001). Post 275
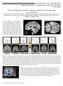
4 Table 1. Fearful vs Happy Comparison in PTSD and Control Groups PTSD Group Control Group Region z Score MNI Coordinates, x, y, z Region z Score MNI Coordinates, x, y, z BOLD Signal Increases Bilateral amygdala , +4, 14 Bilateral amygdala , 6, , 6, , 6, , 8, Right rostral anterior cingulate gyrus , +48, +8 Rostral anterior cingulate gyrus , +38, , +46, , +38, +12 Paracentral cortex , 38, +70 Dorsal anterior cingulate gyrus , +24, +20 Right dorsal medial frontal gyrus , +48, +20 Left hippocampus , 8, 18 Right parahippocampal gyrus , 8, 26 Left middle occipital gyrus , 82, +20 Left inferior temporal gyrus , 22, 18 Right cerebellum , 68, 46 BOLD Signal Decreases Left ventral medial frontal gyrus , +52, 10 Right paracentral cortex , 12, +52 Right rostral anterior cingulate gyrus , +38, +22 Abbreviations: BOLD, blood oxygenation level dependent; MNI, Montreal Neurological Institute; PTSD, posttraumatic stress disorder. A B 0.8 Fearful Happy MR Signal Change PTSD Group Control Figure 1. The functional magnetic resonance (MR) image (A) displays activation to fearful vs happy facial expressions in the amygdala (z=3.14; Montreal Neurological Institute [MNI] coordinates, +22, +2, 14 [arrow]; and z=3.03; MNI coordinates, +22, 0, 26) that were greater in the posttraumatic stress disorder (PTSD) group (n=13) vs control group (n=13) (ie, condition diagnosis interaction). Functional data are superimposed on a standard SPM99 T1 template (Wellcome Department of Cognitive Neurology, London, England), displayed according to neurological convention. The bar graph (B) shows MR signal change in the amygdala (MNI coordinates, +22, +2, 14) in each condition (relative to fixation baseline) for each group. Error bars represent standard error of the mean. hoc testing revealed lower arousal ratings for the neutral faces (1.6±1.3) than for the fearful (3.5±2.0) (t 25 =5.8; P.001) and happy faces (3.1±1.6) (t 24 =4.8; P.001); arousal ratings of the latter 2 expression types did not differ significantly from each other (t 24 =1.1; P=.27). fmri RESULTS Fearful vs Happy Comparison: First Run In the PTSD group, BOLD signal increases occurred in bilateral amygdala and rostral anterior cingulate gyrus (Table 1). BOLD signal decreases occurred in the ventral medial frontal and rostral anterior cingulate gyri. In the control group, BOLD signal increases occurred in bilateral amygdala, rostral and dorsal anterior cingulate gyri, and dorsal medial frontal gyrus, as well as other regions about which we had no specific a priori hypotheses (Table 1). The voxelwise condition diagnosis interaction revealed greater BOLD signal increases in the PTSD group (or greater decreases in the control group) in the right amygdala (Figure 1 and Table 2) among other regions. Greater BOLD signal increases in the control group (or greater decreases in the PTSD group) occurred in the 276
5 rostral anterior cingulate (Figure 2 and Table 2), ventral medial frontal, and dorsal medial frontal gyri. This pattern of results remained when BDI scores were controlled via analysis of covariance and when 4 subjects with PTSD and comorbid major depressive disorder were removed from the analyses. Amygdala/Medial Prefrontal Correlations Within all PTSD participants in the first run, BOLD signal changes in the right amygdala (MNI coordinates, +22, +4, 14) were negatively correlated with BOLD signal changes in the dorsal medial frontal gyrus (z=3.09; MNI coordinates, +14, +50, +16) (Figure 3). BOLD signal changes in the more posterior right amygdala activation (MNI coordinates, +18, 6, 20) were negatively correlated with BOLD signal changes in the dorsal medial frontal gyrus (z=5.30; MNI coordinates, 10, +50, +14). Signal changes in the right and left amygdala were also significantly negatively correlated with ventral medial prefrontal regions; however, the removal of 1 extreme score in each of these latter correlations resulted in subthreshold ( 3.09) z scores. This pattern of results persisted when subjects with PTSD and comorbid depression were removed from the analyses. No such significant correlations were observed in the control group. Symptom Severity Correlations In the PTSD group, PTSD symptom severity (as measured by the CAPS) was negatively correlated with BOLD signal changes in the rostral anterior cingulate gyrus (z=3.62; MNI coordinates, 4, +44, +8) (Figure 4) but was not significantly correlated with signal changes in the amygdala. Depression symptom severity in the PTSD group (as measured by the BDI) was positively correlated with BOLD signal changes in the ventral medial frontal/orbitofrontal gyrus (z=3.60; MNI coordinates, +14, +44, 16) and negatively correlated with signal changes in the right amygdala (z=3.25; MNI coordinates, +28, 4, 16). Thus, the CAPS and BDI appeared to be related very differently to BOLD signal changes in our regions of interest. Habituation Analyses In the control group, fearful vs happy responses decreased from run 1 to run 4 in the right amygdala (z=4.33; MNI coordinates, +18, 6, 18), rostral anterior cingulate gyrus (z=3.18; MNI coordinates, 0, +36, 8), and medial frontal gyrus (z=3.22; MNI coordinates, 12, +56, +4). In the PTSD group, fearful vs happy responses decreased from runs 1 to 4 in the amygdala/periamygdaloid cortex (z=3.00; MNI coordinates, +22, +6, 14) and ventral medial frontal gyrus (z=3.56; MNI coordinates, 6, +48, 16; z=3.25; MNI coordinates, 0, +46, 10). The decline in fearful vs happy responses in the right amygdala tended to be greater in the control vs PTSD group, but did not exceed a priori statistical thresholds (z=2.08; MNI coordinates, +18, 4, 18; z=2.04; MNI coordinates, +26, 0, 16). The decline in fearful vs happy responses in rostral anterior cingulate gyrus was greater Table 2. Condition Diagnosis Interaction Region z Score MNI Coordinates, x, y, z Greater increases in PTSD group or greater decreases in control group Right amygdala , +2, , 0, 26 Right cerebellum , 72, 40 Left posterior cingulate gyrus , 52, +38 Greater decreases in PTSD group or greater increases in control group Bilateral rostral anterior cingulate gyrus Left ventral medial frontal gyrus Right dorsal medial frontal gyrus in the control vs PTSD group (z=3.18; MNI coordinates, 2, +32, 10). COMMENT , +38, , +38, , +52, , +48, +20 Abbreviations: MNI, Montreal Neurological Institute; PTSD, posttraumatic stress disorder. In the present study, the PTSD group exhibited exaggerated amygdala responses and diminished medial prefrontal cortex responses to overtly presented fearful vs happy facial expressions. In addition, only in the PTSD group were BOLD signal changes in the amygdala negatively correlated with signal changes in the medial prefrontal cortex. Furthermore, in the PTSD group, symptom severity was negatively related to BOLD signal changes in the medial prefrontal cortex. Finally, relative to the control group, the PTSD group tended to exhibit diminished habituation of fearful vs happy responses in the right amygdala over functional runs, although this effect did not exceed our a priori statistical threshold. Our finding of relatively greater amygdala responses in PTSD is consistent with previous research using traumatic reminders 1-6 and masked facial expressions. 12 Taken together, the present results and those of Rauch et al 12 show that amygdala hyperresponsivity in PTSD can be demonstrated using facial expression stimuli that are above or below the threshold of conscious recognition. 6 In general, studies using overt facial expressions 8,10,24,25,32 have reported BOLD signal changes in a greater number of brain regions, compared with studies using masked facial expressions. 11,33 Thus, using stimuli that are above the threshold of conscious recognition might afford the ability to examine BOLD signal changes in brain regions other than the amygdala. In the present study, we report relatively diminished BOLD signal changes in medial prefrontal regions in PTSD and a negative correlation between symptom severity and BOLD signal changes in the medial prefrontal cortex. These findings are consistent with those of previous functional 277
6 A B 0.4 Fearful Happy 0.3 MR Signal Change PTSD Group Control Figure 2. The functional magnetic resonance (MR) image (A) shows fearful vs happy signal change in the rostral anterior cingulate gyrus (arrow) (z=3.61; Montreal Neurological Institute [MNI] coordinates, 10, +38, 12) that was greater in the control group than in the posttraumatic stress disorder (PTSD) group (ie, condition diagnosis interaction). Functional data are superimposed on a standard SPM99 T1 template (Wellcome Department of Cognitive Neurology, London, England). The bar graph (B) shows MR signal change in the rostral anterior cingulate gyrus (MNI coordinates, 10, +38, 12) in each condition (relative to fixation baseline) for each group. Error bars represent standard error of the mean. Fearful vs Happy Response in Dorsal Medial Frontal Gyrus r = 0.65 P = Fearful vs Happy Response in Right Amygdala Figure 3. In the posttraumatic stress disorder group, fearful vs happy responses in the right amygdala (Montreal Neurological Institute [MNI] coordinates, +22, +4, 14) were negatively correlated with fearful vs happy responses in dorsal medial frontal gyrus (z=3.09; MNI coordinates, +14, +50, +16) in the first functional run. neuroimaging studies of PTSD using other types of stimuli 5,13-17 and, along with recent reports of structural abnormalities in these regions, further implicate medial prefrontal cortex in the pathophysiology of this disorder. In the PTSD group, we found that BOLD signal changes in the amygdala were negatively correlated with signal changes in the medial frontal gyrus. This finding, which is similar to one recently reported in the context of a symptom provocation paradigm, 5 supports the presence of a reciprocal relationship between these regions, but cannot offer information regarding the direction of causality. Medial prefrontal regions send projections to the amygdala in primates, modulate amygdala output, 41 and play an important role in the process of extinction of fear conditioning Conversely, the amygdala may modulate prefrontal neuronal activity. 45 Although the current finding of a reciprocal relationship between the amygdala and medial prefrontal cortex is consistent with that of a previous report, 5 it also differs from the results of a recent positron emission tomographic symptom provocation study 46 in which multivariate structural equation modeling suggested the possibility of a positive relationship between the amygdala and cingulate/subcallosal cortex blood flow in PTSD. Additional research is needed to further clarify this issue. The present and previous studies suggest functional abnormalities in medial prefrontal regions in PTSD. However, across studies, these functional abnormalities have differed in location within the medial prefrontal cortex, including the medial frontal gyrus and anterior cingulate gyrus. However, it is unlikely that all medial prefrontal regions perform a unitary function. Insights regarding the functional differences between various medial prefrontal regions in humans with PTSD will likely arise from the findings of future basic science studies of conditioning and extinction. 47,48 Analyses of BOLD signal changes from runs 1 to 4 suggested that fearful vs happy responses in the right amygdala decreased in the control group, consistent with the findings of previous studies of habituation in healthy individuals. 31,32,49,50 There was a nonsignificant trend for the PTSD group to show diminished habituation in the right amygdala, compared with the control group. However, we must emphasize that this pattern occurred in only 2 small loci in the right amygdala and that future studies involving larger numbers of subjects are needed to confirm this finding. If replicated, diminished habituation of amygdala responses may be seen as broadly consistent with previous findings of slower habituation of psychophysiologic responses to loud tones in PTSD Habituation of medial prefrontal cortex responses occurred in both groups, although it was significantly greater in the control vs PTSD group in the rostral anterior cingulate gyrus. We attribute this group difference in re- 278
7 A B r = P =.0004 Fearful vs Happy Response in Rostral AC Gyrus CAPS Figure 4. Correlation of posttraumatic stress disorder (PTSD) symptom severity (as assessed by the Clinician-Administered PTSD Scale [CAPS]) with fearful vs happy responses in the rostral anterior cingulate (AC) gyrus (A; arrow) (z=3.62; Montreal Neurological Institute [MNI] coordinates, 4, +44, +8) in the PTSD group. Functional data are superimposed on a standard SPM99 T1 template (Wellcome Department of Cognitive Neurology, London, England). The graph (B) shows negative correlation of CAPS scores with fearful vs happy responses. sponse changes over time to a floor effect: the PTSD group started out (in run 1) with lower rostral anterior cingulate responses compared with the control group, and further decreases were necessarily smaller in magnitude. Although amygdala responses to fearful vs happy faces were bilateral within each group, the condition diagnosis interaction occurred in the right amygdala. Indeed, most 1,3,12,54,55 but not all 2,5 findings of amygdala hyperresponsivity in PTSD have been right-sided. In addition, correlations between PTSD symptom severity and amygdala activity also have been right-sided. 3,5,7 Functional abnormalities in the right amygdala may be associated with a failure of habituation or of sensitization to emotional stimuli. 56 Exaggerated amygdala responses to fearful facial expressions have been observed in PTSD, 12 but not in specific (small-animal) phobia 57 or in social phobia. 58 Further research on panic disorder, obsessive-compulsive disorder, and generalized anxiety disorder would be needed to determine the specificity of these findings to PTSD vs other anxiety disorders. However, individuals with social phobia appear to exhibit exaggerated amygdala responses to neutral faces alone, 59 as well as to contemptuous and angry faces vs happy faces. 58 Thus, whether exaggerated amygdala activation is demonstrated in a particular disorder may depend on the facial expressions used and their relevance to the disorder in question. Fearful faces may be particularly relevant to patients with PTSD, whereas contemptuous, angry, or even ambiguous neutral faces might be particularly relevant to patients with social phobia. Definitive conclusions from the present study are limited by the presence of comorbidity in the PTSD group, although the key results remained significant even after controlling for BDI scores and excluding participants with current major depression. In addition, responsivity of our regions of interest was related to BDI and CAPS scores in very different ways, suggesting that depression symptoms cannot explain the main findings. The relatively small sample size precluded comparisons between firefighters and combat veterans with PTSD. In addition, the participants in this study were all men; whether the findings can be generalized to women with PTSD remains to be determined. Finally, as with any passive viewing paradigm, behavioral data could not be collected or compared between groups. CONCLUSIONS The present results are consistent with exaggerated amygdala responsivity, diminished medial prefrontal cortex responsivity, and a reciprocal relationship between these 2 regions in PTSD. Additional studies are needed to confirm the finding of diminished habituation of amygdala responses over repeated fearful vs happy facial expressions in PTSD, and to determine whether such habituation abnormalities occur in response to other types of stimuli. Submitted for Publication: December 15, 2003; final revision received July 27, 2004; accepted August 20, Author Affiliations: Department of Psychology, Tufts University, Medford, Mass (Drs Shin and Krangel and Ms Cavanagh); Department of Psychiatry, Massachusetts General Hospital and Harvard Medical School (Drs Shin, Wright, Cannistraro, Lasko, Orr, Pitman, and Rauch and Mss Wedig and McMullin), and Brigham Behavioral Neurology Group, Department of Neurology, Brigham and Women s Hospital (Dr Wright), Boston, Mass; Department of Psychiatry, University of Michigan, Ann Arbor (Dr Martis); Veterans Affairs Research Service, Manchester, NH (Drs Lasko and Orr and Mr Macklin); and Departments of Psychiatry and Psychology, University of Wisconsin, Madison (Dr Whalen). Correspondence: Lisa M. Shin, PhD, Department of Psychology, Tufts University, 490 Boston Ave, Medford, MA (lisa.shin@tufts.edu). 279
8 Funding/Support: This study was supported by a Young Investigator Award from the National Alliance for Research on Schizophrenia and Depression, Great Neck, NY (Dr Shin); the Robert Wood Johnson Foundation, Princeton, NJ (Dr Wright); grants MH (Dr Wright) and MH (Dr Rauch) from the National Institute of Mental Health, Bethesda, Md; and a Medical Research Service Merit Review grant from the Department of Veterans Affairs, Washington, DC (Dr Orr). Acknowledgment: We thank Mohammed R. Milad for his comments on this manuscript; the individuals who served as research participants; and Mary Foley, Jennifer Holmes, and Lawrence White for their technical assistance. REFERENCES 1. Rauch SL, van der Kolk BA, Fisler RE, Alpert NM, Orr SP, Savage CR, Fischman AJ, Jenike MA, Pitman RK. A symptom provocation study of posttraumatic stress disorder using positron emission tomography and script-driven imagery. Arch Gen Psychiatry. 1996;53: Liberzon I, Taylor SF, Amdur R, Jung TD, Chamberlain KR, Minoshima S, Koeppe RA, Fig LM. Brain activation in PTSD in response to trauma-related stimuli. Biol Psychiatry. 1999;45: Pissiota A, Frans O, Fernandez M, von Knorring L, Fischer H, Fredrikson M. Neurofunctional correlates of posttraumatic stress disorder: a PET symptom provocation study. Eur Arch Psychiatry Clin Neurosci. 2002;252: Shin LM, Kosslyn SM, McNally RJ, Alpert NM, Thompson WL, Rauch SL, Macklin ML, Pitman RK. Visual imagery and perception in posttraumatic stress disorder: a positron emission tomographic investigation. Arch Gen Psychiatry. 1997; 54: Shin LM, Orr SP, Carson MA, Rauch SL, Macklin ML, Lasko NB, Marzol Peters P, Metzger L, Dougherty DD, Cannistraro PA, Alpert NM, Fischman AJ, Pitman RK. Regional cerebral blood flow in amygdala and medial prefrontal cortex during traumatic imagery in male and female Vietnam veterans with PTSD. Arch Gen Psychiatry. 2004;61: Hendler T, Rotshtein P, Yeshurun Y, Weizmann T, Kahn I, Ben-Bashat D, Malach R, Bleich A. Sensing the invisible: differential sensitivity of visual cortex and amygdala to traumatic context. Neuroimage. 2003;19: Fredrikson M, Furmark T. Amygdaloid regional cerebral blood flow and subjective fear during symptom provocation in anxiety disorders. Ann N Y Acad Sci. 2003;985: Breiter HC, Etcoff NL, Whalen PJ, Kennedy WA, Rauch SL, Buckner RL, Strauss MM, Hyman SE, Rosen BR. Response and habituation of the human amygdala during visual processing of facial expression. Neuron. 1996;17: Morris JS, Frith CD, Perrett DI, Rowland D, Young AW, Calder AJ, Dolan RJ. A differential neural response in the human amygdala to fearful and happy facial expressions. Nature. 1996;383: Phillips ML, Young AW, Senior C, Brammer M, Andrew C, Calder AJ, Bullmore ET, Perrett DI, Rowland D, Williams SC, Gray JA, David AS. A specific neural substrate for perceiving facial expressions of disgust. Nature. 1997;389: Whalen PJ, Rauch SL, Etcoff NL, McInerney SC, Lee MB, Jenike MA. Masked presentations of emotional facial expressions modulate amygdala activity without explicit knowledge. J Neurosci. 1998;18: Rauch SL, Whalen PJ, Shin LM, McInerney SC, Macklin ML, Lasko NB, Orr SP, Pitman RK. Exaggerated amygdala response to masked facial stimuli in posttraumatic stress disorder: a functional MRI study. Biol Psychiatry. 2000;47: Bremner JD, Staib LH, Kaloupek D, Southwick SM, Soufer R, Charney DS. Neural correlates of exposure to traumatic pictures and sound in Vietnam combat veterans with and without posttraumatic stress disorder: a positron emission tomography study. Biol Psychiatry. 1999;45: Bremner JD, Narayan M, Staib LH, Southwick SM, McGlashan T, Charney DS. Neural correlates of memories of childhood sexual abuse in women with and without posttraumatic stress disorder. Am J Psychiatry. 1999;156: Shin LM, McNally RJ, Kosslyn SM, Thompson WL, Rauch SL, Alpert NM, Metzger LJ, Lasko NB, Orr SP, Pitman RK. Regional cerebral blood flow during scriptdriven imagery in childhood sexual abuse-related PTSD: a PET investigation. Am J Psychiatry. 1999;156: Lanius RA, Williamson PC, Densmore M, Boksman K, Gupta MA, Neufeld RW, Gati JS, Menon RS. Neural correlates of traumatic memories in posttraumatic stress disorder: a functional MRI investigation. Am J Psychiatry. 2001;158: Shin LM, Whalen PJ, Pitman RK, Bush G, Macklin ML, Lasko NB, Orr SP, McInerney SC, Rauch SL. An fmri study of anterior cingulate function in posttraumatic stress disorder. Biol Psychiatry. 2001;50: Weathers FW, Keane TM, Davidson JR. Clinician-Administered PTSD Scale: a review of the first ten years of research. Depress Anxiety. 2001;13: Oldfield RC. The assessment and analysis of handedness: the Edinburgh inventory. Neuropsychologia. 1971;9: Beck AT, Steer RA. Manual for the Revised Beck Depression Inventory. San Antonio, Tex: Psychological Corp; Beck AT, Steer RA. Beck Anxiety Inventory Manual. San Antonio, Tex: Psychological Corp; First M, Spitzer R, Gibbon M, Williams J. Structured Clinical Interview for DSM-IV. New York: New York State Psychiatric Institute, Biometrics Research Dept; Ekman P, Friesen WV. Pictures of Facial Affect. Palo Alto, Calif: Consulting Psychologists; Whalen PJ, Shin LM, McInerney SC, Fischer H, Wright CI, Rauch SL. A functional MRI study of human amygdala responses to facial expressions of fear versus anger. Emotion. 2001;1: Lange K, Williams LM, Young AW, Bullmore ET, Brammer MJ, Williams SC, Gray JA, Phillips ML. Task instructions modulate neural responses to fearful facial expressions. Biol Psychiatry. 2003;53: Taylor SF, Phan KL, Decker LR, Liberzon I. Subjective rating of emotionally salient stimuli modulates neural activity. Neuroimage. 2003;18: Reese TG, Davis TL, Weisskoff RM. Automated shimming at 1.5 T using echoplanar image frequency maps. J Magn Reson Imaging. 1995;5: Kwong KK, Belliveau JW, Chesler DA, Goldberg IE, Weisskoff RM, Poncelet BP, Kennedy DN, Hoppel BE, Cohen MS, Turner R, Cheng HM, Brady TJ, Rosen BR. Dynamic magnetic resonance imaging of human brain activity during primary sensory stimulation. Proc Natl Acad Sci U S A. 1992;89: Friston KJ, Frith CD, Liddle PF, Frackowiak RS. Comparing functional (PET) images: the assessment of significant change. J Cereb Blood Flow Metab. 1991; 11: Poldrack RA, Gabrieli JD. Characterizing the neural mechanisms of skill learning and repetition priming: evidence from mirror reading. Brain. 2001;124: Fischer H, Wright CI, Whalen PJ, McInerney SC, Shin LM, Rauch SL. Brain habituation during repeated exposure to fearful and neutral faces: a functional MRI study. Brain Res Bull. 2003;59: Wright CI, Fischer H, Whalen PJ, McInerney SC, Shin LM, Rauch SL. Differential prefrontal cortex and amygdala habituation to repeatedly presented emotional stimuli. Neuroreport. 2001;12: Morris JS, Ohman A, Dolan RJ. Conscious and unconscious emotional learning in the human amygdala. Nature. 1998;393: Rauch SL, Shin LM, Segal E, Pitman RK, Carson MA, McMullin K, Whalen PJ, Makris N. Selectively reduced regional cortical volumes in post-traumatic stress disorder. Neuroreport. 2003;14: De Bellis MD, Keshavan MS, Spencer S, Hall J. N-Acetylaspartate concentration in the anterior cingulate of maltreated children and adolescents with PTSD. Am J Psychiatry. 2000;157: Yamasue H, Kasai K, Iwanami A, Ohtani T, Yamada H, Abe O, Kuroki N, Fukuda R, Tochigi M, Furukawa S, Sadamatsu M, Sasaki T, Aoki S, Ohtomo K, Asukai N, Kato N. Voxel-based analysis of MRI reveals anterior cingulate gray-matter volume reduction in posttraumatic stress disorder due to terrorism. Proc Natl Acad Sci U S A. 2003;100: Aggleton JP, Burton MJ, Passingham RE. Cortical and subcortical afferents to the amygdala of the rhesus monkey (Macaca mulatta). Brain Res. 1980;190: Chiba T, Kayahara T, Nakano K. Efferent projections of infralimbic and prelimbic areas of the medial prefrontal cortex in the Japanese monkey, Macaca fuscata. Brain Res. 2001;888: Stefanacci L, Amaral DG. Some observations on cortical inputs to the macaque monkey amygdala: an anterograde tracing study. J Comp Neurol. 2002;451: Ghashghaei HT, Barbas H. Pathways for emotion: interactions of prefrontal and anterior temporal pathways in the amygdala of the rhesus monkey. Neuroscience. 2002;115: Quirk GJ, Likhtik E, Pelletier JG, Pare D. Stimulation of medial prefrontal cortex decreases the responsiveness of central amygdala output neurons. J Neurosci. 2003;23: Morgan MA, Romanski LM, LeDoux JE. Extinction of emotional learning: contribution of medial prefrontal cortex. Neurosci Lett. 1993;163:
9 43. Quirk GJ, Russo GK, Barron JL, Lebron K. The role of ventromedial prefrontal cortex in the recovery of extinguished fear. J Neurosci. 2000;20: Milad MR, Quirk GJ. Neurons in medial prefrontal cortex signal memory for fear extinction. Nature. 2002;420: Garcia R, Vouimba RM, Baudry M, Thompson RF. The amygdala modulates prefrontal cortex activity relative to conditioned fear. Nature. 1999;402: Gilboa A, Shalev AY, Laor L, Lester H, Louzoun Y, Chisin R, Bonne O. Functional connectivity of the prefrontal cortex and the amygdala in posttraumatic stress disorder. Biol Psychiatry. 2004;55: Barrett D, Shumake J, Jones D, Gonzalez-Lima F. Metabolic mapping of mouse brain activity after extinction of a conditioned emotional response. J Neurosci. 2003;23: Bush G, Luu P, Posner MI. Cognitive and emotional influence in anterior cingulate cortex. Trends Cogn Sci. 2000;4: Phillips ML, Medford N, Young AW, Williams L, Williams SC, Bullmore ET, Gray JA, Brammer MJ. Time courses of left and right amygdalar responses to fearful facial expressions. Hum Brain Mapp. 2001;12: Feinstein JS, Goldin PR, Stein MB, Brown GG, Paulus MP. Habituation of attentional networks during emotion processing. Neuroreport. 2002;13: Orr SP, Lasko NB, Shalev AY, Pitman RK. Physiologic responses to loud tones in Vietnam veterans with posttraumatic stress disorder. J Abnorm Psychol. 1995; 104: Orr SP, Solomon Z, Peri T, Pitman RK, Shalev AY. Physiologic responses to loud tones in Israeli veterans of the 1973 Yom Kippur War. Biol Psychiatry. 1997; 41: Shalev AY, Orr SP, Peri T, Schreiber S, Pitman RK. Physiologic responses to loud tones in Israeli patients with posttraumatic stress disorder. Arch Gen Psychiatry. 1992;49: Driessen M, Beblo T, Mertens M, Piefke M, Rullkoetter N, Silva-Saavedra A, Reddemann L, Rau H, Markowitsch HJ, Wulff H, Lange W, Woermann FG. Posttraumatic stress disorder and fmri activation patterns of traumatic memory in patients with borderline personality disorder. Biol Psychiatry. 2004;55: Semple WE, Goyer PF, McCormick R, Donovan B, Muzic RF Jr, Rugle L, McCutcheon K, Lewis C, Liebling D, Kowaliw S, Vapenik K, Semple MA, Flener CR, Schulz SC. Higher brain blood flow at amygdala and lower frontal cortex blood flow in PTSD patients with comorbid cocaine and alcohol abuse compared with normals. Psychiatry. 2000;63: Rauch SL, Shin LM, Wright CI. Neuroimaging studies of amygdala function in anxiety disorders. Ann N Y Acad Sci. 2003;985: Wright CI, Martis B, McMullin K, Shin LM, Rauch SL. Amygdala and insular responses to emotionally valenced human faces in small animal specific phobia. Biol Psychiatry. 2003;54: Stein MB, Goldin PR, Sareen J, Zorrilla LT, Brown GG. Increased amygdala activation to angry and contemptuous faces in generalized social phobia. Arch Gen Psychiatry. 2002;59: Birbaumer N, Grodd W, Diedrich O, Klose U, Erb M, Lotze M, Schneider F, Weiss U, Flor H. fmri reveals amygdala activation to human faces in social phobics. Neuroreport. 1998;9:
Diminished rostral anterior cingulate cortex activation during trauma-unrelated emotional interference in PTSD
 Offringa et al. Biology of Mood & Anxiety Disorders 2013, 3:10 Biology of Mood & Anxiety Disorders BRIEF REPORT Diminished rostral anterior cingulate cortex activation during trauma-unrelated emotional
Offringa et al. Biology of Mood & Anxiety Disorders 2013, 3:10 Biology of Mood & Anxiety Disorders BRIEF REPORT Diminished rostral anterior cingulate cortex activation during trauma-unrelated emotional
Brain responses to symptom provocation and trauma-related short-term memory recall in coal mining accident survivors with acute severe PTSD
 BRAIN RESEARCH 1144 (2007) 165 174 available at www.sciencedirect.com www.elsevier.com/locate/brainres Research Report Brain responses to symptom provocation and trauma-related short-term memory recall
BRAIN RESEARCH 1144 (2007) 165 174 available at www.sciencedirect.com www.elsevier.com/locate/brainres Research Report Brain responses to symptom provocation and trauma-related short-term memory recall
Methods and Materials
 PRIORITY COMMUNICATION Exaggerated Amygdala Response to Masked Facial Stimuli in Posttraumatic Stress Disorder: A Functional MRI Study Scott L. Rauch, Paul J. Whalen, Lisa M. Shin, Sean C. McInerney, Michael
PRIORITY COMMUNICATION Exaggerated Amygdala Response to Masked Facial Stimuli in Posttraumatic Stress Disorder: A Functional MRI Study Scott L. Rauch, Paul J. Whalen, Lisa M. Shin, Sean C. McInerney, Michael
Neurobehavioral Mechanisms of Fear Generalization in PTSD. Lei Zhang. Duke University, Durham Veteran Affairs Medical Center
 Running head: NEUROBEHAVIORAL MECHANISMS OF FEAR GENERALIZATION 1 Neurobehavioral Mechanisms of Fear Generalization in PTSD Lei Zhang Duke University, Durham Veteran Affairs Medical Center Running head:
Running head: NEUROBEHAVIORAL MECHANISMS OF FEAR GENERALIZATION 1 Neurobehavioral Mechanisms of Fear Generalization in PTSD Lei Zhang Duke University, Durham Veteran Affairs Medical Center Running head:
Procedia - Social and Behavioral Sciences 159 ( 2014 ) WCPCG 2014
 Available online at www.sciencedirect.com ScienceDirect Procedia - Social and Behavioral Sciences 159 ( 2014 ) 743 748 WCPCG 2014 Differences in Visuospatial Cognition Performance and Regional Brain Activation
Available online at www.sciencedirect.com ScienceDirect Procedia - Social and Behavioral Sciences 159 ( 2014 ) 743 748 WCPCG 2014 Differences in Visuospatial Cognition Performance and Regional Brain Activation
Attention-deficit/hyperactivity disorder and dissociative disorder among abused children
 Blackwell Publishing AsiaMelbourne, AustraliaPCNPsychiatry and Clinical Neurosciences1323-13162006 Folia Publishing Society2006604434438Original ArticleADHD and dissociation in abused childrent. Endo et
Blackwell Publishing AsiaMelbourne, AustraliaPCNPsychiatry and Clinical Neurosciences1323-13162006 Folia Publishing Society2006604434438Original ArticleADHD and dissociation in abused childrent. Endo et
Supplementary Information Methods Subjects The study was comprised of 84 chronic pain patients with either chronic back pain (CBP) or osteoarthritis
 Supplementary Information Methods Subjects The study was comprised of 84 chronic pain patients with either chronic back pain (CBP) or osteoarthritis (OA). All subjects provided informed consent to procedures
Supplementary Information Methods Subjects The study was comprised of 84 chronic pain patients with either chronic back pain (CBP) or osteoarthritis (OA). All subjects provided informed consent to procedures
SUPPLEMENT: DYNAMIC FUNCTIONAL CONNECTIVITY IN DEPRESSION. Supplemental Information. Dynamic Resting-State Functional Connectivity in Major Depression
 Supplemental Information Dynamic Resting-State Functional Connectivity in Major Depression Roselinde H. Kaiser, Ph.D., Susan Whitfield-Gabrieli, Ph.D., Daniel G. Dillon, Ph.D., Franziska Goer, B.S., Miranda
Supplemental Information Dynamic Resting-State Functional Connectivity in Major Depression Roselinde H. Kaiser, Ph.D., Susan Whitfield-Gabrieli, Ph.D., Daniel G. Dillon, Ph.D., Franziska Goer, B.S., Miranda
Twelve right-handed subjects between the ages of 22 and 30 were recruited from the
 Supplementary Methods Materials & Methods Subjects Twelve right-handed subjects between the ages of 22 and 30 were recruited from the Dartmouth community. All subjects were native speakers of English,
Supplementary Methods Materials & Methods Subjects Twelve right-handed subjects between the ages of 22 and 30 were recruited from the Dartmouth community. All subjects were native speakers of English,
Supporting Online Material for
 www.sciencemag.org/cgi/content/full/324/5927/646/dc1 Supporting Online Material for Self-Control in Decision-Making Involves Modulation of the vmpfc Valuation System Todd A. Hare,* Colin F. Camerer, Antonio
www.sciencemag.org/cgi/content/full/324/5927/646/dc1 Supporting Online Material for Self-Control in Decision-Making Involves Modulation of the vmpfc Valuation System Todd A. Hare,* Colin F. Camerer, Antonio
Supplementary Online Content
 Supplementary Online Content Redlich R, Opel N, Grotegerd D, et al. Prediction of individual response to electroconvulsive therapy via machine learning on structural magnetic resonance imaging data. JAMA
Supplementary Online Content Redlich R, Opel N, Grotegerd D, et al. Prediction of individual response to electroconvulsive therapy via machine learning on structural magnetic resonance imaging data. JAMA
Previous neuroimaging studies of patients with posttraumatic
 Article The Nature of Traumatic Memories: A 4-T fmri Functional Connectivity Analysis Ruth A. Lanius, M.D., Ph.D. Peter C. Williamson, M.D. Maria Densmore, B.Sc. Kristine Boksman, M.A. R.W. Neufeld, Ph.D.
Article The Nature of Traumatic Memories: A 4-T fmri Functional Connectivity Analysis Ruth A. Lanius, M.D., Ph.D. Peter C. Williamson, M.D. Maria Densmore, B.Sc. Kristine Boksman, M.A. R.W. Neufeld, Ph.D.
Comparing event-related and epoch analysis in blocked design fmri
 Available online at www.sciencedirect.com R NeuroImage 18 (2003) 806 810 www.elsevier.com/locate/ynimg Technical Note Comparing event-related and epoch analysis in blocked design fmri Andrea Mechelli,
Available online at www.sciencedirect.com R NeuroImage 18 (2003) 806 810 www.elsevier.com/locate/ynimg Technical Note Comparing event-related and epoch analysis in blocked design fmri Andrea Mechelli,
Brain Imaging Investigation of the Impairing Effect of Emotion on Cognition
 Brain Imaging Investigation of the Impairing Effect of Emotion on Cognition Gloria Wong 1,2, Sanda Dolcos 1,3, Ekaterina Denkova 1, Rajendra A. Morey 4,5, Lihong Wang 4,5, Nicholas Coupland 1, Gregory
Brain Imaging Investigation of the Impairing Effect of Emotion on Cognition Gloria Wong 1,2, Sanda Dolcos 1,3, Ekaterina Denkova 1, Rajendra A. Morey 4,5, Lihong Wang 4,5, Nicholas Coupland 1, Gregory
Behind the mask: the influence of mask-type on amygdala response to fearful faces
 doi:10.1093/scan/nsq014 SCAN (2010) 5, 363^368 Behind the mask: the influence of mask-type on amygdala response to fearful faces M. Justin Kim, 1 Rebecca A. Loucks, 1 Maital Neta, 1 F. Caroline Davis,
doi:10.1093/scan/nsq014 SCAN (2010) 5, 363^368 Behind the mask: the influence of mask-type on amygdala response to fearful faces M. Justin Kim, 1 Rebecca A. Loucks, 1 Maital Neta, 1 F. Caroline Davis,
State Anxiety Modulation of the Amygdala Response to Unattended Threat-Related Stimuli
 10364 The Journal of Neuroscience, November 17, 2004 24(46):10364 10368 Behavioral/Systems/Cognitive State Anxiety Modulation of the Amygdala Response to Unattended Threat-Related Stimuli Sonia J. Bishop,
10364 The Journal of Neuroscience, November 17, 2004 24(46):10364 10368 Behavioral/Systems/Cognitive State Anxiety Modulation of the Amygdala Response to Unattended Threat-Related Stimuli Sonia J. Bishop,
Authors: Paul A. Frewen, PhD 1,2,*, David J. A. Dozois, PhD 1,2, Richard W. J. Neufeld 1,2,3, &Ruth A. Lanius, MD, PhD 2,3 Departments of Psychology
 Authors: Paul A. Frewen, PhD 1,2,*, David J. A. Dozois, PhD 1,2, Richard W. J. Neufeld 1,2,3, &Ruth A. Lanius, MD, PhD 2,3 Departments of Psychology 1, Psychiatry 2, Neuroscience 3, The University of Western
Authors: Paul A. Frewen, PhD 1,2,*, David J. A. Dozois, PhD 1,2, Richard W. J. Neufeld 1,2,3, &Ruth A. Lanius, MD, PhD 2,3 Departments of Psychology 1, Psychiatry 2, Neuroscience 3, The University of Western
Altered effective connectivity network of the thalamus in post traumatic stress disorder: a resting state FMRI study with granger causality method
 DOI 10.1186/s40535-016-0025-y RESEARCH Open Access Altered effective connectivity network of the thalamus in post traumatic stress disorder: a resting state FMRI study with granger causality method Youxue
DOI 10.1186/s40535-016-0025-y RESEARCH Open Access Altered effective connectivity network of the thalamus in post traumatic stress disorder: a resting state FMRI study with granger causality method Youxue
Literature Review: Posttraumatic Stress Disorder as a Physical Injury Dao 1. Literature Review: Posttraumatic Stress Disorder as a Physical Injury
 Literature Review: Posttraumatic Stress Disorder as a Physical Injury Dao 1 Literature Review: Posttraumatic Stress Disorder as a Physical Injury Amanda Dao University of California, Davis Literature Review:
Literature Review: Posttraumatic Stress Disorder as a Physical Injury Dao 1 Literature Review: Posttraumatic Stress Disorder as a Physical Injury Amanda Dao University of California, Davis Literature Review:
Reproducibility of Visual Activation During Checkerboard Stimulation in Functional Magnetic Resonance Imaging at 4 Tesla
 Reproducibility of Visual Activation During Checkerboard Stimulation in Functional Magnetic Resonance Imaging at 4 Tesla Atsushi Miki*, Grant T. Liu*, Sarah A. Englander, Jonathan Raz, Theo G. M. van Erp,
Reproducibility of Visual Activation During Checkerboard Stimulation in Functional Magnetic Resonance Imaging at 4 Tesla Atsushi Miki*, Grant T. Liu*, Sarah A. Englander, Jonathan Raz, Theo G. M. van Erp,
SUPPLEMENTARY METHODS. Subjects and Confederates. We investigated a total of 32 healthy adult volunteers, 16
 SUPPLEMENTARY METHODS Subjects and Confederates. We investigated a total of 32 healthy adult volunteers, 16 women and 16 men. One female had to be excluded from brain data analyses because of strong movement
SUPPLEMENTARY METHODS Subjects and Confederates. We investigated a total of 32 healthy adult volunteers, 16 women and 16 men. One female had to be excluded from brain data analyses because of strong movement
Methods to examine brain activity associated with emotional states and traits
 Methods to examine brain activity associated with emotional states and traits Brain electrical activity methods description and explanation of method state effects trait effects Positron emission tomography
Methods to examine brain activity associated with emotional states and traits Brain electrical activity methods description and explanation of method state effects trait effects Positron emission tomography
Neuroimaging Studies of Emotional Responses in PTSD
 Neuroimaging Studies of Emotional Responses in PTSD ISRAEL LIBERZON AND BRIAN MARTIS Trauma Stress and Anxiety Research Group, VA Ann Arbor Healthcare System, Department of Psychiatry, University of Michigan,
Neuroimaging Studies of Emotional Responses in PTSD ISRAEL LIBERZON AND BRIAN MARTIS Trauma Stress and Anxiety Research Group, VA Ann Arbor Healthcare System, Department of Psychiatry, University of Michigan,
A review of neuroimaging studies in PTSD: Heterogeneity of response to symptom provocation
 Journal of Psychiatric Research xxx (2005) xxx xxx JOURNAL OF PSYCHIATRIC RESEARCH www.elsevier.com/locate/jpsychires A review of neuroimaging studies in PTSD: Heterogeneity of response to symptom provocation
Journal of Psychiatric Research xxx (2005) xxx xxx JOURNAL OF PSYCHIATRIC RESEARCH www.elsevier.com/locate/jpsychires A review of neuroimaging studies in PTSD: Heterogeneity of response to symptom provocation
Personal Space Regulation by the Human Amygdala. California Institute of Technology
 Personal Space Regulation by the Human Amygdala Daniel P. Kennedy 1, Jan Gläscher 1, J. Michael Tyszka 2 & Ralph Adolphs 1,2 1 Division of Humanities and Social Sciences 2 Division of Biology California
Personal Space Regulation by the Human Amygdala Daniel P. Kennedy 1, Jan Gläscher 1, J. Michael Tyszka 2 & Ralph Adolphs 1,2 1 Division of Humanities and Social Sciences 2 Division of Biology California
Supporting online material. Materials and Methods. We scanned participants in two groups of 12 each. Group 1 was composed largely of
 Placebo effects in fmri Supporting online material 1 Supporting online material Materials and Methods Study 1 Procedure and behavioral data We scanned participants in two groups of 12 each. Group 1 was
Placebo effects in fmri Supporting online material 1 Supporting online material Materials and Methods Study 1 Procedure and behavioral data We scanned participants in two groups of 12 each. Group 1 was
Amy Garrett, Ph.D., Victor Carrion, M.D., and Allan Reiss, M.D. Stanford University School of Medicine Department of Psychiatry
 Amy Garrett, Ph.D., Victor Carrion, M.D., and Allan Reiss, M.D. Stanford University School of Medicine Department of Psychiatry Center for Interdisciplinary Brain Sciences Research and Stanford Early Life
Amy Garrett, Ph.D., Victor Carrion, M.D., and Allan Reiss, M.D. Stanford University School of Medicine Department of Psychiatry Center for Interdisciplinary Brain Sciences Research and Stanford Early Life
Focal brain damage protects against post-traumatic stress disorder in
 Page 1 of 10 Nat Neurosci. Author manuscript; available in PMC 2009 Jun 9. Published in final edited form as: Nat Neurosci. 2008 Feb; 11(2): 232 237. Published online 2007 Dec 23. doi: 10.1038/nn2032 PMCID:
Page 1 of 10 Nat Neurosci. Author manuscript; available in PMC 2009 Jun 9. Published in final edited form as: Nat Neurosci. 2008 Feb; 11(2): 232 237. Published online 2007 Dec 23. doi: 10.1038/nn2032 PMCID:
Left Anterior Prefrontal Activation Increases with Demands to Recall Specific Perceptual Information
 The Journal of Neuroscience, 2000, Vol. 20 RC108 1of5 Left Anterior Prefrontal Activation Increases with Demands to Recall Specific Perceptual Information Charan Ranganath, 1 Marcia K. Johnson, 2 and Mark
The Journal of Neuroscience, 2000, Vol. 20 RC108 1of5 Left Anterior Prefrontal Activation Increases with Demands to Recall Specific Perceptual Information Charan Ranganath, 1 Marcia K. Johnson, 2 and Mark
fmri: What Does It Measure?
 fmri: What Does It Measure? Psychology 355: Cognitive Psychology Instructor: John Miyamoto 04/02/2018: Lecture 02-1 Note: This Powerpoint presentation may contain macros that I wrote to help me create
fmri: What Does It Measure? Psychology 355: Cognitive Psychology Instructor: John Miyamoto 04/02/2018: Lecture 02-1 Note: This Powerpoint presentation may contain macros that I wrote to help me create
Supplemental information online for
 Supplemental information online for Sleep contributes to the strengthening of some memories over others, depending on hippocampal activity at learning. Géraldine Rauchs (1,2), Dorothée Feyers (1), Brigitte
Supplemental information online for Sleep contributes to the strengthening of some memories over others, depending on hippocampal activity at learning. Géraldine Rauchs (1,2), Dorothée Feyers (1), Brigitte
Probing reward function in post-traumatic stress disorder with beautiful facial images
 Psychiatry Research 135 (2005) 179 183 www.elsevier.com/locate/psychres Probing reward function in post-traumatic stress disorder with beautiful facial images Igor Elman a, *, Dan Ariely b, Nina Mazar
Psychiatry Research 135 (2005) 179 183 www.elsevier.com/locate/psychres Probing reward function in post-traumatic stress disorder with beautiful facial images Igor Elman a, *, Dan Ariely b, Nina Mazar
Selective attention to emotional stimuli in a verbal go/no-go task: an fmri study
 BRAIN IMAGING NEUROREPORT Selective attention to emotional stimuli in a verbal go/no-go task: an fmri study Rebecca Elliott, CA Judy S. Rubinsztein, 1 Barbara J. Sahakian 1 and Raymond J. Dolan 2 Neuroscience
BRAIN IMAGING NEUROREPORT Selective attention to emotional stimuli in a verbal go/no-go task: an fmri study Rebecca Elliott, CA Judy S. Rubinsztein, 1 Barbara J. Sahakian 1 and Raymond J. Dolan 2 Neuroscience
Do women with fragile X syndrome have problems in switching attention: Preliminary findings from ERP and fmri
 Brain and Cognition 54 (2004) 235 239 www.elsevier.com/locate/b&c Do women with fragile X syndrome have problems in switching attention: Preliminary findings from ERP and fmri Kim Cornish, a,b, * Rachel
Brain and Cognition 54 (2004) 235 239 www.elsevier.com/locate/b&c Do women with fragile X syndrome have problems in switching attention: Preliminary findings from ERP and fmri Kim Cornish, a,b, * Rachel
Functional Magnetic Resonance Imaging: Application to Posttraumatic Stress Disorder
 WINDOWS TO THE BRAIN Robin A. Hurley, M.D., L. Anne Hayman, M.D., Katherine H. Taber, Ph.D. Section Editors Functional Magnetic Resonance Imaging: Application to Posttraumatic Stress Disorder Katherine
WINDOWS TO THE BRAIN Robin A. Hurley, M.D., L. Anne Hayman, M.D., Katherine H. Taber, Ph.D. Section Editors Functional Magnetic Resonance Imaging: Application to Posttraumatic Stress Disorder Katherine
The Role of Working Memory in Visual Selective Attention
 Goldsmiths Research Online. The Authors. Originally published: Science vol.291 2 March 2001 1803-1806. http://www.sciencemag.org. 11 October 2000; accepted 17 January 2001 The Role of Working Memory in
Goldsmiths Research Online. The Authors. Originally published: Science vol.291 2 March 2001 1803-1806. http://www.sciencemag.org. 11 October 2000; accepted 17 January 2001 The Role of Working Memory in
Supplementary Online Content
 Supplementary Online Content Green SA, Hernandez L, Tottenham N, Krasileva K, Bookheimer SY, Dapretto M. The neurobiology of sensory overresponsivity in youth with autism spectrum disorders. Published
Supplementary Online Content Green SA, Hernandez L, Tottenham N, Krasileva K, Bookheimer SY, Dapretto M. The neurobiology of sensory overresponsivity in youth with autism spectrum disorders. Published
Resistance to forgetting associated with hippocampus-mediated. reactivation during new learning
 Resistance to Forgetting 1 Resistance to forgetting associated with hippocampus-mediated reactivation during new learning Brice A. Kuhl, Arpeet T. Shah, Sarah DuBrow, & Anthony D. Wagner Resistance to
Resistance to Forgetting 1 Resistance to forgetting associated with hippocampus-mediated reactivation during new learning Brice A. Kuhl, Arpeet T. Shah, Sarah DuBrow, & Anthony D. Wagner Resistance to
QUANTIFYING CEREBRAL CONTRIBUTIONS TO PAIN 1
 QUANTIFYING CEREBRAL CONTRIBUTIONS TO PAIN 1 Supplementary Figure 1. Overview of the SIIPS1 development. The development of the SIIPS1 consisted of individual- and group-level analysis steps. 1) Individual-person
QUANTIFYING CEREBRAL CONTRIBUTIONS TO PAIN 1 Supplementary Figure 1. Overview of the SIIPS1 development. The development of the SIIPS1 consisted of individual- and group-level analysis steps. 1) Individual-person
Neural substrates participating in acquisition of facial familiarity: an fmri study
 NeuroImage 20 (2003) 1734 1742 www.elsevier.com/locate/ynimg Neural substrates participating in acquisition of facial familiarity: an fmri study H. Kosaka, a,b, * M. Omori, a T. Iidaka, c T. Murata, a
NeuroImage 20 (2003) 1734 1742 www.elsevier.com/locate/ynimg Neural substrates participating in acquisition of facial familiarity: an fmri study H. Kosaka, a,b, * M. Omori, a T. Iidaka, c T. Murata, a
BOLD signal compartmentalization based on the apparent diffusion coefficient
 Magnetic Resonance Imaging 20 (2002) 521 525 BOLD signal compartmentalization based on the apparent diffusion coefficient Allen W. Song a,b *, Harlan Fichtenholtz b, Marty Woldorff b a Brain Imaging and
Magnetic Resonance Imaging 20 (2002) 521 525 BOLD signal compartmentalization based on the apparent diffusion coefficient Allen W. Song a,b *, Harlan Fichtenholtz b, Marty Woldorff b a Brain Imaging and
PTSD and the brain: What clinicians need to know
 PTSD and the brain: What clinicians need to know Namik Kirlić, MA Elana Newman, PhD Underexplored Territories in Trauma Education: Charting Frontiers for Clinicians and Researchers The University of Tulsa
PTSD and the brain: What clinicians need to know Namik Kirlić, MA Elana Newman, PhD Underexplored Territories in Trauma Education: Charting Frontiers for Clinicians and Researchers The University of Tulsa
Hallucinations and conscious access to visual inputs in Parkinson s disease
 Supplemental informations Hallucinations and conscious access to visual inputs in Parkinson s disease Stéphanie Lefebvre, PhD^1,2, Guillaume Baille, MD^4, Renaud Jardri MD, PhD 1,2 Lucie Plomhause, PhD
Supplemental informations Hallucinations and conscious access to visual inputs in Parkinson s disease Stéphanie Lefebvre, PhD^1,2, Guillaume Baille, MD^4, Renaud Jardri MD, PhD 1,2 Lucie Plomhause, PhD
Supplementary information Detailed Materials and Methods
 Supplementary information Detailed Materials and Methods Subjects The experiment included twelve subjects: ten sighted subjects and two blind. Five of the ten sighted subjects were expert users of a visual-to-auditory
Supplementary information Detailed Materials and Methods Subjects The experiment included twelve subjects: ten sighted subjects and two blind. Five of the ten sighted subjects were expert users of a visual-to-auditory
The previous three chapters provide a description of the interaction between explicit and
 77 5 Discussion The previous three chapters provide a description of the interaction between explicit and implicit learning systems. Chapter 2 described the effects of performing a working memory task
77 5 Discussion The previous three chapters provide a description of the interaction between explicit and implicit learning systems. Chapter 2 described the effects of performing a working memory task
Functional Connectivity in Youth at Risk for Depression. Nikhil Kurapati. University of Michigan
 Functional Connectivity 1 Running Head: FUNCTIONAL CONNECTIVITY Functional Connectivity in Youth at Risk for Depression Nikhil Kurapati University of Michigan A thesis submitted in partial fulfillment
Functional Connectivity 1 Running Head: FUNCTIONAL CONNECTIVITY Functional Connectivity in Youth at Risk for Depression Nikhil Kurapati University of Michigan A thesis submitted in partial fulfillment
Functional MRI Mapping Cognition
 Outline Functional MRI Mapping Cognition Michael A. Yassa, B.A. Division of Psychiatric Neuro-imaging Psychiatry and Behavioral Sciences Johns Hopkins School of Medicine Why fmri? fmri - How it works Research
Outline Functional MRI Mapping Cognition Michael A. Yassa, B.A. Division of Psychiatric Neuro-imaging Psychiatry and Behavioral Sciences Johns Hopkins School of Medicine Why fmri? fmri - How it works Research
Supplementary Online Content
 Supplementary Online Content Gregg NM, Kim AE, Gurol ME, et al. Incidental cerebral microbleeds and cerebral blood flow in elderly individuals. JAMA Neurol. Published online July 13, 2015. doi:10.1001/jamaneurol.2015.1359.
Supplementary Online Content Gregg NM, Kim AE, Gurol ME, et al. Incidental cerebral microbleeds and cerebral blood flow in elderly individuals. JAMA Neurol. Published online July 13, 2015. doi:10.1001/jamaneurol.2015.1359.
Chantal E. Stern,*, Adrian M. Owen, Irene Tracey,*, Rodney B. Look,* Bruce R. Rosen,* and Michael Petrides
 NeuroImage 11, 392 399 (2000) doi:10.1006/nimg.2000.0569, available online at http://www.idealibrary.com on Activity in Ventrolateral and Mid-Dorsolateral Prefrontal Cortex during Nonspatial Visual Working
NeuroImage 11, 392 399 (2000) doi:10.1006/nimg.2000.0569, available online at http://www.idealibrary.com on Activity in Ventrolateral and Mid-Dorsolateral Prefrontal Cortex during Nonspatial Visual Working
Introduction Childhood sexual abuse is an important cause of
 Neural Correlates of Declarative Memory for Emotionally Valenced Words in Women with Posttraumatic Stress Disorder Related to Early Childhood Sexual Abuse J. Douglas Bremner, Meena Vythilingam, Eric Vermetten,
Neural Correlates of Declarative Memory for Emotionally Valenced Words in Women with Posttraumatic Stress Disorder Related to Early Childhood Sexual Abuse J. Douglas Bremner, Meena Vythilingam, Eric Vermetten,
Differential Patterns of Abnormal Activity and Connectivity in the Amygdala Prefrontal Circuitry in Bipolar-I and Bipolar-NOS Youth
 NEW RESEARCH Differential Patterns of Abnormal Activity and Connectivity in the Amygdala Prefrontal Circuitry in Bipolar-I and Bipolar-NOS Youth Cecile D. adouceur, Ph.D., Tiffany Farchione, M.D., Vaibhav
NEW RESEARCH Differential Patterns of Abnormal Activity and Connectivity in the Amygdala Prefrontal Circuitry in Bipolar-I and Bipolar-NOS Youth Cecile D. adouceur, Ph.D., Tiffany Farchione, M.D., Vaibhav
Supplementary Online Content
 1 Supplementary Online Content Miller CH, Hamilton JP, Sacchet MD, Gotlib IH. Meta-analysis of functional neuroimaging of major depressive disorder in youth. JAMA Psychiatry. Published online September
1 Supplementary Online Content Miller CH, Hamilton JP, Sacchet MD, Gotlib IH. Meta-analysis of functional neuroimaging of major depressive disorder in youth. JAMA Psychiatry. Published online September
Overt vs. Covert Responding. Prior to conduct of the fmri experiment, a separate
 Supplementary Results Overt vs. Covert Responding. Prior to conduct of the fmri experiment, a separate behavioral experiment was conducted (n = 16) to verify (a) that retrieval-induced forgetting is observed
Supplementary Results Overt vs. Covert Responding. Prior to conduct of the fmri experiment, a separate behavioral experiment was conducted (n = 16) to verify (a) that retrieval-induced forgetting is observed
Supplementary materials. Appendix A;
 Supplementary materials Appendix A; To determine ADHD diagnoses, a combination of Conners' ADHD questionnaires and a semi-structured diagnostic interview was used(1-4). Each participant was assessed with
Supplementary materials Appendix A; To determine ADHD diagnoses, a combination of Conners' ADHD questionnaires and a semi-structured diagnostic interview was used(1-4). Each participant was assessed with
Brittany Shane Cassidy
 Brittany Shane Cassidy Department of Psychology, Brandeis University Phone: 781-736-3031 415 South St., MS 062 E-mail: bcassidy@brandeis.edu Waltham, MA 02454 EDUCATION Ph.D. (ongoing) M.A. B.A. Psychology
Brittany Shane Cassidy Department of Psychology, Brandeis University Phone: 781-736-3031 415 South St., MS 062 E-mail: bcassidy@brandeis.edu Waltham, MA 02454 EDUCATION Ph.D. (ongoing) M.A. B.A. Psychology
Neuroimaging serves as an effective
 Neurobiology of PTSD: A Review of Neuroimaging Findings Sarah N. Garfinkel, PhD; and Israel Liberzon, MD Sarah N. Garfinkel, PhD, is with the Department of Psychiatry, University of Michigan, Ann Arbor.
Neurobiology of PTSD: A Review of Neuroimaging Findings Sarah N. Garfinkel, PhD; and Israel Liberzon, MD Sarah N. Garfinkel, PhD, is with the Department of Psychiatry, University of Michigan, Ann Arbor.
FREQUENCY DOMAIN HYBRID INDEPENDENT COMPONENT ANALYSIS OF FUNCTIONAL MAGNETIC RESONANCE IMAGING DATA
 FREQUENCY DOMAIN HYBRID INDEPENDENT COMPONENT ANALYSIS OF FUNCTIONAL MAGNETIC RESONANCE IMAGING DATA J.D. Carew, V.M. Haughton, C.H. Moritz, B.P. Rogers, E.V. Nordheim, and M.E. Meyerand Departments of
FREQUENCY DOMAIN HYBRID INDEPENDENT COMPONENT ANALYSIS OF FUNCTIONAL MAGNETIC RESONANCE IMAGING DATA J.D. Carew, V.M. Haughton, C.H. Moritz, B.P. Rogers, E.V. Nordheim, and M.E. Meyerand Departments of
Classical fear conditioning in functional neuroimaging Christian Büchel* and Raymond J Dolan*
 219 Classical fear conditioning in functional neuroimaging Christian Büchel* and Raymond J Dolan* Classical conditioning, the simplest form of associative learning, is one of the most studied paradigms
219 Classical fear conditioning in functional neuroimaging Christian Büchel* and Raymond J Dolan* Classical conditioning, the simplest form of associative learning, is one of the most studied paradigms
Neural activity to positive expressions predicts daily experience of schizophrenia-spectrum symptoms in adults with high social anhedonia
 1 Neural activity to positive expressions predicts daily experience of schizophrenia-spectrum symptoms in adults with high social anhedonia Christine I. Hooker, Taylor L. Benson, Anett Gyurak, Hong Yin,
1 Neural activity to positive expressions predicts daily experience of schizophrenia-spectrum symptoms in adults with high social anhedonia Christine I. Hooker, Taylor L. Benson, Anett Gyurak, Hong Yin,
HUMAN SOCIAL INTERACTION RESEARCH PROPOSAL C8CSNR
 HUMAN SOCIAL INTERACTION RESEARCH PROPOSAL C8CSNR Applicants Principal Investigator Student ID 4036841 Collaborators Name(s) Thomas Straube Institution(s) University of Jena, Germany Title of project:
HUMAN SOCIAL INTERACTION RESEARCH PROPOSAL C8CSNR Applicants Principal Investigator Student ID 4036841 Collaborators Name(s) Thomas Straube Institution(s) University of Jena, Germany Title of project:
Altered Emotional Interference Processing in the Amygdala and Insula in Women with Post- Traumatic Stress Disorder
 University of Missouri, St. Louis IRL @ UMSL Psychology Faculty Works Department of Psychological Sciences 2013 Altered Emotional Interference Processing in the Amygdala and Insula in Women with Post-
University of Missouri, St. Louis IRL @ UMSL Psychology Faculty Works Department of Psychological Sciences 2013 Altered Emotional Interference Processing in the Amygdala and Insula in Women with Post-
doi: /brain/aws024 Brain 2012: 135; Altered brain mechanisms of emotion processing in pre-manifest Huntington s disease
 doi:10.1093/brain/aws024 Brain 2012: 135; 1165 1179 1165 BRAIN A JOURNAL OF NEUROLOGY Altered brain mechanisms of emotion processing in pre-manifest Huntington s disease Marianne J. U. Novak, 1 Jason D.
doi:10.1093/brain/aws024 Brain 2012: 135; 1165 1179 1165 BRAIN A JOURNAL OF NEUROLOGY Altered brain mechanisms of emotion processing in pre-manifest Huntington s disease Marianne J. U. Novak, 1 Jason D.
Ventrolateral Prefrontal Cortex Activation and Attentional Bias in Response to Angry Faces in Adolescents With Generalized Anxiety Disorder
 Article Ventrolateral Prefrontal Cortex Activation and Attentional Bias in Response to Angry Faces in Adolescents With Generalized Anxiety Disorder Christopher S. Monk, Ph.D. Eric E. Nelson, Ph.D. Erin
Article Ventrolateral Prefrontal Cortex Activation and Attentional Bias in Response to Angry Faces in Adolescents With Generalized Anxiety Disorder Christopher S. Monk, Ph.D. Eric E. Nelson, Ph.D. Erin
Investigating directed influences between activated brain areas in a motor-response task using fmri
 Magnetic Resonance Imaging 24 (2006) 181 185 Investigating directed influences between activated brain areas in a motor-response task using fmri Birgit Abler a, 4, Alard Roebroeck b, Rainer Goebel b, Anett
Magnetic Resonance Imaging 24 (2006) 181 185 Investigating directed influences between activated brain areas in a motor-response task using fmri Birgit Abler a, 4, Alard Roebroeck b, Rainer Goebel b, Anett
Visual Awareness and the Detection of Fearful Faces
 Emotion Copyright 2005 by the American Psychological Association 2005, Vol. 5, No. 2, 243 247 1528-3542/05/$12.00 DOI: 10.1037/1528-3542.5.2.243 Visual Awareness and the Detection of Fearful Faces Luiz
Emotion Copyright 2005 by the American Psychological Association 2005, Vol. 5, No. 2, 243 247 1528-3542/05/$12.00 DOI: 10.1037/1528-3542.5.2.243 Visual Awareness and the Detection of Fearful Faces Luiz
Individuals with posttraumatic stress disorder (PTSD) often
 Neural Correlates of Levels of Emotional Awareness During Trauma Script-Imagery in Posttraumatic Stress Disorder PAUL FREWEN, MA, RICHARD D. LANE, MD, PHD, RICHARD W.J. NEUFELD, PHD, MARIA DENSMORE, BSC,
Neural Correlates of Levels of Emotional Awareness During Trauma Script-Imagery in Posttraumatic Stress Disorder PAUL FREWEN, MA, RICHARD D. LANE, MD, PHD, RICHARD W.J. NEUFELD, PHD, MARIA DENSMORE, BSC,
Event-Related fmri and the Hemodynamic Response
 Human Brain Mapping 6:373 377(1998) Event-Related fmri and the Hemodynamic Response Randy L. Buckner 1,2,3 * 1 Departments of Psychology, Anatomy and Neurobiology, and Radiology, Washington University,
Human Brain Mapping 6:373 377(1998) Event-Related fmri and the Hemodynamic Response Randy L. Buckner 1,2,3 * 1 Departments of Psychology, Anatomy and Neurobiology, and Radiology, Washington University,
Statistical parametric mapping
 350 PRACTICAL NEUROLOGY HOW TO UNDERSTAND IT Statistical parametric mapping Geraint Rees Wellcome Senior Clinical Fellow,Institute of Cognitive Neuroscience & Institute of Neurology, University College
350 PRACTICAL NEUROLOGY HOW TO UNDERSTAND IT Statistical parametric mapping Geraint Rees Wellcome Senior Clinical Fellow,Institute of Cognitive Neuroscience & Institute of Neurology, University College
WHAT DOES THE BRAIN TELL US ABOUT TRUST AND DISTRUST? EVIDENCE FROM A FUNCTIONAL NEUROIMAGING STUDY 1
 SPECIAL ISSUE WHAT DOES THE BRAIN TE US ABOUT AND DIS? EVIDENCE FROM A FUNCTIONAL NEUROIMAGING STUDY 1 By: Angelika Dimoka Fox School of Business Temple University 1801 Liacouras Walk Philadelphia, PA
SPECIAL ISSUE WHAT DOES THE BRAIN TE US ABOUT AND DIS? EVIDENCE FROM A FUNCTIONAL NEUROIMAGING STUDY 1 By: Angelika Dimoka Fox School of Business Temple University 1801 Liacouras Walk Philadelphia, PA
Emotion I: General concepts, fear and anxiety
 C82NAB Neuroscience and Behaviour Emotion I: General concepts, fear and anxiety Tobias Bast, School of Psychology, University of Nottingham 1 Outline Emotion I (first part) Studying brain substrates of
C82NAB Neuroscience and Behaviour Emotion I: General concepts, fear and anxiety Tobias Bast, School of Psychology, University of Nottingham 1 Outline Emotion I (first part) Studying brain substrates of
APNA 25th Annual Conference October 19, Session 1022
 When Words Are Not Enough The Use of Sensory Modulation Techniques to Replace Self- Injurious Behaviors in Patients with Borderline Personality Disorder General Organization of the Brain Lita Sabonis,
When Words Are Not Enough The Use of Sensory Modulation Techniques to Replace Self- Injurious Behaviors in Patients with Borderline Personality Disorder General Organization of the Brain Lita Sabonis,
Brain Mapping of Episodic Memory in Patients with Medial Temporal Lobe Epilepsy Using Activation Positron Emission Tomography
 Brain Mapping of Episodic Memory in Patients with Medial Temporal Lobe Epilepsy Using Activation Positron Emission Tomography Hyunwoo Nam, M.D., Sang-Kun Lee, M.D., Dong Soo Lee, M.D.*, Jae Sung Lee, M.S.*,
Brain Mapping of Episodic Memory in Patients with Medial Temporal Lobe Epilepsy Using Activation Positron Emission Tomography Hyunwoo Nam, M.D., Sang-Kun Lee, M.D., Dong Soo Lee, M.D.*, Jae Sung Lee, M.S.*,
Activation of the left amygdala to a cognitive representation of fear
 Activation of the left amygdala to a cognitive representation of fear Elizabeth A. Phelps 1, Kevin J. O Connor 2, J. Christopher Gatenby 3, John C. Gore 3, Christian Grillon 4 and Michael Davis 5 1 Department
Activation of the left amygdala to a cognitive representation of fear Elizabeth A. Phelps 1, Kevin J. O Connor 2, J. Christopher Gatenby 3, John C. Gore 3, Christian Grillon 4 and Michael Davis 5 1 Department
smokers) aged 37.3 ± 7.4 yrs (mean ± sd) and a group of twelve, age matched, healthy
 Methods Participants We examined a group of twelve male pathological gamblers (ten strictly right handed, all smokers) aged 37.3 ± 7.4 yrs (mean ± sd) and a group of twelve, age matched, healthy males,
Methods Participants We examined a group of twelve male pathological gamblers (ten strictly right handed, all smokers) aged 37.3 ± 7.4 yrs (mean ± sd) and a group of twelve, age matched, healthy males,
Integration of diverse information in working memory within the frontal lobe
 articles Integration of diverse information in working memory within the frontal lobe V. Prabhakaran 1, K. Narayanan 2, Z. Zhao 2 and J. D. E. Gabrieli 1,2 1 Program in Neurosciences and 2 Dept. of Psychology,
articles Integration of diverse information in working memory within the frontal lobe V. Prabhakaran 1, K. Narayanan 2, Z. Zhao 2 and J. D. E. Gabrieli 1,2 1 Program in Neurosciences and 2 Dept. of Psychology,
Functional organization of spatial and nonspatial working memory processing within the human lateral frontal cortex
 Proc. Natl. Acad. Sci. USA Vol. 95, pp. 7721 7726, June 1998 Neurobiology Functional organization of spatial and nonspatial working memory processing within the human lateral frontal cortex ADRIAN M. OWEN*,
Proc. Natl. Acad. Sci. USA Vol. 95, pp. 7721 7726, June 1998 Neurobiology Functional organization of spatial and nonspatial working memory processing within the human lateral frontal cortex ADRIAN M. OWEN*,
Presupplementary Motor Area Activation during Sequence Learning Reflects Visuo-Motor Association
 The Journal of Neuroscience, 1999, Vol. 19 RC1 1of6 Presupplementary Motor Area Activation during Sequence Learning Reflects Visuo-Motor Association Katsuyuki Sakai, 1,2 Okihide Hikosaka, 1 Satoru Miyauchi,
The Journal of Neuroscience, 1999, Vol. 19 RC1 1of6 Presupplementary Motor Area Activation during Sequence Learning Reflects Visuo-Motor Association Katsuyuki Sakai, 1,2 Okihide Hikosaka, 1 Satoru Miyauchi,
Functional Magnetic Resonance Imaging
 Todd Parrish, Ph.D. Director of the Center for Advanced MRI Director of MR Neuroimaging Research Associate Professor Department of Radiology Northwestern University toddp@northwestern.edu Functional Magnetic
Todd Parrish, Ph.D. Director of the Center for Advanced MRI Director of MR Neuroimaging Research Associate Professor Department of Radiology Northwestern University toddp@northwestern.edu Functional Magnetic
Supplemental Data. Inclusion/exclusion criteria for major depressive disorder group and healthy control group
 1 Supplemental Data Inclusion/exclusion criteria for major depressive disorder group and healthy control group Additional inclusion criteria for the major depressive disorder group were: age of onset of
1 Supplemental Data Inclusion/exclusion criteria for major depressive disorder group and healthy control group Additional inclusion criteria for the major depressive disorder group were: age of onset of
Functional dissociation of amygdala-modulated arousal and cognitive appraisal, in Turner syndrome
 doi:10.1093/brain/awh562 Brain (2005), 128, 2084 2096 Functional dissociation of amygdala-modulated arousal and cognitive appraisal, in Turner syndrome D. H. Skuse, 1 J. S. Morris 1 and R. J. Dolan 2 1
doi:10.1093/brain/awh562 Brain (2005), 128, 2084 2096 Functional dissociation of amygdala-modulated arousal and cognitive appraisal, in Turner syndrome D. H. Skuse, 1 J. S. Morris 1 and R. J. Dolan 2 1
The Neuroscientist. Posttraumatic Stress Disorder: The Role of Medial Prefrontal Cortex and Amygdala
 The Neuroscientist http://nro.sagepub.com Posttraumatic Stress Disorder: The Role of Medial Prefrontal Cortex and Amygdala Michael Koenigs and Jordan Grafman Neuroscientist 2009; 15; 540 originally published
The Neuroscientist http://nro.sagepub.com Posttraumatic Stress Disorder: The Role of Medial Prefrontal Cortex and Amygdala Michael Koenigs and Jordan Grafman Neuroscientist 2009; 15; 540 originally published
Involvement of both prefrontal and inferior parietal cortex. in dual-task performance
 Involvement of both prefrontal and inferior parietal cortex in dual-task performance Fabienne Collette a,b, Laurence 01ivier b,c, Martial Van der Linden a,d, Steven Laureys b, Guy Delfiore b, André Luxen
Involvement of both prefrontal and inferior parietal cortex in dual-task performance Fabienne Collette a,b, Laurence 01ivier b,c, Martial Van der Linden a,d, Steven Laureys b, Guy Delfiore b, André Luxen
Men fear other men most: Gender specific brain activations in. perceiving threat from dynamic faces and bodies. An fmri. study.
 Men fear other men most: Gender specific brain activations in perceiving threat from dynamic faces and bodies. An fmri study. Kret, ME, Pichon, S 2,4, Grèzes, J 2, & de Gelder, B,3 Cognitive and Affective
Men fear other men most: Gender specific brain activations in perceiving threat from dynamic faces and bodies. An fmri study. Kret, ME, Pichon, S 2,4, Grèzes, J 2, & de Gelder, B,3 Cognitive and Affective
Brain Imaging Applied to Memory & Learning
 Brain Imaging Applied to Memory & Learning John Gabrieli Department of Brain & Cognitive Sciences Institute for Medical Engineering & Sciences McGovern Institute for Brain Sciences MIT Levels of Analysis
Brain Imaging Applied to Memory & Learning John Gabrieli Department of Brain & Cognitive Sciences Institute for Medical Engineering & Sciences McGovern Institute for Brain Sciences MIT Levels of Analysis
Individual Differences in Psychophysiological Reactivity in Adults with Childhood Abuse
 Clinical Psychology and Psychotherapy Clin. Psychol. Psychother. 9, 7 76 () Individual Differences in Psychophysiological Reactivity in Adults with Childhood Abuse Christian G. Schmahl, Bernet M. Elzinga
Clinical Psychology and Psychotherapy Clin. Psychol. Psychother. 9, 7 76 () Individual Differences in Psychophysiological Reactivity in Adults with Childhood Abuse Christian G. Schmahl, Bernet M. Elzinga
Event-Related Activation in the Human Amygdala Associates with Later Memory for Individual Emotional Experience
 The Journal of Neuroscience, 2000, Vol. 20 RC99 1of5 Event-Related Activation in the Human Amygdala Associates with Later Memory for Individual Emotional Experience Turhan Canli, 1 Zuo Zhao, 1 James Brewer,
The Journal of Neuroscience, 2000, Vol. 20 RC99 1of5 Event-Related Activation in the Human Amygdala Associates with Later Memory for Individual Emotional Experience Turhan Canli, 1 Zuo Zhao, 1 James Brewer,
Funding: NIDCF UL1 DE019583, NIA RL1 AG032119, NINDS RL1 NS062412, NIDA TL1 DA
 The Effect of Cognitive Functioning, Age, and Molecular Variables on Brain Structure Among Carriers of the Fragile X Premutation: Deformation Based Morphometry Study Naomi J. Goodrich-Hunsaker*, Ling M.
The Effect of Cognitive Functioning, Age, and Molecular Variables on Brain Structure Among Carriers of the Fragile X Premutation: Deformation Based Morphometry Study Naomi J. Goodrich-Hunsaker*, Ling M.
Identification of Neuroimaging Biomarkers
 Identification of Neuroimaging Biomarkers Dan Goodwin, Tom Bleymaier, Shipra Bhal Advisor: Dr. Amit Etkin M.D./PhD, Stanford Psychiatry Department Abstract We present a supervised learning approach to
Identification of Neuroimaging Biomarkers Dan Goodwin, Tom Bleymaier, Shipra Bhal Advisor: Dr. Amit Etkin M.D./PhD, Stanford Psychiatry Department Abstract We present a supervised learning approach to
Common and distinct neural responses during direct and incidental processing of multiple facial emotions
 NeuroImage 20 (2003) 84 97 www.elsevier.com/locate/ynimg Common and distinct neural responses during direct and incidental processing of multiple facial emotions J.S. Winston,* J. O Doherty, and R.J. Dolan
NeuroImage 20 (2003) 84 97 www.elsevier.com/locate/ynimg Common and distinct neural responses during direct and incidental processing of multiple facial emotions J.S. Winston,* J. O Doherty, and R.J. Dolan
The effects of single-trial averaging upon the spatial extent of fmri activation
 BRAIN IMAGING NEUROREPORT The effects of single-trial averaging upon the spatial extent of fmri activation Scott A. Huettel,CA and Gregory McCarthy, Brain Imaging and Analysis Center, Duke University Medical
BRAIN IMAGING NEUROREPORT The effects of single-trial averaging upon the spatial extent of fmri activation Scott A. Huettel,CA and Gregory McCarthy, Brain Imaging and Analysis Center, Duke University Medical
Michio Nomura, a, * Hideki Ohira, a Kaoruko Haneda, a Tetsuya Iidaka, a Norihiro Sadato, b Tomohisa Okada, b and Yoshiharu Yonekura c
 Functional association of the amygdala and ventral prefrontal cortex during cognitive evaluation of facial expressions primed by masked angry faces: an event-related fmri study Michio Nomura, a, * Hideki
Functional association of the amygdala and ventral prefrontal cortex during cognitive evaluation of facial expressions primed by masked angry faces: an event-related fmri study Michio Nomura, a, * Hideki
SUPPLEMENTARY MATERIAL. Table. Neuroimaging studies on the premonitory urge and sensory function in patients with Tourette syndrome.
 SUPPLEMENTARY MATERIAL Table. Neuroimaging studies on the premonitory urge and sensory function in patients with Tourette syndrome. Authors Year Patients Male gender (%) Mean age (range) Adults/ Children
SUPPLEMENTARY MATERIAL Table. Neuroimaging studies on the premonitory urge and sensory function in patients with Tourette syndrome. Authors Year Patients Male gender (%) Mean age (range) Adults/ Children
Altered Activity and Functional Connectivity of Superior Temporal Gyri in Anxiety Disorders: A Functional Magnetic Resonance Imaging Study
 Original Article Neuroimaging and Head & Neck http://dx.doi.org/10.3348/kjr.2014.15.4.523 pissn 1229-6929 eissn 2005-8330 Korean J Radiol 2014;15(4):523-529 Altered Activity and Functional Connectivity
Original Article Neuroimaging and Head & Neck http://dx.doi.org/10.3348/kjr.2014.15.4.523 pissn 1229-6929 eissn 2005-8330 Korean J Radiol 2014;15(4):523-529 Altered Activity and Functional Connectivity
STRESS, MEMORY AND BOSNIAN WAR VETERANS
 & STRESS, MEMORY AND BOSNIAN WAR VETERANS Aida Sarač Hadžihalilović ¹*, Amela Kulenović¹, Abdulah Kučukalić² ¹ Department for Anatomy, Faculty of Medicine, University of Sarajevo, Čekaluša 90, 71000 Sarajevo,
& STRESS, MEMORY AND BOSNIAN WAR VETERANS Aida Sarač Hadžihalilović ¹*, Amela Kulenović¹, Abdulah Kučukalić² ¹ Department for Anatomy, Faculty of Medicine, University of Sarajevo, Čekaluša 90, 71000 Sarajevo,
Fear extinction in rats: Implications for human brain imaging and anxiety disorders
 Biological Psychology 73 (2006) 61 71 www.elsevier.com/locate/biopsycho Fear extinction in rats: Implications for human brain imaging and anxiety disorders Mohammed R. Milad a, Scott L. Rauch a, Roger
Biological Psychology 73 (2006) 61 71 www.elsevier.com/locate/biopsycho Fear extinction in rats: Implications for human brain imaging and anxiety disorders Mohammed R. Milad a, Scott L. Rauch a, Roger
Functional Elements and Networks in fmri
 Functional Elements and Networks in fmri Jarkko Ylipaavalniemi 1, Eerika Savia 1,2, Ricardo Vigário 1 and Samuel Kaski 1,2 1- Helsinki University of Technology - Adaptive Informatics Research Centre 2-
Functional Elements and Networks in fmri Jarkko Ylipaavalniemi 1, Eerika Savia 1,2, Ricardo Vigário 1 and Samuel Kaski 1,2 1- Helsinki University of Technology - Adaptive Informatics Research Centre 2-
DIFFERENT BOLD RESPONSES TO EMOTIONAL FACES AND EMOTIONAL FACES AUGMENTED BY CONTEXTUAL INFORMATION. Kyung Hwa Lee
 DIFFERENT BOLD RESPONSES TO EMOTIONAL FACES AND EMOTIONAL FACES AUGMENTED BY CONTEXTUAL INFORMATION by Kyung Hwa Lee B.A., Chungnam National University, 1997 M.A., Chungnam National University, 1999 Submitted
DIFFERENT BOLD RESPONSES TO EMOTIONAL FACES AND EMOTIONAL FACES AUGMENTED BY CONTEXTUAL INFORMATION by Kyung Hwa Lee B.A., Chungnam National University, 1997 M.A., Chungnam National University, 1999 Submitted
Reducing Risk and Preventing Violence, Trauma, and the Use of Seclusion and Restraint Neurobiological & Psychological Effects of Trauma
 Reducing Risk and Preventing Violence, Trauma, and the Use of Seclusion and Restraint Neurobiological & Psychological Effects of Trauma Module created by Glenn Saxe, MD: 2002 revised 2009, 2011, 2013,
Reducing Risk and Preventing Violence, Trauma, and the Use of Seclusion and Restraint Neurobiological & Psychological Effects of Trauma Module created by Glenn Saxe, MD: 2002 revised 2009, 2011, 2013,
For better or for worse: neural systems supporting the cognitive down- and up-regulation of negative emotion
 For better or for worse: neural systems supporting the cognitive down- and up-regulation of negative emotion Kevin N. Ochsner, a, * Rebecca D. Ray, b Jeffrey C. Cooper, b Elaine R. Robertson, b Sita Chopra,
For better or for worse: neural systems supporting the cognitive down- and up-regulation of negative emotion Kevin N. Ochsner, a, * Rebecca D. Ray, b Jeffrey C. Cooper, b Elaine R. Robertson, b Sita Chopra,
