COST-EFFECTIVENESS OF TREATMENT FOR MODERATE-TO-SEVERE PSORIASIS
|
|
|
- Duane Jones
- 5 years ago
- Views:
Transcription
1 COST-EFFECTIVENESS OF TREATMENT FOR MODERATE-TO-SEVERE PSORIASIS Cheryl S. Hankin PhD 1 ; Steven R. Feldman, MD, PhD 2 ; Daniel Pearce, MD 2 1 BioMedEcon, San Jose, CA, USA; 2 Wake Forest University School of Medicine, Winston-Salem, NC, USA Funded by Connetics Corporation
2 OVERVIEW Definition and Prevalence Treatment Cost-Effectiveness Study Background Objective Methods Findings Conclusion Discussion
3 DEFINITION Chronic polygenic disorder that results from predisposing & triggering factors (e.g., trauma, infection) Epidermal proliferation and differentiation Sharply demarcated, scaly erythematous plaques with silvery white scales During exacerbations, lesions often itch
4 PREVALENCE of MODERATE-TO-SEVERE PSORIASIS Affects up to 30% of patients Generally involves > 2% body surface area Disabling Location (face, hands, fingernails, feet, genitals) Symptoms (pain, tightness, bleeding, severe itching)
5 TREATMENT Oral Systemics acitretin (Soriatane - Connetics) cyclosporine (Neoral - Novartis) methotrexate (Trexall - Barr) Biologics alefacept (Amevive -Biogen) efalizumab (Raptiva - Genentech/Xoma) etanercept (Enbrel - Amgen) Phototherapy Ultraviolet B (UVB) Psoralen with Ultraviolet A (PUVA) Combined Phototherapy with acitretin
6 Systemic Treatment Options are Burgeoning methotrexate cyclosporine acitretin
7 Systemic Treatment Options are Burgeoning methotrexate cyclosporine acitretin alefacept efalizumab etanercept
8 Systemic Treatment Options are Burgeoning methotrexate cyclosporine acitretin alefacept efalizumab etanercept rosiglitazone maleate bimosiamose pimecrolimus humira ISA247 AE-941 onercept alitretinoin psoraxine infliximab MEDI-507 IR50 rosiglitazone maleate
9 Systemic Treatment Options are Burgeoning But, Head-to-Head Efficacy Comparisons are Lacking methotrexate alefacept cyclosporine efalizumab acitretin etanercept rosiglitazone maleate bimosiamose pimecrolimus humira ISA247 AE-941 onercept alitretinoin psoraxine infliximab MEDI-507 IR50 rosiglitazone maleate
10 OBJECTIVE From the perspective of U.S. managed healthcare decision makers, to compare efficacy and costs across the range of treatments for moderate-tosevere psoriasis.
11 METHODS Total Costs CE = Clinical Outcomes Johnson NE, 1996.
12 METHODS Drug or UV Total Costs CE = Clinical Outcomes Administration Screening Adverse Events Johnson NE, 1996.
13 METHODS Total Costs CE = Clinical Outcomes Drug or UV Administration Screening Adverse Events Average Wholesale Prices Medicare Reimbursement Rates Johnson NE, 1996.
14 METHODS Total Costs CE = Clinical Outcomes Drug or UV Administration Screening Adverse Events Average Wholesale Prices Medicare Reimbursement Rates Average % Improvement Johnson NE, 1996.
15 METHODS Total Costs CE = Clinical Outcomes Drug or UV Administration Screening Adverse Events Average Wholesale Prices Medicare Reimbursement Rates Average % Improvement Psoriasis Area & Severity Index (PASI) Johnson NE, 1996.
16 METHODS Total Costs CE = Clinical Outcomes Drug or UV Administration Screening Adverse Events Average Wholesale Prices Medicare Reimbursement Rates Average % Improvement Psoriasis Area & Severity Index (PASI) Systematic Literature Review by Expert Panel Consensus Johnson NE, 1996.
17 METHODS Total Costs CE = Clinical Outcomes Drug or UV Administration Screening Adverse Events Average Wholesale Prices Medicare Reimbursement Rates WHAT IS THE COST OF 1% PASI IMPROVEMENT? WHAT IS THE COST OF CLINICALLY MEANINGFUL IMPROVEMENT? (50%, 75%) Johnson NE, Average % Improvement Psoriasis Area & Severity Index (PASI) Systematic Literature Review by Expert Panel Consensus
18 Cost-effectiveness Model Perspective Viewpoint: Health Systems Country of Analysis: United States Target Population: Adults with moderate-to-severe psoriasis Setting of Care: Outpatient Time Frame: 1 year
19 RESULTS OF SYSTEMATIC LITERATURE REVIEW OF PASI EFFICACY MEDLINE 1966-August 2004 Psoriasis AND [Treatment] Published, Peer-Reviewed English Language Total Articles Identified 3,886 Less Excluded Due To: Efficacy Characteristics 1,351 Sample Selection 225 Treatment Characteristics 398 Study Design 1,896 Total Excluded 3,870 Total Efficacy Studies Included 16
20 Treatment Dose PASI% Broadband UVB 3 exp/wk 47%, 80.1%, 83.7% Narrowband UVB 3 exp/wk 73.0% PUVA 3 exp/wk 92.9% 3 exp/wk 61.1% acitretin 48.2 mg/day 60.4% cyclosporine 1.5 mg/kg/day 33.4% 3 mg/kg/day 52.0% methotrexate 7.5 mg/wk 58.4% 15 mg/wk 71.6% alefacept 15 mg/wk 2-12wk courses 45.0% efalizumab 1 mg/kg/wk 51%, 52.0% etanercept 50 mg 2xs weekly 64.2% infliximab 5 mg/kg wks 0, 2, % UVB + acitretin 5-7 exp/wk + 25 mg/d 80.7 % PUVA + acitretin 3 exp/wk + 20mg 97.3%
21 ANNUALIZED COSTS (USD) ANNUAL COSTS TO ACHIEVE Treatment Tx Admin January 2004 Screen AE Average Wholesale Price (AWP) TOTAL COSTS PASI% PASI 1% PASI 75 methotrexate 7.5 mg 595-1,188-1, ,290 methotrexate 15 mg 1,190-1,188-2, ,491 cyclosporine 1.5mg/kg 2,789-1, , ,600 PUVA 3xs/wk 3, , ,111 PUVA 3xs/wk 3, , , Medicare Reimbursement Rates BBUVB+acitretin 25mg 3, , ,149 BB UVB 3xs/wk 4, , ,617 cyclosporine 3 mg/kg 5,019-1, , ,025 PUVA+acitretin 20mg 6, , ,735 acitretin 50 mg 12, , ,217 efalizumab 1 mg/kg 17, , ,960 etanercept 50 mg 21, , ,717 alefacept 15 mg IM 23, ,412-27, ,163 infliximab 5 mg/kg 24,898 1, , ,946
22 ANNUALIZED COSTS (USD) ANNUAL COSTS TO ACHIEVE Treatment Tx Admin Screen AE TOTAL COSTS PASI% PASI 1% PASI 75 methotrexate 7.5 mg 595-1,188-1, ,290 methotrexate 15 mg 1,190-1,188-2, ,491 cyclosporine 1.5mg/kg 2,789-1, , ,600 PUVA 3xs/wk 3, , ,111 PUVA 3xs/wk 3, , ,731 BBUVB+acitretin 25mg 3, , ,149 BB UVB 3xs/wk 4, , ,617 cyclosporine 3 mg/kg 5,019-1, , ,025 Self-Administered PUVA+acitretin 20mg 6, , ,735 acitretin 50 mg 12, , ,217 efalizumab 1 mg/kg 17, , ,960 IV Injection (@ 2004 Medicare Rate) etanercept 50 mg 21, , ,717 alefacept 15 mg IM 23, ,412-27, ,163 IV Infusion (@ 2004 Medicare Rate) infliximab 5 mg/kg 24,898 1, , ,946
23 ANNUALIZED COSTS (USD) ANNUAL COSTS TO ACHIEVE Treatment Tx Admin Screen AE Product TOTAL 2004 Medicare COSTS PASI% Rate PASI 1% PASI 75 methotrexate 7.5 mg 595-1,188-1, ,290 methotrexate 15 mg 1,190-1,188-2, ,491 Consensus 2004 Medicare Rate cyclosporine 1.5mg/kg 2,789-1, , ,600 PUVA 3xs/wk 3, , ,111 PUVA 3xs/wk 3, , ,731 BBUVB+acitretin 25mg 3, , ,149 BB UVB 3xs/wk 4, , ,617 cyclosporine 3 mg/kg 5,019-1, , ,025 PUVA+acitretin 20mg 6, , ,735 acitretin 50 mg 12, , ,217 efalizumab 1 mg/kg 17, , ,960 etanercept 50 mg 21, , ,717 alefacept 15 mg IM 23, ,412-27, ,163 infliximab 5 mg/kg 24,898 1, , ,946
24 ANNUALIZED COSTS (USD) ANNUAL COSTS TO ACHIEVE Treatment Tx Admin Screen AE TOTAL COSTS PASI% PASI 1% PASI 75 methotrexate 7.5 mg 595-1,188-1, ,290 methotrexate 15 mg 1,190-1,188-2, ,491 cyclosporine 1.5mg/kg 2,789-1, , ,600 Risk-adjusted Costs PUVA 3xs/wk 3, , ,111 PUVA 3xs/wk 3, , ,731 BBUVB+acitretin 25mg 3, , ,149 BB UVB 3xs/wk 4, , ,617 cyclosporine 3 mg/kg 5,019-1, , ,025 PUVA+acitretin 20mg 6, , ,735 acitretin 50 mg 12, , ,217 efalizumab 1 mg/kg 17, , ,960 etanercept 50 mg 21, , ,717 alefacept 15 mg IM 23, ,412-27, ,163 infliximab 5 mg/kg 24,898 1, , ,946
25 ANNUALIZED COSTS (USD) ANNUAL COSTS TO ACHIEVE Treatment Tx Admin Screen AE TOTAL COSTS PASI% PASI 1% PASI 75 methotrexate 7.5 mg 595-1,188-1, ,290 methotrexate 15 mg 1,190-1,188-2, ,491 cyclosporine 1.5mg/kg 2,789-1, , ,600 PUVA 3xs/wk 3, , ,111 PUVA 3xs/wk 3, , ,731 BBUVB+acitretin 25mg 3, , ,149 BB UVB 3xs/wk 4, , ,617 cyclosporine 3 mg/kg 5,019-1, , ,025 PUVA+acitretin 20mg 6, , ,735 acitretin 50 mg 12, , ,217 efalizumab 1 mg/kg 17, , ,960 etanercept 50 mg 21, , ,717 alefacept 15 mg IM 23, ,412-27, ,163 infliximab 5 mg/kg 24,898 1, , ,946
26 ANNUALIZED COSTS (USD) ANNUAL COSTS TO ACHIEVE Treatment Tx Admin Screen AE TOTAL COSTS PASI PASI 1% PASI 75 methotrexate 7.5 mg 595-1,188-1, ,290 methotrexate 15 mg 1,190-1,188-2, ,491 cyclosporine 1.5mg/kg 2,789-1, , ,600 Expert Panel Systematic Literature Review PUVA 3xs/wk 3, , ,111 PUVA 3xs/wk 3, , ,731 BBUVB+acitretin 25mg 3, , ,149 BB UVB 3xs/wk 4, , ,617 cyclosporine 3 mg/kg 5,019-1, , ,025 PUVA+acitretin 20mg 6, , ,735 acitretin 50 mg 12, , ,217 efalizumab 1 mg/kg 17, , ,960 etanercept 50 mg 21, , ,717 alefacept 15 mg IM 23, ,412-27, ,163 infliximab 5 mg/kg 24,898 1, , ,946
27 Total Costs / PASI ANNUALIZED COSTS (USD) ANNUAL COSTS TO ACHIEVE Treatment Tx Admin Screen AE TOTAL COSTS PASI PASI 1% PASI 75 methotrexate 7.5 mg 595-1,188-1, ,290 methotrexate 15 mg 1,190-1,188-2, ,491 cyclosporine 1.5mg/kg 2,789-1, , ,600 PUVA 3xs/wk 3, , ,111 PUVA 3xs/wk 3, , ,731 BBUVB+acitretin 25mg 3, , ,149 BB UVB 3xs/wk 4, , ,617 cyclosporine 3 mg/kg 5,019-1, , ,025 PUVA+acitretin 20mg 6, , ,735 acitretin 50 mg 12, , ,217 efalizumab 1 mg/kg 17, , ,960 etanercept 50 mg 21, , ,717 alefacept 15 mg IM 23, ,412-27, ,163 infliximab 5 mg/kg 24,898 1, , ,946
28 PASI 1% * 75 ANNUALIZED COSTS (USD) ANNUAL COSTS TO ACHIEVE Treatment Tx Admin Screen AE TOTAL COSTS PASI PASI 1% methotrexate 7.5 mg 595-1,188-1, ,290 methotrexate 15 mg 1,190-1,188-2, ,491 cyclosporine 1.5mg/kg 2,789-1, , ,600 PUVA 3xs/wk 3, , ,111 PUVA 3xs/wk 3, , ,731 BBUVB+acitretin 25mg 3, , ,149 BB UVB 3xs/wk 4, , ,617 cyclosporine 3 mg/kg 5,019-1, , ,025 PUVA+acitretin 20mg 6, , ,735 acitretin 50 mg 12, , ,217 efalizumab 1 mg/kg 17, , ,960 etanercept 50 mg 21, , ,717 alefacept 15 mg IM 23, ,412-27, ,163 infliximab 5 mg/kg 24,898 1, , ,946 PASI 75%
29 ANNUALIZED COSTS (USD) ANNUAL COSTS TO ACHIEVE Treatment Tx Admin Screen AE TOTAL COSTS PASI PASI 1% methotrexate 7.5 mg 595-1,188-1, ,290 methotrexate 15 mg 1,190-1,188-2, ,491 cyclosporine 1.5mg/kg 2,789-1, , ,600 PUVA 3xs/wk 3, , ,111 PUVA 3xs/wk 3, , ,731 BBUVB+acitretin 25mg 3, , ,149 BB UVB 3xs/wk 4, , ,617 cyclosporine 3 mg/kg 5,019-1, , ,025 PUVA+acitretin 20mg 6, , ,735 acitretin 50 mg 12, , ,217 efalizumab 1 mg/kg 17, , ,960 etanercept 50 mg 21, , ,717 alefacept 15 mg IM 23, ,412-27, ,163 infliximab 5 mg/kg 24,898 1, , ,946 PASI 75%
30 Annualized Total Costs (USD) ( Rx/Tx + Admin + Screen + AE) methotrexate 15 mg PUVA BB UVB+acitretin Annualized Total Costs cyclosporine 1.5 mg/kg BB UVB PUVA + acitretin acitretin 50 mg efalizumab 1mg/kg etanercept 50 mg infliximab 5 mg/kg alefacept 15 mg IM 0 5,000 10,000 15,000 20,000 25,000 30,000 USD
31 MTX 15 mg PUVA 3xs/wk BB UVB+acitretin CyA 1.5mg/kg BB UVB 3xs/w k PUVA+acitretin acitretin 50 mg Annualized Total Costs efalizumab 1 mg/kg etanercept 50 mg infliximab 5 mg/kg alefacept 15 mg IM 0 5,000 10,000 15,000 20,000 25,000 30,000
32 Reported Average PASI % Improvement from Baseline (% Efficacy) CyA 1.5mg/kg alefacept 15 mg IM efalizumab 1 mg/kg acitretin 50 mg etanercept 50 mg PASI % MTX 15 mg BB UVB 3xs/w k BB UVB+acitretin infliximab 5 mg/kg PUVA 3xs/w k PUVA+acitretin 0% 20% 40% 60% 80% 100% PASI % IMPROVEMENT FROM BASELINE
33 MTX 15 mg PUVA 3xs/wk BB UVB+acitretin CyA 1.5mg/kg BB UVB 3xs/w k PUVA+acitretin acitretin 50 mg Annualized Total Costs efalizumab 1 mg/kg etanercept 50 mg infliximab 5 mg/kg alefacept 15 mg IM 0 5,000 10,000 15,000 20,000 25,000 30,000 CyA 1.5mg/kg alefacept 15 mg IM efalizumab 1 mg/kg acitretin 50 mg etanercept 50 mg MTX 15 mg BB UVB 3xs/w k BB UVB+acitretin infliximab 5 mg/kg PASI % PUVA 3xs/w k PUVA+acitretin
34 Annualized Costs to Achieve 1% PASI Improvement (USD) MTX 15 mg PUVA 3xs/w k BBUVB+acitretin PUVA+acitretin CyA 1.5mg/kg acitretin 50 mg infliximab 5 mg/kg etanercept 50 mg efalizumab 1 mg/kg Annualized Costs to Achieve 1% PASI Improvement alefacept 15 mg IM USD
35 MTX 15 mg PUVA 3xs/wk BB UVB+acitretin CyA 1.5mg/kg BB UVB 3xs/w k PUVA+acitretin acitretin 50 mg Annualized Total Costs efalizumab 1 mg/kg etanercept 50 mg infliximab 5 mg/kg alefacept 15 mg IM 0 5,000 10,000 15,000 20,000 25,000 30,000 CyA 1.5mg/kg alefacept 15 mg IM efalizumab 1 mg/kg acitretin 50 mg etanercept 50 mg MTX 15 mg BB UVB 3xs/w k BB UVB+acitretin infliximab 5 mg/kg PASI % PUVA 3xs/w k PUVA+acitretin MTX 15 mg PUVA 3xs/wk BBUVB+acitretin Annualized Costs to Achieve 1% PASI Improvement PUVA+acitretin CyA 1.5mg/kg acitretin 50 mg infliximab 5 mg/kg etanercept 50 mg efalizumab 1 mg/kg alefacept 15 mg IM
36 Annualized Costs to Achieve 75% PASI Improvement (USD) MTX 15 mg PUVA 3xs/w k BBUVB+acitretin Annualized Costs to Achieve 75% PASI Improvement CyA 1.5mg/kg acitretin 50 mg infliximab 5 mg/kg etanercept 50 mg efalizumab 1 mg/kg alefacept 15 mg IM 0 10,000 20,000 30,000 40,000 50,000 USD
37 MTX 15 mg PUVA 3xs/wk BB UVB+acitretin CyA 1.5mg/kg BB UVB 3xs/w k PUVA+acitretin acitretin 50 mg Annualized Total Costs efalizumab 1 mg/kg etanercept 50 mg infliximab 5 mg/kg alefacept 15 mg IM 0 5,000 10,000 15,000 20,000 25,000 30,000 CyA 1.5mg/kg alefacept 15 mg IM efalizumab 1 mg/kg acitretin 50 mg etanercept 50 mg MTX 15 mg BB UVB 3xs/w k BB UVB+acitretin infliximab 5 mg/kg PASI % PUVA 3xs/w k PUVA+acitretin MTX 15 mg PUVA 3xs/wk BBUVB+acitretin Annualized Costs to Achieve % PASI Improvement MTX 15 mg PUVA 3xs/w k BBUVB+acitretin Annualized Costs to Achieve 75% PASI Improvement PUVA+acitretin CyA 1.5mg/kg acitretin 50 mg infliximab 5 mg/kg etanercept 50 mg efalizumab 1 mg/kg alefacept 15 mg IM CyA 1.5mg/kg acitretin 50 mg infliximab 5 mg/kg etanercept 50 mg efalizumab 1 mg/kg alefacept 15 mg IM ,000 20,000 30,000 40,000 50,000
38 Conclusions The most costly treatments are not necessarily the most efficacious. Biologics do not appear to be as costeffective as oral systemics, phototherapies, or combined phototherapy with acitretin. Our results do not justify positioning biologics as first-line therapy for moderateto-severe psoriasis.
39 Discussion Cheryl S. Hankin, Ph.D. President and Chief Scientific Officer BioMedEcon Group San Jose, California
40 Backup
41 STUDY EXCLUSION CRITERIA PASI % improvement from baseline not reported or unavailable for calculation. Number of Ss receiving active treatment <10. Treatment duration <6 or > 14 wks. Diagnosis other than moderate to severe psoriasis or PASI < 8. Sample does not consist predominantly of plaque psoriasis (vs other variants). Mean dose unspecified. For monotherapies, concomitant systemic or phototherapy psoriasis. medication/treatment was received in addition to targeted treatment. Maintenance following stabilization (rather than de novo) treatment. Dose exceeds current recommendation. Duration of study or time at endpoint evaluation unspecified. Study is duplication and previously reviewed by us. Poor Study Quality (did not apply Intent To Treat). Disposition of dropouts unclear. Dropouts excluded from analysis. Biased sample selection (e.g., Ss excluded from participation if poor response to treatment previously). Study Design Retrospective review. Case Study. Randomized assignment to groups?
42 SEARCH PROCEDURE CONDCT SEARCH Medline: /2004 for (Psoriasis AND Tx) Limit English N=xxx Handsearch (Using References Obtained from Medline Articles) N=xxx REVIEW TITLES & ABSTRACTS Review Articles Rx Non Tx Case nonplaque News Possible (N=xx) Letter Methods Practice Patterns Child Adverse Events EXAMINE ARTICLES Review Articles Non Psoriasis (=x) Retrospective Study CANDIDATE ARTICLES Adverse Events News Rx Duplicate WRITTEN EVALUATION of ARTICLES Dose? Retrospective Study Dose > No PASI Dropouts? Dropouts Not Included in Analysis INCLUDED AS PASI EFFICACY Small N Endpt? Duplicate Concomitant Rx/ Tx UNCERTAIN: REQUIRES FURTHER REVIEW BY PACE Biased Sample
43 Systemic Psoriasis Drugs Under Development Drug Name Company Description Status Avandia (rosiglitazone maleate) GlaxoSmithKline oral antidiabetic agent phase III for psoriasis Bimosiamose Encysive endothelin A receptor antagonist phase II Elidel (pimecrolimus) Novartis oral anti-inflammatory phase II Humira Abbott monoclonal antibody phase II for psoriasis ISA247 Isotechnika systemic immunosuppressant phase II complete Neovastat (AE-941) Aeterna angiogenesis inhibitor phase II complete Onercept Serono anti-tnf protein phase III Panretin (alitretinoin) Ligand oral vitamin A derivative phase II Psoraxine Astralis injectable immuno-therapeutic agent phase I/II Remicade (infliximab) Centocor anti-tnf phase III for psoriatic Siplizumab (MEDI- 507) Zorcell (IR502) Medimmune Immune Response monoclonal antibody against T-cell receptor (CD2) causes immune system to shut down activated T cells phase II phase II Source: National Psoriasis Foundation updated June 23, 2004
44 PASI Scoring (0 to 72) Head 0.1 (Erythema H + Infiltration H + Desquamation H ) * Area H 0.3 (Erythema T + Infiltration T + Desquamation T ) * Area T + Torso 0.2 (Erythema u + Infiltration u + Desquamation u ) * Area u + Upper Extremities 0.4 (Erythema L + Infiltration L + Desquamation L ) * Area L + Lower Extremities Note: Severity of Erythema, Infiltration, Desquamation assessed on a 5=pt scale (0=no involvement, 4=extreme involvement); Area assessed on a 7-pt scale (0=<10% involvement, 6= % involvement)
TRANSPARENCY COMMITTEE OPINION. 26 April 2006
 TRANSPARENCY COMMITTEE OPINION 26 April 2006 REMICADE 100 mg powder for concentrate for solution for infusion Box of 1 (CIP code: 562 070.1) Applicant : laboratoires Schering Plough List I Drug for hospital
TRANSPARENCY COMMITTEE OPINION 26 April 2006 REMICADE 100 mg powder for concentrate for solution for infusion Box of 1 (CIP code: 562 070.1) Applicant : laboratoires Schering Plough List I Drug for hospital
ETANERCEPT Generic Brand HICL GCN Exception/Other ETANERCEPT ENBREL GUIDELINES FOR USE INITIAL CRITERIA (NOTE: FOR RENEWAL CRITERIA SEE BELOW)
 Generic Brand HICL GCN Exception/Other ETANERCEPT ENBREL 18830 GUIDELINES FOR USE INITIAL CRITERIA (NOTE: FOR RENEWAL CRITERIA SEE BELOW) 1. Does the patient have a diagnosis of moderate to severe rheumatoid
Generic Brand HICL GCN Exception/Other ETANERCEPT ENBREL 18830 GUIDELINES FOR USE INITIAL CRITERIA (NOTE: FOR RENEWAL CRITERIA SEE BELOW) 1. Does the patient have a diagnosis of moderate to severe rheumatoid
USTEKINUMAB Generic Brand HICL GCN Exception/Other USTEKINUMAB STELARA GUIDELINES FOR USE
 Generic Brand HICL GCN Exception/Other USTEKINUMAB STELARA 36187 GUIDELINES FOR USE INITIAL CRITERIA (NOTE: FOR RENEWAL CRITERIA SEE BELOW) 1. Does the patient have a diagnosis of psoriatic arthritis (PsA)
Generic Brand HICL GCN Exception/Other USTEKINUMAB STELARA 36187 GUIDELINES FOR USE INITIAL CRITERIA (NOTE: FOR RENEWAL CRITERIA SEE BELOW) 1. Does the patient have a diagnosis of psoriatic arthritis (PsA)
NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE SCOPE
 NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE 1 Guideline title SCOPE Psoriasis: the management of psoriasis 1.1 Short title Psoriasis 2 The remit The Department of Health has asked NICE: 'to produce
NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE 1 Guideline title SCOPE Psoriasis: the management of psoriasis 1.1 Short title Psoriasis 2 The remit The Department of Health has asked NICE: 'to produce
An otherwise healthy 12-year-old
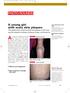 1105 Photo Rounds.finalREV 10/19/05 2:05 PM Page 947 A young girl with scaly skin plaques The patient had numerous thick red plaques on her back and the extensor surfaces of elbows, knees, and forearms
1105 Photo Rounds.finalREV 10/19/05 2:05 PM Page 947 A young girl with scaly skin plaques The patient had numerous thick red plaques on her back and the extensor surfaces of elbows, knees, and forearms
A Cost Comparison of Treatments of Moderate to Severe Psoriasis
 University of Massachusetts Medical School escholarship@umms Meyers Primary Care Institute Publications and Presentations Meyers Primary Care Institute 5-2005 A Cost Comparison of Treatments of Moderate
University of Massachusetts Medical School escholarship@umms Meyers Primary Care Institute Publications and Presentations Meyers Primary Care Institute 5-2005 A Cost Comparison of Treatments of Moderate
PSORIASIS IS A CHRONIC AUTOIMmune
 STUDY Recent Trends in Systemic Psoriasis Treatment Costs Vivianne Beyer, MD; Stephen E. Wolverton, MD Objectives: To analyze the current total cost of systemic therapy for psoriasis and to compare annual
STUDY Recent Trends in Systemic Psoriasis Treatment Costs Vivianne Beyer, MD; Stephen E. Wolverton, MD Objectives: To analyze the current total cost of systemic therapy for psoriasis and to compare annual
Josephine Mauskopf Miny Samuel Doreen McBride Usha G. Mallya Steven R. Feldman
 PharmacoEconomics (2014) 32:395 409 DOI 10.1007/s40273-014-0130-5 SYSTEMATIC REVIEW Treatment Sequencing After Failure of the First Biologic in Cost-Effectiveness Models of Psoriasis: A Systematic Review
PharmacoEconomics (2014) 32:395 409 DOI 10.1007/s40273-014-0130-5 SYSTEMATIC REVIEW Treatment Sequencing After Failure of the First Biologic in Cost-Effectiveness Models of Psoriasis: A Systematic Review
Clinical Policy: Ustekinumab (Stelara) Reference Number: ERX.SPA.01 Effective Date:
 Clinical Policy: (Stelara) Reference Number: ERX.SPA.01 Effective Date: 04.01.17 Last Review Date: 11.17 Revision Log See Important Reminder at the end of this policy for important regulatory and legal
Clinical Policy: (Stelara) Reference Number: ERX.SPA.01 Effective Date: 04.01.17 Last Review Date: 11.17 Revision Log See Important Reminder at the end of this policy for important regulatory and legal
Injectable Biologic Case Studies
 ROBERT J. LIPSY, PharmD, FASHP, BCPS ABSTRACT OBJECTIVE: To identify strategies used by a commercial managed care organization (MCO) to affect appropriate cost-effective use and prioritize payment for
ROBERT J. LIPSY, PharmD, FASHP, BCPS ABSTRACT OBJECTIVE: To identify strategies used by a commercial managed care organization (MCO) to affect appropriate cost-effective use and prioritize payment for
Clinical Policy: Secukinumab (Cosentyx) Reference Number: ERX.SPA.165 Effective Date:
 Clinical Policy: (Cosentyx) Reference Number: ERX.SPA.165 Effective Date: 10.01.16 Last Review Date: 11.17 Revision Log See Important Reminder at the end of this policy for important regulatory and legal
Clinical Policy: (Cosentyx) Reference Number: ERX.SPA.165 Effective Date: 10.01.16 Last Review Date: 11.17 Revision Log See Important Reminder at the end of this policy for important regulatory and legal
A systematic review of treatments for severe psoriasis Griffiths C E, Clark C M, Chalmers R J, Li Wan Po A, Williams H C
 A systematic review of treatments for severe psoriasis Griffiths C E, Clark C M, Chalmers R J, Li Wan Po A, Williams H C Authors' objectives To compare the effectiveness of currently available treatments
A systematic review of treatments for severe psoriasis Griffiths C E, Clark C M, Chalmers R J, Li Wan Po A, Williams H C Authors' objectives To compare the effectiveness of currently available treatments
ustekinumab (Stelara )
 Applies to all products administered or underwritten by Blue Cross and Blue Shield of Louisiana and its subsidiary, HMO Louisiana, Inc.(collectively referred to as the Company ), unless otherwise provided
Applies to all products administered or underwritten by Blue Cross and Blue Shield of Louisiana and its subsidiary, HMO Louisiana, Inc.(collectively referred to as the Company ), unless otherwise provided
Drugs and Applicable Coding: J-code: Enbrel-J1438; Humira-J0135; Remicade-J1745; Inflectra-Q5102; Cimzia-J0718; Simponi-J1602 Renflexis - pending
 Policy Subject: Anti-TNF Agents Policy Number: SHS PBD16 Category: Rheumatology & Autoimmune Policy Type: Medical Pharmacy Department: Pharmacy Product (check all that apply): Group HMO/POS Individual
Policy Subject: Anti-TNF Agents Policy Number: SHS PBD16 Category: Rheumatology & Autoimmune Policy Type: Medical Pharmacy Department: Pharmacy Product (check all that apply): Group HMO/POS Individual
Follow this and additional works at: Part of the Skin and Connective Tissue Diseases Commons
 Philadelphia College of Osteopathic Medicine DigitalCommons@PCOM PCOM Physician Assistant Studies Student Scholarship Student Dissertations, Theses and Papers 2014 Is the Addition of a Topical Agent to
Philadelphia College of Osteopathic Medicine DigitalCommons@PCOM PCOM Physician Assistant Studies Student Scholarship Student Dissertations, Theses and Papers 2014 Is the Addition of a Topical Agent to
Clinical Policy: Ixekizumab (Taltz) Reference Number: CP.PHAR.257 Effective Date: Last Review Date: 05.18
 Clinical Policy: (Taltz) Reference Number: CP.PHAR.257 Effective Date: 08.01.16 Last Review Date: 05.18 Line of Business: Medicaid Revision Log See Important Reminder at the end of this policy for important
Clinical Policy: (Taltz) Reference Number: CP.PHAR.257 Effective Date: 08.01.16 Last Review Date: 05.18 Line of Business: Medicaid Revision Log See Important Reminder at the end of this policy for important
Predicting the Response to Phototherapy for Psoriasis Patients
 A*STAR-NHG-NTU Skin Research Grant Joint Workshop 17 October 2015 Predicting the Response to Phototherapy for Psoriasis Patients Is it possible? Dr Eugene Tan Consultant Dermatologist National Skin Centre
A*STAR-NHG-NTU Skin Research Grant Joint Workshop 17 October 2015 Predicting the Response to Phototherapy for Psoriasis Patients Is it possible? Dr Eugene Tan Consultant Dermatologist National Skin Centre
The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION. 13 May 2009
 The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 13 May 2009 STELARA 45 mg, solution for injection B/1 x 0.5 ml vial (CIP code: 392 586-2) JANSSEN-CILAG Ustekinumab
The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 13 May 2009 STELARA 45 mg, solution for injection B/1 x 0.5 ml vial (CIP code: 392 586-2) JANSSEN-CILAG Ustekinumab
Prior Authorization Conditions for Approval of Enbrel (etanercept) Website Form Submit request via: Fax
 Prior Authorization Conditions for Approval of Enbrel (etanercept) Website Form www.highmarkhealthoptions.com Submit request via: Fax - 1-855-476-4158 All requests for Enbrel (etanercept) require a prior
Prior Authorization Conditions for Approval of Enbrel (etanercept) Website Form www.highmarkhealthoptions.com Submit request via: Fax - 1-855-476-4158 All requests for Enbrel (etanercept) require a prior
2017 Blue Cross and Blue Shield of Louisiana
 Applies to all products administered or underwritten by Blue Cross and Blue Shield of Louisiana and its subsidiary, HMO Louisiana, Inc.(collectively referred to as the Company ), unless otherwise provided
Applies to all products administered or underwritten by Blue Cross and Blue Shield of Louisiana and its subsidiary, HMO Louisiana, Inc.(collectively referred to as the Company ), unless otherwise provided
Clinical Policy: Apremilast (Otezla) Reference Number: CP.PHAR.245 Effective Date: Last Review Date: Line of Business: HIM, Medicaid
 Clinical Policy: (Otezla) Reference Number: CP.PHAR.245 Effective Date: 08.16 Last Review Date: 11.18 Line of Business: HIM, Medicaid Revision Log See Important Reminder at the end of this policy for important
Clinical Policy: (Otezla) Reference Number: CP.PHAR.245 Effective Date: 08.16 Last Review Date: 11.18 Line of Business: HIM, Medicaid Revision Log See Important Reminder at the end of this policy for important
TNF Inhibitors: Lessons From Immunogenicity
 TNF Inhibitors: Lessons From Immunogenicity Edward Keystone, MD, FRCP(C) Professor of Medicine University of Toronto Toronto, Canada Edward Keystone, MD FRCP(C) Disclosures Sources of Funding for Research:
TNF Inhibitors: Lessons From Immunogenicity Edward Keystone, MD, FRCP(C) Professor of Medicine University of Toronto Toronto, Canada Edward Keystone, MD FRCP(C) Disclosures Sources of Funding for Research:
Cigna Drug and Biologic Coverage Policy
 Cigna Drug and Biologic Coverage Policy Subject Apremilast Table of Contents Coverage Policy... 1 General Background... 2 Coding/Billing Information... 4 References... 4 Effective Date... 1/1/2018 Next
Cigna Drug and Biologic Coverage Policy Subject Apremilast Table of Contents Coverage Policy... 1 General Background... 2 Coding/Billing Information... 4 References... 4 Effective Date... 1/1/2018 Next
Economic Evaluation of Apremilast in the Treatment of Moderate to Severe Psoriasis in the United States
 1142 Economic Evaluation of Apremilast in the Treatment of Moderate to Severe Psoriasis in the United States Tom Tencer, PhD 1 ; Zoe Clancy, PharmD, MS 1 ; Vidya Damera, MS 2 ; Frank Zhang MD, MPH 1 ;
1142 Economic Evaluation of Apremilast in the Treatment of Moderate to Severe Psoriasis in the United States Tom Tencer, PhD 1 ; Zoe Clancy, PharmD, MS 1 ; Vidya Damera, MS 2 ; Frank Zhang MD, MPH 1 ;
QUAN V. DOAN, PharmD; CHIUN-FANG CHIOU, PhD; and ROBERT W. DUBOIS, MD, PhD
 SUBJECT REVIEW Review of Eight Pharmacoeconomic Studies of the Value of Biologic DMARDs (Adalimumab, Etanercept, and Infliximab) in the Management of Rheumatoid Arthritis QUAN V. DOAN, PharmD; CHIUN-FANG
SUBJECT REVIEW Review of Eight Pharmacoeconomic Studies of the Value of Biologic DMARDs (Adalimumab, Etanercept, and Infliximab) in the Management of Rheumatoid Arthritis QUAN V. DOAN, PharmD; CHIUN-FANG
They are updated regularly as new NICE guidance is published. To view the latest version of this NICE Pathway see:
 bring together everything NICE says on a topic in an interactive flowchart. are interactive and designed to be used online. They are updated regularly as new NICE guidance is published. To view the latest
bring together everything NICE says on a topic in an interactive flowchart. are interactive and designed to be used online. They are updated regularly as new NICE guidance is published. To view the latest
Horizon Scanning Centre March Tildrakizumab for moderate to severe plaque psoriasis SUMMARY NIHR HSC ID: 6798
 Horizon Scanning Centre March 2015 Tildrakizumab for moderate to severe plaque psoriasis SUMMARY NIHR HSC ID: 6798 This briefing is based on information available at the time of research and a limited
Horizon Scanning Centre March 2015 Tildrakizumab for moderate to severe plaque psoriasis SUMMARY NIHR HSC ID: 6798 This briefing is based on information available at the time of research and a limited
Pediatric Use: Safety and effectiveness of Ustekinumab (STELARA ) in pediatric patients have not been evaluated.
 Original Issue Date (Created): January 1, 2010 Most Recent Review Date (Revised): January 28, 2014 Effective Date: April 1, 2014 I. POLICY Preauthorization Requirements for Ustekinumab (STELARA ) Note:
Original Issue Date (Created): January 1, 2010 Most Recent Review Date (Revised): January 28, 2014 Effective Date: April 1, 2014 I. POLICY Preauthorization Requirements for Ustekinumab (STELARA ) Note:
Clinical Policy: Secukinumab (Cosentyx) Reference Number: CP.PHAR.261 Effective Date: Last Review Date: Line of Business: HIM, Medicaid
 Clinical Policy: (Cosentyx) Reference Number: CP.PHAR.261 Effective Date: 08.16 Last Review Date: 11.18 Line of Business: HIM, Medicaid Coding Implications Revision Log See Important Reminder at the end
Clinical Policy: (Cosentyx) Reference Number: CP.PHAR.261 Effective Date: 08.16 Last Review Date: 11.18 Line of Business: HIM, Medicaid Coding Implications Revision Log See Important Reminder at the end
Clinical Policy: Ixekizumab (Taltz) Reference Number: ERX.SPA.122 Effective Date:
 Clinical Policy: (Taltz) Reference Number: ERX.SPA.122 Effective Date: 10.01.16 Last Review Date: 11.18 Revision Log See Important Reminder at the end of this policy for important regulatory and legal
Clinical Policy: (Taltz) Reference Number: ERX.SPA.122 Effective Date: 10.01.16 Last Review Date: 11.18 Revision Log See Important Reminder at the end of this policy for important regulatory and legal
Clinical Policy: Ixekizumab (Taltz) Reference Number: CP.PHAR.257 Effective Date: Last Review Date: 11.18
 Clinical Policy: (Taltz) Reference Number: CP.PHAR.257 Effective Date: 08.01.16 Last Review Date: 11.18 Line of Business: Medicaid Revision Log See Important Reminder at the end of this policy for important
Clinical Policy: (Taltz) Reference Number: CP.PHAR.257 Effective Date: 08.01.16 Last Review Date: 11.18 Line of Business: Medicaid Revision Log See Important Reminder at the end of this policy for important
The implication of an immunologic
 Evidence-Based Review of Biologic Therapy for Psoriasis: Infliximab, Etanercept, Adalimumab, Efalizumab, and Alefacept Jeffrey M. Weinberg, MD; Robin Buchholz, MD; Noah Scheinfeld, MD DERMATOLOGY ABSTRACT
Evidence-Based Review of Biologic Therapy for Psoriasis: Infliximab, Etanercept, Adalimumab, Efalizumab, and Alefacept Jeffrey M. Weinberg, MD; Robin Buchholz, MD; Noah Scheinfeld, MD DERMATOLOGY ABSTRACT
Psoriasis. and Biologic Treatments. Choose the best treatment to regain your quality of life.
 Psoriasis and Biologic Treatments Choose the best treatment to regain your quality of life. PSORIASIS, What is this disease? Psoriasis is a common disease, that affects up to 4% of the population. It usually
Psoriasis and Biologic Treatments Choose the best treatment to regain your quality of life. PSORIASIS, What is this disease? Psoriasis is a common disease, that affects up to 4% of the population. It usually
BJD. Summary. British Journal of Dermatology THERAPEUTICS
 THERAPEUTICS BJD British Journal of Dermatology Efficacy of psoralen plus ultraviolet A therapy vs. biologics in moderate to severe chronic plaque psoriasis: retrospective data analysis of a patient registry
THERAPEUTICS BJD British Journal of Dermatology Efficacy of psoralen plus ultraviolet A therapy vs. biologics in moderate to severe chronic plaque psoriasis: retrospective data analysis of a patient registry
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE. Proposed Health Technology Appraisal
 NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Proposed Health Technology Appraisal Dimethyl fumarate for treating moderate to severe Draft scope (pre-referral) Draft remit/appraisal objective To appraise
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Proposed Health Technology Appraisal Dimethyl fumarate for treating moderate to severe Draft scope (pre-referral) Draft remit/appraisal objective To appraise
BIOLOGICS THE KEY TO NEXT GENERATION DERMATOLOGY R&D?
 White Paper BIOLOGICS THE KEY TO NEXT GENERATION DERMATOLOGY R&D? Clinical trial considerations and best practices TABLE OF CONTENTS Introduction 3 Making a case for biologics 3 Biologic mechanism of action:
White Paper BIOLOGICS THE KEY TO NEXT GENERATION DERMATOLOGY R&D? Clinical trial considerations and best practices TABLE OF CONTENTS Introduction 3 Making a case for biologics 3 Biologic mechanism of action:
1 Introduction. Kim A. Papp 1 April W. Armstrong. Wendell Valdecantos 4
 Am J Clin Dermatol (216) 17:79 86 DOI 1.17/s4257-15-161-5 ORIGINAL RESEARCH ARTICLE Efficacy in Patients with Psoriasis Who Received or Did Not Respond to Prior Systemic Therapy: A Pooled Post Hoc Analysis
Am J Clin Dermatol (216) 17:79 86 DOI 1.17/s4257-15-161-5 ORIGINAL RESEARCH ARTICLE Efficacy in Patients with Psoriasis Who Received or Did Not Respond to Prior Systemic Therapy: A Pooled Post Hoc Analysis
Canadian Society of Internal Medicine Annual Meeting 2016 Montreal, QC
 Canadian Society of Internal Medicine Annual Meeting 2016 Montreal, QC Update on the Treatment of Rheumatoid Arthritis Sabrina Fallavollita MDCM McGill University Canadian Society of Internal Medicine
Canadian Society of Internal Medicine Annual Meeting 2016 Montreal, QC Update on the Treatment of Rheumatoid Arthritis Sabrina Fallavollita MDCM McGill University Canadian Society of Internal Medicine
Cost per Responder of Apremilast Versus Etanercept, Adalimumab, and Ustekinumab in Patients With Moderate to Severe Psoriasis
 1108 Cost per Responder of Apremilast Versus Etanercept, Adalimumab, and Ustekinumab in Patients With Moderate to Severe Psoriasis Steven R. Feldman, MD, PhD 1 ; Tom Tencer, PhD 2 ; Zoe Clancy, PharmD,
1108 Cost per Responder of Apremilast Versus Etanercept, Adalimumab, and Ustekinumab in Patients With Moderate to Severe Psoriasis Steven R. Feldman, MD, PhD 1 ; Tom Tencer, PhD 2 ; Zoe Clancy, PharmD,
Review Article The Safety of Systemic Treatments That Can Be Used for Geriatric Psoriasis Patients: A Review
 Dermatology Research and Practice Volume 2012, Article ID 367475, 4 pages doi:10.1155/2012/367475 Review Article The Safety of Systemic Treatments That Can Be Used for Geriatric Psoriasis Patients: A Review
Dermatology Research and Practice Volume 2012, Article ID 367475, 4 pages doi:10.1155/2012/367475 Review Article The Safety of Systemic Treatments That Can Be Used for Geriatric Psoriasis Patients: A Review
Psoriasis. and Biologic Treatments. Choose the best treatment to regain your quality of life. Edition 4
 Psoriasis and Biologic Treatments Choose the best treatment to regain your quality of life. Edition 4 PSORIASIS, What is this disease? Psoriasis is a common disease, that affects up to 4% of the population.
Psoriasis and Biologic Treatments Choose the best treatment to regain your quality of life. Edition 4 PSORIASIS, What is this disease? Psoriasis is a common disease, that affects up to 4% of the population.
Horizon Scanning Centre March Ixekizumab for moderate to severe chronic plaque psoriasis SUMMARY NIHR HSC ID: 5209
 Horizon Scanning Centre March 2015 Ixekizumab for moderate to severe chronic plaque psoriasis SUMMARY NIHR HSC ID: 5209 This briefing is based on information available at the time of research and a limited
Horizon Scanning Centre March 2015 Ixekizumab for moderate to severe chronic plaque psoriasis SUMMARY NIHR HSC ID: 5209 This briefing is based on information available at the time of research and a limited
BASIC/CLINICAL SCIENCE. Resident Education: Effect of the Sixth National Psoriasis Foundation Chief Residents Meeting
 BASIC/CLINICAL SCIENCE Resident Education: Effect of the Sixth National Psoriasis Foundation Chief Residents Meeting Heather M. Greist, Daniel J. Pearce, Molly Blauvelt, and Steven R. Feldman Background:
BASIC/CLINICAL SCIENCE Resident Education: Effect of the Sixth National Psoriasis Foundation Chief Residents Meeting Heather M. Greist, Daniel J. Pearce, Molly Blauvelt, and Steven R. Feldman Background:
ADALIMUMAB Generic Brand HICL GCN Exception/Other ADALIMUMAB HUMIRA GUIDELINES FOR USE INITIAL CRITERIA (NOTE: FOR RENEWAL CRITERIA SEE BELOW)
 Generic Brand HICL GCN Exception/Other ADALIMUMAB HUMIRA 24800 GUIDELINES FOR USE INITIAL CRITERIA (NOTE: FOR RENEWAL CRITERIA SEE BELOW) 1. Does the patient have a diagnosis of moderate to severe rheumatoid
Generic Brand HICL GCN Exception/Other ADALIMUMAB HUMIRA 24800 GUIDELINES FOR USE INITIAL CRITERIA (NOTE: FOR RENEWAL CRITERIA SEE BELOW) 1. Does the patient have a diagnosis of moderate to severe rheumatoid
STUDY. Extent and Clinical Consequences of Antibody Formation Against Adalimumab in Patients With Plaque Psoriasis
 STUDY Extent and Clinical Consequences of Antibody Formation Against Adalimumab in Patients With Plaque Psoriasis Lidian L. A. Lecluse, MD; Rieke J. B. Driessen, MD; Phyllis I. Spuls, MD, PhD; Elke M.
STUDY Extent and Clinical Consequences of Antibody Formation Against Adalimumab in Patients With Plaque Psoriasis Lidian L. A. Lecluse, MD; Rieke J. B. Driessen, MD; Phyllis I. Spuls, MD, PhD; Elke M.
Medical Policy An independent licensee of the Blue Cross Blue Shield Association
 Otezla (apremilast) Page 1 of 7 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: Otezla (apremilast) Prime Therapeutics will review Prior Authorization requests Prior
Otezla (apremilast) Page 1 of 7 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: Otezla (apremilast) Prime Therapeutics will review Prior Authorization requests Prior
Psoriasis. Causes of Psoriasis
 Psoriasis Psoriasis is a common, chronic, relapsing/remitting, immune-mediated systemic disease characterized by skin lesions including red, scaly patches, papules, and plaques, which usually itch. The
Psoriasis Psoriasis is a common, chronic, relapsing/remitting, immune-mediated systemic disease characterized by skin lesions including red, scaly patches, papules, and plaques, which usually itch. The
STELARA (ustekinumab)
 STELARA (ustekinumab) Non-Discrimination Statement and Multi-Language Interpreter Services information are located at the end of this document. Coverage for services, procedures, medical devices and drugs
STELARA (ustekinumab) Non-Discrimination Statement and Multi-Language Interpreter Services information are located at the end of this document. Coverage for services, procedures, medical devices and drugs
BJD. Summary. British Journal of Dermatology THERAPEUTICS
 THERAPEUTICS BJD British Journal of Dermatology Efficacy and safety of infliximab vs. methotrexate in patients with moderate-to-severe plaque psoriasis: results of an open-label, active-controlled, randomized
THERAPEUTICS BJD British Journal of Dermatology Efficacy and safety of infliximab vs. methotrexate in patients with moderate-to-severe plaque psoriasis: results of an open-label, active-controlled, randomized
Elements of Successful PBS Applications. Barbara Radulski RN. Copyright
 Elements of Successful PBS Applications Barbara Radulski RN PBS Requirements April 1 2006 THE RULES PBS Requirements 18 years and over Psoriasis x 6 months Failed to achieve an adequate response to 3 systemic
Elements of Successful PBS Applications Barbara Radulski RN PBS Requirements April 1 2006 THE RULES PBS Requirements 18 years and over Psoriasis x 6 months Failed to achieve an adequate response to 3 systemic
Bilaga 1.till rapport. Bilaga 1 Tabell över inkluderade studier/ Appendix 1 Description of included studies
 Bilaga 1.till rapport Ljusbehandling och systemisk behandling av psoriasis, rapport nr 278 (2018) Bilaga 1 Tabell över inkluderade studier/ Appendix 1 Description of included studies Description of included
Bilaga 1.till rapport Ljusbehandling och systemisk behandling av psoriasis, rapport nr 278 (2018) Bilaga 1 Tabell över inkluderade studier/ Appendix 1 Description of included studies Description of included
Scottish Medicines Consortium
 Scottish Medicines Consortium ustekinumab, 45mg solution for injection (Stelara ) No. (572/09) Janssen-Cilag Ltd 15 January 2010 The Scottish Medicines Consortium (SMC) has completed its assessment of
Scottish Medicines Consortium ustekinumab, 45mg solution for injection (Stelara ) No. (572/09) Janssen-Cilag Ltd 15 January 2010 The Scottish Medicines Consortium (SMC) has completed its assessment of
Background AN UPDATED LOOK AT TREATMENTS FOR PLAQUE PSORIASIS JULY 2018 PLAQUE PSORIASIS TARGETED IMMUNOMODULATORS AS A TREATMENT OPTION
 JULY 2018 Background PLAQUE PSORIASIS Plaque psoriasis is a common disease affecting 3% of the US population that causes itchy, red, scaly, raised lesions on the skin, most commonly on the elbows, knees,
JULY 2018 Background PLAQUE PSORIASIS Plaque psoriasis is a common disease affecting 3% of the US population that causes itchy, red, scaly, raised lesions on the skin, most commonly on the elbows, knees,
Pragmatic Approach to Psoriasis 2010
 Pragmatic Approach to Psoriasis 21 Ted Rosen, MD Professor of Dermatology Baylor College of Medicine Houston, Texas USA Conflict of Interest Disclosure Honoraria for Speaker s s Bureau Abbott Amgen Canada
Pragmatic Approach to Psoriasis 21 Ted Rosen, MD Professor of Dermatology Baylor College of Medicine Houston, Texas USA Conflict of Interest Disclosure Honoraria for Speaker s s Bureau Abbott Amgen Canada
1. Background: Infliximab is administered parenterally; therefore, it is not covered under retail pharmacy benefits.
 Subject: Infliximab (Remicade ) Original Original Committee Approval: October 13, 2006 Revised Last Committee Approval: December 3, 2008 Last Review: October 19, 2007 1. Background: Infliximab is a genetically
Subject: Infliximab (Remicade ) Original Original Committee Approval: October 13, 2006 Revised Last Committee Approval: December 3, 2008 Last Review: October 19, 2007 1. Background: Infliximab is a genetically
COMMON SKIN CONDITIONS IN PRIMARY CARE. Ibrahim M. Zayneh, MD Dermatology Private Practice, Portsmouth, Ohio
 COMMON SKIN CONDITIONS IN PRIMARY CARE Ibrahim M. Zayneh, MD Dermatology Private Practice, Portsmouth, Ohio DISCLOSURE The Speaker and members of the planning committee do not have a conflict of interest
COMMON SKIN CONDITIONS IN PRIMARY CARE Ibrahim M. Zayneh, MD Dermatology Private Practice, Portsmouth, Ohio DISCLOSURE The Speaker and members of the planning committee do not have a conflict of interest
Ustekinumab for the treatment of moderate to severe psoriasis
 DOI: 10.3310/hta13suppl3/10 Health Technology Assessment 2009; Vol. 13: Suppl. 3 Ustekinumab for the treatment of moderate to severe psoriasis E Gospodarevskaya, J Picot, K Cooper, E Loveman* and A Takeda
DOI: 10.3310/hta13suppl3/10 Health Technology Assessment 2009; Vol. 13: Suppl. 3 Ustekinumab for the treatment of moderate to severe psoriasis E Gospodarevskaya, J Picot, K Cooper, E Loveman* and A Takeda
Corporate Medical Policy
 Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: abatacept_orencia 4/2008 2/2018 2/2019 2/2018 Description of Procedure or Service Abatacept (Orencia ), a
Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: abatacept_orencia 4/2008 2/2018 2/2019 2/2018 Description of Procedure or Service Abatacept (Orencia ), a
The Natural History of Psoriasis and Treatment Goals
 The Natural History of Psoriasis and Treatment Goals Psoriasis Epidemiology Prevalence Affects 2 3% of adult population (>7 million in US) Caucasians: 25% 2.5% African Americans: 1.3% (more likely to have
The Natural History of Psoriasis and Treatment Goals Psoriasis Epidemiology Prevalence Affects 2 3% of adult population (>7 million in US) Caucasians: 25% 2.5% African Americans: 1.3% (more likely to have
P2866 P2867. Author disclosure: Consultant to Genentech, Inc. 100% sponsored by Genentech, Inc.
 P2864 Case studies of efalizumab in hand and foot psoriasis Jeffrey Sobell, Tufts-New England Medical Center, Boston, MA, United ; Scott Fretzin, MD, Indiana University Medical Center, Indianapolis, IN,
P2864 Case studies of efalizumab in hand and foot psoriasis Jeffrey Sobell, Tufts-New England Medical Center, Boston, MA, United ; Scott Fretzin, MD, Indiana University Medical Center, Indianapolis, IN,
Appendix 1: Frequently Asked Questions
 Appendix 1: Frequently Asked Questions 1. What is the funding status of Inflectra (infliximab)? Effective February 25 2016, Inflectra (infliximab) will be added to the Ontario Drug Benefit (ODB) Formulary
Appendix 1: Frequently Asked Questions 1. What is the funding status of Inflectra (infliximab)? Effective February 25 2016, Inflectra (infliximab) will be added to the Ontario Drug Benefit (ODB) Formulary
Clinical Policy: Brodalumab (Siliq) Reference Number: CP.PHAR.375 Effective Date: Last Review Date: 05.18
 Clinical Policy: (Siliq) Reference Number: CP.PHAR.375 Effective Date: 06.01.18 Last Review Date: 05.18 Line of Business: Medicaid Revision Log See Important Reminder at the end of this policy for important
Clinical Policy: (Siliq) Reference Number: CP.PHAR.375 Effective Date: 06.01.18 Last Review Date: 05.18 Line of Business: Medicaid Revision Log See Important Reminder at the end of this policy for important
Clinical Policy: Ustekinumab (Stelara) Reference Number: CP.PHAR.264 Effective Date: Last Review Date: Line of Business: HIM, Medicaid
 Clinical Policy: (Stelara) Reference Number: CP.PHAR.264 Effective Date: 08.16 Last Review Date: 05.18 Line of Business: HIM, Medicaid Coding Implications Revision Log See Important Reminder at the end
Clinical Policy: (Stelara) Reference Number: CP.PHAR.264 Effective Date: 08.16 Last Review Date: 05.18 Line of Business: HIM, Medicaid Coding Implications Revision Log See Important Reminder at the end
UnitedHealthcare Pharmacy Clinical Pharmacy Programs
 UnitedHealthcare Pharmacy Clinical Pharmacy Programs Program Number 2018 P 1097-7 Program Prior Authorization/Notification Medication *Stelara (ustekinumab) *This program applies to the subcutaneous formulation
UnitedHealthcare Pharmacy Clinical Pharmacy Programs Program Number 2018 P 1097-7 Program Prior Authorization/Notification Medication *Stelara (ustekinumab) *This program applies to the subcutaneous formulation
KEY MESSAGES. Psoriasis patients are more prone to cardiovascular diseases, stroke, lymphoma and non-melanoma skin cancers, and increased mortality.
 KEY MESSAGES Psoriasis is a genetically determined, systemic immune-mediated chronic inflammatory disease that affects primarily the skin and joints. Psoriasis Vulgaris is characterised by well-demarcated
KEY MESSAGES Psoriasis is a genetically determined, systemic immune-mediated chronic inflammatory disease that affects primarily the skin and joints. Psoriasis Vulgaris is characterised by well-demarcated
COSENTYX (secukinumab)
 COSENTYX (secukinumab) Non-Discrimination Statement and Multi-Language Interpreter Services information are located at the end of this document. Coverage for services, procedures, medical devices and drugs
COSENTYX (secukinumab) Non-Discrimination Statement and Multi-Language Interpreter Services information are located at the end of this document. Coverage for services, procedures, medical devices and drugs
USTEKINUMAB. London New Drugs Group APC/DTC Briefing Document. May 2009
 Page 1 London New Drugs Group APC/DTC Briefing Document USTEKINUMAB Contents Summary 1 Background 4 Clinical efficacy 5 Cost implications 9 Reference list 10 Appendices 11 Produced for the London New Drugs
Page 1 London New Drugs Group APC/DTC Briefing Document USTEKINUMAB Contents Summary 1 Background 4 Clinical efficacy 5 Cost implications 9 Reference list 10 Appendices 11 Produced for the London New Drugs
Update on systemic therapies and emerging treatments How do I choose a systemic agent?
 Update on systemic therapies and emerging treatments How do I choose a systemic agent? Amy S. Paller, M.D. Walter J. Hamlin Professor and Chair of Dermatology Professor of Pediatrics Northwestern University
Update on systemic therapies and emerging treatments How do I choose a systemic agent? Amy S. Paller, M.D. Walter J. Hamlin Professor and Chair of Dermatology Professor of Pediatrics Northwestern University
Table SIII. Evidence table. First author (Ref), year Design Dosage Outcome measures Adalimumab Ryan (12), Date of evaluation
 Table SIII. Evidence table First author (Ref), year Design Dosage Outcome measures Adalimumab Ryan (12), 2009 (n = 39 a ) van Lümig (13), 2010 (n = 30 a ) Warren (14), 2010 (n = 46 a ) Retrosp b Adalimumab
Table SIII. Evidence table First author (Ref), year Design Dosage Outcome measures Adalimumab Ryan (12), 2009 (n = 39 a ) van Lümig (13), 2010 (n = 30 a ) Warren (14), 2010 (n = 46 a ) Retrosp b Adalimumab
Psoriasis. Andrei Metelitsa, MD, FRCPC, FAAD Clinical Associate Professor, Dermatology, U of C Co-Director, Institute for Skin Advancement
 Psoriasis Andrei Metelitsa, MD, FRCPC, FAAD Clinical Associate Professor, Dermatology, U of C Co-Director, Institute for Skin Advancement Copyright 2017 by Sea Courses Inc. All rights reserved. No part
Psoriasis Andrei Metelitsa, MD, FRCPC, FAAD Clinical Associate Professor, Dermatology, U of C Co-Director, Institute for Skin Advancement Copyright 2017 by Sea Courses Inc. All rights reserved. No part
Horizon Scanning Centre May Brodalumab for moderate to severe plaque psoriasis SUMMARY NIHR HSC ID: 5524
 Horizon Scanning Centre May 2014 Brodalumab for moderate to severe plaque psoriasis SUMMARY NIHR HSC ID: 5524 This briefing is based on information available at the time of research and a limited literature
Horizon Scanning Centre May 2014 Brodalumab for moderate to severe plaque psoriasis SUMMARY NIHR HSC ID: 5524 This briefing is based on information available at the time of research and a limited literature
Ustekinumab (Stelara )
 Applies to all products administered or underwritten by Blue Cross and Blue Shield of Louisiana and its subsidiary, HMO Louisiana, Inc.(collectively referred to as the Company ), unless otherwise provided
Applies to all products administered or underwritten by Blue Cross and Blue Shield of Louisiana and its subsidiary, HMO Louisiana, Inc.(collectively referred to as the Company ), unless otherwise provided
2019 COLLECTION TYPE: MIPS CLINICAL QUALITY MEASURES (CQMS) MEASURE TYPE: Outcome High Priority
 Quality ID #410: Psoriasis: Clinical Response to Systemic Medications National Quality Strategy Domain: Person and Caregiver-Centered Experience and Outcomes Meaningful Measure Area: Management of Chronic
Quality ID #410: Psoriasis: Clinical Response to Systemic Medications National Quality Strategy Domain: Person and Caregiver-Centered Experience and Outcomes Meaningful Measure Area: Management of Chronic
1. Does the patient have a diagnosis of moderate to severe polyarticular juvenile idiopathic arthritis (PJIA)?
 Humira (adalimumab) Medication Request Form (MRF) for Healthy Indiana Plan (HIP) and Hoosier Healthwise (HHW) FAX TO: (858) 790-7100 c/o MedImpact Healthcare Systems, Inc. Attn: Prior Authorization Department
Humira (adalimumab) Medication Request Form (MRF) for Healthy Indiana Plan (HIP) and Hoosier Healthwise (HHW) FAX TO: (858) 790-7100 c/o MedImpact Healthcare Systems, Inc. Attn: Prior Authorization Department
2.0 Synopsis. Adalimumab R&D/04/118. (For National Authority Use Only) Referring to Part of Dossier: Volume:
 2.0 Synopsis Abbott Laboratories Name of Study Drug: Adalimumab Name of Active Ingredient: Adalimumab Title of Study: Individual Study Table Referring to Part of Dossier: Volume: Page: (For National Authority
2.0 Synopsis Abbott Laboratories Name of Study Drug: Adalimumab Name of Active Ingredient: Adalimumab Title of Study: Individual Study Table Referring to Part of Dossier: Volume: Page: (For National Authority
Targeted Immune Modulators
 Oregon Health Resources Commission Targeted Immune Modulators Subcommittee Report Update #1, April 2007 TIMs Subcommittee Report of March 2006. All revisions are highlighted. Produced by: Health Resources
Oregon Health Resources Commission Targeted Immune Modulators Subcommittee Report Update #1, April 2007 TIMs Subcommittee Report of March 2006. All revisions are highlighted. Produced by: Health Resources
Ontario Public Drug Programs. Inflectra (infliximab) Frequently Asked Questions
 Ontario Public Drug Programs Inflectra (infliximab) Frequently Asked Questions 1. What is the funding status of Inflectra (infliximab)? Effective February 25 2016, Inflectra (infliximab) will be added
Ontario Public Drug Programs Inflectra (infliximab) Frequently Asked Questions 1. What is the funding status of Inflectra (infliximab)? Effective February 25 2016, Inflectra (infliximab) will be added
Cost-effectiveness analysis of biological treatments for rheumatoid arthritis Chiou C F, Choi J, Reyes C M
 Cost-effectiveness analysis of biological treatments for rheumatoid arthritis Chiou C F, Choi J, Reyes C M Record Status This is a critical abstract of an economic evaluation that meets the criteria for
Cost-effectiveness analysis of biological treatments for rheumatoid arthritis Chiou C F, Choi J, Reyes C M Record Status This is a critical abstract of an economic evaluation that meets the criteria for
Efficacy of Concomitant Use of PUVA and Methotrexate in Disease Clearance Time in Plaque Type Psoriasis
 Efficacy of Concomitant Use of PUVA and Methotrexate in Disease Clearance Time in Plaque Type Psoriasis T. Shehzad ( Departments of Dermatology Naval Hospital PNS Shifa, Karachi. ) N. R. Dar ( Departments
Efficacy of Concomitant Use of PUVA and Methotrexate in Disease Clearance Time in Plaque Type Psoriasis T. Shehzad ( Departments of Dermatology Naval Hospital PNS Shifa, Karachi. ) N. R. Dar ( Departments
Cyltezo (adalimumab-adbm) CG-DRUG-64, CG-DRUG-65
 Market DC Cyltezo (adalimumab-adbm) CG-DRUG-64, CG-DRUG-65 Override(s) Prior Authorization Quantity Limit Medications Cyltezo (adalimumab-adbm) 40 mg/0.8 ml prefilled syringe #* ^ Approval Duration 1 year
Market DC Cyltezo (adalimumab-adbm) CG-DRUG-64, CG-DRUG-65 Override(s) Prior Authorization Quantity Limit Medications Cyltezo (adalimumab-adbm) 40 mg/0.8 ml prefilled syringe #* ^ Approval Duration 1 year
Evidence-based Practice Center Systematic Review Protocol
 Evidence-based Practice Center Systematic Review Protocol Project Title: Comparative Effectiveness of Drug Therapy for Psoriatic Arthritis in Adults Update Amendment Date(s) if applicable: (Amendments
Evidence-based Practice Center Systematic Review Protocol Project Title: Comparative Effectiveness of Drug Therapy for Psoriatic Arthritis in Adults Update Amendment Date(s) if applicable: (Amendments
3. Has the patient shown improvement in signs and symptoms of the disease? Y N
 Pharmacy Prior Authorization MERC CARE (MEDICAID) Renflexis (Medicaid) This fax machine is located in a secure location as required by HIPAA regulations. Complete/review information, sign and date. Fax
Pharmacy Prior Authorization MERC CARE (MEDICAID) Renflexis (Medicaid) This fax machine is located in a secure location as required by HIPAA regulations. Complete/review information, sign and date. Fax
Clinical Policy: Secukinumab (Cosentyx) Reference Number: ERX.SPA.165 Effective Date:
 Clinical Policy: (Cosentyx) Reference Number: ERX.SPA.165 Effective Date: 10.01.16 Last Review Date: 11.18 Revision Log See Important Reminder at the end of this policy for important regulatory and legal
Clinical Policy: (Cosentyx) Reference Number: ERX.SPA.165 Effective Date: 10.01.16 Last Review Date: 11.18 Revision Log See Important Reminder at the end of this policy for important regulatory and legal
Clinical Policy: Secukinumab (Cosentyx) Reference Number: CP.PHAR.261 Effective Date: 08/16 Last Review Date: 08/17
 Clinical Policy: (Cosentyx) Reference Number: CP.PHAR.261 Effective Date: 08/16 Last Review Date: 08/17 Line of Business: Medicaid Revision Log See Important Reminder at the end of this policy for important
Clinical Policy: (Cosentyx) Reference Number: CP.PHAR.261 Effective Date: 08/16 Last Review Date: 08/17 Line of Business: Medicaid Revision Log See Important Reminder at the end of this policy for important
Request for Special Authorization Enbrel
 Certain prescription drugs call for a more detailed assessment to help ensure that they represent reasonable treatment. Special Authorization requires that you request approval from Great-West Life for
Certain prescription drugs call for a more detailed assessment to help ensure that they represent reasonable treatment. Special Authorization requires that you request approval from Great-West Life for
Clinical Policy: Ustekinumab (Stelara) Reference Number: CP.PHAR.264
 Clinical Policy: (Stelara) Reference Number: CP.PHAR.264 Effective Date: 08/16 Last Review Date: 05/16 Coding Implications Revision Log See Important Reminder at the end of this policy for important regulatory
Clinical Policy: (Stelara) Reference Number: CP.PHAR.264 Effective Date: 08/16 Last Review Date: 05/16 Coding Implications Revision Log See Important Reminder at the end of this policy for important regulatory
Single Technology Appraisal (STA) Tildrakizumab for treating moderate to severe plaque psoriasis
 Single Technology Appraisal (STA) Tildrakizumab for treating moderate to severe plaque psoriasis Response to consultee and commentator comments on the draft remit and draft scope (pre-referral) Please
Single Technology Appraisal (STA) Tildrakizumab for treating moderate to severe plaque psoriasis Response to consultee and commentator comments on the draft remit and draft scope (pre-referral) Please
Insights from the Clalit Health Services database about Psoriasis Research using BIG DATA Arnon D. Cohen, MD, MPH, PHD
 Insights from the Clalit Health Services database about Psoriasis Research using BIG DATA Arnon D. Cohen, MD, MPH, PHD Siaal Research Center for Family Medicine and Primary Care, Faculty of Health Sciences,
Insights from the Clalit Health Services database about Psoriasis Research using BIG DATA Arnon D. Cohen, MD, MPH, PHD Siaal Research Center for Family Medicine and Primary Care, Faculty of Health Sciences,
chemotherapeutic agents in
 Use of biologics and chemotherapeutic agents in cutaneous emergencies: Focus on lifethreatening forms of psoriasis Alice Bendix Gottlieb MD, PhD Professor of Dermatology New York Medical College Metropolitan
Use of biologics and chemotherapeutic agents in cutaneous emergencies: Focus on lifethreatening forms of psoriasis Alice Bendix Gottlieb MD, PhD Professor of Dermatology New York Medical College Metropolitan
Corporate Medical Policy
 Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: golimumab_simponi 8/2013 2/2018 2/2019 3/2018 Description of Procedure or Service Golimumab (Simponi and
Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: golimumab_simponi 8/2013 2/2018 2/2019 3/2018 Description of Procedure or Service Golimumab (Simponi and
Corporate Medical Policy
 Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: ustekinumab_stelara 02/2010 10/2018 10/2019 10/2018 Description of Procedure or Service Plaque psoriasis
Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: ustekinumab_stelara 02/2010 10/2018 10/2019 10/2018 Description of Procedure or Service Plaque psoriasis
Clinical Policy: Abatacept (Orencia) Reference Number: ERX.SPA.123 Effective Date:
 Clinical Policy: (Orencia) Reference Number: ERX.SPA.123 Effective Date: 10.01.16 Last Review Date: 05.18 Revision Log See Important Reminder at the end of this policy for important regulatory and legal
Clinical Policy: (Orencia) Reference Number: ERX.SPA.123 Effective Date: 10.01.16 Last Review Date: 05.18 Revision Log See Important Reminder at the end of this policy for important regulatory and legal
Pravit Asawanonda, MD, DSc, and Yaowalak Nateetongrungsak, MD Bangkok, Thailand
 Methotrexate plus narrowband UVB phototherapy versus narrowband UVB phototherapy alone in the treatment of plaque-type psoriasis: A randomized, placebo-controlled study Pravit Asawanonda, MD, DSc, and
Methotrexate plus narrowband UVB phototherapy versus narrowband UVB phototherapy alone in the treatment of plaque-type psoriasis: A randomized, placebo-controlled study Pravit Asawanonda, MD, DSc, and
STELARA DATA SHEET NAME OF THE MEDICINE DESCRIPTION V L C L V H C H 1 C H 2 C H 3. Fab. F(ab)' 2. hinge
 DATA SHEET NAME OF THE MEDICINE Ustekinumab (rmc). CAS Registry Number: 815610-63-0. DESCRIPTION (ustekinumab) is a human IgG1kappa monoclonal antibody with an approximate molecular weight of 148,600 daltons.
DATA SHEET NAME OF THE MEDICINE Ustekinumab (rmc). CAS Registry Number: 815610-63-0. DESCRIPTION (ustekinumab) is a human IgG1kappa monoclonal antibody with an approximate molecular weight of 148,600 daltons.
Clinical Policy: Ustekinumab (Stelara) Reference Number: ERX.SPMN.167
 Clinical Policy: (Stelara) Reference Number: ERX.SPMN.167 Effective Date: 10/16 Last Review Date: 12/16 Coding Implications Revision Log See Important Reminder at the end of this policy for important regulatory
Clinical Policy: (Stelara) Reference Number: ERX.SPMN.167 Effective Date: 10/16 Last Review Date: 12/16 Coding Implications Revision Log See Important Reminder at the end of this policy for important regulatory
See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: Infliximab (Remicade), Infliximab-dyyb (Inflectra), and Infliximab-abda (Renflexis) Reference Number: ERX.SPA.160 Effective Date: 10.01.16 Last Review Date: 05.18 Revision Log See Important
Clinical Policy: Infliximab (Remicade), Infliximab-dyyb (Inflectra), and Infliximab-abda (Renflexis) Reference Number: ERX.SPA.160 Effective Date: 10.01.16 Last Review Date: 05.18 Revision Log See Important
Ustekinumab (Stelara) for psoriatic arthritis second line after disease modifying anti rheumatic drugs (DMARDs)
 Ustekinumab (Stelara) for psoriatic arthritis second line after disease modifying anti rheumatic drugs (DMARDs) January 2010 This technology summary is based on information available at the time of research
Ustekinumab (Stelara) for psoriatic arthritis second line after disease modifying anti rheumatic drugs (DMARDs) January 2010 This technology summary is based on information available at the time of research
It is estimated that about 26,000 new cases of
 Focus on CME at Dalhousie University Set On Soothing Psoriasis A. H. Murray, MD, FRCP(C) Presented at the 76th Annual Dalhousie Refresher Course It is estimated that about 26,000 new cases of psoriasis
Focus on CME at Dalhousie University Set On Soothing Psoriasis A. H. Murray, MD, FRCP(C) Presented at the 76th Annual Dalhousie Refresher Course It is estimated that about 26,000 new cases of psoriasis
