Hip Joint Prostheses
|
|
|
- Patrick Higgins
- 5 years ago
- Views:
Transcription
1 Translated English of Chinese Standard: YY PHARMACEUTICAL INDUSTRY STANDARD YY ICS OF THE PEOPLE S REPUBLIC OF CHINA C 35 YY Replacing YY Hip Joint Prostheses 髋关节假体 Issued on: December 7, 2005 Implemented on: December 1, 2006 Issued by: China State Food and Drug Administration Page 1 of 27
2 Table of Contents Foreword Scope Normative References Terms, Definitions and Classification Requirements Test Methods Inspection Rules Instructions for Use Marking Packaging Transportation and Storage Appendix A (Normative) List of Material Standards Found Acceptable for the Manufacture of Hip Joint Prostheses Appendix B (Normative) List of Materials Found Acceptable or not Acceptable for the Manufacture of Articulating Surfaces of Hip Joint Prostheses Appendix C (Normative) List of Materials Found Acceptable or Non-acceptable for Metallic Combinations for Non-articulating Contacting Surfaces of Hip Joint Prostheses Bibliography Page 2 of 27
3 Foreword 3.2, 4.3 and 4.4 of this Standard are recommendatory. This Standard is formulated by mainly referencing to ISO "Implants for Surgery - Partial and Total Hip Joint Prostheses - Part 1: Classification and Designation of Dimensions", ISO "Implants for Surgery - Partial and Total Hip Joint Prostheses - Part 2: Articulating Surfaces Made of Metallic, Ceramic and Plastics Materials" (ISO ), ISO "Non-active Surgical Implants - General Requirements", "Non-active Surgical Implants - Joint Replacement Implants - Particular Requirements", ISO "Non-active Surgical Implants - Joint Replacement Implants - Specific Requirements for Hip-joint Replacement Implants", YY/T 0340 "Implants for Surgery - Fundamental Principles", and YY 0341 "General Technological Requirements for Non-active Metallic Surgery Implants of Osteosynthesis". This Standard shall replace YY "Hip Joint Prostheses". Compared with YY , the main changes in this Standard are as follows: - "Definitions" in Chapter 3 adopts the definition part in ISO "Non-active Surgical Implants - Joint Replacement Implants - Specific Requirements for Hip-joint Replacement Implants"; - The technical contents have referenced the content requirements of the latest international standards, national standards, and industry standards; and the various technical contents are re-compiled. Appendixes A, B and C of this Standard are normative. This Standard was proposed by the China State Food and Drug Administration. This Standard shall be under the jurisdiction of National Technical Committee on Implants for Surgery and Orthopaedic Devices of Standardization Administration of China (SAC/TC 110). Drafting organization of this Standard: Baimtec Material Co., Ltd. Chief drafting staffs of this Standard: Tian Bin, Shen Sihong, and Qiao Zhen. Page 3 of 27
4 Hip Joint Prostheses 1 Scope This Standard specifies the definitions, classification, requirements, test methods, inspection rules, marking, packaging, instructions for use, transportation, storage etc. of total-hip and partial-hip joint replacements. This Standard is applicable to the joint prostheses used for the total-hip and partial-hip joint replacement of human body. 2 Normative References The following standards contain provisions which, through reference in this text, constitute provisions of this Standard. For dated reference, the subsequent amendments (excluding corrections) or revisions of these publications do not apply. However, the parties who enter into agreements based on this Standard are encouraged to investigate the possibility of applying the most recent editions of the standards. For undated references, the latest edition of the normative document referred to applies. GB/T Sampling Procedures for Inspection by Attributes - Part 1: Sampling Schemes Indexed by Acceptance Quality Limit (AQL) for Lot-by-lot Inspection (GB/T , ISO , IDT) GB/T 2829 Sampling Procedures and Tables for Periodic Inspection by Attributes (Apply to Inspection of Process Stability) GB/T Test Methods for Infusion, Transfusion, Injection Equipment for Medical Use - Part 1: Chemical Analysis Methods GB/T Test Methods for Infusion, Transfusion, Injection Equipment for Medical Use - Part 2: Biological Test Methods GB/T Biological Evaluation of Medical Devices - Part 7: Ethylene Oxide Sterilization Residuals (GB/T , idt ISO : 1995) GB/T Implants for Surgery Ultra-high Molecular Weight Polyethylene - Part 2: Moulded Forms (GB/T , ISO : 1998, IDT) YY 0117 (All the parts) Implants for Surgery - Forgings, Castings for Bone Joint Prostheses Page 4 of 27
5 YY 0341 General Technological Requirements for Non-active Metallic Surgery Implants of Osteosynthesis YY/T 0343 Liquid Penetrant Inspection of Metallic Surgical Implants (YY/T , ISO 9583: 1993, NEQ) ISO 6474 Implants for Surgery - Ceramic Materials Based on High Purity Alumina ISO Implants for Surgery - Partial and Total Hip Joint Prostheses - Part 1: Classification and Designation of Dimensions ISO Implants for Surgery - Partial and Total Hip Joint Prostheses - Part 2: Articulating Surfaces Made of Metallic, Ceramic and Plastics Materials ISO Implants for Surgery - Partial and Total Hip Joint Prostheses - Part 4: Determination of Endurance Properties and Performance of Stemmed Femoral Components ISO Implants for Surgery - Partial and Total Hip Joint Prostheses - Part 6: Endurance Properties Testing and Performance Requirements of Neck Region of Stemmed Femoral Components ISO Implants for Surgery - Partial and Total Hip Joint Prostheses - Part 8: Endurance Performance of Stemmed Femoral Components with Application of Torsion ISO Implants for Surgery - Ceramic Materials Based on Yttria-stabilized Tetragonal Zirconia (Y-TZP) ISO Hydroxyapatite Implants for Surgery - Hydroxyapatite - Part 2: Coatings of ISO Implants for Surgery - Wear of Total Hip-joint Prostheses - Part 1: Loading and Displacement Parameters for Wear-testing Machines and Corresponding Environmental Conditions for Test ISO Implants for Surgery - Wear of Total Hip-joint Prostheses - Part 2: Methods of Measurement ISO Non-active Surgical Implants - Joint Replacement Implants - Particular Requirements 3 Terms, Definitions and Classification 3.1 Terms and definitions For the purposes of this Standard, the terms and definitions established in ISO Page 5 of 27
6 and ISO and the following ones apply Hip joint replacement Surgical implant that is used to replace one or both of the articulating surfaces of the hip joint Total hip joint replacement Surgical implant that is consisted of a femoral component and an acetabulum component. It is intended to replace both of the articulating surfaces of the hip joints Partial hip joint replacement Surgical implant that is consisted of a femoral component. It is intended to replace the femoral articulating surface of the hip joint. Note: partial hip joint replacement implants incorporate either a bipolar or a unipolar head Acetabulum replacement Surgical implant that is consisted of an acetabulum component. It is intended to replace acetabulum articulating surface of the hip joint Femoral component Monobloc or combined component of a total-hip or partial-hip joint replacement that is intended to be attached to the femur Acetabulum component Monobloc or combined component that is intended to be fixed to biological acetabulum Bipolar head Page 6 of 27
7 4.1.3 The physical properties of the ultra-high-molecular-weight polyethylene materials used for manufacturing hip joint prostheses shall meet the requirements of Table 1 in GB/T The physical and chemical properties of ceramic materials used for manufacturing hip joint prostheses shall meet the requirements of ISO 6474: 1994 or the requirements of Table 1 in ISO 13356: The hydroxyapatite coating of hip joint prostheses shall meet the requirements of ISO Note 1: Appendix B of this Standard provides the list of materials found acceptable or non-acceptable for manufacturing the articulating surfaces of hip joint prostheses; unalloyed titanium and titanium alloys shall not be used as the articulating surfaces of hip joint replacements, unless an appropriate surface treatment is undertaken and demonstrated to be suitable in clinical use. Note 2: Appendix C of this Standard provides the list of acceptable and unacceptable metallic combinations for use in non-articulating bearing surfaces of hip joint prostheses. Note 3: For applications in which two dissimilar metals or alloys, or two metals in different metallurgical states are in contact where articulation is not intended, the material combinations shall not produce unacceptable galvanic effects. 4.2 Surface quality Appearance Metal surfaces The metallic component surfaces of hip joint prostheses shall be free from defects such as scale, tool marks, nicks, scratches, cracks, cavities, sharp edges, or burrs; and shall also be free from embedded or deposited finishing materials or other contaminants Plastic surfaces The articulating surfaces of hip joint prostheses made of plastic materials shall be free from defects such as particulate contamination, chemical discolouration spots, tool marks, nicks, chips, cavities, or cracks Ceramic surfaces The articulating surfaces of hip joint prostheses made of ceramic materials shall be free from defects such as particulate contamination, chemical discolouration spots, tool marks, nicks, chips, cavities, or cracks Surface defects Page 9 of 27
8 results of test shall be recorded Endurance properties of head and neck region of stemmed femoral components The head and neck region of stemmed femoral components shall be tested in accordance with 7.2 of ISO :1992. Note: An international standard with performance requirements is being formulated. 4.4 Wear testing of total hip joint replacements The wear characteristics of total hip joint replacement implants shall be tested in accordance with a controlled, validated and documented procedure. The test implant is consisted of a femoral component with an integral head or a modular head articulating on a metallic, ceramic or UHMWPE acetabulum component. Note: ISO has started to prepare an international standard which will enable the specification of performance requirements based on the test methods given in ISO s Parts 1 and Sterilization Products supplied in sterile The products which are supplied in sterile shall be free from bacterium Implants containing UHMW polyethylene If implants are to be sterilized by ionizing radiation, the accumulated dose of radiation the implants received shall not be greater than 40 kgy Products supplied in non-sterile For products which are supplied in non-sterile, the manufacturer shall specify at least one appropriate method of sterilization. If multiple sterilizations are not allowed, this shall be stated in the product information provided by the manufacturer (see 8.2) Ethylene oxide sterilization residuals For products which are supplied by ethylene oxide sterilization, the manufacturer shall determine the acceptable limit of ethylene oxide sterilization residual that shall not be greater than those specified in GB/T Test Methods 5.1 Materials Page 12 of 27
9 g) When the national quality supervision institution proposes requirement to carry out periodic inspection Prior to periodic inspection, lot-by-lot inspection shall be carried out. Samples shall be taken from the lot that has passed lot-by-lot inspection to undergo the periodic inspection Periodic inspection shall be carried out in accordance with GB/T The rejection classification, test items and sampling scheme of periodic inspection are shown in Table 3. Table 3 Periodic Inspection Rejection classification A B Test group I II III I Test item a b In Sampling scheme 1[0, 1] 3[0, 1] All qualified upon 100% inspection In accordance with relevant standards All qualified accorda nce with relevant standard 3[0, 1] 3[0, 1] s a Products made of materials of the same lot form a material lot. b Products of the same sterilization process form a sterilization lot In principle, if the products are qualified upon periodic inspection, all test items of this period shall be qualified. 7 Instructions for Use In addition to meeting the requirements of the relevant laws and regulations, the instructions for use of hip joint prostheses shall also include the following contents: a) Name, trademark and address of the manufacturer; b) Necessary description for the user to confirm the product; c) Application scope of the product, and the requirements for safety and correct operation; d) Thorough explanation to all disposal to be made prior to use (like sterilization and final assembly, etc.); Page 15 of 27
10 c) Lot number or serial number used for accurate identification; d) Instructions for single-use product; e) Instructions for sterile package contents (e.g.: "STERILE"); f) Sterilization lot number; g) Validity of sterilization; h) Instructions for special purpose (for instance: "Custom Made" or "For Clinical Study Only"); i) Instructions for particular storage and/or delivery conditions and the relevant influence from ambient conditions; j) Warning and/or precautions; k) Sterilization method if necessary. 9 Packaging For each hip joint prosthesis, the packaging shall be designed so that, under conditions specified by the manufacturer for storage, transport and handling (including control of temperature, humidity and ambient pressure, if applicable), it protects against damage and deterioration and does not adversely affect the implant. Implants labelled with "STERILE" shall be packaged so that they remain sterile under normal storage, transport and handling conditions, unless the package which maintains sterility is damaged or opened. 10 Transportation and Storage The requirements for transportation and storage of hip joint prostheses shall comply with the relevant chapters and articles in YY Page 18 of 27
11 Appendix A (Normative) List of Material Standards Found Acceptable for the Manufacture of Hip Joint Prostheses The following materials have been found acceptable through proven use for the manufacture of hip joint prostheses. The inclusion of materials in this appendix does not imply their satisfactory use in any particular application; neither does it relieve the manufacturer from the obligation to undertake a design evaluation. ISO : 1997 Stainless Steel ISO : 1999 Titanium Implants for Surgery - Metallic Materials - Part 1: Wrought Implants for Surgery - Metallic Materials - Part 2: Unalloyed ISO : 1996 Implants for Surgery - Metallic Materials - Part 3: Wrought Titanium 6-aluminium 4-vanadium Alloy ISO : 1996 Implants for Surgery - Metallic Materials - Part 4: Cobalt-chromium-molybdenum Casting Alloy ISO : 1993 Implants for Surgery - Metallic Materials - Part 5: Wrought Cobalt-chromium-tungsten-nickel Alloy ISO : 1997 Implants for Surgery - Metallic Materials - Part 6: Wrought Cobalt-nickel-chromium-molybdenum Alloy ISO : 1994 Implants for Surgery - Metallic Materials - Part 7: Forgeable and Cold-formed Cobalt-chromium-nickel-molybdenum-iron Alloy ISO : 1997 Implants for Surgery - Metallic Materials - Part 8: Wrought Cobalt-nickel-chromium-molybdenum-tungsten-iron Alloy ISO : 1992 Implants for Surgery - Metallic Materials - Part 9: Wrought High Nitrogen Stainless Steel ISO : 1994 Implants for Surgery - Metallic Materials - Part 11: Wrought Titanium 6-aluminium 7-niobium Alloy Page 19 of 27
12 ISO : 1996 Implants for Surgery - Metallic Materials - Part 12: Wrought Cobalt-chromium-molybdenum Alloy ISO 5833: 2002 Implants for Surgery - Acrylic Resin Cements ISO 6474: 1994 Implants for Surgery - Ceramic Materials Based on High Purity Alumina ISO 13356: 1997 Implants for Surgery - Ceramic Materials Based on Yttria-stabilized Tetragonal Zirconia (Y-TZP) ISO : 2000 Implants for Surgery - Hydroxyapatite - Part 2: Coatings of Hydroxyapatite ISO 13782: 1996 Implants for Surgery - Metallic Materials - Unalloyed Tantalum for Surgical Implant Applications GB/T Implants for Surgery - Ultra-high Molecular Weight Polyethylene - Part 1: Powder Form (ISO : 1998, IDT) GB/T Implants for Surgery - Ultra-high Molecular Weight Polyethylene -Part 2: Moulded Forms (ISO : 1998, IDT) GB Stainless Steel for Surgical Implants (ISO : 1997, MOD) GB Casting Co-Cr-Mo Alloy for Surgical Implants GB/T Wrought Titanium and Titanium Alloy for Surgical Implants YY Implants for Surgery - Acrylic Resin Cements (ISO 5833: 1992, IDT) Page 20 of 27
13 Appendix B (Normative) List of Materials Found Acceptable or not Acceptable for the Manufacture of Articulating Surfaces of Hip Joint Prostheses B.1 Suitable material combinations For the articulating surfaces of hip joint replacement implants, the following combinations of the materials listed in Appendix A have been found to be acceptable in particular applications, provided that adequate attention is paid to in design, surface finish and surface treatment: a) Wrought stainless steel (ISO ) / UHMWPE (ISO , ISO ); b) Wrought high nitrogen stainless steel (ISO ) / UHMWPE (ISO , ISO ); c) Cobalt-chromium-molybdenum casting alloy (ISO , GB 17100) / UHMWPE (ISO , ISO ); d) Wrought cobalt-chromium tungsten-nickel alloy (ISO ) / UHMWPE (ISO , ISO ); e) Forgeable and cold-formed cobalt-chromium-nickel-molybdenum-iron alloy (ISO ) / UHMWPE (ISO , ISO ); f) Wrought cobalt-nickel-chromium-molybdenum-tungsten-iron alloy (ISO ) / UHMWPE (ISO , ISO ); g) Wrought titanium 6-aluminium 4-vanadium alloy 1) (ISO ) / UHMWPE (ISO , ISO ); h) Wrought titanium 6-aluminium 7-niobium alloy 1) (ISO ) / UHMWPE (ISO , ISO ); i) Ceramic materials based on alumina (ISO 6474) / UHMWPE (ISO , ISO ); j) Ceramic materials based on zirconia (ISO 13356) / UHMWPE (ISO , ISO ); 1) Particular attention to surface treatment of the articulating surface may be necessary. Page 21 of 27
14 Appendix C (Normative) List of Materials Found Acceptable or Non-acceptable for Metallic Combinations for Non-articulating Contacting Surfaces of Hip Joint Prostheses C.1 Suitable combinations of dissimilar metals for non-articulating contacting surfaces For some applications where one metal or alloy is in contact with another and articulation is not intended, the following metallic combinations involving the metals listed in Appendix A have been found to be acceptable and can be used, provided that the adequate attention is paid to in design, surface finish, surface treatment, and metallurgical conditions: a) Cobalt based alloys (ISO , ISO , ISO , ISO , ISO 58328, ISO ) / titanium based alloys (ISO 58323, ISO , ISO ); b) Cobalt based alloys (ISO , ISO , ISO , ISO , ISO , ISO ) / other cobalt based alloys (ISO , ISO , ISO , ISO , ISO , ISO ); c) Stainless steel (ISO , ISO ) / titanium based alloys (ISO , ISO , ISO ); d) Stainless steel (ISO , ISO ) / stainless steel (ISO , ISO ); e) Stainless steel (ISO ) / cobalt based alloys (ISO , ISO , ISO , ISO , ISO , ISO ). C.2 Non-acceptable metallic combinations for non-articulating contacting surfaces For some applications where one metal or alloy is in contact with another and articulation is not intended, the following combinations of the metals listed in Appendix A have been found not acceptable: a) Stainless steel (excluding the material described in ISO ) / cobalt based alloys (ISO , ISO , ISO , ISO , ISO , Page 24 of 27
15 Bibliography [1] ISO 6018 Orthopaedic implants - General requirements for marking, packaging and labelling [2] ISO Implants for Surgery - Partial and Total Hip Joint Prostheses - Part 4: Determination of Endurance Properties and Performance of Stemmed Femoral Components [3] ISO Implants for Surgery - Partial and Total Hip Joint Prostheses - Part 6: Endurance Properties Testing and Performance Requirements of Neck Region of Stemmed Femoral Components [4] ISO Implants for Surgery - Partial and Total Hip Joint Prostheses - Part 8: Endurance Performance of Stemmed Femoral Components with Application of Torsion [5] ISO Implants for Surgery - Partial and Total Hip Joint Prostheses - Part 9: Determination of Resistance to Torque of Head Fixation of Stemmed Femoral Components [6] ISO 8828 Implants for Surgery - Guidance on Care and Handling of Orthopaedic Implants [7] ISO/TR 9325 Implants for Surgery - Partial and Total Hip Joint Prostheses - Recommendations for Simulators for Evaluation of Hip Joint Prostheses [8] ISO/TR 9326 Implants for Surgery - Partial and Total Hip Joint Prostheses - Guidance for Laboratory Evaluation of Change of Form of Bearing Surfaces [9] GB/T Biological Evaluation of Medical Devices - Part 7: Ethylene Oxide Sterilization Residuals (GB/T , idt ISO : 1995) [10] ISO Implants for Surgery - Wear of Total Hip-joint Prostheses - Part 1: Loading and Displacement Parameters for Wear-testing Machines and Corresponding Environmental Conditions for Test [11] ISO Implants for Surgery - Wear of Total Hip-joint Prostheses - Part 2: Methods of Measurement [12] ISO : 2000 Implants for Surgery - Hydroxyapatite - Part 1: Ceramic Hydroxyapatite [13] ISO : 2000 Implants for Surgery - Hydroxyapatite - Part 2: Coatings of Hydroxyapatite Page 26 of 27
YY Translated English of Chinese Standard: YY
 Translated English of Chinese Standard: YY0341-2009 www.chinesestandard.net Sales@ChineseStandard.net YY ICS 11.040.40 C 35 Pharmaceutical Industry Standard of the People s Republic of China YY/T 0341-2009
Translated English of Chinese Standard: YY0341-2009 www.chinesestandard.net Sales@ChineseStandard.net YY ICS 11.040.40 C 35 Pharmaceutical Industry Standard of the People s Republic of China YY/T 0341-2009
YY/T PHARMACEUTICAL INDUSTRY STANDARD OF THE PEOPLE S REPUBLIC OF CHINA. Translated English of Chinese Standard: YY/T
 PHARMACEUTICAL INDUSTRY STANDARD OF THE PEOPLE S REPUBLIC OF CHINA Translated English of Chinese Standard: YY/T0520-2009 www.chinesestandard.net Sales@ChineseStandard.net YY ICS 11.060.10 C 33 YY/T 0520-2009
PHARMACEUTICAL INDUSTRY STANDARD OF THE PEOPLE S REPUBLIC OF CHINA Translated English of Chinese Standard: YY/T0520-2009 www.chinesestandard.net Sales@ChineseStandard.net YY ICS 11.060.10 C 33 YY/T 0520-2009
YY Translated English of Chinese Standard: YY
 Translated English of Chinese Standard: YY0497-2005 www.chinesestandard.net Sales@ChineseStandard.net YY ICS 11.040.20 C 31 Pharmaceutical National Standard of the People s Republic of China YY 0497-2005
Translated English of Chinese Standard: YY0497-2005 www.chinesestandard.net Sales@ChineseStandard.net YY ICS 11.040.20 C 31 Pharmaceutical National Standard of the People s Republic of China YY 0497-2005
Non-active surgical implants Joint replacement implants Particular requirements (ISO 21534:2007)
 BRITISH STANDARD Non-active surgical implants Joint replacement implants Particular requirements (ISO 21534:2007) ICS 11.040.40 NO COPYING WITHOUT BSI PERMISSION EXCEPT AS PERMITTED BY COPYRIGHT LAW BS
BRITISH STANDARD Non-active surgical implants Joint replacement implants Particular requirements (ISO 21534:2007) ICS 11.040.40 NO COPYING WITHOUT BSI PERMISSION EXCEPT AS PERMITTED BY COPYRIGHT LAW BS
ISO INTERNATIONAL STANDARD. Non-active surgical implants Joint replacement implants Particular requirements
 INTERNATIONAL STANDARD ISO 21534 Second edition 2007-10-01 Non-active surgical implants Joint replacement implants Particular requirements Implants chirurgicaux non actifs Implants de remplacement d'articulation
INTERNATIONAL STANDARD ISO 21534 Second edition 2007-10-01 Non-active surgical implants Joint replacement implants Particular requirements Implants chirurgicaux non actifs Implants de remplacement d'articulation
YY / ISO 11070:1998
 Translated English of Chinese Standard: YY0450.1-2003 www.chinesestandard.net Sales@ChineseStandard.net PHARMACEUTICAL INDUSTRY STANDARD YY OF THE PEOPLE S REPUBLIC OF CHINA ICS 11.120.30 C 48 YY 0450.1-2003
Translated English of Chinese Standard: YY0450.1-2003 www.chinesestandard.net Sales@ChineseStandard.net PHARMACEUTICAL INDUSTRY STANDARD YY OF THE PEOPLE S REPUBLIC OF CHINA ICS 11.120.30 C 48 YY 0450.1-2003
--> Buy True-PDF --> Auto-delivered in 0~10 minutes. YY/T Translated English of Chinese Standard: YY/T
 Translated English of Chinese Standard: YY/T0343-2002 www.chinesestandard.net Sales@ChineseStandard.net PHARMACEUTICAL INDUSTRY STANDARD YY OF THE PEOPLE S REPUBLIC OF CHINA ICS 11.040.40 C 30 Liquid penetrant
Translated English of Chinese Standard: YY/T0343-2002 www.chinesestandard.net Sales@ChineseStandard.net PHARMACEUTICAL INDUSTRY STANDARD YY OF THE PEOPLE S REPUBLIC OF CHINA ICS 11.040.40 C 30 Liquid penetrant
YY/T Translated English of Chinese Standard: YYT PHARMACEUTICAL INDUSTRY STANDARD
 Translated English of Chinese Standard: YYT0325-2016 www.chinesestandard.net Buy True-PDF Auto-delivery. Sales@ChineseStandard.net PHARMACEUTICAL INDUSTRY STANDARD YY OF THE PEOPLE S REPUBLIC OF CHINA
Translated English of Chinese Standard: YYT0325-2016 www.chinesestandard.net Buy True-PDF Auto-delivery. Sales@ChineseStandard.net PHARMACEUTICAL INDUSTRY STANDARD YY OF THE PEOPLE S REPUBLIC OF CHINA
ISO INTERNATIONAL STANDARD
 INTERNATIONAL STANDARD ISO 7206-2 Third edition 2011-04-01 Implants for surgery Partial and total hip joint prostheses Part 2: Articulating surfaces made of metallic, ceramic and plastics materials Implants
INTERNATIONAL STANDARD ISO 7206-2 Third edition 2011-04-01 Implants for surgery Partial and total hip joint prostheses Part 2: Articulating surfaces made of metallic, ceramic and plastics materials Implants
YY/T Translated English of Chinese Standard: YY/T PHARMACEUTICAL INDUSTRY STANDARD
 Translated English of Chinese Standard: YY/T1518-2017 www.chinesestandard.net Buy True-PDF Auto-delivery. Sales@ChineseStandard.net PHARMACEUTICAL INDUSTRY STANDARD YY OF THE PEOPLE S REPUBLIC OF CHINA
Translated English of Chinese Standard: YY/T1518-2017 www.chinesestandard.net Buy True-PDF Auto-delivery. Sales@ChineseStandard.net PHARMACEUTICAL INDUSTRY STANDARD YY OF THE PEOPLE S REPUBLIC OF CHINA
YY Translated English of Chinese Standard: YY PHARMACEUTICAL INDUSTRY STANDARD
 Translated English of Chinese Standard: YY0766-2009 www.chinesestandard.net Sales@ChineseStandard.net PHARMACEUTICAL INDUSTRY STANDARD YY OF THE PEOPLE S REPUBLIC OF CHINA ICS 11.040.70 C 41 YY 0766-2009
Translated English of Chinese Standard: YY0766-2009 www.chinesestandard.net Sales@ChineseStandard.net PHARMACEUTICAL INDUSTRY STANDARD YY OF THE PEOPLE S REPUBLIC OF CHINA ICS 11.040.70 C 41 YY 0766-2009
GB / ISO :2006
 Translated English of Chinese Standard: GB18280.2-2015 www.chinesestandard.net Sales@ChineseStandard.net ICS 11.080.01 C 47 NATIONAL STANDARD OF THE PEOPLE'S REPUBLIC OF CHINA GB / ISO 11137-2:2006 Partly
Translated English of Chinese Standard: GB18280.2-2015 www.chinesestandard.net Sales@ChineseStandard.net ICS 11.080.01 C 47 NATIONAL STANDARD OF THE PEOPLE'S REPUBLIC OF CHINA GB / ISO 11137-2:2006 Partly
YY/T Translated English of Chinese Standard: YY/T
 Translated English of Chinese Standard: YY/T1218-2013 www.chinesestandard.net Email: Sales@ChineseStandard.net YY ICS 11.100 C 44 PHARMACEUTICAL INDUSTRY STANDARD OF THE PEOPLE S REPUBLIC OF CHINA YY/T
Translated English of Chinese Standard: YY/T1218-2013 www.chinesestandard.net Email: Sales@ChineseStandard.net YY ICS 11.100 C 44 PHARMACEUTICAL INDUSTRY STANDARD OF THE PEOPLE S REPUBLIC OF CHINA YY/T
GB/T Translated English of Chinese Standard: GB/T
 Translated English of Chinese Standard: GB/T 19634-2005 www.chinesestandard.net Sales@ChineseStandard.net GB ICS 11.100 C 44 National Standard of the People s Republic of China GB/T 19634-2005 In vitro
Translated English of Chinese Standard: GB/T 19634-2005 www.chinesestandard.net Sales@ChineseStandard.net GB ICS 11.100 C 44 National Standard of the People s Republic of China GB/T 19634-2005 In vitro
ASTM Committee F04 Tuesday 11/13/2012
 ASTM Committee F04 Tuesday 11/13/2012 Centennial Regency Ballroom Ballroom III V 7:00 AM 7:00 AM 8:00 AM 8:00 AM 10:00 AM 10:00 AM 11:00 AM 11:00 AM F04 Celebration of Committee F04's 50th Anniversary
ASTM Committee F04 Tuesday 11/13/2012 Centennial Regency Ballroom Ballroom III V 7:00 AM 7:00 AM 8:00 AM 8:00 AM 10:00 AM 10:00 AM 11:00 AM 11:00 AM F04 Celebration of Committee F04's 50th Anniversary
GB/T Translated English of Chinese Standard: GB/T NATIONAL STANDARD OF THE
 Translated English of Chinese Standard: GB/T24253-2009 www.chinesestandard.net Sales@ChineseStandard.net GB ICS 59.080.30 W 04 NATIONAL STANDARD OF THE PEOPLE'S REPUBLIC OF CHINA GB/T 24253-2009 Textiles
Translated English of Chinese Standard: GB/T24253-2009 www.chinesestandard.net Sales@ChineseStandard.net GB ICS 59.080.30 W 04 NATIONAL STANDARD OF THE PEOPLE'S REPUBLIC OF CHINA GB/T 24253-2009 Textiles
National Food Safety Standard Infant Formula
 Translated English of Chinese Standard: GB 10765-2010 www.chinesestandard.net Email: Sales@ChineseStandard.net NATIONAL STANDARD GB OF THE PEOPLE S REPUBLIC OF CHINA GB 10765-2010 National Food Safety
Translated English of Chinese Standard: GB 10765-2010 www.chinesestandard.net Email: Sales@ChineseStandard.net NATIONAL STANDARD GB OF THE PEOPLE S REPUBLIC OF CHINA GB 10765-2010 National Food Safety
This document is a preview generated by EVS
 INTERNATIONAL STANDARD ISO 7206-12 First edition 2016-10-01 Implants for surgery Partial and total hip joint prostheses Part 12: Deformation test method for acetabular shells Implants chirurgicaux Prothèses
INTERNATIONAL STANDARD ISO 7206-12 First edition 2016-10-01 Implants for surgery Partial and total hip joint prostheses Part 12: Deformation test method for acetabular shells Implants chirurgicaux Prothèses
INTERNATIONAL STANDARD
 INTERNATIONAL STANDARD ISO 4074 First edition 2002-02-15 Natural latex rubber condoms Requirements and test methods Préservatifs masculins en latex de caoutchouc naturel Exigences et méthodes d'essai Reference
INTERNATIONAL STANDARD ISO 4074 First edition 2002-02-15 Natural latex rubber condoms Requirements and test methods Préservatifs masculins en latex de caoutchouc naturel Exigences et méthodes d'essai Reference
--> Buy True-PDF --> Auto-delivered in 0~10 minutes. GB
 Translated English of Chinese Standard GB15980-1995 Translated by: www.chinesestandard.net Email: Sales@ChineseStandard.net ICS 11.120.20 C 59 National Standard of the People s Republic of China GB Hygienic
Translated English of Chinese Standard GB15980-1995 Translated by: www.chinesestandard.net Email: Sales@ChineseStandard.net ICS 11.120.20 C 59 National Standard of the People s Republic of China GB Hygienic
Instructions for Use
 Instructions for Use Non Cemented Stem without Coating Legend of Symbols Use on Packages Number in the Catalogue; Sterile Product Sterilized by Gamma Radiation; Manufacturing Date; Batch Number; Expiration
Instructions for Use Non Cemented Stem without Coating Legend of Symbols Use on Packages Number in the Catalogue; Sterile Product Sterilized by Gamma Radiation; Manufacturing Date; Batch Number; Expiration
--> Buy True-PDF --> Auto-delivered in 0~10 minutes. GB Translated English of Chinese Standard: GB
 Translated English of Chinese Standard: GB1987-2007 www.chinesestandard.net Sales@ChineseStandard.net NATIONAL STANDARD OF THE PEOPLE S REPUBLIC OF CHINA GB ICS 67.220.20 X 41 Replacing GB 1987-1986 Food
Translated English of Chinese Standard: GB1987-2007 www.chinesestandard.net Sales@ChineseStandard.net NATIONAL STANDARD OF THE PEOPLE S REPUBLIC OF CHINA GB ICS 67.220.20 X 41 Replacing GB 1987-1986 Food
GB Translated English of Chinese Standard: GB NATIONAL STANDARD OF
 Translated English of Chinese Standard: GB13078-2001 www.chinesestandard.net Sales@ChineseStandard.net NATIONAL STANDARD OF GB THE PEOPLE S REPUBLIC OF CHINA ICS 65.120 B 46 GB 13078-2001 Replacing GB
Translated English of Chinese Standard: GB13078-2001 www.chinesestandard.net Sales@ChineseStandard.net NATIONAL STANDARD OF GB THE PEOPLE S REPUBLIC OF CHINA ICS 65.120 B 46 GB 13078-2001 Replacing GB
DRAFT GUIDANCE DOCUMENT List of Recognized Standards
 DRAFT GUIDANCE DOCUMENT This guidance document is being distributed for comment purposes only. Published by authority of the Minister of Health Draft Date 2016/04/20 Health Products and Food Branch TABLE
DRAFT GUIDANCE DOCUMENT This guidance document is being distributed for comment purposes only. Published by authority of the Minister of Health Draft Date 2016/04/20 Health Products and Food Branch TABLE
Salto Talaris Total Ankle Prosthesis
 Instructions for Use Salto Talaris Total Ankle Prosthesis Federal (USA) law restricts this device to sale by or on the order of a physician. Important The manufacturer recommends that all personnel responsible
Instructions for Use Salto Talaris Total Ankle Prosthesis Federal (USA) law restricts this device to sale by or on the order of a physician. Important The manufacturer recommends that all personnel responsible
PRO-DENSE Bone Graft Substitute The following languages are included in this packet:
 EN PRO-DENSE Bone Graft Substitute 133486-11 The following languages are included in this packet: English (en) For additional languages, visit our website www.wmt.com Then click on the Prescribing Information
EN PRO-DENSE Bone Graft Substitute 133486-11 The following languages are included in this packet: English (en) For additional languages, visit our website www.wmt.com Then click on the Prescribing Information
Sterile hypodermic needles for single use Requirements and test methods
 Provläsningsexemplar / Preview INTERNATIONAL STANDARD ISO 7864 Fourth edition 2016-08-01 Sterile hypodermic needles for single use Requirements and test methods Aiguilles hypodermiques stériles, non réutilisables
Provläsningsexemplar / Preview INTERNATIONAL STANDARD ISO 7864 Fourth edition 2016-08-01 Sterile hypodermic needles for single use Requirements and test methods Aiguilles hypodermiques stériles, non réutilisables
ISO INTERNATIONAL STANDARD. Male condoms Requirements and test methods for condoms made from synthetic materials
 INTERNATIONAL STANDARD ISO 23409 First edition 2011-02-15 Male condoms Requirements and test methods for condoms made from synthetic materials Préservatifs masculins Exigences et méthodes d'essai pour
INTERNATIONAL STANDARD ISO 23409 First edition 2011-02-15 Male condoms Requirements and test methods for condoms made from synthetic materials Préservatifs masculins Exigences et méthodes d'essai pour
Intravascular catheters Sterile and single-use catheters. Part 5: Over-needle peripheral catheters
 Provläsningsexemplar / Preview INTERNATIONAL STANDARD ISO 10555-5 Second edition 2013-06-15 Intravascular catheters Sterile and single-use catheters Part 5: Over-needle peripheral catheters Cathéters intravasculaires
Provläsningsexemplar / Preview INTERNATIONAL STANDARD ISO 10555-5 Second edition 2013-06-15 Intravascular catheters Sterile and single-use catheters Part 5: Over-needle peripheral catheters Cathéters intravasculaires
Implants for surgery Partial and total hip joint prostheses. Part 12: Deformation test method for acetabular shells
 INTERNATIONAL STANDARD ISO 7206-12 First edition 2016-10-01 Implants for surgery Partial and total hip joint prostheses Part 12: Deformation test method for acetabular shells Implants chirurgicaux Prothèses
INTERNATIONAL STANDARD ISO 7206-12 First edition 2016-10-01 Implants for surgery Partial and total hip joint prostheses Part 12: Deformation test method for acetabular shells Implants chirurgicaux Prothèses
鱼类罐头卫生标准 Hygienic standard for canned fish
 Translated English of Chinese Standard: GB14939-2005 Translated by: www.chinesestandard.net Wayne Zheng et al. Email: Wayne@ChineseStandard.net ICS 67.040 C 53 GB National Standard of the People's Republic
Translated English of Chinese Standard: GB14939-2005 Translated by: www.chinesestandard.net Wayne Zheng et al. Email: Wayne@ChineseStandard.net ICS 67.040 C 53 GB National Standard of the People's Republic
Doc. No.: IU/8 for. Rev. No.: 07 ResTOR Prosthesis
 0434 Page 1 of 5 For use by an Accredited Orthopaedic Surgeon only IMPORTANT MEDICAL INFORMATION PURPOSE: The ResTOR system is designed to restore structural skeletal stability and enable functional joint
0434 Page 1 of 5 For use by an Accredited Orthopaedic Surgeon only IMPORTANT MEDICAL INFORMATION PURPOSE: The ResTOR system is designed to restore structural skeletal stability and enable functional joint
PRO-DENSE BONE GRAFT SUBSTITUTE
 PRO-DENSE BONE GRAFT SUBSTITUTE 152917-0 The following languages are included in this packet: For additional languages, visit our website www.wright.com. Then click on the option. For additional information
PRO-DENSE BONE GRAFT SUBSTITUTE 152917-0 The following languages are included in this packet: For additional languages, visit our website www.wright.com. Then click on the option. For additional information
--> Buy True-PDF --> Auto-delivered in 0~10 minutes. NB/T
 Translated English of Chinese Standard: GB/T47013.5-2015 www.chinesestandard.net Sales@ChineseStandard.net ENERGY INDUSTRY STANDARD OF THE PEOPLE S REPUBLIC OF CHINA NB ICS 77.140.20 H 26 GB/T 47013.5-2015
Translated English of Chinese Standard: GB/T47013.5-2015 www.chinesestandard.net Sales@ChineseStandard.net ENERGY INDUSTRY STANDARD OF THE PEOPLE S REPUBLIC OF CHINA NB ICS 77.140.20 H 26 GB/T 47013.5-2015
Provläsningsexemplar / Preview INTERNATIONAL STANDARD. Rubber condoms - Part 1: Requirements
 INTERNATIONAL STANDARD IS0 4074-I Second edition 1996-l 2-01 Rubber condoms - Part 1: Requirements Prbservatifs masculins en caoutchouc - Parfie 1: Exigences Reference number IS0 4074-I :1996(E) IS0 4074-I
INTERNATIONAL STANDARD IS0 4074-I Second edition 1996-l 2-01 Rubber condoms - Part 1: Requirements Prbservatifs masculins en caoutchouc - Parfie 1: Exigences Reference number IS0 4074-I :1996(E) IS0 4074-I
Individual oncological implants
 Individual oncological implants Catalogue - individual oncological implants ONCOLOGICAL Materials for implants List of materials MATERIAL ISO ČSN DIN ASTM Stainless steel 5832-1 17350 výběr 17 443 W.Nr.
Individual oncological implants Catalogue - individual oncological implants ONCOLOGICAL Materials for implants List of materials MATERIAL ISO ČSN DIN ASTM Stainless steel 5832-1 17350 výběr 17 443 W.Nr.
Retrieval and analysis of surgical implants. Part 2: Analysis of retrieved surgical implants
 INTERNATIONAL STANDARD ISO 12891-2 Second edition 2014-09-15 Retrieval and analysis of surgical implants Part 2: Analysis of retrieved surgical implants Retrait et analyse des implants chirurgicaux Partie
INTERNATIONAL STANDARD ISO 12891-2 Second edition 2014-09-15 Retrieval and analysis of surgical implants Part 2: Analysis of retrieved surgical implants Retrait et analyse des implants chirurgicaux Partie
COMPETENT AUTHORITY (UK) MEDICAL DEVICES DIRECTIVES GUIDANCE NOTES FOR MANUFACTURERS OF DENTAL APPLIANCES
 COMPETENT AUTHORITY (UK) 10 EC MEDICAL DEVICES DIRECTIVES GUIDANCE NOTES FOR MANUFACTURERS OF DENTAL APPLIANCES (CUSTOM MADE DEVICES) Updated March 2008 CONTENTS PAGE Introduction 3 Definition of dental
COMPETENT AUTHORITY (UK) 10 EC MEDICAL DEVICES DIRECTIVES GUIDANCE NOTES FOR MANUFACTURERS OF DENTAL APPLIANCES (CUSTOM MADE DEVICES) Updated March 2008 CONTENTS PAGE Introduction 3 Definition of dental
ISO INTERNATIONAL STANDARD
 INTERNATIONAL STANDARD ISO 22794 First edition 2007-07-15 Corrected version 2009-01-15 Dentistry Implantable materials for bone filling and augmentation in oral and maxillofacial surgery Contents of a
INTERNATIONAL STANDARD ISO 22794 First edition 2007-07-15 Corrected version 2009-01-15 Dentistry Implantable materials for bone filling and augmentation in oral and maxillofacial surgery Contents of a
METAL HEMI SYSTEM
 METAL HEMI SYSTEM 152206-0 The following languages are included in this packet: English (en) Deutsch (de) Nederlands (nl) Français (fr) Español (es) Italiano (it) Português (pt) 中文 - Chinese (sch) Türkçe
METAL HEMI SYSTEM 152206-0 The following languages are included in this packet: English (en) Deutsch (de) Nederlands (nl) Français (fr) Español (es) Italiano (it) Português (pt) 中文 - Chinese (sch) Türkçe
CONSENSUS ORTHOPEDICS INC. UNISYN HIP SYSTEM
 CONSENSUS ORTHOPEDICS INC. UNISYN HIP SYSTEM Instructions for Use (IFU) IMPORTANT INFORMATION FOR SURGEON: PLEASE READ PRIOR TO IMPLANTING THIS DEVICE IN A CLINICAL SETTING. THE SURGEON SHOULD BE FAMILIAR
CONSENSUS ORTHOPEDICS INC. UNISYN HIP SYSTEM Instructions for Use (IFU) IMPORTANT INFORMATION FOR SURGEON: PLEASE READ PRIOR TO IMPLANTING THIS DEVICE IN A CLINICAL SETTING. THE SURGEON SHOULD BE FAMILIAR
This document is a preview generated by EVS
 INTERNATIONAL STANDARD ISO 14708-7 First edition 2013-01-15 Implants for surgery Active implantable medical devices Part 7: Particular requirements for cochlear implant systems Implants chirurgicaux Dispositifs
INTERNATIONAL STANDARD ISO 14708-7 First edition 2013-01-15 Implants for surgery Active implantable medical devices Part 7: Particular requirements for cochlear implant systems Implants chirurgicaux Dispositifs
This document is a preview generated by EVS
 INTERNATIONAL STANDARD ISO 8537 Third edition 2016-03-15 Sterile single-use syringes, with or without needle, for insulin Seringues à insuline, stériles, non réutilisables, avec ou sans aiguille Reference
INTERNATIONAL STANDARD ISO 8537 Third edition 2016-03-15 Sterile single-use syringes, with or without needle, for insulin Seringues à insuline, stériles, non réutilisables, avec ou sans aiguille Reference
This document is a preview generated by EVS
 INTERNATIONAL STANDARD ISO 12891-2 Second edition 2014-09-15 Retrieval and analysis of surgical implants Part 2: Analysis of retrieved surgical implants Retrait et analyse des implants chirurgicaux Partie
INTERNATIONAL STANDARD ISO 12891-2 Second edition 2014-09-15 Retrieval and analysis of surgical implants Part 2: Analysis of retrieved surgical implants Retrait et analyse des implants chirurgicaux Partie
--> Buy True-PDF --> Auto-delivered in 0~10 minutes. YY Translated English of Chinese Standard: YY
 Translated English of Chinese Standard: YY0831-2011 www.chinesestandard.net Email: Sales@ChineseStandard.net ICS 11.040.50 YY C 43 PHARMACEUTICAL INDUSTRY STANDARD OF THE PEOPLE S REPUBLIC OF CHINA YY
Translated English of Chinese Standard: YY0831-2011 www.chinesestandard.net Email: Sales@ChineseStandard.net ICS 11.040.50 YY C 43 PHARMACEUTICAL INDUSTRY STANDARD OF THE PEOPLE S REPUBLIC OF CHINA YY
ISO INTERNATIONAL STANDARD. Dentistry Implants Dynamic fatigue test for endosseous dental implants
 INTERNATIONAL STANDARD ISO 14801 Second edition 2007-11-15 Dentistry Implants Dynamic fatigue test for endosseous dental implants Art dentaire Implants Essai de fatigue dynamique pour implants dentaires
INTERNATIONAL STANDARD ISO 14801 Second edition 2007-11-15 Dentistry Implants Dynamic fatigue test for endosseous dental implants Art dentaire Implants Essai de fatigue dynamique pour implants dentaires
Dentistry Dental amalgam
 Provläsningsexemplar / Preview INTERNATIONAL STANDARD ISO 24234 Second edition 2015-05-01 Dentistry Dental amalgam Médecine bucco-dentaire Amalgame dentaire Reference number ISO 24234:2015(E) ISO 2015
Provläsningsexemplar / Preview INTERNATIONAL STANDARD ISO 24234 Second edition 2015-05-01 Dentistry Dental amalgam Médecine bucco-dentaire Amalgame dentaire Reference number ISO 24234:2015(E) ISO 2015
Part 1: General requirements
 Provläsningsexemplar / Preview INTERNATIONAL STANDARD ISO 10555-1 Second edition 2013-06-15 Corrected version 2014-01-15 Intravascular catheters Sterile and single-use catheters Part 1: General requirements
Provläsningsexemplar / Preview INTERNATIONAL STANDARD ISO 10555-1 Second edition 2013-06-15 Corrected version 2014-01-15 Intravascular catheters Sterile and single-use catheters Part 1: General requirements
CAUTION: Ceramic liners are not approved for use in the United States.
 Total Hip Prostheses, Self-Centering Hip Prostheses and Hemi-Hip Prostheses IMPORTANT: This essential product information sheet does not include all of the information necessary for selection and use of
Total Hip Prostheses, Self-Centering Hip Prostheses and Hemi-Hip Prostheses IMPORTANT: This essential product information sheet does not include all of the information necessary for selection and use of
SURGICAL TECHNIQUE CEMENTED & PRESS-FIT UNIFIED INSTRUMENTATION INTRAOPERATIVE FLEXIBILITY PROVEN BIOMECHANICS
 SURGICAL TECHNIQUE CEMENTED & PRESS-FIT UNIFIED INSTRUMENTATION INTRAOPERATIVE FLEXIBILITY PROVEN BIOMECHANICS INTRODUCTION The Summit Tapered Hip System s comprehensive set of implants and instruments
SURGICAL TECHNIQUE CEMENTED & PRESS-FIT UNIFIED INSTRUMENTATION INTRAOPERATIVE FLEXIBILITY PROVEN BIOMECHANICS INTRODUCTION The Summit Tapered Hip System s comprehensive set of implants and instruments
JJF Translated English of Chinese Standard: JJF
 Translated English of Chinese Standard: JJF1070.2-2011 www.chinesestandard.net Sales@ChineseStandard.net JJF NATIONAL METROLOGICAL TECHNICAL SPECIFICATION OF THE PEOPLE S REPUBLIC OF CHINA JJF 1070.2-2011
Translated English of Chinese Standard: JJF1070.2-2011 www.chinesestandard.net Sales@ChineseStandard.net JJF NATIONAL METROLOGICAL TECHNICAL SPECIFICATION OF THE PEOPLE S REPUBLIC OF CHINA JJF 1070.2-2011
Enhancing stability and increasing range of motion. Metasul LDH Large Diameter Head
 Enhancing stability and increasing range of motion Metasul LDH Large Diameter Head The Metasul large diameter head technology is the result of in-depth research, development and clinical experience that
Enhancing stability and increasing range of motion Metasul LDH Large Diameter Head The Metasul large diameter head technology is the result of in-depth research, development and clinical experience that
Furlong H-A.C. THR System
 Important Information Please read prior to use in a clinical setting. The Surgeon should be familiar with the operative technique. Caution Federal (U.S.A) law restricts this device to sale by or on the
Important Information Please read prior to use in a clinical setting. The Surgeon should be familiar with the operative technique. Caution Federal (U.S.A) law restricts this device to sale by or on the
This document is a preview generated by EVS
 INTERNATIONAL STANDARD ISO 10555-5 Second edition 2013-06-15 Intravascular catheters Sterile and single-use catheters Part 5: Over-needle peripheral catheters Cathéters intravasculaires Cathéters stériles
INTERNATIONAL STANDARD ISO 10555-5 Second edition 2013-06-15 Intravascular catheters Sterile and single-use catheters Part 5: Over-needle peripheral catheters Cathéters intravasculaires Cathéters stériles
CONSENSUS ORTHOPEDICS INC.
 CONSENSUS ORTHOPEDICS INC. CONSENSUS HIP SYSTEMS Instructions for Use (IFU) IMPORTANT INFORMATION FOR SURGEON: PLEASE READ PRIOR TO IMPLANTING THIS DEVICE IN A CLINICAL SETTING. THE SURGEON SHOULD BE FAMILIAR
CONSENSUS ORTHOPEDICS INC. CONSENSUS HIP SYSTEMS Instructions for Use (IFU) IMPORTANT INFORMATION FOR SURGEON: PLEASE READ PRIOR TO IMPLANTING THIS DEVICE IN A CLINICAL SETTING. THE SURGEON SHOULD BE FAMILIAR
ISO INTERNATIONAL STANDARD. Sterile single-use intravascular catheter introducers
 INTERNATIONAL STANDARD ISO 11070 First edition 1998-05-01 Sterile single-use intravascular catheter introducers Introducteurs de cathéters intravasculaires stériles, non réutilisables A Reference number
INTERNATIONAL STANDARD ISO 11070 First edition 1998-05-01 Sterile single-use intravascular catheter introducers Introducteurs de cathéters intravasculaires stériles, non réutilisables A Reference number
TriboFit Hip System. Issue 3 - August 2013 English
 Important Information: Please read prior to use in a clinical setting. The surgeon should be familiar with the operative technique and all the information in this insert. Description: The TriboFit Hip
Important Information: Please read prior to use in a clinical setting. The surgeon should be familiar with the operative technique and all the information in this insert. Description: The TriboFit Hip
ISO INTERNATIONAL STANDARD. Dentistry Reversible-irreversible hydrocolloid impression material systems
 INTERNATIONAL STANDARD ISO 13716 First edition 1999-05-01 Dentistry Reversible-irreversible hydrocolloid impression material systems Art dentaire Systèmes de produits pour empreintes à base d'hydrocolloïdes
INTERNATIONAL STANDARD ISO 13716 First edition 1999-05-01 Dentistry Reversible-irreversible hydrocolloid impression material systems Art dentaire Systèmes de produits pour empreintes à base d'hydrocolloïdes
Metallic Internal Fixation Devices The following languages are included in this packet:
 Metallic Internal Fixation Devices 150848-0 The following languages are included in this packet: English (en) Deutsch (de) Nederlands (nl) Français (fr) Español (es) Italiano (it) Português (pt) 中文 - Chinese
Metallic Internal Fixation Devices 150848-0 The following languages are included in this packet: English (en) Deutsch (de) Nederlands (nl) Français (fr) Español (es) Italiano (it) Português (pt) 中文 - Chinese
BIOLOX delta Option Ceramic Femoral Head System
 BIOLOX delta Option Ceramic Femoral Head System Product Features and Instructions for Use Knees Hips Extremities Cement and Accessories PMI Technology Product Features BIOLOX delta ceramic heads are composed
BIOLOX delta Option Ceramic Femoral Head System Product Features and Instructions for Use Knees Hips Extremities Cement and Accessories PMI Technology Product Features BIOLOX delta ceramic heads are composed
ISO INTERNATIONAL STANDARD. Non-destructive testing Penetrant testing Part 1: General principles
 INTERNATIONAL STANDARD ISO 3452-1 First edition 2008-09-01 Non-destructive testing Penetrant testing Part 1: General principles Essais non destructifs Examen par ressuage Partie 1: Principes généraux Reference
INTERNATIONAL STANDARD ISO 3452-1 First edition 2008-09-01 Non-destructive testing Penetrant testing Part 1: General principles Essais non destructifs Examen par ressuage Partie 1: Principes généraux Reference
DRAFT UGANDA STANDARD
 DRAFT UGANDA STANDARD DUS 1677 First Edition 217 Poultry feed premix Specification Reference number DUS 1677: 217 UNBS 217 DUS 1677: 217 Compliance with this standard does not, of itself confer immunity
DRAFT UGANDA STANDARD DUS 1677 First Edition 217 Poultry feed premix Specification Reference number DUS 1677: 217 UNBS 217 DUS 1677: 217 Compliance with this standard does not, of itself confer immunity
BIOLOX delta Option Ceramic Femoral Head System. Product Features and Instructions for Use
 BIOLOX delta Option Ceramic Femoral Head System Product Features and Instructions for Use Product Features BIOLOX delta ceramic heads are composed of 75% aluminum oxide and approximately 25% zirconia.
BIOLOX delta Option Ceramic Femoral Head System Product Features and Instructions for Use Product Features BIOLOX delta ceramic heads are composed of 75% aluminum oxide and approximately 25% zirconia.
JRI Thompson Hemiarthroplasty
 JRI ORTHOPAEDICS LTD 18 Churchill Way, 35A Business Park, Chapeltown, Sheffield, S35 2PY, UK Instructions for Use JRI Thompson Hemiarthroplasty Page 1 of 6 English 3 Page 2 of 6 Important Information Please
JRI ORTHOPAEDICS LTD 18 Churchill Way, 35A Business Park, Chapeltown, Sheffield, S35 2PY, UK Instructions for Use JRI Thompson Hemiarthroplasty Page 1 of 6 English 3 Page 2 of 6 Important Information Please
Nondestructive Testing Procedure: Liquid Penetrant Testing
 MOGAS INDUSTRIES, INC. Nondestructive Testing Procedure: Liquid Penetrant Testing Specification: NDT 0200 Version: 2.0 November 10, 2014 Approved by: Signature Paul T. Marks Consulting Level III ACCP Professional
MOGAS INDUSTRIES, INC. Nondestructive Testing Procedure: Liquid Penetrant Testing Specification: NDT 0200 Version: 2.0 November 10, 2014 Approved by: Signature Paul T. Marks Consulting Level III ACCP Professional
Document No Rev. B Pg. 1 of 5 Approvals / Date Title: INSTRUCTIONS FOR USE DJO SURGICAL MODULAR REVISION HIP SYSTEM AND ACETABULAR CAGE
 Document No. 0400-0205 Rev. B Pg. 1 of 5 Approvals / Date Title: INSTRUCTIONS FOR USE DJO SURGICAL MODULAR REVISION HIP SYSTEM AND ACETABULAR CAGE Revision ECO Date ECO Summary of Changes Deann Rector
Document No. 0400-0205 Rev. B Pg. 1 of 5 Approvals / Date Title: INSTRUCTIONS FOR USE DJO SURGICAL MODULAR REVISION HIP SYSTEM AND ACETABULAR CAGE Revision ECO Date ECO Summary of Changes Deann Rector
Important Notice to Users in the United States: General Safety Instructions
 SLR-PLUS Revision Stem, SLR-PLUS Revision Stem Lateral Smith & Nephew Orthopaedics AG Oberneuhofstrasse 10d CH-6340 Baar Phone + 41 41 766 22 22 swiss.implants@smith-nephew.com non-sterile cemented non-
SLR-PLUS Revision Stem, SLR-PLUS Revision Stem Lateral Smith & Nephew Orthopaedics AG Oberneuhofstrasse 10d CH-6340 Baar Phone + 41 41 766 22 22 swiss.implants@smith-nephew.com non-sterile cemented non-
POLARCUP Dual Mobility System. General Safety Instructions. Instructions for Use. Duty to inform the patient. Implant passport
 Important Notice to Users in the United States: This package insert is for product distributed in US only. U.S. Federal law restricts this device to sale by or on the order of a physician. General Safety
Important Notice to Users in the United States: This package insert is for product distributed in US only. U.S. Federal law restricts this device to sale by or on the order of a physician. General Safety
Whey powders Specification
 ICS 67.100.10 DMS 1397:2016 First edition CODEX STAN 289:1995 IDT DRAFT MALAWI STANDARD Whey powders Specification NOTE: This is a draft proposal and it shall neither be used nor regarded as a Malawi standard
ICS 67.100.10 DMS 1397:2016 First edition CODEX STAN 289:1995 IDT DRAFT MALAWI STANDARD Whey powders Specification NOTE: This is a draft proposal and it shall neither be used nor regarded as a Malawi standard
CAUTION Federal law (USA) restricts this device to sale, by or on the order of a physician.
 CAUTION Federal law (USA) restricts this device to sale, by or on the order of a physician. ENGLISH Mpact 3D Metal Implants and Augments 3D Metal INSTRUCTION FOR USE Important notice: the device(s) can
CAUTION Federal law (USA) restricts this device to sale, by or on the order of a physician. ENGLISH Mpact 3D Metal Implants and Augments 3D Metal INSTRUCTION FOR USE Important notice: the device(s) can
Ceramic Femoral Heads and Acetabular Cup Liners
 Important Information: Please read prior to use in a clinical setting. The Surgeon should be familiar with the operative technique. Caution: Federal (U.S.A) law restricts this device to sale by or on the
Important Information: Please read prior to use in a clinical setting. The Surgeon should be familiar with the operative technique. Caution: Federal (U.S.A) law restricts this device to sale by or on the
GB Translated English of Chinese Standard: GB NATIONAL STANDARD OF THE
 Translated English of Chinese Standard: GB5009.268-2016 www.chinesestandard.net Buy True-PDF Auto-delivery. Sales@ChineseStandard.net GB NATIONAL STANDARD OF THE PEOPLE S REPUBLIC OF CHINA GB 5009.268-2016
Translated English of Chinese Standard: GB5009.268-2016 www.chinesestandard.net Buy True-PDF Auto-delivery. Sales@ChineseStandard.net GB NATIONAL STANDARD OF THE PEOPLE S REPUBLIC OF CHINA GB 5009.268-2016
ISO INTERNATIONAL STANDARD. Dentistry Casting and baseplate waxes. Art dentaire Cires pour coulée et pour plaque de base
 INTERNATIONAL STANDARD ISO 15854 First edition 2005-07-15 Dentistry Casting and baseplate waxes Art dentaire Cires pour coulée et pour plaque de base Reference number ISO 15854:2005(E) ISO 2005 PDF disclaimer
INTERNATIONAL STANDARD ISO 15854 First edition 2005-07-15 Dentistry Casting and baseplate waxes Art dentaire Cires pour coulée et pour plaque de base Reference number ISO 15854:2005(E) ISO 2005 PDF disclaimer
EVOLVE TRIAD BONE SCREWS
 EN EVOLVE TRIAD BONE SCREWS 146886-1 English (en) The following languages are included in this packet: For additional languages, visit our website www.wmt.com. Then click on the Prescribing Information
EN EVOLVE TRIAD BONE SCREWS 146886-1 English (en) The following languages are included in this packet: For additional languages, visit our website www.wmt.com. Then click on the Prescribing Information
Important Notice to Users in the United States: General Safety Instructions
 POLARSTEM Femoral Stems with Ti/HA (Standard, Lateral, Valgus and Collar) Smith & Nephew Orthopaedics AG Oberneuhofstrasse 10d CH-6340 Baar Phone + 41 41 766 22 22 swiss.implants@smith-nephew.com 02/2017
POLARSTEM Femoral Stems with Ti/HA (Standard, Lateral, Valgus and Collar) Smith & Nephew Orthopaedics AG Oberneuhofstrasse 10d CH-6340 Baar Phone + 41 41 766 22 22 swiss.implants@smith-nephew.com 02/2017
Optimizing function Maximizing survivorship Accelerating recovery
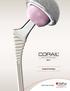 Surgical Technique Optimizing Function Maximizing Survivorship Accelerating Recovery The company believes in an approach to patient treatment that places equal importance on: Optimizing function Maximizing
Surgical Technique Optimizing Function Maximizing Survivorship Accelerating Recovery The company believes in an approach to patient treatment that places equal importance on: Optimizing function Maximizing
This document is a preview generated by EVS
 INTERNATIONAL STANDARD ISO 16408 Second edition 2015-08-15 Dentistry Oral care products Oral rinses Médecine bucco-dentaire Produits de soins bucco-dentaire Bains de bouche Reference number ISO 16408:2015(E)
INTERNATIONAL STANDARD ISO 16408 Second edition 2015-08-15 Dentistry Oral care products Oral rinses Médecine bucco-dentaire Produits de soins bucco-dentaire Bains de bouche Reference number ISO 16408:2015(E)
National Joint Replacement Registry. Lay Summary 2015 Annual Report Hip and Knee Replacement
 National Joint Replacement Registry Lay Summary 2015 Annual Report Hip and Knee Replacement SUPPLEMENTARY REPORT 2015 TABLE OF CONTENTS Introduction... 1 A brief history of the Registry origins... 1 The
National Joint Replacement Registry Lay Summary 2015 Annual Report Hip and Knee Replacement SUPPLEMENTARY REPORT 2015 TABLE OF CONTENTS Introduction... 1 A brief history of the Registry origins... 1 The
For use by an Accredited Orthopaedic Surgeon only
 Page 1 of 5 For use by an Accredited Orthopaedic Surgeon only 1. Purpose: External fixators are intended to aid in surgical stabilization following operative procedures to treat fractures, enable correction
Page 1 of 5 For use by an Accredited Orthopaedic Surgeon only 1. Purpose: External fixators are intended to aid in surgical stabilization following operative procedures to treat fractures, enable correction
Duraloc CONSTRAINED LINER
 SURGICAL TECHNIQUE Duraloc CONSTRAINED LINER A COMPREHENSIVE ACETABULAR REVISION SYSTEM DURALOC CONSTRAINED LINER Introduction Dislocation is the most common postoperative complication in total hip reconstruction.
SURGICAL TECHNIQUE Duraloc CONSTRAINED LINER A COMPREHENSIVE ACETABULAR REVISION SYSTEM DURALOC CONSTRAINED LINER Introduction Dislocation is the most common postoperative complication in total hip reconstruction.
DRAFT UGANDA STANDARD
 DRAFT UGANDA STANDARD DUS 2037 First Edition 2018-mm-dd Kombucha Specification Reference number DUS 2037: 2018 UNBS 2018 DUS 2030: 2018 Compliance with this standard does not, of itself confer immunity
DRAFT UGANDA STANDARD DUS 2037 First Edition 2018-mm-dd Kombucha Specification Reference number DUS 2037: 2018 UNBS 2018 DUS 2030: 2018 Compliance with this standard does not, of itself confer immunity
SL-PLUS MIA Stem, SL-PLUS MIA Stem Lateral, SL-PLUS/ INTEGRATION-PLUS MIA Stem with Ti/HA, SL-PLUS/INTEGRATION-PLUS MIA Stem Lateral with Ti/HA
 SL-PLUS MIA Stem, SL-PLUS MIA Stem Lateral, SL-PLUS/ INTEGRATION-PLUS MIA Stem with Ti/HA, SL-PLUS/INTEGRATION-PLUS MIA Stem Lateral with Ti/HA Smith & Nephew Orthopaedics AG Oberneuhofstrasse 10d CH-6340
SL-PLUS MIA Stem, SL-PLUS MIA Stem Lateral, SL-PLUS/ INTEGRATION-PLUS MIA Stem with Ti/HA, SL-PLUS/INTEGRATION-PLUS MIA Stem Lateral with Ti/HA Smith & Nephew Orthopaedics AG Oberneuhofstrasse 10d CH-6340
Section of total knee replacement. Total Knee Replacement System. Knieendoprothesen System. Système de prothèse totale de genou
 Section of total knee replacement Total Knee Replacement System Knieendoprothesen System Système de prothèse totale de genou Introduction: This knee system features great versality with its modular component
Section of total knee replacement Total Knee Replacement System Knieendoprothesen System Système de prothèse totale de genou Introduction: This knee system features great versality with its modular component
Hip Resurfacing System
 Hip Resurfacing System The Arthrosurface HemiCAP Hip Hemiarthroplasty System restores the articular surface geometry of the femoral head and preserves functional structures using an innovative 3 dimensional
Hip Resurfacing System The Arthrosurface HemiCAP Hip Hemiarthroplasty System restores the articular surface geometry of the femoral head and preserves functional structures using an innovative 3 dimensional
[ICESTM-2018] ISSN Impact Factor
![[ICESTM-2018] ISSN Impact Factor [ICESTM-2018] ISSN Impact Factor](/thumbs/88/117546298.jpg) GLOBAL JOURNAL OF ENGINEERING SCIENCE AND RESEARCHES MODELING AND FINITE ELEMENT ANALYSIS OF KNEE JOINT PROSTHESIS U.D.S.Prathap varma *1,S.Rajesh 2, B.Suresh Kumar 3 & P.Rama Murthy Raju 4 *1 M.TechScholar,
GLOBAL JOURNAL OF ENGINEERING SCIENCE AND RESEARCHES MODELING AND FINITE ELEMENT ANALYSIS OF KNEE JOINT PROSTHESIS U.D.S.Prathap varma *1,S.Rajesh 2, B.Suresh Kumar 3 & P.Rama Murthy Raju 4 *1 M.TechScholar,
SCOPE OF ACCREDITATION TO ISO/IEC 17025:2005
 SCOPE OF ACCREDITATION TO ISO/IEC 17025:2005 EMPIRICAL TESTING CORP. 4628 Northpark Drive Colorado Springs, CO 80918 Dawn Lissy Phone: 719 264 9937 dlissy@empiricaltesting.com MECHANICAL Valid To: January
SCOPE OF ACCREDITATION TO ISO/IEC 17025:2005 EMPIRICAL TESTING CORP. 4628 Northpark Drive Colorado Springs, CO 80918 Dawn Lissy Phone: 719 264 9937 dlissy@empiricaltesting.com MECHANICAL Valid To: January
ISO INTERNATIONAL STANDARD
 INTERNATIONAL STANDARD ISO 15197 First edition 2003-05-01 In vitro diagnostic test systems Requirements for blood-glucose monitoring systems for self-testing in managing diabetes mellitus Systèmes d'essais
INTERNATIONAL STANDARD ISO 15197 First edition 2003-05-01 In vitro diagnostic test systems Requirements for blood-glucose monitoring systems for self-testing in managing diabetes mellitus Systèmes d'essais
ISO INTERNATIONAL STANDARD. Anaesthetic and respiratory equipment Nebulizing systems and components
 INTERNATIONAL STANDARD ISO 27427 First edition 2009-05-01 Anaesthetic and respiratory equipment Nebulizing systems and components Matériel d'anesthésie et de réanimation respiratoire Systèmes de nébulisation
INTERNATIONAL STANDARD ISO 27427 First edition 2009-05-01 Anaesthetic and respiratory equipment Nebulizing systems and components Matériel d'anesthésie et de réanimation respiratoire Systèmes de nébulisation
Implants for surgery Active implantable medical devices. Part 7: Particular requirements for cochlear implant systems
 Provläsningsexemplar / Preview INTERNATIONAL STANDARD ISO 14708-7 First edition 2013-01-15 Implants for surgery Active implantable medical devices Part 7: Particular requirements for cochlear implant systems
Provläsningsexemplar / Preview INTERNATIONAL STANDARD ISO 14708-7 First edition 2013-01-15 Implants for surgery Active implantable medical devices Part 7: Particular requirements for cochlear implant systems
Non-destructive testing Penetrant testing. Part 2: Testing of penetrant materials
 Provläsningsexemplar / Preview INTERNATIONAL STANDARD ISO 3452-2 Third edition 2013-11-15 Non-destructive testing Penetrant testing Part 2: Testing of penetrant materials Essais non destructifs Examen
Provläsningsexemplar / Preview INTERNATIONAL STANDARD ISO 3452-2 Third edition 2013-11-15 Non-destructive testing Penetrant testing Part 2: Testing of penetrant materials Essais non destructifs Examen
ISO INTERNATIONAL STANDARD. Steel products Employer's qualification system for non-destructive testing (NDT) personnel
 INTERNATIONAL STANDARD ISO 11484 Second edition 2009-02-15 Steel products Employer's qualification system for non-destructive testing (NDT) personnel Produits en acier Système de qualification, par l'employeur,
INTERNATIONAL STANDARD ISO 11484 Second edition 2009-02-15 Steel products Employer's qualification system for non-destructive testing (NDT) personnel Produits en acier Système de qualification, par l'employeur,
This document is a preview generated by EVS
 INTERNATIONAL STANDARD ISO 16635-2 First edition 2014-09-15 Dentistry Dental rubber dam instruments Part 2: Clamp forceps Médecine bucco-dentaire Instruments de digue dentaire en caoutchouc Partie 2: Pinces
INTERNATIONAL STANDARD ISO 16635-2 First edition 2014-09-15 Dentistry Dental rubber dam instruments Part 2: Clamp forceps Médecine bucco-dentaire Instruments de digue dentaire en caoutchouc Partie 2: Pinces
ISO INTERNATIONAL STANDARD
 INTERNATIONAL STANDARD ISO 8529-3 First edition 1998-11-15 Reference neutron radiations Part 3: Calibration of area and personal dosimeters and determination of their response as a function of neutron
INTERNATIONAL STANDARD ISO 8529-3 First edition 1998-11-15 Reference neutron radiations Part 3: Calibration of area and personal dosimeters and determination of their response as a function of neutron
Cement Polished Tapered Stems of 12/14 Taper. 96 mm 98 mm 104 mm 110 mm 116 mm 122 mm 128 mm. Ceramic Femoral Head. Outer Diameter
 Design Philosophy Cementless Stems of 12/14 Taper Stem Length Neck shaft Angle STEM-N3 STEM-N4 STEM-N5 STEM-N6 STEM-N7 STEM-N8 STEM-N9 123 mm 125 mm 130 mm 135 mm 140 mm 145 mm 150 mm 132 Cement Polished
Design Philosophy Cementless Stems of 12/14 Taper Stem Length Neck shaft Angle STEM-N3 STEM-N4 STEM-N5 STEM-N6 STEM-N7 STEM-N8 STEM-N9 123 mm 125 mm 130 mm 135 mm 140 mm 145 mm 150 mm 132 Cement Polished
Sterilization of health care products Radiation. Part 2: Establishing the sterilization dose
 Provläsningsexemplar / Preview INTERNATIONAL STANDARD ISO 11137-2 Third edition 2013-06-01 Sterilization of health care products Radiation Part 2: Establishing the sterilization dose Stérilisation des
Provläsningsexemplar / Preview INTERNATIONAL STANDARD ISO 11137-2 Third edition 2013-06-01 Sterilization of health care products Radiation Part 2: Establishing the sterilization dose Stérilisation des
For use by an Accredited Orthopaedic Surgeon only
 Instructions use Page 1 of 6 For use by an Accredited Orthopaedic Surgeon only 1. DEVICE DESCRIPTION: The advancement of partial and total hip replacement has provided surgeons with the means of restoring
Instructions use Page 1 of 6 For use by an Accredited Orthopaedic Surgeon only 1. DEVICE DESCRIPTION: The advancement of partial and total hip replacement has provided surgeons with the means of restoring
DUAL MOBILITY CUP, CEMENTED OR CEMENTLESS
 HIP Documentation DUAL MOBILITY CUP, CEMENTED OR CEMENTLESS >>DUAL MOBILITY ACETABULAR CUPS Dual mobility acetabular cup P r Gilles Bousquet invented the concept of dual mobility more than thirty years
HIP Documentation DUAL MOBILITY CUP, CEMENTED OR CEMENTLESS >>DUAL MOBILITY ACETABULAR CUPS Dual mobility acetabular cup P r Gilles Bousquet invented the concept of dual mobility more than thirty years
