Evaluation of immunomodulatory agents in syngeneic models and immune cell phenotyping of humanized mouse models carrying patient-derived tumors
|
|
|
- Randolph Lamb
- 6 years ago
- Views:
Transcription
1 Evaluation of immunomodulatory agents in syngeneic models and immune cell phenotyping of humanized mouse models carrying patient-derived tumors Philippe Slos 2 nd Annual ICI March 15-17, 2016 Boston
2 Introduction Syngeneic models at Oncodesign Case studies Humanized mouse models PDX panels Conclusions & Perspectives page 2
3 Introduction page 3
4 The needs: to improve predictivity of the models Adapted From Wartha et al. Pharmacology & Therapeutics, 2014 Suitable models for immuno-oncology? Two key components Immune System Tumor Immune system and tumor could be from mouse : standard syngeneic models, GEMM from human : standard xenografts or PDXs on humanized mouse Fit-for purpose use of mouse models to improve predictivity of cancer therapeutics evaluation page 4
5 Quite all is immunomodulation From Galluzzi et al. Oncotarget, 2014, Vol. 5, No. 24, page 5
6 SYNGENEIC MODELS AT ONCODESIGN page 6
7 Syngeneic Models at Oncodesign (1) Good characterization Characterization not complete Limited characterization Model Organ Host rodent species Injection site Tested drugs Metastatic Dissemination 4T1 Breast carcinoma BALB/C mouse OT Radiotherapy, Doxorubicin, PD-1 mab, CTLA- 4 mab A20 B cell lymphoma BALB/C mouse SC IV CTLA-4 mab, PD-1 mab AB12 Mesothelioma BALB/C mouse SC AY27 Bladder transitional carcinoma B16-F10 Malignant melanoma C57BL/6 mouse SC IV IC Ficher rat OT BCG, CDDP Paclitaxel, Doxorubicin, TMZ, PD-1 mab, Cyclophosphamide, CTLA-4 mab, PD-L1 mab C1498 AML C57BL/6 mouse SC IV C26 Colon adenocarcinoma BALB/C mouse IP SC CDDP C38 Colon carcinoma C57BL/6 mouse SC Radiotherapy, CPT-11, L-OHP, CTLA-4 C-51 Colon carcinoma BALB/C mouse SC CPT-11, LOHP C6 Glioma Wistar rat OT SC BCNU CT26 Colon carcinoma BALB/C mouse OT SC *DHD/K12/TRb = PROb Colon adenocarcinoma BDIX rat IP SC *DHD/K12/TSb = REGb Colon adenocarcinoma BDIX rat SC EMT6 Breast carcinoma BALB/C mouse OT SC 5-FU, CPT-11, PD-1 mab, CTLA-4 mab, PD-L1 mab CDDP, Doxorubicin, BCG, 5-FU, L-OHP, Mitoxantrone Doxorubicin, Paclitaxel, Radiotherapy, PD-1 mab, CTLA-4 mab GS-9L Gliosarcoma Ficher rat OT SC BCNU, TMZ page 7 * Ask for rat availability
8 Syngeneic Models at Oncodesign (2) Good characterization Characterization not complete Limited characterization Model Organ Host rodent species Injection site Tested drugs Metastatic Dissemination *GV1A1 Glioma BDIX rat OT SC Hepa1-6 Hepatocarcinoma C57BL/6 mouse OT Sorafenib, CTLA-4 mab L1210 Lymphocytic leukemia DBA/2 mouse IP BCNU, CDDP, Doxorubicin, 5-FU L-OHP, Mitoxantrone L1210/CDDP Lymphocytic leukemia DBA/2 mouse IP CDDP, L-OHP LLC1 Lewis lung carcinoma C57BL/6 mouse IV SC Vinorelbine, Cyclophosphamide CTLA-4 mab, PD-1 mab MAT-LyLu Prostate adenocarcinoma Cop rat SC MBT-2 Bladder carcinoma C3H mouse OT SC BCG, Mitomycin C, PD-1 MPC-11 Plasmacytoma BALB/C mouse SC NBT-II Bladder carcinoma Wistar rat OT P388 Leukemia DBA/2 mouse IP P388/ADR Leukemia DBA/2 mouse IP BCNU, Doxorubicin, Mitoxantrone BCNU, Doxorubicin, Vinblastine R3327-AT3 Prostate adenocarcinoma Cop rat SC R3327H Prostate adenocarcinoma Cop rat OT SC RenCa Kidney carcinoma BALB/C mouse OT SC Paclitaxel, Mitoxantrone, Trastuzumab, Leuroprolin, Castration, Decapeptyl, Bicalutamide, Radiotherapy CDDP, Paclitaxel, Radiotherapy, Gemcitabine, Carboplatin, Sorafenib page 8 * Ask for rat availability
9 CASE STUDIES page 9
10 Tumor volume (mm 3 ) Tumor volume (mm 3 ) Tumor volume (mm 3 ) 4T1 Breast Carcinoma Orthotopic Model Immune checkpoint inhibitors Mice were OT injected with 4T1 murine breast tumor cells at D0. Mice were randomized based on tumor volume at D13 and treated IP with mab against CTLA-4 (clone 9H10) or PD-1 (clone RMP1-14) at 10 mg/kg/inj (Q5Dx3). T/C % (D21) Control 100 CTLA-4 mab 66 PD-1 mab 68 2,000 1,800 1,600 1,400 1,200 1, ,000 1,800 1,600 1,400 1,200 1, ,000 1,800 1,600 1,400 1,200 1, Control Time (Days) CTLA-4 mab Time (Days) PD-1 mab Time (Days) page 10
11 Tumor volume (mm 3 ) Tumor volume (mm 3 ) Tumor volume (mm 3 ) 4T1 Breast Carcinoma Orthotopic Model Correlation of anti-tumoral activity of ICI against tumor and metastasic devolopment Mice were OT injected with 4T1 murine breast tumor cells at D0. Mice were randomized based on tumor volume at D13 and treated IP with mab against CTLA-4 (clone 9H10, Syrian Hamster IgG1) or PD-1 (clone RMP1-14, Rat IgG2a) at 10 mg/kg/inj (Q5Dx3). 2,000 1,800 1,600 1,400 1,200 1, ,000 1,800 1,600 1,400 1,200 1, ,000 1,800 1,600 1,400 1,200 1, Control Time (Days) CTLA-4 mab Time (Days) PD-1 mab Time (Days) page 11
12 Tumor volume (mm 3 ) Tumor volume (mm 3 ) Tumor volume (mm 3 ) Tumor volume (mm 3 ) EMT6 Breast Carcinoma Ectotopic Model Immune checkpoint inhibitors 1,800 1,600 1,400 1,200 1, Control CTLA4 mab PD1 mab Time (Days) Mice were SC injected with EMT6 murine breast tumor cells at D0. Mice were randomized based on tumor volume at D7 and treated IP with mab against CTLA-4 (clone 9H10) or PD-1 (clone RMP1-14) at 10 mg/kg/inj (Q5Dx3). T/C % (D27) Control 100 CTLA-4 mab 4 PD-1 mab 63 2,000 1,800 1,600 1,400 1,200 1, ,000 1,800 1,600 1,400 1,200 1, ,000 1,800 1,600 1,400 1,200 1, Control Time (Days) CTLA-4 mab Time (Days) PD-1 mab Time (Days) page 12
13 EMT6 Breast Carcinoma Ectotopic Model Immune checkpoint inhibitors : Flow Cytometry analysis of FoxP3+ CD4+ T cells TDLN Tumor Control ID#8923 TV 597 mm 3 CTLA-4 mab ID#8901 TV 56 mm 3 Median value Median value without non-responding tumors ( ) CTLA4 mab Non-responding tumors page 13
14 Syngeneic models with immune checkpoint inhibitors : Teff/Treg ratio increase correlated with anti-tumor activity 4T1 Control CT26 Control EMT6 Control CTLA-4 mab CTLA-4 mab CTLA-4 mab Tumor page 14
15 CT26 Colon Carcinoma Ectotopic Model: Teff/Treg ratio increase correlated with anti-tumor activity Combined Immune checkpoint inhibitors Tumor Mice were SC injected with CT26 murine colon tumor cells at D0. Mice were randomized based on tumor volume at D9 and treated IP with mab against CTLA-4 (clone 9H10) at 5 mg/kg/inj, PD-1 (clone RMP1-14) at 10 mg/kg/inj (TWx2) or combination of both. Mice were terminated at D20 and tumors analyzed by Flow Cytometry for T cells content. page 15
16 Checkpoint inhibitor summary table Model Checkpoint Inhibitors NGS Exome Seq. Name Type Site Strain CTLA-4 PD-1 PD-L1 4T1 Breast OT BALB/C X A20 BCL SC BALB/C X B16-F10 Melanoma SC C57BL/6 X C38 Colon SC C57BL/6 CT26 Colon SC BALB/C X EMT6 Breast SC BALB/C X Hepa1-6 Liver OT C57BL/6 LLC Lung SC C57BL/6 X MBT-2 Bladder OT SC C3H RenCa Kidney OT BALB/C X X TC < 42% 42% < TC < 80% TC > 80% page 16
17 Exome sequencing and sensitivity to ICIs Total number of mutations PD-1 / PD-L1 Targeting mabs T/C (%) < 80 is used as cut-off criteria for responder and non-responder populations CTLA-4 Targeting mab Champiat S et al OncoImmunology. Exomics and immunogenics. Bridging mutational load and immune checkpoints efficacy. Rizvi NA et al Science 348:6230 page 17
18 Immunological endpoints Cytokines release quantification ELISA / CBA / Luminex Immune cells phenotyping and quantification Flow Cytometry, IHC Immune cells functionality Cell preparation and/or sorting using FACS or magnetic beads Cytotoxic activity based on Cr 51 release assay Proliferation ability based on H 3 -Thd incorporation assay Specific cytokine release based on ELISpot assay CFU page 18
19 Immune infiltrate phenotyping Immune cell phenotyping using Flow Cytometry page 19
20 Humanized mouse models page 20
21 Recipient mice IL2rg impaired Mouse NOG & upgraded NOG Nude Mouse No T cells Humoral immunity intact NK cell activity ++ Hemolytic complement + CB17-SCID Mouse No T cells or B cells Radiosensitive NK cell activity + Hemolytic complement + NOD-SCID Mouse Defects in innate immunity Impaired macrophage activation NK cell activity +/- Hemolytic complement - Defects in innate & adaptive immunity Impaired macrophage activation NK cell function - Hemolytic complement - BRGS Mouse Defects in innate & adaptive immunity Impaired macrophage activation NK cell function - Hemolytic complement + IMMUNODEFICIENCY ENGRAFTMENT WITH HUMAN IMMUNE CELLS page 21
22 PDX panels page 22
23 PDX models: The IMODI consortium Innovative MODels Initiative Agaisnt Cancer Through collaborative partnerships with Clinicians and scientific experts we are building a collection of > 200 PDX models 4 pharma companies 6 CROs 13 academic groups Total budget 41 M Grant 13.4 M 7 years Oncodesign - 20, rue Jean Mazen - BP Dijon Cedex - France Tel. : +33(0) Fax : +33(0)
24 PDX Collection PDX Available for Services Colon 61 Breast 20 Lung NSCLC 17 Pancreas 13 Brain 5 Ovarian 3 Bladder 3 Head & Neck 3 Lung SCLC 3 Kidney 2 Liver 2 Not only numbers, but an extensive characterization and knowledge of our models (Tumor/Gut microbiota of the host/biomarker identification). PDX Ongoing Development Lung 30 Liver 30 Ovarian 30 Breast 20 Pancreas 20 AML 20 Myeloma 10 Lymphoma 10 Prostate 10 AML 2 Prostate 1 Oncodesign - 20, rue Jean Mazen - BP Dijon Cedex - France Tel. : +33(0) Fax : +33(0)
25 Tumor volume (mm 3 ) Tumor volume (mm 3 ) BRGS mice humanized with HSCs and bearing PDX tumors Growth curves are similar if PDX injected in SWISS-Nude mice 1, LUN-NIC , IM-OVA Time (Days) Time (Days) BRGS mice: BALB/C Rag2 -/- IL-2Rγ c -/- SIRPa NOD (Legrand N et al. PNAS : ) page 25
26 Flow Cytometry analyses General comments Blood, spleen, TDLN, tumor, bone marrow collected Identified immune populations Circulating and central human lymphoid and myeloid cells Lymphoid cells: T cells: CD8, CD4 and Treg evidenced B cells NK cells Myeloid cells: Monocytes others page 26
27 Flow Cytometry analyses : blood Blood : non-tumor bearing mice Lymphoid cells ~90% vs Myeloid cells ~10% T cells: total CD3 ~49% with CD8 (~5%), CD4 (~80%) and Treg (~11%) B cells: ~48% NK cells: ~2% Monocytes: ~5% Other myeloid cells: ~5% Proportion dictated by the murine host! page 27
28 Flow Cytometry analyses : blood Blood : non-tumor bearing mice vs tumor bearing mice Change induced by tumor! increase in Treg change in T/B ratio page 28
29 Flow Cytometry analyses : spleen Spleen: non-tumor bearing mice Lymphoid cells ~98% vs Myeloid cells ~2% T cells: total CD3 ~40% with CD8 (~4%), CD4 (~76%) and Treg (~13%) B cells: ~40% NK cells: ~1% Monocytes: ~0.5% Other myeloid cells: ~1.5% page 29
30 Flow Cytometry analyses : spleen Spleen : non-tumor bearing mice vs tumor bearing mice Change induced by tumor! increase in Treg change in T/B ratio page 30
31 Foxp3 hcd20 Flow Cytometry analyses : spleen Spleen : non-tumor bearing mice vs tumor bearing mice Healthy Tumor-bearing ID20 ID36 ID11 ID37 T/B= 3.0 T/B= 1.3 T/B= 0.1 T/B =0.6 hcd hcd4 page 31
32 Foxp3 Flow Cytometry analyses : Tumor Tumor Lymphoid cells & Myeloid cells T cells: ~55% B cells: ~4% NK cells: ~6% Monocytes: ~3% Other myeloid cells: ~5% hcd4 ID9 Treg identified within the tumor (n=2) page 32
33 BRGS mice humanized with HSCs and bearing PDX tumors Conclusions Growth of SC PDX tumors (ovary and lung) Presence of circulating and central lymphoid and myeloid cells Changes induced by tumor evidenced in blood and spleen increase in Treg change in T/B ratio Presence of Tregs within the tumor Model of interest for further evaluation of compounds targeting Tregs or interfering with CD8/CD4 populations (ipilimumab, nivolumab ) page 33
34 Conclusions & Perspectives page 34
35 Summary table Tumor model HSCs NKs PBMCs T cells Name Histology Injection IV IH IP IV IP IV SC IV 786-O Kidney clear cell adenocarcinoma SC 3 BT-474/matrigel Her2+ Breast ductal carcinoma SC Daudi Burkitt's lymphoma, B cells IV 1 x FaDu Head and Neck carcinoma SC 1 HCT116 Colorectal carcinoma SC x IM-OVA-503 PDX: Ovary SC 1 Jeko-1 Mantle Cell Lymphoma, B cells IV x KARPAS-299 Human T-cell non-hodgkin lymphoma IV 4 LoVo Colorectal adenocarcinoma SC 1 2 LUN-NIC-0084 PDX: NSCLC SC 1 MCF-7 Breast adenocarcinoma SC 2 MKN74/matrigel Gastric carcinoma (stomach) SC 1 MOLM13 acute myeloid leukemia IV x NA (no tumor) NA (no tumor) NCI-H929/matrigel Plasma cell myeloma SC 2 NCI-N87 Gastric carcinoma (stomach) SC 1 NIH:OVCAR-3 Ovarian adenocarcinoma SC 2 Raji* B cell lymphoma IV 1 Ramos B cell lymphoma IV 1 3 RPMI 8226* Multiple myeloma SC 1 * Model not growing properly in humanized condition page 35
36 Immunological endpoints Cytokines release quantification ELISA / CBA / Luminex Immune cells phenotyping and quantification 10-color Flow Cytometry Immune cells functionality Cell preparation and/or sorting using FACS or magnetic beads Cytotoxic activity based on 51 Cr release assay Proliferation ability based on CFSE / 3 H-Thd incorporation assay Specific cytokine release based on ELISpot assay Co-culture assay CFU page 36
37 Ongoing & Future Development To be able to evaluate human specific I-O related compounds Humanization with HSCs Mouse strain : BRGS To validate efficacy of approved immune checkpoint inhibitors Humanization with PBMCs CD4+ and CD8+ T cells from PBMCs To characterize the tumor immune infiltrate To validate efficacy of approved immune checkpoint inhibitors In vitro assays Treg suppresor assay Teff tumor cell killing efficacy page 37
38 Special thanks to : Jean-François Mirjolet - Caroline Mignard Jean-Marie Charpin
Species Tumor Type Comment for in vivo work Lead Time for in vivo studies [weeks] MB-49-luc-2 Mouse urinary bladder carcinoma C57BL/6 2
![Species Tumor Type Comment for in vivo work Lead Time for in vivo studies [weeks] MB-49-luc-2 Mouse urinary bladder carcinoma C57BL/6 2 Species Tumor Type Comment for in vivo work Lead Time for in vivo studies [weeks] MB-49-luc-2 Mouse urinary bladder carcinoma C57BL/6 2](/thumbs/88/116309357.jpg) America, Hershey, PA Australia, Melbourne, VIC Europe, Munich info@vivopharm.com www.vivopharm.com Tissue Bladder Species Tumor Type Comment for in vivo work Lead Time for in vivo studies [weeks] MB-49-luc-
America, Hershey, PA Australia, Melbourne, VIC Europe, Munich info@vivopharm.com www.vivopharm.com Tissue Bladder Species Tumor Type Comment for in vivo work Lead Time for in vivo studies [weeks] MB-49-luc-
America, Hershey, PA Australia, Melbourne, VIC Europe, Munich
 America, Hershey, PA Australia, Melbourne, VIC Europe, Munich info@vivopharm.com www.vivopharm.com Bladder MB-9-luc 2 Mouse urinary bladder carcinoma yes yes yes C57BL/6 2 Bone UMR-106 Rat osteosarcoma
America, Hershey, PA Australia, Melbourne, VIC Europe, Munich info@vivopharm.com www.vivopharm.com Bladder MB-9-luc 2 Mouse urinary bladder carcinoma yes yes yes C57BL/6 2 Bone UMR-106 Rat osteosarcoma
Juyoun Jin, D.V.M., Ph.D. Institute for Refractory Cancer Research, Samsung Medical Center
 Juyoun Jin, D.V.M., Ph.D. Institute for Refractory Cancer Research, Samsung Medical Center Overview of Anticancer Drug Development Discovery Non-clinical development Clinical Trial Target Identification
Juyoun Jin, D.V.M., Ph.D. Institute for Refractory Cancer Research, Samsung Medical Center Overview of Anticancer Drug Development Discovery Non-clinical development Clinical Trial Target Identification
Supplementary Figure 1. Double-staining immunofluorescence analysis of invasive colon and breast cancers. Specimens from invasive ductal breast
 Supplementary Figure 1. Double-staining immunofluorescence analysis of invasive colon and breast cancers. Specimens from invasive ductal breast carcinoma (a) and colon adenocarcinoma (b) were staining
Supplementary Figure 1. Double-staining immunofluorescence analysis of invasive colon and breast cancers. Specimens from invasive ductal breast carcinoma (a) and colon adenocarcinoma (b) were staining
Utilizing Humanized NSG Mice to Evaluate Drug Efficacy in Immuno-Oncology
 Utilizing Humanized NSG Mice to Evaluate Drug Efficacy in Immuno-Oncology Rick Huntress Director Business Development March 15, 2017 Onco-Hu : Humanized Mice for Evaluation of Immuno-Oncology Therapeutics
Utilizing Humanized NSG Mice to Evaluate Drug Efficacy in Immuno-Oncology Rick Huntress Director Business Development March 15, 2017 Onco-Hu : Humanized Mice for Evaluation of Immuno-Oncology Therapeutics
A new Rag2/Il2rg SCID rat. Enabling cell line xenografts. Accelerating PDX establishment. Humanizing the immune system
 A new Rag2/Il2rg SCID rat Enabling cell line xenografts Accelerating PDX establishment Humanizing the immune system Tseten Yeshi, Ph.D. VP R&D services@herabiolabs.com 859-414-0648 About Hera BioLabs SRG
A new Rag2/Il2rg SCID rat Enabling cell line xenografts Accelerating PDX establishment Humanizing the immune system Tseten Yeshi, Ph.D. VP R&D services@herabiolabs.com 859-414-0648 About Hera BioLabs SRG
ONCO-HU TM MICE FOR IMMUNOTHERAPEUTIC DRUG DISCOVERY. Brian W. Soper, Ph.D. Senior Technical Information Scientist
 ONCO-HU TM MICE FOR IMMUNOTHERAPEUTIC DRUG DISCOVERY Brian W. Soper, Ph.D. Senior Technical Information Scientist Presentation Outline Capabilities Humanization Hu-NSG TM versus Hu-NSG TM -SGM3 Immuno-oncology
ONCO-HU TM MICE FOR IMMUNOTHERAPEUTIC DRUG DISCOVERY Brian W. Soper, Ph.D. Senior Technical Information Scientist Presentation Outline Capabilities Humanization Hu-NSG TM versus Hu-NSG TM -SGM3 Immuno-oncology
Juyoun Jin, D.V.M., Ph.D. Institute for Refractory Cancer Research, Samsung Medical Center
 Juyoun Jin, D.V.M., Ph.D. Institute for Refractory Cancer Research, Samsung Medical Center Animal Models for Translational Research How to apply preclinical translational research New trial concept of
Juyoun Jin, D.V.M., Ph.D. Institute for Refractory Cancer Research, Samsung Medical Center Animal Models for Translational Research How to apply preclinical translational research New trial concept of
Immune Checkpoint Inhibitors Drug Combinations: Patients Relevance & Ways Forward
 March 25, 2015 Immune Checkpoint Inhibitors Drug Combinations: Patients Relevance & Ways Forward Dr. Alexandre Passioukov p 1 Therapeutic efficacy of agents targeting immune checkpoints Introduction Deep
March 25, 2015 Immune Checkpoint Inhibitors Drug Combinations: Patients Relevance & Ways Forward Dr. Alexandre Passioukov p 1 Therapeutic efficacy of agents targeting immune checkpoints Introduction Deep
Checkpoint Regulators Cancer Immunotherapy takes centre stage. Dr Oliver Klein Department of Medical Oncology 02 May 2015
 Checkpoint Regulators Cancer Immunotherapy takes centre stage Dr Oliver Klein Department of Medical Oncology 02 May 2015 Adjuvant chemotherapy improves outcome in early breast cancer FDA approval of Imatinib
Checkpoint Regulators Cancer Immunotherapy takes centre stage Dr Oliver Klein Department of Medical Oncology 02 May 2015 Adjuvant chemotherapy improves outcome in early breast cancer FDA approval of Imatinib
Novel RCC Targets from Immuno-Oncology and Antibody-Drug Conjugates
 Novel RCC Targets from Immuno-Oncology and Antibody-Drug Conjugates Christopher Turner, MD Vice President, Clinical Science 04 November 2016 Uveal Melanoma Celldex Pipeline CANDIDATE INDICATION Preclinical
Novel RCC Targets from Immuno-Oncology and Antibody-Drug Conjugates Christopher Turner, MD Vice President, Clinical Science 04 November 2016 Uveal Melanoma Celldex Pipeline CANDIDATE INDICATION Preclinical
CB-1158 Inhibits the Immuno-oncology Target Arginase and Causes an Immune Mediated Anti-Tumor Response
 CB-1158 Inhibits the Immuno-oncology Target Arginase and Causes an Immune Mediated Anti-Tumor Response Francesco Parlati, Ph.D. Calithera Biosciences South San Francisco, CA Arginine Depletion by Arginase
CB-1158 Inhibits the Immuno-oncology Target Arginase and Causes an Immune Mediated Anti-Tumor Response Francesco Parlati, Ph.D. Calithera Biosciences South San Francisco, CA Arginine Depletion by Arginase
Overview for today. NSG and NSG -SGM3 are a proven platform. A growing list of drugs have shown translationally relevant response
 1 Overview for today NSG and NSG -SGM3 are a proven platform A growing list of drugs have shown translationally relevant response Targeted therapeutics and anti-angiogenesis drugs show their expected response
1 Overview for today NSG and NSG -SGM3 are a proven platform A growing list of drugs have shown translationally relevant response Targeted therapeutics and anti-angiogenesis drugs show their expected response
Outline. Case studies & Tumor Immunology platform overview. In vitro assay panel. In vivo models. Clinical and pre-clinical sample analyses
 Immuno-Oncology May, 217 Outline Case studies & Tumor Immunology platform overview In vitro assay panel In vivo models Ex vivo analyses Clinical and pre-clinical sample analyses 2 Case 1: an IS Project
Immuno-Oncology May, 217 Outline Case studies & Tumor Immunology platform overview In vitro assay panel In vivo models Ex vivo analyses Clinical and pre-clinical sample analyses 2 Case 1: an IS Project
Combining ADCs with Immuno-Oncology Agents
 Combining ADCs with Immuno-Oncology Agents Chad May, PhD Senior Director Targeted Immunotherapy Oncology Research Unit, Pfizer 7 th Annual World ADC October 10, 2016 Cancer-Immunity Cycle Innate Immunity
Combining ADCs with Immuno-Oncology Agents Chad May, PhD Senior Director Targeted Immunotherapy Oncology Research Unit, Pfizer 7 th Annual World ADC October 10, 2016 Cancer-Immunity Cycle Innate Immunity
Immuno-Modulatory Drug and Biomarker Discovery Using Onco-Hu Mice
 Immuno-Modulatory Drug and Biomarker Discovery Using Onco-Hu Mice Brian W. Soper, PhD Senior Technical Information Scientist Manager, Technical Information Services Dec, 2017 Presentation Overview 12-16
Immuno-Modulatory Drug and Biomarker Discovery Using Onco-Hu Mice Brian W. Soper, PhD Senior Technical Information Scientist Manager, Technical Information Services Dec, 2017 Presentation Overview 12-16
Beverly A. Teicher, PhD DCTD/NCI. The content reflects my professional opinions, not an NCI policy statement.
 Beverly A. Teicher, PhD DCTD/NCI The content reflects my professional opinions, not an NCI policy statement. Outline 1. Transplantable Syngeneic Tumors 2. Human Tumor Xenografts 3. Disseminated Disease
Beverly A. Teicher, PhD DCTD/NCI The content reflects my professional opinions, not an NCI policy statement. Outline 1. Transplantable Syngeneic Tumors 2. Human Tumor Xenografts 3. Disseminated Disease
Supplementary Figure 1. Characterization of basophils after reconstitution of SCID mice
 Supplementary figure legends Supplementary Figure 1. Characterization of after reconstitution of SCID mice with CD4 + CD62L + T cells. (A-C) SCID mice (n = 6 / group) were reconstituted with 2 x 1 6 CD4
Supplementary figure legends Supplementary Figure 1. Characterization of after reconstitution of SCID mice with CD4 + CD62L + T cells. (A-C) SCID mice (n = 6 / group) were reconstituted with 2 x 1 6 CD4
A Versatile Tool to Study Immune Checkpoint Therapeutics: Syngeneic Tumor Mouse Models in vivo
 The Solution Provider for Drug Discovery in Oncology A Versatile Tool to Study Immune Checkpoint Therapeutics: Syngeneic Tumor Mouse Models in vivo Holger Weber - 1 - Drug discovery platform for oncology
The Solution Provider for Drug Discovery in Oncology A Versatile Tool to Study Immune Checkpoint Therapeutics: Syngeneic Tumor Mouse Models in vivo Holger Weber - 1 - Drug discovery platform for oncology
Syngeneic Models. Progress immunotherapeutic development with CrownBio s panel of over 30 syngeneic models
 Syngeneic Models Progress immunotherapeutic development with CrownBio s panel of over 3 syngeneic models Discover the benefits of using our fully characterized and checkpoint inhibitor benchmarked syngeneic
Syngeneic Models Progress immunotherapeutic development with CrownBio s panel of over 3 syngeneic models Discover the benefits of using our fully characterized and checkpoint inhibitor benchmarked syngeneic
Immunotherapie: algemene principes
 Immunotherapie: algemene principes Prof. dr. Evelien Smits Tumorimmunologie, UAntwerpen 14 Oktober 2017, IKG evelien.smits@uza.be Concept of immune evasion Finn O. J. Ann Oncol. 2012 Sep; 23(Suppl 8):
Immunotherapie: algemene principes Prof. dr. Evelien Smits Tumorimmunologie, UAntwerpen 14 Oktober 2017, IKG evelien.smits@uza.be Concept of immune evasion Finn O. J. Ann Oncol. 2012 Sep; 23(Suppl 8):
Supplemental Materials
 Supplemental Materials Programmed death one homolog maintains the pool size of regulatory T cells by promoting their differentiation and stability Qi Wang 1, Jianwei He 1, Dallas B. Flies 2, Liqun Luo
Supplemental Materials Programmed death one homolog maintains the pool size of regulatory T cells by promoting their differentiation and stability Qi Wang 1, Jianwei He 1, Dallas B. Flies 2, Liqun Luo
Immuno-Oncology. Axel Hoos, MD, PhD Senior Vice President, Oncology R&D. February 24, 2016
 Immuno-Oncology Axel Hoos, MD, PhD Senior Vice President, Oncology R&D February 24, 216 GSK Pipeline Oncology R&D strategy Focusing on 3 areas fundamental to oncology Cancer Epigenetics Long-Term Survival
Immuno-Oncology Axel Hoos, MD, PhD Senior Vice President, Oncology R&D February 24, 216 GSK Pipeline Oncology R&D strategy Focusing on 3 areas fundamental to oncology Cancer Epigenetics Long-Term Survival
In Vitro and Ex Vivo Immuno- Oncology Drug Discovery
 Oncology Drug Discovery CrownBio s integrated Immuno-Oncology Platform allows end-to-end in vitro and ex vivo immunoprofiling, functional assay evaluation, and novel target validation Discover how CrownBio
Oncology Drug Discovery CrownBio s integrated Immuno-Oncology Platform allows end-to-end in vitro and ex vivo immunoprofiling, functional assay evaluation, and novel target validation Discover how CrownBio
Reprogramming Tumor Associated Dendritic Cells for Immunotherapy
 Reprogramming Tumor Associated Dendritic Cells for Immunotherapy Edgar Engleman, M.D. Professor of Pathology and Medicine Stanford University Disclosures: Founder of Dendreon, a biotechnology company that
Reprogramming Tumor Associated Dendritic Cells for Immunotherapy Edgar Engleman, M.D. Professor of Pathology and Medicine Stanford University Disclosures: Founder of Dendreon, a biotechnology company that
Biological Therapies for Cancer: Questions and Answers
 Biological Therapies for Cancer: Questions and Answers Key Points Biological therapies use the body s immune system to fight cancer or to lessen the side effects that may be caused by some cancer treatments
Biological Therapies for Cancer: Questions and Answers Key Points Biological therapies use the body s immune system to fight cancer or to lessen the side effects that may be caused by some cancer treatments
Evaluation of an Agonist Anti-CD27 Human Antibody (Varlilumab) and its Potential for Combination With Checkpoint Inhibitors
 Evaluation of an Agonist Anti-CD27 Human Antibody (Varlilumab) and its Potential for Combination With Checkpoint Inhibitors Tibor Keler, PhD Chief Scientific Officer Enhancing immunity with immune modulating
Evaluation of an Agonist Anti-CD27 Human Antibody (Varlilumab) and its Potential for Combination With Checkpoint Inhibitors Tibor Keler, PhD Chief Scientific Officer Enhancing immunity with immune modulating
Using PDX Models for Preclinical Trials of Oncology Therapeutics: Immunooncology & beyond. Thomas B. Broudy, PhD CSO Crown Bioscience, San Diego
 Using PDX Models for Preclinical Trials of Oncology Therapeutics: Immunooncology & beyond Thomas B. Broudy, PhD CSO Crown Bioscience, San Diego CrownBio: Global Oncology Focused Partner Global capacity
Using PDX Models for Preclinical Trials of Oncology Therapeutics: Immunooncology & beyond Thomas B. Broudy, PhD CSO Crown Bioscience, San Diego CrownBio: Global Oncology Focused Partner Global capacity
An integrative approach to cancer precision medicine
 2017 Korean Society of Gynecologic Oncology Symposium An integrative approach to cancer precision medicine Sung-Yup Cho, M.D., Ph.D. Ewha Institute of Convergence Medicine EWHA WOMANS UNIVERSITY MEDICAL
2017 Korean Society of Gynecologic Oncology Symposium An integrative approach to cancer precision medicine Sung-Yup Cho, M.D., Ph.D. Ewha Institute of Convergence Medicine EWHA WOMANS UNIVERSITY MEDICAL
Immune Checkpoint Inhibitors: The New Breakout Stars in Cancer Treatment
 Immune Checkpoint Inhibitors: The New Breakout Stars in Cancer Treatment 1 Introductions Peter Langecker, MD, PhD Executive Medical Director, Global Oncology Clinipace Worldwide Mark Shapiro Vice President
Immune Checkpoint Inhibitors: The New Breakout Stars in Cancer Treatment 1 Introductions Peter Langecker, MD, PhD Executive Medical Director, Global Oncology Clinipace Worldwide Mark Shapiro Vice President
CHMP Oncology Working Party Workshop on: Histology Independent Indications in Oncology. Non-clinical models: Tumour Models - Proof of Concept
 CHMP Oncology Working Party Workshop on: Histology Independent Indications in Oncology Non-clinical models: Tumour Models - Proof of Concept Edward C. Rosfjord Pfizer Worldwide R. & D. 14 December 2017
CHMP Oncology Working Party Workshop on: Histology Independent Indications in Oncology Non-clinical models: Tumour Models - Proof of Concept Edward C. Rosfjord Pfizer Worldwide R. & D. 14 December 2017
Blocking antibodies and peptides. Rat anti-mouse PD-1 (29F.1A12, rat IgG2a, k), PD-
 Supplementary Methods Blocking antibodies and peptides. Rat anti-mouse PD-1 (29F.1A12, rat IgG2a, k), PD- L1 (10F.9G2, rat IgG2b, k), and PD-L2 (3.2, mouse IgG1) have been described (24). Anti-CTLA-4 (clone
Supplementary Methods Blocking antibodies and peptides. Rat anti-mouse PD-1 (29F.1A12, rat IgG2a, k), PD- L1 (10F.9G2, rat IgG2b, k), and PD-L2 (3.2, mouse IgG1) have been described (24). Anti-CTLA-4 (clone
SUPPLEMENTARY INFORMATION
 SUPPLEMENTARY NFORMATON n format provided by Melero et al. (AUGUST 2015) Supplementary nformation S3 Combinations including two or more immunotherapy agents based on PD-1/PD-L1 blockade. (Source: https://clinicaltrials.gov/
SUPPLEMENTARY NFORMATON n format provided by Melero et al. (AUGUST 2015) Supplementary nformation S3 Combinations including two or more immunotherapy agents based on PD-1/PD-L1 blockade. (Source: https://clinicaltrials.gov/
Immuno-Oncology Applications
 Immuno-Oncology Applications Lee S. Schwartzberg, MD, FACP West Clinic, P.C.; The University of Tennessee Memphis, Tn. ICLIO 1 st Annual National Conference 10.2.15 Philadelphia, Pa. Financial Disclosures
Immuno-Oncology Applications Lee S. Schwartzberg, MD, FACP West Clinic, P.C.; The University of Tennessee Memphis, Tn. ICLIO 1 st Annual National Conference 10.2.15 Philadelphia, Pa. Financial Disclosures
Emerging Concepts of Cancer Immunotherapy
 Emerging Concepts of Cancer Immunotherapy Jeffrey Schlom, Ph.D. Laboratory of Tumor Immunology and Biology (LTIB) Center for Cancer Research National Cancer Institute, NIH Immune Cell Infiltrate in Primary
Emerging Concepts of Cancer Immunotherapy Jeffrey Schlom, Ph.D. Laboratory of Tumor Immunology and Biology (LTIB) Center for Cancer Research National Cancer Institute, NIH Immune Cell Infiltrate in Primary
Immuno-Oncology Clinical Trials Update: Checkpoint Inhibitors Others (not Anti-PD-L1/PD-1) Issue 4 January 2017
 Delivering a Competitive Intelligence Advantage Immuno-Oncology Clinical Trials Update: Checkpoint Inhibitors Others (not Anti-PD-L1/PD-1) Issue 4 January 2017 Immuno-Oncology CLINICAL TRIALS UPDATE The
Delivering a Competitive Intelligence Advantage Immuno-Oncology Clinical Trials Update: Checkpoint Inhibitors Others (not Anti-PD-L1/PD-1) Issue 4 January 2017 Immuno-Oncology CLINICAL TRIALS UPDATE The
Daratumumab, a novel human CD38 monoclonal antibody for the treatment of B cell Non Hodgkin Lymphoma
 Daratumumab, a novel human CD38 monoclonal antibody for the treatment of B cell Non Hodgkin Lymphoma Anna Vidal Department of Hemato Oncology IDIBAPS, Barcelona, Spain B cell lymphoid malignancies seen
Daratumumab, a novel human CD38 monoclonal antibody for the treatment of B cell Non Hodgkin Lymphoma Anna Vidal Department of Hemato Oncology IDIBAPS, Barcelona, Spain B cell lymphoid malignancies seen
NKTR-214 plus NKTR-262, a Scientifically-Guided Rational Combination Approach for Immune Oncology
 plus NKTR-262, a Scientifically-Guided Rational Combination Approach for Immune Oncology Jonathan Zalevsky SVP, Biology and Preclinical Development Nektar Therapeutics World Preclinical Congress, 2017
plus NKTR-262, a Scientifically-Guided Rational Combination Approach for Immune Oncology Jonathan Zalevsky SVP, Biology and Preclinical Development Nektar Therapeutics World Preclinical Congress, 2017
Pediatric Preclinical Testing Program (PPTP) evaluation of the Bcl-2 inhibitor ABT-263
 Pediatric Preclinical Testing Program (PPTP) evaluation of the Bcl-2 inhibitor ABT-263 Malcolm A. Smith, John M. Maris, Stephen T. Keir, Henry S. Friedman, Richard B. Lock, Hernan Carol, Mayamin Tajbakhsh,
Pediatric Preclinical Testing Program (PPTP) evaluation of the Bcl-2 inhibitor ABT-263 Malcolm A. Smith, John M. Maris, Stephen T. Keir, Henry S. Friedman, Richard B. Lock, Hernan Carol, Mayamin Tajbakhsh,
SUPPLEMENTARY INFORMATION
 Table S1: Combination Immunotherapy: Small Molecules that Boost Immune Response in Combination with Other Agents Target Compound (MOA) NLG919 and indoximod 1- MT Combination Model Observation Reference
Table S1: Combination Immunotherapy: Small Molecules that Boost Immune Response in Combination with Other Agents Target Compound (MOA) NLG919 and indoximod 1- MT Combination Model Observation Reference
Human cell line list Production of Exosome Standards - Cell Lysates
 Human cell line list 2016 - Production of Exosome Standards - Cell Lysates 380 peripheral blood leukemia, pre-b cell 1301 blood leukemia, acute lymphoblastic, T cell 5637 bladder carcinoma 8305C thyroid
Human cell line list 2016 - Production of Exosome Standards - Cell Lysates 380 peripheral blood leukemia, pre-b cell 1301 blood leukemia, acute lymphoblastic, T cell 5637 bladder carcinoma 8305C thyroid
Posters and Presentations
 Posters and Presentations June 2017: American Society of Clinical Oncology (ASCO) Annual - Preliminary Correlative Analysis of PD-L1 expression from the SUNRISE Study. View April 2017: American Association
Posters and Presentations June 2017: American Society of Clinical Oncology (ASCO) Annual - Preliminary Correlative Analysis of PD-L1 expression from the SUNRISE Study. View April 2017: American Association
Charles River Immunodeficient Models Xenograft Data Catalog
 Charles River Immunodeficient Models Xenograft Data Catalog Fox Chase SCID Congenic (C.B-17 SCID) Mouse Xenograft Data askcharlesriver@crl.com www.criver.com 214, Charles River Laboratories International,
Charles River Immunodeficient Models Xenograft Data Catalog Fox Chase SCID Congenic (C.B-17 SCID) Mouse Xenograft Data askcharlesriver@crl.com www.criver.com 214, Charles River Laboratories International,
Characterization of immune responses to anti-pd-1 mono and combination immunotherapy in hematopoietic humanized mice implanted with tumor xenografts
 Capasso et al. Journal for ImmunoTherapy of Cancer (2019) 7:37 https://doi.org/10.1186/s40425-019-0518-z RESEARCH ARTICLE Open Access Characterization of immune responses to anti-pd-1 mono and combination
Capasso et al. Journal for ImmunoTherapy of Cancer (2019) 7:37 https://doi.org/10.1186/s40425-019-0518-z RESEARCH ARTICLE Open Access Characterization of immune responses to anti-pd-1 mono and combination
PRELIMINARY PROGRAM MAY 31 - JUNE 4, 2019
 MONDAY, JUNE 3, 2019 7:30 AM - 9:15 AM HIGHLIGHTS OF THE DAY 8:00 AM - 9:00 AM S 8:00 AM - 9:15 AM S S 8:00 AM - 11:00 AM S Highlights of the Day Session II Bringing New and Innovative Glioblastoma Treatments
MONDAY, JUNE 3, 2019 7:30 AM - 9:15 AM HIGHLIGHTS OF THE DAY 8:00 AM - 9:00 AM S 8:00 AM - 9:15 AM S S 8:00 AM - 11:00 AM S Highlights of the Day Session II Bringing New and Innovative Glioblastoma Treatments
The Current Status of Immune Checkpoint Inhibitors: Arvin Yang, MD PhD Oncology Global Clinical Research Bristol-Myers Squibb
 The Current Status of Immune Checkpoint Inhibitors: A Global Overview of the Field Arvin Yang, MD PhD Oncology Global Clinical Research Bristol-Myers Squibb Immune Checkpoint Inhibitors Conference, March
The Current Status of Immune Checkpoint Inhibitors: A Global Overview of the Field Arvin Yang, MD PhD Oncology Global Clinical Research Bristol-Myers Squibb Immune Checkpoint Inhibitors Conference, March
Radiation Therapy as an Immunomodulator
 Radiation Therapy as an Immunomodulator Yvonne Mowery, MD, PhD February 20, 2017 Tumor/Immune System Balance Kalbasi, JCI 2013 UNC-Duke-NC State-Wake Forest Spring 2017 2 RT Can Shift Balance Toward Elimination
Radiation Therapy as an Immunomodulator Yvonne Mowery, MD, PhD February 20, 2017 Tumor/Immune System Balance Kalbasi, JCI 2013 UNC-Duke-NC State-Wake Forest Spring 2017 2 RT Can Shift Balance Toward Elimination
GSK Oncology. Axel Hoos, MD, PhD Senior Vice President, Oncology R&D. March 8, 2017
 GSK Oncology Axel Hoos, MD, PhD Senior Vice President, Oncology R&D March 8, 217 GSK pipeline Oncology R&D Strategy Maximizing survival through transformational medicines and combinations Cancer Epigenetics
GSK Oncology Axel Hoos, MD, PhD Senior Vice President, Oncology R&D March 8, 217 GSK pipeline Oncology R&D Strategy Maximizing survival through transformational medicines and combinations Cancer Epigenetics
Enhanced Cancer Vaccine Effectiveness with NKTR-214, a CD122-Biased Cytokine
 Enhanced Cancer Vaccine Effectiveness with NKTR-214, a CD122-Biased Cytokine Jonathan Zalevsky SVP, Biology and Preclinical Development Nektar Therapeutics SMI Cancer Vaccines, September 2017 Nektar Therapeutics
Enhanced Cancer Vaccine Effectiveness with NKTR-214, a CD122-Biased Cytokine Jonathan Zalevsky SVP, Biology and Preclinical Development Nektar Therapeutics SMI Cancer Vaccines, September 2017 Nektar Therapeutics
Cancer Immunotherapy Survey
 CHAPTER 8: Cancer Immunotherapy Survey All (N=100) Please classify your organization. Academic lab or center Small biopharmaceutical company Top 20 Pharma Mid-size pharma Diagnostics company Other (please
CHAPTER 8: Cancer Immunotherapy Survey All (N=100) Please classify your organization. Academic lab or center Small biopharmaceutical company Top 20 Pharma Mid-size pharma Diagnostics company Other (please
COURSE: Medical Microbiology, PAMB 650/720 - Fall 2008 Lecture 16
 COURSE: Medical Microbiology, PAMB 650/720 - Fall 2008 Lecture 16 Tumor Immunology M. Nagarkatti Teaching Objectives: Introduction to Cancer Immunology Know the antigens expressed by cancer cells Understand
COURSE: Medical Microbiology, PAMB 650/720 - Fall 2008 Lecture 16 Tumor Immunology M. Nagarkatti Teaching Objectives: Introduction to Cancer Immunology Know the antigens expressed by cancer cells Understand
Highlights from AACR 2015: The Emerging Potential of Immunotherapeutic Approaches in Non-Small Cell Lung Cancer
 Transcript Details This is a transcript of a continuing medical education (CME) activity accessible on the ReachMD network. Additional media formats for the activity and full activity details (including
Transcript Details This is a transcript of a continuing medical education (CME) activity accessible on the ReachMD network. Additional media formats for the activity and full activity details (including
Immunotherapy in Lung Cancer - TLR9 as a therapeutic target -
 Immunotherapy in Lung Cancer - TLR9 as a therapeutic target - Wilfried Eberhardt,, MD Head of Outpatient Unit, Dept. of Internal Medicine (Cancer Research) West German Cancer Centre Essen University Hospital
Immunotherapy in Lung Cancer - TLR9 as a therapeutic target - Wilfried Eberhardt,, MD Head of Outpatient Unit, Dept. of Internal Medicine (Cancer Research) West German Cancer Centre Essen University Hospital
Targeting tumour associated macrophages in anti-cancer therapies. Annamaria Gal Seminar Series on Drug Discovery Budapest 5 January 2018
 Targeting tumour associated macrophages in anti-cancer therapies Annamaria Gal Seminar Series on Drug Discovery Budapest 5 January 2018 Macrophages: Professional phagocytes of the myeloid lineage APC,
Targeting tumour associated macrophages in anti-cancer therapies Annamaria Gal Seminar Series on Drug Discovery Budapest 5 January 2018 Macrophages: Professional phagocytes of the myeloid lineage APC,
Predictive Biomarkers for Pembrolizumab. Eric H. Rubin, M.D.
 Predictive Biomarkers for Pembrolizumab Eric H. Rubin, M.D. PD-1 and PD-L1/L2 Pathway PD-1 is an immune checkpoint receptor Binding of PD-1 by its ligands PD-L1 or PD-L2 leads to downregulation of T-cell
Predictive Biomarkers for Pembrolizumab Eric H. Rubin, M.D. PD-1 and PD-L1/L2 Pathway PD-1 is an immune checkpoint receptor Binding of PD-1 by its ligands PD-L1 or PD-L2 leads to downregulation of T-cell
Dual-targeting anti- CD47 bispecific antibodies: Safely provoking macrophages to feast NALM-6 tumor cells (B-ALL) Human macrophages
 Dual-targeting anti- CD47 bispecific antibodies: Safely provoking macrophages to feast Krzysztof Masternak, PhD, Head of Biology 1 2 nd Biologics & BOARD Biosimilars MEETING Congress MARCH 2012 NALM-6
Dual-targeting anti- CD47 bispecific antibodies: Safely provoking macrophages to feast Krzysztof Masternak, PhD, Head of Biology 1 2 nd Biologics & BOARD Biosimilars MEETING Congress MARCH 2012 NALM-6
NKTR-255: Accessing IL-15 Therapeutic Potential through Robust and Sustained Engagement of Innate and Adaptive Immunity
 NKTR-255: Accessing IL-15 Therapeutic Potential through Robust and Sustained Engagement of Innate and Adaptive Immunity Peiwen Kuo Scientist, Research Biology Nektar Therapeutics August 31 st, 2018 Emerging
NKTR-255: Accessing IL-15 Therapeutic Potential through Robust and Sustained Engagement of Innate and Adaptive Immunity Peiwen Kuo Scientist, Research Biology Nektar Therapeutics August 31 st, 2018 Emerging
B Cells Promote Pancreatic Tumorigenesis
 B Cells Promote Pancreatic Tumorigenesis The MIT Faculty has made this article openly available. Please share how this access benefits you. Your story matters. Citation As Published Publisher Roghanian,
B Cells Promote Pancreatic Tumorigenesis The MIT Faculty has made this article openly available. Please share how this access benefits you. Your story matters. Citation As Published Publisher Roghanian,
Immunotherapy and Targeted Therapies: The new face of cancer treatment
 Immunotherapy and Targeted Therapies: The new face of cancer treatment Abdulazeez Salawu MBBS, MSc, PhD, MRCP Academic Clinical Lecturer Weston Park Hospital, Sheffield Novel Systemic Anti-cancer Therapies
Immunotherapy and Targeted Therapies: The new face of cancer treatment Abdulazeez Salawu MBBS, MSc, PhD, MRCP Academic Clinical Lecturer Weston Park Hospital, Sheffield Novel Systemic Anti-cancer Therapies
Treating cancer in HIV infected patients. Professor Mark Bower National Centre for HIV malignancy Chelsea & Westminster Hospital
 Treating cancer in HIV infected patients Professor Mark Bower National Centre for HIV malignancy Chelsea & Westminster Hospital AIDS defining malignancies Malignancy Kaposi s sarcoma Virus KSHV Rate ratio
Treating cancer in HIV infected patients Professor Mark Bower National Centre for HIV malignancy Chelsea & Westminster Hospital AIDS defining malignancies Malignancy Kaposi s sarcoma Virus KSHV Rate ratio
Professor Mark Bower Chelsea and Westminster Hospital, London
 Professor Mark Bower Chelsea and Westminster Hospital, London Cancer immunotherapy & HIV Disclosures: None Lessons for oncology from HIV Awareness and advocacy Activism Rational drug design Prescribing
Professor Mark Bower Chelsea and Westminster Hospital, London Cancer immunotherapy & HIV Disclosures: None Lessons for oncology from HIV Awareness and advocacy Activism Rational drug design Prescribing
Tumor Immunology. Wirsma Arif Harahap Surgical Oncology Consultant
 Tumor Immunology Wirsma Arif Harahap Surgical Oncology Consultant 1) Immune responses that develop to cancer cells 2) Escape of cancer cells 3) Therapies: clinical and experimental Cancer cells can be
Tumor Immunology Wirsma Arif Harahap Surgical Oncology Consultant 1) Immune responses that develop to cancer cells 2) Escape of cancer cells 3) Therapies: clinical and experimental Cancer cells can be
ESMO Preceptorship Breast Cancer. Giuseppe Curigliano MD, PhD Breast Cancer Program Division of Early Drug Development Istituto Europeo di Oncologia
 ESMO Preceptorship Breast Cancer Giuseppe Curigliano MD, PhD Breast Cancer Program Division of Early Drug Development Istituto Europeo di Oncologia Outline Rational for immune-based therapy in BC How to
ESMO Preceptorship Breast Cancer Giuseppe Curigliano MD, PhD Breast Cancer Program Division of Early Drug Development Istituto Europeo di Oncologia Outline Rational for immune-based therapy in BC How to
ICLIO National Conference
 ICLIO National Conference Immuno-oncology In The Clinic Today Lee Schwartzberg, MD, FACP Executive Director, West Cancer Center Chief, Division of Hematology/Oncology University of Tennessee Health Science
ICLIO National Conference Immuno-oncology In The Clinic Today Lee Schwartzberg, MD, FACP Executive Director, West Cancer Center Chief, Division of Hematology/Oncology University of Tennessee Health Science
Humanized Mouse Models for Vaccine Development Michael A. Brehm. Why Do We Need Humanized Mouse Models?
 Humanized Mouse Models for Vaccine Development Michael A. Brehm Diabetes Center of Excellence Dale Greiner Rita Bortell Philip DiIorio Nancy Phillips The Jackson Laboratory Leonard Shultz Human cells &
Humanized Mouse Models for Vaccine Development Michael A. Brehm Diabetes Center of Excellence Dale Greiner Rita Bortell Philip DiIorio Nancy Phillips The Jackson Laboratory Leonard Shultz Human cells &
Corporate Presentation May Transforming Immuno-Oncology Using Next-Generation Immune Cell Engagers
 Corporate Presentation May 2016 Transforming Immuno-Oncology Using Next-Generation Immune Cell Engagers Forward-looking statements / safe harbor This presentation and the accompanying oral commentary contain
Corporate Presentation May 2016 Transforming Immuno-Oncology Using Next-Generation Immune Cell Engagers Forward-looking statements / safe harbor This presentation and the accompanying oral commentary contain
(generic name: ipilimumab) Injection 50 mg ( Yervoy ), a human anti-human CTLA-4 monoclonal. August 21, 2018
 August 21, 2018 Opdivo Approved for Supplemental Applications for Expanded Indications of Malignant Pleural Mesothelioma and Adjuvant Treatment of Melanoma, Change in Dosage and Administration (D&A) of
August 21, 2018 Opdivo Approved for Supplemental Applications for Expanded Indications of Malignant Pleural Mesothelioma and Adjuvant Treatment of Melanoma, Change in Dosage and Administration (D&A) of
Interpreting Therapeutic Response on Immune Cell Number and Spatial Distribution within the Tumor Microenvironment. Lorcan Sherry, CSO OracleBio
 Interpreting Therapeutic Response on Immune Cell Number and Spatial Distribution within the Tumor Microenvironment Lorcan Sherry, CSO OracleBio Company Overview OracleBio is a specialised CRO providing
Interpreting Therapeutic Response on Immune Cell Number and Spatial Distribution within the Tumor Microenvironment Lorcan Sherry, CSO OracleBio Company Overview OracleBio is a specialised CRO providing
L1 on PyMT tumor cells but Py117 cells are more responsive to IFN-γ. (A) Flow
 A MHCI B PD-L1 Fold expression 8 6 4 2 Fold expression 3 2 1 No tx 1Gy 2Gy IFN Py117 Py117 Supplementary Figure 1. Radiation and IFN-γ enhance MHCI expression and PD- L1 on PyMT tumor cells but Py117 cells
A MHCI B PD-L1 Fold expression 8 6 4 2 Fold expression 3 2 1 No tx 1Gy 2Gy IFN Py117 Py117 Supplementary Figure 1. Radiation and IFN-γ enhance MHCI expression and PD- L1 on PyMT tumor cells but Py117 cells
Immunologic Effects of Clinical Stage FAK & PI3K-Delta/Gamma Inhibitors
 1 Immunologic Effects of Clinical Stage FAK & PI3K-Delta/Gamma Inhibitors Jonathan Pachter, Ph.D - Chief Scientific Officer 3rd Annual Immuno-Oncology Congress, May 24, 2018 Disclosures 2 I am an employee
1 Immunologic Effects of Clinical Stage FAK & PI3K-Delta/Gamma Inhibitors Jonathan Pachter, Ph.D - Chief Scientific Officer 3rd Annual Immuno-Oncology Congress, May 24, 2018 Disclosures 2 I am an employee
El contexto molecular de la sobreexpresión de PD-L1 Esther Conde Gallego, MD, PhD
 El contexto molecular de la sobreexpresión de PD-L1 Esther Conde Gallego, MD, PhD Laboratorio de Dianas Terapéuticas Hospital Universitario HM Sanchinarro Madrid, Spain Contents Background PD-L1 expression
El contexto molecular de la sobreexpresión de PD-L1 Esther Conde Gallego, MD, PhD Laboratorio de Dianas Terapéuticas Hospital Universitario HM Sanchinarro Madrid, Spain Contents Background PD-L1 expression
New Developments in Cancer Treatment. Dulcinea Quintana, MD
 New Developments in Cancer Treatment Dulcinea Quintana, MD Mortality Rates Goals of treatment 1 Cure Goal of treatment 2 Prolong life Goals of treatment 3 Improve Quality of Life Goals of treatment 4
New Developments in Cancer Treatment Dulcinea Quintana, MD Mortality Rates Goals of treatment 1 Cure Goal of treatment 2 Prolong life Goals of treatment 3 Improve Quality of Life Goals of treatment 4
We re Reaching Ludicrous Speed: New Immunotherapy Oncology Medications
 We re Reaching Ludicrous Speed: New Immunotherapy Oncology Medications Adam Peele, PharmD, BCPS, BCOP Oncology Pharmacy Manager Cone Health Disclosures Merck Pharmaceuticals Speaker s Bureau 1 Objectives
We re Reaching Ludicrous Speed: New Immunotherapy Oncology Medications Adam Peele, PharmD, BCPS, BCOP Oncology Pharmacy Manager Cone Health Disclosures Merck Pharmaceuticals Speaker s Bureau 1 Objectives
National Cancer Drugs Fund List - Approved
 National Cancer Drugs Fund List - Approved DRUG Abiraterone Aflibercet Albumin Bound Paclitaxel Axitinib CDF INDICATION (EXCLUDING APPROVED CRITERIA ) Metastatic Prostate Cancer Metastatic Colorectal Cancer
National Cancer Drugs Fund List - Approved DRUG Abiraterone Aflibercet Albumin Bound Paclitaxel Axitinib CDF INDICATION (EXCLUDING APPROVED CRITERIA ) Metastatic Prostate Cancer Metastatic Colorectal Cancer
Macmillan Publications
 S1 S2 S3 S3 S3 S4 S5 S6 S7 S8 S8 S9 S10 S11 S11 S12 S13 S14 S15 S17 S18 S19 Bladder Cancer: Non-Invasive, Invasive and Advanced Bone Cancer: Primary, Secondary Colon Cancer, Anal Cancer, Rectal Cancer
S1 S2 S3 S3 S3 S4 S5 S6 S7 S8 S8 S9 S10 S11 S11 S12 S13 S14 S15 S17 S18 S19 Bladder Cancer: Non-Invasive, Invasive and Advanced Bone Cancer: Primary, Secondary Colon Cancer, Anal Cancer, Rectal Cancer
SUPPLEMENTARY FIGURE 1
 SUPPLEMENTARY FIGURE 1 A LN Cell count (1 ) 1 3 1 CD+ 1 1 CDL lo CD hi 1 CD+FoxP3+ 1 1 1 7 3 3 3 % of cells 9 7 7 % of cells CD+ 3 1 % of cells CDL lo CD hi 1 1 % of CD+ cells CD+FoxP3+ 3 1 % of CD+ T
SUPPLEMENTARY FIGURE 1 A LN Cell count (1 ) 1 3 1 CD+ 1 1 CDL lo CD hi 1 CD+FoxP3+ 1 1 1 7 3 3 3 % of cells 9 7 7 % of cells CD+ 3 1 % of cells CDL lo CD hi 1 1 % of CD+ cells CD+FoxP3+ 3 1 % of CD+ T
Rationale for Combining Immunotherapy with Chemotherapy or Targeted Therapy
 Society for Immunotherapy of Cancer (SITC) Rationale for Combining Immunotherapy with Chemotherapy or Targeted Therapy Qing Yi, MD, PhD Staff and Chair, Department of Cancer Biology Betsy B. DeWindt Endowed
Society for Immunotherapy of Cancer (SITC) Rationale for Combining Immunotherapy with Chemotherapy or Targeted Therapy Qing Yi, MD, PhD Staff and Chair, Department of Cancer Biology Betsy B. DeWindt Endowed
Form 2023 R2.0: Ovarian Cancer Pre-HSCT Data
 Key Fields Sequence Number Date Received: - - CIBMTR Center Number: CIBMTR Recipient ID: Today's Date: - - Date of HSCT for which this form is being completed: - - HSCT type: (check all that apply) Autologous
Key Fields Sequence Number Date Received: - - CIBMTR Center Number: CIBMTR Recipient ID: Today's Date: - - Date of HSCT for which this form is being completed: - - HSCT type: (check all that apply) Autologous
Pearson r = P (one-tailed) = n = 9
 8F4-Specific Lysis, % 1 UPN1 UPN3 8 UPN7 6 Pearson r =.69 UPN2 UPN5 P (one-tailed) =.192 4 UPN8 n = 9 2 UPN9 UPN4 UPN6 5 1 15 2 25 8 8F4, % Max MFI Supplementary Figure S1. AML samples UPN1-UPN9 show variable
8F4-Specific Lysis, % 1 UPN1 UPN3 8 UPN7 6 Pearson r =.69 UPN2 UPN5 P (one-tailed) =.192 4 UPN8 n = 9 2 UPN9 UPN4 UPN6 5 1 15 2 25 8 8F4, % Max MFI Supplementary Figure S1. AML samples UPN1-UPN9 show variable
Summary of Research and Writing Activities in Oncology
 Summary of Research and Writing Activities in Oncology Carole Alison Chrvala, PhD 919.545.2149 (Work) 919.951.5230 (Mobile) cchrvala@centurylink.net www.healthmattersmedwriting.com 1 Manuscripts, Posters,
Summary of Research and Writing Activities in Oncology Carole Alison Chrvala, PhD 919.545.2149 (Work) 919.951.5230 (Mobile) cchrvala@centurylink.net www.healthmattersmedwriting.com 1 Manuscripts, Posters,
Jefferies 2017 Global Healthcare Conference. Stephen Doberstein, Ph.D. Senior Vice President & Chief Scientific Officer
 Jefferies 2017 Global Healthcare Conference Stephen Doberstein, Ph.D. Senior Vice President & Chief Scientific Officer June 7, 2017 This presentation includes forward-looking statements regarding Nektar
Jefferies 2017 Global Healthcare Conference Stephen Doberstein, Ph.D. Senior Vice President & Chief Scientific Officer June 7, 2017 This presentation includes forward-looking statements regarding Nektar
NKTR-255: Accessing The Immunotherapeutic Potential Of IL-15 for NK Cell Therapies
 NKTR-255: Accessing The Immunotherapeutic Potential Of IL-15 for NK Cell Therapies Saul Kivimäe Senior Scientist, Research Biology Nektar Therapeutics NK Cell-Based Cancer Immunotherapy, September 26-27,
NKTR-255: Accessing The Immunotherapeutic Potential Of IL-15 for NK Cell Therapies Saul Kivimäe Senior Scientist, Research Biology Nektar Therapeutics NK Cell-Based Cancer Immunotherapy, September 26-27,
Innate Immunity and Inflammation
 SITC Primer on Tumor Immunology and Biological Therapy of Cancer Innate Immunity and Inflammation Willem Overwijk, Ph.D. MD Anderson Cancer Center Center for Cancer Immunology Research Houston, TX www.allthingsbeautiful.com
SITC Primer on Tumor Immunology and Biological Therapy of Cancer Innate Immunity and Inflammation Willem Overwijk, Ph.D. MD Anderson Cancer Center Center for Cancer Immunology Research Houston, TX www.allthingsbeautiful.com
IMMUNOTHERAPY FOR CANCER A NEW HORIZON. Ekaterini Boleti MD, PhD, FRCP Consultant in Medical Oncology Royal Free London NHS Foundation Trust
 IMMUNOTHERAPY FOR CANCER A NEW HORIZON Ekaterini Boleti MD, PhD, FRCP Consultant in Medical Oncology Royal Free London NHS Foundation Trust ASCO Names Advance of the Year: Cancer Immunotherapy No recent
IMMUNOTHERAPY FOR CANCER A NEW HORIZON Ekaterini Boleti MD, PhD, FRCP Consultant in Medical Oncology Royal Free London NHS Foundation Trust ASCO Names Advance of the Year: Cancer Immunotherapy No recent
CANCER 1.7 M 609,000 26% 15.5 M 73% JUST THE FACTS. More Than 1,100 Cancer Treatments in Clinical Testing Offer Hope to Patients
 CANCER MEDICINES IN DEVELOPMENT 2018 REPORT JUST THE FACTS MORE THAN 1.7 M ESTIMATED NEW CASES OF CANCER IN 2018 IN THE UNITED STATES MORE THAN 609,000 U.S. CANCER DEATHS ARE EXPECTED IN 2018 SINCE PEAKING
CANCER MEDICINES IN DEVELOPMENT 2018 REPORT JUST THE FACTS MORE THAN 1.7 M ESTIMATED NEW CASES OF CANCER IN 2018 IN THE UNITED STATES MORE THAN 609,000 U.S. CANCER DEATHS ARE EXPECTED IN 2018 SINCE PEAKING
Supplementary Figure 1. Deletion of Smad3 prevents B16F10 melanoma invasion and metastasis in a mouse s.c. tumor model.
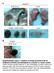 A B16F1 s.c. Lung LN Distant lymph nodes Colon B B16F1 s.c. Supplementary Figure 1. Deletion of Smad3 prevents B16F1 melanoma invasion and metastasis in a mouse s.c. tumor model. Highly invasive growth
A B16F1 s.c. Lung LN Distant lymph nodes Colon B B16F1 s.c. Supplementary Figure 1. Deletion of Smad3 prevents B16F1 melanoma invasion and metastasis in a mouse s.c. tumor model. Highly invasive growth
Cancer Immunotherapy: Active Immunization Approaches
 Cancer Immunotherapy: Active Immunization Approaches Willem W. Overwijk, PhD Department of Melanoma Medical Oncology MD Anderson Cancer Center Houston, TX, USA Disclosures No relevant financial relationships
Cancer Immunotherapy: Active Immunization Approaches Willem W. Overwijk, PhD Department of Melanoma Medical Oncology MD Anderson Cancer Center Houston, TX, USA Disclosures No relevant financial relationships
Engineered Immune Cells for Cancer Therapy : Current Status and Prospects
 When Engineering Meets Immunology Engineered Immune Cells for Cancer Therapy : Current Status and Prospects Yong Taik Lim, Ph.D. Nanomedical Systems Laboratory (http://www.nanomedicalsystems.org) SKKU
When Engineering Meets Immunology Engineered Immune Cells for Cancer Therapy : Current Status and Prospects Yong Taik Lim, Ph.D. Nanomedical Systems Laboratory (http://www.nanomedicalsystems.org) SKKU
and follicular helper T cells is Egr2-dependent. (a) Diagrammatic representation of the
 Supplementary Figure 1. LAG3 + Treg-mediated regulation of germinal center B cells and follicular helper T cells is Egr2-dependent. (a) Diagrammatic representation of the experimental protocol for the
Supplementary Figure 1. LAG3 + Treg-mediated regulation of germinal center B cells and follicular helper T cells is Egr2-dependent. (a) Diagrammatic representation of the experimental protocol for the
Supplementary Figure 1 IL-27 IL
 Tim-3 Supplementary Figure 1 Tc0 49.5 0.6 Tc1 63.5 0.84 Un 49.8 0.16 35.5 0.16 10 4 61.2 5.53 10 3 64.5 5.66 10 2 10 1 10 0 31 2.22 10 0 10 1 10 2 10 3 10 4 IL-10 28.2 1.69 IL-27 Supplementary Figure 1.
Tim-3 Supplementary Figure 1 Tc0 49.5 0.6 Tc1 63.5 0.84 Un 49.8 0.16 35.5 0.16 10 4 61.2 5.53 10 3 64.5 5.66 10 2 10 1 10 0 31 2.22 10 0 10 1 10 2 10 3 10 4 IL-10 28.2 1.69 IL-27 Supplementary Figure 1.
Emerging Tissue and Serum Markers
 Emerging Tissue and Serum Markers for Immune Checkpoint Inhibitors Kyong Hwa Park MD, PhD Medical Oncology Korea University College of Medicine Contents Immune checkpoint inhibitors in clinical practice
Emerging Tissue and Serum Markers for Immune Checkpoint Inhibitors Kyong Hwa Park MD, PhD Medical Oncology Korea University College of Medicine Contents Immune checkpoint inhibitors in clinical practice
NCIC CLINICAL TRIALS GROUP DATA SAFETY MONITORING COMMITTEE Friday, 1 May 2009 SUMMARY REPORT
 NCIC CLINICAL TRIALS GROUP DATA SAFETY MONITORING COMMITTEE Friday, 1 May 2009 SUMMARY REPORT The NCIC CTG DSMC reviewed the following trials with respect to safety, trial conduct, including accrual, and
NCIC CLINICAL TRIALS GROUP DATA SAFETY MONITORING COMMITTEE Friday, 1 May 2009 SUMMARY REPORT The NCIC CTG DSMC reviewed the following trials with respect to safety, trial conduct, including accrual, and
FcγRIIIA (CD16)-expressing monocytes mediate the depletion of tumor-infiltrating Tregs via ipilimumab-dependent ADCC in melanoma patients
 FcγRIIIA (CD16)-expressing monocytes mediate the depletion of tumor-infiltrating Tregs via ipilimumab-dependent ADCC in melanoma patients Emanuela Romano Department of Oncology University of Lausanne and
FcγRIIIA (CD16)-expressing monocytes mediate the depletion of tumor-infiltrating Tregs via ipilimumab-dependent ADCC in melanoma patients Emanuela Romano Department of Oncology University of Lausanne and
Ludwig Presence at 2016 ASCO Annual Meeting
 Ludwig Scientist(s) Affiliation(s) Session and Presentation Time Location Track Session Type/Title Presentation and/or Abstract Title Friday, June 3, 2016 Luisa Lina Villa Ludwig Sao Paulo 1:00pm - 3:15pm
Ludwig Scientist(s) Affiliation(s) Session and Presentation Time Location Track Session Type/Title Presentation and/or Abstract Title Friday, June 3, 2016 Luisa Lina Villa Ludwig Sao Paulo 1:00pm - 3:15pm
Nature Medicine: doi: /nm.3922
 Title: Glucocorticoid-induced tumor necrosis factor receptor-related protein co-stimulation facilitates tumor regression by inducing IL-9-producing helper T cells Authors: Il-Kyu Kim, Byung-Seok Kim, Choong-Hyun
Title: Glucocorticoid-induced tumor necrosis factor receptor-related protein co-stimulation facilitates tumor regression by inducing IL-9-producing helper T cells Authors: Il-Kyu Kim, Byung-Seok Kim, Choong-Hyun
Karolina Palucka, MD, PhD The Jackson Laboratory for Genomic Medicine Farmington, CT
 Humanized mice models of cancer immunotherapy Karolina Palucka, MD, PhD The Jackson Laboratory for Genomic Medicine Farmington, CT Presenter Disclosure Information Karolina Palucka, MD, PhD The following
Humanized mice models of cancer immunotherapy Karolina Palucka, MD, PhD The Jackson Laboratory for Genomic Medicine Farmington, CT Presenter Disclosure Information Karolina Palucka, MD, PhD The following
Converting Novel Therapeutic Models into Early Phase Clinical Trials
 Converting Novel Therapeutic Models into Early Phase Clinical Trials James H. Doroshow, M.D. Deputy Director for Clinical and Translational Research National Cancer Institute, NIH IOM National Cancer Policy
Converting Novel Therapeutic Models into Early Phase Clinical Trials James H. Doroshow, M.D. Deputy Director for Clinical and Translational Research National Cancer Institute, NIH IOM National Cancer Policy
Haematopoietic stem cells
 Haematopoietic stem cells Neil P. Rodrigues, DPhil NIH Centre for Biomedical Research Excellence in Stem Cell Biology Boston University School of Medicine neil.rodrigues@imm.ox.ac.uk Haematopoiesis: An
Haematopoietic stem cells Neil P. Rodrigues, DPhil NIH Centre for Biomedical Research Excellence in Stem Cell Biology Boston University School of Medicine neil.rodrigues@imm.ox.ac.uk Haematopoiesis: An
Dr. Yi-chi M. Kong August 8, 2001 Benjamini. Ch. 19, Pgs Page 1 of 10 TRANSPLANTATION
 Benjamini. Ch. 19, Pgs 379-399 Page 1 of 10 TRANSPLANTATION I. KINDS OF GRAFTS II. RELATIONSHIPS BETWEEN DONOR AND RECIPIENT Benjamini. Ch. 19, Pgs 379-399 Page 2 of 10 II.GRAFT REJECTION IS IMMUNOLOGIC
Benjamini. Ch. 19, Pgs 379-399 Page 1 of 10 TRANSPLANTATION I. KINDS OF GRAFTS II. RELATIONSHIPS BETWEEN DONOR AND RECIPIENT Benjamini. Ch. 19, Pgs 379-399 Page 2 of 10 II.GRAFT REJECTION IS IMMUNOLOGIC
ESMO wishes to thank the following companies for supporting this ESMO Preceptorship Programme
 ESMO wishes to thank the following companies for supporting this ESMO Preceptorship Programme Immunotherapy of cancer Some historical background Rolf Stahel University Hospital Zürich, Switzerland Amsterdam,
ESMO wishes to thank the following companies for supporting this ESMO Preceptorship Programme Immunotherapy of cancer Some historical background Rolf Stahel University Hospital Zürich, Switzerland Amsterdam,
