Keeping the Blood Supply Safe Current and Future Strategies. Marissa Li, MD
|
|
|
- Michael Powers
- 5 years ago
- Views:
Transcription
1 Keeping the Blood Supply Safe Current and Future Strategies Marissa Li, MD
2 Overview Pre-collection Donor Screening Collection Visual Inspection Aseptic Processing Leukoreduction of Apheresis Collections Post-Collection Infectious Disease Testing Bacteria Virus Parasite Blood Culture Emerging Infectious Agents Pathogen Reduction Technologies
3 Donor Screening Questions The first 3 questions on the UDHQ Feeling healthy and well today? Currently taking an antibiotic? Currently taking any other medication for an infection? High risk behavior assessment questions Sexual contacts with prostitutes, persons with HIV or Hepatitis, history of syphilis or gonorrhea, incarceration >72 hrs, etc. vcjd (prion) risk assessment questions Time spent in United Kingdom, Europe or receiving blood transfusions in United Kingdom or France Untested infectious disease questions Malaria, Babesia
4 Value of Donor Questions Protect the health of the donor Lower the risk of collecting an infectious unit Deferral based on risk factors for transmissible infections prevents the collection of contaminated units Risk factor screening compliments laboratory testing by avoiding collections in the window period Safe guard against transmissible agents that are not currently tested (e.g. vcjd) Donor selection may have surrogate value, i.e. deferral of malaria exposure in an endemic area might prevent transfusion risk from an emerging disease
5 Pitfalls of Donor Questions Low sensitivity and specificity May result in significant loss of healthy donors Undermine public confidence in the blood system Trusting that donors are truthful
6 Risk Reduction During Collection Visual inspection Looking for track-marks, or open wounds Aseptic processing Iodine or chlorhexidine gluconate prep 30 seconds of vigorous scrubbing of the venipuncture site Diversion pouch Leukoreduction of apheresis collections
7 Infectious Disease Tests (IDT) Bacteria Syphilis Virus Hepatitis B & C virus HIV 1/2/O HTLV 1/2 WNV CMV Parasite T. cruzi
8 Syphilis Screening test Anti-T. pallidum: microhemagglutination test (MHA-TP) Confirmation test Anti-T. pallidum: EIA Supplemental test RPR
9 Hepatitis B Virus Serologic test Window period: days Risk of TTI: 1:280,000 to 1:357,000 units HBs antigen (1970s) Screening test: ChLIA Confirmatory test: neutralization ChLIA Anti-HBc (1986): ChLIA Nucleic acid test (NAT) Introduced in 2006 Reduced window period to days Risk of TTI: 1:830,000 to 1:2 million units
10 Hepatitis C Virus Serologic test: anti-hcv (1990) Window period: 50 days Screening test Anti-HCV: EIA Third generation EIA screening assay with increased specificity in donor population Confirmation test Anti-HCV: Recombinant Immunoblot Assay (RIBA 3.0) Helps to resolve false positive EIA result NAT introduced in 2000 reduced the risk of transfusion transmitted Hep C to approximately 1:2 million units Shortens window period to ~ days
11 HIV Serologic test Window period: 14 days Screening test Anti-HIV 1/2/O (1985): EIA Confirmation test Anti-HIV 1: IFA Supplemental test Anti-HIV 2: EIA Referral test Anti-HIV 2: Western blot NAT Introduced in 2003 for HIV-1 Reduced the risk of transfusion transmitted HIV to approximately 1:2 million units Shortens window period to 7 days
12 HTLV-I/II Serologic test Risk of TTI: 1:3 million units Screening test Anti-HTLV-I/II (1988): ChLIA Supplemental test Anti-HTLV I/II: Inno-LIA Leukocyte reduction may reduce risk of transmission No confirmed reports of transmission through acellular components (FFP, cryo) or plasma derivatives
13 WNV NAT (2003) Screening test Mini pool of 16 samples year round Individual testing triggered by positive mini pool 1 st positive: individual testing for 7 days 2nd positive: individual testing for 14 days Individual testing during outbreak season: 6/1/11 to 10/31/11 WNV Panel Antibody (IgM/IgG) ELISA Confirmatory test
14 CMV Serologic test Risk of TTI: unknown, presumed rare Anti-CMV Leukocyte reduction decreases the risk of transmission (essentially equivalent to seronegative units) No confirmed reports of transmission through acellular components (FFP, cryo) or plasma derivatives
15 Trypanosoma cruzi Serologic test Risk of TTI: Unknown Screening test Anti-T. cruzi (2007): EIA Referral test Anti-T. cruzi: RIPA One time testing
16 Value of IDT Improve safety of blood supply Individual donor benefit Detecting unsuspected infection Prevent secondary spread Recipient benefit Provide safest blood possible Public health benefit Provide epidemiological data to identify the sources of risk and the safest donors Monitor epidemics, e.g. WNV outbreak
17 Pitfalls of IDT False positive results False negative results Window period
18 Bacterial Detection Platelets Every single unit is cultured (starting in 2004) Bacterial contamination rate: 1:5,000 units Septic reaction rate: between 1:41,000 and 1:193,000 units Fatality rate: 1:500,000 units Most common bacteria Staph epidermidis, Bacillus spp., Staph aureus or Strep spp. Kept at room temperature, optimal for bacterial growth
19 Bacterial Contaminations RBCs Bacterial contamination rate: 1:31,000 units Septic reaction rate: 1:250,000 to 1:500,000 units Fatality rate: between 1:1 million to 1:10 million units Most common bacteria Yersinia enterocolitica, Serratia or Pseudomonas spp. FFP/Cryo
20 Emerging Infectious Agents Dengue Fever Two clusters of TTI has been reported in Hong Kong and Singapore Chikungunya virus (CHIKV) Simian foamy virus (SFV)
21 Pathogen Reduction Technologies (PRT) Exposure of whole blood or blood components to a chemicals, often in combination with UV light which leads to irreversible DNA strand breakage and/or strand linkage This technology has the added benefit of inactivating leukocytes, thus reducing FNHTR, alloimmunization and TAGVHD Plasma pathogen reduction has been in use in many countries other than the US Platelet pathogen reduction has been implemented in some countries Whole blood or RBC pathogen reduction are not available
22 Clinically Approved PRT Solvent-Detergent Treatment Psoralen Phototreatment Methylene Blue Phototreatment Riboflavin (Vitamin B2) Phototreatment
23 Solvent-Detergent Treatment Solvent detergent (SD) system disrupts the lipid bilayers of the plasma membrane of cells/organisms and the lipid coat of enveloped viruses Only approved for non-cellular blood products, e.g. plasma Available in Europe Octoplas Complex process using pooled plasma ( units) Contains variety of antibodies against viruses Reduce the incidence of TRALI through dilution of HLA antibodies
24 Solvent-Detergent Treatment SD plasma has demonstrated significant reduction of lipid-enveloped viruses (>6 log 10 ), bacteria and protozoa Less effective against non-enveloped viruses Hepatitis A virus, Parvovirus B19 Residual solvent (TNBP) and detergent (Triton-X100) are mostly undetectable SD plasma has been shown to reduce pro- and anticoagulation factors Factor V, VIII, antiplasmin and protein S Routine coagulation times are not significantly prolonged
25 Psoralen Phototreatment Amotosalen Psoralen compound found in nature that is able to intercalate between nucleotide base pairs of RNA and DNA Requires photoactivation by UVA at nm INTERCEPT Currently available system Combines amotosalen with a compound absorption device containing activated charcoal to scavenge unincorporated amotosalen and residual photoproducts
26 Psoralen Phototreatment Effective in reducing lipid-enveloped virus (3-6 log 10 ), bacteria and protozoa Less effective against non-enveloped viruses Long term toxicity due to residual psoralen is unknown Reduction in coagulation factors Reduction in platelet count recovery No evidence of antibody formation against psoralen or neoantigens development
27 Methylene Blue Phototreatment Methylene blue Heterocyclic thiazine dye with long history of medical use Primary mechanism of action through photoactivated ( nm) cleavage of nucleic acid It s not incorporated into the nucleic acid product After phototreatment, methylene blue is passed through a specialized filter that adsorbs it Available in Europe, marketed by Macopharma Suitable for treatment of single donor plasma units
28 Methylene Blue Phototreatment Effective in reducing enveloped viruses (>5 log 10 ), some non-enveloped viruses Some effects against parasites (T. cruzi) Unreliable reduction on bacteria or leukocytes Methylene blue do not penetrate bacterial or eukaryotic plasma membrane well Reduction of coagulation factors Long term toxicology is unknown
29 Riboflavin Riboflavin (Vitamin B2) Phototreatment Shares a similar structure and mechanism with methylene blue as a photoreactive compound Photoreactivity can be induced by visible or UV light Mirasol PRT The only commercial system approved for the production of riboflavin-treated plasma and platelets
30 Riboflavin (Vitamin B2) Phototreatment Effective against enveloped viruses (4-6 log 10 reduction), nonenveloped viruses (3-5 log 10 reduction) and bacteria (3-5 log 10 reduction) Advantages Physiological normal metabolic constituent Photoproducts are products of its natural metabolism, no need for posttreatment removal Long history and experience in clinical use Disadvantages Riboflavin-treated platelets stored for 5 days showed greater changes in levels of coagulation, thrombolytic factors and inhibitors without functional deficits
31 Advantage of Pathogen Reduction Proactive approach to improving blood safety Pathogen reduction can kill various kinds of microbs, e.g. virus, bacteria, parasites Prevent spread of unknown blood born pathogens Potential for increasing platelet storage
32 Limitations of Pathogen Reduction Pathogen reduction technology does not lead to complete eradication of infectious agents Bacterial spores are resistant to pathogen reduction methods No technology is available to inactivate prions Not available for prbc or whole blood
33 Limitations of Pathogen Reduction Direct toxicity of the chemical presently used is negligible Indirect effect on blood products have been observed Neoantigen development on RBCs that may induce antibody formation Reduced platelet recovery and survival
34 Conclusion Maintaining a safe and plentiful blood supply is crucial and a tough balancing act Pre collection Collection Post collection Most promising step for reducing infectious disease transmission, especially with the improvements in pathogen reduction technologies
Pathogen Reduction/Inactivation KABB Annual Meeting Elpidio Pena, MD, MA Norton Healthcare Transfusion Services
 Pathogen Reduction/Inactivation KABB Annual Meeting 2017 Elpidio Pena, MD, MA Norton Healthcare Transfusion Services Goals Discuss the approved methods for pathogen reduction/inactivation of blood products
Pathogen Reduction/Inactivation KABB Annual Meeting 2017 Elpidio Pena, MD, MA Norton Healthcare Transfusion Services Goals Discuss the approved methods for pathogen reduction/inactivation of blood products
New Advances in Transfusion EM I LY CO BERLY, M D
 New Advances in Transfusion EM I LY CO BERLY, M D TRANSFUSI ON M EDI CI NE FELLO W VANDERBI LT UNI VERSITY Objectives To discuss the terminology, components, transfusion risks, and dosing guidelines for
New Advances in Transfusion EM I LY CO BERLY, M D TRANSFUSI ON M EDI CI NE FELLO W VANDERBI LT UNI VERSITY Objectives To discuss the terminology, components, transfusion risks, and dosing guidelines for
Screening donors and donations for transfusion transmissible infectious agents. Alan Kitchen
 Screening donors and donations for transfusion transmissible infectious agents Alan Kitchen Aim Not to teach you microbiology To provide and awareness of the big picture To provide an understanding of
Screening donors and donations for transfusion transmissible infectious agents Alan Kitchen Aim Not to teach you microbiology To provide and awareness of the big picture To provide an understanding of
Keeping blood transfusions safe:
 ISP-WIV Seminar Diagnosis and surveillance of infectious diseases May 18 th, 2017 Keeping blood transfusions safe: the challenge of emerging infectious diseases Presented by Roland HÜBNER Superior Health
ISP-WIV Seminar Diagnosis and surveillance of infectious diseases May 18 th, 2017 Keeping blood transfusions safe: the challenge of emerging infectious diseases Presented by Roland HÜBNER Superior Health
Pathogen inactivation in platelet concentrates in France
 Swisstransfusion: Jahrestagung 2011 Fribourg, 8-9 septembre, 2011 Pathogen inactivation in platelet concentrates in France Jean-Pierre Cazenave EFS-Alsace, UMR_S949 INSERM-Université de Strasbourg, France
Swisstransfusion: Jahrestagung 2011 Fribourg, 8-9 septembre, 2011 Pathogen inactivation in platelet concentrates in France Jean-Pierre Cazenave EFS-Alsace, UMR_S949 INSERM-Université de Strasbourg, France
Non-reproductive tissues and cells
 Colour key Minimum requirements as set out in Directive 2004/23/EC and its technical Directives (particularly 2006/17/EC) More stringent -legally binding, applies for all donations and all donor profiles
Colour key Minimum requirements as set out in Directive 2004/23/EC and its technical Directives (particularly 2006/17/EC) More stringent -legally binding, applies for all donations and all donor profiles
Reporting from Council of Europe member states on the collection, testing and use of blood and blood components in Europe The 2006 Survey
 Reporting from Council of Europe member states on the collection, testing and use of blood and blood components in Europe The 2006 Survey This questionnaire consists of three sections: A. Collection and
Reporting from Council of Europe member states on the collection, testing and use of blood and blood components in Europe The 2006 Survey This questionnaire consists of three sections: A. Collection and
Mary Berg, M.D. Medical Director, Transfusion Services Associate Professor of Pathology University of Colorado Hospital
 Transfusion Reactions/Complications Mary Berg, M.D. Medical Director, Transfusion Services Associate Professor of Pathology University of Colorado Hospital Acute Transfusion Reactions Can be seen with
Transfusion Reactions/Complications Mary Berg, M.D. Medical Director, Transfusion Services Associate Professor of Pathology University of Colorado Hospital Acute Transfusion Reactions Can be seen with
Confirmatory Testing Algorithm. Routine Donor Testing Algorithm
 Confirmatory Testing Algorithm Blood Confirmatory ABO/Rh Beckman Coulter PK-ABO/Rh Immucor NEO ABO/Rh Immucor NEO ABO/Rh Immucor Manual ABO/Rh Typing Immucor Manual ABO/Rh Typing Discrepancy Resolution
Confirmatory Testing Algorithm Blood Confirmatory ABO/Rh Beckman Coulter PK-ABO/Rh Immucor NEO ABO/Rh Immucor NEO ABO/Rh Immucor Manual ABO/Rh Typing Immucor Manual ABO/Rh Typing Discrepancy Resolution
Non-reproductive tissues and cells Recommending authority/ association
 Colour key Minimum requirements as set out in Directive 2004/23/EC More stringent - legy binding More stringent - recommended Not legy binding and not recommended Tested pathogen Donor test/ technique
Colour key Minimum requirements as set out in Directive 2004/23/EC More stringent - legy binding More stringent - recommended Not legy binding and not recommended Tested pathogen Donor test/ technique
EDUCATIONAL COMMENTARY EMERGING INFECTIOUS DISEASE AGENTS
 EDUCATIONAL COMMENTARY EMERGING INFECTIOUS DISEASE AGENTS Educational commentary is provided through our affiliation with the American Society for Clinical Pathology (ASCP). To obtain FREE CME/CMLE credits
EDUCATIONAL COMMENTARY EMERGING INFECTIOUS DISEASE AGENTS Educational commentary is provided through our affiliation with the American Society for Clinical Pathology (ASCP). To obtain FREE CME/CMLE credits
Non-reproductive tissues and cells
 Colour key Minimum requirements as set out in Directive 2004/23/EC More stringent testing - legally binding on national level More stringent testing - recommended on national level Not legally binding
Colour key Minimum requirements as set out in Directive 2004/23/EC More stringent testing - legally binding on national level More stringent testing - recommended on national level Not legally binding
Surveillance Report 2014
 Surveillance Report 2014 Executive summary We are pleased to present the third report for our stakeholders describing infectious disease surveillance. High quality and timely surveillance is key to the
Surveillance Report 2014 Executive summary We are pleased to present the third report for our stakeholders describing infectious disease surveillance. High quality and timely surveillance is key to the
The testing of Donated Blood and Components at NHSBT
 The testing of Donated Blood and Components at NHSBT NHSBT is responsible for collecting all donated blood and platelets in England, then processing into components before issuing to client hospitals.
The testing of Donated Blood and Components at NHSBT NHSBT is responsible for collecting all donated blood and platelets in England, then processing into components before issuing to client hospitals.
Non-reproductive tissues and cells
 Colour key Minimum requirements as set out in Directive 2004/23/EC More stringent - legy binding on national level More stringent - recommended on national level Not legy binding and not recommended on
Colour key Minimum requirements as set out in Directive 2004/23/EC More stringent - legy binding on national level More stringent - recommended on national level Not legy binding and not recommended on
Pathogenreduktion mittels UVC-Licht: Entwicklung des THERAFLEX UV-Plättchen-Systems
 Pathogenreduktion mittels UVC-Licht: Entwicklung des THERAFLEX UV-Plättchen-Systems Pathogen reduction by UVC light: Development of the THERAFLEX UV-Platelets system Fribourg 2011 Prof. Dr. Axel Seltsam,
Pathogenreduktion mittels UVC-Licht: Entwicklung des THERAFLEX UV-Plättchen-Systems Pathogen reduction by UVC light: Development of the THERAFLEX UV-Platelets system Fribourg 2011 Prof. Dr. Axel Seltsam,
Babesia from a donor perspective
 Babesia from a donor perspective American Red Cross, Massachusetts Region Bryan Spencer, MPH Research Scientist American Society for Apheresis Annual Meeting May 8, 2015 San Antonio, TX The need is constant.
Babesia from a donor perspective American Red Cross, Massachusetts Region Bryan Spencer, MPH Research Scientist American Society for Apheresis Annual Meeting May 8, 2015 San Antonio, TX The need is constant.
Update on Transfusion- Transmitted Infectious Diseases
 Update on Transfusion- Transmitted Infectious Diseases Scott Koepsell MD PhD Associate Professor University of Nebraska Medical Center I have no conflicts of interest to disclose. Global changes and the
Update on Transfusion- Transmitted Infectious Diseases Scott Koepsell MD PhD Associate Professor University of Nebraska Medical Center I have no conflicts of interest to disclose. Global changes and the
Optimal / Evidence-based Method to Prevent Transmission of Cytomegalovirus (CMV) by Transfusion
 ILABB Fall Meeting 2015 Optimal / Evidence-based Method to Prevent Transmission of Cytomegalovirus (CMV) by Transfusion Presenter Ronald G Strauss, MD Associate Medical Director, LifeSource/ITxM Terms
ILABB Fall Meeting 2015 Optimal / Evidence-based Method to Prevent Transmission of Cytomegalovirus (CMV) by Transfusion Presenter Ronald G Strauss, MD Associate Medical Director, LifeSource/ITxM Terms
Surveillance Report 2015
 Surveillance Report 2015 Executive summary We are pleased to present the fourth report for our stakeholders describing infectious disease surveillance. High quality and timely surveillance is key to the
Surveillance Report 2015 Executive summary We are pleased to present the fourth report for our stakeholders describing infectious disease surveillance. High quality and timely surveillance is key to the
Transfusion transmitted infections in National Haemovigilance Systems: the Greek experience
 Transfusion transmitted infections in National Haemovigilance Systems: the Greek experience Global blood product safety 10 April 2019, Rome, Italy Constantina Politis National Coordinating Haemovigilance
Transfusion transmitted infections in National Haemovigilance Systems: the Greek experience Global blood product safety 10 April 2019, Rome, Italy Constantina Politis National Coordinating Haemovigilance
FDA Reentry Guidance
 FDA Reentry Guidance T. cruzi - Chagas, Hepatitis C and HIV Wednesday 6/6/18 Doug Denyer O Dina Hurlburt, SBB(ASCP)CM Outline Impact to Clients Review of FDA Guidance Documents Reentry Request Process
FDA Reentry Guidance T. cruzi - Chagas, Hepatitis C and HIV Wednesday 6/6/18 Doug Denyer O Dina Hurlburt, SBB(ASCP)CM Outline Impact to Clients Review of FDA Guidance Documents Reentry Request Process
TRANSFUSION ASSOCIATED DISEASE, RECALL, OR COMPLICATION INVESTIGATION POLICY I. FATALITIES AND COMPLICATIONS ASSOCIATED WITH TRANSFUSION:
 I. FATALITIES AND COMPLICATIONS ASSOCIATED WITH TRANSFUSION: A. TRANSFUSION RELATED FATALITY: FDA and MEDIC must be notified immediately, and subsequently in writing, when a possible transfusion related
I. FATALITIES AND COMPLICATIONS ASSOCIATED WITH TRANSFUSION: A. TRANSFUSION RELATED FATALITY: FDA and MEDIC must be notified immediately, and subsequently in writing, when a possible transfusion related
Be ready. The INTERCEPT Blood System for Platelets and Plasma pathogen reduction system
 Be ready. The INTERCEPT Blood System for Platelets and Plasma pathogen reduction system INTERCEPT Blood System for Platelets an Blood safety is changing Safe donated blood is critical to improving certain
Be ready. The INTERCEPT Blood System for Platelets and Plasma pathogen reduction system INTERCEPT Blood System for Platelets an Blood safety is changing Safe donated blood is critical to improving certain
Impact of donor epidemiology and screening strategies on the safety of blood and plasma for fractionation
 IPFA 2 nd Asia Workshop in Yogyakarta Impact of donor epidemiology and screening strategies on the safety of blood and plasma for fractionation Yoshihiko Tani, MD, PhD Japanese Red Cross Osaka Blood Center
IPFA 2 nd Asia Workshop in Yogyakarta Impact of donor epidemiology and screening strategies on the safety of blood and plasma for fractionation Yoshihiko Tani, MD, PhD Japanese Red Cross Osaka Blood Center
Non-reproductive tissues and cells Recommending authority/ association
 Colour key Minimum requirements as set out in Directive 2004/23/EC More stringent - legally binding More stringent - recommended Not legally binding and not recommended Non-reproductive tissues and cells
Colour key Minimum requirements as set out in Directive 2004/23/EC More stringent - legally binding More stringent - recommended Not legally binding and not recommended Non-reproductive tissues and cells
Surveillance Report 2016
 Surveillance Report 2016 Executive summary We are pleased to present the fifth report for our stakeholders describing infectious disease surveillance. High quality and timely surveillance is key to the
Surveillance Report 2016 Executive summary We are pleased to present the fifth report for our stakeholders describing infectious disease surveillance. High quality and timely surveillance is key to the
Non-reproductive tissues and cells
 Colour key Minimum requirements as set out in Directive 2004/23/EC More stringent testing - legally binding on national level More stringent testing - recommended on national level Not legally binding
Colour key Minimum requirements as set out in Directive 2004/23/EC More stringent testing - legally binding on national level More stringent testing - recommended on national level Not legally binding
The Technical Aspects of Pathogen Testing in Canada. Nancy Angus Canadian Blood Services Director, Testing
 PERMISSON TO USE: Please note that, by making their presentations available on-line, primary authors have agreed to share their presentations. However, should you want to use some of the data or slides
PERMISSON TO USE: Please note that, by making their presentations available on-line, primary authors have agreed to share their presentations. However, should you want to use some of the data or slides
Update on THERAFLEX UV-Platelets: a novel system for pathogen reduction of platelets
 Update on THERAFLEX UV-Platelets: a novel system for pathogen reduction of platelets Axel Seltsam, MD, MHBA German Red Cross Blood Service NSTOB, Springe Fribourg 2011 THERAFLEX UV-Platelets Efficient
Update on THERAFLEX UV-Platelets: a novel system for pathogen reduction of platelets Axel Seltsam, MD, MHBA German Red Cross Blood Service NSTOB, Springe Fribourg 2011 THERAFLEX UV-Platelets Efficient
Non-reproductive tissues and cells
 Colour key Tested pathogen VIRAL Minimum requirements as set out in Directive 2004/23/EC More stringent testing - legy binding on national level More stringent testing - recommended on national level Not
Colour key Tested pathogen VIRAL Minimum requirements as set out in Directive 2004/23/EC More stringent testing - legy binding on national level More stringent testing - recommended on national level Not
Pathogen Safety and BSE / variant CJD
 Pathogen Safety and BSE / variant CJD Thomas R. Kreil, Global Pathogen Safety Parenteral Drug Industry Plasma Protein Industry Summit September 7, 2017; Beijing Pathogen Safety Those who cannot remember
Pathogen Safety and BSE / variant CJD Thomas R. Kreil, Global Pathogen Safety Parenteral Drug Industry Plasma Protein Industry Summit September 7, 2017; Beijing Pathogen Safety Those who cannot remember
Are Our Screening Algorithms Appropriate? Prospects for the Next 10 Years
 Are Our Screening Algorithms Appropriate? Prospects for the Next 10 Years IPFA/PEI 18 th International Workshop on Surveillance and Screening of Blood Borne Pathogens 24-25 May 2011, Dublin, Ireland Jay
Are Our Screening Algorithms Appropriate? Prospects for the Next 10 Years IPFA/PEI 18 th International Workshop on Surveillance and Screening of Blood Borne Pathogens 24-25 May 2011, Dublin, Ireland Jay
Prevention of transfusion-transmitted arboviruses in French Polynesia
 Prevention of transfusion-transmitted arboviruses in French Polynesia D Musso 1, V Richard 1, J Green 2,J Broult 3, M Aubry 1 1. Institut Louis Malardé, Tahiti, French Polynesia 2. Cerus Corporation, California,
Prevention of transfusion-transmitted arboviruses in French Polynesia D Musso 1, V Richard 1, J Green 2,J Broult 3, M Aubry 1 1. Institut Louis Malardé, Tahiti, French Polynesia 2. Cerus Corporation, California,
Chapter 17: Transfusion-Transmitted Infections (TTI)
 Annual SHOT Report 2014 Supplementary Information Chapter 17: Transfusion-Transmitted s (TTI) The table below is an excerpt from the full Table 17.2 which can be viewed in the main report. Case reports
Annual SHOT Report 2014 Supplementary Information Chapter 17: Transfusion-Transmitted s (TTI) The table below is an excerpt from the full Table 17.2 which can be viewed in the main report. Case reports
Non-reproductive tissues and cells
 Ministry of Health: Institute for Transplantation and Biomedicine / Colour key VIRAL HIV 1 and HIV 2 Hepatitis B Minimum requirements as set out in Directive 2004/23/EC More stringent - legy binding More
Ministry of Health: Institute for Transplantation and Biomedicine / Colour key VIRAL HIV 1 and HIV 2 Hepatitis B Minimum requirements as set out in Directive 2004/23/EC More stringent - legy binding More
Blood Supply and Wastage
 Blood Supply and Wastage Inga Willett Lab Matters, Oake Manor, 21 st June 2017 Blood supply UK supplied by 4 blood services: SNBTS NIBTS NHSBT WBS http://commons.wikimedia.org/wiki/file:uk_map_home_nations.png
Blood Supply and Wastage Inga Willett Lab Matters, Oake Manor, 21 st June 2017 Blood supply UK supplied by 4 blood services: SNBTS NIBTS NHSBT WBS http://commons.wikimedia.org/wiki/file:uk_map_home_nations.png
Blood Screening Assays. CTS lab locations where assays are performed are indicated by alpha codes D, P, and T.
 Blood Screening Assays CTS lab locations where assays are performed are indicated by alpha codes D, P, and T. D= performed in Dallas lab FEI: 3002580251 P= performed in Phoenix lab FEI: 3000204058 T= performed
Blood Screening Assays CTS lab locations where assays are performed are indicated by alpha codes D, P, and T. D= performed in Dallas lab FEI: 3002580251 P= performed in Phoenix lab FEI: 3000204058 T= performed
Annual SHOT Report 2016 Supplementary Information. Chapter 17: Transfusion-Transmitted Infections (TTI)
 Annual SHOT Report 2016 Supplementary Information Chapter 17: Transfusion-Transmitted s (TTI) The table below is an excerpt from the full Table 17.3 which can be viewed in the main report. Case reports
Annual SHOT Report 2016 Supplementary Information Chapter 17: Transfusion-Transmitted s (TTI) The table below is an excerpt from the full Table 17.3 which can be viewed in the main report. Case reports
Anticipating (re)emerging infections to ensure blood safety
 Anticipating (re)emerging infections to ensure blood safety Pierre Tiberghien Etablissement Français du Sang, St-Denis and Université de Franche-Comté, Besançon, France IPFA/PEI 23rd International Workshop
Anticipating (re)emerging infections to ensure blood safety Pierre Tiberghien Etablissement Français du Sang, St-Denis and Université de Franche-Comté, Besançon, France IPFA/PEI 23rd International Workshop
Non-reproductive tissues and cells Recommending authority/ association
 Colour key Minimum requirements as set out in Directive 2004/23/EC More stringent testing - legy binding on national level More stringent testing - recomd on national level Not legy binding and not recomd
Colour key Minimum requirements as set out in Directive 2004/23/EC More stringent testing - legy binding on national level More stringent testing - recomd on national level Not legy binding and not recomd
Babesia and Blood Safety
 Babesia and Blood Safety American Red Cross, New England Region Bryan Spencer, MPH Manager of Blood Research / REDS-III Coordinator ASM 46 th Annual Region I Meeting 27 October, 2011 Randolph, MA The need
Babesia and Blood Safety American Red Cross, New England Region Bryan Spencer, MPH Manager of Blood Research / REDS-III Coordinator ASM 46 th Annual Region I Meeting 27 October, 2011 Randolph, MA The need
Adverse Reactions Associated with Methylene Blue Inactivated Fresh Frozen Plasma (MB FFP)
 Adverse Reactions Associated with Methylene Blue Inactivated Fresh Frozen Plasma (MB FFP) Constantina Politis Hellenic Coordinating Haemovigilance Centre (SKAE) Background Pathogen inactivation is expected
Adverse Reactions Associated with Methylene Blue Inactivated Fresh Frozen Plasma (MB FFP) Constantina Politis Hellenic Coordinating Haemovigilance Centre (SKAE) Background Pathogen inactivation is expected
A. Anti-HIV-1/2, HIV NAT Lookback: for those identified blood and blood components collected:
 PURPOSE: MATERIALS: To outline a procedure for removing, recalling and/or correcting blood products that do not meet the requirements of the FDA, AABB, or the blood center. Recall/Market Withdrawal Form
PURPOSE: MATERIALS: To outline a procedure for removing, recalling and/or correcting blood products that do not meet the requirements of the FDA, AABB, or the blood center. Recall/Market Withdrawal Form
Blood Transfusion. What is blood transfusion? What are blood banks? When is a blood transfusion needed? Who can donate blood?
 What is blood transfusion? A blood transfusion is a safe, common procedure in which blood is given through an intravenous (IV) line in one of the blood vessels. A blood transfusion usually takes two to
What is blood transfusion? A blood transfusion is a safe, common procedure in which blood is given through an intravenous (IV) line in one of the blood vessels. A blood transfusion usually takes two to
PATHOGEN INACTIVATION:
 PATHOGEN INACTIVATION: She s worth it. Be SURE. With INTERCEPT you know that you are doing everything in your power to deliver safe and effective blood products to patients. Transfusion-transmitted infection
PATHOGEN INACTIVATION: She s worth it. Be SURE. With INTERCEPT you know that you are doing everything in your power to deliver safe and effective blood products to patients. Transfusion-transmitted infection
Non-reproductive tissues and cells Recommending authority/ association
 Colour key Minimum requirements as set out in Directive 2004/23/EC More stringent - legy binding More stringent - recommended Not legy binding and not recommended Non-reproductive tissues and cells VIRAL
Colour key Minimum requirements as set out in Directive 2004/23/EC More stringent - legy binding More stringent - recommended Not legy binding and not recommended Non-reproductive tissues and cells VIRAL
Increased Risk Donors. November 1, 2018 BC Kidney Days. Jagbir Gill MD MPH Associate Professor of Medicine UBC
 Increased Risk Donors November 1, 2018 BC Kidney Days Jagbir Gill MD MPH Associate Professor of Medicine UBC Median dialysis exposure prior to DDKT Case scenarios Which donors should we accept kidneys
Increased Risk Donors November 1, 2018 BC Kidney Days Jagbir Gill MD MPH Associate Professor of Medicine UBC Median dialysis exposure prior to DDKT Case scenarios Which donors should we accept kidneys
Robust Inactivation of Yellow Fever Virus 17D Vaccine Strain can be achieved by Photochemical Treatment of Platelet Concentrates
 Robust Inactivation of Yellow Fever Virus 17D Vaccine Strain can be achieved by Photochemical Treatment of Platelet Concentrates Andrew Laughhunn 1, Jean-Marc Payrat 2, Felicia Santa Maria 1, Yvette Girard
Robust Inactivation of Yellow Fever Virus 17D Vaccine Strain can be achieved by Photochemical Treatment of Platelet Concentrates Andrew Laughhunn 1, Jean-Marc Payrat 2, Felicia Santa Maria 1, Yvette Girard
HCV: FROM HIPPOCRATES TO CURE:
 HCV: FROM HIPPOCRATES TO CURE: A 2000 YEAR RETROSPECTIVE Harvey J. Alter, MD, MACP Department of Transfusion Medicine NIH THE SOMMER SEASONS EARLY SOMMER Discovery of HBV Discovery of NANB/HCV Eradication
HCV: FROM HIPPOCRATES TO CURE: A 2000 YEAR RETROSPECTIVE Harvey J. Alter, MD, MACP Department of Transfusion Medicine NIH THE SOMMER SEASONS EARLY SOMMER Discovery of HBV Discovery of NANB/HCV Eradication
Transfusion therapy: what is new with respect to safety and control of iron loading? Yesim AYDINOK
 Transfusion therapy: what is new with respect to safety and control of iron loading? Yesim AYDINOK Following the Policy of the National Regulation 3.3, page 17, on CME disclosures, dated 5 November 2009,
Transfusion therapy: what is new with respect to safety and control of iron loading? Yesim AYDINOK Following the Policy of the National Regulation 3.3, page 17, on CME disclosures, dated 5 November 2009,
Irish Blood Transfusion Service Seirbhís Fuilaistriúcháin na héireann
 Document Detail Irish Blood Transfusion Service Seirbhís Fuilaistriúcháin na héireann Type: Document No.: Title: PMF IBTS SPEC IBTS/PMF/SPEC/0214[2] PLATELETS, ADULT DOSE WITH PLASMA / PAS, IRRADIATED
Document Detail Irish Blood Transfusion Service Seirbhís Fuilaistriúcháin na héireann Type: Document No.: Title: PMF IBTS SPEC IBTS/PMF/SPEC/0214[2] PLATELETS, ADULT DOSE WITH PLASMA / PAS, IRRADIATED
Within 14 days at 2-8⁰C. DO NOT FREEZE WHOLE BLOOD Micro Typing Systems 50 µl plasma
 Test Name *Minimum Collection Tubes Sample Stability ABO/Rh Blood Typing 50 µl cells EDTA whole blood (lavender-top) Within 14 days at 2-8⁰C. DO NOT FREEZE WHOLE BLOOD Micro Typing Systems 50 µl plasma
Test Name *Minimum Collection Tubes Sample Stability ABO/Rh Blood Typing 50 µl cells EDTA whole blood (lavender-top) Within 14 days at 2-8⁰C. DO NOT FREEZE WHOLE BLOOD Micro Typing Systems 50 µl plasma
Guide to the preparation, use and quality assurance of blood components
 Contents Foreword...3 Members of the European Committee (Partial Agreement) on Blood Transfusion... 8 Members of the GTS working group... 22 Members of the TS066 working group... 30 Recommendation No.
Contents Foreword...3 Members of the European Committee (Partial Agreement) on Blood Transfusion... 8 Members of the GTS working group... 22 Members of the TS066 working group... 30 Recommendation No.
The Alphabet Soup of Viral Hepatitis Testing
 The Alphabet Soup of Viral Hepatitis Testing August 18, 2011 Patricia Slev, PhD, DABCC Medical Director, Serologic Hepatitis and Retrovirus Laboratory, ARUP Laboratories Assistant Professor of Pathology,
The Alphabet Soup of Viral Hepatitis Testing August 18, 2011 Patricia Slev, PhD, DABCC Medical Director, Serologic Hepatitis and Retrovirus Laboratory, ARUP Laboratories Assistant Professor of Pathology,
General Terms: Appendix B. National Marrow Donor Program and The Medical College of Wisconsin
 Glossary of Terms This appendix is divided into two sections. The first section, General Terms, defines terms used throughout the CIBMTR data collection forms. The second section, FormsNet TM 2 Terms,
Glossary of Terms This appendix is divided into two sections. The first section, General Terms, defines terms used throughout the CIBMTR data collection forms. The second section, FormsNet TM 2 Terms,
Blood Products & Transfusion. Karim Rafaat, M.D.
 Blood Products & Transfusion Karim Rafaat, M.D. Compatibility Testing Compatibility testing involves three separate procedures involving both donor and recipient blood. 1. ABO & Rh blood type identification
Blood Products & Transfusion Karim Rafaat, M.D. Compatibility Testing Compatibility testing involves three separate procedures involving both donor and recipient blood. 1. ABO & Rh blood type identification
Table of Contents (continued)
 Emerging Molecular and Immunohematology Blood Typing, Grouping And Infectious Disease NAT Screening Assays And Companies Developing New Technologies and Products Table of Contents 1. Blood Typing and Grouping
Emerging Molecular and Immunohematology Blood Typing, Grouping And Infectious Disease NAT Screening Assays And Companies Developing New Technologies and Products Table of Contents 1. Blood Typing and Grouping
Irish Blood Transfusion Service Seirbhís Fuilaistriúcháin na héireann
 Document Detail Seirbhís Fuilaistriúcháin na héireann Type: Document No.: Title: PMF IBTS SPEC IBTS/PMF/SPEC/0219[1] PLATELETS, ADULT DOSE WITH PLASMA / PAS, WASHED, IRRADIATED Owner: 1895 REBECCA WALDEN
Document Detail Seirbhís Fuilaistriúcháin na héireann Type: Document No.: Title: PMF IBTS SPEC IBTS/PMF/SPEC/0219[1] PLATELETS, ADULT DOSE WITH PLASMA / PAS, WASHED, IRRADIATED Owner: 1895 REBECCA WALDEN
2014/LSIF/PD/025 Malaysia s Approach to Testing Strategies
 2014/LSIF/PD/025 Malaysia s Approach to Testing Strategies Submitted by: Malaysia Policy Dialogue and Workshop on Attaining a Safe and Sustainable Blood Supply Chain Manila, Philippines 30 September 1
2014/LSIF/PD/025 Malaysia s Approach to Testing Strategies Submitted by: Malaysia Policy Dialogue and Workshop on Attaining a Safe and Sustainable Blood Supply Chain Manila, Philippines 30 September 1
Confirmed (Laboratory Tests) Serum positive for IgM anti-hbc or, hepatitis B surface antigen (HbsAg).
 Hepatitis B Hepatitis B is a liver disease that results from infection with the Hepatitis B virus. It can range in severity from a mild illness lasting a few weeks to a serious, lifelong illness. Hepatitis
Hepatitis B Hepatitis B is a liver disease that results from infection with the Hepatitis B virus. It can range in severity from a mild illness lasting a few weeks to a serious, lifelong illness. Hepatitis
Immunity and Infection. Chapter 17
 Immunity and Infection Chapter 17 The Chain of Infection Transmitted through a chain of infection (six links) Pathogen: Disease causing microorganism Reservoir: Natural environment of the pathogen Portal
Immunity and Infection Chapter 17 The Chain of Infection Transmitted through a chain of infection (six links) Pathogen: Disease causing microorganism Reservoir: Natural environment of the pathogen Portal
Duration: 12 months May to April
 SPECIALIST CERTIFICATE IN TRANSFUSION SCIENCE PRACTICE PROGRAMME OF STUDY OVERVIEW Example only Duration: 12 months May to April This document serves as a general programme overview only. To ensure you
SPECIALIST CERTIFICATE IN TRANSFUSION SCIENCE PRACTICE PROGRAMME OF STUDY OVERVIEW Example only Duration: 12 months May to April This document serves as a general programme overview only. To ensure you
Public Health, Infections and Transplantation
 Public Health, Infections and Transplantation Dr Kerry Chant Chief Health Officer & Deputy Director General Population and Public Health NSW Ministry of Health May2014 Public Health Infection and Transplantation
Public Health, Infections and Transplantation Dr Kerry Chant Chief Health Officer & Deputy Director General Population and Public Health NSW Ministry of Health May2014 Public Health Infection and Transplantation
Crossmatching and Issuing Blood Components; Indications and Effects.
 Crossmatching and Issuing Blood Components; Indications and Effects. Alison Muir Blood Transfusion, Blood Sciences, Newcastle Trust Topics Covered Taking the blood sample ABO Group Antibody Screening Compatibility
Crossmatching and Issuing Blood Components; Indications and Effects. Alison Muir Blood Transfusion, Blood Sciences, Newcastle Trust Topics Covered Taking the blood sample ABO Group Antibody Screening Compatibility
Objectives. Methods. Results. Economic
 Octaplas Compared with Fresh Frozen Plasma to Reduce the Risk of Transmitting Lipid-Enveloped Viruses: An Economic Analysis and Budget Impact Analysis [Adapted from Membe SK, Coyle D, Husereau D, Cimon
Octaplas Compared with Fresh Frozen Plasma to Reduce the Risk of Transmitting Lipid-Enveloped Viruses: An Economic Analysis and Budget Impact Analysis [Adapted from Membe SK, Coyle D, Husereau D, Cimon
Immunodeficiency. (2 of 2)
 Immunodeficiency (2 of 2) Acquired (secondary) immunodeficiencies More common Many causes such as therapy, cancer, sarcoidosis, malnutrition, infection & renal disease The most common of which is therapy-related
Immunodeficiency (2 of 2) Acquired (secondary) immunodeficiencies More common Many causes such as therapy, cancer, sarcoidosis, malnutrition, infection & renal disease The most common of which is therapy-related
For more information about how to cite these materials visit
 Author(s): Robertson Davenport, M.D., 2009 License: Unless otherwise noted, this material is made available under the terms of the Creative Commons Attribution Noncommercial Share Alike 3.0 License: http://creativecommons.org/licenses/by-nc-sa/3.0/
Author(s): Robertson Davenport, M.D., 2009 License: Unless otherwise noted, this material is made available under the terms of the Creative Commons Attribution Noncommercial Share Alike 3.0 License: http://creativecommons.org/licenses/by-nc-sa/3.0/
TRANSFUSION REACTIONS
 14 TRANSFUSION REACTIONS 14.1 INTRODUCTION Transfusion of blood and blood products are reported to cause reactions during or after procedure specially in patients who receive multiple transfusions. These
14 TRANSFUSION REACTIONS 14.1 INTRODUCTION Transfusion of blood and blood products are reported to cause reactions during or after procedure specially in patients who receive multiple transfusions. These
Blood transfusion. Dr. J. Potgieter Dept. of Haematology NHLS - TAD
 Blood transfusion Dr. J. Potgieter Dept. of Haematology NHLS - TAD General Blood is collected from volunteer donors >90% is separated into individual components and plasma Donors should be: healthy, have
Blood transfusion Dr. J. Potgieter Dept. of Haematology NHLS - TAD General Blood is collected from volunteer donors >90% is separated into individual components and plasma Donors should be: healthy, have
Dengue & Chikungunya In Asia Strategies for Testing
 Dengue & Chikungunya In Asia Strategies for Testing Sally Lam Blood Services Group Health Sciences Authority Singapore 1 The Viruses - Arboviruses Flavivirus DENGUE Enveloped virus, ~50 nm in diameter
Dengue & Chikungunya In Asia Strategies for Testing Sally Lam Blood Services Group Health Sciences Authority Singapore 1 The Viruses - Arboviruses Flavivirus DENGUE Enveloped virus, ~50 nm in diameter
Risks and Benefits of Blood Transfusions. Objectives. Red Cells (Erythrocytes) Understand the following:
 Risks and Benefits of Blood Transfusions Patient and Family Conference Aplastic Anemia & MDS International Foundation July 10-12 th, 2009 Indianapolis, Indiana Susan M. Carson RN, MSN, CPNP Childrens Hospital
Risks and Benefits of Blood Transfusions Patient and Family Conference Aplastic Anemia & MDS International Foundation July 10-12 th, 2009 Indianapolis, Indiana Susan M. Carson RN, MSN, CPNP Childrens Hospital
Impact of Testing Strategies to Reduce Transmission Risk for HBV. Ravi Reddy, M Vermeulen South African National Blood Service (SANBS) 29 July 2013
 Impact of Testing Strategies to Reduce Transmission Risk for HBV Ravi Reddy, M Vermeulen South African National Blood Service (SANBS) 29 July 2013 Overview of SANBS SANBS is a private not for profit company
Impact of Testing Strategies to Reduce Transmission Risk for HBV Ravi Reddy, M Vermeulen South African National Blood Service (SANBS) 29 July 2013 Overview of SANBS SANBS is a private not for profit company
Emerging TTIs How Singapore secure its blood supply
 Emerging TTIs How Singapore secure its blood supply Ms Sally Lam Acting Division Director I Blood Supply Management I Blood Services Group I Health Sciences Outline Risk Mitigation Strategies to secure
Emerging TTIs How Singapore secure its blood supply Ms Sally Lam Acting Division Director I Blood Supply Management I Blood Services Group I Health Sciences Outline Risk Mitigation Strategies to secure
WHO GUIDELINES ON ESTIMATION OF RESIDUAL RISK OF HIV, HBV OR HCV INFECTIONS VIA CELLULAR BLOOD COMPONENTS AND PLASMA
 ENGLISH ONLY EXPERT COMMITTEE ON BIOLOGICAL STANDARDIZATION Geneva, 17 to 21 October 2016 WHO GUIDELINES ON ESTIMATION OF RESIDUAL RISK OF HIV, HBV OR HCV INFECTIONS VIA CELLULAR BLOOD COMPONENTS AND PLASMA
ENGLISH ONLY EXPERT COMMITTEE ON BIOLOGICAL STANDARDIZATION Geneva, 17 to 21 October 2016 WHO GUIDELINES ON ESTIMATION OF RESIDUAL RISK OF HIV, HBV OR HCV INFECTIONS VIA CELLULAR BLOOD COMPONENTS AND PLASMA
HEPATITIS VIRUSES. Other causes (not exclusively hepatitis v.)also called sporadic hepatitis: HEPATITIS A(infectious hepatitis)
 Dept.of Microbiology/Virology Assist.prof. Shatha F. Abdullah HEPATITIS VIRUSES Medically important hepatitis v. (liver)are: 1.HAV 2.HBV 3.HCV 4.HDV 5.HEV 6.HGV Other causes (not exclusively hepatitis
Dept.of Microbiology/Virology Assist.prof. Shatha F. Abdullah HEPATITIS VIRUSES Medically important hepatitis v. (liver)are: 1.HAV 2.HBV 3.HCV 4.HDV 5.HEV 6.HGV Other causes (not exclusively hepatitis
DHQ Flowcharts v2.0 eff. February 2016
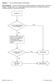 Question: 1. Are you feeling healthy and well today? Donor Eligibility: A person should be free of infectious diseases, including colds, on the day of donation. A person who is not in good health should
Question: 1. Are you feeling healthy and well today? Donor Eligibility: A person should be free of infectious diseases, including colds, on the day of donation. A person who is not in good health should
2017 CST-Astellas Canadian Transplant Fellows Symposium. Optimizing use of organs from Increased Risk Donors
 2017 CST-Astellas Canadian Transplant Fellows Symposium Optimizing use of organs from Increased Risk Donors Atual Humar, MD Atul Humar is a Professor in the Department of Medicine, University of Toronto.
2017 CST-Astellas Canadian Transplant Fellows Symposium Optimizing use of organs from Increased Risk Donors Atual Humar, MD Atul Humar is a Professor in the Department of Medicine, University of Toronto.
Playing Detective: going in search of the source of infection. Su Brailsford Consultant in Epidemiology and Health Protection
 Playing Detective: going in search of the source of infection Su Brailsford Consultant in Epidemiology and Health Protection The Micro Services clinical team Are inquisitive Like problem solving Don t
Playing Detective: going in search of the source of infection Su Brailsford Consultant in Epidemiology and Health Protection The Micro Services clinical team Are inquisitive Like problem solving Don t
Chapter 19: The Genetics of Viruses and Bacteria
 Chapter 19: The Genetics of Viruses and Bacteria What is Microbiology? Microbiology is the science that studies microorganisms = living things that are too small to be seen with the naked eye Microorganisms
Chapter 19: The Genetics of Viruses and Bacteria What is Microbiology? Microbiology is the science that studies microorganisms = living things that are too small to be seen with the naked eye Microorganisms
Laboratory reports of hepatitis A and C infection in England and Wales: January to March
 Volume 4 29 Published on: 23 July 2010 Current News Increased diagnoses of HIV infection in the over-50s in UK Infection Reports Immunisation Laboratory reports of hepatitis A and C infection in England
Volume 4 29 Published on: 23 July 2010 Current News Increased diagnoses of HIV infection in the over-50s in UK Infection Reports Immunisation Laboratory reports of hepatitis A and C infection in England
In Canada and other developed countries, many steps are
 PAEDIATRIC INFECTIOUS DISEASE NOTES Transfusion and risk of infection in Canada: Update 2006 In Canada and other developed countries, many steps are taken to minimize the risk of infection from transfusion
PAEDIATRIC INFECTIOUS DISEASE NOTES Transfusion and risk of infection in Canada: Update 2006 In Canada and other developed countries, many steps are taken to minimize the risk of infection from transfusion
TRANSMISSIBLE SPONGIFORM ENCEPHALOPATHIES ADVISORY COMMITTEE MEETING October2010. Issue Summary
 TRANSMISSIBLE SPONGIFORM ENCEPHALOPATHIES ADVISORY COMMITTEE MEETING 28-29October2010 Issue Summary Informational Topic: FDA s Geographic Donor Deferral Policy to Reduce the Possible Risk of Transmission
TRANSMISSIBLE SPONGIFORM ENCEPHALOPATHIES ADVISORY COMMITTEE MEETING 28-29October2010 Issue Summary Informational Topic: FDA s Geographic Donor Deferral Policy to Reduce the Possible Risk of Transmission
Chapter 08 Lecture Outline
 Chapter 08 Lecture Outline See separate PowerPoint slides for all figures and tables preinserted into PowerPoint without notes. Copyright 2016 McGraw-Hill Education. Permission required for reproduction
Chapter 08 Lecture Outline See separate PowerPoint slides for all figures and tables preinserted into PowerPoint without notes. Copyright 2016 McGraw-Hill Education. Permission required for reproduction
Transfusion Pitfalls. Objectives. Packed Red Blood Cells. TRICC trial (subgroups): Is transfusion always good? Components
 Objectives Transfusion Pitfalls Gregory W. Hendey, MD, FACEP Professor and Chief UCSF Fresno, Emergency Medicine To list risks and benefits of various blood products To discuss controversy over liberal
Objectives Transfusion Pitfalls Gregory W. Hendey, MD, FACEP Professor and Chief UCSF Fresno, Emergency Medicine To list risks and benefits of various blood products To discuss controversy over liberal
Answering your daily challenges. in the ELISA technology I N FECTI OUS DISEASES. bioelisa
 Answering your daily challenges in the ELISA technology I N FECTI OUS DISEASES bioelisa RETROVIRUS HIV is the cause of the most important global pandemic. Due to the lack of vaccination, early diagnosis
Answering your daily challenges in the ELISA technology I N FECTI OUS DISEASES bioelisa RETROVIRUS HIV is the cause of the most important global pandemic. Due to the lack of vaccination, early diagnosis
Inspections, Compliance, Enforcement, and Criminal Investigations. Central Texas Regional Blood & Tissue Center 07-Nov-03
 1 of 7 6/10/2009 2:20 PM Inspections, Compliance, Enforcement, and Criminal Investigations Department of Health and Human Services Public Health Service Food and Drug Administration Dallas District 4040
1 of 7 6/10/2009 2:20 PM Inspections, Compliance, Enforcement, and Criminal Investigations Department of Health and Human Services Public Health Service Food and Drug Administration Dallas District 4040
Chapter 13 Viruses, Viroids, and Prions. Biology 1009 Microbiology Johnson-Summer 2003
 Chapter 13 Viruses, Viroids, and Prions Biology 1009 Microbiology Johnson-Summer 2003 Viruses Virology-study of viruses Characteristics: acellular obligate intracellular parasites no ribosomes or means
Chapter 13 Viruses, Viroids, and Prions Biology 1009 Microbiology Johnson-Summer 2003 Viruses Virology-study of viruses Characteristics: acellular obligate intracellular parasites no ribosomes or means
A Transfusion Reaction What Do I Do Now? Judith A. Sullivan, MS, MT(ASCP)SBB, CQA(ASQ) ASCLS Region III Triennial Meeting Birmingham AL
 A Transfusion Reaction What Do I Do Now? Judith A. Sullivan, MS, MT(ASCP)SBB, CQA(ASQ) ASCLS Region III Triennial Meeting Birmingham AL This promotional educational activity is brought to you by Ortho
A Transfusion Reaction What Do I Do Now? Judith A. Sullivan, MS, MT(ASCP)SBB, CQA(ASQ) ASCLS Region III Triennial Meeting Birmingham AL This promotional educational activity is brought to you by Ortho
Emerging Pathogens that Impact the Canadian Blood Supply Alberta Vein to Vein Conference March 18-19, 2016
 Emerging Pathogens that Impact the Canadian Blood Supply Alberta Vein to Vein Conference March 18-19, 2016 Dr. Margaret Fearon Medical Director, Medical Microbiology, Canadian Blood Services March 18,
Emerging Pathogens that Impact the Canadian Blood Supply Alberta Vein to Vein Conference March 18-19, 2016 Dr. Margaret Fearon Medical Director, Medical Microbiology, Canadian Blood Services March 18,
Special Requirements Lab Matters. 18 th May Katy Cowan PBM Practitioner NHSBT
 Special Requirements Lab Matters. 18 th May 2016 Katy Cowan PBM Practitioner NHSBT Special requirements Frozen components and platelets HEV negative Irradiated CMV negative Frozen components and platelets
Special Requirements Lab Matters. 18 th May 2016 Katy Cowan PBM Practitioner NHSBT Special requirements Frozen components and platelets HEV negative Irradiated CMV negative Frozen components and platelets
Sustainable cleaning of the health care environment.
 Sustainable cleaning of the health care environment. Has the current practice of cleaning the health care environment relying on the use of disinfectants and their label claims and instructions for use
Sustainable cleaning of the health care environment. Has the current practice of cleaning the health care environment relying on the use of disinfectants and their label claims and instructions for use
Structure and duties of Blood service
 NATIONAL CENTER FOR TRANSFUSION MEDICINE Prevalence of infectious diseases among Mongolian blood donors Namjil Erdenebayar General director of The National Center for Transfusion Medicine, Mongolia NCTM,
NATIONAL CENTER FOR TRANSFUSION MEDICINE Prevalence of infectious diseases among Mongolian blood donors Namjil Erdenebayar General director of The National Center for Transfusion Medicine, Mongolia NCTM,
Rassegna. Pathogen inactivation of labile blood products. Andreas Morell
 Rassegna Pathogen inactivation of labile blood products Andreas Morell ZLB Central Laboratory Swiss Red Cross, Bern, Switzerland Introduction Selection of donors and screening of donated blood and plasma
Rassegna Pathogen inactivation of labile blood products Andreas Morell ZLB Central Laboratory Swiss Red Cross, Bern, Switzerland Introduction Selection of donors and screening of donated blood and plasma
Coding for Preventive Services A Guide for HIV Providers
 Coding for Preventive Services A Guide for HIV Providers Jessie Murphy, MPH and Michelle Cataldo, LCSW, April 2016 Implementation of the Patient Protection and Affordable Care Act and other regulatory
Coding for Preventive Services A Guide for HIV Providers Jessie Murphy, MPH and Michelle Cataldo, LCSW, April 2016 Implementation of the Patient Protection and Affordable Care Act and other regulatory
Infection, Detection, Prevention...
 Infection, Detection, Prevention... A disease is any change that disrupts the normal function of one or more body systems. Non infectious diseases are typically caused by exposure to chemicals or are inherited.
Infection, Detection, Prevention... A disease is any change that disrupts the normal function of one or more body systems. Non infectious diseases are typically caused by exposure to chemicals or are inherited.
DHQ v2.0, February 2016 Table Detailing Changes from v1.3 v1.3 Revised language in v2.0 Rationale Question 1: Are you
 DHQ v2.0, February 2016 Table Detailing s from v1.3 v1.3 Revised language in v2.0 Rationale Question 1: Are you Q 1, No Feeling healthy and well today? Question 2: Are you Q 2, No change Currently taking
DHQ v2.0, February 2016 Table Detailing s from v1.3 v1.3 Revised language in v2.0 Rationale Question 1: Are you Q 1, No Feeling healthy and well today? Question 2: Are you Q 2, No change Currently taking
Hepa%%s E Virus Is it a Concern?
 IPFA/PEI 17 th Workshop on Surveillance and Screening of Blood Borne Pathogens 26-27 May 2010, The Regent Esplanade Zagreb Hotel, Zagreb, Croa%a Hepa%%s E Virus Is it a Concern? Keiji Matsubayashi Hokkaido
IPFA/PEI 17 th Workshop on Surveillance and Screening of Blood Borne Pathogens 26-27 May 2010, The Regent Esplanade Zagreb Hotel, Zagreb, Croa%a Hepa%%s E Virus Is it a Concern? Keiji Matsubayashi Hokkaido
PROPOSED TECHNICAL REQUIREMENTS BLOOD AND BLOOD COMPONENTS. File 1 DRAFT FOR CONSULTATION PURPOSES ONLY
 PROPOSED TECHNICAL REQUIREMENTS BLOOD AND BLOOD COMPONENTS File 1 DRAFT FOR CONSULTATION PURPOSES ONLY ANNEX I INFORMATION REQUIREMENTS A. INFORMATION TO BE PROVIDED TO DONORS 1. Accurate but generally
PROPOSED TECHNICAL REQUIREMENTS BLOOD AND BLOOD COMPONENTS File 1 DRAFT FOR CONSULTATION PURPOSES ONLY ANNEX I INFORMATION REQUIREMENTS A. INFORMATION TO BE PROVIDED TO DONORS 1. Accurate but generally
MP Biomedicals Asia Pacific Pte. Ltd. (formerly Genelabs Diagnostics Pte. Ltd.)
 Revision: 12 May 2005 1 WESTERN BLOT / IMMUNOBLOT HIV-1 BLOT Version 1.3* 11010-018 HIV-1 viral lysate Western Blot assay for the 11010-036 detection of antibodies to HIV-1 with serum 11010-108 108 strips
Revision: 12 May 2005 1 WESTERN BLOT / IMMUNOBLOT HIV-1 BLOT Version 1.3* 11010-018 HIV-1 viral lysate Western Blot assay for the 11010-036 detection of antibodies to HIV-1 with serum 11010-108 108 strips
