Laboratory Service Report
|
|
|
- Bennett Golden
- 5 years ago
- Views:
Transcription
1 Specimen Type Peripheral blood CR PDF Report available at: Indication for Test DS CR Pathogenic utations Detected CR 1. JAK2: c.1849g>t;p.val617phe No other pathogenic mutations were detected in the other genes tested on the panel at the reportable limit of assay detection. See below for Variants of Unknown Significance and Additional Notes. Please see the section of "Panel Gene List" below for the complete list of genes tested. Interpretation CR 1. JAK2: c.1849g>t;p.val617phe Normal gene/protein function: The JAK2 gene encodes the Janus kinase 2 (JAK2) protein, which belongs to the Janus family of non-receptor tyrosine kinases (JAK kinases). JAK proteins are pivotal for promoting the signal transduction events of multiple hematopoietic cytokines and growth factors (Ward et al, 2000, ). utation effect: The p.val617phe (V617F) mutation has been shown to be a gain of function resulting in constitutive activation of the JAK2 protein and cytokine-independent growth of hematopoietic cells (Levine et al, 2007, ). Disease associations: The JAK2 V617F mutation is implicated in myeloid neoplasms, especially the BCR-ABL1 negative classical myeloproliferative neoplasms (PN), in which mutations have been reported in at least 95% of polycythemia vera (PV) and 50-60% of essential thrombocythemia (ET) and primary myelofibrosis (PF) patients (Baxter et al, 2005, ; Levine et al, 2005, ; Kralovics et al, 2005, ; James et al, 2005, ). JAK2 V617F has also been described in association in the majority of cases of the rare myeloproliferative/myelodysplastic neoplasm (PN/DS) refractory anemia with ring sideroblasts and thrombocytosis (RARS-T) (alcovati et al, 2009, ), and only infrequently in other myeloid neoplasms, including myelodysplastic syndromes (DS) and other types of PN/DS (e.g. chronic myelomonocytic leukemia (CL), atypical chronic myeloid leukemia (acl)) (Steensma et al, 2005, ; Wang et al, 2014, ). ***Performing Site Legend on Last Page of Report*** Page 1 of 5 >> Continued on Next Page >> * Report times for ayo performed tests are CST/CDT
2 Therapeutic implications: The JAK1/2 inhibitor Ruxolitinib has been approved for treatment in PF and demonstrated efficacy in improving constitutional symptoms and reducing spleen size (Verstovsek et al, 2012, ; Harrison et al 2012, ). In a phase 2 clinical trial, it demonstrated modest antileukemic activity as a single agent in patients with refractory post-pn AL (Eghtedar et al, 2012, ). Other JAK2 inhibitors, PI3K inhibitors and HDAC inhibitors are also being investigated in PN (Geyer et al, 2014, ). Clinical Trials CR Information regarding possible clinical trials for this patient can be found at the following sites: 1). ClinicalTrials.gov: 2). ayo Clinic: 3). National Cancer Institute: 4). olecular atch: Variants of Unknown Significance None CR Additional Notes None CR ethod Summary CR DNA is extracted from the peripheral blood or bone marrow sample and following library preparation by hybrid capture, subjected to next generation sequencing (NGS) with post-sequencing analysis of tumor-associated mutations. Because some regions in the target panel are not consistently resolved by NGS, additional separate clinical laboratory assays may also be used to complete the overall analysis including: CALR (CALR), CEBPA (CEBPA), CSF3R (CSF3R), FLT3 (FLT), JAK2 V617F utation Detection (JAK2B, JAK2, or JAK2V), JAK2 Exon 12 utation Detection (JAKXB or JAKX), KIT (KITB, KITB, KITAS, or KITE), PL (PLB or PL), YD88 (YD88), NP1 (NP1), and TP53 (P53CA). Performance characteristics of NGS panel: Single base substitutions: accuracy >99%; reproducibility 100% (intra- and interassay); sensitivity 5-10% variant allele fraction with a minimum depth coverage of 250X. Insertion/deletion events: accuracy >99%; reproducibility ***Performing Site Legend on Last Page of Report*** Page 2 of 5 >> Continued on Next Page >> * Report times for ayo performed tests are CST/CDT
3 100% (intra- and interassay); sensitivity 5-10% variant allele fraction with a minimum depth coverage of 250X. This test was developed and its performance characteristics determined by ayo Clinic in a manner consistent with CLIA requirements. This test has not been cleared or approved by the U.S. Food and Drug Administration. Disclaimer CR CLINICAL DISCLAIER utation calls detected between 5-10% variant allele fractions (VAF) may indicate low-level (i.e. subclonal) tumor populations, although the clinical significance of these findings may not be clear. Some apparent mutations classified as VUS may represent rare or low frequency polymorphisms. A low incidence of gene mutations associated with myeloid neoplasms can also be detected in hematopoietic cells with advancing age in some individuals without evidence of a hematologic malignancy ("clonal hematopoiesis") and these alterations may not be clearly distinguishable from tumor-associated mutations (PID ; ; ). Prior treatment for hematologic malignancy could affect the results obtained in this assay. In particular, prior allogeneic hematopoietic stem cell transplant (HSCT) may cause difficulties in resolving somatic or polymorphic alterations, or in assigning variant calls correctly to donor and recipient fractions, if pertinent clinical or laboratory information (e.g. chimerism engraftment status) is not provided. Correlation with clinical, histopathologic and additional laboratory findings is required for final interpretation of these results. This assay does not distinguish between somatic and germ line alterations in analyzed gene regions, particularly with VAF near 50% or 100%. If nucleotide alterations in genes associated with germ line mutation syndromes are present and there is also a strong clinical suspicion or family history of malignant disease predisposition, additional genetic testing and appropriate counseling may be indicated. This report interpretation is based on current medical and scientific literature, but clinical significance may not be completely established for all reported target gene abnormalities identified. The final interpretation of results for clinical management of the patient is the responsibility of the managing physician. TECHNICAL DISCLAIER ***Performing Site Legend on Last Page of Report*** Page 3 of 5 >> Continued on Next Page >> * Report times for ayo performed tests are CST/CDT
4 The depth of sequencing coverage may be variable for some target regions, but assay performance below the minimum acceptable criteria, or for failed regions are noted. Analysis of rare (low allele frequency) polymorphisms may be problematic in some cases. A low tumor cell percentage in the sample may affect the true mutation VAF and/or sensitivity. Suboptimal- performing regions (i.e. less than the expected minimum depth of coverage) may affect analytic sensitivity for detecting lower level mutations. This is a qualitative test. The variant read fractions are provided for information only and represent a relative proportion of mutated alleles, but do not indicate a measure of analytical sensitivity for the given genes; assay sensitivity is as stated in the method summary. Some genetic or genomic alterations, such as large insertion/deletion (indel) events, copy number alterations (CNA) and gene translocation events are not detected by this assay. OncoHeme Panel Gene list CR ASXL1 (N_ ) exons 11-14, BCOR (N_ ) exons 5-, BRAF (N_ ) exon 15, CALR (N_ ) exon 9, CBL (N_ ) exon 8, intron 8, and exon 9, CEBPA (N_ ) exon 1, CSF3R (N_ ) exons 14 and 17, DNT3A (N_ ) exons 8-23, ETV6 (N_ ) exons 3-8, EZH2 (N_ ) exons 3-21, FLT3 (N_ ) exons 14-20, GATA1 (N_ ) exons 2 and 4, GATA2 (N_ ) exons 4-8, IDH1 (N_ ) exon 4, IDH2 (N_0028.3) exon 4, JAK2 (N_ ) exons 12-, KIT (N_ ) exons 8-11 and 17, KRAS (N_ ) exons 2-3, PL (N_ ) exons 10-11, YD88 (N_ ) exon 5, NOTCH1 (N_ ) exons 26, 27, and 34, NP1 (N_ ) exons 9, 11, and 12, NRAS (N_ ) exons 2 and 3, PHF6 (N_ ) exons 2-10, PTPN11 (N_ ) exons 3-4 and 12-13, RUNX1 (N_ ) exons 4-10, SETBP1 (N_ ) partial exon 6; amino acids , SF3B1 (N_ ) exons 13-, SRSF2 (N_0030.4) exons 1 and 2, TERT (N_ ) exons 2-, TET2 (N_ ) exons 3-11, TP53 (N_ ) exons 4-9, U2AF1 (N_ ) exons 2, 7, and 9, WT1 (N_ Aas.3) exons 1-11, and ZRSR2 (N_ ) exons For some genes, the transcript IDs used in this analysis may not be the same as in other cancer mutation databases, such as COSIC ( Reviewed By: CR Signing Pathologist: elissa Tricker-Klar ***Performing Site Legend on Last Page of Report*** Page 4 of 5 >> Continued on Next Page >> * Report times for ayo performed tests are CST/CDT
5 RECEIVED: 10/20/20 14:52 REPORTED: 10/20/20 15:03 * Performing Site: ayo Clinic Laboratories - Rochester ain Campus CR 200 First St SW Rochester, N Lab Director: William G. orice, II,.D., Ph.D. Page 5 of 5 ** End of Report ** * Report times for ayo performed tests are CST/CDT
6 PATIENT NAE TESTINGRNV, REPORT PATIENT ID COLLECTED 10/20/20, 6:00 A DLP Rochester Rochester DATE OF BIRTH RECEIVED 10/20/20, 2:52 P N AGE Y SEX ale REPORTED 10/20/20, 3:03 P The collected, received and reported dates and times are in the time zone of the performing location. REQUESTED BY CLIENT CLIENT CLIENT ORDER NUBER CLIENT RN NGSH ORDER NUBER F Specimen Type Indication for Test Peripheral blood DS PATHOGENIC UTATIONS DETECTED 1. JAK2: c.1849g>t;p.val617phe No other pathogenic mutations were detected in the other genes tested on the panel at the reportable limit of assay detection. See below for Variants of Unknown Significance and Additional Notes. Please see the section of "Panel Gene List" below for the complete list of genes tested. INTERPRETATION 1. JAK2: c.1849g>t;p.val617phe Normal gene/protein function: The JAK2 gene encodes the Janus kinase 2 (JAK2) protein, which belongs to the Janus family of non-receptor tyrosine kinases (JAK kinases). JAK proteins are pivotal for promoting the signal transduction events of multiple hematopoietic cytokines and growth factors (Ward et al, 2000, ). utation effect: The p.val617phe (V617F) mutation has been shown to be a gain of function resulting in constitutive activation of the JAK2 protein and cytokineindependent growth of hematopoietic cells (Levine et al, 2007, ). Disease associations: The JAK2 V617F mutation is implicated in myeloid neoplasms, especially the BCR-ABL1 negative classical myeloproliferative neoplasms (PN), in which mutations have been reported in at least 95% of polycythemia vera (PV) and 50-60% of essential thrombocythemia (ET) and primary myelofibrosis (PF) patients (Baxter et al, 2005, ; Levine et al, 2005, ; Kralovics et al, 2005, ; James et al, 2005, ). JAK2 V617F has also been described in association in the majority of cases of the rare myeloproliferative/myelodysplastic neoplasm (PN/DS) refractory anemia with ring sideroblasts and thrombocytosis (RARS-T) (alcovati et al, 2009, ), and only infrequently in other myeloid neoplasms, including myelodysplastic syndromes (DS) and other types of PN/DS (e.g. chronic myelomonocytic leukemia (CL), atypical chronic myeloid leukemia (acl)) (Steensma et al, 2005, ; Wang et al, 2014, ). Therapeutic implications: The JAK1/2 inhibitor Ruxolitinib has been approved for treatment in PF and demonstrated efficacy in improving constitutional symptoms and reducing spleen size (Verstovsek et al, 2012, ; Harrison et al 2012, ). In a phase 2 clinical trial, it demonstrated modest antileukemic activity as a single agent in patients with refractory post-pn AL (Eghtedar et al, 2012, ). Other JAK2 inhibitors, PI3K inhibitors and HDAC inhibitors are also being investigated in PN (Geyer et al, 2014, ). PATIENT NAE TESTINGRNV, REPORT -TL256 Form ID: ETCB Page 1 of 4
7 PATIENT NAE TESTINGRNV, REPORT PATIENT ID COLLECTED 10/20/20, 6:00 A DLP Rochester Rochester DATE OF BIRTH RECEIVED 10/20/20, 2:52 P N AGE Y SEX ale REPORTED 10/20/20, 3:03 P The collected, received and reported dates and times are in the time zone of the performing location. REQUESTED BY CLIENT CLIENT CLIENT ORDER NUBER CLIENT RN NGSH ORDER NUBER F CLINICAL TRIALS Information regarding possible clinical trials for this patient can be found at the following sites: 1). ClinicalTrials.gov: 2). ayo Clinic: 3). National Cancer Institute: 4). olecular atch: VARIANTS OF UNKNOWN SIGNIFICANCE (VUS) None ADDITIONAL NOTES None ETHOD SUARY DNA is extracted from the peripheral blood or bone marrow sample and following library preparation by hybrid capture, subjected to next generation sequencing (NGS) with post-sequencing analysis of tumor-associated mutations. Because some regions in the target panel are not consistently resolved by NGS, additional separate clinical laboratory assays may also be used to complete the overall analysis including: CALR (CALR), CEBPA (CEBPA), CSF3R (CSF3R), FLT3 (FLT), JAK2 V617F utation Detection (JAK2B, JAK2, or JAK2V), JAK2 Exon 12 utation Detection (JAKXB or JAKX), KIT (KITB, KITB, KITAS, or KITE), PL (PLB or PL), YD88 (YD88), NP1 (NP1), and TP53 (P53CA). Performance characteristics of NGS panel: Single base substitutions: accuracy >99%; reproducibility 100% (intra- and interassay); sensitivity 5-10% variant allele fraction with a minimum depth coverage of 250X. Insertion/deletion events: accuracy >99%; reproducibility 100% (intra- and interassay); sensitivity 5-10% variant allele fraction with a minimum depth coverage of 250X. This test was developed and its performance characteristics determined by ayo Clinic in a manner consistent with CLIA requirements. This test has not been cleared or approved by the U.S. Food and Drug Administration. DISCLAIER CLINICAL DISCLAIER utation calls detected between 5-10% variant allele fractions (VAF) may indicate low-level (i.e. subclonal) tumor populations, although the clinical significance of these findings may not be clear. Some apparent mutations classified as VUS may represent rare or low frequency polymorphisms. A low incidence of gene mutations associated with myeloid neoplasms can also be detected in PATIENT NAE TESTINGRNV, REPORT -TL256 Form ID: ETCB Page 2 of 4
8 PATIENT NAE TESTINGRNV, REPORT PATIENT ID COLLECTED 10/20/20, 6:00 A DLP Rochester Rochester DATE OF BIRTH RECEIVED 10/20/20, 2:52 P N AGE Y SEX ale REPORTED 10/20/20, 3:03 P The collected, received and reported dates and times are in the time zone of the performing location. REQUESTED BY CLIENT CLIENT CLIENT ORDER NUBER CLIENT RN NGSH ORDER NUBER F hematopoietic cells with advancing age in some individuals without evidence of a hematologic malignancy ("clonal hematopoiesis") and these alterations may not be clearly distinguishable from tumor-associated mutations (PID ; ; ). Prior treatment for hematologic malignancy could affect the results obtained in this assay. In particular, prior allogeneic hematopoietic stem cell transplant (HSCT) may cause difficulties in resolving somatic or polymorphic alterations, or in assigning variant calls correctly to donor and recipient fractions, if pertinent clinical or laboratory information (e.g. chimerism engraftment status) is not provided. Correlation with clinical, histopathologic and additional laboratory findings is required for final interpretation of these results. This assay does not distinguish between somatic and germ line alterations in analyzed gene regions, particularly with VAF near 50% or 100%. If nucleotide alterations in genes associated with germ line mutation syndromes are present and there is also a strong clinical suspicion or family history of malignant disease predisposition, additional genetic testing and appropriate counseling may be indicated. This report interpretation is based on current medical and scientific literature, but clinical significance may not be completely established for all reported target gene abnormalities identified. The final interpretation of results for clinical management of the patient is the responsibility of the managing physician. TECHNICAL DISCLAIER The depth of sequencing coverage may be variable for some target regions, but assay performance below the minimum acceptable criteria, or for failed regions are noted. Analysis of rare (low allele frequency) polymorphisms may be problematic in some cases. A low tumor cell percentage in the sample may affect the true mutation VAF and/or sensitivity. Suboptimal- performing regions (i.e. less than the expected minimum depth of coverage) may affect analytic sensitivity for detecting lower level mutations. This is a qualitative test. The variant read fractions are provided for information only and represent a relative proportion of mutated alleles, but do not indicate a measure of analytical sensitivity for the given genes; assay sensitivity is as stated in the method summary. Some genetic or genomic alterations, such as large insertion/deletion (indel) events, copy number alterations (CNA) and gene translocation events are not detected by this assay. PANEL GENE LIST ASXL1 (N_ ) exons 11-14, BCOR (N_ ) exons 5-, BRAF (N_ ) exon 15, CALR (N_ ) exon 9, CBL (N_ ) exon 8, intron 8, and exon 9, CEBPA (N_ ) exon 1, CSF3R (N_ ) exons 14 and 17, DNT3A (N_ ) exons 8-23, ETV6 (N_ ) exons 3-8, EZH2 (N_ ) exons 3-21, FLT3 (N_ ) exons 14-20, GATA1 (N_ ) exons 2 and 4, GATA2 (N_ ) exons 4-8, IDH1 (N_ ) exon 4, IDH2 (N_0028.3) exon 4, JAK2 (N_ ) exons 12-, KIT (N_ ) exons 8-11 and 17, KRAS (N_ ) exons 2-3, PL (N_ ) exons 10-11, YD88 (N_ ) exon 5, NOTCH1 (N_ ) exons 26, 27, and 34, NP1 (N_ ) exons 9, 11, and 12, NRAS (N_ ) exons 2 and 3, PHF6 (N_ ) exons 2-10, PTPN11 (N_ ) exons 3-4 and 12-13, RUNX1 (N_ ) exons 4-10, SETBP1 (N_ ) partial exon 6; amino acids , SF3B1 (N_ ) exons 13-, SRSF2 (N_0030.4) exons 1 and 2, TERT (N_ ) exons 2-, TET2 (N_ ) exons 3-11, TP53 (N_ ) exons 4-9, U2AF1 (N_ ) exons 2, 7, and 9, WT1 (N_ Aas.3) exons 1-11, and ZRSR2 (N_ ) exons For some genes, the transcript IDs used in this analysis may not be the same as in other cancer mutation databases, such as COSIC ( REVIEWED BY Signing Pathologist: elissa Tricker-Klar PATIENT NAE TESTINGRNV, REPORT -TL256 Form ID: ETCB Page 3 of 4
9 PATIENT NAE TESTINGRNV, REPORT PATIENT ID COLLECTED 10/20/20, 6:00 A DLP Rochester Rochester DATE OF BIRTH RECEIVED 10/20/20, 2:52 P N AGE Y SEX ale REPORTED 10/20/20, 3:03 P The collected, received and reported dates and times are in the time zone of the performing location. REQUESTED BY CLIENT CLIENT CLIENT ORDER NUBER CLIENT RN NGSH ORDER NUBER F CODE LABORATORY ADDRESS LAB DIRECTOR CR ayo Clinic Laboratories - Rochester ain Campus 200 FIRST STREET SW ROCHESTER N, WILLIA G ORICE, II, D, PhD Report times for Laboratory Name performed tests are CST/CDT. The collected, received, and reported dates and times on the report are in the time zone of the performing location. PATIENT NAE TESTINGRNV, REPORT -TL256 Form ID: ETCB Page 4 of 4
Laboratory Service Report
 Client C7028846-DLP Rochester Rochester, N 55901 Specimen Type Peripheral blood CR PDF Report available at: https://test.mmlaccess.com/reports/c7028846-zwselwql7p.ashx Indication for Test DS CR Pathogenic
Client C7028846-DLP Rochester Rochester, N 55901 Specimen Type Peripheral blood CR PDF Report available at: https://test.mmlaccess.com/reports/c7028846-zwselwql7p.ashx Indication for Test DS CR Pathogenic
Introduction of an NGS gene panel into the Haemato-Oncology MPN service
 Introduction of an NGS gene panel into the Haemato-Oncology MPN service Dr. Anna Skowronska, Dr Jane Bryon, Dr Samuel Clokie, Dr Yvonne Wallis and Professor Mike Griffiths West Midlands Regional Genetics
Introduction of an NGS gene panel into the Haemato-Oncology MPN service Dr. Anna Skowronska, Dr Jane Bryon, Dr Samuel Clokie, Dr Yvonne Wallis and Professor Mike Griffiths West Midlands Regional Genetics
Illumina Trusight Myeloid Panel validation A R FHAN R A FIQ
 Illumina Trusight Myeloid Panel validation A R FHAN R A FIQ G E NETIC T E CHNOLOGIST MEDICAL G E NETICS, CARDIFF To Cover Background to the project Choice of panel Validation process Genes on panel, Protocol
Illumina Trusight Myeloid Panel validation A R FHAN R A FIQ G E NETIC T E CHNOLOGIST MEDICAL G E NETICS, CARDIFF To Cover Background to the project Choice of panel Validation process Genes on panel, Protocol
Next generation sequencing analysis - A UK perspective. Nicholas Lea
 Next generation sequencing analysis - A UK perspective Nicholas Lea King s HMDC LMH is part of an integrated pathology service at King s Haematological Malignancy Diagnostic Centre (HMDC) HMDC serves population
Next generation sequencing analysis - A UK perspective Nicholas Lea King s HMDC LMH is part of an integrated pathology service at King s Haematological Malignancy Diagnostic Centre (HMDC) HMDC serves population
Next Generation Sequencing in Haematological Malignancy: A European Perspective. Wolfgang Kern, Munich Leukemia Laboratory
 Next Generation Sequencing in Haematological Malignancy: A European Perspective Wolfgang Kern, Munich Leukemia Laboratory Diagnostic Methods Cytomorphology Cytogenetics Immunophenotype Histology FISH Molecular
Next Generation Sequencing in Haematological Malignancy: A European Perspective Wolfgang Kern, Munich Leukemia Laboratory Diagnostic Methods Cytomorphology Cytogenetics Immunophenotype Histology FISH Molecular
The Evolving Role of Transplantation for MPN
 The Evolving Role of Transplantation for MPN (PMF, PV, ET) H.Joachim Deeg MD Fred Hutchinson Cancer Research Center, University of Washington, Seattle WA, SCCA jdeeg@fhcrc.org 10 th J. Niblack Conference
The Evolving Role of Transplantation for MPN (PMF, PV, ET) H.Joachim Deeg MD Fred Hutchinson Cancer Research Center, University of Washington, Seattle WA, SCCA jdeeg@fhcrc.org 10 th J. Niblack Conference
Out-Patient Billing CPT Codes
 Out-Patient Billing CPT Codes Updated Date: August 3, 08 Client Billed Molecular Tests HPV DNA Tissue Testing 8764 No Medicare Billed - Molecular Tests NeoARRAY NeoARRAY SNP/Cytogenetic No 89 NeoLAB NeoLAB
Out-Patient Billing CPT Codes Updated Date: August 3, 08 Client Billed Molecular Tests HPV DNA Tissue Testing 8764 No Medicare Billed - Molecular Tests NeoARRAY NeoARRAY SNP/Cytogenetic No 89 NeoLAB NeoLAB
The Center for PERSONALIZED DIAGNOSTICS
 The Center for PERSONALIZED DIAGNOSTICS Precision Diagnostics for Personalized Medicine A joint initiative between The Department of Pathology and Laboratory Medicine & The Abramson Cancer Center The (CPD)
The Center for PERSONALIZED DIAGNOSTICS Precision Diagnostics for Personalized Medicine A joint initiative between The Department of Pathology and Laboratory Medicine & The Abramson Cancer Center The (CPD)
[COMPREHENSIVE GENETIC ASSAY PANEL ON
 2014 SN GENELAB AND RESEARCH CENTER DR. SALIL VANIAWALA, PH.D [COMPREHENSIVE GENETIC ASSAY PANEL ON MYELOPROLIFERATIVE NEOPLASMS] SN Genelab presents one of the most comprehensive genetic assay panel for
2014 SN GENELAB AND RESEARCH CENTER DR. SALIL VANIAWALA, PH.D [COMPREHENSIVE GENETIC ASSAY PANEL ON MYELOPROLIFERATIVE NEOPLASMS] SN Genelab presents one of the most comprehensive genetic assay panel for
Molecular. Oncology & Pathology. Diagnostic, Prognostic, Therapeutic, and Predisposition Tests in Precision Medicine. Liquid Biopsy.
 Molecular Oncology & Pathology Hereditary Cancer Somatic Cancer Liquid Biopsy Next-Gen Sequencing qpcr Sanger Sequencing Diagnostic, Prognostic, Therapeutic, and Predisposition Tests in Precision Medicine
Molecular Oncology & Pathology Hereditary Cancer Somatic Cancer Liquid Biopsy Next-Gen Sequencing qpcr Sanger Sequencing Diagnostic, Prognostic, Therapeutic, and Predisposition Tests in Precision Medicine
Please Silence Your Cell Phones. Thank You
 Please Silence Your Cell Phones Thank You Utility of NGS and Comprehensive Genomic Profiling in Hematopathology Practice Maria E. Arcila M.D. Memorial Sloan Kettering Cancer Center New York, NY Disclosure
Please Silence Your Cell Phones Thank You Utility of NGS and Comprehensive Genomic Profiling in Hematopathology Practice Maria E. Arcila M.D. Memorial Sloan Kettering Cancer Center New York, NY Disclosure
August 17, Dear Valued Client:
 August 7, 08 Re: CMS Announces 6-Month Period of Enforcement Discretion for Laboratory Date of Service Exception Policy Under the Medicare Clinical Laboratory Fee Schedule (the 4 Day Rule ) Dear Valued
August 7, 08 Re: CMS Announces 6-Month Period of Enforcement Discretion for Laboratory Date of Service Exception Policy Under the Medicare Clinical Laboratory Fee Schedule (the 4 Day Rule ) Dear Valued
NeoTYPE Cancer Profiles
 NeoTYPE Cancer Profiles Multimethod Analysis of 25+ Hematologic Diseases and Solid Tumors Anatomic Pathology FISH Molecular The next generation of diagnostic, prognostic, and therapeutic assessment NeoTYPE
NeoTYPE Cancer Profiles Multimethod Analysis of 25+ Hematologic Diseases and Solid Tumors Anatomic Pathology FISH Molecular The next generation of diagnostic, prognostic, and therapeutic assessment NeoTYPE
Should Mutational Status in Primary Myelofibrosis (PMF) Guide Therapy..YES!!!
 Should Mutational Status in Primary Myelofibrosis (PMF) Guide Therapy..YES!!! Lindsay Anne Magura Rein, MD Division of Hematologic Malignancies and Cellular Therapy/BMT A Little Bit of History.. 1665 Advanced
Should Mutational Status in Primary Myelofibrosis (PMF) Guide Therapy..YES!!! Lindsay Anne Magura Rein, MD Division of Hematologic Malignancies and Cellular Therapy/BMT A Little Bit of History.. 1665 Advanced
Opportunities for Optimal Testing in the Myeloproliferative Neoplasms. Curtis A. Hanson, MD
 Opportunities for Optimal Testing in the Myeloproliferative Neoplasms Curtis A. Hanson, MD 2013 MFMER slide-1 DISCLOSURES: Relevant Financial Relationship(s) None Off Label Usage None 2013 MFMER slide-2
Opportunities for Optimal Testing in the Myeloproliferative Neoplasms Curtis A. Hanson, MD 2013 MFMER slide-1 DISCLOSURES: Relevant Financial Relationship(s) None Off Label Usage None 2013 MFMER slide-2
Disclosures for Ayalew Tefferi
 Disclosures for Ayalew Tefferi Principal investigator role Employee Consultant Major Stockholder Speakers Bureau Scientific Advisory Board Janssen, Geron, Celgene, Sanofi-Aventis, Gilead Sciences, Incyte
Disclosures for Ayalew Tefferi Principal investigator role Employee Consultant Major Stockholder Speakers Bureau Scientific Advisory Board Janssen, Geron, Celgene, Sanofi-Aventis, Gilead Sciences, Incyte
ADRL Advanced Diagnostics Research Laboratory
 ADRL Advanced Diagnostics Research Laboratory John DeCoteau, MD FRCP Department of Pathology, Division of Hematopathology University of Saskatchewan Saskatchewan Cancer Agency ADRL Project Objectives New
ADRL Advanced Diagnostics Research Laboratory John DeCoteau, MD FRCP Department of Pathology, Division of Hematopathology University of Saskatchewan Saskatchewan Cancer Agency ADRL Project Objectives New
Precision Medicine and Molecular Testing.
 Precision Medicine and Molecular Testing. David A. Sallman, MD Assistant Member Department of Malignant Hematology Moffitt Cancer Center david.sallman@moffitt.org Disclosures Research funding for Celgene
Precision Medicine and Molecular Testing. David A. Sallman, MD Assistant Member Department of Malignant Hematology Moffitt Cancer Center david.sallman@moffitt.org Disclosures Research funding for Celgene
Technical Bulletin No. 100
 CPAL Central Pennsylvania Alliance Laboratory Technical Bulletin No. 100 August 2, 2012 JAK2 AND MPL 515 MUTATIONAL ANALYSIS Contact: Dr. Jeffrey Wisotzkey, 717-851-1422 Technical Director, CPAL Jill A.
CPAL Central Pennsylvania Alliance Laboratory Technical Bulletin No. 100 August 2, 2012 JAK2 AND MPL 515 MUTATIONAL ANALYSIS Contact: Dr. Jeffrey Wisotzkey, 717-851-1422 Technical Director, CPAL Jill A.
MPL W515L K mutation
 MPL W515L K mutation BCR-ABL genotyping The exact chromosomal defect in Philadelphia chromosome is a translocation. Parts of two chromosomes, 9 and 22, switch places. The result is a fusion gene, created
MPL W515L K mutation BCR-ABL genotyping The exact chromosomal defect in Philadelphia chromosome is a translocation. Parts of two chromosomes, 9 and 22, switch places. The result is a fusion gene, created
Concomitant WT1 mutations predicted poor prognosis in CEBPA double-mutated acute myeloid leukemia
 Concomitant WT1 mutations predicted poor prognosis in CEBPA double-mutated acute myeloid leukemia Feng-Ming Tien, Hsin-An Hou, Jih-Luh Tang, Yuan-Yeh Kuo, Chien-Yuan Chen, Cheng-Hong Tsai, Ming Yao, Chi-Cheng
Concomitant WT1 mutations predicted poor prognosis in CEBPA double-mutated acute myeloid leukemia Feng-Ming Tien, Hsin-An Hou, Jih-Luh Tang, Yuan-Yeh Kuo, Chien-Yuan Chen, Cheng-Hong Tsai, Ming Yao, Chi-Cheng
Prior Authorization Required: Additional Information:
 Genetic Testing for Somatic Tumor Markers MP9486 Covered Service: Prior Authorization Required: Additional Information: Yes when meets criteria below No A first-degree relative is defined as an individual
Genetic Testing for Somatic Tumor Markers MP9486 Covered Service: Prior Authorization Required: Additional Information: Yes when meets criteria below No A first-degree relative is defined as an individual
Leukemia and subsequent solid tumors among patients with myeloproliferative neoplasms
 Leukemia and subsequent solid tumors among patients with myeloproliferative neoplasms Tiziano Barbui (tbarbui@asst-pg23.it Hematology and Research Foundation,Ospedale Papa Giovanni XXIII, Bergamo Italy
Leukemia and subsequent solid tumors among patients with myeloproliferative neoplasms Tiziano Barbui (tbarbui@asst-pg23.it Hematology and Research Foundation,Ospedale Papa Giovanni XXIII, Bergamo Italy
Contractor Information. LCD Information
 Local Coverage Determination (LCD): MolDX: Genetic Testing for BCR-ABL Negative Myeloproliferative Disease (L36044) Links in PDF documents are not guaranteed to work. To follow a web link, please use the
Local Coverage Determination (LCD): MolDX: Genetic Testing for BCR-ABL Negative Myeloproliferative Disease (L36044) Links in PDF documents are not guaranteed to work. To follow a web link, please use the
Management of Myelodysplastic Syndromes
 Management of Myelodysplastic Syndromes Peter L. Greenberg, MD Stanford Cancer Institute Myelodysplastic Syndromes: Clinical & Molecular Advances for Disease Classification and Prognostication MDSs: A
Management of Myelodysplastic Syndromes Peter L. Greenberg, MD Stanford Cancer Institute Myelodysplastic Syndromes: Clinical & Molecular Advances for Disease Classification and Prognostication MDSs: A
West Midlands Regional Genetics Laboratory
 West Midlands Regional Genetics Laboratory Haemato-oncology service update letter October 2017 Dear colleagues, We are writing to outline the latest developments to our service, aiming to support the management
West Midlands Regional Genetics Laboratory Haemato-oncology service update letter October 2017 Dear colleagues, We are writing to outline the latest developments to our service, aiming to support the management
CLINICAL POLICY DEPARTMENT: Medical Management DOCUMENT NAME: JakafiTM REFERENCE NUMBER: NH.PHAR.98
 PAGE: 1 of 6 IMPORTANT REMINDER This Clinical Policy has been developed by appropriately experienced and licensed health care professionals based on a thorough review and consideration of generally accepted
PAGE: 1 of 6 IMPORTANT REMINDER This Clinical Policy has been developed by appropriately experienced and licensed health care professionals based on a thorough review and consideration of generally accepted
JAK2 V617F analysis. Indication: monitoring of therapy
 JAK2 V617F analysis BCR-ABL genotyping The exact chromosomal defect in Philadelphia chromosome is a translocation. Parts of two chromosomes, 9 and 22, switch places. The result is a fusion gene, created
JAK2 V617F analysis BCR-ABL genotyping The exact chromosomal defect in Philadelphia chromosome is a translocation. Parts of two chromosomes, 9 and 22, switch places. The result is a fusion gene, created
Template for Reporting Results of Biomarker Testing for Myeloproliferative Neoplasms
 Template for Reporting Results of Biomarker Testing for Myeloproliferative Neoplasms Version: MPNBiomarkers 1.0.0.2 Protocol Posting Date: June 2017 This biomarker template is NOT required for accreditation
Template for Reporting Results of Biomarker Testing for Myeloproliferative Neoplasms Version: MPNBiomarkers 1.0.0.2 Protocol Posting Date: June 2017 This biomarker template is NOT required for accreditation
ASBMT MDS/MPN Update Sunil Abhyankar, MD
 ASBMT MDS/MPN Update Sunil Abhyankar, MD Professor of Medicine Medical Director, Pheresis and Cell Processing Division of Hematologic Malignancies and Cellular Therapeutics Department of Internal Medicine
ASBMT MDS/MPN Update Sunil Abhyankar, MD Professor of Medicine Medical Director, Pheresis and Cell Processing Division of Hematologic Malignancies and Cellular Therapeutics Department of Internal Medicine
Contractor Information
 Local Coverage Determination (LCD): MolDX: Genetic Testing for BCR-ABL Negative Myeloproliferative Disease (L36186) Links in PDF documents are not guaranteed to work. To follow a web link, please use the
Local Coverage Determination (LCD): MolDX: Genetic Testing for BCR-ABL Negative Myeloproliferative Disease (L36186) Links in PDF documents are not guaranteed to work. To follow a web link, please use the
DISCLOSURE Luca Malcovati, MD. No financial relationships to disclose
 ICUS, CCUS and CHIP Luca Malcovati, MD Department of Molecular Medicine, University of Pavia Medical School, & Department of Hematology Oncology, IRCCS Policlinico S. Matteo Foundation, Pavia, Italy DISCLOSURE
ICUS, CCUS and CHIP Luca Malcovati, MD Department of Molecular Medicine, University of Pavia Medical School, & Department of Hematology Oncology, IRCCS Policlinico S. Matteo Foundation, Pavia, Italy DISCLOSURE
Molecular profiling in confirming the diagnosis of early myelodysplastic syndrome
 Molecular profiling of early MDS Hematopathology - March 2016 Article Molecular profiling in confirming the diagnosis of early myelodysplastic syndrome Maya Thangavelu 1,*, Ryan Olson 2, Li Li 2, Wanlong
Molecular profiling of early MDS Hematopathology - March 2016 Article Molecular profiling in confirming the diagnosis of early myelodysplastic syndrome Maya Thangavelu 1,*, Ryan Olson 2, Li Li 2, Wanlong
Myelodysplastic syndromes and the new WHO 2016 classification
 Myelodysplastic syndromes and the new WHO 2016 classification 32nd General Annual Meeting of the Belgian Hematology Society 10-11 February 2017 Gregor Verhoef, Departement of Hematology, University Hospital
Myelodysplastic syndromes and the new WHO 2016 classification 32nd General Annual Meeting of the Belgian Hematology Society 10-11 February 2017 Gregor Verhoef, Departement of Hematology, University Hospital
Session 4: Summary and Conclusions
 Session 4: Summary and Conclusions Total cases in Session 4 Myeloproliferative neoplasms 16 cases Oral #300 (CEL, NOS) Mastocytosis 2 cases Oral #156 (SM-AHN) Myeloid/lymphoid neoplasms with eosinophilia
Session 4: Summary and Conclusions Total cases in Session 4 Myeloproliferative neoplasms 16 cases Oral #300 (CEL, NOS) Mastocytosis 2 cases Oral #156 (SM-AHN) Myeloid/lymphoid neoplasms with eosinophilia
Corporate Medical Policy. Policy Effective February 23, 2018
 Corporate Medical Policy Genetic Testing for FLT3, NPM1 and CEBPA Mutations in Acute File Name: Origination: Last CAP Review: Next CAP Review: Last Review: genetic_testing_for_flt3_npm1_and_cebpa_mutations_in_acute_myeloid_leukemia
Corporate Medical Policy Genetic Testing for FLT3, NPM1 and CEBPA Mutations in Acute File Name: Origination: Last CAP Review: Next CAP Review: Last Review: genetic_testing_for_flt3_npm1_and_cebpa_mutations_in_acute_myeloid_leukemia
Examining Genetics and Genomics of Acute Myeloid Leukemia in 2017
 Examining Genetics and Genomics of Acute Myeloid Leukemia in 2017 Elli Papaemmanuil, PhD Memorial Sloan Kettering Cancer Center New York, New York, United States Today s Talk Cancer genome introduction
Examining Genetics and Genomics of Acute Myeloid Leukemia in 2017 Elli Papaemmanuil, PhD Memorial Sloan Kettering Cancer Center New York, New York, United States Today s Talk Cancer genome introduction
Myelodysplastic syndromes Impact of Biology. Lionel Adès Hopital Saint Louis Groupe Francophone des SMD. Épidémiologie
 Myelodysplastic syndromes Impact of Biology Lionel Adès Hopital Saint Louis Groupe Francophone des SMD Épidémiologie Incidence : 3 à 6 / 100 000 hab. / An Prédomine chez les sujets âgés Augmentation de
Myelodysplastic syndromes Impact of Biology Lionel Adès Hopital Saint Louis Groupe Francophone des SMD Épidémiologie Incidence : 3 à 6 / 100 000 hab. / An Prédomine chez les sujets âgés Augmentation de
Disclosure: Objectives/Outline. Leukemia: Genealogy of Pathology Practice: Old Diseases New Expectations. Nothing to disclose.
 RC1 Leukemia: Genealogy of Pathology Practice: Old Diseases New Expectations RC2 Disclosure: Nothing to disclose Henry Moon Lecture: UCSF Annual Conference Kathryn Foucar, MD kfoucar@salud.unm.edu May
RC1 Leukemia: Genealogy of Pathology Practice: Old Diseases New Expectations RC2 Disclosure: Nothing to disclose Henry Moon Lecture: UCSF Annual Conference Kathryn Foucar, MD kfoucar@salud.unm.edu May
SESSION 1 Reactive cytopenia and dysplasia
 SESSION 1 Reactive cytopenia and dysplasia Falko Fend, Tübingen & Alexandar Tzankov, Basel 1 Disclosure of speaker s interests (Potential) conflict of interest none Potentially relevant company relationships
SESSION 1 Reactive cytopenia and dysplasia Falko Fend, Tübingen & Alexandar Tzankov, Basel 1 Disclosure of speaker s interests (Potential) conflict of interest none Potentially relevant company relationships
Molecular Markers. Marcie Riches, MD, MS Associate Professor University of North Carolina Scientific Director, Infection and Immune Reconstitution WC
 Molecular Markers Marcie Riches, MD, MS Associate Professor University of North Carolina Scientific Director, Infection and Immune Reconstitution WC Overview Testing methods Rationale for molecular testing
Molecular Markers Marcie Riches, MD, MS Associate Professor University of North Carolina Scientific Director, Infection and Immune Reconstitution WC Overview Testing methods Rationale for molecular testing
7/12/2016 TESTING. Objectives. New Directions in Aplastic Anemia: What's on the Horizon? Better way to evaluate clonal evolution?
 New Directions in Aplastic Anemia: What's on the Horizon? Amy E. DeZern, MD; MHS Assistant Professor Hematologic Malignancies Sidney Kimmel Comprehensive Cancer Center at Johns Hopkins Baltimore, MD Objectives
New Directions in Aplastic Anemia: What's on the Horizon? Amy E. DeZern, MD; MHS Assistant Professor Hematologic Malignancies Sidney Kimmel Comprehensive Cancer Center at Johns Hopkins Baltimore, MD Objectives
WHO Update to Myeloproliferative Neoplasms
 WHO Update to Myeloproliferative Neoplasms Archana M Agarwal, MD, Associate Professor of Pathology University of Utah Department of Pathology/ARUP Laboratories Myeloproliferative Neoplasms The categories
WHO Update to Myeloproliferative Neoplasms Archana M Agarwal, MD, Associate Professor of Pathology University of Utah Department of Pathology/ARUP Laboratories Myeloproliferative Neoplasms The categories
The preclinical efficacy of a novel telomerase inhibitor, imetelstat, in AML: A randomized trial in patient-derived xenografts
 The preclinical efficacy of a novel telomerase inhibitor, imetelstat, in AML: A randomized trial in patient-derived xenografts Claudia Bruedigam, Ph.D Gordon and Jessie Gilmour Leukaemia Research Laboratory
The preclinical efficacy of a novel telomerase inhibitor, imetelstat, in AML: A randomized trial in patient-derived xenografts Claudia Bruedigam, Ph.D Gordon and Jessie Gilmour Leukaemia Research Laboratory
Supporting Information
 Supporting Information Rampal et al. 10.1073/pnas.1407792111 Fig. S1. Genetic events in leukemic transformation of chronic-phase MPNs. (A) Survival of post-mpn AML patients according to mutational status
Supporting Information Rampal et al. 10.1073/pnas.1407792111 Fig. S1. Genetic events in leukemic transformation of chronic-phase MPNs. (A) Survival of post-mpn AML patients according to mutational status
Pathogenesis and management of CMML
 Pathogenesis and management of CMML Raphaël Itzykson, Hôpital Saint-Louis, Paris International Conference of the Korean Society of Hematology March 29th 2018 대한혈액학회 Korean Society of Hematology COI disclosure
Pathogenesis and management of CMML Raphaël Itzykson, Hôpital Saint-Louis, Paris International Conference of the Korean Society of Hematology March 29th 2018 대한혈액학회 Korean Society of Hematology COI disclosure
Myelodysplastic syndrome (MDS) & Myeloproliferative neoplasms
 Myelodysplastic syndrome (MDS) & Myeloproliferative neoplasms Myelodysplastic syndrome (MDS) A multipotent stem cell that can differentiate into any of the myeloid lineage cells (RBCs, granulocytes, megakaryocytes)
Myelodysplastic syndrome (MDS) & Myeloproliferative neoplasms Myelodysplastic syndrome (MDS) A multipotent stem cell that can differentiate into any of the myeloid lineage cells (RBCs, granulocytes, megakaryocytes)
Genetic Testing for Somatic Tumor Markers
 Genetic Testing for Somatic Tumor Markers MP9486 Covered Service: Prior Authorization Required: Additional Information: Yes when meets criteria below Yes-as shown below Genetic testing is covered for a
Genetic Testing for Somatic Tumor Markers MP9486 Covered Service: Prior Authorization Required: Additional Information: Yes when meets criteria below Yes-as shown below Genetic testing is covered for a
Targeted next-generation sequencing in blast phase myeloproliferative neoplasms
 REGULAR ARTICLE Targeted next-generation sequencing in blast phase myeloproliferative neoplasms Terra L. Lasho, Mythri Mudireddy, Christy M. Finke, Curtis A. Hanson, Rhett P. Ketterling, Natasha Szuber,
REGULAR ARTICLE Targeted next-generation sequencing in blast phase myeloproliferative neoplasms Terra L. Lasho, Mythri Mudireddy, Christy M. Finke, Curtis A. Hanson, Rhett P. Ketterling, Natasha Szuber,
Juan Ma 1, Jennifer Dunlap 2, Lisong Shen 1, Guang Fan 2 1
 Juan Ma 1, Jennifer Dunlap 2, Lisong Shen 1, Guang Fan 2 1 Xin Hua Hospital, Shanghai, China 2 Oregon Health & Science University, Portland, OR, United States AML is a hematopoietic neoplasms characterized
Juan Ma 1, Jennifer Dunlap 2, Lisong Shen 1, Guang Fan 2 1 Xin Hua Hospital, Shanghai, China 2 Oregon Health & Science University, Portland, OR, United States AML is a hematopoietic neoplasms characterized
Novità nelle MDS. Matteo G Della Porta. Cancer Center IRCCS Humanitas Research Hospital & Humanitas University Rozzano Milano, Italy
 Novità nelle MDS Matteo G Della Porta Cancer Center IRCCS Humanitas Research Hospital & Humanitas University Rozzano Milano, Italy matteo.della_porta@hunimed.eu Outline ARCH Predictive value of somatic
Novità nelle MDS Matteo G Della Porta Cancer Center IRCCS Humanitas Research Hospital & Humanitas University Rozzano Milano, Italy matteo.della_porta@hunimed.eu Outline ARCH Predictive value of somatic
Blastic Plasmacytoid Dendritic Cell Neoplasm with DNMT3A and TET2 mutations (SH )
 Blastic Plasmacytoid Dendritic Cell Neoplasm with DNMT3A and TET2 mutations (SH2017-0314) Habibe Kurt, Joseph D. Khoury, Carlos E. Bueso-Ramos, Jeffrey L. Jorgensen, Guilin Tang, L. Jeffrey Medeiros, and
Blastic Plasmacytoid Dendritic Cell Neoplasm with DNMT3A and TET2 mutations (SH2017-0314) Habibe Kurt, Joseph D. Khoury, Carlos E. Bueso-Ramos, Jeffrey L. Jorgensen, Guilin Tang, L. Jeffrey Medeiros, and
Published Ahead of Print on April 14, 2016, as doi: /haematol Copyright 2016 Ferrata Storti Foundation.
 Published Ahead of Print on April 14, 2016, as doi:10.3324/haematol.2016.143214. Copyright 2016 Ferrata Storti Foundation. Immunohistochemical pattern of p53 is a measure of TP53 mutation burden and adverse
Published Ahead of Print on April 14, 2016, as doi:10.3324/haematol.2016.143214. Copyright 2016 Ferrata Storti Foundation. Immunohistochemical pattern of p53 is a measure of TP53 mutation burden and adverse
NeoTYPE Cancer Profiles
 NeoTYPE Cancer Profiles 30+ Multimethod Assays for Hematologic Diseases and Solid Tumors Molecular FISH Anatomic Pathology The next generation of diagnostic, prognostic, and therapeutic assessment What
NeoTYPE Cancer Profiles 30+ Multimethod Assays for Hematologic Diseases and Solid Tumors Molecular FISH Anatomic Pathology The next generation of diagnostic, prognostic, and therapeutic assessment What
Related Policies None
 Medical Policy MP 2.04.60 BCBSA Ref. Policy: 2.04.60 Last Review: 07/25/2018 Effective Date: 07/25/2018 Section: Medicine Related Policies None DISCLAIMER Our medical policies are designed for informational
Medical Policy MP 2.04.60 BCBSA Ref. Policy: 2.04.60 Last Review: 07/25/2018 Effective Date: 07/25/2018 Section: Medicine Related Policies None DISCLAIMER Our medical policies are designed for informational
Heme 9 Myeloid neoplasms
 Heme 9 Myeloid neoplasms The minimum number of blasts to diagnose acute myeloid leukemia is 5% 10% 20% 50% 80% AML with the best prognosis is AML with recurrent cytogenetic abnormality AML with myelodysplasia
Heme 9 Myeloid neoplasms The minimum number of blasts to diagnose acute myeloid leukemia is 5% 10% 20% 50% 80% AML with the best prognosis is AML with recurrent cytogenetic abnormality AML with myelodysplasia
Supplementary Information
 Supplementary Information Table of Contents Supplementary methods... 2 Figure S1 - Variable DNA yield proportional to bone marrow aspirate cellularity.... 3 Figure S2 - Mutations by clinical ontogeny group....
Supplementary Information Table of Contents Supplementary methods... 2 Figure S1 - Variable DNA yield proportional to bone marrow aspirate cellularity.... 3 Figure S2 - Mutations by clinical ontogeny group....
Supplemental Material. The new provisional WHO entity RUNX1 mutated AML shows specific genetics without prognostic influence of dysplasia
 Supplemental Material The new provisional WHO entity RUNX1 mutated AML shows specific genetics without prognostic influence of dysplasia Torsten Haferlach, 1 Anna Stengel, 1 Sandra Eckstein, 1 Karolína
Supplemental Material The new provisional WHO entity RUNX1 mutated AML shows specific genetics without prognostic influence of dysplasia Torsten Haferlach, 1 Anna Stengel, 1 Sandra Eckstein, 1 Karolína
TEST MENU TEST CPT CODES TAT. Chromosome Analysis Bone Marrow x 2, 88264, x 3, Days
 TEST MENU CANCER/LEUKEMIA CHROMOSOME ANALYSIS Chromosome Analysis Bone Marrow 88237 x 2, 88264, 88280 x 3, 88291 4 Days Chromosome Analysis Bone Marrow Core 88237 x 2, 88264, 88280 x 3, 88291 4 Days Chromosome
TEST MENU CANCER/LEUKEMIA CHROMOSOME ANALYSIS Chromosome Analysis Bone Marrow 88237 x 2, 88264, 88280 x 3, 88291 4 Days Chromosome Analysis Bone Marrow Core 88237 x 2, 88264, 88280 x 3, 88291 4 Days Chromosome
Practical Considerations in the Treatment Myeloproliferative Neoplasms: Prognostication and Current Treatment Indy Hematology
 Practical Considerations in the Treatment Myeloproliferative Neoplasms: Prognostication and Current Treatment Indy Hematology Angela Fleischman MD PhD UC Irvine March 9, 2019 Disclosures: Angela Fleischman
Practical Considerations in the Treatment Myeloproliferative Neoplasms: Prognostication and Current Treatment Indy Hematology Angela Fleischman MD PhD UC Irvine March 9, 2019 Disclosures: Angela Fleischman
Session II: Summary. Robert P Hasserjian, MD Professor of Pathology Massachusetts General Hospital and Harvard Medical School
 Session II: Summary Robert P Hasserjian, MD Professor of Pathology Massachusetts General Hospital and Harvard Medical School Disclosure of speaker s interests (Prefix and Last Name) (Potential) conflict
Session II: Summary Robert P Hasserjian, MD Professor of Pathology Massachusetts General Hospital and Harvard Medical School Disclosure of speaker s interests (Prefix and Last Name) (Potential) conflict
Pediatric Oncology & Pathology Services
 Pediatric Oncology & Pathology Services Anatomic Pathology Flow Cytometry Cytogenetics Pharma Services Diagnostic, Prognostic, Predictive, and Predisposition Testing Pediatric Oncology & Pathology Services
Pediatric Oncology & Pathology Services Anatomic Pathology Flow Cytometry Cytogenetics Pharma Services Diagnostic, Prognostic, Predictive, and Predisposition Testing Pediatric Oncology & Pathology Services
Patricia Aoun MD, MPH Professor and Vice-Chair for Clinical Affairs Medical Director, Clinical Laboratories Department of Pathology City of Hope
 Patricia Aoun MD, MPH Professor and Vice-Chair for Clinical Affairs Medical Director, Clinical Laboratories Department of Pathology City of Hope National Medical Center Disclosures I have no disclosures
Patricia Aoun MD, MPH Professor and Vice-Chair for Clinical Affairs Medical Director, Clinical Laboratories Department of Pathology City of Hope National Medical Center Disclosures I have no disclosures
Genetic analysis and preclinical modeling of leukemic transformation of myeloproliferative neoplasms: Implications for therapeutic strategies
 Genetic analysis and preclinical modeling of leukemic transformation of myeloproliferative neoplasms: Implications for therapeutic strategies Raajit Rampal, MD, PhD Assistant Attending Physician Leukemia
Genetic analysis and preclinical modeling of leukemic transformation of myeloproliferative neoplasms: Implications for therapeutic strategies Raajit Rampal, MD, PhD Assistant Attending Physician Leukemia
Calreticulin Exon 9 Mutations in Myeloproliferative Neoplasms
 Original Article Diagnostic Hematology Ann Lab Med 2015;35:22-27 http://dx.doi.org/10.3343/alm.2015.35.1.22 ISSN 2234-3806 eissn 2234-3814 Calreticulin Exon 9 Mutations in Myeloproliferative Neoplasms
Original Article Diagnostic Hematology Ann Lab Med 2015;35:22-27 http://dx.doi.org/10.3343/alm.2015.35.1.22 ISSN 2234-3806 eissn 2234-3814 Calreticulin Exon 9 Mutations in Myeloproliferative Neoplasms
Acute Myeloid Leukemia with RUNX1 and Several Co-mutations
 Case SH2017-0281 Acute Myeloid Leukemia with RUNX1 and Several Co-mutations James Bauer, MD, PhD David Yang, MD Erik Ranheim, MD, PhD Catherine Leith, MB, Bchir Clinical History Chief Complaint: 72 year
Case SH2017-0281 Acute Myeloid Leukemia with RUNX1 and Several Co-mutations James Bauer, MD, PhD David Yang, MD Erik Ranheim, MD, PhD Catherine Leith, MB, Bchir Clinical History Chief Complaint: 72 year
Overview. Methods 9/11/2017. Next Generation Sequencing and Precision Medicine in Hematological Malignancies. Genotyping in hematology
 Overview Next Generation Sequencing and Precision Medicine in Hematological Malignancies Sharathkumar Bhagavathi, MD University of Iowa Carver College of Medicine NGS as a genotyping platform in hematopathology
Overview Next Generation Sequencing and Precision Medicine in Hematological Malignancies Sharathkumar Bhagavathi, MD University of Iowa Carver College of Medicine NGS as a genotyping platform in hematopathology
Recommended Timing for Transplant Consultation
 REFERRAL GUIDELINES Recommended Timing for Transplant Consultation Published jointly by the National Marrow Donor Program /Be The Match and the American Society for Blood and Marrow Transplantation BeTheMatchClinical.org
REFERRAL GUIDELINES Recommended Timing for Transplant Consultation Published jointly by the National Marrow Donor Program /Be The Match and the American Society for Blood and Marrow Transplantation BeTheMatchClinical.org
WHO Classification of Myeloid Neoplasms with Defined Molecular Abnormalities
 WHO Classification of Myeloid Neoplasms with Defined Molecular Abnormalities Robert W. McKenna, M.D. 1/2009 WHO Classification of Myeloid Neoplasms (4th Edition)--2008 Incorporates new information that
WHO Classification of Myeloid Neoplasms with Defined Molecular Abnormalities Robert W. McKenna, M.D. 1/2009 WHO Classification of Myeloid Neoplasms (4th Edition)--2008 Incorporates new information that
Ferrata Storti Foundation
 DNMT3A mutations occur early or late in patients with myeloproliferative neoplasms and mutation order influences phenotype Somatic mutations in JAK2, CALR and MPL are found in the majority of myeloproliferative
DNMT3A mutations occur early or late in patients with myeloproliferative neoplasms and mutation order influences phenotype Somatic mutations in JAK2, CALR and MPL are found in the majority of myeloproliferative
A. ILEA 1* A. M. VLADAREANU 2 E. NICULESCU-MIZIL 3
 Bulletin of the Transilvania University of Braşov Series VI: Medical Sciences Vol. 9 (58) No. 1-2016 THE SIMULTANEOUS OCCURRENCE OF THE BCR-ABL TRANSLOCATION AND THE Jak2V617F MUTATION. DETECTION AND DYNAMICS
Bulletin of the Transilvania University of Braşov Series VI: Medical Sciences Vol. 9 (58) No. 1-2016 THE SIMULTANEOUS OCCURRENCE OF THE BCR-ABL TRANSLOCATION AND THE Jak2V617F MUTATION. DETECTION AND DYNAMICS
Supplementary Figure 1
 Count Count Supplementary Figure 1 Coverage per amplicon for error-corrected sequencing experiments. Errorcorrected consensus sequence (ECCS) coverage was calculated for each of the 568 amplicons in the
Count Count Supplementary Figure 1 Coverage per amplicon for error-corrected sequencing experiments. Errorcorrected consensus sequence (ECCS) coverage was calculated for each of the 568 amplicons in the
NGS Gateway Lab Services
 TM NGS Gateway Lab Services Accelerating Precision Medicine Design a Complete Genomic Testing Portfolio with Turnkey Assays About NGS Gateway Lab Services TM Designed to provide a gateway to your own in-house
TM NGS Gateway Lab Services Accelerating Precision Medicine Design a Complete Genomic Testing Portfolio with Turnkey Assays About NGS Gateway Lab Services TM Designed to provide a gateway to your own in-house
BHS Annual Meeting
 BHS Annual Meeting 2014 01.02.2014 Implementing next-generation deepsequencing assays in diagnostic algorithms in hematological malignancies Dr. Alexander Kohlmann Medical Need for Molecular Characterization
BHS Annual Meeting 2014 01.02.2014 Implementing next-generation deepsequencing assays in diagnostic algorithms in hematological malignancies Dr. Alexander Kohlmann Medical Need for Molecular Characterization
Session 5. Pre-malignant clonal hematopoietic proliferations. Chairs: Frank Kuo and Valentina Nardi
 Session 5 Pre-malignant clonal hematopoietic proliferations Chairs: Frank Kuo and Valentina Nardi Pre-malignant clonal hematopoietic proliferations Clonal LYMPHOID proliferations: - Monoclonal gammopathy
Session 5 Pre-malignant clonal hematopoietic proliferations Chairs: Frank Kuo and Valentina Nardi Pre-malignant clonal hematopoietic proliferations Clonal LYMPHOID proliferations: - Monoclonal gammopathy
Enhancing Assessment of Myeloid Disorders in the Era of Precision Medicine
 Enhancing Assessment of Myeloid Disorders in the Era of Precision Medicine Michelle Afkhami, M.D. Medical Director, Clinical Molecular Diagnostic Laboratory City of Hope National Medical Center Objectives
Enhancing Assessment of Myeloid Disorders in the Era of Precision Medicine Michelle Afkhami, M.D. Medical Director, Clinical Molecular Diagnostic Laboratory City of Hope National Medical Center Objectives
Myeloproliferative Disorders: Diagnostic Enigmas, Therapeutic Dilemmas. James J. Stark, MD, FACP
 Myeloproliferative Disorders: Diagnostic Enigmas, Therapeutic Dilemmas James J. Stark, MD, FACP Medical Director, Cancer Program and Palliative Care Maryview Medical Center Professor of Medicine, EVMS
Myeloproliferative Disorders: Diagnostic Enigmas, Therapeutic Dilemmas James J. Stark, MD, FACP Medical Director, Cancer Program and Palliative Care Maryview Medical Center Professor of Medicine, EVMS
5/21/2018. Disclosures. Objectives. Normal blood cells production. Bone marrow failure syndromes. Story of DNA
 AML: Understanding your diagnosis and current and emerging treatments Nothing to disclose. Disclosures Mohammad Abu Zaid, MD Assistant Professor of Medicine Indiana University School of Medicine Indiana
AML: Understanding your diagnosis and current and emerging treatments Nothing to disclose. Disclosures Mohammad Abu Zaid, MD Assistant Professor of Medicine Indiana University School of Medicine Indiana
SUPPLEMENTARY INFORMATION
 Supplementary Information S1 Frequency of DNMT3A mutations in hematologic disorders and their associated clinical phenotypes. Disease Patient population Frequency (%) Associated Clinical Characteristics
Supplementary Information S1 Frequency of DNMT3A mutations in hematologic disorders and their associated clinical phenotypes. Disease Patient population Frequency (%) Associated Clinical Characteristics
Spectrum of somatically acquired mutations identified by combining WES and genome-wide DNA array analysis in the discovery cohort of 30 JMML cases.
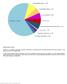 Supplementary Figure 1 Spectrum of somatically acquired mutations identified by combining WES and genome-wide DNA array analysis in the discovery cohort of 30 JMML cases. A total of 85 somatically acquired
Supplementary Figure 1 Spectrum of somatically acquired mutations identified by combining WES and genome-wide DNA array analysis in the discovery cohort of 30 JMML cases. A total of 85 somatically acquired
Intro alla patologia. Giovanni Barosi. Fondazione IRCCS Policlinico San Matteo Pavia
 Settima Giornata Fiorentina dedicata ai pazienti con malattie mieloproliferative croniche Sabato 13 Maggio 2017 CRIMM Centro di Ricerca e Innovazione per le Malattie Mieloproliferative AOU Careggi Intro
Settima Giornata Fiorentina dedicata ai pazienti con malattie mieloproliferative croniche Sabato 13 Maggio 2017 CRIMM Centro di Ricerca e Innovazione per le Malattie Mieloproliferative AOU Careggi Intro
Disclosures. I do not have anything to disclose. Shared Features of MPNs. Overview. Diagnosis and Molecular Monitoring in the
 Myeloproliferative Neoplasms: Diagnosis and Molecular Monitoring in the Target Therapy Era C. Cameron Yin, M.D., Ph.D. Department of Hematopathology UT MD Anderson Cancer Center Disclosures I do not have
Myeloproliferative Neoplasms: Diagnosis and Molecular Monitoring in the Target Therapy Era C. Cameron Yin, M.D., Ph.D. Department of Hematopathology UT MD Anderson Cancer Center Disclosures I do not have
Mayo alliance prognostic system for mastocytosis: clinical and hybrid clinical-molecular models
 REGULAR ARTICLE Mayo alliance prognostic system for mastocytosis: clinical and hybrid clinical-molecular models Animesh Pardanani, 1 Sahrish Shah, 1 Francesco Mannelli, 2 Yoseph C. Elala, 1 Paola Guglielmelli,
REGULAR ARTICLE Mayo alliance prognostic system for mastocytosis: clinical and hybrid clinical-molecular models Animesh Pardanani, 1 Sahrish Shah, 1 Francesco Mannelli, 2 Yoseph C. Elala, 1 Paola Guglielmelli,
Update on Myelodysplastic Syndromes and Myeloproliferative Neoplasms. Kaaren Reichard Mayo Clinic Rochester
 Update on Myelodysplastic Syndromes and Myeloproliferative Neoplasms Kaaren Reichard Mayo Clinic Rochester Reichard.kaaren@mayo.edu Nothing to disclose Conflict of Interest Learning Objectives Present
Update on Myelodysplastic Syndromes and Myeloproliferative Neoplasms Kaaren Reichard Mayo Clinic Rochester Reichard.kaaren@mayo.edu Nothing to disclose Conflict of Interest Learning Objectives Present
Click to edit Master /tle style
 Click to edit Master /tle style Tel: (314) 747-7337 Toll Free: (866) 450-7697 Fax: (314) 747-7336 Email: gps@wustl.edu Website: gps.wustl.edu GENETIC TESTING IN CANCER Ka/nka Vigh-Conrad, PhD Genomics
Click to edit Master /tle style Tel: (314) 747-7337 Toll Free: (866) 450-7697 Fax: (314) 747-7336 Email: gps@wustl.edu Website: gps.wustl.edu GENETIC TESTING IN CANCER Ka/nka Vigh-Conrad, PhD Genomics
Acute leukemia and myelodysplastic syndromes
 11/01/2012 Post-ASH meeting 1 Acute leukemia and myelodysplastic syndromes Peter Vandenberghe Centrum Menselijke Erfelijkheid & Afdeling Hematologie, UZ Leuven 11/01/2012 Post-ASH meeting 2 1. Acute myeloid
11/01/2012 Post-ASH meeting 1 Acute leukemia and myelodysplastic syndromes Peter Vandenberghe Centrum Menselijke Erfelijkheid & Afdeling Hematologie, UZ Leuven 11/01/2012 Post-ASH meeting 2 1. Acute myeloid
JAK inhibitors in Phmyeloproliferative
 Disclosures for A Pardanani Research Support/P.I. Employee Consultant Major Stockholder Speakers Bureau Scientific Advisory Board TargeGen, Cytopia/YM BioSciences, PharmaMar None None None None None Presentation
Disclosures for A Pardanani Research Support/P.I. Employee Consultant Major Stockholder Speakers Bureau Scientific Advisory Board TargeGen, Cytopia/YM BioSciences, PharmaMar None None None None None Presentation
Published Ahead of Print on June 22, 2017, as doi: /haematol Copyright 2017 Ferrata Storti Foundation.
 Published Ahead of Print on June 22, 2017, as doi:10.3324/haematol.2017.166173. Copyright 2017 Ferrata Storti Foundation. Molecular analysis of myelodysplastic syndrome with isolated del(5q) reveals a
Published Ahead of Print on June 22, 2017, as doi:10.3324/haematol.2017.166173. Copyright 2017 Ferrata Storti Foundation. Molecular analysis of myelodysplastic syndrome with isolated del(5q) reveals a
Disclosures for Ayalew Tefferi
 Disclosures for Ayalew Tefferi Principal investigator role Employee Consultant Major Stockholder Speakers Bureau Scientific Advisory Board Janssen, Geron, Celgene, Sanofi-Aventis, Gilead Sciences, Incyte
Disclosures for Ayalew Tefferi Principal investigator role Employee Consultant Major Stockholder Speakers Bureau Scientific Advisory Board Janssen, Geron, Celgene, Sanofi-Aventis, Gilead Sciences, Incyte
74y old Female with chronic elevation of Platelet count. August 18, 2005 Faizi Ali, MD Hematopathology Fellow
 74y old Female with chronic elevation of Platelet count August 18, 2005 Faizi Ali, MD Hematopathology Fellow Clinical History Patient is a 74y old otherwise healthy Caucasian female with no major complaint
74y old Female with chronic elevation of Platelet count August 18, 2005 Faizi Ali, MD Hematopathology Fellow Clinical History Patient is a 74y old otherwise healthy Caucasian female with no major complaint
Myelodysplastic/Myeloproliferative Neoplasms (MDS/MPN) Updated
 Myelodysplastic/Myeloproliferative Neoplasms (MDS/MPN) Updated Attilio Orazi, MD, FRCPath. (Engl.) Professor of Pathology and Laboratory Medicine Weill Cornell Medical College/NYP Hospital New York, NY
Myelodysplastic/Myeloproliferative Neoplasms (MDS/MPN) Updated Attilio Orazi, MD, FRCPath. (Engl.) Professor of Pathology and Laboratory Medicine Weill Cornell Medical College/NYP Hospital New York, NY
Test Name Results Units Bio. Ref. Interval. Positive
 LL - LL-ROHINI (NATIONAL REFERENCE 135091534 Age 36 Years Gender Female 1/9/2017 120000AM 1/9/2017 105316AM 2/9/2017 104147AM Ref By Final LEUKEMIA GENETIC ROFILE ANY SIX MARKERS, CR QUALITATIVE AML ETO
LL - LL-ROHINI (NATIONAL REFERENCE 135091534 Age 36 Years Gender Female 1/9/2017 120000AM 1/9/2017 105316AM 2/9/2017 104147AM Ref By Final LEUKEMIA GENETIC ROFILE ANY SIX MARKERS, CR QUALITATIVE AML ETO
Disclosures for Ayalew Tefferi
 Disclosures for Ayalew Tefferi Principal investigator role Employee Consultant Major Stockholder Speakers Bureau Scientific Advisory Board Janssen, Geron, Celgene, Sanofi-Aventis, Gilead Sciences, Incyte
Disclosures for Ayalew Tefferi Principal investigator role Employee Consultant Major Stockholder Speakers Bureau Scientific Advisory Board Janssen, Geron, Celgene, Sanofi-Aventis, Gilead Sciences, Incyte
Myeloproliferative Neoplasms: Diagnosis and Molecular Monitoring in the Era of Target Therapy
 Myeloproliferative Neoplasms: Diagnosis and Molecular Monitoring in the Era of Target Therapy C. Cameron Yin, M.D., Ph.D. Department of Hematopathology UT MD Anderson Cancer Center Disclosures I do not
Myeloproliferative Neoplasms: Diagnosis and Molecular Monitoring in the Era of Target Therapy C. Cameron Yin, M.D., Ph.D. Department of Hematopathology UT MD Anderson Cancer Center Disclosures I do not
Molecular Advances in Hematopathology
 Molecular Advances in Hematopathology HOW MOLECULAR METHODS HAVE CHANGED MY PRACTICE Objectives Understand the importance of cytogenetic/molecular studies in hematolymphoid diseases Know some of the important
Molecular Advances in Hematopathology HOW MOLECULAR METHODS HAVE CHANGED MY PRACTICE Objectives Understand the importance of cytogenetic/molecular studies in hematolymphoid diseases Know some of the important
Template for Reporting Results of Biomarker Testing for Myeloproliferative Neoplasms
 Template for Reporting Results of Biomarker Testing for Myeloproliferative Neoplasms Template web posting date: December 2014 Authors Todd W. Kelley, MD, FCAP University of Utah and ARUP Laboratories,
Template for Reporting Results of Biomarker Testing for Myeloproliferative Neoplasms Template web posting date: December 2014 Authors Todd W. Kelley, MD, FCAP University of Utah and ARUP Laboratories,
MDS/MPN: What it is and How it Should be Treated?
 MDS/MPN: What it is and How it Should be Treated? MDS MPN Rachel Salit, MD Assistant Member Fred Hutchinson Cancer Research Center rsalit@fredhutch.org MDS Founda>on Pa>ent & Family Forum: May 20, 2017
MDS/MPN: What it is and How it Should be Treated? MDS MPN Rachel Salit, MD Assistant Member Fred Hutchinson Cancer Research Center rsalit@fredhutch.org MDS Founda>on Pa>ent & Family Forum: May 20, 2017
