Analysis and Validation of Putative Substances Involved in Fatal Poisonings
|
|
|
- Bruce Pearson
- 5 years ago
- Views:
Transcription
1 J. Med. Toxicol. (2012) 8: DOI /s TOXICOLOGY INVESTIGATION Analysis and Validation of Putative Substances Involved in Fatal Poisonings Christopher K. Hansen & John Kashani & Bruce Ruck & Steven Marcus Published online: 8 December 2011 # American College of Medical Toxicology 2011 Abstract Each year, poison control centers throughout the United States respond to over 4 million calls for help in treating individuals exposed to toxic substances. Although most cases develop no or minimal clinical effects, a small proportion of patients who receive medical care for overdoses with poison center consultation expire. When such cases are investigated by a medical examiner, the postmortem toxicology results may show substances other than those considered in the consultation with the poison center. We sought to determine the characteristics of discordance in fatal cases between the toxic substances reported to a regional poison control center and postmortem toxicology results. We conducted a retrospective study of the New Jersey regional poison control center records of all fatal cases between the years 1986 and Substances reported as putative agents to the poison center were compared to the postmortem toxicology results obtained by the medical examiner. The frequencies and characteristics surrounding discordance were examined. Of the 708 fatal cases reported to our poison center within the study Funding The authors did not receive funding of any kind for this study. Data Presentation Preliminary data presented at the 2008 North American Congress of Clinical Toxicology, Toronto, Canada. C. K. Hansen : J. Kashani : B. Ruck : S. Marcus New Jersey Poison Information and Education System, Department of Preventive Medicine and Community Health, UMDNJ-New Jersey Medical School, Newark, NJ, USA S. Marcus (*) New Jersey Poison Information and Education System, 140 Bergen Street G1600, Newark, NJ , USA smarcus@njpies.org period, complete postmortem toxicological evaluations were available for 206 (29.0%). Comparison of putative agents between information obtained by history and at postmortem evaluation showed discordance in 41 (19.9%). In a substantial number of fatal cases receiving poison center consultation, substances were found at the time of postmortem examination that were not considered in the poison center consultation. The reasons for this discordance may include a lack of thorough history-taking or a cognitive bias to the substances initially reported. Keywords Fatal poisoning. Poison control center. Postmortem Introduction Poisoning and drug overdose both intentional and unintentional are leading causes of morbidity and mortality in the United States [1]. In 2007, there were 40,059 intentional and unintentional poisoning deaths reported in the United States [2]. The economic costs of poisoning are approximately $26 billion in medical expenses annually, making up 6% of the economic costs of all injuries in the United States [3]. Nationally, regional poison control centers (RPCCs) respond to over 4 million calls for help in treating individuals thought to be exposed to toxic substances. Although the vast majority of such calls are determined to be non life-threatening and are treated outside of a health care facility, some cases are deemed to be potentially severe enough to warrant emergent management in a hospital. For these patients, RPCCs play an important role by providing telephone toxicology consultation to the treating medical professionals [4]. Despite this consultation, a proportion of patients expire.
2 J. Med. Toxicol. (2012) 8: In most of the United States, accidental deaths are reportable illnesses and are often investigated with postmortem examinations. If a postmortem is performed, the investigation routinely includes an autopsy and toxicologic testing of blood and other tissue specimens for a core panel of substances as well as others chosen at the discretion of the medical examiner. It was our experience that, in a substantial number of patients who received RPCC consultation and later expired, the postmortem toxicology results did not correspond to the data collected during the initial call to the RPCC. To our knowledge, no previous study has specifically examined discrepancies in the medications or substances reported to the poison center and those found postmortem. Our main objective was to identify and analyze such discrepancies and to describe the frequencies and characteristics surrounding any discordance. Methods We conducted a retrospective analysis of deceased individuals whose healthcare provider contacted the New Jersey Poison Information and Education System (NJPIES) for consultation following a drug overdose or toxic exposure between 1986 and NJPIES is the only RPCC for the entire state of New Jersey (population 8,707,739) and operates a toll-free telephone hotline that provides consultation to the lay public and healthcare providers on the medical management of poisonings and toxic exposures. Every call to the center generates a medical record that is stored in a centralized computer database where patient demographics and data pertinent to the presentation and management of the patient are recorded. All cases are managed by specialists in poison information, credentialed as either physicians, nurses, or clinical pharmacists with oversight provided by medical toxicologists. If a patient is determined to have expired, the hospital record and results of the postmortem examination are requested by the RPCC's medical director. New Jersey has a medical examiner system that encompasses three regional medical examiner offices covering seven of the 21 counties in the state. The remaining 14 counties each operate their own medical examiner office for deaths that occur within their respective county. All autopsies were conducted by a board certified forensic pathologist. The seven counties in the regional medical examiner system utilize a centralized state toxicology laboratory for forensic toxicological analysis. This laboratory is run by a board certified forensic toxicologist. The remaining medical examiner offices utilize a private laboratory that tests for an identical core panel of substances. The basic panel did not change drastically over the study period and is listed in Table 1. Any substances added to the original 1986 core panel are noted. Additionally, if there was any suspicion by the medical examiner of a substance not found in the core panel such as an insecticide or detergent, this could be specially added to the toxicological analysis. We analyzed all deaths between Janurary 1, 1986 and December 31, Demographic information on callers including the year, age, sex, location of the call by county, medical qualifications of the caller (i.e. physician, nurse, paramedic, etc.), the substances believed to have been taken or exposed to and the estimated amount of the exposure, and any follow-up advice given by the poison specialist over the phone were extracted and entered into the dataset. These were mandatory data fields completed by the Specialist in Poison Information (SPI). If a patient's substance list was updated by follow-up calls to the RPCC during their treatment, these substances were included in the dataset up until the time of death. Postmortem toxicology results were also extracted for these cases and entered into the dataset. Cases were included in the study only if they had a complete RPCC call record and had an autopsy performed that included a postmortem toxicological analysis of blood, bile, urine, and brain matter. Cases were excluded if they did not have an autopsy performed, had only an external examination performed, or were missing a postmortem toxicological analysis from their autopsy record. All deaths occurred in either the ED or inpatient unit of a hospital facility. This study did not examine cases where death occurred in the prehospital setting. The result of the toxicological evaluation was compared with the substances reported to the RPCC as extracted from the RPCC case records. We only included toxicology studies performed on blood or other tissues obtained after death. For descriptive purposes, we categorized toxic substances into the following three groups: opioid analgesics and sedatives with abuse potential, other prescription medications that do not fall in the above group, and nonprescription medications and other toxic substances. These categories were not mutually exclusive and cases in which substances fell into more than one category were placed in a fourth category: multiple drugs or toxic substances identified. Substances that were therapeutically administered during the patient s hospital course such as morphine, diazepam, or phenytoin were excluded from the dataset if they were documented for therapeutic use in the patient s record. Cases for which the substances reported to the RPCC were not found in the postmortem toxicological analysis were classified as discordant. If the substances were not known when the RPCC was contacted but were subsequently discovered on postmortem toxicology, the case was classified as unknown substance. If the substances initially reported were not known but were later identified during the course of treatment up until the time of death, the case
3 96 J. Med. Toxicol. (2012) 8: Table 1 Core panel of substances tested 1986 panel of substances (n=98) Volatiles: cannaboids doxepin methamphetamine promethazine acetone carbamazepine doxylamine methapyriline propoxyphene ethanol chloral hydrate flurazepam methaqualone protriptyline isopropanol chlordiazepoxide glutethimide methphenytoin pyrilamine methanol chlorpheniramine hydromorphone morphine (free) quinidine ethylene glycol chlorpromazine haloperidol nitrazepam quinine Drugs and Metabolites: clonazepam hydrocodone nordiazepam salicylates 6-mono-acetyl morphine cocaethylene imipramine norpropoxyphene scopolamine acetaminophen cocaine ketamine nortriptyline secobarbital alprazolam codeine lidocaine oxazepam talbutal amitriptyline cyanide lorazepam papaverine temazepam amobarbital cyproheptadine loxapine pentazocine thiopental amoxapine demoxepam maprotiline pentobarbital thioridazine amphetamine desalkylflurazepam MDA phencyclidine tranylcypromine barbital desipramine MDMA phenmetrazine trazodone benztropine dextromethorphan meperidine phenobarbital triazolam benzoylecgonine diazepam mephobarbital phenylpropanolamine trihexyphenidyl brompheniramine diphenhydramine meprobamate phenytoin trimipramine butabarbital diphenoxylate mesoridazine procainamide tripelennamine butalbital disopyramide methadone procyclidine The following substances were added in 1996: bupivacaine, carisoprodol, chlorpropamide, cyclobenzaprine, diltiazem, ephedrine, estazolam, ethosuximide, fenfluramine, fluoxetine, hydroxyzine, levorphanol, methsuximide, metoprolol, n-acetyl procainamide, oxycodone, paroxitine, phendimetrazine, phenethylamine, phensuximide, phentermine, prazepam, primidone, propranolol, pseudoephedrine, sertraline, theophylline, venlafaxine, and verapamil. Ethchlorvynol was removed from the panel The following substances were added in 2006: bupropion, buspirone, chlorzoxazone, citolapram, colchicine, fentanyl, ibuprofen, mirtazepine, naproxen, nefazodone, olanzepine, oxcarbazepine, propofol, quetiapine, tramadol, warfarin, and zolpidem was classified as either discordant or concordant depending upon the results of the postmortem toxicology. All other cases for which the substances reported were present postmortem were classified as concordant. All data was entered directly into a Microsoft Excel spreadsheet. Data analysis was performed using SPSS (IBM, Somers, NY). Means were analyzed using a oneway analysis of variance. The University of Medicine and Dentistry of New Jersey Institutional Review Board approved this study. Results Between the years 1986 and 2006, there were 708 cases of toxic exposures to the RPCC that resulted in fatality. Of these, 502 were excluded due to incomplete information with 206 (29.0%) fitting the inclusion criteria previously described. The mean age for all cases was 37.8±17.6 years (range 3 days 89 years). One hundred and twenty-two (59.0%) were male. The substances most frequently involved were nonprescription products and other substances (35.0%) followed by multiple drugs or toxic substances (26.2%). Prescription medications accounted for 21.8% of cases and 17% of cases involved opioid analgesics and sedatives with abuse potential. The types of substances involved varied by age group (Fig. 1). The 0 19 year-old age group comprised 12.1% of the study population. The most frequent substances involved in this age group were nonprescription products and other toxic substances (52% of these cases). The year-old age group had the highest proportion of cases in our study with 28.6% of all cases. Nonprescription products and other substances were involved in 39.0% of cases in this age group. The level of medical training of the initial caller to the poison center included 144 (69.6%) physicians, 52 (25.2%) nurses, 5 (2.4%) other healthcare providers, and 5 (2.4%) nonhealthcare providers. The callers classified as other healthcare providers included 1 (0.5%) physician assistant, 2 (1%) emergency medical technicians, and 2 (1%) paramedics. Our analysis of substances reported to the RPCC and those found upon postmortem toxicology testing by the medical examiner showed 41 (19.9%) cases that demonstrated discordance. In 17 of these cases, the substances reported to have been taken did not correspond to the
4 J. Med. Toxicol. (2012) 8: Fig. 1 Types of substances involved in fatal poisonings by age group substances found in the postmortem toxicology analysis (Table 2). In one case, an organophosphate was reported that did not correspond to the dinitrophenol that was discovered in the postmortem toxicology. This was discovered only after additional testing beyond the core panel. In the other 24 cases, no substances were found postmortem (Table 3). For cases in which uncommon substances were reported such as bifenthrin, piroxicam, kelthane, orthene, detergents, and pesticides, specific testing did not reveal these substances. In five cases (2.4%), the substances involved were unable to be identified during the initial call or subsequent follow up with the RPCC but were revealed on postmortem toxicology (Table 4). Among the concordant cases, the mean age was 36.8±17.9 years (range 3 days 89 years) and among discordant cases, the mean age was 41.7±17.0 years (range 4 months 81 years). Among the cases where the substances reported the RPCC were unknown, the mean age was 38.2±9.0 years (range years) (Table 5). Of the discordant cases, 53.7% were male and 61.5% of the concordant cases were male. Forty percent of the unknown substance cases were male. The toxin type most frequently encountered in discordant cases was over-thecounter drugs and other toxic substances, including carbon Table 2 Substances believed to have been taken compared to substances found postmortem when there was discordance (n=17) a Indicates a substance was reported to the RPCC and found postmortem. Substances found postmortem were identified in blood or other tissues obtained after death Substances initially reported to RPCC Substances found postmortem Year Beta-adrenergic antagonist Fluoxetine, hydrocodone 2005 Acetaminophen, bupropion, clonazepam, Diazepam, nordiazepam, oxazepam, 2004 olanzapine, oxcarbazepine propoxyphene, norpropoxyphene Citalopram, diltiazem Benzodiazepines 2002 Amitriptyline a, aspirin a, chlordiazepoxide, Amitriptyline a, salicylate a, nortriptyline, 1999 triazolam, paroxetine, clonazepam nordiazepam Unknown organophosphate Dinitrophenol 1999 Nifedipine Benzodiazapine, morphine, opiates 1999 Raid roach spray Cocaine 1997 Theophylline a Theophylline a, clonazepam, doxepin, nortriptyline 1995 Copper dust Carbon monoxide 1992 Methanol, nortriptyline Ethylene glycol 1991 Codeine a, acetaminophen, ethanol, Codeine a, glutethimide, morphine 1990 propoxyphene napsylate Red Cross toothache (85% Eugenol) Amitriptyline, codeine, ethanol, nortriptyline 1989 Acyclovir, cocaine, lorazepam, mesoridazine, Chlorpheniramine, dextromethorphan, 1989 phenelzine pseudoephedrine Ethanol a Ethanol a, chlordiazepoxide, demoxepam, 1989 doxepin Methadone, phenteramine Benzoylecgonine, cocaine 1989 Lysergic acid diethylamide, phencyclidine Benzoylecgonine, cocaine 1988 Aspirin, butalbital, caffeine, diazepam Ethylene glycol 1986
5 98 J. Med. Toxicol. (2012) 8: Table 3 Substances reported to the RPCC for which no substances were detected in postmortem toxicology (n=24) Substances Year Fenfluramine, phentermine 2006 Acetaminophen, hydrocodone 2005 Bifenthrin 2005 Amlodipine, benzodiazepine, doxepin, valsartan 2004 Amlodipine, clonidine, cocaine, ethanol, lansoprazole, 2003 metformin Piroxicam 2002 Acetaminophen, ethanol, nifedipine 2002 Haloperidol, lorazepam, risperidone, St. John's wort 2001 Botulism toxin 2001 Amitriptyline, heroin 2000 Unknown drug (2 cases) Atenolol, diazepam, fluoxetine, simvastatin 1999 Doxepin, lorazepam, olanzapine, trazedone, venlafaxine 1998 Kelthane, orthene 1997 Doxepin 1997 Phenytoin 1995 Albuterol, cephalexin, theophylline 1995 Laundary detergent 1992 Ethanol, ethylene glycol (2 cases) D-con fogger (insecticide) 1990 Verapamil 1990 Cocaine 1988 monoxide, with 15 (32.6%) cases. Multiple drugs or toxic substances were identified in 11 (26.8%) cases showing discordance and in 41 (25.6%) cases showing concordance. Finally, opioid analgesics and sedatives with abuse potential and other prescription drugs were identified in 9 (22.0%) and 6 (14.6%) cases showing discordance, respectively. We analyzed the time elapsed from the original call to the RPCC until the patient was pronounced dead. Of all the Table 4 Substances found postmortem when an unknown drug was initially reported (n=5) Substances initially reported to RPCC Unknown drug Substances found postmortem Phenytoin, acetone Amitriptyline, nortriptyline Nortriptyline, codeine, oxycodone, desmethylsertraline, phenytoin Benzoylecgonine, ecgonine methyl ester, morphine Cocaine, benzoylecgonine cases included in the study, the mean elapsed time until death was 25.5 hours (SD±64.3 h). The longest time until death was days and the shortest time was 0 h in two cases. These two patients were pronounced dead as the toxic substances were simultaneously being reported to the RPCC. The mean elapsed time until death was 25.4 h (SD± 67.1 h) in the concordant cases, 27.6 h (SD±57.3 h) in the discordant cases, and 14.5 h (SD±18.2 h) in the unknown substances reported group. A one-way analysis of variance showed that mean time until death was not statistically significant among the discordant, concordant, and unknown substance groups, F(2,203) =.01, p=0.91. There were no significant trends over time noted in either the total number of cases or the number of discordant or unknown cases over the course of the study period. Over 3-year intervals, the mean numbers of total (F(6,14) =1.23, p=0.35), discordant (F(6,14) =0.53, p=0.77), and unknown cases (F(6,14)=0.33, p=0.9) in our study were not significantly different. Discussion Over the 20-year study period, the RPCC encountered an average of 35 cases per year that resulted in fatality. Our results demonstrate that a significant amount of fatal cases in which the RPCC was utilized for treatment consultation showed discordance between the initial substances reported and the postmortem toxicology results. Among the types of substances involved in poison related deaths, the most discordant was seen in nonprescription and other toxic substances with 32.6% of cases. This is likely due to the inherent difficulty present in diagnosing the toxic effects of nonpharmaceutical substances such as industrial chemicals. In addition, over-the-counter products and herbal products are often not considered as medications or are considered safe by the nature of being considered over-the-counter. Further, the postmortem toxicological evaluation for OTC substances is limited. Careful attention to history of exposure to these substances is often forgotten. Several studies have described cognitive biases that may affect clinical decision-making. The phenomenon known as anchoring may occur during history-taking in the critically ill patient and cause the clinician to be unduly persuaded by salient features in the initial presentation [5, 6]. Thus, the healthcare provider might anchor to the patients' symptoms early in the presentation and fail to consider alternative diagnoses. In several of the cases in which there was discordance, the substances reported to the RPCC have vastly different clinical presentations than the substances that were identified postmortem. In such cases, an overreliance on history may lead to therapeutic error, which can adversely affect clinical outcomes.
6 J. Med. Toxicol. (2012) 8: Table 5 Characteristics of concordant and discordant cases Case variables Concordant (n=160) Discordant (n=41) Unknown (n=5) All cases (n=206) Age, years, mean±sd 36.8± ± ± ±17.6 Sex, no. (%) Male 98 (61.3) 22 (53.7) 2 (40.0) 122 (59.2) Female 62 (38.8) 19 (46.3) 3 (60.0) 84 (40.8) Drug or toxin types, no. (%) Opioid analgesics and sedatives 24 (15.0) 9 (22.0) 2 (40.0) 35 (17.0) Other prescription medications 38 (23.8) 6 (14.6) 1 (20.0) 45 (21.8) Nonprescription drugs and other toxic substances 57 (35.6) 15 (32.6) 72 (35.0) Multiple drugs or toxic substances identified 41 (25.6) 11 (26.8) 2 (40.0) 54 (26.2) Level of medical training of caller, no. (%) Physician 111 (69.4) 30 (73.2) 3 (60.0) 144 (69.6) Registered nurse 41 (25.6) 9 (22.0) 2 (40.0) 52 (25.2) Other healthcare provider 4 (2.5) 1 (2.4) 5 (2.4) Nonhealthcare provider 4 (2.5) 1 (2.4) 5 (2.4) Time elapsed from initial RPCC call to death, mean±sd 25.4 h±67.1 h 27.6 h±57.3 h 14.5 h±18.2 h 25.5 h±64.3 h SD standard deviation It is unlikely that time available for history-taking played a significant role as the mean time elapsed between the initial call and death of the victim was essentially the same. The interval for concordant and discordant cases was 25.4 h and 27.6 h, respectively. The fact that, on average, providers (both hospital treating staff and RPCC staff) had h to ascertain the substances involved makes it unlikely that history-taking was inhibited by time. It might be informative to study, however, what prompted the call to the RPCC in the first place and at what point in the treatment course the call was made. It has been shown that patients who receive prompt RPCC assistance have measurable reductions in average hospital lengths of stay [7]. Although the elapsed time until death is approximately 1 day, which might seem like ample time to investigate the substances involved, if RPCC consultation is delayed until the patient is in the midst of a downhill course, the treatment options become severely limited and patient prognosis may become worse. It should also be considered that discordance may be related to the difficulty in identifying many of these products and the fact that the postmortem specimens may have been obtained well after the victim's body cleared the substances but had been fatally afflicted before the substance vanished from the lab s ability to identify it. In many instances, the initial caller had spent the longest amount of time with the patient and likely had the most detailed initial history available. An analysis of their level of training found that among the cases in which a physician contacted the RPCC, 22.9% showed discordance, while other and nonhealthcare providers demonstrated discordance in 20% of cases. The numbers are too small to do a statistically meaningful analysis and the finding is complicated by the fact that the initial caller may not have been the primary history taker, but the trend is interesting. It is unclear if the outcome in cases of discordance would have been any different with concordance. In several cases, although the substances reported to the poison control center were discordant with the substances found during postmortem examination, the clinical effects and treatment given may have been identical. The implications of discordance may also affect the way in which medical examiners determine poisoning as the cause of death, placing more emphasis on analysis of the clinical course of the patient. It has been reported that medical examiners and medical toxicologists may disagree on as many as 34% of cases in which poisoning was suspected as the cause of death [8]. It has also been suggested that collaboration between the RPCC and the medical examiners office may enhance the accuracy of identifying poisoning related fatalities, presumably by providing a more in-depth analysis of the clinical course of the patient [9]. The retrospective nature of this study and use of RPCC records present several limitations to our analysis. In each case, information was collected and recorded by SPIs via telephone. Although the data was collected for patient care purposes at the time of first contact and not a research study, SPIs attempted to determine all substances involved during the initial call. The key demographic and substance information were mandatory data fields in the RPCC medical record. While substances were excluded from the dataset if given
7 100 J. Med. Toxicol. (2012) 8: therapeutically in the patient's course, it is possible that not all therapies were recorded in the patient's record. It has been shown, however, that completeness and accuracy of poison center data fields is relatively strong [10, 11]. The small number of cases for which we had medical examiner toxicology results limits our ability to identify strong statistical associations in our data and precluded a more detailed analysis of the potential relationship between age, sex, location, toxic substances identified, medical qualifications of the caller, and trend over time. While the core panel of substances tested by the medical examiner postmortem has changed somewhat during the 20-year period our study covered, many of the substances involved such as the tricyclic antidepressants and benzodiazepines were included in the panel since Furthermore, substances such as zolpidem were not yet in production in 1986 and thus would not have been involved in a toxic ingestion. The fact that there were no significant trends over time noted in either the total number of cases or the number of discordant cases over the course of the study period supports the assumption that changes in the core panel or their sensitivities, over the study period, had little to no effect on our analysis. It is also important to remember that the postmortem examination may have been performed an appreciable time after the patient presented to the hospital and therapeutic interventions may have played a role in obscuring the toxicological evaluation. It should be noted that because a substance is detected in postmortem analysis does not imply a causal relationship with the presenting clinical picture or the death of the patient. However, because the patient's condition prompted a call to the RPCC, there must have been some strong suspicion on the part of the treating professional that a toxic substance was involved. Although our study did not examine premortem laboratory results, toxic levels on these tests may have prompted the initial call to the RPCC. Conclusion It is generally accepted that poison control center consultation enhances management of the poisoned patient. Despite this, there exists an appreciable percentage (19.9%) of cases in which there is a major difference between the substance thought to be responsible for a patients clinical course as reported to a poison center and that found on postmortem evaluation. The reasons for this discordance are currently unknown but may include a lack of thorough history-taking or a cognitive bias to the substances initially reported. It remains to be shown if the outcome in cases of discordance would have been any different with concordance. Additional studies will be needed to determine the implications of such discordance. We suggest that prehospital and immediate hospital treating teams attempt to collect pretreatment blood on all poisoning victims and embargo such bloods until either the patient is discharged from the hospital or the blood is provided to the medical examiner for toxicological evaluation. Further, although our study did not reveal any evidence that discordance impacted on quality of care, the potential does exist. A close working relationship between regional poison centers providing opportunity for review of fatal cases with feedback provided to the treating physicians might be useful in promoting an improved quality of care in poisoned patients. Conflict of Interest References The authors report no conflicts of interest. 1. Bronstein AC, Spyker DA, Cantilena LR Jr et al (2008) 2007 Annual report of the american association of poison control centers' national poison data system (NPDS): 25th annual report. Clin Toxicol (Phila) 46: Centers for Disease Control and Prevention NCfIPaC. Web-based injury statistics query and reporting system (WISQARS) Finkelstein E, Corso P, Miller T (2006) The incidence and economic costs of injury in the United States. Oxford University Press, New York 4. Linakis JG, Frederick KA (1993) Poisoning deaths not reported to the regional poison control center. Ann Emerg Med 22: Croskerry P (2000) The cognitive imperative: thinking about how we think. Acad Emerg Med 7: Kovacs G, Croskerry P (1999) Clinical decision making: an emergency medicine perspective. Acad Emerg Med 6: Vassilev ZP, Marcus SM (2007) The impact of a poison control center on the length of hospital stay for patients with poisoning. J Toxicol Environ Health A 70: Manini AF, Nelson LS, Olsen D, Vlahov D, Hoffman RS (2011) Medical examiner and medical toxicologist agreement on cause of death. Forensic Sci Int 206: Seifert SA, Letsky MC, Farooqi MF, Benson BE, Zumwalt RE (2009) A novel collaboration between a regional poison control center and state medical examiner. Clin Toxicol 47: Jaramillo JE, Marchbanks B, Willis B, Forrester MB (2010) Evaluation of completeness of selected poison control center data fields. J Med Syst 34: Lubbert J, McVoy J, Seifert SA, Jacobitz K (2005) Accuracy and completeness of initial substance identification: is what you hear, what they got. Clin Toxicol (Phila) 43:764
INTOXICATION DEATHS ASSOCIATED WITH DRUGS OF ABUSE OR ALCOHOL BALTIMORE, MARYLAND QUARTERLY REPORT: FOURTH QUARTER, 2008 AND 2008 SUMMARY
 INTOXICATION DEATHS ASSOCIATED WITH DRUGS OF ABUSE OR ALCOHOL BALTIMORE, MARYLAND QUARTERLY REPORT: FOURTH QUARTER, 2008 AND 2008 SUMMARY A report from the Office of Epidemiology and Planning Baltimore
INTOXICATION DEATHS ASSOCIATED WITH DRUGS OF ABUSE OR ALCOHOL BALTIMORE, MARYLAND QUARTERLY REPORT: FOURTH QUARTER, 2008 AND 2008 SUMMARY A report from the Office of Epidemiology and Planning Baltimore
INTOXICATION DEATHS ASSOCIATED WITH DRUGS OF ABUSE OR ALCOHOL BALTIMORE CITY
 2009 FINAL REPORT INTOXICATION DEATHS ASSOCIATED WITH DRUGS OF ABUSE OR ALCOHOL BALTIMORE CITY This report was prepared by: Dr. Jose Arbelaez, M.D. of Baltimore Substance Abuse Systems, and Ryan J. Petteway,
2009 FINAL REPORT INTOXICATION DEATHS ASSOCIATED WITH DRUGS OF ABUSE OR ALCOHOL BALTIMORE CITY This report was prepared by: Dr. Jose Arbelaez, M.D. of Baltimore Substance Abuse Systems, and Ryan J. Petteway,
Forensic Toxicology Scope of Testing and Detection Limits
 Forensic Toxicology Scope of Testing and Detection Limits Table of Contents QUALITATIVE ANALYSES... 2 Volatile Screen by GC/FID... 2 Carbon Monoxide by Microdiffusion... 2 Ethylene Glycol by GC/MS... 2
Forensic Toxicology Scope of Testing and Detection Limits Table of Contents QUALITATIVE ANALYSES... 2 Volatile Screen by GC/FID... 2 Carbon Monoxide by Microdiffusion... 2 Ethylene Glycol by GC/MS... 2
1. Amphetamine (Adderall, desoxyn, Dexedrine,dextrostat, spancap, oxydess II)
 Cross reactivity of various pharmaceuticals against the Drugs of Abuse Screening Panel (posted 2/3/09) The following tables are only applicable to the specific analyzer and reagents used at HRHS main lab.
Cross reactivity of various pharmaceuticals against the Drugs of Abuse Screening Panel (posted 2/3/09) The following tables are only applicable to the specific analyzer and reagents used at HRHS main lab.
3703 Camino del Rio South 100-A San Diego, CA, Phone Fax CLIA# 05D Director: David J.
 Drug Adherence Assessment Report Prescribed Medications: NO MEDICATION LIST PROVIDED CONSISTENT RESULTS - REPORTED MEDICATION DETECTED (PARENT DRUG AND/OR METABOLITE) REPORTED PRESCRIPTION FLAG ANTICIPATED
Drug Adherence Assessment Report Prescribed Medications: NO MEDICATION LIST PROVIDED CONSISTENT RESULTS - REPORTED MEDICATION DETECTED (PARENT DRUG AND/OR METABOLITE) REPORTED PRESCRIPTION FLAG ANTICIPATED
DrugSmartCup & DrugSmartDip Accuracy Report as produced by Ameditech, Inc.
 Accuracy The accuracy of the DrugSmart Drugs of Abuse Tests was evaluated in comparison to commercially available drug screen tests. Sixty (60) negative urine samples collected from presumed non-user volunteers
Accuracy The accuracy of the DrugSmart Drugs of Abuse Tests was evaluated in comparison to commercially available drug screen tests. Sixty (60) negative urine samples collected from presumed non-user volunteers
May Revised Urine Drug Screen Test Menu Effective mid-june Lauren Anthony, MD, MT(ASCP) SBB Medical Director, Allina Health Laboratory
 May 2014 Lauren Anthony, MD, MT(ASCP) SBB Medical Director, Allina Health Laboratory Revised Urine Drug Test Menu Effective mid-june 2014 To better address the complexities of drug screening for different
May 2014 Lauren Anthony, MD, MT(ASCP) SBB Medical Director, Allina Health Laboratory Revised Urine Drug Test Menu Effective mid-june 2014 To better address the complexities of drug screening for different
3703 Camino del Rio South 100-A San Diego, CA, Phone Fax CLIA# 05D years
 Drug Adherence Assessment Report CleanAssure TM (DRIED BLOOD SPOT): Detection Range see NOTES. Prescribed Medications: HYDROMORPHONE (DILAUDID, EXALGO), CYCLOBENZAPRINE (FLEXERIL), METHADONE (METHADOSE),
Drug Adherence Assessment Report CleanAssure TM (DRIED BLOOD SPOT): Detection Range see NOTES. Prescribed Medications: HYDROMORPHONE (DILAUDID, EXALGO), CYCLOBENZAPRINE (FLEXERIL), METHADONE (METHADOSE),
September HCMC Toxicology Transition: Additional information and Frequently Asked Questions
 September 2016 HCMC Toxicology Transition: Additional information and Frequently Asked Questions Many clinicians have asked for more information about the Urine Drug Compliance Analysis (LAB8742) switch
September 2016 HCMC Toxicology Transition: Additional information and Frequently Asked Questions Many clinicians have asked for more information about the Urine Drug Compliance Analysis (LAB8742) switch
1/27/ New Release, Quest Diagnostics Nichols Institute, Valencia
 NEW TESTS Please Note: Not all test codes assigned to each assay are listed in the table of contents. Please refer to the complete listing on the page numbers indicated. Test Code Test Name Effective Date
NEW TESTS Please Note: Not all test codes assigned to each assay are listed in the table of contents. Please refer to the complete listing on the page numbers indicated. Test Code Test Name Effective Date
CDIA TM Marijuana Colloidal Gold Test Cassette (Saliva)
 CDIA TM Marijuana Colloidal Gold Test Cassette (Saliva) Cat. No.: DTSJZ023 Pkg. Size: Intended Use The CDIA TM Marijuana Colloidal Gold Test Cassette (Saliva) is an immunochromatography based one step
CDIA TM Marijuana Colloidal Gold Test Cassette (Saliva) Cat. No.: DTSJZ023 Pkg. Size: Intended Use The CDIA TM Marijuana Colloidal Gold Test Cassette (Saliva) is an immunochromatography based one step
Physician s Reference for Urine and Blood Drug Testing and Interpretation
 Physician s Reference for Urine and Blood Drug Testing and Interpretation DETECTIMED PANEL Urine (test code 70195) Screen Confirmation Screen Confirmation Alcohol Ethanol Amphetamines Amphetamine Methamphetamine
Physician s Reference for Urine and Blood Drug Testing and Interpretation DETECTIMED PANEL Urine (test code 70195) Screen Confirmation Screen Confirmation Alcohol Ethanol Amphetamines Amphetamine Methamphetamine
Drugs Found in Medical Examiner Cases
 s Found in Medical Examiner Cases 1,1 difluoroethane propellant in aerosols 10-Hydroxycarbazepine 11-Hydroxy Delta-9 THC 6-monoacetylmorphine 7-amino clonazepam acetaminophen acetone alpha-hydroxyalprazolam
s Found in Medical Examiner Cases 1,1 difluoroethane propellant in aerosols 10-Hydroxycarbazepine 11-Hydroxy Delta-9 THC 6-monoacetylmorphine 7-amino clonazepam acetaminophen acetone alpha-hydroxyalprazolam
Urine Drug Testing Methods 3-5
 Urine Drug Testing Methods 3-5 Type of Test Logistics Pearls Initial Screening Test: Immunoassay Confirmatory Test: Gas chromatography-mass spectrometry (GCMS) + or Liquid chromatography-mass spectrometry
Urine Drug Testing Methods 3-5 Type of Test Logistics Pearls Initial Screening Test: Immunoassay Confirmatory Test: Gas chromatography-mass spectrometry (GCMS) + or Liquid chromatography-mass spectrometry
Determination of 78 Banned or Controlled Racing Industry Drugs in Horse Urine Using SPE and LC-MS/MS
 Determination of 78 Banned or Controlled Racing Industry Drugs in Horse Urine Using SPE and LC-MS/MS UCT Part Numbers XRDAH203 Gravity Flow XtrackT DAU 200 mg, 3 ml column SPHPHO6001-5 Select ph Buffer
Determination of 78 Banned or Controlled Racing Industry Drugs in Horse Urine Using SPE and LC-MS/MS UCT Part Numbers XRDAH203 Gravity Flow XtrackT DAU 200 mg, 3 ml column SPHPHO6001-5 Select ph Buffer
3703 Camino del Rio South 100-A San Diego, CA, Phone Fax CLIA# 05D years
 Drug Adherence Assessment Report CleanAssure TM (DRIED BLOOD SPOT): Detection Range see NOTES. Prescribed Medications: NO MEDICATION LIST PROVIDED CONSISTENT RESULTS - MEDICATION DETECTED (PARENT DRUG
Drug Adherence Assessment Report CleanAssure TM (DRIED BLOOD SPOT): Detection Range see NOTES. Prescribed Medications: NO MEDICATION LIST PROVIDED CONSISTENT RESULTS - MEDICATION DETECTED (PARENT DRUG
C O P E. Milwaukee County Opioid-Related Overdose Report MILWAUKEE COMMUNITY OPIOID PREVENTION EFFORT
 Milwaukee County Opioid-Related Overdose Report 2012 2016 MILWAUKEE C O P E COMMUNITY OPIOID PREVENTION EFFORT For additional information or if you have questions about the data presented in this report,
Milwaukee County Opioid-Related Overdose Report 2012 2016 MILWAUKEE C O P E COMMUNITY OPIOID PREVENTION EFFORT For additional information or if you have questions about the data presented in this report,
Drug Adherence Assessment Report
 Prescribed Medications: Drug Adherence Assessment Report FENTANYL, OXYCODONE CONSISTENT RESULTS - REPORTED MEDICATION DETECTED (PARENT DRUG AND/OR METABOLITE) REPORTED ANTICIPATED TEST PRESCRIPTION (S)
Prescribed Medications: Drug Adherence Assessment Report FENTANYL, OXYCODONE CONSISTENT RESULTS - REPORTED MEDICATION DETECTED (PARENT DRUG AND/OR METABOLITE) REPORTED ANTICIPATED TEST PRESCRIPTION (S)
ORAL FLUID AS A CHEMICAL TEST FOR THE DRE PROGRAM : HISTORY, THE FUTURE, AND PRACTICAL CONSIDERATIONS
 ORAL FLUID AS A CHEMICAL TEST FOR THE DRE PROGRAM : HISTORY, THE FUTURE, AND PRACTICAL CONSIDERATIONS Barry K Logan PhD, DABFT National Director of Forensic Services, NMS Labs Willow Grove PA Disclaimer
ORAL FLUID AS A CHEMICAL TEST FOR THE DRE PROGRAM : HISTORY, THE FUTURE, AND PRACTICAL CONSIDERATIONS Barry K Logan PhD, DABFT National Director of Forensic Services, NMS Labs Willow Grove PA Disclaimer
Bree Collaborative AMDG Opioid Prescribing Guidelines Workgroup. Opioid Prescribing Metrics - DRAFT
 Bree Collaborative AMDG Opioid Prescribing Guidelines Workgroup Opioid Prescribing Metrics - DRAFT Definitions: Days Supply: The total of all opioid prescriptions dispensed during the calendar quarter
Bree Collaborative AMDG Opioid Prescribing Guidelines Workgroup Opioid Prescribing Metrics - DRAFT Definitions: Days Supply: The total of all opioid prescriptions dispensed during the calendar quarter
Dynacare Laboratories
 Dynacare Laboratories Affiliated with Froedtert & the Medical College of Wisconsin January 2015 2015 CPT Code Updates Dear Client: The American Medical Association (AMA) publishes the Current Procedural
Dynacare Laboratories Affiliated with Froedtert & the Medical College of Wisconsin January 2015 2015 CPT Code Updates Dear Client: The American Medical Association (AMA) publishes the Current Procedural
Alcohol. Ethanol Highlands Parkway, Suite 100 Smyrna, GA 30082
 Alcohol The alcohol of interest is ethanol. Ethanol has a sedative effect in the brain. Ethanol intoxication symptoms include blurred vision, slurred speech, poor coordination and difficulty thinking depending
Alcohol The alcohol of interest is ethanol. Ethanol has a sedative effect in the brain. Ethanol intoxication symptoms include blurred vision, slurred speech, poor coordination and difficulty thinking depending
ChemaTox Blood Drug Testing Matrix (Updated May 24th, 2016)
 ChemaTox Blood Drug Testing Matrix (Updated May 24th, 2016) Email: DREtox@chematox.com; Phone: (303)440-4500 Always identify any medications/drugs the suspect states they have taken, or that you suspect
ChemaTox Blood Drug Testing Matrix (Updated May 24th, 2016) Email: DREtox@chematox.com; Phone: (303)440-4500 Always identify any medications/drugs the suspect states they have taken, or that you suspect
LCMS-8050 Drugs of Abuse: 113 Analytes with Polarity Switching
 Liquid Chromatography Mass Spectrometry SSI-LCMS-8 LCMS-8 Drugs of Abuse: Analytes with Polarity Switching LCMS-8 Summary Seventy six analytes and their internal standards are described below. Multiple
Liquid Chromatography Mass Spectrometry SSI-LCMS-8 LCMS-8 Drugs of Abuse: Analytes with Polarity Switching LCMS-8 Summary Seventy six analytes and their internal standards are described below. Multiple
Pain Management and Compliance Toxicology. Greg Jellick, MSFS, D-ABFT-FT Technical Director Quality Toxicology San Antonio, TX
 Pain Management and Compliance Toxicology Greg Jellick, MSFS, D-ABFT-FT Technical Director Quality Toxicology San Antonio, TX Prescription Drug Abuse: A National Problem Prescription drug abuse is a growing
Pain Management and Compliance Toxicology Greg Jellick, MSFS, D-ABFT-FT Technical Director Quality Toxicology San Antonio, TX Prescription Drug Abuse: A National Problem Prescription drug abuse is a growing
Toxicological Investigation of Drug Impaired Driving Toxicology Laboratory Survey
 Toxicological Investigation of Drug Impaired Driving Toxicology Laboratory Survey Kayla J. Lowrie, MS, Jennifer Turri, BS, Jill Yeakel, MSFS, Barry K. Logan, PhD, DABFT, Arcadia University, 450 S. Easton
Toxicological Investigation of Drug Impaired Driving Toxicology Laboratory Survey Kayla J. Lowrie, MS, Jennifer Turri, BS, Jill Yeakel, MSFS, Barry K. Logan, PhD, DABFT, Arcadia University, 450 S. Easton
ToxCup Drug Screen Cup Step-by Step Instructions
 ToxCup Drug Screen Cup Step-by Step Instructions This is a preliminary screening test that detects drug-of-abuse in urine at specified detection levels. To confirm preliminary positive results, a more
ToxCup Drug Screen Cup Step-by Step Instructions This is a preliminary screening test that detects drug-of-abuse in urine at specified detection levels. To confirm preliminary positive results, a more
Effective Date: Approved by: Laboratory Executive Director, Ed Hughes (electronic signature)
 1 Policy #: 803 (PLH-803-02) Effective Date: NA Reviewed Date: 4/11/2008 Subject: URINE DRUG SCREENS Approved by: Laboratory Executive Director, Ed Hughes (electronic signature) Approved by: Laboratory
1 Policy #: 803 (PLH-803-02) Effective Date: NA Reviewed Date: 4/11/2008 Subject: URINE DRUG SCREENS Approved by: Laboratory Executive Director, Ed Hughes (electronic signature) Approved by: Laboratory
ALAMEDA COUNTY BEHAVIORAL HEALTH CARE SERVICES MEDICATION FORMULARY
 ANTIDEPRESSANTS Serotonin Selective Reuptake Inhibitors citalopram 10, 20, 40 mg, 10 mg/5cc $ 0.40 No escitalopram 10, 20 mg $ 2.60 Yes fluoxetine 10, 20 mg, 20 mg/5 ml $ 0.40 Yes fluvoxamine 25, 50, 100
ANTIDEPRESSANTS Serotonin Selective Reuptake Inhibitors citalopram 10, 20, 40 mg, 10 mg/5cc $ 0.40 No escitalopram 10, 20 mg $ 2.60 Yes fluoxetine 10, 20 mg, 20 mg/5 ml $ 0.40 Yes fluvoxamine 25, 50, 100
2015 Annual Physician Notice
 0 Annual Physician Notice The Office of Inspector General (OIG) recommends clinical laboratories send notices to physicians and other providers who use their services, at least once a year, to inform the
0 Annual Physician Notice The Office of Inspector General (OIG) recommends clinical laboratories send notices to physicians and other providers who use their services, at least once a year, to inform the
VIRGINIA DEPARTMENT OF HEALTH OFFICE OF THE CHIEF MEDICAL EXAMINER
 VIRGINIA DEPARTMENT OF HEALTH OFFICE OF THE CHIEF MEDICAL EXAMINER To promote and protect the health of all Virginians Fatal Drug Overdose Quarterly Report Edition 216.3 Publication Date: January 217 METHODS,
VIRGINIA DEPARTMENT OF HEALTH OFFICE OF THE CHIEF MEDICAL EXAMINER To promote and protect the health of all Virginians Fatal Drug Overdose Quarterly Report Edition 216.3 Publication Date: January 217 METHODS,
Prepared by Date Adopted Supersedes Procedure # Review Date Revision Date Signature. Distributed to # of Copies Distributed to # of Copies
 Procedure: Status DS Prepared by Date Adopted Supersedes Procedure # Review Date Revision Date Signature Distributed to # of Copies Distributed to # of Copies PRINCIPLE: The Status DS 10 Panel (MET/OPI/COC/THC/PCP/BZO/BAR/MTD/TCA/AMP)
Procedure: Status DS Prepared by Date Adopted Supersedes Procedure # Review Date Revision Date Signature Distributed to # of Copies Distributed to # of Copies PRINCIPLE: The Status DS 10 Panel (MET/OPI/COC/THC/PCP/BZO/BAR/MTD/TCA/AMP)
A Rapid Method for Detection of Drugs of Abuse in Blood Samples Using the Thermal Separation Probe and the 5975T LTM GC/MS
 A Rapid Method for Detection of Drugs of Abuse in Blood Samples Using the Thermal Separation Probe and the 5975T LTM GC/MS Application Note Forensics Authors Suli Zhao Agilent Technologies, Inc. Shanghai
A Rapid Method for Detection of Drugs of Abuse in Blood Samples Using the Thermal Separation Probe and the 5975T LTM GC/MS Application Note Forensics Authors Suli Zhao Agilent Technologies, Inc. Shanghai
Validation of a Benzodiazepine and Z-Drug Method Using an Agilent 6430 LC/MS/MS
 Validation of a Benzodiazepine and Z-Drug Method Using an Agilent 6430 LC/MS/MS Application Note Forensics Authors Jason Hudson, Ph.D., James Hutchings, Ph.D., and Rebecca Wagner, Ph.D. Virginia Department
Validation of a Benzodiazepine and Z-Drug Method Using an Agilent 6430 LC/MS/MS Application Note Forensics Authors Jason Hudson, Ph.D., James Hutchings, Ph.D., and Rebecca Wagner, Ph.D. Virginia Department
Fully Automated Online Sample Preparation and LC-MS/MS Analysis of Drugs of Abuse in Oral Fluids
 PO-CON1753E Fully Automated Online Sample Preparation and LC-MS/MS Analysis of Drugs of Abuse in Oral Fluids ASMS 2017 TP-442 Joshua F. Emory 1, Nathan DeFreitas 2, Michael Roberts 1, Manoj Tyagi 2, M.
PO-CON1753E Fully Automated Online Sample Preparation and LC-MS/MS Analysis of Drugs of Abuse in Oral Fluids ASMS 2017 TP-442 Joshua F. Emory 1, Nathan DeFreitas 2, Michael Roberts 1, Manoj Tyagi 2, M.
Laboratory Testing to Support Pain Management: Methods, Concepts and Case Studies
 Laboratory Testing to Support Pain Management: Methods, Concepts and Case Studies Frederick G. Strathmann, PhD, DABCC, (CC,TC) Medical Director, Toxicology Associate Scientific Director of MS ARUP Laboratories
Laboratory Testing to Support Pain Management: Methods, Concepts and Case Studies Frederick G. Strathmann, PhD, DABCC, (CC,TC) Medical Director, Toxicology Associate Scientific Director of MS ARUP Laboratories
PROFILE -V MEDTOXScan Drugs of Abuse Test System INSERT
 PROFILE -V MEDTOXScan Drugs of Abuse Test System INSERT 1. INTENDED USE The PROFILE -V MEDTOXScan Drugs of Abuse Test System consists of the PROFILE -V MEDTOXScan Test Devices and the MEDTOXScan Reader.
PROFILE -V MEDTOXScan Drugs of Abuse Test System INSERT 1. INTENDED USE The PROFILE -V MEDTOXScan Drugs of Abuse Test System consists of the PROFILE -V MEDTOXScan Test Devices and the MEDTOXScan Reader.
DRUGS OF ABUSE TEST CUPS 14-DRUG PANEL WITH ADULTERANTS Step by Step Instructions
 DRUGS OF ABUSE TEST CUPS 14-DRUG PANEL WITH ADULTERANTS Step by Step Instructions For in vitro diagnostic use The McKesson Consult Drugs of Abuse Test Cup is a screening test for the rapid qualitative
DRUGS OF ABUSE TEST CUPS 14-DRUG PANEL WITH ADULTERANTS Step by Step Instructions For in vitro diagnostic use The McKesson Consult Drugs of Abuse Test Cup is a screening test for the rapid qualitative
SUPPLEMENTARY MATERIALS. Appendix 1. Definition of Suicidal Behavior Using ICD-9 Diagnostic Codes.
 SUPPLEMENTARY MATERIALS Appendix 1. Definition of Suicidal Behavior Using ICD-9 Diagnostic Codes. For this study, suicidal behavior was defined using ICD-9 diagnostic codes and death certificates from
SUPPLEMENTARY MATERIALS Appendix 1. Definition of Suicidal Behavior Using ICD-9 Diagnostic Codes. For this study, suicidal behavior was defined using ICD-9 diagnostic codes and death certificates from
Lyndsey Knoy, D-ABFT-FT Forensic Scientist Washington State Toxicology Laboratory.
 Lyndsey Knoy, D-ABFT-FT Forensic Scientist Washington State Toxicology Laboratory Lyndsey.Knoy@wsp.wa.gov Forensic Toxicology is an interdisciplinary science that analyzes blood, fluid and/or tissues for
Lyndsey Knoy, D-ABFT-FT Forensic Scientist Washington State Toxicology Laboratory Lyndsey.Knoy@wsp.wa.gov Forensic Toxicology is an interdisciplinary science that analyzes blood, fluid and/or tissues for
PROFILE -V MEDTOXScan Drugs of Abuse Test System PACKAGE INSERT
 PROFILE -V MEDTOXScan Drugs of Abuse Test System PACKAGE INSERT 1. INTENDED USE The PROFILE -V MEDTOXScan Drugs of Abuse Test System consists of the PROFILE -V MEDTOXScan Test Devices and the MEDTOXScan
PROFILE -V MEDTOXScan Drugs of Abuse Test System PACKAGE INSERT 1. INTENDED USE The PROFILE -V MEDTOXScan Drugs of Abuse Test System consists of the PROFILE -V MEDTOXScan Test Devices and the MEDTOXScan
Rapid Drug Test Cup Step-by Step Instructions
 Rapid Drug Test Cup The CLIAwaived Inc. Rapid Drug Test Cup is an in vitro screening test for the rapid detection of multiple drugs in human urine at or above the following cutoff concentration: THC 11-nor-Δ9-Tetrahydrocannabinol-9-carboxylic
Rapid Drug Test Cup The CLIAwaived Inc. Rapid Drug Test Cup is an in vitro screening test for the rapid detection of multiple drugs in human urine at or above the following cutoff concentration: THC 11-nor-Δ9-Tetrahydrocannabinol-9-carboxylic
E XCEL LENCE JUST GOT BETTER UCT FORENSICS CLEAN SCREEN XCEL SPE COLUMNS
 FORENSICS UCT E XCEL LENCE JUST GOT BETTER CLEAN SCREEN XCEL SPE COLUMNS CLEAN SCREEN XCEL COLUMNS EXTRACTION OF BASIC DRUGS AND METABOLITES FROM URINE/ BLOOD USING SPE CARTRIDGES 130mg Clean Screen Xcel
FORENSICS UCT E XCEL LENCE JUST GOT BETTER CLEAN SCREEN XCEL SPE COLUMNS CLEAN SCREEN XCEL COLUMNS EXTRACTION OF BASIC DRUGS AND METABOLITES FROM URINE/ BLOOD USING SPE CARTRIDGES 130mg Clean Screen Xcel
Evaluation of the Impact of Expanding ELISA Screening in DUID Investigations. Aileen Lu*, Karen S. Scott, Aya Chan-Hosokawa, and Barry K.
 Evaluation of the Impact of Expanding ELISA Screening in DUID Investigations Aileen Lu*, Karen S. Scott, Aya Chan-Hosokawa, and Barry K. Logan FSF Emerging Forensic Scientist Award Oral Presentation Disclosure
Evaluation of the Impact of Expanding ELISA Screening in DUID Investigations Aileen Lu*, Karen S. Scott, Aya Chan-Hosokawa, and Barry K. Logan FSF Emerging Forensic Scientist Award Oral Presentation Disclosure
2012 DRUG RELATED DEATH STATISTICS JANUARY 1, 2012 THROUGH DECEMBER 31, 2012
 JANUARY 1, 2012 THROUGH DECEMBER 31, 2012 All deaths counted in the column labeled ACCIDENTAL OVERDOSE were the direct result of the drugs ingested by the decedent. The drugs noted as being present came
JANUARY 1, 2012 THROUGH DECEMBER 31, 2012 All deaths counted in the column labeled ACCIDENTAL OVERDOSE were the direct result of the drugs ingested by the decedent. The drugs noted as being present came
Medicines Formulary BNF Section 4 Central Nervous System
 Medicines BNF Section 4 4.1 Hypnotics and anxiolytics Chloral Hydrate 500mg/5ml Solution Clomethiazole 192mg Capsules Lormetazepam Tablets Melatonin Capsules Nitrazepam Suspension Nitrazepam Tablets Temazepam
Medicines BNF Section 4 4.1 Hypnotics and anxiolytics Chloral Hydrate 500mg/5ml Solution Clomethiazole 192mg Capsules Lormetazepam Tablets Melatonin Capsules Nitrazepam Suspension Nitrazepam Tablets Temazepam
10/16/2017. Objectives. Drug Testing Interpretation in Addiction Care. Background. Which is Nonadherent?
 Objectives Drug Testing Interpretation in Addiction Care Brandi Puet, Pharm.D. Describe the difference between immunoassay and confirmatory testing. List explanations for unexpected negative or positive
Objectives Drug Testing Interpretation in Addiction Care Brandi Puet, Pharm.D. Describe the difference between immunoassay and confirmatory testing. List explanations for unexpected negative or positive
PROJECT ECHO OPIOID USE DISORDER IN PREGNANT WOMEN
 PROJECT ECHO OPIOID USE DISORDER IN PREGNANT WOMEN 1 OPIOID USE DISORDER IN WOMEN Recognition & Prevention Date: August 14 th, Time: 8 am Presenters: Deepa Nagar MD, Andria Peterson PharmD Maternal Treatment
PROJECT ECHO OPIOID USE DISORDER IN PREGNANT WOMEN 1 OPIOID USE DISORDER IN WOMEN Recognition & Prevention Date: August 14 th, Time: 8 am Presenters: Deepa Nagar MD, Andria Peterson PharmD Maternal Treatment
Measure #238 (NQF 0022): Use of High-Risk Medications in the Elderly National Quality Strategy Domain: Patient Safety
 Measure #238 (NQF 0022): Use of High-Risk Medications in the Elderly National Quality Strategy Domain: Patient Safety 2017 OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY MEASURE TYPE: Process DESCRIPTION:
Measure #238 (NQF 0022): Use of High-Risk Medications in the Elderly National Quality Strategy Domain: Patient Safety 2017 OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY MEASURE TYPE: Process DESCRIPTION:
Using Liquid Chromatography Tandem Mass Spectrometry Urine Drug Testing to Identify Licit and Illicit Drug-Use in a Community-based Patient Population
 Using Liquid Chromatography Tandem Mass Spectrometry Urine Drug Testing to Identify Licit and Illicit Drug-Use in a Community-based Patient Population Adam S. Ptolemy 1, Colleen Murray 2, Edward Dunn 3,
Using Liquid Chromatography Tandem Mass Spectrometry Urine Drug Testing to Identify Licit and Illicit Drug-Use in a Community-based Patient Population Adam S. Ptolemy 1, Colleen Murray 2, Edward Dunn 3,
The Drug Testing Process. Employer or Practice
 Disclosures Clinical Professor, Jefferson Medical College BOD MROCC [Medical Review Officer Certification Council] BOD National Sleep Foundation BOD POEMS [Pennsylvania Occupational & Environmental Medicine
Disclosures Clinical Professor, Jefferson Medical College BOD MROCC [Medical Review Officer Certification Council] BOD National Sleep Foundation BOD POEMS [Pennsylvania Occupational & Environmental Medicine
Oregon Health & Science University Department of Pathology, Division of Laboratory Medicine Core Laboratory
 Oregon Health & Science University Department of Pathology, Division of Laboratory Medicine Core Laboratory Summary of s that may be detected by OHSU Drugs of Abuse Screening Methods Amphetamine/Methamphetamine
Oregon Health & Science University Department of Pathology, Division of Laboratory Medicine Core Laboratory Summary of s that may be detected by OHSU Drugs of Abuse Screening Methods Amphetamine/Methamphetamine
Measure #238 (NQF 0022): Use of High-Risk Medications in the Elderly National Quality Strategy Domain: Patient Safety
 Measure #238 (NQF 0022): Use of High-Risk Medications in the Elderly National Quality Strategy Domain: Patient Safety 2015 PHYSICIAN QUALITY REPTING OPTIONS F INDIVIDUAL MEASURES REGISTRY ONLY DESCRIPTION:
Measure #238 (NQF 0022): Use of High-Risk Medications in the Elderly National Quality Strategy Domain: Patient Safety 2015 PHYSICIAN QUALITY REPTING OPTIONS F INDIVIDUAL MEASURES REGISTRY ONLY DESCRIPTION:
Intercept i2he Methamphetamine Oral Fluid Assay
 Intercept i2he Methamphetamine FOR FORENSIC USE ONLY For Use with Intercept i2he Oral Fluid Collection Device 1001-0386 (65 ml Kit) INTENDED USE The Intercept i2he Methamphetamine is intended for use in
Intercept i2he Methamphetamine FOR FORENSIC USE ONLY For Use with Intercept i2he Oral Fluid Collection Device 1001-0386 (65 ml Kit) INTENDED USE The Intercept i2he Methamphetamine is intended for use in
Supplementary Online Content
 Supplementary Online Content Raebel MA, Newcomer SR, Reifler LM, et al. Chronic use of opioid medications before and after bariatric surgery. JAMA. doi:10.1001/jama.2013.278344 efigure. Opioid Dispensings
Supplementary Online Content Raebel MA, Newcomer SR, Reifler LM, et al. Chronic use of opioid medications before and after bariatric surgery. JAMA. doi:10.1001/jama.2013.278344 efigure. Opioid Dispensings
Centre of Forensic Sciences
 Introduction Centre of Forensic Sciences Investigators and Submitters Technical Information Sheets Toxicology The performs analyses on biological samples (e.g., blood, urine, liver) to determine the absence/presence/concentration(s)
Introduction Centre of Forensic Sciences Investigators and Submitters Technical Information Sheets Toxicology The performs analyses on biological samples (e.g., blood, urine, liver) to determine the absence/presence/concentration(s)
A Simple and Accurate Method for the Rapid Quantitation of Drugs of Abuse in Urine Using Liquid Chromatography
 Application Note LCMS-109 A Simple and Accurate Method for the Rapid Quantitation of Drugs of Abuse in Urine Using Liquid Chromatography Time of Flight (LC-TOF) Mass Spectrometry Introduction Many clinical
Application Note LCMS-109 A Simple and Accurate Method for the Rapid Quantitation of Drugs of Abuse in Urine Using Liquid Chromatography Time of Flight (LC-TOF) Mass Spectrometry Introduction Many clinical
Multi-Drug Screen Test
 Multi-Drug Screen Test Package Insert for OTC/CLIA-Waived- and Professional Use For in vitro diagnostic use only The Multi-Drug Screen Test offers a variety of solutions for fast and reliable drug testing
Multi-Drug Screen Test Package Insert for OTC/CLIA-Waived- and Professional Use For in vitro diagnostic use only The Multi-Drug Screen Test offers a variety of solutions for fast and reliable drug testing
EDUCATIONAL COMMENTARY rd TEST EVENT Chemistry Urine Drug Testing
 EDUCATIONAL COMMENTARY 2003 3 rd TEST EVENT Chemistry Urine Drug Testing Educational commentary is provided through our affiliation with the American Society for Clinical Pathology (ASCP). To obtain FREE
EDUCATIONAL COMMENTARY 2003 3 rd TEST EVENT Chemistry Urine Drug Testing Educational commentary is provided through our affiliation with the American Society for Clinical Pathology (ASCP). To obtain FREE
Frequently Asked Questions: Opiate Dependency and Methadone Maintenance Treatment program follow-up
 Frequently Asked Questions: Opiate Dependency and Methadone Maintenance Treatment program follow-up Dr. Bhushan M. Kapur Associate Professor Department of Laboratory Medicine and Pathobiology, Faculty
Frequently Asked Questions: Opiate Dependency and Methadone Maintenance Treatment program follow-up Dr. Bhushan M. Kapur Associate Professor Department of Laboratory Medicine and Pathobiology, Faculty
Oxycontin The Concept of a "Ghost Pill" and the Postmortem Tissue Distribution of Oxycodone in 36 Cases*
 Journal of Analytical Toxicology, Voi. 26, October 2002 The Concept of a "Ghost Pill" and the Postmortem Tissue Distribution of Oxycodone in 36 Cases* Daniel T. Anderson, Kristina L. Fritz, and Joseph
Journal of Analytical Toxicology, Voi. 26, October 2002 The Concept of a "Ghost Pill" and the Postmortem Tissue Distribution of Oxycodone in 36 Cases* Daniel T. Anderson, Kristina L. Fritz, and Joseph
METHAMPHETAMINE & AMPHETAMINE
 METHAMPHETAMINE & AMPHETAMINE Enantiomers from Urine Rapid and Accurate Chiral Separation Polysaccharide Chiral Chiral LC LC Columns and SFC PHEN-RUO-49 Revision: Lux 3 µm AMP Column High Efficiency LC/MS/MS
METHAMPHETAMINE & AMPHETAMINE Enantiomers from Urine Rapid and Accurate Chiral Separation Polysaccharide Chiral Chiral LC LC Columns and SFC PHEN-RUO-49 Revision: Lux 3 µm AMP Column High Efficiency LC/MS/MS
Linking Opioid Treatment in Primary Care. Roxanne Lewin M.D.
 Roxanne Lewin M.D. The Facts Fewer than 10 percent of individuals with an alcohol use disorder and only about 20 percent of individuals with an opioid use disorder receive specialty treatment. Many individuals
Roxanne Lewin M.D. The Facts Fewer than 10 percent of individuals with an alcohol use disorder and only about 20 percent of individuals with an opioid use disorder receive specialty treatment. Many individuals
Test Definition: PCDSO Pain Clinic Drug Screen, Urine
 Reporting Title: Pain Clinic Drug Screen, U Performing Location: Rochester Specimen Requirements: Collection Container/Tube: Plastic urine container Submission Container/Tube: Plastic, 60-mL urine bottle
Reporting Title: Pain Clinic Drug Screen, U Performing Location: Rochester Specimen Requirements: Collection Container/Tube: Plastic urine container Submission Container/Tube: Plastic, 60-mL urine bottle
AppNote 11/2006. Performance Evaluation of a Thermal Desorption System for Detection of Basic Drugs in Forensic Samples by GC/MS KEYWORDS ABSTRACT
 AppNote 11/2006 Performance Evaluation of a Thermal Desorption System for Detection of Basic Drugs in Forensic Samples by GC/MS Joseph A. Crifasi, Michael F. Bruder, Christopher W. Long Saint Louis University
AppNote 11/2006 Performance Evaluation of a Thermal Desorption System for Detection of Basic Drugs in Forensic Samples by GC/MS Joseph A. Crifasi, Michael F. Bruder, Christopher W. Long Saint Louis University
Test Definition: PDSOX Pain Clinic Drug Screen, Chain of Custody, Urine
 Reporting Title: Pain Clinic Drug Screen, CoC, U Performing Location: Rochester Specimen Requirements: Container/Tube: Chain-of-Custody Kit (Supply T282) containing the specimen containers, seals, and
Reporting Title: Pain Clinic Drug Screen, CoC, U Performing Location: Rochester Specimen Requirements: Container/Tube: Chain-of-Custody Kit (Supply T282) containing the specimen containers, seals, and
Laboratory Service Report
 4 05/25/19 Client C702884-DLP ROCHESTER Amphetamines, Confirmation Positive Confirmed POSITIVE by LC-S/S for the following: Amphetamine = 52 ethamphetamine = 124 ethamphetamine exists in the d- and l-isomeric
4 05/25/19 Client C702884-DLP ROCHESTER Amphetamines, Confirmation Positive Confirmed POSITIVE by LC-S/S for the following: Amphetamine = 52 ethamphetamine = 124 ethamphetamine exists in the d- and l-isomeric
Intercept i2he Amphetamine Oral Fluid Assay
 FOR FORENSIC USE ONLY For Use with Oral Fluid Collection Device 1001-0385 (65 ml Kit) INTENDED USE The is intended for use in the qualitative determination of amphetamine in human oral fluid at a cutoff
FOR FORENSIC USE ONLY For Use with Oral Fluid Collection Device 1001-0385 (65 ml Kit) INTENDED USE The is intended for use in the qualitative determination of amphetamine in human oral fluid at a cutoff
OneStep Methadone Urine RapiCard InstaTest. Cat. #
 CORTEZ DIAGNOSTICS, INC. 23961 Craftsman Road, Suite D/E/F Calabasas, CA 91302 USA Tel: (818) 591-3030 Fax: (818) 591-8383 E-mail: onestep@rapidtest.com Web site: www.rapidtest.com See external label Σ=25
CORTEZ DIAGNOSTICS, INC. 23961 Craftsman Road, Suite D/E/F Calabasas, CA 91302 USA Tel: (818) 591-3030 Fax: (818) 591-8383 E-mail: onestep@rapidtest.com Web site: www.rapidtest.com See external label Σ=25
Potentially Inappropriate Medication Use in Older Adults 2015 Latest Research
 Home Resources Potentially Inappropriate Medication Use in Older Adults 2015 Resources Potentially Inappropriate Medication Use in Older Adults 2015 Latest Research Drugs and Categories of Drugs What these
Home Resources Potentially Inappropriate Medication Use in Older Adults 2015 Resources Potentially Inappropriate Medication Use in Older Adults 2015 Latest Research Drugs and Categories of Drugs What these
New Test Announcements
 27027 Tourney Road Valencia, CA 91355 800 421 7110 www.specialtylabs.com New Test Announcements Dear Colleague: We are pleased to announce many new test offerings this month. January 25, 2007 In oncology,
27027 Tourney Road Valencia, CA 91355 800 421 7110 www.specialtylabs.com New Test Announcements Dear Colleague: We are pleased to announce many new test offerings this month. January 25, 2007 In oncology,
Palliative Care Drug Plan (Plan P) Formulary List of drugs PharmaCare covers
 Palliative Care Drug Plan (Plan P) Formulary List of drugs PharmaCare covers This formulary is current as of February 11, 2010. Important Notes: Pharmacists must submit a claim on PharmaNet at the time
Palliative Care Drug Plan (Plan P) Formulary List of drugs PharmaCare covers This formulary is current as of February 11, 2010. Important Notes: Pharmacists must submit a claim on PharmaNet at the time
Intercept i2he Opiate Oral Fluid Assay
 FOR FORENSIC USE ONLY For Use with Oral Fluid Collection Device 1001-0387 (65 ml Kit) INTENDED USE The is intended for use in the qualitative determination of Opiate in human oral fluid at a cutoff concentration
FOR FORENSIC USE ONLY For Use with Oral Fluid Collection Device 1001-0387 (65 ml Kit) INTENDED USE The is intended for use in the qualitative determination of Opiate in human oral fluid at a cutoff concentration
Measure #238 (NQF 0022): Use of High-Risk Medications in the Elderly National Quality Strategy Domain: Patient Safety
 Measure #238 (NQF 0022): Use of High-Risk Medications in the Elderly National Quality Strategy Domain: Patient Safety 2016 PHYSICIAN QUALITY REPORTING OPTIONS FOR INDIVIDUAL MEASURES REGISTRY ONLY DESCRIPTION:
Measure #238 (NQF 0022): Use of High-Risk Medications in the Elderly National Quality Strategy Domain: Patient Safety 2016 PHYSICIAN QUALITY REPORTING OPTIONS FOR INDIVIDUAL MEASURES REGISTRY ONLY DESCRIPTION:
Automated ion trap LC-MS screening for xenobiotics in vitreous humour
 Toxichem Krimtech 2015;82(Special Issue):168 Automated ion trap LC-MS screening for xenobiotics in vitreous humour Jürgen Kempf*,Christine Stronczek, Laura M. Huppertz, Volker Auwärter, Susanne Vogt University
Toxichem Krimtech 2015;82(Special Issue):168 Automated ion trap LC-MS screening for xenobiotics in vitreous humour Jürgen Kempf*,Christine Stronczek, Laura M. Huppertz, Volker Auwärter, Susanne Vogt University
nextgen precision Test Report
 nextgen precision TM Test Report SUMMARY OF PRECISION S NEXTGEN TEST REPORT In an effort to provide as much clinical insight as possible to our providers, we created our comprehensive NextGen Precision
nextgen precision TM Test Report SUMMARY OF PRECISION S NEXTGEN TEST REPORT In an effort to provide as much clinical insight as possible to our providers, we created our comprehensive NextGen Precision
Cross-reactivity reactivity in EMIT
 Blood Drug Analysis at the State Bureau of Investigation Crime Laboratory Richard W. Waggoner, Jr. Sample Requirements 2-10 ml gray top vacutainers At least 10 ml of urine sample for drug facilitated sexual
Blood Drug Analysis at the State Bureau of Investigation Crime Laboratory Richard W. Waggoner, Jr. Sample Requirements 2-10 ml gray top vacutainers At least 10 ml of urine sample for drug facilitated sexual
CATECHOLAMINE, FREE, RANDOM URINE
 Lab Dept: Test Name: Urine/Stool CATECHOLAMINE, FREE, RANDOM URINE General Information Lab Order Codes: Synonyms: CPT Codes: Test Includes: CATCR Catecholamine Fractionation, Free, Random Urine 82384 Catecholamines;
Lab Dept: Test Name: Urine/Stool CATECHOLAMINE, FREE, RANDOM URINE General Information Lab Order Codes: Synonyms: CPT Codes: Test Includes: CATCR Catecholamine Fractionation, Free, Random Urine 82384 Catecholamines;
Urine Opioid Dependency Panel (UODP) 1
 Drug Testing Panels Urine Opioid Dependency Panel (UODP) 1 Amphetamines Amphetamine Methamphetamine Methylenedioxyamphetamine Adderall, Dexedrine, Vyvanse, Lisdexamfetamine (Speed, Bennies, Crystal Meth,
Drug Testing Panels Urine Opioid Dependency Panel (UODP) 1 Amphetamines Amphetamine Methamphetamine Methylenedioxyamphetamine Adderall, Dexedrine, Vyvanse, Lisdexamfetamine (Speed, Bennies, Crystal Meth,
Quality ID #238 (NQF 0022): Use of High-Risk Medications in the Elderly National Quality Strategy Domain: Patient Safety
 Quality ID #238 (NQF 0022): Use of High-Risk Medications in the Elderly National Quality Strategy Domain: Patient Safety 2018 OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY MEASURE TYPE: Process DESCRIPTION:
Quality ID #238 (NQF 0022): Use of High-Risk Medications in the Elderly National Quality Strategy Domain: Patient Safety 2018 OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY MEASURE TYPE: Process DESCRIPTION:
Validation of an Automated Method to Remove
 Shahana Wahab Huq 1, Richard Thomas 2, Agnes Cua 2, Seyed Sadjadi 1 1 Phenomenex, 411 Madrid Avenue, Torrance, CA 951 2 Precision Toxicology, 33 Bunker Hill Street, San Diego, CA 9219 Validation of an
Shahana Wahab Huq 1, Richard Thomas 2, Agnes Cua 2, Seyed Sadjadi 1 1 Phenomenex, 411 Madrid Avenue, Torrance, CA 951 2 Precision Toxicology, 33 Bunker Hill Street, San Diego, CA 9219 Validation of an
CLIAwaived, Inc. CLIA X Multiple Drug Cup Test
 CLIAwaived CLIAwaived, Inc. CLIA X Multiple Drug Cup Test URINE SCREENING TEST /HOME TESTING KITS Results in 5 Minutes CLIA-Waived Instructions IVD 2 INTENDED USE This CLIA-X Multiple Drug Cup Test - is
CLIAwaived CLIAwaived, Inc. CLIA X Multiple Drug Cup Test URINE SCREENING TEST /HOME TESTING KITS Results in 5 Minutes CLIA-Waived Instructions IVD 2 INTENDED USE This CLIA-X Multiple Drug Cup Test - is
Executive Report was an active year for the Regional Center for Poison Control and Prevention Serving Massachusetts and Rhode Island.
 Executive Report 2011 was an active year for the Regional Center for Poison Control and Prevention Serving Massachusetts and Rhode Island. In 2011, the Poison Center managed 52,581 poison exposure and
Executive Report 2011 was an active year for the Regional Center for Poison Control and Prevention Serving Massachusetts and Rhode Island. In 2011, the Poison Center managed 52,581 poison exposure and
Pain Medication Management Program Monitors Patient Compliance
 PeaceHealth Laboratories UPDATE 2014/15 Edition Pain Medication Management Program Monitors Patient Compliance BENEFITS Monitors analgesic medication adherence to ensure patient safety and protect your
PeaceHealth Laboratories UPDATE 2014/15 Edition Pain Medication Management Program Monitors Patient Compliance BENEFITS Monitors analgesic medication adherence to ensure patient safety and protect your
cause of death; death certificates; International Classification of Diseases; poisoning; SuperMICAR
 American Journal of Epidemiology The Author 1. Published by Oxford University Press on behalf of the Johns Hopkins Bloomberg School of Public Health. All rights reserved. For permissions, please e-mail:
American Journal of Epidemiology The Author 1. Published by Oxford University Press on behalf of the Johns Hopkins Bloomberg School of Public Health. All rights reserved. For permissions, please e-mail:
Montgomery County Poisoning Death Review:
 Wright State University CORE Scholar Unintentional Prescription Drug Poisoning Project Center for Interventions, Treatment and Addictions Research 5-6-214 Montgomery County Poisoning Death Review: 21-213
Wright State University CORE Scholar Unintentional Prescription Drug Poisoning Project Center for Interventions, Treatment and Addictions Research 5-6-214 Montgomery County Poisoning Death Review: 21-213
C O P E. Milwaukee County Opioid Related Overdose Report MILWAUKEE COMMUNITY OPIOID PREVENTION EFFORT
 Milwaukee County Opioid Related Overdose Report 2012 2016 MILWAUKEE C O P E COMMUNITY OPIOID PREVENTION EFFORT For addi onal informa on or if you have ques ons about the data presented in this report,
Milwaukee County Opioid Related Overdose Report 2012 2016 MILWAUKEE C O P E COMMUNITY OPIOID PREVENTION EFFORT For addi onal informa on or if you have ques ons about the data presented in this report,
Pinellas County Forensic Laboratory. Annual Report 2006
 Pinellas County Forensic Laboratory Annual Report 26 Published April 27 Analytical Services Provided Post Mortem/Medical Examiner Toxicology Seized Drug (Controlled Substances) Analysis Fire Debris and
Pinellas County Forensic Laboratory Annual Report 26 Published April 27 Analytical Services Provided Post Mortem/Medical Examiner Toxicology Seized Drug (Controlled Substances) Analysis Fire Debris and
Rapid screening and semi-quantitative analysis for forensic drugs in blood using liquid chromatography triple quadrupole mass spectrometry
 PO-CON E Rapid screening and semi-quantitative analysis for forensic drugs in blood using liquid chromatography triple quadrupole mass spectrometry ASMS 2013 WP-121 Thomas Hayes 1, Keiko Kudo 2, Toshikazu
PO-CON E Rapid screening and semi-quantitative analysis for forensic drugs in blood using liquid chromatography triple quadrupole mass spectrometry ASMS 2013 WP-121 Thomas Hayes 1, Keiko Kudo 2, Toshikazu
Schedule of Accreditation issued by United Kingdom Accreditation Service 2 Pine Trees, Chertsey Lane, Staines-upon-Thames, TW18 3HR, UK
 Schedule of ccreditation United Kingdom ccreditation Service 2 Pine Trees, Chertsey Lane, Staines-upon-Thames, TW18 3HR, UK ccredited to Laboratory locations: Gavenny Court Brecon Road bergavenny Monmouthshire
Schedule of ccreditation United Kingdom ccreditation Service 2 Pine Trees, Chertsey Lane, Staines-upon-Thames, TW18 3HR, UK ccredited to Laboratory locations: Gavenny Court Brecon Road bergavenny Monmouthshire
Jonathan Rochon, Pier-Luc Plante, Serge Auger, Réal Paquin, Jacques Corbeil, Pierre Picard
 Dried Blood Spots in Tips Targeted Drug Screening in 9 Seconds per Sample Using Laser Diode Thermal Desorption Mass Spectrometry (LDTD-MS/MS) Jonathan Rochon, Pier-Luc Plante, Serge Auger, Réal Paquin,
Dried Blood Spots in Tips Targeted Drug Screening in 9 Seconds per Sample Using Laser Diode Thermal Desorption Mass Spectrometry (LDTD-MS/MS) Jonathan Rochon, Pier-Luc Plante, Serge Auger, Réal Paquin,
Validation of a New Homogeneous Immunoassay for the Detection of Carisoprodol in Urine
 Validation of a New Homogeneous Immunoassay for the Detection of Carisoprodol in Urine Guohong Wang*, Kim Huynh, Rekha Barhate, Warren Rodrigues, Christine Moore, Cynthia Coulter, Michael Vincent, and
Validation of a New Homogeneous Immunoassay for the Detection of Carisoprodol in Urine Guohong Wang*, Kim Huynh, Rekha Barhate, Warren Rodrigues, Christine Moore, Cynthia Coulter, Michael Vincent, and
TITLE: Guidelines for Treatment of Chronic Pain EFFECTIVE DATE: LAST REVISED DATE: LAST REVIEWED DATE:
 TITLE: Guidelines for Treatment of Chronic Pain EFFECTIVE DATE: 4.19.16 LAST REVISED DATE: 4.19.16 LAST REVIEWED DATE: 4.19.16 Table of Contents 1. How do I conduct my first few visits with a patient who
TITLE: Guidelines for Treatment of Chronic Pain EFFECTIVE DATE: 4.19.16 LAST REVISED DATE: 4.19.16 LAST REVIEWED DATE: 4.19.16 Table of Contents 1. How do I conduct my first few visits with a patient who
CNS DEPRESSANT OVERDOSE
 Signs and symptoms of CNS depressant overdose include: altered mental status, respiratory depression, hypotension, bradycardia, pulmonary edema, coma, and constricted pupils (opioids only). The following
Signs and symptoms of CNS depressant overdose include: altered mental status, respiratory depression, hypotension, bradycardia, pulmonary edema, coma, and constricted pupils (opioids only). The following
INTERPATH LABORATORY, INC. TEST UPDATES
 EFFECTIVE DATE: February 16, 2016 INTERPATH LABORATORY, INC. TEST UPDATES Please take note of the following test updates and make the appropriate changes in your Interpath Service Manual. Feel free to
EFFECTIVE DATE: February 16, 2016 INTERPATH LABORATORY, INC. TEST UPDATES Please take note of the following test updates and make the appropriate changes in your Interpath Service Manual. Feel free to
Drugs of Abuse I Serum
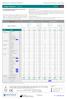 Drugs of Abuse I Serum 2017-11 1.10 For quality monitoring of quantitative measurements by chromatographic methods These reference materials are produced on the basis of human matrices. The highly accurate
Drugs of Abuse I Serum 2017-11 1.10 For quality monitoring of quantitative measurements by chromatographic methods These reference materials are produced on the basis of human matrices. The highly accurate
A Review of the Methods, Interpretation, and Limitations of the Urine Drug Screen
 Section Editor: Kelly M. Smith, PharmD, BCPS, FASHP, FCCP A Review of the Methods, Interpretation, and Limitations of the Urine Drug Screen E. Claire Markway, PharmD; Stephanie N. Baker, PharmD, BCPS Abstract:
Section Editor: Kelly M. Smith, PharmD, BCPS, FASHP, FCCP A Review of the Methods, Interpretation, and Limitations of the Urine Drug Screen E. Claire Markway, PharmD; Stephanie N. Baker, PharmD, BCPS Abstract:
C O P E MILWAUKEE. Milwaukee County Opioid-Related Overdose Report January 1, 2012 June 30, 2018 COMMUNITY OPIOID PREVENTION EFFORT
 Milwaukee County Opioid-Related Overdose Report January 1, 2012 June 30, 2018 MILWAUKEE C O P E COMMUNITY OPIOID PREVENTION EFFORT For additional information or if you have questions about the data presented
Milwaukee County Opioid-Related Overdose Report January 1, 2012 June 30, 2018 MILWAUKEE C O P E COMMUNITY OPIOID PREVENTION EFFORT For additional information or if you have questions about the data presented
Katee Kindler, PharmD, BCACP
 Speaker Introduction Katee Kindler, PharmD, BCACP Current Practice: Clinical Pharmacy Specialist Ambulatory Care, St. Vincent Indianapolis Assistant Professor of Pharmacy Practice, Manchester University,
Speaker Introduction Katee Kindler, PharmD, BCACP Current Practice: Clinical Pharmacy Specialist Ambulatory Care, St. Vincent Indianapolis Assistant Professor of Pharmacy Practice, Manchester University,
Rapid LC/TOF MS for Analytical Screening of Drugs in the Clinical Research Lab
 Application Note Clinical Research Rapid LC/TOF MS for Analytical Screening of Drugs in the Clinical Research Lab Authors Adam Barker 1,2, Frederick G. Strathmann 3, Natalie N. Rasmussen 4, and Carrie
Application Note Clinical Research Rapid LC/TOF MS for Analytical Screening of Drugs in the Clinical Research Lab Authors Adam Barker 1,2, Frederick G. Strathmann 3, Natalie N. Rasmussen 4, and Carrie
