An Overview of the Design and Implementation of FDA s Prospective Routine Observational Monitoring Program Tools for Safety Surveillance
|
|
|
- Diana Powell
- 5 years ago
- Views:
Transcription
1 2013, The Brookings Institution Brookings Roundtable on Active Medical Product Surveillance An Overview of the Design and Implementation of FDA s Prospective Routine Observational Monitoring Program Tools for Safety Surveillance Jennifer Nelson, Group Health Research Institute and University of Washington Elizabeth Chrischilles, University of Iowa Department of Epidemiology Monday, September 16, 2013
2 2013, The Brookings Institution Mini-Sentinel s Distributed Database 1- User creates and submits query (a computer program) 2- Data partners retrieve query 3- Data partners review and run query against their local data 4- Data partners review results 5- Data partners return results via secure network 6- Results are aggregated 3 Source: Mini-Sentinel
3 2013, The Brookings Institution Mini-Sentinel Analyses Modular Programs Rapid assessments Near real-time Executed by each data partner behind their firewall For additional information, please see the Mini-Sentinel Presentation on Modular Programs Protocol-Based Assessments Formal and detailed evaluations Customized study designs and protocols More resource- and time- intensive than modular programs For examples of past Mini-Sentinel Protocol-Based Assessments, please see here or here Prospective Routine Observational Monitoring Program Tools (PROMPT) Relatively standardized Semi-automated Routine prospective surveillance program Can examine a number of products simultaneously 4
4 2013, The Brookings Institution Housekeeping To minimize feedback, please confirm that the microphone on your telephone is muted. To mute your phone, press the mute button or *6. (To un-mute, press *7 ) There will be opportunities for questions and discussion at the end of today s presentations. Please use the Q & A tab on the top of your screen to submit your questions into the queue at any point and we will call upon you to state your question. Call the Level 3 Conferencing at with technical problems. 5
5 2013, The Brookings Institution Brookings Roundtable on Active Medical Product Surveillance An Overview of the Design and Implementation of FDA s Prospective Routine Observational Monitoring Program Tools for Safety Surveillance Jennifer Nelson, Group Health Research Institute and University of Washington Elizabeth Chrischilles, University of Iowa Department of Epidemiology Monday, September 16, 2013
6 7 PROMPT: Prospective Routine Observational Monitoring Program Tools Elizabeth Chrischilles, U of Iowa Jennifer Nelson, Group Health and U of WA Mini-Sentinel Protocol Core and Methods Core September 16, 2013
7 8 FDA s Vision for a Semi-Automated Surveillance System as Another Surveillance Tool
8 9 What constitutes a comprehensive safety surveillance system? Semi-automated routine surveillance, applying general tools with minor adaptations to address the specific product But also Ability to bring specialized expertise to bear on specific issue(s) that may arise in product lifecycle
9 10 Complements other approaches Other Mini Sentinel activities describe populations and treatment (e.g., uptake) one-time, retrospective customized protocol-based assessments for older products monitor impact of FDA actions (e.g., a label change) FDA activities beyond Mini Sentinel surveillance based on spontaneous reports untargeted surveillance (data mining) of healthcare data customized protocol-based studies using healthcare data
10 11 Overview of Anticipated Procedures for Active Surveillance of New Medical Products Using the PROMPT System
11 12 Goal Relatively standardized Prospective A number of products simultaneously Signal potential excess risk for subsequent follow-up Tools: Cohort and outcome library Core confounder definition Module selection tool Analysis modules User guides
12 13 PROMPT surveillance at a glance Newly marketed product Define exposures, outcomes, etc Choose analysis approach Estimate the risk Aggregate results over time Apply alerting rules Report to FDA FDA reports to public when appropriate
13 PROMPT surveillance: who, what, when Newly marketed product Define exposures, outcomes, etc FDA: Selects product for surveillance 6 mo prior to desired start Planning team: Which outcomes? Post-exposure time window? Who is eligible? Comparators? Which confounders? Which design? Plan for promptly evaluating signals? info@mini-sentinel.org 14
14 15 PROMPT Outcome selection occurs in association with several other medical products of that type (e.g., acute liver injury for drugs; febrile seizures for vaccines) or another product in the class and is thus of general interest or reason to suspect that that product in particular might increase the risk of that HOI, for example because of a signal identified in pre-approval animal studies or clinical trials Generation Signal Generation Signal Refinement Signal Evaluation Signal refinement is a process for evaluating the magnitude and clinical significance of a suspected association 15
15 Standard outcome algorithms GI bleeding Systemic lupus erythematosis Pancreatitis Inflammatory Bowel Disease Premature delivery Juvenile RA Pulmonary Fibrosis Tuberculosis Hypertensive crisis Erythema multiforme major Agranulocytosis Idiopathic thrombocytopenic purpura Aplastic Anemia Thrombocytopenia Bronchospasm Henoch Schonlein purpura CVA Peripheral neuropathy Venous Thromboembolism Guillan-Barre syndrome Hemorrhagic CVA Tendon rupture Ischemic CVA Seizure, febrile Neutropenia Suicide Bell s Palsy Valvulopathy Spontaneous abortion/stillbirth Hip fracture Acute Respiratory Failure Pulmonary hypertension Sepsis Rhabdomyolysis Deafness Sudden cardiac death Thrombotic thrombocytopenic purpura Diabetes info@mini-sentinel.org 16
16 Standard Outcome Definitions: Outcome Algorithm Rationale Reference Acute Ischemic Stroke Recommended Primary: 434, 436 in first position of a hospital claim Recommended Secondary: 433.x1, 434 (excluding 434.x0), 436 in first position of a hospital claim Some algorithms also included x1 and excluded 434.x0 (see Recommended Secondary), however 433.x1 may also have low PPV and the PPV for 434 (without exclusion) in first position is good (>85%). PPV diminishes slightly when any position, or when community vs tertiary care. Algorithm that included 433.x1, 434 (excluding 434.x0), and 436 performed well. 433 (other than 433.x0) had very low PPV. One study found 433.x1 PPV=71% Also Observed (but not 433 had very low PPV. 433.x0 PPV was 2%, recommended): 433, 434, 436 in first 433.x1 PPV was only 20% position info@mini-sentinel.org 17
17 18 PROMPT Cohort selection Exclude prior history of event? Separate cohorts for each product indication? Separate cohorts for individuals with major risk factors? High priority subgroups
18 19 Standard cohort algorithms Persons with coronary artery disease Persons with mood disorders Persons with end-stage renal disease Hypertensives Smokers Asthmatics Persons with dementia Persons who received fluoroquinolones for postexposure prophylaxis First responders Nursing home residents Pregnant women Live births Premature babies Persons at high risk for influenza complications Immunocompromised persons Type 1 diabetics Type 2 diabetics Obese persons
19 PROMPT Core confounder definitions Demographics Age Sex Calendar time* Data Partner* Healthcare utilization in baseline period # of visits to emergency departments # of ambulatory visits # of hospitalizations # of distinct drugs ordered/dispensed # of prescriptions ordered/dispensed Lifestyle Factors Smoking**, per algorithm developed by the 15 Cohorts workgroup Body mass index** i, if available in the common data model; otherwise, per algorithm developed by the 15 Cohorts workgroup Combined Charlson-Elixhauser comorbidity index * Requires special consideration given sequential analyses ** Discreetly-captured data field not currently in the Mini Sentinel Common Data Model, therefore alternate diagnosis-based algorithm suggested info@mini-sentinel.org 20
20 21 PROMPT surveillance: how Newly marketed product Define exposures, outcomes, etc Choose analysis approach Cohort or self-controlled? Relative risk? Risk difference?
21 22 What affects the choice? Exposure-outcome characteristics
22 23 Additional characteristics that might affect design and analytic choice
23 PROMPT Module Selection Tool MS Taxonomy project aimed at pre-thinking major design and analysis considerations Outlined methodological decisions in routine monitoring Identified methodological options at each decision node Determined the scenarios characteristics that might influence the decisions Mapped scenario characteristics to recommended options Developed interactive tool to expedite decisionmaking process for routine monitoring 24
24 PROMPT Analysis Modules 25
25 26 Overview: Modular approach to drug safety monitoring in a distributed database system Build pre-programmed modules that can be quickly activated to monitor the use and safety effects of new and existing drugs Construct an analytic dataset based on pre-specified inputs: Population eligibility Outcome, exposure/comparator, and confounder definitions Describe the population of exposed and comparators Estimate adjusted safety effects across data partners Modules can be run once as single analyses or conducted repeatedly over time as data are refreshed and uptake occurs using sequential testing methods and pre-specified inputs: Overall Type 1 (false positive) error to control across multiple tests Frequency of testing and shape of signaling threshold over time Maximum sample size after which surveillance will stop if no signal
26 27 Some Details: Modular approach to drug safety monitoring in a distributed database system Validated programming code Can be run asynchronously across data partners as data get refreshed while preserving data privacy Methods involve standard epidemiologic designs and statistical methods Confounding adjustment via a self-controlled design, PS matching, regression, or inverse weighting approaches Can estimate difference and ratio measures (rate or risk) Addresses heterogeneity across data partners Can flexibly employ a variety of sequential designs and analyses Frequency of sequential analyses Signaling rules for alerting a difference in risk between groups over time
27 28 Current Status: Modular approach to drug safety monitoring in a distributed database system Version 1 of code has been developed and tested in the MSDD Code development was conducted using several example pairs where association was deemed to be known : True positive drug-event pair: ACEis and angioedema True negative drug-event pair: clindamycin and AMI True positive vaccine-event pair: MMRV and fever/seizure Version 1 of module user s manuals have been written Includes needed module inputs and resulting standardized output Describes the epidemiological design and statistical methods employed Version 1 of surveillance reports have been created Next steps: further testing and enhancement of code, manual and reports
28 29 PROMPT surveillance: estimate risk Newly marketed product Define exposures, outcomes, etc Choose analysis approach Estimate the risk Module 1 Module 2 Module 3 Module 4 Self-controlled design Exposure matching by propensity score (cohort design) Regression (cohort design) IPTW regression (cohort design)
29 30 Self-controlled design module (PROMPT 1) Variant of the self-controlled case series Utilizes cases (those who experience an adverse event) only Compares risk in exposed and comparator time windows within the same person Time-fixed confounders are implicitly controlled Future version will allow adjustment for time-varying confounders (Sequential) test is based on a LRT statistic comparing observed versus expected event counts Sequential boundaries extend maxsprt method Flat boundary; can delay 1st analysis, then continuous subsequent tests
30 31 Lisinopril-angioedema Report date: 1/31/2014 Self-controlled group sequential analysis Last exposure: 7/31/2013 Test no. 5 Cumulative exposed: 71,892 Sequential analysis history and results Tracking New events Cumulative Risk estimates Hypothesis-testing statistics and results Test Most No. of No. of No. of No. of No. of Expec- Relative Risk Log Target Actual No. of H0 # recent new new exposed events events ted no. risk differ- likeli- alpha to alpha cases rejected batch(es) events events patients in risk in of cases ence hood spend spent needed? included in risk in interval control ratio to reject interval control ( cases ) interval test H0 (CV) ( cases ) interval ( con- statistic ( con- trols ) trols ) Date of last analysis: 5/21/2013
31 32 PS matching module (PROMPT 2) First (of three) cohort approaches (e.g., new user design) Utilize individual-level data that remain at each data partner to Estimate a PS (based on pre-specified confounders or using hd-ps) Match exposed to unexposed patients using the PS (fixed matching ratio) Evaluate diagnostics Minimal data combined for central analysis at MSOC Can compute HR, RR, RD comparing exposed and comparators, stratified by data partner Future version will allow disease risk score matching (Sequential) test is based on a LRT statistic comparing observed versus expected event counts Sequential boundaries extend maxsprt method Flat boundary; can delay 1st analysis, then continuous subsequent tests
32 33 PS Matching Dataset Creation Capabilities Substantial flexibility in defining exposures and outcomes, exposure windows, washout period, minimum exposures, blackout periods, etc. Use enrollment data to reconcile overlaps and gaps; define continuous enrollment periods Create continuous exposure periods, incorporating stockpiling Identify incident exposure episodes and outcomes Can restrict cohort based on eligibility criteria Identifies and defines a wide range of core confounders (demographics, healthcare utilization, and comorbid conditions)
33 34 Inputs for Prospective Routine Observational Monitoring Program Tool: cohort matching program ELIGIBILITY INFORMATION Enrollment gap: specifies the number of days bridged between two consecutive enrollment periods to create a single continuous enrollment period. Inclusion/exclusion conditions: defined by creating a SAS dataset with codes defining the inclusion or exclusion of conditions(s) of interest. EXPOSURE INFORMATION Medical product of interest: defined by creating a SAS dataset with codes (i.e., NDCs or CPTs) to identify the product of interest. Comparator of interest: defined by creating a SAS dataset with codes (i.e., NDCs or CPTs) to identify the comparator product of interest.
34 35 PS Matching Module in detail Coordinating center Specify input parameters Start PROMPT 2 Transmit code Multiple data partners Identify Cohort, Outcomes Covariates Calculate confounder scores (PS, hd-ps, DRS) Evaluate diagnostics and aggregate data across partners Transmit data Run diagnostics Create de-identified result files Apply alerting algorithms and interpret results Iterate at next data refresh
35 Diagnostics: Balance before/after matching 36
36 37 PS Module Results over time PS-match PS-match PS-match
37 38 Regression module (PROMPT 3) A second cohort data approach (e.g., new user design) Individual-level data remain at each data partner De-identified data are then combined for central analysis at MSOC Can fit any generalized linear model of interest Logistic regression to estimate an OR Poisson regression to estimate a RR (and incorporate person-time data) Linear regression to estimate a RD (Sequential) test is based on a score statistic Signaling boundaries leverage and extend RCT group sequential methods (unifying family by Kittleson et al.) Allows flexible boundary shape specification (O Brien-Fleming, Pocock, etc.) Can customize testing plan (any continuous or group sequential plan) Uses exact method boundary formulation (vs large sample assumptions)
38 39 Example Individual-level Dataset that remains at each Data Partner StudyID Age Sex Smoker Weight Height Flu Vaccine 1 28 F Current Yes 2 45 M Quit No 100, M Never Yes
39 40 Example of combined aggregated data sent to MSOC: event counts by strata Site Age Cat Sex Flu Vaccine Events N A F Yes A F No A M Yes A M No A M No B F Yes B F No
40 41 IPTW regression module (PROMPT 4) A third cohort approach (e.g., new user design) Utilizes individual-level data that remain at each data partner to Estimate a PS based on pre-specified confounders Estimate a RD using linear regression, inverse weighted by the PS Summary statistics are combined across data partners for analysis Acknowledges site heterogeneity by computing a stratified RD estimate that incorporates variability in propensity score models (Sequential) test is based on a Wald test statistic Sequential boundaries leverage and extend RCT methods Allows flexible boundary shape specification (O Brien-Fleming, Pocock, etc.) Can customize testing plan (any continuous or group sequential plan) Uses exact method boundary formulation (vs. large sample assumptions)
41 42 IPTW regression module Involves a stratified IPTW risk difference Construct site-specific propensity scores (logistic regression) Calculate site-specific IPTW risk difference V( ) & variance (Lunceford & Davidian 2004); send with N s to a central location S S Site 1 1, V( 1) Site 2 2, V( 2 ) Site 9 9 V(, 9 ) Site 10 10, V( 10 ) CENTRAL 10 S = 1 = 10 N S = 1 S N S S
42 43 IPTW regression module Uses a non-parametric permutation approach for rare events Fix outcomes and confounders and permute exposure w/in site ( 1) (1) ( P) ( P) Sites also send permuted datasets {(, V( )),, (, V( ))} S S S S Site 1 1, (, V( )) 1 {( (1) (1) ) ( ( P) ( P), V( ),,, V( ))} 1 N 1 1 1, 1 Site 8, (,V( )) {( (1) (1) ) ( ( P) ( P), V( ),,,V( ))} 8 N , 8 Repeat for each analysis time t=1, 2 T) CENTRAL Overall adjusted risk difference Distribution of permuted risk difference under H 0 Use unifying family (Kittleson) to derive stopping boundaries
43 Example IPTW module reports 44
44 Example IPTW module reports 45
45 Example IPTW module reports 46
46 47 What happens when we find something? Prompt, pre-planned product-specific assessment of positive signal (or in the absence of signal) Examples of post-monitoring follow-up activities: Data checks, analytic code checks Subgroup analyses Adjust for additional confounders Test against other comparators Vary population, O, or E definitions Medical chart validation of cases Quantitative bias analysis Detailed epidemiologic investigation to assess causality
47 Example report: demographics over time 48
48 Example report: surveillance results by site 49
49 50 PROMPT surveillance: reporting Newly marketed product Define exposures, outcomes, etc Choose analysis approach Estimate the risk Aggregate results over time Apply alerting rules Report to FDA FDA reports to public when appropriate
50 51 Information from Mini Sentinel s PROMPT is used to complement other FDA data to help inform regulatory action
51 52 Thank You!
52 2013, The Brookings Institution Brookings Roundtable on Active Medical Product Surveillance An Overview of the Design and Implementation of FDA s Prospective Routine Observational Monitoring Program Tools for Safety Surveillance Jennifer Nelson, Group Health Research Institute and University of Washington Elizabeth Chrischilles, University of Iowa Department of Epidemiology Monday, September 16, 2013
Overview of Anticipated Procedures for Active Surveillance of New Medical Products
 info@mini-sentinel.org 1 Overview of Anticipated Procedures for Active Surveillance of New Medical Products Elizabeth Chrischilles, PhD Mini-Sentinel Protocol Core January 31, 2013 info@mini-sentinel.org
info@mini-sentinel.org 1 Overview of Anticipated Procedures for Active Surveillance of New Medical Products Elizabeth Chrischilles, PhD Mini-Sentinel Protocol Core January 31, 2013 info@mini-sentinel.org
Highlights from the Fifth Annual Sentinel Initiative Public Workshop
 2013, The Brookings Institution Highlights from the Fifth Annual Sentinel Initiative Public Workshop Patrick Archdeacon, Medical Officer, Office of Medical Policy, Center for Drug Evaluation and Research,
2013, The Brookings Institution Highlights from the Fifth Annual Sentinel Initiative Public Workshop Patrick Archdeacon, Medical Officer, Office of Medical Policy, Center for Drug Evaluation and Research,
1 Cohort Identification within FDA's Mini-Sentinel Program
 info@mini-sentinel.org 1 Cohort Identification within FDA's Mini-Sentinel Program October 31, 2013 Mini-Sentinel partner organizations Lead HPHC Institute Data and scientific partners Scientific partners
info@mini-sentinel.org 1 Cohort Identification within FDA's Mini-Sentinel Program October 31, 2013 Mini-Sentinel partner organizations Lead HPHC Institute Data and scientific partners Scientific partners
MINI-SENTINEL METHODS PROSPECTIVE ROUTINE OBSERVATIONAL MONITORING PROGRAM TOOLS (PROMPT) USERS GUIDE
 MINI-SENTINEL METHODS PROSPECTIVE ROUTINE OBSERVATIONAL MONITORING PROGRAM TOOLS (PROMPT) USERS GUIDE Prepared by: Richard Platt, MD, MSc, 1 Patrick Archdeacon, MD, MS, 2 Carlos Bell, MPH, 2 Jeffrey Brown,
MINI-SENTINEL METHODS PROSPECTIVE ROUTINE OBSERVATIONAL MONITORING PROGRAM TOOLS (PROMPT) USERS GUIDE Prepared by: Richard Platt, MD, MSc, 1 Patrick Archdeacon, MD, MS, 2 Carlos Bell, MPH, 2 Jeffrey Brown,
Brookings Roundtable on Active Medical Product Surveillance:
 2012, The Brookings Institution Brookings Roundtable on Active Medical Product Surveillance: Findings from a Mini-Sentinel Medical Product Assessment Marsha Reichman, U.S. Food and Drug Administration
2012, The Brookings Institution Brookings Roundtable on Active Medical Product Surveillance: Findings from a Mini-Sentinel Medical Product Assessment Marsha Reichman, U.S. Food and Drug Administration
Modular Program Report
 Modular Program Report The following report(s) provides findings from an FDA initiated query using its Mini Sentinel pilot. While Mini Sentinel queries may be undertaken to assess potential medical product
Modular Program Report The following report(s) provides findings from an FDA initiated query using its Mini Sentinel pilot. While Mini Sentinel queries may be undertaken to assess potential medical product
MINI SENTINEL METHODS
 MINI SENTINEL METHODS IMPROVING SEQUENTIAL SAFETY SURVEILLANCE PLANNING METHODS FOR ROUTINE ASSESSMENTS THAT USE REGRESSION ADJUSTMENT OR WEIGHTING TO CONTROL CONFOUNDING Prepared by: Jennifer C. Nelson,
MINI SENTINEL METHODS IMPROVING SEQUENTIAL SAFETY SURVEILLANCE PLANNING METHODS FOR ROUTINE ASSESSMENTS THAT USE REGRESSION ADJUSTMENT OR WEIGHTING TO CONTROL CONFOUNDING Prepared by: Jennifer C. Nelson,
FDA Sentinel Initiative: Status Update and Examples of Contributions to Regulatory Decisions
 FDA Sentinel Initiative: Status Update and Examples of Contributions to Regulatory Decisions Marsha E. Reichman, Ph.D. Scientific Lead for Surveillance Programs CDER Lead for the Sentinel Initiative OPE/OSE/CDER/FDA
FDA Sentinel Initiative: Status Update and Examples of Contributions to Regulatory Decisions Marsha E. Reichman, Ph.D. Scientific Lead for Surveillance Programs CDER Lead for the Sentinel Initiative OPE/OSE/CDER/FDA
Using the NIH Collaboratory's and PCORnet's distributed data networks for clinical trials and observational research - A preview
 Using the NIH Collaboratory's and PCORnet's distributed data networks for clinical trials and observational research - A preview Millions of people. Strong collaborations. Privacy first. Jeffrey Brown,
Using the NIH Collaboratory's and PCORnet's distributed data networks for clinical trials and observational research - A preview Millions of people. Strong collaborations. Privacy first. Jeffrey Brown,
Integrating Effectiveness and Safety Outcomes in the Assessment of Treatments
 Integrating Effectiveness and Safety Outcomes in the Assessment of Treatments Jessica M. Franklin Instructor in Medicine Division of Pharmacoepidemiology & Pharmacoeconomics Brigham and Women s Hospital
Integrating Effectiveness and Safety Outcomes in the Assessment of Treatments Jessica M. Franklin Instructor in Medicine Division of Pharmacoepidemiology & Pharmacoeconomics Brigham and Women s Hospital
Modular Program Report
 Modular Program Report The following report(s) provides findings from an FDA initiated query using its Mini Sentinel pilot. While Mini Sentinel queries may be undertaken to assess potential medical product
Modular Program Report The following report(s) provides findings from an FDA initiated query using its Mini Sentinel pilot. While Mini Sentinel queries may be undertaken to assess potential medical product
Modular Program Report
 Modular Program Report The following report(s) provides findings from an FDA initiated query using its Mini Sentinel pilot. While Mini Sentinel queries may be undertaken to assess potential medical product
Modular Program Report The following report(s) provides findings from an FDA initiated query using its Mini Sentinel pilot. While Mini Sentinel queries may be undertaken to assess potential medical product
Modular Program Report
 Modular Program Report The following report(s) provides findings from an FDA initiated query using its Mini Sentinel pilot. While Mini Sentinel queries may be undertaken to assess potential medical product
Modular Program Report The following report(s) provides findings from an FDA initiated query using its Mini Sentinel pilot. While Mini Sentinel queries may be undertaken to assess potential medical product
eappendix A. Simulation study parameters
 eappendix A. Simulation study parameters Parameter type Notation Values or distributions Definition i {1 20} Indicator for each of 20 monitoring periods N i 500*i Number of exposed patients in each period,
eappendix A. Simulation study parameters Parameter type Notation Values or distributions Definition i {1 20} Indicator for each of 20 monitoring periods N i 500*i Number of exposed patients in each period,
OBSERVATIONAL MEDICAL OUTCOMES PARTNERSHIP
 OBSERVATIONAL Patient-centered observational analytics: New directions toward studying the effects of medical products Patrick Ryan on behalf of OMOP Research Team May 22, 2012 Observational Medical Outcomes
OBSERVATIONAL Patient-centered observational analytics: New directions toward studying the effects of medical products Patrick Ryan on behalf of OMOP Research Team May 22, 2012 Observational Medical Outcomes
Disclaimer. The following report contains a description of the request, request specifications, and results from the modular program run(s).
 Disclaimer The following report(s) provides findings from an FDA initiated query using its Mini Sentinel pilot. While Mini Sentinel queries may be undertaken to assess potential medical product safety
Disclaimer The following report(s) provides findings from an FDA initiated query using its Mini Sentinel pilot. While Mini Sentinel queries may be undertaken to assess potential medical product safety
Plans for Surveillance of Acute Myocardial Infarction in users of Oral Anti Diabetes Drugs
 Plans for Surveillance of Acute Myocardial Infarction in users of Oral Anti Diabetes Drugs Bruce Fireman Kaiser Permanente, Oakland Brookings, Washington DC, 1 12 2011 Aims Develop and assess a framework
Plans for Surveillance of Acute Myocardial Infarction in users of Oral Anti Diabetes Drugs Bruce Fireman Kaiser Permanente, Oakland Brookings, Washington DC, 1 12 2011 Aims Develop and assess a framework
Modular Program Report
 Modular Program Report Disclaimer The following report(s) provides findings from an FDA initiated query using Sentinel. While Sentinel queries may be undertaken to assess potential medical product safety
Modular Program Report Disclaimer The following report(s) provides findings from an FDA initiated query using Sentinel. While Sentinel queries may be undertaken to assess potential medical product safety
VACCINE ACTIVE SURVEILLANCE I
 VACCINE ACTIVE SURVEILLANCE I Caitlin N Dodd, MS Biostatistician, Global Child Health Cincinnati Children s Hospital Medical Center CONTENTS M1 Compare epidemiological study methods Discuss the benefits
VACCINE ACTIVE SURVEILLANCE I Caitlin N Dodd, MS Biostatistician, Global Child Health Cincinnati Children s Hospital Medical Center CONTENTS M1 Compare epidemiological study methods Discuss the benefits
Division of Pharmacoepidemiology And Pharmacoeconomics Technical Report Series
 Division of Pharmacoepidemiology And Pharmacoeconomics Technical Report Series Year: 2012 #002 Taxonomy for monitoring methods within a medical product safety surveillance system: Year two report of the
Division of Pharmacoepidemiology And Pharmacoeconomics Technical Report Series Year: 2012 #002 Taxonomy for monitoring methods within a medical product safety surveillance system: Year two report of the
MINI-SENTINEL SURVEILLANCE PLAN MINI-SENTINEL PROSPECTIVE ROUTINE OBSERVATIONAL MONITORING PROGRAM TOOLS (PROMPT): RIVAROXABAN SURVEILLANCE
 MINI-SENTINEL SURVEILLANCE PLAN MINI-SENTINEL PROSPECTIVE ROUTINE OBSERVATIONAL MONITORING PROGRAM TOOLS (PROMPT): ABAN SURVEILLANCE Prepared by: Ryan Carnahan, PharmD, MS, BCPP, 1 Joshua J. Gagne, PharmD,
MINI-SENTINEL SURVEILLANCE PLAN MINI-SENTINEL PROSPECTIVE ROUTINE OBSERVATIONAL MONITORING PROGRAM TOOLS (PROMPT): ABAN SURVEILLANCE Prepared by: Ryan Carnahan, PharmD, MS, BCPP, 1 Joshua J. Gagne, PharmD,
Modular Program Report
 Disclaimer The following report(s) provides findings from an FDA initiated query using Sentinel. While Sentinel queries may be undertaken to assess potential medical product safety risks, they may also
Disclaimer The following report(s) provides findings from an FDA initiated query using Sentinel. While Sentinel queries may be undertaken to assess potential medical product safety risks, they may also
Online Supplementary Material
 Section 1. Adapted Newcastle-Ottawa Scale The adaptation consisted of allowing case-control studies to earn a star when the case definition is based on record linkage, to liken the evaluation of case-control
Section 1. Adapted Newcastle-Ottawa Scale The adaptation consisted of allowing case-control studies to earn a star when the case definition is based on record linkage, to liken the evaluation of case-control
Observational Study Designs. Review. Today. Measures of disease occurrence. Cohort Studies
 Observational Study Designs Denise Boudreau, PhD Center for Health Studies Group Health Cooperative Today Review cohort studies Case-control studies Design Identifying cases and controls Measuring exposure
Observational Study Designs Denise Boudreau, PhD Center for Health Studies Group Health Cooperative Today Review cohort studies Case-control studies Design Identifying cases and controls Measuring exposure
Mini-Sentinel Common Data Model
 info@mini-sentinel.org 1 Mini-Sentinel Common Data Model Lesley Curtis on behalf of the Mini-Sentinel Data Core May 8, 2013 info@mini-sentinel.org 2 Guiding principles (selected) Accommodates all requirements
info@mini-sentinel.org 1 Mini-Sentinel Common Data Model Lesley Curtis on behalf of the Mini-Sentinel Data Core May 8, 2013 info@mini-sentinel.org 2 Guiding principles (selected) Accommodates all requirements
Tuning Epidemiological Study Design Methods for Exploratory Data Analysis in Real World Data
 Tuning Epidemiological Study Design Methods for Exploratory Data Analysis in Real World Data Andrew Bate Senior Director, Epidemiology Group Lead, Analytics 15th Annual Meeting of the International Society
Tuning Epidemiological Study Design Methods for Exploratory Data Analysis in Real World Data Andrew Bate Senior Director, Epidemiology Group Lead, Analytics 15th Annual Meeting of the International Society
Risk of serious infections associated with use of immunosuppressive agents in pregnant women with autoimmune inflammatory conditions: cohor t study
 Risk of serious infections associated with use of immunosuppressive agents in pregnant women with autoimmune inflammatory conditions: cohor t study BMJ 2017; 356 doi: https://doi.org/10.1136/bmj.j895 (Published
Risk of serious infections associated with use of immunosuppressive agents in pregnant women with autoimmune inflammatory conditions: cohor t study BMJ 2017; 356 doi: https://doi.org/10.1136/bmj.j895 (Published
Sentinel Initiative Public Workshop. The Brookings Institution Marriott at Metro Center Washington, DC Tuesday, January 14, 2014
 Sentinel Initiative Public Workshop The Brookings Institution Marriott at Metro Center Washington, DC Tuesday, January 14, 2014 State of CBER s Mini-Sentinel Activities Michael Nguyen, MD Division of Epidemiology
Sentinel Initiative Public Workshop The Brookings Institution Marriott at Metro Center Washington, DC Tuesday, January 14, 2014 State of CBER s Mini-Sentinel Activities Michael Nguyen, MD Division of Epidemiology
OHDSI Tutorial: Design and implementation of a comparative cohort study in observational healthcare data
 OHDSI Tutorial: Design and implementation of a comparative cohort study in observational healthcare data Faculty: Martijn Schuemie (Janssen Research and Development) Marc Suchard (UCLA) Patrick Ryan (Janssen
OHDSI Tutorial: Design and implementation of a comparative cohort study in observational healthcare data Faculty: Martijn Schuemie (Janssen Research and Development) Marc Suchard (UCLA) Patrick Ryan (Janssen
PCORnet Use Cases. Observational study: Dabigatran vs warfarin and ischemic and hemorrhagic stroke / extra-cranial bleeding
 PCORnet Use Cases Observational study: Dabigatran vs warfarin and ischemic and hemorrhagic stroke / extra-cranial bleeding 1 Observational study: Dabigatran vs warfarin and stroke / bleeding Goal: Compare
PCORnet Use Cases Observational study: Dabigatran vs warfarin and ischemic and hemorrhagic stroke / extra-cranial bleeding 1 Observational study: Dabigatran vs warfarin and stroke / bleeding Goal: Compare
BIOSTATISTICAL METHODS
 BIOSTATISTICAL METHODS FOR TRANSLATIONAL & CLINICAL RESEARCH PROPENSITY SCORE Confounding Definition: A situation in which the effect or association between an exposure (a predictor or risk factor) and
BIOSTATISTICAL METHODS FOR TRANSLATIONAL & CLINICAL RESEARCH PROPENSITY SCORE Confounding Definition: A situation in which the effect or association between an exposure (a predictor or risk factor) and
Improved Transparency in Key Operational Decisions in Real World Evidence
 PharmaSUG 2018 - Paper RW-06 Improved Transparency in Key Operational Decisions in Real World Evidence Rebecca Levin, Irene Cosmatos, Jamie Reifsnyder United BioSource Corp. ABSTRACT The joint International
PharmaSUG 2018 - Paper RW-06 Improved Transparency in Key Operational Decisions in Real World Evidence Rebecca Levin, Irene Cosmatos, Jamie Reifsnyder United BioSource Corp. ABSTRACT The joint International
CARDIOVASCULAR EFFECTS OF INCRETIN-BASED ANTIHYPERGLYCEMIC DRUGS RELATIVE TO TREATMENT ALTERNATIVES IN OLDER ADULTS. Mugdha Gokhale.
 CARDIOVASCULAR EFFECTS OF INCRETIN-BASED ANTIHYPERGLYCEMIC DRUGS RELATIVE TO TREATMENT ALTERNATIVES IN OLDER ADULTS Mugdha Gokhale A dissertation submitted to the faculty at the University of North Carolina
CARDIOVASCULAR EFFECTS OF INCRETIN-BASED ANTIHYPERGLYCEMIC DRUGS RELATIVE TO TREATMENT ALTERNATIVES IN OLDER ADULTS Mugdha Gokhale A dissertation submitted to the faculty at the University of North Carolina
Varenicline and cardiovascular and neuropsychiatric events: Do Benefits outweigh risks?
 Varenicline and cardiovascular and neuropsychiatric events: Do Benefits outweigh risks? Sonal Singh M.D., M.P.H, Johns Hopkins University Presented by: Sonal Singh, MD MPH September 19, 2012 1 CONFLICTS
Varenicline and cardiovascular and neuropsychiatric events: Do Benefits outweigh risks? Sonal Singh M.D., M.P.H, Johns Hopkins University Presented by: Sonal Singh, MD MPH September 19, 2012 1 CONFLICTS
Methodologies for vaccine safety surveillance. Nick Andrews, Statistics Unit Public Health England October 2015
 Methodologies for vaccine safety surveillance Nick Andrews, Statistics Unit Public Health England October 2015 Some dramas of the past 20 years which do you think turned out to be real adverse reactions?
Methodologies for vaccine safety surveillance Nick Andrews, Statistics Unit Public Health England October 2015 Some dramas of the past 20 years which do you think turned out to be real adverse reactions?
Connecting the data to the science: A biostatistician s role in advancing safety knowledge
 Connecting the data to the science: A biostatistician s role in advancing safety knowledge Jennifer Clark Nelson, PhD Director of Biostatistics Group Health Research Institute Affiliate Professor of Biostatistics
Connecting the data to the science: A biostatistician s role in advancing safety knowledge Jennifer Clark Nelson, PhD Director of Biostatistics Group Health Research Institute Affiliate Professor of Biostatistics
Epidemiology: Overview of Key Concepts and Study Design. Polly Marchbanks
 Epidemiology: Overview of Key Concepts and Study Design Polly Marchbanks Lecture Outline (1) Key epidemiologic concepts - Definition - What epi is not - What epi is - Process of epi research Lecture Outline
Epidemiology: Overview of Key Concepts and Study Design Polly Marchbanks Lecture Outline (1) Key epidemiologic concepts - Definition - What epi is not - What epi is - Process of epi research Lecture Outline
Use of combined oral contraceptives and risk of venous thromboembolism: nested case-control studies using the QResearch and CPRD databases
 open access Use of combined oral contraceptives and risk of venous thromboembolism: nested case-control studies using the and databases Yana Vinogradova, Carol Coupland, Julia Hippisley-Cox Division of
open access Use of combined oral contraceptives and risk of venous thromboembolism: nested case-control studies using the and databases Yana Vinogradova, Carol Coupland, Julia Hippisley-Cox Division of
Working Document on Monitoring and Evaluating of National ART Programmes in the Rapid Scale-up to 3 by 5
 Working Document on Monitoring and Evaluating of National ART Programmes in the Rapid Scale-up to 3 by 5 Introduction Currently, five to six million people infected with HIV in the developing world need
Working Document on Monitoring and Evaluating of National ART Programmes in the Rapid Scale-up to 3 by 5 Introduction Currently, five to six million people infected with HIV in the developing world need
Finland and Sweden and UK GP-HOSP datasets
 Web appendix: Supplementary material Table 1 Specific diagnosis codes used to identify bladder cancer cases in each dataset Finland and Sweden and UK GP-HOSP datasets Netherlands hospital and cancer registry
Web appendix: Supplementary material Table 1 Specific diagnosis codes used to identify bladder cancer cases in each dataset Finland and Sweden and UK GP-HOSP datasets Netherlands hospital and cancer registry
Immunization Safety Office: Overview and Considerations on Safety of Alternative Immunization Schedules
 Immunization Safety Office: Overview and Considerations on Safety of Alternative Immunization Schedules Frank DeStefano MD, MPH Immunization Safety Office Division of Healthcare Quality Promotion, National
Immunization Safety Office: Overview and Considerations on Safety of Alternative Immunization Schedules Frank DeStefano MD, MPH Immunization Safety Office Division of Healthcare Quality Promotion, National
Technical Guidance Note for Global Fund HIV Proposals
 Technical Guidance Note for Global Fund HIV Proposals UNAIDS I World Health Organization I August 2011 Rationale for including pharmacovigilance in the proposal Pharmacovigilance is the science and activities
Technical Guidance Note for Global Fund HIV Proposals UNAIDS I World Health Organization I August 2011 Rationale for including pharmacovigilance in the proposal Pharmacovigilance is the science and activities
DECLARATION OF CONFLICT OF INTEREST
 DECLARATION OF CONFLICT OF INTEREST Warfarin and the risk of major bleeding events in patients with atrial fibrillation: a population-based study Laurent Azoulay PhD 1,2, Sophie Dell Aniello MSc 1, Teresa
DECLARATION OF CONFLICT OF INTEREST Warfarin and the risk of major bleeding events in patients with atrial fibrillation: a population-based study Laurent Azoulay PhD 1,2, Sophie Dell Aniello MSc 1, Teresa
Outcomes: Initially, our primary definitions of pneumonia was severe pneumonia, where the subject was hospitalized
 The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
Research in Real-World Settings: PCORI s Model for Comparative Clinical Effectiveness Research
 Research in Real-World Settings: PCORI s Model for Comparative Clinical Effectiveness Research David Hickam, MD, MPH Director, Clinical Effectiveness & Decision Science Program April 10, 2017 Welcome!
Research in Real-World Settings: PCORI s Model for Comparative Clinical Effectiveness Research David Hickam, MD, MPH Director, Clinical Effectiveness & Decision Science Program April 10, 2017 Welcome!
Practical Lessons Learned for Identification of Thromboembolic Events and Intravenous Immunoglobulin Exposure in the Sentinel Distributed Database
 Practical Lessons Learned for Identification of Thromboembolic Events and Intravenous Immunoglobulin Exposure in the Sentinel Distributed Database Crystal Garcia, MPH, 1 Candace C. Fuller, PhD, MPH, 1
Practical Lessons Learned for Identification of Thromboembolic Events and Intravenous Immunoglobulin Exposure in the Sentinel Distributed Database Crystal Garcia, MPH, 1 Candace C. Fuller, PhD, MPH, 1
The Clinical Information Data Entry Screen is the main screen in the DQCMS application.
 DATA ENTRY Clinical Information The Clinical Information Data Entry Screen is the main screen in the DQCMS application. To enter data, a patient must first be selected from the Patient pull-down list.
DATA ENTRY Clinical Information The Clinical Information Data Entry Screen is the main screen in the DQCMS application. To enter data, a patient must first be selected from the Patient pull-down list.
MINI-SENTINEL METHODS 16 HEALTH OUTCOMES OF INTEREST FOR SURVEILLANCE PREPAREDNESS
 MINI-SENTINEL METHODS 16 HEALTH OUTCOMES OF INTEREST FOR SURVEILLANCE PREPAREDNESS Prepared by: Cristin Freeman, MPH, MBE, 1,2 Charles Leonard, PharmD, MSCE, 1,2 Patrick Archdeacon, MD, 3 Ryan Carnahan,
MINI-SENTINEL METHODS 16 HEALTH OUTCOMES OF INTEREST FOR SURVEILLANCE PREPAREDNESS Prepared by: Cristin Freeman, MPH, MBE, 1,2 Charles Leonard, PharmD, MSCE, 1,2 Patrick Archdeacon, MD, 3 Ryan Carnahan,
11. NATIONAL DAFNE CLINICAL AND RESEARCH DATABASE
 11. NATIONAL DAFNE CLINICAL AND RESEARCH DATABASE The National DAFNE Clinical and Research database was set up as part of the DAFNE QA programme (refer to section 12) to facilitate Audit and was updated
11. NATIONAL DAFNE CLINICAL AND RESEARCH DATABASE The National DAFNE Clinical and Research database was set up as part of the DAFNE QA programme (refer to section 12) to facilitate Audit and was updated
MINI-SENTINEL PROSPECTIVE SURVEILLANCE PLAN. PROSPECTIVE ROUTINE OBSERVATIONAL MONITORING OF MIRABEGRON Report 1 of 4
 MINI-SENTINEL PROSPECTIVE SURVEILLANCE PLAN PROSPECTIVE ROUTINE OBSERVATIONAL MONITORING OF MIRABEGRON Report 1 of 4 Version 1.0 Prepared by: Charles E. Leonard, PharmD, MSCE 1 ; Marsha E. Reichman, PhD
MINI-SENTINEL PROSPECTIVE SURVEILLANCE PLAN PROSPECTIVE ROUTINE OBSERVATIONAL MONITORING OF MIRABEGRON Report 1 of 4 Version 1.0 Prepared by: Charles E. Leonard, PharmD, MSCE 1 ; Marsha E. Reichman, PhD
Findings from a Mini-Sentinel Medical Product Assessment: Influenza Vaccines and Risk of Febrile Seizures
 Brookings Webinar on Active Medical Product Surveillance Findings from a Mini-Sentinel Medical Product Assessment: Influenza Vaccines and Risk of Febrile Seizures Engelberg Center for Health Care Reform
Brookings Webinar on Active Medical Product Surveillance Findings from a Mini-Sentinel Medical Product Assessment: Influenza Vaccines and Risk of Febrile Seizures Engelberg Center for Health Care Reform
Adverse Outcomes After Hospitalization and Delirium in Persons With Alzheimer Disease
 Adverse Outcomes After Hospitalization and Delirium in Persons With Alzheimer Disease J. Sukanya 05.Jul.2012 Outline Background Methods Results Discussion Appraisal Background Common outcomes in hospitalized
Adverse Outcomes After Hospitalization and Delirium in Persons With Alzheimer Disease J. Sukanya 05.Jul.2012 Outline Background Methods Results Discussion Appraisal Background Common outcomes in hospitalized
Confounding in influenza VE studies in seniors, and possible solutions
 Confounding in influenza VE studies in seniors, and possible solutions Michael L. Jackson Group Health Research Institute 4 th December, 2012 1 Outline Focus is on non-specific outcomes (e.g. community-acquired
Confounding in influenza VE studies in seniors, and possible solutions Michael L. Jackson Group Health Research Institute 4 th December, 2012 1 Outline Focus is on non-specific outcomes (e.g. community-acquired
Leveraging Electronic Health Data in a Multinational Clinical Trial: Early Learnings from the HARMONY- OUTCOMES EHR Ancillary Study
 Leveraging Electronic Health Data in a Multinational Clinical Trial: Early Learnings from the HARMONY- OUTCOMES EHR Ancillary Study Emily O Brien, PhD Assistant Professor Lesley Curtis, PhD Professor Department
Leveraging Electronic Health Data in a Multinational Clinical Trial: Early Learnings from the HARMONY- OUTCOMES EHR Ancillary Study Emily O Brien, PhD Assistant Professor Lesley Curtis, PhD Professor Department
Adjunct Lecturer. National Yang-Ming University, Taiwan. Taipei Veterans General Hospital, Taiwan
 Kuan-Po Peng MD Adjunct Lecturer National Yang-Ming University, Taiwan Taipei Veterans General Hospital, Taiwan Nothing to declare Disclosure Experience from the Taiwan National Health Insurance Research
Kuan-Po Peng MD Adjunct Lecturer National Yang-Ming University, Taiwan Taipei Veterans General Hospital, Taiwan Nothing to declare Disclosure Experience from the Taiwan National Health Insurance Research
NQF-ENDORSED VOLUNTARY CONSENSUS STANDARDS FOR HOSPITAL CARE. Measure Information Form Collected For: CMS Outcome Measures (Claims Based)
 Last Updated: Version 4.3 NQF-ENDORSED VOLUNTARY CONSENSUS STANDARDS FOR HOSPITAL CARE Measure Information Form Collected For: CMS Outcome Measures (Claims Based) Measure Set: CMS Mortality Measures Set
Last Updated: Version 4.3 NQF-ENDORSED VOLUNTARY CONSENSUS STANDARDS FOR HOSPITAL CARE Measure Information Form Collected For: CMS Outcome Measures (Claims Based) Measure Set: CMS Mortality Measures Set
Uses of the NIH Collaboratory Distributed Research Network
 Uses of the NIH Collaboratory Distributed Research Network Jeffrey Brown, PhD for the DRN Team Harvard Pilgrim Care Institute and Harvard Medical School March 11, 2016 The Goal The NIH Collaboratory DRN
Uses of the NIH Collaboratory Distributed Research Network Jeffrey Brown, PhD for the DRN Team Harvard Pilgrim Care Institute and Harvard Medical School March 11, 2016 The Goal The NIH Collaboratory DRN
Nighttime Dosing of Anti-Hypertensive Medications
 Nighttime Dosing of Anti-Hypertensive Medications Gary E. Rosenthal, MD Professor of Internal Medicine & Health Management & Policy Director, Institute for Clinical and Translational Science Elizabeth
Nighttime Dosing of Anti-Hypertensive Medications Gary E. Rosenthal, MD Professor of Internal Medicine & Health Management & Policy Director, Institute for Clinical and Translational Science Elizabeth
Is There An Association?
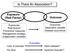 Is There An Association? Exposure (Risk Factor) Outcome Exposures Risk factors Preventive measures Management strategy Independent variables Outcomes Dependent variable Disease occurrence Lack of exercise
Is There An Association? Exposure (Risk Factor) Outcome Exposures Risk factors Preventive measures Management strategy Independent variables Outcomes Dependent variable Disease occurrence Lack of exercise
Supplementary Appendix
 Supplementary Appendix This appendix has been provided by the authors to give readers additional information about their work. Supplement to: Weintraub WS, Grau-Sepulveda MV, Weiss JM, et al. Comparative
Supplementary Appendix This appendix has been provided by the authors to give readers additional information about their work. Supplement to: Weintraub WS, Grau-Sepulveda MV, Weiss JM, et al. Comparative
Propensity Score Methods to Adjust for Bias in Observational Data SAS HEALTH USERS GROUP APRIL 6, 2018
 Propensity Score Methods to Adjust for Bias in Observational Data SAS HEALTH USERS GROUP APRIL 6, 2018 Institute Institute for Clinical for Clinical Evaluative Evaluative Sciences Sciences Overview 1.
Propensity Score Methods to Adjust for Bias in Observational Data SAS HEALTH USERS GROUP APRIL 6, 2018 Institute Institute for Clinical for Clinical Evaluative Evaluative Sciences Sciences Overview 1.
Incorporating Clinical Information into the Label
 ECC Population Health Group LLC expertise-driven consulting in global maternal-child health & pharmacoepidemiology Incorporating Clinical Information into the Label Labels without Categories: A Workshop
ECC Population Health Group LLC expertise-driven consulting in global maternal-child health & pharmacoepidemiology Incorporating Clinical Information into the Label Labels without Categories: A Workshop
Accelerating Patient-Centered Outcomes Research and Methodological Research
 Accelerating Patient-Centered Outcomes Research and Methodological Research Jason Gerson, PhD CER Methods JSM 2016 July 31, 2016 In This Session PCORI Overview Methods Program Overview Methods Program
Accelerating Patient-Centered Outcomes Research and Methodological Research Jason Gerson, PhD CER Methods JSM 2016 July 31, 2016 In This Session PCORI Overview Methods Program Overview Methods Program
Safety Assessment in Clinical Trials and Beyond
 Safety Assessment in Clinical Trials and Beyond Yuliya Yasinskaya, MD Medical Team Leader Division of Anti-Infective Products Center for Drug Evaluation and Research FDA Clinical Investigator Training
Safety Assessment in Clinical Trials and Beyond Yuliya Yasinskaya, MD Medical Team Leader Division of Anti-Infective Products Center for Drug Evaluation and Research FDA Clinical Investigator Training
HEALTH CARE EXPENDITURES ASSOCIATED WITH PERSISTENT EMERGENCY DEPARTMENT USE: A MULTI-STATE ANALYSIS OF MEDICAID BENEFICIARIES
 HEALTH CARE EXPENDITURES ASSOCIATED WITH PERSISTENT EMERGENCY DEPARTMENT USE: A MULTI-STATE ANALYSIS OF MEDICAID BENEFICIARIES Presented by Parul Agarwal, PhD MPH 1,2 Thomas K Bias, PhD 3 Usha Sambamoorthi,
HEALTH CARE EXPENDITURES ASSOCIATED WITH PERSISTENT EMERGENCY DEPARTMENT USE: A MULTI-STATE ANALYSIS OF MEDICAID BENEFICIARIES Presented by Parul Agarwal, PhD MPH 1,2 Thomas K Bias, PhD 3 Usha Sambamoorthi,
MINI-SENTINEL MEDICAL PRODUCT ASSESSMENT A PROTOCOL FOR ASSESSMENT OF DABIGATRAN
 MINI-SENTINEL MEDICAL PRODUCT ASSESSMENT A PROTOCOL FOR ASSESSMENT OF DABIGATRAN Version 3 March 27, 2015 Prior versions: Version 1: December 31, 2013 Version 2: March 18, 2014 Prepared by: Alan S. Go,
MINI-SENTINEL MEDICAL PRODUCT ASSESSMENT A PROTOCOL FOR ASSESSMENT OF DABIGATRAN Version 3 March 27, 2015 Prior versions: Version 1: December 31, 2013 Version 2: March 18, 2014 Prepared by: Alan S. Go,
Jacqueline C. Barrientos, Nicole Meyer, Xue Song, Kanti R. Rai ASH Annual Meeting Abstracts 2015:3301
 Characterization of atrial fibrillation and bleeding risk factors in patients with CLL: A population-based retrospective cohort study of administrative medical claims data in the U.S. Jacqueline C. Barrientos,
Characterization of atrial fibrillation and bleeding risk factors in patients with CLL: A population-based retrospective cohort study of administrative medical claims data in the U.S. Jacqueline C. Barrientos,
Data Fusion: Integrating patientreported survey data and EHR data for health outcomes research
 Data Fusion: Integrating patientreported survey data and EHR data for health outcomes research Lulu K. Lee, PhD Director, Health Outcomes Research Our Development Journey Research Goals Data Sources and
Data Fusion: Integrating patientreported survey data and EHR data for health outcomes research Lulu K. Lee, PhD Director, Health Outcomes Research Our Development Journey Research Goals Data Sources and
Real World Patients: The Intersection of Real World Evidence and Episode of Care Analytics
 PharmaSUG 2018 - Paper RW-05 Real World Patients: The Intersection of Real World Evidence and Episode of Care Analytics David Olaleye and Youngjin Park, SAS Institute Inc. ABSTRACT SAS Institute recently
PharmaSUG 2018 - Paper RW-05 Real World Patients: The Intersection of Real World Evidence and Episode of Care Analytics David Olaleye and Youngjin Park, SAS Institute Inc. ABSTRACT SAS Institute recently
ESM1 for Glucose, blood pressure and cholesterol levels and their relationships to clinical outcomes in type 2 diabetes: a retrospective cohort study
 ESM1 for Glucose, blood pressure and cholesterol levels and their relationships to clinical outcomes in type 2 diabetes: a retrospective cohort study Statistical modelling details We used Cox proportional-hazards
ESM1 for Glucose, blood pressure and cholesterol levels and their relationships to clinical outcomes in type 2 diabetes: a retrospective cohort study Statistical modelling details We used Cox proportional-hazards
HL7 s Version 2 standards and submission to immunization registries
 HL7 s Version 2 standards and submission to immunization registries Alean Kirnak Cochair HL7 Public Health and Emergency Response Workgroup American Immunization Registry Association Software Partners
HL7 s Version 2 standards and submission to immunization registries Alean Kirnak Cochair HL7 Public Health and Emergency Response Workgroup American Immunization Registry Association Software Partners
Influenza Vaccine Safety Monitoring Update
 Influenza Vaccine Safety Monitoring Update Advisory Committee on Immunization Practices October 28, 2010 Tom Shimabukuro, MD, MPH, MBA Immunization Safety Office Division of Healthcare Quality Promotion,
Influenza Vaccine Safety Monitoring Update Advisory Committee on Immunization Practices October 28, 2010 Tom Shimabukuro, MD, MPH, MBA Immunization Safety Office Division of Healthcare Quality Promotion,
TENNCARE Bundled Payment Initiative: Description of Bundle Risk Adjustment for Wave 2 Episodes
 TENNCARE Bundled Payment Initiative: Description of Bundle Risk Adjustment for Wave 2 Episodes Acute COPD exacerbation (COPD); Screening and surveillance colonoscopy (COL); and Outpatient and non-acute
TENNCARE Bundled Payment Initiative: Description of Bundle Risk Adjustment for Wave 2 Episodes Acute COPD exacerbation (COPD); Screening and surveillance colonoscopy (COL); and Outpatient and non-acute
CLINICAL REGISTRIES Use and Emerging Best Practices
 CLINICAL REGISTRIES Use and Emerging Best Practices Tim Friede Department of Medical Statistics University Medical Center Göttingen DZHK (German Center for Cardiovascular Research) OUTLINE Background:
CLINICAL REGISTRIES Use and Emerging Best Practices Tim Friede Department of Medical Statistics University Medical Center Göttingen DZHK (German Center for Cardiovascular Research) OUTLINE Background:
Tobacco Use: Screening & Cessation Intervention
 Tobacco Use: Screening and Cessation Intervention MSSP ACO Measure Tobacco Use: Screening & Cessation Intervention Domain: Preventive Care and Screening ACO 17 PREV- 10 PQRS - 226 NQF 0028 Measure Steward:
Tobacco Use: Screening and Cessation Intervention MSSP ACO Measure Tobacco Use: Screening & Cessation Intervention Domain: Preventive Care and Screening ACO 17 PREV- 10 PQRS - 226 NQF 0028 Measure Steward:
New Patient Questionnaire Pediatric Orthopaedic Surgery
 Page 1 of 5 New Patient Questionnaire Pediatric Orthopaedic Surgery First Name: Last Name: Middle: DOB: Height: Weight: Primary Care Physician/Pediatrician Name: Address: Phone Number: Chief Compliant
Page 1 of 5 New Patient Questionnaire Pediatric Orthopaedic Surgery First Name: Last Name: Middle: DOB: Height: Weight: Primary Care Physician/Pediatrician Name: Address: Phone Number: Chief Compliant
BlueBayCT - Warfarin User Guide
 BlueBayCT - Warfarin User Guide December 2012 Help Desk 0845 5211241 Contents Getting Started... 1 Before you start... 1 About this guide... 1 Conventions... 1 Notes... 1 Warfarin Management... 2 New INR/Warfarin
BlueBayCT - Warfarin User Guide December 2012 Help Desk 0845 5211241 Contents Getting Started... 1 Before you start... 1 About this guide... 1 Conventions... 1 Notes... 1 Warfarin Management... 2 New INR/Warfarin
Status Update on the National Cardiovascular Prevention Guidelines - JNC 8, ATP 4, and Obesity 2
 TABLE OF CONTENTS Status Update on the National Cardiovascular Prevention Guidelines 1 Drosperinone-Containing Oral Contraceptives and Venous Thromboembolism Risk 1-4 P&T Committee Formulary Action 5 Status
TABLE OF CONTENTS Status Update on the National Cardiovascular Prevention Guidelines 1 Drosperinone-Containing Oral Contraceptives and Venous Thromboembolism Risk 1-4 P&T Committee Formulary Action 5 Status
Evolution of Active Surveillance: An Industry Perspective
 Evolution of Active Surveillance: An Industry Perspective Midwest Biopharmaceutical Statistics Workshop May 21, 2013 Stephen Motsko, PharmD, PhD Ken Hornbuckle, DVM, PhD Office of Risk Management and Pharmacoepidemiology
Evolution of Active Surveillance: An Industry Perspective Midwest Biopharmaceutical Statistics Workshop May 21, 2013 Stephen Motsko, PharmD, PhD Ken Hornbuckle, DVM, PhD Office of Risk Management and Pharmacoepidemiology
Discontinuation and restarting in patients on statin treatment: prospective open cohort study using a primary care database
 open access Discontinuation and restarting in patients on statin treatment: prospective open cohort study using a primary care database Yana Vinogradova, 1 Carol Coupland, 1 Peter Brindle, 2,3 Julia Hippisley-Cox
open access Discontinuation and restarting in patients on statin treatment: prospective open cohort study using a primary care database Yana Vinogradova, 1 Carol Coupland, 1 Peter Brindle, 2,3 Julia Hippisley-Cox
NQF-ENDORSED VOLUNTARY CONSENSUS STANDARD FOR HOSPITAL CARE. Measure Information Form Collected For: CMS Outcome Measures (Claims Based)
 Last Updated: Version 4.3 NQF-ENDORSED VOLUNTARY CONSENSUS STANDARD FOR HOSPITAL CARE Measure Information Form Collected For: CMS Outcome Measures (Claims Based) Measure Set: CMS Readmission Measures Set
Last Updated: Version 4.3 NQF-ENDORSED VOLUNTARY CONSENSUS STANDARD FOR HOSPITAL CARE Measure Information Form Collected For: CMS Outcome Measures (Claims Based) Measure Set: CMS Readmission Measures Set
PubH 7405: REGRESSION ANALYSIS. Propensity Score
 PubH 7405: REGRESSION ANALYSIS Propensity Score INTRODUCTION: There is a growing interest in using observational (or nonrandomized) studies to estimate the effects of treatments on outcomes. In observational
PubH 7405: REGRESSION ANALYSIS Propensity Score INTRODUCTION: There is a growing interest in using observational (or nonrandomized) studies to estimate the effects of treatments on outcomes. In observational
Documenting Patient Immunization. New Brunswick 2018/19
 Documenting Patient Immunization New Brunswick 2018/19 Table of Contents Documenting Patient Immunization New Brunswick...3 Immunization Module Features...4 Configuration...5 Marketing Message Setup...6
Documenting Patient Immunization New Brunswick 2018/19 Table of Contents Documenting Patient Immunization New Brunswick...3 Immunization Module Features...4 Configuration...5 Marketing Message Setup...6
Progress from the Patient-Centered Outcomes Research Institute (PCORI)
 Progress from the Patient-Centered Outcomes Research Institute (PCORI) Anne Beal, Chief Operating Officer of the Patient-Centered Outcomes Research Institute Sharon-Lise Normand, Vice Chair, Methodology
Progress from the Patient-Centered Outcomes Research Institute (PCORI) Anne Beal, Chief Operating Officer of the Patient-Centered Outcomes Research Institute Sharon-Lise Normand, Vice Chair, Methodology
Statistical Considerations for Post Market Surveillance
 Statistical Considerations for Post Market Surveillance 30 th International Conference on Phamacoepidemiology and Therapeutic Risk Management Ted Lystig, Ph.D. MEDTRONIC INC. Distinguished Statistician
Statistical Considerations for Post Market Surveillance 30 th International Conference on Phamacoepidemiology and Therapeutic Risk Management Ted Lystig, Ph.D. MEDTRONIC INC. Distinguished Statistician
Public Health Outcomes Framework Key changes and updates for Peterborough: November 2017
 Public Health Outcomes Framework Key changes and updates for Peterborough: November 2017 Introduction and overview The Department of Health first published the Public Health Outcomes Framework (PHOF) for
Public Health Outcomes Framework Key changes and updates for Peterborough: November 2017 Introduction and overview The Department of Health first published the Public Health Outcomes Framework (PHOF) for
A European model for the Automatic Production of Standardized Performance Indicators: the BIRO statistical engine
 EUropean Best Information through Regional Outcomes in Diabetes A European model for the Automatic Production of Standardized Performance Indicators: the BIRO statistical engine Fabrizio Carinci Serectrix
EUropean Best Information through Regional Outcomes in Diabetes A European model for the Automatic Production of Standardized Performance Indicators: the BIRO statistical engine Fabrizio Carinci Serectrix
Mahoning County Public Health. Epidemiology Response Annex
 Mahoning County Public Health Epidemiology Response Annex Created: May 2006 Updated: February 2015 Mahoning County Public Health Epidemiology Response Annex Table of Contents Epidemiology Response Document
Mahoning County Public Health Epidemiology Response Annex Created: May 2006 Updated: February 2015 Mahoning County Public Health Epidemiology Response Annex Table of Contents Epidemiology Response Document
SENTINEL METHODS WHITE PAPER EXPLORING THE FEASIBILITY OF CONDUCTING VACCINE EFFECTIVENESS STUDIES IN SENTINEL S PRISM PROGRAM
 SENTINEL METHODS WHITE PAPER EXPLORING THE FEASIBILITY OF CONDUCTING VACCINE EFFECTIVENESS STUDIES IN SENTINEL S PRISM PROGRAM Prepared by: Catherine A. Panozzo, MPH, PhD, 1 Maria Said, MD, MHS, 2 Deepa
SENTINEL METHODS WHITE PAPER EXPLORING THE FEASIBILITY OF CONDUCTING VACCINE EFFECTIVENESS STUDIES IN SENTINEL S PRISM PROGRAM Prepared by: Catherine A. Panozzo, MPH, PhD, 1 Maria Said, MD, MHS, 2 Deepa
ADVANCING CANCER RESEARCH THROUGH A VIRTUAL POOLED REGISTRY. Dennis Deapen, DrPH NAACCR Annual Meeting June 16, 2015 Charlotte, NC
 ADVANCING CANCER RESEARCH THROUGH A VIRTUAL POOLED REGISTRY Dennis Deapen, DrPH NAACCR Annual Meeting June 16, 2015 Charlotte, NC 1 THE OPPORTUNITY Cancer epidemiology cohort studies may have participants
ADVANCING CANCER RESEARCH THROUGH A VIRTUAL POOLED REGISTRY Dennis Deapen, DrPH NAACCR Annual Meeting June 16, 2015 Charlotte, NC 1 THE OPPORTUNITY Cancer epidemiology cohort studies may have participants
Challenges in design and analysis of large register-based epidemiological studies
 FMS/DSBS autumn meeting 2014 Challenges in design and analysis of large register-based epidemiological studies Caroline Weibull & Anna Johansson Department of Medical Epidemiology and Biostatistics (MEB)
FMS/DSBS autumn meeting 2014 Challenges in design and analysis of large register-based epidemiological studies Caroline Weibull & Anna Johansson Department of Medical Epidemiology and Biostatistics (MEB)
Registry to Evaluate Early and Long-Term PAH Disease Management. Transforming PAH Experience Into Knowledge
 Registry to Evaluate Early and Long-Term PAH Disease Management Transforming PAH Experience Into Knowledge Protocol and Goals of the REVEAL Registry Robyn J. Barst, MD REVEAL Study Objectives To characterize
Registry to Evaluate Early and Long-Term PAH Disease Management Transforming PAH Experience Into Knowledge Protocol and Goals of the REVEAL Registry Robyn J. Barst, MD REVEAL Study Objectives To characterize
Confounding by indication developments in matching, and instrumental variable methods. Richard Grieve London School of Hygiene and Tropical Medicine
 Confounding by indication developments in matching, and instrumental variable methods Richard Grieve London School of Hygiene and Tropical Medicine 1 Outline 1. Causal inference and confounding 2. Genetic
Confounding by indication developments in matching, and instrumental variable methods Richard Grieve London School of Hygiene and Tropical Medicine 1 Outline 1. Causal inference and confounding 2. Genetic
Propensity Score Methods for Estimating Causality in the Absence of Random Assignment: Applications for Child Care Policy Research
 2012 CCPRC Meeting Methodology Presession Workshop October 23, 2012, 2:00-5:00 p.m. Propensity Score Methods for Estimating Causality in the Absence of Random Assignment: Applications for Child Care Policy
2012 CCPRC Meeting Methodology Presession Workshop October 23, 2012, 2:00-5:00 p.m. Propensity Score Methods for Estimating Causality in the Absence of Random Assignment: Applications for Child Care Policy
Costing report: Lipid modification Implementing the NICE guideline on lipid modification (CG181)
 Putting NICE guidance into practice Costing report: Lipid modification Implementing the NICE guideline on lipid modification (CG181) Published: July 2014 This costing report accompanies Lipid modification:
Putting NICE guidance into practice Costing report: Lipid modification Implementing the NICE guideline on lipid modification (CG181) Published: July 2014 This costing report accompanies Lipid modification:
A re-randomisation design for clinical trials
 Kahan et al. BMC Medical Research Methodology (2015) 15:96 DOI 10.1186/s12874-015-0082-2 RESEARCH ARTICLE Open Access A re-randomisation design for clinical trials Brennan C Kahan 1*, Andrew B Forbes 2,
Kahan et al. BMC Medical Research Methodology (2015) 15:96 DOI 10.1186/s12874-015-0082-2 RESEARCH ARTICLE Open Access A re-randomisation design for clinical trials Brennan C Kahan 1*, Andrew B Forbes 2,
Lead the Way with Advanced Care Management. Workbook
 Lead the Way with Advanced Care Management Workbook TPCA Training 10.2018 Section 1: Using i2itracks for Chronic Disease Management Chronic Disease Tracking in 2018 Disease Management Definition A system
Lead the Way with Advanced Care Management Workbook TPCA Training 10.2018 Section 1: Using i2itracks for Chronic Disease Management Chronic Disease Tracking in 2018 Disease Management Definition A system
Part 1: The Rationale for Sentinel Surveillance to Monitor Seasonal and Pandemic Influenza
 1 Part 1: The Rationale for Sentinel Surveillance to Monitor Seasonal and Pandemic Influenza Participant Guide This section should take 3 hours Instructions During this session the moderator will lead
1 Part 1: The Rationale for Sentinel Surveillance to Monitor Seasonal and Pandemic Influenza Participant Guide This section should take 3 hours Instructions During this session the moderator will lead
Documenting Patient Immunization. Ontario 2018/19
 Documenting Patient Immunization Ontario 2018/19 Table of Contents Documenting Patient Immunization Ontario...3 Immunization Module Features...4 Configuration...5 Marketing Message Setup...6 Paper Mode...9
Documenting Patient Immunization Ontario 2018/19 Table of Contents Documenting Patient Immunization Ontario...3 Immunization Module Features...4 Configuration...5 Marketing Message Setup...6 Paper Mode...9
