Complex ilio-caval revascularization in chronic venous obstruction with the Venovo Stent. Michael K. W. Lichtenberg MD, FESC
|
|
|
- Lawrence Jesse Gallagher
- 5 years ago
- Views:
Transcription
1 Complex ilio-caval revascularization in chronic venous obstruction with the Venovo Stent Michael K. W. Lichtenberg MD, FESC German Venous Center Arnsberg, Germany Not available for sale or distribution in the U.S.
2 Conflict of Interest - Disclosure Within the past 12 months, I or my spouse/partner have had a financial interest/arrangement or affiliation with the organization(s) listed below. Affiliation/Financial Relationship Company 1. Honoraria for lectures: CR Bard, Veniti, AB Medica, Volcano, Optimed GmbH, Straub Medical, Terumo, Biotronik, Veryan 2. Honoraria for advisory board activities: Veniti, Optimed GmbH, Straub Medical, Biotronik, Veryan, Boston Scientific 3. Participation in clinical trials: Biotronik, CR Bard, Veryan, Straub Medical, Veniti, TVA Medical, Boston Scientific, LimFlow 4. Research funding: Biotronik, Boston Scientific, Veryan, Veniti, AB Medica Not available for sale or distribution in the U.S.
3 Current Regulatory Status The VENOVO Venous Stent has CE Mark approval for sale and distribution in Europe. The VENOVO Venous Stent is currently undergoing an investigational device exemption (IDE) in the United States. This device is limited by United States law to investigational use only. This device is not available for sale or distribution in the U.S. Not available for sale or distribution in the U.S.
4 BARD DISCLAIMER The speakers presentation today is on behalf of Bard Peripheral Vascular, Inc. Any discussion regarding Bard products during the presentation today is limited to information that is consistent with the Bard labeling for those products. Please consult Bard product labels and inserts for any indications, contraindications, hazards, warnings, cautions and instructions for use. The opinions and clinical experiences presented herein are for informational purposes only. The results from these case studies may not be predictive for all patients. Individual results may vary depending on a variety of patient specific attributes. These physicians have been compensated by Bard Peripheral Vascular.
5 Venous Stent Attributes Self-expandable Crush resistant across length of stent Sufficient chronic outward force Sufficient wall coverage Flexibility sufficient to resist kink at physiological angles Durability allowing repeated shortening, twisting, and bending at the groin Minimal foreshortening on deployment and balloon dilation Predictable, consistent deployment Strength Flexibility Lumen quality
6 Stent options! Boston Wallstent Optimed Cook Zilver Vena VIVO (EU) Trial Veniti Vici VIRTUS Trial Optimed Sinus obliquus Sinus Obliquus-01-NIS Bard Venovo VERNACULAR Trial Upcoming: Medtronic, Gore, ab Medica, INTACT, Abbott Vascular
7
8
9 Venovo Stent 14 x 120 mm and 14 x 80 mm on both CIV and EIV Not available for sale or distribution in the U.S.
10 Stent Design Self expanding nitinol Flexible, fine tubular mesh prosthesis Design alternates between 3 and 6 nitinol connectors in the main body of the stent 6 markers at each end, 3 are radiopaque tantalum, 3 are nitinol Ends flared 3mm to ensure adequate wall apposition Outward radial force established vessel patency Not available for sale or distribution in the U.S.
11 Venovo Venous Stent Tri-axial system 0.035, over-the-wire Dual speed thumbwheel Large thumbwheel for slow deployment Small thumbwheel for fast deployment Shaft Length: 80 & 120 cm Not available for sale or distribution in the U.S.
12 Stent Lengths Venovo Venous Stent 20 mm 40 mm 60 mm 80 mm 100 mm 120 mm 140 mm 160 mm Stent Diameters 10 mm 12 mm 14 mm 16 mm 18 mm 20 mm 8F 9F 10F This product is not available for sale in the US
13 Visibility Ovine Model, AP View Post stent deployment Ovine Model, AP View Compressed stent prior to deployment Ovine Model, AP View During stent deployment Not available for sale or distribution in the U.S. Images presented above are from a Bard GLP animal study in an ovine model.
14 Not available for sale or distribution in the U.S.
15 Stent Strength Attributes FOCAL LOAD CIRCUMFERENTIAL LOAD Crush Resistance Ability to withstand non-uniform FOCAL load. More closely represents condition of diseased veins. Radial Resistive Force Ability to withstand uniform CIRCUMFERENTIAL load Not closely representative of diseased veins.
16 Radial Resistive Force at 1mm oversize (N/mm) Local Compression Force (N) Radial Force and Crush Resistance Radial Resistive Force Crush Resistance BARD VENOVO 14X160 N=20 OPTIMED SINUS VENOUS 14X80 N=3 COOK ZILVER VENA 14X100 N=2 BARD VENOVO 14X160 N=20 OPTIMED SINUS VENOUS 14X80 N=3 COOK ZILVER VENA 14X100 N=3 Bench testing may not be indicative of clinical performance. Different test methods may yield different results. Competitive testing samples represent commercially available venous stents with CE mark as of June This product is not available for sale in the US
17 3 Point Bending Stiffness (N) Stent Flexibility BARD VENOVO 14X160 OPTIMED SINUS VENOUS 14X80 COOK ZILVER VENA 14X100 Bard Bard N=20 Optimed Sinus Venous N=3 Cook Zilver Vena N=3 Bench testing may not be indicative of clinical performance. Different test methods may yield different results. Competitive testing samples represent commercially available venous stents with CE mark as of June Optimed Cook This product is not available for sale in the US
18 Choose wisely
19 Not available for sale or distribution in the U.S.
20
21 C6, 58 year female with chronic outflow obstruction Not available for sale or distribution in the U.S.
22 Not available for sale or distribution in the U.S.
23 2 Sinus XL Stent (22 x 80 mm) 4 x Venovo Stent (16 x 120 mm + 14 x 60 mm) Not available for sale or distribution in the U.S.
24 34 y, male, PTS, CEAP IV Venovo Stent 16 x 100 mm + 14 x 60 mm Venovo Stent Not available for sale or distribution in the U.S.
25 Take home message General Use dedicated venous stents! Choose wisely - based on lesion morphology Choose wisely based on stent technology Venovo Stent Flexibility: proven Strength: proven Lumen quality: proven
26 Thank you for your attention
27 VENOVO Venous Stent Indication for Use The Venovo Venous Stent System is indicated for the treatment of stenoses and occlusions in the iliac and femoral veins. Contraindications The Venovo Venous Stent System is contraindicated for use in: Patients with a known hypersensitivity to nitinol (nickel-titanium), and tantalum. Patients who cannot receive recommended antiplatelet and/or anti-coagulation therapy. Patients who are judged to have a lesion that prevents complete inflation of a balloon dilatation catheter or proper placement of the stent or the stent delivery system. Warnings The Venovo Venous Stent System is supplied sterile and is intended for SINGLE USE ONLY. DO NOT resterilize and/ or reuse the device. Reuse, resterilization, reprocessing and/or repackaging may create a risk to the patient or user, may lead to infection or compromise the structural integrity and/or essential material and design characteristics of the device, which may lead to device failure, and/or lead to injury, illness, or death of the patient. Reusing this medical device bears the risk of cross-patient contamination as medical devices particularly those with long and small lumina, joints, and/or crevices between components are difficult or impossible to clean once body fluids or tissues with potential pyrogenic or microbial contamination have had contact with the medical device for an indeterminable period of time. The residue of biological material can promote the contamination of the device with pyrogens or microorganisms which may lead to infectious complications or death. DO NOT use in patients with total venous occlusion that can not be dilated to allow passage of the guidewire. DO NOT use the device with contralateral access. DO NOT use if pouch is opened or damaged. DO NOT use the device after the Use By date specified on the label. Persons with allergic reactions to nitinol (nickel-titanium) alloy and/or tantalum may suffer an allergic response to this implant. DO NOT expose the delivery system to organic solvents, e.g., alcohol. The stent is not designed for repositioning or recapturing. Stenting across a major branch could cause difficulties during future diagnostic or therapeutic procedures. If a long lesion needs to be stented consider using the longest available stent rather than overlapping stents. If multiple stents are placed in an overlapping fashion, they should be of similar composition (i.e., nitinol). The long-term outcomes following repeat dilatation of endothelialized stents are unknown. The safety and effectiveness of this device for use in the arterial system have not been established. Not available for sale or distribution in the U.S.
28 VENOVO Venous Stent Precautions The device is intended for use by physicians who have received appropriate training. During system flushing, observe that saline exits at the catheter tip. The delivery system is not designed for use with power injection systems. Recrossing a partially or fully deployed stent with adjunct devices must be performed with caution. Prior to stent deployment, remove slack from the delivery system catheter outside the patient. If excessive force is felt during stent deployment, do not force the delivery system. Remove the delivery system and replace with a new unit. Store in a cool, dark, dry place. Do not attempt to break, damage, or disrupt the stent after placement. P otential Complications and Adverse Events Complications and Adverse Events which may occur include, but are not limited to the following: Allergic/anaphylactic reaction Amputation Aneurysm Arteriovenous fistula Death related to procedure Death unrelated to procedure Dissection Embolization, venous Embolization, stent Extravasation Fever Hemorrhage/bleeding requiring a blood transfusion Hematoma, remote site Hematoma, puncture site Hypotension/hypertension Incorrect positioning of the stent requiring further stenting or surgery Intimal injury/dissection Ischemia/infarction of tissue/organ Local infection Malposition (failure to deliver the stent to the intended site) Open surgical repair Pain Pulmonary embolism Pseudoaneurysm Renal failure Respiratory arrest Restenosis Rupture Septicemia/bacteremia Stent Fracture Stent Migration Vasospasm Venous occlusion/thrombosis, remote from puncture site Venous occlusion/thrombosis, near the puncture site Venous occlusion/restenosis of the treated vessel Please consult package insert for more detailed safety information and instructions for use. Bard and Venovo are trademarks and/or registered trademarks of C. R. Bard, Inc. All other trademarks are property of their respective owners. Copyright 2016, C. R. Bard, Inc. All Rights Reserved. Not available for sale or distribution in the U.S.
29 Complex ilio-caval revascularization in chronic venous obstruction with the Venovo Stent Michael K. W. Lichtenberg MD, FESC German Venous Center Arnsberg, Germany Not available for sale or distribution in the U.S.
REKANALISATION CHRONISCH VENÖSER VERSCHLÜSSE. Michael K. W. Lichtenberg, FESC
 REKANALISATION CHRONISCH VENÖSER VERSCHLÜSSE Michael K. W. Lichtenberg, FESC Conflict of Interest - Disclosure Within the past 12 months, I or my spouse/partner have had a financial interest/arrangement
REKANALISATION CHRONISCH VENÖSER VERSCHLÜSSE Michael K. W. Lichtenberg, FESC Conflict of Interest - Disclosure Within the past 12 months, I or my spouse/partner have had a financial interest/arrangement
Clinical results of venous stents. Michael K. W. Lichtenberg MD, FESC
 Clinical results of venous stents Michael K. W. Lichtenberg MD, FESC Conflict of Interest - Disclosure Within the past 12 months, I or my spouse/partner have had a financial interest/arrangement or affiliation
Clinical results of venous stents Michael K. W. Lichtenberg MD, FESC Conflict of Interest - Disclosure Within the past 12 months, I or my spouse/partner have had a financial interest/arrangement or affiliation
VERNACULAR Trial & Clinical Experience with the VENOVO Venous Stent
 Stephen Black, MD VERNACULAR Trial & Clinical Experience with the VENOVO Venous Stent 1 Speaker Disclaimers The speakers presentation today is on behalf of Bard Peripheral Vascular, Inc. Any discussion
Stephen Black, MD VERNACULAR Trial & Clinical Experience with the VENOVO Venous Stent 1 Speaker Disclaimers The speakers presentation today is on behalf of Bard Peripheral Vascular, Inc. Any discussion
Patency rates and clinical results of the Veniti VICI Stent for treatment of iliac vein lesion Data from the Arnsberg Venous Registry
 Patency rates and clinical results of the Veniti VICI Stent for treatment of iliac vein lesion Data from the Arnsberg Venous Registry Michael K. W. Lichtenberg MD, FESC Conflict of Interest - Disclosure
Patency rates and clinical results of the Veniti VICI Stent for treatment of iliac vein lesion Data from the Arnsberg Venous Registry Michael K. W. Lichtenberg MD, FESC Conflict of Interest - Disclosure
Venous stent experience in Arnsberg Michael K. W. Lichtenberg MD, FESC
 Venous stent experience in Arnsberg Michael K. W. Lichtenberg MD, FESC IMPORTANT INFORMATION: These materials are intended to describe common clinical considerations and procedural steps for the on-label
Venous stent experience in Arnsberg Michael K. W. Lichtenberg MD, FESC IMPORTANT INFORMATION: These materials are intended to describe common clinical considerations and procedural steps for the on-label
VIRTUS: Trial Design and Primary Endpoint Results
 VIRTUS: Trial Design and Primary Endpoint Results Mahmood K. Razavi, MD St. Joseph Cardiac and Vascular Center Orange, CA, USA IMPORTANT INFORMATION: These materials are intended to describe common clinical
VIRTUS: Trial Design and Primary Endpoint Results Mahmood K. Razavi, MD St. Joseph Cardiac and Vascular Center Orange, CA, USA IMPORTANT INFORMATION: These materials are intended to describe common clinical
Future Devices of Venous Interventions
 Future Devices of Venous Interventions Director of Peripheral Vascular Medicine Department of Shin Kong Wu Ho-Su Memorial Hospital, Taiwan Interventional Cardiologist Tien-Yu Wu MD Disclosure Speaker name:...
Future Devices of Venous Interventions Director of Peripheral Vascular Medicine Department of Shin Kong Wu Ho-Su Memorial Hospital, Taiwan Interventional Cardiologist Tien-Yu Wu MD Disclosure Speaker name:...
Assurant Cobalt Iliac BALLOON EXPANDABLE STENT SYSTEM
 Assurant Cobalt Iliac BALLOON EXPANDABLE STENT SYSTEM Innovating for life. CONFORMABILIT Y 6 F S H E AT H C O M PAT I B I L I T Y THE ASSURANT COBALT ILIAC STENT, WITH ITS UNIQUE COBALT CHROMIUM MODULAR
Assurant Cobalt Iliac BALLOON EXPANDABLE STENT SYSTEM Innovating for life. CONFORMABILIT Y 6 F S H E AT H C O M PAT I B I L I T Y THE ASSURANT COBALT ILIAC STENT, WITH ITS UNIQUE COBALT CHROMIUM MODULAR
The SplitWire Percutaneous Transluminal Angioplasty Scoring Device. Instructions for Use
 The SplitWire Percutaneous Transluminal Angioplasty Scoring Device Instructions for Use Contents Contains one (1) SplitWire device. Sterile. Sterilized with ethylene oxide gas. Radiopaque. For single use
The SplitWire Percutaneous Transluminal Angioplasty Scoring Device Instructions for Use Contents Contains one (1) SplitWire device. Sterile. Sterilized with ethylene oxide gas. Radiopaque. For single use
Venous Stents Placed Below the Inguinal Ligament: No Worries
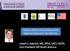 Venous Stents Placed Below the Inguinal Ligament: No Worries Disclosure Speaker name: Lowell S. Kabnick, MD, FACS, FACPh, RPhS... I have the following potential conflicts of interest to report: Consulting:
Venous Stents Placed Below the Inguinal Ligament: No Worries Disclosure Speaker name: Lowell S. Kabnick, MD, FACS, FACPh, RPhS... I have the following potential conflicts of interest to report: Consulting:
Advanced Innovation for Exceptional Performance
 Advanced Innovation for Exceptional Performance longest BALLOON LENGTHS ON THE MARKET Bard Peripheral Vascular Ultraverse 014 300 mm Boston Scientific Coyote 220 mm Cordis SLEEK RX 220 mm Aviator Plus
Advanced Innovation for Exceptional Performance longest BALLOON LENGTHS ON THE MARKET Bard Peripheral Vascular Ultraverse 014 300 mm Boston Scientific Coyote 220 mm Cordis SLEEK RX 220 mm Aviator Plus
Sustained Release. Superior Results.
 ELUVIA Drug-Eluting Vascular Stent System Sustained Release. Superior Results. Superiority determined in Post Hoc Superiority Analysis. 12-Month Primary Patency rate of 86.8% in the Eluvia arm (n=309)
ELUVIA Drug-Eluting Vascular Stent System Sustained Release. Superior Results. Superiority determined in Post Hoc Superiority Analysis. 12-Month Primary Patency rate of 86.8% in the Eluvia arm (n=309)
DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service
 M AY. 6. 2011 10:37 A M F D A - C D R H - O D E - P M O N O. 4147 P. 1 DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service Food and Drug Administration 10903 New Hampshire Avenue Document Control
M AY. 6. 2011 10:37 A M F D A - C D R H - O D E - P M O N O. 4147 P. 1 DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service Food and Drug Administration 10903 New Hampshire Avenue Document Control
DURABLE. CONSISTENT. SAFE. IN.PACT Admiral Drug-Coated Balloon
 DURABLE. CONSISTENT. SAFE. Drug-Coated Balloon THE SCIENCE BEHIND THE OUTCOMES DCB has proven, long-term durable outcomes across multiple clinical trials, as well as across complex patient and lesion types.
DURABLE. CONSISTENT. SAFE. Drug-Coated Balloon THE SCIENCE BEHIND THE OUTCOMES DCB has proven, long-term durable outcomes across multiple clinical trials, as well as across complex patient and lesion types.
Instructions for Use Cordis PRECISE PRO Rx Nitinol Stent System
 100000017144.3 100000017144.3 Instructions for Use Cordis PRECISE PRO Rx Nitinol Stent System Figure 1.065" (1.65 mm) OR.078" (1.98 mm) 16 4 13 10 11 15 8 3 14.062" (1.57 mm) OR.065" (1.65 mm).038" (0.97
100000017144.3 100000017144.3 Instructions for Use Cordis PRECISE PRO Rx Nitinol Stent System Figure 1.065" (1.65 mm) OR.078" (1.98 mm) 16 4 13 10 11 15 8 3 14.062" (1.57 mm) OR.065" (1.65 mm).038" (0.97
Chronic deep venous occlusions: Case planning, recanalization and stent technique
 Chronic deep venous occlusions: Case planning, recanalization and stent technique Michael K. W. Lichtenberg, FESC German Venous Center Arnsberg, Germany Conflict of Interest - Disclosure Within the past
Chronic deep venous occlusions: Case planning, recanalization and stent technique Michael K. W. Lichtenberg, FESC German Venous Center Arnsberg, Germany Conflict of Interest - Disclosure Within the past
Talent Abdominal Stent Graft
 Talent Abdominal with THE Xcelerant Hydro Delivery System Expanding the Indications for EVAR Treat More Patients Short Necks The Talent Abdominal is the only FDA-approved device for proximal aortic neck
Talent Abdominal with THE Xcelerant Hydro Delivery System Expanding the Indications for EVAR Treat More Patients Short Necks The Talent Abdominal is the only FDA-approved device for proximal aortic neck
Proven Performance Through Innovative Design *
 Proven Performance Through Innovative Design * Deliver Our Next Generation AV Covered Stent Results The COVERA Vascular Covered Stent builds upon proven technologies from the category leader in AV Access.
Proven Performance Through Innovative Design * Deliver Our Next Generation AV Covered Stent Results The COVERA Vascular Covered Stent builds upon proven technologies from the category leader in AV Access.
Case Study of Implantation of a VICI VENOUS STENT - Combined NIVL and PTS Stenting
 Case Study of Implantation of a VICI VENOUS STENT - Combined NIVL and PTS Stenting Courtesy of Mr. Stephen Black United kingdom Patient History 25 y/o female 2011: Conservatively treated ilio-femoral DVT
Case Study of Implantation of a VICI VENOUS STENT - Combined NIVL and PTS Stenting Courtesy of Mr. Stephen Black United kingdom Patient History 25 y/o female 2011: Conservatively treated ilio-femoral DVT
COVERA Vascular Covered Stents Innovation in AV Access
 IL0118 Rev.0 1 COVERA Vascular Covered Stents Innovation in AV Access Dheeraj Rajan, MD, FRCPC, FSIR Head and Associate Professor, Division of Vascular & Interventional Radiology Department of Medical
IL0118 Rev.0 1 COVERA Vascular Covered Stents Innovation in AV Access Dheeraj Rajan, MD, FRCPC, FSIR Head and Associate Professor, Division of Vascular & Interventional Radiology Department of Medical
Access More Patients. Customize Each Seal.
 Access More. Customize Each Seal. The Least Invasive Path Towards Proven Patency ULTRA LOW PROFILE TO EASE ADVANCEMENT The flexible, ultra-low 12F ID Ovation ix delivery system enables you to navigate
Access More. Customize Each Seal. The Least Invasive Path Towards Proven Patency ULTRA LOW PROFILE TO EASE ADVANCEMENT The flexible, ultra-low 12F ID Ovation ix delivery system enables you to navigate
RANGER SFA REGISTRY Interim Analysis. Bernd Gehringhoff, MD On behalf the Ranger SFA Registry Investigators
 RANGER SFA REGISTRY Interim Analysis Bernd Gehringhoff, MD On behalf the Ranger SFA Registry Investigators Conflict of Interest - Disclosure Within the past 12 months, I or my spouse/partner have had a
RANGER SFA REGISTRY Interim Analysis Bernd Gehringhoff, MD On behalf the Ranger SFA Registry Investigators Conflict of Interest - Disclosure Within the past 12 months, I or my spouse/partner have had a
PTA Balloon Dilatation Catheter
 PTA Balloon Dilatation Catheter March 2016 1/6(3214-0) Catalogue number Serial number Lot number Use by Do not reuse Do not resterilize Do not use if package is damaged Consult instructions for use Keep
PTA Balloon Dilatation Catheter March 2016 1/6(3214-0) Catalogue number Serial number Lot number Use by Do not reuse Do not resterilize Do not use if package is damaged Consult instructions for use Keep
1 Comparison of Warfarin and Aspirin for Symptomatic Intracranial Arterial Stenosis. N Engl J Med 352;13, March 31, 2005
 The risk of ischemic stroke in patients with Intracranial Atherosclerotic Disease (ICAD) ranges from 7 to 24%. 1,2 Developed specifically for the treatment of ICAD, the Wingspan Stent System and Gateway
The risk of ischemic stroke in patients with Intracranial Atherosclerotic Disease (ICAD) ranges from 7 to 24%. 1,2 Developed specifically for the treatment of ICAD, the Wingspan Stent System and Gateway
Michael K. W. Lichtenberg MD, FESC on behalf of KANSHAS 1 investigators; Tepe G, Müller-Hülsbeck S, Deloose K, Verbist J, Goverde P, Zeller T
 Interim 30-day analysis from the KANSHAS 1 study of the novel KANSHAS drug coated balloon for treatment of femoropopliteal occlusive disease; a latest first-in-human study Michael K. W. Lichtenberg MD,
Interim 30-day analysis from the KANSHAS 1 study of the novel KANSHAS drug coated balloon for treatment of femoropopliteal occlusive disease; a latest first-in-human study Michael K. W. Lichtenberg MD,
System.
 System www.penumbrainc.com POD System Case Examples 45 cm of POD Packing Coil in 1.5 to 3 mm diameter dilating vessel Bronchial Artery Embolization Dr. Amit Kakkar and Dr. Aksim Rivera, Bronx, NY Inflow:
System www.penumbrainc.com POD System Case Examples 45 cm of POD Packing Coil in 1.5 to 3 mm diameter dilating vessel Bronchial Artery Embolization Dr. Amit Kakkar and Dr. Aksim Rivera, Bronx, NY Inflow:
When Outcomes Matter, Design Matters
 to receive our latest news and key activities. Cordis S.M.A.R.T. Self-Expanding Nitinol Lower Extremity Solutions When Outcomes Matter, Design Matters For superficial femoral artery (SFA) and iliac lesions.
to receive our latest news and key activities. Cordis S.M.A.R.T. Self-Expanding Nitinol Lower Extremity Solutions When Outcomes Matter, Design Matters For superficial femoral artery (SFA) and iliac lesions.
Michael K.W. Lichtenberg, MD
 Ranger All-Comer Registry Treatment of femoropopliteal atherosclerotic lesions using the Drug eluting Balloon Ranger: An All Comers Registry Michael K.W. Lichtenberg, MD Klinikum Arnsberg Arnsberg, Germany
Ranger All-Comer Registry Treatment of femoropopliteal atherosclerotic lesions using the Drug eluting Balloon Ranger: An All Comers Registry Michael K.W. Lichtenberg, MD Klinikum Arnsberg Arnsberg, Germany
Ruby Coil. Large Volume Detachable Coils
 Ruby Coil Ruby Case Examples 38 mm Hepatic Artery Aneurysm Pulmonary AVM 2 Coils Dr. James Benenati Miami Cardiac and Vascular Institute, FL Y90 Embolization Type 2 Endoleak Dr. J Moskovitz Florida Hospital,
Ruby Coil Ruby Case Examples 38 mm Hepatic Artery Aneurysm Pulmonary AVM 2 Coils Dr. James Benenati Miami Cardiac and Vascular Institute, FL Y90 Embolization Type 2 Endoleak Dr. J Moskovitz Florida Hospital,
VIVO-EU Results: Prospective European Study of the Zilver Vena TM Venous Stent in the Treatment of Symptomatic Iliofemoral Venous Outflow Obstruction
 VIVO-EU Results: Prospective European Study of the Zilver Vena TM Venous Stent in the Treatment of Symptomatic Iliofemoral Venous Outflow Obstruction Gerard J O Sullivan, M.D. and Jennifer McCann-Brown,
VIVO-EU Results: Prospective European Study of the Zilver Vena TM Venous Stent in the Treatment of Symptomatic Iliofemoral Venous Outflow Obstruction Gerard J O Sullivan, M.D. and Jennifer McCann-Brown,
B05680 Vers. 6/ Figure 2. LifeStent Vascular Stent
 B05680 Vers. 6/12-10 BARD LIFESTENT Stent and Delivery System Vascular Application Recommended Guidewire Length Table Catheter Working Length Recommended Guidewire Length 130 cm 300 cm 80 cm 260 cm 160
B05680 Vers. 6/12-10 BARD LIFESTENT Stent and Delivery System Vascular Application Recommended Guidewire Length Table Catheter Working Length Recommended Guidewire Length 130 cm 300 cm 80 cm 260 cm 160
AXS Catalyst Distal Access Catheter
 AXS Catalyst Distal Access Catheter Directions for Use 2 AXS Catalyst Distal Access Catheter ONLY Caution: Federal Law (USA) restricts this device to sale by or on the order of a physician. warning Contents
AXS Catalyst Distal Access Catheter Directions for Use 2 AXS Catalyst Distal Access Catheter ONLY Caution: Federal Law (USA) restricts this device to sale by or on the order of a physician. warning Contents
Finally, the Control You Need to Deliver Accurate Treatment
 Finally, the Control You Need to Deliver Accurate Treatment LIFESTREAM Covered Stent. has joined BD When you reach for a balloon expandable stent, you require accuracy. The LIFESTREAM Balloon Expandable
Finally, the Control You Need to Deliver Accurate Treatment LIFESTREAM Covered Stent. has joined BD When you reach for a balloon expandable stent, you require accuracy. The LIFESTREAM Balloon Expandable
ER REBOA Catheter. Instructions for Use
 ER REBOA Catheter Instructions for Use Prytime Medical Devices, Inc. 229 N. Main Street Boerne, TX 78006, USA feedback@prytimemedical.com www.prytimemedical.com US 1 210 340 0116 U.S. and Foreign Patents
ER REBOA Catheter Instructions for Use Prytime Medical Devices, Inc. 229 N. Main Street Boerne, TX 78006, USA feedback@prytimemedical.com www.prytimemedical.com US 1 210 340 0116 U.S. and Foreign Patents
The Ideal Venous Stent and Early Results from Venous Stent Trials PNEC, Seattle
 The Ideal Venous Stent and Early Results from Venous Stent Trials 2017 PNEC, Seattle Bill Marston MD Professor, Div of Vascular Surgery University of N. Carolina DISCLOSURES William Marston, MD Consultant/Advisory
The Ideal Venous Stent and Early Results from Venous Stent Trials 2017 PNEC, Seattle Bill Marston MD Professor, Div of Vascular Surgery University of N. Carolina DISCLOSURES William Marston, MD Consultant/Advisory
CP STENT. Large Diameter, Balloon Expandable Stent
 CP STENT Large, Expandable CP STENT OPTIONS 12mm to Expansion 26mm to Expansion CP Matrix (number of zigs) 1.6 2.2 2.8 3.4 3.9 4.5 5 5.5 6 12 14 15 16 18 20 22 24 26 28 30 CP Details: CP is composed of
CP STENT Large, Expandable CP STENT OPTIONS 12mm to Expansion 26mm to Expansion CP Matrix (number of zigs) 1.6 2.2 2.8 3.4 3.9 4.5 5 5.5 6 12 14 15 16 18 20 22 24 26 28 30 CP Details: CP is composed of
- Our patients with iliofemoral DVT - Effective thrombus removal with purely mechanical thrombectomy can lead to better outcomes
 - Our patients with iliofemoral DVT - Effective thrombus removal with purely mechanical thrombectomy can lead to better outcomes Michael K. W. Lichtenberg, FESC Conflict of Interest - Disclosure Within
- Our patients with iliofemoral DVT - Effective thrombus removal with purely mechanical thrombectomy can lead to better outcomes Michael K. W. Lichtenberg, FESC Conflict of Interest - Disclosure Within
PTA Balloon Dilatation Catheter
 PTA Balloon Dilatation Catheter 1/6(3211-0) February 2016 TIS-488-06272017 Catalogue number Serial number Lot number Use by Do not reuse Do not resterilize Do not use if package is damaged Consult instructions
PTA Balloon Dilatation Catheter 1/6(3211-0) February 2016 TIS-488-06272017 Catalogue number Serial number Lot number Use by Do not reuse Do not resterilize Do not use if package is damaged Consult instructions
Bard LifeStar Vascular Stent System
 Bard LifeStar Vascular Stent System Instructions For Use (IFU) Bard LifeStar Vascular Stent System Delivery System Diagram 8x60 B05685 Vers.2/12-11 1 Instructions for use Read the Bard LifeStar Vascular
Bard LifeStar Vascular Stent System Instructions For Use (IFU) Bard LifeStar Vascular Stent System Delivery System Diagram 8x60 B05685 Vers.2/12-11 1 Instructions for use Read the Bard LifeStar Vascular
PERFORMANCE YOU CAN TRUST. EverFlex Self-expanding Peripheral Stent with Entrust Delivery System
 PERFORMANCE YOU CAN TRUST EverFlex Self-expanding Peripheral Stent with Entrust Delivery System The Entrust Delivery System is designed to provide improved patient outcomes and procedural efficiency. The
PERFORMANCE YOU CAN TRUST EverFlex Self-expanding Peripheral Stent with Entrust Delivery System The Entrust Delivery System is designed to provide improved patient outcomes and procedural efficiency. The
Single Pass MCA Revascularization with Trevo
 Single Pass MCA Revascularization with Trevo By Concentric Medical ProVueTM Retriever Case Reported By Sascha Prothmann, MD Klinikum rechts der Isar, Munich, Germany Images contained herein courtesy of
Single Pass MCA Revascularization with Trevo By Concentric Medical ProVueTM Retriever Case Reported By Sascha Prothmann, MD Klinikum rechts der Isar, Munich, Germany Images contained herein courtesy of
FROM THE EVERYDAY TO THE EXTRAORDINARY
 FROM THE EVERYDAY TO THE EXTRAORDINARY Created with the collaboration of more than 250 physicians around the world, ENDURANT empowers you to create stronger outcomes for more patients, including those
FROM THE EVERYDAY TO THE EXTRAORDINARY Created with the collaboration of more than 250 physicians around the world, ENDURANT empowers you to create stronger outcomes for more patients, including those
INSTRUCTIONS FOR USE (IFU)
 INSTRUCTIONS FOR USE (IFU) B05696 Vers.3/11-09 BARD E LUMINEXX Vascular Stent Delivery System Diagrams BARD S.A.F.E. Delivery System with the PERFORMAXX Grip BARD S.A.F.E. Delivery System after removal
INSTRUCTIONS FOR USE (IFU) B05696 Vers.3/11-09 BARD E LUMINEXX Vascular Stent Delivery System Diagrams BARD S.A.F.E. Delivery System with the PERFORMAXX Grip BARD S.A.F.E. Delivery System after removal
NON-COMPLIANT PTCA RAPID EXCHANGE DILATATION CATHETER
 Page 1 of 5 NON-COMPLIANT PTCA RAPID EXCHANGE DILATATION CATHETER STERILE. SINGLE USE ONLY. Sterilized with ethylene oxide gas. Non pyrogenic. Do not resterilize. Do not use opened or damaged packages.
Page 1 of 5 NON-COMPLIANT PTCA RAPID EXCHANGE DILATATION CATHETER STERILE. SINGLE USE ONLY. Sterilized with ethylene oxide gas. Non pyrogenic. Do not resterilize. Do not use opened or damaged packages.
Directions For Use. All directions should be read before use
 Directions For Use All directions should be read before use DEVICE DESCRIPTION: The CLEANER.XT Rotational Thrombectomy System is a percutaneous, 6Fr catheter based system (single piece construction) that
Directions For Use All directions should be read before use DEVICE DESCRIPTION: The CLEANER.XT Rotational Thrombectomy System is a percutaneous, 6Fr catheter based system (single piece construction) that
The Role of LUTONIX 035 DCB in AV Fistula Dysfunction Management in our Practice
 The Role of LUTONIX 035 DCB in AV Fistula Dysfunction Management in our Practice Dr Kate Steiner Consultant Interventional Radiologist East and North Hertfordshire NHS Trust Disclosure Speaker name: Dr
The Role of LUTONIX 035 DCB in AV Fistula Dysfunction Management in our Practice Dr Kate Steiner Consultant Interventional Radiologist East and North Hertfordshire NHS Trust Disclosure Speaker name: Dr
Christian Wissgott MD, PhD Assistant Director, Radiology Westküstenkliniken Heide
 2-Year Results Of The Tack Optimized Balloon Angioplasty (TOBA) Trial For Fem- Pop Lesions Demonstrates Safety and Efficacy Of The Tack Endovascular System In Repairing Focal Post-PTA Dissections Christian
2-Year Results Of The Tack Optimized Balloon Angioplasty (TOBA) Trial For Fem- Pop Lesions Demonstrates Safety and Efficacy Of The Tack Endovascular System In Repairing Focal Post-PTA Dissections Christian
INSTRUCTIONS FOR USE FOR:
 INSTRUCTIONS FOR USE FOR: en English cs Čeština da Dansk nl Nederlands fi Suomi fr Français de Deutsch el Ελληνικά hu Magyar it Italiano lt Lietuvių no Norsk pl Polski pt Português es Español sv Svenska
INSTRUCTIONS FOR USE FOR: en English cs Čeština da Dansk nl Nederlands fi Suomi fr Français de Deutsch el Ελληνικά hu Magyar it Italiano lt Lietuvių no Norsk pl Polski pt Português es Español sv Svenska
Pulsar stent technology
 Pulsar stent technology Michael K. W. Lichtenberg Vascular Center Arnsberg Disclosures Speaker name: Michael Lichtenberg... I have the following potential conflicts of interest to report: X Consulting
Pulsar stent technology Michael K. W. Lichtenberg Vascular Center Arnsberg Disclosures Speaker name: Michael Lichtenberg... I have the following potential conflicts of interest to report: X Consulting
Is a Stent or Scaffold Necessary in The SFA?
 1 2 3 Is a Stent or Scaffold Necessary in The SFA? Stents were developed to optimize acute results after angioplasty Specifically, stents are universally accepted to manage flow limiting dissections and
1 2 3 Is a Stent or Scaffold Necessary in The SFA? Stents were developed to optimize acute results after angioplasty Specifically, stents are universally accepted to manage flow limiting dissections and
1 Description. 2 Indications. 3 Warnings ASPIRATION CATHETER
 Page 1 of 5 ASPIRATION CATHETER Carefully read all instructions prior to use, observe all warnings and precautions noted throughout these instructions. Failure to do so may result in complications. STERILE.
Page 1 of 5 ASPIRATION CATHETER Carefully read all instructions prior to use, observe all warnings and precautions noted throughout these instructions. Failure to do so may result in complications. STERILE.
MICHAEL R. JAFF, DO MASSACHUSETTS, UNITED STATES. Medtronic Further. Together
 DRUG-COATED BALL0ON TREATMENT FOR PATIENTS WITH INTERMITTENT CLAUDICATION: INSIGHTS FROM THE IN.PACT GLOBAL FULL CLINICAL COHORT MICHAEL R. JAFF, DO MASSACHUSETTS, UNITED STATES Medtronic Further. Together
DRUG-COATED BALL0ON TREATMENT FOR PATIENTS WITH INTERMITTENT CLAUDICATION: INSIGHTS FROM THE IN.PACT GLOBAL FULL CLINICAL COHORT MICHAEL R. JAFF, DO MASSACHUSETTS, UNITED STATES Medtronic Further. Together
Understanding of the importance of venous
 The Critical Need for an Iliofemoral Venous Obstruction Classification System An overview of a potential classification system to better identify and treat iliofemoral venous outflow obstruction. BY WILLIAM
The Critical Need for an Iliofemoral Venous Obstruction Classification System An overview of a potential classification system to better identify and treat iliofemoral venous outflow obstruction. BY WILLIAM
JETSTREAM Atherectomy System DELIVERING VERSATILITY TO RESTORE FLOW
 JETSTREAM Atherectomy System DELIVERING VERSATILITY TO RESTORE FLOW DISCOVER THE VALUE OF VERSATILITY Versatility means not having to guess the morphology! Peripheral arterial lesions can present with
JETSTREAM Atherectomy System DELIVERING VERSATILITY TO RESTORE FLOW DISCOVER THE VALUE OF VERSATILITY Versatility means not having to guess the morphology! Peripheral arterial lesions can present with
Lutonix AV Clinical Trial
 Lutonix AV Clinical Trial Long Term Effects of LUTONIX 035 DCB Catheter 18 month Interim Results Scott O. Trerotola, MD Stanley Baum Professor of Radiology Professor of Surgery Associate Chair and Chief,
Lutonix AV Clinical Trial Long Term Effects of LUTONIX 035 DCB Catheter 18 month Interim Results Scott O. Trerotola, MD Stanley Baum Professor of Radiology Professor of Surgery Associate Chair and Chief,
Instructions for Use
 Instructions for Use B05692 Vers.4/09-11 LifeStent Solo Vascular Stent System Figure 1 CAUTION: U.S. federal law restricts this device to sale by or on the order of a physician This device is supplied
Instructions for Use B05692 Vers.4/09-11 LifeStent Solo Vascular Stent System Figure 1 CAUTION: U.S. federal law restricts this device to sale by or on the order of a physician This device is supplied
Balloon in Balloon (BIB )
 Balloon in Balloon (BIB ) Stent Placement Catheter INSTRUCTIONS FOR USE CAUTION: FEDERAL (USA) LAW RESTRICTS THIS DEVICE TO SALE BY OR ON THE ORDER OF A PHYSICIAN. Read all instructions prior to use. Distributed
Balloon in Balloon (BIB ) Stent Placement Catheter INSTRUCTIONS FOR USE CAUTION: FEDERAL (USA) LAW RESTRICTS THIS DEVICE TO SALE BY OR ON THE ORDER OF A PHYSICIAN. Read all instructions prior to use. Distributed
Clinically proven. ordering info. Vascular Intervention // Peripheral Self-Expanding Stent System/0.018 /OTW. Pulsar-18
 140 μm thin struts Clinically proven 4F low profile Vascular Intervention // Peripheral Self-Expanding Stent System/0.018 /OTW Pulsar-18 Technical data / ordering info 140 μm thin struts - thinner than
140 μm thin struts Clinically proven 4F low profile Vascular Intervention // Peripheral Self-Expanding Stent System/0.018 /OTW Pulsar-18 Technical data / ordering info 140 μm thin struts - thinner than
MAXIMIZE RADIAL SOLUTIONS TO PERIPHERAL CHALLENGES
 MAXIMIZE RADIAL SOLUTIONS TO PERIPHERAL CHALLENGES PUSHING BOUNDARIES Terumo Interventional Systems is committed to your success with innovative procedural solutions and ongoing support for your most challenging
MAXIMIZE RADIAL SOLUTIONS TO PERIPHERAL CHALLENGES PUSHING BOUNDARIES Terumo Interventional Systems is committed to your success with innovative procedural solutions and ongoing support for your most challenging
VENOVO Venous Stent Update on the Vernacular Trial
 VENOVO Venous Stent Update on the Vernacular Trial Dr. Sunder Narasimhan, MBBS, MS Senior Consultant Vascular and Endovascular Surgeon Apollo Hospital, Bangalore, India VENOVO is a registered trademark
VENOVO Venous Stent Update on the Vernacular Trial Dr. Sunder Narasimhan, MBBS, MS Senior Consultant Vascular and Endovascular Surgeon Apollo Hospital, Bangalore, India VENOVO is a registered trademark
Advancing Lives and the Delivery of Health Care. The High-Flow Port Designed for Apheresis
 Advancing Lives and the Delivery of Health Care The High-Flow Port Designed for Apheresis Optimized for Long Device Life Bench Tested up to 1,000 Accesses 1 Bard is proud to introduce the first and only
Advancing Lives and the Delivery of Health Care The High-Flow Port Designed for Apheresis Optimized for Long Device Life Bench Tested up to 1,000 Accesses 1 Bard is proud to introduce the first and only
Straub Endovascular System &
 Straub Endovascular System & S t r a u b E n d o v a s c u l a r To o l s Straub Endovascular System Effective debulking in occluded arteries and veins Effective debulking in many indications Rotarex
Straub Endovascular System & S t r a u b E n d o v a s c u l a r To o l s Straub Endovascular System Effective debulking in occluded arteries and veins Effective debulking in many indications Rotarex
Disclosures. Iliac Stenting: How could I mess this up? Surgery vs. Stenting: Gold Standard?
 Disclosures Boston Scientific, Medical Advisory Board Endologix Corp., Consultant and trainer, AFX Aorto-Iliac Disease: Bare Metal, Covered or Stent Grafts How I Decide John S. Lane III MD, FACS Professor
Disclosures Boston Scientific, Medical Advisory Board Endologix Corp., Consultant and trainer, AFX Aorto-Iliac Disease: Bare Metal, Covered or Stent Grafts How I Decide John S. Lane III MD, FACS Professor
REBEL. Platinum Chromium Coronary Stent System. Patient Information Guide
 REBEL Patient Information Guide REBEL PATIENT INFORMATION GUIDE You have recently had a REBEL bare metal stent implanted in the coronary arteries of your heart. The following information is important for
REBEL Patient Information Guide REBEL PATIENT INFORMATION GUIDE You have recently had a REBEL bare metal stent implanted in the coronary arteries of your heart. The following information is important for
Quick Reference Guide
 Quick Reference Guide Indications for Use The AFX Endovascular AAA System is indicated for endovascular treatment in patients with AAA. The devices are indicated for patients with suitable aneurysm morphology
Quick Reference Guide Indications for Use The AFX Endovascular AAA System is indicated for endovascular treatment in patients with AAA. The devices are indicated for patients with suitable aneurysm morphology
GORE EXCLUDER AAA Endoprosthesis ANNUAL CLINICAL UPDATE OCTOBER Section I Clinical experience. Section II Worldwide commercial experience
 GORE EXCLUDER AAA Endoprosthesis ANNUAL CLINICAL UPDATE OCTOBER 2018 Abstract This annual clinical update provides a review of the ongoing experience with the GORE EXCLUDER AAA Endoprosthesis used in the
GORE EXCLUDER AAA Endoprosthesis ANNUAL CLINICAL UPDATE OCTOBER 2018 Abstract This annual clinical update provides a review of the ongoing experience with the GORE EXCLUDER AAA Endoprosthesis used in the
My personal experience with INCRAFT in standard and challenging cases
 My personal experience with INCRAFT in standard and challenging cases G Pratesi, MD Vascular Surgery University of Rome Tor Vergata giovanni.pratesi@uniroma2.it Disclosure Speaker name: Giovanni Pratesi,
My personal experience with INCRAFT in standard and challenging cases G Pratesi, MD Vascular Surgery University of Rome Tor Vergata giovanni.pratesi@uniroma2.it Disclosure Speaker name: Giovanni Pratesi,
Instructions for Use Reprocessed LASSO Circular Mapping Diagnostic Electrophysiology (EP) Catheter
 Instructions for Use Reprocessed LASSO Circular Mapping Diagnostic Electrophysiology (EP) Catheter Caution: Federal (USA) law restricts this device to sale by or on the order of a physician. DEVICE DESCRIPTION
Instructions for Use Reprocessed LASSO Circular Mapping Diagnostic Electrophysiology (EP) Catheter Caution: Federal (USA) law restricts this device to sale by or on the order of a physician. DEVICE DESCRIPTION
Peripheral Intravascular Lithotripsy (IVL) Catheter Instructions for Use (IFU)
 Peripheral Intravascular Lithotripsy (IVL) Catheter Instructions for Use (IFU) For use with the Shockwave Medical, Inc. IVL Generator and Connector Cable Indication for Use The Shockwave Medical IVL System
Peripheral Intravascular Lithotripsy (IVL) Catheter Instructions for Use (IFU) For use with the Shockwave Medical, Inc. IVL Generator and Connector Cable Indication for Use The Shockwave Medical IVL System
Instructions for Use 1
 Instructions for Use 1 Instructions for Use IMPORTANT! Please read before use. Description The Sniper is a dual lumen catheter comprised of an inner guidewire/infusion lumen and an outer, annular lumen
Instructions for Use 1 Instructions for Use IMPORTANT! Please read before use. Description The Sniper is a dual lumen catheter comprised of an inner guidewire/infusion lumen and an outer, annular lumen
Peel-Apart Percutaneous Introducer Kits for
 Bard Access Systems Peel-Apart Percutaneous Introducer Kits for Table of Contents Contents Page Bard Implanted Ports Hickman*, Leonard*, Broviac*, Tenckhoff*, and Groshong* Catheters Introduction....................................
Bard Access Systems Peel-Apart Percutaneous Introducer Kits for Table of Contents Contents Page Bard Implanted Ports Hickman*, Leonard*, Broviac*, Tenckhoff*, and Groshong* Catheters Introduction....................................
Chronic Iliocaval Venous Occlusive Disease
 none Chronic Iliocaval Venous Occlusive Disease David Rigberg, M.D. Clinical Professor of Surgery Division of Vascular Surgery University of California Los Angeles Chronic Venous Occlusive Disease Chronic
none Chronic Iliocaval Venous Occlusive Disease David Rigberg, M.D. Clinical Professor of Surgery Division of Vascular Surgery University of California Los Angeles Chronic Venous Occlusive Disease Chronic
LeMaitre Embolectomy Catheter
 Instructions for Use - English Lemaitre Embolectomy Catheter (Model Numbers 1601-24, e1601-24, 1601-26, e1601-26, 1601-28, e1601-28, 1601-34, e1601-34, 1601-38, e1601-38, 1601-44, e1601-44, 1601-48, e1601-48,
Instructions for Use - English Lemaitre Embolectomy Catheter (Model Numbers 1601-24, e1601-24, 1601-26, e1601-26, 1601-28, e1601-28, 1601-34, e1601-34, 1601-38, e1601-38, 1601-44, e1601-44, 1601-48, e1601-48,
LUTONIX DCB in BTK Update on the BTK clinical program & single center experience
 LUTONIX DCB in BTK Update on the BTK clinical program & single center experience Prof. Dr. med. Dierk Scheinert Department of Interventional Angiology University Hospital Leipzig Disclaimer 1. The information
LUTONIX DCB in BTK Update on the BTK clinical program & single center experience Prof. Dr. med. Dierk Scheinert Department of Interventional Angiology University Hospital Leipzig Disclaimer 1. The information
Mechanical Properties of the GORE TIGRIS Vascular Stent
 Mechanical Properties of the GORE TIGRIS The nitinol frame makes it strong. The fluoropolymers make it flexible. Together, they make it unique. Purpose The association of target vessel restenosis with
Mechanical Properties of the GORE TIGRIS The nitinol frame makes it strong. The fluoropolymers make it flexible. Together, they make it unique. Purpose The association of target vessel restenosis with
Expanding to every demand: The GORE VIABAHN VBX Stent Graft
 Expanding to every demand: The GORE VIABAHN VBX Stent Graft GORE, VIABAHN, and designs are trademarks of W. L. Gore & Associates. 2017 W. L. Gore & Associates, Inc. Program Faculty Martin Austermann, MD
Expanding to every demand: The GORE VIABAHN VBX Stent Graft GORE, VIABAHN, and designs are trademarks of W. L. Gore & Associates. 2017 W. L. Gore & Associates, Inc. Program Faculty Martin Austermann, MD
Patient Brochure. Clearstream Technologies, Ltd. Moyne Upper Enniscorthy Co. Wexford, Ireland. PK Rev. 0 05/17
 Patient Brochure Clearstream Technologies, Ltd. Moyne Upper Enniscorthy Co. Wexford, Ireland PK1411100 Rev. 0 05/17 LIFESTREAM Patient Brochure If you or a member of your family has been diagnosed with
Patient Brochure Clearstream Technologies, Ltd. Moyne Upper Enniscorthy Co. Wexford, Ireland PK1411100 Rev. 0 05/17 LIFESTREAM Patient Brochure If you or a member of your family has been diagnosed with
INTERVENTIONAL VASCULAR DIAGNOSTICS AND THERAPY. VascuFlex Peripheral Stent Systems. Self-Expanding Peripheral Nitinol Stents
 INTERVENTIONAL VASCULAR DIAGNOSTICS AND THERAPY VascuFlex Peripheral Stent Systems Self-Expanding Peripheral Nitinol Stents VascuFlex Multi-LOC MULTIPLE STENT DELIVERY SYSTEM (MSDS) Biomechanical challenges
INTERVENTIONAL VASCULAR DIAGNOSTICS AND THERAPY VascuFlex Peripheral Stent Systems Self-Expanding Peripheral Nitinol Stents VascuFlex Multi-LOC MULTIPLE STENT DELIVERY SYSTEM (MSDS) Biomechanical challenges
Indications for use. Contraindications within the United States
 Indications for use Indications within the United States The GORE TAG Thoracic Endoprosthesis is intended for endovascular repair of all lesions of the descending thoracic aorta, including: Isolated lesions
Indications for use Indications within the United States The GORE TAG Thoracic Endoprosthesis is intended for endovascular repair of all lesions of the descending thoracic aorta, including: Isolated lesions
Polyurethane Dual Lumen Occlusion Catheter English Instructions for Use. Polyurethane Dual Lumen Occlusion Catheter
 Polyurethane Dual Lumen Occlusion Catheter English Instructions for Use Polyurethane Dual Lumen Occlusion Catheter Polyurethane Dual Lumen Occlusion Catheter (Model Numbers 4F-2L-40, 4F-2L-80, 5F-2L-40,
Polyurethane Dual Lumen Occlusion Catheter English Instructions for Use Polyurethane Dual Lumen Occlusion Catheter Polyurethane Dual Lumen Occlusion Catheter (Model Numbers 4F-2L-40, 4F-2L-80, 5F-2L-40,
Innovation by design. Technology that sets a new standard
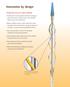 Innovation by design Technology that sets a new standard Flexible nitinol scoring element with three rectangular spiral struts works in tandem with a semi-compliant balloon to score the target lesion Balloon
Innovation by design Technology that sets a new standard Flexible nitinol scoring element with three rectangular spiral struts works in tandem with a semi-compliant balloon to score the target lesion Balloon
Watchman. Left Atrial Appendage Closure Device. Uniquely engineered for the LAA 1-3 with proven safety and longterm efficacy. 4-8
 TM Watchman Left Atrial Appendage Closure Device PROOF OFLEADERSHIP Uniquely engineered for the LAA 1-3 with proven safety and longterm efficacy. 4-8 Patients with AF have a 5x increased risk of stroke.
TM Watchman Left Atrial Appendage Closure Device PROOF OFLEADERSHIP Uniquely engineered for the LAA 1-3 with proven safety and longterm efficacy. 4-8 Patients with AF have a 5x increased risk of stroke.
COVERA Vascular Covered Stents in the Management of Dysfunctional AV Access
 COVERA Vascular Covered Stents in the Management of Dysfunctional AV Access Bart L. Dolmatch, M.D., FSIR Palo Alto Medical Foundation Mountain View, CA USA This presentation is being made on behalf of
COVERA Vascular Covered Stents in the Management of Dysfunctional AV Access Bart L. Dolmatch, M.D., FSIR Palo Alto Medical Foundation Mountain View, CA USA This presentation is being made on behalf of
Per-Q-Cath* PICC Catheters with Excalibur Introducer* System
 Bard Access Systems Per-Q-Cath* PICC and Catheters with Excalibur Introducer* System Instructions For Use Table of Contents Table of Contents Page Contents 1 Product Description, Indications & Contraindications
Bard Access Systems Per-Q-Cath* PICC and Catheters with Excalibur Introducer* System Instructions For Use Table of Contents Table of Contents Page Contents 1 Product Description, Indications & Contraindications
Shaping the future with the swirling flow stent
 NEW SIZES AVAILABLE the swirling flow THE SWIRLING FLOW STENT Significantly better performance than straight control s* in a randomised fempop study ¹ GET WITH THE FLOW the swirling flow IMPROVED BIOMECHANICAL
NEW SIZES AVAILABLE the swirling flow THE SWIRLING FLOW STENT Significantly better performance than straight control s* in a randomised fempop study ¹ GET WITH THE FLOW the swirling flow IMPROVED BIOMECHANICAL
Zenith Renu AAA Converter Graft. Device Description Planning and Sizing Deployment Sequence Patient Follow-Up
 Zenith Renu AAA Converter Graft Device Description Planning and Sizing Deployment Sequence Patient Follow-Up Device description: Device indications The Zenith Renu AAA Converter Graft with Z-Trak Introduction
Zenith Renu AAA Converter Graft Device Description Planning and Sizing Deployment Sequence Patient Follow-Up Device description: Device indications The Zenith Renu AAA Converter Graft with Z-Trak Introduction
Directions For Use. All directions should be read before use. Page 1 of 8
 Directions For Use All directions should be read before use Page 1 of 8 WARNING: For single use only. Do not reuse, reprocess or re-sterilize. Reuse, reprocessing or re-sterilization may compromise the
Directions For Use All directions should be read before use Page 1 of 8 WARNING: For single use only. Do not reuse, reprocess or re-sterilize. Reuse, reprocessing or re-sterilization may compromise the
Allium Trans-hepatic Biliary Stent (BIS)
 Allium Trans-hepatic Biliary (BIS) Instructions for Use Manufactured by Allium Ltd. DEVICE NAME Allium Trans-hepatic Biliary (BIS) DEVICE DESCRIPTION The Trans-hepatic BIS is a flexible, self-expanding
Allium Trans-hepatic Biliary (BIS) Instructions for Use Manufactured by Allium Ltd. DEVICE NAME Allium Trans-hepatic Biliary (BIS) DEVICE DESCRIPTION The Trans-hepatic BIS is a flexible, self-expanding
Azur CX 35 Peripheral Coil System (Detachable) Instructions for Use
 Azur CX 35 Peripheral Coil System (Detachable) Instructions for Use DEVICE DESCRIPTION The Detachable Azur CX 35 Peripheral Coil System (Azur system) consists of a coil implant attached to a delivery system.
Azur CX 35 Peripheral Coil System (Detachable) Instructions for Use DEVICE DESCRIPTION The Detachable Azur CX 35 Peripheral Coil System (Azur system) consists of a coil implant attached to a delivery system.
Update on the Levant 2 Clinical Trial Programme. Dierk Scheinert, MD University Hospital Leipzig Leipzig, Germany
 Update on the Levant 2 Clinical Trial Programme Dierk Scheinert, MD University Hospital Leipzig Leipzig, Germany Disclosure Speaker name: Dierk Scheinert I have the following potential conflicts of interest
Update on the Levant 2 Clinical Trial Programme Dierk Scheinert, MD University Hospital Leipzig Leipzig, Germany Disclosure Speaker name: Dierk Scheinert I have the following potential conflicts of interest
CTO Product Portfolio
 CTO Product Portfolio 1 2 CTO Recanalization Via ATHERECTOMY Occluded anterior tibial artery Tip of Crosser Catheter 3 4 Central lumen vessel recanalization by the Crosser Catheter The Crosser Catheter
CTO Product Portfolio 1 2 CTO Recanalization Via ATHERECTOMY Occluded anterior tibial artery Tip of Crosser Catheter 3 4 Central lumen vessel recanalization by the Crosser Catheter The Crosser Catheter
Excimer Laser for Coronary Intervention: Case Study RADIAL APPROACH: CORONARY LASER ATHERECTOMY FOR CTO OF THE LAD FOLLOWED BY PTCA NO STENTING
 Excimer Laser for Coronary Intervention: Case Study RADIAL APPROACH: CORONARY LASER ATHERECTOMY FOR CTO OF THE LAD FOLLOWED BY PTCA NO STENTING 1 2013 Spectranetics. All Rights Reserved. Approved for External
Excimer Laser for Coronary Intervention: Case Study RADIAL APPROACH: CORONARY LASER ATHERECTOMY FOR CTO OF THE LAD FOLLOWED BY PTCA NO STENTING 1 2013 Spectranetics. All Rights Reserved. Approved for External
Understanding aneurysms and flow diversion treatment
 Surpass Streamline Flow Diverter See package insert for complete indications, contraindications, warnings and instructions for use. INTENDED USE / INDICATIONS FOR USE The Surpass Streamline Flow Diverter
Surpass Streamline Flow Diverter See package insert for complete indications, contraindications, warnings and instructions for use. INTENDED USE / INDICATIONS FOR USE The Surpass Streamline Flow Diverter
THE THE MORE MORE NATURAL APPROACH TO OPTIMAL FIT
 THE THE MORE MORE NATURAL APPROACH Natural Approach TO Optimal Fit TO OPTIMAL FIT Conformability without Compromise THE STANDARD IN Conformability AND Designed for flexibility and conformability in tortuous
THE THE MORE MORE NATURAL APPROACH Natural Approach TO Optimal Fit TO OPTIMAL FIT Conformability without Compromise THE STANDARD IN Conformability AND Designed for flexibility and conformability in tortuous
Indications. The AngioVac cannula is intended for use as a venous drainage cannula and for the removal of fresh, soft thrombi or emboli
 Indications Straight Cannula or Cannula with 20 O Angle 17F Working Channel The AngioVac cannula is intended for use as a venous drainage cannula and for the removal of fresh, soft thrombi or emboli Radiopaque
Indications Straight Cannula or Cannula with 20 O Angle 17F Working Channel The AngioVac cannula is intended for use as a venous drainage cannula and for the removal of fresh, soft thrombi or emboli Radiopaque
Durable outcomes. Proven performance.
 Durable outcomes. Proven performance. GORE EXCLUDER AAA Endoprosthesis GORE EXCLUDER Iliac Branch Endoprosthesis GORE EXCLUDER AAA Endoprosthesis The most-studied* EVAR stent graft designed for durable
Durable outcomes. Proven performance. GORE EXCLUDER AAA Endoprosthesis GORE EXCLUDER Iliac Branch Endoprosthesis GORE EXCLUDER AAA Endoprosthesis The most-studied* EVAR stent graft designed for durable
Bifurcated system Proximal suprarenal stent Modular (aortic main body and two iliac legs) Full thickness woven polyester graft material Fully
 Physician Training Bifurcated system Proximal suprarenal stent Modular (aortic main body and two iliac legs) Full thickness woven polyester graft material Fully supported by self-expanding z-stents H&L-B
Physician Training Bifurcated system Proximal suprarenal stent Modular (aortic main body and two iliac legs) Full thickness woven polyester graft material Fully supported by self-expanding z-stents H&L-B
FLEXIBLE, BALOON EXPANDABLE
 EARLY RESULTS OF A CLINICAL TRIAL OF FLEXIBLE, BALOON EXPANDABLE COVERED STENT GRAFT IN ILIAC OCCLUSIVE DISEASE Chris LeCroy Coastal Vascular and Interventional Pensacola, Florida Clinical Trial WL GORE
EARLY RESULTS OF A CLINICAL TRIAL OF FLEXIBLE, BALOON EXPANDABLE COVERED STENT GRAFT IN ILIAC OCCLUSIVE DISEASE Chris LeCroy Coastal Vascular and Interventional Pensacola, Florida Clinical Trial WL GORE
The High-Flow Port Designed & Indicated for Apheresis
 The High-Flow Port Designed & Indicated for Apheresis Advancing Lives and the Delivery of Health Care Bard is proud to introduce the first and only high-flow, power-injectable port designed and indicated
The High-Flow Port Designed & Indicated for Apheresis Advancing Lives and the Delivery of Health Care Bard is proud to introduce the first and only high-flow, power-injectable port designed and indicated
AXIOS Stent and Delivery System
 PRESCRIPTIVE INFORMATION AXIOS Stent and Delivery System Warning REFER TO THE DEVICE DIRECTIONS FOR USE FOR COMPLETE INSTRUCTIONS ON DEVICE USE. RX ONLY. CAUTION: FEDERAL LAW (USA) RESTRICTS THIS DEVICE
PRESCRIPTIVE INFORMATION AXIOS Stent and Delivery System Warning REFER TO THE DEVICE DIRECTIONS FOR USE FOR COMPLETE INSTRUCTIONS ON DEVICE USE. RX ONLY. CAUTION: FEDERAL LAW (USA) RESTRICTS THIS DEVICE
