ORIGINAL RESEARCH. With an increased incidence of 13.1 per 1000 inpatients 1,2 and an attributable mortality of 6.1%, 3 in
|
|
|
- Brett Nelson
- 5 years ago
- Views:
Transcription
1 ORIGINAL RESEARCH Toxin Assay Is More Reliable than ICD-9 Data and Less Time-Consuming Than Chart Review for Public Reporting of Clostridium difficile Hospital Case Rates James A. Welker, DO 1* and J.B. Bertumen, MD 2 1 University of Maryland School of Medicine, MedStar Research Institute, Anne Arundel Health System, Baltimore, Maryland; 2 Brown University School of Medicine, Providence, Rhode Island. OBJECTIVE: Clostridium difficile associated disease (CDAD) is common and has a 6.1% mortality. Governmental agencies have recommended surveillance, but reporting increases health care costs. We sought to identify a reliable method of reporting CDAD that will not significantly increase health care costs. METHODS: Patients were identified via database query for International Statistical Classification of Diseases and Related Health Problems, 9th Edition (ICD-9) codes and C. difficile toxin positivity. All identified patients underwent a chart review, which was used to determine the accuracy of the database query methods. Methods of determining whether CDAD was acquired at the reporting institution were studied, and time required to perform each method was measured. RESULTS: The toxin assay reported 96.1% (369/384) of cases and had a positive predictive value of 100%. No difference was found in comparison of the toxin assay case rate of 15.7 per 1000 discharged patients to the rate of 16.3 identified by chart review (P ¼ 0.440; 95% confidence interval [CI], ), whereas the ICD-9 method was found to be significantly different by reporting 116.1% (446/ 384) of cases for a case rate of 19.0 per 1000 discharges (P ¼ 0.001; 95% CI, ). The time for data extraction via the toxin assay method required only 842 minutes, while the chart review method consumed 21,899 minutes. CONCLUSION: A positive C. difficile toxin assay accurately reports the institutional incidence of disease and is more reliable than ICD-9 query. This process can be instituted at a fraction of the cost of the standard chart review, and enables governmental agencies to inexpensively add CDAD to their list of reportable diseases. Journal of Hospital Medicine 2012;7: VC 2011 Society of Hospital Medicine. With an increased incidence of 13.1 per 1000 inpatients 1,2 and an attributable mortality of 6.1%, 3 in 2006 the Canadian government added Clostridium difficile associated disease (CDAD) to its list of reportable diseases. 4 The Centers for Disease Control and Prevention and the Infectious Disease Society of America subsequently created definitions of the disease and recommended surveillance of rates of health care facility associated CDAD, which has been found to double a patient s length of stay and cost of hospitalization. 5 7 While not included in the current list of hospital-acquired conditions for which payment is declined, 8 the Centers for Medicare & Medicaid Services (CMS) has noted CDAD as under consideration for addition to the list, 9 which would require hospital reporting of CDAD rates. This reporting is typically a labor-intensive, medical record review process that *Address for correspondence and reprint requests: James A. Welker, DO, 2001 Medical Parkway, Annapolis, MD 21401; Tel.: ; Fax: ; jimwelker@hotmail.com Additional Supporting Information may be found in the online version of this article. Received: June 16, 2011; Revised: September 16, 2011; Accepted: September 22, Society of Hospital Medicine DOI /jhm.990 Published online in Wiley Online Library (Wileyonlinelibrary.com). increases the cost of delivering health care. Health care institutions have reported spending as much as $21 million per year or $400 per discharged patient on quality improvement. 10 These costs are passed on to payers such as those governed by the CMS that are projected to pay for more than half of all national health spending in Therefore, it is prudent to examine alternatives for determining rates of disease for public reporting and quality improvement initiatives such as infection control and antibiotic stewardship programs. There are 3 methods that can be used for this reporting. First, medical record review has been used for determining the case rate of CDAD by published reports. 1,2 This procedure allows the CMS to define the desired data to be collected, but it is labor-intensive. Second, the use of International Statistical Classification of Diseases and Related Health Problems, 9th Edition (ICD-9) codes from hospital databases offers the speed of database query. However, due to diagnostic and coding errors, it may report inaccurate rates of disease. Previous reports include a universitybased hospital that found a sensitivity of 78% and specificity of 99.7%, 12 whereas a Veterans Administration medical center found nearly two-thirds of patients with CDAD did not have the ICD-9 code for C. difficile infection noted in their database An Official Publication of the Society of Hospital Medicine Journal of Hospital Medicine Vol 7 No 3 March 2012
2 Toxin Assay and ICD-9 for Reporting C. difficile Welker and Bertumen TABLE 1. Diagnostic Criteria Chart Review Criteria for CDAD Pseudomembranous colitis seen during endoscopy OR biopsy or resection with surgical pathology consistent with CDAD If neither of the above are present, then Clostridium difficile toxin or WBC count >25,000 plus one of the following: Diarrhea Fever without other cause Abdominal pain without other cause Colonic ileus without other cause Abbreviations: CDAD, Clostridium difficile associated disease; WBC, white blood cell. Chart Review Criteria for CDAD Obtained While Patient Was Not at Our Hospital One of the above symptoms or objective criteria positive within the first 3 days of hospitalization AND patient was not cared for at our insitution within the last 7 days Chart Review Criteria for CDAD Obtained While Patient Was at Our Hospital Patient was cared for at our institution within 7 days of presentation OR the patient s symptoms were not present upon arrival at the hospital and the symptoms began after the third day of hospitalization Therefore, the accuracy of ICD-9 code at a community hospital that better represents the majority of United States hospitals is needed. The third method of identifying CDAD is the presence of toxin identified in the microbiology laboratory. Although this method offers ease of obtainment, it suffers from potential inaccuracy arising from duplicate patient samples, positive samples in patients who are symptom-free carriers, and patients with CDAD despite having a negative toxin. The same organizations that recommend surveillance for CDAD have identified that there are inadequate data on which to base a decision regarding how to proceed with routine community and hospital surveillance. 6 In the setting of public reporting and nonpayment, the method of identifying CDAD cases must be accurate to ensure fairness, while being inexpensive. To identify the potential value of these less labor-intensive methods of reporting the incidence of CDAD, we evaluated the use of ICD-9 codes and C. difficile toxin to accurately report the incidence of CDAD at a community hospital, as well as the labor hours required for each reporting method. METHODS Patients >18 years of age and potentially having CDAD were identified via database queries for ICD-9 codes and positive C. difficile toxin assays at our institution from November 1, 2006, through August 31, Our institution is a 379-bed university-affiliated community teaching hospital in a socio-economically diverse area of Baltimore with approximately 110,000 emergency department visits, 30,000 discharges, and 110,000 inpatient days each year that uses a handwritten paper chart for all provider orders and patient documentation. The C. difficile toxin assay method used during the study period was an enzyme immunoassay that detects both toxins A and B (Meridan Bioscience, Cincinnati, OH). ICD-9 codes were queried in the CareScience database(premier, Inc; Charlotte, NC), while C. difficile toxin was queried via the hospital laboratory database. All identified patients underwent a medical record review to confirm the diagnosis of CDAD and the patient s location at the time of disease acquisition. To eliminate duplicate reporting of a single episode of disease, all duplicate patient visits with a diagnosis of CDAD within 30 days were removed. Our diagnostic criteria (Table 1) used the recommended criteria of a combination of symptoms and positive toxin assay, visualization of pseudomembranes on colonoscopy or pathology-proven CDAD. 6 Based on the presence of CDAD in 25% of patients with a white blood cell count >30,000 and the recommendation that CDAD be considered in all patients with a white blood cell count >15,000, we included the criterion of clinical findings with severe leukocytosis to identify patients with toxin-negative CDAD. 14 Patients were identified by querying the CareScience database for patients having an ICD-9 code of Duplicate cases for the same patient within 30 days were removed. These cases were compared with the cases of CDAD determined by the medical record review for analysis. Patients were identified via positive C. difficile toxin by querying the microbiology laboratory database. A query of all patients with a positive C. difficile toxin was performed. Duplicate samples for the same patient within 30 days were removed. Patients are considered to be outside the hospital when CDAD was acquired if their toxin was positive within the first 3 days of arrival to the hospital, and they were not hospitalized at our institution within 1 week before arrival. All other cases were considered to be acquired while in our hospital. The number of patients with a toxin-positive stool sample was compared with the medical record review. Recognizing that it is unrealistic to review the medical records of the 23,495 discharges during the 10- month study period, a random sample review of 500 charts not included in our CDAD-identified patient list was performed to identify the rate at which CDAD patients failed to be identified by our methods. From a list of discharges during the study period, as long as they were not in our study population, every An Official Publication of the Society of Hospital Medicine Journal of Hospital Medicine Vol 7 No 3 March
3 Welker and Bertumen Toxin Assay and ICD-9 for Reporting C. difficile TABLE 2. Comparison of ICD-9 and Toxin Assay with Chart Review ICD-9 Toxin Assay Chart Review No. of patients identified Case rate per 1000 discharges* % confidence interval NA Case rate compared with chart review, P P¼ P ¼ NA CDAD rate reported compared with chart review 116.1% 96.1% NA Accuracy 83.9% 96.1% NA PPV 74.9% 100% NA Portion of cases acquired at our hospital, % NA 48.2% 40.6% Portion of cases acquired at our hospital, P NA P ¼ NA Minutes consumed for data collection ,899 Estimated annual cost per hospital $ $ $16, Abbreviations: CDAD CDAD, Clostridium difficile associated disease; ICD-9, International Statistical Classification of Diseases and Related Health Problems, 9th Edition; NA, not available; PPV, positive predictive value. * Total discharges during study period: 23,495. Assumes employee cost of $30/hr þ benefits. thirtieth patient was selected for review up to a total of 500 patient charts. Then, the identified case rate was used to predict the prevalence of disease at our institution during the study period. Sample selection was based on the absence of previous studies evaluating the performance of C. difficile toxin assay, and the desire to have the same assay used during the entire study. The lack of data from which to perform sample size determination would result in an inaccurate estimate. Therefore, we chose a period that offered the maximum sample size, during which a single assay was used at our institution. Compared with medical record review, the accuracy and positive predictive values of the ICD-9 code and positive stool toxin methods to identify the cases of CDAD were compared. A true positive was when the ICD-9 or laboratory query method identified a patient who had CDAD based on chart review; a false positive was when the ICD-9 or laboratory query method identified a patient who did not have CDAD based on chart review. Rates of over- or underdiagnosis, case rates, and acquisition location were determined. Statistical analysis of the case rates and acquisition location were performed via chisquare test. The time to perform these queries was collected with accuracy to the minute. RESULTS Of the 23,495 discharges during the study period, the combination of ICD-9 and C. difficile toxin assay identified a total cohort of 496 patients, 319 of whom were identified by both the ICD-9 method and the toxin assay, 50 of whom were identified only by the toxin assay, and 127 of whom were identified only by the ICD-9 method. Chart review confirmed the presence of CDAD in 384 of these 496 cases, for a case rate of 16.3 per 1000 discharged patients (Table 2). The diagnostic criteria for each of these confirmed cases are listed in Table 3. Of the 384 confirmed TABLE 3. Diagnostic Criteria Confirming CDAD in 384 Cases Criterion No. of Cases Case Rate* Endoscopy 2 0.5% Surgical pathology 9 2.3% Positive toxin and diarrhea % WBC >25.0 and diarrhea % WBC >25.0 and fever without other source 9 2.3% WBC >25.0 and unexplained abdominal pain % WBC >25.0 and colonic ileus 2 0.5% Abbreviations: CDAD, Clostridium difficile associated disease; WBC, white blood cell count. * Case rate sum is >100% because some patients met more than 1 criterion. CDAD cases, 319 were identified by both the ICD-9 and toxin assay, 50 were identified only by the toxin assay, and 15 were identified only by the ICD-9 query. Of the 50 cases identified by the toxin assay that were not identified via the ICD-9 method, all 50 (100%) were confirmed to have CDAD by chart review. In contrast, of the 127 cases identified via the ICD-9 method that were not found via the toxin assay, only 15 (11.8%) were confirmed to have CDAD by chart review (Figure 1). Of these 384 cases, 369 were identified via the toxin assay for a case rate of 15.7 per 1000 patient discharges, which was not found to be different from the rate of 16.3 determined by chart review (P ¼ 0.440; 95% confidence interval [CI], ). Compared with chart review, the toxin assay reported 96.1% (369/384) of cases. Chart review demonstrated that every patient who had a positive toxin assay met the diagnostic criteria for CDAD for a positive predictive value (PPV) of 100%. The ICD-9 method identified 446 patients thought to have CDAD, 334 of whom were confirmed by chart review for a PPV of 74.9% (334/446). Compared with chart review, the ICD-9 method reported 116.1% (446/384) of cases for a case rate of 19.0 per 1000 discharges and was significantly different than the rate of 16.3 reported by chart review (P ¼ 0.001; 95% CI, ). Chart review identified 156 of 384 (40.6%) patients who acquired CDAD while at our hospital and 228 of 384 (59.4%) who acquired it elsewhere. In comparison, the toxin assay criteria identified 369 cases of CDAD, of which 48.2% (178/369) were acquired while at our hospital and 51.8% (191/369) were acquired elsewhere (P ¼ 0.003). The time for data extraction via these 3 methods differed greatly. The ICD-9 method only consumed 312 minutes and the toxin assay method 842 minutes, whereas the chart review method consumed 21,899 minutes. These times reported include the database query and data analysis for the ICD-9 and toxin assay methods, while it includes the database query and list generation along with the manual chart review and data analysis for the chart review method. The review 172 An Official Publication of the Society of Hospital Medicine Journal of Hospital Medicine Vol 7 No 3 March 2012
4 Toxin Assay and ICD-9 for Reporting C. difficile Welker and Bertumen FIG. 1. Source of patient identification and confirmation. of the random sample of patients believed not to have CDAD was not included in any of the reported times. Chart review on a random sample of 500 patients not previously identified for review found no additional cases of CDAD. DISCUSSION Our study demonstrates that use of positive C. difficile toxin assay data from the microbiology laboratory alone is an efficient method of identifying patients with CDAD. This method only consumed 842 minutes versus 21,899 minutes consumed by the chart review method. The C. difficile toxin method reduces the workforce required to collect and analyze this data, but more importantly, it was found to be reliable by reporting an institutional case rate that is similar to that of chart review. In contrast, the ICD-9 method was efficient but less reliable. It only consumed 312 minutes, but it overreported the institutional rate of CDAD by 16.1%, had a PPV of only 74.9%, and of those patients who were identified by ICD-9 but not toxin assay, only 11.8% actually had CDAD. This finding is in conflict with the previously noted underreporting of this method. 13,15 We believe this difference to be associated with institutional differences, because previous reports originated from a veterans hospital and an academic medical center, and previous authors have failed to use predefined diagnostic criteria and a complete chart review to confirm cases of CDAD. Similar to our study, an academic medical center identified that listing of CDAD in a patient s medical history in their chart was associated with a false-positive ICD-9 code for CDAD. 15 This observation appears to bring clarity to one of the causes of the variance of ICD-9 code accuracy between institutions. Institutions seem to vary on the method of attaching diagnoses to the patient s final hospital record. Some institutions include only what is listed as an active problem, whereas others list diagnoses listed in the chart as previous problems and those listed as a potential diagnosis without confirmation. Another potential cause of the ICD-9 inaccuracy is the potential of clinicians to diagnose and treat a patient for CDAD in the absence of the diagnostic criteria used for chart review. Physician practices such as these are known to vary between institutions leading to a variance in the ICD- 9 code accuracy. In total, 15 cases of CDAD were identified in the absence of a positive toxin assay, and of these, 12 cases were identified using leukocytosis-based criteria (Table 4). This resulted in 3.1% of our cases being toxin-negative, based on leukocytosis criteria, and is lower than the previously identified 35% of cases. 14 Because it was the only assay available at the time, this previous research used a toxin A only assay, which is more likely to have false-negative results than the toxin A/B assay used at our institution during the study period. The investigators also required all toxin-negative patients to have been recently treated with antibiotics. Based on the increasing rates of community-acquired CDAD, including those that are antibiotic-naïve patients, we felt a history of antibiotic exposure was no longer a prerequisite for CDAD and thus excluded it from our diagnostic criteria. 16,17 Based on these differences, we feel our results are likely an accurate reflection of the number of cases identified by ICD-9 query in the absence of toxin positivity. However, concerns should be further alleviated through the realization that nonuse of this strategy improves the accuracy of the toxin assay method, while reducing the accuracy of the ICD-9 method and thereby strengthening the validity of our conclusions. Mathematically, this would result in 369 of 369 patients identified by toxin assay and 369 patients identified by the ICD-9 method. This would reduce the case rate of the chart review method to the same 15.7 of the toxin assay, while the ICD-9 method would remain at We considered whether the toxin assay method may overestimate the number of cases due to a C. difficile asymptomatic carrier rate as high as 50% of hospitalized patients. 6 However, we found no difference in the case rate when compared with that of chart review, and there were no false-positive cases. We believe this is attributable to the 30-day window that was used to identify a single episode of CDAD and the absence of toxin assays being performed on asymptomatic individuals. To avoid overrepresentation of the actual number of CDAD cases, we chose to label all repeat positive toxins and repeat hospitalizations within 30 days as a single episode of CDAD. This was based on the identification that 56% of patients remained toxin-positive An Official Publication of the Society of Hospital Medicine Journal of Hospital Medicine Vol 7 No 3 March
5 Welker and Bertumen Toxin Assay and ICD-9 for Reporting C. difficile 2 6 weeks after adequate treatment for CDAD. 18 The enzyme-linked immunosorbent assay (ELISA) method used by our laboratory is the method used to report 94% of CDAD cases in a national point prevalence study that collected data from United States acute care hospitals with representation from 47 states 1,6,19 (Table 5). This use of ELISA in the majority of United States hospitals suggests that our data can be extrapolated for use throughout the United States. While laboratories are increasing their use of 2-step algorithms involving glutamate dehydrogenase antigen assay followed by cytotoxin neutralization, and more recently beginning the use of polymerase chain reaction assays, both of these methods have been found to increase the accuracy of detecting C. difficile compared with ELISA. 20 Therefore, as laboratories evolve to use more accurate assays to detect CDAD, the methods described herein will be expected to increase in reliability. The toxin assay methodology used to determine the rate of CDAD cases acquired while the patient was at our hospital overreported these cases. Based on this result, identification of individual cases of CDAD that are obtained at a specific hospital would continue to require manual chart review. This expensive method may be avoided by instead choosing to use institutional case rates for reporting, monitoring, and incentivizing hospitals. However, a discussion of the methods of this approach and its confluence with our societal goal to move toward Accountable Care Organizations is beyond the scope of this discussion section. TABLE 4. Diagnostic Criteria in 15 CDAD Cases with Negative Toxin Criterion Endoscopy 0 Surgical pathology 3 WBC >25.0 and diarrhea 10 WBC >25.0 and fever without other source 1 WBC >25.0 and unexplained abdominal pain 3 WBC >25.0 and colonic ileus 1 No. of Cases* Abbreviations: CDAD, Clostridium difficile associated disease; WBC, white blood cell count. * Sum is >15 because some patients met more than 1 criterion. Although it appears that we identified all cases of CDAD occurring at our institution, a limitation of this study is its inability to review all charts during the 10- month study period. We used a combination of ICD-9 and positive C. difficile toxin assay data to identify all possible cases of C. difficile. The current approach to case identification for reported hospital conditions is limited to an ICD-9 database query. This query is followed by chart review to collect data for hospital performance that is published in locations such as www. hospitalcompare.hhs.gov. Although our approach expands upon this current method of patient identification, it may still fail to identify some cases. To investigate the reliability of our strategy, we performed a chart review on a random sample of patients not previously identified for review. In this portion of the study, 500 charts were reviewed, and no cases of CDAD were found. Considering the identified case rates of 16 to 19 per 1000 discharges, one would expect as many as 10 cases of CDAD to be identified if our methods were unreliable. The identification of zero cases supports our methods as identifying all cases of CDAD during this period. Considering the hurdle of 23,495 charts for a complete review and the inability to identify an adequate number of CDAD cases if 100% chart review over a shorter period was the selected strategy, our study design is the only realistic method of studying this subject. Increased automation is expected in the future of reporting. The Centers for Disease Control and Prevention found increased rates of disease reporting and increased accuracy when reporting is electronically automated via their software system, Electronic Support for Public Health, which is designed to communicate with and perform automated data queries on providers electronic medical records. 21 While use of this model is creeping into the health system for reporting to public health authorities, 22 universal hospital electronic medical record implementation and full connectivity with such reporting systems is many years from fruition. In addition to its practical use for reporting CDAD in our current health system, our work easily transitions into automated reporting within an electronically integrated health system once achieved. TABLE 5. Clostridium difficile Testing Methods Method Sensitivity Specificity Cost Ease of Performance Typical Results Reporting Culture Gold standard NA $$$$ Difficult Days Slow turnaround time; is the standard upon which other test methods are based, but not all organisms are toxin-producing Cell cytotoxicity assay 67% 100% Gold standard $$$ Intermediate Next day Is the standard upon which other test methods are based to identify toxin-producing stains of Clostridium difficile EIA for toxin A/B 63% 94% 75% 100% $ Easy Same day Used by >90% of laboratories in the United States EIA for detection of Clostridium difficile common antigen (GDH) 85% 95% 89% 99% $ Easy Same day Provides no information regarding the toxigenicity of the isolate, typically used in combination with cell cytotoxicity assay to identify toxin-producing strains Polymerase chain reaction 96% 100% 88% 91% $$ Intermediate Same day More data are needed before recommendation for routine testing Abbreviations: EIA, enzyme immunoassay; NA, not available. Notes 174 An Official Publication of the Society of Hospital Medicine Journal of Hospital Medicine Vol 7 No 3 March 2012
6 Toxin Assay and ICD-9 for Reporting C. difficile Welker and Bertumen In conclusion, ICD-9 data were found to be unreliable, and consideration must be given to cessation of their use for CDAD case rate research and reporting. Use of a positive C. difficile toxin assay accurately reports the institutional incidence of disease, can be used by individual institutions to self-monitor case rates or by the government to determine regionally acceptable intuitional rates of CDAD on which incentives and penalties can be based, and will increase in efficiency as reporting continues to be automated. This process can be instituted at a fraction of the cost of the standard chart review that is currently used for most reporting. Disclosure: The authors have no conflicts of interest to report. References 1. Jarvis WR, Schlosser J, Jarvis AA, Chinn RY. National point prevalence of Clostridium difficile in US health care facility inpatients, Am J Infect Control. 2009;37: Archibald LK, Banerjee SN, Jarvis WR. Secular trends in hospitalacquired Clostridium difficile disease in the United States, J Infect Dis. 2004;189: Kenneally C, Rosini JM, Skrupky LP, et al. Analysis of 30-day mortality for Clostridium difficile-associated disease in the ICU setting. Chest. 2007;132: Doherty JA. Final report and recommendations from the National Notifiable Diseases Working Group. Can Commun Dis Rep. 2006; 32: McDonald LC, Coignard B, Dubberke E, et al. Recommendations for surveillance of Clostridium difficile-associated disease. Infect Control Hosp Epidemiol. 2007;28: Cohen SH, Gerding DN, Johnson S, et al. Clinical practice guidelines for Clostridium difficile infection in adults: 2010 update by the society for healthcare epidemiology of America (SHEA) and the infectious diseases society of America (IDSA). Infect Control Hosp Epidemiol. 2010;31: Dubberke ER, Wertheimer AI. Review of current literature on the economic burden of Clostridium difficile infection. Infect Control Hosp Epidemiol. 2009;30: Centers for Medicare & Medicaid Services. Hospital-acquired conditions. Available at: pital-acquired_conditions.asp#topofpage. Accessed April 10, Centers for Medicare & Medicaid Services. CMS proposes to expand quality program for hospital inpatient services in FY Available at: Accessed April 10, Chen LM, Rein MS, Bates DW. Costs of quality improvement: a survey of four acute care hospitals. Jt Comm J Qual Patient Saf. 2009; 35: Sisko A, Truffer C, Smith S, et al. Health spending projections through 2018: recession effects add uncertainty to the outlook. Health Aff (Millwood). 2009;28:w346 w Dubberke ER, Reske KA, McDonald LC, Fraser VJ. ICD-9 codes and surveillance for Clostridium difficile-associated disease. Emerg Infect Dis. 2006;12: McFarland LV, Clarridge JE, Beneda HW, Raugi GJ. Fluoroquinolone use and risk factors for Clostridium difficile-associated disease within a Veterans Administration health care system. Clin Infect Dis. 2007;45: Wanahita A, Goldsmith EA, Musher DM. Conditions associated with leukocytosis in a tertiary care hospital, with particular attention to the role of infection caused by clostridium difficile. Clin Infect Dis. 2002;34: Scheurer DB, Hicks LS, Cook EF, Schnipper JL. Accuracy of ICD-9 coding for Clostridium difficile infections: a retrospective cohort. Epidemiol Infect. 2007;135: McFarland LV, Beneda HW, Clarridge JE, Raugi GJ. Implications of the changing face of Clostridium difficile disease for health care practitioners. Am J Infect Control. 2007;35: Pituch H. Clostridium difficile is no longer just a nosocomial infection or an infection of adults. Int J Antimicrob Agents. 2009;33(suppl 1): S42 S Fekety R, Silva J, Kauffman C, Buggy B, Deery HG. Treatment of antibiotic-associated Clostridium difficile colitis with oral vancomycin: comparison of two dosage regimens. Am J Med. 1989;86: Knetsch CW, Bakker D, de Boer RF, et al. Comparison of realtime PCR techniques to cytotoxigenic culture methods for diagnosing Clostridium difficile infection. J Clin Microbiol. 2011;49: Kvach EJ, Ferguson D, Riska PF, Landry ML. Comparison of BD GeneOhm Cdiff real-time PCR assay with a two-step algorithm and a toxin A/B enzyme-linked immunosorbent assay for diagnosis of toxigenic Clostridium difficile infection. J Clin Microbiol. 2010;48: Centers for Disease Control and Prevention. Automated detection and reporting of notifiable diseases using electronic medical records versus passive surveillance Massachusetts, June 2006-July MMWR Morb Mortal Wkly Rep. 2008;57: Rajeev D, Staes CJ, Evans RS, et al. Development of an electronic public health case report using HL7 v2.5 to meet public health needs. J Am Med Inform Assoc. 2010;17: An Official Publication of the Society of Hospital Medicine Journal of Hospital Medicine Vol 7 No 3 March
EDUCATIONAL COMMENTARY CLOSTRIDIUM DIFFICILE UPDATE
 EDUCATIONAL COMMENTARY CLOSTRIDIUM DIFFICILE UPDATE Educational commentary is provided through our affiliation with the American Society for Clinical Pathology (ASCP). To obtain FREE CME/CMLE credits click
EDUCATIONAL COMMENTARY CLOSTRIDIUM DIFFICILE UPDATE Educational commentary is provided through our affiliation with the American Society for Clinical Pathology (ASCP). To obtain FREE CME/CMLE credits click
DUKE ANTIMICROBIAL STEWARDSHIP OUTREACH NETWORK (DASON) Antimicrobial Stewardship News. Volume 3, Number 6, June 2015
 DUKE ANTIMICROBIAL STEWARDSHIP OUTREACH NETWORK (DASON) Antimicrobial Stewardship News Volume 3, Number 6, June 2015 Diagnostic Testing for Clostridium difficile Infection Background Clostridium difficile
DUKE ANTIMICROBIAL STEWARDSHIP OUTREACH NETWORK (DASON) Antimicrobial Stewardship News Volume 3, Number 6, June 2015 Diagnostic Testing for Clostridium difficile Infection Background Clostridium difficile
ABSTRACT PURPOSE METHODS
 ABSTRACT PURPOSE The purpose of this study was to characterize the CDI population at this institution according to known risk factors and to examine the effect of appropriate evidence-based treatment selection
ABSTRACT PURPOSE The purpose of this study was to characterize the CDI population at this institution according to known risk factors and to examine the effect of appropriate evidence-based treatment selection
C. DIFF QUIK CHEK COMPLETE. Get the complete diagnostic picture with just one test. TAP HERE TO SEE THE NEXT PAGE
 C. DIFF QUIK CHEK COMPLETE Get the complete diagnostic picture with just one test. TAP HERE TO SEE THE PAGE CLINICIAN Would actionable C. difficile test results in less than 30 minutes improve patient
C. DIFF QUIK CHEK COMPLETE Get the complete diagnostic picture with just one test. TAP HERE TO SEE THE PAGE CLINICIAN Would actionable C. difficile test results in less than 30 minutes improve patient
Atypical Presentation of Clostridium Difficille Infection (CDI).
 Article ID: WMC004648 ISSN 2046-1690 Atypical Presentation of Clostridium Difficille Infection (CDI). Peer review status: No Corresponding Author: Dr. Syed A Gardezi, CT1, Medicine,NevillHall Hospital
Article ID: WMC004648 ISSN 2046-1690 Atypical Presentation of Clostridium Difficille Infection (CDI). Peer review status: No Corresponding Author: Dr. Syed A Gardezi, CT1, Medicine,NevillHall Hospital
Diagnosis, Management, and Prevention of Clostridium difficile infection in Long-Term Care Facilities: A Review
 Diagnosis, Management, and Prevention of Clostridium difficile infection in Long-Term Care Facilities: A Review October 18, 2010 James Kahn and Carolyn Kenney, MSIV Overview Burden of disease associated
Diagnosis, Management, and Prevention of Clostridium difficile infection in Long-Term Care Facilities: A Review October 18, 2010 James Kahn and Carolyn Kenney, MSIV Overview Burden of disease associated
L. Clifford McDonald, MD. Senior Advisor for Science and Integrity September 16, 2015
 Controversies and Current Issues in Diagnosis, Surveillance, and Treatment of Clostridium difficile infeciton L. Clifford McDonald, MD Senior Advisor for Science and Integrity September 16, 2015 Division
Controversies and Current Issues in Diagnosis, Surveillance, and Treatment of Clostridium difficile infeciton L. Clifford McDonald, MD Senior Advisor for Science and Integrity September 16, 2015 Division
Clostridium difficile infection surveillance: Applying the case definition
 Clostridium difficile infection surveillance: Applying the case definition PICNet Conference March 3 rd 2016 Presented by: Tara Leigh Donovan, MSc Managing Consultant (Former Epidemiologist) 1 Disclaimer
Clostridium difficile infection surveillance: Applying the case definition PICNet Conference March 3 rd 2016 Presented by: Tara Leigh Donovan, MSc Managing Consultant (Former Epidemiologist) 1 Disclaimer
C. Difficile Testing Protocol
 C. Difficile Testing Protocol Caroline Donovan, RN, BSN, ONC- Infection Control Practitioner Abegail Pangan, RN, MSN, CIC- Infection Control Practitioner U.S. NEWS & WORLD REPORT 2017 2018 RANKINGS Acute
C. Difficile Testing Protocol Caroline Donovan, RN, BSN, ONC- Infection Control Practitioner Abegail Pangan, RN, MSN, CIC- Infection Control Practitioner U.S. NEWS & WORLD REPORT 2017 2018 RANKINGS Acute
Modern approach to Clostridium Difficile Infection
 Modern approach to Clostridium Difficile Infection Pseudomembranous Colitis: Principles for diagnosis and treatment Aggelos Stefos Internist, Infectious diseases Specialist Department of Medicine and Research
Modern approach to Clostridium Difficile Infection Pseudomembranous Colitis: Principles for diagnosis and treatment Aggelos Stefos Internist, Infectious diseases Specialist Department of Medicine and Research
Stony Brook Adult Clostridium difficile Management Guidelines. Discontinue all unnecessary antibiotics
 Stony Brook Adult Clostridium difficile Management Guidelines Summary: Use of the C Diff Infection (CDI) PowerPlan (Adult) Required Patient with clinical findings suggestive of Clostridium difficile infection
Stony Brook Adult Clostridium difficile Management Guidelines Summary: Use of the C Diff Infection (CDI) PowerPlan (Adult) Required Patient with clinical findings suggestive of Clostridium difficile infection
Clostridium difficile Infection (CDI) Management Guideline
 Clostridium difficile Infection (CDI) Management Guideline Do not test all patients with loose or watery stools for CDI o CDI is responsible for
Clostridium difficile Infection (CDI) Management Guideline Do not test all patients with loose or watery stools for CDI o CDI is responsible for
TITLE OF PRESENTATION
 TITLE OF PRESENTATION Can C. Subtitle difficile of Infection Presentation Rates Be Used to Judge Prevention Success for Hospitals? David P. Calfee, MD, MS Associate Professor of Medicine and Public Health
TITLE OF PRESENTATION Can C. Subtitle difficile of Infection Presentation Rates Be Used to Judge Prevention Success for Hospitals? David P. Calfee, MD, MS Associate Professor of Medicine and Public Health
Long-Term Care Updates
 Long-Term Care Updates April 2018 By Austin Smith, PharmD Candidate and Lindsay Slowiczek, PharmD is the most common healthcare-acquired infection (HAI) in the United States. 1,2 A 2014 prevalence survey
Long-Term Care Updates April 2018 By Austin Smith, PharmD Candidate and Lindsay Slowiczek, PharmD is the most common healthcare-acquired infection (HAI) in the United States. 1,2 A 2014 prevalence survey
DETECTION OF TOXIGENIC CLOSTRIDIUM DIFFICILE
 CLINICAL GUIDELINES For use with the UnitedHealthcare Laboratory Benefit Management Program, administered by BeaconLBS DETECTION OF TOXIGENIC CLOSTRIDIUM DIFFICILE Policy Number: PDS 021 Effective Date:
CLINICAL GUIDELINES For use with the UnitedHealthcare Laboratory Benefit Management Program, administered by BeaconLBS DETECTION OF TOXIGENIC CLOSTRIDIUM DIFFICILE Policy Number: PDS 021 Effective Date:
Clostridium Difficile Infection: Applying New Treatment Guidelines and Strategies to Reduce Recurrence Rate
 Clostridium Difficile Infection: Applying New Treatment Guidelines and Strategies to Reduce Recurrence Rate Objectives Summarize the changing epidemiology and demographics of patients at risk for Clostridium
Clostridium Difficile Infection: Applying New Treatment Guidelines and Strategies to Reduce Recurrence Rate Objectives Summarize the changing epidemiology and demographics of patients at risk for Clostridium
Clostridium difficile Infection: Diagnosis and Management
 Clostridium difficile Infection: Diagnosis and Management Brian Viviano D.O. Case study 42 year old female with history of essential hypertension and COPD presents to ED complaining of 24 hours of intractable,
Clostridium difficile Infection: Diagnosis and Management Brian Viviano D.O. Case study 42 year old female with history of essential hypertension and COPD presents to ED complaining of 24 hours of intractable,
ENGLISH FOR PROFESSIONAL PURPOSES UNIT 3 HOW TO DEAL WITH CLOSTRIDIUM DIFFICILE
 ENGLISH FOR PROFESSIONAL PURPOSES UNIT 3 HOW TO DEAL WITH CLOSTRIDIUM DIFFICILE The diagnosis of CDI should be based on a combination of clinical and laboratory findings. A case definition for the usual
ENGLISH FOR PROFESSIONAL PURPOSES UNIT 3 HOW TO DEAL WITH CLOSTRIDIUM DIFFICILE The diagnosis of CDI should be based on a combination of clinical and laboratory findings. A case definition for the usual
Learning Objectives. A Critical Analysis of Recent Fidaxomicin & Nesiritide Trials. Fidaxomicin vs. Vancomycin. Outline 1/3/2012
 Learning Objectives A Critical Analysis of Recent Fidaxomicin & Nesiritide Trials Amy E. Lodolce, PharmD, BCPS Assistant Director, Drug Information Group UIC College of Pharmacy Describe the methods and
Learning Objectives A Critical Analysis of Recent Fidaxomicin & Nesiritide Trials Amy E. Lodolce, PharmD, BCPS Assistant Director, Drug Information Group UIC College of Pharmacy Describe the methods and
The Bristol Stool Scale and Its Relationship to Clostridium difficile Infection
 JCM Accepts, published online ahead of print on 16 July 2014 J. Clin. Microbiol. doi:10.1128/jcm.01303-14 Copyright 2014, American Society for Microbiology. All Rights Reserved. 1 The Bristol Stool Scale
JCM Accepts, published online ahead of print on 16 July 2014 J. Clin. Microbiol. doi:10.1128/jcm.01303-14 Copyright 2014, American Society for Microbiology. All Rights Reserved. 1 The Bristol Stool Scale
Objectives Clostridium difficile Infections, So Many Tests, Which One to Choose?
 Objectives Clostridium difficile Infections, So Many Tests, Which One to Choose? March 9, 0 http://www.slh.wisc.edu/outreach-data/event-detail.php?id=03 Raymond P. Podzorski, Ph.D., D(ABMM) Clinical Microbiologist
Objectives Clostridium difficile Infections, So Many Tests, Which One to Choose? March 9, 0 http://www.slh.wisc.edu/outreach-data/event-detail.php?id=03 Raymond P. Podzorski, Ph.D., D(ABMM) Clinical Microbiologist
ACG Clinical Guideline: Diagnosis, Treatment, and Prevention of Clostridium difficile Infections
 ACG Clinical Guideline: Diagnosis, Treatment, and Prevention of Clostridium difficile Infections Christina M. Surawicz, MD 1, Lawrence J. Brandt, MD 2, David G. Binion, MD 3, Ashwin N. Ananthakrishnan,
ACG Clinical Guideline: Diagnosis, Treatment, and Prevention of Clostridium difficile Infections Christina M. Surawicz, MD 1, Lawrence J. Brandt, MD 2, David G. Binion, MD 3, Ashwin N. Ananthakrishnan,
3/23/2012. Impact of Laboratory Testing on Detection and Treatment of Healthcare Associated Infection: The Case of CDI
 Impact of Laboratory Testing on Detection and Treatment of Healthcare Associated Infection: The Case of CDI L. Clifford McDonald, MD Lance R. Peterson, MD March 27, 2012 1 Potential COI L. Clifford McDonald,
Impact of Laboratory Testing on Detection and Treatment of Healthcare Associated Infection: The Case of CDI L. Clifford McDonald, MD Lance R. Peterson, MD March 27, 2012 1 Potential COI L. Clifford McDonald,
Clostridium difficile Infection (CDI) Guideline Update:
 Clostridium difficile Infection (CDI) Guideline Update: Understanding the Data Behind the Recommendations Erik R. Dubberke, MD, MSPH A Webinar for HealthTrust Members Professor of Medicine September 24,
Clostridium difficile Infection (CDI) Guideline Update: Understanding the Data Behind the Recommendations Erik R. Dubberke, MD, MSPH A Webinar for HealthTrust Members Professor of Medicine September 24,
March 3, To: Hospitals, Long Term Care Facilities, and Local Health Departments
 March 3, 2010 To: Hospitals, Long Term Care Facilities, and Local Health Departments From: NYSDOH Bureau of Healthcare Associated Infections HEALTH ADVISORY: GUIDANCE FOR PREVENTION AND CONTROL OF HEALTHCARE
March 3, 2010 To: Hospitals, Long Term Care Facilities, and Local Health Departments From: NYSDOH Bureau of Healthcare Associated Infections HEALTH ADVISORY: GUIDANCE FOR PREVENTION AND CONTROL OF HEALTHCARE
Does Extending Clostridium Difficile Treatment In Patients Who Are Receiving Concomitant Antibiotics Reduce The Rate Of Relapse?
 ISPUB.COM The Internet Journal of Infectious Diseases Volume 15 Number 1 Does Extending Clostridium Difficile Treatment In Patients Who Are Receiving Concomitant Antibiotics Reduce The Rate Of Relapse?
ISPUB.COM The Internet Journal of Infectious Diseases Volume 15 Number 1 Does Extending Clostridium Difficile Treatment In Patients Who Are Receiving Concomitant Antibiotics Reduce The Rate Of Relapse?
JMSCR Vol 05 Issue 07 Page July 2017
 www.jmscr.igmpublication.org Impact Factor 5.84 Index Copernicus Value: 83.27 ISSN (e)-2347-176x ISSN (p) 2455-0450 DOI: https://dx.doi.org/10.18535/jmscr/v5i7.15 Rapid Diagnosis of Toxigenic Clostridium
www.jmscr.igmpublication.org Impact Factor 5.84 Index Copernicus Value: 83.27 ISSN (e)-2347-176x ISSN (p) 2455-0450 DOI: https://dx.doi.org/10.18535/jmscr/v5i7.15 Rapid Diagnosis of Toxigenic Clostridium
The Epidemiology of Clostridium difficile Infection in Children: A Population-Based Study
 Clinical Infectious Diseases Advance Access published March 5, 2013 MAJOR ARTICLE The Epidemiology of Clostridium difficile Infection in Children: A Population-Based Study Sahil Khanna, 1 Larry M. Baddour,
Clinical Infectious Diseases Advance Access published March 5, 2013 MAJOR ARTICLE The Epidemiology of Clostridium difficile Infection in Children: A Population-Based Study Sahil Khanna, 1 Larry M. Baddour,
ADJUVANT TIGECYCLINE FOR SEVERE CLOSTRIDIUM DIFFICILE-ASSOCIATED DIARRHEA
 ADJUVANT TIGECYCLINE FOR SEVERE CLOSTRIDIUM DIFFICILE-ASSOCIATED DIARRHEA Candace Marr, DO Catholic Health System University at Buffalo, NY Kevin Shiley, MD Catholic Health System Buffalo, NY FINANCIAL
ADJUVANT TIGECYCLINE FOR SEVERE CLOSTRIDIUM DIFFICILE-ASSOCIATED DIARRHEA Candace Marr, DO Catholic Health System University at Buffalo, NY Kevin Shiley, MD Catholic Health System Buffalo, NY FINANCIAL
A Pharmacist Perspective
 Leveraging Technology to Reduce CDI A Pharmacist Perspective Ed Eiland, Pharm.D., MBA, BCPS (AQ-ID) Clinical Practice and Business Supervisor Huntsville Hospital System Huntsville Hospital 881 licensed
Leveraging Technology to Reduce CDI A Pharmacist Perspective Ed Eiland, Pharm.D., MBA, BCPS (AQ-ID) Clinical Practice and Business Supervisor Huntsville Hospital System Huntsville Hospital 881 licensed
Pros and Cons of Alternative Diagnostic Testing Strategies for C. difficile Infection
 Pros and Cons of Alternative Diagnostic Testing Strategies for C. difficile Infection Christopher R. Polage, MD, MAS Associate Professor of Pathology and Infectious Diseases UC Davis Disclosures Test materials
Pros and Cons of Alternative Diagnostic Testing Strategies for C. difficile Infection Christopher R. Polage, MD, MAS Associate Professor of Pathology and Infectious Diseases UC Davis Disclosures Test materials
Clostridium difficile: An Overview
 Clostridium difficile: An Overview CDI Webinar July 11, 2017 PUBLIC HEALTH DIVISION Acute and Communicable Disease Prevention Section Outline Background Microbiology Burden Pathogenesis Diagnostic testing
Clostridium difficile: An Overview CDI Webinar July 11, 2017 PUBLIC HEALTH DIVISION Acute and Communicable Disease Prevention Section Outline Background Microbiology Burden Pathogenesis Diagnostic testing
Clinical Infectious Diseases Advance Access published December 7, 2012
 Clinical Infectious Diseases Advance Access published December 7, 2012 1 Physician Attitudes Towards the Use of Fecal Transplantation for Recurrent Clostridium Difficile Infection in a Large Metropolitan
Clinical Infectious Diseases Advance Access published December 7, 2012 1 Physician Attitudes Towards the Use of Fecal Transplantation for Recurrent Clostridium Difficile Infection in a Large Metropolitan
Clostridium difficile (C difficile)
 Patient Knowledge and Attitudes About for Clostridium difficile Infection Colin Goodman, MD; Nicholas O Rourke, PharmD; Carla Amundson, MA; and Dimitri Drekonja, MD, MS In a survey of patients with Clostridium
Patient Knowledge and Attitudes About for Clostridium difficile Infection Colin Goodman, MD; Nicholas O Rourke, PharmD; Carla Amundson, MA; and Dimitri Drekonja, MD, MS In a survey of patients with Clostridium
Los Angeles County Department of Public Health: Your Partner in CDI Prevention
 Los Angeles County Department of Public Health: Your Partner in CDI Prevention Dawn Terashita, MD, MPH Acute Communicable Disease Control Los Angeles County Department of Public Health dterashita@ph.lacounty.gov
Los Angeles County Department of Public Health: Your Partner in CDI Prevention Dawn Terashita, MD, MPH Acute Communicable Disease Control Los Angeles County Department of Public Health dterashita@ph.lacounty.gov
AN OUTBREAK OF TOXIN A NEGATIVE, TOXIN B POSITIVE CLOSTRIDIUM DIFFICILE -ASSOCIATED DIARRHEA IN A CANADIAN TERTIARY-CARE HOSPITAL
 Vol. 25-7 Date of publication: 1 April 1999 Contained in this FAX issue: (No. of pages: 6) Official page numbers: AN OUTBREAK OF TOXIN A NEGATIVE, TOXIN B POSITIVE CLOSTRIDIUM DIFFICILE-ASSOCIATED DIARRHEA
Vol. 25-7 Date of publication: 1 April 1999 Contained in this FAX issue: (No. of pages: 6) Official page numbers: AN OUTBREAK OF TOXIN A NEGATIVE, TOXIN B POSITIVE CLOSTRIDIUM DIFFICILE-ASSOCIATED DIARRHEA
Division of GIM Lecture Series Case Presentation David A. Erickson, M.D October 9th, 2013
 Division of GIM Lecture Series Case Presentation David A. Erickson, M.D October 9th, 2013 Financial Disclosures No financial disclosures Objectives Review a case of recurrent Clostridium difficile infection
Division of GIM Lecture Series Case Presentation David A. Erickson, M.D October 9th, 2013 Financial Disclosures No financial disclosures Objectives Review a case of recurrent Clostridium difficile infection
Antibiotic treatment comparison in patients with diarrhea
 Original Research Article Antibiotic treatment comparison in patients with diarrhea Deva Lal Kast * Senior Consultant Physician, Department of General Medicine, Krishna Hospital, Ex senior Specialist and
Original Research Article Antibiotic treatment comparison in patients with diarrhea Deva Lal Kast * Senior Consultant Physician, Department of General Medicine, Krishna Hospital, Ex senior Specialist and
Long-Term Care Updates
 Long-Term Care Updates April 2017 Bezlotoxumab to Prevent Recurrent Infection By Amy Wilson, PharmD and Zara Risoldi Cochrane, PharmD, MS, FASCP Introduction The Gram-positive bacteria is a common cause
Long-Term Care Updates April 2017 Bezlotoxumab to Prevent Recurrent Infection By Amy Wilson, PharmD and Zara Risoldi Cochrane, PharmD, MS, FASCP Introduction The Gram-positive bacteria is a common cause
New Molecular Diagnostic Assays for Solana Performance Assessment, Workflow Analysis, and Clinical Utility
 New Molecular Diagnostic Assays for Solana Performance Assessment, Workflow Analysis, and Clinical Utility Gerald A. Capraro, Ph.D., D(ABMM) Director, Clinical Microbiology Carolinas Pathology Group Atrium
New Molecular Diagnostic Assays for Solana Performance Assessment, Workflow Analysis, and Clinical Utility Gerald A. Capraro, Ph.D., D(ABMM) Director, Clinical Microbiology Carolinas Pathology Group Atrium
6/14/2012. Welcome! PRESENTATION OUTLINE CLOSTRIDIUM DIFFICILE PREVENTION. Teaming Up to Prevent Infections! 1) Impact. 2) Testing Recommendations
 CLOSTRIDIUM DIFFICILE PREVENTION Beth Goodall, RN, BSN Board Certified in Infection Prevention and Control DCH Health System Epidemiology Director Welcome! Teaming Up to Prevent Infections! CLOSTRIDIUM
CLOSTRIDIUM DIFFICILE PREVENTION Beth Goodall, RN, BSN Board Certified in Infection Prevention and Control DCH Health System Epidemiology Director Welcome! Teaming Up to Prevent Infections! CLOSTRIDIUM
Original Article. Infection Control & Hospital Epidemiology (2019), 1 5 doi: /ice
 Infection Control & Hospital Epidemiology (2019), 1 5 doi:10.1017/ice.2018.347 Original Article Healthcare provider diagnostic testing practices for identification of Clostridioides (Clostridium) difficile
Infection Control & Hospital Epidemiology (2019), 1 5 doi:10.1017/ice.2018.347 Original Article Healthcare provider diagnostic testing practices for identification of Clostridioides (Clostridium) difficile
-2002: Rectal blood loss, UC? (no definite diagnosis) rectal mesalazine. -June 2008: Recurrence of rectal blood loss and urgency
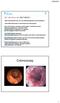 SD, male 40 yrs. old. (680718M467.) -2002: Rectal blood loss, UC? (no definite diagnosis) rectal mesalazine -June 2008: Recurrence of rectal blood loss and urgency Total colonoscopy: ulcerative rectitis,
SD, male 40 yrs. old. (680718M467.) -2002: Rectal blood loss, UC? (no definite diagnosis) rectal mesalazine -June 2008: Recurrence of rectal blood loss and urgency Total colonoscopy: ulcerative rectitis,
CDI The Impact. Disclosures. Acknowledgments. Objectives and Agenda. What s in the Name? 11/14/2012. Lets Talk Numbers
 Disclosures No conflict of interest to declare Acknowledgments Objectives and Agenda Society for Healthcare Epidemiology of America (SHEA) and the Infectious Diseases Society of America (IDSA) Guidelines
Disclosures No conflict of interest to declare Acknowledgments Objectives and Agenda Society for Healthcare Epidemiology of America (SHEA) and the Infectious Diseases Society of America (IDSA) Guidelines
Rapid and Sensitive Loop-Mediated Isothermal Amplification (LAMP) Test for. Gold Standard
 JCM Accepts, published online ahead of print on 24 November 2010 J. Clin. Microbiol. doi:10.1128/jcm.01824-10 Copyright 2010, American Society for Microbiology and/or the Listed Authors/Institutions. All
JCM Accepts, published online ahead of print on 24 November 2010 J. Clin. Microbiol. doi:10.1128/jcm.01824-10 Copyright 2010, American Society for Microbiology and/or the Listed Authors/Institutions. All
Loyola s C diff Journey
 Loyola s C diff Journey Kevin Smith, MD Associate Chief Medical Officer, Quality & Safety Aziz Ansari, DO Associate Chief Medical Officer, Clinical Optimization/Revenue Integrity Background Loyola Medicine
Loyola s C diff Journey Kevin Smith, MD Associate Chief Medical Officer, Quality & Safety Aziz Ansari, DO Associate Chief Medical Officer, Clinical Optimization/Revenue Integrity Background Loyola Medicine
CONSUMERS UNION POLICY BRIEF HOSPITAL-ACQUIRED C. DIFFICILE INFECTIONS (CDI) 11-08
 CONSUMERS UNION POLICY BRIEF HOSPITAL-ACQUIRED C. DIFFICILE INFECTIONS (CDI) 11-08 The growth of infections caused by Clostridium difficile (C.diff) is a highly alarming trend in hospitals today. In 1993
CONSUMERS UNION POLICY BRIEF HOSPITAL-ACQUIRED C. DIFFICILE INFECTIONS (CDI) 11-08 The growth of infections caused by Clostridium difficile (C.diff) is a highly alarming trend in hospitals today. In 1993
CLOSTRIDIUM DIFICILE. Negin N Blattman Infectious Diseases Phoenix VA Healthcare System
 CLOSTRIDIUM DIFICILE Negin N Blattman Infectious Diseases Phoenix VA Healthcare System ANTIBIOTIC ASSOCIATED DIARRHEA 1978: C diff first identified 1989-1992: Four large outbreaks in the US caused by J
CLOSTRIDIUM DIFICILE Negin N Blattman Infectious Diseases Phoenix VA Healthcare System ANTIBIOTIC ASSOCIATED DIARRHEA 1978: C diff first identified 1989-1992: Four large outbreaks in the US caused by J
Historical Perspective
 The Diagnosis of C. difficile Infections: Does Molecular Make it More or Less difficile? Stephen M. Brecher Ph.D. VA Boston Health Care System BU School of Medicine The opinions expressed in this presentation
The Diagnosis of C. difficile Infections: Does Molecular Make it More or Less difficile? Stephen M. Brecher Ph.D. VA Boston Health Care System BU School of Medicine The opinions expressed in this presentation
Clostridium difficile CRISTINA BAKER, MD, MPH INFECTIOUS DISEASE PARK NICOLLET/HEALTH PARTNERS 11/9/2018
 Clostridium difficile CRISTINA BAKER, MD, MPH INFECTIOUS DISEASE PARK NICOLLET/HEALTH PARTNERS 11/9/2018 Disclosures None Objectives Highlight important changes in the management of Clostridium difficile
Clostridium difficile CRISTINA BAKER, MD, MPH INFECTIOUS DISEASE PARK NICOLLET/HEALTH PARTNERS 11/9/2018 Disclosures None Objectives Highlight important changes in the management of Clostridium difficile
Clostridium difficile Diagnostic and Clinical Challenges
 Papers in Press. Published December 11, 2015 as doi:10.1373/clinchem.2015.243717 The latest version is at http://hwmaint.clinchem.org/cgi/doi/10.1373/clinchem.2015.243717 Clinical Chemistry 62:2 000 000
Papers in Press. Published December 11, 2015 as doi:10.1373/clinchem.2015.243717 The latest version is at http://hwmaint.clinchem.org/cgi/doi/10.1373/clinchem.2015.243717 Clinical Chemistry 62:2 000 000
MAJOR ARTICLE. Clostridium difficile infection; vancomycin; metronidazole; mortality; sepsis.
 MAJOR ARTICLE The Addition of Intravenous Metronidazole to Oral Vancomycin is Associated With Improved Mortality in Critically Ill Patients With Clostridium difficile Infection Kristina E. E. Rokas, 1
MAJOR ARTICLE The Addition of Intravenous Metronidazole to Oral Vancomycin is Associated With Improved Mortality in Critically Ill Patients With Clostridium difficile Infection Kristina E. E. Rokas, 1
Activity C: ELC Prevention Collaboratives
 Clostridium difficile il (CDI) Infections Toolkit Activity C: ELC Prevention Collaboratives Carolyn Gould, MD MSCR Cliff McDonald, MD, FACP Division of Healthcare Quality Promotion Centers for Disease
Clostridium difficile il (CDI) Infections Toolkit Activity C: ELC Prevention Collaboratives Carolyn Gould, MD MSCR Cliff McDonald, MD, FACP Division of Healthcare Quality Promotion Centers for Disease
Predictors of Mortality and Morbidity in Clostridium Difficile Infection
 Predictors of Mortality and Morbidity in Clostridium Difficile Infection Jill Dixon, Brian F. Menezes Corresponding author: dippers82@hotmail.com Pages 23-26 ISSN 1840-4529 http://www.iomcworld.com/ijcrimph
Predictors of Mortality and Morbidity in Clostridium Difficile Infection Jill Dixon, Brian F. Menezes Corresponding author: dippers82@hotmail.com Pages 23-26 ISSN 1840-4529 http://www.iomcworld.com/ijcrimph
! Macrolide antibacterial. Fidaxomicin (Dificid ) package labeling. Optimer Pharmaceuticals, Inc. May 2011.
 Disclosure! I have no conflicts of interest related to this presentation Nina Naeger Murphy, Pharm.D., BCPS Clinical Pharmacy Specialist Infectious Diseases MetroHealth Medical Center Learning Objectives!
Disclosure! I have no conflicts of interest related to this presentation Nina Naeger Murphy, Pharm.D., BCPS Clinical Pharmacy Specialist Infectious Diseases MetroHealth Medical Center Learning Objectives!
Indicator Definition
 Hospital Acquired Clostridium difficile Infection (CDI) Rate Full data definition sign-off completed. Name of Measure Name of Measure (short) Domain Type of Measure Business Context Rationale Hospital
Hospital Acquired Clostridium difficile Infection (CDI) Rate Full data definition sign-off completed. Name of Measure Name of Measure (short) Domain Type of Measure Business Context Rationale Hospital
Predictors of serious complications due to Clostridium difficile infection
 Alimentary Pharmacology & Therapeutics Predictors of serious complications due to Clostridium difficile infection D. GUJJA* & F. K. FRIEDENBERG *Department of Medicine, Abington Memorial Hospital, Abington,
Alimentary Pharmacology & Therapeutics Predictors of serious complications due to Clostridium difficile infection D. GUJJA* & F. K. FRIEDENBERG *Department of Medicine, Abington Memorial Hospital, Abington,
Bezlotoxumab (Zinplava) as Adjunct Treatment for Clostridium difficile. Janel Liane Cala, RPh Medical Center Hospital
 Bezlotoxumab (Zinplava) as Adjunct Treatment for Clostridium difficile Janel Liane Cala, RPh Medical Center Hospital Objectives Review pathophysiology, risk factors, prevention, and treatment options of
Bezlotoxumab (Zinplava) as Adjunct Treatment for Clostridium difficile Janel Liane Cala, RPh Medical Center Hospital Objectives Review pathophysiology, risk factors, prevention, and treatment options of
C.Difficile Associated Diarrhea Overview & Management. Sajal Chopra Pharm D. Candidate
 C.Difficile Associated Diarrhea Overview & Management Sajal Chopra Pharm D. Candidate Learning Objectives 1. Recognize the increasing incidence of C.difficile associated diarrhea (CDAD) and its risk factors
C.Difficile Associated Diarrhea Overview & Management Sajal Chopra Pharm D. Candidate Learning Objectives 1. Recognize the increasing incidence of C.difficile associated diarrhea (CDAD) and its risk factors
Cigdem Benlice, Ipek Sapci, T. Bora Cengiz, Luca Stocchi, Michael Valente, Tracy Hull, Scott R. Steele, Emre Gorgun 07/23/2018
 Does preoperative oral antibiotic or mechanical bowel preparation increase Clostridium difficile colitis after colon surgery? An assessment from ACS-NSQIP procedure-targeted database Cigdem Benlice, Ipek
Does preoperative oral antibiotic or mechanical bowel preparation increase Clostridium difficile colitis after colon surgery? An assessment from ACS-NSQIP procedure-targeted database Cigdem Benlice, Ipek
Updated Clostridium difficile Treatment Guidelines
 Updated Clostridium difficile Treatment Guidelines Arielle Arnold, PharmD, BCPS Clinical Pharmacist Saint Alphonsus Regional Medical Center September 29 th, 2018 Disclosures Nothing to disclose Learning
Updated Clostridium difficile Treatment Guidelines Arielle Arnold, PharmD, BCPS Clinical Pharmacist Saint Alphonsus Regional Medical Center September 29 th, 2018 Disclosures Nothing to disclose Learning
Questions and answers about the laboratory diagnosis of Clostridium difficile infection (CDI)
 Questions and answers about the laboratory diagnosis of Clostridium difficile infection (CDI) The NHS Centre for Evidence based Purchasing (CEP) has published the results of an evaluation of the performance
Questions and answers about the laboratory diagnosis of Clostridium difficile infection (CDI) The NHS Centre for Evidence based Purchasing (CEP) has published the results of an evaluation of the performance
International Journal of Pediatrics and Neonatal Health
 Research Article International Journal of Pediatrics and Neonatal Health Open Access Clostridium difficile Infection in Pediatric Inflammatory Bowel Disease Patients in the Era of Molecular Diagnostics
Research Article International Journal of Pediatrics and Neonatal Health Open Access Clostridium difficile Infection in Pediatric Inflammatory Bowel Disease Patients in the Era of Molecular Diagnostics
Clostridium difficile infection in an endemic setting in the Netherlands
 Eur J Clin Microbiol Infect Dis (2011) 30:587 593 DOI 10.1007/s10096-010-1127-4 ARTICLE Clostridium difficile infection in an endemic setting in the Netherlands M. P. M. Hensgens & A. Goorhuis & C. M.
Eur J Clin Microbiol Infect Dis (2011) 30:587 593 DOI 10.1007/s10096-010-1127-4 ARTICLE Clostridium difficile infection in an endemic setting in the Netherlands M. P. M. Hensgens & A. Goorhuis & C. M.
Case 1. Which of the following would be next appropriate investigation/s regarding the pts diarrhoea?
 Case 1 21 yr old HIV +ve, Cd4-100 HAART naïve Profuse diarrhoea for 3/52. Stool MC&S ve Which of the following would be next appropriate investigation/s regarding the pts diarrhoea? Repeat stool MC&S Stool
Case 1 21 yr old HIV +ve, Cd4-100 HAART naïve Profuse diarrhoea for 3/52. Stool MC&S ve Which of the following would be next appropriate investigation/s regarding the pts diarrhoea? Repeat stool MC&S Stool
Impact of clinical symptoms on interpretation of diagnostic assays for Clostridium difficile infections
 Washington University School of Medicine Digital Commons@Becker Open Access Publications 2011 Impact of clinical symptoms on interpretation of diagnostic assays for Clostridium difficile infections Erik
Washington University School of Medicine Digital Commons@Becker Open Access Publications 2011 Impact of clinical symptoms on interpretation of diagnostic assays for Clostridium difficile infections Erik
Each copy of any part of a JSTOR transmission must contain the same copyright notice that appears on the screen or printed page of such transmission.
 Clinical Practice Guidelines for Clostridium difficile Infection in Adults: 2010 Update by the Society for Healthcare Epidemiology of America (SHEA) and the Infectious Diseases Society of America (IDSA)
Clinical Practice Guidelines for Clostridium difficile Infection in Adults: 2010 Update by the Society for Healthcare Epidemiology of America (SHEA) and the Infectious Diseases Society of America (IDSA)
Clostridium difficile Testing Algorithms Using Glutamate Dehydrogenase Antigen and C.
 JCM Accepts, published online ahead of print on 18 January 2012 J. Clin. Microbiol. doi:10.1128/jcm.05620-11 Copyright 2012, American Society for Microbiology. All Rights Reserved. 1 2 3 Clostridium difficile
JCM Accepts, published online ahead of print on 18 January 2012 J. Clin. Microbiol. doi:10.1128/jcm.05620-11 Copyright 2012, American Society for Microbiology. All Rights Reserved. 1 2 3 Clostridium difficile
C. difficile Infection: How it all comes out
 C. difficile Infection: How it all comes out Larry Danziger, Pharm.D. Professor of Pharmacy and Medicine College of Pharmacy University of Illinois at Chicago The speaker has no conflicts to disclose.
C. difficile Infection: How it all comes out Larry Danziger, Pharm.D. Professor of Pharmacy and Medicine College of Pharmacy University of Illinois at Chicago The speaker has no conflicts to disclose.
In recent years, Clostridium difficile infection (CDI) has
 BRIEF REPORTS Poor Functional Status as a Risk Factor for Severe Clostridium difficile Infection in Hospitalized Older Adults Krishna Rao, MD,* Dejan Micic, MD,* Elizabeth Chenoweth, MD, Lili Deng, MD,
BRIEF REPORTS Poor Functional Status as a Risk Factor for Severe Clostridium difficile Infection in Hospitalized Older Adults Krishna Rao, MD,* Dejan Micic, MD,* Elizabeth Chenoweth, MD, Lili Deng, MD,
Clostridium Difficile colitismore
 Clostridium Difficile colitismore virulent than ever ECHO- February 18, 2016 Charles Krasner, M.D. UNR School of Medicine Sierra NV Veterans Affairs Hospital Growing problem of pseudomembranous colitis
Clostridium Difficile colitismore virulent than ever ECHO- February 18, 2016 Charles Krasner, M.D. UNR School of Medicine Sierra NV Veterans Affairs Hospital Growing problem of pseudomembranous colitis
What s New for Clostridium difficile John Lynch MD MPH Harborview Medical Center University of Washington
 What s New for Clostridium difficile 2013 John Lynch MD MPH Harborview Medical Center University of Washington Pathogenic Mechanisms of Diarrhea Toxins: Preformed: S aureus, C perfringens, B cereus Formed
What s New for Clostridium difficile 2013 John Lynch MD MPH Harborview Medical Center University of Washington Pathogenic Mechanisms of Diarrhea Toxins: Preformed: S aureus, C perfringens, B cereus Formed
Clostridium difficile Asymptomatic Carriers The Hidden Part of the Iceberg?
 Clostridium difficile Asymptomatic Carriers The Hidden Part of the Iceberg? Disclosures Merck Canada, BD Diagnostics, AMD Medical, Canadian Institute for Health Research Merck Canada, Pfizer OBJECTIVES
Clostridium difficile Asymptomatic Carriers The Hidden Part of the Iceberg? Disclosures Merck Canada, BD Diagnostics, AMD Medical, Canadian Institute for Health Research Merck Canada, Pfizer OBJECTIVES
Clostridium difficile infection (CDI) Week 52 (Ending 30/12/2017)
 Clostridium difficile infection (CDI) Week 52 (Ending 30/12/2017) What is Clostridium difficile? Clostridium difficile is a Gram-positive anaerobic spore forming bacillus. It is ubiquitous in nature and
Clostridium difficile infection (CDI) Week 52 (Ending 30/12/2017) What is Clostridium difficile? Clostridium difficile is a Gram-positive anaerobic spore forming bacillus. It is ubiquitous in nature and
MALDI-TOF MS: Translating Microbiology Laboratory Alphabet Soup to Optimize Antibiotic Therapy
 MALDI-TOF MS: Translating Microbiology Laboratory Alphabet Soup to Optimize Antibiotic Therapy September 8, 2017 Amy Carr, PharmD PGY-2 Infectious Diseases Pharmacy Resident Seton Healthcare Family amy.carr@ascension.org
MALDI-TOF MS: Translating Microbiology Laboratory Alphabet Soup to Optimize Antibiotic Therapy September 8, 2017 Amy Carr, PharmD PGY-2 Infectious Diseases Pharmacy Resident Seton Healthcare Family amy.carr@ascension.org
Annex C: - CDI What s the diff? 4 th Annual Outbreak Management Workshop September 19, 2013 Naideen Bailey & Grace Volkening
 Annex C: - CDI What s the diff? 4 th Annual Outbreak Management Workshop September 19, 2013 Naideen Bailey & Grace Volkening There s an updated Annex C Annex C is an extension to the PIDAC Infection Prevention
Annex C: - CDI What s the diff? 4 th Annual Outbreak Management Workshop September 19, 2013 Naideen Bailey & Grace Volkening There s an updated Annex C Annex C is an extension to the PIDAC Infection Prevention
Protocol for the Scottish Surveillance Programme for Clostridium difficile infection.
 National Services Scotland Protocol for the Scottish Surveillance Programme for Clostridium difficile infection. User manual. Version 4.0 Revised January 2017 Health Protection Scotland is a division of
National Services Scotland Protocol for the Scottish Surveillance Programme for Clostridium difficile infection. User manual. Version 4.0 Revised January 2017 Health Protection Scotland is a division of
Literature Scan: Antibiotics for Clostridium difficile Infection. Month/Year of Review: May 2015 Date of Last Review: April 2012
 Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Treatment of Clostridium Difficile Infection in Community Teaching Hospital: A Retrospective Study
 International Journal of Infectious Diseases and Therapy 2018; 3(3): 52-61 http://www.sciencepublishinggroup.com/j/ijidt doi: 10.11648/j.ijidt.20180303.12 ISSN: 2578-9651 (Print); ISSN: 2578-966X (Online)
International Journal of Infectious Diseases and Therapy 2018; 3(3): 52-61 http://www.sciencepublishinggroup.com/j/ijidt doi: 10.11648/j.ijidt.20180303.12 ISSN: 2578-9651 (Print); ISSN: 2578-966X (Online)
Probiotics for Primary Prevention of Clostridium difficile Infection
 Probiotics for Primary Prevention of Clostridium difficile Infection Objectives Review risk factors for Clostridium difficile infection (CDI) Describe guideline recommendations for CDI prevention Discuss
Probiotics for Primary Prevention of Clostridium difficile Infection Objectives Review risk factors for Clostridium difficile infection (CDI) Describe guideline recommendations for CDI prevention Discuss
HEALTHCARE- ASSOCIATED CLOSTRIDIUM DIFFICILE INFECTIONS IN CANADIAN ACUTE- CARE HOSPITALS
 HEALTHCARE- ASSOCIATED CLOSTRIDIUM DIFFICILE INFECTIONS IN CANADIAN ACUTE- CARE HOSPITALS SURVEILLANCE REPORT JANUARY 1 st, 2007 TO DECEMBER 31 st, 2012 TO PROMOTE AND PROTECT THE HEALTH OF CANADIANS THROUGH
HEALTHCARE- ASSOCIATED CLOSTRIDIUM DIFFICILE INFECTIONS IN CANADIAN ACUTE- CARE HOSPITALS SURVEILLANCE REPORT JANUARY 1 st, 2007 TO DECEMBER 31 st, 2012 TO PROMOTE AND PROTECT THE HEALTH OF CANADIANS THROUGH
Update on Clostridium difficile infection.
 Update on Clostridium difficile infection. K. Honein Gastroenterologist, HDF Associate Professor Head of Medicine Department St Joseph University-Beirut. Introduction Gram+anaerobic bacillus responsible
Update on Clostridium difficile infection. K. Honein Gastroenterologist, HDF Associate Professor Head of Medicine Department St Joseph University-Beirut. Introduction Gram+anaerobic bacillus responsible
Incidence of and risk factors for communityassociated Clostridium difficile infection
 University of Iowa Iowa Research Online Theses and Dissertations 2010 Incidence of and risk factors for communityassociated Clostridium difficile infection Jennifer Lee Kuntz University of Iowa Copyright
University of Iowa Iowa Research Online Theses and Dissertations 2010 Incidence of and risk factors for communityassociated Clostridium difficile infection Jennifer Lee Kuntz University of Iowa Copyright
Updates to pharmacological management in the prevention of recurrent Clostridium difficile
 Updates to pharmacological management in the prevention of recurrent Clostridium difficile Julia Shlensky, PharmD PGY2 Internal Medicine Resident September 12, 2017 2017 MFMER slide-1 Clinical Impact Increasing
Updates to pharmacological management in the prevention of recurrent Clostridium difficile Julia Shlensky, PharmD PGY2 Internal Medicine Resident September 12, 2017 2017 MFMER slide-1 Clinical Impact Increasing
INFECTION CONTROL AND HOSPITAL EPIDEMIOLOGY MAY 2010, VOL. 31, NO. 5
 INFECTION CONTROL AND HOSPITAL EPIDEMIOLOGY MAY 2010, VOL. 31, NO. 5 SHEA-IDSA GUIDELINE Clinical Practice Guidelines for Clostridium difficile Infection in Adults: 2010 Update by the Society for Healthcare
INFECTION CONTROL AND HOSPITAL EPIDEMIOLOGY MAY 2010, VOL. 31, NO. 5 SHEA-IDSA GUIDELINE Clinical Practice Guidelines for Clostridium difficile Infection in Adults: 2010 Update by the Society for Healthcare
C lostridium difficile is an anaerobic Gram positive bacillus
 673 GASTROINTESTINAL INFECTION Clostridium difficile associated diarrhoea in hospitalised patients: onset in the community and hospital and role of S S Johal, J Hammond, K Solomon, P D James, Y R Mahida...
673 GASTROINTESTINAL INFECTION Clostridium difficile associated diarrhoea in hospitalised patients: onset in the community and hospital and role of S S Johal, J Hammond, K Solomon, P D James, Y R Mahida...
AdvaMed Medtech Value Assessment Framework in Practice
 AdvaMed Medtech Value Assessment Framework in Practice Application of the Medtech Value Assessment Framework to Cymedica s e-vive System Value Framework Overview In response to the growing need to demonstrate
AdvaMed Medtech Value Assessment Framework in Practice Application of the Medtech Value Assessment Framework to Cymedica s e-vive System Value Framework Overview In response to the growing need to demonstrate
Clostridium Difficile Infection in Adults Treatment and Prevention
 Clostridium Difficile Infection in Adults Treatment and Prevention Definition: Clostridium Difficile colonizes the human intestinal tract after the normal gut flora has been altered by antibiotic therapy
Clostridium Difficile Infection in Adults Treatment and Prevention Definition: Clostridium Difficile colonizes the human intestinal tract after the normal gut flora has been altered by antibiotic therapy
Validation of Clinical Outcomes in Electronic Data Sources
 Validation of Clinical Outcomes in Electronic Data Sources Vincent Lo Re, MD, MSCE Assistant Professor of Medicine and Epidemiology Center for Clinical Epidemiology and Biostatistics Center for Pharmacoepidemiology
Validation of Clinical Outcomes in Electronic Data Sources Vincent Lo Re, MD, MSCE Assistant Professor of Medicine and Epidemiology Center for Clinical Epidemiology and Biostatistics Center for Pharmacoepidemiology
Clostridium difficile Infection in the Department of Defense (DOD):
 1 1 Clostridium difficile Infection in the Department of Defense (DOD): 2007-2013 NMCPHC-EDC- TR-271-2015 By Charlotte Neumann and Uzo Chukwuma February 2015 Approved for public release. Distribution is
1 1 Clostridium difficile Infection in the Department of Defense (DOD): 2007-2013 NMCPHC-EDC- TR-271-2015 By Charlotte Neumann and Uzo Chukwuma February 2015 Approved for public release. Distribution is
Clostridium difficile associated diarrhea (CDAD) has emerged. Incidence of Clostridium difficile Infection in Inflammatory Bowel Disease
 CLINICAL GASTROENTEROLOGY AND HEPATOLOGY 2007;5:339 344 Incidence of Clostridium difficile Infection in Inflammatory Bowel Disease JOSEPH F. RODEMANN,* ERIK R. DUBBERKE, KIMBERLY A. RESKE, DA HEA SEO,*
CLINICAL GASTROENTEROLOGY AND HEPATOLOGY 2007;5:339 344 Incidence of Clostridium difficile Infection in Inflammatory Bowel Disease JOSEPH F. RODEMANN,* ERIK R. DUBBERKE, KIMBERLY A. RESKE, DA HEA SEO,*
Prevention of Cdiff recurrence: Evidence from a Cdiff Antitoxin
 Prevention of Cdiff recurrence: Evidence from a Cdiff Antitoxin Prof. Emilio Bouza Departament of Medicine. Hospital Gregorio Marañon and Complutense University. Madrid. Spain Disclosures Last year Participation
Prevention of Cdiff recurrence: Evidence from a Cdiff Antitoxin Prof. Emilio Bouza Departament of Medicine. Hospital Gregorio Marañon and Complutense University. Madrid. Spain Disclosures Last year Participation
(Facility Name and Address) (1D) Surveillance of Urinary Tract Infections in the Long-Term Care Setting
 Policy Number: 1D Date: 4/16/14 Version: 1 (1D) Surveillance of Urinary Tract Infections in the Long-Term Care Setting Introduction: One-quarter of the older adult population in the United States will
Policy Number: 1D Date: 4/16/14 Version: 1 (1D) Surveillance of Urinary Tract Infections in the Long-Term Care Setting Introduction: One-quarter of the older adult population in the United States will
SC P. on POOP. Susan E. Sharp, Ph.D., DABMM Director of Microbiology Portland, OR
 The Straight SC P on POOP Susan E. Sharp, Ph.D., DABMM Director of Microbiology Portland, OR 97230 susan.e.sharp@kp.org But FIRST - - Quiz time! But FIRST - - Quiz time! Here s the QUIZ 4 Here s the QUIZ
The Straight SC P on POOP Susan E. Sharp, Ph.D., DABMM Director of Microbiology Portland, OR 97230 susan.e.sharp@kp.org But FIRST - - Quiz time! But FIRST - - Quiz time! Here s the QUIZ 4 Here s the QUIZ
Clostridium difficile Infection (CDI)
 18.09.10 월요집담회 Clostridium difficile Infection (CDI) R4 송주혜 Clostridium difficile infection (CDI) Anaerobic gram (+), spore-forming, toxin(tcda&tcdb)-producing bacillus Transmitted among humans through
18.09.10 월요집담회 Clostridium difficile Infection (CDI) R4 송주혜 Clostridium difficile infection (CDI) Anaerobic gram (+), spore-forming, toxin(tcda&tcdb)-producing bacillus Transmitted among humans through
The Epidemiology of Clostridium difficile DANIEL SAMAN, DRPH, MPH RESEARCH SCIENTIST ESSENTIA INSTITUTE OF RURAL HEALTH
 The Epidemiology of Clostridium difficile DANIEL SAMAN, DRPH, MPH RESEARCH SCIENTIST ESSENTIA INSTITUTE OF RURAL HEALTH Some history first Clostridium difficile, a spore-forming gram-positive (i.e., thick
The Epidemiology of Clostridium difficile DANIEL SAMAN, DRPH, MPH RESEARCH SCIENTIST ESSENTIA INSTITUTE OF RURAL HEALTH Some history first Clostridium difficile, a spore-forming gram-positive (i.e., thick
Fidaxomicin for Clostridium difficile-associated Diarrhoea
 ORIGINAL RESEARCH ARTICLE Clin Drug Investig 2012; 32 (8): e17-e24 1173-2563/12/0008-0017 Adis ª 2012 Sclar et al., publisher and licensee Springer International Publishing AG. This is an open access article
ORIGINAL RESEARCH ARTICLE Clin Drug Investig 2012; 32 (8): e17-e24 1173-2563/12/0008-0017 Adis ª 2012 Sclar et al., publisher and licensee Springer International Publishing AG. This is an open access article
Reevaluation of Epidemiological Criteria for Identifying Outbreaks of Acute Gastroenteritis Due to Norovirus: United States,
 MAJOR ARTICLE Reevaluation of Epidemiological Criteria for Identifying Outbreaks of Acute Gastroenteritis Due to Norovirus: United States, 1998 2000 Reina M. Turcios, 1 Marc-Alain Widdowson, 1 Alana C.
MAJOR ARTICLE Reevaluation of Epidemiological Criteria for Identifying Outbreaks of Acute Gastroenteritis Due to Norovirus: United States, 1998 2000 Reina M. Turcios, 1 Marc-Alain Widdowson, 1 Alana C.
ECHO-Antibiotic Stewardship Program
 ECHO-Antibiotic Stewardship Program More Interesting Recent Literature Updates April 20, 2017 Charles Krasner, M.D. University of NV, Reno School of Medicine Sierra NV Veterans Affairs Medical Center Antibiotic
ECHO-Antibiotic Stewardship Program More Interesting Recent Literature Updates April 20, 2017 Charles Krasner, M.D. University of NV, Reno School of Medicine Sierra NV Veterans Affairs Medical Center Antibiotic
The disease formerly called Clostridium difficile associated
 Improving Patient Care Annals of Internal Medicine Does My Patient Have Clostridium difficile Infection? Lance R. Peterson, MD, and Ari Robicsek, MD Clostridium difficile infection (CDI) seems to be changing
Improving Patient Care Annals of Internal Medicine Does My Patient Have Clostridium difficile Infection? Lance R. Peterson, MD, and Ari Robicsek, MD Clostridium difficile infection (CDI) seems to be changing
