Successes and Evolving Challenges Posed by the Comprehensive In Vitro Proarrhythmia (CiPA) Initiative
|
|
|
- Alfred Hunter
- 6 years ago
- Views:
Transcription
1 Successes and Evolving Challenges Posed by the Comprehensive In Vitro Proarrhythmia (CiPA) Initiative Gary Gintant Dept. Integrative Pharmacology Integrated Sciences and Technology AbbVie For the CiPA Initiative
2 Outline Introduction and Rationale for CiPA Definition and Overarching Goals Scientific Underpinnings Components: Progress and Challenges Ionic Currents In Silico Reconstruction Stem-Cell Derived Cardiomyocytes Compound Selection/Regulatory Paths Forward and Conclusions 2
3 CiPA: Comprehensive In Vitro Proarrhythmia Assay Goal: Develop a new in vitro paradigm for cardiac safety evaluation of new drugs that provides a more accurate and comprehensive mechanistic-based assessment of proarrhythmic potential - focus on proarrhythmia (not QT prolongation) to improve specificity compared to preclinical herg and clinical TQT studies How? - Define drug effects on multiple human cardiac currents, - Characterize integrated electrophysiologic response using in silico reconstructions of human ventricular electrophysiology, - Verify effects on human stem-cell derived ventricular myocytes 3
4 herg Is Insufficient to Predict Delayed Repolarization REPOLARIZATION: integrated response, multiple currents DELAYED REPOLARIZATION: reduced NET outward current - balance of outward repolarizing current (ikr/herg) vs. inward depolarizing plateau current (Na or Ca) Present (ikr, herg) focus results in premature and unwarranted drug attrition, misclassification of risk 4
5 Drug Effects on Multiple Cardiac Currents Must Be Considered. Lack of selectivity of drugs on cardiac currents (ICaL vs. herg) Effects on multiple channels (multi-channel block) define overall drug response (e.g., verapamil) 5
6 Cellular Proarrhythmia: Abnormal Emergent Responses That Affect Repolarization (not simply hapd or hqtc) Focus on proarrhythmic vulnerability: impaired cellular repolarization, electrical instability - In extreme form, instability manifest as early after-depolarizations (EAD s) associated with initiation of proarrhythmia and l - Proarrhythmic liability (an integrated, emergent effect) manifest on a cellular level with human responses Impaired repolarization used to rank proarrhythmic risk based on comparisons with known clinical TdP drugs 6
7 Comprehensive In Vitro Proarrhythmia Assay: Four Overall Components Drug Effects on Multiple Human Cardiac Currents In Silico Reconstruction Human Ventricular Cellular Electrophysiology In Vitro Effects Human Stem- Cell Derived Ventricular Myocytes Evaluation of Clinical Drugs for Proarrhythmic TdP Liability High Risk Intermediate Risk Low Risk Preclinical ECG & Phase 1 ECG Studies: Complementary Data 7
8 Core Component I. Human Ionic Currents, Voltage Clamp Studies, Heterologous Expression - Ion Channel Working Group (SPS): develop protocols to assess drug effects on currents - Standardized voltage clamp protocols to establish best practices, reduce bias and variability, allow comparisons of automated platforms across laboratories - Information on kinetics-, voltage- and use-dependence to parameterize models (essential for herg) for Robust characterization of drug effects on human currents enables in silico reconstructions of integrated responses 8
9 Core Component II: Computer Reconstructions of Drug Effects on Human Cellular Electrophysiology + = - In Silico Group (FDA): Drug effect multiple human currents integrated to describe cellular electrophysiologic effects - Define changes in repolarization instability, early afterdepolarizations, reduced upstroke velocity using select model (modified O Hara-Rudy model considered) - Ranking of integrated responses compared with clinical examples of different TdP liabilities 9
10 Core Component II. Computer Reconstructions: Progress (FDA Lead) Initial Test Case: d,l Sotalol (known torsadogen) - Recognized for QT prolongation and TdP - O Hara-Rudy model recapitulates delayed repolarization in human ventricular tissues based on IC 50 values for herg, Cav1.2 and Nav1.5 (Kramer et al. (2013) - Not replicated by Grandi or ten Tusscher in silico models - EAD s with prominent ikr/herg block not generated in either Ten Tusscher or Grandi in silico models 10
11 Micro-Electrode Array (MEA) Voltage-Sensitive Dye (VSO) Core Component III: In vitro Effects, Human Stem Cell-Derived Ventricular Cardiomyocytes - Myocyte Group (HESI): Verification of ion channel effects and in silico reconstructions with well characterized human stem-cell cardiomyocytes - Inform on repolarization effects not anticipated from other approaches - channel modulation - emergent effects - physiologic conditions (37 o C) - MEA & VSO approaches 11
12 Core Component III. Cardiomyocytes- Microelectrode Array (MEA) Pilot Study 12 site Pilot Study 12
13 Core Component III. Stem-cell Derived Cardiomyocytes: Progress Test Protocol: - Confluent monolayers prepared according to manufacturer instructions minutes drug exposure - Individual or sequential (cumulative) exposure - 4 concentrations; 3 wells/conc. - Quality controls: stable baseline spontaneous rate, stable conduction across monolayers, post-dose signal quality 8 compound test set (blinded) - 4 Calibration cmpds to test assay sensitivity: IKr, IKs, INaF, ICaL - 4 Test cmpds : preliminary test set, high and low risk examples Pilot Studies - Stem Cell Derived Cardiomyocytes "Calibration" Cmpd & Current "Test" Cmpds Mexiletine (ina) Flecainide Nifedipine* (ical) Moxifloxacin E-4031 (ikr) Quinidine JNJ303 (iks) Ranolazine = "High Risk" Category = "Low Risk" Category = Not Categorized, Channel Specific Prelim. analysis; concordance across VSO & MEA; review ongoing 13
14 Core Component III. Stem-cell Derived Cardiomyocytes: Progress MEA VSO 2 approaches - Multi-electrode arrays (MEA-8 sites) - Voltage-sensing optical (VSO-4 sites) - Dyes or integral sensing proteins 3 Myocyte Vendors - CDI, Axiogenesis, (Stanford University) - Myocytes prepared to vendor specifications; site familiarity 14
15 Sites A & B Cells 1 & 2 Plate 1 Pilot MEA Study: Trends Across 4 sites, 3 compounds, & 2 cells. Compounds 3, 1, 5 Cell A Cell B 15
16 Core Component III. Stem-cell Derived Cardiomyocytes. Voltage sensitive optical probes (VSO) Study Cell providers: Assay companies/groups: B I O S C I E N C E S Voltage sensor Di-4-ANEPPS (small molecule) QuasAr1 (protein) FluoVolt (small molecule) Di-4-ANEPPS (small molecule) Detector Photomultiplier CMOS camera CMOS camera CMOS camera 16
17 VSO: Endpoints Minimum end-points include measures of repolarization timings and arrhythmia detection. Primary end points: Spontaneous beat rate and regularity (adult human 1s) Baseline Drug Action potential rise time (10-90%) (adult human 2-2.5ms) APD at 30%, 50% & 90% (adult human 200ms, 220, 270) AP triangulation index (APD 90 -APD 30 ) (adult human 70ms) 200ms Incidence of early after depolarisation (EADs) Rate sensitivity of the above parameters (paced only) 17
18 APD 30 (% of baseline) APD 50 (% of baseline) APD 90 (% of baseline) Examples: Di-4-ANEPPS as Voltage Sensor C1_Baseline C1_Drug Baseline Drug Baseline Drug Baseline Drug 200ms 200ms 200ms 200ms Vehicle (DMSO) Drug Vehicle (DMSO) Drug * Vehicle (DMSO) Drug *** * *** C1 C2 C3 C4 [Drug] C1 C2 C3 C4 [Drug] C1 C2 C3 C4 [Drug] 18
19 CiPA Pilot Myocyte Workstreams: Opportunities to Assess Experimental Variability CiPA Stem Cell Cardiomyocyte Pilot Studies MicroElectrode Array (MEA) Voltage-Sensing Optical (VSO) CiPA Myocyte Workstream Multiple Multiple MEA Multiple Volunteer List Replicates, Stem Cells Platforms Type Stem Cells 1 Myocyte Type Across 1 Platform 1 Myocyte Type Indicator Across 1 Platform MES Site 1 MEA Site 2 MEA Site 3 MEA Site 4 MEA Site 5 MEA Site 6 MEA Site 7 MEA Site 8 MEA Site 9 MEA Site 10 VSO Site 1 VSO Site 2 VSO Site 3 Di-4-ANEPPS Di-4-ANEPPS QuasAr1 VSO Site 4 FluoVolt 19
20 Clinical Foundations: Compound Selection & Clinical Regulatory Group TdP Risk Compound Selection/Regulatory (CSRC): Categorization available drugs for proarrhythmic risk: - High, Intermediate, Low risk categories - Evaluate clinical data for proarrhythmia - Overall experience, history, patient population, pharmacokinetics - 29 drugs nominated for model development and verification Input from clinical developers/regulators - Validation & acceptance of CiPA paradigm Define Phase 1 ECG Verification Efforts Provides Gold Standard for Calibrating CiPA Studies 20
21 Criteria for Compound Selection - No prominent proarrhythmic / active metabolites - Good solubility - Defined cellular electrophysiology - Ranking compounds based on clinically demonstrated torsadogenic risk and proarrhythmic incidence - published reports, FDA AERS database, additional data sources, expert opinion - Compounds grouped into high, intermediate and very low (i.e., none) risk categories - Intermediate risk compounds grouped into one category - Very low risk no discernable risk - Culled from much larger list of candidates 21
22 Compounds List: CiPA Redfern CiPA Redfern CiPA Redfern HIGH RISK Category INTERMEDIATE RISK Category LOW RISK Category Azimilide 1 Astemizole 2 Diltiazem 5 Bepridil 3 Chlorpromazine - Loratadine 5 Dofetilide 1 Cisapride 2 Metoprolol - Ibutilide 1 Clarithromycin 4 Mexiletine - Quinidine 1 Clozapine - Nifedipine 4 Vandetanib - Domperidone 4 Nitrendipine 5 Methadone - Droperidol - Ranolazine - D,l Sotalol 1 Terfenadine 2 Tamoxifen 5 Pimozide 3 Verapamil 5 Risperidone 5 Ondansetron - 22
23 Group Efforts Against Timelines Parties Involved: Cardiac Safety Research Consortium: Cmpd Selection Team FDA: In Silico Working Group ILSI-HESI: Myocyte Stem Cell Working Group Safety Pharm. Society: Ion Channel Working Group J-iCSA 2015(?) Regulators: FDA, Health Canada, EMEA, Japan Japanese Safety Pharmacology Society Academic Contributors Vendors and Suppliers Timelines: Progress Review: 1Q
24 Future Activities: CiPA 2015 Refinement-Evaluations of Voltage Clamp Protocols Preliminary test of In Silico Reconstructions Based on Ionic Current Studies Assessment of CiPA Stem Cell Cardiomyocyte Pilot Study Results - Planning / Initiation of Validation Studies - Define set of validation compounds - Consult/collaborate/compare with J-iCSA Further discussion with Regulators - Expectations of CiPA Package of regulators, industry - Relation to eqt Clinical Efforts - How to integrate in vitro/in silico preclinical results with emerging clinical eqt approaches 24
25 CiPA: proarrhythmic risk assessment based on mechanistic understanding of integrated, cellular emergent drug effects on multiple human cardiac currents Expectations: - reduce unwarranted attrition early candidates - enable rapid progression of low risk TdP drugs to phase 1 studies (and confirmatory clinical ECG findings) 25
In Vitro Assessment to Replace the Clinical TQT Study: The Comprehensive In Vitro ProArrhythmia Assay (CiPA) Initiative
 In Vitro Assessment to Replace the Clinical TQT Study: The Comprehensive In Vitro ProArrhythmia Assay (CiPA) Initiative Gary Gintant, AbbVie for the Comprehensive in Vitro ProArrhythmia Assay Group Hot
In Vitro Assessment to Replace the Clinical TQT Study: The Comprehensive In Vitro ProArrhythmia Assay (CiPA) Initiative Gary Gintant, AbbVie for the Comprehensive in Vitro ProArrhythmia Assay Group Hot
hipsc Cardiomyocytes: Validation Study Results
 CENTER FOR DEVICES AND RADIOLOGICAL HEALTH OFFICE OF SCIENCE AND ENGINEERING LABORATORIES hipsc Cardiomyocytes: Validation Study Results Ksenia Blinova, PhD US Food and Drug Administration HESI-CSRC CiPA
CENTER FOR DEVICES AND RADIOLOGICAL HEALTH OFFICE OF SCIENCE AND ENGINEERING LABORATORIES hipsc Cardiomyocytes: Validation Study Results Ksenia Blinova, PhD US Food and Drug Administration HESI-CSRC CiPA
Evolution of strategies to improve preclinical cardiac safety testing
 OPINION Evolution of strategies to improve preclinical cardiac safety testing Gary Gintant, Philip T. Sager and Norman Stockbridge Abstract The early and efficient assessment of cardiac safety liabilities
OPINION Evolution of strategies to improve preclinical cardiac safety testing Gary Gintant, Philip T. Sager and Norman Stockbridge Abstract The early and efficient assessment of cardiac safety liabilities
Benchmarking In Silico Models and Candidate Metrics for Assessing the Risk of Torsade de Pointes
 Benchmarking In Silico Models and Candidate Metrics for Assessing the Risk of Torsade de Pointes Sara Dutta Division of Applied Regulatory Science/OCP/CDER CRSC/HESI/SPS/FDA Meeting December 11, 2014 Silver
Benchmarking In Silico Models and Candidate Metrics for Assessing the Risk of Torsade de Pointes Sara Dutta Division of Applied Regulatory Science/OCP/CDER CRSC/HESI/SPS/FDA Meeting December 11, 2014 Silver
Metrion Biosciences: the ion channel specialists
 Metrion Biosciences: the ion channel specialists In depth profiling of human ipsc cardiomyocytes: From electrophysiology to phenotypic assays Saïd El-Haou, PhD 30 November 2017 www.metrionbiosciences.com
Metrion Biosciences: the ion channel specialists In depth profiling of human ipsc cardiomyocytes: From electrophysiology to phenotypic assays Saïd El-Haou, PhD 30 November 2017 www.metrionbiosciences.com
TCP Transl Clin Pharmacol
 TCP 2018;26(4):145-149 https://doi.org/10.12793/tcp.2018.26.4.145 Five years of the CiPA project (2013 2018) - what did we learn? Dong-Seok Yim* Department of Clinical Pharmacology and Therapeutics, Seoul
TCP 2018;26(4):145-149 https://doi.org/10.12793/tcp.2018.26.4.145 Five years of the CiPA project (2013 2018) - what did we learn? Dong-Seok Yim* Department of Clinical Pharmacology and Therapeutics, Seoul
Japanese Stem Cell Efforts
 Japanese Stem Cell Efforts Yuko Sekino, Ph. D. Organizer of JiCSA Head, Division of Pharmacology, National Institute of Health Sciences (NIHS) Pharmaceutical and Medical Safety Bureau NI HS Since 1874
Japanese Stem Cell Efforts Yuko Sekino, Ph. D. Organizer of JiCSA Head, Division of Pharmacology, National Institute of Health Sciences (NIHS) Pharmaceutical and Medical Safety Bureau NI HS Since 1874
MEA assays using human ipsc-derived cardiomyocytes; challenges and opportunities
 MEA assays using human ipsc-derived cardiomyocytes; challenges and opportunities Event Presenter Date EMA Workshop 2017 Tessa de Korte, MSc 2017, October 5 Page 1 Ncardia at a glance Foundation Ncardia
MEA assays using human ipsc-derived cardiomyocytes; challenges and opportunities Event Presenter Date EMA Workshop 2017 Tessa de Korte, MSc 2017, October 5 Page 1 Ncardia at a glance Foundation Ncardia
Developing and Validating an In Silico Model for Proarrhythmia Risk Assessment Under the CiPA Initiative
 Developing and Validating an In Silico Model for Proarrhythmia Risk Assessment Under the CiPA Initiative May 2018 Zhihua Li, PhD for the CiPA In Silico Working Group US Food and Drug Administration This
Developing and Validating an In Silico Model for Proarrhythmia Risk Assessment Under the CiPA Initiative May 2018 Zhihua Li, PhD for the CiPA In Silico Working Group US Food and Drug Administration This
Ncardia. Assessment of pro-arrhythmic effects in Pluricyte Cardiomyocytes. using the Axion BioSystems Maestro TM MEA system
 Ncardia Stem cell experts Assessment of pro-arrhythmic effects in Pluricyte Cardiomyocytes using the Axion BioSystems Maestro TM MEA system Application note Version 2.0 Contents 1. Introduction 1 2. Assessment
Ncardia Stem cell experts Assessment of pro-arrhythmic effects in Pluricyte Cardiomyocytes using the Axion BioSystems Maestro TM MEA system Application note Version 2.0 Contents 1. Introduction 1 2. Assessment
Assessment of pro-arrhythmic effects using Pluricyte Cardiomyocytes. on the ACEA xcelligence RTCA CardioECR
 Assessment of pro-arrhythmic effects using Pluricyte Cardiomyocytes on the ACEA xcelligence RTCA CardioECR Application Note Version 2.1 / March 2018 Contents 1. Introduction 1 2. Assessment of pro-arrhythmic
Assessment of pro-arrhythmic effects using Pluricyte Cardiomyocytes on the ACEA xcelligence RTCA CardioECR Application Note Version 2.1 / March 2018 Contents 1. Introduction 1 2. Assessment of pro-arrhythmic
TdP Mechanisms and CiPA
 TdP Mechanisms and CiPA Craig T. January, MD, PhD Division of Cardiovascular Medicine University of Wisconsin-Madison Cardiac Safety Research Consortium Hilton Washington DC December 6, 2016 Disclosures
TdP Mechanisms and CiPA Craig T. January, MD, PhD Division of Cardiovascular Medicine University of Wisconsin-Madison Cardiac Safety Research Consortium Hilton Washington DC December 6, 2016 Disclosures
CiPA, Pre-clinical Testing. Derek Leishman PhD DSP & ICH S7B
 CiPA, Pre-clinical Testing Derek Leishman PhD DSP & ICH S7B Outline Introduction Scenarios Conclusion Expressing an opinion What is not covered? Analysis/critique of the components too little time to try
CiPA, Pre-clinical Testing Derek Leishman PhD DSP & ICH S7B Outline Introduction Scenarios Conclusion Expressing an opinion What is not covered? Analysis/critique of the components too little time to try
Preclinical Perspectives of QT Outlook from ICH Guidelines
 Biometry in Early Clinical Research QT/QTc Interval Workshop in Heidelberg, November 17-19, 2005 Preclinical Perspectives of QT Outlook from ICH Guidelines Gerd Bode, M.D.,Ph.D. Frelance Consultant, e-mail
Biometry in Early Clinical Research QT/QTc Interval Workshop in Heidelberg, November 17-19, 2005 Preclinical Perspectives of QT Outlook from ICH Guidelines Gerd Bode, M.D.,Ph.D. Frelance Consultant, e-mail
Mechanisms of Arrhythmogenesis: Focus on Long QT Syndrome (LQTS)
 Mechanisms of Arrhythmogenesis: Focus on Long QT Syndrome (LQTS) Craig T. January, MD, PhD Division of Cardiovascular Medicine University of Wisconsin-Madison CSRC-HESI-FDA Rechanneling the Current Cardiac
Mechanisms of Arrhythmogenesis: Focus on Long QT Syndrome (LQTS) Craig T. January, MD, PhD Division of Cardiovascular Medicine University of Wisconsin-Madison CSRC-HESI-FDA Rechanneling the Current Cardiac
Antiarrhythmic Drugs
 Antiarrhythmic Drugs DR ATIF ALQUBBANY A S S I S T A N T P R O F E S S O R O F M E D I C I N E / C A R D I O L O G Y C O N S U L T A N T C A R D I O L O G Y & I N T E R V E N T I O N A L E P A C H D /
Antiarrhythmic Drugs DR ATIF ALQUBBANY A S S I S T A N T P R O F E S S O R O F M E D I C I N E / C A R D I O L O G Y C O N S U L T A N T C A R D I O L O G Y & I N T E R V E N T I O N A L E P A C H D /
Breakout #4 Phase 1 ECG:
 Breakout #4 Phase 1 ECG: Potential Role of ECG under CiPA HESI CSRC CiPA Meeting Washington, DC - May 22, 2018 Jose Vicente, PhD Christine Garnett, PharmD Division of Cardiovascular and Renal Products
Breakout #4 Phase 1 ECG: Potential Role of ECG under CiPA HESI CSRC CiPA Meeting Washington, DC - May 22, 2018 Jose Vicente, PhD Christine Garnett, PharmD Division of Cardiovascular and Renal Products
Cardiac Safety Assessment. In-Vitro/In-Vivo Extrapolation
 Cardiac Safety Assessment In-Vitro/In-Vivo Extrapolation Drugs removed from the market for arrhythmia risk Courtesy of Dr. Norman Stockbridge Copyright 2014 Certara, L.P. All rights reserved. 2 Let s have
Cardiac Safety Assessment In-Vitro/In-Vivo Extrapolation Drugs removed from the market for arrhythmia risk Courtesy of Dr. Norman Stockbridge Copyright 2014 Certara, L.P. All rights reserved. 2 Let s have
Can the Thorough QT Study Be Replaced? Norman Stockbridge Division of Cardiovascular and Renal Products FDA/CDER
 Can the Thorough QT Study Be Replaced? Norman Stockbridge Division of Cardiovascular and Renal Products FDA/CDER Can the TQT study be replaced? Yes. Thank you. Any questions? Four genre of replacement
Can the Thorough QT Study Be Replaced? Norman Stockbridge Division of Cardiovascular and Renal Products FDA/CDER Can the TQT study be replaced? Yes. Thank you. Any questions? Four genre of replacement
QT prolongation and drug-drug interactions. Filip Josephson M.D., Ph.D Clinical Assessor Swedish Medical Products Agency
 QT prolongation and drug-drug interactions Filip Josephson M.D., Ph.D Clinical Assessor Swedish Medical Products Agency Disposition Introduction to QT prolongation Three relevant DDI scenarios Concluding
QT prolongation and drug-drug interactions Filip Josephson M.D., Ph.D Clinical Assessor Swedish Medical Products Agency Disposition Introduction to QT prolongation Three relevant DDI scenarios Concluding
Preparing for Changing Cardiac Safety Regulations. Joy Olbertz, PharmD, PhD
 Preparing for Changing Cardiac Safety Regulations Joy Olbertz, PharmD, PhD Questions? Do I still have to assess proarrhythmic potential of my compound in a Thorough QT (TQT) study? If I have a TQT to conduct,
Preparing for Changing Cardiac Safety Regulations Joy Olbertz, PharmD, PhD Questions? Do I still have to assess proarrhythmic potential of my compound in a Thorough QT (TQT) study? If I have a TQT to conduct,
Assessment of pro-arrhythmic effects using Pluricyte Cardiomyocytes on the ACEA xcelligence RTCA CardioECR
 Assessment of pro-arrhythmic effects using Pluricyte Cardiomyocytes on the ACEA xcelligence RTCA CardioECR Version 1.2 CONTENTS Getting started... 3 Technical support and training... 3 1. Introduction...
Assessment of pro-arrhythmic effects using Pluricyte Cardiomyocytes on the ACEA xcelligence RTCA CardioECR Version 1.2 CONTENTS Getting started... 3 Technical support and training... 3 1. Introduction...
Industry Experience of the Concentration-QTc Relationship in Phase 1 Studies. Corina Dota, AstraZeneca
 Industry Experience of the Concentration-QTc Relationship in Phase 1 Studies Corina Dota, AstraZeneca CSRC-2 th February 2012 ICH E14 2.2.5 Alternative Strategies to Assess QT/QTc Interval Effects Alternatives
Industry Experience of the Concentration-QTc Relationship in Phase 1 Studies Corina Dota, AstraZeneca CSRC-2 th February 2012 ICH E14 2.2.5 Alternative Strategies to Assess QT/QTc Interval Effects Alternatives
Assessment of pro-arrhythmic effects using Pluricyte Cardiomyocytes on the Axion Maestro system
 Assessment of pro-arrhythmic effects using Pluricyte Cardiomyocytes on the Axion Maestro system Version 1.0 CONTENTS Getting started... 3 Technical support and training... 3 1. Introduction... 4 2. Workflow...
Assessment of pro-arrhythmic effects using Pluricyte Cardiomyocytes on the Axion Maestro system Version 1.0 CONTENTS Getting started... 3 Technical support and training... 3 1. Introduction... 4 2. Workflow...
Cardiomyocyte? March 14 th 2013
 What Makes a Cardiomyocyte a Cardiomyocyte? Kyle Kolaja March 14 th 2013 What Makes a Cardiomyocyte a Cardiomyocyte? Shouldn t proliferate Should beat Should be fairly pure Should look like and express
What Makes a Cardiomyocyte a Cardiomyocyte? Kyle Kolaja March 14 th 2013 What Makes a Cardiomyocyte a Cardiomyocyte? Shouldn t proliferate Should beat Should be fairly pure Should look like and express
Assessment of pro-arrhythmic effects using Pluricyte Cardiomyocytes on the Axion BioSystems Maestro TM MEA system
 Assessment of pro-arrhythmic effects using Pluricyte Cardiomyocytes on the Axion BioSystems Maestro TM MEA system Version 1.2 CONTENTS Getting started... 3 1. Introduction... 4 2. Workflow... 5 Important
Assessment of pro-arrhythmic effects using Pluricyte Cardiomyocytes on the Axion BioSystems Maestro TM MEA system Version 1.2 CONTENTS Getting started... 3 1. Introduction... 4 2. Workflow... 5 Important
Drugs, herg and sudden death
 Cell Calcium 35 () 53 57 Drugs, herg and sudden death A.M. Brown a,b, a MetroHealth Campus, Case Western Reserve University, Cleveland, OH 8, USA b ChanTest, Inc., 656 Neo Parkway, Cleveland, OH 8, USA
Cell Calcium 35 () 53 57 Drugs, herg and sudden death A.M. Brown a,b, a MetroHealth Campus, Case Western Reserve University, Cleveland, OH 8, USA b ChanTest, Inc., 656 Neo Parkway, Cleveland, OH 8, USA
xcelligence System RTCA Cardio Instrument
 xcelligence System Application Note No. 15 January 2013 xcelligence System RTCA Cardio Instrument Preclinical Cardiac Safety Assessment using Human ipsc Derived icell TM Cardiomyocytes Introduction Over
xcelligence System Application Note No. 15 January 2013 xcelligence System RTCA Cardio Instrument Preclinical Cardiac Safety Assessment using Human ipsc Derived icell TM Cardiomyocytes Introduction Over
mesc Derived Cardiomyocytes and the RTCA Cardio Instrument - The Perfect Match for Screening Cardiac Effects
 mesc Derived Cardiomyocytes and the RTCA Cardio Instrument - The Perfect Match for Screening Cardiac Effects Dr. Kristina Tressat, Axiogenesis AG ELRIG Drug Discovery II 7-8 Sep 2011, Manchester Challange
mesc Derived Cardiomyocytes and the RTCA Cardio Instrument - The Perfect Match for Screening Cardiac Effects Dr. Kristina Tressat, Axiogenesis AG ELRIG Drug Discovery II 7-8 Sep 2011, Manchester Challange
Comparison of different proarrhythmia biomarkers in isolated rabbit hearts
 Comparison of different proarrhythmia biomarkers in isolated rabbit hearts Summary of PhD Thesis Szabolcs Orosz, MSc Supervisor: Attila Farkas MD, PhD 2nd Dept. of Internal Medicine and Cardiology Centre
Comparison of different proarrhythmia biomarkers in isolated rabbit hearts Summary of PhD Thesis Szabolcs Orosz, MSc Supervisor: Attila Farkas MD, PhD 2nd Dept. of Internal Medicine and Cardiology Centre
The QT Interval Safety Endpoint for DR- TB trials. Kelly Dooley & Gary Maartens (Disclaimer: I know very little cardiac electrophysiology)
 The QT Interval Safety Endpoint for DR- TB trials Kelly Dooley & Gary Maartens (Disclaimer: I know very little cardiac electrophysiology) The ECG tracing Physiology of a cardiac myocyte Flow of ions (Na
The QT Interval Safety Endpoint for DR- TB trials Kelly Dooley & Gary Maartens (Disclaimer: I know very little cardiac electrophysiology) The ECG tracing Physiology of a cardiac myocyte Flow of ions (Na
HHS Public Access Author manuscript Circ Res. Author manuscript; available in PMC 2016 June 05.
 Tissue and Animal Models of Sudden Cardiac Death Karim Sallam, MD *,1,2,3, Yingxin Li, PhD *,1,2,3, Philip T. Sager, MD 1,2, Steven R. Houser, PhD 4, and Joseph C. Wu, MD, PhD 1,2,3 1 Stanford Cardiovascular
Tissue and Animal Models of Sudden Cardiac Death Karim Sallam, MD *,1,2,3, Yingxin Li, PhD *,1,2,3, Philip T. Sager, MD 1,2, Steven R. Houser, PhD 4, and Joseph C. Wu, MD, PhD 1,2,3 1 Stanford Cardiovascular
Comparison of Electrophysiological Data From Human-Induced Pluripotent Stem Cell Derived Cardiomyocytes to Functional Preclinical Safety Assays
 toxicological sciences 134(2), 412 426 2013 doi:10.1093/toxsci/kft113 Advance Access publication May 20, 2013 Comparison of Electrophysiological Data From Human-Induced Pluripotent Stem Cell Derived Cardiomyocytes
toxicological sciences 134(2), 412 426 2013 doi:10.1093/toxsci/kft113 Advance Access publication May 20, 2013 Comparison of Electrophysiological Data From Human-Induced Pluripotent Stem Cell Derived Cardiomyocytes
PHARMACOLOGY OF ARRHYTHMIAS
 PHARMACOLOGY OF ARRHYTHMIAS Course: Integrated Therapeutics 1 Lecturer: Dr. E. Konorev Date: November 27, 2012 Materials on: Exam #5 Required reading: Katzung, Chapter 14 1 CARDIAC ARRHYTHMIAS Abnormalities
PHARMACOLOGY OF ARRHYTHMIAS Course: Integrated Therapeutics 1 Lecturer: Dr. E. Konorev Date: November 27, 2012 Materials on: Exam #5 Required reading: Katzung, Chapter 14 1 CARDIAC ARRHYTHMIAS Abnormalities
xcelligence RTCA Cardio Instrument Monitor Cardiomyocyte Beating in Real Time for Drug Discovery Research
 xcelligence RTCA Cardio Instrument Monitor Cardiomyocyte Beating in Real Time for Drug Discovery Research xcelligence RTCA Cardio Instrument A new way to test for compound cardiotoxicity Cardiac liability
xcelligence RTCA Cardio Instrument Monitor Cardiomyocyte Beating in Real Time for Drug Discovery Research xcelligence RTCA Cardio Instrument A new way to test for compound cardiotoxicity Cardiac liability
Advances in Safety Pharmacology: Utilizing ipsc-derived cardiomyocytes in early stage safety pharmacology investigations. Blake Anson, PhD.
 Advances in Safety Pharmacology: Utilizing ipsc-derived cardiomyocytes in early stage safety pharmacology investigations Blake Anson, PhD. Presentation Outline I. Safety Pharmacology and in-vitro models
Advances in Safety Pharmacology: Utilizing ipsc-derived cardiomyocytes in early stage safety pharmacology investigations Blake Anson, PhD. Presentation Outline I. Safety Pharmacology and in-vitro models
IMPLEMENTATION OF NOVEL ECG BIOMARKERS
 IMPLEMENTATION OF NOVEL ECG BIOMARKERS CSRC CiPA meeting May 21, 2018 Washington DC Borje Darpo, MD, PhD, FESC Chief Scientific Officer, Cardiac Safety, ERT 1 Disclosures I work as a consultant for ERT,
IMPLEMENTATION OF NOVEL ECG BIOMARKERS CSRC CiPA meeting May 21, 2018 Washington DC Borje Darpo, MD, PhD, FESC Chief Scientific Officer, Cardiac Safety, ERT 1 Disclosures I work as a consultant for ERT,
Drug-induced arrhythmogenesis. Prediction and Mechanisms. Blanca Rodriguez and Computational Biology Group University of Oxford
 Drug-induced arrhythmogenesis. Prediction and Mechanisms Blanca Rodriguez and Computational Biology Group University of Oxford Feb 3 rd, 29 Work done by Esther Pueyo, Lucia Romero, Alberto Corrias, Martin
Drug-induced arrhythmogenesis. Prediction and Mechanisms Blanca Rodriguez and Computational Biology Group University of Oxford Feb 3 rd, 29 Work done by Esther Pueyo, Lucia Romero, Alberto Corrias, Martin
Drug-induced effects on the heart: insights from HPC simulations
 Drug-induced effects on the heart: insights from HPC simulations Blanca Rodriguez Computational Biology Group University of Oxford July 1 st, 2009 Acknowledgements Esther Pueyo, Lucia Romero, Alberto Corrias,
Drug-induced effects on the heart: insights from HPC simulations Blanca Rodriguez Computational Biology Group University of Oxford July 1 st, 2009 Acknowledgements Esther Pueyo, Lucia Romero, Alberto Corrias,
Cover Page. The handle holds various files of this Leiden University dissertation.
 Cover Page The handle http://hdl.handle.net/1887/19939 holds various files of this Leiden University dissertation. Author: Chain, Anne S.Y. Title: Mind the gap : predicting cardiovascular risk during drug
Cover Page The handle http://hdl.handle.net/1887/19939 holds various files of this Leiden University dissertation. Author: Chain, Anne S.Y. Title: Mind the gap : predicting cardiovascular risk during drug
This is the author s final accepted version.
 Blinova, K. et al. (2017) Comprehensive translational assessment of humaninduced pluripotent stem cell derived cardiomyocytes for evaluating druginduced arrhythmias. Toxicological Sciences, 155(1), pp.
Blinova, K. et al. (2017) Comprehensive translational assessment of humaninduced pluripotent stem cell derived cardiomyocytes for evaluating druginduced arrhythmias. Toxicological Sciences, 155(1), pp.
Measuring Cardiac Activity:
 icell Cardiomyocytes 2 Application Protocol Measuring Cardiac Activity: Impedance and Extracellular Field Potential Detection with CardioExcyte 96 System Introduction icell Cardiomyocytes 2, human cardiomyocytes
icell Cardiomyocytes 2 Application Protocol Measuring Cardiac Activity: Impedance and Extracellular Field Potential Detection with CardioExcyte 96 System Introduction icell Cardiomyocytes 2, human cardiomyocytes
Measuring Cardiac Activity:
 icell Cardiomyocytes 2 Application Protocol Measuring Cardiac Activity: Impedance Detection with xcelligence RTCA Cardio System Introduction icell Cardiomyocytes 2, human cardiomyocytes derived from induced
icell Cardiomyocytes 2 Application Protocol Measuring Cardiac Activity: Impedance Detection with xcelligence RTCA Cardio System Introduction icell Cardiomyocytes 2, human cardiomyocytes derived from induced
Blocking the Late Sodium Current
 Non-classical Targets in Antiarrhythmic Therapy Blocking the Late Sodium Current Luiz Belardinelli, MD SVP, Cardiovascular Therapeutics Gilead Sciences, CA, USA Madrid June 28, 2011 Disclosures: Full time
Non-classical Targets in Antiarrhythmic Therapy Blocking the Late Sodium Current Luiz Belardinelli, MD SVP, Cardiovascular Therapeutics Gilead Sciences, CA, USA Madrid June 28, 2011 Disclosures: Full time
InPulse: Development of an ipsc cardiomyocyte (ipsc-cm) platform to assess drug-induced contractility liabilities.
 InPulse: Development of an ipsc cardiomyocyte (ipsc-cm) platform to assess drug-induced contractility liabilities. Sponsors: Arun Sridhar, Peter J. Clements, Brian Berridge, Drug induced contractility
InPulse: Development of an ipsc cardiomyocyte (ipsc-cm) platform to assess drug-induced contractility liabilities. Sponsors: Arun Sridhar, Peter J. Clements, Brian Berridge, Drug induced contractility
New ECG Biomarkers and their Potential Role in CiPA: Results and Implications
 New ECG Biomarkers and their Potential Role in CiPA: Results and Implications HESI CSRC CiPA Meeting Washington, DC - May 21, 2018 Jose Vicente, PhD for the ECG Biomarker Working Group Division of Cardiovascular
New ECG Biomarkers and their Potential Role in CiPA: Results and Implications HESI CSRC CiPA Meeting Washington, DC - May 21, 2018 Jose Vicente, PhD for the ECG Biomarker Working Group Division of Cardiovascular
12/19/16. Disclosures
 @atriumrx Don t Miss a Beat! Practical Tips on Managing Drug-Induced QTc Prolongation Sandeep Devabhakthuni, PharmD, BCPS- AQ Cardiology Assistant Professor University of Maryland School of Pharmacy Disclosures
@atriumrx Don t Miss a Beat! Practical Tips on Managing Drug-Induced QTc Prolongation Sandeep Devabhakthuni, PharmD, BCPS- AQ Cardiology Assistant Professor University of Maryland School of Pharmacy Disclosures
Proarrhythmia Risk Assessment in Human Induced Pluripotent Stem Cell-Derived Cardiomyocytes Using the Maestro MEA Platform
 TOXICOLOGICAL SCIENCES, 147(1), 2015, 286 295 doi: 10.1093/toxsci/kfv128 Advance Access Publication Date: June 27, 2015 Research Article Proarrhythmia Risk Assessment in Human Induced Pluripotent Stem
TOXICOLOGICAL SCIENCES, 147(1), 2015, 286 295 doi: 10.1093/toxsci/kfv128 Advance Access Publication Date: June 27, 2015 Research Article Proarrhythmia Risk Assessment in Human Induced Pluripotent Stem
Anti-HERG activity and the risk of drug-induced arrhythmias and sudden death
 European Heart Journal (2005) 26, 590 597 doi:10.1093/eurheartj/ehi092 Clinical research Anti-HERG activity and the risk of drug-induced arrhythmias and sudden death M.L. De Bruin 1,2 *, M. Pettersson
European Heart Journal (2005) 26, 590 597 doi:10.1093/eurheartj/ehi092 Clinical research Anti-HERG activity and the risk of drug-induced arrhythmias and sudden death M.L. De Bruin 1,2 *, M. Pettersson
Mr. Eknath Kole M.S. Pharm (NIPER Mohali)
 M.S. Pharm (NIPER Mohali) Drug Class Actions Therapeutic Uses Pharmacokinetics Adverse Effects Other Quinidine IA -Binds to open and inactivated Na+ -Decreases the slope of Phase 4 spontaneous depolarization
M.S. Pharm (NIPER Mohali) Drug Class Actions Therapeutic Uses Pharmacokinetics Adverse Effects Other Quinidine IA -Binds to open and inactivated Na+ -Decreases the slope of Phase 4 spontaneous depolarization
Low safety index of domperidone: mechanism for increased odds ratio for sudden cardiac death
 Acta Cardiol 2011; 66(4): 421-425 doi: 10.2143/AC.66.4.2126588 421 Low safety index of domperidone: mechanism for increased odds ratio for sudden cardiac death Luc M. HONDEGHEM, MD, PhD Dept. of Pharmacology,
Acta Cardiol 2011; 66(4): 421-425 doi: 10.2143/AC.66.4.2126588 421 Low safety index of domperidone: mechanism for increased odds ratio for sudden cardiac death Luc M. HONDEGHEM, MD, PhD Dept. of Pharmacology,
Chapter 12: Cardiovascular Physiology System Overview
 Chapter 12: Cardiovascular Physiology System Overview Components of the cardiovascular system: Heart Vascular system Blood Figure 12-1 Plasma includes water, ions, proteins, nutrients, hormones, wastes,
Chapter 12: Cardiovascular Physiology System Overview Components of the cardiovascular system: Heart Vascular system Blood Figure 12-1 Plasma includes water, ions, proteins, nutrients, hormones, wastes,
A New Comprehensive Translational Paradigm Using Human ipsc-derived Cardiomyocytes for Safety Assessment of Drug-Induced Arrhythmias
 A New Comprehensive Translational Paradigm Using Human ipsc-derived Cardiomyocytes for Safety Assessment of Drug-Induced Arrhythmias Armando Lagrutta Safety & Exploratory Pharmacology (SEP) Department
A New Comprehensive Translational Paradigm Using Human ipsc-derived Cardiomyocytes for Safety Assessment of Drug-Induced Arrhythmias Armando Lagrutta Safety & Exploratory Pharmacology (SEP) Department
QT Assessment: The new paradigm, but now what? Joy Olbertz PharmD, PhD Senior Director, Cardiovascular Safety Services
 QT Assessment: The new paradigm, but now what? Joy Olbertz PharmD, PhD Senior Director, Cardiovascular Safety Services The Evolution of ICH E14 Points to Consider Joint Health Canada/FDA Concept Paper
QT Assessment: The new paradigm, but now what? Joy Olbertz PharmD, PhD Senior Director, Cardiovascular Safety Services The Evolution of ICH E14 Points to Consider Joint Health Canada/FDA Concept Paper
Drug induced QT prolongation and Torsades de Pointes (TdP) Mark Friesen, PharmD March 13, 2013
 Drug induced QT prolongation and Torsades de Pointes (TdP) Mark Friesen, PharmD March 13, 2013 None Conflict of Interest TdP: Learning objectives To review the pathophysiology of QT prolongation and TdP
Drug induced QT prolongation and Torsades de Pointes (TdP) Mark Friesen, PharmD March 13, 2013 None Conflict of Interest TdP: Learning objectives To review the pathophysiology of QT prolongation and TdP
EXPOSURE RESPONSE ANALYSIS TO EVALUATE A DRUG'S EFFECT ON ECG PARAMETERS
 EXPOSURE RESPONSE ANALYSIS TO EVALUATE A DRUG'S EFFECT ON ECG PARAMETERS Christine Garnett, PharmD DCRP, FDA April 6, 2016 CSRC/FDA Workshop: The Proarrhythmic Assessment of New Chemical Entities Disclosures
EXPOSURE RESPONSE ANALYSIS TO EVALUATE A DRUG'S EFFECT ON ECG PARAMETERS Christine Garnett, PharmD DCRP, FDA April 6, 2016 CSRC/FDA Workshop: The Proarrhythmic Assessment of New Chemical Entities Disclosures
Early drug development: assessment of proarrhythmic risk and cardiovascular safety
 EXPERT REVIEW OF CLINICAL PHARMACOLOGY, 2016 VOL. 9, NO. 12, 1611 1618 http://dx.doi.org/10.1080/17512433.2016.1245142 REVIEW Early drug development: assessment of proarrhythmic risk and cardiovascular
EXPERT REVIEW OF CLINICAL PHARMACOLOGY, 2016 VOL. 9, NO. 12, 1611 1618 http://dx.doi.org/10.1080/17512433.2016.1245142 REVIEW Early drug development: assessment of proarrhythmic risk and cardiovascular
The mechanism of the cardiac arrhythmias Norbert Jost, PhD
 The mechanism of the cardiac arrhythmias Norbert Jost, PhD Department of Pharmacology & Pharmacotherapy, University of Szeged Division for Cardiovascular Pharmacology, Hungarian Academy of Sciences Szeged,
The mechanism of the cardiac arrhythmias Norbert Jost, PhD Department of Pharmacology & Pharmacotherapy, University of Szeged Division for Cardiovascular Pharmacology, Hungarian Academy of Sciences Szeged,
Drug-induced cardiotoxicity: screening methods, regulatory context, commercial environment. Beremans Ltd / Insight Pharma Reports April 2008
 Drug-induced cardiotoxicity: screening methods, regulatory context, commercial environment. Overview Beremans Ltd / Insight Pharma Reports April 2008 This report identifies and discusses methods, products
Drug-induced cardiotoxicity: screening methods, regulatory context, commercial environment. Overview Beremans Ltd / Insight Pharma Reports April 2008 This report identifies and discusses methods, products
Phase 2 Early Afterdepolarization as a Trigger of Polymorphic Ventricular Tachycardia in Acquired Long-QT Syndrome
 Phase 2 Early Afterdepolarization as a Trigger of Polymorphic Ventricular Tachycardia in Acquired Long-QT Syndrome Direct Evidence From Intracellular Recordings in the Intact Left Ventricular Wall Gan-Xin
Phase 2 Early Afterdepolarization as a Trigger of Polymorphic Ventricular Tachycardia in Acquired Long-QT Syndrome Direct Evidence From Intracellular Recordings in the Intact Left Ventricular Wall Gan-Xin
Drugs Controlling Myocyte Excitability and Conduction at the AV node Singh and Vaughan-Williams Classification
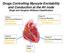 Drugs Controlling Myocyte Excitability and Conduction at the AV node Singh and Vaughan-Williams Classification Class I Na Channel Blockers Flecainide Propafenone Class III K channel Blockers Dofetilide,
Drugs Controlling Myocyte Excitability and Conduction at the AV node Singh and Vaughan-Williams Classification Class I Na Channel Blockers Flecainide Propafenone Class III K channel Blockers Dofetilide,
Predicting critical drug concentrations and torsadogenic risk using a multiscale exposure-response simulator
 Predicting critical drug concentrations and torsadogenic risk using a multiscale exposure-response simulator Francisco Sahli Costabal a, Jiang Yao b, Anna Sher c, Ellen Kuhl a, a Department of Mechanical
Predicting critical drug concentrations and torsadogenic risk using a multiscale exposure-response simulator Francisco Sahli Costabal a, Jiang Yao b, Anna Sher c, Ellen Kuhl a, a Department of Mechanical
Guidance for Industry
 Reprinted from FDA s website by Guidance for Industry E14 Clinical Evaluation of QT/QTc Interval Prolongation and Proarrhythmic Potential for Non-Antiarrhythmic Drugs Questions and Answers (R1) U.S. Department
Reprinted from FDA s website by Guidance for Industry E14 Clinical Evaluation of QT/QTc Interval Prolongation and Proarrhythmic Potential for Non-Antiarrhythmic Drugs Questions and Answers (R1) U.S. Department
Antidysrhythmics HST-151 1
 HST-151 1 Antidysrhythmics I. Ventricular muscle cell action potential a. Phase 0: Upstroke b. Phase 1: Early-fast repolarization c. Phase 2: Plateau d. Phase 3: Repolarization e. Phase 4: Diastole HST-151
HST-151 1 Antidysrhythmics I. Ventricular muscle cell action potential a. Phase 0: Upstroke b. Phase 1: Early-fast repolarization c. Phase 2: Plateau d. Phase 3: Repolarization e. Phase 4: Diastole HST-151
Assessment of pro-arrhythmic effects using Pluricyte Cardiomyocytes. on the ACEA xcelligence RTCA CardioECR
 Assessment of pro-arrhythmic effects using Pluricyte Cardiomyocytes on the ACEA xcelligence RTCA CardioECR User Guide Version 3.0 / March 2018 Contents 1. Introduction 2 2. Workflow 3 3. Important recommendations
Assessment of pro-arrhythmic effects using Pluricyte Cardiomyocytes on the ACEA xcelligence RTCA CardioECR User Guide Version 3.0 / March 2018 Contents 1. Introduction 2 2. Workflow 3 3. Important recommendations
Safety Pharmacology Post Meeting Survey 2014
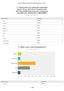 Q1 Please enter your personal information below. (If you want to be included in the 2014 Annual Meeting survey for a chance to win $200, this information is REQUIRED) Answered: 132 Skipped: 14 Name: Company:
Q1 Please enter your personal information below. (If you want to be included in the 2014 Annual Meeting survey for a chance to win $200, this information is REQUIRED) Answered: 132 Skipped: 14 Name: Company:
Can non-clinical repolarization assays predict the results of clinical thorough QT studies? Results from a research consortium
 BJP British Journal of Pharmacology RESEARCH PAPER British Journal of Pharmacology (2018) 175 606 617 606 Can non-clinical repolarization assays predict the results of clinical thorough QT studies? Results
BJP British Journal of Pharmacology RESEARCH PAPER British Journal of Pharmacology (2018) 175 606 617 606 Can non-clinical repolarization assays predict the results of clinical thorough QT studies? Results
The QT interval as it relates to the safety of non-cardiac drugs
 European Heart Journal Supplements (2007) 9 (Supplement G), G3 G8 doi:10.1093/eurheartj/sum047 The QT interval as it relates to the safety of non-cardiac drugs Peter R. Kowey 1 * and Marek Malik 2,3 1
European Heart Journal Supplements (2007) 9 (Supplement G), G3 G8 doi:10.1093/eurheartj/sum047 The QT interval as it relates to the safety of non-cardiac drugs Peter R. Kowey 1 * and Marek Malik 2,3 1
Electrocardiographic Markers Associated with Sotalol-induced Torsades de Pointes
 Electrocardiographic Markers Associated with Sotalol-induced Torsades de Pointes Based on Data from the Telemetric and Holter ECG Warehouse (THEW) Initiative Jean-PhiIippe Couderc, PhD Center for Quantitative
Electrocardiographic Markers Associated with Sotalol-induced Torsades de Pointes Based on Data from the Telemetric and Holter ECG Warehouse (THEW) Initiative Jean-PhiIippe Couderc, PhD Center for Quantitative
Modeling and Simulation Approaches for Cardiovascular Function and Their Role in Safety Assessment
 Citation: CPT Pharmacometrics Syst. Pharmacol. (2015) 4, 175 188; VC 2015 ASCPT All rights reserved doi:10.1002/psp4.18 REVIEW Modeling and Simulation Approaches for Cardiovascular Function and Their Role
Citation: CPT Pharmacometrics Syst. Pharmacol. (2015) 4, 175 188; VC 2015 ASCPT All rights reserved doi:10.1002/psp4.18 REVIEW Modeling and Simulation Approaches for Cardiovascular Function and Their Role
THE IMPACT OF DRUG-INDUCED QT INTERVAL PROLONGATION ON DRUG DISCOVERY AND DEVELOPMENT
 THE IMPACT OF DRUG-INDUCED QT INTERVAL PROLONGATION ON DRUG DISCOVERY AND DEVELOPMENT Bernard Fermini and Anthony A. Fossa During the past decade, a number of non-cardiovascular drugs have had their label
THE IMPACT OF DRUG-INDUCED QT INTERVAL PROLONGATION ON DRUG DISCOVERY AND DEVELOPMENT Bernard Fermini and Anthony A. Fossa During the past decade, a number of non-cardiovascular drugs have had their label
The overly-sensitive heart: sodium channel block and QRS interval prolongation
 BJP British Journal of Pharmacology DOI:10.1111/j.1476-5381.2011.01433.x www.brjpharmacol.org COMMENTARYbph_1433 254..259 The overly-sensitive heart: sodium channel block and QRS interval prolongation
BJP British Journal of Pharmacology DOI:10.1111/j.1476-5381.2011.01433.x www.brjpharmacol.org COMMENTARYbph_1433 254..259 The overly-sensitive heart: sodium channel block and QRS interval prolongation
PHARMACOLOGICAL INTERVENTION OF ARRHYTHMIAS. Ashish Parikh. Bachelor of Biomedical Engineering, University of Mumbai, 2006
 PHARMACOLOGICAL INTERVENTION OF ARRHYTHMIAS by Ashish Parikh Bachelor of Biomedical Engineering, University of Mumbai, 2006 Master of Biomedical Engineering, Cornell University, 2007 Submitted to the Graduate
PHARMACOLOGICAL INTERVENTION OF ARRHYTHMIAS by Ashish Parikh Bachelor of Biomedical Engineering, University of Mumbai, 2006 Master of Biomedical Engineering, Cornell University, 2007 Submitted to the Graduate
CSRC White Papers. An update Dec. 2010
 CSRC White Papers An update Dec. 2010 Ignacio Rodriguez, MD Clinical Safety Risk Management Roche ignacio.rodriguez.ir1@roche.com CSRC White Papers Position papers usually cover challenging areas of cardiovascular
CSRC White Papers An update Dec. 2010 Ignacio Rodriguez, MD Clinical Safety Risk Management Roche ignacio.rodriguez.ir1@roche.com CSRC White Papers Position papers usually cover challenging areas of cardiovascular
Pluricyte Cardiomyocytes. Axion BioSystems Maestro TM MEA system
 Pluricyte Cardiomyocytes Axion BioSystems Maestro TM MEA system User Guide Version 3.0 / March 2018 Contents 1. Introduction 2 2. Workflow 3 3. Important recommendations 4 4. Equipment, Materials and
Pluricyte Cardiomyocytes Axion BioSystems Maestro TM MEA system User Guide Version 3.0 / March 2018 Contents 1. Introduction 2 2. Workflow 3 3. Important recommendations 4 4. Equipment, Materials and
Models of Cardiac Electrophysiology:
 Models of Cardiac Electrophysiology: A Useful Tool for Enhancing Experimental Research T. Alexander Quinn, PhD National Heart and Lung Institute, Imperial College London Department of Computer Science,
Models of Cardiac Electrophysiology: A Useful Tool for Enhancing Experimental Research T. Alexander Quinn, PhD National Heart and Lung Institute, Imperial College London Department of Computer Science,
Pluricyte Cardiomyocytes. using the Multiwell MEA System from Multi Channel Systems
 Pluricyte Cardiomyocytes using the Multiwell MEA System from Multi Channel Systems User Guide Version 3.1 / March 2018 Contents 1. Introduction 2 2. Workflow 3 3. Important Recommendations 4 4. Equipment,
Pluricyte Cardiomyocytes using the Multiwell MEA System from Multi Channel Systems User Guide Version 3.1 / March 2018 Contents 1. Introduction 2 2. Workflow 3 3. Important Recommendations 4 4. Equipment,
Effects of ropinirole and rosiglitazone on action potential characteristics and ion currents in canine ventricular cells. by József Simkó, M.D.
 SHORT THESIS FOR THE DEGREE OF DOCTOR OF PHILOSOPHY (Ph.D.) Effects of ropinirole and rosiglitazone on action potential characteristics and ion currents in canine ventricular cells by József Simkó, M.D.
SHORT THESIS FOR THE DEGREE OF DOCTOR OF PHILOSOPHY (Ph.D.) Effects of ropinirole and rosiglitazone on action potential characteristics and ion currents in canine ventricular cells by József Simkó, M.D.
Multiparameter In Vitro Assessment of Compound Effects on Cardiomyocyte Physiology Using ipsc Cells
 457590JBXXXX10.1177/1087057112457590Jo urnal of Biomolecular ScreeningSirenko et al. 2012 Multiparameter In Vitro Assessment of Compound Effects on Cardiomyocyte Physiology Using ipsc Cells Journal of
457590JBXXXX10.1177/1087057112457590Jo urnal of Biomolecular ScreeningSirenko et al. 2012 Multiparameter In Vitro Assessment of Compound Effects on Cardiomyocyte Physiology Using ipsc Cells Journal of
Modulation of the late sodium current by ATX-II and ranolazine affects the reverse use-dependence and proarrhythmic liability of I Kr blockade
 308..316 British Journal of Pharmacology DOI:10.1111/j.1476-5381.2010.01181.x www.brjpharmacol.org RESEARCH PAPERbph_1181 Modulation of the late sodium current by ATX-II and ranolazine affects the reverse
308..316 British Journal of Pharmacology DOI:10.1111/j.1476-5381.2010.01181.x www.brjpharmacol.org RESEARCH PAPERbph_1181 Modulation of the late sodium current by ATX-II and ranolazine affects the reverse
Late Sodium Current Block for Drug-Induced Long QT Syndrome: Results From a Prospective Clinical Trial
 Late Sodium Current Block for Drug-Induced Long QT Syndrome: Results From a Prospective Clinical Trial L Johannesen 1,2, J Vicente 1,3,4, JW Mason 5,6, C Erato 5, C Sanabria 5, K Waite-Labott 5, M Hong
Late Sodium Current Block for Drug-Induced Long QT Syndrome: Results From a Prospective Clinical Trial L Johannesen 1,2, J Vicente 1,3,4, JW Mason 5,6, C Erato 5, C Sanabria 5, K Waite-Labott 5, M Hong
Antiarrhythmic Drugs 1/31/2018 1
 Antiarrhythmic Drugs 1/31/2018 1 Normal conduction pathway: 1- SA node generates action potential and delivers it to the atria and the AV node 2- The AV node delivers the impulse to purkinje fibers Other
Antiarrhythmic Drugs 1/31/2018 1 Normal conduction pathway: 1- SA node generates action potential and delivers it to the atria and the AV node 2- The AV node delivers the impulse to purkinje fibers Other
Chapter 14. Agents used in Cardiac Arrhythmias
 Chapter 14 Agents used in Cardiac Arrhythmias Cardiac arrhythmia Approximately 50% of post-myocardial infarction fatalities result from ventricular tachycarida (VT) or ventricular fibrillation (VF). These
Chapter 14 Agents used in Cardiac Arrhythmias Cardiac arrhythmia Approximately 50% of post-myocardial infarction fatalities result from ventricular tachycarida (VT) or ventricular fibrillation (VF). These
TQT studies - this is the end!
 TQT studies - this is the end! Borje Darpo MD, PhD Associate Professor of Cardiology Chief Scientific Officer icardiac Technologies Yes as the only way to confidently exclude that a new drug has a clinically
TQT studies - this is the end! Borje Darpo MD, PhD Associate Professor of Cardiology Chief Scientific Officer icardiac Technologies Yes as the only way to confidently exclude that a new drug has a clinically
Comparison of Two Highly Automated ECG Algorithms for Detection of Drug-Induced Cardiac Ion Channel Block
 Comparison of Two Highly Automated ECG Algorithms for Detection of Drug-Induced Cardiac Ion Channel Block Marina Brockway 1, Anthony A. Fossa 2 and Jay W. Mason 3,4 US Food and Drug Administration (FDA)
Comparison of Two Highly Automated ECG Algorithms for Detection of Drug-Induced Cardiac Ion Channel Block Marina Brockway 1, Anthony A. Fossa 2 and Jay W. Mason 3,4 US Food and Drug Administration (FDA)
Perspectives on Evaluating and Managing Cardiovascular Safety Risk for Anticancer Drugs
 Perspectives on Evaluating and Managing Cardiovascular Safety Risk for Anticancer Drugs DCTD/DTP/TPB Myrtle Davis, DVM, Ph.D. Chief, Toxicology and Pharmacology Branch Division of Cancer Treatment and
Perspectives on Evaluating and Managing Cardiovascular Safety Risk for Anticancer Drugs DCTD/DTP/TPB Myrtle Davis, DVM, Ph.D. Chief, Toxicology and Pharmacology Branch Division of Cancer Treatment and
ΚΟΛΠΙΚΗ ΜΑΡΜΑΡΥΓΗ ΦΑΡΜΑΚΕΥΤΙΚΗ ΗΛΕΚΤΡΙΚΗ ΑΝΑΤΑΞΗ. ΣΠΥΡΟΜΗΤΡΟΣ ΓΕΩΡΓΙΟΣ Καρδιολόγος, Ε/Α, Γ.Ν.Κατερίνης. F.E.S.C
 ΚΟΛΠΙΚΗ ΜΑΡΜΑΡΥΓΗ ΦΑΡΜΑΚΕΥΤΙΚΗ ΗΛΕΚΤΡΙΚΗ ΑΝΑΤΑΞΗ ΣΠΥΡΟΜΗΤΡΟΣ ΓΕΩΡΓΙΟΣ Καρδιολόγος, Ε/Α, Γ.Ν.Κατερίνης. F.E.S.C Definitions of AF: A Simplified Scheme Term Definition Paroxysmal AF AF that terminates
ΚΟΛΠΙΚΗ ΜΑΡΜΑΡΥΓΗ ΦΑΡΜΑΚΕΥΤΙΚΗ ΗΛΕΚΤΡΙΚΗ ΑΝΑΤΑΞΗ ΣΠΥΡΟΜΗΤΡΟΣ ΓΕΩΡΓΙΟΣ Καρδιολόγος, Ε/Α, Γ.Ν.Κατερίνης. F.E.S.C Definitions of AF: A Simplified Scheme Term Definition Paroxysmal AF AF that terminates
ΝΕΟΤΕΡΑ ΚΑΙ ΜΕΛΛΟΝΤΙΚΑ ΑΝΤΙΑΡΡΥΘΜΙΚΑ ΦΑΡΜΑΚΑ (ΑΑΦ-AAD)
 ΝΕΟΤΕΡΑ ΚΑΙ ΜΕΛΛΟΝΤΙΚΑ ΑΝΤΙΑΡΡΥΘΜΙΚΑ ΦΑΡΜΑΚΑ (ΑΑΦ-AAD) ΣΤΕΛΙΟΣ ΠΑΡΑΣΚΕΥΑÏΔΗΣ ΔΙΕΥΘΥΝΤΗΣ ΕΣΥ Α Καρδιολογική Κλινική ΑΠΘ, Νοσοκομείο ΑΧΕΠΑ, Θεσσαλονίκη NO CONFLICT OF INTEREST 1985 selectivity and effectiveness
ΝΕΟΤΕΡΑ ΚΑΙ ΜΕΛΛΟΝΤΙΚΑ ΑΝΤΙΑΡΡΥΘΜΙΚΑ ΦΑΡΜΑΚΑ (ΑΑΦ-AAD) ΣΤΕΛΙΟΣ ΠΑΡΑΣΚΕΥΑÏΔΗΣ ΔΙΕΥΘΥΝΤΗΣ ΕΣΥ Α Καρδιολογική Κλινική ΑΠΘ, Νοσοκομείο ΑΧΕΠΑ, Θεσσαλονίκη NO CONFLICT OF INTEREST 1985 selectivity and effectiveness
Short QT Syndrome: Pharmacological Treatment
 Journal of the American College of Cardiology Vol. 43, No. 8, 2004 2004 by the American College of Cardiology Foundation ISSN 0735-1097/04/$30.00 Published by Elsevier Inc. doi:10.1016/j.jacc.2004.02.034
Journal of the American College of Cardiology Vol. 43, No. 8, 2004 2004 by the American College of Cardiology Foundation ISSN 0735-1097/04/$30.00 Published by Elsevier Inc. doi:10.1016/j.jacc.2004.02.034
Antiarrhythmic Drugs. Munir Gharaibeh MD, PhD, MHPE School of Medicine, The University of Jordan November 2017
 Antiarrhythmic Drugs Munir Gharaibeh MD, PhD, MHPE School of Medicine, The University of Jordan November 2017 Types of Cardiac Arrhythmias Abnormalities of Impulse Formation: Rate disturbances. Triggered
Antiarrhythmic Drugs Munir Gharaibeh MD, PhD, MHPE School of Medicine, The University of Jordan November 2017 Types of Cardiac Arrhythmias Abnormalities of Impulse Formation: Rate disturbances. Triggered
Antiarrhythmic Drugs. Munir Gharaibeh MD, PhD, MHPE School of Medicine, The University of Jordan November 2018
 Antiarrhythmic Drugs Munir Gharaibeh MD, PhD, MHPE School of Medicine, The University of Jordan November 2018 2 Ion Permeability Changes Potential Changes Genes and Proteins 3 Cardiac Na+ channels 5 6
Antiarrhythmic Drugs Munir Gharaibeh MD, PhD, MHPE School of Medicine, The University of Jordan November 2018 2 Ion Permeability Changes Potential Changes Genes and Proteins 3 Cardiac Na+ channels 5 6
BJP. British Journal of Pharmacology. MPaci 1,JHyttinen 1,BRodriguez 2 and S Severi 3
 British Journal of Pharmacology RESEARCH PAPER Human induced pluripotent stem cell-derived versus adult cardiomyocytes: an in silico electrophysiological study on effects of ionic current block MPaci 1,JHyttinen
British Journal of Pharmacology RESEARCH PAPER Human induced pluripotent stem cell-derived versus adult cardiomyocytes: an in silico electrophysiological study on effects of ionic current block MPaci 1,JHyttinen
ECG CONVENTIONS AND INTERVALS
 1 ECG Waveforms and Intervals ECG waveforms labeled alphabetically P wave== represents atrial depolarization QRS complex=ventricular depolarization ST-T-U complex (ST segment, T wave, and U wave)== V repolarization.
1 ECG Waveforms and Intervals ECG waveforms labeled alphabetically P wave== represents atrial depolarization QRS complex=ventricular depolarization ST-T-U complex (ST segment, T wave, and U wave)== V repolarization.
HESI Technical Committee on Cardiovascular Safety: Advancing the clinical relevance of preclinical assessments
 HESI Technical Committee on Cardiovascular Safety: Advancing the clinical relevance of preclinical assessments Brian R. Berridge, DVM, PhD, DACVP Committee Co-Chair ILSI Health and Environmental Sciences
HESI Technical Committee on Cardiovascular Safety: Advancing the clinical relevance of preclinical assessments Brian R. Berridge, DVM, PhD, DACVP Committee Co-Chair ILSI Health and Environmental Sciences
IQ-CSRC PROSPECTIVE QTc STUDY Study Design and Choice of Drugs
 IQ-CSRC PROSPECTIVE QTc STUDY Study Design and Choice of Drugs Nenad Sarapa, MD IQ-CSRC meeting, Silver Spring, MD 12 December 2014 Background SAD studies can reliably assess QTc prolongation if proper
IQ-CSRC PROSPECTIVE QTc STUDY Study Design and Choice of Drugs Nenad Sarapa, MD IQ-CSRC meeting, Silver Spring, MD 12 December 2014 Background SAD studies can reliably assess QTc prolongation if proper
Modeling of Anatomy, Electrophysiology and Tension Development in the Human Heart
 European Functional Cardiac Modeling Meeting Modeling of Anatomy, Electrophysiology and Tension Development in the Human Heart Dr.-Ing. Gunnar Seemann Overview Electrophysiology Tension development Anatomy
European Functional Cardiac Modeling Meeting Modeling of Anatomy, Electrophysiology and Tension Development in the Human Heart Dr.-Ing. Gunnar Seemann Overview Electrophysiology Tension development Anatomy
AXION PLATES INDUSTRY-LEADING 768 ELECTRODES AT THE THROUGHPUT YOU NEED AXION MEA PLATES
 AXION PLATES INDUSTRY-LEADING 768 ELECTRODES AT THE THROUGHPUT YOU NEED Your research requires insightful, high-quality data from microelectrode array (MEA) experiments. Axion BioSystems MEA plates meet
AXION PLATES INDUSTRY-LEADING 768 ELECTRODES AT THE THROUGHPUT YOU NEED Your research requires insightful, high-quality data from microelectrode array (MEA) experiments. Axion BioSystems MEA plates meet
Is There a Genomic Basis to Acquired Channelopathic disease
 Is There a Genomic Basis to Acquired Channelopathic disease Yaniv Bar-Cohen, M.D. Associate Professor of Pediatrics Division of Cardiology / Electrophysiology Children s Hospital Los Angeles Keck School
Is There a Genomic Basis to Acquired Channelopathic disease Yaniv Bar-Cohen, M.D. Associate Professor of Pediatrics Division of Cardiology / Electrophysiology Children s Hospital Los Angeles Keck School
Droperidol and ondansetron in vitro electrophysiological drug interaction study
 ORIGINAL ARTICLE doi: 10.1111/j.1472-8206.2009.00735.x Droperidol and ondansetron in vitro electrophysiological drug interaction study Jacques Weissenburger a *, Christian Funck-Brentano b, Patrice Jaillon
ORIGINAL ARTICLE doi: 10.1111/j.1472-8206.2009.00735.x Droperidol and ondansetron in vitro electrophysiological drug interaction study Jacques Weissenburger a *, Christian Funck-Brentano b, Patrice Jaillon
Domperidone: Drug Interactions & Cautions (Mayo Clinic)
 Domperidone: Drug Interactions & Cautions (Mayo Clinic) Before Using In deciding to use a medicine, the risks of taking the medicine must be weighed against the good it will do. This is a decision you
Domperidone: Drug Interactions & Cautions (Mayo Clinic) Before Using In deciding to use a medicine, the risks of taking the medicine must be weighed against the good it will do. This is a decision you
