GLOBALTRAINING. IMPLANON NXT : Reference Guide
|
|
|
- Joel Russell
- 6 years ago
- Views:
Transcription
1 GLOBALTRAINING IMPLANON NXT : Reference Guide
2 IMPLANON NXT (etonogestrel implant): Clinical Information Applicator for IMPLANON NXT A double-blind, parallel-group, randomized trial in 108 women has conclusively shown that IMPLANON and IMPLANON NXT are bioequivalent. Therefore, the trial data for IMPLANON NXT used to support the statements made throughout this document are accurate and relevant for both products. IMPLANON NXT is a subdermal, long-acting, progestagenonly contraceptive effective for up to 3 years. IMPLANON NXT is radiopaque and comparable to IMPLANON (etonogestrel implant). 1 The mechanisms of action of IMPLANON NXT consist of inhibition of ovulation and increases in the viscosity of cervical mucus. Efficacy does not depend on daily, weekly, or monthly self-administration. IMPLANON NXT is effective from day one, when inserted according to instructions in the label. No method of contraception is 100% effective IMPLANON and IMPLANON NXT have been shown to be over 99% effective when inserted correctly. 2 Studies show a continuation rate of 82% after 1 year of use. 3 11% of women studied discontinued IMPLANON due to bleeding irregularities. Bleeding irregularities may include amenorrhea or infrequent, frequent, and/or prolonged bleeding. Information, counseling, and the use of a bleeding diary can improve the woman s acceptance of a bleeding pattern. Following removal of IMPLANON NXT, the hormone is below detectable levels within 7 days. 4 Bone Mineral Density A comparative study of IMPLANON and a non-hormonal IUD showed that bone mineral density remained unaltered over 2 years, with no detectable difference between users of each contraceptive method. Adverse Events Headache Weight increase Acne Breast pain Irregular menstruation Vaginal infections Please refer to the regulatory-approved full Prescribing Information for complete list of adverse events. Contraindications Use of I MPLANON NXT is contraindicated in patients with: Known or suspected pregnancy Active venous thromboembolic disorder Known or suspected sex steroid-sensitive malignancies Presence of history of liver tumors (benign or malignant) Presence or history of severe hepatic disease as long as liver function values have not returned to normal Undiagnosed vaginal bleeding Hypersensitivity to the active substance or to any of the excipients of I MPLANON NXT Interactions The Prescribing Information of concomitant medications used by the patient should be consulted to identify potential interactions. Interactions can occur with medicinal products that induce hepatic enzymes, specifically cytochrome P450 enzymes, which can result in increased clearance of sex hormones (eg, phenytoin, phenobarbital, primidone, bosentan, carbamazapine, rifampicin, and HIV medication [eg, ritonavir, nelfinavir, nevirapine], and possibly also oxcarbazepine, topiramate, felbamate, griseofulvin, and the herbal remedy St. John s wort). Also, HIV protease (eg, ritonavir, nelfinavir), non-nucleoside reverse transcriptase inhibitors (eg, nevirapine, efavirenz), and combinations of them have been reported to potentially affect hepatic metabolism. top view side view bottom view Breast Milk In clinical studies, indicate that IMPLANON NXT does not influence the quantity or quality (protein, lactose, or fat concentrations) of breast milk in nursing mothers. However, small amounts of etonogestrel are excreted in breast milk. Evaluation of growth and physical and psychomotor development of 38 children breast-fed for an average of 14 months did not indicate any differences in comparison to nursing infants whose mothers used an IUD (n=33); each group was followed-up to 36 months of age. Based on the available data, IMPLANON NXT may be used during lactation and should be inserted after the 4th postpartum week. Women on treatment with any of the above mentioned drugs should use a non-hormonal contraceptive method in addition to I MPLANON NXT. Please refer to the regulatory-approved full Prescribing Information for additional interactions. Pregnancy I MPLANON NXT is not indicated during pregnancy. If pregnancy occurs during use of I MPLANON NXT, the implant should be removed. Animal studies have shown that very high doses of progestagenic substances may cause masculinization of female fetuses. Extensive epidemiological studies have revealed neither an increased risk of birth defects in children born to women who used oral contraceptives (OCs) prior to pregnancy, nor a teratogenic effect when OCs were inadvertently used during pregnancy. Although this probably applies to all OCs, it is not clear whether this is also the case for I MPLANON NXT. Key Points for Patient Counseling Women are likely to have changes in their menstrual bleeding pattern with IMPLANON NXT. These may include changes in bleeding frequency, intensity, or duration; however, the bleeding pattern experienced during the first 3 months is broadly predictive of future bleeding patterns for many women. Amenorrhea was reported in about 1 of 5 women, while another 1 of 5 women reported frequent and/or prolonged bleeding. Dysmenorrhea tended to improve while using IMPLANON NXT. Appropriate counseling may make bleeding changes more acceptable for women. Key counseling points include: Discussion of the likelihood of alterations in bleeding patterns Discussion of the risks, benefits, and possible side effects Explain the insertion and removal procedures; emphasizing that the implant should be palpable and that scars or complications may occur. If possible, provide patient education materials. Allow sufficient time for the patient to review the regulatoryapproved Patient Information and Prescribing Information, consider options, and ask questions. PAGE: 1 References: 1 Schnabel 2012, 2 Graesslin 2008, 3 Blumenthal 2008, 4 Davies 1993 PAGE: 2
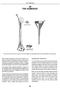
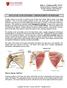
3 How to Insert IMPLANON NXT Guiding Mark Insertion Site Figure 1 Figure 2 Medial Epicondyle Insertion of IMPLANON NXT should be performed under aseptic conditions and only by a qualified health care provider who is familiar with the procedure. Insertion of the implant should only be performed with the preloaded applicator strictly in accordance with the insertion instructions outlined in the regulatory-approved Prescribing Information. It is recommended that the health care provider is in a seated position during the entire insertion procedure so that the insertion site and the movement of the needle can be clearly seen. Have the woman lie on her back on the examination table with her non-dominant arm flexed at the elbow and externally rotated so that her wrist is parallel to her ear or her hand is positioned next to her head (Figure 1). Identify the insertion site, which is at the inner side of the non-dominant upper arm about 8 to 10 cm (3 to 4 inches) above the medial epicondyle of the humerus. The implant should be inserted subdermally just under the skin to avoid the large blood vessels and nerves that lie deeper in the subcutaneous tissue in the sulcus between the triceps and biceps muscles. If the applicator is not kept in the same position during this procedure or if the purple slider is not completely moved to the back, the implant will not be inserted properly. Always verify the presence of the implant in the woman s arm immediately after insertion by palpation. By palpating both ends of the implant, you should be able to confirm the presence of the 4 cm rod (Figure 8). If you cannot feel the implant or doubt its presence: Check the applicator. The needle should be fully retracted and only the purple tip of the obturator should be visible. Use other methods to confirm its presence. Suitable methods are: 2-dimensional X-ray, X-ray computerized tomography (CT scan), ultrasound scanning (USS) with a high-frequency linear array transducer (10 MHz or greater), or magnetic resonance imaging (MRI). Prior to the application of X-ray CT, USS, or MRI for the localization of the implant, it is recommended to consult the local supplier of IMPLANON NXT for instructions. In case these imaging methods fail, it is advised to verify the presence of the implant by measuring the etonogestrel level in a blood sample of the subject. In this case, the local supplier will provide the appropriate procedure. Until you have verified the presence of the implant, a non-hormonal contraceptive method must be used. Apply a small adhesive bandage over the insertion site. Request that the woman palpate the implant. Apply sterile gauze with a pressure bandage to minimize bruising. The woman may remove the pressure bandage in 24 hours and the small bandage over the insertion site after 3 to 5 days. Complete the User Card and give it to the woman to keep. Complete the adhesive labels and affix to the woman s medical record. The applicator is for single use only and must be adequately disposed of, in accordance with local regulations for the handling of biohazardous waste. Figure 3 Figure 4 Figure 5 Figure 6 Make 2 marks with a sterile marker; first, mark the spot where the implant will be inserted, and second, mark a spot a few centimeters proximal to the first mark (Figure 2). This second mark will later serve as a direction guide during insertion. Clean the insertion site with an antiseptic solution. Anesthetize the insertion area (for example with anesthetic spray or by injecting 2 ml of 1% lidocaine just under the skin along the planned insertion tunnel). Remove the sterile preloaded disposable applicator for IMPLANON NXT carrying the implant from its blister. Hold the applicator just above the needle at the textured surface area and remove the transparent protection cap from the needle which contains the implant (Figure 3). If the cap does not come off easily, the applicator should not be used. You may see the white-colored implant by looking into the tip of the needle. Do not touch the purple slider until you have fully inserted the needle subdermally, as it will retract the needle and release the implant from the applicator. With your free hand, stretch the skin around the insertion site with thumb and index finger (Figure 4). Puncture the kin with the tip of the needle angled about 30 (Figure 5). Lower the applicator to a horizontal position. While lifting the skin with the tip of the needle, slide the needle to its full length. You may feel slight resistance but do not exert excessive force (Figure 6). If the needle is not inserted to its full length, the implant will not be inserted properly. Localizing IMPLANON NXT Localization is an essential component of the insertion and removal process. Palpation is the first step in the localization process. Always localize by palpation: Immediately after insertion Immediately prior to removal If the patient has retained and can provide a completed User Card, review it to determine: Whether the implant is radiopaque (IMPLANON NXT) or not radiopaque (IMPLANON) The location noted when the implant was originally inserted If the implant is not palpable after insertion, check the applicator. The needle should be fully retracted and only the purple tip of the obturator should be visible. Confirm its presence in the arm with imaging techniques (X-ray, CT, USS, MRI) as soon as possible. The patient must use a back-up method of contraception until the presence of IMPLANON NXT has been confirmed. If these imaging methods fail to locate the implant, etonogestrel blood level determination can be used for verification of the presence of the implant. Please contact your local supplier for further guidance. IMPLANON NXT is radiopaque and is visible on X-rays and CT images as well as via MRI or ultrasound imaging techniques. NOTE: IMPLANON is not radiopaque and is not visible on X-ray or CT images. Attempts to localize a nonradiopaque implant via X-ray or CT will not be successful. USS characteristics of IMPLANON NXT: Sharp acoustic shadow below the implant in the transverse position Implant is a small echogenic spot (2 mm) when viewed in the transverse position MRI Implant appears as a round hypodense area of 2-mm diameter, over a length of 4 cm. It is especially important to differentiate the implant from blood vessels. X-Ray The implant will be visible as a 4-cm long, 2-mm wide white stick. CT scan The implant will be visible as a round 2-mm wide radiopaque spot over a length of 4 cm. Figure 7 Figure 8 While keeping the applicator in the same position and the needle inserted to its full length, unlock the purple slider by pushing it slightly down. Move the slider fully back until it stops, leaving the implant now in its final subdermal position and locking the needle inside the body of the applicator (Figure 7). PAGE: 3 PAGE: 4
4 Removing IMPLANON NXT Indications for removal Patient request Medical indication At the end of 3 years of use If the patient does not wish to become pregnant, another contraceptive method should be started immediately (return to normal menstrual cycle may be very rapid). Before initiating the removal procedure, consult the User Card for the location of the implant. Verify the exact location of the implant in the arm by palpation. Figure A Figure C Figure B Figure D If the implant is not palpable, 2-dimensional X-ray can be performed to verify its presence. A non-palpable implant should always be first located prior to removal. Suitable methods for localization include X-ray computer tomography (CT), ultrasound scanning (USS) with a high-frequency linear array transducer (10 MHZ or greater), or magnetic resonance imaging (MRI). If these imaging methods fail to locate the implant, etonogestrel determination can be used for verification of the presence of the implant. Please contact your local supplier for further guidance. After localization of a non-palpable implant, consider conducting removal with ultrasound guidance. There have been occasional reports of migration of the implant; usually this involves minor movement relative to the original position unless inserted too deeply. This may complicate localization of the implant by palpation, USS, and/or MRI, and removal may require a larger incision and more time. Removal of IMPLANON NXT should be performed under aseptic conditions and only by a health care provider who is familiar with the removal technique. Exploratory surgery without knowledge of the exact localization of the implant is strictly discouraged. Removal of deeply inserted implants should be conducted with caution in order to prevent damage to deeper neural or vascular structures in the arm and should be performed only by health care providers familiar with the anatomy of the arm. If the implant cannot be removed, please contact your local supplier for further guidance. Clean the site where the incision will be made and apply an antiseptic. Locate the implant by palpation and mark the distal end (end closest to the elbow), for example, with a sterile marker (Figure A). Anesthetize the arm, for example, with 0.5 to 1 ml 1% lidocaine at the marked site where the incision will be made (Figure B). Be sure to inject the local anesthetic under the implant to keep it close to the skin surface. Push down the proximal end of the implant (Figure C) to stabilize it; a bulge may appear indicating the distal end of implant. Starting at the distal tip of the implant, make a longitudinal incision of 2 mm toward the elbow. Gently push the implant towards the incision until the tip is visible. Grasp the implant with forceps (preferably curved mosquito forceps) and remove the implant (Figure D). If the implant is encapsulated, make an incision into the tissue sheath and then remove the implant with forceps. (Figures E and F). If the tip of the implant does not become visible in the incision, gently insert a forceps into the incision (Figure G). Flip the forceps over into your other hand (Figure H). With a second pair of forceps carefully dissect the tissue around the implant and grasp the implant (Figure I). The implant can then be removed. Confirm that the entire rod, which is 4 cm long, has been removed by measuring its length. There have been occasional reports of migration of the implant. This may complicate localization of the implant by palpation, USS, and/or MRI, and removal may require a larger incision and more time. If the woman would like to continue using IMPLANON NXT, a new implant may be inserted immediately after the old implant is removed (see How to Replace IMPLANON NXT ) Close the incision with a steri-strip and apply an adhesive bandage. Apply sterile gauze with a pressure bandage to minimize bruising. If the woman does not wish to continue using IMPLANON NXT and does not want to become pregnant, another contraceptive method should be recommended. How to Replace IMPLANON NXT Immediate replacement can be done after removal of the previous implant and is similar to the insertion procedure described in the section How to Insert Implanon NXT. The new implant may be inserted in the same arm, and through the same incision from which the previous implant was removed. If the same incision is being used to insert a new implant, anesthetize the insertion site (eg, 2 ml lidocaine [1%]) applied just under the skin commencing at the removal incision along the insertion canal. Then follow the subsequent steps in the insertion instructions. Before administering IMPLANON NXT, please read the Prescribing Information. Figure G Figure H Figure I Figure E Figure F PAGE: 5 PAGE: 6
5 Before administering IMPLANON NXT, please read the Prescribing Information. Copyright 2013 Merck Sharp & Dohme B.V., a subsidiary of Merck & Co., Inc., Whitehouse Station, NJ, USA. All rights reserved. WOMN /13
GLOBALTRAINING. IMPLANON : Reference Guide
 GLOBALTRAINING IMPLANON : Reference Guide IMPLANON : Clinical Information IMPLANON is a subdermal, long-acting, progestagen-only contraceptive effective for up to 3 years. The mechanisms of action of IMPLANON
GLOBALTRAINING IMPLANON : Reference Guide IMPLANON : Clinical Information IMPLANON is a subdermal, long-acting, progestagen-only contraceptive effective for up to 3 years. The mechanisms of action of IMPLANON
Jadelle Contraceptive Implants up to 5 years Insertion and Removal
 Contraceptive Implants up to 5 years Bayer AG Global HealthCare Programs Family Planning 13342 Berlin, Germany www.bayer.com September 2017 Global HealthCare Programs Family Planning Contraceptive Independence»»
Contraceptive Implants up to 5 years Bayer AG Global HealthCare Programs Family Planning 13342 Berlin, Germany www.bayer.com September 2017 Global HealthCare Programs Family Planning Contraceptive Independence»»
Implanon NXT Insertion and Removal Training Course for Registered Nurses & Midwives Course Guide
 Implanon NXT Insertion and Removal Training Course for Registered Nurses & Midwives Course Guide DELIVERED BY 2 July 2015 CONTENTS Learning Outcomes 4 Course Structure 5 Workplace Scope of Practice 5 Assessment
Implanon NXT Insertion and Removal Training Course for Registered Nurses & Midwives Course Guide DELIVERED BY 2 July 2015 CONTENTS Learning Outcomes 4 Course Structure 5 Workplace Scope of Practice 5 Assessment
Emergency contraception is an occasional method. It should in no instance replace a regular contraceptive method.
 1. NAME OF THE MEDICINAL PRODUCT: Levonorgestrel Tablets 1.5 mg 2. QUALITATIVE AND QUANTITATIVE COMPOSITION: Each tablet contains levonorgestrel 1.5 mg. Excipient with known effect: Each tablet contains
1. NAME OF THE MEDICINAL PRODUCT: Levonorgestrel Tablets 1.5 mg 2. QUALITATIVE AND QUANTITATIVE COMPOSITION: Each tablet contains levonorgestrel 1.5 mg. Excipient with known effect: Each tablet contains
PROBUPHINE is available only through a restricted program called the PROBUPHINE REMS Program. (5.2)
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use PROBUPHINE safely and effectively. See full prescribing information for PROBUPHINE. PROBUPHINE (buprenorphine)
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use PROBUPHINE safely and effectively. See full prescribing information for PROBUPHINE. PROBUPHINE (buprenorphine)
ARROW EZ-IO Intraosseous Vascular Access System Procedure Template
 ARROW EZ-IO Intraosseous Vascular Access System Procedure Template PURPOSE To provide procedural guidance for establishment of intraosseous vascular access using the ARROW EZ-IO Intraosseous Vascular Access
ARROW EZ-IO Intraosseous Vascular Access System Procedure Template PURPOSE To provide procedural guidance for establishment of intraosseous vascular access using the ARROW EZ-IO Intraosseous Vascular Access
WARNINGS AND PRECAUTIONS
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use PROBUPHINE safely and effectively. See full prescribing information for PROBUPHINE. PROBUPHINE (buprenorphine)
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use PROBUPHINE safely and effectively. See full prescribing information for PROBUPHINE. PROBUPHINE (buprenorphine)
Draft Label HIGHLIGHTS OF PRESCRIBING INFORMATION INDICATIONS AND USAGE
 1 2 3 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use PROBUPHINE safely and effectively. See full prescribing information for PROBUPHINE. PROBUPHINE
1 2 3 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use PROBUPHINE safely and effectively. See full prescribing information for PROBUPHINE. PROBUPHINE
Title: EZ-IO. Effective Date: January SOG Number: EMS Rescinds:
 S O G Title: EZ-IO Effective Date: January 2010 SOG Number: EMS - 25 Rescinds: Scope: Providers Authorized are AIC s in the following certifications EMT-I and EMT-P who have been trained and cleared by
S O G Title: EZ-IO Effective Date: January 2010 SOG Number: EMS - 25 Rescinds: Scope: Providers Authorized are AIC s in the following certifications EMT-I and EMT-P who have been trained and cleared by
Example CLINICAL GUIDELINES for Postpartum IUD insertion
 Example CLINICAL GUIDELINES for Postpartum IUD insertion Postpartum Intrauterine Device Insertion 1.0 Indications: 1.1 Insertion of an intrauterine device (IUD) for long-acting reversible contraception
Example CLINICAL GUIDELINES for Postpartum IUD insertion Postpartum Intrauterine Device Insertion 1.0 Indications: 1.1 Insertion of an intrauterine device (IUD) for long-acting reversible contraception
New Zealand Datasheet
 New Zealand Datasheet 1 PRODUCT NAME POSTINOR-1 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Levonorgestrel 1.5 mg 3 PHARMACEUTICAL FORM Each round white tablet contains 1.5 mg of levonorgestrel. The tablet
New Zealand Datasheet 1 PRODUCT NAME POSTINOR-1 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Levonorgestrel 1.5 mg 3 PHARMACEUTICAL FORM Each round white tablet contains 1.5 mg of levonorgestrel. The tablet
EndoRelease ENDOSCOPIC CUBITAL TUNNEL RELEASE SYSTEM
 EndoRelease ENDOSCOPIC CUBITAL TUNNEL RELEASE SYSTEM SURGICAL TECHNIQUE Up p e r Ex t r e m i t y So l u t i o n s ENDOSCOPIC CUBITAL TUNNEL RELEASE SYSTEM Description: The EndoRelease Endoscopic Cubital
EndoRelease ENDOSCOPIC CUBITAL TUNNEL RELEASE SYSTEM SURGICAL TECHNIQUE Up p e r Ex t r e m i t y So l u t i o n s ENDOSCOPIC CUBITAL TUNNEL RELEASE SYSTEM Description: The EndoRelease Endoscopic Cubital
Patient Guide Levonorgestrel and Ethinyl Estradiol Tablets USP (0.1 mg/0.02 mg) and Ethinyl Estradiol Tablets USP (0.
 Patient Guide Levonorgestrel and Ethinyl Estradiol Tablets USP (0.1 mg/0.02 mg) and Ethinyl Estradiol Tablets USP (0.01 mg) Rx Only This product (like all oral contraceptives) is intended to prevent pregnancy.
Patient Guide Levonorgestrel and Ethinyl Estradiol Tablets USP (0.1 mg/0.02 mg) and Ethinyl Estradiol Tablets USP (0.01 mg) Rx Only This product (like all oral contraceptives) is intended to prevent pregnancy.
Example Clinical Guideline for Immediate Postpartum LARC Insertion
 Example Clinical Guideline for Immediate Postpartum LARC Insertion RATIONALE Delay in contraceptive provision until the six week postpartum appointment can leave some women at risk for rapid repeat pregnancy.
Example Clinical Guideline for Immediate Postpartum LARC Insertion RATIONALE Delay in contraceptive provision until the six week postpartum appointment can leave some women at risk for rapid repeat pregnancy.
Contraceptives. Kim Dawson October 2010
 Contraceptives Kim Dawson October 2010 Objectives: You will learn about: The about the different methods of birth control. How to use each method of birth control. Emergency contraception What are they?
Contraceptives Kim Dawson October 2010 Objectives: You will learn about: The about the different methods of birth control. How to use each method of birth control. Emergency contraception What are they?
CAREFULLY READ ALL INSTRUCTIONS PRIOR TO USE
 CAREFULLY READ ALL INSTRUCTIONS PRIOR TO USE INDICATIONS FOR USE The LATERA Absorbable Nasal Implant is indicated for supporting upper and lower lateral nasal cartilage. CAUTION: Federal law restricts
CAREFULLY READ ALL INSTRUCTIONS PRIOR TO USE INDICATIONS FOR USE The LATERA Absorbable Nasal Implant is indicated for supporting upper and lower lateral nasal cartilage. CAUTION: Federal law restricts
If viewing a printed copy of this policy, please note it could be expired. Got to to view current policies.
 If viewing a printed copy of this policy, please note it could be expired. Got to www.fairview.org/fhipolicies to view current policies. Department Policy Code: D: PC-5575 Entity: Fairview Pharmacy Services
If viewing a printed copy of this policy, please note it could be expired. Got to www.fairview.org/fhipolicies to view current policies. Department Policy Code: D: PC-5575 Entity: Fairview Pharmacy Services
The tablet is white, round, biconvex and 5 mm in diameter. On one side it is coded KV above 2 and on
 1. NAME OF THE MEDICINAL PRODUCT Cerazette 75 microgram film-coated tablets. 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each tablet contains 75 microgram desogestrel. Excipient (s) with known effect:
1. NAME OF THE MEDICINAL PRODUCT Cerazette 75 microgram film-coated tablets. 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each tablet contains 75 microgram desogestrel. Excipient (s) with known effect:
SARASOTA MEMORIAL HOSPITAL. NURSING PROCEDURE INTRAOSSEOUS NEEDLE: INSERTION, CARE, AND REMOVAL (inv08) 12/18 12/18 1 of 7 RESPONSIBILITY:
 SARASOTA MEMORIAL HOSPITAL TITLE: ISSUED FOR: NURSING PROCEDURE INTRAOSSEOUS NEEDLE: INSERTION, CARE, AND REMOVAL (inv08) Nursing DATE: REVIEWED: PAGES: 12/18 12/18 1 of 7 RESPONSIBILITY: PS1094 Insertion-
SARASOTA MEMORIAL HOSPITAL TITLE: ISSUED FOR: NURSING PROCEDURE INTRAOSSEOUS NEEDLE: INSERTION, CARE, AND REMOVAL (inv08) Nursing DATE: REVIEWED: PAGES: 12/18 12/18 1 of 7 RESPONSIBILITY: PS1094 Insertion-
Stratis INSTRUCTIONS FOR USE. Needle-free Injection System. 0.5mL volume (+/-5%)
 Stratis Needle-free Injection System INSTRUCTIONS FOR USE 0.5mL volume (+/-5%) Stratis English Symbols Glossary (Note: All symbols are derived from ISO 15223-1, Medical Devices - Symbols to be used with
Stratis Needle-free Injection System INSTRUCTIONS FOR USE 0.5mL volume (+/-5%) Stratis English Symbols Glossary (Note: All symbols are derived from ISO 15223-1, Medical Devices - Symbols to be used with
MODUS TABLETS. Medroxyprogesterone Tablets IP
 For the use only of a Registered Medical Practitioner or a Hospital or a Laboratory MODUS TABLETS Medroxyprogesterone Tablets IP QUALITATIVE AND QUANTITATIVE COMPOSITION MODUS TABLET 2.5 mg Each uncoated
For the use only of a Registered Medical Practitioner or a Hospital or a Laboratory MODUS TABLETS Medroxyprogesterone Tablets IP QUALITATIVE AND QUANTITATIVE COMPOSITION MODUS TABLET 2.5 mg Each uncoated
INSTRUCTIONS FOR USE TYMLOS (tim lows ) (abaloparatide) injection, for subcutaneous use
 INSTRUCTIONS FOR USE TYMLOS (tim lows ) (abaloparatide) injection, for subcutaneous use Instructions for Use Read and follow this Instructions for Use so that you inject TYMLOS pen the right way. Call
INSTRUCTIONS FOR USE TYMLOS (tim lows ) (abaloparatide) injection, for subcutaneous use Instructions for Use Read and follow this Instructions for Use so that you inject TYMLOS pen the right way. Call
Package leaflet: Information for the user. Levonorgestrel 1.5 mg Tablet. levonorgestrel
 Package leaflet: Information for the user Levonorgestrel 1.5 mg Tablet levonorgestrel Read all of this leaflet carefully before you start taking this medicine because it contains important information
Package leaflet: Information for the user Levonorgestrel 1.5 mg Tablet levonorgestrel Read all of this leaflet carefully before you start taking this medicine because it contains important information
KINGSTON GENERAL HOSPITAL NURSING POLICY AND PROCEDURE
 KINGSTON GENERAL HOSPITAL NURSING POLICY AND PROCEDURE SUBJECT Sample (Adult): Advanced Competency (AC) for Nurses (Registered Nurses and Registered Practical Nurses) PAGE 1 of 5 ORIGINAL ISSUE 1985 January
KINGSTON GENERAL HOSPITAL NURSING POLICY AND PROCEDURE SUBJECT Sample (Adult): Advanced Competency (AC) for Nurses (Registered Nurses and Registered Practical Nurses) PAGE 1 of 5 ORIGINAL ISSUE 1985 January
Family Planning UNMET NEED. The Nurse Mildred Radio Talk Shows
 Family Planning UNMET NEED The Nurse Mildred Radio Talk Shows TOPIC 9: IUD/COIL Guests FP counsellor from MSU, RHU& UHMG Nurse Mildred Nurse Betty Objectives of the programme: To inform listeners about
Family Planning UNMET NEED The Nurse Mildred Radio Talk Shows TOPIC 9: IUD/COIL Guests FP counsellor from MSU, RHU& UHMG Nurse Mildred Nurse Betty Objectives of the programme: To inform listeners about
Trigger Finger and Trigger Thumb A Patient's Guide to Trigger Finger & Trigger Thumb
 Trigger Finger and Trigger Thumb A Patient's Guide to Trigger Finger & Trigger Thumb Introduction Trigger finger and trigger thumb are conditions affecting the movement of the tendons as they bend the
Trigger Finger and Trigger Thumb A Patient's Guide to Trigger Finger & Trigger Thumb Introduction Trigger finger and trigger thumb are conditions affecting the movement of the tendons as they bend the
FDA-Approved Patient Labeling Patient Information Mirena (mur-ā-nah) (levonorgestrel-releasing intrauterine system)
 FDA-Approved Patient Labeling Patient Information Mirena (mur-ā-nah) (levonorgestrel-releasing intrauterine system) Mirena does not protect against HIV infection (AIDS) and other sexually transmitted infections
FDA-Approved Patient Labeling Patient Information Mirena (mur-ā-nah) (levonorgestrel-releasing intrauterine system) Mirena does not protect against HIV infection (AIDS) and other sexually transmitted infections
Patient Information Publications Warren Grant Magnuson Clinical Center National Institutes of Health
 Warren Grant Magnuson Clinical Center National Institutes of Health What is a subcutaneous injection? A subcutaneous injection is given in the fatty layer of tissue just under the skin. A subcutaneous
Warren Grant Magnuson Clinical Center National Institutes of Health What is a subcutaneous injection? A subcutaneous injection is given in the fatty layer of tissue just under the skin. A subcutaneous
BACTISEAL Endoscopic Ventricular Catheter (REF & )
 BACTISEAL Endoscopic Ventricular Catheter (REF 82-3087 & 82-3088) LCN 206966-001/E 2011 2017 DePuy Synthes. All rights reserved. Revised 07/17 206966-001-E.indd 1 ENGLISH IMPORTANT INFORMATION Please Read
BACTISEAL Endoscopic Ventricular Catheter (REF 82-3087 & 82-3088) LCN 206966-001/E 2011 2017 DePuy Synthes. All rights reserved. Revised 07/17 206966-001-E.indd 1 ENGLISH IMPORTANT INFORMATION Please Read
BLOOD COLLECTION GUIDELINES
 I. Patient Identification Lee Memorial Health System Lee County, FL CLINICAL LABORATORY BLOOD COLLECTION GUIDELINES A. Inpatient / Outpatient with armband 1. When possible, ask patient to state their name
I. Patient Identification Lee Memorial Health System Lee County, FL CLINICAL LABORATORY BLOOD COLLECTION GUIDELINES A. Inpatient / Outpatient with armband 1. When possible, ask patient to state their name
Breast Cancer Screening
 Scan for mobile link. Breast Cancer Screening What is breast cancer screening? Screening examinations are tests performed to find disease before symptoms begin. The goal of screening is to detect disease
Scan for mobile link. Breast Cancer Screening What is breast cancer screening? Screening examinations are tests performed to find disease before symptoms begin. The goal of screening is to detect disease
Guidance for Pharmacists on the Safe Supply of Non-Prescription Levonorgestrel 1500mcg for Emergency Hormonal Contraception
 Guidance for Pharmacists on the Safe Supply of Non-Prescription Levonorgestrel 1500mcg for Emergency Hormonal Contraception Pharmaceutical Society of Ireland Version 4 December 2016 Updates made following
Guidance for Pharmacists on the Safe Supply of Non-Prescription Levonorgestrel 1500mcg for Emergency Hormonal Contraception Pharmaceutical Society of Ireland Version 4 December 2016 Updates made following
IMPORTANT: PLEASE READ
 PART III: CONSUMER INFORMATION ALPROLIX [pronounced all prō liks] Coagulation Factor IX (Recombinant), Fc Fusion Protein This leaflet is part III of a three-part "Product Monograph" published when ALPROLIX
PART III: CONSUMER INFORMATION ALPROLIX [pronounced all prō liks] Coagulation Factor IX (Recombinant), Fc Fusion Protein This leaflet is part III of a three-part "Product Monograph" published when ALPROLIX
INTRODUCTION Cubital Tunnel Syndrome
 INTRODUCTION Cubital Tunnel Syndrome Diagram of the ulnar nerve supplying the muscles of forearm and hand Cubital Tunnel is a condition that refers to the ulnar nerve being compressed around the elbow.
INTRODUCTION Cubital Tunnel Syndrome Diagram of the ulnar nerve supplying the muscles of forearm and hand Cubital Tunnel is a condition that refers to the ulnar nerve being compressed around the elbow.
Children's (Pediatric) PICC Line Placement
 Scan for mobile link. Children's (Pediatric) PICC Line Placement A peripherally inserted central catheter (PICC line) is most often used to deliver medication over a long period. The doctor or nurse inserts
Scan for mobile link. Children's (Pediatric) PICC Line Placement A peripherally inserted central catheter (PICC line) is most often used to deliver medication over a long period. The doctor or nurse inserts
IMPORTANT: PLEASE READ
 PART III: CONSUMER INFORMATION ALPROLIX [pronounced all prō liks] Coagulation Factor IX (Recombinant), Fc Fusion Protein This leaflet is part III of a three-part "Product Monograph" published when ALPROLIX
PART III: CONSUMER INFORMATION ALPROLIX [pronounced all prō liks] Coagulation Factor IX (Recombinant), Fc Fusion Protein This leaflet is part III of a three-part "Product Monograph" published when ALPROLIX
How to Use ENBREL : Vial Adapter Method
 How to Use ENBREL : Vial Adapter Method SETTING UP FOR AN INJECTION Select a clean, well-lit, flat working surface, such as a table. Take the ENBREL dose tray out of the refrigerator and place it on your
How to Use ENBREL : Vial Adapter Method SETTING UP FOR AN INJECTION Select a clean, well-lit, flat working surface, such as a table. Take the ENBREL dose tray out of the refrigerator and place it on your
The following lesson on contraception (birth control) is not intended to infer that you will be sexually active as a teen. This is information that
 The following lesson on contraception (birth control) is not intended to infer that you will be sexually active as a teen. This is information that may be used in the future Abstinence Choosing not to
The following lesson on contraception (birth control) is not intended to infer that you will be sexually active as a teen. This is information that may be used in the future Abstinence Choosing not to
READ THIS FOR SAFE AND EFFECTIVE USE OF YOUR MEDICINE PATIENT MEDICATION INFORMATION. AIM-oh-vig Pr AIMOVIG (erenumab injection)
 READ THIS FOR SAFE AND EFFECTIVE USE OF YOUR MEDICINE PATIENT MEDICATION INFORMATION AIM-oh-vig Pr AIMOVIG (erenumab injection) Single-Use Pre-filled SureClick Autoinjector Read this carefully before you
READ THIS FOR SAFE AND EFFECTIVE USE OF YOUR MEDICINE PATIENT MEDICATION INFORMATION AIM-oh-vig Pr AIMOVIG (erenumab injection) Single-Use Pre-filled SureClick Autoinjector Read this carefully before you
Inspection Window. Syringe Body
 Guide to Parts Before use Instructions for Use UDENYCA (yoo-den-i-kah) (pegfilgrastim-cbqv) Injection Single-Dose Prefilled Syringe Inspection Window Needle Cap Needle Label Plunger Plunger Head Needle
Guide to Parts Before use Instructions for Use UDENYCA (yoo-den-i-kah) (pegfilgrastim-cbqv) Injection Single-Dose Prefilled Syringe Inspection Window Needle Cap Needle Label Plunger Plunger Head Needle
The Balanced Counseling Strategy Plus: A Toolkit for Family Planning Service Providers Working in High STI/HIV Prevalence Settings.
 The Balanced Counseling Strategy Plus: A Toolkit for Family Planning Service Providers Working in High STI/HIV Prevalence Settings Counseling Cards Checklist to be reasonably sure a woman is not pregnant
The Balanced Counseling Strategy Plus: A Toolkit for Family Planning Service Providers Working in High STI/HIV Prevalence Settings Counseling Cards Checklist to be reasonably sure a woman is not pregnant
LONG-ACTING REVERSIBLE CONTRACEPTION. Summary Tables
 LONG-ACTING REVERSIBLE CONTRACEPTION Summary Tables Bridging the Divide: A Project of the Jacobs Institute of Women s Health June 2016 Table 1. Summary of LARC Methods Available Years Since Effective Copper
LONG-ACTING REVERSIBLE CONTRACEPTION Summary Tables Bridging the Divide: A Project of the Jacobs Institute of Women s Health June 2016 Table 1. Summary of LARC Methods Available Years Since Effective Copper
Postpartum LARC. (Long Acting Reversible Contraception) NURSING EDUCATION
 Postpartum LARC (Long Acting Reversible Contraception) NURSING EDUCATION What is LARC Long-acting reversible contraception (LARC) methods include the intrauterine device (IUD) and the birth control implant.
Postpartum LARC (Long Acting Reversible Contraception) NURSING EDUCATION What is LARC Long-acting reversible contraception (LARC) methods include the intrauterine device (IUD) and the birth control implant.
Instructions For Use PRALUENT (PRAHL-u-ent) (alirocumab) Injection, for Subcutaneous Injection Single-Dose Pre-Filled Syringe (75 mg/ml)
 Instructions For Use PRALUENT (PRAHL-u-ent) (alirocumab) Injection, for Subcutaneous Injection Single-Dose Pre-Filled Syringe (75 mg/ml) Important Information The device is a single-dose pre-filled syringe.
Instructions For Use PRALUENT (PRAHL-u-ent) (alirocumab) Injection, for Subcutaneous Injection Single-Dose Pre-Filled Syringe (75 mg/ml) Important Information The device is a single-dose pre-filled syringe.
CLARIVEIN INFUSION CATHETER
 CLARIVEIN INFUSION CATHETER General Product Description Overview The ClariVein Infusion Catheter (ClariVein -IC) is an infusion catheter system designed to introduce physician-specified medicaments into
CLARIVEIN INFUSION CATHETER General Product Description Overview The ClariVein Infusion Catheter (ClariVein -IC) is an infusion catheter system designed to introduce physician-specified medicaments into
Instructions for Use Neulasta (nu-las-tah) (pegfilgrastim) Injection, for subcutaneous use Single-Dose Prefilled Syringe. Plunger rod Used plunger rod
 Instructions for Use Neulasta (nu-las-tah) (pegfilgrastim) Injection, for subcutaneous use Single-Dose Prefilled Syringe Guide to parts Before use After use Plunger rod Used plunger rod Finger grip Label
Instructions for Use Neulasta (nu-las-tah) (pegfilgrastim) Injection, for subcutaneous use Single-Dose Prefilled Syringe Guide to parts Before use After use Plunger rod Used plunger rod Finger grip Label
THE WOMAN-FRIENDLY STERILIZATION METHOD
 THE WOMAN-FRIENDLY STERILIZATION METHOD Urogyn BV Transistorweg 5a 6534 AT Nijmegen The Netherlands t +31(0) 24 711 41 30 info@urogynbv.com www.urogynbv.com THE MOST WOMAN-FRIENDLY STERILIZATION METHOD
THE WOMAN-FRIENDLY STERILIZATION METHOD Urogyn BV Transistorweg 5a 6534 AT Nijmegen The Netherlands t +31(0) 24 711 41 30 info@urogynbv.com www.urogynbv.com THE MOST WOMAN-FRIENDLY STERILIZATION METHOD
Before you use the TALTZ autoinjector, read and carefully follow all the step-by-step instructions.
 1 INSTRUCTIONS FOR USE TALTZ (tȯl(t)s) (ixekizumab) injection, for subcutaneous use Autoinjector Before you use the TALTZ autoinjector, read and carefully follow all the step-by-step instructions. Important
1 INSTRUCTIONS FOR USE TALTZ (tȯl(t)s) (ixekizumab) injection, for subcutaneous use Autoinjector Before you use the TALTZ autoinjector, read and carefully follow all the step-by-step instructions. Important
ATI Skills Modules Checklist for Central Venous Access Devices
 For faculty use only Educator s name Score Date ATI Skills Modules Checklist for Central Venous Access Devices Student s name Date Verify order Patient record Assess for procedure need Identify, gather,
For faculty use only Educator s name Score Date ATI Skills Modules Checklist for Central Venous Access Devices Student s name Date Verify order Patient record Assess for procedure need Identify, gather,
Patient Information. EGRIFTA (eh-grif-tuh) (tesamorelin for injection) for subcutaneous use
 Patient Information EGRIFTA (eh-grif-tuh) (tesamorelin for injection) for subcutaneous use Read the Patient Information that comes with EGRIFTA before you start to take it and each time you get a refill.
Patient Information EGRIFTA (eh-grif-tuh) (tesamorelin for injection) for subcutaneous use Read the Patient Information that comes with EGRIFTA before you start to take it and each time you get a refill.
Unintended Pregnancy is Common LEARNING OBJECTIVES. Distribution Of Contraception Use By Women In The Us. Unintended Pregnancy And Contraceptive Use
 3:45 4:30 pm Beyond the Pill: Long Acting Contraceptives and IUDs Presenter Disclosure Information The following relationships exist related to this presentation: Christine L. Curry, MD, PhD: No financial
3:45 4:30 pm Beyond the Pill: Long Acting Contraceptives and IUDs Presenter Disclosure Information The following relationships exist related to this presentation: Christine L. Curry, MD, PhD: No financial
TREMFYA (guselkumab) Information for patients
 TREMFYA (guselkumab) Information for patients Pre-filled syringe Please read this booklet carefully as it contains important information about TREMFYA, the treatment that your doctor has prescribed to
TREMFYA (guselkumab) Information for patients Pre-filled syringe Please read this booklet carefully as it contains important information about TREMFYA, the treatment that your doctor has prescribed to
WHAT ARE CONTRACEPTIVES?
 CONTRACEPTION WHAT ARE CONTRACEPTIVES? Methods used to prevent fertilization *Also referred to as birth control methods With contraceptives, it is important to look at what works for you and your body.
CONTRACEPTION WHAT ARE CONTRACEPTIVES? Methods used to prevent fertilization *Also referred to as birth control methods With contraceptives, it is important to look at what works for you and your body.
Instructions for Use Enbrel (en-brel) (etanercept) injection, for subcutaneous use Single-dose Prefilled Syringe
 Instructions for Use Enbrel (en-brel) (etanercept) injection, for subcutaneous use Single-dose Prefilled Syringe How do I prepare and give an injection with Enbrel Single-dose Prefilled Syringe? There
Instructions for Use Enbrel (en-brel) (etanercept) injection, for subcutaneous use Single-dose Prefilled Syringe How do I prepare and give an injection with Enbrel Single-dose Prefilled Syringe? There
Important Safety Information for Adlyxin (lixisenatide) injection
 A GLP-1 receptor agonist (RA) Adlyxin (lixisenatide) injection 20 mcg Information To Help Answer Patients Questions This booklet contains information about Adlyxin to help you answer patients questions
A GLP-1 receptor agonist (RA) Adlyxin (lixisenatide) injection 20 mcg Information To Help Answer Patients Questions This booklet contains information about Adlyxin to help you answer patients questions
Subacromial Bursa Injection
 Subacromial Bursa Injection 5 cc syringe, 21 gauge 1.5 inch needle 1% lidocaine - 4cc 40mg triamcinolone - 1 cc of 40mg/ml identify site-seat the patient with weight of arm hanging down, palpate the lateral
Subacromial Bursa Injection 5 cc syringe, 21 gauge 1.5 inch needle 1% lidocaine - 4cc 40mg triamcinolone - 1 cc of 40mg/ml identify site-seat the patient with weight of arm hanging down, palpate the lateral
YASMIN 0.03 mg/3 mg BAYER MIDDLE EAST
 08-15 YASMIN 0.03 mg/3 mg BAYER MIDDLE EAST film-coated tablets Ethinylestradiol / Drospirenone 1. WHAT YASMIN IS AND WHAT IT IS USED FOR Yasmin is a contraceptive pill and is used to prevent pregnancy.
08-15 YASMIN 0.03 mg/3 mg BAYER MIDDLE EAST film-coated tablets Ethinylestradiol / Drospirenone 1. WHAT YASMIN IS AND WHAT IT IS USED FOR Yasmin is a contraceptive pill and is used to prevent pregnancy.
Peel-Apart Percutaneous Introducer Kits for
 Bard Access Systems Peel-Apart Percutaneous Introducer Kits for Table of Contents Contents Page Bard Implanted Ports Hickman*, Leonard*, Broviac*, Tenckhoff*, and Groshong* Catheters Introduction....................................
Bard Access Systems Peel-Apart Percutaneous Introducer Kits for Table of Contents Contents Page Bard Implanted Ports Hickman*, Leonard*, Broviac*, Tenckhoff*, and Groshong* Catheters Introduction....................................
LESSON 9. How to counsel clients on Sayana Press self-injection
 LESSON 9 How to counsel clients on Sayana Press self-injection 1 LESSON 9 Learning objectives In this session, you will learn how to advise clients on: Proper storage of Sayana Press. Proper disposal of
LESSON 9 How to counsel clients on Sayana Press self-injection 1 LESSON 9 Learning objectives In this session, you will learn how to advise clients on: Proper storage of Sayana Press. Proper disposal of
Gammanorm, 165 mg/ml, solution for injection Human normal immunoglobulin
 PACKAGE LEAFLET: INFORMATION FOR THE USER Gammanorm, 165 mg/ml, solution for injection Human normal immunoglobulin Read all of this leaflet carefully before you start using this medicine. Keep this leaflet.
PACKAGE LEAFLET: INFORMATION FOR THE USER Gammanorm, 165 mg/ml, solution for injection Human normal immunoglobulin Read all of this leaflet carefully before you start using this medicine. Keep this leaflet.
THE HUMERUS 20 THE HUMERUS* CROSS SECTION CROSS SECTION SUPERIOR VIEW
 20 THE HUMERUS* CROSS SECTION CROSS SECTION SUPERIOR VIEW The marrow canal of the humerus is funnel-shaped. Its successful pinning is influenced by many factors. With a few exceptions, the entire humerus
20 THE HUMERUS* CROSS SECTION CROSS SECTION SUPERIOR VIEW The marrow canal of the humerus is funnel-shaped. Its successful pinning is influenced by many factors. With a few exceptions, the entire humerus
ROTATOR CUFF INJURIES / IMPINGEMENT SYNDROME
 ROTATOR CUFF INJURIES / IMPINGEMENT SYNDROME Shoulder injuries are common in patients across all ages, from young, athletic people to the aging population. Two of the most common problems occur in the
ROTATOR CUFF INJURIES / IMPINGEMENT SYNDROME Shoulder injuries are common in patients across all ages, from young, athletic people to the aging population. Two of the most common problems occur in the
Instructions for Use Enbrel (en-brel) (etanercept) for injection, for subcutaneous use Multiple-dose Vial
 Instructions for Use Enbrel (en-brel) (etanercept) for injection, for subcutaneous use Multiple-dose Vial How do I prepare and give an injection with Enbrel multiple-dose vial? A multiple-dose vial contains
Instructions for Use Enbrel (en-brel) (etanercept) for injection, for subcutaneous use Multiple-dose Vial How do I prepare and give an injection with Enbrel multiple-dose vial? A multiple-dose vial contains
NHS Lothian Patient Group Direction Version: 007. Title: Levonorgestrel 1500mcg for Emergency Contraception
 PATIENT GROUP DIRECTION FOR THE ADMINISTRATION OF LEVORGESTREL 1500mcg FOR EMERGENCY CONTRACEPTION BY DESIGNATED HEALTHCARE PROFESSIONALS PROVIDING SEXUAL HEALTH SERVICES, UNSCHEDULED CARE SERVICES OR
PATIENT GROUP DIRECTION FOR THE ADMINISTRATION OF LEVORGESTREL 1500mcg FOR EMERGENCY CONTRACEPTION BY DESIGNATED HEALTHCARE PROFESSIONALS PROVIDING SEXUAL HEALTH SERVICES, UNSCHEDULED CARE SERVICES OR
Training slides for Tuberculin Skin Testing (TST)
 Disclaimer This presentation is produced by McGill International TB Centre solely for educational purposes. This presentation content is intended to train health care providers to perform Tuberculin skin
Disclaimer This presentation is produced by McGill International TB Centre solely for educational purposes. This presentation content is intended to train health care providers to perform Tuberculin skin
Package leaflet: Information for the patient. Provera 2.5mg, 5mg and 10mg Tablets Medroxyprogesterone acetate
 Package leaflet: Information for the patient Provera 2.5mg, 5mg and 10mg Tablets Medroxyprogesterone acetate Read all of this leaflet carefully before you start using this medicine because it contains
Package leaflet: Information for the patient Provera 2.5mg, 5mg and 10mg Tablets Medroxyprogesterone acetate Read all of this leaflet carefully before you start using this medicine because it contains
Integra. Endoscopic Gastrocnemius Release System SURGICAL TECHNIQUE
 Integra Endoscopic Gastrocnemius Release System SURGICAL TECHNIQUE Table of Contents Indications... 2 Contraindications... 2 System Description... 2 Features and Benefits... 2 Surgical Site Preparation...3
Integra Endoscopic Gastrocnemius Release System SURGICAL TECHNIQUE Table of Contents Indications... 2 Contraindications... 2 System Description... 2 Features and Benefits... 2 Surgical Site Preparation...3
SURGICAL TECHNIQUE GUIDE
 DANGER indicates an imminently hazardous situation which, if not avoided, will result in death or serious injury. WARNING indicates a potentially hazardous situation which, if not avoided, could result
DANGER indicates an imminently hazardous situation which, if not avoided, will result in death or serious injury. WARNING indicates a potentially hazardous situation which, if not avoided, could result
CATHETER ACCESS KIT. For use with Prometra Programmable Infusion Systems
 CATHETER ACCESS KIT Caution: Federal (USA) Law restricts this device to sale by or on the order of a physician. Table of Contents Contents... 3 Description... 3 Indications... 3 Contraindications... 3
CATHETER ACCESS KIT Caution: Federal (USA) Law restricts this device to sale by or on the order of a physician. Table of Contents Contents... 3 Description... 3 Indications... 3 Contraindications... 3
Parkland Health & Hospital System Women & Infant Specialty Health
 Parkland Health & Hospital System Women & Infant Specialty Health NS 1700.04 Nursery Services Procedure Manual Arterial Puncture Practice Statement Upon the written order of the provider, the credentialled
Parkland Health & Hospital System Women & Infant Specialty Health NS 1700.04 Nursery Services Procedure Manual Arterial Puncture Practice Statement Upon the written order of the provider, the credentialled
Package leaflet: Information for the user
 Package leaflet: Information for the user Aimovig 70 mg solution for injection in pre-filled pen erenumab This medicine is subject to additional monitoring. This will allow quick identification of new
Package leaflet: Information for the user Aimovig 70 mg solution for injection in pre-filled pen erenumab This medicine is subject to additional monitoring. This will allow quick identification of new
Instructions for Use. Welcome!
 Instructions for Use Welcome! The AMJEVITA SureClick autoinjector is a single-use prefilled autoinjector. Consult your doctor if you have any questions about your dose. Your doctor has prescribed AMJEVITA
Instructions for Use Welcome! The AMJEVITA SureClick autoinjector is a single-use prefilled autoinjector. Consult your doctor if you have any questions about your dose. Your doctor has prescribed AMJEVITA
LEARNING OBJECTIVES. Beyond the Pill: Long Acting Contraception. Distribution Of Contraception Use By Women In The Us. Unintended Pregnancy is Common
 4:15 5 pm Beyond the Pill: Long Acting Contraceptives and IUDs Presenter Disclosure Information The following relationships exist related to this presentation: Christine L. Curry, MD, PhD: No financial
4:15 5 pm Beyond the Pill: Long Acting Contraceptives and IUDs Presenter Disclosure Information The following relationships exist related to this presentation: Christine L. Curry, MD, PhD: No financial
In Activity 12, What s Happening Inside? you learned about the functions
 16 Support System: Bones, Joints and Muscles l a b o r at o ry In Activity 12, What s Happening Inside? you learned about the functions of the skeletal and muscular systems in supporting and moving your
16 Support System: Bones, Joints and Muscles l a b o r at o ry In Activity 12, What s Happening Inside? you learned about the functions of the skeletal and muscular systems in supporting and moving your
Instructions for Use HEMLIBRA (hem-lee-bruh) (emicizumab-kxwh) injection, for subcutaneous use
 Instructions for Use HEMLIBRA (hem-lee-bruh) (emicizumab-kxwh) injection, for subcutaneous use Be sure that you read, understand, and follow the Instructions for Use before injecting HEMLIBRA. Your healthcare
Instructions for Use HEMLIBRA (hem-lee-bruh) (emicizumab-kxwh) injection, for subcutaneous use Be sure that you read, understand, and follow the Instructions for Use before injecting HEMLIBRA. Your healthcare
Venous sampling. What is venous sampling? What are some common uses of the procedure?
 Scan for mobile link. Venous sampling Venous sampling is a diagnostic procedure that uses imaging guidance to insert a catheter into a specific vein and remove blood samples for laboratory analysis. Abnormal
Scan for mobile link. Venous sampling Venous sampling is a diagnostic procedure that uses imaging guidance to insert a catheter into a specific vein and remove blood samples for laboratory analysis. Abnormal
Patient Group Direction for LEVONELLE (Version 02) Valid From 1 October September 2019
 Version Control This PGD has been agreed by the following organisations FCMS PDS Medical Doncaster CCG Lancashire CCGs including East Lancashire, Fylde and Wyre and North Lancashire CCGs Change history
Version Control This PGD has been agreed by the following organisations FCMS PDS Medical Doncaster CCG Lancashire CCGs including East Lancashire, Fylde and Wyre and North Lancashire CCGs Change history
UW MEDICINE PATIENT EDUCATION. Cushing s Syndrome DRAFT. What is Cushing s syndrome? What is cortisol? What are the symptoms of Cushing s syndrome?
 UW MEDICINE PATIENT EDUCATION Cushing s Syndrome Causes, symptoms, diagnosis, and treatments This handout explains Cushing s syndrome, its causes, symptoms, and how it is diagnosed. It also includes a
UW MEDICINE PATIENT EDUCATION Cushing s Syndrome Causes, symptoms, diagnosis, and treatments This handout explains Cushing s syndrome, its causes, symptoms, and how it is diagnosed. It also includes a
Specimen Collection Policies
 Specimen Collection Policies Purpose Great River Medical Center Laboratory is a hospital-based and outreach laboratory with specific standards of excellence. To best serve our patients, all specimens will
Specimen Collection Policies Purpose Great River Medical Center Laboratory is a hospital-based and outreach laboratory with specific standards of excellence. To best serve our patients, all specimens will
Linear Array Transducer
 User Guide Type 8809 Linear Array Transducer English BB 0911-H August 2012 For Professional Users Only WORLD HEADQUARTERS Mileparken 34 DK-2730 Herlev Denmark Tel.:+45 44528100 / Fax:+45 44528199 www.bkmed.com
User Guide Type 8809 Linear Array Transducer English BB 0911-H August 2012 For Professional Users Only WORLD HEADQUARTERS Mileparken 34 DK-2730 Herlev Denmark Tel.:+45 44528100 / Fax:+45 44528199 www.bkmed.com
Package leaflet: Information for the patient. Orgalutran 0.25 mg/0.5 ml solution for injection Ganirelix
 Package leaflet: Information for the patient Orgalutran 0.25 mg/0.5 ml solution for injection Ganirelix Read all of this leaflet carefully before you start using this medicine because it contains important
Package leaflet: Information for the patient Orgalutran 0.25 mg/0.5 ml solution for injection Ganirelix Read all of this leaflet carefully before you start using this medicine because it contains important
SELF-INJECTION GUIDE
 SELF-INJECTION GUIDE (asfotase alfa) 40 mg/ml solution for injection 18 mg/0.45 ml 28 mg/0.7 ml 40 mg/1 ml 100 mg/ml solution for injection 80 mg/0.8 ml asfotase alfa Introduction Contents This Self-Injection
SELF-INJECTION GUIDE (asfotase alfa) 40 mg/ml solution for injection 18 mg/0.45 ml 28 mg/0.7 ml 40 mg/1 ml 100 mg/ml solution for injection 80 mg/0.8 ml asfotase alfa Introduction Contents This Self-Injection
Procedures/Risks:central venous catheter
 Procedures/Risks:central venous catheter Central Venous Catheter Placement Procedure: Placement of the central venous catheter will take place in the Interventional Radiology Department (IRD) at The Ohio
Procedures/Risks:central venous catheter Central Venous Catheter Placement Procedure: Placement of the central venous catheter will take place in the Interventional Radiology Department (IRD) at The Ohio
Notes to Teacher continued Contraceptive Considerations
 Abstinence a conscious decision to refrain from sexual intercourse 100% pregnancy will not occur if close contact between the penis and vagina does not take place. The risk of a number of STDs, including
Abstinence a conscious decision to refrain from sexual intercourse 100% pregnancy will not occur if close contact between the penis and vagina does not take place. The risk of a number of STDs, including
Tips for Successful Venipuncture
 Page 1 of 5 Patient Positioning Have patient lie or sit down. Never draw blood on a patient who is standing. Make the patient as comfortable as you possibly can. Always watch and ask patient if he/she
Page 1 of 5 Patient Positioning Have patient lie or sit down. Never draw blood on a patient who is standing. Make the patient as comfortable as you possibly can. Always watch and ask patient if he/she
Intravascular Ultrasound
 Scan for mobile link. Intravascular Ultrasound Intravascular ultrasound (IVUS) uses a transducer or probe to generate sound waves and produce pictures of the coronary arteries. IVUS can show the entire
Scan for mobile link. Intravascular Ultrasound Intravascular ultrasound (IVUS) uses a transducer or probe to generate sound waves and produce pictures of the coronary arteries. IVUS can show the entire
PRODUCTS FOR THE DIFFICULT AIRWAY. Courtesy of Cook Critical Care
 PRODUCTS FOR THE DIFFICULT AIRWAY Courtesy of Cook Critical Care EMERGENCY CRICOTHYROTOMY Thyroid Cartilage Access Site Cricoid Cartilage Identify the cricothyroid membrane between the cricoid and thyroid
PRODUCTS FOR THE DIFFICULT AIRWAY Courtesy of Cook Critical Care EMERGENCY CRICOTHYROTOMY Thyroid Cartilage Access Site Cricoid Cartilage Identify the cricothyroid membrane between the cricoid and thyroid
The use of long-acting reversible contraceptive
 Overcoming LARC complications: 7 case challenges The strings to your patient s intrauterine device (IUD) are missing. Clinical experience and ACOG direction guide the management plans for this and more
Overcoming LARC complications: 7 case challenges The strings to your patient s intrauterine device (IUD) are missing. Clinical experience and ACOG direction guide the management plans for this and more
Directions For Use. All directions should be read before use
 Directions For Use All directions should be read before use DEVICE DESCRIPTION: The CLEANER.XT Rotational Thrombectomy System is a percutaneous, 6Fr catheter based system (single piece construction) that
Directions For Use All directions should be read before use DEVICE DESCRIPTION: The CLEANER.XT Rotational Thrombectomy System is a percutaneous, 6Fr catheter based system (single piece construction) that
Women and epilepsy. Introduction. Puberty and young women
 Women and epilepsy Introduction Epilepsy can affect women at any age in their lives, from childhood to puberty, during the reproductive years and pregnancy, through to menopause and later life. This guide
Women and epilepsy Introduction Epilepsy can affect women at any age in their lives, from childhood to puberty, during the reproductive years and pregnancy, through to menopause and later life. This guide
PACKAGE LEAFLET: INFORMATION FOR THE USER Pulmicort Turbohaler budesonide. 1. What Pulmicort Turbohaler is and what it is used for
 PACKAGE LEAFLET: INFORMATION FOR THE USER Pulmicort Turbohaler budesonide Read all of this leaflet carefully before you start taking this medicine. Keep this leaflet. You may need to read it again. If
PACKAGE LEAFLET: INFORMATION FOR THE USER Pulmicort Turbohaler budesonide Read all of this leaflet carefully before you start taking this medicine. Keep this leaflet. You may need to read it again. If
SUMMARY OF PRODUCT CHARACTERISTICS
 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT ellaone 30 mg tablet 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each tablet contains 30 mg ulipristal acetate. Excipients with known
SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT ellaone 30 mg tablet 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each tablet contains 30 mg ulipristal acetate. Excipients with known
INSTRUCTIONS FOR USE. EMGALITY TM (em-gal-it-ē) (galcanezumab-gnlm)
 1 INSTRUCTIONS FOR USE EMGALITY TM (em-gal-it-ē) (galcanezumab-gnlm) injection, for subcutaneous use Prefilled Pen For subcutaneous injection only. Before you use the EMGALITY prefilled pen (Pen), read
1 INSTRUCTIONS FOR USE EMGALITY TM (em-gal-it-ē) (galcanezumab-gnlm) injection, for subcutaneous use Prefilled Pen For subcutaneous injection only. Before you use the EMGALITY prefilled pen (Pen), read
This information is intended as an overview only
 This information is intended as an overview only Please refer to the INSTRUCTIONS FOR USE included with this device for indications, contraindications, warnings, precautions and other important information
This information is intended as an overview only Please refer to the INSTRUCTIONS FOR USE included with this device for indications, contraindications, warnings, precautions and other important information
ALPROLIX Coagulation Factor IX (Recombinant), Fc Fusion Protein INSTRUCTIONS FOR USE Do not Do not YOUR KIT CONTAINS:
 ALPROLIX Coagulation Factor IX (Recombinant), Fc Fusion Protein INSTRUCTIONS FOR USE Read the Instructions for Use before you start using ALPROLIX and each time you get a refill. There may be new information.
ALPROLIX Coagulation Factor IX (Recombinant), Fc Fusion Protein INSTRUCTIONS FOR USE Read the Instructions for Use before you start using ALPROLIX and each time you get a refill. There may be new information.
Stereotactic Breast Biopsy
 Scan for mobile link. Stereotactic Breast Biopsy Stereotactic breast biopsy uses mammography a specific type of breast imaging that uses low-dose x-rays to help locate a breast abnormality and remove a
Scan for mobile link. Stereotactic Breast Biopsy Stereotactic breast biopsy uses mammography a specific type of breast imaging that uses low-dose x-rays to help locate a breast abnormality and remove a
BUTTONHOLE CANNULATION
 BUTTONHOLE CANNULATION What is a Buttonhole? Technique in which an AV fistula is cannulated in the exact same spot, at the same angle and at the same depth of penetration every time. Benefits Reduction
BUTTONHOLE CANNULATION What is a Buttonhole? Technique in which an AV fistula is cannulated in the exact same spot, at the same angle and at the same depth of penetration every time. Benefits Reduction
Cardiva Catalyst III INSTRUCTIONS FOR USE
 Cardiva Catalyst III INSTRUCTIONS FOR USE IFU 2469 Rev. G CAUTION Federal (USA) law restricts this device to sale by or on the order of a physician. For single use only GRAPHICAL SYMBOLS ON THE CARDIVA
Cardiva Catalyst III INSTRUCTIONS FOR USE IFU 2469 Rev. G CAUTION Federal (USA) law restricts this device to sale by or on the order of a physician. For single use only GRAPHICAL SYMBOLS ON THE CARDIVA
17. Preventing pregnancy
 17. Preventing pregnancy Objectives By the end of this session, group members will be able to: Define contraception. List ways young people can prevent pregnancy. Background notes What is contraception?
17. Preventing pregnancy Objectives By the end of this session, group members will be able to: Define contraception. List ways young people can prevent pregnancy. Background notes What is contraception?
