Diagnosis of Adrenal Cortical Dysfunction by Liquid Chromatography-Tandem Mass Spectrometry
|
|
|
- Jerome Strickland
- 5 years ago
- Views:
Transcription
1 Annals of Clinical & Laboratory Science, vol. 31, no. 2, Diagnosis of Adrenal Cortical Dysfunction by Liquid Chromatography-Tandem Mass Spectrometry Pai C. Kao, Dwaine A. Machacek, Mark J. Magera, Jean M. Lacey, and Piero Rinaldo Department of Laboratory Medicine and Pathology, Mayo Clinic, Rochester, Minnesota Abstract. Liquid chromatography-tandem mass spectrometry (LC-MS/MS) was used to measure 6 metabolic compounds of the adrenocorticosteroid pathway simultaneously on residual specimens from patients who had previously been previously diagnosed, on the basis of immunoassays, as having congenital adrenal hyperplasia (CAH), 11 β-hydroxylase deficiency, 21-hydroxylase deficiency, or Addison disease (adrenal insufficiency). Two subjects with normal adrenal function had serum cortisol values of 13.6 and 8.9 µg/dl and serum cortisone values of 2.1 and 0.6 µg/dl, but the rest of the compounds were undetectable. Two patients with 11 β-hydroxylase deficiency had serum 11 β-deoxycortisol values of 14.9 and 10.0 µg/dl and serum 11-deoxycorticosterone values of 3.9 and 1.0 µg/dl, but their serum levels of cortisol and cortisone were diminished. A patient with 21- hydroxylase deficiency had a highly increased serum 17-hydroxyprogesterone concentration of 28.5 µg/dl (or 28,500 ng/dl, the traditional unit to report this assay) and a serum 21-deoxycortisol concentration of 6.9 ug/dl (this is a pathologic marker of 21-hydroxylase deficiency that is nondetectable in sera of healthy subjects). This patient also had diminished concentrations of serum cortisol and cortisone (0.9 and 0.3 µg/dl, respectively). At 30 and 60 min after corticotropin (ACTH) stimulation, serum cortisol was the only compound that showed a dramatic increase in the normal subjects; the patient with 21-hydroxylase deficiency showed an increase of serum 17-hydroxyprogesterone level, but no increase of serum cortisol level; the patient with Addison disease showed no increase in the levels of serum cortisol or other compounds. Metyrapone, which blocks 11 β-hydroxylase activity, increased the serum 11-deoxycorticosteroid levels and decreased the serum cortisol level. This pilot study demonstrates that it is feasible to use LC-MS/MS for the laboratory diagnosis of adrenal cortical dysfunction. The authors envision that LC-MS/MS may soon become an ideal analytical technique for the diagnosis of such endocrine diseases. (received 1 November 2000, accepted 6 February 2001) Keywords: Liquid chromatography-tandem mass spectrometry, serum steroid analysis, adrenal cortical dysfunction Introduction New analytical technologies will change clinical laboratory operations in the new millennium. In an article on future trends in clinical chemistry, Parker [1] recently commented that flow cytometry and mass spectrometry are currently peripheral; they will become mainstream technologies tomorrow. A pertinent question is, When is the tomorrow? This pilot study suggests that the tomorrow is very soon and may be Address correspondence to Pai C. Kao, Ph.D., Department of Laboratory Medicine and Pathology, Mayo Clinic, 200 First Street SW, Rochester, MN 55905, USA; tel: ; fax: ; kao.pai@mayo.edu within the next five years. Currently, liquid chromatography-tandem mass spectrometry (LC-MS/ MS) is available in only a few medical centers. To make LC-MS/MS a mainstream technology that will benefit the entire medical community, instrument manufacturers should take the initiative to introduce relevant clinical tests for this instrumentation and to demonstrate its practical clinical applications. A good candidate for such applications is the use of LC-MS/MS to obtain multiple test results for complicated adrenal disorders. Performing multiple analyses of hormones in a single determination has long been a goal of our laboratory. In the past, many clinically useful endocrine tests were developed that /01/0200/0197 $1.50; 2001 by the Association of Clinical Scientists, Inc.
2 200 Annals of Clinical & Laboratory Science provided multiple analyses (eg, 25-hydroxyvitamin D and 1,25-dihydroxyvitamin D, cortisol and cortisone, and the catecholamines) by high-performance liquid chromatography (HPLC). In these methods, only 2 or 3 analytes could be detected using spectrometric or amperometric detectors [2-4]. LC-MS/MS can be considered as an HPLC apparatus that is equipped with a mass spectrometric detector that measures the mass and charge ratios (m/z) of analytes and their fragments. Numerous analytes can now be measured simultaneously by LC-MS/MS with positive identification. Immunoassays lack positive identification, even with the use of specific antibodies, and occasionally they can give equivocal results owing to crossreactivities, or the presence of human anti-animal antibodies in a patient s serum [5-7]. In particular, modern, highly sensitive, immunometric assays, which require no preanalytical extraction to concentrate and purify the analyte, tend to magnify the problems of nonspecificity and interferences. Because LC-MS/MS can provide multiple determinations in a single step, it simplifies a laboratory s operation and helps to avoid an excessive amount of blood being drawn from a patient [8]. All of the critical steroid metabolites in serum can be analyzed simultaneously in a single determination, and the frequency and volume of blood collection are substantially reduced. The LC-MS/MS technology is also economical, as demonstrated by cost accounting studies of other assays. In a study of homocysteine assays, the relative costs per test, including supplies and equipment, using LC-MS/MS, HPLC, or a commercial immunoassay kit, were estimated to be 1.00, 1.15, and 5.08, respectively [9]. Cost-effectiveness of LC-MS/MS was also demonstrated in assays of methylmalonic acid [10]. Materials and Methods Specimens for analysis. This study used residual specimens of serum or plasma (~0.5 ml) from patients who had previously been diagnosed, on the basis of immunoassays, as having adrenal disorders (eg, 11 β- hydroxylase deficiency, 21-hydroxylase deficiency, Addison disease). Morning fasting serum specimens from 36 healthy subjects, including 11 men (28 to 68 yr) and 25 women (25 to 61 yr), were also analyzed. This project was approved by the Mayo Clinic s Institutional Review Board. Instrumentation and reagents. The tandem mass spectrometer (API 2000, PE Biosystems, Foster City, CA) included an electrospray ionization device, a triplequadruple mass spectrometer, and an electron multiplier detector. Cortisol, cortisone, 17-hydroxyprogesterone, 21-deoxycortisol, 11-deoxycortisol, 11- deoxycorticosterone were from Sigma Chemical Co. (St. Louis, MO). Deuterium-labeled cortisol (cortisol- 9,-11,-12,-12,-d4) was from Cambridge Isotope Labs (Andover, MA). HPLC grade methanol and methylene chloride were from Fisher Scientific (Pittsburgh, PA). Steroid stock standards were prepared in 100% methanol and stored at -20 C. The reverse-phase chromatographic column was Supelcosil LC-18 (33 x 4.6 mm) from Supelco (Bellefonte, PA). Sample preparation. Serum or plasma (0.5 ml) was placed in a 10 x 75-mm glass tube and mixed with 7 µl of 10 µg/ml cortisol-d4. For extraction, methylene chloride (4.5 ml) was dispensed into each tube; the tube was capped, mixed gently on a rotating mixer for 30 min, and centrifuged at 2500 rpm for 5 min. The upper aqueous layer was removed by aspiration; the lower organic phase was evaporated to dryness under nitrogen gas at 45 C. The dried extract was reconstituted with 125 µl of a 70:30 (v/v) methanolwater mixture, which was transferred to a screw-capped injection vial. A 12-point calibration curve containing each steroid to be tested, with concentrations from 0.5 to 25 µg/dl, was prepared prior to analysis. A 17-µL injection volume was used for standards and samples. LC-MS/MS analysis. Reverse-phase LC, which separated each steroid in the sample, involved a 10- min analysis with a linear gradient (53% to 75%) of methanol-water at a flow rate of 1 ml/min. The chromatographic column was connected directly to the ion source of the TurboIonSpray (PE Biosystems) for ionization probe; the column effluent flow was split to deliver approximately 100 µl/min to the mass spectrometer. The instrument was set to use 3 separate scan periods over a 10-min running time. The positive ion mode was used throughout the analysis. The following software was used: Mass-Chrom (version
3 Liquid chromatography-tandem mass spectrometry for adrenal diagnosis 201 Table 1. Metabolites of the adrenal steroid biosynthetic pathway measured in serum (µg/dl) by LC/MS/MS. a Test subjects Cortisol Cortisone 11 β-deoxy- Deoxycortico- 17-Hydroxy- 21-Deoxcortisol cortisol sterone (DOC) progesterone Healthy subjects b Subject # (19) 2.1 nd nd nd nd Subject # trace nd nd nd 11 β-hydroxylase deficiency Patient #1 0.5 (4.7) nd 14.9 (15) c nd Patient #2 0.5 (3.9) nd 10.0 (13) c nd 21-Hydroxylase deficiency Patient #1 0.9 (7.0) (15.9) 6.9 a The numbers in parentheses are results that were previously obtained by immunoassays to establish the diagnoses. The reference values for the immunoassays are: cortisol (am specimens), 7 to 25 µg/dl; deoxycorticosteroids (a combined measurement of 11 β-deoxycortisol and DOC) c, 0 to 5 µg/dl; 17-hydroxyprogesterone, <0.2 µg/dl. b Assays of sera from 36 healthy subjects (fasting morning specimens) gave the following results (mean ± SD): cortisol 14.3 ± 8.1 µg/dl; cortisone 2.2 ± 0.6 µg/dl; the other four steroid compounds were nd (not detected) ) and Multi-View (version 1.4) for chromatographic and spectral interpretations, and TurboIon- Quan (version 1.0, PE Biosystems) for data processing on a Macintosh computer [11]. The sensitivity for cortisol detection using d4-cortisol calibration was 0.1 µg/dl. The sensitivities of the other metabolites were 0.5 µg/dl. The assay CVwas <5%, similar to HPLC. Results Healthy subjects. Of 2 healthy subjects, one had serum cortisol and cortisone values of 13.6 µg/dl and 2.1 µg/dl, respectively. The other had a serum cortisol value of 8.9 µg/dl, a serum cortisone value of 0.6 µg/ dl, and a trace of 11-deoxycortisol (<0.5 µg/dl (Table 1). In serum specimens from 36 healthy subjects, the cortisol concentration (mean ± SD) was 14.3 ± 8.1 µg/dl (range 2 to 30 µg/dl). The cortisone concentration was 2.2 ± 0.6 µg/dl (range 0.2 to 3.2 µg/dl). The remaining steroid compounds were undetectable in serum specimens from healthy subjects.. 11 β-hydroxylase deficiency. Two patients with 11 β- hydroxylase deficiency had only trace amounts of serum cortisol (0.5 µg/dl) and undetectable amounts of cortisone, but they had high levels of 11 β-deoxycortisol (14.9 and 10.0 µg/dl, respectively). These patients had serum 11-deoxycorticosterone values of 3.9 and 1.0 µg/dl and measurable amounts of serum 17- hydroxyprogesterone (0.9 and 1.2 µg/dl) (Table 1). 21-Hydroxylase deficiency. A patient with 21- hydroxylase deficiency had a high level of serum 17- hydroxyprogesterone (28.5 µg/dl, ie, 28,500 ng/dl) and trace amounts of cortisol (0.9 µg/dl) and cortisone (0.3 µg/dl). A unique compound, 21-deoxycortisol, is found only in serum from patients with 21- hydroxylase deficiency. 21-Deoxycortisol is a marker of 21-hydroxylase deficiency and is not a normal precursor in the pathway of adrenocortical hormone biosynthesis. The concentration of serum 21- deoxycortisol in this patient was 6.9 µg/dl. Trace amounts of 11-deoxycortisol and DOC also were present in this patient s serum (0.9 µg/dl and 0.3 µg/ dl, respectively) (Table 1). ACTH stimulation test. Fig. 1 shows illustrative data for two patients who gave normal responses during the (1-24) ACTH stimulation test. Both patients had increases of serum cortisol levels, which peaked at 30 or 60 min after the injection of cosyntropin (0.5 mg/ patient). The serum concentrations of precursors in the glucocorticoid and mineral corticoid pathways showed little change (Fig. 1). In contrast, a patient with Addison disease, whose baseline ACTH level was 1,900 pg/ml (reference range, <23 pg/ml), showed
4 202 Annals of Clinical & Laboratory Science Fig. 1. Illustrative data for two patients with normal responses during (1-24) ACTH stimulation. Serum specimens were collected at baseline (before injection of ACTH) and at 30 and 60 min after ACTH injection. The dose of (1-24) ACTH (cosyntropin) was 0.5 mg/ patient. Fig. 3. Illustrative data for two patients who received an overnight metyrapone test, to show the steroid profiles before and after metyrapone administration. Metyrapone (3 g/ patient) was given at 11 pm and the second serum specimen was collected on the following morning. no change in the serum levels of cortisol or its precursors at 30 or 60 min after an ACTH injection (Fig. 2). In the patient with 21-hydroxylase deficiency, serum 17-hydroxyprogesterone concentration was 5.1 µg/dl at baseline and increased to 21.3 µg/dl at 30 min after the ACTH injection and 20.3 µg/dl at 60 min. The serum cortisol value remained unchanged (4.9 µg/dl) during this period. This patient s ACTH stimulation test was performed at the beginning of our study, before we could measure serum 21-deoxycortisol. Fig. 2. Steroid profiles of a patient with Addison disease before and after ACTH stimulation. There were essentially no changes in serum steroid concentrations at 30 and 60 min after the ACTH injection. The dose of (1-24) ACTH (cosyntropin) was 0.5 mg/ patient. Metyrapone test. In two patients, overnight metyrapone tests were performed by administering 3 g of metyrapone at 11 pm in order to block 11 β-hydroxylase activity. Serum obtained on the next morning had increased 11-deoxycortisol and decreased cortisol levels. In one patient, serum cortisol decreased from 5.1 to 1.3 µg/dl, 11-deoxycortisol increased from 0.6 to 12.5 µg/dl, and DOC increased from undetectable
5 Liquid chromatography-tandem mass spectrometry for adrenal diagnosis 203 to 3.9 µg/dl. In the other patient, serum cortisol decreased from 4.4 to 2.1 µg/dl, 11-deoxycortisol increased from 0.6 to 10.4 µg/dl, and DOC increased from undetectable to 1.8 µg/dl (Fig. 3). Discussion The authors predict that LC-MS/MS will soon become a mainstream technology for the diagnosis of adrenal cortical dysfunction, since multiple compounds of the steroid-biosynthetic pathway can be measured simultaneously in a single serum analysis. Availability of this technology will make differential diagnosis of enzyme deficiency in congenital adrenal hyperplasia (CAH) simpler than the customary procedure of ordering multiple immunoassays of individual metabolites according to algorithms or diagnostic flowcharts. Many endocrinologists, geneticists, and pediatricians are enthusiastic about this new method, because by a single test they can detect the phenotype of congenital adrenal enzyme deficiency. They can readily make a complete diagnosis without referring the patient for time-consuming confirmatory studies. Economics will be a factor that either delays or accelerates the introduction of LC-MS/MS for routine use in clinical laboratories. Although LC-MS/MS is generally viewed as an expensive analytical procedure, cost-comparison studies of assays for homocysteine and methylmalonic acid indicate that LC-MS/MS is actually less costly than HPLC or immunoassays, provided the test volume is large enough [9,10]. It is difficult to predict the costs of using LC-MS/ MS to measure six steroid metabolites in a single determination, as in this study The economical use of mass spectrometry will require deuterium-labeled compounds as internal standards for each specific analyte, and such standards were unavailable to us. The use of non-labeled standards to prepare a 12-point standard curve is too inefficient and time-consuming. We envision that, in response to growing clinical demand, the biomedical industry will supply the requisite deuterium standards, as well as specific analytical and interpretative computer software. The price of instruments for tandem mass spectrometry should decrease with its increasing use. Tandem mass spectrometry gives positive identification to each assayed metabolite and thus is superior to HPLC and immunoassays, which do not provide positive identification [8]. Immunoassays may cross-react with other compounds of the steroid pathway or with drugs of similar structure. A patient s serum may contain human anti-animal immunoglobulins (HAMA), which also cause interference. The HAMA interference gives either falsely high or low results and may lead to an incorrect diagnosis [3,7,8]. This pilot study demonstrated the usefulness of tandem mass spectrometry for the differential diagnosis of 11 β-hydroxylase and 21-hydroxylase deficiencies and for the diagnosis of Addison disease. In regard to 21-hydroxylase (Cyp 21 A2) deficiency, patients with this condition have thousand-fold increase of serum nonconjugated 17-hydroxyprogesterone and their serum contains 21-deoxycortisol, which is a marker of the 21-hydroxylase deficiency. In addition, the concentrations of serum cortisol and cortisone are reduced. These results are obtained simultaneously in a single determination by LC-MS/MS. Other assays cannot accomplish this. The corticotropin stimulation test with synthetic (1-24) ACTH revealed that the endocrine response was very rapid. When specimens were collected at 30 and 60 min after ACTH stimulation, cortisol was the only serum metabolite that increased in the normal subjects. Upstream intermediate compounds such as 11- deoxycortisol or 17-hydroxyprogesterone did not accumulate in serum at these intervals after ACTH injection; the precursors were converted to cortisol rapidly, within 30 min. After pharmaceutical blocking of 11 β-hydroxylase by metyrapone, patients had decreases of serum cortisol and cortisone and increases of serum 11-deoxycortisol and DOC. This situation is similar to CAH (11- hydroxylase deficiency). LC-MS/MS enhances the ability to diagnose such enzyme deficiency. ACTH stimulates cholesterol side-chain cleavage by P450scc enzyme (which, in the new genetic terminology, is CYP 11 A1), starting the pathway of steroid biosynthesis and increasing serum cortisol. Metyrapone blocks 11-hydroxylase (P450c, or CYP 11 B1) activity and inhibits the conversion of 11-doxycortisol to cortisol. The data obtained after ACTH stimulation and metyrapone blockade of enzyme pathways indicate that LC-MS/MS analysis can facilitate pathway profiling to find new drugs that target specific enzymes.
6 204 Annals of Clinical & Laboratory Science References 1. Parker KM. Peeking behind the curtain. Clin Lab News 1999;25(10):4. 2. Kao PC, Heser DW. Simultaneous determination of 25-hydroxy- and 1,25-dihydroxyvitamin D from a single sample by dual-cartridge extraction. Clin Chem 1984;30: Lin C-L, Wu T-J, Machacek DA, Jiang, N-S, Kao PC. Urinary free cortisol and cortisone determined by high performance liquid chromatography in the diagnosis of Cushing s syndrome. J Clin Endocrinol Metab 1997;82: Jiang NS, Machacek D. Measurement of catecholamines in blood and urine by liquid chromatography with amperometric detection: Prog HPLC 1987;2: Kricka LJ. Human anti-animal antibody interferences in immunological assays. Clin Chem 1999;45: Fitzgerald RL. Serum total testosterone: immunoassay compared with negative chemical ionization gas chromatography-mass spectrometry. Clin Chem 1996; 42: Müller U. Hagendorff G, Stibbe W, Schuff-Werner P. Prostate specific antigen (PSA) in serum samples of female patients: possible pitfalls in evaluating the analytical sensitivity of ultrasensitive PSA-assay. Anticancer Res 1997;17: Hicks JM. Excessive blood drawing for laboratory tests. N Engl J Med 1999;340: Magera M, Lacey J, Casetta B, Rinaldo P. Method for the determination of total homocysteine in plasma and urine by stable isotope dilution and electrospray tandem mass spectrometry. Clin Chem 1999;45: Magera M, Helgeson J, Matern D, Rinaldo P. Methylmalonic acid measured in plasma and urine by stable-isotope dilution and electrospray tandem mass spectrometry. Clin Chem 2000;46: Weinmann W, Wiedemann A, Eppinger B, Renz M, Svoboda M. Screening for drugs in serum by electrospray ionization/collision-induced dissociation and library searching. J Am Soc Mass Spectrom 1999;10: Findling JW, Pinkstaff SM, Shaker JL, Raff H, Nelson JC. Pseudohypercortisoluria: spurious elevation of urinary cortisol due to carbamazepine. Endocrinologist 1998;8:51-54.
All stocks and calibration levels were prepared in water: methanol (50:50) v/v to cover range of all steroid concentrations (refer Table 1).
 Application LCMS-8040 Simultaneous determination of 11 steroids and Vitamin D2/D3 in human serum using LC/MS/MS - Introduction Quantification of endogenous hormonal steroids and their precursors is essential
Application LCMS-8040 Simultaneous determination of 11 steroids and Vitamin D2/D3 in human serum using LC/MS/MS - Introduction Quantification of endogenous hormonal steroids and their precursors is essential
Determination of 6-Chloropicolinic Acid (6-CPA) in Crops by Liquid Chromatography with Tandem Mass Spectrometry Detection. EPL-BAS Method No.
 Page 1 of 10 Determination of 6-Chloropicolinic Acid (6-CPA) in Crops by Liquid Chromatography with Tandem Mass Spectrometry Detection EPL-BAS Method No. 205G881B Method Summary: Residues of 6-CPA are
Page 1 of 10 Determination of 6-Chloropicolinic Acid (6-CPA) in Crops by Liquid Chromatography with Tandem Mass Spectrometry Detection EPL-BAS Method No. 205G881B Method Summary: Residues of 6-CPA are
Determination of β2-agonists in Pork Using Agilent SampliQ SCX Solid-Phase Extraction Cartridges and Liquid Chromatography-Tandem Mass Spectrometry
 Determination of β2-agonists in Pork Using Agilent SampliQ SCX Solid-Phase Extraction Cartridges and Liquid Chromatography-Tandem Mass Spectrometry Application Note Food Safety Authors Chenhao Zhai Agilent
Determination of β2-agonists in Pork Using Agilent SampliQ SCX Solid-Phase Extraction Cartridges and Liquid Chromatography-Tandem Mass Spectrometry Application Note Food Safety Authors Chenhao Zhai Agilent
Determination of Bath Salts (Pyrovalerone Analogs) in Biological Samples
 Determination of Bath Salts (Pyrovalerone Analogs) in Biological Samples Application Note Forensic Toxicology Authors Joe Crifasi Saint Louis University Forensic Toxicology Laboratory Saint Louis, Mo.
Determination of Bath Salts (Pyrovalerone Analogs) in Biological Samples Application Note Forensic Toxicology Authors Joe Crifasi Saint Louis University Forensic Toxicology Laboratory Saint Louis, Mo.
Ultra Performance Liquid Chromatography Coupled to Orthogonal Quadrupole TOF MS(MS) for Metabolite Identification
 22 SEPARATION SCIENCE REDEFINED MAY 2005 Ultra Performance Liquid Chromatography Coupled to Orthogonal Quadrupole TOF MS(MS) for Metabolite Identification In the drug discovery process the detection and
22 SEPARATION SCIENCE REDEFINED MAY 2005 Ultra Performance Liquid Chromatography Coupled to Orthogonal Quadrupole TOF MS(MS) for Metabolite Identification In the drug discovery process the detection and
The analysis of Glucocorticoid Steroids in Plasma, Urine and Saliva by UPLC/MS/MS
 The analysis of Glucocorticoid Steroids in Plasma, Urine and Saliva by UPLC/MS/MS Brett McWhinney, Supervising Scientist, HPLC Section, Pathology Central, Pathology Queensland Overview 1. Overview of Pathology
The analysis of Glucocorticoid Steroids in Plasma, Urine and Saliva by UPLC/MS/MS Brett McWhinney, Supervising Scientist, HPLC Section, Pathology Central, Pathology Queensland Overview 1. Overview of Pathology
Amphetamines, Phentermine, and Designer Stimulant Quantitation Using an Agilent 6430 LC/MS/MS
 Amphetamines, Phentermine, and Designer Stimulant Quantitation Using an Agilent 643 LC/MS/MS Application Note Forensics Authors Jason Hudson, Ph.D., James Hutchings, Ph.D., and Rebecca Wagner, Ph.D. Virginia
Amphetamines, Phentermine, and Designer Stimulant Quantitation Using an Agilent 643 LC/MS/MS Application Note Forensics Authors Jason Hudson, Ph.D., James Hutchings, Ph.D., and Rebecca Wagner, Ph.D. Virginia
Diagnostic Testing in Cushing's Syndrome: Reassessment of 17-hydroxycorticosteroid and 17-ketosteroid Measurements
 CRTCAL REVEW [ K e i t h D u n c a n, M. D. March, 1985 Diagnostic Testing in Cushing's Syndrome: Reassessment of 17-hydroxycorticosteroid and 17-ketosteroid Measurements ntroduction The measurement of
CRTCAL REVEW [ K e i t h D u n c a n, M. D. March, 1985 Diagnostic Testing in Cushing's Syndrome: Reassessment of 17-hydroxycorticosteroid and 17-ketosteroid Measurements ntroduction The measurement of
Robust extraction, separation, and quantitation of structural isomer steroids from human plasma by SPE-UHPLC-MS/MS
 TECHNICAL NOTE 21882 Robust extraction, separation, and quantitation of structural isomer steroids human plasma by SPE-UHPLC-MS/MS Authors Jon Bardsley 1, Kean Woodmansey 1, and Stacy Tremintin 2 1 Thermo
TECHNICAL NOTE 21882 Robust extraction, separation, and quantitation of structural isomer steroids human plasma by SPE-UHPLC-MS/MS Authors Jon Bardsley 1, Kean Woodmansey 1, and Stacy Tremintin 2 1 Thermo
Screening of Antihistamine Agents (Diphenhydramine) with Blood and Urine Samples by REMEDi-HS System
 Screening of Antihistamine Agents (Diphenhydramine) with Blood and Urine Samples by REMEDi-HS System Ohtsuji M, Ohshima T, Takayasu T, Nishigami J, Kondo T, Lin Z, Minamino T Department of Legal Medicine,
Screening of Antihistamine Agents (Diphenhydramine) with Blood and Urine Samples by REMEDi-HS System Ohtsuji M, Ohshima T, Takayasu T, Nishigami J, Kondo T, Lin Z, Minamino T Department of Legal Medicine,
Validation of a Reliable LC-MS/MS Method for Analysis of Simultaneously in Clinical Laboratory
 Validation of a Reliable LC-MS/MS Method for Analysis of Simultaneously in Clinical Laboratory Five Steroids Atecla Nunciata Lopes Alves, Berenice Bilharinho Mendonca, Helena Panteliou Lima Valassi. Laboratório
Validation of a Reliable LC-MS/MS Method for Analysis of Simultaneously in Clinical Laboratory Five Steroids Atecla Nunciata Lopes Alves, Berenice Bilharinho Mendonca, Helena Panteliou Lima Valassi. Laboratório
O O H. Robert S. Plumb and Paul D. Rainville Waters Corporation, Milford, MA, U.S. INTRODUCTION EXPERIMENTAL. LC /MS conditions
 Simplifying Qual/Quan Analysis in Discovery DMPK using UPLC and Xevo TQ MS Robert S. Plumb and Paul D. Rainville Waters Corporation, Milford, MA, U.S. INTRODUCTION The determination of the drug metabolism
Simplifying Qual/Quan Analysis in Discovery DMPK using UPLC and Xevo TQ MS Robert S. Plumb and Paul D. Rainville Waters Corporation, Milford, MA, U.S. INTRODUCTION The determination of the drug metabolism
Application Note. Author. Abstract. Introduction. Food Safety
 Determination of β2-agonists in Pork with SPE eanup and LC-MS/MS Detection Using Agilent BondElut PCX Solid-Phase Extraction Cartridges, Agilent Poroshell 120 column and Liquid Chromatography-Tandem Mass
Determination of β2-agonists in Pork with SPE eanup and LC-MS/MS Detection Using Agilent BondElut PCX Solid-Phase Extraction Cartridges, Agilent Poroshell 120 column and Liquid Chromatography-Tandem Mass
Core E Analysis of Neutral Lipids from Human Plasma June 4, 2010 Thomas J. Leiker and Robert M. Barkley
 Core E Analysis of Neutral Lipids from Human Plasma June 4, 2010 Thomas J. Leiker and Robert M. Barkley This protocol describes the extraction and direct measurement of cholesterol esters (CEs) and triacylglycerols
Core E Analysis of Neutral Lipids from Human Plasma June 4, 2010 Thomas J. Leiker and Robert M. Barkley This protocol describes the extraction and direct measurement of cholesterol esters (CEs) and triacylglycerols
Quantitative Analysis of Drugs of Abuse in Urine using UHPLC Coupled to Accurate Mass AxION 2 TOF Mass Spectrometer
 application Note Liquid Chromatography/ Mass Spectrometry Authors Sharanya Reddy Blas Cerda PerkinElmer, Inc. Shelton, CT USA Quantitative Analysis of Drugs of Abuse in Urine using UHPLC Coupled to Accurate
application Note Liquid Chromatography/ Mass Spectrometry Authors Sharanya Reddy Blas Cerda PerkinElmer, Inc. Shelton, CT USA Quantitative Analysis of Drugs of Abuse in Urine using UHPLC Coupled to Accurate
SUPPLEMENTARY APPENDIX. COU-AA-301 enrolled men with pathologically confirmed mcrpc who had received previous
 SUPPLEMENTARY APPENDIX Methods Subjects COUAA30 enrolled men with pathologically confirmed mcrpc who had received previous treatment with docetaxel chemotherapy and had documented PSA progression according
SUPPLEMENTARY APPENDIX Methods Subjects COUAA30 enrolled men with pathologically confirmed mcrpc who had received previous treatment with docetaxel chemotherapy and had documented PSA progression according
Measurement of Renin Activity using Tandem Mass Spectrometry Ravinder J Singh, PhD, DABCC Mayo Clinic, Rochester, MN
 Measurement of Renin Activity using Tandem Mass Spectrometry Ravinder J Singh, PhD, DABCC Mayo Clinic, Rochester, MN Renin Angiotension System Weber, et al. NEJM 2001; 345:1690. Renin Angiotension System
Measurement of Renin Activity using Tandem Mass Spectrometry Ravinder J Singh, PhD, DABCC Mayo Clinic, Rochester, MN Renin Angiotension System Weber, et al. NEJM 2001; 345:1690. Renin Angiotension System
Analysis of Testosterone, Androstenedione, and Dehydroepiandrosterone Sulfate in Serum for Clinical Research
 Analysis of Testosterone, Androstenedione, and Dehydroepiandrosterone Sulfate in Serum for Clinical Research Dominic Foley, Michelle Wills, and Lisa Calton Waters Corporation, Wilmslow, UK APPLICATION
Analysis of Testosterone, Androstenedione, and Dehydroepiandrosterone Sulfate in Serum for Clinical Research Dominic Foley, Michelle Wills, and Lisa Calton Waters Corporation, Wilmslow, UK APPLICATION
LC-MS/MS Method for the Determination of Tenofovir from Plasma
 LC-MS/MS Method for the Determination of Tenofovir from Plasma Kimberly Phipps, Thermo Fisher Scientific, Runcorn, Cheshire, UK Application Note 687 Key Words SPE, SOLA CX, Hypersil GOLD, tenofovir Abstract
LC-MS/MS Method for the Determination of Tenofovir from Plasma Kimberly Phipps, Thermo Fisher Scientific, Runcorn, Cheshire, UK Application Note 687 Key Words SPE, SOLA CX, Hypersil GOLD, tenofovir Abstract
Abstract. Introduction
 A Fast Dilute-And-Shoot Method for Simultaneous 5-Hydroxyindoleacetic Acid (5-HIAA), Vanillylmandelic Acid (VMA), and Homovanillic Acid (HVA) LC-MS/MS Analysis in Human Urine By Shun-Hsin Liang and Sue
A Fast Dilute-And-Shoot Method for Simultaneous 5-Hydroxyindoleacetic Acid (5-HIAA), Vanillylmandelic Acid (VMA), and Homovanillic Acid (HVA) LC-MS/MS Analysis in Human Urine By Shun-Hsin Liang and Sue
Neosolaniol. [Methods listed in the Feed Analysis Standards]
![Neosolaniol. [Methods listed in the Feed Analysis Standards] Neosolaniol. [Methods listed in the Feed Analysis Standards]](/thumbs/86/94003482.jpg) Neosolaniol [Methods listed in the Feed Analysis Standards] 1 Simultaneous analysis of mycotoxins by liquid chromatography/ tandem mass spectrometry [Feed Analysis Standards, Chapter 5, Section 1 9.1 ]
Neosolaniol [Methods listed in the Feed Analysis Standards] 1 Simultaneous analysis of mycotoxins by liquid chromatography/ tandem mass spectrometry [Feed Analysis Standards, Chapter 5, Section 1 9.1 ]
Dienes Derivatization MaxSpec Kit
 Dienes Derivatization MaxSpec Kit Item No. 601510 www.caymanchem.com Customer Service 800.364.9897 Technical Support 888.526.5351 1180 E. Ellsworth Rd Ann Arbor, MI USA TABLE OF CONTENTS GENERAL INFORMATION
Dienes Derivatization MaxSpec Kit Item No. 601510 www.caymanchem.com Customer Service 800.364.9897 Technical Support 888.526.5351 1180 E. Ellsworth Rd Ann Arbor, MI USA TABLE OF CONTENTS GENERAL INFORMATION
Application Note LCMS-108 Quantitation of benzodiazepines and Z-drugs in serum with the EVOQ TM LC triple quadrupole mass spectrometer
 Application Note LCMS-108 Quantitation of benzodiazepines and Z-drugs in serum with the EVOQ TM LC triple quadrupole mass spectrometer Abstract This study demonstrates a sensitive, rapid and reliable research
Application Note LCMS-108 Quantitation of benzodiazepines and Z-drugs in serum with the EVOQ TM LC triple quadrupole mass spectrometer Abstract This study demonstrates a sensitive, rapid and reliable research
Detection of Low Level of Chloramphenicol in Milk and Honey with MIP SPE and LC-MS-MS
 Detection of Low Level of Chloramphenicol in Milk and Honey with MIP SPE and LC-MS-MS Olga Shimelis, An Trinh, and Michael Ye Supelco, Div. of Sigma-Aldrich, Bellefonte, PA T407125 Introduction Molecularly
Detection of Low Level of Chloramphenicol in Milk and Honey with MIP SPE and LC-MS-MS Olga Shimelis, An Trinh, and Michael Ye Supelco, Div. of Sigma-Aldrich, Bellefonte, PA T407125 Introduction Molecularly
The Third Department of Internal Medicine, University of Tokyo Faculty of Medicine, Hongo, Tokyo 113
 Endocrinol. Japon. 1974, 21 (2), 115 ` 119 A Radioimmunoassay for Serum Dehydroepiandrosterone HISAHIKO SEKIHARA, TOHRU YAMAJI, NAKAAKI OHSAWA AND HIROSHI IBAYASHI * The Third Department of Internal Medicine,
Endocrinol. Japon. 1974, 21 (2), 115 ` 119 A Radioimmunoassay for Serum Dehydroepiandrosterone HISAHIKO SEKIHARA, TOHRU YAMAJI, NAKAAKI OHSAWA AND HIROSHI IBAYASHI * The Third Department of Internal Medicine,
Integration of steroids analysis in serum using LC-MS/MS with full-automated sample preparation
 PO-CON69E Integration of steroids analysis in serum using LC-MS/MS with full-automated sample preparation MSACL 6 EU Stéphane Moreau, Daisuke Kawakami, Toshikazu Minohata Shimadzu Europe GmbH, Duisburg,
PO-CON69E Integration of steroids analysis in serum using LC-MS/MS with full-automated sample preparation MSACL 6 EU Stéphane Moreau, Daisuke Kawakami, Toshikazu Minohata Shimadzu Europe GmbH, Duisburg,
Rapid and Accurate LC-MS/MS Analysis of Nicotine and Related Compounds in Urine Using Raptor Biphenyl LC Columns and MS-Friendly Mobile Phases
 Clinical, Forensic & Toxicology Applications Rapid and Accurate LC-MS/MS Analysis of Nicotine and Related Compounds in Urine Using Raptor Biphenyl LC Columns and MS-Friendly Mobile Phases By Shun-Hsin
Clinical, Forensic & Toxicology Applications Rapid and Accurate LC-MS/MS Analysis of Nicotine and Related Compounds in Urine Using Raptor Biphenyl LC Columns and MS-Friendly Mobile Phases By Shun-Hsin
LC-Based Lipidomics Analysis on QTRAP Instruments
 LC-Based Lipidomics Analysis on QTRAP Instruments Junhua Wang and Paul RS Baker SCIEX LC-Based Lipidomics Analysis Topics Covered Lipid extraction techniques Hydrophilic Interaction Chromatography (HILIC)
LC-Based Lipidomics Analysis on QTRAP Instruments Junhua Wang and Paul RS Baker SCIEX LC-Based Lipidomics Analysis Topics Covered Lipid extraction techniques Hydrophilic Interaction Chromatography (HILIC)
ULTIMATE BEAUTY OF BIOCHEMISTRY. Dr. Veena Bhaskar S Gowda Dept of Biochemistry 30 th Nov 2017
 ULTIMATE BEAUTY OF BIOCHEMISTRY Dr. Veena Bhaskar S Gowda Dept of Biochemistry 30 th Nov 2017 SUSPECTED CASE OF CUSHING S SYNDROME Clinical features Moon face Obesity Hypertension Hunch back Abdominal
ULTIMATE BEAUTY OF BIOCHEMISTRY Dr. Veena Bhaskar S Gowda Dept of Biochemistry 30 th Nov 2017 SUSPECTED CASE OF CUSHING S SYNDROME Clinical features Moon face Obesity Hypertension Hunch back Abdominal
In-stream attenuation of neuro-active pharmaceuticals and their
 In-stream attenuation of neuro-active pharmaceuticals and their metabolites Supporting Information Analytical Methods and Figures (10 pages) Jeffrey H. Writer, Ronald C. Antweiler, Imma Ferrer, Joseph
In-stream attenuation of neuro-active pharmaceuticals and their metabolites Supporting Information Analytical Methods and Figures (10 pages) Jeffrey H. Writer, Ronald C. Antweiler, Imma Ferrer, Joseph
Method Development for the Analysis of Endogenous Steroids Using Convergence Chromatography with Mass Spectrometric Detection
 Method Development for the Analysis of Endogenous Steroids Using Convergence Chromatography with Mass Spectrometric Detection Christopher J. Hudalla, Stuart Chadwick, Fiona Liddicoat, Andrew Peck, and
Method Development for the Analysis of Endogenous Steroids Using Convergence Chromatography with Mass Spectrometric Detection Christopher J. Hudalla, Stuart Chadwick, Fiona Liddicoat, Andrew Peck, and
SCIEX Vitamin D 200M Assay for the Topaz System
 The First FDA-Cleared LC-MS/MS Assay for Vitamin D SCIEX Vitamin D 200M Assay for the Topaz System The first FDA-cleared LC-MS/MS assay for Vitamin D Vitamin D is an important building block for human
The First FDA-Cleared LC-MS/MS Assay for Vitamin D SCIEX Vitamin D 200M Assay for the Topaz System The first FDA-cleared LC-MS/MS assay for Vitamin D Vitamin D is an important building block for human
UPLC/MS Monitoring of Water-Soluble Vitamin Bs in Cell Culture Media in Minutes
 UPLC/MS Monitoring of Water-Soluble Vitamin Bs in Cell Culture Media in Minutes Catalin E. Doneanu, Weibin Chen, and Jeffrey R. Mazzeo Waters Corporation, Milford, MA, U.S. A P P L I C AT ION B E N E F
UPLC/MS Monitoring of Water-Soluble Vitamin Bs in Cell Culture Media in Minutes Catalin E. Doneanu, Weibin Chen, and Jeffrey R. Mazzeo Waters Corporation, Milford, MA, U.S. A P P L I C AT ION B E N E F
Fast and simultaneous analysis of ethanol metabolites and barbiturates using the QTRAP 4500 LC-MS/MS system
 Fast and simultaneous analysis of ethanol metabolites and barbiturates using the QTRAP 4500 LC-MS/MS system Xiang He 1, Adrian Taylor 2 and Alexandre Wang 1 1 SCIEX, Redwood City, USA. 2 SCIEX, Concord,
Fast and simultaneous analysis of ethanol metabolites and barbiturates using the QTRAP 4500 LC-MS/MS system Xiang He 1, Adrian Taylor 2 and Alexandre Wang 1 1 SCIEX, Redwood City, USA. 2 SCIEX, Concord,
Sample Concentration and Analysis of Human Hormones in Drinking Water
 Sample Concentration and Analysis of Human Hormones in Drinking Water Carl Fisher Applications Chemist Ion Chromatography/Sample Preparation Thermo Fisher Scientific March 1, 215 1 The world leader in
Sample Concentration and Analysis of Human Hormones in Drinking Water Carl Fisher Applications Chemist Ion Chromatography/Sample Preparation Thermo Fisher Scientific March 1, 215 1 The world leader in
Piero Rinaldo, Si Houn Hahn, Dietrich Matern. Biochemical Genetics Laboratory Mayo Clinic College of Medicine Rochester, MN
 Improved Specificity of Newborn Screening for Congenital Adrenal Hyperplasia (CAH) by Second Tier Steroid Profiling using Tandem Mass Spectrometry (MS/MS) Piero Rinaldo, Si Houn Hahn, Dietrich Matern Biochemical
Improved Specificity of Newborn Screening for Congenital Adrenal Hyperplasia (CAH) by Second Tier Steroid Profiling using Tandem Mass Spectrometry (MS/MS) Piero Rinaldo, Si Houn Hahn, Dietrich Matern Biochemical
Analytical Method for 2, 4, 5-T (Targeted to Agricultural, Animal and Fishery Products)
 Analytical Method for 2, 4, 5-T (Targeted to Agricultural, Animal and Fishery Products) The target compound to be determined is 2, 4, 5-T. 1. Instrument Liquid Chromatograph-tandem mass spectrometer (LC-MS/MS)
Analytical Method for 2, 4, 5-T (Targeted to Agricultural, Animal and Fishery Products) The target compound to be determined is 2, 4, 5-T. 1. Instrument Liquid Chromatograph-tandem mass spectrometer (LC-MS/MS)
Rapid Screening and Quantitation of Postharvest Fungicides on Citrus Fruits Using AxION DSA/TOF and Flexar SQ MS
 APPLICATION NOTE Mass Spectrometry Author: Avinash Dalmia PerkinElmer, Inc. Shelton, CT USA Rapid Screening and Quantitation of Postharvest Fungicides on Citrus Fruits Using AxION DSA/TOF and Flexar SQ
APPLICATION NOTE Mass Spectrometry Author: Avinash Dalmia PerkinElmer, Inc. Shelton, CT USA Rapid Screening and Quantitation of Postharvest Fungicides on Citrus Fruits Using AxION DSA/TOF and Flexar SQ
Detection of Cannabinoids in Oral Fluid with the Agilent 7010 GC-MS/MS System
 Application Note Forensics, Workplace Drug Testing Detection of Cannabinoids in Oral Fluid with the Agilent 7010 GC-MS/MS System Authors Fred Feyerherm and Anthony Macherone Agilent Technologies, Inc.
Application Note Forensics, Workplace Drug Testing Detection of Cannabinoids in Oral Fluid with the Agilent 7010 GC-MS/MS System Authors Fred Feyerherm and Anthony Macherone Agilent Technologies, Inc.
The use of mass spectrometry in lipidomics. Outlines
 The use of mass spectrometry in lipidomics Jeevan Prasain jprasain@uab.edu 6-2612 utlines Brief introduction to lipidomics Analytical methodology: MS/MS structure elucidation of phospholipids Phospholipid
The use of mass spectrometry in lipidomics Jeevan Prasain jprasain@uab.edu 6-2612 utlines Brief introduction to lipidomics Analytical methodology: MS/MS structure elucidation of phospholipids Phospholipid
Characterization of an Unknown Compound Using the LTQ Orbitrap
 Characterization of an Unknown Compound Using the LTQ rbitrap Donald Daley, Russell Scammell, Argenta Discovery Limited, 8/9 Spire Green Centre, Flex Meadow, Harlow, Essex, CM19 5TR, UK bjectives unknown
Characterization of an Unknown Compound Using the LTQ rbitrap Donald Daley, Russell Scammell, Argenta Discovery Limited, 8/9 Spire Green Centre, Flex Meadow, Harlow, Essex, CM19 5TR, UK bjectives unknown
Analysis of Acrylamide in French Fries using Agilent Bond Elut QuEChERS AOAC kit and LC/MS/MS
 Analysis of Acrylamide in French Fries using Agilent Bond Elut QuEChERS AOAC kit and LC/MS/MS Food Application Author Fadwa Al-Taher Institute for Food Safety and Health Illinois Institute of Technology
Analysis of Acrylamide in French Fries using Agilent Bond Elut QuEChERS AOAC kit and LC/MS/MS Food Application Author Fadwa Al-Taher Institute for Food Safety and Health Illinois Institute of Technology
Measuring Lipid Composition LC-MS/MS
 Project: Measuring Lipid Composition LC-MS/MS Verification of expected lipid composition in nanomedical controlled release systems by liquid chromatography tandem mass spectrometry AUTHORED BY: DATE: Sven
Project: Measuring Lipid Composition LC-MS/MS Verification of expected lipid composition in nanomedical controlled release systems by liquid chromatography tandem mass spectrometry AUTHORED BY: DATE: Sven
A RAPID AND SENSITIVE ANALYSIS METHOD OF SUDAN RED I, II, III & IV IN TOMATO SAUCE USING ULTRA PERFORMANCE LC MS/MS
 A RAPID AD SESITIVE AALYSIS METD OF SUDA RED I, II, III & IV I TOMATO SAUCE USIG ULTRA PERFORMACE LC MS/MS Choon Keow G, aomi TAAKA, Michelle KIM, Swee Lee YAP Waters Asia, Regional Technology Center,
A RAPID AD SESITIVE AALYSIS METD OF SUDA RED I, II, III & IV I TOMATO SAUCE USIG ULTRA PERFORMACE LC MS/MS Choon Keow G, aomi TAAKA, Michelle KIM, Swee Lee YAP Waters Asia, Regional Technology Center,
Cannabinoid Quantitation Using an Agilent 6430 LC/MS/MS
 Cannabinoid Quantitation Using an Agilent 643 LC/MS/MS Application Note Forensics Authors Jason Hudson, Ph.D., James Hutchings, Ph.D., and Rebecca Wagner, Ph.D. Virginia Department of Forensic Science
Cannabinoid Quantitation Using an Agilent 643 LC/MS/MS Application Note Forensics Authors Jason Hudson, Ph.D., James Hutchings, Ph.D., and Rebecca Wagner, Ph.D. Virginia Department of Forensic Science
Extraction of a Comprehensive Steroid Panel from Human Serum Using ISOLUTE. SLE+ Prior to LC/MS-MS Analysis
 Application Note AN890 Extraction of a Comprehensive Steroid Panel from Human Serum Using ISOLUTE SLE+ Page Extraction of a Comprehensive Steroid Panel from Human Serum Using ISOLUTE SLE+ Prior to LC/MS-MS
Application Note AN890 Extraction of a Comprehensive Steroid Panel from Human Serum Using ISOLUTE SLE+ Page Extraction of a Comprehensive Steroid Panel from Human Serum Using ISOLUTE SLE+ Prior to LC/MS-MS
Analytes. Sample Preparation Procedure. Introduction. Format: Sample Pre-treatment. Sample Loading. Analyte Extraction and Derivatization.
 Application Note AN857 Ultra-Sensitive Method for the Determination of 1,25 di-oh Vitamin D2 and 1,25 di-oh Vitamin D3 Page 1 Ultra-Sensitive Method for the Determination of 1,25 di-oh Vitamin D2 and 1,25
Application Note AN857 Ultra-Sensitive Method for the Determination of 1,25 di-oh Vitamin D2 and 1,25 di-oh Vitamin D3 Page 1 Ultra-Sensitive Method for the Determination of 1,25 di-oh Vitamin D2 and 1,25
Shuguang Li, Jason Anspach, Sky Countryman, and Erica Pike Phenomenex, Inc., 411 Madrid Ave., Torrance, CA USA PO _W
 Simple, Fast and Accurate Quantitation of Human Plasma Vitamins and Their Metabolites by Protein Precipitation Combined with Columns Using HPLC-UV, HPLC-FLD or LC/MS/MS Shuguang Li, Jason Anspach, Sky
Simple, Fast and Accurate Quantitation of Human Plasma Vitamins and Their Metabolites by Protein Precipitation Combined with Columns Using HPLC-UV, HPLC-FLD or LC/MS/MS Shuguang Li, Jason Anspach, Sky
High-Throughput, Cost-Efficient LC-MS/MS Forensic Method for Measuring Buprenorphine and Norbuprenorphine in Urine
 High-Throughput, Cost-Efficient LC-MS/MS Forensic Method for Measuring and in Urine Xiaolei Xie, Joe DiBussolo, Marta Kozak; Thermo Fisher Scientific, San Jose, CA Application Note 627 Key Words, norbuprenorphine,
High-Throughput, Cost-Efficient LC-MS/MS Forensic Method for Measuring and in Urine Xiaolei Xie, Joe DiBussolo, Marta Kozak; Thermo Fisher Scientific, San Jose, CA Application Note 627 Key Words, norbuprenorphine,
The Investigation of Factors Contributing to Immunosuppressant Drugs Response Variability in LC-MS/MS Analysis
 The Investigation of Factors Contributing to Immunosuppressant Drugs Variability in LC-MS/MS Analysis Joseph Herman, Dayana Argoti, and Sarah Fair Thermo Fisher Scientific, Franklin, MA, USA Overview Purpose:
The Investigation of Factors Contributing to Immunosuppressant Drugs Variability in LC-MS/MS Analysis Joseph Herman, Dayana Argoti, and Sarah Fair Thermo Fisher Scientific, Franklin, MA, USA Overview Purpose:
Quantitative Analysis of Vit D Metabolites in Human Plasma using Exactive System
 Quantitative Analysis of Vit D Metabolites in Human Plasma using Exactive System Marta Kozak Clinical Research Applications Group Thermo Fisher Scientific San Jose CA Clinical Research use only, Not for
Quantitative Analysis of Vit D Metabolites in Human Plasma using Exactive System Marta Kozak Clinical Research Applications Group Thermo Fisher Scientific San Jose CA Clinical Research use only, Not for
Simultaneous Quantitative Analysis of Total Catecholamines and Metanephrines in Urine Using CLEAN UP CCX2 and LC-MS/MS
 Simultaneous Quantitative Analysis of Total Catecholamines and Metanephrines in Urine Using CLEAN UP CCX2 and LC-MS/MS UCT Part Numbers CUCCX256 Clean Up CCX2 (C8 + Carboxylic Acid) 500mg / 6mL SPE Cartridge
Simultaneous Quantitative Analysis of Total Catecholamines and Metanephrines in Urine Using CLEAN UP CCX2 and LC-MS/MS UCT Part Numbers CUCCX256 Clean Up CCX2 (C8 + Carboxylic Acid) 500mg / 6mL SPE Cartridge
SPE-LC-MS/MS Method for the Determination of Nicotine, Cotinine, and Trans-3-hydroxycotinine in Urine
 SPE-LC-MS/MS Method for the Determination of Nicotine, Cotinine, and Trans-3-hydroxycotinine in Urine J. Jones, Thermo Fisher Scientific, Runcorn, Cheshire, UK Application Note 709 Key Words SPE, SOLA
SPE-LC-MS/MS Method for the Determination of Nicotine, Cotinine, and Trans-3-hydroxycotinine in Urine J. Jones, Thermo Fisher Scientific, Runcorn, Cheshire, UK Application Note 709 Key Words SPE, SOLA
LC/MS/MS Separation of Cholesterol and Related Sterols in Plasma on an Agilent InfinityLab Poroshell 120 EC C18 Column
 Application Note Clinical Research LC/MS/MS Separation of Cholesterol and Related Sterols in Plasma on an Agilent InfinityLab Poroshell EC C8 Column Authors Rongjie Fu and Zhiming Zhang Agilent Technologies
Application Note Clinical Research LC/MS/MS Separation of Cholesterol and Related Sterols in Plasma on an Agilent InfinityLab Poroshell EC C8 Column Authors Rongjie Fu and Zhiming Zhang Agilent Technologies
Determination of Gamma-Hydroxy-Butyrate (GHB) in Biological Samples
 Determination of Gamma-Hydroxy-Butyrate (GHB) in Biological Samples Application Note Forensic Toxicology Authors Joe Crifasi Saint Louis University Forensic Toxicology Laboratory Saint Louis, MO, USA Ron
Determination of Gamma-Hydroxy-Butyrate (GHB) in Biological Samples Application Note Forensic Toxicology Authors Joe Crifasi Saint Louis University Forensic Toxicology Laboratory Saint Louis, MO, USA Ron
Comparison of Full Scan MS2 and MS3 Linear Ion Trap Approaches for Quantitation of Vitamin D
 Comparison of Full Scan MS2 and MS3 Linear Ion Trap Approaches for Quantitation of Vitamin D Julie A. Horner 1, Marta Kozak 1, Subodh Nimkar 1, and August A. Specht 1 1 Thermo Fisher Scientific, San Jose,
Comparison of Full Scan MS2 and MS3 Linear Ion Trap Approaches for Quantitation of Vitamin D Julie A. Horner 1, Marta Kozak 1, Subodh Nimkar 1, and August A. Specht 1 1 Thermo Fisher Scientific, San Jose,
The endocrine system is made up of a complex group of glands that secrete hormones.
 1 10. Endocrinology I MEDCHEM 535 Diagnostic Medicinal Chemistry Endocrinology The endocrine system is made up of a complex group of glands that secrete hormones. These hormones control reproduction, metabolism,
1 10. Endocrinology I MEDCHEM 535 Diagnostic Medicinal Chemistry Endocrinology The endocrine system is made up of a complex group of glands that secrete hormones. These hormones control reproduction, metabolism,
Identification of Steroids in Water by Ion Trap LC/MS/MS Application
 Identification of Steroids in Water by Ion Trap LC/MS/MS Application Environmental Author Paul Zavitsanos and Christine Miller Agilent Technologies, Inc. Centerville Road Wilmington, DE 9- USA Abstract
Identification of Steroids in Water by Ion Trap LC/MS/MS Application Environmental Author Paul Zavitsanos and Christine Miller Agilent Technologies, Inc. Centerville Road Wilmington, DE 9- USA Abstract
C h a p t e r 3 8 Cushing s Syndrome : Current Concepts in Diagnosis and Management
 C h a p t e r 3 8 Cushing s Syndrome : Current Concepts in Diagnosis and Management Padma S Menon Professor of Endocrinology, Seth G S Medical College & KEM Hospital, Mumbai A clinical syndrome resulting
C h a p t e r 3 8 Cushing s Syndrome : Current Concepts in Diagnosis and Management Padma S Menon Professor of Endocrinology, Seth G S Medical College & KEM Hospital, Mumbai A clinical syndrome resulting
Detection, Confirmation, and Quantification of Chloramphenicol in Honey, Shrimp and Chicken Using the Agilent 6410 LC/MS Triple Quadrupole
 Detection, Confirmation, and Quantification of Chloramphenicol in Honey, Shrimp and Chicken Using the Agilent LC/MS Triple Quadrupole Application Food Safety Authors Yanyan Fang Agilent Technologies (Shanghai),
Detection, Confirmation, and Quantification of Chloramphenicol in Honey, Shrimp and Chicken Using the Agilent LC/MS Triple Quadrupole Application Food Safety Authors Yanyan Fang Agilent Technologies (Shanghai),
LC/MS/MS Analysis of Metabolites of Synthetic Cannabinoids JWH-018 and JWH-073 in Urine
 Abstract A liquid chromatographic method was developed to resolve a comprehensive set of metabolites of JWH-018 and JWH-073. In addition to the chromatographic analysis method, an extraction method was
Abstract A liquid chromatographic method was developed to resolve a comprehensive set of metabolites of JWH-018 and JWH-073. In addition to the chromatographic analysis method, an extraction method was
Monitoring of plasmatic concentrations of AEDs is important to improve patient s theraphy.
 ANTIEPILEPTICS in plasma by LC-MS Deuterated internal Std - Code LC05015 (Levetiracetam, Lamotrigine, Primidone, Oxcarbazepine, Carbamazepine Epoxide, 10,11-Dihydro- 10-Hydroxy-Carbamazepine, Carbamazepine,
ANTIEPILEPTICS in plasma by LC-MS Deuterated internal Std - Code LC05015 (Levetiracetam, Lamotrigine, Primidone, Oxcarbazepine, Carbamazepine Epoxide, 10,11-Dihydro- 10-Hydroxy-Carbamazepine, Carbamazepine,
Determination of Aflatoxins in Food by LC/MS/MS. Application. Authors. Abstract. Experimental. Introduction. Food Safety
 Determination of Aflatoxins in Food by LC/MS/MS Application Food Safety Authors Masahiko Takino Agilent Technologies 9-1 Takakura-Cho Hachiouji-Shi, Tokyo Japan Toshitsugu Tanaka Kobe Institute of Health
Determination of Aflatoxins in Food by LC/MS/MS Application Food Safety Authors Masahiko Takino Agilent Technologies 9-1 Takakura-Cho Hachiouji-Shi, Tokyo Japan Toshitsugu Tanaka Kobe Institute of Health
A UPLC/MS Approach for the Diagnosis of Inborn Errors of Metabolism
 Elizabeth Want, 1 Stephen McDonald 2 1 Imperial College, London, UK 2 Waters Corporation, Milford, MA, USA A P P L I C AT ION B E N E F I T S UPLC /MS E was used to profile diagnostic metabolites in a
Elizabeth Want, 1 Stephen McDonald 2 1 Imperial College, London, UK 2 Waters Corporation, Milford, MA, USA A P P L I C AT ION B E N E F I T S UPLC /MS E was used to profile diagnostic metabolites in a
New Solvent Grade Targeted for Trace Analysis by UHPLC-MS
 New Solvent Grade Targeted for Trace Analysis by UHPLC-MS Subhra Bhattacharya, Deva H. Puranam, and Stephen C. Roemer Thermo Fisher Scientific Fisher Chemical, One Reagent Lane, Fair Lawn, NJ Material
New Solvent Grade Targeted for Trace Analysis by UHPLC-MS Subhra Bhattacharya, Deva H. Puranam, and Stephen C. Roemer Thermo Fisher Scientific Fisher Chemical, One Reagent Lane, Fair Lawn, NJ Material
Rapid, Simple Impurity Characterization with the Xevo TQ Mass Spectrometer
 Robert Plumb, Michael D. Jones, and Marian Twohig Waters Corporation, Milford, MA, USA INTRODUCTION The detection and characterization of impurities and degradation products of an active pharmaceutical
Robert Plumb, Michael D. Jones, and Marian Twohig Waters Corporation, Milford, MA, USA INTRODUCTION The detection and characterization of impurities and degradation products of an active pharmaceutical
Measuring Phytosterols in Health Supplements by LC/MS. Marcus Miller and William Schnute Thermo Fisher Scientific, San Jose, CA, USA
 Measuring Phytosterols in Health Supplements by LC/MS Marcus Miller and William Schnute Thermo Fisher Scientific, San Jose, CA, USA Overview Purpose: Develop a method for the extraction of phytosterols
Measuring Phytosterols in Health Supplements by LC/MS Marcus Miller and William Schnute Thermo Fisher Scientific, San Jose, CA, USA Overview Purpose: Develop a method for the extraction of phytosterols
Quantification of lovastatin in human plasma by LC/ESI/MS/MS using the Agilent 6410 Triple Quadrupole LC/MS system
 Quantification of lovastatin in human plasma by LC/ESI/MS/MS using the Agilent 641 Triple Quadrupole LC/MS system Application Note Clinical Research Author Siji Joseph Agilent Technologies Bangalore, India
Quantification of lovastatin in human plasma by LC/ESI/MS/MS using the Agilent 641 Triple Quadrupole LC/MS system Application Note Clinical Research Author Siji Joseph Agilent Technologies Bangalore, India
CHOLESTEROL IS THE PRECURSOR OF STERIOD HORMONES
 HORMONES OF ADRENAL CORTEX R. Mohammadi Biochemist (Ph.D.) Faculty member of Medical Faculty CHOLESTEROL IS THE PRECURSOR OF STERIOD HORMONES CONVERSION OF CHOLESTROL TO PREGNENOLONE MINERALOCORTICOCOIDES
HORMONES OF ADRENAL CORTEX R. Mohammadi Biochemist (Ph.D.) Faculty member of Medical Faculty CHOLESTEROL IS THE PRECURSOR OF STERIOD HORMONES CONVERSION OF CHOLESTROL TO PREGNENOLONE MINERALOCORTICOCOIDES
Simultaneous Quantitative Analysis of Total Catecholamines and Metanephrines in Urine Using 500 MG CLEAN UP CCX2 and LC-MS/MS
 Simultaneous Quantitative Analysis of Total Catecholamines and Metanephrines in Urine Using 500 MG CLEAN UP CCX2 and LC-MS/MS UCT Part Numbers: CUCCX256 - Clean-Up CCX2 (C8 + Carboxylic Acid) 500mg/6mL
Simultaneous Quantitative Analysis of Total Catecholamines and Metanephrines in Urine Using 500 MG CLEAN UP CCX2 and LC-MS/MS UCT Part Numbers: CUCCX256 - Clean-Up CCX2 (C8 + Carboxylic Acid) 500mg/6mL
Rapid and Robust Detection of THC and Its Metabolites in Blood
 Rapid and Robust Detection of THC and Its Metabolites in Blood Application Note Forensics/Doping Control Author Stephan Baumann Agilent Technologies, Inc. Santa Clara CA 95051 USA Abstract A robust method
Rapid and Robust Detection of THC and Its Metabolites in Blood Application Note Forensics/Doping Control Author Stephan Baumann Agilent Technologies, Inc. Santa Clara CA 95051 USA Abstract A robust method
Application Note. Abstract. Authors. Pharmaceutical
 Analysis of xycodone and Its Metabolites-oroxycodone, xymorphone, and oroxymorphone in Plasma by LC/MS with an Agilent ZRBAX StableBond SB-C18 LC Column Application ote Pharmaceutical Authors Linda L.
Analysis of xycodone and Its Metabolites-oroxycodone, xymorphone, and oroxymorphone in Plasma by LC/MS with an Agilent ZRBAX StableBond SB-C18 LC Column Application ote Pharmaceutical Authors Linda L.
Supporting Information
 Supporting Information Development of a High Coverage Pseudotargeted Lipidomics Method Based on Ultra-High Performance Liquid Chromatography-Mass Spectrometry Qiuhui Xuan 1,2#, Chunxiu Hu 1#, Di Yu 1,2,
Supporting Information Development of a High Coverage Pseudotargeted Lipidomics Method Based on Ultra-High Performance Liquid Chromatography-Mass Spectrometry Qiuhui Xuan 1,2#, Chunxiu Hu 1#, Di Yu 1,2,
Author. Introduction. Abstract
 Simultaneous Measurement of Sirolimus and Tacrolimus Concentrations in Blood by Semi-Automated Extraction and Liquid Chromatography-Electrospray Ionization Mass Spectrometry Application Author Gary L.
Simultaneous Measurement of Sirolimus and Tacrolimus Concentrations in Blood by Semi-Automated Extraction and Liquid Chromatography-Electrospray Ionization Mass Spectrometry Application Author Gary L.
PosterREPRINT RAPID, SELECTIVE SCREENING OF URINE SAMPLES FOR GLUCURONIDES BY LC/MS/MS INTRODUCTION ABSTRACT
 ABSTRACT The traditional study of in vivo drug metabolism in plasma or urine samples is often complicated by the presence of many endogenous compounds. Several mass spectrometric techniques are often applied
ABSTRACT The traditional study of in vivo drug metabolism in plasma or urine samples is often complicated by the presence of many endogenous compounds. Several mass spectrometric techniques are often applied
See external label 2 C-8 C Σ=96 tests Cat # 6101Z. Cortisol. Cat # 6101Z
 DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
Mass-Based Purification of Natural Product Impurities Using an Agilent 1260 Infinity II Preparative LC/MSD System
 Application Note Food Testing and Agriculture Mass-Based Purification of Natural Product Impurities Using an Agilent 126 Infinity II Preparative LC/MSD System Authors Florian Rieck and Jörg Hippler Agilent
Application Note Food Testing and Agriculture Mass-Based Purification of Natural Product Impurities Using an Agilent 126 Infinity II Preparative LC/MSD System Authors Florian Rieck and Jörg Hippler Agilent
Simplified Gas Chromatographic Assay for Paracetamol
 Ann. clin. Biochem. 12 (1975) 4 Simplified Gas Chromatographic Assay for Paracetamol M. J. STEWART AND R. G. WILLIS Department of' Clinical Chemistry, Ninewells Hospital and Medical School, Dundee, DD2
Ann. clin. Biochem. 12 (1975) 4 Simplified Gas Chromatographic Assay for Paracetamol M. J. STEWART AND R. G. WILLIS Department of' Clinical Chemistry, Ninewells Hospital and Medical School, Dundee, DD2
Jose Castro-Perez, Henry Shion, Kate Yu, John Shockcor, Emma Marsden-Edwards, Jeff Goshawk Waters Corporation, Milford, MA, U.S. and Manchester, UK
 HIGH-THRUGHPUT REACTIVE METABLITE SCREEIG FR DICLFEAC BY UPLC AD XEV TQ MS WITH SCAWAVE Jose Castro-Perez, Henry Shion, Kate Yu, John Shockcor, Emma Marsden-Edwards, Jeff Goshawk Waters Corporation, Milford,
HIGH-THRUGHPUT REACTIVE METABLITE SCREEIG FR DICLFEAC BY UPLC AD XEV TQ MS WITH SCAWAVE Jose Castro-Perez, Henry Shion, Kate Yu, John Shockcor, Emma Marsden-Edwards, Jeff Goshawk Waters Corporation, Milford,
Adrenocorticotropic Hormone (ACTH) ELISA
 For Research Use Only. Not for use in Diagnostic Procedures. INTENDED USE The GenWay, Inc. ACTH ELISA Kit is intended for the quantitative determination of ACTH Adrenocorticotropic Hormone) in human plasma
For Research Use Only. Not for use in Diagnostic Procedures. INTENDED USE The GenWay, Inc. ACTH ELISA Kit is intended for the quantitative determination of ACTH Adrenocorticotropic Hormone) in human plasma
Simple Method (IS-MRM) to Monitor Lysophospholipids and Phospholipids During LC-MS Method Development via In-Source CID
 Simple Method (IS-MRM) to Monitor Lysophospholipids and Phospholipids During LC-MS Method Development via In-Source CID James Little, Eastman Chemical Company, Kingsport, TN Overview Phospholipids and
Simple Method (IS-MRM) to Monitor Lysophospholipids and Phospholipids During LC-MS Method Development via In-Source CID James Little, Eastman Chemical Company, Kingsport, TN Overview Phospholipids and
Neonatal Screening using Shimadzu LCMS- 8040
 SEG- 2050 Application Note for The Analytical Scientist 11-13 Neonatal Screening using Shimadzu LCMS- 8040 Introduction The development of electrospray tandem mass spectrometry (LCMS/MS) in recent years
SEG- 2050 Application Note for The Analytical Scientist 11-13 Neonatal Screening using Shimadzu LCMS- 8040 Introduction The development of electrospray tandem mass spectrometry (LCMS/MS) in recent years
Vitamin D Metabolite Analysis in Biological Samples Using Agilent Captiva EMR Lipid
 Vitamin D Metabolite Analysis in Biological Samples Using Agilent Captiva EMR Lipid Application Note Clinical Research Authors Derick Lucas and Limian Zhao Agilent Technologies, Inc. Abstract Lipids from
Vitamin D Metabolite Analysis in Biological Samples Using Agilent Captiva EMR Lipid Application Note Clinical Research Authors Derick Lucas and Limian Zhao Agilent Technologies, Inc. Abstract Lipids from
Overview. Introduction. Experimental. Cliquid Software for Routine LC/MS/MS Analysis
 A Fast and Sensitive LC/MS/MS Method for the Quantification and Confirmation of 3 Benzodiazepines and Nonbenzodiazepine Hypnotics in Forensic Urine Samples Cliquid Software for Routine LC/MS/MS Analysis
A Fast and Sensitive LC/MS/MS Method for the Quantification and Confirmation of 3 Benzodiazepines and Nonbenzodiazepine Hypnotics in Forensic Urine Samples Cliquid Software for Routine LC/MS/MS Analysis
Bioanalytical Quantitation of Biotherapeutics Using Intact Protein vs. Proteolytic Peptides by LC-HR/AM on a Q Exactive MS
 Bioanalytical Quantitation of Biotherapeutics Using Intact Protein vs. Proteolytic Peptides by LC-HR/AM on a Q Exactive MS Jenny Chen, Hongxia Wang, Zhiqi Hao, Patrick Bennett, and Greg Kilby Thermo Fisher
Bioanalytical Quantitation of Biotherapeutics Using Intact Protein vs. Proteolytic Peptides by LC-HR/AM on a Q Exactive MS Jenny Chen, Hongxia Wang, Zhiqi Hao, Patrick Bennett, and Greg Kilby Thermo Fisher
Development and Validation of a Liquid Chromatography- Mass Spectrometry method for analysis of Oxysterols in Human Plasma
 1 Development and Validation of a Liquid Chromatography- Mass Spectrometry method for analysis of Oxysterols in Human Plasma Rohini Narayanaswamy University At Buffalo, The State University of New York
1 Development and Validation of a Liquid Chromatography- Mass Spectrometry method for analysis of Oxysterols in Human Plasma Rohini Narayanaswamy University At Buffalo, The State University of New York
Determination of Clarithromycin in Human Plasma by LC-EI Tandem Mass Spectrometry: Application to Bioequivalence Study
 Determination of Clarithromycin in Human Plasma by LC-EI Tandem Mass Spectrometry: Application to Bioequivalence Study Syed N Alvi, Ph.D Clinical Studies & Empirical Ethics Department King Faisal Specialist
Determination of Clarithromycin in Human Plasma by LC-EI Tandem Mass Spectrometry: Application to Bioequivalence Study Syed N Alvi, Ph.D Clinical Studies & Empirical Ethics Department King Faisal Specialist
Fig. 1: Chemical structure of arachidonic acid COOH CH 3
 Elimination of Matrix Effects Using Mixed-mode SPE Plate for High Throughput Analysis of Free Arachidonic Acid in Plasma by LC-MS/MS Wan Wang, Suzi Qin, Linsen Li, Warren Chen, Jerry Wang 179, Southern
Elimination of Matrix Effects Using Mixed-mode SPE Plate for High Throughput Analysis of Free Arachidonic Acid in Plasma by LC-MS/MS Wan Wang, Suzi Qin, Linsen Li, Warren Chen, Jerry Wang 179, Southern
SUPPLEMENTARY MATERIAL
 SUPPLEMENTARY MATERIAL Artepillin C, is it a good marker for quality control of Brazilian Green Propolis? Cui-ping Zhang 1, Xiao-ge Shen 1, Jia-wei Chen 1, Xia-sen Jiang 1, Kai Wang 2, Fu-liang Hu 1 *
SUPPLEMENTARY MATERIAL Artepillin C, is it a good marker for quality control of Brazilian Green Propolis? Cui-ping Zhang 1, Xiao-ge Shen 1, Jia-wei Chen 1, Xia-sen Jiang 1, Kai Wang 2, Fu-liang Hu 1 *
Glycerolipid Analysis. LC/MS/MS Analytical Services
 Glycerolipid Analysis LC/MS/MS Analytical Services Molecular Characterization and Quantitation of Glycerophospholipids in Commercial Lecithins by High Performance Liquid Chromatography with Mass Spectrometric
Glycerolipid Analysis LC/MS/MS Analytical Services Molecular Characterization and Quantitation of Glycerophospholipids in Commercial Lecithins by High Performance Liquid Chromatography with Mass Spectrometric
PHOTOCATALYTIC DECONTAMINATION OF CHLORANTRANILIPROLE RESIDUES IN WATER USING ZnO NANOPARTICLES. DR. A. RAMESH, Ph.D, D.Sc.,
 PHOTOCATALYTIC DECONTAMINATION OF CHLORANTRANILIPROLE RESIDUES IN WATER USING ZnO NANOPARTICLES DR. A. RAMESH, Ph.D, D.Sc., raamesh_a@yahoo.co.in 1 OBJECTIVES Determination of persistence and photolysis
PHOTOCATALYTIC DECONTAMINATION OF CHLORANTRANILIPROLE RESIDUES IN WATER USING ZnO NANOPARTICLES DR. A. RAMESH, Ph.D, D.Sc., raamesh_a@yahoo.co.in 1 OBJECTIVES Determination of persistence and photolysis
Determination and pharmacokinetics of manidipine in human plasma by HPLC/ESIMS
 BIOMEDICAL CHROMATOGRAPHY Biomed. Chromatogr. 21: 836 840 (2007) Published 836 online ORIGINAL 12 April RESEARCH 2007 in Wiley InterScience ORIGINAL RESEARCH (www.interscience.wiley.com).827 Determination
BIOMEDICAL CHROMATOGRAPHY Biomed. Chromatogr. 21: 836 840 (2007) Published 836 online ORIGINAL 12 April RESEARCH 2007 in Wiley InterScience ORIGINAL RESEARCH (www.interscience.wiley.com).827 Determination
Result ID Reporting Name Type Unit LOINC. Z1886 Cortisol Alphanumeric ug/dl Z0226 Deoxycorticosterone (DOC) Alphanumeric ng/dl
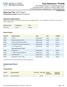 Reporting Title: CAH Profile 6 Performing Location: Esoterix Endocrinol Specimen Requirements: Draw blood in a plain, red-top tube(s). Spin down and separate within 1 hour of collection and send 3.5 ml
Reporting Title: CAH Profile 6 Performing Location: Esoterix Endocrinol Specimen Requirements: Draw blood in a plain, red-top tube(s). Spin down and separate within 1 hour of collection and send 3.5 ml
A Definitive Lipidomics Workflow for Human Plasma Utilizing Off-line Enrichment and Class Specific Separation of Phospholipids
 A Definitive Lipidomics Workflow for Human Plasma Utilizing Off-line Enrichment and Class Specific Separation of Phospholipids Jeremy Netto, 1 Stephen Wong, 1 Federico Torta, 2 Pradeep Narayanaswamy, 2
A Definitive Lipidomics Workflow for Human Plasma Utilizing Off-line Enrichment and Class Specific Separation of Phospholipids Jeremy Netto, 1 Stephen Wong, 1 Federico Torta, 2 Pradeep Narayanaswamy, 2
SUPPORTING INFORMATION FOR: CONCENTRATIONS OF POLYBROMINATED DIPHENYL ETHERS, HEXABROMOCYCLODODECANES AND TETRABROMOBISPHENOL-A IN BREAST MILK FROM
 SUPPORTING INFORMATION FOR: CONCENTRATIONS OF POLYBROMINATED DIPHENYL ETHERS, HEXABROMOCYCLODODECANES AND TETRABROMOBISPHENOL-A IN BREAST MILK FROM UNITED KINGDOM WOMEN DO NOT DECREASE OVER TWELVE MONTHS
SUPPORTING INFORMATION FOR: CONCENTRATIONS OF POLYBROMINATED DIPHENYL ETHERS, HEXABROMOCYCLODODECANES AND TETRABROMOBISPHENOL-A IN BREAST MILK FROM UNITED KINGDOM WOMEN DO NOT DECREASE OVER TWELVE MONTHS
25-Hydroxyvitamin D2-D3 Serum LC-MS/MS APCI Analysis Kit User Manual
 Page 1 / 15 25-Hydroxyvitamin D2-D3 Serum LC-MS/MS APCI Analysis Kit User Manual ZV-3046-0200-10 200 Page 2 / 15 Table of Contents 1. INTENDED USE... 3 2. SUMMARY AND EXPLANATION... 3 3. IVD SYMBOLS...
Page 1 / 15 25-Hydroxyvitamin D2-D3 Serum LC-MS/MS APCI Analysis Kit User Manual ZV-3046-0200-10 200 Page 2 / 15 Table of Contents 1. INTENDED USE... 3 2. SUMMARY AND EXPLANATION... 3 3. IVD SYMBOLS...
Liquid Liquid Extraction of Gamma Hydroxybutyric Acid (GHB) in Blood and Urine for GC-MS analysis
 Liquid Liquid Extraction of Gamma Hydroxybutyric Acid (GHB) in Blood and Urine for GC-MS analysis 1.0 Purpose - This procedure specifies the required elements for the extraction of GHB from blood, urine
Liquid Liquid Extraction of Gamma Hydroxybutyric Acid (GHB) in Blood and Urine for GC-MS analysis 1.0 Purpose - This procedure specifies the required elements for the extraction of GHB from blood, urine
Screening by immunoassay and confirmation & quantitation by GC-MS of buprenorphine and norbuprenorphine in urine, whole blood and serum
 Screening by immunoassay and confirmation & quantitation by GC-MS of buprenorphine and norbuprenorphine in urine, whole blood and serum NINA KANGAS, SIRPA MYKKÄNEN, SANNA KYLLÖNEN, PÄIVI RAJALA, KARI ARINIEMI
Screening by immunoassay and confirmation & quantitation by GC-MS of buprenorphine and norbuprenorphine in urine, whole blood and serum NINA KANGAS, SIRPA MYKKÄNEN, SANNA KYLLÖNEN, PÄIVI RAJALA, KARI ARINIEMI
