PRODUCT INFORMATION H 3 CO. paracetamol MW codeine phosphate hemihydrate MW
|
|
|
- Calvin Berry
- 5 years ago
- Views:
Transcription
1 RODUCT INFORMATION RODEINE NAME OF THE MEDICINE Non-proprietary Name aracetamol, codeine phosphate hemihydrate Chemical Structure H N CH 3 HO O H.H 3 O 4. 1/2 H 2 0 NH CH 3 H 3 CO paracetamol MW codeine phosphate hemihydrate MW CAS Number CAS (paracetamol). CAS (codeine phosphate hemihydrate) DESCRITION rodeine contains paracetamol 500 mg and codeine phosphate hemihydrate 9.6 mg. aracetamol is an odourless, crystalline powder or crystals with a bitter taste. Codeine phosphate hemihydrate is an odourless, crystalline powder or small colourless crystals with a bitter taste. rodeine also contains compap L, sodium starch glycollate, purified talc and magnesium stearate. rodeine is aspirin-free. O OH HARMACOLOGY aracetamol is an effective and fast-acting analgesic which relieves mild to moderate pain. It is rapidly absorbed from the gastrointestinal tract with peak plasma levels usually reached half to one hour after oral administration. It also reduces fever by a direct effect on the heat-regulating centres to increase dissipation of body heat. Codeine phosphate hemihydrate is an effective oral analgesic which provides relief from mild to moderate pain. It is also well absorbed from the gastrointestinal tract after oral administration. The abuse potential of codeine is lower than that of other opiates. harmacokinetics Absorption Food intake delays paracetamol absorption. Codeine has about one-sixth of morphine's analgesic activity. It is well absorbed from the gastrointestinal tract and does not interfere with paracetamol absorption. 1
2 Distribution aracetamol is uniformly distributed throughout most body fluids; the apparent volume of distribution is 1 to 1.2 L/kg. aracetamol can cross the placenta and is excreted in milk. lasma protein binding is negligible at usual therapeutic concentrations but increases with increasing concentrations. Metabolism aracetamol is metabolised by the hepatic microsomal enzyme system. In adults at therapeutic doses, paracetamol is mainly conjugated with glucuronide (45-55%) or sulfate (20-30%). A minor proportion (less than 20%) is metabolised to catechol derivatives, and mercapturic acid compounds via oxidation. The metabolites of paracetamol include a minor hydroxylated intermediate which has hepatotoxic activity. This intermediate metabolite is detoxified by conjugation with glutathione, however, it can accumulate following paracetamol overdosage (more than 150 mg/kg or 10 mg total paracetamol ingested) and if left untreated can cause irreversible liver damage. aracetamol is metabolised differently by infants and children compared to adults, the sulfate conjugate being predominant. Excretion aracetamol is excreted in the urine mainly as the glucuronide and sulphate conjugates. Less than 5% is excreted as unchanged paracetamol with 85-90% of the administered dose eliminated in the urine within 24 hours of ingestion. The elimination half-life varies from 1 to 4 hours. Codeine is metabolised in the liver to morphine and norcodeine, which with codeine, are excreted in the urine, partly as conjugates with glucuronic acid. Excretion is almost complete within 24 hours. atients who metabolise drugs poorly via CY2D6 are likely to obtain reduced benefit from codeine due to reduced formation of the active metabolite. The plasma half-life of codeine has been reported to be between 3 and 4 hours after oral administration. INDICATIONS For the relief of acute moderate pain and fever. CONTRAINDICATIONS rodeine is contraindicated in patients: with known hypersensitivity to paracetamol, codeine or any of the excipients used in this product. with pre-existing respiratory depression. with glucose-6-phosphate-dehydrogenase deficiency. with severely impaired liver or renal function. with diarrhoea caused by pseudomembranous colitis or poisoning (until the causative organism or toxin has been eliminated from the gastrointestinal tract, since codeine may slow down the elimination, thereby prolonging the diarrhoea). with chronic constipation. 2
3 with active alcoholism. during labour when delivery of a premature infant is anticipated as it may produce codeine withdrawal symptoms in neonate. during breast-feeding (see RECAUTIONS Use in Lactation) Aged between years in whom respiratory function might be compromised, including post tonsillectomy and/or adenoidectomy for obstructive sleep apnoea, due to an increased risk of developing serious and life-threatening adverse reactions. (see RECAUTIONS aediatric Use). who are CY 2D6 ultra-rapid metabolisers (see RECAUTIONS- CY2D6 metabolism).. rodeine is contraindicated for use in patients younger than 12 years (see RECAUTIONS aediatric Use). RECAUTIONS rodeine should be used with caution in hepatic or renal dysfunction. Hepatotoxicity may occur with paracetamol even at therapeutic doses, after short treatment duration and in patients without pre-existing liver dysfunction. To avoid the risk of overdose: Check that paracetamol is absent from the composition of other medicinal products taken concomitantly. Dosage should be reduced in elderly or debilitated patients because of the danger of respiratory or cardiac depression. Elderly people may be more sensitive to the effects of this medicinal product, especially respiratory depression. They are also more prone to suffering hypertrophy, prostatic obstruction and age-related renal impairment and they have a higher likelihood of undesirable effects due to opioidinduced urinary retention. Codeine should be used with caution in patients with CNS depression or decreased respiratory reserve eg emphysema, kyphoscoliosis, or severe obesity. Codeine should be administered with caution in patients with impaired cardiac, hepatic or renal function, and in cases of urethral stenosis, gallbladder conditions, multiple sclerosis, hypothyroidism, adrenocortical insufficiency, shock, acute alcohol intoxication or prostatic hyperplasia, hypotension, myasthenia gravis, inflammatory or obstructive bowel disorders, recent gastrointestinal tract surgery, raised intracranial pressure or head injury. atients who have had a cholecystectomy should be treated with caution. The contraction of the sphincter of Oddi can cause symptoms resembling those of myocardial infarction or intensify the symptoms in patients with pancreatitis. Codeine should be used with caution in patients with convulsive disorders. Extensive use of analgesics to relieve headaches or migraines, especially at high doses, may induce headaches that must not be treated with increased doses of the drug. In such cases the analgesic should not continue to be taken without medical advice. rolonged use of high dose of codeine may produce dependence and or addiction. Codeine has a primary potential for dependence. Tolerance, psychological and physical dependence develop with prolonged use of high doses with withdrawal symptoms after sudden discontinuation of the drug. Cross-tolerance with other opioids exists. Rapid relapses can be expected in patients with pre-existing opiate dependence (including those in remission). 3
4 Administration must be discontinued gradually after prolonged treatments. Monitoring after prolonged use should include blood count, liver function and renal function. Codeine should only be used after careful risk-benefit assessment in case of: Opioid dependence Conditions with elevated intracranial pressure and head trauma. Codeine can increase the pressure of cerebrospinal fluid and may increase the respiratory depressant effect. Like other narcotics, it causes adverse reactions that can obscure the clinical course of patients with head injury. Impaired consciousness Compromised respiratory function (due to emphysema, kyphoscoliosis, severe obesity) and chronic obstructive airway disease The codeine in rodeine may cause drowsiness, disturbances of visuomotor coordination and visual acuity, impairing the mental and or physical ability required for the performance of potentially dangerous tasks, in some people. Those affected should not drive or operate machinery or engage in activities which require them to be alert. roducts containing codeine should not be taken in large amounts for prolonged periods as they may be habit-forming and should be used with caution in patients with a history of drug abuse. This medication may be dangerous when used in large amounts or for long periods. Hepatotoxicity may develop following a dose of 10 g of paracetamol and hepatic failure is known to occur occasionally with the long term use of paracetamol. atients with known analgesic intolerance or known bronchial asthma must only use rodeine after having consulted a physician (hypersensitivity reactions including bronchospasm are possible). Caution is advised in patients with underlying sensitivity to aspirin and/or to nonsteroidal anti-inflammatory drugs (NSAIDs).* Severe cutaneous adverse reactions (SCARs): Life threatening cutaneous reactions Stevens-Johnson syndrome (SJS), and Toxic epidermal necrolysis (TEN) have been reported with the use of paracetamol. atients should be advised of the signs and symptoms and monitored closely for skin reactions. If symptoms or signs of SJS and TEN (e.g. progressive skin rash often with blisters or mucosal lesions) occur, patients should stop paracetamol treatment immediately and seek medical advice. aracetamol should be used upon medical advice in patients with: mild to moderate hepatocellular insufficiency severe renal insufficiency chronic alcohol use including recent cessation of alcohol intake low glutathione reserves Gilbert s syndrome CY2D6 metabolism rodeine is contraindicated for use in patients who are CY2D6 ultra-rapid metabolisers. 4
5 Codeine is metabolised by the liver enzyme CY2D6 into morphine, its active metabolite. If a patient has a deficiency or is completely lacking this enzyme an adequate analgesic effect will not be obtained. However, if the patient is an extensive or ultra-rapid metaboliser, there is an increased risk of developing side effects of opioid toxicity even at commonly prescribed doses. These patients convert codeine into morphine rapidly resulting in higher than expected serum morphine levels. General symptoms of opioid toxicity include confusion, somnolence, shallow breathing, small pupils, nausea, vomiting, constipation and lack of appetite. In severe cases this may include symptoms of circulatory and respiratory depression, which may be life-threatening and very rarely fatal. Children are particularly susceptible due to their immature airway anatomy. Deaths have been reported in children with rapid metabolism who were given codeine for analgesia post adenotonsillectomy. Morphine can also be ingested by infants through breast milk, causing risk of respiratory depression to infants of rapid metaboliser mothers who take codeine. The prevalence of codeine ultra-rapid metabolism by CY 2D6 in children is not known, but is assumed to be similar to that reported in adults. The prevalence of ultra-rapid metabolisers differs according to racial and ethnic group. It is estimated to be 1% in those of Chinese, Japanese and Hispanic descent, 3% in African Americans and 1%-10% in Caucasians. The highest prevalence (16%-28%) occurs in North African, Ethiopian and Arab populations. (See also the sections on aediatric Use and Use in Lactation.) Codeine is not recommended for use in children in whom respiratory function might be compromised. Risks from Concomitant Use of Opioids and Benzodiazepines Concomitant use of opioids, including codeine, with benzodiazepines may result in sedation, respiratory depression, coma and death. Because of these risks, reserve concomitant prescribing of opioids and benzodiazepines for use in patients for whom alternative treatment options are inadequate. If a decision is made to prescribe codeine concomitantly with benzodiazepines, prescribe the lowest effective dosages and minimum durations of concomitant use, and follow patients closely for signs and symptoms of sedation and respiratory depression (see INTERACTIONS). atients should be advised to first consult their healthcare professional before taking codeine if they are taking a benzodiazepine (see INTERACTIONS). Risks from Concomitant Use of Opioids and Alcohol Concomitant use of opioids, including codeine, with alcohol may result in sedation, respiratory depression, coma and death. Concomitant use with alcohol is not recommended (see INTERACTIONS). Use in regnancy Category A. There have been no observations of an increase in the frequency of malformations or other direct or indirect harmful effects on the foetus in pregnant women and women of child-bearing age who have taken the active ingredients in rodeine. However, prolonged high-dose use of codeine prior to delivery may produce codeine withdrawal symptoms in the neonate. Opioid analgesics may cause respiratory 5
6 depression in the newborn infant. Codeine may cause respiratory depression and withdrawal syndrome in neonates born to mothers who use codeine during the third trimester of pregnancy. As a precautionary measure, use of rodeine should be avoided during the third trimester of pregnancy and during labour. rodeine should only be used during pregnancy under medical supervision if the potential benefit justifies the potential risk to the foetus. If administered during pregnancy, morphinomimetic properties of codeine should be taken into account. Use in Lactation rodeine is contraindicated during breast-feeding (see CONTRAINDICATIONS see also RECAUTIONS-CY2D6 metabolism) due to risk of respiratory depression in the infant.). There is no data available on the use of rodeine during lactation. If rodeine is administered to a breastfeeding mother, alternative arrangements should be made for feeding the infant. aracetamol and codeine pass into breast milk. Analgesic doses excreted in breast milk are generally low. However, infants of breastfeeding mothers taking codeine may have an increased risk of morphine overdose if the mother is an ultrarapid metaboliser of codeine. Codeine is excreted into human breast milk. Codeine is partially metabolized by cytochrome 450 2D6 (CY2D6) into morphine, which is excreted into breast milk. If nursing mothers are CY2D6 ultra-rapid metabolisers, higher levels of morphine may be present in their breast milk. This may result in symptoms of opioid toxicity in both mother and the breast-fed infant. Life-threatening adverse events or neonatal death may occur even at therapeutic doses (see RECAUTIONS CY2D6 metabolism). Therefore, rodeine is contraindicated for use during breastfeeding. However, in circumstances where a breastfeeding mother requires codeine therapy, breastfeeding should be suspended and alternative arrangements should be made for feeding the infant for any period during codeine treatment. Breastfeeding mothers should be told how to recognise signs of high morphine levels in themselves and their babies. For example, in a mother symptoms include extreme sleepiness and trouble caring for the baby. In the baby, symptoms include signs of increased sleepiness (more than usual), difficulty breastfeeding, breathing difficulties or limpness. Medical advice should be sought immediately. aediatric Use This product is contraindicated for use in children: Younger than 12 years. Aged between years in whom respiratory function might be compromised, including post tonsillectomy and/or adenoidectomy for obstructive sleep apnoea. Respiratory depression and death have occurred in some children who received codeine following tonsillectomy and/or adenoidectomy and had evidence of being ultra-rapid metabolisers of codeine due to a CY2D6 polymorphism. (See also RECAUTIONS CY2D6 metabolism.) Effects on laboratory tests Uric acid and blood glucose: Intake of paracetamol may affect the laboratory determination of uric acid by phosphotungstic acid and of blood glucose by glucose oxidase-peroxidase. 6
7 INTERACTIONS WITH OTHER MEDICINES atients receiving other narcotic analgesics, antitussives, antihypertensives, antihistamines, antipsychotics, antianxiety or other CNS depressants eg. hypnotics, sedatives, tranquillisers, including alcohol, concomitantly may exhibit an additive CNS depression. Concurrent use of codeine and other opioid analgesics is usually inappropriate as additive CNS depression, respiratory depression and other hypotensive effects may occur. Morphinic agonists-antagonists: Concomitant use of codeine with a partial agonist (e.g. buphenorphine) or antagonist (e.g. naltrexone) can precipitate or delay codeine effects. The concomitant use of benzodiazepines and opioids increases the risk of sedation, respiratory depression, coma and death because of additive CNS depressant effect. Limit dosage and duration of concomitant use of benzodiazepines and opioids (see RECAUTIONS). The concomitant use of alcohol and opioids increases the risk of sedation, respiratory depression, coma and death because of additive CNS depressant effect. Concomitant use with alcohol is not recommended (see RECAUTIONS). Concurrent administration or use within 14 days of ceasing monoamine oxidase inhibitors (MAOIs) may enhance the potential respiratory depressant effects of codeine and other side effects of unpredictable severity. A codeine-induced respiratory depression can be potentiated by tricyclic antidepressants. Codeine may increase the hypotensive effects of antihypertensive agents. Other potential interactions with codeine include anticholinergic agents or antidiarrhoeal agents (due to the risk of severe constipation and CNS depression); metoclopramide (as codeine may antagonise the effects of this medicine on gastrointestinal motility); and drugs that can inhibit CY2D6 such as quinidine, phenothiazines and antipsychotic agents as they can interfere with the metabolism of codeine to morphine, reducing the analgesic effect of codeine. The risk of paracetamol toxicity may be increased in patients receiving other potentially hepatotoxic drugs or drugs that induce liver microsomal enzymes, such as barbiturates and other antiepileptics (such as phenobarbital, phenytoin, carbamazepine, topiramate), rifampicin and alcohol. aracetamol may increase the risk of bleeding in patients taking warfarin and other antivitamin K. atients taking paracetamol and antivitamin K should be monitored for appropriate coagulation and bleeding complications. aracetamol may considerably slow down the excretion of chloramphenicol, entailing the risk of increased toxicity. When used concurrently with zidovudine, an increased tendency for neutropenia may develop. Combination of rodeine and zidovudine should be avoided. Concurrent intake of drugs, which delay gastric emptying, such as propantheline, may slow down the uptake of paracetamol, thereby retarding its onset of action. Conversely, drugs, which accelerate gastric emptying, such as metoclopramide or domperidine, may accelerate the uptake of paracetamol and its onset of action. Cholestyramine reduces the absorption of paracetamol if given within 1 hour of paracetamol. aracetamol excretion may be affected and plasma concentrations altered when given with probenecid. Chelating resin can decrease the intestinal absorption of paracetamol and potentially decrease its efficacy if taken simultaneously. In general, there must be an interval of more than 2 hours between taking the resin and taking paracetamol, if possible. 7
8 Co-administration of flucloxacillin with paracetamol may lead to metabolic acidosis, particularly in patients presenting risk factors of glutathione depletion, such as sepsis, malnutrition or chronic alcoholism. ADVERSE EFFECTS Reports of adverse reactions with paracetamol are rare. Dyspepsia, nausea, sweating, erythema, urticaria, anaphylactic shock, angioneurotic oedema, difficulty in breathing, skin rashes, drop in blood pressure, allergic and haematological reactions (thrombocytopenia, leukopenia, neutropenia, anaemia, agranulocytosis & pancytopenia) have been reported. A causal relationship to the administration of paracetamol has not been confirmed. Toxic epidermal necrolysis (TEN), Stevens- Johnson syndrome (SJS), acute generalised exanthematous pustulosis, fixed drug eruption (see RECAUTIONS) and cytolytic hepatitis, which may lead to acute hepatic failure, have also been reported. Bronchospasm may be triggered in patients having a tendency of analgesic asthma. Constipation, dry mouth, nausea and vomiting, dizziness and drowsiness may occur in association with codeine. Other side effects are rare, especially at dosage levels provided with rodeine. These include cough suppression, respiratory depression, confusional state, seizure, headache, somnolence, fatigue, sedation, euphoria, dysphoria, skin rashes, pruritis, histamine release (hypotension, flushing of the face, tachycardia, breathlessness) and other allergic reactions. Long term use also entails the risk of drug dependence. Tinnitus and miosis have been associated with codeine use. Visuomotor coordination and visual acuity may be adverse affected in a dose-dependent manner at higher doses or in particularly sensitive patients. Very rarely, skin rashes may occur in patients hypersensitive to codeine. ancreatitis has been reported very rarely. Renal failure, uraemia, urinary retention or hesitance have been reported less frequently to rarely when both paracetamol and codeine have been administered. Haemolytic anaemia in patients with underlying glucose 6-phosphate-dehydrogenase deficiency has been reported. Kounis syndrome and bronchospasm have also been reported. DOSAGE AND ADMINISTRATION Adults and children 12 years of age and older: One or two caplets every 3 to 4 hours as needed for relief. Do not exceed 8 caplets in 24-hour period. rodeine is not recommended for use over extended periods of time. Use in children under 12 years is contraindicated. OVERDOSAGE Elderly persons, small children, patients with liver disorders, chronic alcohol consumption or chronic malnutrition, as well as patients concomitantly treated with enzyme-inducing drugs are at an increased risk of intoxication, including fatal outcome. It has been reported that paracetamol may produce symptoms of acute toxicity in adults, following the ingestion of more than 15g. Hepatotoxicity may develop after 8
9 the ingestion of a single dose of 10 to 15g (200 to 250 mg/kg) and a dose of more than 25g is potentially fatal. Nausea, vomiting, anorexia, pallor and abdominal pain generally appear during the first 24 hours of overdosage with paracetamol. Overdosage with paracetamol may cause hepatic cytolysis which can lead to hepatocellular insufficiency, gastrointestinal bleeding, metabolic acidosis, encephalopathy, coma and death. Increased levels of hepatic transaminases, lactate dehydrogenase and bilirubin with a reduction in prothrombin level can appear 12 to 48 hours after acute overdosage. It can also lead to pancreatitis, acute renal failure and pancytopenia. atients may be asymptomatic for several days following ingestion of large doses of paracetamol and laboratory evidence of hepatotoxicity may be delayed for up to one week. Non-fatal hepatic damage is usually reversible. The antidote, N-acetylcysteine, should be administered as early as possible. Despite lack of significant early symptoms, patients should be referred to hospital urgently for immediate medical attention. Determinations of the plasma concentration of paracetamol are recommended. lasma concentration of paracetamol should be measured at 4 hours or later after ingestion (earlier concentrations are unreliable). Where paracetamol intoxication is suspected, intravenous administration of SH group donators such as acetylcysteine within the first 10 hours after ingestion is indicated. Although acetylcysteine is most effective if initiated within this period, it can still offer some degree of protection if given as late as 48 hours after ingestion; in this case it is taken for longer. In an evaluation of codeine intoxication in children, symptoms ranked by decreasing order of frequency included sedation, rash, miosis, vomiting, itching, ataxia and swelling of the skin. Respiratory failure may occur. Blood concentrations of codeine ranged from 1.4 to 5.6 g/ml in eight adults whose deaths were attributed primarily to codeine overdosage. The ingestion of very high doses of codeine can cause initial excitation, anxiety, insomnia followed by drowsiness in certain cases, areflexia progressing to stupor or coma, headache, miosis, alterations in blood pressure, arrhythmias, dry mouth, hypersensitivity reactions, cold clammy skin, bradycardia, tachycardia, convulsions, gastrointestinal disorders, nausea, vomiting and respiratory depression. Severe intoxication can lead to apnoea, circulatory collapse, cardiac arrest and death. Relating to codeine component: In general, treatment should be symptomatic: re-establish adequate respiratory exchange by ensuring a clear airway and using mechanical ventilation. When treatment for paracetamol toxicity has been initiated the opioid antagonist naloxone hydrochloride is an antidote to respiratory depression; naloxone 400 microgram may be administered SC, IM or IV. Further measures will depend on the severity, nature and course of clinical symptoms of intoxication and should follow standard intensive care protocols. For information on the management of overdose contact the oisons Information Centre on (Australia). RESENTATION AND STORAGE CONDITIONS rodeine is available as caplets of 12*, 24, 36*, or 40, each containing paracetamol 500mg and codeine phosphate hemihydrate 9.6 mg. 9
10 The caplets are white capsule shaped tablets, plain on one side and a breakline on the other. Storage: Store below 30 C *Not marketed NAME AND ADDRESS OF THE SONSOR sanofi-aventis australia pty ltd Talavera Road Macquarie ark NSW 2113 OISON SCHEDULE OF THE MEDICINE RESCRITION ONLY MEDICINE (Schedule 4) DATE OF FIRST INCLUSION IN THE ARTG 4 September 2012 DATE OF MOST RECENT AMENDMENT 26 September 2017 * Changes of clinical significance 10
PRODUCT INFORMATION CODAPANE XTRA Paracetamol 500 mg and Codeine Phosphate 15 mg Tablets
 PRODUCT INFORMATION CODAPANE XTRA Paracetamol 500 mg and Codeine Phosphate 15 mg Tablets NAME OF THE MEDICINE Active Ingredients: Paracetamol and Codeine Phosphate Paracetamol: Molecular Formula: C 8 H
PRODUCT INFORMATION CODAPANE XTRA Paracetamol 500 mg and Codeine Phosphate 15 mg Tablets NAME OF THE MEDICINE Active Ingredients: Paracetamol and Codeine Phosphate Paracetamol: Molecular Formula: C 8 H
PRODUCT INFORMATION Panadeine EXTRA
 PRODUCT INFORMATION Panadeine EXTRA COMPOSITION Each caplet brand of capsule-shaped tablet contains: Paracetamol 500 mg Codeine phosphate 15 mg and Maize Starch Purified Talc Pregelatinised Maize Starch
PRODUCT INFORMATION Panadeine EXTRA COMPOSITION Each caplet brand of capsule-shaped tablet contains: Paracetamol 500 mg Codeine phosphate 15 mg and Maize Starch Purified Talc Pregelatinised Maize Starch
PRODUCT INFORMATION PANADEINE FORTE
 PRODUCT INFORMATION PANADEINE FORTE NAME OF THE MEDICINE Non-proprietary Name Paracetamol and codeine phosphate DESCRIPTION Each capsule-shaped tablet contains Paracetamol 500 mg, Codeine phosphate 30
PRODUCT INFORMATION PANADEINE FORTE NAME OF THE MEDICINE Non-proprietary Name Paracetamol and codeine phosphate DESCRIPTION Each capsule-shaped tablet contains Paracetamol 500 mg, Codeine phosphate 30
AUSTRALIAN PRODUCT INFORMATION, PAINSTOP DAY-TIME PAIN RELIEVER (PARACETAMOL 120MG IN 5ML AND CODEINE PHOSPHATE HEMIHYDRATE 5MG IN 5ML)
 AUSTRALIAN PRODUCT INFORMATION, PAINSTOP DAY-TIME PAIN RELIEVER (PARACETAMOL 120MG IN 5ML AND CODEINE PHOSPHATE HEMIHYDRATE 5MG IN 5ML) 1. NAME OF THE MEDICINE Paracetamol and codeine phosphate hemihydrate.
AUSTRALIAN PRODUCT INFORMATION, PAINSTOP DAY-TIME PAIN RELIEVER (PARACETAMOL 120MG IN 5ML AND CODEINE PHOSPHATE HEMIHYDRATE 5MG IN 5ML) 1. NAME OF THE MEDICINE Paracetamol and codeine phosphate hemihydrate.
PRODUCT INFORMATION MERSYNDOL FORTE TABLETS
 PRODUCT INFORMATION MERSYNDOL FORTE TABLETS NAME OF THE MEDICINE Non-proprietary Name Paracetamol, codeine phosphate and doxylamine succinate DESCRIPTION Each tablet contains paracetamol 450 mg, codeine
PRODUCT INFORMATION MERSYNDOL FORTE TABLETS NAME OF THE MEDICINE Non-proprietary Name Paracetamol, codeine phosphate and doxylamine succinate DESCRIPTION Each tablet contains paracetamol 450 mg, codeine
PRODUCT INFORMATION PRODEINE FORTE H 3 CO. Paracetamol MW Codeine phosphate hemihydrate MW
 PRODUCT INFORMATION PRODEINE FORTE NAME OF THE MEDICINE Australian Approved Name Paracetamol and codeine phosphate hemihydrate Chemical Structure H N CH 3 HO O H.H 3 PO 4. 1/2 H 2 0 NH CH 3 H 3 CO Paracetamol
PRODUCT INFORMATION PRODEINE FORTE NAME OF THE MEDICINE Australian Approved Name Paracetamol and codeine phosphate hemihydrate Chemical Structure H N CH 3 HO O H.H 3 PO 4. 1/2 H 2 0 NH CH 3 H 3 CO Paracetamol
PRODUCT INFORMATION APO-Paracetamol/Codeine 500/30
 PRODUCT INFORMATION APO-Paracetamol/Codeine 500/30 NAME OF THE MEDICINE Non-proprietary Name Paracetamol and codeine phosphate Chemical Structure Paracetamol MW 151.17 Codeine phosphate MW 406.37 CAS Numbers
PRODUCT INFORMATION APO-Paracetamol/Codeine 500/30 NAME OF THE MEDICINE Non-proprietary Name Paracetamol and codeine phosphate Chemical Structure Paracetamol MW 151.17 Codeine phosphate MW 406.37 CAS Numbers
PRODUCT INFORMATION PRODEINE 15 H 3 CO. Paracetamol MW Codeine phosphate MW
 PRODUCT INFORMATION PRODEINE 15 NAME OF THE MEDICINE Non-proprietary Name Paracetamol and codeine phosphate. Chemical Structure HO O H N CH 3 NH CH 3 H H 3 CO Paracetamol MW 151.17 Codeine phosphate MW
PRODUCT INFORMATION PRODEINE 15 NAME OF THE MEDICINE Non-proprietary Name Paracetamol and codeine phosphate. Chemical Structure HO O H N CH 3 NH CH 3 H H 3 CO Paracetamol MW 151.17 Codeine phosphate MW
PRODUCT INFORMATION ACTACODE
 PRODUCT INFORMATION ACTACODE NAME OF THE MEDICINE Actacode codeine linctus contains codeine phosphate 5 mg/ml. Codeine phosphate is (5R,6S)-7,8-didehydro-4,5-epoxy-3-methoxy-N-methylmorphinan-6-ol dihydrogen
PRODUCT INFORMATION ACTACODE NAME OF THE MEDICINE Actacode codeine linctus contains codeine phosphate 5 mg/ml. Codeine phosphate is (5R,6S)-7,8-didehydro-4,5-epoxy-3-methoxy-N-methylmorphinan-6-ol dihydrogen
Migraleve, Migraleve Pink and Migraleve Yellow Product Information
 Migraleve, Migraleve Pink and Migraleve Yellow Product Information Adverse events should be reported. Reporting forms and information can be found at www.mhra.gov.uk/yellowcard Adverse events should also
Migraleve, Migraleve Pink and Migraleve Yellow Product Information Adverse events should be reported. Reporting forms and information can be found at www.mhra.gov.uk/yellowcard Adverse events should also
PRODUCT INFORMATION APOHEALTH PARACETAMOL PLUS CODEINE & CALMATIVE paracetamol 500mg codeine phosphate 10mg and doxylamine succinate 5.
 Product Information Page 1 PRODUCT INFORMATION APOHEALTH PARACETAMOL PLUS CODEINE & CALMATIVE paracetamol 500mg codeine phosphate 10mg and doxylamine succinate 5.1mg tablets NAME OF THE MEDICINE Paracetamol
Product Information Page 1 PRODUCT INFORMATION APOHEALTH PARACETAMOL PLUS CODEINE & CALMATIVE paracetamol 500mg codeine phosphate 10mg and doxylamine succinate 5.1mg tablets NAME OF THE MEDICINE Paracetamol
Molecular formula: Molecular weight: C 8 H 9 NO 2 CAS Registry no.:
 Parapane Paracetamol PRODUCT INFORMATION NAME OF THE MEDICINE Active ingredient: Chemical name: Paracetamol N-(4-hydroxyphenyl) Structural formula: Molecular formula: 151.20 Molecular weight: C 8 H 9 NO
Parapane Paracetamol PRODUCT INFORMATION NAME OF THE MEDICINE Active ingredient: Chemical name: Paracetamol N-(4-hydroxyphenyl) Structural formula: Molecular formula: 151.20 Molecular weight: C 8 H 9 NO
PRODUCT INFORMATION. Codral* Original Cold & Flu Tablets
 PRODUCT INFORMATION Codral* Original Cold & Flu Tablets Product description Codral* Original Cold & Flu tablets contain paracetamol 500 mg, pseudoephedrine hydrochloride 30 mg and codeine phosphate 6 mg.
PRODUCT INFORMATION Codral* Original Cold & Flu Tablets Product description Codral* Original Cold & Flu tablets contain paracetamol 500 mg, pseudoephedrine hydrochloride 30 mg and codeine phosphate 6 mg.
Panadeine Tablet, Caplets and Rapid Soluble tablets PRODUCT INFORMATION
 Panadeine Tablet, Caplets and Rapid Soluble tablets PRODUCT INFORMATION DESCRIPTION Active Ingredients Paracetamol 500 mg Codeine Phosphate 8 mg Excipients Tablets - Starch - maize, talc - purified, stearic
Panadeine Tablet, Caplets and Rapid Soluble tablets PRODUCT INFORMATION DESCRIPTION Active Ingredients Paracetamol 500 mg Codeine Phosphate 8 mg Excipients Tablets - Starch - maize, talc - purified, stearic
NEW ZEALAND DATA SHEET. Each Paracetamol + Codeine contains 500 mg of paracetamol and 8 mg of codeine phosphate.
 NEW ZEALAND DATA SHEET PARACETAMOL + CODEINE 1. Product Name Paracetamol + Codeine, 500 mg + 8 mg, tablets. 2. Qualitative and Quantitative Composition Each Paracetamol + Codeine contains 500 mg of paracetamol
NEW ZEALAND DATA SHEET PARACETAMOL + CODEINE 1. Product Name Paracetamol + Codeine, 500 mg + 8 mg, tablets. 2. Qualitative and Quantitative Composition Each Paracetamol + Codeine contains 500 mg of paracetamol
Paracetamol, codeine phosphate hemihydrate and promethazine hydrochloride.
 AUSTRALIAN PRODUCT INFORMATION, PAINSTOP NIGHT-TIME PAIN RELIEVER (PARACETAMOL 120MG IN 5ML,CODEINE PHOSPHATE HEMIHYDRATE 5MG IN 5ML AND PROMETHAZINE HYDROCHLORIDE 6.5MG IN 5ML) 1. NAME OF THE MEDICINE
AUSTRALIAN PRODUCT INFORMATION, PAINSTOP NIGHT-TIME PAIN RELIEVER (PARACETAMOL 120MG IN 5ML,CODEINE PHOSPHATE HEMIHYDRATE 5MG IN 5ML AND PROMETHAZINE HYDROCHLORIDE 6.5MG IN 5ML) 1. NAME OF THE MEDICINE
PARACOD Tablets (Paracetamol + Codeine phosphate)
 Published on: 22 Sep 2014 PARACOD Tablets (Paracetamol + Codeine phosphate) Composition PARACOD Tablets Each effervescent tablet contains: Paracetamol IP...650 mg Codeine Phosphate IP... 30 mg Dosage Form/s
Published on: 22 Sep 2014 PARACOD Tablets (Paracetamol + Codeine phosphate) Composition PARACOD Tablets Each effervescent tablet contains: Paracetamol IP...650 mg Codeine Phosphate IP... 30 mg Dosage Form/s
PRODUCT INFORMATION. (RS)-N,N-Dimethyl-2-[(2-methylphenyl)phenylmethoxy]ethanamine dihydrogen 2-hydroxypropane-1,2,3-tricarboxylate
![PRODUCT INFORMATION. (RS)-N,N-Dimethyl-2-[(2-methylphenyl)phenylmethoxy]ethanamine dihydrogen 2-hydroxypropane-1,2,3-tricarboxylate PRODUCT INFORMATION. (RS)-N,N-Dimethyl-2-[(2-methylphenyl)phenylmethoxy]ethanamine dihydrogen 2-hydroxypropane-1,2,3-tricarboxylate](/thumbs/81/82665370.jpg) NORGESIC Orphenadrine citrate and paracetamol PRODUCT INFORMATION NAME OF THE MEDICINE Active ingredient: Chemical name: CAS number: 4682-36-4 Chemical structure: Orphenadrine citrate (RS)-N,N-Dimethyl-2-[(2-methylphenyl)phenylmethoxy]ethanamine
NORGESIC Orphenadrine citrate and paracetamol PRODUCT INFORMATION NAME OF THE MEDICINE Active ingredient: Chemical name: CAS number: 4682-36-4 Chemical structure: Orphenadrine citrate (RS)-N,N-Dimethyl-2-[(2-methylphenyl)phenylmethoxy]ethanamine
Summary of Product Characteristics
 1 NAME OF THE MEDICINAL PRODUCT Tipol 75mg Suppositories Summary of Product Characteristics 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each suppository contains 75mg of paracetamol For a full list of excipients,
1 NAME OF THE MEDICINAL PRODUCT Tipol 75mg Suppositories Summary of Product Characteristics 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each suppository contains 75mg of paracetamol For a full list of excipients,
Active ingredients: Paracetamol Codeine phosphate. Chemical names: N-(4-Hydroxyphenyl)acetamide 4,5a-epoxy-3-methoxy-17-methyl-7,8-
 Codapane Paracetamol / Codeine phosphate PRODUCT INFORMATION NAME OF THE MEDICINE Active ingredients: Paracetamol Codeine phosphate Chemical names: N-(4-Hydroxyphenyl)acetamide 4,5a-epoxy-3-methoxy-17-methyl-7,8-
Codapane Paracetamol / Codeine phosphate PRODUCT INFORMATION NAME OF THE MEDICINE Active ingredients: Paracetamol Codeine phosphate Chemical names: N-(4-Hydroxyphenyl)acetamide 4,5a-epoxy-3-methoxy-17-methyl-7,8-
PRODEINE paracetamol and codeine phosphate hemihydrate
 PRODEINE paracetamol and codeine phosphate hemihydrate Consumer Medicine Information (CMI) What is in this leaflet This leaflet answers some common questions about Prodeine. It does not, however, contain
PRODEINE paracetamol and codeine phosphate hemihydrate Consumer Medicine Information (CMI) What is in this leaflet This leaflet answers some common questions about Prodeine. It does not, however, contain
PANADOL COLD & FLU RELIEF ORIGINAL FORMULA is a white, capsule-shaped tablet with flat edges, one face marked with C&F, PANADOL on the other.
 Product description PANADOL COLD & FLU RELIEF ORIGINAL FORMULA is a white, capsule-shaped tablet with flat edges, one face marked with C&F, PANADOL on the other. Ingredients Active Ingredients: Paracetamol
Product description PANADOL COLD & FLU RELIEF ORIGINAL FORMULA is a white, capsule-shaped tablet with flat edges, one face marked with C&F, PANADOL on the other. Ingredients Active Ingredients: Paracetamol
PRODUCT INFORMATION PANADOL COLD & FLU MAX HOT LEMON POWDER NAME OF THE MEDICINE. Chemical structure: CAS 2 Registry Number: DESCRIPTION
 PRODUCT INFORMATION PANADOL COLD & FLU MAX HOT LEMON POWDER NAME OF THE MEDICINE Active ingredient: Paracetamol Chemical structure: CAS 2 Registry Number: 103-90-2 DESCRIPTION Paracetamol is a white, crystalline
PRODUCT INFORMATION PANADOL COLD & FLU MAX HOT LEMON POWDER NAME OF THE MEDICINE Active ingredient: Paracetamol Chemical structure: CAS 2 Registry Number: 103-90-2 DESCRIPTION Paracetamol is a white, crystalline
Your Pharmacy Paracetamol Plus, capsule shaped tablets (caplets)
 New Zealand Data Sheet Product Description Your Pharmacy Plus, capsule shaped tablets (caplets) 500 mg per tablet & Codeine Phosphate 10 mg per tablet Blister packs of white, capsule-shaped tablets Composition
New Zealand Data Sheet Product Description Your Pharmacy Plus, capsule shaped tablets (caplets) 500 mg per tablet & Codeine Phosphate 10 mg per tablet Blister packs of white, capsule-shaped tablets Composition
APO-PARACETAMOL/CODEINE 500/30
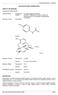 APO-PARACETAMOL/CODEINE 500/30 NAME OF THE MEDICINE Paracetamol/Codeine 500/30. Chemical Name: Paracetamol: N-(4-Hydroxyphenyl) acetamide Codeine: (5R, 6S)-7, 8-didehydro-4,5-epoxy-3-methoxy-Nmethylmorphinan-6-ol
APO-PARACETAMOL/CODEINE 500/30 NAME OF THE MEDICINE Paracetamol/Codeine 500/30. Chemical Name: Paracetamol: N-(4-Hydroxyphenyl) acetamide Codeine: (5R, 6S)-7, 8-didehydro-4,5-epoxy-3-methoxy-Nmethylmorphinan-6-ol
PRESCRIBING INFORMATION. N MERSYNDOL with codeine (acetaminophen 325 mg, codeine phosphate 8 mg and doxylamine succinate 5 mg)
 PRESCRIBING INFORMATION N MERSYNDOL with codeine (acetaminophen 325 mg, codeine phosphate 8 mg and doxylamine succinate 5 mg) Analgesic, Antihistamine sanofi-aventis Canada Inc. 2905 Place Louis R.-Renaud
PRESCRIBING INFORMATION N MERSYNDOL with codeine (acetaminophen 325 mg, codeine phosphate 8 mg and doxylamine succinate 5 mg) Analgesic, Antihistamine sanofi-aventis Canada Inc. 2905 Place Louis R.-Renaud
RAFEN PLUS Ibuprofen 200mg/Codeine phosphate 12.8mg tablets PRODUCT INFORMATION
 RAFEN PLUS Ibuprofen 200mg/Codeine phosphate 12.8mg tablets PRODUCT INFORMATION Composition RAFEN PLUS tablet is a white to off-white capsule-shaped, biconvex tablet, containing ibuprofen 200mg and codeine
RAFEN PLUS Ibuprofen 200mg/Codeine phosphate 12.8mg tablets PRODUCT INFORMATION Composition RAFEN PLUS tablet is a white to off-white capsule-shaped, biconvex tablet, containing ibuprofen 200mg and codeine
Farmadol. Paracetamol 10 mg/ml INFUSION SOLUTION
 Farmadol Paracetamol 10 mg/ml INFUSION SOLUTION Composition Each ml contains: Paracetamol 10 mg Pharmacology Pharmacodynamic properties The precise mechanism of the analgesic and antipyretic properties
Farmadol Paracetamol 10 mg/ml INFUSION SOLUTION Composition Each ml contains: Paracetamol 10 mg Pharmacology Pharmacodynamic properties The precise mechanism of the analgesic and antipyretic properties
Mersyndol Forte Paracetamol, codeine phosphate hemihydrate, doxylamine succinate
 Mersyndol Forte Paracetamol, codeine phosphate hemihydrate, doxylamine succinate Consumer Medicine Information What is in this leaflet This leaflet answers some common questions about Mersyndol Forte.
Mersyndol Forte Paracetamol, codeine phosphate hemihydrate, doxylamine succinate Consumer Medicine Information What is in this leaflet This leaflet answers some common questions about Mersyndol Forte.
SANDOMIGRAN (pizotifen malate)
 SANDOMIGRAN (pizotifen malate) S N CH 3 Pizotifen. COOH CH OH CH 2 COOH MALATE DESCRIPTION Pizotifen is a cycloheptathiophene derivative structurally related to cyproheptadine and the tricyclic antidepressants.
SANDOMIGRAN (pizotifen malate) S N CH 3 Pizotifen. COOH CH OH CH 2 COOH MALATE DESCRIPTION Pizotifen is a cycloheptathiophene derivative structurally related to cyproheptadine and the tricyclic antidepressants.
PRODUCT INFORMATION. Sudafed* Sinus Day + Night Relief Tablets
 PRODUCT INFORMATION Sudafed* Sinus Day + Night Relief Tablets Product description Sudafed* Sinus Day + Night Relief tablets contain two separate formulations: day tablets and night tablets. Each Sudafed*
PRODUCT INFORMATION Sudafed* Sinus Day + Night Relief Tablets Product description Sudafed* Sinus Day + Night Relief tablets contain two separate formulations: day tablets and night tablets. Each Sudafed*
PRODUCT INFORMATION. Sudafed* Sinus Day + Night Relief Tablets
 PRODUCT INFORMATION Sudafed* Sinus Day + Night Relief Tablets Name of the Medicine Paracetamol CAS 2 Registry Number: 103-90-2 Pseudoephedrine Hydrochloride CAS 2 Registry Number: 345-78-8 Triprolidine
PRODUCT INFORMATION Sudafed* Sinus Day + Night Relief Tablets Name of the Medicine Paracetamol CAS 2 Registry Number: 103-90-2 Pseudoephedrine Hydrochloride CAS 2 Registry Number: 345-78-8 Triprolidine
PRODUCT INFORMATION. Sudafed* Sinus + Allergy & Pain Relief Tablets
 PRODUCT INFORMATION Sudafed* Sinus + Allergy & Pain Relief Tablets Name of the Medicine Paracetamol CAS 2 Registry Number: 103-90-2 Pseudoephedrine Hydrochloride CAS 2 Registry Number: 345-78-8 Triprolidine
PRODUCT INFORMATION Sudafed* Sinus + Allergy & Pain Relief Tablets Name of the Medicine Paracetamol CAS 2 Registry Number: 103-90-2 Pseudoephedrine Hydrochloride CAS 2 Registry Number: 345-78-8 Triprolidine
DATA SHEET. PANADOL Mini Caps Capsule shaped tablet with a gelatin coating which is one half green and the other half white.
 PANADOL TABLETS PANADOL MINI CAPS DATA SHEET Proprietary (Trade) Name: PANADOL Active ingredient: Paracetamol (BP) 500 mg/tablet PRESENTATIONS White, film-coated tablet with bevelled edge, shallow convex,
PANADOL TABLETS PANADOL MINI CAPS DATA SHEET Proprietary (Trade) Name: PANADOL Active ingredient: Paracetamol (BP) 500 mg/tablet PRESENTATIONS White, film-coated tablet with bevelled edge, shallow convex,
PRODUCT INFORMATION. Ammonium chloride is an expectorant that has an irritant effect on mucous membranes.
 PRODUCT INFORMATION BENADRYL Original Oral Liquid (New Formula) Name of the Medicine Diphenhydramine hydrochloride Ammonium chloride The chemical name for diphenhydramine hydrochloride is 2-(diphenylmethoxy)-N,Ndimethylethanamine
PRODUCT INFORMATION BENADRYL Original Oral Liquid (New Formula) Name of the Medicine Diphenhydramine hydrochloride Ammonium chloride The chemical name for diphenhydramine hydrochloride is 2-(diphenylmethoxy)-N,Ndimethylethanamine
PANADOL SINUS DAY & NIGHT TABLETS PRODUCT INFORMATION
 Product description PANADOL SINUS DAY tablet is a white, film-coated capsule-shaped tablet with flat edges, front and back faces marked with DAY. PANADOL SINUS NIGHT tablet is a white, film-coated round
Product description PANADOL SINUS DAY tablet is a white, film-coated capsule-shaped tablet with flat edges, front and back faces marked with DAY. PANADOL SINUS NIGHT tablet is a white, film-coated round
PANADOL COLD & FLU MAX HOT LEMON Powder for Oral Solution DATA SHEET
 PANADOL COLD & FLU MAX HOT LEMON Powder for Oral Solution Paracetamol (BP) 1000mg/sachet DATA SHEET Presentation Pale yellow, free flowing heterogeneous powder with and odour of lemon Indications Fast,
PANADOL COLD & FLU MAX HOT LEMON Powder for Oral Solution Paracetamol (BP) 1000mg/sachet DATA SHEET Presentation Pale yellow, free flowing heterogeneous powder with and odour of lemon Indications Fast,
2. QUALITATIVE AND QUANTITATIVE COMPOSITION. Each capsule contains PARACETAMOL 500mg For a full list of excipients, see section 6.1.
 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT PARACETAMOL 500mg CAPSULES Boots Paracetamol 500mg Capsules 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each capsule contains PARACETAMOL
SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT PARACETAMOL 500mg CAPSULES Boots Paracetamol 500mg Capsules 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each capsule contains PARACETAMOL
NEW ZEALAND DATASHEET
 NEW ZEALAND DATASHEET COLDREX HOT REMEDY COLD & FLU HOT LEMON Powder for Oral Solution Paracetamol (BP) 1000mg/sachet Presentation Pale yellow, free flowing heterogeneous powder with and odour of lemon
NEW ZEALAND DATASHEET COLDREX HOT REMEDY COLD & FLU HOT LEMON Powder for Oral Solution Paracetamol (BP) 1000mg/sachet Presentation Pale yellow, free flowing heterogeneous powder with and odour of lemon
2 QUALITATIVE AND QUANTITATIVE COMPOSITION
 1. Name of Medicinal Product Co-Codamol 8/500 mg Capsules 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Paracetamol 500.0 mg Codeine Phosphate 8.0 mg For excipients, see section 6.1 3 PHARMACEUTICAL FORM
1. Name of Medicinal Product Co-Codamol 8/500 mg Capsules 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Paracetamol 500.0 mg Codeine Phosphate 8.0 mg For excipients, see section 6.1 3 PHARMACEUTICAL FORM
Metabolism Paracetamol is metabolised in the liver and excreted in the urine mainly as glucuronide and sulphate conjugates.
 FEBRAMOL Composition Febramol 150 Suppositories Each suppository contains Paracetamol 150 mg. Suppositories, Tablets & Syrup Febramol 300 Suppositories Each suppository contains Paracetamol 300 mg. Each
FEBRAMOL Composition Febramol 150 Suppositories Each suppository contains Paracetamol 150 mg. Suppositories, Tablets & Syrup Febramol 300 Suppositories Each suppository contains Paracetamol 300 mg. Each
NEW ZEALAND DATA SHEET
 1. PRODUCT NAME Sudomyl, Tablet, 60 mg 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Name and strength of the active substance Pseudoephedrine Hydrochloride 60mg Excipient(s) with known effect For the full
1. PRODUCT NAME Sudomyl, Tablet, 60 mg 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Name and strength of the active substance Pseudoephedrine Hydrochloride 60mg Excipient(s) with known effect For the full
PRODUCT INFORMATION. Sudafed* Sinus + Anti-inflammatory Pain Relief Caplets
 PRODUCT INFORMATION Sudafed* Sinus + Anti-inflammatory Pain Relief Caplets Product description Sudafed* Sinus + Anti-inflammatory Pain Relief caplets contain pseudoephedrine hydrochloride 30 mg and ibuprofen
PRODUCT INFORMATION Sudafed* Sinus + Anti-inflammatory Pain Relief Caplets Product description Sudafed* Sinus + Anti-inflammatory Pain Relief caplets contain pseudoephedrine hydrochloride 30 mg and ibuprofen
NEW ZEALAND DATA SHEET
 1. PRODUCT NAME PANADOL TABLETS, Paracetamol 500 mg, film coated tablet PANADOL MINI CAPS, Paracetamol 500 mg, coated tablet 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Active ingredient: Paracetamol (BP)
1. PRODUCT NAME PANADOL TABLETS, Paracetamol 500 mg, film coated tablet PANADOL MINI CAPS, Paracetamol 500 mg, coated tablet 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Active ingredient: Paracetamol (BP)
AUSTRALIAN PRODUCT INFORMATION PANADOL OPTIZORB TABLETS (PARACETAMOL) TABLETS PANADOL OPTIZORB CAPLETS (PARACETAMOL) TABLETS
 AUSTRALIAN PRODUCT INFORMATION PANADOL OPTIZORB TABLETS (PARACETAMOL) TABLETS PANADOL OPTIZORB CAPLETS (PARACETAMOL) TABLETS 1 NAME OF THE MEDICINE Paracetamol 2 QUALITATIVE AND QUANTITATIVE COMPOSITION
AUSTRALIAN PRODUCT INFORMATION PANADOL OPTIZORB TABLETS (PARACETAMOL) TABLETS PANADOL OPTIZORB CAPLETS (PARACETAMOL) TABLETS 1 NAME OF THE MEDICINE Paracetamol 2 QUALITATIVE AND QUANTITATIVE COMPOSITION
PRODUCT INFORMATION. SUDAFED Sinus 12 Hour Relief Tablets
 PRODUCT INFORMATION SUDAFED Sinus 12 Hour Relief Tablets NAME OF THE MEDICINE Pseudoephedrine Hydrochloride CAS 2 Registry Number: 345-78-8 DESCRIPTION SUDAFED Sinus 12 Hour Relief prolonged-release tablets
PRODUCT INFORMATION SUDAFED Sinus 12 Hour Relief Tablets NAME OF THE MEDICINE Pseudoephedrine Hydrochloride CAS 2 Registry Number: 345-78-8 DESCRIPTION SUDAFED Sinus 12 Hour Relief prolonged-release tablets
SCHEDULING STATUS: S0 For pack sizes of 24 tablets or less. For pack sizes of more than 24 tablets
 SCHEDULING STATUS: S0 For pack sizes of 24 tablets or less S1 For pack sizes of more than 24 tablets PROPRIETARY NAME: AND DOSAGE FORM PANADO MELTABS (Tablets) COMPOSITION: Each tablet contains 500 mg
SCHEDULING STATUS: S0 For pack sizes of 24 tablets or less S1 For pack sizes of more than 24 tablets PROPRIETARY NAME: AND DOSAGE FORM PANADO MELTABS (Tablets) COMPOSITION: Each tablet contains 500 mg
NEW ZEALAND DATA SHEET ACUPAN TM. 3. PHARMACEUTICAL FORM White, round, biconvex, film-coated tablets (7 mm diameter) engraved APN on one face.
 1. PRODUCT NAME ACUPAN 30 mg tablets 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each tablet contains nefopam hydrochloride 30 mg. For a full list of excipients, see section 6.1. 3. PHARMACEUTICAL FORM
1. PRODUCT NAME ACUPAN 30 mg tablets 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each tablet contains nefopam hydrochloride 30 mg. For a full list of excipients, see section 6.1. 3. PHARMACEUTICAL FORM
2 QUALITATIVE AND QUANTITATIVE COMPOSITION
 SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Paracetamol 80mg Suppositories 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each suppository contains 80mg Paracetamol For a full list of
SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Paracetamol 80mg Suppositories 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each suppository contains 80mg Paracetamol For a full list of
M0BCore Safety Profile. Active substance: Bromazepam Pharmaceutical form(s)/strength: Tablets 6 mg FR/H/PSUR/0066/001 Date of FAR:
 M0BCore Safety Profile Active substance: Bromazepam Pharmaceutical form(s)/strength: Tablets 6 mg P-RMS: FR/H/PSUR/0066/001 Date of FAR: 26.11.2013 4.3 Contraindications Bromazepam must not be administered
M0BCore Safety Profile Active substance: Bromazepam Pharmaceutical form(s)/strength: Tablets 6 mg P-RMS: FR/H/PSUR/0066/001 Date of FAR: 26.11.2013 4.3 Contraindications Bromazepam must not be administered
SUMMARY OF PRODUCT CHARACTERISTICS FOR BENZODIAZEPINES AS ANXIOLYTICS OR HYPNOTICS
 SUMMARY OF PRODUCT CHARACTERISTICS FOR BENZODIAZEPINES AS ANXIOLYTICS OR HYPNOTICS Guideline Title Summary of Product Characteristics for Benzodiazepines as Anxiolytics or Hypnotics Legislative basis Directive
SUMMARY OF PRODUCT CHARACTERISTICS FOR BENZODIAZEPINES AS ANXIOLYTICS OR HYPNOTICS Guideline Title Summary of Product Characteristics for Benzodiazepines as Anxiolytics or Hypnotics Legislative basis Directive
Chemists Own Strong Pain Extra Paracetamol and Codeine Phosphate
 Chemists Own Strong Pain Extra Paracetamol and Codeine Phosphate Consumer Medicine Information(CMI) Please read this information before you start taking this medicine. What is in this CMI? This CMI answers
Chemists Own Strong Pain Extra Paracetamol and Codeine Phosphate Consumer Medicine Information(CMI) Please read this information before you start taking this medicine. What is in this CMI? This CMI answers
NEW ZEALAND DATA SHEET
 1. PRODUCT NAME Codeine Phosphate Tablets (PSM) 15 mg, 30 mg & 60 mg tablets 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Name and strength of the active substance Codeine Phosphate Hemihydrate BP 15 mg
1. PRODUCT NAME Codeine Phosphate Tablets (PSM) 15 mg, 30 mg & 60 mg tablets 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Name and strength of the active substance Codeine Phosphate Hemihydrate BP 15 mg
PANADOL EXTRA PRODUCT INFORMATION
 PANADOL EXTRA PRODUCT INFORMATION DESCRIPTION Active Ingredients: Paracetamol 500 mg and Caffeine 65mg per tablet. Excipients: Maize starch Starch - Pregelatinised maize Povidone Potassium sorbate Purified
PANADOL EXTRA PRODUCT INFORMATION DESCRIPTION Active Ingredients: Paracetamol 500 mg and Caffeine 65mg per tablet. Excipients: Maize starch Starch - Pregelatinised maize Povidone Potassium sorbate Purified
CORE SAFETY PROFILE OXYCODONE HYDROCHLORIDE NL/H/PSUR/0054/ January 2013
 CORE SAFETY PROFILE OXYCODONE HYDROCHLORIDE NL/H/PSUR/0054/001 16 January 2013 1 4.2 Posology and method of administration (safety aspects only) Posology Elderly patients For oral preparations A dose adjustment
CORE SAFETY PROFILE OXYCODONE HYDROCHLORIDE NL/H/PSUR/0054/001 16 January 2013 1 4.2 Posology and method of administration (safety aspects only) Posology Elderly patients For oral preparations A dose adjustment
Immodium / loprarmide
 Immodium / loprarmide IMODIUM (loperamide hydrochloride) is indicated for the control and symptomatic relief of acute nonspecific diarrhea and of chronic diarrhea associated with inflammatory bowel disease.
Immodium / loprarmide IMODIUM (loperamide hydrochloride) is indicated for the control and symptomatic relief of acute nonspecific diarrhea and of chronic diarrhea associated with inflammatory bowel disease.
Summary of Product Characteristics
 1 NAME OF THE MEDICINAL PRODUCT Maxilief Effervescent Tablets Paracetamol 500mg Codeine Phosphate Hemihydrate 8mg Caffeine 30mg Summary of Product Characteristics 2 QUALITATIVE AND QUANTITATIVE COMPOSITION
1 NAME OF THE MEDICINAL PRODUCT Maxilief Effervescent Tablets Paracetamol 500mg Codeine Phosphate Hemihydrate 8mg Caffeine 30mg Summary of Product Characteristics 2 QUALITATIVE AND QUANTITATIVE COMPOSITION
Prodeine Forte Tablets Paracetamol & Codeine Phosphate Hemihydrate
 Prodeine Forte Tablets Paracetamol & Codeine Phosphate Hemihydrate CONSUMER MEDICINE INFORMATION What is in this leaflet This leaflet answers some common questions about Prodeine Forte tablets. It does
Prodeine Forte Tablets Paracetamol & Codeine Phosphate Hemihydrate CONSUMER MEDICINE INFORMATION What is in this leaflet This leaflet answers some common questions about Prodeine Forte tablets. It does
PRODUCT INFORMATION LOFENOXAL
 PRODUCT INFORMATION LOFENOXAL Diphenoxylate hydrochloride 2.5 mg and Atropine sulfate 25 microgram Name of the medicines Diphenoxylate Hydrochloride: C30H32N2O2, HCl M. W. = 489.1 CAS registry no.: 3810-80-8
PRODUCT INFORMATION LOFENOXAL Diphenoxylate hydrochloride 2.5 mg and Atropine sulfate 25 microgram Name of the medicines Diphenoxylate Hydrochloride: C30H32N2O2, HCl M. W. = 489.1 CAS registry no.: 3810-80-8
Maxilief Effervescent Tablets
 Package Leaflet: Information for the user Maxilief Effervescent Tablets Paracetamol Codeine Phosphate Hemihydrate Caffeine 500mg 8mg 30mg Read all of this leaflet carefully before you start taking this
Package Leaflet: Information for the user Maxilief Effervescent Tablets Paracetamol Codeine Phosphate Hemihydrate Caffeine 500mg 8mg 30mg Read all of this leaflet carefully before you start taking this
PRODUCT INFORMATION PANADOL TABLETS PANADOL MINI CAPS PANADOL SUPPOSITORIES
 PRODUCT INFORMATION PANADOL TABLETS PANADOL MINI CAPS PANADOL SUPPOSITORIES NAME OF THE MEDICINE Active ingredients Chemical structure CAS Registry Number Paracetamol 103-90-2 DESCRIPTION Paracetamol is
PRODUCT INFORMATION PANADOL TABLETS PANADOL MINI CAPS PANADOL SUPPOSITORIES NAME OF THE MEDICINE Active ingredients Chemical structure CAS Registry Number Paracetamol 103-90-2 DESCRIPTION Paracetamol is
PANADOL OSTEO PRODUCT INFORMATION
 PANADOL OSTEO PRODUCT INFORMATION NAME OF THE MEDICINE Active ingredients Chemical structure CAS Registry Number Paracetamol 103-90-2 DESCRIPTION White to off-white film coated capsule shaped tablets with
PANADOL OSTEO PRODUCT INFORMATION NAME OF THE MEDICINE Active ingredients Chemical structure CAS Registry Number Paracetamol 103-90-2 DESCRIPTION White to off-white film coated capsule shaped tablets with
Each 5ml of Sinarest LP New Syrup contains: Phenylephrine
 Composition: Each 5ml of Sinarest LP New Syrup contains: Paracetamol Phenylephrine Levocetrizine 250mg 5mg 1.25mg Pharmacokinetic properties: Paracetamol is readily absorbed from the gastrointestinal tract
Composition: Each 5ml of Sinarest LP New Syrup contains: Paracetamol Phenylephrine Levocetrizine 250mg 5mg 1.25mg Pharmacokinetic properties: Paracetamol is readily absorbed from the gastrointestinal tract
Core Safety Profile. Pharmaceutical form(s)/strength: Tablets 5 mg and 10 mg BE/H/PSUR/0002/002 Date of FAR:
 Core Safety Profile Active substance: Clotiazepam Pharmaceutical form(s)/strength: Tablets 5 mg and 10 mg P-RMS: BE/H/PSUR/0002/002 Date of FAR: 16.06.2011 4.3 Contraindications is contraindicated
Core Safety Profile Active substance: Clotiazepam Pharmaceutical form(s)/strength: Tablets 5 mg and 10 mg P-RMS: BE/H/PSUR/0002/002 Date of FAR: 16.06.2011 4.3 Contraindications is contraindicated
PRODUCT INFORMATION. Active ingredients Chemical structure CAS Registry Number. Paracetamol
 PRODUCT INFORMATION PANADOL SINUS PAIN & CONGESTION RELIEF CAPLETS PANADOL COLD & FLU PLUS DECONGESTANT CAPLETS NAME OF THE MEDICINE Active ingredients Chemical structure CAS Registry Number Paracetamol
PRODUCT INFORMATION PANADOL SINUS PAIN & CONGESTION RELIEF CAPLETS PANADOL COLD & FLU PLUS DECONGESTANT CAPLETS NAME OF THE MEDICINE Active ingredients Chemical structure CAS Registry Number Paracetamol
DATA SHEET. Paracetamol (BP) 500 mg and Phenylephrine Hydrochloride (BP) 5 mg
 COLDREX PE PHENYLEPHRINE SINUS COLDREX PE PHENYLEPHRINE CONGESTION CLEAR PANADOL SINUS PAIN & CONGESTION RELIEF PANADOL COLD & FLU MAX + DECONGESTANT DATA SHEET Paracetamol (BP) 500 mg and Phenylephrine
COLDREX PE PHENYLEPHRINE SINUS COLDREX PE PHENYLEPHRINE CONGESTION CLEAR PANADOL SINUS PAIN & CONGESTION RELIEF PANADOL COLD & FLU MAX + DECONGESTANT DATA SHEET Paracetamol (BP) 500 mg and Phenylephrine
European PSUR Work Sharing Project CORE SAFETY PROFILE. Lendormin, 0.25mg, tablets Brotizolam
 European PSUR Work Sharing Project CORE SAFETY PROFILE Lendormin, 0.25mg, tablets Brotizolam 4.2 Posology and method of administration Unless otherwise prescribed by the physician, the following dosages
European PSUR Work Sharing Project CORE SAFETY PROFILE Lendormin, 0.25mg, tablets Brotizolam 4.2 Posology and method of administration Unless otherwise prescribed by the physician, the following dosages
TRAPADOL INJECTION FOR I.V./I.M. USE ONLY
 TRAPADOL INJECTION FOR I.V./I.M. USE ONLY Composition : Each 2ml. contains : Tramadol Hydrochloride I.P. Water for injection I.P. 100mg. q.s. CLINICAL PHARMACOLOGY : Pharmacodynamics Tramadol is a centrally
TRAPADOL INJECTION FOR I.V./I.M. USE ONLY Composition : Each 2ml. contains : Tramadol Hydrochloride I.P. Water for injection I.P. 100mg. q.s. CLINICAL PHARMACOLOGY : Pharmacodynamics Tramadol is a centrally
Patient leaflet: Information for the user. Paracetamol Galpharm 500mg tablets paracetamol
 Patient leaflet: Information for the user Paracetamol Galpharm 500mg tablets paracetamol [For medicines available only on prescription:] Read all of this leaflet carefully before you start taking this
Patient leaflet: Information for the user Paracetamol Galpharm 500mg tablets paracetamol [For medicines available only on prescription:] Read all of this leaflet carefully before you start taking this
4.4 Special warnings and precautions for use
 SUMMARY OF PRODUCT CHARACTERISTICS 4.3 Contraindications Durogesic is contraindicated in patients with known hypersensitivity to fentanyl or to the excipients present in the patch. Acute or postoperative
SUMMARY OF PRODUCT CHARACTERISTICS 4.3 Contraindications Durogesic is contraindicated in patients with known hypersensitivity to fentanyl or to the excipients present in the patch. Acute or postoperative
Active ingredients: Metoclopramide Hydrochloride mg Equivalent to metoclopramide hydrochloride anhydrous mg
 Name Primperan 10 mg / 2 ml Metoclopramide hydrochloride anhydrous Solution for I.M. or I.V. injection (Ampoules) Composition Each 2 ml ampoule contains: Active ingredients: Metoclopramide Hydrochloride.
Name Primperan 10 mg / 2 ml Metoclopramide hydrochloride anhydrous Solution for I.M. or I.V. injection (Ampoules) Composition Each 2 ml ampoule contains: Active ingredients: Metoclopramide Hydrochloride.
PRODUCT INFORMATION. AKINETON (biperiden hydrochloride 2 mg tablets)
 PRODUCT INFORMATION AKINETON (biperiden hydrochloride 2 mg tablets) NAME OF THE MEDICINE Non-proprietary name: biperiden hydrochloride Structural formula (biperiden): Chemical name (biperiden): α-5-norbornen-2-yl-α-phenyl-1-piperidine
PRODUCT INFORMATION AKINETON (biperiden hydrochloride 2 mg tablets) NAME OF THE MEDICINE Non-proprietary name: biperiden hydrochloride Structural formula (biperiden): Chemical name (biperiden): α-5-norbornen-2-yl-α-phenyl-1-piperidine
LEAFLET: INFORMATION FOR THE USER. Freshalgin 500th mg tablets Metamizole sodium (M etamizol sodium)
 LEAFLET: INFORMATION FOR THE USER Freshalgin 500th mg tablets Metamizole sodium (M etamizol sodium) Read this leaflet carefully before you start taking this medicine. This medicine is available without
LEAFLET: INFORMATION FOR THE USER Freshalgin 500th mg tablets Metamizole sodium (M etamizol sodium) Read this leaflet carefully before you start taking this medicine. This medicine is available without
PRODUCT INFORMATION. Benadryl* for the Family Nightime Oral Liquid
 Product description PRODUCT INFORMATION Benadryl* for the Family Nightime Oral Liquid Each 5 ml of Benadryl* Nightime oral liquid contains dextromethorphan hydrobromide 10 mg and diphenhydramine hydrochloride
Product description PRODUCT INFORMATION Benadryl* for the Family Nightime Oral Liquid Each 5 ml of Benadryl* Nightime oral liquid contains dextromethorphan hydrobromide 10 mg and diphenhydramine hydrochloride
Elements for a Public Summary
 VI.2 Elements for a Public Summary VI.2.1 Overview of disease epidemiology Sudden pain is an unpleasant sensation resulting from physical hurt caused by injuries of the body, illness or emotional illness.
VI.2 Elements for a Public Summary VI.2.1 Overview of disease epidemiology Sudden pain is an unpleasant sensation resulting from physical hurt caused by injuries of the body, illness or emotional illness.
Now available. A maintenance dose of SUBOXONE mg once daily is clinically effective for most patients*1. Once-daily dosing in a single tablet
 Now available SUBOXONE Once-daily dosing1 12 mg and 16 mg tablets A maintenance dose of SUBOXONE 12-16 mg once daily is clinically effective for most patients*1 Effective maintenance dosing with SUBOXONE
Now available SUBOXONE Once-daily dosing1 12 mg and 16 mg tablets A maintenance dose of SUBOXONE 12-16 mg once daily is clinically effective for most patients*1 Effective maintenance dosing with SUBOXONE
PRODUCT INFORMATION ANAGRAINE. Each ANAGRAINE tablet contains 5 mg metoclopramide and 500 mg paracetamol.
 PRODUCT INFORMATION ANAGRAINE COMPOSITION Each ANAGRAINE tablet contains 5 mg metoclopramide and 500 mg paracetamol. ACTIONS Metoclopramide stimulates gastrointestinal tract motility and accelerates gastric
PRODUCT INFORMATION ANAGRAINE COMPOSITION Each ANAGRAINE tablet contains 5 mg metoclopramide and 500 mg paracetamol. ACTIONS Metoclopramide stimulates gastrointestinal tract motility and accelerates gastric
PRODUCT INFORMATION. Active ingredients Chemical structure CAS Registry Number. Paracetamol
 PRODUCT INFORMATION PANADOL COLD & FLU MAX + DECONGESTANT HOT LEMON POWDER NAME OF THE MEDICINE Active ingredients Chemical structure CAS Registry Number Paracetamol 103-90-2 Phenylephrine Hydrochloride
PRODUCT INFORMATION PANADOL COLD & FLU MAX + DECONGESTANT HOT LEMON POWDER NAME OF THE MEDICINE Active ingredients Chemical structure CAS Registry Number Paracetamol 103-90-2 Phenylephrine Hydrochloride
Package leaflet: Information for the patient. Morphine Unimedic 1 mg/ml solution for injection morphine hydrochloride trihydrate
 Package leaflet: Information for the patient Morphine Unimedic 1 mg/ml solution for injection morphine hydrochloride trihydrate Read all of this leaflet carefully before you start using this medicine because
Package leaflet: Information for the patient Morphine Unimedic 1 mg/ml solution for injection morphine hydrochloride trihydrate Read all of this leaflet carefully before you start using this medicine because
CHILDREN S PANADOL COLOURFREE SUSPENSION PANADOL SUPPOSITORIES 125 MG PANADOL SUPPOSITORIES 250 MG DATA SHEET. Proprietary (Trade) Name: PANADOL
 CHILDREN S PANADOL COLOURFREE SUSPENSION PANADOL SUPPOSITORIES 125 MG PANADOL SUPPOSITORIES 250 MG DATA SHEET Proprietary (Trade) Name: PANADOL Active ingredient: Paracetamol (BP) 120 mg/5ml (Children
CHILDREN S PANADOL COLOURFREE SUSPENSION PANADOL SUPPOSITORIES 125 MG PANADOL SUPPOSITORIES 250 MG DATA SHEET Proprietary (Trade) Name: PANADOL Active ingredient: Paracetamol (BP) 120 mg/5ml (Children
SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT 2. QUALITATIVE AND QUANTITATIVE COMPOSITION
 SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT PARACETAMOL 500mg CAPSULES 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Paracetamol 500mg For the full list of excipients see section6.1.
SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT PARACETAMOL 500mg CAPSULES 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Paracetamol 500mg For the full list of excipients see section6.1.
Summary of Product Characteristics
 1 NAME OF THE MEDICINAL PRODUCT Panadol Baby 120 mg/5 ml, Oral Suspension. Summary of Product Characteristics 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each 5 ml spoonful of suspension contains paracetamol
1 NAME OF THE MEDICINAL PRODUCT Panadol Baby 120 mg/5 ml, Oral Suspension. Summary of Product Characteristics 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each 5 ml spoonful of suspension contains paracetamol
PRODUCT INFORMATION RESONIUM A. Na m
 PRODUCT INFORMATION RESONIUM A NAME OF THE MEDICINE Non-proprietary Name Sodium polystyrene sulfonate Chemical Structure CH - 2 CH SO 3 Na + n CAS Number 28210-41-5 [9003-59-2] CH 2 CH SO - 3 m DESCRIPTION
PRODUCT INFORMATION RESONIUM A NAME OF THE MEDICINE Non-proprietary Name Sodium polystyrene sulfonate Chemical Structure CH - 2 CH SO 3 Na + n CAS Number 28210-41-5 [9003-59-2] CH 2 CH SO - 3 m DESCRIPTION
Summary of Product Characteristics
 1 NAME OF THE MEDICINAL PRODUCT Tipol 250mg Suppositories Summary of Product Characteristics 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each suppository contains 250mg of paracetamol For a full list of
1 NAME OF THE MEDICINAL PRODUCT Tipol 250mg Suppositories Summary of Product Characteristics 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each suppository contains 250mg of paracetamol For a full list of
Cetirizine Proposed Core Safety Profile
 Cetirizine Proposed Core Safety Profile Posology and method of administration Elderly subjects: data do not suggest that the dose needs to be reduced in elderly subjects provided that the renal function
Cetirizine Proposed Core Safety Profile Posology and method of administration Elderly subjects: data do not suggest that the dose needs to be reduced in elderly subjects provided that the renal function
Annex III. Amendments to relevant sections of the product information
 Annex III Amendments to relevant sections of the product information Note: These amendments to the relevant sections of the Summary of Product Characteristics and Package Leaflet are the outcome of the
Annex III Amendments to relevant sections of the product information Note: These amendments to the relevant sections of the Summary of Product Characteristics and Package Leaflet are the outcome of the
PACKAGE LEAFLET: INFORMATION FOR THE USER. Meptid 100 mg/ml Solution for Injection. Meptazinol Hydrochloride
 PACKAGE LEAFLET: INFORMATION FOR THE USER Meptid 100 mg/ml Solution for Injection Meptazinol Hydrochloride Read all of this leaflet carefully before you start taking this medicine. - Keep this leaflet.
PACKAGE LEAFLET: INFORMATION FOR THE USER Meptid 100 mg/ml Solution for Injection Meptazinol Hydrochloride Read all of this leaflet carefully before you start taking this medicine. - Keep this leaflet.
RE: Shortage of DBL MORPHINE SULFATE 30mg/1mL injection BP ampoule and alternative supply arrangement under Section 19A of the Therapeutic Goods Act
 22 January 2019 RE: Shortage of DBL MORPHINE SULFATE 30mg/1mL injection BP ampoule and alternative supply arrangement under Section 19A of the Therapeutic Goods Act Dear Healthcare Professional, This notification
22 January 2019 RE: Shortage of DBL MORPHINE SULFATE 30mg/1mL injection BP ampoule and alternative supply arrangement under Section 19A of the Therapeutic Goods Act Dear Healthcare Professional, This notification
PARACIP Injection (Paracetamol )
 Published on: 22 Sep 2014 PARACIP Injection (Paracetamol ) Black Box Warning: For Medication Error And Hepatotoxicity Vigilance is advised when prescribing and administering I.V. paracetamol 10 mg/ml solution
Published on: 22 Sep 2014 PARACIP Injection (Paracetamol ) Black Box Warning: For Medication Error And Hepatotoxicity Vigilance is advised when prescribing and administering I.V. paracetamol 10 mg/ml solution
PACKAGE INSERT TEMPLATE FOR SALBUTAMOL TABLET & SALBUTAMOL SYRUP
 PACKAGE INSERT TEMPLATE FOR SALBUTAMOL TABLET & SALBUTAMOL SYRUP Brand or Product Name [Product name] Tablet 2mg [Product name] Tablet 4mg [Product name] Syrup 2mg/5ml Name and Strength of Active Substance(s)
PACKAGE INSERT TEMPLATE FOR SALBUTAMOL TABLET & SALBUTAMOL SYRUP Brand or Product Name [Product name] Tablet 2mg [Product name] Tablet 4mg [Product name] Syrup 2mg/5ml Name and Strength of Active Substance(s)
PACKAGE LEAFLET: INFORMATION FOR THE USER. MEPHENON 10mg/1ml, solution for injection MEPHENON 5mg, tablets. Methadone hydrochloride
 PACKAGE LEAFLET: INFORMATION FOR THE USER MEPHENON 10mg/1ml, solution for injection MEPHENON 5mg, tablets Methadone hydrochloride Read all of this leaflet carefully before you start using this medicine
PACKAGE LEAFLET: INFORMATION FOR THE USER MEPHENON 10mg/1ml, solution for injection MEPHENON 5mg, tablets Methadone hydrochloride Read all of this leaflet carefully before you start using this medicine
Package leaflet: Information for the user. Co-Codamol 30mg/500mg Capsules (Codeine Phosphate Hemihydrate and Paracetamol)
 Package leaflet: Information for the user Co-Codamol 30mg/500mg Capsules (Codeine Phosphate Hemihydrate and Paracetamol) Read all of this leaflet carefully before you start taking this medicine because
Package leaflet: Information for the user Co-Codamol 30mg/500mg Capsules (Codeine Phosphate Hemihydrate and Paracetamol) Read all of this leaflet carefully before you start taking this medicine because
NEW ZEALAND DATA SHEET
 NEW ZEALAND DATA SHEET VERGO 16 1. Product Name Vergo 16, 16 mg, tablet. 2. Qualitative and Quantitative Composition Each tablet contains 16 mg of betahistine dihydrochloride. For the full list of excipients,
NEW ZEALAND DATA SHEET VERGO 16 1. Product Name Vergo 16, 16 mg, tablet. 2. Qualitative and Quantitative Composition Each tablet contains 16 mg of betahistine dihydrochloride. For the full list of excipients,
M0BCore Safety Profile
 M0BCore Safety Profile Active substance: Aciclovir Pharmaceutical form(s)/strength: Tablets 200, 400 or 800 mg Dispersible tablets 200, 400 or 800 mg Oral suspensions 200 mg or 400 mg per 5 ml. Freeze
M0BCore Safety Profile Active substance: Aciclovir Pharmaceutical form(s)/strength: Tablets 200, 400 or 800 mg Dispersible tablets 200, 400 or 800 mg Oral suspensions 200 mg or 400 mg per 5 ml. Freeze
Fact Sheet. Zohydro ER (hydrocodone bitartrate) Extended-Release Capsules, CII
 Zohydro ER (hydrocodone bitartrate) Extended-Release Capsules, CII Fact Sheet Zohydro ER (hydrocodone bitartrate) Extended-Release Capsule, CII, is a long-acting (extendedrelease) type of pain medication
Zohydro ER (hydrocodone bitartrate) Extended-Release Capsules, CII Fact Sheet Zohydro ER (hydrocodone bitartrate) Extended-Release Capsule, CII, is a long-acting (extendedrelease) type of pain medication
SUMMARY OF PRODUCT CHARACTERISTICS 2 QUALITATIVE AND QUANTITATIVE COMPOSITION
 SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Co-Codamol 8mg/500mg Tablets Boots Paracetamol and Codeine Tablets 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Paracetamol 500mg Codeine
SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Co-Codamol 8mg/500mg Tablets Boots Paracetamol and Codeine Tablets 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Paracetamol 500mg Codeine
Dilaudid, a narcotic analgesic, is prescribed for the relief of moderate to severe pain such as that due to:
 DrugFAQs Dilaudid Generic name: Hydromorphone hydrochloride Brand names: Dilaudid Why is Dilaudid prescribed? Dilaudid, a narcotic analgesic, is prescribed for the relief of moderate to severe pain such
DrugFAQs Dilaudid Generic name: Hydromorphone hydrochloride Brand names: Dilaudid Why is Dilaudid prescribed? Dilaudid, a narcotic analgesic, is prescribed for the relief of moderate to severe pain such
