NIH Public Access Author Manuscript Cell Rep. Author manuscript; available in PMC 2015 June 12.
|
|
|
- Rosamund Small
- 6 years ago
- Views:
Transcription
1 NIH Public Access Author Manuscript Published in final edited form as: Cell Rep June 12; 7(5): doi: /j.celrep Break-Induced Replication is a Source of Mutation Clusters Underlying Kataegis Cynthia J. Sakofsky 1,4,*, Steven A. Roberts 2,*, Ewa Malc 3, Piotr A. Mieczkowski 3, Michael A. Resnick 2, Dmitry A. Gordenin 2,#, and Anna Malkova 1,4,# 1 Department of Biology, College of Liberal Arts and Sciences, University of Iowa, Iowa City, Iowa , USA 2 National Institute of Environmental Health Sciences, Research Triangle Park, NC 27709, USA 3 Department of Genetics, Lineberger Comprehensive Cancer Center and Carolina Center for Genome Sciences, University of North Carolina, Chapel Hill, NC 27599, USA 4 Department of Biology, School of Science, IUPUI, Indianapolis, IN, , USA Summary Clusters of simultaneous multiple mutations can be a source of rapid change during carcinogenesis and evolution. Such mutation clusters have been recently shown to originate from DNA damage within long single-strand (ss) DNA formed at resected double-strand breaks and dysfunctional replication forks. We identify here double-strand break (DSB)-induced replication (BIR) as another powerful source of mutation clusters that formed in nearly half of wild-type yeast cells undergoing BIR in the presence of alkylating damage. Clustered mutations were primarily formed along the track of DNA synthesis and were frequently associated with additional breakage and rearrangements. Moreover, the base specificity, strand coordination and strand bias of the mutation spectrum was consistent with mutations arising from damage in persistent ssdna stretches within unconventional replication intermediates. Together, these features closely resemble kataegic events in cancers, suggesting that replication intermediates during BIR may be the most prominent source of mutation clusters across species. Introduction DNA mutations provide genetic variation that promotes evolution and carcinogenesis. Since most individual genetic changes result in neutral or deleterious effects, the establishment of 2014 Elsevier Inc. All rights reserved. # Contact: anna-malkova@uiowa.edu (Anna Malkova), and gordenin@niehs.nih.gov (Dmitry Gordenin). * These authors contributed equally to this work. Author Contributions: C.J.S., S.A.R., D.A.G., and A.M. designed the experiments. C.J.S. performed BIR experiments. E.M. and P.A.M performed whole-genomic sequencing. S.A.R., C.J.S., M.A.R., D.A.G., and A.M. analyzed the data. C.J.S., S.A.R., D.A.G., and A.M. wrote the manuscript. A.M. and D.A.G. supervised the project. C.J.S. and S.A.R. contributed equally to the work. Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
2 Sakofsky et al. Page 2 novel protein characteristics often requires multiple mutations arising in concert within a single gene (Camps et al., 2007). Processes that over-produce scattered mutations throughout the genome (i.e., defects in mismatch repair and proofreading) are unlikely sources of spatially clustered multiple mutations since they increase the overall mutation load, which would negatively affect fitness. Therefore, processes that produce simultaneous mutations clustered within small sections of the genome are more likely to play a role in promoting rapid genetic change. Recent genome studies of many cancer types have demonstrated that clustered mutations frequently occur during carcinogenesis (Roberts et al., 2012; Nik-Zainal et al., 2012; Alexandrov et al., 2013). In addition, analyses of mutations in cancer and experiments conducted in yeast have provided evidence that damage introduced into ssdna is a primary source of clustered mutations. Among various environmental and intracellular DNA damaging agents that may contribute to cluster formation, a sub-family of AID/APOBEC cytidine deaminases known to target ssdna, is a major source of DNA damage that leads to mutation clusters in cancer (Roberts et al., 2012; Nik-Zainal et al., 2012; Alexandrov et al., 2013; Roberts et al., 2013; Taylor et al., 2013; Burns et al., 2013; Chan et al., 2012; Lada et al., 2012; Lada et al., 2013). Importantly, when APOBECs or other damaging agents are present in a cell, it is the accumulation of ssdna that becomes the limiting factor in cluster formation. Common mechanisms promoting the formation of ssdna include dysfunctional replication forks and DSBs, both of which arise by various cellular processes and conditions including oncogene-induced replication stress (Halazonetis et al., 2008). ssdna formed by 5 to 3 resection during DSB repair was suggested to be one source of kataegic events (Roberts et al., 2012; Nik-Zainal et al., 2012; Taylor et al., 2013; Yang et al., 2008). However, the switching pattern of strand coordinated mutations expected to result from bidirectional DSB resection was rarely observed suggesting that other sources of ssdna may exist (Roberts et al., 2012). In addition, multi-kilobase resection tracts that have been demonstrated in yeast (Chung et al., 2010) have never been observed in mammalian systems, which further supports the idea that alternative sources of ssdna likely exist. It has recently been demonstrated (Saini et al., 2013; Wilson et al., 2013) that ssdna accumulates during one type of DSB repair - break-induced replication (BIR), which is conserved from viruses and bacteria to eukaryotes, including human cells (Costantino et al., 2014). BIR commonly repairs DSBs possessing only one repairable end that frequently occur at collapsed replication forks or at eroded telomeres (reviewed in (Malkova and Ira, 2013)). DNA synthesis during BIR is drastically different from S-phase replication. Instead of a replication fork, BIR is driven by a migrating bubble where lagging strand synthesis is substantially delayed as compared to leading strand synthesis, which results in the accumulation of ssdna behind the replication bubble (Figure 1A) (Saini et al., 2013; Wilson et al., 2013). We hypothesized that the ssdna accumulating during BIR could be a substrate for damageinduced clustered mutations. It remained unclear, however, whether the regions of ssdna formed during BIR would be sufficiently long or stable enough to lead to clustered mutations recoverable as viable BIR repair outcomes. Here, we demonstrate in yeast that BIR is a novel source of damage-induced clustered mutations formed along the track of BIR,
3 Sakofsky et al. Page 3 as well as mutation clusters associated with additional DNA breakage and chromosomal rearrangements, similar to those commonly found in human cancers. Results Half of BIR events completed in the presence of DNA damage result in mutation clusters To test the hypothesis that BIR creates a substrate for the formation of damage-induced mutation clusters, we used a yeast strain where a galactose-inducible DSB is made at MAT in one copy of chromosome III (Deem et al., 2008). The DSB was predominately repaired by BIR (see Supplemental Results) using a second (uncut) copy of chromosome III as a template, while other outcomes including gene conversion (GC), chromosome loss (Loss) and half-crossovers (HC) occurred less frequently (Figure 1B, Table S1, see Supplemental Results for details). BIR was completed in liquid medium in the presence of 1.5mM MMS, a DNA alkylating agent with a ssdna-specific mutation signature (Yang et al., 2010). There was practically no loss of viability after treatment with MMS and the majority of DSB repair outcomes displayed an Ade + Leu - Nat R phenotype with a chromosomal structure expected from BIR (Figure 1B, 2A, Table S1). To determine whether BIR promoted the formation of damage-induced mutation clusters, we sequenced the genomes of 18 independent Ade + Leu - Nat R BIR outcomes formed in the presence of 1.5 mm MMS. Coverage maps generated by whole-genome sequencing showed that in each isolate approximately 120kbs of the rightarm portion of chromosome III from MAT to the end of the chromosome (Figure 2B) was duplicated, as expected for BIR (Deem et al., 2008; Malkova et al., 2005). We found that over 50% of the analyzed BIR outcomes (11/18) contained mutation clusters varying in length from 4kbs to 115kbs in the area of BIR on the right arm of chromosome III (Figure 2C, 2D, Table S2A, S3). The association between BIR and mutation clusters could in principle be even higher, since some isolates that lacked clusters could have resulted from interrupted BIR that led to half-crossovers (HCs) which segregated with an intact copy of the donor chromosome during mitosis (Deem et al., 2008); an outcome that would be indistinguishable from BIR. Also, the interruption of BIR could generate varying lengths of ssdna tracts providing one explanation for the variations in mutation cluster length. The median density of mutations in clusters was 1 mutation per 6.3kb, which was 900 times higher than the density of scattered mutations in the rest of the genome (Table S2A, S3). In addition to 11 clusters found in a relatively small (<1% of the yeast genome) region surrounding the HO-break site on chromosome III, we identified only 4 clusters occurring on one of the other 15 chromosomes (Table S2A, S3) (P-value for random co-localization <1E-15). No mutation clusters were found in the 6 whole-genome sequenced clones that resulted from BIR repair in the absence of MMS (Table S2A). Taken together, our data indicate that the DNA region associated with BIR is a target for damage-induced clustered mutations. Based on what is known about the mechanism of BIR, we hypothesized that clustered mutations resulted from lesions induced by MMS in ssdna formed during BIR as a result of asynchronous leading and lagging strand synthesis (Saini et al., 2013) or during 5 to 3 DSB resection preceding BIR (Chung et al., 2010) (Figure 1A). BIR synthesis begins on the 5 -side of the DSB at the point of strand invasion and can continue for more than 100kbs
4 Sakofsky et al. Page 4 until it reaches the end of the chromosome. (5 - and 3 -sides relative to the DSB are defined along the top (Watson) DNA strand and correspond respectively to the left and right sides on Figure 2D). We observed that BIR-induced clusters occurred on both sides of the break (6/11) as well as only on one side i.e., either on the 3 -side (4/11) or on the 5 -side (1/11). Mutations clustered on the 5 -side of the break could have resulted from damage to ssdna that was generated either by resection or by BIR synthesis. Importantly, the mutation clusters on the 3 -side of the break were located along the track of BIR, and therefore likely stemmed from BIR synthesis. Based on the established signature of MMS mutagenesis in ssdna (Yang et al., 2010; Roberts et al., 2012), we expected that clustered mutations would most frequently result from N3-methyl cytosine lesions in the ssdna strand, and N1-methyl adenine lesions would be the second most frequent cause of clustered mutations. Note: the ssdna specific MMS mutations of cytosines have no particular signature, such as TpC or CpG mutations reported in cancer genomes that are assigned to APOBEC and 5-methyl cytosine deamination in ssdna, respectively (Nik-Zainal et al., 2012; Roberts et al., 2012; Burns et al., 2013; Nikolaev et al., 2012). During BIR, the same DNA strand (Watson or top strand) was expected to be single-stranded regardless of whether it was formed by either 5 3 resection or by asynchronous synthesis of leading and lagging strands (Figure 1A). Thus, cytosines in mutated C:G pairs (and possibly adenines in mutated A:T pairs) were expected to be found mostly in this strand of DNA. Indeed, mutations in BIR-induced clusters on either side of the DSB were biased towards C's located in the Watson strand (P= and 8.02E-9, 5 and 3 of the break respectively, by two-sided goodness-of-fit test) (Table S2B). There was also an overall statistically significant bias towards A's in the Watson strand despite the smaller number of changes in A:T pairs (P= ) (Table S2B). As expected for cytosine-specific MMS mutagenesis in ssdna, changes in neighboring mutations of C:G pairs were strand-coordinated (i.e., mutations observed in cytosines in the same strand (P =2.60E-9 by goodness-of-fit test) (Table S4), and C to T and C to A mutations prevailed over C to G mutations (P=4.32E-13, by two-sided goodness-of-fit) (Table S2B), in good agreement with the known mutation signature of MMS in ssdna (Delaney and Essigmann, 2004; Yang et al., 2010). We conclude that ssdna formed during BIR synthesis is vulnerable to hyper-mutation by low levels of alkylating damage resulting in mutation clusters. The conservative inheritance of newly synthesized DNA during BIR (Saini et al., 2013; Donnianni and Symington, 2013) should result in the majority of clustered mutations being heterozygous and located only in the recipient. Indeed, 61 out of 64 clustered mutations were heterozygous (Table S2A). In addition, the vast majority of BIR-associated clustered mutations occurred only on the recipient chromosome (55 out of 64 mutations; Figure 2E, 2F, Table S2A). Altogether our data show that ssdna accumulated during uncoupled leading strand synthesis in the course of BIR is a source of damage-induced clustered mutations.
5 Sakofsky et al. Page 5 Complex mutation clusters associated with DNA breakage are formed by BIR in the presence of an increased level of DNA damage To further evaluate the role of DNA damage in BIR-induced mutation clusters, we sequenced 30 independent BIR (Ade + Leu - ) isolates that were formed in the presence of higher concentrations of MMS (6 mm). In addition to Ade + Leu -, we used the information about other phenotypes including Nat status and the presence of additional rearrangements in chromosome III to choose isolates representing a complete spectrum of changes (see Supplemental Experimental Procedures for details). We found that approximately half (17/30) of the analyzed isolates contained mutation clusters, whose location strongly associated with the tract of BIR (P< comparing 13 clusters on chromosome III (2.5% of the genome) to 8 clusters on all other chromosomes combined (97.5% of the genome) by binomial test; Figure 3, Table S2A). As in the case of 1.5mM MMS, the majority of clusters (10 out of 13) were located on both sides of the break (Table S2), contained mutations that were biased toward cytosines and adenines in the Watson strand (P = 6.26E-14 and 0.021, respectively by two-sided goodness-of-fit test) (Table S2B), and were strand coordinated (P = 2.38E-9 by goodness-of-fit test) (Figure 3C, Table S4). In addition, BIR-induced clusters were equally frequent among repair outcomes with and without chromosome III rearrangements. Surprisingly though, we observed that a high concentration of MMS changed the distribution of mutation clusters between the donor and recipient molecules. In particular, unlike clusters from 1.5mM MMS exposure, only 2 out of 13 clusters (in ALM_4 and ALM_11) had heterozygous mutations limited to the recipient chromosome, while the majority (10 out of 13) of BIR clusters had heterozygous and/or homozygous mutations in both the recipient and donor chromosomes (Figure 3D, Figure S1, Table S2A). We called such clusters complex and proposed that they formed as a result of a disruption during BIR by MMS-induced damage in dsdna located in front of the BIR bubble. This event could lead to resolution of the HJ migrating behind the BIR bubble causing the donor chromosome to break, which could then initiate a second round of BIR where clustered mutations in the donor could form (Figure 4H-J). To explain the formation of complex clusters in individual outcomes, we utilized the results from a combination of analyses (phenotype, chromosome size, whole genome sequencing and Sanger sequencing). Our results suggested that the formation of complex mutation clusters in all repair outcomes proceeded in general, through the following steps (Figure 4, Figures S2-S4): (i) DSB resection and invasion of the recipient into the donor chromosome initiating BIR synthesis (primary BIR) producing clustered mutations in the recipient from damage in ssdna, (ii) interruption of primary BIR leading to a half-crossover event (similar to (Deem et al., 2008; Smith et al., 2009)) and a fragmented donor chromosome, (iii) resection and invasion of the fragmented donor into homologous DNA initiating a secondary BIR event and generating clustered mutations in the donor chromosome from damage in ssdna. The exact details of how these steps were completed varied among different repair outcomes, which can explain the variety of phenotypes observed in the outcomes (see Supplemental Results for details). For example, the fact that ALM_24 was Nat S and had homozygous clustered mutations while ALM_1 was Nat R and had heterozygous clustered
6 Sakofsky et al. Page 6 Discussion mutations suggested that differences existed between these outcomes with respect to the extent of DSB resection and the location of strand invasion (Figure S2). We propose that primary BIR in ALM_24 was preceded by extensive resection and strand invasion into the donor chromosome at a position centromere proximal to NAT leading to a Nat S phenotype, while strand invasion in ALM_1 occurred telomere proximal to NAT, which resulted in a Nat R phenotype. Furthermore, in ALM_24 the broken donor invaded the HC product at a position centromere proximal to its mutation cluster allowing the mutation cluster to be copied during secondary BIR, thus forming homozygous mutations. For outcome ALM_1, which had heterozygous mutations instead of homozygous mutations, we propose that the broken donor invaded the HC at a position centromere distal to the clustered mutations in the recipient, thus precluding the mutation cluster from being copied. Heterozygous mutations could result from MMS damage introduced in ssdna formed during DNA resection of the donor and/or during secondary BIR. Alternatively, heterozygous mutations in ALM_1 could have also formed by the invasion of the broken donor into another homologous DNA template (i.e., sister chromatid) instead of the HC product when initiating secondary BIR. The use of a sister chromatid as a template for secondary BIR was likely to occur in other isolates and is strongly supported by the analysis of ALM_18 (Figure 3D, S1C, S3), where a 20kb deletion between two pairs of Ty elements, FS1 and FS2, likely resulted from recombination between FS1 of the broken donor chromosome and FS2 of a sister chromatid (see Supplemental Results for details). Thus far, our data indicate that complex clusters form by at least one HC followed by a secondary BIR event. Additional rounds of HCs and BIR events could however lead to even more complex outcomes. This was supported by the analysis of ALM_26, which likely resulted from the following events: formation of 6 clustered mutations, two HC events, and an ectopic recombination event that led to a chromosomal rearrangement (Figure 3D, S1D, S4; also see Supplemental Results for details). Overall our data suggest that increased levels of DNA damage can disrupt BIR and result in multiple rounds of repair leading to the formation of complex clusters that can be associated with chromosomal rearrangements. Recent studies have revealed that cancer genomes frequently contain clusters of strandcoordinated mutations (also called kataegis) caused by ssdna specific enzymes APOBEC cytidine deaminases (Roberts et al., 2012; Nik-Zainal et al., 2012; Alexandrov et al., 2013; Roberts et al., 2013; Burns et al., 2013). A fraction of kataegic clusters co-localized with breakpoints of chromosomal rearrangements (Nik-Zainal et al., 2012; Roberts et al., 2012). However, the source of ssdna and the mechanistic connection between clusters and chromosomal rearrangements remained unexplained. Here, we demonstrate that an unusual type of DNA synthesis, break-induced replication (BIR) completed in the presence of MMS, leads to repair outcomes containing clustered mutations 50% of the time. Importantly these clusters were frequently associated with DNA breakage and chromosomal rearrangements, suggesting that DNA damage to BIR intermediates may account for both clustering of mutations and associated rearrangements that characterize kataegis in human cancer.
7 Sakofsky et al. Page 7 We propose that the formation of mutation clusters and chromosomal rearrangements associated with BIR result from the interplay between DNA damage and two specific structural features of BIR replication (Figure 4B-E). First, ssdna accumulated during BIR from asynchronous leading and lagging strand synthesis and from long resection prior to BIR initiation provides the substrate for clustered mutagenesis. Second, the HJ migrating behind the BIR bubble is a substrate for resolution, which is promoted by stalling of BIR synthesis and leads to breaking and re-invading events that can manifest as half-crossovers, HC-initiated cascades, and other chromosomal rearrangements (Deem et al., 2008; Smith et al., 2009; Vasan et al., 2014). We propose that the formation of mutation clusters in this study were promoted by MMS-induced damage in ssdna (N3-me C and to a lesser extent N1-me A) (Yang et al., 2010). We further propose that stalling of the replication bubble and the consequent breakage of DNA through HJ resolution was likely induced by either N3-me A, a common MMS-induced lesion in dsdna, or abasic sites created by unfinished repair of MMS damage (Yang et al., 2010). The idea that two destabilizing outcomes of BIR- mutation clusters and DNA breakage promoting chromosomal rearrangements- are stimulated by different DNA lesions, predicts that these two outcomes of BIR could be independently modulated and therefore together can give rise to a variety of outcomes. In agreement with this idea, different doses of MMS resulted in clusters with varying length and complexity. We propose that BIR is not only a prominent source of damage-induced mutation clusters in yeast, but also in cancer genomes including clusters associated with DNA breakage and rearrangements. The variety of mutation clusters found in cancer such as mutation clusters with and without various chromosomal rearrangements (i.e., LOH, CNV and translocations), could each result from combining DNA damage induced by APOBEC family enzymes (modeled in our experiments by MMS) with DNA breakage resulting from other sources of DNA damage or from special features of cancer cells such as checkpoint defects. Supporting this, our recent data obtained in yeast demonstrated that checkpoint defects can promote cascades of chromosomal breakage and rearrangements in cells undergoing BIR even in the absence of additional DNA lesions (Vasan et al., 2014). In addition, Costantino and colleagues (Costantino et al., 2014) have recently demonstrated that a massive collapse of replication forks resulting from the overexpression of an oncogene in human cancer cells led to the initiation of BIR, which in turn promoted a burst of chromosomal rearrangements known to lead to cancer. Our results predict that the exposure of such cells to DNA damage will lead to kataegic mutation clusters associated with chromosomal rearrangements. Moreover, short stretches of ssdna resulting from interrupted BIR could contribute to some of the genome-wide mutagenesis in individual cancers where APOBEC mutagenesis in clusters often represents only a small fraction (e.g. less than 10%) of all mutations in APOBEC motifs (Roberts et al., 2012; Alexandrov et al., 2013). Indeed the rates of single mutations, including mutations with APOBEC motifs were increased in the immediate vicinity of rearrangement breakpoints in many cancer samples (Drier et al., 2013), suggesting DSB repair processes including BIR may be targets for scattered mutations as well as for mutation clusters. Additional APOBEC-induced single mutations that are not
8 Sakofsky et al. Page 8 located close to the positions of rearrangement breakpoints could also result from BIR since not all BIR events are associated with detectable rearrangements. All together, this study suggests that BIR could be one of the most powerful sources of destabilizing genomes of somatic cells, capable of producing a wide range of multiple simultaneous genomic changes. We expect that future studies will reveal the scale to which BIR contributes to genome destabilization in both normal and cancer cells as well as uncover how such instability is regulated. Experimental Procedures Yeast strains and growth conditions All experiments were performed using yeast strain AM1003 (Deem et al., 2008). Rich medium (yeast extract-peptone-dextrose [YEPD]) and synthetic complete medium were made as described in (Guthrie and Fink, 1991). YEP-lactate (YEP-Lac) and YEP-galactose (YEP-Gal) used for DSB induction were similar to (Deem et al., 2008). MMS was added to YEP-Gal to the final concentrations of 1.5mM and 6mM. See Supplemental Experimental Procedures for further details. Characterization of DSB repair isolates DSB repair isolates were characterized by phenotype, similar to (Deem et al., 2008). The chromosomal structure of BIR (Ade + Leu - ) outcomes to determine the presence of chromosomal rearrangements was analyzed using PFGE, similar to (Deem et al., 2008) (see Supplemental Experimental Procedures for details). The preparation of yeast genomic DNA for sequencing, library construction, mapping of reads to a reference genome, and identification of mutations were performed similar to (Roberts et al., 2012). See Supplemental Experimental Procedures for details of whole genome sequencing, reference sequence construction, and mutation calling. The identification of mutation clusters and analyses of strand bias, strand-coordination, and co-localization of mutation clusters with breakpoints were performed similar to (Roberts et al., 2012). Coverage maps for chromosome III were created using CLC Genomics Workbench 6.0. The assignment of mutations to the recipient or donor copy of chromosome III was performed similar to (Saini et al., 2013). See Supplemental Experimental Procedures for further details. Supplementary Material Acknowledgments Refer to Web version on PubMed Central for supplementary material. This work was funded by NIH grants R01GM to AM, and 5R01GM to P.A.M, and by the Intramural Research Program of the NIH, National Institute of Environmental Health Sciences, (project ES to M.A.R). S.A.R. was supported by NIH Pathway to Independence Award K99ES C.J.S. was supported in part by the Department of Biology at IUPUI.
9 Sakofsky et al. Page 9 References Alexandrov LB, Nik-Zainal S, Wedge DC, Aparicio SA, Behjati S, Biankin AV, Bignell GR, Bolli N, Borg A, Borresen-Dale AL, et al. Signatures of mutational processes in human cancer. Nature. 2013; 500: [PubMed: ] Burns MB, Lackey L, Carpenter MA, Rathore A, Land AM, Leonard B, Refsland EW, Kotandeniya D, Tretyakova N, Nikas JB, et al. APOBEC3B is an enzymatic source of mutation in breast cancer. Nature. 2013; 494: [PubMed: ] Camps M, Herman A, Loh E, Loeb LA. Genetic constraints on protein evolution. Crit Rev Biochem Mol Biol. 2007; 42: [PubMed: ] Chan K, Sterling JF, Roberts SA, Bhagwat AS, Resnick MA, Gordenin DA. Base damage within single-strand DNA underlies in vivo hypermutability induced by a ubiquitous environmental agent. PLoS Genetics. 2012; 8:e [PubMed: ] Chung WH, Zhu Z, Papusha A, Malkova A, Ira G. Defective resection at DNA double-strand breaks leads to de novo telomere formation and enhances gene targeting. PLoS Genetics. 2010; 6:e [PubMed: ] Costantino L, Sotiriou SK, Rantala JK, Magin S, Mladenov E, Helleday T, Haber JE, Iliakis G, Kallioniemi O, Halazonetis TD. Break-Induced replication repair of damaged forks induces genomic duplications in human cells. Science. 2014; 343: Deem A, Barker K, Vanhulle K, Downing B, Vayl A, Malkova A. Defective break-induced replication leads to half-crossovers in Saccharomyces cerevisiae. Genetics. 2008; 179: [PubMed: ] Delaney JC, Essigmann JM. Mutagenesis, genotoxicity, and repair of 1-methyladenine, 3- alkylcytosines, 1-methylguanine, and 3-methylthymine in alkb Escherichia coli. Proceedings of the National Academy of Sciences of the United States of America. 2004; 101: [PubMed: ] Donnianni RA, Symington LS. Break-induced replication occurs by conservative DNA synthesis. Proc Natl Acad Sci USA. 2013; 33: [PubMed: ] Drier Y, Lawrence MS, Carter SL, Stewart C, Gabriel SB, Lander ES, Meyerson M, Beroukhim R, Getz G. Somatic rearrangements across cancer reveal classes of samples with distinct patterns of DNA breakage and rearrangement-induced hypermutability. Genome research. 2013; 23: [PubMed: ] Guthrie, C.; Fink, G. Guide to Yeast Genetics and Molecular Biology. San Diego: Academic Press; Halazonetis TD, Gorgoulis VG, Bartek J. An oncogene-induced DNA damage model for cancer development. Science. 2008; 319: [PubMed: ] Lada AG, Dhar A, Boissy RJ, Hirano M, Rubel AA, Rogozin IB, Pavlov YI. AID/APOBEC cytosine deaminase induces genome-wide kataegis. Biology direct. 2012; 7:47. [PubMed: ] Lada AG, Stepchenkova EI, Waisertreiger IS, Noskov VN, Dhar A, Eudy JD, Boissy RJ, Hirano M, Rogozin IB, Pavlov YI. Genome-wide mutation avalanches induced in diploid yeast cells by a base analog or an APOBEC deaminase. PLoS Genetics. 2013; 9:e [PubMed: ] Malkova A, Naylor ML, Yamaguchi M, Ira G, Haber JE. RAD51-dependent break-induced replication differs in kinetics and checkpoint responses from RAD51-mediated gene conversion. Molecular and cellular biology. 2005; 25: [PubMed: ] Malkova A, Ira G. Break-induced replication functions and molecular mechanism. Current Opinion in Genetics and Development. 2013; 23: [PubMed: ] Nikolaev SI, Sotiriou SK, Pateras IS, Santoni F, Sougioultzis S, Edgren H, Almusa H, Robyr D, Guipponi M, Saarela J, Gorgoulis VG, Antonarakis SE, Halazonetis TD. A single-nucleotide substitution mutator phenotype revealed by exome sequencing of human colon adenomas. Cancer Res. 2012; 72: [PubMed: ] Nik-Zainal S, Alexandrov LB, Wedge DC, Van Loo P, Greenman CD, Raine K, Jones D, Hinton J, Marshall J, Stebbings LA, et al. Mutational Processes Molding the Genomes of 21 Breast Cancers. Cell. 2012; 149: [PubMed: ]
10 Sakofsky et al. Page 10 Roberts SA, Lawrence MS, Klimczak LJ, Grimm SA, Fargo D, Stojanov P, Kiezun A, Kryukov GV, Carter SL, Saksena G, et al. An APOBEC cytidine deaminase mutagenesis pattern is widespread in human cancers. Nat Genet. 2013; 45: [PubMed: ] Roberts SA, Sterling J, Thompson C, Harris S, Mav D, Shah R, Klimczak LJ, Kryukov GV, Malc E, Mieczkowski PA, et al. Clustered mutations in yeast and in human cancers can arise from damaged long single-strand DNA regions. Molecular Cell. 2012; 46: [PubMed: ] Saini N, Ramakrishnan S, Elango R, Ayyar S, Zhang Y, Deem A, Ira G, Haber JE, Lobachev KS, Malkova A. Migrating bubble during break-induced replication drives conservative DNA synthesis. Nature. 2013; 502: [PubMed: ] Smith CE, Lam AF, Symington LS. Aberrant double-strand break repair resulting in half-crossovers in mutants defective for Rad51 or the DNA polymerase delta complex. Mol Cell Biol. 2009; 29: [PubMed: ] Taylor BJ, Nik-Zainal S, Wu YL, Stebbings LA, Raine K, Campbell PJ, Rada C, Stratton MR, Neuberger MS. DNA deaminases induce break-associated mutation showers with implication of APOBEC3B and 3A in breast cancer kataegis. Elife. 2013; 2:e [PubMed: ] Vasan S, Deem A, Ramakrishnan S, Argueso J, Malkova A. Cascades of genetic instability resulting from compromised break-induced replication. PLoS genetics. 2014; 10:e [PubMed: ] Wilson MA, Kwon Y, Xu Y, Chung WH, Chi P, Niu H, Mayle R, Chen X, Malkova A, Sung P, et al. Pif1 helicase and Poldelta promote recombination-coupled DNA synthesis via bubble migration. Nature. 2013; 502: [PubMed: ] Yang Y, Gordenin DA, Resnick MA. A single-strand specific lesion drives MMS-induced hypermutability at a double-strand break in yeast. DNA repair. 2010; 9: [PubMed: ] Yang Y, Sterling J, Storici F, Resnick MA, Gordenin DA. Hypermutability of damaged single-strand DNA formed at double-strand breaks and uncapped telomeres in yeast Saccharomyces cerevisiae. PLoS genetics. 2008; 4:e [PubMed: ]
11 Sakofsky et al. Page 11 Highlights Damage in ssdna formed during BIR can cause simultaneous clustered mutations. Mutation clusters occur in ssdna formed during uncoupled conservative DNA synthesis. BIR-generated mutation clusters co-localize with additional breaks and rearrangements. BIR-generated mutation clusters closely resemble kataegic clusters in cancer.
12 Sakofsky et al. Page 12 Figure 1. DSB repair by BIR (A) Model of BIR. Dotted lines: newly synthesized DNA. (B) Experimental system to study BIR in yeast. The percent occurrence of the main DSB repair outcomes and their relevant phenotypes are indicated with total isolates scored in parenthesis. Asterisks: statistically significant increase of half-crossovers in MMS versus no-mms. See Supplemental Experimental Procedures and Table S1 for details.
13 Sakofsky et al. Page 13 Figure 2. Mutagenesis associated with BIR in the presence of 1.5mM MMS (A) The structure of one representative outcome (ALM_31). Left: ethidium bromide-stained PFGE gel. Middle and right: Southern blot analysis of PFGE gel using ADE1- and ADE3- specific probes, respectively. (B) Coverage of Illumina sequencing reads for BIR event (ALM_31) is increased 2x centromere distal to MAT (positions >194180bp) as compared to the parental strain. (C) MMS-induced mutations (blue lines) in ALM_31. Enlarged: mutation cluster on the track of BIR. (D) Clustered mutations in Ade + Leu - BIR isolates. Positions of mutated bases (colored circles) are depicted along the chromosome III reference. (E and F) Select BIR-associated mutation clusters with mutations located in recipient (E) or donor (F) chromosomes.
14 Sakofsky et al. Page 14 Figure 3. Mutagenesis associated with BIR in the presence of 6mM MMS (A) Chromosome structures of representative BIR outcomes. Upper panel: ethidium bromide stained PFGE gel. Middle and lower panels: Southern blot analysis with ADE1- or ADE3- specific probes, respectively. (B) Coverage of Illumina sequencing reads derived from representative Ade + Leu - isolates (2x and 3x: fold increases as compared with the parental strain). See Supplemental Experimental Procedures for details. (C) Clustered mutations on chromosome III in Ade + Leu - isolates indicated as in Figure 2D. (D) Distribution of mutations between the recipient and donor chromosomes for the representative isolates indicated in (C). Complete information about cluster structure and formation is given in Figures S1-S4.
15 Sakofsky et al. Page 15 Figure 4. Model of BIR-induced cluster formation DNA lesions (red stars) from MMS are shown in ssdna formed by resection and BIR synthesis. Red squares: mutations from TLS bypass. Scissors: Resolution of HJ-like structure. Details of resolution are unknown. Yellow rectangle: damage in dsdna. One possible scenario of lagging strand synthesis is shown. See text and Supplemental Figures S2, S3 and S4 for details.
Repair of Broken Chromosomes and Maintenance of Chromosome Stability
 Repair of Broken Chromosomes and Maintenance of Chromosome Stability Jim Haber Brandeis University Genome instability in tumor cells Truncations Translocations Inversions Duplications Amplifications Deletions
Repair of Broken Chromosomes and Maintenance of Chromosome Stability Jim Haber Brandeis University Genome instability in tumor cells Truncations Translocations Inversions Duplications Amplifications Deletions
Package ClusteredMutations
 Package ClusteredMutations April 29, 2016 Version 1.0.1 Date 2016-04-28 Title Location and Visualization of Clustered Somatic Mutations Depends seriation Description Identification and visualization of
Package ClusteredMutations April 29, 2016 Version 1.0.1 Date 2016-04-28 Title Location and Visualization of Clustered Somatic Mutations Depends seriation Description Identification and visualization of
Polyomaviridae. Spring
 Polyomaviridae Spring 2002 331 Antibody Prevalence for BK & JC Viruses Spring 2002 332 Polyoma Viruses General characteristics Papovaviridae: PA - papilloma; PO - polyoma; VA - vacuolating agent a. 45nm
Polyomaviridae Spring 2002 331 Antibody Prevalence for BK & JC Viruses Spring 2002 332 Polyoma Viruses General characteristics Papovaviridae: PA - papilloma; PO - polyoma; VA - vacuolating agent a. 45nm
Genomic structural variation
 Genomic structural variation Mario Cáceres The new genomic variation DNA sequence differs across individuals much more than researchers had suspected through structural changes A huge amount of structural
Genomic structural variation Mario Cáceres The new genomic variation DNA sequence differs across individuals much more than researchers had suspected through structural changes A huge amount of structural
A. Incorrect! Cells contain the units of genetic they are not the unit of heredity.
 MCAT Biology Problem Drill PS07: Mendelian Genetics Question No. 1 of 10 Question 1. The smallest unit of heredity is. Question #01 (A) Cell (B) Gene (C) Chromosome (D) Allele Cells contain the units of
MCAT Biology Problem Drill PS07: Mendelian Genetics Question No. 1 of 10 Question 1. The smallest unit of heredity is. Question #01 (A) Cell (B) Gene (C) Chromosome (D) Allele Cells contain the units of
1) DNA unzips - hydrogen bonds between base pairs are broken by special enzymes.
 Biology 12 Cell Cycle To divide, a cell must complete several important tasks: it must grow, during which it performs protein synthesis (G1 phase) replicate its genetic material /DNA (S phase), and physically
Biology 12 Cell Cycle To divide, a cell must complete several important tasks: it must grow, during which it performs protein synthesis (G1 phase) replicate its genetic material /DNA (S phase), and physically
Nature Biotechnology: doi: /nbt.1904
 Supplementary Information Comparison between assembly-based SV calls and array CGH results Genome-wide array assessment of copy number changes, such as array comparative genomic hybridization (acgh), is
Supplementary Information Comparison between assembly-based SV calls and array CGH results Genome-wide array assessment of copy number changes, such as array comparative genomic hybridization (acgh), is
Chromothripsis: A New Mechanism For Tumorigenesis? i Fellow s Conference Cheryl Carlson 6/10/2011
 Chromothripsis: A New Mechanism For Tumorigenesis? i Fellow s Conference Cheryl Carlson 6/10/2011 Massive Genomic Rearrangement Acquired in a Single Catastrophic Event during Cancer Development Cell 144,
Chromothripsis: A New Mechanism For Tumorigenesis? i Fellow s Conference Cheryl Carlson 6/10/2011 Massive Genomic Rearrangement Acquired in a Single Catastrophic Event during Cancer Development Cell 144,
Chapter 9. Cells Grow and Reproduce
 Chapter 9 Cells Grow and Reproduce DNA Replication DNA polymerase Addition of a nucleotide to the 3 end of a growing strand Use dntps as substrate Release of pyrophosphate Initiation of Replication Replication
Chapter 9 Cells Grow and Reproduce DNA Replication DNA polymerase Addition of a nucleotide to the 3 end of a growing strand Use dntps as substrate Release of pyrophosphate Initiation of Replication Replication
2015 AP Biology Unit #4 Test Cell Communication, Cancer, Heredity and The Cell Cycle Week of 30 November
 Class: Date: 2015 AP Biology Unit #4 Test Cell Communication, Cancer, Heredity and The Cell Cycle Week of 30 November Multiple Choice 1 point each Identify the choice that best completes the statement
Class: Date: 2015 AP Biology Unit #4 Test Cell Communication, Cancer, Heredity and The Cell Cycle Week of 30 November Multiple Choice 1 point each Identify the choice that best completes the statement
Single-strand DNA library preparation improves sequencing of formalin-fixed and paraffin-embedded (FFPE) cancer DNA
 www.impactjournals.com/oncotarget/ Oncotarget, Supplementary Materials 2016 Single-strand DNA library preparation improves sequencing of formalin-fixed and paraffin-embedded (FFPE) DNA Supplementary Materials
www.impactjournals.com/oncotarget/ Oncotarget, Supplementary Materials 2016 Single-strand DNA library preparation improves sequencing of formalin-fixed and paraffin-embedded (FFPE) DNA Supplementary Materials
CURRICULUM VITAE. Artem Georgievich Lada
 CURRICULUM VITAE Artem Georgievich Lada Eppley Institute for Research in Cancer and Allied Diseases, ESH7026 986805 Nebraska Medical Center Omaha NE 68198-6805 United States Phones: Work +1 (402) 559-6062;
CURRICULUM VITAE Artem Georgievich Lada Eppley Institute for Research in Cancer and Allied Diseases, ESH7026 986805 Nebraska Medical Center Omaha NE 68198-6805 United States Phones: Work +1 (402) 559-6062;
Variations in Chromosome Structure & Function. Ch. 8
 Variations in Chromosome Structure & Function Ch. 8 1 INTRODUCTION! Genetic variation refers to differences between members of the same species or those of different species Allelic variations are due
Variations in Chromosome Structure & Function Ch. 8 1 INTRODUCTION! Genetic variation refers to differences between members of the same species or those of different species Allelic variations are due
Introduction to Cancer Biology
 Introduction to Cancer Biology Robin Hesketh Multiple choice questions (choose the one correct answer from the five choices) Which ONE of the following is a tumour suppressor? a. AKT b. APC c. BCL2 d.
Introduction to Cancer Biology Robin Hesketh Multiple choice questions (choose the one correct answer from the five choices) Which ONE of the following is a tumour suppressor? a. AKT b. APC c. BCL2 d.
p.r623c p.p976l p.d2847fs p.t2671 p.d2847fs p.r2922w p.r2370h p.c1201y p.a868v p.s952* RING_C BP PHD Cbp HAT_KAT11
 ARID2 p.r623c KMT2D p.v650fs p.p976l p.r2922w p.l1212r p.d1400h DNA binding RFX DNA binding Zinc finger KMT2C p.a51s p.d372v p.c1103* p.d2847fs p.t2671 p.d2847fs p.r4586h PHD/ RING DHHC/ PHD PHD FYR N
ARID2 p.r623c KMT2D p.v650fs p.p976l p.r2922w p.l1212r p.d1400h DNA binding RFX DNA binding Zinc finger KMT2C p.a51s p.d372v p.c1103* p.d2847fs p.t2671 p.d2847fs p.r4586h PHD/ RING DHHC/ PHD PHD FYR N
Chapter 2. Mitosis and Meiosis
 Chapter 2. Mitosis and Meiosis Chromosome Theory of Heredity What structures within cells correspond to genes? The development of genetics took a major step forward by accepting the notion that the genes
Chapter 2. Mitosis and Meiosis Chromosome Theory of Heredity What structures within cells correspond to genes? The development of genetics took a major step forward by accepting the notion that the genes
Asingle inherited mutant gene may be enough to
 396 Cancer Inheritance STEVEN A. FRANK Asingle inherited mutant gene may be enough to cause a very high cancer risk. Single-mutation cases have provided much insight into the genetic basis of carcinogenesis,
396 Cancer Inheritance STEVEN A. FRANK Asingle inherited mutant gene may be enough to cause a very high cancer risk. Single-mutation cases have provided much insight into the genetic basis of carcinogenesis,
DAPI ASY1 DAPI/ASY1 DAPI RAD51 DAPI/RAD51. Supplementary Figure 1. Additional information on meiosis in R. pubera. a) The
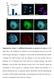 a % 10 Number of crossover per bivalent b 0 1 c DAPI/telomere 80 1 60 40 1 2 20 d 0 0 1 2 >=3 DAPI ASY1 DAPI/ASY1 e DAPI RAD51 DAPI/RAD51 Supplementary Figure 1. Additional information on meiosis in R.
a % 10 Number of crossover per bivalent b 0 1 c DAPI/telomere 80 1 60 40 1 2 20 d 0 0 1 2 >=3 DAPI ASY1 DAPI/ASY1 e DAPI RAD51 DAPI/RAD51 Supplementary Figure 1. Additional information on meiosis in R.
Introduction to Genetics
 Introduction to Genetics Table of contents Chromosome DNA Protein synthesis Mutation Genetic disorder Relationship between genes and cancer Genetic testing Technical concern 2 All living organisms consist
Introduction to Genetics Table of contents Chromosome DNA Protein synthesis Mutation Genetic disorder Relationship between genes and cancer Genetic testing Technical concern 2 All living organisms consist
Generation of antibody diversity October 18, Ram Savan
 Generation of antibody diversity October 18, 2016 Ram Savan savanram@uw.edu 441 Lecture #10 Slide 1 of 30 Three lectures on antigen receptors Part 1 : Structural features of the BCR and TCR Janeway Chapter
Generation of antibody diversity October 18, 2016 Ram Savan savanram@uw.edu 441 Lecture #10 Slide 1 of 30 Three lectures on antigen receptors Part 1 : Structural features of the BCR and TCR Janeway Chapter
CHAPTER 17 CHROMOSOME REARRANGEMENTS
 CHROMOSOME REARRANGEMENTS CHAPTER 17 Figure 1. Comparing an ideogram of the human chromosome 2 to the equivalent chromosomes in chimpanzees, we notice that the human chromosome 2 likely came from a fusion
CHROMOSOME REARRANGEMENTS CHAPTER 17 Figure 1. Comparing an ideogram of the human chromosome 2 to the equivalent chromosomes in chimpanzees, we notice that the human chromosome 2 likely came from a fusion
An APOBEC3A hypermutation signature is distinguishable from the signature of background mutagenesis by APOBEC3B in human cancers
 An APOBEC3A hypermutation signature is distinguishable from the signature of background mutagenesis by APOBEC3B in human cancers The Harvard community has made this article openly available. Please share
An APOBEC3A hypermutation signature is distinguishable from the signature of background mutagenesis by APOBEC3B in human cancers The Harvard community has made this article openly available. Please share
Structural Variation and Medical Genomics
 Structural Variation and Medical Genomics Andrew King Department of Biomedical Informatics July 8, 2014 You already know about small scale genetic mutations Single nucleotide polymorphism (SNPs) Deletions,
Structural Variation and Medical Genomics Andrew King Department of Biomedical Informatics July 8, 2014 You already know about small scale genetic mutations Single nucleotide polymorphism (SNPs) Deletions,
BIOLOGY 111. CHAPTER 9: The Links in Life s Chain Genetics and Cell Division
 BIOLOGY 111 CHAPTER 9: The Links in Life s Chain Genetics and Cell Division The Links in Life s Chain: Genetics and Cell Division 9.1 An Introduction to Genetics 9.2 An Introduction to Cell Division 9.3
BIOLOGY 111 CHAPTER 9: The Links in Life s Chain Genetics and Cell Division The Links in Life s Chain: Genetics and Cell Division 9.1 An Introduction to Genetics 9.2 An Introduction to Cell Division 9.3
Relationship between genomic features and distributions of RS1 and RS3 rearrangements in breast cancer genomes.
 Supplementary Figure 1 Relationship between genomic features and distributions of RS1 and RS3 rearrangements in breast cancer genomes. (a,b) Values of coefficients associated with genomic features, separately
Supplementary Figure 1 Relationship between genomic features and distributions of RS1 and RS3 rearrangements in breast cancer genomes. (a,b) Values of coefficients associated with genomic features, separately
The Chromosomal Basis of Inheritance
 LECTURE PRESENTATIONS For CAMPBELL BIOLOGY, NINTH EDITION Jane B. Reece, Lisa A. Urry, Michael L. Cain, Steven A. Wasserman, Peter V. Minorsky, Robert B. Jackson Chapter 15 The Chromosomal Basis of Inheritance
LECTURE PRESENTATIONS For CAMPBELL BIOLOGY, NINTH EDITION Jane B. Reece, Lisa A. Urry, Michael L. Cain, Steven A. Wasserman, Peter V. Minorsky, Robert B. Jackson Chapter 15 The Chromosomal Basis of Inheritance
Lecture -2- Environmental Biotechnology
 Lecture -2-1-General Bioassay in pollution Monitoring 1 1 Genotoxicity test At the early testing stages, the genotoxicity assays for predicting potential heritable germ cell damage are the same as used
Lecture -2-1-General Bioassay in pollution Monitoring 1 1 Genotoxicity test At the early testing stages, the genotoxicity assays for predicting potential heritable germ cell damage are the same as used
Regulators of Cell Cycle Progression
 Regulators of Cell Cycle Progression Studies of Cdk s and cyclins in genetically modified mice reveal a high level of plasticity, allowing different cyclins and Cdk s to compensate for the loss of one
Regulators of Cell Cycle Progression Studies of Cdk s and cyclins in genetically modified mice reveal a high level of plasticity, allowing different cyclins and Cdk s to compensate for the loss of one
LESSON 3.2 WORKBOOK. How do normal cells become cancer cells? Workbook Lesson 3.2
 For a complete list of defined terms, see the Glossary. Transformation the process by which a cell acquires characteristics of a tumor cell. LESSON 3.2 WORKBOOK How do normal cells become cancer cells?
For a complete list of defined terms, see the Glossary. Transformation the process by which a cell acquires characteristics of a tumor cell. LESSON 3.2 WORKBOOK How do normal cells become cancer cells?
The Chromosomal Basis of Inheritance
 The Chromosomal Basis of Inheritance Factors and Genes Mendel s model of inheritance was based on the idea of factors that were independently assorted and segregated into gametes We now know that these
The Chromosomal Basis of Inheritance Factors and Genes Mendel s model of inheritance was based on the idea of factors that were independently assorted and segregated into gametes We now know that these
Practice Problems 8. a) What do we define as a beneficial or advantageous mutation to the virus? Why?
 Life Sciences 1a Practice Problems 8 1. You have two strains of HIV one is a wild type strain of HIV and the second has acquired a mutation in the gene encoding the protease. This mutation has a dual effect
Life Sciences 1a Practice Problems 8 1. You have two strains of HIV one is a wild type strain of HIV and the second has acquired a mutation in the gene encoding the protease. This mutation has a dual effect
Development of Carcinoma Pathways
 The Construction of Genetic Pathway to Colorectal Cancer Moriah Wright, MD Clinical Fellow in Colorectal Surgery Creighton University School of Medicine Management of Colon and Diseases February 23, 2019
The Construction of Genetic Pathway to Colorectal Cancer Moriah Wright, MD Clinical Fellow in Colorectal Surgery Creighton University School of Medicine Management of Colon and Diseases February 23, 2019
The Cell Cycle CAMPBELL BIOLOGY IN FOCUS SECOND EDITION URRY CAIN WASSERMAN MINORSKY REECE
 CAMPBELL BIOLOGY IN FOCUS URRY CAIN WASSERMAN MINORSKY REECE 9 The Cell Cycle Lecture Presentations by Kathleen Fitzpatrick and Nicole Tunbridge, Simon Fraser University SECOND EDITION Overview: The Key
CAMPBELL BIOLOGY IN FOCUS URRY CAIN WASSERMAN MINORSKY REECE 9 The Cell Cycle Lecture Presentations by Kathleen Fitzpatrick and Nicole Tunbridge, Simon Fraser University SECOND EDITION Overview: The Key
Section 9. Junaid Malek, M.D.
 Section 9 Junaid Malek, M.D. Mutation Objective: Understand how mutations can arise, and how beneficial ones can alter populations Mutation= a randomly produced, heritable change in the nucleotide sequence
Section 9 Junaid Malek, M.D. Mutation Objective: Understand how mutations can arise, and how beneficial ones can alter populations Mutation= a randomly produced, heritable change in the nucleotide sequence
A Mechanism of Gene Amplification Driven by Small DNA Fragments
 A Mechanism of Gene Amplification Driven by Small DNA Fragments Kuntal Mukherjee, Francesca Storici* School of Biology, Georgia Institute of Technology, Atlanta, Georgia, United States of America Abstract
A Mechanism of Gene Amplification Driven by Small DNA Fragments Kuntal Mukherjee, Francesca Storici* School of Biology, Georgia Institute of Technology, Atlanta, Georgia, United States of America Abstract
The Case of the Cumbersome Chromosomes: An Introduction to Workshop Genetics.
 Volume 24: Mini Workshops 253 The Case of the Cumbersome Chromosomes: An Introduction to Workshop Genetics. Tammy Tobin-Janzen Biology Department Susquehanna University Selinsgrove, PA 17870 Tammy is an
Volume 24: Mini Workshops 253 The Case of the Cumbersome Chromosomes: An Introduction to Workshop Genetics. Tammy Tobin-Janzen Biology Department Susquehanna University Selinsgrove, PA 17870 Tammy is an
Studying The Role Of DNA Mismatch Repair In Brain Cancer Malignancy
 Kavya Puchhalapalli CALS Honors Project Report Spring 2017 Studying The Role Of DNA Mismatch Repair In Brain Cancer Malignancy Abstract Malignant brain tumors including medulloblastomas and primitive neuroectodermal
Kavya Puchhalapalli CALS Honors Project Report Spring 2017 Studying The Role Of DNA Mismatch Repair In Brain Cancer Malignancy Abstract Malignant brain tumors including medulloblastomas and primitive neuroectodermal
Supplementary Figure 1
 Supplementary Figure 1 Supplementary Fig. 1: Quality assessment of formalin-fixed paraffin-embedded (FFPE)-derived DNA and nuclei. (a) Multiplex PCR analysis of unrepaired and repaired bulk FFPE gdna from
Supplementary Figure 1 Supplementary Fig. 1: Quality assessment of formalin-fixed paraffin-embedded (FFPE)-derived DNA and nuclei. (a) Multiplex PCR analysis of unrepaired and repaired bulk FFPE gdna from
Mutagenesis. David M. DeMarini, Ph.D. University of North Carolina at Chapel Hill.
 Mutagenesis David M. DeMarini, Ph.D. University of North Carolina at Chapel Hill ddemarini@nc.rr.com 1. DNA as the Target Mutagenesis Overview 2. DNA Damage (The Mutagenesis Paradigm) A. Types of Damage
Mutagenesis David M. DeMarini, Ph.D. University of North Carolina at Chapel Hill ddemarini@nc.rr.com 1. DNA as the Target Mutagenesis Overview 2. DNA Damage (The Mutagenesis Paradigm) A. Types of Damage
The preference for error-free or error-prone postreplication repair is cell-cycle- dependent in Saccharomyces cerevisiae in response to low-dose
 MCB Accepts, published online ahead of print on 4 February 2013 Mol. Cell. Biol. doi:10.1128/mcb.01392-12 Copyright 2013, American Society for Microbiology. All Rights Reserved. 1 2 3 The preference for
MCB Accepts, published online ahead of print on 4 February 2013 Mol. Cell. Biol. doi:10.1128/mcb.01392-12 Copyright 2013, American Society for Microbiology. All Rights Reserved. 1 2 3 The preference for
5/25/2015. Replication fork. Replication fork. Replication fork. Replication fork
 Mutations Chapter 5 Cellular Functions Lecture 3: and Cell Division Most DNA mutations alter the protein product May Make it function better (rarely) Change its function Reduce its function Make it non-functional
Mutations Chapter 5 Cellular Functions Lecture 3: and Cell Division Most DNA mutations alter the protein product May Make it function better (rarely) Change its function Reduce its function Make it non-functional
Targeting the ATR Kinase in Cancer Therapy
 Targeting the ATR Kinase in Cancer Therapy 2017 Chabner Colloquium October 30, 2017 Lee Zou MGH Cancer Center Harvard Medical School Disclosure Consultant/advisory role: Loxo Oncology DNA Damage and Replication
Targeting the ATR Kinase in Cancer Therapy 2017 Chabner Colloquium October 30, 2017 Lee Zou MGH Cancer Center Harvard Medical School Disclosure Consultant/advisory role: Loxo Oncology DNA Damage and Replication
Integration of Cancer Genome into GECCO- Genetics and Epidemiology of Colorectal Cancer Consortium
 Integration of Cancer Genome into GECCO- Genetics and Epidemiology of Colorectal Cancer Consortium Ulrike Peters Fred Hutchinson Cancer Research Center University of Washington U01-CA137088-05, PI: Peters
Integration of Cancer Genome into GECCO- Genetics and Epidemiology of Colorectal Cancer Consortium Ulrike Peters Fred Hutchinson Cancer Research Center University of Washington U01-CA137088-05, PI: Peters
of TERT, MLL4, CCNE1, SENP5, and ROCK1 on tumor development were discussed.
 Supplementary Note The potential association and implications of HBV integration at known and putative cancer genes of TERT, MLL4, CCNE1, SENP5, and ROCK1 on tumor development were discussed. Human telomerase
Supplementary Note The potential association and implications of HBV integration at known and putative cancer genes of TERT, MLL4, CCNE1, SENP5, and ROCK1 on tumor development were discussed. Human telomerase
LECTURE PRESENTATIONS
 LECTURE PRESENTATIONS For CAMPBELL BIOLOGY, NINTH EDITION Jane B. Reece, Lisa A. Urry, Michael L. Cain, Steven A. Wasserman, Peter V. Minorsky, Robert B. Jackson Chapter 12 The Cell Cycle Lectures by Erin
LECTURE PRESENTATIONS For CAMPBELL BIOLOGY, NINTH EDITION Jane B. Reece, Lisa A. Urry, Michael L. Cain, Steven A. Wasserman, Peter V. Minorsky, Robert B. Jackson Chapter 12 The Cell Cycle Lectures by Erin
Why do cells reproduce?
 Outline Cell Reproduction 1. Overview of Cell Reproduction 2. Cell Reproduction in Prokaryotes 3. Cell Reproduction in Eukaryotes 1. Chromosomes 2. Cell Cycle 3. Mitosis and Cytokinesis Examples of Cell
Outline Cell Reproduction 1. Overview of Cell Reproduction 2. Cell Reproduction in Prokaryotes 3. Cell Reproduction in Eukaryotes 1. Chromosomes 2. Cell Cycle 3. Mitosis and Cytokinesis Examples of Cell
Brian T Burgess, DO, PhD, GYN Oncology Fellow Rachel W. Miller, MD, GYN Oncology
 Brian T Burgess, DO, PhD, GYN Oncology Fellow Rachel W. Miller, MD, GYN Oncology Epithelial Ovarian Cancer - Standard Current Treatment: Surgery with De-bulking + Platinum-Taxane based Chemotherapy - No
Brian T Burgess, DO, PhD, GYN Oncology Fellow Rachel W. Miller, MD, GYN Oncology Epithelial Ovarian Cancer - Standard Current Treatment: Surgery with De-bulking + Platinum-Taxane based Chemotherapy - No
LECTURE PRESENTATIONS
 LECTURE PRESENTATIONS For CAMPBELL BIOLOGY, NINTH EDITION Jane B. Reece, Lisa A. Urry, Michael L. Cain, Steven A. Wasserman, Peter V. Minorsky, Robert B. Jackson Chapter 12 The Cell Cycle Lectures by Erin
LECTURE PRESENTATIONS For CAMPBELL BIOLOGY, NINTH EDITION Jane B. Reece, Lisa A. Urry, Michael L. Cain, Steven A. Wasserman, Peter V. Minorsky, Robert B. Jackson Chapter 12 The Cell Cycle Lectures by Erin
Unit 5: Genetics Notes
 Unit 5: Genetics Notes https://goo.gl/fgtzef Name: Period: Test Date: Table of Contents Title of Page Page Number Date Warm-ups 3-4 Mendelian Genetics Notes 5-6 Mendelian Genetics Lets Practice 7 Monohybrid
Unit 5: Genetics Notes https://goo.gl/fgtzef Name: Period: Test Date: Table of Contents Title of Page Page Number Date Warm-ups 3-4 Mendelian Genetics Notes 5-6 Mendelian Genetics Lets Practice 7 Monohybrid
MCB140: Second Midterm Spring 2010
 MCB140: Second Midterm Spring 2010 Before you start, print your name and student identification number (S.I.D) at the top of each page. There are 11 pages including this page. You will have 150 minutes
MCB140: Second Midterm Spring 2010 Before you start, print your name and student identification number (S.I.D) at the top of each page. There are 11 pages including this page. You will have 150 minutes
LTA Analysis of HapMap Genotype Data
 LTA Analysis of HapMap Genotype Data Introduction. This supplement to Global variation in copy number in the human genome, by Redon et al., describes the details of the LTA analysis used to screen HapMap
LTA Analysis of HapMap Genotype Data Introduction. This supplement to Global variation in copy number in the human genome, by Redon et al., describes the details of the LTA analysis used to screen HapMap
Genetics 275 Examination February 10, 2003.
 Genetics 275 Examination February 10, 2003. Do all questions in the spaces provided. The value for this examination is twenty marks (20% of the grade for the course). The value for individual questions
Genetics 275 Examination February 10, 2003. Do all questions in the spaces provided. The value for this examination is twenty marks (20% of the grade for the course). The value for individual questions
The Cell Cycle. Chapter 12. PowerPoint Lecture Presentations for Biology Eighth Edition Neil Campbell and Jane Reece
 Chapter 12 The Cell Cycle PowerPoint Lecture Presentations for Biology Eighth Edition Neil Campbell and Jane Reece Lectures by Chris Romero, updated by Erin Barley with contributions from Joan Sharp Overview:
Chapter 12 The Cell Cycle PowerPoint Lecture Presentations for Biology Eighth Edition Neil Campbell and Jane Reece Lectures by Chris Romero, updated by Erin Barley with contributions from Joan Sharp Overview:
Supplementary Information
 Supplementary Information HBV maintains electrostatic homeostasis by modulating negative charges from phosphoserine and encapsidated nucleic acids Authors: Pei-Yi Su 1,2,3, Ching-Jen Yang 2, Tien-Hua Chu
Supplementary Information HBV maintains electrostatic homeostasis by modulating negative charges from phosphoserine and encapsidated nucleic acids Authors: Pei-Yi Su 1,2,3, Ching-Jen Yang 2, Tien-Hua Chu
Section D: The Molecular Biology of Cancer
 CHAPTER 19 THE ORGANIZATION AND CONTROL OF EUKARYOTIC GENOMES Section D: The Molecular Biology of Cancer 1. Cancer results from genetic changes that affect the cell cycle 2. Oncogene proteins and faulty
CHAPTER 19 THE ORGANIZATION AND CONTROL OF EUKARYOTIC GENOMES Section D: The Molecular Biology of Cancer 1. Cancer results from genetic changes that affect the cell cycle 2. Oncogene proteins and faulty
Ch. 18 Regulation of Gene Expression
 Ch. 18 Regulation of Gene Expression 1 Human genome has around 23,688 genes (Scientific American 2/2006) Essential Questions: How is transcription regulated? How are genes expressed? 2 Bacteria regulate
Ch. 18 Regulation of Gene Expression 1 Human genome has around 23,688 genes (Scientific American 2/2006) Essential Questions: How is transcription regulated? How are genes expressed? 2 Bacteria regulate
Biology 105: Introduction to Genetics Midterm EXAM. Part1. Definitions. 1 Recessive allele. Name. Student ID. 2 Homologous chromosomes
 Biology 105: Introduction to Genetics Midterm EXAM Part1 Definitions 1 Recessive allele Name Student ID 2 Homologous chromosomes Before starting, write your name on the top of each page Make sure you have
Biology 105: Introduction to Genetics Midterm EXAM Part1 Definitions 1 Recessive allele Name Student ID 2 Homologous chromosomes Before starting, write your name on the top of each page Make sure you have
Supplementary Materials for
 www.sciencetranslationalmedicine.org/cgi/content/full/7/283/283ra54/dc1 Supplementary Materials for Clonal status of actionable driver events and the timing of mutational processes in cancer evolution
www.sciencetranslationalmedicine.org/cgi/content/full/7/283/283ra54/dc1 Supplementary Materials for Clonal status of actionable driver events and the timing of mutational processes in cancer evolution
Chapter 8 DNA Replication, Binary Fission, and Mitosis
 Chapter 8 DNA Replication, Binary Fission, and Mitosis World s tallest man Frederic J. Brown/AFP/Getty Images Copyright McGraw-Hill Education. All rights reserved. No reproduction or distribution without
Chapter 8 DNA Replication, Binary Fission, and Mitosis World s tallest man Frederic J. Brown/AFP/Getty Images Copyright McGraw-Hill Education. All rights reserved. No reproduction or distribution without
Genetic Variation Junior Science
 2018 Version Genetic Variation Junior Science http://img.publishthis.com/images/bookmarkimages/2015/05/d/5/c/d5cf017fb4f7e46e1c21b874472ea7d1_bookmarkimage_620x480_xlarge_original_1.jpg Sexual Reproduction
2018 Version Genetic Variation Junior Science http://img.publishthis.com/images/bookmarkimages/2015/05/d/5/c/d5cf017fb4f7e46e1c21b874472ea7d1_bookmarkimage_620x480_xlarge_original_1.jpg Sexual Reproduction
Chromosome Structure & Recombination
 Chromosome Structure & Recombination (CHAPTER 8- Brooker Text) April 4 & 9, 2007 BIO 184 Dr. Tom Peavy Genetic variation refers to differences between members of the same species or those of different
Chromosome Structure & Recombination (CHAPTER 8- Brooker Text) April 4 & 9, 2007 BIO 184 Dr. Tom Peavy Genetic variation refers to differences between members of the same species or those of different
Unit 9: The Cell Cycle
 Unit 9: The Cell Cycle Name: Period: Test Date: 1 Table of Contents Title of Page Page Number Teacher Stamp Unit 9 Warm-Ups 3-4 Cell Cycle/Interphase Notes 5 DNA Replication Video 6 Cancer Notes 15-16
Unit 9: The Cell Cycle Name: Period: Test Date: 1 Table of Contents Title of Page Page Number Teacher Stamp Unit 9 Warm-Ups 3-4 Cell Cycle/Interphase Notes 5 DNA Replication Video 6 Cancer Notes 15-16
Computational Systems Biology: Biology X
 Bud Mishra Room 1002, 715 Broadway, Courant Institute, NYU, New York, USA L#4:(October-0-4-2010) Cancer and Signals 1 2 1 2 Evidence in Favor Somatic mutations, Aneuploidy, Copy-number changes and LOH
Bud Mishra Room 1002, 715 Broadway, Courant Institute, NYU, New York, USA L#4:(October-0-4-2010) Cancer and Signals 1 2 1 2 Evidence in Favor Somatic mutations, Aneuploidy, Copy-number changes and LOH
Cytogenetics 101: Clinical Research and Molecular Genetic Technologies
 Cytogenetics 101: Clinical Research and Molecular Genetic Technologies Topics for Today s Presentation 1 Classical vs Molecular Cytogenetics 2 What acgh? 3 What is FISH? 4 What is NGS? 5 How can these
Cytogenetics 101: Clinical Research and Molecular Genetic Technologies Topics for Today s Presentation 1 Classical vs Molecular Cytogenetics 2 What acgh? 3 What is FISH? 4 What is NGS? 5 How can these
Chapter 6. Cell Reproduction
 Chapter 6 Cell Reproduction Chromosomes The growth and development of your body are controlled by instructions contained in your DNA DNA decides what you will be.. DNA & Chromosomes Very long molecules
Chapter 6 Cell Reproduction Chromosomes The growth and development of your body are controlled by instructions contained in your DNA DNA decides what you will be.. DNA & Chromosomes Very long molecules
Ex vivo functional assays for Homologous Recombination deficiency in breast cancer. Dik C. van Gent
 Ex vivo functional assays for Homologous Recombination deficiency in breast cancer Dik C. van Gent Breast cancer types treatments ER/PR: anti-hormonal therapy HER2: Herceptin Triple negative (TNBC): no
Ex vivo functional assays for Homologous Recombination deficiency in breast cancer Dik C. van Gent Breast cancer types treatments ER/PR: anti-hormonal therapy HER2: Herceptin Triple negative (TNBC): no
A n a ly s i s. DNA replication timing 4 and long-range DNA interactions 6. predict mutational landscapes of cancer genomes
 DNA replication timing and long-range DNA interactions predict mutational landscapes of cancer genomes Subhajyoti De 1,2 & Franziska Michor 1,2 211 Nature America, Inc. All rights reserved. Somatic copy-number
DNA replication timing and long-range DNA interactions predict mutational landscapes of cancer genomes Subhajyoti De 1,2 & Franziska Michor 1,2 211 Nature America, Inc. All rights reserved. Somatic copy-number
Hands-On Ten The BRCA1 Gene and Protein
 Hands-On Ten The BRCA1 Gene and Protein Objective: To review transcription, translation, reading frames, mutations, and reading files from GenBank, and to review some of the bioinformatics tools, such
Hands-On Ten The BRCA1 Gene and Protein Objective: To review transcription, translation, reading frames, mutations, and reading files from GenBank, and to review some of the bioinformatics tools, such
Chapt 15: Molecular Genetics of Cell Cycle and Cancer
 Chapt 15: Molecular Genetics of Cell Cycle and Cancer Student Learning Outcomes: Describe the cell cycle: steps taken by a cell to duplicate itself = cell division; Interphase (G1, S and G2), Mitosis.
Chapt 15: Molecular Genetics of Cell Cycle and Cancer Student Learning Outcomes: Describe the cell cycle: steps taken by a cell to duplicate itself = cell division; Interphase (G1, S and G2), Mitosis.
Breaking Up is Hard to Do (At Least in Eukaryotes) Mitosis
 Breaking Up is Hard to Do (At Least in Eukaryotes) Mitosis Prokaryotes Have a Simpler Cell Cycle Cell division in prokaryotes takes place in two stages, which together make up a simple cell cycle 1. Copy
Breaking Up is Hard to Do (At Least in Eukaryotes) Mitosis Prokaryotes Have a Simpler Cell Cycle Cell division in prokaryotes takes place in two stages, which together make up a simple cell cycle 1. Copy
Biochemistry 201: DNA repair January 24, 26, 2000 Gilbert Chu
 1) Why study DNA repair? Biochemistry 201: DNA repair January 24, 26, 2000 Gilbert Chu The genome is assaulted by a myriad of different agents that cause DNA damage. Sources of damage can be both exogenous
1) Why study DNA repair? Biochemistry 201: DNA repair January 24, 26, 2000 Gilbert Chu The genome is assaulted by a myriad of different agents that cause DNA damage. Sources of damage can be both exogenous
CNV Detection and Interpretation in Genomic Data
 CNV Detection and Interpretation in Genomic Data Benjamin W. Darbro, M.D., Ph.D. Assistant Professor of Pediatrics Director of the Shivanand R. Patil Cytogenetics and Molecular Laboratory Overview What
CNV Detection and Interpretation in Genomic Data Benjamin W. Darbro, M.D., Ph.D. Assistant Professor of Pediatrics Director of the Shivanand R. Patil Cytogenetics and Molecular Laboratory Overview What
Overview: Conducting the Genetic Orchestra Prokaryotes and eukaryotes alter gene expression in response to their changing environment
 Overview: Conducting the Genetic Orchestra Prokaryotes and eukaryotes alter gene expression in response to their changing environment In multicellular eukaryotes, gene expression regulates development
Overview: Conducting the Genetic Orchestra Prokaryotes and eukaryotes alter gene expression in response to their changing environment In multicellular eukaryotes, gene expression regulates development
The Cell Cycle. Chapter 12. Biology Eighth Edition Neil Campbell and Jane Reece. PowerPoint Lecture Presentations for
 Chapter 12 The Cell Cycle PowerPoint Lecture Presentations for Biology Eighth Edition Neil Campbell and Jane Reece Lectures by Chris Romero, updated by Erin Barley with contributions from Joan Sharp 1
Chapter 12 The Cell Cycle PowerPoint Lecture Presentations for Biology Eighth Edition Neil Campbell and Jane Reece Lectures by Chris Romero, updated by Erin Barley with contributions from Joan Sharp 1
Name: PS#: Biol 3301 Midterm 1 Spring 2012
 Name: PS#: Biol 3301 Midterm 1 Spring 2012 Multiple Choice. Circle the single best answer. (4 pts each) 1. Which of the following changes in the DNA sequence of a gene will produce a new allele? a) base
Name: PS#: Biol 3301 Midterm 1 Spring 2012 Multiple Choice. Circle the single best answer. (4 pts each) 1. Which of the following changes in the DNA sequence of a gene will produce a new allele? a) base
Human Genetics 542 Winter 2017 Syllabus
 Human Genetics 542 Winter 2017 Syllabus Monday, Wednesday, and Friday 9 10 a.m. 5915 Buhl Course Director: Tony Antonellis Module I: Mapping and characterizing simple genetic diseases Jan 4 th Wed Mapping
Human Genetics 542 Winter 2017 Syllabus Monday, Wednesday, and Friday 9 10 a.m. 5915 Buhl Course Director: Tony Antonellis Module I: Mapping and characterizing simple genetic diseases Jan 4 th Wed Mapping
Generating Spontaneous Copy Number Variants (CNVs) Jennifer Freeman Assistant Professor of Toxicology School of Health Sciences Purdue University
 Role of Chemical lexposure in Generating Spontaneous Copy Number Variants (CNVs) Jennifer Freeman Assistant Professor of Toxicology School of Health Sciences Purdue University CNV Discovery Reference Genetic
Role of Chemical lexposure in Generating Spontaneous Copy Number Variants (CNVs) Jennifer Freeman Assistant Professor of Toxicology School of Health Sciences Purdue University CNV Discovery Reference Genetic
2.1 The Importance of Cell Division
 2.1 The Importance of Cell Division Functions of cell division Growth Repair Reproduction Growth All organisms begin as a single cell. Cell divisions will increase as an organism s size increases. There
2.1 The Importance of Cell Division Functions of cell division Growth Repair Reproduction Growth All organisms begin as a single cell. Cell divisions will increase as an organism s size increases. There
Part I: The Cell Cycle
 Cellular Differentiation Part I: The Cell Cycle During your lifetime, trillions of your cells will undergo the cell cycle. This process allows you to grow, heal, and maintain your vital tissues and organs.
Cellular Differentiation Part I: The Cell Cycle During your lifetime, trillions of your cells will undergo the cell cycle. This process allows you to grow, heal, and maintain your vital tissues and organs.
Introduction. Cancer Biology. Tumor-suppressor genes. Proto-oncogenes. DNA stability genes. Mechanisms of carcinogenesis.
 Cancer Biology Chapter 18 Eric J. Hall., Amato Giaccia, Radiobiology for the Radiologist Introduction Tissue homeostasis depends on the regulated cell division and self-elimination (programmed cell death)
Cancer Biology Chapter 18 Eric J. Hall., Amato Giaccia, Radiobiology for the Radiologist Introduction Tissue homeostasis depends on the regulated cell division and self-elimination (programmed cell death)
LECTURE PRESENTATIONS
 LECTURE PRESENTATIONS For CAMPBELL BIOLOGY, NINTH EDITION Jane B. Reece, Lisa A. Urry, Michael L. Cain, Steven A. Wasserman, Peter V. Minorsky, Robert B. Jackson Chapter 12 The Cell Cycle Lectures by Erin
LECTURE PRESENTATIONS For CAMPBELL BIOLOGY, NINTH EDITION Jane B. Reece, Lisa A. Urry, Michael L. Cain, Steven A. Wasserman, Peter V. Minorsky, Robert B. Jackson Chapter 12 The Cell Cycle Lectures by Erin
Example: Distance in M.U. % Crossing Over Why? Double crossovers
 Example: Distance in M.U. % Crossing Over 1 5 10 15 50 80 100 107 Why? Double crossovers 232 .. A B. a b. 1. A fully heterozygous gray-bodied (b+), normal winged (vg+) female F 1 fruit fly crossed with
Example: Distance in M.U. % Crossing Over 1 5 10 15 50 80 100 107 Why? Double crossovers 232 .. A B. a b. 1. A fully heterozygous gray-bodied (b+), normal winged (vg+) female F 1 fruit fly crossed with
DNA/RNA: polynucleotide chains
 What is DNA? DNA/RNA: polynucleotide chains Phosphate Base Sugar (2 OH=ribose, 2 H=deoxyribose) Nucleotide =sugar+phosphate+base DNA is a double helix DNA damage and repair How is DNA damaged? How is DNA
What is DNA? DNA/RNA: polynucleotide chains Phosphate Base Sugar (2 OH=ribose, 2 H=deoxyribose) Nucleotide =sugar+phosphate+base DNA is a double helix DNA damage and repair How is DNA damaged? How is DNA
Genomic analysis of essentiality within protein networks
 Genomic analysis of essentiality within protein networks Haiyuan Yu, Dov Greenbaum, Hao Xin Lu, Xiaowei Zhu and Mark Gerstein Department of Molecular Biophysics and Biochemistry, 266 Whitney Avenue, Yale
Genomic analysis of essentiality within protein networks Haiyuan Yu, Dov Greenbaum, Hao Xin Lu, Xiaowei Zhu and Mark Gerstein Department of Molecular Biophysics and Biochemistry, 266 Whitney Avenue, Yale
Chapter 8: Cellular Reproduction
 Chapter 8: Cellular Reproduction 1. The Cell Cycle 2. Mitosis 3. Meiosis 2 Types of Cell Division 2n 1n Mitosis: occurs in somatic cells (almost all cells of the body) generates cells identical to original
Chapter 8: Cellular Reproduction 1. The Cell Cycle 2. Mitosis 3. Meiosis 2 Types of Cell Division 2n 1n Mitosis: occurs in somatic cells (almost all cells of the body) generates cells identical to original
Unit 5 Part B Cell Growth, Division and Reproduction
 Unit 5 Part B Cell Growth, Division and Reproduction Cell Size Are whale cells the same size as sea stars cells? Yes! Cell Size Limitations Cells that are too big will have difficulty diffusing materials
Unit 5 Part B Cell Growth, Division and Reproduction Cell Size Are whale cells the same size as sea stars cells? Yes! Cell Size Limitations Cells that are too big will have difficulty diffusing materials
AS90163 Biology Describe the transfer of genetic information Part 1 - DNA structure & Cell division
 AS90163 Biology Describe the transfer of genetic information Part 1 - DNA structure & Cell division This achievement standard involves the description of the transfer of genetic information. Achievement
AS90163 Biology Describe the transfer of genetic information Part 1 - DNA structure & Cell division This achievement standard involves the description of the transfer of genetic information. Achievement
Deep-Sequencing of HIV-1
 Deep-Sequencing of HIV-1 The quest for true variants Alexander Thielen, Martin Däumer 09.05.2015 Limitations of drug resistance testing by standard-sequencing Blood plasma RNA extraction RNA Reverse Transcription/
Deep-Sequencing of HIV-1 The quest for true variants Alexander Thielen, Martin Däumer 09.05.2015 Limitations of drug resistance testing by standard-sequencing Blood plasma RNA extraction RNA Reverse Transcription/
Chapter 10 Chromosomes and Cell Reproduction
 Chapter 10 Chromosomes and Cell Reproduction Chromosomes Organisms grow by dividing of cells Binary Fission form of asexual reproduction that produces identical offspring (Bacteria) Eukaryotes have two
Chapter 10 Chromosomes and Cell Reproduction Chromosomes Organisms grow by dividing of cells Binary Fission form of asexual reproduction that produces identical offspring (Bacteria) Eukaryotes have two
Introduction retroposon
 17.1 - Introduction A retrovirus is an RNA virus able to convert its sequence into DNA by reverse transcription A retroposon (retrotransposon) is a transposon that mobilizes via an RNA form; the DNA element
17.1 - Introduction A retrovirus is an RNA virus able to convert its sequence into DNA by reverse transcription A retroposon (retrotransposon) is a transposon that mobilizes via an RNA form; the DNA element
Chromosome Abnormalities
 Chromosome Abnormalities Chromosomal abnormalities vs. molecular mutations Simply a matter of size Chromosomal abnormalities are big errors Two types of abnormalities 1. Constitutional problem present
Chromosome Abnormalities Chromosomal abnormalities vs. molecular mutations Simply a matter of size Chromosomal abnormalities are big errors Two types of abnormalities 1. Constitutional problem present
Human Genetics 542 Winter 2018 Syllabus
 Human Genetics 542 Winter 2018 Syllabus Monday, Wednesday, and Friday 9 10 a.m. 5915 Buhl Course Director: Tony Antonellis Jan 3 rd Wed Mapping disease genes I: inheritance patterns and linkage analysis
Human Genetics 542 Winter 2018 Syllabus Monday, Wednesday, and Friday 9 10 a.m. 5915 Buhl Course Director: Tony Antonellis Jan 3 rd Wed Mapping disease genes I: inheritance patterns and linkage analysis
Cancer Genomics. Nic Waddell. Winter School in Mathematical and Computational Biology. July th
 Cancer Genomics Nic Waddell Winter School in Mathematical and Computational Biology 6th July 2015 Time Line of Key Events in Cancer Genomics Michael R. Stratton Science 2011;331:1553-1558 The Cancer Genome
Cancer Genomics Nic Waddell Winter School in Mathematical and Computational Biology 6th July 2015 Time Line of Key Events in Cancer Genomics Michael R. Stratton Science 2011;331:1553-1558 The Cancer Genome
CELL CYCLE REGULATION AND CANCER. Cellular Reproduction II
 CELL CYCLE REGULATION AND CANCER Cellular Reproduction II THE CELL CYCLE Interphase G1- gap phase 1- cell grows and develops S- DNA synthesis phase- cell replicates each chromosome G2- gap phase 2- cell
CELL CYCLE REGULATION AND CANCER Cellular Reproduction II THE CELL CYCLE Interphase G1- gap phase 1- cell grows and develops S- DNA synthesis phase- cell replicates each chromosome G2- gap phase 2- cell
CRISPR/Cas9 Enrichment and Long-read WGS for Structural Variant Discovery
 CRISPR/Cas9 Enrichment and Long-read WGS for Structural Variant Discovery PacBio CoLab Session October 20, 2017 For Research Use Only. Not for use in diagnostics procedures. Copyright 2017 by Pacific Biosciences
CRISPR/Cas9 Enrichment and Long-read WGS for Structural Variant Discovery PacBio CoLab Session October 20, 2017 For Research Use Only. Not for use in diagnostics procedures. Copyright 2017 by Pacific Biosciences
Breaking Up is Hard to Do (At Least in Eukaryotes) Mitosis
 Breaking Up is Hard to Do (At Least in Eukaryotes) Mitosis Chromosomes Chromosomes were first observed by the German embryologist Walther Fleming in 1882. Chromosome number varies among organisms most
Breaking Up is Hard to Do (At Least in Eukaryotes) Mitosis Chromosomes Chromosomes were first observed by the German embryologist Walther Fleming in 1882. Chromosome number varies among organisms most
The Need for a PARP in vivo Pharmacodynamic Assay
 The Need for a PARP in vivo Pharmacodynamic Assay Jay George, Ph.D. Chief Scientific Officer Trevigen, Inc. Gaithersburg, MD Poly(ADP-ribose) polymerases are promising therapeutic targets. In response
The Need for a PARP in vivo Pharmacodynamic Assay Jay George, Ph.D. Chief Scientific Officer Trevigen, Inc. Gaithersburg, MD Poly(ADP-ribose) polymerases are promising therapeutic targets. In response
BIOLOGY. Cell Cycle - Mitosis. Outline. Overview: The Key Roles of Cell Division. identical daughter cells. I. Overview II.
 2 Cell Cycle - Mitosis CAMPBELL BIOLOGY TENTH EDITION Reece Urry Cain Wasserman Minorsky Jackson Outline I. Overview II. Mitotic Phase I. Prophase II. III. Telophase IV. Cytokinesis III. Binary fission
2 Cell Cycle - Mitosis CAMPBELL BIOLOGY TENTH EDITION Reece Urry Cain Wasserman Minorsky Jackson Outline I. Overview II. Mitotic Phase I. Prophase II. III. Telophase IV. Cytokinesis III. Binary fission
Campbell Biology in Focus (Urry) Chapter 9 The Cell Cycle. 9.1 Multiple-Choice Questions
 Campbell Biology in Focus (Urry) Chapter 9 The Cell Cycle 9.1 Multiple-Choice Questions 1) Starting with a fertilized egg (zygote), a series of five cell divisions would produce an early embryo with how
Campbell Biology in Focus (Urry) Chapter 9 The Cell Cycle 9.1 Multiple-Choice Questions 1) Starting with a fertilized egg (zygote), a series of five cell divisions would produce an early embryo with how
Introduction to LOH and Allele Specific Copy Number User Forum
 Introduction to LOH and Allele Specific Copy Number User Forum Jonathan Gerstenhaber Introduction to LOH and ASCN User Forum Contents 1. Loss of heterozygosity Analysis procedure Types of baselines 2.
Introduction to LOH and Allele Specific Copy Number User Forum Jonathan Gerstenhaber Introduction to LOH and ASCN User Forum Contents 1. Loss of heterozygosity Analysis procedure Types of baselines 2.
