COMMITTEE FOR PROPRIETARY MEDICINAL PRODUCTS (CPMP) NOTE FOR GUIDANCE ON CLINICAL INVESTIGATION OF MEDICINAL PRODUCTS IN THE TREATMENT OF HYPERTENSION
|
|
|
- Eleanore O’Connor’
- 6 years ago
- Views:
Transcription
1 The European Agency for the Evaluation of Medicinal Products Human Medicines Evaluation Unit London, 19 November 1998 CPMP/EWP/238/95 Rev. 1 COMMITTEE FOR PROPRIETARY MEDICINAL PRODUCTS (CPMP) NOTE FOR GUIDANCE ON CLINICAL INVESTIGATION OF MEDICINAL PRODUCTS IN THE TREATMENT OF HYPERTENSION DISCUSSION IN THE EFFICACY WORKING PARTY (EWP) June/October 1994 TRANSMISSION TO THE CPMP December 1994 TRANSMISSION TO INTERESTED PARTIES December 1994 DEADLINE FOR COMMENTS June 1995 RE-SUBMISSION TO THE EFFICACY WORKING PARTY October 1995 February/March 1996 RE-SUBMISSION TO THE CPMP March 1996 RE-SUBMISSION TO THE EFFICACY WORKING PARTY October 1996 February/March 1997 APPROVAL BY THE CPMP May 1997 DATE FOR COMING INTO OPERATION November 1997 Note: This revision incorporates an addendum to the previous version 7 Westferry Circus, Canary Wharf, London E14 4HB, UK Switchboard: ( ) Fax: ( ) E_Mail: mail@emea.eudra.org Reproduction and/or distribution of this document is authorised for non commercial purposes only provided the EMEA is acknowledged
2 CLINICAL INVESTIGATION OF MEDICINAL PRODUCTS IN THE TREATMENT OF HYPERTENSION (CPMP/EWP/238/95) These Notes are intended to provide guidance for the evaluation of drugs in the treatment of hypertension. They should be read in conjunction with the Directives 75/318/EEC and 83/570/EEC, and current and future EU and ICH guidelines, especially those on (ICH) Studies in support of special populations: Geriatrics (ICH) The extent of population exposure to assess clinical safety of drugs intended or long-term treatment in non-life threatening conditions (EC) Biostatistical methodology in clinical trials (EC) Fixed-combination products (EC) Pharmacokinetic studies in man They are intended to assist applicants in the interpretation of the latter with respect to specific problems presented by anti-hypertensive products. 1. INTRODUCTION There is a continuous increase of cardiovascular risk associated with the level of blood pressure: the higher the blood pressure, the higher the risk of both stroke and coronary events. Nonfatal and fatal cardiovascular diseases - including coronary heart disease, stroke and congestive heart failure - as well as renal disease and all-cause mortality increase progressively with higher levels of both systolic blood pressure (SBP) and diastolic blood pressure (DBP). At every level of elevated DBP, risks increase in association with elevation of SBP. Recent data underscore the importance of elevations in SBP, as well as in DBP for diagnosis and therapy. The dividing line between normotension and hypertension is arbitrary and might vary with age. The current definition is that this line is the level of blood pressure above which intervention has been shown to reduce the risk. In the otherwise healthy adult population values below 140/90 mmhg are considered within the normal range and values of 140/90 mmhg and greater in the hypertensive range. Hypertension may be classified according to etiology: essential or primary hypertension vs. secondary hypertension; severity: according to WHO/ISH or JNC V; type: systolic, diastolic or both; extent or progression (e.g. malignant hypertension) of target organ damage (heart, brain, eyes, vessels, kidney). 2. ASSESSMENT OF EFFICACY CRITERIA 2.1 Blood pressure The goal of treating hypertension is to prevent morbidity and mortality associated with high blood pressure. Two classes of antihypertensives (ß-blockers and diuretics) have been shown to reduce cardiovascular morbidity and mortality and they may be considered as gold standards for the treatment of hypertension. Large morbidity/mortality studies are ongoing 1/10
3 with other antihypertensive agents, such as different types of calcium channel blockers, ACEinhibitors, alpha-blockers and angiotensin II-antagonist and these studies will verify if there are differences between drug classes as regards reduction in morbidity/mortality. Reduction in blood pressure has usually been accepted as a valid surrogate endpoint in order to assess whether this goal can be achieved by an antihypertensive agent. Notwithstanding, even if an antihypertensive effect has been proven, a new antihypertensive agent is only acceptable for registration when there is no suspicion o a detrimental effect on mortality and cardiovascular morbidity (see 6.9) 2.2 Morbidity and mortality Positive effects on mortality and cardiovascular morbidity can only be evaluated properly in large-scale and long-term controlled clinical trials. Until the results are available, it should be specifically mentioned in the SPC that beneficial effects on mortality and cardiovascular morbidity are unknown. 2.3 Target organ damage Although the prognostic relevance of target organ damage of heart, brain, eyes, kidneys and blood vessels has not yet been fully evaluated in valid clinical studies, it is presumably and plausibly associated with morbidity and mortality; this holds particularly true for left ventricular hypertrophy and proteinuria/microalbuminuria. Trials on outcomes of antihypertensive therapy, monitoring progression and regression of organ damage may provide relevant information on the comparative effectiveness of a new antihypertensive agent, but the prognostic value of drug effects with regard to morbidity and mortality remains to be established. Thus, these endpoints are considered of secondary value and specific studies are only mandatory when specific claims are made or when there are suspicions of a detrimental effect. 3. METHODS TO ASSESS EFFICACY 3.1 Blood pressure Blood pressure lowering effects of anti-hypertensive therapy should be documented as the pre-/post-treatment reduction of blood pressure. As a secondary endpoint these effects can also be assessed with respect to response criteria. Arbitrarily, response criteria for antihypertensive therapy include the percentage of patients with a normalisation of blood pressure (reduction SBP < 140 mmhg and DBP < 90 mmhg) and/or reduction of SBP 20 mmhg and/or DBP 10 mmhg. Results obtained should be discussed in terms of statistical significance and in relation to their clinical relevance. Blood pressure should be measured frequently with emphasis on the maximum and minimum effects of the drug, i.e. before the next dose is given (peak-trough ratio). The main endpoint should be blood pressure at trough which is defined as the residual effect at the end of the dose interval. The peak effect is the maximum blood pressure reduction (at steady state) identified in each patient following repeated blood pressure measurements across a dose interval. All measurements should be performed under standardised conditions. Assessment of trough-peak ratio has to take into account methodological issues and a minimum value should be pre-specified (e.g. 50%) for the recommended dose range. The following methods are available: ad a) Sphygmomanometry Measurements with a calibrated mercury sphygmomanometer are the standard. If not available, another device may be used which is calibrated carefully in proportion to a mercury 2/10
4 sphygmomanometer. Aneroid manometer is not recommended. Appropriate cuff size must be used to ensure accurate measurement. Both SBP and DBP should be recorded. The disappearance of sound (Korotkov phase V) should be used for the diastolic reading. Two or more readings separated by 2 minutes should be averaged. If the first two readings of DBP differ by more than 5 mmhg, additional readings should be obtained. Blood pressure should be checked in both arms, at least once. Blood pressure should be recorded in the arm with the higher pressure; if differences greater than 20 mmhg for SBP and 10 mmhg for DBP are present on 3 consecutive readings, the patient should be excluded from the study. Blood pressure should be measured in either supine or sitting position or both. Additional measurements of standing blood pressure are of value for evaluating postural changes and the risk of postural hypotension. No shift from one position to another should be made during the study. Supine or sitting should be for at least 5 minutes before measurement, standing should be for at least 1 minute before measurement. Blood pressure should be measured under standardised conditions, as nearly as possible at the same time each day, on the same arm, by the same personnel, with the same apparatus. Blood pressure measurement during exercise may provide supportive evidence for efficacy. ad b) Intra-arterial measurements Intra-arterial measurement of blood pressure has been used in phase II studies to investigate the relation between dose and height and duration of effect, to assess changes during exercise and to measure 24-hour efficacy. However, the method is complicated and the interpretation of the results is difficult since its prognostic value is not evaluated. Thus, intra-arterial measurement of blood pressure is not considered to be useful in the setting of clinical routine. ad c) Non-invasive ambulatory blood pressure monitoring As ambulatory blood pressure monitoring (ABPM) provides a better insight to blood pressure changes during every-day activities and is better standardised than casual readings, ABPM is required for the evaluation of new antihypertensive agents. The recorders used must fulfill international acknowledged validation procedures (e.g. AAMI/BHS). Recorders using auscultation and oscillometry as combined methods should be preferred since the numbers of errors can be reduced. Repetitive investigations should be performed on a comparable (work-) day using the same recorder. During daytime (06.00 hrs hrs) readings should be done at least at 15-minute intervals and during night-time (22.00 hrs hrs) at 30 minute intervals. For evaluation purposes at least 64 readings/24 hours have to be evaluable, including at least 52 readings during day-time and 12 readings at night. In day-time at least 2 readings and during night-time at least 1 reading/hour have to be available. Regarding the analysis of the results, mean values (± SD) for day- and night-time, periods should be evaluated separately. Special problems (e.g. trough-to-peak ratio, early morning rise) may be worked out by calculating hourly blood pressure or using time series analysis, respectively. ad d) automatic self (home) measurement Self (home) measurement of blood pressure with the help of automatic devices has been advocated as an alternative approach to better characterise a patient's blood pressure level and to estimate the effect of antihypertensive treatment, also in case of treatment cessation. Validation of the device used is necessary. 3.2 Target organ damage Compared to ECG and chest radiography, echocardiography combines a higher sensitivity for LVH with a more precise assessment of the degree of LVH (i.e. as a continuous variable reflected by magnitude of LV mass). Vascular Doppler echography and echo tracking events 3/10
5 can be used to study LV diastolic function and arterial compliance. Changes in renal function can be assessed in terms of serum creatinine concentrations, 24-hour creatinine clearance and urinary protein excretion. The most objective method to assess renal blood flow and/or glomerular filtration rate is by using radio-isotopes, but this method is limited, among other reasons, by exposure to radioactivity. Clearance of PAH and inulin can be used as alternatives. Opticus fundoscopy can provide evidence about retinal arteries, retina, and papilla. Ultrasound of the large vessels and/or angiography can provide evidence of arteriosclerotic plaques or increased vascular mass or increased intimal-medial thickness. 3.3 Morbidity and mortality Special emphasis should be placed on the effects in certain populations (e.g. elderly patients, subjects with co-morbidity, e.g. diabetic patients). The very old (above 75 years) need a special attention. The evaluation of cardiovascular morbidity should especially take into account sequelae of severe organ damage (e.g. myocardial infarction, stroke, renal insufficiency), and respective therapeutic interventions (e.g. co-medication, need for bypass surgery or PTCA). When planning an all-cause mortality study, further distinction should be made with regard to cardiovascular mortality and sudden death. 4. SELECTION OF PATIENTS 4.1 Study population Generally, the study population will depend on etiology and the type of hypertension for which the drug is intended. Studies for the evaluation of efficacy or safety of a new antihypertensive drug are mainly performed in patients with primary or essential hypertension of mild to moderate severity with elevated systolic and diastolic blood pressure. Patients of both genders should be included in studies in a balanced way. Patients with more severe stages of hypertension also need to be evaluated in add-on designed studies. Attention should be placed on ethnic peculiarities and concomitant illnesses (e.g. diabetes mellitus, renal disease). There is a special need for data in elderly patients, including specific pharmacokinetic studies, dose-response curves and safety data and the number of subjects above 60 years should be proportional to the frequency of prescriptions. Specific attention should be paid to people between 70 and 90 years of age. Salt intake and other non-- pharmacological measures should be kept constant during the trial duration. Patients with disorders causing secondary hypertension (e.g. phaeochromocytoma, adrenal adenoma, renal artery stenosis) and isolated systolic hypertension should be studied separately, if the indication is specifically claimed. This also refers to the treatment of hypertension in pregnancy which should also take into account the obstetrical and pediatric aspects of the problem. 5. STRATEGY-DESIGN Studies involving the first administration of medicinal products for hypertension to man do not differ essentially from those dealing with other cardioactive medicinal products. Patients receiving antihypertensive therapy who are to be included should be withdrawn from treatment during a wash-out period. The time needed will depend on the half-life of the agent(s) used and time taken for the blood pressure to return to pre-treatment levels. The period will be variable but may take weeks to months. Patients with markedly elevated blood pressure readings may require a continuous underlying antihypertensive drug therapy. Initial elevated readings should be confirmed on at least two subsequent visits during one to several weeks. A run-in period of 2, preferably 4 weeks is essential before commencing a 4/10
6 clinical trial of a new antihypertensive agent. An allocation of an individual patient to a study drug should only be performed if the basic blood pressure is stable. 5.1 Pharmacodynamics These studies should include evaluations of tolerability, duration of action, haemodynamic parameters (e.g. stroke volume, PCWP, SVR), heart rate (e.g. Holter), neurohumoral parameters (e.g. RAA-system, sympathetic nervous system) and renal function. Further studies - depending on the mechanism of action of the drug - may include evaluations of (intra) cardiac contractility, impulse formation and conduction, diastolic function, myocardial oxygen consumption, and coronary and regional blood flow. 5.2 Pharmacokinetics Special studies should be performed in the elderly and, depending of the way of elimination, in patients with varying degrees of renal dysfunction and/or hepatic dysfunction. 5.3 Interactions Interaction studies can provide information which may help to define the position of the new drug in the therapeutic schemes used in antihypertensive patients. Special attention should be devoted to potentially useful or unwanted interactions with other drugs which might be used alongside the investigational drug for combined treatment. These will be other antihypertensive agents of each of the major classes, but also other drugs which are likely to be used especially in the elderly patients. Special pharmacokinetic and pharmacodynamic interaction studies should be performed if results of clinical trials or the pharmacokinetic and pharmacodynamic properties of the drug give reason to suspect interaction problems. 5.4 Therapeutic studies Evaluation of efficacy Dose-response studies should be randomised, placebo-controlled and double-blinded using at least 3 dosages to establish the clinically useful dose-range as well as the optimal dose. The dose schedule selected for pivotal studies must be justified on the basis of the results of the dose-finding studies in the target population. Dose schedules should be clearly defined for elderly patients and those with various risk factors. The results of the dose-response studies of a new antihypertensive agent should provide robust evidence of its efficacy as compared to placebo for each recommended dose. Controlled trials with reference therapy should be performed aiming at demonstration of (at least) a similar efficacy/safety ratio of the drug under investigation in comparison to an acknowledged standard antihypertensive agent of the same and of other therapeutic classes. Placebo-controlled withdrawal phases can be introduced at the end of the study. A combination study with at least one other standard antihypertensive agent is mandatory. Special attention should be paid to reduction of the antihypertensive effect (tachyphylaxis). Careful consideration should be given to the results of those patients who fail to complete the study per protocol (e.g. drop-outs due to adverse events or lack of efficacy). Patients The efficacy studies will mainly include patients with mild to moderate essential hypertension, but a certain number of patients with (very) severe hypertension should be enrolled as well. The sample size depends, among others, on the target variable and its variance. Subgroup analyses for gender, race, age, etc. are desirable, A distinction should be made between in- and out- patients. 5/10
7 Design and study duration The dose-response studies should preferably be designed as parallel group studies. Following a run-in period of 2, preferably 4 weeks, the comparative studies with reference agents should be double-blind and randomised. The dose should be increased according dosing rules expressed in the protocol, and at each dose level the duration of treatment should be long enough to estimate the effect of the respective dose. The parallel group design using fixed doses should be applied in some studies, instead of escalating doses. The investigational drug may either be given as monotherapy or combined with underlying therapy. Drug therapy in the main dose-response studies should last at least 2-3 months in order to demonstrate efficacy in terms of the antihypertensive effect and each tested dose should be maintained over at least 4 weeks when more than one dose is used. Controlled studies with reference agents should last even longer up to 6 months, in order to allow a comparison with respect to adverse drug reactions as well. 6. SAFETY ASPECTS All adverse events occurring during the course of clinical trials should be fully documented with separate analysis of adverse drug events/reactions, drop-outs and patients who died while on therapy. Long-term controlled studies may be necessary. Any information available concerning clinical features and therapeutic measures in accidental overdosage or deliberate self-poisoning should be provided. Special efforts should be made to assess potential adverse effects/reactions that are characteristic for the class of drug being investigated. High-risk groups (e.g. elderly patients, patients with renal dysfunction or heart failure or coronary heart disease) require special consideration. Particular attention should be paid to the following specific side effects: 6.1 Hypotension This may be either symptomatic or asymptomatic. Special attention should be paid to orthostasis and first-dose phenomenon, especially at initiation of therapy or at increase of dosage. 6.2 Rebound hypertension Withdrawal phenomena, especially rebound hypertension, should be studied specifically. 6.3 Effects on cardiac rhythm This includes specifically (tachycardiac) pro-arrhythmic effects and effects on impulseconduction. Depending on the particular pharmacodynamic properties of the drug, heart rate, ECG and Holter monitoring should be performed at frequent intervals throughout the study. 6.4 Pro-ischemic effects Coronary steal effects due to coronary vasodilation, together with potential hypotensive effects, may lead to angina pectoris and myocardial infarction. When suspected, this needs to be studied specifically. 6.5 Effects on target organ damage Data on blood chemistry, urine analysis and other general laboratory investigations should be submitted. Effects of alterations in regional blood flow in other organ systems, especially the kidney and heart and brain can be studied. Special emphasis should be placed on renal function, electrolyte homeostasis, and LVH. Depending on suspicion of ophthalmological 6/10
8 side effects, ophthalmological examination should be performed throughout the study. Special emphasis should be placed on cognitive functions and CNS-effects (dizziness, blurred vision, syncope and TIA), especially in the elderly. 6.6 Effects on concomitant diseases Concomitant diseases include diabetes mellitus, renal diseases, ischemic heart disease, heart failure, cerebrovascular diseases and, more rarely, peripheral arterial occlusive disease. When specific claims are made, studies on hypertensive patients with concomitant diseases are required. 6.7 Effects an concomitant risk factors As concomitant risk factors are often present at the same time, effects on glucose and lipid metabolism should be studied specifically. 6.8 Immunological reactions Special attention should be paid to hypersensitivity reactions of the skin and other organs (especially liver, kidney, lung), changes in blood cells, and hepatitis. 6.9 Long-term effects on mortality and cardiovascular morbidity Although the risk of cardiovascular morbidity and mortality is strongly associated with the degree of hypertension, the risk of cardiovascular disease is also determined by many other factors, which may also be affected to a different extent by antihypertensive therapy. Results of pharmaco-epidemiological studies have raised the issue whether, despite an equal blood pressure lowering effect, the influence of antihypertensive drug classes on (cardiovascular) morbidity and mortality may not be alike. Even negative effects have been suggested. Therefore, a sufficient cohort of patients of both sexes and all ages should be continuously exposed to the drug for at least one year. The available data on mortality and cardiovascular morbidity from the clinical trial program should be thoroughly analysed, taking also into account preclinical data and the results obtained from other drugs of the same antihypertensive class and other classes as well. A new antihypertensive agent is only acceptable for registration if there is no suspicion of a detrimental effect on cardiovascular morbidity and mortality. Otherwise, additional studies to clarify the drug effect on these parameters are mandatory 7. FIXED COMBINATIONS 7.1 General remarks Combination therapy in hypertension is commonly applied to improve efficacy and/or safety as compared to the respective monotherapies. Mono-substances for the treatment of hypertension are generally combined in a fixed manner if: the combination of the individual components is plausible since complementary modes of action exist which result in additive antihypertensive effects or a reduction of ADRs; efficacy and safety of the individual components have been proven in confirmatory clinical studies; the individual suitable dosage ratio evaluated in confirmatory clinical trials with the free combination has corresponded with that of the fixed combination; 7/10
9 the joint application of the two components has proven to be efficacious, safe and thus clinically useful. In order to obtain a marketing authorisation for a fixed combination, it is mandatory to prove that each active component in the scheduled dosage independently contributes towards the positive evaluation of the combination drug. Concerning morbidity and mortality data the same requirements apply as to the monocomponents. 7.2 The clinical development of a fixed combination (Refer to the Addendum) Dose-finding studies are necessary for identifying the appropriate dosages of the components of a fixed combination. Preferentially, the factorial design should be used, allowing the simultaneous comparison of various dosage combinations with their respective components and with placebo. Ascending dosages (e.g. in a range of dose equal or superior to two) of the fixed combination could be tested in patients with insufficient response. The results of the dose-finding studies should be the basis for further, confirmatory, clinical trials. It is important that the clinical studies should be designed in accordance with the indication claimed and the wording of the indication must state clearly, whether the fixed combination should be used as first-line or as second-line therapy in hypertension Second-line therapy Usually, a fixed combination serves as a substitute for a free combination provided that a normalisation of blood pressure has been reached with the same doses of the individual components and the respective combination is well tolerated. Generally, the (fixed) combination should not be administered at the beginning of therapy. The following strategies in conducting confirmatory clinical studies are acceptable, but it is mandatory that at least one pivotal clinical study is performed in a population of patients whose blood pressure cannot be normalised with monotherapy of one of the monocomponents, if this indication is claimed: 1. Add-on therapy: Add the second drug to non-responders to the first drug, and vice versa. Dose-titration will usually be indicated. It is necessary to demonstrate a statistically significant and clinically relevant additional blood pressure reduction of the combination in patients who did not respond sufficiently to therapeutic doses of either monotherapy. Furthermore, it is necessary to show that any additional safety concerns (incidence/seriousness/severity/outcome of adverse events/adverse drug reactions) do not outweigh the additional benefit of the combination. In non-responders it is usually sufficient to show a clinically relevant and statistically significant superiority of the combination regarding the mean supine or sitting diastolic and systolic blood pressure, but it would be optimal, if such a trial could show a statistically significant improvement in response rates (blood pressure < 140/90 mmhg) for the fixed combination, as well. Sufficient duration of time should be taken into account to ensure that blood pressure levels are stable before the second drug is added to the medication. 2. Parallel group comparison of the combination with the individual components using the same therapeutic doses: Demonstration of statistically significant superior efficacy of the combination and no additional safety concerns outweighing the additional benefits of the fixed combination. Results of confirmatory dose-finding studies (e.g. using the factorial design) can also be supportive for the proof of efficacy. 8/10
10 In some cases (e.g. the fixed combination of two diuretics one of which is assumed to have a potassium-sparing effect) it can be mandatory to show a statistically significant and clinically relevantly superior safety while accepting a comparable efficacy. In such a case the studies should primarily aim at safety and the indication should be worded accordingly First-line therapy The (fixed) combination of two antihypertensive agents in subtherapeutic dose (i.e. dosage lower than the respective lowest approved individual dosage for antihypertensive monotherapy) aims at the reduction of (dose-dependent) adverse drug reactions (taking into account the anticipated increased frequency of idiosyncratic reactions if the patient is simultaneously confronted with two antihypertensive agents new to him). Recognising that patients with (mild to moderate) hypertension normally are treated with antihypertensive monotherapy which usually will be titrated to the individually optimised dosage, in certain patients first-line therapy with a fixed low-dose combination could be considered. At least the following is required if first-line therapy is claimed for a fixed low-dose combination: 1. Demonstration that each substance (component) has a documented contribution within the (fixed) combination: It is necessary (but not sufficient) that the results of a valid clinical trial evaluating a fixed low-dose combination document a statistically significant and clinically relevant greater blood pressure lowering effect (e.g. > 2 mmhg with respect to sdbp) than placebo, whereas the difference to each component (same subtherapeutic low dose as in the fixed combination) given separately has to be at least statistically significant. In addition, the response rate on the low-dose fixed combination should exceed that on placebo by an amount which is statistically significant and clinically valuable. If these objectives are addressed by means of a factorial design which includes groups of patients on additional doses and combinations of doses, then the conclusions regarding the low dose fixed combination of interest should still be based on the pairwise comparisons described above. 2. Indication for a reduction of (dose-dependent) adverse drug reactions by the lowdose fixed combination as compared to the components in the lowest approved dosages: It is necessary (but not sufficient) that the blood pressure lowering effect of the lowdose fixed combination is similar, i.e. at least not inferior (e.g. decrease in mean sdbp < 2 mmhg lower than the active comparator) than those of the lowest approved dosage of each component. Moreover, there should be a trend towards better safety and response rate regarding the low-dose fixed combination as compared to each component administered at the lowest approved dosage. Accordingly, the inclusion of a placebo arm in this study is helpful to underline these claims. 9/10
11 ADDENDUM FIXED COMBINATION ANTIHYPERTENSIVE MEDICINAL PRODUCTS IN SECOND LINE THERAPY The three following relevant issues were identified regarding applications for fixed combination antihypertensives in second line therapy. 1. Indication It was concluded that, provided sufficient evidence is included in the application, the second line indication for fixed combination medicinal product mentioned under section 4.1. should read as follows: Treatment of essential hypertension, <medicinal product Z> fixed dose combination (X mg /Y mg) is indicated in patients whose blood pressure is not adequately controlled on X or Y alone 2. Posology It was agreed that in section 4.2. Posology and method of administration" the two following recommendations should be included: Individual dose titration with the components can be recommended and When clinically appropriate, direct change from monotherapy to the fixed combination may be considered. 3. Clinical trials requirements for second line indication In the Note for Guidance on clinical investigation of medicinal products in the treatment of hypertension, two types of trials are discussed: trials in patients who are non-responders to the monotherapy, and trials in general population of hypertensive patients (including potential responders). It was agreed that different trial requirements might be needed to support the three different following indications: 3.1 In order to support the indication "Treatment of essential hypertension, <medicinal product Z> fixed dose combination (X mg /Y mg) is indicated in patients whose blood pressure is not adequately controlled on X alone", at least one add-on trial to active treatment in non-responders to X should be carried out. 3.2 In order to support the indication "Treatment of essential hypertension, <medicinal product Z> fixed dose combination (X mg /Y mg) is indicated in patients whose blood pressure is not adequately controlled on Y alone", at least one add-on trial to active treatment in non-responders to Y should be carried out. 3.3 In order to support the indication "Treatment of essential hypertension, <medicinal product Z> fixed dose combination (X mg /Y mg) is indicated in patients whose blood pressure is not adequately controlled on X or Y alone", two add-on studies one in nonresponders to X and one with non-responders to Y should be carried out. In some cases where only one add-on clinical study in non-responders has been carried out, data from appropriately designed parallel group comparative studies of the combination with the individual components may support a broader indication in both categories of non-responders. 10/10
CLINICAL INVESTIGATION OF ANTI-ANGINAL MEDICINAL PRODUCTS IN STABLE ANGINA PECTORIS
 CLINICAL INVESTIGATION OF ANTI-ANGINAL MEDICINAL PRODUCTS IN STABLE ANGINA PECTORIS Guideline Title Clinical Investigation of Anti-Anginal Medicinal Products in Stable Angina Pectoris Legislative basis
CLINICAL INVESTIGATION OF ANTI-ANGINAL MEDICINAL PRODUCTS IN STABLE ANGINA PECTORIS Guideline Title Clinical Investigation of Anti-Anginal Medicinal Products in Stable Angina Pectoris Legislative basis
COMMITTEE FOR MEDICINAL PRODUCTS FOR HUMAN USE (CHMP)
 European Medicines Agency Evaluation of Medicines for Human Use London, 29 July 2004 CPMP/EWP/3020/03 COMMITTEE FOR MEDICINAL PRODUCTS FOR HUMAN USE (CHMP) NOTE FOR GUIDANCE ON CLINICAL INVESTIGATION OF
European Medicines Agency Evaluation of Medicines for Human Use London, 29 July 2004 CPMP/EWP/3020/03 COMMITTEE FOR MEDICINAL PRODUCTS FOR HUMAN USE (CHMP) NOTE FOR GUIDANCE ON CLINICAL INVESTIGATION OF
Interested parties (organisations or individuals) that commented on the draft document as released for consultation.
 23 June 2016 EMA/CHMP/345847/2015 Committee for Medicinal products for Human Use Overview of comments received on ''Guideline on clinical investigation of medicinal products in the treatment of hypertension'
23 June 2016 EMA/CHMP/345847/2015 Committee for Medicinal products for Human Use Overview of comments received on ''Guideline on clinical investigation of medicinal products in the treatment of hypertension'
COMMITTEE FOR PROPRIETARY MEDICINAL PRODUCTS (CPMP)
 The European Agency for the Evaluation of Medicinal Products Evaluation of Medicines for Human Use London, 16 December 1999 COMMITTEE FOR PROPRIETARY MEDICINAL PRODUCTS (CPMP) NOTE FOR GUIDANCE ON CLINICAL
The European Agency for the Evaluation of Medicinal Products Evaluation of Medicines for Human Use London, 16 December 1999 COMMITTEE FOR PROPRIETARY MEDICINAL PRODUCTS (CPMP) NOTE FOR GUIDANCE ON CLINICAL
MEDICINAL PRODUCTS IN THE TREATMENT OF CARDIAC FAILURE
 MEDICINAL PRODUCTS IN THE TREATMENT OF CARDIAC FAILURE Guideline Title Medicinal Products in the Treatment of Cardiac Failure Legislative basis Directive 75/318/EEC as amended Date of first adoption November
MEDICINAL PRODUCTS IN THE TREATMENT OF CARDIAC FAILURE Guideline Title Medicinal Products in the Treatment of Cardiac Failure Legislative basis Directive 75/318/EEC as amended Date of first adoption November
COMMITTEE FOR PROPRIETARY MEDICINAL PRODUCTS (CPMP)
 The European Agency for the Evaluation of Medicinal Products Evaluation of Medicines for Human Use plondon, 20 February 2003 COMMITTEE FOR PROPRIETARY MEDICINAL PRODUCTS (CPMP) APPENDIX TO THE NOTE FOR
The European Agency for the Evaluation of Medicinal Products Evaluation of Medicines for Human Use plondon, 20 February 2003 COMMITTEE FOR PROPRIETARY MEDICINAL PRODUCTS (CPMP) APPENDIX TO THE NOTE FOR
COMMITTEE FOR MEDICINAL PRODUCTS FOR HUMAN USE (CHMP)
 European Medicines Agency Evaluation of Medicines for Human Use London, 29 July 04 CPMP/EWP/2986/03 COMMITTEE FOR MEDICINAL PRODUCTS FOR HUMAN USE (CHMP) NOTE FOR GUIDANCE ON CLINICAL INVESTIGATION OF
European Medicines Agency Evaluation of Medicines for Human Use London, 29 July 04 CPMP/EWP/2986/03 COMMITTEE FOR MEDICINAL PRODUCTS FOR HUMAN USE (CHMP) NOTE FOR GUIDANCE ON CLINICAL INVESTIGATION OF
5.2 Key priorities for implementation
 5.2 Key priorities for implementation From the full set of recommendations, the GDG selected ten key priorities for implementation. The criteria used for selecting these recommendations are listed in detail
5.2 Key priorities for implementation From the full set of recommendations, the GDG selected ten key priorities for implementation. The criteria used for selecting these recommendations are listed in detail
Reflection paper on assessment of cardiovascular safety profile of medicinal products
 25 February 2016 EMA/CHMP/50549/2015 Committee for Medicinal Products for Human Use (CHMP) Reflection paper on assessment of cardiovascular safety profile of medicinal products Draft agreed by Cardiovascular
25 February 2016 EMA/CHMP/50549/2015 Committee for Medicinal Products for Human Use (CHMP) Reflection paper on assessment of cardiovascular safety profile of medicinal products Draft agreed by Cardiovascular
Reflection paper on assessment of cardiovascular risk of medicinal products for the treatment of cardiovascular and metabolic diseases Draft
 1 2 3 21 May 2015 EMA/CHMP/50549/2015 Committee for Medicinal Products for Human Use (CHMP) 4 5 6 7 Reflection paper on assessment of cardiovascular risk of medicinal products for the treatment of cardiovascular
1 2 3 21 May 2015 EMA/CHMP/50549/2015 Committee for Medicinal Products for Human Use (CHMP) 4 5 6 7 Reflection paper on assessment of cardiovascular risk of medicinal products for the treatment of cardiovascular
Candesartan Antihypertensive Survival Evaluation in Japan (CASE-J) Trial of Cardiovascular Events in High-Risk Hypertensive Patients
 1/5 This site became the new ClinicalTrials.gov on June 19th. Learn more. We will be updating this site in phases. This allows us to move faster and to deliver better services. Show less IMPORTANT: Listing
1/5 This site became the new ClinicalTrials.gov on June 19th. Learn more. We will be updating this site in phases. This allows us to move faster and to deliver better services. Show less IMPORTANT: Listing
Cardiac Pathophysiology
 Cardiac Pathophysiology Evaluation Components Medical history Physical examination Routine laboratory tests Optional tests Medical History Duration and classification of hypertension. Patient history of
Cardiac Pathophysiology Evaluation Components Medical history Physical examination Routine laboratory tests Optional tests Medical History Duration and classification of hypertension. Patient history of
Egyptian Hypertension Guidelines
 Egyptian Hypertension Guidelines 2014 Egyptian Hypertension Guidelines Dalia R. ElRemissy, MD Lecturer of Cardiovascular Medicine Cairo University Why Egyptian Guidelines? Guidelines developed for rich
Egyptian Hypertension Guidelines 2014 Egyptian Hypertension Guidelines Dalia R. ElRemissy, MD Lecturer of Cardiovascular Medicine Cairo University Why Egyptian Guidelines? Guidelines developed for rich
Paediatric addendum to the note for guidance on the clinical investigation on medicinal products in the treatment of hypertension
 26 February 2015 EMA/CHMP/206815/2013 Committee for Medicinal Products for Human Use (CHMP) Paediatric addendum to the note for guidance on the clinical investigation on medicinal products in the treatment
26 February 2015 EMA/CHMP/206815/2013 Committee for Medicinal Products for Human Use (CHMP) Paediatric addendum to the note for guidance on the clinical investigation on medicinal products in the treatment
Systemic Hypertension Dr ahmed almutairi Assistant professor Internal medicine dept
 Systemic Hypertension Dr ahmed almutairi Assistant professor Internal medicine dept Continents 1- introduction 2- classification/definition 3- classification/etiology 4-etiology in both categories 5- complications
Systemic Hypertension Dr ahmed almutairi Assistant professor Internal medicine dept Continents 1- introduction 2- classification/definition 3- classification/etiology 4-etiology in both categories 5- complications
A European Regulator s perspective on Fixed Dose Combinations
 A European Regulator s perspective on Fixed Dose Combinations Peter Mol -Head assessor CBG-MEB, the Netherlands -SAWP member, EMA -Assistant professor, Clinical Pharmacology, University Medical Center
A European Regulator s perspective on Fixed Dose Combinations Peter Mol -Head assessor CBG-MEB, the Netherlands -SAWP member, EMA -Assistant professor, Clinical Pharmacology, University Medical Center
Amlodipine plus Lisinopril Tablets AMLOPRES-L
 Amlodipine plus Lisinopril Tablets AMLOPRES-L COMPOSITION AMLOPRES-L Each uncoated tablet contains: Amlodipine besylate equivalent to Amlodipine 5 mg and Lisinopril USP equivalent to Lisinopril (anhydrous)
Amlodipine plus Lisinopril Tablets AMLOPRES-L COMPOSITION AMLOPRES-L Each uncoated tablet contains: Amlodipine besylate equivalent to Amlodipine 5 mg and Lisinopril USP equivalent to Lisinopril (anhydrous)
COMMITTEE FOR MEDICINAL PRODUCTS FOR HUMAN USE (CHMP)
 European Medicines Agency London, 15 November 2007 Doc. Ref. EMEA/CHMP/EWP/517497/2007 COMMITTEE FOR MEDICINAL PRODUCTS FOR HUMAN USE (CHMP) GUIDELINE ON CLINICAL EVALUATION OF MEDICINAL PRODUCTS USED
European Medicines Agency London, 15 November 2007 Doc. Ref. EMEA/CHMP/EWP/517497/2007 COMMITTEE FOR MEDICINAL PRODUCTS FOR HUMAN USE (CHMP) GUIDELINE ON CLINICAL EVALUATION OF MEDICINAL PRODUCTS USED
MEDICINAL PRODUCTS FOR THE TREATMENT OF ARRHYTHMIAS
 MEDICINAL PRODUCTS FOR THE TREATMENT OF ARRHYTHMIAS Guideline Title Medicinal Products for the Treatment of Arrhythmias Legislative basis Directive 75/318/EEC as amended Date of first adoption November
MEDICINAL PRODUCTS FOR THE TREATMENT OF ARRHYTHMIAS Guideline Title Medicinal Products for the Treatment of Arrhythmias Legislative basis Directive 75/318/EEC as amended Date of first adoption November
Younger adults with a family history of premature artherosclerotic disease should have their cardiovascular risk factors measured.
 Appendix 2A - Guidance on Management of Hypertension Measurement of blood pressure All adults from 40 years should have blood pressure measured as part of opportunistic cardiovascular risk assessment.
Appendix 2A - Guidance on Management of Hypertension Measurement of blood pressure All adults from 40 years should have blood pressure measured as part of opportunistic cardiovascular risk assessment.
COMMITTEE FOR MEDICINAL PRODUCTS FOR HUMAN USE (CHMP) DRAFT
 European Medicines Agency Evaluation of Medicines for Human Use London, 17 February 2005 COMMITTEE FOR MEDICINAL PRODUCTS FOR HUMAN USE (CHMP) DRAFT GUIDELINE ON NON-CLINICAL AND CLINICAL DEVELOPMENT OF
European Medicines Agency Evaluation of Medicines for Human Use London, 17 February 2005 COMMITTEE FOR MEDICINAL PRODUCTS FOR HUMAN USE (CHMP) DRAFT GUIDELINE ON NON-CLINICAL AND CLINICAL DEVELOPMENT OF
Hypertension Update Warwick Jaffe Interventional Cardiologist Ascot Hospital
 Hypertension Update 2008 Warwick Jaffe Interventional Cardiologist Ascot Hospital Definition of Hypertension Continuous variable At some point the risk becomes high enough to justify treatment Treatment
Hypertension Update 2008 Warwick Jaffe Interventional Cardiologist Ascot Hospital Definition of Hypertension Continuous variable At some point the risk becomes high enough to justify treatment Treatment
This clinical study synopsis is provided in line with Boehringer Ingelheim s Policy on Transparency and Publication of Clinical Study Data.
 abcd Clinical Study Synopsis for Public Disclosure This clinical study synopsis is provided in line with s Policy on Transparency and Publication of Clinical Study Data. The synopsis which is part of the
abcd Clinical Study Synopsis for Public Disclosure This clinical study synopsis is provided in line with s Policy on Transparency and Publication of Clinical Study Data. The synopsis which is part of the
PUBLIC SUMMARY OF RISK MANAGEMENT PLAN (RMP) CANDESARTAN/HYDROCHLOROTHIAZIDE ORION 8 MG/12.5 MG, 16 MG/12.5 MG, 32 MG/12.5 MG, 32 MG/25 MG ORION
 PUBLIC SUMMARY OF RISK MANAGEMENT PLAN (RMP) CANDESARTAN/HYDROCHLOROTHIAZIDE ORION 8 MG/12.5 MG, 16 MG/12.5 MG, 32 MG/12.5 MG, 32 MG/25 MG ORION CORPORATION DATE: 17-04-2015, VERSION 2 VI.2 Elements for
PUBLIC SUMMARY OF RISK MANAGEMENT PLAN (RMP) CANDESARTAN/HYDROCHLOROTHIAZIDE ORION 8 MG/12.5 MG, 16 MG/12.5 MG, 32 MG/12.5 MG, 32 MG/25 MG ORION CORPORATION DATE: 17-04-2015, VERSION 2 VI.2 Elements for
Verapamil SR and trandolapril combination therapy in hypertension a clinical trial of factorial design
 Br J Clin Pharmacol 1998; 45: 491 495 Verapamil SR and trandolapril combination therapy in hypertension a clinical trial of factorial design Juergen Scholze, 1 Peter Zilles 2 & Daniele Compagnone 2 on
Br J Clin Pharmacol 1998; 45: 491 495 Verapamil SR and trandolapril combination therapy in hypertension a clinical trial of factorial design Juergen Scholze, 1 Peter Zilles 2 & Daniele Compagnone 2 on
COMMITTEE FOR PROPRIETARY MEDICINAL PRODUCTS (CPMP)
 The European Agency for the Evaluation of Medicinal Products Evaluation of Medicines for Human Use London, 30 May 2002 COMMITTEE FOR PROPRIETARY MEDICINAL PRODUCTS (CPMP) NOTE FOR GUIDANCE ON CLINICAL
The European Agency for the Evaluation of Medicinal Products Evaluation of Medicines for Human Use London, 30 May 2002 COMMITTEE FOR PROPRIETARY MEDICINAL PRODUCTS (CPMP) NOTE FOR GUIDANCE ON CLINICAL
COMMITTEE FOR PROPRIETARY MEDICINAL PRODUCTS (CPMP) NOTE FOR GUIDANCE ON EVALUATION OF ANTICANCER MEDICINAL PRODUCTS IN MAN
 The European Agency for the Evaluation of Medicinal Products Evaluation of Medicines for Human Use London, 24 July 2003 EMEA/ COMMITTEE FOR PROPRIETARY MEDICINAL PRODUCTS (CPMP) NOTE FOR GUIDANCE ON EVALUATION
The European Agency for the Evaluation of Medicinal Products Evaluation of Medicines for Human Use London, 24 July 2003 EMEA/ COMMITTEE FOR PROPRIETARY MEDICINAL PRODUCTS (CPMP) NOTE FOR GUIDANCE ON EVALUATION
Dr Doris M. W Kinuthia
 Dr Doris M. W Kinuthia Objectives Normal blood pressures in children Measurement of blood pressure in children Aetiology of Hypertension in children Evaluation of children with hypertension Treatment of
Dr Doris M. W Kinuthia Objectives Normal blood pressures in children Measurement of blood pressure in children Aetiology of Hypertension in children Evaluation of children with hypertension Treatment of
Hypertension The normal radial artery blood pressures in adults are: Systolic arterial pressure: 100 to 140 mmhg. Diastolic arterial pressure: 60 to
 Hypertension The normal radial artery blood pressures in adults are: Systolic arterial pressure: 100 to 140 mmhg. Diastolic arterial pressure: 60 to 90 mmhg. These pressures are called Normal blood pressure
Hypertension The normal radial artery blood pressures in adults are: Systolic arterial pressure: 100 to 140 mmhg. Diastolic arterial pressure: 60 to 90 mmhg. These pressures are called Normal blood pressure
Phase 3 investigation of aprocitentan for resistant hypertension management. Investor Webcast June 2018
 Phase 3 investigation of aprocitentan for resistant hypertension management Investor Webcast June 2018 The following information contains certain forward-looking statements, relating to the company s business,
Phase 3 investigation of aprocitentan for resistant hypertension management Investor Webcast June 2018 The following information contains certain forward-looking statements, relating to the company s business,
EMEA PUBLIC STATEMENT ON THE RECOMMENDATION TO SUSPEND THE MARKETING AUTHORISATION FOR ORLAAM (LEVACETYLMETHADOL) IN THE EUROPEAN UNION
 The European Agency for the Evaluation of Medicinal Products Evaluation of Medicines for Human Use London, 19 April 2001 EMEA/8776/01 EMEA PUBLIC STATEMENT ON THE RECOMMENDATION TO SUSPEND THE MARKETING
The European Agency for the Evaluation of Medicinal Products Evaluation of Medicines for Human Use London, 19 April 2001 EMEA/8776/01 EMEA PUBLIC STATEMENT ON THE RECOMMENDATION TO SUSPEND THE MARKETING
Clinical guideline Published: 24 August 2011 nice.org.uk/guidance/cg127
 Hypertension in adults: diagnosis and management Clinical guideline Published: 24 August 2011 nice.org.uk/guidance/cg127 NICE 2017. All rights reserved. Subject to Notice of rights (https://www.nice.org.uk/terms-and-conditions#notice-ofrights).
Hypertension in adults: diagnosis and management Clinical guideline Published: 24 August 2011 nice.org.uk/guidance/cg127 NICE 2017. All rights reserved. Subject to Notice of rights (https://www.nice.org.uk/terms-and-conditions#notice-ofrights).
How Low Do We Go? Update on Hypertension
 How Low Do We Go? Update on Beth L. Abramson, MD, FRCPC, FACC As presented at the University of Toronto s Saturday at the University Session (September 2003) Arecent World Health Organization report states
How Low Do We Go? Update on Beth L. Abramson, MD, FRCPC, FACC As presented at the University of Toronto s Saturday at the University Session (September 2003) Arecent World Health Organization report states
Sponsor Novartis. Generic Drug Name. Valsartan and amlodipine Trial Indication(s) Hypertension Protocol Number CVAA489A2306 Protocol Title
 Sponsor Novartis Generic Drug Name Valsartan and amlodipine Trial Indication(s) Hypertension Protocol Number CVAA489A2306 Protocol Title A randomized, double-blind, multi-center, active-controlled, parallel
Sponsor Novartis Generic Drug Name Valsartan and amlodipine Trial Indication(s) Hypertension Protocol Number CVAA489A2306 Protocol Title A randomized, double-blind, multi-center, active-controlled, parallel
The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION. 9 March 2011
 The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 9 March 2011 TAREG 3 mg/ml oral solution B/1 160 ml (CIP code: 491 474-8) Applicant: NOVARTIS PHARMA SAS valsartan
The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 9 March 2011 TAREG 3 mg/ml oral solution B/1 160 ml (CIP code: 491 474-8) Applicant: NOVARTIS PHARMA SAS valsartan
Management of Hypertension
 Clinical Practice Guidelines Management of Hypertension Definition and classification of blood pressure levels (mmhg) Category Systolic Diastolic Normal
Clinical Practice Guidelines Management of Hypertension Definition and classification of blood pressure levels (mmhg) Category Systolic Diastolic Normal
Paediatric addendum to CHMP guideline on the clinical investigations of medicinal products for the treatment of pulmonary arterial hypertension
 5 December 2011 EMA/CHMP/213972/2010 Committee for Medicinal Products for Human use (CHMP) Paediatric addendum to CHMP guideline on the clinical investigations of medicinal products for the treatment of
5 December 2011 EMA/CHMP/213972/2010 Committee for Medicinal Products for Human use (CHMP) Paediatric addendum to CHMP guideline on the clinical investigations of medicinal products for the treatment of
Slide notes: References:
 1 2 3 Cut-off values for the definition of hypertension are systolic blood pressure (SBP) 135 and/or diastolic blood pressure (DBP) 85 mmhg for home blood pressure monitoring (HBPM) and daytime ambulatory
1 2 3 Cut-off values for the definition of hypertension are systolic blood pressure (SBP) 135 and/or diastolic blood pressure (DBP) 85 mmhg for home blood pressure monitoring (HBPM) and daytime ambulatory
COMMITTEE FOR MEDICINAL PRODUCTS FOR HUMAN USE (CHMP) GUIDELINE ON THE EVALUATION OF NEW MEDICINAL PRODUCTS IN THE TREATMENT OF PRIMARY OSTEOPOROSIS
 European Medicines Agency Evaluation of Medicines for Human Use London, 14 December 2005 Doc. Ref. CPMP/EWP/552/95 Rev. 2 COMMITTEE FOR MEDICINAL PRODUCTS FOR HUMAN USE (CHMP) DRAFT GUIDELINE ON THE EVALUATION
European Medicines Agency Evaluation of Medicines for Human Use London, 14 December 2005 Doc. Ref. CPMP/EWP/552/95 Rev. 2 COMMITTEE FOR MEDICINAL PRODUCTS FOR HUMAN USE (CHMP) DRAFT GUIDELINE ON THE EVALUATION
Off-white, round, with faceted edges, scored on one side and bearing the inscription IK10 (10mg) or IK20 (20mg).
 1 TRADE NAME OF THE MEDICINAL PRODUCT Ikorel 10mg and 20mg Tablet 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Nicorandil 10mg or 20mg For a full list of excipients, see section 6.1 3 PHARMACEUTICAL FORM
1 TRADE NAME OF THE MEDICINAL PRODUCT Ikorel 10mg and 20mg Tablet 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Nicorandil 10mg or 20mg For a full list of excipients, see section 6.1 3 PHARMACEUTICAL FORM
Byvalson. (nebivolol, valsartan) New Product Slideshow
 Byvalson (nebivolol, valsartan) New Product Slideshow Introduction Brand name: Byvalson Generic name: Nebivolol, valsartan Pharmacological class: Beta-blocker + angiotensin II receptor blocker (ARB) Strength
Byvalson (nebivolol, valsartan) New Product Slideshow Introduction Brand name: Byvalson Generic name: Nebivolol, valsartan Pharmacological class: Beta-blocker + angiotensin II receptor blocker (ARB) Strength
Chapter 23. Media Directory. Cardiovascular Disease (CVD) Hypertension: Classified into Three Categories
 Chapter 23 Drugs for Hypertension Slide 37 Slide 41 Media Directory Nifedipine Animation Doxazosin Animation Upper Saddle River, New Jersey 07458 All rights reserved. Cardiovascular Disease (CVD) Includes
Chapter 23 Drugs for Hypertension Slide 37 Slide 41 Media Directory Nifedipine Animation Doxazosin Animation Upper Saddle River, New Jersey 07458 All rights reserved. Cardiovascular Disease (CVD) Includes
COMPOSITION. A film coated tablet contains. Active ingredient: irbesartan 75 mg, 150 mg or 300 mg. Rotazar (Film coated tablets) Irbesartan
 Rotazar (Film coated tablets) Irbesartan Rotazar 75 mg, 150 mg, 300 mg COMPOSITION A film coated tablet contains Active ingredient: irbesartan 75 mg, 150 mg or 300 mg. Rotazar 75 mg, 150 mg, 300 mg PHARMACOLOGICAL
Rotazar (Film coated tablets) Irbesartan Rotazar 75 mg, 150 mg, 300 mg COMPOSITION A film coated tablet contains Active ingredient: irbesartan 75 mg, 150 mg or 300 mg. Rotazar 75 mg, 150 mg, 300 mg PHARMACOLOGICAL
Treatment A Placebo to match COREG CR 20 mg OD + Lisinopril 10 mg OD (Days 1-7) Placebo to match COREG CR 40 mg OD + Lisinopril 10 mg OD (Days 8-14)
 The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
COMMITTEE FOR PROPRIETARY MEDICINAL PRODUCTS (CPMP)
 The European Agency for the Evaluation of Medicinal Products Evaluation of Medicines for Human Use London, 26 June 2003 COMMITTEE FOR PROPRIETARY MEDICINAL PRODUCTS (CPMP) POINTS TO CONSIDER ON THE CLINICAL
The European Agency for the Evaluation of Medicinal Products Evaluation of Medicines for Human Use London, 26 June 2003 COMMITTEE FOR PROPRIETARY MEDICINAL PRODUCTS (CPMP) POINTS TO CONSIDER ON THE CLINICAL
COMMITTEE FOR PROPRIETARY MEDICINAL PRODUCTS (CPMP)
 The Eu ropea nag ency for the Eva lu a tionof M edicina lprodu cts Human Medicines Evaluation Unit London, 26 February 1998 CPMP/EWP/559/95 COMMITTEE FOR PROPRIETARY MEDICINAL PRODUCTS (CPMP) NOTE FOR
The Eu ropea nag ency for the Eva lu a tionof M edicina lprodu cts Human Medicines Evaluation Unit London, 26 February 1998 CPMP/EWP/559/95 COMMITTEE FOR PROPRIETARY MEDICINAL PRODUCTS (CPMP) NOTE FOR
COMMITTEE FOR MEDICINAL PRODUCTS FOR HUMAN USE (CHMP) DRAFT
 European Medicines Agency London, 18 December 2008 Doc. Ref. EMEA/CHMP/EWP/356954/2008 COMMITTEE FOR MEDICINAL PRODUCTS FOR HUMAN USE (CHMP) DRAFT CHMP GUIDELINE ON THE CLINICAL INVESTIGATIONS OF MEDICINAL
European Medicines Agency London, 18 December 2008 Doc. Ref. EMEA/CHMP/EWP/356954/2008 COMMITTEE FOR MEDICINAL PRODUCTS FOR HUMAN USE (CHMP) DRAFT CHMP GUIDELINE ON THE CLINICAL INVESTIGATIONS OF MEDICINAL
From the desk of the: THE VIRTUAL NEPHROLOGIST
 Hypertension, also referred to as high blood pressure or HTN, is a medical condition in which the blood pressure is chronically elevated. It is a very common illness. One out of three American adults has
Hypertension, also referred to as high blood pressure or HTN, is a medical condition in which the blood pressure is chronically elevated. It is a very common illness. One out of three American adults has
EMEA WORKING PARTY ON HERBAL MEDICINAL PRODUCTS
 The European Agency for the Evaluation of Medicinal Products Evaluation of Medicines for Human Use 25 October 1999 EMEA/HMPWP/23/99 EMEA WORKING PARTY ON HERBAL MEDICINAL PRODUCTS UPDATED DRAFT POINTS
The European Agency for the Evaluation of Medicinal Products Evaluation of Medicines for Human Use 25 October 1999 EMEA/HMPWP/23/99 EMEA WORKING PARTY ON HERBAL MEDICINAL PRODUCTS UPDATED DRAFT POINTS
Hypertension. Most important public health problem in developed countries
 Hypertension Strategy for Continued Success in Treatment for the 21st Century November 15, 2005 Arnold B. Meshkov, M.D. Associate Professor of Medicine Temple University School of Medicine Philadelphia,
Hypertension Strategy for Continued Success in Treatment for the 21st Century November 15, 2005 Arnold B. Meshkov, M.D. Associate Professor of Medicine Temple University School of Medicine Philadelphia,
Hypertension. Risk of cardiovascular disease beginning at 115/75 mmhg doubles with every 20/10mm Hg increase. (Grade B)
 Practice Guidelines and Principles: Guidelines and principles are intended to be flexible. They serve as reference points or recommendations, not rigid criteria. Guidelines and principles should be followed
Practice Guidelines and Principles: Guidelines and principles are intended to be flexible. They serve as reference points or recommendations, not rigid criteria. Guidelines and principles should be followed
LACIPIL QUALITATIVE AND QUANTITATIVE COMPOSITION
 LACIPIL lacidipine QUALITATIVE AND QUANTITATIVE COMPOSITION Lacidipine, 2 mg - round shaped white engraved on one face. Lacidipine, 4 mg - oval white with break line on both faces. Lacidipine, 6 mg - oval
LACIPIL lacidipine QUALITATIVE AND QUANTITATIVE COMPOSITION Lacidipine, 2 mg - round shaped white engraved on one face. Lacidipine, 4 mg - oval white with break line on both faces. Lacidipine, 6 mg - oval
Hypertension and Cardiovascular Disease
 Hypertension and Cardiovascular Disease Copyright 2017 by Sea Courses Inc. All rights reserved. No part of this document may be reproduced, copied, stored, or transmitted in any form or by any means graphic,
Hypertension and Cardiovascular Disease Copyright 2017 by Sea Courses Inc. All rights reserved. No part of this document may be reproduced, copied, stored, or transmitted in any form or by any means graphic,
Antihypertensive Trial Design ALLHAT
 1 U.S. Department of Health and Human Services Major Outcomes in High Risk Hypertensive Patients Randomized to Angiotensin-Converting Enzyme Inhibitor or Calcium Channel Blocker vs Diuretic National Institutes
1 U.S. Department of Health and Human Services Major Outcomes in High Risk Hypertensive Patients Randomized to Angiotensin-Converting Enzyme Inhibitor or Calcium Channel Blocker vs Diuretic National Institutes
The P&T Committee Lisinopril (Qbrelis )
 Situation Background Assessment The P&T Committee Lisinopril (Qbrelis ) Qbrelis, 1 mg/ml lisinopril oral solution, has recently become an FDA- approved formulation. Current practice at UK Chandler Medical
Situation Background Assessment The P&T Committee Lisinopril (Qbrelis ) Qbrelis, 1 mg/ml lisinopril oral solution, has recently become an FDA- approved formulation. Current practice at UK Chandler Medical
hypertension Head of prevention and control of CVD disease office Ministry of heath
 hypertension t. Samavat MD,Cadiologist,MPH Head of prevention and control of CVD disease office Ministry of heath RECOMMENDATIONS FOR HYPERTENSION DIAGNOSIS, ASSESSMENT, AND TREATMENT Definition of hypertension
hypertension t. Samavat MD,Cadiologist,MPH Head of prevention and control of CVD disease office Ministry of heath RECOMMENDATIONS FOR HYPERTENSION DIAGNOSIS, ASSESSMENT, AND TREATMENT Definition of hypertension
WORK PLAN FOR THE EFFICACY WORKING PARTY (EWP) CHAIRPERSON: Barbara van Zwieten-Boot
 European Medicines Agency London, 17 December 2009 EMA/CHMP/EWP/248088/2009 Rev. 1 WORK PLAN FOR THE EFFICACY WORKING PARTY (EWP) 2010 CHAIRPERSON: Barbara van Zwieten-Boot 1. MEETINGS SCHEDULED FOR 2010
European Medicines Agency London, 17 December 2009 EMA/CHMP/EWP/248088/2009 Rev. 1 WORK PLAN FOR THE EFFICACY WORKING PARTY (EWP) 2010 CHAIRPERSON: Barbara van Zwieten-Boot 1. MEETINGS SCHEDULED FOR 2010
VA/DoD Clinical Practice Guideline for the Diagnosis and Management of Hypertension - Pocket Guide Update 2004 Revision July 2005
 VA/DoD Clinical Practice Guideline for the Diagnosis and Management of Hypertension - Pocket Guide Update 2004 Revision July 2005 1 Any adult in the health care system 2 Obtain blood pressure (BP) (Reliable,
VA/DoD Clinical Practice Guideline for the Diagnosis and Management of Hypertension - Pocket Guide Update 2004 Revision July 2005 1 Any adult in the health care system 2 Obtain blood pressure (BP) (Reliable,
ICH Topic E 4 Dose Response Information to Support Drug Registration. Step 5
 European Medicines Agency November 1994 CPMP/ICH/378/95 ICH Topic E 4 Dose Response Information to Support Drug Registration Step 5 NOTE FOR GUIDANCE ON DOSE RESPONSE INFORMATION TO SUPPORT DRUG REGISTRATION
European Medicines Agency November 1994 CPMP/ICH/378/95 ICH Topic E 4 Dose Response Information to Support Drug Registration Step 5 NOTE FOR GUIDANCE ON DOSE RESPONSE INFORMATION TO SUPPORT DRUG REGISTRATION
LXIV: DRUGS: 4. RAS BLOCKADE
 LXIV: DRUGS: 4. RAS BLOCKADE ACE Inhibitors Components of RAS Actions of Angiotensin i II Indications for ACEIs Contraindications RAS blockade in hypertension RAS blockade in CAD RAS blockade in HF Limitations
LXIV: DRUGS: 4. RAS BLOCKADE ACE Inhibitors Components of RAS Actions of Angiotensin i II Indications for ACEIs Contraindications RAS blockade in hypertension RAS blockade in CAD RAS blockade in HF Limitations
ESSENTIAL HYPERTENSION
 E S S E N T I A L H Y P E R T E N S I O N Elevated blood pressure is one of the most important causes of cardiovascular disease. J A Ker MB ChB, MMed, MD Professor and Deputy Dean Faculty of Health Sciences
E S S E N T I A L H Y P E R T E N S I O N Elevated blood pressure is one of the most important causes of cardiovascular disease. J A Ker MB ChB, MMed, MD Professor and Deputy Dean Faculty of Health Sciences
Clinical Updates in the Treatment of Hypertension JNC 7 vs. JNC 8. Lauren Thomas, PharmD PGY1 Pharmacy Practice Resident South Pointe Hospital
 Clinical Updates in the Treatment of Hypertension JNC 7 vs. JNC 8 Lauren Thomas, PharmD PGY1 Pharmacy Practice Resident South Pointe Hospital Objectives Review the Eighth Joint National Committee (JNC
Clinical Updates in the Treatment of Hypertension JNC 7 vs. JNC 8 Lauren Thomas, PharmD PGY1 Pharmacy Practice Resident South Pointe Hospital Objectives Review the Eighth Joint National Committee (JNC
Managing HTN in the Elderly: How Low to Go
 Managing HTN in the Elderly: How Low to Go Laxmi S. Mehta, MD, FACC The Ohio State University Medical Center Assistant Professor of Clinical Internal Medicine Clinical Director of the Women s Cardiovascular
Managing HTN in the Elderly: How Low to Go Laxmi S. Mehta, MD, FACC The Ohio State University Medical Center Assistant Professor of Clinical Internal Medicine Clinical Director of the Women s Cardiovascular
COMMITTEE FOR PROPRIETARY MEDICINAL PRODUCTS (CPMP) POINTS TO CONSIDER ON MISSING DATA
 The European Agency for the Evaluation of Medicinal Products Evaluation of Medicines for Human Use London, 15 November 2001 CPMP/EWP/1776/99 COMMITTEE FOR PROPRIETARY MEDICINAL PRODUCTS (CPMP) POINTS TO
The European Agency for the Evaluation of Medicinal Products Evaluation of Medicines for Human Use London, 15 November 2001 CPMP/EWP/1776/99 COMMITTEE FOR PROPRIETARY MEDICINAL PRODUCTS (CPMP) POINTS TO
COMMITTEE ON HERBAL MEDICINAL PRODUCTS (HMPC) FINAL COMMUNITY HERBAL MONOGRAPH ON SOLIDAGO VIRGAUREA L., HERBA
 European Medicines Agency Evaluation of Medicines for Human Use London, 4 September 2008 Doc. Ref. EMEA/HMPC/285758/2007 COMMITTEE ON HERBAL MEDICINAL PRODUCTS (HMPC) FINAL COMMUNITY HERBAL MONOGRAPH ON
European Medicines Agency Evaluation of Medicines for Human Use London, 4 September 2008 Doc. Ref. EMEA/HMPC/285758/2007 COMMITTEE ON HERBAL MEDICINAL PRODUCTS (HMPC) FINAL COMMUNITY HERBAL MONOGRAPH ON
This clinical study synopsis is provided in line with Boehringer Ingelheim s Policy on Transparency and Publication of Clinical Study Data.
 abcd Clinical Study Synopsis for Public Disclosure This clinical study synopsis is provided in line with Boehringer Ingelheim s Policy on Transparency and Publication of Clinical Study Data. The synopsis
abcd Clinical Study Synopsis for Public Disclosure This clinical study synopsis is provided in line with Boehringer Ingelheim s Policy on Transparency and Publication of Clinical Study Data. The synopsis
Risk Management Plan
 Management Plan Active substance: VI.2 Elements for a Public Summary VI.2.1 Overview of disease epidemiology Hypertension is generally defined as blood pressure higher than 140/90 mmhg. Hypertension may
Management Plan Active substance: VI.2 Elements for a Public Summary VI.2.1 Overview of disease epidemiology Hypertension is generally defined as blood pressure higher than 140/90 mmhg. Hypertension may
Scientific conclusions and detailed explanation of the scientific grounds for the differences from the PRAC recommendation
 Annex I Scientific conclusions, grounds for variation to the terms of the marketing authorisations and detailed explanation of the scientific grounds for the differences from the PRAC recommendation 1
Annex I Scientific conclusions, grounds for variation to the terms of the marketing authorisations and detailed explanation of the scientific grounds for the differences from the PRAC recommendation 1
Metoprolol Succinate SelokenZOC
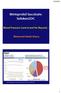 Metoprolol Succinate SelokenZOC Blood Pressure Control and Far Beyond Mohamed Abdel Ghany World Health Organization - Noncommunicable Diseases (NCD) Country Profiles, 2014. 1 Death Rates From Ischemic
Metoprolol Succinate SelokenZOC Blood Pressure Control and Far Beyond Mohamed Abdel Ghany World Health Organization - Noncommunicable Diseases (NCD) Country Profiles, 2014. 1 Death Rates From Ischemic
Angina pectoris due to coronary atherosclerosis : Atenolol is indicated for the long term management of patients with angina pectoris.
 Lonet Tablet Description Lonet contains Atenolol, a synthetic β1 selective (cardioselective) adrenoreceptor blocking agent without membrane stabilising or intrinsic sympathomimetic (partial agonist) activity.
Lonet Tablet Description Lonet contains Atenolol, a synthetic β1 selective (cardioselective) adrenoreceptor blocking agent without membrane stabilising or intrinsic sympathomimetic (partial agonist) activity.
CLINICAL INVESTIGATION OF MEDICAL PRODUCTS IN THE TREATMENT OF GENERALISED ANXIETY DISORDER, PANIC DISORDER AND OBSESSIVE- COMPULSIVE DISORDER
 CLINICAL INVESTIGATION OF MEDICAL PRODUCTS IN THE TREATMENT OF GENERALISED ANXIETY DISORDER, PANIC DISORDER AND OBSESSIVE- COMPULSIVE DISORDER Guideline Title Clinical Investigation of Medical Products
CLINICAL INVESTIGATION OF MEDICAL PRODUCTS IN THE TREATMENT OF GENERALISED ANXIETY DISORDER, PANIC DISORDER AND OBSESSIVE- COMPULSIVE DISORDER Guideline Title Clinical Investigation of Medical Products
ADVANCES IN MANAGEMENT OF HYPERTENSION
 Prevalence 29%; Blacks 33.5% About 72.5% treated; 53.5% uncontrolled (>140/90) Risk for poor control: Latinos, Blacks, age 18-44 and 80,
Prevalence 29%; Blacks 33.5% About 72.5% treated; 53.5% uncontrolled (>140/90) Risk for poor control: Latinos, Blacks, age 18-44 and 80,
Summary of the risk management plan (RMP) for Duaklir Genuair (aclidinium / formoterol fumarate dihydrate)
 EMA/605453/2014 Summary of the risk management plan (RMP) for Duaklir Genuair (aclidinium / formoterol fumarate dihydrate) This is a summary of the risk management plan (RMP) for Duaklir Genuair, which
EMA/605453/2014 Summary of the risk management plan (RMP) for Duaklir Genuair (aclidinium / formoterol fumarate dihydrate) This is a summary of the risk management plan (RMP) for Duaklir Genuair, which
major public health burden
 HYPERTENSION INTRODUCTION Hypertension is one of the major public health burden in the recent times. Hypertension remains a challenging medical condition among the noncommunicable diseases of ever growing
HYPERTENSION INTRODUCTION Hypertension is one of the major public health burden in the recent times. Hypertension remains a challenging medical condition among the noncommunicable diseases of ever growing
2018 OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY. MEASURE TYPE: Process
 Quality ID #317: Preventive Care and Screening: Screening for High Blood Pressure and Follow-Up Documented National Quality Strategy Domain: Community / Population Health 2018 OPTIONS F INDIVIDUAL MEASURES:
Quality ID #317: Preventive Care and Screening: Screening for High Blood Pressure and Follow-Up Documented National Quality Strategy Domain: Community / Population Health 2018 OPTIONS F INDIVIDUAL MEASURES:
Summary of recommendations
 Summary of recommendations Measuring blood pressure (BP) Use the recommended technique at every BP reading to ensure accurate measurements and avoid common errs. Pay particular attention to the following:
Summary of recommendations Measuring blood pressure (BP) Use the recommended technique at every BP reading to ensure accurate measurements and avoid common errs. Pay particular attention to the following:
Reducing proteinuria
 Date written: May 2005 Final submission: October 2005 Author: Adrian Gillin Reducing proteinuria GUIDELINES a. The beneficial effect of treatment regimens that include angiotensinconverting enzyme inhibitors
Date written: May 2005 Final submission: October 2005 Author: Adrian Gillin Reducing proteinuria GUIDELINES a. The beneficial effect of treatment regimens that include angiotensinconverting enzyme inhibitors
Drug treatments in hypertension outcome studies
 The Second Australian National Blood Pressure Study Page 1 of 6 Background Outcome of treatment of hypertension Over the past 25 years studies of the drug treatment of mild-moderate hypertension have demonstrated
The Second Australian National Blood Pressure Study Page 1 of 6 Background Outcome of treatment of hypertension Over the past 25 years studies of the drug treatment of mild-moderate hypertension have demonstrated
Importance of Ambulatory Blood Pressure Monitoring in Adolescents
 Importance of Ambulatory Blood Pressure Monitoring in Adolescents Josep Redon, MD, PhD, FAHA Internal Medicine Hospital Clinico Universitario de Valencia University of Valencia CIBERObn Instituto de Salud
Importance of Ambulatory Blood Pressure Monitoring in Adolescents Josep Redon, MD, PhD, FAHA Internal Medicine Hospital Clinico Universitario de Valencia University of Valencia CIBERObn Instituto de Salud
Guideline on clinical investigation of medicinal products in the treatment of lipid disorders
 19 December 2013 Committee for Medicinal Products for Human Use (CHMP) Guideline on clinical investigation of medicinal products in the treatment of lipid disorders Adopted by CHMP July 2004 Date for coming
19 December 2013 Committee for Medicinal Products for Human Use (CHMP) Guideline on clinical investigation of medicinal products in the treatment of lipid disorders Adopted by CHMP July 2004 Date for coming
Incidental Findings; Management of patients presenting with high BP. Phil Swales
 Incidental Findings; Management of patients presenting with high BP Phil Swales Consultant Physician Acute & General Medicine University Hospitals of Leicester NHS Trust Objectives The approach to an incidental
Incidental Findings; Management of patients presenting with high BP Phil Swales Consultant Physician Acute & General Medicine University Hospitals of Leicester NHS Trust Objectives The approach to an incidental
High-dose monotherapy vs low-dose combination therapy of calcium channel blockers and angiotensin receptor blockers in mild to moderate hypertension
 (2005) 19, 491 496 & 2005 Nature Publishing Group All rights reserved 0950-9240/05 $30.00 www.nature.com/jhh ORIGINAL ARTICLE High-dose monotherapy vs low-dose combination therapy of calcium channel blockers
(2005) 19, 491 496 & 2005 Nature Publishing Group All rights reserved 0950-9240/05 $30.00 www.nature.com/jhh ORIGINAL ARTICLE High-dose monotherapy vs low-dose combination therapy of calcium channel blockers
PRODUCT CIRCULAR. Tablets COZAAR (losartan potassium) I. THERAPEUTIC CLASS II. INDICATIONS III. DOSAGE AND ADMINISTRATION PAK-CZR-T
 PRODUCT CIRCULAR Tablets I. THERAPEUTIC CLASS, the first of a new class of agents for the treatment of hypertension, is an angiotensin II receptor (type AT 1 ) antagonist. also provides a reduction in
PRODUCT CIRCULAR Tablets I. THERAPEUTIC CLASS, the first of a new class of agents for the treatment of hypertension, is an angiotensin II receptor (type AT 1 ) antagonist. also provides a reduction in
COMMITTEE ON HERBAL MEDICINAL PRODUCTS (HMPC) FINAL COMMUNITY HERBAL MONOGRAPH ON AESCULUS HIPPOCASTANUM L., SEMEN
 European Medicines Agency Evaluation of Medicines for Human Use London, 16 July 2009 Doc. Ref.: EMEA/HMPC/225319/2008 COMMITTEE ON HERBAL MEDICINAL PRODUCTS (HMPC) FINAL COMMUNITY HERBAL MONOGRAPH ON AESCULUS
European Medicines Agency Evaluation of Medicines for Human Use London, 16 July 2009 Doc. Ref.: EMEA/HMPC/225319/2008 COMMITTEE ON HERBAL MEDICINAL PRODUCTS (HMPC) FINAL COMMUNITY HERBAL MONOGRAPH ON AESCULUS
ADVANCES IN MANAGEMENT OF HYPERTENSION
 Advances in Management of Robert B. Baron MD Professor of Medicine Associate Dean for GME and CME Declaration of full disclosure: No conflict of interest Current Status of Prevalence 29%; Blacks 33.5%
Advances in Management of Robert B. Baron MD Professor of Medicine Associate Dean for GME and CME Declaration of full disclosure: No conflict of interest Current Status of Prevalence 29%; Blacks 33.5%
COMMITTEE ON HERBAL MEDICINAL PRODUCTS (HMPC) DRAFT COMMUNITY HERBAL MONOGRAPH ON EQUISETUM ARVENSE L., HERBA
 European Medicines Agency Evaluation of Medicines for Human Use London, 31 October 2007 Doc. Ref. EMEA/HMPC/394894/2007 COMMITTEE ON HERBAL MEDICINAL PRODUCTS (HMPC) DRAFT COMMUNITY HERBAL MONOGRAPH ON
European Medicines Agency Evaluation of Medicines for Human Use London, 31 October 2007 Doc. Ref. EMEA/HMPC/394894/2007 COMMITTEE ON HERBAL MEDICINAL PRODUCTS (HMPC) DRAFT COMMUNITY HERBAL MONOGRAPH ON
Long-Term Care Updates
 Long-Term Care Updates August 2015 By Darren Hein, PharmD Hypertension is a clinical condition in which the force of blood pushing on the arteries is higher than normal. This increases the risk for heart
Long-Term Care Updates August 2015 By Darren Hein, PharmD Hypertension is a clinical condition in which the force of blood pushing on the arteries is higher than normal. This increases the risk for heart
DISCLOSURE PHARMACIST OBJECTIVES 9/30/2014 JNC 8: A REVIEW OF THE LONG-AWAITED/MUCH-ANTICIPATED HYPERTENSION GUIDELINES. I have nothing to disclose.
 JNC 8: A REVIEW OF THE LONG-AWAITED/MUCH-ANTICIPATED HYPERTENSION GUIDELINES Tiffany Dickey, PharmD Assistant Professor, UAMS COP Clinical Pharmacy Specialist, Mercy Hospital Northwest AR DISCLOSURE I
JNC 8: A REVIEW OF THE LONG-AWAITED/MUCH-ANTICIPATED HYPERTENSION GUIDELINES Tiffany Dickey, PharmD Assistant Professor, UAMS COP Clinical Pharmacy Specialist, Mercy Hospital Northwest AR DISCLOSURE I
What s In the New Hypertension Guidelines?
 American College of Physicians Ohio/Air Force Chapters 2018 Scientific Meeting Columbus, OH October 5, 2018 What s In the New Hypertension Guidelines? Max C. Reif, MD, FACP Objectives: At the end of the
American College of Physicians Ohio/Air Force Chapters 2018 Scientific Meeting Columbus, OH October 5, 2018 What s In the New Hypertension Guidelines? Max C. Reif, MD, FACP Objectives: At the end of the
The Failing Heart in Primary Care
 The Failing Heart in Primary Care Hamid Ikram How fares the Heart Failure Epidemic? 4357 patients, 57% women, mean age 74 years HFSA 2010 Practice Guideline (3.1) Heart Failure Prevention A careful and
The Failing Heart in Primary Care Hamid Ikram How fares the Heart Failure Epidemic? 4357 patients, 57% women, mean age 74 years HFSA 2010 Practice Guideline (3.1) Heart Failure Prevention A careful and
Individual Study Table Referring to Item of the Submission: Volume: Page:
 2.0 Synopsis Name of Company: Abbott Laboratories Name of Study Drug: Meridia Name of Active Ingredient: Sibutramine hydrochloride monohydrate Individual Study Table Referring to Item of the Submission:
2.0 Synopsis Name of Company: Abbott Laboratories Name of Study Drug: Meridia Name of Active Ingredient: Sibutramine hydrochloride monohydrate Individual Study Table Referring to Item of the Submission:
Hypertension Management Controversies in the Elderly Patient
 Hypertension Management Controversies in the Elderly Patient Juan Bowen, MD Geriatric Update for the Primary Care Provider November 17, 2016 2016 MFMER slide-1 Disclosure No financial relationships No
Hypertension Management Controversies in the Elderly Patient Juan Bowen, MD Geriatric Update for the Primary Care Provider November 17, 2016 2016 MFMER slide-1 Disclosure No financial relationships No
M2 TEACHING UNDERSTANDING PHARMACOLOGY
 M2 TEACHING UNDERSTANDING PHARMACOLOGY USING CVS SYSTEM AS AN EXAMPLE NIGEL FONG 2 JAN 2014 TODAY S OBJECTIVE Pharmacology often seems like an endless list of mechanisms and side effects to memorize. To
M2 TEACHING UNDERSTANDING PHARMACOLOGY USING CVS SYSTEM AS AN EXAMPLE NIGEL FONG 2 JAN 2014 TODAY S OBJECTIVE Pharmacology often seems like an endless list of mechanisms and side effects to memorize. To
...SELECTED ABSTRACTS...
 The following abstracts, from peer-reviewed journals containing literature on vascular compliance and hypertension, were selected for their relevance to this conference and to a managed care perspective.
The following abstracts, from peer-reviewed journals containing literature on vascular compliance and hypertension, were selected for their relevance to this conference and to a managed care perspective.
ICH Topic S1C(R2) Dose Selection for Carcinogenicity Studies of Pharmaceuticals. Step 5
 European Medicines Agency October 2008 EMEA/CHMP/ICH/383/1995 ICH Topic S1C(R2) Dose Selection for Carcinogenicity Studies of Pharmaceuticals Step 5 NOTE FOR GUIDANCE ON DOSE SELECTION FOR CARCINOGENICITY
European Medicines Agency October 2008 EMEA/CHMP/ICH/383/1995 ICH Topic S1C(R2) Dose Selection for Carcinogenicity Studies of Pharmaceuticals Step 5 NOTE FOR GUIDANCE ON DOSE SELECTION FOR CARCINOGENICITY
COMMITTEE FOR MEDICINAL PRODUCTS FOR HUMAN USE (CHMP)
 European Medicines Agency London, 22 February 2007 Doc. Ref. CPMP/EWP/2284/99 Rev. 1 COMMITTEE FOR MEDICINAL PRODUCTS FOR HUMAN USE (CHMP) DRAFT GUIDELINE ON THE DEVELOPMENT OF NEW MEDICINAL PRODUCTS FOR
European Medicines Agency London, 22 February 2007 Doc. Ref. CPMP/EWP/2284/99 Rev. 1 COMMITTEE FOR MEDICINAL PRODUCTS FOR HUMAN USE (CHMP) DRAFT GUIDELINE ON THE DEVELOPMENT OF NEW MEDICINAL PRODUCTS FOR
SECONDARY HYPERTENSION
 HYPERTENSION Hypertension is the clinical term used to describe a high blood pressure of 140/90 mmhg or higher (National Institute of Health 1997). It is such a health risk the World Health Organisation
HYPERTENSION Hypertension is the clinical term used to describe a high blood pressure of 140/90 mmhg or higher (National Institute of Health 1997). It is such a health risk the World Health Organisation
Effects of felodipine on haemodynamics and exercise capacity in patients with angina pectoris
 Br. J. clin. Pharmac. (1987), 23, 391-396 Effects of felodipine on haemodynamics and exercise capacity in patients with angina pectoris J. V. SHERIDAN, P. THOMAS, P. A. ROUTLEDGE & D. J. SHERIDAN Departments
Br. J. clin. Pharmac. (1987), 23, 391-396 Effects of felodipine on haemodynamics and exercise capacity in patients with angina pectoris J. V. SHERIDAN, P. THOMAS, P. A. ROUTLEDGE & D. J. SHERIDAN Departments
IRBES_R_04320 Generic drug name: Irbesartan + amlodipine Date: Study Code: 31 active centers: 20 in Korea, 7 in India and 4 in Philippines.
 These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert in the country of prescription Sponsor/company: sanofi-aventis ClinicalTrials.gov
These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert in the country of prescription Sponsor/company: sanofi-aventis ClinicalTrials.gov
Cardiac Output MCQ. Professor of Cardiovascular Physiology. Cairo University 2007
 Cardiac Output MCQ Abdel Moniem Ibrahim Ahmed, MD Professor of Cardiovascular Physiology Cairo University 2007 90- Guided by Ohm's law when : a- Cardiac output = 5.6 L/min. b- Systolic and diastolic BP
Cardiac Output MCQ Abdel Moniem Ibrahim Ahmed, MD Professor of Cardiovascular Physiology Cairo University 2007 90- Guided by Ohm's law when : a- Cardiac output = 5.6 L/min. b- Systolic and diastolic BP
