PRODUCT INFORMATION. Concentrate for IV infusion. AGGRASTAT (tirofiban hydrochloride) DESCRIPTION
|
|
|
- Joella Harris
- 6 years ago
- Views:
Transcription
1 ARS s 1 PRODUCT INFORMATION Concentrate for IV infusion AGGRASTAT (tirofiban hydrochloride) DESCRIPTION AGGRASTAT (tirofiban hydrochloride, MSD), a non-peptide antagonist of the platelet glycoprotein (GP) IIb/IIIa receptor, is a platelet aggregation inhibitor. Tirofiban hydrochloride monohydrate is chemically described as N-(butylsulfonyl)-O- [4-(4-piperidinyl)butyl]-L-tyrosine monohydrochloride monohydrate. The CAS No is Its empirical formula is C 22 H 36 N 2 O 5 S HCl H 2 O, and its structural formula is: HN CH 2 CH 2 CH 2 CH 2 O CH 2 C COOH. HCl H2 O H NHSO 2 CH 2 CH 2 CH 2 CH 3. Tirofiban hydrochloride monohydrate is a white to off-white non-hygroscopic freeflowing powder, with a molecular weight of It is very slightly soluble in water. AGGRASTAT Concentrate for Infusion is a sterile concentrated solution for intravenous infusion after dilution and is supplied in a 50 ml glass vial. Each ml of the solution contains mg of tirofiban hydrochloride monohydrate equivalent to 0.25 mg of tirofiban and the following inactive ingredients: 0.16 mg citric acid anhydrous, 2.7 mg sodium citrate and 8 mg sodium chloride and water for injections. The ph ranges from 5.5 to 6.5 and may have been adjusted with hydrochloric acid and/or sodium hydroxide. Registered Trademark of Iroko Cardio LLC, Philadelphia, PA, U.S.A
2 ARS s 2 PHARMACOLOGY Mechanism of Action Platelet activation, adhesion and aggregation represent critical initiating steps in the formation of arterial thrombus overlying disrupted atherosclerotic plaque. Thrombus formation is central to the pathophysiology of the acute coronary ischaemic syndromes of unstable angina and myocardial infarction, as well as to cardiac ischaemic complications following coronary angioplasty. AGGRASTAT is a non-peptide antagonist of the GP IIb/IIIa receptor, the major platelet surface receptor involved in platelet aggregation. AGGRASTAT prevents binding of fibrinogen to GP IIb/IIIa, thereby blocking the cross-linking of platelets and platelet aggregation. Pharmacokinetics Distribution Tirofiban is not highly bound to plasma proteins and protein binding is concentration independent over the range of 0.01 to 25 µg/ml. Unbound fraction in human plasma is 35%. The steady state volume of distribution of tirofiban ranges from 22 to 42 litres. Metabolism Profiling of 14 C-labelled tirofiban in urine and faeces indicates that the radioactivity was accounted for mainly by unchanged tirofiban. Circulating plasma radioactivity is accounted for mainly by unchanged tirofiban (up to 10 hours postdose). These data suggest limited metabolism of tirofiban. Elimination Following an intravenous dose of 14 C-labelled tirofiban in healthy subjects, 66% of radioactivity is recovered in the urine and 23% in the faeces. Total radioactivity recovery is about 91%. Both urinary and biliary excretion contribute significantly to the elimination of tirofiban. In healthy subjects, the plasma clearance of tirofiban ranges from 213 to 314 ml/min. Renal clearance accounts for 39 to 69% of plasma clearance. Half-life ranges from 1.4 to 1.8 hours. In patients with coronary artery disease, the plasma clearance of tirofiban ranges from 152 to 267 ml/min. Renal clearance accounts for 39% of plasma clearance. Half-life ranges from 1.9 to 2.2 hours. Characteristics in Patients Gender Plasma clearance of tirofiban in patients with coronary artery disease is similar in males and females.
3 ARS s 3 Elderly Plasma clearance of tirofiban is about 19 to 26% lower in elderly (>65 years) patients with coronary artery disease compared to younger ( 65 years) patients. Race No difference in plasma clearance was detected in patients of different races. Hepatic Insufficiency In patients with mild to moderate hepatic insufficiency, plasma clearance of tirofiban is not significantly different compared to healthy subjects. Renal Insufficiency Plasma clearance of tirofiban is lower to a clinically significant extent (>50%) in patients with creatinine clearance <30 ml/min, including patients requiring haemodialysis (see DOSAGE AND ADMINISTRATION, Patients with Severe Renal Insufficiency). Tirofiban is removed by haemodialysis. Pharmacodynamics AGGRASTAT causes potent inhibition of platelet function as demonstrated by its ability to inhibit ex vivo adenosine phosphate (ADP)-induced platelet aggregation and prolong bleeding time (BT) in healthy subjects and patients with coronary artery disease. The time course of inhibition parallels the plasma concentration profile of the drug. Following discontinuation of an infusion of AGGRASTAT, platelet function rapidly returns to baseline. Coadministration of a 4-hour infusion of 0.15 µg/kg/min of AGGRASTAT and aspirin results in the anticipated near maximal inhibition of platelet aggregation and a modest additive effect of BT prolongation. In patients with unstable angina, a two-staged intravenous infusion regimen of AGGRASTAT (loading infusion of 0.4 µg/kg/min for 30 minutes followed by 0.1 µg/kg/min for up to 48 hours in the presence of heparin and aspirin), produces approximately 90% inhibition of ex vivo ADP-induced platelet aggregation with a 2.9- fold prolongation of bleeding time during the infusion. Inhibition was achieved rapidly with the 30-minute loading infusion and was maintained over the duration of the infusion. Clinical Studies Unstable Angina/Non-Q-Wave Myocardial Infarction PRISM-PLUS (Platelet Receptor Inhibition for Ischaemic Syndrome Management - Patients Limited by Unstable Signs and Symptoms) In the multi-centre, randomised, parallel, double-blind PRISM-PLUS trial, the use of AGGRASTAT in combination with heparin versus heparin alone was evaluated in patients with documented unstable angina/non-q-wave myocardial infarction. In this study, patients were randomised to either AGGRASTAT (30 minute loading infusion of 0.4 µg/kg/min followed by a maintenance infusion of 0.10 µg/kg/min) and heparin (bolus of 5,000 units (U) followed by an infusion of 1,000 U/hr titrated to
4 ARS s 4 maintain an activated partial thromboplastin time (APTT) of approximately 2 times control), or heparin alone (bolus of 5,000 U followed by an infusion of 1,000 U/hr titrated to maintain an APTT of approximately 2 times control). A third group of patients was initially randomised to AGGRASTAT (30 minute loading dose of 0.6 µg/kg/min followed by a maintenance infusion of 0.15µg/kg/min) with no heparin. This arm was discontinued when an increase in mortality was noted at seven days [16 deaths (4.6%) with tirofiban, as compared with 4 deaths (1.1%) with heparin]. All patients received concomitant aspirin unless contraindicated. Therapy with AGGRASTAT commenced within 12 hours of the last episode of chest pain. Patients underwent 48 hours of medical stabilisation on study drug therapy, after which angiography and angioplasty/atherectomy remained options while continuing on AGGRASTAT. AGGRASTAT was generally administered for a minimum of 48 hours and was continued up to 108 hours; on average, patients received AGGRASTAT for 71.3 hours. The primary endpoint of the study was a composite of refractory ischaemia, new myocardial infarction and death at 7 days following initiation of AGGRASTAT. At the primary endpoint, there was a 31.6% risk reduction in the overall composite, a 46.6% risk reduction in myocardial infarction, and a 42.8% risk reduction in the composite of myocardial infarction and death. The results are shown in Table 1: Endpoint Table 1 Cardiac Ischaemic Events (7 Days) AGGRASTAT +Heparin (n=773) Heparin (n=797) Risk Reduction p-value Composite Endpoint 12.9% 17.9% 31.6% Components Myocardial Infarction 4.9% 8.3% 42.8% and Death Myocardial Infarction 3.9% 7.0% 46.6% Death 1.9% 1.9% - - Refractory Ischaemia 9.3% 12.7% 29.6% At 30 days, the risk of the composite endpoint was reduced by 22% (p=0.029) and there was a 30% reduction in the composite of death and myocardial infarction (p=0.027). At 6 months, the risk of the composite endpoint was reduced by 18.9% (p=0.024); in addition, there was 22.5% risk reduction in the composite of myocardial infarction and death. The risk reduction in the composite endpoint at 7 days, 30 days and 6 months is shown in the Kaplan-Meier curve below.
5 ARS s 5 In the 30% of patients who underwent angioplasty/atherectomy in the PRISM-PLUS study, there was a 45.7% risk reduction in the composite endpoint following the procedure at day 30 after start of study drug, as well as a 43.2% risk reduction in the composite of death and myocardial infarction. A sub-study in PRISM-PLUS found that there was a significant decrease in the extent of angiographically apparent thrombus in patients treated with AGGRASTAT in combination with heparin compared to heparin alone. In addition, flow in the affected coronary artery was significantly improved. PRISM-PLUS was not designed to provide definitive results in subsets of the overall population. Nonetheless, demographic results were examined for age, gender and race. No difference in benefit was noticed for age and gender but too few non- Caucasians were enrolled to make a definite statement about racial differences in treatment effect. PRISM (Platelet Receptor Inhibition for Ischaemic Syndrome Management). In the PRISM study, a randomised, parallel, double-blind, active control study, AGGRASTAT alone (n=1616) was compared to heparin (n=1616) alone as medical management in patients with unstable angina/non-q-wave myocardial infarction. In this study, the drug was started within 24 hours of the time the patient experienced chest pain. Patients were randomised to either AGGRASTAT alone (30 minute loading dose of 0.6 µg/kg/min, followed by a maintenance infusion of 0.15 µg/kg/min) or heparin alone (bolus of 5,000 U followed by an infusion of 1000U/hr titrated to maintain an APTT of approximately 2 times control). The mean age of the population was 62 years; 32% of the population was female and 25% had non-q-wave myocardial infarction on presentation. Thirty percent had no ECG evidence of cardiac ischaemia. Exclusion criteria were similar to PRISM-PLUS. The primary, prospectively identified endpoint was the composite endpoint of refractory ischaemia, myocardial infarction or death after a 48-hour drug infusion with AGGRASTAT. The results are shown in Table 2.
6 ARS s 6 Table 2 Cardiac Ischaemic Events Composite AGGRASTAT Heparin Risk Reduction p-value Endpoint (n=1616) (n=1616) 2 Days 3.8% 5.6% 33% Days 10.3% 11.3% 10% Days 15.9% 17.1% 8% 0.34 In the PRISM study, no adverse effect of AGGRASTAT on mortality at either 7 or 30 days was detected. INDICATIONS AGGRASTAT, in combination with heparin, is indicated for patients with unstable angina or non-q-wave myocardial infarction to prevent cardiac ischaemic events. (See PHARMACOLOGY and DOSAGE AND ADMINISTRATION.) CONTRAINDICATIONS AGGRASTAT is contraindicated in patients with known hypersensitivity to any component of the product active internal bleeding or a history of bleeding diathesis within the previous 30 days. a history of intracranial haemorrhage, intracranial neoplasm, arteriovenous malformation, or aneurysm a history of thrombocytopenia following prior exposure to AGGRASTAT history of stroke within 30 days or any history of haemorrhagic stroke major surgical procedure (including epidural or spinal anaesthesia) or severe physical trauma within 1 month history, symptoms, or findings suggestive of aortic dissection severe uncontrolled hypertension (systolic blood pressure >180 mmhg and/or diastolic blood pressure >110 mmhg) concomitant use of another parenteral GP IIb/IIIa inhibitor acute pericarditis PRECAUTIONS AGGRASTAT should be used with caution in the following patients: recent (<1 year) bleeding, including a history of gastrointestinal bleeding, or genitourinary bleeding of clinical significance known coagulopathy, platelet disorder, or history of thrombocytopenia platelet count <150,000 cells/mm 3 history of cerebrovascular disease within 1 year recent epidural procedure haemorrhagic retinopathy chronic haemodialysis
7 ARS s 7 Bleeding Precautions Because AGGRASTAT inhibits platelet aggregation, caution should be employed when it is used with other drugs that affect haemostasis. The safety of AGGRASTAT when used in combination with thrombolytic agents has not been established. During therapy with AGGRASTAT, patients should be monitored for potential bleeding. When treatment of bleeding is required, discontinuation of the drug should be considered. Consideration may also be given to transfusions. Fatal bleedings have been reported (see ADVERSE REACTIONS). Femoral artery access site AGGRASTAT is associated with minor increases in bleeding rates particularly at the site of arterial access for femoral sheath placement. Care should be taken when attempting vascular access that only the anterior wall of the femoral artery is punctured, avoiding a Seldinger (through and through) technique for obtaining sheath access. Care should be taken to obtain proper haemostasis after removal of the sheaths followed by close observation. Laboratory Monitoring Platelet counts, and haemoglobin and haematocrit should be monitored prior to treatment, within 6 hours following the bolus or loading infusion, and at least daily thereafter during therapy with AGGRASTAT (or more frequently if there is evidence of significant decline). In patients who have previously received GP IIb/IIIa receptor antagonists, consideration should be given to earlier monitoring of platelet count. If the patient experiences a platelet count decrease to <90,000 cells/mm3, additional platelet counts should be performed to exclude pseudothrombocytopenia. If thrombocytopenia is confirmed, AGGRASTAT and heparin should be discontinued and the condition appropriately monitored and treated. In addition, the activated partial thromboplastin time (APTT) should be determined before treatment and the anticoagulant effects of heparin should be carefully monitored by repeated determinations of APTT and the dose adjusted accordingly (see also DOSAGE and ADMINISTRATION).Potentially life-threatening bleeding may occur especially when heparin is administered with other products affecting haemostasis, such as GP IIb/IIIa receptor antagonists.. Severe Renal Insufficiency In clinical studies, patients with severe renal insufficiency (creatinine clearance <30 ml/min) demonstrated decreased plasma clearance of AGGRASTAT. The dosage of AGGRASTAT should be reduced in these patients (see DOSAGE AND ADMINISTRATION and PHARMACOLOGY-Clinical Studies). CARCINOGENICITY/MUTAGENICITY AND IMPAIRMENT OF FERTILITY The carcinogenic potential of tirofiban hydrochloride has not been evaluated. Tirofiban was not genotoxic in a series of assays for gene mutations (Salmonella typhimurium, Escherichia coli and Chinese hamster lung cells), chromosomal aberrations (Chinese hamster ovary cell in vitro and mouse bone marrow in vivo) or DNA damage (alkaline elution assay).
8 ARS s 8 Fertility and reproductive performance were not affected in studies with male and female rats given intravenous doses of tirofiban hydrochloride up to 5 mg/kg/day. USE IN PREGNANCY (Category B1) There are no adequate and well-controlled studies in pregnant women. AGGRASTAT should be used during pregnancy only if the potential benefit justifies the potential risk to the foetus. Studies of developmental toxicity in rats and rabbits showed no evidence of maternal or foetal toxicity. In addition, a study of the potential developmental toxicity through sexual maturity of rats exposed in utero and during lactation showed no drug-related effects on mortality, growth, development, and sexual maturation of the F 1 generation. In the developmental toxicity studies, dams were given tirofiban hydrochloride intravenously at doses up to 5 mg/kg/day. USE IN LACTATION It is not known whether AGGRASTAT is excreted in human milk, but it is excreted in rat milk. Because many drugs are excreted in human milk, and because of the potential for adverse effects on the nursing infant, a decision should be made whether to discontinue nursing or discontinue the drug, taking into account the importance of the drug to the mother. Tirofiban hydrochloride crosses the placenta in rats and rabbits. USE IN CHILDREN Safety and effectiveness in children have not been established. USE IN THE ELDERLY In clinical studies the efficacy of AGGRASTAT in the elderly ( 65 years) was comparable to that seen in younger patients (<65 years). Elderly patients receiving AGGRASTAT with heparin or heparin alone had a higher incidence of bleeding complications than younger patients. The incremental risk of bleeding in patients treated with AGGRASTAT in combination with heparin over the risk in patients treated with heparin alone was comparable regardless of age. The overall incidence of non-bleeding adverse events was higher in older patients (compared to younger patients); however, the incidence of non-bleeding adverse events in these patients was comparable between the AGGRASTAT with heparin and the heparin alone groups. No dose adjustment is recommended (see DOSAGE AND ADMINISTRATION). This drug is known to be excreted by the kidney. A 50% reduction in dose is recommended for patients with severe renal insufficiency (creatinine clearance <30 ml/min) (see DOSAGE AND ADMINISTRATION, and PRECAUTIONS). Plasma clearance of AGGRASTAT is about 19 to 26% lower in elderly (>65 years) patients with coronary artery disease compared to younger ( 65 years) patients.
9 ARS s 9 INTERACTIONS WITH OTHER DRUGS AGGRASTAT has been studied on a background of aspirin and heparin. The use of AGGRASTAT, in combination with heparin and aspirin, has been associated with an increase in bleeding compared to heparin and aspirin alone (see ADVERSE REACTIONS). Caution should be employed when AGGRASTAT is used with other drugs that affect haemostasis (e.g., warfarin) (see PRECAUTIONS- Bleeding Precautions). The safety of AGGRASTAT when used in combination with thrombolytic agents has not been established. AGGRASTAT has been used concomitantly in clinical studies with beta-blockers, calcium channel blockers, non-steroidal anti-inflammatory drugs (NSAIDs) and nitrate preparations without evidence of clinically significant adverse interactions. In a sub-set of patients (n=762) in the PRISM study, the plasma clearance of tirofiban in patients receiving one of the following drugs was compared to that in patients not receiving that drug. There were no clinically significant interactions of these drugs on the plasma clearance of tirofiban: acebutolol, alprazolam, amlodipine, aspirin preparations, atenolol, bromazepam, captopril, diazepam, digoxin, diltiazem, docusate sodium, enalapril, frusemide, glibenclamide, heparin, insulin, isosorbide, thyroxine sodium, lorazepam, lovastatin, metoclopramide, metoprolol, morphine, nifedipine, nitrate preparations, omeprazole, oxazepam, paracetamol, potassium chloride, propranolol, ranitidine, simvastatin, sucralfate and temazepam. EFFECTS ON LABORATORY TESTS The most frequently observed laboratory adverse events in patients receiving AGGRASTAT concomitantly with heparin were related to bleeding. Decreases in haemoglobin and haematocrit, and platelet count were observed. Increases in the presence of urine and faecal occult blood were also observed. ADVERSE REACTIONS Bleeding: The most common drug-related adverse event reported during therapy with AGGRASTAT when used concomitantly with heparin and aspirin, was bleeding, usually reported by the investigators as oozing or mild. (See also PRECAUTIONS- Effects on Laboratory Tests.) The incidences of major and minor bleeding using the TIMI ** Criteria in the PRISM PLUS (Platelet Receptor Inhibition for Ischaemic Syndrome Management - Patients Limited by Unstable Signs and Symptoms) study are shown below: PRISM PLUS (UAP/Non-Q-Wave MI Study) ** Bovill, E.G.; et al: Haemorrhagic Events during Therapy with Recombinant Tissue-Type Plasminogen Activator, Heparin, and Aspirin for Acute Myocardial Infarction, Results of the Thrombolysis in Myocardial Infarction (TIMI), Phase II Trial, Annals of Internal Medicine, 115(4): , 1991.
10 ARS s 10 Bleeding AGGRASTAT + Heparin (n=773) % Heparin (n=797) % Major Bleeding (TIMI Criteria) Minor Bleeding (TIMI Criteria) Transfusions Patients received aspirin unless contraindicated. Haemoglobin drop of >50 g/l with or without an identified site, intracranial haemorrhage, or cardiac tamponade. Haemoglobin drop of >30 g/l with bleeding from a known site, spontaneous gross haematuria, haematemesis or haemoptysis. There were no reports of intracranial bleeding in the PRISM PLUS study for AGGRASTAT in combination with heparin or in the control group (which received heparin). In the PRISM PLUS Study, the incidences of retroperitoneal bleeding reported for AGGRASTAT in combination with heparin, and for the control group were 0.0% and 0.1%, respectively. Female patients and elderly patients receiving AGGRASTAT with heparin or heparin alone had a higher incidence of bleeding complications than male patients or younger patients, respectively. The incremental risk of bleeding in patients treated with AGGRASTAT in combination with heparin over the risk in patients treated with heparin alone was comparable regardless of age or gender. No dose adjustment is recommended for these populations (see DOSAGE AND ADMINISTRATION). Thrombocytopenia Patients treated with AGGRASTAT, with heparin, were more likely to experience decreases in platelet counts than the control group. These decreases were reversible upon discontinuation of AGGRASTAT. The percentage of patients with a decrease of platelets to <90,000 cells/mm 3 was 1.5%. The percentage of patients with a decrease of platelets to <50,000 cells/mm 3 was 0.3%. Platelet decreases have been observed in patients with no prior history of thrombocytopenia upon readministration of GP IIb/IIIa receptor antagonists Other adverse reactions The most frequent drug-related non-bleeding adverse reactions reported with AGGRASTAT, administered concomitantly with heparin, occurring at an incidence of >1% were: nausea (1.7% v 1.4% control) fever (1.5% v 1.1% control) headache (1.1% v 1.2% control). In clinical studies, the incidences of adverse events were generally similar among different races, patients with or without hypertension, patients with or without diabetes mellitus, and patients with or without hypercholesteremia.
11 ARS s 11 The overall incidence of non-bleeding adverse events was higher in female patients (compared to male patients) and older patients (compared to younger patients). However, the incidences of non-bleeding adverse events in these patients were comparable between the AGGRASTAT with heparin and the heparin alone groups. (See above for bleeding adverse events.) The following additional adverse reactions have been reported in post-marketing experience: Bleeding: intracranial bleeding, retroperitoneal bleeding, haemopericardium,pulmonary (alveolar) haemorrhage and spinal-epidural hematoma. Fatal bleedings have been reported rarely. Body as a Whole: Acute and/or severe decreases in platelet counts which may be associated with chills, low-grade fever, or bleeding complications (see above). Hypersensitivity: rash and urticaria. Severe allergic reactions including anaphylactic reactions. The reported cases have occurred during the first day of tirofiban infusion, during initial treatment, and during readministration of tirofiban. Some cases have been associated with severe thrombocytopenia (platelet counts < 10,000/mm 3 ). DOSAGE AND ADMINISTRATION The vial of AGGRASTAT (concentrate) must be diluted prior to administration (see INSTRUCTIONS FOR USE) AGGRASTAT is for intravenous use only using sterile equipment. AGGRASTAT may be co-administered with heparin through the same line. AGGRASTAT should not be administered in the same intravenous line as diazepam. AGGRASTAT is recommended for use with a calibrated infusion device. Care should be taken to avoid a prolonged loading infusion. Care should also be taken in calculating the bolus dose and infusion rates based on patient weight. In clinical studies patients received aspirin, unless contraindicated. AGGRASTAT should be administered intravenously at the initial infusion rate of 0.4 µg/kg/min for 30 minutes. Upon completion of the initial infusion, AGGRASTAT should be continued at a maintenance infusion rate of 0.1 µg/kg/min. AGGRASTAT should be given with heparin (usually an intravenous bolus dose of 5000 U simultaneously with the start of therapy with AGGRASTAT, then approximately 1000 U per hour titrated on the basis of APTT, which should be about twice the normal value). In the study that demonstrated efficacy, AGGRASTAT in combination with heparin was generally continued for a minimum of 48 hours and up to 108 hours. This infusion can be continued through angiography and should be continued up to 12 to 24 hours post-angioplasty/atherectomy. Arterial sheaths should be removed when the patient s activated clotting time is <180 seconds or 2-6 hours following cessation of heparin. (See PHARMACOLOGY-Clinical Studies.)
12 ARS s 12 Patients With Severe Renal Insufficiency The dosage of AGGRASTAT should be decreased by 50% in patients with severe renal insufficiency (creatinine clearance <30 ml/min). (See PRECAUTIONS-Severe Renal Insufficiency and PHARMACOLOGY-Pharmacokinetics, Characteristics in Patients, Renal Insufficiency.) The following table is provided as a guide to dosage adjustment by weight. Patient Weight (kg) 30 Min Loading Infusion Rate (ml/hr) Most Patients Maintenance Infusion Rate (ml/hr) Severe Kidney Failure 30 Min Loading Infusion Rate (ml/hr) Maintenance Infusion Rate (ml/hr) Other Patient Populations No dosage adjustment is recommended for elderly patients (see Precautions-Use in the Elderly) or female patients.
13 ARS s 13 INSTRUCTIONS FOR USE Parenteral drug products should be inspected visually for particulate matter and discoloration prior to use, whenever solution and container permit. Concentrate for Infusion The vial of AGGRASTAT (concentrate) must be diluted prior to administration. It contains no antimicrobial agent. Use once only and discard any residue. Directions for Preparation of AGGRASTAT Solution for Infusion from Concentrate 1. Withdraw and discard 50mL from a 250mL bag of sterile 0.9% saline or 5% glucose in water and replace it with 50mL of AGGRASTAT (from one 50 ml vial) to achieve a final concentration of 0.05mg/mL. Mix well before administration. Alternatively, withdraw and discard 100 ml from a 500 ml bag of sterile 0.9% saline or 5% glucose in water and replace it with 100 ml of AGGRASTAT (from two 50 ml vials) to achieve a final concentration of 0.05mg/mL. Mix well before administration. 2. Administer according to the appropriate dosage calculations above. 3. To reduce microbiological hazard, use as soon as practicable after dilution. Discard any unused intravenous solution after 24 hours following start of infusion. If storage is necessary, hold at 2-8 C for not more than 24 hours. OVERDOSAGE In clinical trials, inadvertent overdosage with tirofiban occurred in doses up to 5 times and 2 times the recommended dose for bolus administration and loading infusion, respectively. Inadvertent overdosage occurred in doses up to 9.8 times the 0.15 µg/kg/min maintenance infusion rate. The most frequently reported manifestation of overdosage was bleeding, primarily minor mucocutaneous bleeding events and minor bleeding at the sites of cardiac catheterisation (see PRECAUTIONS-Bleeding Precautions). Overdosage of tirofiban should be treated by assessment of the patient s clinical condition and cessation or adjustment of the drug infusion as appropriate. AGGRASTAT can be removed by haemodialysis.
14 ARS s 14 PRESENTATION AND STORAGE AGGRASTAT Concentrate for Infusion 0.25mg/mL 50 ml glass vials. Must be diluted prior to administration (see INSTRUCTIONS FOR USE), to give a concentration of 0.05mg/mL. Store below 30 C. Do not freeze. Protect from light during storage. SPONSOR Iroko Cardio Australia Pty Ltd Level 43, AMP Centre, 50 Bridge Street, NSW DISTRIBUTED BY Aspen Pharmacare Australia Pty Ltd Chandos Street, St Leonards, NSW 2065 Australia This document was approved by the Therapeutic Goods Administration on 22 April Safety Related Change notified to the Therapeutic Goods Administration on 19 January Updated 19 March 2009
CONTRAINDICATIONS INDICATIONS AND USAGE WARNINGS AND PRECAUTIONS ADVERSE REACTIONS DOSAGE AND ADMINISTRATION DRUG INTERACTIONS
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use AGGRASTAT (tirofiban hydrochloride) safely and effectively. See full prescribing information for
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use AGGRASTAT (tirofiban hydrochloride) safely and effectively. See full prescribing information for
DOSING AND ADMINISTRATION
 DOSING AND ADMINISTRATION USING 100 ML VIAL OR 250 ML BAG (50 MCG/ML UNITS) 1 2 BOLUS 25 mcg/kg within 5 minutes INFUSION CrCl >60 ml/min: 0.15 mcg/kg/min CrCl
DOSING AND ADMINISTRATION USING 100 ML VIAL OR 250 ML BAG (50 MCG/ML UNITS) 1 2 BOLUS 25 mcg/kg within 5 minutes INFUSION CrCl >60 ml/min: 0.15 mcg/kg/min CrCl
AGGRASTAT (tirofiban hydrochloride) injection, for intravenous use Initial U.S. Approval: 1998
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use AGGRASTAT (tirofiban hydrochloride) safely and effectively. See full prescribing information for
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use AGGRASTAT (tirofiban hydrochloride) safely and effectively. See full prescribing information for
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS
 ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS IV 1. NAME OF THE MEDICINAL PRODUCT AGGRASTAT/AGRASTAT * concentrate for solution for infusion 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 1 ml of concentrate
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS IV 1. NAME OF THE MEDICINAL PRODUCT AGGRASTAT/AGRASTAT * concentrate for solution for infusion 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 1 ml of concentrate
PRE-MIXED BAGS DOSING AND ADMINISTRATION. Rate (ml/hr)
 Weight (lb) Weight (kg) PRE-MIXED BAGS DOSING AND ADMINISTRATION 1 2 BOLUS Using Pre-Mixed Bags 25 mcg/kg within 5 minutes Volume (ml) Rate (ml/hr) INFUSION Using Pre-Mixed Bags CrCl >60 ml/min: 0.15 mcg/kg/min
Weight (lb) Weight (kg) PRE-MIXED BAGS DOSING AND ADMINISTRATION 1 2 BOLUS Using Pre-Mixed Bags 25 mcg/kg within 5 minutes Volume (ml) Rate (ml/hr) INFUSION Using Pre-Mixed Bags CrCl >60 ml/min: 0.15 mcg/kg/min
BOLUS VIAL + INFUSION
 INDICATIONS AND USAGE AGGRASTAT is indicated to reduce the rate of thrombotic cardiovascular events (combined endpoint of death, myocardial infarction, or refractory ischemia/ repeat cardiac procedure)
INDICATIONS AND USAGE AGGRASTAT is indicated to reduce the rate of thrombotic cardiovascular events (combined endpoint of death, myocardial infarction, or refractory ischemia/ repeat cardiac procedure)
COMMITTEE FOR PROPRIETARY MEDICINAL PRODUCTS (CPMP)
 The European Agency for the Evaluation of Medicinal Products Human Medicines Evaluation Unit 18 June 1999 CPMP/990/99-EN COMMITTEE FOR PROPRIETARY MEDICINAL PRODUCTS (CPMP) OPINION FOLLOWING AN ARTICLE
The European Agency for the Evaluation of Medicinal Products Human Medicines Evaluation Unit 18 June 1999 CPMP/990/99-EN COMMITTEE FOR PROPRIETARY MEDICINAL PRODUCTS (CPMP) OPINION FOLLOWING AN ARTICLE
P-RMS: LT/H/PSUR/0004/001
 Core Safety Profile Active substance: Dalteparine Pharmaceutical form(s)/strength: Solution for injection, 2500 I.U./0.2ml, 2500 I.U./ml, 5000 I.U./0.2ml, 7500 I.U./0.3ml, 7500 I.U./0.75ml, 10000 I.U./0.4ml,
Core Safety Profile Active substance: Dalteparine Pharmaceutical form(s)/strength: Solution for injection, 2500 I.U./0.2ml, 2500 I.U./ml, 5000 I.U./0.2ml, 7500 I.U./0.3ml, 7500 I.U./0.75ml, 10000 I.U./0.4ml,
PRODUCT MONOGRAPH AGGRASTAT. tirofiban hydrochloride injection mg / 250 ml tirofiban. (5 mg / 100 ml in bags of 250 ml)
 PRODUCT MONOGRAPH Pr AGGRASTAT tirofiban hydrochloride injection 12.5 mg / 250 ml tirofiban (5 mg / 100 ml in bags of 250 ml) Sterile Solution for Intravenous Infusion only Platelet aggregation inhibitor
PRODUCT MONOGRAPH Pr AGGRASTAT tirofiban hydrochloride injection 12.5 mg / 250 ml tirofiban (5 mg / 100 ml in bags of 250 ml) Sterile Solution for Intravenous Infusion only Platelet aggregation inhibitor
NURSING DEPARTMENT CRITICAL CARE POLICY MANUAL CRITICAL CARE PROTOCOLS EPTIFIBATIDE (INTEGRILIN) PROTOCOL
 NURSING DEPARTMENT CRITICAL CARE POLICY MANUAL CRITICAL CARE S EPTIFIBATIDE (INTEGRILIN) I. PURPOSE: A. Integrilin (Eptifibatide) is a specific and potent inhibitor of the platelet receptor glycoprotein
NURSING DEPARTMENT CRITICAL CARE POLICY MANUAL CRITICAL CARE S EPTIFIBATIDE (INTEGRILIN) I. PURPOSE: A. Integrilin (Eptifibatide) is a specific and potent inhibitor of the platelet receptor glycoprotein
NEW ZEALAND DATA SHEET INTEGRILIN SOLUTION FOR INJECTION. INTEGRILIN Eptifibatide (20mg in 10mL and 75mg in 100mL) solution for injection.
 NEW ZEALAND DATA SHEET INTEGRILIN SOLUTION FOR INJECTION NAME OF MEDICINE INTEGRILIN Eptifibatide (20mg in 10mL and 75mg in 100mL) solution for injection. PRESENTATION INTEGRILIN is a clear, colourless
NEW ZEALAND DATA SHEET INTEGRILIN SOLUTION FOR INJECTION NAME OF MEDICINE INTEGRILIN Eptifibatide (20mg in 10mL and 75mg in 100mL) solution for injection. PRESENTATION INTEGRILIN is a clear, colourless
PACKAGE LEAFLET: INFORMATION FOR THE USER. Aggrastat (250 micrograms/ml) concentrate for solution for infusion tirofiban
 PACKAGE LEAFLET: INFORMATION FOR THE USER Aggrastat (250 micrograms/ml) concentrate for solution for infusion tirofiban Read all of this leaflet carefully before you start using this medicine because it
PACKAGE LEAFLET: INFORMATION FOR THE USER Aggrastat (250 micrograms/ml) concentrate for solution for infusion tirofiban Read all of this leaflet carefully before you start using this medicine because it
AGGRASTAT Solution for Infusion
 PRODUCT MONOGRAPH AGGRASTAT Solution for Infusion tirofiban hydrochloride injection 5 mg/100 ml in 250 ml bags Platelet aggregation inhibitor Correvio (UK) Ltd. Lakeside House 1 Furzeground Way, Stockley
PRODUCT MONOGRAPH AGGRASTAT Solution for Infusion tirofiban hydrochloride injection 5 mg/100 ml in 250 ml bags Platelet aggregation inhibitor Correvio (UK) Ltd. Lakeside House 1 Furzeground Way, Stockley
*Sections or subsections omitted from the full prescribing information are not listed.
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use GIAPREZA TM safely and effectively. See full prescribing information for GIAPREZA. GIAPREZA (angiotensin
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use GIAPREZA TM safely and effectively. See full prescribing information for GIAPREZA. GIAPREZA (angiotensin
NURSING DEPARTMENT CRITICAL CARE POLICY MANUAL CRITICAL CARE PROTOCOLS. ACTIVASE (t-pa) INFUSION PROTOCOL FOR ACUTE MYOCARDIAL INFARCTION
 NURSING DEPARTMENT CRITICAL CARE POLICY MANUAL CRITICAL CARE PROTOCOLS ACTIVASE (t-pa) FOR ACUTE MYOCARDIAL INFARCTION I. PURPOSE: A. To reduce the extent of myocardial infarction by lysing the clot in
NURSING DEPARTMENT CRITICAL CARE POLICY MANUAL CRITICAL CARE PROTOCOLS ACTIVASE (t-pa) FOR ACUTE MYOCARDIAL INFARCTION I. PURPOSE: A. To reduce the extent of myocardial infarction by lysing the clot in
Appendix IV - Prescribing Guidance for Apixaban
 Appendix IV - Prescribing Guidance for Apixaban Patient Factors Dose of Apixaban If your patient has any of the following MAJOR risk factors: Hypersensitivity to the active substance or to any of the excipients
Appendix IV - Prescribing Guidance for Apixaban Patient Factors Dose of Apixaban If your patient has any of the following MAJOR risk factors: Hypersensitivity to the active substance or to any of the excipients
Package leaflet: Information for the user. ReoPro 2 mg/ml solution for injection or infusion. abciximab
 Package leaflet: Information for the user ReoPro 2 mg/ml solution for injection or infusion abciximab Read all of this leaflet carefully before you start using this medicine because it contains important
Package leaflet: Information for the user ReoPro 2 mg/ml solution for injection or infusion abciximab Read all of this leaflet carefully before you start using this medicine because it contains important
PACKAGE LEAFLET: INFORMATION FOR THE USER. Aggrastat (50 micrograms/ml) solution for infusion tirofiban
 PACKAGE LEAFLET: INFORMATION FOR THE USER Aggrastat (50 micrograms/ml) solution for infusion tirofiban Read all of this leaflet carefully before you start using this medicine because it contains important
PACKAGE LEAFLET: INFORMATION FOR THE USER Aggrastat (50 micrograms/ml) solution for infusion tirofiban Read all of this leaflet carefully before you start using this medicine because it contains important
(angiotensin II) injection for intravenous infusion
 ADMINISTERING GIAPREZA TM (angiotensin II) injection for intravenous infusion Visit www.giapreza.com INITIATE Recommended starting dose of GIAPREZA is 20 ng/kg/min, which is equivalent to 0.02 mcg/kg/min
ADMINISTERING GIAPREZA TM (angiotensin II) injection for intravenous infusion Visit www.giapreza.com INITIATE Recommended starting dose of GIAPREZA is 20 ng/kg/min, which is equivalent to 0.02 mcg/kg/min
ROSOBAC-1GM / ROSOBAC-FORT
 ROSOBAC-1GM / ROSOBAC-FORT ROSOBAC - 1GM. COMPOSITION : Each vial contains Sterile Cefoperazone Sodium IP Eq. to Anhydrous Cefoperazone - Sterile Sulbactam Sodium USP Eq. to Anhydrous Sulbactam - ROSOBAC
ROSOBAC-1GM / ROSOBAC-FORT ROSOBAC - 1GM. COMPOSITION : Each vial contains Sterile Cefoperazone Sodium IP Eq. to Anhydrous Cefoperazone - Sterile Sulbactam Sodium USP Eq. to Anhydrous Sulbactam - ROSOBAC
LACIPIL QUALITATIVE AND QUANTITATIVE COMPOSITION
 LACIPIL lacidipine QUALITATIVE AND QUANTITATIVE COMPOSITION Lacidipine, 2 mg - round shaped white engraved on one face. Lacidipine, 4 mg - oval white with break line on both faces. Lacidipine, 6 mg - oval
LACIPIL lacidipine QUALITATIVE AND QUANTITATIVE COMPOSITION Lacidipine, 2 mg - round shaped white engraved on one face. Lacidipine, 4 mg - oval white with break line on both faces. Lacidipine, 6 mg - oval
Farmadol. Paracetamol 10 mg/ml INFUSION SOLUTION
 Farmadol Paracetamol 10 mg/ml INFUSION SOLUTION Composition Each ml contains: Paracetamol 10 mg Pharmacology Pharmacodynamic properties The precise mechanism of the analgesic and antipyretic properties
Farmadol Paracetamol 10 mg/ml INFUSION SOLUTION Composition Each ml contains: Paracetamol 10 mg Pharmacology Pharmacodynamic properties The precise mechanism of the analgesic and antipyretic properties
PRODUCT INFORMATION. Colistin Link. Colistin 150 mg/2 ml (as colistimethate sodium) powder for injection vial
 PRODUCT INFORMATION Colistin Link Colistin 150 mg/2 ml (as colistimethate sodium) powder for injection vial For Intramuscular and Intravenous use. NAME OF THE MEDICINE Colistimethate sodium for injection,
PRODUCT INFORMATION Colistin Link Colistin 150 mg/2 ml (as colistimethate sodium) powder for injection vial For Intramuscular and Intravenous use. NAME OF THE MEDICINE Colistimethate sodium for injection,
PHENTOLAMINE MESYLATE INJECTION SANDOZ STANDARD 5 mg/ ml THERAPEUTIC CLASSIFICATION Alpha-adrenoreceptor Blocker
 PACKAGE INSERT Pr PHENTOLAMINE MESYLATE INJECTION SANDOZ STANDARD 5 mg/ ml THERAPEUTIC CLASSIFICATION Alpha-adrenoreceptor Blocker ACTIONS AND CLINICAL PHARMACOLOGY Phentolamine produces an alpha-adrenergic
PACKAGE INSERT Pr PHENTOLAMINE MESYLATE INJECTION SANDOZ STANDARD 5 mg/ ml THERAPEUTIC CLASSIFICATION Alpha-adrenoreceptor Blocker ACTIONS AND CLINICAL PHARMACOLOGY Phentolamine produces an alpha-adrenergic
DBL NALOXONE HYDROCHLORIDE INJECTION USP
 Name of medicine Naloxone hydrochloride Data Sheet New Zealand DBL NALXNE HYDRCHLRIDE INJECTIN USP Presentation DBL Naloxone Hydrochloride Injection USP is a sterile, clear, colourless solution, free from
Name of medicine Naloxone hydrochloride Data Sheet New Zealand DBL NALXNE HYDRCHLRIDE INJECTIN USP Presentation DBL Naloxone Hydrochloride Injection USP is a sterile, clear, colourless solution, free from
Inhixa (Enoxaparin Sodium)
 Inhixa (Enoxaparin Sodium) P R E V ENTIS SAFETY D E V I C E P R E V E N T I S I S A N AU TO M AT I C N E E D L E S H I E L D I N G S Y S T E M, W H I C H H A S A C O V E R T H AT E X T E N D S O V E R
Inhixa (Enoxaparin Sodium) P R E V ENTIS SAFETY D E V I C E P R E V E N T I S I S A N AU TO M AT I C N E E D L E S H I E L D I N G S Y S T E M, W H I C H H A S A C O V E R T H AT E X T E N D S O V E R
PHARMACEUTICAL INFORMATION AZILSARTAN
 AZEARLY Tablets Each Tablet Contains Azilsartan 20/40/80 mg PHARMACEUTICAL INFORMATION AZILSARTAN Generic name: Azilsartan Chemical name: 2-Ethoxy-1-{[2'-(5-oxo-2,5-dihydro-1,2,4-oxadiazol-3-yl)-4-biphenylyl]methyl}-
AZEARLY Tablets Each Tablet Contains Azilsartan 20/40/80 mg PHARMACEUTICAL INFORMATION AZILSARTAN Generic name: Azilsartan Chemical name: 2-Ethoxy-1-{[2'-(5-oxo-2,5-dihydro-1,2,4-oxadiazol-3-yl)-4-biphenylyl]methyl}-
CLINICAL GUIDELINES ID TAG
 Title: Author: Speciality/ Division: Directorate: Date Uploaded: Review Date: September 2019 CG ID TAG CG0423 CLINICAL GUIDELINES ID TAG Clinical Guideline for Alteplase in intra-arterial thrombolysis
Title: Author: Speciality/ Division: Directorate: Date Uploaded: Review Date: September 2019 CG ID TAG CG0423 CLINICAL GUIDELINES ID TAG Clinical Guideline for Alteplase in intra-arterial thrombolysis
BIVASTAT Injection (Bivalirudin)
 Published on: 1 Aug 2014 BIVASTAT Injection (Bivalirudin) Compostion BIVASTAT Each vial contains: Bivalirudin.. 250 mg Excipients.q.s. Supplied with 5 ml Sterile Water for Injection, IP Dosage Form Sterile
Published on: 1 Aug 2014 BIVASTAT Injection (Bivalirudin) Compostion BIVASTAT Each vial contains: Bivalirudin.. 250 mg Excipients.q.s. Supplied with 5 ml Sterile Water for Injection, IP Dosage Form Sterile
New Zealand Data Sheet COLISTIN-LINK
 New Zealand Data Sheet COLISTIN-LINK Colistimethate sodium equivalent to colistin 150mg powder for injection For intramuscular and intravenous use. Description Colistimethate sodium for injection, USP
New Zealand Data Sheet COLISTIN-LINK Colistimethate sodium equivalent to colistin 150mg powder for injection For intramuscular and intravenous use. Description Colistimethate sodium for injection, USP
BECLOT 500MG. INJECTION Composition : Each ml. contains : Tranexamic Acid I.P. 100mg.
 BECLOT 500MG. INJECTION Composition : Each ml. contains : Tranexamic Acid I.P. 100mg. DESCRIPTION Tranexamic acid is a synthetic analog of the amino acid lysine. It is used to treat or prevent excessive
BECLOT 500MG. INJECTION Composition : Each ml. contains : Tranexamic Acid I.P. 100mg. DESCRIPTION Tranexamic acid is a synthetic analog of the amino acid lysine. It is used to treat or prevent excessive
CONTRAINDICATIONS _ Major bleeding (4) History of hypersensitivity to argatroban (4)
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use Argatroban Injection safely and effectively. See full prescribing information for Argatroban Injection.
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use Argatroban Injection safely and effectively. See full prescribing information for Argatroban Injection.
PRODUCT INFORMATION UROCIT -K. Wax matrix tablets
 PRODUCT INFORMATION UROCIT -K Wax matrix tablets NAME OF DRUG Potassium Citrate CAS number- 6100-05-6 The empirical formula of potassium citrate is K 3 C 6 H 5 0 7.H 2 O, and its structural formula is:
PRODUCT INFORMATION UROCIT -K Wax matrix tablets NAME OF DRUG Potassium Citrate CAS number- 6100-05-6 The empirical formula of potassium citrate is K 3 C 6 H 5 0 7.H 2 O, and its structural formula is:
DOSAGE FORMS AND STRENGTHS
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use ARGATROBAN INJECTION safely and effectively. See full prescribing information for ARGATROBAN INJECTION.
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use ARGATROBAN INJECTION safely and effectively. See full prescribing information for ARGATROBAN INJECTION.
DBL MAGNESIUM SULFATE CONCENTRATED INJECTION
 DBL MAGNESIUM SULFATE CONCENTRATED INJECTION NAME OF MEDICINE Magnesium Sulfate BP DESCRIPTION DBL Magnesium Sulfate Concentrated Injection is a clear, colourless, sterile solution. Each ampoule contains
DBL MAGNESIUM SULFATE CONCENTRATED INJECTION NAME OF MEDICINE Magnesium Sulfate BP DESCRIPTION DBL Magnesium Sulfate Concentrated Injection is a clear, colourless, sterile solution. Each ampoule contains
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS
 ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1 1. NAME OF THE MEDICINAL PRODUCT INTEGRILIN 0.75 mg/ml solution for infusion 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each ml of solution for infusion contains
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1 1. NAME OF THE MEDICINAL PRODUCT INTEGRILIN 0.75 mg/ml solution for infusion 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each ml of solution for infusion contains
Role of Clopidogrel in Acute Coronary Syndromes. Hossam Kandil,, MD. Professor of Cardiology Cairo University
 Role of Clopidogrel in Acute Coronary Syndromes Hossam Kandil,, MD Professor of Cardiology Cairo University ACS Treatment Strategies Reperfusion/Revascularization Therapy Thrombolysis PCI (with/ without
Role of Clopidogrel in Acute Coronary Syndromes Hossam Kandil,, MD Professor of Cardiology Cairo University ACS Treatment Strategies Reperfusion/Revascularization Therapy Thrombolysis PCI (with/ without
Amlodipine plus Lisinopril Tablets AMLOPRES-L
 Amlodipine plus Lisinopril Tablets AMLOPRES-L COMPOSITION AMLOPRES-L Each uncoated tablet contains: Amlodipine besylate equivalent to Amlodipine 5 mg and Lisinopril USP equivalent to Lisinopril (anhydrous)
Amlodipine plus Lisinopril Tablets AMLOPRES-L COMPOSITION AMLOPRES-L Each uncoated tablet contains: Amlodipine besylate equivalent to Amlodipine 5 mg and Lisinopril USP equivalent to Lisinopril (anhydrous)
PRODUCT MONOGRAPH. Pr INTEGRILIN. Eptifibatide Injection Intravenous Solution. 2 mg/ml bolus Injection 0.75 mg/ml Injection (as infused)
 PRODUCT MONOGRAPH Pr INTEGRILIN Injection Intravenous Solution 2 mg/ml bolus Injection 0.75 mg/ml Injection (as infused) Platelet Aggregation Inhibitor Merck Canada Inc. Date of Preparation: 16750 route
PRODUCT MONOGRAPH Pr INTEGRILIN Injection Intravenous Solution 2 mg/ml bolus Injection 0.75 mg/ml Injection (as infused) Platelet Aggregation Inhibitor Merck Canada Inc. Date of Preparation: 16750 route
2 QUALITATIVE AND QUANTITATIVE COMPOSITION
 1 PRODUCT NAME TROPISETRON-AFT tropisetron hydrochloride (equivalent to 2 mg or 5 mg tropisetron) per ampoule. 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each ml contains 1 mg of tropisetron 1 2 ml ampoule
1 PRODUCT NAME TROPISETRON-AFT tropisetron hydrochloride (equivalent to 2 mg or 5 mg tropisetron) per ampoule. 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each ml contains 1 mg of tropisetron 1 2 ml ampoule
PACKAGE INSERT TEMPLATE FOR SALBUTAMOL TABLET & SALBUTAMOL SYRUP
 PACKAGE INSERT TEMPLATE FOR SALBUTAMOL TABLET & SALBUTAMOL SYRUP Brand or Product Name [Product name] Tablet 2mg [Product name] Tablet 4mg [Product name] Syrup 2mg/5ml Name and Strength of Active Substance(s)
PACKAGE INSERT TEMPLATE FOR SALBUTAMOL TABLET & SALBUTAMOL SYRUP Brand or Product Name [Product name] Tablet 2mg [Product name] Tablet 4mg [Product name] Syrup 2mg/5ml Name and Strength of Active Substance(s)
Diagnosis and Management of Acute Myocardial Infarction
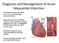 Diagnosis and Management of Acute Myocardial Infarction Acute Myocardial Infarction (AMI) occurs as a result of prolonged myocardial ischemia Atherosclerosis leads to endothelial rupture or erosion that
Diagnosis and Management of Acute Myocardial Infarction Acute Myocardial Infarction (AMI) occurs as a result of prolonged myocardial ischemia Atherosclerosis leads to endothelial rupture or erosion that
ReoPro (abciximab rmc)
 ReoPro (abciximab rmc) NAME OF THE MEDICINE ReoPro (abciximab) The active ingredient is abciximab. DESCRIPTION ReoPro (abciximab rmc) is the Fab fragment of the chimeric monoclonal antibody 7E3 (c7e3 Fab).
ReoPro (abciximab rmc) NAME OF THE MEDICINE ReoPro (abciximab) The active ingredient is abciximab. DESCRIPTION ReoPro (abciximab rmc) is the Fab fragment of the chimeric monoclonal antibody 7E3 (c7e3 Fab).
PRODUCT INFORMATION. DESCRIPTION Addaven is a concentrated trace element solution for infusion which is clear and colourless to slightly yellow.
 PRODUCT INFORMATION Addaven NAME OF MEDICINE Addaven is a concentrated trace element solution containing the following simple salts: chromic chloride hexahydrate (CAS No.:10060-12-5), cupric chloride dihydrate
PRODUCT INFORMATION Addaven NAME OF MEDICINE Addaven is a concentrated trace element solution containing the following simple salts: chromic chloride hexahydrate (CAS No.:10060-12-5), cupric chloride dihydrate
2.2 Dosing in Patients with Heparin-Induced Thrombocytopenia. 2.3 Dosing in Patients Undergoing Percutaneous Coronary Intervention
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use ARGATROBAN INJECTION safely and effectively. See full prescribing information for ARGATROBAN INJECTION.
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use ARGATROBAN INJECTION safely and effectively. See full prescribing information for ARGATROBAN INJECTION.
Elements for a public summary. VI.2.1 Overview of disease epidemiology
 VI.2 Elements for a public summary VI.2.1 Overview of disease epidemiology Coronary artery disease and as anticoagulant (inhibiting the clotting of the blood) in patients undergoing surgery to treat blockages
VI.2 Elements for a public summary VI.2.1 Overview of disease epidemiology Coronary artery disease and as anticoagulant (inhibiting the clotting of the blood) in patients undergoing surgery to treat blockages
PRODUCT INFORMATION. NAME OF THE MEDICINE Compound Sodium Lactate (Hartmann's) Solution for Injection
 PRODUCT INFORMATION NAME OF THE MEDICINE Compound Sodium Lactate (Hartmann's) Solution for Injection DESCRIPTION Molecular formulae. Potassium chloride: KCl; sodium chloride: NaCl; calcium chloride dihydrate:
PRODUCT INFORMATION NAME OF THE MEDICINE Compound Sodium Lactate (Hartmann's) Solution for Injection DESCRIPTION Molecular formulae. Potassium chloride: KCl; sodium chloride: NaCl; calcium chloride dihydrate:
Major bleeding (4) History of hypersensitivity to this product (4)
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use ARGATROBAN INJECTION safely and effectively. See full prescribing information for ARGATROBAN INJECTION.
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use ARGATROBAN INJECTION safely and effectively. See full prescribing information for ARGATROBAN INJECTION.
azilsartan medoxomil
 azilsartan medoxomil edarbi 40mg Tablet 80mg Tablet ANTIHYPERTENSIVE Angiotensin II Receptor Antagonist FORMULATION: Each tablet contains 40mg Azilsartan medoxomil (as potassium) Each tablet contains 80mg
azilsartan medoxomil edarbi 40mg Tablet 80mg Tablet ANTIHYPERTENSIVE Angiotensin II Receptor Antagonist FORMULATION: Each tablet contains 40mg Azilsartan medoxomil (as potassium) Each tablet contains 80mg
DATA SHEET. REOPRO vial 5 ml contains 10 mg abciximab rmc. REOPRO is a clear, colourless, sterile solution for intravenous use.
 DATA SHEET REOPRO Abciximab rmc, solution for injection, vials 2 mg/ml. Presentation REOPRO vial 5 ml contains 10 mg abciximab rmc. REOPRO is a clear, colourless, sterile solution for intravenous use.
DATA SHEET REOPRO Abciximab rmc, solution for injection, vials 2 mg/ml. Presentation REOPRO vial 5 ml contains 10 mg abciximab rmc. REOPRO is a clear, colourless, sterile solution for intravenous use.
Thrombolysis administration
 Thrombolysis administration Liz Mackey Stroke Nurse Practitioner Western Health Sunshine & Footscray Hospital, Melbourne Thanks ASNEN committee members Skye Coote, Acute Stroke Nurse, Eastern Health (slide
Thrombolysis administration Liz Mackey Stroke Nurse Practitioner Western Health Sunshine & Footscray Hospital, Melbourne Thanks ASNEN committee members Skye Coote, Acute Stroke Nurse, Eastern Health (slide
Reference ID:
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use TRIFERIC safely and effectively. See full prescribing information for TRIFERIC. TRIFERIC (ferric
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use TRIFERIC safely and effectively. See full prescribing information for TRIFERIC. TRIFERIC (ferric
Angina pectoris due to coronary atherosclerosis : Atenolol is indicated for the long term management of patients with angina pectoris.
 Lonet Tablet Description Lonet contains Atenolol, a synthetic β1 selective (cardioselective) adrenoreceptor blocking agent without membrane stabilising or intrinsic sympathomimetic (partial agonist) activity.
Lonet Tablet Description Lonet contains Atenolol, a synthetic β1 selective (cardioselective) adrenoreceptor blocking agent without membrane stabilising or intrinsic sympathomimetic (partial agonist) activity.
50% Concentrated Injection
 NAME OF THE MEDICINE. The molecular weight of the compound is 246.5 and the CAS registry number is 10034-99-8. The molecular formula is MgSO4, 7H2O. DESCRIPTION MAGNESIUM SULFATE HEPTAHYDRATE 50% CONCENTRATED
NAME OF THE MEDICINE. The molecular weight of the compound is 246.5 and the CAS registry number is 10034-99-8. The molecular formula is MgSO4, 7H2O. DESCRIPTION MAGNESIUM SULFATE HEPTAHYDRATE 50% CONCENTRATED
BRICANYL INJECTION. terbutaline sulfate PRODUCT INFORMATION
 BRICANYL INJECTION terbutaline sulfate PRODUCT INFORMATION NAME OF THE MEDICINE Terbutaline sulfate, 2-(tert-butylamino)-1-(3,5-dihydroxyphenyl) ethanol sulfate, a sympathomimetic bronchodilator with a
BRICANYL INJECTION terbutaline sulfate PRODUCT INFORMATION NAME OF THE MEDICINE Terbutaline sulfate, 2-(tert-butylamino)-1-(3,5-dihydroxyphenyl) ethanol sulfate, a sympathomimetic bronchodilator with a
ARGATROBAN Injection PRESCRIBING INFORMATION
 ARGATROBAN Injection PRESCRIBING INFORMATION DESCRIPTION Argatroban is a synthetic direct thrombin inhibitor derived from L-arginine. The chemical name for Argatroban is 1-[5-[(aminoiminomethyl)amino]-1-oxo-2-[[(1,2,3,4-tetrahydro-3-
ARGATROBAN Injection PRESCRIBING INFORMATION DESCRIPTION Argatroban is a synthetic direct thrombin inhibitor derived from L-arginine. The chemical name for Argatroban is 1-[5-[(aminoiminomethyl)amino]-1-oxo-2-[[(1,2,3,4-tetrahydro-3-
1 INDICATIONS AND USAGE. 1.1 Limitation of Use FULL PRESCRIBING INFORMATION
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use TRIFERIC safely and effectively. See full prescribing information for TRIFERIC. TRIFERIC (ferric
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use TRIFERIC safely and effectively. See full prescribing information for TRIFERIC. TRIFERIC (ferric
Protamine sulphate LEO Pharma 1400 anti-heparin IU/ml solution for injection and infusion.
 1. NAME OF THE MEDICINAL PRODUCT Protamine sulphate LEO Pharma 1400 anti-heparin IU/ml solution for injection and infusion. 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Protamine sulphate 1400 anti-heparin
1. NAME OF THE MEDICINAL PRODUCT Protamine sulphate LEO Pharma 1400 anti-heparin IU/ml solution for injection and infusion. 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Protamine sulphate 1400 anti-heparin
GALLIUM CITRATE Ga 67 INJECTION
 511945-0903 September 2003 USA Bristol-Myers Squibb Medical Imaging 331 Treble Cove Road N. Billerica, MA 01862 USA GALLIUM CITRATE Ga 67 INJECTION FOR DIAGNOSTIC USE DESCRIPTION: Gallium Citrate Ga 67
511945-0903 September 2003 USA Bristol-Myers Squibb Medical Imaging 331 Treble Cove Road N. Billerica, MA 01862 USA GALLIUM CITRATE Ga 67 INJECTION FOR DIAGNOSTIC USE DESCRIPTION: Gallium Citrate Ga 67
ELSPAR (asparaginase) For injection, intravenous or intramuscular Initial U.S. Approval: 1978
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use Elspar safely and effectively. See full prescribing information for Elspar. ELSPAR (asparaginase)
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use Elspar safely and effectively. See full prescribing information for Elspar. ELSPAR (asparaginase)
TRANSPARENCY COMMITTEE OPINION. 4 November 2009
 The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 4 November 2009 RANEXA 375 mg extended release tablet Pack of 60 (CIP: 394 370-7) RANEXA 500 mg extended release tablet
The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 4 November 2009 RANEXA 375 mg extended release tablet Pack of 60 (CIP: 394 370-7) RANEXA 500 mg extended release tablet
0BCore Safety Profile. Pharmaceutical form(s)/strength: Film-coated tablet 40, 80, 160, 320 mg SE/H/PSUR/0024/003 Date of FAR:
 0BCore Safety Profile Active substance: Valsartan Pharmaceutical form(s)/strength: Film-coated tablet 40, 80, 160, 320 mg P-RMS: SE/H/PSUR/0024/003 Date of FAR: 28.02.2013 4.2 Posology and method of administration
0BCore Safety Profile Active substance: Valsartan Pharmaceutical form(s)/strength: Film-coated tablet 40, 80, 160, 320 mg P-RMS: SE/H/PSUR/0024/003 Date of FAR: 28.02.2013 4.2 Posology and method of administration
Younger adults with a family history of premature artherosclerotic disease should have their cardiovascular risk factors measured.
 Appendix 2A - Guidance on Management of Hypertension Measurement of blood pressure All adults from 40 years should have blood pressure measured as part of opportunistic cardiovascular risk assessment.
Appendix 2A - Guidance on Management of Hypertension Measurement of blood pressure All adults from 40 years should have blood pressure measured as part of opportunistic cardiovascular risk assessment.
Composition Each ml of Ventol solution for inhalation contains 5 mg Salbutamol (as sulphate).
 VENTOL Composition Each ml of Ventol solution for inhalation contains 5 mg Salbutamol (as sulphate). Respiratory Solution Action Salbutamol is a short-acting, relatively selective beta2-adrenoceptor agonist.
VENTOL Composition Each ml of Ventol solution for inhalation contains 5 mg Salbutamol (as sulphate). Respiratory Solution Action Salbutamol is a short-acting, relatively selective beta2-adrenoceptor agonist.
PRODUCT INFORMATION. Sudafed* Sinus + Anti-inflammatory Pain Relief Caplets
 PRODUCT INFORMATION Sudafed* Sinus + Anti-inflammatory Pain Relief Caplets Product description Sudafed* Sinus + Anti-inflammatory Pain Relief caplets contain pseudoephedrine hydrochloride 30 mg and ibuprofen
PRODUCT INFORMATION Sudafed* Sinus + Anti-inflammatory Pain Relief Caplets Product description Sudafed* Sinus + Anti-inflammatory Pain Relief caplets contain pseudoephedrine hydrochloride 30 mg and ibuprofen
* Adults. NSAID associated peptic ulceration: - Acute treatment: 150 mg twice daily for 8 to 12 weeks, or 300mg nocte.
 Trade Name Aciloc 75 mg & 300 mg Film-coated tablets Generic Name Ranitidine Composition Each Aciloc 300 mg film-coated tablet contains: - Active ingredient: Ranitidine hydrochloride 336 mg equivalent
Trade Name Aciloc 75 mg & 300 mg Film-coated tablets Generic Name Ranitidine Composition Each Aciloc 300 mg film-coated tablet contains: - Active ingredient: Ranitidine hydrochloride 336 mg equivalent
PRODUCT INFORMATION RESONIUM A. Na m
 PRODUCT INFORMATION RESONIUM A NAME OF THE MEDICINE Non-proprietary Name Sodium polystyrene sulfonate Chemical Structure CH - 2 CH SO 3 Na + n CAS Number 28210-41-5 [9003-59-2] CH 2 CH SO - 3 m DESCRIPTION
PRODUCT INFORMATION RESONIUM A NAME OF THE MEDICINE Non-proprietary Name Sodium polystyrene sulfonate Chemical Structure CH - 2 CH SO 3 Na + n CAS Number 28210-41-5 [9003-59-2] CH 2 CH SO - 3 m DESCRIPTION
Granisetron Kabi, 1mg/ml, concentrate for solution for injection/infusion
 SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Granisetron Kabi, 1mg/ml, concentrate for solution for injection/infusion 2 QUALITATIVE AND QUANTITATIVE COMPOSITION 1 ml contains granisetron
SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Granisetron Kabi, 1mg/ml, concentrate for solution for injection/infusion 2 QUALITATIVE AND QUANTITATIVE COMPOSITION 1 ml contains granisetron
*Sections or subsections omitted from the full prescribing information are not listed.
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use ARGATROBAN INJECTION safely and effectively. See full prescribing information for ARGATROBAN INJECTION.
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use ARGATROBAN INJECTION safely and effectively. See full prescribing information for ARGATROBAN INJECTION.
NURSING DEPARTMENT CRITICAL CARE POLICY MANUAL CRITICAL CARE PROTOCOLS. ACUTE CEREBROVASCULAR ACCIDENT TPA (ACTIVASE /alteplase) FOR THROMBOLYSIS
 NURSING DEPARTMENT CRITICAL CARE POLICY MANUAL CRITICAL CARE PROTOCOLS ACUTE CEREBROVASCULAR ACCIDENT TPA (ACTIVASE /alteplase) FOR THROMBOLYSIS I. Purpose : A. To reduce morbidity and mortality associated
NURSING DEPARTMENT CRITICAL CARE POLICY MANUAL CRITICAL CARE PROTOCOLS ACUTE CEREBROVASCULAR ACCIDENT TPA (ACTIVASE /alteplase) FOR THROMBOLYSIS I. Purpose : A. To reduce morbidity and mortality associated
For the use of a Registered Medical Practitioner or a Hospital or a Laboratory only.
 For the use of a Registered Medical Practitioner or a Hospital or a Laboratory only. GRANIZ (Granisetron Injection) Compostion : Each ml contains : Granisetron Hydrochloride BP equivalent to Granisetron...1
For the use of a Registered Medical Practitioner or a Hospital or a Laboratory only. GRANIZ (Granisetron Injection) Compostion : Each ml contains : Granisetron Hydrochloride BP equivalent to Granisetron...1
PRESCRIBING INFORMATION. PENTASPAN* (10% Pentastarch in 0.9% Sodium Chloride Injection) Injection THERAPEUTIC CLASSIFICATION. Plasma Volume Expander
 PRESCRIBING INFORMATION PENTASPAN* (10% Pentastarch in 0.9% Sodium Chloride Injection) Injection THERAPEUTIC CLASSIFICATION Plasma Volume Expander ACTION AND CLINICAL PHARMACOLOGY The colloidal properties
PRESCRIBING INFORMATION PENTASPAN* (10% Pentastarch in 0.9% Sodium Chloride Injection) Injection THERAPEUTIC CLASSIFICATION Plasma Volume Expander ACTION AND CLINICAL PHARMACOLOGY The colloidal properties
Meningococcal polysaccharide vaccine Group A, Group C, Group Y and Group W-135
 MENOMUNE ACYW-135 Meningococcal polysaccharide vaccine Group A, Group C, Group Y and Group W-135 Description The vaccine is a freeze-dried preparation of the group specific antigens from Neisseria meningitidis,
MENOMUNE ACYW-135 Meningococcal polysaccharide vaccine Group A, Group C, Group Y and Group W-135 Description The vaccine is a freeze-dried preparation of the group specific antigens from Neisseria meningitidis,
PRODUCT MONOGRAPH. fondaparinux sodium injection. 2.5 mg/0.5 ml 5.0 mg/0.4 ml 7.5 mg/0.6 ml 10.0 mg/0.8 ml. ATC Classification: B01AX05
 PRODUCT MONOGRAPH Pr ARIXTRA fondaparinux sodium injection 2.5 mg/0.5 ml 5.0 mg/0.4 ml 7.5 mg/0.6 ml 10.0 mg/0.8 ml ATC Classification: B01AX05 Synthetic Antithrombotic Aspen Pharmacare Canada Inc 111
PRODUCT MONOGRAPH Pr ARIXTRA fondaparinux sodium injection 2.5 mg/0.5 ml 5.0 mg/0.4 ml 7.5 mg/0.6 ml 10.0 mg/0.8 ml ATC Classification: B01AX05 Synthetic Antithrombotic Aspen Pharmacare Canada Inc 111
SUMMARY OF PRODUCT CHARACTERISTICS. Albuman 40 g/l is a solution containing 40 g/l (4%) of total protein of which at least 95% is human albumin.
 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Albuman 40 g/l solution for infusion 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Albuman 40 g/l is a solution containing 40 g/l (4%)
SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Albuman 40 g/l solution for infusion 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Albuman 40 g/l is a solution containing 40 g/l (4%)
Powder for reconstitution for use in central venous access devices
 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 27 28 29 30 Cathflo Activase (Alteplase) Powder for reconstitution for use in central venous access devices DESCRIPTION Cathflo Activase
1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 27 28 29 30 Cathflo Activase (Alteplase) Powder for reconstitution for use in central venous access devices DESCRIPTION Cathflo Activase
METALYSE Tenecteplase
 METALYSE Tenecteplase Consumer Medicine Information What is in this leaflet This leaflet answers some common questions about Metalyse. It does not contain all the available information. It does not take
METALYSE Tenecteplase Consumer Medicine Information What is in this leaflet This leaflet answers some common questions about Metalyse. It does not contain all the available information. It does not take
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use BIVALIRUDIN safely and effectively. See full prescribing information for BIVALIRUDIN. BIVALIRUDIN
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use BIVALIRUDIN safely and effectively. See full prescribing information for BIVALIRUDIN. BIVALIRUDIN
SUMMARY OF PRODUCT CHARACTERISTICS
 MUTUAL RECOGNITION PROCEDURE Page 1 of 5 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT, syrup 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each ml of syrup contains 1 mg loratadine.
MUTUAL RECOGNITION PROCEDURE Page 1 of 5 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT, syrup 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each ml of syrup contains 1 mg loratadine.
Bivalirudin APOTEX Powder for Injection
 Bivalirudin APOTEX Powder for Injection NAME OF THE MEDICINE Bivalirudin Chemical Name: D-phenylalanyl-L-prolyl-L-arginyl-L-prolyl-glycyl-glycyl-glycyl-glycyl-Lasparagyl-glycyl-L-α-aspartyl-L-phenylalanyl-L-α-glutamyl-L-α-glutamyl-Lisoleucyl-L-prolyL-L-α-glutamyl-L-α-glutamyl-L-tyrosyl-L-leucine
Bivalirudin APOTEX Powder for Injection NAME OF THE MEDICINE Bivalirudin Chemical Name: D-phenylalanyl-L-prolyl-L-arginyl-L-prolyl-glycyl-glycyl-glycyl-glycyl-Lasparagyl-glycyl-L-α-aspartyl-L-phenylalanyl-L-α-glutamyl-L-α-glutamyl-Lisoleucyl-L-prolyL-L-α-glutamyl-L-α-glutamyl-L-tyrosyl-L-leucine
GUIDELINES FOR THE EARLY MANAGEMENT OF PATIENTS WITH ACUTE ISCHEMIC STROKE
 2018 UPDATE QUICK SHEET 2018 American Heart Association GUIDELINES FOR THE EARLY MANAGEMENT OF PATIENTS WITH ACUTE ISCHEMIC STROKE A Summary for Healthcare Professionals from the American Heart Association/American
2018 UPDATE QUICK SHEET 2018 American Heart Association GUIDELINES FOR THE EARLY MANAGEMENT OF PATIENTS WITH ACUTE ISCHEMIC STROKE A Summary for Healthcare Professionals from the American Heart Association/American
COMPANY CORE PACKAGE INSERT CCPI (PI/CORE/ENGLISH)
 COMPANY CORE PACKAGE INSERT CCPI (PI/CORE/ENGLISH) HUMAN ALBUMIN 20 % BEHRING Rev.: 05-MAR-2008 / PEI approval 26.02.08 Supersedes previous versions Rev.: 28-NOV-2007 / Adaptation to Core SPC Rev.: 02-JAN-2007
COMPANY CORE PACKAGE INSERT CCPI (PI/CORE/ENGLISH) HUMAN ALBUMIN 20 % BEHRING Rev.: 05-MAR-2008 / PEI approval 26.02.08 Supersedes previous versions Rev.: 28-NOV-2007 / Adaptation to Core SPC Rev.: 02-JAN-2007
Horizon Scanning Centre November 2012
 Horizon Scanning Centre November 2012 Cangrelor to reduce platelet aggregation and thrombosis in patients undergoing percutaneous coronary intervention99 SUMMARY NIHR HSC ID: 2424 This briefing is based
Horizon Scanning Centre November 2012 Cangrelor to reduce platelet aggregation and thrombosis in patients undergoing percutaneous coronary intervention99 SUMMARY NIHR HSC ID: 2424 This briefing is based
INSPRA 25 & 50 mg TABLETS
 INSPRA 25 & 50 mg TABLETS SCHEDULING STATUS: Schedule 4 PROPRIETARY NAMES (and dosage forms): INSPRA 25 (Tablets) INSPRA 50 (Tablets) COMPOSITION: INSPRA 25: INSPRA 50: Each tablet contains 25 mg eplerenone
INSPRA 25 & 50 mg TABLETS SCHEDULING STATUS: Schedule 4 PROPRIETARY NAMES (and dosage forms): INSPRA 25 (Tablets) INSPRA 50 (Tablets) COMPOSITION: INSPRA 25: INSPRA 50: Each tablet contains 25 mg eplerenone
Salapin: Salbutamol BP 2mg as sulphate in each 5mL of a raspberry cola flavoured, sugar free syrup.
 Salapin Salbutamol Syrup 2mg/5mL Qualitative and quantitative composition Salapin: Salbutamol BP 2mg as sulphate in each 5mL of a raspberry cola flavoured, sugar free syrup. Clinical particulars Therapeutic
Salapin Salbutamol Syrup 2mg/5mL Qualitative and quantitative composition Salapin: Salbutamol BP 2mg as sulphate in each 5mL of a raspberry cola flavoured, sugar free syrup. Clinical particulars Therapeutic
FLOLAN QUALITATIVE AND QUANTITATIVE COMPOSITION
 FLOLAN epoprostenol sodium with sterile diluent (ph 12) QUALITATIVE AND QUANTITATIVE COMPOSITION Vials containing sterile, freeze-dried epoprostenol sodium equivalent to 0.5 mg or 1.5 mg epoprostenol.
FLOLAN epoprostenol sodium with sterile diluent (ph 12) QUALITATIVE AND QUANTITATIVE COMPOSITION Vials containing sterile, freeze-dried epoprostenol sodium equivalent to 0.5 mg or 1.5 mg epoprostenol.
DATA SHEET. Product Summary. 1. Trade Name of Medicinal Product. Protamine Sulphate Injection BP. 2. Qualitative and Quantitative Composition
 DATA SHEET Product Summary 1. Trade Name of Medicinal Product Protamine Sulphate Injection BP 2. Qualitative and Quantitative Composition Protamine Sulphate 10mg/ml 3. Pharmaceutical Form Solution for
DATA SHEET Product Summary 1. Trade Name of Medicinal Product Protamine Sulphate Injection BP 2. Qualitative and Quantitative Composition Protamine Sulphate 10mg/ml 3. Pharmaceutical Form Solution for
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS
 ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1 1. NAME OF THE MEDICINAL PRODUCT Tenecteplase Boehringer Ingelheim Pharma KG 6,000 units Powder and solvent for solution for injection 2. QUALITATIVE AND QUANTITATIVE
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1 1. NAME OF THE MEDICINAL PRODUCT Tenecteplase Boehringer Ingelheim Pharma KG 6,000 units Powder and solvent for solution for injection 2. QUALITATIVE AND QUANTITATIVE
EFFIENT (prasugrel hydrochloride)
 EFFIENT (prasugrel hydrochloride) NAME OF THE MEDICINE EFFIENT (prasugrel hydrochloride) The active ingredient is prasugrel hydrochloride. DESCRIPTION Prasugrel, an adenosine diphosphate (ADP) receptor
EFFIENT (prasugrel hydrochloride) NAME OF THE MEDICINE EFFIENT (prasugrel hydrochloride) The active ingredient is prasugrel hydrochloride. DESCRIPTION Prasugrel, an adenosine diphosphate (ADP) receptor
PRODUCT CIRCULAR. Tablets COZAAR (losartan potassium) I. THERAPEUTIC CLASS II. INDICATIONS III. DOSAGE AND ADMINISTRATION PAK-CZR-T
 PRODUCT CIRCULAR Tablets I. THERAPEUTIC CLASS, the first of a new class of agents for the treatment of hypertension, is an angiotensin II receptor (type AT 1 ) antagonist. also provides a reduction in
PRODUCT CIRCULAR Tablets I. THERAPEUTIC CLASS, the first of a new class of agents for the treatment of hypertension, is an angiotensin II receptor (type AT 1 ) antagonist. also provides a reduction in
Otamixaban for non-st-segment elevation acute coronary syndrome
 Otamixaban for non-st-segment elevation acute coronary syndrome September 2011 This technology summary is based on information available at the time of research and a limited literature search. It is not
Otamixaban for non-st-segment elevation acute coronary syndrome September 2011 This technology summary is based on information available at the time of research and a limited literature search. It is not
SUMMARY OF PRODUCT CHARACTERISTICS
 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT DigiFab, 40 mg/vial digoxin immune Fab, Powder for solution for infusion 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each glass vial
SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT DigiFab, 40 mg/vial digoxin immune Fab, Powder for solution for infusion 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each glass vial
PRODUCT INFORMATION CODAPANE XTRA Paracetamol 500 mg and Codeine Phosphate 15 mg Tablets
 PRODUCT INFORMATION CODAPANE XTRA Paracetamol 500 mg and Codeine Phosphate 15 mg Tablets NAME OF THE MEDICINE Active Ingredients: Paracetamol and Codeine Phosphate Paracetamol: Molecular Formula: C 8 H
PRODUCT INFORMATION CODAPANE XTRA Paracetamol 500 mg and Codeine Phosphate 15 mg Tablets NAME OF THE MEDICINE Active Ingredients: Paracetamol and Codeine Phosphate Paracetamol: Molecular Formula: C 8 H
3. PHARMACEUTICAL FORM Solution for infusion. A clear, slightly viscous liquid; it is almost colourless, yellow, amber or green.
 1. NAME OF THE MEDICINAL PRODUCT Albutein 250 g/l, solution for infusion. 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Human albumin Albutein 250 g/l is a solution containing 250 g/l of total protein of
1. NAME OF THE MEDICINAL PRODUCT Albutein 250 g/l, solution for infusion. 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Human albumin Albutein 250 g/l is a solution containing 250 g/l of total protein of
Medicine Dr. Omed Lecture 2 Stable and Unstable Angina
 Medicine Dr. Omed Lecture 2 Stable and Unstable Angina Risk stratification in stable angina. High Risk; *post infarct angina, *poor effort tolerance, *ischemia at low workload, *left main or three vessel
Medicine Dr. Omed Lecture 2 Stable and Unstable Angina Risk stratification in stable angina. High Risk; *post infarct angina, *poor effort tolerance, *ischemia at low workload, *left main or three vessel
Clodronate BE/H/PSUR/001/001 October 2011 Agreed CSP
 Clodronate BE/H/PSUR/001/001 October 2011 Agreed CSP 4. CLINICAL PARTICULARS 4.1 Therapeutic indications Intravenous use Treatment of hypercalcemia due to malignancy. Oral use Treatment of hypercalcemia
Clodronate BE/H/PSUR/001/001 October 2011 Agreed CSP 4. CLINICAL PARTICULARS 4.1 Therapeutic indications Intravenous use Treatment of hypercalcemia due to malignancy. Oral use Treatment of hypercalcemia
Solution for cardiac perfusion in viaflex plastic container
 CARDIOPLEGIA SOLUTION A Solution for cardiac perfusion in viaflex plastic container DESCRIPTION Cardioplegia Solution A is a sterile, non-pyrogenic solution in a Viaflex bag. It is used to induce cardiac
CARDIOPLEGIA SOLUTION A Solution for cardiac perfusion in viaflex plastic container DESCRIPTION Cardioplegia Solution A is a sterile, non-pyrogenic solution in a Viaflex bag. It is used to induce cardiac
