Commissioning Statement. Flash Glucose Monitoring system (FreeStyle Libre ) March 2018
|
|
|
- Horace Nelson
- 6 years ago
- Views:
Transcription
1 Technology Commissioning Statement Flash Glucose Monitoring system (FreeStyle Libre ) March 2018 FreeStyle Libre (Abbott) Flash Glucose Monitoring System for use in adults, young people and children. Recommendation The Medicines Commissioning Committee has considered the recommendation issued by the Regional Medicines Optimisation Committee on the 25 th October 2017 along with additional committee discussions. NHS Vale of York and NHS Scarborough and Ryedale CCGs do not routinely commission the use of Freestyle Libre. FreeStyle Libre is only commissioned for: patients with Type 1 diabetes mellitus (T1DM) and; aged four and above and; under specialist care and; using multiple daily injections of insulin, or insulin pump therapy, and; whom the specialist considers the use of the device will be costeffective. The use of FreeStyle Libre is expected to be cost neutral if a patient is currently finger prick testing 8 or more times daily. It will be regarded as cost effective when the introduction of FreeStyle Libre reduces the testing frequency by at least seven times daily. In addition to all of the above the patient must meet one or more of the following criteria: Meet current NICE criteria for insulin pump therapy (HbA1c 69.4 mmol/mol ( 8.5%) or disabling hypoglycaemia as described in NICE TA151) where a successful trial of FreeStyle Libre may avoid the need for pump therapy Recently developed impaired awareness of hypoglycaemia such that there is an inability to detect the onset of hypoglycaemia because of absence of warning symptoms. The individual must also demonstrate a willingness to engage with further education where applicable and a high level of engagement with glucose testing and management. (Note that for persistent hypoglycaemia unawareness, NICE recommend continuous glucose monitoring with alarms and FreeStyle Libre does not currently have this function). 1
2 One severe hypoglycaemic episode, defined as an episode of hypoglycaemia requiring the assistance of another person, with no obvious precipitating cause and with clear evidence of impairment of hypoglycaemia unawareness on subsequent assessment by specialist diabetes team. The individual must also demonstrate a willingness to engage with further education where applicable and a high level of engagement with glucose testing and management. Two or more admissions to hospital per year with diabetic ketoacidosis or hypoglycaemia Patients who have considerable difficulties in finger prick testing due to a physical limitation Patients with functional impairment that impacts on their ability to interpret standard finger prick testing results Pregnant women with T1DM or T2DM on a basal bolus insulin regime and in women with T1DM who are trying to conceive. Use of FreeStyle Libre in women with gestational diabetes is excluded unless the above criteria are met. After giving birth, women will be expected to return to their previous method of blood glucose monitoring Reluctance to carry out finger prick testing (e.g. due to distress or inconvenience) alone is not considered to be criteria qualifying the use of FreeStyle Libre. FreeStyle Libre will be provided initially on a 6 month trial basis. The decision to continue will be made by the diabetes specialist team only if one or more of the following are demonstrated: Reduction in usage of blood glucose test strips [approximate target to be agreed] (Trial data showed a reduction in blood glucose testing to an average of 0.5 times per day in patients using FreeStyle Libre ; however it is acknowledged that more frequent testing may be required in certain circumstances e.g. during periods of illness or to fulfil DVLA requirements). Reduction in severe/non- severe hypoglycaemia frequency Reversal of impaired awareness of hypoglycaemia Reduction in episodes of diabetic ketoacidosis HbA1c reduction of 0.5% (5 mmol/mol) within 6 months Reduction in hospital admissions Improved quality of life using validated rating scales The provision of FreeStyle Libre sensors will remain the responsibility of the diabetes specialist team in secondary care. All patients (or carers) must be willing to undertake training in the use of FreeStyle Libre. They must commit to regular scans of the device demonstrating evidence of FreeStyle Libre use in self-management, 2
3 and commit to ongoing regular follow-up and monitoring. They must also agree the expected outcomes with usage and that NHS provision of FreeStyle Libre will be withdrawn if one or more of the above criteria are not met. The diabetes specialist team in secondary care must gain consent from the patient (or patient representative) at the point of initiation of FreeStyle Libre, commissioned by the NHS, to permit NHS staff to undertake an audit of data from both the specialist setting and the patient s GP Practice to enable the benefit of initiation and continuation of FreeStyle Libre to be assessed. Secondary care specialists teams are responsible for collecting the audit data at the end of a six month trial from primary and secondary care in order for the commissioning of FreeStyle Libre to continue. If data is not presented to the CCG within four weeks of the six month trial commissioning of FreeStyle Libre will not continue for that individual patient. The diabetes specialist team in secondary care must complete a proforma for each patient demonstrating that the patient meets the above criteria and submit this pro-forma to the CCG. The CCG will audit the uptake on FreeStyle Libre on a monthly basis. The commissioning statement will be formally reviewed at either 12 months or when the number of patients exceeds the anticipated uptake (in January 2018), whichever is soonest. Summary of Clinical Evidence Treatment outcomes must be audited in all patients started on FreeStyle Libre by specialist teams. This audit data will be reviewed regularly by the CCGs and will inform the review of this recommendation twelve months following publication. Adjunct blood testing strips should be prescribed according to locally agreed best value guidelines with an expectation that demand/frequency of supply will be reduced. Use of Freestyle Libre in patients with T2DM (other than in pregnancy) is not recommended. Patients already purchasing FreeStyle Libre who do not meet the above criteria for initiation OR continuation will not be entitled to NHS prescriptions. A clinician can make an Individual Funding Request (IFR) for treatment when a patient does not meet the stated criteria for funding. Funding can only be approved if a case of exceptional clinical need has been demonstrated. FreeStyle Libre measures interstitial glucose levels from a sensor applied to the skin as an alternative to routine finger-prick blood glucose testing, and can produce a near-continuous record of measurements which can be accessed on demand. It can also indicate glucose level trends over time. Glucose readings can be seen anytime by scanning the sensor with a FreeStyle Libre reader or an android mobile device with Near-field Communication (NFC) capabilities via the LibreLink companion app. 3
4 FreeStyle Libre is indicated in people aged 4 or over with diabetes mellitus, who have multiple daily injections of insulin or who use insulin pumps and are self-managing their diabetes. FreeStyle Libre received European CE mark certification in August For more details on the device, please refer to NICE Medtech innovation briefing 110. The main points from the evidence are from 5 studies involving 700 people. These include 2 randomised controlled trials, 1 including people with type 1 diabetes (n=241; the IMPACT study) and the other including people with type 2 diabetes (n=224; the REPLACE study). Three of the studies reported device accuracy compared with selfmonitored blood glucose, with results ranging from 84% to 88% accuracy and from 99% to 100% clinical acceptability, using an error grid. One study reported device accuracy and acceptability of 97% to 99% compared with venous blood sampling. The evidence suggests that using FreeStyle Libre for up to 12 months reduces time spent in hypoglycaemia compared with self-monitoring of blood glucose using finger-prick tests, and reduces the average number of finger-prick blood glucose tests needed. In the IMPACT study, patients using FreeStyle Libre experienced less time in hypoglycaemia than patients using self-monitored blood glucose (SMBG), averaging 1.24 hours per day (SE 0.24) or 38% less time (p<0.0001) in hypoglycaemia and 1 hour more per day in euglycaemia (p=0.0006). The number of hypoglycaemic events per day reduced by mean of 0.45 (by over 25%; p<0.0001). The mean number of SMBG tests per day reduced from 5.5 (SD 2.0) to 0.5 (SD 0.7) in the FreeStyle Libre group. FreeStyle Libre does not include an alarm that alerts users when glucose levels are too high or too low. The device measures interstitial glucose levels and finger-prick blood glucose testing would still be needed: During times of rapidly changing glucose levels when interstitial fluid glucose levels may not accurately reflect blood glucose levels If FreeStyle Libre shows hypoglycaemia or impending hypoglycaemia When symptoms do not match the system readings To fulfil Driving and Vehicle Licensing Authority requirements to assess fitness to drive. Safety There are currently limited safety data on the use of FreeStyle Libre. The most commonly reported adverse effect related to sensor use in trials was skin reactions e.g. itching, rash, erythema, allergy, oedema and blisters. Some users may need to use a skin covering in order to be able to use the sensor. Accuracy of FreeStyle Libre readings compared to capillary blood glucose testing has been found to be broadly comparable. However capillary blood glucose testing is still recommended during times of rapidly changing glucose levels when interstitial fluid glucose levels 4
5 Cost Date effective from may not accurately reflect blood glucose levels (e.g. acute illness such as Influenza, diarrhoea and vomiting), or if hypoglycaemia or impending hypoglycaemia is reported, or the symptoms do not match the system readings. The annual cost of sensors is 910 per patient. The reader is not prescribable on the NHS but provided free of charge by the manufacturer. The use of FreeStyle Libre is expected to be cost neutral if a patient is currently finger prick testing 8 or more times daily, and the introduction of FreeStyle Libre reduces the testing frequency to an average of 0.5 times daily. The resource impact depends upon the extent to which improved glucose control through the adoption of FreeStyle Libre translates into fewer complications (hypoglycaemia and the longer term microvascular and macrovascular complications of hyperglycaemia), reduced admissions and reduced use of glucose test strips. 01 March 2018 Date published 01 March 2018 Review date March 2019 Author Responsible officer York and Scarborough Medicines Commissioning Committee Dr Shaun O Connell 5
Commissioning Statement
 Commissioning Statement Treatment/ device For the treatment of Commissioning position Flash Glucose Monitoring Systems (including Freestyle Libre ) Monitoring glucose levels in adults and children over
Commissioning Statement Treatment/ device For the treatment of Commissioning position Flash Glucose Monitoring Systems (including Freestyle Libre ) Monitoring glucose levels in adults and children over
THE SHEFFIELD AREA PRESCRIBING GROUP. Position Statement for Prescribing of Freestyle Libre In Type 1 Diabetes. Date: March 2018.
 THE SHEFFIELD AREA PRESCRIBING GROUP Position Statement for Prescribing of Freestyle Libre In Type 1 Diabetes Date: March 2018 Overview Freestyle Libre is a flash glucose sensor device that measures *interstitial
THE SHEFFIELD AREA PRESCRIBING GROUP Position Statement for Prescribing of Freestyle Libre In Type 1 Diabetes Date: March 2018 Overview Freestyle Libre is a flash glucose sensor device that measures *interstitial
FreeStyle Libre for glucose monitoring: Interim Position Statement for GPs & Patient FAQ
 North Central London Joint Formulary Committee FreeStyle Libre for glucose monitoring: Interim Position Statement for GPs & Patient FAQ GPs should not prescribe FreeStyle Libre sensors on the NHS until
North Central London Joint Formulary Committee FreeStyle Libre for glucose monitoring: Interim Position Statement for GPs & Patient FAQ GPs should not prescribe FreeStyle Libre sensors on the NHS until
NHS Leeds CCG. Policy for the Funding of Flash Glucose Monitoring (FlashGM) in Paediatrics and Adults
 NHS Leeds CCG Policy for the Funding of Flash Glucose Monitoring (FlashGM) in Paediatrics and Adults Produced by: Jo Alldred, Medicines Effectiveness Lead, NHS Leeds CCG Dr Bryan Power, Long Term Conditions
NHS Leeds CCG Policy for the Funding of Flash Glucose Monitoring (FlashGM) in Paediatrics and Adults Produced by: Jo Alldred, Medicines Effectiveness Lead, NHS Leeds CCG Dr Bryan Power, Long Term Conditions
NHS GG&C Introduction of Freestyle Libre flash glucose monitoring system
 NHS GG&C Introduction of Freestyle Libre flash glucose monitoring system The Freestyle Libre flash glucose monitoring system is a sensor based, factory-calibrated system that measures interstitial fluid
NHS GG&C Introduction of Freestyle Libre flash glucose monitoring system The Freestyle Libre flash glucose monitoring system is a sensor based, factory-calibrated system that measures interstitial fluid
Placename CCG. Policies for the Commissioning of Healthcare
 Placename CCG Policies for the Commissioning of Healthcare Policy for the Provision of Continuous Glucose Monitoring and Flash Glucose Monitoring to patients with Diabetes Mellitus. This document is part
Placename CCG Policies for the Commissioning of Healthcare Policy for the Provision of Continuous Glucose Monitoring and Flash Glucose Monitoring to patients with Diabetes Mellitus. This document is part
In keeping with the Scottish Diabetes Group criteria, use should be restricted to those who:
 Advice Statement 009-18 July 2018 Advice Statement What is the clinical and cost effectiveness of Freestyle Libre flash glucose monitoring for patients with diabetes mellitus treated with intensive insulin
Advice Statement 009-18 July 2018 Advice Statement What is the clinical and cost effectiveness of Freestyle Libre flash glucose monitoring for patients with diabetes mellitus treated with intensive insulin
Commissioning Policy
 Commissioning Policy Abbott FreeStyle Libre Flash Glucose Monitoring System Individual Funding Request Date Adopted: 09 September 2018 Version: 1819.1.02 Title of document: Authors job title(s): Document
Commissioning Policy Abbott FreeStyle Libre Flash Glucose Monitoring System Individual Funding Request Date Adopted: 09 September 2018 Version: 1819.1.02 Title of document: Authors job title(s): Document
Policy for the Provision of Continuous Glucose Monitoring and Flash Glucose Monitoring to patients with Diabetes Mellitus
 Policy for the Provision of Continuous Glucose Monitoring and Flash Glucose Monitoring to patients with Diabetes Mellitus Version No. Changes Made Version of 05.10.2018 V1 Policy ratified by Healthier
Policy for the Provision of Continuous Glucose Monitoring and Flash Glucose Monitoring to patients with Diabetes Mellitus Version No. Changes Made Version of 05.10.2018 V1 Policy ratified by Healthier
DERBYSHIRE JOINT AREA PRESCRIBING COMMITTEE (JAPC) JAPC Briefing FreeStyle Libre
 DERBYSHIRE JOINT AREA PRESCRIBING COMMITTEE (JAPC) JAPC Briefing FreeStyle Libre JAPC has classified Freestyle Libre as BROWN after diabetes consultant/specialist initiation within a Derbyshire diabetes
DERBYSHIRE JOINT AREA PRESCRIBING COMMITTEE (JAPC) JAPC Briefing FreeStyle Libre JAPC has classified Freestyle Libre as BROWN after diabetes consultant/specialist initiation within a Derbyshire diabetes
Policy for Continuous Glucose Monitoring for Type 1 Diabetic Paediatric Patients (<18 years of age)
 Policy for Continuous Glucose Monitoring for Type 1 Diabetic Paediatric Patients (
Policy for Continuous Glucose Monitoring for Type 1 Diabetic Paediatric Patients (
medicines_management/correspondence/pathway-for-the-managed- Access-of-FreeStyle-Libre.
 Pathway for the Managed Access of FreeStyle Libre (Flash Glucose monitoring) for Adults and Children in the care of Trust Specialist Diabetes Clinics in Northern Ireland www.hscboard.hscni.net/download/publications/pharmacy_and_
Pathway for the Managed Access of FreeStyle Libre (Flash Glucose monitoring) for Adults and Children in the care of Trust Specialist Diabetes Clinics in Northern Ireland www.hscboard.hscni.net/download/publications/pharmacy_and_
Implementation of Freestyle Libre prescribing guidance across the NHS in London
 Implementation of FreeStyle Libre prescribing guidance across the NHS in London Contents Section 1 Background to the document (pages 2-5) Section 2 Implementation guidance pathways 1. Recommended implementation
Implementation of FreeStyle Libre prescribing guidance across the NHS in London Contents Section 1 Background to the document (pages 2-5) Section 2 Implementation guidance pathways 1. Recommended implementation
Flash Glucose Monitoring (Flash GM) Frequently asked questions (November 2018)
 Flash Glucose Monitoring (Flash GM) Frequently asked questions (November 2018) What is Flash Glucose Monitoring (Flash GM)? Flash glucose monitoring is a small sensor that you wear on your skin (usually
Flash Glucose Monitoring (Flash GM) Frequently asked questions (November 2018) What is Flash Glucose Monitoring (Flash GM)? Flash glucose monitoring is a small sensor that you wear on your skin (usually
Description of the technology
 Advice Note 2017/ 001 What is the clinical effectiveness, safety and budget impact of the Freestyle Libre System compared with current glucose monitoring methods for people aged 4 years and over with diabetes
Advice Note 2017/ 001 What is the clinical effectiveness, safety and budget impact of the Freestyle Libre System compared with current glucose monitoring methods for people aged 4 years and over with diabetes
Placename CCG. Policies for the Commissioning of Healthcare
 Placename CCG Policies for the Commissioning of Healthcare Policy for the funding of insulin pumps and continuous glucose monitoring devices for patients with diabetes 1 Introduction 1.1 This document
Placename CCG Policies for the Commissioning of Healthcare Policy for the funding of insulin pumps and continuous glucose monitoring devices for patients with diabetes 1 Introduction 1.1 This document
DIABETES BLOOD GLUCOSE MONITORING AT HOME (ADULTS)
 Type 2 Diabetes DIABETES BLOOD GLUCOSE MONITORING AT HOME (ADULTS) Clinical Guideline Self-monitoring of blood glucose (SMBG) should be considered but not be routinely offered for adults with type 2 diabetes
Type 2 Diabetes DIABETES BLOOD GLUCOSE MONITORING AT HOME (ADULTS) Clinical Guideline Self-monitoring of blood glucose (SMBG) should be considered but not be routinely offered for adults with type 2 diabetes
DIABETES BLOOD GLUCOSE MONITORING AT HOME (ADULTS)
 DIABETES BLOOD GLUCOSE MONITORING AT HOME (ADULTS) Type 2 Diabetes Clinical Guideline Self-monitoring of blood glucose (SMBG) should be considered but not routinely offered for adults with type 2 diabetes
DIABETES BLOOD GLUCOSE MONITORING AT HOME (ADULTS) Type 2 Diabetes Clinical Guideline Self-monitoring of blood glucose (SMBG) should be considered but not routinely offered for adults with type 2 diabetes
Policy for the Provision of Insulin Pumps for Patients with Diabetes Mellitus
 Policy for the Provision of Insulin Pumps for Patients with Diabetes Mellitus Version No. Changes Made Version of July 2018 V0.5 Changes made to the policy following patient engagement including: - the
Policy for the Provision of Insulin Pumps for Patients with Diabetes Mellitus Version No. Changes Made Version of July 2018 V0.5 Changes made to the policy following patient engagement including: - the
Commissioning Policy Individual Funding Request
 Commissioning Policy Individual Funding Request Continuous Glucose Monitors Prior Approval Policy Date Adopted: 13 October 2017 Version: 1718.2 Document Control Title of document Continuous Glucose Monitors
Commissioning Policy Individual Funding Request Continuous Glucose Monitors Prior Approval Policy Date Adopted: 13 October 2017 Version: 1718.2 Document Control Title of document Continuous Glucose Monitors
Agenda Item 10 - Glucose Monitoring Device for Individuals with Diabetes
 Dorset Health Scrutiny Committee 17 October 2018 Public Participation Agenda Item 10 - Glucose Monitoring Device for Individuals with Diabetes Questions 1. Rosie and Kirsty Edwardes, Bridport Residents
Dorset Health Scrutiny Committee 17 October 2018 Public Participation Agenda Item 10 - Glucose Monitoring Device for Individuals with Diabetes Questions 1. Rosie and Kirsty Edwardes, Bridport Residents
Healthcare professionals involved in care of children and young people with Type 1 Diabetes Mellitus
 Clinical Guideline A Practical Approach to the Management of Continuous Glucose Monitoring (CGM) / Real-Time Flash Glucose Scanning (FGS) in Type 1 Diabetes Mellitus in Children and Young People Under
Clinical Guideline A Practical Approach to the Management of Continuous Glucose Monitoring (CGM) / Real-Time Flash Glucose Scanning (FGS) in Type 1 Diabetes Mellitus in Children and Young People Under
DIABETES BLOOD GLUCOSE MONITORING AT HOME (ADULTS)
 Type 2 Diabetes DIABETES BLOOD GLUCOSE MONITORING AT HOME (ADULTS) Clinical Guideline Self-monitoring of blood glucose should be offered to all newly diagnosed type 2 diabetic patients as an integral part
Type 2 Diabetes DIABETES BLOOD GLUCOSE MONITORING AT HOME (ADULTS) Clinical Guideline Self-monitoring of blood glucose should be offered to all newly diagnosed type 2 diabetic patients as an integral part
Healthcare professionals involved in care of children and young people with Type 1 Diabetes Mellitus. Children and young people with diabetes mellitus
 ssociation of Children s Diabetes Clinical Guideline A Practical Approach to the Management of Continuous Glucose Monitoring (CGM) / Real-Time Flash Glucose Scanning (FGS) in Type 1 Diabetes Mellitus in
ssociation of Children s Diabetes Clinical Guideline A Practical Approach to the Management of Continuous Glucose Monitoring (CGM) / Real-Time Flash Glucose Scanning (FGS) in Type 1 Diabetes Mellitus in
Walsall CCG Supported Blood Glucose Meters April 2018
 Meter Name Picture Type Strips Lancets Information Company PRIMARY CARE INITIATION TEE2+ Uncomplicated Type 2 diabetes patient on TEE2 test CareSens Lancets Easy to use. Large clear display Bluetooth functionality
Meter Name Picture Type Strips Lancets Information Company PRIMARY CARE INITIATION TEE2+ Uncomplicated Type 2 diabetes patient on TEE2 test CareSens Lancets Easy to use. Large clear display Bluetooth functionality
Implementation of Flash Glucose Monitoring
 Equality Analysis (Health Inequalities, Human Rights, Social Value) Implementation of Flash Glucose Monitoring Before completing this equality analysis it is recommended that you: Contact your equality
Equality Analysis (Health Inequalities, Human Rights, Social Value) Implementation of Flash Glucose Monitoring Before completing this equality analysis it is recommended that you: Contact your equality
Corporate Medical Policy
 Corporate Medical Policy Continuous Monitoring of Glucose in the Interstitial Fluid File Name: Origination: Last CAP Review: Next CAP Review: Last Review: continuous_monitoring_of_glucose_in_the_interstitial_fluid
Corporate Medical Policy Continuous Monitoring of Glucose in the Interstitial Fluid File Name: Origination: Last CAP Review: Next CAP Review: Last Review: continuous_monitoring_of_glucose_in_the_interstitial_fluid
Continuous Glucose Monitoring (CGM)/Real-Time Flash Glucose Scanning (FGS) Training for Healthcare Professionals and Patients
 ssociation of Children s Diabetes Continuous Glucose Monitoring (CGM)/Real-Time Flash Glucose Scanning (FGS) Training for Healthcare Professionals and Patients 1 ssociation of Children s Diabetes Contents
ssociation of Children s Diabetes Continuous Glucose Monitoring (CGM)/Real-Time Flash Glucose Scanning (FGS) Training for Healthcare Professionals and Patients 1 ssociation of Children s Diabetes Contents
GDm-Health is free to download and use. Its use may result in efficiency savings from reducing face-to-face clinic appointments.
 pat hways Health app: GDm-Health for people with gestational diabetes Medtech innovation briefing Published: 13 November 2017 nice.org.uk/guidance/mib131 Summary About this app GDm-Health is a health application
pat hways Health app: GDm-Health for people with gestational diabetes Medtech innovation briefing Published: 13 November 2017 nice.org.uk/guidance/mib131 Summary About this app GDm-Health is a health application
NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE. Health Technology Appraisal
 NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE Health Technology Appraisal Continuous subcutaneous insulin infusion for the treatment of diabetes (review) Final scope Appraisal objective To review
NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE Health Technology Appraisal Continuous subcutaneous insulin infusion for the treatment of diabetes (review) Final scope Appraisal objective To review
COPYRIGHTED MATERIAL. Chapter 1 An Introduction to Insulin Pump Therapy WHAT IS INSULIN PUMP THERAPY?
 Chapter 1 An Introduction to Insulin Pump Therapy This chapter will provide information on what insulin pump therapy is, and how insulin pumps have developed from the early models introduced in the 1970s
Chapter 1 An Introduction to Insulin Pump Therapy This chapter will provide information on what insulin pump therapy is, and how insulin pumps have developed from the early models introduced in the 1970s
CONTINUOUS OR INTERMITTENT GLUCOSE MONITORING IN INTERSTITIAL FLUID
 FLUID Non-Discrimination Statement and Multi-Language Interpreter Services information are located at the end of this document. Coverage for services, procedures, medical devices and drugs are dependent
FLUID Non-Discrimination Statement and Multi-Language Interpreter Services information are located at the end of this document. Coverage for services, procedures, medical devices and drugs are dependent
Quick Reference Guide
 FLASH GLUCOSE MONITORING SYSTEM Quick Reference Guide FreeStyle LibreLink app A FreeStyle Libre product IMPORTANT USER INFORMATION Before you use your System, review all the product instructions and the
FLASH GLUCOSE MONITORING SYSTEM Quick Reference Guide FreeStyle LibreLink app A FreeStyle Libre product IMPORTANT USER INFORMATION Before you use your System, review all the product instructions and the
Diagnostics guidance Published: 12 February 2016 nice.org.uk/guidance/dg21
 Integrated sensor-augmented pump therapy systems for managing blood glucose levels els in type 1 diabetes (the MiniMed Paradigm adigm Veo system and the Vibe and G4 PLATINUM CGM system) Diagnostics guidance
Integrated sensor-augmented pump therapy systems for managing blood glucose levels els in type 1 diabetes (the MiniMed Paradigm adigm Veo system and the Vibe and G4 PLATINUM CGM system) Diagnostics guidance
How can I access flash glucose monitoring if I need it? Support pack. This pack will help you to find out more about flash and how you can access it.
 How can I access flash glucose monitoring if I need it? Support pack This pack will help you to find out more about flash and how you can access it. Reviewed March 2019 Introduction Following several major
How can I access flash glucose monitoring if I need it? Support pack This pack will help you to find out more about flash and how you can access it. Reviewed March 2019 Introduction Following several major
Type 2 Diabetes Recommended SMBG
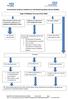 Herefordshire Diabetes Guideline for Self-Monitoring Blood Glucose (SMBG) Type 2 Diabetes Recommended SMBG Diet controlled/ metformin only/ combination of metformin and pioglitazone/dpp4/sglt2 or GLP1
Herefordshire Diabetes Guideline for Self-Monitoring Blood Glucose (SMBG) Type 2 Diabetes Recommended SMBG Diet controlled/ metformin only/ combination of metformin and pioglitazone/dpp4/sglt2 or GLP1
Diabetes UK Consensus Guideline for Flash Glucose Monitoring
 Diabetes UK Consensus Guideline for Flash Glucose Monitoring Date published Introduction Our recommendations Overview Who is flash glucose monitoring suitable for? What is the clinical evidence for flash
Diabetes UK Consensus Guideline for Flash Glucose Monitoring Date published Introduction Our recommendations Overview Who is flash glucose monitoring suitable for? What is the clinical evidence for flash
Quick Reference Guide
 FLASH GLUCOSE MONITORING SYSTEM Quick Reference Guide IMPORTANT USER INFORMATION Before you use your System, review all the product instructions and the Interactive Tutorial. The Quick Reference Guide
FLASH GLUCOSE MONITORING SYSTEM Quick Reference Guide IMPORTANT USER INFORMATION Before you use your System, review all the product instructions and the Interactive Tutorial. The Quick Reference Guide
Guidance on the Self-Monitoring of Blood Glucose in Adults with Diabetes
 Introduction Guidance on the Self-Monitoring of Blood Glucose in Adults with Diabetes This guideline is designed to offer guidance for primary and secondary care on the use of selfmonitoring of blood glucose
Introduction Guidance on the Self-Monitoring of Blood Glucose in Adults with Diabetes This guideline is designed to offer guidance for primary and secondary care on the use of selfmonitoring of blood glucose
DO IT TIPS & TRICKS YOU CAN WITHOUT LANCETS * with FreeStyle Libre FLASH GLUCOSE MONITORING SYSTEM
 FLASH GLUCOSE MONITORING SYSTEM YOU CAN DO IT WITHOUT LANCETS * TIPS & TRICKS with FreeStyle Libre *Scanning the sensor to obtain glucose values does not require lancets. FreeStyle Libre overview 1 The
FLASH GLUCOSE MONITORING SYSTEM YOU CAN DO IT WITHOUT LANCETS * TIPS & TRICKS with FreeStyle Libre *Scanning the sensor to obtain glucose values does not require lancets. FreeStyle Libre overview 1 The
Insulin Pumps and Glucose Monitors in Adults Policy
 Insulin Pumps and Glucose Monitors in Adults Policy Version: 2016-19 Ratified by: NHS Leeds West CCG Assurance Committee on; 16 November 2016 NHS Leeds North CCG Governance on Performance and Risk Committee
Insulin Pumps and Glucose Monitors in Adults Policy Version: 2016-19 Ratified by: NHS Leeds West CCG Assurance Committee on; 16 November 2016 NHS Leeds North CCG Governance on Performance and Risk Committee
Regional Medicines Optimisation Committee (North)
 Present: Regional Medicines Optimisation Committee (North) October 26 th 2017 Minutes Quarry House, Leeds 10:00-1:00 pm Name Title Organisation Jun Oct Dr Mike Prentice (MP) Chair Michele Cossey (MC) Dr
Present: Regional Medicines Optimisation Committee (North) October 26 th 2017 Minutes Quarry House, Leeds 10:00-1:00 pm Name Title Organisation Jun Oct Dr Mike Prentice (MP) Chair Michele Cossey (MC) Dr
QOF Indicator DM013:
 QOF Indicator DM013: The percentage of patients with diabetes, on the register, who have a record of a dietary review by a suitably competent professional in the preceding 12 months Note: the bold signposts
QOF Indicator DM013: The percentage of patients with diabetes, on the register, who have a record of a dietary review by a suitably competent professional in the preceding 12 months Note: the bold signposts
Quick Reference Guide
 FLASH GLUCOSE MONITORING SYSTEM Quick Reference Guide IMPORTANT USER INFORMATION Before you use your System, review all the product instructions and the Interactive Tutorial. The Quick Reference Guide
FLASH GLUCOSE MONITORING SYSTEM Quick Reference Guide IMPORTANT USER INFORMATION Before you use your System, review all the product instructions and the Interactive Tutorial. The Quick Reference Guide
Insulin Pumps and Glucose Monitors in Adults, Children and Young People Policy
 Insulin Pumps and Glucose Monitors in Adults, Children and Young People Policy Version: 2017-20 Ratified by: NHS Leeds West CCG Assurance Committee on; 16 November 2016 NHS Leeds North CCG Governance on
Insulin Pumps and Glucose Monitors in Adults, Children and Young People Policy Version: 2017-20 Ratified by: NHS Leeds West CCG Assurance Committee on; 16 November 2016 NHS Leeds North CCG Governance on
June 2016 Review: June 2019
 BEDFORDSHIRE & LUTON JOINT PRESCRIBING COMMITTEE June 2016 Review: June 2019 Bulletin 241 : Guidelines on self-monitoring of blood glucose (SMBG) in non-insulin treated Type 2 diabetic patients for maintaining
BEDFORDSHIRE & LUTON JOINT PRESCRIBING COMMITTEE June 2016 Review: June 2019 Bulletin 241 : Guidelines on self-monitoring of blood glucose (SMBG) in non-insulin treated Type 2 diabetic patients for maintaining
Audit support for continuous subcutaneous insulin infusion for the treatment of diabetes mellitus (review of technology appraisal guidance 57)
 Audit support for continuous subcutaneous insulin (review of technology appraisal guidance 57) Issue date: 2008 Audit support Continuous subcutaneous insulin infusion for the treatment of diabetes mellitus
Audit support for continuous subcutaneous insulin (review of technology appraisal guidance 57) Issue date: 2008 Audit support Continuous subcutaneous insulin infusion for the treatment of diabetes mellitus
NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE GUIDANCE EXECUTIVE (GE)
 NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE GUIDANCE EXECUTIVE (GE) Review of TA151 Continuous subcutaneous insulin infusion for the treatment of diabetes mellitus This guidance was issued in
NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE GUIDANCE EXECUTIVE (GE) Review of TA151 Continuous subcutaneous insulin infusion for the treatment of diabetes mellitus This guidance was issued in
In response to an enquiry from the Scottish Diabetes Group (SDG)
 Evidence Note Number 81 July 2018 In response to an enquiry from the Scottish Diabetes Group (SDG) What is the clinical and cost effectiveness of Freestyle Libre flash glucose monitoring for patients with
Evidence Note Number 81 July 2018 In response to an enquiry from the Scottish Diabetes Group (SDG) What is the clinical and cost effectiveness of Freestyle Libre flash glucose monitoring for patients with
FLIPS FreeStyle Libre in Pregnancy Study
 FLIPS FreeStyle Libre in Pregnancy Study Evaluation of the Accuracy of the FreeStyle Libre Flash Glucose Monitoring System Use in Pregnancy Label Extension Study (CE) Section 1: PARTICIPANT INFORMATION
FLIPS FreeStyle Libre in Pregnancy Study Evaluation of the Accuracy of the FreeStyle Libre Flash Glucose Monitoring System Use in Pregnancy Label Extension Study (CE) Section 1: PARTICIPANT INFORMATION
Diabetes Subcommittee of PTAC meeting held 3 March (minutes for web publishing)
 Diabetes Subcommittee of PTAC meeting held 3 March 2011 (minutes for web publishing) Diabetes Subcommittee minutes are published in accordance with the Terms of Reference for the Pharmacology and Therapeutics
Diabetes Subcommittee of PTAC meeting held 3 March 2011 (minutes for web publishing) Diabetes Subcommittee minutes are published in accordance with the Terms of Reference for the Pharmacology and Therapeutics
NEW TECHNOLOGIES FOR MANAGING DIABETES ANGELA THOMPSON DNP, FNP-C, BC-ADM, CDE, FAANP
 NEW TECHNOLOGIES FOR MANAGING DIABETES ANGELA THOMPSON DNP, FNP-C, BC-ADM, CDE, FAANP No commercial support or sponsorship was received for this project I have nothing to disclose OBJECTIVES Identify at
NEW TECHNOLOGIES FOR MANAGING DIABETES ANGELA THOMPSON DNP, FNP-C, BC-ADM, CDE, FAANP No commercial support or sponsorship was received for this project I have nothing to disclose OBJECTIVES Identify at
Executive Summary Management of Type 1 Diabetes Mellitus during illness in children and young people under 18 years (Sick Day Rules)
 Executive Summary Management of Type 1 Diabetes Mellitus during illness in children and young people under 18 years (Sick Day Rules) SETTING FOR STAFF PATIENTS Medical and nursing staff Children and young
Executive Summary Management of Type 1 Diabetes Mellitus during illness in children and young people under 18 years (Sick Day Rules) SETTING FOR STAFF PATIENTS Medical and nursing staff Children and young
People with type 1 diabetes and Do It Yourself (DIY) technology solutions ABOUT THIS POSITION STATEMENT
 Position Statement People with type 1 diabetes and Do It Yourself (DIY) technology solutions ABOUT THIS POSITION STATEMENT Diabetes Australia believes that people with diabetes should have choice and access
Position Statement People with type 1 diabetes and Do It Yourself (DIY) technology solutions ABOUT THIS POSITION STATEMENT Diabetes Australia believes that people with diabetes should have choice and access
Guidance on the Self-Monitoring of Blood Glucose in Adults with Diabetes
 Introduction Guidance on the Self-Monitoring of Blood Glucose in Adults with Diabetes This guideline is designed to offer guidance for primary and secondary care on the use of selfmonitoring of blood glucose
Introduction Guidance on the Self-Monitoring of Blood Glucose in Adults with Diabetes This guideline is designed to offer guidance for primary and secondary care on the use of selfmonitoring of blood glucose
Advances in Diabetes Care Technologies
 1979 Advances in Diabetes Care Technologies 2015 Introduction Insulin pump use: ~ 20% - 30% of patients with T1DM < 1% of insulin-treated patients with T2DM 2007 FDA estimates ~375,000 insulin pumps for
1979 Advances in Diabetes Care Technologies 2015 Introduction Insulin pump use: ~ 20% - 30% of patients with T1DM < 1% of insulin-treated patients with T2DM 2007 FDA estimates ~375,000 insulin pumps for
Priorities Advisory Committee
 Insulin degludec (Tresiba ) PAC - Insulin degludec (Tresiba ) 2.1 The East of England Priorities Advisory Committee A function of GUIDANCE STATEMENT PAC Recommendations 1. Insulin degludec is NOT recommended
Insulin degludec (Tresiba ) PAC - Insulin degludec (Tresiba ) 2.1 The East of England Priorities Advisory Committee A function of GUIDANCE STATEMENT PAC Recommendations 1. Insulin degludec is NOT recommended
Medical Policy An independent licensee of the Blue Cross Blue Shield Association
 Continuous or Intermittent Monitoring of Glucose in Interstitial Fluid Page 1 of 26 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: Continuous or Intermittent Monitoring
Continuous or Intermittent Monitoring of Glucose in Interstitial Fluid Page 1 of 26 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: Continuous or Intermittent Monitoring
What is the role of insulin pumps in the modern day care of patients with Type 1 diabetes?
 What is the role of insulin pumps in the modern day care of patients with Type 1 diabetes? Dr. Fiona Wotherspoon Consultant in Diabetes and Endocrinology Dorset County Hospital Fiona.Wotherspoon@dchft.nhs.uk
What is the role of insulin pumps in the modern day care of patients with Type 1 diabetes? Dr. Fiona Wotherspoon Consultant in Diabetes and Endocrinology Dorset County Hospital Fiona.Wotherspoon@dchft.nhs.uk
Diabetes in England NHS Medical Directorate. Dr Rowan Hillson MBE National Clinical Director for Diabetes
 Diabetes in England 2010 Dr Rowan Hillson MBE National Clinical Director for Diabetes I strongly believe that Everyone with diabetes deserves the highest standards of personalised diabetes care, no matter
Diabetes in England 2010 Dr Rowan Hillson MBE National Clinical Director for Diabetes I strongly believe that Everyone with diabetes deserves the highest standards of personalised diabetes care, no matter
Sandwell & West Birmingham integrated community care diabetes model (DICE) the future of diabetes services?
 Sandwell & West Birmingham integrated community care diabetes model (DICE) the future of diabetes services? Dr PARIJAT DE DUK Clinical Champion Clinical Lead for Diabetes & Endocrinology, Sandwell & West
Sandwell & West Birmingham integrated community care diabetes model (DICE) the future of diabetes services? Dr PARIJAT DE DUK Clinical Champion Clinical Lead for Diabetes & Endocrinology, Sandwell & West
The management of diabetes
 MEDICINE DIGEST STATINS The management of diabetes should include efforts to prevent, or delay, the onset of cardiovascular disease. Statins have been shown to lower cardiovascular risk in diabetes and
MEDICINE DIGEST STATINS The management of diabetes should include efforts to prevent, or delay, the onset of cardiovascular disease. Statins have been shown to lower cardiovascular risk in diabetes and
BEDFORDSHIRE AND LUTON JOINT PRESCRIBING COMMITTEE (JPC)
 BEDFORDSHIRE AND LUTON JOINT PRESCRIBING COMMITTEE (JPC) June 2017 Review: June 2020 (earlier if required see recommendations) Bulletin 255: Insulin aspart New Formulation - Fiasp JPC Recommendations:
BEDFORDSHIRE AND LUTON JOINT PRESCRIBING COMMITTEE (JPC) June 2017 Review: June 2020 (earlier if required see recommendations) Bulletin 255: Insulin aspart New Formulation - Fiasp JPC Recommendations:
Networking for success: A burning platform in Berkshire West
 SERVICE REDESIGN CASE STUDY 1: NOVEMBER 2014 Networking for success: A burning platform in Berkshire West SUMMARY In 2012, four federated CCGs set up a network to redesign diabetes services in Berkshire
SERVICE REDESIGN CASE STUDY 1: NOVEMBER 2014 Networking for success: A burning platform in Berkshire West SUMMARY In 2012, four federated CCGs set up a network to redesign diabetes services in Berkshire
EFFECTIVE SHARE CARE AGREEMENT. FOR THE off license use of GLP1 mimetics in combination with insulin IN DUDLEY
 Specialist details Patient identifier Name Tel: EFFECTIVE SHARE CARE AGREEMENT FOR THE off license use of GLP1 mimetics in combination with insulin IN DUDLEY The aim of Effective Shared Care Guidelines
Specialist details Patient identifier Name Tel: EFFECTIVE SHARE CARE AGREEMENT FOR THE off license use of GLP1 mimetics in combination with insulin IN DUDLEY The aim of Effective Shared Care Guidelines
Blood Glucose Level (BGL) greater than or equal to 15.0 mmol/l
 DIABETES ACTION PLAN 2019 EARLY CHILDHOOD SETTINGS Use in conjunction with Diabetes Management Plan. This plan should be reviewed every year. Twice daily injections LOW Hypoglycaemia (Hypo) Blood Glucose
DIABETES ACTION PLAN 2019 EARLY CHILDHOOD SETTINGS Use in conjunction with Diabetes Management Plan. This plan should be reviewed every year. Twice daily injections LOW Hypoglycaemia (Hypo) Blood Glucose
Commissioning for Outcomes in Diabetes. Joanne Taylor Primary Care Commissioning Manager
 Commissioning for Outcomes in Diabetes Joanne Taylor Primary Care Commissioning Manager Current Diabetes Model in Dudley History and Current Diabetes Pathway Primary Care 46 2 no LIS practices Annual review
Commissioning for Outcomes in Diabetes Joanne Taylor Primary Care Commissioning Manager Current Diabetes Model in Dudley History and Current Diabetes Pathway Primary Care 46 2 no LIS practices Annual review
Control of Glycemic Variability for Reducing Hypoglycemia Jae Hyeon Kim
 Control of Glycemic Variability for Reducing Hypoglycemia Jae Hyeon Kim Division of Endocrinology and Metabolism, Samsung Medical Center, Sungkyunkwan University School of Medicine Conflict of interest
Control of Glycemic Variability for Reducing Hypoglycemia Jae Hyeon Kim Division of Endocrinology and Metabolism, Samsung Medical Center, Sungkyunkwan University School of Medicine Conflict of interest
GOVERNING BOARD. Assisted Conception (IVF): Review of access criteria. Date of Meeting 21 January 2015 Agenda Item No 13. Title
 GOVERNING BOARD Date of Meeting 21 January 2015 Agenda Item No 13 Title Assisted Conception (IVF): Review of access criteria Purpose of Paper The SHIP (Southampton, Hampshire, Isle of Wight and Portsmouth)
GOVERNING BOARD Date of Meeting 21 January 2015 Agenda Item No 13 Title Assisted Conception (IVF): Review of access criteria Purpose of Paper The SHIP (Southampton, Hampshire, Isle of Wight and Portsmouth)
Advances in Diabetes Care Technologies
 1979 Advances in Diabetes Care Technologies 2015 Introduction Roughly 20% - 30% of patients with T1DM and fewer than 1% of insulin-treated patients with T2DM use an insulin pump In 2007, the US FDA estimated
1979 Advances in Diabetes Care Technologies 2015 Introduction Roughly 20% - 30% of patients with T1DM and fewer than 1% of insulin-treated patients with T2DM use an insulin pump In 2007, the US FDA estimated
Country Health SA Local Health Network. Title: Continuous Glucose Monitoring (CGM) and Flash Glucose Monitoring (FGM) Ambulatory Service
 Protocol (Clinical) Title: Continuous Glucose Monitoring (CGM) and Flash Glucose Monitoring (FGM) Ambulatory Service Protocol developed by: CHSALHN Diabetes Service Protocol Sponsor: CHSALHN, Executive
Protocol (Clinical) Title: Continuous Glucose Monitoring (CGM) and Flash Glucose Monitoring (FGM) Ambulatory Service Protocol developed by: CHSALHN Diabetes Service Protocol Sponsor: CHSALHN, Executive
Continuous or Intermittent Glucose Monitoring in Interstitial Fluid
 Continuous or Intermittent Glucose Monitoring in Interstitial Fluid Policy Number: 1.01.20 Last Review: 9/2018 Origination: 11/2001 Next Review: 9/2019 Policy Blue Cross and Blue Shield of Kansas City
Continuous or Intermittent Glucose Monitoring in Interstitial Fluid Policy Number: 1.01.20 Last Review: 9/2018 Origination: 11/2001 Next Review: 9/2019 Policy Blue Cross and Blue Shield of Kansas City
Integrated Community Diabetes Services (ICDS) GP Referral Guide Version 3 - October 2014
 Integrated Community Diabetes Services (ICDS) GP Referral Guide Version 3 - October 2014 Introduction The Integrated Community Diabetes Service (ICDS) will deliver high quality care to individuals who
Integrated Community Diabetes Services (ICDS) GP Referral Guide Version 3 - October 2014 Introduction The Integrated Community Diabetes Service (ICDS) will deliver high quality care to individuals who
Monitoring in Type 2 Diabetes. Learning Outcomes. Type 2 Diabetes. Senga Hunter Community Diabetes Specialist Nurse
 Monitoring in Type 2 Diabetes Senga Hunter Community Diabetes Specialist Nurse Learning Outcomes Understand why blood monitoring is necessary Understand the blood tests for monitoring diabetes Understand
Monitoring in Type 2 Diabetes Senga Hunter Community Diabetes Specialist Nurse Learning Outcomes Understand why blood monitoring is necessary Understand the blood tests for monitoring diabetes Understand
Health technology assessment report in support of county council decisionmaking
 Health technology assessment report in support of county council decisionmaking FreeStyle Libre Method for self-monitoring of glucose level in people with type 1 and 2 diabetes treated with basal insulin
Health technology assessment report in support of county council decisionmaking FreeStyle Libre Method for self-monitoring of glucose level in people with type 1 and 2 diabetes treated with basal insulin
Continuous Glucose Monitoring in Women With Type 1 Diabetes in Pregnancy Trial
 Continuous Glucose Monitoring in Women With Type 1 Diabetes in Pregnancy Trial Dr Sandra Neoh on behalf of: Dr Denice Feig and Professor Helen Murphy Funders: JDRF (Juvenile Diabetes Research Foundation),
Continuous Glucose Monitoring in Women With Type 1 Diabetes in Pregnancy Trial Dr Sandra Neoh on behalf of: Dr Denice Feig and Professor Helen Murphy Funders: JDRF (Juvenile Diabetes Research Foundation),
GUIDELINE FOR SELF MONITORING OF BLOOD GLUCOSE IN ADULTS WITH DIABETES
 It is recognised that self- monitoring of blood glucose (SMBG) may have an important role to play for some patients with. It can allow individuals to see what impact particular behaviours, such as dietary
It is recognised that self- monitoring of blood glucose (SMBG) may have an important role to play for some patients with. It can allow individuals to see what impact particular behaviours, such as dietary
New NICE guidance: Changes in practice for multidisciplinary teams. Part 1: Type 1 diabetes in children and young people
 Article New NICE guidance: Changes in practice for multidisciplinary teams. Part 1: Type 1 diabetes in children and young people Helen Thornton This is the first of two articles on the 2015 NICE NG18 guideline,
Article New NICE guidance: Changes in practice for multidisciplinary teams. Part 1: Type 1 diabetes in children and young people Helen Thornton This is the first of two articles on the 2015 NICE NG18 guideline,
Insulin Pumps and Continuous Glucose Sensors- Embracing Technology. Susan Cavalier, BS, RN, CDE Manager, Diabetes Educator Sanford Diabetes Education
 Insulin Pumps and Continuous Glucose Sensors- Embracing Technology Susan Cavalier, BS, RN, CDE Manager, Diabetes Educator Sanford Diabetes Education Diabetes Management Tools in 1974 Insulin pump history
Insulin Pumps and Continuous Glucose Sensors- Embracing Technology Susan Cavalier, BS, RN, CDE Manager, Diabetes Educator Sanford Diabetes Education Diabetes Management Tools in 1974 Insulin pump history
DATE RECEIVED 09/10/2017 SUBJECT Formulary PASSED TO Mary DATE PASSED 09/10/2017 RESPOND BY 27/10/2017 CATEGORY Business FoI NUMBER
 DATE RECEIVED 09/10/2017 SUBJECT Formulary PASSED TO Mary DATE PASSED 09/10/2017 RESPOND BY 27/10/2017 CATEGORY Business FoI NUMBER 2017-404 Question/s to be Answered Can you please provide the following
DATE RECEIVED 09/10/2017 SUBJECT Formulary PASSED TO Mary DATE PASSED 09/10/2017 RESPOND BY 27/10/2017 CATEGORY Business FoI NUMBER 2017-404 Question/s to be Answered Can you please provide the following
Preconception advice for women with type 1 and 2 diabetes. Points to consider before or as soon as you learn that you are pregnant.
 Preconception advice for women with type 1 and 2 diabetes Points to consider before or as soon as you learn that you are pregnant. General advice for women planning pregnancy Folic acid tablets: Doctors
Preconception advice for women with type 1 and 2 diabetes Points to consider before or as soon as you learn that you are pregnant. General advice for women planning pregnancy Folic acid tablets: Doctors
15 th Annual DAFNE collaborative meeting Tuesday 28 th June 2016
 15 th Annual DAFNE collaborative meeting Tuesday 28 th June 2016 Sponsored by: Abbott Diabetes Care and Lilly Diabetes The REPOSE Trial (Relative Effectiveness of Pumps over Structured Education) Background
15 th Annual DAFNE collaborative meeting Tuesday 28 th June 2016 Sponsored by: Abbott Diabetes Care and Lilly Diabetes The REPOSE Trial (Relative Effectiveness of Pumps over Structured Education) Background
Blood Glucose Level (BGL) greater than or equal to 15.0 mmol/l
 DIABETES ACTION PLAN 2019 SCHOOL SETTING Use in conjunction with Diabetes Management Plan. This plan should be reviewed every year. Multiple daily injections LOW Hypoglycaemia (Hypo) Blood Glucose Level
DIABETES ACTION PLAN 2019 SCHOOL SETTING Use in conjunction with Diabetes Management Plan. This plan should be reviewed every year. Multiple daily injections LOW Hypoglycaemia (Hypo) Blood Glucose Level
The FreeStyle Libre Flash Glucose Monitoring System NEW STARTER CLINIC - LONDON
 The FreeStyle Libre Flash Glucose Monitoring System NEW STARTER CLINIC - LONDON The FreeStyle Libre Flash Glucose Monitoring System Indication of Use The FreeStyle Libre flash glucose monitoring system
The FreeStyle Libre Flash Glucose Monitoring System NEW STARTER CLINIC - LONDON The FreeStyle Libre Flash Glucose Monitoring System Indication of Use The FreeStyle Libre flash glucose monitoring system
Blood Glucose monitoring during extra-corporeal renal therapy and plasmapheresis.
 Blood Glucose monitoring during extra-corporeal renal therapy and plasmapheresis. Lead Clinician: Dr. R. Diwakar Implementation date: July 2013 Last updated: August 2017 Last review date: Planned review
Blood Glucose monitoring during extra-corporeal renal therapy and plasmapheresis. Lead Clinician: Dr. R. Diwakar Implementation date: July 2013 Last updated: August 2017 Last review date: Planned review
Temporary interruption in supply of APIDRA (insulin glulisine) - OPTISET and SOLOSTAR prefilled pens and CLIKSTAR reusable cartridges.
 September 2011 Dear Health Care Professional, Temporary interruption in supply of APIDRA (insulin glulisine) - OPTISET and SOLOSTAR prefilled pens and CLIKSTAR reusable cartridges. We would like to inform
September 2011 Dear Health Care Professional, Temporary interruption in supply of APIDRA (insulin glulisine) - OPTISET and SOLOSTAR prefilled pens and CLIKSTAR reusable cartridges. We would like to inform
Community alcohol detoxification in primary care
 Community alcohol detoxification in primary care 1. Purpose The purpose of this primary care enhanced service is to improve the health and quality of life of people whose health may be compromised by their
Community alcohol detoxification in primary care 1. Purpose The purpose of this primary care enhanced service is to improve the health and quality of life of people whose health may be compromised by their
Advances in Diabetes Care Technologies
 Advances in Diabetes Care Technologies 1979 2015 Introduction Roughly 20% to 30% of patients with T1DM and fewer than 1% of insulin-treated patients with T2DM use an insulin pump In 2007, the U.S. FDA
Advances in Diabetes Care Technologies 1979 2015 Introduction Roughly 20% to 30% of patients with T1DM and fewer than 1% of insulin-treated patients with T2DM use an insulin pump In 2007, the U.S. FDA
UvA-DARE (Digital Academic Repository) The artificial pancreas Kropff, J. Link to publication
 UvA-DARE (Digital Academic Repository) The artificial pancreas Kropff, J. Link to publication Citation for published version (APA): Kropff, J. (2017). The artificial pancreas: From logic to life General
UvA-DARE (Digital Academic Repository) The artificial pancreas Kropff, J. Link to publication Citation for published version (APA): Kropff, J. (2017). The artificial pancreas: From logic to life General
Update on Continuous Glucose Monitoring (CGM) Technology in Diabetes. Elena Toschi, MD November 12, 2016
 Update on Continuous Glucose Monitoring (CGM) Technology in Diabetes Elena Toschi, MD November 12, 2016 Presenter Disclosure Information Elena Toschi, MD No financial disclosure Objectives: Use of CGM
Update on Continuous Glucose Monitoring (CGM) Technology in Diabetes Elena Toschi, MD November 12, 2016 Presenter Disclosure Information Elena Toschi, MD No financial disclosure Objectives: Use of CGM
Guidance for the choice of blood glucose testing meters, strips and lancets
 Guidance for the choice of blood glucose testing meters, strips and lancets This guidance is intended to assist healthcare professionals in their selection of appropriate blood glucose meters and testing
Guidance for the choice of blood glucose testing meters, strips and lancets This guidance is intended to assist healthcare professionals in their selection of appropriate blood glucose meters and testing
All patients put on this combination must have been seen by a dietitian and made appropriate efforts to lose weight
 Shared Care Protocol: Addition of a glucagonlike peptide-1 (GLP-1) analogues (exenatide or liraglutide) to patients already on insulin who have poorly controlled type 2 diabetes All patients put on this
Shared Care Protocol: Addition of a glucagonlike peptide-1 (GLP-1) analogues (exenatide or liraglutide) to patients already on insulin who have poorly controlled type 2 diabetes All patients put on this
NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE SCOPE
 NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE 1 Guideline title SCOPE Diabetes in children and young people: diagnosis and management of type 1 and type 2 diabetes in children and young people
NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE 1 Guideline title SCOPE Diabetes in children and young people: diagnosis and management of type 1 and type 2 diabetes in children and young people
National Paediatric Diabetes Audit
 National Paediatric Diabetes Audit Parent and Carers Report 2015-16 Commissioned by the Healthcare Quality Improvement Partnership Managed by the Royal College of Paediatrics and Child Health National
National Paediatric Diabetes Audit Parent and Carers Report 2015-16 Commissioned by the Healthcare Quality Improvement Partnership Managed by the Royal College of Paediatrics and Child Health National
