Assurant Cobalt Iliac BALLOON EXPANDABLE STENT SYSTEM
|
|
|
- Matilda Gardner
- 5 years ago
- Views:
Transcription
1 Assurant Cobalt Iliac BALLOON EXPANDABLE STENT SYSTEM Innovating for life.
2 CONFORMABILIT Y 6 F S H E AT H C O M PAT I B I L I T Y THE ASSURANT COBALT ILIAC STENT, WITH ITS UNIQUE COBALT CHROMIUM MODULAR DESIGN, PROVIDES EXCELLENT CONFORMABILITY WITHOUT SACRIFICING RADIAL STRENGTH. * Allows for immediate postprocedure MRI imaging (MRI conditional) Greater conformability* Excellent radial strength* By combining its unique cobalt chromium modular design with our patented Secure Technology, Assurant Cobalt Iliac achieves sheath compatibility for every stent size.* sheath compatibility for every stent size Sheath Compatibility Matrix Modular design for providing radial strength with conformability EXPRESS LD Omnilink Elite ASSURANT COBALT ILIAC Increasing Radial Force Radial Force (Compression Force) (N/mm).3 Assurant Cobalt Iliac Express LD Omnilink Elite 7F 7F Lower Crossing Profile design makes the stent extremely trackable.** Increasing Conformability Stent Conformability (Post-Deployment) (N) Smaller Consistent Cell Size* is designed to conform to the vessel wall..8 Medtronic Assurant Cobalt Iliac (8 mm x 6 mm) Medtronic Assurant Cobalt Iliac (8 mm x 6 mm).87 Boston Scientific Express LD (8 mm x 57 mm).85 Abbott Omnilink Elite (8 mm x 59 mm) Boston Scientific Express LD (8 mm x 57 mm) *Bench data on file at Medtronic, Inc., compares the Assurant Cobalt Iliac stent with the Boston Scientific Express LD stent and Abbott Omnilink Elite stent. Bench test results may not be indicative of clinical performance. Cell area bench test was not performed for Abbott Omnilink Elite. *Boston Scientific Express LD IFU and Abbott Omnilink Elite IFU **Bench data on file at Medtronic, Inc., compares the Assurant Cobalt Iliac stent with the Boston Scientific Express LD stent and Abbott Omnilink Elite stent. Bench test results may not be indicative of clinical performance.
3 C L I N I C A L R E S U LT S ACTIVE US CLINICAL TRIAL 9-MONTH FOLLOW-UP IMPROVEMENT IN CLINICAL STATUS, ABI AND WALKING CAPACITY ACTIVE US Clinical Trial Overview Fontaine Class Trial Design Prospective, multicenter, non-randomized Primary Endpoint Major adverse events at s Secondary Endpoints Primary Patency, Acute Success, Clinical Success, ABI, and Capacity at s Number of Subjects 123 subjects with symptomatic PAD Lesions (Procedural) Direct Stenting () 46.8 Mean lesion length ± Post Proc. Stenosis ( ± SD) ± 6.99 Stenosis (most severe) ± Post Procedure MLD (mm ± SD) MLD Primary Endpoint Shows Low MAE Rates Major Adverse Event (MAE) rates include target lesion revascularization (TLR), target vessel revascularization (TVR), target limb loss, and death, including both device-related and procedure-related. During the ACTIVE Trial, one of 123 subjects received a TVR/TLR (.8). No death or target limb loss occurred in the study, thus the composite 9-month MAE is m MAE Death total (device and proc. related)..8.8 Target limb loss TLR TVR * 53.6* (11) (14/141) (119/123) 9-m MAE Death Target TLR total (device and limb loss proc. related) (132/146) TVR (14/141) (119/123) 9.4 (132/146) (/121) Primary patency rate1 Device success2 Lesion success3 Procedural success4 Clinical success5 2 All cause mortality. (/121) Primary patency rate1 Device success2 Lesion success3 Procedural success4 Clinical success5 61.7* Stair impairment distance speed climbing ABI Distribution.6 Cumulative Frequency. I IIa IIb III (N = 147) ACTIVE Data Show Increased Capacity Secondary Endpoints Show 99.3 Primary Patency * (19) Days (122) Months 71.4 (N = 157) All cause mortality 1. I n the ACTIVE study, Primary Patency is reported as 1. It is defined as the blood flow through the treated vessel segment into the distal vasculature (e.g. the common femoral artery and/or the deep femoral artery) as evidenced by duplex ultrasound scan at 3 days and at s for all subjects enrolled with evaluable Duplex scans. Primary patency as defined in this study is independent of interval revascularization procedure. The study definition did not include TLR or TVR. Including TLR and TVR in primary patency calculation yields a primary patency rate of Device success defined as angiographic evidence of <3 final residual stenosis of the target lesion using only the assigned device. 3. Lesion success defined as angiographic evidence of <3 final residual stenosis of the target lesion using any percutaneous method. 4. Procedure success defined as angiographic evidence of <3 final residual stenosis of the target lesion after stent placement and no occurrence of a procedure-related MAE prior to hospital discharge (for subjects with more than one lesion stented the worse case is counted). 5. Clinical success defined as the improvement of Fontaine classification by at least one stage above the pretreated (pre-procedure) clinical value. Assessment, () (N = 147) ABI ± ± (N = 157) Assessment, () of Limbs of Patients 7.62 ± 1.18 I IIa IIb III 4 2 Lesions () Characteristics RVD 79 8 of Patients * * (11) impairment 22.9 distance 45.6* (19) 22.7 speed 61.7* 31.8 (122) Stair climbing All values are mean (n). The walking impairment questionnaire (WIQ) scores range from (unable to perform due to severe claudication) to 1 (no impairment). *P value (baseline vs 9-m) <.1
4 UNIQUE MODULAR DESIGN PROVIDES GREATER CONFORMABILITY The Assurant Cobalt Iliac stent s modular design starts with an advanced cobalt chromium alloy, which is used to create individual rings that are then formed into sinusoidal elements. These elements are laser-fused using customized fusion patterns specifically designed to provide an optimal balance of strength and conformability, then electropolished to create a smooth, edgeless finish. Our patented Secure Technology stent retention system features distal and proximal pillows intended to secure the stent to the catheter during delivery. Indications: The Assurant Cobalt Iliac Balloon-Expandable Stent System is indicated for improving iliac luminal diameter in patients with de novo and restenotic lesions in the common and external iliac arteries with reference vessel diameters between 6mm and 1mm and lesion lengths up to 61mm. The stent is intended as a permanent implant. Contraindications: There are no known contraindications. Warnings and Precautions: The Assurant Cobalt Iliac Balloon-Expandable Stent System is provided sterile for single use only. Do not reuse, reprocess, or resterilize this product. Reuse, reprocessing, or resterilization may compromise the mechanical/structural integrity of the device, compromise the essential material and design characteristics of the device, and/or create a risk of contamination of the device, which could result in patient injury, illness, or death. Do not modify the packaging or labeling as this may lead to the use of an incorrect or expired product, which could result in patient injury or death. Use prior to the Use By date noted on the package. Patients allergic to cobalt-chromium alloy may suffer an allergic reaction to this implant. Administration of appropriate anticoagulant therapy and/or antiplatelet therapy is critical to the success of the procedure. When multiple stents are required and placement may result in overlap or stent-to-stent contact, stent materials should be of similar composition to avoid the potential for dissimilar metal corrosion. Use only appropriate balloon-inflation media. Do not use air or gas to inflate the balloon as it may cause uneven expansion and stent deployment difficulties. Subsequent restenosis may require repeat dilatation of the arterial segment containing the stent. The long-term outcome following repeat dilatation of endothelialized iliac stents is unknown at present. Caution: Read all the instructions carefully in the Instructions for Use. Failure to properly follow the instructions, warnings, and precautions may lead to serious consequences or injury to the patient. Use of the Assurant Cobalt Iliac Balloon-Expandable Stent System requires advanced iliac angioplasty technical skills. The following instructions will provide technical guidance but do not obviate the need for adequate training prior to use of the device. Stent placement should only be performed at hospitals where emergency surgery can be performed. Do not prepare or preinflate the stent delivery balloon prior to stent deployment other than as directed in these Instructions for Use. If the position of the stent is not optimal or appropriate for the vessel, it should not be deployed. The stent cannot be repositioned once deployed. Prior to stent deployment, utilize high resolution fluoroscopy to verify that the stent has not been damaged or dislodged during positioning. The Assurant Cobalt Iliac Balloon-Expandable Stent System is designed for use as a unit. The stent must not be removed from the delivery system. The Assurant Cobalt stent is not designed to be crimped onto another delivery system. Special care must be taken not to handle or in any way disrupt the stent position on the delivery system balloon. Removing the stent from its delivery system or excessively manipulating the stent (eg, rolling the mounted stent) may cause dislodgement, damage to the stent, and stent embolization. Once deployment is initiated, the stent cannot be removed. Prior to completion of the procedure, utilize fluoroscopy to ensure proper positioning of the deployed stent. If the target lesion is not completely stented, use additional Assurant Cobalt Iliac stents as necessary to adequately treat the lesion. Use caution when crossing the stented area with ancillary equipment to avoid dislodgment of the stent. Judicious selection of patients is necessary since the use of this device carries the associated risk of subacute thrombosis, vascular complications, and bleeding events. The Assurant Cobalt Iliac Balloon-Expandable Stent System does not provide for distal contrast injections or pressure measurements through the guidewire lumen. Stent retrieval methods (use of additional wires, snares, or forceps) may result in additional trauma to the vascular access site. Complications may include bleeding, hematoma, or pseudoaneurysm. MRI Safety and Compatibility: The Assurant Cobalt Iliac stent was determined to be MR conditional. It can be scanned safely in both 1.5T & 3.T MR systems under certain conditions as described in the product Instructions for Use. For additional information regarding MR conditions of use, please refer to the product Instructions for Use. Adverse Events: Potential adverse events include (not arranged in any particular order): acute myocardial infarction, allergic reaction, aneurysm, pseudoaneurysm, arteriovenous fistula, angina, arrhythmias, bleeding complications, death, detachment and/or implantation of a component of the system, embolization, emergent or urgent surgery, fever, hematoma or hemorrhagic event, hypotension or hypertension, infection, ischemia, pain at catheter insertion site, pulmonary embolism, renal failure, restenosis, stent malapposition or migration, stent fracture, stent thrombosis, stroke (CVA or TIA), limb loss, vessel dissection, vascular thrombosis or occlusion at puncture site, treatment site, or remote site, vessel spasm, worsening claudication or rest pain. Please reference product Instructions for Use for more information regarding all adverse events. Caution Federal law (USA) restricts this device for sale by or on order of a physician. FDA approved by PMA1111. Assurant Cobalt Iliac balloon-expandable stent system is a registered trademark of Medtronic, Inc. Trademarks are the property of their respective owners.
5 Order Information Assurant Cobalt Iliac Product Code ASC 6 2 SV Catheter Working Length (SV = 8cm, LV = 13cm) Stent Length Stent Product Code Proximal and distal radiopaque markers Stent Minimum Sheath Size (F) Stent Length ASC62SV ASC62LV ASC63SV ASC63LV ASC64SV ASC66SV 7 6 ASC72SV ASC72LV ASC73SV ASC73LV ASC74SV ASC76SV 8 6 ASC82SV ASC82LV ASC83SV ASC83LV ASC84SV ASC86SV 9 6 ASC93SV ASC93LV ASC94SV ASC96SV 1 6 ASC13SV ASC13LV ASC14SV ASC16SV 8cm catheter working length 13cm catheter working length Bilumen shaft Over-the-wire catheter DEPLOYED CONFIGURATIONS ** Nominal Nominal Stent Length Deployed length at nominal pressure Deployed diameter at nominal pressure Assurant Cobalt ILIAC Compliance Pressure **Data on file at Medtronic, Inc. Medtronic Vascular, Inc Unocal Place Santa Rosa, CA 9543 USA Product Services Support Center Tel: Fax: Deployed Stent I.D. (kpa) (atm) Nominal pressure Rated burst pressure The Assurant Cobalt Iliac balloon-expandable stent system is compatible with a maximum guidewire of.35 in. CardioVascular LifeLine Customer Support Tel: Tel: UC212144b EN 213 Medtronic, Inc. All rights reserved. Printed in USA. For distribution in the USA only. 3/13
DURABLE. CONSISTENT. SAFE. IN.PACT Admiral Drug-Coated Balloon
 DURABLE. CONSISTENT. SAFE. Drug-Coated Balloon THE SCIENCE BEHIND THE OUTCOMES DCB has proven, long-term durable outcomes across multiple clinical trials, as well as across complex patient and lesion types.
DURABLE. CONSISTENT. SAFE. Drug-Coated Balloon THE SCIENCE BEHIND THE OUTCOMES DCB has proven, long-term durable outcomes across multiple clinical trials, as well as across complex patient and lesion types.
Talent Abdominal Stent Graft
 Talent Abdominal with THE Xcelerant Hydro Delivery System Expanding the Indications for EVAR Treat More Patients Short Necks The Talent Abdominal is the only FDA-approved device for proximal aortic neck
Talent Abdominal with THE Xcelerant Hydro Delivery System Expanding the Indications for EVAR Treat More Patients Short Necks The Talent Abdominal is the only FDA-approved device for proximal aortic neck
DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service
 M AY. 6. 2011 10:37 A M F D A - C D R H - O D E - P M O N O. 4147 P. 1 DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service Food and Drug Administration 10903 New Hampshire Avenue Document Control
M AY. 6. 2011 10:37 A M F D A - C D R H - O D E - P M O N O. 4147 P. 1 DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service Food and Drug Administration 10903 New Hampshire Avenue Document Control
The SplitWire Percutaneous Transluminal Angioplasty Scoring Device. Instructions for Use
 The SplitWire Percutaneous Transluminal Angioplasty Scoring Device Instructions for Use Contents Contains one (1) SplitWire device. Sterile. Sterilized with ethylene oxide gas. Radiopaque. For single use
The SplitWire Percutaneous Transluminal Angioplasty Scoring Device Instructions for Use Contents Contains one (1) SplitWire device. Sterile. Sterilized with ethylene oxide gas. Radiopaque. For single use
PERFORMANCE YOU CAN TRUST. EverFlex Self-expanding Peripheral Stent with Entrust Delivery System
 PERFORMANCE YOU CAN TRUST EverFlex Self-expanding Peripheral Stent with Entrust Delivery System The Entrust Delivery System is designed to provide improved patient outcomes and procedural efficiency. The
PERFORMANCE YOU CAN TRUST EverFlex Self-expanding Peripheral Stent with Entrust Delivery System The Entrust Delivery System is designed to provide improved patient outcomes and procedural efficiency. The
1 Comparison of Warfarin and Aspirin for Symptomatic Intracranial Arterial Stenosis. N Engl J Med 352;13, March 31, 2005
 The risk of ischemic stroke in patients with Intracranial Atherosclerotic Disease (ICAD) ranges from 7 to 24%. 1,2 Developed specifically for the treatment of ICAD, the Wingspan Stent System and Gateway
The risk of ischemic stroke in patients with Intracranial Atherosclerotic Disease (ICAD) ranges from 7 to 24%. 1,2 Developed specifically for the treatment of ICAD, the Wingspan Stent System and Gateway
MICHAEL R. JAFF, DO MASSACHUSETTS, UNITED STATES. Medtronic Further. Together
 DRUG-COATED BALL0ON TREATMENT FOR PATIENTS WITH INTERMITTENT CLAUDICATION: INSIGHTS FROM THE IN.PACT GLOBAL FULL CLINICAL COHORT MICHAEL R. JAFF, DO MASSACHUSETTS, UNITED STATES Medtronic Further. Together
DRUG-COATED BALL0ON TREATMENT FOR PATIENTS WITH INTERMITTENT CLAUDICATION: INSIGHTS FROM THE IN.PACT GLOBAL FULL CLINICAL COHORT MICHAEL R. JAFF, DO MASSACHUSETTS, UNITED STATES Medtronic Further. Together
Peripheral Intravascular Lithotripsy (IVL) Catheter Instructions for Use (IFU)
 Peripheral Intravascular Lithotripsy (IVL) Catheter Instructions for Use (IFU) For use with the Shockwave Medical, Inc. IVL Generator and Connector Cable Indication for Use The Shockwave Medical IVL System
Peripheral Intravascular Lithotripsy (IVL) Catheter Instructions for Use (IFU) For use with the Shockwave Medical, Inc. IVL Generator and Connector Cable Indication for Use The Shockwave Medical IVL System
FROM THE EVERYDAY TO THE EXTRAORDINARY
 FROM THE EVERYDAY TO THE EXTRAORDINARY Created with the collaboration of more than 250 physicians around the world, ENDURANT empowers you to create stronger outcomes for more patients, including those
FROM THE EVERYDAY TO THE EXTRAORDINARY Created with the collaboration of more than 250 physicians around the world, ENDURANT empowers you to create stronger outcomes for more patients, including those
CP STENT. Large Diameter, Balloon Expandable Stent
 CP STENT Large, Expandable CP STENT OPTIONS 12mm to Expansion 26mm to Expansion CP Matrix (number of zigs) 1.6 2.2 2.8 3.4 3.9 4.5 5 5.5 6 12 14 15 16 18 20 22 24 26 28 30 CP Details: CP is composed of
CP STENT Large, Expandable CP STENT OPTIONS 12mm to Expansion 26mm to Expansion CP Matrix (number of zigs) 1.6 2.2 2.8 3.4 3.9 4.5 5 5.5 6 12 14 15 16 18 20 22 24 26 28 30 CP Details: CP is composed of
Advanced Innovation for Exceptional Performance
 Advanced Innovation for Exceptional Performance longest BALLOON LENGTHS ON THE MARKET Bard Peripheral Vascular Ultraverse 014 300 mm Boston Scientific Coyote 220 mm Cordis SLEEK RX 220 mm Aviator Plus
Advanced Innovation for Exceptional Performance longest BALLOON LENGTHS ON THE MARKET Bard Peripheral Vascular Ultraverse 014 300 mm Boston Scientific Coyote 220 mm Cordis SLEEK RX 220 mm Aviator Plus
Express LD Iliac. Premounted Stent System ONLY. over-the-wire
 50455844-01 STERILE - DO NOT RESTERILIZE - SINGLE USE ONLY ONLY 2017-03 < EN > Express LD Iliac over-the-wire Premounted Stent System Caution: Federal Law (USA) restricts this device to sale by or on the
50455844-01 STERILE - DO NOT RESTERILIZE - SINGLE USE ONLY ONLY 2017-03 < EN > Express LD Iliac over-the-wire Premounted Stent System Caution: Federal Law (USA) restricts this device to sale by or on the
RECOMMENDED INSTRUCTIONS FOR USE
 Rapid Exchange PTCA Dilatation Catheter RECOMMENDED INSTRUCTIONS FOR USE Available in diameters 1.25mm to 4.5mm and in lengths 09mm to 40mm Caution: This device should be used only by physicians trained
Rapid Exchange PTCA Dilatation Catheter RECOMMENDED INSTRUCTIONS FOR USE Available in diameters 1.25mm to 4.5mm and in lengths 09mm to 40mm Caution: This device should be used only by physicians trained
Sustained Release. Superior Results.
 ELUVIA Drug-Eluting Vascular Stent System Sustained Release. Superior Results. Superiority determined in Post Hoc Superiority Analysis. 12-Month Primary Patency rate of 86.8% in the Eluvia arm (n=309)
ELUVIA Drug-Eluting Vascular Stent System Sustained Release. Superior Results. Superiority determined in Post Hoc Superiority Analysis. 12-Month Primary Patency rate of 86.8% in the Eluvia arm (n=309)
Finally, the Control You Need to Deliver Accurate Treatment
 Finally, the Control You Need to Deliver Accurate Treatment LIFESTREAM Covered Stent. has joined BD When you reach for a balloon expandable stent, you require accuracy. The LIFESTREAM Balloon Expandable
Finally, the Control You Need to Deliver Accurate Treatment LIFESTREAM Covered Stent. has joined BD When you reach for a balloon expandable stent, you require accuracy. The LIFESTREAM Balloon Expandable
ER REBOA Catheter. Instructions for Use
 ER REBOA Catheter Instructions for Use Prytime Medical Devices, Inc. 229 N. Main Street Boerne, TX 78006, USA feedback@prytimemedical.com www.prytimemedical.com US 1 210 340 0116 U.S. and Foreign Patents
ER REBOA Catheter Instructions for Use Prytime Medical Devices, Inc. 229 N. Main Street Boerne, TX 78006, USA feedback@prytimemedical.com www.prytimemedical.com US 1 210 340 0116 U.S. and Foreign Patents
MAXIMIZE RADIAL SOLUTIONS TO PERIPHERAL CHALLENGES
 MAXIMIZE RADIAL SOLUTIONS TO PERIPHERAL CHALLENGES PUSHING BOUNDARIES Terumo Interventional Systems is committed to your success with innovative procedural solutions and ongoing support for your most challenging
MAXIMIZE RADIAL SOLUTIONS TO PERIPHERAL CHALLENGES PUSHING BOUNDARIES Terumo Interventional Systems is committed to your success with innovative procedural solutions and ongoing support for your most challenging
REBEL. Platinum Chromium Coronary Stent System. Patient Information Guide
 REBEL Patient Information Guide REBEL PATIENT INFORMATION GUIDE You have recently had a REBEL bare metal stent implanted in the coronary arteries of your heart. The following information is important for
REBEL Patient Information Guide REBEL PATIENT INFORMATION GUIDE You have recently had a REBEL bare metal stent implanted in the coronary arteries of your heart. The following information is important for
PTA Balloon Dilatation Catheter
 PTA Balloon Dilatation Catheter March 2016 1/6(3214-0) Catalogue number Serial number Lot number Use by Do not reuse Do not resterilize Do not use if package is damaged Consult instructions for use Keep
PTA Balloon Dilatation Catheter March 2016 1/6(3214-0) Catalogue number Serial number Lot number Use by Do not reuse Do not resterilize Do not use if package is damaged Consult instructions for use Keep
DISRUPT PAD. (( Data Summary )) DISRUPT PAD Data Summary SPL Rev. B 2016 Shockwave Medical Inc. All rights reserved.
 DISRUPT PAD (( Data Summary )) DISRUPT PAD Data Summary SPL 60971 Rev. B 1 Summary of the key findings from the DISRUPT PAD Study 99% of femoropopliteal lesions treated were moderately or severely calcified.
DISRUPT PAD (( Data Summary )) DISRUPT PAD Data Summary SPL 60971 Rev. B 1 Summary of the key findings from the DISRUPT PAD Study 99% of femoropopliteal lesions treated were moderately or severely calcified.
TOBA II 12-Month Results Tack Optimized Balloon Angioplasty
 TOBA II 12-Month Results Tack Optimized Balloon Angioplasty William Gray, MD System Chief, Cardiovascular Division Main Line Health, Philadelphia, PA Dissection: The Primary Mechanism of Angioplasty Lesions
TOBA II 12-Month Results Tack Optimized Balloon Angioplasty William Gray, MD System Chief, Cardiovascular Division Main Line Health, Philadelphia, PA Dissection: The Primary Mechanism of Angioplasty Lesions
NON-COMPLIANT PTCA RAPID EXCHANGE DILATATION CATHETER
 Page 1 of 5 NON-COMPLIANT PTCA RAPID EXCHANGE DILATATION CATHETER STERILE. SINGLE USE ONLY. Sterilized with ethylene oxide gas. Non pyrogenic. Do not resterilize. Do not use opened or damaged packages.
Page 1 of 5 NON-COMPLIANT PTCA RAPID EXCHANGE DILATATION CATHETER STERILE. SINGLE USE ONLY. Sterilized with ethylene oxide gas. Non pyrogenic. Do not resterilize. Do not use opened or damaged packages.
Instructions for Use Cordis PRECISE PRO Rx Nitinol Stent System
 100000017144.3 100000017144.3 Instructions for Use Cordis PRECISE PRO Rx Nitinol Stent System Figure 1.065" (1.65 mm) OR.078" (1.98 mm) 16 4 13 10 11 15 8 3 14.062" (1.57 mm) OR.065" (1.65 mm).038" (0.97
100000017144.3 100000017144.3 Instructions for Use Cordis PRECISE PRO Rx Nitinol Stent System Figure 1.065" (1.65 mm) OR.078" (1.98 mm) 16 4 13 10 11 15 8 3 14.062" (1.57 mm) OR.065" (1.65 mm).038" (0.97
INSTRUCTIONS FOR USE
 PERICARDIUM COVERED STENT (PCS) INSTRUCTIONS FOR USE STERILE - Ethylene Oxide Sterilized. For single use only. Do not re-sterilize. THE CLINICAL TECHNIQUES AND PROCEDURES DESCRIBED HERE DO NOT REPRESENT
PERICARDIUM COVERED STENT (PCS) INSTRUCTIONS FOR USE STERILE - Ethylene Oxide Sterilized. For single use only. Do not re-sterilize. THE CLINICAL TECHNIQUES AND PROCEDURES DESCRIBED HERE DO NOT REPRESENT
Complex ilio-caval revascularization in chronic venous obstruction with the Venovo Stent. Michael K. W. Lichtenberg MD, FESC
 Complex ilio-caval revascularization in chronic venous obstruction with the Venovo Stent Michael K. W. Lichtenberg MD, FESC German Venous Center Arnsberg, Germany Not available for sale or distribution
Complex ilio-caval revascularization in chronic venous obstruction with the Venovo Stent Michael K. W. Lichtenberg MD, FESC German Venous Center Arnsberg, Germany Not available for sale or distribution
PTA Balloon Dilatation Catheter
 PTA Balloon Dilatation Catheter 1/6(3211-0) February 2016 TIS-488-06272017 Catalogue number Serial number Lot number Use by Do not reuse Do not resterilize Do not use if package is damaged Consult instructions
PTA Balloon Dilatation Catheter 1/6(3211-0) February 2016 TIS-488-06272017 Catalogue number Serial number Lot number Use by Do not reuse Do not resterilize Do not use if package is damaged Consult instructions
Instructions for Use Reprocessed LASSO Circular Mapping Diagnostic Electrophysiology (EP) Catheter
 Instructions for Use Reprocessed LASSO Circular Mapping Diagnostic Electrophysiology (EP) Catheter Caution: Federal (USA) law restricts this device to sale by or on the order of a physician. DEVICE DESCRIPTION
Instructions for Use Reprocessed LASSO Circular Mapping Diagnostic Electrophysiology (EP) Catheter Caution: Federal (USA) law restricts this device to sale by or on the order of a physician. DEVICE DESCRIPTION
AXS Catalyst Distal Access Catheter
 AXS Catalyst Distal Access Catheter Directions for Use 2 AXS Catalyst Distal Access Catheter ONLY Caution: Federal Law (USA) restricts this device to sale by or on the order of a physician. warning Contents
AXS Catalyst Distal Access Catheter Directions for Use 2 AXS Catalyst Distal Access Catheter ONLY Caution: Federal Law (USA) restricts this device to sale by or on the order of a physician. warning Contents
Innovation by design. Technology that sets a new standard
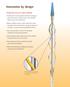 Innovation by design Technology that sets a new standard Flexible nitinol scoring element with three rectangular spiral struts works in tandem with a semi-compliant balloon to score the target lesion Balloon
Innovation by design Technology that sets a new standard Flexible nitinol scoring element with three rectangular spiral struts works in tandem with a semi-compliant balloon to score the target lesion Balloon
Understanding aneurysms and flow diversion treatment
 Surpass Streamline Flow Diverter See package insert for complete indications, contraindications, warnings and instructions for use. INTENDED USE / INDICATIONS FOR USE The Surpass Streamline Flow Diverter
Surpass Streamline Flow Diverter See package insert for complete indications, contraindications, warnings and instructions for use. INTENDED USE / INDICATIONS FOR USE The Surpass Streamline Flow Diverter
Access More Patients. Customize Each Seal.
 Access More. Customize Each Seal. The Least Invasive Path Towards Proven Patency ULTRA LOW PROFILE TO EASE ADVANCEMENT The flexible, ultra-low 12F ID Ovation ix delivery system enables you to navigate
Access More. Customize Each Seal. The Least Invasive Path Towards Proven Patency ULTRA LOW PROFILE TO EASE ADVANCEMENT The flexible, ultra-low 12F ID Ovation ix delivery system enables you to navigate
System.
 System www.penumbrainc.com POD System Case Examples 45 cm of POD Packing Coil in 1.5 to 3 mm diameter dilating vessel Bronchial Artery Embolization Dr. Amit Kakkar and Dr. Aksim Rivera, Bronx, NY Inflow:
System www.penumbrainc.com POD System Case Examples 45 cm of POD Packing Coil in 1.5 to 3 mm diameter dilating vessel Bronchial Artery Embolization Dr. Amit Kakkar and Dr. Aksim Rivera, Bronx, NY Inflow:
NIRxcell CoCr Coronary Stent System
 cordis.com/nirxcell NIRxcell CoCr Coronary Stent System Exceptional Outcomes by Design Exceptionally low TLR rate of 5.1 % at 9 months 1 Superior Deliverability and Crossability 2 Best-in-class Conformability
cordis.com/nirxcell NIRxcell CoCr Coronary Stent System Exceptional Outcomes by Design Exceptionally low TLR rate of 5.1 % at 9 months 1 Superior Deliverability and Crossability 2 Best-in-class Conformability
Christian Wissgott MD, PhD Assistant Director, Radiology Westküstenkliniken Heide
 2-Year Results Of The Tack Optimized Balloon Angioplasty (TOBA) Trial For Fem- Pop Lesions Demonstrates Safety and Efficacy Of The Tack Endovascular System In Repairing Focal Post-PTA Dissections Christian
2-Year Results Of The Tack Optimized Balloon Angioplasty (TOBA) Trial For Fem- Pop Lesions Demonstrates Safety and Efficacy Of The Tack Endovascular System In Repairing Focal Post-PTA Dissections Christian
Balloon in Balloon (BIB )
 Balloon in Balloon (BIB ) Stent Placement Catheter INSTRUCTIONS FOR USE CAUTION: FEDERAL (USA) LAW RESTRICTS THIS DEVICE TO SALE BY OR ON THE ORDER OF A PHYSICIAN. Read all instructions prior to use. Distributed
Balloon in Balloon (BIB ) Stent Placement Catheter INSTRUCTIONS FOR USE CAUTION: FEDERAL (USA) LAW RESTRICTS THIS DEVICE TO SALE BY OR ON THE ORDER OF A PHYSICIAN. Read all instructions prior to use. Distributed
Long Lesions: Primary stenting or DCB first? John Laird MD Adventist Heart and Vascular Institute, St. Helena, CA
 Long Lesions: Primary stenting or DCB first? John Laird MD Adventist Heart and Vascular Institute, St. Helena, CA Disclosures John R. Laird Within the past 12 months, I or my spouse/partner have had a
Long Lesions: Primary stenting or DCB first? John Laird MD Adventist Heart and Vascular Institute, St. Helena, CA Disclosures John R. Laird Within the past 12 months, I or my spouse/partner have had a
VERNACULAR Trial & Clinical Experience with the VENOVO Venous Stent
 Stephen Black, MD VERNACULAR Trial & Clinical Experience with the VENOVO Venous Stent 1 Speaker Disclaimers The speakers presentation today is on behalf of Bard Peripheral Vascular, Inc. Any discussion
Stephen Black, MD VERNACULAR Trial & Clinical Experience with the VENOVO Venous Stent 1 Speaker Disclaimers The speakers presentation today is on behalf of Bard Peripheral Vascular, Inc. Any discussion
1 Description. 2 Indications. 3 Warnings ASPIRATION CATHETER
 Page 1 of 5 ASPIRATION CATHETER Carefully read all instructions prior to use, observe all warnings and precautions noted throughout these instructions. Failure to do so may result in complications. STERILE.
Page 1 of 5 ASPIRATION CATHETER Carefully read all instructions prior to use, observe all warnings and precautions noted throughout these instructions. Failure to do so may result in complications. STERILE.
LIBERTY 360 Study. 15-Jun-2018 Data 1. Olinic Dm, et al. Int Angiol. 2018;37:
 LIBERTY 360 Study LIBERTY is a prospective, observational, multi-center study to evaluate procedural and long-term clinical and economic outcomes of endovascular device interventions in patients with symptomatic
LIBERTY 360 Study LIBERTY is a prospective, observational, multi-center study to evaluate procedural and long-term clinical and economic outcomes of endovascular device interventions in patients with symptomatic
Single Pass MCA Revascularization with Trevo
 Single Pass MCA Revascularization with Trevo By Concentric Medical ProVueTM Retriever Case Reported By Sascha Prothmann, MD Klinikum rechts der Isar, Munich, Germany Images contained herein courtesy of
Single Pass MCA Revascularization with Trevo By Concentric Medical ProVueTM Retriever Case Reported By Sascha Prothmann, MD Klinikum rechts der Isar, Munich, Germany Images contained herein courtesy of
Stellarex Drug-coated 0.035" Angioplasty Balloon. Featuring EnduraCoat Technology. The Clear DCB Choice
 Stellarex Drug-coated 0.035" Angioplasty Balloon Featuring EnduraCoat Technology The Clear DCB Choice Top-tier outcomes with the lowest therapeutic drug dose in all patients common to complex 89.0 % patency
Stellarex Drug-coated 0.035" Angioplasty Balloon Featuring EnduraCoat Technology The Clear DCB Choice Top-tier outcomes with the lowest therapeutic drug dose in all patients common to complex 89.0 % patency
When Outcomes Matter, Design Matters
 to receive our latest news and key activities. Cordis S.M.A.R.T. Self-Expanding Nitinol Lower Extremity Solutions When Outcomes Matter, Design Matters For superficial femoral artery (SFA) and iliac lesions.
to receive our latest news and key activities. Cordis S.M.A.R.T. Self-Expanding Nitinol Lower Extremity Solutions When Outcomes Matter, Design Matters For superficial femoral artery (SFA) and iliac lesions.
Comparison Of Primary Long Stenting Versus Primary Short Stenting For Long Femoropopliteal Artery Disease (PARADE)
 Comparison Of Primary Long Stenting Versus Primary Short Stenting For Long Femoropopliteal Artery Disease (PARADE) Young-Guk Ko, M.D. Severance Cardiovascular Hospital, Yonsei University Health System,
Comparison Of Primary Long Stenting Versus Primary Short Stenting For Long Femoropopliteal Artery Disease (PARADE) Young-Guk Ko, M.D. Severance Cardiovascular Hospital, Yonsei University Health System,
BTK Case Studies Joseph Cardenas, MD AZ Heart & Vascular, Yuma, AZ
 BTK Case Studies Joseph Cardenas, MD AZ Heart & Vascular, Yuma, AZ 1 Case 1 78 yr. old female Rutherford Class II/III lesion 1 block claudicant 2 Pre Treatment Post Treatment Anterior Tibial Artery Occlusion
BTK Case Studies Joseph Cardenas, MD AZ Heart & Vascular, Yuma, AZ 1 Case 1 78 yr. old female Rutherford Class II/III lesion 1 block claudicant 2 Pre Treatment Post Treatment Anterior Tibial Artery Occlusion
Turbo-Power. Laser atherectomy catheter. The standard. for ISR
 Turbo-Power Laser atherectomy catheter The standard for ISR Vaporize the ISR challenge In-stent restenosis (ISR) Chance of recurring 7 115,000 + /year (U.S.) 1-6 Repeated narrowing of the arteries after
Turbo-Power Laser atherectomy catheter The standard for ISR Vaporize the ISR challenge In-stent restenosis (ISR) Chance of recurring 7 115,000 + /year (U.S.) 1-6 Repeated narrowing of the arteries after
Drug-Coated Balloon Treatment for Patients with Intermittent Claudication: Insights from the IN.PACT Global Full Clinical Cohort
 Drug-Coated Balloon Treatment for Patients with Intermittent Claudication: Insights from the IN.PACT Global Full Clinical Cohort a.o. Univ. Prof. Dr. Marianne Brodmann Medical University of Graz Graz,
Drug-Coated Balloon Treatment for Patients with Intermittent Claudication: Insights from the IN.PACT Global Full Clinical Cohort a.o. Univ. Prof. Dr. Marianne Brodmann Medical University of Graz Graz,
Ruby Coil. Large Volume Detachable Coils
 Ruby Coil Ruby Case Examples 38 mm Hepatic Artery Aneurysm Pulmonary AVM 2 Coils Dr. James Benenati Miami Cardiac and Vascular Institute, FL Y90 Embolization Type 2 Endoleak Dr. J Moskovitz Florida Hospital,
Ruby Coil Ruby Case Examples 38 mm Hepatic Artery Aneurysm Pulmonary AVM 2 Coils Dr. James Benenati Miami Cardiac and Vascular Institute, FL Y90 Embolization Type 2 Endoleak Dr. J Moskovitz Florida Hospital,
Directions For Use. All directions should be read before use
 Directions For Use All directions should be read before use DEVICE DESCRIPTION: The CLEANER.XT Rotational Thrombectomy System is a percutaneous, 6Fr catheter based system (single piece construction) that
Directions For Use All directions should be read before use DEVICE DESCRIPTION: The CLEANER.XT Rotational Thrombectomy System is a percutaneous, 6Fr catheter based system (single piece construction) that
B05680 Vers. 6/ Figure 2. LifeStent Vascular Stent
 B05680 Vers. 6/12-10 BARD LIFESTENT Stent and Delivery System Vascular Application Recommended Guidewire Length Table Catheter Working Length Recommended Guidewire Length 130 cm 300 cm 80 cm 260 cm 160
B05680 Vers. 6/12-10 BARD LIFESTENT Stent and Delivery System Vascular Application Recommended Guidewire Length Table Catheter Working Length Recommended Guidewire Length 130 cm 300 cm 80 cm 260 cm 160
LUTONIX DCB in BTK Update on the BTK clinical program & single center experience
 LUTONIX DCB in BTK Update on the BTK clinical program & single center experience Prof. Dr. med. Dierk Scheinert Department of Interventional Angiology University Hospital Leipzig Disclaimer 1. The information
LUTONIX DCB in BTK Update on the BTK clinical program & single center experience Prof. Dr. med. Dierk Scheinert Department of Interventional Angiology University Hospital Leipzig Disclaimer 1. The information
NC EMERGE TM PTCA Dilatation Catheter
 LEARN MORE Indications, contraindications, warnings and instructions for use can be found in the product labeling supplied with each device. Information for use only in countries with applicable health
LEARN MORE Indications, contraindications, warnings and instructions for use can be found in the product labeling supplied with each device. Information for use only in countries with applicable health
Innovating for life. CardioVascular LifeLine Customer Support Tel: Tel:
 Indications The Valiant Thoracic with the Captivia Delivery System is intended for the endovascular repair of fusiform aneurysms and saccular aneurysms/penetrating ulcers of the descending thoracic aorta
Indications The Valiant Thoracic with the Captivia Delivery System is intended for the endovascular repair of fusiform aneurysms and saccular aneurysms/penetrating ulcers of the descending thoracic aorta
Balloon in Balloon (BIB )
 in (BIB ) Dilatation Catheter INSTRUCTIONS FOR USE CAUTION: FEDERAL (USA) LAW RESTRICTS THIS DEVICE TO SALE BY OR ON THE ORDER OF A PHYSICIAN. Read all instructions prior to use. Distributed by: B. Braun
in (BIB ) Dilatation Catheter INSTRUCTIONS FOR USE CAUTION: FEDERAL (USA) LAW RESTRICTS THIS DEVICE TO SALE BY OR ON THE ORDER OF A PHYSICIAN. Read all instructions prior to use. Distributed by: B. Braun
Emerge. PTCA Dilatation Catheter ONLY. monorail. over-the-wire
 90990229-01 Emerge monorail over-the-wire PTCA Dilatation Catheter Table of Contents 2014-09 < EN > warning...1 device description...1 Table 1. Emerge Balloon Coatings...1 Contents...1 Intended use/indications
90990229-01 Emerge monorail over-the-wire PTCA Dilatation Catheter Table of Contents 2014-09 < EN > warning...1 device description...1 Table 1. Emerge Balloon Coatings...1 Contents...1 Intended use/indications
Atherectomy: Jetstream and Directional. George S. Chrysant, M.D.
 Atherectomy: Jetstream and Directional George S. Chrysant, M.D. Disclosures Abbott Vascular: MAB, consultant, proctor Abiomed: consultant Boston Scientific: MAB, consultant, proctor Medicines Company:
Atherectomy: Jetstream and Directional George S. Chrysant, M.D. Disclosures Abbott Vascular: MAB, consultant, proctor Abiomed: consultant Boston Scientific: MAB, consultant, proctor Medicines Company:
INSTRUCTIONS FOR USE FOR:
 INSTRUCTIONS FOR USE FOR: en English cs Čeština da Dansk nl Nederlands fi Suomi fr Français de Deutsch el Ελληνικά hu Magyar it Italiano lt Lietuvių no Norsk pl Polski pt Português es Español sv Svenska
INSTRUCTIONS FOR USE FOR: en English cs Čeština da Dansk nl Nederlands fi Suomi fr Français de Deutsch el Ελληνικά hu Magyar it Italiano lt Lietuvių no Norsk pl Polski pt Português es Español sv Svenska
JETSTREAM Atherectomy System DELIVERING VERSATILITY TO RESTORE FLOW
 JETSTREAM Atherectomy System DELIVERING VERSATILITY TO RESTORE FLOW DISCOVER THE VALUE OF VERSATILITY Versatility means not having to guess the morphology! Peripheral arterial lesions can present with
JETSTREAM Atherectomy System DELIVERING VERSATILITY TO RESTORE FLOW DISCOVER THE VALUE OF VERSATILITY Versatility means not having to guess the morphology! Peripheral arterial lesions can present with
LIFESTREAM TM First look at the BOLSTER 2-year follow up data
 LIFESTREAM TM First look at the BOLSTER 2-year follow up data Dierk Scheinert, MD, on behalf of the Investigators Department of Angiology University Hospital Leipzig, Germany Disclosure Dierk Scheinert,
LIFESTREAM TM First look at the BOLSTER 2-year follow up data Dierk Scheinert, MD, on behalf of the Investigators Department of Angiology University Hospital Leipzig, Germany Disclosure Dierk Scheinert,
Instructions for Use
 Instructions for Use B05692 Vers.4/09-11 LifeStent Solo Vascular Stent System Figure 1 CAUTION: U.S. federal law restricts this device to sale by or on the order of a physician This device is supplied
Instructions for Use B05692 Vers.4/09-11 LifeStent Solo Vascular Stent System Figure 1 CAUTION: U.S. federal law restricts this device to sale by or on the order of a physician This device is supplied
Outcomes Of DCB Use In Real World Registries: 2 Year Results From The INPACT Global Registry
 Outcomes Of DCB Use In Real World Registries: 2 Year Results From The INPACT Global Registry Marianne Brodmann, MD Head of the Clinical Division of Angiology Department of Internal Medicine Medical University
Outcomes Of DCB Use In Real World Registries: 2 Year Results From The INPACT Global Registry Marianne Brodmann, MD Head of the Clinical Division of Angiology Department of Internal Medicine Medical University
Chronic Total Occlusion (CTO) Technologies
 to receive our latest news and key activities. Chronic Total Occlusion (CTO) Technologies Re-open vital channels LinkedIn page Follow us on CORDIS EMEA OUTBACK LTD Re-Entry Catheter True Lumen Re-Entry
to receive our latest news and key activities. Chronic Total Occlusion (CTO) Technologies Re-open vital channels LinkedIn page Follow us on CORDIS EMEA OUTBACK LTD Re-Entry Catheter True Lumen Re-Entry
Shockwave Medical Lithoplasty. Thomas Zeller MD Universitäts-Herzzentrum Freiburg & Bad Krozingen, Germany
 Shockwave Medical Lithoplasty Thomas Zeller MD Universitäts-Herzzentrum Freiburg & Bad Krozingen, Germany Disclosure Speaker name: Thomas Zeller, MD I have the following potential conflicts of interest
Shockwave Medical Lithoplasty Thomas Zeller MD Universitäts-Herzzentrum Freiburg & Bad Krozingen, Germany Disclosure Speaker name: Thomas Zeller, MD I have the following potential conflicts of interest
VIRTUS: Trial Design and Primary Endpoint Results
 VIRTUS: Trial Design and Primary Endpoint Results Mahmood K. Razavi, MD St. Joseph Cardiac and Vascular Center Orange, CA, USA IMPORTANT INFORMATION: These materials are intended to describe common clinical
VIRTUS: Trial Design and Primary Endpoint Results Mahmood K. Razavi, MD St. Joseph Cardiac and Vascular Center Orange, CA, USA IMPORTANT INFORMATION: These materials are intended to describe common clinical
INSTRUCTIONS FOR USE FOR: English
 INSTRUCTIONS FOR USE FOR: H E P A R I N B I O A C T I V E S U R F A C E en English INSTRUCTIONS FOR USE FOR Endoprosthesis Endoprosthesis with Heparin Bioactive Surface Carefully read all instructions
INSTRUCTIONS FOR USE FOR: H E P A R I N B I O A C T I V E S U R F A C E en English INSTRUCTIONS FOR USE FOR Endoprosthesis Endoprosthesis with Heparin Bioactive Surface Carefully read all instructions
XIENCE Sierra INTRODUCING
 INTRODUCING Sierra With the safety you ve always relied on and the deliverability you ve always wanted, choosing the right stent is now an easier decision. START 2017 Abbott. All rights reserved. AP2944678-OUS
INTRODUCING Sierra With the safety you ve always relied on and the deliverability you ve always wanted, choosing the right stent is now an easier decision. START 2017 Abbott. All rights reserved. AP2944678-OUS
COIL SYSTEM ORDERING INFORMATION
 EMBOLIZATION SYSTEM RUBY COIL SYSTEM FRAME with Ruby Standard FILL with Ruby Soft Now with 3 & 40 mm sizes! SYSTEM ANCHOR with Now designed to embolize 3 4 mm vessels! PACK with Coil Now with WAVE Shape
EMBOLIZATION SYSTEM RUBY COIL SYSTEM FRAME with Ruby Standard FILL with Ruby Soft Now with 3 & 40 mm sizes! SYSTEM ANCHOR with Now designed to embolize 3 4 mm vessels! PACK with Coil Now with WAVE Shape
Straub Endovascular System &
 Straub Endovascular System & S t r a u b E n d o v a s c u l a r To o l s Straub Endovascular System Effective debulking in occluded arteries and veins Effective debulking in many indications Rotarex
Straub Endovascular System & S t r a u b E n d o v a s c u l a r To o l s Straub Endovascular System Effective debulking in occluded arteries and veins Effective debulking in many indications Rotarex
PTCA Dilatation Catheter. Instructions for Use
 PTCA Dilatation Catheter Instructions for Use Rx ONLY Caution: Federal law restricts this device to sale by or on the order of a physician STERILE. Sterilized with ethylene oxide gas Distributed by Medinol
PTCA Dilatation Catheter Instructions for Use Rx ONLY Caution: Federal law restricts this device to sale by or on the order of a physician STERILE. Sterilized with ethylene oxide gas Distributed by Medinol
CLARIVEIN INFUSION CATHETER
 CLARIVEIN INFUSION CATHETER General Product Description Overview The ClariVein Infusion Catheter (ClariVein -IC) is an infusion catheter system designed to introduce physician-specified medicaments into
CLARIVEIN INFUSION CATHETER General Product Description Overview The ClariVein Infusion Catheter (ClariVein -IC) is an infusion catheter system designed to introduce physician-specified medicaments into
Balloon Expandable Covered Stents. Suddenly a Crowded Space
 Novel Balloon Expandable Covered Stents for Aorto-iliac Occlusive Disease: New Trial Data makes for an Interesting Balloon Expandable Covered Stents Comparison in Aorto-iliac Occlusive Disease: Andrew
Novel Balloon Expandable Covered Stents for Aorto-iliac Occlusive Disease: New Trial Data makes for an Interesting Balloon Expandable Covered Stents Comparison in Aorto-iliac Occlusive Disease: Andrew
Excimer Laser for Coronary Intervention: Case Study RADIAL APPROACH: CORONARY LASER ATHERECTOMY FOR CTO OF THE LAD FOLLOWED BY PTCA NO STENTING
 Excimer Laser for Coronary Intervention: Case Study RADIAL APPROACH: CORONARY LASER ATHERECTOMY FOR CTO OF THE LAD FOLLOWED BY PTCA NO STENTING 1 2013 Spectranetics. All Rights Reserved. Approved for External
Excimer Laser for Coronary Intervention: Case Study RADIAL APPROACH: CORONARY LASER ATHERECTOMY FOR CTO OF THE LAD FOLLOWED BY PTCA NO STENTING 1 2013 Spectranetics. All Rights Reserved. Approved for External
Update on Tack Optimized Balloon Angioplasty (TOBA) Below the Knee. Marianne Brodmann, MD Medical University Graz Graz, Austria
 Update on Tack Optimized Balloon Angioplasty (TOBA) Below the Knee Marianne Brodmann, MD Medical University Graz Graz, Austria Critical Limb Ischemia Infrapopliteal arterial disease is a leading source
Update on Tack Optimized Balloon Angioplasty (TOBA) Below the Knee Marianne Brodmann, MD Medical University Graz Graz, Austria Critical Limb Ischemia Infrapopliteal arterial disease is a leading source
Lutonix AV Clinical Trial
 Lutonix AV Clinical Trial Long Term Effects of LUTONIX 035 DCB Catheter 18 month Interim Results Scott O. Trerotola, MD Stanley Baum Professor of Radiology Professor of Surgery Associate Chair and Chief,
Lutonix AV Clinical Trial Long Term Effects of LUTONIX 035 DCB Catheter 18 month Interim Results Scott O. Trerotola, MD Stanley Baum Professor of Radiology Professor of Surgery Associate Chair and Chief,
Bifurcated system Proximal suprarenal stent Modular (aortic main body and two iliac legs) Full thickness woven polyester graft material Fully
 Physician Training Bifurcated system Proximal suprarenal stent Modular (aortic main body and two iliac legs) Full thickness woven polyester graft material Fully supported by self-expanding z-stents H&L-B
Physician Training Bifurcated system Proximal suprarenal stent Modular (aortic main body and two iliac legs) Full thickness woven polyester graft material Fully supported by self-expanding z-stents H&L-B
Off-Label Use Expansion: The CTO Example
 Off-Label Use Expansion: The CTO Example Physician/Scientist Perspective Chuck Simonton MD, FACC, FSCAI Chief Medical Officer Divisional Vice President Abbott Vascular Santa Clara, CA TCT 2011 1 Disclosures
Off-Label Use Expansion: The CTO Example Physician/Scientist Perspective Chuck Simonton MD, FACC, FSCAI Chief Medical Officer Divisional Vice President Abbott Vascular Santa Clara, CA TCT 2011 1 Disclosures
My personal experience with INCRAFT in standard and challenging cases
 My personal experience with INCRAFT in standard and challenging cases G Pratesi, MD Vascular Surgery University of Rome Tor Vergata giovanni.pratesi@uniroma2.it Disclosure Speaker name: Giovanni Pratesi,
My personal experience with INCRAFT in standard and challenging cases G Pratesi, MD Vascular Surgery University of Rome Tor Vergata giovanni.pratesi@uniroma2.it Disclosure Speaker name: Giovanni Pratesi,
Device Preparation (all steps to be performed per standard interventional technique)
 sertera Biopsy Device Instructions for Use Please read all information carefully. Failure to properly follow the instructions may lead to unintended consequences. Important: This package insert is designed
sertera Biopsy Device Instructions for Use Please read all information carefully. Failure to properly follow the instructions may lead to unintended consequences. Important: This package insert is designed
GORE EXCLUDER AAA Endoprosthesis ANNUAL CLINICAL UPDATE OCTOBER Section I Clinical experience. Section II Worldwide commercial experience
 GORE EXCLUDER AAA Endoprosthesis ANNUAL CLINICAL UPDATE OCTOBER 2018 Abstract This annual clinical update provides a review of the ongoing experience with the GORE EXCLUDER AAA Endoprosthesis used in the
GORE EXCLUDER AAA Endoprosthesis ANNUAL CLINICAL UPDATE OCTOBER 2018 Abstract This annual clinical update provides a review of the ongoing experience with the GORE EXCLUDER AAA Endoprosthesis used in the
Patient Brochure. Clearstream Technologies, Ltd. Moyne Upper Enniscorthy Co. Wexford, Ireland. PK Rev. 0 05/17
 Patient Brochure Clearstream Technologies, Ltd. Moyne Upper Enniscorthy Co. Wexford, Ireland PK1411100 Rev. 0 05/17 LIFESTREAM Patient Brochure If you or a member of your family has been diagnosed with
Patient Brochure Clearstream Technologies, Ltd. Moyne Upper Enniscorthy Co. Wexford, Ireland PK1411100 Rev. 0 05/17 LIFESTREAM Patient Brochure If you or a member of your family has been diagnosed with
Introduction 3. What is Peripheral Vascular Disease? 5. What Are Some of the Symptoms of Peripheral Vascular Disease? 6
 Patient Information Table of Contents Introduction 3 What is Peripheral Vascular Disease? 5 What Are Some of the Symptoms of Peripheral Vascular Disease? 6 What Causes Peripheral Vascular Disease? 7 How
Patient Information Table of Contents Introduction 3 What is Peripheral Vascular Disease? 5 What Are Some of the Symptoms of Peripheral Vascular Disease? 6 What Causes Peripheral Vascular Disease? 7 How
Introduction What Causes Peripheral Vascular Disease? How Do Doctors Treat Peripheral Vascular Disease?... 9
 Patient Information Table of Contents Introduction... 3 What is Peripheral Vascular Disease?... 5 What Are Some of the Symptoms of Peripheral Vascular Disease?... 7 What Causes Peripheral Vascular Disease?...
Patient Information Table of Contents Introduction... 3 What is Peripheral Vascular Disease?... 5 What Are Some of the Symptoms of Peripheral Vascular Disease?... 7 What Causes Peripheral Vascular Disease?...
The Endurant Stent Graft System
 The Stent Graft System Instructions for Use (IFU) IMPORTANT! Do not attempt to use the Stent Graft System before completely reading and understanding the information contained in the Instructions for Use.
The Stent Graft System Instructions for Use (IFU) IMPORTANT! Do not attempt to use the Stent Graft System before completely reading and understanding the information contained in the Instructions for Use.
Quick Reference Guide
 Quick Reference Guide Indications for Use The AFX Endovascular AAA System is indicated for endovascular treatment in patients with AAA. The devices are indicated for patients with suitable aneurysm morphology
Quick Reference Guide Indications for Use The AFX Endovascular AAA System is indicated for endovascular treatment in patients with AAA. The devices are indicated for patients with suitable aneurysm morphology
PTCA Dilatation Catheter. Instructions for Use
 PTCA Dilatation Catheter Instructions for Use Rx ONLY Caution: Federal law restricts this device to sale by or on the order of a physician STERILE. Sterilized with ethylene oxide gas Distributed by Medinol
PTCA Dilatation Catheter Instructions for Use Rx ONLY Caution: Federal law restricts this device to sale by or on the order of a physician STERILE. Sterilized with ethylene oxide gas Distributed by Medinol
Brachytherapy for In-Stent Restenosis: Is the Concept Still Alive? Matthew T. Menard, M.D. Brigham and Women s Hospital Boston, Massachussetts
 Brachytherapy for In-Stent Restenosis: Is the Concept Still Alive? Matthew T. Menard, M.D. Brigham and Women s Hospital Boston, Massachussetts Disclosure Speaker name: Matthew T. Menard... x I do not have
Brachytherapy for In-Stent Restenosis: Is the Concept Still Alive? Matthew T. Menard, M.D. Brigham and Women s Hospital Boston, Massachussetts Disclosure Speaker name: Matthew T. Menard... x I do not have
DISRUPT CAD. Todd J. Brinton, MD Clinical Associate Professor of Medicine Adjunct Professor of Bioengineering Stanford University
 DISRUPT CAD A multicenter, prospective, single-arm study of percutaneous Lithoplasty prior to stent implantation in heavily calcified coronary lesions Todd J. Brinton, MD Clinical Associate Professor of
DISRUPT CAD A multicenter, prospective, single-arm study of percutaneous Lithoplasty prior to stent implantation in heavily calcified coronary lesions Todd J. Brinton, MD Clinical Associate Professor of
Instructions for Use 1
 Instructions for Use 1 Instructions for Use IMPORTANT! Please read before use. Description The Sniper is a dual lumen catheter comprised of an inner guidewire/infusion lumen and an outer, annular lumen
Instructions for Use 1 Instructions for Use IMPORTANT! Please read before use. Description The Sniper is a dual lumen catheter comprised of an inner guidewire/infusion lumen and an outer, annular lumen
COMPARE-Pilot RCT: 1-year results of a randomised comparison of RANGER DCB vs. IN.PACT DCB in complex SFA lesions. Dierk Scheinert
 COMPARE-Pilot RCT: 1-year results of a randomised comparison of RANGER DCB vs. IN.PACT DCB in complex SFA lesions Dierk Scheinert Department of Angiology University Hospital Leipzig, Germany Disclosure
COMPARE-Pilot RCT: 1-year results of a randomised comparison of RANGER DCB vs. IN.PACT DCB in complex SFA lesions Dierk Scheinert Department of Angiology University Hospital Leipzig, Germany Disclosure
Cardiva Catalyst III INSTRUCTIONS FOR USE
 Cardiva Catalyst III INSTRUCTIONS FOR USE IFU 2469 Rev. G CAUTION Federal (USA) law restricts this device to sale by or on the order of a physician. For single use only GRAPHICAL SYMBOLS ON THE CARDIVA
Cardiva Catalyst III INSTRUCTIONS FOR USE IFU 2469 Rev. G CAUTION Federal (USA) law restricts this device to sale by or on the order of a physician. For single use only GRAPHICAL SYMBOLS ON THE CARDIVA
2019 ABBOTT REIMBURSEMENT GUIDE CMS Physician Fee Schedule
 ABBOTT REIMBURSEMENT GUIDE CMS Physician Fee Schedule This document and the information contained herein is for general information purposes only and is not intended and does not constitute legal, reimbursement,
ABBOTT REIMBURSEMENT GUIDE CMS Physician Fee Schedule This document and the information contained herein is for general information purposes only and is not intended and does not constitute legal, reimbursement,
OCT Guided Atherectomy: Initial Results of the VISION Trial Using the Pantheris Catheter. Patrick Muck, MD
 OCT Guided Atherectomy: Initial Results of the VISION Trial Using the Pantheris Catheter Patrick Muck, MD Chief, Division of Vascular Surgery Good Samaritan Hospital Cincinna
OCT Guided Atherectomy: Initial Results of the VISION Trial Using the Pantheris Catheter Patrick Muck, MD Chief, Division of Vascular Surgery Good Samaritan Hospital Cincinna
Solitaire FR Revascularization Device
 Solitaire FR Revascularization Device FEATURING PARAMETRIC DESIGN Restore. Retrieve. Revive. 5 Revolutionizing Mechanical Thrombectomy with Parametric Design Solitaire FR Device The Solitaire FR revascularization
Solitaire FR Revascularization Device FEATURING PARAMETRIC DESIGN Restore. Retrieve. Revive. 5 Revolutionizing Mechanical Thrombectomy with Parametric Design Solitaire FR Device The Solitaire FR revascularization
Z-MED II BALLOON AORTIC AND PULMONIC VALVULOPLASTY CATHETER. Instructions for Use
 Z-MED II BALLOON AORTIC AND PULMONIC VALVULOPLASTY CATHETER Instructions for Use CAUTION: Federal (USA) Law restricts this device to sale by or on the order of a physician. Manufactured for: B. Braun Interventional
Z-MED II BALLOON AORTIC AND PULMONIC VALVULOPLASTY CATHETER Instructions for Use CAUTION: Federal (USA) Law restricts this device to sale by or on the order of a physician. Manufactured for: B. Braun Interventional
Proven Performance Through Innovative Design *
 Proven Performance Through Innovative Design * Deliver Our Next Generation AV Covered Stent Results The COVERA Vascular Covered Stent builds upon proven technologies from the category leader in AV Access.
Proven Performance Through Innovative Design * Deliver Our Next Generation AV Covered Stent Results The COVERA Vascular Covered Stent builds upon proven technologies from the category leader in AV Access.
Copyright HMP Communications
 Ocelot With Wildcat in a Complicated Superficial Femoral Artery Chronic Total Occlusion Soundos K. Moualla, MD, FACC, FSCAI; Richard R. Heuser, MD, FACC, FACP, FESC, FSCAI From Phoenix Heart Center, Phoenix,
Ocelot With Wildcat in a Complicated Superficial Femoral Artery Chronic Total Occlusion Soundos K. Moualla, MD, FACC, FSCAI; Richard R. Heuser, MD, FACC, FACP, FESC, FSCAI From Phoenix Heart Center, Phoenix,
Atrium Advanta V12. Balloon Expandable Covered Stents
 Atrium Advanta V12 Balloon Expandable Covered Stents This document is intended to provide information to an international audience outside of the US. V12 is not approved or available in the US. Improving
Atrium Advanta V12 Balloon Expandable Covered Stents This document is intended to provide information to an international audience outside of the US. V12 is not approved or available in the US. Improving
Figure 1: Revolution TM Peripheral Atherectomy System Diagram. Table 1: Revolution TM Peripheral Atherectomy System Specifications Minimum Burr
 Instructions For Use All instructions should be read before use CAUTION Investigational Device. Limited by Federal (or United States) law to investigational use. DEVICE DESCRIPTION: The Revolution TM Peripheral
Instructions For Use All instructions should be read before use CAUTION Investigational Device. Limited by Federal (or United States) law to investigational use. DEVICE DESCRIPTION: The Revolution TM Peripheral
Pipeline Embolization Device
 Pipeline Embolization Device The power to redefine aneurysm treatment. REDEFINE The Pipeline device redefines treatment for large or giant wide-necked aneurysms by reconstructing the parent artery and
Pipeline Embolization Device The power to redefine aneurysm treatment. REDEFINE The Pipeline device redefines treatment for large or giant wide-necked aneurysms by reconstructing the parent artery and
Treating In-Stent Restenosis with Brachytherapy: Does it Actually Work?
 Treating In-Stent Restenosis with Brachytherapy: Does it Actually Work? Matthew T. Menard, M.D. Brigham and Women s Hospital Pacific Northwest Endovascular Conference June 15, 2018 DISCLOSURE Matthew Menard,
Treating In-Stent Restenosis with Brachytherapy: Does it Actually Work? Matthew T. Menard, M.D. Brigham and Women s Hospital Pacific Northwest Endovascular Conference June 15, 2018 DISCLOSURE Matthew Menard,
Koen Keirse, MD RZ Tienen, Belgium
 Clinical Benefits of the Vanguard IEP Peripheral Balloon Angioplasty System with Integrated Embolic Protection from the ENTRAP Study Koen Keirse, MD RZ Tienen, Belgium Disclosure Speaker name: Koen Keirse...
Clinical Benefits of the Vanguard IEP Peripheral Balloon Angioplasty System with Integrated Embolic Protection from the ENTRAP Study Koen Keirse, MD RZ Tienen, Belgium Disclosure Speaker name: Koen Keirse...
