Q21. Where can I find more information on meningococcal disease and vaccines?
|
|
|
- Christal Bridges
- 6 years ago
- Views:
Transcription
1 FREQUENTLY ASKED QUESTIONS This fact sheet provides responses to some common questions about meningococcal vaccines, focusing on quadrivalent meningococcal conjugate vaccines (4vMenCVs). More detailed information about meningococcal disease and the available meningococcal vaccines can be found in the NCIRS fact sheet Meningococcal vaccines for Australians and in The Australian Immunisation Handbook (Chapter 4.10 Meningococcal disease). Additional information on the use of the meningococcal B vaccine (Bexsero ) is available in the Australian Technical Advisory Group on Immunisation s advice for immunisation providers regarding the use of Bexsero. Questions about meningococcal vaccines and vaccine schedules Q1. Who should be vaccinated with quadrivalent (A, C, W, Y) meningococcal conjugate vaccines (4vMenCV)? Q2. Which 4vMenCVs are available in Australia, and are there any differences between them? Q3. How many doses of 4vMenCV are required? Q4. Do 4vMenCVs protect against all meningococcal disease? Q5. Which vaccine is more important to give, 4vMenCV or Bexsero? Q6. Is there a different vaccine schedule for people at increased risk of meningococcal disease? Q7. Are 4vMenCVs safe? Q8. Are there any contraindications to using 4vMenCV? Q9. Is 4vMenCV or MenB vaccine available for free? Q10. Can a different brand of 4vMenCV be used to complete a vaccination course (i.e. are the brands interchangeable), particularly if there are shortages in vaccine supply? Q11. If a 12-month-old child receives 4vMenCV, should they still receive Menitorix (Hib MenCCV)? Q12. Can a meningococcal vaccine be given concomitantly with other meningococcal vaccines? Q13. Can 4vMenCV be given at the same time as other non-meningococcal vaccines? Q14. For older children and adolescents (aged years) who require a catch-up dose of MenC vaccine, is vaccination with a dose of 4vMenCV sufficient? Q15. An adolescent who is currently eligible for a dose of 4vMenCV through the state-based program has received 4vMenCV or a quadrivalent polysaccharide meningococcal vaccine in the past; should they receive this dose through the current program? Q16. My patient has received 4vMenCV in the past; do they require a booster dose? Q17. My patient has been in close contact with someone who has been diagnosed with meningococcal disease. Do they require vaccination? Questions about meningococcal disease Q18. How has meningococcal disease changed recently? Q19 Does the type of meningococcal disease vary by state or territory? Q20. Which age groups are most at risk of serogroups W and Y meningococcal disease? For further information Q21. Where can I find more information on meningococcal disease and vaccines? The Australian Technical Advisory Group on Immunisation (ATAGI) is currently reviewing the use of meningococcal vaccines in Australia and will be updating the meningococcal chapter of The Australian Immunisation Handbook. Some of the information contained within this FAQ and the NCIRS fact sheet is not currently in the Handbook. Meningococcal vaccines FAQ NCIRS Fact sheet: September
2 Questions about meningococcal vaccines and vaccine schedules Q1. Who should be vaccinated with quadrivalent (A, C, W, Y) meningococcal conjugate vaccines (4vMenCV)? Any person 2 months of age or older can be vaccinated with 4vMenCV to reduce their likelihood of becoming ill with meningococcal disease caused by serogroup A, C, W or Y. Children aged under 2 years have the highest rates of meningococcal W and Y disease, followed by older adolescents (refer also to Q20). Meningococcal bacteria are carried and spread more frequently in older adolescents and young adults, so people living in close conditions, such as military recruits or those in residential accommodation, may particularly benefit from vaccination. Individuals travelling to parts of the world where serogroup A, C, W or Y disease is more common should also receive 4vMenCV, and 4vMenCV is required for pilgrims attending the annual Hajj in Mecca. Vaccination with both 4vMenCV and the meningococcal B (MenB) vaccine is recommended for people with medical conditions associated with an increased risk of meningococcal disease (refer to Q6), and laboratory personnel who frequently handle Neisseria meningitidis. Q2. Which 4vMenCVs are available in Australia, and are there any differences between them? There are three brands of 4vMenCVs available for use in Australia (Table 1). They differ in the carrier protein that the meningococcal serogroup A, C, W and Y antigens are conjugated to: Menactra (Sanofi Pasteur) diphtheria toxoid conjugate Menveo (GlaxoSmithKline) CRM 197 conjugate Nimenrix (Pfizer) tetanus toxoid conjugate. All three vaccines provide protection against meningococcal A, C, W and Y disease. However, the level of immune response that each of them induces in different age groups and to different serogroups varies to some extent. Therefore, there are differences in the dosing schedules for each vaccine. The age ranges for which they are registered for use in Australia also vary. There is currently no preferential recommendation for the use of one brand over another within age groups (Table 1). Q3. How many doses of 4vMenCV are required? The number of doses of 4vMenCV required varies by age at which vaccination commences, as shown in Table 1. The intervals given in Table 1 are the minimum intervals between doses; administering subsequent doses at longer intervals does not reduce vaccine effectiveness or necessitate repeating prior doses. Although Menveo is the only brand that is currently registered in Australia for use in those aged over 55 years, both Menactra and Nimenrix can be given to people in this age group, as per The Australian Immunisation Handbook. Given the current supply shortages of all three brands of 4vMenCVs in Australia, there may be times when a particular brand is not available; hence alternative schedules are also provided. The alternative schedules are based on clinical trials demonstrating that they induce adequate antibody responses compared with 4vMenCV schedules registered in Australia, and they are safe. These schedules are also approved by regulatory authorities in Europe or the USA. Refer to the Appendix for key references regarding these vaccination schedules. Meningococcal vaccines FAQ NCIRS Fact sheet: September
3 Table 1: Vaccination schedules for 4vMenCV by age group, for healthy individuals, travellers and laboratory personnel Age at commencement of vaccine course 4vMenCV vaccine brand Registered schedule in Australia number of doses required (and minimum intervals) 2 6 months Menveo 4 doses (8 weeks between doses; 4th dose at 12 months of age or 8 weeks after 3rd dose, whichever is later) Alternative schedule 3 doses (8 weeks between 1st and 2nd doses; 3rd dose at 12 months of age) Nimenrix N/R 3 doses (8 weeks between 1st and 2nd doses; 3rd dose at 12 months of age) Source of evidence for alternative schedule Clinical trial reports 1-3 Clinical trial report; 4 registered in Europe Menactra N/R Not suitable for use* No published clinical trial data in ages <9 months to date 7 8 months Menveo 2 doses (2nd dose at 12 months of age or 8 weeks after 1st dose, whichever is later) N/A Nimenrix N/R 3 doses (8 weeks between 1st and 2nd doses; 3rd dose at 12 months of age) Clinical trial report; 4 registered in Europe Menactra N/R Not suitable for use* No published clinical trial data in ages <9 months to date 9 11 months Menveo 2 doses (2nd dose at 12 months of age or 8 weeks after 1st dose, whichever is later) N/A Nimenrix N/R 2 doses (2nd dose at 12 months of age or 8 weeks after 1st dose, whichever is later) Clinical trial report 5 Menactra N/R 2 doses (2nd dose at 12 months of age or 12 weeks after 1st dose, whichever is later) months Menveo 2 doses (8 weeks between doses) N/A Nimenrix 1 dose N/A Menactra N/R 2 doses (12 weeks between doses) 2 years Menveo 1 dose N/A Nimenrix Menactra N/A not applicable, i.e. there is no alternative schedule available for this vaccine brand and the specified age group N/R not currently registered in Australia for use in the specified age group Clinical trial report; 6 registered in 2-dose schedule with 3 months interval in USA Clinical trial report; 6 registered in 2-dose schedule with 3 months interval in USA * There are no published clinical trials data on the use of Menactra in infants younger than 9 months of age. Given that infants typically have weaker immune responses than toddlers and older children, the clinical effectiveness of Menactra in young infants is not yet known. Therefore Menactra is not recommended in this age group. There is no registered upper age limit for use of Menveo. Although both Menactra and Nimenrix are registered for use up to 55 years of age only, either of these brands can be given to people over 55 years of age, as per The Australian Immunisation Handbook. Meningococcal vaccines FAQ NCIRS Fact sheet: September
4 Q4. Do 4vMenCVs protect against all meningococcal disease? No. 4vMenCVs (Menactra, Menveo and Nimenrix ) provide protection against four meningococcal serogroups (A, C, W and Y), but not against serogroup B. Serogroups W and Y are the two emerging serogroups in Australia (refer to Q18). There are also a number of other serogroups that are very rare in Australia and are not currently vaccine preventable. A meningococcal B vaccine (Bexsero ) is available to protect against serogroup B but may also provide some crossprotective benefit against the currently circulating strains of serogroup W (MenW). 7 This may be particularly relevant for infants (refer to Q5), in whom the rate of MenB disease has remained higher than that of MenW disease (refer also to Q20). However, vaccination with 4vMenCV is still needed to adequately protect against the A, C, W and Y serogroups. No vaccine is 100% effective, and an individual may still become infected even after immunisation. However, 4vMenCVs have demonstrated good vaccine effectiveness in populations in which they have been used. 8,9 Q5. Which vaccine is more important to give, 4vMenCV or Bexsero? Epidemiologic data show that the rate of meningococcal disease varies by age and by state and territory. In general, MenB is more common than MenW in young children, especially infants, while MenW tends to affect older age groups. However, given that the epidemiology is constantly changing, ideally both vaccines would be given. They can be administered together on the same day. As the vaccines are currently available through private prescription, financial constraints may also apply. (Refer also to Q18, Q19 and Q20.) Q6. Is there a different vaccine schedule for people at increased risk of meningococcal disease? Additional doses of 4vMenCV are recommended for individuals with certain medical conditions associated with an increased risk of meningococcal disease, including: inherited defects or deficiency of properdin or complement components current or future treatment with eculizumab functional or anatomical asplenia HIV infection haematopoetic stem cell transplant. Refer to Table in The Australian Immunisation Handbook online for details on dosing requirements for these population groups, including requirements for booster doses. MenB vaccine is also recommended for people with these conditions. For dosing guidelines, refer to Table in The Australian Immunisation Handbook online. Q7. Are 4vMenCVs safe? Meningococcal conjugate vaccines (including MenCCV and 4vMenCVs) are generally safe and well tolerated, even in children as young as 6 weeks of age. Limited data suggest no significant differences in frequency or severity of adverse events between brands of 4vMenCV. Additional information on adverse events following meningococcal vaccination can be found in Chapter 4.10 of The Australian Immunisation Handbook online. Any reports of adverse events following immunisation can be made to the Therapeutic Goods Administration (TGA) and jurisdictional adverse event surveillance systems. Further information on reporting can be found on the NCIRS website. Q8. Are there any contraindications to using 4vMenCV? The only absolute contraindications for 4vMenCV are anaphylaxis following a previous dose of the respective vaccine, or anaphylaxis following any component of the vaccine. Previous meningococcal disease, regardless of the serogroup, is not a contraindication for vaccination. 10 Meningococcal vaccines FAQ NCIRS Fact sheet: September
5 Q9. Is 4vMenCV or MenB vaccine available for free? As of July 2017, five states (New South Wales, Queensland, Tasmania, Victoria and Western Australia) have statefunded 4vMenCV vaccination programs for older adolescents via school-based immunisation and/or primary care providers. Refer to state and territory health department websites for further details (refer to Q21 for relevant links). In addition, B Part of It is a clinical research study offering the MenB vaccine to students in school years 10 through 12 in participating schools in South Australia. Eligible students will be offered the vaccine in either 2017 or South Australia does not currently have a 4vMenCV vaccination program. For other individuals aged 2 months or older, 4vMenCV and MenB vaccines are available through private prescription. Neither the 4vMenCV nor the MenB vaccine is currently free under the National Immunisation Program (NIP). Q10. Can a different brand of 4vMenCV be used to complete a vaccination course (i.e. are the brands interchangeable), particularly if there are shortages in vaccine supply? It is preferable to use the same brand of 4vMenCV when giving subsequent doses, especially for completing a primary vaccination course for young children. However, a different brand may be substituted in cases where the initial brand used is unavailable or not known. Q11. If a 12-month-old child receives 4vMenCV, should they still receive Menitorix (Hib MenCCV)? Administration of Menitorix at 12 months of age is funded under the NIP and is necessary in accordance with Australian Immunisation Register (AIR) due/overdue rules. To ensure parents remain eligible for Family Tax Benefits A and Child Care Benefits, a dose of MenC vaccine and a Hib booster dose must be given before age 13 months. As 4vMenCV provides protection against MenC as well as other meningococcal serogroups, it may be given in lieu of Menitorix to satisfy MenC vaccination requirements. However, a booster dose of Hib is also required in the second year of life, ideally before 13 months of age. Presently, monovalent Hib vaccine is in limited supply in Australia and is not available for use under the NIP; therefore, in most instances Menitorix will be required as the Hib booster dose even if 4vMenCV is given. A minimal interval of 4 weeks between 4vMenCV and Menitorix is suggested (refer to Q12). Q12. Can a meningococcal vaccine be given concomitantly with other meningococcal vaccines? 4vMenCV with Menitorix (Hib MenCCV) It is preferable that 4vMenCV is not administered concurrently with Menitorix. There is the potential for interference between the immune responses to the various conjugate carrier proteins and polysaccharide antigens in the vaccines resulting in lower antibody responses. Ideally, the vaccines should be administered at least 4 weeks apart. Either Menitorix or a 4vMenCV can be administered first. However, in circumstances where the child may not be able to return to clinic for timely administration of the other vaccine, Menitorix can be co-administered with a 4vMenCV. Bexsero (MenB vaccine) with Menitorix (Hib MenCCV) or 4vMenCV Bexsero can be co-administered with Menitorix or 4vMenCV. Prophylactic administration of paracetamol prior to administration is recommended before and after administration of Bexsero (refer to The Australian Immunisation Handbook and ATAGI s advice on the use of Bexsero ). Q13. Can 4vMenCV be given at the same time as other non-meningococcal vaccines? Yes, 4vMenCV can be administered concurrently with other routine childhood vaccinations. Clinical trials have reported some variations in antibody responses when 4vMenCVs are co-administered with either 7-valent or 10-valent pneumococcal conjugate vaccines (PCVs) (both of which are not currently used in the NIP); 6,11 however, differences are mostly minor and protective levels of antibodies are attained. Trials on co-administration of 4vMenCVs and 13-valent PCV (Prevenar 13 ) are ongoing and results will be reviewed when available. Meningococcal vaccines FAQ NCIRS Fact sheet: September
6 Q14. For older children and adolescents (aged years) who require a catch-up dose of MenC vaccine, is vaccination with a dose of 4vMenCV sufficient? Yes, vaccination with 4vMenCV provides protection against MenC disease and is accepted by the AIR as a dose of MenC vaccine. It can be given in lieu of monovalent MenC vaccines. Catch-up of the Hib booster dose is not required for children aged over 5 years. Table in The Australian Immunisation Handbook online provides further details on catch-up requirements in people aged 10 years or older, and an Australian Government Department of Health fact sheet provides information on catch-up doses available at no cost under the NIP. Q15. An adolescent who is currently eligible for a dose of 4vMenCV through the state-based program has received 4vMenCV or a quadrivalent polysaccharide meningococcal vaccine in the past; should they receive this dose through the current program? Yes. Eligible adolescents can receive a dose of 4vMenCV through a state-based program if they have been vaccinated with 4vMenCV in the past (with at least 4 weeks between doses), even if the previous dose was within the past 5 years. Repeat vaccination can offer a benefit by boosting immunity, and carries minimal risk of adverse events. Adolescents who have previously received a polysaccharide meningococcal vaccine (at any age), which would have provided protection for only a limited period, can be vaccinated with 4vMenCV now, provided that at least 6 months have passed since receiving the last dose of polysaccharide vaccine. Q16. My patient has received 4vMenCV in the past; do they require a booster dose? In healthy individuals of any age (i.e. those without risk factors for meningococcal disease, refer to Q6), the need for booster doses of 4vMenCV following vaccination with an age-appropriate number of doses has not been established. However, repeat vaccination can benefit the individual as it can boost immunity and clinical trials show no increase in adverse events with multi-dose schedules. There are countries that routinely recommend multiple doses even in older age groups; for example, 2 doses of vaccine with 4 5 years between doses is recommended for adolescents in the USA. Individuals at increased risk of meningococcal disease due to certain medical conditions (refer to Q6), laboratory personnel who handle Neisseria meningitidis and repeat travellers to areas with increased risk of meningococcal disease have ongoing risk of infection and should receive regular booster doses, as per the schedules given in Chapter 4.10 of The Australian Immunisation Handbook online. Serologic testing to determine immune status is not necessary, nor recommended. Q17. My patient has been in close contact with someone who has been diagnosed with meningococcal disease. Do they require vaccination? The relevant state or territory public health authority should be contacted as soon as possible for advice on determining the risk of disease and for guidance on management, including whether to offer clearance antibiotics or vaccination. (Refer also to the NCIRS fact sheet Meningococcal vaccines for Australians, Management of meningococcal disease.) Questions about meningococcal disease Q18. How has meningococcal disease changed recently? Nationally, from 2002 to 2015, MenB was the most common serogroup causing invasive meningococcal disease (IMD) in Australia. However, the incidence of IMD caused by MenW has increased since 2013, and more rapidly since mid (Figure 1). A smaller increase in serogroup Y (MenY) disease incidence has occurred since Approximately 20% of MenW cases had an atypical presentation. A higher case fatality rate for MenW compared with MenB disease has also been observed (about 7% for MenW versus about 4% for MenB). More information on trends in meningococcal disease can be found in the NCIRS fact sheet Meningococcal vaccines for Australians. Information about meningococcal W disease is available on the Australian Government Department of Health website. Meningococcal vaccines FAQ NCIRS Fact sheet: September
7 Figure 1: Notification rates of invasive meningococcal disease by selected serogroups and year, Australia, Q19. Does the type of meningococcal disease vary by state or territory? Yes. While MenW was the dominant strain in some states in 2016 and a growing proportion of all IMD cases compared with previous years, MenB was still the prominent serogroup causing disease in several states. However, the epidemiology of meningococcal disease is constantly changing. Information on the latest trends can be found on the Australian Government Department of Health website. State and territory government health authorities can also provide up-to-date information for your state or territory. Q20. Which age groups are most at risk of serogroups W and Y meningococcal disease? Age-specific rates of MenW IMD, while still low overall, have increased in most age groups since Children aged under 5 years (especially those under 2 years) and adolescents/young adults (aged years) have the highest rates of MenW disease. However, since its emergence in Australia, a substantial proportion of all MenW disease has also occurred in adults aged 45 years and over. There has also been an increase in MenY IMD. Table 2 shows age-specific rates of meningococcal disease in Australia in Table 2: Age-specific numbers and rates of invasive meningococcal disease (IMD) in Australia, 2016 Age group Meningococcal W and Y disease Meningococcal B disease Number of cases Rate (per 100,000) Number of cases Rate (per 100,000) <12 months months years years years years years years years Meningococcal vaccines FAQ NCIRS Fact sheet: September
8 For further information Q21. Where can I find more information on meningococcal disease and vaccines? The following links contain the latest updates on meningococcal disease epidemiology, availability of vaccines and general information on meningococcal disease: The Australian Immunisation Handbook, 10th edition NCIRS fact sheet on meningococcal disease and meningococcal vaccines for Australians Immunise Australia website National Immunisation Program Schedule Australian Government Department of Health Meningococcal W surveillance Centers for Disease Control and Prevention (USA) Meningococcal disease Contact details for Australian, state and territory government health authorities and communicable disease control (Appendix of The Australian Immunisation Handbook) tools~handbook10-appendices~handbook10-appendix1 Jurisdictional meningococcal vaccination programs (as of July 2017): o New South Wales o Queensland o Tasmania ation_program/meningococcal_w o Victoria www2.health.vic.gov.au/public-health/immunisation/vaccination-adolescents/meningococcal-acwy o Western Australia ww2.health.wa.gov.au/articles/j_m/meningococcal-acwy-statewide-vaccination-program B Part of It South Australian meningococcal B clinical study Catch-up schedule to meet No Jab, No Pay new immunisation requirements for Family Assistance Payments $File/No-Jab-No-Pay-FSheet.pdf Communicable Diseases Network Australia (CDNA) National Guidelines for Public Health Units Invasive meningococcal disease Meningococcal vaccines FAQ NCIRS Fact sheet: September
9 Appendix Several alternative dosing schedules for use of 4vMenCVs in infants and children under 2 years of age are summarised below. While these schedules are not currently registered in Australia, use of the schedules may be considered based on published clinical trials and/or registration by regulatory bodies in Europe (EMA) and the USA (FDA). Infants aged under 12 months Menactra : A trial of Menactra in 9-month old infants found the vaccine was highly immunogenic when given in a 2-dose schedule at 9 and 12 months of age. 6 There are no trials of Menactra in infants younger than 9 months of age. Nimenrix : A 3-dose schedule of Nimenrix (given at 2, 4 and 12 months of age) in infants as young as 6 weeks of age induced an antibody response as good as that with a 4 dose schedule (2, 3, 4, 12 months). 4 In infants aged 9 12 months, giving 2 doses, at 9 and 12 months of age, was found to be better than giving only 1 dose at 12 months of age. 5 Menveo : Clinical trials indicate a 3-dose schedule of Menveo (2 doses spaced 4 8 weeks apart plus a booster at 12 months of age) induces a comparable antibody response to the 4-dose schedule (3 doses spaced 8 weeks apart plus a booster at 12 months of age) currently registered for infants aged 2 6 months. 1-3,12-14 One study of a 3-dose schedule (given at 2, 4 and 12 months of age) suggests adequate immunity after the first 2 (primary course) doses with a robust booster response at 12 months. 14 Although the immune response after the primary course doses is better with 3 doses than with 2 doses, the differences are less or absent after the 12-month booster vaccination which induces a robust response. 1-3 The level of antibodies 5 years after vaccination does not differ between the two schedules. 15 Toddlers aged months Menactra : There are currently no published studies of Menactra in children aged months. However, given that Menactra has been shown to induce a good immune response using a 2-dose schedule in infants aged 9 12 months, it is likely that this 2-dose schedule would be similarly immunogenic in month olds. Menveo : In a clinical trial, 1 dose of Menveo was moderately immunogenic in children aged months. There is some variation in response according to meningococcal serogroup. The immune response was significantly improved with an additional dose given 1 month later. 16 References 1. Block SL, Shepard J, Garfield H, et al. Immunogenicity and safety of a 3- and 4-dose vaccination series of a meningococcal ACWY conjugate vaccine in infants: results of a phase 3b, randomized, open-label trial. Pediatric Infectious Disease Journal 2016;35:e Tregnaghi M, Lopez P, Stamboulian D, et al. Immunogenicity and safety of a quadrivalent meningococcal polysaccharide CRM conjugate vaccine in infants and toddlers. International Journal of Infectious Diseases 2014;26: Snape MD, Perrett KP, Ford KJ, et al. Immunogenicity of a tetravalent meningococcal glycoconjugate vaccine in infants: a randomized controlled trial. [erratum appears in JAMA Mar 16;305(11):1096]. JAMA 2008;299: Merino Arribas JM, Carmona Martinez A, Horn M, et al. Safety and immunogenicity of the quadrivalent meningococcal serogroups A, C, W and Y tetanus toxoid conjugate vaccine coadministered with routine childhood vaccines in European infants: an open, randomized trial. Pediatric Infectious Disease Journal 2017;36:e98-e Klein NP, Baine Y, Bianco V, et al. One or two doses of quadrivalent meningococcal serogroups A, C, W-135 and Y tetanus toxoid conjugate vaccine is immunogenic in 9- to 12-month-old children. Pediatric Infectious Disease Journal 2013;32: Pina LM, Bassily E, Machmer A, Hou V, Reinhardt A. Safety and immunogenicity of a quadrivalent meningococcal polysaccharide diphtheria toxoid conjugate vaccine in infants and toddlers: three multicenter phase III studies. Pediatric Infectious Disease Journal 2012;31: Ladhani SN, Giuliani MM, Biolchi A, et al. Effectiveness of meningococcal B vaccine against endemic hypervirulent Neisseria meningitidis W strain, England. Emerging Infectious Diseases 2016;22: Centers for Disease Control and Prevention, Cohn AC, MacNeil JR, et al. Prevention and control of meningococcal disease: recommendations of the Advisory Committee on Immunization Practices (ACIP). MMWR Recommendations and Reports 2013;62(RR-2):1-28. Meningococcal vaccines FAQ NCIRS Fact sheet: September
10 9. MacNeil JR, Cohn AC, Zell ER, et al. Early estimate of the effectiveness of quadrivalent meningococcal conjugate vaccine. Pediatric Infectious Disease Journal 2011;30: Australian Technical Advisory Group on Immunisation (ATAGI). The Australian immunisation handbook. 10th ed (2015 update). Canberra: Australian Government Department of Health; Ruiz-Palacios GM, Huang LM, Lin TY, et al. Immunogenicity and safety of a booster dose of the 10-valent pneumococcal Haemophilus influenzae protein D conjugate vaccine coadministered with the tetravalent meningococcal serogroups A, C, W-135 and Y tetanus toxoid conjugate vaccine in toddlers: a randomized trial. Pediatric Infectious Disease Journal 2013;32: Klein NP, Shepard J, Bedell L, Odrljin T, Dull P. Immunogenicity and safety of a quadrivalent meningococcal conjugate vaccine administered concomitantly with measles, mumps, rubella, varicella vaccine in healthy toddlers. Vaccine 2012;30: Nolan TM, Nissen MD, Naz A, et al. Immunogenicity and safety of a CRM-conjugated meningococcal ACWY vaccine administered concomitantly with routine vaccines starting at 2 months of age. Human Vaccines and Immunotherapeutics 2014;10: Perrett KP, Snape MD, Ford KJ, et al. Immunogenicity and immune memory of a nonadjuvanted quadrivalent meningococcal glycoconjugate vaccine in infants. Pediatric Infectious Disease Journal 2009;28: Khatami A, Snape MD, Davis E, et al. Persistence of the immune response at 5 years of age following infant immunisation with investigational quadrivalent MenACWY conjugate vaccine formulations. Vaccine 2012;30: Novartis Vaccines. Safety and immunogenicity of Novartis meningococcal ACWY conjugate vaccine in healthy children aged months (Study V59P7) Available from: (Accessed 23 May 2017). Meningococcal vaccines FAQ NCIRS Fact sheet: September
MENINGITIS WHAT S NEW? 3 RD NOVEMBER 2018
 MENINGITIS WHAT S NEW? 3 RD NOVEMBER 2018 CONFLICTS OF INTEREST Educational travel grant - Gilead JIM NEWCOMBE FRCPA FRACP ID PHYSICIAN, ROYAL NORTH SHORE HOSPITAL, SYDNEY CLINICAL MICROBIOLOGIST, DOUGLASS
MENINGITIS WHAT S NEW? 3 RD NOVEMBER 2018 CONFLICTS OF INTEREST Educational travel grant - Gilead JIM NEWCOMBE FRCPA FRACP ID PHYSICIAN, ROYAL NORTH SHORE HOSPITAL, SYDNEY CLINICAL MICROBIOLOGIST, DOUGLASS
BACTERIAL MENINGITIS IN CHILDREN
 Overview Bacterial meningitis in children : rare, severe, mostly preventable What should GPs advise Australian parents in 2018? Peter McIntyre Professorial Fellow, National Centre for Immunisation Research
Overview Bacterial meningitis in children : rare, severe, mostly preventable What should GPs advise Australian parents in 2018? Peter McIntyre Professorial Fellow, National Centre for Immunisation Research
Meningococci and meningococcal disease
 Australian Technical Advisory Group on Immunisation (ATAGI) Statement Advice for immunisation providers regarding the use of Bexsero a recombinant multicomponent meningococcal B vaccine (4CMenB) March
Australian Technical Advisory Group on Immunisation (ATAGI) Statement Advice for immunisation providers regarding the use of Bexsero a recombinant multicomponent meningococcal B vaccine (4CMenB) March
See Spot Run Meningococcal update 2017
 See Spot Run Meningococcal update 2017 Jim Buttery Infection and Immunity Monash Children s Hospital Monash Health Monash University SAEFVIC How serious a disease is N meningitidis? Meningococcal disease
See Spot Run Meningococcal update 2017 Jim Buttery Infection and Immunity Monash Children s Hospital Monash Health Monash University SAEFVIC How serious a disease is N meningitidis? Meningococcal disease
Maternal immunisation and infant immunisation: core business for GPs What s new in 2017?
 Maternal immunisation and infant immunisation: core business for GPs What s new in 2017? Peter McIntyre Director, National Centre for Immunisation Research & Surveillance Senior Staff Specialist, Children
Maternal immunisation and infant immunisation: core business for GPs What s new in 2017? Peter McIntyre Director, National Centre for Immunisation Research & Surveillance Senior Staff Specialist, Children
Immunization Guidelines for the Use of State Supplied Vaccine May 17, 2015
 DTaP / DT DTaP/IPV/Hep B Combination (Pediarix ) Children from 6 weeks of age up to the 7 th birthday Children from 2 months of age up to the 7th birthday: Indicated for the primary doses of DTaP, IPV,
DTaP / DT DTaP/IPV/Hep B Combination (Pediarix ) Children from 6 weeks of age up to the 7 th birthday Children from 2 months of age up to the 7th birthday: Indicated for the primary doses of DTaP, IPV,
2017 Vaccination Update
 2017 Vaccination Update NATHAN BOONSTRA, MD General Pediatrician, Blank Pediatric Clinic TODAY S OBJECTIVES Review the latest recommendations for immunizations, including HPV Meningococcal vaccines Influenza
2017 Vaccination Update NATHAN BOONSTRA, MD General Pediatrician, Blank Pediatric Clinic TODAY S OBJECTIVES Review the latest recommendations for immunizations, including HPV Meningococcal vaccines Influenza
Adult Pneumococcal Disease
 Adult Pneumococcal Disease Reviewed and updated July 2018 S. pneumoniae and pneumococcal disease The bacterium Streptococcus pneumoniae causes pneumococcal disease S. pneumoniae is commonly found in human
Adult Pneumococcal Disease Reviewed and updated July 2018 S. pneumoniae and pneumococcal disease The bacterium Streptococcus pneumoniae causes pneumococcal disease S. pneumoniae is commonly found in human
9/12/2018. Meningococcal Disease and Meningococcal Vaccine. Neisseria meningitidis. Meningococcal Disease Pathogenesis. Aerobic gram-negative bacteria
 Centers for Disease Control and Prevention National Center for Immunization and Respiratory Diseases Meningococcal Disease and Meningococcal Vaccine Adult Track Chapter 14 Photographs and images included
Centers for Disease Control and Prevention National Center for Immunization and Respiratory Diseases Meningococcal Disease and Meningococcal Vaccine Adult Track Chapter 14 Photographs and images included
Bacterial diseases caused by Streptoccus pneumoniae in children
 Bacterial diseases caused by Streptoccus pneumoniae in children Bactermia 85% Bacterial pneumonia 66% Bacterial meningitis 50% Otitis media 40% Paranasal sinusitis 40% 0% 10% 20% 30% 40% 50% 60% 70% 80%
Bacterial diseases caused by Streptoccus pneumoniae in children Bactermia 85% Bacterial pneumonia 66% Bacterial meningitis 50% Otitis media 40% Paranasal sinusitis 40% 0% 10% 20% 30% 40% 50% 60% 70% 80%
Recommended Immunization Schedules for Persons Aged 0 Through 18 Years UNITED STATES, 2016
 Recommended Immunization Schedules for Persons Aged 0 Through 18 Years UNITED STATES, 2016 This schedule includes recommendations in effect as of January 1, 2016. Any dose not administered at the recommended
Recommended Immunization Schedules for Persons Aged 0 Through 18 Years UNITED STATES, 2016 This schedule includes recommendations in effect as of January 1, 2016. Any dose not administered at the recommended
Immunization Update: New CDC Recommendations. Blaise L. Congeni M.D. 2012
 Immunization Update: New CDC Recommendations Blaise L. Congeni M.D. 2012 Polysaccharide Vaccines Vaccine Hib capsule polysaccharide PRP (polyribose ribitol phosphate) Not protective in infants
Immunization Update: New CDC Recommendations Blaise L. Congeni M.D. 2012 Polysaccharide Vaccines Vaccine Hib capsule polysaccharide PRP (polyribose ribitol phosphate) Not protective in infants
Immunization Guidelines for the Use of State Supplied Vaccine April 18, 2013
 DTaP / DT DTaP/IPV/Hep B Combination (Pediarix ) Children from 6 weeks of age up to the 7 th birthday Children from 2 months of age up to the 7th birthday: Indicated for the primary doses of DTaP, IPV,
DTaP / DT DTaP/IPV/Hep B Combination (Pediarix ) Children from 6 weeks of age up to the 7 th birthday Children from 2 months of age up to the 7th birthday: Indicated for the primary doses of DTaP, IPV,
Platforms. Adolescent Immunization Update and the 16 Year Old Platform. Advisory Committee on Immunization Practices (ACIP)
 Adolescent Immunization Update and the 16 Year Old Platform William Atkinson, MD, MPH Associate Director for Immunization Education Immunization Action Coalition Advisory Committee on Immunization Practices
Adolescent Immunization Update and the 16 Year Old Platform William Atkinson, MD, MPH Associate Director for Immunization Education Immunization Action Coalition Advisory Committee on Immunization Practices
What DO the childhood immunization footnotes reveal? Questions and answers
 What DO the childhood immunization footnotes reveal? Questions and answers Stanley E. Grogg, DO, FACOP, FAAP he Advisory Committee on Immunization Practices (ACIP) recommends the childhood vaccination
What DO the childhood immunization footnotes reveal? Questions and answers Stanley E. Grogg, DO, FACOP, FAAP he Advisory Committee on Immunization Practices (ACIP) recommends the childhood vaccination
2/16/2015 IMMUNIZATION UPDATE Kelly Ridgway, RPh February 21, Today s Overview NEW RECOMMENDATIONS
 IMMUNIZATION UPDATE 2015 Kelly Ridgway, RPh February 21, 2015 Today s Overview 1 2 3 4 5 6 Pneumococcal Vaccine Recommendations Meningococcal Vaccine Recommendations HPV Vaccine Recommendations Patient
IMMUNIZATION UPDATE 2015 Kelly Ridgway, RPh February 21, 2015 Today s Overview 1 2 3 4 5 6 Pneumococcal Vaccine Recommendations Meningococcal Vaccine Recommendations HPV Vaccine Recommendations Patient
Andrew Kroger, MD, MPH National Center for Immunization and Respiratory Diseases MCH & Immunization Conference Anchorage, AK September 28, 2010
 2010 Immunization Update Andrew Kroger, MD, MPH National Center for Immunization and Respiratory Diseases MCH & Immunization Conference Anchorage, AK September 28, 2010 Disclosures No financial conflict
2010 Immunization Update Andrew Kroger, MD, MPH National Center for Immunization and Respiratory Diseases MCH & Immunization Conference Anchorage, AK September 28, 2010 Disclosures No financial conflict
ACIP Meeting Update, New Recommendations and Pending Influenza Season
 ACIP Meeting Update, New Recommendations and Pending Influenza Season February 17 th 2011 www.immunizetexas.com ACIP Upcoming Agenda and New Recommendations ACIP (February 23-24 th 2011) Topics for meeting
ACIP Meeting Update, New Recommendations and Pending Influenza Season February 17 th 2011 www.immunizetexas.com ACIP Upcoming Agenda and New Recommendations ACIP (February 23-24 th 2011) Topics for meeting
3 rd dose. 3 rd or 4 th dose, see footnote 5. see footnote 13. for certain high-risk groups
 Figure 1. Recommended immunization schedule for persons aged 0 through 18 years 2013. (FOR THOSE WHO FALL BEHIND OR START LATE, SEE THE CATCH-UP SCHEDULE [FIGURE 2]). These recommendations must be read
Figure 1. Recommended immunization schedule for persons aged 0 through 18 years 2013. (FOR THOSE WHO FALL BEHIND OR START LATE, SEE THE CATCH-UP SCHEDULE [FIGURE 2]). These recommendations must be read
No Jab, No Pay New Immunisation Requirements for Family Assistance Payments
 No Jab, No Pay New Immunisation Requirements for Family Assistance Payments SUMMARY From 1 January 2016: Only parents of children (less than 20 years of age) who are fully immunised or are on a recognised
No Jab, No Pay New Immunisation Requirements for Family Assistance Payments SUMMARY From 1 January 2016: Only parents of children (less than 20 years of age) who are fully immunised or are on a recognised
Vaccination and prophylaxis for asplenia: Guideline for clinicians
 Vaccination and prophylaxis for asplenia: Guideline for clinicians Adults better health * better care * better value Acknowledgements The Western Australian Committee for Antimicrobials (WACA) would like
Vaccination and prophylaxis for asplenia: Guideline for clinicians Adults better health * better care * better value Acknowledgements The Western Australian Committee for Antimicrobials (WACA) would like
31/10/2017 COI. Travel Vaccination Update. Outline. General Advice. Steffen graph: travel epi. Checklist. Current nil
 Travel Vaccination Update COI Current nil Nigel Crawford HealthEd, Brisbane October 2017 Historical pharmaceutical industry funding to my research institute (MCRI) for special risk and vaccine safety projects
Travel Vaccination Update COI Current nil Nigel Crawford HealthEd, Brisbane October 2017 Historical pharmaceutical industry funding to my research institute (MCRI) for special risk and vaccine safety projects
Changes to the Meningococcal C conjugate (MenC) vaccine schedule. Questions and Answers
 Changes to the Meningococcal C conjugate (MenC) vaccine schedule Questions and Answers Background The meningococcal C (MenC) vaccination programme was first introduced into the UK routine immunisation
Changes to the Meningococcal C conjugate (MenC) vaccine schedule Questions and Answers Background The meningococcal C (MenC) vaccination programme was first introduced into the UK routine immunisation
What are the new active vaccine recommendations in the Canadian Immunization Guide?
 154 CCDR 17 April 2014 Volume 40-8 https://doi.org/10.14745/ccdr.v40i08a03 1 What are the new active vaccine recommendations in the Canadian Immunization Guide? Warshawsky B 1 and Gemmill I 2 on behalf
154 CCDR 17 April 2014 Volume 40-8 https://doi.org/10.14745/ccdr.v40i08a03 1 What are the new active vaccine recommendations in the Canadian Immunization Guide? Warshawsky B 1 and Gemmill I 2 on behalf
NOTE: The above recommendations must be read along with the footnotes of this schedule.
 Figure 1. Recommended immunization schedule for persons aged 0 through 18 years 2013. (FOR THOSE WHO FALL BEHIND OR START LATE, SEE THE CATCH-UP SCHEDULE [FIGURE 2]). These recommendations must be read
Figure 1. Recommended immunization schedule for persons aged 0 through 18 years 2013. (FOR THOSE WHO FALL BEHIND OR START LATE, SEE THE CATCH-UP SCHEDULE [FIGURE 2]). These recommendations must be read
BabyJabs Vaccines. All vaccines are mercury-free We use aluminium-free vaccines wherever possible
 BabyJabs Vaccines All vaccines are mercury-free We use aluminium-free vaccines wherever possible BabyJabs is a dedicated children s immunisation service, offering a choice of single and small combination
BabyJabs Vaccines All vaccines are mercury-free We use aluminium-free vaccines wherever possible BabyJabs is a dedicated children s immunisation service, offering a choice of single and small combination
CVU: What s new? 2 nd December 2013
 CVU: What s new? 2 nd December 2013 Dr Margie Danchin Paediatrician, Department of General Medicine, RCH Senior Research Fellow, MCRI Senior Fellow, Department of Paediatrics, The University of Melbourne
CVU: What s new? 2 nd December 2013 Dr Margie Danchin Paediatrician, Department of General Medicine, RCH Senior Research Fellow, MCRI Senior Fellow, Department of Paediatrics, The University of Melbourne
NOTE: The above recommendations must be read along with the footnotes of this schedule.
 Figure 1. Recommended immunization schedule for persons aged 0 through 18 years United States, 2014. (FOR THOSE WHO FALL BEHIND OR START LATE, SEE THE CATCH-UP SCHEDULE [FIGURE 2]). These recommendations
Figure 1. Recommended immunization schedule for persons aged 0 through 18 years United States, 2014. (FOR THOSE WHO FALL BEHIND OR START LATE, SEE THE CATCH-UP SCHEDULE [FIGURE 2]). These recommendations
VACCINES-WHAT HAS HAPPENED IN THE LAST YEAR. Mark H. Sawyer UCSD School of Medicine Rady Children s Hospital San Diego
 VACCINES-WHAT HAS HAPPENED IN THE LAST YEAR Mark H. Sawyer UCSD School of Medicine Rady Children s Hospital San Diego LOTS HAS HAPPENED AND IT CAN BE A LITTLE CONFUSING Some people can get 2 doses of HPV
VACCINES-WHAT HAS HAPPENED IN THE LAST YEAR Mark H. Sawyer UCSD School of Medicine Rady Children s Hospital San Diego LOTS HAS HAPPENED AND IT CAN BE A LITTLE CONFUSING Some people can get 2 doses of HPV
Splenectomy Vaccine Protocol PIDPIC
 Splenectomy Vaccine Protocol PIDPIC 6.24.14 Rationale Spleen clears encapsulated bacteria and infected erythrocytes Serves as one of the largest lymphoid tissues where B cells are educated against encapsulated
Splenectomy Vaccine Protocol PIDPIC 6.24.14 Rationale Spleen clears encapsulated bacteria and infected erythrocytes Serves as one of the largest lymphoid tissues where B cells are educated against encapsulated
Immunization Guidelines For the Use of State Supplied Vaccine July 1, 2011
 DTaP / DT DTaP/IPV/Hep B Combination (Pediarix ) Children from 6 weeks of age up to the 7 th birthday Children from 2 months of age up to the 7th birthday: Indicated for the primary doses of DTaP, IPV,
DTaP / DT DTaP/IPV/Hep B Combination (Pediarix ) Children from 6 weeks of age up to the 7 th birthday Children from 2 months of age up to the 7th birthday: Indicated for the primary doses of DTaP, IPV,
BCG vaccine and tuberculosis
 PART 2: Vaccination for special risk groups 2.1 Vaccination for Aboriginal and Torres Strait Islander people Aboriginal and Torres Strait Islander people historically had a very high burden of infectious
PART 2: Vaccination for special risk groups 2.1 Vaccination for Aboriginal and Torres Strait Islander people Aboriginal and Torres Strait Islander people historically had a very high burden of infectious
Introduction of a meningococcal ACWY immunisation programme for adolescents
 Introduction of a meningococcal ACWY immunisation programme for adolescents Information for registered healthcare practitioners Acknowledgement: This resource has been adapted with the permission of Public
Introduction of a meningococcal ACWY immunisation programme for adolescents Information for registered healthcare practitioners Acknowledgement: This resource has been adapted with the permission of Public
Hemagglutinin Neuraminidase
 Evolving Vaccine Guidance: Influenza, Meningococcal & HPV Vaccines H. Cody Meissner, M.D. Professor of Pediatrics Tufts University School of Medicine Maine Chapter AAP Bar Harbor, Maine October 16, 2016
Evolving Vaccine Guidance: Influenza, Meningococcal & HPV Vaccines H. Cody Meissner, M.D. Professor of Pediatrics Tufts University School of Medicine Maine Chapter AAP Bar Harbor, Maine October 16, 2016
A Licensed Combined Haemophilus influenzae Type b- Serogroups C and Y Meningococcal Conjugate Vaccine
 Infect Dis Ther (2013) 2:1 13 DOI 10.1007/s40121-013-0007-5 REVIEW A Licensed Combined Haemophilus influenzae Type b- Serogroups C and Y Meningococcal Conjugate Vaccine Kirsten P. Perrett Terry M. Nolan
Infect Dis Ther (2013) 2:1 13 DOI 10.1007/s40121-013-0007-5 REVIEW A Licensed Combined Haemophilus influenzae Type b- Serogroups C and Y Meningococcal Conjugate Vaccine Kirsten P. Perrett Terry M. Nolan
New NSW Immunisation Schedule
 New NSW Immunisation Schedule GP Update Prepared by Kym Bush Immunisation Coordinator Public Health Unit July 2018 Rationale for changes on 1 July 2018 Increase in cases of IPD in kiddies > 12 months Increase
New NSW Immunisation Schedule GP Update Prepared by Kym Bush Immunisation Coordinator Public Health Unit July 2018 Rationale for changes on 1 July 2018 Increase in cases of IPD in kiddies > 12 months Increase
Early Learning Centre Immunisation Policy Legislation ACT Public Health Regulations (2000)
 Early Learning Centre Immunisation Policy Legislation ACT Public Health Regulations (2000) National Quality Standard / Education and Care Services National Regulations Standard 2.1 Each child s health
Early Learning Centre Immunisation Policy Legislation ACT Public Health Regulations (2000) National Quality Standard / Education and Care Services National Regulations Standard 2.1 Each child s health
MEDICAL ASSISTANCE BULLETIN
 ISSUE DATE August 30, 2010 EFFECTIVE DATE August 30, 2010 MEDICAL ASSISTANCE BULLETIN NUMBER 01-10-27, 08-10-28, 09-10-29, 31-10-30, 33-10-05 SUBJECT: Updates to the Medical Assistance Program Fee Schedule
ISSUE DATE August 30, 2010 EFFECTIVE DATE August 30, 2010 MEDICAL ASSISTANCE BULLETIN NUMBER 01-10-27, 08-10-28, 09-10-29, 31-10-30, 33-10-05 SUBJECT: Updates to the Medical Assistance Program Fee Schedule
Vaccination Update. Pregnancy. 24-May-17. Disclosures. Overview. Paul Van Buynder Professor, Griffith University Chairman, Immunisation Coalition
 Vaccination Update Paul Van Buynder Professor, Griffith University Chairman, Immunisation Coalition Disclosures I have been involved in vaccine studies funded by CSL, Sanofi Pasteur, Merck and Novartis
Vaccination Update Paul Van Buynder Professor, Griffith University Chairman, Immunisation Coalition Disclosures I have been involved in vaccine studies funded by CSL, Sanofi Pasteur, Merck and Novartis
ACIP Recommendation Update. Mark H. Sawyer, MD UCSD School of Medicine Rady Children s Hospital San Diego
 ACIP Recommendation Update Mark H. Sawyer, MD UCSD School of Medicine Rady Children s Hospital San Diego What has ACIP been up to lately? New Recommendations Tdap vaccination for each pregnancy Meningococcal
ACIP Recommendation Update Mark H. Sawyer, MD UCSD School of Medicine Rady Children s Hospital San Diego What has ACIP been up to lately? New Recommendations Tdap vaccination for each pregnancy Meningococcal
Review Article Conjugate Meningococcal Vaccines Development: GSK Biologicals Experience
 SAGE-Hindawi Access to Research Advances in Preventive Medicine Volume 2011, Article ID 846756, 17 pages doi:10.4061/2011/846756 Review Article Conjugate Meningococcal Vaccines Development: GSK Biologicals
SAGE-Hindawi Access to Research Advances in Preventive Medicine Volume 2011, Article ID 846756, 17 pages doi:10.4061/2011/846756 Review Article Conjugate Meningococcal Vaccines Development: GSK Biologicals
Updated: 26 August 2010
 Australian Technical Advisory Group on Immunisation (ATAGI) Updated advice on the use of pandemic and seasonal influenza vaccines in children
Australian Technical Advisory Group on Immunisation (ATAGI) Updated advice on the use of pandemic and seasonal influenza vaccines in children
Changes to the meningococcal C conjugate (MenC) vaccine schedule 2014 first-time university entrants under the age of 25 An update for registered
 Changes to the meningococcal C conjugate (MenC) vaccine schedule 2014 first-time university entrants under the age of 25 An update for registered healthcare practitioners Frequently asked questions August
Changes to the meningococcal C conjugate (MenC) vaccine schedule 2014 first-time university entrants under the age of 25 An update for registered healthcare practitioners Frequently asked questions August
BabyJabs Vaccines. All vaccines are mercury- free We use aluminium- free vaccines wherever possible
 BabyJabs Vaccines All vaccines are mercury- free We use aluminium- free vaccines wherever possible BabyJabs is a dedicated children s immunisation service, offering a choice of single and small combination
BabyJabs Vaccines All vaccines are mercury- free We use aluminium- free vaccines wherever possible BabyJabs is a dedicated children s immunisation service, offering a choice of single and small combination
BabyJabs Vaccines. All vaccines are mercury-free We use aluminium-free vaccines wherever possible
 BabyJabs Vaccines All vaccines are mercury-free We use aluminium-free vaccines wherever possible BabyJabs is a dedicated children s immunisation service, offering a choice of single and small combination
BabyJabs Vaccines All vaccines are mercury-free We use aluminium-free vaccines wherever possible BabyJabs is a dedicated children s immunisation service, offering a choice of single and small combination
Meningococcal Conjugate (Groups A, C, Y and W-135) Vaccine Biological Page
 Meningococcal Conjugate (Groups A, C, Y and W-135) Vaccine Biological Page Section 7: Biological Product Information Standard #: 07.281 Created by: Approved by: Province-wide Immunization Program Standards
Meningococcal Conjugate (Groups A, C, Y and W-135) Vaccine Biological Page Section 7: Biological Product Information Standard #: 07.281 Created by: Approved by: Province-wide Immunization Program Standards
Vaccinations for Adults
 Case: Vaccinations for Adults Lisa Winston, MD University of California, San Francisco San Francisco General Hospital A 30-year old healthy woman comes for a routine visit. She is recently married and
Case: Vaccinations for Adults Lisa Winston, MD University of California, San Francisco San Francisco General Hospital A 30-year old healthy woman comes for a routine visit. She is recently married and
Immunisation Update for Occupational Health
 Immunisation Update for Occupational Health Dr Gayatri Amirthalingam Immunisation, Hepatitis & Blood Safety department Public Health England 29 th April 2016 Session Outline Epidemiology of vaccine preventable
Immunisation Update for Occupational Health Dr Gayatri Amirthalingam Immunisation, Hepatitis & Blood Safety department Public Health England 29 th April 2016 Session Outline Epidemiology of vaccine preventable
PEDIATRIC PHARMACOTHERAPY A Monthly Newsletter for Health Care Professionals from the University of Virginia Children s Hospital
 PEDIATRIC PHARMACOTHERAPY A Monthly Newsletter for Health Care Professionals from the University of Virginia Children s Hospital Volume 17 Number 10 October 2011 M An Update on Quadrivalent Meningococcal
PEDIATRIC PHARMACOTHERAPY A Monthly Newsletter for Health Care Professionals from the University of Virginia Children s Hospital Volume 17 Number 10 October 2011 M An Update on Quadrivalent Meningococcal
2016/17 Vaccination and Immunisation list of additional services and enhanced services
 2016/17 Vaccination and Immunisation list of additional services and enhanced services 2016/17 Vaccination and Immunisation list of additional services and enhanced services Version number: 1 First published:
2016/17 Vaccination and Immunisation list of additional services and enhanced services 2016/17 Vaccination and Immunisation list of additional services and enhanced services Version number: 1 First published:
2017/18 Immunisation programmes list of additional and enhanced services
 2017/18 Immunisation programmes list of additional and enhanced services 2017/18 Vaccination and Immunisation list of additional and enhanced services Version number: 1 First published: April 2017 Prepared
2017/18 Immunisation programmes list of additional and enhanced services 2017/18 Vaccination and Immunisation list of additional and enhanced services Version number: 1 First published: April 2017 Prepared
CONJUGATE MENINGOCOCCAL (ACWY) VACCINE
 Cumbria & Lancashire Vaccine PGD Group ADVISORY INFORMATION FOR USE WITH PATIENT SPECIFIC DIRECTION OR INDIVIDUAL PRESCRIPTION SIGNED BY GP OR NON-MEDICAL PRESCRIBER. CONJUGATE MENINGOCOCCAL (ACWY) VACCINE
Cumbria & Lancashire Vaccine PGD Group ADVISORY INFORMATION FOR USE WITH PATIENT SPECIFIC DIRECTION OR INDIVIDUAL PRESCRIPTION SIGNED BY GP OR NON-MEDICAL PRESCRIBER. CONJUGATE MENINGOCOCCAL (ACWY) VACCINE
Appendix An Assessment Tool to Determine the Validity of Vaccine Doses
 Appendix 4.4 - An Assessment Tool to Determine the Validity of Vaccine Doses Note: Refer to the Canadian Immunization Guide and New Brunswick (NB) immunization program directives for recommendations for
Appendix 4.4 - An Assessment Tool to Determine the Validity of Vaccine Doses Note: Refer to the Canadian Immunization Guide and New Brunswick (NB) immunization program directives for recommendations for
Syrian Programme Refugees Advice on assessment of immunisation status and recommendations for additional immunisation
 Syrian Programme Refugees Advice on assessment of immunisation status and recommendations for additional immunisation Background Information Immunisation services in Syria Syria had good immunisation services,
Syrian Programme Refugees Advice on assessment of immunisation status and recommendations for additional immunisation Background Information Immunisation services in Syria Syria had good immunisation services,
Victorian immunisation catch-up tool for 10 to 19 year olds
 Victorian immunisation catch-up tool for 10 to 19 year olds January 2018 Guidelines for 10 to 19 year olds with no documented history of vaccine administration The objective of catch-up vaccination is
Victorian immunisation catch-up tool for 10 to 19 year olds January 2018 Guidelines for 10 to 19 year olds with no documented history of vaccine administration The objective of catch-up vaccination is
Immunization Update & focus on meningococcal vaccine PART 2
 Immunization Update & focus on meningococcal vaccine PART 2 Gregory Hussey Vaccines for Africa Initiative Institute of Infectious Diseases University of Cape Town www.vacfa.uct.ac.za Meningococcal Disease
Immunization Update & focus on meningococcal vaccine PART 2 Gregory Hussey Vaccines for Africa Initiative Institute of Infectious Diseases University of Cape Town www.vacfa.uct.ac.za Meningococcal Disease
2018/19 Immunisation programmes list of additional and enhanced services
 2018/19 Immunisation programmes list of additional and enhanced services 2018/19 Vaccination and Immunisation list of additional and enhanced services Version number: 1 First published: April 2018 Prepared
2018/19 Immunisation programmes list of additional and enhanced services 2018/19 Vaccination and Immunisation list of additional and enhanced services Version number: 1 First published: April 2018 Prepared
Meningococcal disease and vaccination in the UK
 Meningococcal disease and vaccination in the UK Ray Borrow Professor of Vaccine Preventable Diseases, Vaccine Evaluation Unit, Health Protection Agency, Manchester Royal Infirmary, Manchester, U.K. ray.borrow@hpa.org.uk
Meningococcal disease and vaccination in the UK Ray Borrow Professor of Vaccine Preventable Diseases, Vaccine Evaluation Unit, Health Protection Agency, Manchester Royal Infirmary, Manchester, U.K. ray.borrow@hpa.org.uk
7.0 Nunavut Childhood and Adult Immunization Schedules and Catch-up Aids
 7.0 Nunavut Childhood and Adult Immunization Schedules and Catch-up Aids Contents Introduction Nunavut Recommended Childhood Immunization Schedule Nunavut Routine Adult Immunization Schedule Nunavut Immunization
7.0 Nunavut Childhood and Adult Immunization Schedules and Catch-up Aids Contents Introduction Nunavut Recommended Childhood Immunization Schedule Nunavut Routine Adult Immunization Schedule Nunavut Immunization
Victorian immunisation catch-up tool for 10 to 19 year olds
 Victorian immunisation catch-up tool for 10 to 19 year olds For healthy children with no documented history of vaccine administration The objective of catch-up vaccination is to complete a course of age
Victorian immunisation catch-up tool for 10 to 19 year olds For healthy children with no documented history of vaccine administration The objective of catch-up vaccination is to complete a course of age
S404- Meningococcal Vaccines: Updated Policy Statement 2014
 S404- Meningococcal Vaccines: Updated Policy Statement 2014 Michael T. Brady, MD Associate Medical Director Nationwide Children s Hospital Columbus, Ohio Disclosure of Relevant Relationship Dr. Brady (or
S404- Meningococcal Vaccines: Updated Policy Statement 2014 Michael T. Brady, MD Associate Medical Director Nationwide Children s Hospital Columbus, Ohio Disclosure of Relevant Relationship Dr. Brady (or
Changes to the meningococcal C conjugate (MenC) vaccine schedule 2014 first-time university entrants under the age of 25
 Changes to the meningococcal C conjugate (MenC) vaccine schedule 2014 first-time university entrants under the age of 25 An update for registered healthcare practitioners August 2014 Quality Education
Changes to the meningococcal C conjugate (MenC) vaccine schedule 2014 first-time university entrants under the age of 25 An update for registered healthcare practitioners August 2014 Quality Education
Monitoring vaccine-preventable diseases is
 New South Wales annual vaccinepreventable disease report, 2013 Surveillance Report Alexander Rosewell, a Paula Spokes a and Robin Gilmour a Correspondence to Robin Gilmour (e-mail: rgilm@doh.health.nsw.gov.au).
New South Wales annual vaccinepreventable disease report, 2013 Surveillance Report Alexander Rosewell, a Paula Spokes a and Robin Gilmour a Correspondence to Robin Gilmour (e-mail: rgilm@doh.health.nsw.gov.au).
UNSCHEDULED VACCINATION OF CHILDREN AND YOUNG PEOPLE WHO HAVE OUTSTANDING ROUTINE IMMUNISATIONS. Service Specification
 UNSCHEDULED VACCINATION OF CHILDREN AND YOUNG PEOPLE WHO HAVE OUTSTANDING ROUTINE IMMUNISATIONS Service Specification National Enhanced Service Specification For The Unscheduled Vaccination Of Children
UNSCHEDULED VACCINATION OF CHILDREN AND YOUNG PEOPLE WHO HAVE OUTSTANDING ROUTINE IMMUNISATIONS Service Specification National Enhanced Service Specification For The Unscheduled Vaccination Of Children
Study No.: Title: Rationale: Note: Phase: Study Period: Study Design: Centres: Indication: Treatment: Hib-MenCY F1 Group Hib-MenCY F2 Group
 The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The Australian Immunisation Handbook, 10th Edition, 2013 What s new?
 The Australian Immunisation Handbook, 10th Edition, 2013 What s new? This slide set has been developed by the staff of the NCIRS for education and teaching purposes Last updated: 8 April 2013 1 Presentation
The Australian Immunisation Handbook, 10th Edition, 2013 What s new? This slide set has been developed by the staff of the NCIRS for education and teaching purposes Last updated: 8 April 2013 1 Presentation
NHS public health functions agreement
 NHS public health functions agreement 2016-17 Service specification No. 31 Meningococcal group B (MenB) programme Classification: official NHS England INFORMATION READER BOX Directorate Medical Commissioning
NHS public health functions agreement 2016-17 Service specification No. 31 Meningococcal group B (MenB) programme Classification: official NHS England INFORMATION READER BOX Directorate Medical Commissioning
Invasive Meningococcal Disease - prevention through vaccination
 Invasive Meningococcal Disease - prevention through vaccination Dr Shamez Ladhani Paediatric Infectious Diseases Consultant Public Health England Email: shamez.ladhani@phe.gov.uk a pain you cannot describe
Invasive Meningococcal Disease - prevention through vaccination Dr Shamez Ladhani Paediatric Infectious Diseases Consultant Public Health England Email: shamez.ladhani@phe.gov.uk a pain you cannot describe
PRODUCT MONOGRAPH NIMENRIX. Meningococcal polysaccharide groups A, C, W-135 and Y conjugate vaccine. Powder and diluent for solution for injection
 PRODUCT MONOGRAPH Meningococcal polysaccharide groups A, C, W-135 and Y conjugate vaccine Powder and diluent for solution for injection Active Immunizing Agent Pfizer Canada Inc. 17,300 Trans-Canada Highway
PRODUCT MONOGRAPH Meningococcal polysaccharide groups A, C, W-135 and Y conjugate vaccine Powder and diluent for solution for injection Active Immunizing Agent Pfizer Canada Inc. 17,300 Trans-Canada Highway
Disclosure Statement. Encapsulated Bacteria. Functions of the Spleen 10/25/2017. Pharmacist Learning Objectives
 Pharmacist Learning Objectives No Spleen? No Problem. A Review of Vaccinations Indicated for the Asplenic Patient SCSHP Fall Meeting October 26, 2017 Explain the rationale for vaccinations in Select the
Pharmacist Learning Objectives No Spleen? No Problem. A Review of Vaccinations Indicated for the Asplenic Patient SCSHP Fall Meeting October 26, 2017 Explain the rationale for vaccinations in Select the
Meningococcal Disease and College Students
 MASSACHUSETTS PUBLIC HEALTH FACT SHEET Meningococcal Disease and College Students May 2018 Page 1 of 4 What is meningococcal disease? Meningococcal disease is caused by infection with bacteria called Neisseria
MASSACHUSETTS PUBLIC HEALTH FACT SHEET Meningococcal Disease and College Students May 2018 Page 1 of 4 What is meningococcal disease? Meningococcal disease is caused by infection with bacteria called Neisseria
2015 ACIP Immunization Schedules & Recommendations and MDPH Updates
 Massachusetts Department of Public Health Division of Epidemiology and Immunization 2015 ACIP Immunization Schedules & Recommendations and MDPH Updates Susan M. Lett, MD, MPH Medical Director, Immunization
Massachusetts Department of Public Health Division of Epidemiology and Immunization 2015 ACIP Immunization Schedules & Recommendations and MDPH Updates Susan M. Lett, MD, MPH Medical Director, Immunization
Immunisation schedule Victoria
 Infants and children July 2018 Birth H-B-Vax-II Paediatric or Paediatric 2 months (from 6 weeks) and 4 months Diphtheria-tetanus-pertussis, poliomyelitis-hepatitis B- Haemophilus influenzae type b Anterolateral
Infants and children July 2018 Birth H-B-Vax-II Paediatric or Paediatric 2 months (from 6 weeks) and 4 months Diphtheria-tetanus-pertussis, poliomyelitis-hepatitis B- Haemophilus influenzae type b Anterolateral
NHS public health functions agreement Service specification No.6 Meningococcal C (MenC) containing vaccine immunisation programme
 NHS public health functions agreement 2018-19 Service specification No.6 Meningococcal C (MenC) containing vaccine immunisation programme 1 NHS public health functions agreement 2018-19 Service specification
NHS public health functions agreement 2018-19 Service specification No.6 Meningococcal C (MenC) containing vaccine immunisation programme 1 NHS public health functions agreement 2018-19 Service specification
Immunisation Policy. Country Children s Early Learning Ph: M:
 Immunisation Policy Published June 2018 Review Date December 2019 Sources Education and Care Services National Regulations, October 2017 Guide to the Education and Care Services National Law and the Education
Immunisation Policy Published June 2018 Review Date December 2019 Sources Education and Care Services National Regulations, October 2017 Guide to the Education and Care Services National Law and the Education
Decision to amend access criteria for some vaccines
 3 July 2015 Decision to amend access criteria for some vaccines PHARMAC is pleased to announce the following changes to the funding access criteria for a number of vaccines. The changes in criteria follow
3 July 2015 Decision to amend access criteria for some vaccines PHARMAC is pleased to announce the following changes to the funding access criteria for a number of vaccines. The changes in criteria follow
MenC. MenW MenY
 The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
Immunization Documentation Upload instructions
 Immunization Documentation Upload instructions 1. Log into Health-e-Messaging using your Kerberos ID and password. 2. Enter your student ID number. 3. From the left side of the screen choose either: Immunizations
Immunization Documentation Upload instructions 1. Log into Health-e-Messaging using your Kerberos ID and password. 2. Enter your student ID number. 3. From the left side of the screen choose either: Immunizations
Meningococcal Update. Disclosure. Meningococal and Influenza Vaccines Update! Robert Wittler, MD Sept 12, 2014 KAAP Fall CME Meeting
 1 Meningococal and Influenza Vaccines Update! Robert Wittler, MD Sept 12, 2014 KAAP Fall CME Meeting 2 Disclosure Speakers Bureau: Sanofi Pasteur Vaccines and Novartis! I do not intend to discuss an unapproved/
1 Meningococal and Influenza Vaccines Update! Robert Wittler, MD Sept 12, 2014 KAAP Fall CME Meeting 2 Disclosure Speakers Bureau: Sanofi Pasteur Vaccines and Novartis! I do not intend to discuss an unapproved/
type b (Hib) disease
 6 Haemophilus influenzae Haemophilus influenzae Key information Mode of transmission Incubation period Period of communicability Disease burden Funded vaccines Dose, presentation, route By inhalation of
6 Haemophilus influenzae Haemophilus influenzae Key information Mode of transmission Incubation period Period of communicability Disease burden Funded vaccines Dose, presentation, route By inhalation of
Advisory Committee on Immunization Practices VACCINE ACRONYMS
 May 1, 2015 Vaccine Acronyms Page 1 of 5 Advisy Committee on Immunization Practices VACCINE ACRONYMS Vaccines Included in the Immunization Schedules f Children, Adolescents, and Adults Following is a table
May 1, 2015 Vaccine Acronyms Page 1 of 5 Advisy Committee on Immunization Practices VACCINE ACRONYMS Vaccines Included in the Immunization Schedules f Children, Adolescents, and Adults Following is a table
Immunization Update. William Atkinson, MD, MPH. Immunization Action Coalition
 Immunization Update William Atkinson, MD, MPH Immunization Action Coalition Coordinated School Health Conference Lake Ozark, Missouri December 3, 2016 1 Disclosures William Atkinson has no financial relationships
Immunization Update William Atkinson, MD, MPH Immunization Action Coalition Coordinated School Health Conference Lake Ozark, Missouri December 3, 2016 1 Disclosures William Atkinson has no financial relationships
Patient Group Direction For the supply and administration of
 Patient Group Direction For the supply and administration of HAEMOPHILUS INFLUENZAE TYPE B AND MENINGOCOCCAL C CONJUGATE VACCINE (HIB-MenC) MENITORIX Vaccine By Registered Nurse/Midwife/Health Visitor
Patient Group Direction For the supply and administration of HAEMOPHILUS INFLUENZAE TYPE B AND MENINGOCOCCAL C CONJUGATE VACCINE (HIB-MenC) MENITORIX Vaccine By Registered Nurse/Midwife/Health Visitor
- Infanrix hexa (DTPa-HBV-IPV/Hib): GSK Biologicals combined diphtheria, tetanus, acellular pertussis, hepatitis
 The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
Enhanced immunisation schedule Victoria
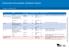 Children from March 2016 Age Disease Vaccine brand Birth Hepatitis B H-B-Vax-II Paediatric 2 months - from 6 weeks Diphtheria-tetanus-pertussis, poliomyelitis-hepatitis B- Reconstitute Site given Route
Children from March 2016 Age Disease Vaccine brand Birth Hepatitis B H-B-Vax-II Paediatric 2 months - from 6 weeks Diphtheria-tetanus-pertussis, poliomyelitis-hepatitis B- Reconstitute Site given Route
Study No.: Title: Rationale: Phase: Study Period: Study Design Centers: Indication: Treatment: Objectives: Primary Outcome/Efficacy Variable:
 The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
New Jersey Department of Health Vaccine Preventable Disease Program Childhood and Adolescent Recommended Vaccines
 New Jersey Department of Health Vaccine Preventable Disease Program Childhood and Adolescent Recommended Vaccines Antigens Vaccine Approved Age Daptacel Diphtheria, Tetanus, and acellular Pertussis (DTaP)
New Jersey Department of Health Vaccine Preventable Disease Program Childhood and Adolescent Recommended Vaccines Antigens Vaccine Approved Age Daptacel Diphtheria, Tetanus, and acellular Pertussis (DTaP)
Significant events in immunisation policy and practice* in Australia
 Significant events in immunisation policy and practice* in Australia Year 1804 First vaccine (for smallpox) used in Australia 1916 Commonwealth Serum Laboratories (CSL) established in Victoria to produce
Significant events in immunisation policy and practice* in Australia Year 1804 First vaccine (for smallpox) used in Australia 1916 Commonwealth Serum Laboratories (CSL) established in Victoria to produce
Current Immunisation Issues in the Nepean Blue Mountains Presented as part of the Immunisation Update education series in October 2017
 Current Immunisation Issues in the Nepean Blue Mountains 2017 Presented as part of the Immunisation Update education series in October 2017 Additional funded vaccines Catch up vaccines for all individuals
Current Immunisation Issues in the Nepean Blue Mountains 2017 Presented as part of the Immunisation Update education series in October 2017 Additional funded vaccines Catch up vaccines for all individuals
PHN - Western Victoria Immunisation Forum Stephen Pellissier Manager Immunisation, DHHS 29 July 2017
 PHN - Western Victoria Immunisation Forum 2017 Stephen Pellissier Manager Immunisation, DHHS 29 July 2017 Herding Cats 2016-17 Again a huge increase in political and media focus on immunisation and vaccination
PHN - Western Victoria Immunisation Forum 2017 Stephen Pellissier Manager Immunisation, DHHS 29 July 2017 Herding Cats 2016-17 Again a huge increase in political and media focus on immunisation and vaccination
PHAA Immunisaton Conference. Adelaide 5-7 June 2018
 PHAA Immunisaton Conference Adelaide 5-7 June 2018 Good Public Health Messaging Reporting back Cold Chain Nurse Practitioner Immunisation Cold Chain Use of and access to thermostability data has been
PHAA Immunisaton Conference Adelaide 5-7 June 2018 Good Public Health Messaging Reporting back Cold Chain Nurse Practitioner Immunisation Cold Chain Use of and access to thermostability data has been
Vaccines available in 2016
 Endorsed ATAGI 59 Australian Technical Advisory Group on Immunisation (ATAGI) advice for immunisation providers regarding the administration of seasonal influenza vaccines in 2016 This document provides
Endorsed ATAGI 59 Australian Technical Advisory Group on Immunisation (ATAGI) advice for immunisation providers regarding the administration of seasonal influenza vaccines in 2016 This document provides
NHS Greater Glasgow & Clyde SOP No. BMT Haemopoietic Stem Cell Transplantation Services Vaccination Policy
 This policy was written with advice from Dr S Ahmed, Consultant in Public Health, Greater Glasgow & Clyde. We would like to thank him for his guidance. 1. Background It is recommended by EBMT and CDC that
This policy was written with advice from Dr S Ahmed, Consultant in Public Health, Greater Glasgow & Clyde. We would like to thank him for his guidance. 1. Background It is recommended by EBMT and CDC that
PHN - Western Victoria Immunisation Forum Chelsea Taylor Senior Policy Advisor Immunisation, DHHS 21 October 2017
 PHN - Western Victoria Immunisation Forum 2017 Chelsea Taylor Senior Policy Advisor Immunisation, DHHS 21 October 2017 Australian Immunisation Register (AIR) quarterly immunisation coverage data Victoria,
PHN - Western Victoria Immunisation Forum 2017 Chelsea Taylor Senior Policy Advisor Immunisation, DHHS 21 October 2017 Australian Immunisation Register (AIR) quarterly immunisation coverage data Victoria,
Whose Calling the Shots? - A 2019 Vaccine Update. Frank Bell Swedish Pediatric Infectious Disease Jan 2019
 Whose Calling the Shots? - A 2019 Vaccine Update Frank Bell Swedish Pediatric Infectious Disease Jan 2019 2019 Vaccine Update Vaccines Meningococcal serogroup B HPV Pertussis Influenza Children with uncertain
Whose Calling the Shots? - A 2019 Vaccine Update Frank Bell Swedish Pediatric Infectious Disease Jan 2019 2019 Vaccine Update Vaccines Meningococcal serogroup B HPV Pertussis Influenza Children with uncertain
12 Meningococcal disease
 12 Meningococcal disease Key information Mode of transmission By respiratory droplets or direct contact with nasopharyngeal secretions from a carrier or case. Incubation period Period of communicability
12 Meningococcal disease Key information Mode of transmission By respiratory droplets or direct contact with nasopharyngeal secretions from a carrier or case. Incubation period Period of communicability
PedsCases Podcast Scripts
 PedsCases Podcast Scripts This is a text version of a podcast from Pedscases.com on CPS Meningococcal Vaccination These podcasts are designed to give medical students an overview of key topics in pediatrics.
PedsCases Podcast Scripts This is a text version of a podcast from Pedscases.com on CPS Meningococcal Vaccination These podcasts are designed to give medical students an overview of key topics in pediatrics.
Objectives 1. Become familiar with current immunization schedules for children
 Sarah Matches, DO, FAAP Assistant Professor UNTHSC, Dept. of Pediatrics Objectives 1. Become familiar with current immunization schedules for children 2. Describe the international progress in reducing
Sarah Matches, DO, FAAP Assistant Professor UNTHSC, Dept. of Pediatrics Objectives 1. Become familiar with current immunization schedules for children 2. Describe the international progress in reducing
Update on Vaccine Recommendations. Objectives. Childhood Immunization Schedule At the Turn of the Century. New Horizons in Pediatrics April 30, 2017
 Centers for for Disease Disease Control Control and and Prevention Prevention National Center for Immunization and Respiratory Diseases Update on Vaccine Recommendations New Horizons in Pediatrics April
Centers for for Disease Disease Control Control and and Prevention Prevention National Center for Immunization and Respiratory Diseases Update on Vaccine Recommendations New Horizons in Pediatrics April
