Adrenal adrenaline- and noradrenaline-containing cells and. celiac sympathetic ganglia are differentially controlled by
|
|
|
- Frederica Goodman
- 5 years ago
- Views:
Transcription
1 * Manuscript Adrenal adrenaline- and noradrenaline-containing cells and celiac sympathetic ganglia are differentially controlled by centrally administered corticotropin-releasing factor and arginine-vasopressin in rats Naoko Yamaguchi-Shima a,*, Shoshiro Okada a, Takahiro Shimizu a, Daisuke Usui a,b, Kumiko Nakamura a, Lianyi Lu a, Kunihiko Yokotani a a Department of Pharmacology, Graduate School of Medicine, Kochi University, Nankoku, Kochi , Japan bdepartment of Pediatrics, Graduate School of Medicine, Kochi University, Nankoku, Kochi , Japan * Correspondence to: Naoko Yamaguchi-Shima, Department of Pharmacology, Graduate School of Medicine, Kochi University, Nankoku, Kochi , Japan. Tel and Fax: address: ynaoko@kochi-u.ac.jp 1
2 Abstract The adrenal glands and sympathetic celiac ganglia are innervated mainly by the greater splanchnic nerves, which contain preganglionic sympathetic nerves originated from thoracic spinal cord. The adrenal medulla has two separate populations of chromaffin cells, adrenaline-containing cells (Acells) and noradrenaline-containing cells (NA-cells), which have been shown to be differentially innervated by separate groups of the preganglionic sympathetic neurons. The present study was designed to characterize the centrally activating mechanisms of the adrenal A-cells, NA-cells and celiac sympathetic ganglia with expression of cfos (a marker for neural excitation), in regard to the brain prostanoids, in anesthetized rats. Intracerebroventricularly (i.c.v.) administered corticotropinreleasing factor (CRF) induced cfos expression in the adrenal A- cells, but not NA-cells, and celiac ganglia. On the other hand, i.c.v. administered arginine-vasopressin (AVP) resulted in cfos induction in both A-cells and NA-cells in the adrenal medulla, but not in the celiac ganglia. Intracerebroventricular pretreatment with indomethacin (an inhibitor of cyclooxygenase) abolished the CRF- and AVP-induced cfos expression in all regions described above. On the other hand, intracerebroventricular pretreatment with furegrelate (an inhibitor of thromboxane A 2 synthase) abolished the CRF-induced cfos expression in the adrenal A-cells, but not in the celiac ganglia, and also abolished the AVP-induced cfos expression in both A-cells and NA-cells in the adrenal medulla. These results suggest that centrally administered CRF activates adrenal A- 2
3 cells and celiac sympathetic ganglia by brain thromboxane A 2 - mediated and other prostanoid than thromboxane A 2 (probably prostaglandin E 2 )-mediated mechanisms, respectively. On the other hand, centrally administered AVP activates adrenal A-cells and NA-cells by brain thromboxane A 2 -mediated mechanisms in rats. Key words: Brain arachidonic acid cascade; Brain thromboxane A 2 ; cfos expression; Central sympatho-adrenomedullary outflow 3
4 1. Introduction The relative importance of the adrenal medulla and sympathetic nervous system in various pathophysiological conditions has generally been inferred from measurement of plasma adrenaline and noradrenaline, since plasma adrenaline and noradrenaline have been shown to reflect the activity of adrenal medulla and sympathetic nervous system, respectively. Hypoglycemia causes the elevation of plasma adrenaline (Young et al., 1984; Fujino and Fujii, 1995; Vollmer et al., 1997), while hypotension elevates both catecholamines (noradrenaline > adrenaline) (Brown and Fisher, 1984; Vollmer et al., 2000). Immobilization stress elicits a robust increase in the plasma adrenaline and noradrenaline (Kvetnansky and Mikulaj, 1970; Jezova et al., 1999; Dronjak et al., 2004), while cold stress triggers a robust increase in plasma noradrenaline (Dronjak et al., 2004). Likewise, centrally administered neuropeptides such as corticotropin-releasing factor (CRF), arginine-vasopressin (AVP), bombesin, thyrotropin-releasing hormone, and -calcitonin gene-related peptide also produce differential changes in the plasma catecholamines (Brown et al., 1979, 1985; Fisher et al., 1983; Brown and Fisher, 1984; Feuerstein et al., 1984; Hasegawa et al., 1993; Okuma et al., 1996; Yokotani et al., 2001; Okada et al., 2002). In the adrenal medulla of both humans and rats, adrenaline and noradrenaline are localized in separate populations of cells with the number of adrenaline-containing cells (A-cells) about 4-fold greater than that of noradrenaline-containing cells (NAcells) (Verhofstad et al., 1985; Suzuki and Kachi, 1996). 4
5 Despite the constancy of the stored amounts of adrenaline and noradrenaline, the proportion of each catecholamine secreted seems to vary depending on the strength and type of stimulus, suggesting a differential control of A-cells and NA-cells (Vollmer, 1996). Recently, adrenal A- and NA-cells have been shown to be innervated by separate groups of preganglionic sympathetic neurons located in the spinal cord (Edwards et al., 1996; Vollmer et al., 2000). Adrenaline secreted into circulation is almost produced within the adrenal A-cells (Axelrod, 1962; Wurtman, 2002), while plasma noradrenaline seems to reflect the secretion from adrenal NA-cells in addition to the release from sympathetic nerves (Folkow and von Euler, 1954; Vollmer et al., 1997; Yokotani et al., 2001, 2005; Okada et al., 2003a). However, little is known about centrally controlling mechanisms of adrenal A-cells, NA-cells and sympathetic nerves. Arachidonic acid is metabolized rapidly to oxygenated products, such as prostaglandin E 2 and thromboxane A 2, by several distinct enzymes including cyclooxygenase, prostaglandin E synthase and thromboxane A synthase (Flower and Blackwell, 1976; Irvine, 1982; Axelrod, 1990). Previously, we reported that centrally administered prostaglandin E 2 evokes noradrenaline release from sympathetic nerves by activation of the brain prostanoid EP 3 receptors (Yokotani et al., 1995, 2005). Recently we also reported that centrally administered CRF evokes the brain thromboxane A 2 -mediated adrenal adrenaline secretion and the other prostanoid (probably prostaglandin E 2 )-mediated sympathetic noradrenaline release, while centrally administered AVP, bombesin and histamine evoke the brain thromboxane A 2-5
6 mediated adrenal secretion of both adrenaline and noradrenaline in rats (Yokotani et al., 2001, 2005; Okada et al., 2003a; Shimizu et al., 2006). Since the greater splanchnic nerve, which contains sympathetic preganglionic nerves originated from spinal cord, ramifies into the adrenal gland and celiac sympathetic ganglia (Yokotani et al., 1983), these regions are very useful to identify the centrally activated adrenal A-, NA-cells and celiac sympathetic ganglia with nuclear expression of cfos, immediate early gene product (Sagar et al., 1988; Herrera and Robertson, 1996). In the present study, therefore, we examined the effects of centrally administered CRF and AVP on the cfos expression in these regions, in regard to the brain arachidonic acid cascade, to further clarify the presence of differential activating mechanisms for sympathetic nerves and adrenal medulla. 6
7 2. Materials and methods 2.1. Animals Male Wistar rats weighing about 350 g were maintained in an air-conditioned room at C under a constant day night rhythm for more than 2 weeks and given food (laboratory chow, CE-2; Clea Japan, Hamamatsu, Japan) and water ad libitum. All effort was made to minimize animal suffering and the number of animals used. All rats were treated in accordance with the Guiding Principles for the Care and Use of laboratory animals approved by the Graduate School of Medicine, Kochi University Experimental design Under urethane anesthesia (1.2 g/kg, i.p.), the animal was placed in a stereotaxic apparatus, as shown in our previous paper (Yokotani et al., 2001). The skull was drilled for intracerebroventricular administration of test substances using stainless-steel cannula (0.3 mm outer diameter) or a double lumens cannula (0.5 mm outer diameter). The stereotaxic coordinates of the tip of cannula were as follows (in mm): AP - 0.8, L 1.5, V 4.0 (AP, anterior from the bregma; L, lateral from the midline; V, below the surface of the brain), according to the rat brain atlas (Paxinos and Watson, 1986). Then, 3 h were allowed to elapse before the application of test reagents. The peptides (CRF and AVP) were slowly injected into the right lateral ventricle in a volume of 10 µl using a 10 µl Hamilton syringe. CRF and AVP were dissolved in sterile saline. Water- 7
8 soluble indomethacin-na and furegrelate dissolved in sterile saline were also administered into the right lateral ventricle in a volume of 10 µl 30 min before the application of CRF or AVP. Correct placement of the cannula was confirmed at the end of each experiment by verifying that a blue dye, injected through the cannula, had spread throughout the entire ventricular system. After several periods of time from injection of these peptide (CRF and AVP) into the right lateral ventricle (at 0, 10, 60 or 120 min after injection of CRF; at 0, 10 or 60 min after injection of AVP), each rat was perfused through the left cardiac ventricle with 100 ml of 0.1 M phosphate buffered saline (ph 7.4) followed by 300 ml of ice-cold 4% paraformaldehyde in 0.1 M phosphate buffer (ph 7.4), as shown in our previous paper (Shima et al., 2003). Then, celiac ganglia around the celiac artery and adrenal glands were immediately removed, post-fixed over night with the same fixative for the celiac ganglia or with 3% potassium dichromate and 4% paraformaldehyde in 0.1 M phosphate buffer for the adrenal gland with a slight modification of the previously reported methods (Coupland et al., 1964; Piezzi et al., 1983), and then cryoprotected in 20% sucrose in 0.1 M phosphate buffer for 24 h at 4 C. Frozen sections (20 µm-thickness) were cut on a cryostat, thaw-mounted on silane-coated slides, and then stored at -80 C until used Immunohistochemistry Sections on the slides were washed in phosphate buffered saline and then pretreated with 5% normal donkey serum (for 8
9 adrenal glands) or normal goat serum (for celiac ganglia) in phosphate buffered saline containing 0.3% Triton X-100 for 1 h at room temperature to eliminate non-specific antibody binding. To codetect by double-immunostaining of cfos/phenylethanolamine N-methyltransferase (PNMT) in the adrenal medulla or cfos/dopamine-β-hydroxylase (DBH) in the celiac ganglia, sections were incubated in a cocktail of rabbit anti-cfos (1:500) and sheep anti-pnmt (1:200) antibodies for adrenal glands, or rabbit anti-cfos (1:500) and mouse anti-dbh (1:500) antibodies for celiac ganglia for 4 h at room temperature. After incubation, the sections were washed three times for 10 min each in phosphate buffered saline, and then incubated for 2 h at room temperature in a mixture of secondary antisera; fluorescein isothiocyanate-conjugated donkey anti-rabbit IgG (1:200) and rhodamine-conjugated donkey anti-sheep IgG (1:200), or fluorescein isothiocyanate-conjugated goat anti-rabbit IgG (1:200) and rhodamine-conjugated goat anti-mouse IgG (1:200). After washing in phosphate buffered saline, nuclei were counterstained with 4,6-diamidine-2 -phenylindole dihydrochloride (1:1000) using sections from adrenal glands for chromaffin cell counting. After these procedures, the sections were coverslipped using mounting medium VECTASHIELD. All antisera were diluted in phosphate buffered saline containing 0.3% Triton X-100 and 10% normal serum produced in the animal species from which the respective secondary antibodies were obtained. Control experiments were performed by omitting primary antibodies as a test of cross-reactivity of secondary antibodies, and resulted in the absence of staining. 9
10 2.4. Image analysis and statistics Photomicrographs were captured using a digital camera (DP70, Olympus, Tokyo, Japan) attached to a fluorescent microscope (AX70, Olympus). The chromaffin reaction method demonstrated chromaffin cells with a yellow cytoplasmic staining. In addition, immunohistochemical method for PNMT divided chromaffin cells into two cell-types, A-cells (PNMT-positive) and NA-cells (PNMTnegative). Therefore, cfos/pnmt double-staining cells indicate activated A-cells, while cfos-staining cells in the PNMTnegative chromaffin cells indicate activated NA-cells. In the celiac ganglia, DBH was used as a marker of noradrenergic neurons, and cfos/dbh double-staining indicate activated noradrenergic neurons. Quantification of the number of immunolabeled cells was carried out within a square of defined size (200 x 200 µm) placed on each section of adrenal medulla and celiac ganglia. The number of cfos-positive cells, adrenal A-cells, NA-cells and ganglionic noradrenergic neurons were assessed by cell counting in five sections of each area per animal. The percentage of cfos-positive cells in the total A- cells, NA-cells and ganglionic noradrenergic neurons was calculated and expressed as the means ± S.E.M. Data were analyzed by one-way analysis of variance, followed by post-hoc analysis with the Bonferroni method. When only two means were compared, the data were analyzed by ANOVA followed by unpaired Student s t-test. P values less than 0.05 were taken to indicate significance. 10
11 2.5. Drugs The following reagents were used: arginine-vasopressin, corticotropin-releasing factor (rat/human) (Peptide Institute, Osaka, Japan); indomethacin sodium trihydrate (a kind gift from Merck, Rahway, NJ, U.S.A.); furegrelate sodium (Biomol Research Lab., Plymouth Meeting, PA, U.S.A); anti-cfos (Calbiochem, San Diego, CA, U.S.A.); anti-dopamine-β-hydroxylase, antiphenylethanolamine N-methyltransferase (Chemicon, Temecula, CA, U.S.A.); fluorescein isothiocyanate-conjugated donkey antirabbit IgG, fluorescein isothiocyanate-conjugated goat antirabbit IgG, rhodamine-conjugated donkey anti-sheep IgG, rhodamine-conjugated goat anti-mouse IgG (Jackson ImmunoResearch Laboratories, West Grove, PA, U.S.A.); 4,6- diamidine-2 -phenylindole dihydrochloride (Kirkegaard & Perry Laboratories, Gaithersburg, MD, U.S.A.); VECTASHIELD (Vector Laboratories, Burlingame, CA, U.S.A.). All other reagents were the highest grade available (Nacalai Tesque, Kyoto, Japan). 11
12 3. Results 3.1. CRF- and AVP-induced cfos expression in the adrenal medulla and celiac ganglia In the adrenal medulla, A-cells (PNMT-staining cells in the total chromaffin cells) were 74.2±5.9% and NA-cells (PNMTnegative cells in the total chromaffin cells) were 25.8±5.9% (n=44). Administration of CRF (1.5 and 3.0 nmol/animal, i.c.v.) effectively increased cfos expression in the adrenal A-cells and noradrenergic neurons in the celiac ganglia, and these responses obtained with CRF (1.5 nmol/animal, i.c.v.) was greater than those obtained with CRF (3.0 nmol/animal, i.c.v.) (Fig. 1A,B,C,D,E,F,K,M). cfos expression in the adrenal A-cells gradually increased and reached a maximum 60 min after administration of CRF (1.5 and 3.0 nmol/animal) (Fig. 1K,M). In noradrenergic neurons in the celiac ganglia, a large dose of CRF (3.0 nmol/animal, i.c.v.) quickly evoked cfos expression than a small dose of CRF (1.5 nmol/animal, i.c.v.): maximal response was obtained at 60 min for CRF (1.5 nmol/animal, i.c.v.) and at 10 min for CRF (3.0 nmol/animal, i.c.v.), respectively (Fig. 1K,M). On the other hand, the peptide (1.5 and 3.0 nmol/animal, i.c.v.) had no effect on the adrenal NA-cells throughout the experimental period (from 0 min to 120 min) (Fig. 1C,E,K,M). Administration of AVP (0.2 and 0.5 nmol/animal, i.c.v.) effectively increased the levels of cfos expression in the adrenal A-cells and NA-cells, and these responses were almost the same in both doses of AVP. The maximal cfos expressions in 12
13 these regions were obtained at 10 min after AVP administration (0.2 and 0.5 nmol/animal, i.c.v.) (Fig. 1G,I,L,N). On the other hand, the peptide (0.2 and 0.5 nmol/animal, i.c.v.) had no effect on noradrenergic neurons in the celiac ganglia throughout the experimental period (from 0 min to 60 min) (Fig. 1H,J,L,N). In the following experiments, we examined further cfos expression at 60 min after the CRF (1.5 nmol/animal, i.c.v.) administration and 10 min after the AVP (0.2 nmol/animal, i.c.v.) administration, respectively Effects of indomethacin and furegrelate on the CRF-induced cfos expression in the adrenal medulla and celiac ganglia Sixty min after intracerebroventricular treatments with vehicle-1 (10 µl saline) plus vehicle-2 (10 µl saline), indomethacin (an inhibitor of cyclooxygenase) [1.2 µmol (500 µg)/animal] plus vehicle-2, or furegrelate [1.8 µmol (500 µg)/animal] plus vehicle-2, there were little cfos-positive cells in each cell type in the adrenal medulla (A-cells and NAcells) and noradrenergic neurons in the celiac ganglia: percentage of cfos-positive A-cells in total adrenal A-cells [4.2±1.0% for vehicle-1 plus vehicle-2 (n=4), 4.0±0.6% for indomethacin plus vehicle-2 (n=3), 5.5±0.3% for furegrelate plus vehicle-2 (n=3)]; percentage of cfos-positive NA-cells in total adrenal NA-cells [3.9±1.6 for vehicle-1 plus vehicle-2 (n=4), 3.0±1.0% for indomethacin plus vehicle-2 (n=3), 4.5±0.1% for furegrelate plus vehicle-2 (n=3)]; percentage of cfos-positive neurons in total noradrenergic neurons in the celiac ganglia 13
14 [1.1±1.1% for vehicle-1 plus vehicle-2 (n=4), 2.6±0.4% for indomethacin plus vehicle-2 (n=3), 1.8±0.4% for furegrelate plus vehicle-2 (n=3)] (Fig. 2A,B,I). CRF (1.5 nmol/animal, i.c.v.) significantly increased cfos expression in the adrenal A-cells and noradrenergic neurons in the celiac ganglia, but had no effect on the adrenal NA-cells: percentage of cfos-positive cells in total cells in each group [A-cells, 32.4±1.1%; NA-cells, 2.1±2.1%; noradrenergic neurons in the celiac ganglia, 48.5±6.4% (n=4)] (Fig. 2C,D,I). Pretreatment with indomethacin (1.2 µmol/animal, i.c.v.) abolished the CRF (1.5 nmol/animal, i.c.v.)-induced cfos expression in all regions (adrenal A-cells and noradrenergic neurons in the celiac ganglia): percentage of cfos-positive cells in total cells in each group [A-cells, 3.9±1.1%; NA-cells, 3.3±1.7%; noradrenergic neurons in the celiac ganglia, 6.1±4.6 (n=4)] (Fig. 2E,F,I). Pretreatment with furegrelate (1.8 µmol/animal, i.c.v.) abolished the CRF-induced cfos expression in the adrenal A-cells, but had no effect on the noradrenergic neurons in the celiac ganglia: percentage of cfos-positive cells in total cells in each group [A-cells, 5.1±2.2%; NA-cells, 4.8±3.3%; noradrenergic neurons in the celiac ganglia, 41.4±8.6% (n=4)] (Fig. 2G,H,I) Effects of indomethacin and furegrelate on the AVP-induced cfos expression in the adrenal medulla and celiac ganglia 14
15 Ten min after intracerebroventricular treatments with vehicle-1 (10 µl saline) plus vehicle-2 (10 µl saline), indomethacin (an inhibitor of cyclooxygenase) [1.2 µmol (500 µg)/animal] plus vehicle-2, or furegrelate [1.8 µmol (500 µg)/animal] plus vehicle-2, there were little cfos-positive cells in each cell type in the adrenal medulla (A-cells and NAcells) and noradrenergic neurons in the celiac ganglia: percentage of cfos-positive cells in total A-cells [3.5±0.9% for vehicle-1 plus vehicle-2 (n=4), 5.6±1.4% for indomethacin plus vehicle-2 (n=3), 4.1±1.4% for furegrelate plus vehicle-2 (n=3)]; percentage of cfos-positive cells in total NA-cells [5.0±0.7% for vehicle-1 plus vehicle-2 (n=4), 4.7±0.9% for indomethacin plus vehicle-2 (n=3), 4.0±1.2% for furegrelate plus vehicle-2 (n=3)]; percentage of cfos-positive cells in total noradrenergic neurons in the celiac ganglia [2.5±1.0% for vehicle-1 plus vehicle-2 (n=4), 2.5±0.6% for indomethacin plus vehicle-2 (n=3), 3.8±0.8 for furegrelate plus vehicle-2 (n=3)] (Fig. 3A,B,I). AVP (0.2 nmol/animal, i.c.v.) significantly increased cfos expression in the adrenal A-cells and NA-cells, but had no effect on the noradrenergic neurons in the celiac ganglia: percentage of cfos-positive cells in total cells in each group [A-cells, 37.2±1.0%; NA-cells, 40.2±5.8%; noradrenergic neurons in the celiac ganglia, 2.0±1.0% (n=4)] (Fig. 3C, D and I). Indomethacin [1.2 µmol (500 µg)/animal, i.c.v.] abolished the AVP (0.2 nmol/animal)-induced cfos expression in all regions (the adrenal A-cells and NA-cells): percentage of cfos-positive cells in total cells in each group [A-cells, 5.3±0.8%; NA-cells, 15
16 6.2±3.1%; noradrenergic neurons in the celiac ganglia, 3.2±3.2% (n=4)] (Fig. 3E,F,I). Furegrelate [1.8 µmol (500 µg)/animal, i.c.v.] also abolished the AVP-induced cfos expression in all regions such as the adrenal A-cells and NA-cells: percentage of cfos-positive cells in total cells in each group [A-cells, 7.9±3.2%; NA-cells, 3.4±2.7%; noradrenergic neurons in the celiac ganglia, 1.1±1.1% (n=4)] (Fig. 3G,H,I). 16
17 4. Discussion In many mammalian species, adrenomedullary chromaffin cells consist of A-cells and NA-cells. The former cells contain the enzyme PNMT, which converts noradrenaline to adrenaline. Previous studies with immunohistochemical staining for PNMT show that A-cells occupied large areas in the adrenal medulla, and NA-cells were scattered among A-cell regions (Edwards et al., 1996; Suzuki and Kachi, 1996; Phillips et al., 2001). In the present experiment, the number of A-cells is about 3-fold greater than that of NA-cells in the rat adrenal medulla. In the celiac ganglia around the celiac artery, there are many noradrenergic neurons containing the enzyme DBH, which converts dopamine to noradrenaline. In the present experiment, we used cfos expression as an indicator of neuronal and cellular activity. Although the DBH and the PNMT promoter regions do not contain an AP-1 site (Ebert et al., 1994; Morita et al., 1995; Sabban and Kvetnansky, 2001), suggesting that direct effects of cfos on the synthesis of these enzymes are unlikely, tyrosine hydroxylase (a rate-limiting enzyme in catecholamine synthesis) contains an AP-1 site in its promotor region (Chambi et al., 1989; Liu et al., 1997). Many previous studies have shown the colocalization of cfos and PNMT immunoreactivities to prove the neural activity in the adrenergic neuron in the brain (Jung et al., 2004; Lee et al., 2000; Ritter et al., 1998; Temel et al., 2002; Yang and Voogt, 2001). Previously we reported that centrally administered CRF (0.5, 1.5 and 3.0 nmol/animal) and AVP (0.1, 0.2 and 0.5 nmol/animal) dose-dependently elevated plasma levels of noradrenaline and 17
18 adrenaline in anesthetized rats (Yokotani et al., 2001; Okada et al., 2002). In the first experiment, we examined the time course of cfos induction in the adrenal A- and NA-cells and noradrenergic neurons in the celiac ganglia in response to centrally administered CRF (1.5 and 3.0 nmol/animal) or AVP (0.2 and 0.5 nmol/animal). CRF effectively induced cfos expression in the adrenal A-cells and noradrenergic neurons in the celiac ganglia, although CRF even at a higher dose had no effect on the adrenal NA-cells throughout the experimental period (120 min). AVP also effectively induced cfos expression in both A- and NAcells in the adrenal medulla, however AVP even at a higher dose had no effect on noradrenergic neurons in the celiac ganglia throughout the experimental period (60 min). Intravenous administration of these peptides had no effect on cfos expression in any regions. These results suggest that centrally administered CRF activates adrenal A-cells and celiac sympathetic ganglia, while centrally administered AVP activates both the adrenal A-cells and NA-cells in rats. These results are well consistent with our previous paper in which bilateral adrenalectomy abolished the AVP-induced elevations of plasma noradrenaline and adrenaline, while the procedure abolished only the CRF-induced elevation of plasma adrenaline in rats (Okada et al., 2003a). The differences in the time course of cfos expression between CRF- and AVP-treated rats may be, in part, due to the differences in their signal transductions in the brain. We previously reported the involvement of brain inducible nitric oxide synthase in the centrally administered CRF-induced increase of plasma catecholamines (Okada et al., 2003b). Brain nitric oxide has been shown to activate the brain cyclooxygenase, 18
19 thereby elevating plasma catecholamines in rats (Murakami et al., 1998). In the second experiments, we examined the effects of indomethacin, a non-selective inhibitor of cyclooxygenase (Insel, 1996), on the centrally administered CRF- and AVP-induced cfos expression. The central pretreatment with indomethacin abolished the CRF-induced all cfos expression in the adrenal A-cells and noradrenergic neurons in the celiac ganglia and also abolished the AVP-induced all cfos expression in the adrenal A-cells and NA-cells. These results suggest the involvement of the brain arachidonic acid cascade in the CRF- and AVP-induced responses. The present results are well consistent with those of our previous report in which central pretreatment with indomethacin abolished the centrally administered CRF- and AVP-induced elevation of plasma catecholamines in rats (Yokotani et al., 2001; Okada et al., 2003a). Finally, we examined the effects of furegrelate, a selective inhibitor of thromboxane A 2 synthase (Gorman et al., 1983), on the centrally administered CRF- and AVP-induced cfos expression. Central pretreatment with furegrelate abolished the CRF-induced cfos expression in the adrenal A-cells (but not noradrenergic neurons in the celiac ganglia), and also abolished the AVPinduced all cfos expression in the adrenal A-cells and NA-cells. These results indicate the involvement of brain thromboxane A 2 in the centrally administered CRF-mediated activation of adrenal A-cells and AVP-mediated activation of adrenal A- and NA-cells in rats. The present results are well consistent with our previous paper, in which the central pretreatment with 19
20 furegrelate abolished the CRF-induced elevation of adrenaline (but not noradrenaline) and the AVP-induced elevations of both adrenaline and noradrenaline in rats (Yokotani et al., 2001; Okada et al., 2003a). A question has arisen which type of prostanoids is involved in the centrally administered CRF-induced cfos expression in the noradrenergic neurons in the celiac ganglia. In previous studies, we reported that centrally administered prostaglandin E 2 (but not prostaglandin D 2, prostaglandin F 2 or prostaglandin I 2 ) elevated plasma levels of noradrenaline alone by activation of the brain prostanoid EP 3 receptors in rats (Yokotani et al., 1995; Murakami et al., 2002) and this elevation was not influenced by bilateral adrenalectomy (Yokotani et al., 2005). These results suggest the involvement of brain prostaglandin E 2 in the centrally administered CRF-induced cfos expression in the noradrenergic neurons in the celiac ganglia. The hypothalamus, especially the paraventricular nucleus, has been considered to be the control center of the sympathoadrenomedullary outflow (Swanson and Sawchenko, 1980). Retrograde tracer studies suggest a possible connection between the sympatho-adrenomedullary system and the paraventricular nucleus through the splanchnic nerve and spinal cord (Luiten et al., 1987; Jansen et al., 1995; Ranson et al., 1998). Recently we reported that N-methyl-D-aspartate applied into the rat paraventricular nucleus using microdialysis technique produced thromboxane A 2 release in this region and elevation of plasma catecholamines in rats (Okada et al., 2000). Microinjection of thromboxane A 2 mimetic into the rat paraventricular nucleus 20
21 elevated plasma catecholamines (Murakami et al., 2002). Further immunohistochemical studies are required to clarify the brain regions involved in the CRF- or AVP-induced activation of sympatho-adrenomedullary outflow in rats. In summary, we demonstrated here that centrally administered CRF activates adrenal A-cells and noradrenergic neurons in the celiac ganglia by the brain thromboxane A 2 -mediated and the other prostanoid than thromboxane A 2 (probably prostaglandin E 2 )- mediated mechanisms, respectively. On the other hand, centrally administered AVP activates adrenal A-cells and NA-cells by brain thromboxane A 2 -mediated mechanisms in rats. 21
22 Acknowledgements This work was supported in part by a grant from The Smoking Research Foundation in Japan and The Dean Research Fund of the Kochi University. 22
23 References Axelrod, J., Receptor-mediated activation of phospholipase A 2 and arachidonic acid release in signal transduction. Biochem. Soc. Trans. 18, Axelrod, L., Purification and properties of phenylethanolamine-n-methyl transferase. J. Biol. Chem. 237, Brown, M., Tache, Y., Fisher, D., Central nervous system action of bombesin: mechanism to induce hyperglycemia. Endocrinology 105, Brown, M.R., Fisher, L.A., Brain peptide regulation of adrenal epinephrine secretion. Am. J. Physiol. 247, E41-E46. Brown, M.R., Fisher, L.A., Webb, V., Vale, W.W., Rivier, J.E., Corticotropin-releasing factor: a physiologic regulator of adrenal epinephrine secretion. Brain Res. 328, Chambi, F., Fung, B., Chikaraishi, D., flanking DNA sequences direct cell-specific expression of rat tyrosine hydroxylase. J. Neurochem. 53, Coupland, R.E., Pyper, A.S., Hopwood, D., A method for differentiating between noradrenaline- and adrenalinestoring cells in the light and electron microscope. Nature 201,
24 Dronjak, S., Gavrilovic, L., Filipovic, D., Radojcic, M.B., Immobilization and cold stress affect sympathoadrenomedullary system and pituitary-adrenocortical axis of rats exposed to long-term isolation and crowding. Physiol. Behav. 81, Ebert, S.N., Balt, S.L., Hunter, J.P., Gashler, A., Sukhatme, V., Wong, D.L., Egr-1 activation of rat adrenal phenylethanolamine N-methyltransferase gene. J. Biol. Chem. 269, Edwards, S.L., Anderson, C.R., Southwell, B.R., McAllen, R.M Distinct preganglionic neurons innervate noradrenaline and adrenaline cells in the cat adrenal medulla. Neuroscience 70, Feuerstein, G., Zerbe, R.L., Faden, A.I., Central cardiovascular effects of vasotocin, oxytocin and vasopressin in conscious rats. J. Pharmacol. Exp. Ther. 228, Fisher, L.A., Kikkawa, D.O., Rivier, J.E., Amara, S.G., Evans, R.M., Rosenfeld, M.G., Vale, W.W., Brown, M.R., Stimulation of noradrenergic sympathetic outflow by calcitonin gene-related peptide. Nature 305, Flower, R.J., Blackwell, G.J., The importance of phospholipase A 2 in prostaglandin biosynthesis. Biochem. Pharmacol. 25, Folkow, B., von Euler, U.S., Selective activation of noradrenaline and adrenaline producing cells in the cat's 24
25 adrenal gland by hypothalamic stimulation. Cric. Res. 2, Fujino, Y., Fujii, T., Insulin-induced hypoglycemia stimulates both adrenaline and noradrenaline release from adrenal medulla in 21-day-old rats. Jpn. J. Pharmacol. 69, Gorman, R.R., Johnson, R.A., Spilman, C.H., Aiken, J.W., Inhibition of platelet thromboxane A2 synthase activity by sodium 5-(3'-pyridinylmethyl)benzofuran-2-carboxylate. Prostaglandins 26, Hasegawa, T., Yokotani, K., Okuma, Y., Manabe, M., Hirakawa, M., Osumi, Y., Microinjection of α-calcitonin gene-related peptide into the hypothalamus activates sympathetic outflow in rats. Jpn. J. Pharmacol. 61, Herrera, D.G., Robertson, H.A., Activation of c-fos in the brain. Prog. Neurobiol. 50, Insel, P.A., Analgesic-antipyretic and antiinflammatory agents and drugs employed in the treatment of gout. In: Hardman, J.G., Limbird, L.E., Molinoff, P.B., Ruddonm, R.W., Gilman, A.G. (Eds.), Goodman and Gilman s The Pharmacological Basis of Therapeutics, McGraw-Hill, New York, pp Irvine, R.F., How is the level of free arachidonic acid controlled in mammalian cells? Biochem. J. 2004,
26 Jansen, A.S., Nguyen, X.V., Karpitskiy, V., Mettenleiter, T.C., Loewy, A.D., Central command neurons of the sympathetic nervous system: basis of the fight-or-flight response. Science 270, Jezova, D., Ochedalski, T., Glickman, M., Kiss, A., Aguilera, G., Central corticotropin-releasing hormone receptors modulate hypothalamic-pituitary-adrenocortical and sympathoadrenal activity during stress. Neuroscience 94, Jung, J., Lee, J., Kim, W., Enhanced activity of central adrenergic neurons in two-kidney, one clip hypertension in Sprague-Dawley rats. Neurosci. Lett. 369, Kvetnansky, R., Mikulaj, L., Adrenal and urinary catecholamines in rats during adaptation to repeated immobilization stress. Endocrinology 87, Lee, E.J., Moore, C.T., Hosny, S., Centers, A., Jennes, L., Expression of estrogen receptor-α and c-fos in adrenergic neurons of the female rat during the steroid-induced LH surge. Brain Res. 875, Liu, J., Merlie, J.P., Todd, R.D., Identification of cell type-specific promotor elements associated with the rat tyrosine hydroxylase gene using transgenic founder analysis. Mol. Brain Res. 50, Luiten, P.G.M., Ter Horst, G.J., Steffens, A.B., The hypothalamus, intrinsic connections and outflow pathways to 26
27 the endocrine system in relation to the control of feeding and metabolism. Prog. Neurobiol. 28, Morita, K., Ebert, S.N., Wong, D.L., Role of transcription factor Egr-1 in phorbol ester-induced phenylethanolamine N- methyltransferase gene expression. J. Biol. Chem. 270, Murakami, Y., Yokotani, K., Okuma, Y., Osumi, Y., Thromboxane A 2 is involved in the nitric oxide-induced central activation of adrenomedullary outflow in rats. Neuroscience 87, Murakami, Y., Okada, S., Nishihara, M., Yokotani, K., Roles of brain prostaglandin E 2 and thromboxane A 2 in the activation of the central sympatho-adrenomedullary outflow in rats. Eur. J. Pharmacol. 452, Okada, S., Murakami, Y., Nishihara, M., Yokotani, K., Osumi, Y., Perfusion of the hypothalamic paraventricular nucleus with N-methyl-D-aspartate produces thromboxane A 2 and centrally activates adrenomedullary outflow in rats. Neuroscience 96, Okada, S., Murakami, Y., Nakamura, K., Yokotani, K., Vasopressin V 1 receptor-mediated activation of central sympatho-adrenomedullary outflow in rats. Eur. J. Pharmacol. 457, Okada, S., Murakami, Y., Yokotani, K., 2003a. Role of brain thromboxane A 2 in the release of noradrenaline and adrenaline 27
28 from adrenal medulla in rats. Eur. J. Pharmacol. 467, Okada, S., Yokotani, K., Yokotani, K., 2003b. Inducible nitric oxide synthase is involved in corticotropin-releasing hormone-mediated central sympatho-adrenal outflow in rats. Eur. J. Pharmacol. 477, Okuma, Y., Yokotani, K., Osumi, Y., Brain prostaglandins mediate the bombesin-induced increase in plasma levels of catecholamines. Life Sci. 59, Paxinos, G., Watson, C., The Rat Brain in Stereotaxic Coordinates. Academic Press, Boston. Phillips, J.K., Dubey, R., Sesiashvilvi, E., Takeda, M., Christie, D.L., Lipski, J., Differential expression of the noradrenaline transporter in adrenergic chromaffin cells, ganglion cells and nerve fibres of the rat adrenal medulla. J. Chem. Neuroanat. 21, Piezzi, R.S., Bianchi, R.A., Miranda, J.C., 1983, Chromaffin reactions and fluorometric determination of catecholamines in the neonatal adrenal medulla of the rat. J. Histochem. Cytochem. 31, Ranson, R.N., Motawei, K., Pyner, S., Coote, J.H., The paraventricular nucleus of the hypothalamus sends efferents to the spinal cord of the rat that closely appose sympathetic preganglionic neurones projecting to the stellate ganglion. Exp. Brain Res. 120,
29 Ritter, S., Llewellyn-Smith, I., Dinh, T.T., Subgroups of hindbrain catecholamine neurons are selectively activated by 2-deoxy-D-glucose induced metabolic challenge. Brain Res. 805, Sabban, E.L., Kvetnansky, R., Stress-triggered activation of gene expression in catecholaminergic systems: dynamics of transcriptional events. Trends Neurosci. 24, Sagar, S.M., Sharp, F.R., Curran, T., Expression of c-fos protein in brain: metabolic mapping at the cellular level. Science 240, Shima, N., Yamaguchi, Y., Yuri, K., Distribution of estrogen receptor beta mrna-containing cells in ovariectomized and estrogen-treated female rat brain. Ant. Sci. Int. 78, Shimizu, T., Okada, S., Yamaguchi, N., Sasaki, T., Lu, L., Yokotani, K., Centrally administered histamine evokes the adrenal secretion of noradrenaline and adrenaline by brain cyclooxygenase-1- and thromboxane A2-mediated mechanisms in rats. Eur. J. Pharmacol. 541, Suzuki, T., Kachi, T., Similarities and differences in supporting and chromaffin cells in the mammalian adrenal medullae: and immunohistochemical study. Anat. Rec. 244, Swanson, L.W., Sawchenko, P.E., Paraventricular nucleus: a site for the integration of neuroendocrine and autonomic mechanisms. Neuroendocrinology 31,
30 Temel, S., Lin, W., Lakhlani, S., Jennes, L., Expression of estrogen receptor-α and cfos in Norepinephrine and epinephrine neurons of young and middle-aged rats during the steroid-induced luteinizeing hormone surge. Endocrinology 143, Verhofstad, A.A., Coupland, R.E., Parker, T.R., Goldstein, M., Immunohistochemical and biochemical study on the development of the noradrenaline- and adrenaline-storing cells of the adrenal medulla of the rat. Cell Tissue Res. 242, Vollmer, R.R., Selective neural regulation of epinephrine and norepinephrine cells in the adrenal medullacardiovascular implications. Clin. Exp. Hypertens. 18, Vollmer, R.R., Balcita, J.J., Sved, A.F., Edwards, D.J., Adrenal epinephrine and norepinephrine release to hypoglycemia measured by microdialysis in conscious rats. Am. J. Physiol. 273, R1758-R1763. Vollmer, R.R., Balcita-Pedicino, J.J., Debnam, A.J., Edwards, D.J., Adrenal medullary catecholamine secretion patterns in rats evoked by reflex and direct neural stimulation. Clin. Exp. Hypertens. 22, Wurtman, R.J., Stress and the adrenocortical control of epinephrine synthesis. Metabolism 51,
31 Yang, S., Voogt, J.L., Mating-activated brainstem catecholaminergic neurons in the female rat. Brain Res. 894, Yokotani, K., Muramatsu, I., Fujiwara, M., Osumi, Y., Effects of the sympathoadrenal system on vagally induced gastric acid secretion and mucosal blood flow in rats. J. Pharmacol. Exp. Ther. 224, Yokotani, K., Nishihara, M., Murakami, Y., Hasegawa, T., Okuma, Y., Osumi, Y., Elevation of plasma noradrenaline levels in urethane-anaesthetized rats by activation of central prostanoid EP 3 receptors. Br. J. Pharmacol. 115, Yokotani, K., Murakami, Y., Okada, S., Hirata, M., Role of brain arachidonic acid cascade on central CRF 1 receptormediated activation of sympatho-adrenomedullary outflow in rats. Eur. J. Pharmacol. 419, Yokotani, K., Okada, S., Nakamura, K., Yamaguchi-Shima, N., Shimizu, T., Arai, J., Wakiguchi, H., Yokotani, K., Brain prostanoid TP receptor-mediated adrenal noradrenaline secretion and EP 3 receptor-mediated sympathetic noradrenaline release in rats. Eur. J. Pharmacol. 512, Young, J.B., Rosa, R.M., Landsberg, L., Dissociation of sympathetic nervous system and adrenal medullary responses. Am. J. Physiol. 247, E35-E40. 31
32 Legends to figures Fig. 1. CRF- and AVP-induced cfos expression in the adrenal medulla and celiac ganglia. CRF (1.5 and 3.0 nmol/animal, i.c.v.) or AVP (0.2 and 0.5 nmol/animal, i.c.v.) was administered in anesthetized rats. cfos expression in the adrenal medulla (A,C,E,G,I) and celiac ganglia (B,D,F,H,J) was examined at 0 min (A,B) and 60 min (C,D) after CRF (1.5 nmol/animal) administration and at 120 min (E,F) after CRF (3.0 nmol/animal) administration, or 0 min (A,B) and 10 min (G,H) after AVP (0.2 nmol/animal) administration and at 60 min (I,J) after AVP (0.5 nmol/animal) administration. In the adrenal medulla, cfos is shown in the left panel, PNMT is shown in the middle panel and both in the right panel. In the celiac ganglia, cfos is shown in the left panel, DBH is shown in the middle panel and both in the right panel. Scale bar = 100 µm. (K-N) Quantitative analysis of the percentage of cfos-positive cells in all cells in each group (adrenal A-cells and NA-cells and noradrenergic neurons in the celiac ganglia) after intracerebroventricular administration of CRF (1.5 nmol/animal, K; 3.0 nmol/animal, M) or AVP (0.2 nmol/animal, L; 0.5 nmol/animal, N)., adernal A cells;, adrenal NA cells;, celiac ganglia. Results represent the means±s.e.m. (n=4 in each group). #Significantly different (P<0.05) from 0 min before intracerebroventricular administration of CRF or AVP with the Bonferroni method. Fig. 2. Effects of indomethacin and furegrelate on the CRFinduced cfos expression in the adrenal medulla and celiac 32
33 ganglia. Indomethacin (IND) [1.2 µmol (500 µg)/animal], furegrelate [1.8 µmol (500 µg)/animal] (FUR) or vehicle-1 (10 µl saline) was administered 30 min before application of CRF (1.5 nmol/animal, i.c.v.) or vehicle-2 (10 µl saline, i.c.v.). cfos expression in the adrenal medulla (A,C,E,G) and celiac ganglia (B,D,F,H) was examined 60 after CRF (or vehicle-2) administration. (A,B) vehicle-1 plus vehicle-2; (C,D) vehicle-1 plus CRF; (E,F) indomethacin (IND) plus CRF; (G,H) furegrelate (FUR) plus CRF. (I) Quantitative analysis of the percentage of cfos-positive cells in all cells in each group (adrenal A-cells and NA-cells and noradrenergic neurons in the celiac ganglia) after intracerebroventricular administration of CRF (n=4 in each group). Other conditions were the same as those in Fig. 1. *Significantly different (P<0.05) from the group treated with vehicle-1 plus vehicle-2 with the Student s t-test; #significantly different (P<0.05) from the group treated with vehicle-1 plus CRF with the Bonferroni method. Fig. 3. Effects of indomethacin and furegrelate on the AVPinduced cfos expression in the adrenal medulla and celiac ganglia. Indomethacin (IND) [1.2 µmol (500 µg)/animal], furegrelate [1.8 µmol (500 µg)/animal] (FUR) or vehicle-1 (10 µl saline) was i.c.v. administered 30 min before the application of AVP (0.2 nmol/animal) or vehicle-2 (10 µl saline). cfos expression in the adrenal medulla (A,C,E,G) and celiac ganglia (B,D,F,H) was examined 10 min after AVP (or vehicle-2) administration. (A,B) vehicle-1 plus vehicle-2; (C,D) vehicle-1 plus AVP; (E,F) indomethacin (IND) plus AVP; (G,H) furegrelate 33
34 (FUR) plus AVP. (I) Quantitative analysis of the percentage of cfos-positive cells in all cells in each group (adrenal A-cells and NA-cells and noradrenergic neurons in the celiac ganglia) after intracerebroventricular administration of AVP (n=4 in each group). Other conditions were the same as those in Figs. 1 and 2. *Significantly different (P<0.05) from the group treated with vehicle-1 plus vehicle-2 with the Student s t-test; #significantly different (P<0.05) from the group treated with vehicle-1 plus CRF with the Bonferroni method. 34
35 Figure Click here to download high resolution image
36 Figure Click here to download high resolution image
37 Figure Click here to download high resolution image
Brain cyclooxygenase and prostanoid TP receptors are involved in centrally
 Manuscript Click here to view linked References Brain cyclooxygenase and prostanoid TP receptors are involved in centrally administered epibatidine-induced secretion of noradrenaline and adrenaline from
Manuscript Click here to view linked References Brain cyclooxygenase and prostanoid TP receptors are involved in centrally administered epibatidine-induced secretion of noradrenaline and adrenaline from
Centrally administered neuromedin U elevates plasma adrenaline by brain prostanoid
 Revised manuscript Click here to view linked References Centrally administered neuromedin U elevates plasma adrenaline by brain prostanoid TP receptor-mediated mechanisms in rats Tsuyoshi Sasaki a,b, Takahiro
Revised manuscript Click here to view linked References Centrally administered neuromedin U elevates plasma adrenaline by brain prostanoid TP receptor-mediated mechanisms in rats Tsuyoshi Sasaki a,b, Takahiro
Hormones and the Endocrine System Chapter 45. Intercellular communication. Paracrine and Autocrine Signaling. Signaling by local regulators 11/26/2017
 Hormones and the Endocrine System Chapter 45 Intercellular communication Endocrine signaling Local regulators Paracrine and autocrine signaling Neuron signaling Synaptic and neuroendocrine signaling Paracrine
Hormones and the Endocrine System Chapter 45 Intercellular communication Endocrine signaling Local regulators Paracrine and autocrine signaling Neuron signaling Synaptic and neuroendocrine signaling Paracrine
T. Laitinen Departments of Physiology and Clinical Physiology, University of Kuopio and Kuopio University Hospital, Kuopio, Finland
 AUTONOMOUS NEURAL REGULATION T. Laitinen Departments of Physiology and Clinical Physiology, University of Kuopio and Kuopio University Hospital, Kuopio, Finland Keywords: Autonomic nervous system, sympathetic
AUTONOMOUS NEURAL REGULATION T. Laitinen Departments of Physiology and Clinical Physiology, University of Kuopio and Kuopio University Hospital, Kuopio, Finland Keywords: Autonomic nervous system, sympathetic
Hypothalamic Control of Posterior Pituitary
 Hypothalamic Control of Posterior Pituitary Hypothalamus neuron cell bodies produce ADH: supraoptic nuclei Oxytocin: paraventricular nuclei Transported along the hypothalamohypophyseal tract Stored in
Hypothalamic Control of Posterior Pituitary Hypothalamus neuron cell bodies produce ADH: supraoptic nuclei Oxytocin: paraventricular nuclei Transported along the hypothalamohypophyseal tract Stored in
Central catecholamine pathways in stress reactions
 Central catecholamine pathways in stress reactions Palkovits Miklós Semmelweis University, Budapest 2016 Selye János (1907-1982) In 1936, when this definition was formulated, we knew of only three objective
Central catecholamine pathways in stress reactions Palkovits Miklós Semmelweis University, Budapest 2016 Selye János (1907-1982) In 1936, when this definition was formulated, we knew of only three objective
Endocrine secretion cells secrete substances into the extracellular fluid
 Animal Hormones Concept 30.1 Hormones Are Chemical Messengers Endocrine secretion cells secrete substances into the extracellular fluid Exocrine secretion cells secrete substances into a duct or a body
Animal Hormones Concept 30.1 Hormones Are Chemical Messengers Endocrine secretion cells secrete substances into the extracellular fluid Exocrine secretion cells secrete substances into a duct or a body
BIOLOGY - CLUTCH CH.45 - ENDOCRINE SYSTEM.
 !! www.clutchprep.com Chemical signals allow cells to communicate with each other Pheromones chemical signals released to the environment to communicate with other organisms Autocrine signaling self-signaling,
!! www.clutchprep.com Chemical signals allow cells to communicate with each other Pheromones chemical signals released to the environment to communicate with other organisms Autocrine signaling self-signaling,
Chapter 16: Endocrine System 1
 Ch 16 Endocrine System Bi 233 Endocrine system Endocrine System: Overview Body s second great controlling system Influences metabolic activities of cells by means of hormones Slow signaling Endocrine glands
Ch 16 Endocrine System Bi 233 Endocrine system Endocrine System: Overview Body s second great controlling system Influences metabolic activities of cells by means of hormones Slow signaling Endocrine glands
Systems Neuroscience November 21, 2017 The autonomic nervous system
 Systems Neuroscience November 21, 2017 The autonomic nervous system Daniel C. Kiper kiper@ini.phys.ethz.ch http: www.ini.unizh.ch/~kiper/system_neurosci.html How is the organization of the autonomic nervous
Systems Neuroscience November 21, 2017 The autonomic nervous system Daniel C. Kiper kiper@ini.phys.ethz.ch http: www.ini.unizh.ch/~kiper/system_neurosci.html How is the organization of the autonomic nervous
Chp. 17 FUNCTIONAL ORG. Char.of the Endocrine System
 Chp. 17 FUNCTIONAL ORG. Char.of the Endocrine System Glands that secrete chemical signals (hormones) into circulatory system Hormone characteristics Produced in small quantities Secreted into intercellular
Chp. 17 FUNCTIONAL ORG. Char.of the Endocrine System Glands that secrete chemical signals (hormones) into circulatory system Hormone characteristics Produced in small quantities Secreted into intercellular
BIOL 2458 A&P II CHAPTER 18 SI Both the system and the endocrine system affect all body cells.
 BIOL 2458 A&P II CHAPTER 18 SI 1 1. Both the system and the endocrine system affect all body cells. 2. Affect on target cells by the system is slow. Affect on target cells by the system is fast. INTERCELLULAR
BIOL 2458 A&P II CHAPTER 18 SI 1 1. Both the system and the endocrine system affect all body cells. 2. Affect on target cells by the system is slow. Affect on target cells by the system is fast. INTERCELLULAR
Introduction to Autonomic
 Part 2 Autonomic Pharmacology 3 Introduction to Autonomic Pharmacology FUNCTIONS OF THE AUTONOMIC NERVOUS SYSTEM The autonomic nervous system (Figure 3 1) is composed of the sympathetic and parasympathetic
Part 2 Autonomic Pharmacology 3 Introduction to Autonomic Pharmacology FUNCTIONS OF THE AUTONOMIC NERVOUS SYSTEM The autonomic nervous system (Figure 3 1) is composed of the sympathetic and parasympathetic
Chapter 9. The Endocrine System. Lecture Presentation by Patty Bostwick-Taylor Florence-Darlington Technical College Pearson Education, Inc.
 Chapter 9 The Endocrine System Lecture Presentation by Patty Bostwick-Taylor Florence-Darlington Technical College Intro to the Endocrine System Chief Complaint:8-year-old girl with excessive thirst, frequent
Chapter 9 The Endocrine System Lecture Presentation by Patty Bostwick-Taylor Florence-Darlington Technical College Intro to the Endocrine System Chief Complaint:8-year-old girl with excessive thirst, frequent
AUTONOMIC NERVOUS SYSTEM PART I: SPINAL CORD
 AUTONOMIC NERVOUS SYSTEM PART I: SPINAL CORD How is the organization of the autonomic nervous system different from that of the somatic nervous system? Peripheral Nervous System Divisions Somatic Nervous
AUTONOMIC NERVOUS SYSTEM PART I: SPINAL CORD How is the organization of the autonomic nervous system different from that of the somatic nervous system? Peripheral Nervous System Divisions Somatic Nervous
Chapter 20. Endocrine System Chemical signals coordinate body functions Chemical signals coordinate body functions. !
 26.1 Chemical signals coordinate body functions Chapter 20 Endocrine System! Hormones Chemical signals Secreted by endocrine glands Usually carried in the blood Cause specific changes in target cells Secretory
26.1 Chemical signals coordinate body functions Chapter 20 Endocrine System! Hormones Chemical signals Secreted by endocrine glands Usually carried in the blood Cause specific changes in target cells Secretory
Hormones, Receptors and Receptor-Hormone Interactions
 Classification of Hormones Hormones, Receptors and Receptor-Hormone Interactions Synthesis of Protein Hormones and Amine Hormones Hormone Activity Locations of Receptors Mechanisms of Hormone Action Types
Classification of Hormones Hormones, Receptors and Receptor-Hormone Interactions Synthesis of Protein Hormones and Amine Hormones Hormone Activity Locations of Receptors Mechanisms of Hormone Action Types
Chp. 16: AUTONOMIC N.S. (In Review: Peripheral N. S.)
 Chp. 16: AUTONOMIC N.S. (In Review: Peripheral N. S.) Peripheral nerves contain both motor and sensory neurons Among the motor neurons, some of these are somatic and innervate skeletal muscles while some
Chp. 16: AUTONOMIC N.S. (In Review: Peripheral N. S.) Peripheral nerves contain both motor and sensory neurons Among the motor neurons, some of these are somatic and innervate skeletal muscles while some
Downregulation of angiotensin type 1 receptor and nuclear factor-κb. by sirtuin 1 contributes to renoprotection in unilateral ureteral
 Supplementary Information Downregulation of angiotensin type 1 receptor and nuclear factor-κb by sirtuin 1 contributes to renoprotection in unilateral ureteral obstruction Shao-Yu Yang 1,2, Shuei-Liong
Supplementary Information Downregulation of angiotensin type 1 receptor and nuclear factor-κb by sirtuin 1 contributes to renoprotection in unilateral ureteral obstruction Shao-Yu Yang 1,2, Shuei-Liong
4/23/2018. Endocrine System: Overview. Endocrine System: Overview
 Endocrine System: Overview With nervous system, coordinates and integrates activity of body cells Influences metabolic activities via hormones transported in blood Response slower but longer lasting than
Endocrine System: Overview With nervous system, coordinates and integrates activity of body cells Influences metabolic activities via hormones transported in blood Response slower but longer lasting than
significantly suppressed this increase in plasma ACTH.
 Journal of Physiology (1991), 443, pp. 431-439 431 With 3 figures Printed in Great Britain ACTH RELEASE INDUCED IN RATS BY NORADRENALINE IS MEDIATED BY PROSTAGLANDIN E2 BY TATSUO WATANABE, AKIO MORIMOTO,
Journal of Physiology (1991), 443, pp. 431-439 431 With 3 figures Printed in Great Britain ACTH RELEASE INDUCED IN RATS BY NORADRENALINE IS MEDIATED BY PROSTAGLANDIN E2 BY TATSUO WATANABE, AKIO MORIMOTO,
Chapter 11 - Endocrine System
 Chapter 11 - Endocrine System 11.1 Introduction A. The endocrine system is made up of the cells, tissues, and organs that secrete hormones into body fluids. B. The body has two kinds of glands, exocrine
Chapter 11 - Endocrine System 11.1 Introduction A. The endocrine system is made up of the cells, tissues, and organs that secrete hormones into body fluids. B. The body has two kinds of glands, exocrine
Stress and the aging brain
 Stress and the aging brain Stress and the aging brain: What are the issues? Aging makes us less able to adjust to change Reactions of elderly to change generate stress Stress response involves acute reactions
Stress and the aging brain Stress and the aging brain: What are the issues? Aging makes us less able to adjust to change Reactions of elderly to change generate stress Stress response involves acute reactions
Lujain Hamdan. Ayman Musleh & Yahya Salem. Mohammed khatatbeh
 12 Lujain Hamdan Ayman Musleh & Yahya Salem Mohammed khatatbeh the last lecture, we have studied the differences between the two divisions of the ANS: sympathetic and parasympathetic pathways which work
12 Lujain Hamdan Ayman Musleh & Yahya Salem Mohammed khatatbeh the last lecture, we have studied the differences between the two divisions of the ANS: sympathetic and parasympathetic pathways which work
Chapter 20 Endocrine System
 Chapter 20 Endocrine System The endocrine system consists of glands and tissues that secrete Hormones are chemicals that affect other glands or tissues, many times far away from the site of hormone production
Chapter 20 Endocrine System The endocrine system consists of glands and tissues that secrete Hormones are chemicals that affect other glands or tissues, many times far away from the site of hormone production
Ch45: Endocrine System
 Ch45: Endocrine System Endocrine System Homeostasis is the tendency to maintain a stable internal environment. Function = coordinate and control the body with hormones to maintain homeostasis Works with
Ch45: Endocrine System Endocrine System Homeostasis is the tendency to maintain a stable internal environment. Function = coordinate and control the body with hormones to maintain homeostasis Works with
Endocrine System. Chapter 20. Endocrine Glands and Hormones. The Endocrine System. Endocrine glands
 Chapter 20 Endocrine System Endocrine Glands and Hormones The endocrine system consists of glands and tissues that secrete hormones Hormones are chemicals that affect other glands or tissues, many times
Chapter 20 Endocrine System Endocrine Glands and Hormones The endocrine system consists of glands and tissues that secrete hormones Hormones are chemicals that affect other glands or tissues, many times
Art labeling Activity: Figure 16.1
 ANP 1105D Winter 2013 Assignment 6 part I: The Endocrine Sy... Assignment 6 part I: The Endocrine System, Chapter 16 Due: 11:59pm on Monday, March 4, 2013 Note: To understand how points are awarded, read
ANP 1105D Winter 2013 Assignment 6 part I: The Endocrine Sy... Assignment 6 part I: The Endocrine System, Chapter 16 Due: 11:59pm on Monday, March 4, 2013 Note: To understand how points are awarded, read
norepinephrinee." 2 PNMT activity is stimulated by certain adrenocortical markedly,3' 4 but can be restored to normal by the administration of
 IMPAIRED SECRETION OF EPINEPHRINE IN RESPONSE TO INSULIN AMONG HYPOPHYSECTOMIZED DOGS* BY RICHARD J. WURTMAN, ALFRED CASPER, LARISSA A. POHORECKY, AND FREDERIC C. BARTTER DEPARTMENT OF NUTRITION AND FOOD
IMPAIRED SECRETION OF EPINEPHRINE IN RESPONSE TO INSULIN AMONG HYPOPHYSECTOMIZED DOGS* BY RICHARD J. WURTMAN, ALFRED CASPER, LARISSA A. POHORECKY, AND FREDERIC C. BARTTER DEPARTMENT OF NUTRITION AND FOOD
Chapter 26. Hormones and the Endocrine System. Lecture by Edward J. Zalisko
 Chapter 26 Hormones and the Endocrine System PowerPoint Lectures for Biology: Concepts & Connections, Sixth Edition Campbell, Reece, Taylor, Simon, and Dickey Copyright 2009 Pearson Education, Inc. Lecture
Chapter 26 Hormones and the Endocrine System PowerPoint Lectures for Biology: Concepts & Connections, Sixth Edition Campbell, Reece, Taylor, Simon, and Dickey Copyright 2009 Pearson Education, Inc. Lecture
Adrenocorticotropin Responses to Corticotropin Releasing Factor and Vasopressin in Spontaneously Hypertensive Rats
 Laboratory Studies Adrenocorticotropin Responses to Corticotropin Releasing Factor and Vasopressin in Spontaneously Hypertensive Rats TERUHIKO HATTORI, KOZO HASHIMOTO, AND ZENSUKE OTA SUMMARY The effects
Laboratory Studies Adrenocorticotropin Responses to Corticotropin Releasing Factor and Vasopressin in Spontaneously Hypertensive Rats TERUHIKO HATTORI, KOZO HASHIMOTO, AND ZENSUKE OTA SUMMARY The effects
Neuropsychiatry Block
 Neuropsychiatry Block Physiology of the Autonomic Nervous System By Laiche Djouhri, PhD Dept. of Physiology Email: ldjouhri@ksu.edu.sa Ext:71044 References The Autonomic Nervous System and the Adrenal
Neuropsychiatry Block Physiology of the Autonomic Nervous System By Laiche Djouhri, PhD Dept. of Physiology Email: ldjouhri@ksu.edu.sa Ext:71044 References The Autonomic Nervous System and the Adrenal
Chapter 16. APR Enhanced Lecture Slides
 Chapter 16 APR Enhanced Lecture Slides See separate PowerPoint slides for all figures and tables pre-inserted into PowerPoint without notes and animations. Copyright The McGraw-Hill Companies, Inc. Permission
Chapter 16 APR Enhanced Lecture Slides See separate PowerPoint slides for all figures and tables pre-inserted into PowerPoint without notes and animations. Copyright The McGraw-Hill Companies, Inc. Permission
Receptors Functions and Signal Transduction L1- L2
 Receptors Functions and Signal Transduction L1- L2 Faisal I. Mohammed, MD, PhD University of Jordan 1 Introduction to Physiology (0501110) Spring 2013 Subject Receptors: types and adaptation - Membrane
Receptors Functions and Signal Transduction L1- L2 Faisal I. Mohammed, MD, PhD University of Jordan 1 Introduction to Physiology (0501110) Spring 2013 Subject Receptors: types and adaptation - Membrane
Autonomic regulation of islet hormone secretion
 Autonomic regulation of islet hormone secretion Implications for health and disease Billy & Bree Paper 1: Autonomic regulation of islet hormone secretion : Implications for health and disease By Team BBB
Autonomic regulation of islet hormone secretion Implications for health and disease Billy & Bree Paper 1: Autonomic regulation of islet hormone secretion : Implications for health and disease By Team BBB
Chapter 14 The Autonomic Nervous System Chapter Outline
 Chapter 14 The Autonomic Nervous System Chapter Outline Module 14.1 Overview of the Autonomic Nervous System (Figures 14.1 14.3) A. The autonomic nervous system (ANS) is the involuntary arm of the peripheral
Chapter 14 The Autonomic Nervous System Chapter Outline Module 14.1 Overview of the Autonomic Nervous System (Figures 14.1 14.3) A. The autonomic nervous system (ANS) is the involuntary arm of the peripheral
Autonomic Nervous System
 Autonomic Nervous System Keri Muma Bio 6 Organization of the Nervous System Efferent Division Somatic Nervous System Voluntary control Effector = skeletal muscles Muscles must be excited by a motor neuron
Autonomic Nervous System Keri Muma Bio 6 Organization of the Nervous System Efferent Division Somatic Nervous System Voluntary control Effector = skeletal muscles Muscles must be excited by a motor neuron
Biology 2100 Human Physiology C. Iltis SLCC March 8, Midterm Examination #2
 Biology 2100 Human Physiology Name: KEY C. Iltis SLCC March 8, 2000 Midterm Examination #2 Multiple Choice Questions (2 POINTS EACH) 1. When glucose levels are above 100 mg/dl, which of the following is
Biology 2100 Human Physiology Name: KEY C. Iltis SLCC March 8, 2000 Midterm Examination #2 Multiple Choice Questions (2 POINTS EACH) 1. When glucose levels are above 100 mg/dl, which of the following is
Chapter 6 Communication, Integration, and Homeostasis
 Chapter 6 Communication, Integration, and Homeostasis About This Chapter Cell-to-cell communication Signal pathways Novel signal molecules Modulation of signal pathways Homeostatic reflex pathways Cell-to-Cell
Chapter 6 Communication, Integration, and Homeostasis About This Chapter Cell-to-cell communication Signal pathways Novel signal molecules Modulation of signal pathways Homeostatic reflex pathways Cell-to-Cell
Pregnanolone effects on the blood pressure of stress-induced hypertension in rats
 Acta Physiologica Sinica August June 25 25 2004 2004 56 56 (3) (4) 269-274 471-475 http//www.actaps.com.cn 471 * 130021 (pregnanolone ) (stress-induced hypertension SIH) (0.24 mg/kg) (angiotensin Ang )
Acta Physiologica Sinica August June 25 25 2004 2004 56 56 (3) (4) 269-274 471-475 http//www.actaps.com.cn 471 * 130021 (pregnanolone ) (stress-induced hypertension SIH) (0.24 mg/kg) (angiotensin Ang )
Chapter 41. Lecture 14. Animal Hormones. Dr. Chris Faulkes
 Chapter 41 Lecture 14 Animal Hormones Dr. Chris Faulkes Animal Hormones Aims: To appreciate the variety and roles of hormones in the body To understand the basic types of hormones To understand how hormones
Chapter 41 Lecture 14 Animal Hormones Dr. Chris Faulkes Animal Hormones Aims: To appreciate the variety and roles of hormones in the body To understand the basic types of hormones To understand how hormones
Chapter 17. Lecture and Animation Outline
 Chapter 17 Lecture and Animation Outline To run the animations you must be in Slideshow View. Use the buttons on the animation to play, pause, and turn audio/text on or off. Please Note: Once you have
Chapter 17 Lecture and Animation Outline To run the animations you must be in Slideshow View. Use the buttons on the animation to play, pause, and turn audio/text on or off. Please Note: Once you have
Human Anatomy and Physiology - Problem Drill 16: The Endocrine System
 Human Anatomy and Physiology - Problem Drill 16: The Endocrine System Question No. 1 of 10 The endocrine system is made up of a number of organs and glands. Which one of the following is not an organ or
Human Anatomy and Physiology - Problem Drill 16: The Endocrine System Question No. 1 of 10 The endocrine system is made up of a number of organs and glands. Which one of the following is not an organ or
Hormones. Prof. Dr. Volker Haucke Institut für Chemie-Biochemie Takustrasse 6
 Hormones Prof. Dr. Volker Haucke Institut für Chemie-Biochemie Takustrasse 6 Tel. 030-8385-6920 (Sekret.) 030-8385-6922 (direkt) e-mail: vhaucke@chemie.fu-berlin.de http://userpage.chemie.fu-berlin.de/biochemie/aghaucke/teaching.html
Hormones Prof. Dr. Volker Haucke Institut für Chemie-Biochemie Takustrasse 6 Tel. 030-8385-6920 (Sekret.) 030-8385-6922 (direkt) e-mail: vhaucke@chemie.fu-berlin.de http://userpage.chemie.fu-berlin.de/biochemie/aghaucke/teaching.html
The endocrine system -- a brief overview.
 The endocrine system -- a brief overview. I. Introduction - the endocrine system is an integration system that influences the metabolic activities of cells. - acts via hormones, chemical messengers produced
The endocrine system -- a brief overview. I. Introduction - the endocrine system is an integration system that influences the metabolic activities of cells. - acts via hormones, chemical messengers produced
Drugs Affecting The Autonomic Nervous System(ANS)
 Drugs Affecting The Autonomic Nervous System(ANS) ANS Pharmacology Lecture 1 Dr. Hiwa K. Saaed College of Pharmacy, University of Sulaimani 2018-2019 AUTOMATIC NERVOUS SYSTEM (ANS) The ANS is the major
Drugs Affecting The Autonomic Nervous System(ANS) ANS Pharmacology Lecture 1 Dr. Hiwa K. Saaed College of Pharmacy, University of Sulaimani 2018-2019 AUTOMATIC NERVOUS SYSTEM (ANS) The ANS is the major
Cholecystokinin antagonist, proglumide, stimulates growth hormone release in the rat
 J. Biosci., Vol. 15, Number 1, March 1990, pp. 17 21. Printed in India. Cholecystokinin antagonist, proglumide, stimulates growth hormone release in the rat E. VIJAYAN* and S. M. McCANN Department of Physiology,
J. Biosci., Vol. 15, Number 1, March 1990, pp. 17 21. Printed in India. Cholecystokinin antagonist, proglumide, stimulates growth hormone release in the rat E. VIJAYAN* and S. M. McCANN Department of Physiology,
Possible inhibitory roles of endogenous 2-arachidonoylglycerol during corticotropinreleasing
 *Manuscript Click here to view linked References Possible inhibitory roles of endogenous 2-arachidonoylglycerol during corticotropinreleasing factor-induced activation of central sympatho-adrenomedullary
*Manuscript Click here to view linked References Possible inhibitory roles of endogenous 2-arachidonoylglycerol during corticotropinreleasing factor-induced activation of central sympatho-adrenomedullary
Chemical Regulation. Chapter 26. Testosterone and Male Aggression: Is There a Link? THE NATURE OF CHEMICAL REGULATION
 Chapter 6 Chemical Regulation PowerPoint Lectures for Biology: Concepts and Connections, Fifth Edition Campbell, Reece, Taylor, and Simon Testosterone and Male Aggression: Is There a Link? Among male animals,
Chapter 6 Chemical Regulation PowerPoint Lectures for Biology: Concepts and Connections, Fifth Edition Campbell, Reece, Taylor, and Simon Testosterone and Male Aggression: Is There a Link? Among male animals,
NOTES: ENDOCRINE SYSTEM (CH 9)
 NOTES: ENDOCRINE SYSTEM (CH 9) Endocrine System *The endocrine system consists of a range of glands and tissues throughout the body Functions of the Endocrine System: 1) Maintain balance within body (homeostasis)
NOTES: ENDOCRINE SYSTEM (CH 9) Endocrine System *The endocrine system consists of a range of glands and tissues throughout the body Functions of the Endocrine System: 1) Maintain balance within body (homeostasis)
Biology 218 Human Anatomy
 Chapter 20 Adapted form Tortora 10 th ed. LECTURE OUTLINE A. Introduction (p. 632) 1. The autonomic nervous system (ANS) regulates the activity of smooth muscle, cardiac muscle, and certain glands. 2.
Chapter 20 Adapted form Tortora 10 th ed. LECTURE OUTLINE A. Introduction (p. 632) 1. The autonomic nervous system (ANS) regulates the activity of smooth muscle, cardiac muscle, and certain glands. 2.
Analysis of AVP functions via V1a and V1b receptors with knockout mice. Akito Tanoue
 Analysis of AVP functions via V1a and V1b receptors with knockout mice Akito Tanoue Department of Pharmacology, National Research Institute for Child Health and Development Arginine-Vasopressin (AVP) is
Analysis of AVP functions via V1a and V1b receptors with knockout mice Akito Tanoue Department of Pharmacology, National Research Institute for Child Health and Development Arginine-Vasopressin (AVP) is
Endocrine System Notes
 Endocrine System Notes is the tendency to maintain a stable internal environment. - parts of the body that secrete hormones directly into the body. - parts of the body that make secretions which travel
Endocrine System Notes is the tendency to maintain a stable internal environment. - parts of the body that secrete hormones directly into the body. - parts of the body that make secretions which travel
Testosterone and other male hormones seem to be related to aggressive behavior in some species
 Testosterone and Male Aggression Testosterone and other male hormones seem to be related to aggressive behavior in some species In the fish species Oreochromis mossambicus, elevated levels have been found
Testosterone and Male Aggression Testosterone and other male hormones seem to be related to aggressive behavior in some species In the fish species Oreochromis mossambicus, elevated levels have been found
I. Neural Control of Involuntary Effectors. Chapter 9. Autonomic Motor Nerves. Autonomic Neurons. Autonomic Ganglia. Autonomic Neurons 9/19/11
 Chapter 9 I. Neural Control of Involuntary Effectors The Autonomic Nervous System Lecture PowerPoint Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction or display. Autonomic
Chapter 9 I. Neural Control of Involuntary Effectors The Autonomic Nervous System Lecture PowerPoint Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction or display. Autonomic
ADRENAL GLAND. Introduction 4/21/2009. Among most important and vital endocrine organ. Small bilateral yellowish retroperitoneal organ
 Introduction Among most important and vital endocrine organ ADRENAL GLAND D.Hammoudi.MD Small bilateral yellowish retroperitoneal organ Lies just above kidney in gerota s fascia 2 1 The Adrenal Gland Anatomy
Introduction Among most important and vital endocrine organ ADRENAL GLAND D.Hammoudi.MD Small bilateral yellowish retroperitoneal organ Lies just above kidney in gerota s fascia 2 1 The Adrenal Gland Anatomy
GLUCOSE TRANSPORTERS IN THE BLOOD-BRAIN BARRIER
 Papers on Anthropology XXI, 2012, pp. 50 55 GLUCOSE TRANSPORTERS IN THE BLOOD-BRAIN BARRIER PIRET HUSSAR 1, INDREK BENNO 2, ÜLO HUSSAR 1 1 Institute of Anatomy, Faculty of Medicine, University of Tartu,
Papers on Anthropology XXI, 2012, pp. 50 55 GLUCOSE TRANSPORTERS IN THE BLOOD-BRAIN BARRIER PIRET HUSSAR 1, INDREK BENNO 2, ÜLO HUSSAR 1 1 Institute of Anatomy, Faculty of Medicine, University of Tartu,
Laith Abu Shekha. Omar Sami. Ebaa Alzayadneh
 24 Laith Abu Shekha Omar Sami Ebaa Alzayadneh Signal Transduction Please note that it s very important to refer to the slides. Introduction: Through these five lectures, we should know the basics of signal
24 Laith Abu Shekha Omar Sami Ebaa Alzayadneh Signal Transduction Please note that it s very important to refer to the slides. Introduction: Through these five lectures, we should know the basics of signal
Chapter 11. Endocrine System
 Chapter 11 Endocrine System 1 Introduction A. The endocrine system is made up of the cells, tissues, and organs that secrete hormones into body fluids. B. Hormones diffuse into the bloodstream to act target
Chapter 11 Endocrine System 1 Introduction A. The endocrine system is made up of the cells, tissues, and organs that secrete hormones into body fluids. B. Hormones diffuse into the bloodstream to act target
NROSCI/BIOSC 1070 and MSNBIO 2070 September 11, 2017 Control Mechanisms 2: Endocrine Control
 NROSCI/BIOSC 1070 and MSNBIO 2070 September 11, 2017 Control Mechanisms 2: Endocrine Control Hormones are chemical messengers that are secreted into the blood by endocrine cells or specialized neurons.
NROSCI/BIOSC 1070 and MSNBIO 2070 September 11, 2017 Control Mechanisms 2: Endocrine Control Hormones are chemical messengers that are secreted into the blood by endocrine cells or specialized neurons.
Autonomic nervous system
 Autonomic nervous system Key notes Autonomic: an independent system that runs on its own The ANS is a visceral and involuntary sensory and motor system The visceral motor fibers in the autonomic nerves
Autonomic nervous system Key notes Autonomic: an independent system that runs on its own The ANS is a visceral and involuntary sensory and motor system The visceral motor fibers in the autonomic nerves
Nature Neuroscience: doi: /nn Supplementary Figure 1. Splenic atrophy and leucopenia caused by T3 SCI.
 Supplementary Figure 1 Splenic atrophy and leucopenia caused by T3 SCI. (a) Gross anatomy of representative spleens from control and T3 SCI mice at 28 days post-injury. (b and c) Hematoxylin and eosin
Supplementary Figure 1 Splenic atrophy and leucopenia caused by T3 SCI. (a) Gross anatomy of representative spleens from control and T3 SCI mice at 28 days post-injury. (b and c) Hematoxylin and eosin
Autonomic Nervous System and Hypothalamus
 Lu Chen LSA room 201 Phone: (510) 643-8163 Email: luchen@berkeley.edu Office hours: M,W,F, 10-11 am 1 Autonomic Nervous System and Hypothalamus Lu Chen, Ph.D. MCB, UC Berkeley 2 Hypothalamus Brain stem
Lu Chen LSA room 201 Phone: (510) 643-8163 Email: luchen@berkeley.edu Office hours: M,W,F, 10-11 am 1 Autonomic Nervous System and Hypothalamus Lu Chen, Ph.D. MCB, UC Berkeley 2 Hypothalamus Brain stem
Chapter 13 worksheet
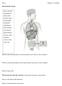 Name: Chapter 13 worksheet The Endocrine System Please label the: hypothalamus pineal gland pituitary gland thyroid gland parathyroid gland thymus heart stomach liver adrenal glands kidneys pancreas small
Name: Chapter 13 worksheet The Endocrine System Please label the: hypothalamus pineal gland pituitary gland thyroid gland parathyroid gland thymus heart stomach liver adrenal glands kidneys pancreas small
Endocrine System Hormones. AP Biology
 Endocrine System Hormones 2007-2008 Regulation Why are hormones needed? u chemical messages from one body part to another u communication needed to coordinate whole body u daily homeostasis & regulation
Endocrine System Hormones 2007-2008 Regulation Why are hormones needed? u chemical messages from one body part to another u communication needed to coordinate whole body u daily homeostasis & regulation
The Autonomic Nervous System Outline of class lecture for Physiology
 The Autonomic Nervous System Outline of class lecture for Physiology 1 After studying the endocrine system you should be able to: 1. Describe the organization of the nervous system. 2. Compare and contrast
The Autonomic Nervous System Outline of class lecture for Physiology 1 After studying the endocrine system you should be able to: 1. Describe the organization of the nervous system. 2. Compare and contrast
Adrenal Medulla. Amelyn R. Rafael, M.D.
 Adrenal Medulla Amelyn R. Rafael, M.D. Adrenal Medulla Exodermal in origin Cells derived from the sympathogonia of the primitive neuroectoderm A sympathetic ganglion in which the post-ganglionic cells
Adrenal Medulla Amelyn R. Rafael, M.D. Adrenal Medulla Exodermal in origin Cells derived from the sympathogonia of the primitive neuroectoderm A sympathetic ganglion in which the post-ganglionic cells
Homeostasis Through Chemistry. The Endocrine System Topic 6.6
 Homeostasis Through Chemistry The Endocrine System Topic 6.6 Comparing NS & ES Animals have two systems of internal communication and regulation The nervous system Response time: Fast, quick Signals: electrical
Homeostasis Through Chemistry The Endocrine System Topic 6.6 Comparing NS & ES Animals have two systems of internal communication and regulation The nervous system Response time: Fast, quick Signals: electrical
PHRM20001: Pharmacology - How Drugs Work!
 PHRM20001: Pharmacology - How Drugs Work Drug: a chemical that affects physiological function in a specific way. Endogenous substances: hormones, neurotransmitters, antibodies, genes. Exogenous substances:
PHRM20001: Pharmacology - How Drugs Work Drug: a chemical that affects physiological function in a specific way. Endogenous substances: hormones, neurotransmitters, antibodies, genes. Exogenous substances:
Tyrosine hydroxylase, the rate-limiting enzyme in catecholamine biosynthesis could be an index of functionality in pheochromocytoma diagnosis
 Tyrosine hydroxylase, the rate-limiting enzyme in catecholamine biosynthesis could be an index of functionality in pheochromocytoma diagnosis Ana-Maria Stefanescu 1, *, Sorina Schipor 1, and Corin Badiu
Tyrosine hydroxylase, the rate-limiting enzyme in catecholamine biosynthesis could be an index of functionality in pheochromocytoma diagnosis Ana-Maria Stefanescu 1, *, Sorina Schipor 1, and Corin Badiu
Endocrine system. Coordination & regulation Glands Hormones
 Endocrine system Coordination & regulation Glands Hormones Endocrine system structures Anatomy - Dispersed system of glands that communicate with each other & all body cells via hormones. Endocrine glands:
Endocrine system Coordination & regulation Glands Hormones Endocrine system structures Anatomy - Dispersed system of glands that communicate with each other & all body cells via hormones. Endocrine glands:
Integrated Cardiopulmonary Pharmacology Third Edition
 Integrated Cardiopulmonary Pharmacology Third Edition Chapter 3 Pharmacology of the Autonomic Nervous System Multimedia Directory Slide 19 Slide 37 Slide 38 Slide 39 Slide 40 Slide 41 Slide 42 Slide 43
Integrated Cardiopulmonary Pharmacology Third Edition Chapter 3 Pharmacology of the Autonomic Nervous System Multimedia Directory Slide 19 Slide 37 Slide 38 Slide 39 Slide 40 Slide 41 Slide 42 Slide 43
RELEASE OF MEDULLARY AMINES FROM THE ISOLATED PERFUSED ADRENAL GLAND OF THE DOG
 Brit. J. Pharmacol. (1965), 24, 561-565. RELEASE OF MEDULLARY AMINES FROM THE ISOLATED PERFUSED ADRENAL GLAND OF THE DOG BY MARTHE VOGT From the Agricultural Research Council Institute of Animal Physiology,
Brit. J. Pharmacol. (1965), 24, 561-565. RELEASE OF MEDULLARY AMINES FROM THE ISOLATED PERFUSED ADRENAL GLAND OF THE DOG BY MARTHE VOGT From the Agricultural Research Council Institute of Animal Physiology,
Endocrine System. Chemical Control
 Endocrine System Chemical Control Endocrine System - the system that secretes hormones in the body - hormones can last for minutes or for hours - a major gland, once called the master gland, is the pituitary
Endocrine System Chemical Control Endocrine System - the system that secretes hormones in the body - hormones can last for minutes or for hours - a major gland, once called the master gland, is the pituitary
GENERAL CHARACTERISTICS OF THE ENDOCRINE SYSTEM FIGURE 17.1
 GENERAL CHARACTERISTICS OF THE ENDOCRINE SYSTEM FIGURE 17.1 1. The endocrine system consists of glands that secrete chemical signals, called hormones, into the blood. In addition, other organs and cells
GENERAL CHARACTERISTICS OF THE ENDOCRINE SYSTEM FIGURE 17.1 1. The endocrine system consists of glands that secrete chemical signals, called hormones, into the blood. In addition, other organs and cells
Endocrine System Hormones (Ch. 45)
 Endocrine System Hormones (Ch. 45) Regulation Why are hormones needed? chemical messages from one body part to another communication needed to coordinate whole body daily homeostasis & regulation of large
Endocrine System Hormones (Ch. 45) Regulation Why are hormones needed? chemical messages from one body part to another communication needed to coordinate whole body daily homeostasis & regulation of large
Composed by Natalia Leonidovna Svintsitskaya, Associate professor of the Chair of Human Anatomy, Candidate of Medicine
 Theoretical background to the study of the autonomic nervous system. Sympathetic and parasympathetic divisions of the autonomic nervous system. Features of the structure, function Composed by Natalia Leonidovna
Theoretical background to the study of the autonomic nervous system. Sympathetic and parasympathetic divisions of the autonomic nervous system. Features of the structure, function Composed by Natalia Leonidovna
Central vs. Peripheral Nervous System
 Nervous System 2 C 1 2 : A N A L Y Z E T H E F U N C T I O N A L I N T E R R E L A T I O N S H I P S O F T H E D I V I S I O N S O F T H E N E R V O U S S Y S T E M Central vs. Peripheral Nervous System
Nervous System 2 C 1 2 : A N A L Y Z E T H E F U N C T I O N A L I N T E R R E L A T I O N S H I P S O F T H E D I V I S I O N S O F T H E N E R V O U S S Y S T E M Central vs. Peripheral Nervous System
CHAPTER 15 LECTURE OUTLINE
 CHAPTER 15 LECTURE OUTLINE I. INTRODUCTION A. The autonomic nervous system (ANS) regulates the activity of smooth muscle, cardiac muscle, and certain glands. B. Operation of the ANS to maintain homeostasis,
CHAPTER 15 LECTURE OUTLINE I. INTRODUCTION A. The autonomic nervous system (ANS) regulates the activity of smooth muscle, cardiac muscle, and certain glands. B. Operation of the ANS to maintain homeostasis,
Central Progesterone Involvement in Estrogen- Induced Prolactin and Luteinizing Hormone Secretion Surges in Female Rats
 Southern Illinois University Carbondale OpenSIUC Honors Theses University Honors Program 5-10-2014 Central Progesterone Involvement in Estrogen- Induced Prolactin and Luteinizing Hormone Secretion Surges
Southern Illinois University Carbondale OpenSIUC Honors Theses University Honors Program 5-10-2014 Central Progesterone Involvement in Estrogen- Induced Prolactin and Luteinizing Hormone Secretion Surges
8/26/13. Announcements
 Announcements THM questions will start for points on Wednesday. Make sure you are registered correctly! Problems registering for BioPortal? Make sure you are using the link from the syllabus or FAQ. 30
Announcements THM questions will start for points on Wednesday. Make sure you are registered correctly! Problems registering for BioPortal? Make sure you are using the link from the syllabus or FAQ. 30
Medical Endocrinology / Introduction 4 Medical Endocrinology
 Medical Endocrinology / Introduction 4 Medical Endocrinology 1 2 : Positive feedback control of labor contractions during birth of a baby. The solid return arrow symbolizes positive feedback. If the response
Medical Endocrinology / Introduction 4 Medical Endocrinology 1 2 : Positive feedback control of labor contractions during birth of a baby. The solid return arrow symbolizes positive feedback. If the response
Hormonal regulation of. Physiology Department Medical School, University of Sumatera Utara
 Hormonal regulation of nutrient metabolism Physiology Department Medical School, University of Sumatera Utara Homeostasis & Controls Successful compensation Homeostasis reestablished Failure to compensate
Hormonal regulation of nutrient metabolism Physiology Department Medical School, University of Sumatera Utara Homeostasis & Controls Successful compensation Homeostasis reestablished Failure to compensate
Autocrine. Control of Lactation. Lactation. Illinois Module :
 Autocrine Control of Lactation Illinois Module : Lactation Autocrine Control of Lactation Control of lactation is clearly regulated by hormones, However local factors are also important Evidences Cows
Autocrine Control of Lactation Illinois Module : Lactation Autocrine Control of Lactation Control of lactation is clearly regulated by hormones, However local factors are also important Evidences Cows
Ch 8: Endocrine Physiology
 Ch 8: Endocrine Physiology Objectives 1. Review endocrine glands of body. 2. Understand how hypothalamus controls endocrine system & sympathetic epinephrine response. 3. Learn anterior pituitary hormones
Ch 8: Endocrine Physiology Objectives 1. Review endocrine glands of body. 2. Understand how hypothalamus controls endocrine system & sympathetic epinephrine response. 3. Learn anterior pituitary hormones
Ch45: Endocrine System
 Ch45: Endocrine System Endocrine System Homeostasis is the tendency to maintain a stable internal environment. Function = with hormones to maintain homeostasis Works with nervous system Anatomy Location:
Ch45: Endocrine System Endocrine System Homeostasis is the tendency to maintain a stable internal environment. Function = with hormones to maintain homeostasis Works with nervous system Anatomy Location:
HORMONES (Biomedical Importance)
 hormones HORMONES (Biomedical Importance) Hormones are the chemical messengers of the body. They are defined as organic substances secreted into blood stream to control the metabolic and biological activities.
hormones HORMONES (Biomedical Importance) Hormones are the chemical messengers of the body. They are defined as organic substances secreted into blood stream to control the metabolic and biological activities.
ENDOCRINE SYSTEM ENDOCRINE SYSTEM
 Endocrine system consists of organs that produce and secrete hormones "endocrine" = internal secretion into capillaries Hormones carried by the blood to another organ; exert effects Hormones manipulate
Endocrine system consists of organs that produce and secrete hormones "endocrine" = internal secretion into capillaries Hormones carried by the blood to another organ; exert effects Hormones manipulate
Enhanced Norepinephrine Release in Hypothalamus from Locus Coeruleus in SHR
 Enhanced Norepinephrine Release in Hypothalamus from Locus Coeruleus in SHR Shingo KAWASAKI, M.D., Kazuo TAKEDA, M.D., Motoo TANAKA, M.D., Hiroshi ITOH, M.D., Masahiro HIRATA, M.D., Tetsuo NAKATA, M.D.,
Enhanced Norepinephrine Release in Hypothalamus from Locus Coeruleus in SHR Shingo KAWASAKI, M.D., Kazuo TAKEDA, M.D., Motoo TANAKA, M.D., Hiroshi ITOH, M.D., Masahiro HIRATA, M.D., Tetsuo NAKATA, M.D.,
The Endocrine System. By Carl Antiado, Marco Habana, Cameron Loewen, and Nikki Ooka
 The Endocrine System By Carl Antiado, Marco Habana, Cameron Loewen, and Nikki Ooka Standards 2.C.2 : Organisms respond to changes in their external environments. 4.A.4: Organisms exhibit complex properties
The Endocrine System By Carl Antiado, Marco Habana, Cameron Loewen, and Nikki Ooka Standards 2.C.2 : Organisms respond to changes in their external environments. 4.A.4: Organisms exhibit complex properties
INSULIN AND THE SUPRARENAL GLAND OF THE RABBIT
 Brit. J. Phawmacol. (1951), 6, 289. INSULIN AND THE SUPRARENAL GLAND OF THE RABBIT BY From the Pharmacological Laboratory, University of St. Andrews, Medical School, Dundee (Received February 2, 1951)
Brit. J. Phawmacol. (1951), 6, 289. INSULIN AND THE SUPRARENAL GLAND OF THE RABBIT BY From the Pharmacological Laboratory, University of St. Andrews, Medical School, Dundee (Received February 2, 1951)
The Adrenal Glands. Bởi: OpenStaxCollege
 The Adrenal Glands Bởi: OpenStaxCollege The adrenal glands are wedges of glandular and neuroendocrine tissue adhering to the top of the kidneys by a fibrous capsule ([link]). The adrenal glands have a
The Adrenal Glands Bởi: OpenStaxCollege The adrenal glands are wedges of glandular and neuroendocrine tissue adhering to the top of the kidneys by a fibrous capsule ([link]). The adrenal glands have a
ENDOCRINE SYSTEM. Endocrine
 ENDOCRINE SYSTEM Endocrine Function Help regulate internal functions Use chemical messengers Recall: Endocrine vs. Exocrine glands Nervous System vs Endocrine System Target Specificity Lock n Key action
ENDOCRINE SYSTEM Endocrine Function Help regulate internal functions Use chemical messengers Recall: Endocrine vs. Exocrine glands Nervous System vs Endocrine System Target Specificity Lock n Key action
Close to site of release (at synapse); binds to receptors in
 Chapter 18: The Endocrine System Chemical Messengers 1. Neural 2. Endocrine 3. Neuroendocrine 4. Paracrine 5. Autocrine Endocrine System --Endocrine and nervous systems work together --Endocrine vs. Nervous
Chapter 18: The Endocrine System Chemical Messengers 1. Neural 2. Endocrine 3. Neuroendocrine 4. Paracrine 5. Autocrine Endocrine System --Endocrine and nervous systems work together --Endocrine vs. Nervous
Endocrine system. Coordination & regulation Glands Hormones
 Endocrine system Coordination & regulation Glands Hormones Endocrine system structures Anatomy - Dispersed system of glands that communicate with each other & all body cells via hormones. Endocrine glands:
Endocrine system Coordination & regulation Glands Hormones Endocrine system structures Anatomy - Dispersed system of glands that communicate with each other & all body cells via hormones. Endocrine glands:
Endocrine Glands: Hormone-secreting organs are called endocrine glands
 University of Jordan Department of Physiology and Biochemistry Nursing students, Academic year 2017/2018. ******************************************************************* Ref: Principles of Anatomy
University of Jordan Department of Physiology and Biochemistry Nursing students, Academic year 2017/2018. ******************************************************************* Ref: Principles of Anatomy
Autonomic Nervous System Dr. Ali Ebneshahidi
 Autonomic Nervous System Dr. Ali Ebneshahidi Nervous System Divisions of the nervous system The human nervous system consists of the central nervous System (CNS) and the Peripheral Nervous System (PNS).
Autonomic Nervous System Dr. Ali Ebneshahidi Nervous System Divisions of the nervous system The human nervous system consists of the central nervous System (CNS) and the Peripheral Nervous System (PNS).
9.3 Stress Response and Blood Sugar
 9.3 Stress Response and Blood Sugar Regulate Stress Response Regulate Blood Sugar Stress Response Involves hormone pathways that regulate metabolism, heart, rate and breathing The Adrenal Glands a pair
9.3 Stress Response and Blood Sugar Regulate Stress Response Regulate Blood Sugar Stress Response Involves hormone pathways that regulate metabolism, heart, rate and breathing The Adrenal Glands a pair
Autonomic Nervous System
 ANS..??? Autonomic Nervous System Nervous system CNS PNS Autonomic Somatic Symp Parasymp Enteric SOMATIC AUTONOMIC Organ supplied Skeletal muscle Other organs Distal most synapse Nerve fibre Peripheral
ANS..??? Autonomic Nervous System Nervous system CNS PNS Autonomic Somatic Symp Parasymp Enteric SOMATIC AUTONOMIC Organ supplied Skeletal muscle Other organs Distal most synapse Nerve fibre Peripheral
Cells and viruses. Human isolates (A/Kawasaki/173/01 [H1N1], A/Yokohama/2057/03 [H3N2],
![Cells and viruses. Human isolates (A/Kawasaki/173/01 [H1N1], A/Yokohama/2057/03 [H3N2], Cells and viruses. Human isolates (A/Kawasaki/173/01 [H1N1], A/Yokohama/2057/03 [H3N2],](/thumbs/86/93801604.jpg) Supplementary information Methods Cells and viruses. Human isolates (A/Kawasaki/173/01 [H1N1], A/Yokohama/2057/03 [H3N2], and A/Hong Kong/213/03 [H5N1]) were grown in Madin-Darby canine kidney (MDCK) cells
Supplementary information Methods Cells and viruses. Human isolates (A/Kawasaki/173/01 [H1N1], A/Yokohama/2057/03 [H3N2], and A/Hong Kong/213/03 [H5N1]) were grown in Madin-Darby canine kidney (MDCK) cells
