2017 CST-Astellas Canadian Transplant Fellows Symposium. Optimizing use of organs from Increased Risk Donors
|
|
|
- Gwen Powell
- 5 years ago
- Views:
Transcription
1 2017 CST-Astellas Canadian Transplant Fellows Symposium Optimizing use of organs from Increased Risk Donors Atual Humar, MD Atul Humar is a Professor in the Department of Medicine, University of Toronto. Dr. Humar received his medical degree from the University of Ottawa. He completed his residency and did further training in Transplant Infectious Diseases in Toronto and Boston. Dr. Humar s research interests are in virology with a focus on the pathogenesis of herpesvirus infections posttransplant. He is involved in both basic and clinical research assessing immunologic and virologic determinants of infection. Dr. Humar is the Director of Multi Organ Transplant Program at the University Health Network and the University of Toronto Transplant Institute. He is also active in the Canadian Society of Transplantation as a President and has been very active in both the AST and TTS. Dr. Humar operates a joint research lab with his wife, Dr. Deepali Kumar, who is also a faculty member at the University of Toronto.
2 Optimizing use of organs from Increased Risk Donors ATUL HUMAR M.D. University Health Network Toronto
3 Conflict of Interest Disclosure No relevant COI for this talk
4 HIV, HCV,HBV Potentially infected donor Known pathogen Unknown pathogen Bacterial infection CMV, EBV Others
5 Potential DDI Pathogens VIRAL Hepatitis A, B, C, D HIV HHV 1-8 Rabies West Nile Virus LCMV PARASITIC Malaria Chagas disease Strongyloides Schistosomiasis Flukes PATHOGENS BACTERIAL Gram positive Gram negative Mycobacterial Spirochetes PRION vcjd FUNGAL Candida Cryptocococcus Coccidioides Histoplasma Aspergillus
6 Notable Organ Transplant- Transmitted Infections: Published Literature HIV, 1985 HCV, 2000 Chagas Disease (T. cruzi), 2001 West Nile Virus (WNV), GA 2002 Lymphocytic Choriomeningitis Virus (LCMV) 2003; 2005 Rabies, 2004, 2005 WNV, NY/PA 2005 Chagas, 2006 M. TB HIV/HCV 2007 HCV 2009 Balamuthia mandrillaris HCV 2011 HIV 2011
7 Standard Donor Testing 2017 Step 1. Screening by history Step 2. Screening by laboratory results HIV antibody [Ab/Ag assay] Hepatitis B surface antigen, HepB core Ab Hepatitis C antibody syphilis serology CMV antibody EBV antibody HTLV-I (and II) (no HTLV testing in US) Cultures
8 Additional Donor Testing NAT testing (geographic variation) HIV, HCV, HBV West Nile Virus Serologies Fungal, Chagas/T.cruzi, Strongyloides (selected situations)
9 Issues with Donor Testing 2017 Serologies can be confounded Blood products Hemodilution Recent exposure (window period) Turn around time especially NAT Single tests done especially NAT False positives organ discard
10 CASE # 1: The High risk donor You are called about a 25 year old deceased donor HCV Antibody negative HIV Antibody negative HBV testing negative [HbSAg] AST, ALT, ALP normal Donor found in a bathroom stall with a needle in arm In hospital x 1 day
11 CASE # 1 NAT for HIV/HCV/HBV negative. Would you use the organs? a) Yes for any consenting recipient b) Yes higher-status or special case recipient only c) Decline organs in most cases except certain exceptional circumstances
12 Summary of Canadian Standards Association increased-risk donor criteria CSA criteria for increased-risk donors Nonmedical intravenous, intramuscular, or subcutaneous injection of drugs in the preceding 5 years Men who have had sex with another man in the preceding 5 years Persons who have engaged in sex in exchange for money or drugs in the preceding five years Persons who have had sex in the preceding 12 months with any of the above persons or a person known or suspected to have HIV, HCV or HBV infection. Exposure in preceding 12 months through percutaneous inoculation or open wound Prison, lock up, jail or juvenile detention >72 hours in the past 12 months Non-sterile tattooing, piercings in the past 12 months Close contact with anyone with clinically active viral hepatitis (living in the same house where kitchen and bathroom are shared) in the past 12 months
13 CHICAGO TRANSMISSION CASE 2007: Multiple organs transplanted from a single donor Donor HCV Ab and HIV Ab negative Classified as CDC-IRD (high risk donor) 4-recipients infected with HIV and HCV Retrospective testing of donor sera showed NAT + for HIV/HCV
14 What is the residual risk? If the serology and NAT are negative what is the risk of transmission? Answer not very often but sometimes Depends on risk behaviours in donor
15 Window Period
16 What is the Window Period Pathogen serology Nucleic Acid Testing HIV days 5-6 days HCV ~ 70 days 3 5 days (best test; many assume 7 days) HBV days days Humar et al; NAT Consensus conference. Am J Transplant 2010
17 Likelihood of being infected in last 7 days Using drugs for 10 years Negative for HIV/HCV
18 Risk per 10,000 donors of an HIV infection occurring during the window period, by ELISA and NAT Risk Category Men who have sex with men ELISA Per 10, ( ) Intravenous drug use 6.6 ( ) Commercial sex worker 3.7 ( ) Sex with a partner in above categories 0.7 ( ) NAT Per 10, ( ) 2.7 ( ) 1.5 ( ) 0.3 ( ) Risk of window period infection expressed as ratio 1: :3704 1:6667 1:33,333 HIV Exposed through blood 1.5 ( ) Incarcerated 1.0 ( ) 0.6 ( ) 0.4 ( ) 1:16,667 1: 25,000 CST/CNTRP Consensus guideline; Transplantation 2014
19 Risk per 10,000 donors of an HCV infection occurring during the window period, by ELISA and NAT Risk Category ELISA Per 10,000 Men who have sex with men 14.3 ( ) Intravenous drug use ( ) Commercial sex worker ( ) Sex with a partner in above categories ( ) Exposed through blood 13.9 ( ) Incarcerated ( ) NAT Per 10, ( ) 40.8 ( ) 29.1 ( ) 18.0 ( ) 1.4 ( ) 11.5 ( ) Risk of window period infection expressed as ratio 1: :245 1:344 1:556 1:7143 1: 870
20 HOWEVER HCV NOW VERY TREATABLE Risk Category Men who have sex with men Intravenous drug use Commercial sex worker Sex with a partner in above categories Risk of window period infection expressed as ratio Chance of cure with DAA Chance of chronic HCV infection 1: : 133,340 1: : 4,900 1: : 6,800 1: : 11,120 Exposed through blood 1: : 142,860 Incarcerated 1: : 17,400
21 Estimated Canadian wait list numbers and mortality Number on the wait list Death on the wait list Heart % Lung % Liver % Kidney % Kidney Pancreas 56 9% Source: Canadian Organ Replacement Register, 2012, CIHI; Data extracted for year 2009
22 RISKY DONORS: CONSIDERATIONS Individual Risk vs. benefit of transplantation Risk of waiting until next available offer Is patient on an alternate list [e.g. HCV +] Societal Acceptance beneficial from a societal perspective (Schwietzer et al. AJT 2007) by adding organs into the pool Programmatic An transmission incident may reflect poorly on a transplant program.
23 The New York Times Sun, Jul 10, 2005
24
25
26 Considerations Many donor are substance abuse but not IVDU we count as HRD but lower risk In hospital time is important In hospital x 7d with NAT means near normal risk History in donors often not reliable
27 Likelihood of accepting organ Kumar et al. Transplantation 2015
28 REASONS FOR DECLINE
29 STANDARDIZED INFORMED CONSENT Read from a script Describe the risk to the patient Provide the alternatives Document the informed consent process
30 FOLLOW-UP TESTING IS ESSENTIAL Post-transplant Test Timing of Test HIV, HCV NAT HBV NAT or HBsAg At 1 and 3 months post-transplant Anti-HBs, anti-hbc, and either HBV NAT or HBsAg At 12 months post-transplant CST/CNTRP Consensus guideline; Transplantation 2014
31 What about the HCV positive donor? HCV Ab + /NAT + HCV Ab+ /NAT-ve Recipient may also be any one of these
32 How many donors? Estimated Risk of Human Immunodeficiency Virus and Hepatitis C Virus Infection among Potential Organ Donors from 17 Organ Procurement Organizations in the United States The overall adjusted prevalence for anti-hcv was 5.58% (CI = %). The adjusted anti-hcv prevalence was lowest for normal risk donors at 3.45% (CI = ), and highest for high risk donors at 18.20% (CI = %). Ellingsona K et al. AJT 2011; 11:
33 If you are HCV Ab+ / NAT negative is there residual virus? This result strongly suggests that SVR may be considered to show eradication of HCV infection. Maylin et al. Gastroenterology 2008
34 WHAT ABOUT THE HCV NAT POSITIVE DONOR? ASSUME 100% RISK OF TRANSMISSION
35 SVR12 (%) ARE IMPLICATIONS OF TRANSMISSION CHANGING? Sofosbuvir/Ledipasvir ± RBV for 12 weeks Nuc Pol Inhibitor NS5A Inhibitor Most Cured with 1 pill a day for 12 weeks! No cirrhosis Cirrhosis 20 0 SOF/LDV SOF/LDV + RBV SOF/LDV SOF/LDV + RBV 12 Wks 24 Wks ION 1 Study Afdhal N, et al. N Engl J Med. 2014;370:
36 Case 3 Donor is 35 y.o. female NDD; ICH 2% to endocarditis from IVDU; in hospital x 2 weeks Adequate antibiotic treatment for strep viridans endocarditis LFTs, Cr, imaging normal HCV NAT+, HCV Ab+ HIV NAT -ve
37 Would you: a) Decline all together b) Accept only for HCV NAT+ recipient c) Accept for any recipient (HCV NAT -ve) d) Accept for special list recipient (HCV NATve but eg. highly sensitized, very sick etc)
38 HCV D+/R-
39 HCV KIDNEY TRIAL (Goldberg et al) HCV NAT +ve Donor Transplant to HCV NAT -ve recipient Age >50 1 st Kidney Decreased donor PRA <20% Treat with Grazoprevir/Elbasvir (Zepatier) +/- Ribavirin +/- Sofosbuvir Depending on genotype
40 10 patients Median wait time 58 days IAQR days Median KDPI score 42% Goldberg et al. NEJM 2017
41 HCV LUNG TRIAL Cypel/Humar HCV NAT Positive Donor Ex Vivo lung with high volume wash 6 hours Transplant in HCV NAT -ve recipient Negative: NO Treatment Week PCR X 12 Positive: Treat with Sofosbuvir/ velpatasvir (Epclusa)
42 Recommendations for utilization of increased risk donors Transplant physicians and surgeons should consider the utilization of organs from IRDs. This should be done in conjunction with NAT testing for HCV and HIV, and HBsAg or NAT for HBV. Some development work is required to ensure that NAT is available across all jurisdictions in Canada (strong recommendation, moderate quality evidence). All potential recipients should be made aware of the option to consider accepting organs from IRDs and that declining such an offer will not impact their waiting time for a standard risk (non-ird) organ.
43 Recommendations for utilization of increased risk donors Appropriate informed consent from potential recipients should be obtained. It is suggested that a standardized informed consent process be used Information provided at the time of listing Standardized IC at time of transplant Decisions around utilization or non-utilization may take into account the timing of increased risk behaviour, the window period for the specific test used, and the status of the recipient as well as other recipient-specific circumstances.
44 Recommendations RE HCV Definition of HCV positive donor should include NAT results ( HCV Viremic donor ) HCV Ab + / NAT negative donor Consider using all organs for any appropriately consented recipient Appropriate f/u testing important We should keep a national database of such transplants
45 Recommendations HCV NAT Positive donor Use preferentially in HCV NAT positive recipient Consider using in selected NAT negative recipients with following considerations For now should normally be in the context of a research study May be very sick or special circumstance Should have access to DAAs post transplant We should track all such cases nationally
46 Thank You Questions / Discussion atul.humar@uhn.ca
Increased Risk Donors. November 1, 2018 BC Kidney Days. Jagbir Gill MD MPH Associate Professor of Medicine UBC
 Increased Risk Donors November 1, 2018 BC Kidney Days Jagbir Gill MD MPH Associate Professor of Medicine UBC Median dialysis exposure prior to DDKT Case scenarios Which donors should we accept kidneys
Increased Risk Donors November 1, 2018 BC Kidney Days Jagbir Gill MD MPH Associate Professor of Medicine UBC Median dialysis exposure prior to DDKT Case scenarios Which donors should we accept kidneys
Increased Risk Donors for Organ Transplantation
 A Tool Kit to Assist Transplant Programs in the Use of Increased Risk Donors for Organ Transplantation February 2016 Table of Contents Table of Contents... 2 1.0 Increased Risk Donor Tool Kit... 3 1.1
A Tool Kit to Assist Transplant Programs in the Use of Increased Risk Donors for Organ Transplantation February 2016 Table of Contents Table of Contents... 2 1.0 Increased Risk Donor Tool Kit... 3 1.1
RISK OF DISEASE TRANSMISSION FROM ORGAN DONORS PATIENT INFORMATION GUIDE
 Receiving an organ transplant carries many risks, including the risk of getting a disease from the June 2017 donor. This is true for every organ we transplant. BC Transplant makes every effort to minimize
Receiving an organ transplant carries many risks, including the risk of getting a disease from the June 2017 donor. This is true for every organ we transplant. BC Transplant makes every effort to minimize
Current Infectious Disease Screening for the Live Organ Donor
 2013 Public Health and Safety Guidelines for reducing HIV, HBV and HCV Transmission Though Organ Transplantation: Implications for Counseling and Disclosure Dianne LaPointe Rudow DNP, ANP-BC, CCTC Director
2013 Public Health and Safety Guidelines for reducing HIV, HBV and HCV Transmission Though Organ Transplantation: Implications for Counseling and Disclosure Dianne LaPointe Rudow DNP, ANP-BC, CCTC Director
Update on organ donation in British Columbia
 Update on organ donation in British Columbia Jagbir Gill MD MPH FRCPC Associate Professor of Medicine, UBC Vice President, CORR Board 2017 BC Kidney Days October 5, 2017 Canadian Institute for Health Information
Update on organ donation in British Columbia Jagbir Gill MD MPH FRCPC Associate Professor of Medicine, UBC Vice President, CORR Board 2017 BC Kidney Days October 5, 2017 Canadian Institute for Health Information
Patient Name: MRN: DOB: Treatment Location:
 Page 1 of 5 I. TO (Required) This Section is required to be completed by all patients who undergo kidney transplant surgery. I hereby consent to and authorize Dr. and his/her assistant(s), including supervised
Page 1 of 5 I. TO (Required) This Section is required to be completed by all patients who undergo kidney transplant surgery. I hereby consent to and authorize Dr. and his/her assistant(s), including supervised
Risky Business Preventing Disease Transmission From Donor Organs. Peter Chin-Hong, MD UCSF September 2012
 Risky Business Preventing Disease Transmission From Donor Organs Peter Chin-Hong, MD UCSF September 2012 Case 1: Something rare? 54 yo WM with HBV/HCV/HCC Day 5: Fever to 102.4, mild frontal HA since time
Risky Business Preventing Disease Transmission From Donor Organs Peter Chin-Hong, MD UCSF September 2012 Case 1: Something rare? 54 yo WM with HBV/HCV/HCC Day 5: Fever to 102.4, mild frontal HA since time
Risk of Other Donor-Derived Infections (nonhiv, nonhcv) Daniel Kaul MD Associate Professor University of Michigan
 Risk of Other Donor-Derived Infections (nonhiv, nonhcv) Daniel Kaul MD Associate Professor University of Michigan Conflict of Interest Disclosure I have no relevant financial relationships to disclose
Risk of Other Donor-Derived Infections (nonhiv, nonhcv) Daniel Kaul MD Associate Professor University of Michigan Conflict of Interest Disclosure I have no relevant financial relationships to disclose
Screening donors and donations for transfusion transmissible infectious agents. Alan Kitchen
 Screening donors and donations for transfusion transmissible infectious agents Alan Kitchen Aim Not to teach you microbiology To provide and awareness of the big picture To provide an understanding of
Screening donors and donations for transfusion transmissible infectious agents Alan Kitchen Aim Not to teach you microbiology To provide and awareness of the big picture To provide an understanding of
2017 CST-Astellas Canadian Transplant Fellows Symposium. EBV Post Transplantation Implications and Approach to Management
 2017 CST-Astellas Canadian Transplant Fellows Symposium EBV Post Transplantation Implications and Approach to Management Atul Humar, MD Atul Humar is a Professor in the Department of Medicine, University
2017 CST-Astellas Canadian Transplant Fellows Symposium EBV Post Transplantation Implications and Approach to Management Atul Humar, MD Atul Humar is a Professor in the Department of Medicine, University
Improving Utilization of Organs from Increased Risk Donors. Matthew J. Kuehnert, MD CDC
 Improving Utilization of Organs from Increased Risk Donors Matthew J. Kuehnert, MD CDC Conflict of Interest Disclosure I have no relevant financial relationships to disclose. 2016 AST Improving Utilization
Improving Utilization of Organs from Increased Risk Donors Matthew J. Kuehnert, MD CDC Conflict of Interest Disclosure I have no relevant financial relationships to disclose. 2016 AST Improving Utilization
Public Health, Infections and Transplantation
 Public Health, Infections and Transplantation Dr Kerry Chant Chief Health Officer & Deputy Director General Population and Public Health NSW Ministry of Health May2014 Public Health Infection and Transplantation
Public Health, Infections and Transplantation Dr Kerry Chant Chief Health Officer & Deputy Director General Population and Public Health NSW Ministry of Health May2014 Public Health Infection and Transplantation
HEALTH SCREENING QUESTIONNAIRE. Work-up. Donor ID EdgeCell #:
 HEALTH SCREENING QUESTIONNAIRE Héma-Québec Registre des Donneurs de Cellules Souches 4045 Côte-vertu, St-Laurent, QC, Canada, H4R 2W7 Tél : + 514-832-1031 Fax : + 514-832-0266 www.hema-quebec.qc.ca CT
HEALTH SCREENING QUESTIONNAIRE Héma-Québec Registre des Donneurs de Cellules Souches 4045 Côte-vertu, St-Laurent, QC, Canada, H4R 2W7 Tél : + 514-832-1031 Fax : + 514-832-0266 www.hema-quebec.qc.ca CT
Non-reproductive tissues and cells Recommending authority/ association
 Colour key Minimum requirements as set out in Directive 2004/23/EC More stringent - legy binding More stringent - recommended Not legy binding and not recommended Tested pathogen Donor test/ technique
Colour key Minimum requirements as set out in Directive 2004/23/EC More stringent - legy binding More stringent - recommended Not legy binding and not recommended Tested pathogen Donor test/ technique
Directed Semen Donor Eligibility Requirements
 Directed Semen Donor Eligibility Requirements Directed (known) reproductive donors using the University Andrology Sperm Banking Program must follow FDA requirements to prevent the transmission of relevant
Directed Semen Donor Eligibility Requirements Directed (known) reproductive donors using the University Andrology Sperm Banking Program must follow FDA requirements to prevent the transmission of relevant
Non-reproductive tissues and cells
 Colour key Minimum requirements as set out in Directive 2004/23/EC More stringent - legy binding on national level More stringent - recommended on national level Not legy binding and not recommended on
Colour key Minimum requirements as set out in Directive 2004/23/EC More stringent - legy binding on national level More stringent - recommended on national level Not legy binding and not recommended on
Hot Topics in Tissue Safety
 Hot Topics in Tissue Safety 37 th Annual AATB Meeting National Harbor, Maryland 03 October 2013 Scott A. Brubaker, CTBS Chief Policy Officer PHS Guideline for Reducing Human Immunodeficiency Virus, Hepatitis
Hot Topics in Tissue Safety 37 th Annual AATB Meeting National Harbor, Maryland 03 October 2013 Scott A. Brubaker, CTBS Chief Policy Officer PHS Guideline for Reducing Human Immunodeficiency Virus, Hepatitis
Hepatitis Case Investigation
 * indicates required fields Does patient also have: Hepatitis Case Investigation West Virginia Electronic Disease Surveillance System Division of Surveillance and Disease Control Infectious Disease Epidemiology
* indicates required fields Does patient also have: Hepatitis Case Investigation West Virginia Electronic Disease Surveillance System Division of Surveillance and Disease Control Infectious Disease Epidemiology
HEALTH SCREENING QUESTIONNAIRE CT Work-up. Donor ID EdgeCell #:
 HEALTH SCREENING QUESTIONNAIRE CT Work-up Héma-Québec Registre des Donneurs de Cellules Souches 4045 Côte-vertu, St-Laurent, QC, Canada, H4R 2W7 Tél : + 514-832-1031 Fax : + 514-832-0266 www.hema-quebec.qc.ca
HEALTH SCREENING QUESTIONNAIRE CT Work-up Héma-Québec Registre des Donneurs de Cellules Souches 4045 Côte-vertu, St-Laurent, QC, Canada, H4R 2W7 Tél : + 514-832-1031 Fax : + 514-832-0266 www.hema-quebec.qc.ca
Non-reproductive tissues and cells
 Ministry of Health: Institute for Transplantation and Biomedicine / Colour key VIRAL HIV 1 and HIV 2 Hepatitis B Minimum requirements as set out in Directive 2004/23/EC More stringent - legy binding More
Ministry of Health: Institute for Transplantation and Biomedicine / Colour key VIRAL HIV 1 and HIV 2 Hepatitis B Minimum requirements as set out in Directive 2004/23/EC More stringent - legy binding More
2017 CST-Astellas Canadian Transplant Fellows Symposium
 2017 CST-Astellas Canadian Transplant Fellows Symposium The Future of Transplantation Atul Humar, MD Atul Humar is a Professor in the Department of Medicine, University of Toronto. Dr. Humar received his
2017 CST-Astellas Canadian Transplant Fellows Symposium The Future of Transplantation Atul Humar, MD Atul Humar is a Professor in the Department of Medicine, University of Toronto. Dr. Humar received his
2017 CST-Astellas Canadian Transplant Fellows Symposium. Optimizing use of organs from Increased Risk Donors
 2017 CST-Astellas Canadian Transplant Fellows Symposium Optimizing use of organs from Increased Risk Donors Atual Humar, MD Atul Humar is a Professor in the Department of Medicine, University of Toronto.
2017 CST-Astellas Canadian Transplant Fellows Symposium Optimizing use of organs from Increased Risk Donors Atual Humar, MD Atul Humar is a Professor in the Department of Medicine, University of Toronto.
[Submitted Electronically]
![[Submitted Electronically] [Submitted Electronically]](/thumbs/82/85246007.jpg) [Submitted Electronically] Dr. Matthew J. Kuehnert, Director Office of Blood, Organ, and Other Tissue Safety Division of Healthcare Quality Promotion National Center for Emerging and Zoonotic Infectious
[Submitted Electronically] Dr. Matthew J. Kuehnert, Director Office of Blood, Organ, and Other Tissue Safety Division of Healthcare Quality Promotion National Center for Emerging and Zoonotic Infectious
Non-reproductive tissues and cells
 Colour key Tested pathogen VIRAL Minimum requirements as set out in Directive 2004/23/EC More stringent testing - legy binding on national level More stringent testing - recommended on national level Not
Colour key Tested pathogen VIRAL Minimum requirements as set out in Directive 2004/23/EC More stringent testing - legy binding on national level More stringent testing - recommended on national level Not
HEPATITIS C UPDATES. Sanaa S. Said 10 th April, 2014
 HEPATITIS C UPDATES Sanaa S. Said 10 th April, 2014 CONTENTS Introduction Epidemiology Transmission and Natural history Kenyan guidelines What is new? References INTRODUCTION Hepacivirus genus, Flaviviridae
HEPATITIS C UPDATES Sanaa S. Said 10 th April, 2014 CONTENTS Introduction Epidemiology Transmission and Natural history Kenyan guidelines What is new? References INTRODUCTION Hepacivirus genus, Flaviviridae
Potential etiologies of infection in these patients are diverse, including common and uncommon opportunistic infections.
 In the name of God Principles of post Tx infections 1: Potential etiologies of infection in these patients are diverse, including common and uncommon opportunistic infections. Infection processes can progress
In the name of God Principles of post Tx infections 1: Potential etiologies of infection in these patients are diverse, including common and uncommon opportunistic infections. Infection processes can progress
Non-reproductive tissues and cells Recommending authority/ association
 Colour key Minimum requirements as set out in Directive 2004/23/EC More stringent - legally binding More stringent - recommended Not legally binding and not recommended Non-reproductive tissues and cells
Colour key Minimum requirements as set out in Directive 2004/23/EC More stringent - legally binding More stringent - recommended Not legally binding and not recommended Non-reproductive tissues and cells
Non-reproductive tissues and cells
 Colour key Minimum requirements as set out in Directive 2004/23/EC More stringent testing - legally binding on national level More stringent testing - recommended on national level Not legally binding
Colour key Minimum requirements as set out in Directive 2004/23/EC More stringent testing - legally binding on national level More stringent testing - recommended on national level Not legally binding
Viral hepatitis. Supervised by: Dr.Gaith. presented by: Shaima a & Anas & Ala a
 Viral hepatitis Supervised by: Dr.Gaith presented by: Shaima a & Anas & Ala a Etiology Common: Hepatitis A Hepatitis B Hepatitis C Hepatitis D Hepatitis E Less common: Cytomegalovirus EBV Rare: Herpes
Viral hepatitis Supervised by: Dr.Gaith presented by: Shaima a & Anas & Ala a Etiology Common: Hepatitis A Hepatitis B Hepatitis C Hepatitis D Hepatitis E Less common: Cytomegalovirus EBV Rare: Herpes
Surveillance Report 2014
 Surveillance Report 2014 Executive summary We are pleased to present the third report for our stakeholders describing infectious disease surveillance. High quality and timely surveillance is key to the
Surveillance Report 2014 Executive summary We are pleased to present the third report for our stakeholders describing infectious disease surveillance. High quality and timely surveillance is key to the
Hepatitis C Virus (HCV)
 Clinical Practice Guidelines Hepatitis C Virus (HCV) OBJECTIVE The purpose is to guide the appropriate diagnosis and management of Hepatitis C Virus (HCV). GUIDELINE These are only guidelines, and are
Clinical Practice Guidelines Hepatitis C Virus (HCV) OBJECTIVE The purpose is to guide the appropriate diagnosis and management of Hepatitis C Virus (HCV). GUIDELINE These are only guidelines, and are
Infectious Disease Considerations in Organ Donation. Nikole A Neidlinger MD FACS Chief Medical Officer
 Infectious Disease Considerations in Organ Donation Nikole A Neidlinger MD FACS Chief Medical Officer Screening Donors and Infectious Disease considerations: Deceased Donor Screening Donors at increased
Infectious Disease Considerations in Organ Donation Nikole A Neidlinger MD FACS Chief Medical Officer Screening Donors and Infectious Disease considerations: Deceased Donor Screening Donors at increased
Management of Acute HCV Infection
 Management of Acute HCV Infection This section provides guidance on the diagnosis and medical management of acute HCV infection, which is defined as presenting within 6 months of the exposure. During this
Management of Acute HCV Infection This section provides guidance on the diagnosis and medical management of acute HCV infection, which is defined as presenting within 6 months of the exposure. During this
The Alphabet Soup of Viral Hepatitis Testing
 The Alphabet Soup of Viral Hepatitis Testing August 18, 2011 Patricia Slev, PhD, DABCC Medical Director, Serologic Hepatitis and Retrovirus Laboratory, ARUP Laboratories Assistant Professor of Pathology,
The Alphabet Soup of Viral Hepatitis Testing August 18, 2011 Patricia Slev, PhD, DABCC Medical Director, Serologic Hepatitis and Retrovirus Laboratory, ARUP Laboratories Assistant Professor of Pathology,
Confirmed (Laboratory Tests) Serum positive for IgM anti-hbc or, hepatitis B surface antigen (HbsAg).
 Hepatitis B Hepatitis B is a liver disease that results from infection with the Hepatitis B virus. It can range in severity from a mild illness lasting a few weeks to a serious, lifelong illness. Hepatitis
Hepatitis B Hepatitis B is a liver disease that results from infection with the Hepatitis B virus. It can range in severity from a mild illness lasting a few weeks to a serious, lifelong illness. Hepatitis
HEPATITIS C, ACUTE CRUDE DATA. Number of Cases 5 Annual Incidence a LA County 0.05 California b 0.10 United States b 0.68 Age at Diagnosis Mean 38
 2016 Annual Morbidity Report HEPATITIS C, ACUTE a Rates calculated based on less than 19 cases or events are considered unreliable b Calculated from: CDC. Notice to Readers: Final 2016 Reports of Nationally
2016 Annual Morbidity Report HEPATITIS C, ACUTE a Rates calculated based on less than 19 cases or events are considered unreliable b Calculated from: CDC. Notice to Readers: Final 2016 Reports of Nationally
BVHG/BASL/BSG/BHIVA/BIA/CVN Guidelines for management of chronic HCV infection
 BVHG/BASL/BSG/BHIVA/BIA/CVN Guidelines for management of chronic HCV infection Headline Recommendations 1. We recommend that NHSE considers commissioning pan-genotypic regimens for use in the community
BVHG/BASL/BSG/BHIVA/BIA/CVN Guidelines for management of chronic HCV infection Headline Recommendations 1. We recommend that NHSE considers commissioning pan-genotypic regimens for use in the community
The testing of Donated Blood and Components at NHSBT
 The testing of Donated Blood and Components at NHSBT NHSBT is responsible for collecting all donated blood and platelets in England, then processing into components before issuing to client hospitals.
The testing of Donated Blood and Components at NHSBT NHSBT is responsible for collecting all donated blood and platelets in England, then processing into components before issuing to client hospitals.
Update on Real-World Experience With HARVONI
 Update on Real-World Experience With A RESOURCE FOR PAYERS MAY 217 This information is intended for payers only. The HCV-TARGET study was supported by Gilead Sciences, Inc. Real-world experience data were
Update on Real-World Experience With A RESOURCE FOR PAYERS MAY 217 This information is intended for payers only. The HCV-TARGET study was supported by Gilead Sciences, Inc. Real-world experience data were
HCV Infection: EASL Clinical Practice Guidelines Francesco Negro University Hospital Geneva Switzerland
 HCV Infection: EASL Clinical Practice Guidelines 2016 Francesco Negro University Hospital Geneva Switzerland Panel Codinat: Jean-Michel Pawlotsky Panel: Alessio Aghemo David Back Geoffrey Dusheiko Xavier
HCV Infection: EASL Clinical Practice Guidelines 2016 Francesco Negro University Hospital Geneva Switzerland Panel Codinat: Jean-Michel Pawlotsky Panel: Alessio Aghemo David Back Geoffrey Dusheiko Xavier
Arvind R. Murali, MD Assistant Professor of Medicine Gastroenterology & Hepatology Organ Transplant Center UIHC, Carver College of Medicine
 Arvind R. Murali, MD Assistant Professor of Medicine Gastroenterology & Hepatology Organ Transplant Center UIHC, Carver College of Medicine No financial disclosures No conflicts of interest No affiliations
Arvind R. Murali, MD Assistant Professor of Medicine Gastroenterology & Hepatology Organ Transplant Center UIHC, Carver College of Medicine No financial disclosures No conflicts of interest No affiliations
Hepatitis C Infection: Updated Information for Front Line Workers in Primary Care Settings MAMTA K. JAIN, MD, MPH 2/14/18
 Hepatitis C Infection: Updated Information for Front Line Workers in Primary Care Settings MAMTA K. JAIN, MD, MPH 2/14/18 Overview Hepatitis C Virus Prevalence Effects of Hepatitis C Prevention Diagnosis
Hepatitis C Infection: Updated Information for Front Line Workers in Primary Care Settings MAMTA K. JAIN, MD, MPH 2/14/18 Overview Hepatitis C Virus Prevalence Effects of Hepatitis C Prevention Diagnosis
Emerging Infections in Solid Organ Transplantation
 Emerging Infections in Solid Organ Transplantation Sherif R. Zaki, MD, PhD Chief, Infectious Diseases Pathology Branch Division of High-consequence Pathogens and Pathology National Center of Emerging and
Emerging Infections in Solid Organ Transplantation Sherif R. Zaki, MD, PhD Chief, Infectious Diseases Pathology Branch Division of High-consequence Pathogens and Pathology National Center of Emerging and
Viral Hepatitis. Dr Melissa Haines Gastroenterologist Waikato Hospital
 Viral Hepatitis Dr Melissa Haines Gastroenterologist Waikato Hospital Viral Hepatitis HAV HBV HCV HDV HEV Other viral: CMV, EBV, HSV Unknown Hepatitis A Hepatitis A Transmitted via the faecal-oral route
Viral Hepatitis Dr Melissa Haines Gastroenterologist Waikato Hospital Viral Hepatitis HAV HBV HCV HDV HEV Other viral: CMV, EBV, HSV Unknown Hepatitis A Hepatitis A Transmitted via the faecal-oral route
DHQ Flowcharts v2.0 eff. February 2016
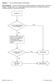 Question: 1. Are you feeling healthy and well today? Donor Eligibility: A person should be free of infectious diseases, including colds, on the day of donation. A person who is not in good health should
Question: 1. Are you feeling healthy and well today? Donor Eligibility: A person should be free of infectious diseases, including colds, on the day of donation. A person who is not in good health should
Update on Hepatitis B and Hepatitis C
 Update on Hepatitis B and Hepatitis C Catherine Stedman Department of Gastroenterology, Christchurch Hospital and University of Otago, Christchurch Disclosures I have the following financial relationships
Update on Hepatitis B and Hepatitis C Catherine Stedman Department of Gastroenterology, Christchurch Hospital and University of Otago, Christchurch Disclosures I have the following financial relationships
Non-reproductive tissues and cells
 Colour key Minimum requirements as set out in Directive 2004/23/EC and its technical Directives (particularly 2006/17/EC) More stringent -legally binding, applies for all donations and all donor profiles
Colour key Minimum requirements as set out in Directive 2004/23/EC and its technical Directives (particularly 2006/17/EC) More stringent -legally binding, applies for all donations and all donor profiles
The ABCs of Viral Hepatitis Diagnosis. Ila Singh, M.D., Ph.D. P & S Viral Hepatitis. Hepatitis A, B, C, D, E and G viruses
 The ABCs of Viral Hepatitis Diagnosis Ila Singh, M.D., Ph.D. P & S 14-453 is132@columbia.edu Viral Hepatitis Hepatotropic viruses Hepatitis A, B, C, D, E and G viruses Generalized infection plus infection
The ABCs of Viral Hepatitis Diagnosis Ila Singh, M.D., Ph.D. P & S 14-453 is132@columbia.edu Viral Hepatitis Hepatotropic viruses Hepatitis A, B, C, D, E and G viruses Generalized infection plus infection
Learning Objectives: Hepatitis Update. Primary Causes of Chronic Liver Disease in the U.S. Hepatitis Definition. Hepatitis Viruses.
 Learning Objectives: Hepatitis Update ASCLS-Michigan March 31, 2016 Dr. Kathleen Hoag Upon attendance of this seminar and review of material provided, the attendees will be able to: 1. List hepatitis viruses
Learning Objectives: Hepatitis Update ASCLS-Michigan March 31, 2016 Dr. Kathleen Hoag Upon attendance of this seminar and review of material provided, the attendees will be able to: 1. List hepatitis viruses
risk of Transfusion-Transmitted Transmitted Viral Infections (HIV, HCV and HBV)
 Prevention and ResidualR risk of Transfusion-Transmitted Transmitted Viral Infections (HIV, HCV and HBV) Syria Laperche L (Centre National de référence r rence pour les hépatites h B et C en Transfusion)
Prevention and ResidualR risk of Transfusion-Transmitted Transmitted Viral Infections (HIV, HCV and HBV) Syria Laperche L (Centre National de référence r rence pour les hépatites h B et C en Transfusion)
Question: 1. Are you currently taking an antibiotic?
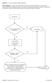 Question: 1. Are you currently taking an antibiotic? Donor Eligibility: A donor with an infection that could cause bacteremia/sepsis or other relevant communicable disease should not donate. The reason
Question: 1. Are you currently taking an antibiotic? Donor Eligibility: A donor with an infection that could cause bacteremia/sepsis or other relevant communicable disease should not donate. The reason
Hepatitis C Update on New Treatments
 Hepatitis C Update on New Treatments Kevork M. Peltekian, MD, FRCPC 44th Annual Dalhousie Spring Refresher Course - Therapeutics April 5 - April 7, 2018 Halifax Convention Centre Disclosures Conflicts
Hepatitis C Update on New Treatments Kevork M. Peltekian, MD, FRCPC 44th Annual Dalhousie Spring Refresher Course - Therapeutics April 5 - April 7, 2018 Halifax Convention Centre Disclosures Conflicts
Viral hepatitis Blood Born hepatitis. Dr. MONA BADR Assistant Professor College of Medicine & KKUH
 Viral hepatitis Blood Born hepatitis Dr. MONA BADR Assistant Professor College of Medicine & KKUH Outline Introduction to hepatitis Characteristics of viral hepatitis Mode of transmission Markers of hepatitis
Viral hepatitis Blood Born hepatitis Dr. MONA BADR Assistant Professor College of Medicine & KKUH Outline Introduction to hepatitis Characteristics of viral hepatitis Mode of transmission Markers of hepatitis
Hepatitis C Update: Screening, Diagnosis, and Treatment
 Mountain West AIDS Education and Training Center Hepatitis C Update: Screening, Diagnosis, and Treatment Brian R. Wood, MD (bwood2@uw.edu) Assistant Professor of Medicine, University of Washington Medical
Mountain West AIDS Education and Training Center Hepatitis C Update: Screening, Diagnosis, and Treatment Brian R. Wood, MD (bwood2@uw.edu) Assistant Professor of Medicine, University of Washington Medical
Question: 1. Are you currently taking an antibiotic?
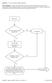 Question: 1. Are you currently taking an antibiotic? Donor Eligibility: A donor with an infection that could cause bacteremia/sepsis or other relevant communicable disease should not donate. The reason
Question: 1. Are you currently taking an antibiotic? Donor Eligibility: A donor with an infection that could cause bacteremia/sepsis or other relevant communicable disease should not donate. The reason
Non-reproductive tissues and cells Recommending authority/ association
 Colour key Minimum requirements as set out in Directive 2004/23/EC More stringent testing - legy binding on national level More stringent testing - recomd on national level Not legy binding and not recomd
Colour key Minimum requirements as set out in Directive 2004/23/EC More stringent testing - legy binding on national level More stringent testing - recomd on national level Not legy binding and not recomd
Epidemiology of Hepatitis C Iran
 Seyed Moayed Alavian M.D. Professor of Gastro & Hepatology Editor-in-chief of Glob Hepat Comm Editor-in-chief of Hepatitis Monthly E mail: editor@hepatmon.com Epidemiology of Hepatitis C Iran Epidemiology
Seyed Moayed Alavian M.D. Professor of Gastro & Hepatology Editor-in-chief of Glob Hepat Comm Editor-in-chief of Hepatitis Monthly E mail: editor@hepatmon.com Epidemiology of Hepatitis C Iran Epidemiology
The Technical Aspects of Pathogen Testing in Canada. Nancy Angus Canadian Blood Services Director, Testing
 PERMISSON TO USE: Please note that, by making their presentations available on-line, primary authors have agreed to share their presentations. However, should you want to use some of the data or slides
PERMISSON TO USE: Please note that, by making their presentations available on-line, primary authors have agreed to share their presentations. However, should you want to use some of the data or slides
The Changing World of Hepatitis C
 The Changing World of Hepatitis C Alnoor Ramji Gastroenterology & Hepatology Clinical Associate Professor Division of Gastroenterology University Of British Columbia St. Paul s Hospital Site Disclosures
The Changing World of Hepatitis C Alnoor Ramji Gastroenterology & Hepatology Clinical Associate Professor Division of Gastroenterology University Of British Columbia St. Paul s Hospital Site Disclosures
The Hepatitis B-e antigen-positive
 The Hepatitis B-e antigen-positive Dental Student Developing an Equitable Policy Hepatitis B Virus Serology 1 HBsAg A protein on the surface of HBV; it can be detected in high levels in serum during acute
The Hepatitis B-e antigen-positive Dental Student Developing an Equitable Policy Hepatitis B Virus Serology 1 HBsAg A protein on the surface of HBV; it can be detected in high levels in serum during acute
Hepatitis C Best Practice Guidelines For Local Health Departments
 Hepatitis C Best Practice Guidelines For Local Health Departments LHDs are responsible for investigating and reporting all physician reported cases of acute hepatitis C (HCV). For clients known to have
Hepatitis C Best Practice Guidelines For Local Health Departments LHDs are responsible for investigating and reporting all physician reported cases of acute hepatitis C (HCV). For clients known to have
HBV, HCV, HIV and Kidney Transplantation. Simin Goral MD University of Pennsylvania Medical Center Philadelphia, Pennsylvania
 HBV, HCV, HIV and Kidney Transplantation Simin Goral MD University of Pennsylvania Medical Center Philadelphia, Pennsylvania Objectives Prevalence of hepatitis B (HBV), hepatitis C (HCV) and human immunodeficiency
HBV, HCV, HIV and Kidney Transplantation Simin Goral MD University of Pennsylvania Medical Center Philadelphia, Pennsylvania Objectives Prevalence of hepatitis B (HBV), hepatitis C (HCV) and human immunodeficiency
CORD BLOOD TRANSPLANTATION STUDY CBU EXCLUSION AND QUARANTINE RELEASE FORM
 (Any answer of Yes indicates exclusion) 1. Informed Consent and Maternal/Infant Demographics a. Lack of confirmation of signed and witnessed informed consent 1 Yes 2 No Is the following information missing
(Any answer of Yes indicates exclusion) 1. Informed Consent and Maternal/Infant Demographics a. Lack of confirmation of signed and witnessed informed consent 1 Yes 2 No Is the following information missing
Reporting from Council of Europe member states on the collection, testing and use of blood and blood components in Europe The 2006 Survey
 Reporting from Council of Europe member states on the collection, testing and use of blood and blood components in Europe The 2006 Survey This questionnaire consists of three sections: A. Collection and
Reporting from Council of Europe member states on the collection, testing and use of blood and blood components in Europe The 2006 Survey This questionnaire consists of three sections: A. Collection and
Commonly Asked Questions About Chronic Hepatitis C
 Commonly Asked Questions About Chronic Hepatitis C From the American College of Gastroenterology 1. How common is the hepatitis C virus? The hepatitis C virus is the most common cause of chronic viral
Commonly Asked Questions About Chronic Hepatitis C From the American College of Gastroenterology 1. How common is the hepatitis C virus? The hepatitis C virus is the most common cause of chronic viral
@PremierHA #AdvisorLive. Download today s slides at
 @PremierHA #AdvisorLive Download today s slides at www.premierinc.com/events Logistics Audio Use your computer speakers or dial in with the number on your screen Notes Download today s slides from the
@PremierHA #AdvisorLive Download today s slides at www.premierinc.com/events Logistics Audio Use your computer speakers or dial in with the number on your screen Notes Download today s slides from the
Worldwide Causes of HCC
 Approach to HCV Treatment in Patients with HCC JORGE L. HERRERA, MD, MACG UNIVERSITY OF SOUTH ALABAMA COLLEGE OF MEDICINE Worldwide Causes of HCC 60% 50% 40% 54% 30% 20% 10% 31% 15% 0% Hepatitis B Hepatitis
Approach to HCV Treatment in Patients with HCC JORGE L. HERRERA, MD, MACG UNIVERSITY OF SOUTH ALABAMA COLLEGE OF MEDICINE Worldwide Causes of HCC 60% 50% 40% 54% 30% 20% 10% 31% 15% 0% Hepatitis B Hepatitis
Hepatitis C epidemiology, screening and treatment
 Hepatitis C epidemiology, screening and treatment Date Presenter Wednesday 29 August 2018: 7.00-8.00pm Professor Greg Dore This education activity has been developed in association with: Kirby Institute,
Hepatitis C epidemiology, screening and treatment Date Presenter Wednesday 29 August 2018: 7.00-8.00pm Professor Greg Dore This education activity has been developed in association with: Kirby Institute,
Hepatitis C Management and Treatment
 Hepatitis C Management and Treatment Kaya Süer Near East University Faculty of Medicine Infectious Diseases and Clinical Microbiology 1 Discovery of Hepatitis C Key facts Hepatitis C: the virus can cause
Hepatitis C Management and Treatment Kaya Süer Near East University Faculty of Medicine Infectious Diseases and Clinical Microbiology 1 Discovery of Hepatitis C Key facts Hepatitis C: the virus can cause
Non-reproductive tissues and cells Recommending authority/ association
 Colour key Minimum requirements as set out in Directive 2004/23/EC More stringent - legy binding More stringent - recommended Not legy binding and not recommended Non-reproductive tissues and cells VIRAL
Colour key Minimum requirements as set out in Directive 2004/23/EC More stringent - legy binding More stringent - recommended Not legy binding and not recommended Non-reproductive tissues and cells VIRAL
HEPATITIS C: UPDATE AND MANAGEMENT
 HEPATITIS C: UPDATE AND MANAGEMENT José Franco, MD Professor of Medicine Associate Dean for Educational Improvement Associate Director, Kern Institute STAR Center Director José Franco, MD Disclosures I
HEPATITIS C: UPDATE AND MANAGEMENT José Franco, MD Professor of Medicine Associate Dean for Educational Improvement Associate Director, Kern Institute STAR Center Director José Franco, MD Disclosures I
Viral Hepatitis Diagnosis and Management
 Viral Hepatitis Diagnosis and Management CLINICAL BACKGROUND Viral hepatitis is a relatively common disease (25 per 100,000 individuals in the United States) caused by a diverse group of hepatotropic agents
Viral Hepatitis Diagnosis and Management CLINICAL BACKGROUND Viral hepatitis is a relatively common disease (25 per 100,000 individuals in the United States) caused by a diverse group of hepatotropic agents
Hepatitis B and Hepatitis C Virus in non-liver Transplant Recipients. Karim Qumosani MD, FRCPC, ABIM, MdMEd Multi-organ Transplant Unit, London
 Hepatitis B and Hepatitis C Virus in non-liver Transplant Recipients Karim Qumosani MD, FRCPC, ABIM, MdMEd Multi-organ Transplant Unit, London Financial Disclosures Research Grants Merck, Gilead, Abbvie,
Hepatitis B and Hepatitis C Virus in non-liver Transplant Recipients Karim Qumosani MD, FRCPC, ABIM, MdMEd Multi-organ Transplant Unit, London Financial Disclosures Research Grants Merck, Gilead, Abbvie,
FORM TITLE: Risk Behavior Assessment. 1. Are you a male who has had sex with another male in the preceding five years?
 RELESE DTE: 12/07/2012 EFFECTIVE DTE: 12/13/2012 1. re you a male who has had sex with another male in the preceding five years? 2. Have you injected drugs for non-medical reason in the preceding five
RELESE DTE: 12/07/2012 EFFECTIVE DTE: 12/13/2012 1. re you a male who has had sex with another male in the preceding five years? 2. Have you injected drugs for non-medical reason in the preceding five
Viral Hepatitis: Dr Erana Gray General and Infectious Diseases Physician
 Viral Hepatitis: Dr Erana Gray General and Infectious Diseases Physician Outline: Virology HBV + pregnancy HCV + new treatments Cases Hepatitis Foundation HBV: virus HBsAg far in excess of complete virions,
Viral Hepatitis: Dr Erana Gray General and Infectious Diseases Physician Outline: Virology HBV + pregnancy HCV + new treatments Cases Hepatitis Foundation HBV: virus HBsAg far in excess of complete virions,
Structure and duties of Blood service
 NATIONAL CENTER FOR TRANSFUSION MEDICINE Prevalence of infectious diseases among Mongolian blood donors Namjil Erdenebayar General director of The National Center for Transfusion Medicine, Mongolia NCTM,
NATIONAL CENTER FOR TRANSFUSION MEDICINE Prevalence of infectious diseases among Mongolian blood donors Namjil Erdenebayar General director of The National Center for Transfusion Medicine, Mongolia NCTM,
Tough Cases in HIV/HCV Coinfection
 NORTHWEST AIDS EDUCATION AND TRAINING CENTER Tough Cases in HIV/HCV Coinfection John Scott, MD, MSc Assistant Professor University of Washington Presentation prepared by: J Scott Last Updated: Jun 5, 2014
NORTHWEST AIDS EDUCATION AND TRAINING CENTER Tough Cases in HIV/HCV Coinfection John Scott, MD, MSc Assistant Professor University of Washington Presentation prepared by: J Scott Last Updated: Jun 5, 2014
HIV/hepatitis co-infection. Christoph Boesecke Department of Medicine I University Hospital Bonn Germany
 HIV/hepatitis co-infection Christoph Boesecke Department of Medicine I University Hospital Bonn Germany Clinical Management and Treatment of HBV and HCV Co-infection in HIVpositive Persons Hepatitis B
HIV/hepatitis co-infection Christoph Boesecke Department of Medicine I University Hospital Bonn Germany Clinical Management and Treatment of HBV and HCV Co-infection in HIVpositive Persons Hepatitis B
Taken From VBA s Adjudication Procedure Manual Section on Hepatitis
 Taken From VBA s Adjudication Procedure Manual Section on Hepatitis M21-1, Part III, Subpart iv, 4.I.2 Updated January 11, 2017 2. Hepatitis and Other Liver Disabilities Introduction Change Date January
Taken From VBA s Adjudication Procedure Manual Section on Hepatitis M21-1, Part III, Subpart iv, 4.I.2 Updated January 11, 2017 2. Hepatitis and Other Liver Disabilities Introduction Change Date January
Ari Bunim, M.D. Director of Hepatology New York Hospital Queens Assistant Professor of Clinical Medicine Weill Cornell Medical College
 Ari Bunim, M.D. Director of Hepatology New York Hospital Queens Assistant Professor of Clinical Medicine Weill Cornell Medical College New York State Law Goes into Effect January 1, 2014 Hepatitis C Virus
Ari Bunim, M.D. Director of Hepatology New York Hospital Queens Assistant Professor of Clinical Medicine Weill Cornell Medical College New York State Law Goes into Effect January 1, 2014 Hepatitis C Virus
Hepatitis C. Core slides
 Hepatitis C Core slides This material was prepared by the Viral Hepatitis Prevention Board The slides (or subsets) can be reproduced for educational use only, with reference to the original source and
Hepatitis C Core slides This material was prepared by the Viral Hepatitis Prevention Board The slides (or subsets) can be reproduced for educational use only, with reference to the original source and
Only one take home point for the talk 9/26/2018. Infectious Diseases and Donor Derived Infections. Don t forget about donor-derived infections
 Shane Colombo 1993 2018 Infectious Diseases and Donor Derived Infections Peter Chin-Hong, MD Division of Infectious Diseases UCSF Only one take home point for the talk Don t forget about donor-derived
Shane Colombo 1993 2018 Infectious Diseases and Donor Derived Infections Peter Chin-Hong, MD Division of Infectious Diseases UCSF Only one take home point for the talk Don t forget about donor-derived
HIV Basics: Clinical Tests and Guidelines
 HIV Basics: Clinical Tests and Guidelines ACTHIV 2010 Zelalem Temesgen MD Mayo Clinic Topics Baseline laboratory evaluation Laboratory monitoring through the continuum of care Patients not on antiretroviral
HIV Basics: Clinical Tests and Guidelines ACTHIV 2010 Zelalem Temesgen MD Mayo Clinic Topics Baseline laboratory evaluation Laboratory monitoring through the continuum of care Patients not on antiretroviral
ACTIVITY DISCLAIMER. Kurt Cook, MD, MSc DISCLOSURE. Audience Engagement System. Learning Objectives
 On the Liver Disease Front Lines: Hepatitis A, B and C Prevention and Treatment Kurt Cook, MD, MSc ACTIVITY DISCLAIMER The material presented here is being made available by the American Academy of Family
On the Liver Disease Front Lines: Hepatitis A, B and C Prevention and Treatment Kurt Cook, MD, MSc ACTIVITY DISCLAIMER The material presented here is being made available by the American Academy of Family
Surveillance Report 2015
 Surveillance Report 2015 Executive summary We are pleased to present the fourth report for our stakeholders describing infectious disease surveillance. High quality and timely surveillance is key to the
Surveillance Report 2015 Executive summary We are pleased to present the fourth report for our stakeholders describing infectious disease surveillance. High quality and timely surveillance is key to the
Hepatitis C Elimination: Screening, Linkage and Treatment. Eric Lawitz, MD The Texas Liver Institute San Antonio, Texas
 Hepatitis C Elimination: Screening, Linkage and Treatment Eric Lawitz, MD The Texas Liver Institute San Antonio, Texas Hepatitis C: Worldwide Presence Worldwide prevalence: 130-150 million Viral hepatitis
Hepatitis C Elimination: Screening, Linkage and Treatment Eric Lawitz, MD The Texas Liver Institute San Antonio, Texas Hepatitis C: Worldwide Presence Worldwide prevalence: 130-150 million Viral hepatitis
Next generation DAAs: Combining efficacy and safety profile. Jiannis Vlachogiannakos
 Next generation DAAs: Combining efficacy and safety profile. Jiannis Vlachogiannakos Associate Professor of Gastroenterology, Academic Department of Gastroenterology, National and Kapodistrian University
Next generation DAAs: Combining efficacy and safety profile. Jiannis Vlachogiannakos Associate Professor of Gastroenterology, Academic Department of Gastroenterology, National and Kapodistrian University
Why make this statement?
 HCV Council 2014 10 clinical practice statements were evaluated by the Council A review of the available literature was conducted The level of support and level of evidence for the statements were discussed
HCV Council 2014 10 clinical practice statements were evaluated by the Council A review of the available literature was conducted The level of support and level of evidence for the statements were discussed
Achieving Consensus on Increased Risk Donors to Improve Access to Organ Transplantation Executive Summary
 Achieving Consensus on Increased Risk Donors to Improve Access to Organ Transplantation Executive Summary Introduction To address the current shortage of organs for transplantation, (114,651 candidates
Achieving Consensus on Increased Risk Donors to Improve Access to Organ Transplantation Executive Summary Introduction To address the current shortage of organs for transplantation, (114,651 candidates
Hepatitis C in Correctional Facilities: Big Problem, Bigger Opportunity. Cody A. Chastain, MD
 Hepatitis C in Correctional Facilities: Big Problem, Bigger Opportunity Cody A. Chastain, MD Disclosures Research supported by Gilead Sciences Inc.: Site investigator for HIV/HCV SWITCH Registry Study
Hepatitis C in Correctional Facilities: Big Problem, Bigger Opportunity Cody A. Chastain, MD Disclosures Research supported by Gilead Sciences Inc.: Site investigator for HIV/HCV SWITCH Registry Study
Exploring the risks of liver cancer after successful treatment for hepatitis C virus
 CATIE-News CATIE s bite-sized HIV and hepatitis C news bulletins. Exploring the risks of liver cancer after successful treatment for hepatitis C virus 11 June 2013 In Canada and other high-income countries,
CATIE-News CATIE s bite-sized HIV and hepatitis C news bulletins. Exploring the risks of liver cancer after successful treatment for hepatitis C virus 11 June 2013 In Canada and other high-income countries,
Monitoring Patients Who Are Starting HCV Treatment, Are On Treatment, Or Have Completed Therapy
 Monitoring Patients Who Are Starting HCV Treatment, Are On Treatment, Or Have Completed Therapy WV ECHO August 10, 2017 Selection of patients for HCV treatment Despite current guidance to treat everyone,
Monitoring Patients Who Are Starting HCV Treatment, Are On Treatment, Or Have Completed Therapy WV ECHO August 10, 2017 Selection of patients for HCV treatment Despite current guidance to treat everyone,
Update in the Management of Hepatitis C: What Does the Future Hold
 Update in the Management of Hepatitis C: What Does the Future Hold Paul Y Kwo, MD, FACG Professor of Medicine Mdi Medical ldirector, Liver Transplantation tti Gastroenterology/Hepatology Division Indiana
Update in the Management of Hepatitis C: What Does the Future Hold Paul Y Kwo, MD, FACG Professor of Medicine Mdi Medical ldirector, Liver Transplantation tti Gastroenterology/Hepatology Division Indiana
Keeping the Blood Supply Safe Current and Future Strategies. Marissa Li, MD
 Keeping the Blood Supply Safe Current and Future Strategies Marissa Li, MD Overview Pre-collection Donor Screening Collection Visual Inspection Aseptic Processing Leukoreduction of Apheresis Collections
Keeping the Blood Supply Safe Current and Future Strategies Marissa Li, MD Overview Pre-collection Donor Screening Collection Visual Inspection Aseptic Processing Leukoreduction of Apheresis Collections
EPIDEMIOLOGY, CLINICAL FEATURES AND OUTCOME OF ACUTE HEPATITIS C IN HIV-POSITIVE PATIENTS: PRESENTATION OF OUR EXPERIENCE
 EPIDEMIOLOGY, CLINICAL FEATURES AND OUTCOME OF ACUTE HEPATITIS C IN HIV-POSITIVE PATIENTS: PRESENTATION OF OUR EXPERIENCE E. Angeli, A. Mainini, C. Atzori, G. Gubertini and G. Rizzardini II Dept. Infectious
EPIDEMIOLOGY, CLINICAL FEATURES AND OUTCOME OF ACUTE HEPATITIS C IN HIV-POSITIVE PATIENTS: PRESENTATION OF OUR EXPERIENCE E. Angeli, A. Mainini, C. Atzori, G. Gubertini and G. Rizzardini II Dept. Infectious
Phase 3. Treatment Experienced. Ledipasvir-Sofosbuvir +/- Ribavirin in HCV Genotype 1 ION-2. Afdhal N, et al. N Engl J Med. 2014;370:
 Phase 3 Treatment Experienced Ledipasvir-Sofosbuvir +/- Ribavirin in HCV Genotype 1 ION-2 Afdhal N, et al. N Engl J Med. 2014;370:1483-93. Ledipasvir-Sofosbuvir +/- Ribavirin in Treatment-Experienced HCV
Phase 3 Treatment Experienced Ledipasvir-Sofosbuvir +/- Ribavirin in HCV Genotype 1 ION-2 Afdhal N, et al. N Engl J Med. 2014;370:1483-93. Ledipasvir-Sofosbuvir +/- Ribavirin in Treatment-Experienced HCV
Factsheet: New Treatments for hepatitis C Direct Acting Antivirals (DAAs)
 Factsheet: New Treatments for hepatitis C Direct Acting Antivirals (DAAs) For more information about anything in this factsheet, phone the Hepatitis Infoline on 1800 703 003 or go to www.hepvic.org.au
Factsheet: New Treatments for hepatitis C Direct Acting Antivirals (DAAs) For more information about anything in this factsheet, phone the Hepatitis Infoline on 1800 703 003 or go to www.hepvic.org.au
Is Treatment cost effective HCV and Organ Transplantation
 Is Treatment cost effective HCV and Organ Transplantation Dr Kosh Agarwal Institute of Liver Studies King s College Hospital Barcelona 2016 Disclosures: BoJo Pharma support: AbbVie/Achillion/ Astellas/
Is Treatment cost effective HCV and Organ Transplantation Dr Kosh Agarwal Institute of Liver Studies King s College Hospital Barcelona 2016 Disclosures: BoJo Pharma support: AbbVie/Achillion/ Astellas/
Hepatitis STARS Program. Geri Brown, M.D. Associate Professor Department of Internal Medicine October 4, 2003
 Hepatitis 2003 STARS Program Geri Brown, M.D. Associate Professor Department of Internal Medicine October 4, 2003 Outline n Hepatitis A Epidemiology and screening Transmission n Hepatitis B Epidemiology
Hepatitis 2003 STARS Program Geri Brown, M.D. Associate Professor Department of Internal Medicine October 4, 2003 Outline n Hepatitis A Epidemiology and screening Transmission n Hepatitis B Epidemiology
