VERNACULAR Trial & Clinical Experience with the VENOVO Venous Stent
|
|
|
- Augustus Wilkerson
- 5 years ago
- Views:
Transcription
1 Stephen Black, MD VERNACULAR Trial & Clinical Experience with the VENOVO Venous Stent 1
2 Speaker Disclaimers The speakers presentation today is on behalf of Bard Peripheral Vascular, Inc. Any discussion regarding Bard products during the presentation today is limited to information that is consistent with the Bard labeling for those products. Please consult Bard product labels and inserts for any indications, contraindications, hazards, warnings, cautions and instructions for use. The opinions and clinical experiences presented herein are for informational purposes only. The results from these case studies may not be predictive for all patients. Individual results may vary depending on a variety of patient specific attributes. These physicians have been compensated by Bard Peripheral Vascular. 2
3 VENOVO Venous Stent System The VENOVO Venous Stent has CE Mark approval for sale and distribution in Europe. The VENOVO Venous Stent is currently undergoing an investigational device exemption (IDE) in the United States. This device is limited by United States law to investigational use only. This device is not available for sale or distribution in the U.S. VENOVO Venous Stent Indication for Use: The Venovo Venous Stent System is indicated for the treatment of stenoses and occlusions in the iliac and femoral veins. VENOVO Venous Stent Contraindications: The Venovo Venous Stent System is contraindicated for use in patients with a known hypersensitivity to nitinol (nickel-titanium), and tantalum, who cannot receive recommended antiplatelet and/or anti-coagulation therapy, or who are judged to have a lesion that prevents complete inflation of a balloon dilatation catheter or proper placement of the stent or the stent delivery system. 3
4 Consequences of DVT/PE 60, ,000 in U.S. die each year 1, % die within 1 month of diagnosis 1 33% recurrence over 10 years 1 50% develop post thrombotic syndrome (PTS) 1,2 Swelling, pain, skin discoloration, varicose veins, chronic ulcers Avg. 548,000 hospitalizations each year due to VTE 3 1. Venous Thromboembolism: Data & Statistics. Centers for Disease Control and Prevention. June 22, Retrieved August 24, 2016, from 2. Venous Thromboembolism (VTE). National Center on Birth Defects and Developmental Disabilities 2012 Annual Fiscal Report. Retrieved August 24, 2016 from 3. Venous Thromboembolism in Adult Hospitalizations United States, CDC Morbidity and Mortality Weekly Report. June 8, Retrieved August 24, 2016 from 4
5 VTE Risk Factors Trauma Obesity Pregnancy Hormone Therapy Age Prolonged Immobility Nursing Home Surgery Cancer Oral Contraceptives Smoking Genetic Clotting Disorders Hospitalization Prolonged Travel The Surgeon General s Call to Action to Prevent Deep Vein Thrombosis and Pulmonary Embolism. Office of the Surgeon General (US); National Heart, Lung, and Blood Institute (US)
6 Costs of VTE Hospital claims from 30 hospitals over 7 years % patients had readmission within 1 year 1 Costs 21% higher for readmission Stays longer Annual cost for DVT - $9,321 1 Avg. stay 5.6 days Annual cost for PE - $15,655 1 Avg. stay 7.0 days Avg. cost for DVT + PE - $27,909 1 Annual US healthcare cost of $2-$10 Billion 2 1. Spyropoulos AC, Lin J. Direct Medical Costs of Venous Thromboembolism and Subsequent Hospital Readmission Rates: An Administrative Claims Analysis From 30 Managed Care Organizations. J Manag Care Pharm Jul-Aug;13(6): Annual costs include both primary and secondary diagnosis. 2. Beckman M, et al. Venous Thromboembolism A Public Health Concern. Am J Prev Med Apr;38(4 Suppl):S
7 Venous Stenting in the Literature Meta analysis of 37 studies, 2,869 patients Analyzed stent placement for treatment of iliofemoral venous outflow Primary Patency at 12 months Nonthrombotic lesions: 96% (95% CI, 93% 98%) Acute DVT: 87% (95% CI, 80% 92%) Chronic DVT: 79% (95% CI, 76% 83%) High technical success (94-96%) Low complication rate (<1% bleeding, PE, and mortality) Razavi MK, Jaff MR, Miller LE. Safety and Effectiveness of Stent Placement for Iliofemoral Venous Outflow Obstruction. Circ Cardiovasc Interv Oct;8(10) 7
8 VERNACULAR Trial The BARD VENOVO Venous Stent Study - A Prospective, Non- Randomized, Multi-Center, Single-Arm Study of the Treatment of Iliofemoral Occlusive Disease an Assessment for Effectiveness and Safety Design: Prospective, multi-center, non-randomized, single-arm Core lab & DSMB Purpose: to assess the safety and effectiveness of the VENOVO Venous Stent for the treatment of iliofemoral occlusive disease including Acute or Chronic Deep Vein Thrombosis (DVT), May- Thurner Syndrome, or any combination of the above. Investigative Sites: 35 sites in the US, Europe, and Australia/NZ Subjects: 170 subjects 8
9 Key Inclusion/Exclusion Inclusion Unilateral disease of common femoral, common/external iliac Symptomatic venous outflow obstruction > 50% by venography CEAP >3 or VCSS >2 RVD 7 mm - 19 mm Exclusion Contralateral disease and lesions that extend into IVC or below lesser trochanter Uncorrectable bleeding diathesis or active coagulopathy Previous venous stents Can t cross occlusion 9
10 VERNACULAR Trial Primary endpoints: Primary patency (12 months) Freedom from MAE (30 days) Evaluated against literature derived performance goals Key secondary endpoints: VCSS/ CEAP/ QoL assessment Procedure/technical success Freedom from TVR/TLR Primary patency at 24 and 36 mo. Stent fracture 10
11 VERNACULAR : VENOVO Stent IDE Clinical Study 10 US Sites 7 OUS sites Enrollment at 77 patients 11 11
12 The ATTRACT Study Acute Venous Thrombosis: Thrombus Removal with Adjunctive Catheter-Directed Thrombolysis BPV/STNT/0916/0051 NIH-funded, Phase III, multicenter, open-label, assessor-blinded, randomized controlled trial Randomized to receive PMT + standard therapy vs. standard therapy alone 692 patients with symptomatic proximal iliofemoral DVT 58 study sites, enrollment closed Primary endpoints: Cumulative incidence of PTS (Villalta Scale) at 24 months Secondary endpoints: Severity of PTS, DVT symptoms, valve reflux, residual thrombus, clot lysis, cost effectiveness Major bleeding, symptomatic PE, recurrent VTE, death Vedantham S, et al. Rationale and design of the ATTRACT Study: a multicenter randomized trial to evaluate pharmacomechanical catheter-directed thrombolysis for the prevention of postthrombotic syndrome in patients with proximal deep vein thrombosis. Am Heart J Apr;165(4): ClinicalTrials.gov NCT
13 May-Thurner Syndrome Compression of left iliac vein by right iliac artery 22-24% of lower extremity DVT cases Prevalence may be underestimated Peters M, Syed RK, Katz M, et al. May-Thurner syndrome: a not so uncommon cause of a common condition. Proc (Bayl Univ Med Cent) July;25(3):
14 VENOVO Stent Delivery System 14
15 Deployment 15
16 Clinical Use Visibility is good Deployment is easy with minimal foreshortening Long term data will come from the VERNACULAR Study Key points: May-Thurner Pelvis Ligament 16
17 Flexibility BPV/STNT/0916/
18 Summary Slide VTE and PTS are major health and economic concerns Current clinical guidelines vary on appropriate treatment algorithms Multiple endovascular treatment options needed VENOVO Venous Stent is being investigated for treatment of iliofemoral disease 18
19 Indication for Use: The Venovo Venous Stent System is indicated for the treatment of stenoses and occlusions in the iliac and femoral veins. Contraindications: The Venovo Venous Stent System is contraindicated for use in patients with a known hypersensitivity to nitinol (nickel-titanium), and tantalum, who cannot receive recommended antiplatelet and/or anticoagulation therapy, or who are judged to have a lesion that prevents complete inflation of a balloon dilatation catheter or proper placement of the stent or the stent delivery system Warnings: The Venovo Venous Stent System is supplied sterile and is intended for single use only. Do not resterilize and/or reuse the device. Do not use in patients with total venous occlusion that cannot be dilated to allow passage of the guidewire. Do not use the device with contralateral access. Do not use if pouch is opened or damaged. Do not use the device after the Use By date specified on the label. Persons with allergic reactions to nitinol (nickel-titanium) alloy and/or tantalum may suffer an allergic response to this implant. Do not expose the delivery system to organic solvents, e.g., alcohol. The stent is not designed for repositioning or recapturing. Stenting across a major branch could cause difficulties during future diagnostic or therapeutic procedures. If a long lesion needs to be stented consider using the longest available stent rather than overlapping stents. If multiple stents are placed in an overlapping fashion, they should be of similar composition (i.e., nitinol). The long-term outcomes following repeat dilatation of endothelialized stents are unknown. The safety and effectiveness of this device for use in the arterial system have not been established. Precautions: The device is intended for use by physicians who have received appropriate training. During system flushing, observe that saline exits at the catheter tip. The delivery system is not designed for use with power injection systems. Recrossing a partially or fully deployed stent with adjunct devices must be performed with caution. Prior to stent deployment, remove slack from the delivery system catheter outside the patient. If excessive force is felt during stent deployment, do not force the delivery system. Remove the delivery system and replace with a new unit. Store in a cool, dark, dry place. Do not attempt to break, damage, or disrupt the stent after placement. Potential Complications and Adverse Events: Allergic/anaphylactic reaction; Amputation; Aneurysm; Arteriovenous fistula; Death related/unrelated to procedure; Dissection; Embolization; Extravasation; Fever; Hemorrhage/bleeding requiring a blood transfusion; Hematoma; Hypotension/hypertension; Incorrect positioning of the stent requiring further stenting or surgery; Intimal injury/dissection; Ischemia/infarction of tissue/organ; Local infection; Malposition (failure to deliver the stent to the intended site); Open surgical repair; Pain; Pulmonary embolism; Pseudoaneurysm; Renal failure; Respiratory arrest; Restenosis; Rupture; Septicemia/bacteremia; Stent Fracture; Stent Migration; Vasospasm; Venous occlusion/thrombosis/restenosis Please consult package insert for more detailed safety information and instructions for use. Bard and Venovo are trademarks and/or registered trademarks of C. R. Bard, Inc. All other trademarks are property of their respective owners. Copyright 2016, C. R. Bard, Inc. All Rights Reserved. 19
20 Stephen Black, MD VERNACULAR Trial & Clinical Experience with the VENOVO Venous Stent 20
Complex ilio-caval revascularization in chronic venous obstruction with the Venovo Stent. Michael K. W. Lichtenberg MD, FESC
 Complex ilio-caval revascularization in chronic venous obstruction with the Venovo Stent Michael K. W. Lichtenberg MD, FESC German Venous Center Arnsberg, Germany Not available for sale or distribution
Complex ilio-caval revascularization in chronic venous obstruction with the Venovo Stent Michael K. W. Lichtenberg MD, FESC German Venous Center Arnsberg, Germany Not available for sale or distribution
Future Devices of Venous Interventions
 Future Devices of Venous Interventions Director of Peripheral Vascular Medicine Department of Shin Kong Wu Ho-Su Memorial Hospital, Taiwan Interventional Cardiologist Tien-Yu Wu MD Disclosure Speaker name:...
Future Devices of Venous Interventions Director of Peripheral Vascular Medicine Department of Shin Kong Wu Ho-Su Memorial Hospital, Taiwan Interventional Cardiologist Tien-Yu Wu MD Disclosure Speaker name:...
VIRTUS: Trial Design and Primary Endpoint Results
 VIRTUS: Trial Design and Primary Endpoint Results Mahmood K. Razavi, MD St. Joseph Cardiac and Vascular Center Orange, CA, USA IMPORTANT INFORMATION: These materials are intended to describe common clinical
VIRTUS: Trial Design and Primary Endpoint Results Mahmood K. Razavi, MD St. Joseph Cardiac and Vascular Center Orange, CA, USA IMPORTANT INFORMATION: These materials are intended to describe common clinical
VIVO-EU Results: Prospective European Study of the Zilver Vena TM Venous Stent in the Treatment of Symptomatic Iliofemoral Venous Outflow Obstruction
 VIVO-EU Results: Prospective European Study of the Zilver Vena TM Venous Stent in the Treatment of Symptomatic Iliofemoral Venous Outflow Obstruction Gerard J O Sullivan, M.D. and Jennifer McCann-Brown,
VIVO-EU Results: Prospective European Study of the Zilver Vena TM Venous Stent in the Treatment of Symptomatic Iliofemoral Venous Outflow Obstruction Gerard J O Sullivan, M.D. and Jennifer McCann-Brown,
DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service
 M AY. 6. 2011 10:37 A M F D A - C D R H - O D E - P M O N O. 4147 P. 1 DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service Food and Drug Administration 10903 New Hampshire Avenue Document Control
M AY. 6. 2011 10:37 A M F D A - C D R H - O D E - P M O N O. 4147 P. 1 DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service Food and Drug Administration 10903 New Hampshire Avenue Document Control
VENOVO Venous Stent Update on the Vernacular Trial
 VENOVO Venous Stent Update on the Vernacular Trial Dr. Sunder Narasimhan, MBBS, MS Senior Consultant Vascular and Endovascular Surgeon Apollo Hospital, Bangalore, India VENOVO is a registered trademark
VENOVO Venous Stent Update on the Vernacular Trial Dr. Sunder Narasimhan, MBBS, MS Senior Consultant Vascular and Endovascular Surgeon Apollo Hospital, Bangalore, India VENOVO is a registered trademark
Assurant Cobalt Iliac BALLOON EXPANDABLE STENT SYSTEM
 Assurant Cobalt Iliac BALLOON EXPANDABLE STENT SYSTEM Innovating for life. CONFORMABILIT Y 6 F S H E AT H C O M PAT I B I L I T Y THE ASSURANT COBALT ILIAC STENT, WITH ITS UNIQUE COBALT CHROMIUM MODULAR
Assurant Cobalt Iliac BALLOON EXPANDABLE STENT SYSTEM Innovating for life. CONFORMABILIT Y 6 F S H E AT H C O M PAT I B I L I T Y THE ASSURANT COBALT ILIAC STENT, WITH ITS UNIQUE COBALT CHROMIUM MODULAR
Venous stent experience in Arnsberg Michael K. W. Lichtenberg MD, FESC
 Venous stent experience in Arnsberg Michael K. W. Lichtenberg MD, FESC IMPORTANT INFORMATION: These materials are intended to describe common clinical considerations and procedural steps for the on-label
Venous stent experience in Arnsberg Michael K. W. Lichtenberg MD, FESC IMPORTANT INFORMATION: These materials are intended to describe common clinical considerations and procedural steps for the on-label
Clinical results of venous stents. Michael K. W. Lichtenberg MD, FESC
 Clinical results of venous stents Michael K. W. Lichtenberg MD, FESC Conflict of Interest - Disclosure Within the past 12 months, I or my spouse/partner have had a financial interest/arrangement or affiliation
Clinical results of venous stents Michael K. W. Lichtenberg MD, FESC Conflict of Interest - Disclosure Within the past 12 months, I or my spouse/partner have had a financial interest/arrangement or affiliation
REKANALISATION CHRONISCH VENÖSER VERSCHLÜSSE. Michael K. W. Lichtenberg, FESC
 REKANALISATION CHRONISCH VENÖSER VERSCHLÜSSE Michael K. W. Lichtenberg, FESC Conflict of Interest - Disclosure Within the past 12 months, I or my spouse/partner have had a financial interest/arrangement
REKANALISATION CHRONISCH VENÖSER VERSCHLÜSSE Michael K. W. Lichtenberg, FESC Conflict of Interest - Disclosure Within the past 12 months, I or my spouse/partner have had a financial interest/arrangement
DURABLE. CONSISTENT. SAFE. IN.PACT Admiral Drug-Coated Balloon
 DURABLE. CONSISTENT. SAFE. Drug-Coated Balloon THE SCIENCE BEHIND THE OUTCOMES DCB has proven, long-term durable outcomes across multiple clinical trials, as well as across complex patient and lesion types.
DURABLE. CONSISTENT. SAFE. Drug-Coated Balloon THE SCIENCE BEHIND THE OUTCOMES DCB has proven, long-term durable outcomes across multiple clinical trials, as well as across complex patient and lesion types.
Interventional Treatment VTE: Radiologic Approach
 Interventional Treatment VTE: Radiologic Approach Hae Giu Lee, MD Professor, Dept of Radiology Seoul St. Mary s Hospital The Catholic University of Korea Introduction Incidence High incidence: 250,000-1,000,000/year
Interventional Treatment VTE: Radiologic Approach Hae Giu Lee, MD Professor, Dept of Radiology Seoul St. Mary s Hospital The Catholic University of Korea Introduction Incidence High incidence: 250,000-1,000,000/year
Finally, the Control You Need to Deliver Accurate Treatment
 Finally, the Control You Need to Deliver Accurate Treatment LIFESTREAM Covered Stent. has joined BD When you reach for a balloon expandable stent, you require accuracy. The LIFESTREAM Balloon Expandable
Finally, the Control You Need to Deliver Accurate Treatment LIFESTREAM Covered Stent. has joined BD When you reach for a balloon expandable stent, you require accuracy. The LIFESTREAM Balloon Expandable
THERE IS NO ROLE FOR SURGICAL THERAPY FOR DVT
 THERE IS NO ROLE FOR SURGICAL THERAPY FOR DVT Tara D. Balint, MD FACS Sentara RMH Thursday, June 14, 2018 1 Objectives of treatment for DVT Prevent death from PE Prevent recurrent VTE Prevent post-thrombotic
THERE IS NO ROLE FOR SURGICAL THERAPY FOR DVT Tara D. Balint, MD FACS Sentara RMH Thursday, June 14, 2018 1 Objectives of treatment for DVT Prevent death from PE Prevent recurrent VTE Prevent post-thrombotic
How to best approach chronic venous occlusions?
 How to best approach chronic venous occlusions? Prof. Nils Kucher Director Venous Thromboembolism Reseach Group University Hospital Bern nilskucher.com Disclosure Speaker name: Nils Kucher X X I have the
How to best approach chronic venous occlusions? Prof. Nils Kucher Director Venous Thromboembolism Reseach Group University Hospital Bern nilskucher.com Disclosure Speaker name: Nils Kucher X X I have the
Should We Be More Aggressive in the Treatment of Acute DVT?
 DISCLOSURES Consultant Penumbra, Inc. UCSF Vascular Surgery Symposium April 6, 2017 K. Pallav Kolli, MD Assistant Professor of Clinical Radiology University of California, San Francisco 17 yo male, DVT
DISCLOSURES Consultant Penumbra, Inc. UCSF Vascular Surgery Symposium April 6, 2017 K. Pallav Kolli, MD Assistant Professor of Clinical Radiology University of California, San Francisco 17 yo male, DVT
VIRTUS Trial: Pivotal Cohort 12-Month Primary Safety and Efficacy Results of the VICI Venous Stent System
 VIRTUS Trial: Pivotal Cohort 12-Month Primary Safety and Efficacy Results of the VICI Venous Stent System Mahmood K. Razavi, MD St. Joseph Cardiac and Vascular Center Orange, CA, USA Disclosure Speaker
VIRTUS Trial: Pivotal Cohort 12-Month Primary Safety and Efficacy Results of the VICI Venous Stent System Mahmood K. Razavi, MD St. Joseph Cardiac and Vascular Center Orange, CA, USA Disclosure Speaker
The SplitWire Percutaneous Transluminal Angioplasty Scoring Device. Instructions for Use
 The SplitWire Percutaneous Transluminal Angioplasty Scoring Device Instructions for Use Contents Contains one (1) SplitWire device. Sterile. Sterilized with ethylene oxide gas. Radiopaque. For single use
The SplitWire Percutaneous Transluminal Angioplasty Scoring Device Instructions for Use Contents Contains one (1) SplitWire device. Sterile. Sterilized with ethylene oxide gas. Radiopaque. For single use
The Ideal Venous Stent and Early Results from Venous Stent Trials PNEC, Seattle
 The Ideal Venous Stent and Early Results from Venous Stent Trials 2017 PNEC, Seattle Bill Marston MD Professor, Div of Vascular Surgery University of N. Carolina DISCLOSURES William Marston, MD Consultant/Advisory
The Ideal Venous Stent and Early Results from Venous Stent Trials 2017 PNEC, Seattle Bill Marston MD Professor, Div of Vascular Surgery University of N. Carolina DISCLOSURES William Marston, MD Consultant/Advisory
Venous interventions in DVT
 Venous interventions in DVT Sriram Narayanan Chief of Vascular and Endovascular Surgery, Tan Tock Seng Hospital A/Prof of Surgery, National University of Singapore ANTI-COAGULATION LMWH Warfarin x 6m Acute
Venous interventions in DVT Sriram Narayanan Chief of Vascular and Endovascular Surgery, Tan Tock Seng Hospital A/Prof of Surgery, National University of Singapore ANTI-COAGULATION LMWH Warfarin x 6m Acute
PERFORMANCE YOU CAN TRUST. EverFlex Self-expanding Peripheral Stent with Entrust Delivery System
 PERFORMANCE YOU CAN TRUST EverFlex Self-expanding Peripheral Stent with Entrust Delivery System The Entrust Delivery System is designed to provide improved patient outcomes and procedural efficiency. The
PERFORMANCE YOU CAN TRUST EverFlex Self-expanding Peripheral Stent with Entrust Delivery System The Entrust Delivery System is designed to provide improved patient outcomes and procedural efficiency. The
Intervention for Deep Venous Thrombosis and Pulmonary Embolus
 Intervention for Deep Venous Thrombosis and Pulmonary Embolus Michael R. Jaff, DO Paul and Phyllis Fireman Endowed Chair in Vascular Medicine Massachusetts General Hospital Professor of Medicine Harvard
Intervention for Deep Venous Thrombosis and Pulmonary Embolus Michael R. Jaff, DO Paul and Phyllis Fireman Endowed Chair in Vascular Medicine Massachusetts General Hospital Professor of Medicine Harvard
Instructions for Use Cordis PRECISE PRO Rx Nitinol Stent System
 100000017144.3 100000017144.3 Instructions for Use Cordis PRECISE PRO Rx Nitinol Stent System Figure 1.065" (1.65 mm) OR.078" (1.98 mm) 16 4 13 10 11 15 8 3 14.062" (1.57 mm) OR.065" (1.65 mm).038" (0.97
100000017144.3 100000017144.3 Instructions for Use Cordis PRECISE PRO Rx Nitinol Stent System Figure 1.065" (1.65 mm) OR.078" (1.98 mm) 16 4 13 10 11 15 8 3 14.062" (1.57 mm) OR.065" (1.65 mm).038" (0.97
1 Comparison of Warfarin and Aspirin for Symptomatic Intracranial Arterial Stenosis. N Engl J Med 352;13, March 31, 2005
 The risk of ischemic stroke in patients with Intracranial Atherosclerotic Disease (ICAD) ranges from 7 to 24%. 1,2 Developed specifically for the treatment of ICAD, the Wingspan Stent System and Gateway
The risk of ischemic stroke in patients with Intracranial Atherosclerotic Disease (ICAD) ranges from 7 to 24%. 1,2 Developed specifically for the treatment of ICAD, the Wingspan Stent System and Gateway
Sustained Release. Superior Results.
 ELUVIA Drug-Eluting Vascular Stent System Sustained Release. Superior Results. Superiority determined in Post Hoc Superiority Analysis. 12-Month Primary Patency rate of 86.8% in the Eluvia arm (n=309)
ELUVIA Drug-Eluting Vascular Stent System Sustained Release. Superior Results. Superiority determined in Post Hoc Superiority Analysis. 12-Month Primary Patency rate of 86.8% in the Eluvia arm (n=309)
Techniques for thrombus removal in acute DVT Benefits of an Endovascular Approach for Rapid Flow Restoration in DVT
 Techniques for thrombus removal in acute DVT Benefits of an Endovascular Approach for Rapid Flow Restoration in DVT Michael K. W. Lichtenberg, MD, FESC Vascular Centre Arnsberg, Germany Disclosure Speaker
Techniques for thrombus removal in acute DVT Benefits of an Endovascular Approach for Rapid Flow Restoration in DVT Michael K. W. Lichtenberg, MD, FESC Vascular Centre Arnsberg, Germany Disclosure Speaker
Update on Tack Optimized Balloon Angioplasty (TOBA) Below the Knee. Marianne Brodmann, MD Medical University Graz Graz, Austria
 Update on Tack Optimized Balloon Angioplasty (TOBA) Below the Knee Marianne Brodmann, MD Medical University Graz Graz, Austria Critical Limb Ischemia Infrapopliteal arterial disease is a leading source
Update on Tack Optimized Balloon Angioplasty (TOBA) Below the Knee Marianne Brodmann, MD Medical University Graz Graz, Austria Critical Limb Ischemia Infrapopliteal arterial disease is a leading source
A Dedicated Venous Self-expanding Oblique Hybrid Nitinol Stent (Sinus-Obliquus Stent)
 A Dedicated Venous Self-expanding Oblique Hybrid Nitinol Stent (Sinus-Obliquus Stent) Anna Stuck, Rolf P. Engelberger, Nils Kucher Division of Angiology Cantonal Hospital Fribourg & Bern University Hospital
A Dedicated Venous Self-expanding Oblique Hybrid Nitinol Stent (Sinus-Obliquus Stent) Anna Stuck, Rolf P. Engelberger, Nils Kucher Division of Angiology Cantonal Hospital Fribourg & Bern University Hospital
Patency rates and clinical results of the Veniti VICI Stent for treatment of iliac vein lesion Data from the Arnsberg Venous Registry
 Patency rates and clinical results of the Veniti VICI Stent for treatment of iliac vein lesion Data from the Arnsberg Venous Registry Michael K. W. Lichtenberg MD, FESC Conflict of Interest - Disclosure
Patency rates and clinical results of the Veniti VICI Stent for treatment of iliac vein lesion Data from the Arnsberg Venous Registry Michael K. W. Lichtenberg MD, FESC Conflict of Interest - Disclosure
What Really Matters to Patient is QOL: Veniti Virtus Venous Feasibility Trial
 DISCLOSURES Speaker name: Lowell S. Kabnick, MD, FACS... I have the following potential conflicts of interest: Consultant and shareholder, VENITI, Inc. Consultant to BARD What Really Matters to Patient
DISCLOSURES Speaker name: Lowell S. Kabnick, MD, FACS... I have the following potential conflicts of interest: Consultant and shareholder, VENITI, Inc. Consultant to BARD What Really Matters to Patient
MICHAEL R. JAFF, DO MASSACHUSETTS, UNITED STATES. Medtronic Further. Together
 DRUG-COATED BALL0ON TREATMENT FOR PATIENTS WITH INTERMITTENT CLAUDICATION: INSIGHTS FROM THE IN.PACT GLOBAL FULL CLINICAL COHORT MICHAEL R. JAFF, DO MASSACHUSETTS, UNITED STATES Medtronic Further. Together
DRUG-COATED BALL0ON TREATMENT FOR PATIENTS WITH INTERMITTENT CLAUDICATION: INSIGHTS FROM THE IN.PACT GLOBAL FULL CLINICAL COHORT MICHAEL R. JAFF, DO MASSACHUSETTS, UNITED STATES Medtronic Further. Together
Proven Performance Through Innovative Design *
 Proven Performance Through Innovative Design * Deliver Our Next Generation AV Covered Stent Results The COVERA Vascular Covered Stent builds upon proven technologies from the category leader in AV Access.
Proven Performance Through Innovative Design * Deliver Our Next Generation AV Covered Stent Results The COVERA Vascular Covered Stent builds upon proven technologies from the category leader in AV Access.
Acoustic Pulse Thrombolysis Treatment
 Acoustic Pulse Thrombolysis Treatment BTGVascular.com SETTING THE STANDARD FOR VASCULAR THERAPIES Quickly & safely dissolve thrombus with the EKOS System. The Acoustic Pulse Difference Acoustic Pulse Thrombolysis
Acoustic Pulse Thrombolysis Treatment BTGVascular.com SETTING THE STANDARD FOR VASCULAR THERAPIES Quickly & safely dissolve thrombus with the EKOS System. The Acoustic Pulse Difference Acoustic Pulse Thrombolysis
Starting with deep venous treatment
 Starting with deep venous treatment Carsten Arnoldussen, MD Interventional Radiologist Maastricht University Medical Centre, Maastricht VieCuri Medical Centre, Venlo The Netherlands Background Maastricht
Starting with deep venous treatment Carsten Arnoldussen, MD Interventional Radiologist Maastricht University Medical Centre, Maastricht VieCuri Medical Centre, Venlo The Netherlands Background Maastricht
BC Vascular Day. Contents. November 3, Abdominal Aortic Aneurysm 2 3. Peripheral Arterial Disease 4 6. Deep Venous Thrombosis 7 8
 BC Vascular Day Contents Abdominal Aortic Aneurysm 2 3 November 3, 2018 Peripheral Arterial Disease 4 6 Deep Venous Thrombosis 7 8 Abdominal Aortic Aneurysm Conservative Management Risk factor modification
BC Vascular Day Contents Abdominal Aortic Aneurysm 2 3 November 3, 2018 Peripheral Arterial Disease 4 6 Deep Venous Thrombosis 7 8 Abdominal Aortic Aneurysm Conservative Management Risk factor modification
Iliofemoral DVT: Miminizing Post-Thrombotic Syndrome
 Iliofemoral DVT: Miminizing Post-Thrombotic Syndrome Catherine K. Chang, MD FACS Vascular Surgery San Diego Southern California Permanente Medical Group Acute Deep Venous Thrombosis Incidence & Outcomes
Iliofemoral DVT: Miminizing Post-Thrombotic Syndrome Catherine K. Chang, MD FACS Vascular Surgery San Diego Southern California Permanente Medical Group Acute Deep Venous Thrombosis Incidence & Outcomes
COVERA Vascular Covered Stents in the Management of Dysfunctional AV Access
 COVERA Vascular Covered Stents in the Management of Dysfunctional AV Access Bart L. Dolmatch, M.D., FSIR Palo Alto Medical Foundation Mountain View, CA USA This presentation is being made on behalf of
COVERA Vascular Covered Stents in the Management of Dysfunctional AV Access Bart L. Dolmatch, M.D., FSIR Palo Alto Medical Foundation Mountain View, CA USA This presentation is being made on behalf of
Management of Acute DVT Extending From the Tibial Veins to the Common Iliac Vein Using the AngioJet Thrombectomy System
 FEATURED TECHNOLOGY: ANGIOJET THROMBECTOMY SYSTEM Sponsored by Boston Scientific Corporation Management of Acute DVT Extending From the Tibial Veins to the Common Iliac Vein Using the AngioJet Thrombectomy
FEATURED TECHNOLOGY: ANGIOJET THROMBECTOMY SYSTEM Sponsored by Boston Scientific Corporation Management of Acute DVT Extending From the Tibial Veins to the Common Iliac Vein Using the AngioJet Thrombectomy
B05680 Vers. 6/ Figure 2. LifeStent Vascular Stent
 B05680 Vers. 6/12-10 BARD LIFESTENT Stent and Delivery System Vascular Application Recommended Guidewire Length Table Catheter Working Length Recommended Guidewire Length 130 cm 300 cm 80 cm 260 cm 160
B05680 Vers. 6/12-10 BARD LIFESTENT Stent and Delivery System Vascular Application Recommended Guidewire Length Table Catheter Working Length Recommended Guidewire Length 130 cm 300 cm 80 cm 260 cm 160
Case Study of Implantation of a VICI VENOUS STENT - Combined NIVL and PTS Stenting
 Case Study of Implantation of a VICI VENOUS STENT - Combined NIVL and PTS Stenting Courtesy of Mr. Stephen Black United kingdom Patient History 25 y/o female 2011: Conservatively treated ilio-femoral DVT
Case Study of Implantation of a VICI VENOUS STENT - Combined NIVL and PTS Stenting Courtesy of Mr. Stephen Black United kingdom Patient History 25 y/o female 2011: Conservatively treated ilio-femoral DVT
The evidence for venous interventions is evolving- many patients do actually benefit. Nils Kucher University Hospital Bern Switzerland
 The evidence for venous interventions is evolving- many patients do actually benefit Nils Kucher University Hospital Bern Switzerland Disclosure Speaker name: Nils Kucher X X I have the following potential
The evidence for venous interventions is evolving- many patients do actually benefit Nils Kucher University Hospital Bern Switzerland Disclosure Speaker name: Nils Kucher X X I have the following potential
INFERIOR VENA CAVA FILTERS QUIZ 10 QUESTIONS MAY 5, 2014
 INFERIOR VENA CAVA FILTERS QUIZ 10 QUESTIONS MAY 5, 2014 QUESTION 1 1. Anatomically Transposition of the IVC is observed in what % of individuals A) 1-5% B).2-.05% C) 3-5% D) 5-10% ANSWER QUESTION 1 Answer
INFERIOR VENA CAVA FILTERS QUIZ 10 QUESTIONS MAY 5, 2014 QUESTION 1 1. Anatomically Transposition of the IVC is observed in what % of individuals A) 1-5% B).2-.05% C) 3-5% D) 5-10% ANSWER QUESTION 1 Answer
Anticoagulation therapy following endovascular treatment of iliofemoral deep vein thrombosis
 Anticoagulation therapy following endovascular treatment of iliofemoral deep vein thrombosis Tim Sebastian, M.D. University Hospital Zurich Clinic for Angiology Disclosure Speaker name: Tim Sebastian I
Anticoagulation therapy following endovascular treatment of iliofemoral deep vein thrombosis Tim Sebastian, M.D. University Hospital Zurich Clinic for Angiology Disclosure Speaker name: Tim Sebastian I
TOBA II 12-Month Results Tack Optimized Balloon Angioplasty
 TOBA II 12-Month Results Tack Optimized Balloon Angioplasty William Gray, MD System Chief, Cardiovascular Division Main Line Health, Philadelphia, PA Dissection: The Primary Mechanism of Angioplasty Lesions
TOBA II 12-Month Results Tack Optimized Balloon Angioplasty William Gray, MD System Chief, Cardiovascular Division Main Line Health, Philadelphia, PA Dissection: The Primary Mechanism of Angioplasty Lesions
Ultrasound-assisted catheter-directed thrombolysis: Does it really work? The BERNUTIFUL trial
 Ultrasound-assisted catheter-directed thrombolysis: Does it really work? The BERNUTIFUL trial Rolf P. Engelberger Division of Angiology CHUV, Lausanne & Inselspital, Bern Switzerland Disclosure Speaker
Ultrasound-assisted catheter-directed thrombolysis: Does it really work? The BERNUTIFUL trial Rolf P. Engelberger Division of Angiology CHUV, Lausanne & Inselspital, Bern Switzerland Disclosure Speaker
NOTE: Deep Vein Thrombosis (DVT) Risk Factors
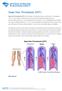 Deep Vein Thrombosis (DVT) Deep Vein Thrombosis (DVT) is the formation of a blood clot, known as a thrombus, in the deep leg vein. It is a very serious condition that can cause permanent damage to the
Deep Vein Thrombosis (DVT) Deep Vein Thrombosis (DVT) is the formation of a blood clot, known as a thrombus, in the deep leg vein. It is a very serious condition that can cause permanent damage to the
Not all Leg DVT s are the Same: Which Patients Benefit from Interventional Therapy? Case 1:
 12/16/2015 Not all Leg DVT s are the Same: Which Patients Benefit from Interventional Therapy? Constantino S.Peña, FSIR, FSCCT, FAHA Interventional Radiologist Medical Director, Vascular Imaging Miami
12/16/2015 Not all Leg DVT s are the Same: Which Patients Benefit from Interventional Therapy? Constantino S.Peña, FSIR, FSCCT, FAHA Interventional Radiologist Medical Director, Vascular Imaging Miami
Aspirex for Upper and Lower Extremity DVT
 Aspirex for Upper and Lower Extremity DVT Steven Kum MD Vascular & Endovascular Surgeon Director of Vascular Service Changi General Hospital Singapore Disclosure Speaker name:... I have the following potential
Aspirex for Upper and Lower Extremity DVT Steven Kum MD Vascular & Endovascular Surgeon Director of Vascular Service Changi General Hospital Singapore Disclosure Speaker name:... I have the following potential
Straub Endovascular System &
 Straub Endovascular System & S t r a u b E n d o v a s c u l a r To o l s Straub Endovascular System Effective debulking in occluded arteries and veins Effective debulking in many indications Rotarex
Straub Endovascular System & S t r a u b E n d o v a s c u l a r To o l s Straub Endovascular System Effective debulking in occluded arteries and veins Effective debulking in many indications Rotarex
EKOS. Interventional Vascular 3 February, Imagine where we can go.
 EKOS Interventional Vascular 3 February, 2015 Imagine where we can go. Forward-looking statement This presentation and information communicated verbally to you may contain certain projections and other
EKOS Interventional Vascular 3 February, 2015 Imagine where we can go. Forward-looking statement This presentation and information communicated verbally to you may contain certain projections and other
Implications from the ACCP 2012 Consensus Guidelines for the Management of Thrombosis: a case based approach
 Implications from the ACCP 2012 Consensus Guidelines for the Management of Thrombosis: a case based approach Prof. I. Baumgartner Head Clinical and Interventional Angiology About the ACCP guidelines Widely
Implications from the ACCP 2012 Consensus Guidelines for the Management of Thrombosis: a case based approach Prof. I. Baumgartner Head Clinical and Interventional Angiology About the ACCP guidelines Widely
The Evidence Base for Treating Acute DVT
 The Evidence Base for Treating Acute DVT Mr Chung Sim Lim Consultant Vascular Surgeon and Honorary Lecturer Royal Free London NHS Foundation Trust and University College London NIHR UCLH Biomedical Research
The Evidence Base for Treating Acute DVT Mr Chung Sim Lim Consultant Vascular Surgeon and Honorary Lecturer Royal Free London NHS Foundation Trust and University College London NIHR UCLH Biomedical Research
Christian Wissgott MD, PhD Assistant Director, Radiology Westküstenkliniken Heide
 2-Year Results Of The Tack Optimized Balloon Angioplasty (TOBA) Trial For Fem- Pop Lesions Demonstrates Safety and Efficacy Of The Tack Endovascular System In Repairing Focal Post-PTA Dissections Christian
2-Year Results Of The Tack Optimized Balloon Angioplasty (TOBA) Trial For Fem- Pop Lesions Demonstrates Safety and Efficacy Of The Tack Endovascular System In Repairing Focal Post-PTA Dissections Christian
Talent Abdominal Stent Graft
 Talent Abdominal with THE Xcelerant Hydro Delivery System Expanding the Indications for EVAR Treat More Patients Short Necks The Talent Abdominal is the only FDA-approved device for proximal aortic neck
Talent Abdominal with THE Xcelerant Hydro Delivery System Expanding the Indications for EVAR Treat More Patients Short Necks The Talent Abdominal is the only FDA-approved device for proximal aortic neck
Instructions for Use
 Instructions for Use B05692 Vers.4/09-11 LifeStent Solo Vascular Stent System Figure 1 CAUTION: U.S. federal law restricts this device to sale by or on the order of a physician This device is supplied
Instructions for Use B05692 Vers.4/09-11 LifeStent Solo Vascular Stent System Figure 1 CAUTION: U.S. federal law restricts this device to sale by or on the order of a physician This device is supplied
CP STENT. Large Diameter, Balloon Expandable Stent
 CP STENT Large, Expandable CP STENT OPTIONS 12mm to Expansion 26mm to Expansion CP Matrix (number of zigs) 1.6 2.2 2.8 3.4 3.9 4.5 5 5.5 6 12 14 15 16 18 20 22 24 26 28 30 CP Details: CP is composed of
CP STENT Large, Expandable CP STENT OPTIONS 12mm to Expansion 26mm to Expansion CP Matrix (number of zigs) 1.6 2.2 2.8 3.4 3.9 4.5 5 5.5 6 12 14 15 16 18 20 22 24 26 28 30 CP Details: CP is composed of
System.
 System www.penumbrainc.com POD System Case Examples 45 cm of POD Packing Coil in 1.5 to 3 mm diameter dilating vessel Bronchial Artery Embolization Dr. Amit Kakkar and Dr. Aksim Rivera, Bronx, NY Inflow:
System www.penumbrainc.com POD System Case Examples 45 cm of POD Packing Coil in 1.5 to 3 mm diameter dilating vessel Bronchial Artery Embolization Dr. Amit Kakkar and Dr. Aksim Rivera, Bronx, NY Inflow:
COVERA Vascular Covered Stents Innovation in AV Access
 IL0118 Rev.0 1 COVERA Vascular Covered Stents Innovation in AV Access Dheeraj Rajan, MD, FRCPC, FSIR Head and Associate Professor, Division of Vascular & Interventional Radiology Department of Medical
IL0118 Rev.0 1 COVERA Vascular Covered Stents Innovation in AV Access Dheeraj Rajan, MD, FRCPC, FSIR Head and Associate Professor, Division of Vascular & Interventional Radiology Department of Medical
Patient Brochure. Clearstream Technologies, Ltd. Moyne Upper Enniscorthy Co. Wexford, Ireland. PK Rev. 0 05/17
 Patient Brochure Clearstream Technologies, Ltd. Moyne Upper Enniscorthy Co. Wexford, Ireland PK1411100 Rev. 0 05/17 LIFESTREAM Patient Brochure If you or a member of your family has been diagnosed with
Patient Brochure Clearstream Technologies, Ltd. Moyne Upper Enniscorthy Co. Wexford, Ireland PK1411100 Rev. 0 05/17 LIFESTREAM Patient Brochure If you or a member of your family has been diagnosed with
Aggressive endovascular management of ilio-femoral DVT. thrombotic syndrome. is the key in preventing post
 CACVS 2017 Aggressive endovascular management of ilio-femoral DVT is the key in preventing post thrombotic syndrome ALI AMIN MD, FACS,FACC, RVT CHIEF OF ENDOVASCULAR INTERVENTIONS READING HEALTH SYSTEM
CACVS 2017 Aggressive endovascular management of ilio-femoral DVT is the key in preventing post thrombotic syndrome ALI AMIN MD, FACS,FACC, RVT CHIEF OF ENDOVASCULAR INTERVENTIONS READING HEALTH SYSTEM
DISRUPT PAD. (( Data Summary )) DISRUPT PAD Data Summary SPL Rev. B 2016 Shockwave Medical Inc. All rights reserved.
 DISRUPT PAD (( Data Summary )) DISRUPT PAD Data Summary SPL 60971 Rev. B 1 Summary of the key findings from the DISRUPT PAD Study 99% of femoropopliteal lesions treated were moderately or severely calcified.
DISRUPT PAD (( Data Summary )) DISRUPT PAD Data Summary SPL 60971 Rev. B 1 Summary of the key findings from the DISRUPT PAD Study 99% of femoropopliteal lesions treated were moderately or severely calcified.
Directions For Use. All directions should be read before use
 Directions For Use All directions should be read before use DEVICE DESCRIPTION: The CLEANER.XT Rotational Thrombectomy System is a percutaneous, 6Fr catheter based system (single piece construction) that
Directions For Use All directions should be read before use DEVICE DESCRIPTION: The CLEANER.XT Rotational Thrombectomy System is a percutaneous, 6Fr catheter based system (single piece construction) that
Chronic Iliocaval Venous Occlusive Disease
 none Chronic Iliocaval Venous Occlusive Disease David Rigberg, M.D. Clinical Professor of Surgery Division of Vascular Surgery University of California Los Angeles Chronic Venous Occlusive Disease Chronic
none Chronic Iliocaval Venous Occlusive Disease David Rigberg, M.D. Clinical Professor of Surgery Division of Vascular Surgery University of California Los Angeles Chronic Venous Occlusive Disease Chronic
Optimal Utilization of Thrombolytics
 April 8-9, 2011 New York LaGuardia Marriott COMPLETE MANAGEMENT OF VENOUS DISEASE Optimal Utilization of Thrombolytics Anthony J. Comerota, MD, FACS, FACC Director, Jobst Vascular Institute Adjunct Professor
April 8-9, 2011 New York LaGuardia Marriott COMPLETE MANAGEMENT OF VENOUS DISEASE Optimal Utilization of Thrombolytics Anthony J. Comerota, MD, FACS, FACC Director, Jobst Vascular Institute Adjunct Professor
Disclosures. DVT: Diagnosis and Treatment. Questions To Ask. Dr. Susanna Shin - DVT: Diagnosis and Treatment. Acute Venous Thromboembolism (VTE) None
 Disclosures DVT: Diagnosis and Treatment None Susanna Shin, MD, FACS Assistant Professor University of Washington Acute Venous Thromboembolism (VTE) Deep Venous Thrombosis (DVT) Pulmonary Embolism (PE)
Disclosures DVT: Diagnosis and Treatment None Susanna Shin, MD, FACS Assistant Professor University of Washington Acute Venous Thromboembolism (VTE) Deep Venous Thrombosis (DVT) Pulmonary Embolism (PE)
Pharmaco-mechanical techniques stand alone procedures? Peter Neglén, MD, PhD SP Vascular Center Limassol Cyprus
 Pharmaco-mechanical techniques stand alone procedures? Peter Neglén, MD, PhD SP Vascular Center Limassol Cyprus Faculty Disclosure Peter Neglén, M.D., Ph.D Stockholder/Founder of Veniti, Inc. Member, Medical
Pharmaco-mechanical techniques stand alone procedures? Peter Neglén, MD, PhD SP Vascular Center Limassol Cyprus Faculty Disclosure Peter Neglén, M.D., Ph.D Stockholder/Founder of Veniti, Inc. Member, Medical
Endovascular treatment of acquired arteriovenous fistula with severe hemodynamic effects: a case report
 Endovascular treatment of acquired arteriovenous fistula with severe hemodynamic effects: a case report The Leipzig Interventional Course, January 24 27, 2017 El Samman K., Šedivý P., Šnajdrová A., Přindišová
Endovascular treatment of acquired arteriovenous fistula with severe hemodynamic effects: a case report The Leipzig Interventional Course, January 24 27, 2017 El Samman K., Šedivý P., Šnajdrová A., Přindišová
Percutaneously Inserted AngioVac Suction Thrombectomy for the Treatment of Filter-Related. Iliocaval Thrombosis
 Percutaneously Inserted AngioVac Suction Thrombectomy for the Treatment of Filter-Related Iliocaval Thrombosis Faiz D. Francis, DO; Gianvito Salerno, MD; Sabbah D. Butty, MD Abstract In the setting of
Percutaneously Inserted AngioVac Suction Thrombectomy for the Treatment of Filter-Related Iliocaval Thrombosis Faiz D. Francis, DO; Gianvito Salerno, MD; Sabbah D. Butty, MD Abstract In the setting of
Acute Venous Thrombosis: Thrombus Removal with Adjunctive Catheter-Directed Thrombolysis (ATTRACT Trial)
 Acute Venous Thrombosis: Thrombus Removal with Adjunctive Catheter-Directed Thrombolysis (ATTRACT Trial) N Engl J Med. Volume 377(23):2240-2252. December 7, 2017 Wednesday, July 11, 2018, 1:00pm ET Guest
Acute Venous Thrombosis: Thrombus Removal with Adjunctive Catheter-Directed Thrombolysis (ATTRACT Trial) N Engl J Med. Volume 377(23):2240-2252. December 7, 2017 Wednesday, July 11, 2018, 1:00pm ET Guest
Successful recanalisation of venous thrombotic occlusions with Aspirex mechanical thrombectomy. Michael K. W. Lichtenberg
 Successful recanalisation of venous thrombotic occlusions with Aspirex mechanical thrombectomy Michael K. W. Lichtenberg Disclosure Speaker name: Michael Lichtenberg... I have the following potential conflicts
Successful recanalisation of venous thrombotic occlusions with Aspirex mechanical thrombectomy Michael K. W. Lichtenberg Disclosure Speaker name: Michael Lichtenberg... I have the following potential conflicts
Introduction 3. What is Peripheral Vascular Disease? 5. What Are Some of the Symptoms of Peripheral Vascular Disease? 6
 Patient Information Table of Contents Introduction 3 What is Peripheral Vascular Disease? 5 What Are Some of the Symptoms of Peripheral Vascular Disease? 6 What Causes Peripheral Vascular Disease? 7 How
Patient Information Table of Contents Introduction 3 What is Peripheral Vascular Disease? 5 What Are Some of the Symptoms of Peripheral Vascular Disease? 6 What Causes Peripheral Vascular Disease? 7 How
Varicose Veins are a Symptom of Vein Disease. Now you can treat the source of your varicose veins with non-surgical endovenous laser treatment.
 Varicose Veins are a Symptom of Vein Disease. Now you can treat the source of your varicose veins with non-surgical endovenous laser treatment. Approximately 1 in 5 adult Americans suffer from superficial
Varicose Veins are a Symptom of Vein Disease. Now you can treat the source of your varicose veins with non-surgical endovenous laser treatment. Approximately 1 in 5 adult Americans suffer from superficial
AXS Catalyst Distal Access Catheter
 AXS Catalyst Distal Access Catheter Directions for Use 2 AXS Catalyst Distal Access Catheter ONLY Caution: Federal Law (USA) restricts this device to sale by or on the order of a physician. warning Contents
AXS Catalyst Distal Access Catheter Directions for Use 2 AXS Catalyst Distal Access Catheter ONLY Caution: Federal Law (USA) restricts this device to sale by or on the order of a physician. warning Contents
1 Description. 2 Indications. 3 Warnings ASPIRATION CATHETER
 Page 1 of 5 ASPIRATION CATHETER Carefully read all instructions prior to use, observe all warnings and precautions noted throughout these instructions. Failure to do so may result in complications. STERILE.
Page 1 of 5 ASPIRATION CATHETER Carefully read all instructions prior to use, observe all warnings and precautions noted throughout these instructions. Failure to do so may result in complications. STERILE.
REBEL. Platinum Chromium Coronary Stent System. Patient Information Guide
 REBEL Patient Information Guide REBEL PATIENT INFORMATION GUIDE You have recently had a REBEL bare metal stent implanted in the coronary arteries of your heart. The following information is important for
REBEL Patient Information Guide REBEL PATIENT INFORMATION GUIDE You have recently had a REBEL bare metal stent implanted in the coronary arteries of your heart. The following information is important for
Copy Here. The Easy One.. What is the Role of Thrombus Removal in Acute Proximal DVT after ATTRACT? Deep Venous Thrombosis Spectrum
 What is the Role of Thrombus Removal in Acute Proximal DVT after ATTRACT? Mitchell J. Silver DO FACC FSVM RPVI Director, Center for Critical Limb Care Riverside Methodist Hospital Ohio Health Heart and
What is the Role of Thrombus Removal in Acute Proximal DVT after ATTRACT? Mitchell J. Silver DO FACC FSVM RPVI Director, Center for Critical Limb Care Riverside Methodist Hospital Ohio Health Heart and
Use of EKOS Catheter in the management of Venous Mr. Manoj Niverthi, Mr. Sarang Pujari, and Ms. Nupur Dandavate, The GTF Group
 Use of EKOS Catheter in the management of Venous Thromboembolism @ Mr. Manoj Niverthi, Mr. Sarang Pujari, and Ms. Nupur Dandavate, The GTF Group Introduction Georgia Thrombosis Forum (GTF, www.gtfonline.net)
Use of EKOS Catheter in the management of Venous Thromboembolism @ Mr. Manoj Niverthi, Mr. Sarang Pujari, and Ms. Nupur Dandavate, The GTF Group Introduction Georgia Thrombosis Forum (GTF, www.gtfonline.net)
Venous Thrombosis. Magnitude of the Problem. DVT 2 Million PE 600,000. Death 60,000. Estimated Cost of VTE Care $1.5 Billion/year.
 Venous Thrombosis Magnitude of the Problem DVT 2 Million Postthrombotic Syndrome 800,000 PE 600,000 Death 60,000 Silent PE 1 Million Pulmonary Hypertension 30,000 Estimated Cost of VTE Care $1.5 Billion/year
Venous Thrombosis Magnitude of the Problem DVT 2 Million Postthrombotic Syndrome 800,000 PE 600,000 Death 60,000 Silent PE 1 Million Pulmonary Hypertension 30,000 Estimated Cost of VTE Care $1.5 Billion/year
Proper Diagnosis of Venous Thromboembolism (VTE)
 Proper Diagnosis of Venous Thromboembolism (VTE) Whal Lee, M.D. Seoul National University Hospital Department of Radiology 2 nd EFORT Asia Symposium, 3 rd November 2010, Taipei DVT - Risk Factors Previous
Proper Diagnosis of Venous Thromboembolism (VTE) Whal Lee, M.D. Seoul National University Hospital Department of Radiology 2 nd EFORT Asia Symposium, 3 rd November 2010, Taipei DVT - Risk Factors Previous
- Our patients with iliofemoral DVT - Effective thrombus removal with purely mechanical thrombectomy can lead to better outcomes
 - Our patients with iliofemoral DVT - Effective thrombus removal with purely mechanical thrombectomy can lead to better outcomes Michael K. W. Lichtenberg, FESC Conflict of Interest - Disclosure Within
- Our patients with iliofemoral DVT - Effective thrombus removal with purely mechanical thrombectomy can lead to better outcomes Michael K. W. Lichtenberg, FESC Conflict of Interest - Disclosure Within
PTA Balloon Dilatation Catheter
 PTA Balloon Dilatation Catheter March 2016 1/6(3214-0) Catalogue number Serial number Lot number Use by Do not reuse Do not resterilize Do not use if package is damaged Consult instructions for use Keep
PTA Balloon Dilatation Catheter March 2016 1/6(3214-0) Catalogue number Serial number Lot number Use by Do not reuse Do not resterilize Do not use if package is damaged Consult instructions for use Keep
Percutaneous Mechanical Thrombectomy for Acute Iliofemoral DVT with the Aspirex Catheter: The Dijon Experience
 JFICV 2018, Beaune Percutaneous Mechanical Thrombectomy for Acute Iliofemoral DVT with the Aspirex Catheter: The Dijon Experience Prof. Romaric LOFFROY, MD, PhD, FCIRSE Chief, Department of Vascular and
JFICV 2018, Beaune Percutaneous Mechanical Thrombectomy for Acute Iliofemoral DVT with the Aspirex Catheter: The Dijon Experience Prof. Romaric LOFFROY, MD, PhD, FCIRSE Chief, Department of Vascular and
The Role of LUTONIX 035 DCB in AV Fistula Dysfunction Management in our Practice
 The Role of LUTONIX 035 DCB in AV Fistula Dysfunction Management in our Practice Dr Kate Steiner Consultant Interventional Radiologist East and North Hertfordshire NHS Trust Disclosure Speaker name: Dr
The Role of LUTONIX 035 DCB in AV Fistula Dysfunction Management in our Practice Dr Kate Steiner Consultant Interventional Radiologist East and North Hertfordshire NHS Trust Disclosure Speaker name: Dr
FROM THE EVERYDAY TO THE EXTRAORDINARY
 FROM THE EVERYDAY TO THE EXTRAORDINARY Created with the collaboration of more than 250 physicians around the world, ENDURANT empowers you to create stronger outcomes for more patients, including those
FROM THE EVERYDAY TO THE EXTRAORDINARY Created with the collaboration of more than 250 physicians around the world, ENDURANT empowers you to create stronger outcomes for more patients, including those
Surgical approach for DVT. Division of Vascular Surgery Department of Surgery Seoul National University College of Medicine
 Surgical approach for DVT Seung-Kee Min Division of Vascular Surgery Department of Surgery Seoul National University College of Medicine Treatment Options for Venous Thrombosis Unfractionated heparin &
Surgical approach for DVT Seung-Kee Min Division of Vascular Surgery Department of Surgery Seoul National University College of Medicine Treatment Options for Venous Thrombosis Unfractionated heparin &
Treatment of Chronic DVT with EKOS: Reproducing ACCESS PTS Data in Every Day Clinical Practice
 Treatment of Chronic DVT with EKOS: Reproducing ACCESS PTS Data in Every Day Clinical Practice Mert Dumantepe, MD Acibadem Altunizade Hospital, Istanbul, Turkey Department of Cardiovascular Surgery Disclosure
Treatment of Chronic DVT with EKOS: Reproducing ACCESS PTS Data in Every Day Clinical Practice Mert Dumantepe, MD Acibadem Altunizade Hospital, Istanbul, Turkey Department of Cardiovascular Surgery Disclosure
LUTONIX DCB in BTK Update on the BTK clinical program & single center experience
 LUTONIX DCB in BTK Update on the BTK clinical program & single center experience Prof. Dr. med. Dierk Scheinert Department of Interventional Angiology University Hospital Leipzig Disclaimer 1. The information
LUTONIX DCB in BTK Update on the BTK clinical program & single center experience Prof. Dr. med. Dierk Scheinert Department of Interventional Angiology University Hospital Leipzig Disclaimer 1. The information
Watchman. Left Atrial Appendage Closure Device. Uniquely engineered for the LAA 1-3 with proven safety and longterm efficacy. 4-8
 TM Watchman Left Atrial Appendage Closure Device PROOF OFLEADERSHIP Uniquely engineered for the LAA 1-3 with proven safety and longterm efficacy. 4-8 Patients with AF have a 5x increased risk of stroke.
TM Watchman Left Atrial Appendage Closure Device PROOF OFLEADERSHIP Uniquely engineered for the LAA 1-3 with proven safety and longterm efficacy. 4-8 Patients with AF have a 5x increased risk of stroke.
Introduction What Causes Peripheral Vascular Disease? How Do Doctors Treat Peripheral Vascular Disease?... 9
 Patient Information Table of Contents Introduction... 3 What is Peripheral Vascular Disease?... 5 What Are Some of the Symptoms of Peripheral Vascular Disease?... 7 What Causes Peripheral Vascular Disease?...
Patient Information Table of Contents Introduction... 3 What is Peripheral Vascular Disease?... 5 What Are Some of the Symptoms of Peripheral Vascular Disease?... 7 What Causes Peripheral Vascular Disease?...
The Conservative and Active Management of Post Thrombotic Syndrome
 The Conservative and Active Management of Post Thrombotic Syndrome Stephen Black Consultant Vascular Surgeon Clinical Lead for Venous and Lymphoedema Surgery Guys and St Thomas Hospital London How important
The Conservative and Active Management of Post Thrombotic Syndrome Stephen Black Consultant Vascular Surgeon Clinical Lead for Venous and Lymphoedema Surgery Guys and St Thomas Hospital London How important
On Which Criteria Do You Select Your Stent for Ilio-femoral Venous Obstruction? North American Point of View
 On Which Criteria Do You Select Your Stent for Ilio-femoral Venous Obstruction? North American Point of View Peter Gloviczki, MD Ying Huang, MD, PhD Division of Vascular and Endovascular Surgery, Mayo
On Which Criteria Do You Select Your Stent for Ilio-femoral Venous Obstruction? North American Point of View Peter Gloviczki, MD Ying Huang, MD, PhD Division of Vascular and Endovascular Surgery, Mayo
Understanding aneurysms and flow diversion treatment
 Surpass Streamline Flow Diverter See package insert for complete indications, contraindications, warnings and instructions for use. INTENDED USE / INDICATIONS FOR USE The Surpass Streamline Flow Diverter
Surpass Streamline Flow Diverter See package insert for complete indications, contraindications, warnings and instructions for use. INTENDED USE / INDICATIONS FOR USE The Surpass Streamline Flow Diverter
Improved clinical outcomes Evidence on venous thrombectomy followed by stenting
 Improved clinical outcomes Evidence on venous thrombectomy followed by stenting Michael K. W. Lichtenberg, MD, FESC Vascular Centre Arnsberg, Germany Venous Centre Arnsberg, Germany Disclosure Speaker
Improved clinical outcomes Evidence on venous thrombectomy followed by stenting Michael K. W. Lichtenberg, MD, FESC Vascular Centre Arnsberg, Germany Venous Centre Arnsberg, Germany Disclosure Speaker
PEARL Registry Update Overview Venous Arterial AV Access
 PEARL Registry Update Overview Venous Arterial AV Access PEARL Registry Overview (as of 10 Sep12*) Overview Venous Arterial AV Access HOME Topic Data Support Comments Study Design Prospective, non-randomized,
PEARL Registry Update Overview Venous Arterial AV Access PEARL Registry Overview (as of 10 Sep12*) Overview Venous Arterial AV Access HOME Topic Data Support Comments Study Design Prospective, non-randomized,
Durable outcomes. Proven performance.
 Durable outcomes. Proven performance. GORE EXCLUDER AAA Endoprosthesis GORE EXCLUDER Iliac Branch Endoprosthesis GORE EXCLUDER AAA Endoprosthesis The most-studied* EVAR stent graft designed for durable
Durable outcomes. Proven performance. GORE EXCLUDER AAA Endoprosthesis GORE EXCLUDER Iliac Branch Endoprosthesis GORE EXCLUDER AAA Endoprosthesis The most-studied* EVAR stent graft designed for durable
4/30/2018 CLOT+ In patients with an acute proximal deep vein thrombosis, pharmacomechanical catheter-directed thrombolysis does not reduce t
 In patients with an acute proximal deep vein thrombosis, pharmacomechanical catheter-directed thrombolysis does not reduce the rate of post-thrombotic syndrome Question In patients who have symptomatic
In patients with an acute proximal deep vein thrombosis, pharmacomechanical catheter-directed thrombolysis does not reduce the rate of post-thrombotic syndrome Question In patients who have symptomatic
Image-Guided Approach to Treatment of Patients with Nonthrombotic
 Image-Guided Approach to Treatment of Patients with Nonthrombotic May Thurner Syndrome Brian DeRubertis, MD, FACS Associate Professor of Surgery Division of Vascular Surgery UCLA School of Medicine Los
Image-Guided Approach to Treatment of Patients with Nonthrombotic May Thurner Syndrome Brian DeRubertis, MD, FACS Associate Professor of Surgery Division of Vascular Surgery UCLA School of Medicine Los
GORE EXCLUDER AAA Endoprosthesis ANNUAL CLINICAL UPDATE OCTOBER Section I Clinical experience. Section II Worldwide commercial experience
 GORE EXCLUDER AAA Endoprosthesis ANNUAL CLINICAL UPDATE OCTOBER 2018 Abstract This annual clinical update provides a review of the ongoing experience with the GORE EXCLUDER AAA Endoprosthesis used in the
GORE EXCLUDER AAA Endoprosthesis ANNUAL CLINICAL UPDATE OCTOBER 2018 Abstract This annual clinical update provides a review of the ongoing experience with the GORE EXCLUDER AAA Endoprosthesis used in the
Inferior Venacaval Filters Valuable vs. Dangerous Valuable Annie Kulungowski. Department of Surgery Grand Rounds March 24, 2008
 Inferior Venacaval Filters Valuable vs. Dangerous Valuable Annie Kulungowski Department of Surgery Grand Rounds March 24, 2008 History of Vena Cava Filters Virchow-1846-Proposes PE originate from veins
Inferior Venacaval Filters Valuable vs. Dangerous Valuable Annie Kulungowski Department of Surgery Grand Rounds March 24, 2008 History of Vena Cava Filters Virchow-1846-Proposes PE originate from veins
Bard LifeStar Vascular Stent System
 Bard LifeStar Vascular Stent System Instructions For Use (IFU) Bard LifeStar Vascular Stent System Delivery System Diagram 8x60 B05685 Vers.2/12-11 1 Instructions for use Read the Bard LifeStar Vascular
Bard LifeStar Vascular Stent System Instructions For Use (IFU) Bard LifeStar Vascular Stent System Delivery System Diagram 8x60 B05685 Vers.2/12-11 1 Instructions for use Read the Bard LifeStar Vascular
