Independent Health Facilities
|
|
|
- Jemima Gordon
- 5 years ago
- Views:
Transcription
1 Independent Health Facilities Assessment Protocol for Pulmonary Function Studies INSTRUCTIONS: Please complete ( ) the attached protocol during the assessment. Ensure that all the questions have been answered, please do not leave any questions blank. A space has been provided at the bottom of each section for your comments.
2 FACILITY NAME/NO.: Date: STAFF s s with Recommendations 1. Do the physicians providing interpreting services meet the qualifications as outlined on Pg 5 of the CPPs&FS? 2. Do the medical staff ensure: The safe, accurate and reliable performance of those tests which the physician will be interpreting? Prompt communication of test results to the referring physician? Assistance is provided to the Quality Advisor to perform other responsibilities that may be assigned to a physician who has had appropriate training? Preparation of written reports for the Quality Advisor detailing any concerns the physician may have as to the safe and proper conduct of the facility? 3. Do the technologists meet the qualifications as outlined on pg 6 of the CPPs & FS? 4. Is there a Chief Technologist appointed? If appointed, does the Chief Technologist meet the qualifications as outlined on pg 6 of the CPPs & FS? 5. Do the Technologists: Ensure they are current with the changing technical trends in the cardiopulmonary field by attending conferences, meetings or other forms of continuing education and reading current relevant literature? Arrange patient appointments and staff work schedules? Distribute to referring physicians and agencies the test requisition and the completed test reports? Maintain proper policies & procedures? Maintain records of equipment calibration, maintenance and repair procedures? Maintain copies of test observations and reports? N/A Comments
3 STAFF s s with Recommendations Maintain administrative records? Ensure that safety policies and the equipment and facilities necessary for their implementation are in place and in working order? Ensure the safe and reliable performance of tests? Observe and follow documented infection control measures? Maintain all necessary facility supplies? Ensure safe and accurate performance of testing procedures? Implement current policies & procedures? Provide assistance to the Quality Advisor? 6. Are written job descriptions available for all staff? 7. Are continuing professional development activities documented by technologists and physicians? 8. Is all staff certified and current in BCLS? N/A Comments POLICIES AND PROCEDURES s s with Recommendations Are there written policies/procedures guidelines for the following? Description of the proper methodology for performing each test offered by the facility, including criteria to ensure that the results obtained is reliable? The predicted normal values for each test offered by the facility including the references from which these values were obtained? Procedures to be followed to maintain proper infection control/body substance precautions as described in the CPSO Guidelines Infection Control in the Physicians Office 2004? Procedures to be followed for each test to ensure that the test is performed only on those patients for whom it can be performed safely? meet N/A Comments Independent Health Facilities Pulmonary Function Protocol 3
4 POLICIES AND PROCEDURES CONT D s s with Recommendations General safety precautions to be followed in operating the facility and performing the tests to prevent adverse health effects from occurring in the facility? Specific first aid measures to be followed in the event of adverse health effect including a description of the arrangement made to transfer patients to an acute care facility when required? A list of safety equipment and medications with expiry dates to be maintained by the facility? Routine maintenance procedures to be followed to ensure reliable and accurate testing equipment? Documentation of regular routine calibration and validation measures on test equipment? Patient consent based on scope of practice in the facility in compliance with the Health Care Consent Act? Latex anaphylaxis? Material Safety Data Sheets (MSDS) current within 3 years for all chemicals maintained in the facility A copy of the Workplace Hazardous Materials Information Systems (WHMIS) manual? Fire safety including fire prevention; classes of fires and extinguishers, steps on discovery of fires, plans for reporting fires, fire evacuation plans and maps? 2. Are policies/procedures reviewed annually and dated accordingly? 3. Is there evidence that the staff know the policy or where to look for the policy if needed? 4. Is there evidence that the policy and procedures are implemented? *Assessor may select staff at random to ask of knowledge or use of policies meet N/A Independent Health Facilities Pulmonary Function Protocol 4
5 QUALITY CONTROL ACTIVITIES s s with Recommendations 1. Are Quality Control Activities implemented: Oxygen Saturation by Oximetry Calibration performed according to manufacturer s recommendations? Biological controls monitored and documented at least monthly? Carbon Monoxide Diffusing Capacity Spirometer volume calibrated to an accuracy of ± 3.5% of the syringe volume (3 ±.105L)? For peumotachometers, varying flow rates (2-12 L/sec) must be injectedusing a 3 L syringe to check for the same recovered volume accuracy range For continuous real-time gas analysis, a 2-point calibration of the infrared analyzer using zero and test gas concentration to within ± 0.5% of the test gas prior to each test For gas chromatography analysis, separation of the test gas concentration into its component gases and detection by a thermal conductivity analyzer to g single chromatograph tracing with a Ne and CO peak is required before each test. For infrared CO analyzers with electronic linearization, and He analyzers, a linear response is established for each test (i.e., the analyzer is adjusted to zero and full scale using the test gas concentration). Linearity check using a minimum of 3 serial gases or primary standards (with a maximum error of ±0.0015%) is completed every 3 months. Time is checked for accuracy within ±1% over 10 sec every 3 months. Biological controls monitored and documented at least monthly? FRC by Closed Circuit Helium Dilution Spirometer calibrated every three months over the full range of the instrument and accurate over 7 L to ± 3.5% of the volume added. A daily 3 L volume calibration (verification) and leak check is performed. Independent Health Facilities Pulmonary Function Protocol 5 N/A Comments
6 QUALITY CONTROL ACTIVITIES CONT D s s with Recommendations Two point (zero to full scale) calibration of the helium analyzer performed daily Biological controls monitored and documented at least monthly FRC by Nitrogen Washout Spirometer calibrated every three months over the full range of the instrument and accurate over 7 L to ± 3.5% of the volume added. Daily 3 L volume check and leak check Accuracy and linearity of the N2 analyzer checked before each patient by performing a 2 point calibration using 0 and 80% Nitrogen. A 3-point linear check is required initially and every 6 months thereafter (0%, 40% and 80% Nitrogen). If Nitrogen is derived from deductions of CO 2 and O 2, the respective analyzers must be checked according to the manufacturer. The pneumotach must be calibrated using room air for the inhaled check and O 2 for the exhaled check. Biological controls monitored and documented at least monthly FRC and Airways Resistance by Body Plethysmography Transducers are calibrated initially and checked every six months for noise and drift interference FRC and Airways Resistance by Body Plethysmography Box pressure transducer calibrated using a cc piston pump at 2 Hz Mouth pressure transducer calibrated using a water or mercury manometer and rotameter for flow Linearity is checked over the entire range of use Physical calibration is performed monthly Biological controls monitored and documented at least monthly Does N/A Independent Health Facilities Pulmonary Function Protocol 6
7 QUALITY CONTROL ACTIVITIES CONT D s s with Recommendations Non-specific Bronchial Provocative Test A new nebulizer s output must be determined by full calibration prior to use. The corresponding flow rate required to deliver the appropriate output must be recorded and used consistently. Checks of nebulizer output every 6 months need only test the output at that flow rate. If it varies by more than 10% during verification, a full calibration must be performed. Spirometer volume calibrated to an accuracy of ± 3.5% of the syringe volume (3 ±.105L). For peumotachometers, varying flow rates (2-12 L/sec) must be injected using a 3 L syringe to check for the same recovered volume accuracy range. Methacholine/provomethacholine and histamine stored according to the parameters and standards (refrigerated at 4 degrees Celsius for no more than 3 months). Stage 1 Exercise Testing Are mechanical bicycles tested for accuracy using known weights at least yearly? Are electronic bicycles calibrated with physical balance such as a torque calibrator at least yearly? Is the treadmill s belt velocity and grade tested yearly? Is each gas analyzer calibrated (2 point with range 0-7% for CO 2 and 0-100% for 0 2 with accuracy of ± 0.03%) prior to use? Three point calibration for O 2 analyzers using 0% O 2, 16% O 2, and 100% O 2 with an accuracy of ± 0.03% and five point calibration for CO2 between0-7% quarterly If the system has a linearizing algorithm a 2 point gas calibration is adequate Spirometer volume calibrated to an accuracy of ± 3.5% of the syringe volume (3 ±.105L)? Does N/A Independent Health Facilities Pulmonary Function Protocol 7
8 QUALITY CONTROL ACTIVITIES CONT D s s with Recommendations For peumotachometers, varying flow rates (2-12 L/sec) must be injected using a 3 L syringe to check for the same recovered volume accuracy range Is the respiratory circuit checked for leaks weekly Are biologic controls performed at least quarterly? Maximal Inspiratory and Expiratory Pressure Check the calibration by applying 100 cmh20 for both positive and negative pressures daily. The measured reading should be within 5% of the expected value. Is at least a 2 point calibration done with a water or mercury manometer over the pressure range used at least quarterly? ARTERIAL BLOOD GASES Is there a one point calibration performed every 30 minutes or at least prior to the patient sample? For ph is a one-point calibration is performed using the calibration solution with normal ph to determine the status of the electrode? Is a two-point calibration is performed every 8 hours using 2 ph buffer solutions (e.g., 7.4 and 6.8) to determine the sensitivity of the electrode? For PCO 2 is a one-point calibration is performed using a precision CO 2 gas mixture (e.g. 5%) to determine the status of the electrode? Is a two-point calibration is performed using 2 precise mixtures of CO 2 concentrations (e.g., 5% and 10%) to determine the sensitivity of the electrode? For PO 2 is a zero point value performed using 0% O 2? Is a one-point calibration performed using one O 2 concentration (e.g., 20%) to determine the sensitivity? Is the spectrophotometer calibrated over the fixed wavelengths using a water sample to determine the zero point and the drift? Is a thg calibration performed every 3 months? Does N/A Comments Independent Health Facilities Pulmonary Function Protocol 8
9 ENVIRONMENT, EQUIPMENT AND SUPPLIES s s with Recommendations 1. Is the facility clean? 2. Is all equipment used in the facility CSA approved? 3. Are electrical cords, plugs, outlets routinely checked for damage? 4. Are gas cylinders Properly labeled and secured to a wall or placed in a stationary cart whether or not they are in use? 5. Are the physicians and staff familiar with the current recommendations of the ATS/ERS regarding pulmonary function standards?* 6. Is mandatory compliance with the minimal recommendations for spirometry, lung diffusion and lung volumes as published in 2005 by the joint publications of the ATS/ERS Task Force be adhered to by staff? 7. Are the physicians and staff familiar with the Clinical Practice Parameters and Facility Standards? 8. In rooms where pharmacological challenge testing is done: Is there adequate ventilation? Are filters used on the expiratory circuit of the mouthpiece apparatus? 9. Are all tubings and valves sterilized after each use? 10. Are clean mouthpieces and noseclips used for each patient? 11. Are disposable bacterial filters used unless the circuitry is changed after each patient? 12. Is equipment that cannot be subjected to heat or chemicals sterilized using a cold sterilizing solution? 13. Does the facility follow manufacturer s recommendations for disinfecting and sterilization equipment? 14. Is the following equipment available in facilities performing exercise testing and bronchoprovocative testing? Sphygmomanometer and stethoscope Wheelchair O 2 source with mask Independent Health Facilities Pulmonary Function Protocol 9 N/A Comments
10 ENVIRONMENT, EQUIPMENT AND SUPPLIES s s with Recommendations Connective tubing Resuscitation equipment Airway management equipment Appropriate drugs 15. Are all resuscitation equipment, drugs and sterile equipment checked monthly for expiry dates? Is this activity documented? 16. Are fire evacuation plans/maps posted throughout the facility? 17. Are fire extinguishers easily accessible and checked each month and replaced if outdated or used? 18. Is the fire evacuation plan practiced periodically? Is fire safety plan activities documented? N/A Independent Health Facilities Pulmonary Function Protocol 10
11 RECORDS s s with Recommendations 1. Are written requisitions completed for all pulmonary function tests? 2. When an order for a test(s) has been dictated over the telephone, is the following written on the requisition by the person to whom the order was dictated? Test(s) Name of requisitioning physician Date and time of order Signature of person receiving the order 3. Does the report contain the following information? Personal data sufficient to identify the patient, the patient s age, height and weight; the referring and reporting physicians, the name of the facility performing the test and the test date? The technologist s comments as to the reliability of the patient s performance during the test, where necessary A summary of the original data obtained, the calculations made during the test and graphic records The reporting physician s interpretation of the original data as well as where appropriate comments as to the relevance of the results to the patient s presenting problem or suggestions as to patient management arising from the results 4. Does the facility maintain a log of activities that include but are not limited to: All maintenance, repair and calibration procedures performed (including biologic controls), results obtained, and where appropriate, corrective action taken 5. Are copies of all reports retained with the requisition and original data for the period of time as specified by the IHFA Regulations? N/A Comments Independent Health Facilities Pulmonary Function Protocol 11
12 QUALITY MANAGEMENT PROGRAM s s with Recommendations 1. Has a Quality Advisory Committee been established as outlined in the IHF Act? 2. Do quality management activities include the following? Establishing a mechanism for periodic review of selected original data for all types of tests performed by the facility to establish that tests are properly performed and reliable Regular review of calibration and validation data on equipment, noting any deviations from accepted norms and recording corrective action taken, if required Reporting and review of all adverse health effects occurring during testing, the action taken, the outcome achieved and documentation of recommendations made for future prevention, if required Establishing a mechanism for periodic review of the pattern of tests that the facility is requested to perform. This review includes but is not limited to: Whether the tests ordered are appropriate to the presenting clinic problem Whether effective use of the facility is being made in assessing these various clinical problems Whether results of these reviews are used to further educate referring physicians and facility staff? Periodic review of reports issued to ensure that: Test results are issued within two weeks accurately? Physicians interpret the studies accurately? Periodic surveys of patients to determine their satisfaction with the services Periodic surveys of referring physicians to determine their satisfaction and whether results have influenced patient outcomes. Independent Health Facilities Pulmonary Function Protocol 12 N/A Comments
13 QUALITY MANAGEMENT PROGRAM s s with Recommendations 3. Do staff participate in planning strategies to overcome any deficiencies and to continuously improve the services provided? 4. Based on the information above, has the facility established a quality management program appropriate for its volume and types of services provided? N/A QUALITY ADVISOR s s with Recommendations 1. Is there a designated Quality Advisor? 2. Is there a formal written agreement for the Quality Advisor to advise the facility owner/operator with respect to the quality of services provided? 3. Does the Quality Advisor fulfill the responsibilities of the appointed role which include but are not limited to the following: The qualifications and work performed by other physicians employed in the facility? The qualifications and work performed by pulmonary function technologists employed in the facility and where the nature and size of the facility warrants the appointment of a chief technologist and office support staff? The accuracy and reliability of the equipment used in performing pulmonary function tests? Whether the tests performed by the facility are done accurately and reliably? Whether the tests performed by the facility are conducted safely, and whether procedures and equipment are available within the facility to manage any adverse reaction that may occur? The appropriate design, staffing, and equipping of the facility so as to ensure patient comfort and safety and the proper performance and reporting of pulmonary function tests? The proper design of pulmonary function test requisitions and reports? The maintenance of all necessary records? Independent Health Facilities Pulmonary Function Protocol 13 N/A Comments
14 QUALITY ADVISOR s s with Recommendations Whether the pulmonary function test results are properly interpreted and promptly communicated to the referring physician? The establishment of a quality assurance program for the facility, including matters related to maintenance of a safe work environment? 4. Based on the information above, has the Quality Advisory fulfilled his/her role as Quality Advisor for the facility? N/A Independent Health Facilities Pulmonary Function Protocol 14
15 FACILITY NAME/NO. PATIENT IDENTIFIER (e: either a chart # or patient initials) Date of Test: Tests Ordered by Referring Physician (please check as applicable) : Oxygen Saturation by Oximetry (J323, J332, J334) Non-specific Bronchial Provocative Test (J333) Carbon Monoxide Diffusing Capacity (J309, 310) MIPs & MEPs (J340) Function Residual Capacity (J311, 307) Stage 1 Exercise Testing(J315, E450, E451) Exercise Challenge Testing for Asthma (J330) Other TESTING GUIDELINES s s with 1. Does the requisition include: patient demographics; clinical information; working diagnosis or reason for test; referring physician s name and signature 2. Are the clinical indications for the procedure in compliance with those listed in the parameters? 3. Are these studies directly related to the patient s clinical condition? 4. Are these confirmed by the information documented in the patient s chart? 5. Was informed consent obtained from the patient based on the test being performed? 6. Are the pre-requisites met: Is spirometry properly performed? Are spirometry results known prior to the test? 7. Are the test results reproducible as outlined in the parameters and standards for: J323, J332, J334 J333 J309, J310 J340 J311, J307 J315, E450, E451 J For J309,310 -Do the acceptable tests reported meet ATS standards for reproducibility; collection volumes; breath hold time; inspiratory volume; inspiratory & expiratory times? 8. For J315, J330, E450, E451 Is a resting ECG done and results known? (exception young healthy individuals) 9. For J330, J315, E450, E451: Is blood pressure taken at 1-2 minute intervals during exercise & recovery from exercise? Are flow-volume loops pre-and post exercise properly performed and reported.? Is a physician in attendance at all times during the test? Independent Health Facilities Pulmonary Function Protocol 15
16 REPORTING GUIDELINES s s with 10. Does the report include the following: Patient demographics Interpretation of the data, and where appropriate comments as to the relevance of the results to the patient s presenting problem For J323, J332, J334 Interpretation of the data taking into account the limitations of the study For J333 Results of baseline spirometry Summary of the data and calculations made If the study is positive, provision of some quantification of hyperactivity For J309, J310 Technologist s comments as to the reliability of patient s performance where necessary Summary of the original data and calculations made during test and where feasible, of the graphical records. For J340 Results of baseline spirometry Technologists comments as to the reliability of patient s performance where necessary Comparison of patient measurement to predicted normal value as to patient management For J311, J307 Results of baseline spirometry Comparison of patient measurement to predicted normal values In the case of abnormal results, type of ventilatory abnormality expected For J330, J315, E450,E451 Description of the exercise study protocol completed or when and why the test was stopped prematurely. Symptoms reported by patients Presence or absence of clinically significant changes in BP or heart rate or rhythm Evaluation of exercise performance and the ventilatory response to exercise Precise identification of the factor(s) that limit(s) exercise performance Results of baseline spirometry and changes seen Comments: Independent Health Facilities Pulmonary Function Protocol 16
17 FACILITY NAME/NO. PULMONARY FUNCTION STUDIES CHART REVIEW FORM PATIENT IDENTIFIER (e: either a chart # or patient initials) Date of Test: Tests Ordered by Referring Physician (please check as applicable) : Oxygen Saturation by Oximetry (J323, J332, J334) Non-specific Bronchial Provocative Test (J333) Carbon Monoxide Diffusing Capacity (J309, 310) MIPs & MEPs (J340) Function Residual Capacity (J311, 307) Stage 1 Exercise Testing(J315, E450, E451) Exercise Challenge Testing for Asthma (J330) Other TESTING GUIDELINES s s with 1. Does the requisition include: patient demographics; clinical information; working diagnosis or reason for test; referring physician s name and signature 2. Are the clinical indications for the procedure in compliance with those listed in the parameters? 3. Are these studies directly related to the patient s clinical condition? 4. Are these confirmed by the information documented in the patient s chart? 5. Was informed consent obtained from the patient based on the test being performed? 6. Are the pre-requisites met: Is spirometry properly performed? Are spirometry results known prior to the test? 8. Are the test results reproducible as outlined in the parameters and standards for: J323, J332, J334 J333 J309, J310 J340 J311, J307 J315, E450, E451 J For J309,310 -Do the acceptable tests reported meet ATS standards for reproducibility; collection volumes; breath hold time; inspiratory volume; inspiratory & expiratory times? 8. For J315, J330, E450, E451 Is a resting ECG done and results known? (exception young healthy individuals) 9. For J330, J315, E450, E451: Is blood pressure taken at 1-2 minute intervals during exercise & recovery from exercise? Are flow-volume loops pre-and post exercise properly performed and reported.? Is a physician in attendance at all times during the test? Independent Health Facilities Pulmonary Function Protocol 17
18 REPORTING GUIDELINES s s with 10. Does the report include the following: Patient demographics Interpretation of the data, and where appropriate comments as to the relevance of the results to the patient s presenting problem For J323, J332, J334 Interpretation of the data taking into account the limitations of the study For J333 Results of baseline spirometry Summary of the data and calculations made If the study is positive, provision of some quantification of hyperactivity For J309, J310 Technologist s comments as to the reliability of patient s performance where necessary Summary of the original data and calculations made during test and where feasible, of the graphical records. For J340 Results of baseline spirometry Technologists comments as to the reliability of patient s performance where necessary Comparison of patient measurement to predicted normal value as to patient management For J311, J307 Results of baseline spirometry Comparison of patient measurement to predicted normal values In the case of abnormal results, type of ventilatory abnormality expected For J330, J315, E450,E451 Description of the exercise study protocol completed or when and why the test was stopped prematurely. Symptoms reported by patients Presence or absence of clinically significant changes in BP or heart rate or rhythm Evaluation of exercise performance and the ventilatory response to exercise Precise identification of the factor(s) that limit(s) exercise performance Results of baseline spirometry and changes seen Comments: Independent Health Facilities Pulmonary Function Protocol 18
19 FACILITY NAME/NO. PULMONARY FUNCTION STUDIES CHART REVIEW FORM PATIENT IDENTIFIER (e: either a chart # or patient initials) Date of Test: Tests Ordered by Referring Physician (please check as applicable) : Oxygen Saturation by Oximetry (J323, J332, J334) Non-specific Bronchial Provocative Test (J333) Carbon Monoxide Diffusing Capacity (J309, 310) MIPs & MEPs (J340) Function Residual Capacity (J311, 307) Stage 1 Exercise Testing(J315, E450, E451) Exercise Challenge Testing for Asthma (J330) Other TESTING GUIDELINES s s with 1. Does the requisition include: patient demographics; clinical information; working diagnosis or reason for test; referring physician s name and signature 2. Are the clinical indications for the procedure in compliance with those listed in the parameters? 3. Are these studies directly related to the patient s clinical condition? 4. Are these confirmed by the information documented in the patient s chart? 5. Was informed consent obtained from the patient based on the test being performed? 6. Are the pre-requisites met: Is spirometry properly performed? Are spirometry results known prior to the test? 9. Are the test results reproducible as outlined in the parameters and standards for: J323, J332, J334 J333 J309, J310 J340 J311, J307 J315, E450, E451 J For J309,310 -Do the acceptable tests reported meet ATS standards for reproducibility; collection volumes; breath hold time; inspiratory volume; inspiratory & expiratory times? 8. For J315, J330, E450, E451 Is a resting ECG done and results known? (exception young healthy individuals) 9. For J330, J315, E450, E451: Is blood pressure taken at 1-2 minute intervals during exercise & recovery from exercise? Are flow-volume loops pre-and post exercise properly performed and reported.? Is a physician in attendance at all times during the test? Independent Health Facilities Pulmonary Function Protocol 19
20 REPORTING GUIDELINES s s with 10. Does the report include the following: Patient demographics Interpretation of the data, and where appropriate comments as to the relevance of the results to the patient s presenting problem For J323, J332, J334 Interpretation of the data taking into account the limitations of the study For J333 Results of baseline spirometry Summary of the data and calculations made If the study is positive, provision of some quantification of hyperactivity For J309, J310 Technologist s comments as to the reliability of patient s performance where necessary Summary of the original data and calculations made during test and where feasible, of the graphical records. For J340 Results of baseline spirometry Technologists comments as to the reliability of patient s performance where necessary Comparison of patient measurement to predicted normal value as to patient management For J311, J307 Results of baseline spirometry Comparison of patient measurement to predicted normal values In the case of abnormal results, type of ventilatory abnormality expected For J330, J315, E450,E451 Description of the exercise study protocol completed or when and why the test was stopped prematurely. Symptoms reported by patients Presence or absence of clinically significant changes in BP or heart rate or rhythm Evaluation of exercise performance and the ventilatory response to exercise Precise identification of the factor(s) that limit(s) exercise performance Results of baseline spirometry and changes seen Comments: Independent Health Facilities Pulmonary Function Protocol 20
21 FACILITY NAME/NO. PULMONARY FUNCTION STUDIES CHART REVIEW FORM PATIENT IDENTIFIER (e: either a chart # or patient initials) Date of Test: Tests Ordered by Referring Physician (please check as applicable) : Oxygen Saturation by Oximetry (J323, J332, J334) Non-specific Bronchial Provocative Test (J333) Carbon Monoxide Diffusing Capacity (J309, 310) MIPs & MEPs (J340) Function Residual Capacity (J311, 307) Stage 1 Exercise Testing(J315, E450, E451) Exercise Challenge Testing for Asthma (J330) Other TESTING GUIDELINES s s with 1. Does the requisition include: patient demographics; clinical information; working diagnosis or reason for test; referring physician s name and signature 2. Are the clinical indications for the procedure in compliance with those listed in the parameters? 3. Are these studies directly related to the patient s clinical condition? 4. Are these confirmed by the information documented in the patient s chart? 5. Was informed consent obtained from the patient based on the test being performed? 6. Are the pre-requisites met: Is spirometry properly performed? Are spirometry results known prior to the test? 10. Are the test results reproducible as outlined in the parameters and standards for: J323, J332, J334 J333 J309, J310 J340 J311, J307 J315, E450, E451 J For J309,310 -Do the acceptable tests reported meet ATS standards for reproducibility; collection volumes; breath hold time; inspiratory volume; inspiratory & expiratory times? 8. For J315, J330, E450, E451 Is a resting ECG done and results known? (exception young healthy individuals) 9. For J330, J315, E450, E451: Is blood pressure taken at 1-2 minute intervals during exercise & recovery from exercise? Are flow-volume loops pre-and post exercise properly performed and reported.? Is a physician in attendance at all times during the test? Independent Health Facilities Pulmonary Function Protocol 21
22 REPORTING GUIDELINES s s with 10. Does the report include the following: Patient demographics Interpretation of the data, and where appropriate comments as to the relevance of the results to the patient s presenting problem For J323, J332, J334 Interpretation of the data taking into account the limitations of the study For J333 Results of baseline spirometry Summary of the data and calculations made If the study is positive, provision of some quantification of hyperactivity For J309, J310 Technologist s comments as to the reliability of patient s performance where necessary Summary of the original data and calculations made during test and where feasible, of the graphical records. For J340 Results of baseline spirometry Technologists comments as to the reliability of patient s performance where necessary Comparison of patient measurement to predicted normal value as to patient management For J311, J307 Results of baseline spirometry Comparison of patient measurement to predicted normal values In the case of abnormal results, type of ventilatory abnormality expected For J330, J315, E450,E451 Description of the exercise study protocol completed or when and why the test was stopped prematurely. Symptoms reported by patients Presence or absence of clinically significant changes in BP or heart rate or rhythm Evaluation of exercise performance and the ventilatory response to exercise Precise identification of the factor(s) that limit(s) exercise performance Results of baseline spirometry and changes seen Comments: Independent Health Facilities Pulmonary Function Protocol 22
23 FACILITY NAME/NO. PULMONARY FUNCTION STUDIES CHART REVIEW FORM PATIENT IDENTIFIER (e: either a chart # or patient initials) Date of Test: Tests Ordered by Referring Physician (please check as applicable) : Oxygen Saturation by Oximetry (J323, J332, J334) Non-specific Bronchial Provocative Test (J333) Carbon Monoxide Diffusing Capacity (J309, 310) MIPs & MEPs (J340) Function Residual Capacity (J311, 307) Stage 1 Exercise Testing(J315, E450, E451) Exercise Challenge Testing for Asthma (J330) Other TESTING GUIDELINES s s with 1. Does the requisition include: patient demographics; clinical information; working diagnosis or reason for test; referring physician s name and signature 2. Are the clinical indications for the procedure in compliance with those listed in the parameters? 3. Are these studies directly related to the patient s clinical condition? 4. Are these confirmed by the information documented in the patient s chart? 5. Was informed consent obtained from the patient based on the test being performed? 6. Are the pre-requisites met: Is spirometry properly performed? Are spirometry results known prior to the test? 11. Are the test results reproducible as outlined in the parameters and standards for: J323, J332, J334 J333 J309, J310 J340 J311, J307 J315, E450, E451 J For J309,310 -Do the acceptable tests reported meet ATS standards for reproducibility; collection volumes; breath hold time; inspiratory volume; inspiratory & expiratory times? 8. For J315, J330, E450, E451 Is a resting ECG done and results known? (exception young healthy individuals) 9. For J330, J315, E450, E451: Is blood pressure taken at 1-2 minute intervals during exercise & recovery from exercise? Are flow-volume loops pre-and post exercise properly performed and reported.? Is a physician in attendance at all times during the test? Independent Health Facilities Pulmonary Function Protocol 23
24 REPORTING GUIDELINES s s with 10. Does the report include the following: Patient demographics Interpretation of the data, and where appropriate comments as to the relevance of the results to the patient s presenting problem For J323, J332, J334 Interpretation of the data taking into account the limitations of the study For J333 Results of baseline spirometry Summary of the data and calculations made If the study is positive, provision of some quantification of hyperactivity For J309, J310 Technologist s comments as to the reliability of patient s performance where necessary Summary of the original data and calculations made during test and where feasible, of the graphical records. For J340 Results of baseline spirometry Technologists comments as to the reliability of patient s performance where necessary Comparison of patient measurement to predicted normal value as to patient management For J311, J307 Results of baseline spirometry Comparison of patient measurement to predicted normal values In the case of abnormal results, type of ventilatory abnormality expected For J330, J315, E450,E451 Description of the exercise study protocol completed or when and why the test was stopped prematurely. Symptoms reported by patients Presence or absence of clinically significant changes in BP or heart rate or rhythm Evaluation of exercise performance and the ventilatory response to exercise Precise identification of the factor(s) that limit(s) exercise performance Results of baseline spirometry and changes seen Comments: Independent Health Facilities Pulmonary Function Protocol 24
25 FACILITY NAME/NO. PULMONARY FUNCTION STUDIES CHART REVIEW FORM PATIENT IDENTIFIER (e: either a chart # or patient initials) Date of Test: Tests Ordered by Referring Physician (please check as applicable) : Oxygen Saturation by Oximetry (J323, J332, J334) Non-specific Bronchial Provocative Test (J333) Carbon Monoxide Diffusing Capacity (J309, 310) MIPs & MEPs (J340) Function Residual Capacity (J311, 307) Stage 1 Exercise Testing(J315, E450, E451) Exercise Challenge Testing for Asthma (J330) Other TESTING GUIDELINES s s with 1. Does the requisition include: patient demographics; clinical information; working diagnosis or reason for test; referring physician s name and signature 2. Are the clinical indications for the procedure in compliance with those listed in the parameters? 3. Are these studies directly related to the patient s clinical condition? 4. Are these confirmed by the information documented in the patient s chart? 5. Was informed consent obtained from the patient based on the test being performed? 6. Are the pre-requisites met: Is spirometry properly performed? Are spirometry results known prior to the test? 12. Are the test results reproducible as outlined in the parameters and standards for: J323, J332, J334 J333 J309, J310 J340 J311, J307 J315, E450, E451 J For J309,310 -Do the acceptable tests reported meet ATS standards for reproducibility; collection volumes; breath hold time; inspiratory volume; inspiratory & expiratory times? 8. For J315, J330, E450, E451 Is a resting ECG done and results known? (exception young healthy individuals) 9. For J330, J315, E450, E451: Is blood pressure taken at 1-2 minute intervals during exercise & recovery from exercise? Are flow-volume loops pre-and post exercise properly performed and reported.? Is a physician in attendance at all times during the test? Independent Health Facilities Pulmonary Function Protocol 25
26 REPORTING GUIDELINES s s with 10. Does the report include the following: Patient demographics Interpretation of the data, and where appropriate comments as to the relevance of the results to the patient s presenting problem For J323, J332, J334 Interpretation of the data taking into account the limitations of the study For J333 Results of baseline spirometry Summary of the data and calculations made If the study is positive, provision of some quantification of hyperactivity For J309, J310 Technologist s comments as to the reliability of patient s performance where necessary Summary of the original data and calculations made during test and where feasible, of the graphical records. For J340 Results of baseline spirometry Technologists comments as to the reliability of patient s performance where necessary Comparison of patient measurement to predicted normal value as to patient management For J311, J307 Results of baseline spirometry Comparison of patient measurement to predicted normal values In the case of abnormal results, type of ventilatory abnormality expected For J330, J315, E450,E451 Description of the exercise study protocol completed or when and why the test was stopped prematurely. Symptoms reported by patients Presence or absence of clinically significant changes in BP or heart rate or rhythm Evaluation of exercise performance and the ventilatory response to exercise Precise identification of the factor(s) that limit(s) exercise performance Results of baseline spirometry and changes seen Comments: Independent Health Facilities Pulmonary Function Protocol 26
27 FACILITY NAME/NO. PULMONARY FUNCTION STUDIES CHART REVIEW FORM PATIENT IDENTIFIER (e: either a chart # or patient initials) Date of Test: Tests Ordered by Referring Physician (please check as applicable) : Oxygen Saturation by Oximetry (J323, J332, J334) Non-specific Bronchial Provocative Test (J333) Carbon Monoxide Diffusing Capacity (J309, 310) MIPs & MEPs (J340) Function Residual Capacity (J311, 307) Stage 1 Exercise Testing(J315, E450, E451) Exercise Challenge Testing for Asthma (J330) Other TESTING GUIDELINES s s with 1. Does the requisition include: patient demographics; clinical information; working diagnosis or reason for test; referring physician s name and signature 2. Are the clinical indications for the procedure in compliance with those listed in the parameters? 3. Are these studies directly related to the patient s clinical condition? 4. Are these confirmed by the information documented in the patient s chart? 5. Was informed consent obtained from the patient based on the test being performed? 6. Are the pre-requisites met: Is spirometry properly performed? Are spirometry results known prior to the test? 13. Are the test results reproducible as outlined in the parameters and standards for: J323, J332, J334 J333 J309, J310 J340 J311, J307 J315, E450, E451 J For J309,310 -Do the acceptable tests reported meet ATS standards for reproducibility; collection volumes; breath hold time; inspiratory volume; inspiratory & expiratory times? 8. For J315, J330, E450, E451 Is a resting ECG done and results known? (exception young healthy individuals) 9. For J330, J315, E450, E451: Is blood pressure taken at 1-2 minute intervals during exercise & recovery from exercise? Are flow-volume loops pre-and post exercise properly performed and reported.? Is a physician in attendance at all times during the test? Independent Health Facilities Pulmonary Function Protocol 27
28 REPORTING GUIDELINES s s with 10. Does the report include the following: Patient demographics Interpretation of the data, and where appropriate comments as to the relevance of the results to the patient s presenting problem For J323, J332, J334 Interpretation of the data taking into account the limitations of the study For J333 Results of baseline spirometry Summary of the data and calculations made If the study is positive, provision of some quantification of hyperactivity For J309, J310 Technologist s comments as to the reliability of patient s performance where necessary Summary of the original data and calculations made during test and where feasible, of the graphical records. For J340 Results of baseline spirometry Technologists comments as to the reliability of patient s performance where necessary Comparison of patient measurement to predicted normal value as to patient management For J311, J307 Results of baseline spirometry Comparison of patient measurement to predicted normal values In the case of abnormal results, type of ventilatory abnormality expected For J330, J315, E450,E451 Description of the exercise study protocol completed or when and why the test was stopped prematurely. Symptoms reported by patients Presence or absence of clinically significant changes in BP or heart rate or rhythm Evaluation of exercise performance and the ventilatory response to exercise Precise identification of the factor(s) that limit(s) exercise performance Results of baseline spirometry and changes seen Comments: Independent Health Facilities Pulmonary Function Protocol 28
29 FACILITY NAME/NO. PULMONARY FUNCTION STUDIES CHART REVIEW FORM PATIENT IDENTIFIER (e: either a chart # or patient initials) Date of Test: Tests Ordered by Referring Physician (please check as applicable) : Oxygen Saturation by Oximetry (J323, J332, J334) Non-specific Bronchial Provocative Test (J333) Carbon Monoxide Diffusing Capacity (J309, 310) MIPs & MEPs (J340) Function Residual Capacity (J311, 307) Stage 1 Exercise Testing(J315, E450, E451) Exercise Challenge Testing for Asthma (J330) Other TESTING GUIDELINES s s with 1. Does the requisition include: patient demographics; clinical information; working diagnosis or reason for test; referring physician s name and signature 2. Are the clinical indications for the procedure in compliance with those listed in the parameters? 3. Are these studies directly related to the patient s clinical condition? 4. Are these confirmed by the information documented in the patient s chart? 5. Was informed consent obtained from the patient based on the test being performed? 6. Are the pre-requisites met: Is spirometry properly performed? Are spirometry results known prior to the test? 14. Are the test results reproducible as outlined in the parameters and standards for: J323, J332, J334 J333 J309, J310 J340 J311, J307 J315, E450, E451 J For J309,310 -Do the acceptable tests reported meet ATS standards for reproducibility; collection volumes; breath hold time; inspiratory volume; inspiratory & expiratory times? 8. For J315, J330, E450, E451 Is a resting ECG done and results known? (exception young healthy individuals) 9. For J330, J315, E450, E451: Is blood pressure taken at 1-2 minute intervals during exercise & recovery from exercise? Are flow-volume loops pre-and post exercise properly performed and reported.? Is a physician in attendance at all times during the test? Independent Health Facilities Pulmonary Function Protocol 29
30 REPORTING GUIDELINES s s with 10. Does the report include the following: Patient demographics Interpretation of the data, and where appropriate comments as to the relevance of the results to the patient s presenting problem For J323, J332, J334 Interpretation of the data taking into account the limitations of the study For J333 Results of baseline spirometry Summary of the data and calculations made If the study is positive, provision of some quantification of hyperactivity For J309, J310 Technologist s comments as to the reliability of patient s performance where necessary Summary of the original data and calculations made during test and where feasible, of the graphical records. For J340 Results of baseline spirometry Technologists comments as to the reliability of patient s performance where necessary Comparison of patient measurement to predicted normal value as to patient management For J311, J307 Results of baseline spirometry Comparison of patient measurement to predicted normal values In the case of abnormal results, type of ventilatory abnormality expected For J330, J315, E450,E451 Description of the exercise study protocol completed or when and why the test was stopped prematurely. Symptoms reported by patients Presence or absence of clinically significant changes in BP or heart rate or rhythm Evaluation of exercise performance and the ventilatory response to exercise Precise identification of the factor(s) that limit(s) exercise performance Results of baseline spirometry and changes seen Comments: Independent Health Facilities Pulmonary Function Protocol 30
31 FACILITY NAME/NO. PULMONARY FUNCTION STUDIES CHART REVIEW FORM PATIENT IDENTIFIER (e: either a chart # or patient initials) Date of Test: Tests Ordered by Referring Physician (please check as applicable) : Oxygen Saturation by Oximetry (J323, J332, J334) Non-specific Bronchial Provocative Test (J333) Carbon Monoxide Diffusing Capacity (J309, 310) MIPs & MEPs (J340) Function Residual Capacity (J311, 307) Stage 1 Exercise Testing(J315, E450, E451) Exercise Challenge Testing for Asthma (J330) Other TESTING GUIDELINES s s with 1. Does the requisition include: patient demographics; clinical information; working diagnosis or reason for test; referring physician s name and signature 2. Are the clinical indications for the procedure in compliance with those listed in the parameters? 3. Are these studies directly related to the patient s clinical condition? 4. Are these confirmed by the information documented in the patient s chart? 5. Was informed consent obtained from the patient based on the test being performed? 6. Are the pre-requisites met: Is spirometry properly performed? Are spirometry results known prior to the test? 15. Are the test results reproducible as outlined in the parameters and standards for: J323, J332, J334 J333 J309, J310 J340 J311, J307 J315, E450, E451 J For J309,310 -Do the acceptable tests reported meet ATS standards for reproducibility; collection volumes; breath hold time; inspiratory volume; inspiratory & expiratory times? 8. For J315, J330, E450, E451 Is a resting ECG done and results known? (exception young healthy individuals) 9. For J330, J315, E450, E451: Is blood pressure taken at 1-2 minute intervals during exercise & recovery from exercise? Are flow-volume loops pre-and post exercise properly performed and reported.? Is a physician in attendance at all times during the test? Independent Health Facilities Pulmonary Function Protocol 31
32 REPORTING GUIDELINES s s with 10. Does the report include the following: Patient demographics Interpretation of the data, and where appropriate comments as to the relevance of the results to the patient s presenting problem For J323, J332, J334 Interpretation of the data taking into account the limitations of the study For J333 Results of baseline spirometry Summary of the data and calculations made If the study is positive, provision of some quantification of hyperactivity For J309, J310 Technologist s comments as to the reliability of patient s performance where necessary Summary of the original data and calculations made during test and where feasible, of the graphical records. For J340 Results of baseline spirometry Technologists comments as to the reliability of patient s performance where necessary Comparison of patient measurement to predicted normal value as to patient management For J311, J307 Results of baseline spirometry Comparison of patient measurement to predicted normal values In the case of abnormal results, type of ventilatory abnormality expected For J330, J315, E450,E451 Description of the exercise study protocol completed or when and why the test was stopped prematurely. Symptoms reported by patients Presence or absence of clinically significant changes in BP or heart rate or rhythm Evaluation of exercise performance and the ventilatory response to exercise Precise identification of the factor(s) that limit(s) exercise performance Results of baseline spirometry and changes seen Comments: Independent Health Facilities Pulmonary Function Protocol 32
Independent Health Facilities
 Independent Health Facilities Clinical Practice Parameters and Facility Standards Pulmonary Function Studies -3 rd Edition, April 2008 First Edition, October 1993 Members of the Respiratory Disease Task
Independent Health Facilities Clinical Practice Parameters and Facility Standards Pulmonary Function Studies -3 rd Edition, April 2008 First Edition, October 1993 Members of the Respiratory Disease Task
Credential Maintenance Program
 First Quarter of the Calendar 5 I. INSTRUMENTATION / EQUIPMENT 1 4 5 A. Set Up, Maintain, Calibrate 1 2 3 1. Blood gas analyzers 2. CO-oximeters / hemoximeters 3. Spirometers (for example, diagnostic,
First Quarter of the Calendar 5 I. INSTRUMENTATION / EQUIPMENT 1 4 5 A. Set Up, Maintain, Calibrate 1 2 3 1. Blood gas analyzers 2. CO-oximeters / hemoximeters 3. Spirometers (for example, diagnostic,
Pulmonary Function Technologists (PFT) Detailed Content Outline
 I. INSTRUMENTATION / EQUIPMENT 7 15 8 30 A. Set Up, Maintain, Calibrate 3 5 2 10 1. Blood gas analyzers 2. CO-oximeters / hemoximeters 3. Spirometers (for example, diagnostic, screening, portable) 4. Peak
I. INSTRUMENTATION / EQUIPMENT 7 15 8 30 A. Set Up, Maintain, Calibrate 3 5 2 10 1. Blood gas analyzers 2. CO-oximeters / hemoximeters 3. Spirometers (for example, diagnostic, screening, portable) 4. Peak
Spirometry and Flow Volume Measurements
 Spirometry and Flow Volume Measurements Standards & Guidelines December 1998 To serve the public and guide the medical profession Revision Dates: December 1998 Approval Date: June 1998 Originating Committee:
Spirometry and Flow Volume Measurements Standards & Guidelines December 1998 To serve the public and guide the medical profession Revision Dates: December 1998 Approval Date: June 1998 Originating Committee:
Quality Assurance Mapping Your QC Program Equipment and Test Quality. Susan Blonshine RRT, RPFT, FAARC, AE-C
 Quality Assurance Mapping Your QC Program Equipment and Test Quality Susan Blonshine RRT, RPFT, FAARC, AE-C How to Begin Gather resources Define PF scope of service Procedures performed Equipment Describe
Quality Assurance Mapping Your QC Program Equipment and Test Quality Susan Blonshine RRT, RPFT, FAARC, AE-C How to Begin Gather resources Define PF scope of service Procedures performed Equipment Describe
NBRC Exam RPFT Registry Examination for Advanced Pulmonary Function Technologists Version: 6.0 [ Total Questions: 111 ]
![NBRC Exam RPFT Registry Examination for Advanced Pulmonary Function Technologists Version: 6.0 [ Total Questions: 111 ] NBRC Exam RPFT Registry Examination for Advanced Pulmonary Function Technologists Version: 6.0 [ Total Questions: 111 ]](/thumbs/76/73199826.jpg) s@lm@n NBRC Exam RPFT Registry Examination for Advanced Pulmonary Function Technologists Version: 6.0 [ Total Questions: 111 ] https://certkill.com NBRC RPFT : Practice Test Question No : 1 Using a peak
s@lm@n NBRC Exam RPFT Registry Examination for Advanced Pulmonary Function Technologists Version: 6.0 [ Total Questions: 111 ] https://certkill.com NBRC RPFT : Practice Test Question No : 1 Using a peak
DIAGNOSTIC ACCREDITATION PROGRAM. Spirometry Quality Control Plan
 DIAGNOSTIC ACCREDITATION PROGRAM Spirometry Quality Control Plan Table of Contents Introduction...1 Spirometry Definitions and Requirements...2 Spirometry Requirements... 2...4 Daily Quality Control (see
DIAGNOSTIC ACCREDITATION PROGRAM Spirometry Quality Control Plan Table of Contents Introduction...1 Spirometry Definitions and Requirements...2 Spirometry Requirements... 2...4 Daily Quality Control (see
Performing a Methacholine Challenge Test
 powder for solution, for inhalation Performing a Methacholine Challenge Test Provocholine is a registered trademark of Methapharm Inc. Copyright Methapharm Inc. 2016. All rights reserved. Healthcare professionals
powder for solution, for inhalation Performing a Methacholine Challenge Test Provocholine is a registered trademark of Methapharm Inc. Copyright Methapharm Inc. 2016. All rights reserved. Healthcare professionals
Chapter 3. Pulmonary Function Study Assessments. Mosby items and derived items 2011, 2006 by Mosby, Inc., an affiliate of Elsevier Inc.
 Chapter 3 Pulmonary Function Study Assessments 1 Introduction Pulmonary function studies are used to: Evaluate pulmonary causes of dyspnea Differentiate between obstructive and restrictive pulmonary disorders
Chapter 3 Pulmonary Function Study Assessments 1 Introduction Pulmonary function studies are used to: Evaluate pulmonary causes of dyspnea Differentiate between obstructive and restrictive pulmonary disorders
Facility Information for Initial Assessment
 DIAGNOSTIC ACCREDITATION PROGRAM 300 669 Howe Street Telephone: 604-733-7758 Vancouver BC V6C 0B4 Toll Free: 1-800-461-3008 (in BC) www.cpsbc.ca Fax: 604-733-3503 Facility Information for Initial Assessment
DIAGNOSTIC ACCREDITATION PROGRAM 300 669 Howe Street Telephone: 604-733-7758 Vancouver BC V6C 0B4 Toll Free: 1-800-461-3008 (in BC) www.cpsbc.ca Fax: 604-733-3503 Facility Information for Initial Assessment
PULMONARY FUNCTION TESTING. Purposes of Pulmonary Tests. General Categories of Lung Diseases. Types of PF Tests
 PULMONARY FUNCTION TESTING Wyka Chapter 13 Various AARC Clinical Practice Guidelines Purposes of Pulmonary Tests Is lung disease present? If so, is it reversible? If so, what type of lung disease is present?
PULMONARY FUNCTION TESTING Wyka Chapter 13 Various AARC Clinical Practice Guidelines Purposes of Pulmonary Tests Is lung disease present? If so, is it reversible? If so, what type of lung disease is present?
Pulmonary Function Laboratory ATS Accreditation Are you prepared? Susan Blonshine RRT, RPFT, FAARC, AE-C
 Pulmonary Function Laboratory ATS Accreditation Are you prepared? Susan Blonshine RRT, RPFT, FAARC, AE-C Why Quality Consistent, accurate, reliable results Cost-effective Diagnosis Misclassification Reduce
Pulmonary Function Laboratory ATS Accreditation Are you prepared? Susan Blonshine RRT, RPFT, FAARC, AE-C Why Quality Consistent, accurate, reliable results Cost-effective Diagnosis Misclassification Reduce
Spirometry Training Courses. Spirometry for. Thoracic Society of Australia and New Zealand. June Developed in partnership with
 Standards for Spirometry Training Courses Companion Document to Standards for the Delivery of Spirometry for Coal Mine Workers Thoracic Society of Australia and New Zealand June 2017 Developed in partnership
Standards for Spirometry Training Courses Companion Document to Standards for the Delivery of Spirometry for Coal Mine Workers Thoracic Society of Australia and New Zealand June 2017 Developed in partnership
UNIT TWO: OVERVIEW OF SPIROMETRY. A. Definition of Spirometry
 UNIT TWO: OVERVIEW OF SPIROMETRY A. Definition of Spirometry Spirometry is a medical screening test that measures various aspects of breathing and lung function. It is performed by using a spirometer,
UNIT TWO: OVERVIEW OF SPIROMETRY A. Definition of Spirometry Spirometry is a medical screening test that measures various aspects of breathing and lung function. It is performed by using a spirometer,
6- Lung Volumes and Pulmonary Function Tests
 6- Lung Volumes and Pulmonary Function Tests s (PFTs) are noninvasive diagnostic tests that provide measurable feedback about the function of the lungs. By assessing lung volumes, capacities, rates of
6- Lung Volumes and Pulmonary Function Tests s (PFTs) are noninvasive diagnostic tests that provide measurable feedback about the function of the lungs. By assessing lung volumes, capacities, rates of
NATIONAL COMPETENCY SKILL STANDARDS FOR PERFORMING POLYSOMNOGRAPHY/SLEEP TECHNOLOGY
 NATIONAL COMPETENCY SKILL STANDARDS FOR PERFORMING POLYSOMNOGRAPHY/SLEEP TECHNOLOGY Polysomnography/Sleep Technology providers practice in accordance with the facility policy and procedure manual which
NATIONAL COMPETENCY SKILL STANDARDS FOR PERFORMING POLYSOMNOGRAPHY/SLEEP TECHNOLOGY Polysomnography/Sleep Technology providers practice in accordance with the facility policy and procedure manual which
Biology 236 Spring 2002 Campos/Wurdak/Fahey Laboratory 4. Cardiovascular and Respiratory Adjustments to Stationary Bicycle Exercise.
 BACKGROUND: Cardiovascular and Respiratory Adjustments to Stationary Bicycle Exercise. The integration of cardiovascular and respiratory adjustments occurring in response to varying levels of metabolic
BACKGROUND: Cardiovascular and Respiratory Adjustments to Stationary Bicycle Exercise. The integration of cardiovascular and respiratory adjustments occurring in response to varying levels of metabolic
PORTO 2 VENT CPAP OS. Operator s Manual. PORTO 2VENT CPAP OS System Operator s Manual Part Number Rev I
 PORTO 2 VENT CPAP OS Operator s Manual 1 2 TABLE of CONTENTS 1. Introduction 3. Operating Instructions 1a. Definitions 3a. Setting the CPAP Level 1b. General Description 3b. Applying the Breathing Circuit
PORTO 2 VENT CPAP OS Operator s Manual 1 2 TABLE of CONTENTS 1. Introduction 3. Operating Instructions 1a. Definitions 3a. Setting the CPAP Level 1b. General Description 3b. Applying the Breathing Circuit
Content Indica c tion Lung v olumes e & Lung Indica c tions i n c paci c ties
 Spirometry Content Indication Indications in occupational medicine Contraindications Confounding factors Complications Type of spirometer Lung volumes & Lung capacities Spirometric values Hygiene &
Spirometry Content Indication Indications in occupational medicine Contraindications Confounding factors Complications Type of spirometer Lung volumes & Lung capacities Spirometric values Hygiene &
SPIROMETRY. Marijke Currie (CRFS) Care Medical Ltd Phone: Copyright CARE Medical ltd
 SPIROMETRY Marijke Currie (CRFS) Care Medical Ltd Phone: 0800 333 808 Email: sales@caremed.co.nz What is spirometry Spirometry is a physiological test that measures the volume of air an individual can
SPIROMETRY Marijke Currie (CRFS) Care Medical Ltd Phone: 0800 333 808 Email: sales@caremed.co.nz What is spirometry Spirometry is a physiological test that measures the volume of air an individual can
1.40 Prevention of Nosocomial Pneumonia
 1.40 Prevention of Nosocomial Pneumonia Purpose Audience Policy Statement: The guideline is designed to reduce the incidence of pneumonia and other acute lower respiratory tract infections. All UTMB healthcare
1.40 Prevention of Nosocomial Pneumonia Purpose Audience Policy Statement: The guideline is designed to reduce the incidence of pneumonia and other acute lower respiratory tract infections. All UTMB healthcare
COURSE OUTLINE (Page 1 of 5)
 (Page 1 of 5) Date form completed: June 2018 Course Title: Advanced Respiratory Care Course Prefix & Number: Lecture: RESP 2100 3.5 Lab: 3.0 Credit Hours: 4.5 COURSE DESCRIPTION: This course includes advanced
(Page 1 of 5) Date form completed: June 2018 Course Title: Advanced Respiratory Care Course Prefix & Number: Lecture: RESP 2100 3.5 Lab: 3.0 Credit Hours: 4.5 COURSE DESCRIPTION: This course includes advanced
I. Subject: Continuous Positive Airway Pressure CPAP by Continuous Flow Device
 I. Subject: Continuous Positive Airway Pressure CPAP by Continuous Flow Device II. Policy: Continuous Positive Airway Pressure CPAP by the Down's system will be instituted by Respiratory Therapy personnel
I. Subject: Continuous Positive Airway Pressure CPAP by Continuous Flow Device II. Policy: Continuous Positive Airway Pressure CPAP by the Down's system will be instituted by Respiratory Therapy personnel
Pulmonary Function Testing: Concepts and Clinical Applications. Potential Conflict Of Interest. Objectives. Rationale: Why Test?
 Pulmonary Function Testing: Concepts and Clinical Applications David M Systrom, MD Potential Conflict Of Interest Nothing to disclose pertinent to this presentation BRIGHAM AND WOMEN S HOSPITAL Harvard
Pulmonary Function Testing: Concepts and Clinical Applications David M Systrom, MD Potential Conflict Of Interest Nothing to disclose pertinent to this presentation BRIGHAM AND WOMEN S HOSPITAL Harvard
-SQA-SCOTTISH QUALIFICATIONS AUTHORITY. Hanover House 24 Douglas Street GLASGOW G2 7NQ NATIONAL CERTIFICATE MODULE DESCRIPTOR
 -SQA-SCOTTISH QUALIFICATIONS AUTHORITY Hanover House 24 Douglas Street GLASGOW G2 7NQ NATIONAL CERTIFICATE MODULE DESCRIPTOR -Module Number- 0099111 -Session-1989-90 -Superclass- PB -Title- PRINCIPLES
-SQA-SCOTTISH QUALIFICATIONS AUTHORITY Hanover House 24 Douglas Street GLASGOW G2 7NQ NATIONAL CERTIFICATE MODULE DESCRIPTOR -Module Number- 0099111 -Session-1989-90 -Superclass- PB -Title- PRINCIPLES
MEDIUM-FLOW PNEUMOTACH TRANSDUCER
 MEDIUM-FLOW PNEUMOTACH TRANSDUCER SS11LA for MP3x and MP45 System TSD117 & TSD117-MRI for MP150/MP100 System RX117 Replacement Airflow Head See also: AFT series of accessories for airflow and gas analysis
MEDIUM-FLOW PNEUMOTACH TRANSDUCER SS11LA for MP3x and MP45 System TSD117 & TSD117-MRI for MP150/MP100 System RX117 Replacement Airflow Head See also: AFT series of accessories for airflow and gas analysis
Independent Health Facilities. Clinical Practice Parameters and Facility Standards. Pulmonary Function Studies - 4th Edition, 2014 DRAFT
 Independent Health Facilities Clinical Practice Parameters and Facility Standards Pulmonary Function Studies - 4th Edition, 2014 DRAFT First Edition, October 1993: Members of the Respiratory Disease Task
Independent Health Facilities Clinical Practice Parameters and Facility Standards Pulmonary Function Studies - 4th Edition, 2014 DRAFT First Edition, October 1993: Members of the Respiratory Disease Task
Course Description A study of pulmonary testing functions and cardiac dysrhythmias interpretation
 Cardiopulmonary Testing (RSPT 1335) Credit: 3 semester credit hours (2 hours lecture, 2 hours lab) Prerequisite: RSPT 1329, RSPT 1207, RSPT 2310, RSPT 1113, RSPT 1325 Co-requisite: RSPT 1331, RSPT 1360,
Cardiopulmonary Testing (RSPT 1335) Credit: 3 semester credit hours (2 hours lecture, 2 hours lab) Prerequisite: RSPT 1329, RSPT 1207, RSPT 2310, RSPT 1113, RSPT 1325 Co-requisite: RSPT 1331, RSPT 1360,
Small Volume Nebulizer Treatment (Hand-Held)
 Small Volume Aerosol Treatment Page 1 of 6 Purpose Policy Physician's Order Small Volume Nebulizer Treatment To standardize the delivery of inhalation aerosol drug therapy via small volume (hand-held)
Small Volume Aerosol Treatment Page 1 of 6 Purpose Policy Physician's Order Small Volume Nebulizer Treatment To standardize the delivery of inhalation aerosol drug therapy via small volume (hand-held)
PULMONARY FUNCTION TESTS
 Chapter 4 PULMONARY FUNCTION TESTS M.G.Rajanandh, Department of Pharmacy Practice, SRM College of Pharmacy, SRM University. OBJECTIVES Review basic pulmonary anatomy and physiology. Understand the reasons
Chapter 4 PULMONARY FUNCTION TESTS M.G.Rajanandh, Department of Pharmacy Practice, SRM College of Pharmacy, SRM University. OBJECTIVES Review basic pulmonary anatomy and physiology. Understand the reasons
#7 - Respiratory System
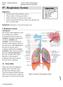 #7 - Respiratory System Objectives: Study the parts of the respiratory system Observe slides of the lung and trachea Perform spirometry to measure lung volumes Define and understand the lung volumes and
#7 - Respiratory System Objectives: Study the parts of the respiratory system Observe slides of the lung and trachea Perform spirometry to measure lung volumes Define and understand the lung volumes and
Effective Date: August 31, 2006 SUBJECT: CARE AND USE OF NEBULIZER AND INTERMITTENT POSITIVE PRESSURE BREATHING DEVICE
 COALINGA STATE HOSPITAL NURSING POLICY AND PROCEDURE MANUAL SECTION - Treatments POLICY NUMBER: 421 Effective Date: August 31, 2006 SUBJECT: CARE AND USE OF NEBULIZER AND INTERMITTENT POSITIVE PRESSURE
COALINGA STATE HOSPITAL NURSING POLICY AND PROCEDURE MANUAL SECTION - Treatments POLICY NUMBER: 421 Effective Date: August 31, 2006 SUBJECT: CARE AND USE OF NEBULIZER AND INTERMITTENT POSITIVE PRESSURE
Compressor Nebulizer System
 MedPro The Professional Choice Le choix des professionnels MedProTM MC Compressor Nebulizer System 705-445 Instruction Manual Index 1. Introduction...2 2. Product Identification...2 3. Important Safeguards...3
MedPro The Professional Choice Le choix des professionnels MedProTM MC Compressor Nebulizer System 705-445 Instruction Manual Index 1. Introduction...2 2. Product Identification...2 3. Important Safeguards...3
Teacher : Dorota Marczuk Krynicka, MD., PhD. Coll. Anatomicum, Święcicki Street no. 6, Dept. of Physiology
 Title: Spirometry Teacher : Dorota Marczuk Krynicka, MD., PhD. Coll. Anatomicum, Święcicki Street no. 6, Dept. of Physiology I. Measurements of Ventilation Spirometry A. Pulmonary Volumes 1. The tidal
Title: Spirometry Teacher : Dorota Marczuk Krynicka, MD., PhD. Coll. Anatomicum, Święcicki Street no. 6, Dept. of Physiology I. Measurements of Ventilation Spirometry A. Pulmonary Volumes 1. The tidal
PULMONARY FUNCTION. VOLUMES AND CAPACITIES
 PULMONARY FUNCTION. VOLUMES AND CAPACITIES The volume of air a person inhales (inspires) and exhales (expires) can be measured with a spirometer (spiro = breath, meter = to measure). A bell spirometer
PULMONARY FUNCTION. VOLUMES AND CAPACITIES The volume of air a person inhales (inspires) and exhales (expires) can be measured with a spirometer (spiro = breath, meter = to measure). A bell spirometer
DTSS Practice Inspections
 DTSS Practice Inspections A number of Quality and Safety validation checks have been introduced as part of the approval process prior to awarding DTSS contracts. The initial stage of a DTSS contract application
DTSS Practice Inspections A number of Quality and Safety validation checks have been introduced as part of the approval process prior to awarding DTSS contracts. The initial stage of a DTSS contract application
Getting Spirometry Right It Matters! Performance, Quality Assessment, and Interpretation. Susan Blonshine RRT, RPFT, AE-C, FAARC
 Getting Spirometry Right It Matters! Performance, Quality Assessment, and Interpretation Susan Blonshine RRT, RPFT, AE-C, FAARC Objectives Sample Title Recognize acceptable spirometry that meets the start
Getting Spirometry Right It Matters! Performance, Quality Assessment, and Interpretation Susan Blonshine RRT, RPFT, AE-C, FAARC Objectives Sample Title Recognize acceptable spirometry that meets the start
Portage Health Cardiopulmonary Policy. Cardiopulmonary. To provide guidelines for process and procedure with pulmonary function tests.
 Subject: Pulmonary Function Testing Scope: Cardiopulmonary Effective Date: 6/90 Revised: 3/99, 5/02, 6/09, 6/12 Approved by: Deb Young Approval Date: Portage Health Cardiopulmonary Policy Cardiopulmonary
Subject: Pulmonary Function Testing Scope: Cardiopulmonary Effective Date: 6/90 Revised: 3/99, 5/02, 6/09, 6/12 Approved by: Deb Young Approval Date: Portage Health Cardiopulmonary Policy Cardiopulmonary
. Type of solution/medication. Amount/dose to be delivered. Frequency/duration. Mode of administration.
 Page 1 of 9 (Large, Medium Volume, and Aerogen Continuous Nebulizer) Purpose Scope Physician's Order To standardize the use of continuous aerosol therapy as a modality used for the rescue of patients with
Page 1 of 9 (Large, Medium Volume, and Aerogen Continuous Nebulizer) Purpose Scope Physician's Order To standardize the use of continuous aerosol therapy as a modality used for the rescue of patients with
June 2011 Bill Streett-Training Section Chief
 Capnography 102 June 2011 Bill Streett-Training Section Chief Terminology Capnography: the measurement and numerical display of end-tidal CO2 concentration, at the patient s airway, during a respiratory
Capnography 102 June 2011 Bill Streett-Training Section Chief Terminology Capnography: the measurement and numerical display of end-tidal CO2 concentration, at the patient s airway, during a respiratory
Basic approach to PFT interpretation. Dr. Giulio Dominelli BSc, MD, FRCPC Kelowna Respiratory and Allergy Clinic
 Basic approach to PFT interpretation Dr. Giulio Dominelli BSc, MD, FRCPC Kelowna Respiratory and Allergy Clinic Disclosures Received honorarium from Astra Zeneca for education presentations Tasked Asked
Basic approach to PFT interpretation Dr. Giulio Dominelli BSc, MD, FRCPC Kelowna Respiratory and Allergy Clinic Disclosures Received honorarium from Astra Zeneca for education presentations Tasked Asked
behaviour are out of scope of the present review.
 explained about the test, a trial may be done before recording the results. The maneuver consists initially of normal tidal breathing. The subject then inhales to maximally fill the lungs. This is followed
explained about the test, a trial may be done before recording the results. The maneuver consists initially of normal tidal breathing. The subject then inhales to maximally fill the lungs. This is followed
BETTER SPIROMETRY. Marijke Currie (CRFS) Care Medical Ltd Phone: Copyright CARE Medical ltd
 BETTER SPIROMETRY Marijke Currie (CRFS) Care Medical Ltd Phone: 0800 333 808 Email: sales@caremed.co.nz What is spirometry Spirometry is a physiological test that measures the volume of air an individual
BETTER SPIROMETRY Marijke Currie (CRFS) Care Medical Ltd Phone: 0800 333 808 Email: sales@caremed.co.nz What is spirometry Spirometry is a physiological test that measures the volume of air an individual
COMPREHENSIVE RESPIROMETRY
 INTRODUCTION Respiratory System Structure Complex pathway for respiration 1. Specialized tissues for: a. Conduction b. Gas exchange 2. Position in respiratory pathway determines cell type Two parts Upper
INTRODUCTION Respiratory System Structure Complex pathway for respiration 1. Specialized tissues for: a. Conduction b. Gas exchange 2. Position in respiratory pathway determines cell type Two parts Upper
Split Night Protocols for Adult Patients - Updated July 2012
 Split Night Protocols for Adult Patients - Updated July 2012 SUMMARY: Sleep technologists are team members who work under the direction of a physician practicing sleep disorders medicine. Sleep technologists
Split Night Protocols for Adult Patients - Updated July 2012 SUMMARY: Sleep technologists are team members who work under the direction of a physician practicing sleep disorders medicine. Sleep technologists
1. When a patient fails to ventilate or oxygenate adequately, the problem is caused by pathophysiological factors such as hyperventilation.
 Chapter 1: Principles of Mechanical Ventilation TRUE/FALSE 1. When a patient fails to ventilate or oxygenate adequately, the problem is caused by pathophysiological factors such as hyperventilation. F
Chapter 1: Principles of Mechanical Ventilation TRUE/FALSE 1. When a patient fails to ventilate or oxygenate adequately, the problem is caused by pathophysiological factors such as hyperventilation. F
Competency Title: Continuous Positive Airway Pressure
 Competency Title: Continuous Positive Airway Pressure Trainee Name: ------------------------------------------------------------- Title: ---------------------------------------------------------------
Competency Title: Continuous Positive Airway Pressure Trainee Name: ------------------------------------------------------------- Title: ---------------------------------------------------------------
Compressor Nebulizer. Guidebook
 NSTRU Compressor Nebulizer Guidebook MODEL: MQ6002 READ THIS INSTRUCTION MANUAL CAREFULLY BEFORE USE Compressor Nebulizer MODEL NO: MQ6002 INSTRUCTIONS INDEX 1. Introduction ----------------------------------------------------------------
NSTRU Compressor Nebulizer Guidebook MODEL: MQ6002 READ THIS INSTRUCTION MANUAL CAREFULLY BEFORE USE Compressor Nebulizer MODEL NO: MQ6002 INSTRUCTIONS INDEX 1. Introduction ----------------------------------------------------------------
Pulmonary Function Testing. Ramez Sunna MD, FCCP
 Pulmonary Function Testing Ramez Sunna MD, FCCP Lecture Overview General Introduction Indications and Uses Technical aspects Interpretation Patterns of Abnormalities When to perform a PFT 1. Evaluation
Pulmonary Function Testing Ramez Sunna MD, FCCP Lecture Overview General Introduction Indications and Uses Technical aspects Interpretation Patterns of Abnormalities When to perform a PFT 1. Evaluation
Asthma Coding Fact Sheet for Primary Care Pediatricians
 01/01/2017 Asthma Coding Fact Sheet for Primary Care Pediatricians Physician Evaluation & Management Services Outpatient 99201 99202 99203 99204 99205 Office or other outpatient visit, new patient; self
01/01/2017 Asthma Coding Fact Sheet for Primary Care Pediatricians Physician Evaluation & Management Services Outpatient 99201 99202 99203 99204 99205 Office or other outpatient visit, new patient; self
SPIROMETRY METHOD. COR-MAN IN / EN Issue A, Rev INNOVISION ApS Skovvænget 2 DK-5620 Glamsbjerg Denmark
 SPIROMETRY METHOD COR-MAN-0000-006-IN / EN Issue A, Rev. 2 2013-07 INNOVISION ApS Skovvænget 2 DK-5620 Glamsbjerg Denmark Tel.: +45 65 95 91 00 Fax: +45 65 95 78 00 info@innovision.dk www.innovision.dk
SPIROMETRY METHOD COR-MAN-0000-006-IN / EN Issue A, Rev. 2 2013-07 INNOVISION ApS Skovvænget 2 DK-5620 Glamsbjerg Denmark Tel.: +45 65 95 91 00 Fax: +45 65 95 78 00 info@innovision.dk www.innovision.dk
Interfacility Protocol Protocol Title:
 Interfacility Protocol Protocol Title: Mechanical Ventilator Monitoring & Management Original Adoption Date: 05/2009 Past Protocol Updates 05/2009, 12/2013 Date of Most Recent Update: March 23, 2015 Medical
Interfacility Protocol Protocol Title: Mechanical Ventilator Monitoring & Management Original Adoption Date: 05/2009 Past Protocol Updates 05/2009, 12/2013 Date of Most Recent Update: March 23, 2015 Medical
Capnography (ILS/ALS)
 Capnography (ILS/ALS) Clinical Indications: 1. Capnography shall be used as soon as possible in conjunction with any airway management adjunct, including endotracheal, Blind Insertion Airway Devices (BIAD)
Capnography (ILS/ALS) Clinical Indications: 1. Capnography shall be used as soon as possible in conjunction with any airway management adjunct, including endotracheal, Blind Insertion Airway Devices (BIAD)
Spirometry: Introduction
 Spirometry: Introduction Dr. Badri Paudel 1 2 GMC Spirometry Spirometry is a method of assessing lung function by measuring the volume of air the patient can expel from the lungs after a maximal expiration.
Spirometry: Introduction Dr. Badri Paudel 1 2 GMC Spirometry Spirometry is a method of assessing lung function by measuring the volume of air the patient can expel from the lungs after a maximal expiration.
What do pulmonary function tests tell you?
 Pulmonary Function Testing Michael Wert, MD Assistant Professor Clinical Department of Internal Medicine Division of Pulmonary, Critical Care, and Sleep Medicine The Ohio State University Wexner Medical
Pulmonary Function Testing Michael Wert, MD Assistant Professor Clinical Department of Internal Medicine Division of Pulmonary, Critical Care, and Sleep Medicine The Ohio State University Wexner Medical
Continuous Aerosol Therapy
 PROCEDURE - : Page 1 of 5 Purpose Policy Physician's Order To standardize the administration of continuous aerosol therapy. Respiratory Care Services provides equipment and therapy according to physician
PROCEDURE - : Page 1 of 5 Purpose Policy Physician's Order To standardize the administration of continuous aerosol therapy. Respiratory Care Services provides equipment and therapy according to physician
PORTABLE COMPRESSOR NEBULIZER Guidebook & Manual Reorder No. 5606
 PORTABLE COMPRESSOR NEBULIZER Guidebook & Manual Reorder No. 5606 Please read this guidebook carefully before operating this unit. Your Nebulizer is intended for use in the treatment of asthma, COPD and
PORTABLE COMPRESSOR NEBULIZER Guidebook & Manual Reorder No. 5606 Please read this guidebook carefully before operating this unit. Your Nebulizer is intended for use in the treatment of asthma, COPD and
MASTER SYLLABUS
 MASTER SYLLABUS 2018-2019 A. Academic Division: Health Science B. Discipline: Respiratory Care C. Course Number and Title: RESP 2490 Practicum IV D. Course Coordinator: Tricia Winters, BBA, RRT, RCP Assistant
MASTER SYLLABUS 2018-2019 A. Academic Division: Health Science B. Discipline: Respiratory Care C. Course Number and Title: RESP 2490 Practicum IV D. Course Coordinator: Tricia Winters, BBA, RRT, RCP Assistant
REB SOP 04 Page 1 of 6. VO 2 Max - Maximal Oxygen Uptake Testing. Short Title. Effective Date July 4, Approved by REB July 4, 2017
 REB SOP 04 Page 1 of 6 Short Title VO 2 Max Testing Effective Date July 4, 2017 Approved by REB July 4, 2017 Version Number 1 A. PURPOSE AND BACKGROUND A graded maximal exertion test is used to determine
REB SOP 04 Page 1 of 6 Short Title VO 2 Max Testing Effective Date July 4, 2017 Approved by REB July 4, 2017 Version Number 1 A. PURPOSE AND BACKGROUND A graded maximal exertion test is used to determine
It is recommended that a mask and protective eyewear be worn when providing care to a patient with a cough
 UNIVERSITY HEALTH NETWORK POLICY #: PAGE 1 OF 7 POLICY AND PROCEDURE MANUAL: RESPIRATORY THERAPY DEPT PATIENT CARE SECTION ORIGINAL DATE: 04/03 ISSUED BY: SITE LEADER APPROVED BY: Infection Prevention
UNIVERSITY HEALTH NETWORK POLICY #: PAGE 1 OF 7 POLICY AND PROCEDURE MANUAL: RESPIRATORY THERAPY DEPT PATIENT CARE SECTION ORIGINAL DATE: 04/03 ISSUED BY: SITE LEADER APPROVED BY: Infection Prevention
Compressor Nebulizer. Guidebook
 NSTRU Compressor Nebulizer Guidebook MODEL: CN02WS (MQ6003) READ ALL INSTRUCTION BEFORE USE Airial TM Compressor Nebulizer INSTRUCTIONS INDEX 1. Introduction ----------------------------------------------------------------
NSTRU Compressor Nebulizer Guidebook MODEL: CN02WS (MQ6003) READ ALL INSTRUCTION BEFORE USE Airial TM Compressor Nebulizer INSTRUCTIONS INDEX 1. Introduction ----------------------------------------------------------------
SPIROMETRY. Performance and Interpretation for Healthcare Professionals. Faculty of Respiratory Physiology IICMS. Version 1 April 2015
 SPIROMETRY Performance and Interpretation for Healthcare Professionals Faculty of Respiratory Physiology IICMS Version 1 April 2015 Authors: Maria Mc Neill & Geraldine Nolan www.iars.ie Contents Page Background
SPIROMETRY Performance and Interpretation for Healthcare Professionals Faculty of Respiratory Physiology IICMS Version 1 April 2015 Authors: Maria Mc Neill & Geraldine Nolan www.iars.ie Contents Page Background
Mechanical Ventilation Principles and Practices
 Mechanical Ventilation Principles and Practices Dr LAU Chun Wing Arthur Department of Intensive Care Pamela Youde Nethersole Eastern Hospital 6 October 2009 In this lecture, you will learn Major concepts
Mechanical Ventilation Principles and Practices Dr LAU Chun Wing Arthur Department of Intensive Care Pamela Youde Nethersole Eastern Hospital 6 October 2009 In this lecture, you will learn Major concepts
SS11LA Pneumotach Transducer
 BIOPAC Systems, Inc. 42 Aero Camino Goleta, CA 93117 Ph (805) 685-0066 Fax (805) 685-0067 www.biopac.com info@biopac.com SS11LA Pneumotach Transducer The SS11LA can be used to measure respiratory flow
BIOPAC Systems, Inc. 42 Aero Camino Goleta, CA 93117 Ph (805) 685-0066 Fax (805) 685-0067 www.biopac.com info@biopac.com SS11LA Pneumotach Transducer The SS11LA can be used to measure respiratory flow
ISO INTERNATIONAL STANDARD
 INTERNATIONAL STANDARD ISO 26782 First edition 2009-07-15 Anaesthetic and respiratory equipment Spirometers intended for the measurement of time forced expired volumes in humans Matériel d'anesthésie et
INTERNATIONAL STANDARD ISO 26782 First edition 2009-07-15 Anaesthetic and respiratory equipment Spirometers intended for the measurement of time forced expired volumes in humans Matériel d'anesthésie et
Medescan Nebuliser Med - S600A Instructions Manual
 Medescan Nebuliser Med - S600A Instructions Manual Please read this guidebook carefully before operating this unit Your Nebuliser is intended for use in the treatment of asthma, COPD and other respiratory
Medescan Nebuliser Med - S600A Instructions Manual Please read this guidebook carefully before operating this unit Your Nebuliser is intended for use in the treatment of asthma, COPD and other respiratory
Airway Clearance Devices
 Print Page 1 of 11 Wisconsin.gov home state agencies subject directory department of health services Search Welcome» August 2, 2018 5:18 PM Program Name: BadgerCare Plus and Medicaid Handbook Area: Durable
Print Page 1 of 11 Wisconsin.gov home state agencies subject directory department of health services Search Welcome» August 2, 2018 5:18 PM Program Name: BadgerCare Plus and Medicaid Handbook Area: Durable
CLINICAL CONSIDERATIONS FOR THE BUNNELL LIFE PULSE HIGH-FREQUENCY JET VENTILATOR
 CLINICAL CONSIDERATIONS FOR THE BUNNELL LIFE PULSE HIGH-FREQUENCY JET VENTILATOR 801-467-0800 Phone 800-800-HFJV (4358) Hotline TABLE OF CONTENTS Respiratory Care Considerations..3 Physician Considerations
CLINICAL CONSIDERATIONS FOR THE BUNNELL LIFE PULSE HIGH-FREQUENCY JET VENTILATOR 801-467-0800 Phone 800-800-HFJV (4358) Hotline TABLE OF CONTENTS Respiratory Care Considerations..3 Physician Considerations
FOLLOW-UP MEDICAL CARE OF SERVICE MEMBERS AND VETERANS CARDIOPULMONARY EXERCISE TESTING
 Cardiopulmonary Exercise Testing Chapter 13 FOLLOW-UP MEDICAL CARE OF SERVICE MEMBERS AND VETERANS CARDIOPULMONARY EXERCISE TESTING WILLIAM ESCHENBACHER, MD* INTRODUCTION AEROBIC METABOLISM ANAEROBIC METABOLISM
Cardiopulmonary Exercise Testing Chapter 13 FOLLOW-UP MEDICAL CARE OF SERVICE MEMBERS AND VETERANS CARDIOPULMONARY EXERCISE TESTING WILLIAM ESCHENBACHER, MD* INTRODUCTION AEROBIC METABOLISM ANAEROBIC METABOLISM
Basic Standards for Osteopathic Fellowship Training in Sleep Medicine
 Basic Standards for Osteopathic Fellowship Training in Sleep Medicine American Osteopathic Association and the American College of Osteopathic Neurologists and Psychiatrists and the American College of
Basic Standards for Osteopathic Fellowship Training in Sleep Medicine American Osteopathic Association and the American College of Osteopathic Neurologists and Psychiatrists and the American College of
PEDIATRIC PAP TITRATION PROTOCOL
 PURPOSE In order to provide the highest quality care for our patients, our sleep disorders facility adheres to the AASM Standards of Accreditation. The accompanying policy and procedure on pediatric titrations
PURPOSE In order to provide the highest quality care for our patients, our sleep disorders facility adheres to the AASM Standards of Accreditation. The accompanying policy and procedure on pediatric titrations
S P I R O M E T R Y. Objectives. Objectives 3/12/2018
 S P I R O M E T R Y Dewey Hahlbohm, PA-C, AE-C Objectives To understand the uses and importance of spirometry testing To perform spirometry testing including reversibility testing To identify normal and
S P I R O M E T R Y Dewey Hahlbohm, PA-C, AE-C Objectives To understand the uses and importance of spirometry testing To perform spirometry testing including reversibility testing To identify normal and
Respiratory Protection for Exposures to the Influenza A (H1N1) Virus. Frequently Asked Questions (FAQs)
 3M Occupational Health and 3M Center Environmental Safety Division St. Paul, MN 55144-1000 651 733 1110 Respiratory Protection for Exposures to the Influenza A (H1N1) Virus Frequently Asked Questions (FAQs)
3M Occupational Health and 3M Center Environmental Safety Division St. Paul, MN 55144-1000 651 733 1110 Respiratory Protection for Exposures to the Influenza A (H1N1) Virus Frequently Asked Questions (FAQs)
SUPPLEMENT TO CHAPTER 6: QUALITY MANAGEMENT
 SUPPLEMENT TO CHAPTER 6: QUALITY MANAGEMENT Clinical Practice Parameters and Standards Sleep Medicine 4th Edition, October 2016 Supplement to Chapter 6 - Quality Management Guide to Selecting and Preparing
SUPPLEMENT TO CHAPTER 6: QUALITY MANAGEMENT Clinical Practice Parameters and Standards Sleep Medicine 4th Edition, October 2016 Supplement to Chapter 6 - Quality Management Guide to Selecting and Preparing
You Take My Breath Away. Student Information Page 5C Part 1
 You Take My Breath Away Student Information Page 5C Part 1 Students with asthma or other respiratory problems should not participate in this activity because it involves repeated maximal inhalations and
You Take My Breath Away Student Information Page 5C Part 1 Students with asthma or other respiratory problems should not participate in this activity because it involves repeated maximal inhalations and
Cardiopulmonary exercise testing (CPET)
 Cardiopulmonary exercise testing (CPET) A patient s guide 1 What is a cardiopulmonary exercise test? The test is used to assess the response of your heart and lungs to exercise. It can give useful information
Cardiopulmonary exercise testing (CPET) A patient s guide 1 What is a cardiopulmonary exercise test? The test is used to assess the response of your heart and lungs to exercise. It can give useful information
Locally Enhanced Service for Stopping Smoking
 NHS Devon Locally Enhanced Service for Stopping Smoking This Local Enhanced Service (LES) Specification details the agreement between Devon PCT (the commissioner) and community pharmacies (the service
NHS Devon Locally Enhanced Service for Stopping Smoking This Local Enhanced Service (LES) Specification details the agreement between Devon PCT (the commissioner) and community pharmacies (the service
HEARING CONSERVATION PROGRAM
 CALIFORNIA STATE UNIVERSITY, CHICO HEARING CONSERVATION PROGRAM PREPARED BY THE OFFICE OF ENVIRONMENTAL HEALTH AND SAFETY REVISED June 2008 TABLE OF CONTENTS Section Page 1.0 Introduction... 1-1 2.0 Exposure
CALIFORNIA STATE UNIVERSITY, CHICO HEARING CONSERVATION PROGRAM PREPARED BY THE OFFICE OF ENVIRONMENTAL HEALTH AND SAFETY REVISED June 2008 TABLE OF CONTENTS Section Page 1.0 Introduction... 1-1 2.0 Exposure
Pulmonary Function Tests. Mohammad Babai M.D Occupational Medicine Specialist
 Pulmonary Function Tests Mohammad Babai M.D Occupational Medicine Specialist www.drbabai.com Pulmonary Function Tests Pulmonary Function Tests: Spirometry Peak-Flow metry Bronchoprovocation Tests Body
Pulmonary Function Tests Mohammad Babai M.D Occupational Medicine Specialist www.drbabai.com Pulmonary Function Tests Pulmonary Function Tests: Spirometry Peak-Flow metry Bronchoprovocation Tests Body
In order to diagnose lung diseases doctors
 You Take My Breath Away Activity 5C NOTE: This activity is designed to follow You Really Are Full of Hot Air! Activity Objectives: After completing You Really Are Full of Hot Air! Activity 5B, students
You Take My Breath Away Activity 5C NOTE: This activity is designed to follow You Really Are Full of Hot Air! Activity Objectives: After completing You Really Are Full of Hot Air! Activity 5B, students
Scope of Practice Athletic Rehabilitation Therapist, Certified (A.R.T.C.)
 Scope of Practice Athletic Rehabilitation Therapist, Certified (A.R.T.C.) An Athletic Rehabilitation Therapist Certified (A.R.T.C) is an autonomous health care practitioner to whom members of the public
Scope of Practice Athletic Rehabilitation Therapist, Certified (A.R.T.C.) An Athletic Rehabilitation Therapist Certified (A.R.T.C) is an autonomous health care practitioner to whom members of the public
Pulmonary Function Test
 Spirometry: Introduction Dr. Badri Paudel GMC Spirometry Pulmonary Function Test! Spirometry is a method of assessing lung function by measuring the volume of air the patient can expel from the lungs after
Spirometry: Introduction Dr. Badri Paudel GMC Spirometry Pulmonary Function Test! Spirometry is a method of assessing lung function by measuring the volume of air the patient can expel from the lungs after
To deliver the Program of Care, you will be required to meet the following criteria:
 Acknowledgements The WSIB would like to acknowledge the significant contributions of the following associations, and workplace representatives in the development of the Program of Care for Noise Induced
Acknowledgements The WSIB would like to acknowledge the significant contributions of the following associations, and workplace representatives in the development of the Program of Care for Noise Induced
Breathing and pulmonary function
 EXPERIMENTAL PHYSIOLOGY EXPERIMENT 5 Breathing and pulmonary function Ying-ying Chen, PhD Dept. of Physiology, Zhejiang University School of Medicine bchenyy@zju.edu.cn Breathing Exercise 1: Tests of pulmonary
EXPERIMENTAL PHYSIOLOGY EXPERIMENT 5 Breathing and pulmonary function Ying-ying Chen, PhD Dept. of Physiology, Zhejiang University School of Medicine bchenyy@zju.edu.cn Breathing Exercise 1: Tests of pulmonary
PULMONARY FUNCTION TESTING. By: Gh. Pouryaghoub. MD Center for Research on Occupational Diseases (CROD) Tehran University of Medical Sciences (TUMS)
 PULMONARY FUNCTION TESTING By: Gh. Pouryaghoub. MD Center for Research on Occupational Diseases (CROD) Tehran University of Medical Sciences (TUMS) PULMONARY FUNCTION TESTS CATEGORIES Spirometry Lung volumes
PULMONARY FUNCTION TESTING By: Gh. Pouryaghoub. MD Center for Research on Occupational Diseases (CROD) Tehran University of Medical Sciences (TUMS) PULMONARY FUNCTION TESTS CATEGORIES Spirometry Lung volumes
LEAD SAFETY PROGRAM. Purpose. Scope. Responsibilities. Southern Heat Exchanger Services Safety Program
 Page: Page 1 of 5 Purpose The purpose of this procedure is to identify the controls and actions necessary to prevent adverse health effects to employees from occupational exposure to lead, and to ensure
Page: Page 1 of 5 Purpose The purpose of this procedure is to identify the controls and actions necessary to prevent adverse health effects to employees from occupational exposure to lead, and to ensure
Cough Assist. Information for patients, families and carers Therapy Services
 Cough Assist Information for patients, families and carers Therapy Services PROUD TO MAKE A DIFFERENCE SHEFFIELD TEACHING HOSPITALS NHS FOUNDATION TRUST page 2 of 16 Table of contents Why do I need a Cough
Cough Assist Information for patients, families and carers Therapy Services PROUD TO MAKE A DIFFERENCE SHEFFIELD TEACHING HOSPITALS NHS FOUNDATION TRUST page 2 of 16 Table of contents Why do I need a Cough
Medical Emergency Management for the Dental Clinic
 Medical Emergency Management for the Dental Clinic Revised: Spring 2017 1 Medical Emergency Management Florida State College at Jacksonville Dental Clinic Topic Page Emergency Equipment and Supplies 3
Medical Emergency Management for the Dental Clinic Revised: Spring 2017 1 Medical Emergency Management Florida State College at Jacksonville Dental Clinic Topic Page Emergency Equipment and Supplies 3
PFT Interpretation and Reference Values
 PFT Interpretation and Reference Values September 21, 2018 Eric Wong Objectives Understand the components of PFT Interpretation of PFT Clinical Patterns How to choose Reference Values 3 Components Spirometry
PFT Interpretation and Reference Values September 21, 2018 Eric Wong Objectives Understand the components of PFT Interpretation of PFT Clinical Patterns How to choose Reference Values 3 Components Spirometry
POSITION DESCRIPTION / PERFORMANCE EVALUATION
 POSITION DESCRIPTION / PERFORMANCE EVALUATION Job Title: Cardiopulmonary Services Manager Prepared by: Date: Supervised by: Cardiopulmonary Services Medical Director, CEO Approved by: Date: Job Summary:
POSITION DESCRIPTION / PERFORMANCE EVALUATION Job Title: Cardiopulmonary Services Manager Prepared by: Date: Supervised by: Cardiopulmonary Services Medical Director, CEO Approved by: Date: Job Summary:
Subject Index. Carbon monoxide (CO) disease effects on levels 197, 198 measurement in exhaled air 197 sources in exhaled air 197
 Subject Index Airway resistance airflow interruption measurement in preschoolers, see Forced oscillation technique; Interrupter technique plethysmography, see Plethysmography; Whole-body plethysmography
Subject Index Airway resistance airflow interruption measurement in preschoolers, see Forced oscillation technique; Interrupter technique plethysmography, see Plethysmography; Whole-body plethysmography
FACTORIES ACT (CHAPTER 104, SECTION 102 (1)) FACTORIES (NOISE) REGULATIONS
 FACTORIES ACT (CHAPTER 104, SECTION 102 (1)) FACTORIES (NOISE) REGULATIONS [1st January 1997] Arrangement of Provisions PART I PRELIMINARY 1 Citation 2 Definitions 3 Application PART II NOISE CONTROL 4
FACTORIES ACT (CHAPTER 104, SECTION 102 (1)) FACTORIES (NOISE) REGULATIONS [1st January 1997] Arrangement of Provisions PART I PRELIMINARY 1 Citation 2 Definitions 3 Application PART II NOISE CONTROL 4
INVESTIGATING BREATHING: SHEET A
 Activity 7.13a Student Sheet INVESTIGATING BREATHING: SHEET A Purpose To investigate tidal volume, vital capacity, rate of breathing, respiratory minute ventilation and oxygen consumption. To interpret
Activity 7.13a Student Sheet INVESTIGATING BREATHING: SHEET A Purpose To investigate tidal volume, vital capacity, rate of breathing, respiratory minute ventilation and oxygen consumption. To interpret
Niche News August 2017 August 2017
 Niche News August 2017 August 2017 In This Issue Greetings! Introducing the new EasyOne Air Spirometer LiteAire Spacer - Get Better Pricing with Bulk Buy FeNO "Value Proposition" White Paper EasyOne Pro
Niche News August 2017 August 2017 In This Issue Greetings! Introducing the new EasyOne Air Spirometer LiteAire Spacer - Get Better Pricing with Bulk Buy FeNO "Value Proposition" White Paper EasyOne Pro
HCS70004P, HCS70004G HCS70004C, HCS70004BL HCS70004DP, HCS70004 AEROMIST COLORS NEBULIZER COMPRESSOR KIT. Instruction Manual
 HCS70004P, HCS70004G HCS70004C, HCS70004BL HCS70004DP, HCS70004 AEROMIST COLORS NEBULIZER COMPRESSOR KIT Instruction Manual TABLE OF CONTENTS IEC Symbols...2 Important Safeguards...2 Introduction...3 Specifications...4
HCS70004P, HCS70004G HCS70004C, HCS70004BL HCS70004DP, HCS70004 AEROMIST COLORS NEBULIZER COMPRESSOR KIT Instruction Manual TABLE OF CONTENTS IEC Symbols...2 Important Safeguards...2 Introduction...3 Specifications...4
NHA Certified EKG Technician (CET) Test Plan for the CET Exam
 NHA Certified EKG Technician (CET) Test Plan for the CET Exam 100 scored items Exam Time: 2 hours *Based on the results of a job analysis completed in 2017 This document provides both a summary and detailed
NHA Certified EKG Technician (CET) Test Plan for the CET Exam 100 scored items Exam Time: 2 hours *Based on the results of a job analysis completed in 2017 This document provides both a summary and detailed
Drew University Health Service 36 Madison Avenue Madison, New Jersey Tel: Fax:
 Dear Student, Enclosed you will find our policies, procedures and student consent form for your allergy immunotherapy. We ask that you read them carefully, sign the consent form, and take the physician
Dear Student, Enclosed you will find our policies, procedures and student consent form for your allergy immunotherapy. We ask that you read them carefully, sign the consent form, and take the physician
REQUISITION FOR DIAGNOSTIC SERVICES
 2300 McPhillips Street, Winnipeg, Manitoba R2V 3M3 REQUISITION FOR DIAGNOSTIC SERVICES PATIENT NAME ADDRESS BIRTH M.H.# P.H.I.N.# TELEPHONE HOME BUSINESS MALE FEMALE POSTAL CODE HOSPITAL USE ONLY ACCOUNT
2300 McPhillips Street, Winnipeg, Manitoba R2V 3M3 REQUISITION FOR DIAGNOSTIC SERVICES PATIENT NAME ADDRESS BIRTH M.H.# P.H.I.N.# TELEPHONE HOME BUSINESS MALE FEMALE POSTAL CODE HOSPITAL USE ONLY ACCOUNT
COMMISSION ON ACCREDITATION FOR RESPIRATORY CARE TMC DETAILED CONTENT OUTLINE COMPARISON
 A. Evaluate Data in the Patient Record I. PATIENT DATA EVALUATION AND RECOMMENDATIONS 1. Patient history e.g., admission data orders medications progress notes DNR status / advance directives social history
A. Evaluate Data in the Patient Record I. PATIENT DATA EVALUATION AND RECOMMENDATIONS 1. Patient history e.g., admission data orders medications progress notes DNR status / advance directives social history
Environmental Health & Safety Policy Manual
 Environmental Health & Safety Policy Manual Issue Date: 5/31/2017 Policy # EHS-400.17 Isoflurane Use and Exposure Control Procedures 1.0 PURPOSE: LSUHSC is committed to keeping all exposures to hazardous
Environmental Health & Safety Policy Manual Issue Date: 5/31/2017 Policy # EHS-400.17 Isoflurane Use and Exposure Control Procedures 1.0 PURPOSE: LSUHSC is committed to keeping all exposures to hazardous
