Effect of Attentive Fixation in Macaque Thalamus and Cortex
|
|
|
- Clifton Davidson
- 5 years ago
- Views:
Transcription
1 Effect of Attentive Fixation in Macaque Thalamus and Cortex D. B. BENDER AND M. YOUAKIM Department of Physiology and Biophysics, School of Medicine and Biomedical Sciences, University at Buffalo, State University of New York, Buffalo, New York Received 29 December 1999; accepted in final form 21 September 2000 Bender, D. B. and M. Youakim. Effect of attentive fixation in macaque thalamus and cortex. J Neurophysiol 85: , Attentional modulation of neuronal responsiveness is common in many areas of visual cortex. We examined whether attentional modulation in the visual thalamus was quantitatively similar to that in cortex. Identical procedures and apparatus were used to compare attentional modulation of single neurons in seven different areas of the visual system: the lateral geniculate, three visual subdivisions of the pulvinar [inferior, lateral, dorsomedial part of lateral pulvinar (Pdm)], and three areas of extrastriate cortex representing early, intermediate, and late stages of cortical processing (V2, V4/PM, area 7a). A simple fixation task controlled transitions among three attentive states. The animal waited for a fixation point to appear (ready state), fixated the point until it dimmed (fixation state), and then waited idly to begin the next trial (idle state). Attentional modulation was estimated by flashing an identical, irrelevant stimulus in a neuron s receptive field during each of the three states; the three responses defined a response vector whose deviation from the line of equal response in all three states (the main diagonal) indicated the character and magnitude of attentional modulation. Attentional modulation was present in all visual areas except the lateral geniculate, indicating that modulation was of central origin. Prevalence of modulation was modest (26%) in pulvinar, and increased from 21% in V2 to 43% in 7a. Modulation had a push-pull character (as many cells facilitated as suppressed) with respect to the fixation state in all areas except Pdm where all cells were suppressed during fixation. The absolute magnitude of attentional modulation, measured by the angle between response vector and main diagonal expressed as a percent of the maximum possible angle, differed among brain areas. Magnitude of modulation was modest in the pulvinar (19 26%), and increased from 22% in V2 to 41% in 7a. However, average trial-to-trial variability of response, measured by the coefficient of variation, also increased across brain areas so that its difference among areas accounted for more than 90% of the difference in modulation magnitude among areas. We also measured attentional modulation by the ratio of cell discharge due to attention divided by discharge variability. The resulting signal-tonoise ratio of attention was small and constant, %, across all areas of pulvinar and cortex. We conclude that the pulvinar, but not the lateral geniculate, is as strongly affected by attentional state as any area of visual cortex we studied and that attentional modulation amplitude is closely tied to intrinsic variability of response. INTRODUCTION It is now clear that attention can affect the responsiveness of neurons throughout visual cortex. Visually responsive cortex includes a number of distinct areas beyond striate cortex, or V1. Beginning with V2, these extrastriate areas are organized into two partially segregated, roughly hierarchical systems Address for reprint requests: D. B. Bender, Dept. of Physiology and Biophysics, 329 Cary Hall, SUNY/AB Medical School, Buffalo, NY (reviews in Felleman and Van Essen 1991; Maunsell and Newsome 1987; Ungerleider and Mishkin 1982; Van Essen 1985). One includes dorsally located areas such as V3A, MT, and MST and leads into area 7a in the inferior parietal lobule. The other includes more ventrally located areas such as V4 and TEO and leads into area TE in the temporal lobe. Recordings from single neurons in many of these areas show that neuronal excitability depends on the animal s attentive state (reviews in Colby 1991; Desimone and Duncan 1995; Lock and Bender 1999; Maunsell 1995; Motter 1998). Typically the effect of attention is modest: a small increase or decrease in magnitude of response to a visual stimulus relative to a control condition. Such modulation can be found at virtually every level of the cortical hierarchy, including V1. A variety of behavioral paradigms have been used to manipulate attention, and these show that the prevalence and magnitude of attentional modulation can depend substantially on both the behavioral paradigm and the cortical area in which its effects are measured. Furthermore factors such as task difficulty, the extent to which a task engages the functions of an area, and whether multiple stimuli compete for attention all can affect the modulation (Luck et al. 1997; Motter 1993; Richmond and Sato 1987). To what extent does the thalamus contribute to, or participate in, the attentional modulation that is so widespread throughout visual cortex? Three thalamic nuclei are closely interrelated with visual cortex: the lateral geniculate nucleus, the pulvinar, and the reticular nucleus of the thalamus. All have been thought to be involved in one form of attention or another (e.g., Guillery et al. 1998; Koch and Ullman 1985; Olshausen et al. 1993). The lateral geniculate projects almost exclusively to V1 with little or no output to extrastriate cortex. Layer 6 of both extrastriate and striate cortex project back to the geniculate, potentially modulating transmission through it. The pulvinar has at least three distinct visual subdivisions. The inferior (PI) and lateral pulvinar (PL) contain two separate visuotopic maps (Bender 1981). PI is driven by input from V1 (Bender 1983) but also receives input from extrastriate cortex and the superior colliculus. It projects mainly to V2, V3, V3A, and MT. PL likewise receives input from V1 and extrastriate cortex, but may have a particular affinity for V2: both areas have the distinctive visuotopic organization that is characterized by a second-order transformation of the hemifield (Allman and Kaas 1974; Bender 1981). PL projects extensively to more The costs of publication of this article were defrayed in part by the payment of page charges. The article must therefore be hereby marked advertisement in accordance with 18 U.S.C. Section 1734 solely to indicate this fact /01 $5.00 Copyright 2001 The American Physiological Society 219
2 220 D. B. BENDER AND M. YOUAKIM ventral areas of extrastriate cortex, including V4, TEO, and TE. Both PI and PL could thus influence, or be influenced by, attentional modulation in the early and intermediate stages of the cortical hierarchy. A third area, the dorsomedial portion of the lateral pulvinar, is visually responsive but its visuotopic organization has not yet been determined (Bender 1981). Referred to as Pdm (Petersen et al. 1985), it has connections with area 7a (Asanuma et al. 1985; Baleydier and Morel 1992) and is thus more closely related to the highest level of the dorsal pathway. The reticular nucleus of the thalamus is driven both by descending input from striate and extrastriate cortex and by branches of ascending thalamocortical axons from geniculate and pulvinar. It projects not to cortex but rather back on pulvinar and geniculate, providing an indirect route by which cortex potentially could modulate excitability in these nuclei (reviews in Guillery et al. 1998; Mitrofanis and Guillery 1993). There is thus ample opportunity for thalamic involvement in the attentional modulation found in cortex. However, evidence of that involvement has been ambiguous. For the pulvinar, glucose utilization in human subjects and reversible inactivation in monkeys both suggest a role in attention (Desimone et al. 1990; LaBerge and Buchsbaum 1990), but lesions of the pulvinar generally do not. Pulvinar lesions do not impair visual search, and those deficits that have been found with attentiondependent tasks (e.g., Chalupa et al. 1976; Ogren et al. 1984; Ungerleider and Christensen 1979) either have not been replicated or may have resulted from damage to neighboring structures such as the corticotectal tract (Bender and Baizer 1984, 1990; Bender and Butter 1987; Nagel-Leiby et al. 1984). Attentional modulation of single neurons has been found in Pdm (Petersen et al. 1985), but there has been no quantitative comparison with neuronal modulation in cortex. For the lateral geniculate, there has been little attempt to find attentional modulation like that in cortex at the single neuron level. Our main goal in this study was thus to see whether attentional modulation like that in cortex was detectable in single neurons of the lateral geniculate and pulvinar and, if so, to compare its prevalence and strength with modulation in cortex. Because attentional modulation can differ among cortical areas, it was important to compare across different levels of the cortical hierarchy as well as across all three visual subdivisions of the pulvinar. Looking at an object and fixating it is an elemental act of attention. To manipulate attention, we used a simple fixation task that required an animal to wait for a spot of light to appear, look at the spot until it dimmed, and then wait idly for a chance to begin the next trial. We estimated attentional modulation by comparing responses to another stimulus, irrelevant to the animal, flashed in a neuron s receptive field during each of those three states. Although simple, this widely used fixation task has the advantage that all animals were likely to find it equally easy and perform it in similar fashion. Further, the stimulus used to probe cell excitability was not itself an object of attention, thereby avoiding the potential confounding of receptive-field selectivity and attentional specificity. Such a fundamental behavior as fixation can generate strong attentional modulation, and with a character that differs, it has been argued, among cortical areas. In area 7a, for example, responses are typically threefold larger during fixation than when waiting for the fixation point to appear or performing no task at all (Mountcastle et al. 1981). In area V4, by contrast, responses during fixation are the same as when waiting for the fixation point (Mountcastle et al. 1987), and in area TE, responses are suppressed during fixation (Richmond et al. 1983). It was thus also of interest to see which cortical pattern of modulation thalamic modulation might resemble. For comparison we chose cortical areas V2, V4, and 7a. As a group, they span the cortical hierarchy and are easily accessible from a single recording cylinder. The choice also permitted an independent evaluation of previous findings in V4 and 7a (Mountcastle et al. 1981, 1987). In this study, we found that almost all brain areas had a push-pull form of modulation in which some cells were facilitated and others were suppressed during fixation. Allowing for that, and the difference among brain areas in response variability, we found the strength of modulation was about the same in all areas of both pulvinar and cortex. METHODS Animal preparation and procedures Eight juvenile male macaques weighing between 3.5 and 7.3 kg were used. Naive animals were first trained on the fixation tasks described in the following text. Standard aseptic technique was used to implant the skull with hardware for recording and painless fixation of the head. A subscleral, magnetic-field search-coil was also implanted in one eye (Judge et al. 1980) for monitoring eye position. For surgery, animals were anesthetized with halothane in 70% N 2 O-O 2, and rectal temperature, electrocardiogram (EKG), and respiration were continually monitored. Penicillin G was given as prophylaxis against infection. All surgical procedures and animal use protocols were reviewed and approved by the Institutional Animal Care and Use Committee. Animals continued training for 3 8 mo with head fixed until performance was consistently accurate despite repeated and irregular stoppages of the task. Apparatus Stimuli were rear-projected on a Polacoat tangent screen, 57 cm in front of the animal. The screen extended 30 in all directions from straight ahead; beyond that, a black matte surface filled the visible field. A lever and small ready-light (see following text) were mounted in front of the animal at waist level. A 0.2 diam He-Ne laser beam provided the fixation point, and a tungsten-illuminated rectangle of adjustable height, width, orientation, and color served to probe neuronal excitability. Fixation point and probe luminance were controlled by liquid-crystal film shutters with rise and fall times 1 ms and 10 ms, respectively. Maximum probe luminance was 30 cd/m 2, 15 db above background luminance. Independent, temperature-stabilized, servo-controlled, mirror galvanometers positioned the stimuli. Galvanometer and eye position signals were modified by second-order polynomials in screen coordinates so that fixation point, probe, and eye position all tracked to better than 0.2 over the central 15 of the screen. This was verified physiologically by electronically locking a 0.4 square spot relative to the fixation point so that the spot fell on the receptive field of a lateral geniculate nucleus (LGN) neuron and showing that the spot always evoked a response from the cell as the animal tracked the fixation point over the central 15. Behavioral tasks All animals performed a simple fixation task for which we operationally defined three different behavioral states. The animal had to wait for a small fixation point to appear (the ready state), fixate the
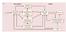
3 ATTENTIVE FIXATION IN PULVINAR AND CORTEX 221 spot and wait for it to dim (the fixation state), and then respond quickly to the dimming to receive reward. There was also a period of idleness (the idle state) during which responding was discouraged witha4stime-out. Neuron excitability was probed by flashing a visual stimulus at the same retinal location, regardless of eye position, during each state. The probe stimulus was irrelevant to the animal in that its appearance was uncorrelated with reward. We used three minor variants of this task, described in the following text, to address potentially troublesome methodological issues in previous studies (Mountcastle et al. 1981, 1987). Those studies had measured excitability for the idle state in a time period different from that for the other two states, potentially introducing an order effect, and there had been no behavioral indication of readiness. Thus in task 1, we randomly interleaved stimulus presentations for all three states on a trial-to-trial basis and required an explicit ready response to define the ready state. Task 2 was virtually identical to that used by Mountcastle. Task 3 eliminated uncertainty in fixation point location, a factor common to both tasks 1 and 2 and the Mountcastle task. In the end, task variants had no effect on the main findings of this study. TASK 1. This task (Fig. 1, top) was used for testing the majority (61%) of cells. A small, dim ready-light turned on to indicate that the animal could start a trial by depressing a lever. Doing so turned off the ready-light and, after a delay (the ready state), turned on the fixation point at a randomly chosen position within 15 of straight ahead. After another delay (the fixation state), the fixation point dimmed and the animal had to release the lever within 0.7 s to receive reward. There followed a 3 9 s idle period (the idle state) after which the ready-light turned on again. On each trial, the probe stimulus was flashed with equal probability during one of the three states. TASK 2. This task (Fig. 1, middle) was used for 31% of the cells, and closely matched the paradigm used by Mountcastle et al. (1987). There was no ready-light, and the idle state was not interleaved with fixation trials. Without warning, the fixation point appeared, and the animal had to depress the lever within 0.7 s to start the trial. After a delay (the fixation state), the fixation point dimmed and the animal had to release the lever within 0.7 s for reward. After another delay (the ready state), the fixation point reappeared at a different location for the next trial. The probe was flashed twice on every trial, once during the fixation state and once during the ready state. Each block of 8 16 such trials was followed by a 1- to 2-min idle period during which the probe was flashed every 5 8 s. The entire sequence, fixation trials followed by idleness, was repeated two to five times to minimize potential order effects. TASK 3. For 8% of the cells, the position of the fixation point was fixed at the center of the tangent screen (Fig. 1, bottom). As in task 1, the ready-light turned on to signal that a trial could be started. Depressing the lever turned it off and started one of two types of trials, each equally likely. Either the fixation point turned on (the fixation state) and then dimmed or there was no fixation point and the animal was simply assumed to be in the ready state. In either case, the probe was flashed within s. There followed a 3- to 9-s idle period during which the probe was flashed with probability 0.5. For all task variants, we imposed constraints to minimize potentially confounding effects of eye and arm movement and to ensure that probe (and eye) positions were comparable across states. The probe was presented only if gaze direction was within 15 of straight ahead, eye position had been stationary for ms, and no trial event (e.g., lever movement, fixation point change) had occurred in the previous 400 ms. Rapid foveation and accurate fixation were encouraged by aborting trials if gaze failed to acquire the fixation point within 350 ms or left it by more than 1 at any time. During the ready and idle states, any saccade during probe presentation immediately extinguished the probe and data were discarded; after training, animals showed no inclination to look in the probe s direction. Recording methods and procedure Epoxy-coated tungsten and glass-coated Pt-Ir microelectrodes were used to record from thalamus and cortex, respectively. To ensure recording from single isolated neurons, waveforms were digitized at 17 khz and matched against a stored template. We studied every reliably isolated cell. The receptive field was plotted if possible, and an optimal probe selected, while the animal fixated. Next the state-dependent modulation of excitability was estimated as the animal ran a block of trials on one of the three tasks described in the preceding text. The same probe was used for the entire block. For LGN cells, the probe was typically 0.8 square so as to always cover the receptive field despite slight galvo positioning errors. Moving stimuli were often used, and these were swept so that receptive-field stimulation was the same despite small positioning errors. These precautions eliminated variations in receptive-field stimulation as a source of response variance. Probe duration was typically 250 ms although durations as short as 150 ms and as long as 300 ms were occasionally used. Gaze direction was used to electronically lock the probe to the retina, thereby providing nearly identical retinal stimulation on every presentation in each behavioral state. FIG. 1. Sequence of events during 3 variants of fixation task. Dashed lines indicate variable length intervals. Stimulus event lines: upward and downward deflections indicate onset and offset, respectively; half-steps indicate dimming. rdy L, ready light; fp, fixation point; probe, visual stimulus flashed in receptive field (vertical displacements of the probe event lines indicate probe appears only once during trial). Data analysis Each cell yielded three histograms of response evoked by the probe, one for each behavioral state. During the ready and idle states, there were frequent eye movements (though rarely toward the probe). When
4 222 D. B. BENDER AND M. YOUAKIM discharge related to those saccades appeared to contaminate a response evoked by the probe, data from that probe presentation were discarded, as were any data for which the eye moved during probe presentation. This often resulted in different numbers of probe presentations per histogram. Only cells with 15 presentations were used; 75% of the cells had 30 presentations. Response magnitude was computed as spikes/second for counts within a time window. For each cell, the window had a fixed width, just wide enough to encompass the largest of the three responses, but 500 ms. The window was positioned for each histogram to maximize counts, thereby eliminating the effect of small differences in response timing that could result from moving stimuli with small positioning errors. Window widths averaged 240 ms in the pulvinar and V2, and slightly longer (290 ms) in the LGN, V4/PM, and area 7a, reflecting the larger proportion of sustained responses in those areas. A measure of spontaneous, i.e., ongoing, discharge was defined similarly, except the window always ended just prior to stimulus onset. We did not subtract ongoing from stimulus-evoked discharge (see DISCUSSION). Only cells for which the response exceeded ongoing discharge in at least one behavioral state (t-test, P 0.01) were used. To determine whether behavioral state affected responses, a one-way repeatedmeasures ANOVA with three levels of state was done on the response measures (P 0.02). We tested all cells for inhomogeneity of variance among states (Bartlett s test) because an inhomogeneity can lead to false positives. Few cells showed it (8%, P 0.02), and when such inhomogeneity was found it clearly could not have led to false positives. Inspection of the individual histograms for such cells showed that the largest effects of attention naturally led to greater inhomogeneity: at least one attentional state necessarily had a small response, and thus small variance, producing inhomogeneity. For neurons significantly affected by behavioral state, we explored that effect using an analysis that gave equal weight to all three states. Each cell with a significant ANOVA was represented by a single vector, r, in a three-dimensional space whose axes represented response strength (in spikes/s) in the ready, fixation, and idle state. The deviation of that vector from the main diagonal of the space (i.e., the locus of equal response, and thus of no attentional modulation) preserved all information about the differential effect of the three states on excitability. This approach, which differs from that of Mountcastle et al. (1981, 1987) who compared responses in only two states at a time, was needed because no single pair of states was sufficient to characterize state-dependent excitability for all brain areas. Proportions were compared using 2 or Fisher s exact probability test. Unless otherwise noted, magnitudes were compared using the Mann-Whitney U test or Kruskal-Wallis test. In the latter case, z scores based on within-group ranks were used to interpret inter-group differences. Histological procedures At the end of recording, marking penetrations were made to aid in reconstruction of recording locations. Frozen sections were cut in the coronal plane at 50 and stained with cresyl-echt. The cut surface of the brain was photographed every 500 as an aid to reconstruction of the cortical surface. Cortical recording sites were located on a reconstruction of the cortical surface made from the section photographs (Fig. 2A). Sites were assigned to area 7a, the dorsal part of the exposed prelunate gyrus, or area V2, based on the stained sections. Sites on the prelunate gyrus extended from a point several mm medial to the medial terminus of the lateral sulcus to a point just lateral to the medial end of the superior temporal sulcus; this area is termed PM by Maguire and Baizer (1984), is included as part of V4 by other authors (Gattass et al. 1988), and is essentially the same region in which the V4 cells reported in Mountcastle et al. (1987) were located. We refer to this area as V4/PM. Some sites could have been located in the adjacent area DP (Andersen et al. 1990a) since criteria for distinguishing FIG. 2. A: standardized drawing of left hemisphere showing regions where penetrations entered to record from cortical area V2 (horizontal hatching), area V4/PM (vertical hatching), and area 7a (left hatching). Scale: 1 cm. B: standardized coronal sections through left thalamus indicating regions in PI (left hatching), PL (horizontal hatching), and Pdm (vertical hatching) where recorded cells were estimated to lie. cs, central sulcus; ec, external calcarine s.; ios, inferior occipital s.; ip, intraparietal s.; lat, lateral s.; ls, lunate s.; sts, superior temporal s.; LGN, lateral geniculate nucleus; MG, medial geniculate n.; PI, inferior pulvinar n.; PL, lateral pulvinar n.; PM, medial pulvinar n.; SC, superior colliculus. among the three areas have yet to be clearly defined. Ninety-five percent of the cells were recorded within 1.7 mm of the cortical surface, except in V2 where 80% were buried in the posterior bank of the lunate sulcus. Thalamic recording sites (Fig. 2B) were assigned to the geniculate, and inferior (PI), lateral (PL), or dorsomedial part of the lateral pulvinar (Pdm) on the basis of receptive-field properties (Bender 1982), visuotopic organization (Bender 1981), and location of the penetration relative to the posterior tip of the geniculate. PI and PL were easily identifiable by their distinctive mirror-image visuotopic maps. Pdm is a visually responsive zone, just dorsomedial and adjacent to the periphery of the lower field representation of PL (labeled visual in Fig. 11, C E, Bender 1981); although penetrations through this area can yield systematic shifts in receptive-field position, its visuotopic organization has not been reported, and it may contain more than one visual area. Almost all lateral geniculate cells were estimated to lie within layers 5 6, based on depth, ocular dominance and whether responses were sustained or transient. RESULTS We found that both prevalence and magnitude of attentional modulation differed substantially across brain areas. Because the character of the modulation, i.e., the relative response magnitudes among the three attentional states, also differed among brain areas, we had to devise a measure of the magnitude that was independent of that character. Using that measure, we found that modulation was absent in the geniculate, modest in the pulvinar, and increased from modest in area V2
5 ATTENTIVE FIXATION IN PULVINAR AND CORTEX 223 to reach, in area 7a, the highest level seen in any of the brain areas studied. However, we also found systematic differences among brain areas in trial-to-trial variability of the response evoked by a visual stimulus. When we scaled the magnitude of attentional modulation in terms of this variability, using a kind of signal-to-noise ratio, we found that modulation was about the same in all brain areas beyond the lateral geniculate nucleus. In the following sections, we compare first the prevalence, then the character, and then the absolute magnitude of attentional modulation among brain areas. We next describe how brain areas differ in response variability, and show that the signal-to-noise ratio of attention is roughly constant at 1.3. Because task variant had no significant effect on modulation (with minor exceptions described at the end of RESULTS), we pooled data across all cells, regardless of variant. Prevalence of attentional modulation We recorded from 699 neurons in the thalamus and cortex of eight monkeys (Table 1). For each cell, we compared the responses evoked by the receptive-field stimulus flashed in the three behavioral states, ready, fixation, and idle, using a repeated measures, one-way ANOVA with three levels of state. A cell was considered to be significantly affected by attentional state if the F ratio from the ANOVA was significant at P THALAMUS. The prevalence of attentional modulation differed markedly between LGN and pulvinar. There was no evidence of modulation in the LGN. Only 2% of the cells (2/98) had a significant F ratio (P 0.02), no more than expected by chance. By contrast, about a quarter of the pulvinar cells (26%, 61/237) were affected by attention (see Fig. 3). Prevalence did not differ significantly among pulvinar subdivisions, but in the inferior and lateral subdivisions attentional modulation was slightly more common in central vision. A third of the cells (33%, 32/94) with receptive-field eccentricities 10 were affected by behavioral state, whereas only 20% (16/81) of the cells with more eccentric receptive-fields were affected ( 2 - test, P 0.03, excluding 6 cells with incomplete receptivefield data). CORTEX. As shown in Fig. 3, the prevalence of attentional modulation increased at higher levels of the cortical hierarchy ( 2 -test, df 2, P 0.001). Compared with the pulvinar, TABLE 1. Animal Cells tested for attentional modulation No. of Cells LGN Pulvinar Cortex Total P P35 12 P P P44* P45* V02 98 V Total * Thalamic cells were from one hemisphere, cortical cells from the opposite hemisphere. FIG. 3. Proportions of cells significantly affected by behavioral state in different areas of thalamus and cortex. Bars, based on ANOVA with P 0.02; dots, based on ANOVA with P 0.05 (see text); error bars, 1 SE based on binomial variance; N, number of neurons recorded in each area. prevalence in area V2 (21%, 19/91) and in area V4/PM (31%, 28/91) was about the same, whereas prevalence in area 7a (43%, 79/182) was significantly higher (P ). As in the pulvinar, prevalence in area 7a was slightly higher (57%, 17/30) among cells with more centrally located receptive fields (probe eccentricity 0.10 ) than for cells with more peripheral fields (41%, 62/152, probe eccentricity 10 ), but the difference did not reach significance. In estimating prevalence, we used an 0.02 so that for subsequent analyses there would be no more than one to two false positives in the sample of significantly affected cells. Had we used a more conventional 0.05 (see DISCUSSION), the corresponding estimates of prevalence would have been 36% for the pulvinar and 26 56% for cortex (Fig. 3, dots). Character of attentional modulation The ANOVA results identified cells affected by behavioral state but not the nature of the effect. To examine that, we discarded all unaffected cells and then looked at the patterns of relative response among the three states. Mountcastle et al. (1981) had found that almost all cells in area 7a gave their largest responses during fixation. However, we found virtually all possible patterns of relative excitability in both thalamus and cortex with no marked preponderance of high responsiveness during fixation in any area. For any one cell, the stimulusevoked response in one state might be either smaller or larger than that evoked in any other state. Figure 4 shows some typical examples of the different patterns of excitability seen in pulvinar neurons. In Fig. 4A, a cell in PL gave its strongest response during fixation with responses during both the ready and idle states only about half as strong. In Fig. 4B, a cell in Pdm shows the opposite pattern, giving no response during fixation and strong responses during the ready and idle states. Other cells gave their strongest response in the ready state (Fig. 4C) and still others in the idle state (Fig. 4D). In cortex, we saw the same diversity in patterns of excitability, though differences between states often seemed more extreme in area 7a. Figure 5A shows an area 7a cell giving a characteristically strong response during fixation. Other 7a cells gave similarly strong responses during the idle state (Fig. 5B) or ready state. Figure 5C shows a V2 cell with a pattern of excitability similar to that in Fig. 5A, though less pronounced.
6 224 D. B. BENDER AND M. YOUAKIM responses, in spikes/second, evoked in the fixation, ready, and idle states. Figure 6A illustrates the response vector for the cell in Fig. 4A; it points above the main diagonal (representing equal responses in all 3 states) because the response during fixation is about twice that in the other two states. Because response magnitudes, and thus response vector lengths, differed substantially among cells, we used a unit vector in the direction of r to represent the relative excitability of the cell in the three states. Each cell, regardless of overall response strength, could then have its relative excitability represented by a single point on a unit sphere. Figure 6B shows the unit response vector representation of relative excitability for the four cells illustrated in Fig. 4. A cell that showed no attentional modulation (equal responses in all three states) would be represented by the in Fig. 6B, where the main diagonal intersects the unit sphere. Relative excitabilities were plotted as unit vectors for all cells significantly affected by behavioral state. Figure 7 shows unit vector plots for cells in the three pulvinar subdivisions, Fig. 8 shows vector plots for cells in the three cortical areas. The plots suggested not only a general similarity between FIG. 4. Responses from 4 different pulvinar neurons to identical stimuli flashed in the ready (Rdy), fixation (Fix), and idle (Idl) states. A: PL neuron, stimulus speed 25 /s. B and D: dorsal portion of the lateral pulvinar (Pdm) neurons. C: PI neuron. Top: individual trials; bottom: averaged histogram (smoothed with 40 ms, 0 phase shift, triangular impulse-response filter). Responses for each state plotted in order of occurrence, but those on the same line did not necessarily occur on the same trial. Horizontal bar indicates stimulus duration; time marker, 100 ms; Sp/s, spikes/second. Despite the variety among individual cells in patterns of relative excitability, when populations of cells in different brain regions were compared, clear though subtle differences among regions were apparent. To compare cells, each was represented by a single vector in three-dimensional space, the cell s response vector, r; the three components of r were the FIG. 5. Responses from 3 different cortical neurons. A and B: area 7a neurons. C: V2 neuron. Conventions as in Fig. 4.
7 ATTENTIVE FIXATION IN PULVINAR AND CORTEX 225 plane was tilted 15 off normal to the plane that included both the fixation-state axis and main diagonal (Figs. 7 and 8, dotted lines). Thus for all brain areas, the fixation state was just about the best single-axis discriminant of the effect of behavioral state on excitability. Another similarity between thalamus and cortex was that (with the sole exception of Pdm) there were roughly similar FIG. 6. A: response vector, r, for neuron whose responses are shown in Fig. 4A; dashed line, main diagonal; dotted lines, components of r for the fixation, ready, and idle states. B: unit response vectors shown as points (large dots) on the surface of a unit sphere for the 4 correspondingly labeled neurons in Fig. 4, A D. For each cell, the relative excitability in each state can be read directly from the nearest grid lines since both grid lines and point are drawn on the sphere s surface. Viewer s perspective line for all coordinate systems of unit spheres is down main diagonal;, unit vector along main diagonal; dashed lines, constant relative-response contours for fixation state; solid lines, constant relative-response contours for ready and idle states. thalamus and cortex but also subtle differences among brain areas. The most obvious similarity between thalamus and cortex was that cells in all brain regions segregated into one of two groups: those that gave stronger responses during fixation (which we call F cells), and those that gave weaker responses during fixation ( F cells). To show this, we computed for the cells in each brain region that plane which both included the main diagonal of the unit sphere and maximized the root-mean-square distance of all the unit response vectors from the plane. It is thus the decision plane that maximally divides the cells of a given brain region into two groups from the point of no attentional modulation. 1 The intersection of the plane for each brain area with the unit sphere is shown as a heavy dashed line in Figs. 7 and 8. For every brain area, the 1 If pˆ is a unit vector normal to the discriminant plane, and dˆ is a unit vector on the main diagonal, and rˆi is the unit response vector of the ith cell in a brain area, then we chose the pˆ for that area to maximize pˆ rˆi 2 1 2, subject to i the constraint pˆ dˆ 0. FIG. 7. Unit response vectors for all pulvinar neurons significantly affected by behavioral state. Heavy dashed lines: optimal discriminant plane that includes main diagonal; heavy dotted lines: ideal discriminant plane based on fixation as a sole factor. A: neurons in PI. B: neurons in PL. C: neurons in Pdm. Other conventions as in Fig. 6B.
8 226 D. B. BENDER AND M. YOUAKIM neurons simply shut down when the animal fixated. This made Pdm a frustrating area to record from and may in part be responsible for our low estimate of prevalence there: some cells may have been passed over as unresponsive when in fact they were unresponsive only during fixation. There were also subtle differences among brain areas. First, excitability in the ready and idle states could be highly correlated or not. Thus the ready and idle components of the unit response vectors were strongly correlated in PL (Spearman rank-order correlation 0.59) and in area V2 ( 0.59), but essentially uncorrelated in PI ( 0.05) and in areas V4/PM ( 0.08) and 7a ( 0.15). Second, and reminiscent of the findings of Mountcastle et al. (1981), area 7a was unique in having many cells responding almost exclusively during fixation; 37% of the significantly modulated area 7a cells had a fixation-state component of 0.8 or more (above latitude 0.8 in Fig. 8C). By contrast, only 10% of the cells in V2, and 11% in V4/PM, had such a high relative responsiveness during fixation, with even fewer (5%) in the pulvinar. In showing that cells divided into two groups with respect to the point of zero modulation, we had constrained the discriminant plane to include the main diagonal. To avoid any potential bias from that constraint, we also did a principal components analysis based on the covariance matrix for the normalized discharge rates in the fixation, ready, and idle states, analyzing each brain area separately. The results confirmed the dominance of fixation in determining excitability. For every brain area, the first principal component (i.e., eigenvector with the largest eigenvalue) accounted for 65 88% of total variance, and its direction was the same as the discriminant plane s normal within 3 9 (except for Pdm). Furthermore, the second (orthogonal) principal component accounted for only 12 14% of variance for PL and V2 cells but for more than twice that in other brain areas, thus confirming the relatively high correlation of ready and idle state excitability in PL and V2. In summary, attentional modulation had a push-pull character with respect to the fixation state in all brain areas but one, Pdm, where there was exclusively depression of excitability during fixation. Relative excitabilities in the ready and idle states could be strongly correlated as in PL and area V2, or not, as in PI and cortical areas V4/PM and 7a. Magnitude of attentional modulation FIG. 8. Unit response vectors for all cortical neurons significantly affected by behavioral state. A: neurons in V2. B: neurons in V4/PM. C: neurons in area 7a. Conventions as in Fig. 7. proportions of F and F cells in both pulvinar and cortex. In the inferior and lateral pulvinar, 60% of the neurons were F cells (PI, 16/28; PL, 14/22). Likewise in cortex, 52% of the neurons were F cells (V2, 11/19; V4/PM, 12/28; 7a, 42/79). Thus in all regions but Pdm, the act of attentive fixation exerted a push-pull effect on neuronal excitability, with some cells gaining, and others losing, responsiveness. Pdm was unique in having only F cells (see Fig. 7C). In effect, these pulvinar Because the character of attentional modulation could vary substantially among cells and brain areas, we needed a measure of the magnitude of attentional modulation that, for each cell, would be unaffected by that cell s particular pattern of statedependent excitability. As a dimensionless measure of modulation amplitude, we used the angle between the response vector and the main diagonal, expressed as a percent of the largest possible angle (54.7 ); it is completely independent of the character of modulation. We call this measure of attentional modulation the percent angular modulation. A cell that responded equally in all three states would have 0% angular modulation, while a cell that responded in one state but not the other two would have 100% modulation; the intermediate case, equal responses in two states with none in the third, corresponds to 64% modulation. On this measure, we found clear differences among brain regions. Figure 9 shows for each brain area the average percent
9 ATTENTIVE FIXATION IN PULVINAR AND CORTEX 227 FIG. 9. Average amplitude of attentional modulation expressed as percent angular modulation for all neurons significantly affected by behavioral state. Error bars, 1 SE; N, number of neurons significantly affected by state in the indicated brain area. Response variability in different brain areas We had noticed that trial-to-trial variability in responsiveness seemed high in area 7a, where modulation was largest, and thus wondered whether modulation strength might be linked to the variability of an area for other brain areas as well. We looked at the variability of all responsive cells, not just those significantly affected by state, because we wanted to estimate variability of the whole brain area, not just of a particular subset of cells within it. Because we had to have a dimensionless measure of variability or noise to compare with the dimensionless measure of modulation strength, we used the percent error in cell response strength in the usual form of a coefficient of variation, SD err /gm, where SD err is the standard deviation based on the error sum of squares, and gm the grand mean, from the cell s ANOVA. Thus represents the percent noise left after removing all effects of behavioral state. (The percentage, rather than absolute, error was essential because response magnitude varied systematically across brain areas, see following text.) In the thalamus, percent variability was least in the LGN (average 0.25) where attentional modulation was undetectable (see Fig. 10A). For the pulvinar, percent variability was half again as large in PI and PL, where attentional modulation was modest, and largest of all in Pdm, the pulvinar subdivision with largest attentional modulation (average 0.45; P 0.001, Kruskal-Wallis, df 2). In cortex, percent variability increased with level in the cortical hierarchy (P , Kruskal-Wallis, df 2). In area V2, percent variability (average 0.41) was comparable to that in the pulvinar. It was larger in area V4/PM (average 0.54) and still larger in area 7a where it reached the highest value (average 0.60) of any brain region studied. Variability in area V4/PM was significantly larger than in either PI or PL (P , Kruskal-Wallis, df 3) although not different from Pdm, while in area 7a percent variability was angular modulation for all cells that were significantly affected by attention. In the pulvinar, angular modulation was modest, averaging just 21% of the maximum possible. This roughly corresponds to a 45% increase (or decrease) in response during fixation relative to that, for example, in the idle state. Average modulation was slightly larger in Pdm (26%) than in PI (19%) or PL (20%), but the difference among subdivisions did not reach significance. In cortex, angular modulation increased with level in the cortical hierarchy (P , Kruskal-Wallis, df 2). Average modulation was just 22% in V2, no different from that in the pulvinar. Modulation was larger in area V4/PM (32%) and larger still in area 7a (41%), where it reached the largest value of any brain area studied (representing roughly a 2.5-fold difference in response between fixation and idle states). Compared to the pulvinar, modulation in area V4/PM was significantly larger than in either PI or PL though not different from Pdm (P 0.001, Kruskal-Wallis, df 3), while modulation in area 7a was significantly larger than for all of the pulvinar divisions (P , Kruskal-Wallis, df 3). FIG. 10. A: average trial-to-trial response variability for all neurons recorded (N) in each brain area. B: comparison of average attentional modulation (significantly affected neurons only) per brain area and average response variability (all neurons) per area , linear regression of the 6 points. C: average response vector magnitude for all neurons in the indicated brain area. All error bars represent 1 SE.
10 228 D. B. BENDER AND M. YOUAKIM significantly larger than in any pulvinar division (P , Kruskal-Wallis, df 3). Comparing across all brain regions (except the geniculate), to a first approximation, there was a roughly constant relation between the average magnitude of attentional modulation and the average noise in an area. Figure 10B plots, for each area, the percent angular modulation averaged over all significantly affected cells in the area against the average for all cells in the area. Viewed in this way, both thalamic and cortical areas were essentially the same in their magnitude of attentional modulation. Linear regression of modulation on variability (Fig. 10B, - - -) showed that modulation was about threefourths of the noise (regression slope 78.8, intercept not significantly different from 0). Furthermore, the average of an area accounted for virtually all (adjusted R 2 94%) of the variance in angular modulation across areas. It should be noted that response strength, in spikes/second, also varied among brain regions but in the opposite sense: discharge went down as attention and noise went up. We used the length of a cell s response vector, r, to represent response strength. Figure 10C shows the average response strength for all cells in an area whether or not a cell was significantly affected by attention. Responses were strongest in the LGN and decreased by about half in the pulvinar. In cortex, responses were slightly stronger in V2 than in the pulvinar, but decreased in area V4/PM and again in area 7a to be the smallest, on average, of any brain area. Signal-to-noise ratio of attention The fact that the average magnitude of attentional modulation was a roughly constant fraction of a brain area s variability suggested that a signal-to noise ratio might be a more appropriate measure of attentional modulation than the absolute measure, percent angular modulation, that we had first computed. Thus for each cell that was significantly affected by attention, we defined a signal-to-noise measure of the attentional effect, S/N, as follows. The signal was defined by resolving a cell s raw response vector into two orthogonal components (see Fig. 11A). One (at, the attention vector) was perpendicular to the main diagonal and thus represented the entire effect of attentional state, in spikes/second. The other component, in the direction of the main diagonal, represented the amount of discharge, in spikes/second, unaffected by attention and played no role in this analysis. The noise, in spikes/second, was simply taken to be the error term in the cell s ANOVA (SD err as defined in the preceding text). S/N was then computed as the dimensionless ratio of the magnitude of the attention vector, at, to the noise: S/N at /SD err. Figure 11B shows the average signal-to-noise ratios for each brain area. On this measure of attentional modulation, brain areas hardly differed at all. The signal-to-noise ratio was roughly constant, to within 10%, for all areas, in both thalamus and cortex. Furthermore, attention had a rather modest effect, just 30% greater than noise; the average S/N for all affected cells, in all areas, was Interestingly, the uniqueness of area 7a in having many cells with a high relative responsiveness during fixation ( 0.8) disappeared: the average S/N of those cells (1.40) was no different from that for the rest of the significantly affected area 7a cells (1.41). FIG. 11. A: resolution of response vector, r, in Fig. 6A into attention vector, at, and an orthogonal vector along main diagonal (- - -). B: average signal-tonoise ratio of attention for all neurons significantly affected by behavioral state in the indicated brain area , average of all significantly affected neurons, regardless of brain area; error bars, 1 SE. Ongoing discharge and behavioral task variants Attentional state affected the ongoing or spontaneous discharge for a relatively small proportion of cells. Prevalence (repeated-measures, 1-way ANOVA, P 0.02) was about the same among pulvinar (18%, 43/238) and cortical neurons (17%, 61/356) and did not differ among areas. Prevalence was still smaller among lateral geniculate neurons (12%, 12/98). The most common effect was suppression of ongoing discharge during fixation. In all brain areas, modulation of ongoing and stimulus-evoked discharge were statistically independent ( 2 -test) so that very few cells showed modulation of both ongoing and stimulus-evoked discharge. We had used three minor variants of the fixation task (see Fig. 1), the major differences being whether an explicit ready response was required (task 1), whether the idle state appeared in a separate block of time (task 2), and whether the fixation point appeared in a predictable location (task 3) or not. In general, task variant did not significantly affect any quantitative measure related to attentional modulation, so that the major aspects of attentional modulation appear not to depend on details of the task. Prevalence, percent angular modulation, response variability, response magnitude, and the S/N of attention were all unaffected by task variant in both pulvinar and cortex as a whole. The character of attentional modulation, as reflected in the proportion of F cells, was likewise unaffected by task variant. Furthermore the trends of increasing percent angular modulation and percent response variability with level
Effects of Attention on MT and MST Neuronal Activity During Pursuit Initiation
 Effects of Attention on MT and MST Neuronal Activity During Pursuit Initiation GREGG H. RECANZONE 1 AND ROBERT H. WURTZ 2 1 Center for Neuroscience and Section of Neurobiology, Physiology and Behavior,
Effects of Attention on MT and MST Neuronal Activity During Pursuit Initiation GREGG H. RECANZONE 1 AND ROBERT H. WURTZ 2 1 Center for Neuroscience and Section of Neurobiology, Physiology and Behavior,
Attention Response Functions: Characterizing Brain Areas Using fmri Activation during Parametric Variations of Attentional Load
 Attention Response Functions: Characterizing Brain Areas Using fmri Activation during Parametric Variations of Attentional Load Intro Examine attention response functions Compare an attention-demanding
Attention Response Functions: Characterizing Brain Areas Using fmri Activation during Parametric Variations of Attentional Load Intro Examine attention response functions Compare an attention-demanding
CSE511 Brain & Memory Modeling. Lect21-22: Vision Central Pathways
 CSE511 Brain & Memory Modeling CSE511 Brain & Memory Modeling Lect02: BOSS Discrete Event Simulator Lect21-22: Vision Central Pathways Chapter 12 of Purves et al., 4e Larry Wittie Computer Science, StonyBrook
CSE511 Brain & Memory Modeling CSE511 Brain & Memory Modeling Lect02: BOSS Discrete Event Simulator Lect21-22: Vision Central Pathways Chapter 12 of Purves et al., 4e Larry Wittie Computer Science, StonyBrook
Exploring the pulvinar path to visual cortex
 C. Kennard & R.J. Leigh (Eds.) Progress in Brain Research, Vol. 171 ISSN 0079-6123 Copyright r 2008 Elsevier B.V. All rights reserved CHAPTER 5.14 Exploring the pulvinar path to visual cortex Rebecca A.
C. Kennard & R.J. Leigh (Eds.) Progress in Brain Research, Vol. 171 ISSN 0079-6123 Copyright r 2008 Elsevier B.V. All rights reserved CHAPTER 5.14 Exploring the pulvinar path to visual cortex Rebecca A.
Area MT Neurons Respond to Visual Motion Distant From Their Receptive Fields
 J Neurophysiol 94: 4156 4167, 2005. First published August 24, 2005; doi:10.1152/jn.00505.2005. Area MT Neurons Respond to Visual Motion Distant From Their Receptive Fields Daniel Zaksas and Tatiana Pasternak
J Neurophysiol 94: 4156 4167, 2005. First published August 24, 2005; doi:10.1152/jn.00505.2005. Area MT Neurons Respond to Visual Motion Distant From Their Receptive Fields Daniel Zaksas and Tatiana Pasternak
Reading Assignments: Lecture 5: Introduction to Vision. None. Brain Theory and Artificial Intelligence
 Brain Theory and Artificial Intelligence Lecture 5:. Reading Assignments: None 1 Projection 2 Projection 3 Convention: Visual Angle Rather than reporting two numbers (size of object and distance to observer),
Brain Theory and Artificial Intelligence Lecture 5:. Reading Assignments: None 1 Projection 2 Projection 3 Convention: Visual Angle Rather than reporting two numbers (size of object and distance to observer),
LISC-322 Neuroscience Cortical Organization
 LISC-322 Neuroscience Cortical Organization THE VISUAL SYSTEM Higher Visual Processing Martin Paré Assistant Professor Physiology & Psychology Most of the cortex that covers the cerebral hemispheres is
LISC-322 Neuroscience Cortical Organization THE VISUAL SYSTEM Higher Visual Processing Martin Paré Assistant Professor Physiology & Psychology Most of the cortex that covers the cerebral hemispheres is
Retinotopy & Phase Mapping
 Retinotopy & Phase Mapping Fani Deligianni B. A. Wandell, et al. Visual Field Maps in Human Cortex, Neuron, 56(2):366-383, 2007 Retinotopy Visual Cortex organised in visual field maps: Nearby neurons have
Retinotopy & Phase Mapping Fani Deligianni B. A. Wandell, et al. Visual Field Maps in Human Cortex, Neuron, 56(2):366-383, 2007 Retinotopy Visual Cortex organised in visual field maps: Nearby neurons have
CS294-6 (Fall 2004) Recognizing People, Objects and Actions Lecture: January 27, 2004 Human Visual System
 CS294-6 (Fall 2004) Recognizing People, Objects and Actions Lecture: January 27, 2004 Human Visual System Lecturer: Jitendra Malik Scribe: Ryan White (Slide: layout of the brain) Facts about the brain:
CS294-6 (Fall 2004) Recognizing People, Objects and Actions Lecture: January 27, 2004 Human Visual System Lecturer: Jitendra Malik Scribe: Ryan White (Slide: layout of the brain) Facts about the brain:
PHY3111 Mid-Semester Test Study. Lecture 2: The hierarchical organisation of vision
 PHY3111 Mid-Semester Test Study Lecture 2: The hierarchical organisation of vision 1. Explain what a hierarchically organised neural system is, in terms of physiological response properties of its neurones.
PHY3111 Mid-Semester Test Study Lecture 2: The hierarchical organisation of vision 1. Explain what a hierarchically organised neural system is, in terms of physiological response properties of its neurones.
Vision II. Steven McLoon Department of Neuroscience University of Minnesota
 Vision II Steven McLoon Department of Neuroscience University of Minnesota 1 Ganglion Cells The axons of the retinal ganglion cells form the optic nerve and carry visual information into the brain. 2 Optic
Vision II Steven McLoon Department of Neuroscience University of Minnesota 1 Ganglion Cells The axons of the retinal ganglion cells form the optic nerve and carry visual information into the brain. 2 Optic
Lateral Geniculate Nucleus (LGN)
 Lateral Geniculate Nucleus (LGN) What happens beyond the retina? What happens in Lateral Geniculate Nucleus (LGN)- 90% flow Visual cortex Information Flow Superior colliculus 10% flow Slide 2 Information
Lateral Geniculate Nucleus (LGN) What happens beyond the retina? What happens in Lateral Geniculate Nucleus (LGN)- 90% flow Visual cortex Information Flow Superior colliculus 10% flow Slide 2 Information
THE INFLUENCE OF THE ANGLE OF GAZE UPON THE EXCITABILITY OF THE LIGHT-SENSITIVE NEURONS OF THE POSTERIOR PARIETAL CORTEX1
 0270~6474830303-0532$02.00O Copyright 0 Society for Neuroscience Printed in U.S.A. The Journal of Neuroscience Vol. 3, No. 3, pp. 532-548 March 1983 THE INFLUENCE OF THE ANGLE OF GAZE UPON THE EXCITABILITY
0270~6474830303-0532$02.00O Copyright 0 Society for Neuroscience Printed in U.S.A. The Journal of Neuroscience Vol. 3, No. 3, pp. 532-548 March 1983 THE INFLUENCE OF THE ANGLE OF GAZE UPON THE EXCITABILITY
Spatial Distribution of Contextual Interactions in Primary Visual Cortex and in Visual Perception
 Spatial Distribution of Contextual Interactions in Primary Visual Cortex and in Visual Perception MITESH K. KAPADIA, 1,2 GERALD WESTHEIMER, 1 AND CHARLES D. GILBERT 1 1 The Rockefeller University, New
Spatial Distribution of Contextual Interactions in Primary Visual Cortex and in Visual Perception MITESH K. KAPADIA, 1,2 GERALD WESTHEIMER, 1 AND CHARLES D. GILBERT 1 1 The Rockefeller University, New
Privileged Processing of the Straight-Ahead Direction in Primate Area V1
 Article Privileged Processing of the Straight-Ahead Direction in Primate Area V1 Jean-Baptiste Durand, 1,2 Yves Trotter, 1,2 and Simona Celebrini 1,2, * 1 Université de Toulouse; UPS; Centre de Recherche
Article Privileged Processing of the Straight-Ahead Direction in Primate Area V1 Jean-Baptiste Durand, 1,2 Yves Trotter, 1,2 and Simona Celebrini 1,2, * 1 Université de Toulouse; UPS; Centre de Recherche
Multiple Visual Areas in the Caudal Superior Temporal Sulcus of the Macaque
 THE JOURNAL OF COMPARATIVE NEUROLOGY 248:164-189 (1986) Multiple Visual Areas in the Caudal Superior Temporal Sulcus of the Macaque ROBERT DESIMONE AND LESLIE G. UNGERLEIDER Laboratory of Neuropsychology,
THE JOURNAL OF COMPARATIVE NEUROLOGY 248:164-189 (1986) Multiple Visual Areas in the Caudal Superior Temporal Sulcus of the Macaque ROBERT DESIMONE AND LESLIE G. UNGERLEIDER Laboratory of Neuropsychology,
Required Slide. Session Objectives
 Vision: CNS 2018 Required Slide Session Objectives Visual system: CNS At the end of this session, students will be able to: 1. Understand how axons from the eyes travel through the optic nerves and tracts
Vision: CNS 2018 Required Slide Session Objectives Visual system: CNS At the end of this session, students will be able to: 1. Understand how axons from the eyes travel through the optic nerves and tracts
Parallel streams of visual processing
 Parallel streams of visual processing RETINAL GANGLION CELL AXONS: OPTIC TRACT Optic nerve Optic tract Optic chiasm Lateral geniculate nucleus Hypothalamus: regulation of circadian rhythms Pretectum: reflex
Parallel streams of visual processing RETINAL GANGLION CELL AXONS: OPTIC TRACT Optic nerve Optic tract Optic chiasm Lateral geniculate nucleus Hypothalamus: regulation of circadian rhythms Pretectum: reflex
Key questions about attention
 Key questions about attention How does attention affect behavioral performance? Can attention affect the appearance of things? How does spatial and feature-based attention affect neuronal responses in
Key questions about attention How does attention affect behavioral performance? Can attention affect the appearance of things? How does spatial and feature-based attention affect neuronal responses in
Pathway from the eye to the cortex
 Vision: CNS 2017 Pathway from the eye to the cortex Themes of this lecture Visual information is analyzed in more complicated ways than in the retina. One major pathway from the eye leads to the striate
Vision: CNS 2017 Pathway from the eye to the cortex Themes of this lecture Visual information is analyzed in more complicated ways than in the retina. One major pathway from the eye leads to the striate
Neuroscience Tutorial
 Neuroscience Tutorial Brain Organization : cortex, basal ganglia, limbic lobe : thalamus, hypothal., pituitary gland : medulla oblongata, midbrain, pons, cerebellum Cortical Organization Cortical Organization
Neuroscience Tutorial Brain Organization : cortex, basal ganglia, limbic lobe : thalamus, hypothal., pituitary gland : medulla oblongata, midbrain, pons, cerebellum Cortical Organization Cortical Organization
Early Learning vs Early Variability 1.5 r = p = Early Learning r = p = e 005. Early Learning 0.
 The temporal structure of motor variability is dynamically regulated and predicts individual differences in motor learning ability Howard Wu *, Yohsuke Miyamoto *, Luis Nicolas Gonzales-Castro, Bence P.
The temporal structure of motor variability is dynamically regulated and predicts individual differences in motor learning ability Howard Wu *, Yohsuke Miyamoto *, Luis Nicolas Gonzales-Castro, Bence P.
Spectro-temporal response fields in the inferior colliculus of awake monkey
 3.6.QH Spectro-temporal response fields in the inferior colliculus of awake monkey Versnel, Huib; Zwiers, Marcel; Van Opstal, John Department of Biophysics University of Nijmegen Geert Grooteplein 655
3.6.QH Spectro-temporal response fields in the inferior colliculus of awake monkey Versnel, Huib; Zwiers, Marcel; Van Opstal, John Department of Biophysics University of Nijmegen Geert Grooteplein 655
Substructure of Direction-Selective Receptive Fields in Macaque V1
 J Neurophysiol 89: 2743 2759, 2003; 10.1152/jn.00822.2002. Substructure of Direction-Selective Receptive Fields in Macaque V1 Margaret S. Livingstone and Bevil R. Conway Department of Neurobiology, Harvard
J Neurophysiol 89: 2743 2759, 2003; 10.1152/jn.00822.2002. Substructure of Direction-Selective Receptive Fields in Macaque V1 Margaret S. Livingstone and Bevil R. Conway Department of Neurobiology, Harvard
The individual animals, the basic design of the experiments and the electrophysiological
 SUPPORTING ONLINE MATERIAL Material and Methods The individual animals, the basic design of the experiments and the electrophysiological techniques for extracellularly recording from dopamine neurons were
SUPPORTING ONLINE MATERIAL Material and Methods The individual animals, the basic design of the experiments and the electrophysiological techniques for extracellularly recording from dopamine neurons were
Changing expectations about speed alters perceived motion direction
 Current Biology, in press Supplemental Information: Changing expectations about speed alters perceived motion direction Grigorios Sotiropoulos, Aaron R. Seitz, and Peggy Seriès Supplemental Data Detailed
Current Biology, in press Supplemental Information: Changing expectations about speed alters perceived motion direction Grigorios Sotiropoulos, Aaron R. Seitz, and Peggy Seriès Supplemental Data Detailed
Visual Physiology. Perception and Attention. Graham Hole. Problems confronting the visual system: Solutions: The primary visual pathways: The eye:
 Problems confronting the visual system: Visual Physiology image contains a huge amount of information which must be processed quickly. image is dim, blurry and distorted. Light levels vary enormously.
Problems confronting the visual system: Visual Physiology image contains a huge amount of information which must be processed quickly. image is dim, blurry and distorted. Light levels vary enormously.
Supplementary materials for: Executive control processes underlying multi- item working memory
 Supplementary materials for: Executive control processes underlying multi- item working memory Antonio H. Lara & Jonathan D. Wallis Supplementary Figure 1 Supplementary Figure 1. Behavioral measures of
Supplementary materials for: Executive control processes underlying multi- item working memory Antonio H. Lara & Jonathan D. Wallis Supplementary Figure 1 Supplementary Figure 1. Behavioral measures of
The Visual System. Cortical Architecture Casagrande February 23, 2004
 The Visual System Cortical Architecture Casagrande February 23, 2004 Phone: 343-4538 Email: vivien.casagrande@mcmail.vanderbilt.edu Office: T2302 MCN Required Reading Adler s Physiology of the Eye Chapters
The Visual System Cortical Architecture Casagrande February 23, 2004 Phone: 343-4538 Email: vivien.casagrande@mcmail.vanderbilt.edu Office: T2302 MCN Required Reading Adler s Physiology of the Eye Chapters
7 Grip aperture and target shape
 7 Grip aperture and target shape Based on: Verheij R, Brenner E, Smeets JBJ. The influence of target object shape on maximum grip aperture in human grasping movements. Exp Brain Res, In revision 103 Introduction
7 Grip aperture and target shape Based on: Verheij R, Brenner E, Smeets JBJ. The influence of target object shape on maximum grip aperture in human grasping movements. Exp Brain Res, In revision 103 Introduction
Representation of negative motivational value in the primate
 Representation of negative motivational value in the primate lateral habenula Masayuki Matsumoto & Okihide Hikosaka Supplementary Figure 1 Anticipatory licking and blinking. (a, b) The average normalized
Representation of negative motivational value in the primate lateral habenula Masayuki Matsumoto & Okihide Hikosaka Supplementary Figure 1 Anticipatory licking and blinking. (a, b) The average normalized
OPTO 5320 VISION SCIENCE I
 OPTO 5320 VISION SCIENCE I Monocular Sensory Processes of Vision: Color Vision Mechanisms of Color Processing . Neural Mechanisms of Color Processing A. Parallel processing - M- & P- pathways B. Second
OPTO 5320 VISION SCIENCE I Monocular Sensory Processes of Vision: Color Vision Mechanisms of Color Processing . Neural Mechanisms of Color Processing A. Parallel processing - M- & P- pathways B. Second
Neurons in Dorsal Visual Area V5/MT Signal Relative
 17892 The Journal of Neuroscience, December 7, 211 31(49):17892 1794 Behavioral/Systems/Cognitive Neurons in Dorsal Visual Area V5/MT Signal Relative Disparity Kristine Krug and Andrew J. Parker Oxford
17892 The Journal of Neuroscience, December 7, 211 31(49):17892 1794 Behavioral/Systems/Cognitive Neurons in Dorsal Visual Area V5/MT Signal Relative Disparity Kristine Krug and Andrew J. Parker Oxford
2/3/17. Visual System I. I. Eye, color space, adaptation II. Receptive fields and lateral inhibition III. Thalamus and primary visual cortex
 1 Visual System I I. Eye, color space, adaptation II. Receptive fields and lateral inhibition III. Thalamus and primary visual cortex 2 1 2/3/17 Window of the Soul 3 Information Flow: From Photoreceptors
1 Visual System I I. Eye, color space, adaptation II. Receptive fields and lateral inhibition III. Thalamus and primary visual cortex 2 1 2/3/17 Window of the Soul 3 Information Flow: From Photoreceptors
LISC-322 Neuroscience. Visual Field Representation. Visual Field Representation. Visual Field Representation. Visual Field Representation
 LISC-3 Neuroscience THE VISUAL SYSTEM Central Visual Pathways Each eye sees a part of the visual space that defines its visual field. The s of both eyes overlap extensively to create a binocular. eye both
LISC-3 Neuroscience THE VISUAL SYSTEM Central Visual Pathways Each eye sees a part of the visual space that defines its visual field. The s of both eyes overlap extensively to create a binocular. eye both
Attention: Neural Mechanisms and Attentional Control Networks Attention 2
 Attention: Neural Mechanisms and Attentional Control Networks Attention 2 Hillyard(1973) Dichotic Listening Task N1 component enhanced for attended stimuli Supports early selection Effects of Voluntary
Attention: Neural Mechanisms and Attentional Control Networks Attention 2 Hillyard(1973) Dichotic Listening Task N1 component enhanced for attended stimuli Supports early selection Effects of Voluntary
Computational model of MST neuron receptive field and interaction effect for the perception of selfmotion
 Rochester Institute of Technology RIT Scholar Works Theses Thesis/Dissertation Collections 2008 Computational model of MST neuron receptive field and interaction effect for the perception of selfmotion
Rochester Institute of Technology RIT Scholar Works Theses Thesis/Dissertation Collections 2008 Computational model of MST neuron receptive field and interaction effect for the perception of selfmotion
9.14 Classes #21-23: Visual systems
 9.14 Classes #21-23: Visual systems Questions based on Schneider chapter 20 and classes: 1) What was in all likelihood the first functional role of the visual sense? Describe the nature of the most primitive
9.14 Classes #21-23: Visual systems Questions based on Schneider chapter 20 and classes: 1) What was in all likelihood the first functional role of the visual sense? Describe the nature of the most primitive
Competitive Mechanisms Subserve Attention in Macaque Areas V2 and V4
 The Journal of Neuroscience, March 1, 1999, 19(5):1736 1753 Competitive Mechanisms Subserve Attention in Macaque Areas V2 and V4 John H. Reynolds, 1 Leonardo Chelazzi, 2 and Robert Desimone 1 1 Laboratory
The Journal of Neuroscience, March 1, 1999, 19(5):1736 1753 Competitive Mechanisms Subserve Attention in Macaque Areas V2 and V4 John H. Reynolds, 1 Leonardo Chelazzi, 2 and Robert Desimone 1 1 Laboratory
 Peripheral facial paralysis (right side). The patient is asked to close her eyes and to retract their mouth (From Heimer) Hemiplegia of the left side. Note the characteristic position of the arm with
Peripheral facial paralysis (right side). The patient is asked to close her eyes and to retract their mouth (From Heimer) Hemiplegia of the left side. Note the characteristic position of the arm with
Nature Neuroscience: doi: /nn Supplementary Figure 1. Trial structure for go/no-go behavior
 Supplementary Figure 1 Trial structure for go/no-go behavior a, Overall timeline of experiments. Day 1: A1 mapping, injection of AAV1-SYN-GCAMP6s, cranial window and headpost implantation. Water restriction
Supplementary Figure 1 Trial structure for go/no-go behavior a, Overall timeline of experiments. Day 1: A1 mapping, injection of AAV1-SYN-GCAMP6s, cranial window and headpost implantation. Water restriction
Sum of Neurally Distinct Stimulus- and Task-Related Components.
 SUPPLEMENTARY MATERIAL for Cardoso et al. 22 The Neuroimaging Signal is a Linear Sum of Neurally Distinct Stimulus- and Task-Related Components. : Appendix: Homogeneous Linear ( Null ) and Modified Linear
SUPPLEMENTARY MATERIAL for Cardoso et al. 22 The Neuroimaging Signal is a Linear Sum of Neurally Distinct Stimulus- and Task-Related Components. : Appendix: Homogeneous Linear ( Null ) and Modified Linear
Supplementary Information Methods Subjects The study was comprised of 84 chronic pain patients with either chronic back pain (CBP) or osteoarthritis
 Supplementary Information Methods Subjects The study was comprised of 84 chronic pain patients with either chronic back pain (CBP) or osteoarthritis (OA). All subjects provided informed consent to procedures
Supplementary Information Methods Subjects The study was comprised of 84 chronic pain patients with either chronic back pain (CBP) or osteoarthritis (OA). All subjects provided informed consent to procedures
Supplementary Figure 1 Information on transgenic mouse models and their recording and optogenetic equipment. (a) 108 (b-c) (d) (e) (f) (g)
 Supplementary Figure 1 Information on transgenic mouse models and their recording and optogenetic equipment. (a) In four mice, cre-dependent expression of the hyperpolarizing opsin Arch in pyramidal cells
Supplementary Figure 1 Information on transgenic mouse models and their recording and optogenetic equipment. (a) In four mice, cre-dependent expression of the hyperpolarizing opsin Arch in pyramidal cells
Photoreceptors Rods. Cones
 Photoreceptors Rods Cones 120 000 000 Dim light Prefer wavelength of 505 nm Monochromatic Mainly in periphery of the eye 6 000 000 More light Different spectral sensitivities!long-wave receptors (558 nm)
Photoreceptors Rods Cones 120 000 000 Dim light Prefer wavelength of 505 nm Monochromatic Mainly in periphery of the eye 6 000 000 More light Different spectral sensitivities!long-wave receptors (558 nm)
C:\Documents and Settings\sstensaas\Desktop\dental visual 2010\VisualPath dental 2010.docVisualPath dental 2010.doc
 Neuroanatomy Suzanne Stensaas April 8, 2010, 10:00-12:00 p.m. Reading: Waxman Ch. 15, Computer Resources: HyperBrain Ch 7 THE VISUAL PATHWAY Objectives: 1. Describe the pathway of visual information from
Neuroanatomy Suzanne Stensaas April 8, 2010, 10:00-12:00 p.m. Reading: Waxman Ch. 15, Computer Resources: HyperBrain Ch 7 THE VISUAL PATHWAY Objectives: 1. Describe the pathway of visual information from
Medial Superior Temporal Area Neurons Respond to Speed Patterns in Optic Flow
 The Journal of Neuroscience, April 15, 1997, 17(8):2839 2851 Medial Superior Temporal Area Neurons Respond to Speed Patterns in Optic Flow Charles J. Duffy 1 and Robert H. Wurtz 2 1 Departments of Neurology,
The Journal of Neuroscience, April 15, 1997, 17(8):2839 2851 Medial Superior Temporal Area Neurons Respond to Speed Patterns in Optic Flow Charles J. Duffy 1 and Robert H. Wurtz 2 1 Departments of Neurology,
Plasticity of Cerebral Cortex in Development
 Plasticity of Cerebral Cortex in Development Jessica R. Newton and Mriganka Sur Department of Brain & Cognitive Sciences Picower Center for Learning & Memory Massachusetts Institute of Technology Cambridge,
Plasticity of Cerebral Cortex in Development Jessica R. Newton and Mriganka Sur Department of Brain & Cognitive Sciences Picower Center for Learning & Memory Massachusetts Institute of Technology Cambridge,
Selective Attention. Modes of Control. Domains of Selection
 The New Yorker (2/7/5) Selective Attention Perception and awareness are necessarily selective (cell phone while driving): attention gates access to awareness Selective attention is deployed via two modes
The New Yorker (2/7/5) Selective Attention Perception and awareness are necessarily selective (cell phone while driving): attention gates access to awareness Selective attention is deployed via two modes
Adapting internal statistical models for interpreting visual cues to depth
 Journal of Vision (2010) 10(4):1, 1 27 http://journalofvision.org/10/4/1/ 1 Adapting internal statistical models for interpreting visual cues to depth Anna Seydell David C. Knill Julia Trommershäuser Department
Journal of Vision (2010) 10(4):1, 1 27 http://journalofvision.org/10/4/1/ 1 Adapting internal statistical models for interpreting visual cues to depth Anna Seydell David C. Knill Julia Trommershäuser Department
Extrastriate Visual Areas February 27, 2003 A. Roe
 Extrastriate Visual Areas February 27, 2003 A. Roe How many extrastriate areas are there? LOTS!!! Macaque monkey flattened cortex Why? How do we know this? Topography Functional properties Connections
Extrastriate Visual Areas February 27, 2003 A. Roe How many extrastriate areas are there? LOTS!!! Macaque monkey flattened cortex Why? How do we know this? Topography Functional properties Connections
Discharge Characteristics of Pursuit Neurons in MST During Vergence Eye Movements
 J Neurophysiol 93: 2415 2434, 2005. First published December 8, 2004; doi:10.1152/jn.01028.2004. Discharge Characteristics of Pursuit Neurons in MST During Vergence Eye Movements Teppei Akao, 1 Michael
J Neurophysiol 93: 2415 2434, 2005. First published December 8, 2004; doi:10.1152/jn.01028.2004. Discharge Characteristics of Pursuit Neurons in MST During Vergence Eye Movements Teppei Akao, 1 Michael
Cortical Control of Movement
 Strick Lecture 2 March 24, 2006 Page 1 Cortical Control of Movement Four parts of this lecture: I) Anatomical Framework, II) Physiological Framework, III) Primary Motor Cortex Function and IV) Premotor
Strick Lecture 2 March 24, 2006 Page 1 Cortical Control of Movement Four parts of this lecture: I) Anatomical Framework, II) Physiological Framework, III) Primary Motor Cortex Function and IV) Premotor
1. The responses of on-center and off-center retinal ganglion cells
 1. The responses of on-center and off-center retinal ganglion cells 2. Responses of an on-center ganglion cell to different light conditions 3. Responses of an on-center ganglion cells to different light
1. The responses of on-center and off-center retinal ganglion cells 2. Responses of an on-center ganglion cell to different light conditions 3. Responses of an on-center ganglion cells to different light
Adventures into terra incognita
 BEWARE: These are preliminary notes. In the future, they will become part of a textbook on Visual Object Recognition. Chapter VI. Adventures into terra incognita In primary visual cortex there are neurons
BEWARE: These are preliminary notes. In the future, they will become part of a textbook on Visual Object Recognition. Chapter VI. Adventures into terra incognita In primary visual cortex there are neurons
Functional Imaging of the Human Lateral Geniculate Nucleus and Pulvinar
 J Neurophysiol 91: 438 448, 2004. First published September 17, 2003; 10.1152/jn.00553.2003. Functional Imaging of the Human Lateral Geniculate Nucleus and Pulvinar Sabine Kastner, Daniel H. O Connor,
J Neurophysiol 91: 438 448, 2004. First published September 17, 2003; 10.1152/jn.00553.2003. Functional Imaging of the Human Lateral Geniculate Nucleus and Pulvinar Sabine Kastner, Daniel H. O Connor,
Senses are transducers. Change one form of energy into another Light, sound, pressure, etc. into What?
 1 Vision 2 TRANSDUCTION Senses are transducers Change one form of energy into another Light, sound, pressure, etc. into What? Action potentials! Sensory codes Frequency code encodes information about intensity
1 Vision 2 TRANSDUCTION Senses are transducers Change one form of energy into another Light, sound, pressure, etc. into What? Action potentials! Sensory codes Frequency code encodes information about intensity
Ch 5. Perception and Encoding
 Ch 5. Perception and Encoding Cognitive Neuroscience: The Biology of the Mind, 2 nd Ed., M. S. Gazzaniga, R. B. Ivry, and G. R. Mangun, Norton, 2002. Summarized by Y.-J. Park, M.-H. Kim, and B.-T. Zhang
Ch 5. Perception and Encoding Cognitive Neuroscience: The Biology of the Mind, 2 nd Ed., M. S. Gazzaniga, R. B. Ivry, and G. R. Mangun, Norton, 2002. Summarized by Y.-J. Park, M.-H. Kim, and B.-T. Zhang
Neural Activity in Primate Parietal Area 7a Related to Spatial Analysis of Visual Mazes
 Neural Activity in Primate Parietal Area 7a Related to Spatial Analysis of Visual Mazes David A. Crowe 1,2, Matthew V. Chafee 1,3, Bruno B. Averbeck 1,2 and Apostolos P. Georgopoulos 1 5 1 Brain Sciences
Neural Activity in Primate Parietal Area 7a Related to Spatial Analysis of Visual Mazes David A. Crowe 1,2, Matthew V. Chafee 1,3, Bruno B. Averbeck 1,2 and Apostolos P. Georgopoulos 1 5 1 Brain Sciences
Response profiles to texture border patterns in area V1
 Visual Neuroscience (2000), 17, 421 436. Printed in the USA. Copyright 2000 Cambridge University Press 0952-5238000 $12.50 Response profiles to texture border patterns in area V1 HANS-CHRISTOPH NOTHDURFT,
Visual Neuroscience (2000), 17, 421 436. Printed in the USA. Copyright 2000 Cambridge University Press 0952-5238000 $12.50 Response profiles to texture border patterns in area V1 HANS-CHRISTOPH NOTHDURFT,
COGS 101A: Sensation and Perception
 COGS 101A: Sensation and Perception 1 Virginia R. de Sa Department of Cognitive Science UCSD Lecture 5: LGN and V1: Magno and Parvo streams Chapter 3 Course Information 2 Class web page: http://cogsci.ucsd.edu/
COGS 101A: Sensation and Perception 1 Virginia R. de Sa Department of Cognitive Science UCSD Lecture 5: LGN and V1: Magno and Parvo streams Chapter 3 Course Information 2 Class web page: http://cogsci.ucsd.edu/
Ch 5. Perception and Encoding
 Ch 5. Perception and Encoding Cognitive Neuroscience: The Biology of the Mind, 2 nd Ed., M. S. Gazzaniga,, R. B. Ivry,, and G. R. Mangun,, Norton, 2002. Summarized by Y.-J. Park, M.-H. Kim, and B.-T. Zhang
Ch 5. Perception and Encoding Cognitive Neuroscience: The Biology of the Mind, 2 nd Ed., M. S. Gazzaniga,, R. B. Ivry,, and G. R. Mangun,, Norton, 2002. Summarized by Y.-J. Park, M.-H. Kim, and B.-T. Zhang
(Visual) Attention. October 3, PSY Visual Attention 1
 (Visual) Attention Perception and awareness of a visual object seems to involve attending to the object. Do we have to attend to an object to perceive it? Some tasks seem to proceed with little or no attention
(Visual) Attention Perception and awareness of a visual object seems to involve attending to the object. Do we have to attend to an object to perceive it? Some tasks seem to proceed with little or no attention
Comparison of Shape Encoding in Primate Dorsal and Ventral Visual Pathways
 J Neurophysiol 97: 37 319, 27. First published October 4, 26; doi:1.1152/jn.168.26. Comparison of Shape Encoding in Primate Dorsal and Ventral Visual Pathways Sidney R. Lehky 1,2 and Anne B. Sereno 2,3
J Neurophysiol 97: 37 319, 27. First published October 4, 26; doi:1.1152/jn.168.26. Comparison of Shape Encoding in Primate Dorsal and Ventral Visual Pathways Sidney R. Lehky 1,2 and Anne B. Sereno 2,3
The Eye. Cognitive Neuroscience of Language. Today s goals. 5 From eye to brain. Today s reading
 Cognitive Neuroscience of Language 5 From eye to brain Today s goals Look at the pathways that conduct the visual information from the eye to the visual cortex Marielle Lange http://homepages.inf.ed.ac.uk/mlange/teaching/cnl/
Cognitive Neuroscience of Language 5 From eye to brain Today s goals Look at the pathways that conduct the visual information from the eye to the visual cortex Marielle Lange http://homepages.inf.ed.ac.uk/mlange/teaching/cnl/
Nature Neuroscience doi: /nn Supplementary Figure 1. Characterization of viral injections.
 Supplementary Figure 1 Characterization of viral injections. (a) Dorsal view of a mouse brain (dashed white outline) after receiving a large, unilateral thalamic injection (~100 nl); demonstrating that
Supplementary Figure 1 Characterization of viral injections. (a) Dorsal view of a mouse brain (dashed white outline) after receiving a large, unilateral thalamic injection (~100 nl); demonstrating that
Consistency of Encoding in Monkey Visual Cortex
 The Journal of Neuroscience, October 15, 2001, 21(20):8210 8221 Consistency of Encoding in Monkey Visual Cortex Matthew C. Wiener, Mike W. Oram, Zheng Liu, and Barry J. Richmond Laboratory of Neuropsychology,
The Journal of Neuroscience, October 15, 2001, 21(20):8210 8221 Consistency of Encoding in Monkey Visual Cortex Matthew C. Wiener, Mike W. Oram, Zheng Liu, and Barry J. Richmond Laboratory of Neuropsychology,
DATA MANAGEMENT & TYPES OF ANALYSES OFTEN USED. Dennis L. Molfese University of Nebraska - Lincoln
 DATA MANAGEMENT & TYPES OF ANALYSES OFTEN USED Dennis L. Molfese University of Nebraska - Lincoln 1 DATA MANAGEMENT Backups Storage Identification Analyses 2 Data Analysis Pre-processing Statistical Analysis
DATA MANAGEMENT & TYPES OF ANALYSES OFTEN USED Dennis L. Molfese University of Nebraska - Lincoln 1 DATA MANAGEMENT Backups Storage Identification Analyses 2 Data Analysis Pre-processing Statistical Analysis
Cortical Connections of Area V4 in the Macaque
 Cerebral Cortex March 2008;18:477-499 doi:10.1093/cercor/bhm061 Advance Access publication June 4, 2007 Cortical Connections of Area V4 in the Macaque Leslie G. Ungerleider 1, Thelma W. Galkin 2, Robert
Cerebral Cortex March 2008;18:477-499 doi:10.1093/cercor/bhm061 Advance Access publication June 4, 2007 Cortical Connections of Area V4 in the Macaque Leslie G. Ungerleider 1, Thelma W. Galkin 2, Robert
Discrimination and Generalization in Pattern Categorization: A Case for Elemental Associative Learning
 Discrimination and Generalization in Pattern Categorization: A Case for Elemental Associative Learning E. J. Livesey (el253@cam.ac.uk) P. J. C. Broadhurst (pjcb3@cam.ac.uk) I. P. L. McLaren (iplm2@cam.ac.uk)
Discrimination and Generalization in Pattern Categorization: A Case for Elemental Associative Learning E. J. Livesey (el253@cam.ac.uk) P. J. C. Broadhurst (pjcb3@cam.ac.uk) I. P. L. McLaren (iplm2@cam.ac.uk)
Supplemental Material
 Supplemental Material Recording technique Multi-unit activity (MUA) was recorded from electrodes that were chronically implanted (Teflon-coated platinum-iridium wires) in the primary visual cortex representing
Supplemental Material Recording technique Multi-unit activity (MUA) was recorded from electrodes that were chronically implanted (Teflon-coated platinum-iridium wires) in the primary visual cortex representing
NO EVIDENCE FOR ANIMATE IMPLIED MOTION PROCESSING IN MACAQUE AREAS MT AND MST
 CHAPTER 4 NO EVIDENCE FOR ANIMATE IMPLIED MOTION PROCESSING IN MACAQUE AREAS MT AND MST Abstract In this study we investigated animate implied motion processing in macaque motion sensitive cortical neurons.
CHAPTER 4 NO EVIDENCE FOR ANIMATE IMPLIED MOTION PROCESSING IN MACAQUE AREAS MT AND MST Abstract In this study we investigated animate implied motion processing in macaque motion sensitive cortical neurons.
Nonlinear processing in LGN neurons
 Nonlinear processing in LGN neurons Vincent Bonin *, Valerio Mante and Matteo Carandini Smith-Kettlewell Eye Research Institute 2318 Fillmore Street San Francisco, CA 94115, USA Institute of Neuroinformatics
Nonlinear processing in LGN neurons Vincent Bonin *, Valerio Mante and Matteo Carandini Smith-Kettlewell Eye Research Institute 2318 Fillmore Street San Francisco, CA 94115, USA Institute of Neuroinformatics
M Cells. Why parallel pathways? P Cells. Where from the retina? Cortical visual processing. Announcements. Main visual pathway from retina to V1
 Announcements exam 1 this Thursday! review session: Wednesday, 5:00-6:30pm, Meliora 203 Bryce s office hours: Wednesday, 3:30-5:30pm, Gleason https://www.youtube.com/watch?v=zdw7pvgz0um M Cells M cells
Announcements exam 1 this Thursday! review session: Wednesday, 5:00-6:30pm, Meliora 203 Bryce s office hours: Wednesday, 3:30-5:30pm, Gleason https://www.youtube.com/watch?v=zdw7pvgz0um M Cells M cells
Processing of the incomplete representation of the visual world
 Processing of the incomplete representation of the visual world Andrzej W. Przybyszewski, and Tomasz M. Rutkowski Dept Psychology McGill University Montreal, Canada Schepens Eye Res Institute, Harvard
Processing of the incomplete representation of the visual world Andrzej W. Przybyszewski, and Tomasz M. Rutkowski Dept Psychology McGill University Montreal, Canada Schepens Eye Res Institute, Harvard
SUPPLEMENTARY INFORMATION. Table 1 Patient characteristics Preoperative. language testing
 Categorical Speech Representation in the Human Superior Temporal Gyrus Edward F. Chang, Jochem W. Rieger, Keith D. Johnson, Mitchel S. Berger, Nicholas M. Barbaro, Robert T. Knight SUPPLEMENTARY INFORMATION
Categorical Speech Representation in the Human Superior Temporal Gyrus Edward F. Chang, Jochem W. Rieger, Keith D. Johnson, Mitchel S. Berger, Nicholas M. Barbaro, Robert T. Knight SUPPLEMENTARY INFORMATION
Cortical Organization. Functionally, cortex is classically divided into 3 general types: 1. Primary cortex:. - receptive field:.
 Cortical Organization Functionally, cortex is classically divided into 3 general types: 1. Primary cortex:. - receptive field:. 2. Secondary cortex: located immediately adjacent to primary cortical areas,
Cortical Organization Functionally, cortex is classically divided into 3 general types: 1. Primary cortex:. - receptive field:. 2. Secondary cortex: located immediately adjacent to primary cortical areas,
Clinical and Experimental Neuropsychology. Lecture 3: Disorders of Perception
 Clinical and Experimental Neuropsychology Lecture 3: Disorders of Perception Sensation vs Perception Senses capture physical energy from environment that are converted into neural signals and elaborated/interpreted
Clinical and Experimental Neuropsychology Lecture 3: Disorders of Perception Sensation vs Perception Senses capture physical energy from environment that are converted into neural signals and elaborated/interpreted
Gaze-Dependent Visual Neurons in Area V3A of Monkey Prestriate Cortex
 The Journal of Neuroscience, April 1989, g(4): 1112-l 125 Gaze-Dependent Visual Neurons in Area V3A of Monkey Prestriate Cortex Claudio Galletti and Piero Paolo Battaglini Cattedra di Fisiologia generale
The Journal of Neuroscience, April 1989, g(4): 1112-l 125 Gaze-Dependent Visual Neurons in Area V3A of Monkey Prestriate Cortex Claudio Galletti and Piero Paolo Battaglini Cattedra di Fisiologia generale
Rules of apparent motion: The shortest-path constraint: objects will take the shortest path between flashed positions.
 Rules of apparent motion: The shortest-path constraint: objects will take the shortest path between flashed positions. The box interrupts the apparent motion. The box interrupts the apparent motion.
Rules of apparent motion: The shortest-path constraint: objects will take the shortest path between flashed positions. The box interrupts the apparent motion. The box interrupts the apparent motion.
Encoding model of temporal processing in human visual cortex
 Encoding model of temporal processing in human visual cortex Anthony Stigliani a, Brianna Jeska a, and Kalanit Grill-Spector a,b, PNAS PLUS a Department of Psychology, Stanford University, Stanford, CA
Encoding model of temporal processing in human visual cortex Anthony Stigliani a, Brianna Jeska a, and Kalanit Grill-Spector a,b, PNAS PLUS a Department of Psychology, Stanford University, Stanford, CA
Disparity- and velocity- based signals for 3D motion perception in human MT+ Bas Rokers, Lawrence K. Cormack, and Alexander C. Huk
 Disparity- and velocity- based signals for 3D motion perception in human MT+ Bas Rokers, Lawrence K. Cormack, and Alexander C. Huk Supplementary Materials fmri response (!% BOLD) ).5 CD versus STS 1 wedge
Disparity- and velocity- based signals for 3D motion perception in human MT+ Bas Rokers, Lawrence K. Cormack, and Alexander C. Huk Supplementary Materials fmri response (!% BOLD) ).5 CD versus STS 1 wedge
The detection of visual signals by macaque frontal eye field during masking
 articles The detection of visual signals by macaque frontal eye field during masking Kirk G. Thompson and Jeffrey D. Schall Vanderbilt Vision Research Center, Department of Psychology, Wilson Hall, Vanderbilt
articles The detection of visual signals by macaque frontal eye field during masking Kirk G. Thompson and Jeffrey D. Schall Vanderbilt Vision Research Center, Department of Psychology, Wilson Hall, Vanderbilt
Morton-Style Factorial Coding of Color in Primary Visual Cortex
 Morton-Style Factorial Coding of Color in Primary Visual Cortex Javier R. Movellan Institute for Neural Computation University of California San Diego La Jolla, CA 92093-0515 movellan@inc.ucsd.edu Thomas
Morton-Style Factorial Coding of Color in Primary Visual Cortex Javier R. Movellan Institute for Neural Computation University of California San Diego La Jolla, CA 92093-0515 movellan@inc.ucsd.edu Thomas
Attentional Control of Visual Perception: Cortical and Subcortical Mechanisms
 Attentional Control of Visual Perception: Cortical and Subcortical Mechanisms R. Desimone, M. Wessinger, L. Thomas, et al. Cold Spring Harb Symp Quant Biol 1990 55: 963-971 Access the most recent version
Attentional Control of Visual Perception: Cortical and Subcortical Mechanisms R. Desimone, M. Wessinger, L. Thomas, et al. Cold Spring Harb Symp Quant Biol 1990 55: 963-971 Access the most recent version
Limits to the Use of Iconic Memory
 Limits to Iconic Memory 0 Limits to the Use of Iconic Memory Ronald A. Rensink Departments of Psychology and Computer Science University of British Columbia Vancouver, BC V6T 1Z4 Canada Running Head: Limits
Limits to Iconic Memory 0 Limits to the Use of Iconic Memory Ronald A. Rensink Departments of Psychology and Computer Science University of British Columbia Vancouver, BC V6T 1Z4 Canada Running Head: Limits
Light passes through the lens, through the inner layer of ganglion cells and bipolar cells to reach the rods and cones. The retina
 The visual system Light passes through the lens, through the inner layer of ganglion cells and bipolar cells to reach the rods and cones. The retina 0.5 mm thick The retina 0.5 mm thick The photosensors
The visual system Light passes through the lens, through the inner layer of ganglion cells and bipolar cells to reach the rods and cones. The retina 0.5 mm thick The retina 0.5 mm thick The photosensors
Neural Correlates of Structure-from-Motion Perception in Macaque V1 and MT
 The Journal of Neuroscience, July 15, 2002, 22(14):6195 6207 Neural Correlates of Structure-from-Motion Perception in Macaque V1 and MT Alexander Grunewald, David C. Bradley, and Richard A. Andersen Division
The Journal of Neuroscience, July 15, 2002, 22(14):6195 6207 Neural Correlates of Structure-from-Motion Perception in Macaque V1 and MT Alexander Grunewald, David C. Bradley, and Richard A. Andersen Division
Continuous transformation learning of translation invariant representations
 Exp Brain Res (21) 24:255 27 DOI 1.17/s221-1-239- RESEARCH ARTICLE Continuous transformation learning of translation invariant representations G. Perry E. T. Rolls S. M. Stringer Received: 4 February 29
Exp Brain Res (21) 24:255 27 DOI 1.17/s221-1-239- RESEARCH ARTICLE Continuous transformation learning of translation invariant representations G. Perry E. T. Rolls S. M. Stringer Received: 4 February 29
Attention March 4, 2009
 Attention March 4, 29 John Maunsell Mitchell, JF, Sundberg, KA, Reynolds, JH (27) Differential attention-dependent response modulation across cell classes in macaque visual area V4. Neuron, 55:131 141.
Attention March 4, 29 John Maunsell Mitchell, JF, Sundberg, KA, Reynolds, JH (27) Differential attention-dependent response modulation across cell classes in macaque visual area V4. Neuron, 55:131 141.
Spectrograms (revisited)
 Spectrograms (revisited) We begin the lecture by reviewing the units of spectrograms, which I had only glossed over when I covered spectrograms at the end of lecture 19. We then relate the blocks of a
Spectrograms (revisited) We begin the lecture by reviewing the units of spectrograms, which I had only glossed over when I covered spectrograms at the end of lecture 19. We then relate the blocks of a
Biological Bases of Behavior. 6: Vision
 Biological Bases of Behavior 6: Vision Sensory Systems The brain detects events in the external environment and directs the contractions of the muscles Afferent neurons carry sensory messages to brain
Biological Bases of Behavior 6: Vision Sensory Systems The brain detects events in the external environment and directs the contractions of the muscles Afferent neurons carry sensory messages to brain
Perceptual Read-Out of Conjoined Direction and Disparity Maps in Extrastriate Area MT
 Perceptual Read-Out of Conjoined Direction and Disparity Maps in Extrastriate Area MT Gregory C. DeAngelis 1*, William T. Newsome 2 PLoS BIOLOGY 1 Department of Anatomy and Neurobiology, Washington University
Perceptual Read-Out of Conjoined Direction and Disparity Maps in Extrastriate Area MT Gregory C. DeAngelis 1*, William T. Newsome 2 PLoS BIOLOGY 1 Department of Anatomy and Neurobiology, Washington University
Supplementary Information
 Supplementary Information The neural correlates of subjective value during intertemporal choice Joseph W. Kable and Paul W. Glimcher a 10 0 b 10 0 10 1 10 1 Discount rate k 10 2 Discount rate k 10 2 10
Supplementary Information The neural correlates of subjective value during intertemporal choice Joseph W. Kable and Paul W. Glimcher a 10 0 b 10 0 10 1 10 1 Discount rate k 10 2 Discount rate k 10 2 10
Double dissociation of value computations in orbitofrontal and anterior cingulate neurons
 Supplementary Information for: Double dissociation of value computations in orbitofrontal and anterior cingulate neurons Steven W. Kennerley, Timothy E. J. Behrens & Jonathan D. Wallis Content list: Supplementary
Supplementary Information for: Double dissociation of value computations in orbitofrontal and anterior cingulate neurons Steven W. Kennerley, Timothy E. J. Behrens & Jonathan D. Wallis Content list: Supplementary
Supplementary Figure 1. Recording sites.
 Supplementary Figure 1 Recording sites. (a, b) Schematic of recording locations for mice used in the variable-reward task (a, n = 5) and the variable-expectation task (b, n = 5). RN, red nucleus. SNc,
Supplementary Figure 1 Recording sites. (a, b) Schematic of recording locations for mice used in the variable-reward task (a, n = 5) and the variable-expectation task (b, n = 5). RN, red nucleus. SNc,
Tuning properties of individual circuit components and stimulus-specificity of experience-driven changes.
 Supplementary Figure 1 Tuning properties of individual circuit components and stimulus-specificity of experience-driven changes. (a) Left, circuit schematic with the imaged component (L2/3 excitatory neurons)
Supplementary Figure 1 Tuning properties of individual circuit components and stimulus-specificity of experience-driven changes. (a) Left, circuit schematic with the imaged component (L2/3 excitatory neurons)
Vision Research 49 (2009) Contents lists available at ScienceDirect. Vision Research. journal homepage:
 Vision Research 49 (29) 29 43 Contents lists available at ScienceDirect Vision Research journal homepage: www.elsevier.com/locate/visres A framework for describing the effects of attention on visual responses
Vision Research 49 (29) 29 43 Contents lists available at ScienceDirect Vision Research journal homepage: www.elsevier.com/locate/visres A framework for describing the effects of attention on visual responses
Just One View: Invariances in Inferotemporal Cell Tuning
 Just One View: Invariances in Inferotemporal Cell Tuning Maximilian Riesenhuber Tomaso Poggio Center for Biological and Computational Learning and Department of Brain and Cognitive Sciences Massachusetts
Just One View: Invariances in Inferotemporal Cell Tuning Maximilian Riesenhuber Tomaso Poggio Center for Biological and Computational Learning and Department of Brain and Cognitive Sciences Massachusetts
