THE UPTAKE AND LOCALIZATION OF CATECHOLAMINES IN CHICK EMBRYO SYMPATHETIC NEURONS IN TISSUE CULTURE
|
|
|
- Moris Banks
- 5 years ago
- Views:
Transcription
1 J. Cell Sci. 4, (1969) 677 Printed in Great Britain THE UPTAKE AND LOCALIZATION OF CATECHOLAMINES IN CHICK EMBRYO SYMPATHETIC NEURONS IN TISSUE CULTURE J. M. ENGLAND AND M. N. GOLDSTEIN Department of Anatomy, Washington University School of Medicine, St Louis, Missouri, U.S.A. SUMMARY The uptake of exogenous ['Hjdopamine, PHJnorepinephrine, and [ 3 H]epinephrine by dissociated chick embryo sympathetic neurons growing in tissue culture was studied by autoradiography. The neurons, growing in a medium containing nerve growth factor, rapidly and specifically took up all three catecholamines for at least 60 days, while no uptake was observed in several other cell types, including satellite cells and chick dorsal-root ganglion cells. The uptake was dependent on the concentration of the catecholamine and the duration of the pulse and was inhibited by cocaine and several sympathomimetic amines. Labelling was visualized only with fixatives which react with catecholamines to form waterinsoluble compounds. Autoradiographs showed that the label was much denser over the axons than the cell bodies. The label was distributed uniformly along the axons and did not seem to be preferentially localized at the axon terminals or varicosities which contain aggregates of dense core granules. These observations indicate that a large portion of the exogenous 3 [H]catecholamine is localized in an extragranular compartment and suggest that the differentiated function of the sympathetic neuronal cell membrane, which plays an important role in uptake, is retained after prolonged tissue culture. INTRODUCTION Postganglionic sympathetic neurons take up exogenous norepinephrine and structurally related amines (for review see Iversen, 1967). The kinetics of this uptake process and the effect of a variety of drugs which alter this process have been studied in intact animals (Whitby, Hertting & Axelrod, i960; Whitby, Axelrod & Weil-Malherbe, 1961), perfused organs with heavy adrenergic innervation (Kopin, Hertting & Gordon, 1962; Iversen, 1963), and sympathetic ganglia (Fischer & Snyder, 1965). [ 3 H]norepinephrine has been used with light- and electron-microscope autoradiography to localize uptake in the pineal body (Wolfe, Potter, Richardson & Axelrod, 1962), atrial myocardium (Wolfe & Potter, 1963), hypothalamus (Aghajanian & Bloom, 1966, 1967), spleen (Gillespie & Kirpekar, 1966), brain (Lenn, 1967), and vascular sympathetic axons (Devine & Simpson, 1968). In these organs the label was predominantly found in relation to small unmyelinated axons and nerve endings, many of which contained dense core granules. Levi-Montalcini & Angeletti (1963) reported that dissociated embryonic chick sympathetic neurons survive in tissue culture and undergo axonal regeneration when grown
2 678 J. M. England and M. N. Goldstein in a medium containing nerve growth factor (Levi-Montalcini & Booker, i960). The purpose of this study is to examine the uptake of phjcatecholamines by chick sympathetic neurons in tissue culture, in order: (1) to see if these cells retain this differentiated function after prolonged growth in vitro, and (2) to determine the disposition of exogenous catecholamines in the entire neuron, rather than in only its terminal axons. METHODS Sympathetic ganglia were dissected from 10-day-old chick embryos and incubated for 90 min in 0-3% crude trypsin (Difco 1-300) in Hanks's solution. The cells were dissociated by gentle pipetting in a small volume of trypsin solution, then washed 3 times in the culture medium consisting of 70% medium 199, 30% calf serum, 100 units/ml purified nerve growth factor (Levi-Montalcini & Booker, i960), and 100 units/ml penicillin. The washed cells were suspended in a small volume of medium and about 0-2 ml of suspension was placed on a 22 x 22 mm collagen-coated coverslip in a 35-mm plastic Petri dish. Eight to 10 cultures can be made from the pooled ganglia of 3 dozen embryos. Each culture was fed with 2 ml of medium and gently swirled to return the small cellular aggregates to the centre of the coverslip. Cultures were incubated at 36-5 C in a humidified atmosphere of 95% air and 5% CO 2 and re-fed at 48-h intervals. After 3 68 days in vitro the uptake of catecholamines was studied by incubating cultures for 1-60 min in medium containing 1-10/tc/ml of the following radioactive catecholamines: DL-norepinephrine-7-H 3.HCl (specific activity 7-27 c/mm), 3,4- dihydroxyphenylethyl-2-h 3 amine.hcl (phjdopamine) (specific activity 1-84 c/mm), and DL-epinephrine-7-H 3 (specific activity IO-I c/mm). After the pulse, the cultures were rinsed 4 times in warm Hanks's solution and fixed or chased in normal medium or in medium containing io~ B to \o~* M unlabelled catecholamines for periods ranging up to 24 h. Several fixatives were tested, including: 10% formalin in Hanks's; acetic acid:ethanol (2:6); acetic acid:ethanol:formalin (10:85:5); Bouin's solution; 3% glutaraldehyde in Hanks's (ph ); and buffered formol-dichromate (Wood, 1963). After fixation, autoradiographs were prepared using AR-10 stripping film as described previously (Goldstein, 1967). These preparations were exposed for 7 days at room temperature and stained with either 0-02% toluidine blue in 0-15 M (ph 6) phosphate buffer or 0-04 % aqueous crystal violet. Inhibition of [ 3 H]norepinephrine uptake was studied by incubating cultures in medium containing dopamine HC1, L-norepinephrine bitartrate, L-epinephrine bitartrate, or cocaine for 5-30 min and then adding [ 3 H]norepinephrine to this medium for the desired pulse period. For histological study, cultures were also fixed in 3% glutaraldehyde in Hanks's and stained by the Bodian method, or mounted in the fixative for study with positive phase-contrast optics.
3 Catecholamine uptake by chick neurons 679 RESULTS Dissociated sympathetic neurons in vitro Within 1 h after explantation the dissociated sympathetic ganglion cells reaggregated into small clumps on the surface of the collagen and by 24 h axons began to grow from the cellular aggregates. These early axonal outgrowths were typically very fine and had a complex pattern of terminal arborizations. With time the axons elongated greatly and by 15 days in vitro a dense network of axons was observed (Fig. 1). With a clump of ganglion cells as a centre of orientation, the axons were arranged differently in various regions of the culture. Immediately adjacent to the aggregate of cell bodies, the axons coursed randomly to form a dense mat of nerve fibres. As these fibres proceeded more peripherally, they established a parallel orientation with each other and formed bundles of axons which characteristically branched and rejoined, forming a complex network. Many axons terminated as free endings at the periphery, while others became entangled in the axonal network or within the clumps of cell bodies. After approximately 15 days in culture, small groups of neurons formed epitheliallike arrangements. These neurons had large nuclei and prominent nucleoli and were surrounded by small fusiform satellite cells with ovoid nuclei. Most of the satellite cells were in the aggregates of ganglion cell bodies, while only a few were associated with the bundles of axons. Fine neurites coursed over and around the neurons and their satellites (Figs. 2, 3). Long axons and shorter dendrites were observed arising from the neuron cell bodies (Fig. 4). The terminal axons and the axons which projected into the peripheral network of nerve fibres had many varicosities. A small number of fibroblasts were also observed in these cultures, but they did not survive under these culture conditions and often appeared vacuolated by the second week and degenerated shortly thereafter. The nerve cells on the other hand survived and appeared healthy even after 68 days in culture. Uptake of [ 3 H]catecholamines Autoradiographs of sympathetic ganglion cultures pulsed with [ 3 H]dopamine, [ 3 H]norepinephrine, or [ 3 H]epinephrine showed graining specifically concentrated over the axons. The distribution of the label was identical for the three catecholamines and was uniform along the entire length of the axons with no preferential accumulation at the axon terminals (Fig. 5). The visualization of this radioactive label was dependent on the method of fixation; graining resulting from [ 3 H]dopamine or [ 3 H]norepinephrine was observed after fixation with 3 % glutaraldehyde in Hanks's. However, [ 3 H]epinephrine could not be visualized with glutaraldehyde, although it, as well as other catecholamines, could be demonstrated after formol-dichromate fixation. The other fixatives which were used failed to show label over axons in cultures pulsed with any of the ph]catecholamines. The intensity of the graining, indicative of catecholamine uptake, was dependent on the concentration of the [ 3 H]catecholamine and the duration of the pulse. Because of the limits of resolution and the variability in axonal volume, it is difficult to quanti-
4 680 J. M. England and M. N. Goldstein tate these data precisely. Therefore, only a simple comparison can be made. The axons of cultures pulsed for 60 min with 5 /(c/ml [ 3 H]norepinephrine were more densely labelled than those pulsed with 1 /tc/ml for the same time. Similarly those pulsed with 5 /tc/ml phjnorepinephrine for 60 min were more densely labelled than those pulsed for 1 min. There was a marked disparity in the density of graining over the cell bodies and the axons. In contrast to the densely labelled axons, the large neuronal cell bodies were much less densely labelled (Fig. 6). A portion of the graining over the aggregate of cell bodies in Fig. 6 appears to be originating from axons coursing around and over the cell bodies. This close relationship between cell bodies and neuron fibres can be easily seen in the Bodian preparations in Figs. 2 and 3. The relatively lighter labelling of cell bodies as compared to axons does not appear to be an artifact of this autoradiographic technique, because the cell bodies in cultures pulsed with [ 3 H]leucine or PHjuridine were very densely labelled (unpublished observations). An apparent paradox was encountered when cell bodies lying on dense bundles of axons appeared densely labelled. However, close examination revealed that the graining pattern over these cell bodies was identical to and continuous with that over the adjacent bundles of axons. This dense graining may be an artifact caused by radiation from the [ 3 H]- catecholamines taken up by the axons which lie under these cell bodies. [ 3 H]norepinephrine uptake, a differentiated function of sympathetic neurons, was maintained even after prolonged culture. Neurons growing in culture for more than 60 days showed moderate to heavy graining over their axons when pulsed for 60 min with 5 /tc/ml [ 3 H]norepinephrine. In this system the labelling resulting from [ 3 H]- catecholamine uptake was specific for sympathetic neurons. No labelling was observed in HeLa cells, Chinese hamster fibroblasts or in chick fibroblasts, satellite cells, or the cell bodies and axons of dorsal root ganglion cells. Turnover of \*H]catecholamines After a pulse of 5 min with 5 /tc/ml [ 3 H]dopamine or phjnorepinephrine, there was a gradual decrease in the density of graining over the axons as cultures were incubated for increasing periods of time in a medium free from catecholamines. After a 24-h chase, the graining was still significant over the axons. However, when 5 x io" 4 M unlabelled dopamine or norepinephrine was added to the chase medium of cultures previously pulsed with 5 /tc/ml [ 3 H]dopamine or [ 3 H]norepinephrine for 5 min, there was a more rapid disappearance of label from the axons than in cultures chased with catecholamine-free medium. This decrease was very striking by the 5th hour of incubation in the chase medium. Inhibition of [ 3 H]norepinephrine uptake The uptake of phjnorepinephrine was partially or totally inhibited by cocaine and several unlabelled catecholamines. Cultures which were incubated with io~ 5 M cocaine for 30 min and then pulsed with 5 /ic/ml phjnorepinephrine for 20 min in the presence of this concentration of cocaine, showed a partial inhibition of uptake (Fig. 8) as compared to cultures pulsed without cocaine in the medium (Fig. 7). Total inhibi-
5 Catecholamine uptake by chick neurons 681 tion of uptake, detectable by autoradiography, was obtained with IO^M cocaine. These cultures showed no graining over either axons or cell bodies and appeared identical to Fig. 9 (in which uptake was completely inhibited by unlabelled dopamine). The uptake of 2 /tc/ml [ 3 H]norepinephrine during a 5-min pulse was completely inhibited by incubating cultures for 5 min with either 6x IO~ 5 M L-epinephrine bitartrate or 6 x io~ 5 M dopamine. HC1 and pulsing in the presence of the unlabelled catecholamines. Inhibition of uptake by dopamine is shown in Fig. 9, where axons are completely unlabelled. Identical results were obtained when uptake was inhibited with epinephrine. In all cases graining in the control cultures was uniformly dense along the entire length of the axon. DISCUSSION Dissociated chick sympathetic neurons actively grow in tissue culture by axonal proliferation and elongation. The retention of differentiated function by these cells after prolonged growth in vitro provides a system for analysing catecholamine uptake and turnover in both the cell bodies and axons of growing sympathetic neurons. At all stages of axonal outgrowth the cultured neurons rapidly took up PH] dopamine, [ 3 H]norepinephrine, and phjepinephrine from the culture medium so that there was intense axonal labelling after pulse periods as short as 1 min. Examples of this phenomenon have been reported after the in vivo administration or in vitro organ perfusion of [ 3 H]catecholamines (Iversen, 1967). The present work demonstrates that sympathetic neurons isolated from presynaptic input and end-organ connexions are able to take up exogenous catecholamines. The label resulting from the exogenous [ 3 H] catecholamines was uniformly distributed along the entire axon. It did not appear to be preferentially localized at the varicosities or in the terminal portion of the axons. Electron microscopy demonstrated concentrations of dense core granules in these dilatations and relatively few granules in other portions of the axon (unpublished observations). This supports the observations of Malmfors (1965), who found that, when catecholamine-specific fluorescence was restored to reserpine-depleted axons by exogenous norepinephrine with a monoamine oxidase inhibitor, the terminals appeared more uniformly fluorescent than the controls. In addition, Van Orden, Bensch & Giarman (1967) found this restoration of fluorescence did not result in a concomitant increase in the number of dense-core granules. This evidence suggests that in addition to the [ 3 H]catecholamine taken up and localized at nerve endings in association with dense-core granules (Wolfe et al. 1962; Wolfe & Potter, 1963; Aghajanian & Bloom, 1966, 1967; Lenn, 1967; Devine & Simpson, 1968) some is stored, at least temporarily, in the axoplasm independent of the dense-core granules. The present data support the work of Van Orden et al. (1967), who demonstrated an extragranular catecholamine storage compartment. Both studies emphasize the importance of a functionally differentiated axonal membrane in the uptake process. Glutaraldehyde preserved [ 3 H]dopamine and ph]norepinephrine label best of the fixatives tested, although label could be visualized also with formol-dichromate. How- 43 Cell Sci. 4
6 682 J. M. England and M. N. Goldstein ever, [ 3 H]epinephrine was preserved only by formol-dichromate. Hopwood & Coupland (1965) demonstrated that several biogenic amines, including norepinephrine and dopamine, react with glutaraldehyde to form an insoluble complex while epinephrine fails to react. On the other hand, epinephrine, as well as other catecholamines, after exposure to an oxidizing agent such as potassium dichromate, is transformed by the chromaffine reaction to a dark, water-insoluble precipitate (Hillarp & Hokfelt, 1955). This supports the hypothesis that at least a portion of the [ 3 H]catecholamines are not firmly bound in the axon, but free in the axoplasm. Catecholamines appear to be washed out of the axons when fixation does not convert them into water-insoluble compounds. Thus, they appear to be visualized only when precipitated in situ by the fixative. There appears to be a striking non-uniformity in the distribution of [ 3 H]catecholamines in the neurons. Most of the label appears to be localized within the axons, with much less in the cell bodies. In fact, a portion of the sparse label seen over the cell bodies may actually be due to the ph]catecholamine in the small nerve fibres in this region. It is impossible to determine the precise site of the radioactivity in the cellular aggregates by using whole-mount preparations. It is hoped that thin-section autoradiography, now in progress, will provide conclusive results. Fischer & Snyder (1965) reported rapid ph] norepinephrine uptake by the superior cervical ganglion; however, it is difficult in their study to assess the relative contribution of the cell bodies and intraganglionic neurites to this uptake. Three possible explanations for this unequal distribution of label are: (1) the exogenous catecholamine may be degraded more rapidly in the cell body than in the axon to compounds which possibly do not undergo the glutaraldehyde or chromaffine reactions; (2) there may be a very rapid flow of catecholamines from the cell body into the axons; or (3) there may be a selective difference in the uptake mechanism in the membrane of the cell body and axon. Studies are in progress to determine which of these hypothesized mechanisms is mainly responsible for this unequal distribution. The uptake of [ 3 H]norepinephrine by cultured neurons is inhibited by cocaine and several sympathomimetic amines, a finding similar to the results described for other systems, including the isolated rat heart (Burgen & Iversen, 1965; Iversen, 19*65). This functional similarity to other more conventional systems and the relative stability of this differentiated function in prolonged tissue culture suggests that sympathetic neurons and perhaps selected regions of the central nervous system in vitro may provide useful models for studying the uptake, storage, and possibly release of catecholamines in an accessible and isolated environment. Investigations with human foetal sympathetic and human sympathicoblastomas in tissue culture have given similar results (Goldstein, 1967). This investigation was supported by U.S.P.H.S. grant number GM
7 Catecholamine uptake by chick neurons 683 REFERENCES AGHAJANIAN, G. K. & BLOOM, F. E. (1966). Electron-microscopic autoradiography of rat hypothalamus after intraventricular H 3 norepinephrine. Science, N.Y. 153, AGHAJANIAN, G. K. & BLOOM, F. E. (1967). Electron-microscopic localization of tritiated norepinephrine in rat brain: effect of drugs. J. Pharmac. exp. Ther. 156, BURGEN, A. S. V. & IVERSEN, L. L. (1965). The inhibition of noradrenaline uptake by sympathomimetic amines in the rat isolated heart. Br. J. Pharmac. Chemother. 25, DEVINE, C. E- & SIMPSON, F. O. (1968). Localization of tritiated norepinephrine in vascular sympathetic axons of the rat intestine and mesentery by electron microscope radioautography. J. CellBiol. 38, FISCHER, J. E. & SNYDER, S. (1965). Disposition of norepinephrine H 3 in sympathetic ganglia. J. Pharmac. exp. Ther. 150, GILLESPIE, J. S. & KIRPEKAR, S. M. (1966). The histological localization of noradrenaline in the cat spleen. J. PhysioL, Lond. 187, GOLDSTEIN, M. N. (1967). Incorporation and release of H 3 catecholamines by cultured fetal human sympathetic nerve cells and neuroblastoma cells. Proc. Soc. exp. Biol. Med. 125, HILLARP, N.-A. & HOKFELT, B. (19SS). Histochemical demonstration of noradrenaline and adrenaline in the adrenal medulla. J. Histochem. Cytochem. 3, 1-5. HOPWOOD, D. & COUPLAND, R. E. (1965). Further observations on the reaction between biogenic amines and glutaraldehyde. J. Anat. 99, 414. IVERSEN, L. L. (1963). Uptake of noradrenaline by the isolated perfused rat heart. Br. J. Pharmac. Chemother. 21, IVERSEN, L. L. (1965). The inhibition of noradrenaline uptake by drugs. Adv. Drug Res. 2, IVERSEN, L. L. (1967). The uptake of catecholamines by sympathetic nerves. In The Uptake and Storage of Noradrenaline in Sympathetic Nerves, pp Cambridge University Press. KOPIN, I. J., HERTTINC, G. & GORDON, E. K. (1962). Fate of norepinephrine-h 3 in the isolated perfused rat heart. J. Pharmac. exp. Ther. 138, LENN, N. J. (1967). Localization of uptake of tritiated norepinephrine by rat brain in vivo and in vitro using electron microscope autoradiography. Am. J. Anat. 120, LEVI-MONTALCINI, R. & ANCELETTI, P. U. (1963). Essential role of the nerve growth factor in the survival and maintenance of dissociated sensory and sympathetic embryonic nerve cells in vitro. Devi Biol. 7, LEVI-MONTALCINI, R. & BOOKER, B. (i960). Excessive growth of sympathetic ganglia evoked by a protein isolated from mouse salivary glands. Proc. natn. Acad. Sci. U.S.A. 46, MALMFORS, T. (1965). Studies on adrenergic nerves. Actaphysiol. scand. 64, Suppl. 248, VAN ORDEN, L. S., Ill, BENSCH, K. G. & GIARMAN, N. J. (1967). Histochemical and functional relationships of catecholamines in adrenergic nerve endings. II. Extravesicular norepinephrine. J. Pharmac. exp. Ther. 155, WHITBY, L. G., AXELROD, J. & WEIL-MALHERBE, H. (1961). The fate of H 3 -norepinephrine in animals. J. Pharmac. exp. Ther. 132, WHITBY, L. G., HERTTING, G. & AXELROD, J. (i960). Effect of cocaine on the disposition of noradrenaline labelled with tritium. Nature, Lond. 187, WOLFE, D. E. & POTTER, L. T. (1963). Localization of norepinephrine in the atrial myocardium. Anat. Rec. 145, 301. WOLFE, D. E., POTTER, L. T., RICHARDSON, K. C. & AXELROD, J. (1962). Localizing tritiated norepinephrine in sympathetic axons by electron microscopic autoradiography. Science, N. Y. 138, WOOD, J. G. (1963). Identification of and observations on epinephrine and norepinephrine containing cells in the adrenal medulla. Am. J. Anat. 112, (Received 12 August 1968) 43-2
8 684 J- M - England and M. N. Goldstein The photomicrographs are of dissociated io-day-old chick embryo sympathetic ganglia grown for various periods in tissue culture. Fig. i. Note the dense fascicles of axons which extend from the aggregates of neuron cell bodies and satellite cells. Fifteen days in vitro, x 40.
9 Catecholamine uptake by chick neurons 685 /.:
10 686 J. M. England and M. N. Goldstein Figs. 2, 3. Large flattened ganglion cell bodies with associated neurites are surrounded by numerous small satellite cells with dense ovoid nuclei. Note the axons and dendrites which course below and above the ganglion cells. Bodian stain. Thirty days in vitro. Fig. 2, x 1250; Fig. 3, X750. Fig. 4. Numerous neurites arise from the large ganglion cells to form a dense network. Satellite cells adjacent to neurons are out of the plane of focus. Fixed in 3 % glutaraldehyde; phase-contrast. Fifteen days in vitro, x 900.
11 Catecholamine uptake by chick neurons 687
12 688 J. M. England and M. N. Goldstein Fig. 5. Neurons pulsed with 2-5 fic/m\ PH]norepinephrine for iomin; washed and chased for 1 h. Note the even distribution of graining along the axon bundles and terminals. This distribution is seen in experiments fixed immediately after pulsing. Eight days in vitro, x 475. Fig. 6. Neurons pulsed with 5 fic/m\ PHJnorepinephrine for 60 min; washed and chased for 10 min. There is much less graining over the cell bodies than over the adjacent axons. A portion of the graining over the aggregate of cell bodies appears to be derived from axons which course over and around these cell bodies. Because of the thickness of the preparation, two photographs taken at different focal planes were superimposed LO show graining accurately over both cell bodies and axons. Ten days in vitro, x 950.
13 Catecholamine uptake by chick neurons
14 690 J. M. England and M. N. Goldstein Fig. 7. Neurons pulsed with 5 /ic/ml [ 3 H]norepinephrine for 20 min; washed and chased for 10 min. Note the very dense graining over the bundles of axons. Five days in vitro, x 900. Fig. 8. Neurons pretreated with io~ B M cocaine for 30 min, then pulsed in the presence of cocaine with 5 /ic/ml [ 3 H]norepinephrine for 20 min; washed and chased for 10 min. Note the marked decrease in graining over axons as compared with control in Fig. 7. Five days in vitro, x 900. Fig. 9. Neurons pretreated with 6x io~ 5 M dopamine.hc1 for 5 min, then pulsed in the presence of dopamine with 2 /tc/ml fjm-tlnorepinephrine; washed and chased for 10 min. Note complete absence of graining over axons; comparable controls were densely labelled. Eight days in vitro, x 900.
15 Catecholamine uptake by chick neurons
16
ENHANCEMENT OF THE GRANULATION OF ADRFNERGIC STORAGE VESICLES IN DRUG-FREE SOLUTION
 ENHANCEMENT OF THE GRANULATION OF ADRFNERGIC STORAGE VESICLES IN DRUG-FREE SOLUTION TAKASHI IWAYAMA and J. B. FURNESS. From the Department of Zoology, University of Melbourne, Victoria, Australia. Dr.
ENHANCEMENT OF THE GRANULATION OF ADRFNERGIC STORAGE VESICLES IN DRUG-FREE SOLUTION TAKASHI IWAYAMA and J. B. FURNESS. From the Department of Zoology, University of Melbourne, Victoria, Australia. Dr.
Monoamine oxidase in sympathetic nerves: a transmitter specific enzyme type
 Br. J. Pharmac. (1971), 43, 814-818. Monoamine oxidase in sympathetic nerves: a transmitter specific enzyme type C. GORIDIS AND N. H. NEFF Laboratory of Preclinical Pharmacology, National Institute of
Br. J. Pharmac. (1971), 43, 814-818. Monoamine oxidase in sympathetic nerves: a transmitter specific enzyme type C. GORIDIS AND N. H. NEFF Laboratory of Preclinical Pharmacology, National Institute of
A comparison of the sensitivities of innervated and denervated rat vasa deferentia to agonist drugs
 Br. J. Pharmac. (1970), 39, 748-754. A comparison of the sensitivities of innervated and denervated rat vasa deferentia to agonist drugs A. T. BIRMINGHAM*, G. PATRSON AND J. W6JCICKIt Department of Pharmacology,
Br. J. Pharmac. (1970), 39, 748-754. A comparison of the sensitivities of innervated and denervated rat vasa deferentia to agonist drugs A. T. BIRMINGHAM*, G. PATRSON AND J. W6JCICKIt Department of Pharmacology,
Sympathetic Nerve Cell Destruction in Newborn Mammals by 6-Hydroxydopamine P. U. Angeletti and R. Levi-Montalcini
 Proceedings of the National Academy of Sciences Vol. 65, No. 1, pp. 114-121, January 1970 Sympathetic Nerve Cell Destruction in Newborn Mammals by 6-Hydroxydopamine P. U. Angeletti and R. Levi-Montalcini
Proceedings of the National Academy of Sciences Vol. 65, No. 1, pp. 114-121, January 1970 Sympathetic Nerve Cell Destruction in Newborn Mammals by 6-Hydroxydopamine P. U. Angeletti and R. Levi-Montalcini
Disappearance of small vesicles from adrenergic nerve endings in the rat vas deferens caused by red back spider venom
 Journal of Neurocytology z, 465-469 (I973) Disappearance of small vesicles from adrenergic nerve endings in the rat vas deferens caused by red back spider venom R. C. HAMILTON 1 and P. M. ROBINSON~ 1Commonwealth
Journal of Neurocytology z, 465-469 (I973) Disappearance of small vesicles from adrenergic nerve endings in the rat vas deferens caused by red back spider venom R. C. HAMILTON 1 and P. M. ROBINSON~ 1Commonwealth
Dopamine-s-Hydroxylase in the Rat Superior Cervical Ganglia
 Proc. Nat. Acad. Sci. USA Vol. 68, No. 7, pp. 1598-1602, July 1971 Selective Induction by Nerve Growth Factor of Tyrosine Hydroxylase and Dopamine-s-Hydroxylase in the Rat Superior Cervical Ganglia (dopa
Proc. Nat. Acad. Sci. USA Vol. 68, No. 7, pp. 1598-1602, July 1971 Selective Induction by Nerve Growth Factor of Tyrosine Hydroxylase and Dopamine-s-Hydroxylase in the Rat Superior Cervical Ganglia (dopa
ACTIONS OF BRETYLIUM AND GUANETHIDINE ON THE UPTAKE AND RELEASE OF [3H]-NORADRENALINE
![ACTIONS OF BRETYLIUM AND GUANETHIDINE ON THE UPTAKE AND RELEASE OF [3H]-NORADRENALINE ACTIONS OF BRETYLIUM AND GUANETHIDINE ON THE UPTAKE AND RELEASE OF [3H]-NORADRENALINE](/thumbs/74/71109260.jpg) Brit. J. Pharmacol. (1962), 18, 161-166. ACTIONS OF BRETYLIUM AND GUANETHIDINE ON THE UPTAKE AND RELEASE OF [3H]-NORADRENALINE BY G. HERTTING,* J. AXELROD AND R. W. PATRICK From the Laboratory of Clinical
Brit. J. Pharmacol. (1962), 18, 161-166. ACTIONS OF BRETYLIUM AND GUANETHIDINE ON THE UPTAKE AND RELEASE OF [3H]-NORADRENALINE BY G. HERTTING,* J. AXELROD AND R. W. PATRICK From the Laboratory of Clinical
suggesting that the release of noradrenaline from sympathetic fibres was dependent on the concentration of Ca2+ outside the fibre.
 214 J. Phy8iol. (1965), 181, pp. 214-223 With 4 text-figurem Printed in Great Britain THE RELEASE OF NORADRENALINE FROM SYMPATHETIC FIBRES IN RELATION TO CALCIUM CONCENTRATION BY J. H. BURN AND W. R. GIBBONS
214 J. Phy8iol. (1965), 181, pp. 214-223 With 4 text-figurem Printed in Great Britain THE RELEASE OF NORADRENALINE FROM SYMPATHETIC FIBRES IN RELATION TO CALCIUM CONCENTRATION BY J. H. BURN AND W. R. GIBBONS
PENTOBARBITONE AND COLCHICINE ON DEGENERATION
 Br. J. Pharmac. (1978), 62, 55-561 TIME COURSE OF DEGENERATION OF SHORT AND LONG POSTGANGLIONIC SYMPATHETIC NERVE FIBRES AND EFFECT OF PENTOBARBITONE AND COLCHICINE ON DEGENERATION ARUN R. WAKADE Department
Br. J. Pharmac. (1978), 62, 55-561 TIME COURSE OF DEGENERATION OF SHORT AND LONG POSTGANGLIONIC SYMPATHETIC NERVE FIBRES AND EFFECT OF PENTOBARBITONE AND COLCHICINE ON DEGENERATION ARUN R. WAKADE Department
THE QUESTION OF RELATIONSHIP BETWEEN GOLGI VESICLES AND SYNAPTIC VESICLES IN OCTOPUS NEURONS
 J. Cell Set. 7, 89- (97) Printed in Great Britain THE QUESTION OF RELATIONSHIP BETWEEN GOLGI VESICLES AND SYNAPTIC VESICLES IN OCTOPUS NEURONS E. G. GRAY Department of Anatomy, University College London,
J. Cell Set. 7, 89- (97) Printed in Great Britain THE QUESTION OF RELATIONSHIP BETWEEN GOLGI VESICLES AND SYNAPTIC VESICLES IN OCTOPUS NEURONS E. G. GRAY Department of Anatomy, University College London,
Replacement of Nerve-Growth Factor by Ganglionic Non-Neuronal Cells for the Survival In Vitro of Dissociated Ganglionic Neurons (culture neuroglia)
 Proc. Nat. Acad. Sci. USA VoL 69, No. 12, pp. 3556-3560, December 1972 Replacement of Nerve-Growth Factor by Ganglionic Non-Neuronal Cells for the Survival In Vitro of Dissociated Ganglionic Neurons (culture
Proc. Nat. Acad. Sci. USA VoL 69, No. 12, pp. 3556-3560, December 1972 Replacement of Nerve-Growth Factor by Ganglionic Non-Neuronal Cells for the Survival In Vitro of Dissociated Ganglionic Neurons (culture
Adrenergic fibres in the human intestine
 Gut, 1968, 9, 678-682 Adrenergic fibres in the human intestine L. CAPURSO,1 C. A. FRIEDMANN, AND A. G. PARKS From the Research Department, St Mark's Hospital, London, and the London Hospital, Whitechapel,
Gut, 1968, 9, 678-682 Adrenergic fibres in the human intestine L. CAPURSO,1 C. A. FRIEDMANN, AND A. G. PARKS From the Research Department, St Mark's Hospital, London, and the London Hospital, Whitechapel,
Fine Structure of the Normal Trigeminal Ganglion in the Cat and Monkey*
 Fine Structure of the Normal Trigeminal Ganglion in the Cat and Monkey* DAVID S. MAXWELL, PH.D. Principal Contributor and Leader of Discussion HE inclusion of animal material m a y be justified as a means
Fine Structure of the Normal Trigeminal Ganglion in the Cat and Monkey* DAVID S. MAXWELL, PH.D. Principal Contributor and Leader of Discussion HE inclusion of animal material m a y be justified as a means
ROLE OF ADRENAL HORMONES IN MAINTAINING TISSUE STORES OF NORADRENALINE DURING INCREASED SYMPATHETIC ACTIVITY
 Br. J. Pharmac. Chemother. (1966), 27, 532-535. ROLE OF ADRENAL HORMONES IN MAINTAINING TISSUE STORES OF NORADRENALINE DURING INCREASED SYMPATHETIC ACTIVITY BY V. M. AVAKIAN* AND MARTHE VOGT From the Agricultural
Br. J. Pharmac. Chemother. (1966), 27, 532-535. ROLE OF ADRENAL HORMONES IN MAINTAINING TISSUE STORES OF NORADRENALINE DURING INCREASED SYMPATHETIC ACTIVITY BY V. M. AVAKIAN* AND MARTHE VOGT From the Agricultural
LOCALIZATION OF MONOAMINE OXIDASE IN MAMMALIAN
 Brit. Heart J., 1965, 27, 116. LOCALIZATION OF MONOAMINE OXIDASE IN MAMMALIAN AND REPTILIAN HEART BY E. MULLER* AND A. G. E. PEARSEt From the Department ofpathology, Postgraduate Medical School, London
Brit. Heart J., 1965, 27, 116. LOCALIZATION OF MONOAMINE OXIDASE IN MAMMALIAN AND REPTILIAN HEART BY E. MULLER* AND A. G. E. PEARSEt From the Department ofpathology, Postgraduate Medical School, London
Lesson 33. Objectives: References: Chapter 16: Reading for Next Lesson: Chapter 16:
 Lesson 33 Lesson Outline: Nervous System Structure and Function Neuronal Tissue Supporting Cells Neurons Nerves Functional Classification of Neuronal Tissue Organization of the Nervous System Peripheral
Lesson 33 Lesson Outline: Nervous System Structure and Function Neuronal Tissue Supporting Cells Neurons Nerves Functional Classification of Neuronal Tissue Organization of the Nervous System Peripheral
ANTAGONISM OF THE BLOCKING ACTION OF BRETYLIUM AND GUANETHIDINE ON THE PRESSOR EFFECT OF PHYSOSTIGMINE IN THE RAT
 Brit. J. Pharmacol. (1965), 24, 624-631. ANTAGONISM OF THE BLOCKING ACTION OF BRETYLIUM AND GUANETHIDINE ON THE PRESSOR EFFECT OF PHYSOSTIGMINE IN THE RAT BY S. D. GOKHALE, 0. D. GULATI AND N. Y. JOSHI
Brit. J. Pharmacol. (1965), 24, 624-631. ANTAGONISM OF THE BLOCKING ACTION OF BRETYLIUM AND GUANETHIDINE ON THE PRESSOR EFFECT OF PHYSOSTIGMINE IN THE RAT BY S. D. GOKHALE, 0. D. GULATI AND N. Y. JOSHI
C'4-tryptophan in rat pineals in organ culture. Other compounds related in
 STIMULATION OF C14-MELATONIN SYNTHESIS FROM C14-TRYPTOPHAN BY NORADRENALINE IN RAT PINEAL IN ORGAN CULTURE* BY JULIUS AXELROD, HARVEY M. SHEIN, AND RICHARD J. WURTMAN LABORATORY OF CLINICAL SCIENCE, NATIONAL
STIMULATION OF C14-MELATONIN SYNTHESIS FROM C14-TRYPTOPHAN BY NORADRENALINE IN RAT PINEAL IN ORGAN CULTURE* BY JULIUS AXELROD, HARVEY M. SHEIN, AND RICHARD J. WURTMAN LABORATORY OF CLINICAL SCIENCE, NATIONAL
H. istochemical 1 " 0 and chemical 7 ' 8 evidence
 Dopamine: A retinal neurotransmitter II. Autoradiographic localization of H3-dopamine in the retina Steven G. Kramer, Albert M. Potts, and Yvonne Mangnall Retinal uptake of tritiated dopamine was induced
Dopamine: A retinal neurotransmitter II. Autoradiographic localization of H3-dopamine in the retina Steven G. Kramer, Albert M. Potts, and Yvonne Mangnall Retinal uptake of tritiated dopamine was induced
Introduction to Autonomic
 Part 2 Autonomic Pharmacology 3 Introduction to Autonomic Pharmacology FUNCTIONS OF THE AUTONOMIC NERVOUS SYSTEM The autonomic nervous system (Figure 3 1) is composed of the sympathetic and parasympathetic
Part 2 Autonomic Pharmacology 3 Introduction to Autonomic Pharmacology FUNCTIONS OF THE AUTONOMIC NERVOUS SYSTEM The autonomic nervous system (Figure 3 1) is composed of the sympathetic and parasympathetic
THE ACTION OF GUANETHIDINE WITH PARTICULAR REFERENCE TO THE SYMPATHETIC NERVOUS SYSTEM
 Brit. J. Pharinacol. (1963), 20, 171-177. THE ACTION OF GUANETHIDINE WITH PARTICULAR REFERENCE TO THE SYMPATHETIC NERVOUS SYSTEM BY G. F. ABERCROMBIE AND B. N. DAVIES From the Department of Physiology,
Brit. J. Pharinacol. (1963), 20, 171-177. THE ACTION OF GUANETHIDINE WITH PARTICULAR REFERENCE TO THE SYMPATHETIC NERVOUS SYSTEM BY G. F. ABERCROMBIE AND B. N. DAVIES From the Department of Physiology,
SOME PHARMACOLOGICAL ACTIONS OF DIETHYLDITHIOCARBAMATE ON RABBIT AND RAT ILEUM
 Br. J. Pharmac. Chemother. (1968), 32, 42-49. SOME PHARMACOLOGICAL ACTIONS OF DIETHYLDITHIOCARBAMATE ON RABBIT AND RAT ILEUM BY G. G. S. COLLINS* AND G. B. WESTt From the Department of Pharmacology, School
Br. J. Pharmac. Chemother. (1968), 32, 42-49. SOME PHARMACOLOGICAL ACTIONS OF DIETHYLDITHIOCARBAMATE ON RABBIT AND RAT ILEUM BY G. G. S. COLLINS* AND G. B. WESTt From the Department of Pharmacology, School
Department of Anatomy, Glasgow University
 THE UPTAKE OF LABELLED SULPHATE INJECTED INTO THE HOST ANIMAL BY CARTILAGE HOMOGRAFTS By G. M. WYBURN, D.Sc., F.R.F.P.S.G., and P. BACSICH, D.Sc., M.D. Department of Anatomy, Glasgow University INTRODUCTION
THE UPTAKE OF LABELLED SULPHATE INJECTED INTO THE HOST ANIMAL BY CARTILAGE HOMOGRAFTS By G. M. WYBURN, D.Sc., F.R.F.P.S.G., and P. BACSICH, D.Sc., M.D. Department of Anatomy, Glasgow University INTRODUCTION
Chapter 14 The Autonomic Nervous System Chapter Outline
 Chapter 14 The Autonomic Nervous System Chapter Outline Module 14.1 Overview of the Autonomic Nervous System (Figures 14.1 14.3) A. The autonomic nervous system (ANS) is the involuntary arm of the peripheral
Chapter 14 The Autonomic Nervous System Chapter Outline Module 14.1 Overview of the Autonomic Nervous System (Figures 14.1 14.3) A. The autonomic nervous system (ANS) is the involuntary arm of the peripheral
MODIFICATIONS BY PROPRANOLOL OF THE RESPONSE OF ISOLATED RABBIT ATRIA TO ENDOGENOUS AND EXOGENOUS NORADRENALINE
 Br. J. Pharmac. Chemother. (1968), 32, 539-545. MODIFICATIONS BY PROPRANOLOL OF THE RESPONSE OF ISOLATED RABBIT ATRIA TO ENDOGENOUS AND EXOGENOUS NORADRENALINE BY K. SHIMAMOTO AND N. TODA From the Department
Br. J. Pharmac. Chemother. (1968), 32, 539-545. MODIFICATIONS BY PROPRANOLOL OF THE RESPONSE OF ISOLATED RABBIT ATRIA TO ENDOGENOUS AND EXOGENOUS NORADRENALINE BY K. SHIMAMOTO AND N. TODA From the Department
ELECTRON MICROSCOPIC RADIOAUTOGRAPHIC STUDIES OF THE CAROTID BODY FOLLOWING INJECTIONS OF LABELED BIOGENIC AMINE PRECURSORS
 ELECTRON MICROSCOPIC RADIOAUTOGRAPHIC STUDIES OF THE CAROTID BODY FOLLOWING INJECTIONS OF LABELED BIOGENIC AMINE PRECURSORS I-LI CHEN and ROBERT D. YATES From the Department of Anatomy, The Universty of
ELECTRON MICROSCOPIC RADIOAUTOGRAPHIC STUDIES OF THE CAROTID BODY FOLLOWING INJECTIONS OF LABELED BIOGENIC AMINE PRECURSORS I-LI CHEN and ROBERT D. YATES From the Department of Anatomy, The Universty of
Nervous tissue. Lab. 7
 Nervous tissue Lab. 7 Nervous tissue :- is responsible for transport nervous impulse (motor and sensory impulse), and it is formed by network more than 100 million nerve cell (neurons), nerve fiber and
Nervous tissue Lab. 7 Nervous tissue :- is responsible for transport nervous impulse (motor and sensory impulse), and it is formed by network more than 100 million nerve cell (neurons), nerve fiber and
Neurotransmitter Systems I Identification and Distribution. Reading: BCP Chapter 6
 Neurotransmitter Systems I Identification and Distribution Reading: BCP Chapter 6 Neurotransmitter Systems Normal function of the human brain requires an orderly set of chemical reactions. Some of the
Neurotransmitter Systems I Identification and Distribution Reading: BCP Chapter 6 Neurotransmitter Systems Normal function of the human brain requires an orderly set of chemical reactions. Some of the
The developmental history of a sensory ganglion cell
 (bozza v1, luglio 2016 per VC) The developmental history of a sensory ganglion cell by Brian Freeman, School of Medical Sciences, UNSW (b.freeman@unsw.edu.au) The sensory ganglion cell (e.g., in spinal
(bozza v1, luglio 2016 per VC) The developmental history of a sensory ganglion cell by Brian Freeman, School of Medical Sciences, UNSW (b.freeman@unsw.edu.au) The sensory ganglion cell (e.g., in spinal
Sympathetic Innervation of the Developing Rabbit Heart
 Sympathetic Innervation of the Developing Rabbit Heart BIOCHEMICAL AND HISTOCHEMICAL COMPARISONS OF FETAL, NEONATAL, AND ADULT MYOCARDIUM By William F. Friedman, M.D., Peter E. Pool, M.D., David Jacobowitx,
Sympathetic Innervation of the Developing Rabbit Heart BIOCHEMICAL AND HISTOCHEMICAL COMPARISONS OF FETAL, NEONATAL, AND ADULT MYOCARDIUM By William F. Friedman, M.D., Peter E. Pool, M.D., David Jacobowitx,
[1920], in studies on the human pleural membrane, pointed out the
![[1920], in studies on the human pleural membrane, pointed out the [1920], in studies on the human pleural membrane, pointed out the](/thumbs/84/89127006.jpg) 'ca -.101 6II.25:6II.OI8.86 NERVES AND NERVE ENDINGS IN THE VISCERAL PLEURA OF THE CAT. BY A. I. G. McLAUGHLIN. (From the Unit Laboratories, University College Hospital Medical School.) (Received September
'ca -.101 6II.25:6II.OI8.86 NERVES AND NERVE ENDINGS IN THE VISCERAL PLEURA OF THE CAT. BY A. I. G. McLAUGHLIN. (From the Unit Laboratories, University College Hospital Medical School.) (Received September
Autonomic Nervous System
 ANS..??? Autonomic Nervous System Nervous system CNS PNS Autonomic Somatic Symp Parasymp Enteric SOMATIC AUTONOMIC Organ supplied Skeletal muscle Other organs Distal most synapse Nerve fibre Peripheral
ANS..??? Autonomic Nervous System Nervous system CNS PNS Autonomic Somatic Symp Parasymp Enteric SOMATIC AUTONOMIC Organ supplied Skeletal muscle Other organs Distal most synapse Nerve fibre Peripheral
THE ACTION OF NICOTINE ON THE CILIARY GANGLION
 Brit. J. Pharmnacol. (1952), 7, 665. THE ACTION OF NICOTINE ON THE CILIARY GANGLION BY BRENDA M. SCHOFIELD From the Department of Pharmacology, University of Oxford (Received June 7, 1952) The existing
Brit. J. Pharmnacol. (1952), 7, 665. THE ACTION OF NICOTINE ON THE CILIARY GANGLION BY BRENDA M. SCHOFIELD From the Department of Pharmacology, University of Oxford (Received June 7, 1952) The existing
Chapter 15: The Autonomic Nervous System. Copyright 2009, John Wiley & Sons, Inc.
 Chapter 15: The Autonomic Nervous System Comparison of Somatic and Autonomic Nervous Systems Comparison of Somatic and Autonomic Nervous Systems Anatomy of Autonomic Motor Pathways Preganglionic neuron
Chapter 15: The Autonomic Nervous System Comparison of Somatic and Autonomic Nervous Systems Comparison of Somatic and Autonomic Nervous Systems Anatomy of Autonomic Motor Pathways Preganglionic neuron
AUTONOMIC NERVOUS SYSTEM PART I: SPINAL CORD
 AUTONOMIC NERVOUS SYSTEM PART I: SPINAL CORD How is the organization of the autonomic nervous system different from that of the somatic nervous system? Peripheral Nervous System Divisions Somatic Nervous
AUTONOMIC NERVOUS SYSTEM PART I: SPINAL CORD How is the organization of the autonomic nervous system different from that of the somatic nervous system? Peripheral Nervous System Divisions Somatic Nervous
Action of drugs on denervated myoepithelial cells of salivary glands
 Br. J. Pharmac. (1973), 48, 73-79. Action of drugs on denervated myoepithelial cells of salivary glands N. EMMELIN AND A. THULIN Institute of Physiology, University of Lund, Sweden Summary 1. The pressure
Br. J. Pharmac. (1973), 48, 73-79. Action of drugs on denervated myoepithelial cells of salivary glands N. EMMELIN AND A. THULIN Institute of Physiology, University of Lund, Sweden Summary 1. The pressure
Biology 218 Human Anatomy
 Chapter 17 Adapted form Tortora 10 th ed. LECTURE OUTLINE A. Overview of the Nervous System (p. 537) 1. The nervous system and the endocrine system are the body s major control and integrating centers.
Chapter 17 Adapted form Tortora 10 th ed. LECTURE OUTLINE A. Overview of the Nervous System (p. 537) 1. The nervous system and the endocrine system are the body s major control and integrating centers.
I. OVERVIEW DIRECT. Drugs affecting the autonomic nervous system (ANS) are divided into two groups according to the type of
 THE CHOLINERGIC NEURON 1 I. OVERVIEW DIRECT Drugs affecting the autonomic nervous system (ANS) are divided into two groups according to the type of ACTING neuron involved in their mechanism of action.
THE CHOLINERGIC NEURON 1 I. OVERVIEW DIRECT Drugs affecting the autonomic nervous system (ANS) are divided into two groups according to the type of ACTING neuron involved in their mechanism of action.
A NEW TYPE OF DRUG ENHANCEMENT: INCREASED MAXIMUM RESPONSE TO CUMULATIVE NORADREN- ALINE IN THE ISOLATED RAT VAS DEFERENS
 Br. J. Pharmac. Chemother. (1968), 33, 171-176. A NEW TYPE OF DRUG ENHANCEMENT: NCREASED MAXMUM RESPONSE TO CUMULATVE NORADREN- ALNE N THE SOLATED RAT VAS DEFERENS BY A. BARNETT, D. D. GREENHOUSE AND R..
Br. J. Pharmac. Chemother. (1968), 33, 171-176. A NEW TYPE OF DRUG ENHANCEMENT: NCREASED MAXMUM RESPONSE TO CUMULATVE NORADREN- ALNE N THE SOLATED RAT VAS DEFERENS BY A. BARNETT, D. D. GREENHOUSE AND R..
Dopamine as a Synaptic Transmitter and Modulator in Sympathetic Ganglia: A Different Mode of Synaptic Action*
 Proceedings of the National Academy of Sciences Vol. 67, No. 2, pp. 667-673, October 1970 Dopamine as a Synaptic Transmitter and Modulator in Sympathetic Ganglia: A Different Mode of Synaptic Action* Benjamin
Proceedings of the National Academy of Sciences Vol. 67, No. 2, pp. 667-673, October 1970 Dopamine as a Synaptic Transmitter and Modulator in Sympathetic Ganglia: A Different Mode of Synaptic Action* Benjamin
by nervous activity. (Received 7 December 1965) chloralosed cats at rates of or 1-25,tg/min for 10 or 20 min and the infusion.
 J. Physiol. (1966), 187, pp. 51-68 51 With 8 text-figures Printed in Great Britain THE UPTAKE AND RELEASE OF RADIOACTIVE NORADRENALINE BY THE SPLENIC NERVES OF CATS BY J. S. GILLESPIE* AND S. M. KIRPEKAR
J. Physiol. (1966), 187, pp. 51-68 51 With 8 text-figures Printed in Great Britain THE UPTAKE AND RELEASE OF RADIOACTIVE NORADRENALINE BY THE SPLENIC NERVES OF CATS BY J. S. GILLESPIE* AND S. M. KIRPEKAR
The Effect of Cortisone on Cell Proliferation and Migration in Peripheral Nerves undergoing Wallerian degeneration
 The Effect of Cortisone on Cell Proliferation and Migration in Peripheral Nerves undergoing Wallerian by G. A. THOMAS 1 From the Department of Anatomy, Guy's Hospital Medical School, London INTRODUCTION
The Effect of Cortisone on Cell Proliferation and Migration in Peripheral Nerves undergoing Wallerian by G. A. THOMAS 1 From the Department of Anatomy, Guy's Hospital Medical School, London INTRODUCTION
Biology 218 Human Anatomy
 Chapter 20 Adapted form Tortora 10 th ed. LECTURE OUTLINE A. Introduction (p. 632) 1. The autonomic nervous system (ANS) regulates the activity of smooth muscle, cardiac muscle, and certain glands. 2.
Chapter 20 Adapted form Tortora 10 th ed. LECTURE OUTLINE A. Introduction (p. 632) 1. The autonomic nervous system (ANS) regulates the activity of smooth muscle, cardiac muscle, and certain glands. 2.
I. Neural Control of Involuntary Effectors. Chapter 9. Autonomic Motor Nerves. Autonomic Neurons. Autonomic Ganglia. Autonomic Neurons 9/19/11
 Chapter 9 I. Neural Control of Involuntary Effectors The Autonomic Nervous System Lecture PowerPoint Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction or display. Autonomic
Chapter 9 I. Neural Control of Involuntary Effectors The Autonomic Nervous System Lecture PowerPoint Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction or display. Autonomic
THE ACTION OF PHYSOSTIGMINE AND THE DISTRIBUTION OF CHOLINESTERASES IN THE CHICKEN OESOPHAGUS
 Br. J. Phannac. Chemother. (1968), 33, 531-536. THE ACTION OF PHYSOSTIGMINE AND THE DISTRIBUTION OF CHOLINESTERASES IN THE CHICKEN OESOPHAGUS BY A. L. BARTLET AND T. HASSAN From the Department of Veterinary
Br. J. Phannac. Chemother. (1968), 33, 531-536. THE ACTION OF PHYSOSTIGMINE AND THE DISTRIBUTION OF CHOLINESTERASES IN THE CHICKEN OESOPHAGUS BY A. L. BARTLET AND T. HASSAN From the Department of Veterinary
The Nervous System PART A
 7 The Nervous System PART A PowerPoint Lecture Slide Presentation by Jerry L. Cook, Sam Houston University ESSENTIALS OF HUMAN ANATOMY & PHYSIOLOGY EIGHTH EDITION ELAINE N. MARIEB Structural Classification
7 The Nervous System PART A PowerPoint Lecture Slide Presentation by Jerry L. Cook, Sam Houston University ESSENTIALS OF HUMAN ANATOMY & PHYSIOLOGY EIGHTH EDITION ELAINE N. MARIEB Structural Classification
Principles of Anatomy and Physiology
 Principles of Anatomy and Physiology 14 th Edition CHAPTER 15 The Autonomic Nervous System Comparison of Somatic and Autonomic Nervous Systems The somatic nervous system includes both sensory and motor
Principles of Anatomy and Physiology 14 th Edition CHAPTER 15 The Autonomic Nervous System Comparison of Somatic and Autonomic Nervous Systems The somatic nervous system includes both sensory and motor
Composed by Natalia Leonidovna Svintsitskaya, Associate professor of the Chair of Human Anatomy, Candidate of Medicine
 Theoretical background to the study of the autonomic nervous system. Sympathetic and parasympathetic divisions of the autonomic nervous system. Features of the structure, function Composed by Natalia Leonidovna
Theoretical background to the study of the autonomic nervous system. Sympathetic and parasympathetic divisions of the autonomic nervous system. Features of the structure, function Composed by Natalia Leonidovna
THE UPTAKE OF CATECHOL AMINES AT HIGH PERFUSION CONCENTRATIONS IN THE RAT ISOLATED HEART: A NOVEL CATECHOL AMINE UPTAKE PROCESS
 Brit. J. Pharmacol. (1965), 25, 18-33. THE UPTAKE OF CATECHOL AMINES AT HIGH PERFUSION CONCENTRATIONS IN THE RAT ISOLATED HEART: A NOVEL CATECHOL AMINE UPTAKE PROCESS BY L. L. IVERSEN* From the Departments
Brit. J. Pharmacol. (1965), 25, 18-33. THE UPTAKE OF CATECHOL AMINES AT HIGH PERFUSION CONCENTRATIONS IN THE RAT ISOLATED HEART: A NOVEL CATECHOL AMINE UPTAKE PROCESS BY L. L. IVERSEN* From the Departments
ENDOCRINE SYSTEM ENDOCRINE SYSTEM
 Endocrine system consists of organs that produce and secrete hormones "endocrine" = internal secretion into capillaries Hormones carried by the blood to another organ; exert effects Hormones manipulate
Endocrine system consists of organs that produce and secrete hormones "endocrine" = internal secretion into capillaries Hormones carried by the blood to another organ; exert effects Hormones manipulate
Sympathetic Nervous System
 Sympathetic Nervous System Lecture Objectives Review the subdivisions of the nervous system. Review the general arrangement and compare the sympathetic and parasympathetic parts. Describe the following
Sympathetic Nervous System Lecture Objectives Review the subdivisions of the nervous system. Review the general arrangement and compare the sympathetic and parasympathetic parts. Describe the following
8.2. Types of Neurons
 Chapter 8 Nervous Tissue The neuron is the functional and the structural unit of the nervous system. It displays two highly developed physiological traits: 1. Irritability - the capacity to generate a
Chapter 8 Nervous Tissue The neuron is the functional and the structural unit of the nervous system. It displays two highly developed physiological traits: 1. Irritability - the capacity to generate a
CATION AND METABOLIC REQUIREMENTS FOR RETENTION OF METARAMINOL BY RAT UTERINE HORNS
 Br. J. Pharmac. Chemother. (1968), 33, 277-286. CATION AND METABOLIC REQUIREMENTS FOR RETENTION OF METARAMINOL BY RAT UTERINE HORNS BY From the Department of Pharmacology, University of Alberta! Edmonton,
Br. J. Pharmac. Chemother. (1968), 33, 277-286. CATION AND METABOLIC REQUIREMENTS FOR RETENTION OF METARAMINOL BY RAT UTERINE HORNS BY From the Department of Pharmacology, University of Alberta! Edmonton,
NERVOUS SYSTEM 1 CHAPTER 10 BIO 211: ANATOMY & PHYSIOLOGY I
 BIO 211: ANATOMY & PHYSIOLOGY I 1 Ch 10 A Ch 10 B This set CHAPTER 10 NERVOUS SYSTEM 1 BASIC STRUCTURE and FUNCTION Dr. Lawrence G. Altman www.lawrencegaltman.com Some illustrations are courtesy of McGraw-Hill.
BIO 211: ANATOMY & PHYSIOLOGY I 1 Ch 10 A Ch 10 B This set CHAPTER 10 NERVOUS SYSTEM 1 BASIC STRUCTURE and FUNCTION Dr. Lawrence G. Altman www.lawrencegaltman.com Some illustrations are courtesy of McGraw-Hill.
The influence of light and dark on the catecholamine content of the retina and choroid. Charles W. Nichols, David Jacobowitz, and Marianne Hottenstein
 The influence of light and dark on the catecholamine content of the retina and choroid Charles W. Nichols, David Jacobowitz, and Marianne Hottenstein Recent histochemical studies with the use of a fluorescence
The influence of light and dark on the catecholamine content of the retina and choroid Charles W. Nichols, David Jacobowitz, and Marianne Hottenstein Recent histochemical studies with the use of a fluorescence
Screening and bioassay of Sympatholytics. Dr. Magdy M. Awny Lecture 4
 Screening and bioassay of Sympatholytics by Dr. Magdy M. Awny Lecture 4 1 They are classified into: Sympatholytics = Antagonist of adrenergic activity Drugs that interfere with the activity of the sympathetic
Screening and bioassay of Sympatholytics by Dr. Magdy M. Awny Lecture 4 1 They are classified into: Sympatholytics = Antagonist of adrenergic activity Drugs that interfere with the activity of the sympathetic
Neuropsychiatry Block
 Neuropsychiatry Block Physiology of the Autonomic Nervous System By Laiche Djouhri, PhD Dept. of Physiology Email: ldjouhri@ksu.edu.sa Ext:71044 References The Autonomic Nervous System and the Adrenal
Neuropsychiatry Block Physiology of the Autonomic Nervous System By Laiche Djouhri, PhD Dept. of Physiology Email: ldjouhri@ksu.edu.sa Ext:71044 References The Autonomic Nervous System and the Adrenal
EFFECT OF MUSCARINE ON RELEASE OF CATECHOLAMINES
 Br. J. Pharmac. (1982), 77, 455-46 EFFECT OF MUSCARINE ON RELEASE OF CATECHOLAMINES FROM THE PERFUSED ADRENAL GLAND OF THE CAT S.M. KIRPEKAR, J.C. PRAT & M.T. SCHIAVONE Department of Pharmacology, Downstate
Br. J. Pharmac. (1982), 77, 455-46 EFFECT OF MUSCARINE ON RELEASE OF CATECHOLAMINES FROM THE PERFUSED ADRENAL GLAND OF THE CAT S.M. KIRPEKAR, J.C. PRAT & M.T. SCHIAVONE Department of Pharmacology, Downstate
Nervous Tissue and Histology of CNS
 Nervous Tissue and Histology of CNS Functions of Nervous System Like the CPU of a computer, the nervous system is the master controlling system of the body. It is designed to constantly and rapidly adjust
Nervous Tissue and Histology of CNS Functions of Nervous System Like the CPU of a computer, the nervous system is the master controlling system of the body. It is designed to constantly and rapidly adjust
The Nervous System: Autonomic Nervous System Pearson Education, Inc.
 17 The Nervous System: Autonomic Nervous System Introduction The autonomic nervous system: Functions outside of our conscious awareness Makes routine adjustments in our body s systems The autonomic nervous
17 The Nervous System: Autonomic Nervous System Introduction The autonomic nervous system: Functions outside of our conscious awareness Makes routine adjustments in our body s systems The autonomic nervous
Ultrastructural Contributions to Desensitization at the Cerebellar Mossy Fiber to Granule Cell Synapse
 Ultrastructural Contributions to Desensitization at the Cerebellar Mossy Fiber to Granule Cell Synapse Matthew A.Xu-Friedman and Wade G. Regehr Department of Neurobiology, Harvard Medical School, Boston,
Ultrastructural Contributions to Desensitization at the Cerebellar Mossy Fiber to Granule Cell Synapse Matthew A.Xu-Friedman and Wade G. Regehr Department of Neurobiology, Harvard Medical School, Boston,
Chapter 7 Nerve tissue 1 Liu Jiamei
 Chapter 7 Nerve tissue 1 Liu Jiamei General description: nerve tissue nerve cells (neurons): show numerous long processes receive the stimulation make contact with each other, conduct the nerve impulse
Chapter 7 Nerve tissue 1 Liu Jiamei General description: nerve tissue nerve cells (neurons): show numerous long processes receive the stimulation make contact with each other, conduct the nerve impulse
NERVOUS SYSTEM CELLS. a. afferent division CHAPTER 12 ORGANIZATION OF THE NERVOUS SYSTEM. Student Name
 Student Name CHAPTER 12 NERVOUS SYSTEM CELLS T he nervous system organizes and coordinates the millions of impulses received each day to make communication with and enjoyment of our environment possible.
Student Name CHAPTER 12 NERVOUS SYSTEM CELLS T he nervous system organizes and coordinates the millions of impulses received each day to make communication with and enjoyment of our environment possible.
9.01 Introduction to Neuroscience Fall 2007
 MIT OpenCourseWare http://ocw.mit.edu 9.01 Introduction to Neuroscience Fall 2007 For information about citing these materials or our Terms of Use, visit: http://ocw.mit.edu/terms. 9.01 Recitation (R02)
MIT OpenCourseWare http://ocw.mit.edu 9.01 Introduction to Neuroscience Fall 2007 For information about citing these materials or our Terms of Use, visit: http://ocw.mit.edu/terms. 9.01 Recitation (R02)
The Nervous System. Nervous System Functions 1. gather sensory input 2. integration- process and interpret sensory input 3. cause motor output
 The Nervous System Nervous System Functions 1. gather sensory input 2. integration- process and interpret sensory input 3. cause motor output The Nervous System 2 Parts of the Nervous System 1. central
The Nervous System Nervous System Functions 1. gather sensory input 2. integration- process and interpret sensory input 3. cause motor output The Nervous System 2 Parts of the Nervous System 1. central
EFFECT OF DENERVATION AND OF COCAINE ON THE ACTION OF SYMPATHOMIMETIC AMINES
 Brit. J. Pharmacol. (1960), 15, 328. EFFECT OF DENERVATION AND OF COCAINE ON THE ACTION OF SYMPATHOMIMETIC AMINES BY B. C. R. STROMBLAD From the Institute of Physiology, Lund, Sweden (RECEIVED FEBRUARY
Brit. J. Pharmacol. (1960), 15, 328. EFFECT OF DENERVATION AND OF COCAINE ON THE ACTION OF SYMPATHOMIMETIC AMINES BY B. C. R. STROMBLAD From the Institute of Physiology, Lund, Sweden (RECEIVED FEBRUARY
Nervous system. Dr. Rawaa Salim Hameed
 Nervous system Dr. Rawaa Salim Hameed Central nervous system (CNS) CNS consists of the brain (cerebrum, cerebellum, and brainstem) and spinal cord CNS is covered by connective tissue layers, the meninges
Nervous system Dr. Rawaa Salim Hameed Central nervous system (CNS) CNS consists of the brain (cerebrum, cerebellum, and brainstem) and spinal cord CNS is covered by connective tissue layers, the meninges
Autonomic Nervous System. Lanny Shulman, O.D., Ph.D. University of Houston College of Optometry
 Autonomic Nervous System Lanny Shulman, O.D., Ph.D. University of Houston College of Optometry Peripheral Nervous System A. Sensory Somatic Nervous System B. Autonomic Nervous System 1. Sympathetic Nervous
Autonomic Nervous System Lanny Shulman, O.D., Ph.D. University of Houston College of Optometry Peripheral Nervous System A. Sensory Somatic Nervous System B. Autonomic Nervous System 1. Sympathetic Nervous
The Autonomic Nervous
 Autonomic Nervous System The Autonomic Nervous Assess Prof. Fawzia Al-Rouq System Department of Physiology College of Medicine King Saud University LECTUR (1) Functional Anatomy & Physiology of Autonomic
Autonomic Nervous System The Autonomic Nervous Assess Prof. Fawzia Al-Rouq System Department of Physiology College of Medicine King Saud University LECTUR (1) Functional Anatomy & Physiology of Autonomic
Nervous System Dr. Naim Kittana Department of Biomedical Sciences Faculty of Medicine & Health Sciences An-Najah National University
 Nervous System Department of Biomedical Sciences Faculty of Medicine & Health Sciences An-Najah National University Declaration The content and the figures of this seminar were directly adopted from the
Nervous System Department of Biomedical Sciences Faculty of Medicine & Health Sciences An-Najah National University Declaration The content and the figures of this seminar were directly adopted from the
LOCALIZATION OF SODIUM CHANNELS IN CULTURED NEURAL CELLS
 0270~6474/81/0107-0777$02.00/O Copyright 0 Society for Neuroscience Printed in U.S.A. The Journal of Neuroscience Vol. 1, No. 7, pp. 777-783 July 1981 LOCALIZATION OF SODIUM CHANNELS IN CULTURED NEURAL
0270~6474/81/0107-0777$02.00/O Copyright 0 Society for Neuroscience Printed in U.S.A. The Journal of Neuroscience Vol. 1, No. 7, pp. 777-783 July 1981 LOCALIZATION OF SODIUM CHANNELS IN CULTURED NEURAL
THE UPTAKE OF CATECHOL AMINES AT HIGH PERFUSION CONCENTRATIONS IN THE RAT ISOLATED HEART: A NOVEL CATECHOL AMINE UPTAKE PROCESS
 Brit. J. Pharmacol. (1965), 25, 18-33. THE UPTAKE OF CATECHOL AMINES AT HIGH PERFUSION CONCENTRATIONS IN THE RAT ISOLATED HEART: A NOVEL CATECHOL AMINE UPTAKE PROCESS BY L. L. IVERSEN* From the Departments
Brit. J. Pharmacol. (1965), 25, 18-33. THE UPTAKE OF CATECHOL AMINES AT HIGH PERFUSION CONCENTRATIONS IN THE RAT ISOLATED HEART: A NOVEL CATECHOL AMINE UPTAKE PROCESS BY L. L. IVERSEN* From the Departments
Autonomic Nervous System (ANS):
 Autonomic Nervous System (ANS): ANS is the major involuntary, unconscious, automatic portion of the nervous system. involuntary voluntary The motor (efferent)portion of the ANS is the major pathway for
Autonomic Nervous System (ANS): ANS is the major involuntary, unconscious, automatic portion of the nervous system. involuntary voluntary The motor (efferent)portion of the ANS is the major pathway for
NURSE-UP INTRODUCTION TO THE NERVOUS SYSTEM
 NURSE-UP INTRODUCTION TO THE NERVOUS SYSTEM FUNCTIONS OF THE NERVOUS SYSTEM Body s primary communication and control system. Integrates and regulates body function Collects information specialized nervous
NURSE-UP INTRODUCTION TO THE NERVOUS SYSTEM FUNCTIONS OF THE NERVOUS SYSTEM Body s primary communication and control system. Integrates and regulates body function Collects information specialized nervous
periphery of the cells, whereas in the smooth muscle of arteries the fluorescence was
 Br. J. Pharmac. Chemother. (1968), 32, 168-184. UPTAKE OF NORADRENALINE BY ADRENERGIC NERVES, SMOOTH MUSCLE AND CONNECTIVE TISSUE IN ISOLATED PERFUSED ARTERIES AND ITS CORRELATION WITH THE VASOCONSTRICTOR
Br. J. Pharmac. Chemother. (1968), 32, 168-184. UPTAKE OF NORADRENALINE BY ADRENERGIC NERVES, SMOOTH MUSCLE AND CONNECTIVE TISSUE IN ISOLATED PERFUSED ARTERIES AND ITS CORRELATION WITH THE VASOCONSTRICTOR
Autonomic Nervous System
 Autonomic Nervous System Autonomic nervous system organization Sympathetic Nervous System division of the autonomic nervous system that arouses the body, mobilizing its energy in stressful situations
Autonomic Nervous System Autonomic nervous system organization Sympathetic Nervous System division of the autonomic nervous system that arouses the body, mobilizing its energy in stressful situations
Autonomic Nervous System. Introduction
 Autonomic Nervous System Introduction 1 The nervous system is divided into: 1- the central nervous system (CNS; the brain and spinal cord) 2- the peripheral nervous system (PNS; neuronal tissues outside
Autonomic Nervous System Introduction 1 The nervous system is divided into: 1- the central nervous system (CNS; the brain and spinal cord) 2- the peripheral nervous system (PNS; neuronal tissues outside
The removal of noradrenaline in the pulmonary circulation of rat isolated lungs
 Br. J. Pharmac. (1973), 47, 325-331. The removal of noradrenaline in the pulmonary circulation of rat isolated lungs VALERIE A. ALABASTER AND Y. S. BAKHLE Departtent of Pharmacology, Institute of Basic
Br. J. Pharmac. (1973), 47, 325-331. The removal of noradrenaline in the pulmonary circulation of rat isolated lungs VALERIE A. ALABASTER AND Y. S. BAKHLE Departtent of Pharmacology, Institute of Basic
The Nervous System. Functions of the Nervous System input gathering To monitor occurring inside and outside the body Changes =
 The Nervous System Functions of the Nervous System input gathering To monitor occurring inside and outside the body Changes = To process and sensory input and decide if is needed output A response to integrated
The Nervous System Functions of the Nervous System input gathering To monitor occurring inside and outside the body Changes = To process and sensory input and decide if is needed output A response to integrated
DEPLETION AND REPLACEMENT OF THE ADRENALINE AND NORADRENALINE CONTENTS OF THE RAT ADRENAL GLAND, FOLLOWING TREATMENT WITH RESERPINE
 Brit. J. Pharmacol. (1962), 18, 138-149. DEPLETION AND REPLACEMENT OF THE ADRENALINE AND NORADRENALINE CONTENTS OF THE RAT ADRENAL GLAND, FOLLOWING TREATMENT WITH RESERPINE BY B. A. CALLINGHAM AND MONICA
Brit. J. Pharmacol. (1962), 18, 138-149. DEPLETION AND REPLACEMENT OF THE ADRENALINE AND NORADRENALINE CONTENTS OF THE RAT ADRENAL GLAND, FOLLOWING TREATMENT WITH RESERPINE BY B. A. CALLINGHAM AND MONICA
Autonomic nervous system
 Autonomic nervous system Key notes Autonomic: an independent system that runs on its own The ANS is a visceral and involuntary sensory and motor system The visceral motor fibers in the autonomic nerves
Autonomic nervous system Key notes Autonomic: an independent system that runs on its own The ANS is a visceral and involuntary sensory and motor system The visceral motor fibers in the autonomic nerves
Major Structures of the Nervous System. Brain, cranial nerves, spinal cord, spinal nerves, ganglia, enteric plexuses and sensory receptors
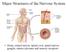 Major Structures of the Nervous System Brain, cranial nerves, spinal cord, spinal nerves, ganglia, enteric plexuses and sensory receptors Nervous System Divisions Central Nervous System (CNS) consists
Major Structures of the Nervous System Brain, cranial nerves, spinal cord, spinal nerves, ganglia, enteric plexuses and sensory receptors Nervous System Divisions Central Nervous System (CNS) consists
THE LACK OF CROSSED TACHYPHYLAXIS BETWEEN TYRAMINE AND SOME OTHER INDIRECTLY ACTING SYMPATHOMIMETIC AMINES
 Br. J. Pharmac. Chenother. (1967). 30, 631-643. THE LACK OF CROSSED TACHYPHYLAXIS BETWEEN TYRAMINE AND SOME OTHER INDIRECTLY ACTING SYMPATHOMIMETIC AMINES BY M. D. DAY* From the Pharmacology Laboratories,
Br. J. Pharmac. Chenother. (1967). 30, 631-643. THE LACK OF CROSSED TACHYPHYLAXIS BETWEEN TYRAMINE AND SOME OTHER INDIRECTLY ACTING SYMPATHOMIMETIC AMINES BY M. D. DAY* From the Pharmacology Laboratories,
Franklin, 1933; Waterman, 1933]; indeed, the only negative findings, [Waterman, 1933]. Inasmuch, then, as Donegan was misled with
![Franklin, 1933; Waterman, 1933]; indeed, the only negative findings, [Waterman, 1933]. Inasmuch, then, as Donegan was misled with Franklin, 1933; Waterman, 1933]; indeed, the only negative findings, [Waterman, 1933]. Inasmuch, then, as Donegan was misled with](/thumbs/91/106294335.jpg) 381 6I2.I34:6I2.893 THE CONSTRICTOR RESPONSE OF THE INFERIOR VENA CAVA TO STIMULATION OF THE SPLANCHNIC NERVE BY K. J. FRANKLIN AND A. D. McLACHLIN (From the University Department of Pharmacology, Oxford)
381 6I2.I34:6I2.893 THE CONSTRICTOR RESPONSE OF THE INFERIOR VENA CAVA TO STIMULATION OF THE SPLANCHNIC NERVE BY K. J. FRANKLIN AND A. D. McLACHLIN (From the University Department of Pharmacology, Oxford)
however, reduced after parasympathetic denervation [Nordenfelt et al., 1960]. opposite to those caused by parasympathetic denervation.
![however, reduced after parasympathetic denervation [Nordenfelt et al., 1960]. opposite to those caused by parasympathetic denervation. however, reduced after parasympathetic denervation [Nordenfelt et al., 1960]. opposite to those caused by parasympathetic denervation.](/thumbs/90/101412039.jpg) CHOLINE ACETYLASE IN SALIVARY GLANDS OF THE CAT AFTER SYMPATHETIC DENERVATION. By IVAR NORDENFELT. From the Institute of Physiology, University of Lund, Sweden. (Received for publication 20th April 1964)
CHOLINE ACETYLASE IN SALIVARY GLANDS OF THE CAT AFTER SYMPATHETIC DENERVATION. By IVAR NORDENFELT. From the Institute of Physiology, University of Lund, Sweden. (Received for publication 20th April 1964)
Chapter 16. APR Enhanced Lecture Slides
 Chapter 16 APR Enhanced Lecture Slides See separate PowerPoint slides for all figures and tables pre-inserted into PowerPoint without notes and animations. Copyright The McGraw-Hill Companies, Inc. Permission
Chapter 16 APR Enhanced Lecture Slides See separate PowerPoint slides for all figures and tables pre-inserted into PowerPoint without notes and animations. Copyright The McGraw-Hill Companies, Inc. Permission
Development of the myelin sheath of the hypogastric nerves in a human foetus aged 23 weeks
 O R I G I N A L A R T I C L E Folia Morphol. Vol. 63, No. 3, pp. 289 301 Copyright 2004 Via Medica ISSN 0015 5659 www.fm.viamedica.pl Development of the myelin sheath of the hypogastric nerves in a human
O R I G I N A L A R T I C L E Folia Morphol. Vol. 63, No. 3, pp. 289 301 Copyright 2004 Via Medica ISSN 0015 5659 www.fm.viamedica.pl Development of the myelin sheath of the hypogastric nerves in a human
Human Anatomy. Autonomic Nervous System
 Human Anatomy Autonomic Nervous System 1 Autonomic Nervous System ANS complex system of nerves controls involuntary actions. Works with the somatic nervous system (SNS) regulates body organs maintains
Human Anatomy Autonomic Nervous System 1 Autonomic Nervous System ANS complex system of nerves controls involuntary actions. Works with the somatic nervous system (SNS) regulates body organs maintains
Chapter 17. Nervous System Nervous systems receive sensory input, interpret it, and send out appropriate commands. !
 Chapter 17 Sensory receptor Sensory input Integration Nervous System Motor output Brain and spinal cord Effector cells Peripheral nervous system (PNS) Central nervous system (CNS) 28.1 Nervous systems
Chapter 17 Sensory receptor Sensory input Integration Nervous System Motor output Brain and spinal cord Effector cells Peripheral nervous system (PNS) Central nervous system (CNS) 28.1 Nervous systems
Experimental Neoplastic Formation in Embryonic Chick Brains
 Experimental Neoplastic Formation in Embryonic Chick Brains by BENGT KALLEN 1 From the Tornblad Institute of Comparative Embryology, Lund WITH TWO PLATES IN mammalian teratology, a malformation consisting
Experimental Neoplastic Formation in Embryonic Chick Brains by BENGT KALLEN 1 From the Tornblad Institute of Comparative Embryology, Lund WITH TWO PLATES IN mammalian teratology, a malformation consisting
LOCALIZATION OF SUBSTANCE P-IMMUNOREACTIVITY IN THE DEVELOPING HUMAN URINARY BLADDER
 ACTA HISTOCHEM. CYTOCHEM. Vol. 21, No. 2, 1988 LOCALIZATION OF SUBSTANCE P-IMMUNOREACTIVITY IN THE DEVELOPING HUMAN URINARY BLADDER SHASHI WADHWA AND VEENA BIJLANI Department of Anatomy, All-India Institute
ACTA HISTOCHEM. CYTOCHEM. Vol. 21, No. 2, 1988 LOCALIZATION OF SUBSTANCE P-IMMUNOREACTIVITY IN THE DEVELOPING HUMAN URINARY BLADDER SHASHI WADHWA AND VEENA BIJLANI Department of Anatomy, All-India Institute
Muscle Tissue. General concepts. Classification of muscle. I. Functional classification is based on the type of neural control.
 Muscle Tissue LEARNING OBJECTIVES 1. Identify the three types of muscle tissue at the light microscopic level. 2. List and compare the structural and functional features of each of the three muscle fiber
Muscle Tissue LEARNING OBJECTIVES 1. Identify the three types of muscle tissue at the light microscopic level. 2. List and compare the structural and functional features of each of the three muscle fiber
Tyrosinase Activity in the Pigmented Cells of the Nucleus Substantiae Nigrae. I. Monophenolase and Diphenolase Activity. By C. D.
 4 7 Tyrosinase Activity in the Pigmented Cells of the Nucleus Substantiae Nigrae. I. Monophenolase and Diphenolase Activity By C. D. MARSDEN (From the Department of Anatomy, St. Thomas's Hospital Medical
4 7 Tyrosinase Activity in the Pigmented Cells of the Nucleus Substantiae Nigrae. I. Monophenolase and Diphenolase Activity By C. D. MARSDEN (From the Department of Anatomy, St. Thomas's Hospital Medical
The Egyptian journal of hospital medicine (2009)vol.,36:
 The Egyptian journal of hospital medicine (2009)vol.,36: 468-482 QUANTITATIVE AND HISTOCHEMICAL STUDY ON THE ADRENAL MEDULLA IN POST NATAL PERIOD OF ALBINO RAT Wagieh k.baiomy; Abdel-mawgood Anas; Mamdooh
The Egyptian journal of hospital medicine (2009)vol.,36: 468-482 QUANTITATIVE AND HISTOCHEMICAL STUDY ON THE ADRENAL MEDULLA IN POST NATAL PERIOD OF ALBINO RAT Wagieh k.baiomy; Abdel-mawgood Anas; Mamdooh
Autonomic Division of NS
 Autonomic Division of NS Compare and contrast the structures of the sympathetic and the parasympathetic divisions, including functions and neurotransmitters. Show the levels of integration in the ANS,
Autonomic Division of NS Compare and contrast the structures of the sympathetic and the parasympathetic divisions, including functions and neurotransmitters. Show the levels of integration in the ANS,
Yara Saddam. Amr Alkhatib. Ihsan
 1 Yara Saddam Amr Alkhatib Ihsan NOTE: Yellow highlighting=correction/addition to the previous version of the sheet. Histology (micro anatomy) :- the study of tissues and how they are arranged into organs.
1 Yara Saddam Amr Alkhatib Ihsan NOTE: Yellow highlighting=correction/addition to the previous version of the sheet. Histology (micro anatomy) :- the study of tissues and how they are arranged into organs.
THE EFFECT OF ESERINE ON THE RESPONSE OF THE VAS DEFERENS TO HYPOGASTRIC NERVE STIMULATION
 Brit. J. Pharmacol. (1963), 20, 74-82. THE EFFECT OF ESERINE ON THE RESPONSE OF THE VAS DEFERENS TO HYPOGASTRIC NERVE STIMULATION BY J. H. BURN AND D. F. WEETMAN From the Biological Research Laboratories,
Brit. J. Pharmacol. (1963), 20, 74-82. THE EFFECT OF ESERINE ON THE RESPONSE OF THE VAS DEFERENS TO HYPOGASTRIC NERVE STIMULATION BY J. H. BURN AND D. F. WEETMAN From the Biological Research Laboratories,
EM: myelin sheath shows a series of concentrically arranged lamellae
 EM: myelin sheath shows a series of concentrically arranged lamellae ---- how to form myelin sheath? Schwann cell invagination and envelop the axon form mesaxon mesaxon become longer and longer winding
EM: myelin sheath shows a series of concentrically arranged lamellae ---- how to form myelin sheath? Schwann cell invagination and envelop the axon form mesaxon mesaxon become longer and longer winding
Do Now pg What is the fight or flight response? 2. Give an example of when this response would kick in.
 Do Now pg 81 1. What is the fight or flight response? 2. Give an example of when this response would kick in. Autonomic Nervous System The portion of the PNS that functions independently (autonomously)
Do Now pg 81 1. What is the fight or flight response? 2. Give an example of when this response would kick in. Autonomic Nervous System The portion of the PNS that functions independently (autonomously)
