Min Number of Patients Agreed. Target Date to Recruit Patients Agreed?
|
|
|
- Emil Chapman
- 5 years ago
- Views:
Transcription
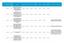
1 NIHR 70 day Performance in Delivery Report Q REC IRAS Name of Trial Target of Patients Min of Patients Max of Patients Target Date to Recruit Patients? Date to Recruit Target of Patients Total of Patients Recruited at the Target Date Date the Trial Closed to Recruitment Total of Study Participants Recruited Reason for Closure of Trial 1 12/LO/ Safety and Performance study of the Edwards SAPIEN 3 Transcatheter Heart Valve 8 8 Date 15/07/ /07/ Recruitment 2 17/EM/ A Parallel Group, Double- Blind, Randomized, Placebo Controlled Phase 3 Trial to Evaluate the Efficacy and Safety of ALD403 Administered Intravenously in Patients with Chronic Migraine 5 5 Date 06/07/ /07/ Recruitment 3 15/NE/ PROSPER: Prospective Real World Outcomes Study of Hepatic Encephalopathy Patients? Experience on Rifaximin-a (TARGAXAN?/XIFAXAN?) 5 5 Date 29/09/ /09/ Recruitment Page 1 of 21
2 550 mg 4 13/LO/ A Randomized, Multicountry, Multicenter, Double-Blind, Parallel, Placebo-Controlled Study of the Effects of Atrasentan on Renal Outcomes in Subjects with Type 2 Diabetes and Nephropathy SONAR: Study Of Diabetic Nephropathy with Atrasentan 6 6 Date 28/02/ /11/ Withdrawn By Sponsor 5 15/LO/ A phase I, randomised, double-blind, placebocontrolled, multi-centre, ascending-dose trial to evaluate the safety, tolerability and immunogenicity of Vaccine FP-02.2 in HBeAgnegative hepatitis B patients as an add-on treatment to entecavir or tenof 5 5 Date 15/08/ /08/ Recruitment 6 15/LO/ A Phase III Double-blind, Randomised, Parallel Group Comparison of the 4 4 Date 31/12/ /12/ Recruitment Page 2 of 21
3 Efficacy and Safety of FP lyo (Recombinant Human Interferon beta- 1a) and Placebo in the Treatment of Patients with Moderate or Severe Acute Respiratory Distress Syndrome 7 16/EE/ A PHASE IB, BLINDED, RANDOMIZED, MULTICENTER, MULTIPLE ASCENDING-DOSE STUDY OF THE SAFETY, TOLERABILITY, PHARMACOKINETICS, AND PHARMACODYNAMICS OF UTTR1147A ADMINISTERED TOPICALLY AND BY SUBCUTANEOUS INJECTION IN PATIENTS WITH NON HEALING NEUROPATHIC 3 5 Date 09/08/ /08/ Recruitment 8 15/LO/ Clinical and device functional assessment of real world ICD patients 6 6 Date 31/10/ /08/ Recruitment 9 16/LO/ PEPTIDE RECEPTOR RADIONUCLIDE THERAPY (PRRT) IN COMBINATION WITH LANREOTIDE 5 5 Date 25/07/ /07/ Recruitment Page 3 of 21
4 AUTOGEL: A RETROSPECTIVE STUDY IN PROGRESSIVE DIGESTIVE AND BRONCHOPULMONARY NEUROENDOCRINE TUMOURS /SW/ A Phase 2, Randomized, Double-Blind, Placebo- Controlled Study to Assess the Efficacy and Tolerability of Entospletinib, a Selective SYK Inhibitor, in Combination with Systemic Corticosteroids as First-Line Therapy in Subjects with Chronic Graft Versu 1 3 Date 28/07/ /07/ Recruitment 11 16/EE/ Efficacy and safety of opicapone in clinical practice in Parkinson?s Disease patients with wearing-off motor fluctuations 4 4 Date 31/12/ /12/ Recruitment 12 15/NW/ A Feasibility Study of the ReGenerCell? Autologous Cell Harvesting Device for Diabetic Foot Ulcers 2 10 Date 21/07/ /07/ Recruitment Page 4 of 21
5 13 16/NW/ Phase 3, Randomized, Open-Label, Active- Controlled Study of ALXN1210 Versus Eculizumab in Complement Inhibitor- Na?ve Adult Patients with Paroxysmal Nocturnal Hemoglobinuria (PNH) 1 2 Date 06/07/ /07/ Recruitment 14 16/SS/ A prospective multi-centre observational cohort study assessing the effectiveness of Cortiment? for the treatment of mild-tomoderate active ulcerative colitis in routine clinical practice 5 8 Date 31/08/ /10/ Recruitment 15 16/NE/ A Randomized, Phase 2a, Open-Label Study to Evaluate the Safety, Pharmacokinetics and Efficacy of the Combination of AL-335, Odalasvir, and Simeprevir in Treatment-Na?ve or Treatment-Experienced Subjects with Genotype 1 and Genotype 3 Chronic Hepatit Date 11/09/ /09/ Recruitment Page 5 of 21
6 16 16/HRA/ Multicenter, Non- Interventional, Retrospective, Matched Cohort Study of Patients Monoinfected with Chronic Hepatitis B and with Moderate or Severe Renal Impairment Treated with Viread? or Baraclude? 5 5 Date 14/08/ /08/ Recruitment 17 14/LO/ A Phase II Proof of Concept (PoC), Double- Blind, Randomised, Placebo-controlled Study to Evaluate the Efficacy, Safety, and Pharmacokinetics of VSN16R for the Treatment of Spasticity in Subjects with Multiple Sclerosis 7 7 Date 28/07/ /07/ Recruitment 18 17/LO/ A Follow Up study to assess Resistance and Durability of Response to Abbvie Direct Acting Antiviral Agent (DAA) Therapy (ABT-493 and/or ABT-530) in Subjects Who Participated in Phase 2 or 3 Clinical Studies for the Treatment of Chronic Date 14/08/ /08/ Recruitment Page 6 of 21
7 Hepatitis C Vi 19 17/NE/ An Open-Label Study to Evaluate the Safety And Efficacy of Sofosbuvir/Velpatasvir/Vo xilaprevir Fixed-Dose Combination for 12 Weeks in Subjects who Participated in a Prior Gilead-Sponsored HCV Treatment Study 1 1 Date 30/09/ /09/ Recruitment 20 13/LO/ Phase III Non-dialysis CKD 4 4 Date 10/07/ /08/ Recruitment 21 16/EE/ Safety and proof of principle study of ATX-GD- 59 in male and female subjects with Graves? disease not currently treated with anti-thyroid therapy: An Open label study, with an upward titration over five dose levels administered by Intradermal injection 1 3 Date 01/06/ /07/ Recruitment Page 7 of 21
8 22 16/LO/ Incidence and outcomes associated with the management of adenovirus infections in allogeneic hematopoietic cell transplant recipients: AdVance 1 5 Date 11/09/ /09/ Recruitment 23 13/NW/ A Phase 3, Randomized, DoubleBlind, PlaceboControlled Study to Compare Efficacy and Safety of Oral Azacitidine Plus Best Supportive Care Versus Best Supportive Care as Maintenance Therapy in Subjects with Acute Myeloid Leukaemia in Complete Remission 5 5 Date 31/12/ /10/ Withdrawn By Sponsor 24 15/LO/ Phase 3 multi-center, outpatient, randomized, double-blind, placebocontrolled study of LY for the prevention of episodic cluster headache 2 2 Date 31/12/ /12/ Withdrawn By Sponsor 25 14/LO/ A Phase III Clinical Trial of Intra-arterial TheraSphere? in the Treatment of Patients with 2 4 Date 31/01/ /08/ Recruitment Page 8 of 21
9 Unresectable Hepatocellular Carcinoma (HCC) 26 15/EM/ A Phase 2, randomised, double-masked, shamcontrolled, multi-centre study to evaluate the efficacy and safety of ocriplasmin in inducing posterior vitreous detachment (PVD) in subjects with nonproliferative diabetic retinopathy (NPDR) (CIRCLE) 1 5 Date 31/12/ /12/ Recruitment 27 15/NW/ A Randomized, Multicenter, Open-Label, Non-Inferiority, Phase 3 Study of ACP-196 Versus Ibrutinib in Previously Treated Subjects with High Risk Chronic Lymphocytic Leukemia 3 5 Date 08/11/ /10/ Withdrawn By Sponsor 28 16/LO/ A Phase 2a, Single-Blind, Randomized, Placebo- Controlled Study Evaluating the Safety, Anti-Viral Activity, and Pharmacokinetics of ARB- 4 4 Date 20/01/ /01/ Withdrawn By Sponsor Page 9 of 21
10 in Non-Cirrhotic, HBeAg-Negative and Positive Subjects with Chronic HBV Infection Receiving Nucleos(t)ide An 29 16/SC/ A RANDOMIZED, MULTICENTER, OPEN- LABEL, PHASE 2 STUDY EVALUATING THE EFFICACY AND SAFETY OF AZACITIDINE SUBCUTANEOUS IN COMBINATION WITH DURVALUMAB (MEDI4736) IN PREVIOUSLY NTREATED SUBJECTS WITH HIGHER- RISK MYELODYSPLASTIC SYNDROMES MDS) OR IN ELDERL 1 3 Date 15/07/ /10/ Recruitment 30 16/LO/ A Phase II Study of Pembrolizumab (MK-3475) as Monotherapy in Subjects with Previously Systemically Treated Advanced Hepatocellular Carcinoma 2 4 Date 06/03/ /02/ Recruitment Page 10 of 21
11 31 17/LO/ Clinical safety & efficacy of a new infant formula with specific medical purpose (FSMP) containing 2 human milk oligosaccharides (HMOs). 2 5 Date 28/02/ /02/ Withdrawn By Sponsor 32 16/LO/ A Phase III Study of Pembrolizumab (MK-3475) vs. Best Supportive Care as Second-Line Therapy in Subjects with Previously Systemically Treated Advanced Hepatocellular Carcinoma (KEYNOTE-240) 2 4 Date 30/09/ /11/ Recruitment 33 17/LO/ A Phase 3b, 12-month, Open-label, Multicenter Study to Evaluate the Efficacy and Safety of BIIB019, Daclizumab, in Subjects with Relapsing- Remitting Multiple Sclerosis (RRMS) Switching from Natalizumab (SUSTAIN) 6 6 Date 31/03/ /03/ Withdrawn By Sponsor 34 16/LO/ A Randomized, Multicenter, Open-Label, Phase 3 Study of Acalabrutinib (ACP-196) Versus Investigator?s 1 3 Date 28/02/ /12/ Recruitment Page 11 of 21
12 Choice of Either Idelalisib Plus Rituximab or Bendamustine Plus Rituximab in Subjects with Relapsed or Refractory Chronic Lymphocytic Leukemia 35 17/LO/ CONSERVE: Coversin Long Term Safety and Efficacy Surveillance Study 2 2 Date 31/12/ /12/ Recruitment 36 17/SC/ A Phase 3, Randomized, Double-Blind Study to Evaluate the Efficacy and Safety of Switching from Tenofovir Disoproxil Fumarate (TDF) 300 mg QD to Tenofovir Alafenamide (TAF) 25mg QD in Subjects with Chronic Hepatitis B who are Virologically Suppressed Date 31/10/ /10/ Recruitment 37 16/LO/ A Phase 2, Double-Blind, Randomized Study Evaluating the Safety, Tolerability, and Efficacy of GS-4997 in Combination with Prednisolone versus Prednisolone Alone in 1 3 Date 30/08/ /12/ Recruitment Page 12 of 21
13 Subjects with Severe Alcoholic Hepatitis (AH) 38 17/WM/ A non-interventional observational clinical study to document the clinical course of patients at 12 months or more following an ischaemic stroke 1 5 Date 31/10/ /11/ Recruitment 39 16/YH/ RA : A MULTICENTER, OPEN- LABEL, UNCONTROLLED, EXTENSION STUDY OF RA IN SUBJECTS WITH PAROXYSMAL NOCTURNAL HEMOGLOBINURIA WHO HAVE COMPLETED A RA CLINICAL STUDY 1 3 Date 15/11/ /11/ Recruitment 40 16/LO/ Non-interventional Study to Investigate the Short and Longterm Real-life Safety, Effectiveness, and Adherence of Velphoro? in Patients with hyperphosphataemia Undergoing Haemodialysis or Peritoneal Dialysis 1 10 Date 30/04/ /03/ Recruitment Page 13 of 21
14 41 12/NE/ An Open-Label, First-in- Human Study of the Safety, Tolerability, and Pharmacokinetics of VX- 970 in Combination with Either Gemcitabine or Cisplatin and Etoposide in Subjects with Advanced Solid Tumors 2 3 Date 11/09/ /09/ Recruitment 42 13/EE/ Revacept, an inhibitor of platelet adhesion in symptomatic carotid stenosis: a phase II, multicentre; randomised, dose-finding, double-blind and placebo-controlled superiority study with parallel groups Date 31/12/ /05/ Recruitment 43 13/LO/ A Phase 3, Randomized, Double-blind, Controlled Study of Cabozantinib (XL184) vs. Placebo in Previously Treated Subjects with Hepatocellular Carcinoma 4 4 Date 01/11/ /08/ Recruitment 44 14/EM/ A Phase 3, Open-Label, Randomized Study of Quizartinib (AC220) Monotherapy vs. Salvage 1 1 Date 01/09/ /09/ Recruitment Page 14 of 21
15 Chemotherapy in Subjects with FLT3-ITD Positive Acute Myeloid Leukemia (AML) Refractory to, or Relapsed after, First-Line Treatment with or without Hematopoietic S 45 14/NE/ A Phase 2b, Randomized, Double-Blind, Placebo- Controlled Multi-Center Study Evaluating Antiviral Effects, Pharmacokinetics, Safety, and Tolerability of GS-5806 in Hematopoietic Cell Transplant (HCT) Recipientswith Respiratory Syncytial Virus (RSV) In 2 4 Date 21/09/ /09/ Recruitment 46 14/WS/ Multicenter, Randomized, Double-blind, Doubledummy, Activecomparator, Event-driven, Superiority Phase III Study of Secondary Prevention of Stroke and Prevention of Systemic Embolism in Patients With a Recent Embolic Stroke of Undetermined Source (E Date 20/09/ /09/ Recruitment Page 15 of 21
16 47 15/SS/ A Multi-national, Multicenter, Prospective, Randomized, Double Blinded, Placebocontrolled Trial to Evaluate the Efficacy of HyperBox Cyclical Topical Wound Oxygen Therapy (TWO2) in the Treatment of Chronic Diabetic Foot Ulcers Date 31/12/ /02/ Recruitment 48 16/EM/ Global study comparing a rivaroxaban-based antithrombotic strategy to an antiplatelet-based strategy after transcatheter aortic valve replacement to Optimize clinical outcomes Date 31/10/ /10/ Recruitment 49 16/LO/ DUOdopa in Patients with Advanced Parkinson's Disease (PD) - A GLobal OBservational Study Evaluating Long-Term Effectiveness (DUOGLOBE) Date 30/12/ /11/ Recruitment 50 16/LO/ Coversin Global Study: An Open-Label, Safety and Efficacy Trial in PNH Patients 1 1 Date 18/09/ /09/ Recruitment Page 16 of 21
17 51 16/LO/ A Phase 3, Multicenter, Randomized, Double Blind Study of Bortezomib and Dexamethasone in Combination with Either Venetoclax or Placebo in Subjects with Relapsed or Refractory Multiple Myeloma Who are Sensitive or Na?ve to Proteasome Inhibitors. 2 4 Date 06/07/ /07/ Recruitment 52 16/SC/ A Phase 2 Randomised Double Blind Placebo controlled study evaluating the Safety, Tolerability and Efficacy of GS-9674 in Subjects with Primary Biliary Cholangitis without Cirrhosis 3 3 Date 30/11/ /03/ Recruitment 53 16/WM/ Phase 3, randomised, open-label, activecontrolled, parallel-group, multi-centre study to evaluate the safety and efficacy of GSK compared to recombinant human erythropoietin (rhepo) in dialysis subjects with anemia associated 4 6 Date 30/04/ /06/ Recruitment Page 17 of 21
18 with chronic kid 54 17/EM/ A Multicenter, Open-Label Study to Evaluate the Safety and Tolerability of Tozadenant as Adjunctive Therapy in Levodopa- Treated Patients with Parkinson?s Disease Experiencing End of Dose?Wearing-Off? 2 2 Date 31/12/ /11/ Withdrawn By Sponsor 55 17/LO/ A Phase 2, Multicenter, Open-Label Study to Evaluate the Efficacy and Safety of Sofosbuvir/Velpatasvir for 12 Weeks in Subjects with Chronic HCV Infection Who are on Dialysis for End Stage Renal Disease 1 3 Date 31/01/ /01/ Recruitment 56 17/LO/ A Double-Blind, Randomised, Placebo- Controlled, Parallel-Group, Phase 2, Dose Ranging Trial to Evaluate the Efficacy, Safety, and Tolerability of Oral Litoxetine 10mg,20mg and 40mg Twice Daily (BID) 2 2 Date 04/06/ /05/ Recruitment Page 18 of 21
19 versus Placebo in Women with Mixed Urinary Incontin 57 17/LO/ A Multicenter, Parallel- Group, Randomized, Cross-Over Trial to Compare the Efficacy of LUMINITY? and SonoVue? in the Evaluation of Left Ventricular Endocardial Border Definition Date 31/05/ /03/ Recruitment 58 17/LO/ A Phase 3, Randomized, Double-Blind, Placebo- Controlled Study Evaluating the Safety and Efficacy of Selonsertib in Subjects with Nonalcoholic Steatohepatitis (NASH) and Bridging (F3) Fibrosis 2 3 Date 19/08/ /04/ Recruitment 59 17/LO/ A Phase 3, Randomized, Double-Blind, Placebo- Controlled Study Evaluating the Safety and Efficacy of Selonsertib in Subjects with Compensated Cirrhosis due to Nonalcoholic Steatohepatitis (NASH) 2 3 Date 19/08/ /01/ Recruitment Page 19 of 21
20 60 17/LO/ A Phase 1b/2a, Dose- Ranging Study of the Safety, Tolerability, Pharmacokinetics, and Initial Efficacy of ABI- H0731 in Patients with Chronic Hepatitis B 5 5 Date 30/06/ /04/ Recruitment 61 17/LO/ An open label, single-arm, Phase I/II study of vandetanib-eluting radiopaque embolic beads (BTG ) in patients with hepatocellular carcinoma (HCC) without curative options. 3 6 Date 31/01/ /05/ Withdrawn By Sponsor 62 17/SC/ A Phase 1/2 Proof-of- Concept Study of the Combination of Acalabrutinib and Vistusertib in Subjects with Relapsed/Refractory B-cell Malignancies 2 4 Date 30/04/ /05/ Withdrawn By Sponsor 63 17/SC/ A Double-blind, Placebocontrolled Crossover Study to Evaluate the Safety, Tolerability, Pharmacokinetics and Effects on Transcranial Magnetic Stimulation of Date 31/05/ /05/ Recruitment Page 20 of 21
21 Oral Administration of XEN1101 in Healthy Male Subjects 64 Registry for Patients with Digital Ulcers Associated with Systemic Sclerosis (DU/SSc) Not Available / Not 16 19/11/ Recruitment Page 21 of 21
The Pennine Acute Hospitals NHS Trust Performance in delivering clinical research Reporting period 1 October 2017 to 30 September 2018
 The Pennine Acute Hospitals NHS Trust Performance in delivering clinical research Reporting period 1 October 2017 to 30 September 2018 REC reference IRAS reference Name of trial Target number of? Minimum
The Pennine Acute Hospitals NHS Trust Performance in delivering clinical research Reporting period 1 October 2017 to 30 September 2018 REC reference IRAS reference Name of trial Target number of? Minimum
The Pennine Acute Hospitals NHS Trust Performance in initiating clinical research Reporting period 1 October 2017 to 30 September 2018
 The Pennine Acute Hospitals NHS Trust Performance in initiating clinical research Reporting period 1 October 2017 to 30 September 2018 REC reference IRAS reference Name of trial First patient recruited?
The Pennine Acute Hospitals NHS Trust Performance in initiating clinical research Reporting period 1 October 2017 to 30 September 2018 REC reference IRAS reference Name of trial First patient recruited?
Perfomance in Delivering (Commercial Trials)
 Perfomance in Delivering (Commercial Trials) REC Ref IRAS ID Name of Trial Target Of? Minimum Of Maximum Of Target Date To Recruit? Total Date Of to recruit target number Recruited At of patients The Target
Perfomance in Delivering (Commercial Trials) REC Ref IRAS ID Name of Trial Target Of? Minimum Of Maximum Of Target Date To Recruit? Total Date Of to recruit target number Recruited At of patients The Target
Barnes, Maisha. Habib, Adil. Kedia, Prashant
 Study Title PI Area of Discipline A Phase 2, Randomized, Double-Blind, Placebo-Controlled, 12-Month, Multiple-Dose Study to Evaluate the Safety, Tolerability and Efficacy of Three Dose Levels of MSDC-
Study Title PI Area of Discipline A Phase 2, Randomized, Double-Blind, Placebo-Controlled, 12-Month, Multiple-Dose Study to Evaluate the Safety, Tolerability and Efficacy of Three Dose Levels of MSDC-
Target date to recruit patients agreed? Target range minimum. Target range maximum
 No REC Reference IRAS No Title Target number of patients agreed? Target range minimum Target range maximum Target date to recruit patients agreed? Planned Recruitment end date Total no of patients recruited
No REC Reference IRAS No Title Target number of patients agreed? Target range minimum Target range maximum Target date to recruit patients agreed? Planned Recruitment end date Total no of patients recruited
Target date to recruit patients agreed? Target range minimum. Target range maximum. Agreed 1 1 Agreed 01/01/ /01/2016 Recruitment Finished
 No REC Reference IRAS No Title Target number of patients agreed? Target range minimum Target range maximum Target date to recruit patients agreed? Planned Recruitment end date No of patients recruited
No REC Reference IRAS No Title Target number of patients agreed? Target range minimum Target range maximum Target date to recruit patients agreed? Planned Recruitment end date No of patients recruited
HRA Approval Date. confirmed by sponsor 07/04/ /04/ /04/ /04/ /04/ /04/ /05/2016
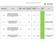
 Reference Reason for delay in recruiting first 1 15/NE/0144 179243 AN OPEN-LABEL EXTENSION AND SAFETY MONITORING STUDY OF PATIENTS WITH MODERATELY TO SEVERELY ACTIVE CROHN S DISEASE PREVIOUSLY ENROLLED
Reference Reason for delay in recruiting first 1 15/NE/0144 179243 AN OPEN-LABEL EXTENSION AND SAFETY MONITORING STUDY OF PATIENTS WITH MODERATELY TO SEVERELY ACTIVE CROHN S DISEASE PREVIOUSLY ENROLLED
Performance in delivering Clinical Research at PHT Q3 14/15 study met the 70 day target study within 70 day target study not met 70 day target
 Performance in delivering Clinical Research at PHT Q3 14/15 study met the 70 day target study within 70 day target study not met 70 day target Research Ethics Committee 14/ES/0004 ALA-BCC-CT008 - A randomized,
Performance in delivering Clinical Research at PHT Q3 14/15 study met the 70 day target study within 70 day target study not met 70 day target Research Ethics Committee 14/ES/0004 ALA-BCC-CT008 - A randomized,
Description of Commitment
 AbbVie U.S. Postmarketing s Table: Name of ANDROGEL (testosterone gel) CREON (pancrelipase) CREON (pancrelipase) / 022309 and 021015 20725 20725 125057/0 125057/114 Description of A randomized, double-blind,
AbbVie U.S. Postmarketing s Table: Name of ANDROGEL (testosterone gel) CREON (pancrelipase) CREON (pancrelipase) / 022309 and 021015 20725 20725 125057/0 125057/114 Description of A randomized, double-blind,
Integrated Research Application System Number. Total Number Of Study Participants Recruited. Research Ethics Committee Reference Number
 Research Ethics Committee Reference Number Integrated Research Application System Number Name of Trial Minimum Number Of Patients Agreed Maximum Number Of Patients Agreed Date That The Trial Closed To
Research Ethics Committee Reference Number Integrated Research Application System Number Name of Trial Minimum Number Of Patients Agreed Maximum Number Of Patients Agreed Date That The Trial Closed To
Performance in Initiating and Delivering Clinical Research Why are we doing this?
 Performance in Initiating and Delivering Clinical Research Why are we doing this? Through the NIHR (National Institute for Health Research) the Government wishes to see a dramatic and sustained improvement
Performance in Initiating and Delivering Clinical Research Why are we doing this? Through the NIHR (National Institute for Health Research) the Government wishes to see a dramatic and sustained improvement
Performance in Initiating Clinical Research Q2 2016/17
 Performance in Initiating Clinical Research Q2 2016/17 Research Ethics Committee Reference Number Integrated Research Application System Number Name of Trial Date of Receipt of Valid Research Application
Performance in Initiating Clinical Research Q2 2016/17 Research Ethics Committee Reference Number Integrated Research Application System Number Name of Trial Date of Receipt of Valid Research Application
Table 1 Salford Royal NHS Foundation Trust: Performance in initiating clinical research (HRA Approval) Reporting period 1 April March 2017
 Table 1 Salford Royal NHS Foundation Trust: Performance in initiating clinical research (HRA Approval) Reporting period 1 April 2016 31 March 2017 REC reference IRAS number Study title 16/NW/0428 207062
Table 1 Salford Royal NHS Foundation Trust: Performance in initiating clinical research (HRA Approval) Reporting period 1 April 2016 31 March 2017 REC reference IRAS number Study title 16/NW/0428 207062
Target date to recruit patients agreed? Target range. maximum. minimum. Date. 5 Agreed 26/01/ /11/2015 Recruitment Finished.
 No REC Reference IRAS No Title Target number of agreed? Target range minimum Target range maximum Target date to recruit agreed? Planned Recruitment end date Total no of recruited at the agreed target
No REC Reference IRAS No Title Target number of agreed? Target range minimum Target range maximum Target date to recruit agreed? Planned Recruitment end date Total no of recruited at the agreed target
Clinical Trial List
 Clinical Trial List 2005 2008 Service Driven. Quality Focused. Global Perspective. Clinical Trial List 2005-2008 1 Pharmacokinetic A Two-Period, Single Dose Pharmacokinetic Study of Three Sustained-Release
Clinical Trial List 2005 2008 Service Driven. Quality Focused. Global Perspective. Clinical Trial List 2005-2008 1 Pharmacokinetic A Two-Period, Single Dose Pharmacokinetic Study of Three Sustained-Release
Curriculum Vitae, Joseph J. Savon, M.D.
 CONTACT INFORMATION: Joseph J. Savon, M.D. Hassman Research Institute, LLC. 175 Cross Keys Road Centennial Center, Bldg. 300 B Berlin, NJ 08009 Site Selection and Information: Bobbie Theodore, Alliance
CONTACT INFORMATION: Joseph J. Savon, M.D. Hassman Research Institute, LLC. 175 Cross Keys Road Centennial Center, Bldg. 300 B Berlin, NJ 08009 Site Selection and Information: Bobbie Theodore, Alliance
Hepatitis B Virus. Taylor Page PharmD Candidate 2019 February 1, 2019
 Hepatitis B Virus Taylor Page PharmD Candidate 2019 February 1, 2019 Epidemiology 3218 cases of acute HBV reported in 2016 847,000 non-institutionalized persons living with chronic HBV in 2011-2012 Viral
Hepatitis B Virus Taylor Page PharmD Candidate 2019 February 1, 2019 Epidemiology 3218 cases of acute HBV reported in 2016 847,000 non-institutionalized persons living with chronic HBV in 2011-2012 Viral
Performance in Initiating Clinical Research
 Performance in Initiating Clinical Research January 2016 December 2016 NHS Permissions Site approval Research Ethics Committee Reference Number Integrated Research Application System Number Name of Trial
Performance in Initiating Clinical Research January 2016 December 2016 NHS Permissions Site approval Research Ethics Committee Reference Number Integrated Research Application System Number Name of Trial
Performance in Delivering Q4. Target number of patients
 Performance in Delivering Q4 Research Ethics Committee Reference Number Name of Trial Target number of patients Date Agreed to recruit target number of patients Trial Status Target met within the agreed
Performance in Delivering Q4 Research Ethics Committee Reference Number Name of Trial Target number of patients Date Agreed to recruit target number of patients Trial Status Target met within the agreed
Gilead to Present Wide-Ranging New Data on Treatment and Diagnosis of Liver Diseases at The Liver Meeting 2018
 Gilead to Present Wide-Ranging New Data on Treatment and Diagnosis of Liver Diseases at The Liver Meeting 2018 October 11, 2018 3:24 PM ET -- More Than 50 Abstracts Across NASH, PSC, HBV and HCV Reflect
Gilead to Present Wide-Ranging New Data on Treatment and Diagnosis of Liver Diseases at The Liver Meeting 2018 October 11, 2018 3:24 PM ET -- More Than 50 Abstracts Across NASH, PSC, HBV and HCV Reflect
Table 1: Salford Royal NHS Foundation Trust: Performance in initiating clinical research: Reporting period 1 July June 2013
 Table 1: Salford Royal NHS Foundation Trust: Performance in initiating clinical research: Reporting period 1 July 2012-30 June 2013 Name of trial Note, some study titles are shortened 1 Long-Term Study
Table 1: Salford Royal NHS Foundation Trust: Performance in initiating clinical research: Reporting period 1 July 2012-30 June 2013 Name of trial Note, some study titles are shortened 1 Long-Term Study
Min number of patients agreed to be recruited. Max number of patients agreed to be recruited
 be 12/SC/0139 98125 A PHASE III PROSPECTIVE, TWO-COHORT NON-RANDOMIZED, MULTI- CENTRE, MULTINATIONAL, OPEN LABEL STUDY TO ASSESS THE SAFETY OF ASSISTED- AND SELF-ADMINISTERED SUBCUTANEOUS TRASTUZUMAB AS
be 12/SC/0139 98125 A PHASE III PROSPECTIVE, TWO-COHORT NON-RANDOMIZED, MULTI- CENTRE, MULTINATIONAL, OPEN LABEL STUDY TO ASSESS THE SAFETY OF ASSISTED- AND SELF-ADMINISTERED SUBCUTANEOUS TRASTUZUMAB AS
Studies proceeding under pre HRA-Approval system (NHS Permission)
 15/NW/0004 162679 Studies proceeding under pre HRA-roval system (NHS Permission) A MULTI-CENTRE RANDOMISED CLINICAL TRIAL OF BIOMARKER-DRIVEN MAINTENANCE TREATMENT FOR FIRST- LINE METASTATIC COLORECTAL
15/NW/0004 162679 Studies proceeding under pre HRA-roval system (NHS Permission) A MULTI-CENTRE RANDOMISED CLINICAL TRIAL OF BIOMARKER-DRIVEN MAINTENANCE TREATMENT FOR FIRST- LINE METASTATIC COLORECTAL
Table 1: Salford Royal NHS Foundation Trust: Performance in initiating clinical research: Reporting period 1 October September 2013
 Table 1: Salford Royal NHS Foundation Trust: Performance in initiating clinical research: Reporting period 1 October 2012-30 September 2013 Research Ethics Committee reference number Name of Trial 1 12/NW/0321
Table 1: Salford Royal NHS Foundation Trust: Performance in initiating clinical research: Reporting period 1 October 2012-30 September 2013 Research Ethics Committee reference number Name of Trial 1 12/NW/0321
Performance in Initiating Clinical Research Q4 2015/16
 Performance in Initiating Clinical Research Q4 2015/16 Research Ethics Committee Reference Number Integrated Research Application System Number Name of Trial Date of Receipt of Valid Research Application
Performance in Initiating Clinical Research Q4 2015/16 Research Ethics Committee Reference Number Integrated Research Application System Number Name of Trial Date of Receipt of Valid Research Application
Table 1: Salford Royal NHS Foundation Trust: Performance in initiating clinical research: Reporting period 1 April March 2014
 Table 1: Salford Royal NHS Foundation Trust: Performance in initiating clinical research: Reporting period 1 April 2013-31 March 2014 Research Ethics Committee Name of Trial reference number 1 08/H0906/140
Table 1: Salford Royal NHS Foundation Trust: Performance in initiating clinical research: Reporting period 1 April 2013-31 March 2014 Research Ethics Committee Name of Trial reference number 1 08/H0906/140
HMO: Medical (provider setting); Rx (out patient) PPO/CDHP: Rx
 BENEFIT DESCRIPTION AND LIMITATIONS OF COVERAGE ITEM: PRODUCT LINES: COVERED UNDER: DESCRIPTION: CPT/HCPCS Code: Company Supplying: Setting: Epogen, Procrit (epoetin alfa, injection) Commercial HMO/PPO/CDHP
BENEFIT DESCRIPTION AND LIMITATIONS OF COVERAGE ITEM: PRODUCT LINES: COVERED UNDER: DESCRIPTION: CPT/HCPCS Code: Company Supplying: Setting: Epogen, Procrit (epoetin alfa, injection) Commercial HMO/PPO/CDHP
NICE TECHNOLOGY APPRAISAL MEDICINES REPORT 2017
 NICE TECHNOLOGY PPRISL MEDICINES REPORT St George s Healthcare NHS Trust key for medicine-related NICE Technology ppraisals. has been approved by the Drugs and Therapeutics Committee as recommended within
NICE TECHNOLOGY PPRISL MEDICINES REPORT St George s Healthcare NHS Trust key for medicine-related NICE Technology ppraisals. has been approved by the Drugs and Therapeutics Committee as recommended within
Directorate General of Health Services Office of Drugs Controller General (India) (Biological Division)
 Directorate General of Health Services Office of Drugs Controller General (India) (Biological Division) List of r-dna drug products approved in the country (Form 45 and 45A) from 1 st Jan 2015 to 30 th
Directorate General of Health Services Office of Drugs Controller General (India) (Biological Division) List of r-dna drug products approved in the country (Form 45 and 45A) from 1 st Jan 2015 to 30 th
Intron A Hepatitis B. Intron A (interferon alfa-2b) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.01.01 Subject: Intron A Hepatitis B Page: 1 of 7 Last Review Date: November 30, 2018 Intron A Hepatitis
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.01.01 Subject: Intron A Hepatitis B Page: 1 of 7 Last Review Date: November 30, 2018 Intron A Hepatitis
The Impact of HBV Therapy on Fibrosis and Cirrhosis
 The Impact of HBV Therapy on Fibrosis and Cirrhosis Jordan J. Feld, MD, MPH Associate Professor of Medicine University of Toronto Hepatologist Toronto Centre for Liver Disease Sandra Rotman Centre for
The Impact of HBV Therapy on Fibrosis and Cirrhosis Jordan J. Feld, MD, MPH Associate Professor of Medicine University of Toronto Hepatologist Toronto Centre for Liver Disease Sandra Rotman Centre for
NIHR Performance Metrics - Initiating Clinical Research. Quarter 4 ( )
 NIHR Performance Metrics - Initiating Clinical Research Quarter 4 (2013-2014) Research Ethics Committee Reference Number Name of Trial Date of Receipt of Valid Research Application Date of First Patient
NIHR Performance Metrics - Initiating Clinical Research Quarter 4 (2013-2014) Research Ethics Committee Reference Number Name of Trial Date of Receipt of Valid Research Application Date of First Patient
Open and Pending Trials Listing For Arizona Oncology Associates P.C. HOPE Division/ Tucson, Arizona
 Open and Pending Trials Listing For Arizona Oncology Associates P.C. HOPE Division/ Tucson, Arizona Solid Tumor: USON 15180 / A Multicenter, Open-Label Phase 1b/2 Trial of Lenvatinib (E7080) Plus Pembrolizumab
Open and Pending Trials Listing For Arizona Oncology Associates P.C. HOPE Division/ Tucson, Arizona Solid Tumor: USON 15180 / A Multicenter, Open-Label Phase 1b/2 Trial of Lenvatinib (E7080) Plus Pembrolizumab
Hepatitis B: Future treatment developments
 Hepatitis B: Future treatment developments VIII International Update Workshop in Hepatology Curitiba, 27.08.2016 Christoph Sarrazin St. Josefs-Hospital Wiesbaden and Goethe-University, Frankfurt am Main
Hepatitis B: Future treatment developments VIII International Update Workshop in Hepatology Curitiba, 27.08.2016 Christoph Sarrazin St. Josefs-Hospital Wiesbaden and Goethe-University, Frankfurt am Main
Inarigivir ACHIEVE Trial Results and HBV Clinical Program Update. August 2, 2018
 Inarigivir ACHIEVE Trial Results and HBV Clinical Program Update August 2, 2018 FORWARD LOOKING STATEMENT This presentation includes forward-looking statements within the meaning of the Private Securities
Inarigivir ACHIEVE Trial Results and HBV Clinical Program Update August 2, 2018 FORWARD LOOKING STATEMENT This presentation includes forward-looking statements within the meaning of the Private Securities
Hepatitis B Virus therapy. Maria Buti Hospital Universitario Valle Hebron Barcelona Spain
 Hepatitis B Virus therapy Maria Buti Hospital Universitario Valle Hebron Barcelona Spain Disclosures Advisor: AbbVie, Boehringer Ingelheim, Bristol-Myers Squibb, Gilead Sciences, Janssen, Merck Sharp &
Hepatitis B Virus therapy Maria Buti Hospital Universitario Valle Hebron Barcelona Spain Disclosures Advisor: AbbVie, Boehringer Ingelheim, Bristol-Myers Squibb, Gilead Sciences, Janssen, Merck Sharp &
Swiss Summary of the Risk Management Plan (RMP) for. Vosevi, film-coated tablets
 Swiss Summary of the Management Plan (RMP) for, film-coated tablets (Sofosbuvir/Velpatasvir/Voxilaprevir Fixed-Dose Combination) 400mg/100mg/100mg Version 1.0 (February 2018) final Based on EU RMP version
Swiss Summary of the Management Plan (RMP) for, film-coated tablets (Sofosbuvir/Velpatasvir/Voxilaprevir Fixed-Dose Combination) 400mg/100mg/100mg Version 1.0 (February 2018) final Based on EU RMP version
Date of NHS Permission. Date of First Patient Recruited. Date Site Invited
 Reference of Receipt of of NHS of First 14/WA/1075 150715 NHS HeadPoST Version 2.1 23/11/2015 05/01/2016 11/01/2016 N/A 12/YH/0452 106985 NHS INCA 10/12/2015 15/01/2016 17/03/2016 N/A Excess Treatment
Reference of Receipt of of NHS of First 14/WA/1075 150715 NHS HeadPoST Version 2.1 23/11/2015 05/01/2016 11/01/2016 N/A 12/YH/0452 106985 NHS INCA 10/12/2015 15/01/2016 17/03/2016 N/A Excess Treatment
Hepatitis B Virus therapy. Maria Buti Hospital Universitario Valle Hebron Barcelona Spain
 Hepatitis B Virus therapy Maria Buti Hospital Universitario Valle Hebron Barcelona Spain Disclosures Advisor: AbbVie, Boehringer Ingelheim, Bristol-Myers Squibb, Gilead Sciences, Janssen, Merck Sharp &
Hepatitis B Virus therapy Maria Buti Hospital Universitario Valle Hebron Barcelona Spain Disclosures Advisor: AbbVie, Boehringer Ingelheim, Bristol-Myers Squibb, Gilead Sciences, Janssen, Merck Sharp &
Summary of Strategic Competitive Analysis and Publication Planning
 Summary of Strategic Competitive Analysis and Publication Planning Carole Alison Chrvala, PhD 919.545.2149 (Work) 919.951.5230 (Mobile) cchrvala@centurylink.net www.healthmattersmedwriting.com 1 Briefing
Summary of Strategic Competitive Analysis and Publication Planning Carole Alison Chrvala, PhD 919.545.2149 (Work) 919.951.5230 (Mobile) cchrvala@centurylink.net www.healthmattersmedwriting.com 1 Briefing
Performance in Initiating and Delivering Clinical Research
 Performance in Initiating and Delivering Clinical Research As a global leading centre for research in Eyes and Vision, it is a key priority to offer as many patients as possible the opportunity to get
Performance in Initiating and Delivering Clinical Research As a global leading centre for research in Eyes and Vision, it is a key priority to offer as many patients as possible the opportunity to get
entecavir, 0.5mg and 1mg film-coated tablets and 0.05 mg/ml oral solution, Baraclude SMC No. (747/11) Bristol-Myers Squibb Pharmaceuticals Ltd
 entecavir, 0.5mg and 1mg film-coated tablets and 0.05 mg/ml oral solution, Baraclude SMC No. (747/11) Bristol-Myers Squibb Pharmaceuticals Ltd 09 December 2011 The Scottish Medicines Consortium (SMC) has
entecavir, 0.5mg and 1mg film-coated tablets and 0.05 mg/ml oral solution, Baraclude SMC No. (747/11) Bristol-Myers Squibb Pharmaceuticals Ltd 09 December 2011 The Scottish Medicines Consortium (SMC) has
Future strategies with new DAAs
 Future strategies with new DAAs Ola Weiland professor New direct antiviral drugs Case no 1 male with genotype 2b Male with gt 2b chronic HCV Male with gt 2b relapse afer peg-ifn + RBV during 24 weeks
Future strategies with new DAAs Ola Weiland professor New direct antiviral drugs Case no 1 male with genotype 2b Male with gt 2b chronic HCV Male with gt 2b relapse afer peg-ifn + RBV during 24 weeks
New York State HCV Provider Webinar Series. Side Effects of Therapy
 New York State HCV Provider Webinar Series Side Effects of Therapy Objectives Understand the basics of HCV therapy Review the currently available regimens for treatment of HCV Appreciate side effects related
New York State HCV Provider Webinar Series Side Effects of Therapy Objectives Understand the basics of HCV therapy Review the currently available regimens for treatment of HCV Appreciate side effects related
NIHR Performance Metrics - Initiating Clinical Research. Quarter 1 ( )
 NIHR Performance Metrics - Initiating Clinical Research Quarter 1 (2014-2015) Research Ethics Committee Reference Number Name of Trial Date of Receipt of Valid Research Application Date of First Patient
NIHR Performance Metrics - Initiating Clinical Research Quarter 1 (2014-2015) Research Ethics Committee Reference Number Name of Trial Date of Receipt of Valid Research Application Date of First Patient
Date of HRA. Date Site Invited. Date Site Selected. Approval. Recruited. Date
 14/LO/0259 134883 RE-AKT: A randomised Phase II study of enzalutamide (MDV3100) in combination with AZD5363 in s with Metastatic Castration - Resistant Prostate Cancer Yes 08/01/2018 01/07/2016 01/07/2017
14/LO/0259 134883 RE-AKT: A randomised Phase II study of enzalutamide (MDV3100) in combination with AZD5363 in s with Metastatic Castration - Resistant Prostate Cancer Yes 08/01/2018 01/07/2016 01/07/2017
Table 2: Salford Royal NHS Foundation Trust: Performance in delivering clinical research: Reporting period 1 January December 2014
 Table 2: Salford Royal HS Foundation Trust: Performance in delivering clinical research: Reporting period 1 January 2014 1 December 2014 Research Ethics Committee reference number 1 12/W/0321 2 11/LO/1986
Table 2: Salford Royal HS Foundation Trust: Performance in delivering clinical research: Reporting period 1 January 2014 1 December 2014 Research Ethics Committee reference number 1 12/W/0321 2 11/LO/1986
Tenofovir as a drug of choice for the chronic hepatitis B treatment
 EASL endorsed conference White Nights of Hepatology 2013 Symposium Perspectives of chronic viral hepatitis B and C treatment June 6-7 Saint-Petersburg Tenofovir as a drug of choice for the chronic hepatitis
EASL endorsed conference White Nights of Hepatology 2013 Symposium Perspectives of chronic viral hepatitis B and C treatment June 6-7 Saint-Petersburg Tenofovir as a drug of choice for the chronic hepatitis
Hepatitis B Prior Authorization Policy
 Hepatitis B Prior Authorization Policy Line of Business: Medi-Cal P&T Approval Date: November 15, 2017 Effective Date: January 1, 2018 This policy has been developed through review of medical literature,
Hepatitis B Prior Authorization Policy Line of Business: Medi-Cal P&T Approval Date: November 15, 2017 Effective Date: January 1, 2018 This policy has been developed through review of medical literature,
BC Cancer Protocol Summary for Therapy of Acute Myeloid Leukemia Using azacitidine and SORAfenib
 BC Cancer Protocol Summary for Therapy of Acute Myeloid Leukemia Using azacitidine and SORAfenib Protocol Code Tumour Group Contact Physician ULKAMLAS Leukemia/BMT Dr. Donna Hogge ELIGIBILITY: Acute myeloid
BC Cancer Protocol Summary for Therapy of Acute Myeloid Leukemia Using azacitidine and SORAfenib Protocol Code Tumour Group Contact Physician ULKAMLAS Leukemia/BMT Dr. Donna Hogge ELIGIBILITY: Acute myeloid
Performance in Initiating and Delivering Clinical Research
 Performance in Initiating and Delivering Clinical Research As a global leading centre for research in Eyes and Vision, it is a key priority to offer as many patients as possible the opportunity to get
Performance in Initiating and Delivering Clinical Research As a global leading centre for research in Eyes and Vision, it is a key priority to offer as many patients as possible the opportunity to get
HBV Therapy in Special Populations: Liver Cirrhosis
 HBV Therapy in Special Populations: Liver Cirrhosis Universitätsklinikum Leipzig Thomas Berg Sektion Hepatologie Klinik und Poliklinik für Gastroenterologie und Rheumatologie Leber- und Studienzentrum
HBV Therapy in Special Populations: Liver Cirrhosis Universitätsklinikum Leipzig Thomas Berg Sektion Hepatologie Klinik und Poliklinik für Gastroenterologie und Rheumatologie Leber- und Studienzentrum
Clinical Research Institute HUCH Sponsored, fully signed Clinical Trial Agreements (except grants)
 , fully signed Clinical Trial Agreements (except grants) Sponsor Full name of the Hospital clinic where the is details of the January x x January x NightstaRx Ltd A Randomised, Open Label, Outcomes-Assessor
, fully signed Clinical Trial Agreements (except grants) Sponsor Full name of the Hospital clinic where the is details of the January x x January x NightstaRx Ltd A Randomised, Open Label, Outcomes-Assessor
Update on Real-World Experience With HARVONI
 Update on Real-World Experience With A RESOURCE FOR PAYERS MAY 217 This information is intended for payers only. The HCV-TARGET study was supported by Gilead Sciences, Inc. Real-world experience data were
Update on Real-World Experience With A RESOURCE FOR PAYERS MAY 217 This information is intended for payers only. The HCV-TARGET study was supported by Gilead Sciences, Inc. Real-world experience data were
April May For adults for the treatment of visual impairment due to macular oedema secondary to central retinal vein occlusion.
 April May 2014 Recommended for use within NHS Scotland April May 2014 aflibercept intravitreal (Eylea ) 954/14 For adults for the treatment of visual impairment due to macular oedema secondary to central
April May 2014 Recommended for use within NHS Scotland April May 2014 aflibercept intravitreal (Eylea ) 954/14 For adults for the treatment of visual impairment due to macular oedema secondary to central
Update on Real-World Experience With HARVONI
 Update on Real-World Experience With A RESOURCE FOR PAYERS This information is intended for payers only. The HCV-TARGET and TRIO studies were supported by Gilead Sciences, Inc. Real-world experience data
Update on Real-World Experience With A RESOURCE FOR PAYERS This information is intended for payers only. The HCV-TARGET and TRIO studies were supported by Gilead Sciences, Inc. Real-world experience data
Supplementary Digital Content
 Geissler et al: Sirolimus and Hepatocellular Carcinoma in Liver Transplantation Page 1 of 10 Supplementary Digital Content Supplementary Table 1. Surgical procedures used Total Transplant technique Piggy
Geissler et al: Sirolimus and Hepatocellular Carcinoma in Liver Transplantation Page 1 of 10 Supplementary Digital Content Supplementary Table 1. Surgical procedures used Total Transplant technique Piggy
Scottish Medicines Consortium
 Scottish Medicines Consortium tenofovir disoproxil (as fumarate), 245 mg film-coated tablet (Viread ) No. (479/08) Gilead Sciences 06 June 2008 The Scottish Medicines Consortium has completed its assessment
Scottish Medicines Consortium tenofovir disoproxil (as fumarate), 245 mg film-coated tablet (Viread ) No. (479/08) Gilead Sciences 06 June 2008 The Scottish Medicines Consortium has completed its assessment
Commissioning policies agreed by PCTs in Yorkshire and the Humber at Board meeting of YH SCG on December
 Commissioning policies agreed by PCTs in Yorkshire and the Humber at Board meeting of YH SCG on December 17 2010. 32/10 Imatinib for gastrointestinal stromal tumours (unresectable/metastatic) (update on
Commissioning policies agreed by PCTs in Yorkshire and the Humber at Board meeting of YH SCG on December 17 2010. 32/10 Imatinib for gastrointestinal stromal tumours (unresectable/metastatic) (update on
HRA Approval Date Site Date Site Sponsor. Non- Confirmation Status. Date Site Ready To Start
 Research Ethics Committee Reference Number Integrated Research Application System Number Submission Type Name of Trial Date of Receipt of Date of Valid Research Application First Patient Recruited? Date
Research Ethics Committee Reference Number Integrated Research Application System Number Submission Type Name of Trial Date of Receipt of Date of Valid Research Application First Patient Recruited? Date
Hepatocellular Carcinoma: Can We Slow the Rising Incidence?
 Hepatocellular Carcinoma: Can We Slow the Rising Incidence? K.Rajender Reddy M.D. Professor of Medicine Director of Hepatology Medical Director of Liver Transplantation University of Pennsylvania Outline
Hepatocellular Carcinoma: Can We Slow the Rising Incidence? K.Rajender Reddy M.D. Professor of Medicine Director of Hepatology Medical Director of Liver Transplantation University of Pennsylvania Outline
Novedades en el tratamiento de la hepatitis B: noticias desde la EASL. Maria Buti Hospital Universitario Valle Hebrón Barcelona
 Novedades en el tratamiento de la hepatitis B: noticias desde la EASL Maria Buti Hospital Universitario Valle Hebrón Barcelona Milestones in CHB treatment Conventional IFN 1991 Lamivudine (LAM) 1998 Adefovir
Novedades en el tratamiento de la hepatitis B: noticias desde la EASL Maria Buti Hospital Universitario Valle Hebrón Barcelona Milestones in CHB treatment Conventional IFN 1991 Lamivudine (LAM) 1998 Adefovir
PART VI: SUMMARY OF THE RISK MANAGEMENT PLAN BY PRODUCT
 PAGE 175 PART VI: SUMMARY OF THE RISK MANAGEMENT PLAN BY PRODUCT Summary of risk management plan for pembrolizumab This is a summary of the risk management plan (RMP) for pembrolizumab. The RMP details
PAGE 175 PART VI: SUMMARY OF THE RISK MANAGEMENT PLAN BY PRODUCT Summary of risk management plan for pembrolizumab This is a summary of the risk management plan (RMP) for pembrolizumab. The RMP details
Pegylated Interferons and Ribavirins
 Pegylated Interferons and Ribavirins Goal(s): Cover drugs only for those clients where there is evidence of effectiveness and safety Length of Authorization: 16 weeks plus 12-36 additional weeks or 12
Pegylated Interferons and Ribavirins Goal(s): Cover drugs only for those clients where there is evidence of effectiveness and safety Length of Authorization: 16 weeks plus 12-36 additional weeks or 12
The HCV pipeline: Will IFN-free treatment be possible? Heiner Wedemeyer. Hannover Medical School Germany
 : Will IFN-free treatment be possible? Heiner Wedemeyer Hannover Medical School Germany Interferon-free regimens to treat hepatitis C What should be the goal of interferon-free treatment regimens: Sustained
: Will IFN-free treatment be possible? Heiner Wedemeyer Hannover Medical School Germany Interferon-free regimens to treat hepatitis C What should be the goal of interferon-free treatment regimens: Sustained
Why do we need new HBV treatment and what is our definition for cure?
 Why do we need new HBV treatment and what is our definition for cure? Harry L.A. Janssen Francis Family Chair of Hepatology Director Toronto Centre for Liver Disease University Health Network University
Why do we need new HBV treatment and what is our definition for cure? Harry L.A. Janssen Francis Family Chair of Hepatology Director Toronto Centre for Liver Disease University Health Network University
Hepatitis B Treatment Pearls. Agenda
 Hepatitis B Treatment Pearls Fredric D. Gordon, MD Vice Chair Dept. of Transplantation and Hepatobiliary Diseases Lahey Hospital & Medical Center Associate Professor of Medicine Tufts Medical School Boston,
Hepatitis B Treatment Pearls Fredric D. Gordon, MD Vice Chair Dept. of Transplantation and Hepatobiliary Diseases Lahey Hospital & Medical Center Associate Professor of Medicine Tufts Medical School Boston,
Drug Class Monograph
 Drug Class Monograph Class: Chronic Hepatitis B Drug: Baraclude (entecavir), Epivir (lamivudine), Hepsera (adefovir), Intron A (interferon alfa- 2b), Pegasys (peginterferon alfa-2a), Tyzeka (telbivudine),
Drug Class Monograph Class: Chronic Hepatitis B Drug: Baraclude (entecavir), Epivir (lamivudine), Hepsera (adefovir), Intron A (interferon alfa- 2b), Pegasys (peginterferon alfa-2a), Tyzeka (telbivudine),
Viral Hepatitis The Preventive Potential of Antiviral Therapy. Thomas Berg
 Viral Hepatitis The Preventive Potential of Antiviral Therapy Thomas Berg Therapeutic and preventive strategies in patients with hepatitis virus infection Treatment of acute infection Treatment of chronic
Viral Hepatitis The Preventive Potential of Antiviral Therapy Thomas Berg Therapeutic and preventive strategies in patients with hepatitis virus infection Treatment of acute infection Treatment of chronic
OPEN TRIALS Accruals counted until 30-April Current Accrual
 Disease group Breast OPEN TRIALS Trial Title Accrual Target IBCSG 48-14 POSITIVE IBCSG 50-14 OLYMPIA IBCSG 52-15 PALLAS SAKK 21/12 A study evaluating the pregnancy outcomes and safety of interrupting endocrine
Disease group Breast OPEN TRIALS Trial Title Accrual Target IBCSG 48-14 POSITIVE IBCSG 50-14 OLYMPIA IBCSG 52-15 PALLAS SAKK 21/12 A study evaluating the pregnancy outcomes and safety of interrupting endocrine
UNIVERSITY HOSPITAL OF HERAKLION DEPARTMENT OF HAEMATOLOGY
 UNIVERSITY HOSPITAL OF HERAKLION DEPARTMENT OF HAEMATOLOGY HEAD: Helen Papadaki, Professor of Haematology PO Box 1352, Heraklion, Crete, Greece Email: e.papadaki@uoc.gr Tel/Fax: 0030 2810 392805, http://haematology.med.uoc.gr
UNIVERSITY HOSPITAL OF HERAKLION DEPARTMENT OF HAEMATOLOGY HEAD: Helen Papadaki, Professor of Haematology PO Box 1352, Heraklion, Crete, Greece Email: e.papadaki@uoc.gr Tel/Fax: 0030 2810 392805, http://haematology.med.uoc.gr
Curriculum Vitae, Nirav S. Patel, M.D. Nirav S. Patel, M.D. Collaborative Neuroscience Network, LLC.
 CONTACT INFORMATION: Site Selection and Information: Bobbie Theodore, Alliance Director Tel. (916) 939-6696 Fax (208) 575-3169 Email: clinicaltrials@alliancesites.com AFFILIATIONS: Collaborative Neuroscience
CONTACT INFORMATION: Site Selection and Information: Bobbie Theodore, Alliance Director Tel. (916) 939-6696 Fax (208) 575-3169 Email: clinicaltrials@alliancesites.com AFFILIATIONS: Collaborative Neuroscience
Management of HBV in KidneyTransplanted Patients Dr.E.Nemati
 Management of HBV in KidneyTransplanted Patients Dr.E.Nemati Hepatitis B virus (HBV) infection Hepatitis B virus (HBV) infection confers a significantly negative impact on the clinical outcomes of kidney
Management of HBV in KidneyTransplanted Patients Dr.E.Nemati Hepatitis B virus (HBV) infection Hepatitis B virus (HBV) infection confers a significantly negative impact on the clinical outcomes of kidney
HBV Diagnosis and Treatment
 HBV Diagnosis and Treatment Anna S. F. Lok, MD Alice Lohrman Andrews Professor in Hepatology Director of Clinical Hepatology Assistant Dean for Clinical Research University of Michigan Ann Arbor, MI, USA
HBV Diagnosis and Treatment Anna S. F. Lok, MD Alice Lohrman Andrews Professor in Hepatology Director of Clinical Hepatology Assistant Dean for Clinical Research University of Michigan Ann Arbor, MI, USA
Hepatocellular Carcinoma HCC Updated November 2015 by: Dr. Mohammed Alghamdi (Medical Oncology Fellow, University of Calgary)
 Hepatocellular Carcinoma HCC Updated November 2015 by: Dr. Mohammed Alghamdi (Medical Oncology Fellow, University of Calgary) Staff Reviewers: Dr. Yoo Joung Ko (Medical Oncologist, Sunnybrook Odette Cancer
Hepatocellular Carcinoma HCC Updated November 2015 by: Dr. Mohammed Alghamdi (Medical Oncology Fellow, University of Calgary) Staff Reviewers: Dr. Yoo Joung Ko (Medical Oncologist, Sunnybrook Odette Cancer
HRA Approval date. Date site was confirmed by Sponsor. Date site was confimred
 Sourceof the delay in recruiting 1 16/YH/0154 202759 A Multicenter, Double-blind, Randomized, Placebo-controlled, Parallel-group, Prospective Study to Evaluate the Safety and Efficacy of Domperidone in
Sourceof the delay in recruiting 1 16/YH/0154 202759 A Multicenter, Double-blind, Randomized, Placebo-controlled, Parallel-group, Prospective Study to Evaluate the Safety and Efficacy of Domperidone in
Inarigivir: A novel RIG-I agonist for chronic hepatitis B
 : A novel RIG-I agonist for chronic hepatitis B Stephen Locarnini, Danny Wong, Kathy Jackson, Renae Walsh, Ros Edwards, Rachel Hammond, Carla S. Coffin, Magdy Elkhashab, Susan Greenbloom, Alnoor Ramji,
: A novel RIG-I agonist for chronic hepatitis B Stephen Locarnini, Danny Wong, Kathy Jackson, Renae Walsh, Ros Edwards, Rachel Hammond, Carla S. Coffin, Magdy Elkhashab, Susan Greenbloom, Alnoor Ramji,
tenofovir disoproxil (as fumarate), 245mg, film-coated tablet (Viread ) SMC No. (720/11) Gilead Sciences Ltd
 tenofovir disoproxil (as fumarate), 245mg, film-coated tablet (Viread ) SMC No. (720/11) Gilead Sciences Ltd 05 August 2011 The Scottish Medicines Consortium (SMC) has completed its assessment of the above
tenofovir disoproxil (as fumarate), 245mg, film-coated tablet (Viread ) SMC No. (720/11) Gilead Sciences Ltd 05 August 2011 The Scottish Medicines Consortium (SMC) has completed its assessment of the above
Summary of late stage clinical trials. As of Jul. 22, 2016
 Summary of late stage clinical trials As of Jul. 22, 2016 List of abbreviations AE BID DLT GFR iv MTD ORR OS PD PFS PK po PPK Q2W Q3W Q4W QD QW sc TID Adverse Events Twice daily Dose Limiting Toxicity
Summary of late stage clinical trials As of Jul. 22, 2016 List of abbreviations AE BID DLT GFR iv MTD ORR OS PD PFS PK po PPK Q2W Q3W Q4W QD QW sc TID Adverse Events Twice daily Dose Limiting Toxicity
Hepatitis B and Hepatitis C Virus in non-liver Transplant Recipients. Karim Qumosani MD, FRCPC, ABIM, MdMEd Multi-organ Transplant Unit, London
 Hepatitis B and Hepatitis C Virus in non-liver Transplant Recipients Karim Qumosani MD, FRCPC, ABIM, MdMEd Multi-organ Transplant Unit, London Financial Disclosures Research Grants Merck, Gilead, Abbvie,
Hepatitis B and Hepatitis C Virus in non-liver Transplant Recipients Karim Qumosani MD, FRCPC, ABIM, MdMEd Multi-organ Transplant Unit, London Financial Disclosures Research Grants Merck, Gilead, Abbvie,
Direct acting anti-virals: the near future
 Direct acting anti-virals: the near future Heiner Wedemeyer Hannover Medical School Germany Will IFN-free treatment be possible in the near future? Interferon-free regimens to treat hepatitis C What should
Direct acting anti-virals: the near future Heiner Wedemeyer Hannover Medical School Germany Will IFN-free treatment be possible in the near future? Interferon-free regimens to treat hepatitis C What should
HBV Novel Therapies Maria Buti MD, PhD
 HBV Novel Therapies Maria Buti MD, PhD Liver Unit, Internal Medicine Department Vall d Hebron Hospital CONFLICT OF INTEREST I have financial relationships to disclose within the past 12 months relevant
HBV Novel Therapies Maria Buti MD, PhD Liver Unit, Internal Medicine Department Vall d Hebron Hospital CONFLICT OF INTEREST I have financial relationships to disclose within the past 12 months relevant
Waldenstrom s Macroglobulinemia
 Waldenstrom s Macroglobulinemia : Introduction Waldenstrom s macroglobulinemia (WM) is a lymphoma, or cancer of the lymphatic system. It occurs in a type of white blood cell called a B-lymphocyte or B-cell,
Waldenstrom s Macroglobulinemia : Introduction Waldenstrom s macroglobulinemia (WM) is a lymphoma, or cancer of the lymphatic system. It occurs in a type of white blood cell called a B-lymphocyte or B-cell,
A - Nonconfirmation Suspended by Sponsor B - D - Date site. C - Closed ready to start. Permissions. Sponsor delays ied. delayed/den.
 No REC Reference Number IRAS No Title Submission Type Valid Research Application Date of First Patient Recruitment Benchmark Met invited selected Approval date confirmed by sponsor confirmed A - Nonconfirmation
No REC Reference Number IRAS No Title Submission Type Valid Research Application Date of First Patient Recruitment Benchmark Met invited selected Approval date confirmed by sponsor confirmed A - Nonconfirmation
AASLD 2017 Viral Hepatitis Debrief
 AASLD 2017 Viral Hepatitis Debrief Paul Y Kwo MD Professor of Medicine Director of Hepatology Stanford University School of Medicine 750 Welch Road #210 Palo Alto, CA 94304-1507 P (650) 498-6080 F (650)
AASLD 2017 Viral Hepatitis Debrief Paul Y Kwo MD Professor of Medicine Director of Hepatology Stanford University School of Medicine 750 Welch Road #210 Palo Alto, CA 94304-1507 P (650) 498-6080 F (650)
Tenofovir alafenamide (chronic hepatitis B)
 IQWiG Reports Commission No. A17-13 Tenofovir alafenamide (chronic hepatitis B) Benefit assessment according to 35a Social Code Book V 1 Extract 1 Translation of Sections 2.1 to 2.7 of the dossier assessment
IQWiG Reports Commission No. A17-13 Tenofovir alafenamide (chronic hepatitis B) Benefit assessment according to 35a Social Code Book V 1 Extract 1 Translation of Sections 2.1 to 2.7 of the dossier assessment
Medication Policy Manual. Topic: Arzerra, ofatumumab Date of Origin: January 15, 2010
 Independent licensees of the Blue Cross and Blue Shield Association Medication Policy Manual Policy No: dru196 Topic: Arzerra, ofatumumab Date of Origin: January 15, 2010 Committee Approval Date: January
Independent licensees of the Blue Cross and Blue Shield Association Medication Policy Manual Policy No: dru196 Topic: Arzerra, ofatumumab Date of Origin: January 15, 2010 Committee Approval Date: January
Current Status of HBV and Liver Transplant
 Current Status of HBV and Liver HBV as Indication for Liver ation in U.S. Significant decrease in rate of wait listing for decompensated cirrhosis since 2003 (since s) No change in rate of wait listing
Current Status of HBV and Liver HBV as Indication for Liver ation in U.S. Significant decrease in rate of wait listing for decompensated cirrhosis since 2003 (since s) No change in rate of wait listing
AASLD 2017 Viral Hepatitis Debrief
 AASLD 2017 Viral Hepatitis Debrief Paul Y Kwo MD Professor of Medicine Director of Hepatology Stanford University School of Medicine 750 Welch Road #210 Palo Alto, CA 94304-1507 P (650) 498-6080 F (650)
AASLD 2017 Viral Hepatitis Debrief Paul Y Kwo MD Professor of Medicine Director of Hepatology Stanford University School of Medicine 750 Welch Road #210 Palo Alto, CA 94304-1507 P (650) 498-6080 F (650)
Treatment Op+ons for Chronic Hepa++s B. Judith Feinberg, MD Project ECHO Jan. 19, 2017
 Treatment Op+ons for Chronic Hepa++s B Judith Feinberg, MD Project ECHO Jan. 19, 2017 Treatment of Chronic Hepatitis B can be prevented by vaccinacon (part of infant series since 1992-3) goal of drug therapy
Treatment Op+ons for Chronic Hepa++s B Judith Feinberg, MD Project ECHO Jan. 19, 2017 Treatment of Chronic Hepatitis B can be prevented by vaccinacon (part of infant series since 1992-3) goal of drug therapy
Antiviral agents in HCV
 Antiviral agents in HCV : Upcoming Therapeutic Options Su Jong Yu, M.D., Ph.D. Department of Internal Medicine, Liver Research Institute, Seoul National University College of Medicine Estimated 170 Million
Antiviral agents in HCV : Upcoming Therapeutic Options Su Jong Yu, M.D., Ph.D. Department of Internal Medicine, Liver Research Institute, Seoul National University College of Medicine Estimated 170 Million
Who to Treat? Consider biopsy Treat. > 2 ULN Treat Treat Treat Treat CIRRHOTIC PATIENTS Compensated Treat HBV DNA detectable treat
 Who to Treat? Parameter AASLD US Algorithm EASL APASL HBV DNA CRITERIA HBeAg+ >, IU/mL > 2, IU/mL > 2, IU/mL >, IU/mL HBeAg- > 2, IU/mL > 2, IU/mL > 2, IU/mL > 2, IU/mL ALT CRITERIA PNALT 1-2 ULN Monitor
Who to Treat? Parameter AASLD US Algorithm EASL APASL HBV DNA CRITERIA HBeAg+ >, IU/mL > 2, IU/mL > 2, IU/mL >, IU/mL HBeAg- > 2, IU/mL > 2, IU/mL > 2, IU/mL > 2, IU/mL ALT CRITERIA PNALT 1-2 ULN Monitor
Management of Chronic HCV 2017 and Beyond
 Management of Chronic HCV 2017 and Beyond Blaire E Burman, MD Virginia Mason Gastroenterology & Hepatology Relevant Disclosures No financial disclosures to report Leaning Objectives Burden of HCV Prevalence
Management of Chronic HCV 2017 and Beyond Blaire E Burman, MD Virginia Mason Gastroenterology & Hepatology Relevant Disclosures No financial disclosures to report Leaning Objectives Burden of HCV Prevalence
Curriculum Vitae, Nirav S. Patel, M.D. Nirav S. Patel, M.D. Collaborative Neuroscience Network, LLC Redondo Avenue, Suites 415 & 500
 CONTACT INFORMATION: Site Selection and Information: Bobbie Theodore, Alliance Director Tel. (916) 939-6696 Fax (208) 575-3169 Email: clinicaltrials@alliancesites.com AFFILIATIONS: Collaborative Neuroscience
CONTACT INFORMATION: Site Selection and Information: Bobbie Theodore, Alliance Director Tel. (916) 939-6696 Fax (208) 575-3169 Email: clinicaltrials@alliancesites.com AFFILIATIONS: Collaborative Neuroscience
SASKATCHEWAN FORMULARY BULLETIN Update to the 62nd Edition of the Saskatchewan Formulary
 April 1, 2017 Bulletin #165 ISSN 1923-0761 SASKATCHEWAN FORMULARY BULLETIN Update to the 62nd Edition of the Saskatchewan Formulary Related Information for Prescribers: Only prescribers who have completed
April 1, 2017 Bulletin #165 ISSN 1923-0761 SASKATCHEWAN FORMULARY BULLETIN Update to the 62nd Edition of the Saskatchewan Formulary Related Information for Prescribers: Only prescribers who have completed
Slides are the property of the author and AASLD. Permission is required from both AASLD and the author for reuse.
 Inarigivir Demonstrates Potent Dose Dependent Anti-Viral Activity in HBV Treatment-Naïve Patients: Role of HBeAg Status and Baseline HBsAg in Anti-Viral Response MF Yuen, M. Elkhashab, CY Chen, YF Chen,
Inarigivir Demonstrates Potent Dose Dependent Anti-Viral Activity in HBV Treatment-Naïve Patients: Role of HBeAg Status and Baseline HBsAg in Anti-Viral Response MF Yuen, M. Elkhashab, CY Chen, YF Chen,
Primary Care Approach to Diagnosis and Management of Chronic Hepatitis C Brian Viviano, D.O.
 Primary Care Approach to Diagnosis and Management of Chronic Hepatitis C Brian Viviano, D.O. Objectives Epidemiology of chronic hepatitis C CDC guidelines on screening or hepatitis C Diagnosing hepatitis
Primary Care Approach to Diagnosis and Management of Chronic Hepatitis C Brian Viviano, D.O. Objectives Epidemiology of chronic hepatitis C CDC guidelines on screening or hepatitis C Diagnosing hepatitis
Harvoni (sofosbuvir/ledipasvir
 Market DC Override(s) Prior Authorization Quantity Limit (sofosbuvir/ledipasvir) Approval Duration Based on Genotype, Treatment status, Baseline HCV RNA status, Cirrhosis status, Transplant status, or
Market DC Override(s) Prior Authorization Quantity Limit (sofosbuvir/ledipasvir) Approval Duration Based on Genotype, Treatment status, Baseline HCV RNA status, Cirrhosis status, Transplant status, or
South London and Maudsley NHS Foundation Trust: FY1819 Q2 Performance in Initiating
 of First 17/YH/0195 228739 17/YH/0196 228435 17/LO/0987 215947 17/LO/1314 224111 17/WM/0441 224759 17/LO/1867 229005 A Randomized, Double-Blind, Placebo Controlled, Two-Period Cross-Over, Proof of Activity
of First 17/YH/0195 228739 17/YH/0196 228435 17/LO/0987 215947 17/LO/1314 224111 17/WM/0441 224759 17/LO/1867 229005 A Randomized, Double-Blind, Placebo Controlled, Two-Period Cross-Over, Proof of Activity
