Significant events in immunisation policy and practice* in Australia
|
|
|
- Lisa Hood
- 6 years ago
- Views:
Transcription
1 Significant events in immunisation policy and practice* in Australia Year 1804 First vaccine (for smallpox) used in Australia 1916 Commonwealth Serum Laboratories (CSL) established in Victoria to produce vaccine for Australia 1924 First mass vaccination program in Australia (with diphtheria toxoid in Victoria) 1932 First school-based vaccination programs (for diphtheria) commenced First time vaccine provided (for diphtheria-tetanus-pertussis) at no cost to recipients in the public sector 1st edition of Immunisation Procedures (now The Australian Immunisation Handbook) published by of the National Health and Medical Research Council (NHMRC). 2nd edition of Immunisation Procedures (now The Australian Immunisation Handbook) published by of the NHMRC. 3rd edition of Immunisation Procedures (now The Australian Immunisation Handbook) published by of the NHMRC. Responsibility for immunisation programs transferred to the states and territories. This resulted in variation across Australia in implementation of the national immunisation program. Arrangements for funding immunisation programs were negotiated through the Australian Health Ministers Advisory Council (AHMAC). Communicable Diseases Network Australia (CDNA) established as the Communicable Diseases Control Network, a joint initiative of the NHMRC and AHMAC Legislation in Victoria required documentation of immunisation status when a child enrolled in primary school Health Amendment Act 1990 (now in the Public Health and Wellbeing Act 2008) 4th edition of Immunisation Procedures (now The Australian Immunisation Handbook) published by of the NHMRC. Northern Territory Immunisation Register (NTIR) launched, capturing vaccines administered to both children and adults Legislation in New South Wales required documentation of immunisation status when a child enrolled in childcare or primary school introduced in a 1992 amendment of the Public Health Act 1991 (now in the Public Health Act 2010) First National Immunisation Strategy ( ), produced by a panel of experts on behalf of the NHMRC, included a common vaccination schedule and vaccine pricing in all states and territories National Immunisation Committee (NIC) established as a sub-committee of the AHMAC to oversee the implementation of the 1993 National Immunisation Strategy and to provide advice to the AHMAC on immunisation and vaccine-preventable disease issues Vaccination Information and Vaccination Administration System (VIVAS) launched in Queensland, capturing vaccines administered to both children and adults 5th edition of The Australian Immunisation Procedures Handbook (now The Australian Immunisation Handbook) published by the Australian Government. Vaccine recommendations were made by an expert advisory sub-committee of the NHMRC. Legislation in the Australian Capital Territory required documentation of immunisation status when a child enrolled in childcare, pre-school or primary school ACT Public Health Regulations 2000
2 1996 Australian Childhood Immunisation Register (ACIR) launched by collating data on all children <7 years of age enrolled in Medicare. This was the first complete purpose-built national childhood immunisation register in the world and replaced ad hoc regional registers. The Register is administered by the Australian Government Department of Human Services as per the Health Insurance Act 1973 s.46b. Immunisation provider incentives introduced by the Australian government. An ACIR notification payment of $6 per notification is made to all immunisation providers who notify the ACIR of a vaccination that completes one of the age-based schedules. In Queensland, immunisation providers receive $3 per notification, in recognition that a separate register (VIVAS) is maintained in that state. The Immunise Australia Program launched, also known as the National Immunisation Program (NIP). The Program included a Seven Point Plan outlining a series of initiatives designed to increase immunisation coverage: 1. Incentives for parents 2. General practice immunisation incentive scheme 3. Monitoring and evaluation of immunisation targets 4. Immunisation days 5. Measles elimination strategy 6. Education and research through the establishment of the National Centre for Immunisation Research and Surveillance of Vaccine Preventable Diseases (NCIRS) 7. School-entry legislative requirements Public Health Outcome Funding Agreements (PHOFAs) established between the Australian and state and territory governments to fund the NIP 1997 National Health Act 1953 amended to list designated vaccines that may be provided free of charge to eligible people under the NIP on the National Health (Immunisation Program Designated Vaccines) Determination 6th edition of The Australian Immunisation Handbook published by the Australian Government. Vaccine recommendations were made by an expert advisory sub-committee of the NHMRC. Australian Technical Advisory Group on Immunisation (ATAGI) established as part of the development of the Immunise Australia Program to provide technical advice related to government vaccine funding decisions directly to the federal health minister. ATAGI replaced the expert sub-committee of the NHMRC that had previously produced national clinical guidelines on immunisation for health professionals. NCIRS established by the Australian Government Department of Health and Ageing as part of the development of the Immunise Australia Program to provide technical support to ATAGI Legislation in Tasmania required documentation of immunisation status when a child enrolled in day care or school Tasmania Public Health Act 1997 First Victorian Immunisation Strategy (1997) developed Australian Childhood Immunisation Charter (ACIC) Protecting Our Children developed as part of the Immunise Australia Program. This described the fundamental principles and practices governing childhood immunisation and articulated goals related to vaccine coverage to be achieved by year 2000.
3 1998 Australian Government introduced a nationwide scheme of financial immunisation incentives for: 1. General practitioners (GPs) a. ACIR notification payment (paid to general practitioners since 1996) b. General Practice Immunisation Incentives (GPII) scheme direct and indirect payments to GPs including: 2. Parents i. Service Incentive Payment (SIP) for individual GPs who report the completion of ageappropriate vaccinations for children <7 years of age $18.50 per report ii. Outcomes Bonus Payment (OBP) scheme for general practices $3.50 per fully immunised whole-patient equivalent if practice coverage is 90% for children aged <7 years a. The Maternity Immunisation Allowance (MIA) $200 per fully immunised child at 19 months of age (means-tested) b. Childcare Assistance Rebate and/or the Childcare Cash Rebate in 1998, $20 $122 per child per week (means-tested) National serosurveillance program commenced. The first serosurvey was conducted using specimens collected before ( ) and after (1999) the national Measles Control Campaign. NIC repositioned under the auspices of the National Public Health Partnership due to a realignment of AHMAC sub-committees 2000 Child Care Benefit replaced Childcare Assistance Rebate and Childcare Cash Rebate linked to a child s immunisation status ACIR legislation amended to allow immunisations given overseas to be recorded if a provider endorsed their validity 7th edition of The Australian Immunisation Handbook published by the Australian Government NIC repositioned again to report to National Public Health Partnership through CDNA Second national serosurvey conducted using specimens collected in 2002 NHMRC approved the new National Immunisation Program Schedule (NIPS). For the first time since 1994 when all vaccines recommended on the schedule were funded for children under the National Immunisation Strategy the recommended childhood schedule contained vaccines (IPV, varicella and 7vPCV) that were not available free of charge to parents. 8th edition of The Australian Immunisation Handbook published by the Australian Government. First New South Wales Immunisation strategy ( ) developed First Tasmanian Immunisation strategy (2003) developed 2004 Means-testing removed from the Maternity Immunisation Allowance National Heath Act 1953 amended to provide for the evaluation of cost-effectiveness of vaccines by the Pharmaceutical Benefits Advisory Committee (PBAC). The amendment included provisions that the Health Minister could not approve a vaccine for funding under the NIP unless it was deemed by PBAC to be cost-effective for inclusion on the Australian Pharmaceutical Benefits Scheme (PBS) First National Vaccine Storage Guidelines (Strive for 5) published, to assist immunisation service providers to maintain the safety and viability of vaccines. Legislation in Queensland included school exclusion provisions for contagious conditions but no requirement for documentation of immunisation status when a child enrolled in school Queensland Public Health Act 2005 and Public Health Regulation 2005
4 2007 Third national serosurvey conducted using specimens collected in 2007 First Australian Capital Territory Immunisation Strategy ( ) developed 9th edition of The Australian Immunisation Handbook published by the Australian Government National HPV Vaccination Program Register established to support the National HPV Vaccination Program. A notification payment ($6 per dose notified) was introduced and paid only to GPs who had registered and who notified vaccines administered under the community-based catch-up component of the program. Australian Government introduced the Healthy Start for School check for all 4-year-old children to ensure that every child has a basic health check prior to starting school. Documented completion of a Healthy Start for School check is linked to receipt of the Family Tax Benefit Part A supplement. Immunisation status is reviewed by a health professional during a health check. General Practice Immunisation Incentive scheme Service Incentive Payment (GPII SIP) ceased Second New South Wales Immunisation Strategy ( ) developed 2009 The Maternity Immunisation Allowance and childcare-related parent incentive payments split into two payments of $129 (2009 to end June 2012) for a fully immunised child aged months and 4 5 years: 1. continuing financial incentives for vaccines due by 12 months of age; and 2. introducing a financial incentive for vaccines due at 4 years of age, when coverage and timeliness were lowest Western Australia established a statewide Grade 8 vaccination database for school-based vaccination to help ensure students were appropriately immunised. The database permits calculation of vaccination coverage rates and generates a paper-based vaccination record provided to each student at the end of the year. Second Victorian Immunisation Strategy ( ) developed 2010 Report on the management of adverse events associated with Panvax and Fluvax (the Horvath Review ) published, examining the vaccine safety system in Australia following adverse events associated with seasonal influenza vaccine in children in 2010 National HPV Vaccination Program Register notification payment to GPs ceased 2012 Maternity Immunisation Allowance discontinued; instead immunisation status became linked to the existing means-tested Family Tax Benefit Part A supplement for each child at ages 1, 2 and 5 years (maximum of $726 per child, per age milestone). Linkage of immunisation status to eligibility for Australian Government Child Care Benefit was unchanged. Fourth national serosurvey conducted using specimens collected in Second Australian Capital Territory Immunisation Strategy ( ) developed 10th edition of The Australian Immunisation Handbook published by the Australian Government. General Practice Immunisation Incentive scheme Outcomes Bonus Payment (GPII OBP) ceased National HPV Vaccination Program Register began to record HPV vaccinations given to males 2013 First Western Australian Immunisation Strategy ( ) developed Second National Immunisation Strategy for Australia developed Pharmacy Board of Australia announced that vaccination was within the current scope of practice of pharmacists once the pharmacist completes a training course accredited by the Australian Pharmacy Council and acts in accordance with any practice standards approved by the Director-General 2nd edition of National Vaccine Storage Guidelines Strive for 5 published
5 First Queensland Immunisation Strategy developed Legislation in Western Australia amended to allow pharmacists to administer influenza vaccine Poisons and Therapeutic Goods Act th edition of The Australian Immunisation Handbook updated and endorsed by the NHMRC AusVaxSafety implemented. This is a national, collaborative active vaccine safety surveillance initiative led by NCIRS and funded by the Australian Government Department of Health. Essential Vaccines Procurement Strategy developed. Under the new arrangement, the Commonwealth is responsible for procurement of NIP vaccines while the implementation will remain the responsibility of the states and territories. Legislation in South Australia amended to allow appropriately trained pharmacists to administer influenza vaccine to people aged over 16 years who are not eligible for vaccine through the NIP Poisons and Therapeutic Goods Act Legislation in New South Wales amended to allow appropriately trained pharmacists to administer influenza vaccine to people aged 18 years or over at a retail pharmacy other than on prescription Poisons and Therapeutic Goods Regulation 2008 Legislation in the Northern Territory amended to allow pharmacists in the course of practising the pharmacy profession to supply and administer a vaccine to a person in accordance with the approved scheduled substance treatment protocol Medicines, Poisons and Therapeutic Goods Act 10th edition of The Australian Immunisation Handbook updated and endorsed by the NHMRC New legislated immunisation requirements for family assistance payments introduced by the Australian Government known as the No Jab, No Pay measure Only parents of children (aged <20 years) who are, according to the ACIR, immunised with all NIP vaccines or on a recognised catch-up schedule can receive the Child Care Benefit, the Child Care Rebate and the Family Tax Benefit Part A supplement. Vaccinations must be recorded on the ACIR. Children with medical contraindications or natural immunity for certain diseases continue to be exempt from the requirements Conscientious objection and vaccination objection on non-medical grounds that were formerly permissible no longer a valid exemption from immunisation requirements Interim Vaccination Objection Form for Enrolment in NSW Child Care Centres introduced in New South Wales while the Public Health Act underwent scheduled review process 2016 ACIR expanded to become the Australian Immunisation Register (AIR), capturing all vaccines administered throughout a person s life (birth to death), given through general practice and community clinics Legislation in the Australian Capital Territory amended to allow appropriately trained pharmacists to administer influenza vaccine to people aged over 18 years without a prescription Medicines, Poisons and Therapeutic Good Regulation 2008, s.352 Legislation in Queensland amended to allow appropriately trained pharmacists to administer influenza, measles and pertussis vaccines to people aged 18 years or over in a community pharmacy setting Health (Drugs and Poisons) Regulation 1996 Legislation in Tasmania amended to allow appropriately trained pharmacists to administer influenza vaccine to people aged over 18 years in an approved setting Tasmanian Poisons Regulations 2008 (S.R. 2008, No. 162) Legislation in Victoria amended to allow an approved pharmacist to administer influenza and pertussis vaccines to eligible people aged 18 years or over Secretary Approval for Pharmacist Immunisers s.140. New legislated immunisation requirements introduced in Victoria for enrolment into all early childhood education and care services conscientious objection no longer an exemption
6 10th edition of The Australian Immunisation Handbook updated and endorsed by the NHMRC Second Western Australia Immunisation Strategy ( ) developed 2017 Legislation in the Australian Capital Territory amended to allow appropriately trained pharmacists to administer the diphtheria-tetanus-acellular pertussis (dtpa) vaccine to people aged over 18 years without a prescription Medicines, Poisons and Therapeutic Good Regulation 2008, s.352 NIP expanded to provide free catch-up vaccination for all people aged years and refugees and humanitarian entrants aged 20 years or over * Information relating to disease-specific policies and programs (e.g. school-based programs) can be found in the disease-specific history tables at
Early Learning Centre Immunisation Policy Legislation ACT Public Health Regulations (2000)
 Early Learning Centre Immunisation Policy Legislation ACT Public Health Regulations (2000) National Quality Standard / Education and Care Services National Regulations Standard 2.1 Each child s health
Early Learning Centre Immunisation Policy Legislation ACT Public Health Regulations (2000) National Quality Standard / Education and Care Services National Regulations Standard 2.1 Each child s health
No Jab, No Pay New Immunisation Requirements for Family Assistance Payments
 No Jab, No Pay New Immunisation Requirements for Family Assistance Payments SUMMARY From 1 January 2016: Only parents of children (less than 20 years of age) who are fully immunised or are on a recognised
No Jab, No Pay New Immunisation Requirements for Family Assistance Payments SUMMARY From 1 January 2016: Only parents of children (less than 20 years of age) who are fully immunised or are on a recognised
IMMUNISATION POLICY. Explanation: It is imperative that children are kept safe and healthy at all times in the centre environment.
 IMMUNISATION POLICY Aim: Immunisation is a simple, safe and effective way of protecting people against harmful diseases before they come into contact with them in the community. Immunisation not only protects
IMMUNISATION POLICY Aim: Immunisation is a simple, safe and effective way of protecting people against harmful diseases before they come into contact with them in the community. Immunisation not only protects
RACGP Immunisation Position Paper
 THE ROYAL AUSTRALIAN COLLEGE OF GENERAL PRACTITIONERS RACGP Immunisation Position Paper Aim The prevention of disease through vaccination is readily available to all Australian children Principles Prevention
THE ROYAL AUSTRALIAN COLLEGE OF GENERAL PRACTITIONERS RACGP Immunisation Position Paper Aim The prevention of disease through vaccination is readily available to all Australian children Principles Prevention
Immunisation Policy. Country Children s Early Learning Ph: M:
 Immunisation Policy Published June 2018 Review Date December 2019 Sources Education and Care Services National Regulations, October 2017 Guide to the Education and Care Services National Law and the Education
Immunisation Policy Published June 2018 Review Date December 2019 Sources Education and Care Services National Regulations, October 2017 Guide to the Education and Care Services National Law and the Education
Policy Document. Vaccination Policy. Background
 Policy Document Vaccination Policy Background The Australian Medical Students Association (AMSA) is the peak representative body of Australia s medical students. AMSA believes that all communities have
Policy Document Vaccination Policy Background The Australian Medical Students Association (AMSA) is the peak representative body of Australia s medical students. AMSA believes that all communities have
PHN - Western Victoria Immunisation Forum Chelsea Taylor Senior Policy Advisor Immunisation, DHHS 21 October 2017
 PHN - Western Victoria Immunisation Forum 2017 Chelsea Taylor Senior Policy Advisor Immunisation, DHHS 21 October 2017 Australian Immunisation Register (AIR) quarterly immunisation coverage data Victoria,
PHN - Western Victoria Immunisation Forum 2017 Chelsea Taylor Senior Policy Advisor Immunisation, DHHS 21 October 2017 Australian Immunisation Register (AIR) quarterly immunisation coverage data Victoria,
PHN - Western Victoria Immunisation Forum Stephen Pellissier Manager Immunisation, DHHS 29 July 2017
 PHN - Western Victoria Immunisation Forum 2017 Stephen Pellissier Manager Immunisation, DHHS 29 July 2017 Herding Cats 2016-17 Again a huge increase in political and media focus on immunisation and vaccination
PHN - Western Victoria Immunisation Forum 2017 Stephen Pellissier Manager Immunisation, DHHS 29 July 2017 Herding Cats 2016-17 Again a huge increase in political and media focus on immunisation and vaccination
Australian Technical Advisory Group on Immunisation Then and Now. Professor Terry Nolan AO
 Australian Technical Advisory Group on Immunisation Then and Now Professor Terry Nolan AO Declarations See World Health Organisation SAGE website http://www.who.int/immunization/sage/meetings/2017/october/sa
Australian Technical Advisory Group on Immunisation Then and Now Professor Terry Nolan AO Declarations See World Health Organisation SAGE website http://www.who.int/immunization/sage/meetings/2017/october/sa
General Practice Nurses
 General Practice Nurses Annual Update 2016 Prepared by Kym Bush Immunisation Coordinator Public Health Unit April 2016 Topics to be covered Vaccine preventable diseases update Current schedule and changes
General Practice Nurses Annual Update 2016 Prepared by Kym Bush Immunisation Coordinator Public Health Unit April 2016 Topics to be covered Vaccine preventable diseases update Current schedule and changes
A PERTUSSIS EPIDEMIC IN NSW: HOW EPIDEMIOLOGY REFLECTS VACCINATION POLICY
 A PERTUSSIS EPIDEMIC IN NSW: HOW EPIDEMIOLOGY REFLECTS VACCINATION POLICY Julia Brotherton and Jeremy McAnulty Communicable Diseases Branch NSW Department of Health Pertussis has traditionally been considered
A PERTUSSIS EPIDEMIC IN NSW: HOW EPIDEMIOLOGY REFLECTS VACCINATION POLICY Julia Brotherton and Jeremy McAnulty Communicable Diseases Branch NSW Department of Health Pertussis has traditionally been considered
A WORD FROM SIR GUSTAV NOSSAL ABOUT THE NCIRS. Our Vision. Our Purpose To lead and support collaborative research,
 1997 217 ABOUT THE NCIRS A WORD FROM SIR GUSTAV NOSSAL Our Vision To be a world leader in translational research optimising the population health benefits of immunisation The National Centre for Immunisation
1997 217 ABOUT THE NCIRS A WORD FROM SIR GUSTAV NOSSAL Our Vision To be a world leader in translational research optimising the population health benefits of immunisation The National Centre for Immunisation
Victorian immunisation catch-up tool for 10 to 19 year olds
 Victorian immunisation catch-up tool for 10 to 19 year olds January 2018 Guidelines for 10 to 19 year olds with no documented history of vaccine administration The objective of catch-up vaccination is
Victorian immunisation catch-up tool for 10 to 19 year olds January 2018 Guidelines for 10 to 19 year olds with no documented history of vaccine administration The objective of catch-up vaccination is
The Australian Immunisation Register. Presented by Tracie Hibbard and Lauren Patron
 The Australian Immunisation Register Presented by Tracie Hibbard and Lauren Patron 21 November 2018 Page 2 The Australian Immunisation Register The AIR is a national register that records all vaccinations
The Australian Immunisation Register Presented by Tracie Hibbard and Lauren Patron 21 November 2018 Page 2 The Australian Immunisation Register The AIR is a national register that records all vaccinations
INFORMATION FOR RESEARCHERS REQUESTING DATA FROM THE NHVPR
 INFORMATION FOR RESEARCHERS REQUESTING DATA FROM THE NHVPR What is the NHVPR? The National Human Papillomavirus Vaccination Program Register (NHVPR) is the Australian register which records HPV vaccine
INFORMATION FOR RESEARCHERS REQUESTING DATA FROM THE NHVPR What is the NHVPR? The National Human Papillomavirus Vaccination Program Register (NHVPR) is the Australian register which records HPV vaccine
The Australian Immunisation Handbook, 10th Edition, 2013 What s new?
 The Australian Immunisation Handbook, 10th Edition, 2013 What s new? This slide set has been developed by the staff of the NCIRS for education and teaching purposes Last updated: 8 April 2013 1 Presentation
The Australian Immunisation Handbook, 10th Edition, 2013 What s new? This slide set has been developed by the staff of the NCIRS for education and teaching purposes Last updated: 8 April 2013 1 Presentation
OVERVIEW OF THE NATIONAL CHILDHOOD IMMUNISATION PROGRAMME IN SINGAPORE
 OVERVIEW OF THE NATIONAL CHILDHOOD IMMUNISATION PROGRAMME IN SINGAPORE Dr Tiong Wei Wei, MD, MPH Senior Assistant Director Policy and Control Branch, Communicable Diseases Division Ministry of Health 9
OVERVIEW OF THE NATIONAL CHILDHOOD IMMUNISATION PROGRAMME IN SINGAPORE Dr Tiong Wei Wei, MD, MPH Senior Assistant Director Policy and Control Branch, Communicable Diseases Division Ministry of Health 9
Current Immunisation Issues in the Nepean Blue Mountains Presented as part of the Immunisation Update education series in October 2017
 Current Immunisation Issues in the Nepean Blue Mountains 2017 Presented as part of the Immunisation Update education series in October 2017 Additional funded vaccines Catch up vaccines for all individuals
Current Immunisation Issues in the Nepean Blue Mountains 2017 Presented as part of the Immunisation Update education series in October 2017 Additional funded vaccines Catch up vaccines for all individuals
NATIONAL PARTNERSHIP AGREEMENT ON ESSENTIAL VACCINES
 NATIONAL PARTNERSHIP AGREEMENT ON ESSENTIAL VACCINES Council of Australian Governments Anagreement between the Commonwealth ofaustralia and the States andterritories, being: TheStateof New SouthWales TheStateof
NATIONAL PARTNERSHIP AGREEMENT ON ESSENTIAL VACCINES Council of Australian Governments Anagreement between the Commonwealth ofaustralia and the States andterritories, being: TheStateof New SouthWales TheStateof
Childhood Immunisation Strategy
 Childhood Immunisation Strategy 2015-18 1 P a g e 1. Purpose Childhood immunisation rates is one of the four National Headline Performance Indicators for PHNs. This document provides a strategy for, and
Childhood Immunisation Strategy 2015-18 1 P a g e 1. Purpose Childhood immunisation rates is one of the four National Headline Performance Indicators for PHNs. This document provides a strategy for, and
2018 Immunisation Update
 2018 Immunisation Update A puzzling time?!! Sometimes the hardest pieces of a puzzle to assemble, are the ones missing from the box. Dixie Waters, Author The Puzzle Pieces Immunisation coverage rates NSW
2018 Immunisation Update A puzzling time?!! Sometimes the hardest pieces of a puzzle to assemble, are the ones missing from the box. Dixie Waters, Author The Puzzle Pieces Immunisation coverage rates NSW
Review of the management of adverse events associated with Panvax and Fluvax. Professor John Horvath ao mbbs fracp
 Review of the management of adverse events associated with Panvax and Fluvax Professor John Horvath ao mbbs fracp Final Report 10 March 2011 Review of the management of adverse events associated with Panvax
Review of the management of adverse events associated with Panvax and Fluvax Professor John Horvath ao mbbs fracp Final Report 10 March 2011 Review of the management of adverse events associated with Panvax
Measles is a highly infectious viral
 YOUTH Challenges in managing a school-based measles outbreak in Melbourne, Australia, 2014 Katherine B. Gibney, 1,2 Aicha Brahmi, 1 Miriam O Hara, 1 Rosemary Morey, 1 Lucinda Franklin 1 Measles is a highly
YOUTH Challenges in managing a school-based measles outbreak in Melbourne, Australia, 2014 Katherine B. Gibney, 1,2 Aicha Brahmi, 1 Miriam O Hara, 1 Rosemary Morey, 1 Lucinda Franklin 1 Measles is a highly
Victorian immunisation catch-up tool for 10 to 19 year olds
 Victorian immunisation catch-up tool for 10 to 19 year olds For healthy children with no documented history of vaccine administration The objective of catch-up vaccination is to complete a course of age
Victorian immunisation catch-up tool for 10 to 19 year olds For healthy children with no documented history of vaccine administration The objective of catch-up vaccination is to complete a course of age
Monitoring vaccine-preventable diseases is
 New South Wales annual vaccinepreventable disease report, 2013 Surveillance Report Alexander Rosewell, a Paula Spokes a and Robin Gilmour a Correspondence to Robin Gilmour (e-mail: rgilm@doh.health.nsw.gov.au).
New South Wales annual vaccinepreventable disease report, 2013 Surveillance Report Alexander Rosewell, a Paula Spokes a and Robin Gilmour a Correspondence to Robin Gilmour (e-mail: rgilm@doh.health.nsw.gov.au).
Update of Childhood Vaccination on the Gold Coast
 Update of Childhood Vaccination on the Gold Coast 1 Percentage IMMUNISATION GPII Division % Coverage Rates 88.5 88 87.5 87 Preliminary Recalc 86.5 86 Feb-09 May-09 Aug-09 Nov-09 Quarter Percentage IMMUNISATION
Update of Childhood Vaccination on the Gold Coast 1 Percentage IMMUNISATION GPII Division % Coverage Rates 88.5 88 87.5 87 Preliminary Recalc 86.5 86 Feb-09 May-09 Aug-09 Nov-09 Quarter Percentage IMMUNISATION
IMMUNISATION POLICY AND HEALTH RELATED EXCLUSION POLICY
 Issue Date Version Amendment Quality Area September 2016 1.1 Policy Reviewed Approved by (Name, Position Title) Next Revision Date Mandatory QA2 Committee of Management October 2018 Introduction When groups
Issue Date Version Amendment Quality Area September 2016 1.1 Policy Reviewed Approved by (Name, Position Title) Next Revision Date Mandatory QA2 Committee of Management October 2018 Introduction When groups
Primary Health Care forum August 2015 Sue Casey
 Primary Health Care forum August 2015 Sue Casey A needs analysis of catch-up immunisation in refugee background and asylum seeker communities in Victoria Authors: Dr Georgia Paxton Dr Hamish Graham Peter
Primary Health Care forum August 2015 Sue Casey A needs analysis of catch-up immunisation in refugee background and asylum seeker communities in Victoria Authors: Dr Georgia Paxton Dr Hamish Graham Peter
Stable coverage with persistent gap. Talk Outline. Vaccine Acceptance spectrum
 Declaration of conflicts of interest HealthEd Immunisation Update: No Jab No Pay the solution to improving immunisation uptake? A/Prof Peter Richmond UWA School of Paediatrics and Child Health, Princess
Declaration of conflicts of interest HealthEd Immunisation Update: No Jab No Pay the solution to improving immunisation uptake? A/Prof Peter Richmond UWA School of Paediatrics and Child Health, Princess
Welcome to the National Indigenous Immunisation message stick (NIIMS) January 2014 A vaccine preventable diseases newsletter for health services
 Welcome to the National Indigenous Immunisation message stick (NIIMS) January 2014 A vaccine preventable diseases newsletter for health services Wishing you all a wonderful New Year What s new in Indigenous
Welcome to the National Indigenous Immunisation message stick (NIIMS) January 2014 A vaccine preventable diseases newsletter for health services Wishing you all a wonderful New Year What s new in Indigenous
standards for vaccinators and guidelines for organisations offering immunisation services
 standards for vaccinators and guidelines for organisations offering immunisation services A3.1 Purpose The Immunisation standards for vaccinators (see section A3.3) are quality levels all vaccinators should
standards for vaccinators and guidelines for organisations offering immunisation services A3.1 Purpose The Immunisation standards for vaccinators (see section A3.3) are quality levels all vaccinators should
up to CPD Credits GROUP 3 Immunisation Health promotion & professional services workbook Print Post approved PP
 up to 22 CPD Credits GROUP 3 Immunisation Health promotion & professional services workbook Print Post approved PP 100009758 Pharmaceutical Society of Australia Ltd., 2013 This handbook contains material
up to 22 CPD Credits GROUP 3 Immunisation Health promotion & professional services workbook Print Post approved PP 100009758 Pharmaceutical Society of Australia Ltd., 2013 This handbook contains material
GSK Medicine: Study No.: Title: Rationale: Objectives: Indication: Study Investigators/ Centres: Research Methods: Data Source Study Design
 GSK Medicine: Rotarix (HRV1): GlaxoSmithKline (GSK) Biologicals oral live attenuated human rotavirus vaccine Study No.: 114910 (EPI-ROTA-025 VE AU DB) Title: A study on the impact of rotavirus vaccination
GSK Medicine: Rotarix (HRV1): GlaxoSmithKline (GSK) Biologicals oral live attenuated human rotavirus vaccine Study No.: 114910 (EPI-ROTA-025 VE AU DB) Title: A study on the impact of rotavirus vaccination
UNSCHEDULED VACCINATION OF CHILDREN AND YOUNG PEOPLE WHO HAVE OUTSTANDING ROUTINE IMMUNISATIONS. Service Specification
 UNSCHEDULED VACCINATION OF CHILDREN AND YOUNG PEOPLE WHO HAVE OUTSTANDING ROUTINE IMMUNISATIONS Service Specification National Enhanced Service Specification For The Unscheduled Vaccination Of Children
UNSCHEDULED VACCINATION OF CHILDREN AND YOUNG PEOPLE WHO HAVE OUTSTANDING ROUTINE IMMUNISATIONS Service Specification National Enhanced Service Specification For The Unscheduled Vaccination Of Children
Maternal immunisation and infant immunisation: core business for GPs What s new in 2017?
 Maternal immunisation and infant immunisation: core business for GPs What s new in 2017? Peter McIntyre Director, National Centre for Immunisation Research & Surveillance Senior Staff Specialist, Children
Maternal immunisation and infant immunisation: core business for GPs What s new in 2017? Peter McIntyre Director, National Centre for Immunisation Research & Surveillance Senior Staff Specialist, Children
Seventh-day Adventist Schools (South Queensland) Limited
 Seventh-day Adventist Schools (South Queensland) Limited Document Name: Immunisation Issue Date: 10 February 2016 Vaccination Table of Contents 1. Scope... 2 2. Purpose... 2 3. Policy... 2 3.1.1 Children...
Seventh-day Adventist Schools (South Queensland) Limited Document Name: Immunisation Issue Date: 10 February 2016 Vaccination Table of Contents 1. Scope... 2 2. Purpose... 2 3. Policy... 2 3.1.1 Children...
Updated: 26 August 2010
 Australian Technical Advisory Group on Immunisation (ATAGI) Updated advice on the use of pandemic and seasonal influenza vaccines in children
Australian Technical Advisory Group on Immunisation (ATAGI) Updated advice on the use of pandemic and seasonal influenza vaccines in children
Immunisation coverage How much do we need? How do we get there?
 Immunisation coverage How much do we need? How do we get there? Peter McIntyre Director, NCIRS Julie Leask Associate Professor University of Sydney and ghost writer www.ncirs.usyd.edu.au www.ncirs.usyd.edu.au
Immunisation coverage How much do we need? How do we get there? Peter McIntyre Director, NCIRS Julie Leask Associate Professor University of Sydney and ghost writer www.ncirs.usyd.edu.au www.ncirs.usyd.edu.au
Refer to the PCS Clinical Audit Tool Quick Reference Guide for installation instructions and an overview of CAT features.
 Getting Started Installation Refer to the PCS Clinical Audit Tool Quick Reference Guide for installation instructions and an overview of CAT features. CAT Immunisations > Childhood Schedule Graph and Worksheet
Getting Started Installation Refer to the PCS Clinical Audit Tool Quick Reference Guide for installation instructions and an overview of CAT features. CAT Immunisations > Childhood Schedule Graph and Worksheet
Q21. Where can I find more information on meningococcal disease and vaccines?
 FREQUENTLY ASKED QUESTIONS This fact sheet provides responses to some common questions about meningococcal vaccines, focusing on quadrivalent meningococcal conjugate vaccines (4vMenCVs). More detailed
FREQUENTLY ASKED QUESTIONS This fact sheet provides responses to some common questions about meningococcal vaccines, focusing on quadrivalent meningococcal conjugate vaccines (4vMenCVs). More detailed
Immunization Report Public Health September 2013
 Immunization Report Public Health September 2013 Daycare, school entry and school program immunization enrollment rates, up to 2012 Table of Contents 1. Introduction... 2 2. Data Source... 2 3. Limitations...
Immunization Report Public Health September 2013 Daycare, school entry and school program immunization enrollment rates, up to 2012 Table of Contents 1. Introduction... 2 2. Data Source... 2 3. Limitations...
Questions & Answers 1 November 2005 Vaccine Schedule
 Questions & Answers 1 November 2005 Vaccine Schedule General Questions Q. When can clinics order the new vaccine? A. From early October 2005. Orders will be placed on back order & stock will be sent out
Questions & Answers 1 November 2005 Vaccine Schedule General Questions Q. When can clinics order the new vaccine? A. From early October 2005. Orders will be placed on back order & stock will be sent out
Purpose To provide a policy through which children and staff are protected against harmful infection diseases.
 Dealing with Infectious Disease and Immunisation. Purpose To provide a policy through which children and staff are protected against harmful infection diseases. Early Years @ Phoenix Park (EY@PP) is committed
Dealing with Infectious Disease and Immunisation. Purpose To provide a policy through which children and staff are protected against harmful infection diseases. Early Years @ Phoenix Park (EY@PP) is committed
GUIDELINES FOR INFECTIOUS DISEASES POLICIES AND PROGRAMS FOR MEDICAL STUDENTS
 1 Committee of Deans of Australian Medical Schools GUIDELINES FOR INFECTIOUS DISEASES POLICIES AND PROGRAMS FOR MEDICAL STUDENTS Prepared by Dr Philip Jones, UNSW, and Danielle Brown, CDAMS Overview These
1 Committee of Deans of Australian Medical Schools GUIDELINES FOR INFECTIOUS DISEASES POLICIES AND PROGRAMS FOR MEDICAL STUDENTS Prepared by Dr Philip Jones, UNSW, and Danielle Brown, CDAMS Overview These
BUILDING TRUST, MANAGING RISK: VACCINE CONFIDENCE AND HUMAN PAPILLOMAVIRUS VACCINATION
 BUILDING TRUST, MANAGING RISK: VACCINE CONFIDENCE AND HUMAN PAPILLOMAVIRUS VACCINATION Associate Professor Kristine Macartney Deputy Director, National Centre for Immunisation Research & Surveillance www.ncirs.usyd.edu.au
BUILDING TRUST, MANAGING RISK: VACCINE CONFIDENCE AND HUMAN PAPILLOMAVIRUS VACCINATION Associate Professor Kristine Macartney Deputy Director, National Centre for Immunisation Research & Surveillance www.ncirs.usyd.edu.au
School-based HPV Immunisation Programs: The WA Experience
 School-based HPV Immunisation Programs: The WA Experience Professor Paul Effler, MD, MPH, FAFPHM Medical Coordinator, Prevention and Control Program Communicable Disease Control Directorate WA School Based
School-based HPV Immunisation Programs: The WA Experience Professor Paul Effler, MD, MPH, FAFPHM Medical Coordinator, Prevention and Control Program Communicable Disease Control Directorate WA School Based
NSW Annual Vaccine-Preventable Disease Report, 2011
 NSW Annual Vaccine-Preventable Disease Report, 211 Alexander Rosewell A,B, Paula J. Spokes A and Robin E. Gilmour A A Health Protection NSW B Corresponding author. Email: arosw@doh.health.nsw.gov.au Abstract:
NSW Annual Vaccine-Preventable Disease Report, 211 Alexander Rosewell A,B, Paula J. Spokes A and Robin E. Gilmour A A Health Protection NSW B Corresponding author. Email: arosw@doh.health.nsw.gov.au Abstract:
Rationale for recommendations
 Australian Technical Advisory Group on Immunisation (ATAGI) Statement Clinical advice for immunisation providers on resumption of the use of 2010 trivalent seasonal vaccines in children less than 5 years
Australian Technical Advisory Group on Immunisation (ATAGI) Statement Clinical advice for immunisation providers on resumption of the use of 2010 trivalent seasonal vaccines in children less than 5 years
Wherever possible it is recommended that a child receive their vaccination on a day when they will not be attending the centre..
 Policy Hierarchy link Responsible Officer Contact Officer Superseded Documents Children (Education and Care Services National Law Application) Act 2010 Education and Care Services National Regulations
Policy Hierarchy link Responsible Officer Contact Officer Superseded Documents Children (Education and Care Services National Law Application) Act 2010 Education and Care Services National Regulations
SHRS Student Requirements
 SHRS Student Requirements All students enrolled in a School of Health and Rehabilitation Sciences (SHRS) program have to comply with various program requirements. These requirements are to be satisfied
SHRS Student Requirements All students enrolled in a School of Health and Rehabilitation Sciences (SHRS) program have to comply with various program requirements. These requirements are to be satisfied
Student Verification Pack 2019
 Student Verification Pack 2019 Important Start this Now Page 1 Clinical Placements in NSW Health facilities require students to complete a verification process to ensure the safety of themselves, other
Student Verification Pack 2019 Important Start this Now Page 1 Clinical Placements in NSW Health facilities require students to complete a verification process to ensure the safety of themselves, other
Appendix An Assessment Tool to Determine the Validity of Vaccine Doses
 Appendix 4.4 - An Assessment Tool to Determine the Validity of Vaccine Doses Note: Refer to the Canadian Immunization Guide and New Brunswick (NB) immunization program directives for recommendations for
Appendix 4.4 - An Assessment Tool to Determine the Validity of Vaccine Doses Note: Refer to the Canadian Immunization Guide and New Brunswick (NB) immunization program directives for recommendations for
Enhanced immunisation schedule Victoria
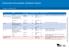 Children from March 2016 Age Disease Vaccine brand Birth Hepatitis B H-B-Vax-II Paediatric 2 months - from 6 weeks Diphtheria-tetanus-pertussis, poliomyelitis-hepatitis B- Reconstitute Site given Route
Children from March 2016 Age Disease Vaccine brand Birth Hepatitis B H-B-Vax-II Paediatric 2 months - from 6 weeks Diphtheria-tetanus-pertussis, poliomyelitis-hepatitis B- Reconstitute Site given Route
8: Applicability
 Chapter 14 New Jersey State Sanitary Code Immunization of Pupils in Schools (New Jersey Administrative Code Citation 8:57-4.1 to 8:57-4.20) (Readopted with amendments September 20, 2003. Effective Date:
Chapter 14 New Jersey State Sanitary Code Immunization of Pupils in Schools (New Jersey Administrative Code Citation 8:57-4.1 to 8:57-4.20) (Readopted with amendments September 20, 2003. Effective Date:
UICC HPV and CERVICAL CANCER CURRICULUM. UICC HPV and Cervical Cancer Curriculum Chapter 5. Application of HPV vaccines Prof. Suzanne Garland MD
 UICC HPV and CERVICAL CANCER CURRICULUM 01 Chapter 5. Application of HPV vaccines Director of Microbiological Research Director of Clinical Microbiology and Infectious Diseases The Royal Women's Hospital
UICC HPV and CERVICAL CANCER CURRICULUM 01 Chapter 5. Application of HPV vaccines Director of Microbiological Research Director of Clinical Microbiology and Infectious Diseases The Royal Women's Hospital
Manitoba Annual Immunization Surveillance Report
 Annual Immunization Surveillance Report January 1 to December 31, 2014 Epidemiology & Surveillance Public Branch Public and Primary Care Division, y Living and Seniors Released: January 2016 TABLE OF CONTENTS
Annual Immunization Surveillance Report January 1 to December 31, 2014 Epidemiology & Surveillance Public Branch Public and Primary Care Division, y Living and Seniors Released: January 2016 TABLE OF CONTENTS
Changes in hospitalisations for acute gastroenteritis in Australia after the national rotavirus vaccination program
 Changes in hospitalisations for acute gastroenteritis in Australia after the national rotavirus vaccination program Rotavirus gastroenteritis is the leading cause of severe acute gastroenteritis (AGE)
Changes in hospitalisations for acute gastroenteritis in Australia after the national rotavirus vaccination program Rotavirus gastroenteritis is the leading cause of severe acute gastroenteritis (AGE)
Directed Enhanced Service (DES) for H1N1 Vaccination Programme JCVI priority groups
 Directed Enhanced Service (DES) for H1N1 Vaccination Programme JCVI priority groups October 2009 Introduction NHS Employers and the General Practitioners Committee (GPC) of the BMA have agreed arrangements
Directed Enhanced Service (DES) for H1N1 Vaccination Programme JCVI priority groups October 2009 Introduction NHS Employers and the General Practitioners Committee (GPC) of the BMA have agreed arrangements
Immunisation records and data explained: A guide for immunisation providers
 Immunisation records and data explained: A guide for immunisation providers 2 nd edition, April 2017 Immunisation records and data explained: A guide for immunisation providers, 2 nd edition Published
Immunisation records and data explained: A guide for immunisation providers 2 nd edition, April 2017 Immunisation records and data explained: A guide for immunisation providers, 2 nd edition Published
RE-REGISTRATION FORM
 RE-REGISTRATION FORM (please print) Name of Child: Male / Female Home Phone #: street city/state/zip Date of Birth: E-mail address: Second e-mail: Mother s Social Security #: Employer s Father s Social
RE-REGISTRATION FORM (please print) Name of Child: Male / Female Home Phone #: street city/state/zip Date of Birth: E-mail address: Second e-mail: Mother s Social Security #: Employer s Father s Social
Your guide to understanding childhood immunisation
 Your guide to understanding childhood immunisation Your guide to understanding childhood immunisation ISBN: 978-1-74186-077-1 Publications approval number: 10549 Commonwealth of Australia 2013 This work
Your guide to understanding childhood immunisation Your guide to understanding childhood immunisation ISBN: 978-1-74186-077-1 Publications approval number: 10549 Commonwealth of Australia 2013 This work
immunisation in New Zealand
 This appendix details the history of. Section A1.1 is a brief summary of when each vaccine was introduced to the National Immunisation Schedule (the Schedule). This summary includes vaccines which were
This appendix details the history of. Section A1.1 is a brief summary of when each vaccine was introduced to the National Immunisation Schedule (the Schedule). This summary includes vaccines which were
Health benefits versus intussusception risk of rotavirus vaccination in Australia
 Health benefits versus intussusception risk of rotavirus vaccination in Australia Julie Bines 1 On behalf of co-authors: John Carlin 1, Kristine Macartney 2, Katherine Lee 1, Helen Quinn 2, Jim Buttery
Health benefits versus intussusception risk of rotavirus vaccination in Australia Julie Bines 1 On behalf of co-authors: John Carlin 1, Kristine Macartney 2, Katherine Lee 1, Helen Quinn 2, Jim Buttery
Dear Parents. To obtain your child s immunisation history you can:
 Dear Parents The Public Health Act require schools and child care centres to keep information about students immunisation records with respect to diphtheria, pertussis (whooping cough), polio, haemophilus
Dear Parents The Public Health Act require schools and child care centres to keep information about students immunisation records with respect to diphtheria, pertussis (whooping cough), polio, haemophilus
NHS Sheffield Community Pharmacy Catch Up Seasonal Flu Vaccination Programme for hard to reach at risk groups
 NHS Sheffield Community Pharmacy Catch Up Seasonal Flu Vaccination Programme for hard to reach at risk groups 2011-12 Service Evaluation Supported by Sheffield Local Pharmaceutical Committee Supporting
NHS Sheffield Community Pharmacy Catch Up Seasonal Flu Vaccination Programme for hard to reach at risk groups 2011-12 Service Evaluation Supported by Sheffield Local Pharmaceutical Committee Supporting
Director of Public Health Board Paper No. 13/13
 Greater Glasgow and Clyde NHS Board Director of Public Health Board Paper No. 13/13 Report of the Director of Public Health : Major Development to Immunisation Programmes in Scotland Implications for NHSGGC
Greater Glasgow and Clyde NHS Board Director of Public Health Board Paper No. 13/13 Report of the Director of Public Health : Major Development to Immunisation Programmes in Scotland Implications for NHSGGC
IMMUNISATION: A PUBLIC HEALTH SUCCESS CONTENTS
 NSW Public Health Bulletin IMMUNISATION: A PUBLIC HEALTH SUCCESS GUEST EDITORIAL Margaret Burgess National Centre for Immunisation Research The University of Sydney and The Children s Hospital at Westmead
NSW Public Health Bulletin IMMUNISATION: A PUBLIC HEALTH SUCCESS GUEST EDITORIAL Margaret Burgess National Centre for Immunisation Research The University of Sydney and The Children s Hospital at Westmead
2016/17 Vaccination and Immunisation list of additional services and enhanced services
 2016/17 Vaccination and Immunisation list of additional services and enhanced services 2016/17 Vaccination and Immunisation list of additional services and enhanced services Version number: 1 First published:
2016/17 Vaccination and Immunisation list of additional services and enhanced services 2016/17 Vaccination and Immunisation list of additional services and enhanced services Version number: 1 First published:
Non-judgmental conversations with parents worried about vaccines side-effects. Dr Jenny Royle
 Non-judgmental conversations with parents worried about vaccines side-effects Nov 3 rd, 2017 Dr Jenny Royle NEST Family Clinic, Elsternwick Multicentre NHMRC Centre of Research Excellence Immunisation
Non-judgmental conversations with parents worried about vaccines side-effects Nov 3 rd, 2017 Dr Jenny Royle NEST Family Clinic, Elsternwick Multicentre NHMRC Centre of Research Excellence Immunisation
The English immunization programme
 The English immunization programme Mary Ramsay Head of Immunisation Public Health England National Health Service Constitution (2009) You have the right to receive the vaccinations that the Joint Committee
The English immunization programme Mary Ramsay Head of Immunisation Public Health England National Health Service Constitution (2009) You have the right to receive the vaccinations that the Joint Committee
Interstate dispensing : A case for uniform, intuitive legislation
 Interstate dispensing : A case for uniform, intuitive legislation Author Bernaitis, Nijole, King, Michelle, Hope, Denise Published 2014 Journal Title Journal of Law and Medicine Copyright Statement 2014
Interstate dispensing : A case for uniform, intuitive legislation Author Bernaitis, Nijole, King, Michelle, Hope, Denise Published 2014 Journal Title Journal of Law and Medicine Copyright Statement 2014
Student Immunisation Record Faculty of Medicine. Section 1: Information. Notes
 Student Immunisation Record Faculty of Section 1: Information Students enrolled in programs offered by the Faculty of are REQUIRED to provide evidence of their immunisation status for the diseases listed
Student Immunisation Record Faculty of Section 1: Information Students enrolled in programs offered by the Faculty of are REQUIRED to provide evidence of their immunisation status for the diseases listed
Submission to the Senate Community Affairs Legislation Committee inquiry into the Therapeutic Goods Amendment (2016 Measures No.
 Submission to the Senate Community Affairs Legislation Committee inquiry into the Therapeutic Goods Amendment (2016 Measures No. 1) Bill 2016 Submitted by: Submitted electronically to: Complementary Medicines
Submission to the Senate Community Affairs Legislation Committee inquiry into the Therapeutic Goods Amendment (2016 Measures No. 1) Bill 2016 Submitted by: Submitted electronically to: Complementary Medicines
Immunization Requirements for School Entry - Ohio
 Immunization Requirements for School Entry - Ohio Kindergarten through 12 th Grade Andrew Heffron Cuyahoga County Board of Health This information will help your school better understand Immunization entry
Immunization Requirements for School Entry - Ohio Kindergarten through 12 th Grade Andrew Heffron Cuyahoga County Board of Health This information will help your school better understand Immunization entry
Nursing and Midwifery students only. Section 1: Information
 Nursing and Midwifery students only. Section 1: Information Students enrolled in programs offered by our School are REQUIRED to provide evidence of their immunisation status for the diseases listed in
Nursing and Midwifery students only. Section 1: Information Students enrolled in programs offered by our School are REQUIRED to provide evidence of their immunisation status for the diseases listed in
Vaccine preventable diseases and vaccination coverage in Australia, 1999 to 2000
 Vaccine preventable diseases and vaccination coverage in Australia, 1999 to 2000 This report was written at the National Centre for Immunisation Research and Surveillance of Vaccine Preventable Diseases
Vaccine preventable diseases and vaccination coverage in Australia, 1999 to 2000 This report was written at the National Centre for Immunisation Research and Surveillance of Vaccine Preventable Diseases
Virginia Health Value Dashboard. March 2019
 Virginia Health Value Dashboard March 2019 WHO WE ARE In Virginia, the major community partners engaged in health reform -- health care providers, health systems, health plans, pharmaceutical manufacturers
Virginia Health Value Dashboard March 2019 WHO WE ARE In Virginia, the major community partners engaged in health reform -- health care providers, health systems, health plans, pharmaceutical manufacturers
Enhanced service specification Childhood seasonal influenza vaccination programme NHS England gateway reference: 01641
 Enhanced service specification Childhood seasonal influenza vaccination programme NHS England gateway reference: 01641 Introduction 1. All GMS practices are expected to provide essential and those additional
Enhanced service specification Childhood seasonal influenza vaccination programme NHS England gateway reference: 01641 Introduction 1. All GMS practices are expected to provide essential and those additional
Vaccine Finance. Overview of stakeholder input and NVAC working group draft white paper. Walt Orenstein, MD
 Vaccine Finance Overview of stakeholder input and NVAC working group draft white paper Walt Orenstein, MD Consultant to the National Vaccine Program Office July 24, 2008 Number of Vaccines in the Routine
Vaccine Finance Overview of stakeholder input and NVAC working group draft white paper Walt Orenstein, MD Consultant to the National Vaccine Program Office July 24, 2008 Number of Vaccines in the Routine
Dreamers Child Care Enrollment Application
 Dreamers Child Care Enrollment Application Child s Full Name Gender Birth Date Address Home Phone Chronic Physical Problems / Pertinent Developmental Information / Special Accommodations Needed Previous
Dreamers Child Care Enrollment Application Child s Full Name Gender Birth Date Address Home Phone Chronic Physical Problems / Pertinent Developmental Information / Special Accommodations Needed Previous
Adult Pneumococcal Disease
 Adult Pneumococcal Disease Reviewed and updated July 2018 S. pneumoniae and pneumococcal disease The bacterium Streptococcus pneumoniae causes pneumococcal disease S. pneumoniae is commonly found in human
Adult Pneumococcal Disease Reviewed and updated July 2018 S. pneumoniae and pneumococcal disease The bacterium Streptococcus pneumoniae causes pneumococcal disease S. pneumoniae is commonly found in human
2018/19 Immunisation programmes list of additional and enhanced services
 2018/19 Immunisation programmes list of additional and enhanced services 2018/19 Vaccination and Immunisation list of additional and enhanced services Version number: 1 First published: April 2018 Prepared
2018/19 Immunisation programmes list of additional and enhanced services 2018/19 Vaccination and Immunisation list of additional and enhanced services Version number: 1 First published: April 2018 Prepared
California Vaccines for Adults (VFA) Program - Q&A Session for 2017 Program Enrollees -
 California Vaccines for Adults (VFA) Program - Q&A Session for 2017 Program Enrollees - Immunization Branch, California Department of Public Health November 15, 2017 California Department of Public Health
California Vaccines for Adults (VFA) Program - Q&A Session for 2017 Program Enrollees - Immunization Branch, California Department of Public Health November 15, 2017 California Department of Public Health
The National Immunisation Program in the modern era providing, nudging, and cajoling
 The National Immunisation Program in the modern era providing, nudging, and cajoling Professor Terry Nolan PHAA National Immunisation Conference 2016 Plenary One: Completing the Jigsaw Declarations Research
The National Immunisation Program in the modern era providing, nudging, and cajoling Professor Terry Nolan PHAA National Immunisation Conference 2016 Plenary One: Completing the Jigsaw Declarations Research
NHS public health functions agreement Service specification No.6 Meningococcal C (MenC) containing vaccine immunisation programme
 NHS public health functions agreement 2018-19 Service specification No.6 Meningococcal C (MenC) containing vaccine immunisation programme 1 NHS public health functions agreement 2018-19 Service specification
NHS public health functions agreement 2018-19 Service specification No.6 Meningococcal C (MenC) containing vaccine immunisation programme 1 NHS public health functions agreement 2018-19 Service specification
131. Public school enrollees' immunization program; exemptions
 TITLE 14. EDUCATION PART I. FREE PUBLIC SCHOOLS CHAPTER 1. DEPARTMENT OF EDUCATION SUBCHAPTER II. POWERS AND DUTIES 14 Del. C. 131 (2007) 131. Public school enrollees' immunization program; exemptions
TITLE 14. EDUCATION PART I. FREE PUBLIC SCHOOLS CHAPTER 1. DEPARTMENT OF EDUCATION SUBCHAPTER II. POWERS AND DUTIES 14 Del. C. 131 (2007) 131. Public school enrollees' immunization program; exemptions
Submission to Health and Ambulance Services Committee Public Health (Childcare Vaccination) and Other Legislation Amendment Bill 2015
 Submission No: 037 Submission to Health and Ambulance Services Committee Public Health (Childcare Vaccination) and Other Legislation Amendment Bill 2015 A submission by: Julie Leask PhD, MPH, DipHealthSci
Submission No: 037 Submission to Health and Ambulance Services Committee Public Health (Childcare Vaccination) and Other Legislation Amendment Bill 2015 A submission by: Julie Leask PhD, MPH, DipHealthSci
Recognition of Skills and Training Q. Does the Greens support direct referrals to selected medical specialist services?
 22 September 2014 Colleen Hartland, MLC 75 Victoria Street SEDDON VIC 3011 Dear Ms Hartland, The Australian Physiotherapy Association represents more than 4,100 Victorian physiotherapists and over 16,500
22 September 2014 Colleen Hartland, MLC 75 Victoria Street SEDDON VIC 3011 Dear Ms Hartland, The Australian Physiotherapy Association represents more than 4,100 Victorian physiotherapists and over 16,500
Camden Clinical Commissioning Group
 Locally Commissioned Service Clinical Lead Commissioner Reporting Mechanism/Frequency Payment Frequency Payment Contact Childhood Immunisation Dr Oliver Anglin Camden Clinical Commissioning Group Partially
Locally Commissioned Service Clinical Lead Commissioner Reporting Mechanism/Frequency Payment Frequency Payment Contact Childhood Immunisation Dr Oliver Anglin Camden Clinical Commissioning Group Partially
2018 Immunisation Update
 2018 Immunisation Update A puzzling time?!! Sometimes the hardest pieces of a puzzle to assemble, are the ones missing from the box. Dixie Waters, Author Authority to Immunise NSW Immunisation Schedule
2018 Immunisation Update A puzzling time?!! Sometimes the hardest pieces of a puzzle to assemble, are the ones missing from the box. Dixie Waters, Author Authority to Immunise NSW Immunisation Schedule
Daycare, school entry and school program immunization report. Data for school year 2016/17
 Daycare, school entry and school program immunization report Data for school year 2016/17 Table of Contents 1. Introduction... 1 2. Data Source... 1 3. Limitations... 2 4. Daycare - Proof of Immunization...
Daycare, school entry and school program immunization report Data for school year 2016/17 Table of Contents 1. Introduction... 1 2. Data Source... 1 3. Limitations... 2 4. Daycare - Proof of Immunization...
AUSTRALIAN IMMUNISATION REGISTER - ENHANCEMENTS
 AUSTRALIAN IMMUNISATION REGISTER - ENHANCEMENTS Updates September 2016 June 2018... 1 AIR Enhancements - June 2018... 2 AIR Enhancements planned for release September 2018... 3 Using the AIR Online Secure
AUSTRALIAN IMMUNISATION REGISTER - ENHANCEMENTS Updates September 2016 June 2018... 1 AIR Enhancements - June 2018... 2 AIR Enhancements planned for release September 2018... 3 Using the AIR Online Secure
Q Can a local government elect to cease fluoridating communities that are currently fluoridated?
 Changes to the Water Fluoridation Act 2008 Frequently Asked Questions Schemes currently fluoridating Q Can a local government elect to cease fluoridating communities that are currently fluoridated? A Yes
Changes to the Water Fluoridation Act 2008 Frequently Asked Questions Schemes currently fluoridating Q Can a local government elect to cease fluoridating communities that are currently fluoridated? A Yes
INFECTIOUS DISEASES PROCEDURE
 INFECTIOUS DISEASES PROCEDURE Policy Hierarchy link Responsible Officer Contact Officer Superseded Documents Associated Documents Children (Education and Care Services National Law Application) Act 2010
INFECTIOUS DISEASES PROCEDURE Policy Hierarchy link Responsible Officer Contact Officer Superseded Documents Associated Documents Children (Education and Care Services National Law Application) Act 2010
NHS Sheffield Community Pharmacy Seasonal Flu Vaccination Programme for hard to reach at risk groups (and catch up campaign for over 65s)
 NHS Sheffield Community Pharmacy Seasonal Flu Vaccination Programme for hard to reach at risk groups 2012-13 (and catch up campaign for over 65s) Service Evaluation! Supported by Sheffield!Local!Pharmaceutical!Committee!
NHS Sheffield Community Pharmacy Seasonal Flu Vaccination Programme for hard to reach at risk groups 2012-13 (and catch up campaign for over 65s) Service Evaluation! Supported by Sheffield!Local!Pharmaceutical!Committee!
BCG vaccine and tuberculosis
 PART 2: Vaccination for special risk groups 2.1 Vaccination for Aboriginal and Torres Strait Islander people Aboriginal and Torres Strait Islander people historically had a very high burden of infectious
PART 2: Vaccination for special risk groups 2.1 Vaccination for Aboriginal and Torres Strait Islander people Aboriginal and Torres Strait Islander people historically had a very high burden of infectious
Daycare, school entry and school program immunization report. Data for school year 2015/16
 Daycare, school entry and school program immunization report Data for school year 2015/16 Table of Contents Table of Content2 1. Introduction... 1 2. Data Source... 1 3. Limitations... 2 4. Daycare - Proof
Daycare, school entry and school program immunization report Data for school year 2015/16 Table of Contents Table of Content2 1. Introduction... 1 2. Data Source... 1 3. Limitations... 2 4. Daycare - Proof
Classification: official 1
 NHS public health functions agreement 2018-19 Service specification No.4 Immunisation against diphtheria, tetanus, poliomyelitis, pertussis, Hib and HepB programme 1 NHS public health functions agreement
NHS public health functions agreement 2018-19 Service specification No.4 Immunisation against diphtheria, tetanus, poliomyelitis, pertussis, Hib and HepB programme 1 NHS public health functions agreement
WHO WE ARE WHAT WE DO
 WHO WE ARE In Virginia, the major community partners engaged in health reform -- health care providers, health systems, health plans, pharmaceutical manufacturers and laboratory companies, employers, consumers,
WHO WE ARE In Virginia, the major community partners engaged in health reform -- health care providers, health systems, health plans, pharmaceutical manufacturers and laboratory companies, employers, consumers,
