Target date to recruit patients agreed? Target range. maximum. minimum. Date. 5 Agreed 26/01/ /11/2015 Recruitment Finished.
|
|
|
- Basil Harris
- 5 years ago
- Views:
Transcription
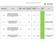
1 No REC Reference IRAS No Title Target number of agreed? Target range minimum Target range maximum Target date to recruit agreed? Planned Recruitment end date Total no of recruited at the agreed target date Total no of recruited study actually closed to recruitment at STH Reason for closure to recruitment at STH 1 10/H1307/ A Compassionate Use Open-Label Study of Anti IL-5 (Mepolizumab) Treatment in Subjects with Hypereosinophilic Syndrome Agreed 1 1 Agreed 01/01/ /01/2016 Recruitment Finished 2 11/LO/ LUX Head & Neck 2; A randomised, doubleblind, placebo controlled, phase III study to evaluate the efficacy and safety of afatinib (BIBW 2992) as adjuvant therapy after chemoradiotherapy in primary unresected with stage III, IVa, or IVb locoregionally advanced head and neck squamous cell carcinoma. Agreed 5 5 Agreed 26/01/ /11/2015 Recruitment Finished International non-randomized, single arm, long-term follow-up study of with uncontrolled 3 12/LO/ hypertension A randomised, multicentre, open-label Phase III study to evaluate the efficacy and safety of Trastuzumab Emtansine versus Trastuzumab as adjuvant therapy for with 4 12/LO/ HER2-positive primary breast cancer Agreed Agreed 31/01/ /09/2016 Recruitment Finished Agreed 3 3 Agreed 30/04/ /01/2016 Recruitment Finished Efficacy and Safety of AM-101 in the Treatment of Acute Peripheral 5 13/EE/ Tinnitus 3 (TACTT3) 6 13/LO/ Agreed 3 5 Agreed 30/06/ /07/2016 Recruitment Finished A Phase II Simon Two Stage Efficacy Study of Intracerebral CTX0E03 DP in Patients with Stable Paresis of the Arm Following an Ischaemic Stroke. Agreed 2 4 Agreed 05/08/ /08/2016 Recruitment Finished
2 A randomized, double-blind, parallelgroup, placebo and active controlled, multicenter study to evaluate the efficacy, safety and tolerability of combinations of solifenacin succinate 5mg and mirabegron 25 and 50mg compared to slifenacin succinate and mirabegron monotherapy in the 7 13/LO/ treatment of overactive bladder. CALM-NET: A Phase IV, Multicentre, Open label, Single Group Exploratory Study to Assess the Clinical Value of 8 13/LO/ Enumeration of Circulating Tumour 9 13/NI/ Agreed 8 8 Agreed 13/03/ /02/2016 Recruitment Finished Agreed 4 4 Agreed 22/07/ /07/2016 Recruitment Finished A randomised, double-blind, placebo controlled, multicentre trial to measure the oral corticosteroid-sparing effect of lebrikizumab in with severe corticosteroid dependent asthma - VOCALS Agreed 7 7 Agreed 02/02/ /02/2016 Recruitment Finished A Phase III randomized, 3-arm, open label, multicenter study of LGX818 plus MEK162 and LGX818 monotherapy compared with vemurafenib in with unresectable or metastatic BRAF 10 13/SC/ V600 mutant melanoma Study of Oral Pacritinib versus Best Available Therapy in Patients with Thrombocytopenia and Primary Myelofibrosis, Post-Polycythemia Vera Myelofibrosis, or Post-Essential Thrombocythemia Myelofibrosis (PERSIST-2) 11 14/EM/ Agreed 3 3 Agreed 31/03/ /10/2015 Recruitment Finished Agreed 3 4 Agreed 01/01/ /02/2016 Recruitment Finished 12 14/EM/ An open-label, multicenter study to provide with chronic heart failure and reduced ejection fraction access to LCZ696 following participation in PARADIGM-HF Agreed 2 2 Agreed 29/02/ /02/2016 Recruitment Finished
3 Prospective, randomized, placebocontrolled, double-blind, multicenter, parallel-group, 24-week study to assess the efficacy, safety and tolerability of macitentan in subjects with inoperable chronic 13 14/LO/ thromboembolic pulmonary Agreed 2 2 Agreed 18/03/ /03/2016 Recruitment Finished 14 14/LO/ /LO/ A Phase 3, Randomized, Placebo- Controlled, Double-Blind Study of Oral MLN9708 Maintenance Therapy in Patients with Multiple Myeloma Following Autologous Stem Cell Transplant - Millennium A Multi-center, Prospective, Randomized, Controlled Trial of Endobronchial Valve (EBV) Therapy vs. Standard of Care (SoC) in Heterogeneous Emphysema Agreed 3 3 Agreed 01/12/ /03/2016 Recruitment Finished Agreed 5 10 Agreed 30/06/ /06/2016 Recruitment Finished 16 14/LO/ /LO/ /NW/ Long term, multicenter, single-arm, open-label extension study of the MERIT-1 study, to assess the safety, tolerability and efficacy of macitentan in subjects with inoperable chronic thromboembolic pulmonary hypertension (CTEPH) Study : Albiglutide versus Placebo in Subjects with new- onset type 1 diabetes mellitus A 4 week phase 2a, multicentre, randomised, doubleblind, placebocontrolled add-on study into safety, tolerability and efficacy of 200 mg t.i.d. of PL37 in with peripheral neuropathic pain of diabetic origin treated with pregabalin or gabapentin Agreed 1 1 Agreed 27/06/ /05/2016 Recruitment Finished Agreed 3 3 Agreed 26/05/ /05/2016 Recruitment Finished Agreed 7 7 Agreed 15/03/ /03/2016 Recruitment Finished
4 Comparative testing of Transfix Vacuum blood collection tubes and Cytochex blood collection tubes for FDA 510 (k) submission 19 14/NW/ A phase 3 randomized, double-blind study assessing the efficacy and safety of pf and 20 14/SC/ adalimumab in combination with 21 14/SC/ A Multicenter, Randomized, Double Blind, Placebo controlled, Parallel Group, Phase 3 Study to Evaluate the Efficacy and Safety of Dapagliflozin as an Add on to Insulin Therapy in Subjects with Type 1 Diabetes Mellitus Agreed Agreed 25/02/ /02/2016 Recruitment Finished Agreed 1 4 Agreed 28/02/ /02/2016 Recruitment Finished Agreed 5 5 Agreed 31/10/ /04/2016 Recruitment Finished A Phase 3b, Multi-center, Randomized-withdrawal, Placebocontrolled, Double-blind, Parallelgroup Trial to Compare the Efficacy and Safety of Tolvaptan (45 to 120 mg/day, split dose) in Subjects with Chronic Kidney Disease Between Late Stage 2 to Early Stage 4 Due to Autosomal Dominant Polycystic 22 14/SS/ Kidney Disease A multinational, randomised, double blind, placebo controlled trial to evaluate the effect of ticagrelor 90mg twice daily on the incidence of cardiovascular death, myocardial infarction or stroke in with 23 14/WM/ type 2 diabetes mellitus. (THEMIS) 24 14/WS/ A phase 3, multicentre, open label, randomised trial of nab-paclitaxel plus Gemcitabine versus Gemcitabine alone as adjuvant therapy in subjects with surgically resected pancreatic Not Agreed 5 Available / 5 Not Agreed /01/2016 Recruitment Finished Agreed Agreed 03/06/ /05/2016 Withdrawn By Sponsor Agreed Agreed 30/06/ /03/2016 Recruitment Finished
5 A Phase 2, Randomized, Placebo Controlled, Double Blind, Proof of concept Study of the Efficacy and Safety of PF in Subjects 25 14/YH/ with Huntington s disease 26 15/EM/ A Multi Center, Open Label Study Evaluating the Safety, Tolerability, and Efficacy of Pridopidine in Patients with Huntington s Disease (Open PRIDE HD) Agreed 4 10 Agreed 19/02/ /03/2016 Recruitment Finished Agreed 0 3 Agreed 30/07/ /02/2016 Recruitment Finished 27 15/LO/ Safety and Efficacy of Brimonidine Posterior Segment Drug Delivery System in Patients with Geographic Atrophy Secondary to Age-related Macular Degeneration. Agreed 8 10 Agreed 09/09/ /09/2016 Recruitment Finished 28 15/LO/ ARMOR3-SV A Phase 3, randomised, open label, multicentre, controlled study of Galeterone compared to Enzalutamide in men expressing androgen receptor splice variant-7 mrna (AR-V7) metastatic (M1) castrate resistant prostate cancer (CRPC). Effects of ODM-109 on respiratory function in with ALS. A randomised, double blind, placebocontrolled, cross-over, 3 period, multicentre study with open-label 29 15/LO/ follow-up extension /LO/ An Open-Label, Multicenter Clinical Trial with Nivolumab (BMS ) Monotherapy in Subjects with Advanced or Metastatic Squamous Cell (Sq) Non-Small Cell Lung Cancer (NSCLC) who Have Received at Least One Prior Systemic Regimen for the Treatment of Stage IIIb/IV SqNSCLC Agreed 0 0 Agreed 31/12/ /07/2016 Withdrawn By Sponsor Agreed 5 10 Agreed 31/07/ /07/2016 Recruitment Finished Agreed 9 9 Agreed 15/06/ /06/2016 Recruitment Finished
6 An open-label, multi-center, nonrandomized Phase Ib study to investigate safety and tolerability of radium Ra 223 dichloride (BAY ) administered in combination with paclitaxel in cancer subjects with 31 15/LO/ bone lesions. Agreed 2 4 Agreed 28/02/ /02/2016 Recruitment Finished 32 15/NW/ A Phase 3, Randomized, Doubleblind, Placebo-controlled, Parallel- Group, Multicenter Study to Evaluate the Efficacy, Safety and Tolerability of LX4211 as Adjunct Therapy in Adults Patients with Type 1 Diabetes Mellitus Who Have Inadequate Glycemic Control with Insulin Therapy. Agreed 5 8 Agreed 30/04/ /04/2016 Recruitment Finished 33 15/NW/ /SC/ A Prospective Observational Trial to Evaluate the Correlation of T-SPOT Response to CMV Infection and T cellmediated Acute Graft Rejection. The PROTECT Study. Agreed Agreed 31/01/ /09/2016 Recruitment Finished A randomised, double-blind, placebocontrolled, phase 3 trial of doxorubicin plus olaratumab versus doxorubicin plus placebo in with advanced or metastatic soft tissue sarcoma. Agreed Agreed 30/06/ /05/2016 Withdrawn By Sponsor 35 15/SC/ A Randomized, double blind, placebocontrolled, phase 3 study to assess the efficacy and safety of KRN23 in adults with x-linked hypophosphotaemia (XLH). Agreed 6 6 Agreed 30/06/ /06/2016 Recruitment Finished
7 A Randomized, Open-label, Phase 3 Study Assessing Safety and Efficacy in Subjects with Relapsed and Refractory Multiple Myeloma Receiving Once-weekly Carfilzomib Compared to Twice-weekly Carfilzomib in Combination with Dexamethasone /WM/ /YH/ Acceptability study on Nutricomp Drink Plus in adult Agreed 4 5 Agreed 30/08/ /08/2016 Recruitment Finished Agreed Agreed 31/10/ /10/2015 Recruitment Finished 38 15/YH/ A phase 3, randomized, double-blind, placebo-controlled study investigating the efficacy and safety of multiple Dupilumab dose regimens administered as monotherapy for maintaining treatment response in with atopic dermatitis. Agreed 0 5 Agreed 01/12/ /12/2015 Recruitment Finished 39 15/YH/ /EM/ Multi-center, Open-Label, Randomized, Two-Arm, Parallel- Group Study to Assess Efficacy and Safety of Envarsus Compared with Tacrolimus Used as Per Current Clinical Practice in the Initial Maintenance Setting in De Novo Kidney Transplant Patients The BAHA Attract System with Cochlear BAHA 5 SuperPower. Agreed 4 4 Agreed 30/08/ /07/2016 Recruitment Finished Agreed 3 5 Agreed 30/07/ /09/2016 Recruitment Finished 41 GTAC A Phase I study of the safety, tolerability and biological effect of single and repeat administration of the selectively replication-competent herpes virus HSV1716 into the tumourbearing pleural cavity (intrapleural) in with inoperable malignant pleural mesothelioma Agreed Agreed 26/09/ /09/2016 Recruitment Finished
Target date to recruit patients agreed? Target range minimum. Target range maximum. Agreed 1 1 Agreed 01/01/ /01/2016 Recruitment Finished
 No REC Reference IRAS No Title Target number of patients agreed? Target range minimum Target range maximum Target date to recruit patients agreed? Planned Recruitment end date No of patients recruited
No REC Reference IRAS No Title Target number of patients agreed? Target range minimum Target range maximum Target date to recruit patients agreed? Planned Recruitment end date No of patients recruited
Target date to recruit patients agreed? Target range minimum. Target range maximum
 No REC Reference IRAS No Title Target number of patients agreed? Target range minimum Target range maximum Target date to recruit patients agreed? Planned Recruitment end date Total no of patients recruited
No REC Reference IRAS No Title Target number of patients agreed? Target range minimum Target range maximum Target date to recruit patients agreed? Planned Recruitment end date Total no of patients recruited
Min number of patients agreed to be recruited. Max number of patients agreed to be recruited
 be 12/SC/0139 98125 A PHASE III PROSPECTIVE, TWO-COHORT NON-RANDOMIZED, MULTI- CENTRE, MULTINATIONAL, OPEN LABEL STUDY TO ASSESS THE SAFETY OF ASSISTED- AND SELF-ADMINISTERED SUBCUTANEOUS TRASTUZUMAB AS
be 12/SC/0139 98125 A PHASE III PROSPECTIVE, TWO-COHORT NON-RANDOMIZED, MULTI- CENTRE, MULTINATIONAL, OPEN LABEL STUDY TO ASSESS THE SAFETY OF ASSISTED- AND SELF-ADMINISTERED SUBCUTANEOUS TRASTUZUMAB AS
Studies proceeding under pre HRA-Approval system (NHS Permission)
 15/NW/0004 162679 Studies proceeding under pre HRA-roval system (NHS Permission) A MULTI-CENTRE RANDOMISED CLINICAL TRIAL OF BIOMARKER-DRIVEN MAINTENANCE TREATMENT FOR FIRST- LINE METASTATIC COLORECTAL
15/NW/0004 162679 Studies proceeding under pre HRA-roval system (NHS Permission) A MULTI-CENTRE RANDOMISED CLINICAL TRIAL OF BIOMARKER-DRIVEN MAINTENANCE TREATMENT FOR FIRST- LINE METASTATIC COLORECTAL
The Pennine Acute Hospitals NHS Trust Performance in delivering clinical research Reporting period 1 October 2017 to 30 September 2018
 The Pennine Acute Hospitals NHS Trust Performance in delivering clinical research Reporting period 1 October 2017 to 30 September 2018 REC reference IRAS reference Name of trial Target number of? Minimum
The Pennine Acute Hospitals NHS Trust Performance in delivering clinical research Reporting period 1 October 2017 to 30 September 2018 REC reference IRAS reference Name of trial Target number of? Minimum
Perfomance in Delivering (Commercial Trials)
 Perfomance in Delivering (Commercial Trials) REC Ref IRAS ID Name of Trial Target Of? Minimum Of Maximum Of Target Date To Recruit? Total Date Of to recruit target number Recruited At of patients The Target
Perfomance in Delivering (Commercial Trials) REC Ref IRAS ID Name of Trial Target Of? Minimum Of Maximum Of Target Date To Recruit? Total Date Of to recruit target number Recruited At of patients The Target
Performance in Initiating Clinical Research Q2 2016/17
 Performance in Initiating Clinical Research Q2 2016/17 Research Ethics Committee Reference Number Integrated Research Application System Number Name of Trial Date of Receipt of Valid Research Application
Performance in Initiating Clinical Research Q2 2016/17 Research Ethics Committee Reference Number Integrated Research Application System Number Name of Trial Date of Receipt of Valid Research Application
Date of end of Recruitment Window at STH. Target No of Patients. No REC Ref Title
 o REC Ref Title Target o of Patients Date of end of Recruitment Window at STH Trial Status Target Met 1 06/MRE12/35 2 06/MRE04/84 3 07/MRE01/68 A randomised, double-blind, placebo-controlled, multicentre,
o REC Ref Title Target o of Patients Date of end of Recruitment Window at STH Trial Status Target Met 1 06/MRE12/35 2 06/MRE04/84 3 07/MRE01/68 A randomised, double-blind, placebo-controlled, multicentre,
The Pennine Acute Hospitals NHS Trust Performance in initiating clinical research Reporting period 1 October 2017 to 30 September 2018
 The Pennine Acute Hospitals NHS Trust Performance in initiating clinical research Reporting period 1 October 2017 to 30 September 2018 REC reference IRAS reference Name of trial First patient recruited?
The Pennine Acute Hospitals NHS Trust Performance in initiating clinical research Reporting period 1 October 2017 to 30 September 2018 REC reference IRAS reference Name of trial First patient recruited?
Performance in Initiating Clinical Research
 Performance in Initiating Clinical Research January 2016 December 2016 NHS Permissions Site approval Research Ethics Committee Reference Number Integrated Research Application System Number Name of Trial
Performance in Initiating Clinical Research January 2016 December 2016 NHS Permissions Site approval Research Ethics Committee Reference Number Integrated Research Application System Number Name of Trial
A - Nonconfirmation Suspended by Sponsor B - D - Date site. C - Closed ready to start. Permissions. Sponsor delays ied. delayed/den.
 No REC Reference Number IRAS No Title Submission Type Valid Research Application Date of First Patient Recruitment Benchmark Met invited selected Approval date confirmed by sponsor confirmed A - Nonconfirmation
No REC Reference Number IRAS No Title Submission Type Valid Research Application Date of First Patient Recruitment Benchmark Met invited selected Approval date confirmed by sponsor confirmed A - Nonconfirmation
HRA Approval Date Site Date Site Sponsor. Non- Confirmation Status. Date Site Ready To Start
 Research Ethics Committee Reference Number Integrated Research Application System Number Submission Type Name of Trial Date of Receipt of Date of Valid Research Application First Patient Recruited? Date
Research Ethics Committee Reference Number Integrated Research Application System Number Submission Type Name of Trial Date of Receipt of Date of Valid Research Application First Patient Recruited? Date
F - No Eligible G - No Patients. D - Sponsor delays. C - Closed by Sponsor. E - Staff Availability issues. patient seen)
 No REC Reference Number IRAS No Title Valid Research Application Date of First Patient Recruitment A - Permissions delayed/deni ed B - Suspended by C - Closed by D - delays E - Staff Availability issues
No REC Reference Number IRAS No Title Valid Research Application Date of First Patient Recruitment A - Permissions delayed/deni ed B - Suspended by C - Closed by D - delays E - Staff Availability issues
NIHR Performance Metrics - Initiating Clinical Research. Quarter 2 ( )
 NIHR Performance Metrics - Initiating Clinical Research Quarter 2 (2014-2015) Research Ethics Committee Reference Number Name of Trial Date of Receipt of Valid Research Application Date of First Patient
NIHR Performance Metrics - Initiating Clinical Research Quarter 2 (2014-2015) Research Ethics Committee Reference Number Name of Trial Date of Receipt of Valid Research Application Date of First Patient
Date of First Patient Recruitment. Benchm ark Met. A - Permissions delayed/denied
 No REC Ref Title Authorisation Valid Research Date Application 1 13/EE/0011 2 14/SS/1043 Date of First Patient Recruitment A Phase III Randomised Trial of Gemcitabine plus Docetaxel followed by Doxorubicin
No REC Ref Title Authorisation Valid Research Date Application 1 13/EE/0011 2 14/SS/1043 Date of First Patient Recruitment A Phase III Randomised Trial of Gemcitabine plus Docetaxel followed by Doxorubicin
Clinical Research Institute HUCH Sponsored, fully signed Clinical Trial Agreements (except grants)
 , fully signed Clinical Trial Agreements (except grants) Sponsor Full name of the Hospital clinic where the is details of the January x x January x NightstaRx Ltd A Randomised, Open Label, Outcomes-Assessor
, fully signed Clinical Trial Agreements (except grants) Sponsor Full name of the Hospital clinic where the is details of the January x x January x NightstaRx Ltd A Randomised, Open Label, Outcomes-Assessor
NCIC CLINICAL TRIALS GROUP DATA SAFETY MONITORING COMMITTEE Friday, 1 May 2009 SUMMARY REPORT
 NCIC CLINICAL TRIALS GROUP DATA SAFETY MONITORING COMMITTEE Friday, 1 May 2009 SUMMARY REPORT The NCIC CTG DSMC reviewed the following trials with respect to safety, trial conduct, including accrual, and
NCIC CLINICAL TRIALS GROUP DATA SAFETY MONITORING COMMITTEE Friday, 1 May 2009 SUMMARY REPORT The NCIC CTG DSMC reviewed the following trials with respect to safety, trial conduct, including accrual, and
National Cancer Drugs Fund List - Approved
 National Cancer Drugs Fund List - Approved DRUG Abiraterone Aflibercet Albumin Bound Paclitaxel Axitinib CDF INDICATION (EXCLUDING APPROVED CRITERIA ) Metastatic Prostate Cancer Metastatic Colorectal Cancer
National Cancer Drugs Fund List - Approved DRUG Abiraterone Aflibercet Albumin Bound Paclitaxel Axitinib CDF INDICATION (EXCLUDING APPROVED CRITERIA ) Metastatic Prostate Cancer Metastatic Colorectal Cancer
Performance in Initiating Clinical Research Q4 2015/16
 Performance in Initiating Clinical Research Q4 2015/16 Research Ethics Committee Reference Number Integrated Research Application System Number Name of Trial Date of Receipt of Valid Research Application
Performance in Initiating Clinical Research Q4 2015/16 Research Ethics Committee Reference Number Integrated Research Application System Number Name of Trial Date of Receipt of Valid Research Application
NCIC CLINICAL TRIALS GROUP DATA SAFETY MONITORING COMMITTEE Fall Conference Call 23 November 2009 SUMMARY REPORT
 NCIC CLINICAL TRIALS GROUP DATA SAFETY MONITORING COMMITTEE Fall Conference Call 23 November 2009 SUMMARY REPORT The NCIC CTG DSMC reviewed the following trials with respect to safety, trial conduct, including
NCIC CLINICAL TRIALS GROUP DATA SAFETY MONITORING COMMITTEE Fall Conference Call 23 November 2009 SUMMARY REPORT The NCIC CTG DSMC reviewed the following trials with respect to safety, trial conduct, including
Integrated Research Application System Number. Total Number Of Study Participants Recruited. Research Ethics Committee Reference Number
 Research Ethics Committee Reference Number Integrated Research Application System Number Name of Trial Minimum Number Of Patients Agreed Maximum Number Of Patients Agreed Date That The Trial Closed To
Research Ethics Committee Reference Number Integrated Research Application System Number Name of Trial Minimum Number Of Patients Agreed Maximum Number Of Patients Agreed Date That The Trial Closed To
Performance in Delivering Q4. Target number of patients
 Performance in Delivering Q4 Research Ethics Committee Reference Number Name of Trial Target number of patients Date Agreed to recruit target number of patients Trial Status Target met within the agreed
Performance in Delivering Q4 Research Ethics Committee Reference Number Name of Trial Target number of patients Date Agreed to recruit target number of patients Trial Status Target met within the agreed
CLINICAL TRIALS ACC. Jul 2016
 CLINICAL TRIALS ACC Jul 2016 Glioblastoma BRAIN A071102 A Phase II/III Randomized Trial of Veliparib or Placebo in Combination With Temozolomide in Newly Diagnosed Glioblastoma With MGMT Promoter Hypermethylation
CLINICAL TRIALS ACC Jul 2016 Glioblastoma BRAIN A071102 A Phase II/III Randomized Trial of Veliparib or Placebo in Combination With Temozolomide in Newly Diagnosed Glioblastoma With MGMT Promoter Hypermethylation
Open Clinical Trials: What s Out There Now Paula D. Ryan, MD, PhD
 Open Clinical Trials: What s Out There Now Paula D. Ryan, MD, PhD Hanahan and Weinberg, 2000 Acquired Capabilities of Cancer Clinical Trials When should I consider a clinical trial? How do I find the right
Open Clinical Trials: What s Out There Now Paula D. Ryan, MD, PhD Hanahan and Weinberg, 2000 Acquired Capabilities of Cancer Clinical Trials When should I consider a clinical trial? How do I find the right
London Cancer New Drugs Group. February London Cancer New Drugs Group (LCNDG) Work Plan for the London Cancer Drugs Fund list.
 February 2013 London Cancer New s Group (LCNDG) Work Plan for the London Cancer s Fund London Cancer s Fund List This Cancer s Fund (CDF) list of medicines and s is in two parts. 1. The standard list of
February 2013 London Cancer New s Group (LCNDG) Work Plan for the London Cancer s Fund London Cancer s Fund List This Cancer s Fund (CDF) list of medicines and s is in two parts. 1. The standard list of
Date of HRA. Date Site Invited. Date Site Selected. Approval. Recruited. Date
 14/LO/0259 134883 RE-AKT: A randomised Phase II study of enzalutamide (MDV3100) in combination with AZD5363 in s with Metastatic Castration - Resistant Prostate Cancer Yes 08/01/2018 01/07/2016 01/07/2017
14/LO/0259 134883 RE-AKT: A randomised Phase II study of enzalutamide (MDV3100) in combination with AZD5363 in s with Metastatic Castration - Resistant Prostate Cancer Yes 08/01/2018 01/07/2016 01/07/2017
SUPPLEMENTARY INFORMATION
 SUPPLEMENTARY NFORMATON n format provided by Melero et al. (AUGUST 2015) Supplementary nformation S3 Combinations including two or more immunotherapy agents based on PD-1/PD-L1 blockade. (Source: https://clinicaltrials.gov/
SUPPLEMENTARY NFORMATON n format provided by Melero et al. (AUGUST 2015) Supplementary nformation S3 Combinations including two or more immunotherapy agents based on PD-1/PD-L1 blockade. (Source: https://clinicaltrials.gov/
Performance in delivering Clinical Research at PHT Q3 14/15 study met the 70 day target study within 70 day target study not met 70 day target
 Performance in delivering Clinical Research at PHT Q3 14/15 study met the 70 day target study within 70 day target study not met 70 day target Research Ethics Committee 14/ES/0004 ALA-BCC-CT008 - A randomized,
Performance in delivering Clinical Research at PHT Q3 14/15 study met the 70 day target study within 70 day target study not met 70 day target Research Ethics Committee 14/ES/0004 ALA-BCC-CT008 - A randomized,
Date of NHS Permission. Date of First Patient Recruited. Date Site Invited
 Reference of Receipt of of NHS of First 14/WA/1075 150715 NHS HeadPoST Version 2.1 23/11/2015 05/01/2016 11/01/2016 N/A 12/YH/0452 106985 NHS INCA 10/12/2015 15/01/2016 17/03/2016 N/A Excess Treatment
Reference of Receipt of of NHS of First 14/WA/1075 150715 NHS HeadPoST Version 2.1 23/11/2015 05/01/2016 11/01/2016 N/A 12/YH/0452 106985 NHS INCA 10/12/2015 15/01/2016 17/03/2016 N/A Excess Treatment
All Studies by Indication
 Therapeutic Area Protocol Number Drug/Device Phase Description Advanced malignancies 337 20101132 AMG 337 I " A Phase 1, First-in-Human Study Evaluating the Safety, Tolerability, and Pharmacokinetics of
Therapeutic Area Protocol Number Drug/Device Phase Description Advanced malignancies 337 20101132 AMG 337 I " A Phase 1, First-in-Human Study Evaluating the Safety, Tolerability, and Pharmacokinetics of
Performance in Initiating and Delivering Clinical Research
 Performance in Initiating and Delivering Clinical Research As a global leading centre for research in Eyes and Vision, it is a key priority to offer as many patients as possible the opportunity to get
Performance in Initiating and Delivering Clinical Research As a global leading centre for research in Eyes and Vision, it is a key priority to offer as many patients as possible the opportunity to get
Table 1 Salford Royal NHS Foundation Trust: Performance in initiating clinical research (HRA Approval) Reporting period 1 April March 2017
 Table 1 Salford Royal NHS Foundation Trust: Performance in initiating clinical research (HRA Approval) Reporting period 1 April 2016 31 March 2017 REC reference IRAS number Study title 16/NW/0428 207062
Table 1 Salford Royal NHS Foundation Trust: Performance in initiating clinical research (HRA Approval) Reporting period 1 April 2016 31 March 2017 REC reference IRAS number Study title 16/NW/0428 207062
Performance in Initiating and Delivering Clinical Research
 Performance in Initiating and Delivering Clinical Research As a global leading centre for research in Eyes and Vision, it is a key priority to offer as many patients as possible the opportunity to get
Performance in Initiating and Delivering Clinical Research As a global leading centre for research in Eyes and Vision, it is a key priority to offer as many patients as possible the opportunity to get
NIHR Performance Metrics - Initiating Clinical Research. Quarter 3 ( )
 NIHR Performance Metrics - Initiating Clinical Research Quarter 3 (2014-2015) Research Ethics Committee Reference Number 13/LO/0291 Name of Trial Insight Insulin Pump EU Study: A European Multicenter Study
NIHR Performance Metrics - Initiating Clinical Research Quarter 3 (2014-2015) Research Ethics Committee Reference Number 13/LO/0291 Name of Trial Insight Insulin Pump EU Study: A European Multicenter Study
PERFORMANCE IN DELIVERING CLINICAL RESEARCH (Q1, 2016/17)
 PERFORMANCE IN DELIVERING CLINICAL RESEARCH (Q1, 2016/17) Research Ethics Committee Reference Name of Trial Target Of Patients? Minimum Of Patients (Enter Same In Both If Only One ) Maximum Of Patients
PERFORMANCE IN DELIVERING CLINICAL RESEARCH (Q1, 2016/17) Research Ethics Committee Reference Name of Trial Target Of Patients? Minimum Of Patients (Enter Same In Both If Only One ) Maximum Of Patients
Table 1: Salford Royal NHS Foundation Trust: Performance in initiating clinical research: Reporting period 1 July June 2013
 Table 1: Salford Royal NHS Foundation Trust: Performance in initiating clinical research: Reporting period 1 July 2012-30 June 2013 Name of trial Note, some study titles are shortened 1 Long-Term Study
Table 1: Salford Royal NHS Foundation Trust: Performance in initiating clinical research: Reporting period 1 July 2012-30 June 2013 Name of trial Note, some study titles are shortened 1 Long-Term Study
Performance in Initiating and Delivering Clinical Research
 Performance in Initiating and Delivering Clinical Research As a global leading centre for research in Eyes and Vision, it is a key priority to offer as many patients as possible the opportunity to get
Performance in Initiating and Delivering Clinical Research As a global leading centre for research in Eyes and Vision, it is a key priority to offer as many patients as possible the opportunity to get
Title Cancer Drug Phase Status
 Clinical Trial Identifier Title Cancer Drug Phase Status NCT01164995 Study With Wee-1 Inhibitor MK-1775 and Carboplatin to Treat p53 Mutated Refractory and Resistant Ovarian Cancer Epithelial Ovarian Cancer
Clinical Trial Identifier Title Cancer Drug Phase Status NCT01164995 Study With Wee-1 Inhibitor MK-1775 and Carboplatin to Treat p53 Mutated Refractory and Resistant Ovarian Cancer Epithelial Ovarian Cancer
NIHR Performance Metrics - Initiating Clinical Research. Quarter 1 ( )
 NIHR Performance Metrics - Initiating Clinical Research Quarter 1 (2014-2015) Research Ethics Committee Reference Number Name of Trial Date of Receipt of Valid Research Application Date of First Patient
NIHR Performance Metrics - Initiating Clinical Research Quarter 1 (2014-2015) Research Ethics Committee Reference Number Name of Trial Date of Receipt of Valid Research Application Date of First Patient
Summary of Strategic Competitive Analysis and Publication Planning
 Summary of Strategic Competitive Analysis and Publication Planning Carole Alison Chrvala, PhD 919.545.2149 (Work) 919.951.5230 (Mobile) cchrvala@centurylink.net www.healthmattersmedwriting.com 1 Briefing
Summary of Strategic Competitive Analysis and Publication Planning Carole Alison Chrvala, PhD 919.545.2149 (Work) 919.951.5230 (Mobile) cchrvala@centurylink.net www.healthmattersmedwriting.com 1 Briefing
N. Ireland Cancer Trials Centre/ N. Ireland Cancer Trials Network Trials Open to Recruitment June 2013
 N. Ireland Cancer Trials Centre/ N. Ireland Cancer Trials Network Trials Open to Recruitment June 2013 Bladder HABIO Haematuria Biomarker Study Bowel Cancer NSCCG National Study Of Colorectal Cancer Genetics
N. Ireland Cancer Trials Centre/ N. Ireland Cancer Trials Network Trials Open to Recruitment June 2013 Bladder HABIO Haematuria Biomarker Study Bowel Cancer NSCCG National Study Of Colorectal Cancer Genetics
General Information, efficacy and safety data
 Horizon Scanning in Oncology Horizon Scanning in Oncology 28 th Prioritization 3 rd quarter 2016 General Information, efficacy and safety data Nicole Grössmann Claudia Wild Please note: Within this document
Horizon Scanning in Oncology Horizon Scanning in Oncology 28 th Prioritization 3 rd quarter 2016 General Information, efficacy and safety data Nicole Grössmann Claudia Wild Please note: Within this document
Table 2: Salford Royal NHS Foundation Trust: Performance in delivering clinical research: Reporting period 1 January December 2014
 Table 2: Salford Royal HS Foundation Trust: Performance in delivering clinical research: Reporting period 1 January 2014 1 December 2014 Research Ethics Committee reference number 1 12/W/0321 2 11/LO/1986
Table 2: Salford Royal HS Foundation Trust: Performance in delivering clinical research: Reporting period 1 January 2014 1 December 2014 Research Ethics Committee reference number 1 12/W/0321 2 11/LO/1986
NIHR Performance Metrics - Initiating Clinical Research. Quarter 4 ( )
 NIHR Performance Metrics - Initiating Clinical Research Quarter 4 (2013-2014) Research Ethics Committee Reference Number Name of Trial Date of Receipt of Valid Research Application Date of First Patient
NIHR Performance Metrics - Initiating Clinical Research Quarter 4 (2013-2014) Research Ethics Committee Reference Number Name of Trial Date of Receipt of Valid Research Application Date of First Patient
The Royal Marsden NHS Foundation Trust Medicines Formulary. NICE register for medicines initiated at the trust. Type and code. Publication.
 NICE register for medicines initiated at the trust. CG denotes Clinical Guideline TA denotes Technical Appraisal For further information about NICE and full guidelines please refer to www.nice.org.uk Title
NICE register for medicines initiated at the trust. CG denotes Clinical Guideline TA denotes Technical Appraisal For further information about NICE and full guidelines please refer to www.nice.org.uk Title
PART VI: SUMMARY OF THE RISK MANAGEMENT PLAN BY PRODUCT
 PAGE 175 PART VI: SUMMARY OF THE RISK MANAGEMENT PLAN BY PRODUCT Summary of risk management plan for pembrolizumab This is a summary of the risk management plan (RMP) for pembrolizumab. The RMP details
PAGE 175 PART VI: SUMMARY OF THE RISK MANAGEMENT PLAN BY PRODUCT Summary of risk management plan for pembrolizumab This is a summary of the risk management plan (RMP) for pembrolizumab. The RMP details
General Information, efficacy and safety data
 Horizon Scanning in Oncology Horizon Scanning in Oncology 23 rd Prioritization 2 nd quarter 2015 General Information, efficacy and safety data Eleen Rothschedl Anna Nachtnebel Priorisierung XXIII HSS Onkologie
Horizon Scanning in Oncology Horizon Scanning in Oncology 23 rd Prioritization 2 nd quarter 2015 General Information, efficacy and safety data Eleen Rothschedl Anna Nachtnebel Priorisierung XXIII HSS Onkologie
Date Site Confirmed By Sponsor. Non- Confirmation Status. HRA Approval Date. Date Site Confirmed
 Research Ethics Committee Reference Number Integrated Research Application System Number Submission Type Name of Trial First Patient Recruited? Date of First Patient Recruited Invited Selected Approval
Research Ethics Committee Reference Number Integrated Research Application System Number Submission Type Name of Trial First Patient Recruited? Date of First Patient Recruited Invited Selected Approval
Currently recruiting trials and/or near future recruitment
 Clinical Trials at the Bank of Cyprus Oncology Centre (As of Jan 2018) Currently recruiting trials and/or near future recruitment Development of a module to supplement the EORTC Core instruments for assessment
Clinical Trials at the Bank of Cyprus Oncology Centre (As of Jan 2018) Currently recruiting trials and/or near future recruitment Development of a module to supplement the EORTC Core instruments for assessment
29 August 2016 Page 1 of 7. How does the NHS board decide which new medicines to make available for patients?
 NHS Greater Glasgow and Clyde: New Medicines Decisions In Scotland, a newly licensed medicine is routinely available for use in an NHS board only after it has been: accepted for use in the NHSScotland
NHS Greater Glasgow and Clyde: New Medicines Decisions In Scotland, a newly licensed medicine is routinely available for use in an NHS board only after it has been: accepted for use in the NHSScotland
NIHR Performance Metrics - Initiating Clinical Research. Quarter 4 (
 NIHR Performance Metrics - Initiating Clinical Research Quarter 4 (2014-2015 Research Ethics Committee Reference Number Name of Trial Date of Receipt of Valid Research Application Date of First Patient
NIHR Performance Metrics - Initiating Clinical Research Quarter 4 (2014-2015 Research Ethics Committee Reference Number Name of Trial Date of Receipt of Valid Research Application Date of First Patient
Open NCT NCT Open NCT
 Updated Feb 18/16. Please note, although every effort is made to keep this list as up-to-date as possible, some trial status updates may not be reflected in the list below. DST Project Title Project Display
Updated Feb 18/16. Please note, although every effort is made to keep this list as up-to-date as possible, some trial status updates may not be reflected in the list below. DST Project Title Project Display
MEDICAL PRIOR AUTHORIZATION
 MEDICAL PRIOR AUTHORIZATION TAXOTERE (docetaxel) DOCEFREZ(docetaxel) docetaxel (generic) POLICY I. INDICATIONS The indications below including FDA-approved indications and compendial uses are considered
MEDICAL PRIOR AUTHORIZATION TAXOTERE (docetaxel) DOCEFREZ(docetaxel) docetaxel (generic) POLICY I. INDICATIONS The indications below including FDA-approved indications and compendial uses are considered
Performance in Initiating and Delivering Clinical Research
 Performance in Initiating and Delivering Clinical Research As a global leading centre for research in Eyes and Vision, it is a key priority to offer as many as possible the opportunity to get involved
Performance in Initiating and Delivering Clinical Research As a global leading centre for research in Eyes and Vision, it is a key priority to offer as many as possible the opportunity to get involved
Performance in Delivering - Q2 2015/2016. Date Agreed to recruit target number of patients. Trial Status. 2 01/12/2015 Open N/A
 12/SC/0394 11/SC/0272 12/LO/0482 A Randomized, Open Label Study of Ofatumumab and Bendamustine Combination Therapy Compared with Bendamustine Monotherapy in Indolent Bcell onhodgkin?s Lymphoma Unresponsive
12/SC/0394 11/SC/0272 12/LO/0482 A Randomized, Open Label Study of Ofatumumab and Bendamustine Combination Therapy Compared with Bendamustine Monotherapy in Indolent Bcell onhodgkin?s Lymphoma Unresponsive
Date of First Patient Recruitment. Bench mark met. A - Permissions delayed/denied
 No REC Ref Title Authorisation Date Valid Research Application Date of First Patient Recruitment Bench mark met A - Permissions delayed/denied B - Suspended by Sponsor C - Closed by Sponsor D - Sponsor
No REC Ref Title Authorisation Date Valid Research Application Date of First Patient Recruitment Bench mark met A - Permissions delayed/denied B - Suspended by Sponsor C - Closed by Sponsor D - Sponsor
Performance in Initiating and Delivering Clinical Research Why are we doing this?
 Performance in Initiating and Delivering Clinical Research Why are we doing this? Through the NIHR (National Institute for Health Research) the Government wishes to see a dramatic and sustained improvement
Performance in Initiating and Delivering Clinical Research Why are we doing this? Through the NIHR (National Institute for Health Research) the Government wishes to see a dramatic and sustained improvement
HRA Approval Date. confirmed by sponsor 07/04/ /04/ /04/ /04/ /04/ /04/ /05/2016
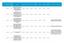 Reference Reason for delay in recruiting first 1 15/NE/0144 179243 AN OPEN-LABEL EXTENSION AND SAFETY MONITORING STUDY OF PATIENTS WITH MODERATELY TO SEVERELY ACTIVE CROHN S DISEASE PREVIOUSLY ENROLLED
Reference Reason for delay in recruiting first 1 15/NE/0144 179243 AN OPEN-LABEL EXTENSION AND SAFETY MONITORING STUDY OF PATIENTS WITH MODERATELY TO SEVERELY ACTIVE CROHN S DISEASE PREVIOUSLY ENROLLED
University Hospitals of Leicester NHS Trust Research & Innovation Directorate Performance in Delivering Research 01/04/2015 to 31/03/2016
 University Hospitals of Leicester NHS Trust Research & Innovation Directorate Performance in Delivering Research 01/04/2015 to 31/03/2016 REC Date 10/H0808/109 58307 EXCEL Clinical Trial 1.0 8th June 2010
University Hospitals of Leicester NHS Trust Research & Innovation Directorate Performance in Delivering Research 01/04/2015 to 31/03/2016 REC Date 10/H0808/109 58307 EXCEL Clinical Trial 1.0 8th June 2010
Opdivo. Opdivo (nivolumab) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.21.53 Subsection: Antineoplastic Agents Original Policy Date: January 16, 2015 Subject: Opdivo Page:
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.21.53 Subsection: Antineoplastic Agents Original Policy Date: January 16, 2015 Subject: Opdivo Page:
A - Permissions delayed/den ied. Date site ready to start. B - Suspended by Sponsor. C - Closed D - Sponsor
 No REC Reference IRAS No Number Title Date of First Patient Recruitment Benchmark Met invited selected HRA Approval date confirmed by sponsor confirmed ready to start A - Permissions delayed/den ied B
No REC Reference IRAS No Number Title Date of First Patient Recruitment Benchmark Met invited selected HRA Approval date confirmed by sponsor confirmed ready to start A - Permissions delayed/den ied B
Clinical Research Institute HUCH Sponsored, fully signed Clinical Trial Agreements (except grants)
 1. 2. The is Sponsor Full name of the Hospital clinic where the is details of the January January NightstaR Ltd A Randomised, Open Label, Outcomes-Assessor Masked, Prospective, Parallel Controlled Group,
1. 2. The is Sponsor Full name of the Hospital clinic where the is details of the January January NightstaR Ltd A Randomised, Open Label, Outcomes-Assessor Masked, Prospective, Parallel Controlled Group,
National Horizon Scanning Centre. Sunitinib (Sutent) for advanced and/or metastatic breast cancer. December 2007
 Sunitinib (Sutent) for advanced and/or metastatic breast cancer December 2007 This technology summary is based on information available at the time of research and a limited literature search. It is not
Sunitinib (Sutent) for advanced and/or metastatic breast cancer December 2007 This technology summary is based on information available at the time of research and a limited literature search. It is not
Now Available: Final Rule for FDAAA 801 and NIH Policy on Clinical Trial Reporting
 A service of the U.S. National Institutes of Health Now Available: Final Rule for FDAAA 801 and NIH Policy on Clinical Trial Reporting Trial record 1 of 1 for: Keynote 355 Previous Study Return to List
A service of the U.S. National Institutes of Health Now Available: Final Rule for FDAAA 801 and NIH Policy on Clinical Trial Reporting Trial record 1 of 1 for: Keynote 355 Previous Study Return to List
Curriculum Vitae ARUN K. KALRA, MD
 Curriculum Vitae ARUN K. KALRA, MD Office: CCARE California Cancer Associates in Research and Excellence 39755 Date St. Suite 103 Murrieta, California 92563 01/2017 to current. Arun Kalra MD Inc. 09/2016
Curriculum Vitae ARUN K. KALRA, MD Office: CCARE California Cancer Associates in Research and Excellence 39755 Date St. Suite 103 Murrieta, California 92563 01/2017 to current. Arun Kalra MD Inc. 09/2016
A JOHNS HOPKINS SINGAPORE ANNUAL RESEARCH REPORT 2015
 A JOHNS HOPKINS SINGAPORE ANNUAL RESEARCH REPORT 2015 15 years of excellence in patient care, research, and education The Promise of Medicine: The research team at Johns Hopkins Singapore provides unparalleled
A JOHNS HOPKINS SINGAPORE ANNUAL RESEARCH REPORT 2015 15 years of excellence in patient care, research, and education The Promise of Medicine: The research team at Johns Hopkins Singapore provides unparalleled
Ontario s Referral and Listing Criteria for Adult Pancreas-After- Kidney Transplantation
 Ontario s Referral and Listing Criteria for Adult Pancreas-After- Kidney Transplantation Version 2.0 Trillium Gift of Life Network Adult Pancreas-After-Kidney Transplantation Referral & Listing Criteria
Ontario s Referral and Listing Criteria for Adult Pancreas-After- Kidney Transplantation Version 2.0 Trillium Gift of Life Network Adult Pancreas-After-Kidney Transplantation Referral & Listing Criteria
Avastin (bevacizumab) DRUG.00028, CG-DRUG-68
 Avastin (bevacizumab) DRUG.00028, CG-DRUG-68 Override(s) Prior Authorization Approval Duration 1 year Medications Avastin (bevacizumab) APPROVAL CRITERIA Requests for Avastin (bevacizumab) may be approved
Avastin (bevacizumab) DRUG.00028, CG-DRUG-68 Override(s) Prior Authorization Approval Duration 1 year Medications Avastin (bevacizumab) APPROVAL CRITERIA Requests for Avastin (bevacizumab) may be approved
Etudes cliniques Service d Oncologie - Radiothérapie
 Etudes cliniques Service d Oncologie - Radiothérapie Décembre 2017 SEIN NEO ADJUVANT WO39392 (Impassion 031) : A phase III randomized study to investigate the efficacy and safety of Atezolizumab in combination
Etudes cliniques Service d Oncologie - Radiothérapie Décembre 2017 SEIN NEO ADJUVANT WO39392 (Impassion 031) : A phase III randomized study to investigate the efficacy and safety of Atezolizumab in combination
Immune Checkpoint Inhibitors: The New Breakout Stars in Cancer Treatment
 Immune Checkpoint Inhibitors: The New Breakout Stars in Cancer Treatment 1 Introductions Peter Langecker, MD, PhD Executive Medical Director, Global Oncology Clinipace Worldwide Mark Shapiro Vice President
Immune Checkpoint Inhibitors: The New Breakout Stars in Cancer Treatment 1 Introductions Peter Langecker, MD, PhD Executive Medical Director, Global Oncology Clinipace Worldwide Mark Shapiro Vice President
Drugs approved from 1st January 2015 till present. S.NO Name Of Drug Indication Date of Approval 1 Levocetirizine ODS 2.5mg /5mg (Additional
 Drugs approved from 1st January 2015 till present S.NO Name Of Drug Indication Date of 1 Levocetirizine ODS 2.5mg /5mg dosage form) For allergic rhinitis and chronic urticaria. 14.01.15 2 Decitabine Injection
Drugs approved from 1st January 2015 till present S.NO Name Of Drug Indication Date of 1 Levocetirizine ODS 2.5mg /5mg dosage form) For allergic rhinitis and chronic urticaria. 14.01.15 2 Decitabine Injection
University of Groningen. Health economics of targeted cancer therapies Mihajlovic, Jovan
 University of Groningen Health economics of targeted therapies Mihajlovic, Jovan IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's PDF) if you wish to cite from it. Please
University of Groningen Health economics of targeted therapies Mihajlovic, Jovan IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's PDF) if you wish to cite from it. Please
Molecular Profiling METASTATIC, REFRACTORY, HER2+, BREAST PHASE 1, METASTATIC, RELAPSED/REFRACTORY, PROSTATE OVARIAN
 FOR MORE INFORMATION OR TO REFER A PATIENT TO ANY OF OUR CLINICAL TRIALS PLEASE CONTACT NICOLE GIVEN, BS, OHC RESEARCH DEPARTEMTN AT 513-751- 2273, EXT 27110 Phase 1 REFMAL 381 (Phase 1 open at BAM only)
FOR MORE INFORMATION OR TO REFER A PATIENT TO ANY OF OUR CLINICAL TRIALS PLEASE CONTACT NICOLE GIVEN, BS, OHC RESEARCH DEPARTEMTN AT 513-751- 2273, EXT 27110 Phase 1 REFMAL 381 (Phase 1 open at BAM only)
List of Qualifying Conditions
 List of Qualifying Conditions Cancer Conditions 1) Adrenal cancer 2) Bladder cancer 3) Bone cancer all forms 4) Brain cancer 5) Breast cancer 6) Cervical cancer 7) Colon cancer 8) Colorectal cancer 9)
List of Qualifying Conditions Cancer Conditions 1) Adrenal cancer 2) Bladder cancer 3) Bone cancer all forms 4) Brain cancer 5) Breast cancer 6) Cervical cancer 7) Colon cancer 8) Colorectal cancer 9)
Clinical Policy: Nivolumab (Opdivo) Reference Number: CP.PHAR.121 Effective Date: Last Review Date: Line of Business: Medicaid
 Clinical Policy: (Opdivo) Reference Number: CP.PHAR.121 Effective Date: 07.15 Last Review Date: 01.18 Line of Business: Medicaid Revision Log See Important Reminder at the end of this policy for important
Clinical Policy: (Opdivo) Reference Number: CP.PHAR.121 Effective Date: 07.15 Last Review Date: 01.18 Line of Business: Medicaid Revision Log See Important Reminder at the end of this policy for important
Summary of late stage clinical trials. As of Jul. 22, 2016
 Summary of late stage clinical trials As of Jul. 22, 2016 List of abbreviations AE BID DLT GFR iv MTD ORR OS PD PFS PK po PPK Q2W Q3W Q4W QD QW sc TID Adverse Events Twice daily Dose Limiting Toxicity
Summary of late stage clinical trials As of Jul. 22, 2016 List of abbreviations AE BID DLT GFR iv MTD ORR OS PD PFS PK po PPK Q2W Q3W Q4W QD QW sc TID Adverse Events Twice daily Dose Limiting Toxicity
Practical Considerations in Multiple Myeloma: Optimizing Therapy With New Proteasome Inhibitors
 Welcome to Managing Myeloma. My name is Dr. Donald Harvey. I am Director of Phase 1 Clinical Trials Section and an Associate Professor in Hematology, Medical Oncology, and Pharmacology at the Winship Cancer
Welcome to Managing Myeloma. My name is Dr. Donald Harvey. I am Director of Phase 1 Clinical Trials Section and an Associate Professor in Hematology, Medical Oncology, and Pharmacology at the Winship Cancer
CHAPTER 3 DEATHS. Stephen McDonald Leonie Excell Brian Livingston
 CHAPTER 3 DEATHS Stephen McDonald Leonie Excell Brian Livingston DEATHS ANZDATA Registry 2008 Report INTRODUCTION AUSTRALIA NEW ZEALAND The total number of deaths was 1,452 (15.4 deaths per 100 patient
CHAPTER 3 DEATHS Stephen McDonald Leonie Excell Brian Livingston DEATHS ANZDATA Registry 2008 Report INTRODUCTION AUSTRALIA NEW ZEALAND The total number of deaths was 1,452 (15.4 deaths per 100 patient
Immuno-Oncology Applications
 Immuno-Oncology Applications Lee S. Schwartzberg, MD, FACP West Clinic, P.C.; The University of Tennessee Memphis, Tn. ICLIO 1 st Annual National Conference 10.2.15 Philadelphia, Pa. Financial Disclosures
Immuno-Oncology Applications Lee S. Schwartzberg, MD, FACP West Clinic, P.C.; The University of Tennessee Memphis, Tn. ICLIO 1 st Annual National Conference 10.2.15 Philadelphia, Pa. Financial Disclosures
Dennis J Slamon, MD, PhD
 I N T E R V I E W Dennis J Slamon, MD, PhD Dr Slamon is Professor of Medicine, Chief of the Division of Hematology/Oncology and Director of Clinical and Translational Research at UCLA s David Geffen School
I N T E R V I E W Dennis J Slamon, MD, PhD Dr Slamon is Professor of Medicine, Chief of the Division of Hematology/Oncology and Director of Clinical and Translational Research at UCLA s David Geffen School
Dr. Andres Wiernik. Lung Cancer
 Dr. Andres Wiernik Lung Cancer Lung Cancer Facts - Demographics World Incidence: 1 8 million / year World Mortality: 1 6 million / year 5-year survival rates vary from 4 17% depending on stage and regional
Dr. Andres Wiernik Lung Cancer Lung Cancer Facts - Demographics World Incidence: 1 8 million / year World Mortality: 1 6 million / year 5-year survival rates vary from 4 17% depending on stage and regional
A randomised controlled clinical trial in type 2 diabetes comparing semaglutide to placebo and liraglutide
 A randomised controlled clinical trial in type 2 diabetes comparing semaglutide to placebo and liraglutide This trial was conducted in Europe,Asia and Africa. Study participants were randomised evenly
A randomised controlled clinical trial in type 2 diabetes comparing semaglutide to placebo and liraglutide This trial was conducted in Europe,Asia and Africa. Study participants were randomised evenly
Directorate General of Health Services Office of Drugs Controller General (India) (Biological Division)
 Directorate General of Health Services Office of Drugs Controller General (India) (Biological Division) List of r-dna drug products approved in the country (Form 45 and 45A) from 1 st Jan 2015 to 30 th
Directorate General of Health Services Office of Drugs Controller General (India) (Biological Division) List of r-dna drug products approved in the country (Form 45 and 45A) from 1 st Jan 2015 to 30 th
Keytruda (pembrolizumab)
 Keytruda (pembrolizumab) Line(s) of Business: HMO; PPO; QUEST Integration Akamai Advantage Original Effective Date: 10/01/2015 Current Effective Date: 07/24/2017TBD03/01/2018 POLICY A. INDICATIONS The
Keytruda (pembrolizumab) Line(s) of Business: HMO; PPO; QUEST Integration Akamai Advantage Original Effective Date: 10/01/2015 Current Effective Date: 07/24/2017TBD03/01/2018 POLICY A. INDICATIONS The
Open and Pending Trials Listing For Arizona Oncology Associates P.C. HOPE Division/ Tucson, Arizona
 Open and Pending Trials Listing For Arizona Oncology Associates P.C. HOPE Division/ Tucson, Arizona Solid Tumor: USON 15180 / A Multicenter, Open-Label Phase 1b/2 Trial of Lenvatinib (E7080) Plus Pembrolizumab
Open and Pending Trials Listing For Arizona Oncology Associates P.C. HOPE Division/ Tucson, Arizona Solid Tumor: USON 15180 / A Multicenter, Open-Label Phase 1b/2 Trial of Lenvatinib (E7080) Plus Pembrolizumab
Curriculum Vitae, Joseph J. Savon, M.D.
 CONTACT INFORMATION: Joseph J. Savon, M.D. Hassman Research Institute, LLC. 175 Cross Keys Road Centennial Center, Bldg. 300 B Berlin, NJ 08009 Site Selection and Information: Bobbie Theodore, Alliance
CONTACT INFORMATION: Joseph J. Savon, M.D. Hassman Research Institute, LLC. 175 Cross Keys Road Centennial Center, Bldg. 300 B Berlin, NJ 08009 Site Selection and Information: Bobbie Theodore, Alliance
The Clinical Research E-News
 The Clinical Research E-News Volume 7: ISSUE 1: January 16, 2015 Jefferson Kimmel Cancer Center Network: For urgent clinical trial questions or assistance please page: 877-656-9004 New Featured Trials
The Clinical Research E-News Volume 7: ISSUE 1: January 16, 2015 Jefferson Kimmel Cancer Center Network: For urgent clinical trial questions or assistance please page: 877-656-9004 New Featured Trials
Etudes cliniques Service d Oncologie - Radiothérapie
 Etudes cliniques Service d Oncologie - Radiothérapie Juillet 2017 SEIN NEO ADJUVANT Neo-RHEA : NEOadjuvant Biomarker ResearcH Study of Palbociclib in Estrogen Receptor Positive HER2 Negative Breast CAncer
Etudes cliniques Service d Oncologie - Radiothérapie Juillet 2017 SEIN NEO ADJUVANT Neo-RHEA : NEOadjuvant Biomarker ResearcH Study of Palbociclib in Estrogen Receptor Positive HER2 Negative Breast CAncer
Bone Marrow Transplant Gynecologic Immunotherapy Phase 1. Breast Head & Neck Melanoma/Skin Sarcoma
 Karmanos offers clinical trials for many different forms of cancer. Below are the most current clinical trials open to new participants as of this publication's issue date. If you have any questions regarding
Karmanos offers clinical trials for many different forms of cancer. Below are the most current clinical trials open to new participants as of this publication's issue date. If you have any questions regarding
KEYTRUDA is also indicated in combination with pemetrexed and platinum chemotherapy for the
 FDA-Approved Indication for KEYTRUDA (pembrolizumab) in Combination With Carboplatin and Either Paclitaxel or Nab-paclitaxel for the Firstline Treatment of Patients With Metastatic Squamous Non Small Cell
FDA-Approved Indication for KEYTRUDA (pembrolizumab) in Combination With Carboplatin and Either Paclitaxel or Nab-paclitaxel for the Firstline Treatment of Patients With Metastatic Squamous Non Small Cell
Clinical Policy: Nivolumab (Opdivo) Reference Number: CP.PHAR.121
 Clinical Policy: (Opdivo) Reference Number: CP.PHAR.121 Effective Date: 07/15 Last Review Date: 04/17 Coding Implications Revision Log See Important Reminder at the end of this policy for important regulatory
Clinical Policy: (Opdivo) Reference Number: CP.PHAR.121 Effective Date: 07/15 Last Review Date: 04/17 Coding Implications Revision Log See Important Reminder at the end of this policy for important regulatory
JOHNS HOPKINS SINGAPORE ANNUAL RESEARCH REPORT 2014
 JOHNS HOPKINS SINGAPORE ANNUAL RESEARCH REPORT 2014 15 years of excellence in patient care, research, and education The Promise of Medicine: The research team at Johns Hopkins Singapore provides unparalleled
JOHNS HOPKINS SINGAPORE ANNUAL RESEARCH REPORT 2014 15 years of excellence in patient care, research, and education The Promise of Medicine: The research team at Johns Hopkins Singapore provides unparalleled
Table 1: Salford Royal NHS Foundation Trust: Performance in initiating clinical research: Reporting period 1 October September 2013
 Table 1: Salford Royal NHS Foundation Trust: Performance in initiating clinical research: Reporting period 1 October 2012-30 September 2013 Research Ethics Committee reference number Name of Trial 1 12/NW/0321
Table 1: Salford Royal NHS Foundation Trust: Performance in initiating clinical research: Reporting period 1 October 2012-30 September 2013 Research Ethics Committee reference number Name of Trial 1 12/NW/0321
Personalized Gene Profile
 Sequence Variants: 6 4 ALTERATION MUTANT FRACTION FDA GUIDANCE (for indication) FDA GUIDANCE (for other indications) TRIALS (details below) c.38g>a; p.g13d 21.0% Cetuximab Contraindicated 22 Panitumumab
Sequence Variants: 6 4 ALTERATION MUTANT FRACTION FDA GUIDANCE (for indication) FDA GUIDANCE (for other indications) TRIALS (details below) c.38g>a; p.g13d 21.0% Cetuximab Contraindicated 22 Panitumumab
New Targeted Agents Demonstrate Greater Efficacy and Tolerability in the Treatment of HER2-positive Breast Cancer
 New Evidence reports on presentations given at ASCO 2012 New Targeted Agents Demonstrate Greater Efficacy and Tolerability in the Treatment of HER2-positive Breast Cancer Presentations at ASCO 2012 Breast
New Evidence reports on presentations given at ASCO 2012 New Targeted Agents Demonstrate Greater Efficacy and Tolerability in the Treatment of HER2-positive Breast Cancer Presentations at ASCO 2012 Breast
Ontario s Referral and Listing Criteria for Adult Kidney Transplantation
 Ontario s Referral and Listing Criteria for Adult Kidney Transplantation Version 3.0 Trillium Gift of Life Network Adult Kidney Transplantation Referral & Listing Criteria PATIENT REFERRAL CRITERIA: The
Ontario s Referral and Listing Criteria for Adult Kidney Transplantation Version 3.0 Trillium Gift of Life Network Adult Kidney Transplantation Referral & Listing Criteria PATIENT REFERRAL CRITERIA: The
This clinical study synopsis is provided in line with Boehringer Ingelheim s Policy on Transparency and Publication of Clinical Study Data.
 abcd Clinical Study for Public Disclosure This clinical study synopsis is provided in line with s Policy on Transparency and Publication of Clinical Study Data. The synopsis which is part of the clinical
abcd Clinical Study for Public Disclosure This clinical study synopsis is provided in line with s Policy on Transparency and Publication of Clinical Study Data. The synopsis which is part of the clinical
Development status of ONO-4538(nivolumab)1
 Development status of ONO-4538(nivolumab)1 Development Code Target Disease JAPAN US/EU KOR/TAI ONO-4538 Melanoma(2 nd ~) Launched Launched (US) Filing (EU) Approved(KOR) Filing(TAI) ONO-4538 Melanoma(1
Development status of ONO-4538(nivolumab)1 Development Code Target Disease JAPAN US/EU KOR/TAI ONO-4538 Melanoma(2 nd ~) Launched Launched (US) Filing (EU) Approved(KOR) Filing(TAI) ONO-4538 Melanoma(1
