Minimum effective dose of midazolam for sedation of mechanically ventilated neonates
|
|
|
- Willis Carson
- 6 years ago
- Views:
Transcription
1 Journal of Clinical Pharmacy and Therapeutics (2005) 30, ORIGINAL ARTICLE Minimum effective dose of midazolam for sedation of mechanically ventilated neonates J.-M. Treluyer* MD PhD, S.Zohar PhD, E.Rey* Pharm D, P.Hubertà MD, F. Iserinà MD, M. Jugie MD, R.Lenclen MD, S.Chevret MD PhD and G. Pons* MD PhD *Pharmacologie, Hôpital Cochin-Saint-Vincent de Paul, AP-HP, Université Paris V, Paris, Département de Biostatistique et Informatique Médicale, Hôpital Saint-Louis, AP-HP, Université Paris VII, U717 INSERM, Paris, àréanimation Pédiatrique, Cardiologie Hôpital Necker Enfants malades, AP-HP, Université Paris V, Paris and Réanimation, Hôpital de Poissy-Saint Germain, France SUMMARY Objective: To determine the minimal effective dose (MED) of intravenous midazolam, required for appropriate sedation in 95% of patients, 1 h after drug administration. Methods: A double-blind dose-finding study using the continual reassessment method, a Bayesian sequential design. Twenty-three newborn infants hospitalized in intensive care unit participated. Inclusion criteria were: (i) postnatal age <28 days, (ii) gestational age >33 weeks, (iii) intubation and ventilatory support required for respiratory distress syndrome, (iv) need for sedation (i.e. one of the six following criteria: agitation or grimacing or crying facial expression before tracheal suctioning, agitation or grimacing or crying facial expression during tracheal suctioning). Each neonate was allocated to a loading dose, ranging from 75 to 200 lg/kg, and a maintenance dose ranging from 37Æ5 to 100 lg/kg/h. Results: The primary endpoint was the level of sedation 1 h after the onset of infusion. The sedation procedure was classified as a success if all the following clinical criteria were met: no agitation, no grimacing and no crying facial expression before as well as during tracheal suctioning. Based on the 23 patients, the final Received 18 March 2005, Accepted 5 July 2005 Correspondence: Jean-Marc Treluyer, Pharmacologie Clinique, Hôpital Saint-Vincent de Paul, 82 avenue Denfert Rochereau, Paris cedex 14, France. Tel.: 33/ ; fax: 33/ ; jm.treluyer@svp.ap-hop-paris.fr estimated probability of success was 76Æ9% (95% credibility interval: 56Æ6 91Æ4%) for the 200 lg/kg loading dose. No significant adverse effect was observed. Conclusions: Continual reassessment is a new approach, suitable for dose-finding study in neonates. This method overcomes some of the ethical, statistical and practical problems associated with this population. Using this method, the MED was estimated to be the 200 lg/kg loading dose of midazolam. Keywords: continual reassessment method, dosefinding study, midazolam, neonates, stopping rules for decision making INTRODUCTION Proper sedation of patients is part of good clinical practice in intensive care units (ICU) in children as well as in adults. The main goal of sedation is to increase patients comfort by decreasing stress and anxiety (1). Moreover, adequate sedation during mechanical ventilation may facilitate effective ventilation so that complications such as pneumothorax and intraventricular haemorrhage may be prevented (2, 3). Benzodiazepines, such as midazolam and lorazepam, are frequently chosen for sedation in the neonatal ICU. Intravenous midazolam differs from other benzodiazepines by its water solubility, short elimination half-life, and generally short duration of action. Many authors have studied the pharmacokinetic properties and clinical efficacy of midazolam in ventilated neonates. However, the dosage used to initiate and Ó 2005 Blackwell Publishing Ltd 479
2 480 J.-M. Treluyer et al. maintain sedation show dramatic differences. Most reports to date have been case series of midazolam used in patients of diverse age groups (from 3 days to 18 years of age), with doses ranging from 25 to 300 lg/kg administered as a bolus injection, and lg/kg/h administered as a continuous infusion (4 8). In a multicentre pilot study, Anand et al. (9), administered midazolam with 200 lg/kg loading dose followed by an infusion of 20, 40, or 60 lg/kg/h for infants of gestational ages 24 26, 27 29, or weeks respectively. Jacqz-Aigrain et al. (10), administered midazolam as a 60 lg/kg/h infusion for up to 5 days in infants >33 week gestation, or 60 lg/kg/h for 1 day followed by 30 lg/kg/h for up to a total of 5 days in infants 33 week gestation without any loading dose. As with many drugs used in children, little is known about midazolam in neonates. In using midazolam as a sedative in ventilated neonates, the dosing regimen for effective and safe treatment is a key issue and no adequate evaluation has been reported. However, in this patient group such a dosefinding study meets with distinct ethical, statistical and practical problems. A new approach proposed and used previously by us for dose-finding of ibuprofen in patent ductus arteriosus (12) is proposed for dose-finding of midazolam for sedation of ventilated neonates, to overcome some of the ethical and recruitment issues. The main objective was to determine the dose of intravenous midazolam, required to obtain appropriate sedation in 95% of ventilated neonates within 1 h of administration. MATERIALS AND METHODS The study was approved by the local ethics committee (Cochin Hospital, Paris) and written informed consent was obtained from the parents. Inclusion criteria were: (i) post-natal age <28 days, (ii) gestational age >33 weeks, (iii) intubation and ventilatory support required for respiratory distress syndrome, (iv) need for sedation (i.e. one of the six following criteria: agitation or grimacing or crying facial expression before tracheal suctioning, agitation or grimacing or crying facial expression during tracheal suctioning). We excluded babies if they or their mother had previously received benzodiazepines or opioids during the 15 days before the time of inclusion. The other exclusion criteria were abnormalities of renal, hepatic, neurologic or haemodynamic functions. Midazolam was administered intravenously at a 0Æ6 ml/kg loading dose over 1 h, followed by an infusion of 0Æ3 ml/kg/h over 47 h using the same infusion rate for all the dose levels. Only the concentration of the solution was modified. The concentrations of the solution ranged from 6Æ2 to 16Æ7 mg/50 ml. Each neonate was double-blindedly allocated to a loading dose of lg/kg and a maintenance dose of 37Æ5 100 lg/kg/h. Six dose regimens were tested: 75, 100, 125, 150, 175, 200 and 37Æ5, 50, 62Æ5, 75, 87Æ5, 100 lg/kg for loading doses and maintenance doses respectively. Physicians, nurses and all NICU staff were blind to the dose of midazolam administered. The primary endpoint was the level of sedation 1 h after the onset of the infusion (H1). The sedation procedure was classified as a success if all the following clinical criteria were absent: agitation, grimacing and crying facial expression before as well as during tracheal suctioning. The following secondary endpoints were also studied: level of sedation at H4, H12, H18, H24, H48 using the same criteria used at H1, heart rate, blood pressure, oxygen saturation measured by pulse oximetry, oxygenation index, and triggering of ventilator breathing by the patients at H0, H1, H4, H12, H18, H24 and H48. Additional midazolam bolus doses or increases in the rate of infusion were not allowed. Sedative drugs that may interact with the assessment of sedation (benzodiazepines, opioids, muscle relaxants) were not allowed. Infusion was continued for as long as clinically necessary but no longer than 48 h. At completion of the study, depending on the level of sedation achieved, physicians were allowed to prescribe midazolam or another sedative drug according to their own practice. After the assessment of the primary endpoint, the treatment could be stopped before the end of the study if the level of sedation was considered insufficient by the physicians. Midazolam was also stopped after H1 if respiratory distress rapidly improved and extubation of the child was considered. Dose-finding statistical analysis The continual reassessment method (CRM) (11), a Bayesian sequential design, was used in order to
3 Minimal effective dose of midazolam 481 determine the minimal effective dose (MED) of midazolam for the sedation of 95% of newborn infants. The CRM is an iterative Bayesian method based on a one-parameter model, which aims at estimating the percentile of among k distinct dose levels d i (i = 1,, k). Each of the six dose levels was arbitrarily chosen by the investigator (according to his/her personal experience and literature data) with the following prior estimated success probability, p i (i = 1,, 6), that is 0Æ3, 0Æ5, 0Æ7, 0Æ9, 0Æ95 and 1Æ0 for the 75, 100, 125, 150, 175 and 200 lg/kg loading dose respectively. Then, a one-parameter logistic model (the scale parameter was fixed at 3) was used to fit the dose response curve, with a unit exponential distribution for the model parameter. The posterior response probability of each dose level was re-estimated after each new patient s inclusion. The dose allocated to each new patient was the dose level with the updated posterior response probability closest to 0Æ95. In the present study, the first patient received a loading dose of 175 lg/kg with the prior success probability closest to the target (0Æ95). The MED was defined as the dose level among the six chosen doses that had a final response probability closest to the target. The decision to end the study was based on stopping criteria, in order to detect whether all doses were likely to be inefficient, or a suitable estimation of the MED has been obtained (13). RESULTS Description of the population Twenty-three newborns entered the study. The demographic and clinical variables for the neonates are presented in Table 1. Twenty-one (91Æ3%) children stopped the study before H48, seven (30Æ4%) because the level of sedation was considered insufficient by the physicians and 14 (60Æ9%) because the respiratory distress rapidly improved and children were extubated. The median observation time period was 12 h (1 60). Dose-finding analysis The estimated success probability, updated after each patient, was <95% for all dose level, and led to allocation of the maximum loading dose Table 1. Demographic and clinical variables of the neonates at inclusion Patients N (%)/Median (range) Sex (male) 16 (69Æ6) Post-natal age (days) 0 (0 4) Gestational age >33 weeks 23 (100) Weight (g) 2650 ( ) FiO 2 (%) 45 (21 80) Serum creatinine (lmol/l) 86Æ5 (50 122) Prothrombin time (%) 59 (20 79) Plasma fibrine (g/l) 1Æ9 (0Æ2 5Æ5) Blood platelets (10 3 /mm 3 ) 198 ( ) (200 lg/kg) from the fifth patient to the 23rd patient (Table 2). The final estimated probability of success of that dose level was 76Æ9% (95% credibility interval: 56Æ6 91Æ4%) after 23 infants had been included (Fig. 1). The posterior success probability for the six doses 75, 100, 125, 150, 175 and 200 lg/kg corresponded to 27Æ7%, 32Æ7%, 38Æ1%, 47Æ3%, 52Æ5% and 76Æ9% respectively (Table 2). Because the target was fixed at 95%, the MED was selected to be 200 lg/kg. Failures were recorded in five (25%) of 20 patients receiving a 200 lg/kg midazolam loading dose. Among these patients, the failure was assessed on 6, 4, 3, 3, 1 of the 6 failure criteria. Classification as a failure was the result of agitation (n = 2), grimacing (n = 3) and crying facial expression (n = 3) before tracheal suctioning, agitation (n = 3), grimacing (n = 4) and crying facial expression (n = 3) during tracheal suctioning. The stopping decision has been made after the analysis of the stopping criteria. The first decision was based on the probability that the posterior estimated response probability of success of either dose was lower than the targeted response probability. This probability was higher than 95% after the inclusion of the 14th patient. The second stopping decision was associated with four stopping criteria, based on the predictive mean and the maximum gain from further patient inclusions on the response probability and the width of the associated credibility interval. These stopping criteria (3 from 4) were lower than 5%, after the 20th patient with a success probability at 78Æ7% (95% credibility interval: 57Æ6 93Æ3%). The response probability associated with the 200 lg/kg dose level would have changed for <5%, even if more
4 482 J.-M. Treluyer et al. Table 2. Posterior estimated probabilities of success of the five tested doses, updated after each newly included patient Midazolam loading dose (lg/kg) Prior estimated probability of success (%) Subject Dose (lg/kg) Clinical response 30Æ0 50Æ0 70Æ0 90Æ0 95Æ0* 100 Posterior estimated probability of success (%) Success 30Æ8 55Æ8 78Æ2 95Æ0 98Æ Success 31Æ4 60Æ8 84Æ0 97Æ3 99Æ Failure 28Æ5 38Æ6 49Æ7 67Æ1 75Æ3 96Æ Failure 27Æ5 30Æ9 34Æ6 40Æ9 44Æ5 63Æ Success 27Æ6 32Æ0 36Æ7 44Æ7 49Æ3 72Æ Success 27Æ8 33Æ0 38Æ6 48Æ3 53Æ7 78Æ Success 27Æ9 33Æ8 40Æ3 51Æ2 57Æ3 83Æ Success 28Æ0 34Æ5 41Æ6 53Æ6 60Æ2 86Æ Failure 27Æ7 32Æ1 36Æ9 45Æ2 49Æ8 73Æ Success 27Æ7 32Æ7 38Æ0 47Æ1 55Æ3 76Æ Success 27Æ8 33Æ1 38Æ9 48Æ8 54Æ4 79Æ Success 27Æ9 33Æ5 39Æ8 50Æ3 56Æ2 81Æ Success 27Æ9 33Æ9 40Æ5 51Æ6 57Æ7 83Æ Failure 27Æ7 32Æ6 37Æ9 46Æ8 51Æ9 76Æ Success 27Æ8 32Æ9 38Æ5 48Æ0 53Æ3 78Æ Success 27Æ8 33Æ2 39Æ0 49Æ0 54Æ6 79Æ Success 27Æ8 33Æ4 39Æ6 49Æ9 55Æ7 81Æ Success 27Æ9 33Æ7 40Æ0 50Æ8 56Æ8 82Æ Success 27Æ9 33Æ9 40Æ5 51Æ6 57Æ7 83Æ Failure 27Æ8 33Æ0 38Æ7 48Æ3 53Æ8 78Æ Success 27Æ8 33Æ2 39Æ1 49Æ1 54Æ7 79Æ Success 27Æ8 33Æ4 39Æ5 49Æ7 55Æ5 81Æ Failure 27Æ7 32Æ7 38Æ1 47Æ3 52Æ5 76Æ9 *Targeted probability of success. Stopping criteria Maximum predictive gain on the response probability Maximum predictive gain on the width of the credibility interval Mean predictive gain on the response probability Mean predictive gain on the width of the credibility interval Stopping bound Patient number Fig. 1. Estimated posterior probability of success of the 200 lg/kg midazolam dose and associated 95% credibility interval along the trial. This dose level was administered from the fourth patient up to the last patient.
5 Minimal effective dose of midazolam Fig. 2. Stopping criteria based on the predicted gain from further patient inclusions on the response probability (mean and maximum) and on the width of its credibility interval (mean and maximum). Three criteria out of the four were lower than 5% from the 20th patient. Probability Estimated probability of success Upper bound of credibility interval Lower bound of credibility interval Patient number patients were included (Fig. 2). No significant effect of sedation on pulse oximetry, oxygenation index or triggering of ventilator breathing was evidenced after the onset of midazolam administration. Adverse events No serious adverse event was reported during the study. Pneumothorax was reported in two children who had received a 200 lg/kg initial loading dose, and were classified as success for sedation. However, the sedation study was stopped after the diagnosis of pneumothorax to administer morphine for prevention of pain. Relative median variations between H0 and H1 of systolic, diastolic and mean blood pressure were )2% ()18%; 27%), )7% ()43%; 19%), )6% ()40%; 15%), respectively, but none was found different from 0. A decrease of more than 30% for systolic, diastolic and mean blood pressure was observed during the study inclusion in none, two (8Æ7%) and one (4Æ3%) children respectively (all received the 200 lg/kg loading dose level). However, this event was very transient and no haemodynamic support was required. Relative variation of heart rate was )4% ()22%; 16%) between H0 and H1. DISCUSSION Midazolam is suitable for sedation of neonates as it is a short-acting benzodiazepine with a high clearance and a short elimination half-life. However, consequent to immature hepatic cytochrome P450 activity, midazolam clearance is reduced, and half-life and time to reach steady-state increased in neonates compared with children and adults (14 20). Consequently, a specific phase II study was conducted to determine the optimal dose for sedation of ventilated neonates. A CRM was chosen for this study because of the potential advantages of such an approach in paediatric patients: small number of patients required (25 or less), no placebo group necessary and ethical suitability as each subject takes advantage of the previous inclusions to receive the current estimated MED. As our objective was to obtain an optimal sedation 1 h after the onset of midazolam administration, a loading dose was used. The final estimated probability of success of the highest dose (i.e. 200 lg/kg for the loading dose and 100 lg/kg/h for the maintenance dose) was 76Æ9% (95% credibility interval: 56Æ6 91Æ4%). Based upon the estimated probabilities of success, the optimal dose to obtain 95% of success (the initial objective of our study) appears to be higher than the maximum dose used in this study. One can wonder if the criteria used to define sedation were too stringent. There is a paucity of tools to measure the level of sedation in preterm neonates and those who require mechanical ventilation. The scoring systems used in previous studies concerning use of midazolam for sedation in neonates were not validated and were not assessed during clinical care. We used a behavioural scale adapted from the scoring system by Barrier et al. (21) and Jacqz-Aigrain et al. (10). We modified these scores as we chose to assess sedation before but also during tracheal
6 484 J.-M. Treluyer et al. suctioning. As tracheal suction is an acute and repetitively stressful in mechanically ventilated neonates, and as the main objective of sedation is to decrease the deleterious effects of clinical care for the patients, we assessed sedation during tracheal suction. The other difference in the design of our study compared with previously published studies is that sedation was assessed as early as 1 h after the onset of midazolam infusion to obtain an effective and very rapid sedation. This may explain the highest dose of midazolam being selected at the end of the study. The aim of this study was to improve management of non-painful stress (anxiety). Analgesics were administered only if painful procedures were anticipated. However, concomitant use of opioids and benzodiazepines necessitates a decrease in the total dose of opioids and benzodiazepines (22). Consequently, results of the present study are applicable only when midazolam is used singly. No severe side-effect was observed. Transient hypotension was noticed in two children. In the study by Jacqz-Aigrain et al. (10) the number of infants with haemodynamic instability was not significantly different between the two midazolam and the placebo groups, although blood pressure was significantly decreased in the midazolam group. In most cases, hypotension observed with midazolam occurred only during concomitant use of opioids (23, 24). Transient neurologic effects have been associated with the use of midazolam, but no such side-effect was noticed in the present study (22). In conclusion, the probability of success for sedation in ventilated neonates, even during tracheal suctioning, using a 200 lg/kg loading dose of midazolam during 1 h followed by a 100 lg/kg/h maintenance dose, was estimated to be 76Æ9% in the absence of analgesics. Given the small number of children included, tolerability of this dose schedule needs to be confirmed in a larger group of neonates. REFERENCES 1. Quinn MW, Wild J, Dean HG, Hartley R, Rushforth JA, Puntis JW, Levene MI (1993) Randomised double-blind controlled trial of effect of morphine on catecholamine concentrations in ventilated pre-term babies. Lancet, 342, Greenough A, Morley C, Davis J (1983) Interaction of spontaneous respiration with artificial ventilation in preterm babies. Journal of Pediatrics, 103, Perlman JM, Goodman S, Kreusser KL, Volpe JJ (1985) Reduction in intraventricular hemorrhage by elimination of fluctuating cerebral blood-flow velocity in preterm infants with respiratory distress syndrome. New England Journal of Medicine, 312, Hartwig S, Roth B, Theisohn M (1991) Clinical experience with continuous intravenous sedation using midazolam and fentanyl in the paediatric intensive care unit. European Journal of Pediatrics, 150, Pellier I, Monrigal JP, Le Moine P, Rod B, Rialland X, Granry JC (1999) Use of intravenous ketamine-midazolam association for pain procedures in children with cancer. A prospective study. Paediatric Anaesthesia, 9, Rosen DA, Rosen KR (1991) Midazolam for sedation in the paediatric intensive care unit. Intensive Care Medicine, 17 (Suppl. 1), S15 S Hogberg L, Nordvall M, Tjellstrom B, Stenhammar L (1995) Intranasal versus intravenous administration of midazolam to children undergoing small bowel biopsy. Acta Paediatrica, 84, Ng E, Taddio A, Ohlsson A (2000) Intravenous midazolam infusion for sedation of infants in the neonatal intensive care unit. Cochrane Database of Systematic Review, CD Anand KJS, McIntosh N, Lagercrantz H, Pelausa E, Young TE, Vasa R (1999) Analgesia and sedation in preterm neonates who require ventilatory support results from the NOPAIN trial. Archives of Pediatrics and Adolescent Medicine, 153, Jacqz-Aigrain E, Daoud P, Burtin P, Desplanques L, Beaufils F (1994) Placebo-controlled trial of midazolam sedation in mechanically ventilated newborn babies [see comments]. Lancet, 344, Desfrere L, Zohar S, Morville P et al. (2003) Dosefinding study of ibuprofen in patent ductus arteriosus using the continual assessment method. Journal of Clinical Pharmacy and Therapeutics, 30, O Quigley J, Pepe M, Fisher L (1990) Continual reassessment method: a practical design for phase 1 clinical trials in cancer. Biometrics, 46, Zohar S, Chevret S (2001) The continual reassessment method: comparison of Bayesian stopping rules for dose-ranging studies. Statistics in Medicine, 20, de Wildt SN, Kearns GL, Hop WC, Murry DJ, Abdel- Rahman SM, van den Anker JN (2001) Pharmacokinetics and metabolism of intravenous midazolam in
7 Minimal effective dose of midazolam 485 preterm infants. Clinical Pharmacology and Therapeutics, 70, Nahara MC, McMorrow J, Jones PR, Anglin D, Rosenberg R (2000) Pharmacokinetics of midazolam in critically ill pediatric patients. European Journal of Drug Metabolism and Pharmacokinetics, 25, Jacqz-Aigrain E, Daoud P, Burtin P, Maherzi S, Beaufils F (1992) Pharmacokinetics of midazolam during continuous infusion in critically ill neonates. European Journal of Clinical Pharmacology, 42, Jacqz-Aigrain E, Wood C, Robieux I (1990) Pharmacokinetics of midazolam in critically ill neonates. European Journal of Clinical Pharmacology, 39, Burtin P, Jacqz-Aigrain E, Girard P et al. (1994) Population pharmacokinetics of midazolam in neonates. Clinical Pharmacology and Therapeutics, 56, Lee TC, Charles BG, Harte GJ, Gray PH, Steer PA, Flenady VJ (1999) Population pharmacokinetic modeling in very premature infants receiving midazolam during mechanical ventilation: midazolam neonatal pharmacokinetics. Anesthesiology, 90, Harte GJ, Gray PH, Lee TC, Steer PA, Charles BG (1997) Haemodynamic responses and population pharmacokinetics of midazolam following administration to ventilated, preterm neonates. Journal of Paediatrics and Child Health, 33, Barrier G, Attia J, Mayer MN, Amiel-Tison C, Shnider SM (1989) Measurement of post-operative pain and narcotic administration in infants using a new clinical scoring system. Intensive Care Medicine, 15 (Suppl. 1), S37 S Fetus and Newborn Committee (2000) Prevention and management of pain and stress in the neonate. American Academy of Pediatrics. Committee on Fetus and Newborn. Committee on Drugs. Section on Anesthesiology. Section on Surgery. Canadian Paediatric Society. Pediatrics, 105, Van den Anker J, Sauer P. (1992) The use of midazolam in the preterm neonate. European Journal of Pediatrics, 151, Burtin P, Daoud P, Jacqz-Aigrain E, Mussat P, Moriette G (1991) Hypotension with midazolam and fentanyl in the newborn. Lancet, 337,
SEDATION FOR SMALL PROCEDURES
 SEDATION FOR SMALL PROCEDURES Sinno Simons Erasmus MC Sophia Children s Hospital Rotterdam, the Netherlands s.simons@erasmusmc.nl SEDATION in newborns How and when How to evaluate How to dose Why to use
SEDATION FOR SMALL PROCEDURES Sinno Simons Erasmus MC Sophia Children s Hospital Rotterdam, the Netherlands s.simons@erasmusmc.nl SEDATION in newborns How and when How to evaluate How to dose Why to use
P ain management of neonates admitted to neonatal
 F46 ORIGINAL ARTICLE Morphine in ventilated neonates: its effects on arterial blood pressure S H P Simons, D W E Roofthooft, M van Dijk, R A van Lingen, H J Duivenvoorden, J N van den Anker, D Tibboel...
F46 ORIGINAL ARTICLE Morphine in ventilated neonates: its effects on arterial blood pressure S H P Simons, D W E Roofthooft, M van Dijk, R A van Lingen, H J Duivenvoorden, J N van den Anker, D Tibboel...
Of the following, the BEST nutritional strategy for this infant would be to add:
 NeoReviewsPlus 2009 August 09 Assessment Your Score Page 1234 5678910 My Learning Plan 1 2 3 January 09 February 09 March 09 A 700-g infant regained her birthweight 14 days after birth. She is receiving
NeoReviewsPlus 2009 August 09 Assessment Your Score Page 1234 5678910 My Learning Plan 1 2 3 January 09 February 09 March 09 A 700-g infant regained her birthweight 14 days after birth. She is receiving
Pain Control in the NICU
 Pain Control in the NICU By Robert Cicco, MD Introduction There is ample documentation in the literature that newborn infants experience pain and exhibit a variety of both physiologic and behavioral responses
Pain Control in the NICU By Robert Cicco, MD Introduction There is ample documentation in the literature that newborn infants experience pain and exhibit a variety of both physiologic and behavioral responses
Haemodynamics of intravenous paracetamol in neonates
 Haemodynamics of intravenous paracetamol in neonates Karel Allegaert, Gunnar Naulaers To cite this version: Karel Allegaert, Gunnar Naulaers. Haemodynamics of intravenous paracetamol in neonates. European
Haemodynamics of intravenous paracetamol in neonates Karel Allegaert, Gunnar Naulaers To cite this version: Karel Allegaert, Gunnar Naulaers. Haemodynamics of intravenous paracetamol in neonates. European
Specialist neonatal respiratory care for babies born preterm
 National Institute for Health and Care Excellence Draft for Consultation Specialist neonatal respiratory care for babies born preterm [E] Evidence reviews for sedation and analgesia NICE guideline
National Institute for Health and Care Excellence Draft for Consultation Specialist neonatal respiratory care for babies born preterm [E] Evidence reviews for sedation and analgesia NICE guideline
Review Article Clinical Pharmacology of Midazolam in Neonates and Children: Effect of Disease A Review
 International Journal of Pediatrics, Article ID 309342, 20 pages http://dx.doi.org/10.1155/2014/309342 Review Article Clinical Pharmacology of Midazolam in Neonates and Children: Effect of Disease A Review
International Journal of Pediatrics, Article ID 309342, 20 pages http://dx.doi.org/10.1155/2014/309342 Review Article Clinical Pharmacology of Midazolam in Neonates and Children: Effect of Disease A Review
Ventilator-Associated Event Prevention: Innovations
 Ventilator-Associated Event Prevention: Innovations Michael J. Apostolakos, MD Professor of Medicine Director, Adult Critical Care University of Rochester Mobility/Sedation in the ICU Old teaching: Keep
Ventilator-Associated Event Prevention: Innovations Michael J. Apostolakos, MD Professor of Medicine Director, Adult Critical Care University of Rochester Mobility/Sedation in the ICU Old teaching: Keep
Sedation in Children
 CHILDREN S SERVICES Sedation in Children See text for full explanation and drug doses Patient for Sedation Appropriate staffing Resuscitation equipment available Monitoring equipment Patient suitability
CHILDREN S SERVICES Sedation in Children See text for full explanation and drug doses Patient for Sedation Appropriate staffing Resuscitation equipment available Monitoring equipment Patient suitability
Baby It Hurts. Deb Fraser, MN, RNC
 Baby It Hurts Deb Fraser, MN, RNC Outline What is pain? Misconceptions about pain Problems with neonatal pain management Pain assessment Our view of pain? definitions Pain: An unpleasant sensory or emotional
Baby It Hurts Deb Fraser, MN, RNC Outline What is pain? Misconceptions about pain Problems with neonatal pain management Pain assessment Our view of pain? definitions Pain: An unpleasant sensory or emotional
Sedation and delirium- drugs and clinical management
 Sedation and delirium- drugs and clinical management Shannon S. Carson, MD Associate Professor and Chief Division of Pulmonary and Critical Care Medicine University of North Carolina Probability of transitioning
Sedation and delirium- drugs and clinical management Shannon S. Carson, MD Associate Professor and Chief Division of Pulmonary and Critical Care Medicine University of North Carolina Probability of transitioning
DEEP SEDATION TEST QUESTIONS
 Mailing Address: Phone: Fax: The Study Guide is provided for those physicians eligible to apply for Deep Sedation privileges. The Study Guide is approximately 41 pages, so you may consider printing only
Mailing Address: Phone: Fax: The Study Guide is provided for those physicians eligible to apply for Deep Sedation privileges. The Study Guide is approximately 41 pages, so you may consider printing only
Interaction between Sedation and Weaning: How to Balance Them? Guillermo Castorena MD Fundacion Clinica Medica Sur Mexico
 Interaction between Sedation and Weaning: How to Balance Them? Guillermo Castorena MD Fundacion Clinica Medica Sur Mexico Balance is not that easy! Weaning Weaning is the liberation of a patient from
Interaction between Sedation and Weaning: How to Balance Them? Guillermo Castorena MD Fundacion Clinica Medica Sur Mexico Balance is not that easy! Weaning Weaning is the liberation of a patient from
MORPHINE ADMINISTRATION
 Introduction Individualised Administration Drug of Choice Route of Administration & Doses Monitoring of Neonates & high risk patients Team Management Responsibility Morphine Protocol Flow Chart Introduction
Introduction Individualised Administration Drug of Choice Route of Administration & Doses Monitoring of Neonates & high risk patients Team Management Responsibility Morphine Protocol Flow Chart Introduction
Patent Ductus Arteriosus: Philosophy or Pathology?
 Patent Ductus Arteriosus: Philosophy or Pathology? Disclosure Ray Sato, MD is a speaker for Prolacta Biosciences, Inc. This presentation will discuss off-label uses of acetaminophen and ibuprofen. RAY
Patent Ductus Arteriosus: Philosophy or Pathology? Disclosure Ray Sato, MD is a speaker for Prolacta Biosciences, Inc. This presentation will discuss off-label uses of acetaminophen and ibuprofen. RAY
Surfactant Administration
 Approved by: Surfactant Administration Gail Cameron Senior Director Operations, Maternal, Neonatal & Child Health Programs Dr. Paul Byrne Medical Director, Neonatology Neonatal Policy & Procedures Manual
Approved by: Surfactant Administration Gail Cameron Senior Director Operations, Maternal, Neonatal & Child Health Programs Dr. Paul Byrne Medical Director, Neonatology Neonatal Policy & Procedures Manual
CPAP failure in preterm infants: incidence, predictors and consequences
 CPAP failure in preterm infants: incidence, predictors and consequences SUPPLEMENTAL TEXT METHODS Study setting The Royal Hobart Hospital has an 11-bed combined Neonatal and Paediatric Intensive Care Unit
CPAP failure in preterm infants: incidence, predictors and consequences SUPPLEMENTAL TEXT METHODS Study setting The Royal Hobart Hospital has an 11-bed combined Neonatal and Paediatric Intensive Care Unit
POST-INTUBATION ANALGESIA AND SEDATION. August 2012 J Pelletier
 POST-INTUBATION ANALGESIA AND SEDATION August 2012 J Pelletier Intubated patients experience pain and anxiety Mechanical ventilation, endotracheal tube Blood draws, positioning, suctioning Surgical procedures,
POST-INTUBATION ANALGESIA AND SEDATION August 2012 J Pelletier Intubated patients experience pain and anxiety Mechanical ventilation, endotracheal tube Blood draws, positioning, suctioning Surgical procedures,
Date 8/95; Rev.12/97; 7/98; 2/99; 5/01, 3/03, 9/03; 5/04; 8/05; 3/07; 10/08; 10/09; 10/10 Manual of Administrative Policy Source Sedation Committee
 Code No. 711 Section Subject Moderate Sedation (formerly termed Conscious Sedation ) Date 8/95; Rev.12/97; 7/98; 2/99; 5/01, 3/03, 9/03; 5/04; 8/05; 3/07; 10/08; Manual of Administrative Policy Source
Code No. 711 Section Subject Moderate Sedation (formerly termed Conscious Sedation ) Date 8/95; Rev.12/97; 7/98; 2/99; 5/01, 3/03, 9/03; 5/04; 8/05; 3/07; 10/08; Manual of Administrative Policy Source
ULTIVA. Remifentanil hydrochloride
 ULTIVA Remifentanil hydrochloride QUALITATIVE AND QUANTITATIVE COMPOSITION Remifentanil for injection is a sterile, preservative-free, white to off white, lyophilised powder, to be reconstituted before
ULTIVA Remifentanil hydrochloride QUALITATIVE AND QUANTITATIVE COMPOSITION Remifentanil for injection is a sterile, preservative-free, white to off white, lyophilised powder, to be reconstituted before
The Impact of Sedation and Analgesia on the Developing Preterm Brain. Christopher McPherson, PharmD
 The Impact of Sedation and Analgesia on the Developing Preterm Brain Christopher McPherson, PharmD Disclosures I have no conflicts of interest to disclose. I will be discussing off-label or unapproved
The Impact of Sedation and Analgesia on the Developing Preterm Brain Christopher McPherson, PharmD Disclosures I have no conflicts of interest to disclose. I will be discussing off-label or unapproved
DATA SHEET. Remifentanil hydrochloride for injection1mg and 2mg (remifentanil base) vials
 DATA SHEET 1. PRODUCT NAME ULTIVA Remifentanil hydrochloride for injection1mg and 2mg (remifentanil base) vials 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Active substance Remifentanil hydrochloride 1mg
DATA SHEET 1. PRODUCT NAME ULTIVA Remifentanil hydrochloride for injection1mg and 2mg (remifentanil base) vials 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Active substance Remifentanil hydrochloride 1mg
An approach for dose finding of drugs in infants: sedation by midazolam studied using the continual reassessment method
 Br J Clin Pharmacol 1998; 46: 395 401 An approach for dose finding of drugs in infants: sedation by midazolam studied using the continual reassessment method Emmanuelle Fabre, 1 Sylvie Chevret, 2 Jean-François
Br J Clin Pharmacol 1998; 46: 395 401 An approach for dose finding of drugs in infants: sedation by midazolam studied using the continual reassessment method Emmanuelle Fabre, 1 Sylvie Chevret, 2 Jean-François
Pain & Sedation Management in PICU. Marut Chantra, M.D.
 Pain & Sedation Management in PICU Marut Chantra, M.D. Pain Diseases Trauma Procedures Rogers Textbook of Pediatric Intensive Care, 5 th ed, 2015 Emotional Distress Separation from parents Unfamiliar
Pain & Sedation Management in PICU Marut Chantra, M.D. Pain Diseases Trauma Procedures Rogers Textbook of Pediatric Intensive Care, 5 th ed, 2015 Emotional Distress Separation from parents Unfamiliar
DELIRIUM IN ICU: Prevention and Management. Milind Baldi
 DELIRIUM IN ICU: Prevention and Management Milind Baldi Contents Introduction Risk factors Assessment Prevention Management Introduction Delirium is a syndrome characterized by acute cerebral dysfunction
DELIRIUM IN ICU: Prevention and Management Milind Baldi Contents Introduction Risk factors Assessment Prevention Management Introduction Delirium is a syndrome characterized by acute cerebral dysfunction
Hazards and Benefits of Postnatal Steroids. David J. Burchfield, MD Professor and Chief, Neonatology University of Florida
 Hazards and Benefits of Postnatal Steroids David J. Burchfield, MD Professor and Chief, Neonatology University of Florida Disclosures I have no financial affiliations or relationships to disclose. I will
Hazards and Benefits of Postnatal Steroids David J. Burchfield, MD Professor and Chief, Neonatology University of Florida Disclosures I have no financial affiliations or relationships to disclose. I will
ULTIVA GlaxoSmithKline
 ULTIVA GlaxoSmithKline Remifentanil QUALITATIVE AND QUANTITATIVE COMPOSITION Remifentanil for injection is a sterile, endotoxin-free, preservative-free, white to off white, lyophilised powder, to be reconstituted
ULTIVA GlaxoSmithKline Remifentanil QUALITATIVE AND QUANTITATIVE COMPOSITION Remifentanil for injection is a sterile, endotoxin-free, preservative-free, white to off white, lyophilised powder, to be reconstituted
PHYSICIAN'S ORDERS Mark in for desired orders. If is blank, order is inactive. VENTILATOR SEDATION / ANALGESIC / DELIRIUM ORDER
 Nursing Daily awakenings PHYSICIAN'S ORDERS Mark in for desired orders. If is blank, order is inactive. VENTILATOR SEDATION / ANALGESIC / DELIRIUM ORDER Do not perform daily awakenings: Rationale: Daily
Nursing Daily awakenings PHYSICIAN'S ORDERS Mark in for desired orders. If is blank, order is inactive. VENTILATOR SEDATION / ANALGESIC / DELIRIUM ORDER Do not perform daily awakenings: Rationale: Daily
Optimal sedation and management of anxiety in patients undergoing endobronchial ultrasound (EBUS)
 Optimal sedation and management of anxiety in patients undergoing endobronchial ultrasound (EBUS) Georgios Dadoudis Anesthesiologist ICU DIRECTOR INTERBALKAN MEDICAL CENTER Optimal performance requires:
Optimal sedation and management of anxiety in patients undergoing endobronchial ultrasound (EBUS) Georgios Dadoudis Anesthesiologist ICU DIRECTOR INTERBALKAN MEDICAL CENTER Optimal performance requires:
PFIZER INC. THERAPEUTIC AREA AND FDA APPROVED INDICATIONS: See USPI.
 PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
FANNP 28TH NATIONAL NNP SYMPOSIUM: CLINICAL UPDATE AND REVIEW OCTOBER 17-21, 2017
 Pulse Oximetry in the Delivery Room: Principles and Practice GS2 3 Jonathan P. Mintzer, MD, FAAP Assistant Professor of Pediatrics Stony Brook Children s Hospital, Division of Neonatal-Perinatal Medicine,
Pulse Oximetry in the Delivery Room: Principles and Practice GS2 3 Jonathan P. Mintzer, MD, FAAP Assistant Professor of Pediatrics Stony Brook Children s Hospital, Division of Neonatal-Perinatal Medicine,
COMPARISON OF THE EFFICIENCY OF CAFFEINE VERSUS AMINOPHYLLINE FOR THE TREATMENT OF APNOEA OF PREMATURITY
 CASE STUDIES COMPARISON OF THE EFFICIENCY OF CAFFEINE VERSUS AMINOPHYLLINE FOR THE TREATMENT OF APNOEA OF PREMATURITY Gabriela Ildiko Zonda 1, Andreea Avasiloaiei 1, Mihaela Moscalu 2, Maria Stamatin 1
CASE STUDIES COMPARISON OF THE EFFICIENCY OF CAFFEINE VERSUS AMINOPHYLLINE FOR THE TREATMENT OF APNOEA OF PREMATURITY Gabriela Ildiko Zonda 1, Andreea Avasiloaiei 1, Mihaela Moscalu 2, Maria Stamatin 1
British Journal of Clinical Pharmacology
 British Journal of Clinical Pharmacology DOI:1.1111/j.1365-2125.8.3118.x An optimized ibuprofen dosing scheme for preterm neonates with patent ductus arteriosus, based on a population pharmacokinetic and
British Journal of Clinical Pharmacology DOI:1.1111/j.1365-2125.8.3118.x An optimized ibuprofen dosing scheme for preterm neonates with patent ductus arteriosus, based on a population pharmacokinetic and
Update on mangement of patent ductus arteriosus in preterm infants. Dr. Trinh Thi Thu Ha
 Update on mangement of patent ductus arteriosus in preterm infants Dr. Trinh Thi Thu Ha Outline 1. Overview of PDA 2. Timing of screening PDA? 3. When to treat PDA? Timing of ductal closure Prenatal
Update on mangement of patent ductus arteriosus in preterm infants Dr. Trinh Thi Thu Ha Outline 1. Overview of PDA 2. Timing of screening PDA? 3. When to treat PDA? Timing of ductal closure Prenatal
Sedation and Analgesia in the Critically Ill
 12th Congress of the World Federation of Societies of Intensive and Critical Care Medicine August 29 (Sat.) September 1 (Tue.), 2015 COEX, Seoul, Korea ONE STEP FURTHER: THE PURSUIT OF EXCELLENCE IN CRITICAL
12th Congress of the World Federation of Societies of Intensive and Critical Care Medicine August 29 (Sat.) September 1 (Tue.), 2015 COEX, Seoul, Korea ONE STEP FURTHER: THE PURSUIT OF EXCELLENCE IN CRITICAL
Newborn Life Support. NLS guidance.
 Kelly Harvey, ANNP NWNODN, previously Wythenshawe Hospital has shared this presentation with the understanding that it is for personal use following your attendance at the 8th Annual Senior Neonatal Nursing
Kelly Harvey, ANNP NWNODN, previously Wythenshawe Hospital has shared this presentation with the understanding that it is for personal use following your attendance at the 8th Annual Senior Neonatal Nursing
Printed copies of this document may not be up to date, obtain the most recent version from
 Children s Acute Transport Service Clinical Guidelines Septic Shock Document Control Information Author Claire Fraser P.Ramnarayan Author Position tanp CATS Consultant Document Owner E. Polke Document
Children s Acute Transport Service Clinical Guidelines Septic Shock Document Control Information Author Claire Fraser P.Ramnarayan Author Position tanp CATS Consultant Document Owner E. Polke Document
ADMINISTRATIVE POLICY AND PROCEDURE MANUAL. Subject: Moderate Sedation/Analgesia- Procedural ( Conscious Sedation ) Policy
 BRYN MAWR HOSPITAL LANKENAU HOSPITAL PAOLI HOSPITAL Working Together to Serve the Community ADMINISTRATE POLICY AND PROCEDURE MANUAL Subject: Moderate Sedation/Analgesia- Procedural ( Conscious Sedation
BRYN MAWR HOSPITAL LANKENAU HOSPITAL PAOLI HOSPITAL Working Together to Serve the Community ADMINISTRATE POLICY AND PROCEDURE MANUAL Subject: Moderate Sedation/Analgesia- Procedural ( Conscious Sedation
SWISS SOCIETY OF NEONATOLOGY. Prolonged arterial hypotension due to propofol used for endotracheal intubation in a newborn infant
 SWISS SOCIETY OF NEONATOLOGY Prolonged arterial hypotension due to propofol used for endotracheal intubation in a newborn infant July 2001 2 Wagner B, Intensive Care Unit, University Children s Hospital
SWISS SOCIETY OF NEONATOLOGY Prolonged arterial hypotension due to propofol used for endotracheal intubation in a newborn infant July 2001 2 Wagner B, Intensive Care Unit, University Children s Hospital
Addendum to the NRP Provider Textbook 6 th Edition Recommendations for specific modifications in the Canadian context
 Addendum to the NRP Provider Textbook 6 th Edition Recommendations for specific modifications in the Canadian context A subcommittee of the Canadian Neonatal Resuscitation Program (NRP) Steering Committee
Addendum to the NRP Provider Textbook 6 th Edition Recommendations for specific modifications in the Canadian context A subcommittee of the Canadian Neonatal Resuscitation Program (NRP) Steering Committee
Clinical Guideline: Premedication for non-emergency endotracheal intubation in the neonate
 Clinical Guideline: Premedication for non-emergency endotracheal intubation in the neonate EOE Neonatal ODN Guideline no: Clinical guideline: Author: Adapted/Modified from: NEO-ODN-2016-1 Premedication
Clinical Guideline: Premedication for non-emergency endotracheal intubation in the neonate EOE Neonatal ODN Guideline no: Clinical guideline: Author: Adapted/Modified from: NEO-ODN-2016-1 Premedication
Neonatal Life Support Provider (NLSP) Certification Preparatory Materials
 Neonatal Life Support Provider (NLSP) Certification Preparatory Materials NEONATAL LIFE SUPPORT PROVIDER (NRP) CERTIFICATION TABLE OF CONTENTS NEONATAL FLOW ALGORITHM.2 INTRODUCTION 3 ANTICIPATION OF RESUSCITATION
Neonatal Life Support Provider (NLSP) Certification Preparatory Materials NEONATAL LIFE SUPPORT PROVIDER (NRP) CERTIFICATION TABLE OF CONTENTS NEONATAL FLOW ALGORITHM.2 INTRODUCTION 3 ANTICIPATION OF RESUSCITATION
Infection. Risk factor for infection ACoRN alerting sign with * Clinical deterioration. Problem List. Respiratory. Cardiovascular
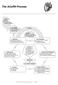 The ACoRN Process Baby at risk Unwell Risk factors Post-resuscitation requiring stabilization Resuscitation Ineffective breathing Heart rate < 100 bpm Central cyanosis Support Infection Risk factor for
The ACoRN Process Baby at risk Unwell Risk factors Post-resuscitation requiring stabilization Resuscitation Ineffective breathing Heart rate < 100 bpm Central cyanosis Support Infection Risk factor for
Morphine and morphine-6-glucuronide concentrations and their correlation with pain responses
 Archives of Perinatal Medicine 16(2), 92-96, 2010 ORGNAL PAPER Morphine and morphine-6-glucuronide concentrations and their correlation with pain responses JOANNA RÓŻYCKA 1, JANUSZ GADZNOWSK 1, DHARMAPUR
Archives of Perinatal Medicine 16(2), 92-96, 2010 ORGNAL PAPER Morphine and morphine-6-glucuronide concentrations and their correlation with pain responses JOANNA RÓŻYCKA 1, JANUSZ GADZNOWSK 1, DHARMAPUR
TRANSPARENCY COMMITTEE OPINION. 21 January 2009
 The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 21 January 2009 BRIDION 100 mg/ml, solution for injection Box containing 10 x 2 ml bottles, CIP: 573 553-9 Box containing
The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 21 January 2009 BRIDION 100 mg/ml, solution for injection Box containing 10 x 2 ml bottles, CIP: 573 553-9 Box containing
POST TEST: PROCEDURAL SEDATION
 POST TEST: PROCEDURAL SEDATION Name: Date: Instructions: Complete the Post-Test (an 85% is required to pass). If there are areas that you are unsure of, please review the relevant portions of the learning
POST TEST: PROCEDURAL SEDATION Name: Date: Instructions: Complete the Post-Test (an 85% is required to pass). If there are areas that you are unsure of, please review the relevant portions of the learning
PFIZER INC. Study Center(s): A total of 6 centers took part in the study, including 2 in France and 4 in the United States.
 PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
Noah Hillman M.D. IPOKRaTES Conference Guadalajaira, Mexico August 23, 2018
 Postnatal Steroids Use for Bronchopulmonary Dysplasia in 2018 + = Noah Hillman M.D. IPOKRaTES Conference Guadalajaira, Mexico August 23, 2018 AAP Policy Statement - 2002 This statement is intended for
Postnatal Steroids Use for Bronchopulmonary Dysplasia in 2018 + = Noah Hillman M.D. IPOKRaTES Conference Guadalajaira, Mexico August 23, 2018 AAP Policy Statement - 2002 This statement is intended for
PROFESSOR DR. NUMAN NAFIE HAMEED الاستاذ الدكتور نعمان نافع الحمداني
 Lecture 6 PROFESSOR DR. NUMAN NAFIE HAMEED الاستاذ الدكتور نعمان نافع الحمداني Neonatal Resuscitation Program (NRP) 2010 MCQ? In neonatal resuscitation program, the preterm neonates need special preparations
Lecture 6 PROFESSOR DR. NUMAN NAFIE HAMEED الاستاذ الدكتور نعمان نافع الحمداني Neonatal Resuscitation Program (NRP) 2010 MCQ? In neonatal resuscitation program, the preterm neonates need special preparations
University of Mississippi Medical Center University of Mississippi Health Care. Pharmacy and Therapeutics Committee Medication Use Evaluation
 University of Mississippi Medical Center University of Mississippi Health Care Pharmacy and Therapeutics Committee Medication Use Evaluation Cisatracurium (Nimbex ) and Vecuronium (Norcuron ) PURPOSE The
University of Mississippi Medical Center University of Mississippi Health Care Pharmacy and Therapeutics Committee Medication Use Evaluation Cisatracurium (Nimbex ) and Vecuronium (Norcuron ) PURPOSE The
MEDICAL ADVISORY COUNCIL Position Statement PREHOSPITAL PAIN MANAGEMENT
 MEDICAL ADVISORY COUNCIL Position Statement PREHOSPITAL PAIN MANAGEMENT MAC PS 2013-002 Appropriate treatment of acute pain in the prehospital arena offers an opportunity to positively impact many patients.
MEDICAL ADVISORY COUNCIL Position Statement PREHOSPITAL PAIN MANAGEMENT MAC PS 2013-002 Appropriate treatment of acute pain in the prehospital arena offers an opportunity to positively impact many patients.
Benefits of Caffeine Citrate: Neurodevelopmental Outcomes of ELBW Infants
 St. Catherine University SOPHIA Master of Arts in Nursing Theses Nursing 12-2011 Benefits of Caffeine Citrate: Neurodevelopmental Outcomes of ELBW Infants Teri Johnson St. Catherine University Follow this
St. Catherine University SOPHIA Master of Arts in Nursing Theses Nursing 12-2011 Benefits of Caffeine Citrate: Neurodevelopmental Outcomes of ELBW Infants Teri Johnson St. Catherine University Follow this
Pain Management in the NICU. Tamorah Lewis MD, PhD
 Pain Management in the NICU & Iatrogenic Opiate Withdrawal Tamorah Lewis MD, PhD Assistant Professor Divisions of Neonatology & Clinical Pharmacology, Toxicology and Therapeutic Innovation The Children's
Pain Management in the NICU & Iatrogenic Opiate Withdrawal Tamorah Lewis MD, PhD Assistant Professor Divisions of Neonatology & Clinical Pharmacology, Toxicology and Therapeutic Innovation The Children's
CLINICAL POLICY FOR THE USE OF INTRANASAL DIAMORPHINE FOR ANALGESIA IN CHILDREN ATTENDING THE PAEDIATRIC EMERGENCY DEPARTMENT, SASH
 CLINICAL POLICY FOR THE USE OF INTRANASAL DIAMORPHINE FOR ANALGESIA IN CHILDREN ATTENDING THE PAEDIATRIC EMERGENCY DEPARTMENT, SASH Background Adequate analgesia is a vital aspect of early management of
CLINICAL POLICY FOR THE USE OF INTRANASAL DIAMORPHINE FOR ANALGESIA IN CHILDREN ATTENDING THE PAEDIATRIC EMERGENCY DEPARTMENT, SASH Background Adequate analgesia is a vital aspect of early management of
PHYSICIAN COMPETENCY FOR ADULT DEEP SEDATION (Ages 14 and older)
 Name Score PHYSICIAN COMPETENCY FOR ADULT DEEP SEDATION (Ages 14 and older) 1. Pre-procedure evaluation for moderate sedation should involve all of the following EXCEPT: a) Airway Exam b) Anesthetic history
Name Score PHYSICIAN COMPETENCY FOR ADULT DEEP SEDATION (Ages 14 and older) 1. Pre-procedure evaluation for moderate sedation should involve all of the following EXCEPT: a) Airway Exam b) Anesthetic history
Results of a one-year, retrospective medication use evaluation. Joseph Ladd, PharmD PGY-1 Pharmacy Resident BHSF Homestead Hospital
 Results of a one-year, retrospective medication use evaluation Joseph Ladd, PharmD PGY-1 Pharmacy Resident BHSF Homestead Hospital Briefly review ketamine s history, mechanism of action, and unique properties
Results of a one-year, retrospective medication use evaluation Joseph Ladd, PharmD PGY-1 Pharmacy Resident BHSF Homestead Hospital Briefly review ketamine s history, mechanism of action, and unique properties
Sedation Hold/Interruption and Weaning Protocol ( Wake-up and Breathe )
 PROTOCOL Sedation Hold/Interruption and Weaning Protocol ( Wake-up and Breathe ) Page 1 of 6 Scope: Population: Outcome: Critical care clinicians and providers. All ICU patients intubated or mechanically
PROTOCOL Sedation Hold/Interruption and Weaning Protocol ( Wake-up and Breathe ) Page 1 of 6 Scope: Population: Outcome: Critical care clinicians and providers. All ICU patients intubated or mechanically
Satellite Symposium. Sponsored by
 Satellite Symposium Sponsored by Management of fluids and electrolytes in the preterm infant in the first week of life Pam Cairns St Michaels Hospital Bristol Healthy, term, breast fed babies Limited intake
Satellite Symposium Sponsored by Management of fluids and electrolytes in the preterm infant in the first week of life Pam Cairns St Michaels Hospital Bristol Healthy, term, breast fed babies Limited intake
Interprofessional Trauma Conference September 28th 2018 Montreal
 Interprofessional Trauma Conference September 28th 2018 Montreal Marc Perreault & Marc Alexandre Duceppe ICU Pharmacists MGH & RVH-CUSM Faculté de Pharmacie Université de Montréal I have no potential conflict
Interprofessional Trauma Conference September 28th 2018 Montreal Marc Perreault & Marc Alexandre Duceppe ICU Pharmacists MGH & RVH-CUSM Faculté de Pharmacie Université de Montréal I have no potential conflict
Sedation For Cardiac Procedures A Review of
 Sedation For Cardiac Procedures A Review of Sedative Agents Dr Simon Chan Consultant Anaesthesiologist Department of Anaesthesia and Intensive Care Prince of Wales Hospital Hong Kong 21 February 2009 Aims
Sedation For Cardiac Procedures A Review of Sedative Agents Dr Simon Chan Consultant Anaesthesiologist Department of Anaesthesia and Intensive Care Prince of Wales Hospital Hong Kong 21 February 2009 Aims
Ali Mohammadshahi 1, Ali Omraninava 1 *, Amir Masoud Hashemian 2 and Mohammad Mehdi Forouzanfar 3
 BIOSCIENCES BIOTECHNOLOGY RESEARCH ASIA, August 2014. Vol. 11(2), 993-997 Comparission Between Midazolam and Midazolam Plus Diphenhydramine Efficacy of Sedation in Children Undergoing CT-scan admitted
BIOSCIENCES BIOTECHNOLOGY RESEARCH ASIA, August 2014. Vol. 11(2), 993-997 Comparission Between Midazolam and Midazolam Plus Diphenhydramine Efficacy of Sedation in Children Undergoing CT-scan admitted
STARSHIP WITHDRAWAL OF ANALGESIA AND SEDATION
 STARSHIP WITHDRAWAL OF ANALGESIA AND SEDATION Patients receiving analgesia and/or sedation for longer than 5-7 days may suffer withdrawal if these drugs are suddenly stopped. To prevent this happening
STARSHIP WITHDRAWAL OF ANALGESIA AND SEDATION Patients receiving analgesia and/or sedation for longer than 5-7 days may suffer withdrawal if these drugs are suddenly stopped. To prevent this happening
GE Healthcare. Non Invasive Ventilation (NIV) For the Engström Ventilator. Relief, Relax, Recovery
 GE Healthcare Non Invasive Ventilation (NIV) For the Engström Ventilator Relief, Relax, Recovery COPD is currently the fourth leading cause of death in the world, and further increases in the prevalence
GE Healthcare Non Invasive Ventilation (NIV) For the Engström Ventilator Relief, Relax, Recovery COPD is currently the fourth leading cause of death in the world, and further increases in the prevalence
Etomidate is a short-acting, sedative hypnotic
 Hosp Pharm 2014;49(2):177 183 2014 Thomas Land Publishers, Inc. www.hospital-pharmacy.com doi: 10.1310/hpj4902-177 Original Article Effects of Etomidate on Adrenal Suppression: A Review of Intubated Septic
Hosp Pharm 2014;49(2):177 183 2014 Thomas Land Publishers, Inc. www.hospital-pharmacy.com doi: 10.1310/hpj4902-177 Original Article Effects of Etomidate on Adrenal Suppression: A Review of Intubated Septic
Addendum D. Procedural Sedation Test MERCY MEDICAL CENTER- SIOUX CITY. Procedural Sedation Questions
 Addendum D. Procedural Sedation Test MERCY MEDICAL CENTER- SIOUX CITY Procedural Sedation Questions Individuals applying for moderate sedation privileges must achieve a score of 80%. PRACTITIONER NAME
Addendum D. Procedural Sedation Test MERCY MEDICAL CENTER- SIOUX CITY Procedural Sedation Questions Individuals applying for moderate sedation privileges must achieve a score of 80%. PRACTITIONER NAME
Awake regional versus general anesthesia in preterms and ex-preterm infants for herniotomy
 Awake regional versus general anesthesia in preterms and ex-preterm infants for herniotomy Department of Anaesthesia University Children s Hospital Zurich Switzerland Epidemiology Herniotomy needed in
Awake regional versus general anesthesia in preterms and ex-preterm infants for herniotomy Department of Anaesthesia University Children s Hospital Zurich Switzerland Epidemiology Herniotomy needed in
Uses 1,2,3 : Labeled: Prevention of respiratory distress syndrome in premature infants
 Brand Name: Surfaxin Generic Name: lucinactant Manufacturer 1 : Discovery Laboratories, Inc. Drug Class 2,3 : Synthetic lung surfactant Uses 1,2,3 : Labeled: Prevention of respiratory distress syndrome
Brand Name: Surfaxin Generic Name: lucinactant Manufacturer 1 : Discovery Laboratories, Inc. Drug Class 2,3 : Synthetic lung surfactant Uses 1,2,3 : Labeled: Prevention of respiratory distress syndrome
TRAINING NEONATOLOGY SILVANA PARIS
 TRAINING ON NEONATOLOGY SILVANA PARIS RESUSCITATION IN DELIVERY ROOM INTRODUCTION THE GLOBAL RESUSCITATION BURDEN IN NEWBORN 136 MILL NEWBORN BABIES EACH YEAR (WHO WORLD REPORT) 5-8 MILL NEWBORN INFANTS
TRAINING ON NEONATOLOGY SILVANA PARIS RESUSCITATION IN DELIVERY ROOM INTRODUCTION THE GLOBAL RESUSCITATION BURDEN IN NEWBORN 136 MILL NEWBORN BABIES EACH YEAR (WHO WORLD REPORT) 5-8 MILL NEWBORN INFANTS
CHEST PHYSIOTHERAPY IN NICU PURPOSE POLICY STATEMENTS SITE APPLICABILITY PRACTICE LEVEL/COMPETENCIES. The role of chest physiotherapy in the NICU
 PURPOSE The role of chest physiotherapy in the NICU POLICY STATEMENTS In principle chest physiotherapy should be limited to those infants considered most likely to benefit with significant respiratory
PURPOSE The role of chest physiotherapy in the NICU POLICY STATEMENTS In principle chest physiotherapy should be limited to those infants considered most likely to benefit with significant respiratory
FOR REPRESENTATIVE EDUCATION
 Neuromuscular Blockade in the ICU NIMBEX Indication 1 NIMBEX (cisatracurium besylate) is indicated as an adjunct to general anesthesia to facilitate tracheal intubation in adults and in pediatric patients
Neuromuscular Blockade in the ICU NIMBEX Indication 1 NIMBEX (cisatracurium besylate) is indicated as an adjunct to general anesthesia to facilitate tracheal intubation in adults and in pediatric patients
POLICY and PROCEDURE
 Misericordia Community Hospital Administration of Intravenous FentaNYL During Labour POLICY and PROCEDURE Labour and Delivery Manual Original Date Revised Date Approved by: Director, Women s Health, Covenant
Misericordia Community Hospital Administration of Intravenous FentaNYL During Labour POLICY and PROCEDURE Labour and Delivery Manual Original Date Revised Date Approved by: Director, Women s Health, Covenant
Pharmacodynamics of midazolam in pediatric intensive care patients. Chapter
 Pharmacodynamics of midazolam in pediatric intensive care patients 11 Chapter S.N. de Wildt, M. de Hoog, A.A. Vinks, K.F.M. Joosten, J.N. van den Anker Aim Methods Results Discussion Summary To aim of
Pharmacodynamics of midazolam in pediatric intensive care patients 11 Chapter S.N. de Wildt, M. de Hoog, A.A. Vinks, K.F.M. Joosten, J.N. van den Anker Aim Methods Results Discussion Summary To aim of
INTRAVENOUS FLUIDS PRINCIPLES
 INTRAVENOUS FLUIDS PRINCIPLES Postnatal physiological weight loss is approximately 5-10% Postnatal diuresis is delayed in Respiratory Distress Syndrome (RDS) Preterm babies have limited capacity to excrete
INTRAVENOUS FLUIDS PRINCIPLES Postnatal physiological weight loss is approximately 5-10% Postnatal diuresis is delayed in Respiratory Distress Syndrome (RDS) Preterm babies have limited capacity to excrete
Objectives. Apnea Definition and Pitfalls. Pathophysiology of Apnea. Apnea of Prematurity and hypoxemia episodes 5/18/2015
 Apnea of Prematurity and hypoxemia episodes Deepak Jain MD Care of Sick Newborn Conference May 2015 Objectives Differentiating between apnea and hypoxemia episodes. Pathophysiology Diagnosis of apnea and
Apnea of Prematurity and hypoxemia episodes Deepak Jain MD Care of Sick Newborn Conference May 2015 Objectives Differentiating between apnea and hypoxemia episodes. Pathophysiology Diagnosis of apnea and
New Zealand Data Sheet. Poractant alfa (Phospholipid fraction of porcine lung) 80 mg/ml
 CUROSURF New Zealand Data Sheet Poractant alfa (Phospholipid fraction of porcine lung) 80 mg/ml Presentation Sterile suspension in single-dose vials for intratracheal or intrabronchial administration.
CUROSURF New Zealand Data Sheet Poractant alfa (Phospholipid fraction of porcine lung) 80 mg/ml Presentation Sterile suspension in single-dose vials for intratracheal or intrabronchial administration.
Surviving Sepsis Campaign. Guidelines for Management of Severe Sepsis/Septic Shock. An Overview
 Surviving Sepsis Campaign Guidelines for Management of Severe Sepsis/Septic Shock An Overview Mechanical Ventilation of Sepsis-Induced ALI/ARDS ARDSnet Mechanical Ventilation Protocol Results: Mortality
Surviving Sepsis Campaign Guidelines for Management of Severe Sepsis/Septic Shock An Overview Mechanical Ventilation of Sepsis-Induced ALI/ARDS ARDSnet Mechanical Ventilation Protocol Results: Mortality
Pharmacokinetics and pharmacology of drugs in children
 Pharmacokinetics and pharmacology of drugs in children Saskia N. de Wildt Professor of Clinical Pharmacology Pediatric Intensivist-Clinical Pharmacologist Understanding pediatric PK and PD Absorption 1.
Pharmacokinetics and pharmacology of drugs in children Saskia N. de Wildt Professor of Clinical Pharmacology Pediatric Intensivist-Clinical Pharmacologist Understanding pediatric PK and PD Absorption 1.
Surgical Care at the District Hospital. EMERGENCY & ESSENTIAL SURGICAL CARE
 Surgical Care at the District Hospital 1 14 Practical Anesthesia Key Points 2 14.1 General Anesthesia Have a clear plan before starting anesthesia Never use an unfamiliar anesthetic technique in an emergency
Surgical Care at the District Hospital 1 14 Practical Anesthesia Key Points 2 14.1 General Anesthesia Have a clear plan before starting anesthesia Never use an unfamiliar anesthetic technique in an emergency
Prolonged opioid therapy in the Critically Ill Pediatric Patient
 PBLD Table #22 Prolonged opioid therapy in the Critically Ill Pediatric Patient Arlyne K. Thung, M.D. Assistant Professor, Anesthesiology & Shu-Ming Wang, M.D. Associate Professor, Anesthesiology Yale
PBLD Table #22 Prolonged opioid therapy in the Critically Ill Pediatric Patient Arlyne K. Thung, M.D. Assistant Professor, Anesthesiology & Shu-Ming Wang, M.D. Associate Professor, Anesthesiology Yale
Procedural Sedation and Analgesia in the ED
 Overview Procedural Sedation and Analgesia in the ED Susan Lambe, MD Assistant Clinical Professor UCSF Division of Emergency Medicine Terminology Goals Indications Presedation Assessment Consent Issues
Overview Procedural Sedation and Analgesia in the ED Susan Lambe, MD Assistant Clinical Professor UCSF Division of Emergency Medicine Terminology Goals Indications Presedation Assessment Consent Issues
Responding to The Joint Commission Alert on Safe Use of Opioids in Hospitals
 Responding to The Joint Commission Alert on Safe Use of Opioids in Hospitals Suzanne A Nesbit, PharmD, CPE Clinical Pharmacy Specialist, Pain Management The Johns Hopkins Hospital Objectives and Disclosures
Responding to The Joint Commission Alert on Safe Use of Opioids in Hospitals Suzanne A Nesbit, PharmD, CPE Clinical Pharmacy Specialist, Pain Management The Johns Hopkins Hospital Objectives and Disclosures
ORAL GLUCOSE SOLUTION BEFORE VENEPUNCTURE FOR NEONATAL PAIN RELIEF
 ORAL GLUCOSE SOLUTION BEFORE VENEPUNCTURE FOR NEONATAL PAIN RELIEF DR I Wayan Retayasa Department of Child Health Wangaya General Hospital Medical Faculty Udayana University Denpasar Bali Indonesia Training
ORAL GLUCOSE SOLUTION BEFORE VENEPUNCTURE FOR NEONATAL PAIN RELIEF DR I Wayan Retayasa Department of Child Health Wangaya General Hospital Medical Faculty Udayana University Denpasar Bali Indonesia Training
AUSTRALIAN PRODUCT INFORMATION REMIFENTANIL APOTEX (REMIFENTANIL HYDROCHLORIDE) POWDER FOR INJECTION
 AUSTRALIAN PRODUCT INFORMATION REMIFENTANIL APOTEX (REMIFENTANIL HYDROCHLORIDE) POWDER FOR INJECTION 1 NAME OF THE MEDICINE Remifentanil (as hydrochloride) 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each
AUSTRALIAN PRODUCT INFORMATION REMIFENTANIL APOTEX (REMIFENTANIL HYDROCHLORIDE) POWDER FOR INJECTION 1 NAME OF THE MEDICINE Remifentanil (as hydrochloride) 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each
PATENT DUCTUS ARTERIOSUS IN THE PRETERM INFANT EVIDENCE FOR & AGAINST TREATMENT
 PATENT DUCTUS ARTERIOSUS IN THE PRETERM INFANT EVIDENCE FOR & AGAINST TREATMENT Dr. Youssef Abou Zanouna, FRCPI, FACC Consultant Pediatric Cardiologist King Fahd Military Medical Complex Dhahran Introduction
PATENT DUCTUS ARTERIOSUS IN THE PRETERM INFANT EVIDENCE FOR & AGAINST TREATMENT Dr. Youssef Abou Zanouna, FRCPI, FACC Consultant Pediatric Cardiologist King Fahd Military Medical Complex Dhahran Introduction
Name and title of the investigators responsible for conducting the research: Dr Anna Lavizzari, Dr Mariarosa Colnaghi
 Protocol title: Heated, Humidified High-Flow Nasal Cannula vs Nasal CPAP for Respiratory Distress Syndrome of Prematurity. Protocol identifying number: Clinical Trials.gov NCT02570217 Name and title of
Protocol title: Heated, Humidified High-Flow Nasal Cannula vs Nasal CPAP for Respiratory Distress Syndrome of Prematurity. Protocol identifying number: Clinical Trials.gov NCT02570217 Name and title of
Tarek M Sarhan, Assistant professor of Anesthesiology, Faculty of Medicine, Alexandria University
 7 ANALGESIA FOR TRACHEOESOPHAGEAL FISTULA REPAIR IN NEONATES : A COMPARISON OF SINGLE SHOT THORACIC PARAVERTEBRAL BLOCK AND EPIDURAL BLOCK WITH ROPIVACAINE Tarek M Sarhan, Assistant professor of Anesthesiology,
7 ANALGESIA FOR TRACHEOESOPHAGEAL FISTULA REPAIR IN NEONATES : A COMPARISON OF SINGLE SHOT THORACIC PARAVERTEBRAL BLOCK AND EPIDURAL BLOCK WITH ROPIVACAINE Tarek M Sarhan, Assistant professor of Anesthesiology,
CLINICAL PROCEDURE MUHC
 CLINICAL PROCEDURE MUHC Medication included No Medication included THIS IS NOT A MEDICAL ORDER Title: This procedure is attached to: Administration of sucrose 24% in pediatric population MUHC Patient Double
CLINICAL PROCEDURE MUHC Medication included No Medication included THIS IS NOT A MEDICAL ORDER Title: This procedure is attached to: Administration of sucrose 24% in pediatric population MUHC Patient Double
Disclosure. Hospira Pharmaceuticals. Unrestricted research funding Honoraria for CME education administered via France Foundation
 Disclosure Hospira Pharmaceuticals Unrestricted research funding Honoraria for CME education administered via France Foundation Economics in Sedation: Responsible Use of the ICU Budget John W. Devlin,
Disclosure Hospira Pharmaceuticals Unrestricted research funding Honoraria for CME education administered via France Foundation Economics in Sedation: Responsible Use of the ICU Budget John W. Devlin,
Source of effectiveness data The evidence for the final outcomes and resource use were derived from a single study.
 Gastroenterologist-administered propofol versus meperidine and midazolam for advanced upper endoscopy: a prospective, randomized trial Vargo J J, Zuccaro G, Dumot J A, Shermock K M, Morrow J B, Conwell
Gastroenterologist-administered propofol versus meperidine and midazolam for advanced upper endoscopy: a prospective, randomized trial Vargo J J, Zuccaro G, Dumot J A, Shermock K M, Morrow J B, Conwell
A SYNOPSIS BY ILCOR PEDIATRIC TASK FORCE. Pediatric Basic Life Support, Pediatric Advanced Life Support and Neonatal Resuscitation 2015
 Vol. 2 - No.4 October - December 2015 83 Vol. 2 - No.4 October - December 2015 84 There is new evidence that when treating pediatric septic shock in specific settings, the use of restricted volume of isotonic
Vol. 2 - No.4 October - December 2015 83 Vol. 2 - No.4 October - December 2015 84 There is new evidence that when treating pediatric septic shock in specific settings, the use of restricted volume of isotonic
Supportive Care. End of Life Phase
 Supportive Care End of Life Phase Guidelines for Health Care Professionals In the care of patients with established renal failure who are in the last days of life References: Chambers E J (2004) End of
Supportive Care End of Life Phase Guidelines for Health Care Professionals In the care of patients with established renal failure who are in the last days of life References: Chambers E J (2004) End of
ASSESSMENT OF THE PAEDIATRIC NEEDS ANAESTHESIOLOGY DISCLAIMER
 European Medicines Agency Evaluation of Medicines for Human Use ASSESSMENT OF THE PAEDIATRIC NEEDS ANAESTHESIOLOGY London, October 2006 Doc. Ref: EMEA/405166/2006 DISCLAIMER The Paediatric Working Party
European Medicines Agency Evaluation of Medicines for Human Use ASSESSMENT OF THE PAEDIATRIC NEEDS ANAESTHESIOLOGY London, October 2006 Doc. Ref: EMEA/405166/2006 DISCLAIMER The Paediatric Working Party
I. Subject. Moderate Sedation
 I. Subject II. III. Moderate Sedation Purpose To establish criteria for the monitoring and management of patients receiving moderate throughout the hospital Definitions A. Definitions of three levels of
I. Subject II. III. Moderate Sedation Purpose To establish criteria for the monitoring and management of patients receiving moderate throughout the hospital Definitions A. Definitions of three levels of
5 Million neonatal deaths each year worldwide. 20% caused by neonatal asphyxia. Improvement of the outcome of 1 million newborns every year
 1 5 Million neonatal deaths each year worldwide 20% caused by neonatal asphyxia Improvement of the outcome of 1 million newborns every year International Liaison Committee on Resuscitation (ILCOR) American
1 5 Million neonatal deaths each year worldwide 20% caused by neonatal asphyxia Improvement of the outcome of 1 million newborns every year International Liaison Committee on Resuscitation (ILCOR) American
Ventilated children are often sedated in order
 Paediatrica Indonesiana Volume 5 January 0 Number Original Article Continuous sedation vs. daily sedation interruption in mechanically-ventilated children Henri Azis, Silvia Triratna, Erial Bahar Abstract
Paediatrica Indonesiana Volume 5 January 0 Number Original Article Continuous sedation vs. daily sedation interruption in mechanically-ventilated children Henri Azis, Silvia Triratna, Erial Bahar Abstract
Effective dose of nefopam in 80% of patients (ED 80 ): a study using the continual reassessment method
 British Journal of Clinical Pharmacology DOI:10.1046/j.0306-5251.2007.02960.x Effective dose of nefopam in 80% of patients (ED 80 ): a study using the continual reassessment method Hélène Beloeil, Mathilde
British Journal of Clinical Pharmacology DOI:10.1046/j.0306-5251.2007.02960.x Effective dose of nefopam in 80% of patients (ED 80 ): a study using the continual reassessment method Hélène Beloeil, Mathilde
Weaning and extubation in PICU An evidence-based approach
 Weaning and extubation in PICU An evidence-based approach Suchada Sritippayawan, MD. Div. Pulmonology & Crit Care Dept. Pediatrics Faculty of Medicine Chulalongkorn University Kanokporn Udomittipong, MD.
Weaning and extubation in PICU An evidence-based approach Suchada Sritippayawan, MD. Div. Pulmonology & Crit Care Dept. Pediatrics Faculty of Medicine Chulalongkorn University Kanokporn Udomittipong, MD.
Siesta: Should Dexmedetomidine Be Used for Sedation in the Neonatal Intensive Care Unit?
 Siesta: Should Dexmedetomidine Be Used for Sedation in the Neonatal Intensive Care Unit? Letitia M. DeLaine, B.S., Pharm.D. PGY1 Pharmacy Resident 2013 2014 Children s Hospital of San Antonio/CHRISTUS
Siesta: Should Dexmedetomidine Be Used for Sedation in the Neonatal Intensive Care Unit? Letitia M. DeLaine, B.S., Pharm.D. PGY1 Pharmacy Resident 2013 2014 Children s Hospital of San Antonio/CHRISTUS
Hummi Micro Draw Blood Transfer Device. An Important Addition to Your IVH Bundle
 Hummi Micro Draw Blood Transfer Device An Important Addition to Your IVH Bundle Hummi Micro Draw & Micro T Connector For Infec6on Control and IVH Risk Reduc6on The Next Genera6on System for Closed Micro
Hummi Micro Draw Blood Transfer Device An Important Addition to Your IVH Bundle Hummi Micro Draw & Micro T Connector For Infec6on Control and IVH Risk Reduc6on The Next Genera6on System for Closed Micro
