The Journal of Physiology
|
|
|
- Marsha Parker
- 5 years ago
- Views:
Transcription
1 J Physiol (2012) pp Positive allosteric modulation reveals a specific role for mglu2 receptors in sensory processing in the thalamus C. S. Copeland, S. A. Neale and T. E. Salt Department of Visual Neuroscience, Institute of Ophthalmology, University College London, London, UK The Journal of Physiology Non-technicalsummary Neurones in the ventrobasal thalamic nucleus (VB) are the major source of somatosensory input to the cerebral cortex. Thorough investigation has identified neuronal circuits formed between the VB and the adjacent thalamic reticular nucleus (TRN); and Group II metabotropic glutamate (mglu) receptors located within this circuitry have been demonstrated as capable of modulating somatosensory transmission. There are two Group II mglu receptor subtypes, mglu2 and mglu3, and this study has demonstrated that there is an mglu2 component to the Group II mglu receptor effect on sensory transmission within the VB, and that the mglu2 receptor subtype may be activated physiologically upon sensory stimulation. It is believed that this circuitry is of importance in the control of sensory discriminative processes, and we propose that mglu2 functions within this circuitry to enable relevant sensory information to be discerned from background activity. Abstract Group II metabotropic glutamate receptor (mglu) modulation of sensory processing in the rat ventrobasal thalamic nucleus (VB) has been extensively studied in vivo. However, it is not yet known what the relative contributions are of the Group II mglu receptor subtypes (mglu2 and mglu3) to this modulation, nor to what extent these receptors may be activated under physiological conditions during this process. Using single-neurone recording in the rat VB in vivo with local application of the selective Group II agonist LY and the subtype selective mglu2 positive allosteric modulator (PAM) LY487379, our findings were twofold. Firstly, we found that there is an mglu2 component to the effects of LY on sensory responses in the VB. Secondly, we have demonstrated that application of the PAM alone can modulate sensory responses of single neurones in vivo. This indicates that mglu2 receptors can be activated by endogenous agonist following physiological sensory stimulation. We speculate that the mglu2 subtype could be activated under physiological stimulus-evoked conditions by glutamate spillover from synapses between excitatory sensory afferents and VB neurones that can lead to a reduction in sensory-evoked inhibition arising from the thalamic reticular nucleus (TRN). We propose that this potential mglu2 receptor modulation of inhibition could play an important role in discerning relevant information from background activity upon physiological sensory stimulation. Furthermore, this could be a site of action for mglu2 PAMs to modulate cognitive processes. (Received 29 August 2011; accepted after revision 20 December 2011; first published online 23 December 2011) Correspondingauthor C. S. Copeland: Department of Visual Neuroscience, UCL Institute of Ophthalmology, University College London, Bath Street, London EC1V 9EL, UK. c.copeland@ucl.ac.uk Abreviations mglu, metabotropic glutamate receptor; mglu1, metabotropic glutamate receptor subtype 1; mglu2, metabotropic glutamate receptor subtype 2, mglu3, metabotropic glutamate receptor subtype 3; mglu4, metabotropic glutamate receptor subtype 4; mglu5, metabotropic glutamate receptor subtype 5; mglu6, metabotropic glutamate receptor subtype 6; mglu8, metabotropic glutamate receptor subtype 8; MWE, excitation to multiple whiskers; PAM, positive allosteric modulator; PSTH, poststimulus time histogram; SWE, excitation to a single whisker; TRN, thalamic reticular nucleus; VB, ventrobasal thalamic nucleus. DOI: /jphysiol
2 938 C. S. Copeland and others J Physiol Introduction Somatosensory information ascends to the cerebral cortex via the ventrobasal thalamic nucleus (VB). Common to all thalamic nuclei, the VB possesses excitatory glutamatergic and inhibitory GABAergic synapses that contribute to the overall gating and processing of sensory information. TherodentVBcontainsonlyonemajorcelltype,the thalamocortical relay neurone (Peschanski et al. 1984; Harris, 1986; Ohara & Havton, 1994), and is largely devoid of intrinsic Golgi II inhibitory interneurons, receiving its GABAergic input almost exclusively from the adjacent thalamic reticular nucleus (TRN) (Ralston, 1983; Barbaresi et al. 1986; Harris & Hendrickson, 1987). The TRN is innervated by collaterals from VB relay neurone axons and by layer VI-originating corticothalamic fibres, both of which traverse the TRN. Thus the TRN is thought to coordinate the activation of appropriate thalamocortical circuits during selective attention (Crick, 1984; Pinault, 2004). GABAergic innervation from the TRN strongly influences how VB neurones process sensory information, making it important to understand how this inhibition is controlled. The eight known metabotropic glutamate receptor subtypes (mglu1 mglu8) are divided into three groups based upon their sequence homology, signal transduction mechanism and pharmacology (Nakanishi, 1992; Conn & Pin, 1997; Niswender & Conn, 2010). In general, the Group I receptors (mglu1, mglu5) have predominately postsynaptic actions, whereas the Group II (mglu2, mglu3) and Group III (mglu4, mglu6 8) receptors have predominately presynaptic actions. However, the relative locations of these receptor subtypes is now evidently more complex than this, with receptors from all three groups possessing post-, pre- or extrasynaptic actions (Conn & Pin, 1997; Niswender & Conn, 2010). There is considerable evidence to indicate that local application of selective Group II mglu receptor agonists in vivo can modulate somatosensory transmission within the rat VB by reducing TRN-originating inhibition (Salt & Eaton, 1995a; Salt & Turner, 1998). These findings are supported by ultra-structural studies of the rodent VB: Group II mglu receptors appear to be localised in glial processes surrounding GABAergic terminals (Liu et al. 1998; Mineff & Valtschanoff, 1999), and mglu3 can be found on TRN-originating axons (Tamaru et al. 2001). However, the relative contributions of the mglu2 and mglu3 subtypes to the observed Group II receptor effects is not known, nor to what extent these receptors may be activated under physiological conditions. The recent development of selective positive allosteric modulators (PAMs) for mglu2 (Niswender & Conn, 2010) enables the specific identification of mglu2 receptor-mediated components to responses. Such compounds typically potentiate orthosteric agonist-evoked responses whilst having relatively little direct effect on receptor activity (Ehlert, 1986; Christopoulos, 2002). Therefore, using an in vivo preparation with different sensory stimulation protocols and the PAM LY (Johnson et al. 2003) as a tool, the aim of the present study was twofold. Firstly, to identify whether there is an mglu2 component to the Group II effect on GABAergic inhibition in VB by investigating whether LY potentiates the effects of an exogenous Group II agonist, LY (Schoepp et al. 2003), on neurone responses. Secondly, to examine if mglu2 receptors are activated by endogenous neurotransmitter during the synaptic processing of sensory inputs by applying LY alone. Methods All experiments were conducted using adult male Wistar rats ( g). Animals were purchased from Harlan and were housed on a 12 h light dark cycle with unlimited access to food and water. All experimental conditions and procedures were in accordance with the UK Animals (Scientific Procedures) Act 1986 and associated guidelines, and the experiments comply with the policies and regulations of The Journal of Physiology (Drummond, 2009). Subjects (n = 47) were anaesthetised with urethane (1.2 g kg 1 I.P.) and were prepared for recording as detailed previously (Salt, 1987, 1989). Throughout the experiments electroencephalogram and electrocardiogram were monitored. Additional urethane anaesthetic was administered I.P. as required, and the experiment was terminated with an overdose of the same anaesthetic. Neuromuscular blocking agents were not used at any stage of the experiment. Seven-barrel recording and iontophoretic glass pipettes were advanced into the VB. Extracellular recordings were made from single VB neurones responsive to somatosensory input through the central barrel (filled with 4 M NaCl). Iontophoretic drug applications were performed using the outer barrels (Salt, 1987, 1989). On each occasion, one of the outer barrels was filled with 1 M NaCl for current balancing. The remaining outer barrels each contained one of the following substances: NMDA (50 mm, ph 8.0 in 150 mm NaCl), AMPA (25mM, ph 8.0 in 150 mm NaCl), (1S,2S,5R,6S)-2- aminobicyclo[3.1.0]hexane-2,6-dicarboxylic acid (LY354740; 5 mm, ph 8.0 in 75 mm NaCl), (2S)-2-amino- 2-[(1S,2S)-2-carboxycycloprop-1-yl]-3-(xanth-9-yl) propanoic acid (LY341495; 5 mm, ph 8.5 in 75 mm NaCl), as Na + salts, ejected as anions, and 2,2,2-trifluoro-N-[4-(2-methoxyphenoxy)phenyl]-N- (3-pyridinylmethyl)ethanesulfonamide hydrochloride (LY487379; 1 mm, ph 6.0, in 1% dimethyl sulfoxide (DMSO), 75 mm NaCl) and 1% DMSO (ph 6.0, 75 mm
3 J Physiol mglu2 positive allosteric modulation in the ventrobasal thalamus 939 NaCl), ejected as cations. All compounds were prevented from diffusing out of the pipette by using a retaining current (10 20 na) of opposite polarity to that of the ejection current. All compounds were obtained from Tocris. Throughout the study, extracellular single neurone action potentials were gated, timed and counted using a window discriminator, a CED1401 interface and Spike2 software, which recorded the output from the iontophoresis unit and also triggered some of the sensory stimuli sequences. Data were analysed by plotting poststimulus time histograms (PSTHs) from these recordings by counting the spikes evoked by either agonist ejection or sensory stimulation. Neurones were identified as VB neurones on the basis of stereotaxic location (Paxinos & Watson, 1998) and responses to vibrissa deflection. Vibrissa deflection was performed using fine air jets directed through 23 gauge needles mounted on micromanipulators positioned and orientated close to the vibrissa to ensure deflection of a single vibrissa was achieved. Air jets were electronically gated with solenoid valves that produced a rising air pulse at the vibrissa 8 ms after switching. Response latencies were calculated from the start of the gating pulse. Using such an approach it is possible to use air jets on adjacent vibrissae and only evoke an excitatory response from one of the vibrissa stimuli (Fig. 1), indicating the specificity of the stimulation procedure, as described previously (Salt, 1989). Prior to the beginning of each of the experimental protocols described below, the principal vibrissa (i.e. the vibrissa at the centre of the receptive field) for each neurone was identified, and responses to additional vibrissae were noted. Neurones could thus be classified as responding with excitation to a single whisker (SWE) or multiple whiskers (MWE), as described by other workers (Ito, 1988; Brecht & Sakmann, 2002). All neurones recorded from were quiescent. The effects of the Group II agonist and the mglu2 PAM on sensory responses Cycles of sensory stimulation (10 s long) were established and repeated continuously whilst recording from neurones. Cycles contained two types of sensory stimuli, which consisted of electronically gated short (10 30 ms) and long ( ms) duration air jets directed at the principal vibrissa, with 4 5 s interstimulus intervals. Depending upon the response profile of the neurone undergoing investigation, the long air jet was applied as either a continual stimulation or as a train of stimuli at a frequency of 5 7 Hz. After several control cycles displaying consistent neurone responses had been recorded, LY354740, LY and LY were iontophoretically ejected either alone or in conjunction with each other for 2 20 min as required. After cessation of LY354740, LY and/or LY ejection, sensory stimulation cycles were continued until neurone responses had returned to control levels. An inter-stimulus interval of 4 5 s was sufficient to ensure that any post-stimulus effects from either stimulus type were no longer apparent upon subsequent stimulation (Salt, 1989; Turner & Salt, 2003). This experimental protocol was also followed when the DMSO vehicle was iontophoretically ejected either alone or in conjunction with LY as appropriate. The effects of the Group II agonist and the mglu2 PAM on inhibitory sensory responses Individual VB neurones receive direct dominant input from a principal vibrissa via the trigeminal nuclei (Diamond et al. 1992; Nicolelis & Chapin, 1994; Pinault & Deschenes, 1998; Crabtree, 1999; Lavallee & Deschenes, 2004). VB neurone projections reach the cerebral cortex and the TRN, with the TRN inhibitory neurones receiving innervation from several vibrissae, which in turn make reciprocal projections back to the thalamus (Jones, 1985; Salt & Eaton, 1989; Shosaku et al. 1989; Pinault & Deschenes, 1998; Salt & Turner, 1998). Therefore, upon principal vibrissal stimulation, the responses of a VB neurone are the result of excitatory glutamatergic input and recurrent GABAergic inhibition (Salt, 1989; Shosaku et al. 1989; Lee et al. 1994; Hartings & Simons, 2000; Binns et al. 2003), whereas upon stimulation of surrounding secondary vibrissae a lateral GABAergic inhibition via the TRN is evoked (Salt, 1989; Pinault & Deschenes, 1998; Crabtree, 1999) (Fig. 1). In order to reveal these GABAergic inhibitory processes arising from the TRN, we used a condition-test protocol with two air jets each directed at an adjacent receptive field area (Salt, 1989). A conditioning stimulus presented to a secondary vibrissa preceding a test stimulus to a principal vibrissa by ms can inhibit the response of a VB neurone to stimulation of the principal vibrissa (Salt, 1989; Turner & Salt, 2003) (Fig. 1). The precise interval between presentation of the conditioning stimulus and the test stimulus was altered within the ms time window to produce the maximum inhibition achievable for each individual cell. The secondary vibrissa was normally in the same row as the principal vibrissa, but removed from the principal vibrissa by one or two positions (Lavallee & Deschenes, 2004). Several control cycles which each contained two sensory stimulations (presentation of the test stimulus alone and then the condition test stimuli, separated by an inter-stimulus interval of 4 5 s) that demonstrated the establishment of sensory inhibition were recorded, and these were then repeated during the concurrent application of LY and/or LY for 2 10 min as required. After cessation
4 940 C. S. Copeland and others J Physiol of LY and/or LY ejection, cycles were continued until neurone responses had returned to control levels. This experimental protocol was also followed when the DMSO vehicle was iontophoretically ejected either alone or in conjunction with LY as required. The degree of inhibition was quantified as a percentage as follows: Percentage inhibition = (1 T C /T) 100% where T is the total number of action potentials evoked to the test stimulus and T C is the total number of action potentials evoked to the test stimulus when preceded by the condition stimulus. Using this calculation, the maximum inhibitory response that can be achieved is 100%. Action potentials evoked ms following test stimuli were counted in analysis, as all responses occurred within 100 ms of stimulus presentation: the approximate time scale during which inhibition evoked by principal or secondary vibrissa stimulation is likely to reach its peak (Salt, 1989; Turner & Salt, 2003). It is important to note that the condition stimulus used to evoke an inhibition of a test response may in itself produce an excitatory response in some neurones. Aa Ba b b Figure 1. Circuitry underlying responses of VB neurones to vibrissa stimulation Aa, stimulation of the principal vibrissa drives excitation in the recorded VB neurone (VB), followed by recurrent inhibition via the TRN. Ab, PSTH record showing responses of the principal vibrissa when stimulated with a short-duration stimulus. Ba, stimulation of an adjacent secondary vibrissa drives lateral inhibition onto the recorded VB neurone, via the TRN. Bb, PSTH record showing responses when stimulation of the principal vibrissa is preceded by stimulation of the secondary vibrissa. Note the reduction in the response to the test stimulus (arrow). T: 20 ms short-duration test stimulation to the principal vibrissa; C: 20 ms conditioning stimulation to a secondary vibrissa; spike count: the total number of action potentials recorded per 2 ms bin over 6 trials. PSTHs were constructed using responses from the same cell (CVB050b).
5 J Physiol mglu2 positive allosteric modulation in the ventrobasal thalamus 941 The effects of the Group II agonist and the mglu2 PAM on lateral and recurrent inhibition As an alternative means of revealing functional recurrent or lateral inhibition onto VB neurones, NMDA was continuously applied to neurones whilst alternate presentations of short duration stimuli to either the principal or secondary vibrissa took place (separated by 5 s). NMDA ejections were adjusted to produce stable submaximal increases in action potential firing of neurones, typically into the range of Hz. The degree of inhibition produced when either the principal or secondary vibrissa was stimulated was quantified from cumulative PSTHs of action potential spikes before, during and after the stimulus was applied. The first 100 ms of evoked inhibition was used in the quantification analysis as this is the approximate time scale during which inhibition evoked by principal or secondary vibrissa stimulation is likely to reach its peak (Salt, 1989; Turner & Salt, 2003). Spikes counted during this period of inhibition are expressed as a percentage of the background firing rate. After several control cycles displaying consistent neurone responses had been recorded, either LY or LY was iontophoretically ejected for min as required. After cessation of LY or LY ejection, cycles were continued until neurone responses had returned to control levels. The effects of the Group II agonist and the mglu2 PAM on responses to ionotropic glutamate receptor agonist application In order to directly assess the effects of Group II mglu agents on postsynaptic responses to NMDA and AMPA receptor activation, cycles consisting of brief ejections (10 15 s) of NMDA or AMPA with intervals of s, were established and repeated continuously. NMDA and AMPA ejection parameters were adjusted to ensure that excitatory neuronal responses evoked by these ejections were submaximal. After several cycles of NMDA and AMPA responses had been recorded under control conditions, LY or LY was iontophoretically ejected during one to three cycles of NMDA/AMPA ejection as required. After cessation of LY or LY ejection, NMDA/AMPA ejection cycles were continued until neurone responses had returned to control levels. Statistical analysis Data are expressed as a percentage of control responses prior to Group II agonist and/or mglu2 PAM application (±SEM) and comparisons were made using Wilcoxon s matched-pairs test (P < 0.05). Results It has been previously demonstrated that the selective activation of Group II mglu receptors can modulate GABAergic afferent inhibition in VB (Salt & Eaton, 1995a; Salt & Turner, 1998; Turner & Salt, 2003). The results presented in this study complement these previous data whilst also providing evidence that there is an mglu2 component to this modulation. In addition, the present study has also elucidated a possible mechanism of action by which the mglu2 receptor subtype is activated in vivo during the synaptic processing of sensory inputs. The effects of the Group II agonist LY and the mglu2 PAM LY on sensory responses to short-duration vibrissa stimulation Initial investigation began by testing the effects of the Group II agonist and the mglu2 PAM on VB neurones with vibrissal receptive fields that were responsive to a short-duration stimulus directed at the principal vibrissa. Neuronal responses to short-duration stimuli were at a latency of ms (mean = 18.9 ± 0.71 ms) from stimulus onset. When this is adjusted for the time delay introduced by the air-jet stimulator (8 ms), these values correspond closely to those described by others (Ito, 1988; Brecht & Sakmann, 2002). Iontophoretic ejections of the Group II agonist alone significantly increased the responses of neurones to short-duration stimuli (128 ± 4% of control, n = 42) (Fig. 2Aa), as did application of the mglu2 PAM alone (128 ± 6% of control, n = 38) (Fig. 3A). In order to confirm the specificity of the agonist and PAM applications, the Group II antagonist LY was subsequently co-applied with these agents in a subset of the same neurones. LY reversed the effects of both the Group II agonist (LY alone: 138 ± 2% of control; LY plus LY ± 5% of control, n = 5), and the mglu2 PAM (LY alone: 127 ± 7% of control; LY plus LY ± 4% of control, n = 7) on neuronal responses to short-duration stimuli. On a subset of the neurones upon which the effects of the Group II agonist alone were tested, the mglu2 PAM was co-ejected with the Group II agonist and was found to significantly potentiate the agonist effect on neurone responses to the short-duration stimuli when compared to responses from the same neurones when the Group II agonist was applied alone (LY alone: 131 ± 6% of control; LY plus LY487379: 166 ± 12% of control, n = 22) (Fig. 2Ba). As a control for the use of DMSO vehicle, on some neurones an iontophoretic current was passed through the 1% DMSO solution that the mglu2 PAM was dissolved in at the same current as the PAM ejection on the same neurones, either alone or in conjunction with the Group II agonist as appropriate.
6 942 C. S. Copeland and others J Physiol Passing iontophoretic current through the vehicle barrel did not significantly alter responses of neurones to short-duration stimuli when applied alone (100 ± 2% of control, n = 10), nor did it significantly potentiate the responses of neurones when co-applied with the Group II agonist (LY alone: 124 ± 11% of control; LY plus 1% DMSO: 135 ± 13% of control, n = 10). Aa b Ba b Figure 2. VB neurone responses before, during and after iontophoretic application of the Group II agonist LY either alone or co-applied with the mglu2 PAM LY upon short-duration stimulation of the principal vibrissa and execution of the condition test protocol Aa, the top three PSTHs represent responses from a single neurone to short-duration stimulation of the principal vibrissa under control conditions, during application of the Group II agonist, and during recovery. The bottom bar graph represents the mean responses of a group of neurones (n = 44) to short-duration stimulation of the principal vibrissa under the same conditions. Ab, the top three PSTHs represent responses from a single neurone upon execution of the condition test protocol under control conditions, during application of the Group II agonist, and during recovery. The bottom bar graph represents the mean inhibitions of a group of neurones (n = 25) to the condition test protocol under the same conditions. Ba, the top three PSTHs represent responses from a single neurone to short-duration stimulation of the principal vibrissa under control conditions, upon co-application of the Group II agonist and the mglu2 PAM and during recovery. The bottom bar graph represents the mean responses of a group of neurones (n = 22) to short-duration stimulation of the principal vibrissa under control conditions, upon application of the Group II agonist alone, and upon co-application of the Group II agonist and the mglu2 PAM. Bb, the top three PSTHs represent responses from a single neurone to the condition test protocol under control conditions, during co-application of the Group II agonist and the mglu2 PAM, and during recovery. The bottom bar graph represents the mean reduction in sensory inhibition of a group of neurones (n = 14) compared to control to application of the Group II agonist alone, or co-applied with the mglu2 PAM upon execution of the condition test protocol. T: 20 ms short-duration test stimulation to the principal vibrissa; C: 20 ms conditioning stimulation to a secondary vibrissa; spike count: the total number of action potentials recorded per 2 ms bin over 6 trials. All PSTHs are constructed using responses from the same cell (CVB050b). Bar graph data are expressed as means ± SEM; P < 0.05.
7 J Physiol mglu2 positive allosteric modulation in the ventrobasal thalamus 943 The effects of the Group II agonist LY and the mglu2 PAM LY on inhibitory sensory responses On a subset of the VB neurones upon which the responses to a single short-duration vibrissa stimulus during Group II agonist and/or mglu2 PAM application had been examined, an additional sensory stimulation protocol was performed. Condition test protocols, which reveal GABAergic inputs from the TRN (Salt, 1989) (see Methods), were carried out under control conditions and in the presence of the Group II agonist and/or the mglu2 PAM.ForthispopulationofVBneuronesthedegreeof sensory inhibition observed under control conditions was 82± 2%. The properties of the afferent-evoked inhibition in these data are similar to those that have been previously presented (Salt, 1989; Salt & Eaton, 1995a;Salt&Turner, 1998). Iontophoretic ejections of the Group II agonist alone significantly reduced sensory inhibition (21 ± 3% reduction in sensory inhibition compared to control, n = 25) (Fig. 2Ab). The effect on sensory inhibition was reversed upon co-application of the Group II antagonist LY (LY354740: 22 ± 4% reduction in sensory inhibition compared to control; LY plus LY341495: 0.5± 3% reduction in sensory inhibition compared to control, n = 5) in a subpopulation of the same neurones. By contrast, iontophoretic ejections of the mglu2 PAM alone had no significant effect on the degree of sensory inhibition compared to control (1 ± 5% reduction in sensory inhibition, n = 7) (Fig. 3B). This result is of particular interest, as although the mglu2 PAM when applied alone had no effect on sensory inhibition, responses to short-duration sensory stimulation of the principal vibrissa were significantly potentiated in the same population of neurones (134 ± 14% of control, n = 7); a response profile to the short-duration stimulus which is reflected in the overall population of neurones upon which the mglu2 PAM was applied alone (Fig. 3A). On a subset of the neurones upon which the effects of the Group II agonist alone were tested, the mglu2 PAM was co-ejected with the Group II agonist and was found to significantly potentiate the agonist effect on sensory inhibition when compared to the reduction in sensory inhibition in the same neurones when the Group II agonist was applied alone (LY alone: 20 ± 4% reduction in sensory inhibition; LY plus LY487379: 30 ± 7% reduction in sensory inhibition, n = 14) (Fig. 2Bb). Similarly to before, vehicle control experiments in which iontophoretic current was passed through the 1% DMSO barrel at the same current as the mglu2 PAM application while ejecting the Group II agonist on the same neurones indicated that there was no significant effect of vehicle (LY alone: 23 ± 6% reduction in sensory inhibition; LY plus 1% DMSO: 20 ± 5% reduction in sensory inhibition, n = 7). The effects of the Group II agonist LY and the mglu2 PAM LY on lateral and recurrent inhibition This experimental protocol was performed in order to further investigate why application of the mglu2 PAM A Figure 3. VB neurone responses before, during and after iontophoretic application of the mglu2 PAM LY upon short-duration stimulation of the principal vibrissa and execution of the condition test protocol A, the top three PSTHs represent responses from a single neurone to short-duration stimulation of the principal vibrissa under control conditions, during application of the mglu2 PAM, and during recovery. The bottom bar graph represents the mean responses of a group of neurones (n = 39) to short-duration stimulation of the principal vibrissa under the same conditions. B, the top three PSTHs represent responses from a single neurone upon execution of the condition test protocol under control conditions, during application of the mglu2 PAM, and during recovery. The bottom bar graph represents the mean inhibitions of a group of neurones (n = 7) to the condition test protocol under the same conditions. T: 20 ms short-duration test stimulation to the principal vibrissa; C: 20 ms conditioning stimulation to a secondary vibrissa; spike count: the total number of action potentials recorded per 2ms bin over 6 trials. All PSTHs are constructed using responses from the same cell (CVB049c). Bar graph data are expressed as means ± SEM; P < B
8 944 C. S. Copeland and others J Physiol potentiated the VB neurone response to test stimulation at the principal vibrissa, but had no effect on the degree of sensory inhibition evoked by preceding test stimulation of the principal vibrissa with a conditioning stimulation A B of a secondary vibrissa. In this protocol, action potential firing rate of the neurone was elevated with a continuous ejection of NMDA while principal and secondary vibrissa stimuli were alternately delivered, and the degree of recurrent and lateral inhibition respectively was revealed by the reduction in NMDA-evoked neuronal firing (see Methods). Iontophoretic application of the Group II agonist significantly reduced both the lateral and recurrent sensory inhibition to a similar degree in the same group of neurones (Lateral: 45 ± 9% reduction in inhibition compared to control; Recurrent 45 ± 7% reduction in inhibition compared to control, n = 6) (Fig.4). In contrast, application of the mglu2 PAM significantly reduced recurrent inhibition (29 ± 5% reduction in inhibition compared to control, n = 6), but did not alter the level of lateral inhibition ( 3 ± 5% reduction in inhibition compared to control, n = 6) in the same group of neurones (Fig. 5). No significant effects of the Group II agonist or the mglu2 PAM on the neural firingevokedbynmdawereseen(seebelowforfurther details). Figure 4. VB neurone responses before, during and after iontophoretic application of the Group II agonist LY upon stimulation of either the principal or secondary vibrissa to evoke recurrent and lateral inhibition respectively A, the top three PSTHs represent responses from a single neurone during continuous NMDA application (see Methods) to short-duration stimulation of the principal vibrissa under control conditions, during application of the Group II agonist, and during recovery. The bottom bar graph represents the mean inhibitions of a group of neurones (n = 6) to short-duration stimulation of the principal vibrissa under the same conditions. B, the top three PSTHs represent responses from a single neurone during continuous NMDA application to short-duration stimulation of a secondary vibrissa under control conditions, during application of the Group II agonist, and during recovery. The bottom bar graph represents the mean inhibitions of a group of neurones (n = 6) to short-duration stimulation of a secondary vibrissa under the same conditions. T: 30 ms short-duration test stimulation to the principal vibrissa; C: 30 ms conditioning stimulation to a secondary vibrissa; spike count: the total number of action potentials recorded per 50 ms bin over 30 trials. All PSTHs are constructed using responses from the same cell (CVB060b). Bar graph data are expressed as means ± SEM; P < The effects of the Group II agonist LY and the mglu2 PAM LY on sensory responses to long-duration vibrissa stimulation As synaptic activation of mglu receptors has been often shown to be dependent upon repetitive stimulation of pathways in vitro in a number of brain regions (Ohishi et al. 1994; Fitzsimonds & Dichter, 1996; Wada et al. 1998; Vogt & Nicoll, 1999; Mitchell & Silver, 2000; Semyanov & Kullmann, 2000; Turner & Salt, 2000; Arnth-Jensen et al. 2002; Piet et al. 2003; Piet et al. 2004; Neale & Salt, 2006), we thought it important to investigate whether such activity-dependent activation occurs in vivo in response to physiological stimuli. This was done by examining the effects of Group II activation on neuronal responses to repetitive vibrissal stimulation using a long-duration stimulus directed at the principal vibrissa. Iontophoretic ejections of the Group II agonist alone significantly increased the responses of neurones to long-duration stimuli (154 ± 10% of control, n = 19) (Fig. 6A), as did application of the mglu2 PAM alone (135 ± 5% of control, n = 29) (Fig. 7). Co-ejection of the mglu2 PAM along with the Group II agonist significantly potentiated the agonist effect on neurone responses to long-duration stimuli when compared to responses from the same neurones when the Group II agonist was applied alone (LY alone: 170 ± 20% of control; LY plus LY487379: 246 ± 30% of control, n = 8) (Fig. 6B). As in previous protocols, passing iontophoretic current through the 1% DMSO barrel did not significantly alter responses of neurones when applied alone (100 ± 2% of
9 J Physiol mglu2 positive allosteric modulation in the ventrobasal thalamus 945 control, n = 10), nor did it further potentiate the responses of neurones when co-applied with the Group II agonist (LY alone: 159 ± 20% of control; LY plus 1% DMSO: 159 ± 21% of control, n = 6). A B The effects of the Group II antagonist LY on sensory responses to short- and long-duration vibrissa stimulation As the mglu2 PAM was able to potentiate neuronal responses to both the short- and long-duration A B Figure 5. VB neurone responses before, during and after iontophoretic application of the mglu2 PAM LY upon stimulation of either the principal or secondary vibrissa to evoke recurrent and lateral inhibition respectively A, the top three PSTHs represent responses from a single neurone during continuous NMDA application (see Methods) to short-duration stimulation of the principal vibrissa under control conditions, during application of the mglu2 PAM, and during recovery. The bottom bar graph represents the mean inhibitions of a group of neurones (n = 6) to short-duration stimulation of the principal vibrissa under the same conditions. B, the top three PSTHs represent responses from a single neurone during continuous NMDA application to short-duration stimulation of a secondary vibrissa under control conditions, during application of the mglu2 PAM, and during recovery. The bottom bar graph represents the mean inhibitions of a group of neurones (n = 6) to short-duration stimulation of a secondary vibrissa under the same conditions. T: 30 ms short-duration test stimulation to the principal vibrissa; C: 30 ms conditioning stimulation to a secondary vibrissa; spike count: the total number of action potentials recorded per 50 ms bin over 30 trials. All PSTHs are constructed using responses from the same cell (CVB060b). Bar graph data are expressed as means ± SEM; P < Figure 6. VB neurone responses before, during and after iontophoretic application of the Group II agonist LY either alone or co-applied with the mglu2 PAM LY upon long-duration stimulation of the principal vibrissa A, the top three PSTHs represent responses from a single neurone to long-duration stimulation of the principal vibrissa under control conditions, during application of the Group II agonist, and during recovery. The bottom bar graph represents the mean responses of a group of neurones (n = 21) to long-duration stimulation of the principal vibrissa under the same conditions. B, the top three PSTHs represent responses from a single neurone to long-duration stimulation of the principal vibrissa under control conditions, upon co-application of the Group II agonist and the mglu2 PAM, and during recovery. The bottom bar graph represents the mean responses of a group of neurones (n = 8) to long-duration stimulation of the principal vibrissa under control conditions, upon application of the Group II agonist alone and upon co-application of the Group II agonist and the mglu2 PAM. T: 1000 ms long-duration test stimulation to the principal vibrissa; spike count: the total number of action potentials recorded per 200 ms bin over 18 trials. All PSTHs are constructed using responses from the same cell (CVB038d). Bar graph data are expressed as means ± SEM; P < 0.05.
10 946 C. S. Copeland and others J Physiol stimulation of the principal vibrissa, the Group II antagonist LY was applied as an additional means to reveal the release of endogenous agonist during these sensory stimulation protocols. Iontophoretic ejections of the Group II antagonist significantly reduced the responses of neurones to both short- (91 ± 4% compared to control, n = 12) and long- (86 ± 4% compared to control, n = 7) duration stimuli of the principal vibrissa (Fig. 8). Furthermore, on a subset of the neurones upon which the effects of the mglu2 PAM alone were tested, the Group II antagonist was co-applied with the mglu2 PAM and was found to reverse the mglu2 PAM effect on neuronal responses to short- (LY alone: 127 ± 7% of control; LY plus LY341495: 104 ± 4% of control, n = 7) and long- (LY alone: 136 ± 6% of control; LY plus LY341495: 102 ± 9% of control, n = 7) duration stimuli. This verifies that the mglu2 PAM is exerting its effects via Group II receptors. A B Figure 7. VB neurone responses before, during and after iontophoretic application of the mglu2 PAM LY upon long-duration stimulation of the principal vibrissa The top three PSTHs represent responses from a single neurone to long-duration stimulation of the principal vibrissa under control conditions, during application of the mglu2 PAM, and during recovery. The bottom bar graph represents the mean responses of a group of neurones (n = 22) to long-duration stimulation of the principal vibrissa under the same conditions. T: 1000 ms long-duration test stimulation to the principal vibrissa; spike count: the total number of action potentials recorded per 100 ms bin over 18 trials. All PSTHs are constructed using responses from the same cell (CVB043a). Bar graph data are expressed as means ± SEM; P < Figure 8. VB neurone responses before, during and after iontophoretic application of the Group II antagonist LY upon short- and long-duration stimulation of the principal vibrissa A, the top three PSTHs represent responses from a single neurone to short-duration stimulation of the principal vibrissa under control conditions, during application of the Group II antagonist, and during recovery. The bottom bar graph represents the mean responses of a group of neurones (n = 12) to short-duration stimulation of the principal vibrissa under the same conditions. B, the top three PSTHs represent responses from a single neurone upon to long-duration stimulation of the principal vibrissa under control conditions, during application of the Group II antagonist, and during recovery. The bottom bar graph represents the mean inhibitions of a group of neurones (n = 7) to the condition test protocol under the same conditions. T 1 : 30 ms short-duration test stimulation to the principal vibrissa; T 2 : 500 ms long-duration test stimulation to the principal vibrissa; spike count: the total number of action potentials recorded per bin (20 ms bins in A, 100 ms bins in B) over 6 trials. All PSTHs are constructed using responses from the same cell (CVB085c). Bar graph data are expressed as means ± SEM; P < 0.05.
11 J Physiol mglu2 positive allosteric modulation in the ventrobasal thalamus 947 The effects of the Group II agonist LY and the mglu2 PAM LY on ionotropic glutamate receptor agonist responses As it is known that VB neurone responses to vibrissal stimulation are mediated via AMPA and NMDA receptors (Salt, 1986; 1987), a final experimental protocol was performed in order to investigate whether the Group II agonist and the mglu2 PAM could exert their effects on VB neurone responses via a postsynaptic interaction with NMDA and/or AMPA receptors. AMPA and NMDA were applied iontophoretically and were found to excite VB neurones in a manner concordant with that of previous work from this and other laboratories (Salt & Eaton, 1996; Binns et al. 2003). Iontophoretic ejections of the Group II agonist did not significantly alter the responses of neurones to agonist ejections (AMPA: 108 ± 7% of control; NMDA: 105 ± 7% of control, n = 23); nor did iontophoretic ejections of the mglu2 PAM (AMPA: 102 ± 8% of control; NMDA: 102 ± 6% of control, n = 13). Discussion It has been well established that the Group II mglu receptors can modulate somatosensory transmission in the rat VB (Salt & Eaton, 1995a, b; Saltet al. 1996; Salt & Turner, 1998; Turner & Salt, 2003). However, the relative contributions of the Group II mglu receptor subtypes in modulating sensory transmission are not known, nor to what extent and under what conditions these receptors may be activated upon physiological sensory stimulation. The data obtained in this study using in vivo electrophysiology and iontophoresis in the rat VB clearly demonstrate an mglu2 component to the observed Group II effect on sensory inhibition. Additionally, we have shown that mglu2 is likely to be activated during sensory afferent transmission to VB, which leads to a reduction in inhibition. We speculate this could be occurring via glutamate spillover from the sensory afferent synapse upon physiological sensory stimulation. The rat VB is well known to contain only relay neurones of a relatively homogeneous morphology (Peschanski et al. 1984; Harris, 1986; Ohara & Havton, 1994), but it is possible to categorise neurones functionally into SWE or MWE neurones (Ito, 1988; Brecht & Sakmann, 2002). In this study we recorded from both SWE (74% of cells) and MWE (26% of cells) neurones and did not find any differences in our results between these cell types. This suggests that our findings and interpretation are pertinent across different functional categories of thalamic relay neurones. LY354740, the best-studied selective Group II orthosteric agonist (Schoepp et al. 2003), has been extensively used to probe Group II mglu receptor function in behavioural (Schoeppet al. 2003; Nordquist et al. 2008) and physiological (Flor et al. 2002; Moldrich et al. 2003) responses in both the human and rodent CNS in vivo and in vitro. LY487379, a highly selective mglu2 PAM, which possesses no intrinsic agonist activity but does enhance responses to submaximal glutamate without activity at other receptors or ion channels (Johnson et al. 2003), has been used in behavioural and in vitro electrophysiological studies in the rodent CNS (Schaffhauser et al. 2003; Galici et al. 2005; Poisik et al. 2005; Harich et al. 2007; Hermes & Renaud, 2010; Nikiforuk et al. 2010). The orthosteric antagonist LY has a relatively high selectivity with a nanomolar potency for the Group II mglu receptors, and with submicromolar potencies at all other mglu receptor subtypes (Kingston et al. 1998; Schoepp et al. 1999). However, the iontophoretic parameters used for LY in this study have been demonstrated by ourselves to produce selective antagonism for the Group II mglu receptors (Cirone et al. 2002). The pharmacological specificity of our drug applications is clearly crucial to the interpretation of the results of the present study. Our finding that the Group II antagonist LY reverses the effects of both the Group II agonist and the mglu2 PAM indicates that we are applying the agents in pharmacologically appropriate quantities. Furthermore, application of either the Group II agonist or mglu2 PAM had no effect on responses evoked by NMDA or AMPA, indicating that non-specific effects are not being produced by our drug application protocols. Group II mglu receptor activation facilitates responses to sensory stimulation of the principal vibrissa A potentiation of VB neurone responses to sensory stimulation of the principal vibrissa was observed upon application of the Group II agonist LY VB neurone responses to sensory stimulation are mediated by NMDA and AMPA receptors (Salt, 1986, 1987; Salt & Eaton, 1989), but excitatory responses to these ionotropic agonists were not perturbed upon LY application. Furthermore, in vitro experiments show that LY can directly reduce TRN-evoked inhibitory postsynaptic potentials without affecting postsynaptic membrane properties of VB neurones (Turner & Salt, 2003). Thus the Group II agonist is likely to be potentiating sensory responses by reducing recurrent inhibition, therefore enabling neurones to elicit enhanced responses. Indeed, use of the same sensory stimulation protocol revealed a similar effect on VB neurone responses when GABAergic transmission originating from the TRN was reduced by the GABA antagonist bicuculline (Salt, 1989). Co-application of the mglu2 PAM further potentiated the Group II agonist effect, indicating an mglu2 component to this effect. Interestingly, the percentage increase in VB neurone response to Group II agonist application either alone or in conjunction
12 948 C. S. Copeland and others J Physiol with the mglu2 PAM was significantly greater for the long-duration stimulus compared to the short-duration stimulus. The long-duration stimulus not only provides the VB neurone with a longer duration of excitatory input than the short-duration stimulus, but also evokes a longer recurrent inhibition. When this is reduced by Group II agonist application, either alone or in conjunction with themglu2pam,thevbneuroneisabletorespondtoa greater extent to the longer duration excitatory drive. A particularly novel and important finding of our study is that application of the mglu2 PAM alone potentiated excitatory VB neurone responses to sensory stimulation, an effect that was reversed upon co-application of the Group II antagonist. Additionally, application of the Group II antagonist alone reduced neuronal responses to sensory stimulation. This indicates that the Group II receptors are normally activated in vivo by endogenous ligand upon stimulation of principal vibrissae, and that thereisanmglu2componenttothiseffect.howthismay occur and its physiological relevance are considered in detail below. Group II mglu receptor activation reduces inhibition in VB evoked by sensory stimulation of the secondary vibrissa A reduction in VB neurone inhibition evoked by stimulation of a secondary vibrissa shortly before stimulation of the principal vibrissa was observed upon application of the Group II agonist LY This finding is consistent with previous in vivo studies that have used alternative Group II agonists (e.g. (1S,3R)-ACPD, L-CCG-l, (2R,4R)-APDC) (Salt & Eaton, 1995a, b; Salt et al. 1996; Salt & Turner, 1998). In our present study, the mglu2 PAM potentiated the agonist effect on sensory inhibition, indicating an mglu2 component. mglu2 receptors may be activated by glutamate spillover from the sensory afferent synapse As application of the mglu2 PAM potentiated VB neurone responses to principal vibrissa stimulation, this indicates that mglu2 receptors are normally activated in vivo upon physiological sensory stimulation. However, when stimulation of the principal vibrissa was preceded by stimulation of a secondary vibrissa, application of the PAM alone did not alter the level of inhibition in VB. Although puzzling at first, this outcome provides a possible explanation as to how mglu2 receptors are activated in vivo. As the PAM possesses no intrinsic activity (Johnson et al. 2003), it is possible to speculate that it is potentiating the effects of endogenously released glutamate, which could lead to an increase in response to principal vibrissa stimulation. The source of this glutamate may be from excitatory afferent terminals, which could be sensory or cortical in origin. Ultrastructural studies indicate that there are no axo-axonic contacts onto GABAergic terminals, and cortical afferent terminals are not closely associated with TRN terminals in the rat VB (Ohara & Lieberman, 1993). Therefore, transmission from and receptors associated with cortico-thalamic afferent terminals are unlikely to be involved in this mechanism. However, ultrastructural studies do indicate that sensory afferent terminals are closely located to TRN afferent GABAergic terminals and glial processes on the soma or proximal dendrites of neurones in the rat VB (Ralston, 1983; Ohara & Lieberman, 1993). Glutamate may therefore be spilling over from the sensory afferent synapse to activate extrasynaptic mglu2 receptors leading to a reduction in inhibition with consequent facilitation of responses to sensory stimuli. Similar glutamate spillover has been shown to activate Group II mglu receptors in vitro (Alexander & Godwin, 2005; Chen & Bonham, 2005; Linden et al. 2005; Alexander & Godwin, 2006), making it appropriate to speculate that mglu2 receptors in VB may be activated via this mechanism in vivo. It has been demonstrated that Group II mglu receptors can be localised on glial processes surrounding GABAergic terminals (Liu et al. 1998; Mineff & Valtschanoff, 1999), and mglu3 can be found on TRN-originating GABAergic axons (Tamaru et al. 2001) in the rat VB. Group II mglu receptors have been demonstrated to regulate non-vesicular release of glutamate from glial cells via the cysteine-glutamate transporter in other brain regions (Xi et al. 2002; Moran et al. 2003; Baker et al. 2008). Such locations are therefore ideally suited for these receptors to modulate GABAergic transmission due to their close association with sensory afferent terminals on the soma and proximal dendrites of VB neurones (Ralston, 1983; Ohara & Lieberman, 1993). However, in the alternative stimulation protocol when principal vibrissa stimulation is preceded by stimulation of a secondary vibrissa, the above mechanism could not take place. In this scenario, inhibition in VB via the TRN is provided laterally upon stimulation of the secondary vibrissa (Pinault & Deschenes, 1998; Crabtree, 1999). This postsynaptic inhibition precedes the principal vibrissa excitation; therefore the subsequent release of glutamate from the sensory afferent terminal would be unable to reduce the GABAergic transmission from the TRN by acting at mglu2 receptors, as inhibition of the VB neurone has already taken place. An additional stimulation protocol was devised to further investigate the above hypothesis that mglu2 receptors may be activated by glutamate spillover from the sensory afferent synapse upon sensory stimulation. If such a mechanism does occur, then application of the mglu2 PAM should only influence inhibition if there is direct driving of the VB neurone by its principal vibrissa
Thalamo-Cortical Relationships Ultrastructure of Thalamic Synaptic Glomerulus
 Central Visual Pathways V1/2 NEUR 3001 dvanced Visual Neuroscience The Lateral Geniculate Nucleus () is more than a relay station LP SC Professor Tom Salt UCL Institute of Ophthalmology Retina t.salt@ucl.ac.uk
Central Visual Pathways V1/2 NEUR 3001 dvanced Visual Neuroscience The Lateral Geniculate Nucleus () is more than a relay station LP SC Professor Tom Salt UCL Institute of Ophthalmology Retina t.salt@ucl.ac.uk
Astrocytes modulate thalamic sensory processing via mglu2 receptor activation
 1 2 3 4 5 6 7 8 9 Astrocytes modulate thalamic sensory processing via mglu2 receptor activation To appear in: Neuropharmacology Received Date: 4 August 2016 Revised Date: 27 March 2017 Accepted Date: 13
1 2 3 4 5 6 7 8 9 Astrocytes modulate thalamic sensory processing via mglu2 receptor activation To appear in: Neuropharmacology Received Date: 4 August 2016 Revised Date: 27 March 2017 Accepted Date: 13
Embryological origin of thalamus
 diencephalon Embryological origin of thalamus The diencephalon gives rise to the: Thalamus Epithalamus (pineal gland, habenula, paraventricular n.) Hypothalamus Subthalamus (Subthalamic nuclei) The Thalamus:
diencephalon Embryological origin of thalamus The diencephalon gives rise to the: Thalamus Epithalamus (pineal gland, habenula, paraventricular n.) Hypothalamus Subthalamus (Subthalamic nuclei) The Thalamus:
SUPPLEMENTARY INFORMATION. Supplementary Figure 1
 SUPPLEMENTARY INFORMATION Supplementary Figure 1 The supralinear events evoked in CA3 pyramidal cells fulfill the criteria for NMDA spikes, exhibiting a threshold, sensitivity to NMDAR blockade, and all-or-none
SUPPLEMENTARY INFORMATION Supplementary Figure 1 The supralinear events evoked in CA3 pyramidal cells fulfill the criteria for NMDA spikes, exhibiting a threshold, sensitivity to NMDAR blockade, and all-or-none
TNS Journal Club: Interneurons of the Hippocampus, Freund and Buzsaki
 TNS Journal Club: Interneurons of the Hippocampus, Freund and Buzsaki Rich Turner (turner@gatsby.ucl.ac.uk) Gatsby Unit, 22/04/2005 Rich T. Introduction Interneuron def = GABAergic non-principal cell Usually
TNS Journal Club: Interneurons of the Hippocampus, Freund and Buzsaki Rich Turner (turner@gatsby.ucl.ac.uk) Gatsby Unit, 22/04/2005 Rich T. Introduction Interneuron def = GABAergic non-principal cell Usually
Lecture 22: A little Neurobiology
 BIO 5099: Molecular Biology for Computer Scientists (et al) Lecture 22: A little Neurobiology http://compbio.uchsc.edu/hunter/bio5099 Larry.Hunter@uchsc.edu Nervous system development Part of the ectoderm
BIO 5099: Molecular Biology for Computer Scientists (et al) Lecture 22: A little Neurobiology http://compbio.uchsc.edu/hunter/bio5099 Larry.Hunter@uchsc.edu Nervous system development Part of the ectoderm
STRUCTURAL ELEMENTS OF THE NERVOUS SYSTEM
 STRUCTURAL ELEMENTS OF THE NERVOUS SYSTEM STRUCTURE AND MAINTENANCE OF NEURONS (a) (b) Dendrites Cell body Initial segment collateral terminals (a) Diagrammatic representation of a neuron. The break in
STRUCTURAL ELEMENTS OF THE NERVOUS SYSTEM STRUCTURE AND MAINTENANCE OF NEURONS (a) (b) Dendrites Cell body Initial segment collateral terminals (a) Diagrammatic representation of a neuron. The break in
Neuropharmacology 92 (2015) 16e24. Contents lists available at ScienceDirect. Neuropharmacology. journal homepage:
 Neuropharmacology 92 (2015) 16e24 Contents lists available at ScienceDirect Neuropharmacology journal homepage: www.elsevier.com/locate/neuropharm Neuronal activity patterns in the mediodorsal thalamus
Neuropharmacology 92 (2015) 16e24 Contents lists available at ScienceDirect Neuropharmacology journal homepage: www.elsevier.com/locate/neuropharm Neuronal activity patterns in the mediodorsal thalamus
COGNITIVE SCIENCE 107A. Sensory Physiology and the Thalamus. Jaime A. Pineda, Ph.D.
 COGNITIVE SCIENCE 107A Sensory Physiology and the Thalamus Jaime A. Pineda, Ph.D. Sensory Physiology Energies (light, sound, sensation, smell, taste) Pre neural apparatus (collects, filters, amplifies)
COGNITIVE SCIENCE 107A Sensory Physiology and the Thalamus Jaime A. Pineda, Ph.D. Sensory Physiology Energies (light, sound, sensation, smell, taste) Pre neural apparatus (collects, filters, amplifies)
CYTOARCHITECTURE OF CEREBRAL CORTEX
 BASICS OF NEUROBIOLOGY CYTOARCHITECTURE OF CEREBRAL CORTEX ZSOLT LIPOSITS 1 CELLULAR COMPOSITION OF THE CEREBRAL CORTEX THE CEREBRAL CORTEX CONSISTS OF THE ARCHICORTEX (HIPPOCAMPAL FORMA- TION), PALEOCORTEX
BASICS OF NEUROBIOLOGY CYTOARCHITECTURE OF CEREBRAL CORTEX ZSOLT LIPOSITS 1 CELLULAR COMPOSITION OF THE CEREBRAL CORTEX THE CEREBRAL CORTEX CONSISTS OF THE ARCHICORTEX (HIPPOCAMPAL FORMA- TION), PALEOCORTEX
Supplementary Figure 1. Basic properties of compound EPSPs at
 Supplementary Figure 1. Basic properties of compound EPSPs at hippocampal CA3 CA3 cell synapses. (a) EPSPs were evoked by extracellular stimulation of the recurrent collaterals and pharmacologically isolated
Supplementary Figure 1. Basic properties of compound EPSPs at hippocampal CA3 CA3 cell synapses. (a) EPSPs were evoked by extracellular stimulation of the recurrent collaterals and pharmacologically isolated
QUIZ/TEST REVIEW NOTES SECTION 7 NEUROPHYSIOLOGY [THE SYNAPSE AND PHARMACOLOGY]
![QUIZ/TEST REVIEW NOTES SECTION 7 NEUROPHYSIOLOGY [THE SYNAPSE AND PHARMACOLOGY] QUIZ/TEST REVIEW NOTES SECTION 7 NEUROPHYSIOLOGY [THE SYNAPSE AND PHARMACOLOGY]](/thumbs/86/94924826.jpg) QUIZ/TEST REVIEW NOTES SECTION 7 NEUROPHYSIOLOGY [THE SYNAPSE AND PHARMACOLOGY] Learning Objectives: Explain how neurons communicate stimulus intensity Explain how action potentials are conducted along
QUIZ/TEST REVIEW NOTES SECTION 7 NEUROPHYSIOLOGY [THE SYNAPSE AND PHARMACOLOGY] Learning Objectives: Explain how neurons communicate stimulus intensity Explain how action potentials are conducted along
1) Drop off in the Bi 150 box outside Baxter 331 or to the head TA (jcolas).
 Bi/CNS/NB 150 Problem Set 3 Due: Tuesday, Oct. 27, at 4:30 pm Instructions: 1) Drop off in the Bi 150 box outside Baxter 331 or e-mail to the head TA (jcolas). 2) Submit with this cover page. 3) Use a
Bi/CNS/NB 150 Problem Set 3 Due: Tuesday, Oct. 27, at 4:30 pm Instructions: 1) Drop off in the Bi 150 box outside Baxter 331 or e-mail to the head TA (jcolas). 2) Submit with this cover page. 3) Use a
Relative contributions of cortical and thalamic feedforward inputs to V2
 Relative contributions of cortical and thalamic feedforward inputs to V2 1 2 3 4 5 Rachel M. Cassidy Neuroscience Graduate Program University of California, San Diego La Jolla, CA 92093 rcassidy@ucsd.edu
Relative contributions of cortical and thalamic feedforward inputs to V2 1 2 3 4 5 Rachel M. Cassidy Neuroscience Graduate Program University of California, San Diego La Jolla, CA 92093 rcassidy@ucsd.edu
To Accompany: Thalamic Synchrony and the Adaptive Gating of Information Flow to Cortex Wang, Webber, & Stanley
 SUPPLEMENTARY MATERIAL To Accompany: Thalamic Synchrony and the Adaptive Gating of Information Flow to Cortex Wang, Webber, & Stanley Supplementary Note 1: Parametric fits of spike count distributions
SUPPLEMENTARY MATERIAL To Accompany: Thalamic Synchrony and the Adaptive Gating of Information Flow to Cortex Wang, Webber, & Stanley Supplementary Note 1: Parametric fits of spike count distributions
When cells are already maximally potentiated LTP is occluded.
 When cells are already maximally potentiated LTP is occluded. Stein, V et al., (2003) J Neurosci, 23:5503-6606. Also found in Rat Barrel Cortex Ehrlich & Malinow (2004) J. Neurosci. 24:916-927 Over-expression
When cells are already maximally potentiated LTP is occluded. Stein, V et al., (2003) J Neurosci, 23:5503-6606. Also found in Rat Barrel Cortex Ehrlich & Malinow (2004) J. Neurosci. 24:916-927 Over-expression
Plasticity of Cerebral Cortex in Development
 Plasticity of Cerebral Cortex in Development Jessica R. Newton and Mriganka Sur Department of Brain & Cognitive Sciences Picower Center for Learning & Memory Massachusetts Institute of Technology Cambridge,
Plasticity of Cerebral Cortex in Development Jessica R. Newton and Mriganka Sur Department of Brain & Cognitive Sciences Picower Center for Learning & Memory Massachusetts Institute of Technology Cambridge,
The Cerebellum. Outline. Lu Chen, Ph.D. MCB, UC Berkeley. Overview Structure Micro-circuitry of the cerebellum The cerebellum and motor learning
 The Cerebellum Lu Chen, Ph.D. MCB, UC Berkeley 1 Outline Overview Structure Micro-circuitry of the cerebellum The cerebellum and motor learning 2 Overview Little brain 10% of the total volume of the brain,
The Cerebellum Lu Chen, Ph.D. MCB, UC Berkeley 1 Outline Overview Structure Micro-circuitry of the cerebellum The cerebellum and motor learning 2 Overview Little brain 10% of the total volume of the brain,
Neural Basis of Motor Control
 Neural Basis of Motor Control Central Nervous System Skeletal muscles are controlled by the CNS which consists of the brain and spinal cord. Determines which muscles will contract When How fast To what
Neural Basis of Motor Control Central Nervous System Skeletal muscles are controlled by the CNS which consists of the brain and spinal cord. Determines which muscles will contract When How fast To what
Applied Neuroscience. Conclusion of Science Honors Program Spring 2017
 Applied Neuroscience Conclusion of Science Honors Program Spring 2017 Review Circle whichever is greater, A or B. If A = B, circle both: I. A. permeability of a neuronal membrane to Na + during the rise
Applied Neuroscience Conclusion of Science Honors Program Spring 2017 Review Circle whichever is greater, A or B. If A = B, circle both: I. A. permeability of a neuronal membrane to Na + during the rise
File name: Supplementary Information Description: Supplementary Figures, Supplementary Table and Supplementary References
 File name: Supplementary Information Description: Supplementary Figures, Supplementary Table and Supplementary References File name: Supplementary Data 1 Description: Summary datasheets showing the spatial
File name: Supplementary Information Description: Supplementary Figures, Supplementary Table and Supplementary References File name: Supplementary Data 1 Description: Summary datasheets showing the spatial
LESSON 3.3 WORKBOOK. Why does applying pressure relieve pain?
 Postsynaptic potentials small changes in voltage (membrane potential) due to the binding of neurotransmitter. Receptor-gated ion channels ion channels that open or close in response to the binding of a
Postsynaptic potentials small changes in voltage (membrane potential) due to the binding of neurotransmitter. Receptor-gated ion channels ion channels that open or close in response to the binding of a
Supporting information
 Supporting information Buckley CL, Toyoizumi T (2018) A theory of how active behavior stabilises neural activity: Neural gain modulation by closed-loop environmental feedback. PLoS Comput Biol 14(1): e1005926.
Supporting information Buckley CL, Toyoizumi T (2018) A theory of how active behavior stabilises neural activity: Neural gain modulation by closed-loop environmental feedback. PLoS Comput Biol 14(1): e1005926.
Communication within a Neuron
 Neuronal Communication, Ph.D. Communication within a Neuron Measuring Electrical Potentials of Axons The Membrane Potential The Action Potential Conduction of the Action Potential 1 The withdrawal reflex
Neuronal Communication, Ph.D. Communication within a Neuron Measuring Electrical Potentials of Axons The Membrane Potential The Action Potential Conduction of the Action Potential 1 The withdrawal reflex
Functioning of Circuits Connecting Thalamus and Cortex
 Functioning of Circuits Connecting Thalamus and Cortex S. Murray Sherman *1 ABSTRACT Glutamatergic pathways in thalamus and cortex are divided into two distinct classes: driver, which carries the main
Functioning of Circuits Connecting Thalamus and Cortex S. Murray Sherman *1 ABSTRACT Glutamatergic pathways in thalamus and cortex are divided into two distinct classes: driver, which carries the main
MOLECULAR AND CELLULAR NEUROSCIENCE
 MOLECULAR AND CELLULAR NEUROSCIENCE BMP-218 November 4, 2014 DIVISIONS OF THE NERVOUS SYSTEM The nervous system is composed of two primary divisions: 1. CNS - Central Nervous System (Brain + Spinal Cord)
MOLECULAR AND CELLULAR NEUROSCIENCE BMP-218 November 4, 2014 DIVISIONS OF THE NERVOUS SYSTEM The nervous system is composed of two primary divisions: 1. CNS - Central Nervous System (Brain + Spinal Cord)
MOLECULAR BIOLOGY OF DRUG ADDICTION. Sylvane Desrivières, SGDP Centre
 1 MOLECULAR BIOLOGY OF DRUG ADDICTION Sylvane Desrivières, SGDP Centre Reward 2 Humans, as well as other organisms engage in behaviours that are rewarding The pleasurable feelings provide positive reinforcement
1 MOLECULAR BIOLOGY OF DRUG ADDICTION Sylvane Desrivières, SGDP Centre Reward 2 Humans, as well as other organisms engage in behaviours that are rewarding The pleasurable feelings provide positive reinforcement
Cholinergic Activation of M2 Receptors Leads to Context- Dependent Modulation of Feedforward Inhibition in the Visual Thalamus
 Cholinergic Activation of M2 Receptors Leads to Context- Dependent Modulation of Feedforward Inhibition in the Visual Thalamus Miklos Antal., Claudio Acuna-Goycolea., R. Todd Pressler, Dawn M. Blitz, Wade
Cholinergic Activation of M2 Receptors Leads to Context- Dependent Modulation of Feedforward Inhibition in the Visual Thalamus Miklos Antal., Claudio Acuna-Goycolea., R. Todd Pressler, Dawn M. Blitz, Wade
Lisa M. Giocomo & Michael E. Hasselmo
 Mol Neurobiol (2007) 36:184 200 DOI 10.1007/s12035-007-0032-z Neuromodulation by Glutamate and Acetylcholine can Change Circuit Dynamics by Regulating the Relative Influence of Afferent Input and Excitatory
Mol Neurobiol (2007) 36:184 200 DOI 10.1007/s12035-007-0032-z Neuromodulation by Glutamate and Acetylcholine can Change Circuit Dynamics by Regulating the Relative Influence of Afferent Input and Excitatory
Memory Systems II How Stored: Engram and LTP. Reading: BCP Chapter 25
 Memory Systems II How Stored: Engram and LTP Reading: BCP Chapter 25 Memory Systems Learning is the acquisition of new knowledge or skills. Memory is the retention of learned information. Many different
Memory Systems II How Stored: Engram and LTP Reading: BCP Chapter 25 Memory Systems Learning is the acquisition of new knowledge or skills. Memory is the retention of learned information. Many different
Chapter 11 Introduction to the Nervous System and Nervous Tissue Chapter Outline
 Chapter 11 Introduction to the Nervous System and Nervous Tissue Chapter Outline Module 11.1 Overview of the Nervous System (Figures 11.1-11.3) A. The nervous system controls our perception and experience
Chapter 11 Introduction to the Nervous System and Nervous Tissue Chapter Outline Module 11.1 Overview of the Nervous System (Figures 11.1-11.3) A. The nervous system controls our perception and experience
Astrocyte signaling controls spike timing-dependent depression at neocortical synapses
 Supplementary Information Astrocyte signaling controls spike timing-dependent depression at neocortical synapses Rogier Min and Thomas Nevian Department of Physiology, University of Berne, Bern, Switzerland
Supplementary Information Astrocyte signaling controls spike timing-dependent depression at neocortical synapses Rogier Min and Thomas Nevian Department of Physiology, University of Berne, Bern, Switzerland
Introduction to Computational Neuroscience
 Introduction to Computational Neuroscience Lecture 7: Network models Lesson Title 1 Introduction 2 Structure and Function of the NS 3 Windows to the Brain 4 Data analysis 5 Data analysis II 6 Single neuron
Introduction to Computational Neuroscience Lecture 7: Network models Lesson Title 1 Introduction 2 Structure and Function of the NS 3 Windows to the Brain 4 Data analysis 5 Data analysis II 6 Single neuron
All questions below pertain to mandatory material: all slides, and mandatory homework (if any).
 ECOL 182 Spring 2008 Dr. Ferriere s lectures Lecture 6: Nervous system and brain Quiz Book reference: LIFE-The Science of Biology, 8 th Edition. http://bcs.whfreeman.com/thelifewire8e/ All questions below
ECOL 182 Spring 2008 Dr. Ferriere s lectures Lecture 6: Nervous system and brain Quiz Book reference: LIFE-The Science of Biology, 8 th Edition. http://bcs.whfreeman.com/thelifewire8e/ All questions below
Supplementary figure 1: LII/III GIN-cells show morphological characteristics of MC
 1 2 1 3 Supplementary figure 1: LII/III GIN-cells show morphological characteristics of MC 4 5 6 7 (a) Reconstructions of LII/III GIN-cells with somato-dendritic compartments in orange and axonal arborizations
1 2 1 3 Supplementary figure 1: LII/III GIN-cells show morphological characteristics of MC 4 5 6 7 (a) Reconstructions of LII/III GIN-cells with somato-dendritic compartments in orange and axonal arborizations
Part 11: Mechanisms of Learning
 Neurophysiology and Information: Theory of Brain Function Christopher Fiorillo BiS 527, Spring 2012 042 350 4326, fiorillo@kaist.ac.kr Part 11: Mechanisms of Learning Reading: Bear, Connors, and Paradiso,
Neurophysiology and Information: Theory of Brain Function Christopher Fiorillo BiS 527, Spring 2012 042 350 4326, fiorillo@kaist.ac.kr Part 11: Mechanisms of Learning Reading: Bear, Connors, and Paradiso,
Chapter 7. The Nervous System: Structure and Control of Movement
 Chapter 7 The Nervous System: Structure and Control of Movement Objectives Discuss the general organization of the nervous system Describe the structure & function of a nerve Draw and label the pathways
Chapter 7 The Nervous System: Structure and Control of Movement Objectives Discuss the general organization of the nervous system Describe the structure & function of a nerve Draw and label the pathways
Physiology of synapses and receptors
 Physiology of synapses and receptors Dr Syed Shahid Habib Professor & Consultant Clinical Neurophysiology Dept. of Physiology College of Medicine & KKUH King Saud University REMEMBER These handouts will
Physiology of synapses and receptors Dr Syed Shahid Habib Professor & Consultant Clinical Neurophysiology Dept. of Physiology College of Medicine & KKUH King Saud University REMEMBER These handouts will
Tonic and burst firing: dual modes of thalamocortical relay
 1 Review Vol. No. February 1 Tonic and burst firing: dual modes of thalamocortical relay S. Murray Sherman All thalamic relay cells exhibit two distinct response modes tonic and burst that reflect the
1 Review Vol. No. February 1 Tonic and burst firing: dual modes of thalamocortical relay S. Murray Sherman All thalamic relay cells exhibit two distinct response modes tonic and burst that reflect the
Cellular Neurobiology / BIPN 140
 SECOND MIDTERM EXAMINATION Fall, 2015 GENERAL INSTRUCTIONS 1. Please write your name on ALL 6 pages. 2. Please answer each question IN THE SPACE ALLOTTED. 1) /10 pts 2) /10 pts 3) /15 pts 4) /15 pts 5)
SECOND MIDTERM EXAMINATION Fall, 2015 GENERAL INSTRUCTIONS 1. Please write your name on ALL 6 pages. 2. Please answer each question IN THE SPACE ALLOTTED. 1) /10 pts 2) /10 pts 3) /15 pts 4) /15 pts 5)
Chapter 7. Objectives
 Chapter 7 The Nervous System: Structure and Control of Movement Objectives Discuss the general organization of the nervous system Describe the structure & function of a nerve Draw and label the pathways
Chapter 7 The Nervous System: Structure and Control of Movement Objectives Discuss the general organization of the nervous system Describe the structure & function of a nerve Draw and label the pathways
Response Transformation and Receptive-Field Synthesis in the Lemniscal Trigeminothalamic Circuit
 J Neurophysiol 90: 1556 1570, 2003. First published April 30, 2003; 10.1152/jn.00111.2003. Response Transformation and Receptive-Field Synthesis in the Lemniscal Trigeminothalamic Circuit Brandon S. Minnery,
J Neurophysiol 90: 1556 1570, 2003. First published April 30, 2003; 10.1152/jn.00111.2003. Response Transformation and Receptive-Field Synthesis in the Lemniscal Trigeminothalamic Circuit Brandon S. Minnery,
LESSON 3.3 WORKBOOK. Why does applying pressure relieve pain? Workbook. Postsynaptic potentials
 Depolarize to decrease the resting membrane potential. Decreasing membrane potential means that the membrane potential is becoming more positive. Excitatory postsynaptic potentials (EPSP) graded postsynaptic
Depolarize to decrease the resting membrane potential. Decreasing membrane potential means that the membrane potential is becoming more positive. Excitatory postsynaptic potentials (EPSP) graded postsynaptic
Modeling Depolarization Induced Suppression of Inhibition in Pyramidal Neurons
 Modeling Depolarization Induced Suppression of Inhibition in Pyramidal Neurons Peter Osseward, Uri Magaram Department of Neuroscience University of California, San Diego La Jolla, CA 92092 possewar@ucsd.edu
Modeling Depolarization Induced Suppression of Inhibition in Pyramidal Neurons Peter Osseward, Uri Magaram Department of Neuroscience University of California, San Diego La Jolla, CA 92092 possewar@ucsd.edu
Activity-Dependent Regulation of Retinogeniculate Signaling by Metabotropic Glutamate Receptors
 12820 The Journal of Neuroscience, September 12, 2012 32(37):12820 12831 Behavioral/Systems/Cognitive Activity-Dependent Regulation of Retinogeniculate Signaling by Metabotropic Glutamate Receptors Gubbi
12820 The Journal of Neuroscience, September 12, 2012 32(37):12820 12831 Behavioral/Systems/Cognitive Activity-Dependent Regulation of Retinogeniculate Signaling by Metabotropic Glutamate Receptors Gubbi
Supporting Information
 ATP from synaptic terminals and astrocytes regulates NMDA receptors and synaptic plasticity through PSD- 95 multi- protein complex U.Lalo, O.Palygin, A.Verkhratsky, S.G.N. Grant and Y. Pankratov Supporting
ATP from synaptic terminals and astrocytes regulates NMDA receptors and synaptic plasticity through PSD- 95 multi- protein complex U.Lalo, O.Palygin, A.Verkhratsky, S.G.N. Grant and Y. Pankratov Supporting
Thalamocortical Feedback and Coupled Oscillators
 Thalamocortical Feedback and Coupled Oscillators Balaji Sriram March 23, 2009 Abstract Feedback systems are ubiquitous in neural systems and are a subject of intense theoretical and experimental analysis.
Thalamocortical Feedback and Coupled Oscillators Balaji Sriram March 23, 2009 Abstract Feedback systems are ubiquitous in neural systems and are a subject of intense theoretical and experimental analysis.
1) Drop off in the Bi 150 box outside Baxter 331 or to the head TA (jcolas).
 Bi/CNS/NB 150 Problem Set 3 Due: Tuesday, Oct. 27, at 4:30 pm Instructions: 1) Drop off in the Bi 150 box outside Baxter 331 or e-mail to the head TA (jcolas). 2) Submit with this cover page. 3) Use a
Bi/CNS/NB 150 Problem Set 3 Due: Tuesday, Oct. 27, at 4:30 pm Instructions: 1) Drop off in the Bi 150 box outside Baxter 331 or e-mail to the head TA (jcolas). 2) Submit with this cover page. 3) Use a
How Nicotinic Signaling Shapes Neural Networks
 How Nicotinic Signaling Shapes Neural Networks Darwin K. Berg Division of Biological Sciences University of California, San Diego Nicotinic Cholinergic Signaling Uses the transmitter ACh to activate cation-selective
How Nicotinic Signaling Shapes Neural Networks Darwin K. Berg Division of Biological Sciences University of California, San Diego Nicotinic Cholinergic Signaling Uses the transmitter ACh to activate cation-selective
Cellular Bioelectricity
 ELEC ENG 3BB3: Cellular Bioelectricity Notes for Lecture 24 Thursday, March 6, 2014 8. NEURAL ELECTROPHYSIOLOGY We will look at: Structure of the nervous system Sensory transducers and neurons Neural coding
ELEC ENG 3BB3: Cellular Bioelectricity Notes for Lecture 24 Thursday, March 6, 2014 8. NEURAL ELECTROPHYSIOLOGY We will look at: Structure of the nervous system Sensory transducers and neurons Neural coding
Dep. Control Time (min)
 aa Control Dep. RP 1s 1 mv 2s 1 mv b % potentiation of IPSP 2 15 1 5 Dep. * 1 2 3 4 Time (min) Supplementary Figure 1. Rebound potentiation of IPSPs in PCs. a, IPSPs recorded with a K + gluconate pipette
aa Control Dep. RP 1s 1 mv 2s 1 mv b % potentiation of IPSP 2 15 1 5 Dep. * 1 2 3 4 Time (min) Supplementary Figure 1. Rebound potentiation of IPSPs in PCs. a, IPSPs recorded with a K + gluconate pipette
Neuroscience: Exploring the Brain, 3e. Chapter 4: The action potential
 Neuroscience: Exploring the Brain, 3e Chapter 4: The action potential Introduction Action Potential in the Nervous System Conveys information over long distances Action potential Initiated in the axon
Neuroscience: Exploring the Brain, 3e Chapter 4: The action potential Introduction Action Potential in the Nervous System Conveys information over long distances Action potential Initiated in the axon
Psychology 320: Topics in Physiological Psychology Lecture Exam 2: March 19th, 2003
 Psychology 320: Topics in Physiological Psychology Lecture Exam 2: March 19th, 2003 Name: Student #: BEFORE YOU BEGIN!!! 1) Count the number of pages in your exam. The exam is 8 pages long; if you do not
Psychology 320: Topics in Physiological Psychology Lecture Exam 2: March 19th, 2003 Name: Student #: BEFORE YOU BEGIN!!! 1) Count the number of pages in your exam. The exam is 8 pages long; if you do not
The Role of Mitral Cells in State Dependent Olfactory Responses. Trygve Bakken & Gunnar Poplawski
 The Role of Mitral Cells in State Dependent Olfactory Responses Trygve akken & Gunnar Poplawski GGN 260 Neurodynamics Winter 2008 bstract Many behavioral studies have shown a reduced responsiveness to
The Role of Mitral Cells in State Dependent Olfactory Responses Trygve akken & Gunnar Poplawski GGN 260 Neurodynamics Winter 2008 bstract Many behavioral studies have shown a reduced responsiveness to
Chapter 2: Cellular Mechanisms and Cognition
 Chapter 2: Cellular Mechanisms and Cognition MULTIPLE CHOICE 1. Two principles about neurons were defined by Ramón y Cajal. The principle of connectional specificity states that, whereas the principle
Chapter 2: Cellular Mechanisms and Cognition MULTIPLE CHOICE 1. Two principles about neurons were defined by Ramón y Cajal. The principle of connectional specificity states that, whereas the principle
Modeling synaptic facilitation and depression in thalamocortical relay cells
 College of William and Mary W&M ScholarWorks Undergraduate Honors Theses Theses, Dissertations, & Master Projects 5-2011 Modeling synaptic facilitation and depression in thalamocortical relay cells Olivia
College of William and Mary W&M ScholarWorks Undergraduate Honors Theses Theses, Dissertations, & Master Projects 5-2011 Modeling synaptic facilitation and depression in thalamocortical relay cells Olivia
Synaptic Integration
 Synaptic Integration 3 rd January, 2017 Touqeer Ahmed PhD Atta-ur-Rahman School of Applied Biosciences National University of Sciences and Technology Excitatory Synaptic Actions Excitatory Synaptic Action
Synaptic Integration 3 rd January, 2017 Touqeer Ahmed PhD Atta-ur-Rahman School of Applied Biosciences National University of Sciences and Technology Excitatory Synaptic Actions Excitatory Synaptic Action
Neurons. Pyramidal neurons in mouse cerebral cortex expressing green fluorescent protein. The red staining indicates GABAergic interneurons.
 Neurons Pyramidal neurons in mouse cerebral cortex expressing green fluorescent protein. The red staining indicates GABAergic interneurons. MBL, Woods Hole R Cheung MSc Bioelectronics: PGEE11106 1 Neuron
Neurons Pyramidal neurons in mouse cerebral cortex expressing green fluorescent protein. The red staining indicates GABAergic interneurons. MBL, Woods Hole R Cheung MSc Bioelectronics: PGEE11106 1 Neuron
Computational cognitive neuroscience: 2. Neuron. Lubica Beňušková Centre for Cognitive Science, FMFI Comenius University in Bratislava
 1 Computational cognitive neuroscience: 2. Neuron Lubica Beňušková Centre for Cognitive Science, FMFI Comenius University in Bratislava 2 Neurons communicate via electric signals In neurons it is important
1 Computational cognitive neuroscience: 2. Neuron Lubica Beňušková Centre for Cognitive Science, FMFI Comenius University in Bratislava 2 Neurons communicate via electric signals In neurons it is important
Supplementary Figure 1. ACE robotic platform. A. Overview of the rig setup showing major hardware components of ACE (Automatic single Cell
 2 Supplementary Figure 1. ACE robotic platform. A. Overview of the rig setup showing major hardware components of ACE (Automatic single Cell Experimenter) including the MultiClamp 700B, Digidata 1440A,
2 Supplementary Figure 1. ACE robotic platform. A. Overview of the rig setup showing major hardware components of ACE (Automatic single Cell Experimenter) including the MultiClamp 700B, Digidata 1440A,
Neurons! John A. White Dept. of Bioengineering
 Neurons! John A. White Dept. of Bioengineering john.white@utah.edu What makes neurons different from cardiomyocytes? Morphological polarity Transport systems Shape and function of action potentials Neuronal
Neurons! John A. White Dept. of Bioengineering john.white@utah.edu What makes neurons different from cardiomyocytes? Morphological polarity Transport systems Shape and function of action potentials Neuronal
Antiepileptic agents
 Antiepileptic agents Excessive excitability of neurons in the CNS Abnormal function of ion channels Spread through neural networks Abnormal neural activity leads to abnormal motor activity Suppression
Antiepileptic agents Excessive excitability of neurons in the CNS Abnormal function of ion channels Spread through neural networks Abnormal neural activity leads to abnormal motor activity Suppression
Bursting dynamics in the brain. Jaeseung Jeong, Department of Biosystems, KAIST
 Bursting dynamics in the brain Jaeseung Jeong, Department of Biosystems, KAIST Tonic and phasic activity A neuron is said to exhibit a tonic activity when it fires a series of single action potentials
Bursting dynamics in the brain Jaeseung Jeong, Department of Biosystems, KAIST Tonic and phasic activity A neuron is said to exhibit a tonic activity when it fires a series of single action potentials
Neural Basis of Motor Control. Chapter 4
 Neural Basis of Motor Control Chapter 4 Neurological Perspective A basic understanding of the physiology underlying the control of voluntary movement establishes a more comprehensive appreciation and awareness
Neural Basis of Motor Control Chapter 4 Neurological Perspective A basic understanding of the physiology underlying the control of voluntary movement establishes a more comprehensive appreciation and awareness
BIPN 140 Problem Set 6
 BIPN 140 Problem Set 6 1) The hippocampus is a cortical structure in the medial portion of the temporal lobe (medial temporal lobe in primates. a) What is the main function of the hippocampus? The hippocampus
BIPN 140 Problem Set 6 1) The hippocampus is a cortical structure in the medial portion of the temporal lobe (medial temporal lobe in primates. a) What is the main function of the hippocampus? The hippocampus
Neurons Chapter 7 2/19/2016. Learning Objectives. Cells of the Nervous System. Cells of the Nervous System. Cells of the Nervous System
 Learning Objectives Neurons Chapter 7 Identify and describe the functions of the two main divisions of the nervous system. Differentiate between a neuron and neuroglial cells in terms of structure and
Learning Objectives Neurons Chapter 7 Identify and describe the functions of the two main divisions of the nervous system. Differentiate between a neuron and neuroglial cells in terms of structure and
biological psychology, p. 40 The study of the nervous system, especially the brain. neuroscience, p. 40
 biological psychology, p. 40 The specialized branch of psychology that studies the relationship between behavior and bodily processes and system; also called biopsychology or psychobiology. neuroscience,
biological psychology, p. 40 The specialized branch of psychology that studies the relationship between behavior and bodily processes and system; also called biopsychology or psychobiology. neuroscience,
Function of the Nervous System
 Nervous System Function of the Nervous System Receive sensory information, interpret it, and send out appropriate commands to form a response Composed of neurons (functional unit of the nervous system)
Nervous System Function of the Nervous System Receive sensory information, interpret it, and send out appropriate commands to form a response Composed of neurons (functional unit of the nervous system)
Neurotransmitter Systems III Neurochemistry. Reading: BCP Chapter 6
 Neurotransmitter Systems III Neurochemistry Reading: BCP Chapter 6 Neurotransmitter Systems Normal function of the human brain requires an orderly set of chemical reactions. Some of the most important
Neurotransmitter Systems III Neurochemistry Reading: BCP Chapter 6 Neurotransmitter Systems Normal function of the human brain requires an orderly set of chemical reactions. Some of the most important
Ligand-Gated Ion Channels
 Ligand-Gated Ion Channels The Other Machines That Make It Possible... Topics I Introduction & Electrochemical Gradients Passive Membrane Properties Action Potentials Voltage-Gated Ion Channels Topics II
Ligand-Gated Ion Channels The Other Machines That Make It Possible... Topics I Introduction & Electrochemical Gradients Passive Membrane Properties Action Potentials Voltage-Gated Ion Channels Topics II
Chapter 5 subtitles GABAergic synaptic transmission
 CELLULAR NEUROPHYSIOLOGY CONSTANCE HAMMOND Chapter 5 subtitles GABAergic synaptic transmission INTRODUCTION (2:57) In this fifth chapter, you will learn how the binding of the GABA neurotransmitter to
CELLULAR NEUROPHYSIOLOGY CONSTANCE HAMMOND Chapter 5 subtitles GABAergic synaptic transmission INTRODUCTION (2:57) In this fifth chapter, you will learn how the binding of the GABA neurotransmitter to
Human Brain and Senses
 Human Brain and Senses Outline for today Levels of analysis Basic structure of neurons How neurons communicate Basic structure of the nervous system Levels of analysis Organism Brain Cell Synapses Membrane
Human Brain and Senses Outline for today Levels of analysis Basic structure of neurons How neurons communicate Basic structure of the nervous system Levels of analysis Organism Brain Cell Synapses Membrane
BIPN 140 Problem Set 6
 BIPN 140 Problem Set 6 1) Hippocampus is a cortical structure in the medial portion of the temporal lobe (medial temporal lobe in primates. a) What is the main function of the hippocampus? The hippocampus
BIPN 140 Problem Set 6 1) Hippocampus is a cortical structure in the medial portion of the temporal lobe (medial temporal lobe in primates. a) What is the main function of the hippocampus? The hippocampus
Exploring the Functional Significance of Dendritic Inhibition In Cortical Pyramidal Cells
 Neurocomputing, 5-5:389 95, 003. Exploring the Functional Significance of Dendritic Inhibition In Cortical Pyramidal Cells M. W. Spratling and M. H. Johnson Centre for Brain and Cognitive Development,
Neurocomputing, 5-5:389 95, 003. Exploring the Functional Significance of Dendritic Inhibition In Cortical Pyramidal Cells M. W. Spratling and M. H. Johnson Centre for Brain and Cognitive Development,
Theme 2: Cellular mechanisms in the Cochlear Nucleus
 Theme 2: Cellular mechanisms in the Cochlear Nucleus The Cochlear Nucleus (CN) presents a unique opportunity for quantitatively studying input-output transformations by neurons because it gives rise to
Theme 2: Cellular mechanisms in the Cochlear Nucleus The Cochlear Nucleus (CN) presents a unique opportunity for quantitatively studying input-output transformations by neurons because it gives rise to
Effect of Subthreshold Up and Down States on the Whisker-Evoked Response in Somatosensory Cortex
 J Neurophysiol 92: 3511 3521, 2004. First published July 14, 2004; doi:10.1152/jn.00347.2004. Effect of Subthreshold Up and Down States on the Whisker-Evoked Response in Somatosensory Cortex Robert N.
J Neurophysiol 92: 3511 3521, 2004. First published July 14, 2004; doi:10.1152/jn.00347.2004. Effect of Subthreshold Up and Down States on the Whisker-Evoked Response in Somatosensory Cortex Robert N.
Synaptic plasticityhippocampus. Neur 8790 Topics in Neuroscience: Neuroplasticity. Outline. Synaptic plasticity hypothesis
 Synaptic plasticityhippocampus Neur 8790 Topics in Neuroscience: Neuroplasticity Outline Synaptic plasticity hypothesis Long term potentiation in the hippocampus How it s measured What it looks like Mechanisms
Synaptic plasticityhippocampus Neur 8790 Topics in Neuroscience: Neuroplasticity Outline Synaptic plasticity hypothesis Long term potentiation in the hippocampus How it s measured What it looks like Mechanisms
Active Control of Spike-Timing Dependent Synaptic Plasticity in an Electrosensory System
 Active Control of Spike-Timing Dependent Synaptic Plasticity in an Electrosensory System Patrick D. Roberts and Curtis C. Bell Neurological Sciences Institute, OHSU 505 N.W. 185 th Avenue, Beaverton, OR
Active Control of Spike-Timing Dependent Synaptic Plasticity in an Electrosensory System Patrick D. Roberts and Curtis C. Bell Neurological Sciences Institute, OHSU 505 N.W. 185 th Avenue, Beaverton, OR
QUIZ YOURSELF COLOSSAL NEURON ACTIVITY
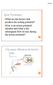 QUIZ YOURSELF What are the factors that produce the resting potential? How is an action potential initiated and what is the subsequent flow of ions during the action potential? 1 COLOSSAL NEURON ACTIVITY
QUIZ YOURSELF What are the factors that produce the resting potential? How is an action potential initiated and what is the subsequent flow of ions during the action potential? 1 COLOSSAL NEURON ACTIVITY
Supporting Online Material for
 www.sciencemag.org/cgi/content/full/317/5841/183/dc1 Supporting Online Material for Astrocytes Potentiate Transmitter Release at Single Hippocampal Synapses Gertrudis Perea and Alfonso Araque* *To whom
www.sciencemag.org/cgi/content/full/317/5841/183/dc1 Supporting Online Material for Astrocytes Potentiate Transmitter Release at Single Hippocampal Synapses Gertrudis Perea and Alfonso Araque* *To whom
What do you notice? Edited from
 What do you notice? Edited from https://www.youtube.com/watch?v=ffayobzdtc8&t=83s How can a one brain region increase the likelihood of eliciting a spike in another brain region? Communication through
What do you notice? Edited from https://www.youtube.com/watch?v=ffayobzdtc8&t=83s How can a one brain region increase the likelihood of eliciting a spike in another brain region? Communication through
Neuroscience 201A (2016) - Problems in Synaptic Physiology
 Question 1: The record below in A shows an EPSC recorded from a cerebellar granule cell following stimulation (at the gap in the record) of a mossy fiber input. These responses are, then, evoked by stimulation.
Question 1: The record below in A shows an EPSC recorded from a cerebellar granule cell following stimulation (at the gap in the record) of a mossy fiber input. These responses are, then, evoked by stimulation.
Synaptic Transmission: Ionic and Metabotropic
 Synaptic Transmission: Ionic and Metabotropic D. Purves et al. Neuroscience (Sinauer Assoc.) Chapters 5, 6, 7. C. Koch. Biophysics of Computation (Oxford) Chapter 4. J.G. Nicholls et al. From Neuron to
Synaptic Transmission: Ionic and Metabotropic D. Purves et al. Neuroscience (Sinauer Assoc.) Chapters 5, 6, 7. C. Koch. Biophysics of Computation (Oxford) Chapter 4. J.G. Nicholls et al. From Neuron to
CASE 48. What part of the cerebellum is responsible for planning and initiation of movement?
 CASE 48 A 34-year-old woman with a long-standing history of seizure disorder presents to her neurologist with difficulty walking and coordination. She has been on phenytoin for several days after having
CASE 48 A 34-year-old woman with a long-standing history of seizure disorder presents to her neurologist with difficulty walking and coordination. She has been on phenytoin for several days after having
What effect would an AChE inhibitor have at the neuromuscular junction?
 CASE 4 A 32-year-old woman presents to her primary care physician s office with difficulty chewing food. She states that when she eats certain foods that require a significant amount of chewing (meat),
CASE 4 A 32-year-old woman presents to her primary care physician s office with difficulty chewing food. She states that when she eats certain foods that require a significant amount of chewing (meat),
SUPPLEMENTARY INFORMATION
 Supplementary Figure 1. Normal AMPAR-mediated fepsp input-output curve in CA3-Psen cdko mice. Input-output curves, which are plotted initial slopes of the evoked fepsp as function of the amplitude of the
Supplementary Figure 1. Normal AMPAR-mediated fepsp input-output curve in CA3-Psen cdko mice. Input-output curves, which are plotted initial slopes of the evoked fepsp as function of the amplitude of the
Introduction to CNS 1
 Introduction to CNS 1 Types of ion channels 1- voltage-gated 2-legends-gated Voltage-gated channel A voltage Sensor component of the protein controls the gating (broken arrow) of the channel. Voltage-gated
Introduction to CNS 1 Types of ion channels 1- voltage-gated 2-legends-gated Voltage-gated channel A voltage Sensor component of the protein controls the gating (broken arrow) of the channel. Voltage-gated
Analysis of in-vivo extracellular recordings. Ryan Morrill Bootcamp 9/10/2014
 Analysis of in-vivo extracellular recordings Ryan Morrill Bootcamp 9/10/2014 Goals for the lecture Be able to: Conceptually understand some of the analysis and jargon encountered in a typical (sensory)
Analysis of in-vivo extracellular recordings Ryan Morrill Bootcamp 9/10/2014 Goals for the lecture Be able to: Conceptually understand some of the analysis and jargon encountered in a typical (sensory)
Basics of Computational Neuroscience: Neurons and Synapses to Networks
 Basics of Computational Neuroscience: Neurons and Synapses to Networks Bruce Graham Mathematics School of Natural Sciences University of Stirling Scotland, U.K. Useful Book Authors: David Sterratt, Bruce
Basics of Computational Neuroscience: Neurons and Synapses to Networks Bruce Graham Mathematics School of Natural Sciences University of Stirling Scotland, U.K. Useful Book Authors: David Sterratt, Bruce
Modelling corticothalamic feedback and the gating of the thalamus by the cerebral cortex
 J. Physiol. (Paris) 94 (2000) 391 410 2000 Elsevier Science Ltd. Published by Éditions scientifiques et médicales Elsevier SAS. All rights reserved PII: S0928-4257(00)01093-7/FLA Modelling corticothalamic
J. Physiol. (Paris) 94 (2000) 391 410 2000 Elsevier Science Ltd. Published by Éditions scientifiques et médicales Elsevier SAS. All rights reserved PII: S0928-4257(00)01093-7/FLA Modelling corticothalamic
Basic Neuroscience. Sally Curtis
 The Physiology of Pain Basic Neuroscience Sally Curtis sac3@soton.ac.uk The behaviour of humans is a result of the actions of nerves. Nerves form the basis of Thoughts, sensations and actions both reflex
The Physiology of Pain Basic Neuroscience Sally Curtis sac3@soton.ac.uk The behaviour of humans is a result of the actions of nerves. Nerves form the basis of Thoughts, sensations and actions both reflex
Introduction to Neurobiology
 Biology 240 General Zoology Introduction to Neurobiology Nervous System functions: communication of information via nerve signals integration and processing of information control of physiological and
Biology 240 General Zoology Introduction to Neurobiology Nervous System functions: communication of information via nerve signals integration and processing of information control of physiological and
Brain anatomy and artificial intelligence. L. Andrew Coward Australian National University, Canberra, ACT 0200, Australia
 Brain anatomy and artificial intelligence L. Andrew Coward Australian National University, Canberra, ACT 0200, Australia The Fourth Conference on Artificial General Intelligence August 2011 Architectures
Brain anatomy and artificial intelligence L. Andrew Coward Australian National University, Canberra, ACT 0200, Australia The Fourth Conference on Artificial General Intelligence August 2011 Architectures
Neurotransmitter Systems I Identification and Distribution. Reading: BCP Chapter 6
 Neurotransmitter Systems I Identification and Distribution Reading: BCP Chapter 6 Neurotransmitter Systems Normal function of the human brain requires an orderly set of chemical reactions. Some of the
Neurotransmitter Systems I Identification and Distribution Reading: BCP Chapter 6 Neurotransmitter Systems Normal function of the human brain requires an orderly set of chemical reactions. Some of the
Nervous System. 2. Receives information from the environment from CNS to organs and glands. 1. Relays messages, processes info, analyzes data
 Nervous System 1. Relays messages, processes info, analyzes data 2. Receives information from the environment from CNS to organs and glands 3. Transmits impulses from CNS to muscles and glands 4. Transmits
Nervous System 1. Relays messages, processes info, analyzes data 2. Receives information from the environment from CNS to organs and glands 3. Transmits impulses from CNS to muscles and glands 4. Transmits
Section: Chapter 5: Multiple Choice. 1. The structure of synapses is best viewed with a(n):
 Section: Chapter 5: Multiple Choice 1. The structure of synapses is best viewed with a(n): p.155 electron microscope. light microscope. confocal microscope. nissle-stained microscopic procedure. 2. Electron
Section: Chapter 5: Multiple Choice 1. The structure of synapses is best viewed with a(n): p.155 electron microscope. light microscope. confocal microscope. nissle-stained microscopic procedure. 2. Electron
Lateral view of human brain! Cortical processing of touch!
 Lateral view of human brain! Cortical processing of touch! How do we perceive objects held in the hand?! Touch receptors deconstruct objects to detect local features! Information is transmitted in parallel
Lateral view of human brain! Cortical processing of touch! How do we perceive objects held in the hand?! Touch receptors deconstruct objects to detect local features! Information is transmitted in parallel
Pharmacology of Pain Transmission and Modulation
 Pharmacology of Pain Transmission and Modulation 2 Jürg Schliessbach and Konrad Maurer Nociceptive Nerve Fibers Pain is transmitted to the central nervous system via thinly myelinated Aδ and unmyelinated
Pharmacology of Pain Transmission and Modulation 2 Jürg Schliessbach and Konrad Maurer Nociceptive Nerve Fibers Pain is transmitted to the central nervous system via thinly myelinated Aδ and unmyelinated
Concept 48.1 Neuron organization and structure reflect function in information transfer
 Name Chapter 48: Neurons, Synapses, and Signaling Period Chapter 48: Neurons, Synapses, and Signaling Concept 48.1 Neuron organization and structure reflect function in information transfer 1. What is
Name Chapter 48: Neurons, Synapses, and Signaling Period Chapter 48: Neurons, Synapses, and Signaling Concept 48.1 Neuron organization and structure reflect function in information transfer 1. What is
Biomarkers in Schizophrenia
 Biomarkers in Schizophrenia David A. Lewis, MD Translational Neuroscience Program Department of Psychiatry NIMH Conte Center for the Neuroscience of Mental Disorders University of Pittsburgh Disease Process
Biomarkers in Schizophrenia David A. Lewis, MD Translational Neuroscience Program Department of Psychiatry NIMH Conte Center for the Neuroscience of Mental Disorders University of Pittsburgh Disease Process
